 2014 Financial
Guidance
Conference Call
January 7, 2014
Exhibit 99.1 |
 1
Forward-looking Statements
Note 1: The guidance in this presentation is only effective as of the date
given, January 7, 2014, and will not be updated or affirmed unless and until
the Company publicly announces updated or affirmed guidance.
Forward-looking Statements
Non-GAAP Information
Certain statements made in this presentation may constitute forward-looking statements, including,
but not limited to, statements regarding preliminary results and guidance with respect to
expected revenues, non-GAAP cash earnings per share, adjusted cash flows from
operations, organic product sales growth, integration-related activities and benefits, synergies, launches and approvals of
products, assumptions with respect to 2014 guidance, and the 2014 strategic initiatives of Valeant
Pharmaceuticals International, Inc. (the “Company”). Forward-looking statements may
be identified by the use of the words “anticipates,” “expects,” “intends,” “plans,”
“could,” “should,” “would,” “may,” “will,”
“believes,” “estimates,” “potential,” or “continue” and variations or similar expressions.
These statements are based upon the current expectations and beliefs of management and are subject to
certain risks and uncertainties that could cause actual results to differ materially from those
described in the forward-looking statements. These risks and uncertainties include, but are
not limited to, risks and uncertainties discussed in the company's most recent annual or quarterly report
filed with the Securities and Exchange Commission ("SEC") and other risks and uncertainties
detailed from time to time in the Company's filings with the SEC and the Canadian Securities
Administrators ("CSA"), which factors are incorporated herein by reference. Readers are
cautioned not to place undue reliance on any of these forward-looking statements. The Company undertakes no
obligation to update any of these forward-looking statements to reflect events or circumstances
after the date of this presentation or to reflect actual outcomes.
To supplement the financial measures prepared in accordance with generally accepted accounting
principles (GAAP), the Company uses non-GAAP financial measures that exclude certain items.
Management uses non-GAAP financial measures internally for strategic decision making,
forecasting future results and evaluating current performance. By disclosing non-GAAP financial measures,
management intends to provide investors with a meaningful, consistent comparison of the Company’s
core operating results and trends for the periods presented. Non-GAAP financial measures are
not prepared in accordance with GAAP; therefore, the information is not necessarily comparable to
other companies and should be considered as a supplement to, not a substitute for, or superior to, the
corresponding measures calculated in accordance with GAAP. The Company has provided preliminary
results and guidance with respect to cash earnings per share, adjusted cash flows from operations
and organic product growth rates, which are non-GAAP financial measures. The Company has not
provided a reconciliation of these preliminary and forward-looking non-GAAP financial
measures due to the difficulty in forecasting and quantifying the exact amount of the items excluded
from the non-GAAP financial measures that will be included in the comparable GAAP financial
measures. Reconciliations of historical non-GAAP financials can be found at www.valeant.com.
|
 2
Agenda
2013 Review –
J. Michael Pearson
2014 Priorities –
Howard Schiller
2014 Financial Guidance
Other Updates –
J. Michael Pearson
New 2014 Strategic Initiatives |
 3
Previous Q4 Guidance Unchanged
Fourth Quarter 2013 Guidance
Revenue expected to be >$2.0 billion
Cash EPS expected to be between $2.05 -
$2.10
Adjusted cash flows from operations expected to be greater than $625
million
Organic growth, unadjusted, is positive in the fourth quarter, both same
store sales and pro forma
See Note 1 regarding guidance |
 4
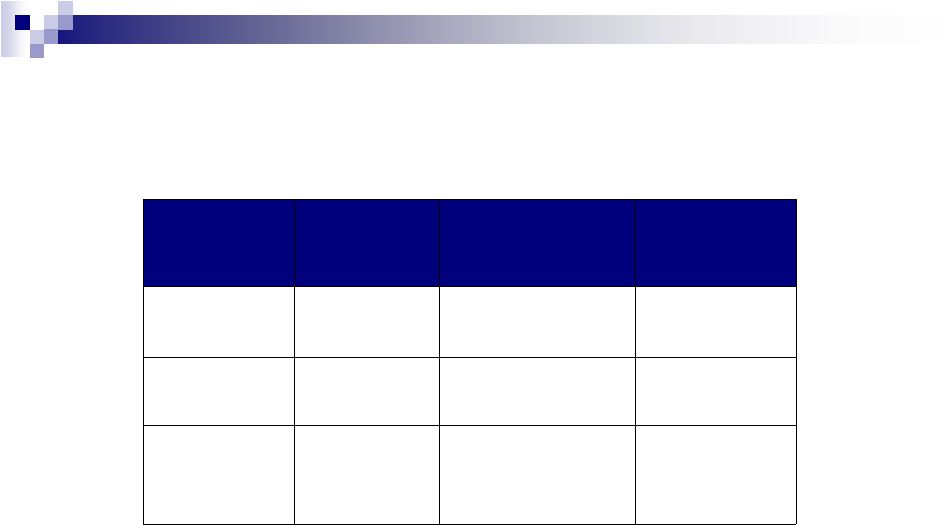
2013 Full Year Performance
See Note 1 regarding guidance
2012
As Reported
2013
Guidance
Growth
Revenue
$3.4 billion*
$5.7 -
$5.9 billion
~60% -
65%
Cash EPS
$4.14*
$6.11
-
$6.16
~35% -
37%
Adjusted Cash
Flow from
Operations
$1.2 billion*
>$1.8 billion
~50%
* Excludes $66 M from divestiture of IDP-111 generic Efudex and $45M Retigabine
Milestone. Cash EPS also excludes $.08/share one time Fx gain
|
 5
2013 Highlights
Operations:
Added
another
significant
Specialty
platform
–
Eye
Health
Achieved nearly $2.0 billion sales in Emerging Markets
Significantly expanded our aesthetic business in U.S. & Canada
MVP Program
Completed plant consolidations in Brazil, Mexico and Canada
Returned U.S. Neuro & Other to growth
Business Development:
Completed over 25 transactions
Bausch + Lomb, Euvipharm (Vietnam), Natur Produkt, Obagi,
Majority
between
1.5-3
x
sales
Established new growth platforms
China, Eye Health, Middle East, Turkey, U.S. generics, Vietnam
Added scale to a number of priority businesses
Brazil, Medical Devices, U.S. OTC, Russia, South East Asia
Acquired significant number of pipeline/launch products
Bensal, Biotrue ONEDay for Presbyopia, BV Metrogel, Luzu, Neotensil (partnership
with Living Proof), Peroxiclear, PureVision2 Multifocal for Presbyopia,
Trulign IOL, Zeus Balance Sheet Management:
Raised $10.7 billion in capital, including both debt and equity
|
 6
2013 Highlights (continued)
Strengthened Senior Management Team:
Dr.
Ari
Kellen
-
Company
Group
Chairman
Laizer
Kornwasser
-
Company
Group
Chairman
Dan
Wechsler
–
Company
Group
Chairman
Tom
Appio,
GM,
North
Asia
Muhittin
Bilgutay
–
GM
Turkey,
MENA
Andy
Chang
–
Sales
&
Marketing,
Surgical
Angelo
Conti
–
U.S.
Manufacturing
Craig
Dashefsky
–
U.S.
Commercial
Operations
Dr.
Richard
D’Souza
–
R&D,
Eye
Health
Joe
Gordon
–
U.S.
Consumer
Kym
Hampton
–
GM,
South
Africa
Deb
Jorn
–
Sales/Marketing
Rx
Derm
Jacques
Lacomb
–
EU
Manufacturing
Linda
LaGorga
-
Treasurer
Ray
Larwood
–
Sales,
Eye
Health
Pharma
Dr.
Calvin
Roberts,
CMO,
Eye
Health
Dr.
Baldo
Sforzolini
–
Medical/Clinical,
Eye
Health
Tracy
Valorie
–
Marketing,
Eye
Health
Pharma
Gaelle
Waltinger
–
GM,
Western
Europe |
 7
2013 Achievements
Average Growth
2009 -
2012
Y/Y Growth
2013
Revenue
65%
64%
Cash EPS
30%
36%
Total Shareholder Return
64%
96% |
 8
Agenda
2013 Review –
J. Michael Pearson
2014 Priorities –
Howard Schiller
2014 Financial Guidance
Other Updates –
J. Michael Pearson
New 2014 Strategic Initiatives |
 9
2014 Priorities
Continue Strong Organic Growth
All business units are expected to have positive growth in 2014
Business units expected to grow double digit in 2014
Aesthetics
Consumer
Contact Lens
Neurology & Other
Ophthalmic Rx
Oral Health
Significant sales force expansions
Aesthetics
–
94
reps
going
to
~200
Oral Health –
100 reps going to ~150
New Launches planned
Aesthetics
–
Emervel,
Neotensil,
Small
Particle
Hyaluronic
Acid
for
the
Lips
(SPHAL)
Derm Rx –
Bensal, CeraVe Baby Line, Jublia, Luzu
Eye Health –
BioTrue ONEday for Presbyopia, enVista Inserter, Peroxiclear, Victus
Fragmentation, Zeus
Branded generic launches >300
Asia
Central and Eastern Europe
Latin America
Middle East / N. Africa |
 10
2014 Expected Product Launches
Product
Business
Unit
Description
Estimated Launch Date
Bensal
Derm Rx
Topical
treatment for the inflammation and irritation
associated with many forms of dermatitis
Q1
CeraVe Baby Line
Consumer
OTC Moisturizer
Q1
Luzu
Derm Rx
Topical antifungal approved for tinea cruris, corporis, and
pedis
Q1
Neotensil
Aesthetics
Topical product that reduces the appearance of under-eye
bags within one hour
Q1
Jublia
Derm Rx
Topical antifungal for onychomycosis
2H 2014
(pending FDA approval)
enVista Inserter
Surgical
Latest enhancements
enVista inserter Jan/Feb approval
and Q1 launch
Trulign ranges
Surgical
Broader range of powers
Q2
BioTrue Oneday for
Presbyopia
Contact Lens
Daily Contact Lens
Launching regionally in 2014
Peroxiclear
Consumer
New hydrogen peroxide-based contact lens solution
Q2
Zeus
Contact Lens
Silicon Hydrogel monthly disposable contact lenses
Q2
Victus Enhancements
Surgical
Launch of multiple VICTUS enhancements
FDA corneal incision clearance
estimated 1H of 2014 / FDA lens
fragmentation estimated 2H of 2014
Hyaluronic Acid for Lips
Aesthetics
Small-particle filler
Q4 2014
Emervel
Aesthetics
Facial injectable filler line
Q4 2014/Q1 2015 |
 11
2014 Priorities (continued)
Complete Bausch + Lomb Integration
Achieve >$850 million run rate synergies by year end
Synergy capture to accelerate 2H 2014 due to heavy R&D spend 1H and European
Workers’
Council process
Maintain double digit organic growth
Launch Key Products
Continue and/or complete Bausch + Lomb R&D projects
Brimonidine Phase 3 (Eye Whitener)
Latanoprostene bunod Phase 3 (glaucoma)
Lotemax Gel Next Generation (post op inflammation)
MIM-D3 Phase 3 (Dry Eye)
Zeus Toric & Multifocal contact lens
Continue to pursue Value Added Business Development with Tuck-In
Acquisitions
Continue to be opportunistic
Focus
on
Emerging
markets
–
especially
Middle
East,
Asia
and
Russia
Continue to pursue value added Merger of Equals (MOE)
Reduce leverage ratio to less than 4x adjusted pro forma EBITDA
|
 12
2014 Guidance Assumptions
Exchange rates are based on current spot rates
Planned impact from generics to be >$200m in revenues vs. 2013 to
hurt organic growth, particularly Q1 and Q2
Atralin,
Lotemax
suspension,
Retin
A
Micro,
Vanos,
Wellbutrin
XL
(Canada),
Zovirax,
~$50m negative total revenue impact from divestitures
Includes Solta
Expected to close in January
No other acquisitions included in guidance
Jublia (efinaconazole) U.S. approval expected 2H 2014
Potential launch to be breakeven
Gross Margins expected to be in low 70’s
Cash Flow Items
Cap Ex -
~$200m
Depreciation -
~$150m
Stock Based Comp -
~$75m |
 13
2014 Guidance Assumptions (continued)
Cash EPS expected to be 40% / 60% 1H vs. 2H
Q1 expected to be lowest quarter; Q4 expected to be highest quarter
Completion of late stage R&D programs
~$200m spend expected in 1H out of $300m total 2014 R&D spend
Expected synergy capture of >$850 million run rate by year end to accelerate
2H 2014 due to heavy
R&D
spend
1H
and
European
Workers’
Council
process
Net realized annual synergies projected to be at least $650 million
Launch costs from Luzu, Neotensil, Zeus
Expansion of Oral Health and Aesthetics sales forces; revenue expected to
accelerate in 2H Cash tax rate expected <5%
Leverage reduced to <4x adjusted pro forma EBITDA by the end of
year |
 14
Financial Guidance for 2014
See Note 1 regarding guidance
2014
% over 2013
Revenue
~40%
Cash EPS
~40%
Adjusted Cash Flow from
Operations
~40%
$8.25 - $8.75
$2.4 - $2.6 billion
$8.2 - $8.6 billion |
 15
Agenda
2012 Review –
J. Michael Pearson
2014 Priorities –
Howard Schiller
2014 Financial Guidance
Other Updates –
J. Michael Pearson
New 2014 Strategic Initiatives |
 16
New 2014 Strategic Initiatives
1.
Achieve Bausch + Lomb annual run-rate synergies of greater
than $900 million.
2.
Successfully
launch
key
new
products
–
Bensal,
Jublia,
Luzu,
Neotensil, PeroxiClear, Zeus.
3.
Build out high priority businesses in Middle East, Russia/CIS,
and South East Asia.
4.
Reduce leverage ratio below 4x.
5.
Do at least one significant deal that creates substantial
shareholder value.
6. Become one of the 5 most valuable pharma companies (as
measured by Market Cap) by the end of 2016. |
 2014 Financial
Guidance
Conference Call
January 7, 2014 |