Forward Looking Statements This presentation contains forward-looking information within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, and other securities laws. Forward-looking statements are statements that are not historical facts. Words such as “will,” “underway,” “allow,” “expect(ed),” “pursuing,” “may,” “would,” “addressing,” “creating,” “intends,” “anticipate(s),” “plan,” “partner,” “could,” “enables,” “potential(ly),” and similar expressions are intended to identify forward-looking statements. These statements include, but are not limited to, statements regarding future events such as: the continued commercial performance of our marketed products, including but not limited to BENDEKA, which is marketed by our partner Teva, Argatroban, which is marketed by Chiesi USA and Sandoz pursuant to separate agreements and Ryanodex, which we market ourselves, as well as our ability to replicate our marketing successes for our other product candidates such as Ryanodex for EHS or other additional indications, our pemetrexed product candidate, or our fulvestrant product candidate, either through joint or direct marketing efforts; the potential lack of a need for human safety and efficacy data for the submission of an NDA for Ryanodex and the adequacy of the regulatory pathway to complete an NDA submission; the Company’s share repurchase authorization and timing and ability to continue to repurchase shares of the Company’s common stock under a share repurchase program; the business path forward for the Company between now and beyond 2026; the label expansions of Ryanodex for EHS patients and for the treatment of ecstasy and methamphetamine intoxication and for the treatment of neurological impact and nerve agent exposure; the strength of the Company’s cash position and the ability to optimize the deployment of capital and take advantage of market opportunities; the potential of the Company’s pipeline to drive value between now and beyond 2026; the contribution of the Ryanodex portfolio to the Company’s growth; the timing of Ryanodex for EHS obtaining FDA approval, if ever, and, entering the market; and the advancement of any of the Company’s other product candidates including but not limited to fulvestrant and pemetrexed, through the development process including FDA approval and the ability of any such products to have commercial success and to access significant new markets; and our ability to use the acquisition of Arsia Therapeutics (now Eagle Biologics) to enter into the biologics market and to effectively carry out our strategy in this new market. All of such statements are subject to certain risks and uncertainties, many of which are difficult to predict and generally beyond Eagle’s control, that could cause actual results to differ materially from those expressed in, or implied or projected by, the forward-looking information and statements. Such risks include, but are not limited to: whether our animal studies will support the safety and efficacy of Ryanodex for the treatment of EHS, ecstasy and methamphetamine intoxication, and neurological impact of nerve gas exposure; whether the FDA will ultimately approve Ryanodex for these indications; whether the FDA will approve our application for pemetrexed, and, if filed, fulvestrant; fluctuations in the trading volume and market price of shares of the Company's common stock, general business and market conditions and management's determination of alternative needs and uses of the Company's cash resources which may affect the Company's share repurchase program; the success of our commercial relationship with Teva and our other marketing partners and the parties’ ability to work effectively together; whether Eagle and Teva and our other marketing partners will successfully perform their respective obligations under their agreements with us; difficulties or delays in manufacturing; the availability and pricing of third party sourced products and materials; the outcome of litigation involving any of our products or product candidates or that may have an impact on any of our products or product candidates, successful compliance with FDA and other governmental regulations applicable to product approvals, manufacturing facilities, products and/or businesses; general economic conditions; the strength and enforceability of our intellectual property rights or the rights of third parties; competition from other pharmaceutical and biotechnology companies; the timing of product launches; the successful marketing of our products; the risks inherent in the early stages of drug development and in conducting clinical trials; and other factors that are discussed in Eagle’s Annual Report on Form 10-K for the year ended December 31, 2016, its Quarterly Reports on Form 10-Q for each of the quarters ended March 31, 2017 and June 30, 2017 and its other filings with the U.S. Securities and Exchange Commission. Readers are cautioned not to place undue reliance on these forward-looking statements that speak only as of the date hereof, and we do not undertake any obligation to revise and disseminate forward-looking statements to reflect events or circumstances after the date hereof, or to reflect the occurrence of or non-occurrence of any events. 2

Eagle 2017 At A Glance 3 Driving long term value Launched Bendeka: 97% market conversion Bendamustine Unique J-Code effective January 1, 2017 13 Orange Book Listed patents for Bendeka Sued three ANDA filers for patent infringement Promoted by Eagle salesforce; pursuing add’l indications Ryanodex R&D for additional indications and administration routes Received CRL on EHS from the FDA on July 26, 2017 Phase II clinical trial for MDMA/meth intoxication underway Potential next indication: treatment of neurological impact of exposure to nerve agents NDA accepted for filing; October 30, 2017 PDUFA date Pemetrexed Began patent litigation Innovative formulation offers opportunity similar to Bendeka Fulvestrant Positive regulatory feedback Pivotal trial dosing expected to begin Q4 2017 Acquisition provides entry into fastest growing pharma sector Eagle Biologics “Biobetter” product potential Orphan Drug Designation In litigation for ODE Licensed to SymBio in Japan
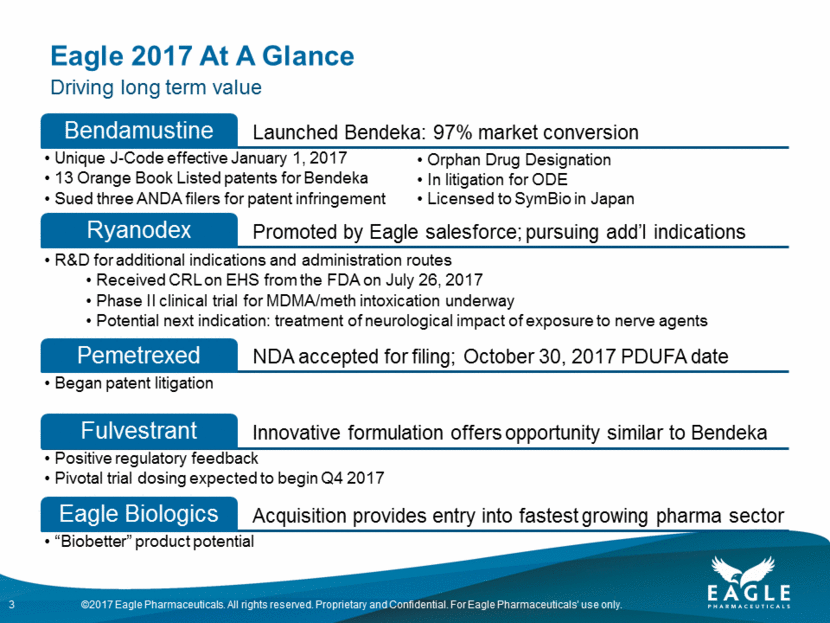
Bendeka: Long Life Cycle 4 2016 97% market share Unique J-code Royalty increase from 20% to 25% 8 newly listed patents through 2033 $89mm in royalties earned in 2016 $89mm in milestones earned 2015/2016 Now - 2019 $70mm in royalties earned in 1H2017 Royalty of 25% of US net sales $25mm milestone earned in 2017 US and expansion to OUS markets (20% royalty) 2020 – 2026 6 years of royalty of 25% net sales Assuming large market share held post generic Treanda® entry Improved product profile and J-code
Thirteen Orange Book Patents Running from 2026-2033 5 U.S. Patent No. Patent Expiration 8,609,707 1/28/2031 8,791,270*PED (owned by Teva Pharmaceutical Industries Ltd.) 7/12/2026 9,000,021 3/15/2033 9,034,908 3/15/2033 9,144,568 3/15/2033 9,265,831 1/28/2031 9,572,796 1/28/2031 9,572,797 1/28/2031 9,572,887 3/15/2033 9,579,384 3/15/2033 9,579,397 3/15/2033 9,579,398 3/15/2033 9,579,399 3/15/2033 Protecting the longevity of the bendamustine franchise Eagle/Teva asserting all patents challenged by ANDA filers

Develop, market and sell Eagle’s bendamustine hydrochloride ready-to-dilute (RTD) and rapid infusion injection (RI) products Begins process to maximize value of Eagle’s product portfolio worldwide $12.5 million upfront payment plus future potential milestones and royalty payments SymBio will be responsible for securing regulatory approval of the RTD and RI injection in Japan Target for approval of a product in 2020 SymBio markets TREAKISYM® in Japan, a lyophilized powder formulation of bendamustine HCl for chronic lymphocytic leukemia (CLL); relapsed or refractory low-grade Hodgkin’s lymphoma (NHL); mantle cell lymphoma (MCL); and as a first line treatment of low-grade NHL and MCL 12-month sales ended June 30, 2017 for TREAKISYM in Japan were $52 million SymBio has estimated that sales of TREAKISYM will grow to $90 million in 2018 Symbio is conducting a Ph 3 clinical trial for relapsed/refractory diffuse large B-cell lymphoma Licensed Japanese Rights for Bendamustine Hydrochloride Products to SymBio Pharmaceuticals Limited 6

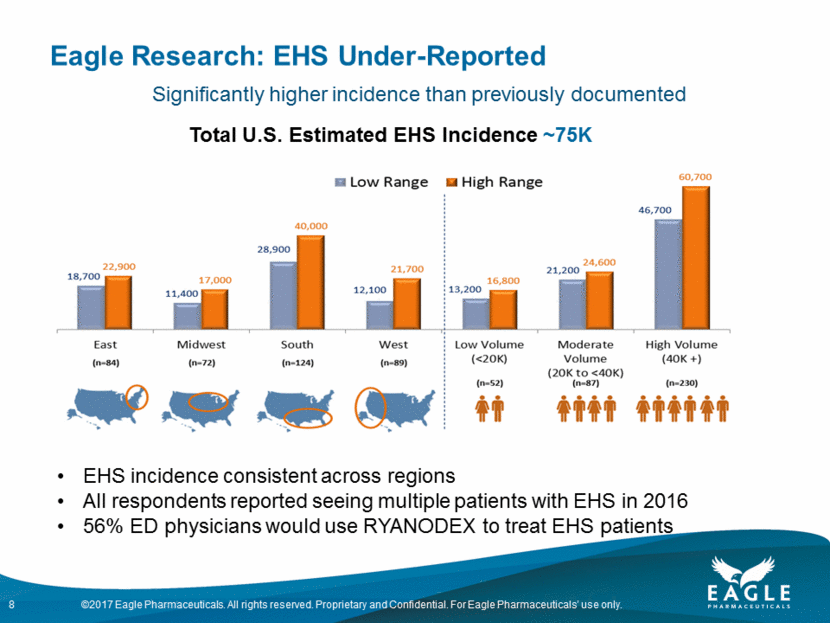
RYANODEX®: Multiple Label Expansion Opportunities Creating additional value by addressing life-threatening, unmet needs Breakthrough formulation of dantrolene sodium Approved in July 2014; launched in August 2014 Potential to be the first drug to market for EHS Type A meeting scheduled with FDA Vigorously pursuing approval Total U.S. Estimated EHS Incidence ~75K Orphan Drug Designation Preclinical studies by NIDA/NIH completed Phase II clinical trial for MDMA & meth intoxication underway Broadened endpoint to include severe organ dysfunction and damage 125K U.S. ED visits in 2011 due to MDMA & meth intoxication New patent allowed Marketed Potential Label Expansion Malignant Hyperthermia Exertional Heat Stroke MDMA & Methamphetamine Intoxication 7 Nerve Agent Treatment of neurological impact of nerve agent exposure as potential next indication Positive results of an initial study to evaluate the neuroprotective effects of RYANODEX® in an established rodent model Plan to meet with FDA to discuss next steps

Eagle Research: EHS Under-Reported 8 Total U.S. Estimated EHS Incidence ~75K EHS incidence consistent across regions All respondents reported seeing multiple patients with EHS in 2016 56% ED physicians would use RYANODEX to treat EHS patients Significantly higher incidence than previously documented
Received a CRL from the FDA on July 26, 2017 Type A meeting with FDA in September Label expansion for an already approved drug No safety or CMC issues Priority review and Fast Track designation We believe the results of our human and animal studies are consistent with what the FDA requested and previous animal pilot work performed Our independent consultants agree with our conclusion that we have met all of our commitments and that the data is quite strong and supports approval Availing ourselves of mechanisms available to us: dispute process possible Believe strongly in RYANODEX given that there is no pharmacological option available for patients in need for this life-threatening condition RYANODEX® for Exertional Heat Stroke 9

References: 1. Center for Behavioral Health Statistics and Quality, SAMHSA, Drug Abuse Warning Network, National Estimates of Drug-Related Emergency Department Visits, 2011. 2. E Musselman M, Saely S. Diagnosis and treatment of drug-induced hyperthermia. Am J Health-Syst Pharm. 2013 Vol 70. Use of illegal stimulants constitutes a growing public health problem in the US and EU1 Over 125,000 ED visits related to MDMA (ecstasy) and methamphetamine use in the US alone (2011)1 ED visits involving MDMA among patients 21 years and younger grew over 128% between 2005-20111 In 2011, 42% of ED visits among people 18-29 years old involved illicit stimulant drugs1 Associated with body and brain hyperthermia, high incidence of severe cardiovascular and neurologic complications, and lifetime neurologic sequelae2 Can be fatal or lead to permanent damage if not treated promptly2 MDMA (Ecstasy) and Methamphetamine Intoxication 10

MDMA (Ecstasy) & Methamphetamine Intoxication: Positive pre-IND Meeting with FDA FDA suggested and we are broadening the indication to evaluate ‘organ damage and/or severe dysfunction’ in patients with MDMA and Methamphetamine intoxication No additional preclinical work required to support the efficacy of RYANODEX for this indication A single robust, controlled and well powered clinical trial may be sufficient for filing the NDA Phase II clinical trial for MDMA & Methamphetamine intoxication underway; anticipate pivotal study to begin Q4 2017, if needed Patent directed to this indication allowed in August 2017 11

Pemetrexed Opportunity At this time, Lilly’s Alimta patent infringement lawsuit prevents current ANDA filers from launching until May 24, 2022 Eagle’s Pemetrexed RTD NDA was filed December 2016 NDA accepted with PDUFA date of October 30, 2017 Lilly sued Eagle on August 14, 2017 There are three 505(b)(2) filers (DRL, Hospira, Actavis/Teva) with a similar approach to Eagle’s (ours is a differentiated product) DRL and Hospira both filed Motions for Summary Judgment of Noninfringement in late July 2017 (Lilly’s opposition to DRL’s motion due on or before 9/25/17) Eagle continues evaluating all litigations and outcomes $1.1B market opportunity1 12 1 Alimta® (pemetrexed) (Eli Lilly & Co.). Source: Eli Lilly & Co. Q2 2016 earnings for MAT 12 mos. ending 6/30/16: (U.S. sales)

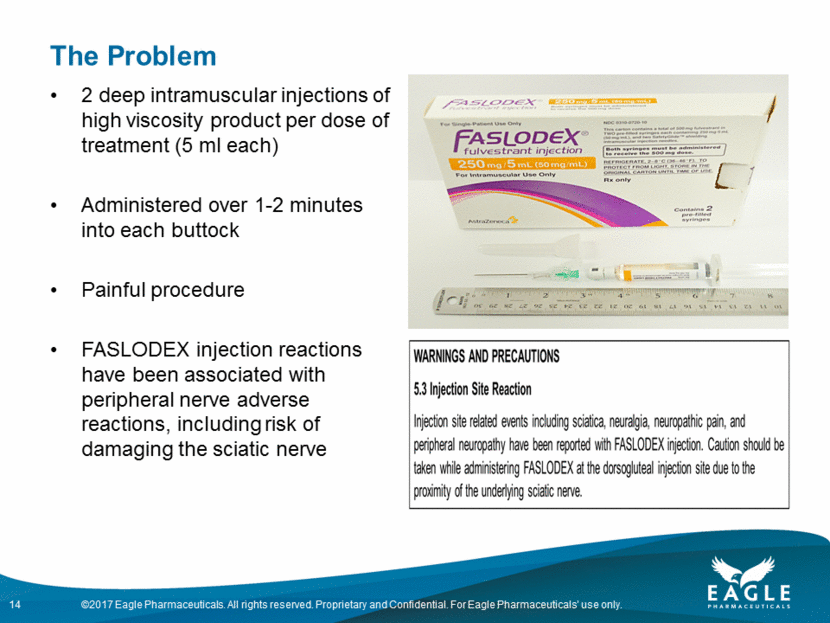
Fulvestrant Opportunity INDICATIONS for FASLODEX ® Monotherapy FASLODEX is indicated for the treatment of hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced breast cancer in postmenopausal women not previously treated with endocrine therapy, or HR-positive advanced breast cancer in postmenopausal women with disease progression following endocrine therapy Combination Therapy FASLODEX in combination with palbociclib is indicated for the treatment of HR-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced or metastatic breast cancer in women with disease progression after endocrine therapy. Currently marketed by AstraZeneca Administered monthly in a doctor’s office as 2 separate intramuscular injections, one in each buttock FDA has recently required revising the FASLODEX label 13 Faslodex ® is a registered trademark of AstraZeneca. Label expanded in the US to first line breast cancer in August 2017

The Problem 2 deep intramuscular injections of high viscosity product per dose of treatment (5 ml each) Administered over 1-2 minutes into each buttock Painful procedure FASLODEX injection reactions have been associated with peripheral nerve adverse reactions, including risk of damaging the sciatic nerve 14
Positive Regulatory Feedback from FDA regarding Fulvestrant Clinical Protocol Pre-IND meeting responses are aligned with Eagle’s development plan A single PK and safety study in healthy female volunteers may be sufficient for filing Expect study to recruit and complete within 12 months FDA has agreed to consider not including Injection Site Reaction (Warning & Precautions) language in Eagle’s label, provided that our clinical data demonstrates a significant safety improvement 15

Eagle’s Solution Innovative formulation administered in far less time (seconds) vs. 1-2 minutes per injection Eagle formulation may allow warning on insert to be eliminated Eagle formulation does not contain castor oil 1X Low viscosity 5mL dose 2X High viscosity 5mL doses vs. 16

FASLODEX Market Opportunity $400mm+ O.U.S. Source: AstraZeneca Financial Summary Full Year and Q4 2016 & AstraZeneca H1 2017 Results Results)www.londonstockexchange.com/exchange/news/market-news/market-news 17 12 months ended June 30th 2017 total sales of the branded form of fulvestrant, FASLODEX, were up 15% to $891 million worldwide 16% growth in US sales to $468mm 13% growth in EU sales to $248mm 22% growth in Established ROW sales to $72mm 12% growth in Emerging Markets sales to $103mm
Eagle Biologics (Arsia Acquisition) Marks Eagle’s entry into Biologics Fastest growing pharmaceutical sector Enhances Eagle’s formulation capabilities and expands product development opportunities Secures a patent portfolio of viscosity-reducing technology Provides access to leading minds in the field who will work on additional Eagle formulations Extends Eagle’s strategy: plan to partner with key Biosimilar or Bioinnovator companies to alter their existing pipeline into “Biobetters” $78 million investment: largely dependent upon achievement of milestones 18

Biologics Market Opportunity The global biologics market could exceed $390 billion in value over the next five years1 Growing at nearly 2X the rate of pharma1 By the end of 2020, biologics could account for 28% of the global pharmaceuticals market1 The global biosimilar market may reach $20 - $26 billion by 20202 19 References: 1. PRA Health Sciences Whitepaper. The Value of Biobetters. December 2015. 2. IMS Medicines Use and Spending in the U.S. – A Review of 2015 and Outlook to 2020. April 2015.

Financial Highlights LTM 6/30/17 EBITDA: $96mm 6/30/17: $55.4mm cash; $53.2mm A/R; no debt Share Repurchase Plan $75.8mm repurchased (1.1mm shares at $65.91) since August 2016 $100mm additional authorization approved by the Board August 2017 14.9mm basic shares outstanding as of 9/05/17 $150mm credit facility August 2017 $100mm term loan (of which $50mm was drawn 8/8/17) $50mm revolver 20

Poised for Continued Growth Beyond 2020 Ongoing Reinvestment of Cash Including Additional $100 Million Share Repurchase 21 Potential for multiple in-market products over the long-term 2017 - 2019 2020 - 2026 2016 2026+ Multiple in-market products Use of Capital R&D success Bendeka US & WW Ryanodex for MH, EHS, MDMA & Methamphetamine Intoxication and Nerve Agent Argatroban Diclofenac misoprostol Docetaxel Pemetrexed Fulvestrant Biobetters Bendeka US & WW Ryanodex for MH Ryanodex for EHS Ryanodex for MDMA & Methamphetamine Intoxication Argatroban Diclofenac misoprostol Docetaxel Fulvestrant Bendeka US Ryanodex MH Argatroban Dicloflenac misoprostol Docetaxel

Thank You September 2017 22

