
Welcome to
AMAG’s Analyst Day
May 24, 2017

2
Forward-Looking Statements
This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 (PSLRA) and
other federal securities laws. Any statements contained herein which do not describe historical facts, including, among others, AMAG’s 2017
financial guidance, including forecasted GAAP and non-GAAP revenues, GAAP net income and operating income, non-GAAP adjusted EBITDA, and
GAAP research and development; AMAG’s strategy for growth, including through acquisitions, its commercial platform, diversification,
profitability and drug development; future growth drivers for Makena, including the launch of the Makena subcutaneous auto-injector, if
approved, positive and negative influences, strategies for increasing market share, increasing the average dose per patient, holding net price
stable and maintaining inventory levels; beliefs that the current Makena subcutaneous auto-injector formulation offers benefits to physicians and
patients, including the potential for greater convenience, alternative administration, increased patient compliance and the competitiveness of
pricing; patient and health care providers preferences to the Makena intramuscular option, launch of an AMAG owned/authorized generic for
Makena, and benefits of product/formulation mix to patients and any related impacts on AMAG’s revenues; expectations regarding timing of the
Makena subcutaneous auto-injector for an FDA decision on the sNDA and commercial launch, if approved; growth drivers for Cord Blood Registry
(CBR), including AMAG’s market position, the steady cash flow and high margin recurring revenue business model, and marketing and
communication strategies for patients and to leverage AMAG’s product portfolio; plans to differentiate CBR offerings and increase engagement
and communications in the industry; growth drivers for Feraheme, including plans to grow in key segments, expectations that the size of the
addressable market, if the broader indication is approved, would double, and the potential for synergies with AMAG’s sales force; establishment
of Feraheme as the IV iron product of choice for adult IDA patients; expected timing for submission of the sNDA for the expanded Feraheme
label (including expected timing for an FDA decision on the sNDA and commercial launch, if approved); the competitive landscape and breadth of
the vulvar and vaginal atrophy markets and Intrarosa’s market potential; the potential benefits, safety and efficacy profile, and commercial
opportunity of Intrarosa; drivers of pricing, including the low degree of payer management, broad access enjoyed by all therapies and patient
assistance programs; expectations on timing of Intrarosa launch, including the full campaign and direct-to-consumer push; the expected timeline
of bremelanotide, including the timing for the drug-drug interaction and safety pharmacology studies, NDA submission, subsequent FDA action
and commercial launch; the competitive landscape and breadth of hypoactive sexual desire disorder (HSDD) market and bremelanotide’s market
potential; AMAG’s strategy in preparation for the early 2019 launch of bremelanotide, including patient segmentation and market and brand
development; AMAG’s capital allocation and expected debt payments; search and evaluation criteria for therapeutic areas; and the expected
timing of initiation of the Velo Phase 2b/3a study and commercial launch, if approved, are forward-looking statements which involve risks and
uncertainties that could cause actual results to differ materially from those discussed in such forward-looking statements.
Such risks and uncertainties include, among others, risks identified in AMAG’s filings with the U.S. Securities and Exchange Commission (the
“SEC”), including its Annual Report on Form 10-K for the year ended December 31, 2016, Quarterly Report on Form 10-Q for the quarter ended
March 31, 2017, and subsequent filings with the SEC. AMAG cautions you not to place undue reliance on any forward-looking statements, which
speak only as of the date they are made. AMAG disclaims any obligation to publicly update or revise any such statements to reflect any change in
expectations or in events, conditions or circumstances on which any such statements may be based, or that may affect the likelihood that actual
results will differ from those set forth in the forward-looking statements.

3
Cautionary Disclosure Regarding AMAG’s Long-Term Outlook
Slides 14, 18, 19, 20 21, 27, 93, 135, 136, 137 and 139 contain forward-looking estimates of AMAG’s growth trajectory in
revenue and EBITDA on a multi-year timeframe based on a strategy of increasing revenue diversification by leveraging its
commercialization and drug-development capabilities. In addition to the risk factors and forward-looking statement
disclosed above, these estimates involve risks and uncertainties related to: (i) AMAG’s plan to build a portfolio of novel
products meeting unmet medical needs; (ii) an increased focus on durable assets; (iii) ongoing efforts to leverage clinical
development capabilities against later-stage, lower-risk development opportunities; (iv) the need to dedicate cash-flow
to the funding of AMAG’s evolving business model; (v) the uncertain commercial potential of therapeutic areas of
interest; and (vi) external pricing/reimbursement. The purpose of these long-term revenue and EBITDA estimates is to
illustrate AMAG’s current growth model based on current plans for the advancement of Intrarosa, bremelanotide,
Feraheme, Makena, CBR and future portfolio expansion. These estimates include assumptions based on current
circumstances with respect to, among other things, (A) design and execution of clinical studies, (B) anticipated timetables
for regulatory filings and related reviews and potential approvals of products, (C) cost and timing for commercial
launches, and (D) forecasted volumes and pricing. In addition, with respect to Makena and Feraheme, these estimates
assume approval of the Makena subcutaneous auto-injector, approval of Feraheme for the broader IDA indication and
does not take into consideration the outcome related to an ongoing patent infringement suit against Sandoz Inc. There
can be no assurance that all or any of the assumptions and estimates built into our long-term models will prove correct,
and we caution you not to place undue reliance on such statements and the overall progression of revenue for specific
products, as the timing of regulatory approvals, clinical study results, commercial launch, volume and pricing may turn
out to be significantly different from our current estimates.
Such risks and uncertainties include, among others, risks identified in AMAG’s SEC filings, including its Annual Report on
Form 10-K for the year ended December 31, 2016, Quarterly Report on Form 10-Q for the quarter ended March 31,
2017, and subsequent filings with the SEC. AMAG cautions you not to place undue reliance on any forward-looking
statements, which speak only as of the date they are made. AMAG disclaims any obligation to publicly update or revise
any such statements to reflect any change in expectations or in events, conditions or circumstances on which any such
statements may be based, or that may affect the likelihood that actual results will differ from those set forth in the
forward-looking statements.
AMAG Pharmaceuticals® and Feraheme® are registered trademark of AMAG Pharmaceuticals, Inc. MuGard® is a registered
trademark of Abeona Therapeutics, Inc. Makena® is a registered trademark of AMAG Pharmaceuticals IP, Ltd. Cord Blood
Registry® and CBR® are registered trademarks of CBR Systems, Inc. IntrarosaTM is a trademark of Endoceutics, Inc.

4
AMAG Executive Team Speakers
Bill Heiden
President and
Chief Executive Officer
Ted Myles
Chief Financial Officer
Nik Grund
Chief Commercial Officer
Julie Krop, MD
Chief Medical Officer

5
1 Dr. Simon and Dr. Kagan were compensated for their time and participation in this event and periodically
provide consulting services to AMAG.
Guest Speakers
Risa Kagan, MD, FACOG, CCD, NCMP1
• Clinical Professor in the Department of Obstetrics, Gynecology, and
Reproductive Sciences, University of California, San Francisco
• Practicing gynecologist with the East Bay Physicians Medical Group,
Sutter Medical Foundation
Fernand Labrie, MD, PhD
• Chief Executive Officer and founder of EndoceuticsTM
• Emeritus Professor, Laval University
James Simon, MD, CCD, NCMP, IF, FACOG1
• Clinical Professor of Obstetrics and Gynecology,
George Washington University
• Medical Director, Women’s Health & Research Consultants®
• Current President-elect of The International Society for the Study of
Women’s Sexual Health (ISSWSH)

6
Agenda – AMAG Analyst Day 2017
Registration and continental breakfast 7:30 am – 8:30 am
Welcome and agenda Linda Lennox, VP, Investor Relations
Strategic overview and outlook Bill Heiden, President and CEO
AMAG products – currently marketed
Makena®
Feraheme®
Cord Blood Registry® (CBR®)
Nik Grund, Chief Commercial Officer
Julie Krop, MD, Chief Medical Officer
AMAG products – future
IntrarosaTM
Bremelanotide
Nik Grund
Julie Krop, MD
Risa Kagan, MD, UCSF
Fernand Labrie, MD, Endoceutics CEO & Founder
James Simon, MD, George Washington University
Q&A with external experts Moderated by Julie Krop, MD
Financial strategy and business development Ted Myles, Chief Financial Officer
Closing remarks
Near-term milestones
Longer-term vision
Bill Heiden

Strategic Overview
& Outlook
Bill Heiden
AMAG President & Chief
Executive Officer
8
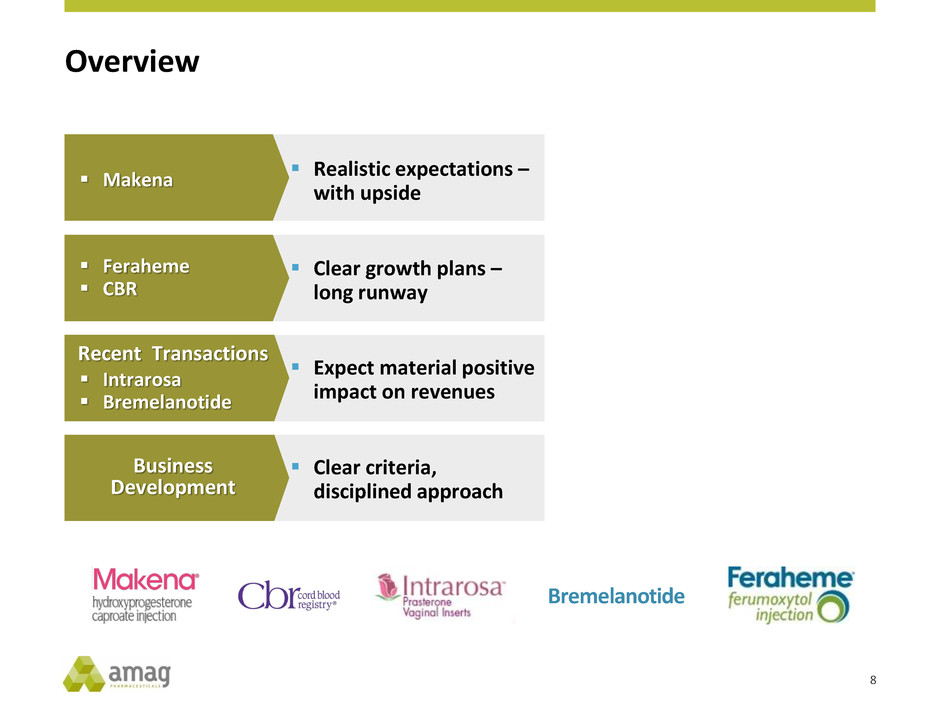
Overview
Bremelanotide
Realistic expectations –
with upside
Makena
Expect material positive
impact on revenues
Recent Transactions
Intrarosa
Bremelanotide
Clear growth plans –
long runway
Feraheme
CBR
Clear criteria,
disciplined approach
Business
Development

9
Agenda
AMAG: Yesterday and Today
AMAG: Tomorrow
10
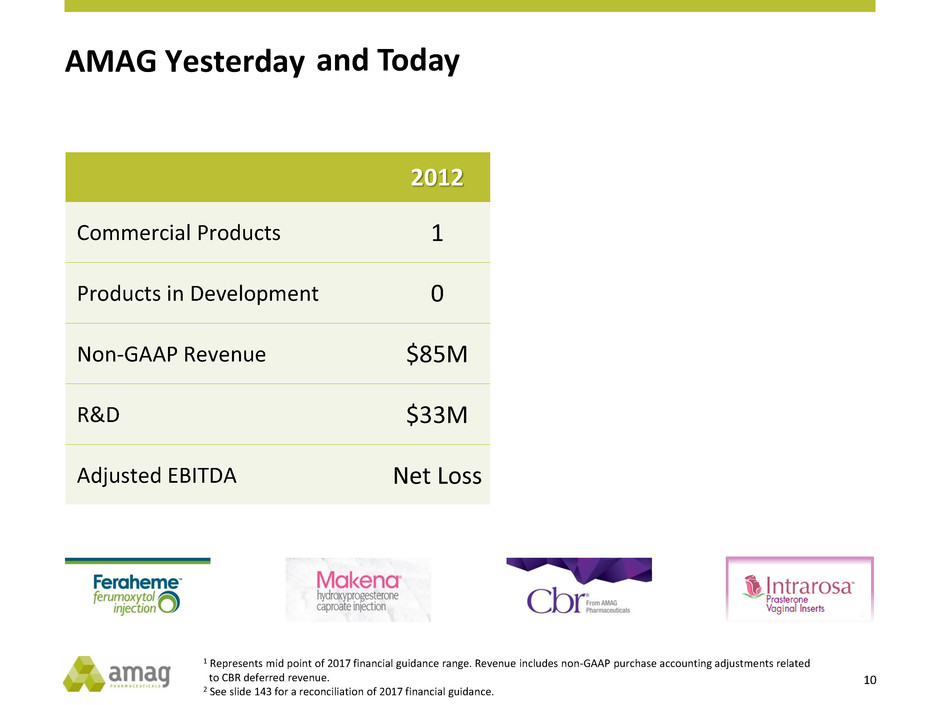
1 Represents mid point of 2017 financial guidance range. Revenue includes non-GAAP purchase accounting adjustments related
to CBR deferred revenue.
2 See slide 143 for a reconciliation of 2017 financial guidance.
AMAG Yesterday
2012 2017 E
Commercial Products 1 5
Products in Development 0 2
Non-GAAP Revenue $85M $660M1
R&D $33M ~$100M
Adjusted EBITDA Net Loss $235M1,2
and Today
11
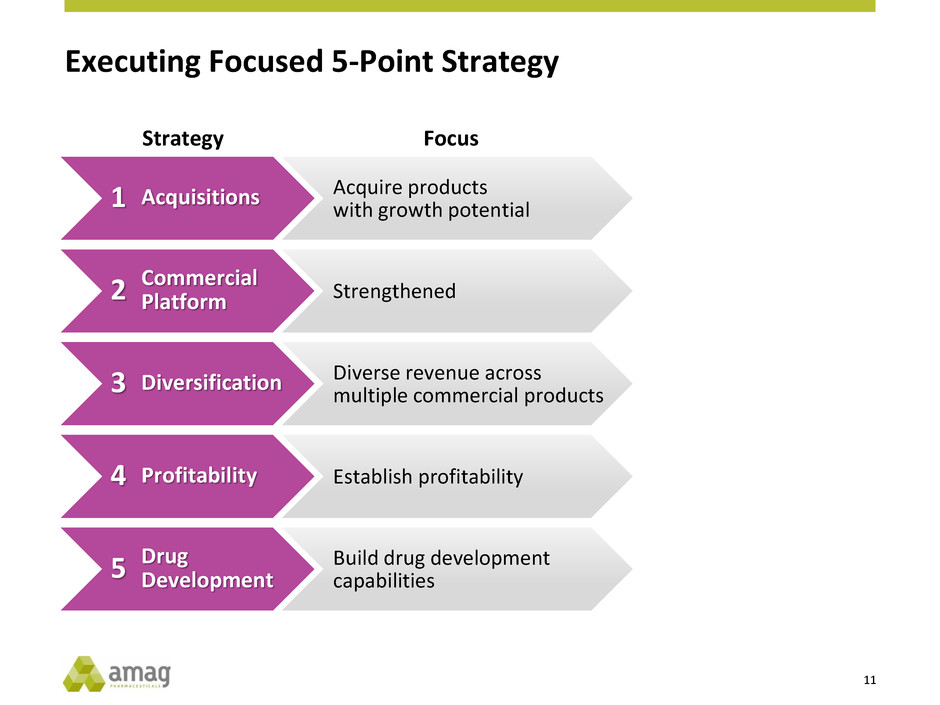
Executing Focused 5-Point Strategy
Acquire products
with growth potential
Strengthened
Diverse revenue across
multiple commercial products
Establish profitability
Build drug development
capabilities
Focus
Acquisitions
Commercial
Platform
Diversification
Profitability
Drug
Development
Strategy
1
2
3
4
5
12
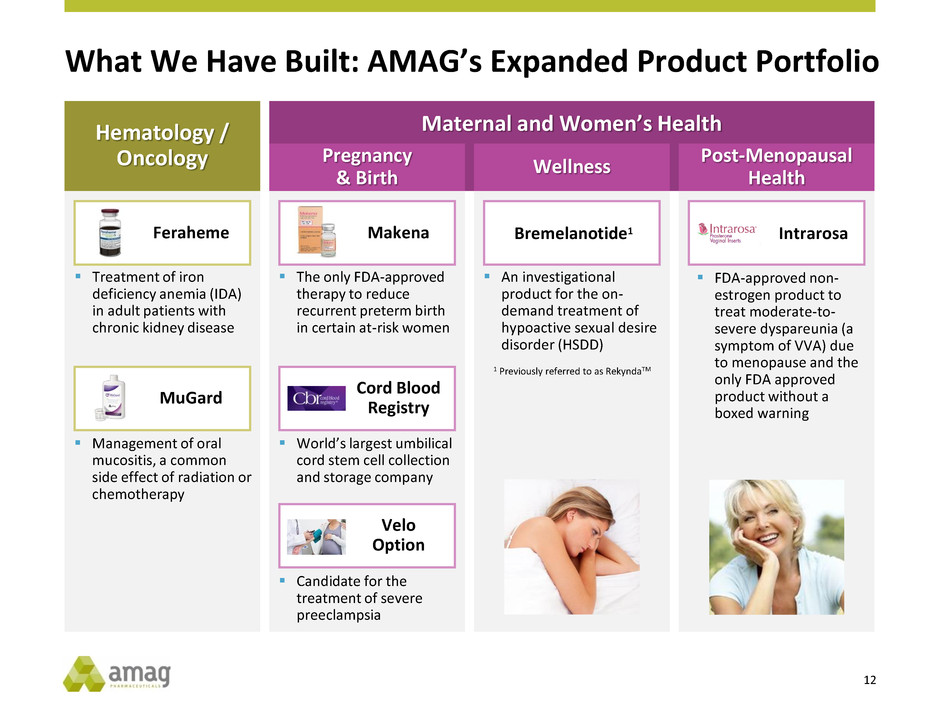
What We Have Built: AMAG’s Expanded Product Portfolio
Feraheme
Treatment of iron
deficiency anemia (IDA)
in adult patients with
chronic kidney disease
The only FDA-approved
therapy to reduce
recurrent preterm birth
in certain at-risk women
World’s largest umbilical
cord stem cell collection
and storage company
Candidate for the
treatment of severe
preeclampsia
An investigational
product for the on-
demand treatment of
hypoactive sexual desire
disorder (HSDD)
MuGard
Management of oral
mucositis, a common
side effect of radiation or
chemotherapy
Maternal and Women’s Health Hematology /
Oncology Pregnancy
& Birth
Wellness
Post-Menopausal
Health
Makena
Velo
Option
Cord Blood
Registry
Bremelanotide1 Intrarosa
1 Previously referred to as RekyndaTM
FDA-approved non-
estrogen product to
treat moderate-to-
severe dyspareunia (a
symptom of VVA) due
to menopause and the
only FDA approved
product without a
boxed warning
13
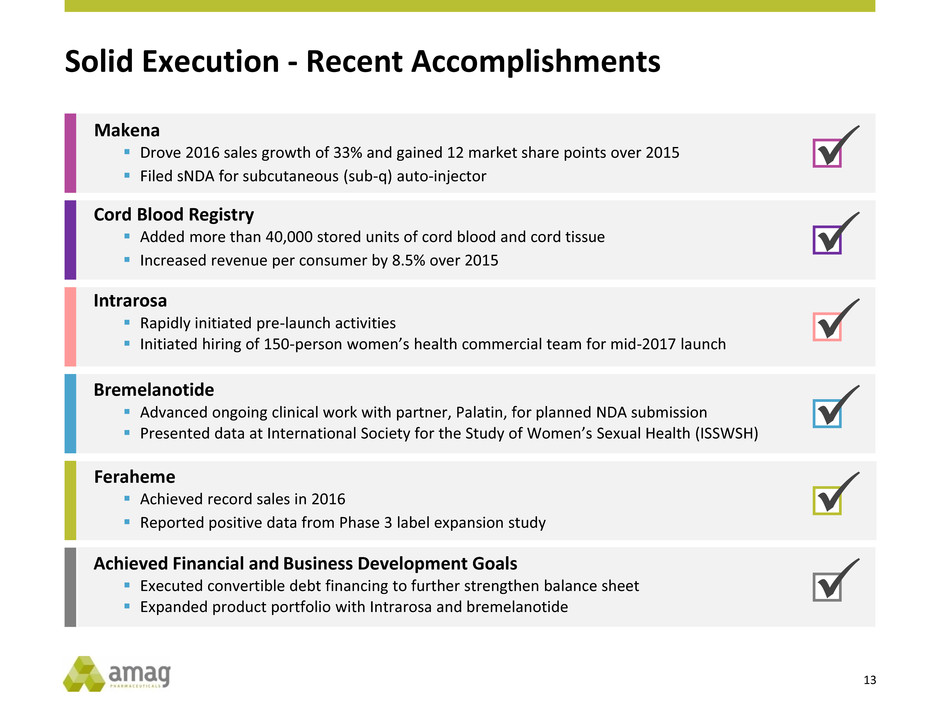
Solid Execution - Recent Accomplishments
Cord Blood Registry
Added more than 40,000 stored units of cord blood and cord tissue
Increased revenue per consumer by 8.5% over 2015
Intrarosa
Rapidly initiated pre-launch activities
Initiated hiring of 150-person women’s health commercial team for mid-2017 launch
Bremelanotide
Advanced ongoing clinical work with partner, Palatin, for planned NDA submission
Presented data at International Society for the Study of Women’s Sexual Health (ISSWSH)
Achieved Financial and Business Development Goals
Executed convertible debt financing to further strengthen balance sheet
Expanded product portfolio with Intrarosa and bremelanotide
Feraheme
Achieved record sales in 2016
Reported positive data from Phase 3 label expansion study
Makena
Drove 2016 sales growth of 33% and gained 12 market share points over 2015
Filed sNDA for subcutaneous (sub-q) auto-injector

14
Agenda
AMAG: Yesterday and Today
AMAG: Tomorrow
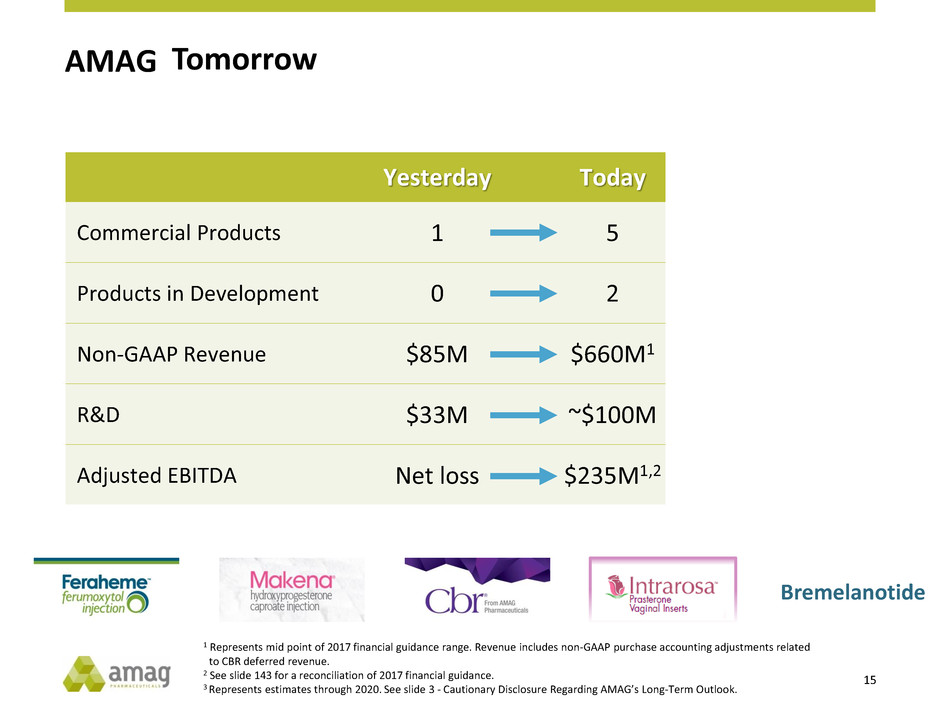
Yesterday Today Tomorrow3
Commercial Products 1 5 7
Products in Development 0 2 3
Non-GAAP Revenue $85M $660M1 >$1B
R&D $33M ~$100M ~$150M
Adjusted EBITDA Net loss $235M1,2 ~$350M
15
AMAG
1 Represents mid point of 2017 financial guidance range. Revenue includes non-GAAP purchase accounting adjustments related
to CBR deferred revenue.
2 See slide 143 for a reconciliation of 2017 financial guidance.
3 Represents estimates through 2020. See slide 3 - Cautionary Disclosure Regarding AMAG’s Long-Term Outlook.
Tomorrow
Bremelanotide
16
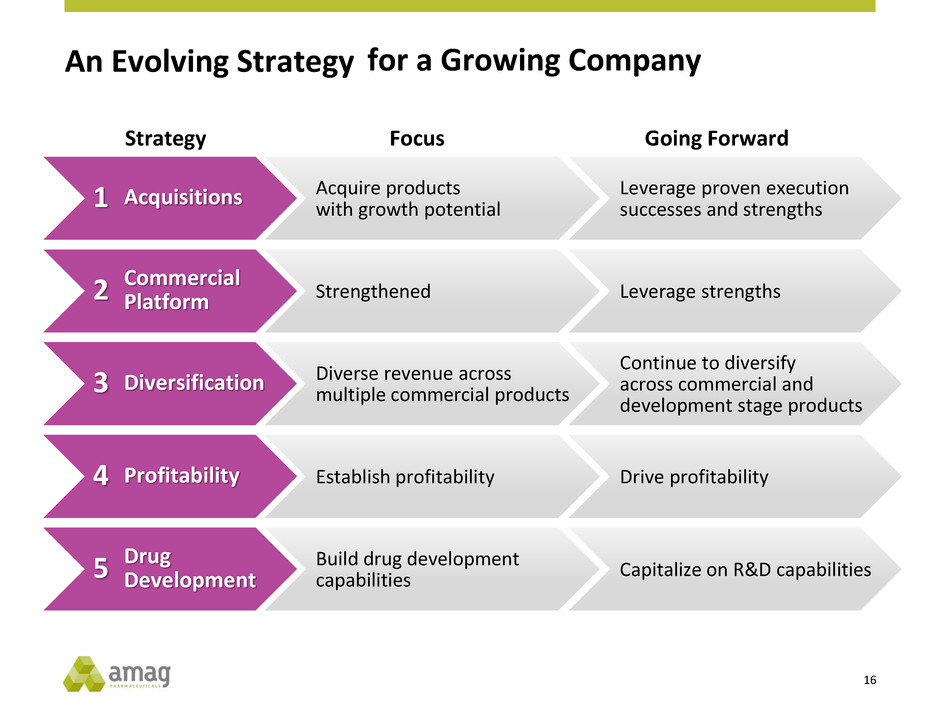
An Evolving Strategy
Acquisitions
Commercial
Platform
Diversification
Profitability
Drug
Development
Acquire products
with growth potential
Strengthened
Diverse revenue across
multiple commercial products
Establish profitability
Build drug development
capabilities
Strategy Focus
1
2
3
4
5
Leverage proven execution
successes and strengths
Leverage strengths
Continue to diversify
across commercial and
development stage products
Drive profitability
Capitalize on R&D capabilities
Going Forward
for a Growing Company

Makena Franchise
2020 Revenue Expectations
18
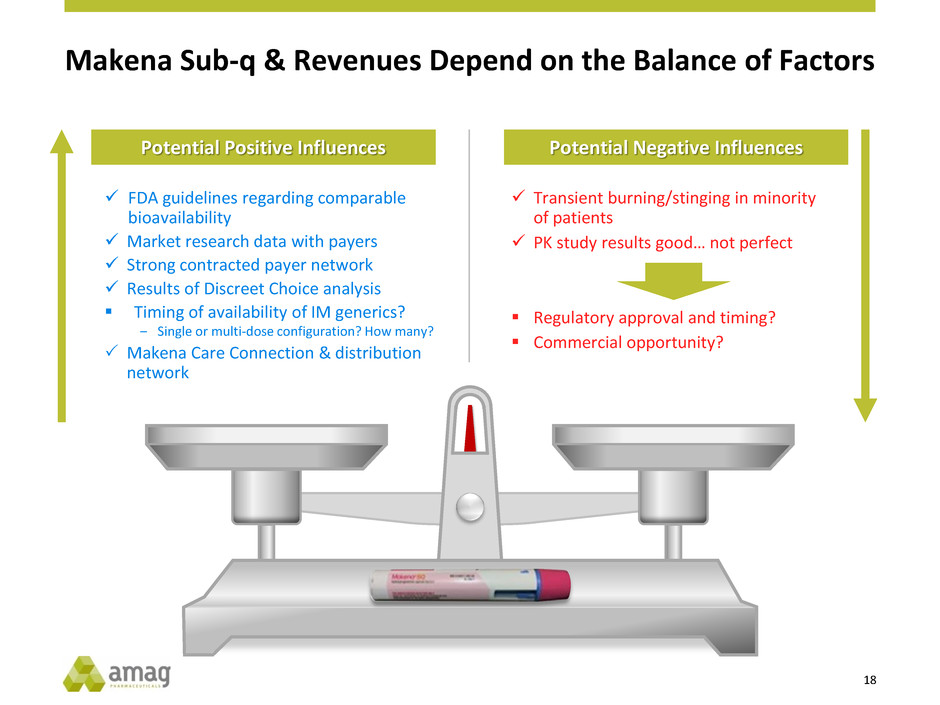
Makena Sub-q & Revenues Depend on the Balance of Factors
FDA guidelines regarding comparable
bioavailability
Market research data with payers
Strong contracted payer network
Results of Discreet Choice analysis
Timing of availability of IM generics?
‒ Single or multi-dose configuration? How many?
Makena Care Connection & distribution
network
Potential Positive Influences Potential Negative Influences
Transient burning/stinging in minority
of patients
PK study results good… not perfect
Regulatory approval and timing?
Commercial opportunity?
19
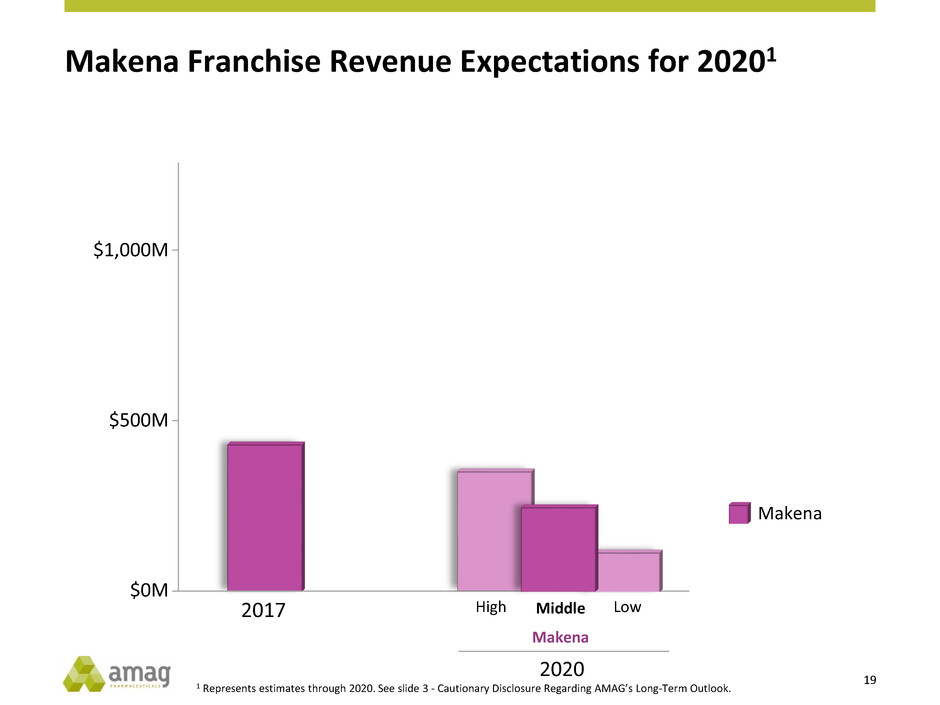
Makena Franchise Revenue Expectations for 20201
$0M
$500M
$1,000M
2017 Low
High Middle
Makena
2020
1 Represents estimates through 2020. See slide 3 - Cautionary Disclosure Regarding AMAG’s Long-Term Outlook.
Makena
High
20
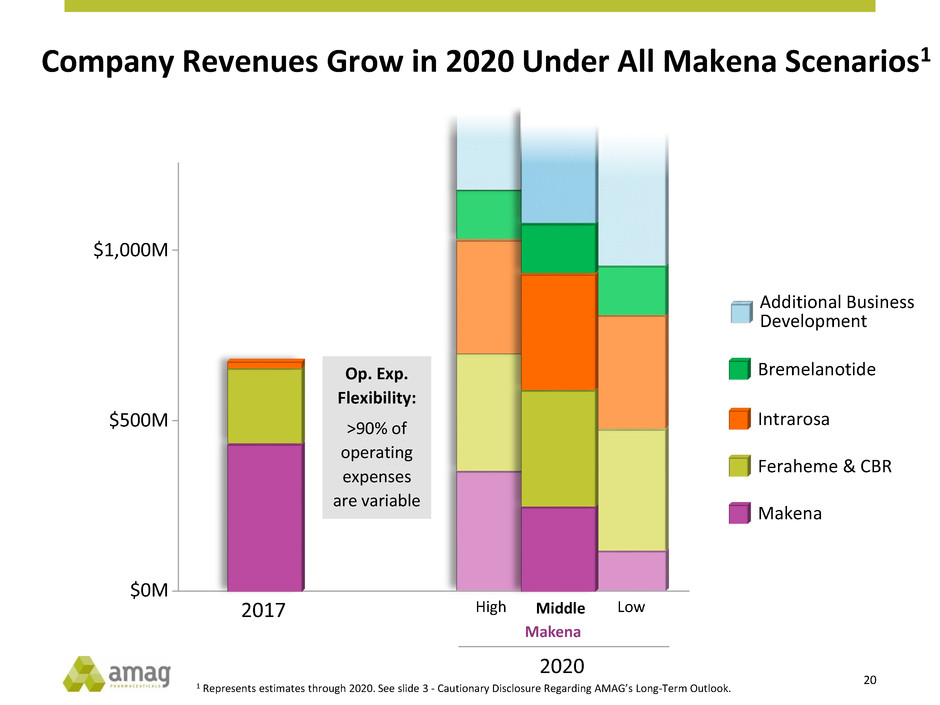
Company Revenues Grow in 2020 Under All Makena Scenarios1
$0M
$500M
$1,000M
2017 Low
Middle
2020
Makena
Op. Exp.
Flexibility:
>90% of
operating
expenses
are variable
Feraheme & CBR
Intrarosa
Bremelanotide
Additional Business
Development
Makena
1 Represents estimates through 2020. See slide 3 - Cautionary Disclosure Regarding AMAG’s Long-Term Outlook.
21
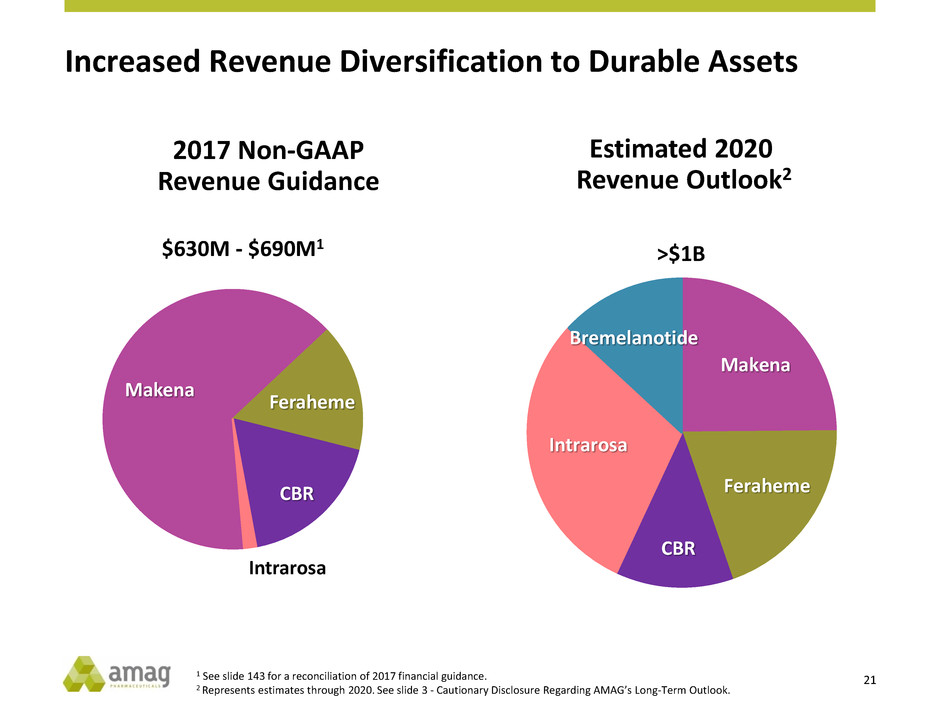
Increased Revenue Diversification to Durable Assets
Makena
Feraheme
CBR
Intrarosa
Bremelanotide
Estimated 2020
Revenue Outlook2
Intrarosa
Makena
Feraheme
CBR
>$1B
2017 Non-GAAP
Revenue Guidance
$630M - $690M1
1 See slide 143 for a reconciliation of 2017 financial guidance.
2 Represents estimates through 2020. See slide 3 - Cautionary Disclosure Regarding AMAG’s Long-Term Outlook.
22
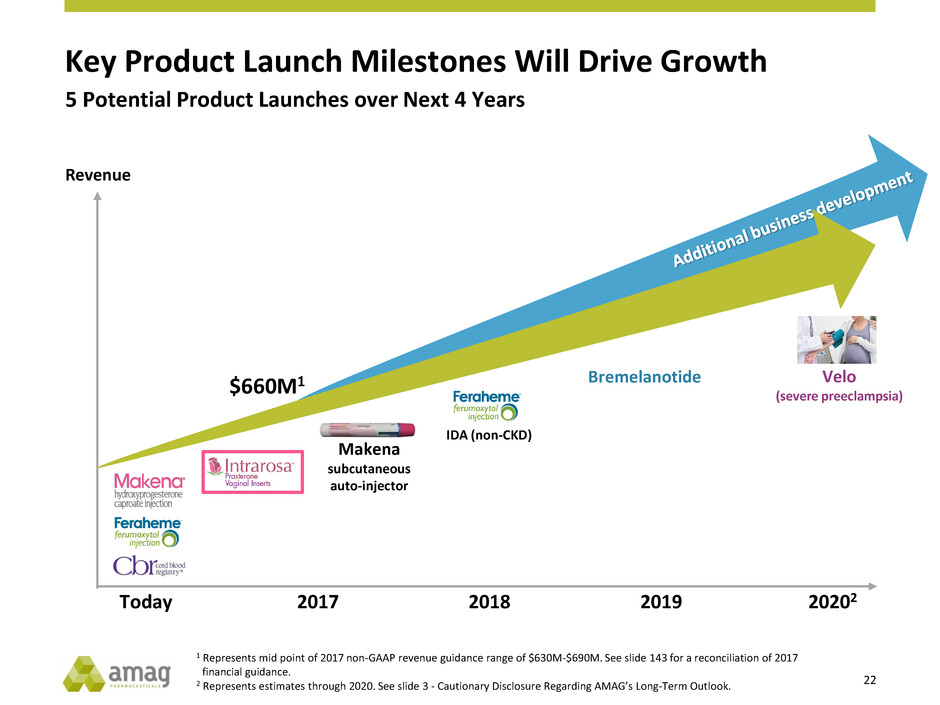
Key Product Launch Milestones Will Drive Growth
Bremelanotide
Makena
subcutaneous
auto-injector
Revenue
Velo
(severe preeclampsia)
Today 2017 2018 2019 20202
$660M1
IDA (non-CKD)
5 Potential Product Launches over Next 4 Years
1 Represents mid point of 2017 non-GAAP revenue guidance range of $630M-$690M. See slide 143 for a reconciliation of 2017
financial guidance.
2 Represents estimates through 2020. See slide 3 - Cautionary Disclosure Regarding AMAG’s Long-Term Outlook.

23
Revenue Potential of >$1 Billion by 20201
Realistic expectations –
with upside
Makena
Expect material positive
impact on revenues
Recent Transactions
Intrarosa
Bremelanotide
Clear growth plans –
long runway
Feraheme
CBR
Clear criteria,
disciplined approach
Business
Development
Revenue >$900 M
More durable assets with
growth potential
Leverage drug
development capabilities
Revenue >$1 billion
More durable assets with
growth potential
Leverage commercialization
and drug development
capabilities
Goals
3 years out
Bremelanotide
1 Represents estimates through 2020. See slide 3 - Cautionary Disclosure Regarding AMAG’s Long-Term Outlook.

Maternal Health:
Makena
Nik Grund
AMAG Chief Commercial Officer

25
Makena Agenda
Continued Strong Growth in 2017
Maximize the Makena Franchise Opportunity
Sub-q Auto-injector Launch Plan
Market Research Supports Acceptance of Sub-q Auto-injector
M A T E R N A L H E A L T H : M A K E N A
26
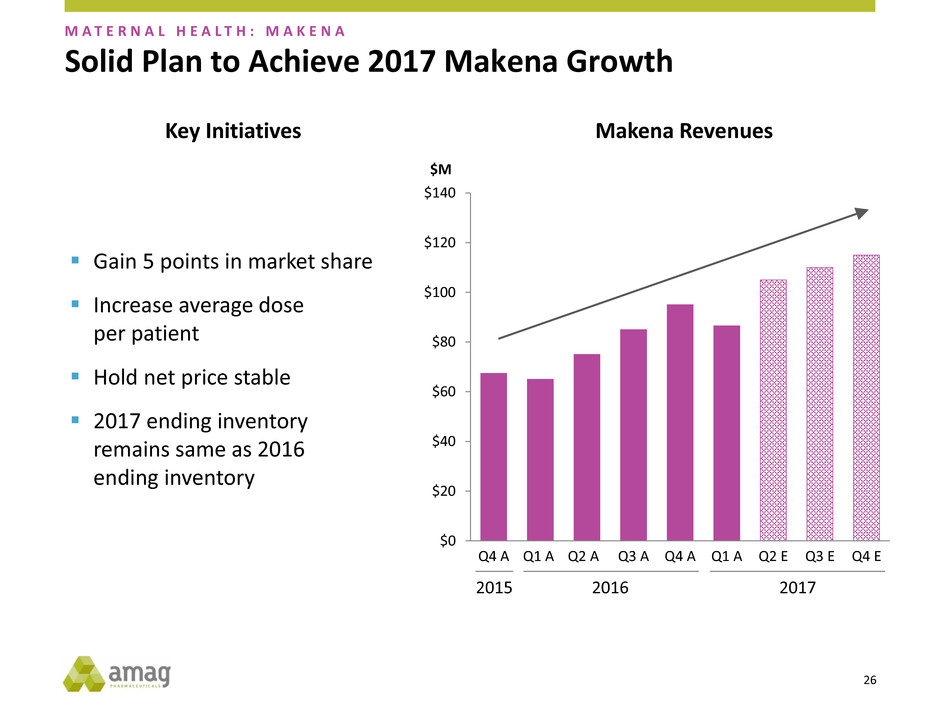
Solid Plan to Achieve 2017 Makena Growth
$0
$20
$40
$60
$80
$100
$120
$140
Q4 A Q1 A Q2 A Q3 A Q4 A Q1 A Q2 E Q3 E Q4 E
M A T E R N A L H E A L T H : M A K E N A
$M
Gain 5 points in market share
Increase average dose
per patient
Hold net price stable
2017 ending inventory
remains same as 2016
ending inventory
Makena Revenues Key Initiatives
2017 2016 2015

27
Makena Agenda
Continued Strong Growth in 2017
Maximize the Makena Franchise Opportunity
Sub-q Auto-injector Launch Plan
Market Research Supports Acceptance of Sub-q Auto-injector
M A T E R N A L H E A L T H : M A K E N A
28
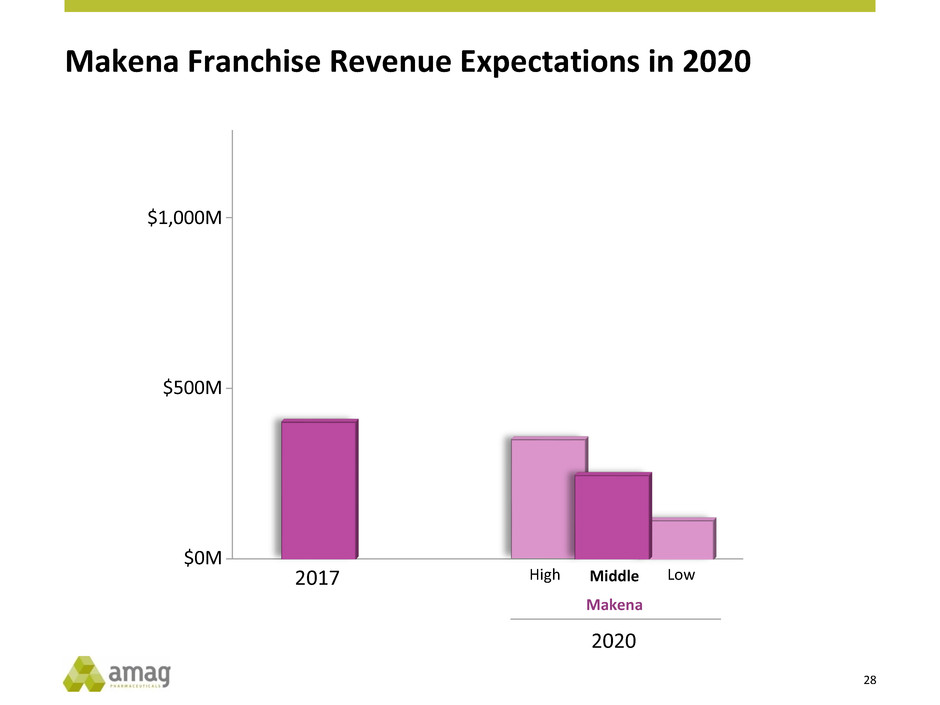
Makena Franchise Revenue Expectations in 2020
$0M
$500M
$1,000M
2017 High Middle
Makena
Low
2020
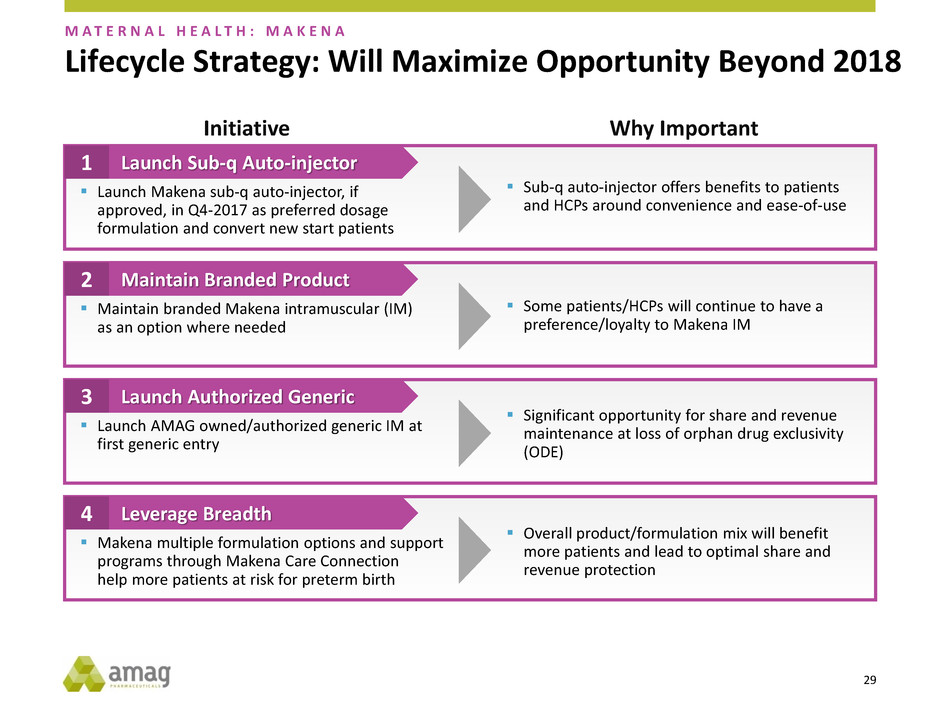
Lifecycle Strategy: Will Maximize Opportunity Beyond 2018
M A T E R N A L H E A L T H : M A K E N A
Initiative Why Important
Launch Sub-q Auto-injector 1
Maintain Branded Product 2
Launch Authorized Generic 3
Leverage Breadth 4
Launch Makena sub-q auto-injector, if
approved, in Q4-2017 as preferred dosage
formulation and convert new start patients
Maintain branded Makena intramuscular (IM)
as an option where needed
Launch AMAG owned/authorized generic IM at
first generic entry
Makena multiple formulation options and support
programs through Makena Care Connection
help more patients at risk for preterm birth
Sub-q auto-injector offers benefits to patients
and HCPs around convenience and ease-of-use
Some patients/HCPs will continue to have a
preference/loyalty to Makena IM
Significant opportunity for share and revenue
maintenance at loss of orphan drug exclusivity
(ODE)
Overall product/formulation mix will benefit
more patients and lead to optimal share and
revenue protection
29

Makena Agenda
Continued Strong Growth in 2017
Maximize the Makena Franchise Opportunity
Sub-q Auto-injector Launch Plan
Market Research Supports Acceptance of Sub-q Auto-injector
M A T E R N A L H E A L T H : M A K E N A
30
31
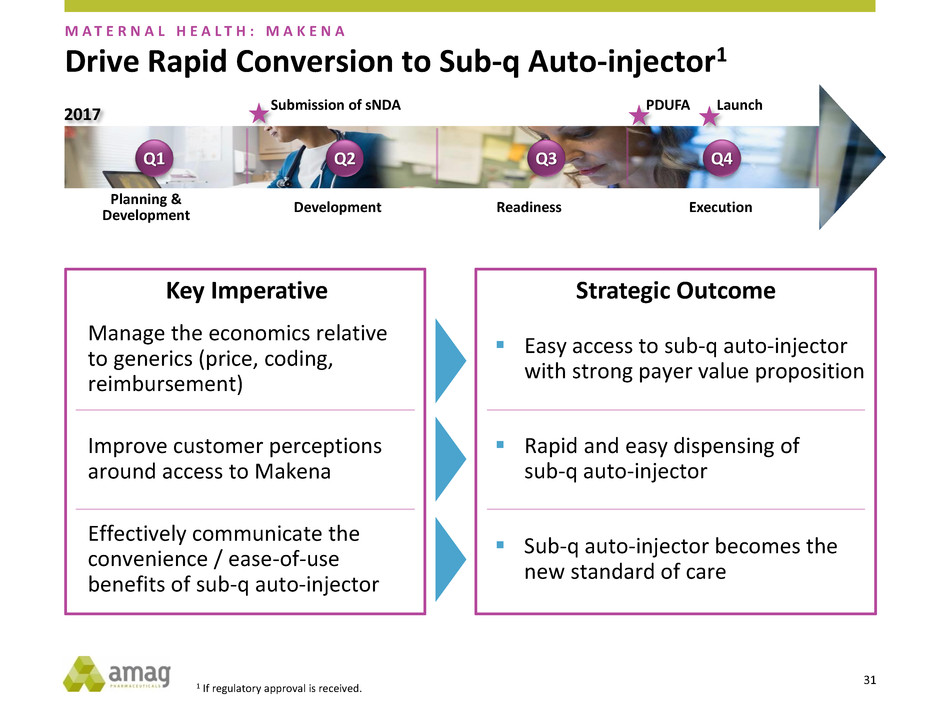
Drive Rapid Conversion to Sub-q Auto-injector1
M A T E R N A L H E A L T H : M A K E N A
Key Imperative
Effectively communicate the
convenience / ease-of-use
benefits of sub-q auto-injector
Manage the economics relative
to generics (price, coding,
reimbursement)
Improve customer perceptions
around access to Makena
Strategic Outcome
Sub-q auto-injector becomes the
new standard of care
Rapid and easy dispensing of
sub-q auto-injector
Easy access to sub-q auto-injector
with strong payer value proposition
Q1 Q2 Q3 Q4
2017
Planning &
Development
Development Readiness Execution
Submission of sNDA PDUFA Launch
1 If regulatory approval is received.

32
Successfully Competing in a Generic Environment
Barrier to Uptake Rationale
Medicaid business
value proposition
M A T E R N A L H E A L T H : M A K E N A
Authorized generic to
compete for exposed
commercial business
Generic formulary status helps share in any leakage
to generic products
Majority of prescriptions
tied to Makena Care
Connection (MCC)
~60% of prescriptions flow through MCC allowing
verification of benefits and physician follow up
Since medicaid is Makena’s most price sensitive and
most deeply discounted business, generics may find
it difficult to price competitively

Makena Agenda
Continued Strong Growth in 2017
Maximize the Makena Franchise Opportunity
Sub-q Auto-injector Launch Plan
Market Research Supports Acceptance of Sub-q Auto-injector
M A T E R N A L H E A L T H : M A K E N A
33

Pre-filled, single-use, ergonomic
device (one-and-done)
Self-contained, small needle
– Sub-q needle: 27 gauge, ½” needle
– IM needle: 21 gauge, 1½” needle
– HPC and patient never see needle
– Prevents inadvertent needle pricks
Once weekly administration
Sub-q in back of arm vs.
IM in buttock
Dosing window shows when
administration is complete
34
Sub-q Auto-injector Offers Improved Profile
IM injection
needle
SQ injection
needle
M A T E R N A L H E A L T H : M A K E N A

Supportive Research on Makena IM versus Makena Sub-q1,2
Unmet need of administration-related attributes around IM
hydroxyprogesterone caproate (HPC)
Makena sub-q auto-injector is perceived to perform better than Makena
IM and HPC: time for administration, needle length, and needle visibility
1
2
M A T E R N A L H E A L T H : M A K E N A
Perception that sub-q will increase compliance 3
Priced similarly to IM, expect major shift to Makena sub-q 4
35
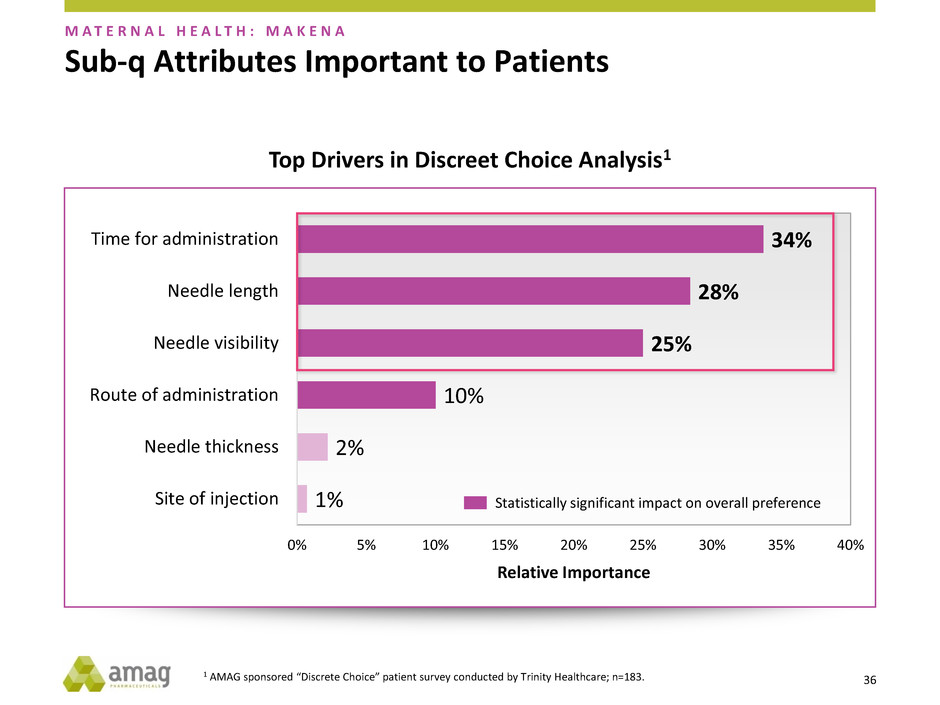
1 AMAG sponsored “Discrete Choice” patient survey conducted by Trinity Healthcare; n=183.
2 AMAG sponsored qualitative research conducted by Thinkgen.
36
1 AMAG sponsored “Discrete Choice” patient survey conducted by Trinity Healthcare; n=183.
Sub-q Attributes Important to Patients
1%
2%
10%
25%
28%
34%
0% 5% 10% 15% 20% 25% 30% 35% 40%
Site of injection
Needle thickness
Route of administration
Needle visibility
Needle length
Time for administration
Relative Importance
Statistically significant impact on overall preference
M A T E R N A L H E A L T H : M A K E N A
Top Drivers in Discreet Choice Analysis1
37
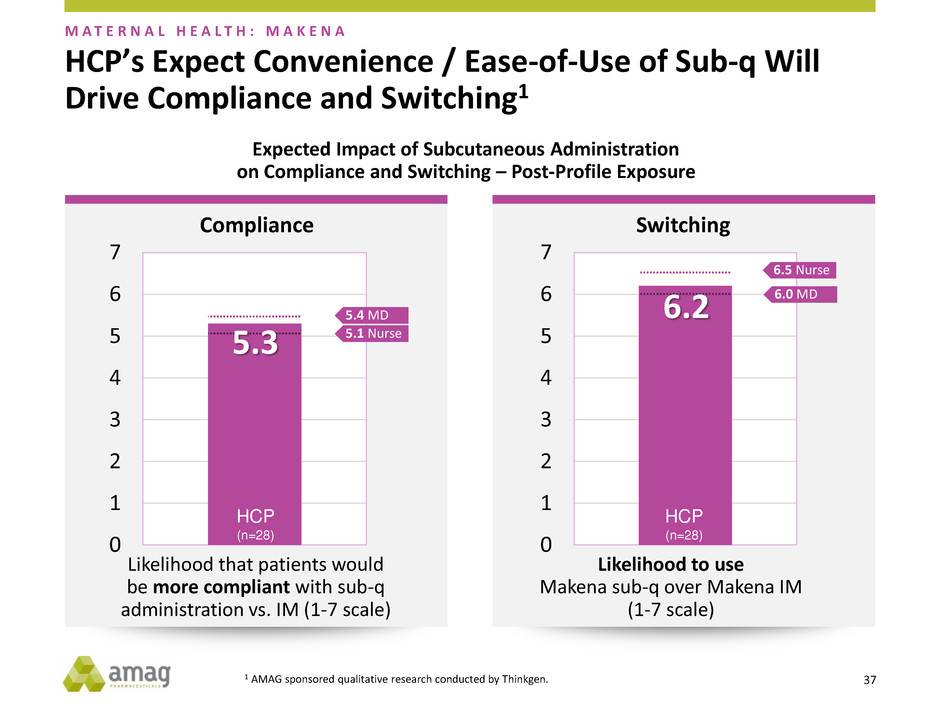
HCP’s Expect Convenience / Ease-of-Use of Sub-q Will
Drive Compliance and Switching1
Expected Impact of Subcutaneous Administration
on Compliance and Switching – Post-Profile Exposure
M A T E R N A L H E A L T H : M A K E N A
Likelihood that patients would
be more compliant with sub-q
administration vs. IM (1-7 scale)
Likelihood to use
Makena sub-q over Makena IM
(1-7 scale)
0
1
2
3
4
5
6
7
5.1 Nurse
5.4 MD
HCP
(n=28)
5.3
0
1
2
3
4
5
6
7
6.5 Nurse
6.0 MD
6.2
HCP
(n=28)
Compliance Switching
1 AMAG sponsored qualitative research conducted by Thinkgen.
38
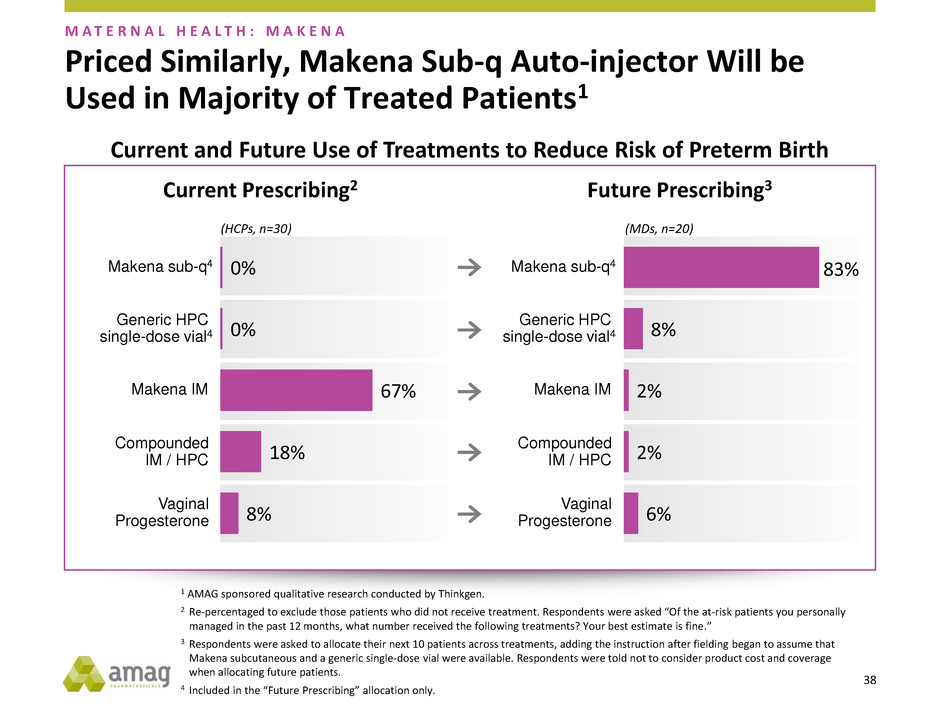
Priced Similarly, Makena Sub-q Auto-injector Will be
Used in Majority of Treated Patients1
M A T E R N A L H E A L T H : M A K E N A
6%
2%
2%
8%
83%
Future Prescribing3
(MDs, n=20)
Current Prescribing2
(HCPs, n=30)
8%
18%
67%
0%
0% Makena sub-q4
Makena IM
Generic HPC
single-dose vial4
Compounded
IM / HPC
Vaginal
Progesterone
Makena sub-q4
Makena IM
Generic HPC
single-dose vial4
Compounded
IM / HPC
Vaginal
Progesterone
Current and Future Use of Treatments to Reduce Risk of Preterm Birth
1 AMAG sponsored qualitative research conducted by Thinkgen.
2 Re-percentaged to exclude those patients who did not receive treatment. Respondents were asked “Of the at-risk patients you personally
managed in the past 12 months, what number received the following treatments? Your best estimate is fine.”
3 Respondents were asked to allocate their next 10 patients across treatments, adding the instruction after fielding began to assume that
Makena subcutaneous and a generic single-dose vial were available. Respondents were told not to consider product cost and coverage
when allocating future patients.
4 Included in the “Future Prescribing” allocation only.

39
Payer Perceptions on Sub-q Auto-injector1
M A T E R N A L H E A L T H : M A K E N A
85%
(n=9)
2%
(n=4) 13%
(n=2)
Perception of
Makena Sub-q Auto-Injector
(n=15, weighted by % commercial and Medicaid lives)
Drivers for “Favorable” Perception:
Perceived ease-of-use given existing self-injection of
Makena IM
Payers expect increased patient comfort
Drivers for “Neutral” and “Unfavorable”
Perception:
Sub-q auto-injector regarded as means of preventing
generic erosion of branded Makena IM
Minimal perceived value or innovation added
to the product
Unfavorable
Neither Favorable nor Unfavorable (Neutral)
Favorable
Substantial coverage cut-off above a 5%-10%
premium to generic HPC; Payer’s are accepting of a
slight premium, driven by the perceived incremental
value of a sub-q auto-injector. “We would pay a modest premium in the range of 5%
to 10%, especially if a sub-q auto-injector incentivizes
patients to go on therapy. But above that, it is not
worth it, and we would not cover the product.”
- Regional HP
1 AMAG sponsored qualitative research conducted by Insight Strategy Advisors.
If physician usage is strong, more likely to
command slight premium

Plan in place to optimize short- and long-term value
– Many possible scenarios, AMAG will be ready
Research indicates sub-q auto-injector provides convenience and ease-
of-use, driving compliance and switching
Payer acceptance with parity pricing should drive conversion to sub-q
sNDA filed in April 2017 with potential approval and launch in Q4-2017
40
Summary: Comprehensive Plan to Maximize Makena
Franchise
M A T E R N A L H E A L T H : M A K E N A

Hematology/Oncology:
Feraheme
Julie Krop, MD
AMAG Chief Medical Officer
Nik Grund
AMAG Chief Commercial Officer

42
Feraheme Agenda
Label Expansion Background
Phase 3 Study Results and Regulatory Timeline
Competitive Landscape
Market Opportunity with Broad Label
Broad Label Launch Strategy
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E
Submitted sNDA to expand label
to include all IDA patients
– Included data from 2 controlled,
multi-center Phase 3 trials with
more the 1,400 patients
– Both studies met the primary
efficacy endpoints related to
improvements in hemoglobin
43
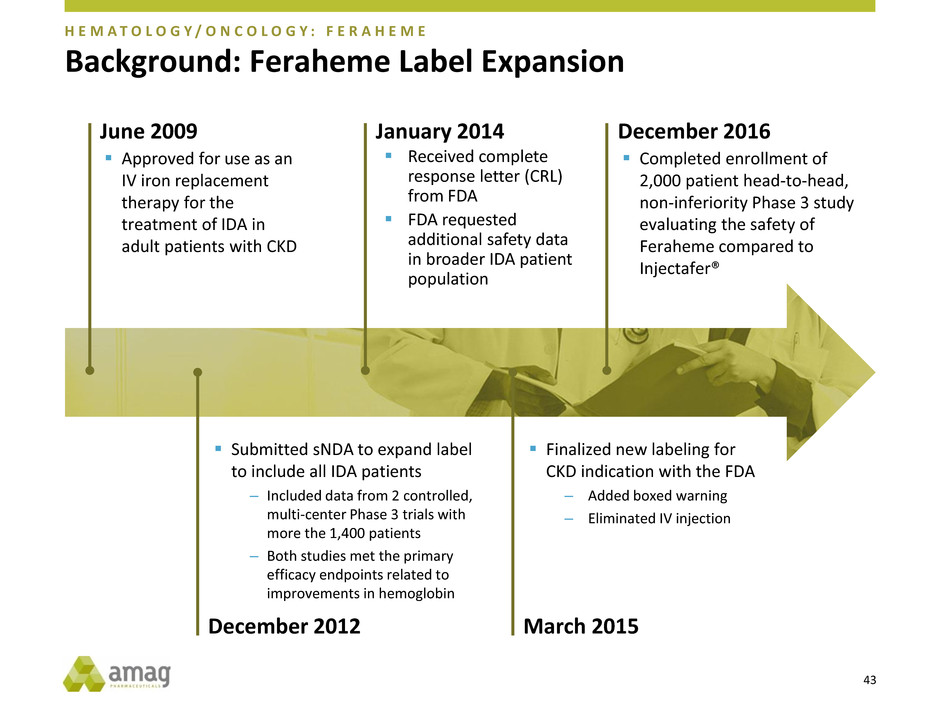
Background: Feraheme Label Expansion
June 2009
Approved for use as an
IV iron replacement
therapy for the
treatment of IDA in
adult patients with CKD
December 2012
January 2014
March 2015
December 2016
Finalized new labeling for
CKD indication with the FDA
– Added boxed warning
– Eliminated IV injection
Completed enrollment of
2,000 patient head-to-head,
non-inferiority Phase 3 study
evaluating the safety of
Feraheme compared to
Injectafer®
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E
Received complete
response letter (CRL)
from FDA
FDA requested
additional safety data
in broader IDA patient
population

44
Large Phase 3 Label Expansion Study
Sample
size
~2,000 patients randomized in a 1:1 ratio
Dosing
regimen
2 doses of Feraheme (1.02g)* or Injectafer® (1.5g)*, dosed 7 days apart
Patient
Population
Subjects with IDA and in whom intravenous iron treatment is indicated
and have failed previous course of oral iron
Primary
endpoint
Incidence of moderate-to-severe hypersensitivity reactions
(including anaphylaxis) and/or moderate-to-severe hypotension
Secondary
endpoints
Incidence of any of the following: moderate-to-severe hypersensitivity
reactions (including anaphylaxis), serious cardiovascular events, death
Mean change in hemoglobin/g of iron delivered at week 5
Mean change in hemoglobin from baseline to week 5
# of sites /
region
~130 sites, global (U.S., Canada, Europe)
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E
* FDA approved dose

45
Rationale for Injectafer as the Comparator
Competitor with a broader label
Similar dosing regimen allows for blinded study
Injectafer requires 50% higher iron dose to achieve
similar rise in hemoglobin levels
Injectafer is fastest growing product in IV iron class
Rationale
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E

46
Feraheme Agenda
Label Expansion Background
Phase 3 Study Results and Regulatory Timeline
Competitive Landscape
Market Opportunity with Broad Label
Broad Label Launch Strategy
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E
47
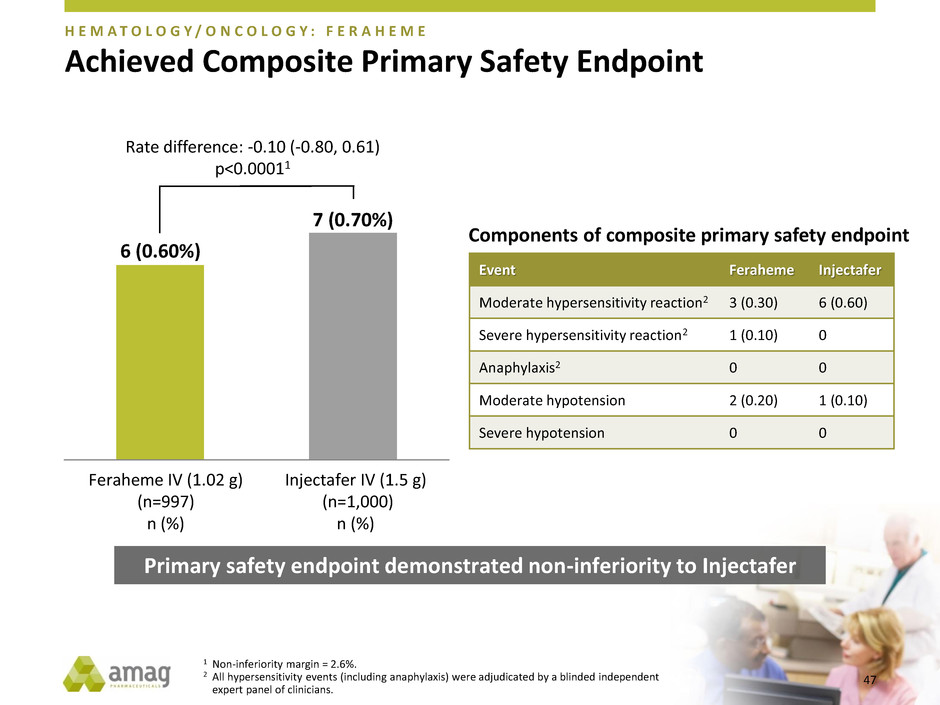
1 Non-inferiority margin = 2.6%.
2 All hypersensitivity events (including anaphylaxis) were adjudicated by a blinded independent
expert panel of clinicians.
Achieved Composite Primary Safety Endpoint
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E
6 (0.60%)
7 (0.70%)
Feraheme IV (1.02 g)
(n=997)
n (%)
Injectafer IV (1.5 g)
(n=1,000)
n (%)
Rate difference: -0.10 (-0.80, 0.61)
p<0.00011
Primary safety endpoint demonstrated non-inferiority to Injectafer
Event Feraheme Injectafer
Moderate hypersensitivity reaction2 3 (0.30) 6 (0.60)
Severe hypersensitivity reaction2 1 (0.10) 0
Anaphylaxis2 0 0
Moderate hypotension 2 (0.20) 1 (0.10)
Severe hypotension 0 0
Components of composite primary safety endpoint
48
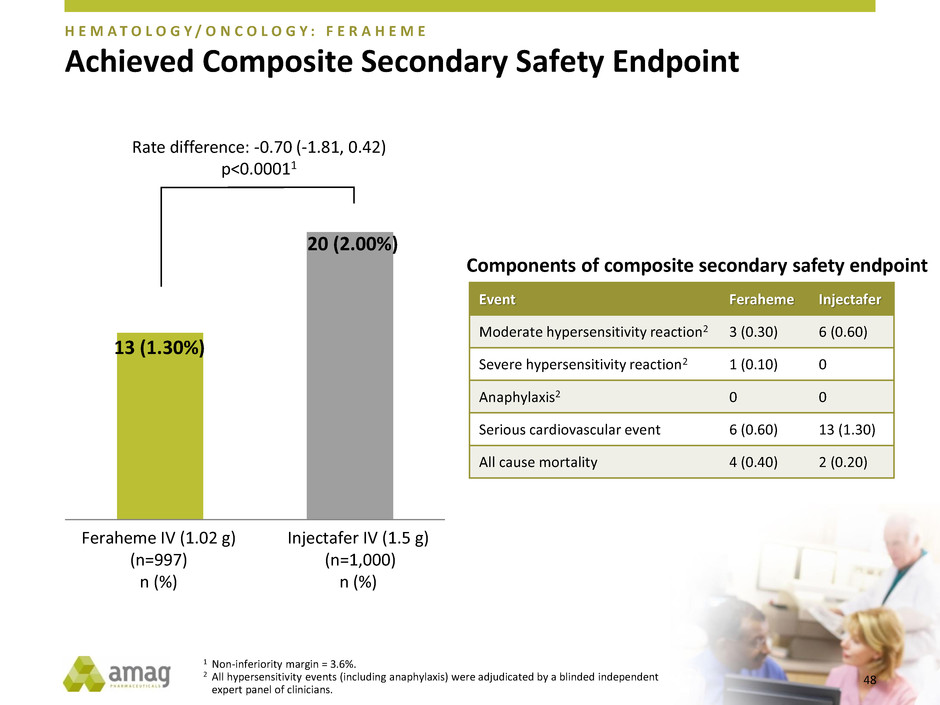
1 Non-inferiority margin = 3.6%.
2 All hypersensitivity events (including anaphylaxis) were adjudicated by a blinded independent
expert panel of clinicians.
Achieved Composite Secondary Safety Endpoint
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E
Feraheme IV (1.02 g)
(n=997)
n (%)
Injectafer IV (1.5 g)
(n=1,000)
n (%)
13 (1.30%)
20 (2.00%)
Rate difference: -0.70 (-1.81, 0.42)
p<0.00011
Event Feraheme Injectafer
Moderate hypersensitivity reaction2 3 (0.30) 6 (0.60)
Severe hypersensitivity reaction2 1 (0.10) 0
Anaphylaxis2 0 0
Serious cardiovascular event 6 (0.60) 13 (1.30)
All cause mortality 4 (0.40) 2 (0.20)
Components of composite secondary safety endpoint

49
1 Blazevic A et al., The Netherlands Journal of Medicine, January 2014, Vol. 72, No. 1.
2 Mani et al., Transplantation: October 15, 2010, Vol. 90, No. 7, pp 804-805.
Higher Incidence of Hypophosphatemia with Injectafer
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E
Moderate
Blood phosphorous <.6 mmol/L
n = 1,997
Severe
Blood phosphorous <.42 mmol/L
n = 1,997
Week 2
Feraheme 0.4% 0%
Injectafer 39% 8%
Week 5
Feraheme 0% 0%
Injectafer 22% 5%
▪ Hypophosphatemia can have important clinical implications
– Acutely: muscle weakness and pain, fatigue
– Chronically: osteomalacia (impaired bone mineralization), bone pain, fractures,
muscle weakness
▪ Majority of cases asymptomatic, but multiple case reports in literature of
symptomatic severe hypophosphatemia1,2
50
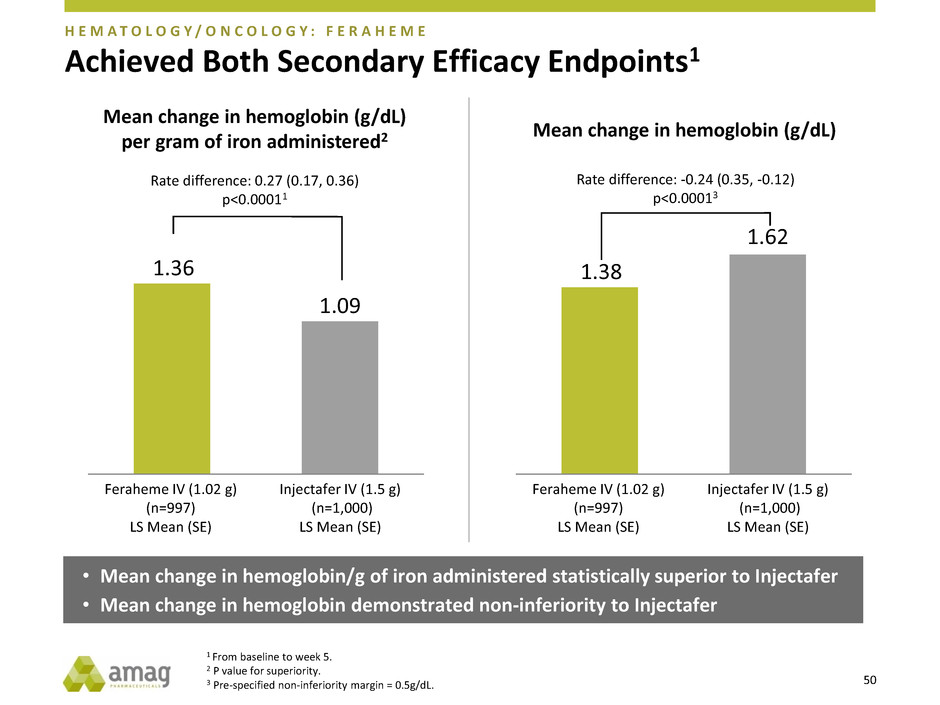
1 From baseline to week 5.
2 P value for superiority.
3 Pre-specified non-inferiority margin = 0.5g/dL.
Achieved Both Secondary Efficacy Endpoints1
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E
• Mean change in hemoglobin/g of iron administered statistically superior to Injectafer
• Mean change in hemoglobin demonstrated non-inferiority to Injectafer
1.36
1.09
Mean change in hemoglobin (g/dL)
per gram of iron administered2
Feraheme IV (1.02 g)
(n=997)
LS Mean (SE)
Injectafer IV (1.5 g)
(n=1,000)
LS Mean (SE)
Rate difference: 0.27 (0.17, 0.36)
p<0.00011
1.38
1.62
Mean change in hemoglobin (g/dL)
Feraheme IV (1.02 g)
(n=997)
LS Mean (SE)
Injectafer IV (1.5 g)
(n=1,000)
LS Mean (SE)
Rate difference: -0.24 (0.35, -0.12)
p<0.00013
2016 2017 2018
51
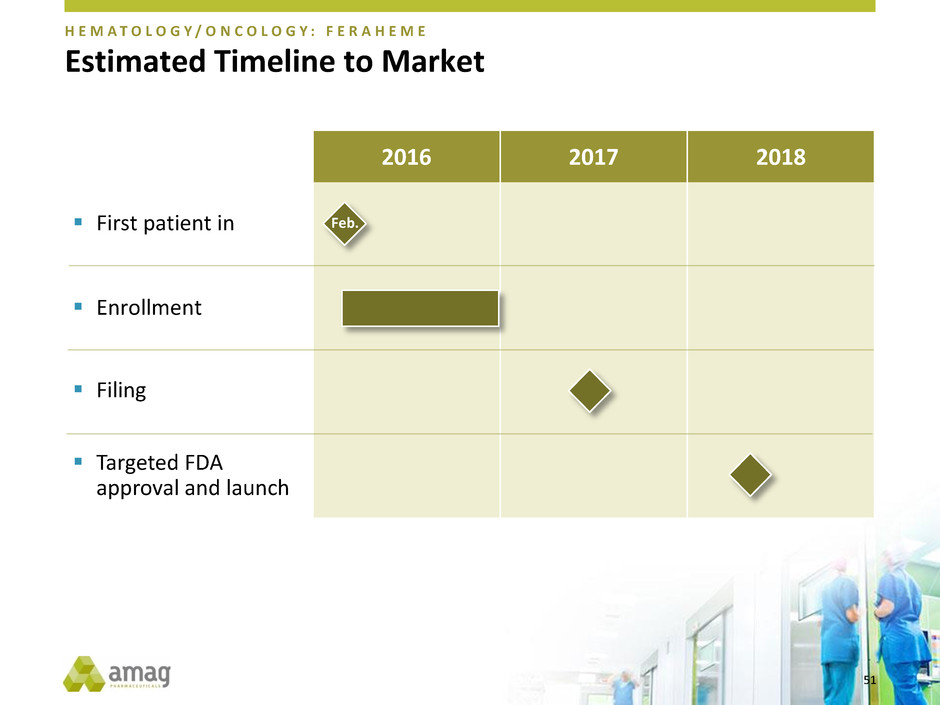
Estimated Timeline to Market
Targeted FDA
approval and launch
First patient in
Enrollment
Filing
Feb.
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E

52
Feraheme Agenda
Label Expansion Background
Phase 3 Study Results and Regulatory Timeline
Competitive Landscape
Market Opportunity with Broad Label
Broad Label Launch Strategy
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E
53
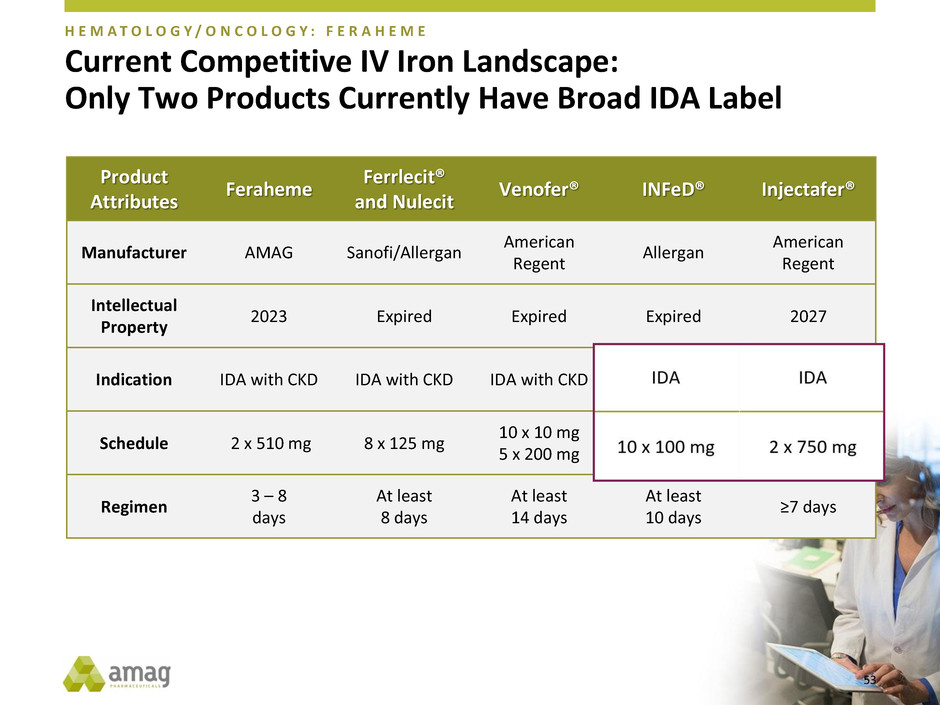
Current Competitive IV Iron Landscape:
Only Two Products Currently Have Broad IDA Label
Product
Attributes
Feraheme
Ferrlecit®
and Nulecit
Venofer® INFeD® Injectafer®
Manufacturer AMAG Sanofi/Allergan
American
Regent
Allergan
American
Regent
Intellectual
Property
2023 Expired Expired Expired 2027
Indication IDA with CKD IDA with CKD IDA with CKD IDA IDA
Schedule 2 x 510 mg 8 x 125 mg
10 x 10 mg
5 x 200 mg
10 x 100 mg 2 x 750 mg
Regimen
3 – 8
days
At least
8 days
At least
14 days
At least
10 days
≥7 days
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E
54
1 Aggregate of Ferrlecit® Brand + generic Sodium Ferric Gluconate.
2 IMS DDD data through week ending 01/02/15.
3 IMS DDD data through week ending 12/30/16.
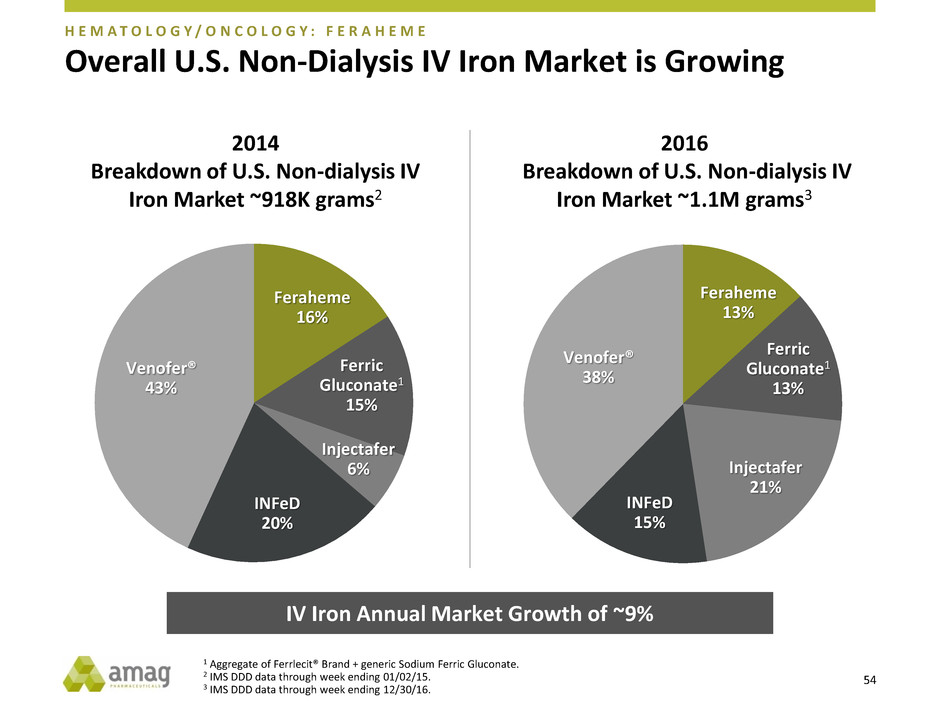
Overall U.S. Non-Dialysis IV Iron Market is Growing
2014
Breakdown of U.S. Non-dialysis IV
Iron Market ~918K grams2
2016
Breakdown of U.S. Non-dialysis IV
Iron Market ~1.1M grams3
IV Iron Annual Market Growth of ~9%
Venofer®
38%
INFeD
15%
Injectafer
21%
Ferric
Gluconate1
13%
Feraheme
13%
Venofer®
43%
INFeD
20%
Injectafer
6%
Ferric
Gluconate1
15%
Feraheme
16%
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E
55
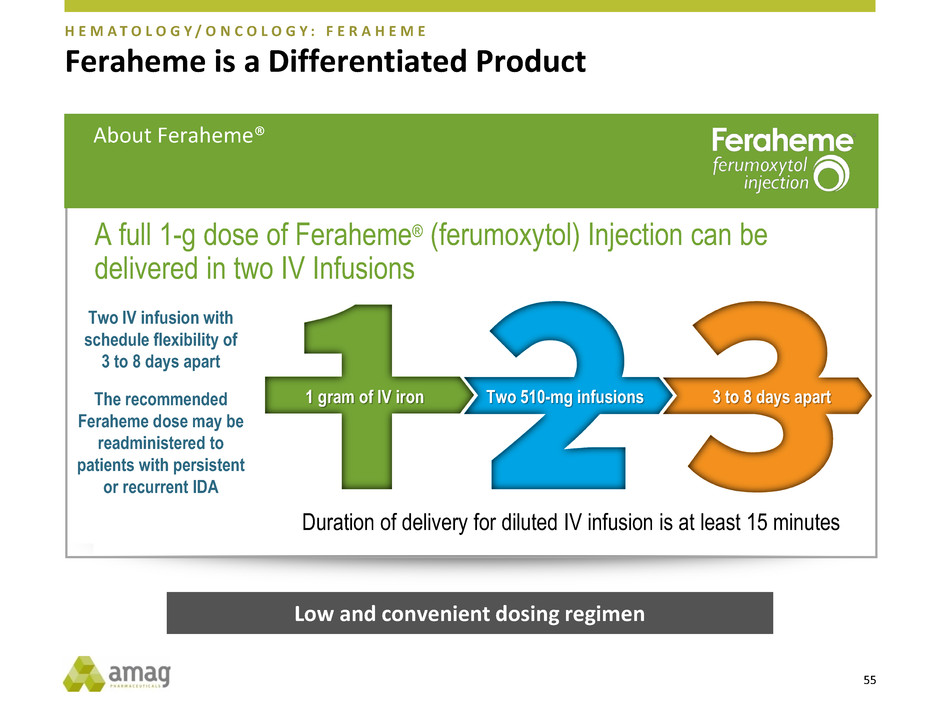
Feraheme is a Differentiated Product
A full 1-g dose of Feraheme® (ferumoxytol) Injection can be
delivered in two IV Infusions
Two IV infusion with
schedule flexibility of
3 to 8 days apart
The recommended
Feraheme dose may be
readministered to
patients with persistent
or recurrent IDA
Duration of delivery for diluted IV infusion is at least 15 minutes
1 gram of IV iron Two 510-mg infusions 3 to 8 days apart
Low and convenient dosing regimen
About Feraheme®
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E

56
Feraheme Agenda
Label Expansion Background
Phase 3 Study Results and Regulatory Timeline
Competitive Landscape
Market Opportunity with Broad Label
Broad Label Launch Strategy
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E
57
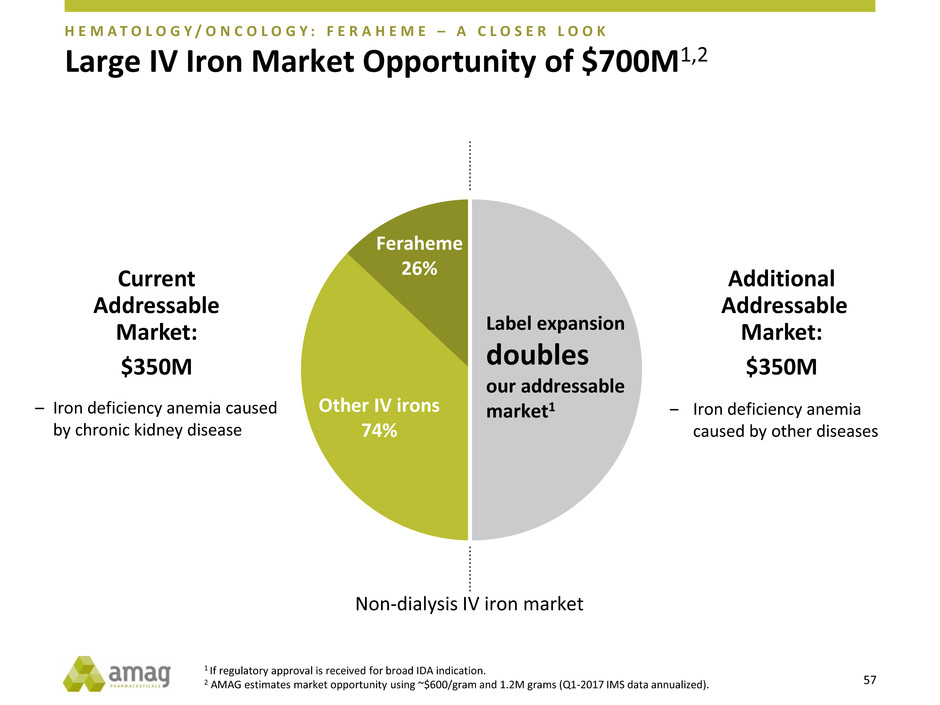
Large IV Iron Market Opportunity of $700M1,2
Feraheme
26%
Other IV irons
74%
Current
Addressable
Market:
$350M
Additional
Addressable
Market:
$350M
‒ Iron deficiency anemia
caused by other diseases
– Iron deficiency anemia caused
by chronic kidney disease
Label expansion
doubles
our addressable
market1
Non-dialysis IV iron market
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E – A C L O S E R L O O K
1 If regulatory approval is received for broad IDA indication.
2 AMAG estimates market opportunity using ~$600/gram and 1.2M grams (Q1-2017 IMS data annualized).
58
1 Global Intravenous (I.V.) Iron Drugs Market Report: 2015 Edition.
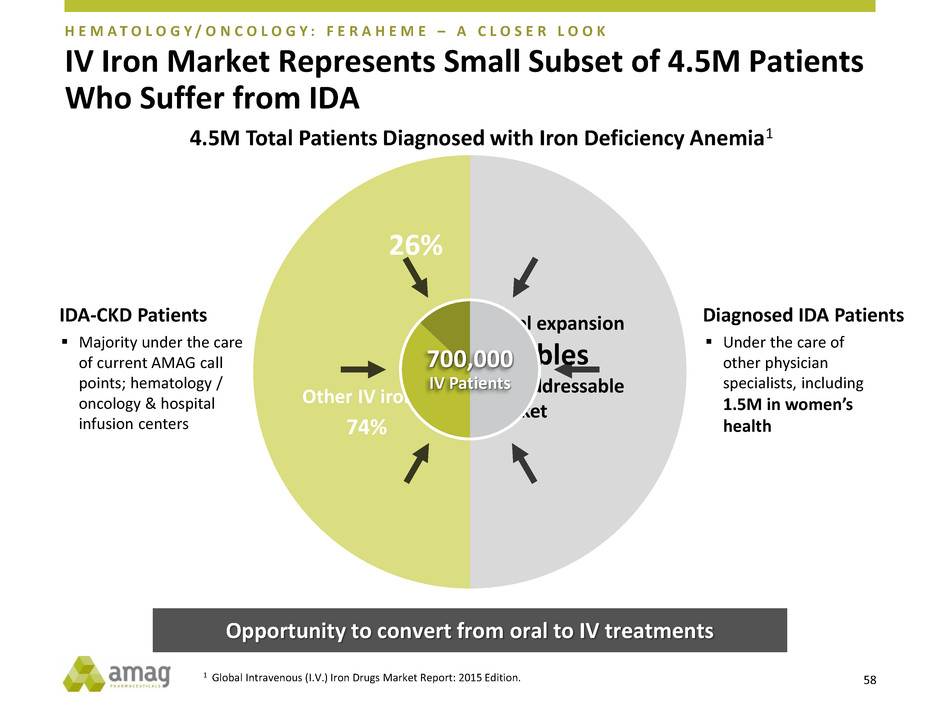
IV Iron Market Represents Small Subset of 4.5M Patients
Who Suffer from IDA
Opportunity to convert from oral to IV treatments
IDA-CKD Patients
Majority under the care
of current AMAG call
points; hematology /
oncology & hospital
infusion centers
Diagnosed IDA Patients
Under the care of
other physician
specialists, including
1.5M in women’s
health
26%
74%
Other IV irons
Label expansion
doubles
our addressable
market
700,000
IV Patients
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E – A C L O S E R L O O K
4.5M Total Patients Diagnosed with Iron Deficiency Anemia1
59
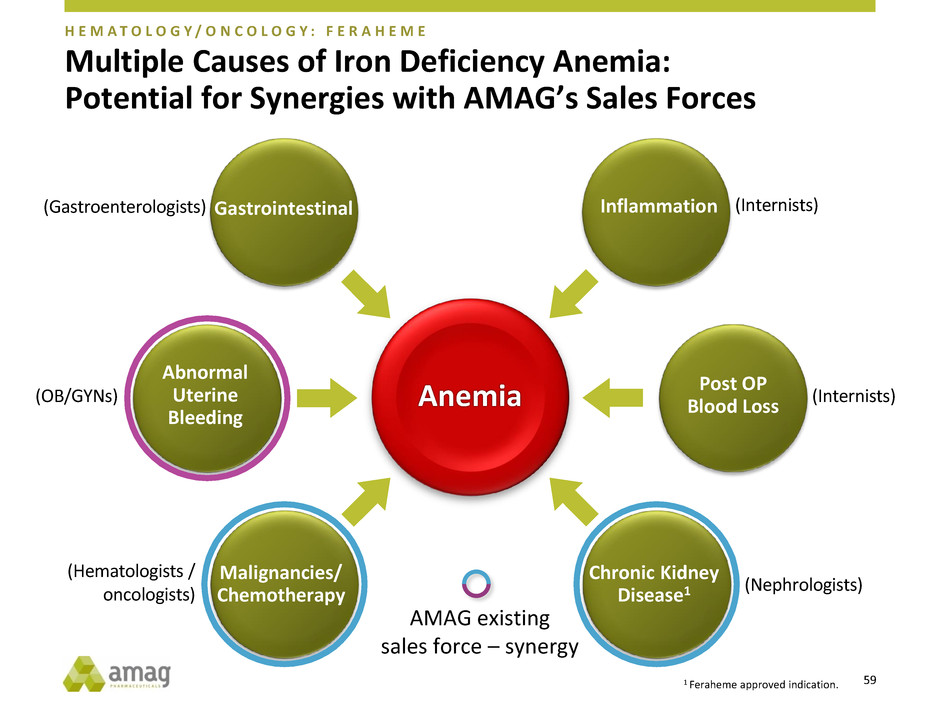
Multiple Causes of Iron Deficiency Anemia:
Potential for Synergies with AMAG’s Sales Forces
Anemia
Gastrointestinal Inflammation
Abnormal
Uterine
Bleeding
Post OP
Blood Loss
Chronic Kidney
Disease1
Malignancies/
Chemotherapy
(Internists)
(Internists)
(Nephrologists)
(Hematologists /
oncologists)
(OB/GYNs)
(Gastroenterologists)
AMAG existing
sales force – synergy
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E
1 Feraheme approved indication.

60
Feraheme Agenda
Label Expansion Background
Phase 3 Study Results and Regulatory Timeline
Competitive Landscape
Market Opportunity with Broad Label
Broad Label Launch Strategy
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E
61
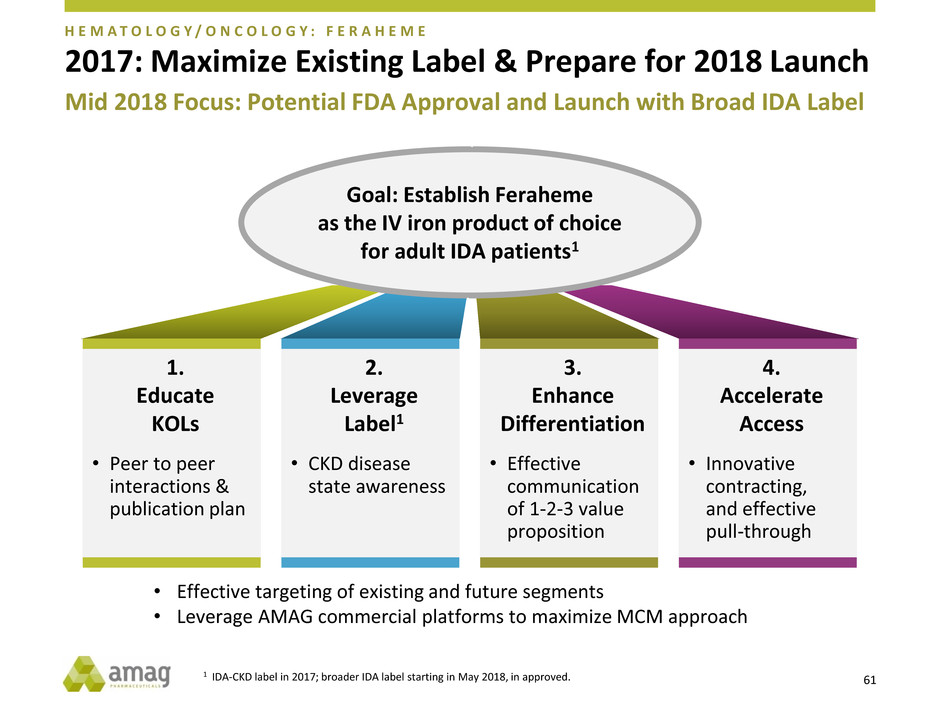
2017: Maximize Existing Label & Prepare for 2018 Launch
• Effective targeting of existing and future segments
• Leverage AMAG commercial platforms to maximize MCM approach
1.
Educate
KOLs
• Peer to peer
interactions &
publication plan
2.
Leverage
Label1
• CKD disease
state awareness
3.
Enhance
Differentiation
• Effective
communication
of 1-2-3 value
proposition
4.
Accelerate
Access
• Innovative
contracting,
and effective
pull-through
Goal: Establish Feraheme
as the IV iron product of choice
for adult IDA patients1
Mid 2018 Focus: Potential FDA Approval and Launch with Broad IDA Label
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E
1 IDA-CKD label in 2017; broader IDA label starting in May 2018, in approved.

62
Beyond 2017 Focus: Maximizing Feraheme Franchise
Opportunity to leverage existing resources for future growth
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E
Current AMAG Hem/Onc sales and strategic accounts
team covers over 90% of total IV iron market
Current AMAG Maternal and Women’s Health teams
covers 25,000 OB/GYNs

Clinical trial showed similar safety and efficacy profile of Feraheme as
compared to Injectafer
Feraheme is a differentiated IV iron replacement therapy
Broad IDA label, if approved, provides opportunity for both indication
expansion and market expansion
63
In Summary: Feraheme Franchise is Strong and Growing
H E M A T O L O G Y / O N C O L O G Y : F E R A H E M E

Maternal Health:
Cord Blood Registry (CBR)
Nik Grund
AMAG Chief Commercial Officer

In 2016, the average age of a first-time mom enrolling with CBR was ~35
~90% of moms having babies today are millennial moms
Adapting messaging
to be relevant to moms
having babies in the
20-34 age range
65
Understanding Today’s Pregnant Moms
M A T E R N A L H E A L T H : C O R D B L O O D R E G I S T R Y
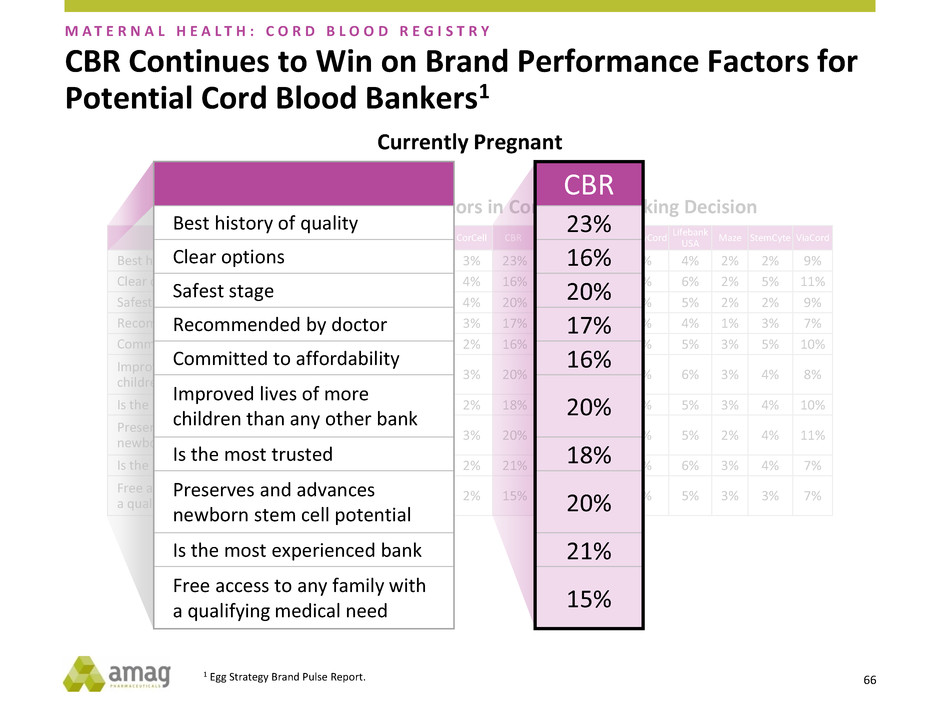
Brand Performance on Key Factors in Cord Blood Banking Decision
Alphacord Americord CariCord CorCell CBR CordUse Cryo-cell FamilyCord
Lifebank
USA
Maze StemCyte ViaCord
Best history of quality 3% 5% 5% 3% 23% 3% 4% 5% 4% 2% 2% 9%
Clear options 3% 6% 7% 4% 16% 2% 3% 5% 6% 2% 5% 11%
Safest storage 3% 5% 4% 4% 20% 3% 3% 4% 5% 2% 2% 9%
Recommended by doctor 4% 5% 5% 3% 17% 3% 4% 6% 4% 1% 3% 7%
Committed to affordability 3% 5% 4% 2% 16% 2% 2% 4% 5% 3% 5% 10%
Improved lives of more
children than any other bank
2% 6% 3% 3% 20% 1% 4% 5% 6% 3% 4% 8%
Is the most trusted 4% 4% 5% 2% 18% 2% 5% 7% 5% 3% 4% 10%
Preserves and advances
newborn stem cell potential
4% 5% 3% 3% 20% 3% 5% 4% 5% 2% 4% 11%
Is the most experienced bank 2% 3% 3% 2% 21% 3% 3% 5% 6% 3% 4% 7%
Free access to any family with
a qualifying medical need
2% 5% 4% 2% 15% 2% 3% 6% 5% 3% 3% 7%
66
CBR Continues to Win on Brand Performance Factors for
Potential Cord Blood Bankers1
M A T E R N A L H E A L T H : C O R D B L O O D R E G I S T R Y
Currently Pregnant
CBR
23%
16
20%
17
16%
20
18%
20%
21%
15%
Best history of quality
Clear options
Safest stage
Recommended by doctor
Committed to affordability
Improved lives of more
children than any other bank
Is the most trusted
Preserves and advances
newborn stem cell potential
Is the most experienced bank
Free access to any family with
a qualifying medical need
1 Egg Strategy Brand Pulse Report.

CBR continues to be the market leader in cord blood and tissue storage
Comprises a business model that provides steady cash flow and high
margin recurring revenue
67
In Summary
M A T E R N A L H E A L T H : C O R D B L O O D R E G I S T R Y
Market research confirms new messaging is
resonating with modern, younger families
(millennials)
Building out direct-to-consumer digital platform
and expertise to be leveraged across AMAG
product portfolio

Q&A
Marketed Products:
• Makena
• Feraheme
• CBR

Women’s Health:
Intrarosa™ (prasterone)
Risa Kagan, MD
University of California, San Francisco,
East Bay Physicians Medical Group Sutter
Medical Foundation
Fernand Labrie, MD
CEO and Founder of Endoceutics
Nik Grund
AMAG Chief Commercial Officer

70
Intrarosa Agenda
Disease Overview
Mechanism of Action (MOA)
Market Size
Competitive Landscape
Market and Pricing Research
Revenue Potential
Commercial Launch Strategy
W O M E N ’ S H E A L T H : I N T R A R O S A

71
Intrarosa Agenda
Disease Overview
Mechanism of Action (MOA)
Market Size
Competitive Landscape
Market and Pricing Research
Revenue Potential
Commercial Launch Strategy
W O M E N ’ S H E A L T H : I N T R A R O S A
72
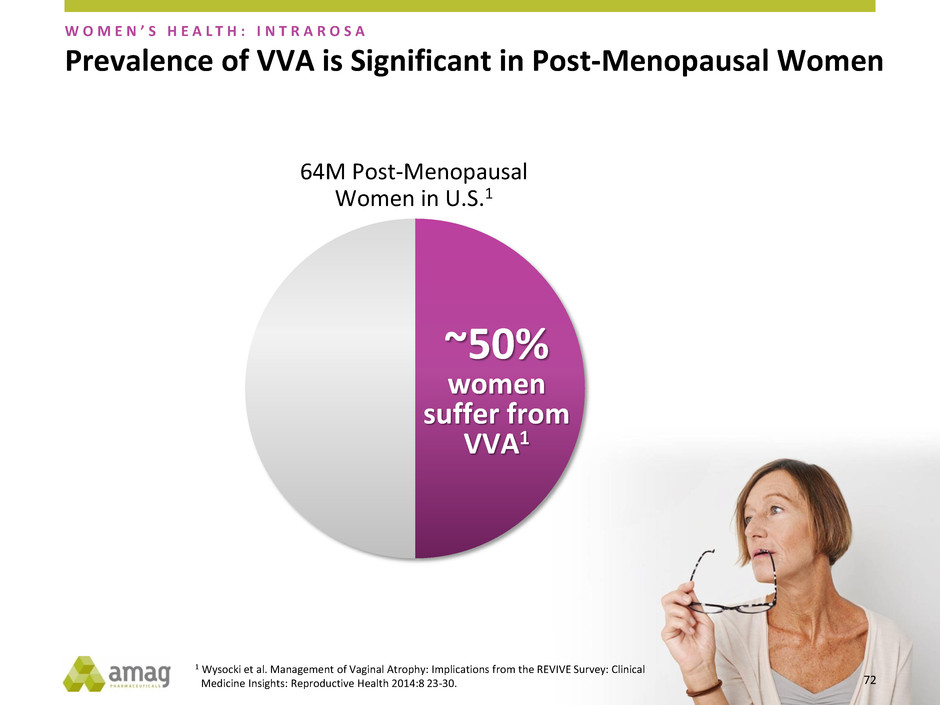
Prevalence of VVA is Significant in Post-Menopausal Women
W O M E N ’ S H E A L T H : I N T R A R O S A
~50%
women
suffer from
VVA1
64M Post-Menopausal
Women in U.S.1
1 Wysocki et al. Management of Vaginal Atrophy: Implications from the REVIVE Survey: Clinical
Medicine Insights: Reproductive Health 2014:8 23-30.
Affected, but not currently seeking treatment
Utilizing OTC treatments
73
1 Based on IMS SMART Tool NSP and NPA data.
2 AMAG estimates based on:
a) Wysocki et al. Management of Vaginal Atrophy: Implications from the REVIVE Survey. Clinical Medicine Insights: Reproductive
Health 2014:8 23–30;
b) Kingsberg et al. Vulvar and Vaginal Atrophy in Postmenopausal Women: Findings from the REVIVE Survey. J Sex Med 2013;101790-1799; and
c) F. Palma et al: Vaginal atrophy of women in postmenopause. Results from a multicentric observational study: The AGATA study.
3 Assumes parity pricing to current therapies (WAC ~$200/month).
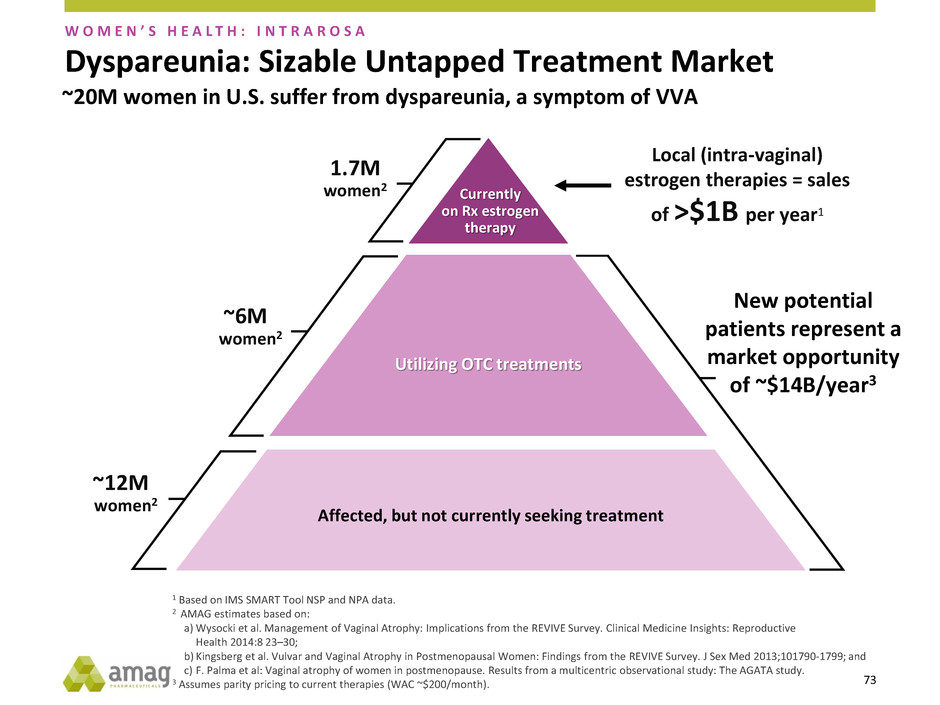
Dyspareunia: Sizable Untapped Treatment Market
Currently
on Rx estrogen
therapy
Local (intra-vaginal)
estrogen therapies = sales
of >$1B per year1
1.7M
women2
~6M
women2
~12M
women2
W O M E N ’ S H E A L T H : I N T R A R O S A
~20M women in U.S. suffer from dyspareunia, a symptom of VVA
New potential
patients represent a
market opportunity
of ~$14B/year3

74
Intrarosa Agenda
Disease Overview
Mechanism of Action (MOA)
Market Size
Competitive Landscape
Market and Pricing Research
Revenue Potential
Commercial Launch Strategy
W O M E N ’ S H E A L T H : I N T R A R O S A

75
Compares Favorably with Existing Intra-vaginal
Treatments in Current $1B Estrogen Therapy Market
Criteria Current Intra-Vaginal Therapies Intrarosa
Administration Vaginal insert or cream by applicator Vaginal insert by applicator
Indication
Approved for various symptoms of VVA,
including dyspareunia, atrophic vaginitis,
dryness, itching and burning
Approved for moderate-
to-severe dyspareunia
(a common symptom of
VVA) due to menopause
Boxed Warning
Class boxed warning regarding increased risk
of cardiovascular disorders, certain cancers
and probable dementia
No boxed warning
Dosing Lowest effective dose for the shortest duration Daily
W O M E N ’ S H E A L T H : I N T R A R O S A
1.7M on
estrogen therapy2
Local (intra-vaginal) estrogen
therapies = sales of >$1B per year1
Safety concerns about estrogen-containing treatment options
1 Based on IMS SMART Tool NSP and NP2008-11A data.
2 AMAG estimates based on:
a) Wysocki et al. Management of Vaginal Atrophy: Implications from the REVIVE Survey. Clinical Medicine Insights: Reproductive Health 2014:8 23–30;
b) Kingsberg et al. Vulvar and Vaginal Atrophy in Postmenopausal Women: Findings from the REVIVE Survey. J Sex Med 2013;101790-1799;
c) F. Palma et al: Vaginal atrophy of women in postmenopause. Results from a multicentric observational study: The AGATA study.
76
1 NSP audit from 4/2011/32017 Growth Dynamics Audit.
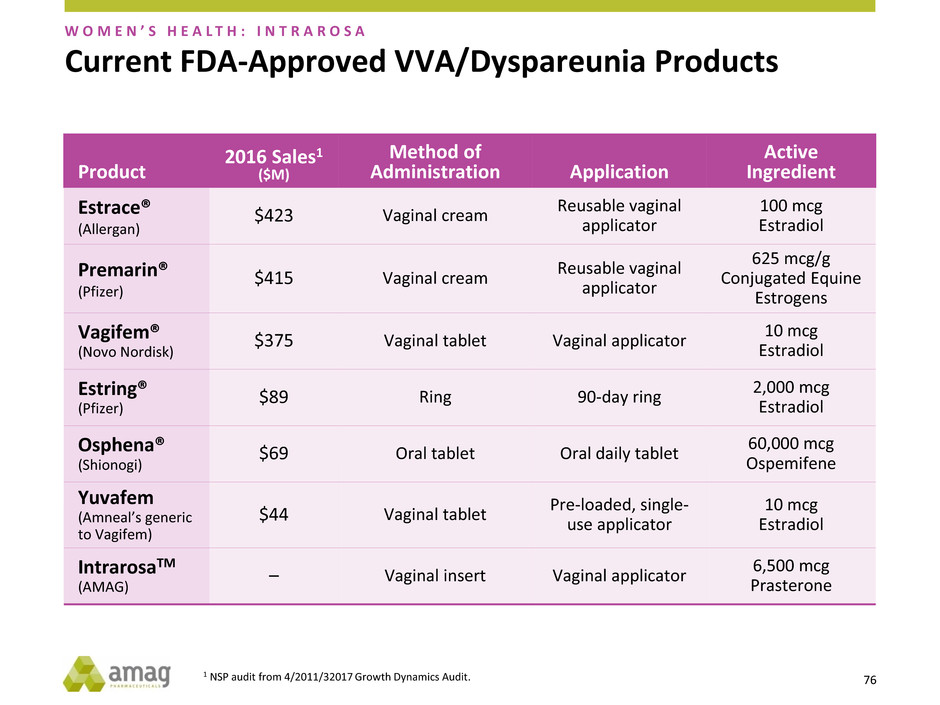
Current FDA-Approved VVA/Dyspareunia Products
W O M E N ’ S H E A L T H : I N T R A R O S A
Product
2016 Sales1
($M)
Method of
Administration Application
Active
Ingredient
Estrace®
(Allergan)
$423 Vaginal cream
Reusable vaginal
applicator
100 mcg
Estradiol
Premarin®
(Pfizer)
$415 Vaginal cream
Reusable vaginal
applicator
625 mcg/g
Conjugated Equine
Estrogens
Vagifem®
(Novo Nordisk)
$375 Vaginal tablet Vaginal applicator
10 mcg
Estradiol
Estring®
(Pfizer)
$89 Ring 90-day ring
2,000 mcg
Estradiol
Osphena®
(Shionogi)
$69 Oral tablet Oral daily tablet
60,000 mcg
Ospemifene
Yuvafem
(Amneal’s generic
to Vagifem)
$44 Vaginal tablet
Pre-loaded, single-
use applicator
10 mcg
Estradiol
IntrarosaTM
(AMAG)
– Vaginal insert Vaginal applicator
6,500 mcg
Prasterone

77
Intrarosa Agenda
Disease Overview
Mechanism of Action (MOA)
Market Size
Competitive Landscape
Market and Pricing Research
Revenue Potential
Commercial Launch Strategy
W O M E N ’ S H E A L T H : I N T R A R O S A

Unique safety
profile
Non-estrogen
treatment2
Differentiated
MOA
78
Research1: Opportunities are Significant to
Differentiate Intrarosa
W O M E N ’ S H E A L T H : I N T R A R O S A
1 Market research sponsored by AMAG and conducted by Roscow Market Research.
2 Locally converted into estrogens and androgens.
79
1 Market research sponsored by AMAG and conducted by Roscow Market Research.
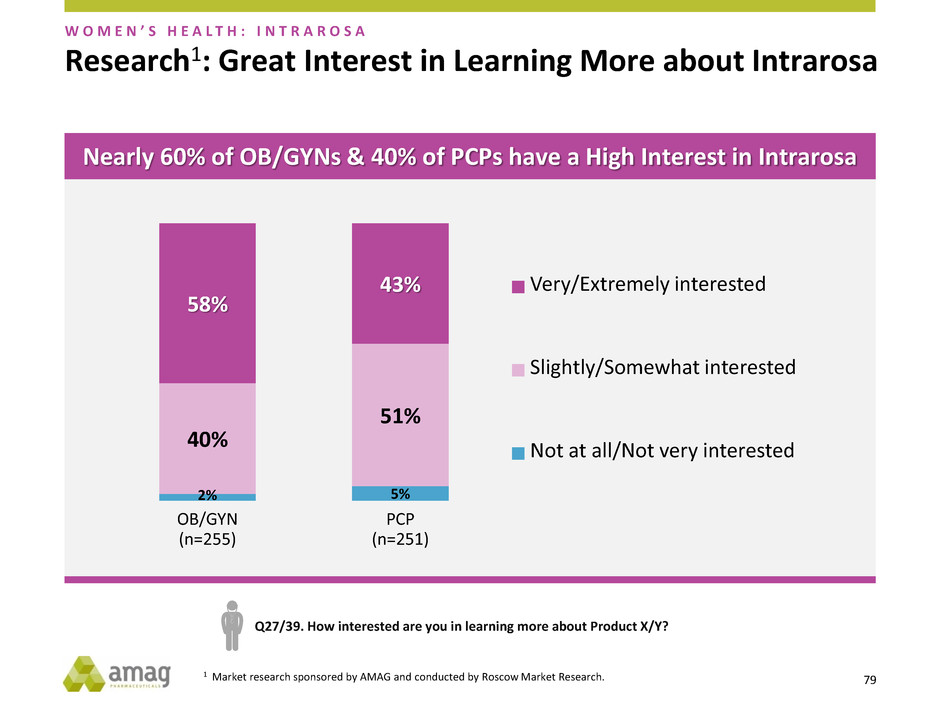
Research1: Great Interest in Learning More about Intrarosa
2% 5%
40%
51%
58%
43%
OB/GYN
(n=255)
PCP
(n=251)
Very/Extremely interested
Slightly/Somewhat interested
Not at all/Not very interested
Q27/39. How interested are you in learning more about Product X/Y?
W O M E N ’ S H E A L T H : I N T R A R O S A
Nearly 60% of OB/GYNs & 40% of PCPs have a High Interest in Intrarosa
80
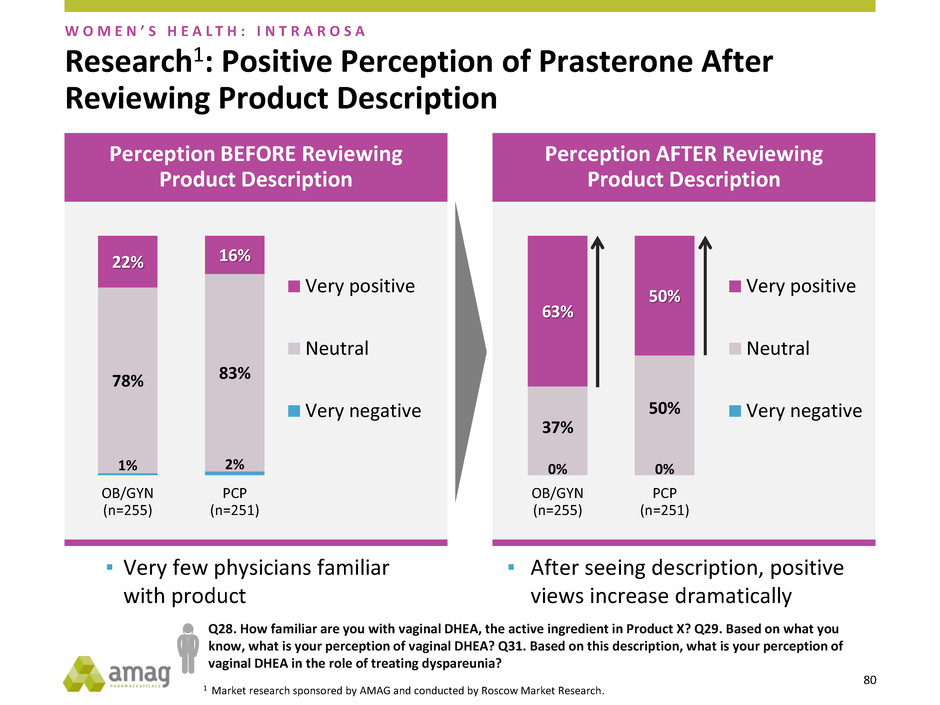
Research1: Positive Perception of Prasterone After
Reviewing Product Description
W O M E N ’ S H E A L T H : I N T R A R O S A
Perception BEFORE Reviewing
Product Description
1% 2%
78% 83%
22% 16%
OB/GYN
(n=255)
PCP
(n=251)
Very positive
Neutral
Very negative
▪ Very few physicians familiar
with product
0% 0%
37%
50%
63%
50%
OB/GYN
(n=255)
PCP
(n=251)
Very positive
Neutral
Very negative
Perception AFTER Reviewing
Product Description
▪ After seeing description, positive
views increase dramatically
1 Market research sponsored by AMAG and conducted by Roscow Market Research.
Q28. How familiar are you with vaginal DHEA, the active ingredient in Product X? Q29. Based on what you
know, what is your perception of vaginal DHEA? Q31. Based on this description, what is your perception of
vaginal DHEA in the role of treating dyspareunia?
81 1 Market research sponsored by AMAG and conducted by Roscow Market Research.
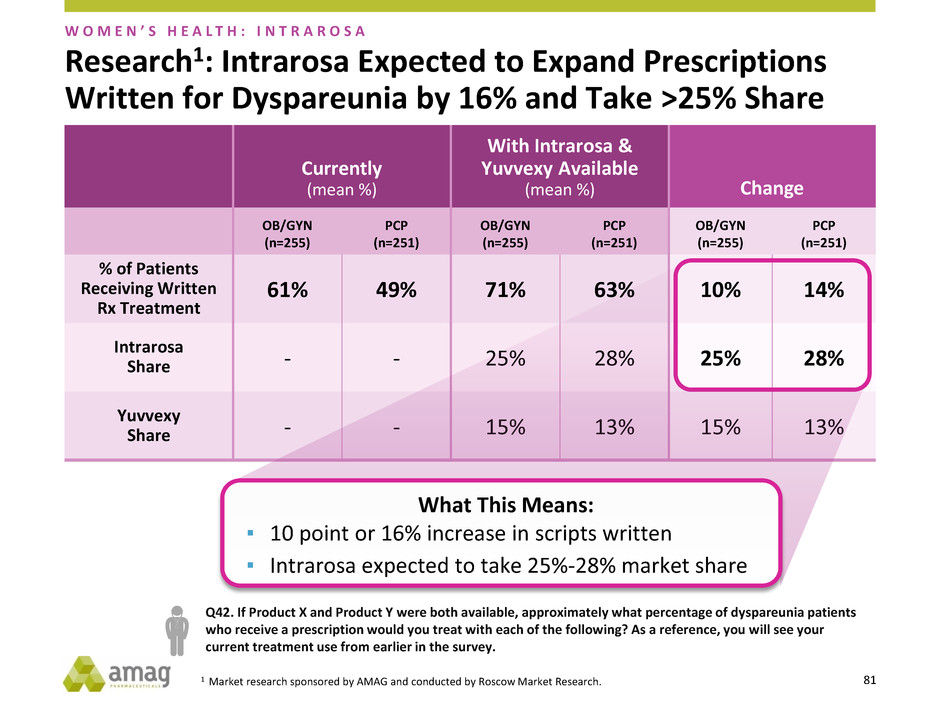
Research1: Intrarosa Expected to Expand Prescriptions
Written for Dyspareunia by 16% and Take >25% Share
W O M E N ’ S H E A L T H : I N T R A R O S A
Currently
(mean %)
With Intrarosa &
Yuvvexy Available
(mean %) Change
OB/GYN
(n=255)
PCP
(n=251)
OB/GYN
(n=255)
PCP
(n=251)
OB/GYN
(n=255)
PCP
(n=251)
% of Patients
Receiving Written
Rx Treatment
61% 49% 71% 63% 10% 14%
Intrarosa
Share - - 25% 28% 25% 28%
Yuvvexy
Share - - 15% 13% 15% 13%
What This Means:
▪ 10 point or 16% increase in scripts written
▪ Intrarosa expected to take 25%-28% market share
Q42. If Product X and Product Y were both available, approximately what percentage of dyspareunia patients
who receive a prescription would you treat with each of the following? As a reference, you will see your
current treatment use from earlier in the survey.

82
Intrarosa Agenda
Disease Overview
Mechanism of Action (MOA)
Market Size
Competitive Landscape
Market and Pricing Research
Revenue Potential
Commercial Launch Strategy
W O M E N ’ S H E A L T H : I N T R A R O S A
83
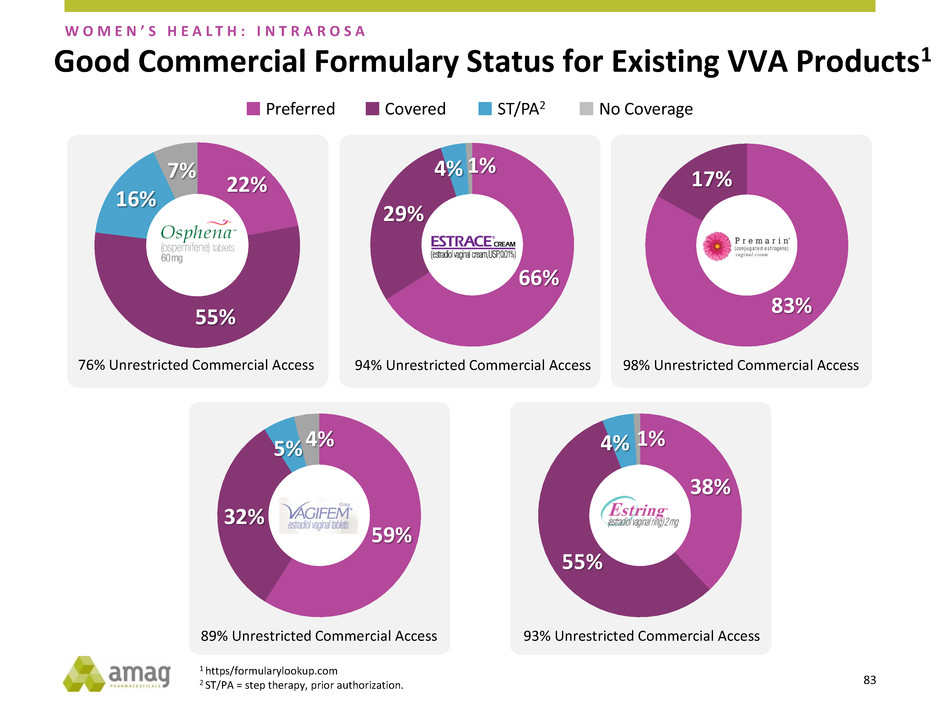
Good Commercial Formulary Status for Existing VVA Products1
W O M E N ’ S H E A L T H : I N T R A R O S A
76% Unrestricted Commercial Access 94% Unrestricted Commercial Access 98% Unrestricted Commercial Access
89% Unrestricted Commercial Access 93% Unrestricted Commercial Access
Preferred Covered ST/PA2 No Coverage
22%
55%
16%
7%
66%
29%
4% 1% 17%
83%
59%
32%
5% 4%
38%
55%
4% 1%
1 https/formularylookup.com
2 ST/PA = step therapy, prior authorization.
84
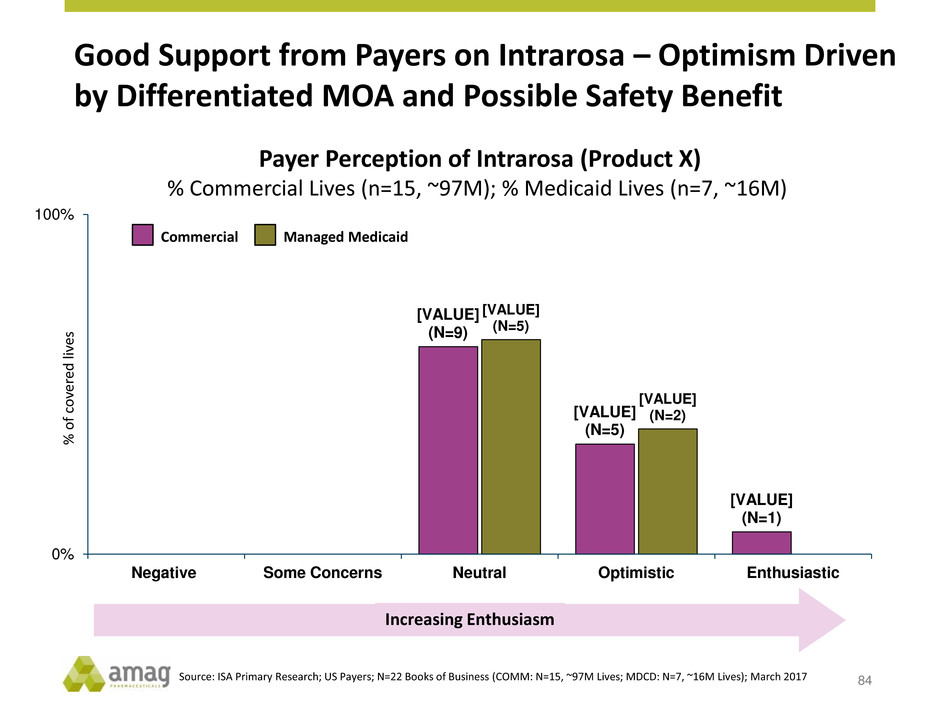
Good Support from Payers on Intrarosa – Optimism Driven
by Differentiated MOA and Possible Safety Benefit
[VALUE]
(N=9)
[VALUE]
(N=5)
[VALUE]
(N=1)
[VALUE]
(N=5)
[VALUE]
(N=2)
0%
100%
Negative Some Concerns Neutral Optimistic Enthusiastic
%
o
f
co
ve
re
d
li
ve
s
Managed Medicaid Commercial
Increasing Enthusiasm
Payer Perception of Intrarosa (Product X)
% Commercial Lives (n=15, ~97M); % Medicaid Lives (n=7, ~16M)
Source: ISA Primary Research; US Payers; N=22 Books of Business (COMM: N=15, ~97M Lives; MDCD: N=7, ~16M Lives); March 2017
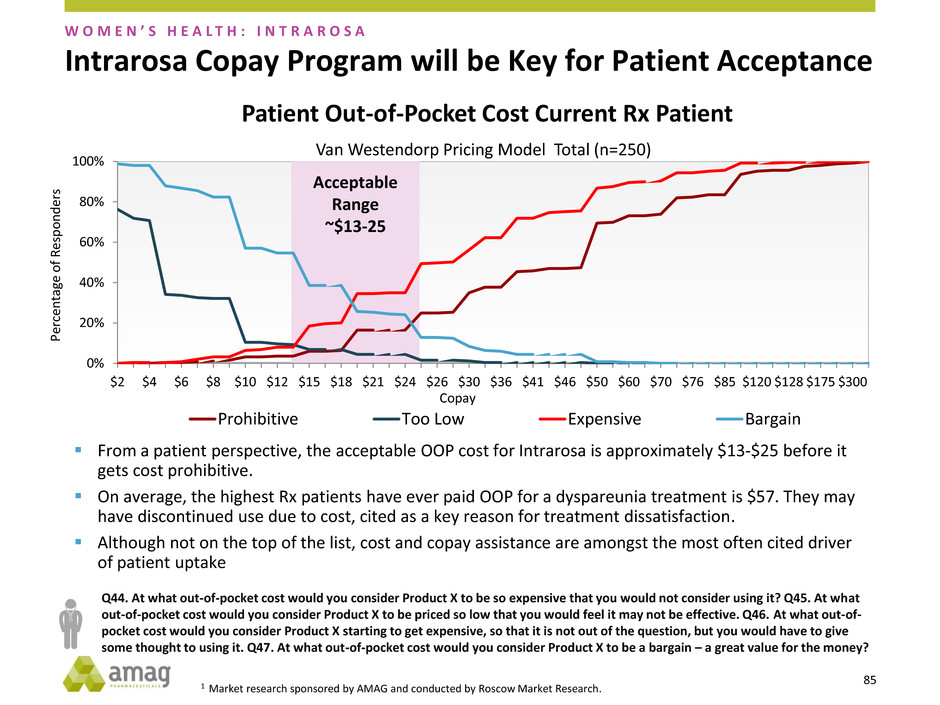
Acceptable
Range
~$13-25
85
Intrarosa Copay Program will be Key for Patient Acceptance
W O M E N ’ S H E A L T H : I N T R A R O S A
0%
20%
40%
60%
80%
100%
$2 $4 $6 $8 $10 $12 $15 $18 $21 $24 $26 $30 $36 $41 $46 $50 $60 $70 $76 $85 $120 $128 $175 $300
Van Westendorp Pricing Model Total (n=250)
Prohibitive Too Low Expensive Bargain
Patient Out-of-Pocket Cost Current Rx Patient
From a patient perspective, the acceptable OOP cost for Intrarosa is approximately $13-$25 before it
gets cost prohibitive.
On average, the highest Rx patients have ever paid OOP for a dyspareunia treatment is $57. They may
have discontinued use due to cost, cited as a key reason for treatment dissatisfaction.
Although not on the top of the list, cost and copay assistance are amongst the most often cited driver
of patient uptake
P
erc
e
n
tage
o
f
Resp
o
n
d
er
s
1 Market research sponsored by AMAG and conducted by Roscow Market Research.
Q44. At what out-of-pocket cost would you consider Product X to be so expensive that you would not consider using it? Q45. At what
out-of-pocket cost would you consider Product X to be priced so low that you would feel it may not be effective. Q46. At what out-of-
pocket cost would you consider Product X starting to get expensive, so that it is not out of the question, but you would have to give
some thought to using it. Q47. At what out-of-pocket cost would you consider Product X to be a bargain – a great value for the money?
Copay

86
Market Research1 – Key Takeaways
High level of physician interest to prescribe Intrarosa and treat
more patients 1
Low degree of payer management and broad access enjoyed
by all therapies 2
W O M E N ’ S H E A L T H : I N T R A R O S A
1 Market research sponsored by AMAG and conducted by Roscow Market Research.
Largest driver of access is patient out-of-pocket; patient assistance
programs important competitive component 3

Affected, but not currently seeking treatment
Utilizing OTC treatments
87
1 Based on IMS SMART Tool NSP and NPA data.
2 AMAG estimates based on:
a) Wysocki et al. Management of Vaginal Atrophy: Implications from the REVIVE Survey. Clinical Medicine Insights: Reproductive
Health 2014:8 23–30;
b) Kingsberg et al. Vulvar and Vaginal Atrophy in Postmenopausal Women: Findings from the REVIVE Survey. J Sex Med 2013;101790-1799;
c) F. Palma et al: Vaginal atrophy of women in postmenopause. Results from a multicentric observational study: The AGATA study.
3 Assumes parity pricing to current therapies (WAC ~$200/month).
Dyspareunia: Sizable Untapped Treatment Market
Currently
on Rx estrogen
therapy
Local (intra-vaginal)
estrogen therapies = sales
of >$1B per year1
1.7M
women2
~6M
women2
~12M
women2
W O M E N ’ S H E A L T H : I N T R A R O S A
~20M women in U.S. suffer from dyspareunia, a symptom of VVA
New potential
patients represent a
market opportunity
of ~$14B/year3
Prasterone
Vaginal Inserts
Intent-to-presc ibe market
research indicates sales potential:
>$500M/year

88
Intrarosa Agenda
Disease Overview
Mechanism of Action (MOA)
Market Size
Competitive Landscape
Market and Pricing Research
Revenue Potential
Commercial Launch Strategy
W O M E N ’ S H E A L T H : I N T R A R O S A
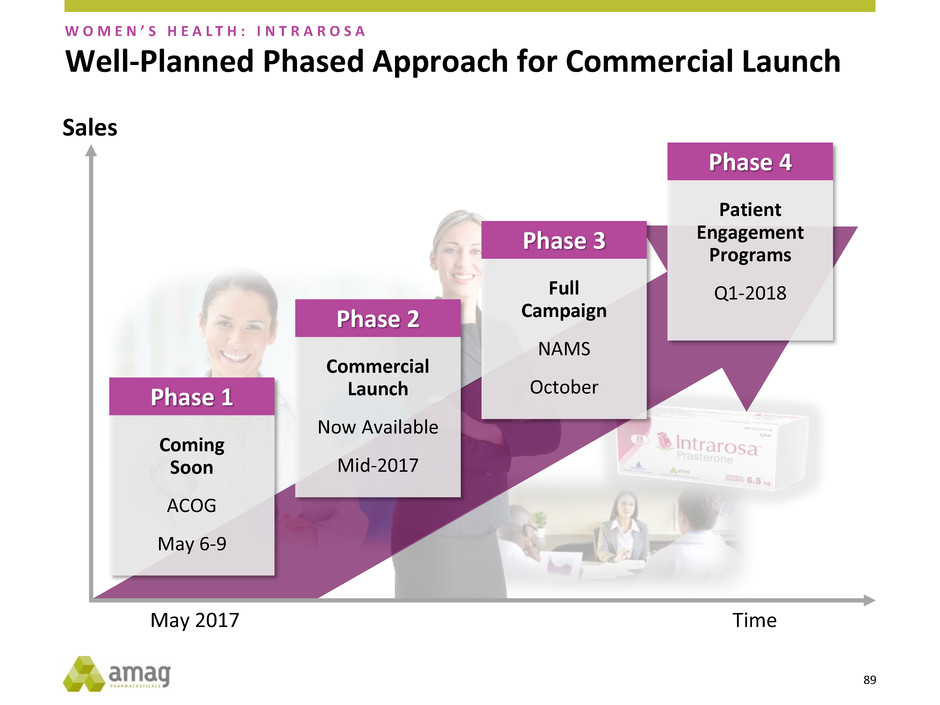
Full
Campaign
NAMS
October
Phase 3
89
Well-Planned Phased Approach for Commercial Launch
W O M E N ’ S H E A L T H : I N T R A R O S A
Coming
Soon
ACOG
May 6-9
Phase 1
Commercial
Launch
Now Available
Mid-2017
Phase 2
Patient
Engagement
Programs
Q1-2018
Phase 4
May 2017 Time
Sales

90
“Uncover DHEA” Debuted at ACOG – Well Received
Greater brand presence at ACOG than past years
W O M E N ’ S H E A L T H : I N T R A R O S A
Elevate awareness and importance of DHEA

91
Launch Day 1 – Now Available
W O M E N ’ S H E A L T H : I N T R A R O S A
Arming the Sales Force
Sales training modules / certification
Annotated PI leave behind
Patient co-pay card brochure
Pharmacy intro sheet
Co-pay flash card
“Now available” ad in
journals / online

Differentiated mechanism of action
Only FDA-approved non-estrogen local product1 for dyspareunia
due to menopause
– Only product without a boxed warning
Strong physician interest to prescribe
Well-planned launch strategy to quickly capture market share
Significant market opportunity with sizeable revenue potential
92
In Summary
W O M E N ’ S H E A L T H : I N T R A R O S A
1 Locally converted to estrogens and androgens.

Women’s Health:
Bremelanotide
James Simon, MD
Clinical Professor, Dept. of OB/GYN,
George Washington University
Julie Krop, MD
AMAG Chief Medical Officer
Nik Grund
AMAG Chief Commercial Officer

94
Bremelanotide Agenda
Disease Overview and Need
Mechanism of Action (MOA)
Phase 3 Clinical Overview
Co-primary Endpoints Met
Key Safety Data
Regulatory Path and Timeline
Significant Market Opportunity
Launch Readiness Strategy
Recent ACOG Initiatives
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E

95
Bremelanotide Agenda
Disease Overview and Need
Mechanism of Action (MOA)
Phase 3 Clinical Overview
Co-primary Endpoints Met
Key Safety Data
Regulatory Path and Timeline
Significant Market Opportunity
Launch Readiness Strategy
Recent ACOG Initiatives
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E

MC4R
MC4R
Dopamine
axon terminal
D1 dopamine
receptor
96
Presumed Mechanism of Action (MOA)
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
HSDD-related dopamine release Treatment of HSDD with bremelanotide
Decreased release
of dopamine
MC4R
MC4R
Dopamine
axon terminal
D1 dopamine
receptor
Bremelanotide
Increased release of
dopamine
In pre-clinical animal studies, efficacy was blocked by dopamine antagonist1
1 Pre-clinical animal studies conducted by Palatin.

97
Bremelanotide Agenda
Disease Overview and Need
Mechanism of Action (MOA)
Phase 3 Clinical Overview
Co-primary Endpoints Met
Key Safety Data
Regulatory Path and Timeline
Significant Market Opportunity
Launch Readiness Strategy
Recent ACOG Initiatives
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
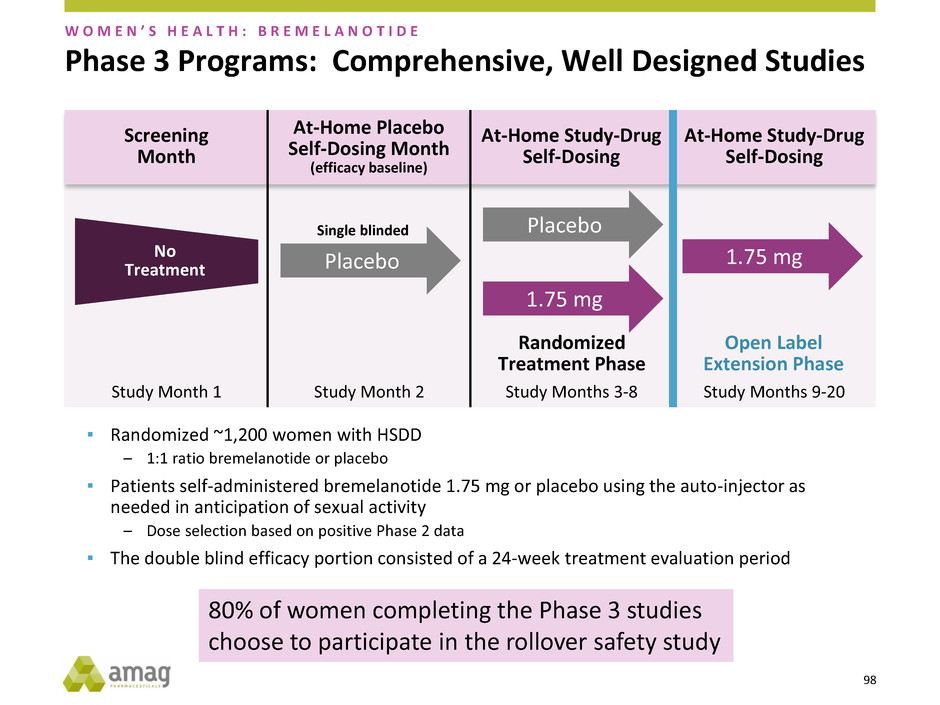
98
Phase 3 Programs: Comprehensive, Well Designed Studies
▪ Randomized ~1,200 women with HSDD
– 1:1 ratio bremelanotide or placebo
▪ Patients self-administered bremelanotide 1.75 mg or placebo using the auto-injector as
needed in anticipation of sexual activity
– Dose selection based on positive Phase 2 data
▪ The double blind efficacy portion consisted of a 24-week treatment evaluation period
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
Screening
Month
At-Home Placebo
Self-Dosing Month
(efficacy baseline)
At-Home Study-Drug
Self-Dosing
At-Home Study-Drug
Self-Dosing
No
Treatment Placebo
Placebo
1.75 mg
1.75 mg
Open Label
Extension Phase
Randomized
Treatment Phase
Study Month 1 Study Month 2 Study Months 3-8 Study Months 9-20
Single blinded
80% of women completing the Phase 3 studies
choose to participate in the rollover safety study

99
Phase 3 Outcome Measures: Clearly Defined Endpoints
▪ Co-primary Efficacy Endpoints
– Change in Female Sexual Function Index (FSFI):
Desire Domain (FSFI-D) Score
– Change in Female Sexual Distress Scale:
Desire/Arousal/Orgasm (FSDS-DAO) Item 13 Score
▪ Key Secondary Endpoints
– Self-assessment of number of satisfying sexual event (SSE)
– Change in FSFI total, arousal, lubrication, orgasm, and
satisfaction scores
▪ Responder Analysis
– Participants self-reporting a score of ≥5 (on a 7-point Likert scale)
in response to question 3 on the General Assessment
Questionnaire (GAQ) “To what degree do you think you benefited
from taking the study drug?”
– The proportion of participants meeting or exceeding predefined
minimal clinically important differences (MCIDs)
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
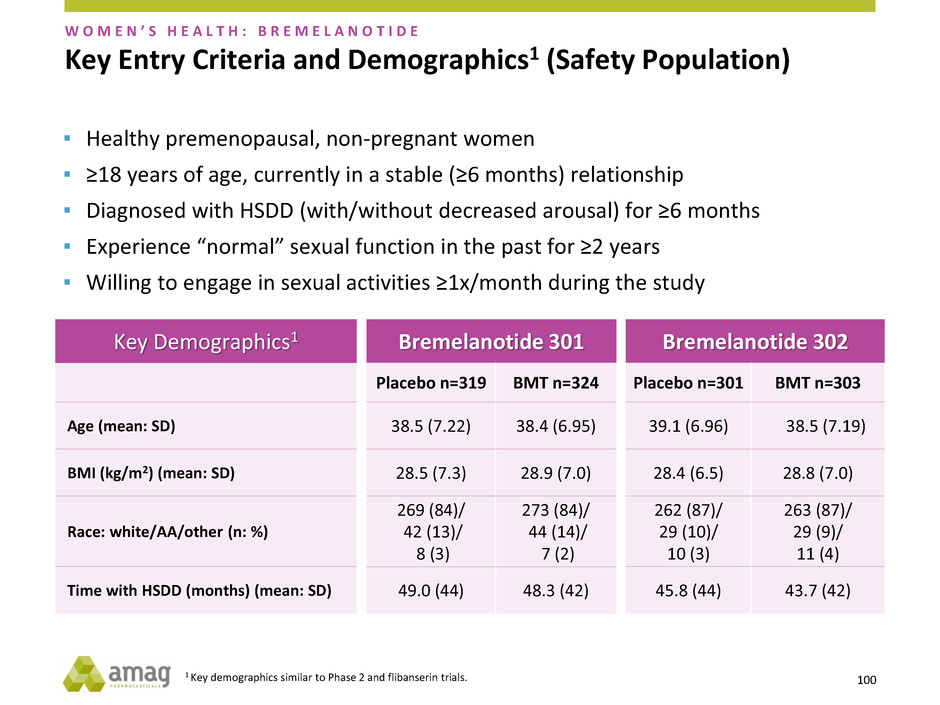
Key Demographics1
Age (mean: SD)
BMI (kg/m2) (mean: SD)
Race: white/AA/other (n: %)
Time with HSDD (months) (mean: SD)
Bremelanotide 301
Placebo n=319 BMT n=324
38.5 (7.22) 38.4 (6.95)
28.5 (7.3) 28.9 (7.0)
269 (84)/
42 (13)/
8 (3)
273 (84)/
44 (14)/
7 (2)
49.0 (44) 48.3 (42)
Bremelanotide 302
Placebo n=301 BMT n=303
39.1 (6.96) 38.5 (7.19)
28.4 (6.5) 28.8 (7.0)
262 (87)/
29 (10)/
10 (3)
263 (87)/
29 (9)/
11 (4)
45.8 (44) 43.7 (42)
1 Key demographics similar to Phase 2 and flibanserin trials. 100
Key Entry Criteria and Demographics1 (Safety Population)
▪ Healthy premenopausal, non-pregnant women
▪ ≥18 years of age, currently in a stable (≥6 months) relationship
▪ Diagnosed with HSDD (with/without decreased arousal) for ≥6 months
▪ Experience “normal” sexual function in the past for ≥2 years
▪ Willing to engage in sexual activities ≥1x/month during the study
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E

101
Bremelanotide Agenda
Disease Overview and Need
Mechanism of Action (MOA)
Phase 3 Clinical Overview
Co-primary Endpoints Met
Key Safety Data
Regulatory Path and Timeline
Significant Market Opportunity
Launch Readiness Strategy
Recent ACOG Initiatives
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
102 P values determined by unadjusted Wilcoxon rank-sum test. Error bars are standard error of the mean.
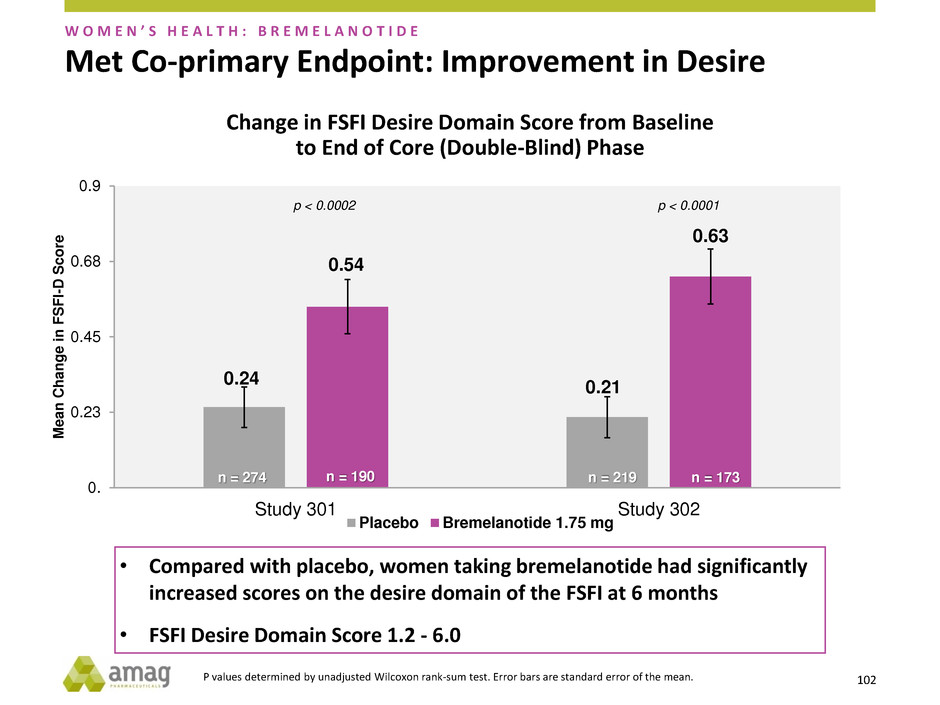
Met Co-primary Endpoint: Improvement in Desire
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
Change in FSFI Desire Domain Score from Baseline
to End of Core (Double-Blind) Phase
0.24 0.21
0.54
0.63
0.
0.23
0.45
0.68
0.9
Study 301 Study 302
M
e
a
n
C
h
a
n
g
e
i
n
F
S
F
I-
D
S
c
o
re
Placebo Bremelanotide 1.75 mg
p < 0.0002 p < 0.0001
n = 190 n = 274 n = 173 n = 219
• Compared with placebo, women taking bremelanotide had significantly
increased scores on the desire domain of the FSFI at 6 months
• FSFI Desire Domain Score 1.2 - 6.0
-0.42
-0.35
-0.71 -0.74
-1.
-0.75
-0.5
-0.25
0.
Study 301 Study 302
M
e
a
n
C
h
a
n
g
e
i
n
F
S
D
S
-D
A
O
I
tem
1
3
Placebo Bremelanotide 1.75 mg
103
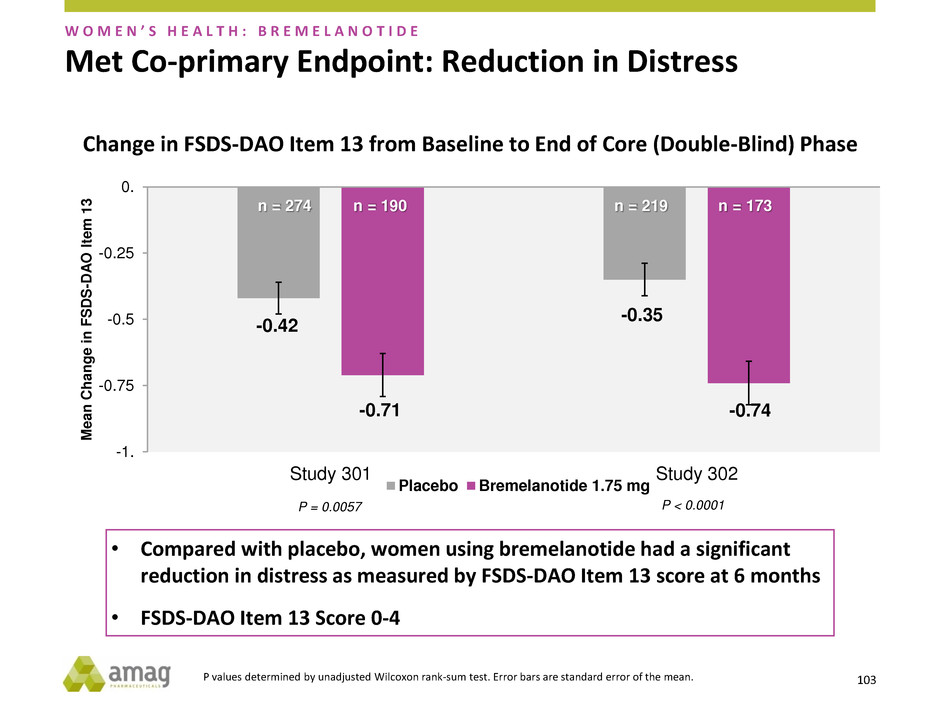
Met Co-primary Endpoint: Reduction in Distress
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
• Compared with placebo, women using bremelanotide had a significant
reduction in distress as measured by FSDS-DAO Item 13 score at 6 months
• FSDS-DAO Item 13 Score 0-4
P = 0.0057 P < 0.0001
n = 190 n = 274 n = 173 n = 219
Change in FSDS-DAO Item 13 from Baseline to End of Core (Double-Blind) Phase
P values determined by unadjusted Wilcoxon rank-sum test. Error bars are standard error of the mean.
104
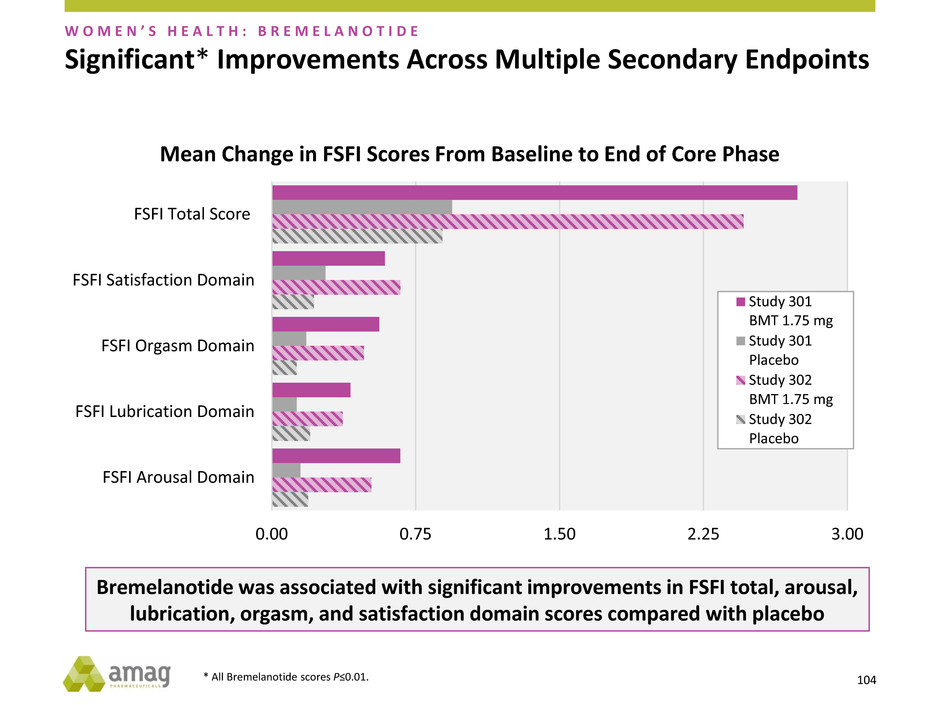
Significant* Improvements Across Multiple Secondary Endpoints
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
0.00 0.75 1.50 2.25 3.00
FSFI Total Score
FSFI Satisfaction Domain
FSFI Orgasm Domain
FSFI Lubrication Domain
FSFI Arousal Domain
Study 301
BMT 1.75 mg
Study 301
Placebo
Study 302
BMT 1.75 mg
Study 302
Placebo
Mean Change in FSFI Scores From Baseline to End of Core Phase
Bremelanotide was associated with significant improvements in FSFI total, arousal,
lubrication, orgasm, and satisfaction domain scores compared with placebo
* All Bremelanotide scores P≤0.01.

Difference in number of SSEs not statistically significant
Expert consensus1 that SSE is a poor endpoint for trials evaluating
treatments for HSDD
– Sexual activity/frequency not considered in the diagnosis
SSEs were required by FDA to attempt to have an objective “countable”
endpoint similar to successful erections in ED trials
Treatment goal is to improve DESIRE and reduce associated distress—
not necessarily increase the frequency of satisfying events
– SSE is a DOWNSTREAM behavior separate from emotion of desire with many potential
confounds (e.g. partner not available, interruptions from others, menstrual cycle etc.
Higher number of SSEs at baseline was actually correlated with increase
in distress in the study population
105
SSEs Not Effective Endpoint for HSDD
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
1 FDA workshop October 2014
106
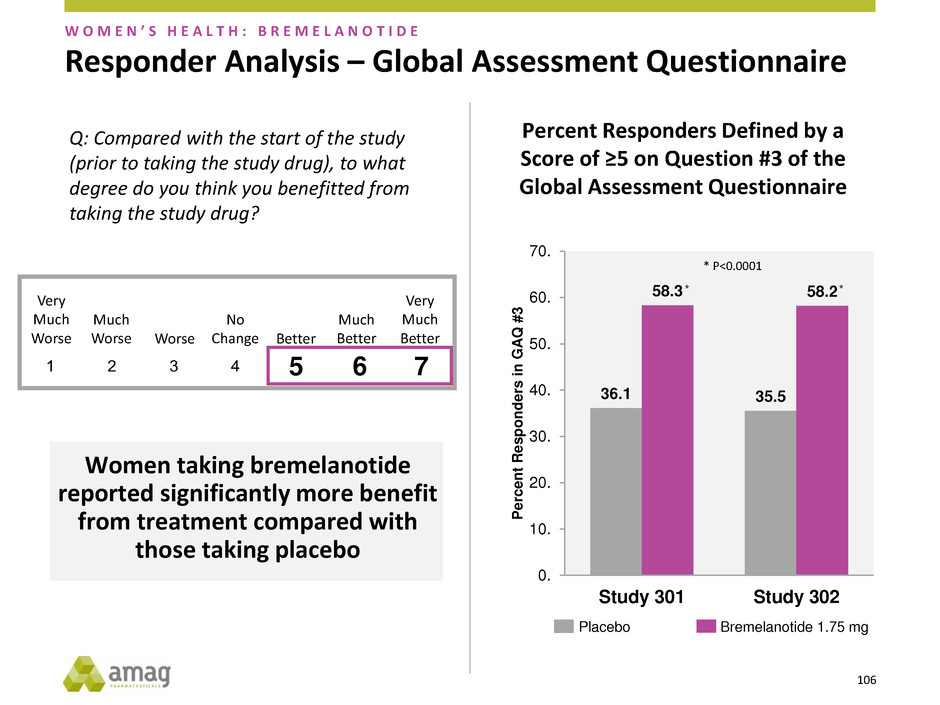
Responder Analysis – Global Assessment Questionnaire
Percent Responders Defined by a
Score of ≥5 on Question #3 of the
Global Assessment Questionnaire
36.1 35.5
58.3 58.2
0.
10.
20.
30.
40.
50.
60.
70.
Study 301 Study 302
P
erce
n
t
R
e
s
p
o
n
d
ers
i
n
G
A
Q
#
3
Bremelanotide 1.75 mg Placebo
Women taking bremelanotide
reported significantly more benefit
from treatment compared with
those taking placebo
* *
* P<0.0001
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
Very
Much
Worse
Much
Worse Worse
No
Change
Very
Much
Better
Much
Better Better
5 6 7
Q: Compared with the start of the study
(prior to taking the study drug), to what
degree do you think you benefitted from
taking the study drug?

107
Bremelanotide Agenda
Disease Overview and Need
Mechanism of Action (MOA)
Phase 3 Clinical Overview
Co-primary Endpoints Met
Key Safety Data
Regulatory Path and Timeline
Significant Market Opportunity
Launch Readiness Strategy
Recent ACOG Initiatives
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
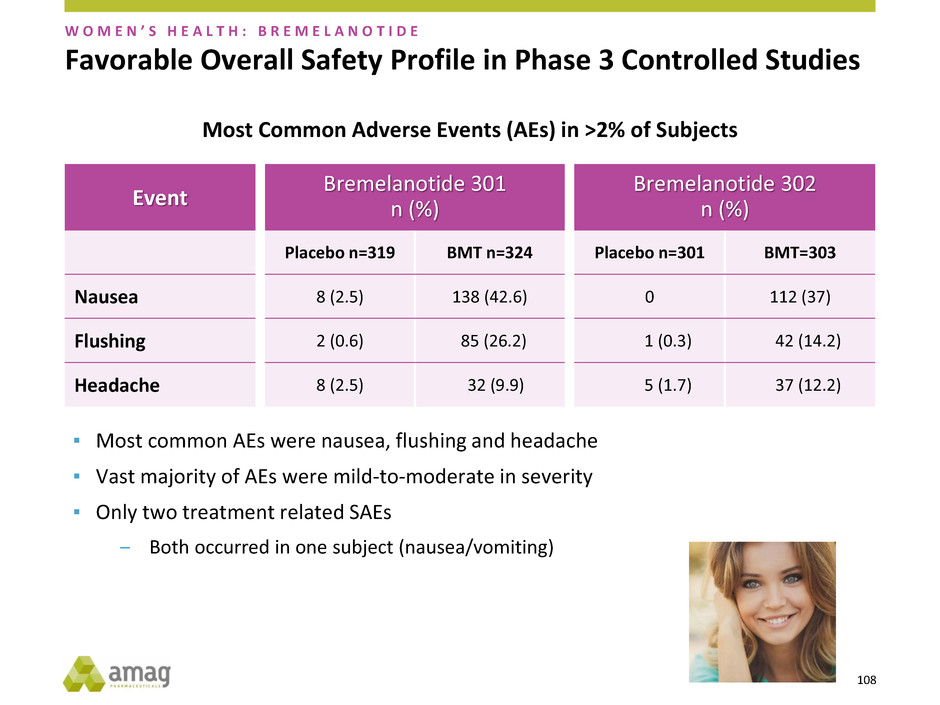
Event
Nausea
Flushing
Headache
Bremelanotide 301
n (%)
Placebo n=319 BMT n=324
8 (2.5) 138 (42.6)
2 (0.6) 85 (26.2)
8 (2.5) 32 (9.9)
Bremelanotide 302
n (%)
Placebo n=301 BMT=303
0 112 (37)
1 (0.3) 42 (14.2)
5 (1.7) 37 (12.2)
108
Favorable Overall Safety Profile in Phase 3 Controlled Studies
▪ Most common AEs were nausea, flushing and headache
▪ Vast majority of AEs were mild-to-moderate in severity
▪ Only two treatment related SAEs
‒ Both occurred in one subject (nausea/vomiting)
Most Common Adverse Events (AEs) in >2% of Subjects
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E

109
Bremelanotide Agenda
Disease Overview and Need
Mechanism of Action (MOA)
Phase 3 Clinical Overview
Co-primary Endpoints Met
Key Safety Data
Regulatory Path and Timeline
Significant Market Opportunity
Launch Readiness Strategy
Recent ACOG Initiatives
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
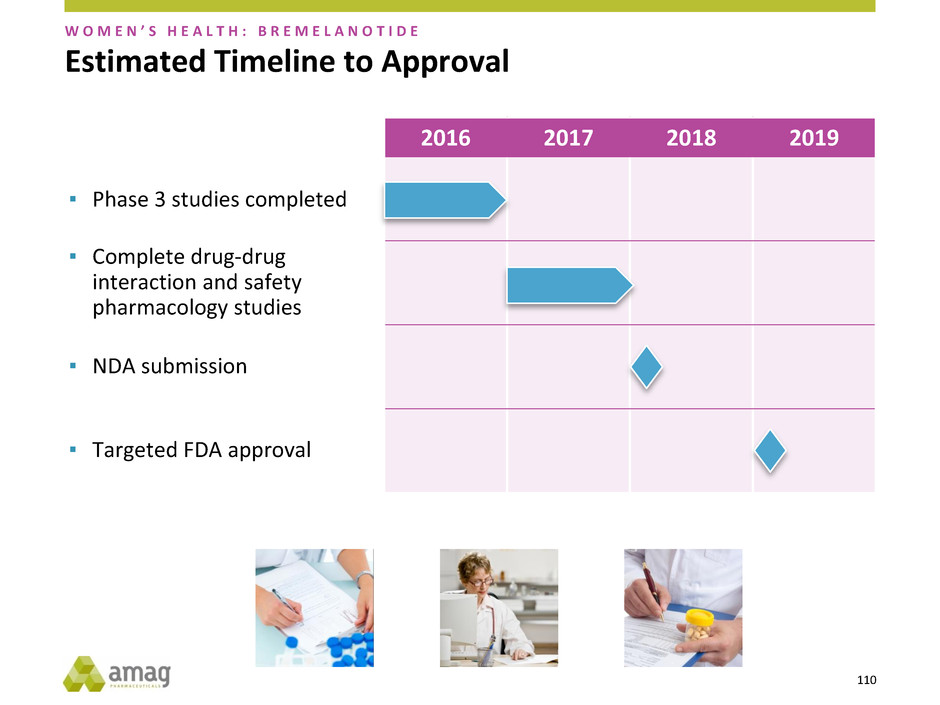
2016 2017 2018 2019
110
Estimated Timeline to Approval
▪ Targeted FDA approval
▪ Phase 3 studies completed
▪ Complete drug-drug
interaction and safety
pharmacology studies
▪ NDA submission
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E

111
Bremelanotide Agenda
Disease Overview and Need
Mechanism of Action (MOA)
Phase 3 Clinical Overview
Co-primary Endpoints Met
Key Safety Data
Regulatory Path and Timeline
Significant Market Opportunity
Launch Readiness Strategy
Recent ACOG Initiatives
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
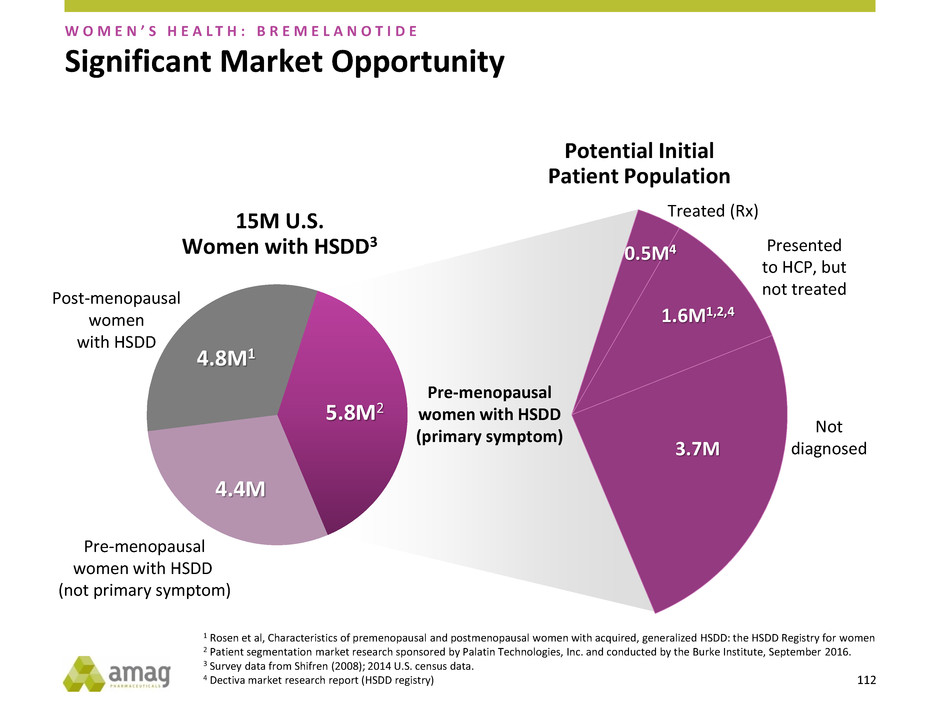
Treated (Rx)
Presented
to HCP, but
not treated
Not
diagnosed
0.5M4
1.6M1,2,4
3.7M
112
1 Rosen et al, Characteristics of premenopausal and postmenopausal women with acquired, generalized HSDD: the HSDD Registry for women
2 Patient segmentation market research sponsored by Palatin Technologies, Inc. and conducted by the Burke Institute, September 2016.
3 Survey data from Shifren (2008); 2014 U.S. census data.
4 Dectiva market research report (HSDD registry)
Significant Market Opportunity
Pre-menopausal
women with HSDD
(primary symptom)
Pre-menopausal
women with HSDD
(not primary symptom)
Post-menopausal
women
with HSDD
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
4.8M1
4.4M
5.8M2
15M U.S.
Women with HSDD3
Potential Initial
Patient Population

113
Bremelanotide Agenda
Disease Overview and Need
Mechanism of Action (MOA)
Phase 3 Clinical Overview
Co-primary Endpoints Met
Key Safety Data
Regulatory Path and Timeline
Significant Market Opportunity
Launch Readiness Strategy
Recent ACOG Initiatives
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
114
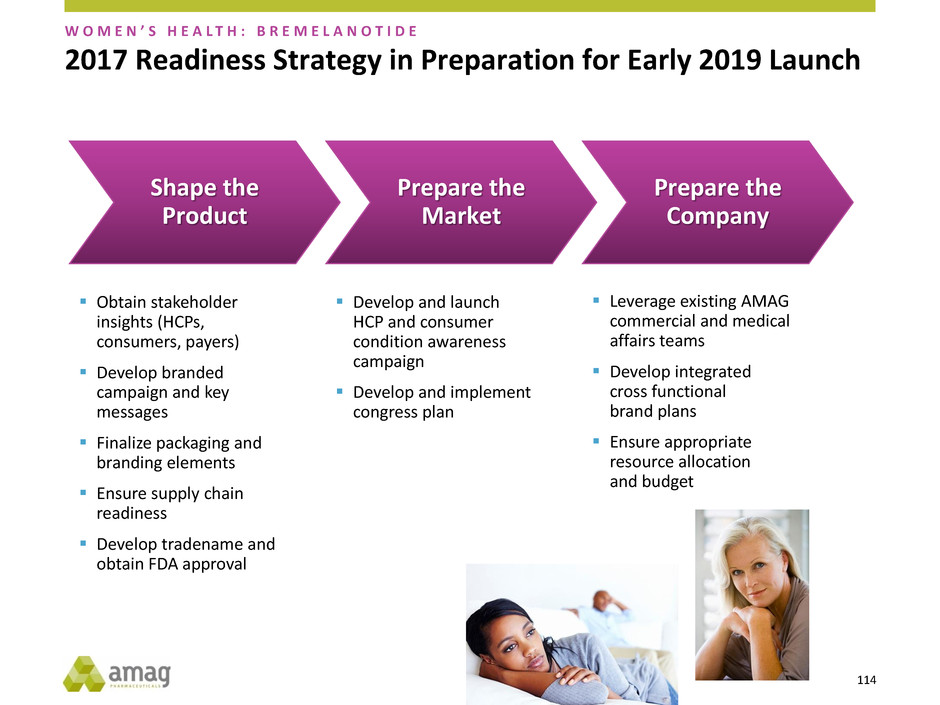
2017 Readiness Strategy in Preparation for Early 2019 Launch
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
Obtain stakeholder
insights (HCPs,
consumers, payers)
Develop branded
campaign and key
messages
Finalize packaging and
branding elements
Ensure supply chain
readiness
Develop tradename and
obtain FDA approval
Shape the
Product
Develop and launch
HCP and consumer
condition awareness
campaign
Develop and implement
congress plan
Prepare the
Market
Leverage existing AMAG
commercial and medical
affairs teams
Develop integrated
cross functional
brand plans
Ensure appropriate
resource allocation
and budget
Prepare the
Company
115
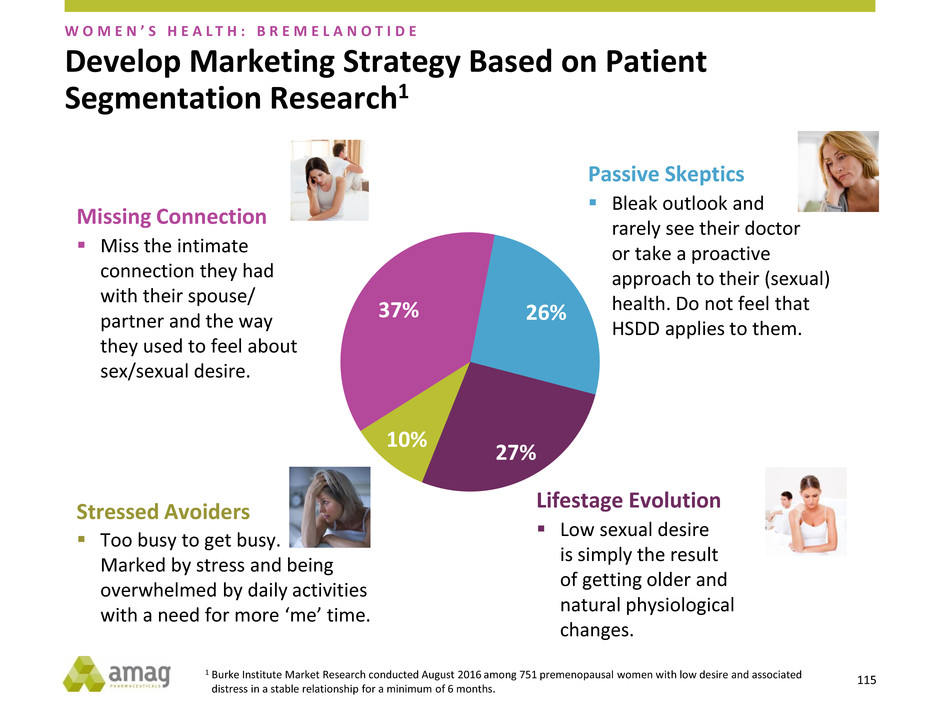
Develop Marketing Strategy Based on Patient
Segmentation Research1
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
Missing Connection
Miss the intimate
connection they had
with their spouse/
partner and the way
they used to feel about
sex/sexual desire.
Stressed Avoiders
Too busy to get busy.
Marked by stress and being
overwhelmed by daily activities
with a need for more ‘me’ time.
Passive Skeptics
Bleak outlook and
rarely see their doctor
or take a proactive
approach to their (sexual)
health. Do not feel that
HSDD applies to them.
Lifestage Evolution
Low sexual desire
is simply the result
of getting older and
natural physiological
changes.
26%
27%
10%
37%
1 Burke Institute Market Research conducted August 2016 among 751 premenopausal women with low desire and associated
distress in a stable relationship for a minimum of 6 months.

116
Bremelanotide Agenda
Disease Overview and Need
Mechanism of Action (MOA)
Phase 3 Clinical Overview
Co-primary Endpoints Met
Key Safety Data
Regulatory Path and Timeline
Significant Market Opportunity
Launch Readiness Strategy
Recent ACOG Initiatives
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E

117
Fostering HSDD Disease Awareness and Education
HSDD Booth HCP HSDD Diagnostic Screener Kit
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E

118
Fostering HSDD Disease Awareness and Education
HSDD Booth HCP HSDD Diagnostic Screener Kit
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E

119
Significant Interest in HSDD
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E
Product Theater at ACOG
“Sexual Desire: Everything You Always
Wanted to Know But Were Afraid to Ask”
Everything You Wanted to Know
But Were Afraid to Ask
▪ Objective: A focused, interactive
forum to gather and discuss issues
on HSDD
▪ Attendees: 120 attended
▪ Speakers and Discussions:
– Dr. Sharon Parrish -
Patient/physician perception of
associated distress with HSDD
– Dr. Sheryl Kingsberg –
“From Dialogue to Diagnosis”
– Dr. James Simon –
Neurobiology of Sexual Desire

Met co-primary, pre-specified endpoints on desire and distress
– Both key symptoms of HSDD
– Responder analysis shows results are clinically meaningful to patients
Consistent efficacy across multiple domains of sexual functioning
Favorable safety profile
On track for NDA submission Q1-2018
Large market opportunity
Significant HSDD education and awareness
campaign taking place prior to potential launch in
early 2019 to ensure successful launch
120
In Summary: Significant Opportunity in Area of High
Unmet Need
W O M E N ’ S H E A L T H : B R E M E L A N O T I D E

121
Q&A with External Experts
Risa Kagan, MD, FACOG, CCD, NCMP
• Clinical Professor in the Department of Obstetrics, Gynecology, and
Reproductive Sciences, University of California, San Francisco
• Practicing gynecologist with the East Bay Physicians Medical Group,
Sutter Medical Foundation
Fernand Labrie, MD, PhD
• Chief Executive Officer and Founder of EndoceuticsTM
• Emeritus Professor, Laval University
James Simon, MD, CCD, NCMP, IF, FACOG
• Clinical Professor of Obstetrics and Gynecology,
George Washington University
• Medical Director, Women’s Health & Research Consultants®
• Current President-elect of The International Society for the Study of
Women’s Sexual Health (ISSWSH)

Financial Overview
Ted Myles
Chief Financial Officer

123
Agenda
AMAG Evolution
Capital Allocation
Business Development
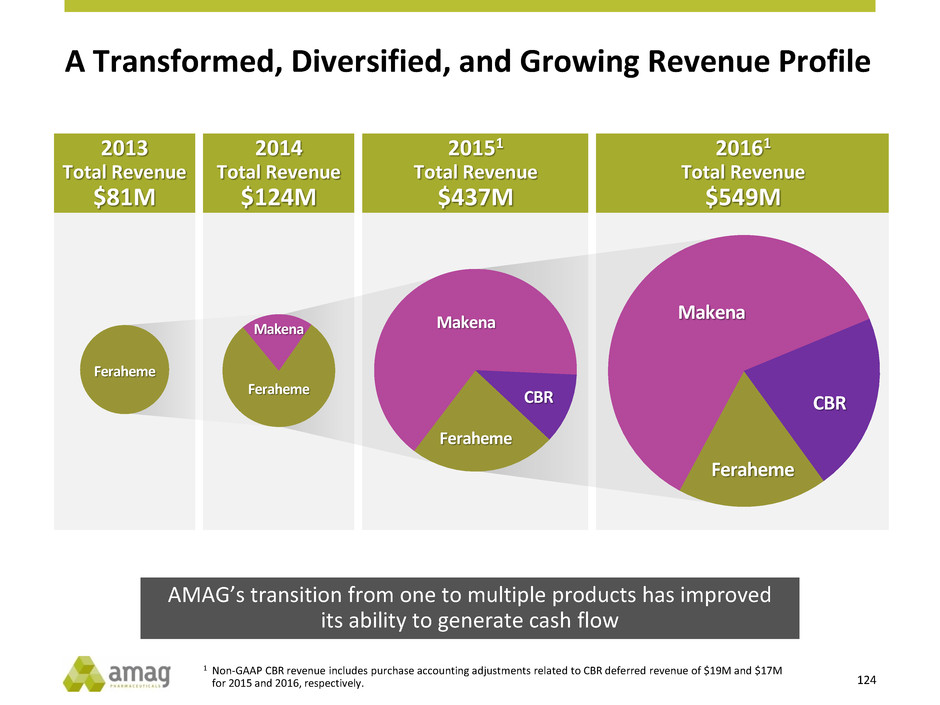
124
1 Non-GAAP CBR revenue includes purchase accounting adjustments related to CBR deferred revenue of $19M and $17M
for 2015 and 2016, respectively.
A Transformed, Diversified, and Growing Revenue Profile
20161
Total Revenue
$549M
20151
Total Revenue
$437M
2014
Total Revenue
$124M
2013
Total Revenue
$81M
Feraheme
Feraheme
Feraheme
Makena Makena
CBR
AMAG’s transition from one to multiple products has improved
its ability to generate cash flow
Feraheme
Makena
CBR
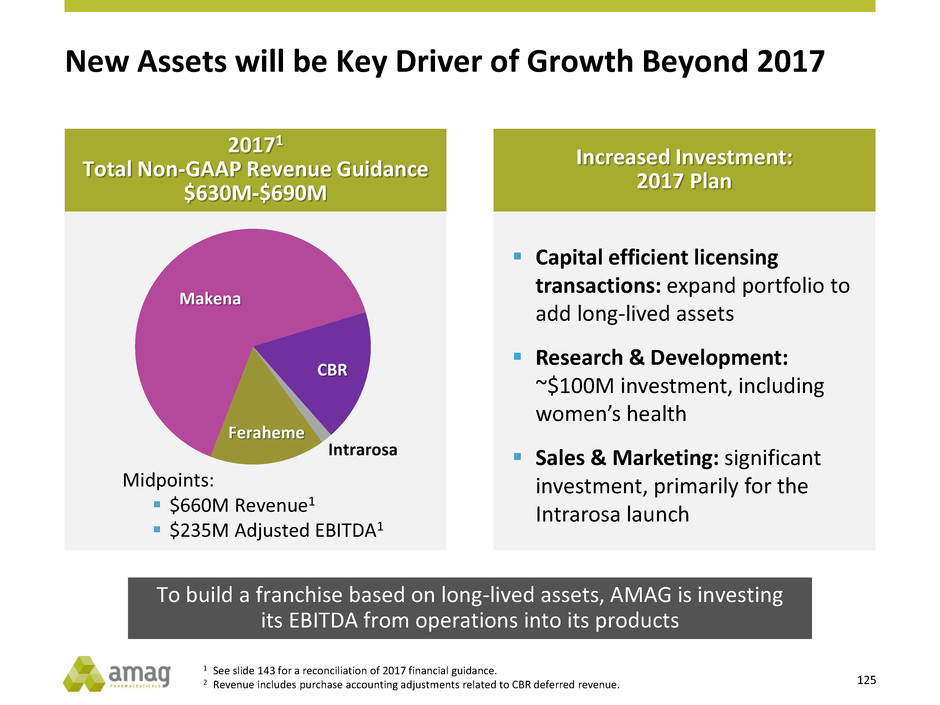
125
1 See slide 143 for a reconciliation of 2017 financial guidance.
2 Revenue includes purchase accounting adjustments related to CBR deferred revenue.
New Assets will be Key Driver of Growth Beyond 2017
20171
Total Non-GAAP Revenue Guidance
$630M-$690M
Feraheme
Makena
CBR
Intrarosa
To build a franchise based on long-lived assets, AMAG is investing
its EBITDA from operations into its products
Increased Investment:
2017 Plan
Midpoints:
$660M Revenue1
$235M Adjusted EBITDA1
Capital efficient licensing
transactions: expand portfolio to
add long-lived assets
Research & Development:
~$100M investment, including
women’s health
Sales & Marketing: significant
investment, primarily for the
Intrarosa launch

126
Agenda
AMAG Evolution
Capital Allocation
Business Development
127
Consistent Capital Allocation Principles
Focus on
Shareholder
Returns
3
Strong cash position allows us to pursue our
strategy, even in challenging markets
Full commitment to value-creating
opportunities that we choose to pursue:
organic or external
Key criterion in making investment decisions
Conviction to
Investments 2
Liquidity 1

128
1 Pro forma as of 12/31/17, debt balance assumes $20M from over-allotment option.
2 Approximates weighted average interest rate.
3 Reflects mid-point of financial guidance range. See slide 143 for a reconciliation of 2017 financial guidance.
Revised Liability Profile to Align with Business Strategy
BEFORE – Early Maturing Debt Profile
Key Items Totals Before
Cash1: $500M
Outstanding debt balance1: $1,028M
Interest rate2: 5.9%
Total annual interest cost: $60M
Projected adjusted EBITDA3: $235M
Leverage: 4.4x
Capital Structure
$18 $18
$218
$18
$258
$500
2017 2018 2019 2020 2021 2022 2023
Debt Payments $M
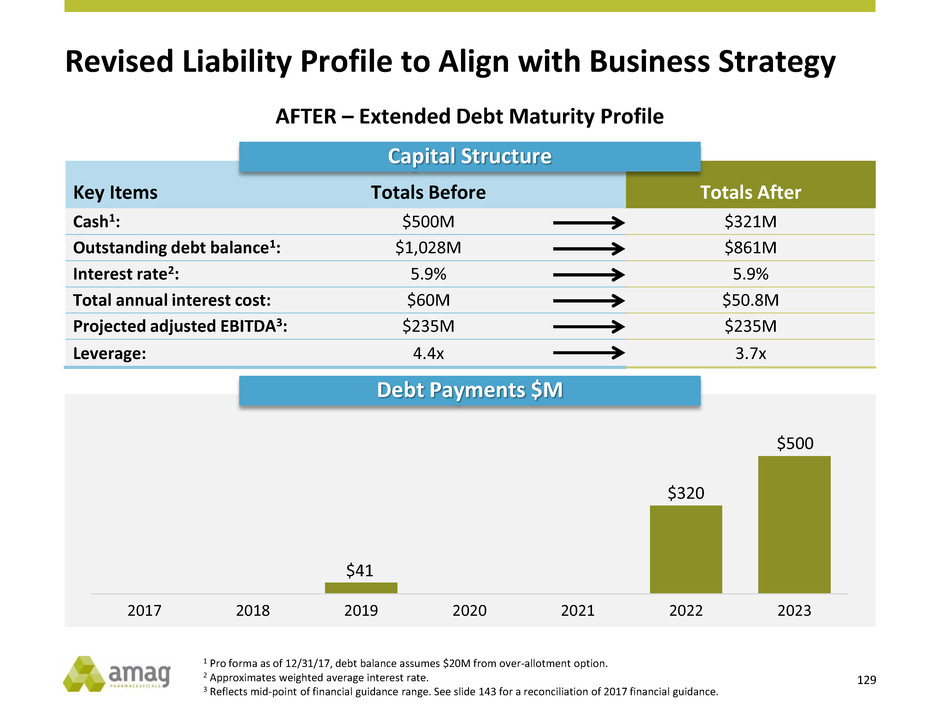
129
Revised Liability Profile to Align with Business Strategy
AFTER – Extended Debt Maturity Profile
Key Items Totals Before Totals After
Cash1: $500M $321M
Outstanding debt balance1: $1,028M $861M
Interest rate2: 5.9% 5.9%
Total annual interest cost: $60M $50.8M
Projected adjusted EBITDA3: $235M $235M
Leverage: 4.4x 3.7x
Capital Structure
Debt Payments $M
$41
$320
$500
2017 2018 2019 2020 2021 2022 2023
1 Pro forma as of 12/31/17, debt balance assumes $20M from over-allotment option.
2 Approximates weighted average interest rate.
3 Reflects mid-point of financial guidance range. See slide 143 for a reconciliation of 2017 financial guidance.
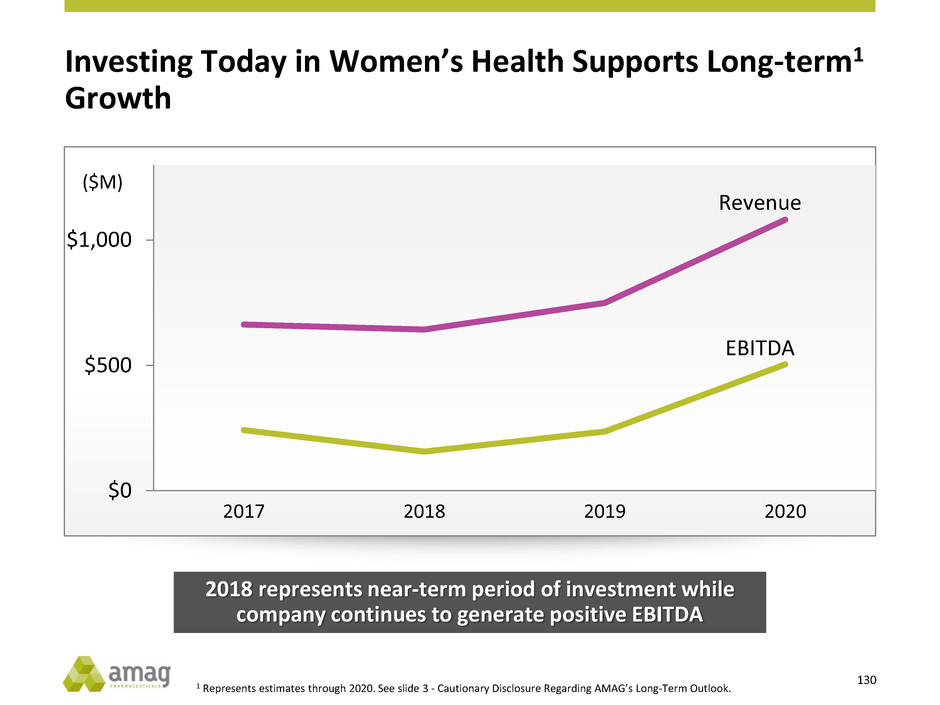
$0
$500
$1,000
2017 2018 2019 2020
130
Investing Today in Women’s Health Supports Long-term1
Growth
2018 represents near-term period of investment while
company continues to generate positive EBITDA
Revenue
EBITDA
($M)
1 Represents estimates through 2020. See slide 3 - Cautionary Disclosure Regarding AMAG’s Long-Term Outlook.
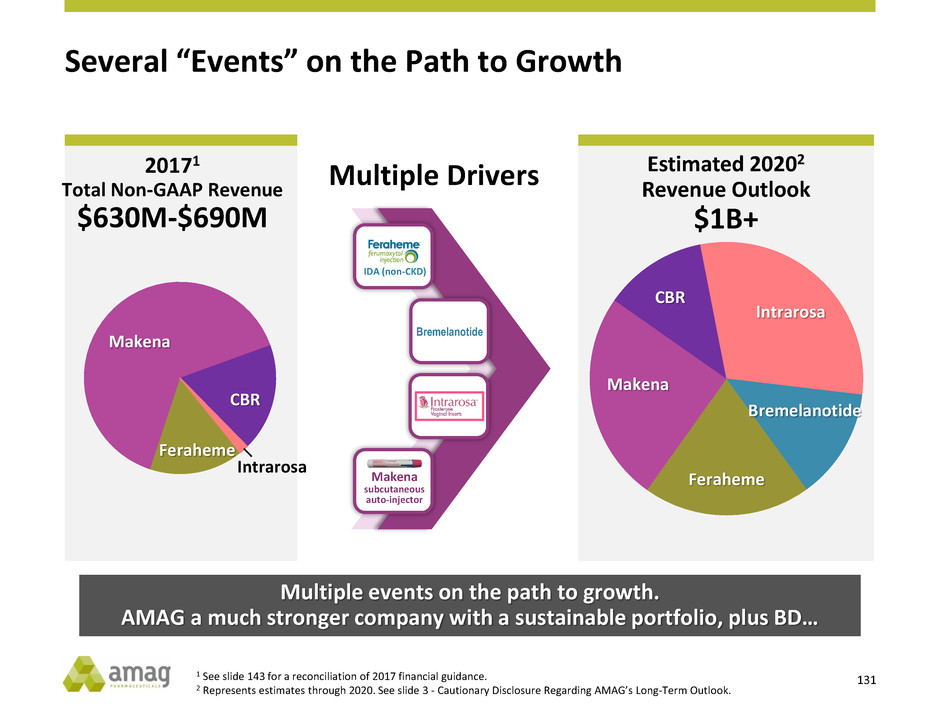
131
Several “Events” on the Path to Growth
20171
Total Non-GAAP Revenue
$630M-$690M
Feraheme
Makena
CBR
Intrarosa
Estimated 20202
Revenue Outlook
$1B+
Feraheme
Makena
CBR
Intrarosa
Bremelanotide
Multiple events on the path to growth.
AMAG a much stronger company with a sustainable portfolio, plus BD…
Multiple Drivers
Bremelanotide
IDA (non-CKD)
Makena
subcutaneous
auto-injector
1 See slide 143 for a reconciliation of 2017 financial guidance.
2 Represents estimates through 2020. See slide 3 - Cautionary Disclosure Regarding AMAG’s Long-Term Outlook.

132
Agenda
AMAG Evolution
Capital Allocation
Business Development
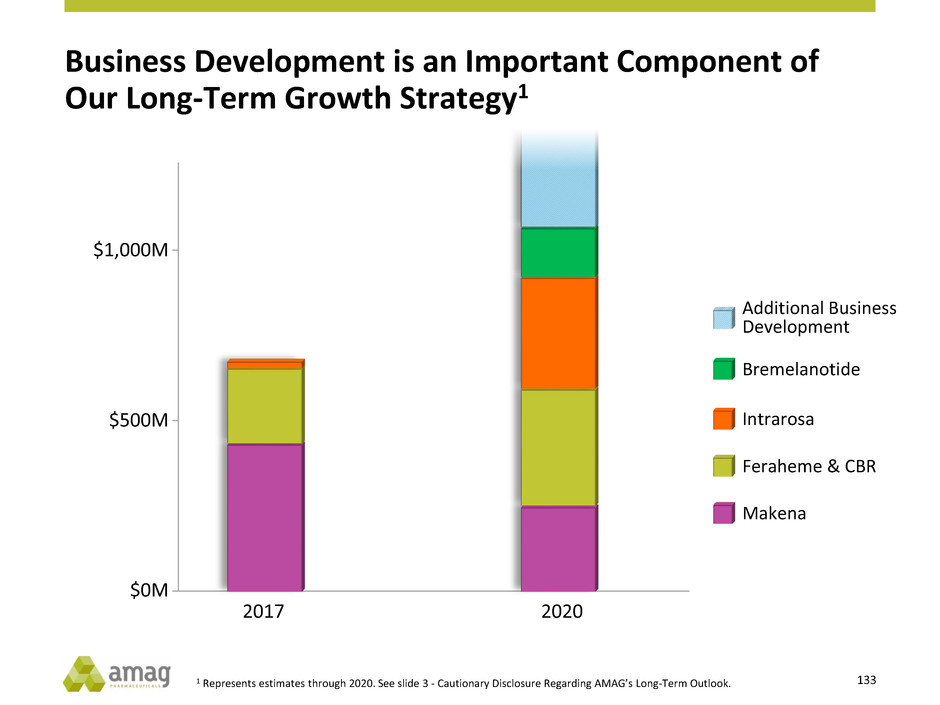
133
$0M
$500M
$1,000M
2017 2020
Makena
Feraheme & CBR
Intrarosa
Bremelanotide
Additional Business
Development
Business Development is an Important Component of
Our Long-Term Growth Strategy1
1 Represents estimates through 2020. See slide 3 - Cautionary Disclosure Regarding AMAG’s Long-Term Outlook.

134
Building Our Portfolio: Consistent Search and
Evaluation Criteria
Core therapeutic areas (TAs) (maternal
health, hem/onc & select hospital)
Adjacent TAs (women’s health, nephrology)
Therapeutic
Criteria
Differentiated & durable
Stage of development
Cash payback period and IRR
Financial
Criteria
Physician relationships
Commercial execution skills
Consumer / digital platform
Leverage
Core
Capabilities
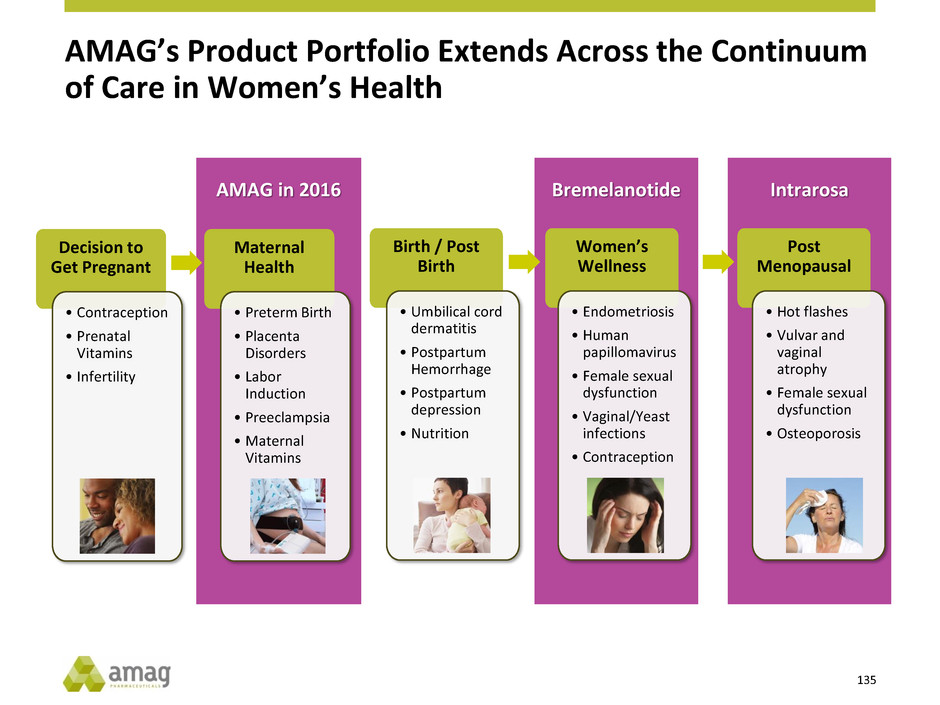
135
AMAG’s Product Portfolio Extends Across the Continuum
of Care in Women’s Health
AMAG in 2016
Decision to
Get Pregnant
• Contraception
• Prenatal
Vitamins
• Infertility
Maternal
Health
• Preterm Birth
• Placenta
Disorders
• Labor
Induction
• Preeclampsia
•Maternal
Vitamins
Bremelanotide Intrarosa
Birth / Post
Birth
• Umbilical cord
dermatitis
• Postpartum
Hemorrhage
• Postpartum
depression
• Nutrition
Women’s
Wellness
• Endometriosis
• Human
papillomavirus
• Female sexual
dysfunction
• Vaginal/Yeast
infections
• Contraception
Post
Menopausal
• Hot flashes
• Vulvar and
vaginal
atrophy
• Female sexual
dysfunction
• Osteoporosis

Balance sheet maturities now align with portfolio
Strong cash balance supportive of additional business
development
Positive EBITDA and strong cash flow generation from current
products enables investment in future products
136
Financial Profile Strengthened to Support Evolving
Business Model
I N S U M M A R Y

Q&A

Closing Remarks
Bill Heiden
AMAG President & Chief
Executive Officer
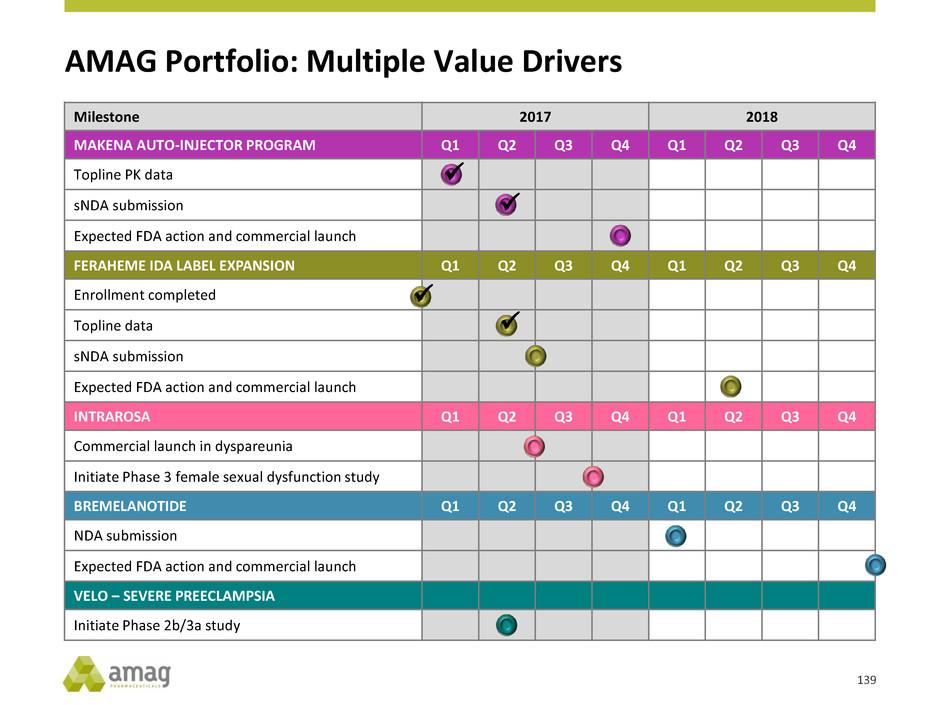
139
AMAG Portfolio: Multiple Value Drivers
Milestone 2017 2018
MAKENA AUTO-INJECTOR PROGRAM Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4
Topline PK data
sNDA submission
Expected FDA action and commercial launch
FERAHEME IDA LABEL EXPANSION Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4
Enrollment completed
Topline data
sNDA submission
Expected FDA action and commercial launch
INTRAROSA Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4
Commercial launch in dyspareunia
Initiate Phase 3 female sexual dysfunction study
BREMELANOTIDE Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4
NDA submission
Expected FDA action and commercial launch
VELO – SEVERE PREECLAMPSIA
Initiate Phase 2b/3a study
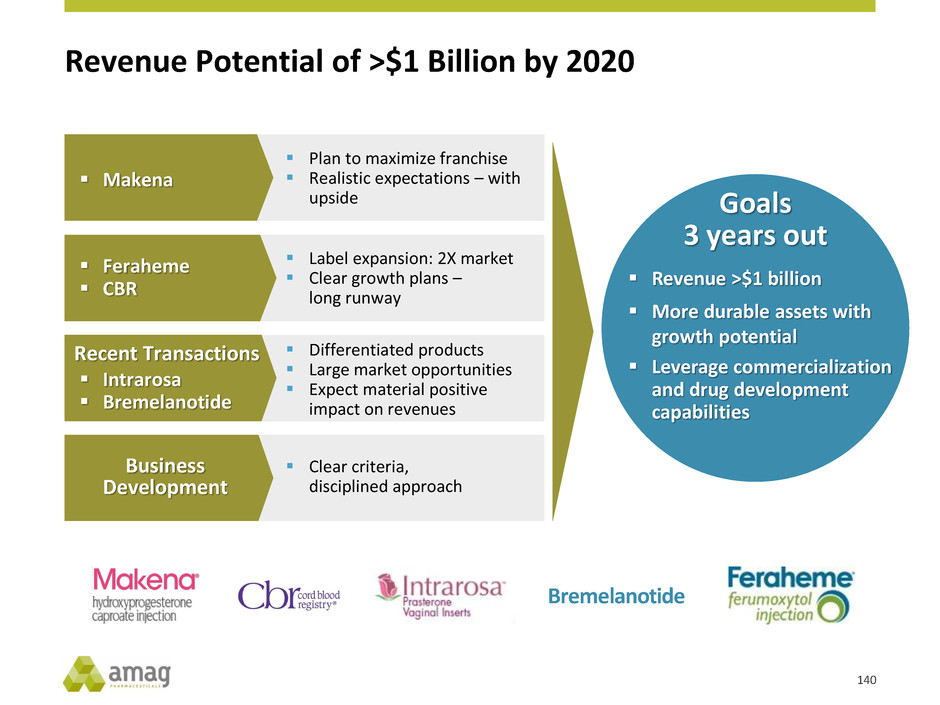
140
Revenue Potential of >$1 Billion by 2020
Revenue >$900 M
More durable assets with
growth potential
Leverage drug
development capabilities
Revenue >$1 billion
More durable assets with
growth potential
Leverage commercialization
and drug development
capabilities
Goals
3 years out
Bremelanotide
Makena
Recent Transactions
Intrarosa
Bremelanotide
Feraheme
CBR
Business
Development
Plan to maximize franchise
Realistic expectations – with
upside
Differentiated products
Large market opportunities
Expect material positive
impact on revenues
Label expansion: 2X market
Clear growth plans –
long runway
Clear criteria,
disciplined approach

AMAG Analyst Day
Thank you for coming!
May 24, 2017

Appendix
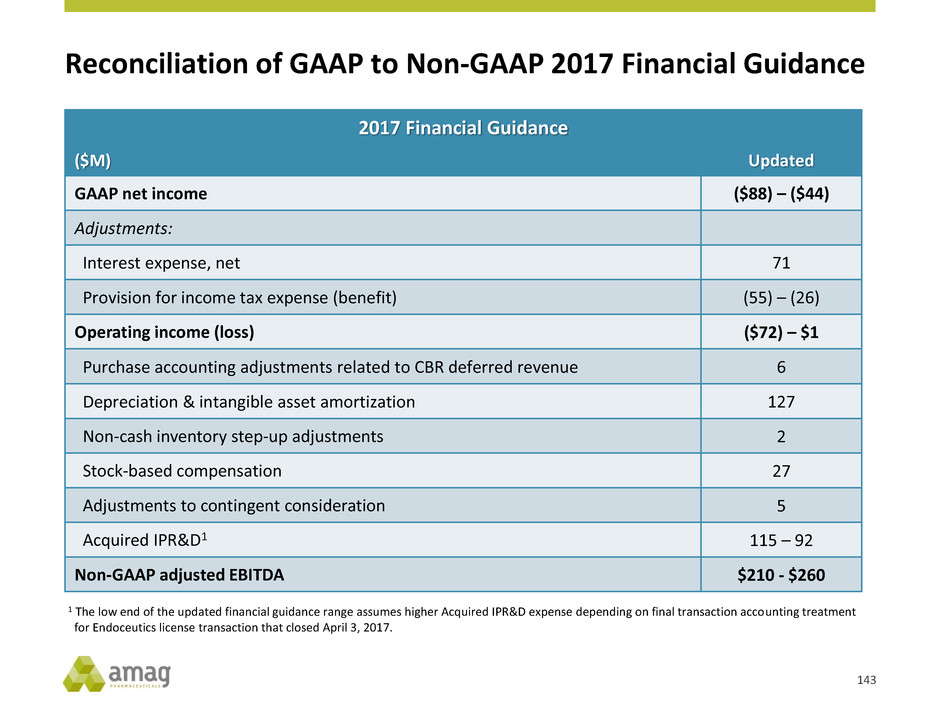
143
Reconciliation of GAAP to Non-GAAP 2017 Financial Guidance
2017 Financial Guidance
($M) Updated
GAAP net income ($88) – ($44)
Adjustments:
Interest expense, net 71
Provision for income tax expense (benefit) (55) – (26)
Operating income (loss) ($72) – $1
Purchase accounting adjustments related to CBR deferred revenue 6
Depreciation & intangible asset amortization 127
Non-cash inventory step-up adjustments 2
Stock-based compensation 27
Adjustments to contingent consideration 5
Acquired IPR&D1 115 – 92
Non-GAAP adjusted EBITDA $210 - $260
1 The low end of the updated financial guidance range assumes higher Acquired IPR&D expense depending on final transaction accounting treatment
for Endoceutics license transaction that closed April 3, 2017.

AMAG Analyst Day
Thank you for coming!
May 24, 2017































































































































































































































































































