Exhibit 99.1
Brian Varnum, Chief Executive Officer Steve Martin, Chief Financial Officer January 5, 2022 NYSE American: ARMP

2 Forward Looking Statements This presentation contains “forward - looking” statements that involve risks, uncertainties and assumptions . If the risks or uncertainties materialize or the assumptions prove incorrect, our results may differ materially from those expressed or implied by such forward - looking statements . All statements other than statements of historical fact could be deemed forward - looking, including, but not limited to : the potential future of antibiotic resistance ; the ability for bacteriophage therapies to disrupt and destroy biofilms and restore sensitivity to antibiotics ; the planned development strategy, presenting data to regulatory agencies and defining planned clinical studies ; the expected timing of additional clinical trials, including Phase 1 b/Phase 2 or registrational clinical trials ; the drug product candidates to be supplied by Armata for clinical trials ; bacteriophage technology being uniquely positioned to address the global threat of antibiotic resistance ; the protection of intellectual property, including pending and issued patents ; the activities to be performed by specific parties in connection with clinical trials ; the potential use of bacteriophages to treat bacterial infections ; research and development plans ; the development of bacteriophage - based therapies ; the ability to select combinations of phages to formulate product candidates ; the ability to manufacture product candidates ; the pursuit of additional indications ; the safety and efficacy of product candidates ; collaborations with third parties and the potential markets and market opportunities for product candidates ; potential market growth ; our partnership with Merck, known as MSD outside of the United States and Canada, the Cystic Fibrosis Foundation, and U . S . Department of Defense ; our ability to achieve our vision, including improvements through engineering and success of clinical trials ; our ability to finance our operations ; Armata's ability to set up or operate R&D and manufacturing facilities ; our ability to meet anticipated milestones for 2022 ; Armata's ability to be a leader in the development of phage - based therapeutics ; the expected use of proceeds from the recent $ 15 million grant ; the expected impact of the COVID - 19 pandemic on the Company’s operations and any statements of assumptions underlying any of the items mentioned . These statements are based on estimates and information available to us at the time of this presentation and are not guarantees of future performance . Actual results could differ materially from our current expectations as a result of these risks and uncertainties, which include, without limitation, risks related to the ability of our lead clinical candidates, AP - PA 02 and AP - SA 02 (including any modifications thereto) to be more effective than previous candidates ; our ability to enhance AP - PA 02 to treat both CF and NCFB patients ; our ability to advance our preclinical and clinical programs and through the uncertain and time - consuming regulatory approval process ; our ability to develop products as expected ; our expected market opportunity for our products ; our ability to sufficiently fund our operations as expected, including obtaining additional funding as needed ; and any delays or adverse events within, or outside of, our control, caused by the recent outbreak of COVID - 19 . You should not rely upon forward - looking statements as predictions of future events . Although we believe that the expectations reflected in the forward - looking statements are reasonable, we cannot guarantee that the future results, levels of activity, performance or events and circumstances reflected in the forward - looking statements will be achieved or occur . Moreover, we undertake no obligation to update publicly any forward - looking statements for any reason to conform these statements to actual results or to changes in our expectations except as required by law . We refer you to the documents that we file from time to time with the Securities and Exchange Commission, including our registration statement, Annual Report on Form 10 - K, Quarterly Reports on Form 10 - Q and Current Reports on Form 8 - K . These documents, including the sections therein entitled “Risk Factors,” identify important factors that could cause the actual results to differ materially from those contained in forward - looking statements .

3 Investment Highlights Phage cocktails targeting acute and chronic infections ▪ P. aeruginosa product candidates for respiratory infections ▪ Cystic fibrosis: First multi - center randomized trial ▪ Non - cystic fibrosis bronchiectasis ▪ Hospitalized pneumonia ▪ S. aureus phage product candidate ▪ Complicated bacteremia ▪ Prosthetic joint infection Phage - specific GMP drug manufacturing facilities ▪ In - house manufacturing and quality systems Strong partnerships ▪ Cystic Fibrosis Foundation ($5M award; $3M equity investment), US Department of Defense ($15M award), Merck Strong board and executive leadership team ▪ Seasoned drug development team ▪ Successful track record in capital raises, M&A, and exits $52 million in equity capital and $20 million in supporting grants received since early 2020
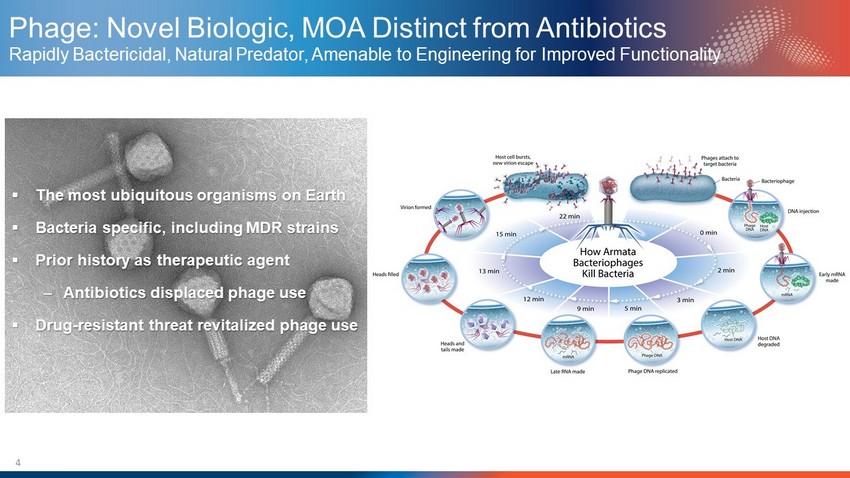
4 Phage: Novel Biologic, MOA Distinct from Antibiotics Rapidly Bactericidal, Natural Predator, Amenable to Engineering for Improved Functionality ▪ The most ubiquitous organisms on Earth ▪ Bacteria specific, including MDR strains ▪ Prior history as therapeutic agent – Antibiotics displaced phage use ▪ Drug - resistant threat revitalized phage use

5 Armata Benefits from Long History of Phage Development Pseudomonas aeruginosa ▪ >600 primary phage isolates; >230 with host range data; >270 sequenced phage ▪ >2,000 bacterial isolates Staphylococcus aureus ▪ >60 primary phage isolates; 11 phages screened for biocompatibility ▪ ~1,000 bacterial isolates Additional Sources ▪ Historical collections: C3J Therapeutics, Synthetic Genomics, Biocontrol, Special Phage Services, Novolytics ▪ R. M. Alden Research Lab Collection of >25,000 primary bacterial isolates ▪ >330 phages against diverse pathogens ( Salmonella, Klebsiella, E. coli, Acinetobacter, Enterococcus …) Comparative Genetics ▪ ~1,000 bacterial genomes sequenced representing different disease states

6 Armata’s Capabilities and Operational Overview Purposely Built for Phage Product Development, Bench to Clinic Discovery • Phage libraries • Pathogen libraries • Synthetic biologists with phage engineering expertise • Computational biology team Preclinical Development • Well - equipped BSL2 labs • Highly trained microbiologists • Experienced translational biologists • Formulation capabilities PD & Analytical Sciences • Fully equipped for all method development • Strong team of biophysical scientists (chemists, structural biologists, etc.) • Aligned with manufacturing for efficient method transfer CMC • Licensed cGMP facility operating 2 clean rooms • In - house Quality Control and Quality Systems • ~100 years combined manufacturing experience from phase 1 to commercialization Clinical Development • Successful filing of INDs and conducting of FIH studies • Execution of mid - stage studies • Conducting registrational studies and achieving approval (BLAs and INDs) • Operational expertise in US and ex - US • Support of product launch
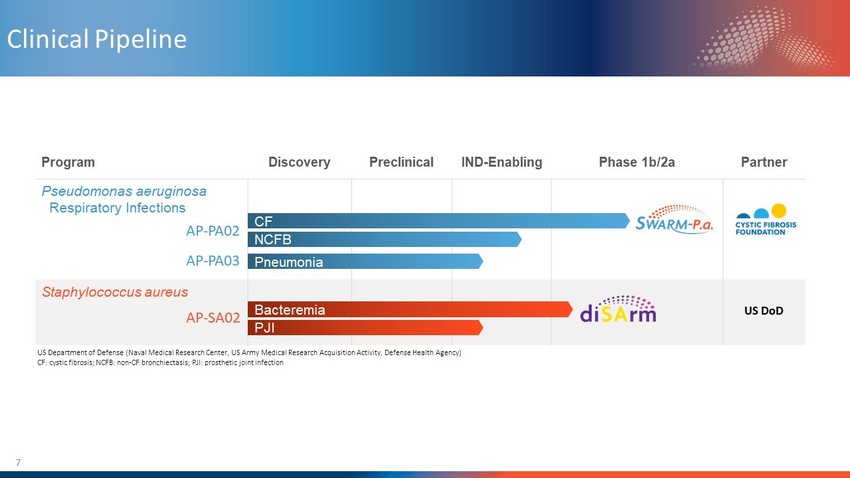
7 Clinical Pipeline Program Discovery Preclinical IND - Enabling Phase 1b/2a Partner Pseudomonas aeruginosa Respiratory Infections Staphylococcus aureus CF Bacteremia US DoD US Department of Defense (Naval Medical Research Center, US Army Medical Research Acquisition Activity, Defense Health Agency ) CF: cystic fibrosis; NCFB: non - CF bronchiectasis; PJI: prosthetic joint infection NCFB Pneumonia PJI AP - PA02 AP - PA03 AP - SA02

Cystic Fibrosis Non - CF Bronchiectasis Pneumonia Pseudomonas aeruginosa Program
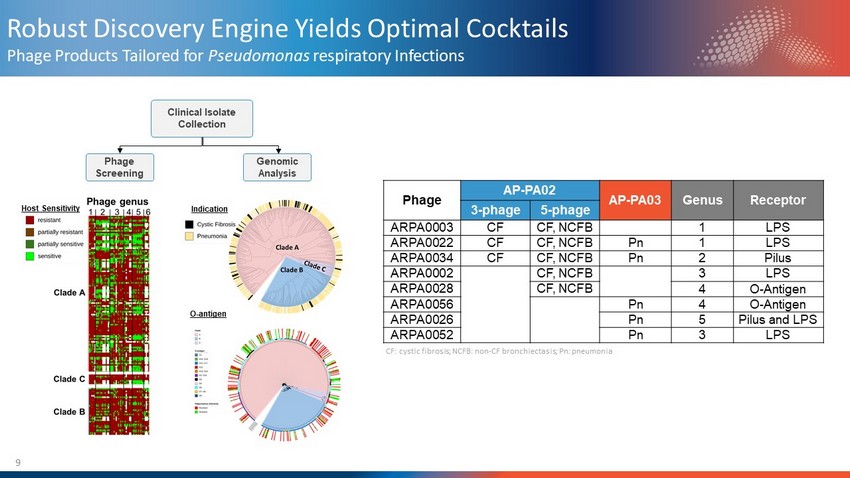
9 Robust Discovery Engine Yields Optimal Cocktails Phage Products Tailored for Pseudomonas respiratory Infections Clinical Isolate Collection Phage Screening Genomic Analysis O - antigen Indication Host Sensitivity Phage AP - PA02 AP - PA03 Genus Receptor 3 - phage 5 - phage ARPA0003 CF CF, NCFB 1 LPS ARPA0022 CF CF, NCFB Pn 1 LPS ARPA0034 CF CF, NCFB Pn 2 Pilus ARPA0002 CF, NCFB 3 LPS ARPA0028 CF, NCFB 4 O - Antigen ARPA0056 Pn 4 O - Antigen ARPA0026 Pn 5 Pilus and LPS ARPA0052 Pn 3 LPS CF: cystic fibrosis; NCFB: non - CF bronchiectasis ; Pn : pneumonia
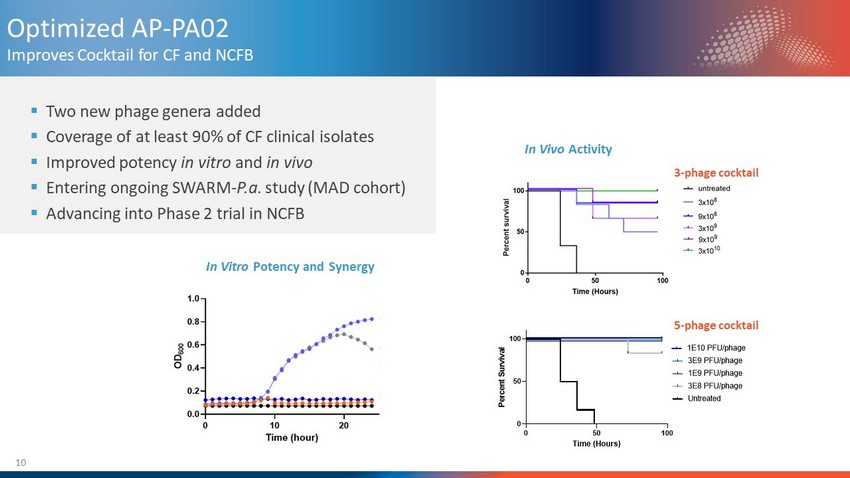
10 ▪ Two new phage genera added ▪ Coverage of at least 90% of CF clinical isolates ▪ Improved potency in vitro and in vivo ▪ Entering ongoing SWARM - P.a. study (MAD cohort) ▪ Advancing into Phase 2 trial in NCFB Optimized AP - PA02 Improves Cocktail for CF and NCFB In Vivo Activity 0 50 100 0 50 100 Time (Hours) P e r c e n t S u r v i v a l Untreated 3E8 PFU/phage 1E9 PFU/phage 3E9 PFU/phage 1E10 PFU/phage AP-PA02 (5-phage cocktail) 3 - phage cocktail 5 - phage cocktail In Vitro Potency and Synergy
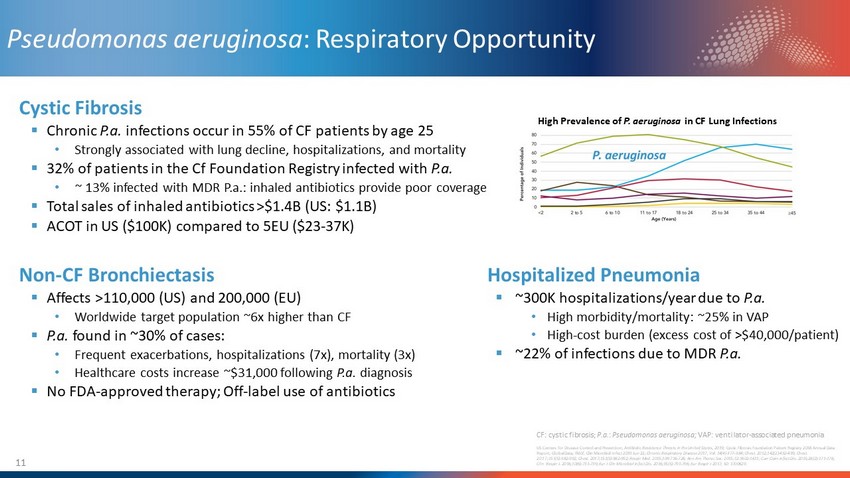
11 Pseudomonas aeruginosa : Respiratory Opportunity Cystic Fibrosis ▪ Chronic P.a. infections occur in 55% of CF patients by age 25 • Strongly associated with lung decline, hospitalizations, and mortality ▪ 32% of patients in the Cf Foundation Registry infected with P.a. • ~ 13% infected with MDR P.a.: inhaled antibiotics provide poor coverage ▪ Total sales of inhaled antibiotics >$1.4B (US: $1.1B) ▪ ACOT in US ($100K) compared to 5EU ($23 - 37K) Non - CF Bronchiectasis ▪ Affects >110,000 (US) and 200,000 (EU) • Worldwide target population ~6x higher than CF ▪ P.a. found in ~30% of cases: • Frequent exacerbations, hospitalizations (7x), mortality (3x) • Healthcare costs increase ~$31,000 following P.a. diagnosis ▪ No FDA - approved therapy; Off - label use of antibiotics High Prevalence of P. aeruginosa in CF Lung Infections US Centers for Disease Control and Prevention , Antibiotic Resistance Threats in the United State s , 2019; Cystic Fibrosis Foundation Patient Registry 2018 Annual Data Report; GlobalData; Eklöf, Clin Microbiol Infect 2019 Jun 22 ; Chronic Respiratory Disease 2017, Vol. 14(4) 377 – 384; Chest. 2012;142(2):432 - 439; Chest. 2017;151(5):982-992; Chest. 2017;151(5):982 - 992; Respir Med. 2015;109:716 - 726; Ann Am Thorac Soc. 2015;12:1602 - 1611; Curr Opin Infect Dis. 2015;28(2):171 - 176; Clin Respir J. 2016;10(6):731 - 739; Eur J Clin Microbiol Infect Dis. 2016;35(5):791 - 796; Eur Respir J 2017; 50: 1700629. Hospitalized Pneumonia ▪ ~300K hospitalizations/year due to P.a. • High morbidity/mortality: ~25% in VAP • High - cost burden (excess cost of >$40,000/patient) ▪ ~22% of infections due to MDR P.a. CF: cystic fibrosis; P.a. : Pseudomonas aeruginosa; VAP: ventilator - associated pneumonia

Staphylococcus aureus Program Complicated Bacteremia Prosthetic Joint Infection

13 Medical Opportunity Methicillin - Resistant S. aureus (MRSA): A Serious Threat 1 ▪ 323,700 new cases in hospitalized patients ▪ 10,600 deaths ▪ $1.7 billion of attributable healthcare costs First indication: S. aureus bacteremia (SAB) 2 ▪ S. aureus is the most commonly identified pathogen in hospital - and community - acquired bacteremia • 40% mortality in SAB ▪ Annually in the US there are approximately 200,000 hospitalizations for SAB • Average hospital costs to patients with nosocomial SAB ranges between $40,000 (MSSA) and $114,000 (MRSA) ▪ Complicated SAB responds poorly to SOC • While biofilms can render traditional antibiotics ineffective, phages have the ability to penetrate the biofilm S. aureus Prosthetic Joint Infection (PJI) 3 ▪ Total number of PJI - related revision surgeries is expected to grow ▪ Rise from 70,000 in 2020 to 144,000 in 2040 in the US and EU5 ▪ US is the largest market for PJI ▪ US accounts for 61% of PJI - related revision surgery in 2020 (71% by 2040) ▪ High rates of S. aureus PJI infection across all regions ▪ S. aureus accounts for up to 47% of all PJI infections across the US and EU5 ▪ Total hospital charge for PJI estimated at $150,000 1 US Centers for Disease Control and Prevention , Antibiotic Resistance Threats in the United State s , 2019. 2 Clinical Leader, Sept 6, 2018; Clin Microbiol Rev. 2012;25(2):362 - 386; ICHE. 2007;28(3):273 - 279; Rev Infect Dis. 1987;9(5):891 - 907; ICHE. 2009; 30(5):453 - 460; JAMA Intern Med. 2013;173(22):2039 - 46. 3 GlobalData PJI Market Assessment
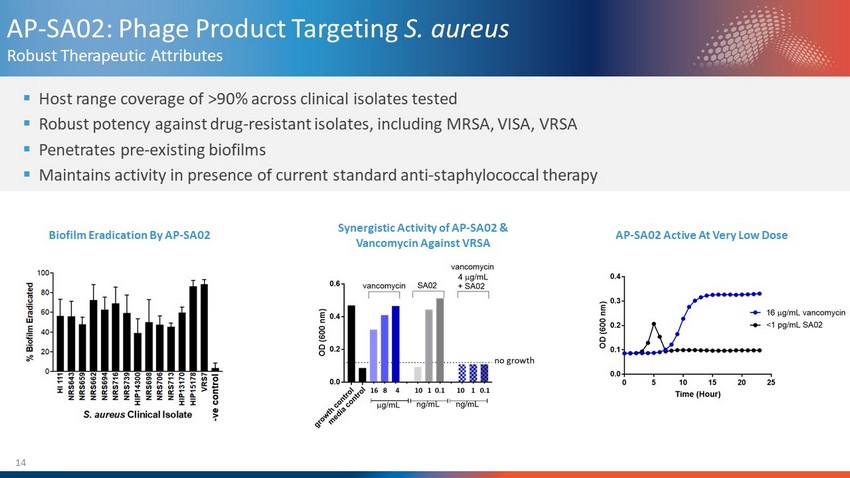
14 AP - SA02: Phage Product Targeting S. aureus Robust Therapeutic Attributes ▪ Host range coverage of >90% across clinical isolates tested ▪ Robust potency against drug - resistant isolates, including MRSA, VISA, VRSA ▪ Penetrates pre - existing biofilms ▪ Maintains activity in presence of current standard anti - staphylococcal therapy Biofilm Eradication By AP - SA02 AP - SA02 Active At Very Low Dose Synergistic Activity of AP - SA02 & Vancomycin Against VRSA

15 AP - SA02: Clinical Outline $15M in Nondilutive Funding from US DoD to Support Phase 1b/2a Bacteremia Study Phase 1b/2a Follow - on study 2022 Regulatory Filing Patient population: Complicated bacteremia stratified for MRSA Route of administration: I.V. as adjunct to best available therapy Goals: Safety and tolerability, pharmacokinetics, dose exploration, exploratory efficacy endpoints Efficacy in bacteremia Fixed dose and schedule Refined patient population Powered for rigorous demonstration of efficacy Periprosthetic joint infection (PJI)

Corporate Summary

17 Strong Global IP Position Through Pending and Issued Patents 15 Patent Families, Long - Life Patents, Patents Granted in all Major Jurisdictions Expiration dates through 2041 Armata’s patents and applications cover: ▪ Therapeutic phage cocktails (Staphylococcus and Pseudomonas) and uses thereof ▪ Synthetic phage and methods of manufacture thereof ▪ Beneficial effects of phage treatment ▪ Phage combinations for treating biofilm infections ▪ Sequential use of phages in combination with antibiotics ▪ Methods to reduce antibiotic resistance ▪ Methods to design therapeutic combination panels of phage ▪ Disinfection methods using bacteriophages ▪ Phage mutants having increased bacterial host spectra Jurisdiction Issued Pending U.S. 11 10 R.O.W. 65 45

18 Recent Accomplishments and Anticipated Milestones Pseudomonas phage respiratory programs ▪ Optimized AP - PA02 and advanced into ongoing SWARM - P.a. CF study ▪ Positioned to obtain topline SWARM - P.a. data in 2022 ▪ Obtain regulatory approval to initiate non - CF bronchiectasis Phase 2 study in 2022 ▪ Manufacture AP - PA03 and submit for regulatory approval for pneumonia Staphylococcus phage program ▪ Obtained clearance from FDA for US IND for bacteremia ▪ Initiate diSArm bacteremia study at US sites in 1H22 and enroll into Phase 1b ▪ Activate Australian and EU sites to facilitate Phase 2a enrolment of diSArm study ▪ Obtain regulatory approval for prosthetic joint infection

19 Leadership and Board of Directors Diverse Public Company Drug Development Expertise Robin Kramer Joseph Patti, PhD Sarah Schlesinger, MD Todd Patrick Todd Peterson, PhD Brian Varnum, PhD Steve Martin CFO Mina Pastagia, MD SVP, Clinical Development Duane Morris VP, Operations Management Board of Directors Brian Varnum, PhD CEO Jules Haimovitz , Chair Odysseas Kostas, MD Syntex

20 Cash Position ▪ $12.1 million at September 30, 2021 ▪ October 2021: Executed $7M private placement of common stock with two investors - Cystic Fibrosis Foundation and a subsidiary of Innoviva , Inc. ▪ Q1 2021: Executed $20M private placement of common stock and warrants with a subsidiary of Innoviva , Inc. ▪ Innoviva (NASDAQ: INVA) is a holding company receiving royalties from GSK; $1.2B mkt cap Capitalization ▪ 27.1 million common shares outstanding; no debt ▪ Trades on NYSE American exchange: ARMP Funding and Capitalization

