Exhibit 99.1

Gilead to Acquire Immunomedics S e p t e m b e r 13, 2020

The tender offer described in this document has not yet commenced . This communication is for informational purposes only and is neither an offer to purchase nor a solicitation of an offer to sell shares of Immunomedics, nor is it a substitute for any tender offer materials that Gilead, Maui Merger Sub, Inc . or Immunomedics will file with the SEC . A solicitation and an offer to buy shares of Immunomedics will be made only pursuant to an offer to purchase and related materials that Gilead intends to file with the SEC . At the time the tender offer is commenced, Gilead will file a Tender Offer Statement on Schedule TO with the SEC, and Immunomedics will file a Solicitation/Recommendation Statement on Schedule 14 D - 9 with the SEC with respect to the tender offer . IMMUNOMEDICS’ STOCKHOLDERS AND OTHER INVESTORS ARE URGED TO READ THE TENDER OFFER MATERIALS (INCLUDING AN OFFER TO PURCHASE, A RELATED LETTER OF TRANSMITTAL AND CERTAIN OTHER TENDER OFFER DOCUMENTS) AND THE SOLICITATION/RECOMMENDATION STATEMENT BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION WHICH SHOULD BE READ CAREFULLY BEFORE ANY DECISION IS MADE WITH RESPECT TO THE TENDER OFFER . The Offer to Purchase, the related Letter of Transmittal and certain other tender offer documents, as well as the Solicitation/Recommendation Statement, will be sent to all stockholders of Immunomedics at no expense to them . The Tender Offer Statement and the Solicitation/Recommendation Statement will be made available for free at the SEC’s web site at www . sec . gov . Additional copies may be obtained for free by contacting Gilead or Immunomedics . Free copies of these materials and certain other offering documents will be made available by Gilead by mail to Gilead Sciences, Inc . , 333 Lakeside Drive, Foster City, CA 94404 , attention : Investor Relations, by phone at 1 - 800 - GILEAD - 5 or 1 - 650 - 574 - 3000 , or by directing requests for such materials to the information agent for the offer, which will be named in the Tender Offer Statement . Copies of the documents filed with the SEC by Immunomedics will be available free of charge under the “Investors” section of Immunomedics’ internet website at Immunomedics . com . In addition to the Offer to Purchase, the related Letter of Transmittal and certain other tender offer documents, as well as the Solicitation/Recommendation Statement, Gilead and Immunomedics file annual, quarterly and current reports, proxy statements and other information with the SEC . Gilead’s and Immunomedics’ filings with the SEC are also available for free to the public from commercial document - retrieval services and at the website maintained by the SEC at www . sec . gov . Forward - Looking Statements This communication contains forward - looking statements, within the meaning of the Private Securities Litigation Reform Act of 1995 , related to Gilead, Immunomedics and the acquisition of Immunomedics by Gilead that are subject to risks, uncertainties and other factors . All statements other than statements of historical fact are statements that could be deemed forward - looking statements, including all statements regarding the intent, belief or current expectation of the companies’ and members of their senior management team . Forward - looking statements include, without limitation, statements regarding the business combination and related matters, prospective performance and opportunities, post - closing operations and the outlook for the companies’ businesses, including, without limitation, the ability of Gilead to advance Immunomedics’ product pipeline and successfully commercialize Trodelvy; expectations for achieving full U . S . Food and Drug Administration approval based on Immunomedics’ confirmatory data for Trodelvy and Immunomedics’ development of Trodelvy for additional indications; clinical trials (including the anticipated timing of clinical data; the funding therefor, anticipated patient enrollment, trial outcomes, timing or associated costs); the possibility of unfavorable results from clinical trials; regulatory applications and related timelines, including the filing and approval timelines for Biologics License Applications and supplements; filings and approvals relating to the transaction; the expected timing of the completion of the transaction; the ability to complete the transaction considering the various closing conditions; difficulties or unanticipated expenses in connection with integrating the companies; and any assumptions underlying any of the foregoing . Investors are cautioned that any such forward - looking statements are not guarantees of future performance and involve risks and uncertainties and are cautioned not to place undue reliance on these forward - looking statements . Actual results may differ materially from those currently anticipated due to a number of risks and uncertainties . Risks and uncertainties that could cause the actual results to differ from expectations contemplated by forward - looking statements include : uncertainties as to the timing of the tender offer and merger; uncertainties as to how many of Immunomedics’ stockholders will tender their stock in the offer; the possibility that competing offers will be made; the possibility that various closing conditions for the transaction may not be satisfied or waived, including that a governmental entity may prohibit, delay or refuse to grant approval for the consummation of the transaction; the effects of the transaction on relationships with employees, other business partners or governmental entities; the difficulty of predicting the timing or outcome of regulatory approvals or actions, if any; Immunomedics’ ability to meet post - approval compliance obligations (on topics including but not limited to product quality, product distribution and supply chain requirements, and promotional and marketing compliance); imposition of significant post - approval regulatory requirements on Immunomedics’ products, including a requirement for a post - approval confirmatory clinical study, or failure to maintain (if received) or obtain full regulatory approval for Immunomedics’ products due to a failure to satisfy post - approval regulatory requirements, such as the submission of sufficient data from a confirmatory clinical study; the impact of competitive products and pricing; other business effects, including the effects of industry, economic or political conditions outside of the companies’ control; transaction costs; actual or contingent liabilities; adverse impacts on business, operating results or financial condition in the future due to pandemics, epidemics or outbreaks, such as COVID - 19 ; and other risks and uncertainties detailed from time to time in the companies’ periodic reports filed with the U . S . Securities and Exchange Commission (the “SEC”), including current reports on Form 8 - K, quarterly reports on Form 10 - Q and annual reports on Form 10 - K, as well as the Schedule 14 D - 9 to be filed by Immunomedics and the Schedule TO and related tender offer documents to be filed by Gilead and Maui Merger Sub, Inc . , a wholly owned subsidiary of Gilead . All forward - looking statements are based on information currently available to Gilead and Immunomedics, and Gilead and Immunomedics assume no obligation and disclaim any intent to update any such forward - looking statements . 2 Additional I n f o r m atio n and Where to Find It

Gilead is Executing Upon our Corporate Strategy 3 Long - T e r m Ambitions S tr a t eg i c P r i o r i t i e s Bring 10+ Transformative Therapies to Patients by 2030 Be the Biotech Employer and Partner of Choice Deliver Shareholder Value in a Sustainable, Responsible Manner Strengthen Portfolio Strategy and Decision - Making Increase Patient Access and Benefit Continue to Evolve our Culture Expand Internal and External Innovation

Gilead Acquires Immunomedics to Further Transform Oncology Portfolio 4 Gilead to acquire Immunomedics for a total purchase price of ~$21 billion Adds Trodelvy TM , a first - in - class antib for 3L+ metastatic triple - negative br ody drug conjugate (ADC) approved east cancer (mTNBC) 1 Significant efficacy benefit vs. chemotherapy driving early adoption with potential to become standard of care in advanced TNBC Ongoing registrational studies in H clinical activity demonstrated across m R+/HER2 - mBC and urothelial cancer; ultiple other solid tumor types Five data presentations at ESMO 2020 this coming week, including phase 3 ASCENT and phase 2 TROPHY U - 01 studies 1 TRODELVY (sacituzumab govitecan - hziy) is indicated for the treatment of adult patients with metastatic triple - negative breast cancer (mTNBC) who have received at least 2 prior therapies for metastatic disease; this indication was approved under accelerated approval based on tumor response rate and duration of response; continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trials. Fully integrated oncology company with 430 full - time employees Trodelvy is a foundational, first - in - class commercial product with potential to be best - in - class and drive significant revenue growth Impressive talent and expertise of Immunomedics’ employees augment our existing capabilities in oncology

Strong Strategic Fit 5 x Approved transformational medicine with potential to provide benefit across a broad range of solid tumors x Accelerates entry into solid tumors with immediate presence in breast cancer x Emerging data supports expansion into multiple solid tumors , including 3L+ HR+/HER2 - metastatic breast cancer and 3L+ urothelial cancer x Complements pipeline with opportunities to explore combinations with internal and external immuno - oncology (IO) and other agents x Provides foundation to advance solid tumor portfolio x Diversifies marketed portfolio beyond antivirals and has potential to deliver significant revenue growth +
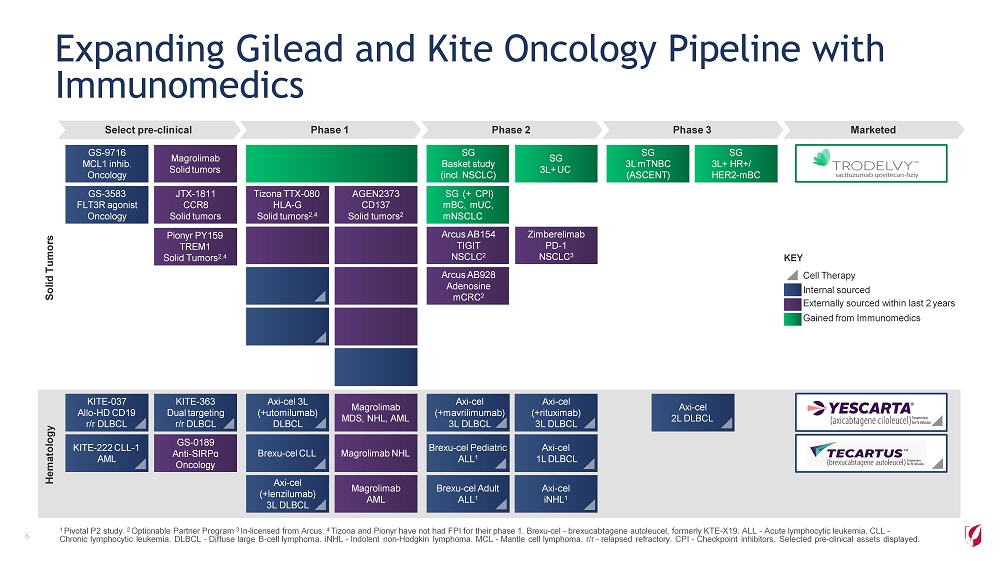
Select pre - clinical Phase 1 Phase 2 Phase 3 Marketed Hematology Solid Tumors Magrolimab NHL KITE - 222 CLL - 1 AML Brexu - cel CLL Brexu - cel Pediatric ALL 1 Axi - cel 1L DLBCL M ag r o li m ab AML Axi - cel ( + l en z il u m ab ) 3L DLBCL Brexu - cel Adult ALL 1 A x i - c e l iNHL 1 SG 3L mTNBC (ASCENT) SG 3L+ HR+/ H E R 2 - m BC SG Basket study (incl. NSCLC) SG 3L+ UC SG (+ CPI) mBC, mUC, mNSCLC Arcus AB154 TIGIT NSCLC 2 Z i m be r e li m a b PD - 1 NSCLC 3 Arcus AB928 Adenosine mCRC 2 JT X - 1811 CCR8 Solid tumors GS - 3583 FLT3R agonist Oncology Magrolimab Solid tumors GS - 9716 MCL1 inhib. Oncology Sacituzumab govitecan (SG) mTNBC, mUC, Ovarian (+ PARPi) A G E N 2373 CD137 Solid tumors 2 AGEN1223 Bi - specific Solid tumors 2 GS - 1423 CD73/TGFβ Solid tumors Ar c us CD 7 3 Solid tumors 2 GS - 422 4 PD - L1 NSCLC Tizona TTX - 080 HLA - G Solid tumors 2,4 Pionyr PY314 TREM2 Solid Tumors 2,4 KITE - 718 MAGE - A3/A6 Solid tumor KITE - 439 HPV - 16 E7 Solid tumor Magrolimab MDS, NHL, AML KITE - 363 Dual targeting r/r DLBCL GS - 0189 An t i - S IR P ɑ Oncology Axi - cel 3L ( + u t o m il u m ab ) DLBCL KITE - 037 Allo - HD CD19 r/r DLBCL Axi - cel ( + m a v rili m u m ab) 3L DLBCL Axi - cel ( + ri t u x i m ab) 3L DLBCL Axi - cel 2L DLBCL 1 Pivotal P2 study. 2 Optionable Partner Program 3 In - licensed from Arcus. 4 Tizona and Pionyr have not had FPI for their phase 1. Brexu - cel - brexucabtagene autoleucel, formerly KTE - X19. ALL - Acute lymphocytic leukemia. CLL - Chronic lymphocytic leukemia. DLBCL - Diffuse large B - cell lymphoma. iNHL - Indolent non - Hodgkin lymphoma. MCL - Mantle cell lymphoma. r/r - relapsed refractory. CPI - Checkpoint inhibitors. Selected pre - clinical assets displayed. Expanding Gilead and Kite Oncology Pipeline with Immunomedics 6 Internal sourced Externally sourced within last 2 years Gained from Immunomedics KEY Cell Therapy Pionyr PY159 TREM1 Solid Tumors 2,4
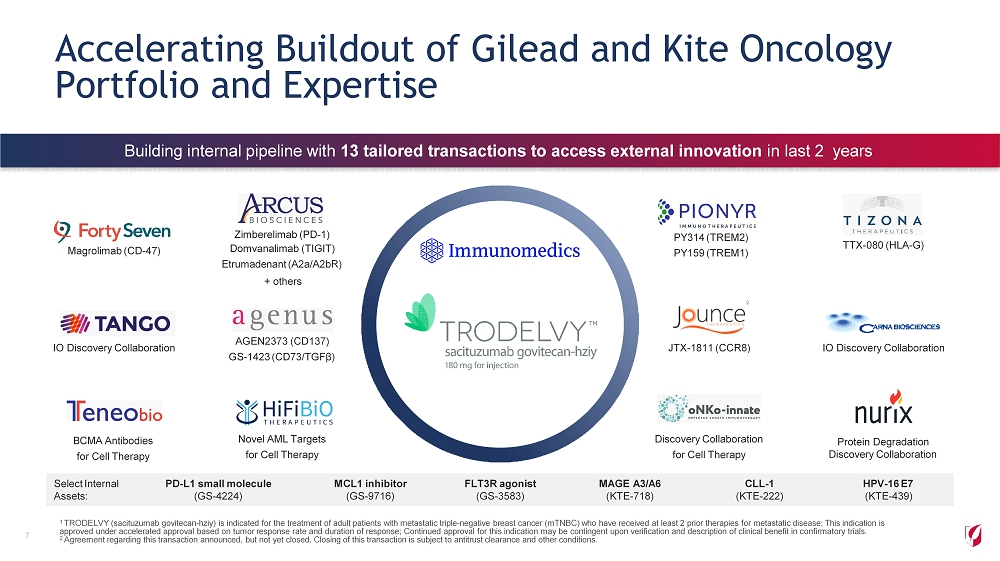
Select Internal Assets: PD - L1 small molecule (GS - 4224) MCL1 inhibitor (GS - 9716) FLT3R agonist (GS - 3583) MAGE A3/A6 (KTE - 718) CLL - 1 (KTE - 222) HPV - 16 E7 (KTE - 439) Accelerating Buildout of Gilead and Kite Oncology Portfolio and Expertise 7 Magrolimab (CD - 47) Zimberelimab (PD - 1) Domvanalimab (TIGIT) Etrumadenant (A2a/A2bR) + others PY314 (TREM2) PY159 (TREM1) 2 TTX - 080 (HLA - G) IO Discovery Collaboration AGEN2373 (CD137) GS - 1423 (CD73/TGFβ) F i r s t - i n - c l as s conjugate for JTX - 1811 (CCR8) antibody drug triple negative IO Discovery Collaboration BCMA Antibodies for Cell Therapy b r eas t Novel AML Targets for Cell Therapy cancer 1 Discovery Collaboration for Cell Therapy Protein Degradation Discovery Collaboration 1 TRODELVY (sacituzumab govitecan - hziy) is indicated for the treatment of adult patients with metastatic triple - negative breast cancer (mTNBC) who have received at least 2 prior therapies for metastatic disease; This indication is approved under accelerated approval based on tumor response rate and duration of response; Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trials. 2 Agreement regarding this transaction announced, but not yet closed. Closing of this transaction is subject to antitrust clearance and other conditions. Building internal pipeline with 13 tailored transactions to access external innovation in last 2 years

Trodelvy Overview 8 • Trodelvy (sacituzumab govitecan) is the first ADC approved for r/r metastatic triple negative breast cancer - Highly specific antibody targets Trop - 2, an epithelial antigen over - expressed across a range of solid tumors - Validated SN - 38 payload kills cancers by damaging DNA - Hydrolysable linker facilitates intra - and extra - cellular (bystander effect) killing of tumor cells • Trodelvy has demonstrated activity across a range of solid tumors , including HR+/HER2 - metastatic breast and urothelial cancers • Broad development program offers potential expansion opportunities Humanized anti - Trop - 2 antibody • Targets Trop - 2, an epithelial antigen expressed on a range of solid tumors SN - 38 payload • SN - 38 is the active metabolite of irinotecan Linker for SN - 38 • Hydrolysable linker for payload release
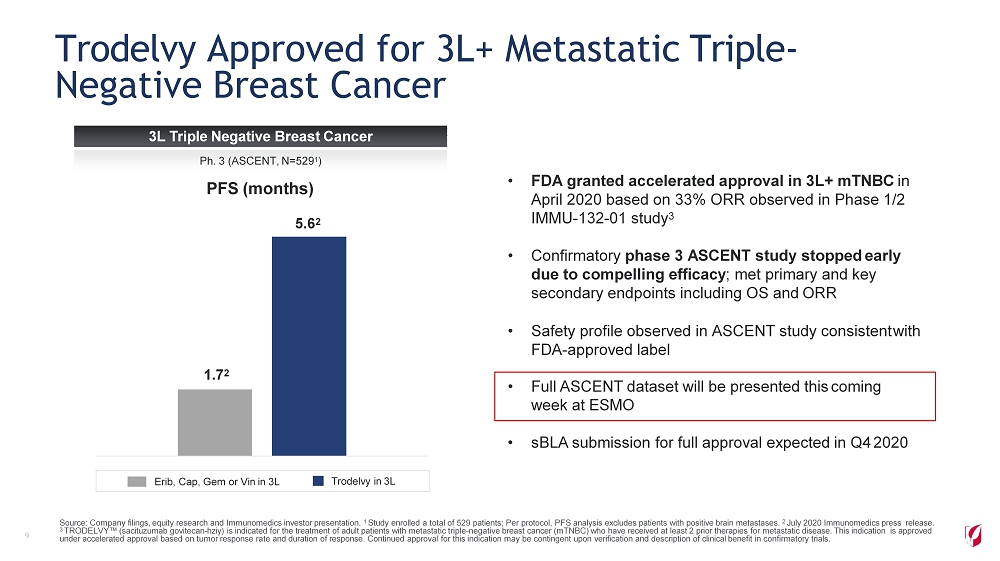
Trodelvy Approved for 3L+ Metastatic Triple - Negative Breast Cancer 9 • FDA granted accelerated approval in 3L+ mTNBC in April 2020 based on 33% ORR observed in Phase 1/2 IMMU - 132 - 01 study 3 • Confirmatory phase 3 ASCENT study stopped early due to compelling efficacy ; met primary and key secondary endpoints including OS and ORR • Safety profile observed in ASCENT study consistent with FDA - approved label • Full ASCENT dataset will be presented this coming week at ESMO • sBLA submission for full approval expected in Q4 2020 PFS (months) Erib, Cap, Gem or Vin in 3L Trodelvy in 3L 1 . 7 2 5 . 6 2 Source: Company filings, equity research and Immunomedics investor presentation. 1 Study enrolled a total of 529 patients; Per protocol, PFS analysis excludes patients with positive brain metastases. 2 July 2020 Immunomedics press release. 3 TRODELVY™ (sacituzumab govitecan - hziy) is indicated for the treatment of adult patients with metastatic triple - negative breast cancer (mTNBC) who have received at least 2 prior therapies for metastatic disease. This indication is approved under accelerated approval based on tumor response rate and duration of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trials. 3L Triple Negative Breast Cancer Ph. 3 (ASCENT, N=529 1 )
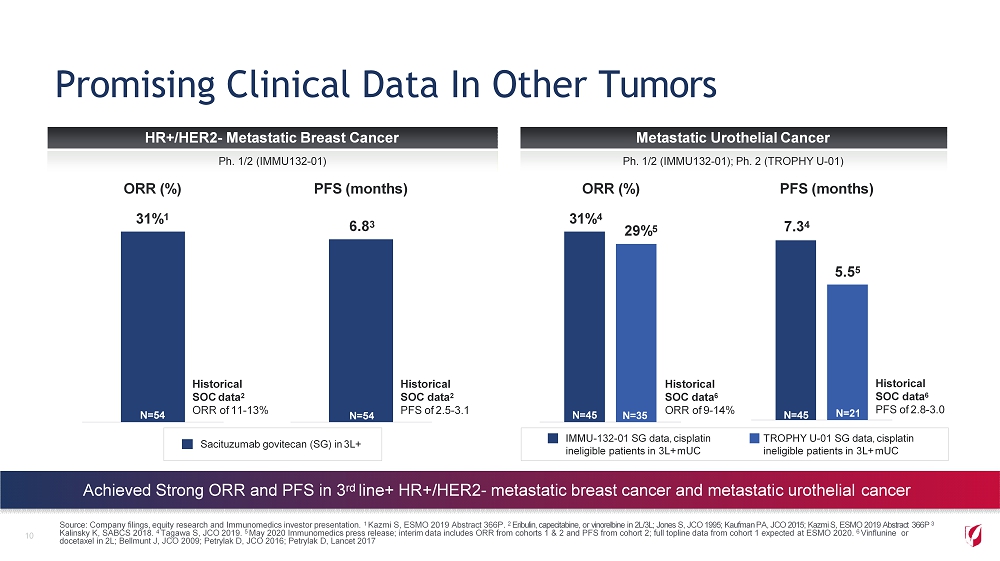
Promising Clinical Data In Other Tumors 10 HR+/HER2 - Metastatic Breast Cancer Metastatic Urothelial Cancer Ph. 1/2 (IMMU132 - 01) Ph. 1/2 (IMMU132 - 01); Ph. 2 (TROPHY U - 01) Source: Company filings, equity research and Immunomedics investor presentation. 1 Kazmi S, ESMO 2019 Abstract 366P. 2 Eribulin, capecitabine, or vinorelbine in 2L/3L; Jones S, JCO 1995; Kaufman PA, JCO 2015; Kazmi S, ESMO 2019 Abstract 366P 3 Kalinsky K, SABCS 2018. 4 Tagawa S, JCO 2019. 5 May 2020 Immunomedics press release; interim data includes ORR from cohorts 1 & 2 and PFS from cohort 2; full topline data from cohort 1 expected at ESMO 2020. 6 Vinflunine or docetaxel in 2L; Bellmunt J, JCO 2009; Petrylak D, JCO 2016; Petrylak D, Lancet 2017 Achieved Strong ORR and PFS in 3 rd line+ HR+/HER2 - metastatic breast cancer and metastatic urothelial cancer Sacituzumab govitecan (SG) in 3L+ Historical SOC data 2 ORR of 11 - 13% N=54 Historical SOC data 2 PFS of 2.5 - 3.1 N=54 6 . 8 3 IMMU - 132 - 01 SG data, cisplatin ineligible patients in 3L+ mUC TROPHY U - 01 SG data, cisplatin ineligible patients in 3L+ mUC Historical SOC data 6 ORR of 9 - 14% 7 . 3 4 29 % 5 5 . 5 5 N=45 N=35 Historical SOC data 6 PFS of 2.8 - 3.0 ORR (%) PFS (months) 31% 4 ORR (%) PFS (months) 31% 1 N=45 N=21
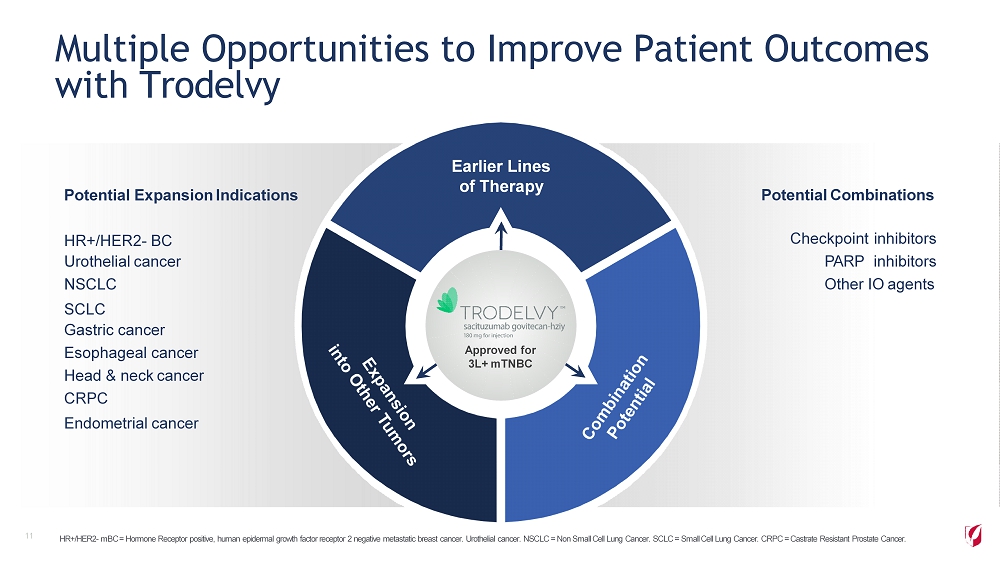
Multiple Opportunities to Improve Patient Outcomes with Trodelvy 11 Potential Expansion Indications HR+/HER2 - BC Urothelial cancer NSCLC SCLC Gastric cancer Esophageal cancer Head & neck cancer CRPC Endometrial cancer Potential Combinations Checkpoint inhibitors PARP inhibitors Other IO agents Earlier Lines of Therapy Approved for 3L+ mTNBC HR+/HER2 - mBC = Hormone Receptor positive, human epidermal growth factor receptor 2 negative metastatic breast cancer. Urothelial cancer. NSCLC = Non Small Cell Lung Cancer. SCLC = Small Cell Lung Cancer. CRPC = Castrate Resistant Prostate Cancer.

Breadth of Trodelvy Studies Provides Opportunity for Expansion 12 Trial Indication Phase 1 Phase 2 Phase 3 Approved IMMU - 132 - 01 mTNBC (3L+) ASCENT mTNBC (3L) S tudy stopped early due to compelling efficacy TROPiCS - 02 HR+/HER2 - mBC (3L+) TROPHY U - 01 Urothelial (3L+) P o t e n tially registrational TROPiCS - 03 Basket (mNSCLC / H&N / mSCLC / endometrial / HCC) MORPHEUS mTNBC (1L) / mUC / mNSCLC (+Tecentriq) SEASTAR mTNBC / mUC / Ovarian (2L+) (+ Rubraca) Source: Company Investor Presentation May 2020 and equity research. Information regarding partnerships is subject to confirmation in legal diligence. 1 Clinical pipeline shown does not include investigator sponsored trials (ISTs). These ISTs include collaborations 1) with German Breast Group to evaluate Trodelvy in HER2 - breast cancer in the post - neoadjuvant setting, 2) with Dana Farber Cancer Institute and Merck to evaluate Trodelvy + Keytruda in advanced breast cancers, 3) with Massachusetts General Hospital to evaluate Trodelvy in TNBC in the neoadjuvant setting and Trodelvy + Talzenna in 2L mTNBC, and 4) has further collaborations with Yale, U of Wisconsin and UT Health at San Antonio to evaluate Trodelvy in other solid tumor types. 2 Partnered with Everest for oncology in Greater China, South Korea and part of Southeast Asia in exchange for 1) $65m upfront; 2) $60m milestone on U.S. FDA Approval in mTNBC; 3) R&D and commercial milestones of up to $710m; and 4) tiered mid - teens royalties.
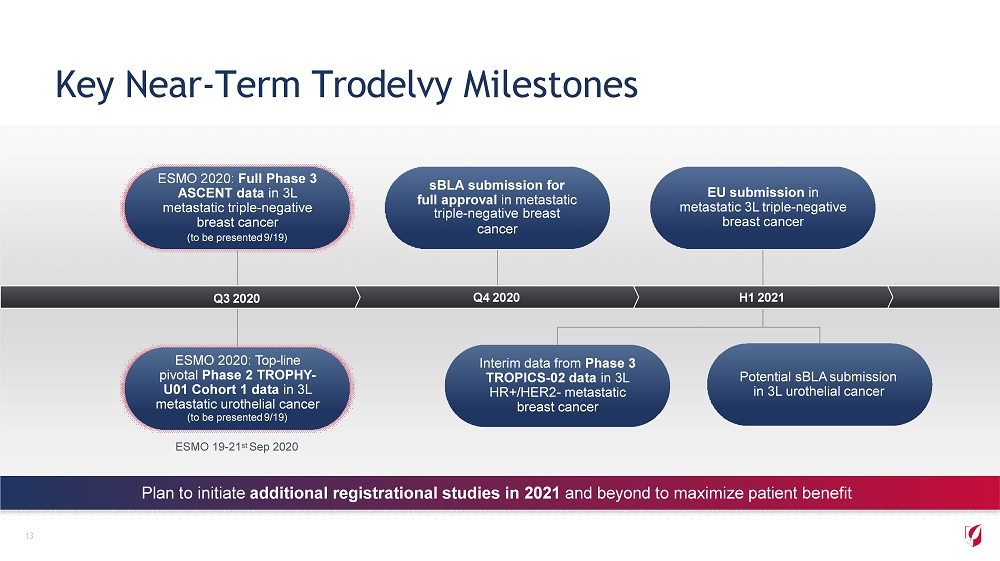
Key Near - Term Trodelvy Milestones ESMO 2020: Full Phase 3 ASCENT data in 3L metastatic triple - negative breast cancer (to be presented 9/19) ESMO 2020: Top - line pivotal Phase 2 TROPHY - U01 Cohort 1 data in 3L metastatic urothelial cancer (to be presented 9/19) sBLA submission for full approval in metastatic triple - negative breast cancer EU submission in metastatic 3L triple - negative breast cancer Interim data from Phase 3 TROPICS - 02 data in 3L HR+/HER2 - metastatic breast cancer Potential sBLA submission in 3L urothelial cancer 13 Plan to initiate additional registrational studies in 2021 and beyond to maximize patient benefit Q3 2020 Q4 2020 H1 2021 ESMO 19 - 21 st Sep 2020

Transaction Details 14 Transaction provides immediate revenue diversification and significantly improves growth prospects • Gilead to acquire Immunome - Tender offer is not subject to a - Expect to fund acquisition with - Does not alter stated capital al dics for $88.00 per share in cash for a total purchase price of ~$21 billion financing condition ~$15 billion of existing cash and ~$6 billion in newly issued debt location strategy or commitment to maintain and grow dividend neutral or accretive to non - GAAP earnings in 2023 and significantly accretive • Transaction anticipated to be thereafter • Close expected in Q4 2020 , subject to regulatory approval and other customary closing conditions

Trodelvy Supports Strategy to Drive Additional Growth and Complements YTD Progress 15 Strategy to Drive Additional Growth x Trodelvy foundational asset for solid tumors x 15 tailored transactions YTD x Accelerating efforts to build IO portfolio and expertise x Optimizing portfolio through strategic evaluation Durable Core Business x Biktarvy #1 across markets x Descovy for PrEP launch success x HCV market share maintained x Growth in China continues Existing Pipeline Opportunities x Remdesivir for COVID - 19 x Tecartus approved for r/r MCL x Filgotinib positive European CHMP opinion for RA x Lenacapavir for long - acting HIV x Magrolimab for MDS and AML + + Well positioned to maximize near - term opportunities and achieve long - term success

THANK YOU CONTACT US investor_relations@gilead.com investors.gilead.com

















































