Use these links to rapidly review the document
TABLE OF CONTENTS
SCHEDULE 14A
(Rule 14a-101)
INFORMATION REQUIRED IN PROXY STATEMENT
SCHEDULE 14A INFORMATION
Proxy
Statement Pursuant to Section 14(a) of
the Securities Exchange Act of 1934 (Amendment No. )
| Filed by the Registrant ý | ||
Filed by a Party other than the Registrant o |
||
Check the appropriate box: |
||
o |
Preliminary Proxy Statement |
|
o |
Soliciting Material under Rule 14a-12 |
|
o |
Confidential, for Use of the Commission Only (as permitted by Rule 14a-6(e)(2)) |
|
ý |
Definitive Proxy Statement |
|
o |
Definitive Additional Materials |
|
| Regeneron Pharmaceuticals, Inc. | ||||
|
(Name of Registrant as Specified In Its Charter) |
||||
|
(Name of Person(s) Filing Proxy Statement, if other than the Registrant) |
||||
Payment of Filing Fee (Check the appropriate box): |
||||
ý |
No fee required. |
|||
o |
Fee computed on table below per Exchange Act Rules 14a-6(i)(1) and 0-11. |
|||
| (1) | Title of each class of securities to which transaction applies: |
|||
| (2) | Aggregate number of securities to which transaction applies: |
|||
| (3) | Per unit price or other underlying value of transaction computed pursuant to Exchange Act Rule 0-11 (set forth the amount on which the filing fee is calculated and state how it was determined): |
|||
| (4) | Proposed maximum aggregate value of transaction: |
|||
| (5) | Total fee paid: |
|||
o |
Fee paid previously with preliminary materials. |
|||
o |
Check box if any part of the fee is offset as provided by Exchange Act Rule 0-11(a)(2) and identify the filing for which the offsetting fee was paid previously. Identify the previous filing by registration statement number, or the Form or Schedule and the date of its filing. |
|||
(1) |
Amount Previously Paid: |
|||
| (2) | Form, Schedule or Registration Statement No.: |
|||
| (3) | Filing Party: |
|||
| (4) | Date Filed: |
|||
|
|
REGENERON 2016 PROXY STATEMENT AND NOTICE OF ANNUAL SHAREHOLDER MEETING |
|
|
PLEASE VIEW OUR FULL ANNUAL ONLINE INVESTOR.REGENERON.COM/2015AR |
|
|
4,000+ Regeneron employees worldwide peer-reviewed publications in 2015 68 consented individuals sequenced by the Regeneron Genetics Center 66organizations through Regeneron in the Community program 2,118 volunteer hours at 3.5 MILLION doses of EYLEA® sold globally in 2015 14% annual reduction in greenhouse gas emissions per employee Most Innovative Company, according to Forbes 4TH Ranked #1 or #2 employer in the global biopharmaceutical industry in Science Top Employers Survey 5 years in a row 5 YRS antibodies in clinical trials across multiple therapeutic areas 12 SHAREHOLDER LET TER REGENERON TODAY |
|
|
Dear Shareholders, 2015 was a busy and rewarding year for Regeneron as we made major strides in advancing our mission of bringing important new medicines to people with serious diseases, over and over again. We delivered EYLEA® (aflibercept) Injection, our therapy for patients with serious vision-threatening diseases, to more and more patients, and we launched PRALUENT® (alirocumab) Injection, a first-in-class therapy for uncontrolled LDL cholesterol in certain patients. Our pipeline of a dozen clinical-stage antibodies continues to progress, with important programs in eye disease, cancer, infectious disease, pain, cardiovascular disease and inflammation. We also continue to invest in technology and innovation that will position us to bring needed new medicines to patients for many years into the future. Likewise, we have made important infrastructure investments to ensure our long-term success, including adding two new buildings at our headquarters in Tarrytown, New York, and expanding our industrial operations facilities in Rensselaer, New York, and Limerick, Ireland. We have always run Regeneron by the principle of “doing well by doing good.” In addition to our work to invent new and needed medicines, we focus on improving our world and operating with the highest standards of integrity. This year, for the first time, our Annual Review integrates reporting on our citizenship priorities and aspirations, in addition to our financial and business performance. We invite you to read more about our business, pipeline and citizenship efforts below, and with supplemental content on our website at investor.regeneron.com/2015AR. Our 2015 Annual Report on Form 10-K is available on the Investor Relations portion of our website. MARKETED MEDICINES EYLEA® (aflibercept) Injection and Retinal Disease Programs Market-leading VEGF-Trap approved in more than 100 countries for the treatment of many blindness-causing retinal conditions, including wet age-related macular degeneration and diabetic macular edema (DME). EYLEA net sales in the U.S. increased 54 percent to $2.676 billion for the full year 2015, from $1.736 billion for the full year 2014. Outside of the U.S., where our collaborator Bayer HealthCare commercializes EYLEA, net sales were $1.413 billion in 2015, compared to $1.039 billion in 2014. Regeneron recognized $467 million from its share of net profit outside the U.S. in 2015, compared to $301 million in 2014. This growth was driven in part by the publication in early 2015 of first-year results from an independent National Institutes of Health (NIH)-sponsored comparative effectiveness study in DME. In the study, at one year, EYLEA demonstrated a significantly greater improvement in mean change in best-corrected visual acuity (BCVA) from baseline compared to ranibizumab and bevacizumab, two other VEGF inhibitors used in retinal disease. The rates of most ocular and systemic adverse events were similar across the three study groups. L-1 SHAREHOLDER LET TER |
|
|
In 2016, we initiated a Phase 3 study of EYLEA in diabetic retinopathy in patients without DME, a common degenerative eye disease that impacts people with diabetes. We continue to explore EYLEA in combination with other mechanisms, and have two ongoing clinical programs in this area in collaboration with Bayer HealthCare: aflibercept+PDGFR-beta and aflibercept+ANG2. PRALUENT® (alirocumab) Injection Only monoclonal antibody targeting PCSK9 (proprotein convertase subtilisin/kexin type 9) available in two doses, allowing for tailored therapy based on a patient’s LDL-C lowering needs. In July 2015, PRALUENT was approved by the U.S. Food and Drug Administration (FDA) as adjunct to diet and maximally tolerated statin therapy for the treatment of adults with heterozygous familial hypercholesterolemia or clinical atherosclerotic cardiovascular disease who require additional lowering of LDL-C (often referred to as “bad cholesterol”). The effect of PRALUENT on cardiovascular morbidity and mortality has not been determined. Together with our collaborator Sanofi, the U.S. launch is underway. We have focused on physician education about this new class, as well as achieving patient access and reimbursement coverage from health plans. PRALUENT was also approved in the E.U., and launches are underway across the region. The ongoing ODYSSEY OUTCOMES clinical trial program, which is evaluating the potential of PRALUENT to prevent heart attacks, stroke and cardiac death, reached full enrollment in 2015, with more than 18,000 patients at more than 2,000 study centers. Interim results are possible in late 2016, and we expect full results in 2017. L-2 |
|
|
P H A S E 2 P H A S E 3 REGN1979 CD20/CD3 Antibody Cancer REGN1908-1909 Allergic disease DUPILUMAB* IL-4R Antibody Atopic dermatitis in children, nasal polyps, eosinophilic esophagitis SARILUMAB* IL-6R Antibody Non-infectious uveitis TREVOGRUMAB GDF8 Antibody Skeletal muscle disorders REGN2176-3^ Rinucumab (PDGFR-beta Antibody) + Aflibercept Wet age-related macular degeneration EVINACUMAB Angptl3 Antibody Lipid disorders REGN910-3^ Nesvacumab (Ang2 Antibody) + Aflibercept Ophthalmology REGN2810* PD-1 Antibody Cancer FASINUMAB† NGF Antibody Chronic lower back pain ALIROCUMAB* PCSK9 Antibody Cardiovascular outcomes AFLIBERCEPT^ VEGF-Trap Diabetic retinopathy without DME SARILUMAB* IL-6R Antibody Rheumatoid arthritis DUPILUMAB* IL-4R Antibody Atopic dermatitis in adults, asthma REGN2222 RSV Antibody Respiratory syncytial virus FASINUMAB† NGF Antibody Pain due to osteoarthritis P H A S E 1 CLINICAL-STAGE PIPELINE Regeneron has a dozen fully human monoclonal antibodies in clinical development, all of which were developed using our proprietary VelocImmune® technology. (as of April 2016) L-3 |
|
|
|
|
|
|
|
|
* in collaboration with Sanofi ^ in collaboration with Bayer HealthCare † in collaboration with Mitsubishi Tanabe LATE-STAGE PIPELINE Sarilumab Anti–IL-6 monoclonal antibody under U.S. regulatory review for the treatment of rheumatoid arthritis (RA). In 2015, we reported positive data from three Phase 3 trials of sarilumab in patients with rheumatoid arthritis. Together with our collaborator Sanofi, we submitted the U.S. Biologics License Application in November 2015 and were assigned a Prescription Drug User Fee Act (PDUFA) date of October 30, 2016. In March 2016, the Phase 3 SARIL-RA-MONARCH monotherapy study met its primary endpoint by demonstrating that sarilumab was superior to adalimumab (Humira®) in improving signs and symptoms of active RA at Week 24. The incidence of adverse events, serious adverse events, infections and serious infections was generally similar between groups. Dupilumab First-in-class investigational monoclonal antibody blocking IL-4 and IL-13, two key cytokines believed to be drivers in allergic inflammation, being studied for the treatment of certain allergic conditions, including atopic dermatitis (AD), uncontrolled asthma and eosinophilic esophagitis. Dupilumab was granted a Breakthrough Therapy designation by the FDA for the treatment of adults with moderate-to-severe atopic dermatitis who are not adequately controlled with topical prescription therapy and/or for whom these treatments are not appropriate. We expect to submit an application for FDA approval later this year. In 2016, we reported positive topline results from two large Phase 3 studies in atopic dermatitis and continue to enroll patients in a second pivotal study in asthma. In the atopic dermatitis studies, the overall rate of adverse events was comparable between the dupilumab groups and the placebo groups. REGN2222 Our fully human monoclonal antibody being investigated for the prevention of serious lower respiratory tract infections associated with Respiratory Syncytial Virus (RSV). In 2015, we initiated the Phase 3 NURSERY-Pre-term trial that will evaluate the efficacy, safety, pharmacokinetics and immunogenicity of REGN2222 in infants under the age of six months. Fasinumab Our antibody targeting nerve growth factor being evaluated for potential to offer a novel, non-opioid approach to addressing chronic pain. Two clinical trials of fasinumab for pain due to osteoarthritis and chronic back pain were initiated in 2016. In 2015, we entered into a collaboration with Mitsubishi Tanabe Pharma Corporation to develop and commercialize fasinumab in Japan, Korea, and nine other Asian countries (excluding China). L-4 |
|
|
EARLY-STAGE PIPELINE AND R&D Immuno-oncology Building on our existing antibody collaboration, we launched a new $2.2B global immuno-oncology collaboration with Sanofi. This will provide important new resources to advance our portfolio in this rapidly developing field, which seeks to harness the body’s immune system to fight cancer. We continued to explore multiple approaches in immuno-oncology, including bispecific antibodies, checkpoint inhibitors and antibody drug conjugates. We have two antibodies, a CD20/CD3 bispecific antibody and a PD-1 inhibitor, in clinical studies with data expected in 2016. A number of additional immuno-oncology antibodies are expected to enter the clinic this year and next. Rapid Response & Infectious Disease Regeneron’s Rapid Response capabilities leverage our core VelociSuite® technologies to significantly compress the time required for discovery and preclinical validation of potential treatments for emerging infectious diseases. In 2015, we identified and validated a novel therapeutic cocktail of three antibodies targeting the Ebola virus, and reached an agreement with the Biomedical Advanced Research and Development Authority of the U.S. Department of Health and Human Services to develop, test and manufacture this potential treatment. A Phase 1 study in healthy volunteers is planned for the first half of 2016. We similarly identified and validated an antibody against MERS (Middle East Respiratory Virus) and are working to advance this program, as well as pursuing antibody therapies for other devastating viral diseases such as Zika and Dengue. Regeneron Genetics Center In its second full year, the Regeneron Genetics Center (RGC) continued to grow rapidly in terms of scope, scale and speed. The RGC was created to elucidate, on a large scale, genetic factors that cause or influence a range of human diseases. The team has sequenced approximately 100,000 exomes to date, and is now delivering new target opportunities and validating existing targets in our preclinical and clinical programs. We continued to bring on board world-class collaborators from industry, academia and leading health-systems, and published the RGC’s first peer-reviewed publication in the New England Journal of Medicine. GROWTH In 2015, we grew in many aspects of our business. We continued construction of our world-class 400,000-square-foot manufacturing facility in Raheen, Ireland, which will significantly expand our biologic supply capabilities for commercial products. We opened new L-5 SHAREHOLDER LET TER |
|
|
buildings in Rensselaer, our Industrial Operations headquarters, and our Tarrytown R&D laboratories and corporate headquarters. And we welcomed our 4,000th Regeneron employee, all while remaining focused on sustaining the innovative culture that makes us unique. SHAREHOLDER ENGAGEMENT One of the priorities of the Board of Directors and Company management is ensuring robust outreach and engagement with our shareholders. We value shareholder views and insights, and we believe that constructive and meaningful dialogue allows us to develop broader relationships with investors over the long-term and builds informed relationships that promote transparency and accountability. In 2015, we continued our shareholder outreach efforts and engaged in discussions with a large number of our shareholders. Please refer to the table on page 53 of our proxy statement for a summary of recent changes we have adopted based on shareholder feedback and other relevant considerations. CITIZENSHIP At Regeneron, we are committed to a better future. In addition to our work to invent new and needed medicines, we are focused on improving our world and operating with the highest standards of integrity. We are proud not only of what we do, but how we do it. Four pillars help us articulate how we view our responsibility and commitment to society: Fostering the Future of Scientific Innovation We believe Science, Technology, Engineering, and Math (STEM) education is a top priority, and are focused on ensuring a strong pipeline of STEM talent for many years to come. Our strategic programs in this area: • Attract, support and reward the best and brightest minds in science research; • Increase the effectiveness of teachers in STEM; and • Bridge STEM skills gaps and career awareness among students historically underrepresented in the sciences. Spanning from elementary school to postdoctoral fellowships, our STEM programs spark interest in science and enhance knowledge, scientific research and careers in biotechnology. One important program allows more than 200 high school and college students to participate in internship opportunities at Regeneron. In addition, we are proud to award the Regeneron Prize for Creative Innovation each year to outstanding graduate and postdoctoral students. Cultivating Sustainable Communities Regeneron employees are passionate about giving back to our communities through volunteerism, fundraising and advocacy. Regeneron In the Community (RIC), our company volunteer program, unites our people through days of service, company-sponsored activities L-6 |
|
|
and employee-led projects. RIC inspires action, fosters collaboration and motivates our people to self-organize around service projects that reflect their individual passions. We want to grow our business while reducing our environmental impact. We proactively seek environmentally responsible ways to better operate our business now and in the future, and we focus on environmental stewardship throughout our value chain. Supporting Patient Communities Our employees are focused on putting science, technology and innovation to work in order to make a difference in patients’ lives. This effort starts in the labs, moves into the clinic and continues with our commitment to ensuring patients can access the therapies they need. Nurturing our High-Engagement, High-Integrity Culture We empower our people to thrive personally and professionally, work together to create positive change and promote an ethical culture of diversity and inclusion. In 2015, we were proud to be named one of the two top employers in the global biopharmaceutical industry by Science for the fifth consecutive year, the fourth most innovative company in the world by Forbes and one of the 100 best companies to work for by Fortune. IN CLOSING Unfortunately, there was also some sadness in 2015. Our longtime friend, mentor, co-founder and Board member, Dr. Alfred G. Gilman, passed away in December. Dr. Gilman was a Nobel Laureate who made lasting contributions to science and medicine. On a personal level, we all benefited greatly from Al’s counsel and wry wit over the years, and we will miss him greatly. We look forward to updating you on our progress as we continue building Regeneron into a leading global biopharmaceutical company. Sincerely, P. Roy Vagelos, MD Leonard S. Schleifer, MD, PhD George D. Yancopoulos, MD, PhD L-7 SHAREHOLDER LET TER |
![]()
REGENERON PHARMACEUTICALS, INC.
777 Old Saw Mill River Road
Tarrytown, New York 10591-6707
NOTICE OF ANNUAL MEETING OF SHAREHOLDERS
The 2016 Annual Meeting of Shareholders of Regeneron Pharmaceuticals, Inc. (the "Company") will be held on Friday, June 10, 2016, commencing at 10:30 a.m., Eastern Time, at the Westchester Marriott Hotel, 670 White Plains Road, Tarrytown, New York, for the following purposes:
- (1)
- to
elect three Class I directors for a term of three years;
- (2)
- to
ratify the appointment of PricewaterhouseCoopers LLP as the Company's independent registered public accounting firm for the fiscal year ending
December 31, 2016; and
- (3)
- to act upon such other matters as may properly come before the meeting and any adjournment(s) or postponement(s) thereof.
The board of directors has fixed the close of business on April 14, 2016 as the record date for determining shareholders entitled to notice of, and to vote at, the Annual Meeting and at any adjournment(s) or postponement(s) thereof.
Pursuant to the rules of the Securities and Exchange Commission, we have elected to use the "Notice and Access" method of providing our proxy materials over the Internet. Accordingly, we will mail, beginning on or about April 27, 2016, a Notice of Internet Availability of Proxy Materials to our shareholders of record and beneficial owners as of the record date (other than (i) those who previously elected to access the proxy materials over the Internet, (ii) those who have previously asked to receive paper copies of the proxy materials, and (iii) shareholders who participate and hold shares of common stock in the Regeneron Pharmaceuticals, Inc. 401(k) Savings Plan). As of the date of mailing of the Notice of Internet Availability of Proxy Materials, all shareholders and beneficial owners will have the ability to access all of the proxy materials on a website referenced in the Notice of Internet Availability of Proxy Materials.
The Notice of Internet Availability of Proxy Materials also contains a toll-free telephone number, an e-mail address, and a website where shareholders can request a paper or electronic copy of the proxy statement, our 2015 annual report, and/or a form of proxy relating to the Annual Meeting. These materials are available free of charge. The Notice also contains information on how to access and vote the form of proxy.
As Authorized by the Board of Directors, |
||

|
||
| Joseph J. LaRosa Senior Vice President, General Counsel and Secretary |
April 26, 2016
REGENERON PHARMACEUTICALS, INC.
![]()
i
Table of Contents
Note Regarding Forward-Looking Statements and Non-GAAP Financial Measures
See Appendix A for important information regarding forward-looking statements and financial measures not calculated in accordance with U.S. Generally Accepted Accounting Principles contained in this proxy statement.
ii
Table of Contents
![]()
Proxy Summary
The summary below highlights information that is described in more detail elsewhere in this proxy statement. This summary does not contain all of the information you should consider, and we urge you to read the entire proxy statement carefully before voting.
General Information (see "General Information about the Meeting" on page 6 for more information)
| Date: | June 10, 2016 | |
| Time: | 10:30 a.m., Eastern Time | |
| Place: | Westchester Marriott Hotel, 670 White Plains Road, Tarrytown, New York 10591 | |
| Record Date: | April 14, 2016 |
Meeting Agenda
| Matter | Board Vote Recommendation | |||
| 1. | Election of three Class I directors for a term of three years | For each director nominee | ||
| 2. | Ratification of the appointment of PricewaterhouseCoopers LLP as the Company's independent registered public accounting firm for the fiscal year ending December 31, 2016 | For | ||
Proposal No. 1 – Our Director Nominees (see "Proposal No. 1: Election of Directors" on page 10 for more information)
The following individuals have been nominated for election at the 2016 Annual Meeting:
|
Director Class |
Name | Age* |
Director Since |
Occupation | Independent |
Committee Memberships |
||||||
| Class I | Michael S. Brown, M.D. | 75 | 1989 | Distinguished Chair in Biomedical Sciences, Regental Professor of Molecular Genetics, and Director of the Jonsson Center for Molecular Genetics, University of Texas Southwestern Medical Center at Dallas | ü |
Technology Committee (Chairman) Corporate Governance and Compliance Committee |
||||||
| Class I | Leonard S. Schleifer, M.D., Ph.D. | 63 | 1988 | President and Chief Executive Officer of Regeneron Pharmaceuticals, Inc. | | Technology Committee (Ex Officio Member) | ||||||
| Class I | George D. Yancopoulos, M.D., Ph.D. | 56 | 2001 | President, Regeneron Laboratories and Chief Scientific Officer of Regeneron Pharmaceuticals, Inc. | | Technology Committee (Ex Officio Member) | ||||||
| | | | | | | | | | | | | |
- *
- As of April 14, 2016.
1
Proxy Summary
Each director nominee is a current director and attended at least 75% of the aggregate of all 2015 meetings of the board of directors and each committee on which he served.
Corporate Governance (see "Corporate Governance" on page 15 for more information)
Regeneron is committed to good corporate governance, which we believe promotes the long-term interests of shareholders, strengthens the accountability of the board of directors and management, and helps build trust in the Company. The following chart summarizes key information regarding our corporate governance.
| Board and Other Governance Information | 2016* | |
| Size of Board | 10 | |
| Number of Independent Directors | 7 | |
| Separate Chairman and Chief Executive Officer | ü | |
| Majority Voting in the Election of Directors | ü | |
| Director Resignation Policy | ü | |
| Number of Meetings of the Board of Directors Held in 2015 | 7 | |
| Independent Directors Meet in Executive Sessions Without Management Present | ü | |
| Code of Business Conduct and Ethics Applicable to All Employees, Officers, and Directors | ü | |
| Annual Board and Committee Self-Evaluations | ü | |
| Stock Ownership Guidelines for Directors and Senior Executives | ü | |
| Active Shareholder Engagement | ü | |
| Shareholder Right to Remove Directors for Cause | ü | |
| Shareholder Right to Call Special Shareholder Meeting | ü | |
| | | |
- *
- As of April 14, 2016.
Proposal No. 2 – Ratification of PricewaterhouseCoopers LLP (see "Proposal No. 2: Ratification of Appointment of Independent Registered Public Accounting Firm" on page 33 for more information)
We ask that our shareholders ratify the appointment of PricewaterhouseCoopers LLP as the Company's independent registered public accounting firm for 2016. Below is a summary of fees related to services provided to the Company by PricewaterhouseCoopers LLP for the years ended December 31, 2015 and 2014.
|
|
2015 | | 2014 | |||
Audit Fees |
$ | 1,721,000 | $ | 1,567,493 | |||
Audit-Related Fees |
| 2,007 | | – | |||
All Other Fees |
| 4,637 | | 4,812 | |||
| | | | | | | | |
Total Fees |
$ | 1,727,644 | $ | 1,572,305 |
2015 Performance Overview (see "Executive Compensation – Compensation Discussion and Analysis – Section 1 – Summary – 2014 Performance Overview" on page 35 for more information)
2015 was another extraordinary year for Regeneron. Our key accomplishments in 2015 included:
- •
- 47% growth in EYLEA® (aflibercept) Injection global net product sales as compared to 2014;
- •
- 46% growth in our total revenues as compared to 2014;
- •
- 19% growth in non-GAAP net income as compared to 2014 (non-GAAP net income is not a measure calculated in accordance with U.S.
Generally Accepted Accounting Principles; see Appendix A for a definition of non-GAAP net income and a reconciliation of non-GAAP net income to net income);
- •
- advances in our EYLEA® franchise, including regulatory approval of EYLEA® for the treatment of visual
impairment due to macular edema secondary to retinal vein occlusion and the treatment of visual impairment secondary to myopic choroidal neovascularization in the European Union; regulatory approval
of EYLEA® for the treatment of diabetic retinopathy in patients with diabetic macular edema in the United States; and regulatory approval of EYLEA® for the treatment of retinal
vein occlusion in Japan;
- •
- regulatory approval and launch of Praluent® (alirocumab) Injection, the first drug approved by the U.S. Food and Drug
Administration ("FDA") in a new class of drugs that lower LDL ("bad") cholesterol;
- •
- positive Phase 3 data for sarilumab from three Phase 3 studies in patients with rheumatoid arthritis (SARIL-RA-TARGET, SARIL-RA-EASY, and SARIL-RA-ASCERTAIN) and submission of a Biologics License Application for sarilumab with the FDA;
2
Proxy Summary
- •
- positive pivotal Phase 2b data for dupilumab in asthma and completion of enrollment of the dupilumab atopic dermatitis
Phase 3 studies;
- •
- new collaboration agreement relating to fasinumab with Mitsubishi Tanabe Pharma Corporation for Japan, Korea, and nine other Asian
countries, excluding China;
- •
- initiation of Phase 3 clinical study of REGN2222 for Respiratory Syncytial Virus;
- •
- continued growth of our clinical development pipeline, as evidenced by the submission of one Investigational New Drug Application
with the FDA in 2015 and 13 product candidates (consisting of one Trap-based and 12 fully-human monoclonal antibody product candidates based on the Company's VelocImmune® technology) in clinical
development as of December 31, 2015;
- •
- new global strategic collaboration with Sanofi to discover, develop, and commercialize antibody-based cancer treatments in the field
of immuno-oncology; and
- •
- further important steps to support our current and future growth, including adding two new buildings in the Tarrytown campus providing nearly 300,000 square feet of additional laboratory and office space; significant progress with the construction of a new manufacturing facility in Limerick, Ireland; and increasing headcount on a year-over-year basis by approximately 47% as of December 31, 2015.
Our strong performance is reflected in the appreciation of our stock price, which increased 32%, 217%, and 1554% over the one-, three-, and five-year periods ended December 31, 2015, respectively. This shareholder return places our common stock performance in the 85th, 90th, and 99th percentile, respectively, of all NASDAQ-listed companies with a market capitalization greater than $5 billion in those periods.
Executive Compensation (see "Executive Compensation" on page 35 for more information)
We believe that the leadership of the current executive team has been instrumental to our success in 2015 and prior years, and that an executive compensation program that attracts, motivates, and helps retain key executives, including the Named Officers, is critical to our long-term success.
The main objectives of our executive compensation program are to pay for performance; closely align the interests of shareholders and management; strike a balance between short- and long-term perspectives and support our long-term growth prospects; and attract and retain highly skilled and talented executives in a competitive marketplace.
These objectives were reflected in our 2015 compensation decisions in a number of ways, including the following:
- •
- We believe in performance-based compensation and long-term incentives. In 2015, we
continued to rely primarily on performance-based compensation, both for our short-term (cash bonus) and long-term incentives (stock option awards). This emphasis on performance-based compensation
(particularly long-term incentives in the form of stock options) has been a consistent part of our philosophy since Regeneron's inception, including prior to the significant appreciation in
Regeneron's stock price that began in early 2011.
- •
- We believe that time-based stock options are inherently performance based, as they provide value to employees
only if there is future stock price appreciation and do not provide any value to employees if the stock price declines below the exercise price. As illustrated by the charts in
"Executive Compensation – Compensation Discussion and Analysis – Section 2 – Analysis of 2015 Executive Compensation Based on
Compensation Objectives," this emphasis on stock options has resulted in close alignment of our Chief Executive Officer's compensation in 2015 and over the last five years with the performance of our
common stock over those periods:
- o
- Both in 2015 and over the five-year period ended December 31, 2015, the
year-over-year increases in our Chief Executive Officer's compensation were principally attributable to the significant appreciation in our stock price, which increased the reported grant date fair
value of our Chief Executive Officer's stock option awards as determined according to the Black-Scholes model for valuing stock options.
- o
- Over the same periods, the Black-Scholes grant date fair value of stock option
grants to our Chief Executive Officer increased less than the appreciation of our stock price, in part because the absolute number of stock options granted to our Chief Executive Officer decreased in
the last three years. The number of shares underlying the annual stock option award to our Chief Executive Officer in 2015 was approximately 39% lower than in 2012, while the stock price appreciated
217% over the same period. As a result, the appreciation in the reported value of our Chief Executive Officer's pay was significantly below the appreciation of our stock price, both cumulatively over
the five-year period and on a year-over-year basis. This means that the value of our long-term shareholders' investment in Regeneron grew more rapidly than our CEO's pay over those periods.
- o
- To further illustrate this point, over the last five years, our Chief Executive Officer's total direct compensation, as a percentage of Regeneron's capitalization in the year in which the compensation was awarded, decreased from 0.20% to 0.08%.
3
Proxy Summary
- o
- As a result of our emphasis on performance-based compensation, on a relative
basis when compared to our Peer Group, the total direct compensation of our Chief Executive Officer over the last three years was also closely aligned with the performance of our common stock even
when taking into account the reported grant date fair value of our Chief Executive Officer's stock option awards as determined according to the Black-Scholes model.
- •
- We believe in year-over-year consistency in making compensation decisions and in striking a balance between
the dilutive impact of equity grants and the competitiveness of our compensation program. In our compensation decisions, we focus on the number of shares underlying equity
awards relative to the number of basic shares of common stock outstanding, rather than the grant date fair value of the award (as determined according to the Black-Scholes model). We believe this
ownership- and dilution-based approach to awarding stock options provides a better measure of the amount of potential increases in shareholder value that would be shared by the awards and allows us to
evaluate such grants on a consistent basis as compared to other companies and regardless of fluctuations in the price of Regeneron's or other companies' common stock. Further, focusing on the number
of shares and the incremental sharing rate of potential future upside (rather than targeting a specific Black-Scholes grant date fair value) avoids rewarding officers with larger grant sizes following
a decline in our stock price.
- o
- As a percentage of the total basic shares outstanding, the 2015 stock option
award to our Chief Executive Officer was significantly below the 75th percentile of the companies included in the 2015 Radford Global Life Sciences Survey and only slightly above
the 50th percentile (at 0.166% compared to 0.290% and 0.154%, respectively). In addition, this award was below the 50th percentile of our Biotech R&D Peers
(which was 0.183%).
- o
- In 2015, the Compensation Committee reduced the number of shares underlying the annual stock option awards to the Named Officers by 15% compared to 2014 (other than
- Mr. Terifay's
award, which remained at the 2014 level due to his promotion to Executive Vice President, Commercial). This decrease
constituted the third consecutive double-digit percentage decrease in the annual grant of stock options to our Named Officers, in each case following outstanding TSR performance. In reducing the size
of 2015 annual stock option awards to executives, the Compensation Committee sought to reduce the potential dilutive impact of new equity awards without adversely affecting the competitiveness of our
executive compensation program, which has successfully motivated our senior management team to deliver high operating performance and shareholder value.
- o
- We continued to pay close attention to our burn rate. Despite the expansive
growth of our employee base, which increased by 121% between 2012 and 2015 (from 1,950 full-time employees to 4,303 full-time employees), our burn rate decreased from 5.4% to 4.4% over the same
period, and we maintained a three-year burn rate average of 4.1% in 2015. We achieved this reduction through implementing three consecutive double-digit percentage decreases in the number of shares
underlying annual stock option awards, without eliminating the broad-based nature of our equity compensation program.
- o
- We believe our approach to equity compensation has helped us to successfully grow and manage employee attrition, as evidenced by our 2015 employee turnover of approximately 6%, which compares favorably to the average employee turnover of approximately 18% for the life sciences sector based on the Fourth Quarter 2015 Radford Global Life Sciences Trends Report.
Our Compensation Policies and Practices
We have compensation policies and practices designed to enhance governance of our executive compensation program and to further our compensation objectives. These policies and practices include:
| Engagement and use of an independent compensation consultant by the Compensation Committee | No "single trigger" change-in-control severance or vesting arrangements for the Named Officers | |
Stock ownership guidelines for senior executives and directors |
Policy against including excise tax gross-up provisions with respect to payments contingent upon a change in control of Regeneron in compensatory arrangements with executive officers, including the Named Officers (other than CEO employment agreement) |
|
| Transparent equity granting process and practices | Limited perquisites | |
| Policy regarding recoupment or reduction of incentive compensation that is applicable to officers, including the Named Officers | Compensation Committee and non-employee director oversight of our compensation program | |
| Prohibition against hedging and pledging of our securities by directors and employees | Prudent management of compensation-related risks |
4
Proxy Summary
| |
|
|
||
|---|---|---|---|---|
| | | | | |
2015 Shareholder Outreach |
|
|||
We have instituted an ongoing shareholder outreach program through which we seek input from our institutional investors and other shareholders regarding our executive compensation and other governance practices, and implement appropriate changes based on this input. We value shareholder views and insights and believe that constructive and meaningful dialogue allows us to develop broader relationships with investors over the long-term and builds informed relationships that promote transparency and accountability. We continued our shareholder outreach efforts in 2015 and engaged in discussions with shareholders collectively representing approximately 47% of the shares of common stock outstanding as of December 31, 2015 (excluding shares held our directors and executive officers and Sanofi). Below is a summary of recent changes we have adopted based on shareholder feedback and other relevant considerations: |
|
|||
| | ||||
| | | | | |
| What We Heard | What We Did | When Implemented | ||
| Concern about size of NEO equity awards | Implemented another double-digit percentage decrease in the number of shares underlying the annual stock option awards to our CEO, CSO, CFO, and EVP, Research & Development | December 2015 (earlier reductions implemented in December 2013 and December 2014) | ||
| Concern about burn rate | Implemented across-the-board decrease in the number of shares underlying employee annual stock option awards; maintained a three-year burn rate average of 4.1% despite a 121% increase in the number of employees over the same period | December 2015 (earlier reductions implemented in December 2013 and December 2014) | ||
| Continue to implement corporate governance best practices | Adopted majority voting standard in the election of directors | January 2016 |
5
Proxy Summary
REGENERON PHARMACEUTICALS, INC.
777 Old Saw Mill River Road
Tarrytown, New York 10591-6707
April 26, 2016
![]()
General Information about the Meeting
Where and when will the 2016 Annual Meeting be held?
The 2016 Annual Meeting of Shareholders of Regeneron Pharmaceuticals, Inc. ("Regeneron," "Company," "we," "us," and "our") is scheduled for June 10, 2016, commencing at 10:30 a.m., Eastern Time, at the Westchester Marriott Hotel, 670 White Plains Road, Tarrytown, New York 10591. If you are planning to attend the meeting, directions to this location are available on our website at http://newsroom.regeneron.com.
Why did you receive a notice in the mail regarding the Internet availability of proxy materials instead of a paper copy of the proxy materials?
The "Notice and Access" rules of the United States Securities and Exchange Commission (the "SEC") permit us to furnish proxy materials, including this proxy statement and our Annual Report on Form 10-K for the fiscal year ended December 31, 2015 filed with the SEC on February 11, 2016 (the "2015 Annual Report"), to our shareholders by providing access to such documents on the Internet instead of mailing printed copies. Most shareholders received a Notice of Internet Availability of Proxy Materials (the "Notice") and will not receive printed copies of the proxy materials unless they request them. The Notice will be mailed beginning on or about April 27, 2016. The Notice includes instructions on how you may access and review all of our proxy materials via the Internet. The Notice also includes instructions on how you may vote your shares. If you would like to receive a paper or electronic copy of our proxy materials, you should follow the instructions in the Notice for requesting such materials. Any request to receive proxy materials by mail or e-mail will remain in effect until you revoke it.
Why didn't you receive a notice in the mail about the Internet availability of the proxy materials?
Shareholders who previously elected to access the proxy materials over the Internet will not receive a notice in the mail about the Internet availability of the proxy materials. Instead,
these shareholders should have received an e-mail with links to the proxy materials and the proxy voting website. In addition, shareholders who have previously asked to receive paper copies of the proxy materials and shareholders who participate and hold shares of common stock in the Regeneron Pharmaceuticals, Inc. 401(k) Savings Plan will receive paper copies of the proxy materials.
Can you vote your shares by filling out and returning the Notice?
No. The Notice identifies the items to be voted on at the Annual Meeting, but you cannot vote by marking the Notice and returning it. The Notice provides instructions on how to vote by Internet, by requesting and returning a paper proxy card, or by submitting a ballot in person at the meeting.
Why did we send you the Notice?
We sent you the Notice regarding this proxy statement because Regeneron's board of directors is asking (technically called soliciting) holders of the Company's common stock, par value $0.001 per share ("common stock"), and Class A stock, par value $0.001 per share ("Class A stock"), to provide proxies to be voted at our 2016 Annual Meeting of Shareholders or at any adjournment(s) or postponement(s) of the meeting.
Who is entitled to vote at the Annual Meeting?
Only shareholders of record at the close of business on the record date, April 14, 2016, are entitled to vote at the Annual Meeting shares of common stock and/or Class A stock held of record on that date. As of April 14, 2016, 103,165,457 shares of common stock and 1,913,136 shares of Class A stock were issued and outstanding. The common stock and the Class A stock vote together on all matters as a single class, with the common stock being entitled to one vote per share and the Class A stock being entitled to ten votes per share.
6
General Information about the Meeting
What are you being asked to vote on?
We are asking you to vote on:
- •
- election of three Class I directors for a term of three years (Proposal No. 1); and
- •
- ratification of the appointment of PricewaterhouseCoopers LLP as the Company's independent registered public accounting firm for the fiscal year ending December 31, 2016 (Proposal No. 2).
What are the board's recommendations?
The board of directors recommends that you vote:
- •
- FOR election of each of the three nominated Class I directors (Proposal
No. 1); and
- •
- FOR ratification of the appointment of PricewaterhouseCoopers LLP as the Company's independent registered public accounting firm for 2016 (Proposal No. 2).
How can you vote?
In person. If you are a shareholder of record, you may vote in person at the Annual Meeting. The Company will give you a ballot when you arrive. If you are a beneficial owner of shares held in the name of your bank, broker, or other nominee, or in "street name," to vote in person at the Annual Meeting you must obtain from your nominee and bring to the meeting a "legal proxy" authorizing you to vote such shares held as of the record date. We recommend you vote by proxy even if you plan to attend the meeting. So long as you meet the applicable requirements, you can always change your vote at the meeting. Instructions on voting by proxy are included below.
Via the Internet. You may vote by proxy via the Internet by visiting www.proxyvote.com. You will need the 12 digit control number included on the Notice or, if you received a paper copy of the proxy materials, the proxy card or voting instruction form you received. You may vote via the Internet through 11:59 p.m., Eastern Time, on June 9, 2016.
Via telephone. If you received printed copies of the proxy materials, you may vote by proxy via telephone by calling the toll free number found on the proxy card or the voting instruction form. You will need the 12 digit control number included on the proxy card or voting instruction form. You may vote via telephone through 11:59 p.m., Eastern Time, on June 9, 2016.
By mail. If you received printed copies of the proxy materials, you may vote by proxy by completing the proxy card or voting instruction form and returning it in the envelope provided.
How are proxies voted?
If you vote by proxy in time for it to be voted at the Annual Meeting, one of the individuals named as your proxy will vote your shares as you have directed. If you submit a proxy, but no indication is given as to how to vote your shares as to a proposal, your shares will be voted in the manner recommended by the board of directors. The board of directors knows of no matter, other than those indicated above under "What are you being asked to vote on?", to be presented at the Annual Meeting. If any other matter properly comes before the Annual Meeting, the persons named and designated as proxies will vote your shares in their discretion.
What constitutes a quorum?
The presence at the Annual Meeting, in person or by proxy, of the holders as of the record date of shares of common stock and Class A stock having a majority of the voting power of all shares of common stock and Class A stock outstanding on the record date will constitute a quorum for the transaction of business at the Annual Meeting. Shares held as of the record date by holders who are present or represented by proxy at the Annual Meeting but who have abstained from voting or have not voted with respect to some or all of such shares on any proposal to be voted on at the Annual Meeting will be counted as present for purposes of establishing a quorum.
7
General Information about the Meeting
What vote is required to approve each proposal?
The following table summarizes the voting requirements applicable to the proposals to be voted on at the Annual Meeting:
| Proposal | Vote Required |
Effect of Abstentions* |
Broker Discretionary Voting Allowed?+ |
|||
| Proposal No. 1: Election of Directors | Majority of the votes cast. In accordance with our director resignation policy, an incumbent director who fails to receive the required number of votes in an uncontested election will be required to tender his or her resignation to the Chairman of the board of directors for consideration by the Corporate Governance and Compliance Committee. | No effect — not considered votes cast on this proposal | No — brokers without voting instructions will not be able to vote on this proposal | |||
Proposal No. 2: Ratification of the Appointment of PricewaterhouseCoopers LLP |
Majority of the votes cast |
No effect — not considered votes cast on this proposal |
Yes — brokers without voting instructions will have discretionary voting authority to vote |
|||
| | | | | | | |
- *
- As noted above, abstentions will be counted as present for purposes of establishing a quorum at the Annual Meeting.
- +
- Only relevant if you are the beneficial owner of shares held in "street name." If you are a shareholder of record and you do not cast your vote, no votes will be cast on your behalf on any of the items of business at the Annual Meeting.
If you are a Regeneron employee or former employee, how do you vote shares in the Company Stock Fund in your 401(k) account?
If you participate and hold shares of common stock in the Regeneron Pharmaceuticals, Inc. 401(k) Savings Plan, you may provide voting instructions to Fidelity Management Trust Company, the plan's trustee, (1) through the Internet at www.proxyvote.com by 11:59 p.m., Eastern Time, on June 7, 2016, (2) by calling 1-800-690-6903 by 11:59 p.m., Eastern Time, on June 7, 2016, or (3) by returning your completed proxy card by mail. The trustee will vote your shares in accordance with your instructions. If you do not provide timely voting instructions to the trustee, the trustee will vote your shares in the same proportion as the shares for which the trustee receives voting instructions from other participants in the plan.
Can you change your vote or revoke your proxy?
Yes. You may change your vote or revoke your proxy at any time before the proxy is exercised. If you voted by proxy electronically through the Internet or by telephone as described above, you may simply vote again at a later date using the same procedures, in which case the later submitted proxy will be recorded and the earlier vote revoked. If you submitted your proxy by mail, you must (i) file with the
Secretary of the Company, at or before the taking of the vote at the Annual Meeting, a written notice of revocation bearing a later date than the proxy you previously submitted or (ii) duly execute a later dated proxy relating to the same shares and deliver it to the Secretary of the Company or other designee before the taking of the vote at the Annual Meeting. Attendance at the Annual Meeting will not have the effect of revoking a proxy unless you give written notice of revocation to the Secretary of the Company before the proxy is exercised or you vote by written ballot at the Annual Meeting. If you hold your shares through a broker, bank, or other nominee in "street name," you will need to contact them or follow the instructions in the voting instruction form used by the firm that holds your shares to revoke your proxy.
Who solicits proxies and bears the cost of solicitation?
Solicitation of proxies may be made by mail, in person, or by telephone by officers, directors, and other employees of the Company and by employees of the Company's transfer agent, American Stock Transfer & Trust Company, LLC ("AST"), and employees of Broadridge Financial Solutions, Inc. ("Broadridge"). We will reimburse AST, Broadridge, and our banks, brokers, and other custodians, nominees, and fiduciaries for their respective reasonable costs in the preparation and mailing of proxy materials to shareholders. In addition, we
8
General Information about the Meeting
have engaged Innisfree M&A Incorporated to assist in the solicitation of proxies and provide related advice and informational support for a services fee and the reimbursement of customary disbursements that are not expected to exceed $25,000 in the aggregate. We will bear all costs of the solicitation of proxies.
Please note that cameras, other photographic equipment, or audio or video recording devices will not be permitted at the Annual Meeting.
9
General Information about the Meeting
![]()
Proposal No. 1: Election of Directors
Pursuant to the Company's Certificate of Incorporation, the board of directors is divided into three classes, denominated Class I, Class II, and Class III, with members of each class holding office for staggered three-year terms. There are currently three members in Class I and Class II and four members in Class III. The respective terms of the directors expire (in all cases, subject to the election and qualification of their successors and to their earlier death, resignation, or removal) as follows:
- •
- The terms of the Class I Directors expire at the 2016 Annual Meeting;
- •
- The terms of the Class II Directors expire at the 2017 Annual Meeting; and
- •
- The terms of the Class III Directors expire at the 2018 Annual Meeting.
The board of directors, upon the recommendation of the Corporate Governance and Compliance Committee, has nominated for election at the 2016 Annual Meeting Michael S.
Brown, M.D., Leonard S. Schleifer, M.D., Ph.D., and George D. Yancopoulos, M.D., Ph.D. as Class I Directors for a three-year term expiring at the 2019 Annual Meeting.
Biographical information is given below, as of April 14, 2016, for each nominee for Class I Director, and for each of the other directors whose term of office will continue after the 2016 Annual Meeting. All the nominees are presently directors and were previously elected by the shareholders. None of the corporations or other organizations referred to below with which a director has been or is currently employed or otherwise associated is a parent, subsidiary, or affiliate of the Company.
The board of directors unanimously recommends a vote FOR the election of Michael S. Brown, M.D., Leonard S. Schleifer, M.D., Ph.D., and George D. Yancopoulos, M.D., Ph.D. as Class I Directors for a three-year term expiring at the 2019 Annual Meeting.
The table below summarizes key qualifications, skills, or attributes most relevant to the decision to nominate the director to serve on the board of directors. A mark indicates a specific area of focus or expertise on which the board of directors relies most. The lack of a mark does not mean the director does not possess that qualification or skill. Each director biography below describes these qualifications and relevant experience in more detail. We believe the table below demonstrates the breadth and diversity of the collective experience, expertise, and skills of our board of directors.
Experience, Expertise, or Attribute |
Charles A. Baker |
Michael S. Brown, M.D. |
Joseph L. Goldstein, M.D. |
Christine A. Poon |
Arthur F. Ryan |
Leonard S. Schleifer, M.D., Ph.D. |
George L. Sing |
Marc Tessier- Lavigne, Ph.D. |
P. Roy Vagelos, M.D. |
George D. Yancopoulos, M.D., Ph.D. |
||||||||||
Industry Experience |
· | · | · | · | · | · | · | · | · | · | ||||||||||
Executive/Leadership Experience |
· | · | · | · | · | · | · | · | · | · | ||||||||||
Science/Biotech Background |
· | · | · | · | | · | · | · | · | · | ||||||||||
Research/Academic Experience |
| · | · | · | | · | | · | · | · | ||||||||||
Business Strategy/Operations Experience |
· | | | · | · | · | · | · | · | · | ||||||||||
Financial Expertise |
· | | | · | · | · | · | | · | | ||||||||||
Public Company CEO Experience |
· | | | | · | · | | | · | | ||||||||||
National Academy of Sciences Membership |
| · | · | | | | | · | · | · | ||||||||||
| | | | | | | | | | | | | | | | | | | | | |
10
Proposal No. 1: Election of Directors
Nominees for Class I Directors for Election at the 2016 Annual Meeting |
| Michael S. Brown, M.D. | ||||
 Director since: 1991
Director since: 1991Age: 75 Independent |
MICHAEL S. BROWN, M.D., 75, has been a Director of the Company since June 1991. Dr. Brown holds the Distinguished Chair in Biomedical Sciences, a position he has held since 1989, is a Regental Professor of Molecular Genetics and Internal Medicine, and the Director of the Jonsson Center for Molecular Genetics, at The University of Texas Southwestern Medical Center at Dallas, positions he has held since 1985. Drs. Brown and Goldstein jointly received the Nobel Prize for Physiology or Medicine in 1985 and the U.S. National Medal of Science in 1988. Dr. Brown is a member of the National Academy of Sciences, the National Academy of Medicine, and Foreign Member of the Royal Society (London). Dr. Brown retired as a member of the board of directors of Pfizer Inc. in 2012. Dr. Brown's distinguished scientific and academic background, including his receipt of the Nobel Prize for Physiology or Medicine in 1985, and his significant industry experience gained through his service on the board of directors of the Company and of a leading pharmaceutical company, led to the board to conclude that Dr. Brown should serve as a director. | |||
| Leonard S. Schleifer, M.D., Ph.D. | ||||
 Director since: 1988
Director since: 1988Age: 63 |
LEONARD S. SCHLEIFER, M.D., Ph.D., 63, co-founded the Company in 1988, has been a Director and its President and Chief Executive Officer since its inception, and served as Chairman of the Board from 1990 through 1994. Dr. Schleifer is a licensed physician and is certified in Neurology by the American Board of Psychiatry and Neurology. With more than 25 years of experience as Chief Executive Officer of the Company, Dr. Schleifer brings to the board an incomparable knowledge of the Company, significant leadership experience, and an in-depth understanding of the complex research, drug development, and business issues facing companies in the biopharmaceutical industry. Dr. Schleifer's significant industry and leadership experience, as well as his extensive knowledge of the Company, led the board to conclude that Dr. Schleifer should serve as a director. | |||
| George D. Yancopoulos, M.D., Ph.D. | ||||
 Director since: 2001
Director since: 2001Age: 56 |
GEORGE D. YANCOPOULOS, M.D., Ph.D., 56, joined the Company in 1989 as its Founding Scientist and is currently President, Regeneron Laboratories and Chief Scientific Officer. While holding leadership positions, Dr. Yancopoulos headed the Company's laboratories and science organization since joining the Company and, in 1998, was named the Company's first Chief Scientific Officer. Dr. Yancopoulos joined the board in 2001. He received his M.D. and Ph.D. from Columbia University. Dr. Yancopoulos was the 11th most highly cited scientist in the world in the 1990s, and in 2004 he was elected to be a member of the National Academy of Sciences. Dr. Yancopoulos, together with key members of his team, is a principal inventor and developer of the four FDA-approved drugs the Company has developed, EYLEA® (aflibercept) Injection, Praluent® (alirocumab) Injection, ZALTRAP® (ziv-aflibercept) Injection for Intravenous Infusion, and ARCALYST® (rilonacept) Injection for Subcutaneous Use, as well as of its foundation technologies, including the TRAP technology, VelociGene®, and VelocImmune®. As one of the few members of the National Academy of Sciences from industry and as an author of a substantial number of scientific publications, Dr. Yancopoulos has a distinguished record of scientific expertise. Dr. Yancopoulos also brings to the board his experience in leading and managing a complex research and development organization and his in-depth knowledge of the Company's technologies and research and development programs. Dr. Yancopoulos's significant industry and scientific experience, as well as his extensive knowledge of the Company, led the board to conclude that Dr. Yancopoulos should serve as a director. |
11
Proposal No. 1: Election of Directors
Class II Directors Continuing in Office |
| Joseph L. Goldstein, M.D. | ||||
 Director since: 1991
Director since: 1991Age: 75 Independent |
JOSEPH L. GOLDSTEIN, M.D., 75, has been a Director of the Company since June 1991. Dr. Goldstein has been a Professor of Molecular Genetics and Internal Medicine and the Chairman of the Department of Molecular Genetics at The University of Texas Southwestern Medical Center at Dallas since 1977. Dr. Goldstein is a member of the National Academy of Sciences, the National Academy of Medicine, and the Royal Society (London). He also serves on the Boards of Trustees of The Rockefeller University and the Howard Hughes Medical Institute. Drs. Goldstein and Brown jointly received the Nobel Prize for Physiology or Medicine in 1985 and the U.S. National Medal of Science in 1988. Dr. Goldstein's extensive research experience, his distinguished scientific and academic credentials, including his receipt of the Nobel Prize for Physiology or Medicine in 1985, and his substantial understanding of the Company gained through his service as a director since 1991, led to the board's decision to nominate Dr. Goldstein for reelection to the board. | |||
| Christine A. Poon | ||||
 Director since: 2010
Director since: 2010Age: 63 Independent |
CHRISTINE A. POON, 63, has been a director of the Company since November 2010. Ms. Poon is an Executive-in-Residence in the Department of Management and Human Resources at The Max M. Fisher College of Business at The Ohio State University, where she served as Dean and the John W. Berry, Sr. Chair in Business from 2009 to 2014. Prior to joining Fisher, Ms. Poon spent eight years at Johnson & Johnson, most recently as vice chairman and worldwide chairman of pharmaceuticals. At Johnson & Johnson, she served on the company's board of directors and executive committee and was responsible for managing the pharmaceutical businesses of the company. Prior to joining Johnson & Johnson, Ms. Poon spent 15 years at Bristol-Myers Squibb Company, a global pharmaceutical company, where she held senior leadership positions including president of international medicines and president of medical devices. Ms. Poon serves on the boards of directors of Prudential Financial, Inc. and The Sherwin-Williams Company and the Supervisory Board of Royal Philips Electronics. Ms. Poon's extensive expertise in domestic and international business operations, including sales and marketing and commercial operations, and her deep strategic and operational knowledge of the pharmaceutical industry, led to the board's decision to nominate Ms. Poon for reelection to the board. | |||
| P. Roy Vagelos, M.D. | ||||
 Director since: 1995
Director since: 1995Age: 86 |
P. ROY VAGELOS, M.D., 86, has been Chairman of the Board of the Company since January 1995. Prior to joining Regeneron, Dr. Vagelos was Chairman of the Board and Chief Executive Officer of Merck & Co., Inc., a global pharmaceutical company. He joined Merck in 1975, became a director in 1984, President and Chief Executive Officer in 1985, and Chairman in 1986. Dr. Vagelos retired from all positions with Merck in 1994. Dr. Vagelos served on the board of directors of Theravance, Inc. through April 2010. Dr. Vagelos is a member of the National Academy of Sciences. During his tenure as Chairman of the Company and previously as Chairman and Chief Executive Officer of Merck, Dr. Vagelos developed an extensive understanding of the complex business, operational, scientific, regulatory, and commercial issues facing the pharmaceutical industry. Dr. Vagelos's tenure and experience with the Company and Merck, his extensive knowledge of the pharmaceutical industry, his substantial leadership experience, and his significant understanding of the Company led to the board's decision to nominate Dr. Vagelos for reelection to the board. |
12
Proposal No. 1: Election of Directors
Class III Directors Continuing in Office |
| Charles A. Baker | ||||
 Director since: 1989
Director since: 1989Age: 83 Independent |
CHARLES A. BAKER, 83, has been a Director of the Company since February 1989. In September 2000, Mr. Baker retired as Chairman, President, and Chief Executive Officer of The Liposome Company, Inc., a biopharmaceutical company, a position he had held since December 1989. During his career, Mr. Baker served in a senior management capacity in various other pharmaceutical companies, including tenures as Group Vice President, Squibb Corporation (now Bristol-Myers Squibb Company) and President, Squibb International, and various senior executive positions at Abbott Laboratories and Pfizer Inc. From 1994 to 2013, Mr. Baker served as a member of the board of directors of Progenics Pharmaceuticals, Inc., a biopharmaceutical company. Mr. Baker's substantial commercial experience gained from leadership roles at biopharmaceutical and pharmaceutical companies, his extensive industry knowledge, his having overseen the approval, manufacture, and marketing of pharmaceutical products throughout the world and having led a biotechnology company to sustained profitability, and his significant understanding of the Company led the board to conclude that Mr. Baker should serve as a director. | |||
| Arthur F. Ryan | ||||
 Director since: 2003
Director since: 2003Age: 73 Independent |
ARTHUR F. RYAN, 73, has been a Director of the Company since January 2003. In 2008, Mr. Ryan retired as the Chairman of the Board of Prudential Financial, Inc., one of the largest diversified financial institutions in the world. He served as Chief Executive Officer of Prudential until December 2007. Prior to joining Prudential in December 1994, Mr. Ryan served as President and Chief Operating Officer of Chase Manhattan Bank since 1990. Mr. Ryan managed Chase's worldwide retail bank between 1984 and 1990. From 2008 to 2013, Mr. Ryan served as a non-executive director of the Royal Bank of Scotland Group plc. Since April 2009, Mr. Ryan has served as a director of Citizens Financial Group, Inc., a retail bank holding company that became publicly traded in September 2014, and currently serves as its lead director, chair of the Compensation and Human Resources Committee, and a member of the Nominating and Corporate Governance Committee. Mr. Ryan's substantial leadership experience as a chief executive officer of leading companies in the banking and insurance industries, and his extensive business experience and financial expertise, led the board to conclude that Mr. Ryan should serve as a director. | |||
| George L. Sing | ||||
 Director since: 1988
Director since: 1988Age: 66 Independent |
GEORGE L. SING, 66, has been a Director of the Company since January 1988. Since 1998, he has been a Managing Director of Lancet Capital, a venture capital investment firm in the healthcare field. From January 2004 to April 2015, Mr. Sing served as Chief Executive Officer of Stemnion, Inc., a biomedical company in the regenerative medicine field. Mr. Sing's extensive healthcare and financial expertise as a healthcare venture capital investor and biomedical company chief executive officer, his executive leadership experience, and his substantial knowledge of the Company led the board to conclude that Mr. Sing should serve as a director. | |||
13
Proposal No. 1: Election of Directors
| Marc Tessier-Lavigne, Ph.D. | ||||
 Director since: 2011
Director since: 2011Age: 56 Independent |
MARC TESSIER-LAVIGNE, Ph.D., 56, has been a Director of the Company since November 2011. Dr. Tessier-Lavigne has been the President of The Rockefeller University since March 2011 and is a Carson Family Professor and head of the Laboratory of Brain Development at The Rockefeller University. In February 2016, he was appointed the President of Stanford University effective September 1, 2016. Previously, he served as Executive Vice President and Chief Scientific Officer at Genentech, Inc., which he joined in 2003. He was a professor at Stanford University from 2001 to 2003 and at the University of California, San Francisco from 1991 to 2001. Dr. Tessier-Lavigne is a member of the National Academy of Sciences, the National Academy of Medicine, and a fellow of the Royal Societies of the United Kingdom and Canada. Dr. Tessier-Lavigne is a member of the Board of Directors of Agios Pharmaceuticals, Inc. and Juno Therapeutics, Inc., and previously served on the board of directors of Pfizer Inc. Dr. Tessier-Lavigne's distinguished scientific and academic background, and his significant industry experience, including experience in senior scientific leadership roles at a leading biopharmaceutical company, led the board to conclude that Dr. Tessier-Lavigne should serve as a director. |
14
Proposal No. 1: Election of Directors
![]()
Regeneron is committed to good corporate governance, which we believe promotes the long-term interests of shareholders, strengthens the accountability of the board of directors
and management, and helps build trust in the Company. The following chart summarizes key information regarding our corporate governance.
Board and Other Governance Information |
2016* | |
Size of Board |
10 | |
Number of Independent Directors |
7 |
|
Separate Chairman and Chief Executive Officer |
ü |
|
Majority Voting in the Election of Directors |
ü |
|
Director Resignation Policy |
ü |
|
Number of Meetings of the Board of Directors Held in 2015 |
7 |
|
Independent Directors Meet in Executive Sessions Without Management Present |
ü |
|
Code of Business Conduct and Ethics Applicable to All Employees, Officers, and Directors |
ü |
|
Annual Board and Committee Self-Evaluations |
ü |
|
Stock Ownership Guidelines for Directors and Senior Executives |
ü |
|
Active Shareholder Engagement |
ü |
|
Shareholder Right to Remove Directors for Cause |
ü |
|
Shareholder Right to Call Special Shareholder Meeting |
ü |
|
| | | |
- *
- As of April 14, 2016.
Procedures Relating to Nominees
The Corporate Governance and Compliance Committee will consider a nominee for election to the board of directors recommended by a shareholder of record if the shareholder submits the nomination in compliance with the requirements of our by-laws and the Guidelines Regarding Director Nominations, which are available on our website at www.regeneron.com under the "Corporate Governance" heading on the "Investors & Media" page.
In considering potential candidates for the board of directors, the Corporate Governance and Compliance Committee considers factors such as whether or not a potential candidate: (1) possesses relevant expertise; (2) brings skills and experience complementary to those of the other members of the board; (3) has sufficient time to devote to the affairs of the Company; (4) has demonstrated excellence in his or her field; (5) has the ability to exercise sound business judgment; (6) has the commitment to rigorously represent the long-term interests of the Company's shareholders; (7) possesses a diverse background and experience, including with respect to race, age, and gender; and (8) such other factors as the Corporate Governance and Compliance Committee may determine from time to time.
Candidates for director are reviewed in the context of the current composition of the board of directors, the operating requirements of the Company, and the long-term interests of
shareholders. In conducting the assessment, the Committee considers the individual's independence, experience, skills, background, and diversity, including with respect to race, age, and gender, along with such other factors as it deems appropriate, given the current needs of the board and the Company to maintain a balance of knowledge, experience, and capabilities. When recommending a slate of director nominees each year, the Corporate Governance and Compliance Committee reviews the current composition of the board of directors in order to recommend a slate of directors who, with the continuing directors, will provide the board with the requisite diversity of skills, expertise, experience, and viewpoints necessary to effectively fulfill its duties and responsibilities.
In the case of an incumbent director whose term of office is set to expire, the Corporate Governance and Compliance Committee reviews such director's overall service to the Company during the director's term and also considers the director's interest in continuing as a member of the board. In the case of a new director candidate, the Corporate Governance and Compliance Committee also reviews whether the nominee is "independent," based on our Corporate Governance Guidelines, applicable listing standards of the NASDAQ Stock Market LLC, and applicable SEC and other relevant rules and regulations, if necessary.
The Corporate Governance and Compliance Committee may employ a variety of methods for identifying and evaluating
15
Corporate Governance
nominees for the board of directors. The Corporate Governance and Compliance Committee may consider candidates recommended by other directors, management, search firms, shareholders, or other sources. When conducting searches for new directors, the Corporate Governance and Compliance Committee will take reasonable steps to include diverse candidates in the pool of nominees and any search firm will affirmatively be instructed to seek to include diverse candidates. Candidates recommended by shareholders will be evaluated on the same basis as candidates recommended by our directors or management or by third party search firms or other sources. Candidates may be evaluated at regular or special meetings of the Corporate Governance and Compliance Committee.
Shareholder Rights to Remove Directors for Cause and to Call Special Shareholder Meeting
Regeneron's charter documents give shareholders the rights to (i) remove directors for cause by an affirmative vote of at least 80% of the outstanding shares of all classes of capital stock entitled to vote for directors; and (ii) call a special shareholder meeting upon the written request of at least 25% of the total number of votes entitled to be cast by shareholders.
Shareholder Communications with Directors
The Company has established a process for shareholders to send communications to the members of the board of directors. Shareholders may send such communications by mail addressed to the full board, a specific member or members of the board, or a particular committee of the board, at 777 Old Saw Mill River Road, Tarrytown, New York 10591-6707,
Attention: Corporate Secretary. All such communications will be opened by our Corporate Secretary for the sole purpose of determining whether the contents represent a message to our directors. Any contents that are not in the nature of advertising, promotions of a product or service, or patently offensive material will be forwarded promptly to the addressee. In the case of communications to the board or any individual director or group or committee of directors, the Corporate Secretary will make sufficient copies of the contents to send to such director or each director who is a member of the group or committee to which the envelope is addressed.
The board has a standing Audit Committee established in accordance with Section 3(a)(58)(A) of the Securities Exchange Act of 1934, as amended (the "Exchange Act"), a Compensation Committee, and a Corporate Governance and Compliance Committee, each of which is comprised entirely of independent directors. The Corporate Governance and Compliance Committee is responsible for reviewing and recommending for the board's selection candidates to serve on our board of directors and for overseeing all aspects of the Company's compliance program other than financial compliance. The board also has a standing Technology Committee. The board has adopted charters for the Audit Committee, Compensation Committee, Corporate Governance and Compliance Committee, and Technology Committee, current copies of which are available on our website at www.regeneron.com under the "Corporate Governance" heading on the "Investors & Media" page.
16
Corporate Governance
We show below information on the membership, key functions, and number of meetings of each board committee during 2015.
|
Name of Committee and Members |
Key Functions of the Committee |
Number of Meetings Held in 2015 |
||||
| AUDIT | ||||||
George L. Sing, Chairman |
• Select the independent registered public accounting firm, review and approve its engagement letter, and monitor its independence and performance. • Review the overall scope and plans for the annual audit by the independent registered public accounting firm. • Approve performance of non-audit services by the independent registered public accounting firm and evaluate the performance and independence of the independent registered public accounting firm. • Review and approve the Company's periodic financial statements and the results of the year-end audit. • Review and discuss the adequacy and effectiveness of the Company's accounting and internal control policies and procedures. • Evaluate the internal audit process for establishing the annual audit plan; review and approve the appointment and replacement of the Company's Chief Audit Executive, if applicable, and any outside entities providing internal audit services and evaluate their performance on an annual basis. • Review the independent registered public accounting firm's recommendations concerning the Company's financial practices and procedures. • Establish procedures for the receipt, retention, and treatment of complaints received by the Company regarding accounting, internal accounting controls, or auditing matters and for the confidential, anonymous submission by employees of concerns regarding questionable accounting or auditing matters. • Review and approve any related person transaction. • Prepare an annual report of the Audit Committee for inclusion in the proxy statement and annually evaluate the Audit Committee Charter. • Oversee the Company's risk management program. • Discuss with management the Company's major financial risk exposures and the steps management has taken to monitor and control such exposures. |
10 |
17
Corporate Governance
|
Name of Committee and Members |
Key Functions of the Committee |
Number of Meetings Held in 2015 |
||||
| COMPENSATION | ||||||
Marc Tessier-Lavigne, Ph.D., Chairman
+ |
• Evaluate the performance of the Chief Executive Officer and other executive officers of the Company. • Approve the total compensation budget for all Company employees. • Oversee the Company's compensation and benefit philosophy and programs generally. • Review and approve annually the corporate goals and objectives applicable to the compensation of the Chief Executive Officer and the goals and objectives of the Company's executive compensation programs. • Prepare an annual report of the Compensation Committee for inclusion in the proxy statement. • Review and approve the Compensation Discussion and Analysis to be included in the Company's proxy statement. |
9 |
||||
CORPORATE GOVERNANCE AND COMPLIANCE |
|
|||||
Alfred G. Gilman, M.D., Ph.D., Chairman (until his resignation as Chairman on June 12, 2015, |
• Identify qualified individuals to become members of the board and recommend such candidates to the board. • Assess the functioning of the board and its committees and make recommendations to the board concerning the appropriate size, function, and needs of the board. • Make recommendations to the board regarding non-employee director compensation. • Make recommendations to the board regarding corporate governance matters and practices. • Oversee all aspects of the Company's comprehensive compliance program other than financial compliance. |
7 |
||||
TECHNOLOGY |
||||||
Michael S. Brown, M.D., Chairman |
• Review and evaluate the Company's research and clinical development programs, plans, and policies. |
2 |
||||
| | | | | | | |
- +
- Dr. Tessier-Lavigne resigned as Chairman and a member of the Compensation Committee effective as of April 1, 2016. Ms. Poon was appointed as Chairperson of the Compensation Committee effective as of Dr. Tessier-Lavigne's resignation date.
- *
- Ex Officio Member.
The board of directors has adopted a code of business conduct and ethics that applies to all of our employees, officers, and directors. You can find links to this code on our website at www.regeneron.com under the "Corporate Governance" heading on the "Investors & Media" page. We may satisfy the disclosure requirement under Item 5.05 of Form 8-K regarding
an amendment to, or a waiver from, a provision of our code of business conduct and ethics that applies to our principal executive officer, principal financial officer, principal accounting officer, or controller, or persons performing similar functions, by posting such information on our website where it is accessible through the same link noted above.
18
Corporate Governance
The board of directors has determined that each of the following currently serving directors is independent as defined in the listing standards of The NASDAQ Stock Market LLC and our Corporate Governance Guidelines: Charles A. Baker, Michael S. Brown, M.D., Joseph L. Goldstein, M.D., Christine A. Poon, Arthur F. Ryan, George L. Sing, and Marc Tessier-Lavigne, Ph.D. These individuals are affiliated with numerous educational institutions, hospitals, charities and corporations, as well as civic organizations and professional associations. The board of directors considered each of these relationships and determined that none of these relationships conflicted with the interests of the Company or would impair their independence or judgment. The board conducts executive sessions of independent directors following each regularly scheduled board meeting.
The board of directors has determined that each of the current members of the Audit Committee, Messrs. Baker, Ryan, and Sing, qualifies as an "audit committee financial expert" as that term is defined in Item 407(d)(5)(ii) of Regulation S-K under the Exchange Act, meets the required standards for independence set forth in Rule 10A-3(b)(1) under the Exchange Act, and is independent as defined for audit committee members in the listing standards of The NASDAQ Stock Market LLC.
In addition, the board of directors has determined that each of the current members of the Compensation Committee, Ms. Poon, Messrs. Baker and Sing, and Dr. Goldstein, meets the additional independence criteria applicable to compensation committee members under the listing standards of The NASDAQ Stock Market LLC and qualifies as a "Non-Employee Director" pursuant to Rule 16b-3 under the Exchange Act and as an "outside director" within the meaning of Section 162(m) of the Internal Revenue Code.
Board Leadership and Role in Risk Oversight
The board of directors recognizes that one of its key responsibilities is to establish and evaluate an appropriate leadership structure for the board so as to provide effective oversight of management. Since 1995, the board has separated the roles of the Chief Executive Officer and the Chairman of the Board, with Dr. Vagelos serving as Chairman and Dr. Schleifer serving as President and Chief Executive Officer. Dr. Vagelos's extensive leadership experience, his business acumen, and his deep understanding of the healthcare industry have made him an invaluable resource to both the board and Dr. Schleifer. The board has determined that this leadership structure is appropriate for the Company at this time.
The board executes its oversight responsibility for risk management directly and through its Committees, as follows:
- •
- The Audit Committee oversees the Company's risk management program. The Company's Chief Audit Executive,
who reports independently to the Committee, facilitates the risk management program. Audit Committee meetings include discussions of specific risk areas throughout the year, as well as annual reports from the Chief Audit Executive on the Company's enterprise risk profile.
- •
- The Compensation, Corporate Governance and Compliance, and Technology Committees oversee risks associated with their respective areas
of responsibility. As part of its overall review of the Company's compensation policies and practices, the Compensation Committee generally considers the risks associated with these policies and
practices. The Corporate Governance and Compliance Committee oversees all aspects of the Company's comprehensive compliance program other than financial compliance and considers legal and regulatory
compliance risks. The Technology Committee considers risks associated with our research and development programs.
- •
- The board is kept abreast of its Committees' risk oversight and other activities via reports of the Committee chairmen to the full board at regular board meetings. The board considers specific risk topics, including risks associated with our strategic plan, our finances, and our development activities. In addition, the board receives detailed regular reports from members of our senior management that include discussions of the risks and exposures involved in their respective areas of responsibility. Further, the board is routinely informed by the appropriate members of senior management of developments internal and external to the Company that could affect our risk profile.
Board Meetings and Attendance of Directors
The board held five regular meetings and two special meetings in 2015. All directors attended more than 75% of the total number of meetings of the board and committees of the board on which they served. According to the Regeneron Board of Directors Corporate Governance Guidelines, board members are expected to attend the Company's Annual Meeting of Shareholders. All of the directors attended our 2015 Annual Meeting of Shareholders, with one director participating through electronic conferencing.
Executive Compensation Processes and Procedures; Role of Compensation Consultants
The Compensation Committee is responsible for overseeing the Company's general compensation objectives and programs. We describe the role of the Compensation Committee, as well as the role of our executive officers, in decisions regarding executive compensation (particularly with respect to our Named Officers referred to below under "Executive Compensation") below under "Executive Compensation – Compensation Discussion and Analysis – Section 3 – Executive Compensation Process and Considerations – Overview."
19
Corporate Governance
As discussed in greater detail under "Executive Compensation – Compensation Discussion and Analysis – Section 3 – Executive Compensation Process and Considerations," the Compensation Committee has the sole authority to retain its own third-party compensation consultants, and in 2015 utilized the services of Frederic W. Cook & Co., Inc. ("Frederic W. Cook & Co."), a compensation consultant. Advice and recommendations provided by Frederic W. Cook & Co. may relate to both executive compensation (discussed under "Executive Compensation" below) and director compensation matters (discussed under " – Compensation of Directors" below). In addition, management retains another compensation consultant for its own use. In 2015, management used the services of Radford, a compensation consultant focused on the technology and life sciences sectors. Radford provided various consulting services to us, including analyzing the competitiveness of specific compensation programs; preparing surveys of competitive pay practices (including the 2015 Radford Global Life Sciences Survey discussed in this proxy statement); and assisting management in the development and analysis of executive compensation recommendations. Reports prepared by Radford that relate to executive compensation may also be shared with the Compensation Committee.
Overview
The general philosophy we have applied to compensation of our non-employee directors and the Chairman of the Board is similar to the executive compensation philosophy outlined in the Compensation Discussion and Analysis section of this proxy statement. This philosophy places an emphasis on equity compensation in the form of stock options, which reward growth in stock price and align the directors' interests with those of our shareholders by providing value to the directors only if there is future stock price appreciation and not rendering any value to the directors if the stock price declines below the applicable exercise price. Similar to executive compensation, the emphasis on long-term incentives in the form of stock options has been a consistent part of Regeneron's director compensation philosophy and preceded the significant appreciation in Regeneron's stock price that began in early 2011. As discussed in greater detail below, the board of directors voluntarily reduced the number of shares underlying the three most recent annual stock option awards to the non-employee directors and the Chairman of the Board by 15% each time, consistent with the reductions in annual awards to executive officers implemented in December 2013, 2014, and 2015.
The Corporate Governance and Compliance Committee makes recommendations to the board of directors regarding, and the board of directors determines, the compensation of non-employee directors. The Corporate Governance and
Compliance Committee evaluates the appropriate level and form of compensation for non-employee directors at least annually and recommends changes to the board of directors when appropriate. Directors who are Company employees receive no additional compensation for serving on our board of directors or its committees. In determining compensation recommendations for the non-employee directors, the Corporate Governance and Compliance Committee considers the qualifications, expertise, demands on our directors, practices of similar companies in the biotechnology industry, and any recommendations provided by the compensation consultants of the Committee or management. The recent changes to the non-employee director compensation program described below were adopted based on such considerations.
Cash Fees and Matching Gift Program
A non-employee director receives an annual retainer of $55,000 and an annual committee retainer of $10,000 for each standing committee on which the director serves. In 2015, the Chairman of the Audit Committee received an additional retainer of $5,000 (increased to $10,000 in the aggregate starting in 2016 as a result of the change described in the following sentence). Starting in 2016, the board of directors approved an annual retainer of $10,000 for each chairperson of the standing committees of the Company's board of directors, which is in addition to the annual retainer payable to all non-employee directors and the annual retainer payable in respect of service as a director on each standing committee (which remained unchanged). Non-employee directors are reimbursed for their actual expenses incurred in connection with their activities as directors, which included travel, hotel, and food and entertainment expenses. In addition, directors are eligible to participate in the Regeneron Matching Gift Program, which was adopted effective January 1, 2013 and is also available to eligible employees. Under this program, the Company matches contributions made to eligible tax-exempt organizations. Effective January 1, 2014, the maximum annual match has been increased to $5,000 (from $2,000) per person.
Annual Stock Option Awards
Pursuant to the terms of the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan and resolutions of the Compensation Committee adopted on December 16, 2014 and December 16, 2015, each non-employee director receives an automatic grant of a stock option to purchase common stock on the first business day of each year, with an exercise price equal to the fair market value of a share of common stock on the date of grant (determined, for so long as our common stock is listed on the NASDAQ Global Select Market, as the average of the high and low sales price per share of common stock on the NASDAQ Global Select Market on the date of grant or, if
20
Corporate Governance
such date is not a trading day, on the last preceding date on which there was a sale of the Company's common stock on the NASDAQ Global Select Market). These stock options become exercisable as to one-third of the shares on the anniversary of the date of grant in each of the three subsequent calendar years, generally subject to continued service on the board, and generally expire ten years following the date of grant. In 2015, similar to the reductions in annual awards to executive officers and other employees discussed under "Executive Compensation" below, the Compensation Committee reduced the automatic grant of stock options to our non-employee directors by 15%, from 10,838 shares to 9,212 shares of common stock underlying each such stock option. Similar to the rationale for the reductions in annual awards to executive officers and other employees discussed under "Executive Compensation" below, the impetus for the reductions in the automatic grants to the non-employee directors was to reduce the potential dilutive impact of these grants; the reductions also took into account the increase in the Company stock price since the prior years' awards, with the resulting increases in the grant date fair values of the automatic grants (as determined according to the Black-Scholes model for valuing stock options). The reduced grants to our non-employee directors were made on January 4, 2016. This decrease constituted the third consecutive double-digit percentage decrease in the annual grant of stock options to our non-employee directors, in each case following high stock appreciation, consistent with the reductions in annual awards to executive officers discussed under "Executive Compensation" below. To the extent they remain unvested and outstanding, stock options granted to a non-employee director continue to vest following the retirement of that director provided applicable conditions relating to the length of the director's service and the director's age have been met. If a non-employee director's service as a member of the board is terminated as a result of his or her death, all of the director's stock options will immediately vest in full.
To the extent they remain unvested and outstanding, stock options granted to non-employee directors become fully vested automatically upon a change of control of the Company. Each non-employee director has the right to nullify this acceleration of vesting, in whole or in part, if it would cause the director to pay excise taxes under the requirements of the Internal Revenue Code.
Stock Option Awards to New Directors
Starting in 2016, each new non-employee director will receive an initial stock option award to purchase a number of shares equal to 5/3rds of the number of shares of common stock
underlying the most recent regular annual stock option award to a non-employee director; and, with respect to the annual stock option award to a non-employee director in respect of the first year of his or her service, the number of shares of common stock underlying such annual award will be prorated based on the date as of which the non-employee director first becomes a member of the board of directors.
Compensation Arrangements of the Chairman of the Board of Directors
On December 31, 1998, we entered into an employment agreement with the Chairman of the board of directors, Dr. Vagelos. Dr. Vagelos did not become an officer of the Company or change his title. Pursuant to the terms of his employment agreement, Dr. Vagelos receives an annual salary of $100,000. In the employment agreement, we agreed to recommend to the Compensation Committee that stock option grants be made to Dr. Vagelos for calendar years 2000 through 2003 in the amount of the greater of (a) 125,000 shares or (b) 125% of the highest annual option award granted to an officer of the Company.
In 2011, the Compensation Committee determined that Dr. Vagelos's target grant would be equal to ten times the annual grant for a non-employee member of the board of directors, setting his target award at 150,000 shares of common stock underlying stock options. In December 2013, 2014, and 2015, the Compensation Committee reduced the award to Dr. Vagelos by 15% each time, in line with the reduction in the annual stock option awards to our executive officers and the reduction in the annual stock option awards to the non-employee directors made in January 2014, 2015, and 2016. On December 16, 2015, the Compensation Committee granted Dr. Vagelos stock options to purchase 92,123 shares of common stock, at an exercise price of $555.67 per share, the fair market value per share of our common stock on the date of grant (determined as the average of the high and low sales price per share of common stock on the NASDAQ Global Select Market on the date of grant). The stock option award granted to Dr. Vagelos vests ratably over four years subject to his continued service and contains change-of-control provisions consistent with those described above for stock option grants to non-employee directors. Pursuant to the terms of his employment agreement, if Dr. Vagelos dies or is disabled during the term of his employment, all stock options granted to him by the Company will immediately become vested and exercisable.
21
Corporate Governance
The following table and explanatory footnotes provide information with respect to compensation paid to Dr. Vagelos and each non-employee director for their service in 2015
in accordance with the policies, plans, and employment agreement described above:
Director Compensation
Name |
|
Fees earned or paid in cash ($) (b) |
Stock awards ($) (c) |
|
Option awards ($) 1,2 (d) |
Non-equity incentive plan compensation ($) (e) |
Change in pension value and non-qualified deferred compensation earnings (f) |
|
All other compensation ($) (g) |
|
Total ($) (h) |
||||||||
Charles A. Baker |
| 75,000 | – | | 1,986,604 | – | – | | – | | 2,061,604 | ||||||||
Michael S. Brown, M.D. |
| 75,000 | – | | 1,986,604 | – | – | | 5,000 | 3 | | 2,066,604 | |||||||
Alfred G. Gilman, M.D., Ph.D. 4 |
| 75,000 | – | | 1,986,604 | – | – | | – | | 2,061,604 | ||||||||
Joseph L. Goldstein, M.D. |
| 75,000 | – | | 1,986,604 | – | – | | – | | 2,061,604 | ||||||||
Robert A. Ingram 5 |
| 65,000 | | | 1,986,604 | – | – | | – | | 2,051,604 | ||||||||
Christine A. Poon |
| 75,000 | – | | 1,986,604 | – | – | | – | | 2,061,604 | ||||||||
Arthur F. Ryan |
| 75,000 | – | | 1,986,604 | – | – | | 2,500 | 3 | | 2,064,104 | |||||||
George L. Sing |
| 80,000 | – | | 1,986,604 | – | – | | 5,000 | 3 | | 2,071,604 | |||||||
Marc Tessier-Lavigne, Ph.D. |
| 75,000 | – | | 1,986,604 | – | – | | – | | 2,061,604 | ||||||||
P. Roy Vagelos, M.D. |
| – | – | | 23,098,558 | – | – | | 109,000 | 6 | | 23,207,558 | |||||||
| | | | | | | | | | | | | | | | | | | | |
- 1
- The amounts in column (d) reflect the aggregate grant date fair value of options awarded during the year ended December 31, 2015 pursuant to the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan. Assumptions used in the calculation of this amount do not take into account expected forfeitures and are otherwise described in Note 14 to the Company's audited financial statements for the fiscal year ended December 31, 2015 included in the 2015 Annual Report.
- 2
- At December 31, 2015, the non-employee directors and Dr. Vagelos had the following stock option awards outstanding: Mr. Baker: 88,588; Dr. Brown: 35,588; Dr. Gilman: 38,588; Dr. Goldstein: 43,588; Mr. Ingram: 0; Ms. Poon: 93,118; Mr. Ryan: 24,338; Mr. Sing: 105,588; Dr. Tessier-Lavigne: 52,867; and Dr. Vagelos: 2,270,363.
- 3
- Consists of a Company contribution paid or payable on or before April 14, 2016 under the Regeneron Matching Gift Program in respect of a charitable gift made in 2015.
- 4
- Dr. Gilman served as a director until his death on December 23, 2015.
- 5
- Mr. Ingram served as a director until his resignation on November 10, 2015.
- 6
- Consists of (i) $100,000 for the salary paid pursuant to the terms of our employment agreement with Dr. Vagelos, (ii) $4,000 for 401(k) Savings Plan matching contributions in respect of 2015 paid in February 2016, and (iii) $5,000 for a Company contribution paid or payable on or before April 14, 2016 under the Regeneron Matching Gift Program in respect of a charitable gift made in 2015.
22
Corporate Governance
![]()
Executive Officers of the Company
All officers of the Company are appointed annually and serve at the pleasure of the board of directors. The names, positions, ages, and background of the Company's executive officers as of April 14, 2016 are set forth below. Except as identified below, there are no family relationships between any of our directors and executive officers. None of the corporations or other organizations referred to below with which an executive officer has previously been employed or otherwise associated is a parent, subsidiary, or affiliate of the Company.
LEONARD S. SCHLEIFER, M.D., Ph.D., 63, co-founded the Company in 1988, has been a Director and its President and Chief Executive Officer since its inception, and served as Chairman of the Board from 1990 through 1994. Dr. Schleifer received his M.D. and Ph.D. in Pharmacology from the University of Virginia. Dr. Schleifer is a licensed physician and is certified in Neurology by the American Board of Psychiatry and Neurology.
GEORGE D. YANCOPOULOS, M.D., Ph.D., 56, joined the Company in 1989 as its Founding Scientist and is currently President, Regeneron Laboratories and Chief Scientific Officer. While holding leadership positions, Dr. Yancopoulos headed the Company's laboratories and science organization since joining the Company and, in 1998, was named the Company's first Chief Scientific Officer. Dr. Yancopoulos joined the board in 2001. He received his M.D. and Ph.D. from Columbia University. Dr. Yancopoulos was the 11th most highly cited scientist in the world in the 1990s, and in 2004 he was elected to be a member of the National Academy of Sciences. Dr. Yancopoulos, together with key members of his team, is a principal inventor and developer of the four FDA-approved drugs the Company has developed, EYLEA®, Praluent®, ZALTRAP®, and ARCALYST®, as well as of its foundation technologies, including the TRAP technology, VelociGene®, and VelocImmune®.
MICHAEL ABERMAN, M.D., 45, has been Senior Vice President, Strategy and Investor Relations since January 2015. From March 2010 to December 2014, he served as Vice President, Strategy and Investor Relations. Prior to joining the Company, he spent six years as a Wall Street analyst covering the biotechnology industry. From March 2006 until joining the Company, he was Director and Senior Biotechnology Analyst at Credit Suisse. Prior to that, from March 2004 until March 2006, he worked as a Biotechnology Analyst at Morgan Stanley, Inc. From February 2002 through March 2004, Dr. Aberman was Director of Business Development at Antigenics Inc., an oncology-focused biotechnology company. Dr. Aberman received his M.D. with honors from the University of Toronto and his M.B.A. from the Wharton School of the University of Pennsylvania.
ROBERT E. LANDRY, 52, has been Senior Vice President, Finance since September 2013 and Chief Financial Officer since October 2013. Previously, Mr. Landry served as Senior Vice President, Treasurer, at Pfizer Inc. from October 2012 to August 2013 and Senior Vice President – Finance, Pfizer's Diversified Business, from October 2009 to October 2012. Prior to those roles, Mr. Landry held a number of positions at Wyeth, which was acquired by Pfizer Inc. in October 2009, including Treasurer and Principal Corporate Officer from 2007 to 2009, Director of Pharmaceutical Marketing and Sales of Wyeth's Australian affiliate from 2006 to 2007, and Chief Financial Officer of Wyeth's Australian and New Zealand affiliates from 2004 to 2006.
JOSEPH J. LAROSA, 57, has been Senior Vice President, General Counsel, and Secretary since September 2011. Before joining Regeneron, Mr. LaRosa was Senior Vice President, General Counsel, and Secretary at Nycomed US Inc. Mr. LaRosa's prior experience includes working in a number of senior legal positions at Schering-Plough Corporation from 1993 to 2009, where he was a corporate officer and served most recently as Vice President, Legal Affairs, and a member of the Operations Management Team. Mr. LaRosa received his J.D. from New York University School of Law.
DOUGLAS S. McCORKLE, 59, has been Vice President, Controller, and Assistant Treasurer since 2007. Prior to that date, he served as Controller and Assistant Treasurer since 1998. Prior to joining the Company, Mr. McCorkle was Controller of Intergen Company, a manufacturer of biopharmaceutical products, a position he held since 1997. From 1990 to 1996, Mr. McCorkle was employed with Coopers & Lybrand L.L.P., where he specialized in biotechnology clients and served in various positions including Audit Manager from 1995 to 1996. As previously reported, Mr. McCorkle has notified the Company that he plans to retire in early 2017.
PETER POWCHIK, M.D., 59, has been Senior Vice President, Clinical Development since joining the Company in October 2006. Prior to joining the Company, Dr. Powchik was employed at several pharmaceutical companies, serving as Senior Vice President and Chief Medical Officer of Chugai Pharma USA, a position he held from May 2005 until October 2006. From April 2001 until May 2005, he held various senior clinical development positions at Novartis Pharmaceuticals Corporation, most recently as Vice President, US Clinical Development and Medical Affairs. Dr. Powchik held various clinical development positions with Sepracor Inc. and Pfizer Inc. from October 1996 to April 2001.
23
Executive Officers of the Company
Dr. Powchik received his M.D. from New York University School of Medicine.
NEIL STAHL, Ph.D., 59, has been Executive Vice President, Research and Development since January 2015. He previously served as Senior Vice President, Research and Development Sciences from January 2007 to December 2014, as Senior Vice President, Preclinical Development and Biomolecular Sciences from December 2000 to December 2007, and as Vice President, Preclinical Development and Biomolecular Sciences from January 2000 to December 2000. He joined the Company in 1991. Before becoming Vice President, Biomolecular Sciences in July 1997, Dr. Stahl was Director, Cytokines and Signal Transduction. Dr. Stahl received his Ph.D. in Biochemistry from Brandeis University.
ROBERT J. TERIFAY, 56, has been Executive Vice President, Commercial since January 2016. From February 2007 to December 2015, he served as Senior Vice President, Commercial. Prior to joining the Company, Mr. Terifay was employed at several biopharmaceutical companies. From January to October 2006, Mr. Terifay served as President and Chief Operating Officer of Arginox Pharmaceuticals. Prior to his employment at Arginox, Mr. Terifay was Senior Vice President, Business Operations at Synta Pharmaceuticals from March to December 2005. From February 2002 until March 2005, he held various senior commercial and marketing positions at Millennium Pharmaceuticals, Inc., most recently as Senior Vice President, Oncology Commercial. Mr. Terifay was Vice President, Marketing at Cor Therapeutics, Inc. from 1996
until its acquisition by Millennium Pharmaceuticals, Inc. in February 2002. Mr. Terifay was Executive Vice President of Strategic Services at Saatchi & Saatchi, an advertising firm, from 1993 to 1996. From 1985 to 1993, he held various commercial and marketing positions at G.D. Searle & Company. Mr. Terifay received his Master of Management degree in Marketing and Health Service Management from the J.L. Kellogg Graduate School of Management, Northwestern University.
DANIEL P. VAN PLEW, 43, has been Executive Vice President and General Manager, Industrial Operations and Product Supply since January 2016. From April 2008 to December 2015, Mr. Van Plew served as Senior Vice President and General Manager, Industrial Operations and Product Supply. Prior to that date, he served as Vice President and General Manager, Industrial Operations and Product Supply since joining the Company in 2007. From 2006 until 2007, Mr. Van Plew served as Executive Vice President, R&D and Technical Operations of Crucell Holland B.V., a global biopharmaceutical company. Between 2004 and 2006, Mr. Van Plew held positions of increasing responsibility at Chiron Biopharmaceuticals, part of Chiron Corporation, a biotechnology company, most recently as Senior Director, Vacaville Operations. From 1998 until 2004, Mr. Van Plew held various managerial positions in the health and life sciences practice at Accenture, Ltd., a management consulting business. Mr. Van Plew received his M.S. in Chemistry from The Pennsylvania State University and his M.B.A. from Michigan State University.
24
Executive Officers of the Company
![]()
Security Ownership of Certain Beneficial Owners and Management
The following table sets forth, as of April 14, 2016, the number of shares of the Company's Class A stock and common stock beneficially owned by each of the Company's directors, each of the Named Officers referred to below under "Executive Compensation," all directors and executive officers as a group, and each other person or group of persons known by the Company to beneficially own more than 5% of the outstanding shares of common stock or Class A stock, based upon (unless indicated otherwise) information obtained from such persons, and the percentage that such shares represent of the number of outstanding shares of Class A stock and common stock, respectively.
The Class A stock is convertible on a share-for-share basis into common stock. The Class A stock is entitled to ten votes per share and the common stock is entitled to one vote per share. We have determined beneficial ownership in accordance with the rules of the SEC. Except as otherwise indicated in the footnotes below, we believe, based on the information furnished or otherwise available to us, that the persons named in the table below have sole voting and investment power with respect to all shares of Class A stock and common stock shown as beneficially owned by them, subject to applicable community property laws. We have based our calculation of percentage of shares of a class beneficially owned on
1,913,136 shares of Class A stock and 103,165,457 shares of common stock outstanding as of April 14, 2016, except that for each person listed who beneficially owns Class A stock (and for directors and executive officers as a group), the number of shares of common stock beneficially owned by that person (and by directors and executive officers as a group) and the percentage ownership of common stock of such person assume the conversion on April 14, 2016 of all shares of Class A stock listed as beneficially owned by such person (or persons in the case of directors and executive officers as a group) into common stock and also that no other shares of Class A stock beneficially owned by others are so converted.
In computing the number of shares of common stock beneficially owned by a person (and by directors and executive officers as a group) and the percentage ownership of common stock of such person (and by directors and executive officers as a group), shares of common stock subject to options held by that person (and by directors and executive officers as a group) that are exercisable as of April 14, 2016 or are exercisable within sixty days after April 14, 2016 are deemed to be outstanding. Such shares are not deemed to be outstanding, however, for the purpose of computing the percentage ownership of common stock of any other person.
25
Security Ownership of Certain Beneficial Owners and Management
|
|
Shares of Class A Stock Beneficially Owned1 |
|
Shares of Common Stock Beneficially Owned1 |
|||||||
| | | | | | | | | | | | |
Name and Address of Beneficial Owner |
|
Number |
Percent of Class |
|
Number2 |
Percent of Class |
|||||
Beneficial Owners of 5% or More of Common Stock or |
|||||||||||
| | | | | | | | | | | | |
Sanofi 3 |
| — | — | | 23,353,665 | 22.6% | |||||
54, rue La Boetie |
| | |||||||||
75008 Paris |
| | |||||||||
France |
| | |||||||||
| | | | | | | | | | | | |
Capital World Investors 4 |
— | — | 6,804,465 | 6.6% | |||||||
333 South Hope Street |
|||||||||||
Los Angeles, California 90071 |
|||||||||||
| | | | | | | | | | | | |
FMR LLC 5 |
| — | — | | 6,223,092 | 6.0% | |||||
245 Summer Street |
| | |||||||||
Boston, Massachusetts 02210 |
| | |||||||||
| | | | | | | | | | | | |
BlackRock, Inc. 6 |
— | — | 5,382,473 | 5.2% | |||||||
55 East 52nd Street |
|||||||||||
New York, New York 10055 |
|||||||||||
| | | | | | | | | | | | |
Directors and Executive Officers: 7 |
| | |||||||||
| | | | | | | | | | | | |
Leonard S. Schleifer, M.D., Ph.D. |
1,726,565 | 8 | 90.3% | 3,993,844 | 9 | 3.7% | |||||
| | | | | | | | | | | | |
P. Roy Vagelos, M.D. |
| — | — | | 2,875,335 | 10 | 2.7% | ||||
| | | | | | | | | | | | |
George D. Yancopoulos, M.D., Ph.D. |
42,750 | 11 | 2.2% | 2,967,079 | 12 | 2.8% | |||||
| | | | | | | | | | | | |
Charles A. Baker |
| 62,384 | 13 | 3.3% | | 148,497 | 14 | * | |||
| | | | | | | | | | | | |
Michael S. Brown, M.D. |
— | — | 47,462 | 15 | * | ||||||
| | | | | | | | | | | | |
Joseph L. Goldstein, M.D. |
| — | — | | 40,113 | 16 | * | ||||
| | | | | | | | | | | | |
Christine A. Poon |
— | — | 82,433 | 17 | * | ||||||
| | | | | | | | | | | | |
Arthur F. Ryan |
| — | — | | 53,363 | 18 | * | ||||
| | | | | | | | | | | | |
George L. Sing |
— | — | 224,385 | 19 | * | ||||||
| | | | | | | | | | | | |
Marc Tessier-Lavigne, Ph.D. |
— | — | 42,579 | 20 | * | ||||||
| | | | | | | | | | | | |
Robert E. Landry |
| — | — | | 41,656 | 21 | * | ||||
| | | | | | | | | | | | |
Neil Stahl, Ph.D. |
— | — | 405,164 | 22 | * | ||||||
| | | | | | | | | | | | |
Robert J. Terifay |
| — | — | | 197,466 | 23 | * | ||||
| | | | | | | | | | | | |
All Directors and Executive Officers as a Group (18 persons) |
1,831,699 | 95.7% | 11,720,333 | 24 | 10.4% | ||||||
| | | | | | | | | | | | |
- *
- Represents less than 1%
- 1
- The inclusion herein of any Class A stock or common stock, as the case may be, deemed beneficially owned does not constitute an admission of beneficial ownership of those shares.
- 2
- For each person listed who beneficially owns Class A stock (and for directors and executive officers as a group), the number of shares of common stock listed includes the number of shares of Class A stock listed as beneficially owned by such person (or persons in the case of directors and executive officers as a group).
- 3
- Based solely on a Form 4 filed by Sanofi with the SEC on February 18, 2016. According to this Form 4, 20,554,113 of the shares are held directly by sanofi-aventis Amerique du Nord and 2,799,552 of the shares are held directly by Aventis Pharmaceuticals Inc. sanofi-aventis Amerique du Nord is a direct, wholly-owned subsidiary of Sanofi, and Aventis Pharmaceuticals Inc. is an indirect, wholly-owned subsidiary of sanofi-aventis Amerique du Nord. Pursuant to the Amended and Restated Investor Agreement, dated as of January 11, 2014, by and among Sanofi, sanofi-aventis US LLC, Aventis Pharmaceuticals Inc., and sanofi-aventis Amerique du Nord (collectively, the "Sanofi Parties"), and the Company, the Sanofi Parties have agreed to vote all shares of our voting securities they are entitled to vote from time to time as recommended by our board of directors, except that they may elect to vote proportionally with the votes cast by all of our other shareholders with respect to certain change-of-control transactions and to vote in their sole discretion with respect to liquidation or dissolution of Regeneron, stock issuances equal to or exceeding 20% of the then outstanding shares or voting rights of common stock and Class A stock (taken together), and new equity compensation plans or amendments if not materially consistent with our historical equity compensation practices. See "Certain Relationships and Related Transactions – Transactions with Related Persons – Amended and Restated Investor Agreement with Sanofi" for further information regarding the Amended and Restated Investor Agreement with Sanofi.
26
Security Ownership of Certain Beneficial Owners and Management
- 4
- Based solely on an amendment to a Schedule 13G filed by Capital World Investors on February 16, 2016. According to this amendment, Capital World Investors, a division of Capital Research and Management Company, has sole voting and dispositive power as to all of the shares reported as beneficially owned. Capital World Investors is deemed to be the beneficial owner of such shares as a result of Capital Research and Management Company acting as investment adviser to various registered investment companies.
- 5
- Based solely on an amendment to a Schedule 13G filed by FMR LLC on February 12, 2016. According to this amendment, FMR LLC has sole voting power as to 639,403 of the shares reported as beneficially owned and sole dispositive power as to all of the shares reported as beneficially owned. Abigail P. Johnson is a Director, the Vice Chairman, the Chief Executive Officer, and the President of FMR LLC. Members of the Johnson family, including Abigail P. Johnson, are the predominant owners, directly or through trusts, of Series B voting common shares of FMR LLC, representing 49% of the voting power of FMR LLC. The Johnson family group and all other Series B shareholders have entered into a shareholders' voting agreement under which all Series B voting common shares will be voted in accordance with the majority vote of Series B voting common shares. Accordingly, through their ownership of voting common shares and the execution of the shareholders' voting agreement, members of the Johnson family may be deemed to form a controlling group with respect to FMR LLC. Neither FMR LLC nor Abigail P. Johnson has the sole power to vote or direct the voting of the shares owned directly by the various investment companies (the "Fidelity Funds") advised by Fidelity Management & Research Company, a wholly owned subsidiary of FMR LLC, which power resides with the Fidelity Funds' Boards of Trustees. Fidelity Management & Research Company carries out the voting of the shares under written guidelines established by the Fidelity Funds' Boards of Trustees.
- 6
- Based solely on an amendment to a Schedule 13G filed by BlackRock, Inc. on February 10, 2016. According to this amendment, BlackRock, Inc. has sole voting power as to 4,830,005, shared voting power as to 5,449, sole dispositive power as to 5,377,024, and shared dispositive power as to 5,449 of the shares reported as beneficially owned.
- 7
- The address for each director and executive officer is c/o Regeneron Pharmaceuticals, Inc., 777 Old Saw Mill River Road, Tarrytown, New York 10591-6707.
- 8
- Includes 15,775 shares of Class A stock held in trust for the benefit of Dr. Schleifer's son, of which Dr. Schleifer is a trustee.
- 9
- Includes (i) 2,218,771 shares of common stock purchasable upon the exercise of options granted pursuant to the Regeneron Pharmaceuticals, Inc. Second Amended and Restated 2000 Long-Term Incentive Plan or the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan that are exercisable or become so within sixty days after April 14, 2016; and (ii) 5,702 shares of common stock held in an account under the Company's 401(k) Savings Plan.
- 10
- Includes (i) 1,995,705 shares of common stock purchasable upon exercise of options granted pursuant to the Regeneron Pharmaceuticals, Inc. Second Amended and Restated 2000 Long-Term Incentive Plan or the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan that are exercisable or become so within sixty days after April 14, 2016; (ii) 2,290 shares of common stock held in an account under the Company's 401(k) Savings Plan; (iii) 152,964 shares of common stock held in a charitable lead annuity trust, of which Dr. Vagelos is the trustee; (iv) 92,947 shares of common stock held in a trust for his grandchildren, of which Dr. Vagelos's wife is the trustee; (v) 1,203 shares of common stock held in trusts for his grandchildren, of which Dr. Vagelos and/or his wife are trustees; and (vi) 56,159 shares of common stock and 241,187 shares of common stock held by the Marianthi Foundation and the Pindaros Foundation, respectively, both of which are charitable foundations of which Dr. Vagelos is a director and an officer. Dr. Vagelos disclaims beneficial ownership of the shares held by these charitable foundations.
- 11
- Of these shares, 23,367 shares are held in trust for the benefit of Dr. Yancopoulos's children and certain other family members; Dr. Yancopoulos is a trustee of the trust. The remaining 19,383 shares are held in custody for the benefit of Dr. Yancopoulos's children.
- 12
- Includes (i) 1,848,756 shares of common stock purchasable upon exercise of options granted pursuant to the Regeneron Pharmaceuticals, Inc. Second Amended and Restated 2000 Long-Term Incentive Plan or the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan that are exercisable or become so within sixty days after April 14, 2016; (ii) 500,000 shares of restricted stock which vest on December 17, 2017; (iii) 5,676 shares of common stock held in an account under the Company's 401(k) Savings Plan; and (iv) 567,976 shares of common stock held in trust for the benefit of Dr. Yancopoulos's children and certain other family members, of which Dr. Yancopoulos is a trustee.
- 13
- All shares of Class A stock are held by a limited partnership, of which Mr. Baker is a general partner.
- 14
- Includes 77,113 shares of common stock purchasable upon exercise of options granted pursuant to the Regeneron Pharmaceuticals, Inc. Second Amended and Restated 2000 Long-Term Incentive Plan or the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan that are exercisable or become so within sixty days after April 14, 2016.
- 15
- Includes (i) 24,113 shares of common stock purchasable upon exercise of options granted pursuant to the Regeneron Pharmaceuticals, Inc. Second Amended and Restated 2000 Long-Term Incentive Plan or the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan that are exercisable or become so within sixty days after April 14, 2016; and (ii) 6,000 shares of common stock held by a family charitable foundation of which Dr. Brown is a director and an officer and his wife is a director. Dr. Brown disclaims beneficial ownership of the shares held by this charitable foundation.
- 16
- Includes 27,113 shares of common stock purchasable upon exercise of options granted pursuant to the Regeneron Pharmaceuticals, Inc. Second Amended and Restated 2000 Long-Term Incentive Plan or the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan that are exercisable or become so within sixty days after April 14, 2016.
- 17
- Includes 81,643 shares of common stock purchasable upon exercise of options granted pursuant to the Regeneron Pharmaceuticals, Inc. Second Amended and Restated 2000 Long-Term Incentive Plan or the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan that are exercisable or become so within sixty days after April 14, 2016.
- 18
- Includes 12,863 shares of common stock purchasable upon exercise of options granted pursuant to the Regeneron Pharmaceuticals, Inc. Second Amended and Restated 2000 Long-Term Incentive Plan or the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan that are exercisable or become so within sixty days after April 14, 2016.
27
Security Ownership of Certain Beneficial Owners and Management
- 19
- Includes (i) 94,113 shares of common stock purchasable upon exercise of options granted pursuant to the Regeneron Pharmaceuticals, Inc. Second Amended and Restated 2000 Long-Term Incentive Plan or the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan that are exercisable or become so within sixty days after April 14, 2016; (ii) 3,000 shares of common stock held by Mr. Sing's spouse; (iii) 4,500 shares of common stock held by Mr. Sing's spouse as custodian for the benefit of their son; and (iv) 10,000 shares of common stock held in a trust for benefit of Mr. Sing's son.
- 20
- Includes 41,392 shares of common stock purchasable upon exercise of options granted pursuant to the Regeneron Pharmaceuticals, Inc. Second Amended and Restated 2000 Long-Term Incentive Plan or the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan that are exercisable or become so within sixty days after April 14, 2016.
- 21
- Includes (i) 34,500 shares of common stock purchasable upon exercise of options granted pursuant to the Regeneron Pharmaceuticals, Inc. Second Amended and Restated 2000 Long-Term Incentive Plan or the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan that are exercisable or become so within sixty days after April 14, 2016; (ii) 5,000 shares of restricted stock which vest on September 9, 2018; and (iii) 57 shares of common stock held in an account under the Company's 401(k) Savings Plan.
- 22
- Includes (i) 352,717 shares of common stock purchasable upon exercise of options granted pursuant to the Regeneron Pharmaceuticals, Inc. Second Amended and Restated 2000 Long-Term Incentive Plan or the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan that are exercisable or become so within sixty days after April 14, 2016; (ii) 20,732 shares of common stock held in separate grantor retained annuity trusts of which Dr. Stahl is the trustee; and (iii) 5,621 shares of common stock held in an account under the Company's 401(k) Savings Plan.
- 23
- Includes (i) 172,500 shares of common stock purchasable upon exercise of options granted pursuant to the Regeneron Pharmaceuticals, Inc. Second Amended and Restated 2000 Long-Term Incentive Plan or the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan that are exercisable or become so within sixty days after April 14, 2016; and (ii) 1,673 shares of common stock held in an account under the Company's 401(k) Savings Plan.
- 24
- Includes (i) 7,483,044 shares of common stock purchasable upon exercise of options granted pursuant to the Regeneron Pharmaceuticals, Inc. Second Amended and Restated 2000 Long-Term Incentive Plan or the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan that are exercisable or become so within sixty days after April 14, 2016; (ii) 515,000 shares of unvested restricted stock; and (iii) 28,853 shares of common stock held in an account under the Company's 401(k) Savings Plan.
28
Security Ownership of Certain Beneficial Owners and Management
![]()
Section 16(a) Beneficial Ownership Reporting Compliance
Based solely upon a review of reports filed pursuant to Section 16(a) of the Exchange Act or written representations from reporting persons, the Company is not aware of any director, executive officer, or beneficial owner of more than 10%
of our common stock who has not filed on a timely basis any report required by such Section 16(a) to be filed during or in respect of our fiscal year ended December 31, 2015.
29
Section 16(a) Beneficial Ownership Reporting Compliance
![]()
Certain Relationships and Related Transactions
Review, Approval, or Ratification of Transactions with Related Persons
The board of directors has adopted a written policy for the review, approval, or ratification of related person transactions. The Company considers transactions (or a series of related transactions) in which the Company is a participant, the amount involved exceeds $10,000 in any calendar year, and a director, officer, more than 5% holder of our voting securities, any immediate family member of any of the foregoing, or any related entity of any of the foregoing has a direct or indirect material interest to constitute related person transactions. As amended in January 2015, the policy provides for a standing pre-approval of transactions with any passive institutional shareholder who holds more than 5% of our voting securities, transactions where all shareholders receive proportional benefits, and certain transactions with Sanofi. With respect to any new transaction that is deemed pre-approved, the Audit Committee receives a summary of each such transaction and retains the ability to require that one or more of such transactions be subject to the standard approval procedures. The policy also requires that the arrangements relating to a permanent, full-time employment of an immediate family member of a director or executive officer hired by the Company be approved in accordance with the policy. In addition, in the event a person is or becomes a director or executive officer of the Company and an immediate family member of such person is a permanent, full-time employee of the Company, no material, outside-of-the-ordinary-course-of-business change in the terms of employment, including compensation, are permitted to be made without the prior approval of the Audit Committee (except, if the immediate family member is himself or herself an executive officer of the Company, any proposed change in the terms of employment are reviewed and approved in the same manner as compensatory arrangements of other executive officers).
The board of directors determined that the members of the Audit Committee are best suited to review and approve related person transactions. Accordingly, each related person transaction (other than a transaction that is deemed pre-approved as described above) must be reviewed and approved or ratified by the members of the Audit Committee, other than any member of the Audit Committee that has an interest in the transaction. Under the policy, the Chairman of the Audit Committee is delegated the authority to approve certain related person transactions that require urgent review and approval.
When reviewing, approving, or ratifying a related person transaction, the Audit Committee will consider several factors,
including the benefits to the Company, the impact on a director's independence in the event that a director or his/her immediate family is involved in the transaction, the terms of the transaction, and the terms available to unrelated third parties or to employees in general, if applicable. Related person transactions are approved only if the Audit Committee (or the Chairman of the Audit Committee pursuant to delegated authority in the circumstances noted above) determines that they are in, or are not inconsistent with, the best interests of the Company and our shareholders.
Transactions with Related Persons
Collaborations with Sanofi
As the beneficial owner of 23,353,665 shares of common stock of the Company, or 22.6% of the common stock outstanding as of April 14, 2016, Sanofi is considered a related person of the Company. In 2015, Sanofi funded $145.0 million of our antibody discovery expenses under the Amended and Restated Discovery and Preclinical Development Agreement, and $747.8 million of our development and other costs (including $157.4 million of commercialization-related expenses) under the Amended and Restated License and Collaboration Agreement. In addition, in 2015, we funded $92.6 million of Sanofi's Phase 3 development costs for Praluent® and sarilumab under the Amended and Restated License and Collaboration Agreement. In 2015, we and Sanofi shared losses in connection with commercialization-related activities for Praluent® (which was launched in 2015 following receipt of regulatory approval in the United States and the European Union) and sarilumab, which resulted in us funding $240.0 million of such costs. In 2016, Sanofi has continued to fund the agreed-upon worldwide research and development expenses incurred by us and Sanofi, we have continued to fund certain Phase 3 development costs, and we and Sanofi have continued to share certain commercialization-related revenues and expenses under the Agreements.
In July 2015, we and Sanofi entered into a collaboration to discover, develop, and commercialize antibody-based cancer treatments in the field of immuno-oncology (the "IO Collaboration"). The IO Collaboration is governed by an Immuno-oncology Discovery and Development Agreement (the "IO Discovery Agreement"), and an Immuno-oncology License and Collaboration Agreement (the "IO License and Collaboration Agreement"). In connection with the IO Discovery Agreement, Sanofi made a $265.0 million non-refundable up-front payment to us in 2015. Pursuant to the IO Discovery Agreement, we will spend up to $1,090.0 million (the "IO Discovery Budget") to identify and validate potential immuno-oncology
30
Certain Relationships and Related Transactions
targets and develop therapeutic antibodies against such targets through clinical proof-of-concept. Sanofi will reimburse us for up to $825.0 million of these costs, subject to certain annual limits. In connection with the IO License and Collaboration Agreement, Sanofi made a $375.0 million non-refundable up-front payment to us in 2015. Under the terms of the IO License and Collaboration Agreement, the parties will also co-develop our antibody product candidate targeting the receptor known as Programmed Cell Death protein 1, or PD-1 ("REGN2810"). The parties will share equally, on an ongoing basis, development expenses for REGN2810 up to a total of $650.0 million. In 2015, Sanofi funded $29.2 million of our research and development expenses under the IO Discovery Agreement and $10.8 million under the IO License and Collaboration Agreement.
In 2013, we acquired from Sanofi exclusive rights to antibodies targeting the platelet derived growth factor (PDGF) family of receptors and ligands. In connection with this arrangement, we made a $10.0 million development milestone payment to Sanofi in 2015.
During 2014, Sanofi agreed to fund up to $17.5 million of agreed-upon costs incurred by us in connection with expanding manufacturing capacity at our Rensselaer, New York facility, of which $13.2 had been received or was receivable as of December 31, 2015.
A description of our antibody collaboration and our IO Collaboration with Sanofi is set forth in Part II, Item 7. "Management's Discussion and Analysis of Financial Condition and Results of Operations" of our 2015 Annual Report under the heading "Liquidity and Capital Resources – Collaborations with Sanofi – Antibodies" and "– Immuno-Oncology," respectively.
In February 2015, we and Sanofi entered into an amended and restated collaboration agreement relating to ZALTRAP® (ziv-aflibercept) Injection for Intravenous Infusion (the "Amended ZALTRAP® Collaboration Agreement"). Under the terms of the Amended ZALTRAP® Collaboration Agreement, Sanofi is solely responsible for the development and commercialization of ZALTRAP® for cancer indications worldwide. Sanofi bears the cost of all development and commercialization activities and reimburses Regeneron for its costs for any such activities. Sanofi will pay Regeneron a percentage of aggregate net sales of ZALTRAP® during each calendar year, which percentage shall be from 15% to 30%, depending on the aggregate net sales of ZALTRAP® in such calendar year. Regeneron is also paid for all quantities of ZALTRAP® manufactured by it pursuant to a supply agreement through the earlier of 2021 or the date Sanofi receives regulatory approval to manufacture ZALTRAP® at one of its facilities or a facility of a third party. Regeneron is no longer required to reimburse Sanofi for fifty percent of the development expenses that Sanofi previously funded for the development of ZALTRAP® under the original ZALTRAP® collaboration agreement. As a
result of entering into the Amended ZALTRAP® Collaboration Agreement, in 2015, we recognized $14.9 million of collaboration revenue, which was previously recorded as deferred revenue under the original ZALTRAP® collaboration agreement. In addition, during the year ended December 31, 2015, we recorded $38.8 million of revenue, primarily related to (i) revenues earned from Sanofi based on a percentage of net sales of ZALTRAP® and (ii) revenues earned from Sanofi for manufacturing ZALTRAP® commercial supplies.
A description of the Amended ZALTRAP® Collaboration Agreement is set forth in Part I, Item 1. "Business" of our 2015 Annual Report, under the heading "Collaboration Agreements – Collaborations with Sanofi – ZALTRAP."
Amended and Restated Investor Agreement with Sanofi
In January 2014, we entered into an Amended and Restated Investor Agreement with Sanofi. Pursuant to the agreement, Sanofi has agreed to vote its shares as recommended by our board of directors, except that it may elect to vote proportionally with the votes cast by all of our other shareholders with respect to certain change-of-control transactions and to vote in its sole discretion with respect to liquidation or dissolution of our company, stock issuances equal to or exceeding 20% of the then outstanding shares or voting rights of common stock and Class A stock (taken together), and new equity compensation plans or amendments if not materially consistent with our historical equity compensation practices.
In addition, upon Sanofi reaching 20% ownership of our then outstanding shares of Class A stock and common stock (taken together), we were required under the agreement to appoint an individual agreed upon by us and Sanofi to our board of directors. Subject to certain exceptions, we are required to use our reasonable efforts (including recommending that our shareholders vote in favor) to cause the election of this designee at our annual shareholder meetings for so long as Sanofi maintains an equity interest in us that is the lower of (i) the highest percentage ownership Sanofi attains following its acquisition of 20% of our then outstanding shares of Class A stock and common stock (taken together) and (ii) 25% of our then outstanding shares of Class A stock and common stock (taken together). This designee is required to be "independent" of Regeneron, as determined under NASDAQ rules, and not to be a current or former officer, director, employee, or paid consultant of Sanofi. In April 2014, Sanofi notified us that it had reached the 20% ownership threshold and designated Robert A. Ingram as its designee. On April 4, 2014, following recommendation of the Corporate Governance and Compliance Committee, the board of directors elected Mr. Ingram as a director and a member of the Compensation Committee. Mr. Ingram was subsequently elected as a Class I director at our 2014 annual shareholder meeting for a term expiring at the 2016 annual shareholder meeting and resigned on
31
Certain Relationships and Related Transactions
November 10, 2015. In December 2015, Sanofi disclosed in an amendment to its Schedule 13D filed with the SEC its intention to designate a successor designee.
Under the Amended and Restated Investor Agreement, Sanofi also has three demand rights to require us to use all reasonable efforts to conduct a registered underwritten offering with respect to shares of our common stock held by Sanofi from time to time; however, shares of our common stock held by Sanofi from time to time may not be sold until the later of (i) December 20, 2020 or (ii) the expiration of our discovery and preclinical development agreement with Sanofi relating to our antibody collaboration (as amended) if the agreement is extended beyond December 20, 2020. These restrictions on dispositions are subject to earlier termination upon the occurrence of certain events, such as the consummation of a change-of-control transaction involving us or a dissolution or liquidation of Regeneron.
Pursuant to the Amended and Restated Investor Agreement, Sanofi is bound by certain "standstill" provisions, which contractually prohibit Sanofi from seeking to directly or indirectly
exert control of Regeneron or acquiring more than 30% of our Class A stock and common stock (taken together). This prohibition will remain in place until the earliest of (i) the later of the fifth anniversaries of the expiration or earlier termination of our license and collaboration agreement with Sanofi relating to our antibody collaboration or our ZALTRAP® collaboration agreement with Sanofi, each as amended; (ii) our announcement recommending acceptance by our shareholders of a tender offer or exchange offer that, if consummated, would constitute a change of control involving us; (iii) the public announcement of any definitive agreement providing for a change of control involving us; (iv) the date of any issuance of shares of common stock by us that would result in another party's having more than 10% of the voting power of our then outstanding Class A stock and common stock (taken together) unless such party enters into a standstill agreement containing certain terms substantially similar to the standstill obligations of Sanofi; or (v) other specified events, such as a liquidation or dissolution of Regeneron.
32
Certain Relationships and Related Transactions
![]()
Proposal No. 2: Ratification of Appointment of Independent Registered Public Accounting Firm
The Audit Committee has appointed PricewaterhouseCoopers LLP as the Company's independent registered public accounting firm for the fiscal year ending December 31, 2016. PricewaterhouseCoopers LLP (or its predecessor) has audited the Company's financial statements for the past 27 years.
The board of directors has directed that the appointment of PricewaterhouseCoopers LLP as the Company's independent registered public accounting firm for fiscal year 2016 be submitted for ratification by the shareholders at the Annual Meeting. Shareholder ratification of the appointment of PricewaterhouseCoopers LLP as the Company's independent registered public accounting firm for fiscal year 2016 is not required by the Company's charter documents or otherwise, but is being pursued as a matter of good corporate practice. If shareholders do not ratify the appointment of PricewaterhouseCoopers LLP as the Company's independent registered public accounting firm for fiscal year 2016, the board of directors will consider the matter at its next meeting.
PricewaterhouseCoopers LLP has advised the Company that it will have in attendance at the 2016 Annual Meeting a representative who will be afforded an opportunity to make a statement, if such representative desires to do so, and will respond to appropriate questions presented at the 2016 Annual Meeting.
Information about Fees
Paid to Independent Registered
Public Accounting Firm
Aggregate fees incurred related to services provided to the Company by PricewaterhouseCoopers LLP for the years ended December 31, 2015 and 2014 were:
|
|
2015 | | 2014 | |||
Audit Fees |
$ | 1,721,000 | $ | 1,567,493 | |||
Audit-Related Fees |
| 2,007 | | – | |||
All Other Fees |
| 4,637 | | 4,812 | |||
| | | | | | | | |
Total Fees |
$ | 1,727,644 | $ | 1,572,305 |
Audit Fees. Audit fees in 2015 and 2014 were primarily for professional services rendered for the audit of the Company's financial statements for the fiscal year, including attestation services required under Section 404 of the Sarbanes-Oxley Act of 2002, technical accounting consultations related to the annual audit, and reviews of the Company's quarterly financial statements included in its Form 10-Q filings.
Audit-Related Fees. Audit-related fees in 2015 were for professional services rendered for the review of a benefit plan of a foreign subsidiary of the Company.
All Other Fees. All other fees in 2015 and 2014 were for an annual subscription to a technical accounting database and for professional services rendered to a foreign subsidiary of the Company.
The Audit Committee has adopted a policy regarding the pre-approval of audit and permitted non-audit services to be performed by the Company's independent registered public accounting firm, PricewaterhouseCoopers LLP. The Audit Committee will, on an annual basis, consider and, if appropriate, approve the provision of audit and non-audit services by PricewaterhouseCoopers LLP. The Audit Committee has approved a general provision of $75,000 for accounting advisory and other permissible consulting engagements. Management is responsible for notifying the Audit Committee of the status of accounting advisory and other permissible consulting engagements at regularly scheduled Audit Committee meetings and, if the Audit Committee so determines, the general provision is replenished to $75,000. The Audit Committee did not utilize the de minimis exception to the pre-approval requirements to approve any services provided by PricewaterhouseCoopers LLP during fiscal 2015 and 2014.
The board of directors unanimously recommends a vote FOR ratification of the appointment of PricewaterhouseCoopers LLP as the Company's independent registered public accounting firm for the fiscal year ending December 31, 2016.
33
Proposal No. 2: Ratification of Appointment of Independent Registered Public Accounting Firm
![]()
The Audit Committee Report below shall not be deemed to be "soliciting material" or to be filed with the SEC or subject to Regulation 14A or 14C under the Exchange Act, or to the liabilities of Section 18 of the Exchange Act. Notwithstanding anything to the contrary set forth in any of the Company's previous filings under the Securities Act of 1933, as amended (the "Securities Act"), or the Exchange Act that might incorporate future filings, including this proxy statement, in whole or in part, the Audit Committee Report below shall not be incorporated by reference into any such filings.
We have reviewed the audited financial statements of the Company for the year ended December 31, 2015, which are included in the Company's Annual Report on Form 10-K for the fiscal year ended December 31, 2015, and met with both management and PricewaterhouseCoopers LLP, the Company's independent registered public accounting firm, to discuss those financial statements. The Audit Committee has discussed with the Company's independent registered public accounting firm the matters required to be discussed by Auditing Standard No. 16, as adopted by the Public Company Accounting Oversight Board (the "PCAOB"), which include, among other items, matters related to the conduct of the audit of the Company's financial statements. The Audit Committee also discussed with the independent registered public accounting firm their independence relative to the Company and received and reviewed the written disclosures and the letter from the independent registered public accounting firm required by PCAOB Rule 3526 (Communication with Audit Committees Concerning Independence).
Based on the foregoing discussions and review, the Audit Committee recommended to the board of directors that the audited financial statements of the Company for the year ended December 31, 2015 be included in the Company's Annual Report on Form 10-K for the fiscal year ended December 31, 2015 for filing with the Securities and Exchange Commission.
We have appointed PricewaterhouseCoopers LLP as the Company's independent registered public accounting firm for the fiscal year ending December 31, 2016. This appointment was based on a variety of factors, including PricewaterhouseCoopers LLP's competence in the fields of accounting and auditing.
| The Audit Committee | ||
George L. Sing, Chairman Charles A. Baker Arthur F. Ryan |
34
Audit Committee Report
![]()
Executive Compensation
Compensation Discussion and Analysis |
This Compensation Discussion and Analysis provides information about our 2015 compensation program for the following executive officers (the "Named Officers"), whose compensation is set forth in the Summary Compensation Table
and the other compensation tables included in this proxy statement:
|
Named Officer |
Position |
|
| Leonard S. Schleifer, M.D., Ph.D. | President and Chief Executive Officer | |
| George D. Yancopoulos, M.D., Ph.D. | President, Regeneron Laboratories and Chief Scientific Officer | |
| Robert E. Landry | Senior Vice President, Finance and Chief Financial Officer | |
| Neil Stahl, Ph.D. | Executive Vice President, Research & Development | |
| Robert J. Terifay | Executive Vice President, Commercial |
It is organized into the following sections:
- •
- Section 1 – Summary (page 35)
- •
- Section 2 – Analysis of 2015 Executive Compensation Based on Compensation Objectives (page 40)
- •
- Section 3 – Executive Compensation Process and Considerations (page 49)
- •
- Section 4 – Elements of Executive Compensation (page 54)
Section 1 – Summary |
2015 Performance Overview
We are a fully integrated biopharmaceutical company that discovers, invents, develops, manufactures, and commercializes medicines for the treatment of serious medical conditions. We commercialize medicines for eye diseases, high LDL cholesterol, and a rare inflammatory condition and have product candidates in development in other areas of high unmet medical need, including oncology, rheumatoid arthritis, asthma, atopic dermatitis, pain, and infectious diseases. For more information about our business, please see Part I, Item 1. "Business" and Part II, Item 7. "Management's Discussion and Analysis of Financial Condition and Results of Operations" of our 2015 Annual Report.
2015 was another extraordinary year for Regeneron. Our key accomplishments in 2015 included:
- •
- 47% growth in EYLEA® (aflibercept) Injection global net product sales as
compared to 2014, from $2.775 billion to $4.089 billion.
- •
- 46% growth in our total revenues as compared to 2014, from $2.820 billion to $4.104 billion.
- •
- 19% growth in non-GAAP net income as compared to 2014, from $1.175 billion to
$1.404 billion. (Non-GAAP net income is not a measure calculated in accordance with U.S. Generally Accepted Accounting Principles. See Appendix A for a definition of non-GAAP net income
and a reconciliation of non-GAAP net income to net income.)
- •
- Advances in our EYLEA® franchise, including:
- o
- Regulatory approval of EYLEA® for the treatment of visual impairment
due to macular edema secondary to retinal vein occlusion and the treatment of visual impairment secondary to myopic choroidal neovascularization in the European Union;
- o
- Regulatory approval of EYLEA® for the treatment of diabetic
retinopathy in patients with diabetic macular edema in the United States; and
- o
- Regulatory approval of EYLEA® for the treatment of retinal vein
occlusion in Japan.
- •
- Regulatory approval and launch of Praluent® (alirocumab) Injection, the first FDA-approved drug in a new class of drugs that lower LDL ("bad") cholesterol.
35
Executive Compensation
- •
- Completion of enrollment in the Praluent® ODYSSEY OUTCOMES trial, an
18,000-patient study prospectively assessing the potential of Praluent® to demonstrate cardiovascular benefit.
- •
- Further advances in our late-stage clinical pipeline, which includes sarilumab, an IL-6
receptor antibody for rheumatoid arthritis; dupilumab, an IL-4 receptor-alpha antibody for allergic diseases; fasinumab, an antibody to nerve growth factor (NGF); and REGN2222, an antibody to the
Respiratory Syncytial Virus-F (RSV-F) protein:
- o
- Reported positive Phase 3 data for sarilumab from three Phase 3
studies in patients with rheumatoid arthritis (SARIL-RA-TARGET, SARIL-RA-EASY, and SARIL-RA-ASCERTAIN) and submitted a Biologics License Application for sarilumab with the FDA.
- o
- Reported positive pivotal Phase 2b data for dupilumab in asthma and
completed enrollment of the dupilumab atopic dermatitis Phase 3 studies.
- o
- Entered into a collaboration agreement relating to fasinumab with Mitsubishi
Tanabe Pharma Corporation for Japan, Korea, and nine other Asian countries, excluding China.
- o
- Initiated a Phase 3 clinical study of REGN2222 for Respiratory Syncytial
Virus.
- •
- Continued growth of our clinical development pipeline, as evidenced by the submission of
one Investigational New Drug Application with the FDA in 2015 and 13 product candidates (consisting of one Trap-based and 12 fully-human monoclonal antibody product candidates based on the Company's VelocImmune® technology) in clinical development as of December 31, 2015.
- •
- New global strategic collaboration with Sanofi to discover, develop, and commercialize antibody-based cancer
treatments in the field of immuno-oncology.
- •
- Further important steps to support our current and future growth, including adding two new buildings in the Tarrytown campus providing nearly 300,000 square feet of additional laboratory and office space; significant progress with the construction of a new manufacturing facility in Limerick, Ireland; and increasing headcount on a year-over-year basis by approximately 47% as of December 31, 2015.
- •
- Further progress of Regeneron Genetics Center. As of
December 31, 2015, the Regeneron Genetics Center expanded on its foundational population-based collaboration with Geisinger Health Systems with over a dozen collaborations with academic
institutions, government entities, and health systems, and had achieved the ability to sequence exomes at the rate of 100,000 per year.
- •
- Collaboration agreement with the Biomedical Advanced Research and Development Authority
(BARDA) of the U.S. Department of Health and Human Services to develop, test, and manufacture a monoclonal antibody therapy for the treatment of Ebola virus infection.
- •
- Named one of the two top employers in the global biopharmaceutical industry by Science Magazine for the fifth consecutive year, the fourth most innovative company in the world by Forbes, and one of Fortune Magazine's 100 best places to work.
These and other recent accomplishments have contributed to the creation of significant value for our shareholders. The Company's strong performance is reflected in the appreciation of our stock price, which increased 32%, 217%, and 1554% over the one-, three-, and five-year periods ended December 31, 2015, respectively. This shareholder return places our common stock performance in the 85th, 90th, and 99th percentile, respectively, of all NASDAQ-listed companies with a market capitalization greater than $5 billion in those periods. In addition, our common stock was the 3rd, 4th, and top performing stock in our Peer Group, discussed further in this Compensation Discussion and Analysis section, over the one-, three-, and five-year periods ended December 31, 2015. See "Section 3 – Executive Compensation Process and Considerations – Peer Group" for the definition of our Peer Group and our Biotech R&D Peers.
As illustrated by the chart below, our total shareholder return ("TSR") significantly outperformed both the S&P 500 Index and the S&P Biotechnology Select Industry Index over the past five years.
36
Executive Compensation
Regeneron 5-Year TSR vs. S&P Indices

TSR data reflect total returns (stock price appreciation and, if applicable, reinvested dividends).
Compensation Objectives and Elements
We believe that the leadership of the current executive team has been instrumental to our success in 2015 and prior years, and that an executive compensation program that attracts, motivates, and helps retain key executives, including the Named Officers, is critical to our long-term success. Our executive compensation program is designed to achieve four main objectives:
- •
- pay for performance;
- •
- closely align the interests of shareholders and management;
- •
- strike a balance between short- and long-term perspectives and support our long-term growth prospects; and
- •
- attract and retain highly skilled and talented executives in a competitive marketplace.
To realize these objectives, we utilize five primary compensation elements, which are summarized in the table below and discussed in detail under "Section 4 – Elements of Executive Compensation":
|
Objective | |||||||
| | | | | | | | | |
Compensation Element |
Pay for Performance |
Shareholder Alignment |
Balance between Short- and Long-Term Perspectives |
Market Competitiveness and Retention |
||||
Base Salary |
| | · | · | ||||
Annual Cash Bonus |
· | · | · | · | ||||
Annual Stock Option Awards |
· | · | · | · | ||||
Perquisites and Other Personal Benefits |
| | | · | ||||
Potential Severance Benefits |
| | · | · | ||||
37
Executive Compensation
The Compensation Committee considered each of our compensation objectives in determining the 2015 compensation of our executives, including the Named Officers, as discussed in greater detail for each of these objectives under "Section 2 – Analysis of 2015 Executive Compensation Based on Compensation Objectives." In particular:
- •
- We believe in performance-based compensation and long-term incentives. In
2015, we continued to rely primarily on performance-based compensation, both for our short-term (cash bonus) and long-term incentives (stock option awards). This emphasis on performance-based
compensation (particularly long-term incentives in the form of stock options) has been a consistent part of our philosophy since Regeneron's inception, including prior to the significant appreciation
in Regeneron's stock price that began in early 2011.
- •
- We believe that time-based stock options are inherently performance based, as they provide value to employees
only if there is future stock price appreciation and do not provide any value to employees if the stock price declines below the exercise price. As illustrated by
the charts in "Section 2 – Analysis of 2015 Executive Compensation Based on Compensation Objectives," this emphasis on stock options has resulted in close alignment of our
Chief Executive Officer's compensation in 2015 and over the last five years with the performance of our common stock over those periods:
- o
- Both in 2015 and over the five-year period ended December 31, 2015, the
year-over-year increases in our Chief Executive Officer's compensation were principally attributable to the significant appreciation in our stock price, which increased the reported grant date fair
value of our Chief Executive Officer's stock option awards as determined according to the Black-Scholes model for valuing stock options.
- o
- Over the same periods, the Black-Scholes grant date fair value of stock option grants to our Chief Executive Officer increased less than the appreciation of our stock price, in part because the absolute number of stock options granted to our Chief Executive Officer decreased in the last three years. The number of shares underlying the annual stock option award to our Chief Executive Officer in 2015 was approximately 39% lower than in 2012, while the stock price appreciated 217% over the same period. As a result, the appreciation in the reported value of our Chief Executive Officer's pay was significantly below the appreciation of our stock price, both cumulatively over the five-year period and on a year-over-year basis. This means that the value of our long-term shareholders' investment in Regeneron grew more rapidly than our CEO's pay over those periods.
- o
- To further illustrate this point, over the last five years, our Chief Executive
Officer's total direct compensation, as a percentage of Regeneron's capitalization in the year in which the compensation was awarded, decreased from 0.20% to 0.08%.
- o
- As a result of our emphasis on performance-based compensation, on a relative
basis when compared to our Peer Group, the total direct compensation of our Chief Executive Officer over the last three years was also closely aligned with the performance of our common stock even
when taking into account the reported grant date fair value of our Chief Executive Officer's stock option awards as determined according to the Black-Scholes model.
- •
- We believe in year-over-year consistency in making compensation decisions and in striking a balance between the
dilutive impact of equity grants and the competitiveness of our compensation program. In our compensation decisions, we focus on the number of shares underlying
equity awards relative to the number of basic shares of common stock outstanding, rather than the grant date fair value of the award (as determined according to the Black-Scholes model). We believe
this ownership- and dilution-based approach to awarding stock options provides a better measure of the amount of potential increases in shareholder value that would be shared by the awards and allows
us to evaluate such grants on a consistent basis as compared to other companies and regardless of fluctuations in the price of Regeneron's or other companies' common stock. Further, focusing on the
number of shares and the incremental sharing rate of potential future upside (rather than targeting a specific Black-Scholes grant date fair value) avoids rewarding officers with larger grant sizes
following a decline in our stock price.
- o
- As a percentage of the total basic shares outstanding, the 2015 stock option
award to our Chief Executive Officer was significantly below the 75th percentile of the companies included in the 2015 Radford Global Life Sciences Survey and only slightly above
the 50th percentile (at 0.166% compared to 0.290% and 0.154%, respectively). In addition, this award was below the 50th percentile of our Biotech R&D Peers
(which was 0.183%).
- o
- In 2015, the Compensation Committee reduced the number of shares underlying the annual stock option awards to the Named Officers by 15% compared to 2014 (other than Mr. Terifay's award, which remained at the 2014 level due to his promotion to Executive Vice President, Commercial). This decrease constituted the third consecutive double-digit percentage decrease in the annual grant of stock options to our Named Officers, in each case following outstanding TSR performance. In reducing the size of 2015 annual stock option awards to executives, the Compensation Committee sought to reduce the
38
Executive Compensation
- o
- We continued to pay close attention to our burn rate. Despite the expansive growth of our employee base, which increased by 121% between 2012 and 2015 (from 1,950 full-time employees to 4,303 full-time employees), our burn rate decreased from 5.4% to 4.4% over the same period, and we maintained a three-year burn rate average of 4.1% in 2015. We achieved this reduction through implementing three consecutive double-digit percentage decreases in the number of shares underlying annual stock option awards, without eliminating the broad-based nature of our equity compensation program.
potential dilutive impact of new equity awards without adversely affecting the competitiveness of our executive compensation program, which has successfully motivated our senior management team to deliver high operating performance and shareholder value.
- o
- We believe our approach to equity compensation has helped us to successfully grow and manage employee attrition, as evidenced by our 2015 employee turnover of approximately 6%, which compares favorably to the average employee turnover of approximately 18% for the life sciences sector based on the Fourth Quarter 2015 Radford Global Life Sciences Trends Report.
Highlights of Compensation Policies and Practices
We are committed to maintaining rigorous corporate governance standards, including those related to the oversight of our executive compensation policies and practices. We have compensation policies and practices designed to enhance governance of our executive compensation program and to further our compensation objectives. These policies and practices include:
|
Policy/Practice |
Description |
|
| Independent Compensation Consultant | The Compensation Committee's consultant, Frederic W. Cook & Co., Inc., is retained directly by the Compensation Committee, performs projects at the Compensation Committee's direction, and does not otherwise perform any other consulting or other services for us. | |
Stock Ownership Guidelines |
To further align the interests of senior management with the interests of shareholders and promote a long-term perspective, our directors and senior officers are subject to stock ownership guidelines with the following share ownership targets: • Chairman of the Board and Chief Executive Officer, six times (6x) base salary; • Chief Scientific Officer, three times (3x) base salary; • Executive/Senior Vice Presidents, two times (2x) base salary; and • Non-employee members of the board of directors, six times (6x) the annual retainer. All of our directors and senior officers subject to the stock ownership guidelines have either met their respective share ownership targets or are still within the five-year period for achieving compliance. |
|
Transparent Equity Granting Process and Practices |
Annual stock option awards to eligible employees are made by the Compensation Committee according to a regular, pre-set schedule. The meetings at which such grants are approved are generally scheduled about a year in advance, without regard to the timing of earnings or other major announcements. |
|
Recoupment Policy |
We have a policy regarding recoupment or reduction of incentive compensation for compliance violations that is applicable to our officers, including the Named Officers, and other specified employees. |
39
Executive Compensation
|
Policy/Practice |
Description |
|
Prohibition Against Hedging and Pledging of Our Securities |
We have a policy against hedging and pledging of our securities by our directors and employees, including the Named Officers. |
|
No "Single Trigger" Severance or Vesting Change-in-Control Arrangements for Named Officers |
Our change in control severance plan, equity award agreements with the Named Officers, and employment agreement with our Chief Executive Officer do not contain "single trigger" provisions (i.e., do not provide for cash severance payments or accelerated vesting of equity awards solely on account of a change in control without a termination of employment). |
|
Policy Regarding Excise Tax Gross-Ups |
We have a policy against including excise tax gross-up provisions with respect to payments contingent upon a change in control of Regeneron in contracts, compensatory plans, or other arrangements with the Company's executive officers, including the Named Officers (other than the existing employment agreement with our Chief Executive Officer or any amendments thereto, which we expressly exempted). |
|
| Limited Perquisites | The perquisites and other personal benefits afforded to our Named Officers are limited and are subject to periodic review by the Compensation Committee. | |
| Compensation Committee Oversight; Executive Sessions | Our Compensation Committee regularly meets in executive sessions without members of management present. | |
| Risk Management | Our Compensation Committee regularly reviews our compensation strategy and practices, including an annual review of our compensation-related risk profile, to ensure that our compensation-related risks are not reasonably likely to have a material adverse effect on the Company. |
Section 2 – Analysis of 2015 Executive Compensation Based on Compensation Objectives |
Pay for Performance
We believe in rewarding performance and establishing a strong link between compensation and both individual and corporate performance at all levels of the Company. We also believe that accountability and total compensation potential should generally increase with position and responsibility and that the proportion of the performance-based component of compensation should increase with position and responsibility. Consistent with this view, individuals with greater responsibility and ability to influence our achievement of corporate goals and strategic initiatives generally are targeted with higher levels of cash compensation, but have a higher proportion of their total cash compensation represented by cash bonus, the amount of which is based on individual and/or corporate performance and is, therefore, at risk. Similarly, equity-based compensation is higher for persons with higher levels of responsibility, making a significant portion of their total compensation dependent on long-term stock appreciation and, therefore, long-term corporate performance.
This pay-for-performance philosophy was strongly reflected in the mix of compensation elements, or "pay mix," of our Named Officers in 2015. The following charts display the mix of 2015 compensation (fixed vs. performance-based and long-term vs. short-term) of our Chief Executive Officer and our other Named Officers (excluding our Chief Executive Officer) and the mix of compensation elements of the companies included in the 2015 Radford Global Life Sciences Survey. As shown in these charts, 97% and 96% of the total direct 2015 compensation of our Chief Executive Officer and our other Named Officers, respectively, consisted of performance-based compensation, which exceeds the average percentage of performance-based compensation paid by the companies included in the 2015 Radford Global Life Sciences Survey by 7% and 14%, respectively. Accordingly, based on these metrics, our 2015 executive compensation program was more heavily weighted toward "at risk," performance-based components.
40
Executive Compensation
| Regeneron CEO Pay Mix | Regeneron Other NEO Pay Mix | |
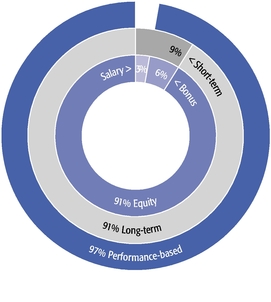
|
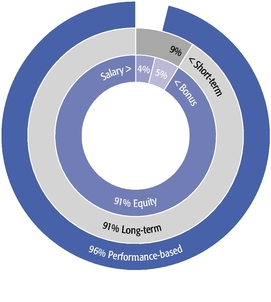
|
|
| Survey CEO Pay Mix | Survey Other NEO Pay Mix | |

|
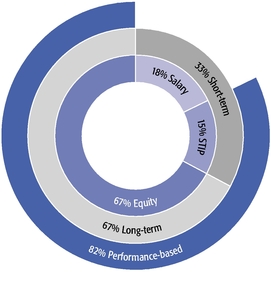
|
Based on 2015 compensation information reported in the Summary Compensation Table on page 61 for Regeneron and on the 2015 Radford Global Life Sciences Survey (comprising U.S. public biotechnology and pharmaceutical companies that have between 800 and 15,000 employees). "Equity" (shown as part of the inner circle in the charts above) reflects the grant date fair value of equity awards; and "STIP" (shown as part of the inner circle in the charts above) consists of bonus and/or other applicable compensation provided under short-term, non-equity incentive plans. "Long-term" compensation (shown as part of the middle circle in the charts above) consists of equity; and "short-term" compensation (shown as part of the middle circle in the charts above) consists of base salary and bonus/STIP. "Performance-based" compensation (shown as part of the outer circle in the charts above) consists of bonus/STIP and equity. Total compensation amounts reflect total direct compensation (total reported compensation, other than amounts reported under "All other compensation").
As shown in the charts above, the Compensation Committee manages the pay mix such that a substantial portion of pay is dedicated to "at risk," performance-based compensation, with that portion to comprise a significantly greater percentage of total direct compensation as compared to the average of the
companies included in the 2015 Radford Global Sciences Survey. The Compensation Committee believes that this mix of pay best aligns the interests of our Named Officers with those of our shareholders over time.
41
Executive Compensation
Shareholder Alignment
We believe that the compensation realized by our leadership team, including the Named Officers, should show a strong relation to the value realized by our shareholders. This principle is reflected both in the pay mix referenced above and in our focus on stock options, as the value realized by the holder of a stock option, if any, is dependent upon, and directly proportionate to, appreciation in the price of our common stock. Since our inception, we have consistently structured our executive compensation based on our belief that stock options naturally align executives with the creation of future shareholder value. We also believe that stock options have been fundamental to our ability to attract, motivate, and retain top talent whose contributions and leadership have driven the Company's achievement of corporate and strategic goals and resulted in a substantial enhancement of shareholder value. Over the one-, three-, and five-year periods ended December 31, 2015, our stock price increased 32%, 217%, and 1554%, respectively. This shareholder return places our common stock performance in the 85th, 90th, and 99th percentile,
respectively, of all NASDAQ-listed companies with a market capitalization greater than $5 billion in those periods. Our use of stock options ensures alignment of the interests of our executives, including the Named Officers, with those of Company shareholders, as our executives not only benefit from our successes, and the resulting appreciation of our stock, but are also impacted by decreases in our stock price.
The following chart compares our TSR over the last five years with the compensation of our Chief Executive Officer over the same period. We do not focus primarily on total direct compensation as a key metric, as it is derived in part from the reported grant date fair value of stock option awards as determined according to the Black-Scholes model. As further discussed below, in assessing stock option awards, we primarily consider the number of shares underlying the awards relative to the number of basic shares of common stock outstanding in order to evaluate such awards on a consistent basis as compared to other companies and regardless of fluctuations in the price of Regeneron's or other companies' common stock.
5-Year CEO Total Direct Compensation vs. Total Shareholder Return
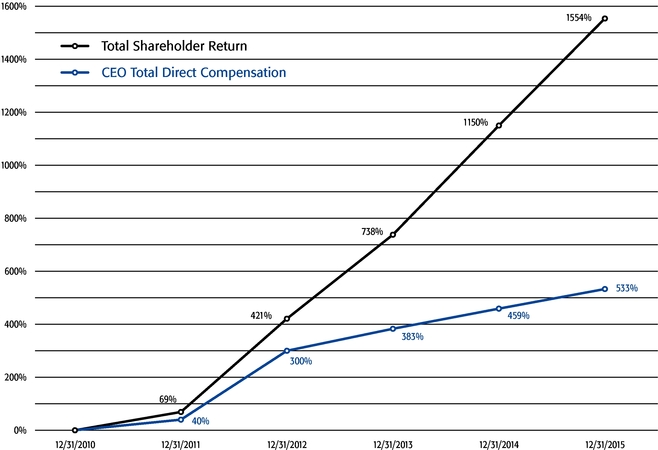
- "CEO
- total direct compensation" means total reported compensation (other than amounts reported under "All other compensation").
42
Executive Compensation
As shown in the table above, compensation awarded to our Chief Executive Officer over the last five years closely tracked our TSR over the same period, and percentage increases in such compensation were considerably smaller than our TSR over the same period (533% increase in total direct compensation compared to 1554% TSR). The overwhelming portion of the increase in our Chief Executive Officer's total direct compensation over this period is directly attributable to the value of the non-cash components of his compensation package, primarily stock option awards valued according to the
Black-Scholes model. Consistent with our objective to align the interests of our executives with the interests of our shareholders, as the price of our common stock appreciated over the last five years, the value of stock option awards, the most significant component of the compensation of our Chief Executive Officer (as well as the other Named Officers), increased as well.
In addition, on a year-over-year basis, percentage increases in our Chief Executive Officer's total direct compensation in the last five years were significantly below the year-over-year appreciation of our stock price, as shown in the chart below.
Year-Over-Year Increase in CEO Total Direct Compensation vs. Total Shareholder Return
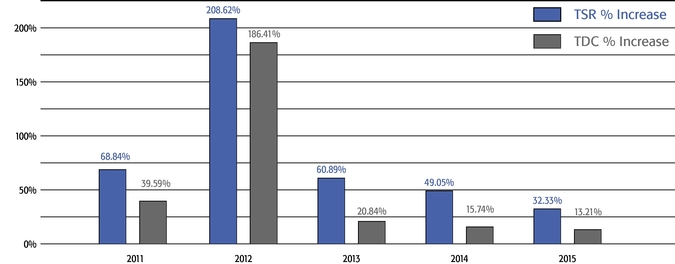
"CEO total direct compensation" means total reported compensation (other than amounts reported under "All other compensation").
43
Executive Compensation
The table below further demonstrates alignment of Regeneron's performance (as determined by its market capitalization) and our Chief Executive Officer's total direct compensation in the last five years. Over this period, our Chief
Executive Officer's total direct compensation, as a percentage of Regeneron's capitalization in the year in which the compensation was awarded, decreased from 0.20% to 0.08%.
CEO Total Direct Compensation as Percentage of Market Capitalization
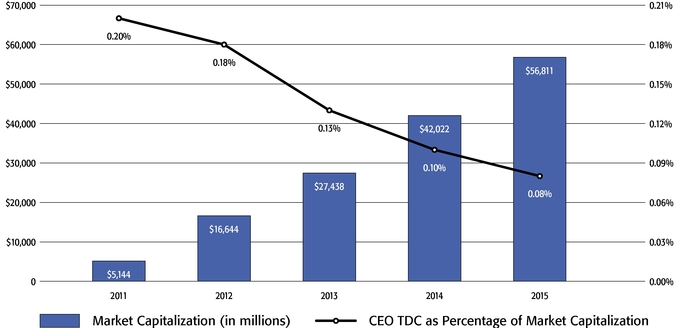
"CEO total direct compensation" means total reported compensation (other than amounts reported under "All other compensation"); and market capitalization is determined based on the number of shares of common stock and Class A stock outstanding as of December 31 of each year.
44
Executive Compensation
We further believe that the compensation awarded to our Chief Executive Officer in recent years showed a strong connection to our TSR performance over the same period both on an absolute basis and relative to the companies in our Peer Group. In addition to being the 4th best performing
company in our Peer Group over the three-year period (as shown in the chart below), we were the 3rd best and the top performing company in our Peer Group over the one- and five-year periods (in each case as measured by TSR).
CEO Reported Pay Percentile versus TSR Percentile

- Compensation information reflects total direct compensation (i.e., total reported compensation, other than amounts reported under "Change in pension value and nonqualified deferred compensation earnings" and "All other compensation") and is based on the 2013 - 2015 compensation information reported in the Summary Compensation Table included in this proxy statement for Regeneron and on the data for the most recently completed three-year periods available as of December 31, 2015 for the companies included in the Peer Group. TSR information is based on 2013 - 2015 TSR for Regeneron and the Peer Group. The shaded area between the two diagonal lines indicates an alignment of pay and performance. See "Section 3 – Executive Compensation Process and Considerations – Peer Group" for more information regarding our Peer Group.
Balance between Short- and Long-Term Perspectives
The Company's compensation program has been designed to strike a balance between short- and long-term compensation in light of the Company's available resources; its growth trajectory; its goal of offering compensation packages that are competitive with those of companies in the Peer Group and the broader biopharmaceutical industry; and its objective to
retain and motivate high-performing employees and closely align their interests with the interests of shareholders. We utilize base salary and annual cash bonuses to compensate our executives over the short term by providing a base level of income and rewarding performance on an annual basis, and we utilize stock options to foster a long-term perspective and reward long-term performance.
45
Executive Compensation
Why stock options? |
|
|
||||
|
Time-based stock options represent the foundation of our compensation philosophy and an integral part of our executive compensation program. Since our inception, we have consistently structured our executive compensation based on our belief that
stock options naturally align executives with the creation of future shareholder value and are the best vehicle to retain and foster the entrepreneurial aspects of our culture. The core group of our senior management (Drs. Schleifer, Yancopoulos, and
Stahl) has been with Regeneron for over 20 years and has been instrumental in driving shareholder value over this period. We discussed our use of time-based stock options as a critical component of our compensation program as part of our 2015
shareholder outreach (discussed in greater detail below) and were again encouraged by the fact that a number of our key shareholders were supportive of time-based stock options and viewed them as an excellent compensation tool for a high-performing,
growth-oriented company. In 2015, as in other recent years, we relied on time-based stock options as the primary vehicle for offering long-term incentives to our employees, including the Named Officers, and for linking compensation and performance. However, we reduced the number of shares underlying the annual stock option awards to the Named Officers by 15% compared to 2014 (other than Mr. Terifay's award, which remained at the 2014 level due to his promotion to Executive Vice President, Commercial). This reduction constituted the third consecutive double-digit percentage decrease in such annual grant. We believe that time-based stock options are inherently performance-based, offer an excellent compensation mechanism for getting our management team to act in ways that ensure the long-term success of Regeneron, and are appropriate and advantageous for the following additional reasons:
• The executive only realizes
value from stock options if the price of the Company's common stock increases and the executive continues to serve through the vesting period. The value is solely tied to an increase in the Company's stock price, aligning the interests of executives
with those of shareholders. |
• The executive has the opportunity to realize value if the price of the Company's common stock continues to increase over the remainder of the ten-year term of the option. Time-based options, therefore, promote the long view and motivate and reward the executive for Company performance not only over the four-year vesting term, but also the ten-year option term.
• Compared to full-value awards, stock options provide for greater leverage and, therefore, alignment of employee incentives with those of our shareholders. They also amplify downside risk, as they do not provide any value to the holder if the stock price declines below the exercise price (determined as of the date of grant).
• Stock options have played an important role in our success and have helped us to conserve the Company's cash.
• Stock options are highly valued by employees, particularly in our industry. They have helped us attract and retain entrepreneurial employees and foster an ownership culture, which we believe has been part of the Company's formula for success.
• Time-based stock options are understood by employees and are straightforward to administer. We also believe that they are less likely to cause employees to pursue attainment of a particular performance metric at the expense of value creation for our shareholders generally.
• Stock options generally qualify as performance-based compensation for purposes of the deduction limit of Section 162(m) of the Internal Revenue Code, and all amounts realized by our Named Officers upon exercise of their stock options are expected to be fully deductible by us. |
|
|||
46
Executive Compensation
As shown in the charts under "– Pay for Performance" above, 91% of the total direct compensation of our Chief Executive Officer and our other Named Officers in 2015 consisted of long-term compensation, which exceeds the corresponding average percentage for the companies included in the 2015 Radford Global Life Sciences Survey by 18% and 24%, respectively. Based on these metrics, our compensation structure is more heavily weighted toward long-term components versus short-term pay, thus promoting a long-term perspective among our executives.
Despite the reductions in the annual grant of stock options to our Named Officers described above, the grant date fair value (as determined according to the Black-Scholes model for valuing stock options) of the stock options granted in 2015 is higher than the Black-Scholes grant date fair value of the larger number of stock options granted to Named Officers in prior years. This increase in the Black-Scholes value of annual stock option awards (and the corresponding increase in the value of total compensation for the Named Officers in 2015) is attributable largely to the higher market value of our common stock on December 16, 2015, the date of these option grants, compared to prior years. On December 16, 2015, the average of the high and low sales price per share of our common stock as quoted on the NASDAQ Global Select Market (used for calculating the grant date fair value) was $555.67, as compared to $399.66 on December 31, 2014 and $270.43 on December 13, 2013, the respective 2014 and 2013 grant dates.
In line with the goal of maintaining year-over-year consistency in making compensation decisions (regardless of stock price fluctuations), the Compensation Committee believes that the grant date fair value of the option award (as determined according to the Black-Scholes model for valuing stock options) is not the most appropriate measure for evaluating the grant. Rather, the Compensation Committee focuses more on the percentage of Regeneron's potential future share price appreciation that is shared by means of the award with the executive, expressed as the number of shares underlying the option award relative to the number of outstanding shares.
The following chart shows the decrease in the 2012 - 2015 annual stock option awards to our Chief Executive Officer both based on the number of shares underlying the awards and as
a percentage of the applicable number of outstanding shares of Regeneron common and Class A stock:
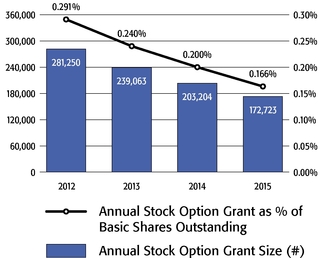
Share percentages are based on 96.6 million, 99.4 million, 101.7 million, and 104.1 million of shares (in each case consisting of common stock and Class A stock) outstanding as of October 12, 2012, October 28, 2013, October 16, 2014, and October 16, 2015, respectively, as reported in Regeneron's Quarterly Report on Form 10-Q for the third quarter of the applicable year.
In recent years, we have experienced significant growth in employees to support our research & development and commercialization efforts. From 2012 to 2015, we increased our employee base by approximately 121%. While the number of recipients of stock awards continued to increase given our practice of making initial stock option awards to all new employees and annual stock option awards to eligible employees whose performance is determined to merit an annual grant, we managed our utilization of stock awards judiciously, reducing our burn rate from the 2012 level and achieving a three-year burn rate average of 4.1% in 2015, in line with our current burn rate goal of 4%. We achieved this reduction through implementing three consecutive double-digit percentage decreases in the number of shares underlying annual stock option awards, without eliminating the broad-based nature of our equity compensation program.
47
Executive Compensation
Regeneron Stock Utilization vs. Headcount
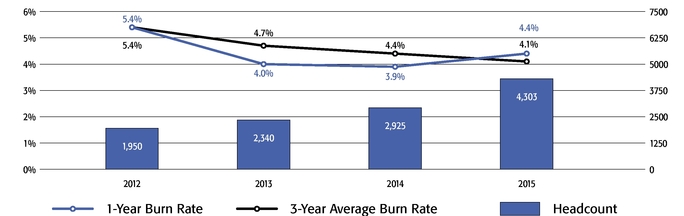
Burn rate calculated by dividing the number of shares subject to equity awards (time-based and performance-based stock options and restricted stock) granted during the year by the weighted-average number of shares of common stock (including unvested restricted stock) and Class A stock outstanding during the year. A multiplier of 2 is applied to restricted stock awards. Headcount numbers based on the number of employees as of December 31 of the applicable year.
In addition, in the last three years when we implemented the stock option award reductions described above, we managed either to decrease our burn rate or to keep the percentage increase in our burn rate significantly below the percentage increase in the number of our employees. The 2015 increase in our burn rate compared to the prior year is wholly attributable to the rapid hiring of new key employees (as evidenced by a 47% increase in the number of employees over the same period), not increased sharing with our Named Officers (whose stock options grants declined).
Regeneron Year-over-Year Change in
Burn Rate and Headcount

Burn rate and headcount calculated as noted above.
Market Competitiveness and Employee Retention
In determining the appropriate size of stock option awards to executives, including the Named Officers, the Compensation Committee primarily considers the number of shares underlying the awards relative to the number of basic shares of common stock outstanding and not the grant date fair value of the award (as determined according to the Black-Scholes model). The Compensation Committee is therefore able to evaluate such grants on a consistent basis as compared to other companies and regardless of fluctuations in the price of Regeneron's or other companies' common stock. Further, focusing on the number of shares and the incremental sharing rate of potential future upside (rather than targeting a specific Black-Scholes grant date fair value) avoids rewarding officers with larger grant sizes following a decline in our stock price. The following chart displays the 2015 annual stock option award to our Chief Executive Officer as a percentage of the total basic shares outstanding, as compared to the 75th percentile and the 50th percentile of the companies included in the 2015 Radford Global Life Sciences Survey, showing that the size of his 2015 annual award was significantly below the 75th percentile and only slightly above the 50th percentile. It also shows that this award was below the 50th percentile of our Biotech R&D Peers.
48
Executive Compensation
CEO Grant as Percentage of Basic Shares Outstanding

- Based on 2015 stock option award information reported in the Summary Compensation Table included in this proxy statement for Regeneron; data relating to grants of equity awards from the 2015 Radford Global Life Sciences Survey (comprising U.S. public biotechnology and pharmaceutical companies that have between 800 and 15,000 employees); and 2014 information available for Regeneron's Biotech R&D Peers (as defined in "Section 3 – Executive Compensation Process and Considerations – Peer Group"). The share information is based on 104.1 million shares (consisting of common stock and Class A stock) of Regeneron outstanding as of October 16, 2015 (as reported in Regeneron's Quarterly Report on Form 10-Q for the quarterly period ended September 30, 2015) and on the number of shares outstanding reported in the 2015 Radford Global Life Sciences Survey for each of the companies included in the survey.
We believe that the structure of our executive compensation program encourages innovation, attracts talent with entrepreneurial spirit, increases employee engagement, and improves employee retention and the continuity of executive leadership during a critical period of growth. We regard our stock option grants as a key employee and executive retention tool. As stock option awards to our employees vest over time, generally over four years, they provide an incentive for the employee to continue to contribute to our success. Historically,
stock options have helped us maintain a motivational level of total compensation for our Named Officers and other eligible employees, while conserving the Company's cash. We believe that this has helped us to successfully grow and manage employee attrition, as evidenced by our 2015 employee turnover of approximately 6%, which compares favorably to the average employee turnover of approximately 18% for the life sciences sector based on the Fourth Quarter 2015 Radford Global Life Sciences Trends Report. In addition, stock options have allowed us to attract and retain entrepreneurial employees and foster an ownership culture. Moreover, granting stock options as long-term incentives to executives is standard practice in our industry and is an important part of our effort to attract, retain, and motivate high-quality talent.
In light of these considerations, we continued our practice of annual stock option grants in 2015, although we again (for the third consecutive year) reduced the number of shares underlying the annual stock option awards to the Named Officers as described above to reduce the potential dilutive impact of executive awards without adversely affecting the effectiveness of our executive compensation program, which has successfully motivated our senior management team to deliver high operating performance and shareholder value. In light of the fierce competition for talent in our industry, we believe that it is important to continue to motivate our existing employees, including the Named Officers, through stock option awards and to ensure that they share in the success of Regeneron.
We provide our Named Officers with a limited number of perquisites and other personal benefits. These benefits, which are described in greater detail under "Section 4 – Elements of Executive Compensation – Perquisites and Other Personal Benefits" below, are periodically reviewed by the Compensation Committee.
The Compensation Committee is responsible for overseeing the Company's general compensation objectives and programs. The Compensation Committee evaluates the performance of our Named Officers and approves compensation for the Named Officers (in the case of the Chief Executive Officer, subject to first obtaining the approval of the non-employee members of the board of directors). The Compensation Committee operates under a written charter adopted by the board of directors and regularly reviews and reassesses the adequacy of its charter. A copy of the current charter is available on our website at www.regeneron.com under the "Corporate Governance" heading on the "Investors & Media" page.
Members of our senior management play a significant role in the overall executive compensation process and assess performance of other officers. They also recommend, for Compensation Committee approval, salary, bonus, and stock option grant budgets for non-officers and make specific recommendations for salary increases, bonuses, and equity award grants for other officers. For our Named Officers (other than our Chief Executive Officer), recommendations to the Compensation Committee regarding their compensation are made by, or with the approval of, our Chief Executive Officer, who also evaluates their performance. Our Chief Executive Officer's performance is evaluated directly by the Compensation Committee based on our overall corporate performance
49
Executive Compensation
against annual goals that are approved by the board of directors at the beginning of each year, as discussed in more detail below.
The Compensation Committee has the sole authority to retain, at our expense, one or more third-party compensation consultants to assist the Compensation Committee in performing its responsibilities and to terminate the services of the consultant if the Compensation Committee deems it appropriate. In 2015, the Compensation Committee utilized the services of Frederic W. Cook & Co. to assist it in fulfilling its responsibilities. In order to maintain its independence, Frederic W. Cook & Co. was retained directly by the Compensation Committee and performed projects at the Compensation Committee's direction. The Compensation Committee's consultant reviews management recommendations on compensation plans, budgets, and strategies and advises the Compensation Committee on regulations and trends in executive compensation nationally and specifically in the pharmaceutical and biopharmaceutical industries. The Compensation Committee's consultant provides comparative compensation information for our Chief Executive Officer and other senior executives (using the Peer Group and other compensation data as described below), reviews senior management's compensation recommendations for other officers, including the other Named Officers, and provides general advice to the Compensation Committee on compensation matters, including facilitating the articulation and periodic review of the Company's compensation philosophy.
Annual salaries for the following year and year-end bonuses and stock option awards or other year-end equity awards for all employees are determined in December of each year based on Company and individual performance, as well as other factors, including compensation trends among our Peer Group and in the biotechnology industry in general. The 2015 salaries and 2014 year-end bonuses and stock option awards for our Named Officers were established by the Compensation Committee in December 2014. In November and December 2015, the Compensation Committee reviewed the performance of each of the Named Officers and presented its recommendations for 2016 salaries and 2015 year-end bonuses and equity awards for the Named Officers to the non-employee members of the board of directors for concurrence. With respect to our Chief Executive Officer, this process is formalized in the charter of the Compensation Committee, which specifies that the Compensation Committee is to annually present the proposed annual compensation of the Chief Executive Officer to the non-employee members of the board of directors for approval.
For purposes of setting compensation of our Chief Executive Officer, our Chief Scientific Officer, the other Named Officers, and other senior executives, we use comparative compensation information from a relevant peer group of companies ("Peer Group"). The companies in the Peer Group are selected by the Compensation Committee, with the assistance of Frederic W. Cook & Co., based on factors including, but not limited to, market capitalization, geographic location, number of employees, therapeutic focus, research and development expenditures, stage of development, total revenues, and product sales. The Company's trailing revenue size, number of employees, operating earnings, and market capitalization are all nearly in the middle of the range of the current Peer Group companies. The Peer Group is also meant to provide a representative sample of companies with which we compete for talent. The Compensation Committee periodically reassesses the composition of the companies within the Peer Group and makes changes as appropriate, taking into account factors such as changes in the Company's market capitalization and merger-and-acquisition activity impacting the existing Peer Group companies. In September 2015, the Compensation Committee approved a new Peer Group based on the recommendation of its compensation consultant, Frederic W. Cook & Co.. The changes to the Peer Group were as follows: (i) elimination of Cubist Pharmaceuticals, Inc. in light of the fact that it had been acquired by Merck & Co., Inc.; (ii) elimination of Seattle Genetics, Inc. due to its market capitalization being significantly below Regeneron's; and (iii) addition of Alkermes plc and Alnylam Pharmaceuticals, Inc., both of which are biopharmaceutical companies with a business model the Compensation Committee considered similar to Regeneron's. In approving the new Peer Group, the Compensation Committee also took into account that, as of the approval date, Regeneron was the median company in the Peer Group based on market capitalization, revenues for the last four completed quarters, and the then-available reported number of employees, as shown in the table below.
50
Executive Compensation
Market Capitalization (in millions) |
Last Four Quarter |
| |||||||||||||||||
| | | | | | | | | | | | | | | | | | | | |
As of 8/31/2015 |
12-Month Average |
Revenue (in millions) |
Employees* |
||||||||||||||||
Gilead |
$ | 154,201 | Gilead | $ | 157,984 | Gilead | $ | 29,194 | Lilly | | 39,135 | ||||||||
Amgen |
$ | 115,087 | Amgen | $ | 119,866 | AbbVie | $ | 20,986 | AbbVie | | 28,000 | ||||||||
AbbVie |
$ | 103,306 | AbbVie | $ | 102,021 | Amgen | $ | 20,765 | BMS | | 25,000 | ||||||||
BMS |
$ | 99,166 | BMS | $ | 101,620 | Lilly | $ | 19,620 | Amgen | | 17,900 | ||||||||
Celgene |
$ | 93,347 | Celgene | $ | 91,033 | BMS | $ | 16,383 | Biogen | | 7,550 | ||||||||
Lilly |
$ | 87,343 | Biogen | $ | 84,590 | Biogen | $ | 10,296 | Gilead | | 7,000 | ||||||||
Biogen |
$ | 69,916 | Lilly | $ | 78,328 | Celgene | $ | 8,426 | Celgene | | 6,012 | ||||||||
Regeneron |
$ | 54,257 | Regeneron | $ | 46,358 | Regeneron | $ | 3,396 | Regeneron | | 3,320 | ||||||||
Alexion |
$ | 38,942 | Alexion | $ | 37,041 | Alexion | $ | 2,391 | Alexion | | 2,273 | ||||||||
Vertex |
$ | 31,198 | Vertex | $ | 29,201 | United Therapeutics | $ | 1,351 | Vertex | | 1,830 | ||||||||
Incyte |
$ | 20,970 | BioMarin | $ | 17,132 | BioMarin | $ | 861 | BioMarin | | 1,681 | ||||||||
BioMarin |
$ | 20,820 | Incyte | $ | 15,294 | Alkermes | $ | 648 | Alkermes | | 1,300 | ||||||||
Alkermes |
$ | 8,898 | Alkermes | $ | 8,873 | Incyte | $ | 644 | United Therapeutics | | 740 | ||||||||
Alnylam |
$ | 8,708 | Alnylam | $ | 8,535 | Vertex | $ | 628 | Incyte | | 588 | ||||||||
United Therapeutics |
$ | 6,859 | United Therarapeutics | $ | 7,103 | Alnylam | $ | 62 | Alnylam | | 256 | ||||||||
75th Percentile |
$ | 100,201 | | $ | 101,720 | | $ | 19,906 | | | 19,675 | ||||||||
Median |
$ | 54,429 | | $ | 57,685 | | $ | 5,409 | | | 4,143 | ||||||||
25th Percentile |
$ | 17,839 | | $ | 13,689 | | $ | 647 | | | 1,160 | ||||||||
REGN Percentile Rank |
| 50th | | | 48th | | | 47th | | | 48th | ||||||||
| | | | | | | | | | | | | | | | | | | | |
Source: Standard & Poor's Compustat.
- *
- Based on information reported in the companies' most recent Annual Reports on Form 10-K available in September 2015.
The Peer Group utilized in 2015 (approved by the Compensation Committee in September 2015 as noted above) consists of the following 14 companies:
| | | | | |
| AbbVie Inc. | Biogen Inc. * | Gilead Sciences, Inc. * | ||
| Alexion Pharmaceuticals, Inc. * | BioMarin Pharmaceutical Inc. * | Incyte Corporation * | ||
| Alkermes plc * | Bristol-Myers Squibb Company | United Therapeutics Corporation * | ||
| Alnylam Pharmaceuticals, Inc. * | Celgene Corporation * | Vertex Pharmaceuticals, Inc. * | ||
| Amgen Inc. | Eli Lilly and Company | | ||
| | | | | |
- *
- Regeneron's Biotech R&D Peer.
In making its compensation decisions in December 2015, the Compensation Committee used data from publicly filed proxy statements of the companies in the Peer Group (as compiled by its compensation consultant) to review each element of compensation of our Named Officers against their peers in the Peer Group as well as their total annual compensation in relation to the Peer Group, while taking into account various factors such as the executive's performance, past compensation history, experience, and the role in the Company's success. We use Peer Group data as a point of reference for measurement, but Peer Group data do not represent the only factor considered and there is no targeted pay level percentile. Further, in its review of the Peer Group data, the Compensation Committee also considers the practices of the 10-company sub-group of peers viewed as having businesses and drug discovery cultures that are most similar to Regeneron's, with similarly-sized employee bases (marked with an asterisk
in the table above and referred to as "Biotech R&D Peers"). The Compensation Committee retains discretion in determining the nature and extent of the use of Peer Group data. In addition, in 2015 management and the Compensation Committee reviewed compensation data for biotechnology companies from the 2015 Radford Global Life Sciences Survey (comprising U.S. public biotechnology and pharmaceutical companies that have between 800 and 15,000 employees) to obtain a general understanding of current compensation practices and to assess overall competitiveness of our compensation program.
Compensation Consultant Independence
In accordance with applicable listing standards of the NASDAQ Stock Market LLC and SEC rules, the Compensation Committee evaluated in 2015 the independence of Frederic W. Cook & Co.,
51
Executive Compensation
including by taking into consideration the following factors:
- •
- the fact that Frederic W. Cook & Co. did not provide any other services to the Company;
- •
- the amount of fees received from the Company by Frederic W. Cook & Co. as a percentage of its revenue;
- •
- the policies and procedures of Frederic W. Cook & Co. designed to prevent conflicts of interest;
- •
- the absence of any significant business or personal relationship between Frederic W. Cook & Co. and any member of the
Compensation Committee;
- •
- the fact that Frederic W. Cook and the representative of Frederic W. Cook & Co. to the Company did not own any stock of
the Company; and
- •
- the absence of any material business or personal relationship between Frederic W. Cook & Co. and any executive officer of the Company.
The Compensation Committee's evaluation was based in part on a representation letter from Frederic W. Cook & Co.. On the basis of this evaluation, the Compensation Committee concluded that the engagement of Frederic W. Cook & Co. did not raise any conflicts of interest.
To further align the interests of senior management with the interests of shareholders and promote a long-term perspective, the board of directors has adopted stock ownership guidelines for members of senior management, including the Named Officers, and the members of the board of directors. The guidelines are reviewed periodically by the Corporate Governance and Compliance Committee. Pursuant to these guidelines, these individuals are expected to meet share ownership targets that are determined based on their position and their base salary. The share ownership targets are as follows:
- •
- Chairman of the Board and Chief Executive Officer, six times (6x) base salary;
- •
- Chief Scientific Officer, three times (3x) base salary;
- •
- Executive/Senior Vice Presidents, two times (2x) base salary; and
- •
- non-employee members of the board of directors, six times (6x) the annual retainer.
Covered individuals who do not currently meet these guidelines have five years from becoming subject to the policy to reach their target. Members of senior management who are
hired or promoted, and directors who join the board of directors, have five years from such date to reach their target. Shares held directly, shares held indirectly through our 401(k) Savings Plan, shares held in trust, and shares held by immediate family members residing in the same household are included in determining an individual's share ownership. Unexercised stock options and unvested restricted stock are not considered owned for purposes of these guidelines. All directors and officers have either met their respective share ownership targets or are still within the five-year period for achieving compliance.
Say-on-Pay Response
Our shareholders are provided with an opportunity to cast a non-binding, advisory vote every three years on our executive compensation program. Our shareholders most recently had the opportunity to cast advisory say-on-pay votes at our annual shareholder meeting held in June 2014, at which approximately 62% of the votes cast supported the advisory vote proposal on our executive compensation program. Management and the Compensation Committee carefully considered the results of the most recent say-on-pay vote. Senior members of our management as well as the Chairman of the Compensation Committee subsequently spent a significant amount of time speaking with some of our key shareholders about executive compensation and corporate governance. As part of our 2014 engagement effort, we discussed these issues with shareholders collectively representing approximately 47% of the shares of common stock outstanding as of December 31, 2014 (excluding shares held our directors and executive officers and Sanofi). Following the 2014 discussions, we implemented several changes to our executive compensation program and continued the implementation of our existing compensation and governance initiatives. A summary of the relevant changes and initiatives adopted after the most recent say-on-pay vote is provided below.
- •
- We reduced the number of shares underlying the 2014 annual stock option awards to the Named Officers by an average of 16% compared to
the prior year (without giving effect to a grant to our Chief Financial Officer, who did not receive an annual stock option award in 2013 because he joined the Company in September 2013).
- •
- We eliminated certain perquisites of our Chief Executive Officer and our Chief Scientific Officer we considered no longer consistent with our overall compensation program, including, in the case of our Chief Executive Officer, a tax gross-up related to legal, tax, and financial planning advisory services.
52
Executive Compensation
- •
- We adopted a policy against including excise tax gross-up provisions with respect to payments contingent upon a change in control of Regeneron in contracts, compensatory plans, or other arrangements with the Company's executive officers, including the Named Officers (other than the existing employment agreement with our Chief Executive Officer or any amendments thereto, which we expressly exempted).
- •
- We provided additional information regarding compensation decisions and our compensation philosophy in the Compensation Discussion and Analysis of our 2015 proxy statement to better communicate to our shareholders what drives compensation decisions at Regeneron.
We will continue to consider the outcome of our past and future advisory vote results. Our shareholder engagement efforts and implementation of compensation and governance initiatives continued in 2015, as described further below under "2015 Shareholder Outreach."
2015 Shareholder Outreach
We have instituted an ongoing shareholder outreach program through which we seek input from our institutional investors and other shareholders regarding our executive compensation and other governance practices, and implement appropriate changes based on this input. We value shareholder views and insights and believe that constructive and meaningful dialogue allows us to develop broader relationships with investors over the long-term and builds informed relationships that promote transparency and accountability. We continued our shareholder outreach efforts in 2015 and engaged in discussions with shareholders collectively representing approximately 47% of the shares of common stock outstanding as of December 31, 2015 (excluding shares held our directors and executive officers and Sanofi). Below is a summary of recent changes we have adopted based on shareholder feedback and other relevant considerations:
| What We Heard | What We Did | When Implemented | ||
| Concern about size of NEO equity awards | Implemented another double-digit percentage decrease in the number of shares underlying the annual stock option awards to our CEO, CSO, CFO, and EVP, Research & Development | December 2015 (earlier reductions implemented in December 2013 and December 2014) | ||
| | | | | |
| Concern about burn rate | Implemented across-the-board decrease in the number of shares underlying employee annual stock option awards; maintained a three-year burn rate average of 4.1% despite a 121% increase in the number of employees over the same period | December 2015 (earlier reductions implemented in December 2013 and December 2014) | ||
| | | | | |
| Continue to implement corporate governance best practices | Adopted majority voting standard in the election of directors | January 2016 | ||
| | | | | |
| |
Consideration of Risk in Company Compensation Policies
The Compensation Committee regularly reviews the Company's compensation and benefits programs, including its executive compensation program and its incentive based compensation programs for commercial personnel. Our compensation and governance-related policies are further enhanced by our stock ownership guidelines applicable to our senior officers and our policy regarding recoupment or reduction of incentive compensation of our officers and other specified employees for compliance violations, as well as a policy against hedging and pledging of our securities by our directors and employees, including the Named Officers. We have also adopted a policy against including excise tax gross-up provisions with respect to payments contingent upon a change in control of Regeneron in
contracts, compensatory plans, or other arrangements with the Company's executive officers, including the Named Officers (other than the existing employment agreement with our Chief Executive Officer or any amendments thereto, which we expressly exempted). These policies evidence Regeneron's continued commitment to robust corporate governance and are meant to reduce compensation-related risks and ensure greater alignment of the interests of our employees, including the Named Officers, and those of the Company and our shareholders.
We believe that the Company's programs balance risk and potential reward in a manner that is appropriate to the Company's circumstances and in the best interests of the Company's shareholders over the long term. We also believe that
53
Executive Compensation
the Company's compensation and benefits programs do not create risks that are reasonably likely to have a material adverse effect on the Company.
Tax Implications
Section 162(m) of the Internal Revenue Code limits the deductibility for federal income tax purposes of compensation in any year paid to the Chief Executive Officer and the other Named Officers (other than the Chief Financial Officer) to the extent such compensation exceeds $1 million and does not qualify as "performance-based" compensation as defined under Section 162(m) of the Internal Revenue Code. The Company has adopted (and the shareholders approved at the 2015 annual shareholder meeting) the Regeneron Pharmaceuticals, Inc. Cash Incentive Bonus Plan. Awards under the Plan (as well as awards under the previously approved Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan) may, but
are not required to, be subject to the attainment of performance goals in order to qualify for this performance-based compensation exception. The Compensation Committee has implemented the Cash Incentive Bonus Plan for annual cash bonuses of the Named Officers in respect of performance in 2016.
The Compensation Committee takes into account the deductibility of compensation in determining Named Officer compensation. However, the Compensation Committee reserves the right to use its judgment to authorize compensation payments that are not deductible, such as when the Compensation Committee believes that such payments are necessary to maintain the flexibility needed to attract talent, promote executive retention, or reward performance, or as required to comply with the Company's contractual commitments. As noted above, compensation attributable to stock options generally qualifies as "performance-based" compensation and, as such, receives favorable treatment under Section 162(m) of the Internal Revenue Code.
Section 4 – Elements of Executive Compensation |
Our executive compensation program currently has five components:
- •
- base salary;
- •
- annual cash bonus;
- •
- annual equity awards, typically consisting of stock options;
- •
- certain perquisites and other personal benefits; and
- •
- potential severance benefits.
Base Salary
We provide the Named Officers and other employees with base salary to compensate them for services rendered during the fiscal year and provide them with a regular monthly income. In determining base salaries for our Named Officers, the Compensation Committee considers the executive's scope of responsibilities, experience, annual performance, and future potential or role in future success. The Compensation Committee also considers base salaries for comparable positions in our geographic region, competitive salary practices of companies in the Peer Group and the broader biopharmaceutical industry, and annual inflation levels.
For each of 2015 and 2016, each of the Named Officers received a merit increase of 3.5% of his 2014 and 2015 base salary, respectively (which were in addition to the other increases discussed for the Named Officers below). In addition, for 2015, Dr. Schleifer received a base salary increase of
$91,300, which aligned his base salary with that of the median of the companies included in the 2014 Radford Global Life Sciences Survey and below the median of the Peer Group (as in effect in 2014), and Dr. Yancopoulos received a base salary increase of $77,600 to set his base salary at 85% of Dr. Schleifer's. Dr. Yancopoulos's cash compensation, including base salary, is set at 85% of Dr. Schleifer's, which we believe is more appropriate for him in light of the absence of meaningful comparative data for similarly situated executives. Further, for 2015, Dr. Stahl received a $39,200 base salary increase in connection with his promotion to Executive Vice President, Research and Development and Mr. Terifay received a $50,000 base salary increase, in each case to better align his base salary with relevant industry comparative information. For 2016, Dr. Stahl and Mr. Landry received a $50,000 base salary increase each in order to better align their base salaries with relevant industry comparative information. The determinations regarding the 2015 and 2016 merit increases were based on broad-based national data for high-performing, larger biotechnology companies. In each of 2015 and 2016, the base salaries of the Named Officers were set at or below the median of the Peer Group (other than with respect to Dr. Yancopoulos, whose cash compensation, including base salary, is set at 85% of Dr. Schleifer's (as noted above)).
54
Executive Compensation
Annual Cash Bonus
It has been our practice to offer annual cash bonus opportunities to our Named Officers. The Compensation Committee focuses exclusively on our overall corporate performance when determining the annual cash bonus for our Chief Executive Officer and Chief Scientific Officer. The cash bonuses of the other Named Officers are based both on overall corporate performance and their individual contributions and performances during the year. We historically had no formal bonus plan and the award of any bonus to a Named Officer was based on an assessment of corporate and, in the case of Named Officers other than Dr. Schleifer, individual performance. We believe that this approach, rather than working from a rigid bonus formula or plan, was beneficial given the growth trajectory of the Company and its rapid transformation over the past few years from a development-stage company to a fully integrated, commercial-stage biopharmaceutical company. In 2015, the Company adopted, and the shareholders approved, the Regeneron Pharmaceuticals, Inc. Cash Incentive Bonus Plan. The Compensation Committee has implemented the Cash Incentive Bonus Plan for annual cash bonuses of the Named Officers in respect of performance in 2016.
The 2015 cash bonus target for the Chief Executive Officer was 100% of his base salary. In December 2015, the board of directors approved an increase in the Chief Executive Officer's cash bonus target to 120% of his base salary to bring Dr. Schleifer's target annual cash compensation to the median of the companies included in the 2015 Radford Global Life Sciences Survey. For 2016, Dr. Schleifer's target cash compensation remained below the median of the Peer Group. Consistent with Regeneron's historical practice, the increase was given effect in the calculation of Dr. Schleifer's cash bonus paid in respect of 2015. The Chief Executive Officer
recommended 2015 target bonuses for the other Named Officers, which were reviewed and approved by the Compensation Committee. In December 2015, the Compensation Committee increased the cash bonus target for Mr. Landry and Mr. Terifay to 50% and 60% of their respective base salaries; consistent with Regeneron's historical practice, these increases were given effect in the calculation of their cash bonuses paid in respect of 2015. In determining the cash bonus targets for 2015 for Mr. Landry, Dr. Stahl, and Mr. Terifay, the Compensation Committee took into consideration the compensation of similarly situated executive officers at companies in the Peer Group and, in the case of Mr. Terifay, also his promotion to Executive Vice President, Commercial.
In determining the cash bonus target for Dr. Yancopoulos, the Compensation Committee took into consideration the importance of his scientific leadership as President of Regeneron Laboratories and Chief Scientific Officer and the significant contributions he has made to the success of the Company and, specifically, to the discovery and development of the Company's commercial products, its pipeline of internally developed product candidates, and its platform technologies.
The Compensation Committee determined that for Dr. Yancopoulos there were no meaningful comparative data relating to similarly situated executives and that his base salary, cash bonus, and annual stock option awards would be set at 85% of Dr. Schleifer's. The 2015 cash bonus target for Dr. Yancopoulos was 120% of his base salary (increased in connection with the increase of Dr. Schleifer's cash bonus target as described above). These cash bonus targets are consistent with the Company's emphasis on performance-based compensation and are set at or below the median of the Peer Group (other than with respect to Dr. Yancopoulos, whose cash compensation, including cash bonus, is set at 85% of Dr. Schleifer's).
In December 2015, our Named Officers were awarded the following cash bonuses, which were paid in January 2016.
Named Officer |
|
Bonus Target (as percentage of base salary) |
|
Personal Performance Multiplier |
|
Company Performance Multiplier |
|
Total Cash Bonus ($) |
||||||
Leonard S. Schleifer, M.D., Ph.D. |
| 120% | | n/a | | 2.0 | | 2,880,000 | | |||||
George D. Yancopoulos, M.D., Ph.D. |
| 120% | | n/a | | 2.0 | | 2,448,000 | | |||||
Robert E. Landry |
| 50% | | 1.5 | | 2.0 | | 465,750 | 1 | | ||||
Neil Stahl, Ph.D. |
| 60% | | 1.5 | | 2.0 | | 594,000 | 1 | | ||||
Robert J. Terifay |
| 60% | | 1.5 | | 2.0 | | 574,668 | 1 | | ||||
| | | | | | | | | | | | | | | |
- 1
- Amount based on Named Officer's cash bonus target and his weighted average performance with a weight of 40% for personal performance and 60% for Company performance.
The cash bonuses were determined through the use of both an individual and a Company performance component with a possible
range of 0 to 1.5 for the personal performance multiplier and a possible range of 0 to 2.0 for the Company performance multiplier,
55
Executive Compensation
depending upon performance during the year. Both the personal performance multiplier and the Company performance multiplier were determined by the Compensation Committee for each Named Officer based on the Committee's assessment of the Company's performance relative to the general corporate goals described below and, in the case of each of Mr. Landry, Dr. Stahl, and Mr. Terifay, the Named Officer's personal performance during the year.
The Compensation Committee determined that the Company's performance in 2015 well exceeded its 2015 corporate goals. These corporate achievements included the following:
- •
- 47% growth in EYLEA® global net product sales as compared to 2014;
- •
- 46% growth in our total revenues as compared to 2014;
- •
- 19% growth in non-GAAP net income as compared to 2014 (non-GAAP net income is not a measure calculated in accordance with U.S.
Generally Accepted Accounting Principles; see Appendix A for a definition of non-GAAP net income and a reconciliation of non-GAAP net income to net income);
- •
- advances in our EYLEA® franchise, including regulatory approval of EYLEA® for the treatment of visual
impairment due to macular edema secondary to retinal vein occlusion and the treatment of visual impairment secondary to myopic choroidal neovascularization in the European Union; regulatory approval
of EYLEA® for the treatment of diabetic retinopathy in patients with diabetic macular edema in the United States; and regulatory approval of EYLEA® for the treatment of retinal
vein occlusion in Japan;
- •
- approval and launch of Praluent® (alirocumab) Injection, the first FDA-approved drug in a new class of drugs that lower LDL
("bad") cholesterol;
- •
- positive Phase 3 data for sarilumab from three Phase 3 studies in patients with rheumatoid arthritis (SARIL-RA-TARGET,
SARIL-RA-EASY, and SARIL-RA-ASCERTAIN) and submission of a Biologics License Application for sarilumab with the FDA;
- •
- positive pivotal Phase 2b data for dupilumab in asthma and completion of enrollment of the dupilumab atopic dermatitis
Phase 3 studies;
- •
- new collaboration agreement relating to fasinumab with Mitsubishi Tanabe Pharma Corporation for Japan, Korea, and nine other Asian
countries, excluding China;
- •
- initiation of Phase 3 clinical study of REGN2222 for Respiratory Syncytial Virus;
- •
- continued growth of our clinical development pipeline, as evidenced by the submission of one Investigational New Drug Application with the FDA in 2015 and 13 product
candidates (consisting of one Trap-based and 12 fully-human monoclonal antibody product candidates based on the Company's VelocImmune® technology) in clinical development as of December 31, 2015;
- •
- new global strategic collaboration with Sanofi to discover, develop, and commercialize antibody-based cancer treatments in the field of
immuno-oncology; and
- •
- further important steps to support our current and future growth, including adding two new buildings in the Tarrytown campus providing nearly 300,000 square feet of additional laboratory and office space; significant progress with the construction of a new manufacturing facility in Limerick, Ireland; and increasing headcount on a year-over-year basis by approximately 47% as of December 31, 2015.
See "Section 1 – Summary – 2015 Performance Overview" above for additional information.
In recognition of these significant achievements, the Company performance multiplier for 2015 was set at 2.0.
With respect to 2015, the Compensation Committee approved a personal performance multiplier of 1.5 for each of Mr. Landry, Dr. Stahl, and Mr. Terifay. The personal performance component accounted for 40% of these officers' bonuses. The Company component was based on a Company performance multiplier that was determined based on the Company's overall corporate performance (as described above) against 2015 goals that were approved by the board of directors in January 2015. This Company performance component accounted for 60% of the bonuses awarded to Mr. Landry, Dr. Stahl, and Mr. Terifay. In the case of Drs. Schleifer and Yancopoulos, the Compensation Committee focused exclusively on our overall Company performance in 2015 (as described above) when determining their cash bonuses and did not utilize a personal performance multiplier.
In determining the personal performance multiplier for Mr. Landry, the Compensation Committee gave special consideration to Mr. Landry's leadership of and accomplishments in the Company's accounting and finance functions and his assumption of additional responsibilities since he joined the Company in September 2013. In the case of Dr. Stahl, the Compensation Committee focused on the progress and continued expansion of the Company's preclinical and clinical development pipeline, including the positive results from the Company's clinical trials reported in 2015, as summarized in "Section 1 – Summary – 2015 Performance Overview" above. In the case of Mr. Terifay, the Compensation Committee focused primarily on the continued commercial success of EYLEA® and the fact that EYLEA® U.S. net product sales in 2015 grew by 54% compared to 2014; the launch of Praluent® in 2015; and his leadership of the Company's commercial group, including the increase in his responsibilities in
56
Executive Compensation
connection with the expansion of the commercialization group to support the launch of Praluent® and the planned launches of sarilumab and dupilumab.
Annual Stock Option Awards
We have used stock option grants as the primary vehicle for offering long-term incentives and rewarding our Named Officers and other eligible employees. These time-based stock options generally vest at a rate of 25% per year over the first four years of the ten-year option term, subject to the executive's continued employment. We grant stock option awards to our Named Officers and other eligible employees based on their annual performance and their position and responsibilities with the Company. Each of our Named Officers generally receives an annual stock option grant and all of our other regular employees are also eligible for an annual stock option grant, subject to satisfactory performance. The number of stock options granted to each Named Officer is determined on a discretionary basis, rather than by a formula. The Compensation Committee primarily considers the number of shares underlying the awards relative to the number of basic shares of common stock outstanding and not the grant date fair value of the award (as determined according to the Black-Scholes model). The Compensation Committee is therefore able to evaluate such grants on a consistent basis as compared to other companies and regardless of fluctuations in the price of Regeneron's or other companies' common stock. Further, focusing on the number of shares and the incremental sharing rate of potential future upside (rather than targeting a specific Black-Scholes grant date fair value) avoids rewarding officers with larger grant sizes following a decline in our stock price. While the Compensation Committee takes the estimated Black-Scholes grant date fair value of annual stock option grants into account, it does not necessarily determine its compensation decisions.
It has been the practice of our Compensation Committee to grant annual stock option awards to eligible employees whose performance is determined to merit an annual grant, including the Named Officers, at a meeting held during December. In 2015, stock option awards (all of which were non-qualified stock options) were granted to our Named Officers and other eligible employees on December 16, 2015. Pursuant to the terms of the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan, stock option awards are granted, for so long as our common stock is listed on the NASDAQ Global Select Market, with an exercise price equal to the
average of the high and low sales price per share of our common stock as quoted on the NASDAQ Global Select Market on the date of the grant or, if such date is not a trading day, on the last preceding date on which there was a sale of our common stock on the NASDAQ Global Select Market.
All of the stock options granted to our Named Officers in 2015 will vest ratably over a period of four years. Except as set forth below under the heading "Post-Employment Compensation" on page 66, stock option vesting ceases, and unvested stock options are forfeited, upon termination of employment. The use of only time-based stock options is consistent with the Company's practice prior to 2008 and since 2012.
Our Named Officers received a grant of time-based stock options on December 16, 2015, as set forth below. The annual grants shown below reflected an average reduction of 15% compared to 2014 (other than Mr. Terifay's award, which remained at the 2014 level due to his promotion to Executive Vice President, Commercial). This decrease constituted the third consecutive double-digit percentage decrease in the annual grant of stock options to our Named Officers, in each case following outstanding TSR performance. In reducing the size of 2015 annual stock option awards to the Named Officers, the Compensation Committee sought to reduce the potential dilutive impact of new equity awards without adversely affecting the effectiveness of our executive compensation program, which has successfully motivated our senior management team to deliver high operating performance and shareholder value. The reduction also took into account the increase in the Company's stock price in recent years. We will continue to assess the number of stock options that will be awarded to the Named Officers as annual merit grants.
Named Officer |
|
Stock Option Award1 |
||
Leonard S. Schleifer, M.D., Ph.D. |
| 172,723 | ||
George D. Yancopoulos, M.D., Ph.D. |
| 146,815 | ||
Robert E. Landry |
| 28,900 | ||
Neil Stahl, Ph.D. |
| 68,638 | ||
Robert J. Terifay |
| 40,000 | ||
| | | | | |
- 1
- These stock options all have an exercise price of $555.67 per share, the average of the high and low sales price per share of our common stock as quoted on the NASDAQ Global Select Market on the date of grant.
57
Executive Compensation
| |
How did we determine the size of our 2015 annual Named Officer stock option awards? |
| | |||
|
The size of the time-based stock option awards granted to our Named Officers were based on the following six factors: • past Company practices (i.e., history of grants to relevant executives); • an assessment of each Named Officer's 2015 performance against the Named Officer's goals, as established by our CEO (in the case of the Named Officers other than our Chief Executive Officer) and the Compensation Committee (in the case of our Chief Executive Officer), as described above under "– Annual Cash Bonus"; • Regeneron's significant corporate accomplishments in 2015; • recognition that cash compensation of the Named Officers is generally positioned at or below the median of the Peer Group despite the fact that Regeneron is a high-growth, high-performing company, which supports a higher level of equity grants; |
• an evaluation of the awards as a percentage of the total basic shares outstanding as compared to Peer Group and survey data, in particular the 2015 Radford Global Life Sciences Survey (see "Section 2 – Analysis of 2015 Executive Compensation Based on Compensation Objectives – Market Competitiveness and Employee Retention"); and • the increase in the Company stock price since the annual stock option awards in 2014 and prior years, with the resulting increase in the grant date fair value of the 2015 annual stock option awards (as determined according to the Black-Scholes model for valuing stock options), which contributed to the decision to implement the third consecutive double-digit percentage reduction in the annual grant of stock options to our Named Officers (for information regarding our burn rate since 2012, see "Regeneron Stock Utilization vs. Headcount" on page 48). As noted above, Dr. Yancopoulos's 2015 annual stock option award was set at 85% of Dr. Schleifer's (consistent with other elements of his compensation and our historical practice). |
|
|||
Perquisites and Other Personal Benefits
The Named Officers are provided with a limited number of perquisites and other personal benefits. The Compensation Committee periodically reviews the perquisites and other personal benefits provided to the Named Officers, including the Chief Executive Officer.
All of the Named Officers are eligible to receive financial and tax planning assistance, which are taxable benefits and are meant to save the executives time in order to focus on business matters as well as to recognize that their tax situations are affected by their employment at Regeneron. Similar to other employees, the Named Officers may participate in company-wide health, disability, life insurance, and other benefit plans, as well as our 401(k) Savings Plan. All employees who participate in our 401(k) Savings Plan are eligible to receive certain matching contributions. In each plan year, we contribute to each participant's account a matching contribution (in the form of shares of our common stock) equal to 50% of a specified percentage of the participant's compensation that the participant has contributed to the plan (which was 6% with respect to each of 2013 and 2014 and 8% with respect to
2015), up to a maximum level established under the Internal Revenue Code. In addition, for 2014, the Company made an additional discretionary contribution equal to 1% of each eligible employee's salary. Each of our Named Officers participated in our 401(k) Savings Plan during 2015 and received a matching contribution in the aggregate amount of $10,600 in the form of shares of our common stock. The contribution was paid in February 2016 and is included in the compensation amounts reported for each of our Named Officers in the Summary Compensation Table included in this proxy statement. As with all employees, the number of shares of common stock that each Named Officer received was determined using the average market price per share of our common stock during the 401(k) Savings Plan year, which for 2015 was $498.52.
Our Chief Executive Officer is entitled to life insurance, long-term disability, and medical malpractice insurance premiums (as well as an additional amount for tax preparation and financial planning services) pursuant to the terms of his employment agreement, as described in footnote 5 to the Summary Compensation Table included in this proxy statement. Pursuant to the terms of our security policy, the Company's Chief Executive Officer and Chief Scientific Officer are
58
Executive Compensation
required, to the extent practicable, to utilize personal travel security services, on-site residential security at their primary residence, and secure car transportation. We calculate the aggregate incremental cost to Regeneron for secure car transportation as the portion of the invoiced amount from our third-party provider of such transportation that is attributable to personal use based on the number of hours used, without including fixed costs that would be incurred in any event.
In addition, in order to ensure increased efficiencies and to provide a more secure traveling environment, the Company utilizes on-demand air transportation for certain executive and director travel in accordance with guidelines approved by our board of directors. Based on the recommendation of an independent, third-party security study, the guidelines and our security policy (as amended in November 2015) require Drs. Schleifer and Yancopoulos (as well as their spouses and dependent children when they accompany them) to use, as much as practicable, Company-provided aircraft for all business and personal air travel. Starting in 2016, Regeneron will cover the cost of any such personal air travel for up to $250,000 in incremental cost annually for each of Drs. Schleifer and Yancopoulos. Family members or other guests may accompany our Named Officers and directors during on-demand air business travel, space permitting, so long as they cover any incremental cost related to such guests (other than with respect to the family members of Drs. Schleifer and Yancopoulos as described above). In addition, in limited circumstances personal use of on-demand air travel by our other Named Officers or directors may be permitted if authorized by the Chairman and any incremental cost is paid by the lead passenger. Any required reimbursement or other payment of the incremental cost is made to the extent permitted by applicable Federal Aviation Administration rules.
There was no unreimbursed personal use, or guest use resulting in any incremental cost to us, of Company-provided aircraft in 2015.
The Corporate Governance and Compliance Committee monitors business and any personal or guest on-demand air travel on a periodic basis.
Additional information regarding perquisites and other personal benefits provided to our Named Officers in, or with respect to, 2015 is given in the applicable footnotes to the Summary Compensation Table included in this proxy statement.
Potential Severance Benefits
Outstanding stock option award agreements (as well as outstanding restricted stock award agreements) for all employees (other than Dr. Vagelos, whose stock option awards contain change-of-control provisions consistent with those of non-employee director stock option awards, as described under "Corporate Governance – Compensation of Directors" above) include a "double trigger" provision for the acceleration of vesting of unvested stock options (or restricted stock) upon a termination by the Company without cause or by the employee for good reason within two years following a change in control. Our Chief Executive Officer has an employment agreement that provides for certain severance benefits following termination, including following death or disability, resignation following defined "good reason" events, or termination in connection with a change in control. The other Named Officers are covered by a change in control severance plan, which provides certain benefits to them and other designated officers if they are terminated in connection with a change in control. In addition, in the case of our Chief Scientific Officer, stock option and restricted stock award agreements applicable to his awards granted starting in December 2015 provide that he would have a "good reason" for terminating his employment with Regeneron upon or within two years after the occurrence of a change in control if the employment of our Chief Executive Officer has ended due to our Chief Executive Officer's involuntary termination (as defined in his employment agreement). Information regarding applicable payments under this employment agreement and change in control severance plan is provided under the heading "Post-Employment Compensation" on page 66.
Except as provided in our employment agreement with our Chief Executive Officer and in our change in control severance plan, our Named Officers will forfeit any unvested time-based stock options or restricted stock upon the termination of their employment for any reason (including disability or retirement) other than death. In the event of the death of an employee, any unvested stock options held by such employee become immediately exercisable, and any shares of restricted stock will become fully vested. For information regarding unvested stock options and shares of restricted stock held by our Named Officers as of December 31, 2015, see "Outstanding Equity Awards at Fiscal Year-End" on page 64. When employees (other than our Chief Executive Officer) retire, they forfeit all unvested time-based stock options and restricted stock. For all stock options granted prior to 2007, an employee (other than our Chief Executive Officer) who retires has up to two years to exercise stock options that are vested as of the date of his or her retirement. Commencing in 2007, we amended our forms of stock option agreement to allow the retired employee the remaining life of the 10-year stock option term to exercise stock options that are vested as of the date of his or her retirement.
The severance benefits provided to our Named Officers are designed to promote stability and continuity of our senior management and are intended to preserve employee morale and productivity and encourage retention in the face of the disruptive impact of an actual, threatened, or rumored change in control of the Company. The severance benefits were established following a review of comparable practices at the Company's peer companies and with the advice of the Compensation Committee's consultant. We have no pension, deferred compensation, or retirement plans, other than our 401(k) Savings Plan described above.
59
Executive Compensation
The Compensation Committee Report below shall not be deemed to be "soliciting material" or to be filed with the SEC or subject to Regulation 14A or 14C under the Exchange Act, or to the liabilities of Section 18 of the Exchange Act. Notwithstanding anything to the contrary set forth in any of the Company's previous filings under the Securities Act or the Exchange Act that might incorporate future filings, including this proxy statement, in whole or in part, the Compensation Committee Report below shall not be incorporated by reference into any such filings.
Compensation Committee Report
We have reviewed and discussed with management the Compensation Discussion and Analysis, beginning on page 35. Based on that review and discussion, we have recommended to the board of directors that the Compensation Discussion and Analysis be included in this proxy statement.
The Compensation Committee
Christine
A. Poon, Chairperson*
Charles A. Baker
Joseph L. Goldstein, M.D.
George L. Sing
- *
- Ms. Poon became Chairperson of the Compensation Committee effective as of April 1, 2016.
Compensation Committee Interlocks and Insider Participation
None of the members of the Compensation Committee is currently, or has been at any time since our formation, one of our officers or employees. None of our executive officers serves as a member of the board of directors or compensation committee of any entity that has one or more executive officers serving as a member of our board of directors or Compensation Committee.
60
Executive Compensation
The following table and accompanying footnotes provide information regarding compensation earned by, or paid to, our Named Officers (our Chief Executive Officer, our Chief Financial Officer, and our three other highest-compensated executive officers in 2015).
Summary Compensation Table
|
Name and principal position (a) |
|
Year (b) |
|
Salary ($) (c) |
|
Bonus ($)1 (d) |
|
Stock awards ($)2 (e) |
|
Option awards ($)2 (f) |
|
All other compensation ($)3 (i) |
|
Total ($) (j) |
||||||||||
| | Leonard S. Schleifer, M.D., Ph.D. | | 2015 | | 1,200,000 | | 2,880,000 | | — | | 43,307,918 | 4 | | 74,608 | 5 | | 47,462,526 | |||||||
|
President and Chief | | 2014 | | 1,071,200 | | 2,142,400 | | — | | 38,644,700 | 6 | | 107,124 | | 41,965,424 | ||||||||
|
Executive Officer | | 2013 | | 1,035,000 | | 2,070,000 | | — | | 33,062,325 | 7 | | 105,340 | | 36,272,665 | ||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | |
| | George D. Yancopoulos, M.D., Ph.D. | | 2015 | | 1,020,000 | | 2,448,000 | | — | | 36,811,837 | 4 | | 22,519 | 8 | | 40,302,356 | |||||||
|
President, Regeneron Laboratories | | 2014 | | 910,500 | | 1,821,040 | | — | | 32,744,746 | 6 | | 30,525 | | 35,506,811 | ||||||||
|
and Chief Scientific Officer | | 2013 | | 879,800 | | 1,759,500 | | — | | 28,096,838 | 7 | | 281,455 | | 31,017,593 | ||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | |
| | Robert E. Landry | | 2015 | | 517,500 | | 465,750 | | — | | 7,246,284 | 4 | | 19,360 | 9 | | 8,248,894 | |||||||
|
Senior Vice President, Finance | | 2014 | | 500,000 | | 455,000 | 10 | | — | | 6,403,138 | 6 | | 14,818 | | 7,372,956 | |||||||
|
and Chief Financial Officer* | | 2013 | | 144,231 | 11 | | 50,000 | 12 | | 1,363,500 | 13 | | 11,054,274 | 7 | | 1,130 | | 12,613,135 | |||||
| | | | | | | | | | | | | | | | | | | | | | | | | |
| | Neil Stahl, Ph.D. | | 2015 | | 550,000 | | 594,000 | | — | | 17,209,995 | 4 | | 20,515 | 14 | | 18,374,510 | |||||||
|
Executive Vice President, | | 2014 | | 493,500 | | 532,980 | | — | | 15,207,420 | 6 | | 20,215 | | 16,254,115 | ||||||||
|
Research & Development | | 2013 | | 476,800 | | 386,208 | | — | | 13,474,403 | 7 | | 17,235 | | 14,354,646 | ||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | |
| | Robert J. Terifay | | 2015 | | 532,100 | | 574,668 | | — | | 10,029,459 | 4 | | 20,515 | 14 | | 11,156,742 | |||||||
|
Executive Vice President, | | 2014 | | 465,800 | | 377,298 | | — | | 7,533,104 | 6 | | 20,125 | | 8,396,327 | ||||||||
|
Commercial | | 2013 | | 450,000 | | 364,500 | | — | | 6,847,486 | 7 | | 17,145 | | 7,679,131 | ||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | |
- *
- Mr. Landry joined the Company in September 2013.
- 1
- Bonuses are shown in the year in which they were accrued and earned.
- 2
- The amounts in column (e) and (f) reflect the respective aggregate grant date fair values (disregarding estimated forfeitures) of stock and option awards granted in 2015, 2014, and 2013, respectively, pursuant to the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan or the Regeneron Pharmaceuticals, Inc. Second Amended and Restated 2000 Long-Term Incentive Plan. Assumptions used in the calculation of these amounts are included in Note 14 to the Company's audited financial statements for the fiscal year ended December 31, 2015 included in the 2015 Annual Report. The 2013, 2014, and 2015 annual option awards reflect a double-digit percentage decrease in the number of shares underlying the awards, in each case as compared to the prior year (other
than Mr. Terifay's 2015 award, which remained at the 2014 level due to his promotion to Executive Vice President, Commercial). Mr. Landry did not receive an annual option award in 2013; his 2013 option award was granted pursuant to his offer letter with the Company in connection with the commencement of his employment.
- 3
- See "Compensation Discussion and Analysis – Section 4 – Elements of Executive Compensation – Perquisites and Other Personal Benefits" for further information. Certain 2015 perquisites and other personal benefits are quantified for each of the Named Officers in the footnotes to this table below based on the actual additional cost incurred by us in providing the perquisite or other personal benefit.
- 4
- Reflects the aggregate grant date fair value of time-based option awards granted in 2015.
61
Executive Compensation
- 5
- Includes (i) $1,655 for life insurance premiums, (ii) $23,961 for long-term disability insurance premiums, (iii) $13,889 for medical malpractice insurance premiums, (iv) $10,600 for 401(k) Savings Plan matching contributions in respect of 2015 paid in February 2016, (v) $9,915 for tax and financial planning advisory services, and (vi) $14,200 for personal use of secure car transportation in accordance with our security policy.
- 6
- Reflects the aggregate grant date fair value of time-based option awards granted in 2014.
- 7
- Reflects the aggregate grant date fair value of time-based option awards granted in 2013. In the case of Mr. Landry, such option award was granted pursuant to his offer letter with the Company in connection with the commencement of his employment.
- 8
- Consists of (i) $10,600 for 401(k) Savings Plan matching contributions in respect of 2015 paid in February 2016, (ii) $9,915 for tax and financial planning advisory services, and (iii) $2,004 for personal use of secure car transportation in accordance with our security policy.
- 9
- Consists of (i) $10,600 for 401(k) Savings Plan matching contributions in respect of 2015 paid in February 2016 and (ii) $8,760 for tax and financial planning advisory services.
- 10
- Includes the second installment in the amount of $50,000 of Mr. Landry's $100,000 sign-on bonus (paid in 2014).
- 11
- Represents Mr. Landry's base salary paid from September 9, 2013 (the starting date of Mr. Landry's employment with the Company) to the end of 2013.
- 12
- Consists of the first installment of Mr. Landry's $100,000 sign-on bonus (paid in 2013).
- 13
- Reflects the aggregate grant date fair value of restricted stock granted to Mr. Landry pursuant to his offer letter with the Company in connection with the commencement of his employment.
- 14
- Consists of (i) $10,600 for 401(k) Savings Plan matching contributions in respect of 2015 paid in February 2016 and (ii) $9,915 for tax and financial planning advisory services.
62
Executive Compensation
The following table and explanatory footnotes provide information regarding each equity award granted to our Named Officers during 2015. There were no non-equity incentive plan awards granted in 2015.
Grants of Plan-Based Awards
|
Name (a) |
|
Grant date (b) |
|
All other stock awards: number of shares of stock or units (#) (i) |
|
All other option awards: number of securities underlying options (#) (j) |
|
Exercise or base price of option awards ($/Sh)1 (k) |
|
Closing price of Company common stock on grant date ($/Sh)1 (l) |
|
Grant date fair value of stock and option awards ($)2 (l) |
|||||||
| Leonard S. Schleifer, M.D., Ph.D. | | 12/16/2015 | 3 | | — | | 172,723 | | 555.67 | | 559.67 | | 43,307,918 | ||||||
| | | | | | | | | | | | | | | | | | | | |
| George D. Yancopoulos, M.D., Ph.D. | | 12/16/2015 | 3 | | — | | 146,815 | | 555.67 | | 559.67 | | 36,811,837 | ||||||
| | | | | | | | | | | | | | | | | | | | |
| Robert E. Landry | | 12/16/2015 | 3 | | — | | 28,900 | | 555.67 | | 559.67 | | 7,246,284 | ||||||
| | | | | | | | | | | | | | | | | | | | |
| Neil Stahl, Ph.D. | | 12/16/2015 | 3 | | — | | 68,638 | | 555.67 | | 559.67 | | 17,209,995 | ||||||
| | | | | | | | | | | | | | | | | | | | |
| Robert J. Terifay | | 12/16/2015 | 3 | | — | | 40,000 | | 555.67 | | 559.67 | | 10,029,459 | ||||||
| | | | | | | | | | | | | | | | | | | | |
- 1
- These options have an exercise price equal to the average of the high and low sales price per share of the Company's common stock on the date of grant. Therefore, the closing price of our common stock on the grant date may be higher or lower than the exercise price of these options.
- 2
- The amounts in this column represent the grant date fair value of the awards made pursuant to the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan. The assumptions used in the calculation of these amounts are included in Note 14 to the Company's
- audited financial statements for the fiscal year ended December 31, 2015 included in the 2015 Annual Report.
- 3
- The Named Officer received a non-qualified stock option award that vests at a rate of 25% per year over the first four years of the ten-year option term.
63
Executive Compensation
The following table and explanatory footnotes provide information regarding unexercised stock options and unvested restricted stock awards held by our Named Officers as of December 31, 2015.
OUTSTANDING EQUITY AWARDS AT FISCAL YEAR-END
|
|
Option Awards |
|
Stock Awards |
||||||||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Name |
|
Number of securities underlying unexercised options (#) exercisable (b) |
|
Number of securities underlying unexercised options (#) unexercisable (c) |
|
Equity incentive plan awards: number of securities underlying unexercised unearned options (#) (d) |
|
Option exercise price ($) (e) |
|
Option expiration date (f) |
|
Number of shares or units of stock that have not vested (#) (g) |
|
Market value of shares or units of stock that have not vested ($)5 (h) |
|
Equity incentive plan awards: number of unearned shares, units or other rights that have not vested (#) (i) |
|
Equity incentive plan awards: market or payout value of unearned shares, units or other rights that have not vested ($) (j) |
||||||||||
Leonard S. Schleifer, M.D., Ph.D. |
| — | | 172,723 | 1 | | — | | 555.67 | | 12/16/2025 | | — | | — | | — | | — | |||||||||
|
| 50,801 | | 152,403 | 2 | | — | | 399.66 | | 12/16/2024 | | — | | — | | — | | — | |||||||||
|
| 119,532 | | 119,531 | 3 | | — | | 270.43 | | 12/13/2023 | | — | | — | | — | | — | |||||||||
|
| 210,938 | | 70,312 | 4 | | — | | 179.13 | | 12/14/2022 | | — | | — | | — | | — | |||||||||
|
| 160,000 | | — | | — | | 52.03 | | 12/16/2021 | | — | | — | | — | | — | ||||||||||
|
| 240,000 | | — | | — | | 52.03 | | 12/16/2021 | | — | | — | | — | | — | ||||||||||
|
| 125,000 | | — | | — | | 30.63 | | 12/14/2020 | | — | | — | | — | | — | ||||||||||
|
| 187,500 | | — | | — | | 30.63 | | 12/14/2020 | | — | | — | | — | | — | ||||||||||
|
| 187,500 | | — | | — | | 21.25 | | 12/18/2019 | | — | | — | | — | | — | ||||||||||
|
| 125,000 | | — | | — | | 21.25 | | 12/18/2019 | | — | | — | | — | | — | ||||||||||
|
| 125,000 | | — | | — | | 16.80 | | 12/17/2018 | | — | | — | | — | | — | ||||||||||
|
| 187,500 | | — | | — | | 16.80 | | 12/17/2018 | | — | | — | | — | | — | ||||||||||
|
| 250,000 | | — | | — | | 21.92 | | 12/17/2017 | | — | | — | | — | | — | ||||||||||
|
| 250,000 | | — | | — | | 20.32 | | 12/18/2016 | | — | | — | | — | | — | ||||||||||
TOTAL |
| 2,218,771 | | 514,969 | | | | | | | | | | | | | | |||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
George D. Yancopoulos, M.D., Ph.D. |
| — | | 146,815 | 1 | | — | | 555.67 | | 12/16/2025 | | — | | — | | — | | — | |||||||||
|
| 43,181 | | 129,542 | 2 | | — | | 399.66 | | 12/16/2024 | | — | | — | | — | | — | |||||||||
|
| 101,602 | | 101,602 | 3 | | — | | 270.43 | | 12/13/2023 | | — | | — | | — | | — | |||||||||
|
| 179,298 | | 59,765 | 4 | | — | | 179.13 | | 12/14/2022 | | — | | — | | — | | — | |||||||||
|
| 158,079 | | — | | — | | 52.03 | | 12/16/2021 | | — | | — | | — | | — | ||||||||||
|
| 240,000 | | — | | — | | 52.03 | | 12/16/2021 | | — | | — | | — | | — | ||||||||||
|
| 96,736 | | — | | — | | 30.63 | | 12/14/2020 | | — | | — | | — | | — | ||||||||||
|
| 150,000 | | — | | — | | 30.63 | | 12/14/2020 | | — | | — | | — | | — | ||||||||||
|
| 150,000 | | — | | — | | 21.25 | | 12/18/2019 | | — | | — | | — | | — | ||||||||||
|
| 95,295 | | — | | — | | 21.25 | | 12/18/2019 | | — | | — | | — | | — | ||||||||||
|
| 94,048 | | — | | — | | 16.80 | | 12/17/2018 | | — | | — | | — | | — | ||||||||||
|
| 150,000 | | — | | — | | 16.80 | | 12/17/2018 | | — | | — | | — | | — | ||||||||||
|
| 195,438 | | — | | — | | 21.92 | | 12/17/2017 | | — | | — | | — | | — | ||||||||||
|
| 195,079 | | — | | — | | 20.32 | | 12/18/2016 | | — | | — | | — | | — | ||||||||||
|
| — | | — | | — | | — | | — | | 500,000 | 6 | | 542.87 | | — | | — | |||||||||
TOTAL |
| 1,848,756 | | 437,724 | | | | | | | | 500,000 | | | | | | |||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Robert E. Landry |
| — | | 28,900 | 1 | | — | | 555.67 | | 12/16/2025 | | — | | — | | — | | — | |||||||||
|
| 8,500 | | 25,500 | 2 | | — | | 399.66 | | 12/16/2024 | | — | | — | | — | | — | |||||||||
|
| 26,000 | | 40,000 | 7 | | — | | 272.70 | | 9/9/2023 | | — | | — | | — | | — | |||||||||
|
| — | | — | | — | | — | | — | | 5,000 | 8 | | 542.87 | | — | | — | |||||||||
TOTAL |
| 34,500 | | 94,400 | | | | | | | | 5,000 | | | | | | |||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
64
Executive Compensation
|
|
Option Awards |
|
Stock Awards |
||||||||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Name |
|
Number of securities underlying unexercised options (#) exercisable (b) |
|
Number of securities underlying unexercised options (#) unexercisable (c) |
|
Equity incentive plan awards: number of securities underlying unexercised unearned options (#) (d) |
|
Option exercise price ($) (e) |
|
Option expiration date (f) |
|
Number of shares or units of stock that have not vested (#) (g) |
|
Market value of shares or units of stock that have not vested ($)5 (h) |
|
Equity incentive plan awards: number of unearned shares, units or other rights that have not vested (#) (i) |
|
Equity incentive plan awards: market or payout value of unearned shares, units or other rights that have not vested ($) (j) |
||||||||||
Neil Stahl, Ph.D. |
| — | | 68,638 | 1 | | — | | 555.67 | | 12/16/2025 | | — | | — | | — | | — | |||||||||
|
| 20,188 | | 60,562 | 2 | | — | | 399.66 | | 12/16/2024 | | — | | — | | — | | — | |||||||||
|
| 47,500 | | 47,500 | 3 | | — | | 270.43 | | 12/13/2023 | | — | | — | | — | | — | |||||||||
|
| 4,527 | | — | | — | | 268.40 | | 12/17/2017 | | — | | — | | — | | — | ||||||||||
|
| 84,375 | | 28,125 | 4 | | — | | 179.13 | | 12/14/2022 | | — | | — | | — | | — | |||||||||
|
| 6,017 | | — | | — | | 145.70 | | 12/17/2017 | | — | | — | | — | | — | ||||||||||
|
| 48,079 | | — | | — | | 52.03 | | 12/16/2021 | | — | | — | | — | | — | ||||||||||
|
| 75,000 | | — | | — | | 52.03 | | 12/16/2021 | | — | | — | | — | | — | ||||||||||
|
| 46,736 | | — | | — | | 30.63 | | 12/14/2020 | | — | | — | | — | | — | ||||||||||
|
| 20,295 | | — | | — | | 21.25 | | 12/18/2019 | | — | | — | | — | | — | ||||||||||
TOTAL |
| 352,717 | | 204,825 | | | | | | | | | | | | | | |||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Robert J. Terifay |
| — | | 40,000 | 1 | | — | | 555.67 | | 12/16/2025 | | — | | — | | — | | — | |||||||||
|
| 10,000 | | 30,000 | 2 | | — | | 399.66 | | 12/16/2024 | | — | | — | | — | | — | |||||||||
|
| 25,000 | | 25,000 | 3 | | — | | 270.43 | | 12/13/2023 | | — | | — | | — | | — | |||||||||
|
| 56,250 | | 18,750 | 4 | | — | | 179.13 | | 12/14/2022 | | — | | — | | — | | — | |||||||||
|
| 1,921 | | — | | — | | 52.03 | | 12/16/2021 | | — | | — | | — | | — | ||||||||||
|
| 30,579 | | — | | — | | 52.03 | | 12/16/2021 | | — | | — | | — | | — | ||||||||||
|
| 48,750 | | — | | — | | 52.03 | | 12/16/2021 | | — | | — | | — | | — | ||||||||||
TOTAL |
| 172,500 | | 113,750 | | | | | | | | | | | | | | |||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
- 1
- This stock option award was granted to the Named Officer on December 16, 2015 and vests at a rate of 25% per year over the first four years of the option term.
- 2
- This stock option award was granted to the Named Officer on December 16, 2014 and vests at a rate of 25% per year over the first four years of the option term.
- 3
- This stock option award was granted to the Named Officer on December 13, 2013 and vests at a rate of 25% per year over the first four years of the option term.
- 4
- This stock option award was granted to the Named Officer on December 14, 2012 and vests at a rate of 25% per year over the first four years of the option term.
- 5
- Reflects the closing price per share of the Company's common stock on the NASDAQ Global Select Market on December 31, 2015.
- 6
- This restricted stock award was granted to the Named Officer on June 27, 2012 and vests 100% on December 17, 2017, subject to the Named Officer's continued employment.
- 7
- This stock option award was granted to the Named Officer on September 9, 2013 and vests at a rate of 25% per year over the first four years of the option term.
- 8
- This restricted stock award was granted to the Named Officer on September 9, 2013 and vests 100% on the fifth anniversary of the date of grant, subject to the Named Officer's continued employment.
65
Executive Compensation
The following table and explanatory footnotes provide information with regard to amounts realized by our Named Officers during 2015 as a result of the exercise of stock options or the vesting of restricted stock awards.
Option Exercises and Stock Vested
|
|
Option awards |
|
Stock awards |
|||||||||
| | | | | | | | | | | | | | |
Name |
|
Number of shares acquired on exercise (#) (b) |
|
Value realized on exercise ($)1 (c) |
|
Number of shares acquired on vesting (#) (d) |
|
Value realized on vesting ($) (e) |
|||||
Leonard S. Schleifer, M.D., Ph.D. |
| 7,226 | | 749,192 | | — | | — | |||||
George D. Yancopoulos, M.D., Ph.D. |
| 184,739 | | 101,049,378 | | — | | — | |||||
Robert E. Landry |
| 14,000 | | 3,048,560 | | — | | — | |||||
Neil Stahl, Ph.D. |
| 101,921 | | 54,901,992 | | — | | — | |||||
Robert J. Terifay |
| 81,250 | | 37,906,863 | | — | | — | |||||
| | | | | | | | | | | | | | |
- 1
- Amounts reflect the difference between the exercise price of the option(s) and the average of the high and low sales price per share of the Company's common stock on the NASDAQ Global Select Market on the exercise date(s).
As discussed under "Section 4 – Elements of Executive Compensation – Potential Severance Benefits" on page 59, our Named Officers are entitled to certain severance benefits upon the voluntary or involuntary termination of their employment. We provide additional information regarding the severance benefits available to our Named Officers in the tables on pages 67 and 69. For our Chief Executive Officer, the table shows the amounts payable under his employment agreement upon his involuntary or not-for-cause termination, termination in connection with a corporate change of control, and in the event of his disability or death. For the other Named Officers, the table shows their post-termination compensation arrangements under our change in control severance plan upon an involuntary or not-for-cause termination in connection with a corporate change of control.
Leonard S. Schleifer, M.D., Ph.D., Employment Agreement
We entered into an employment agreement with our Chief Executive Officer, Dr. Schleifer, effective as of December 20, 2002, providing for his employment with the Company through December 31, 2003 and continuing thereafter on a year-by-year basis. On November 14, 2008, this employment agreement was amended and restated to bring the employment agreement into compliance with Section 409A of the
Internal Revenue Code. Pursuant to this agreement, we agreed that in the event that Dr. Schleifer's employment is terminated by us other than for cause (as defined in the agreement) or is terminated by Dr. Schleifer for good reason (as defined in the agreement to include specified acts of constructive termination, together called an "involuntary termination"), we will pay Dr. Schleifer an amount equal to 125% of the sum of his base salary plus his average bonus paid over the prior three years. This amount will be paid in a lump sum severance payment. In addition, we will continue to provide Dr. Schleifer and his dependents medical, dental, and life insurance benefits for eighteen months. Subject to the discussion in the following paragraph, in the event that Dr. Schleifer's employment is terminated for any reason other than for cause, all of his unvested stock options will continue to vest in accordance with the terms of the applicable award grant and he will be entitled to exercise the stock options throughout their original term, which is generally ten years from the date of grant.
Upon an involuntary termination (i.e., a termination by the Company without cause or by Dr. Schleifer for good reason, each as defined in the agreement) within three years after a change of control of the Company or within three months prior to such a change of control, we will pay Dr. Schleifer an amount equal to three times the sum of his annual base salary plus his average bonus over the prior three years. This amount will be paid in a lump sum severance payment. In addition, we will continue to provide Dr. Schleifer and his dependents medical, dental, and life insurance benefits for thirty-six months. Upon such an involuntary termination in
66
Executive Compensation
connection with a change of control, Dr. Schleifer's outstanding stock options will vest immediately and remain exercisable throughout their original term, which is generally ten years from the date of grant. If aggregate severance payments to Dr. Schleifer in connection with a change of control exceed certain thresholds set forth in the Internal Revenue Code, then we will pay him an additional amount to cover any resulting excise tax obligations, unless the excise taxes could be eliminated by reducing Dr. Schleifer's cash severance payments and benefits under the agreement by less than ten
percent, in which case such benefits and payments will be reduced accordingly.
The following table reflects the potential payments to our Chief Executive Officer under his employment agreement upon his termination, effective December 31, 2015, under different scenarios, including following a change of control, as well as upon death or disability. The information in the table below is based on the assumptions set forth in the footnotes to the table; actual values and amounts may differ from those presented below.
Potential Severance Payments under Dr. Schleifer's Employment Agreement
|
|
Cash Severance |
|
Benefits Continuation |
|
Death Benefits4 |
|
Disability Benefits |
|
Value of Accelerated Stock Options |
|
Cutback/ Gross- up6 |
|
Total Amount |
||||||||
Involuntary Termination Following a Change of Control 1 |
$ | 10,112,400 | 2 | $ | 199,311 | 3 | | — | | — | $ | 79,965,946 | 5 | | — | $ | 90,277,657 | |||||
Involuntary Termination |
$ |
4,213,500 |
7 |
$ |
91,742 |
8 |
|
— |
|
— |
|
— |
|
— |
$ |
4,305,242 |
||||||
Death |
|
— |
$ |
89,260 |
8 |
|
— |
|
— |
|
— |
9 |
|
— |
$ |
89,260 |
||||||
Disability |
|
— |
$ |
91,742 |
8 |
|
— |
$ |
630,000 |
10 |
|
— |
|
— |
$ |
721,742 |
||||||
| | | | | | | | | | | | | | | | | | | | | | | |
- 1
- For purposes of these calculations, (i) we used Dr. Schleifer's 2015 base salary and the annual bonuses paid to Dr. Schleifer for performance in 2012, 2013, and 2014, respectively; (ii) we assumed that Dr. Schleifer received his annual bonus that was earned in 2015 and paid in 2016 (described in the Summary Compensation Table on page 61); (iii) we took into consideration, for purposes of determining whether Dr. Schleifer was entitled to receive a gross-up payment under the terms of his employment agreement, the fact that Dr. Schleifer's stock options continue to vest according to their original vesting schedule following a voluntary or involuntary termination (other than in connection with a change of control); (iv) we assumed an 8% annual increase in medical premiums, 5% annual increase in dental premiums, and an increase in annual life insurance premiums from $1,655 to $7,000 in October 2017; (v) we assumed that the medical and dental insurance benefits received in 2016, 2017, and 2018 would be taxable and that Dr. Schleifer would be eligible for a tax gross-up for these benefits under the terms of his employment agreement; (vi) although Dr. Schleifer's employment agreement provides for restrictive covenants, including a six-month non-compete obligation, no specific value has been ascribed to these covenants solely for purposes of this calculation; and (vii) although certain payments to Dr. Schleifer would be subject to potential delays upon separation of service under Section 409A of the Internal Revenue Code, we did not attempt to determine which, if any, payments would be delayed.
- 2
- Equal to three times the sum of (a) Dr. Schleifer's 2015 base salary and (b) the average annual bonus paid to Dr. Schleifer for performance in the three completed years prior to the termination date. For purposes of this calculation, we used Dr. Schleifer's annual bonuses for performance in 2012, 2013, and 2014.
- 3
- Equal to the estimated cost of providing Dr. Schleifer and his dependents medical, dental, and life insurance benefits for thirty-six months.
- 4
- We maintain $1 million of term life insurance covering Dr. Schleifer payable to his designated beneficiary.
- 5
- Equal to the aggregate amount of the differences between the exercise prices of Dr. Schleifer's accelerated stock options and the closing sales price per share of the Company's common stock on the NASDAQ Global Select Market on December 31, 2015 of $542.87.
- 6
- Under Dr. Schleifer's employment agreement, if payments due in connection with a change of control are subject to excise taxes under Section 280G of the Internal Revenue Code, we will cut back the payments if the excise tax can be eliminated by reducing his cash severance payments and benefits by less than ten percent. Otherwise, we will pay him an additional "gross up" amount so that his after tax benefits are the same as though no excise tax had been applied. We have determined that Dr. Schleifer would not be subject to excise taxes if he had been terminated on December 31, 2015 as a result of a change of control.
- 7
- Equal to 1.25 times the sum of (a) Dr. Schleifer's 2015 base salary and (b) the average annual bonus paid to Dr. Schleifer for performance in the three completed years prior to the termination date. For purposes of this calculation, we used Dr. Schleifer's annual bonuses for performance in 2012, 2013, and 2014.
- 8
- Equal to the estimated cost of providing Dr. Schleifer and his dependents medical, dental, and life insurance benefits for eighteen months.
- 9
- As discussed under "Section 4 – Elements of Executive Compensation – Potential Severance Benefits" on page 59, unvested stock options held by any employee (including Dr. Schleifer) become immediately exercisable upon his or her death.
- 10
- Represents 35% of Dr. Schleifer's 2015 salary over a period of eighteen months. We have assumed long-term disability coverage exists pursuant to Dr. Schleifer's employment agreement for the remaining 65% of Dr. Schleifer's salary.
67
Executive Compensation
Change in Control Severance Plan
Each of the Named Officers, other than our Chief Executive Officer, participates in our change in control severance plan that was adopted by the board of directors on January 20, 2006. The purposes of the plan are (i) to help us retain key employees, (ii) to help maintain the focus of such employees on our business and to mitigate the distractions caused by the possibility that we may be the target of an acquisition, and (iii) to provide certain benefits to such employees in the event their employment is terminated (or constructively terminated) after, or in contemplation of, a change in control. On November 14, 2008, the change in control severance plan was amended and restated to bring it into compliance with Section 409A of the Internal Revenue Code.
Under the plan, each participant is entitled to receive a cash severance payment in an amount equal to one, or, in designated cases, including with respect to the Named Officers other than Dr. Schleifer, two times the sum of the participant's annual base salary and his or her average annual bonus over the prior three years if, within two years after or 180 days before a change in control, either the participant resigns his or her employment for Good Reason (as defined in the plan) or the participant's employment is terminated by the Company for any reason other than Cause (as defined in the plan). This amount will be paid in a lump sum severance payment. A participant so terminated is also entitled to receive a pro rata bonus for the year in which he or she is terminated based on the portion of the year the participant was employed by us. In addition, for either one or two years, as the case may be, plan participants will receive continuation of health care coverage and welfare benefits provided by us, to the extent permitted by our benefit plans, at a cost no greater than what the participant's cost would have been if he or she had continued his or her employment with the Company.
In the event that a plan participant resigns his or her employment for Good Reason (which generally conforms to the definition in Section 409A), or the participant's employment is terminated by the Company for any reason other than Cause, in either case within two years after or 180 days before a change in control, then the participant's stock options and other equity awards granted under our long-term incentive plans that would have vested prior to or upon the change in control will become vested on the change in control date, and the exercise period of such equity awards, and other equity awards held by the participant that otherwise would have expired, will be extended to the later of (i) thirty days following the first date after a change in control in which the shares underlying the equity award may be traded, and (ii) the permitted exercise date in the plan or grant assuming the change in control happened immediately prior to the participant's termination. However, in no event will any stock option or other equity award be extended (i) beyond the expiration date of the grant, or (ii) such that the grant will be subject to the additional tax under Section 409A of the Internal Revenue Code.
In the event that a participant would become subject to a "golden parachute" excise tax under Section 4999 of the Internal Revenue Code as a result of severance benefits and payments, the severance benefits and payments owed to the participant shall be reduced to an amount one dollar less than the amount that would subject the participant to the excise tax, unless the total severance benefits/payments net of the excise taxes are greater than the amount that the participant would receive following any such reduction.
68
Executive Compensation
The following table shows the potential payments to our Named Officers (other than our Chief Executive Officer), upon their hypothetical termination (other than for Cause) or resignation for Good Reason, in the two years following, or the six months prior to, a change in control. The information in the
table below is based on an effective termination or resignation date of December 31, 2015 and on the assumptions set forth in the footnotes to the table; actual values and amounts may differ from those presented below.
Potential Payments under Change in Control Severance Plan
|
|
Cash Severance1 |
|
Benefits Continuation2 |
|
Value of Accelerated Stock Options/Restricted Stock3 |
|
Cutback4 |
|
Total Amount5 |
||||||
George D. Yancopoulos, M.D., Ph.D. |
$ | 5,730,360 | $ | 114,488 | $ | 339,406,080 | | — | $ | 345,250,928 | ||||||
Robert E. Landry |
$ | 1,440,000 | $ | 111,338 | $ | 17,173,005 | | — | $ | 18,724,343 | ||||||
Neil Stahl, Ph.D. |
$ | 1,961,570 | $ | 53,425 | $ | 31,844,172 | | — | $ | 33,859,167 | ||||||
Robert J. Terifay |
$ | 1,833,191 | $ | 53,416 | $ | 17,927,425 | | — | $ | 19,814,032 | ||||||
| | | | | | | | | | | | | | | | | |
- 1
- Equal to two times the sum of (a) the Named Officer's 2015 base salary and (b) the average annual bonus paid to the Named Officer (other than Mr. Landry) over the prior three years. In the case of Mr. Landry (who joined the Company in September 2013), we averaged his 2013 and 2014 annual bonus amounts of $0 and $405,000, respectively, and took into account that he did not receive a bonus in respect of his performance in 2013.
- 2
- Equal to the estimated cost of providing each Named Officer and his dependents medical, dental, vision, disability, and life insurance coverage for twenty-four months, plus the estimated cost of providing each Named Officer tax and financial planning advisory services for twenty-four months.
- 3
- For stock options, equal to the aggregate amount of the differences between the exercise prices of each Named Officer's accelerated "in-the-money" stock options and the closing sales price per share of the Company's common stock on the NASDAQ Global Select Market on December 31, 2015 of $542.87. The amounts also include the value as of December 31, 2015 of unvested restricted stock.
- 4
- In accordance with the terms of the change in control severance plan, the total amount for Mr. Landry has not been "cut back" as he would be in a more favorable net after-tax position without any such reduction. None of the other Named Officers listed in the table above would have been expected to receive severance payments in excess of his applicable "golden parachute" safe harbor amount.
- 5
- For purposes of these calculations, (i) we used base salaries as of December 31, 2015 and annual bonuses paid to the Named Officers (other than Mr. Landry) for performance in 2012, 2013, and 2014, respectively; in the case of Mr. Landry (who joined the Company in September 2013), we averaged his 2013 and 2014 annual bonus amounts of $0 and $405,000, respectively, and took into account that he did not receive a bonus in respect of his performance in 2013; (ii) we assumed that each Named Officer received his annual bonus that was earned in 2015 and paid in 2016 (described in the Summary Compensation Table on page 61); (iii) we took into consideration, for purposes of determining whether each Named Officer was subject to a reduction under the terms of the change in control severance plan, the fact that each Named Officer's stock options vest following an involuntary termination without Cause or termination for Good Reason following a change in control (parachute payments for time vesting stock options and restricted stock were valued using Internal Revenue Code Treas. Reg. Section 1.28G-1 Q&A 24(c)); (iv) we assumed an 8% annual increase in medical premiums, 5% annual increase in dental premiums, 4% increase in vision premiums, and no increase in disability or life insurance premiums or employer cost of tax and financial planning advisory services for 2016 and 2017; (v) we assumed that the medical insurance benefits received in 2016 and 2017 would be taxable and that the Named Officers would be eligible for a tax gross-up for these benefits under the terms of the change in control severance plan; (vi) although the change in control severance plan provides for restrictive covenants, including a one-year covenant prohibiting the solicitation of company employees, no specific value has been ascribed to these covenants; and (vii) although certain payments to the Named Officers would be subject to potential delays upon separation of service under Section 409A of the Internal Revenue Code, we did not attempt to determine which, if any, payments would be delayed.
69
Executive Compensation
Corporate Governance Aspects of the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan
The Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan (referred to in this subsection as the "Plan") is the only plan currently used by the Company to grant equity awards. It has been designed to include a number of provisions that promote best practices by reinforcing the alignment
between equity compensation arrangements for eligible employees and non-employee directors, on the one hand, and shareholders' interests, on the other hand. These provisions include:
|
Provision |
Description |
|
| No Discounted Stock Options | Stock options are not granted with an exercise price less than the fair market value of common stock (as defined in the Plan) on the date of grant. | |
| No Stock Option Re-pricing or Exchange | Except for equitable adjustments in connection with specific corporate transactions (such as stock splits, recapitalizations, reorganizations, mergers, consolidations, and similar transactions), the Plan does not permit a decrease in the exercise price of a stock option granted under the Plan through settlement, cancellation, forfeiture, exchange, surrender, or otherwise below the fair market value of common stock (as defined in the Plan) on the date of grant. | |
| Minimum Vesting Requirements |
If the vesting condition for any equity award (other than an option or a stock appreciation right) made to an employee is based solely upon continued employment, the regular vesting may not be more favorable to the employee than in equal annual
increments over 36 months. If the vesting condition for any equity award (other than an option or a stock appreciation right) made to an employee is based upon the attainment of specified performance measures, the regular performance vesting period may not be less than one year. The Compensation Committee's discretion to deviate from the minimum vesting requirements described above is limited to accelerated vesting upon a change of control or upon a termination of the employee's employment and with respect to grants not in excess of 1,000,000 shares in the aggregate under the Plan. |
|
| Recoupment Policy | Awards granted to our officers and other specified employees under the Plan are subject to recoupment or reduction in accordance with the terms of our policy regarding recoupment or reduction of incentive compensation. | |
| Independent Administration | The Plan is administered by the Compensation Committee, which is intended to be comprised solely of non-employee directors each of whom meets the additional independence criteria applicable to compensation committee members under the listing standards of The NASDAQ Stock Market LLC, qualifies as a "Non-Employee Director" pursuant to Rule 16b-3 under the Exchange Act, and meets the requirements for an "outside director" within the meaning of Section 162(m) of the Internal Revenue Code. | |
| No "Evergreen" Provision | The Plan does not contain an "evergreen" feature pursuant to which the shares authorized for issuance thereunder can be automatically replenished. | |
| No Tax Gross-ups | The Plan does not provide for any tax gross-ups. |
70
Executive Compensation
Equity Compensation Plan Information
The following table shows information with respect to securities authorized for issuance under the equity compensation plans maintained by the Company as of December 31, 2015.
Plan Category |
(a) Number of securities to be issued upon exercise of outstanding options, warrants, and rights |
|
(b) Weighted-average exercise price of outstanding options, warrants, and rights |
(c) Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) |
|||
Equity compensation plans approved by security holders 1 |
23,165,769 shares of common stock |
$ |
236.75 |
9,711,439 shares of common stock 3 |
|||
Equity compensation plans not approved by security holders 2 |
– | $ | – |
44,246 shares of Class A stock |
|||
| | | | | | | | |
Total |
23,165,769 shares of common stock |
$ | 236.75 |
9,755,685 shares of common stock and Class A stock |
|||
| | | | | | | | |
- 1
- The equity compensation plans approved by the security holders are the Regeneron Pharmaceuticals, Inc. Second Amended and Restated 2000 Long-Term Incentive Plan and the Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan. The Regeneron Pharmaceuticals, Inc. 2014 Long-Term Incentive Plan is the only plan currently used by the Company to grant equity awards.
- 2
- The equity compensation plan not approved by the security holders is the Executive Stock Purchase Plan. It was adopted in 1989 and provides for the Compensation Committee of the board of directors to award employees, directors, consultants, and other individuals who render service to the Company the right to
purchase Class A stock at a price set by the Compensation Committee. The Plan provides for the vesting of shares as determined by the Compensation Committee; should the Company's relationship with a Plan participant terminate before all shares are vested, unvested shares will be repurchased by the Company at a price per share equal to the original amount paid by the Plan participant. As of December 31, 2015, there were no unvested shares and 44,246 shares of Class A stock available for future grants under the Plan.
- 3
- Gives effect to 541,700 outstanding shares of restricted stock. As these shares are considered issued and outstanding upon grant, they are not included in the amounts reported in column (a).
71
Executive Compensation
![]()
When are shareholder proposals due for the 2017 Annual Meeting of Shareholders?
A shareholder wishing to present a proposal at the 2017 Annual Meeting of Shareholders must submit the proposal in writing and it must be received by the Company at its principal executive offices at 777 Old Saw Mill River Road, Tarrytown, New York 10591-6707 by December 28, 2016, and must satisfy the other conditions established by the SEC, in order for such proposal to be considered for inclusion in the Company's proxy statement and form of proxy relating to that meeting.
Under our by-laws, proposals of shareholders intended to be submitted for a formal vote (other than proposals to be included in our proxy statement) at the 2017 Annual Meeting may be made only by a shareholder of record who has given notice of the proposal to the Secretary of the Company at our principal executive offices no earlier than 90 days and no later than 60 days prior to the meeting; provided that if less than 70 days' notice or public disclosure of the date of the 2017 Annual Meeting is given or made to shareholders, notice by the shareholder in order to be timely must be received no later than the close of business on the tenth day following the day on which such notice of the annual meeting was first mailed or such public disclosure of the annual meeting was made, whichever first occurs. The notice must contain certain information as specified in our by-laws. Assuming our 2017 Annual Meeting is held on June 9, 2017 in accordance with the Company's past practice, and at least 70 days' notice or prior public disclosure of the date of the 2017 Annual Meeting is given or made to shareholders, notice of such proposals would need to be given no earlier than March 11, 2017 and no later than April 10, 2017. Any proposal received outside of such dates will not be considered "timely" under the federal proxy rules for purposes of determining whether we may use discretionary authority to vote on such proposal.
What happens if multiple shareholders share an address?
Applicable rules permit brokerage firms and the Company to send one Notice of Internet Availability of Proxy Materials (or one annual report, proxy statement, and Notice of Internet Availability of Proxy Materials in the case of shareholders who have elected to receive paper copies of our proxy materials) to multiple shareholders who share the same address under certain circumstances. This practice is known as "householding." We believe that householding will provide greater convenience for our shareholders, as well as cost savings for us by reducing the number of duplicate documents that are sent to your home. Consequently, we have implemented the practice of householding for shares held in "street name" and intend to deliver only one copy of the applicable proxy materials to
multiple shareholders sharing the same address. If you wish to receive separate copies of the proxy statement for the 2016 Annual Meeting, the 2015 Annual Report, or the Notice of Internet Availability of Proxy Materials, you may find these materials at our internet website (www.regeneron.com) or you may stop householding for your account and receive separate printed copies of these materials by contacting our Investor Relations Department, at Regeneron Pharmaceuticals, Inc., 777 Old Saw Mill River Road, Tarrytown, New York 10591-6707, or by calling us at 914-847-7000, and these materials will be promptly delivered to you. If you hold shares registered in your name (sometimes called a shareholder of record), you can elect householding for your account by contacting us in the same manner described above. Any shareholder may stop householding for your account by contacting our Investor Relations Department at the address and/or phone number included above. If you revoke your consent, you will be removed from the householding program within 30 days of receipt of your revocation and each shareholder at your address will receive individual copies of our disclosure documents.
Are there any other matters to be addressed at the Annual Meeting?
We know of no other matters to be brought before the Annual Meeting, except as set forth in this proxy statement. If any other matter is properly presented at the Annual Meeting upon which a vote may properly be taken, shares represented by duly executed and timely submitted proxies will be voted on any such matter in accordance with the judgment of the persons named as proxies in the enclosed proxy card. Discretionary authority for them to do so is contained in the enclosed proxy card.
Who will pay the costs related to this proxy statement and the Annual Meeting?
The solicitation of proxies is being made on behalf of the Company and we will bear the costs of the solicitation. We will be responsible for paying for all expenses to prepare, print, and mail the proxy materials to shareholders. In accordance with the regulations of the SEC, we will make arrangements with brokerage houses and other custodians, nominees, and fiduciaries to send proxies and proxy materials to their principals and will reimburse them for their reasonable expenses in so doing. In addition to the solicitation by use of the mails and the Internet, certain of our officers, directors, and employees may solicit the return of proxies by telephone, e-mail or personal interviews.
72
Other Matters
How can you receive a printed copy of the Company's 2015 Annual Report?
Interested shareholders may obtain without charge a copy of our 2015 Annual Report (without exhibits), which includes our audited financial statements for the fiscal year ended December 31, 2015, required to be filed with the SEC, by making a written request to Regeneron Pharmaceuticals, Inc., 777 Old Saw Mill River Road, Tarrytown, New York 10591-6707, Attention: Investor Relations, or by calling our Investor Relations Department at (914) 847-7000.
How do you elect to receive future proxy materials electronically?
If you previously requested to receive proxy materials through the mail, or by means of an e-mail with links to the proxy materials and the proxy voting website, your election will remain in effect until you revoke it. Shareholders currently receiving paper copies of our proxy materials, and shareholders who received a paper copy of the Notice of Internet
Availability of Proxy Materials, may instead elect to receive all future proxy materials electronically through an e-mail with a link to these documents on the Internet. Receiving these documents online conserves resources, saves the Company the cost of producing and mailing documents to your home or business, and gives you an automatic link to the proxy voting site.
If your shares are registered in your name or you hold shares in the Company Stock Fund in the Company's 401(k) Savings Plan, to enroll in the electronic delivery service, vote your shares through the Internet at www.proxyvote.com and, when prompted, indicate that you agree to receive or access shareholder communications electronically in future years. If your shares are not registered in your name, to enroll in the electronic delivery service, check the information provided to you by your bank or broker, or contact your bank or broker for instructions on how to elect to view future proxy statements and annual reports over the Internet.
73
Other Matters
![]()
Appendix A Note Regarding Forward-Looking Statements andNon-GAAP Financial Measures
This proxy statement includes forward-looking statements that involve risks and uncertainties relating to future events and the future performance of Regeneron Pharmaceuticals, Inc. ("we," "us," "our," "Regeneron," or the "Company"), and actual events or results may differ materially from these forward-looking statements. Words such as "anticipate," "expect," "intend," "plan," "believe," "seek," "estimate," variations of such words and similar expressions are intended to identify such forward-looking statements, although not all forward-looking statements contain these identifying words. These statements concern, and these risks and uncertainties include, among others, the nature, timing, and possible success and therapeutic applications of our products, product candidates, and research and clinical programs now underway or planned, including without limitation EYLEA® (aflibercept) Injection, Praluent® (alirocumab) Injection, sarilumab, dupilumab, fasinumab, REGN 2222, and the immuno-oncology program; unforeseen safety issues resulting from the administration of products and product candidates in patients, including serious complications or side effects in connection with the use of our product candidates in clinical trials; the likelihood and timing of possible regulatory approval and commercial launch of our late-stage product candidates and new indications for marketed products, including without limitation EYLEA®, Praluent®, sarilumab, dupilumab, fasinumab, and REGN 2222; ongoing regulatory obligations and oversight impacting our marketed products (such as EYLEA® and Praluent®), research and clinical programs, and business, including those relating to patient privacy; determinations by regulatory and administrative governmental authorities which may delay or restrict our ability to continue to develop or commercialize our products and product candidates; competing drugs and product candidates that may be superior to our products and product candidates; uncertainty of market acceptance and commercial success of our products and product candidates; our ability to manufacture and manage supply chains for multiple products and product candidates; coverage and reimbursement determinations by third-party payers, including Medicare and Medicaid; unanticipated expenses; the costs of developing, producing, and selling products; our ability to meet any of its sales or other financial projections or guidance and changes to the assumptions underlying those projections or guidance; the potential for any license or collaboration agreement, including our agreements with Sanofi and Bayer HealthCare LLC, to be cancelled or terminated without any further product success; and risks associated with third party intellectual property and
pending or future litigation relating thereto. A more complete description of these and other material risks can be found in our filings with the Securities and Exchange Commission, including our Annual Report on Form 10-K for the fiscal year ended December 31, 2015 (filed with the Securities and Exchange Commission on February 11, 2016), including in the section thereof captioned "Item 1A. Risk Factors." Any forward-looking statements are made based on management's current beliefs and judgment, and the reader is cautioned not to rely on any forward-looking statements made by Regeneron. We do not undertake any obligation to update publicly any forward-looking statement, including without limitation any financial projection or guidance, whether as a result of new information, future events, or otherwise.
This proxy statement uses non-GAAP net income, which is a financial measure that is not calculated in accordance with U.S. Generally Accepted Accounting Principles ("GAAP"). We believe that the presentation of this non-GAAP measure is useful to investors because it excludes (i) non-cash share-based compensation expense, which fluctuates from period to period based on factors that are not within the Company's control, such as the Company's stock price on the dates share-based grants are issued; (ii) the incremental charge recorded in the third quarter of 2014 related to the issuance of the final IRS regulations that provide guidance on the annual fee imposed by the Patient Protection and Affordable Care Act (the final IRS regulations differed from the temporary regulations issued in 2011 which resulted in the recognition of a catch-up adjustment); (iii) non-cash interest expense related to the Company's convertible senior notes, since this is not deemed useful in evaluating the Company's operating performance; (iv) loss on extinguishment of debt, since this non-cash charge is based on factors that are not within the Company's control; and (v) income tax expense for 2014, which was principally a non-cash expense due primarily to utilization of net operating loss and tax credit carryforwards, and deductions related to employee stock option exercises. In 2015, income tax expense adjustments consider the tax effect of reconciling items and an adjustment from GAAP tax expense to the amount of taxes that are paid or payable in cash in respect of the current period. As there is a significant difference between the Company's effective tax rate and actual cash income taxes paid or payable, GAAP income tax expense is not deemed useful in evaluating the Company's operating performance. Management uses these non-GAAP measures for planning, budgeting, forecasting, assessing historical performance, and making financial
A-1
Appendix A Note Regarding Forward-Looking Statements and Non-GAAP Financial Measures
and operational decisions, and also provides forecasts to investors on this basis. However, there are limitations in the use of these and other non-GAAP financial measures as they exclude certain expenses that are recurring in nature. Furthermore, the Company's non-GAAP financial measures may not be comparable with non-GAAP information provided by other
companies. Any non-GAAP financial measure presented by Regeneron should be considered supplemental to, and not a substitute for, measures of financial performance prepared in accordance with GAAP. A reconciliation of our GAAP to non-GAAP results is included below.
Reconciliation of GAAP Net Income to Non-GAAP Net Income
(Unaudited)
(In thousands)
|
|
Year ended December 31, |
|||||
| | | | | | | | |
|
|
2015 |
| 2014* | |||
GAAP net income |
$ | 636,056 | $ | 338,126 | |||
Adjustments: |
| | |||||
R&D: Non-cash share-based compensation expense |
| 255,708 | | 184,347 | |||
SG&A: Non-cash share-based compensation expense |
| 193,026 | | 134,715 | |||
SG&A: Branded Prescription Drug Fee incremental charge |
| — | | 40,600 | |||
COGS and COCM: Non-cash share-based compensation expense |
| 10,315 | | 2,688 | |||
Interest expense: Non-cash interest related to convertible senior notes |
| 2,818 | | 17,821 | |||
Other expense: Loss on extinguishment of debt |
| 18,861 | | 33,469 | |||
Non-cash income taxes |
| 287,110 | | 423,109 | |||
| | | | | | | | |
Non-GAAP net income |
$ | 1,403,894 | $ | 1,174,875 | |||
| | | | | | | | |
- *
- Certain revisions have been made to the amounts originally reported for the year ended December 31, 2014 (see Note 1 to the Company's audited financial statements for the fiscal year ended December 31, 2015 included in its Annual Report on Form 10-K for the year ended December 31, 2015).
A-2
Appendix A Note Regarding Forward-Looking Statements and Non-GAAP Financial Measures
|
|
REGENERON 777 OLD SAW MILL RIVER ROAD TARRYTOWN, NY 10591-6707 |
|
|
REGENERON 777 OLD SAW MILL RIVER ROAD TARRYTOWN, NY 10591-6707 |
If you would like to reduce the costs incurred by our company in mailing proxy 1234567 VOTE BY MAIL 123,456,789,012.12345 TO VOTE, MARK BLOCKS BELOW IN BLUE OR BLACK INK AS FOLLOWS: KEEP THIS PORTION FOR YOUR RECORDS DETACH AND RETURN THIS PORTION ONLY THIS PROXY CARD IS VALID ONLY WHEN SIGNED AND DATED. The Board of Directors recommends you vote FOR the following: 1. Election of Directors Nominees For 0 0 0 For 0 Against 0 0 0 Against 0 Abstain 0 0 0 Abstain 0 01 Michael S. Brown 02 Leonard S. Schleifer 03 George D. Yancopoulos The Board of Directors recommends you vote FOR the following proposal: 2 Ratification of the appointment of PricewaterhouseCoopers LLP as the Company's independent registered public accounting firm for the fiscal year ending December 31, 2016. NOTE: In their discretion, the named proxies may vote on such other matters as may properly come before the meeting and any adjournment(s) or postponement(s) thereof. John Sample attorney, executor, administrator, or other fiduciary, please give full ANY CITY, ON A1A 1A1 partnership name, by authorized officer. Signature [PLEASE SIGN WITHIN BOX] Date Signature (Joint Owners) Date 02 0000000000 1 OF 1 1 2 0000282794_1 R1.0.1.25 Please sign exactly as your name(s) appear(s) hereon. When signing as title as such. Joint owners should each sign personally. All holders must sign. If a corporation or partnership, please sign in full corporate or Investor Address Line 1 Investor Address Line 2 Investor Address Line 3 Investor Address Line 4 Investor Address Line 5 1234 ANYWHERE STREET SHARES CUSIP # JOB #SEQUENCE # VOTE BY INTERNET - www.proxyvote.com Use the Internet to transmit your voting instructions and for electronic delivery of information up until 11:59 P.M. Eastern Time the day before the cut-off date or meeting date. Have your proxy card in hand when you access the web site and follow the instructions to obtain your records and to create an electronic voting instruction form. ELECTRONIC DELIVERY OF FUTURE PROXY MATERIALS materials, you can consent to receiving all future proxy statements, proxy cards and annual reports electronically via e-mail or the Internet. To sign up for electronic delivery, please follow the instructions above to vote using the Internet and, when prompted, indicate that you agree to receive or access proxy materials electronically in future years. VOTE BY PHONE - 1-800-690-6903 Use any touch-tone telephone to transmit your voting instructions up until 11:59 John Sample 23456 7P.M. Eastern Time the day before the cut-off date or meeting date. Have your proxy card in hand when you call and then follow the instructions. 1234567 Mark, sign and date your proxy card and return it in the postage-paid envelope we have provided or return it to Vote Processing, c/o Broadridge, 51 Mercedes Way, Edgewood, NY 11717. NAME THE COMPANY NAME INC. - COMMON THE COMPANY NAME INC. - CLASS A THE COMPANY NAME INC. - CLASS B THE COMPANY NAME INC. - CLASS C THE COMPANY NAME INC. - CLASS D THE COMPANY NAME INC. - CLASS E THE COMPANY NAME INC. - CLASS F THE COMPANY NAME INC. - 401 K CONTROL # SHARES 123,456,789,012.12345 123,456,789,012.12345 123,456,789,012.12345 123,456,789,012.12345 123,456,789,012.12345 123,456,789,012.12345 123,456,789,012.12345 x PAGE 1 OF 2 REGENERON PHARMACEUTICALS, INC. 777 OLD SAW MILL RIVER ROAD TARRYTOWN, NY 10591-6707 Investor Address Line 1 Investor Address Line 2 Investor Address Line 3 Investor Address Line 4 Investor Address Line 5 8 8 8 1 1234 ANYWHERE STREET ANY CITY, ON A1A 1A1 234567 234567 234567 234567

Important Notice Regarding the Availability of Proxy Materials for the Annual Meeting: The Notice & Proxy Statement, Annual Report is/ are available at www.proxyvote.com REGENERON PHARMACEUTICALS, INC. Annual Meeting of Shareholders June 10, 2016 10:30 AM This proxy is solicited by the Board of Directors The undersigned hereby appoints Leonard S. Schleifer, M.D., Ph.D. and Joseph J. LaRosa, and each of them individually, as lawful proxies, each with full power of substitution, to represent the undersigned, with all powers that the undersigned would possess if personally present, and to vote, as indicated on the reverse side of this card, all shares of Common Stock and Class A Stock of Regeneron Pharmaceuticals, Inc. that the undersigned would be entitled to vote if personally present, at the Annual Meeting of Shareholders of the Company to be held on June 10, 2016 or at any adjourned or postponed session thereof. This proxy revokes all prior proxies given by the undersigned. SHARES REPRESENTED BY THIS PROXY WILL BE VOTED AT THE MEETING AND AT ANY ADJOURNMENTS OR POSTPONEMENTS THEREOF IN ACCORDANCE WITH THE SHAREHOLDER'S SPECIFICATIONS ABOVE. IF YOU SIGN AND RETURN YOUR PROXY CARD IN A TIMELY MANNER, BUT DO NOT INDICATE HOW THESE SHARES ARE TO BE VOTED, THIS PROXY WILL BE VOTED FOR THE ELECTION OF THE NOMINEES NAMED ABOVE AS DIRECTORS AND FOR PROPOSAL 2. THIS PROXY ALSO CONFERS DISCRETIONARY AUTHORITY WITH RESPECT TO MATTERS NOT KNOWN OR DETERMINED AT THE TIME OF THE MAILING OF THE NOTICE OF THE ANNUAL MEETING OF SHAREHOLDERS TO THE UNDERSIGNED. Continued and to be signed on reverse side 0000282794_2 R1.0.1.25












