| CONTACT: | Investors | Media | ||
| EVC Group | EVC Group | |||
| Jenifer Kirtland, 415-568-9349 | Christopher Gale, 646-201-5431 | |||
| Douglas Sherk, 415-652-9100 |
STAAR Surgical Reports First Quarter 2012 Financial Results
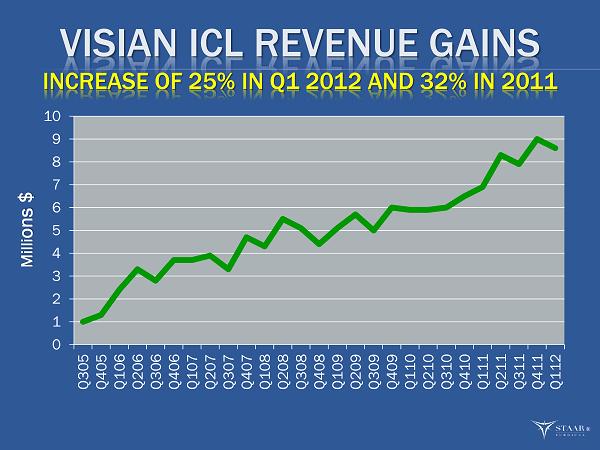
Visian® ICL™ Revenue Increases 25% Year over Year
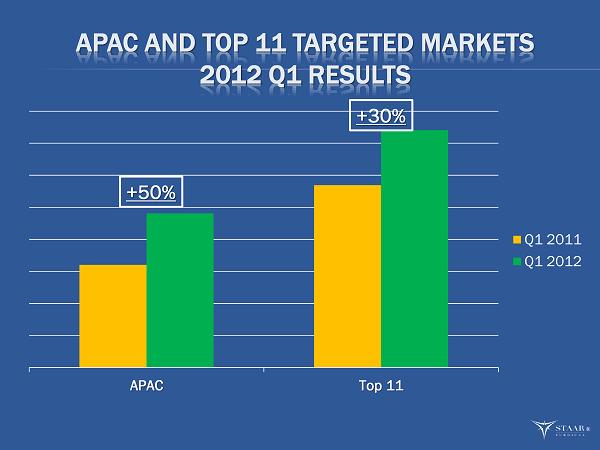
Visian ICL Revenue in Asia/Pacific Region Up 50%
Record Gross Margin of 70.3%, 550 Basis Points Above 1Q 2011
GAAP Net Income of $0.01 Per Diluted Share/Non-GAAP Adjusted Net Income of $0.04
Company Reiterates Confidence in Achieving 2012 Metrics
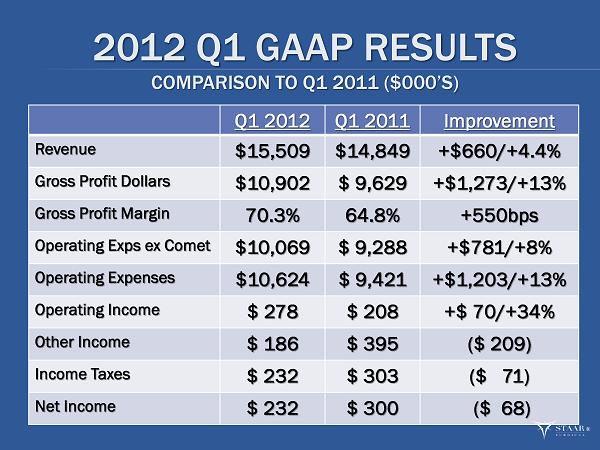
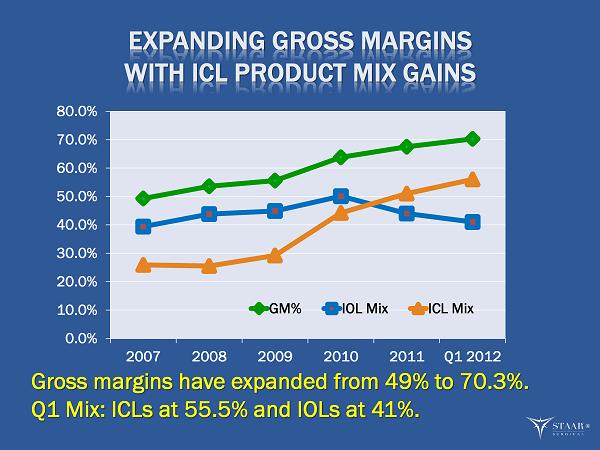
MONROVIA, CA, May 2, 2012 -- STAAR Surgical Company (NASDAQ: STAA), a leading developer, manufacturer and marketer of minimally invasive ophthalmic products, today reported that sales of its Visian ICL product in the first quarter ended March 30, 2012 increased 25% to $8.6 million. Total revenue grew 4% to $15.5 million from $14.8 million in the first quarter of 2011. Gross margin set another quarterly record at 70.3%, 550 basis points above the 64.8% margin in the first quarter of 2011 and 50 basis points above the 69.8% margin in the fourth quarter of 2011. This was the highest gross margin since 1998 and represented the sixth consecutive quarterly gross margin increase. The gross margin improvement continues to reflect the increased contribution of Visian ICL sales and margin improvement in IOLs.
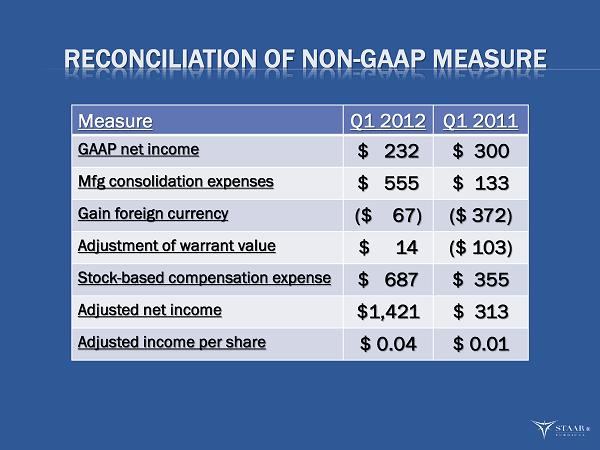
Net income, calculated in accordance with GAAP, was $232,000, or $0.01 per diluted share, compared with net income of $300,000, also $0.01 per diluted share, in the first quarter of 2011. This marked the fifth consecutive quarter of profitability for the Company. Adjusted net income (excluding manufacturing consolidation expenses, gain on foreign currency transactions, fair value adjustment of warrants, and non-cash share-based compensation expense) for the quarter ended March 30, 2012 was $1.4 million, or $0.04 per diluted share. Adjusted net income for the year ago quarter was $313,000, or $0.01 per diluted share. Cash and cash equivalents at March 30, 2012 totaled $16.4 million.
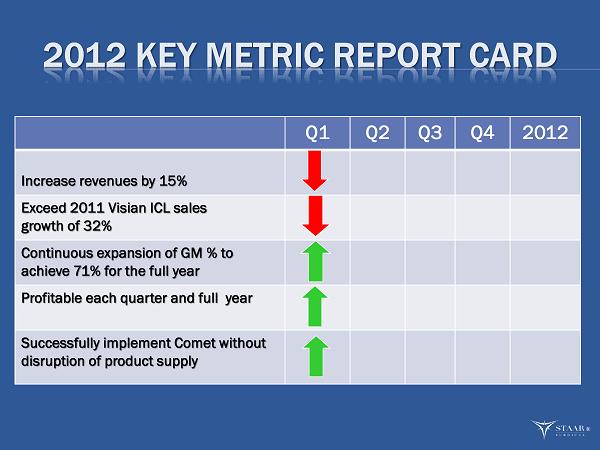
“We continued to make good progress in our business during the first quarter evidenced by our 30% increase of ICL revenues in our top 11 targeted markets,” said Barry G. Caldwell, President and CEO. “This increase in ICL sales combined with our product cost improvements led to significant year over year expansion in gross margin. Our IOL sales were not as strong as expected but we understand why and expect growth for the remainder of the year. Our key metrics are developed on an annualized basis and our first quarter is always the most challenging when compared to our annualized targets. The 2012 start is very similar to the start of 2011 when we missed two of our metrics in the first quarter; only to exceed them as the year progressed. We remain confident that we will achieve our metrics for 2012 which include ICL revenue growth in excess of 32 % and total revenue growth of 15%.”
“We continue to make progress in the roll-out of the V4c ICL in markets where approved,” added Mr. Caldwell. “Our participation at recent ophthalmology conferences in the Middle East and U.S. has confirmed the growing interest in Visian ICL as a true competitor to both standard and customized LASIK with significant patient benefits including improved vision, comfort and safety. The V4c ICL with CentraFLOW technology is gaining traction in Europe and represented 58% of total European ICL sales in the quarter. Surgeons in Europe have already implanted over 1,000 V4c ICLs and the AquaPORT™ has performed as expected in all cases. This data further proves the CentraFLOW technology eliminates the need for an iridotomy prior to surgery making the procedure much more convenient and less complicated for both the surgeon and patient.”
“We reached another key milestone in gross profit margin expansion by achieving a 70.3% gross margin for the first quarter, our highest margin in 14 years,” Mr. Caldwell continued. “Our increased gross margin was due largely to the improved mix toward higher margin ICL sales and improved costs. ICL gross margin for the quarter was 85%, while our IOL margin continued to expand to 60% during the quarter. We expect continued margin improvement during the year due to increased ICL sales and further market penetration of the V4c version.”
“Our operating income in the first quarter increased 34% compared with the first quarter of 2011. We continued execution on our plan to consolidate manufacturing and recorded approximately $0.6 million in expenses for that effort in the first quarter. Net income for the recent quarter of $0.2 million was below last year’s $0.3 million, attributable to the increase in manufacturing consolidation expenses and the $0.3 million lower gain on foreign currency transactions. Looking ahead, we expect enhanced growth in sales of Visian ICL and total revenue based on the ramp of our newly approved product introductions, expanded awareness of the benefits of our technology and the improvements we continue to make in our operations. We expect to meet all of the key metrics we established earlier this year for the full year 2012,” Mr. Caldwell concluded.
2012 Key Metrics
The Company reaffirmed its key target metrics for 2012:
| 1. | Increase in revenues of 15%. |
| 2. | Exceed the 2011 Visian ICL revenue growth rate of 32%. |
| 3. | Continuous expansion of gross margins to achieve 71% for the full year. |
| 4. | Profitable each quarter and for the full year. |
| 5. | Implement manufacturing consolidation with no disruption to customer supply requirements. This metric will be measured by the amount of customer backorders. |
| 2 |
Recent Visian Implantable Collamer® Lens (ICL) Highlights
| · | First quarter Visian ICL sales grew to $8.6 million, a 25% increase from $6.9 million in the first quarter of 2011. |
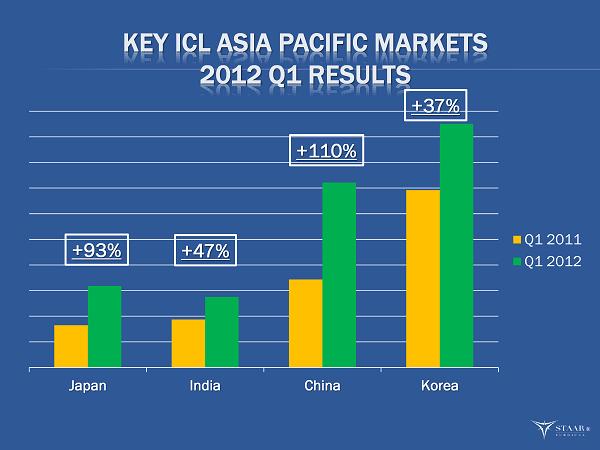
| · | Sales in STAAR’s top 11 targeted markets increased 30% in the first quarter. Asia/Pacific sales grew 50% in the first quarter. Leading the growth were China, up 110%; Japan up 93%; India, up 47%; and Korea, up 37%. |
| · | Sales in EMEA grew an unusually slow 2% in the quarter, primarily due to three factors. First, during March 2011 the Company had two distributors stocking the expanded range for the V4b which had just been introduced. With this introduction there were new powers which needed to be placed into their inventories. Secondly, the WOC held in February slowed refractive surgery particularly in the Mideast. This meeting was not held in 2011. Finally, the Company had two fairly large ICL orders which were delayed at quarter end due to some logistics issues. |
| · | 58% of total ICL sales to European markets during the first quarter of 2012 were of the Visian V4c ICL. There have been over 1,000 implants of the V4c in Europe with the AquaPORT performing as expected in all cases. |
| · | ICL sales in North America increased by 4% during the quarter. U.S. ICL sales in the first quarter grew 2%. Sales to private sector accounts increased by 6% while military sales of ICLs during the quarter decreased 34% from the prior year due to key ICL surgeon deployment or retirement. The Company expects to see an improvement in military ICL sales during the second half of 2012. |
Recent Intraocular Lens (IOL) Highlights
| · | First quarter IOL sales decreased 11% or $0.8 million to $6.4 million compared with $7.1 million in the first quarter of 2011. |
| · | During the first quarter of 2011 the Company transitioned the business in Australia from a direct distribution model to an indirect model. In that transition it sold its inventory to the new distributor creating a revenue comparison for the quarter unrelated to actual demand. That negative impact for comparative purposes was over $0.4 million or approximately half of the quarter over quarter decline. |
| · | Preloaded IOLs in Japan declined by 7% or $0.2 million. Key factors were aggressive competitive pricing during the quarter though the Company’s price actually increased by 2%. Finalizing the KS-SP preloaded single piece acrylic IOL is important for that market. The Company believes it has now solved the technical issues regarding delivery of the lens. A focus remains on gaining incremental university accounts where the average prices are higher. During the month of April the Company saw a return to IOL revenue growth in Japan. |
| · | In the U.S., silicone IOL sales decreased nearly $0.3 million. Sales of nanoFLEX and Silicone Toric IOLs did not outpace the loss in standard silicone lenses. |
| · | IOL gross margins improved by 200 basis points to 60% for the quarter. Cost improved reflecting an improved gross margin percentage in all five categories of IOLs year over year. |
| · | Shipments of the nanoFLEX IOL have occurred for on-going trials with surgeons in two key European markets. The pre-market launch of the newly approved nanoFLEX Toric IOLs are expected to begin during the second quarter, at which time, the Company will introduce both nanoFLEX models to additional European markets. |
| 3 |
First Quarter Financial Highlights
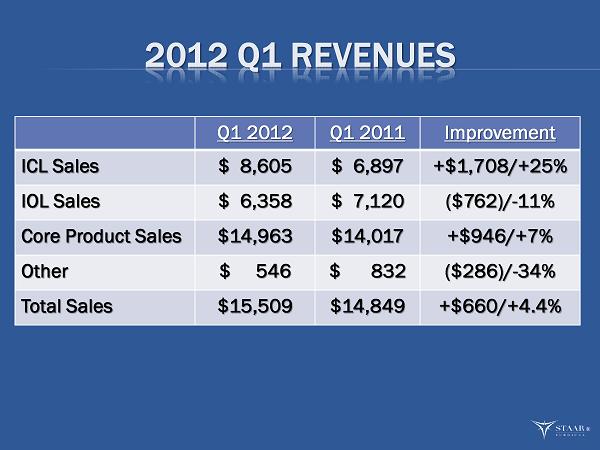
| · | Total net sales in the first quarter increased 4%, or $0.7 million, to $15.5 million from $14.8 million in the first quarter of 2011. Foreign currency transactions had a favorable impact on sales of $0.1 million which is $0.3 million less gain than the prior year first quarter impact. Net sales were also affected by a $0.3 million, or 34%, decline in sales of the “other” product category on which the Company is not focused. |
| · | Gross margin increased to a record 70.3% of revenue compared with 64.8% in the prior year’s first quarter. The increase reflected a higher mix of ICL to total sales of 56%, as compared to 46% during the first quarter of 2011, and a 200 basis point improvement in IOL margins. |
| · | Total operating expenses were $10.6 million, a 13% increase over $9.4 million in the first quarter of 2011. Manufacturing consolidation expenses totaled $0.6 million during the quarter. Excluding consolidation expenses, operating expenses were $10.1 million, a 8% increase over the 2011 first quarter. The non-cash charge during the quarter for stock-based compensation increased by $332,000 despite the issuance of the same number of options, reflecting an increase in the Company’s stock price from the prior year period. |
| · | Other expenses contributed $186,000 versus $395,000 in the prior year period. This was driven by the over $300,000 decrease in gains on foreign currency transactions offset to some degree by an increase in royalty income and a decrease in interest expense. |
| · | GAAP net income in the first quarter of 2012 totaled $232,000, or $0.01 per diluted share, compared with net income in the first quarter last year of $300,000, or $0.01 per diluted share. Adjusted net income was $1.4 million, or $0.04 per diluted share, compared with adjusted net income during the first quarter of 2011 of $0.3 million, or $0.01 per diluted share. |
| · | Cash and cash equivalents and restricted cash totaled $16.4 million at March 30, 2012 compared with $16.7 million at December 30, 2011 and $10.4 million at April 1, 2011. The Company used $0.4 million in cash for operating activities during the first quarter due to expenditures associated with the manufacturing consolidation including an investment in inventories. This investment in inventory is to help guarantee no disruption in customer supply during the transitions and $0.4 million in cash was used for this purpose during the quarter. |
Second Quarter and Remainder of Year Key Events
| · | Continued penetration of the V4c ICL into the currently approved markets. Penetration rate was 58% during first quarter. |
| · | Expanding the approval of the V4c ICL to additional targeted markets outside of Europe. |
| · | Release of nanoFLEX Toric IOL for pre-market launch to Europe which is planned during the second quarter of 2012. |
| · | Complete the transfer of the enhanced KS-SP preloaded single piece acrylic from R&D to manufacturing in order to begin shipping this new product. |
| · | With the completion of the R&D inventions for the V5 Preloaded ICL, complete the file for CE Mark and plan to gain additional surgeon input and support prior to the ESCRS meeting in Milan during September. |
| 4 |
Conference Call
The Company will host a conference call and video webcast today, May 2, 2012 at 4:30 p.m. Eastern / 1:30 p.m. Pacific to discuss the Company's first quarter 2012 financial results and recent corporate developments. The dial-in number for the conference call is 877-941-9205 for domestic participants and 480-629-9771 for international participants.
The Company will also be using slides to illustrate its first quarter 2012 results and operational progress. The slides and live webcast of the call can be accessed from the investor relations section of the STAAR website at www.staar.com.
A taped replay of the conference call will also be available beginning approximately one hour after the call’s conclusion and will be available for seven days. This replay can be accessed by dialing 800-406-7325 for domestic callers and 303-590-3030 for international callers, both using passcode 4531978#. An archived webcast will also be available at www.staar.com.
Use of Non-GAAP Financial Measures
This press release includes supplemental non-GAAP financial information, which STAAR believes investors will find helpful in understanding its operating performance. “Adjusted Net Income” excludes the following items that are included in “Net Income” as calculated in accordance with U.S. generally accepted accounting principles (“GAAP”): manufacturing consolidation expenses, gain or loss on foreign currency transactions, the fair value adjustment of outstanding warrants issued in 2007, and stock-based compensation expenses.
We believe that “Adjusted Net Income” is useful to investors in gauging the outcome of the key drivers of our business performance: our ability to increase sales revenue and our ability to increase profit margin by improving the mix of high value products while reducing the costs over which we have control.
We have excluded manufacturing consolidation expenses because these are non-recurring expenses and their inclusion may mask underlying trends in our business performance.
We have excluded gains and losses on foreign currency transactions and the fair value adjustment of warrants because of the significant fluctuations that can result from period to period as a result of market driven factors.
| 5 |
Stock-based compensation expenses consist of expenses for stock options and restricted stock under Statement of Financial Accounting Standards (“SFAS”) No. 123R. In calculating Adjusted Net Income STAAR excludes these expenses and the fair value adjustment of outstanding warrants because they are non-cash expenses and because of the complexity and considerable judgment involved in calculating their values. In addition, these expenses tend to be driven by fluctuations in the price of our stock and not by the same factors that generally affect our other business expenses.
We have provided below a detailed reconciliation table, which is useful to investors in providing the context to understand our Adjusted Net Income and how it differs from Net Income calculated in accordance with GAAP.
About STAAR Surgical
STAAR, which has been dedicated solely to ophthalmic surgery for over 25 years, designs, develops, manufactures and markets implantable lenses for the eye. All of these lenses are foldable, which permits the surgeon to insert them through a small incision. STAAR’s lens used in refractive surgery as an alternative to LASIK is called an Implantable Collamer® Lens or “ICL.” A lens used to replace the natural lens after cataract surgery is called an intraocular lens or “IOL.” Over 300,000 Visian ICLs have been implanted to date; to learn more about the ICL go to: www.visianinfo.com. STAAR has approximately 300 full time employees and markets lenses in approximately 50 countries. Headquartered in Monrovia, CA, it manufactures in the following locations: Nidau, Switzerland; Ichikawa City, Japan; Aliso Viejo, CA; and Monrovia, CA. For more information, please visit the Company’s website at www.staar.com or call 626-303-7902.
Collamer® is the registered trademark for STAAR’s proprietary biocompatible collagen copolymer lens material.
Safe Harbor
All statements in this press release that are not statements of historical fact are forward-looking statements, including statements about any of the following: any projections of earnings, revenue, sales, profit margins, cash or any other financial items; the plans, strategies, and objectives of management for future operations or prospects for achieving such plans; prospects for increased sales as a result of new product introductions or new product approvals, expected savings from business consolidation plans and the timetable for those plans; the approval of pending regulatory applications; statements of belief; and any statements of assumptions underlying any of the foregoing.
These statements are based on expectations and assumptions as of the date of this press release and are subject to numerous risks and uncertainties, which could cause actual results to differ materially from those described in the forward-looking statements. The risks and uncertainties include the following: our limited capital resources and limited access to financing; the negative effect of poor global economic conditions on sales of products, especially products such as the ICL used in non-reimbursed elective procedures; the challenge of managing our foreign subsidiaries; backlog as we ramp up production to meet rapidly growing demand for our products; the risk of unfavorable changes in currency exchange rate; the discretion of regulatory agencies to approve or reject new products, or to require additional actions before approval; unexpected costs or delays that could reduce or eliminate the expected benefits of our consolidation plans; the risk that research and development efforts will not be successful or may be delayed in delivering for launch; the willingness of surgeons and patients to adopt a new product and procedure; patterns of Visian ICL use that have typically limited our penetration of the refractive surgery market, and a general decline in the demand for refractive surgery particularly in the U.S., which STAAR believes has resulted from both concerns about the safety and effectiveness of laser procedures and current economic conditions. The Visian Toric ICL and the V4c are not yet approved for sale in the United States.
| 6 |
| STAAR Surgical Company | ||||||||
| Condensed Consolidated Balance Sheets | ||||||||
| (in 000's) | ||||||||
| March 30, | December 30 | |||||||
| 2012 | 2011 | |||||||
| ASSETS | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | 16,445 | $ | 16,582 | ||||
| Restricted cash | - | 129 | ||||||
| Accounts receivable trade, net | 8,331 | 9,089 | ||||||
| Inventories, net | 11,145 | 10,933 | ||||||
| Prepaids, deposits, and other current assets | 2,583 | 1,921 | ||||||
| Total current assets | 38,504 | 38,654 | ||||||
| Property, plant, and equipment, net | 4,171 | 4,222 | ||||||
| Intangible assets, net | 2,673 | 2,989 | ||||||
| Goodwill | 1,786 | 1,786 | ||||||
| Deferred income taxes | 151 | 152 | ||||||
| Other assets | 1,135 | 1,203 | ||||||
| Total assets | $ | 48,420 | $ | 49,006 | ||||
| LIABILITIES AND STOCKHOLDERS' EQUITY | ||||||||
| Current liabilities: | ||||||||
| Line of credit | $ | 2,420 | $ | 2,580 | ||||
| Accounts payable | 3,082 | 4,261 | ||||||
| Deferred income taxes | 472 | 472 | ||||||
| Obligations under capital leases | 869 | 597 | ||||||
| Other current liabilities | 5,661 | 6,106 | ||||||
| Total current liabilities | 12,504 | 14,016 | ||||||
| Obligations under capital leases | 736 | 1,124 | ||||||
| Deferred income taxes | 766 | 708 | ||||||
| Other long-term liabilities | 3,691 | 3,700 | ||||||
| Total liabilities | 17,697 | 19,548 | ||||||
| Stockholders' equity: | ||||||||
| Common stock | 362 | 360 | ||||||
| Additional paid-in capital | 158,944 | 157,383 | ||||||
| Accumulated other comprehensive income | 1,875 | 2,405 | ||||||
| Accumulated deficit | (130,458 | ) | (130,690 | ) | ||||
| Total stockholders' equity | 30,723 | 29,458 | ||||||
| Total liabilities and stockholders' equity | $ | 48,420 | $ | 49,006 | ||||
| STAAR Surgical Company | ||||||||||||||||||
| Condensed Consolidated Statements of Operations | ||||||||||||||||||
| (In 000's except for per share data) | ||||||||||||||||||
| Three Months Ended | ||||||||||||||||||
| % of | March 30, | % of | April 1, | Change | ||||||||||||||
| Sales | 2012 | Sales | 2011 | Amount | % | |||||||||||||
| Net sales | 100.0% | $ | 15,509 | 100.0% | $ | 14,849 | $ | 660 | 4.4% | |||||||||
| Cost of sales | 29.7% | 4,607 | 35.2% | 5,220 | 613 | -11.7% | ||||||||||||
| Gross profit | 70.3% | 10,902 | 64.8% | 9,629 | 1,273 | 13.2% | ||||||||||||
| Selling, general and administrative expenses: | ||||||||||||||||||
| General and administrative | 24.8% | 3,860 | 22.9% | 3,397 | 463 | 13.6% | ||||||||||||
| Marketing and selling | 30.1% | 4,663 | 30.0% | 4,459 | 204 | 4.6% | ||||||||||||
| Research and development | 10.0% | 1,546 | 9.6% | 1,432 | 114 | 8.0% | ||||||||||||
| Selling, general, and administrative expenses | 64.9% | 10,069 | 62.5% | 9,288 | 781 | 8.4% | ||||||||||||
| Other general and administrative expenses | 3.6% | 555 | 0.9% | 133 | 422 | 0.0% | ||||||||||||
| Total selling, general and administrative expenses | 68.5% | 10,624 | 63.4% | 9,421 | 1,203 | 12.8% | ||||||||||||
| Operating income | 1.8% | 278 | 1.4% | 208 | 70 | 33.7% | ||||||||||||
| Other income (expense): | ||||||||||||||||||
| Interest income | 0.0% | 0 | 0.1% | 13 | (13 | ) | -99.2% | |||||||||||
| Interest expense | -0.6% | (95 | ) | -1.0% | (153 | ) | 58 | -37.9% | ||||||||||
| Gain on foreign currency transactions | 0.4% | 67 | 2.5% | 372 | (305 | ) | -82.0% | |||||||||||
| Other income, net | 1.4% | 214 | 1.1% | 163 | 51 | 31.3% | ||||||||||||
| Total other income | 1.2% | 186 | 2.7% | 395 | (209 | ) | -52.9% | |||||||||||
| Income before provision for income taxes | 3.0% | 464 | 4.1% | 603 | (139 | ) | -23.0% | |||||||||||
| Provision for income taxes | 1.5% | 232 | 2.1% | 303 | (71 | ) | -23.4% | |||||||||||
| Net income | 1.5% | $ | 232 | 2.0% | $ | 300 | $ | (68 | ) | -22.6% | ||||||||
| Net Income per share-basic | $ | 0.01 | $ | 0.01 | ||||||||||||||
| Net Income per share-diluted | $ | 0.01 | $ | 0.01 | ||||||||||||||
| Weighted average shares outstanding - basic | 36,071 | 35,188 | ||||||||||||||||
| Weighted average shares outstanding - diluted | 38,420 | 36,389 | ||||||||||||||||
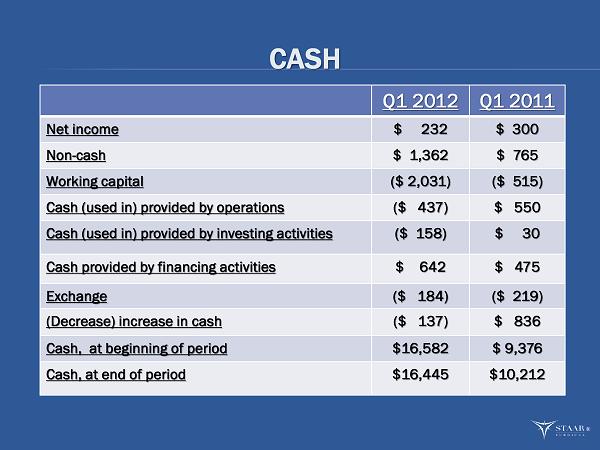
| STAAR Surgical Company | ||||||||
| Condensed Consolidated Statements of Cash Flows | ||||||||
| (in 000's) | ||||||||
| Year Ended | ||||||||
| March 30, | April 1, | |||||||
| 2012 | 2011 | |||||||
| Cash flows from operating activities: | ||||||||
| Net income | $ | 232 | $ | 300 | ||||
| Adjustments to reconcile net income to net cash (used in) provided by operating activities: | ||||||||
| Depreciation of property and equipment | 317 | 307 | ||||||
| Amortization of intangibles | 175 | 197 | ||||||
| Deferred income taxes | 57 | 44 | ||||||
| Fair value adjustment of warrant | 14 | (103 | ) | |||||
| Change in net pension liability | 72 | 60 | ||||||
| Gain on disposal of property and equipment | - | (14 | ) | |||||
| Stock-based compensation expense | 687 | 355 | ||||||
| Other | 40 | (81 | ) | |||||
| Changes in working capital: | ||||||||
| Accounts receivable, net | 556 | 666 | ||||||
| Inventories | (432 | ) | 548 | |||||
| Prepaids, deposits and other current assets | (665 | ) | (473 | ) | ||||
| Accounts payable | (1,100 | ) | (87 | ) | ||||
| Other current liabilities | (390 | ) | (1,169 | ) | ||||
| Net cash (used in) provided by operating activities | (437 | ) | 550 | |||||
| Cash flows from investing activities: | ||||||||
| Acquisition of property and equipment | (287 | ) | (44 | ) | ||||
| Release of restricted cash | 129 | - | ||||||
| Proceeds from the sale of property and equipment | - | 26 | ||||||
| Net change in other assets | - | 48 | ||||||
| Net cash (used in) provided by investing activities | (158 | ) | 30 | |||||
| Cash flows from financing activities: | ||||||||
| Repayment of capital lease lines of credit | (195 | ) | (131 | ) | ||||
| Proceeds from the exercise of stock options | 837 | 606 | ||||||
| Net cash provided by financing activities | 642 | 475 | ||||||
| Effect of exchange rate changes on cash and cash equivalents | (184 | ) | (219 | ) | ||||
| (Decrease) increase in cash and cash equivalents | (137 | ) | 836 | |||||
| Cash and cash equivalents, at beginning of year | 16,582 | 9,376 | ||||||
| Cash and cash equivalents, at end of year | $ | 16,445 | $ | 10,212 | ||||
| STAAR Surgical Company | ||||||||||||||
| Global Sales | ||||||||||||||
| (in 000's) | ||||||||||||||
| Three Months Ended | ||||||||||||||
| March 30, | April 1, | % | ||||||||||||
| Geographic Sales | 2012 | 2011 | Change | |||||||||||
| United States | 20.5% | $ | 3,174 | 23.8% | $ | 3,533 | -10.2% | |||||||
| Japan | 24.9% | 3,857 | 25.9% | 3,845 | 0.3% | |||||||||
| Korea | 12.3% | 1,903 | 9.3% | 1,384 | 37.5% | |||||||||
| China | 13.6% | 2,106 | 8.1% | 1,204 | 74.9% | |||||||||
| Other | 28.7% | 4,469 | 32.9% | 4,883 | -8.5% | |||||||||
| Total International Sales | 79.5% | 12,335 | 76.2% | 11,316 | 9.0% | |||||||||
| Total Sales | 100.0% | $ | 15,509 | 100.0% | $ | 14,849 | 4.4% | |||||||
| Product Sales | ||||||||||||||
| Core products | ||||||||||||||
| ICLs | 55.5% | $ | 8,605 | 46.4% | $ | 6,897 | 24.8% | |||||||
| IOLs | 41.0% | 6,358 | 48.0% | 7,120 | -10.7% | |||||||||
| Total core products | 96.5% | 14,963 | 94.4% | 14,017 | 6.7% | |||||||||
| Non-core products | ||||||||||||||
| Other | 3.5% | 546 | 5.6% | 832 | -34.4% | |||||||||
| Total Sales | 100.0% | $ | 15,509 | 100.0% | $ | 14,849 | 4.4% | |||||||
| STAAR Surgical Company | ||||||||
| Reconciliation of Non-GAAP Financial Measure | ||||||||
| Three Months Ended | ||||||||
| March 30, | April 1, | |||||||
| 2012 | 2011 | |||||||
| Net income - (as reported) | $ | 232 | $ | 300 | ||||
| Less: | ||||||||
| Manufacturing consolidation expenses | 555 | 133 | ||||||
| Gain on foreign currency | (67 | ) | (372 | ) | ||||
| Fair value adjustment of warrants | 14 | (103 | ) | |||||
| Stock-based compensation expense | 687 | 355 | ||||||
| Net income - (adjusted) | $ | 1,421 | $ | 313 | ||||
| Net income per share, basic - (as reported) | $ | 0.01 | $ | 0.01 | ||||
| Manufacturing consolidation expenses | $ | 0.02 | $ | 0.00 | ||||
| Gain on foreign currency | $ | (0.00 | ) | $ | (0.01 | ) | ||
| Fair value adjustment of warrants | $ | 0.00 | $ | (0.00 | ) | |||
| Stock-based compensation expense | $ | 0.02 | $ | 0.01 | ||||
| Net income per share, basic - (adjusted) | $ | 0.04 | $ | 0.01 | ||||
| Net income per share, diluted - (as reported) | $ | 0.01 | $ | 0.01 | ||||
| Manufacturing consolidation expenses | $ | 0.01 | $ | 0.00 | ||||
| Gain on foreign currency | $ | (0.00 | ) | $ | (0.01 | ) | ||
| Fair value adjustment of warrants | $ | 0.00 | $ | (0.00 | ) | |||
| Stock-based compensation expense | $ | 0.02 | $ | 0.01 | ||||
| Net income per share, diluted - (adjusted) | $ | 0.04 | $ | 0.01 | ||||
| Weighted average shares outstanding - Basic | 36,071 | 35,188 | ||||||
| Weighted average shares outstanding - Diluted | 38,420 | 36,389 | ||||||
| Note: Net income per share (adjusted), basic and diluted, may not add up due to rounding | ||||||||