| ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
| TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
Not Applicable | ||
| (State or other jurisdiction of incorporation or organization) |
(loyer Identification No.) |
| Title of each class |
Trading Symbol(s) |
Name of each exchange on which registered | ||
| ☒ | Accelerated filer | ☐ | ||||
| Non-accelerated filer | ☐ | Smaller reporting company | ||||
| Emerging growth company | ||||||
Page |
||||||
1 |
||||||
1 |
||||||
PART I |
||||||
| Item 1. |
2 |
|||||
| Item 1A. |
26 |
|||||
| Item 1B. |
50 |
|||||
| Item 2. |
51 |
|||||
| Item 3. |
51 |
|||||
| Item 4. |
51 |
|||||
PART II |
||||||
| Item 5. |
51 |
|||||
| Item 6. |
52 |
|||||
| Item 7. |
53 |
|||||
| Item 7A. |
82 |
|||||
| Item 8. |
85 |
|||||
| Item 9. |
168 |
|||||
| Item 9A. |
168 |
|||||
| Item 9B. |
169 |
|||||
| Item 9C. |
169 |
|||||
PART III |
||||||
| Item 10. |
169 |
|||||
| Item 11. |
169 |
|||||
| Item 12. |
169 |
|||||
| Item 13. |
169 |
|||||
| Item 14. |
169 |
|||||
PART IV |
||||||
| Item 15. |
170 |
|||||
| Item 16. |
175 |
|||||
| • | our ability to successfully compete in the marketplace, including: that we are substantially dependent on our generic products; consolidation of our customer base and commercial alliances among our customers; delays in launches of new generic products; the increase in the number of competitors targeting generic opportunities and seeking U.S. market exclusivity for generic versions of significant products; our ability to develop and commercialize biopharmaceutical products; competition for our specialty products, including AUSTEDO ® , AJOVY® and COPAXONE® ; our ability to achieve expected results from investments in our product pipeline; our ability to develop and commercialize additional pharmaceutical products; and the effectiveness of our patents and other measures to protect our intellectual property rights; |
| • | our substantial indebtedness, which may limit our ability to incur additional indebtedness, engage in additional transactions or make new investments, may result in a further downgrade of our credit ratings; and our inability to raise debt or borrow funds in amounts or on terms that are favorable to us; |
| • | our business and operations in general, including: uncertainty regarding the COVID-19 pandemic and the governmental and societal responses thereto; our ability to successfully execute and maintain the activities and efforts related to the measures we have taken or may take in response to the COVID-19 pandemic and associated costs therewith; effectiveness of our optimization efforts; our ability to attract, hire and retain highly skilled personnel; manufacturing or quality control problems; interruptions in our supply chain; disruptions of information technology systems; breaches of our data security; variations in intellectual property laws; challenges associated with conducting business globally, including political or economic instability, major hostilities or terrorism; costs and delays resulting from the extensive pharmaceutical regulation to which we are subject or delays in governmental processing time due to travel and work restrictions caused by the COVID-19 pandemic; the effects of reforms in healthcare regulation and reductions in pharmaceutical pricing, reimbursement and coverage; |
| significant sales to a limited number of customers; our ability to successfully bid for suitable acquisition targets or licensing opportunities, or to consummate and integrate acquisitions; and our prospects and opportunities for growth if we sell assets; |
| • | compliance, regulatory and litigation matters, including: failure to comply with complex legal and regulatory environments; increased legal and regulatory action in connection with public concern over the abuse of opioid medications and our ability to reach a final resolution of the remaining opioid-related litigation; scrutiny from competition and pricing authorities around the world, including our ability to successfully defend against the U.S. Department of Justice (“DOJ”) criminal charges of Sherman Act violations; potential liability for patent infringement; product liability claims; failure to comply with complex Medicare and Medicaid reporting and payment obligations; compliance with anti-corruption sanctions and trade control laws; environmental risks; and the impact of Environmental, Social and Governance (“ESG”) issues; |
| • | other financial and economic risks, including: our exposure to currency fluctuations and restrictions as well as credit risks; potential impairments of our intangible assets; potential significant increases in tax liabilities (including as a result of potential tax reform in the United States); and the effect on our overall effective tax rate of the termination or expiration of governmental programs or tax benefits, or of a change in our business; |
ITEM 1. |
BUSINESS |
| • | AJOVY (fremanezumab-vfrm) injection is a fully humanized monoclonal antibody that binds to calcitonin gene-related peptide (“CGRP”) and it is indicated for the preventive treatment of migraine in adults. AJOVY was launched in the U.S. in 2018. AJOVY was approved in Canada in April 2020. |
| • | During 2019, AJOVY was granted a marketing authorization in the European Union by the European Medicines Agency (“EMA”) in a centralized process and began receiving marketing authorizations in |
| various countries in our International Markets segment. AJOVY was launched in Japan in August 2021. By the end of 2021, we launched AJOVY in most European countries and in certain International Markets countries. We are moving forward with plans to launch in other countries around the world. |
| • | Our auto-injector device for AJOVY became commercially available in the U.S. in April 2020 and in Canada in April 2021. We have also received approval from the EMA for AJOVY’s auto-injector submission in the European Union in October 2019, and we commenced launch in March 2020. |
| • | AJOVY is the only anti-CGRP subcutaneous product indicated for quarterly treatment. |
| • | AJOVY is protected by patents expiring in 2026 in Europe and in 2027 in the United States. Applications for patent term extensions have been submitted in various markets around the world, and certain extensions in Europe and other countries have already been granted until 2031. Additional patents relating to the use of AJOVY in the treatment of migraine have also been issued in the United States and will expire between 2035 and 2039. Such patents are also pending in other countries. AJOVY will also be protected by regulatory exclusivity for 12 years from marketing approval in the United States and 10 years from marketing approval in Europe. |
| • | We have filed a lawsuit in the U.S. District Court for the District of Massachusetts alleging that Eli Lilly & Co.’s (“Lilly”) marketing and sale of its galcanezumab product for the treatment of migraine infringes nine Teva patents. Lilly then submitted inter partes review (“IPR”) petitions to the Patent Trial and Appeal Board (“PTAB”), challenging the validity of the nine patents asserted against it in the litigation. The litigation in the district court was stayed pending resolution of the IPR petitions. On February 18, 2020, the PTAB issued decisions on the first six IPRs, finding the six composition of matter patents invalid as being obvious. On March 31, 2020, the PTAB issued a decision upholding the three method of treatment patents. On August 16, 2021, the Court of Appeals for the Federal Circuit affirmed all of the PTAB’s decisions. The litigation is proceeding as to the three method of treatment patents and a trial is expected in 2022. We also filed another suit against Lilly on June 8, 2021, asserting two patents recently granted to Teva, related to the treatment of refractory migraine. Lilly responded to the complaint with a motion to dismiss, which Teva is opposing. In addition, in 2018 we entered into separate agreements with Alder Biopharmaceuticals, Inc. and Lilly resolving the European Patent Office oppositions that they filed against our AJOVY patents. The settlement agreement with Lilly also resolved Lilly’s action to revoke the patent protecting AJOVY in the United Kingdom. |
| • | AUSTEDO (deutetrabenazine) is a deuterated form of a small molecule inhibitor of vesicular monoamine 2 transporter, or VMAT2, that is designed to regulate the levels of a specific neurotransmitter, dopamine, in the brain. The FDA granted deutetrabenazine New Chemical Entity exclusivity until April 2022 and Orphan Drug exclusivity for the treatment of chorea associated with Huntington’s disease until April 2024. |
| • | AUSTEDO was launched in the U.S. in 2017. It is indicated for the treatment of chorea associated with Huntington’s disease and for the treatment of tardive dyskinesia in adults, which is a debilitating, often irreversible movement disorder caused by certain medications used to treat mental health or gastrointestinal conditions. |
| • | AUSTEDO was launched in China for the treatment of chorea associated with Huntington’s disease and for the treatment of tardive dyskinesia in early 2021. In October 2021, we received marketing approval for both indications in Brazil. We continue with additional submissions in various other countries around the world. |
| • | AUSTEDO is protected in the United States by seven Orange Book patents expiring between 2031 and 2038 and in Europe by two patents expiring in 2029. We received notice letters from two ANDA filers regarding the filing of their ANDAs with paragraph (IV) certifications for certain of the patents listed |
| in the Orange Book for AUSTEDO. On July 1, 2021, we filed a complaint against Aurobindo, asserting six of the Orange Book patents, and a separate complaint against Lupin, asserting four of the Orange Book patents. The suits were filed in the U.S. District Court for the District of New Jersey. The seventh patent was issued in November 2021, and listed in the Orange Book in December 2021. In addition, Apotex has filed a petition for IPR by the PTAB of the patent covering the deutetrabenazine compound that expires in 2031. We responded to that petition on December 15, 2021. |
| • | COPAXONE (glatiramer acetate injection) continues to play a major role in the treatment of MS in the United States (according to IQVIA data as of late 2021). COPAXONE is indicated for the treatment of patients with relapsing forms of MS (“RMS”), including the reduction of the frequency of relapses in relapsing-remitting multiple sclerosis (“RRMS”), including in patients who have experienced a first clinical episode and have MRI features consistent with MS. |
| • | COPAXONE is believed to have a unique mechanism of action that works with the immune system, unlike many therapies that are believed to rely on general immune suppression or cell sequestration to exert their effect. COPAXONE provides a proven mix of efficacy, safety and tolerability. |
| • | One European patent protecting COPAXONE 40 mg/mL was found invalid by the Board of Appeal of the European Patent Office in September 2020. Two additional patents expiring in 2030 were found invalid at the European Patent Office in December 2021. In certain countries, Teva remains in litigation against generic companies on an additional COPAXONE 40 mg/mL patent that expires in 2030. |
| • | In December 2018, Teva sued Pharmascience regarding its application to sell a generic version of COPAXONE in Canada. In December 2020, the Canadian Federal Court issued a decision finding the 2028 method of use patent invalid and the 2030 dosing regimen patent valid and infringed. In January 2022, the Canadian Federal Court of Appeals affirmed Teva’s victory against Pharmascience on the 2030 dosing regimen patent. A re-examination proceeding initiated by Pharmascience at the Canadian Patent office that had been stayed, may resume. We previously settled our Canadian litigation with Sandoz, regarding their application for a generic version of COPAXONE in Canada. Additionally, a case against Mylan with respect to its Canadian application for a generic version of COPAXONE was stayed pending the outcome of the Pharmascience appeal, and may resume. Mylan’s 24-month stay for its product will likely be extended at least until 2023. |
| • | The market for MS treatments continues to develop, particularly with generic versions of COPAXONE. Oral treatments for MS, such as Tecfidera ® , Gilenya® and Aubagio® , continue to present significant and increasing competition. COPAXONE also continues to face competition from existing injectable products, as well as from monoclonal antibodies, such as Ocrevus® . |
| • | BENDEKA (bendamustine hydrochloride) injection and TREANDA (bendamustine hydrochloride) for injection are approved in the United States for the treatment of patients with Chronic Lymphocytic Leukemia (“CLL”) and patients with indolent B-cell Non-Hodgkin’s Lymphoma (“NHL”) that has progressed during or within six months of treatment with rituximab or a rituximab-containing regimen. We launched BENDEKA in the United States in January 2016. It is a liquid, low-volume (50 mL) and short-time 10-minute infusion formulation of bendamustine hydrochloride that we licensed from Eagle. |
| • | BENDEKA faces direct competition from Belrapzo ® (a ready-to-dilute bendamustine hydrochloride product from Eagle). Other competitors to BENDEKA include combination therapies such as R-CHOP (a combination of cyclophosphamide, vincristine, doxorubicin and prednisone in combination with rituximab) and CVP-R (a combination of cyclophosphamide, vincristine and prednisolone in combination with rituximab) for the treatment of NHL, as well as a combination of fludarabine, doxorubicin and rituximab for the treatment of CLL and newer targeted oral therapies, such as ibrutinib, idelilisib and venetoclax. |
| • | In July 2018, Eagle prevailed in its suit against the FDA to obtain seven years of orphan drug exclusivity in the United States for BENDEKA. On March 13, 2020, this decision was upheld in the appellate court. As things currently stand, drug applications referencing BENDEKA, TREANDA or any other bendamustine product will not be approved by the FDA until the orphan drug exclusivity expires in December 2022. In April 2019, we signed an amendment to the license agreement with Eagle extending the royalty term applicable to the United States to the full period for which we sell BENDEKA and increased the royalty rate. In consideration, Eagle agreed to assume a portion of BENDEKA-related patent litigation expenses. |
| • | There are 16 patents listed in the U.S. Orange Book for BENDEKA with expiry dates between 2026 and 2031. In September 2019, a patent infringement action against four of six ANDA filers for generic versions of BENDEKA was tried in the U.S. District Court for the District of Delaware. On April 27, 2020, the district court upheld the validity of all of the asserted patents and found that all four ANDA filers infringe at least one of the patents. Three of the four ANDA filers appealed the district court decision. Teva settled with one of the three ANDA filers, and on August 13, 2021, the Federal Circuit issued a Rule 36 affirmance of the district court decision with respect to the other two filers. On December 14, 2021, Apotex filed a Petition for a Writ of Certiorari with the U.S. Supreme Court. Litigation against the fifth ANDA filer was dismissed after the withdrawal of its patent challenge, and the case against a sixth ANDA filer was also settled. Recent suits against two filers of 505(b)(2) NDAs referencing BENDEKA are also in initial stages of litigation. |
| • | Additionally, in July 2018, Teva and Eagle filed suit against Hospira, Inc. (“Hospira”) related to its 505(b)(2) NDA referencing BENDEKA in the U.S. District Court for the District of Delaware. On December 16, 2019, the district court dismissed the case against Hospira on all but one of the asserted patents, which expires in 2031. The trial on the remaining asserted patent has been postponed and is scheduled to begin on April 25, 2022. |
| • | In addition to the settlement with Eagle regarding its bendamustine 505(b)(2) NDA, between 2015 and 2020, we reached final settlements with 22 ANDA filers for generic versions of the lyophilized form of TREANDA and one 505(b)(2) NDA filer for a generic version of the liquid form of TREANDA, providing for the launch of generic versions of TREANDA prior to patent expiration. |
| • | ProAir HFA ® HFA, another albuterol inhaler. Other generic versions of ProAir were launched in 2020. |
| • | ProAir RespiClick |
| • | QVAR |
| • | QVAR RediHaler |
| • | ProAir Digihaler |
| • | ArmonAir Digihaler |
| • | AirDuo Digihaler |
| • | BRALTUS ® inhaler. It was launched in Europe in August 2016. |
| • | CINQAIR/CINQAERO |
| • | AirDuo RespiClick |
| Phase 2 |
Phase 3 |
Pre-Submission |
Under Regulatory Review | |||||
Novel Biologics |
TEV-48574 Irritable Bowel Syndrome |
Fasinumab Osteoarthritic Pain (March 2016) (1) |
||||||
TEV-53275 |
||||||||
Small Molecules |
Deutetrabenazine Dyskinesia in Cerebral Palsy (September 2019) |
Risperidone LAI Schizophrenia (2) | ||||||
Digital Respiratory |
Digihaler ® (budesonide and formoterol fumarate dihydrate) (EU) |
|||||||
QVAR ® Digihaler® (beclomethasone dipropionate HFA)(U.S.) |
| (1) | Developed in collaboration with Regeneron Pharmaceuticals, Inc. (“Regeneron”). Results for two phase 3 clinical trials, FACT OA1 and FACT OA2, were released on August 5, 2020, indicating that the co-primary endpoints for fasinumab 1 mg monthly were achieved. Fasinumab 1 mg monthly demonstrated significant improvements in pain and physical function over placebo at week 16 and week 24, respectively. Fasinumab 1 mg monthly also showed nominally significant benefits in physical function in two trials and pain in one trial, when compared to the maximum FDA-approved prescription doses of non-steroidal anti-inflammatory drugs for osteoarthritis. The FACT OA1 trial included an additional treatment arm, fasinumab 1 mg every two months, which showed numerical benefit over placebo, but did not reach statistical significance. In initial safety analyses from the phase 3 trials, there was an increase in arthropathies reported with fasinumab. In a sub-group of patients from one phase 3 long-term safety trial, there was an increase in joint replacement with fasinumab 1 mg monthly treatment during the off-drug follow-up period, although this increase was not seen in the other trials to date. |
| (2) | Developed under a license agreement with MedinCell. In August 2021, the FDA accepted the NDA for risperidone LAI, based on phase 3 data from two pivotal studies. |
| • | fremanezumab for fibromyalgia and for an additional indication; and |
| • | deutetrabenazine for an additional indication. |
| • | global R&D facilities that enable us to have a broad global generic pipeline and product line, as well as a focused pipeline of specialty products; |
| • | API manufacturing capabilities that offer a stable, high-quality supply of key APIs, vertically integrated with our pharmaceutical operations; |
| • | pharmaceutical manufacturing facilities approved by the FDA, EMA and other regulatory authorities located around the world, which offer a broad range of production technologies and the ability to concentrate production in order to achieve high quality and economies of scale; and |
| • | high-volume, technologically advanced distribution facilities for solid dosage forms, injectable and blow-fill-seal, which are available in North America, Europe, Latin America, India and Israel and that allow us to deliver new products to our customers quickly and efficiently, providing a cost-effective, safe and reliable supply. |
| • | we continued the implementation of our global EHS management system in all countries where we operate, which promotes proactive compliance with applicable EHS requirements, establishes EHS standards throughout our global operations and helps drive continuous improvement in our EHS performance; |
| • | proactively evaluated EHS compliance through self-evaluation and an internal and external audit program, addressing non-conformities through appropriate corrective and preventative action; and |
| • | continued to promote climate change mitigation and adaptation strategy according to international standards. |
December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
| Full-time |
34,713 | 37,100 | 38,130 | |||||||||
| Part-time |
1,266 | 1,272 | 1,158 | |||||||||
| Contractor |
1,558 | 1,844 | 1,497 | |||||||||
| Total |
37,537 | 40,216 | 40,785 | |||||||||
| Total full time equivalent |
37,037 | 39,717 | 40,039 | |||||||||
December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
| North America |
6,302 | 6,918 | 7,336 | |||||||||
| Europe |
18,122 | 18,569 | 18,207 | |||||||||
| International Markets (excluding Israel) |
7,955 | 9,210 | 9,408 | |||||||||
| Israel |
3,600 | 3,675 | 4,337 | |||||||||
| Total (excluding contractors) |
35,979 | 38,372 | 39,288 | |||||||||
Female |
Male |
|||||||
| Total employees |
46 | % | 54 | % | ||||
| Managers |
48 | % | 52 | % | ||||
| Senior management |
29 | % | 71 | % | ||||
ITEM |
1A. RISK FACTORS |
| • | Our future success depends on our ability to maximize the growth and commercial success of AUSTEDO. If our revenues derived from AUSTEDO do not increase as expected or if we lose market share to competing therapies, it may have an adverse effect on our results of operations. |
| • | AJOVY faces strong competition from two products that were introduced into the market around the same time and are competing for market share in the same space, as well as from other emerging competing therapies, including oral calcitonin gene-related peptide (“CGRP”) products. |
| • | COPAXONE faces increasing competition from generic versions in the U.S. and competing glatiramer acetate products in Europe, as well as from orally-administered therapies. Following the approval of generic competition, COPAXONE’s revenues and profitability have decreased. We expect this trend to continue in the future, which may have a significant effect on our financial results and cash flow. |
| • | making it more difficult for us to satisfy our obligations; |
| • | limiting our ability to borrow additional funds and increasing the cost of any such borrowing; |
| • | increasing our vulnerability to, and reducing our flexibility to respond to, general adverse economic and industry conditions; |
| • | limiting our flexibility in planning for, or reacting to, changes in our business and the industry in which we operate; |
| • | placing us at a competitive disadvantage as compared to our competitors, to the extent that they are not as highly leveraged; and |
| • | restricting us from pursuing certain business opportunities. |
| • | some government programs may be discontinued, or the applicable tax rates may increase; |
| • | we may be unable to meet the requirements for continuing to qualify for some programs and the restructuring plan may lead to the loss of certain tax benefits we currently receive; |
| • | these programs and tax benefits may be unavailable at their current levels; |
| • | upon expiration of a particular benefit, we may not be eligible to participate in a new program or qualify for a new tax benefit that would offset the loss of the expiring tax benefit; or |
| • | we may be required to refund previously recognized tax benefits if we are found to be in violation of the stipulated conditions. |
| Business Segment |
Number of Facilities |
Square Feet (in thousands) |
||||||
| North America |
19 | 4,000 | ||||||
| Europe |
30 | 11,200 | ||||||
| International Markets |
30 | 7,000 | ||||||
| |
|
|
|
|||||
| Worldwide Total Manufacturing and R&D Facilities |
79 | 22,200 | ||||||
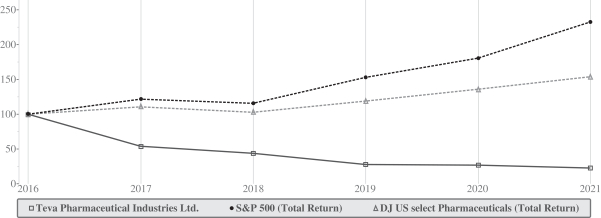
| * | $100 invested on December 31, 2016 in stock or index – including reinvestment of dividends. Indexes calculated on month-end basis. |
| • | Our revenues in 2021 were $15,878 million, a decrease of 5% in U.S. dollars, or 6% in local currency terms, compared to 2020, mainly due to lower revenues from COPAXONE, generic products in the U.S., generic products in Japan resulting from the divestment of a majority of the generic and operational assets of our Japanese business venture, and Anda, partially offset by higher revenues from AUSTEDO and AJOVY. Revenues continued to be affected by the ongoing impact of the COVID-19 pandemic on markets and on customer stocking and purchasing patterns. |
| • | Our North America segment generated revenues of $7,809 million and profit of $2,224 million in 2021. Revenues decreased by 7.5% compared to 2020. Profit decreased by 8% compared to 2020. |
| • | Our Europe segment generated revenues of $4,886 million and profit of $1,494 million in 2021. Revenues increased by 3% in U.S. dollars. In local currency terms, revenues decreased by 2% compared to 2020. Profit increased by 12% compared to 2020. |
| • | Our International Markets segment generated revenues of $2,032 million and profit of $529 million in 2021. Revenues decreased by 6% in U.S. dollars, or 4% in local currency terms compared to 2020. Profit increased by 12% compared to 2020. |
| • | Our revenues from other activities in 2021 were $1,151 million, a decrease of 12% compared to 2020. In local currency terms, revenues decreased by 13%. |
| • | Impairments of identifiable intangible assets were $424 million and $1,502 million in the years ended December 31, 2021 and 2020, respectively. See note 6 to our consolidated financial statements. |
| • | We recorded expenses of $341 million for other asset impairments, restructuring and other items in 2021, compared to expenses of $479 million in 2020. See note 15 to our consolidated financial statements. |
| • | In 2021, we recorded an expense of $717 million in legal settlements and loss contingencies, compared to an expense of $60 million in 2020. See note 11 to our consolidated financial statements. |
| • | Operating income was $1,716 million in 2021, compared to an operating loss of $3,572 million in 2020. |
| • | Financial expenses were $1,058 million in 2021, compared to $834 million in 2020. See note 17 to our consolidated financial statements. |
| • | In 2021, we recognized a tax expense of $211 million, or 32%, on a pre-tax income of $658 million. In 2020, we recognized a tax benefit of $168 million, or 4%, on a pre-tax loss of $4,406 million. See note 13 to our consolidated financial statements. |
| • | As of December 31, 2021, our debt was $23,043 million, compared to $25,919 million as of December 31, 2020. This decrease was mainly due to $4,008 million repurchased upon consummation of a cash tender offer, $3,167 million senior notes repaid at maturity and $710 million exchange rate fluctuations, partially offset by $4,973 million of issued sustainability-linked senior notes net of issuance costs. |
| • | Cash flow generated from operating activities was $798 million in 2021, compared to $1,216 million in 2020. This decrease was mainly due to lower profit in our North America segment during 2021. |
| • | During 2021, we generated free cash flow of $2,196 million, which we define as comprising: $798 million in cash flow generated from operating activities, $1,648 million in beneficial interest collected in exchange for securitized accounts receivables and $311 million in proceeds from divestitures of businesses and other assets, partially offset by $562 million in cash used for capital investments. During 2020, we generated free cash flow of $2,110 million. |
Year ended December 31, |
||||||||||||||||
2021 |
2020 |
|||||||||||||||
(U.S. $ in millions /% of Segment Revenues) |
||||||||||||||||
| Revenues |
$ | 7,809 | 100 | % | $ | 8,447 | 100 | % | ||||||||
| Gross profit |
4,226 | 54.1 | % | 4,489 | 53.1 | % | ||||||||||
| R&D expenses |
618 | 7.9 | % | 622 | 7.4 | % | ||||||||||
| S&M expenses |
988 | 12.7 | % | 1,013 | 12.0 | % | ||||||||||
| G&A expenses |
427 | 5.5 | % | 443 | 5.2 | % | ||||||||||
| Other income |
(31 | ) | § | (10 | ) | § | ||||||||||
| |
|
|
|
|
|
|
|
|||||||||
| Segment profit* |
$ | 2,224 | 28.5 | % | $ | 2,421 | 28.7 | % | ||||||||
| |
|
|
|
|
|
|
|
|||||||||
| * | Segment profit does not include amortization and certain other items. |
| § | Represents an amount less than 0.5%. |
Year ended December 31, |
Percentage Change 2020-2021 |
|||||||||||
2021 |
2020 |
|||||||||||
(U.S. $ in millions) |
||||||||||||
| Generic products |
$ | 3,769 | $ | 4,010 | (6 | %) | ||||||
| AJOVY |
176 | 134 | 31 | % | ||||||||
| AUSTEDO |
802 | 637 | 26 | % | ||||||||
| BENDEKA/TREANDA |
385 | 415 | (7 | %) | ||||||||
| COPAXONE |
577 | 884 | (35 | %) | ||||||||
| ProAir* |
180 | 241 | (25 | %) | ||||||||
| Anda |
1,323 | 1,462 | (9 | %) | ||||||||
| Other |
597 | 664 | (10 | %) | ||||||||
| |
|
|
|
|||||||||
| Total |
$ | 7,809 | $ | 8,447 | (8 | %) | ||||||
| |
|
|
|
|||||||||
| * | Does not include revenues from the ProAir authorized generic, which are included under generic products. |
| Product Name |
Brand Name |
Launch Date |
Total Annual U.S. Branded Sales at Time of Launch (U.S. $ in millions (IQVIA))* |
|||||
| Mesalamine Extended-Release Capsules, 0.375g |
Apriso ® |
January | $ | 344 | ||||
| Etonogestrel and Ethinyl Estradiol Vaginal Ring |
NuvaRing ® |
January | $ | 812 | ||||
| Testosterone Gel, 1.62%, 20.25mg/1.25g & 40.5mg/2.5g |
AndroGel ® |
February | $ | 40 | ||||
| Liothyronine Sodium Tablets USP, 5mcg, 25mcg, 50mcg |
Cytomel ® |
February | $ | 107 | ||||
| Brinzolamide Ophthalmic Suspension, USP, 1% |
Azopt ® |
March | $ | 184 | ||||
| Mesalamine Suppositories |
Canasa ® |
April | $ | 66 | ||||
| Isotretinoin Capsules, USP |
Absorica ® |
April | $ | 156 | ||||
| Erythromycin Tablets, USP |
N/A | May | $ | 45 | ||||
| Tiopronin Tablets |
Thiola ® |
May | $ | 0.2 | ||||
| Ivermectin Cream, 1% |
Soolantra ® |
June | $ | 111 | ||||
| Formoterol Fumarate Inhalation Solution |
Perforomist ® |
June | $ | 300 | ||||
| Bexarotene Capsules 75 mg |
Targretin ® |
November | $ | 48 | ||||
| Adapalene and Benzoyl Peroxide Gel |
Epiduo ® |
December | $ | 281 | ||||
| Arformoterol Tartrate Inhalation, Eq. 0.015 mg base/2 mL |
Brovana ® |
December | $ | 333 | ||||
| Erlotinib Hydrochloride Tablets, 2 mg |
Tarceva ® |
December | $ | 3 | ||||
| Pyrimethamine Tablets USP, 25 mg |
Daraprim ® |
December | $ | 42 | ||||
| Ibuprofen & Famotidine Tablets, 800 mg/26.6 mg |
Duexis ® |
December | $ | 690 | ||||
| Naloxone HCl Nasal Spray, 4 mg/spray |
Narcan ® |
December | $ | 281 | ||||
| Sunitinib Malate Capsules, 12.5 mg, 25 mg, 37.5 mg and 50 mg |
Sutent ® |
December | $ | 193 | ||||
| Buprenorphine Transdermal System, 7.5 mcg |
Butrans ® |
December | $ | 13 | ||||
| * | The figures presented are for the twelve months ended in the calendar quarter immediately prior to our launch or re-launch. |
| Generic Name |
Brand Name |
Total U.S. Annual Branded Market (U.S. $ in millions (IQVIA))* |
||||
| Ibrutinib Caps |
Imbruvica ® |
$ | 781 | |||
| Lubiprostone Caps |
Amitiza ® |
$ | 306 | |||
| Lenalidomide Capsules, 2.5 mg and 20 mg |
Revlimid ® |
$ | 162 | |||
| Pimavanserin Capsules, 34 mg |
Nuplazid ® |
$ | 165 | |||
| Methylnaltrexone Bromide Subcutaneous Injection |
Relistor ® |
$ | 19 | |||
| Tasimelteon Caps |
Hetlioz ® |
$ | 2 | |||
| * | The figures presented are for the twelve months ended in the calendar quarter immediately prior to our launch or re-launch. |
Year ended December 31, |
||||||||||||||||
2021 |
2020 |
|||||||||||||||
(U.S. $ in millions /% of Segment Revenues) |
||||||||||||||||
| Revenues |
$ | 4,886 | 100 | % | $ | 4,757 | 100 | % | ||||||||
| Gross profit |
2,823 | 57.8 | % | 2,666 | 56.0 | % | ||||||||||
| R&D expenses |
244 | 5.0 | % | 247 | 5.2 | % | ||||||||||
| S&M expenses |
846 | 17.3 | % | 830 | 17.4 | % | ||||||||||
| G&A expenses |
244 | 5.0 | % | 261 | 5.5 | % | ||||||||||
| Other (income) expense |
(5 | ) | § | (3 | ) | § | ||||||||||
| |
|
|
|
|
|
|
|
|||||||||
| Segment profit* |
$ | 1,494 | 30.6 | % | $ | 1,331 | 28.0 | % | ||||||||
| |
|
|
|
|
|
|
|
|||||||||
| * | Segment profit does not include amortization and certain other items. |
| § | Represents an amount less than 0.5%. |
Year ended December 31, |
Percentage Change 2020-2021 |
|||||||||||
2021 |
2020 |
|||||||||||
(U.S. $ in millions) |
|
|||||||||||
| Generic products |
$ | 3,569 | $ | 3,513 | 2 | % | ||||||
| AJOVY |
87 | 31 | 184 | % | ||||||||
| COPAXONE |
391 | 400 | (2 | %) | ||||||||
| Respiratory products |
356 | 353 | 1 | % | ||||||||
| Other |
483 | 459 | 5 | % | ||||||||
| |
|
|
|
|||||||||
| Total |
$ | 4,886 | $ | 4,757 | 3 | % | ||||||
| |
|
|
|
|||||||||
| § | Represents an amount less than 0.5%. |
2021 |
2020 |
|||||||||||||||
(U.S. $ in millions / % of Segment Revenues) |
||||||||||||||||
| Revenues |
$ | 2,032 | 100 | % | $ | 2,154 | 100 | % | ||||||||
| Gross profit |
1,118 | 55.0 | % | 1,096 | 50.9 | % | ||||||||||
| R&D expenses |
68 | 3.3 | % | 70 | 3.3 | % | ||||||||||
| S&M expenses |
417 | 20.5 | % | 427 | 19.8 | % | ||||||||||
| G&A expenses |
109 | 5.4 | % | 136 | 6.3 | % | ||||||||||
| Other (income) expense |
(5 | ) | § | (11 | ) | (0.5 | %) | |||||||||
| |
|
|
|
|
|
|
|
|||||||||
| Segment profit* |
$ | 529 | 26.0 | % | $ | 474 | 22.0 | % | ||||||||
| |
|
|
|
|
|
|
|
|||||||||
| * | Segment profit does not include amortization and certain other items. |
| § | Represents an amount less than 0.5%. |
Year ended December 31, |
Percentage Change 2020-2021 |
|||||||||||
2021 |
2020 |
|||||||||||
(U.S. $ in millions) |
||||||||||||
| Generic products |
$ | 1,649 | $ | 1,792 | (8 | %) | ||||||
| AJOVY |
50 | 18 | 179 | % | ||||||||
| COPAXONE |
37 | 53 | (29 | %) | ||||||||
| Other |
295 | 291 | 1 | % | ||||||||
| |
|
|
|
|||||||||
| Total |
$ | 2,032 | $ | 2,154 | (6 | %) | ||||||
| |
|
|
|
|||||||||
Year ended December 31, |
||||||||
2021 |
2020 |
|||||||
(U.S. $ in millions) |
||||||||
| North America profit |
$ | 2,224 | $ | 2,421 | ||||
| Europe profit |
1,494 | 1,331 | ||||||
| International Markets profit |
529 | 474 | ||||||
| |
|
|
|
|||||
| Total reportable segments profit |
4,246 | 4,225 | ||||||
| Profit of other activities |
154 | 163 | ||||||
| |
|
|
|
|||||
| Total segments profit |
4,401 | 4,388 | ||||||
| Amounts not allocated to segments: |
||||||||
| Amortization |
802 | 1,020 | ||||||
| Other assets impairments, restructuring and other items |
341 | 479 | ||||||
| Goodwill impairment |
— | 4,628 | ||||||
| Intangible asset impairments |
424 | 1,502 | ||||||
| Legal settlements and loss contingencies |
717 | 60 | ||||||
| Other unallocated amounts |
402 | 271 | ||||||
| Consolidated operating income (loss) |
1,716 | (3,572 | ) | |||||
| |
|
|
|
|||||
| Financial expenses, net |
1,058 | 834 | ||||||
| |
|
|
|
|||||
| Consolidated income (loss) before income taxes |
$ | 658 | $ | (4,406 | ) | |||
| |
|
|
|
|||||
Payments Due by Period |
||||||||||||||||||||
Total |
Less than 1 year |
1-3 years |
3-5 years |
More than 5 years |
||||||||||||||||
(U.S. $ in millions) |
||||||||||||||||||||
| Long-term debt obligations, including estimated interest* |
$ | 29,778 | $ | 2,340 | $ | 5,805 | $ | 8,301 | $ | 13,332 | ||||||||||
| Purchase obligations (including purchase orders) |
1,382 | 1,135 | 163 | 69 | 15 | |||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| Total |
$ | 31,160 | $ | 3,475 | $ | 5,968 | $ | 8,370 | $ | 13,347 | ||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| * | Long-term debt obligations mainly include senior notes, sustainability-linked senior notes and convertible senior debentures, as disclosed in note 9 to our consolidated financial statements. |
| • | our management and Board of Directors use non-GAAP measures to evaluate our operational performance, to compare against work plans and budgets, and ultimately to evaluate the performance of management; |
| • | our annual budgets are prepared on a non-GAAP basis; and |
| • | senior management’s annual compensation is derived, in part, using these non-GAAP measures. While qualitative factors and judgment also affect annual bonuses, the principal quantitative element in the determination of such bonuses is performance targets tied to the work plan, which is based on the non-GAAP presentation set forth below. |
| • | amortization of purchased intangible assets; |
| • | legal settlements and material litigation fees, and/or loss contingencies, due to the difficulty in predicting their timing and scope; |
| • | impairments of long-lived assets, including intangibles, property, plant and equipment and goodwill; |
| • | restructuring expenses, including severance, retention costs, contract cancellation costs and certain accelerated depreciation expenses primarily related to the rationalization of our plants or to certain other strategic activities, such as the realignment of R&D focus or other similar activities; |
| • | acquisition- or divestment- related items, including changes in contingent consideration, integration costs, banker and other professional fees, inventory step-up and in-process R&D acquired in development arrangements; |
| • | expenses related to our equity compensation; |
| • | significant one-time financing costs, amortization of issuance costs and terminated derivative instruments, and valuation gains or losses; |
| • | unusual tax items; |
| • | other awards or settlement amounts, either paid or received; |
| • | other exceptional items that we believe are sufficiently large that their exclusion is important to facilitate an understanding of trends in our financial results, such as impacts due to changes in accounting, significant costs for remediation of plants, such as inventory write-offs or related consulting costs, or other unusual events; and |
| • | corresponding tax effects of the foregoing items. |
Year Ended December 31, 2021 (U.S. $ and shares in millions, except per share amounts) |
||||||||||||||||||||||||||||||||||||||||||||||||||||
| GAAP | Excluded for non-GAAP measurement | Non- GAAP |
||||||||||||||||||||||||||||||||||||||||||||||||||
| Amortization of purchased intangible assets |
Legal settlements and loss contingencies |
Impairment of long- lived assets |
Other R&D expenses |
Restructuring costs |
Costs related to regulatory actions taken in facilities |
Equity compensation |
Contingent consideration |
Gain on sale of business |
Other non- GAAP items |
Other items |
||||||||||||||||||||||||||||||||||||||||||
| Net revenue |
15,878 | 15,878 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Cost of sales |
8,284 | 702 | 23 | 23 | 270 | 7,266 | ||||||||||||||||||||||||||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||
| Gross profit |
7,594 | 702 | — | — | — | — | 23 | 23 | — | — | 270 | — | 8,612 | |||||||||||||||||||||||||||||||||||||||
| Gross profit margin |
48 | % | 54 | % | ||||||||||||||||||||||||||||||||||||||||||||||||
| R&D expenses |
967 | 15 | 19 | — | 933 | |||||||||||||||||||||||||||||||||||||||||||||||
| S&M expenses |
2,429 | 99 | 33 | — | 2,297 | |||||||||||||||||||||||||||||||||||||||||||||||
| G&A expenses |
1,099 | 43 | 27 | 1,029 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Other (income) expense |
(98 | ) | (51 | ) | (48 | ) | ||||||||||||||||||||||||||||||||||||||||||||||
| Legal settlements and loss contingencies |
717 | 717 | — | |||||||||||||||||||||||||||||||||||||||||||||||||
| Other asset impairments, restructuring and other items |
341 | 160 | 133 | 7 | 40 | — | ||||||||||||||||||||||||||||||||||||||||||||||
| Intangible assets impairment |
424 | 424 | — | |||||||||||||||||||||||||||||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||
| Operating income (loss) |
1,716 | 802 | 717 | 584 | 15 | 133 | 23 | 118 | 7 | (51 | ) | 337 | — | 4,401 | ||||||||||||||||||||||||||||||||||||||
| Financial expenses |
1,058 | 128 | 930 | |||||||||||||||||||||||||||||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||
| Income (loss) before income taxes |
658 | 802 | 717 | 584 | 15 | 133 | 23 | 118 | 7 | (51 | ) | 337 | 128 | 3,471 | ||||||||||||||||||||||||||||||||||||||
| Income taxes |
211 | (360 | ) | 570 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Share in profits (losses) of associated companies – net |
(9 | ) | (1 | ) | (8 | ) | ||||||||||||||||||||||||||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||
| Net income (loss) |
456 | 802 | 717 | 584 | 15 | 133 | 23 | 118 | 7 | (51 | ) | 337 | (232 | ) | 2,909 | |||||||||||||||||||||||||||||||||||||
| Net income (loss) attributable to non-controlling interests |
39 | (15 | ) | 54 | ||||||||||||||||||||||||||||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||
| Net income (loss) attributable to Teva |
417 | 802 | 717 | 584 | 15 | 133 | 23 | 118 | 7 | (51 | ) | 337 | (247 | ) | 2,855 | |||||||||||||||||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||
| EPS—Basic |
0.38 | 2.21 | 2.59 | |||||||||||||||||||||||||||||||||||||||||||||||||
| EPS—Diluted |
0.38 | 2.20 | 2.58 | |||||||||||||||||||||||||||||||||||||||||||||||||
| * | Other non-GAAP items include other exceptional items that we believe are sufficiently large that their exclusion is important to facilitate an understanding of trends in our financial results, such as certain accelerated depreciation expenses and inventory write offs, primarily related to the rationalization of our plants and other unusual events. |
Year ended December 31, 2020 (U.S. $ and shares in millions, except per share amounts) |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GAAP | Excluded for non-GAAP measurement | Non-GAAP | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Amortization of purchased intangible assets |
Legal settlements and loss contingencies |
Goodwill impairment |
Impairment of long- lived assets |
Other R&D expenses |
Restructuring costs |
Costs related to regulatory actions taken in facilities |
Equity compensation |
Contingent consideration |
Gain on sale of business |
Other non- GAAP items |
Other items |
|||||||||||||||||||||||||||||||||||||||||||||
| Net revenue |
16,659 | 16,659 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cost of sales |
8,933 | 894 | 23 | 27 | 63 | 7,925 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||
| Gross profit |
7,726 | 894 | 23 | 27 | 63 | 8,734 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Gross profit margin |
46.4 | % | 52.4 | % | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| R&D expenses |
997 | 37 | 20 | — | 941 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| S&M expenses |
2,498 | 126 | 36 | 14 | 2,322 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| G&A expenses |
1,173 | 46 | 12 | 1,115 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other (income) expense |
(40 | ) | (8 | ) | (31 | ) | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Legal settlements and loss contingencies |
60 | 60 | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other asset impairments, restructuring and other items |
479 | 416 | 120 | (81 | ) | 24 | — | |||||||||||||||||||||||||||||||||||||||||||||||||
| Intangible assets impairment |
1,502 | 1,502 | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Goodwill impairment |
4,628 | 4,628 | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||
| Operating income (loss) |
(3,572 | ) | 1,020 | 60 | 4,628 | 1,918 | 37 | 120 | 23 | 129 | (81 | ) | (8 | ) | 114 | — | 4,388 | |||||||||||||||||||||||||||||||||||||||
| Financial expenses, net |
834 | (85 | ) | 918 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||
| Income (loss) before income taxes |
(4,406 | ) | 1,020 | 60 | 4,628 | 1,918 | 37 | 120 | 23 | 129 | (81 | ) | (8 | ) | 114 | (85 | ) | 3,470 | ||||||||||||||||||||||||||||||||||||||
| Income taxes |
(168 | ) | (745 | ) | 577 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Share in profits (losses) of associated companies – net |
(138 | ) | (134 | ) | (4 | ) | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||
| Net income (loss) |
(4,099 | ) | 1,020 | 60 | 4,628 | 1,918 | 37 | 120 | 23 | 129 | (81 | ) | (8 | ) | 114 | (964 | ) | 2,897 | ||||||||||||||||||||||||||||||||||||||
| Net income (loss) attributable to non-controlling interests |
(109 | ) | (177 | ) | 68 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||
| Net income (loss) attributable to Teva |
(3,990 | ) | 1,020 | 60 | 4,628 | 1,918 | 37 | 120 | 23 | 129 | (81 | ) | (8 | ) | 114 | (1,140 | ) | 2,830 | ||||||||||||||||||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||
| EPS—Basic |
(3.64 | ) | 6.23 | 2.58 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| EPS—Diluted |
(3.64 | ) | 6.22 | 2.57 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| * | Other non-GAAP items include other exceptional items that we believe are sufficiently large that their exclusion is important to facilitate an understanding of trends in our financial results, such as certain accelerated depreciation expenses and inventory write offs, primarily related to the rationalization of our plants and other unusual events. |
| • | ongoing impact of the COVID-19 pandemic on markets and on customer stocking and purchasing patterns. For further details, see “—The COVID-19 Pandemic” above; |
| • | continued success of our specialty products AUSTEDO and AJOVY; |
| • | success of clinical trials and approval of our specialty product risperidone LAI; |
| • | ability to successfully execute key generic launches in a timely manner; |
| • | ability to successfully develop and launch new biosimiliar products; |
| • | a decrease in sales of COPAXONE and other specialty products due to potential loss of exclusivity, generic competition and/or availability of alternative therapies; |
| • | we expect continued competition for our generic products where multiple similar generic products have been launched, resulting in pricing pressure in the generics markets. We do, however, also see certain generic segments in which opportunities exist to grow our business, our portfolio of new drug applications and our portfolio of approved complex products; |
| • | we expect continued increases in prices of raw materials, energy, labor and transportation; |
| • | our disciplined cash management and debt repayment schedule; |
| • | our high debt levels and non-investment grade credit rating may increase the cost of any new borrowing; |
| • | continued impact of currency fluctuations on revenues and operating income, as well as on various balance sheet and statements of income line items; |
| • | ongoing evaluation to further network consolidation activities to achieve additional operational efficiencies, which may affect our business and operations; and |
| • | continued efforts towards achieving our long-term financial goals. |
| • | Revenue Recognition and SR&A in the United States |
| • | Income Taxes |
| • | Contingencies |
| • | Goodwill |
| • | Identifiable Intangible Assets |
| • | A projection or forecast that indicates losses or reduced profits associated with an asset. This could result, for example, from a change in the competitive landscape modifying our assumptions about market share or pricing prospectively, a government reimbursement program that results in an inability to sustain projected product revenues and profitability, or lack of acceptance of a product by patients, physicians or payers limiting our projected growth. |
| • | A significant adverse change in legal factors or in the business climate that could affect the value of the asset. For example, a successful challenge of our patent rights by a competitor would likely result in generic competition earlier than expected. And conversely, a lost challenge of patent rights in connection with our generic file would likely result in delayed entry. |
| • | A significant adverse change in the extent or manner in which an asset is used. For example, restrictions imposed by the FDA or other regulatory authorities could affect our ability to manufacture or sell a product. |
| • | For IPR&D projects, this could result from, among other things, a change in outlook affecting assumptions around competition or timing of entry such as approval success or the related timing of approval, clinical trial data results, other delays in the projected launch dates or additional expenditures required to commercialize the product. |
ITEM |
7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK |
| Net exposure as of December 31, 2021 |
||||
Liability/Asset |
(U.S. $ in millions) |
|||
| USD/CHF |
475 | |||
| BGN/EUR |
291 | |||
| USD/JPY |
285 | |||
| GBP/USD |
180 | |||
| INR/USD |
130 | |||
| USD/MXN |
99 | |||
| PLN/EUR |
75 | |||
| USD/EUR |
71 | |||
| HRK/USD |
50 | |||
| Currency (sold) |
Cross Currency (bought) |
Net Notional Value |
Fair Value |
2021 Weighted Average Cross Currency Prices or Strike Prices |
||||||||||||||||||||
2021 |
2020 |
2021 |
2020 |
|||||||||||||||||||||
(U.S. $ in millions) |
||||||||||||||||||||||||
| Forward: |
||||||||||||||||||||||||
| CHF |
USD | 509 | 464 | (4 | ) | (12 | ) | 0.92 | ||||||||||||||||
| JPY |
USD | 313 | 326 | 4 | (5 | ) | 113.51 | |||||||||||||||||
| USD |
GBP | 133 | * | (1 | ) | — | 1.36 | |||||||||||||||||
| EUR |
USD | 98 | 400 | 4 | (16 | ) | 1.18 | |||||||||||||||||
| MXN |
USD | 96 | 91 | (4 | ) | (2 | ) | 21.38 | ||||||||||||||||
| USD |
INR | 95 | 145 | 1 | 2 | 75.35 | ||||||||||||||||||
| RUB |
USD | 79 | * | (1 | ) | — | 76.92 | |||||||||||||||||
| CAD |
USD | 76 | 70 | 1 | (2 | ) | 1.25 | |||||||||||||||||
| EUR |
PLN | 68 | 103 | 1 | — | 4.66 | ||||||||||||||||||
| CZK |
EUR | 50 | * | (1 | ) | — | 25.63 | |||||||||||||||||
| GBP |
USD | * | 133 | — | (3 | ) | — | |||||||||||||||||
| EUR |
CAD | * | 101 | — | (1 | ) | — | |||||||||||||||||
| Options: |
||||||||||||||||||||||||
| EUR |
USD | 73 | 167 | 1 | (3 | ) | 1.15 | |||||||||||||||||
| CAD |
USD | 53 | * | — | (1 | ) | 1.30 | |||||||||||||||||
| CHF |
USD | 51 | 84 | — | (2 | ) | 0.94 | |||||||||||||||||
| JPY |
USD | * | 89 | — | — | — | ||||||||||||||||||
| GBP |
USD | * | 53 | — | (1 | ) | — | |||||||||||||||||
| * | Represents net notional value of less than $50 million. |
| Currency |
Total Amount |
Interest Rate Ranges |
2022 |
2023 |
2024 |
2025 |
2026 |
2027 & thereafter |
||||||||||||||||||||||||||||
(U.S. dollars in millions) |
||||||||||||||||||||||||||||||||||||
| Fixed Rate: |
||||||||||||||||||||||||||||||||||||
| USD |
13,934 | 2.80 | % | 7.13 | % | 1,453 | 1,250 | 1,000 | 3,496 | 1,000 | 5,019 | |||||||||||||||||||||||||
| Euro |
8,417 | 1.13 | % | 6.00 | % | 670 | 708 | 2,152 | — | 2,037 | 2,543 | |||||||||||||||||||||||||
| CHF |
767 | 0.50 | % | 1.00 | % | — | — | 383 | — | — | — | |||||||||||||||||||||||||
| USD convertible debentures* |
23 | 0.25 | % | 0.25 | % | — | — | — | — | — | — | |||||||||||||||||||||||||
| Variable Rate: |
||||||||||||||||||||||||||||||||||||
| Others |
2 | 1.00 | % | 2.00 | % | — | — | — | — | — | — | |||||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||
| Total: |
23,143 | $ | 2,122 | $ | 1,958 | $ | 3,535 | $ | 3,496 | $ | 3,037 | $ | 7,562 | |||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||
| Less debt issuance costs |
(100 | ) | ||||||||||||||||||||||||||||||||||
| |
|
|||||||||||||||||||||||||||||||||||
| Total: |
$ | 23,043 | ||||||||||||||||||||||||||||||||||
| |
|
|||||||||||||||||||||||||||||||||||
| * | Classified under short-term debt. |
December 31, |
December 31, |
|||||||
2021 |
2020 |
|||||||
ASSETS |
||||||||
Current assets: |
||||||||
Cash and cash equivalents |
$ | $ | ||||||
Accounts receivables, net of allowance for credit losses of $ |
||||||||
Inventories |
||||||||
Prepaid expenses |
||||||||
Other current assets |
||||||||
Assets held for sale |
||||||||
Total current assets |
||||||||
Deferred income taxes |
||||||||
Other non-current assets |
||||||||
Property, plant and equipment, net |
||||||||
Operating lease right-of-use assets |
||||||||
Identifiable intangible assets, net |
||||||||
Goodwill |
||||||||
Total assets |
$ | $ | ||||||
LIABILITIES AND EQUITY |
||||||||
Current liabilities: |
||||||||
Short-term debt |
$ | $ | ||||||
Sales reserves and allowances |
||||||||
Accounts payables |
||||||||
Employee-related obligations |
||||||||
Accrued expenses |
||||||||
Other current liabilities |
||||||||
Total current liabilities |
||||||||
Long-term liabilities: |
||||||||
Deferred income taxes |
||||||||
Other taxes and long-term liabilities |
||||||||
Senior notes and loans |
||||||||
Operating lease liabilities |
||||||||
Total long-term liabilities |
||||||||
Commitments and contingencies |
||||||||
Total liabilities |
||||||||
Equity: |
||||||||
Teva shareholders’ equity: |
||||||||
Ordinary shares of NIS |
||||||||
Additional paid-in capital |
||||||||
Accumulated deficit |
( |
) | ( |
) | ||||
Accumulated other comprehensive loss |
( |
) | ( |
) | ||||
Treasury shares as of December 31, 2021 and December 31, 2020: |
( |
) | ( |
) | ||||
Non-controlling interests |
||||||||
Total equity |
||||||||
Total liabilities and equity |
$ | $ | ||||||
Year ended December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
| Net revenues |
$ | $ | $ | |||||||||
| Cost of sales |
||||||||||||
| |
|
|
|
|
|
|||||||
| Gross profit |
||||||||||||
| Research and development expenses , net |
||||||||||||
| Selling and marketing expenses |
||||||||||||
| General and administrative expenses |
||||||||||||
| Intangible assets impairments |
||||||||||||
| Goodwill impairment |
||||||||||||
| Other asset impairments, restructuring and other items |
||||||||||||
| Legal settlements and loss contingencies |
||||||||||||
| Other income |
( |
) | ( |
) | ( |
) | ||||||
| |
|
|
|
|
|
|||||||
| Operating (loss) income |
( |
) | ( |
) | ||||||||
| Financial expenses—net |
||||||||||||
| |
|
|
|
|
|
|||||||
| Income (loss) before income taxes |
( |
) | ( |
) | ||||||||
| Income taxes (benefit) |
( |
) | ( |
) | ||||||||
| Share in (profits) losses of associated companies—net |
( |
) | ( |
) | ||||||||
| |
|
|
|
|
|
|||||||
| Net income (loss) |
( |
) | ( |
) | ||||||||
| Net income (loss) attributable to non-controlling interests |
( |
) | ( |
) | ||||||||
| |
|
|
|
|
|
|||||||
| Net income (loss) attributable to Teva |
( |
) | ( |
) | ||||||||
| |
|
|
|
|
|
|||||||
| Earnings (loss) per share attributable to ordinary shareholders: |
||||||||||||
| Basic |
$ | $ | ( |
) | $ | ( |
) | |||||
| |
|
|
|
|
|
|||||||
| Diluted |
$ | $ | ( |
) | $ | ( |
) | |||||
| |
|
|
|
|
|
|||||||
| Weighted average number of shares (in millions): |
||||||||||||
| Basic |
||||||||||||
| |
|
|
|
|
|
|||||||
| Diluted |
||||||||||||
| |
|
|
|
|
|
|||||||
Year ended December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
| Net income (loss) |
$ | $ | ( |
) | $ | ( |
) | |||||
| Other comprehensive income (loss), net of tax: |
||||||||||||
| Currency translation adjustment |
( |
) | ( |
) | ||||||||
| Unrealized gain (loss) on derivative financial instruments, net |
||||||||||||
| Unrealized gain (loss) on available-for-sale securities, net |
( |
) | ||||||||||
| Unrealized gain (loss) on defined benefit plans, net |
( |
) | ( |
) | ||||||||
| |
|
|
|
|
|
|||||||
| Total other comprehensive income (loss) |
( |
) | ( |
) | ||||||||
| |
|
|
|
|
|
|||||||
| Total comprehensive income (loss) |
( |
) | ( |
) | ||||||||
| Comprehensive income (loss) attributable to non-controlling interests |
( |
) | ( |
) | ||||||||
| |
|
|
|
|
|
|||||||
| Comprehensive income (loss) attributable to Teva |
$ | $ | ( |
) | $ | ( |
) | |||||
| |
|
|
|
|
|
|||||||
Teva shareholders’ equity |
||||||||||||||||||||||||||||||||||||
Ordinary shares |
||||||||||||||||||||||||||||||||||||
Number of shares (in millions) |
Stated value |
Additional paid-in capital |
Retained earnings (accumulated deficit) |
Accumulated other comprehensive income (loss) |
Treasury shares |
Total Teva share-holders’ equity |
Non- controlling interests |
Total equity |
||||||||||||||||||||||||||||
(U.S. dollars in millions) |
||||||||||||||||||||||||||||||||||||
| Balance at January 1, 2019 |
( |
) | ( |
) | ( |
) | ||||||||||||||||||||||||||||||
| Changes during 2019: |
||||||||||||||||||||||||||||||||||||
| Net income (loss) |
( |
) | ( |
) | ( |
) | ( |
) | ||||||||||||||||||||||||||||
| Other comprehensive income (loss) |
||||||||||||||||||||||||||||||||||||
| Issuance of Treasury Shares |
* | ( |
) | |||||||||||||||||||||||||||||||||
| Stock-based compensation expense |
||||||||||||||||||||||||||||||||||||
| Issuance of shares |
* | * | ||||||||||||||||||||||||||||||||||
| Transactions with non-controlling interests |
( |
) | ( |
) | ||||||||||||||||||||||||||||||||
| Other |
( |
) | ( |
) | ( |
) | ||||||||||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Balance at December 31, 2019 |
( |
) | ( |
) | ( |
) | ||||||||||||||||||||||||||||||
| Changes during 2020: |
||||||||||||||||||||||||||||||||||||
| Net income (loss) |
( |
) | ( |
) | ( |
) | ( |
) | ||||||||||||||||||||||||||||
| Other comprehensive income (loss) |
( |
) | ( |
) | ( |
) | ||||||||||||||||||||||||||||||
| Issuance of Shares |
* | * | ||||||||||||||||||||||||||||||||||
| Stock-based compensation expense |
||||||||||||||||||||||||||||||||||||
| Transactions with non-controlling interests |
( |
) | ( |
) | ||||||||||||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Balance at December 31, 2020 |
( |
) | ( |
) | ( |
) | ||||||||||||||||||||||||||||||
| Changes during 2021: |
||||||||||||||||||||||||||||||||||||
| Net income (loss) |
||||||||||||||||||||||||||||||||||||
| Other comprehensive income (loss) |
( |
) | ( |
) | ( |
) | ( |
) | ||||||||||||||||||||||||||||
| Issuance of Shares |
* | * | * | |||||||||||||||||||||||||||||||||
| Stock-based compensation expense |
||||||||||||||||||||||||||||||||||||
| Transactions with non-controlling interests |
( |
) | ( |
) | ||||||||||||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Balance at December 31, 2021 |
$ | $ | $ | ( |
) | $ | ( |
) | $ | ( |
) | $ | $ | $ | ||||||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| * | Represents an amount less than |
Year ended December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
| Operating activities: |
||||||||||||
| Net income (loss) |
$ | $ | ( |
) | $ | ( |
) | |||||
| Adjustments to reconcile net loss to net cash provided by operations: |
||||||||||||
| Impairment of goodwill, long-lived assets and assets held for sale |
||||||||||||
| Depreciation and amortization |
||||||||||||
| Net change in operating assets and liabilities |
( |
) | ( |
) | ( |
) | ||||||
| Deferred income taxes—net and uncertain tax positions |
( |
) | ( |
) | ( |
) | ||||||
| Stock-based compensation |
||||||||||||
| Other items |
||||||||||||
| Research and development in process |
||||||||||||
| Net loss (gain) from investments and from sale of business and long lived assets |
( |
) | ( |
) | ||||||||
| |
|
|
|
|
|
|||||||
| Net cash provided by operating activities |
||||||||||||
| |
|
|
|
|
|
|||||||
| Investing activities: |
||||||||||||
| Beneficial interest collected in exchange for securitized trade receivables |
||||||||||||
| Proceeds from sale of business and long lived assets |
||||||||||||
| Purchases of property, plant and equipment |
( |
) | ( |
) | ( |
) | ||||||
| Purchases of investments and other assets |
( |
) | ( |
) | ( |
) | ||||||
| Proceeds from sale of investments |
||||||||||||
| Other investing activities |
||||||||||||
| |
|
|
|
|
|
|||||||
| Net cash provided by investing activities |
||||||||||||
| |
|
|
|
|
|
|||||||
| Financing activities: |
||||||||||||
| Repayment of senior notes and loans and other long term liabilities |
( |
) | ( |
) | ( |
) | ||||||
| Proceeds from senior notes, net of issuance costs |
||||||||||||
| Proceeds from short term debt |
||||||||||||
| Repayment of short term debt |
( |
) | ( |
) | ( |
) | ||||||
| Redemption of convertible debentures |
( |
) | ||||||||||
| Other financing activities |
( |
) | ( |
) | ( |
) | ||||||
| Tax withholding payments made on shares and dividends |
( |
) | ||||||||||
| |
|
|
|
|
|
|||||||
| Net cash used in financing activities |
( |
) | ( |
) | ( |
) | ||||||
| |
|
|
|
|
|
|||||||
| Translation adjustment on cash and cash equivalents |
( |
) | ||||||||||
| |
|
|
|
|
|
|||||||
| Net change in cash, cash equivalents and restricted cash |
||||||||||||
| Balance of cash, cash equivalents and restricted cash at beginning of year |
||||||||||||
| |
|
|
|
|
|
|||||||
| Balance of cash, cash equivalents and restricted cash at end of year |
$ | $ | $ | |||||||||
| |
|
|
|
|
|
|||||||
| Reconciliation of cash, cash equivalents and restricted cash reported in the consolidated balance sheets: |
||||||||||||
| Cash and cash equivalents |
||||||||||||
| Restricted cash included in other current assets |
||||||||||||
| |
|
|
|
|
|
|||||||
| Total cash, cash equivalents and restricted cash shown in the statement of cash flows |
||||||||||||
| |
|
|
|
|
|
|||||||
Year ended December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
| Supplemental cash flow information: |
||||||||||||
| Non-cash financing and investing activities: |
||||||||||||
| Beneficial interest obtained in exchange for securitized trade receivables |
$ | $ | $ | |||||||||
| Cash paid during the year for: |
||||||||||||
| Interest |
$ | $ | $ | |||||||||
| Income taxes, net of refunds |
$ | $ | $ | |||||||||
Year ended December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
| Other current assets |
$ | ( |
) | $ | ( |
) | $ | ( |
) | |||
| Trade payables, accrued expenses, employee-related obligations and other liabilities |
( |
) | ||||||||||
| Trade receivables net of sales reserves and allowances |
( |
) | ( |
) | ( |
) | ||||||
| Inventories |
||||||||||||
| |
|
|
|
|
|
|||||||
| $ | ( |
) | $ | ( |
) | $ | ( |
) | ||||
| |
|
|
|
|
|
|||||||
b. |
New accounting pronouncements |
c. |
Acquisitions: |
d. |
Collaborative arrangements: |
e. |
Equity investments: |
f. |
Fair value measurement: |
g. |
Investment in debt securities: |
h. |
Cash and cash equivalents: |
i. |
Restricted cash: |
j. |
Accounts Receivable: |
k. |
Concentration of credit risks: |
l. |
Inventories: |
m. |
Long-lived assets: |
| 1. | An initial qualitative assessment may be performed to determine whether it is more likely than not that the fair value of the reporting unit is less than its carrying amount. |
| 2. | If the Company concludes it is more likely than not that the fair value of the reporting unit is less than its carrying mount, a quantitative fair value test is performed. An impairment charge for the amount by which the carrying amount exceeds the reporting unit’s fair value is recognized. |
n. |
Contingencies: |
o. |
Treasury shares: |
p. |
Stock-based compensation: |
q. |
Deferred income taxes: |
1. |
Taxes that would apply in the event of disposal of investments in subsidiaries, as it is generally the Company’s intention to hold these investments, not to realize them. The determination of the amount of related unrecognized deferred tax liability is not practicable. |
2. |
Amounts of tax-exempt income generated from the Company’s current Approved Enterprises and unremitted earnings from foreign subsidiaries retained for reinvestment in the Group. See note 13f. |
r. |
Uncertain tax positions: |
s. |
Derivatives and hedging: |
t. |
Revenue recognition: |
u. |
Research and development: |
v. |
Shipping and handling costs: |
w. |
Advertising costs: |
x. |
Restructuring: |
y. |
Segment reporting: |
| (a) | North America segment, which includes the United States and Canada. |
| (b) | Europe segment, which includes the European Union, the United Kingdom and certain other European countries. |
| (c) | International Markets segment, which includes all countries in which Teva operates other than those in the North America and Europe segments. |
z. |
Earnings per share: |
aa. |
Securitization |
bb. |
Divestitures |
cc. |
Debt instruments |
dd. |
Leases |
December 31, 2021 |
December 31, 2020 |
|||||||
(U.S. $ in millions) |
||||||||
| Inventories |
||||||||
| Property, plant and equipment, net and others |
||||||||
| Goodwill |
||||||||
| Adjustments of assets held for sale to fair value |
( |
) | ( |
) | ||||
| |
|
|
|
|||||
| Total assets of the disposal group classified as held for sale in the consolidated balance sheets |
$ | $ | ||||||
| |
|
|
|
|||||
| Total liabilities of the disposal group classified as held for sale in the consolidated balance sheets under accrued expenses ($ |
$ | ( |
) | $ | ||||
| |
|
|
|
|||||
Year ended December 31, 2021 |
||||||||||||||||||||
North America |
Europe |
International Markets |
Other activities |
Total |
||||||||||||||||
(U.S.$ in millions) |
||||||||||||||||||||
| Sale of goods |
||||||||||||||||||||
| Licensing arrangements |
||||||||||||||||||||
| Distribution |
||||||||||||||||||||
| Other |
( |
) | ||||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| $ | $ | $ | $ | $ | ||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
Year ended December 31, 2020 |
||||||||||||||||||||
North America |
Europe |
International Markets |
Other activities |
Total |
||||||||||||||||
(U.S.$ in millions) |
||||||||||||||||||||
| Sale of goods |
||||||||||||||||||||
| Licensing arrangements |
||||||||||||||||||||
| Distribution |
||||||||||||||||||||
| Other |
§ | ( |
) | |||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| $ | $ | $ | $ | $ | ||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
Year ended December 31, 2019 |
||||||||||||||||||||
North America |
Europe |
International Markets |
Other activities |
Total |
||||||||||||||||
(U.S.$ in millions) |
||||||||||||||||||||
| Sale of goods |
||||||||||||||||||||
| Licensing arrangements |
||||||||||||||||||||
| Distribution |
||||||||||||||||||||
| Other |
§ | ( |
) | |||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| $ | $ | $ | $ | $ | ||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| § | Represents an amount less than $1 million. |
Sales Reserves and Allowances |
||||||||||||||||||||||||||||||||
Reserves included in Accounts Receivable, net |
Rebates |
Medicaid and other governmental allowances |
Chargebacks |
Returns |
Other |
Total reserves included in Sales Reserves and Allowances |
Total |
|||||||||||||||||||||||||
(U.S.$ in millions) |
||||||||||||||||||||||||||||||||
Balance at January 1, 2020 |
$ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||
Provisions related to sales made in current year period |
$ | |||||||||||||||||||||||||||||||
Provisions related to sales made in prior periods |
( |
) | ( |
) | ( |
) | ( |
) | ( |
) | ( |
) | $ | ( |
) | |||||||||||||||||
Credits and payments |
( |
) | ( |
) | ( |
) | ( |
) | ( |
) | ( |
) | ( |
) | $ | ( |
) | |||||||||||||||
Translation differences |
$ | |||||||||||||||||||||||||||||||
Balance at December 31, 2020 |
$ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||
Provisions related to sales made in current year period |
$ | |||||||||||||||||||||||||||||||
Provisions related to sales made in prior periods |
( |
) | ( |
) | ( |
) | ( |
) | ( |
) | ( |
) | ( |
) | $ | ( |
) | |||||||||||||||
Credits and payments |
( |
) | ( |
) | ( |
) | ( |
) | ( |
) | ( |
) | ( |
) | $ | ( |
) | |||||||||||||||
Translation differences |
( |
) | ( |
) | ( |
) | ( |
) | ( |
) | ( |
) | $ | ( |
) | |||||||||||||||||
Balance at December 31, 2021 |
$ | $ | ||||||||||||||||||||||||||||||
December 31, |
||||||||
2021 |
2020 |
|||||||
(U.S. $ in millions) |
||||||||
Finished products |
$ | $ | ||||||
Raw and packaging materials |
||||||||
Products in process |
||||||||
Materials in transit and payments on account |
||||||||
| $ | $ | |||||||
December 31, |
||||||||
2021 |
2020 |
|||||||
(U.S. $ in millions) |
||||||||
Machinery and equipment |
$ | $ | ||||||
Buildings |
||||||||
Computer equipment and other assets |
||||||||
Assets under construction and payments on account |
||||||||
Land |
||||||||
Less—accumulated depreciation |
( |
) | ( |
) | ||||
| $ |
$ |
|||||||
Gross carrying amount net of impairment |
Accumulated amortization |
Net carrying amount |
||||||||||||||||||||||
December 31, |
||||||||||||||||||||||||
2021 |
2020 |
2021 |
2020 |
2021 |
2020 |
|||||||||||||||||||
(U.S. $ in millions) |
||||||||||||||||||||||||
Product rights |
$ | $ | $ | $ | $ | $ | ||||||||||||||||||
Trade names |
||||||||||||||||||||||||
In-process research and development (IPR&D) |
— | — | ||||||||||||||||||||||
Total |
$ | $ | $ | $ | $ | $ | ||||||||||||||||||
| (a) | Identifiable product rights and trade names of $ ® ), resulting from modified competition assumptions as a result of settlements between the innovator and other generic filers; and |
| (b) | IPR&D assets of $ |
| (a) | IPR&D assets of $ ® ), due to modified competition assumptions as a result of settlements between the innovator and other generic filers; (iii) $ |
| (b) | Identifiable product rights of $ |
| (a) | Identifiable product rights of $ |
| (b) | IPR&D assets of $ ® ), due to modified competition assumptions as a result of settlements between the innovator and other generic filers, and (iii) $ |
North America |
Europe |
International Markets |
Other |
Total |
||||||||||||||||
(U.S. $ in millions) |
||||||||||||||||||||
| Balance as of December 31, 2019 (1) |
$ | $ | $ | $ | $ | |||||||||||||||
| Changes during the period: |
||||||||||||||||||||
| Goodwill reclassified as assets held for sale |
— | ( |
) | ( |
) | — | ( |
) | ||||||||||||
| Goodwill impairment |
( |
) | — | — | — | ( |
) | |||||||||||||
| Translation differences |
( |
) | — | |||||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| Balance as of December 31, 2020 (1) |
$ | $ | $ | $ | $ | |||||||||||||||
| Changes during the period: |
||||||||||||||||||||
| Goodwill reclassified as assets held for sale |
— | ( |
) | — | ( |
) | ( |
) | ||||||||||||
| Translation differences |
( |
) | ( |
) | ( |
) | ||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| Balance as of December 31, 2021 (1) |
$ | $ | $ | $ | $ | |||||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| (1) | Accumulated goodwill impairment as of December 31, 2021, December 31, 2020 and December 31, 2019 was approximately $ |
Year ended December 31, |
Year ended December 31, |
Year ended December 31, |
||||||||||
2021 |
2020 |
2019 |
||||||||||
(U.S. $ in millions) |
(U.S. $ in millions) |
(U.S. $ in millions) |
||||||||||
| Operating lease cost: |
||||||||||||
| Fixed payments and variable payments that depend on an index or rate |
$ | $ | $ | |||||||||
| Variable lease payments not included in the lease liability |
||||||||||||
| Short-term lease cost |
||||||||||||
| |
|
|
|
|
|
|||||||
| $ | $ | $ | ||||||||||
| |
|
|
|
|
|
|||||||
Year ended December 31, |
Year ended December 31, |
Year ended December 31, |
||||||||||
2021 |
2020 |
2019 |
||||||||||
(U.S. $ in millions) |
(U.S. $ in millions) |
(U.S. $ in millions) |
||||||||||
| Cash paid for amounts included in the measurement of lease liabilities: |
||||||||||||
| Operating cash flows from operating leases |
$ | $ | $ | |||||||||
| Right-of-use assets obtained in exchange for lease obligations (non-cash): |
||||||||||||
| Operating leases |
$ | $ | $ | |||||||||
December 31, |
December 31, |
|||||||
2021 |
2020 |
|||||||
(U.S. $ in millions) |
(U.S. $ in millions) |
|||||||
| Operating leases: |
||||||||
| Operating lease ROU assets |
$ | $ | ||||||
| Other current liabilities |
||||||||
| s |
||||||||
| |
|
|
|
|||||
| Total operating lease liabilities |
$ | $ | ||||||
| |
|
|
|
|||||
December 31, |
December 31, |
|||||||
2021 |
2020 |
|||||||
| Weighted average remaining lease term |
||||||||
| Operating leases |
||||||||
| Weighted average discount rate |
||||||||
| Operating leases |
% | % | ||||||
December 31, |
||||
2021 |
||||
(U.S. $ in millions) |
||||
| 2022 |
$ | |||
| 2023 |
||||
| 2024 |
||||
| 2025 |
||||
| 2026 and thereafter |
||||
| |
|
|||
| Total operating lease payments |
$ | |||
| |
|
|||
| Less: imputed interest |
||||
| |
|
|||
| Present value of lease liabilities |
$ | |||
| |
|
|||
a. |
Short-term debt: |
December 31, |
||||||||||||||||
Weighted average interest rate as of December 31, 2021 |
Maturity |
2021 |
2020 |
|||||||||||||
(U.S. $ in millions) |
||||||||||||||||
| Convertible debentures |
% | |||||||||||||||
| Current maturities of long-term liabilities |
|
|||||||||||||||
| |
|
|
|
|||||||||||||
| Total short term debt |
|
$ | $ | |||||||||||||
| |
|
|
|
|||||||||||||
b. |
Long-term debt: |
Weighted average interest rate as of December 31, 2021 |
Maturity |
December 31, 2021 |
December 31, 2020 |
|||||||||||||
% |
(U.S. $ in millions) |
|||||||||||||||
| Senior notes EUR |
% | |||||||||||||||
| Sustainability-linked senior notes EUR |
% | — | ||||||||||||||
| Senior notes EUR |
% | |||||||||||||||
| Sustainability-linked senior notes EUR |
% | — | ||||||||||||||
| Senior notes EUR |
% | |||||||||||||||
| Senior notes EUR |
% | |||||||||||||||
| Senior notes EUR |
% | |||||||||||||||
| Senior notes EUR |
% | |||||||||||||||
| Senior notes EUR |
% | |||||||||||||||
| Senior notes USD |
% | |||||||||||||||
| Senior notes USD |
% | |||||||||||||||
| Senior notes USD |
% | |||||||||||||||
| Senior notes USD |
% | — | ||||||||||||||
| Senior notes USD |
% | |||||||||||||||
| Senior notes USD |
% | |||||||||||||||
| Senior notes USD |
% | |||||||||||||||
| Sustainability-linked senior notes USD |
% | — | ||||||||||||||
| Sustainability-linked senior notes USD |
% | — | ||||||||||||||
| Senior notes USD |
% | |||||||||||||||
| Senior notes USD |
% | |||||||||||||||
Weighted average interest rate as of December 31, 2021 |
Maturity |
December 31, 2021 |
December 31, 2020 |
|||||||||||||
% |
(U.S. $ in millions) |
|||||||||||||||
| Senior notes USD |
% | — | ||||||||||||||
| Senior notes USD |
% | — | ||||||||||||||
| Senior notes CHF |
% | |||||||||||||||
| Senior notes CHF |
% | |||||||||||||||
| |
|
|
|
|||||||||||||
| Total senior notes |
||||||||||||||||
| Other long-term debt |
||||||||||||||||
| Less current maturities |
( |
) | ( |
) | ||||||||||||
| Less debt issuance costs |
( |
) | ( |
) | ||||||||||||
| |
|
|
|
|||||||||||||
| Total senior notes and loans |
$ | $ | ||||||||||||||
| |
|
|
|
|||||||||||||
| (1) | In July 2021, Teva repaid $ |
| (2) | In November 2021, Teva issued sustainability-linked senior notes in an aggregate principal amount of |
| (3) | In November 2021, Teva issued sustainability-linked senior notes in an aggregate principal amount of |
| (4) | In November 2021, Teva issued sustainability-linked senior notes in an aggregate principal amount of $ |
| (5) | In November 2021, Teva issued sustainability-linked senior notes in an aggregate principal amount of $ |
| (6) | In November 2021, Teva consummated a cash tender offer and extinguished |
| (7) | In November 2021, Teva repaid $ |
| (8) | Debt issuance costs as of December 31, 2021 include $ million in connection with the issuance of the sustainability-linked senior notes in November 2021. |
| * | Interest rate adjustments and a potential one-time premium payment related to the sustainability-linked bonds are treated as bifurcated embedded derivatives. See note 10c. |
December 31, 2021 |
||||
(U.S. $ in millions) |
||||
| 2023 |
$ | |||
| 2024 |
||||
| 2025 |
||||
| 2026 * |
||||
| 2027 and thereafter |
||||
| |
|
|||
| $ | ||||
| |
|
|||
| * | including $ |
a. |
Foreign exchange risk management: |
b. |
Interest risk management: |
c. |
Bifurcated embedded derivatives: |
d. |
Derivative instrument outstanding: |
Fair value |
||||||||
Not designated as hedging instruments |
||||||||
December 31, 2021 |
December 31, 2020 |
|||||||
| Reported under |
(U.S. $ in millions) |
|||||||
| Asset derivatives: |
||||||||
| Other current assets: |
||||||||
| Option and forward contracts |
$ | $ | ||||||
| Liability derivatives: |
||||||||
| Other current liabilities: |
||||||||
| Option and forward contracts |
( |
) | ( |
) | ||||
Financial expenses, net |
Other comprehensive income (loss) |
|||||||||||||||||||||||
Year ended December 31, |
Year ended December 31, |
|||||||||||||||||||||||
2021 |
2020 |
2019 |
2021 |
2020 |
2019 |
|||||||||||||||||||
| Reported under |
(U.S. $ in millions) |
|||||||||||||||||||||||
| Line items in which effects of hedges are recorded |
$ | $ | $ | $ | ( |
) | $ | ( |
) | $ | ||||||||||||||
| Cross-currency swaps—cash flow hedge (1) |
( |
) | ( |
) | ||||||||||||||||||||
| Cross-currency swaps—net investment hedge (2) |
( |
) | ( |
) | ( |
) | ( |
) | ||||||||||||||||
| Interest rate swaps—fair value hedge (3) . |
||||||||||||||||||||||||
Financial expenses, net |
Net revenues |
|||||||||||||||||||||||
Year ended December 31, |
Year ended December 31, |
|||||||||||||||||||||||
2021 |
2020 |
2019 |
2021 |
2020 |
2019 |
|||||||||||||||||||
| Reported under |
(U.S. $ in millions) |
|||||||||||||||||||||||
| Line items in which effects of hedges are recorded |
$ | $ | $ | $ | $ | $ | ||||||||||||||||||
| Option and forward contracts (4) |
( |
) | ( |
) | — | — | — | |||||||||||||||||
| Option and forward contracts (5) |
— | — | * | |||||||||||||||||||||
| * | Represents an amount less than $ |
| (1) | With respect to cross-currency swap agreements, Teva recognized gains which mainly reflect the differences between the fixed interest rate and the floating interest rate. In the fourth quarter of 2019, Teva terminated $ |
| (2) | In each of the first and second quarters of 2017, Teva entered into a cross currency swap agreement with a notional amount of $ |
| (3) | In the fourth quarter of 2016, Teva entered into an interest rate swap agreement designated as fair value hedge relating to its |
| (4) | Teva uses foreign exchange contracts (mainly option and forward contracts) to hedge balance sheet items from currency exposure. These foreign exchange contracts are not designated as hedging instruments for accounting purposes. In connection with these foreign exchange contracts, Teva recognizes gains or losses that offset the revaluation of the balance sheet items also recorded under financial expenses, net. |
| (5) | Teva entered into option and forward contracts designed to limit the exposure of foreign exchange fluctuations on projected revenues and expenses recorded in euro, the Swiss franc, the Japanese yen, the British pound, the Russian ruble, the Canadian dollar and some other currencies to protect its projected operating results for 2021 and 2022. These derivative instruments do not meet the criteria for hedge accounting, however, they are accounted for as an economic hedge. These derivative instruments, which may include hedging transactions against future projected revenues and expenses, are recognized on the balance sheet at their fair value on a quarterly basis, while the foreign exchange impact on the underlying revenues and expenses may occur in subsequent quarters. In 2021, the positive impact from these derivatives recognized under revenues was $ |
e. |
Amortizations due to terminated derivative instruments: |
f. |
Securitization: |
As of and for the year ended December 31, |
||||||||
2021 |
2020 |
|||||||
(U.S. $ in millions) |
||||||||
| Sold receivables at the beginning of the year |
$ | $ | ||||||
| Proceeds from sale of receivables |
||||||||
| Cash collections (remitted to the owner of the receivables) |
( |
) | ( |
) | ||||
| Effect of currency exchange rate changes |
( |
) | ||||||
| |
|
|
|
|||||
| Sold receivables at the end of the year |
$ | $ | ||||||
| |
|
|
|
|||||
a. |
Commitments: |
b. |
Contingencies: |
a. |
Income (loss) before income taxes: |
Year ended December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
(U.S. $ in millions) |
||||||||||||
Parent Company and its Israeli subsidiaries |
$ | $ | $ | |||||||||
Non-Israeli subsidiaries |
( |
) | ( |
) | ||||||||
| $ | $ | ( |
) | $ | ( |
) | ||||||
b. |
Income taxes: |
Year ended December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
(U.S. $ in millions) |
||||||||||||
In Israel |
$ | $ | $ | |||||||||
Outside Israel |
( |
) | ( |
) | ||||||||
| $ | $ | ( |
) | $ | ( |
) | ||||||
Current |
$ | $ | $ | |||||||||
Deferred |
( |
) | ( |
) | ( |
) | ||||||
| $ | $ | ( |
) | $ | ( |
) | ||||||
2021 |
2020 |
2019 |
||||||||||
(U.S. $ in millions) |
||||||||||||
Income (loss) before income taxes |
$ | $ | ( |
) | $ | ( |
) | |||||
Statutory tax rate in Israel |
% | % | % | |||||||||
Theoretical provision for income taxes |
$ | $ | ( |
) | $ | ( |
) | |||||
Increase (decrease) in the provision for income taxes due to: |
||||||||||||
The Parent Company and its Israeli subsidiaries - Tax benefits arising from reduced tax rates under benefit programs |
( |
) | ( |
) | ( |
) | ||||||
Mainly nondeductible items and prior year tax |
( |
) |
||||||||||
Non-Israeli subsidiaries, including impairments (*) |
( |
) | ||||||||||
Increase (decrease) in other uncertain tax positions—net |
( |
) | ( |
) | ||||||||
Effective consolidated income taxes |
$ | $ | ( |
) | $ | ( |
) | |||||
(*) |
In 2020, income before income taxes includes goodwill impairment in non-Israeli subsidiaries that did not have a corresponding tax effect. |
c. |
Deferred income taxes: |
December 31, |
||||||||
2021 |
2020 |
|||||||
(U.S. $ in millions) |
||||||||
Deferred tax assets (liabilities), net: |
||||||||
Inventory related |
$ | $ | ||||||
Sales reserves and allowances |
||||||||
Provision for legal settlements |
||||||||
Intangible assets (*) |
( |
) | ( |
) | ||||
Carryforward losses and deductions and credits (**) |
||||||||
Property, plant and equipment |
( |
) | ( |
) | ||||
Deferred interest |
||||||||
Provisions for employee related obligations |
||||||||
Other |
||||||||
Valuation allowance—in respect of carryforward losses and deductions that may not be utilized |
( |
) | ( |
) | ||||
| $ | ( |
) | $ | ( |
) | |||
| (*) | The decrease in deferred tax liability is mainly due to impairment and amortization. |
| (**) | The amounts are shown following a reduction for unrecognized tax benefits of $ |
The amount as of December 31, 2021 represents the tax effect of gross carryforward losses and deductions with the following expirations: - — $ |
December 31, |
||||||||
2021 |
2020 |
|||||||
(U.S. $ in millions) |
||||||||
Long-term assets—deferred income taxes |
||||||||
Long-term liabilities—deferred income taxes |
( |
) | ( |
) | ||||
| $ | ( |
) | $ | ( |
) | |||
d. |
Uncertain tax positions: |
Year ended December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
(U.S. $ in millions) |
||||||||||||
Balance at the beginning of the year |
$ | $ | $ | |||||||||
Increase (decrease) related to prior year tax positions, net |
( |
) | ( |
) | ||||||||
Increase related to current year tax positions |
||||||||||||
Decrease related to settlements with tax authorities and lapse of applicable statutes of limitations |
( |
) | ( |
) | ( |
) | ||||||
Other |
( |
) | ( |
) | — | |||||||
Balance at the end of the year |
$ | $ | $ | |||||||||
e. |
Tax assessments: |
f. |
Basis of taxation: |
| • | Investment of at least |
| • | One of the following: |
| a. | At least |
| b. | A venture capital investment approximately equivalent to at least $ |
| c. | Growth in sales or workforce by an average of |
a. |
Ordinary shares and ADSs |
b. |
Stock-based compensation plans |
Year ended December 31, |
||||||||||||||||||||||||
2021 |
2020 |
2019 |
||||||||||||||||||||||
Number (in thousands) |
Weighted average exercise price |
Number (in thousands) |
Weighted average exercise price |
Number (in thousands) |
Weighted average exercise price |
|||||||||||||||||||
| Balance outstanding at beginning of year |
$ | $ | $ | |||||||||||||||||||||
| Changes during the year: |
||||||||||||||||||||||||
| Exercised |
— | — | — | — | ( |
) | ||||||||||||||||||
| Forfeited |
( |
) | ( |
) | ( |
) | ||||||||||||||||||
| Expired |
( |
) | ( |
) | — | — | ||||||||||||||||||
| |
|
|
|
|
|
|||||||||||||||||||
| Balance outstanding at end of year |
||||||||||||||||||||||||
| |
|
|
|
|
|
|||||||||||||||||||
| Balance exercisable at end of year |
||||||||||||||||||||||||
| |
|
|
|
|
|
|||||||||||||||||||
| (1) Number of ordinary shares issuable upon exercise of outstanding options |
||||||||||||
| Range of exercise prices |
Balance at end of period (in thousands) |
Weighted average exercise price |
Weighted average remaining life |
|||||||||
Number of shares |
$ |
Years |
||||||||||
| Lower than $ |
||||||||||||
| $ |
||||||||||||
| $ |
||||||||||||
| $ |
||||||||||||
| $ |
||||||||||||
| $ |
||||||||||||
| |
|
|||||||||||
| Total |
||||||||||||
| |
|
|||||||||||
(2) Number of ordinary shares issuable upon exercise of vested options |
||||||||||||
Range of exercise prices |
Balance at end of period (in thousands) |
Weighted average exercise price |
Weighted average remaining life |
|||||||||
Number of shares |
$ |
Years |
||||||||||
Lower than $ |
||||||||||||
$ |
||||||||||||
$ |
||||||||||||
$ |
||||||||||||
$ |
||||||||||||
$ |
||||||||||||
Total |
||||||||||||
Year ended December 31, |
||||||||||||||||||||||||
2021 |
2020 |
2019 |
||||||||||||||||||||||
Number (in thousands) |
Weighted average grant date fair value |
Number (in thousands) |
Weighted average grant date fair value |
Number (in thousands) |
Weighted average grant date fair value |
|||||||||||||||||||
Balance outstanding at beginning of year |
$ | $ | $ | |||||||||||||||||||||
Granted |
||||||||||||||||||||||||
Vested |
( |
) | ( |
) | ( |
) | ||||||||||||||||||
Forfeited |
( |
) | ( |
) | ( |
) | ||||||||||||||||||
Balance outstanding at end of year |
||||||||||||||||||||||||
Year ended December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
(U.S. $ in millions) |
||||||||||||
Employee stock options |
$ | $ | $ | |||||||||
RSUs and PSUs |
||||||||||||
Total stock-based compensation expense |
||||||||||||
Tax effect on stock-based compensation expense |
||||||||||||
Net effect |
$ | $ | $ | |||||||||
d. |
Dividends |
e. |
Accumulated other comprehensive loss |
Net Unrealized Gains/(Losses) |
Benefit Plans |
|||||||||||||||||||
Foreign currency translation adjustments |
Available- for-sale securities |
Derivative financial instruments |
Actuarial gains/(losses) and prior service (costs)/credits |
Total |
||||||||||||||||
(U.S. $ in millions) |
||||||||||||||||||||
Balance as of January 1, 2019 |
$ | ( |
) | $ |
$ |
( |
) | $ |
( |
) | $ |
( |
) | |||||||
Other comprehensive income/(loss) before reclassifications |
( |
) | ( |
) | ||||||||||||||||
Amounts reclassified to the statements of income |
( |
) |
||||||||||||||||||
Net other comprehensive income/(loss) before tax |
( |
) | ( |
) | ||||||||||||||||
Corresponding income tax |
( |
) | — | — | ( |
) | ||||||||||||||
Net other comprehensive income/(loss) after tax* |
( |
) | ( |
) | ||||||||||||||||
Balance as of December 31, 2019 |
( |
) | — | ( |
) | ( |
) | ( |
) | |||||||||||
Other comprehensive income/(loss) before reclassifications |
( |
) | — | ( |
) | ( |
) | |||||||||||||
Amounts reclassified to the statements of income |
— |
— |
( |
) | ||||||||||||||||
Net other comprehensive income/(loss) before tax |
( |
) | — | ( |
) | ( |
) | |||||||||||||
Corresponding income tax |
— | — | ||||||||||||||||||
Net other comprehensive income/(loss) after tax* |
( |
) | — | ( |
) | ( |
) | |||||||||||||
Balance as of December 31, 2020 |
( |
) | — | ( |
) | ( |
) | ( |
) | |||||||||||
Other comprehensive income/(loss) before reclassifications |
( |
) | — |
( |
) | |||||||||||||||
Amounts reclassified to the statements of income |
— | — | ||||||||||||||||||
Net other comprehensive income/(loss) before tax |
( |
) | — | ( |
) | |||||||||||||||
Corresponding income tax |
— | — | ( |
) | ||||||||||||||||
Net other comprehensive income/(loss) after tax* |
( |
) | — | ( |
) | |||||||||||||||
Balance as of December 31, 202 1 |
$ | ( |
) | — | $ | ( |
) | $ | ( |
) | $ | ( |
) | |||||||
| * | Amounts do not include foreign currency translation adjustments attributable to non-controlling interests of $ |
Year ended December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
(U.S. $ in millions) |
||||||||||||
Impairment of long-lived tangible assets (1) |
$ | $ | $ | |||||||||
Contingent consideration (see note 20) |
( |
) | ||||||||||
Restructuring |
||||||||||||
Other |
||||||||||||
Total |
$ | $ | $ | |||||||||
| (1) | Including impairments related to exit and disposal activities. |
Year ended December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
(U.S. $ in millions) |
||||||||||||
Restructuring |
||||||||||||
Employee termination |
$ | $ | $ | |||||||||
Other |
||||||||||||
Total |
$ | $ | $ | |||||||||
Employee termination costs |
Other |
Total |
||||||||||
(U.S. $ in millions ) |
||||||||||||
Balance as of January 1, 2019 |
$ | ( |
) | $ | ( |
) | $ | ( |
) | |||
Provision |
( |
) | ( |
) | ( |
) | ||||||
Utilization and other* |
||||||||||||
Balance as of January 1, 2020 |
$ | ( |
) | $ | ( |
) | $ | ( |
) | |||
Provision |
( |
) | ( |
) | ( |
) | ||||||
Utilization and other* |
||||||||||||
Balance as of December 31, 2020 |
$ | ( |
) | $ | ( |
) | $ | ( |
) | |||
Provision |
( |
) | ( |
) | ( |
) | ||||||
Utilization and other* |
||||||||||||
Balance as of December 31, 2021 |
$ | ( |
) | $ | ( |
) | $ | ( |
) | |||
| * | Includes adjustments for foreign currency translation. |
Year ended December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
(U.S. $ in millions) |
||||||||||||
| Gain on divestitures, net of divestitures related costs (1) |
||||||||||||
| Section 8 and similar payments (2) |
— | |||||||||||
| Gain (loss) on sale of assets |
( |
) | ||||||||||
| Other, net |
||||||||||||
| |
|
|
|
|
|
|||||||
| Total other income |
$ | $ | $ | |||||||||
| |
|
|
|
|
|
|||||||
| (1) | In 2021, mainly due to capital gains related to the sale of certain OTC assets. In 2020 and 2019, mainly related to the divestment of several activities in the International Markets segment. |
| (2) | Section 8 of the Patented Medicines (Notice of Compliance) Regulation relates to recoveries of lost revenue related to patent infringement proceedings in Canada. |
Year ended December, 31 |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
(U.S. $ in millions) |
||||||||||||
| Interest expenses and other bank charges |
||||||||||||
| (Income) loss from investments (1) |
( |
) | ( |
) | ||||||||
| Foreign exchange (gains) losses, net |
( |
) | ( |
) | ||||||||
| Other, net (2) |
||||||||||||
| |
|
|
|
|
|
|||||||
| Total finance expense, net |
$ | $ | $ | |||||||||
| |
|
|
|
|
|
|||||||
| |
(1) |
(Income) loss from investments in 2021 and 2020 comprised mainly of revaluation gains and loss of Teva’s investment in American Well Corporation (“American Well”). See note 20. |
(2) |
Amortization of issuance costs and terminated derivative instruments. |
Year ended December, 31 |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
(U.S. $ in millions, except share data) |
||||||||||||
| Net income (loss) used for the computation of basic and diluted earnings (loss) per share |
$ | ( |
) | $ | ( |
) | ||||||
| |
|
|
|
|
|
|||||||
| Weighted average number of shares used in the computation of basic earnings (loss) per share |
||||||||||||
| |
|
|
|
|
|
|||||||
| Weighted average number of shares used in the computation of diluted earnings (loss) per share |
||||||||||||
| |
|
|
|
|
|
|||||||
| (a) | North America segment, which includes the United States and Canada. |
| (b) | Europe segment, which includes the European Union, the United Kingdom and certain other European countries. |
| (c) | International Markets segment, which includes all countries other than those in the North America and Europe segments. |
Year ended December 31, |
||||||||||||
2021 |
||||||||||||
North America |
Europe |
International Markets |
||||||||||
(U.S. $ in millions) |
||||||||||||
| Revenues |
$ | $ | $ | |||||||||
| Gross profit |
||||||||||||
| R&D expenses |
||||||||||||
| S&M expenses |
||||||||||||
| G&A expenses |
||||||||||||
| Other income |
( |
) | ( |
) | ( |
) | ||||||
| |
|
|
|
|
|
|||||||
| Segment profit |
$ | $ | $ | |||||||||
| |
|
|
|
|
|
|||||||
Year ended December 31, |
||||||||||||
2020 |
||||||||||||
North America |
Europe |
International Markets |
||||||||||
(U.S. $ in millions) |
||||||||||||
| Revenues |
$ | $ | $ | |||||||||
| Gross profit |
||||||||||||
| R&D expenses |
||||||||||||
| S&M expenses |
||||||||||||
| G&A expenses |
||||||||||||
| Other income |
( |
) | ( |
) | ( |
) | ||||||
| |
|
|
|
|
|
|||||||
| Segment profit |
$ | $ | $ | |||||||||
| |
|
|
|
|
|
|||||||
Year ended December 31, |
||||||||||||
2019 |
||||||||||||
North America |
Europe |
International Markets |
||||||||||
(U.S. $ in millions) |
||||||||||||
| Revenues |
$ | $ | $ | |||||||||
| Gross profit |
||||||||||||
| R&D expenses |
||||||||||||
| S&M expenses |
||||||||||||
| G&A expenses |
||||||||||||
| Other income |
( |
) | ( |
) | ( |
) | ||||||
| |
|
|
|
|
|
|||||||
| Segment profit |
$ | $ | $ | |||||||||
| |
|
|
|
|
|
|||||||
Year ended |
||||||||||||
December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
(U.S. $ in millions) |
||||||||||||
| North America profit |
$ | $ | $ | |||||||||
| Europe profit |
||||||||||||
| International Markets profit |
||||||||||||
| |
|
|
|
|
|
|||||||
| Total reportable segments profit |
||||||||||||
| Profit of other activities |
||||||||||||
| |
|
|
|
|
|
|||||||
| Total segments profit |
||||||||||||
| Amounts not allocated to segments: |
||||||||||||
| Amortization |
||||||||||||
| Other assets impairments, restructuring and other items |
||||||||||||
| Goodwill impairment |
||||||||||||
| Intangible asset impairments |
||||||||||||
| Legal settlements and loss contingencies |
||||||||||||
| Other unallocated amounts |
||||||||||||
| Consolidated operating income (loss) |
( |
) | ( |
) | ||||||||
| |
|
|
|
|
|
|||||||
| Financial expenses, net |
||||||||||||
| |
|
|
|
|
|
|||||||
| Consolidated income (loss) before income taxes |
$ | $ | ( |
) | $ | ( |
) | |||||
| |
|
|
|
|
|
|||||||
b. |
Segment revenues by major products and activities: |
Year ended December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
(U.S. $ in millions) |
||||||||||||
| Generic products |
$ | $ | $ | |||||||||
| AJOVY |
||||||||||||
| AUSTEDO |
||||||||||||
| BENDEKA/TREANDA |
||||||||||||
| COPAXONE |
||||||||||||
| ProAir* |
||||||||||||
| Anda |
||||||||||||
| Other |
||||||||||||
| |
|
|
|
|
|
|||||||
| Total |
$ | $ | $ | |||||||||
| |
|
|
|
|
|
|||||||
| * | Does not include revenues from the ProAir authorized generic, which are included under generic products. |
Year ended December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
(U.S. $ in millions) |
||||||||||||
| Generic products |
$ | $ | $ | |||||||||
| AJOVY |
||||||||||||
| COPAXONE |
||||||||||||
| Respiratory products |
||||||||||||
| Other |
||||||||||||
| |
|
|
|
|
|
|||||||
| Total |
$ | $ | $ | |||||||||
| |
|
|
|
|
|
|||||||
Year ended December 31, |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
(U.S. $ in millions) |
||||||||||||
| Generic products |
$ | $ | $ | |||||||||
| AJOVY |
§ | |||||||||||
| COPAXONE |
||||||||||||
| Other |
||||||||||||
| |
|
|
|
|
|
|||||||
| Total |
$ | $ | $ | |||||||||
| |
|
|
|
|
|
|||||||
| § | Represents an amount less than $ million . |
c. |
Supplemental data—major customers: |
Percentage of Third Party Net Sales |
||||||||||||
2021 |
2020 |
2019 |
||||||||||
| McKesson Corporation |
% | % | % | |||||||||
| AmerisourceBergen Corporation |
% | % | % | |||||||||
d. |
Property, plant and equipment—by geographical location were as follows: |
December 31, |
||||||||
2021 |
2020 |
|||||||
(U.S. $ in millions) |
||||||||
| Israel |
$ | $ | ||||||
| United States |
||||||||
| Croatia |
||||||||
| Germany |
||||||||
| Czech republic |
||||||||
| Hungary |
||||||||
| Ireland |
||||||||
| Other |
||||||||
| |
|
|
|
|||||
| Total property, plant and equipment |
$ | $ | ||||||
| |
|
|
|
|||||
December 31, 2021 |
||||||||||||||||
Level 1 |
Level 2 |
Level 3 |
Total |
|||||||||||||
(U.S. $ in millions) |
||||||||||||||||
| Cash and cash equivalents: |
||||||||||||||||
| Money markets |
$ | $ | — | $ | — | $ | ||||||||||
| Cash, deposits and other |
— | — | ||||||||||||||
| Investment in securities: |
||||||||||||||||
| Equity securities* |
— | — | ||||||||||||||
| Other |
— | |||||||||||||||
| Restricted cash |
— | — | ||||||||||||||
| Derivatives: |
||||||||||||||||
| Asset derivatives—options and forward contracts |
— | — | ||||||||||||||
| Liabilities derivatives: |
||||||||||||||||
| Options and forward contracts |
— | ( |
) | — | ( |
) | ||||||||||
| Bifurcated embedded derivatives |
— | — | § | |||||||||||||
| Contingent consideration** |
— | — | ( |
) | ( |
) | ||||||||||
| |
|
|
|
|
|
|
|
|||||||||
| Total |
$ | $ | $ | ( |
) | $ | ||||||||||
| |
|
|
|
|
|
|
|
|||||||||
| § | Represents an amount less than 0.5 million. |
December 31, 2020 |
||||||||||||||||
Level 1 |
Level 2 |
Level 3 |
Total |
|||||||||||||
(U.S. $ in millions) |
||||||||||||||||
| Cash and cash equivalents: |
||||||||||||||||
| Money markets |
$ | $ | $ | $ | ||||||||||||
| Cash, deposits and other |
||||||||||||||||
| Investment in securities: |
||||||||||||||||
| Equity securities |
||||||||||||||||
| Other, mainly debt securities |
||||||||||||||||
| Derivatives: |
||||||||||||||||
| Asset derivatives—options and forward contracts |
— | — | ||||||||||||||
| Liability derivatives—options and forward contracts |
— | ( |
) | — | ( |
) | ||||||||||
| Contingent consideration** |
( |
) | ( |
) | ||||||||||||
| |
|
|
|
|
|
|
|
|||||||||
| Total |
$ | $ | $ | ( |
) | $ | ||||||||||
| |
|
|
|
|
|
|
|
|||||||||
| * | During the first quarter of 2021, Teva’s shares in American Well Corporation (“American Well”) moved from a Level 2 measurement to a Level 1 measurement within the fair value hierarchy, since they were no longer subject to a sale restriction. By the end of September, 2021, Teva sold all of its holdings in American Well. |
| ** | Contingent consideration represents liabilities recorded at fair value in connection with acquisitions. |
December 31, 2021 |
December 31, 2020 |
|||||||
(U.S. $ in millions) |
||||||||
| Fair value at the beginning of the period |
$ | ( |
) | $ | ( |
) | ||
| Transfer into Level 3- equity securities |
||||||||
| Revaluation of equity securities |
||||||||
| Redemption of debt securities |
( |
) | ||||||
| Revaluation of debt securities |
( |
) | ||||||
| Reclassification to Level 2- equity securities |
( |
) | ||||||
| Bifurcated embedded derivatives |
§ | |||||||
| Adjustments to provisions for contingent consideration: |
||||||||
| Actavis Generics transaction |
||||||||
| Eagle transaction |
( |
) | ( |
) | ||||
| Settlement of contingent consideration: |
||||||||
| Eagle transaction |
||||||||
| |
|
|
|
|||||
| Fair value at the end of the period |
$ | ( |
) | $ | ( |
) | ||
| |
|
|
|
|||||
| § | Represents an amount less than $0.5 million. |
Estimated fair value* |
||||||||
December 31, |
||||||||
2021 |
2020 |
|||||||
(U.S. $ in millions) |
||||||||
| Senior notes and sustainability-linked senior notes included under senior notes and loans |
$ | $ | ||||||
| Senior notes and convertible senior debentures included under short-term debt |
||||||||
| |
|
|
|
|||||
| Total |
$ | $ | ||||||
| |
|
|
|
|||||
| * | The fair value was estimated based on quoted market prices. |
a. |
Long-term employee-related obligations consisted of the following: |
December 31, |
||||||||
2021 |
2020 |
|||||||
(U.S. $ in millions) |
||||||||
| Accrued severance obligations |
$ | $ | ||||||
| Defined benefit plans |
||||||||
| |
|
|
|
|||||
| Total |
$ | $ | ||||||
| |
|
|
|
|||||
b. |
Terms of arrangements: |
| Column A |
Column B |
Column C |
Column D |
Column E |
||||||||||||||||
Balance at beginning of period |
Charged to costs and expenses |
Charged to other accounts |
Deductions |
Balance at end of period |
||||||||||||||||
| Allowance for doubtful accounts: |
||||||||||||||||||||
| Year ended December 31, 2021 |
$ | $ | ( |
) | $ | $ | ( |
) |
$ | |||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| Year ended December 31, 2020 |
$ | $ | ( |
) | $ | $ | $ | |||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| Year ended December 31, 2019 |
$ | $ | ( |
) | $ | $ | ( |
) | $ | |||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| Allowance in respect of carryforward tax losses and deductions that may not be utilized: |
||||||||||||||||||||
| Year ended December 31, 2021 |
$ | $ | $ | — | $ | ( |
) | $ | ||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| Year ended December 31, 2020 |
$ | $ | $ | — | $ | ( |
) | $ | ||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
| Year ended December 31, 2019 |
$ | $ | $ | — | $ | ( |
) | $ | ||||||||||||
| |
|
|
|
|
|
|
|
|
|
|||||||||||
ITEM 9A. |
CONTROLS AND PROCEDURES |
| (a) | The following financial statements are filed as part of this Annual Report on Form 10-K: |
page |
||||
| 86 | ||||
| Consolidated Financial Statements: |
||||
| 90 | ||||
| 91 | ||||
| 92 | ||||
| 93 | ||||
| 94 | ||||
| 96 | ||||
| Financial Statement Schedule: |
||||
| 167 | ||||
| 101.DEF | Inline XBRL Taxonomy Extension Definition Linkbase Document | |
| 101.LAB | Inline XBRL Taxonomy Extension Labels Linkbase Document | |
| 101.PRE | Inline XBRL Taxonomy Extension Presentation Linkbase Document | |
| 104 | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) | |
| * | Filed herewith |
| 1. | English translation or summary from Hebrew original, which is the official version. |
| 2. | Incorporated by reference to Exhibit 3.1 to Registration Statement on Form F-1(Reg. No. 33-15736). |
| 3. | Incorporated by reference to Exhibit 3.1 to Current Report on Form 8-K filed on December 14, 2018. |
| 4. | Incorporated by reference to Exhibit 3.1 to Current Report on Form 8-K filed on June 9, 2020. |
| 5. | Incorporated by reference to Exhibit 4.1 to Current Report on Form 8-K filed on December 4, 2018. |
| 6. | Incorporated by reference to Exhibit 4.1 to Form 6-K filed on January 31, 2006. |
| 7. | Incorporated by reference to Exhibit 4.2 to Form 6-K filed on January 31, 2006. |
| 8. | Incorporated by reference to Exhibit 4.3 to Form 6-K filed on January 31, 2006. |
| 9. | Incorporated by reference to Exhibit 4.1 to Form 6-K filed on May 4, 2010. |
| 10. | Incorporated by reference to Exhibit 4.1 to Form 6-K filed on November 10, 2011. |
| 11. | Incorporated by reference to Exhibit 4.2 to Form 6-K filed on December 18, 2012. |
| 12. | Incorporated by reference to Exhibit 4.3 to Form 6-K filed on November 10, 2011. |
| 13. | Incorporated by reference to Exhibit 4.4 to Form 6-K filed on November 10, 2011. |
| 14. | Incorporated by reference to Exhibit 4.5 to Form 6-K filed on November 10, 2011. |
| 15. | Incorporated by reference to Exhibit 4.6 to Form 6-K filed on November 10, 2011. |
| 16. | Incorporated by reference to Exhibit 4.1 to Form 6-K filed on March 31, 2015. |
| 17. | Incorporated by reference to Exhibit 4.2 to Form 6-K filed on March 31, 2015. |
| 18. | Incorporated by reference to Exhibit 4.2 to Form 6-K filed on July 25, 2016. |
| 19. | Incorporated by reference to Exhibit 4.1 to Form 6-K filed on July 21, 2016. |
| 20. | Incorporated by reference to Exhibit 4.2 to Form 6-K filed on July 21, 2016. |
| 21. | Incorporated by reference to Exhibit 4.2 to Form 6-K filed on July 28, 2016. |
| 22. | Incorporated by reference to Exhibit 4.3 to Form 6-K filed on July 28, 2016. |
| 23. | Incorporated by reference to Exhibit 4.5 to Form 6-K filed on July 28, 2016. |
| 24. | Incorporated by reference to Exhibit 4.6 to Form 6-K filed on July 28, 2016. |
| 25. | Incorporated by reference to Exhibit 4.1 to Current Report on Form 8-K filed on March 14, 2018. |
| 26. | Incorporated by reference to Exhibit 4.2 to Current Report on Form 8-K filed on March 14, 2018. |
| 27. | Incorporated by reference to Exhibit 4.5 to Current Report on Form 8-K filed on March 14, 2018. |
| 28. | Incorporated by reference to Exhibit 4.6 to Current Report on Form 8-K filed on March 14, 2018. |
| 29. | Incorporated by reference to Exhibit 4.2 to Current Report on Form 8-K filed on November 25, 2019. |
| 30. | Incorporated by reference to Exhibit 4.6 to Current Report on Form 8-K filed on November 25, 2019. |
| 31. | Incorporated by reference to Exhibit 4.2 to Current Report on Form 8-K filed on November 10, 2021. |
| 32. | Incorporated by reference to Exhibit 4.6 to Current Report on Form 8-K filed on November 10, 2021. |
| 33. | Incorporated by reference to Exhibit 4.33 to Annual Report on Form 10-K filed on February 21, 2020. |
| 34. | Incorporated by reference to Exhibit 10.1 to Current Report on Form 8-K filed on April 10, 2019. |
| 35. | Incorporated by reference to Exhibit 10.20 to Annual Report on Form 10-K filed on February 12, 2018. |
| 36. | Incorporated by reference to Exhibit 10.1 to Current Report on Form 8-K filed on June 9, 2020. |
| 37. | Incorporated by reference to Exhibit 10.7 to Annual Report on Form 10-K filed on February 10, 2021. |
| 38. | Incorporated by reference to Exhibit 10.13 to Annual Report on Form 10-K filed on February 21, 2020. |
| 39. | Incorporated by reference to Exhibit 10.32 to Annual Report on Form 10-K filed on February 21, 2020. |
| 40. | Incorporated by reference to Exhibit A to Proxy Statement filed on June 8, 2017. |
| 41. | Incorporated by reference to Exhibit 10.49 to Annual Report on Form 10-K filed on February 12, 2018. |
| 42. | Incorporated by reference to Exhibit 10.50 to Annual Report on Form 10-K filed on February 12, 2018. |
| 43. | Incorporated by reference to Exhibit 10.51 to Annual Report on Form 10-K filed on February 12, 2018. |
| 44. | Incorporated by reference to Exhibit 10.52 to Annual Report on Form 10-K filed on February 12, 2018. |
| 45. | Incorporated by reference to Exhibit 10.54 to Annual Report on Form 10-K filed on February 12, 2018. |
| 46. | Incorporated by reference to Exhibit 10.60 to Annual Report on Form 10-K filed on February 12, 2018. |
| 47. | Incorporated by reference to Exhibit Appendix A to our Definitive Proxy Statement filed on April 22, 2020. |
| 48. | Incorporated by reference to Exhibit 10.63 to Annual Report on Form 10-K filed on February 12, 2018. |
| 49. | Incorporated by reference to Exhibit 10.64 to Annual Report on Form 10-K filed on February 12, 2018. |
| 50. | Incorporated by reference to Exhibit 10.2 to Quarterly Report on Form 10-Q filed on November 5, 2020. |
| 51. | Incorporated by reference to Exhibit 10.30 to Annual Report on Form 10-K filed on February 21, 2020. |
| 52. | Incorporated by reference to Exhibit 10.31 to Annual Report on Form 10-K filed on February 21, 2020. |
| 53. | Incorporated by reference to Exhibit 10.3 to Quarterly Report on Form 10-Q filed on November 5, 2020. |
| 54. | Incorporated by reference to Exhibit 10.37 to Annual Report on Form 10-K filed on February 12, 2018. |
| 55. | Incorporated by reference to Exhibit 10.38 to Annual Report on Form 10-K filed on February 12, 2018. |
| 56. | Incorporated by reference to Exhibit 18 to Quarterly Report on Form 10-Q filed on August 5, 2020. |
| TEVA PHARMACEUTICAL INDUSTRIES LIMITED | ||
| By: | /s/ Kåre Schultz | |
| Name: | Kåre Schultz | |
| Title: | President and Chief Executive Officer | |
| Dated: | February 9, 2022 | |
| Name |
Title |
Date | ||||
| By: |
/s/ Dr. Sol J. Barer Dr. Sol J. Barer |
Chairman of the Board of Directors |
February 9, 2022 | |||
| By: |
/s/ Kåre Schultz Kåre Schultz |
President and Chief Executive Officer and Director | February 9, 2022 | |||
| By: |
/s/ Eli Kalif Eli Kalif |
Executive Vice President, Chief Financial Officer (Principal Financial Officer) |
February 9, 2022 | |||
| By: |
/s/ Amir Weiss Amir Weiss |
Senior Vice President, Chief Accounting Officer (Principal Accounting Officer) |
February 9, 2022 | |||
| By: |
/s/ Rosemary A. Crane Rosemary A. Crane |
Director |
February 9, 2022 | |||
| By: |
/s/ Amir Elstein Amir Elstein |
Director |
February 9, 2022 | |||
| Name |
Title |
Date | ||||
| By: |
/s/ Jean-Michel Halfon Jean-Michel Halfon |
Director |
February 9, 2022 | |||
| By: | /s/ Gerald M. Lieberman |
Director | February 9, 2022 | |||
| Gerald M. Lieberman | ||||||
| By: |
/s/ Roberto A. Mignone Roberto A. Mignone |
Director |
February 9, 2022 | |||
| By: |
/s/ Dr. Perry D. Nisen Dr. Perry D. Nisen |
Director |
February 9, 2022 | |||
| By: |
/s/ Nechemia (Chemi) J. Peres Nechemia (Chemi) J. Peres |
Director |
February 9, 2022 | |||
| By: |
/s/ Prof. Ronit Satchi-Fainaro Prof. Ronit Satchi-Fainaro |
Director |
February 9, 2022 | |||
| By: |
/s/ Janet S. Vergis Janet S. Vergis |
Director |
February 9, 2022 | |||
| By: |
/s/ Dr. Tal Zaks Dr. Tal Zaks |
Director |
February 9, 2022 | |||

