Exhibit 99.1

FOR IMMEDIATE RELEASE
AMAG Pharmaceuticals Reports Third Quarter 2015 Financial Results
Achieves non-GAAP quarterly revenue of $103.5 million and adjusted EBITDA of $52.8 million
Provides new updates on Makena next generation development programs
Closes acquisition and initiates integration of Cord Blood Registry
Conference call scheduled for 8:00 a.m. ET today
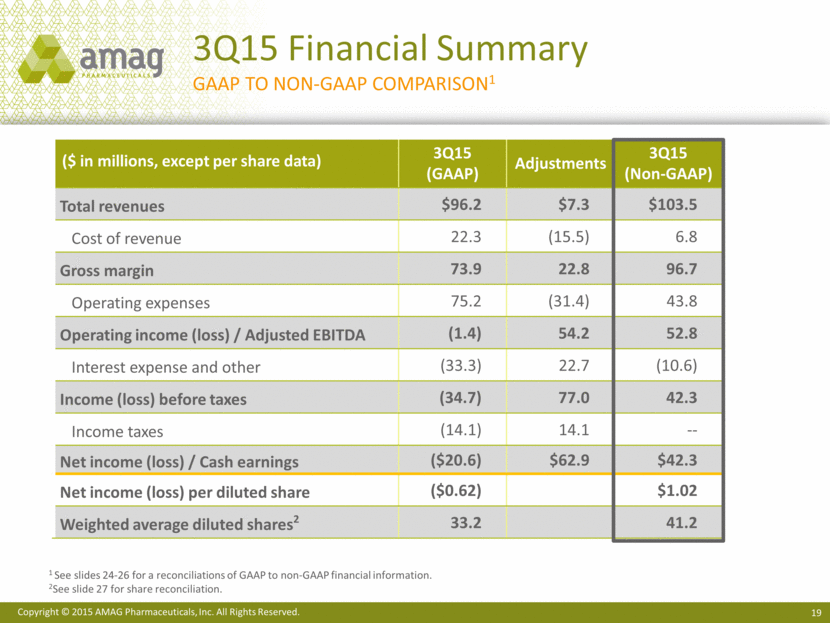
WALTHAM, MA (November 3, 2015) — AMAG Pharmaceuticals, Inc. (NASDAQ: AMAG) today reported unaudited consolidated financial results for the third quarter and year-to-date periods ended September 30, 2015. Total revenues for the third quarter of 2015 increased to $96.2 million, compared with $25.5 million in the third quarter of 2014. This increase is primarily related to the additions of Makena® (hydroxyprogesterone caproate injection) in November 2014 and Cord Blood Registry (CBR) in August 2015 to the company’s portfolio, which contributed revenue of $65.2 million and $7.2 million, respectively, to the third quarter 2015 results. After certain non-cash and one-time costs related to business development activities and acquisitions, the company reported an operating loss of $1.4 million in the third quarter of 2015, compared with operating income of $4.3 million in the same period last year. This resulted in a net loss of $20.6 million, or $0.62 per basic and diluted share, compared with net income of $1.5 million, or $0.07 per basic share and $0.06 per diluted share for the same period last year.
Non-GAAP revenue totaled $103.5 million in the third quarter of 2015, compared with $23.5 million for the same period last year.(1) The company reported adjusted EBITDA of $52.8 million in the third quarter of 2015, compared with adjusted EBITDA of $1.3 million in the third quarter of 2014.(1) Non-GAAP cash earnings for the third quarter of 2015 totaled $42.3 million, or $1.02 per diluted share, compared with $0.3 million, or $0.01 per diluted share, for the same period in 2014.(1),(2)
“While our overall portfolio experienced growth during the third quarter, we believe there is opportunity for even better performance in the future as integration activities are completed and our investments in the next generation development programs for Makena and an expanded label for Feraheme progress toward FDA approvals and launches,” said William Heiden, AMAG’s chief executive officer. “With our business continuing to generate strong cash flows, we will maintain our focus on executing on the opportunities we have today, line extension approvals in the future, as well as continued portfolio expansion through acquisitions and licensing transactions.”
(1) See summaries of non-GAAP adjustments to reconcile GAAP Consolidated Statements of Operations to Non-GAAP Consolidated Statements of Operations for the three and nine months ended September 30, 2015 and 2014 at the conclusion of this press release.
(2) See share count reconciliation at the conclusion of this press release.
|
November 3, 2015 |
AMAG Pharmaceuticals, Inc. |
3Q15 Business Highlights and Recent Developments
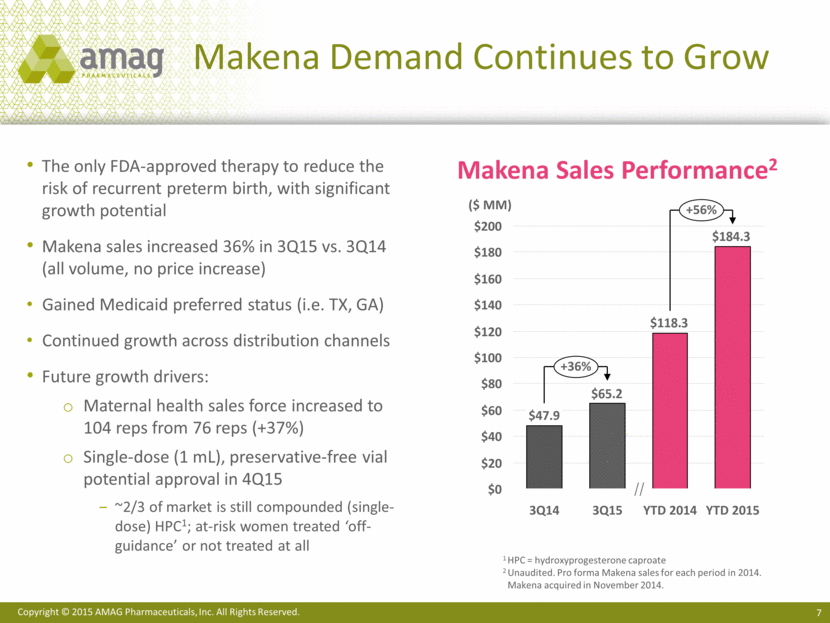
· Makena net sales totaled $65.2 million in the third quarter of 2015, representing 36% growth over the corresponding period in the prior year.(3) The growth in sales was driven by a 42% increase in volume partially offset by a decrease in net revenue per injection due to higher growth in the Medicaid versus commercial segment.
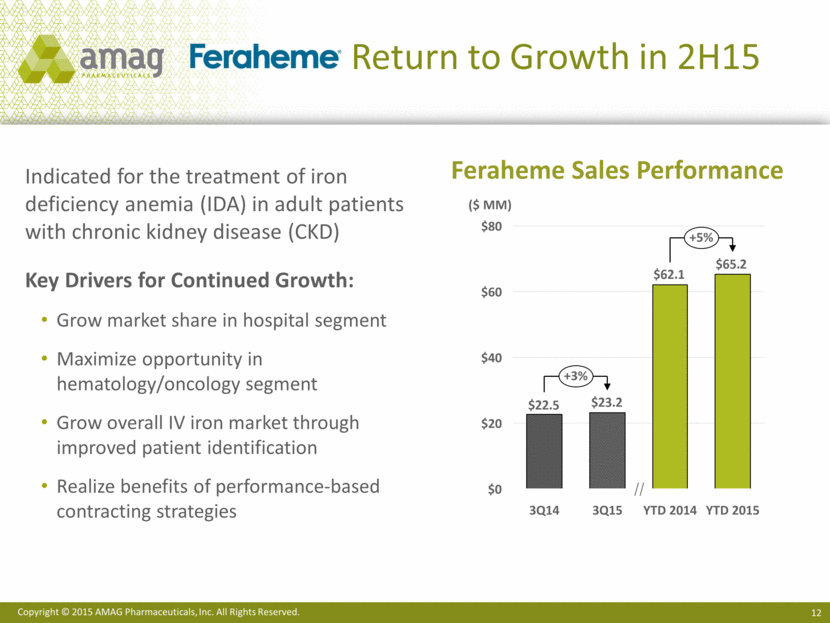
· Sales of Feraheme® (ferumoxytol) injection returned to growth, increasing 3% to $23.2 million in the third quarter of 2015, compared with $22.5 million in the same period last year. Sales of MuGard grew 16% in the third quarter of 2015 over the same period last year.
· The company closed on the acquisition and financing of CBR, the world’s largest stem cell collection and storage company, on August 17, 2015. The CBR business recorded $14.5 million of non-GAAP revenue for the six-week period between closing and September 30, 2015.(1) On a pro forma basis (as if AMAG had acquired CBR at the beginning of the third quarter of 2015), total CBR revenue was $29.3 million, compared with $33.0 million in the third quarter of 2014, with the decrease being partially attributable to new customer discounts in the third quarter of 2015.(4)
· During the third quarter of 2015, the company announced plans to further expand its growing maternal health portfolio by entering into an exclusive option agreement with Velo Bio, LLC, a privately held life sciences company. The agreement gives the company the option to acquire the global rights to an orphan drug candidate in clinical development for use in the treatment of severe preeclampsia, an area of high unmet need in pregnant women.
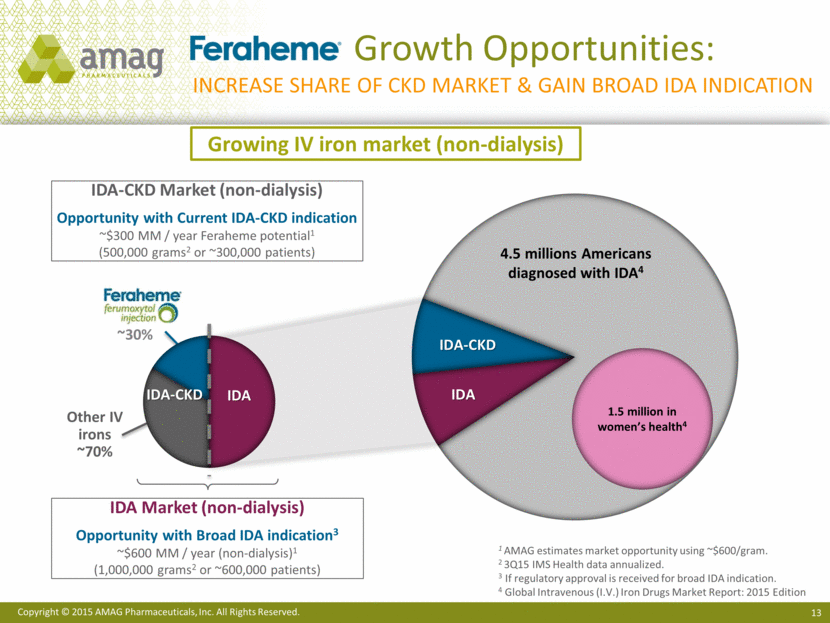
· The Company recently announced start-up activities for a head-to-head, Phase 3 clinical trial evaluating Feraheme in adult patients with iron deficiency anemia (IDA). The study marks a step forward on the pathway to potentially broaden the use of Feraheme beyond the current chronic kidney disease (CKD) indication to include all adult IDA patients who have failed or cannot tolerate oral iron treatment. The company expects to begin enrolling patients in the trial in the first quarter of 2016, with potential approval of a supplemental new drug application for a broader IDA indication and launch in 2018.
· The company advanced its next generation development programs for Makena seeking to enhance the product for patients and their healthcare providers. As previously disclosed, the company has filed for approval of a single-dose, preservative-free version of Makena, with anticipated approval in the fourth quarter of 2015. In addition, the company is developing a device for subcutaneous administration of Makena by an auto-injector. The company has an exclusive agreement in place with a device partner whose technologies are covered by multiple issued patents, and has other approved products in its portfolio. AMAG is also developing a longer-acting formulation of Makena.
Third Quarter Ended September 30, 2015 (unaudited)
Financial Results (GAAP Basis)
Total revenues for the third quarter of 2015 were $96.2 million, compared with $25.5 million for the same period in 2014. Net product sales of Makena, Feraheme and MuGard totaled $88.9 million in the third quarter of
(3) Based on unaudited pro forma sales for the third quarter of 2014. Acquisition of Lumara Health closed in November 2014.
(4) Based on unaudited pro forma sales for the third quarter of 2015 and 2014. Acquisition of CBR closed on August 17, 2015.
2015, compared with $23.0 million in the third quarter of 2014. Service revenue from CBR totaled $7.2 million in the third quarter of 2015.
Total costs and expenses for the third quarter of 2015 were $97.5 million, compared with $21.2 million for the same period in 2014. The increases in costs and expenses were primarily due to the following: (i) higher costs associated with managing the company’s expanded portfolio and infrastructure following the acquisitions of Lumara Health in November 2014 and CBR in August 2015, (ii) increased cost of goods sold from new product and higher sales volumes for each of the company’s products, (iii) non-cash charges associated with inventory step-up and amortization related to the acquisitions, (iv) a $10.0 million upfront payment for the exclusive option to acquire rights to a development stage asset to treat severe preeclampsia, and (v) $9.2 million in acquisition and restructuring costs related to CBR.
The company reported an operating loss of $1.4 million and a net loss of $20.6 million, or $0.62 per basic and diluted share, for the third quarter of 2015, compared with operating income of $4.3 million and net income of $1.5 million, or $0.07 per basic share and $0.06 per diluted share, for the same period in 2014.
Financial Results (Non-GAAP Basis)(1),(2)
Non-GAAP revenues totaled $103.5 million, up from $23.5 million in the third quarter of 2014. Non-GAAP CBR revenue totaled $14.5 million in the third quarter of 2015. The difference between GAAP and non-GAAP revenue for CBR represents purchase accounting adjustments related to deferred revenue.
Total costs and expenses on a non-GAAP basis totaled $50.6 million resulting in a gross margin of 93% and adjusted EBITDA margin of 51% for the third quarter of 2015. This compares to costs and expenses of $22.3 million in the same period of 2014, which resulted in a gross margin of 90% and adjusted EBITDA margin of 5%. Non-GAAP adjusted EBITDA for the third quarter of 2015 was $52.8 million, compared with $1.3 million for the same period in 2014.
After deducting cash interest expense, the company generated third quarter non-GAAP cash earnings of $42.3 million, or $1.27 per non-GAAP basic share and $1.02 per non-GAAP diluted share. The weighted average diluted shares used in calculating the non-GAAP cash earnings per diluted share for the third quarter of 2015 includes the impact of the convertible debt and related bond hedge and warrants.
Balance Sheet Highlights
As of September 30, 2015, the company’s cash and investments totaled approximately $442.9 million and total debt (face value) was $1.05 billion.
“The two transformative acquisitions that we completed in the past twelve months have allowed us to significantly grow our top line while delivering strong cash flows and profitability for our shareholders,” said Frank Thomas, AMAG’s president and chief operating officer. “As we continue to integrate the businesses, we are anticipating even better financial and operating performance, allowing us to serve our patients more effectively and efficiently.”
Nine Months Ended September 30, 2015 (unaudited)
Financial Results (GAAP Basis)
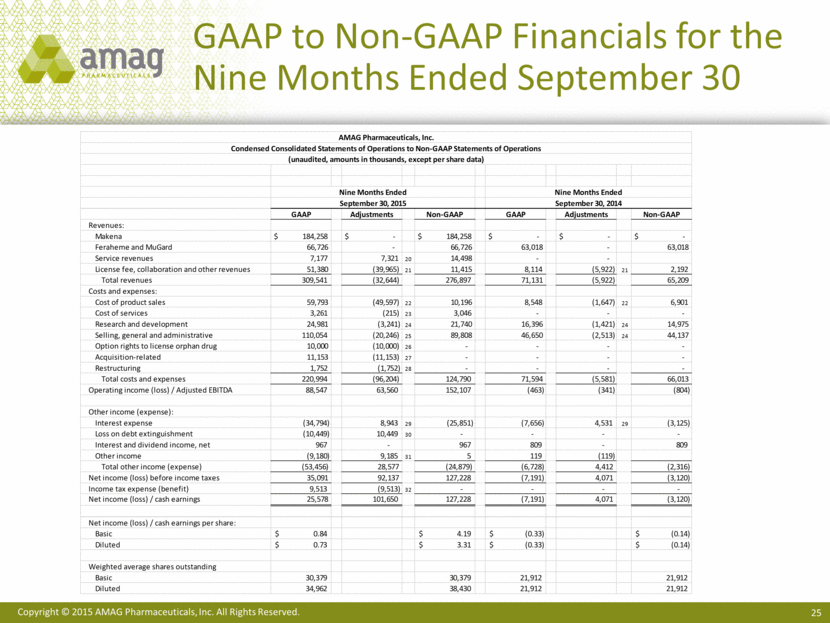
Total revenues for the nine months ended September 30, 2015 were $309.5 million, compared with $71.1 million for the same period in 2014. This increase is primarily related to the addition of Makena in November 2014 and CBR in August 2015, which contributed $184.3 million and $7.2 million, respectively, in product revenue to the year-to-date 2015 results. In addition, the company recognized $39.2 million of collaboration revenue in 2015 related to the termination of the company’s ex-US ferumoxytol marketing agreement with Takeda Pharmaceutical Company Limited.
Net income for the first nine months of 2015 totaled $25.6 million, compared with a net loss of $7.2 million for same period in 2014. Basic net income per share was $0.84, compared with a net loss per basic share of $0.33 in 2014. Diluted net income per share was $0.73 in 2015, compared with a net loss per diluted share of $0.33 in 2014. The weighted average diluted shares used in calculating diluted net income per share for the first nine months of 2015 followed the if-converted method of accounting for the convertible debt.
Financial Results (Non-GAAP Basis)(1),(2)
Non-GAAP adjusted EBITDA for the nine months ended September 30, 2015 was $152.1 million, compared with a loss before interest, taxes, depreciation and amortization of $0.8 million for the same period in 2014. After deducting cash interest expense, the company generated non-GAAP cash earnings of $127.2 million, or $3.31 per non-GAAP diluted share.
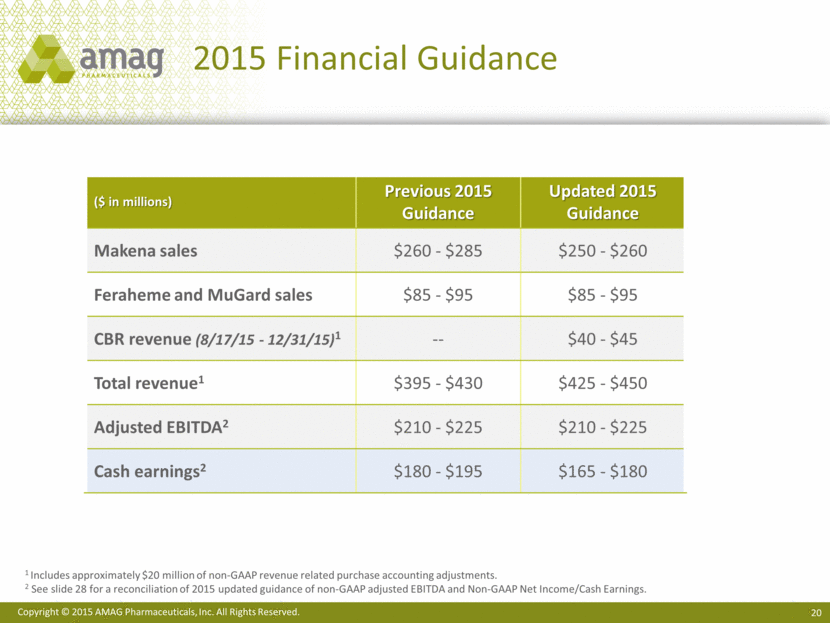
Updating 2015 Financial Outlook(6)
The company is updating its full year 2015 guidance to include revenue from CBR and reflect the business performance for the first nine months and the outlook for the remainder of 2015.
|
$ in millions |
|
Previous 2015 Guidance |
|
Updated 2015 Guidance |
|
|
Makena sales |
|
$260 - $285 |
|
$250 - $260 |
|
|
Feraheme and MuGard sales |
|
$85 - $95 |
|
$85 - $95 |
|
|
CBR revenue (8/17/15-12/31/15)(5) |
|
— |
|
$40 - $45 |
|
|
Total revenue(5) |
|
$395 - $430 |
|
$425 - $450 |
|
|
Adjusted EBITDA (6) |
|
$210 - $225 |
|
$210 - $225 |
|
|
Cash earnings(6) |
|
$180 - $195 |
|
$165 - $180 |
|
Conference Call and Webcast Access
AMAG Pharmaceuticals, Inc. will host a conference call and webcast with slides today at 8:00 a.m. ET, during which management will discuss the company’s financial and operating results and recent developments. To access the conference call via telephone, please dial (877) 412-6083 from the United States or (702) 495-1202 for international access. A telephone replay will be available from approximately 11:00 a.m. ET on November 3, 2015 through midnight on November 10, 2015. To access a replay of the conference call, dial (855) 859-2056 from the United States or (404) 537-3406 for international access. The pass code for the live call and the replay is 59521553.
(5) Includes approximately $20 million of non-GAAP revenue related purchase accounting adjustments.
(6) See Reconciliation of 2015 updated guidance of non-GAAP adjusted EBITDA and Non-GAAP Net Income/Cash Earnings at the conclusion of this press release.
The call will be webcast with slides and accessible through the Investors section of the company’s website at www.amagpharma.com. The webcast replay will be available from approximately 11:00 a.m. ET on November 3, 2015 through midnight on December 3, 2015.
Use of Non-GAAP Financial Measures
AMAG has presented certain non-GAAP financial measures, including non-GAAP revenue, non-GAAP adjusted EBITDA (earnings before income taxes, depreciation and amortization), non-GAAP net income, or cash earnings, and non-GAAP diluted net income, or cash earnings, per share. These non-GAAP financial measures exclude certain amounts, revenue, expenses or income, from the corresponding financial measures determined in accordance with accounting principles generally accepted in the U.S. (GAAP). Management believes this non-GAAP information is useful for investors, taken in conjunction with AMAG’s GAAP financial statements, because it provides greater transparency regarding AMAG’s operating performance. Management uses these measures, among other factors, to assess and analyze operational results and trends and to make financial and operational decisions. Non-GAAP information is not prepared under a comprehensive set of accounting rules and should only be used to supplement an understanding of AMAG’s operating results as reported under GAAP, not as a substitute for GAAP. In addition, these non-GAAP financial measures are unlikely to be comparable with non-GAAP information provided by other companies. The determination of the amounts that are excluded from non-GAAP financial measures is a matter of management judgment and depends upon, among other factors, the nature of the underlying expense or income amounts. Reconciliations between these non-GAAP financial measures and the most comparable GAAP financial measures are included in the tables accompanying this press release after the unaudited condensed consolidated financial statements.
About AMAG
AMAG Pharmaceuticals, Inc. uses its business and clinical expertise to bring medical therapies and other innovations to market that provide clear benefits and improve people’s lives. Based in Waltham, MA, AMAG has a diverse portfolio of products in the areas of maternal health, anemia management and cancer supportive care. AMAG continues to work to expand the impact of these and future products for patients by delivering on its growth strategy, which includes organic growth, as well as the pursuit of products and companies that align with AMAG’s existing therapeutic areas or those that could benefit from its proven core competencies. For additional company information, please visit www.amagpharma.com.
About Makena® (hydroxyprogesterone caproate injection)
Makena® is a progestin indicated to reduce the risk of preterm birth in women with a singleton pregnancy who have a history of singleton spontaneous preterm birth.
The effectiveness of Makena is based on improvement in the proportion of women who delivered <37 weeks of gestation. There are no controlled trials demonstrating a direct clinical benefit, such as improvement in neonatal mortality and morbidity.
Limitation of use: While there are many risk factors for preterm birth, safety and efficacy of Makena has been demonstrated only in women with a prior spontaneous singleton preterm birth. It is not intended for use in women with multiple gestations or other risk factors for preterm birth.
Makena should not be used in women with any of the following conditions: blood clots or other blood clotting problems, breast cancer or other hormone-sensitive cancers, or history of these conditions; unusual vaginal bleeding not related to the current pregnancy, yellowing of the skin due to liver problems during pregnancy, liver problems, including liver tumors, or uncontrolled high blood pressure.
Before patients receive Makena, they should tell their healthcare provider if they have an allergy to hydroxyprogesterone caproate, castor oil, or any of the other ingredients in Makena; diabetes or prediabetes, epilepsy, migraine headaches, asthma, heart problems, kidney problems, depression, or high blood pressure.
In one clinical study, certain complications or events associated with pregnancy occurred more often in women who received Makena. These included miscarriage (pregnancy loss before 20 weeks of pregnancy), stillbirth (fetal death occurring during or after the 20th week of pregnancy), hospital admission for preterm labor, preeclampsia (high blood pressure and too much protein in the urine), gestational hypertension (high blood pressure caused by pregnancy), gestational diabetes, and oligohydramnios (low amniotic fluid levels).
Makena may cause serious side effects including blood clots, allergic reactions, depression, and yellowing of the skin and the whites of the eyes. The most common side effects of Makena include injection site reactions (pain, swelling, itching, bruising, or a hard bump), hives, itching, nausea, and diarrhea.
For additional U.S. product information, including full prescribing information, please visit www.makena.com.
About Feraheme® (ferumoxytol) Injection
Feraheme received marketing approval from the FDA on June 30, 2009 for the treatment of IDA in adult CKD patients and was commercially launched by AMAG in the U.S. shortly thereafter. Ferumoxytol is protected in the U.S. by six issued patents covering the composition and dosage form of the product. Each issued patent is listed in the FDA’s Orange Book, the last of which expires in June 2023.
Fatal and serious hypersensitivity reactions including anaphylaxis have occurred in patients receiving Feraheme. Initial symptoms may include hypotension, syncope, unresponsiveness, cardiac/cardiorespiratory arrest. Feraheme is contraindicated in patients with a known hypersensitivity to Feraheme or any of its components, or a history of allergic reaction to any intravenous iron product.
For additional U.S. product information, please see full Prescribing Information, including Boxed Warning, available at www.feraheme.com.
About Cord Blood Registry (CBR)
CBR is the world’s largest newborn stem cell company. Founded in 1992, CBR is entrusted by parents with storing more than 600,000 cord blood and cord tissue units. CBR is dedicated to advancing the clinical application of newborn stem cells by partnering with leading research institutions to establish conduct FDA-regulated clinical trials for conditions that have no cure today. For more information, visit www.cordblood.com.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 (PSLRA) and other federal securities laws. Any statements contained herein which do not
describe historical facts, including, among others, statements regarding beliefs about AMAG being well-positioned for continued growth; AMAG’s ability to service patients and improve lives; AMAG’s capacity to leverage newly acquired capabilities; plans to expand AMAG’s growing maternal health portfolio; expectations for AMAG’s Phase 3 clinical trial for the broader indication for Feraheme; expectations for AMAG’s lifecycle management program for Makena, including the development and advantages of a subcutaneous auto-injector device and a longer-acting formulation of Makena (including the timing and clinical trial efforts in connection with such activities), as well as the potential approval of a single-dose, preservative free version of Makena; opportunity for improved performance in light of integration activities and lifecycle management investments; AMAG’s ability to generate strong cash flows; expectations regarding line extension approvals and portfolio expansion efforts, and AMAG’s 2015 financial outlook, are forward-looking statements which involve risks and uncertainties that could cause actual results to differ materially from those discussed in such forward-looking statements.
Such risks and uncertainties include, among others, AMAG’s ability to realize the expected benefits and synergies from the CBR acquisition and the success of the CBR services as part of AMAG’s portfolio; AMAG’s inexperience with cord blood and stem cell services, including the attendant regulatory regime and increased legal and compliance risks; the ethical, legal, regulatory, and social implications of stem cell research; AMAG’s ability to successfully commercialize Makena, including if a generic version of hydroxyprogesterone caproate is introduced to the market or if AMAG is unable to otherwise maintain the benefits of Makena’s orphan drug exclusivity; the possibility that AMAG’s lifecycle management program for Makena will not be successful on expected timelines or at all, and that pursuit of the subcutaneous auto-injector device and longer-acting formulation of Makena will absorb significant resources, financial and otherwise, over a considerable period of time, and despite such efforts, might not be deemed adequate by the FDA and may not extend Makena’s commercial viability beyond February 2018; the possibility that AMAG’s level of indebtedness could restrict operations and limit its ability to plan for or respond to changes in its business or acquire additional products for its portfolio, as well as those risks identified in AMAG’s filings with the U.S. Securities and Exchange Commission (SEC), including its Annual Report on Form 10-K for the year ended December 31, 2014, its Quarterly Report on Form 10-Q for the quarter ended June 30, 2015 and subsequent filings with the SEC. Any such risks and uncertainties could materially and adversely affect AMAG’s results of operations, its profitability and its cash flows, which would, in turn, have a significant and adverse impact on AMAG’s stock price. AMAG cautions you not to place undue reliance on any forward-looking statements, which speak only as of the date they are made.
AMAG disclaims any obligation to publicly update or revise any such statements to reflect any change in expectations or in events, conditions or circumstances on which any such statements may be based, or that may affect the likelihood that actual results will differ from those set forth in the forward-looking statements.
AMAG Pharmaceuticals® and Feraheme® are registered trademark of AMAG Pharmaceuticals, Inc. MuGard® is a registered trademark of Abeona Therapeutics, Inc. (formerly known as PlasmaTech Biopharmaceuticals, Inc. and Access Pharmaceuticals, Inc.). Makena® is a registered trademark of Lumara Health Inc. Lumara HealthTM is a registered trademark of Lumara Health. Cord Blood Registry® and CBR® are registered trademarks of CBR Systems, Inc.
AMAG Pharmaceuticals, Inc.
Condensed Consolidated Statements of Operations
(unaudited, amounts in thousands, except for per share data)
|
|
|
Three Months Ended |
|
Nine Months Ended |
| ||||||||
|
|
|
2015 |
|
2014 |
|
2015 |
|
2014 |
| ||||
|
Revenues: |
|
|
|
|
|
|
|
|
| ||||
|
Makena |
|
$ |
65,155 |
|
$ |
— |
|
$ |
184,258 |
|
$ |
— |
|
|
Feraheme/MuGard |
|
23,762 |
|
23,009 |
|
66,726 |
|
63,017 |
| ||||
|
Cord Blood Registry |
|
7,177 |
|
— |
|
7,177 |
|
— |
| ||||
|
License fee, collaboration and other |
|
58 |
|
2,485 |
|
51,380 |
|
8,114 |
| ||||
|
Total revenues |
|
96,152 |
|
25,494 |
|
309,541 |
|
71,131 |
| ||||
|
Operating costs and expenses: |
|
|
|
|
|
|
|
|
| ||||
|
Cost of product sales |
|
19,088 |
|
2,968 |
|
59,793 |
|
8,548 |
| ||||
|
Cost of services |
|
3,261 |
|
— |
|
3,261 |
|
|
| ||||
|
Research and development |
|
9,809 |
|
5,358 |
|
24,981 |
|
16,396 |
| ||||
|
Selling, general and administrative |
|
46,141 |
|
12,875 |
|
110,054 |
|
46,650 |
| ||||
|
Option rights to license orphan drug |
|
10,000 |
|
— |
|
10,000 |
|
— |
| ||||
|
Acquisition-related |
|
8,500 |
|
— |
|
11,153 |
|
— |
| ||||
|
Restructuring |
|
738 |
|
— |
|
1,752 |
|
— |
| ||||
|
Total costs and expenses |
|
97,537 |
|
21,201 |
|
220,994 |
|
71,594 |
| ||||
|
Operating income (loss) |
|
(1,385 |
) |
4,293 |
|
88,547 |
|
(463 |
) | ||||
|
|
|
|
|
|
|
|
|
|
| ||||
|
Other income (expense): |
|
|
|
|
|
|
|
|
| ||||
|
Interest expense |
|
(14,222 |
) |
(3,129 |
) |
(34,794 |
) |
(7,656 |
) | ||||
|
Loss on debt extinguishment |
|
(10,449 |
) |
— |
|
(10,449 |
) |
— |
| ||||
|
Interest and dividend income, net |
|
524 |
|
291 |
|
967 |
|
809 |
| ||||
|
Other income, net |
|
(9,182 |
) |
3 |
|
(9,180 |
) |
119 |
| ||||
|
Total other income (expense) |
|
(33,329 |
) |
(2,835 |
) |
(53,456 |
) |
(6,728 |
) | ||||
|
Net income (loss) before income taxes |
|
(34,714 |
) |
1,458 |
|
35,091 |
|
(7,191 |
) | ||||
|
Income tax expense (benefit) |
|
(14,130 |
) |
— |
|
9,513 |
|
— |
| ||||
|
Net income (loss) / cash earnings |
|
$ |
(20,584 |
) |
$ |
1,458 |
|
$ |
25,578 |
|
$ |
(7,191 |
) |
|
|
|
|
|
|
|
|
|
|
| ||||
|
Net income (loss) per share |
|
|
|
|
|
|
|
|
| ||||
|
Basic |
|
$ |
(0.62 |
) |
$ |
0.07 |
|
$ |
0.84 |
|
$ |
(0.33 |
) |
|
Diluted |
|
$ |
(0.62 |
) |
$ |
0.06 |
|
$ |
0.73 |
|
$ |
(0.33 |
) |
|
|
|
|
|
|
|
|
|
|
| ||||
|
Weighted average shares outstanding used to compute net income (loss) per share: |
|
|
|
|
|
|
|
|
| ||||
|
Basic |
|
33,223 |
|
21,984 |
|
30,379 |
|
21,912 |
| ||||
|
Diluted |
|
33,223 |
|
23,467 |
|
34,962 |
|
21,912 |
| ||||
AMAG Pharmaceuticals, Inc.
Condensed Consolidated Balance Sheets
(unaudited, amounts in thousands)
|
|
|
September 30, 2015 |
|
December 31, 2014 |
| ||
|
Assets: |
|
|
|
|
| ||
|
Cash and cash equivalents |
|
$ |
167,530 |
|
$ |
119,296 |
|
|
Investments |
|
275,343 |
|
24,890 |
| ||
|
Accounts receivable, net |
|
73,689 |
|
38,172 |
| ||
|
Inventories |
|
37,217 |
|
40,610 |
| ||
|
Receivable from collaboration |
|
— |
|
4,518 |
| ||
|
Deferred tax assets |
|
53,164 |
|
32,094 |
| ||
|
Prepaid and other current assets |
|
17,725 |
|
14,456 |
| ||
|
Restricted cash |
|
30,755 |
|
— |
| ||
|
Total current assets |
|
655,423 |
|
274,036 |
| ||
|
Property, plant and equipment, net |
|
30,357 |
|
1,519 |
| ||
|
Goodwill |
|
637,400 |
|
205,824 |
| ||
|
Intangible assets, net |
|
1,211,547 |
|
887,908 |
| ||
|
Restricted cash |
|
2,397 |
|
2,397 |
| ||
|
Other long-term assets |
|
17,289 |
|
17,249 |
| ||
|
Total assets |
|
$ |
2,554,413 |
|
$ |
1,388,933 |
|
|
|
|
|
|
|
| ||
|
Liabilities and stockholders’ equity: |
|
|
|
|
| ||
|
Accounts payable |
|
$ |
3,661 |
|
$ |
7,301 |
|
|
Accrued expenses |
|
102,656 |
|
80,093 |
| ||
|
Payable to former CBR shareholders |
|
36,259 |
|
— |
| ||
|
Current portion of long-term debt |
|
17,500 |
|
34,000 |
| ||
|
Current portion of contingent consideration |
|
96,756 |
|
718 |
| ||
|
Deferred revenues |
|
10,012 |
|
44,376 |
| ||
|
Total current liabilities |
|
266,844 |
|
166,488 |
| ||
|
Long-term debt, net |
|
815,067 |
|
293,905 |
| ||
|
Convertible 2.5% senior notes, net |
|
172,594 |
|
167,441 |
| ||
|
Acquisition-related contingent consideration |
|
126,189 |
|
217,984 |
| ||
|
Deferred income tax liabilities |
|
247,615 |
|
77,619 |
| ||
|
Deferred revenues |
|
1,889 |
|
— |
| ||
|
Other long-term liabilities |
|
4,254 |
|
5,543 |
| ||
|
Total liabilities |
|
1,634,452 |
|
928,980 |
| ||
|
Total stockholders’ equity |
|
919,961 |
|
459,953 |
| ||
|
Total liabilities and stockholders’ equity |
|
$ |
2,554,413 |
|
$ |
1,388,933 |
|
AMAG Pharmaceuticals, Inc.
Condensed Consolidated Statements of Operations to Non-GAAP Statements of Operations
(unaudited, amounts in thousands, except per share data)
|
|
|
Three Months Ended |
|
Three Months Ended |
| ||||||||||||||||||
|
|
|
September 30, 2015 |
|
September 30, 2014 |
| ||||||||||||||||||
|
|
|
GAAP |
|
Adjustments |
|
|
|
Non-GAAP |
|
GAAP |
|
Adjustments |
|
|
|
Non-GAAP |
| ||||||
|
Revenues: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
|
Makena |
|
$ |
65,155 |
|
$ |
— |
|
|
|
$ |
65,155 |
|
$ |
— |
|
$ |
— |
|
|
|
$ |
— |
|
|
Feraheme/MuGard |
|
23,762 |
|
— |
|
|
|
23,762 |
|
23,009 |
|
— |
|
|
|
23,009 |
| ||||||
|
Cord Blood Registry |
|
7,177 |
|
7,321 |
|
(7) |
|
14,498 |
|
— |
|
— |
|
|
|
— |
| ||||||
|
License fee, collaboration and other |
|
58 |
|
— |
|
|
|
58 |
|
2,485 |
|
(1,974 |
) |
(8) |
|
511 |
| ||||||
|
Total revenues |
|
96,152 |
|
7,321 |
|
|
|
103,473 |
|
25,494 |
|
(1,974 |
) |
|
|
23,520 |
| ||||||
|
Operating costs and expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
|
Cost of products sold |
|
19,088 |
|
(15,336 |
) |
(9) |
|
3,752 |
|
2,968 |
|
(554 |
) |
(9) |
|
2,414 |
| ||||||
|
Cost of services |
|
3,261 |
|
(215 |
) |
(10) |
|
3,046 |
|
— |
|
— |
|
|
|
— |
| ||||||
|
Research and development |
|
9,809 |
|
(1,550 |
) |
(11) |
|
8,259 |
|
5,358 |
|
(579 |
) |
(11) |
|
4,779 |
| ||||||
|
Selling, general and administrative |
|
46,141 |
|
(10,561 |
) |
(12) |
|
35,580 |
|
12,875 |
|
2,183 |
|
(12) |
|
15,058 |
| ||||||
|
Option rights to license orphan drug |
|
10,000 |
|
(10,000 |
) |
(13) |
|
— |
|
— |
|
— |
|
|
|
— |
| ||||||
|
Acquisition-related |
|
8,500 |
|
(8,500 |
) |
(14) |
|
— |
|
— |
|
— |
|
|
|
— |
| ||||||
|
Restructuring |
|
738 |
|
(738 |
) |
(15) |
|
— |
|
— |
|
— |
|
|
|
— |
| ||||||
|
Total costs and expenses |
|
97,537 |
|
(46,900 |
) |
|
|
50,637 |
|
21,201 |
|
1,050 |
|
|
|
22,251 |
| ||||||
|
Operating income (loss) / adjusted EBITDA |
|
(1,385 |
) |
54,221 |
|
|
|
52,836 |
|
4,293 |
|
(3,024 |
) |
|
|
1,269 |
| ||||||
|
Other income (expense): |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
|
Interest expense |
|
(14,222 |
) |
3,111 |
|
(16) |
|
(11,111 |
) |
(3,129 |
) |
1,879 |
|
(16) |
|
(1,250 |
) | ||||||
|
Loss on debt extinguishment |
|
(10,449 |
) |
10,449 |
|
(17) |
|
— |
|
— |
|
— |
|
|
|
— |
| ||||||
|
Interest and dividend income, net |
|
524 |
|
— |
|
|
|
524 |
|
291 |
|
— |
|
|
|
291 |
| ||||||
|
Other income, net |
|
(9,182 |
) |
9,187 |
|
(18) |
|
5 |
|
3 |
|
(3 |
) |
|
|
— |
| ||||||
|
Total other income (expense) |
|
(33,329 |
) |
22,747 |
|
|
|
(10,582 |
) |
(2,835 |
) |
1,876 |
|
|
|
(959 |
) | ||||||
|
Net income (loss) before income taxes |
|
(34,714 |
) |
76,968 |
|
|
|
42,254 |
|
1,458 |
|
(1,148 |
) |
|
|
310 |
| ||||||
|
Income tax expense (benefit) |
|
(14,130 |
) |
14,130 |
|
(19) |
|
— |
|
— |
|
— |
|
|
|
— |
| ||||||
|
Net income (loss) / cash earnings |
|
$ |
(20,584 |
) |
$ |
62,838 |
|
|
|
$ |
42,254 |
|
$ |
1,458 |
|
$ |
(1,148 |
) |
|
|
$ |
310 |
|
|
Net income (loss) / cash earnings per share |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
|
Basic |
|
$ |
(0.62 |
) |
|
|
|
|
$ |
1.27 |
|
$ |
0.07 |
|
|
|
|
|
$ |
0.01 |
| ||
|
Diluted |
|
$ |
(0.62 |
) |
|
|
|
|
$ |
1.02 |
|
$ |
0.06 |
|
|
|
|
|
$ |
0.01 |
| ||
|
Weighted average shares outstanding |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
|
Basic |
|
33,223 |
|
|
|
|
|
33,223 |
|
21,984 |
|
|
|
|
|
21,984 |
| ||||||
|
Diluted |
|
33,223 |
|
|
|
|
|
41,229 |
|
23,467 |
|
|
|
|
|
23,098 |
| ||||||
|
(7) |
Adding back period write-down of deferred revenue from purchase accounting. |
|
(8) |
Eliminate non-cash revenue related to recognition of previously deferred revenue on Takeda agreement. |
|
(9) |
Eliminate the following: (i) non-cash step-up of inventory from purchase accounting (2015); (ii) amortization expense related to intangible assets; (iii) depreciation expense; and (iv) stock-based compensation expense. |
|
(10) |
Eliminate the following: (i) depreciation expense; and (ii) stock-based compensation expense. |
|
(11) |
Eliminate the following: (i) non-cash step-up of inventory from purchase accounting (2015); (ii) depreciation expense; and (iii) stock-based compensation expense. |
|
(12) |
Eliminate the following: (i) non-cash adjustments to contingent consideration; (ii) amortization expense related to intangible assets; (iii) certain transaction-related expenses (2015); (iv) depreciation expense; and (v) stock-based compensation expense. |
|
(13) |
Eliminate one-time costs related to Velo option. |
|
(14) |
Eliminate one-time acquisition costs related to Cord Blood Registry. |
|
(15) |
Eliminate one-time restructuring costs related to Cord Blood Registry acquisition. |
|
(16) |
Eliminate non-cash interest expense; amortization of debt discount and other costs. |
|
(17) |
Eliminate non-cash or one-time expenses related to the August 2015 term loan refinancing. |
|
(18) |
Eliminate one-time expenses related to the August 2015 debt financing. |
|
(19) |
Eliminate non-cash income tax expense. |
AMAG Pharmaceuticals, Inc.
Condensed Consolidated Statements of Operations to Non-GAAP Statements of Operations
(unaudited, amounts in thousands, except per share data)
|
|
|
Nine Months Ended |
|
Nine Months Ended |
| ||||||||||||||||||
|
|
|
September 30, 2015 |
|
September 30, 2014 |
| ||||||||||||||||||
|
|
|
GAAP |
|
Adjustments |
|
|
|
Non-GAAP |
|
GAAP |
|
Adjustments |
|
|
|
Non-GAAP |
| ||||||
|
Revenues: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
|
Makena |
|
$ |
184,258 |
|
$ |
— |
|
|
|
$ |
184,258 |
|
$ |
— |
|
$ |
— |
|
|
|
$ |
— |
|
|
Feraheme/MuGard |
|
66,726 |
|
— |
|
|
|
66,726 |
|
63,017 |
|
— |
|
|
|
63,017 |
| ||||||
|
Cord Blood Registry |
|
7,177 |
|
7,321 |
|
(20) |
|
14,498 |
|
— |
|
— |
|
|
|
— |
| ||||||
|
License fee, collaboration and other |
|
51,380 |
|
(39,965 |
) |
(21) |
|
11,415 |
|
8,114 |
|
(5,922 |
) |
(21) |
|
2,192 |
| ||||||
|
Total revenues |
|
309,541 |
|
(32,644 |
) |
|
|
276,897 |
|
71,131 |
|
(5,922 |
) |
|
|
65,209 |
| ||||||
|
Operating costs and expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
|
Cost of products sold |
|
59,793 |
|
(49,597 |
) |
(22) |
|
10,196 |
|
8,548 |
|
(1,647 |
) |
(22) |
|
6,901 |
| ||||||
|
Cost of services |
|
3,261 |
|
(215 |
) |
(23) |
|
3,046 |
|
— |
|
— |
|
|
|
— |
| ||||||
|
Research and development |
|
24,981 |
|
(3,241 |
) |
(24) |
|
21,740 |
|
16,396 |
|
(1,421 |
) |
(24) |
|
14,975 |
| ||||||
|
Selling, general and administrative |
|
110,054 |
|
(20,246 |
) |
(25) |
|
89,808 |
|
46,650 |
|
(2,513 |
) |
(25) |
|
44,137 |
| ||||||
|
Option rights to license orphan drug |
|
10,000 |
|
(10,000 |
) |
(26) |
|
— |
|
— |
|
— |
|
|
|
— |
| ||||||
|
Acquisition-related |
|
11,153 |
|
(11,153 |
) |
(27) |
|
— |
|
— |
|
— |
|
|
|
— |
| ||||||
|
Restructuring |
|
1,752 |
|
(1,752 |
) |
(28) |
|
— |
|
— |
|
— |
|
|
|
— |
| ||||||
|
Total costs and expenses |
|
220,994 |
|
(96,204 |
) |
|
|
124,790 |
|
71,594 |
|
(5,581 |
) |
|
|
66,013 |
| ||||||
|
Operating income (loss) / adjusted EBITDA |
|
88,547 |
|
63,560 |
|
|
|
152,107 |
|
(463 |
) |
(341 |
) |
|
|
(804 |
) | ||||||
|
Other income (expense): |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
|
Interest expense |
|
(34,794 |
) |
8,943 |
|
(29) |
|
(25,851 |
) |
(7,656 |
) |
4,531 |
|
(29) |
|
(3,125 |
) | ||||||
|
Loss on debt extinguishment |
|
(10,449 |
) |
10,449 |
|
(30) |
|
— |
|
— |
|
— |
|
|
|
— |
| ||||||
|
Interest and dividend income, net |
|
967 |
|
— |
|
|
|
967 |
|
809 |
|
— |
|
|
|
809 |
| ||||||
|
Other income, net |
|
(9,180 |
) |
9,185 |
|
(31) |
|
5 |
|
119 |
|
(119 |
) |
|
|
— |
| ||||||
|
Total other income (expense) |
|
(53,456 |
) |
28,577 |
|
|
|
(24,879 |
) |
(6,728 |
) |
4,412 |
|
|
|
(2,316 |
) | ||||||
|
Net income (loss) before income taxes |
|
35,091 |
|
92,137 |
|
|
|
127,228 |
|
(7,191 |
) |
4,071 |
|
|
|
(3,120 |
) | ||||||
|
Income tax expense (benefit) |
|
9,513 |
|
(9,513 |
) |
(32) |
|
|
|
— |
|
— |
|
|
|
— |
| ||||||
|
Net income (loss) / cash earnings |
|
$ |
25,578 |
|
$ |
101,650 |
|
|
|
$ |
127,228 |
|
$ |
(7,191 |
) |
$ |
4,071 |
|
|
|
$ |
(3,120 |
) |
|
Net income (loss) / cash earnings per share |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
|
Basic |
|
$ |
0.84 |
|
|
|
|
|
$ |
4.19 |
|
$ |
(0.33 |
) |
|
|
|
|
$ |
(0.14 |
) | ||
|
Diluted |
|
$ |
0.73 |
|
|
|
|
|
$ |
3.31 |
|
$ |
(0.33 |
) |
|
|
|
|
$ |
(0.14 |
) | ||
|
Weighted average shares outstanding |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
|
Basic |
|
30,379 |
|
|
|
|
|
30,379 |
|
21,912 |
|
|
|
|
|
21,912 |
| ||||||
|
Diluted |
|
34,962 |
|
|
|
|
|
38,430 |
|
21,912 |
|
|
|
|
|
21,912 |
| ||||||
|
(20) |
Adding back period write-down of deferred revenue from purchase accounting. |
|
(21) |
Eliminate non-cash revenue related to recognition of previously deferred revenue on Takeda agreement. |
|
(22) |
Eliminate the following: (i) non-cash step-up of inventory from purchase accounting (2015); (ii) amortization expense related to intangible assets; (iii) depreciation expense; and (iv) stock-based compensation expense. |
|
(23) |
Eliminate the following: (i) depreciation expense; and (ii) stock-based compensation expense. |
|
(24) |
Eliminate the following: (i) non-cash step-up of inventory from purchase accounting (2015); (ii) depreciation expense; and (iii) stock-based compensation expense. |
|
(25) |
Eliminate the following: (i) non-cash adjustments to contingent consideration; (ii) amortization expense related to intangible assets; (iii) certain transaction-related expenses (2015); (iv) depreciation expense; and (v) stock-based compensation expense. |
|
(26) |
Eliminate one-time costs related to Velo option. |
|
(27) |
Eliminate one-time acquisition costs related to Cord Blood Registry. |
|
(28) |
Eliminate one-time restructuring costs related to Cord Blood Registry acquisition. |
|
(29) |
Eliminate non-cash interest expense; amortization of debt discount and other costs. |
|
(30) |
Eliminate non-cash or one-time expenses related to the August 2015 term loan refinancing. |
|
(31) |
Eliminate one-time expenses related to the August 2015 debt financing. |
|
(32) |
Eliminate non-cash income tax expense. |
AMAG Pharmaceuticals, Inc.
Reconciliation of 2015 Updated Guidance of Non-GAAP Adjusted EBITDA
and Non-GAAP Net Income / Cash Earnings
(unaudited, amounts in millions)
|
|
|
2015 Updated |
|
|
GAAP net income |
|
$15 - 30 |
|
|
CBR deferred revenue purchase accounting adjustments |
|
20 |
|
|
Depreciation and amortization |
|
60 |
|
|
Interest expense, net |
|
70 |
|
|
Provision (benefit) for income taxes |
|
15 |
|
|
EBITDA |
|
$180 - 195 |
|
|
Non-cash collaboration revenue |
|
(40 |
) |
|
Non-cash inventory step-up adjustments |
|
15 |
|
|
Stock-based compensation |
|
15 |
|
|
Adjustment to contingent consideration |
|
10 |
|
|
Severance and transaction-related costs |
|
20 |
|
|
Velo option |
|
10 |
|
|
Adjusted EBITDA |
|
$210 – 225 |
|
|
Cash interest expense |
|
(45 |
) |
|
Non-GAAP net income – cash earnings |
|
$165 - 180 |
|
AMAG Pharmaceuticals, Inc.
Share Count Reconciliation
(amounts in millions)
|
|
|
Three Months Ended |
|
Nine Months Ended |
|
|
Weighted average basic shares outstanding |
|
33.2 |
|
30.4 |
|
|
Employee equity incentive awards |
|
1.5 |
|
3.0 |
|
|
Convertible notes |
|
7.4 |
|
— |
|
|
Warrants |
|
3.3 |
|
1.6 |
|
|
GAAP diluted shares outstanding |
|
45.4 |
(33) |
35.0 |
|
|
Adjustment(34) |
|
(4.2 |
) |
3.4 |
|
|
Non-GAAP diluted shares outstanding |
|
41.2 |
|
38.4 |
|
(33) Since the company reported a loss in this period, GAAP diluted shares outstanding are equal to GAAP basic shares outstanding. This information is included for purposes of helping the reader understand the calculation of non-GAAP shares diluted shares outstanding.
(34) Reflects the impact of the non-GAAP benefit of the bond hedge.