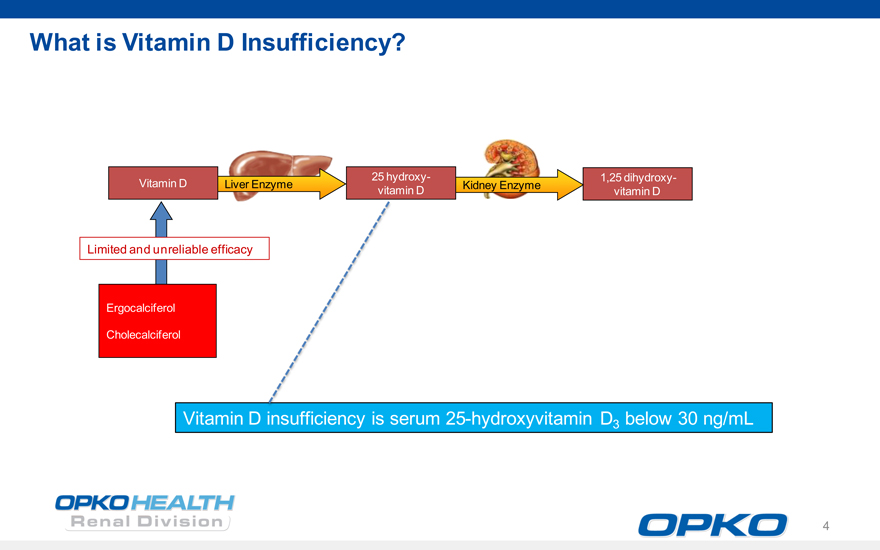
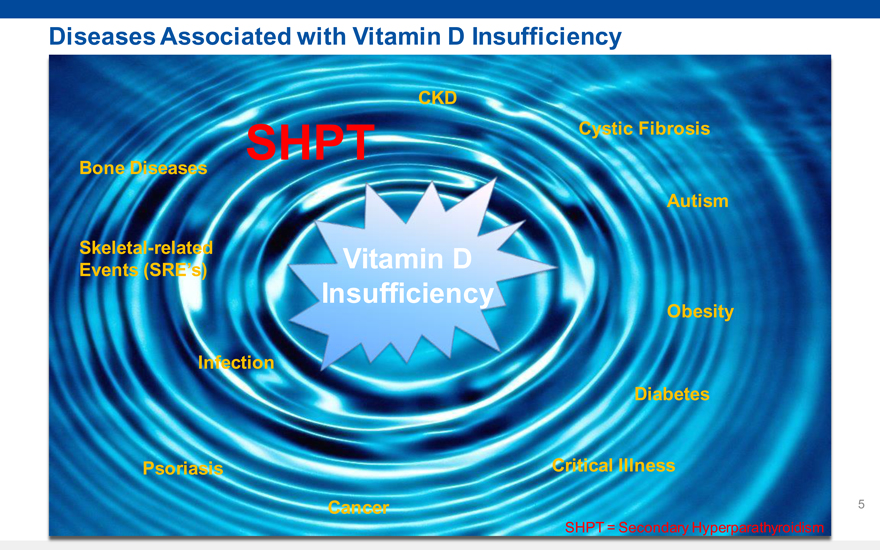
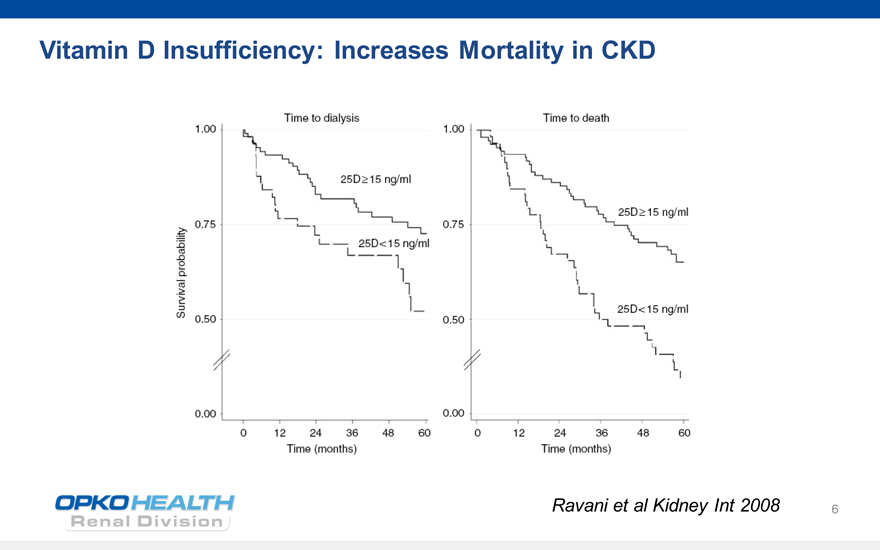
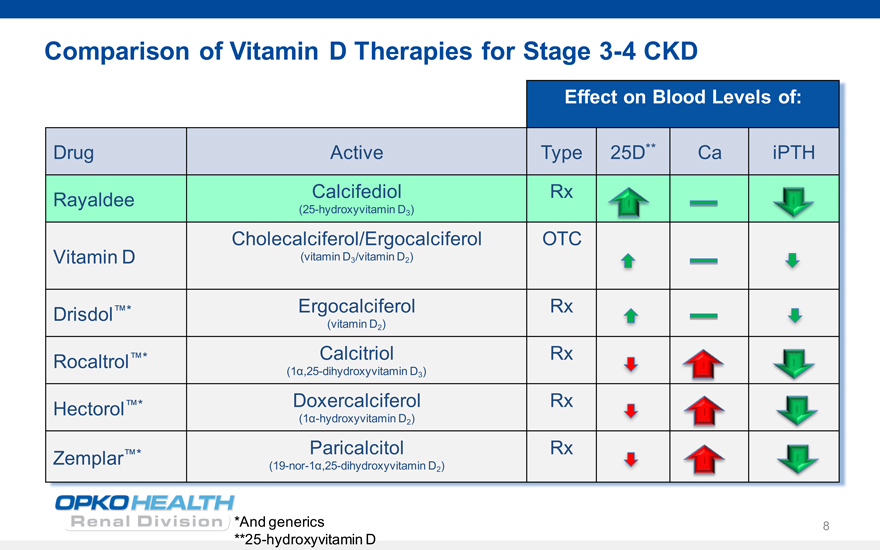
6
M8#(<@N>X7'VSO<1+>VL+9;3;!C,?:W;[^X+IYI
MH8X6,W\GV
M_&,K8?N1CKV^RMQ/C+BRNI)?V1/;NQ.^:&.;)WN0OY1<7)A-Q&70"8RNDDBK
MMUW"Y#R*2U[T8_ZANY?%LV.\O(,-A>,6%ER/'YC&06W[U6-_'RDQ6=W/:\_LR8NM[FXV;P^:]R98RW<_WF
MVQ8UA$9`MO?'ZK;7N3D_JJY'!RZ*XXG?PX?@O"L._(CY.3##DEF(K['V!E\E
MT-IA^-MFEOHXCY4N29-)*'W`+ODD^VRA"D*Z..$;([DJ6HLE!/ZRCV,I*1Z@
MJW'KK&N/[:]PCPW$7'%L)<8[G+>*:ED@QEEOV,#+RUM+VZ;
M;P/(=!M%&L:MBL]]1G8-G=OE./[E'V\%[R
M3+B(2S/FQ&3RN&Q4E]>1-9IMG>W6-FQAES%W!!9V5J]]S%%)-<
M6SC=32SRAP,LTCV2/#I7'%.9[IY/@/;'F&*^L/N3@.81]R^_O#GY?#XSD-IR
M,6G$\5>WV7S&3@BQUU=06EAD(QC;6UL[8Q/%O:6\,UO"8[6,??>GM9"'0`PE
M:]U,--E+BD=_M%Q"BM\+=24CN]$_DW`M_9_BF1QE[QO*V-OG7Q\-PUY>7`?Q
MR/$R_.OL)(9<7/=2?Z9F+J[FD?-,;<3Q23PQ7$DSGOB"D/U:]TN.\DPW<'C&
M8R'"(9^[?+L1B;!T/<&;E5K^R(6O)++&0']D\3QN+M+>&RLV9`V5U;V-
MYU>3XEP
MCMO!8\=?<\OMK;*\CR\C;0.GDR0PUW(VQN9BS=ONLADFV[+:1P$C8'1AA:PT
MM'(?J>XSW5[S_4->YKN#!CNU.0R/&.W_`!2W?E"RRM^/.Y?BH),UC[1LOE^3
MC)QMGE\QG6V=M>W<4UC:7$=Q<2Q&2VG:SD<6@E0[]P
MW'2VPM+*&"YLX@*]P-C]X>Q2QW*)4K8;GX:YN^+\N>_F>2XC99*XMO\`#R'!
MXN6/CO[#9_\`(E=;VD43)9(Z^:X]C\A_CW>X@$6-/RUY
M+':V[W27T;[>#J.'=9Z@]H2C=/5)[$A&X/Q![=]?2;#XFTP&'M,#8`-L;&UB
MMXP.@C@C;$P?8UH7YSN89,WF?4A!4D**A"^X'-[3M[Q:XY+=`.?$-L;
M33WGNZ#73K^!3CM_PNZY[R6WX[:G:)35[OT6CJ=/`!>A7D/Z:7T0_&6^3Q5Y
M(>8G)%?RS4P8,O(8)S"OKIC(L(R9,9V728[QOD3%&)C*@\W'=D%X-+23N"%'
M5O#=U_J*Y59_MGC6#MY\/*YVR0PD@T-"`XW$>[:=-&BE"MHLMVH^GSC-XV201N<"9)*TKK[Q`ZT"K+3L;V=Y)A9\GPZ^N;J
M&)DH>XO!+7LC=(P']7%0';I[NOI*BOB;Z1WT^<4\+>"?)KS5YNY*XAL^3\=Q
M^PN9\;+*FLQ-JZR\6-EC=130DX7?V2E2:".AP$J<"^Q:^X)(&K]G^_?=";GN
M5XKP:SAO&6TSXV4MW2NVQG8YP+7`?&#ZQ4*S8'L?VRM^#X[D_-LE/8275NV1
MS=[&BLE7-^*-[J["VOLZ*3>#OI1?1G\FLBN\(\?/+/E3D?,ZBA=OK"GH>XG`8Y]I!:7EG:.G>]M6QN&M/I(&OL`4(^"?T<9'*O
M'=CY/>;V8S?&GQDJ*)^]BNV$N!C&;Y;7)97[61O.W\.3%PS$^XA4=4EAR=:$
MI2PP$*2XJ6]S_J#CQV:'#>W5L7\_N&X_BP#BQC_=D(UJ7$@$`4%.I/@`I$\6?I>^(?G=RMR?><"9CS
M1@_B)P4\U09)RAG=I03,RYKN%P##6UGR6.TFY?D&UBMXFT-N&TJ9:5W.>7``5`T-.FMXX=V=
MX+S_`#5Q-QZ2X@XKCR3+/+\,P=6@CKH`W82XZZ$*597B/_=VJZ7)KYGFSR'(
MEP7%Q9#T/D%R7#+S*NQYQN7!X?DP'VDJ]7&W%-_8K;5EA[D?4[*&7C,0QUNY
ME0/EB[3K4M#PX>UW57M_;WZ;(B^RDRS_`)ECJ;A,UHKTH"YA!^S\*B[ZF/TH
MO%3QS\2,5\N_%7E?.,MPZYO\'C-0\INJ;*Z3+<;SUJ0:K(<;OJVDH9<-QE2&
MU^TXVZAQI2A^[6D;WKM'WOYES#F4W;_F$$4(FBE!>R)\4D4C!NU)<:#2A`H0
M59.[G9/B'&N%,YAPNZ=(P.:';Y&2->QP.K:-;[U0-?1589T7A#Q1&^G?8>6N
M96><-\@S*ZQF8I25UC7Q\:DN2\P1BV+)E5ZZF3/DB4V%/NA,A'>G8?=ZG6)\
MM]6O57]U:L;:8^5[2QP
ME-8][H"6"4G;(\-:]PUH5KC@,%<\CS]CB[6VEDDNGPLD:RI+:N`<:;13W22=
M5OEE_2F\=%>15!PY5Y'RF]CS7$5_R7E\YVZHE6S,^1EE;BN&PH3Z<>2Q&8G*
M;L7G6W$*4I+"=MMMS\@K+_>*=]9^S5WW'O
M<7RL<'Q.CC:S:X`%VT^Z2#O@[Z5^VA[A,XHV:Z%FW"S3SN,FS8^.>,1G>624
M+V%WYAK2@IU4&7'CU]'W'[FXH+?R?Y#C6U%:V-+;13.>?^4LJJ6]!G1_>B\?
MNQW?9DL*3W(4I)VW!VUE3!][_P#>2\IQ4&;PG#..RXZ=F^-YV"-P`W>\SYUL
MI!`_-/7KI50S)=MOI+PMW+C%FL(/+?#?#/#OX'8\
M3>2LGY+-@B^>S>=D+JU1:9$4P$4\:)[^/8^?>EER0MU8+J4);2#MOK;'Z<>;
M?4=RB&^RWU$8"QQ0V[(FV9:T$[@`YC#FNE5@KNS@NS^&,4?;.ZO
M+_?3WYI""T^N/Y:.OLW!9B^'7TV<"Y$X<8YI\GLLR/CO'LXO:&IXMJJRQKYL&ZJJLK)^VJ[)2WT20&2)GFN@#@#M^78#-.@J:=-E5LXFF3EN02$5R5
M5WX1^(.(1BK*I*@9()<'KMK$?$/JP[C57/4OC+*HV4JK\7QZKM?PQ[2^)-B^MMI
M7N((2R=T]0=9#^I;N=]6/#N78W%_3OQ*UY!QFYB+KJ:>&:4Q&E?BCD92GV*P
M]G.%=E.186>ZY[?RVV4#J!F_:/L_@5L=S_Z;_P!._BC$,;Y!Y%YMY&P_#TW;:'/=Q\#QNSYOANWH**P<4["=B^>('5;(;CPBXGQ'Z=U9
MY795:YNSRED==62*"E;L($;%2]D^6JIJ!+]6[4N3W1^`-F6X!(3W$;@@:TCP
MWU8]QN1_6W-]/.#BL7]NK6Y>R6Z?;2&9H@M6W$_E/8_8X-E<6-+CJ`*K9#(]
MD>,X'Z?H>YV7,L>;GM8WQ,)-'2F[?&UI'H=#1U/0M4"NJE;>FYZ_`]?4=3T.
MOHM)%-`\PW-?F&FCJC::CK5H^$^KP6I#]I>2S1E=/8OG75=4T1-$31$T1-$3
M1$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1
M-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31
M$T1-$31$T1-$31$T1-$31$T1-$7Z22=R23]I.Y_IT))U/5<``=$))VW).WIN
M3T_-]FE2>J4`Z+\W/VZ+E-S]OY?Y=*E%^DD^I)_.=]"2>JX``Z)N3MN2=O3J
M>GYOLTJ3U2@7YN?MT7*_=SOON=_7?<[[_;I4]?%<4'3P3<^NYW]?4^I]3_+I
M4I0+\T7*:(FB)HB:(FB)HB:(O13_`';+':*Q\O>8,BLRPJXQ?@*:WCK"U-_,
M.IR'-L<@73T="B'%+CQ8B$**1NE+VQ("M:G?5S=7T'!;*T;$Y]G/?O+BWJ`R
M(4;]I)(]8TU6V?TE6]D[EU]>2O:RZBLP&!U.KG@$C[-#3TE5[S5^C#]1'GGR
MUY[YBQS$>/,GQODODS(Y0HZX7';H=11M!1;%_&_QPRGZ5OTS>=:?ERWQJ#SWSSD=A
M18_2XY+\/I+%3,4W5O&DSUSWO80MID+605(;4HXCYWRNS[
MQ]SL?=86"Y;QG&6^]Q?U`83)*XDUHVH;2OYS0>JR7Q#BU[VA[;9"US,T'[S9
M*Y;&P-(.C@(V4IU.US^G0&BGKZC68_38XAXN\<_&7SQC9?=8Y3X]56W'6-8?
M%SOL"N.:*)A/XQ9*P6UJWVD)CSUM1T2%*0KN<*1NDG4:[667<[)WT\
M45TY[FO=*80ZKWN>YP,C7'4FII2IU\5*NY>4[:83`V'&>X!E\D01D1QME+=K
M6[!I&0T4VD"NHH*"E%KYXH^HI]%;P3K^0.0_#SBGD^;RID.+O4[5>[29T[)O
M&V'/GZZFD9)R'D<^/CU'(MXS3DI4<*<7[:3[;A2$ZRWG^VGU`=RY+:#F]Y:2
MXJWJ3L,0:-_YSFQ-`=(`T@%VM#2H"Q5A>Z'87@4<]UP>WN3E):-&YK]*>#72
M$N:PEP+@*`T]*SV\D?-BU^GAX$>,'(.4\?4?)W(G+%QC*LRPZ[M'JBOE6V=U
M=QRQR9:PWA`F&7,JK2>IF(A]KL*G4>X1V]<4\3[?2]R.?9C$XU_R]I81%[I&
MBM&0_JF!IJ*-]WP.G@LDC_`%B/"?`N=?#[E'(;>7A*)F0QN%%7*:[',NN(S#2K[",PQ]#P
MCU'*^+.([*YV0HQUA80D^W(:>3?.V_(I.R/<.;`\YL*8Z5\!%X&UG`)?M='*
M1N\EU07`&FM#JTA6[N)Q^/O-P2WS'![T_.6ULZMKN_4O<0TR-=&/=,K:>X2/
M'143Z7/!.?6_T;>;N,>-$1J'G7E&R\DL:EU^2+?H9.-9^^I[`F6D?-&6R:%CZAI]%`?#P6ERH_N_P!]2&PGQZR=@W&5!7J=
MCP7+B9RW12(,6*YLV[8?*5K$RRD,Q&U%7M):#JRGH-;)W?U4]KXV-G8S(.DC
MA+6L`<&N=MH`X5H0/#3KJM?+3Z8>YDLQC>+)L3I07/K3U]H!UZ+85]
M>'-L1X-\1_$?P&PNY@V&08W_``E;9!4P'6A,BXQQAAZ\7QVPLX*"IV"G*LHG
MNR(Z5@*6(RR!L"1AKZ<+*?,[S!%N!_I)I6D,!(\!UIZEE7ZB
M[RPQ7#\/V\LIF27T9C$K&$4/ZLQ`N`\=[J@$>M=3GOR'_P"KS\4_&7&X.`4.
M?6\^@QO"UXWD,V75P6$46'QK>_ME*CP9I??;N)K:.PMC9;O<=NFOF3VB[*O^
MLSZB.>\BOK^_Q&,.3N+L31-+BYT\[Q$T@FFK0X`]0!0:+._,^X]GV&[2\:LV
M6<-]>26443H'O,;3L:3)(:=75V@^FNO11CX;?4;RCRDY[H.*!X^<=8A#D5&1
M9':Y+2VEA/LJ>%15SDAMV/'?I(C!WCD6"L;#="
M^8S-D+W`P`.;HX4\:`]1X+.3C,V=YSEY?*1WGOE6)DWC[
M"%Y;95SDQ?:U&:FY=G'LO.$CVRWN=NT[:@]QHXL+VM[=<4S;[BU^:N[_`"MW
M((F3.MV7TT<,4S8Y0YF]L=N0QY&Y@F<6D$K/^"?=YGFG*N0VK;6YGVPV<4XU+MH.OX%AAA?B#R=
MY6>;N:7GD+QMA?%N%\3,88>2,5XZ<@+PF18Q*"%8T.!5,^N2F')DV\9:9UPY
MW=S$592LI6XC6T?+?J2X=V$^DS%V/8_-7W(N6JRU\JLC\._(^;Q_4SO.S$^+\=XFM&K&EP_"W*.55_Q5326VJ^YFR'RWWN
M4#40,0F6_P!Q'1W%/56XUE[#X;ZD>Q>%R>5;VQN,]F>3VGEONI'!\LEN\E[6
M1[ZNC:Q8;$X:5CF6S6'89(R""`WW0XD?$J3
M]4_CFI\A/%_BSF;CFR@YHG&2'XN,2ID5QI*E+B*R=N"L
M[;);*U=/MOGT#<[O>R/U`\F[<\W#\8+O'33?*/8:6\UH'.C:7]'?JG.>1XAJ
MI/J0XW8=R.V^%YKQB4WV0ANXHQ+&[9M@N`!OH*$B&4-:T=&UIXJ3//'Q(YEY
ME\?.#."N$Z['GJ[`I=.O)47^0-8ZTAC&D%^D'ZC^V/:WO3S7NEW:N+OY^^\_;
M'F/,NV^#X?PHV<<5I+#'(SW6!QCB]PN%*'82=M>E31:3.0_IR^0W#7M/O:$TH/O'CU6FQ)W&VM5?]UCQG*7`YIW6R\6W(Y/(6MD=P]YSS,^[?N)U)=Y\;G5Z
MEK2=0%FOZTLU9VUOQ[B-D\>1:Q^^P:-/34MZ'[0K]^EH]&X,\)?(CR+LX*)2
M56^1WK##SJHK-E6\:XLAN#`&V[N[OU1\1
M[06#;B6,06MOH/O*3?2U<6W;[LWF^X,H:9B^
M^V&,L:'>D-D=N:#H'$D:J%XOUGLNG2(<1CQ4XI=F3WV(S+0R"U4I4V8\EIEH
M=V.*4KW'W-BK;?K^765.W&(]989
MKF'\26U7!"FHL2/B6-I]Z*PS[3(5"BVV1H0C[J5;M#<#/4KYUV1-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31
M$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-
M$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-$31$
MT1-$31$T1-$31$T1-$31$T1-$31$T1-$31$T1-$5W8/R!G7&>10LOXYS+*,#
MRJN#J8.1XA>V6/745M](2^RU8U4F+)$>0D`.-E1;6`.X'8:MF6PV)SUJ++-6
MT-U:`U#)6A[02*5H=*T5RQF9RN%F,^*N);>9P`)C<6D@&H!(UT.JRJ1]23S]
M;2E"/,+R`"4!*4@\B72M@A(2GJIU1.P'J>I^.H:.TG;$:#!8T#_N6J:/[N]R
MI#5^:R!/_?/_`"KIXQ:^=OF1F<>ZQ>Z\A?(K.^,7JW*8$
M?+GE"TQ^K=KZVXS;%LNR>QKJEDT\:^A7CD'$N\/(W#(9NUR5S.&?]L7O<&CT
M5Z#U46O&PAV%1/E5UA$EUEE72W8LZOFL/0K"OGP9"VGHLV(^AN1$F19#:DK;
M<2E:%`@@'<:RQ;S660MQ-:%K[.0`@CHX:T.G7J?O6*KB.]L;@PW.YETTFH/4
M'2OXA]RE3D[R'YWYJK:"GY=Y?Y#Y*J<5<>>QFMS3*;6_@T+K\9J&\[4QI\AU
MJ$ZY$80T5(`/8D)]-6;"\/XMQV>XN<%86MI<7<>R9T3`TRL))+7D=0223[5>
M?>#HUS"X;YEY)XOA9$
M_%DWT/!LON<.6[[7!W]S:VTCMSFQR.:"[TD`]5=^/>
M9GEIB629%F&+^2',V/Y-E\UFRRNXJ<_R"%(R.SCQ6H+-G=-LS$Q;*R1#80U\
MPZVIXH0D%1`&K?<=N.!W=O':W.(L)+>($,:Z)I#0>H;Z`:=.BK[7N)SBRN)+
MNTREY'/+3<6RN&ZG2M#K2I4@/?4=\^);#K$CS`\@'6%@!QO_`%C7K?=UV'WV
MI#;B=OR*&K:>TW:^V8ZY&!QA+?#R6ZU5W'=ON;=N;:NSF0VN/^>?I^%0Q+Q+
MG[DC&^R?%L?BL!:D,-U+
M%$&";9N+!(0=:%SBVO2I5RON2\X[D7%KCR+CWQR\H,-O)$%VKD6^,8;DM/9?A\EQEQZ`9D8-._+/.
ML(*T`[**1OJ*U_RTS0]NZ,U:\M)I5AU%5*<%Q#O
M#QFY=><=L\GCKZ)I87L+FDA^A%:`G=3PT4=9+F_G)P9E<_*^]'&;I]O/?Y*SO+JYA
MDEEVM<]WI*MB;Q!RA6\8TW,T[CW+87$M[=2<9Q_DE^HD,X?:Y!#,Q+])7VY2(K\]E4!
M\%M)W!97^R=3ZPY3@&YY_'RT'S">U([7ME#8@'40R79SM7E^02\MR7',9=6QHD$@HT:4:\!]/2%D!QER
MMY^\V7EKCG$7(?DCR-D5/22\EMZ3#\@R>]L(&/PG66YMQ-8B25.-5T9Y]"5N
M*Z)4L?$ZQ_GNR'TP\5$5UR'C&"Q\DE(F-EM6.KN<*,K_`"G?E4YPO<+OCR6>
M6'`Y?)W?E`RO=',YM`UIJ\^.C0?L4&Y#Y$\^Y1+Q^;D_,/(]Y-P^[;R'&'[?
M*[>:_CF0QDJ81;U2I$A9@V;""I`=1LM(W&^IIQ_LQV?X[:W+N-\:P]E;WUJ^
MVN&PVS&B:WD-7POH/>8XZD>*A^2[C]PK^6WAR67OIG6=QY\.^5SO+F'21E3H
M_P!:LC.>2>0.3K=F_P"19&,8T'T-
M"L'(>4\BY9=B_P"27D][>-Z/E<7N'VE56'S+RS7X+)XP@#Q
M;^Q8Q>2U-E"=+;?IFWTPG4RIB0ZX"D]Z^IWU;KSM=VYR'+CSZ^PN/EYJ9FRF
M]=$#<>8QGE,?YG7Y#K+[:5)(ZA0!U-;
MBUM[NT=8W3!)9NB?&6.U:8Y!MD81^B\&CAXA16WEEMI63VY+)HWA[2-"'#H1
MZPI"Y&Y2Y7Y,F0/]:V=YIF$^B;?8KOXRM[.VE5:)GLNR68K=DZM<5,H(;6L)
M`[P$D[]-0;@O;KMUVYCN[+M[AK##PWDC77#;6(1B9\371L=)2NXL8YS6^@$J
M5B*(LC\UY=L8YS7EK:]`7-::#Q`4;[)'JM)/VE*]S^76
M0C4FKC5RB/L39OXK`_*$K)_F)`UPB_>UK_PO\[9_X"3HB=C?_AA_B+_1HB=K
M7_A?YFS_`,)!T1.UK_PI_P#L1_\`KM<)5.QHG_+J'_QHD?\`J8URN:K]]MG_
M`-:#_P#85#_`L:X2H]"_0U'^,E8_.P3_`('=GL1?_`%J4/_P=?_"[I4KDRO\`T!]ZY4L0]AO,3Z?^LJ]_Y_NVZVA_/^'R-_P"A
MTC1A?ORM8?_ZL1^0UT@_TAT:)[J^A%JOC;G\XK)/_`-^UR$T7[\G4?^OI
MS_Y%/?\`&-<)IZ/PKZ^2ISZWI'_Y(E_\$C35`6^(_$N3Y*D_]?R__D/)_P",
M:+BOJ_$@@4?_`"@<_P#D-)/^&1HN:CT!?:8%#O\`>R!P?_D22>O\D@:ICN3K2/R8[-/_P#%C3YB3]'\:]FPQN:'%U"5
M]?AN-_\`*ES_`-MN9_QO3YB3]$?A7;Y>/])?HK<:W&^4N;?'_P!EN9_QS3YB
M3]$?A7'R\?Z2^_PS%S_\]3H_+_#=AT_+LF9UUS\S)^B%V$4+11QJ4_"L7'_S
MW.?^VQ8?_!3=]<&Y?X-_`5P88G&K74"?A6+[C_V;W1_]#$__`(]I\Q)^BGD1
M_I+[-7BF_3,'?R]V*V!V_-VS=M/F9/T5V;%"T>\X53\)Q3_ED\?_`*$[(?X)
MO77/S,GZ(7.R#TA!3XE_RT?_`)<4LO\`@FZ?,R?H!<>7!Z0N3\(P_P#Y:O)/
MY<2LE;__`([TT^9D/Y@7!BA=T(7[^#X>?3-W=_\`VD+3_CYUT=/,X4:VBX$,
M3>KD_!G\=/)_+_!MF?Z/G]=_F)/T5WC@B#JN=HOT4F%[
M]<]=[?\`VC+3?_V(?;K@W$KA1K-54>3:_I!/P/"BK_Z8#R4_^T3:G;I__<-=
M/.N6>^6UIX+D06ATW`+[_`L'_P"<-X_D_@>U_P!(:?M"?_,C[ES\I8C5\GN^
M.J^OP/"/^<%[_P!L:T_TAK@W]P11D0W^'M\%S\OAO\](GX%@Y_\`TA/C_P"@
M:T_X_KQ^9S?^8C7/R^%_STB^TT6"C?NY">/Y\&M?^">-/F[!'3UI\OA?\`/R?P^Q/X?X__`._P!GUQ_P6F^GS.;_`,Q$GR^%_P`](N08]Q[L-^2U
M_P`O'EV3_/\`BFGS.;_S$7\/M7'RV%_S\J_?X=X[/KR8[_)Q[=@?S?B>GS.;
M_P`Q%_#[5S\OA?\`/2?P^Q/X=X[_`.EKK@W6;_S$?\/M7JRSP[A42O(_
MFN/X00%])QKCC^MR@YZ_#CJ\/3_Y*#7'S6;_`,Q&N_R6(_SC_P#(D_ZR^CC/
M&WPY3='_`+K>]/\`\M=/FLW_`)B-/DL1_G'_`.0__K(<8XV^'*;H_P#=;WA_
M^6NNIN\T/^QB'VM'XP4^2Q'^,E^O+#B-O3?CF\1O_`#VI
MWVUP;W-C_L8C]L9__A3Y##NZW$C/9$]]?^=HAQ/C+_G_P"EM/GLM_JUM_:F?]5=/V=A
M?];E_L[_`/K+D&(\6=-^8%_E'^KF^_TMMI\]EO\`5K;^U,_ZJX_9N&K_`.R_A;6X/JN&//^3MU3]E8;_6YO[,[_K+Z&'<4[=>9G0?_
M`#9WY'_L7UQ\]FO&"(CUO8T?Y6W3VKC]E8;_`%N;^S._ZR_?X.XI_P">AP_^
MZQO_`/2^AOLM0TMK:O\`ZMG_`%5V_96%.GSDW]F?_P!9?G\'\4$C?F=X=1T_
MU89`?\%SKP^>S7^JV[O4+ME3ZA[O4]`N?V/@Z?\`G9O[,_\`ZRYAAO$A]>:7
MQ^0<6Y#U'Y_QOIKO\]G?[GD_MS/^JN/V/@QJ;Z;^S/\`^LOO^#N'O^>2;_LQ
MR+_36NOSO(/[H=_;6?\`57;]G\<_O1O]E?\`]9?2<.X>/1?-$Q()3NH<6Y"2
M!\2-KK39N(9JT'KZ
M5U-=EYIHB:(FB)HB:(FB)HB:(FB)HB:(FB)HB]+/]VW4^.2/,7Y>VCT3Z.#L
M;5&NW@@M4L@9#<*9MY(<(;4Q6.@/D*Z=J#OZZU!^K4M?B\)'-"9X6WKCL:/>
M?[M=H(UJ:4'K6WGTG-(R6;?&Z6*4V31YG_9M]^@-#^<*U_F@K:%X<,>0;WD/
M@+F5?67XG\K*"%_$$NUX'PZIX]1?YY%106:6FH[U7L+5OP!XL\*>9OU+_J4Y5Y'
MX#.IZ#A^=G?(#7$]KDT'%X>'WMM)/?,AB?*UNZ2.L;7%L;7^[YAK7WAXTTZK#G&.#
M\:YQW:Y$[E%G<16]DZ:9D1<6QNVR$5DW&0Y)C[6UAREO(&M=833S1CWJ`/$S1M<14D"HZZK[Y(XQ^FKX?^+7
M@?S%RUXDY;SCG_DAP\+W(X-7R_DN'T*9<*MQ:=D&628[DN8V[;?,7:&X<..(
M\9*''"KJ$G77"9GO-W!Y;R#$83-6MA:8R]=MJQA`8#+MC95AJRC-2XN<:=5Z
M9G"=HN!\6P&6R^(NKRYR5D=WON!+OU=7NHX`.]XT`HW4:*T_)CZ=/!]!]3;Q
M;X(X5P;)++A_R*PC$.5)?&#V<2::SK:E]G)IN2U4+.+-BXL*FK774*'U;IE2
M&=W6V%%1;":_B/=KEU]V=S/(LO=63V4=Q)"'T-'EW1YJ6@;?>ZT6P_$OI_?3AYFSCG
M7A&IXJX#JKWCW!LUG_-\-^0G/&4*@GR7L+GN:1TJUNAK
M6NBRE8]LNV.3O;[&,L\8;RUM)'@07-Q(]E&N#1*QX:T/:ZA=MX[_9K(?>\AW:W/ASV,>ZW!!>*^_M813^5
MJ:?:L;]E6N=W-Q%&R.;'=MT9H0W<[KZJTJMS/U#^<_K(XMYB\Y8]XV.>3@X4
MKKZH;X^_@7B1C(L656JPV@?FBFN%85:*G1_QIR3W*]]PI=*D[[`)U@/MA@^P
MEWVZQMQRPXE_("'^<7RRM>[4@;@R5H'V`+8#N7G^^=CS[(P<:=E!@@]OE!D<
M98`:?"3&:@FOB3Z^BY_K,IS'(^&?I*7'-=<]78CF;R!TT39WO#6N-=@.\-`%=U3H!U4,^6/&
M'BWAGUJ\>X;\C*3*.0N"<\PGBW#K*PSCD[/;B_QO(\SI%L8OD;^:V&0JR%=7
M77R&8[K#TI;#,60M24CM&TDX/DN;7/8:[RW$93;YFSGED_5LC_6,C?ND`:6E
MM1&''0`DCUJ/\TQ/#;+OE:XCE;!/A;N")A+GNK&Y\>UAW-(.CRWU`==`NGG_
M`-,OA#PAX:\_>:_*;#)F8P,4Y!_U5^%M).R.]HAD\O(HRK'%\N>-)8Q)%PS"
M;OH[4E+O>V4T;&-[2-E
M`""-=SQJJ?+]HL!V[X_R'/\`,(&2,;,8<8TNZX$T;J0=!1U?!8V\
MA\94D;Z(OC?R5^)YP]D%[Y2Y1CTRJM`37P!-*JPY7#6CN
MPV+O6R!OG9.AJX[6-)`^RFM3Z%MG@^!OTRZV1PGA^+<>>-V02,UQ/%GK&)S_
M`.1O._%?/&19+>*$$1Z+"&,:F05/3Y^S;276XX$DJ;2V$I!.&).Y7=R^9E+^
M6ZSK+>UDD]^SL[66RAC94D2RF02,VM!)+=S@*$5(66X.W7:ZT9C;*VML&^XN
M8HZMO+VYCNY)'T`\N)K-C@YQHUIHUQT)H:K%WP@\1<5P#ZHOF=P'94V;<84&
M!<$YCD&.8Y@O->7BPK:FSD8):T]>_P`AXR[BMSD]*[$ME*$>:R"`4)?2M;?<
MJ9\TYM>9CLUQO,,G-UE9K\-EFEC:XNVO(&C@0-1U&OH-%$>&\*M,;W:Y#B9X
M1;6$%C(Z.*&1P`K'44H03H>AT/2BQS\+O&_Q8J_IZ;_-OCEF7E/EN+\W'
MC6CXSQK+,HQV'64A5C4,VLI&,%Z4?8E7KC\R2ZT^4M-H2A*-U+-_YYS'F,_<
M2QX%@LS:8;&/QS)7S-#2USR"229`[4TTI0=?8K+PCAW$HN`WO-\UBKK+9%N0
M,38JN!:P=!2,@FGYQ]E%Q^;/C;XJV_T\.'O-OA+QRS+Q6S')^:W.,KSC?(\M
MR;)8-K2)BY0V;=EO*0S*[9,G'6WH4AEN/WMN.)4E?W5"I[:\UYFSNA>\%SF8
MMR=S6-:UU&D$&,"I&[6M>GV*B[@\/XA=]N\;S3$8BYQ-[-D!;/AW/
M.YOO?K")"2/AH#T)/H66G/7"'TLO%3R5\=O'G)_#3->2;;R$Q+B*9.R5KF;)
MZS'\&7F]RO&&I];2.V:["YGS+%2Y,T*E-(0VE"&=CN##N*Y_O?SCBV6Y)8YZ
MUMHL+)'OZO
M=M)^/J?SO1X46E7ZD/CS@OBMYJ\X\$\<&Q_@3#K>CF8RU"XS+9;7(/;,V633WW1N+=
MV@`\*B@"UZ[N<6L.(*Q0#;"*5AB8"3L:\`AM2232M.JQMY708]MB,!]2
MW+&OXOXYCV[CVRGU3Y%`W:-MOK*BXIZ'66$:.0K<@,@?`:FO'FE\,\TCB]_G
MOH2*4%:`#U:5!\:J&9L;'PQM&UAC;4>N@46'UU>VDEH)ZT5C>`UY`Z`K\UV7
M5-$31$T1-$31$T1-$3?;7"Y!(Z+]W/\`V`:43<4W/V_S=-*!I2B;G\G
M\P_1KBI2B;G_`+`--Q"43O]M7^,?TZ4
M"5]0^X)WK_;5_C'].N=1T)'L)'XD^P?<$[U'U43^?K_ATJ[])W^4?RI[-/9I
M^)?GG3^0?HTW.\23[23^.J5)\3]Z_>]
M7V_T#2OJ'W!/O3O5]O\`0-*^H?<$^].]7V_T#]&E?4/N"?:?O7YW'_L`_1I7
MV?<$J?2?O*=Q_P"P#]&FX^&A]6A^\:I4^D_>4W/Y/YA^C3<_])Q'K)(^XE*G
MTG[ROWN/Y/YA^C2OL^X)KZ3]Y3O5^3_%3^C773U)KZ3]Y3O5]O\`0/T:XIZW
M?>?RIKZ3]Y7YW*_:5_.=[U?L_>?RIO=ZON'
MY%^:Y75-$31$T1-$31$T1-$31$T1-$31$T1-$57HF:9^UAL9!,E5]3(<4S+L
M8D8S7JX.-N(:F_(I*5S6(K_:MUI*@XMON"-U;`T5U+-"TR1Z@>&BJK9D4K@Q
MW7TK+OA#E?FCQE7**;@YG6E##*`?6"#X:!6#Q%;9_P`#
M(JL%XYD]=D,F1,JU6U;.I[%33%CB,R$Y\W6V3[*N]M0[7#\=B+ER&"+E6'DP
M.?Q\MQB9=NYAD`KM>'M]YMT':.`.A]NBHN/9"[XKEH\Y@KYL.4B+MC]KG[=S
M"PZ/MG-^%Q'32NFJE'$O(#R5P+FW*/(W#/*["\:YIS>38RLRSBJRIZ/)RE=N
M\Q)L6;ZJ_@U5%91)4F,VX67(I;2XA*D@$`ZLV1XMQS*\?@XM?X..3!6S6MBC
M]P;-HHTAXN@_RR\K,Y.YSI)*O.\N.YP+3;["T
MDUV;=OJ4@\W^:7F9Y&8+*XQY>\P>/\DX^GRXTZTQ"!85>*TES)B2D38JKJ+B
MO'U,;5N/,:0ZEM\K;]Q"5%)*01:^.=O^'<2R<>9P6",>4BKMD?(9G"NG_:WC
MZZ::CIHKIG^X?,>3XY^(S.8\S&2$;HVQB)IIJ#2*U937T=5!O)>?0_&>58?PC0R<8XLI)F0.QH^'T,QJL9DUT%V!AT65(:=:IHJ=Y"WE@,
MIV(Z[R+$XC%8*]O9<.!8?,?[VM#=$-^-VC0`:ZA1W+9O*YRRL
M\?ERGE/C;FVZ\L,&D-\U8R(0+7%J"L3-;BU41,'"8\.3$]JQ?0XF0T][R'5)<[@=M6Z#B/&;;
M#77'H<&P8:]E,LT1+2'O-*NJ;HD'04VD;?"BKY^7+2=D..]RV@I781W'>+VW:#MS:!HAXZ1L>'-K.]Q:6FHH3>F@!
M'0:>I2BY[P=Q+LDS9P511Q%`-/MH/LC<$;[S
MJYQ.-N^36_,+C%.=R.UC='%-N:"UCFO:6[1=!A!$CQJTG6O@%"+7-9.RX[/Q
M.VR`;Q^YD:^2+:2'.8YKFG<;8N!!8TZ.'3UE6AQL[F7$.>8OR=QOS9Q5BN>8
M79HN<7R.%D'L>TR`AP-0?Z#T^!T6
M>G_6A?4EWZ>?.*^@&PG8\D=/_=:;;ZQ:.S';)C0UO&@&CP$S_P#^]63_`/&?
MN:22>0R:_P#LV_\`]II]BQ5YGYF\A/(?-,>Y"YL\IL+Y&R[$4QF\5L[_`#"6
MMK&T1+!FU:125D/$(E57A=C';>=+;`4\M"?<*@D`3CCO'<#Q3&W.*X_A&6UE
M>-VR@;'.>#H1YCKISQ]C@H=G^4<@Y1?P9+/9:2XN[9P=&7;P&D&H.T6X:?M"
MMGFK,^5O(GD&5RMS/Y!\9YQR%,@U%9(RB;DOL/P]-4)[BJS"H+UA,37@MAR0IU0"UD'=:B;5QSB7&^)7DV0
MX[A1;7EP*2/:X$D;MU!NNSM%3J&T!Z'0*Y\AY?R/E=E!CN099US96_\`1L$L2RF5FV.X"<>`/P<>KX]742+A&+7US%A1&DL1DF^N.,9MR^\RVA(#KCRG20"5$]=06
MY[0=N+NX?UPH11UL6BH\6@$>"
M[WCSY1>5?BE!OJG@#RSP+CVER>P%M>T,6\9N**=;!E,TASVN?Z:26S@/L`7QY$>3WE3Y80:*H\@?+/!.1*/&IJ
MK.DQ^7?-5&/P;13"HOXH*;'\'JX#]DF*XIM+SJ%K0A:@DCN5OVXOQ#C'#+B2
M[XW@F6]S+$8WN);(2P]162Z?2OI%"NO*.8\FYE#%;\CR[[BW@-6-`=&UI]-(
M[9H)]9!.G56ER3S'SWR]R)@O+7(_DMQOE/(O&D'&J[!%%2Y7D>82YQ)<]Y-22>I]%?%
M4>6R?[=SY'>_-7LQ!D=M:W<6@`:-:T"E/!H]BQMS3*9F:Y9?Y7-89B/WE
MB],3!C;F+6Q!VLP*N*I0"_E:R`TU':W`/8V/CJ:X^S;CK5MLT4>&@.];J>\?
MM->BAV0NWWETZ0NW,#CMZ#W:Z=`/!6QJL`H*#HJ(DDU/5-ZY^V=*N_2*53W7/VSI5WZ12J>ZY^V=*N_2*53W7/VSI5WZ12J>ZY^V=*N_
M2*53W7/VSI5WZ12J>ZY^V=*N_2*53W7/VSI5WZ12J>ZY^V=*N_2*53W7/VSI
M5WZ12J>ZY^V=*N_2*53W7/VSI5WZ12J>ZY^V=*N_2*53W7/VSI5WZ12J>ZY^
MV=*N_2*53W7/VSI5WZ12J^"HD[D]?MUVWNV[:_;X_?U7=LKF,,8`VGT@%?FN
MB\TURB:(FB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)
MHB:(FB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)HB:(
MFB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)H
MB:(FB)HB:(FB)[C/[8_QD?IUY5=Z_N'Y5[>6?X5_(GN,_MC_`!D?ITJ[U_XS^V/\9'Z=*N]?W#\J>6?X5_(GN,_MC_&1^G2KO7]P_*GEG^%?
MR)[C/[8_QD?ITJ[U_XS^V/\`&1^G2KO7]P_*GEG^%?R)[C/[
M8_QD?ITJ[U_XS^V/\9'Z=*N]?W#\J>6?X5_(GN,_MC_&1^G2
MKO7]P_*GEG^%?R)[C/[8_P`9'Z=*N]?W#\J>6?X5_(GN,_MC_&1^G2KO7]P_
M*GEG^%?R)[C/[8_QD?ITJ[U_XS^V/\9'Z=*N]?W#\J>6?X5_
M(GN,_MC_`!D?ITJ[U_XS^V/\9'Z=*N]?W#\J>6?X5_(GN,_M
MC_&1^G2KO7]P_*GEG^%?R)[C/[8_QD?ITJ[U_XS^V/\`&1^G
M2KO7]P_*GEG^%?R+?9]+;P_\6>>N*T93SMQR_J3L-M$^_W>WG_`^[3.,X'(_*X-V,MYO+,,#_UD
MCY@YVY\;G:AC=-U!30=5OOV$['=ON<]G'\ISN.-SR!N4N(?,$]PS]6R.!S&[
M&2-9H7N-=M374]*;9)'TE/`AFP*D<">]`]Q2FVVN1>4"7&MP$[._Q<=P>X?E
M.VHL>_'=!S6OBRPPG;$$M?BG-<#36XN=?7_`$JO2H^D/].Z
MRBEX^/+C2^]:0%\F\IJ4"D].HS!`V/YNFO%_?KNL#093_P"'MOZI3/<0<-+HM'F`>#2;J[RUBWXKME4F+NL+O
M)"Y&2QY]*4N+0[`CM-RT",2V68Z$@G92.Y1/38:D/%OJ'^H'(
MR_-9',$6ATV&TM!K7K40`]/6L)]UOIV^DKC=LS'\8P>_*^8:O%]?N]S:?!UR
M6_%3P69T[Z17@0KM+'"CL8!)W[.0.25A1/4;^YEJU`C62W=^.ZOF@-RGN;?]
M7MNO_A+6W_`CM;(TNCQ9'O:#YBYZ?^,J8U])#P.87[CW#+LA!`/M.9[R.CT]
M=E-Y8%'?\NN']^.ZP;493_X>V_JERSL-VOK[^,-/_47/]:J+)^E/X(I-DMOA
M!0;84VEO_P!R#R2?;[O7URS=6^_QUY#OUW7_`+T_^'MOZI5$?87M2YU#BS3_
M`-1<_P!O3[,L_+KG_'KNM_>G_P]M_5+U?V
M#[3AI(Q9K_ZFY_KE39/TM_!EM12GA(C91'_S?\C'X?:NZ_P#>G_P]
MM_5*G_P'[5?W8?[1<_UJH,KZ8?A$V"4<,%/KL/X\Y#/IO]N4G0=^NZ_]Z?\`
MP]M_5+LWL-VJ+@#BS2O^L7/]G_MT;===O\>NZ
M_P#>G_P]M_5*I_P"[3_W6?[3=?URI4WZ:OA>P%=O#I!ZGIG/(!VV_/DQZ:'O
MWW7I_P#5/_A[;^J7'^`7:?\`NL_VFY_KECYEG@;XJUMDXQ"XQ^782H;(_B_-
M%_=.^W5R_4KKMJW3?4%W<8=,KX_ZO;?U*]H?I_[2O!+L4:_^INOZY6=-\'O&
MML#V>-%I*@`@#*&+EIQ]'#]@(ZBL,2'\BR^*ROMVZMK=NT>XG-S]2'DGTY=LW_`-#A7_VBZ_KE(.!_38\=(60N.\@\
M5MRZ`L("83&:Y>F8A]95VGMA9`Q(Z`@=3MTUX8_ZK>6N=2[S%:?_`,O;C\42
M]+GZ8NWA:#;XL@T_S]R?QRJ?G?`WZ9]>TA=IP;>,)2$>^^G*.67VFR=]^]3&
M6*V*=]U;#8)V.I);?5%F[DT_:^W_`/`@_JU:7_3+Q6#WOV.7M/3]?<_UJNJC
M^G'],S)]OP'CBFL5K0IU,>/RIR$980A7:M2H;F8)DI#:_NJW3]T^NI39]^.4
MW8#H\PTM(J/U5O\`U:LUUV"X3;$^9AG@CP\^Y_K57'/I6>!*@%,<(DME.Z5?
MZPN2?O[CN3V[Y9][N3U&WJ-7%O=SN#+NDM\HUT8]$-OT_P##5N9V9[;L!9<8
MIS9/"L]S_6JCR/I8>##:P!PDH?>/_P`_W(Y^[Z=3_%1Z;ZZM[P=PG#<,AI_W
M,']6N&]E>VP%'8\D_P#?W']:J5:?2X\'XZ$J9X64C<=3_'G(JO\`U+*CTU[-
M[N=PBW6__P#R8/ZM=3V7[;`__3C_`./M]WM<2%.R3M
M_P"SIG9ZC?[C_OE7&OHX?3F6$__`*ORB2>O
M_N2^5AT^WIF?0:Z?[0_>#^]O_AK7^I7?_9Y[0?W2?[3=?URJ+?T:?IPD#N\>
MR?O;?_3,Y7'\_P#[.G3KI_M#]X/[V_\`AK7^I7'^SQV@_ND_VFZ_KE5(_P!%
M_P"FXYZ^/)(!V)/)_*Z?7\^;`#3_`&A^\'][?_#6O]2G^SQV@_ND_P!INOZY
M=M'T7OIL.J]MKQS*COU7_K0Y;_E`_P#9WV`'VGU^&G^T/W@_O;_X:U_J4_V>
M.T']TG^TW7]./#$G'N2,(E4V4OEC,^0\A-MBE=.0<
MHJE5]SDMFQLNL475.MH2ZVVA:DJ!'6V9'Z@>^PU]R1ECR'"F6RE^%OS=XW7VMG:>JU`T'C)XI9Y2.0:'%Y%5FDZ`TN!6
MNWN5A4:Y80'950I^7:&&](4M):(`5ND%:-^@.N8^J_ZL<7DY;;+(G!U544>1N\6
MIL3038:LJQ=W),QC3_\`D;CY6XY"`X]#\G9#K_\`LZQUE/IU^G/'8"&\FXQY>7M7[7--]?[+LO\`
MS9#\S^J='_V?E[=]?>JLA.*?"KPUSZ(JHF<3OQ;:P9D/X]<,9GG#K=G&>651
MTQA^/-QG;2$E:4EE2![R02`%!0%9G?J.^JG"/%S9YKY^S=3X;.QTKX>[;@Z#
M\:].WO8[Z1>09"7$\CP(L;R&8M(FR5Y$2*-=0AUR/20"-"*'K59=8-]$;@PU
M$O,.3,7J,7PN*I+RYD_*<_C6Z:UO9QG*68.?R$L5^_X0(+(-VT'Q-,&[K74:4]:EG/.S/T4\=L9!C>'MDN(A3?'
ME,E*UQ_2J+PMKZAHLGL%^F']('.9RL>QW@?);>^B1`L19&?\MPG;;VF.]Y^-
M(]\>^-N7SQ9MDMHTZEEI;C9ZB7P"I/6HT6M
MMCV=[(YASI+'";6.<:--U>>Z/`"MP3]Y)4GS_HQ?32K4(E+\8V(\-LE=@Y<<
MV\K1C"@):<6J:IEC,'2I7>D)[>XCK_(;*SZCN[334YZ5[R?A^3M*`^T6X51/
MV$[/L=M?B=A_]5=&O_YY6/O*_P!.#Z6O',B%5L>-U#>6UM!WX]\4(W%3%PPEC&$YIR=RC8Y'>QWUH!RB-CLG+G
M'5PI#BDMP$`*^YWN.I)6VD4F?^K[GN!P<=G=<@C'/G.$D]F+2WK:Q;2`TO,.
MQVYQ:3U<#T-*JVWOT^<(?:?,X7BHDQI>`+N2^NA'(7`[(H&"
MT-%*T.O3G'Q9XVXHI<6#K#"LNOY+TVPQYJPLE/8[4)B#Y>'8ERR>*[1',_P">_P":W\B^QQOAVVYINFW0_.SOM`_]:?M.G[+L
M?T/PN_*G[Q9G_/?\UOY%5K#BS"6(<.2U2=H=4XVZ?GK!6ZPE"V_UI1`W2H[_
M`)M/V78T_H_PN_*J9O)LV9MGG>[_`#6?]54;_5UAO_KG']MG_P#&=/V78_H?
MA/Y54_O#F?\`/?\`-;^15:GXLPN=,;:72]S8V*Q\]/3W=1TW$I)'3[-!B[&O
MP?A=^5=).29EC"[SN@_1;^12%E7!W'57BL>TB8Y[4Q5DS'6O\3M5]S*VGRH=
MCDU;8V4E/PWUX0XZS=(6N9I[3^56FVYAG);@QNGTK^BS\BBC_5QAW_KF_P#Q
MR?\`\9U[_LNQ_0_"?RJ\_O%F/&;3^:W\BJ#'%6(.^RK\(W2XH$_YY/&Z?C_Z
ML[Z?LNPK\'X3^5#R7*TTF_YK?^JON?Q?A,5#1%-L5+*3O.L#_5.W_JT?CKG]
MEV-:;/PG\JZP\CS+QK-_S6_]54O_`%=8;_ZYQ_;9_P#QG7'[+L?T/PG\J[?O
M#F?\]_S6_D7.[QGA[3;*U4W^62I:?\]G=$A12"!\SN0O;<'3]EV/Z'X3^5/W
MBS'^>_YK?R+Y;XUPYPG_`,D`)2DK4?G9W1*!N0?\YW`5Z:?LNQ_0_"[\J?O%
MF?\`/?\`-;^1=EKC'"U17Y"J;<)(:9_SV?LMS;=2DD2ME=H'IKG]F6'^;_"[
M\J?O+E1IYO\`S6_]5=\\3X5%J539=*??D)6[&09U@D-LI3NGH)0[E.'J-^G;
MH,78'\S\+ORJE')\V97-\[W:_HL_(MH/'_@_XT27..FJO-MF\M'FX[&[EK#)+!0;6_#)$''4"O4U5[X5X$^+-Q%Y#K
M+#B\N75-C4^WI7_XMS1"H\NBL(CUC'++=\AM_P!ZK3('WDJ[0@D;$#5KY1F,
MEBY\9>VTM,7-*QDHHT[M[2&FI!(][;T(ZJXX+*9"]@S5G<2%V2M;;S(30#:6
MO#SH``ZL+PW4'X:]:E''AW`Q@6-YP*QD11>.T]DX,]Y#KI[#$F`W)A2(
M3T3)DQ8\A*V']BXP\V=ANDZ\,Y^]$^<%EB;TV\,D(>P>7&X5&AKN:21J/S@1
MZ55XOE]C;8&:[R<9GN89@TN!+:CKT;0:CU*_W_IT^$MW;XO/Q''(L"'<1:F>
MYAN8YGF%;*?CHD*A63,3)TWS=5*?D/PGC]]<(I*MDCH-15O*^88BVNL?GXY9
M9K5SA\U;1M>:D;F^;%2C6@.&K6G0:FNJED[;+,S6V2X[>16\-W&#\M<$!K2!
MM/ER$C>7$%WZV:,U-`"**.;/Z=7`^-6^64>1<.V$*3#J;6;2R)&0YHTV\:N4
MW+^8AJ%V&IS$JKCO=I!<3VJWWW`.I59\TL,G;V%]9W49#Y`V9C:'1P(&XD5:
MX.I4"BB=_;QW3W.;^I>^-HB;IJ0YK1N'K)#WBLO,J*"CC%Q%-D-;4.QF3EF7DLR;BJ1'[TN_CQ=(C7:BH)4HC9)2
M=QJU.OLD,$^Y#ZWMK1=FFQQR4L)8&/:*
M-\1M<:TK\;7>/J75J/![QAGX+G,Q7&05D.+S,=GHD'*\R)%1*F3:>TCF.B^#
M*_\`/I$57<4]R=M@0"=>]_DY8>1XD1O(PM^)8RV@H9=K7QG<1N%`':`T->A5
M;9Y6^N,'EY@^N0L]DC#0>[$*B04I0U)&I!(\"KJI?$?PF9:PZ/E'`CK]?>1(
MRI%Y3\@9_`MXLYFPDU<[WVI61RZJ5%[V4O+:#3+@2L=KB=QJR7UIS*>YR$^(
MOVL%K*[;$^)CFN86AS`"&AXTJ*U=KU!HKK9#O,UH:`*,;7P
M<\>J"XPEJZXHE0&)Y1#OZ^5D.:19"9<6U>@SG'6W[E$B,%1UMN`#MZ[;#KJ7
MV^6M\AB;Z;&7+97PD/AE]WWFN%0RE*&FHK2M%8+F\SF/?;S7D9-L:M?IM)<*
M:F/5S/\`+(/2@HK5<\,_'F/7YM%?X]4FXQN;$<;>.294"F&Q9.U-DU[/XSV*
M27Y+*^X@J3V?`$ZDL0@GNL;=MHZ'2H'J5OBS&2\NX$S
MB)8P'C0:,)IZ/^%5_&?#'QAL,TP&%,X[#M'F%9%AK:7E&7H#5Q)3.HW)29#5
MXEP>S\Z@^`O:YONG3^C);TKX]=5
M><1?S7>7M8)G_P"BS-VD:#WW`M:ZHH?=)#J5IIKHNJSX3\+T.,.9)9<(KMD5
MF4_(OJG7V=-PK""N,^RN-W0\@AA0C3X*@2TM+@4[L3ML->E]=XR^RO[)LKYU
MO*^P$@\L,>X25%"0]KM'!PT(III14\.9R4-C!DIX6FV-R[>9*M:8PYT>RK:>
M\"PN%-:.U\%=-EX4^&&57!J\>Q>VXXMY%.W#Q_[1RUO;Y+&1S&-SFDQW-2\L;5C2(R`=
M:@-J%(G7.$S-R66%U/:.>S
M/8^#D&CC$\/#PS\XT<:$?RA4>M>-IC\O$\ON)!/8ZTD``J:5&C1[?#P6LCW&
M?VQ_C(_3KK5WK^X?E5=Y9_A7\B>XS^V/\9'Z=*N]?W#\J>6?X5_(GN,_MC_&
M1^G2KO7]P_*GEG^%?R)[C/[8_P`9'Z=*N]?W#\J>6?X5_(GN,_MC_&1^G2KO
M7]P_*GEG^%?R)[C/[8_QD?ITJ[U_XS^V/\9'Z=*N]?W#\J>6
M?X5_(GN,_MC_`!D?ITJ[U_XS^V/\9'Z=*N]?W#\J>6?X5_(G
MN,_MC_&1^G2KO7]P_*GEG^%?R)[C/[8_QD?ITJ[U_XS^V/\`
M&1^G2KO7]P_*GEG^%?R)[C/[8_QD?ITJ[U_XS^V/\9'Z=*N]
M?W#\J>6?X5_(GN,_MC_&1^G2KO7]P_*GEG^%?R)[C/[8_P`9'Z=*N]?W#\J>
M6?X5_(GN,_MC_&1^G2KO7]P_*GEG^%?R)[C/[8_QD?ITJ[U_8_MMS_6KZ]_X?\$_N7$_V.V_J
MD_W3_&3_`*)7C'_L.P'_`$3I^_O._P"^\Q_;;G^M3_#_`()_8_MMS_6I_A_P3^Y<3_8[;^J3_`'3_`!D_
MZ)7C'_L.P'_1.G[^\[_OO,?VVY_K4_P_X)_=_P!]YC^VW/\`6I_A_P`$_N7$_P!CMOZI/]T_QD_Z)7C'_L.P
M'_1.G[^\[_OO,?VVY_K4_P`/^"?W+B?[';?U2?[I_C)_T2O&/_8=@/\`HG3]
M_>=_WWF/[;<_UJ?X?\$_N7$_V.V_JD_W3_&3_HE>,?\`L.P'_1.G[^\[_OO,
M?VVY_K4_P_X)_=_WWF/[;<_UJ?X?\$_N7$_V.
MV_JD_P!T_P`9/^B5XQ_[#L!_T3I^_O._[[S']MN?ZU/\/^"?W+B?[';?U2?[
MI_C)_P!$KQC_`-AV`_Z)T_?WG?\`?>8_MMS_`%J?X?\`!/[EQ/\`8[;^J3_=
M/\9/^B5XQ_[#L!_T3I^_O._[[S']MN?ZU/\`#_@G]RXG^QVW]4G^Z?XR?]$K
MQC_V'8#_`*)T_?WG?]]YC^VW/]:G^'_!/[EQ/]CMOZI/]T_QD_Z)7C'_`+#L
M!_T3I^_O._[[S']MN?ZU/\/^"?W+B?[';?U2?[I_C)_T2O&/_8=@/^B=/W]Y
MW_?>8_MMS_6I_A_P3^Y<3_8[;^J3_=/\9/\`HE>,?^P[`?\`1.G[^\[_`+[S
M']MN?ZU/\/\`@G]RXG^QVW]4M:GE?D^(>/G,,7`\.X8HN.<&7BE!D#]K@N)5
MV(X!$N;21:-REO1J*.Q$:MGV8+8D/K1N4AON5L!J%YS]AW':VM!0TZE9@[>WW!..V)XM>,_95@^5TC7P6[!"7
MO#01M8T-W^Z-QI6E/0%(_'7D&]!HT6#[OXQ#V84825!R0S'>W+3K:SN'&E).
MX5L`1J*X?E-Y@HW1975C9"#'3WFF@TW=2I-F>WUKE9_-Q9D,)95DKF[`YM3K
MMZ:^E9HX?EM3EEGHE(`&VO3?J7-#6U
M'@`!]RHI97SO\R8[G^DJDNH(/797W2`/YM8T-52G65$$]-_3;;T!^'
MYQIN+M"O5GOG:[HK/GI"$7+>P(+L<[CT`[1_PZ47N(VM-1U5!93OW)]!N"-_
MS;_X1H>BXE^`JES$C=0[DE6^^P'7KM_1OKJJ16I.;_=N*)`*=^A]?M_P'0=5
MV9\8]JH"DJ)"MTA(V!)^W??[==E6JGRVN\+)*3TW&WJ=_L^/IH>B:>/10U&X
MPML[RJ1[3)8JHRT?,3G4%+*1L%EA*]BI;Q;6"$@;GTW&H?R#*QV+-L;VB;UT
M*OF&L#=R;G,<8#[?QJ>J_BW$\<:2$1H7SL=ON7*FJ:#C7:$[K1[;3KS+?7<#
MN&Q/KK$U]F[J]Z.=]FGXED:RQ5C9Z!FOK-?QKHO4U5:J>C_CL,NL*"2(=@X^
MD%Q!*4G=U[M`Z]-D*Z=!JS>1<^+WGVDJ012PQ?`UGW`JGP\(IZXOKEY'6S'5
M=ZVF9#'NE8"Q^['NO,OE04@]I;4-MO0ZKHFP,TE:VOL5/OH:@-^Y8[\JY?:4
M,.9$@_*J]E#B4RHJG&Y2&.X@%R%+2VY)"TJ!"$A6X]#\-45Q]VXEVIKU*U'\N_(@O[]W>DQ),1QE^OE*2D)4IL
MH22CJTKUUX0YO(6!\R.8[?`>@>A7E^/MKZ-L+XFZ"E2.OK]:I'`WU-<\X&7,
M8NG\@YKXSA!+'\/S;1#V7X,[\PKV),6;(2HRZY?WVUE?W>WM&R"-U99X/W3N
M\=^KR$9D@=T\"0L=;(#)`_X:=1[2LR+EG=I`&VQ!]`3^?K^0^GQU+6!S6[7=0HVYA::
M.ZJ,+&*/OGIO^;_#JH9\*\CH58U@P1W#IMVG?UH#N&VRNGWD^I[@.A`&B+YL:Q%W`M:EWN+%K63*Q];
M>P*&IL9V*XX@$@%3:720-QOMKTA>RWN8KE^K1I]BJ[>9^.O[3*@U$;JT]%'5
MU7G;Y*^F[RWQUG#MOB4>H?IHTU5C66Z)B?:F-0EH>AF-"`=EQK2&H)[F5I4V
MXC?JHCK'>X+,+<,_T*&EL6@]27;B/>UZ]>@\/!;)'O7Q['0_,OD>UEVUK)(]
M33:`TD>BO6H5N76(9Q%>6O*L!:8,.Q<#C['
M:6EM/J=2GMZH&%X<)C+IQ^1O/ELDS4L=K4>&I]*XN<%Q#E<+CA+J-MA/'JR0
M[SO/Q.J[4.!IL/YG@KYXZYSP?!HTO',DQ)4BM4ZN1^%.06X*XMLI3JDR$6E6
M]&L*&QCROO)LF`IET[B0T%;DSWB7+^1X%KK9^R6U+]K:M:XF2@J:FM6D4TZ+
M'W(^Q_$.3V$.`Y?:QR6\30W]J15^9C94D";:09=I)_6.U((;6C0H7S^5R+E2
MYTB;R1E<.3-+#]!%1D#QQ]:4O*^09++9;@L6#;'W6Y>ZV9!W5WI6HI&P^#Q4
M,[[?+9GR&Y-[*_`*@$DT`Z-U59+].V`XEQX8WCS)?V3+[T=_O\V.?3;K&\G9
M/IHTT92AZDJ:_%^XN9%PJGYJY,R7"X>.QVY%9FDE+9L'5-(=C+JH;,2)(3(D
M)40M+S@V]HJ"U'H-9$S&1Q5ABG1^6;VZDU;[Q#64TH6CK7JL17?TV\_N,E'>
MX**_9:1M(>2;=CI"34.+#[K=.FS2G759RH3F(41;D
M62EM9[C)":I*D.+6LJ24'H#T&VHC;6`5_F>6?OKZE`V?\C^#]'5QWL=S;D&1+BJE][U[6M6\9\A`^3[8
M\RWK^T&4"$^RAQ2TG8H).J_'=RLY"#!-;L9&WX-K-M/3TZZ4T/17Z3Z3>69@
M0QS7F-CMW2-W;WNE+FU&X-V-.TT\7:'2BUPY'S'C>/03EV0M6,U^7)=&/8\Q
M6O)O,KM@_P"]&"&_;91%@L+`4W':^XPW]YPDI`U%>0!\.JW.&%[6=FN/VV$QF*CN[P0M:\QQ`OFF<*-#7D%Y&[5U=&M
MU6,.;2',&85N46N/-YKR6ZXU7XHVN`Q8)QEDA,6%%2E?9#E7:(S8"W5`-
ML)3[JP%("QH[R6;CV4RSK9DTCYY)JN+G%S_$:O.I:*T`K0*,VG!.(=O]W.^Z
M$5O:YF4E]I8!U+>V:_WP60@[)-]*R.(KNH"H;Y4X,Y8X_H:CD;E7(&K^WR^X
M=HWU5\\6-)6RXD)RQ_"ZJ:VVF)-$,)<#SL=;C/O%0[U*WV^D_P#NY<7B<5SC
MD<%@X/N#@X"YWJ^;;I]Z^2'^\][J\=[A<*XY98>*%MU;<@N)'.C8�-S`*
M#2E=:*"=?6E?&U?A.VWYP#^8G1=@*E5Q$0NP%+0$DLGW2"2.Y/0$`C[!UUS1
M4Y?1]#TU5T_)&;C,[8;KBI3-:&W17RY[71N.H[FE$[_';14@E8)PK"6UV>VK
M?N2[W=FW_=E`.^W7KI171CHG1[C\2OO%*\B=MVE0#B4]WP((0KI^**
MT,K:2W/3'0DA#+04KJ?UGE%8_E2D`:]("7UW*NMGN(5`C17I!'_<[Z\B]P-`J*6:-C75_I*Z
M*C9=)[W"D;;(!2$C]1#820$)2-@$IVV`^`UZMJ:>M>EJ-S`]^KB-5NP@XQDU
M1Q1P%E<5F,1
M>Y/.8-\[8;5MU-+:K^<6FE;)KPZ'X27.;KU%`K"KHSC5=R%CKZ5"3#CLVJ4JV"T/XY;)CR0.G
M5?X=,=Z#]G\FI-/*SL
MG`H>M(RP._YC7:_:J7/]V7A%#/"]WJ>VMZ9PM#M6E$IN/;11L/O=H6[(V)^[
M]W8:JK`!O)+VRG_HKIH?3I4-K'U\?=#05;+MTMQQV&_:?_F%B7L8?!CB6[*-
MZ?'YAZ:_8I:J.5,GI,OH6UV`GXSD\.B?G4EPTBSIU)NX359:$0IX=1'491?"
MEL%I922.[;4&NN$8&YX[>2/:(LQ#+*&NC)8X;7$LKLINJ*'WJ@GP62;;G.=Q
M_)[&&V?)<866%A0\TX[-879>W)61B,;30-?
MLD`=L+OA<1M%"0#4]5ZV\G`>0S9''6<3\?D_F/-,0870[VUI)1E'MW5.[7;I
MT5M91QQE$+'A7$RVMH#,ANWF+OFWB16HEF+6N5/98;1/K'-IZQ[<8VXN\EBLVU]G<71#A'*-I-8_+<6DZ$58SX21U]*MN5X3G+#'V6
M1QKX+@1[HB8GMD&SS`]K335KJ.EJ'>\`17H%U\>B)AI&\5&IJ%$SQ,G"(ZAU`:DES'>\:^'NO_
M`(_!0PW,LF,\V,;,M87LT(D:*.$+PTL:/^4!4^)!5QK?4O.VWF5N-M9MCZ/:
M=2L(!D7U&2G;M`[2S?(W!'WNY`&_KJSOMHKCBIMKEOF/LKHAS7"HVME=I0^'
ME4]JO]N^.VY`)[6K+">"IH2-[MC03([JXB3?2ITJ0%4*CE>^_@-<7*(5?G5?
MC^11HRX65L&QDQ(%O$4([=?;I=9O*KV9E8>TL2$I2M?5)&J3(\%Q$_*&2XF1
M]@ZYMOU9B-&.ECH7;HZ%CM'#JTDZJZ8_DN0CP3K.^+9+*"X+I&%HKL=HW:?B
M:=#4@ZZ*ZW:KBS-LGDFKMIV&76>XLM:*J]8-ICTJ9ZB^
MDN.X"4E2G-6&'(`/34MQG)./Z_W]:NV%
MU/T3[PU"V=\.\X\1-P[SA[D1J$A%C:2,JJ%W$%B72HQ?)JD9%*;W6AYR+)'S
M2PEUMM;B3N0H;'6K'-.!\S=):'6UI=<.Y1:PF'YD2QF1@(%O*T;@":D.)`(D-K4VI7[M2^X(<3N@=IU9N,][^8\
M3-S@^76DLSY&EVYS0'[V&H-2`*>NG7VJ]\C[0\4SAL,_P&[9'AC-/B&_@W-:^->8\1PGDS#+#&;K,ZB@S&EKJV@K#10Y[>[/>KL([CJ4=Y>[N-Y9VZ_;?$3-89B.]AB?)"]\,C
M6DN<&[XRUVUPW5UHZ@J#177Z?.W^2PO>-W%.;6T-W9-P\TK?,8R2&1[7M:7!
MCPYE6U'A5M3Z2MCZ?$OQB)Z^)?C&.GQX/P$?X*=_WWF/[;<_
MUJ?X?\%_N7$?V.V_JE^_[I/C%\?$OQC_`)>#\!`_HISI^_O._P"_,Q_;;G^M
M3_#_`(+_`'+B?[';?U2_1XE>+^_WO$OQDV^T<(8%_)U-+MKC]_>=_P!^9G^V
MW/\`6KC_``^X+_=_WYF?[;
M<_UJ?X?<%_N7$_V*V_JE^_[I'B[_`-$SQEV_\R&!;_\`L%V_IT_?WG?]^9G^
MVW/]:G^'W!?[EQ/]BMOZI!XD^+A(_P#U3?&4=?\`F1P#_0I_PZ?O[SO^_,Q_
M;;G^M7/^'_!*?_1<37_T=M_5+D_W1_%O_HF^,W^Q'`/]#:?O[SO^_,S_`&VY
M_K5Q_A]P7^Y<3_8K;^J3_='\6_\`HF^,W^Q'`/\`0VG[^\[_`+\S/]MN?ZU/
M\/N"_P!RXG^Q6W]4G^Z-XMGT\3/&8_\`ND,`_P!"Z?O[SO\`OS,_VVY_K4_P
M^X+_`'+B?[%;?U2Y?]T/Q9_Z)OC/_L1X_P#]"Z?O[SO^_,S_`&VY_K4_P^X+
M_+(]?$WQG(_P"UX1P#^G_R'I^_O._[[S']MN?ZU/\`#[@O
M]S8G^Q6W]4OU/B'XL*)W\3?&8_MMU_6I_A]P3^Y<33_T
M=M_5+D'A_P"*I`)\3O&@'[/]2>`_Z"UQ^_O._P"^\Q_;;K^M3_#[@?\`8_MMU_6I_A]P/^Y<3_8[;^J7V
M/#WQ2(Z^*'C.#_YD\!_T#I^_O._[[S']MNOZU/\`#[@?]RXG^QVW]4LL_P`/
M1]G]"/T:BBEZ?AZ/L_H1^C1$_#T?9_0C]&B)^'H^S^A'Z-$3\/1]G]"/T:(G
MX>C[/Z$?HT1/P]'V?T(_1HB?AZ/L_H1^C1$_#T?9_0C]&B)^'H^S^A'Z-$3\
M/1]G]"/T:(GX>C[/Z$?HT1/P]'V?T(_1HB?AZ/L_H1^C1$_#T?9_0C]&B)^'
MH^S^A'Z-$3\/1]G]"/T:(GX>C[/Z$?HT1:._J0Y!?.GRV(9*UD<0'F1FA)%:
M@G6H%>GK62>+6N/N<9&R\FC%;A_ZMU#31OO`=03_`!*&;6#%8K:;*\*=IV*M
MZ'%AO,Q4AZ%(BO.-M+]GVAV^TPL_`;#;6)\E#?-N1/*VES2C@_6IJ?>/37\B
MRY@LK97,$V$R/>[]7(*D>`UIIZ`L<\ZMK#(8V:.W
M9_0G:"?B(.NIIJLD);#/S;ZERI0[%GM;'8>XE75M'3UV.^_PUG!H:29&U(
M^6[?>[3[?N$A'?L>WO(ZA.^O,#8\TI0)Z)2>W;O*#\=<1O@+=DCJ3U^Q>#9P]N^**21HZTKH/2NC,M.06?O
M#&ZM]*4C9*9ZDN+/;NM(W3L"D].NO4L8';-=W5<[W.;YC11E:4/5<&-W>86$
M]QN_Q9FF@%E2FY29R)"RYW']VIH#IND`@_ET:KE;[*=NY)`(!_6`/V;^NAZ+SE^`JE3$@]`GT)]/
M7[==0J9H!<`5:\SN[7!LG8[D@IZ]/Y?L&NU%4B)H-16JH:D!2D`CIOUVZ=01
ML/Y==)'^7&Z2E=H)^Y>E"[W6_$>B^F:Y4IP+46TL,H6])><`]B.R$J)+P]25
MI![1\3JQYW-PX3'?.S.!<]IVCQ!(TK]JK<+CKO.WOR5H*.8X;R>A%=:?96BK
M5,S?Y"@L4`-51(=]I$U3@CRK5Q._O+2ZI+90RX4DE7>D)1\=8$EN+W.W9N+A
MU&>K3\JS3%;V.%MA;PMW2>O57N,/CLM!MAQ^0IUO=^2R8S,=SHH*4[+E-K>D
M;.I([@@]WP/35VBACB^$#[53R.,GQ?@4-YY'QJG:4F1)E29+B5MKBQ+?*9*>
M_P!LI4EMNKEI6G8(/]0(W]=6N\OYH/@#/N_X57VEO'-\=5BSD-]CT=LEFBS)
M(D1KJ=&CEM*&FW61.HWI;2"2G?O>!V&X4/74=ES,LFCH'_9HKX,-:$#
M_2&A8]9E?8W91'XT?)+%I#96J1'MVI4IA"F%J[&'8+_O2&T'K]]M03\=P>H\
M774<@]X.'J79MI-%HP@M\#3JM4/E-`L*B%-OL(L4S6%):C6M8ZZP[7.R''=_
MD#,][VB^\!M&;EO-+"SVM2%$=@\(6-+R=2"?%71I)C:UP%0!6BU48]D]N>7W&ULN>G>=MNT'4XQ7)),;$W(N/3<).V_\`+MO]OY-=][EYD"JL2UC_`+M?IZ'IMZ=/Z=>C#7KZ5U/1;<*9
MO_,*W]3_`,0A?#_]E9ZG\FM=)?Z9_P#/=^,K,D?]&W^:/Q*N`^B0`>T[CM^W
MTZ_DUYKNN_#B][A6O]7X!7HHGJK[O78`]!HBNJ*P`%*^Z`A(WVZ#;X?R'1%4
MV4?,+/;]Q"=NY6^Y7Z;@)^PC1%683`2.Y&Q!/:"H?`'H`.FNP<6^@CUHT[37
MJ/0=0ONVI(EU">K9[8.W(<(2HU-HH-_PM/6=
MU+3_`.*+43NE/ZQR;PWN1"V<0Y"5LKIY0ZK@:1B@&P5.G2OVK*YY+:86Q^?N
M&Q?([Z303%PB<*"IC<'4C!]`:377=K1<6-0*[!Y#C.8X389U&4Q
MGM-')7VR:FYCNL4&:U4E2>QMN/,#S:/O)4L[H$YO.XMI?YHX["S0QB&C2UPW
M%Y.M0:C0=%AKN%W^;AGQQ<#:^V@=J+62%UR)&@T<^"0N!;$#HXFM'467%!2>
M-^:29E;Q?XNY%GF;T\9Y[(./'-?L3@UO):8YDA8U75,@*2])QK#\6"Y:\,QJ
M(S("/FW7T^^VL.#;?MUKO<+;C;6[Y7@GX2:4!J*R>F,#0:U6R?
M%N>77!.-&YS+[SD7
M=8'B,68AN+(;D6K;$-N$A2%?@T>8\CY_(7I*5)#T6%N92]O?4ILA(ON+Q>'=
M/I:&&A!KJ':]/6M>N5R=X^[.5=G^2X[(.MV5;$(FN-O&R
MOP,+RWKU+=3HK>^KW`QBM\>N`(^%MTC.-*Y3OA'7&LH%G>VLF/A+C+EE8?A4
MA^#4PT-J"&H7G77#C05"M5U*YH)'I5X8Y7A
MAYZN>3[C?[Z([W>BVG$EM*MO0=S:^X#7@^5[6U%%;3(6G>3JHXL*%Z&PPTH?
M?@W#T%T;'=2"5=A`^`_=[_RZ]6'FYV_/JW
M6\CC<.!I2JCME,#=:'6J@;\)3\$]?AT/KJXJ1&0^E5]JG6D?J`$)2!NG\F_Q
M(^.NF[P73S-5#4M!'V-O>6`XL;GV&6]UOND==_;:2?S[[?'50T;K4VUVMI`^*B/4:I][O4J&69K15IU5?GP
MA0U2V%=IF/E4F:H;=7U)!#85T*D,H"4@';J"=NNNIZJB)^9>'O/3I18_W#XD
M27^OHG8]?B=]R!^37NTZ!2*&,"$`=2W^);X."N1)@X[X_J*"S1@^3?P1BL&+
M!<=1+X^Y`7'I8;+4*[J;!+]?!OI:4[>ZMM2'7E>NYZ:Z9&5;R1NNQM=!0F@I71?4O@?(>TGU+=OL;PO'SMXKWNP^,M[.V
MCG+#99H6L+8B'1.'EQ22;*^9\;RZM:E2..1L'RNU@RLWQ27@^4T;\."+7#TM
MHA09-,\EIIFPQ::OV4*C.-`=T62P2!T21MJS3<4Y)@[&>TX]=MO^-743Y2Z<
METCS**N+96C3=U&YI'A5:61&F5HJ[GY=]DLNH>JYIK9H6AQ*5H<2S;=Q!V_5V.LP375F[+XR]MY6OB
MF8YNX$4(A#=HXE/PV3JL@BCCY)?6CVM(N/
M*?J-"SRRUU!Z=S14^M45[)(_`8VZB<]C[.,MK7_M!)T=Z1M/317"HLGDC[JN
MV)E\4*2-QVJ1F5(E6ZD[?]ZGS^H'H4_DU:K=A'$-X+OF[&=U:]`Z*3H1Z-H%
M!7Q5YE=Y7(BP.=^S[R%AI6E0YO4.;M=4FO5Q&G14C&[Z\I<>R151:SZNWH;:
MIO([\&0^P^TVXIVGL0'&5I4A/?,CE0_5.P!&O?-8S'92_P`?)D88GPW<+HGF
M@]+7BE:_HD#U$^ENGQL$AK;R?K&NC>T/JT'WFD.)#2'>%-5/\-SF.27"RYBS9
M):,8RMRQPCG<5PWG%.-R+3DK&<*RZ!+L.]]Z+B]^TG'K3
MYBHLFY[:ZZ8^Z]13XBJU;W:L2&G"C;]WUVUQC.<9@6V,R>Y2*UNXC:RLC=2HK5T980#_P`@5\:G50S(8C,87&V#KFUE='&Z
M0/(J#0OW`T(#A\3J$BA`%*T*H,J$(]_R+C[9_=SJNPL8;20`'$0)+.10EH2#
ML0NK2L)^T*Z>NJRVN&_LW$Y)[:-@N',+SK\;?+=[!OVZ^I>=Q"QMS=&T<'-G
MBCI&1JW:237TUJK>1IX%H_$O)NQYL[VI%L)'1R-'PZ-K2GA
MKZU=--D^3X3:X-/YPC+J&NE>BE:QY`P?)H_$V6YWB:&K6YKI>+V.3XD\Q2JC
M2JB:]4NB70EA52_#%/9QU$,B*O;?8[;:@EKQ/D&#N\W@\!?D8RRN&W,45R/-
MW"9@>1O&UU?,#@SX@`*==5-KO,8CD$5CDLQ!_P#,7VD5O*8O<;6-SPT[===A
M;4UU.J_,:K\XQ>IN&^%.41D4_',FB6#6.(E2*'(VTMF56S&)-+92!%G+3)2S
MV_+/R$N=Y2!UVU[Y.^P.9NK-_.\4;L?H[5^XZ>YUI37HM@OBASWR)RCY4X%B'(=`S6S*G
MB3-;9$UZ"_"M9;EBC'$RX[S9:D/(5
M(DA(?;W8```5V]-]9#XOW"P_&,6[!YH/%I=/=[S*;AN`&A((\/05+>/=O.2<
M@C/)<"YA?;$C9)4QG;0FH!!\?2LCN.T23QG7QK>E8QYZ*EADU[#2FJ]IEQH/
M(^16?NNM;G?NV'H=8%YA!9"[FN,>^=UL9/<\P@NH===K6C\"R9Q^XN9,E$R:
M*!MZX;9!&';:CK0%Q-?:5,>./6CEA@\U$AUS_/8HV[@!LU]IIXK/>=6!J)^(!3KJF@APH[D
M%2NFW]8`[[=>FLYQN,4;(A\(:/:M7IAYTCYG:$N/3IH53*R=8S9S+.]A[*%`
MN>ZX@-^WN3U^\>Y*1\!\-53V`@$JC:\BJYIN5/Q)+D4U#SFSBDH<0%$+0%;>
MYZ#M`'\^O"0>4WS&^!_&N[GO>QS6TH0L#_-S,[ON"IYA"
MCVJWV))ZZR9BI77=C!?;@0^+6GIT6O'*K#]G\CN["5CHY&2F@&C=M?9U582U
MT((2=B$@`=3]G3^35PD^%6-@$9W-ZJSK9`2[;I[=MT1=SML1W(1O^0:\:JH9
M(YSJ&BME">KB4_:-M_RIWURO9PW"A5-E#8$$#?<]=NOQ(_PZ47F(FM-156C.
MZI(]-N[<_D/Y?CKE>JI+J>U(V"2G??<[=Q^T#J`?37+7MC(>\5:W4CT@+BCC
MHSX_#VKN.L3;=#-*@"NJF&Q)N+`%OOE`/$.H0`3[G>@=I4OHC^J#K7?EEW)D
M\])%N/R[34,\/L6:>/6<6/Q#+H-VW+Z`D>M21&M(\.#'"6F68L2.3"BN+0RT
MU$2!_G-DL`[1R4!1ZE3RQMZ`C5!#(Q\0B'N@>CJJ\6[O-,C@7./IZ+B5:W^1
MH<O+,QZ^O2$_>6U!BR&7G"P$GN!]D*4D=%;ZN#1AL)
M(0M)ER:QB?7-(_?;%9/=W?``[:MGRESX/-/L_(JZ.6,FDK`/O6.N<':A3\=1^\-P4ZIS;R5H6@GV?
M\*KH;V!QVU<`%A1R;@%';&794$MZD?L''(NL8E(<"RY195$/;8U\1Y3
M_P!Q3XG0E=R2AU!`U2^6]CC3K57-L[*"ATHM?D_QQE_C%@_.K7\J%+`V%="]Q:8W=#_#14LXCJ7U-
M3KZE#_D!5NQ\XXV2U+5*K8U2N-=P7F_>%5/>2TRW[+K)4Y&ZM>VH?J*#86.C
MFVKS'$UN)?:O`,+A6IZ_D_`K.)B(@A\32:>DC7\*TLN+>>UNIK>>N]DK@*]=
MM=/P*B6#.Y)/;OV_F^WX'[=585.[JK(M&`$K_4_45\?3H>GKKNUQ&BX/1;5:
MI17!KT-]`(,(%0&Q),5G[H!_-K7B7^F?_/=^,K,I'PVT158
M('NGUZ`'\^_37!%>A(/J1=*[@,/,JD.):)::+9]W;L2A1W*NOZJT*'0_E(]-
M6/)V0>[YAHW2T`U51;EKR8IJ>33[5BIGU,F5#?E,NHKWV'DK@Y/VN-%(84E+
M8#`4EN48[B0#WE*0/C\-6"/*#'Q.9D6^9&2?U9%8Z'QIUK]O@K%D<;\Q/\QB
M`67#--X^*H\--*+"[D.^XUJE3(_-^;!2PV5RU^XLZ.<:?F_R::>VJFW#N77V/
MO2_E$39[41^5YCP7!C>OPN)9NUK4M)^Q:U^9.;(F(*B,6/S$:A"RJJK9LV%*
MCAE2C[;:,Y^7?MH`<24@-1&>BC]V0G;N&,+`7MEE7U\N&\@:1Y\N[RR[PK0@
MFOM5PSW).VMJS9QY[;W*2NH(IHA=.J?""W:&ACO1*VFTK`/F?R^Y"R9NEJ&F
MY.!U%3.**:1CC+N/9HPVLAV2*KDY],[)Y:'W5;I9M'YD5P="^A)Z7ZXYKRC,
M63<5-(9;RI!?J+(`=-PKOU\/UGI4,=B;G%W9ON67<]I8;-S+.29LN1E:=71Q
MND#VQ1G0%LC7O:"!&YGO5BEV\O\`%:^/>\A)_B3$K24ZY(M+!R5?CN?
M^4,@P2.ZXS<1'V=N^XJVG@D^J'=BH4^-[;6V>O\`YNX<;[.-VGRVN_T.,@FA
M93]:*]'?K:$`55@Y)]6+,)B&\1XN&\;LMY#;=L89+,YOP&6=M-SG$DUZDG5;
M6/#/"XUUQG>[SGDN$)-&EA8&QNC)Z->UYKK5;$:B-R;!@UD+"\(_%LJEL.L
M4QS:H;@RV&&PY'5(B.UJTR(<2--O
M;4/I0M4R:?>*$=J1V]3],O\`=E8[N%CN\_+V<]8#.>,VVR2(.%N3\\VK6AQ<
M=X&I][X:Z+3'_>.YN'*]J>*0VE]>GYH;[O52_0,;[=$;]Z>GV=/3UUY.>2U6.Y?J=W126U7I:E0YG:`'0&'M
MOU5.-'N:"MOVVE';X_=UXN)<*>"LTTX/N?FE,CQI+QM7$)W,R+%MFSL-P_&6
M"^``G<']VK\Y7^37=LCFB@Z)%4CJ@O=X*ENYFD'=Z%/-]4>[@%AN%#LD5R]MOLEI3UW3T'W_Y]6?SQ%IV'0;
M^H^/0[:X$[J]`@G'16A,QL`J(;&Y))Z>IW))]/B3JL+BJED_E]"%77J3\!Q(
M!;:6Y%DM<]U/7HTM!;C)*CL=_:1OM\-]4S+AUQI(`/8O%]UYUY5OW>"A_&,>
M=D2;.V4@>]8O_A=;W?\`@]_\ZD=1T0I8[3UZ)0K[=5HF9)VQ1>6
M.BENNHV8Y$EQ`$.I2&HG=K
MFT8:E1+G%B/W_P!X$;+._P#(0/C\=5C27-#O&@5SQ\+90W=7H/PK']XE:EK4
M"%*!W'IT_EZZJ6]`I&UH:T-\`MNV`MHNHG=1#S)-KB'%YW4I5PU]TU!TIIX&GBO&SR5]BLQ;Y&T>YEQ#*'@@D
M%Q::L!(H0&Z4V%OKJI.M)"K2OB9$X=[*(Y$HCR
M%XN[0NOQW%-.)G8M:S(:-W6REP.BME1SON/N[;?#41XQ9V@%P=]JUBY1?7F1L^*YW?()F-EA>XTJT0@EA`(HUQ!%::>JJXJ
MKG3(4L.P,PK*O/($B$]6/KOF%"\1`DI*'F(V2Q"S<))1MV^ZZ\A*@"$]-5&0
M[78J:?YS!7,UG=1&K6,+3"'#Q=&01J>M-I*\K'NODH9A^VK:.ZBD%'RN:XR.
M;T'O;OS10"HH``JNU%XGRC'+"GQ_)9.&VTFT@VT6JS((?J&Y+,:7#D,Q\HK6
M"I+87+-R69LVW%I#"8C+!XL6[?+AGC:1[0]@H[Q*HN+U^
M!Y#,@%Z/5Q37"XJW8]G3NBGMY$^L6W;5JY5?_G$6<.T^YWD-[$=-A<>/:&O!C>'O:&R-V/H=K2T4T\3JJ/D7$N1XBWQ5[)8S&2.D!CC<
MV8.A828[CSXSL`EW$AI:'-#?>"MU4-#.>931I3V10``_>&@MZT/K5G[F9@CB`"'Z/)4N^YU4I,/(*XLA':3VI0F
M95(^&_W]Z'W):YL$^/#:D=2R0[R/"H:\4/J-526V/CEP<=J2'W%M
MD@6N!-0'M/D@ZTH71NW:>.E%(>33$KSS!U/
ML-RY%58LH@6#;J(J"]-CJ[V%-.!>VQ^(J\UP[C^5RV/N8F_*LN(9`9H3L>7`
M-DJ2.HV!_4'4BOH7OB.69C&XFZ$E;J6&9A$+Q6,-<2RNP4H2\LJ014`^HBZV
MKSB3)+O![^VK[7!K.ZK&X+LJB2F\QMQ<
M*SOQ&Q87%5*J+-$ZC+;=Y"=3'EH*UNQDI`;&Q/KJ1VO
M2%[YX]D&YP:\;9*[7:@@`..A]*M-YPC+08F\ML,QE];1N\]I9,PR,+CM)E;0
M.;'3IM:YV[3HK!6I^/EF'S9S"XC>48ZQ16JG4+2HO+B2L-L4J*D[MN(#"%]H
M^\#MZ'4DM9X'X.[QUA,RX%I=&:-S*$>6UT8S;MH:[9'>G0"GBI7QO'1Y3997;@(7YJWC<8]'1QR4C16'&\U>24N68W=/.;;Q'8=A',:5$W<6L,NN/6#;2D^VKU<
M4-]T[ZPOP[ZF>.YG`06/*&-CRMC<,`+FU;LZ.%#K\)(%2>@*RGRSL5RWC.=G
M.$MG9+$36TDT4\9.FQU`QQ!`)(]%*E7]].L\NT_.^.8OGS-F(E+BO*%#+3<1
M&Y:ZV6FW0VL-#<'8:M'U)'A%_PVYRO&#"7R3V
M4C=AI42>:'^ZTAIZ`U+:@GJLB_2QD,Z.X]K;Y/S([:?'W@$;@*MDA,6X$D5T
M!T%5OJUH,OI,FB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)HB:
M(FB)HB:(FB)HB:(FB)HB:(FB)HB:(FB)HB\W'U7H?X1Y+MYBPPAUR-QEA,24
MA:>Y+L<6^5@%0V]6R_T._36'.97MTWE<5NUY^79"QP;X5+GZ_@6\G8+'6TG:
MNYNGM_6/O9VEWC0,B_*5V*Z6^YPAC,BBE;?MJ"O4$>I&N,?+O9;QNH`90HYS:Q!OKNZ:XU?&XD>&GH6P
M^R#:JIP--MK6AE':EU92A6[:>JE^J?76>&L:YPH3HUOXEJF^5P!:1^<[\:M:
MGDS&9K#2&X:6WB4.K2KW%!`!.P2=R.[\FVJ@FJI@*+AL;6=$FR&P9ZVD/=P(
M""UL!M[824[D*_+KSE`D9Y)Z/\?$4]"[LFCA.Y_6E`%CAY4,T>28%&1+=]NP
M2IQ*H"D$O)AO)+,AYY"?1AM)W4?AOK'7<:W;>8AT-/UK![I'6H"S[V!NKW%\
ME8YI`M9"*U-`:N&GL6)_C7R[R%QXVQ2W>3S-ICF_M=H;:@[&]>IU!U]0*RAWWXKQS*91SL&^O
M("/->T;=NP`AVO7XBU;%G6KB2E$V#=1%0I+:5QW/<;'>TX.Y"M^X]2A6LRF1
MDL0DB-8RM.Y8KBWG=;W30V5I\%TI@4E-FVITOJ4W$[G>GWB&T@[=I4-@=_CK
MR'5=HOC5(;!^]]WJ".NQW]!KLJM4B8D]R@!_65U/IZ?;HBM:8WT*"G?N)'UZQ<"@&*]
MA)`2W$;*@0GKOOUZZUIRUZZZRK[H48YW@.FOMU6P6*M1'B&VY&_:-">NBHEI
M>54)MJUR)]:X#"BXQ71NTBTF(2H=[K*"E+[@4>UELD-L(/+WP=G;_P#F7&7^=3^)1#F?&.5MP)4Q$FRM[4L.
MK+]K(E2W7&G5%:%*,QT1&6W#ZH`*B/1(UVN(,I2M%S#)B:TC]P>I:VN6L%YU
M;>F1H$/C&QIW4>Z_3Y-%KXCK?:HDML3U2Z^5%82K<]S?WT#?M)(&O&*._=_2
MZ!>MS^SW@!G58%9%QUG3,BS?H[5.&7;T2:EZEJ[VMH
MES&8Y:JWH22XDS;%A+[\=2BE83);94XI)V=W+*BE-"]K2\N]:J0YU!X%9X0J
MZJF5\7*(HAFG:9#L-45(>:9)C+<>;B$>XKVD/L%:$]4H1OW)V`([LM?SQ75>
M-S(X,^Q:Y/(_'Z[)N6*S\.:CPF&X1GV$FO4M*7HTQWV694-I"4EQ44]%IV#S
M0)00H)WU77D[H,<&`#I3\:I[*(SW.X^E9I>.S&546,Y'Q_`$!9O=5K/==NKN\>JFY^';C,_\Q#7R)]M:TH#36E%E;8,!7>0D
M?J]`>A.XW_X=9=!JT'Q(6.7-H\CP!5CV:[JJPF5O%#CZ=D#HVW_-NI7P.Q'31%<$=`22.OJ!]
MG3\@VT15%/[L.#8[$A(W_P"VV'_#HBJ\7T&_VIWVZ_#\VB+[F6+,"/*G/*0E
MJ.C?91/"IS-R%P397B$UJ75+2S7R.JG4-*3^]]@A3CBE*/0`'8?9KK?F&U/D1$
M/DI7[_8O'!LR^38V[N&".5^FP5V@>!UUJ5#V?PFN`*[78^/T;P5$D//$[)=D$,I/HE9U8KQE@Z(NR+6-@VUW>-?1JK_`'-W
MB^-Q/N\E*6!C=SOT0#TZ>\37P"TN>1MM37[M\QQU1Q,5J)5>X+'*NO
MJ5I;Q_E/)K7]X[TC'<#?JUSCMEN!U\QC'=&4]W45J#X+4)=<7\CUL5*<2&CB<6:52[*0>U003VL+.Q![=QJM%A'<-AAY#&)[*5S
M2V**ID+O3/UHT>.VFBQ_E.266!,D7`VMM\GY99\SM!E*WEB;)&33"I2&I&+30TW2U;G?^JRE*6D@;.*0-][
MCG>U?,,PV48!]M98QD8<8]VVWD;0D`2NJ_S`*@-\2?!0;'\RX_!9_(\UCN;N
M^\X-9.'.?<"60Z5>ZI,9(&[T#HJ39>+]/PY-=M.=4V6%9*IQI..S(EDN3/QV
MP;+3J8[[*EE;]I.=V*F&TF(^D[I?W[0=>).[5UQ'(G`8026.2M7TF;Z3M)TT6
M:GC/R7&PJYE9+@&0(Q#DVY>[+*W;I7'N->;&D,A+%7E.)5T27$XXS-Y37)VBSX('8JZR9P\V1B;3.&\UP,!X>K:TM9!Q_F%AD>138%[69+2-HL
M,=771846XJ@J)(FI6X7%AM;@2A.Q.^M[/HDAYC!R_.29[$76,Q;\3%Y7G.8X
MND^9%1[H!!#===*>M:>?[Q[MEDN(=DN&<^G5`I3JN`4
MC\9PE:.]Z+VH=(/^4C_KI=`/KL3U/KUUU