Exhibit 99.1

FOR IMMEDIATE RELEASE
AMAG Pharmaceuticals Reports Financial Results for Third Quarter and
Nine Months Ended September 30, 2014
20% Growth Results in Record U.S. Feraheme Sales
Lumara Health Acquisition on Track to Close in Fourth Quarter
Conference call scheduled for 8:00 a.m. EDT today
WALTHAM, MA (October 30, 2014) — AMAG Pharmaceuticals, Inc. (NASDAQ: AMAG), a specialty pharmaceutical company, today reported unaudited consolidated financial results for the third quarter and nine months ended September 30, 2014. Total revenues for the third quarter of 2014 were $25.5 million, of which $22.5 million were U.S. net Feraheme® (ferumoxytol) product sales, the highest Feraheme quarterly sales since launch in 2009. AMAG ended the third quarter with $386.2 million in cash and investments.
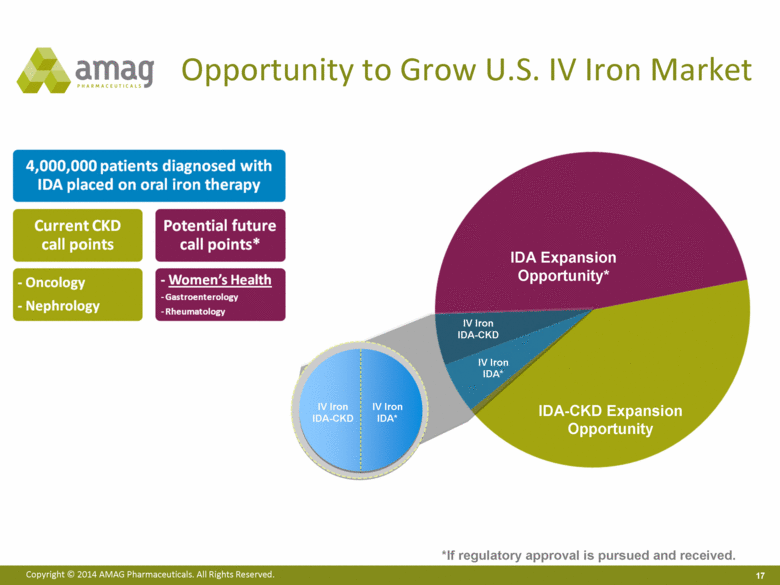
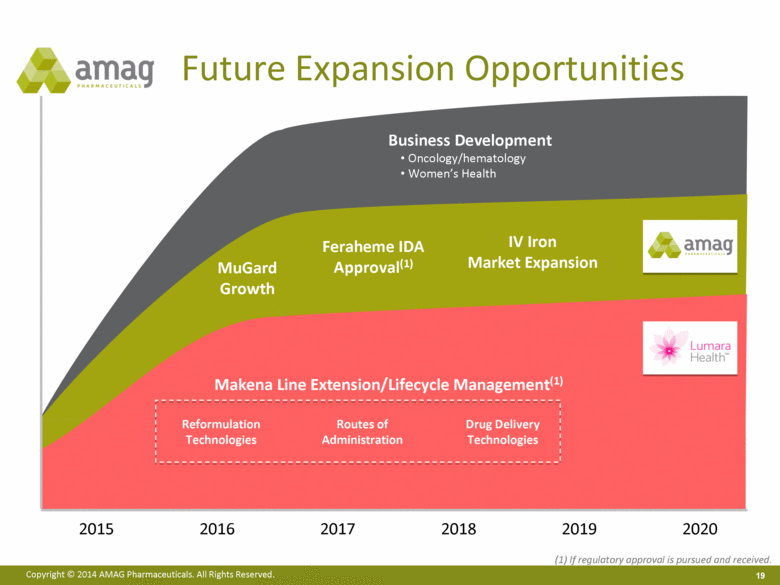
“Our ability to deliver another quarter of record sales in the fifth year since Feraheme’s launch demonstrates that more physicians across the U.S. are prescribing Feraheme for the treatment of iron deficiency anemia (IDA) in a growing number of patients with chronic kidney disease (CKD),” said William Heiden, president and chief executive officer. “In addition to driving growth of Feraheme, we also are executing on our long-term strategic plan to expand and diversify our product portfolio through our recently announced agreement to acquire Lumara Health Inc. We believe the Lumara Health business will facilitate future product acquisitions in an attractive new therapeutic area and is an excellent strategic fit with our Feraheme market expansion plans, if regulatory approval for the broader IDA indication is sought and received.”
Business Highlights
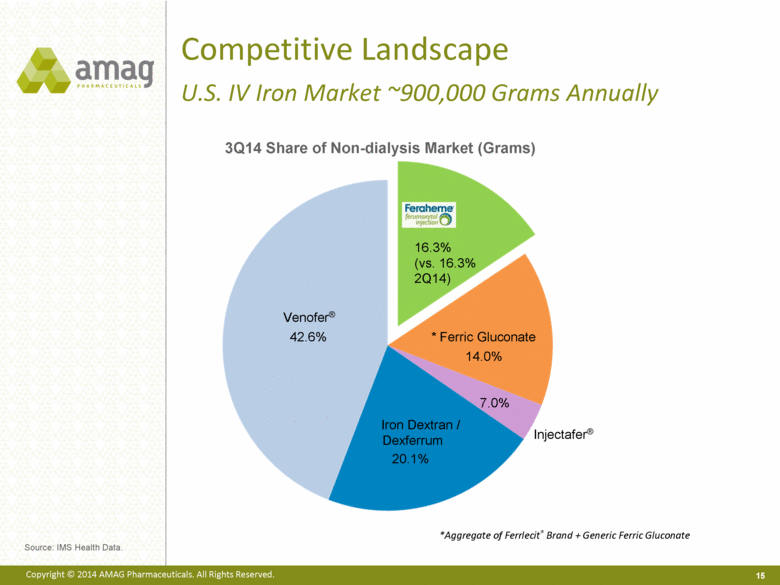
· The company reported record U.S. net Feraheme product sales of $22.5 million in the third quarter of 2014, compared to $19.3 million in the third quarter of 2013, representing a 20% increase (excluding a $0.6 million reduction in reserves for Medicaid rebates recorded in the third quarter of 2013). This growth was driven by higher volume and improved net revenue per gram of Feraheme, each up 10% from the third quarter of 2013.
· Total Feraheme provider demand for the third quarter of 2014 was approximately 37,000 grams, compared to approximately 36,000 in the third quarter of 2013.1 Several strategies led to the sales success during the third quarter of 2014, including greater penetration in the hospital segment and effective commercial contracting strategies.
1 IMS Health
· Feraheme gross margins held relatively steady at 88% in the third quarter of 2014, compared to 87% in the third quarter of 2013. Reported operating expenses for the third quarter of 2014 were $18.2 million. Excluding the favorable impact of a $3.7 million adjustment to MuGard-related contingent consideration expense in the quarter, operating expenses were $21.9 million, representing a 13% increase from the corresponding period in 2013 and a 5% increase from the second quarter of 2014. The increase in operating expenses is due largely to costs associated with the planned acquisition of Lumara Health.
· On September 29, 2014, AMAG announced that it had entered into a definitive agreement to acquire Lumara Health Inc., a privately held pharmaceutical company specializing in women’s health. Lumara Health markets the fast-growing product Makena® (hydroxyprogesterone caproate injection), which was granted 7-year orphan drug exclusivity in February 2011 and is the only FDA-approved product indicated to reduce the risk of preterm birth in women who are pregnant with one baby and who have spontaneously delivered one baby preterm in the past.
· AMAG will acquire Lumara Health for $675 million ($600 million in cash and $75 million in AMAG stock) and additional contingent consideration of up to $350 million based on achievement of certain sales milestones, including $100 million payments at $300 million, $400 million and $500 million in net sales in non-overlapping 12-month periods.
· Based on the last three months ended August 31, 2014, the combined pro forma company (AMAG and Lumara Health) is on a run rate to achieve annualized total revenue of approximately $288 million and annualized pro forma adjusted EBITDA of approximately $147 million2, including $20 million in expected annual synergies.
· Shipments of Makena for the third quarter were 20,421 vials, representing 14% growth over the quarter ended June 30, 2014. AMAG believes that attractive market dynamics, along with the implementation of a new patient-centric business strategy aimed at expanding patient access to and compliance with the product, has contributed to the significant recent growth of Makena.
“AMAG’s acquisition of Lumara Health will position the combined company for accelerated top-line growth and significant cash earnings, provide further business diversification and create value for our shareholders,” said Frank Thomas, executive vice president and chief operating officer. “Plans for integrating these two businesses are well underway, and we believe they will allow us to efficiently and quickly capture value following the close of the acquisition. We also feel confident that we are on track to complete the loan syndication process and close the transaction by the end of the year.”
2 The $147 million in annualized adjusted EBITDA is arrived at as follows: (1) AMAG net loss for the three months ended August 2014 of ($0.4) million, plus $3.1 million of interest expense and amortization of debt discount and deferred financing costs, plus $0.2 million in depreciation and amortization, less $0.3 million in interest income, plus $1.5 million in stock compensation expense, plus $0.4 million due to a fair value adjustment in contingent consideration expense, plus $0.6 million in other items; PLUS (2) Lumara Health net income for the three months ended August 2014 of $7.7 million, plus $0.9 million of interest expense and amortization of debt discount and deferred financing costs, plus $14.9 million in depreciation and amortization, plus $2.3 million in stock compensation expense, plus $0.6 million in other items; PLUS (3) $20 million in expected annual synergies.
Third Quarter and Nine Month 2014 Financial Results (unaudited)
The company reported record U.S. net Feraheme product sales of $22.5 million for the third quarter of 2014, compared to $19.3 million in the third quarter of 2013. For the nine months ended September 30, 2014, AMAG reported U.S. net Feraheme product sales of $62.1 million, compared to $52.4 million for the corresponding period in 2013.
Total revenues for the quarter ended September 30, 2014 were $25.5 million, as compared to $21.6 million for the third quarter of 2013. For the nine months ended September 30, 2014, AMAG reported total revenues of $71.1 million, as compared to revenues of $59.1 million for the corresponding period in 2013.
Feraheme cost of goods sold (COGS) for the quarter ended September 30, 2014 were $2.8 million, or 12% of net Feraheme product sales, compared to $2.5 million, or 13% of net Feraheme product sales for the quarter ended September 30, 2013. For the nine months ended September 30, 2014, Feraheme COGS were $8.2 million, or 13% of net Feraheme product sales, compared to $8.4 million, or 16% of net Feraheme product sales, for the corresponding period in 2013.
Total operating expenses for the quarter ended September 30, 2014 were $18.2 million, compared to $19.5 million for the third quarter of 2013. Total operating expenses for the nine months ended September 30, 2014 were $63.0 million, as compared to operating expenses of $58.1 million for the corresponding period in 2013.
The company reported net income of $1.5 million, or $0.07 per basic share, and $0.06 per diluted share, for the quarter ended September 30, 2014, as compared to net loss of $0.1 million, or $0.01 per basic and diluted share, for the third quarter of 2013. AMAG reported a net loss for the nine months ended September 30, 2014 of $7.2 million, or $0.33 per basic and diluted share, as compared to a net loss of $5.9 million, or $0.28 per basic and diluted share, for the corresponding period in 2013.
As of September 30, 2014, the company’s cash and investments totaled approximately $386.2 million, reflecting net cash usage during the third quarter of approximately $0.3 million. The Company expects to have approximately $100 million in cash and investments following the close of the Lumara Health transaction.
Conference Call and Webcast Access
AMAG Pharmaceuticals, Inc. will host a conference call and webcast with slides today at 8:00 a.m. EDT, during which management will discuss the company’s financial results, the acquisition of Lumara Health and commercial progress. To access the conference call via telephone, please dial (877) 412-6083 from the United States or (702) 495-1202 for international access. A telephone replay will be available from approximately 11:00 a.m. ET on October 30, 2014 through midnight November 6, 2014. To access a replay of the conference call, dial (855) 859-2056 from the United States or (404) 537-3406 for international access. The pass code for the live call and the replay is 25342306.
The call will be webcast with slides and accessible through the Investors section of the company’s website at www.amagpharma.com. The webcast replay will be available from approximately 11:00 a.m. ET on October 30, 2014 through midnight on November 29, 2014.
AMAG Pharmaceuticals, Inc.
Condensed Consolidated Statements of Operations
(unaudited, amounts in thousands, except per share data)
|
|
|
Three Months Ended |
|
Nine Months Ended |
| ||||||||
|
|
|
2014 |
|
2013 |
|
2014 |
|
2013 |
| ||||
|
Revenues: |
|
|
|
|
|
|
|
|
| ||||
|
U.S. Feraheme product sales, net |
|
$ |
22,547 |
|
$ |
19,347 |
|
$ |
62,147 |
|
$ |
52,381 |
|
|
Other product sales and royalties |
|
765 |
|
271 |
|
1,560 |
|
708 |
| ||||
|
License fee and other collaboration revenues |
|
2,182 |
|
1,998 |
|
7,424 |
|
6,056 |
| ||||
|
Total revenues |
|
25,494 |
|
21,616 |
|
71,131 |
|
59,145 |
| ||||
|
Operating costs and expenses: |
|
|
|
|
|
|
|
|
| ||||
|
Cost of product sales |
|
2,968 |
|
2,547 |
|
8,548 |
|
8,634 |
| ||||
|
Research and development expenses |
|
5,358 |
|
4,530 |
|
16,396 |
|
13,983 |
| ||||
|
Selling, general and administrative expenses |
|
12,875 |
|
14,934 |
|
46,650 |
|
44,150 |
| ||||
|
Total operating costs and expenses |
|
21,201 |
|
22,011 |
|
71,594 |
|
66,767 |
| ||||
|
Operating income (loss) |
|
4,293 |
|
(395 |
) |
(463 |
) |
(7,622 |
) | ||||
|
Interest expense |
|
(3,129 |
) |
— |
|
(7,656 |
) |
— |
| ||||
|
Interest and dividend income, net |
|
291 |
|
246 |
|
809 |
|
773 |
| ||||
|
Gains on sale of assets |
|
— |
|
— |
|
102 |
|
865 |
| ||||
|
Gains on investments, net |
|
3 |
|
4 |
|
17 |
|
36 |
| ||||
|
Net income (loss) before income taxes |
|
1,458 |
|
(145 |
) |
(7,191 |
) |
(5,948 |
) | ||||
|
Income tax benefit |
|
— |
|
— |
|
— |
|
— |
| ||||
|
Net income (loss) |
|
$ |
1,458 |
|
$ |
(145 |
) |
$ |
(7,191 |
) |
$ |
(5,948 |
) |
|
|
|
|
|
|
|
|
|
|
| ||||
|
Net income (loss) per basic share |
|
$ |
0.07 |
|
$ |
(0.01 |
) |
$ |
(0.33 |
) |
$ |
(0.28 |
) |
|
Net income (loss) per diluted share |
|
$ |
0.06 |
|
$ |
(0.01 |
) |
$ |
(0.33 |
) |
$ |
(0.28 |
) |
|
|
|
|
|
|
|
|
|
|
| ||||
|
Weighted average shares outstanding used to compute net income (loss) per share: |
|
|
|
|
|
|
|
|
| ||||
|
Basic |
|
21,984 |
|
21,691 |
|
21,912 |
|
21,613 |
| ||||
|
Diluted |
|
23,467 |
|
21,691 |
|
21,912 |
|
21,613 |
| ||||
AMAG Pharmaceuticals, Inc.
Condensed Consolidated Balance Sheets
(unaudited, amounts in thousands)
|
|
|
September 30, 2014 |
|
December 31, 2013 |
| ||
|
Cash and cash equivalents |
|
$ |
196,154 |
|
$ |
26,986 |
|
|
Short-term investments |
|
190,088 |
|
186,803 |
| ||
|
Accounts receivable, net |
|
10,873 |
|
6,842 |
| ||
|
Inventories |
|
19,580 |
|
17,217 |
| ||
|
Receivable from collaboration |
|
932 |
|
278 |
| ||
|
Other current assets |
|
9,357 |
|
6,279 |
| ||
|
Total current assets |
|
426,984 |
|
244,405 |
| ||
|
Property and equipment, net |
|
1,582 |
|
1,846 |
| ||
|
Intangible assets, net |
|
16,597 |
|
16,844 |
| ||
|
Other assets |
|
6,229 |
|
2,364 |
| ||
|
Total assets |
|
$ |
451,392 |
|
$ |
265,459 |
|
|
|
|
|
|
|
| ||
|
Accounts payable |
|
$ |
1,899 |
|
$ |
2,629 |
|
|
Accrued expenses |
|
27,521 |
|
22,266 |
| ||
|
Deferred revenues |
|
9,419 |
|
8,226 |
| ||
|
Total current liabilities |
|
38,839 |
|
33,121 |
| ||
|
Convertible 2.5% senior notes, net |
|
165,778 |
|
— |
| ||
|
Deferred revenues |
|
36,682 |
|
44,534 |
| ||
|
Acquisition-related contingent consideration |
|
11,188 |
|
13,609 |
| ||
|
Other long-term liabilities |
|
1,924 |
|
1,787 |
| ||
|
Total long-term liabilities |
|
215,572 |
|
59,930 |
| ||
|
Total stockholders’ equity |
|
196,981 |
|
172,408 |
| ||
|
Total liabilities and stockholders’ equity |
|
$ |
451,392 |
|
$ |
265,459 |
|
About AMAG
AMAG Pharmaceuticals, Inc. is a specialty pharmaceutical company that markets Feraheme® (ferumoxytol) Injection and MuGard® Mucoadhesive Oral Wound Rinse in the United States. Along with driving continued growth of its products, AMAG intends to continue to expand its portfolio through the in-license or purchase of additional pharmaceutical products or companies, including revenue-generating commercial products and late-stage development assets that leverage its corporate infrastructure, sales force call points and commercial expertise. Our primary goal is to bring to market therapies that provide clear benefits and improve patients’ lives. For additional company information, please visit www.amagpharma.com.
About Lumara Health
Lumara Health™ is a specialty pharmaceutical company committed to advancing the health of women throughout the stages of their lives, with a particular focus on maternal health. At the heart of Lumara Health is our mission to help women achieve healthier lives. For more information on Lumara Health, please visit www.lumarahealth.com.
About Makena® (hydroxyprogesterone caproate injection)
Makena® is a progestin indicated to reduce the risk of preterm birth in women with a singleton pregnancy who have a history of singleton spontaneous preterm birth.
The effectiveness of Makena is based on improvement in the proportion of women who delivered <37 weeks of gestation. There are no controlled trials demonstrating a direct clinical benefit, such as improvement in neonatal mortality and morbidity.
Limitation of use: While there are many risk factors for preterm birth, safety and efficacy of Makena has been demonstrated only in women with a prior spontaneous singleton preterm birth. It is not intended for use in women with multiple gestations or other risk factors for preterm birth.
Makena should not be used in women with any of the following conditions: blood clots or other blood clotting problems, breast cancer or other hormone-sensitive cancers, or history of these conditions; unusual vaginal bleeding not related to the current pregnancy, yellowing of the skin due to liver problems during pregnancy, liver problems, including liver tumors, or uncontrolled high blood pressure.
Before patients receive Makena, they should tell their healthcare provider if they have an allergy to hydroxyprogesterone caproate, castor oil, or any of the other ingredients in Makena; diabetes or prediabetes, epilepsy, migraine headaches, asthma, heart problems, kidney problems, depression, or high blood pressure.
In one clinical study, certain complications or events associated with pregnancy occurred more often in women who received Makena. These included miscarriage (pregnancy loss before 20 weeks of pregnancy), stillbirth (fetal death occurring during or after the 20th week of pregnancy), hospital admission for preterm labor, preeclampsia (high blood pressure and too much protein in the urine), gestational hypertension (high blood pressure caused by pregnancy), gestational diabetes, and oligohydramnios (low amniotic fluid levels).
Makena may cause serious side effects including blood clots, allergic reactions, depression, and yellowing of the skin and the whites of the eyes. The most common side effects of Makena include injection site reactions (pain, swelling, itching, bruising, or a hard bump), hives, itching, nausea, and diarrhea.
For additional U.S. product information, including full prescribing information, please visit www.makena.com.
About Feraheme® (ferumoxytol)/Rienso
Feraheme (ferumoxytol) Injection for IV use received marketing approval from the FDA on June 30, 2009 for the treatment of IDA in adult chronic kidney disease (CKD) patients and was commercially launched by AMAG in the U.S. shortly thereafter. Ferumoxytol is protected in the U.S. by five issued patents covering the composition and dosage form of the product. Each issued patent is listed in the FDA’s Orange Book, the last of which expires in June 2023.
Ferumoxytol received marketing approval in Canada in December 2012, where it is marketed by Takeda as Feraheme, and in the European Union in June 2013 where it is marketed by Takeda as Rienso™. Ferumoxytol received marketing approval in Switzerland in August 2013.
Feraheme is contraindicated in patients with known hypersensitivity to Feraheme or any of its components. Serious hypersensitivity reactions, including anaphylactic-type reactions, some of which have life-threatening and fatal, have been reported in patients receiving Feraheme. Serious adverse reactions of clinically significant hypotension have been reported in the post-marketing experience of Feraheme.
For additional U.S. product information, including full prescribing information, please visit www.feraheme.com.
About MuGard
MuGard® Mucoadhesive Oral Wound Rinse is indicated for the management of oral mucositis/stomatitis (that may be caused by radiotherapy and/or chemotherapy) and all types of oral wounds (mouth sores and injuries), including aphthous ulcers/canker sores and traumatic ulcers, such as those caused by oral surgery or ill-fitting dentures or braces. MuGard is contraindicated in patients with known hypersensitivity to any of the ingredients in the formulation. MuGard was launched in 2010 after receiving 510(k) clearance from the U.S. Food and Drug Administration.
Forward-looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 (PSLRA) and other federal securities laws. Any statements contained herein which do not describe historical facts, including but not limited to, statements regarding: (i) physician behaviors and growth in the number of patients with CKD; (ii) AMAG’s plans to build a multi-product specialty pharmaceutical company; (iii) the transaction with Lumara Health and the addition of Makena to AMAG’s product portfolio, including expected benefits and the impact on shareholder value and AMAG’s business diversification, as well as the timing of the closing of the transaction with Lumara Health; (iv) AMAG’s belief that the Lumara Health business will facilitate future product acquisitions and its strategic fit with AMAG’s Feraheme market expansion plans, if regulatory approval for the broader IDA indication is sought and received; (v) AMAG’s plans to expand the market for Feraheme; (vi) expectations that the combined company, after the completion of AMAG’s acquisition of Lumara Health, will see annualized total revenue of approximately $288 million, adjusted EBITDA of approximately $147 million and expected annual synergies of $20 million; (vii) beliefs and expectations regarding integration efforts, (viii) beliefs regarding the drivers of recent growth in sales of Makena; (ix) AMAG’s belief that this transaction will position AMAG for accelerated top-line growth and significant cash earnings, further portfolio diversification and shareholder value creation; (x) beliefs about the commercial platform and the strategic fit of Lumara Health; (xi) expectations that the combined company will generate cost synergies and other efficiencies; and (xii) AMAG’s beliefs regarding the timing of completion and expected closing of the pending loan syndication process in connection with the Lumara Health transaction are forward-looking statements which involve risks and uncertainties that could cause actual results to differ materially from those discussed in such forward-looking statements. Statements about AMAG’s or Lumara Health’s past financial results do not, and are not meant to, predict future results. AMAG can provide no assurance that such results and performance will continue.
Such risks and uncertainties include, among others: (1) the possibility that the closing conditions set forth in the definitive agreement for the acquisition of Lumara Health will not be met and that the parties will be unable to consummate the proposed transaction, (2) the chance that, despite having a commitment in place for the financing of the Lumara Health acquisition, AMAG will be unable to secure such financing, or financing on satisfactory terms, in amounts sufficient to consummate the acquisition of Lumara Health, (3) the possibility that if the Lumara Health acquisition is consummated, AMAG may not realize the expected benefits, synergies and opportunities anticipated in connection with the transaction, including the anticipated costs synergies of $20 million, annualized sales of more than $180 million, continued revenue growth, annualized EBITDA of $110 million and $1 billion market opportunity, (4) the challenges of integrating the Makena commercial team into AMAG, (5) the impact on sales of Makena from competitive,
commercial payor, government (including federal and state Medicaid reimbursement policies), physician, patient or public responses with respect to product pricing, product access and sales and marketing initiatives, (6) the impact of patient compliance on units sales, (7) the uncertainty of achieving sales of Feraheme to OB/GYN specialists for the treatment of women who suffer from IDA, even assuming FDA approval for the broader IDA indication, (8) AMAG may face challenges in leveraging its in-office injectables commercial expertise, which could result in unforeseen expenses and disrupt business operations, (9) liabilities AMAG assumes from Lumara Health, including the class action litigation In Re K-V Pharmaceutical Company Securities Litigation, Case No. 4:11CV1816 AGF, may be higher than expected, (10) the possibility that sales of Makena will not meet expectations as a result of current and future competition from compounded products and/or future competition from generic alternatives upon expiration of exclusivity in February 2018, (11) the impact of reimbursement policies for Makena and the resulting coverage decisions and/or impact on pricing, (12) the number of preterm birth risk pregnancies for which Makena may be prescribed, its safety and side effects profile and acceptance of pricing, (13) in connection with the Lumara Health acquisition, AMAG will incur a substantial amount of indebtedness and will have to comply with restrictive and affirmative debt covenants, including a requirement that AMAG reduce its leverage over time, (14) the possibility that AMAG will need to raise additional capital from the sale of its common stock, which will cause significant dilution to AMAG’s stockholders, in order to satisfy its contractual obligations, including its debt service, milestone payments that may become payable to Lumara Health’s stockholders and/or in order to pursue business development activities, (15) upon consummation of the Lumara Health transaction, AMAG will be highly leveraged and have limited cash and cash equivalent resources which may limit its ability to take advantage of attractive business development opportunities and execute on its strategic plan, (16) the possibility that AMAG’s stockholders will not approve the amendment to its shareholder rights plan and that AMAG’s tax benefits, including those acquired upon consummation of the Lumara Health acquisition, will not be available in the future, (17) the likelihood and timing of potential approval of Feraheme in the U.S. in the broader IDA indication in light of the complete response letter AMAG received from the FDA informing AMAG that its supplemental new drug application (sNDA) for the broader indication could not be approved in its present form and stating that AMAG had not provided sufficient information to permit labeling of Feraheme for safe and effective use for the proposed broader indication, (18) the possibility that following FDA review of post-marketing safety data, including reports of serious anaphylaxis, cardiovascular events, and death, and/or in light of the label changes requested by the European Medicines Agency’s (EMA) Pharmacovigilance Risk Assessment Committee (PRAC) and confirmed by the Committee for Medicinal Products for Human Use (CHMP), the FDA (or other regulators) will request additional technical or scientific information, new studies or reanalysis of existing data, on-label warnings, post-marketing requirements/commitments or risk evaluation and mitigation strategies (REMS) in the current indication for Feraheme for IDA in adult patients with CKD and the additional costs and expenses that will or may be incurred in connection with such activities, (19) whether AMAG’s proposed label changes will be acceptable to the FDA or other regulatory authorities and what impact such changes, or such additional changes as the FDA, CHMP or other regulators may require, will have on sales of Feraheme/ Rienso™ (Rienso is the trade name for ferumoxytol outside of the U.S. and Canada), (20) AMAG’s and Takeda Pharmaceutical Company Limited’s ability to successfully compete in the IV iron replacement market both in the U.S. and outside the U.S., including the EU, as a result of limitations, restrictions or warnings in Feraheme’s/Rienso’s current or future label, including that Feraheme/Rienso be administered to patients by infusion over at least 15-minutes (replacing injection) and that it be contraindicated for patients with any known history of drug allergy, (21) AMAG’s ability to execute on its long-term strategic plan or to realize the expected results from its long-term strategic plan, (22) Takeda’s ability to obtain regulatory approval for Feraheme in Canada, and Rienso in the
EU, in the broader IDA patient population, (23) the possibility that significant safety or drug interaction problems could arise with respect to Feraheme/Rienso and in turn affect sales, or AMAG’s ability to market the product both in the U.S. and outside of the U.S., including the EU, (24) the relationship between Takeda and AMAG and the impact on commercialization efforts for Feraheme/Rienso in the EU and Canada, (25) the likelihood and timing of milestone payments, if any, in connection with AMAG’s licensing arrangement with Takeda, (26) the manufacture of Feraheme/Rienso or MuGard (or Makena if the acquisition is consummated), including any significant interruption in the supply of raw materials or finished product, (27) AMAG’s patents and proprietary rights both in the U.S. and outside the U.S. (including those that AMAG acquires from the acquisition of Lumara), (28) the risk of an Abbreviated New Drug Application (ANDA) filing for generic ferumoxytol, especially following the FDA’s draft bioequivalence recommendation for ferumoxytol published in December 2012 (or, following the consummation of the Lumara Health acquisition, hydroxyprogesterone caproate), (29) the possibility that AMAG (or Takeda) will disseminate future Dear Healthcare Provider letters, (30) uncertainties regarding AMAG’s ability to compete in the oral mucositis market in the U.S. and in the women’s maternal health market and (31) other risks identified in AMAG’s filings with the U.S. Securities and Exchange Commission (SEC), including its Quarterly Report on Form 10-Q for the quarter ended June 30, 2014 and subsequent filings with the SEC. Any of the above risks and uncertainties could materially and adversely affect AMAG’s results of operations, profitability and cash flows, which would, in turn, have a significant and adverse impact on AMAG’s stock price. Use of the term “including” in the two paragraphs above shall mean in each case “including, but not limited to.” AMAG cautions you not to place undue reliance on any forward-looking statements, which speak only as of the date they are made.
AMAG disclaims any obligation to publicly update or revise any such statements to reflect any change in expectations or in events, conditions or circumstances on which any such statements may be based, or that may affect the likelihood that actual results will differ from those set forth in the forward-looking statements.
AMAG Pharmaceuticals and Feraheme are registered trademarks of AMAG Pharmaceuticals, Inc. MuGard is a registered trademark of Access Pharmaceuticals, Inc. Rienso is a registered trademark of Takeda Pharmaceutical Company Limited. Lumara Health is a trademark of Lumara Health Inc. Makena is a registered trademark of Lumara Health Inc.
AMAG Pharmaceuticals, Inc. Contact
Katie Payne, 617-498-3303