UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K | ||
(Mark One)
x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2017
OR
¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from N/A to N/A
Commission file number: 0-10961
QUIDEL CORPORATION (Exact name of registrant as specified in its charter) |
DELAWARE | 94-2573850 |
(State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) |
12544 High Bluff Drive, Suite 200, San Diego, California | 92130 |
(Address of principal executive offices) | (Zip Code) |
858-552-1100
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
Title of each class | Name of each exchange on which registered |
Common stock, $0.001 par value | Nasdaq Global Market |
Securities registered pursuant to Section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ¨ No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ¨ No x
Indicate by check mark whether the Registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No ¨
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes x No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer”, “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer | x | Accelerated filer | ¨ | |
Non-accelerated filer | ¨ (Do not check if a smaller reporting company) | Smaller reporting company | ¨ | |
Emerging growth company | ¨ | |||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Securities Act. | ¨ | |||
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ¨ No x
The aggregate market value of the registrant’s common stock held by non-affiliates of the registrant was $747,331,220 based on the closing sale price at which the common stock was last sold, as of the last business day of the registrant’s most recently completed second fiscal quarter.
As of February 21, 2018, 34,852,455 shares of the registrant’s common stock were outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
(To the Extent Indicated Herein)
Portions of the registrant’s definitive proxy statement to be filed with the Securities and Exchange Commission in connection with the registrant’s 2018 Annual Meeting of Stockholders (to be held on May 15, 2018) are incorporated by reference into Part III, Items 10, 11, 12, 13 and 14 of this Annual Report on Form 10-K
QUIDEL CORPORATION
FORM 10-K
FOR THE FISCAL YEAR ENDED DECEMBER 31, 2017
TABLE OF CONTENTS
Page | ||
Part I | ||
Part II | ||
Part III | ||
Part IV | ||
2
A Warning About Forward-Looking Statements
This Annual Report on Form 10-K contains forward-looking statements within the meaning of the federal securities laws that involve material risks, assumptions and uncertainties. Many possible events or factors could affect our future financial results and performance, such that our actual results and performance may differ materially from those that may be described or implied in the forward-looking statements. As such, no forward-looking statement can be guaranteed. Differences in actual results and performance may arise as a result of a number of factors including, without limitation, those discussed in this Annual Report on Form 10-K in Part I, Item 1A "Risk Factors." Forward-looking statements typically are identified by the use of terms such as “may,” “will,” “should,” “might,” “expect,” “anticipate,” “estimate,” “plan,” “intend,” “goal,” “project,” “strategy,” “future,” and similar words, although some forward-looking statements are expressed differently. Forward-looking statements in this Annual Report include, among others, statements concerning: our outlook for the upcoming fiscal year regarding revenue growth, gross margins and earnings, projected capital expenditures for the upcoming fiscal year; and our source of funds for such expenditures; the sufficiency of our liquidity and capital resources; our strategy, goals, initiatives and objectives; the anticipated beneficial attributes of products and platforms under development; anticipated new product and development results; that we expect to continue to depend on a few key distributors for sales of our products; expected growth and the sources of that growth; the impact of new accounting standards; that point-of-care testing is increasing; that clinical reference laboratories will continue to be a competitive threat; that we will continue to make substantial expenditures for sales and marketing, manufacturing and product research and development activities; that influenza and cardiology revenues will be a significant portion of our total revenue; industry consolidation and competition trends; competition for management and key personnel; that we may enter into additional foreign currency exchange risk sharing arrangements; the sufficiency of our facilities; the sufficiency of our insurance and our exposure to claims and litigation; our intention to not pay dividends; that we will continue to obtain licenses from third parties; and our intention to continue to evaluate technology and acquisition opportunities. The risks described under “Risk Factors” in Item 1A of this Annual Report and elsewhere herein and in reports and registration statements that we file with the Securities and Exchange Commission (the “SEC”) from time to time, should be carefully considered. You are cautioned not to place undue reliance on these forward-looking statements, which reflect management’s analysis only as of the date of this Annual Report. Except as required by law, we undertake no obligation to publicly release the results of any revision or update of these forward-looking statements, whether as a result of new information, future events or otherwise.
3
Part I
Item 1. Business
All references to “we,” “our,” and “us” in this Annual Report refer to Quidel Corporation and its subsidiaries.
Overview
We have a leadership position in the development, manufacturing and marketing of rapid diagnostic testing solutions. These diagnostic testing solutions are separated into our four product categories: rapid immunoassays, cardiac immunoassays, specialized diagnostic solutions and molecular diagnostic solutions. We sell our products directly to end users and distributors, in each case, for professional use in physician offices, hospitals, clinical laboratories, reference laboratories, leading universities, retail clinics, pharmacies and wellness screening centers. We market our products through a network of distributors, and through a direct sales force. The Company operates in one business segment that develops, manufactures and markets our four product categories.
We commenced our operations in 1979 and launched our first products, dipstick-based pregnancy tests, in 1983. Since such time, our product base and technology platforms have expanded through internal development and acquisitions of other products, technologies and companies, including the recent acquisition of our cardiovascular and toxicology businesses in 2017. Our diagnostic solutions aid in the detection and diagnosis of many critical diseases and other medical conditions, including infectious diseases, cardiovascular diseases and conditions, women’s health, gastrointestinal diseases, autoimmune diseases, bone health and thyroid diseases.
Corporate Information
We are a corporation, originally incorporated as Monoclonal Antibodies, Inc. in California in 1979 and re-incorporated as Quidel Corporation in the State of Delaware in 1987. Our executive offices are located at 12544 High Bluff Drive, Suite 200, San Diego, California 92130, and our telephone number is (858) 552-1100. This Annual Report and each of our other periodic and current reports, including any amendments thereto, are available, free of charge, on our website, www.quidel.com, as soon as reasonably practicable after such material is electronically filed with or furnished to the SEC. In addition, the SEC website contains reports, proxy and information statements, and other information about us at www.sec.gov. The information contained on our website or on the SEC website is not incorporated by reference into this Annual Report and should not be considered part of this Annual Report.
Recent Developments
On October 6, 2017, we closed our acquisition of the Triage® MeterPro® Cardiovascular (CV) and toxicology business ("Triage Business"), and B-type Naturietic Peptide (BNP) assay business run on Beckman Coulter analyzers ("BNP Business" and, together, the "Triage and BNP Businesses") from Alere Inc. ("Alere"). In connection with the acquisition of the Triage Business, the Company paid $399.8 million in cash and assumed certain liabilities. In connection with the acquisition of the BNP Business, the Company: (i) will pay (A) $16.0 million in cash plus up to an additional $24.0 million in contingent consideration, payable in five annual installments of up to $8.0 million, the first of which will be paid on April 30, 2018, (B) $240.0 million in cash, payable in six annual installments of $40.0 million each, the first of which will be paid on April 30, 2018 and (C) $0.2 million in cash for certain inventory related adjustments; and (ii) assumed certain liabilities. This strategic acquisition adds an extensive cardiovascular and toxicology point-of-care (POC) offering to our innovative medical diagnostics portfolio.
Business Strategy
Our primary objective is to increase shareholder value by building a broader-based diagnostic company capable of delivering revenue growth and consistent operating results. Our strategy is to identify potential market segments that provide, or are expected to provide, significant total market opportunities, and in which we can be successful by applying our significant expertise and know-how to develop differentiated technologies and products.
Our diagnostic testing solutions are designed to provide specialized results that serve a broad range of customers, by addressing the market requirements of ease of use, reduced cost, increased test accuracy and reduced time to result. Our current approach to address this diagnostic continuum relative to our strategy is to offer products in the following product categories:
• | rapid immunoassay tests for use in physician offices, hospital laboratories and emergency departments, retail clinics, eye health settings, pharmacies and other urgent care or alternative site settings; |
4
• | cardiac immunoassay tests for use in physician offices, hospital laboratories and emergency departments, and other urgent care or alternative site settings; |
• | specialized diagnostic solutions, including direct fluorescent assays (“DFA”) and culture-based tests for the clinical virology laboratory and other products serving the bone health, autoimmune and complement research communities; and |
• | molecular diagnostic tests across a number of hospitals, moderately complex physician offices, laboratories and other segments. |
Our current focus to accomplish our primary objective includes the following:
• | leveraging our current infrastructure to develop and launch new Rapid Immunoassays and Cardiac Immunoassays such as additional assays for our Sofia® and Sofia® 2 Analyzers and Triage® MeterPro® systems and next-generation immunoassay analyzers; |
• | developing a molecular diagnostics franchise that incorporates three distinct testing platforms, Solana®, AmpliVue®, and Savanna® and that leverages our molecular assay development competencies; and |
• | strengthening our position with distribution partners and our end-user customers to gain more emphasis on our products. |
Our current initiatives to execute this strategy include the following:
• | continue to provide products that can compete effectively in the healthcare market where cost and quality are important; |
• | continue to focus on integrating the Triage and BNP Businesses acquired from Alere in late 2017; |
• | strengthen our international infrastructure to support the integration of the Triage and BNP Businesses and enhance our global footprint to support our international operations and future growth; |
• | continue to focus our research and development efforts on three areas: |
• | new proprietary product platform development; |
• | the creation of improved products and new products for existing markets and unmet clinical needs; and |
• | pursuit of collaborations with, or acquisitions of, other companies for new and existing products and markets that advance our strategy to develop differentiated technologies and products; |
• | strengthen our market and brand leadership in current markets by acquiring and/or developing and introducing clinically superior diagnostic solutions; |
• | strengthen our direct sales force to enhance relationships with integrated delivery networks, laboratories and hospitals, with a goal of driving growth through improved physician and laboratorian satisfaction; |
• | leverage our wireless connectivity and data management systems, including cloud-based tools; |
• | support payer evaluation of diagnostic tests and establishment of favorable reimbursement rates; |
• | provide clinicians with validated studies that encompass the clinical efficacy and economic efficiency of our diagnostic tests for the professional market; |
• | continue to create strong global alliances to support our efforts to achieve leadership in key markets and expand our presence in emerging markets; and |
• | further refine our manufacturing efficiencies and productivity improvements to increase profit, with continued focus on innovative products and markets and our efforts to leverage our core competency in new product development. |
The Overall Market for In Vitro Diagnostics
Customers for In Vitro Diagnostics (“IVD”) products are primarily centralized laboratories and decentralized point-of-care settings.
Centralized testing market
The centralized in vitro diagnostic testing process typically involves obtaining a specimen of blood, urine or other sample from the patient and sending the sample from the healthcare provider’s office, hospital unit or clinic to a central laboratory. In a typical visit to the physician’s office, after the patient’s test specimen is collected, the patient is usually sent home and receives the results of the test several hours or days later. The result of this process is that the patient may leave the physician’s office without confirmation of the diagnosis and the opportunity to begin potentially more effective immediate care.
5
Decentralized POC market
POC testing for certain diseases has become an accepted adjunct to central laboratory and self-testing. The professional POC market is comprised of two general segments: decentralized testing in non-institutional settings, such as physicians’ offices and hospital testing (e.g., emergency rooms and bedside).
• | Out-of-hospital testing sites consist of physicians’ office laboratories, nursing homes, pharmacies, eye health offices, retail clinics and other non-institutional, ambulatory settings in which healthcare providers perform diagnostic tests. |
• | Hospital POC testing is accepted and growing and is generally an extension of the hospital’s central laboratory. Hospitals in the U.S. have progressively sought to reduce the length of patient stays and, consequently, the proportion of cases seen as outpatients have increased. If the U.S. experience is representative of future trends, emergency departments and other critical care units such as intensive care units, operating rooms, trauma and cardiac centers are increasingly becoming the principal centers for the management of moderate and severe acute illness. |
This decentralized POC market utilizes a large variety of IVD products ranging from moderate-sized instrumented diagnostic systems serving larger group practices to single-use, disposable tests. We believe POC testing is increasing due to its clinical benefit, fast results, cost-effectiveness and patient satisfaction.
We believe that the growth in POC testing is in part due to evolving technological improvements creating high quality tests with laboratory accuracy and POC ease-of-use, some of which are capable of being granted a waiver under the Clinical Laboratory Improvement Amendments of 1988 (“CLIA”).
Products
We provide diagnostic testing solutions under various brand names, including, among others, the following: Quidel®, QuickVue®, QuickVue+®, Sofia®, Triage®, AmpliVue®, Solana®, Virena®, MicroVueTM, Lyra®, FreshCellsTM, D3®, FastPoint®, ReadyCells®, Super E-MixTM, InflammaDry®, AdenoPlus®, ELVIRA®, ELVIS® and Thyretain®.
System Platforms:
Our diagnostic testing solutions are separated into our four product categories: rapid immunoassay, cardiac immunoassay, specialized diagnostic solutions and molecular diagnostic solutions. The key product categories and platforms are described below:
Rapid Immunoassay
Sofia and Sofia 2 Analyzers. Sofia is the brand name for our fluorescent immunoassay (“FIA”) systems. The easy-to-use Sofia and Sofia 2 Analyzers combine unique software, when used in conjunction with Sofia FIA tests, yield an automatic, objective result that is readily available on the instrument’s screen, in a hard-copy printout, and in a transmissible electronic form that can network via a lab information system to hospital and medical center databases. We originally launched the Sofia analyzer in 2011 and the next-generation Sofia 2 in 2017. These systems provide for different operational modes to accommodate both small and large laboratories as well as other features designed to facilitate use in a variety of healthcare settings, including hospitals, medical centers, and small clinics. Sofia 2 systems include additional benefits and features at a cost point that allows us to better address the lower-volume segment of the diagnostic testing market and incorporate enhanced optics designed to provide added performance benefits and kinetic reading designed to enable positive test results to be read in as short as a few minutes. The Sofia FIA tests that run on Sofia and Sofia 2 analyzers employ advanced lateral flow and immunofluorescence technologies to provide enhanced performance for several assays as noted in our medical and wellness categories discussion below.
QuickVue. QuickVue is the brand name for our rapid, visually-read, lateral flow immunoassay products. We have been a leader in the development and production of high quality lateral flow diagnostics since the early 1990s and offer a broad portfolio of products to diagnose a wide variety of infectious diseases and medical conditions.
InflammaDry and AdenoPlus. The InflammaDry and AdenoPlus products are rapid, lateral-flow based, POC products for the detection of infectious and inflammatory diseases and conditions of the eye. InflammaDry is a test that detects elevated levels of MMP-9, a key inflammatory marker for dry eye. AdenoPlus is a test that differentiates between a viral and bacterial infection of acute conjunctivitis (pink eye). Both products utilize innovative patented technology, are CE marked, FDA-cleared, CLIA-waived, and complement Quidel’s existing rapid diagnostic testing solutions.
6
Cardiac Immunoassay
Triage MeterPro. Triage MeterPro is our newly acquired portable testing platform, which incorporates a comprehensive menu of tests that enable physicians to promote improved health outcomes through the rapid diagnosis of critical diseases and health conditions, as well as the detection of certain drugs of abuse. This system aids in the diagnosis, assessment and risk stratification of patients having critical care issues, including congestive heart failure, acute coronary syndromes, acute myocardial infarction, or AMI, and can reduce hospital admissions and improve clinical and economic outcomes. Triage cardiovascular rapid tests include immunoassays for B-type Natriuretic Peptide (BNP), creatine kinase-MB (CK-MB), d-dimer, myoglobin, troponin I and N-terminal pro-Brain Natriuretic Peptide (NT-proBNP). Triage tests for troponin I and NT-proBNP, as well as certain test panels which include a combination of immunoassays, are not available for sale in the United States.
We also offer a version of the Triage BNP Test for use on Beckman Coulter lab analyzers.
In addition to the cardiovascular menu, the Triage TOX Drug Screen provides qualitative results for the determination of the presence of drug and/or the major metabolites in urine including assays for acetaminophen/paracetamol, amphetamines, methamphetamines, barbiturates, benzodiazepines, cocaine, methadone, opiates, phencyclidine, THC and tricyclic antidepressants.
Specialized Diagnostic Solutions
Virology. We provide a wide variety of traditional cell lines, specimen collection devices, media and controls for use in laboratories that culture and test for many human viruses, including, among others, respiratory and herpes family viruses. We provide cell-based products under the FreshCells brand in multiple different formats, including tubes, shell vials and multi-well plates. Our Virology product category includes the FDA cleared bioassay, Thyretain, which is used for the differential diagnosis of an autoimmune disease called Graves Disease.
Specialty Products. As a leader in the research space with our biomarkers for bone health, we produce both clinical and research products for the assessment of osteoporosis and the evaluation of bone resorption/formation, which, including our metabolic bone markers, are used to monitor the effectiveness of therapy in pharmaceutical and related research. In the area of autoimmune disease, we have developed Enzyme Linked Immunosorbent Assays (“ELISA”) and reagents for the detection of activation products from the three main complement pathways. Assays are developed on a microwell platform and are currently marketed to clinicians and researchers. We currently sell these products both directly and through select distributors throughout the world under the Quidel and MicroVue brands.
Molecular Diagnostic Solutions
AmpliVue. With our AmpliVue hand-held molecular diagnostic assay platform, the detection of the pathogen is achieved using a hand-held, fully contained cassette that combines isothermal Helicase Dependent Amplification (“HDA”) with lateral flow detection technology, and is currently used in several assays also noted in our medical and wellness categories discussion below.
Solana. The Solana system was developed as an extension to the AmpliVue product line, running the same proprietary HDA technology. Solana is an easy to run amplification and detection system that has the ability to concurrently run up to 12 assays at a time.
Lyra. Our open system molecular assays run on several thermocyclers currently on the market. We have several existing Lyra Molecular Real-Time Polymerase Chain Reaction (“PCR”) assays that provide important benefits to the customer, including, among others, room temperature storage, reduced process time, and ready-to-use reagent configurations. These include several assays as noted in our medical and wellness categories discussion below.
Savanna. We are developing the Savanna system as a low-cost, fully-integrated system with novel extraction, and sample in/result out simplicity. The system is expected to be able to run either PCR or HDA assays from multiple sample types.
Connectivity and Data Management
Virena. Virena is a wireless cellular data management and surveillance system that operates as a cloud-based solution connecting Quidel instruments across a healthcare system and automatically transmitting de-identified test results to a secure database. With Virena, a health system, physician office laboratory (“POL”), urgent care or retail clinic has the ability to compile, analyze, map and generate reports of de-identified test results improving operational efficiencies, quality and patient outcome initiatives.
7
Medical and Wellness Categories:
Our products address the following medical and wellness categories, among others:
Infectious Diseases
Influenza. We offer a variety of products designed to detect the viral antigens of influenza type A and B utilizing fluorescent immunoassay, lateral flow and molecular technologies. Our Sofia Influenza A+B test, used in conjunction with our Sofia and Sofia 2 analyzers, and our QuickVue influenza tests are rapid, qualitative tests for the detection of the viral antigens of influenza type A and B, the two most common types of the influenza virus. In addition, we offer molecular testing options with the recently launched Solana Influenza A+B assay utilizing HDA technology and our Lyra Influenza A+B real-time PCR assay.
Streptococci. We offer a variety of products designed to detect various Streptococcal disease states utilizing fluorescent immunoassay, lateral flow and molecular technologies. Our Sofia Strep A and Strep A+ fluorescent immunoassays, used in conjunction with our Sofia and Sofia 2 analyzers, and our QuickVue Strep A tests are intended for the rapid, qualitative detection of Group A Streptococcal antigen from throat swabs or confirmation of presumptive Group A Streptococcal colonies recovered from culture. Our Solana Strep Complete and Solana Group A Strep assays allow for the rapid, qualitative detection of Group A and pyogenic Group C/G Strep and Group A Strep, respectively, utilizing our molecular HDA technology. In addition, our Lyra Direct Strep Assay is a multiplex real-time PCR assay that detects and differentiates between pyogenic Group A and pyogenic C or G Streptococcal throat infections.
RSV (and hMPV). Our Sofia RSV test and our QuickVue RSV test are rapid immunoassay tests for Respiratory Syncytial Virus (“RSV”). In addition, Quidel offers molecular testing options with the recently launched Solana RSV + human metapneumovirus (hMPV) test and our combo Quidel Lyra RSV + human metapneumovirus (hMPV) test. The majority of upper respiratory tract infections in children are caused by viruses, and RSV is generally recognized as a frequent agent responsible for these infections and shares overlapping symptoms with hMPV.
Herpes and Herpes Family. We offer a variety of products designed to detect various herpes simplex virus (“HSV”) and herpes family viruses utilizing molecular and cell culture technologies. In the fall of 2016, we obtained FDA clearance of our Solana HSV-1+2/VZV Assay, used in conjunction with our Solana instrument, for the detection of HSV type 1, HSV type 2, and varicella-zoster virus (“VZV”). We also offer our Lyra Direct HSV 1+2/VZV and AmpliVue HSV 1+2 assays. In addition, our proprietary engineered cell culture system, ELVIS HSV, is an FDA cleared and highly sensitive system for the isolation and detection of HSV types 1 and 2. We also provide a multiplex cell culture solution using a propriety cell platform called H&V-MixTM that is used to isolate HSV, VZV and Cytomegalovirus, all in the herpes family of viruses. Antibody detection and identification of each of these viruses can be performed with FDA cleared antibody products provided under the D3 DFA brand. HSV is a widespread sexually transmitted infection. VZV is a DNA virus of the family Herpesviridae; infection results in chickenpox (varicella) and may lead to complications such as pneumonia and may reactivate later in life to produce shingles.
Multiplex Respiratory. Our cell culture and DFA detection solutions, including D3 FastPoint technology, are used by reference laboratories, public health labs and acute care hospitals to detect eight major viral respiratory pathogens. Our proprietary cell culture platform R-MixTM combined with our D3 Ultra DFA antibody kit, detects Influenza A and B, RSV, Adenovirus and Parainfluenza types 1, 2 and 3, with turn-around times between 16 and 48 hours. The same D3 Ultra DFA antibody kit can also be used for direct specimen testing for those viruses with turn-around times in less than 90 minutes. Our D3 FastPoint antibody kit detects eight viruses, with human metapneumovirus added to the testing menu, and provides laboratories, in a direct specimen testing format, the ability to produce virus identification in less than 25 minutes from specimen receipt.
Lyme. Our Sofia Lyme FIA, used in conjunction with our Sofia Analyzers, was FDA cleared in 2017. The assay is used to aid in the rapid differential detection of human IgM and IgG antibodies to Borrelia burgdorferi from serum and plasma specimens from patients suspected of B. burgdorferi infection and is intended for use to aid in the diagnosis of Lyme disease, a tickborne disease.
S. pneumoniae. Our Sofia S. Pneumoniae FIA, used in conjunction with our Sofia Analyzer, was CE Marked for sale in the European market in 2016. The assay is used to aid in the detection of both pneumococcal pneumonia and pneumococccal meningitis. Streptococcus pneumoniae is a leading cause of community-acquired pneumonia and bacterial meningitis.
Legionella. Our Sofia Legionella FIA, used in conjunction with our Sofia Analyzer, is CE Marked for sale in the European market. The assay is used to aid in the detection of Legionella pneumophila serogroup 1 antigen, which is the major causative agent of Legionnaires' disease, a disease primarily of pneumonia.
8
Bordetella Pertussis. Our AmpliVue Bordetella Assay is used in detection of Bordetella pertussis. Pertussis, or whooping cough, is a very contagious disease caused by the Bordetella pertussis bacteria and there has been increasing incidence in recent years.
Adenovirus and Parainfluenza. Quidel offers the Lyra Adenovirus Assay, a real-time PCR test for the qualitative detection of human adenovirus (HAdV) viral DNA, and our Lyra Parainfluenza Assay, a real-time PCR test for the qualitative detection and identification of Parainfluenza virus infections for types 1, 2 or 3 viral RNA.
Cardiology
The cardiology diagnostic market includes the markets for heart failure diagnostics, coronary artery disease risk assessment, coagulation testing and acute coronary syndrome. Our 2017 acquisitions of the Triage and BNP Businesses have positioned us to become a leader in this market. The Triage system consists of a portable fluorometer that interprets consumable test devices for cardiovascular conditions, as well as the detection of drugs of abuse. The Triage cardiovascular tests include the following:
Triage BNP Test. An immunoassay that measures B-type Natriuretic Peptide (BNP) in whole blood or plasma, used as an aid in the diagnosis and assessment of severity of heart failure. The test is also used for the risk stratification of patients with acute coronary syndromes and heart failure.
Triage Cardiac Panel. An immunoassay for the quantitative determination of CK-MB, myoglobin and troponin I in whole blood or plasma, as an aid in the diagnosis of acute myocardial infarction.
Triage Profiler S.O.B. An immunoassay for use as an aid in the diagnosis of myocardial infarction, the diagnosis and assessment of severity of congestive heart failure, the assessment and evaluation of patients suspected of having disseminated intravascular coagulation and thromboembolic events, including pulmonary embolism and deep vein thrombosis, and the risk stratification of patients with acute coronary syndromes.
Triage D-Dimer Test. An immunoassay for use as an aid in the assessment and evaluation of patients suspected of having disseminated intravascular coagulation or thromboembolic events, including pulmonary embolism and deep vein thrombosis.
Triage® NT-proBNP Test. A fluorescence immunoassay to be used with the Triage® MeterPro for the quantitative determination of N-terminal pro-Brain Natriuretic Peptide (NT-proBNP) in Ethylenediaminetetraacetic Acid (EDTA) anticoagulated whole blood and plasma specimens. The test is used as an aid in the diagnosis of individuals suspected of having congestive heart failure (also referred to as heart failure). The test is also used as an aid for the risk stratification of patients with heart failure and the risk stratification of patients with acute coronary syndromes (ACS).
Triage BNP Test for Beckman Analyzers. We also offer a version of the Triage BNP Test for use on Beckman Coulter lab analyzers.
Thyroid
Graves Disease. Our FDA cleared bioassay called Thyretain is used for the differential diagnosis of an autoimmune disease called Graves Disease. Graves Disease is caused by antibodies that stimulate the thyroid hormone receptors to create a hyperthyroid condition causing symptoms that include heart palpitations, unexplained weight loss, anxiety, depression and fatigue. Graves Disease is considered the most common autoimmune disorder in the U.S. according to an article published in the New England Journal of Medicine and it predominantly affects women. Thyretain is sold to reference laboratories and select acute care hospitals.
Autoimmune Thyroiditis. In 2017, we received the CE Mark for our Thyretain TBI Reporter BioAssay for the qualitative detection of blocking autoantibodies to the thyroid-stimulating hormone receptors (TSHR) in serum. The assay enables highly complex laboratories to diagnose autoimmune thyroiditis in just a few days, compared to traditional detection methods that could take months or even years.
Women’s and General Health
Pregnancy. Our Sofia hCG fluorescent immunoassay and our QuickVue pregnancy tests are used for the qualitative detection of hCG in serum or urine for the early detection of pregnancy. The early detection of pregnancy enables the physician and patient to institute proper care, helping to promote the health of both the mother and the developing embryo.
9
Chlamydia. Our QuickVue Chlamydia test is a lateral flow immunoassay for the rapid, qualitative detection of Chlamydia trachomatis from endocervical swab and cytology brush specimens. The test is intended for use as an aid in the presumptive diagnosis of Chlamydia. Chlamydia trachomatis is responsible for the most widespread sexually transmitted disease in the U.S. Over one-half of infected women do not have symptoms and, if left untreated, Chlamydia trachomatis can cause sterility.
Group B Streptococcus (GBS). In 2017, we obtained FDA clearance of our Solana GBS assay, used in conjunction with our Solana instrument, for the direct, qualitative detection of Group B Streptococcus from enriched broth cultures of specimens from antepartum women. GBS is commonly carried by pregnant women and can be transmitted to newborns at delivery, resulting in potential life-threatening illness. It is recommended that all pregnant women be tested for GBS during pregnancy.
Trichomonas. In 2016, we obtained FDA clearance of our Solana Trichomonas assay, used in conjunction with our Solana instrument, to aid in the diagnosis of trichomoniasis, a sexually transmitted disease attributable to infection from the Trichomonas vaginalis parasite. Trichomoniasis affects millions of people in the U.S., is more common in women and can be treated with antibiotics upon diagnosis.
Bone Health. Osteoporosis is a systemic skeletal disease characterized by low bone mass and deterioration of the microarchitecture of bone tissue, with a consequent increase in bone fragility and susceptibility to fractures. The risk for fracture increases exponentially with age. A key set of parameters in the monitoring of osteoporosis, both before and after therapy, are biochemical markers of bone metabolism. As a leader in the research space with our biomarkers for bone health, we produce both clinical and research products for the assessment of osteoporosis and the evaluation of bone resorption/formation, which, including our metabolic bone markers, are used by physicians to monitor the effectiveness of therapy in pharmaceutical and related research.
Eye Health
Our InflammaDry and AdenoPlus products are rapid, lateral-flow based, POC products for the detection of infectious and inflammatory diseases and conditions of the eye, which were acquired from RPS Diagnostics in 2017. InflammaDry is a test that detects elevated levels of MMP-9, a key inflammatory marker for dry eye. AdenoPlus is a test that differentiates between a viral and bacterial infection of acute conjunctivitis (pink eye).
Gastrointestinal Diseases
Clostridium difficile. In 2017, we received FDA clearance of our Solana C. difficile assay, used in conjunction with our Solana instrument, for the direct, qualitative detection of the Clostridium difficile DNA in unformed stool specimens of patients suspected of having Clostridium difficile-infection (CDI). In addition, we sell our Lyra Direct C. difficile Assay, a qualitative, multiplexed real-time PCR test for the detection of Clostridium difficile Toxin A or Toxin B genes approved for use on a variety of real-time PCR instruments and our AmpliVue C. difficile Assay, utilizing our HDA technology, for the detection of the Clostridium difficile Toxin A gene. Clostridium difficile can be a life threatening bacterial infection, especially for the elderly and patients on a prolonged antibiotic regimen.
Enterovirus. Enteroviruses reproduce initially in the gastrointestinal tract before spreading to other organs such as the nervous system, heart and skin. Enteroviruses can also infect the respiratory tract. Enteroviruses such as Coxsackievirus A16 are referred to as Hand Foot and Mouth disease and commonly affect infants and children. Our indirect fluorescent antibody (“IFA”) products sold under the name Super E-Mix and D3 IFA Enterovirus kit are used by reference laboratories and acute care hospitals.
Immunoassay fecal occult blood. Our QuickVue fecal immunochemical test (“FIT”) is a rapid test intended to detect the presence of blood in stool specimens. Blood in the stool is an indication of a number of gastrointestinal disorders, including colorectal cancer.
Helicobacter pylori (“H. pylori”). H. pylori is the bacterium associated with patients diagnosed with peptic ulcers. H. pylori is implicated in chronic gastritis and is recognized by the World Health Organization as a Class 1 carcinogen that may increase a person’s risk of developing stomach cancer. Our QuickVue rapid test is a serological test that measures antibodies circulating in the blood caused by the immune response to the H. pylori bacterium.
Toxicology
The toxicology testing market includes testing for substance use, misuse and abuse, including testing in connection with pain management and opioid cessation therapy. The ability to rapidly identify the impact of drug use on a patient’s clinical
10
presentation as well as securely monitor a patient’s therapy compliance is critical to the substance abuse testing market. Our Triage TOX Drug Screen provides qualitative results for the determination of the presence of drug and/or the major metabolites in urine including assays for acetaminophen/paracetamol, amphetamines, methamphetamines, barbiturates, benzodiazepines, cocaine, methadone, opiates, phencyclidine, THC and tricyclic antidepressants.
Seasonality
Sales of our influenza products are subject to, and significantly affected by, the seasonal demands of the cold and flu seasons, prevalent during the fall and winter. As a result of these seasonal demands, we typically experience lower sales volume in the second and third quarters of the calendar year, and typically have higher sales in the first and fourth quarters of the calendar year. Historically, sales of our influenza products have varied from year to year based in large part on the severity, length and timing of the onset of the cold and flu season. For the years ended December 31, 2017, 2016 and 2015, sales of our influenza products accounted for 39%, 37% and 43%, respectively, of total revenue. This percentage is expected to decrease as a result of the late 2017 acquisition of the Triage and BNP Businesses.
Research and Development
We continue to focus our research and development efforts on three areas:
• | new proprietary product platform development, |
• | the creation of improved products and new products for existing markets and unmet clinical needs, and |
• | pursuit of collaborations with, or acquisitions of, other companies for new and existing products and markets that advance our differentiated strategy. |
Research and development expenses were approximately $33.6 million, $38.7 million, and $35.5 million for the years ended December 31, 2017, 2016 and 2015, respectively. We anticipate that we will continue to devote a significant amount of financial resources to product and technology research and development for the foreseeable future.
Marketing and Distribution
Our business strategy is designed around serving the continuum of healthcare delivery needs globally, starting with POC clinicians located in doctor’s office practices to moderately complex POLs through the highly complex environment in hospital and clinical reference laboratories in North America and a variety of settings internationally.
Within the inherent operational diversity of these various segments, we focus on ensuring market leadership and providing points of differentiation by specializing in the diagnosis and monitoring of selected disease states and conditions. Our marketing strategy includes ensuring that our key product portfolios are supported by clinical validation and health economic and outcomes research that demonstrates to hospitals, laboratories, acute care facilities and POC clinicians that these tests deliver fast, high quality results, are cost-effective to use, and improve patient outcomes.
Our North America distribution strategy takes into account the fact that the POC market is highly fragmented, with many small or medium-sized customers. A network of national and regional distributors is utilized, combined with our own sales force, to reach customers using POC diagnostic tests.
We have expanded the size of our North America sales force in the past few years. As of December 31, 2017, we employed more than 100 sales representatives in North America. We are utilizing this expanded sales force to work closely with our key distributors to drive market penetration of our products in the POC market.
The sales, distribution and service of our cell culture and molecular diagnostic tests are controlled primarily by us. Laboratory end-users in hospitals and clinical reference laboratories utilizing these diagnostic tests are reached through our own direct sales force and technical support services that have specialized training and understanding of the product portfolio.
Internationally, we sell products in approximately 90 countries and market and distribute products in a variety of methods, including through a mix of direct and distribution strategies worldwide, and currently through temporary transition support services provided by Abbott Laboratories in connection with the Triage and BNP Businesses.
In Europe, we currently employ more than 60 employees, primarily to support sales and marketing activities in key countries, such as Germany and Italy and are continuing to recruit more sales personnel to support the Triage and BNP Businesses. In addition, we are building out a shared service center in Galway, Ireland to support general and administrative and technical support and customer service functions in Europe.
11
In Asia, we are focused on supporting the acquired Triage and BNP Businesses and continuing to grow our core immunoassay and cell culture businesses. We currently employ more than 30 employees in China, primarily to support sales and marketing efforts in China related to the acquired Triage and BNP Businesses. In addition, we are building out a shared service center in Shanghai, China to support general and administrative and technical support and customer service functions.
We derive a significant portion of our total revenue from a few distributors. Three of our distributors, which are considered to be among the market leaders, collectively accounted for approximately 54%, 44% and 48% of our total revenue for the years ended December 31, 2017, 2016 and 2015, respectively. As a result of the 2017 acquisition of the Triage and BNP Businesses, these percentages may change in the future. See Note 9 “Industry and Geographic Information” and Note 12 "Acquisition" in the Consolidated Financial Statements included in this Annual Report.
Manufacturing
We have three primary manufacturing sites. Two are in San Diego, California and one is located in Athens, Ohio.
Our McKellar Court, Lateral Flow manufacturing facility is located in San Diego, and consists of laboratories devoted to tissue culture, cell culture, protein purification and immunochemistry. Production areas are dedicated to manufacturing and assembly. In the manufacturing process, biological and chemical supplies and equipment are used. We have invested in a high degree of automated inspection, assembly and testing. Since the year 2000, this facility has operated under a Quality Management System certified to International Organization for Standardization (“ISO”) standards. The facility is certified to ISO 13485:2003 / EN 13485:2012 medical device standards as required for medical device manufacturers distributing product within the European Union and other countries. Many of the immunoassay products manufactured in this San Diego facility are packaged and shipped by a local third party.
Our Athens facility consists of a molecular manufacturing laboratory dedicated to the manufacture and assembly of our molecular products, clean rooms (FS-209E Class 1000: ISO Class 6) for the culturing and dispensing of cell cultures under GMP conditions and laboratories devoted to tissue culture for the production of monoclonal antibodies and the development and manufacture of research and MicroVue products. In the manufacturing process, biological and chemical supplies are used, as well as specialized equipment. The facility is certified to ISO 13485:2003 / EN ISO 13485:2012 medical device standards. Packaging and shipping logistics are handled at the facility.
Our Summers Ridge, San Diego facility consists of laboratories that are involved in mammalian cell culture, bacterial fermentation, protein purification and modification, as well as other techniques involved in immunoassay reagent manufacturing. These reagents are used in the manufacture of devices made at the site and are also supplied to a third party as key active ingredients for the Beckman Coulter Immunoassay Systems BNP product. In addition, this site has production areas dedicated to creating and processing plastic components that are subsequently transformed into finished devices (Cardiac and Drugs of Abuse ("DOA") products) using customized manufacturing equipment, including bespoke automation. This facility is certified to ISO 13485:2003 / EN ISO 13485:2012 medical device standards. Most of the products are packaged at this site and subsequently distributed by a third party.
We seek to conduct all of our manufacturing in compliance with the FDA Quality System Regulations (“QSR”) (formerly Good Manufacturing Practices) governing the manufacture of medical devices. Our manufacturing facilities have been registered with the FDA and the Department of Health Services of the State of California for our San Diego facilities (the “State FDA”), and have passed routine federal and state inspections confirming compliance with the QSR regulatory requirements.
Government Regulation
Regulation in the United States
The testing, manufacture and commercialization of our products are subject to regulation by numerous governmental authorities, principally the FDA and corresponding state and foreign regulatory agencies. Pursuant to the U.S. Federal Food, Drug, and Cosmetic Act and the regulations promulgated thereunder, the FDA regulates the preclinical and clinical testing, manufacture, labeling, distribution and promotion of medical devices. Noncompliance with applicable requirements can result in, among other matters, fines, injunctions, civil penalties, recall or seizure of products, total or partial suspension of production, failure of the FDA to grant premarket clearance or premarket approval for devices, withdrawal of marketing clearances or approvals and criminal prosecution. The FDA also has the authority to request a recall, repair, replacement or refund of the cost of any device manufactured or distributed in the U.S. if the device is deemed to be unsafe.
12
In the U.S., devices are classified into one of three classes (Class I, II or III) on the basis of the controls deemed necessary by the FDA to reasonably ensure their safety and effectiveness. Class I and II devices are subject to general controls including, but not limited to, performance standards, premarket notification (“510(k)”) and post market surveillance. Class III devices generally pose the highest risk to the patient and are typically subject to premarket approval to ensure their safety and effectiveness. Our current products are all Class I or II.
Prior to commercialization in the U.S. market, manufacturers of diagnostic assays like our products must obtain FDA clearance through a premarket notification or premarket approval process, which can be lengthy, expensive and uncertain. The FDA has been requiring more rigorous demonstration of product performance as part of the 510(k) process, including submission of extensive clinical data. It generally takes from three to six months to obtain clearance but may take longer. A premarket approval application must be supported by valid scientific evidence to demonstrate the safety and effectiveness of the device, typically including the results of clinical investigations, bench tests and reference laboratory studies. In addition, modifications or enhancements for existing products that could significantly affect safety or effectiveness, or constitute a major change in the intended use of the device, will require new submissions to the FDA.
The FDA's current guidance entitled “Guidance for Industry and FDA Staff Recommendation for CLIA waiver applications” sets forth requirements for obtaining a CLIA waiver that are onerous and have increased the time and cost required to obtain a CLIA waiver.
Any devices we manufacture or distribute pursuant to FDA clearance or approvals are subject to continuing regulation by the FDA and certain state agencies, including adherence to QSR relating to testing, control, documentation and other quality assurance requirements. We must also comply with Medical Device Reporting (“MDR”) requirements mandating reporting to the FDA of any incident in which a device may have caused or contributed to a death or serious injury, or in which a device malfunctioned and, if the malfunction were to recur, would be likely to cause or contribute to a death or serious injury. Labeling and promotional activities are also subject to scrutiny by the FDA and, in certain circumstances, by the Federal Trade Commission. Current FDA enforcement policy prohibits the marketing of approved medical devices for unapproved uses.
Regulation Outside of the United States
For marketing outside the U.S., we are subject to foreign regulatory requirements governing human clinical testing and marketing approval for our products. These requirements vary by jurisdiction, differ from those in the U.S., and may require us to perform additional or different preclinical or clinical testing regardless of whether we have obtained FDA approval. The amount of time required to obtain necessary approvals may be longer or shorter than that required for FDA approval. In many foreign countries, pricing and reimbursement approvals are also required.
Our initial focus for obtaining marketing approval outside the U.S. is typically the European Union (the “EU”), Japan, China and Canada. EU Regulations and Directives generally classify healthcare products either as medicinal products, medical devices or in vitro diagnostics. The CE Mark certification for the EU requires us to receive ISO certification for the manufacture of our products. This certification comes only after the development of an all-inclusive quality system, which is reviewed for compliance with ISO standards by a licensed body working within the EU. After certification is received, a technical file is developed which attests to the product’s compliance with EU directive 98/79/EC for in vitro diagnostic medical devices. Only after this point is the product CE marked. Japanese regulations require registration of in vitro diagnostic products with the Japanese Ministry of Health, Labor and Welfare. Chinese regulations require registration of diagnostic products with the China FDA, or CFDA. Additional clinical trials are typically required for registration purposes. For products marketed in Canada, registration is required with Health Canada and we have our independent party certification under the Canadian Medical Device Regulation.
Intellectual Property
The healthcare industry has traditionally placed considerable importance on obtaining and maintaining patent and trade secret protection for commercially relevant technologies, devices, products and processes. We and other companies engaged in research and development of new diagnostic products actively pursue patents for technologies that are considered novel and patentable. However, important factors, many of which are not within our control, can affect whether and to what extent patent protection in the U.S. and in other important markets worldwide is obtained. By way of example, the speed, accuracy and consistency in application of the law in a patent office within any particular jurisdiction is beyond our control and can be unpredictable. The resolution of issues such as these and their effect upon our long-term success is likewise indeterminable. We have issued patents, both in the U.S. and internationally, with expiration dates ranging from the present through approximately 2035 and have patent applications pending throughout the world.
13
It has been our policy to file for patent protection in the U.S. and other countries with significant markets, such as Western European countries and Japan, if the economics are deemed to justify such filing and our patent counsel advises that relevant patent protection may be obtained.
A large number of individuals and commercial enterprises seek patent protection for technologies, products and processes in fields in, or related to, our areas of product development. To the extent such efforts are successful, we may be required to obtain licenses and pay significant royalties in order to exploit certain of our product strategies. Licenses may not be available to us at all or, if so available, may not be available on acceptable terms.
We are aware of certain patents issued to various developers of diagnostic products with potential applicability to our diagnostic technology. We have licensed certain rights from certain companies to assist with the manufacturing of certain products. In the future, we expect that we will require or desire additional licenses from other parties in order to refine our products further and to allow us to develop, manufacture and market commercially viable or superior products effectively.
We seek to protect our trade secrets and technology by entering into confidentiality agreements with employees and third parties (such as potential licensees, customers, strategic partners and consultants). In addition, we have implemented certain security measures in our laboratories and offices. Also, to the extent that consultants or contracting parties apply technical or scientific information independently developed by them to our projects, disputes may arise as to the proprietary rights to such data.
Under many of our contractual agreements, we have agreed to indemnify the counterparty against costs and liabilities arising out of any patent infringement claims and other intellectual property claims asserted by a third party relating to products sold under those agreements.
Competition
Competition in the development and marketing of IVD products is intense, and innovation, product development, regulatory clearance to market and commercial introduction of new IVD technologies can occur rapidly. We believe that some of the most significant competitive factors in the rapid diagnostic market include convenience, speed to result, specimen flexibility, product menu, clinical needs, price, reimbursement levels and product performance as well as effective distribution, advertising, promotion and brand name recognition. The competitive factors in the central laboratory market are also significant and include price, product performance, reimbursement, compatibility with routine specimen procurement methods, and manufacturing products in testing formats that meet the workflow demands of larger volume laboratories. We believe our success will depend on our ability to remain abreast of technological advances, to develop, gain regulatory clearance and introduce technologically advanced products, to effectively market to customers a differentiated value proposition represented by our commercialized products, to maintain our brand strength and to attract and retain experienced personnel. The majority of diagnostic tests requested by physicians and other healthcare providers are performed by independent clinical reference laboratories. These laboratories, we expect, will continue to compete vigorously to maintain their dominance of the testing market. In order to achieve market acceptance for our products, we will be required to continue to demonstrate that our products provide physicians and central laboratories cost-effective and time-saving alternatives to other competitive products and technologies.
Many of our current and prospective commercial competitors, including several large pharmaceutical and diversified healthcare companies, have substantially greater financial, marketing and other resources than we have. These competitors include, among others, Abbott Laboratories, Beckman Coulter Primary Care Diagnostics, Fisher, Becton Dickinson and Company, Meridian Bioscience, Inc., and Danaher Corporation. We also face competition from our distributors since some have created, and others may decide to create, their own products to compete with ours. Competition may also be provided from large, medium and small development companies whose portfolio and technologies are dedicated to the development of diagnostic solutions in areas in which we currently have relevant market share.
Human Resources
As of December 31, 2017, we had 1,193 employees, none of whom are represented by a labor union. We have experienced no work stoppages and believe that our employee relations are good.
14
Executive Officers of Quidel Corporation
The names, ages and positions of all executive officers as of December 31, 2017 are listed below, followed by a brief account of their business experience. There are no family relationships among these officers, nor any arrangements or understandings between any officer and any other person pursuant to which an officer was selected.
Douglas C. Bryant, 60, was named President, Chief Executive Officer and a member of the Board of Directors in 2009. Prior to joining us, Mr. Bryant served as Executive Vice President and Chief Operating Officer at Luminex Corporation, managing its Bioscience Group, Luminex Molecular Diagnostics (Toronto), manufacturing, R&D, technical operations, and commercial operations. From 1983 to 2007, Mr. Bryant held various worldwide commercial operations positions with Abbott Laboratories including, among others: Vice President of Abbott Vascular for Asia/Japan, Vice President of Abbott Molecular Global Commercial Operations and Vice President of Abbott Diagnostics Global Commercial Operations. Earlier in his career with Abbott, Mr. Bryant was Vice President of Diagnostic Operations in Europe, the Middle East and Africa, and Vice President of Diagnostic Operations Asia Pacific. Mr. Bryant has over 30 years of industry experience in sales and marketing, product development, manufacturing and service and support in both the diagnostics and life sciences markets. Mr. Bryant holds a B.A. in Economics from the University of California at Davis.
Randall J. Steward, 63, became our Chief Financial Officer in October 2011. Prior to joining us, Mr. Steward served as the Chief Financial Officer for Navilyst Medical, Inc., a medical device company based in Massachusetts. From 2008 to January 2011, Mr. Steward served as Chief Operating Officer for SeQual Technologies, Inc., a San Diego-based medical device company, where he was responsible for all aspects of engineering, manufacturing, finance, and information systems. Prior to SeQual Technologies, Mr. Steward spent 11 years with Spectrum Brands as Executive Vice President and Chief Financial Officer. Mr. Steward holds a B.B.A. in Accounting from Southern Methodist University. He is also a Certified Public Accountant and a member of the American Institute of Certified Public Accountants.
Michael D. Abney, Jr., 54, became our Senior Vice President, Distribution in January 2015. Prior to joining us, he served as Vice President, Channel and Distribution for ConvaTec from 2013 to 2014 and held a number of positions at PSS World Medical, Inc. from 1989 to 2013, including most recently as Vice President, Supplier Management. Mr. Abney received his B.A. degree in Finance from the University of Florida in 1989.
Ratan S. Borkar, 44, became our Senior Vice President, International Commercial Operations in October 2017. He joined Quidel in May 2009 as Vice President, Business Development. In February 2012, Mr. Borkar moved into the role of VP, International Commercial Operations. Prior to Quidel, Mr. Borkar had a career in Investment Banking, where he most recently worked at J.P. Morgan as Vice President in the Healthcare Investment Banking Group, advising companies in the diagnostics, life sciences and clinical research areas. Mr. Borkar started his career at KPMG and is a Chartered Accountant. Mr. Borkar has an MBA from the University of Michigan’s Ross School of Business, where he was an Alan Gelband Scholar. Mr. Borkar received his Bachelor of Commerce from the University of Mumbai.
Robert J. Bujarski, J.D., 49, became our Senior Vice President, General Counsel and Corporate Secretary in June 2008 and in 2010 became our Senior Vice President, Business Development, General Counsel and Corporate Secretary. Mr. Bujarski previously served as our Senior Vice President, General Counsel and Corporate Secretary from March 2007 through March 2008. From July 2005 to March 2007, he was our General Counsel and Vice President. Mr. Bujarski was an associate attorney with the law firm of Gibson, Dunn & Crutcher LLP in its transactions practice group from October 2001 to July 2005. Mr. Bujarski received his B.A. degree in 1991 and his law degree in 2001 from the University of Arizona.
Werner Kroll, Ph.D., 61, became our Senior Vice President, R&D in May 2014. Prior to joining us, Dr. Kroll was Vice President and Global Head Research and Innovation for Novartis Molecular since 2009. Prior to holding that position he held a variety of senior positions from 2005 to 2009 at Novartis. Dr. Kroll has also held senior positions at Bayer from 1991 to 2005. Dr. Kroll received his Ph.D. and a Diploma in Chemistry from the University of Marburg.
Edward K. Russell, 50, became our Senior Vice President, Global Commercial Operations in October 2015 and subsequently became our Senior Vice President, North America Operations. Prior to joining the Company, Mr. Russell was employed by Thermo Fisher Scientific, a life sciences company based in Massachusetts, and its predecessor company Life Technologies for ten years. Mr. Russell served in various leadership roles from 2005 through 2015, including North America Commercial Leader of the BioSciences Division, General Manager of Life Technologies’ Global Services & Support Division, and President of Life Technologies Japan. Prior to joining Life Technologies in 2005, Mr. Russell held various leadership positions at FedEx Kinko’s, ExxonMobil and Toyota/Lexus. Mr. Russell started his career as an officer in the U.S. Coast Guard. Mr. Russell holds a B.S. in Civil Engineering from the U.S. Coast Guard Academy and an MBA from The Wharton School, University of Pennsylvania.
15
Item 1A. Risk Factors
Risks Related to Our Business
Our operating results are heavily dependent on sales of our influenza diagnostic tests and if sales or revenues of our influenza tests decline for any reason, our operating results would be materially and adversely affected on a disproportionate basis.
Although we continue to diversify our products, a significant percentage of our total revenues still continue to come from a limited number of our product families. In particular, revenues from the sale of our influenza tests represent a significant portion of our total revenues and are expected to remain so for at least the near future. In addition, the gross margins derived from sales of our influenza tests are significantly higher than the gross margins from many of our other core products. As a result, if sales or revenues of our influenza tests decline for any reason whether as a result of market share loss or price pressure, obsolescence, a mild flu season, regulatory matters or any other reason our operating results would be materially and adversely affected on a disproportionate basis. For the years ended December 31, 2017, 2016 and 2015, sales of our influenza products accounted for 39%, 37%, and 43% respectively, of total revenue.
Our operating results may fluctuate adversely as a result of many factors that are outside our control, which may negatively impact our stock price.
Fluctuations in customer demand, or operating or integration costs, for any reason, could cause our growth or operating results to fall below the expectations of investors and securities analysts.
We base the scope of our operations and related expenses on our estimates of future revenues. A significant portion of our operating expenses are fixed, and we may not be able to rapidly adjust our expenses if our revenues fall short of our expectations. Our revenue estimates for future periods are based, among other factors, on estimated end-user demand for our products. If end-user consumption is less than estimated, revenues from our distribution partners and other distribution channels would be expected to fall short of expectations, and because such a significant portion of our costs are fixed, could result in operating losses.
Factors that are beyond our control and that could affect our operating results in the future include:
• | timing of the onset, length and severity of the cold and flu seasons; |
• | seasonal fluctuations in our sales of influenza disease tests, which are generally highest in fall and winter, thus resulting in generally lower operating results in the second and third calendar quarters and higher operating results in the first and fourth calendar quarters; |
• | government and media attention focused on influenza and the related potential impact on humans from novel influenza viruses, such as H1N1 and avian flu; |
• | changes in the level of competition, such as would occur if one of our competitors introduced a new, better performing or lower priced product to compete with one or more of our products; |
• | changes in the reimbursement systems or reimbursement amounts that end-users may rely upon in choosing to use our products; |
• | changes in economic conditions in our domestic and international markets, such as economic downturns, decreased healthcare spending, reduced consumer demand, inflation and currency fluctuations and changes in government laws and regulations affecting our business; |
• | lower than anticipated market penetration of our new or more recently introduced products; |
• | significant quantities of our products or those of our competitors in our distributors’ inventories or distribution channels; |
• | changes in distributor buying patterns; and |
• | changes in the healthcare market, including consolidation in our customer base. |
To remain competitive, we must continue to develop, obtain and protect our proprietary technology rights; otherwise, we may lose market share or need to reduce prices as a result of competitors selling lower priced or technologically superior products that compete with our products.
Our ability to compete successfully in the diagnostic market depends on continued development and introduction of new proprietary technology and the improvement of existing technology. If we cannot continue to improve upon or develop, obtain and protect proprietary technology, we may lose market share or need to reduce prices as a result of competitors selling lower
16
priced or technologically superior products that compete with our products, and our operating results could be adversely affected.
Our competitive position is heavily dependent on obtaining and protecting our own proprietary technology or obtaining licenses from others. Our ability to obtain patents and licenses, and their benefits, is uncertain.
• | We have issued patents both in the U.S. and internationally, with expiration dates ranging from the present through approximately 2035. In addition to our patents in the U.S., we have patents issued in various other countries including, among others, Australia, Canada, Japan, various European countries, including France, Germany, Italy, Spain and the United Kingdom, and South Africa. Additionally, we have patent applications pending in the U.S. and various foreign jurisdictions. These pending patent applications may not result in the issuance of any patents, or if issued, may not have priority over others’ applications or may not offer meaningful protection against competitors with similar technology or may not otherwise provide commercial value. Moreover, any patents issued to us may be challenged, invalidated, found unenforceable or circumvented in the future. Third parties can make, use and sell products covered by our patents in any country in which we do not have patent protection. |
• | We also license the right to use our products to our customers under label licenses that are for research purposes only. These licenses could be contested and, because we cannot monitor all potential unauthorized uses of our proprietary technology around the world, we might not be aware of an unauthorized use or might not be able to enforce the license restrictions in a cost-effective manner. |
• | Our current and future licenses may not be adequate for the operation of our business. In the future, we expect that we will require or desire additional licenses from other parties in order to refine our products further and to allow us to develop, manufacture and market commercially viable or superior products. We may not be able to obtain licenses for technology patented by others and required to produce our products on commercially reasonable terms, if at all. |
To protect or enforce our patent rights, it may be necessary for us to initiate patent litigation proceedings against third parties, such as infringement suits or interference proceedings. These lawsuits would be expensive, take significant time and could divert management’s attention from other business concerns. In the event that we seek to enforce any of our patents against an infringing party, it is likely that the party defending the claim will seek to invalidate the patents we assert, which could put our patents at risk of being invalidated, held unenforceable, or interpreted narrowly, and our patent applications at risk of not being issued. Further, these lawsuits may provoke the defendants to assert claims against us. If we pursue any such claim, there will be no assurance that will prevail in any of such suits or proceedings or that the damages or other remedies awarded to us, if any, will be economically valuable.
In addition to our patents, we rely on confidentiality agreements and other similar arrangements with our employees and other persons who have access to our proprietary and confidential information, together with trade secrets and other common law rights, to protect our proprietary and confidential technology. These agreements and laws may not provide meaningful protection for our proprietary technology in the event of unauthorized use or disclosure of such information or in the event that our competitors independently develop technologies that are substantially equivalent or superior to ours. Moreover, the laws of some foreign jurisdictions may not protect intellectual property rights to the same extent as those in the U.S. In the event of unauthorized use or disclosure of such information, if we encounter difficulties or are otherwise unable to effectively protect our intellectual property rights domestically or in foreign jurisdictions, our business, operating results and financial condition could be materially and adversely affected.
In order to remain competitive and profitable, we must expend considerable resources to research new technologies and products and develop new markets, and there is no assurance our efforts to develop new technologies, products or markets will be successful or such technologies, products or markets will be commercially viable.
We devote a significant amount of financial and other resources to researching and developing new technologies, new products and new markets. The development, manufacture and sale of diagnostic products and new technologies require a significant investment of resources. The development of new markets also requires a substantial investment of resources, such as new employees, offices and manufacturing facilities. No assurances can be given that our efforts to develop new technologies or products will be successful, that such technologies and products will be commercially viable, or our expansion into new markets will be profitable.
We expect to incur significant operating expenses as a result of continued investment in sales and marketing activities, manufacturing scale-up and new product development associated with our efforts to accomplish our business strategies discussed in “Business - Business Strategy” in Part I of this Annual Report. No assurance can be given that we will be successful in implementing our operational, growth and other strategic efforts. In addition, the funds for our strategic
17
development projects have in the past come primarily from our business operations, borrowings under available lines of credit and the sale of equity or debt securities. If our business slows and we become less profitable, and as a result have less money available to fund research and development, we may have to reduce or eliminate programs. Similarly, if adequate financial, personnel, equipment or other resources are not available, we may be required to delay or scale back our strategic efforts.
Our operations will be adversely affected if our operating results do not correspondingly increase with our increased expenditures or if our technology, product and market development efforts are unsuccessful or delayed. Furthermore, our failure to successfully introduce new technologies or products and develop new markets could have a material adverse effect on our business and prospects.
We rely on a limited number of key distributors that account for a significant portion of our total revenue. The loss of any key distributor or an unsuccessful effort by us to directly distribute our products could lead to reduced sales.
Although we have many distributor relationships in the U.S., the market is dominated by a small number of these distributors. Three of our distributors, which are considered to be among the market leaders, collectively accounted (each individually in excess of 10%) for approximately 54%, 44%, and 48% of our total revenue for the years ended December 31, 2017, 2016 and 2015, respectively. In addition, we rely on a few key distributors for a majority of our international sales, and expect to continue to do so for the foreseeable future. The loss or termination of our relationship with any of these key distributors could significantly disrupt our business unless suitable alternatives are timely found or lost sales to one distributor are absorbed by another distributor. Finding a suitable alternative to a lost or terminated distributor may pose challenges in our industry’s competitive environment, and another suitable distributor may not be found on satisfactory terms, if at all. For instance, some distributors already have exclusive arrangements with our competitors, and others do not have the same level of penetration into our target markets as our existing distributors. If total revenue from these or any of our other significant distributors were to decrease in any material amount in the future or we are not successful in timely transitioning business to new distributors, our business, operating results and financial condition could be materially and adversely affected.
Intellectual property risks and third-party claims of infringement, misappropriation of proprietary rights or other claims against us could adversely affect our ability to market our products, require us to redesign our products or attempt to seek licenses from third parties, and materially adversely affect our operating results. In addition, the defense of such claims could result in significant costs and divert the attention of our management and other key employees.
Companies in or related to our industry often aggressively protect and pursue their intellectual property rights. In developing and producing new products and entering new markets, we may not be able to obtain, at reasonable cost or upon commercially reasonable terms, if at all, licenses to intellectual property of others that is alleged to be part of such new or existing products. From time to time, we have received, and may continue to receive, notices that claim we have infringed upon, misappropriated or misused other parties’ proprietary rights. Moreover, in the past we have been engaged in litigation with parties that claim, among other matters, that we infringed their patents.
We have hired and will continue to hire individuals or contractors who have experience in medical diagnostics and these individuals or contractors may have confidential trade secret or proprietary information of third parties. We cannot assure you that these individuals or contractors will not use this third-party information in connection with performing services for us or otherwise reveal this third-party information to us. Thus, we could be sued for misappropriation of proprietary information and trade secrets. Such claims are expensive to defend and could divert our attention and result in substantial damage awards and injunctions that could have a material adverse effect on our business, financial condition or results of operations. In addition, to the extent that individuals or contractors apply technical or scientific information independently developed by them to our projects, disputes may arise as to the proprietary rights to such data and may result in litigation.
The defense and prosecution of patent and trade secret claims are both costly and time consuming. We or our customers may be sued by other parties that claim that our products have infringed their patents or misappropriated their proprietary rights or that may seek to invalidate one or more of our patents. An adverse determination in any of these types of disputes could prevent us from manufacturing or selling some of our products, limit or restrict the type of work that employees involved with such products may perform for us, increase our costs and expose us to significant liability.
As a general matter, our involvement in litigation or in any claims to determine proprietary rights, as may arise from time to time, could materially and adversely affect our business, financial condition and results of operations for reasons such as:
• | it may of itself cause our distributors or end-users to reduce or terminate purchases of our products; |
• | it may consume a substantial portion of our managerial and financial resources; |
• | the outcome of such litigation would be uncertain and a court may find any third-party patent claims valid and infringed by our products (issuing a preliminary or permanent injunction) that would require us to procure costly |
18
licensing arrangements from third parties or withdraw or recall such products from the market, redesign such products offered for sale or under development or restrict employees from performing work in their areas of expertise;
• | governmental agencies may commence investigations or criminal proceedings against our employees, former employees and us relating to claims of misappropriation or misuse of another party’s proprietary rights; |
• | an adverse outcome could subject us to significant liability in the form of past royalty payments, penalties, special and punitive damages, the opposing party’s attorneys' fees, and future royalty payments significantly affecting our future earnings; and |
• | failure to obtain a necessary license (upon commercially reasonable terms, if at all) upon an adverse outcome could prevent us from selling our current products or other products we may develop. |
Even if licenses to intellectual property rights are available, they can be costly. We have entered into various licensing agreements, which largely require payments based on specified product sales as well as the achievement of specific milestones. Royalty and license expenses under these arrangements collectively totaled $0.6 million, $0.8 million and $1.1 million for the years ended December 31, 2017, 2016 and 2015, respectively.
In addition to the foregoing, we may also be required to indemnify certain customers, distributors and strategic partners under our agreements with such parties if a third party alleges or if a court finds that our products or activities have infringed upon, misappropriated or misused another person’s proprietary rights. Further, our products may contain technology provided to us by other parties such as contractors, suppliers or customers. We may have little or no ability to determine in advance whether such technology infringes the intellectual property rights of a third party. Our contractors, suppliers and licensors may not be required or financially able to indemnify us in the event that a claim of infringement is asserted against us, or they may be required to indemnify us only up to a maximum amount, above which we would be responsible for any further costs or damages.
We may need to raise additional funds to finance our future capital or operating needs, which could have adverse consequences on our operations and the interests of our stockholders.
Seasonal fluctuations in our operating results could limit the cash we have available for research and development and other operating needs. As a result, we may need to seek to raise funds through the issuance of public or private debt or the sale of equity to achieve our business strategy. In addition, we may need funds to complete acquisitions, or may issue equity in connection with acquisitions. If we raise funds or acquire other technologies or businesses through issuance of equity, this could dilute the interests of our stockholders. Moreover, the availability of additional capital, whether debt or equity from private capital sources (including banks) or the public capital markets, fluctuates as our financial condition and industry or market conditions in general change. There may be times when the private capital markets and the public debt or equity markets lack sufficient liquidity or when our securities cannot be sold at attractive prices, in which case we would not be able to access capital from these sources on favorable terms, if at all. We can give no assurance as to the terms or availability of additional capital.
Our results of operations and financial condition may be adversely affected by the financial soundness of our customers and suppliers.
If our customers’ or suppliers’ operating and financial performance deteriorates, or if they are unable to make scheduled payments or obtain credit, our customers may not be able to pay, or may delay payment of, accounts receivable owed to us, and our suppliers may restrict credit or impose different payment terms or reduce or terminate production of products they supply to us. Any inability of customers to pay us for our products and services, or any demands by suppliers for different payment terms, may adversely affect our operating results and financial condition. Additionally, both state and federal government sponsored and private payers, as a result of budget deficits or reductions, may seek to reduce their healthcare expenditures by renegotiating their contracts with us. Any reduction in payments by such government sponsored or private payers may adversely affect our earnings and cash flow.
We may not achieve market acceptance of our products among healthcare providers and physicians, and this would have a negative effect on future sales.
A large part of our business is based on the sale of rapid POC diagnostic tests. Our future sales depend on, among other matters, capture of sales from central laboratories by achieving market acceptance of POC testing from physicians and other healthcare providers. If we do not capture sales at the levels anticipated in our budget, our total revenue will not be at the levels that we expect and the costs we incur or have incurred will be disproportionate to our sales levels. We expect that clinical reference and hospital-based laboratories will continue to compete vigorously against our POC diagnostic products in order to
19
maintain and expand their existing dominance of the overall diagnostic testing market. Moreover, even if we can demonstrate that our products are more cost-effective, save time, or have better performance, physicians and other healthcare providers may resist changing to POC tests. Our failure to achieve market acceptance from physicians and healthcare providers with respect to the use of our POC diagnostic products would have a negative effect on our future sales.
The industry and market segment in which we operate are highly competitive, and intense competition with other providers of diagnostic products may reduce our sales and margins.
Our diagnostic tests compete with similar products made by our competitors. There are a large number of multinational and regional competitors making investments in competing technologies and products, including several large pharmaceutical and diversified healthcare companies. We also face competition from our distributors as some have created, and others may decide to create, their own products to compete with ours. A number of our competitors have competitive advantages, such as substantially greater financial, technical, research and other resources, and larger, more established marketing, sales, distribution and service organizations than we have. Moreover, some competitors offer broader product lines and have greater name recognition than we have. Our operating results could be materially and adversely affected if:
• | our competitors' products are more effective than ours or take market share from our products through more effective marketing or competitive pricing; |
• | our competitors obtain patent protection or other intellectual property rights that prevent us from offering competing products or services; or |
• | our competitors are able to obtain regulatory approvals for products or services or otherwise bring competing products to market earlier than us. |
In addition, there has been a trend toward industry consolidation in our markets over the last few years. We may not be able to compete successfully in an increasingly consolidated industry. We expect this trend toward industry consolidation to continue as companies attempt to strengthen or hold their market positions in an evolving industry and as companies are acquired or are unable to continue operations.
Our business and products are highly regulated by various governmental agencies. Our results of operations would be negatively affected by failures or delays in the receipt of regulatory approvals or clearances, the loss of previously received approvals or other changes to existing laws and regulations that adversely impact our ability to manufacture and market our products.
The testing, manufacture and sale of our products are subject to regulation by numerous governmental authorities in the U.S., principally the FDA and corresponding state and foreign regulatory agencies. The FDA regulates most of our products, which are currently all Class I or II devices. The U.S. Department of Agriculture regulates our veterinary products. Our future performance depends on, among other matters, if, when and at what cost we will receive regulatory approval or clearances for new products. Regulatory review can be a lengthy, expensive and uncertain process, making the timing and costs of clearances and approvals difficult to predict. Similarly, conducting clinical studies that may be required for regulatory approvals or clearances is a complex, time-consuming and expensive process, requiring months or years to complete, and our studies are not guaranteed to generate data that demonstrate safety and effectiveness or substantial equivalence of the evaluated product.
In addition, after we obtain necessary clearances or approvals to market our products, the FDA and other regulatory agencies may require post-market testing and additional surveillance to monitor the performance and use of approved products or may place conditions on any product approvals that could restrict the commercial applications of those products. Our results of operations would be negatively affected by failures or delays in the receipt of regulatory approvals or clearances, changes in laws and regulations, the loss of previously received approvals or clearances or the placement of limits on the manufacture, marketing and use of our products. For example, prior to our acquisition of the Triage products, the Summers Ridge, San Diego facility was subject to a 2012 FDA inspection that resulted in recalls of Triage products and revised release specifications for certain Triage meter-based products, which has not formally been closed-out with the FDA until after a future inspection. We cannot assure you that the government will find efforts to resolve the FDA warning letter to be satisfactory. We cannot predict whether other governments’ regulatory authorities will require additional remedial or corrective actions in the future, and the issues arising out of the FDA inspection may be expanded to cover other matters.
Furthermore, in the ordinary course of business, we must frequently make subjective judgments with respect to compliance with applicable laws and regulations. If regulators subsequently disagree with the manner in which we have sought to comply with these regulations, we could be subjected to substantial civil and criminal penalties, as well as field corrective actions, product recalls, seizures or injunctions with respect to the sale of our products. The assessment of any civil and criminal penalties against us could severely impair our reputation within the industry and affect our operating results, and any limitation on our ability to manufacture and market our products could also have a material adverse effect on our business.
20
Changes in government policy could adversely affect our business and profitability.
Changes in government policy could have a significant impact on our business by increasing the cost of doing business, affecting our ability to sell our products and negatively impacting our profitability. Such changes could include modifications to existing legislation, such as U.S. tax policy, or entirely new legislation, such as the Affordable Healthcare Act ("AHA") in the U.S. Although we cannot fully predict the many ways that healthcare reform might affect our business, the AHA imposed a 2.3% medical device excise tax ("MDET") on certain transactions, including many U.S. sales of medical devices, which includes the majority of our U.S. product sales. This tax took effect January 1, 2013. For the year ended December 31, 2015, we incurred $2.1 million related to the MDET, and although the MDET was suspended for 2016, 2017 and a two-year moratorium was recently implemented for 2018 and 2019, it may be reinstated. It is unclear whether and to what extent, if at all, other anticipated developments, including changes due to governmental administrative priorities, or changes resulting from healthcare reform, such as a change in the number of people with health insurance, may impact us.
We are subject to, and may in the future become subject to, claims and litigation that could result in significant expenses and could ultimately result in an unfavorable outcome for us.
From time to time, we are involved in litigation and other proceedings, including matters related to product liability claims, commercial disputes and intellectual property claims, as well as regulatory, employment, and other claims related to our business. Litigation can be lengthy, expensive and disruptive to our operations, and results cannot be predicted with certainty. An adverse decision could result in significant settlement amounts, monetary damages, fines or injunctive relief that could affect our financial condition or results of operations. Even if lawsuits do not result in an unfavorable outcome, the costs of defending such lawsuits may be material to our business and our operations. Moreover, these lawsuits may divert management’s attention from the operation of our business, which could adversely affect our business and results of operations.
For example, as further described in Part II, Item 8 of this Annual Report, Beckman Coulter, Inc. (“Beckman”) filed a lawsuit against us in November 2017, relating to a contractual arrangement with Beckman we acquired in October 2017 for the supply of antibodies and other inputs related to, and distribution of, the Triage® BNP Test for the Beckman Coulter Access Family of Immunoassay Systems and the outcome of such lawsuit may affect the value of the assets and liabilities we acquired and expose us to monetary liability. While we believe that the claims in the lawsuit are without merit, we can provide no assurance that we will be successful in defending the claims. If this lawsuit, or any other lawsuit filed against us, is resolved against us, we may be liable for significant damages and restraints on our business, which could adversely affect our results of operations and financial condition.
We are subject to numerous government regulations in addition to FDA regulation, and compliance with laws, including changed or new laws, could increase our costs and adversely affect our operations.
In addition to FDA and other regulations referred to above, numerous laws relating to such matters as safe working conditions, manufacturing practices, employment practices, data privacy, environmental protection, fire hazard control and disposal of hazardous or potentially hazardous substances impact our business or operations. If these laws or their interpretation change or new laws regulating any of our businesses or operations are adopted, the costs of compliance with these laws could substantially increase our overall costs. Failure to comply with any laws, including laws regulating the manufacture and marketing of our products, could result in substantial costs and loss of sales or customers. Because of the number and extent of the laws and regulations affecting our industry, and the number of governmental agencies whose actions could affect our operations, it is impossible to reliably predict the full nature and impact of future legislation or regulatory developments relating to our industry and our products. To the extent the costs and procedures associated with meeting new or changing requirements are substantial, our business, results of operations and financial condition could be adversely affected.
We use hazardous materials in our business that may result in unexpected and substantial claims against us relating to handling, storage or disposal.
Our research and development and manufacturing activities involve the controlled use of hazardous materials. Federal, state and local laws and regulations govern the use, manufacture, storage, handling and disposal of hazardous materials. These regulations include federal statutes commonly known as CERCLA, RCRA and the Clean Water Act. Compliance with these laws and regulations is already expensive. If any governmental authorities impose new environmental regulations requiring compliance in addition to that required by existing regulations, or alter their interpretation of the requirements of such existing regulations, such environmental regulations could impair our research, development or production efforts by imposing additional, and possibly substantial, costs, restrictions or compliance procedures on our business or operations. In addition, because of the nature of the penalties provided for in some of these environmental regulations, we could be required to pay sizable fines, penalties or damages in the event of noncompliance with environmental laws. Any environmental violation or remediation requirement could also partially or completely shut down our research and manufacturing facilities and operations, which would have a material adverse effect on our business. The risk of accidental contamination or injury from these
21
hazardous materials cannot be completely eliminated and exposure of individuals to these materials could result in substantial fines, penalties or damages that are not covered by insurance.
Our total revenue could be affected by third-party reimbursement policies and potential cost constraints.
The end-users of our products are primarily physicians and other healthcare providers. In the U.S., healthcare providers such as hospitals and physicians who purchase diagnostic products generally rely on third-party payers, principally private health insurance plans, federal Medicare and state Medicaid, to reimburse all or part of the cost of the procedure. Use of our products would be adversely impacted if physicians and other healthcare providers do not receive adequate reimbursement for the cost of our products by their patients’ third-party payers. Our total revenue could also be adversely affected by changes or trends in reimbursement policies of these governmental or private healthcare payers. We believe that the overall escalating cost of medical products and services has led to, and will continue to lead to, increased pressures on the healthcare industry, both foreign and domestic, to reduce the cost of products and services. Given the efforts to control and reduce healthcare costs in the U.S. in recent years, currently available levels of reimbursement may not continue to be available in the future for our existing products or products under development. Third-party reimbursement and coverage may not be available or adequate in either the U.S. or foreign markets, current reimbursement amounts may be decreased in the future and future legislation, regulation or reimbursement policies of third-party payers may reduce the demand for our products or adversely impact our ability to sell our products on a profitable basis.
Billing and payment for healthcare services are highly regulated, and the failure to comply with applicable laws and regulations can result in civil or criminal sanctions, including exclusion from federal and state healthcare programs.
A portion of our healthcare products and services are paid for by private and governmental third-party payers, such as Medicare and Medicaid. These third-party payers typically have different and complex billing and documentation requirements that we must satisfy in order to receive payment, and they carefully audit and monitor our compliance with these requirements. Such audits may lead to determinations that certain claims should not have been paid, and payors may seek to recoup or offset amounts they assert have been paid in error.
We must also comply with numerous other laws applicable to billing and payment for healthcare services, including privacy laws. Failure to comply with these requirements may result in non-payment, refunds, exclusion from government healthcare programs and civil or criminal liabilities, any of which may have a material adverse effect on our revenues, earnings
and cash flows. In addition, failure by third-party payers to properly process our payment claims in a timely manner could delay our receipt of payment for our products and services, which may have a material adverse effect on our cash flows.
Unexpected increases in, or inability to meet, demand for our products could require us to spend considerable resources to meet the demand or harm our reputation and customer relationships if we are unable to meet demand.
Our inability to meet customer demand for our products, whether as a result of manufacturing problems or supply shortfalls, could harm our customer relationships and impair our reputation within the industry. In addition, our product manufacturing of certain product lines is concentrated in one or more of our manufacturing sites. Weather, natural disasters (including pandemics), fires, terrorism, political change, failure to follow specific internal protocols and procedures, equipment malfunction, environmental factors or damage to one or more of our facilities could adversely affect our ability to manufacture our products. This, in turn, could have a material adverse effect on our business.
If we experience unexpected increases in the demand for our products or supply shortfalls, we may be required to expend additional capital resources to meet these demands. These capital resources could involve the cost of new machinery or even the cost of new manufacturing facilities. This would increase our capital costs, which could adversely affect our earnings and cash resources. If we are unable to develop or obtain necessary manufacturing capabilities in a timely manner, our total revenue could be adversely affected. Failure to cost-effectively increase production volumes, if required, or lower than anticipated yields or production problems, including those encountered as a result of changes that we may make in our manufacturing processes to meet increased demand or changes in applicable laws and regulations, could result in shipment delays as well as increased manufacturing costs, which could also have a material adverse effect on our business, reputation, operating results and financial condition.
Unexpected increases in demand for our products or supply shortfalls could also require us to obtain additional raw materials in order to manufacture products to meet the demand. Some raw materials require significant ordering lead time and we may not be able to timely access sufficient raw materials in the event of an unexpected increase in demand or supply shortfall, particularly those obtained from a sole supplier or a limited group of suppliers.
22
Interruptions in the supply of raw materials and other products and services could adversely affect our operations and financial results.
We depend on third-party manufacturers and suppliers for some of our products, or components and materials used in our products. Some of our raw materials, equipment and components are currently obtained from a sole supplier or a limited group of suppliers. We have long-term supply agreements with many of these suppliers, but these long-term agreements involve risks for us, such as our potential inability to obtain an adequate supply of quality raw materials, equipment or components and our reduced control over pricing, quality and timely delivery. It is also possible that one or more of these suppliers may become unwilling or unable to deliver materials or components to us. In addition, due to regulatory requirements relating to the qualification of suppliers, we may not be able to establish additional or replacement sources on a timely basis or without excessive cost. Any shortfall in our supply of raw materials, equipment or components, or our inability to quickly and cost-effectively obtain alternative sources for this supply, could have a material adverse effect on our business and operating results.
In addition, we use third party packaging companies to ship our products to customers. An interruption in the businesses of these third party packaging companies could result in a delay of shipments to customers.
If one or more of our products is claimed to be defective, we could be subject to claims of liability and harm to our reputation that could adversely affect our business.
Our business involves an inherent risk of product liability claims. Our product development and production processes are complex and could expose our products to claims of defectiveness. Alleged manufacturing and design defects could lead to recalls (either voluntary or required by the FDA or other government authorities) and could result in the removal of one or more of our products from the market. Similarly, a defect in one of our diagnostic products could lead to a false positive or false negative result, affecting the eventual diagnosis or treatment. Any such defects may require the payment of significant amounts in damages in connection with lawsuits. A defect or claim of a defect in the design or manufacture of our products could also have a material adverse effect on our reputation in the industry. Moreover, any product liability or other claim brought against us regardless of merit, could be costly to defend.
We are exposed to business risk which, if not covered by insurance, could have an adverse effect on our results of operations.
We face a number of business risks, including exposure to product liability claims. Although we maintain insurance for a number of these risks, we may face claims for types of damages, or for amounts of damages, that are not covered by our insurance. For example, although we currently carry product liability insurance for liability losses, there is a risk that product liability or other claims may exceed the amount of our insurance coverage or may be excluded from coverage under the terms of our policy. Also, our existing insurance may not be renewed at the same cost and level of coverage as currently in effect, or may not be renewed at all. Further, we do not currently have insurance against many environmental risks we confront in our business. If we are held liable for a claim against which we are not insured or for damages exceeding the limits of our insurance coverage, whether arising out of product liability matters, cybersecurity matters, or from some other matter, that claim could have a material adverse effect on our results of operations.
Failures in our information technology and storage systems could significantly disrupt our business or force us to expend excessive costs.
We utilize complex information technology systems to support our business and process, transmit, and store information, including sensitive personal information and proprietary or confidential information. We cannot be sure that our systems will meet our future business needs or that necessary upgrades will operate as designed, which could result in excessive costs or disruptions in portions of our business. In particular, any disruptions, delays or deficiencies caused by our enterprise resource planning system could adversely affect our ability to process orders, ship products, provide services and customer support, send invoices and track payments, fulfill contractual obligations or otherwise operate our business. Moreover, despite network security and back-up measures, some of our servers are potentially vulnerable to physical or electronic break-ins, computer viruses and similar disruptive problems. Despite the precautionary measures we have taken to prevent unanticipated problems that could affect our systems, sustained or repeated system failures that interrupt our ability to generate and maintain data, could result in a material disruption in our operations.
Our ability to protect our information systems and electronic transmissions of sensitive data from data corruption, cyber-based attacks, security breaches or privacy violations is critical to the success of our business.
Although we invest in security technology designed to protect our data against risks of data security breaches and cyber-attacks, and we have implemented solutions, processes and procedures to help mitigate these risks at various locations, such as encryption, virus protection, security firewalls and information security and privacy policies, our information technology and
23
infrastructure are subject to attacks by hackers and may be breached due to inadequacy or ineffectiveness of the protective measures undertaken, employee errors or omissions, malfeasance or other disruptions. A security breach or privacy violation that leads to disclosure of consumer information (including personally identifiable information or protected health information) could harm our reputation, compel us to comply with disparate state and foreign breach notification laws and otherwise subject us to liability under laws that protect personal data, resulting in increased costs or loss of revenue.
Despite our efforts to protect against cyber-attacks and security breaches, hackers and other cyber criminals are using increasingly sophisticated and constantly evolving techniques, and we may need to expend substantial additional resources to continue to protect against potential security breaches or to address problems caused by such attacks or any breach of our safeguards. In addition, a data security breach could distract management or other key personnel from performing their primary operational duties.
The interpretation and application of consumer and data protection laws in the United States, Europe and elsewhere are often uncertain, contradictory and in flux. Among other things, foreign privacy laws impose significant obligations on U.S. companies to protect the personal information of foreign citizens. It is possible that these laws may be interpreted and applied in a manner that is inconsistent with our data practices, which could have a material adverse effect on our business. Complying with these various laws could cause us to incur substantial costs or require us to change our business practices in a manner adverse to our business.
Our business could be negatively affected by the loss of or the inability to hire key personnel.
Our future success depends in part on our ability to retain our key technical, sales, marketing and executive personnel and our ability to identify and hire additional qualified personnel. Competition for these personnel is intense, both in the industry in which we operate and where our operations are located. Further, we expect to grow our operations, and our needs for additional management and other key personnel are expected to increase. If we are not able to retain existing key personnel, or timely identify and hire replacement or additional qualified personnel to meet expected growth, our business could be adversely impacted. In addition, the loss of any of our key personnel, particularly key research and development personnel, could harm our business and prospects and could impede the achievement of our research and development, operation or strategic objectives.
We face risks relating to our international sales, including inherent economic, political and regulatory risks, that could impact our financial performance, cause interruptions in our current business operations and impede our growth strategy.
Our products are sold internationally, with the majority of our international sales to our customers in Europe and Asia-Pacific. We currently sell and market our products through direct sales, distributor organizations and sales agents. Sales to foreign customers accounted for 18%, 17%, and 14% of our total revenue for the years ended December 31, 2017, 2016 and 2015, respectively. and are expected to increase as a percentage of our total revenue in 2018 and beyond as a result of our acquisition of the Triage and BNP Businesses in the fourth quarter of 2017. Our international operations are subject to inherent economic, political and regulatory risks, which could impact our financial performance, cause interruptions in our current business operations and impede our international growth. These foreign risks include, among others:
• | compliance with multiple different registration requirements and new and changing registration requirements, our inability to benefit from registration for our products inasmuch as registrations may be controlled by a distributor, and the difficulty in transitioning our product registrations; |
• | compliance with complex foreign and U.S. laws and regulations that apply to our international operations, including U.S. laws such as import/export limitations, the Foreign Corrupt Practices Act, and local laws prohibiting corrupt payments to governmental officials, could expose us or our employees to fines and criminal sanctions and damage our reputation; |
• | tariffs or other barriers as we continue to expand into new countries and geographic regions; |
• | exposure to currency exchange fluctuations against the U.S. dollar; |
• | longer payment cycles, generally lower average selling prices and greater difficulty in accounts receivable collection and enforcing agreements with foreign entities; |
• | reduced, or lack of, protection for, and enforcement of, intellectual property rights; |
• | social, political and economic instability in some of the regions where we currently sell our products or that we may expand into in the future; |
• | increased financial accounting and reporting burdens and complexities; |
• | complex and potentially adverse tax consequences; and |
24
• | diversion to the U.S. of our products sold into international markets at lower prices. |
Our international operations are governed by the U.S. Foreign Corrupt Practices Act and similar anti-corruption laws outside the U.S. Global enforcement of anti-corruption laws has increased substantially in recent years, with more enforcement proceedings by U.S. and foreign governmental agencies and the imposition of significant fines and penalties. While we have
implemented policies and procedures to comply with these laws, our international operations, which may involve customer relationships with foreign governments, create the risk that there may be unauthorized payments or offers of payments made by employees, consultants, sales agents or distributors. Any alleged or actual violations of these laws may subject us to government investigations, significant criminal or civil sanctions and other liabilities, and negatively affect our reputation.
Currently, the majority of our international sales are negotiated for and paid in U.S. dollars. Nonetheless, these sales are subject to currency risks, since changes in the values of foreign currencies relative to the value of the U.S. dollar can render our products comparatively more expensive. These exchange rate fluctuations could negatively impact international sales of our products, as could changes in the general economic conditions in those markets. In order to maintain a competitive price for our products internationally, we may have to continue to provide discounts or otherwise effectively reduce our prices, resulting in a lower margin on products sold internationally. Continued change in the values of the Euro, the Chinese Renminbi, the Japanese Yen and other foreign currencies could have a negative impact on our business, financial condition and results of operations.
In addition, we have certain supply agreements with foreign vendors whereby we share the foreign currency exchange fluctuation risk. We may, in the future, enter into similar arrangements.
Changes in our tax rates or exposure to additional income tax liabilities or assessments could affect our profitability.
We are subject to income taxes in the U.S. and in various non-U.S. jurisdictions. In addition, the amount of income taxes we pay is subject to ongoing audits by U.S. federal, state and local tax authorities and by non-U.S. tax authorities. Due to the potential for changes to tax laws (or changes to the interpretation thereof) and the ambiguity of tax laws, the subjectivity of factual interpretations, the complexity of our intercompany arrangements and other factors, our estimates of income tax assets or liabilities may differ from actual payments, assessments or receipts. If these audits result in payments or assessments different from our reserves, our future results may include unfavorable adjustments to our tax liabilities and our financial statements could be adversely affected. If we determine to repatriate earnings from foreign jurisdictions that have been considered permanently re-invested under existing accounting standards, it could also increase our effective tax rate. In addition, any significant change to the tax system in the United States or in other jurisdictions (including changes in the taxation of international income as further described below) could adversely affect our financial statements.
Risks Related to Our Acquisitions
Our acquisition of Alere’s Triage® and BNP Businesses presents certain risks to our business and operations
On October 6, 2017, we acquired the Triage and BNP Businesses from Alere. See Note 12 in this Annual Report for additional information concerning these acquisitions. The acquisition of these businesses, and the transition and integration process related thereto, present certain risks to our business and operations, including, among other things, risks that:
• | we may be unable to successfully transition and integrate the businesses, and we may experience business interruptions during transition and integration; |
• | we may not realize the anticipated benefits of the acquisitions, including those anticipated to arise from reducing costs, making product and process improvements and developing new products; |
• | we may lose management personnel and other key employees and be unable to attract and retain personnel and employees; |
• | management's attention and our other resources may be focused on integration activities instead of on day-to-day management activities, including pursuing other beneficial opportunities; |
• | we may incur substantial unexpected integration or transition related costs; |
• | the deferred consideration payable to Alere for the BNP Business will be payable even if BNP sales are significantly reduced, or even terminate, whether as a result of the introduction of a competing product, a determination that provisions of the contractual arrangement with Beckman are unenforceable or otherwise, and such payment obligations may significantly exceed the revenues from such business; |
• | we may not be able to successfully or efficiently manage our foreign expansion, and the acquired businesses will increase our exposure and risks related to foreign markets; |
• | we may be subject to claims, litigation, other legal proceedings and liabilities and damages in connection with the businesses and assets acquired in the acquisitions, some of which may not be covered in full, if at all, by the |
25
indemnification provisions provided for in the acquisition agreements, and even if indemnified, may be disruptive to our business;
• | we may not be able to receive required regulatory approvals or clearances relating to the acquired businesses and the acquired products, or may lose previously received regulatory approvals or clearances; |
• | in certain international markets, the marketing authorizations to sell the acquired products will continue to be held by Alere post-closing until the authorizations can be transferred to us through the applicable regulatory process, and such deferred transfers have additional risks, including: |
◦ | we may not timely receive such authorizations, if at all, or may encounter unexpected difficulties and costs in receiving the authorizations; |
◦ | we may have less control over the acquired businesses until the deferred transfers occur; |
◦ | completion of the acquisitions may trigger assignment or other provisions in certain commercial contracts to which Alere was a party, such that counterparties may potentially have the right to terminate the contracts; and |
◦ | launching branding or rebranding initiatives may involve substantial costs and may not be favorably received by customers. |
Alere may fail to perform under various transition agreements that were entered into as part of our acquisition of the Triage and BNP Businesses and we may fail to have necessary systems and services in place when certain of the transition services expire.
In connection with the acquisition of the Triage and BNP Businesses, we entered into a number of agreements with Alere, including transition services agreements and a manufacturing and supply agreement. Certain of these agreements will provide for the performance of services by each company for the benefit of the other for a period of time after the closing of the acquisitions of the Triage and BNP Businesses. If Alere is unable or unwilling to satisfy its payment or performance obligations under these agreements, we could incur operational difficulties or losses which could have a material adverse effect on our profitability and business. In addition, if the costs that we must pay for the services under these agreements significantly increase this could affect our profitability. Moreover, if we do not have our own systems and services in place, or if we do not have agreements in place with other providers of these services when the term of a particular transition service terminates (whether at the end of the term or as a result of an early termination), we may not be able to operate our business effectively, and the cost of such systems and services may be greater than what is provided under the transition services.
As we build our information technology infrastructure and transition the Triage and BNP Businesses to our own systems, we could incur substantial additional costs and experience temporary business interruptions.
We are installing and implementing information technology infrastructure to support critical business functions relating to the Triage and BNP Businesses, including accounting and reporting, customer service, inventory control and distribution, billing and receivables collection, as well as order entry, warehousing and other administrative services. Under the transition services agreements with Alere, Alere is required to provide services both inside and outside the United States, including back office services, for up to two years following the closings of the acquisitions of the Triage and BNP Businesses. The services provided include information technology, billing and receivables collection, as well as order entry, warehousing, and certain other administrative services. These transition services agreements allow us to operate the Triage and BNP Businesses prior to establishing our back office infrastructure and information technology systems to accommodate the Triage and BNP Businesses. Our failure to avoid operational interruptions as we implement the new systems and information technology could disrupt our integration of the Triage and BNP Businesses and have a material adverse effect on our profitability.
If goodwill or other intangible assets that we have recorded in connection with our acquisitions of other businesses become impaired, we may be required to take significant charges against earnings.
As a result of our acquisitions, we have recorded, and may continue to record, a significant amount of goodwill and other intangible assets. Under current accounting standards, we must assess, at least annually and potentially more frequently, whether the value of goodwill and other intangible assets has been impaired. Any reduction or impairment of the value of goodwill or other intangible assets will result in additional charges against earnings, which could materially reduce our reported results of operations in future periods.
If we are not able to manage our growth strategy or if we experience difficulties identifying or integrating companies or technologies we may acquire, our operating results may be adversely affected.
Our business strategy contemplates further growth, which we expect to result in expanding the scope of operating and financial systems and the geographical area of our operations, including further expansion outside the U.S., as new products
26
and technologies are developed and commercialized or new geographical markets are entered. Because we have a relatively small executive staff, acquisitions and other future growth may divert management’s attention from other aspects of our business, and place a strain on existing management and our operational, financial and management information systems. Furthermore, we may expand into markets in which we have less experience or incur higher costs. Some of our growth is expected to come from acquisitions of businesses and technologies. However, we cannot be certain that we will be able to successfully identify and acquire attractive targets. Other risks associated with acquiring other technologies or businesses, include: inability to obtain financing for acquisitions on satisfactory terms or at all; difficulties integrating the operations of companies or technologies that we acquire with our own operations; diversion of the attention of management and key personnel from our core business; adverse effects on our existing business relationships; potential loss of key employees of the acquired businesses; write-downs of goodwill, intangible assets or other assets associated with the acquisitions; and we may not realize our anticipated benefits and cost savings within our expected time frame, or at all, or may experience unexpected costs. We can give no assurance that we will be able to successfully identify, complete and integrate strategic acquisitions. Should we encounter difficulties in managing these tasks and risks, our growth strategy may suffer and our revenue and profitability could be adversely affected.
Risk Factors Related to our Indebtedness
Our substantial debt could materially adversely affect our financial condition and results of operations.
On October 6, 2017, we entered into a $270.0 million five-year senior secured credit facility (the “Senior Credit Facility”), consisting of a $245.0 million Term Loan, all of which was borrowed at the closing, and a $25.0 million Revolving Credit Facility, $10.0 million of which was borrowed at the closing. The Senior Credit Facility includes an accordion feature that allows the borrowings under the Senior Credit Facility to be increased by $50 million upon the satisfaction of certain conditions. See Note 3 to the Consolidated Financial Statements for a more detailed description of the Credit Agreement. We also have outstanding indebtedness under our Convertible Senior Notes described in Note 3 to the Consolidated Financial Statements, and may incur other indebtedness from time to time.
The degree to which we are leveraged could have important consequences to our business and operating results, including:
• | our ability to obtain additional financing in the future for working capital, capital expenditures, acquisitions and general corporate purposes may be impaired; |
• | a significant portion of our cash flow from operating activities must be dedicated to the payment of our debt, which reduces the funds available to us for our operations and may limit our ability to engage in acts that may be in our long-term best interests; |
• | some of our debt is and will continue to be at variable rates of interest, which may result in higher interest expense in the event of increases in market interest rates; |
• | our debt agreements contain, and any agreements to refinance our debt likely will contain, financial and other restrictive covenants, and our failure to comply with them may result in an event of default, which, if not cured or waived, could have a material adverse effect on us; |
• | our level of indebtedness will increase our vulnerability to, and reduce our flexibility to respond to, general economic downturns and adverse industry and business conditions; |
• | as our long-term debt ages, we may need to renegotiate or repay such debt or seek additional financing; |
• | to the extent the debt we incur requires collateral to secure such indebtedness, our assets could be at risk and our flexibility related to such assets could be limited; |
• | our debt service obligations could limit our flexibility in planning for, or reacting to, changes in our business and industry; and |
• | our level of indebtedness may place us at a competitive disadvantage relative to less leveraged competitors. |
We may not be able to generate sufficient cash flow to meet our debt service obligations, and any inability to repay our debt when due would have a material adverse effect on our business, financial condition and results of operations.
Our ability to generate sufficient cash flow from operating activities to make scheduled or other required payments on our debt obligations and maintain a desired level of capital expenditures depends on our future performance, which is subject to economic, financial, competitive and other factors, many of which are beyond our control. If we are unable to generate such cash flow, we may be required to adopt one or more alternatives, such as selling assets, restructuring debt or obtaining additional equity capital on terms that may be onerous or highly dilutive. Our ability to undertake these activities may also be restricted by the terms of our various debt instruments then in effect. In addition, our ability to refinance our indebtedness or
27
issue additional equity capital will depend on the capital markets and our financial condition at such time. We may not be able to engage in any of these activities or engage in these activities on desirable terms, which could result in a default on our debt obligations. Any such default on our debt obligations could materially adversely affect our business, financial condition and results of operations.
The Senior Credit Facility is secured by substantially all of our assets and those of our subsidiary guarantors.
Borrowings under the Senior Credit Facility are guaranteed by certain of our material domestic subsidiaries and are secured by liens on substantially all of our assets and those of the guarantor subsidiaries, other than real property and certain other types of excluded assets. If we default under our secured indebtedness, including our Senior Credit Facility, the holders of such debt could proceed against the collateral securing that indebtedness, which could materially adversely affect our business, financial condition and results of operations.
The Senior Credit Facility contains certain prepayment requirements that will limit our ability to use our cash flow from operations and the proceeds of certain assets sales or other monies for other corporate purposes.
Subject to certain limitations, we are required to prepay loans outstanding under the Senior Credit Facility out of excess cash flow and out of the net cash proceeds of property dispositions and certain other amounts received not in the ordinary course of business, such as certain insurance proceeds and condemnation awards. For example, we were required to use a substantial portion of the proceeds of the Summers Ridge Sale Leaseback to repay amounts under the Senior Credit Facility. These requirements to prepay the Senior Credit Facility with excess monies will limit our flexibility to utilize those monies for other purposes that may be in our best interests.
Borrowings under the Senior Credit Facility will be accelerated if certain conditions are not met prior to the maturity of the Convertible Senior Notes.
Although the regular maturity date for the Senior Credit Facility is October 6, 2022, the maturity will be accelerated and all borrowings under the Senior Credit Facility will become due and payable on the date that is 91 days prior to the maturity of the Convertible Senior Notes, if any of the Convertible Senior Notes remain outstanding on that date and certain liquidity and refinancing conditions are not satisfied. If the Senior Credit Facility is accelerated under this provision, we may not be able to refinance the facility on acceptable terms, and could be forced to sell assets or issue stock in order to obtain capital to repay the Senior Credit Facility. Any failure to repay the Senior Credit Facility when due could have a material adverse effect on our liquidity and financial position.
The agreements relating to our indebtedness contain terms that restrict our ability to operate our business, and as a result, may materially and adversely affect our results of operations.
Our Senior Credit Facility contains, and other of our debt agreements may include from time to time, a number of restrictive covenants that impose significant operating and financial restrictions on us and our subsidiaries. Such restrictive covenants significantly limit our ability to:
• | incur additional debt, including guarantees; |
• | allow other liens on our property; |
• | make certain investments and acquisitions; |
• | sell or otherwise dispose of assets; |
• | engage in mergers or consolidations or allow a change in control to occur; |
• | make distributions to our stockholders; |
• | engage in restructuring activities; |
• | enter into transaction with affiliates; |
• | prepay or amend other indebtedness; |
• | engage in certain sale and leaseback transactions; and |
• | issue or repurchase stock or other securities. |
Such agreements also require us to satisfy other requirements, including maintaining certain financial ratios. Our ability to meet these requirements can be affected by events beyond our control and we may be unable to meet them. To the extent we fail to meet any such requirements and are in default under our debt obligations, our financial condition may be materially adversely affected. These restrictions may also limit our ability to engage in activities that could otherwise benefit us. To the extent that we are unable to engage in activities that support the growth, profitability and competitiveness of our business, our results of operations may be materially adversely affected.
28
An event of default under any agreement relating to our outstanding indebtedness or other event that could require outstanding debt to be prepaid or purchased by us could cross default other indebtedness, which could have a material adverse effect on our business, financial condition and results of operations.
If there were an event of default under any of the agreements relating to our outstanding indebtedness, the holders of the defaulted debt could cause all amounts outstanding with respect to that debt to be due and payable immediately, which default or acceleration of debt could cross default other indebtedness. Our Senior Credit Facility could also be cross defaulted as a result of other events that could require us to prepay or purchase outstanding indebtedness. Any cross default with respect to any of our indebtedness would put immediate pressure on our liquidity and financial condition and would amplify the risks described above with regards to being unable to repay our indebtedness when due and payable. We cannot assure you that our assets or cash flow would be sufficient to fully repay borrowings under our outstanding debt instruments if accelerated upon an event of default, and, as described above, any inability to repay our debt when due could have a material adverse effect on our business, financial condition and results of operations.
We may not have the ability to raise the funds necessary to settle conversions of our Convertible Senior Notes, purchase the Convertible Senior Notes as required upon a fundamental change or service or repay the Convertible Notes at maturity, and potential future debt may contain limitations on our ability to pay cash upon conversion or purchase of our Convertible Senior Notes.
Following a fundamental change (as defined in the indenture to our Convertible Senior Notes), the holders of our Convertible Senior Notes will have the right to require us to purchase their notes for cash. In addition, upon conversion of the Convertible Senior Notes, unless we settle our conversion obligation solely in shares of our common stock (other than cash in lieu of any fractional share), we will be required to make cash payments in respect of the Convertible Senior Notes being surrendered for conversion. We may not have sufficient financial resources, or be able to arrange financing, to pay the fundamental change purchase price in cash with respect to any Convertible Senior Notes surrendered by holders for purchase upon a fundamental change or make cash payments upon conversions. Our failure to purchase the Convertible Senior Notes upon a fundamental change or make cash payments upon conversions thereof when required would result in an event of default with respect to the Convertible Senior Notes which could, in turn, constitute a default under the terms of our other then-existing indebtedness. If the repayment of the related indebtedness were to be accelerated after any applicable notice or grace periods, we may not have sufficient funds to repay the indebtedness and purchase the Convertible Senior Notes or make cash payments upon conversions thereof.
The conditional conversion feature of our Convertible Senior Notes, if triggered, may adversely affect our financial condition and operating results.
In the event the conditional conversion feature of the Convertible Senior Notes is triggered, holders of Convertible Senior Notes will be entitled to convert their Convertible Senior Notes at any time during specified periods at their option. If one or more Convertible Senior Note holders elects to convert their notes, unless we satisfy our conversion obligation by delivering solely shares of our common stock, we would be required to settle all or a portion of our conversion obligation through the payment of cash, which could adversely affect our liquidity. Furthermore, even if Convertible Senior Note holders did not elect to convert their notes, we could be required under applicable accounting rules to reclassify all or a portion of the outstanding principal of the Convertible Senior Notes as a current rather than long-term liability, which would result in a material reduction of our net working capital.
We will continue to have the ability to incur debt and our levels of debt may affect our operations and our ability to pay the principal of and interest on our debt.
We and our subsidiaries may be able to incur substantial additional debt in the future. Our indebtedness could be costly or have adverse consequences, such as:
• | requiring us to dedicate a substantial portion of our cash flows from operations to payments on our debt; |
• | limiting our ability to obtain future financing for working capital, capital expenditures, acquisitions, debt obligations and other general corporate requirements; |
• | making us more vulnerable to adverse conditions in the general economy or our industry and to fluctuations in our operating results, including affecting our ability to comply with and maintain any financial tests and ratios required under our indebtedness; |
• | limiting our flexibility to engage in certain transactions or to plan for, or react to, changes in our business and the diagnostics industry; |
• | putting us at a disadvantage compared to competitors that have less relative and/or less restrictive debt; and |
• | subjecting us to additional restrictive financial and other covenants. |
29
If we incur substantial additional indebtedness in the future, these higher levels of indebtedness may affect our ability to pay the principal of and interest on existing indebtedness and our creditworthiness generally. In addition, our ability to make scheduled payments of the principal of, to pay interest on, or to refinance our indebtedness, depends on our future performance, which is subject to economic, financial, competitive and other factors beyond our control. Our business may not continue to generate cash flow from operations in the future sufficient to service or repay our debt. If we are unable to generate such cash flow, we may be required to adopt one or more alternatives, such as reducing capital expenditures, selling assets, restructuring debt or obtaining additional equity capital on terms that may be onerous or highly dilutive. Our ability to refinance our indebtedness will depend on the capital markets and our financial condition at such time. We may not be able to engage in any of these activities or engage in these activities on desirable terms, which could result in a default on our debt obligations.
If interest rates increase, our debt service obligations under our variable rate indebtedness could increase significantly, which could have a material adverse effect on our results of operations.
Borrowings under certain of our facilities from time to time, including under our Senior Credit Facility, are at variable rates of interest and as a result expose us to interest rate risk. If interest rates were to increase, our debt service obligations on the variable rate indebtedness would increase even though the amount borrowed remained the same, and our net income and cash flows will correspondingly decrease. To the extent the risk materializes and is not fully mitigated, the resulting increase in interest expense could have a material adverse effect on our results of operations.
Risks Related to our Common Stock
Sales of our common stock in the public market could lower the market price for our common stock and adversely impact the trading price of our securities.
We may need to seek additional capital. If this additional financing is obtained through the issuance of equity securities, debt convertible into equity or options or warrants to acquire equity securities, our existing stockholders could experience significant dilution upon the issuance, conversion or exercise of such securities. In addition, a substantial number of shares of our common stock is reserved for issuance upon the conversion of our Convertible Senior Notes, exercise of stock options and vesting of other equity awards.
In addition, we may issue shares of our common stock or securities convertible into our common stock from time to time in connection with a debt refinancing or replacement, acquisition, or other transaction. The issuance of additional shares of our common stock, or issuances of additional securities convertible into or exercisable for shares of our common stock or other equity linked securities, including, convertible debt, preferred stock or warrants, could dilute the ownership interest of our common stockholders and could depress the market price of shares of our common stock and impair our ability to raise capital through the sale of additional equity securities. We cannot predict the size of future issuances or the effect, if any, that they may have on the market price for our common stock.
We also have a number of institutional stockholders that own significant blocks of our common stock. If one or more of these stockholders were to sell large portions of their holdings in a relatively short time, for liquidity or other reasons, the prevailing market price of shares of our common stock could be negatively affected.
The price of our stock may fluctuate unpredictably in response to factors unrelated to our operating performance.
The stock market periodically experiences significant price and volume fluctuations that are unrelated to the operating performance of particular companies. These broad market fluctuations may cause the market price of our common stock to drop. In particular, the market price of securities of smaller medical device companies, like ours, has been very unpredictable and may vary in response to:
• | announcements by us or our competitors concerning technological innovations; |
• | introductions of new products; |
• | FDA and foreign regulatory actions; |
• | developments or disputes relating to patents or proprietary rights; |
• | failure to meet the expectations of stock market analysts and investors; |
• | changes in stock market analyst recommendations regarding our common stock; |
• | changes in healthcare policy in the U.S. or other countries; and |
• | general stock market conditions and other factors unrelated to our operating performance. |
30
Some provisions of our charter documents, Delaware law, and our Convertible Senior Notes may make takeover attempts difficult, which could depress the price of our stock and inhibit our stockholders' ability to receive a premium price for their shares.
Provisions of our amended and restated certificate of incorporation could make it more difficult for a third party to acquire control of our business, even if such change in control would be beneficial to our stockholders. Our amended and restated certificate of incorporation allows our board of directors to issue up to five million shares of preferred stock and to fix the rights and preferences of such shares without stockholder approval. Any such issuance could make it more difficult for a third party to acquire our business and may adversely affect the rights of our stockholders. Our amended and restated bylaws include advance notice requirements for stockholder proposals that require stockholders to give written notice of any proposal or director nomination to us within a specified period of time prior to any stockholder meeting and do not permit stockholders to call a special meeting of the stockholders, unless such stockholders hold not less than 50% of our stock entitled to vote at the meeting.
We are also subject to anti-takeover provisions under Delaware law. Together these provisions may delay, deter or prevent a change in control of us, adversely affecting the market price of our common stock.
In addition, the terms of our Convertible Senior Notes require us to offer to purchase the notes for cash in the event of a fundamental change. A non-stock takeover of our company may trigger the requirement that we purchase the Convertible Senior Notes. This feature may have the effect of delaying or preventing a takeover of us that would otherwise be beneficial to investors.
We do not anticipate declaring any cash dividends on our common stock.
We have never declared or paid cash dividends on our common stock and do not plan to pay any cash dividends in the near future. Our current policy is to retain all funds and any earnings for use in the operation and expansion of our business.
Item 1B. Unresolved Staff Comments
None.
Item 2. Properties
At December 31, 2017, we occupied the indicated square footage in the leased and owned facilities described below:
Location | Status | Lease term | Square Footage | Primary Use | ||||
San Diego, CA (McKellar) | Leased | 2020 - options to extend for three additional 5 year periods | 78,000 | Administrative offices, research and development and manufacturing | ||||
San Diego, CA (High Bluff) | Leased | 2022 - options to extend for two additional 5 year periods | 30,000 | Administrative offices, sales and marketing (principal executive offices) | ||||
San Diego, CA (Summers Ridge) | Owned | (1) | N/A | 320,000 | Administrative offices, research and development and manufacturing | |||
Athens, OH | Leased | 2022 - options to extend for one additional 5 year period | 94,000 | Administrative offices, sales and marketing, research and development and manufacturing | ||||
Beverly, MA | Leased | 2020 - options to extend for two additional 5 year periods | 9,700 | Administrative offices, research and development and manufacturing | ||||
(1) | As of December 31, 2017, we owned the four buildings at Summers Ridge and leased two buildings of approximately 150,000 square feet to Abbott. On January 5, 2018, we entered into a sale and leaseback transaction pursuant to which we sold the Summers Ridge property and leased back the other two buildings for an initial term of 15 years. The initial term can be extended at our option for two additional five-year terms upon satisfaction of certain conditions. |
We believe that our facilities are adequate for our current needs, and we currently do not anticipate any material difficulty in renewing any of our leases as they expire or securing additional or replacement facilities, in each case, on commercially reasonable terms. However, in anticipation of our growth strategy, we may pursue alternative facilities.
31
Item 3. Legal Proceedings
The information set forth in "Litigation and Other Legal Proceedings" in Note 8 in the Consolidated Financial Statements in Part II, Item 8 of this Annual Report is incorporated herein by reference.
Item 4. Mine Safety Disclosures
Not applicable.
32
Part II
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
COMMON STOCK PRICE RANGE
Our common stock is traded on the Nasdaq Global Market under the symbol “QDEL.” The following table sets forth the range of high and low sales prices for our common stock for the periods indicated.
Quarter Ended | Low | High | |||||
December 29, 2017 | $ | 43.28 | $ | 44.49 | |||
September 29, 2017 | $ | 42.84 | $ | 44.50 | |||
June 30, 2017 | $ | 27.08 | $ | 27.50 | |||
March 31, 2017 | $ | 22.55 | $ | 22.76 | |||
December 31, 2016 | $ | 20.88 | $ | 21.44 | |||
September 30, 2016 | $ | 20.96 | $ | 22.20 | |||
June 30, 2016 | $ | 17.20 | $ | 18.00 | |||
March 31, 2016 | $ | 16.98 | $ | 17.48 | |||
As of February 21, 2018, we had approximately 369 common stockholders of record. No cash dividends were declared for our common stock during the fiscal years ended in 2017 or 2016, and we do not anticipate paying any dividends in the foreseeable future.
Stock Repurchases
The Company did not make any stock repurchases under its share repurchase program during the year ended December 31, 2017, and there was $35.0 million available under the Company's share repurchase program. During the year ended December 31, 2017, 25,079 shares of outstanding common stock with a value of $0.5 million were repurchased in connection with payment of minimum tax withholding obligations for certain employees relating to the lapse of restrictions on certain restricted stock units ("RSUs"). These shares are not considered repurchases under the Company’s repurchase program. During the three months ended December 31, 2017, we repurchased no shares of common stock from employees in connection with payment of minimum tax withholding obligations related to the lapse of restrictions on certain RSUs.
On January 25, 2016, we announced that the Board of Directors authorized an amendment to the Company's previously announced stock repurchase program to (i) replenish the amount available for repurchase under the program back to the previously authorized repurchase amount of $50.0 million ($35.0 million of which remained available as of December 31, 2017), (ii) approve the addition of repurchases of the Company's Convertible Senior Notes under the program and (iii) extend the expiration date of the program to January 25, 2018. The stock repurchase program expired on January 25, 2018.
33
STOCKHOLDER RETURN PERFORMANCE GRAPH
Set forth below is a line graph comparing the yearly percentage change in the cumulative total stockholder return on our common stock with the cumulative total return of the Nasdaq Composite Index, Nasdaq Health Care Index, and Nasdaq US Benchmark Medical Supplies Index for the period beginning December 31, 2012 and ending December 31, 2017. The graph assumes an initial investment of $100 on December 31, 2012 in our common stock, the Nasdaq Composite Index, the Nasdaq US Benchmark Medical Supplies Index, the Nasdaq Health Care Index and reinvestment of dividends. The stock price performance of our common stock depicted in the graph represents past performance only and is not necessarily indicative of future performance.
COMPARISON OF 5 YEAR TOTAL CUMULATIVE RETURN
Among Quidel Corporation, the NASDAQ Composite, NASDAQ US Benchmark Medical Supplies and NASDAQ Health Care Indices
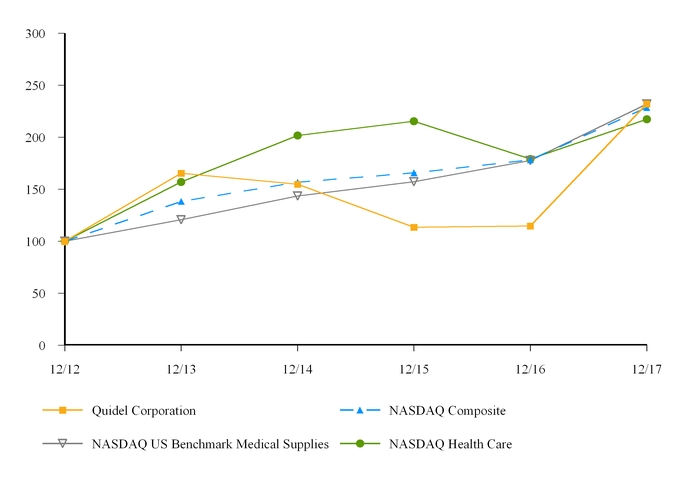
Base Period | |||||||||||||||||||||||
Company/Index | 12/31/2012 | 12/31/2013 | 12/31/2014 | 12/31/2015 | 12/31/2016 | 12/31/2017 | |||||||||||||||||
Quidel Corporation | $ | 100.00 | $ | 165.45 | $ | 154.90 | $ | 113.55 | $ | 114.73 | $ | 232.19 | |||||||||||
NASDAQ Composite | $ | 100.00 | $ | 138.32 | $ | 156.85 | $ | 165.84 | $ | 178.28 | $ | 228.63 | |||||||||||
NASDAQ US Benchmark Medical Supplies | $ | 100.00 | $ | 120.86 | $ | 143.48 | $ | 157.22 | $ | 177.71 | $ | 231.99 | |||||||||||
NASDAQ Health Care | $ | 100.00 | $ | 157.04 | $ | 201.75 | $ | 215.58 | $ | 179.12 | $ | 217.28 | |||||||||||
34
Item 6. Selected Financial Data
The following table presents selected consolidated financial data of Quidel Corporation. This historical data should be read in conjunction with the Consolidated Financial Statements and related Notes thereto in Item 8 and “Management’s Discussion and Analysis of Financial Condition and Results of Operation” in Item 7 in this Annual Report.
Consolidated Statements of Operations | |||||||||||||||||||
Year ended December 31, | |||||||||||||||||||
2017 (1) | 2016 (1) | 2015 | 2014 | 2013 (1) | |||||||||||||||
(in thousands, except per share data) | |||||||||||||||||||
Total revenues | $ | 277,743 | $ | 191,603 | $ | 196,129 | $ | 184,158 | $ | 177,325 | |||||||||
Cost of sales (2) | 121,601 | 79,872 | 78,029 | 80,463 | 73,055 | ||||||||||||||
Gross profit | 156,142 | 111,731 | 118,100 | 103,695 | 104,270 | ||||||||||||||
Research and development | 33,644 | 38,672 | 35,514 | 37,913 | 34,186 | ||||||||||||||
Sales and marketing (3) | 67,248 | 50,436 | 50,401 | 45,621 | 37,836 | ||||||||||||||
General and administrative (4) | 29,192 | 26,351 | 27,057 | 25,811 | 25,581 | ||||||||||||||
Acquisition and integration costs (4) | 16,506 | 711 | 2,390 | — | — | ||||||||||||||
Impairment loss | — | — | — | 3,558 | — | ||||||||||||||
Facility restructuring charge | — | — | — | — | 1,825 | ||||||||||||||
Total operating expenses | 146,590 | 116,170 | 115,362 | 112,903 | 99,428 | ||||||||||||||
Operating income (loss) | 9,552 | (4,439 | ) | 2,738 | (9,208 | ) | 4,842 | ||||||||||||
Interest expense, net | (17,588 | ) | (11,760 | ) | (12,035 | ) | (1,775 | ) | (1,408 | ) | |||||||||
(Loss) income before taxes | (8,036 | ) | (16,199 | ) | (9,297 | ) | (10,983 | ) | 3,434 | ||||||||||
Provision (benefit) for income taxes | 129 | (2,391 | ) | (3,218 | ) | (3,909 | ) | (3,956 | ) | ||||||||||
Net (loss) income | $ | (8,165 | ) | $ | (13,808 | ) | $ | (6,079 | ) | $ | (7,074 | ) | $ | 7,390 | |||||
Basic (loss) earnings per share | $ | (0.24 | ) | $ | (0.42 | ) | $ | (0.18 | ) | $ | (0.21 | ) | $ | 0.22 | |||||
Diluted (loss) earnings per share | $ | (0.24 | ) | $ | (0.42 | ) | $ | (0.18 | ) | $ | (0.21 | ) | $ | 0.21 | |||||
Shares used in basic per share calculation | 33,734 | 32,708 | 34,104 | 34,451 | 33,836 | ||||||||||||||
Shares used in diluted per share calculation | 33,734 | 32,708 | 34,104 | 34,451 | 34,947 | ||||||||||||||
Balance Sheet Data | |||||||||||||||||||
December 31, | |||||||||||||||||||
2017 | 2016 | 2015 | 2014 | 2013 | |||||||||||||||
(in thousands) | |||||||||||||||||||
Cash and cash equivalents | $ | 36,086 | $ | 169,508 | $ | 191,471 | $ | 200,895 | $ | 8,388 | |||||||||
Working capital | $ | 202,881 | $ | 191,782 | $ | 209,834 | $ | 238,096 | $ | 54,610 | |||||||||
Total assets | $ | 935,251 | $ | 388,250 | $ | 406,505 | $ | 447,411 | $ | 271,485 | |||||||||
Long-term debt and lease obligation, net of current portions | $ | 381,110 | $ | 148,319 | $ | 147,329 | $ | 142,575 | $ | 5,126 | |||||||||
Stockholders’ equity | $ | 227,104 | $ | 200,630 | $ | 218,676 | $ | 245,011 | $ | 223,779 | |||||||||
Common shares outstanding | 34,540 | 32,897 | 33,323 | 34,433 | 34,073 | ||||||||||||||
(1) | Includes the results of operations of the BioHelix, AnDiaTec, Immutopics and Triage and BNP Businesses, from dates of acquisition, May 6, 2013, August 26, 2013, March 18, 2016 and October 6, 2017, respectively. |
(2) | Includes reclassification of $6.5 million, $6.3 million, $6.3 million and $6.1 million from amortization of intangible assets from acquired business and technology to cost of sales for the years ended December 31, 2016, December 31, 2015, December 31, 2014 and December 31, 2013, respectively. |
(3) | Includes reclassification of $2.6 million, $2.5 million, $2.5 million and $2.1 million from amortization of intangible assets from acquired business and technology to sales and marketing for the years ended December 31, 2016, December 31, 2015, December 31, 2014 and December 31, 2013, respectively. |
(4) | The Company recorded reclassifications of acquisition and integration costs totaling $0.7 million and $2.4 million for years ended December 31, 2016 and 2015, respectively, from general and administrative expense as previously reported in the Consolidated Statements of Operations to conform to current year presentation. |
35
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
The following discussion of our financial condition and results of operations contains forward-looking statements within the meaning of the federal securities laws that involve material risks and uncertainties. This discussion should be read in conjunction with “A Warning About Forward-Looking Statements” on page 2 and “Risk Factors” under Item 1A of this Annual Report. In addition, our discussion of the financial condition and results of operations of Quidel Corporation in this Item 7 should be read in conjunction with our Consolidated Financial Statements and the related Notes included elsewhere in this Annual Report.
Overview and Executive Summary
We have a leadership position in the development, manufacturing and marketing of diagnostic testing solutions. These diagnostic testing solutions are separated into our four product categories, comprised of the following: rapid immunoassay, cardiac immunoassay, specialized diagnostic solutions and molecular diagnostic solutions. We sell our products directly to end users and distributors, in each case, for professional use in physician offices, hospitals, clinical laboratories, reference laboratories, leading universities, retail clinics, pharmacies and wellness screening centers. We market our products through a network of distributors and a direct sales force.
For the year ended December 31, 2017, total revenue increased 45% to $277.7 million as compared to the year ended December 31, 2016. Historically, a majority of our total revenues relate to three product families: Influenza, Strep A and pregnancy tests. Going forward, we expect our cardiology family of products acquired with the acquisition of the Triage and BNP Businesses from Alere on October 6, 2017 will also constitute a significant portion of our revenue. See further discussion in Note 12 in the Consolidated Financial Statements in Part II, Item 8 of this Annual Report. For the years ended December 31, 2017, 2016 and 2015, we derived approximately 58%, 62% and 65%, respectively, of our total revenues from sales of our influenza, Strep A and pregnancy tests. Additionally, a significant portion of our total revenue is from a relatively small number of distributors. Approximately 54%, 44% and 48% of our total revenue for the years ended December 31, 2017, 2016 and 2015, respectively, were related to sales through our three largest distributors.
Our primary objective is to increase shareholder value by building a broader-based diagnostic company capable of delivering revenue growth and consistent operating results. Our strategy is to identify potential market segments that provide, or are expected to provide, significant total market opportunities, and in which we can be successful by applying our significant expertise and know-how to develop differentiated technologies and products.
Our diagnostic testing solutions are designed to provide specialized results that serve a broad range of customers, by addressing the market requirements of ease of use, reduced cost, increased test accuracy and reduced time to result. Our current approach to address this diagnostic continuum relative to our strategy is to offer products in the following product categories:
• | rapid immunoassay tests for use in physician offices, hospital laboratories and emergency departments, retail clinics, eye health settings, pharmacies and other urgent care or alternative site settings; |
• | cardiac immunoassay tests for use in physician offices, hospital laboratories and emergency departments, and other urgent care or alternative site settings; |
• | specialized diagnostic solutions, including DFA and culture-based tests for the clinical virology laboratory and other products serving the bone health, autoimmune and complement research communities; and |
• | molecular diagnostic tests across a number of hospitals, moderately complex physician offices, laboratories and other segments. |
Our current focus to accomplish our primary objective includes the following:
• | leveraging our current infrastructure to develop and launch new Rapid Immunoassays and Cardiac Immunoassays such as additional assays for our Sofia® and Sofia® 2 Analyzers and Triage® MeterPro® systems and next-generation immunoassay analyzers; |
• | developing a molecular diagnostics franchise that incorporates three distinct testing platforms, Solana®, AmpliVue®, and Savanna® and that leverages our molecular assay development competencies; and |
• | strengthening our position with distribution partners and our end-user customers to gain more emphasis on our products. |
36
Our current initiatives to execute this strategy include the following:
• | continue to provide products that can compete effectively in the healthcare market where cost and quality are important; |
• | continue to focus on integrating the Triage and BNP Businesses acquired in late 2017; |
• | strengthen our international infrastructure to support the integration of the Triage and BNP Businesses and enhance our global footprint to support our international operations and future growth; |
• | continue to focus our research and development efforts on three areas: |
• | new proprietary product platform development; |
• | the creation of improved products and new products for existing markets and unmet clinical needs; and |
• | pursuit of collaborations with, or acquisitions of, other companies for new and existing products and markets that advance our strategy to develop differentiated technologies and products; |
• | strengthen our market and brand leadership in current markets by acquiring and/or developing and introducing clinically superior diagnostic solutions; |
• | strengthen our direct sales force to enhance relationships with integrated delivery networks, laboratories and hospitals, with a goal of driving growth through improved physician and laboratorian satisfaction; |
• | leverage our wireless connectivity and data management systems, including cloud-based tools; |
• | support payer evaluation of diagnostic tests and establishment of favorable reimbursement rates; |
• | provide clinicians with validated studies that encompass the clinical efficacy and economic efficiency of our diagnostic tests for the professional market; |
• | continue to create strong global alliances to support our efforts to achieve leadership in key markets and expand our presence in emerging markets; and |
• | further refine our manufacturing efficiencies and productivity improvements to increase profit, with continued focus on innovative products and markets and our efforts to leverage our core competency in new product development. |
Product development activities are inherently uncertain, and there can be no assurance that we will be able to obtain regulatory body clearance to market any of our products, or if we obtain clearances, that we will successfully commercialize any of our products. In addition, we may terminate our development efforts with respect to one or more of our products under development at any time, including before or during clinical trials.
Outlook
We anticipate revenue growth over the next year and a related positive impact on gross margin and earnings, assuming relatively normal respiratory seasons. This growth is expected to be driven primarily by the full year impact of the Triage/BNP acquisition, and increased sales of our Sofia assays and molecular products. In addition, we expect continued and significant investment in research and development activities as we invest in our next generation immunoassay and molecular platforms. We will continue our focus on prudently managing our business and delivering solid financial results, while at the same time striving to continue to introduce new products to the market and maintaining our emphasis on research and development investments for longer term growth. Finally, we will continue to evaluate opportunities to acquire new product lines, technologies and companies.
37
Results of Operations
Comparison of years ended December 31, 2017 and 2016
Total Revenues
The following table compares total revenues for the years ended December 31, 2017 and 2016 (in thousands, except percentages):
For the year ended December 31, | Increase (decrease) | ||||||||||||||
2017 | 2016 | $ | % | ||||||||||||
Rapid Immunoassay | $ | 165,099 | $ | 121,416 | $ | 43,683 | 36 | % | |||||||
Cardiac Immunoassay | 47,030 | — | 47,030 | 100 | % | ||||||||||
Specialized Diagnostic Solutions | 51,978 | 60,681 | (8,703 | ) | (14 | )% | |||||||||
Molecular Diagnostic Solutions | 13,636 | 9,506 | 4,130 | 43 | % | ||||||||||
Total revenues | $ | 277,743 | $ | 191,603 | $ | 86,140 | 45 | % | |||||||
For the year ended December 31, 2017, total revenues increased 45% to $277.7 million. The increase in total revenues was driven by the incremental Cardiac Immunoassay revenue from the acquisition of the Triage and BNP Businesses in October 2017 discussed further in Note 12 to the Consolidated Financial Statements in Part II, Item 8 of this Annual Report. In addition, the Company realized increases in Rapid Immunoassay revenues due primarily to growth in Influenza and Strep A products, bolstered by a severe cold and flu season. The increase in our Molecular Diagnostic Solutions was driven by continued gains on our Solana platform and higher sales of Strep A due to the cold and flu season. These increases were partially offset by decreased sales within our Specialized Diagnostics solutions category. This new category includes Virology, Bone Health and Complement products and grants and royalty revenues. The year over year decrease was due to the satisfaction of the terms of the Bill and Melinda Gates grant in the third quarter of 2016, leaving no grant revenue recognized in 2017.
Gross Profit
Gross profit increased by 40% over prior year, to $156.1 million, or 56.2% of revenue for the year ended December 31, 2017, compared to $111.7 million, or 58.3% of revenue for the year ended December 31, 2016. Gross margins declined slightly during 2017 due to the amortization of inventory step-up to fair value and intangible assets associated with the acquisition of Triage and BNP Businesses.
Operating Expenses
The following table compares operating expenses for the years ended December 31, 2017 and 2016 (in thousands, except percentages):
For the year ended December 31, | ||||||||||||||||||||
2017 | 2016 | |||||||||||||||||||
Operating expenses | As a % of total revenues | Operating expenses | As a % of total revenues | Increase (decrease) | ||||||||||||||||
$ | % | |||||||||||||||||||
Research and development | $ | 33,644 | 12 | % | $ | 38,672 | 20 | % | $ | (5,028 | ) | (13 | )% | |||||||
Sales and marketing | $ | 67,248 | 24 | % | $ | 50,436 | 26 | % | $ | 16,812 | 33 | % | ||||||||
General and administrative | $ | 29,192 | 11 | % | $ | 26,351 | 14 | % | $ | 2,841 | 11 | % | ||||||||
Acquisition and integration costs | $ | 16,506 | 6 | % | $ | 711 | — | % | $ | 15,795 | 2,222 | % | ||||||||
Research and Development Expense
Research and development expense for the year ended December 31, 2017 decreased from $38.7 million to $33.6 million primarily due to a decrease in development spending for the Savanna MDx platform and lower spend on clinical trial activities. These decreases are partially offset by additional expenses associated with the Triage and BNP Businesses.
38
Research and development expenses include direct external costs such as fees paid to third-party contractors and consultants, and internal direct and indirect costs such as compensation and other expenses for research and development personnel, supplies and materials, clinical trials and studies, facility costs and depreciation.
Due to the risks inherent in the product development process and given the early-stage of development of certain projects, we are unable to estimate with meaningful certainty the costs we will incur in the continued development of our product candidates for commercialization. We expect our research and development costs to be substantial as we move other product candidates into preclinical and clinical trials and advance our existing product candidates into later stages of development.
Sales and Marketing Expense
Sales and marketing expense for the year ended December 31, 2017 increased from $50.4 million to $67.2 million primarily driven by expenses associated with the newly acquired Triage and BNP Businesses in October 2017 and the InflammaDry and AdenoPlus diagnostic business from RPS Diagnostics in May 2017 as discussed in the Notes to the Consolidated Financial Statements in Part II, Item 8 of this Annual Report.
General and Administrative Expense
General and administrative expense for the year ended December 31, 2017 increased from $26.4 million to $29.2 million primarily due to higher incentive compensation and additional costs associated with the Triage and BNP Businesses. General and administrative expense primarily includes personnel costs, information technology, facilities and professional service fees.
Acquisition and Integration Costs
Acquisition and integration costs for the year ended December 31, 2017 increased from $0.7 million last year to $16.5 million this year primarily attributable to due diligence and integration costs related to the acquisition of the Triage and BNP Businesses on October 6, 2017. The Company recorded reclassifications of acquisition and integration costs totaling $0.7 million for the year ended December 31, 2016, from general and administrative expense as previously reported in the Consolidated Statements of Operations to conform to current year presentation.
Interest Expense, Net
Interest expense relates to accrued interest for the coupon and accretion of the discount on our $172.5 million 3.25% Convertible Senior Notes due 2020 ("Convertible Senior Notes") issued in December 2014, accrued interest and amortization of deferred loan costs associated with the Term Loan and the Revolving Credit Facility together (the "Senior Credit Facility"), and interest paid on our lease obligation associated with our San Diego McKellar facility. The increase in interest expense of $5.8 million for the year ended December 31, 2017 was primarily due to the interest incurred under the Senior Credit Facility entered into in connection with the acquisition of the Triage and BNP Businesses.
Income Taxes
On December 22, 2017, the U.S. government enacted comprehensive tax legislation referred to as the Tax Cuts and Jobs Act (the “Tax Act”). Shortly after the Tax Act was enacted, the SEC staff issued Staff Accounting Bulletin No. 118, Income Tax Accounting Implications of the Tax Cuts and Jobs Act (SAB 118), which provides guidance on accounting for the Tax Act’s impact. SAB 118 provides a measurement period, which should not extend beyond one year from the Tax Act enactment date, during which a company acting in good faith may complete the accounting for the impacts of the Tax Act under ASC Topic 740. In accordance with SAB 118, we must reflect the income tax effects of the Tax Act in the reporting period in which the accounting under ASC Topic 740 is complete.
For further information, see Note 4 to Consolidated Financial Statements in this Annual Report.
We recognized an income tax expense of $0.1 million and a benefit of $2.4 million for the years ended December 31, 2017 and 2016, respectively. The primary factors contributing to our tax expense in 2017 is the recognition of $0.4 million of deferred tax expenses from indefinite-lived assets, namely goodwill that is being amortized for tax, and current tax expense of $0.3 million related to state income taxes. These expenses are offset by an income tax benefit of $0.6 million for alternative minimum tax (AMT) credits which are now refundable based on the law changes in the Tax Act.
39
Comparison of years ended December 31, 2016 and 2015
Total Revenues
The following table compares total revenues for the years ended December 31, 2016 and 2015 (in thousands, except percentages):
For the year ended December 31, | Increase (decrease) | |||||||||||||
2016 | 2015 | $ | % | |||||||||||
Rapid Immunoassay | $ | 121,416 | $ | 130,348 | $ | (8,932 | ) | (7 | )% | |||||
Specialized Diagnostic Solutions | 60,681 | 60,358 | 323 | 1 | % | |||||||||
Molecular Diagnostic Solutions | 9,506 | 5,423 | 4,083 | 75 | % | |||||||||
Total revenues | $ | 191,603 | $ | 196,129 | $ | (4,526 | ) | (2 | )% | |||||
For the year ended December 31, 2016, total revenue decreased 2% to $191.6 million. The decrease in total revenues was primarily driven by lower Rapid Immunoassay revenues due to a weaker Influenza season in the first quarter of 2016 compared to the previous year. This decrease was partially offset by growth in our Molecular Diagnostic Solutions. The acquisition of Immutopics, Inc. ("Immutopics") contributed to the growth in our Specialized Diagnostic Solutions category.
Gross Profit
Gross profit for the year ended December 31, 2016 decreased by 5% as compared to the prior year, to $111.7 million, or 58% of revenue, as compared to $118.1 million, or 60% of revenue, for the year ended December 31, 2015. Gross margins decreased in 2016 due to unfavorable product mix, with lower influenza product sales in the same period as compared to the prior year.
Operating Expenses
The following table compares operating expenses for the years ended December 31, 2016 and 2015 (in thousands, except percentages):
For the year ended December 31, | ||||||||||||||||||||
2016 | 2015 | |||||||||||||||||||
Operating | As a % of total | Operating | As a % of total | Increase (decrease) | ||||||||||||||||
expenses | revenues | expenses | revenues | $ | % | |||||||||||||||
Research and development | $ | 38,672 | 20 | % | $ | 35,514 | 18 | % | $ | 3,158 | 9 | % | ||||||||
Sales and marketing | $ | 50,436 | 26 | % | $ | 50,401 | 26 | % | $ | 35 | — | % | ||||||||
General and administrative | $ | 26,351 | 14 | % | $ | 27,057 | 14 | % | $ | (706 | ) | (3 | )% | |||||||
Acquisition and integration costs | $ | 711 | — | % | $ | 2,390 | 1 | % | $ | (1,679 | ) | (70 | )% | |||||||
Research and Development Expense
Research and development expense for the year ended December 31, 2016 increased from $35.5 million to $38.7 million primarily due to an increase in development spending for the Savanna MDx platform and our next generation Sofia instrument, and an increase in clinical trials spending for our Solana and Sofia products. These increases were partially offset by lower spending on development of our Lyra products.
Research and development expenses include direct external costs such as fees paid to third-party contractors and consultants, and internal direct and indirect costs such as compensation and other expenses for research and development personnel, supplies and materials, clinical trials and studies, facility costs and depreciation.
Due to the risks inherent in the product development process and given the early-stage of development of certain projects, we are unable to estimate with meaningful certainty the costs we will incur in the continued development of our product candidates for commercialization. We expect our research and development costs to be substantial as we move other product candidates into preclinical and clinical trials and advance our existing product candidates into later stages of development.
40
Sales and Marketing Expense
Sales and marketing expense for the year ended December 31, 2016 remained relatively flat over prior year. At December 31, 2015, we employed more than 100 U.S. sales representatives. We utilized this sales force to work closely with our key distributors to drive market penetration of our products in the U.S. POC market, with a particular focus on addressing acute care and integrated delivery network customers.
General and Administrative Expense
General and administrative expense for the year ended December 31, 2016 decreased from $27.1 million to $26.4 million. The decline was due primarily to business development expenditures in the prior year period that did not repeat during 2016, as well as the suspension of the medical device excise tax for 2016. These decreases were partially offset by increased integration costs associated with the acquisition of Immutopics.
Acquisition and Integration Costs
Acquisition and integration costs for the year ended for the year ended December 31, 2016 decreased from $2.4 million to $0.7 million and is primarily attributable to one-time fees for professional services and internal costs related to business development activities occurring in 2015. This decrease was partially offset by increased integration costs associated with the acquisition of Immutopics in 2016. The Company recorded reclassifications of acquisition and integration costs totaling $0.7 million and $2.4 million for years ended December 31, 2016 and 2015, respectively, from general and administrative expense as previously reported in the Consolidated Statements of Operations to conform to current year presentation.
Interest Expense, Net
Interest expense in 2016 and 2015 related to accrued interest for the coupon and accretion of the discount on our $172.5 million 3.25% Convertible Senior Notes due 2020 ("Convertible Senior Notes") issued in December 2014 and interest paid on our lease obligation associated with our San Diego McKellar facility. The decrease in interest expense of $0.3 million for the year ended December 31, 2016 was primarily due to a gain on extinguishment of debt related to the repurchase of $5.2 million in principal of our Convertible Senior Notes during the first quarter of 2016.
Income Taxes
We recognized an income tax benefit of $2.4 million and $3.2 million for the years ended December 31, 2016 and 2015, respectively. The decrease in the income tax benefit in 2016 was primarily driven by the increase in the valuation allowance for our federal deferred tax assets.
Liquidity and Capital Resources
As of December 31, 2017 and 2016, our principal sources of liquidity consisted of the following (in thousands):
December 31, | |||||||
2017 | 2016 | ||||||
Cash and cash equivalents | $ | 36,086 | $ | 169,508 | |||
Working capital including cash, cash equivalents, and restricted cash | $ | 202,881 | $ | 191,782 | |||
Amount available to borrow under the Revolving Credit Facility | $ | 15,000 | N/A | ||||
As of December 31, 2017, we had $36.1 million in cash and cash equivalents, a $133.4 million decrease from the prior year. During the year ended December 31, 2017, the Company used $399.8 million in cash to acquire the Triage and BNP Businesses and $13.7 million to acquire the InflammaDry and AdenoPlus diagnostic business from RPS Diagnostics. Our cash requirements fluctuate as a result of numerous factors, such as the extent to which we generate cash from operations, progress in research and development projects and integration activities, competition and technological developments and the time and expenditures required to obtain governmental approval of our products. In addition, we intend to continue to evaluate candidates for new product lines, company or technology acquisitions or technology licensing. If we decide to proceed with any such transactions, we may need to incur additional debt or issue additional equity, to successfully complete the transactions.
41
Our primary source of liquidity, other than our holdings of cash and cash equivalents, has been cash flows from operations and financing. Cash generated from operations provides us with the financial flexibility we need to meet normal operating, investing, and financing needs. We anticipate that our current cash and cash equivalents, together with cash provided by operating activities will be sufficient to fund our near term capital and operating needs for at least the next 12 months. Normal operating needs include the planned costs to operate our business, including amounts required to fund working capital and capital expenditures. Our primary short-term needs for capital, which are subject to change, include expenditures related to:
• | support of commercialization efforts related to our current and future products, including support of our direct sales force and field support resources both in the United States and abroad; |
• | interest on and repayments of our Convertible Senior Notes, Senior Credit Facility, deferred consideration, contingent consideration and lease obligations; |
• | the continued advancement of research and development efforts; |
• | acquisitions of equipment and other fixed assets for use in our current and future manufacturing and research and development facilities; |
• | the integration of our recent strategic acquisitions and investments; and |
• | potential strategic acquisitions and investments. |
In December 2014, we issued Convertible Senior Notes in the aggregate principle amount of $172.5 million. The Convertible Senior Notes have a coupon rate of 3.25% and are due in December 2020. The Convertible Senior Notes were not convertible as of December 31, 2017. For detailed information of the terms of the Convertible Senior Notes, see Note 3 to the Consolidated Financial Statements in Part II, Item 8 of this Annual Report under the heading “3.25% Convertible Senior Notes due 2020,” which is incorporated by reference herein.
As of December 31, 2017, we have $24.3 million in fair value of contingent considerations and $223.2 million of deferred consideration associated with acquisitions to be settled in future periods.
In January 2016, our board of directors authorized an amendment to replenish the amount available under our share repurchase program up to an aggregate of $50.0 million in shares of common stock or Convertible Senior Notes. During 2016, we used $19.6 million to repurchase our common stock under the share repurchase program and $4.5 million to repurchase $5.2 million in principal amount of our outstanding Convertible Senior Notes.
On October 6, 2017, the Company entered into the Credit Agreement, which provided the Company with a $245.0 million Term Loan and a $25.0 million Revolving Credit Facility. The Term Loan and the Revolving Credit Facility will mature on October 6, 2022, provided that if any of Convertible Senior Notes remain outstanding on the date that is 91 days prior to the maturity date of the Convertible Senior Notes and the Company has not satisfied certain Refinancing Conditions, then the maturity date for the Term Loan and the Revolving Credit Facility will be the date that is 91 days prior to the maturity date of the Convertible Senior Notes. On the closing date of the Credit Agreement, the Company borrowed the entire amount of the Term Loan and $10.0 million under the Revolving Credit Facility. The Company used the proceeds of the Term Loan along with its cash on hand, to pay (i) the consideration for the Triage Business and (ii) the fees and expenses incurred in connection with the acquisition of the Triage and BNP Businesses.
We expect our revenue and operating expenses will significantly impact our cash management decisions. Our future capital requirements and the adequacy of our available funds to service our long-term debt and to fund working capital expenditures and business development efforts will depend on many factors, including:
• | our ability to successfully integrate our recently acquired businesses and realize revenue growth from our new technologies and create innovative products in our markets; |
• | our outstanding debt and covenant restrictions; |
• | leveraging our operating expenses to realize operating profits as we grow revenue; |
• | competing technological and market developments; and |
• | the need to enter into collaborations with other companies or acquire other companies or technologies to enhance or complement our product and service offerings. |
42
Cash Flow Summary
Year ended December 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Net cash provided by operating activities | $ | 27,709 | $ | 11,815 | $ | 36,880 | |||||
Net cash used for investing activities | (431,759 | ) | (16,970 | ) | (17,032 | ) | |||||
Net cash provided by (used for) financing activities | 270,608 | (16,799 | ) | (29,255 | ) | ||||||
Effect of exchange rate changes on cash | 20 | (9 | ) | (17 | ) | ||||||
Net decrease in cash and cash equivalents | $ | (133,422 | ) | $ | (21,963 | ) | $ | (9,424 | ) | ||
Cash provided by operating activities was $27.7 million during the year ended December 31, 2017. The Company had a net loss of $8.2 million for the year ended December 31, 2017, including non-cash charges of $59.4 million of depreciation, amortization of intangible assets, amortization of the inventory step-up to fair value related to the Triage and BNP Businesses acquired in 2017, stock-based compensation and accretion of interest on deferred consideration related to the acquired BNP Business. Partially offsetting this was a net working capital use of cash of $30.7 million.
Cash provided by operating activities was $11.8 million during the year ended December 31, 2016. The major contributors to the net cash provided by operating activities is the $36.7 million in non-cash add backs to the $13.8 million loss, associated with depreciation, amortization and stock-based compensation. Partially offsetting this was net working capital use of cash of $5.6 million.
Cash provided by operating activities was $36.9 million during the year ended December 31, 2015. The Company had a net loss of $6.1 million, including non-cash charges of $36.5 million of depreciation and amortization of intangible assets and property and equipment, stock-based compensation and amortization of debt discount and deferred issuance costs. The most significant change in operating assets and liabilities included a decrease in accounts receivable of $16.1 million due to increased collection efforts and a $3.1 million decrease in restricted cash as grant terms were met under the Bill and Melinda Gates Foundation grant agreement. This was offset by a decrease of $3.1 million in payables as a result of decreased production in the fourth quarter of 2015 compared to 2014.
Our investing activities used $431.8 million during the year ended December 31, 2017 primarily for the acquisition of the Triage and BNP Businesses as more fully described in Note 12 in the Consolidated Financial Statements in Part II, Item 8 of this Annual Report. Additionally, we used $13.7 million for the acquisition of the InflammaDry and AdenoPlus diagnostic business from RPS Diagnostics and $17.5 million primarily for the acquisition of production equipment, Sofia and Solana instruments available for sale or lease and building improvements. Our investing activities used $17.0 million during the year ended December 31, 2016; $5.1 million for the acquisition of Immutopics, and $11.9 million primarily for the acquisition of production equipment, Sofia and Solana instruments available for sale or lease and building improvements. Our investing activities used $17.0 million during the year ended December 31, 2015, primarily related to the acquisition of production equipment, building improvements and Sofia instruments available for sale or lease.
We are currently planning approximately $26.0 million in capital expenditures over the next 12 months. The primary purpose for our capital expenditures is to acquire manufacturing and scientific equipment, to purchase or develop information technology, and to implement facility improvements. We plan to fund these capital expenditures with the cash on our balance sheet. We have $5.9 million in firm purchase commitments with respect to planned inventory and capital expenditures as of December 31, 2017.
Cash provided by financing activities was $270.6 million during the year ended December 31, 2017, and was primarily related to proceeds from the issuance of the Term Loan and initial advances under the Revolving Credit Facility as more fully described in Note 3 in the Consolidated Financial Statements in Part II, Item 8 of this Annual Report. Additional cash was provided by the issuance of common stock of $25.4 million, partially offset by payment on debt issuance costs of $8.7 million. Cash used by financing activities was $16.8 million during the year ended December 31, 2016, of which $20.2 million was used for repurchases of common stock primarily related to our share repurchase program, and $4.5 million was used for the repurchase of some of the Convertible Senior Notes. These amounts were partially offset by proceeds from the issuance of common stock of $8.6 million. Cash used by financing activities was $28.7 million during the year ended December 31, 2015 and was driven primarily by $30.4 million of repurchases of common stock under our share repurchase program and $0.5 million of repurchases in connection with payment of minimum tax withholding obligations for certain employees relating to the lapse of restrictions on certain restricted stock units.
43
Off-Balance Sheet Arrangements
At December 31, 2017 and 2016, we did not have any relationships with unconsolidated entities or financial partners, such as entities often referred to as structured finance or special purpose entities, which would have been established for the purpose of facilitating off-balance sheet arrangements or other contractually narrow or limited purposes. As such, we are not materially exposed to any financing, liquidity, market or credit risk that could arise if we had engaged in such relationships.
Contractual Obligations
As of December 31, 2017, our future contractual obligations were as follows (in thousands):
Payment due by period | |||||||||||||||||||
Total | Less than 1 year | 1-3 Years | 3-5 Years | More than 5 years | |||||||||||||||
Senior Convertible Notes (1) | $ | 183,627 | $ | 5,438 | $ | 178,189 | — | $ | — | ||||||||||
Term Loan (2) | 245,000 | 12,250 | 42,875 | 189,875 | — | ||||||||||||||
Revolving Credit Facility (3) | 10,000 | 10,000 | — | — | — | ||||||||||||||
Deferred consideration (4) | 256,000 | 46,000 | 86,000 | 124,000 | |||||||||||||||
Lease obligation (5) | 2,869 | 946 | 956 | 967 | — | ||||||||||||||
Operating lease obligations (6) | 123,943 | 8,638 | 18,284 | 16,462 | 80,559 | ||||||||||||||
Non-cancelable purchase commitment (7) | 5,924 | 2,679 | 1,697 | 434 | 1,114 | ||||||||||||||
Total contractual obligations | $ | 827,363 | $ | 85,951 | $ | 328,001 | $ | 331,738 | $ | 81,673 | |||||||||
(1) | Includes the principal amount of our Convertible Senior Notes due in December 2020, as well as interest payments to be made semi-annually. |
(2) | Reflects the $245.0 million Term Loan that will mature on October 6, 2022. |
(3) | Reflects the $10.0 million of the $25.0 million Revolving Credit Facility that was borrowed at the closing of the acquisition of the Triage and BNP Businesses, which drawn amount was paid off in January 2018. |
(4) | Reflects the deferred consideration payments related to the acquisition of the BNP Business. |
(5) | Reflects our lease obligation on the approximately 78,000 square-foot McKellar San Diego facility in place as of December 31, 2017. The facility is subject to a financing arrangement with payments through December 2020. Our future obligation under this financing arrangement is included in the table above. |
(6) | Reflects future minimum lease obligations on facilities and equipment under operating leases in place as of December 31, 2017. In October of 2013, we entered into a lease for approximately 30,000 square feet of office space in San Diego. The lease expires in 2022 with options to extend the lease for two additional five-year periods. In the fourth quarter of 2016, we exercised our renewal option for the Athens, Ohio location. The amended lease expires in 2022 with the option to extend the lease for one additional five-year period through 2027. On January 5, 2018, we entered into a lease for the Summers Ridge facility, which has been included in the table above. |
(7) | Reflects our $5.9 million of non-cancelable commitments for planned inventory and capital expenditures under contractual arrangements. |
We have entered into various licensing agreements, which largely require payments based on specified product sales as well as the achievement of specific milestones. Royalty and license expenses under these various royalty and licensing agreements collectively totaled $0.6 million, $0.8 million and $1.1 million for the years ended December 31, 2017, 2016 and 2015, respectively.
We exclude liabilities pertaining to uncertain tax positions from our table of contractual obligations as we cannot make a reliable estimate of the period of cash settlement with the respective taxing authorities, nor the amount of the final cash settlement. As of December 31, 2017, we had approximately $1.4 million of liabilities associated with uncertain tax positions. See Note 4 in the Consolidated Financial Statements included in this Annual Report for further discussion of uncertain tax positions. The table also excludes $24.3 million in potential contingent consideration payments primarily related to the acquisition of the BNP Business and achievement of certain revenue targets under other acquisition agreements. We have not included amounts in the table because we cannot make a reasonably reliable estimate regarding the probability of the annual payments for the BNP Business or whether the milestones required for the other acquisition payments will be achieved. See Note 10 in the Consolidated Financial Statements included in this Annual Report for further discussion of our contingent consideration.
44
Recent Accounting Standards
For summary of recent accounting pronouncements applicable to our consolidated financial statements see “Company Operations and Summary of Significant Accounting Policies” in Note 1 to our Consolidated Financial Statements in Part II, Item 8, which is incorporated herein by reference.
Critical Accounting Policies and Estimates
Our discussion and analysis of our financial condition and results of operations are based on our consolidated financial statements, which have been prepared in accordance with accounting principles generally accepted in the U.S. The preparation of these financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, revenues and expenses, and related disclosure of contingent assets and liabilities. On an on-going basis, we evaluate our estimates, including those related to revenue recognition, stock-based compensation, goodwill and intangibles, business combinations, income taxes and convertible debt. We base our estimates on historical experience and on various other assumptions that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions.
We believe the following critical accounting policies affect our more significant judgments and estimates used in the preparation of our consolidated financial statements:
Reserve for Contractual Rebates and Discounts
The Company records revenues primarily from product sales. These revenues are recorded net of rebates and other discounts that are estimated at the time of sale, and are largely driven by various customer program offerings, including special pricing agreements, promotions and other volume-based incentives. Rebates and discounts are recorded as a reduction of sales and trade accounts receivable, based upon historical experience and estimated revenue levels.
Stock-Based Compensation
Compensation expense related to stock options granted is recognized ratably over the service vesting period for the entire option. The total number of stock options expected to vest is adjusted by estimated forfeiture rates. We determine the estimated fair value of each stock option on the date of grant using the Black-Scholes option valuation model. The computation of the expected option life is based on a weighted-average calculation combining the average life of options that have already been exercised and post-vest cancellations with the estimated life of the remaining vested and unexercised options. The expected volatility is based on the historical volatility of our stock. The risk-free interest rate is based on the U.S Treasury yield curve over the expected term of the option. Historically, we have not paid any cash dividends on our common stock, and we do not anticipate paying any cash dividends in the foreseeable future. Consequently, we use an expected dividend yield of zero in the Black-Scholes option valuation model. The estimated forfeiture rate is based on our historical experience and future expectations.
Compensation expense for time-based restricted stock units are measured at the grant date and recognized ratably over the vesting period. We determine the fair value of time-based and performance-based restricted stock units based on the closing market price of our common stock on the grant date. The recognition of compensation expense associated with performance-based restricted stock units requires judgment in assessing the probability of meeting the performance goals, as well as defined criteria for assessing achievement of the performance-related goals. For purposes of measuring compensation expense, the amount of shares ultimately expected to vest is estimated at each reporting date based on management’s expectations regarding the relevant performance criteria. The grant date of the performance-based restricted stock units takes place when the grant is authorized and the specific achievement goals are communicated. The communication date of the performance goals can impact the valuation and associated expense of the restricted stock units.
Goodwill and Intangible Assets
The effective life and related amortization of intangible assets with definite lives will be based on the higher of the percentage of usage or the straight-line method. Useful lives are based on the expected number of years the asset will generate revenue or otherwise be used by us. Goodwill and in-process research and development that have indefinite lives are not amortized but instead are tested at least annually for impairment, or more frequently when events or changes in circumstances indicate that the asset might be impaired. Examples of such events or circumstances include:
• | the asset’s ability to continue to generate income from operations and positive cash flow in future periods; |
• | any volatility or significant decline in our stock price and market capitalization compared to our net book value; |
• | loss of legal ownership or title to an asset; |
45
• | significant changes in our strategic business objectives and utilization of our assets; and |
• | the impact of significant negative industry or economic trends. |
If a change were to occur in any of the above-mentioned factors or estimates, the likelihood of a material change in our reported results would increase.
For goodwill and in-process research and development, a two-step test is used to identify the potential impairment and to measure the amount of impairment, if any. The first step is to compare the fair value of a reporting unit with the carrying amount, including goodwill and in-process research and development. If the fair value of a reporting unit exceeds its carrying amount, goodwill and in-process research and development are considered not impaired; otherwise, goodwill and in-process research and development are impaired and the loss is measured by performing step two. Under step two, the impairment loss is measured by comparing the implied fair value of the reporting unit with the carrying amount of goodwill and in-process research and development. We are required to perform periodic evaluations for impairment of goodwill balances. We completed our annual evaluation for impairment of goodwill and in-process research and development as of December 31, 2017 and determined that no impairment existed.
Business Combinations
The cost of an acquired business is assigned to the tangible and identifiable intangible assets acquired and liabilities assumed on the basis of the estimated fair values at the date of acquisition. We assess fair value, which is the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date, using a variety of methods including, but not limited to, an income approach and a market approach such as the estimation of future cash flows of acquired business and current selling prices of similar assets. Fair value of the assets acquired and liabilities assumed, including intangible assets, in-process research and development (IPR&D), and contingent payments, are measured based on the assumptions and estimations with regards to the variable factors such as the amount and timing of future cash flows for the asset or liability being measured, appropriate risk-adjusted discount rates, nonperformance risk, or other factors that market participants would consider. Upon acquisition, we determine the estimated economic lives of the acquired intangible assets for amortization purposes, which are based on the underlying expected cash flows of such assets. When applicable, adjustments to inventory and property, plant and equipment are based on the fair market value of inventory and amortized into income based on the period in which the underlying inventory is sold. Goodwill is an asset representing the future economic benefits arising from other assets acquired in a business combination that is not individually identified and separately recognized. Actual results may vary from projected results and assumptions used in the fair value assessments.
Income Taxes
Significant judgment is required in determining our provision for income taxes, current tax assets and liabilities, deferred tax assets and liabilities, and our future taxable income, both as a whole and in various tax jurisdictions, for purposes of assessing our ability to realize future benefit from our deferred tax assets. A valuation allowance may be established to reduce our deferred tax assets to the amount that is considered more likely than not to be realized through the generation of future taxable income and other tax planning opportunities. In 2016 and 2017, we evaluated our gross deferred tax assets, including an assessment of cumulative income or loss over the prior three-year period and future periods, to determine if a valuation allowance was required. A significant piece of objective negative evidence evaluated was the cumulative before-tax loss incurred over the three-year periods ended December 31, 2017 and 2016. Such objective evidence limits the ability to consider other subjective evidence such as our projections for future profitability. On the basis of this evaluation, as of December 31, 2017, we recorded a valuation allowance of $15.2 million. This valuation allowance represents the portion of the deferred tax asset that management could no longer conclude was more likely than not to be realized. We will continue to assess the need for a valuation allowance on our deferred tax assets by evaluating both positive and negative evidence that may exist.
We recognize liabilities for uncertain tax positions based on a two-step process. The first step is to evaluate the tax position for recognition by determining if the weight of available evidence indicates that it is more likely than not that the position will be sustained during an audit, including resolution of related appeals or litigation processes, if any. The second step is to measure the tax benefit as the largest amount that is more than 50% likely of being realized upon settlement. While we believe that we have appropriate support for the positions taken on our tax returns, we regularly assess the potential outcome of examinations by tax authorities in determining the adequacy of our provision for income taxes. See Note 4 in the Consolidated Financial Statements included in this Annual Report for more information on income taxes.
Convertible Debt
We account for convertible debt instruments that may be settled in cash upon conversion (including combination settlement of cash equal to the “principal portion” and delivery of the “share amount” in excess of the conversion value over
46
the principal portion in shares of common stock and/or cash) by separating the liability and equity components of the instruments in a manner that reflects our nonconvertible debt borrowing rate. We determine the carrying amount of the liability component by measuring the fair value of similar debt instruments that do not have the conversion feature. If no similar debt instrument exists, we estimate fair value by using assumptions that market participants would use in pricing a debt instrument, including market interest rates, credit standing, yield curves and volatilities. Determining the fair value of the debt component requires the use of accounting estimates and assumptions. These estimates and assumptions are judgmental in nature and could have a significant impact on the determination of the debt component, and the associated non-cash interest expense.
In December 2014, we issued $172.5 million aggregate principal amount of 3.25% Convertible Senior Notes due 2020. We assigned a value to the debt component of our Convertible Senior Notes equal to the estimated fair value of similar debt instruments without the conversion feature, which resulted in us recording the debt at a discount. We are amortizing the debt discount over the life of the Convertible Senior Notes as additional non-cash interest expense utilizing the effective interest method. For additional information, see Note 3 in the Consolidated Financial Statements included in this Annual Report.
Item 7A. Quantitative and Qualitative Disclosures About Market Risk
Interest Rate Risk
We are not subject to interest rate risk on our Convertible Senior Notes as the Notes have a fixed rate of 3.25%. For fixed rate debt, changes in interest rates will generally affect the fair value of the debt instrument, but not our earnings or cash flows. Under our current policies, we do not use interest rate derivative instruments to manage our exposure to changes in interest rates.
The fair market value of our Senior Credit Facility interest rate debt is subject to interest rate risk. Generally, the fair market value of the Senior Credit Facility interest rate debt will vary as interest rates increase or decrease. We had $255.0 million outstanding under our Senior Credit Facility at December 31, 2017. The weighted average interest rate on these borrowings is 5.19% as of December 31, 2017. A hypothetical 100 basis point adverse move in interest rates along the entire interest rate yield curve would increase our annual interest expense by approximately $1.5 million. Based on our market risk sensitive instruments outstanding at December 31, 2017, we have determined that there was no material market risk exposure from such instruments to our consolidated financial position, results of operations or cash flows as of such dates.
Our current investment policy with respect to our cash and cash equivalents focuses on maintaining acceptable levels of interest rate risk and liquidity. Although we continually evaluate our placement of investments, as of December 31, 2017, our cash and cash equivalents were placed in the Company's highly liquid operating accounts.
Foreign Currency Exchange Risk
The majority of our international sales are negotiated for and paid in U.S. dollars. Nonetheless, these sales are subject to currency risks, since changes in the values of foreign currencies relative to the value of the U.S. dollar can render our products comparatively more expensive. These exchange rate fluctuations could negatively impact international sales of our products, as could changes in the general economic conditions in those markets. Continued change in the values of the Euro, the Chinese Renminbi, the Japanese Yen and other foreign currencies could have an impact on our business, financial condition and results of operations. We do not currently hedge against exchange rate fluctuations, which means that we are fully exposed to exchange rate changes. In addition, we have certain agreements whereby we share the foreign currency exchange fluctuation risk. We may, in the future, enter into similar such arrangements.
47
Item 8. Financial Statements and Supplementary Data
Index of Consolidated Financial Statements and Schedule
48
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Shareholders and the Board of Directors of Quidel Corporation
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Quidel Corporation (the “Company“) as of December 31, 2017 and 2016, the related consolidated statements of operations, comprehensive loss, stockholders‘ equity, and cash flows, for each of the three years in the period ended December 31, 2017 and the related notes and schedule listed in the Index at Item 15(a)(2) (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2017 and 2016, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2017, in conformity with US generally accepted accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the Company's internal control over financial reporting as of December 31, 2017, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework), and our report dated February 27, 2018 expressed an unqualified opinion thereon.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company‘s financial statements based on our audits. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the US federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures include examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ Ernst & Young LLP | |
We have served as the Company‘s auditor since 2002. | |
San Diego, California | |
February 27, 2018 | |
49
QUIDEL CORPORATION
CONSOLIDATED BALANCE SHEETS
(in thousands, except par value)
December 31, | |||||||
2017 | 2016 | ||||||
ASSETS | |||||||
Current assets: | |||||||
Cash and cash equivalents | $ | 36,086 | $ | 169,508 | |||
Accounts receivable, net | 67,046 | 24,990 | |||||
Inventories | 67,078 | 26,045 | |||||
Assets held for sale | 146,644 | — | |||||
Prepaid expenses and other current assets | 14,375 | 4,851 | |||||
Total current assets | 331,229 | 225,394 | |||||
Property, plant and equipment, net | 61,585 | 50,858 | |||||
Goodwill | 337,028 | 83,834 | |||||
Intangible assets, net | 203,827 | 27,639 | |||||
Other non-current assets | 1,582 | 525 | |||||
Total assets | $ | 935,251 | $ | 388,250 | |||
LIABILITIES AND STOCKHOLDERS’ EQUITY | |||||||
Current liabilities: | |||||||
Accounts payable | $ | 27,279 | $ | 16,047 | |||
Accrued payroll and related expenses | 15,926 | 9,642 | |||||
Current portion of contingent consideration | 6,293 | 2,826 | |||||
Current portion of deferred consideration | 46,000 | — | |||||
Current portion of Revolving Credit Facility | 10,000 | — | |||||
Current portion of long-term debt | 10,184 | — | |||||
Other current liabilities | 12,666 | 5,097 | |||||
Total current liabilities | 128,348 | 33,612 | |||||
Convertible Senior Notes | 149,868 | 144,340 | |||||
Term Loan | 227,394 | — | |||||
Deferred consideration - non-current | 177,158 | — | |||||
Contingent consideration - non-current | 18,008 | 2,349 | |||||
Deferred tax liability - non-current | 430 | 58 | |||||
Other non-current liabilities | 6,941 | 7,261 | |||||
Commitments and contingencies (Note 8) | |||||||
Stockholders’ equity: | |||||||
Preferred stock, $.001 par value per share; 5,000 shares authorized; none issued or outstanding at December 31, 2017 and 2016 | — | — | |||||
Common stock, $.001 par value per share; 97,500 shares authorized; 34,540 and 32,897 shares issued and outstanding at December 31, 2017 and 2016, respectively | 35 | 33 | |||||
Additional paid-in capital | 239,489 | 204,905 | |||||
Accumulated other comprehensive loss | — | (53 | ) | ||||
Accumulated deficit | (12,420 | ) | (4,255 | ) | |||
Total stockholders’ equity | 227,104 | 200,630 | |||||
Total liabilities and stockholders’ equity | $ | 935,251 | $ | 388,250 | |||
See accompanying notes.
50
QUIDEL CORPORATION
CONSOLIDATED STATEMENTS OF OPERATIONS
(in thousands, except per share data)
Year ended December 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Total revenues | $ | 277,743 | $ | 191,603 | $ | 196,129 | |||||
Cost of sales | 121,601 | 79,872 | 78,029 | ||||||||
Gross profit | 156,142 | 111,731 | 118,100 | ||||||||
Research and development | 33,644 | 38,672 | 35,514 | ||||||||
Sales and marketing | 67,248 | 50,436 | 50,401 | ||||||||
General and administrative | 29,192 | 26,351 | 27,057 | ||||||||
Acquisition and integration costs | 16,506 | 711 | 2,390 | ||||||||
Total operating expenses | 146,590 | 116,170 | 115,362 | ||||||||
Operating income (loss) | 9,552 | (4,439 | ) | 2,738 | |||||||
Interest expense, net | (17,588 | ) | (11,760 | ) | (12,035 | ) | |||||
Loss before taxes | (8,036 | ) | (16,199 | ) | (9,297 | ) | |||||
Provision (benefit) for income taxes | 129 | (2,391 | ) | (3,218 | ) | ||||||
Net loss | $ | (8,165 | ) | $ | (13,808 | ) | $ | (6,079 | ) | ||
Basic and diluted loss per share | $ | (0.24 | ) | $ | (0.42 | ) | $ | (0.18 | ) | ||
Shares used in basic and diluted per share calculation | 33,734 | 32,708 | 34,104 | ||||||||
See accompanying notes.
51
QUIDEL CORPORATION
CONSOLIDATED STATEMENTS OF COMPREHENSIVE LOSS
(in thousands)
Year ended December 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Net loss | $ | (8,165 | ) | $ | (13,808 | ) | $ | (6,079 | ) | ||
Other comprehensive income, net of tax | |||||||||||
Changes in cumulative translation adjustment | 53 | (22 | ) | (2 | ) | ||||||
Comprehensive loss | $ | (8,112 | ) | $ | (13,830 | ) | $ | (6,081 | ) | ||
See accompanying notes.
52
QUIDEL CORPORATION
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
(in thousands)
Common Stock | ||||||||||||||||||||||
Shares | Par | Additional paid-in capital | Accumulated other comprehensive loss | Retained earnings (accumulated deficit) | Total stockholders’ equity | |||||||||||||||||
Balance at January 1, 2015 | 34,433 | $ | 34 | $ | 229,374 | $ | (29 | ) | $ | 15,632 | $ | 245,011 | ||||||||||
Issuance of common stock under equity compensation plans | 308 | — | 3,318 | — | — | 3,318 | ||||||||||||||||
Excess tax benefit from share-based compensation | — | — | 571 | — | — | 571 | ||||||||||||||||
Stock-based compensation expense | — | — | 6,791 | — | — | 6,791 | ||||||||||||||||
Repurchases of common stock | (1,418 | ) | (1 | ) | (30,933 | ) | — | — | (30,934 | ) | ||||||||||||
Changes in cumulative translation adjustment, net of tax | — | — | — | (2 | ) | — | (2 | ) | ||||||||||||||
Net loss | — | — | — | — | (6,079 | ) | (6,079 | ) | ||||||||||||||
Balance at December 31, 2015 | 33,323 | 33 | 209,121 | (31 | ) | 9,553 | 218,676 | |||||||||||||||
Issuance of common stock under equity compensation plans | 755 | — | 9,365 | — | — | 9,365 | ||||||||||||||||
Stock-based compensation expense | — | — | 7,134 | — | — | 7,134 | ||||||||||||||||
Repurchase of Convertible Senior Notes | — | — | (547 | ) | — | — | (547 | ) | ||||||||||||||
Repurchases of common stock | (1,181 | ) | — | (20,168 | ) | — | — | (20,168 | ) | |||||||||||||
Changes in cumulative translation adjustment, net of tax | — | — | — | (22 | ) | — | (22 | ) | ||||||||||||||
Net loss | — | — | — | — | (13,808 | ) | (13,808 | ) | ||||||||||||||
Balance at December 31, 2016 | 32,897 | 33 | 204,905 | (53 | ) | (4,255 | ) | 200,630 | ||||||||||||||
Issuance of common stock under equity compensation plans | 1,669 | 2 | 26,077 | — | — | 26,079 | ||||||||||||||||
Stock-based compensation expense | — | — | 9,048 | — | — | 9,048 | ||||||||||||||||
Repurchases of common stock | (26 | ) | — | (541 | ) | — | — | (541 | ) | |||||||||||||
Changes in cumulative translation adjustment, net of tax | — | — | — | 53 | — | 53 | ||||||||||||||||
Net loss | — | — | — | — | (8,165 | ) | (8,165 | ) | ||||||||||||||
Balance at December 31, 2017 | 34,540 | $ | 35 | $ | 239,489 | $ | — | $ | (12,420 | ) | $ | 227,104 | ||||||||||
See accompanying notes.
53
QUIDEL CORPORATION
CONSOLIDATED STATEMENTS OF CASH FLOWS
(in thousands)
Year ended December 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
OPERATING ACTIVITIES | |||||||||||
Net loss | $ | (8,165 | ) | $ | (13,808 | ) | $ | (6,079 | ) | ||
Adjustments to reconcile net loss to net cash provided by operating activities: | |||||||||||
Depreciation, amortization and other | 30,762 | 22,796 | 23,386 | ||||||||
Stock-based compensation expense | 9,061 | 7,986 | 7,419 | ||||||||
Amortization of debt discount and deferred issuance costs | 6,022 | 5,891 | 5,664 | ||||||||
Change in fair value of acquisition contingencies | (81 | ) | (485 | ) | (88 | ) | |||||
Accretion of interest on deferred consideration | 2,608 | — | — | ||||||||
Amortization of inventory step-up to fair value | 10,950 | — | — | ||||||||
Change in deferred tax assets and liabilities | 365 | (2,603 | ) | (4,027 | ) | ||||||
Gain on extinguishment of Convertible Senior Notes | — | (421 | ) | — | |||||||
Changes in assets and liabilities: | |||||||||||
Accounts receivable | (42,052 | ) | (6,265 | ) | 16,060 | ||||||
Inventories | 362 | 859 | (1,637 | ) | |||||||
Prepaid expenses and other current and non-current assets | (9,113 | ) | (552 | ) | (1,039 | ) | |||||
Restricted cash | — | 63 | 3,064 | ||||||||
Accounts payable | 12,956 | 4,323 | (3,082 | ) | |||||||
Accrued payroll and related expenses | 7,130 | (375 | ) | 1,061 | |||||||
Deferred grant revenue | — | (3,658 | ) | (2,672 | ) | ||||||
Other current and non-current liabilities | 6,904 | (1,936 | ) | (1,150 | ) | ||||||
Net cash provided by operating activities | 27,709 | 11,815 | 36,880 | ||||||||
INVESTING ACTIVITIES | |||||||||||
Acquisitions of property, equipment and intangibles | (17,510 | ) | (11,909 | ) | (17,032 | ) | |||||
Acquisition of other businesses, net of cash acquired | (14,451 | ) | (5,061 | ) | — | ||||||
Acquisition of Triage and BNP Businesses | (399,798 | ) | — | — | |||||||
Net cash used for investing activities | (431,759 | ) | (16,970 | ) | (17,032 | ) | |||||
FINANCING ACTIVITIES | |||||||||||
Proceeds from issuance of Term Loan | 245,000 | — | — | ||||||||
Proceeds from issuance of Revolving Credit Facility | 10,000 | — | — | ||||||||
Proceeds from issuance of common stock | 25,426 | 8,575 | 2,911 | ||||||||
Payments of debt issuance costs | (8,682 | ) | — | (365 | ) | ||||||
Payments on lease obligation | (98 | ) | (540 | ) | (509 | ) | |||||
Repurchases of common stock | (541 | ) | (20,168 | ) | (30,934 | ) | |||||
Repurchases of Convertible Senior Notes | — | (4,459 | ) | — | |||||||
Payments on acquisition contingencies | (497 | ) | (207 | ) | (129 | ) | |||||
Payment for acquisition holdback | — | — | (229 | ) | |||||||
Net cash provided by (used for) financing activities | 270,608 | (16,799 | ) | (29,255 | ) | ||||||
Effect of exchange rate changes on cash | 20 | (9 | ) | (17 | ) | ||||||
Net decrease in cash and cash equivalents | (133,422 | ) | (21,963 | ) | (9,424 | ) | |||||
Cash and cash equivalents, beginning of period | 169,508 | 191,471 | 200,895 | ||||||||
Cash and cash equivalents, at end of period | $ | 36,086 | $ | 169,508 | $ | 191,471 | |||||
See accompanying notes.
54
Year ended December 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
SUPPLEMENTAL DISCLOSURES OF CASH FLOW INFORMATION | |||||||||||
Cash paid during the period for interest | $ | 9,137 | $ | 6,488 | $ | 6,998 | |||||
Cash paid during the period for income taxes | $ | 1,274 | $ | 490 | $ | 1,922 | |||||
NON-CASH INVESTING ACTIVITIES | |||||||||||
Purchase of property, equipment and intangibles by incurring current liabilities | $ | 1,446 | $ | 3,280 | $ | 239 | |||||
NON-CASH FINANCING ACTIVITIES | |||||||||||
Decrease of accrued payroll and related expenses upon issuance of common stock | $ | 903 | $ | 539 | $ | 408 | |||||
Receivable for stock option exercises | $ | — | $ | 251 | $ | — | |||||
Deferred consideration for acquisition of BNP Business | $ | 220,550 | $ | — | $ | — | |||||
See accompanying notes.
55
QUIDEL CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Note 1. Company Operations and Summary of Significant Accounting Policies
Quidel Corporation (the “Company”) commenced operations in 1979. The Company operates in one business segment, which develops, manufactures and markets rapid diagnostic testing solutions. These diagnostic tests can be categorized in the following product categories: Rapid Immunoassay, Cardiac Immunoassay, Specialized Diagnostic Solutions and Molecular Diagnostic Solutions. The Company sells its products directly to end users and distributors, in each case, for professional use in physician offices, hospitals, clinical laboratories, reference laboratories, leading universities, retail clinics and wellness screening centers. The Company markets its products through a network of distributors and a direct sales force and temporary transition service arrangements entered into with Abbott Laboratories in connection with the acquisition of the Triage and BNP Businesses.
The accompanying consolidated financial statements of the Company and its subsidiaries have been prepared in accordance with generally accepted accounting principles in the U.S.
Consolidation—The consolidated financial statements include the accounts of the Company and its wholly owned subsidiaries. All significant intercompany accounts and transactions have been eliminated.
Cash and Cash Equivalents—The Company considers cash equivalents to be highly liquid investments with a maturity at the date of purchase of three months or less. The Company invests its cash equivalents primarily in money market funds and commercial paper. Cash equivalents are maintained with high quality institutions.
Accounts Receivable—The Company sells its products directly to hospitals and reference laboratories as well as to distributors in the U.S. and sells directly to hospitals and labs and through distribution internationally (see Note 9). The Company periodically assesses the financial strength of these customers and establishes reserves for anticipated losses when necessary, which historically have not been material. The Company’s reserves primarily consist of amounts related to cash discounts and contract rebates. The balance of accounts receivable is net of reserves of $12.3 million and $7.2 million at December 31, 2017 and 2016, respectively.
Concentration of Credit Risk—Financial instruments that potentially subject the Company to significant concentrations of credit risk consists principally of trade accounts receivable.
The Company performs credit evaluations of its customers’ financial condition and limits the amount of credit extended when deemed necessary, but generally requires no collateral. Credit quality is monitored regularly by reviewing credit history. The Company believes that the concentration of credit risk in its trade accounts receivables is moderated by its credit evaluation process, relatively short collection terms, the high level of credit worthiness of its customers, and letters of credit issued on the Company’s behalf. Potential credit losses are limited to the gross value of accounts receivable.
Inventories—Inventories are stated at the lower of cost (first-in, first-out) or net realizable value. The Company reviews the components of its inventory on a quarterly basis for excess, obsolete and impaired inventory and makes appropriate dispositions as obsolete stock is identified.
Assets Held for Sale—Assets to be disposed that meet the held for sale criteria are reported at the lower of their carrying amount and fair value, less costs to sell, and are no longer depreciated.
Property, Plant and Equipment—Property, plant and equipment is recorded at cost and depreciated over the estimated useful lives of the assets (three to 15 years) using the straight-line method. Amortization of leasehold improvements is computed on the straight-line method over the estimated useful lives of the assets.
56
Goodwill and Intangible Assets—Intangible assets are recorded at cost and amortized on a straight-line basis over their estimated useful lives, except for indefinite-lived intangibles such as goodwill. Software development costs associated with software to be leased or otherwise marketed are expensed as incurred until technological feasibility has been established. After technological feasibility is established, software development costs are capitalized. The capitalized cost is amortized on a straight-line basis over the estimated product life or on the ratio of current revenues to total projected product revenues, whichever is greater.
Convertible Debt—The Company accounts for convertible debt instruments that may be settled in cash upon conversion (including combination settlement of cash equal to the “principal portion” and delivery of the “share amount” in excess of the conversion value over the principal portion in shares of common stock and/or cash) by separating the liability and equity components of the instruments in a manner that reflects our nonconvertible debt borrowing rate. The Company determines the carrying amount of the liability component by measuring the fair value of similar debt instruments that do not have the conversion feature. If no similar debt instrument exists, the Company estimates fair value by using assumptions that market participants would use in pricing a debt instrument, including market interest rates, credit standing, yield curves and volatilities. Determining the fair value of the debt component requires the use of accounting estimates and assumptions. These estimates and assumptions are judgmental in nature and could have a significant impact on the determination of the debt component, and the associated non-cash interest expense. See Note 3 for additional discussion of the Convertible Senior Notes issued in December 2014.
Revenue Recognition—The Company records revenues primarily from product sales. These revenues are recorded net of rebates and other discounts. These rebates and discounts are estimated at the time of sale, and are largely driven by various customer program offerings, including special pricing agreements, promotions and other volume-based incentives. Revenues from product sales are recorded upon passage of title and risk of loss to the customer. Passage of title to the product and recognition of revenue occurs upon delivery to the customer when sales terms are free on board (“FOB”) destination and at the time of shipment when the sales terms are FOB shipping point and there is no right of return.
A portion of product sales includes revenues for diagnostic kits, which are utilized on leased instrument systems under the Company’s “reagent rental” program. The reagent rental program provides customers the right to use the instruments at no separate cost to the customer in consideration for a multi-year agreement to purchase annual minimum amounts of consumables (“reagents” or “diagnostic kits”). When an instrument is placed with a customer under a reagent rental agreement, the Company retains title to the equipment and it remains capitalized on the Company’s Consolidated Balance Sheets as property and equipment. The instrument is depreciated on a straight-line basis over the life of the instrument. Depreciation expense is recorded in cost of sales included in the Consolidated Statements of Operations. The reagent rental agreements represent one unit of accounting as the instrument and consumables (reagents) are interdependent in producing a diagnostic result and neither has a stand-alone value with respect to these agreements. No revenue is recognized at the time of instrument placement. All revenue is recognized when the title and risk of loss for the diagnostic kits have passed to the customer.
Royalty income from the grant of license rights is recognized during the period in which the revenue is earned and the amount is determinable from the licensee.
From time to time, the Company earns income from grants for research and commercialization activities. For the years ended December 31, 2016 and 2015, the Company recognized $6.5 million and $5.1 million as grant revenue, respectively, related to the Bill and Melinda Gates Foundation to develop, manufacture and validate a quantitative, low-cost, nucleic acid assay for HIV drug treatment monitoring on the integrated Savanna MDx platform for use in limited resource settings. Cash payments received were restricted as to use until expenditures contemplated in the grant were incurred or committed. As of December 31, 2016, all payment related milestones have been achieved and all of the grant revenue of $20.9 million was fully recognized. As such, the Company recognized no grant revenue during the twelve months ended December 31, 2017.
Research and Development Costs—Research and development costs are charged to operations as incurred. In conjunction with certain third party service agreements, the Company is required to make periodic payments based on achievement of certain milestones. The costs related to these research and development services are also charged to operations as incurred.
Product Shipment Costs—Product shipment costs are included in sales and marketing expense in the accompanying Consolidated Statements of Operations. Shipping and handling costs were $3.7 million, $3.8 million and $3.9 million for the years ended December 31, 2017, 2016 and 2015, respectively.
Advertising Costs—Advertising costs are expensed as incurred. Advertising costs were $0.5 million, $0.3 million and $0.7 million for the years ended December 31, 2017, 2016 and 2015, respectively.
57
Deferred Rent—The Company enters into lease arrangements for office space under non-cancelable operating leases. Certain of the operating lease agreements contain rent escalation provisions, which are considered in determining the straight-line rent expense to be recorded over the lease term. The lease term begins on the date of initial possession of the leased property for purposes of recognizing lease expense on a straight-line basis over the term of the lease. The Company does not assume renewals in the determination of the lease term unless the renewals are deemed to be reasonably assured at lease inception. The difference between rent expense and amounts paid under the Company's lease agreements are recorded as deferred rent.
Income Taxes—Deferred income taxes reflect the net tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes, using enacted tax rates in effect for the year in which the differences are expected to reverse. Valuation allowances are established, when necessary, to reduce deferred tax assets to the amount expected to be realized. The Company’s policy is to recognize the interest expense and penalties related to income tax matters as a component of income tax expense.
Fair Value of Financial Instruments— The Company uses the fair value hierarchy established in Accounting Standards Codification (“ASC”) Topic 820, Fair Value Measurements and Disclosures, which requires that the valuation of assets and liabilities subject to fair value measurements be classified and disclosed by the Company in one of the following three categories:
Level 1: Unadjusted quoted prices in active markets that are accessible at the measurement date for identical, unrestricted assets or liabilities;
Level 2: Quoted prices for similar assets and liabilities in active markets, quoted prices in markets that are not active, or inputs which are observable, either directly or indirectly, for substantially the full term of the asset or liability; and
Level 3: Prices or valuation techniques that require inputs that are both significant to the fair value measurement and unobservable (i.e. supported by little or no market activity).
The carrying amounts of the Company’s financial instruments, including cash, receivables, accounts payable and accrued liabilities approximate their fair values due to their short-term nature.
Reclassifications— The Company recorded reclassifications of acquisition and integration costs totaling $0.7 million and $2.4 million for years ended December 31, 2016 and 2015, respectively, from general and administrative expense as previously reported in the Consolidated Statements of Operations to conform to current year presentation. The Company believes these reclassifications provide greater clarity and insight into the consolidated financial statements for the periods presented.
The Company also recorded reclassifications of $6.5 million and $6.3 million for the years ended December 31, 2016 and 2015, respectively, from amortization of intangible assets from acquired business and technology to cost of sales expense as previously reported in the Consolidated Statements of Operations. In addition, the Company recorded reclassifications of $2.6 million and $2.5 million for the years ended December 31, 2016 and 2015, respectively, from amortization of intangible assets from acquired business and technology to sales and marketing expense to conform to current year presentation. These reclassifications did not impact the net loss as previously reported or any prior amounts reported on the Consolidated Balance Sheets, Statements of Cash Flows or Statements of Comprehensive Loss.
Product Warranty—The Company generally sells products with a limited product warranty and certain limited indemnifications. Due to product testing, the short time between product shipment and the detection and correction of product failures and a low historical rate of payments on indemnification claims, the historical activity and the related expense were not significant for the fiscal years presented.
Stock-Based Compensation—Compensation expense related to stock options granted is recognized ratably over the service vesting period for the entire option. The total number of stock options expected to vest is adjusted by estimated forfeiture rates. The Company determined the estimated fair value of each stock option on the date of grant using the Black-Scholes option valuation model. The fair value of restricted stock units is determined based on the closing market price of the Company’s common stock on the grant date. Compensation expense for time-based restricted stock units is measured at the grant date and recognized ratably over the vesting period. For purposes of measuring compensation expense for performance-based restricted stock units, the amount of shares ultimately expected to vest is estimated at each reporting date based on management’s expectations regarding the relevant performance criteria. The recognition of compensation expense associated with the performance-based restricted stock units requires judgment in assessing the probability of meeting the performance goals. This may result in significant expense recognition in the period in which the performance goals are met or when
58
achievement of the goals is deemed probable or may result in the reversal of previously recognized stock-based compensation expense if the performance criteria are deemed not probable of being met.
Computation of (Loss) Earnings Per Share—For the years ended December 31, 2017, 2016 and 2015, basic loss per share was computed by dividing net loss by the weighted-average number of common shares outstanding, including restricted stock units vested during the period. Diluted earnings per share (“EPS”) reflects the potential dilution that could occur if the earnings were divided by the weighted-average number of common shares and potentially dilutive common shares from outstanding stock options, unvested restricted stock units and the 3.25% Convertible Senior Notes due 2020 (“Convertible Senior Notes”). Potentially dilutive common shares from outstanding stock options and unvested restricted stock units are determined using the average share price for each period under the treasury stock method.
Comprehensive Loss—Comprehensive loss includes unrealized gains and losses which are related to the cumulative translation adjustments excluded from the Company’s Consolidated Statements of Operations.
Use of Estimates—The preparation of financial statements in conformity with accounting principles generally accepted in the U.S. requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosures of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from those estimates.
Accounting Periods—Each of the Company’s fiscal quarters end on the Sunday closest to the end of the calendar quarter. The Company’s fiscal year ends are December 31, 2017, January 1, 2017 and January 3, 2016. For ease of reference, the calendar quarter end dates are used herein.
Recent Accounting Pronouncements—In May 2014, the Financial Accounting Standards Board (“FASB”) issued guidance codified in Accounting Standards Update (“ASU”) 2014-09, Revenue from Contracts with Customers, which amends the guidance in former ASC 605, Revenue Recognition ("ASC 605"). The standard’s core principle is that a company will recognize revenue when it transfers promised goods or services to customers in an amount that reflects the consideration to which the company expects to be entitled in exchange for those goods or services. In doing so, companies will need to use more judgment and make more estimates than under current authoritative guidance. These may include identifying performance obligations in the contract, estimating the amount of variable consideration to include in the transaction price and allocating the transaction price to each separate performance obligation. The FASB has issued several amendments to the new standard, which include clarification of accounting guidance related to identification of performance obligations, intellectual property licenses, and principal vs. agent considerations. ASU 2014-09 and all subsequent amendments (collectively, the “ASC 606”) will be effective for public entities for annual reporting periods beginning after December 15, 2017, including interim periods therein.
The Company will adopt ASC 606 on January 1, 2018, using the modified retrospective transition method applied to those contracts which were not completed as of that date. Our evaluation of our contracts subject to this standard is substantially complete and based on procedures completed to date, we do not expect the application of the new standard to these contracts to have a material impact to our Consolidated Statements of Operations or Balance Sheets at initial implementation. We are also evaluating the new disclosures required by the standard to determine what additional information will need to be disclosed.
In February 2016, the FASB issued guidance codified in ASU 2016-02 (Topic 842), Leases. The guidance requires a lessee to recognize a lease liability for the obligation to make lease payments and a right-to-use asset representing the right to use the underlying asset for the lease term on the balance sheet. The guidance is effective for fiscal years beginning after December 15, 2018 including interim periods within those years. The Company is currently evaluating the impact of this guidance and expects to adopt the standard in the first quarter of 2019.
In January 2017, the FASB issued guidance codified in ASU 2017-04, Intangibles-Goodwill and Other (Topic 350) Simplifying the Test for Goodwill Impairment (“ASU 2017-04”). Under this new guidance, an entity will no longer determine goodwill impairment by calculating the implied fair value of goodwill by assigning the fair value of a reporting unit to all of its assets and liabilities as if that reporting unit had been acquired in a business combination. Instead, an entity will compare the fair value of a reporting unit with its carrying amount and recognize an impairment charge for the amount by which the carrying amount exceeds the reporting unit's fair value. The guidance is effective for fiscal years beginning after December 15, 2019 including interim periods therein. The Company is currently evaluating the impact of this guidance and expects to adopt the standard in the first quarter of 2020.
In March 2016, the FASB issued guidance codified in ASU 2016-09 (Topic 718), Improvements to Employee Share-Based Payment Accounting (“ASU 2016-09”). This guidance includes provisions to simplify several aspects of accounting for share-
59
based payment transactions, including income tax consequences, accounting for forfeitures, classification of awards as either equity or liability and classification on the statement of cash flows. ASU 2016-09 includes a requirement that the tax effect related to the settlement of share-based awards be recorded within income tax expense or benefit in the income statement. The simplification of income tax accounting for share-based payment transactions also impacts the computation of weighted-average diluted shares outstanding under the treasury stock method. The Company adopted ASU 2016-09 in the first quarter of 2017 and the impact of the adoption resulted in the following:
• | Upon adoption, the balance of the unrecognized excess tax benefits of $1.8 million was recorded as an increase to deferred tax assets and a corresponding increase to the valuation allowance, resulting in no impact to retained earnings. |
• | Excess tax benefits from share-based arrangements are to be classified within cash flow from operating activities, rather than as cash flow from financing activities. The Company applied this provision on a retrospective basis and the prior period statement of cash flows was adjusted. This adoption did not have a material impact on the Company’s cash flows. |
• | The Company elected to continue to estimate the number of awards expected to be forfeited and adjust the estimate when appropriate, as is currently required. This adoption did not have a material impact on the Company’s consolidated results of operations, financial condition or cash flows. |
• | There was no material impact on the computation of weighted-average diluted shares outstanding. |
Note 2. Balance Sheet Account Details
Inventories
Inventories consisted of the following, net of immaterial excess and obsolete reserves, at December 31, 2017 and 2016 (in thousands):
December 31, | |||||||
2017 | 2016 | ||||||
Raw materials | $ | 22,252 | $ | 9,297 | |||
Work-in-process (materials, labor and overhead) | 22,813 | 7,990 | |||||
Finished goods (materials, labor and overhead) | 22,013 | 8,758 | |||||
Total inventories | $ | 67,078 | $ | 26,045 | |||
Property, Plant and Equipment
The following is a summary of property, plant and equipment (in thousands):
December 31, | |||||||
2017 | 2016 | ||||||
Equipment, furniture and fixtures | $ | 75,728 | $ | 61,972 | |||
Building and improvements | 34,994 | 34,243 | |||||
Leased instruments | 32,458 | 24,014 | |||||
Land | 1,080 | 1,080 | |||||
Total property, plant and equipment, gross | 144,260 | 121,309 | |||||
Less: accumulated depreciation and amortization | (82,675 | ) | (70,451 | ) | |||
Total property, plant and equipment, net | $ | 61,585 | $ | 50,858 | |||
The equipment, furniture and fixtures category above includes construction in progress and instruments that have not been placed at a customer under a lease agreement. These items will be reclassified when the assets are placed in service. The total expense for depreciation of fixed assets and amortization of leasehold improvements was $14.6 million, $13.4 million and $12.7 million for the years ended December 31, 2017, 2016 and 2015, respectively. Maintenance and minor repairs are charged to operations as incurred.
Goodwill and Intangible Assets
The Company had goodwill of $337.0 million as of December 31, 2017, which increased by $253.2 million as compared to $83.8 million as of December 31, 2016 due to the Company's acquisition of the cardiovascular and toxicology Triage® MeterPro business (“Triage Business”) and the Triage® BNP Test for the Beckman Coulter Access Family of Immunoassay
60
Systems business (the “BNP Business”). The Triage Business and the BNP Business are collectively referred to as the “Triage and BNP Businesses.”
On October 6, 2017, the Company acquired the Triage and BNP Businesses from Alere Inc. ("Alere"). As part of this acquisition, the Company identified and recorded $115.0 million in customer relationships, $52.4 million in purchased technology and $17.5 million in trademarks. Refer to Note 12 for additional details regarding the acquisition and the purchase price allocation.
On May 16, 2017, the Company acquired the InflammaDry® and AdenoPlus® diagnostic businesses from RPS
Diagnostics, a developer and manufacturer of rapid, point-of-care (“POC”) diagnostic tests for the eye health and
primary care markets, for approximately $13.7 million in cash. The purchase price has been allocated as follows:
$6.1 million to purchased technology and $7.6 million to goodwill. The acquisition has been accounted for in conformity with
ASC Topic 805, Business Combinations. The InflammaDry and AdenoPlus products are rapid, lateral-flow based, POC
products for the detection of infectious and inflammatory diseases and conditions of the eye. Revenues for these products are
reflected in the Company’s Rapid Immunoassay revenue category.
Amortizable intangible assets consisted of the following (dollar amounts in thousands):
December 31, 2017 | December 31, 2016 | |||||||||||||||||||
Description | Weighted-average useful life (years) | Gross assets | Accumulated amortization | Net | Gross assets | Accumulated amortization | Net | |||||||||||||
Purchased technology | 9.1 | 112,100 | (49,614 | ) | 62,486 | 53,600 | (41,369 | ) | 12,231 | |||||||||||
Customer relationships | 7.0 | 122,404 | (10,960 | ) | 111,444 | 7,157 | (5,928 | ) | 1,229 | |||||||||||
License agreements | 9.9 | 6,515 | (3,980 | ) | 2,535 | 6,009 | (3,222 | ) | 2,787 | |||||||||||
Patent and trademark costs | 10.8 | 28,740 | (4,917 | ) | 23,823 | 11,240 | (3,522 | ) | 7,718 | |||||||||||
Software development costs | 5 | 6,630 | (3,091 | ) | 3,539 | 6,000 | (2,326 | ) | 3,674 | |||||||||||
Total amortizable intangible assets | $ | 276,389 | $ | (72,562 | ) | $ | 203,827 | $ | 84,006 | $ | (56,367 | ) | $ | 27,639 | ||||||
Amortization expense related to the capitalized software costs was $0.8 million, $0.5 million and $0.6 million for the years ended December 31, 2017, 2016 and 2015, respectively. Amortization expense (including capitalized software costs) was $16.1 million, $9.5 million and $10.2 million for the years ended December 31, 2017, 2016 and 2015, respectively.
The expected future annual amortization expense of the Company’s intangible assets is as follows (in thousands):
For the years ending December 31, | Amortization expense | |||
2018 | $ | 28,799 | ||
2019 | 27,542 | |||
2020 | 27,144 | |||
2021 | 26,996 | |||
2022 | 26,472 | |||
Thereafter | 66,874 | |||
Total | $ | 203,827 | ||
61
Other current liabilities
The following is a summary of other current liabilities (in thousands):
December 31, | |||||||
2017 | 2016 | ||||||
Customer incentives | $ | 7,165 | $ | 3,766 | |||
Accrued interest | 442 | 227 | |||||
Other | 5,059 | 1,104 | |||||
Total other current liabilities | $ | 12,666 | $ | 5,097 | |||
Note 3. Debt
3.25% Convertible Senior Notes due 2020
In December 2014, the Company issued $172.5 million aggregate principal amount of 3.25% Convertible Senior Notes due 2020. Debt issuance costs of approximately $5.1 million were primarily comprised of underwriters fees, legal, accounting, and other professional fees of which $4.2 million were capitalized and are recorded as a reduction to long-term debt and are being amortized using the effective interest method to interest expense over the six-year term of the Convertible Senior Notes. The remaining $0.9 million of debt issuance costs were allocated as a component of equity in additional paid-in capital. Deferred issuance costs related to the Convertible Senior Notes were $2.1 million and $2.8 million as of December 31, 2017 and 2016, respectively.
The holders of the Convertible Senior Notes may surrender their notes for conversion, subject to specified circumstances, into cash, shares of common stock, or a combination of cash and shares of common stock, at the election of the Company, based on an initial conversion rate, subject to adjustment, of 31.1891 shares per $1,000 principal amount of the Convertible Senior Notes (which represents an initial conversion price of approximately $32.06 per share) up until the business day immediately preceding September 15, 2020. This conversion may, in the discretion of the holder, occur in the following circumstances and to the following extent: (1) during any calendar quarter commencing after the calendar quarter ending on March 31, 2015, if the last reported sales price of the Company's common stock, for at least 20 trading days (whether or not consecutive) in the period of 30 consecutive trading days ending on the last trading day of the calendar quarter immediately preceding the calendar quarter in which the conversion occurs, is more than 130% of the conversion price of the notes in effect on each applicable trading day; (2) during the five consecutive business day period following any five consecutive trading day period in which the trading price per $1,000 principal amount of the Convertible Senior Note for each such trading day was less than 98% of the product of the last reported sale price of the Company's common stock and the conversion rate on each such day; or (3) upon the occurrence of specified events described in the indenture for the Convertible Senior Notes. On or after September 15, 2020 until the close of business on the second scheduled trading day immediately preceding the stated maturity date, holders may surrender their notes for conversion at any time, regardless of the foregoing circumstances.
In general, for each $1,000 in principal, the “principal portion” of cash upon settlement is defined as the lesser of $1,000, or the conversion value during the 25-day observation period as described in the indenture for the Convertible Senior Notes. The conversion value is the sum of the daily conversion value, which is the product of the effective conversion rate divided by 25 days and the daily volume weighted-average price (“VWAP”) of the Company’s common stock. The “share amount” is the cumulative “daily share amount” during the observation period, which is calculated by dividing the daily VWAP into the difference between the daily conversion value (i.e., conversion rate x daily VWAP) and $1,000.
The Company pays 3.25% interest per annum on the principal amount of the Convertible Senior Notes semi-annually in arrears in cash on June 15 and December 15 of each year. The effective interest rate during fiscal year 2017 was 6.6%. The Convertible Senior Notes mature on December 15, 2020. During the year ended December 31, 2017, the Company recorded total interest expense of $11.0 million related to the Convertible Senior Notes of which $5.5 million related to the amortization of the debt discount and issuance costs and $5.4 million related to the coupon due semi-annually. During the year ended December 31, 2016, the Company recorded total interest expense of $10.9 million related to the Convertible Senior Notes of which $5.4 million related to the amortization of the debt discount and issuance costs and $5.5 million related to the coupon due semi-annually. During the year ended December 31, 2015, the Company recorded total interest expense of $10.9 million related to the Convertible Senior Notes of which $5.3 million related to the amortization of the debt discount and issuance costs and $5.6 million related to the coupon due semi-annually.
62
If a fundamental change, as defined in the indenture for the Convertible Senior Notes, such as certain acquisitions, mergers, or a liquidation of the Company, occurs prior to the maturity date, subject to certain limitations, holders of the Convertible Senior Notes may require the Company to repurchase all or a portion of their Convertible Senior Notes for cash at a repurchase price equal to 100% of the principal amount of the Convertible Senior Notes to be repurchased, plus any accrued and unpaid interest to, but excluding, the repurchase date.
The Company accounts separately for the liability and equity components of the Convertible Senior Notes in accordance with authoritative guidance for convertible debt instruments that may be settled in cash upon conversion. The guidance requires the carrying amount of the liability component to be estimated by measuring the fair value of a similar liability that does not have an associated conversion feature. Because the Company had no outstanding non-convertible public debt, the Company determined that senior, unsecured corporate bonds traded on the market represent a similar liability to the Convertible Senior Notes without the conversion option. Based on market data available for publicly traded, senior, unsecured corporate bonds issued by companies in the same industry with similar credit ratings and with similar maturity, the Company estimated the implied interest rate of its Convertible Senior Notes to be 6.9%, assuming no conversion option. Assumptions used in the estimate represent what market participants would use in pricing the liability component, which were defined as Level 2 observable inputs. The estimated implied interest rate was applied to the Convertible Senior Notes, which resulted in a fair value of the liability component of $141.9 million upon issuance, calculated as the present value of implied future payments based on the $172.5 million aggregate principal amount. The $30.7 million difference between the cash proceeds of $172.5 million and the estimated fair value of the liability component was recorded in additional paid-in capital, net of tax and issuance costs, as the Convertible Senior Notes were not considered redeemable.
In the first quarter of 2016, the Company repurchased and retired $5.2 million in principal amount of the outstanding Convertible Senior Notes. The aggregate cash used for the transaction was $4.5 million. The repurchase resulted in a reduction in debt of $4.4 million and a reduction in additional paid-in capital of $0.5 million with a gain on extinguishment of Convertible Senior Notes of $0.4 million included in interest expense, net in the Consolidated Statements of Operations.
The following table summarizes information about the equity and liability components of the Convertible Senior Notes (dollars in thousands). The fair values of the respective notes outstanding were measured based on quoted market prices:
December 31, | |||||||
2017 | 2016 | ||||||
Principal amount of Convertible Senior Notes outstanding | $ | 167,314 | $ | 167,314 | |||
Unamortized discount of liability component | (15,356 | ) | (20,221 | ) | |||
Unamortized deferred issuance costs | (2,090 | ) | (2,753 | ) | |||
Net carrying amount of liability component | 149,868 | 144,340 | |||||
Less: current portion | — | — | |||||
Long-term debt | $ | 149,868 | $ | 144,340 | |||
Carrying value of equity component, net of issuance costs | $ | 29,211 | $ | 29,211 | |||
Fair value of outstanding Convertible Senior Notes | $ | 257,245 | $ | 165,223 | |||
Remaining amortization period of discount on the liability component | 3 years | 4 years | |||||
Historically, the Company disclosed its intent, ability and policy to settle conversions of its Senior Convertible Notes through combination settlement, which essentially involves repayment of an amount of cash equal to the “principal portion” and delivery of the “share amount” in excess of the principal portion in shares of common stock or cash. This would have resulted in the “share amount” impacting diluted EPS. Based upon the use of funds to acquire the Triage and BNP Businesses, the Company has determined that it currently does not have the available funds to settle the principal in cash and as a result, may choose to settle both the principal and share amount using equity. As a result, both the “principal portion” and “share portion” will impact diluted EPS. This change in assessment had no impact on diluted EPS because the Company incurred a net loss for the year ended December 31, 2017. As of December 31, 2017, the if-converted value exceeded the principal amount of the Convertible Senior Notes by $1.5 million.
As of December 31, 2016 and 2015, the Convertible Senior Notes were not convertible and the if-converted value did not exceed the principal amount of the Convertible Senior Notes, therefore, there was no dilutive impact for the years ended December 31, 2016 and 2015.
63
Senior Credit Agreement
On October 6, 2017, the Company entered into a Credit Agreement (the “Credit Agreement”), which provided the Company with a $245.0 million senior secured term loan facility (the “Term Loan”) and a $25.0 million Revolving Credit Facility ("Revolving Credit Facility") together (the “Senior Credit Facility”). The Term Loan and the Revolving Credit Facility will mature on October 6, 2022. On the closing date of the Credit Agreement, the Company borrowed the entire amount of the Term Loan and $10.0 million under the Revolving Credit Facility. The Company used the proceeds of the Term Loan along with its cash on hand, to pay (i) the consideration for the Triage Business and (ii) the fees and expenses incurred in connection with the acquisition of the Triage and BNP Businesses.
The Credit Agreement includes an accordion feature that allows the facility to be increased by $50.0 million upon the satisfaction of certain conditions. The Financing is guaranteed by certain material domestic subsidiaries of the Company (the “Guarantors”) and is secured by liens on substantially all of the assets of the Company and the Guarantors, excluding real property and certain other types of excluded assets.
Loans under the Credit Agreement bear interest at a rate equal to (i) the London Interbank Offered Rate (“LIBOR”) plus the “applicable rate” or (ii) the “base rate” (defined as the highest of (a) the Bank of America prime rate, (b) the Federal Funds rate plus one-half of one percent and (c) LIBOR plus one percent) plus the “applicable rate.” The initial applicable rate is 2.50% per annum for base rate loans and 3.50% per annum for LIBOR rate loans, and thereafter will be determined in accordance with a pricing grid based on the Company’s Consolidated Leverage Ratio (as defined in the Credit Agreement) ranging from 2.50% to 3.50% per annum for LIBOR rate loans and from 1.50% to 2.50% per annum for base rate loans. In addition, the Company will pay a commitment fee on the unused portion of the Credit Agreement based on the Company’s Consolidated Leverage Ratio ranging from 0.10% to 0.50% per annum.
The Term Loan is subject to quarterly amortization of the principal amount on the last business day of each fiscal quarter of the Company (commencing on March 30, 2018) in such amounts as are set forth in the Credit Agreement. The Term Loan and the Revolving Credit Facility will mature on October 6, 2022, provided that if any of Convertible Senior Notes remain outstanding on the date that is 91 days prior to the maturity date of the Convertible Senior Notes and the Company has not satisfied certain Refinancing Conditions, then the maturity date for the Term Loan and the Revolving Credit Facility will be the date that is 91 days prior to the maturity date of the Convertible Senior Notes.
The Company must prepay loans outstanding under the Credit Agreement in an amount equal to 50% of Excess Cash Flow (as defined in the Credit Agreement) for each fiscal year (commencing with fiscal 2018) less any amount voluntarily prepaid during such fiscal year, but only if the Consolidated Senior Secured Leverage Ratio (as defined in the Credit Agreement) as of the last day of such fiscal year is greater than or equal to 1.25 to 1.00. The Company must also prepay loans outstanding under the Credit Agreement in an amount equal to 100% of the Net Cash Proceeds (as defined in the Credit Agreement) from (i) certain property dispositions and (ii) the receipt of certain other amounts not in the ordinary course of business, in each case, if not reinvested within a specified time period as contemplated in the Credit Agreement, and with a carve out of up to 30% of the Net Cash Proceeds of the contemplated sale leaseback transaction relating to the Company’s Summers Ridge property to the extent the excluded amounts are used for specified purposes.
The Credit Agreement contains affirmative and negative covenants that are customary for credit agreements of this nature. The negative covenants include, among other things, limitations on asset sales, mergers, indebtedness, liens, investments and transactions with affiliates. The Credit Agreement contains two financial covenants: (i) a maximum Consolidated Leverage Ratio as of the last day of each fiscal quarter for the most recently completed four fiscal quarters of (a) 5.00 to 1.00 for the fiscal quarter ending December 31, 2017, (b) 4.25 to 1.00 for the fiscal quarters ending March 31, 2018 through December 31, 2018 and (c) 3.50 to 1.00 for the fiscal quarter ending March 31, 2019 and each fiscal quarter thereafter; and (ii) a minimum Consolidated Fixed Charge Coverage Ratio of 1.25 to 1.00 as of the end of any fiscal quarter for the most recently completed four fiscal quarters. The Company was in compliance with all financial covenants as of December 31, 2017.
64
The Term Loan consists of the following (dollars in thousands):
December 31, 2017 | |||
Principal balance | $ | 245,000 | |
Unamortized deferred issuance costs | (7,422 | ) | |
Term Loan, net | 237,578 | ||
Less: current portion | (10,184 | ) | |
Term Loan, non-current | $ | 227,394 | |
As of December 31, 2017, the aggregate contractual maturities of long-term borrowings for the Term Loan are as follows (dollars in thousands):
Fiscal year: | |||
2018 | $ | 12,250 | |
2019 | 18,375 | ||
2020 | 24,500 | ||
2021 | 24,500 | ||
2022 | 165,375 | ||
Total | $ | 245,000 | |
Interest expense recognized on the Term Loan for the year ended December 31, 2017 totaled $3.0 million for the stated interest. Amortization of debt issuance costs associated with the Term Loan was $0.5 million for the year ended December 31, 2017, and was recorded to interest expense in the Company's Consolidated Statement of Operations.
Interest expense recognized on the Revolving Credit Facility for the year ended December 31, 2017 totaled $0.1 million for the stated interest and unused commitment fee. Amortization of debt issuance costs associated with the Revolving Credit Facility was $40,000 for the year ended December 31, 2017, and was recorded to interest expense in the Company's Consolidated Statement of Operations.
Note 4. Income Taxes
Significant components of the provision (benefit) for income taxes are as follows (in thousands):
December 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Current: | |||||||||||
Federal | $ | (615 | ) | $ | (117 | ) | $ | 948 | |||
State | 314 | 246 | 399 | ||||||||
Foreign | 57 | 84 | 41 | ||||||||
Total current (benefit) provision | (244 | ) | 213 | 1,388 | |||||||
Deferred: | |||||||||||
Federal | 131 | (2,545 | ) | (4,624 | ) | ||||||
State | 238 | (63 | ) | — | |||||||
Foreign | 4 | 4 | 18 | ||||||||
Total deferred benefit | 373 | (2,604 | ) | (4,606 | ) | ||||||
Provision (benefit) for income taxes | $ | 129 | $ | (2,391 | ) | $ | (3,218 | ) | |||
65
The Company’s (loss) before provision (benefit) for income taxes was subject to taxes in the following jurisdictions for the following periods (in thousands):
December 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
United States | $ | (8,198 | ) | $ | (16,426 | ) | $ | (9,480 | ) | ||
Foreign | 162 | 227 | 183 | ||||||||
Loss before benefit for income taxes | $ | (8,036 | ) | $ | (16,199 | ) | $ | (9,297 | ) | ||
Significant components of the Company’s deferred tax assets and deferred tax liabilities as of December 31, 2017 and 2016 are shown below (in thousands):
December 31, | |||||||
2017 | 2016 | ||||||
Deferred tax assets: | |||||||
Net operating loss carryforwards | $ | 3,924 | $ | 3,255 | |||
Intangible assets | 2,935 | 2,351 | |||||
Sale-leaseback, net | 628 | 888 | |||||
Allowance for returns and discounts | 5,358 | 4,043 | |||||
Stock-based compensation | 5,933 | 10,963 | |||||
Tax credit carryforwards | 5,247 | 3,430 | |||||
Other, net | 4,580 | 4,066 | |||||
Total deferred tax assets | 28,605 | 28,996 | |||||
Valuation allowance for deferred tax assets | (15,204 | ) | (7,774 | ) | |||
Total deferred tax assets, net of valuation allowance | 13,401 | 21,222 | |||||
Deferred tax liabilities: | |||||||
Convertible Senior Notes | (3,633 | ) | (7,592 | ) | |||
Intangible assets | (2,935 | ) | (7,557 | ) | |||
Property, plant and equipment | (7,263 | ) | (6,131 | ) | |||
Total deferred tax liabilities | (13,831 | ) | (21,280 | ) | |||
Net deferred tax liabilities | $ | (430 | ) | $ | (58 | ) | |
Management assesses the available positive and negative evidence to estimate if sufficient future taxable income will be generated to use the existing deferred tax assets. A significant piece of objective negative evidence evaluated was the cumulative before-tax loss incurred over the three-year periods ended December 31, 2017 and 2016. Such objective evidence limits the ability to consider other subjective evidence such as the Company's projections for future profitability. On the basis of this evaluation, as of December 31, 2017, the Company had recorded a valuation allowance of $15.2 million, which represents the portion of the deferred tax asset that management could no longer conclude was more likely than not to be realized. The amount of the deferred tax assets considered realizable, however, could be adjusted in the future based on changes in available evidence and additional weight may be given to subjective evidence such as the Company's projections for profitability. During the year ended December 31, 2017, the allowance increased by $7.4 million.
As of December 31, 2017, the Company had federal net operating loss ("NOL") carryforwards of approximately $13.0 million which will begin to expire in 2018, unless previously utilized. The Company also had state NOLs of approximately $23.3 million which will begin to expire in 2026, unless previously utilized. The Company has federal research credits of $4.3 million which will begin to expire on December 31, 2031, unless previously utilized. Additionally, the Company has federal alternative minimum tax credits of $0.6 million which have been reclassified to a tax receivable based on the new federal tax legislation. The Company has state research credits of $9.8 million, of which $9.5 million do not expire. The remaining $0.3 million begin to expire in 2028, unless previously utilized.
Pursuant to Internal Revenue Code Sections 382 and 383, the Company’s use of its NOL and research credit carryforwards may be limited as a result of cumulative changes in ownership of more than 50% over a three-year period.
66
The provision (benefit) for income taxes reconciles to the amount computed by applying the federal statutory rate to loss before taxes as follows (in thousands):
Year ended December 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Tax benefit at statutory tax rate | $ | (2,812 | ) | $ | (5,775 | ) | $ | (3,254 | ) | ||
State tax benefits, net of federal tax benefit | (239 | ) | (390 | ) | (235 | ) | |||||
Permanent differences | 327 | 129 | 157 | ||||||||
Federal and state research credits—current year | (484 | ) | (979 | ) | (722 | ) | |||||
Accrual of uncertain tax positions | 142 | 43 | 101 | ||||||||
Stock-based compensation | (5,851 | ) | — | — | |||||||
Impact of change in federal and state tax rate on revaluing deferred tax assets | 3,357 | (4 | ) | 56 | |||||||
Change in valuation allowance | 5,799 | 4,687 | 756 | ||||||||
Other | (110 | ) | (102 | ) | (77 | ) | |||||
Provision (benefit) for income taxes | $ | 129 | $ | (2,391 | ) | $ | (3,218 | ) | |||
On December 22, 2017, the Tax Cuts and Jobs Act (the "Tax Act") was enacted into legislation, which includes a broad range of provisions affecting businesses. The Tax Act significantly revises how companies compute their U.S corporate tax liability by, among other provisions, reducing the corporate tax rate from 35% to 21% for tax years beginning after December 31, 2017, implementing a territorial tax system, and requiring a mandatory one-time tax on U.S. owned undistributed foreign earnings and profits known as the transition tax.
Pursuant to the SEC Staff Accounting Bulletin No. 118, Income Tax Accounting Implications of the Tax Cuts and Jobs Act ("SAB 118"), a company may select between one of three scenarios to determine a reasonable estimate arising from the Tax Act. Those scenarios are (i) a final estimate which effectively closes the measurement window; (ii) a reasonable estimate leaving the measurement window open for future revisions; and (iii) no estimate, as the law is still being analyzed. The Company was able to provide a reasonable estimate for the revaluation of deferred taxes and the effects of the transition tax on undistributed foreign earnings and profits. As such, the Company has recorded a reduction in deferred tax assets for the revaluation of deferred taxes and a reduction of the net operating losses for the impacts of the transition tax, with both reductions offset by a decrease in the Company's full valuation allowance.
As of December 31, 2017, the Company had $1.0 million of undistributed foreign earnings and profits. Pursuant to the Tax Act, the Company’s undistributed foreign earnings and profits were deemed repatriated as of December 31, 2017. As a result, the Company utilized net operating losses and generated foreign tax credits, each offset by the Company’s change in valuation allowance. Upon the distribution of foreign earnings and profits, certain foreign countries impose withholding taxes. The Company did not provide for foreign withholding taxes on the undistributed earnings and profits from certain non-U.S. subsidiaries that will be permanently reinvested outside the United States. If the foreign earnings and profits were distributed, the Company would need to accrue an additional income tax liability, potentially generating foreign tax credits for use against the Company’s U.S. tax liability, subject to certain limitations.
The Company recognizes liabilities for uncertain tax positions based on a two-step process. The first step is to evaluate the tax position for recognition by determining if the weight of available evidence indicates that it is more likely than not that the position will be sustained on audit, including resolution of related appeals or litigation processes, if any. The second step is to measure the tax benefit as the largest amount that is more than 50% likely of being realized upon settlement. While the Company believes that it has appropriate support for the positions taken on its tax returns, the Company regularly assesses the potential outcome of examinations by tax authorities in determining the adequacy of its provision for income taxes.
67
The following table summarizes the activity related to the Company’s unrecognized tax benefits (in thousands):
Year ended December 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Beginning balance | $ | 8,604 | $ | 7,684 | $ | 7,065 | |||||
Increases (decreases) related to prior year tax positions | 10 | (10 | ) | (12 | ) | ||||||
Increases related to current year tax positions | 951 | 773 | 631 | ||||||||
Other | $ | — | $ | 157 | $ | — | |||||
Ending balance | $ | 9,565 | $ | 8,604 | $ | 7,684 | |||||
As of December 31, 2017 and 2016, the unrecognized tax benefits of $9.6 million and $8.6 million, respectively, of which $8.1 million and $6.4 million, respectively, would reduce the Company’s annual effective tax rate, subject to the valuation allowance. The Company does not anticipate any significant decreases in its unrecognized tax benefits over the next 12 months. The Company's policy is to recognize the interest expense and penalties related to income tax matters as a component of income tax expense. The Company has accrued approximately $0.3 million and $0.2 million of interest and penalties associated with uncertain tax positions for each of the years ended December 31, 2017 and 2016 respectively. There was no interest expense, net of accrued interest (reversed) in 2016. Interest expense, net of accrued interest (reversed) was approximately $0.1 million for both the years ended December 31, 2017 and 2015, respectively.
The Company is subject to periodic audits by domestic and foreign tax authorities; however there are no known audits at this time.
Due to the carryforward of unutilized net operating loss and credit carryovers, the Company's federal tax years from 2009 and forward and state tax years 2001 and forward are subject to examination by tax authorities.
The Company believes that it has appropriate support for the income tax positions taken on its tax returns and that its accruals for tax liabilities are adequate for all open years based on an assessment of many factors, including past experience and interpretations of tax law applied to the facts of each matter.
Note 5. Stockholders’ Equity
Preferred Stock. The Company’s certificate of incorporation, as amended, authorizes the issuance of up to five million preferred shares. The Board of Directors is authorized to fix the number of shares of any series of preferred stock and to determine the designation of such shares. However, the amended certificate of incorporation specifies the initial series and the rights of that series. No shares of preferred stock were outstanding as of December 31, 2017, 2016 or 2015.
Equity Incentive Plan. The Company grants stock options, time-based restricted stock units ("RSUs") and performance-based restricted stock units ("PSUs") to employees and non-employee directors under its 2016 Equity Incentive Plan (the “2016 Plan”). The Company previously granted stock options under the Amended and Restated 2010 Equity Incentive Plan (the "2010 Plan") and the Amended and Restated 2001 Equity Incentive Plan (the "2001 Plan"). The 2010 Plan and 2001 Plan were terminated at the time of adoption of the 2016 Plan, but the terminated Plans continue to govern outstanding options granted thereunder. The Company has stock options, RSUs and PSUs outstanding, which were issued under each of these equity incentive plans to certain employees and directors. Stock options granted under these plans have terms ranging up to ten years, have exercise prices ranging from $8.50 to $41.36 per share, and generally vest over four years. As of December 31, 2017, approximately 1.8 million shares remained available for grant under the 2016 Plan.
Restricted Stock Units. The Company grants both RSUs and PSUs to certain officers, directors and management. Until the restrictions lapse, ownership of the affected restricted stock units granted to the Company’s officers, directors and management is conditional upon continuous employment with the Company.
For the years ended December 31, 2017, 2016 and 2015, the Company granted approximately 0.3 million, 0.2 million and 0.2 million shares, respectively, of restricted stock units to officers and management, which either have a time-based four-year vesting provision or performance-based vesting provisions. For purposes of measuring compensation expense of PSUs, the amount of shares ultimately expected to vest is estimated at each reporting date based on management’s expectations regarding the relevant performance criteria. The grant date of the PSUs takes place when the grant is authorized and the specific achievement goals are communicated. The recognition of compensation expense associated with the PSUs requires judgment in assessing the probability of meeting the performance goals. This may result in significant expense recognition in the period in
68
which the performance goals are met or when achievement of the goals is deemed probable or may result in the reversal of previously recognized stock-based compensation expense if the performance criteria are deemed not probable of being met.
During the year ended December 31, 2017, common stock was issued to certain members of the Board of Directors in lieu of cash compensation for these members that elected to participate and agree to hold the stock for the elected deferral period. During the years ended December 31, 2016 and 2015, RSUs were granted to certain members of the Board of Directors in lieu of cash compensation as a part of the Company’s non-employee director’s deferred compensation program. The compensation expense associated with these RSU grants were $0.1 million, $0.4 million and $0.5 million for the years ended December 31, 2017, 2016 and 2015, respectively.
Employee Deferred Bonus Compensation Program. For the year ended December 31, 2017, the deferred bonus compensation program was suspended temporarily by the Board. For the years ended December 31, 2016 and 2015, certain employees of the Company were eligible to participate in the Company’s deferred bonus compensation program with respect to any payments received under the Company’s cash incentive plan. Participating employees could elect to receive 50% or 100% of the cash value of their cash bonus in the form of fully vested, restricted stock units plus an additional premium as additional restricted stock units, issued under the 2016 Plan. The premium restricted stock units are subject to a one-year vesting requirement from the date of issuance. The additional premium will be determined based on the length of time of the deferral period selected by the participating employee as follows: (i) if one year from the date of grant, a premium of 10% on the amount deferred, (ii) if two years from the date of grant, a premium of 20% on the amount deferred, or (iii) if four years from the date of grant, a premium of 30% on the amount deferred.
Employee Stock Purchase Plan. Under the Company’s Amended and Restated 1983 Employee Stock Purchase Plan (the “ESPP”), full-time employees are allowed to purchase common stock through payroll deductions (which cannot exceed 10% of the employee’s compensation) at the lower of 85% of fair market value at the beginning or end of each six-month purchase period. As of December 31, 2017, 1,272,274 shares had been sold under the Plan, leaving 227,726 shares available for future issuance.
Share Repurchase Program. On January 25, 2016, the Company's Board of Directors authorized an amendment to extend the previously announced stock repurchase program. The Board of Directors has authorized the Company to repurchase up to an aggregate of $50.0 million in shares of our common stock under our stock repurchase program. Any shares of common stock repurchased under this program will no longer be deemed outstanding upon repurchase and will be returned to the pool of authorized shares. During the year ended December 31, 2016, 1,152,386 shares of outstanding common stock were repurchased under the Company's previously announced share repurchase program for approximately $19.6 million. During the year ended December 31, 2015, 1,397,000 shares of outstanding common stock were repurchased under the Company's previously announced share repurchase program for approximately $30.4 million. There were no repurchases during 2017 and at December 31, 2017, $35.0 million remained available under this program. The repurchase program expired on January 25, 2018.
Shares Reserved for Future Issuance. At December 31, 2017, approximately 5.3 million shares of common stock were reserved under the Company’s equity incentive plan and 227,726 shares were reserved for purchases under the ESPP.
Note 6. Stock-Based Compensation
For the years ended December 31, 2017, 2016 and 2015 stock-based compensation expense was $9.1 million, $8.0 million and $7.4 million, respectively, of which $4.1 million, $4.7 million and $4.7 million, respectively, related to stock options and $4.9 million, $2.4 million and $2.0 million, respectively, related to restricted stock units. For the years ended December 31, 2017, 2016 and 2015, the Company recorded $0.1 million, $0.9 million and $0.7 million in stock-based compensation expense, respectively, associated with the deferred bonus compensation program, described in Note 5. During the years ended December 31, 2016 and 2015, $0.9 million and $0.6 million, respectively, was initially recorded as a component of accrued payroll and related expenses. Since the employee Deferred Bonus Compensation Program was suspended in 2017, there was no component of stock-based compensation recorded to accrued payroll related expenses as of December 31, 2017.
69
Stock-based compensation expense related to stock options and restricted stock units was as follows (in thousands):
Year ended December 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Cost of sales | $ | 579 | $ | 617 | $ | 581 | |||||
Research and development | 1,886 | 1,551 | 734 | ||||||||
Sales and marketing | 2,129 | 1,189 | 1,554 | ||||||||
General and administrative | 4,467 | 4,629 | 4,550 | ||||||||
Total stock-based compensation expense | $ | 9,061 | $ | 7,986 | $ | 7,419 | |||||
Stock-based compensation expense capitalized to inventory and compensation expense related to the Company’s ESPP were not material for the years ended December 31, 2017, 2016 and 2015.
Stock Options
Compensation expense related to stock options granted is recognized ratably over the service vesting period for the entire option award. For stock options with graded vesting, the Company ensures that the cumulative amount of compensation expense recognized at the end of any reporting period at least equals the portion of the stock option that has vested at that date. The total number of stock options expected to vest is adjusted by estimated forfeiture rates. The estimated fair value of each stock option was determined on the date of grant using the Black-Scholes option valuation model with the following weighted-average assumptions for the option grants:
Year ended December 31, | ||||||||
2017 | 2016 | 2015 | ||||||
Risk-free interest rate | 2.30 | % | 1.47 | % | 1.50 | % | ||
Expected option life (in years) | 6.63 | 6.59 | 6.24 | |||||
Volatility rate | 36 | % | 36 | % | 40 | % | ||
Dividend rate | — | % | — | % | — | % | ||
The computation of the expected option life is based on a weighted-average calculation combining the average life of options that have already been exercised and post-vest cancellations with the estimated life of the remaining vested and unexercised options. The expected volatility is based on the historical volatility of the Company’s stock. The risk-free interest rate is based on the U.S. Treasury yield curve over the expected term of the option. The Company has never paid any cash dividends on its common stock, and does not anticipate paying any cash dividends in the foreseeable future. Consequently, the Company uses an expected dividend yield of zero in the Black-Scholes option valuation model. The Company’s estimated forfeiture rate is based on its historical experience and future expectations.
The Company’s determination of fair value is affected by the Company’s stock price as well as a number of assumptions that require judgment. The weighted-average fair value per share was $8.99, $6.00 and $9.46 for options granted during the years ended December 31, 2017, 2016 and 2015, respectively. The total intrinsic value was $26.8 million, $4.5 million and $1.6 million for options exercised during the years ended December 31, 2017, 2016 and 2015, respectively. As of December 31, 2017, total unrecognized compensation expense related to stock options was approximately $7.7 million and the related weighted-average period over which it is expected to be recognized is approximately 1.9 years. The maximum contractual term of the Company’s stock options is ten years.
70
A summary of the status of stock option activity for the years ended December 31, 2015, 2016 and 2017 is as follows (in thousands, except price data and years):
Number of Shares | Weighted- average exercise price per share | Weighted- average remaining contractual term (in years) | Aggregate intrinsic value | |||||||||
Outstanding at January 1, 2015 | 3,607 | $ | 16.37 | |||||||||
Granted | 659 | 23.15 | ||||||||||
Exercised | (168 | ) | 12.30 | |||||||||
Cancelled | (131 | ) | 23.41 | |||||||||
Outstanding at December 31, 2015 | 3,967 | 17.44 | ||||||||||
Granted | 677 | 15.48 | ||||||||||
Exercised | (553 | ) | 13.76 | |||||||||
Cancelled | (150 | ) | 20.86 | |||||||||
Outstanding at December 31, 2016 | 3,941 | 17.49 | ||||||||||
Granted | 263 | 22.21 | ||||||||||
Exercised | (1,527 | ) | 16.38 | |||||||||
Cancelled | (18 | ) | 24.91 | |||||||||
Outstanding at December 31, 2017 | 2,659 | $ | 18.54 | 5.69 | $ | 65,969 | ||||||
Vested and expected to vest at December 31, 2017 | 2,574 | $ | 18.51 | 5.61 | $ | 63,914 | ||||||
Exercisable at December 31, 2017 | 1,463 | $ | 17.71 | 3.99 | $ | 37,503 | ||||||
Available for future grant at December 31, 2017 | 1,848 | |||||||||||
Restricted Stock Units
The Company grants both time-based restricted stock units ("RSUs") and performance-based restricted stock units ("PSUs"). The fair value of RSUs and PSUs is determined based on the closing market price of the Company’s common stock on the grant date. Compensation expense for RSUs is measured at the grant date and recognized ratably over the vesting period. A portion of the restricted stock granted in 2017 is performance-based and vesting is tied to achievement of specific Company goals over a three year time period, subject to early vesting upon achievement of the performance goals. For purposes of measuring compensation expense for PSUs, the amount of shares ultimately expected to vest is estimated at each reporting date based on management’s expectations regarding the relevant performance criteria. There was no stock-based compensation expense related to PSUs for the years ended December 31, 2016 or 2015.
A summary of the status of stock awards activity for the years ended December 31, 2015, 2016 and 2017 is as follows (in thousands, except price data):
Shares | Weighted-average grant date fair value | |||||
Non-vested at January 1, 2015 | 402 | $ | 14.84 | |||
Granted | 171 | 22.79 | ||||
Vested | (96 | ) | 18.01 | |||
Forfeited | (18 | ) | 22.87 | |||
Non-vested at December 31, 2015 | 459 | 21.61 | ||||
Granted | 185 | 16.14 | ||||
Vested | (120 | ) | 18.50 | |||
Forfeited | (23 | ) | 20.80 | |||
Non-vested at December 31, 2016 | 501 | 20.37 | ||||
Granted | 349 | 22.34 | ||||
Vested | (100 | ) | 23.49 | |||
Forfeited | (4 | ) | 18.69 | |||
Non-vested at December 31, 2017 | 746 | $ | 20.88 | |||
71
In 2017, 2016 and 2015, the Company issued approximately 0.1 million restricted stock units each year in exchange for the deferred bonus liability of $0.9 million, $0.5 million and $0.4 million, respectively.
The total amount of unrecognized compensation expense related to non-vested restricted stock awards as of December 31, 2017 was approximately $13.3 million, which is expected to be recognized over a weighted-average period of approximately 1.8 years.
Note 7. Earnings (Loss) Per Share
The following table reconciles the weighted-average shares used in computing basic and diluted earnings (loss) per share in the respective periods (in thousands):
Twelve months ended December 31, | ||||||||
2017 | 2016 | 2015 | ||||||
Shares used in basic loss per share (weighted-average common shares outstanding) | 33,734 | 32,708 | 34,104 | |||||
Effect of potentially dilutive shares issuable from stock options, RSUs and Convertible Senior Notes | — | — | — | |||||
Shares used in diluted loss per share calculation | 33,734 | 32,708 | 34,104 | |||||
Potentially dilutive shares excluded from calculation due to anti-dilutive effect | 37 | 2,770 | 1,902 | |||||
Potentially dilutive shares excluded from the calculation above represent stock options when the combined exercise price and unrecognized stock-based compensation are greater than the average market price for the Company’s common stock because their effect is anti-dilutive.
Stock options, RSUs and Convertible Senior Notes that would have been included in the diluted EPS calculation if the Company had earnings amounted to 1.4 million for the year ended December 31, 2017. Stock options and RSUs that would have been included in the diluted EPS calculation if the Company had earnings amounted to 0.8 million and 1.0 million for the years ended December 31, 2016 and 2015, respectively. No conversion premium existed on the Convertible Senior Notes as of December 31, 2016 and 2015; therefore, there was no dilutive impact from the Convertible Senior Notes to diluted EPS during the years ended December 31, 2016 and 2015.
Note 8. Commitments and Contingencies
Leases
The Company leases its facilities and certain equipment. Commitments for minimum rentals under non-cancelable leases at the end of 2017 are as follows (in thousands):
Years ending December 31, | Operating Leases | Lease obligation | ||||||
2018 | $ | 8,638 | $ | 946 | ||||
2019 | 9,072 | 956 | ||||||
2020 | 9,212 | 967 | ||||||
2021 | 9,050 | — | ||||||
2022 | 7,412 | — | ||||||
Thereafter | 80,559 | — | ||||||
Total minimum lease payments | $ | 123,943 | $ | 2,869 | ||||
Operating Leases—Rent expense under operating leases totaled approximately $2.1 million for the year ended December 31, 2017, $2.2 million for the year ended December 31, 2016 and $2.3 million for the year ended December 31, 2015.
In the fourth quarter of 2013, the Company entered into a lease for approximately 30,000 square feet of office space and moved the executive and administrative functions into this facility in the second quarter of 2014. The lease expires in 2022 with options to extend the lease for two additional five-year periods. This operating lease included a lease incentive for tenant
72
improvements of $1.7 million which has been included as a leasehold improvement in property, plant and equipment and as deferred rent in other current liabilities and non-current deferred rent.
On January 5, 2018, the Company entered into a sales and leaseback transaction for the San Diego property on Summers Ridge Road that was acquired as part of the Triage Business discussed in Note 12. As part of the transaction, the Company sold the Summers Ridge property and entered into a lease agreement with the buyer to lease two of the four buildings for a term of 15 years. The initial term can be extended for two additional five year terms upon satisfaction of certain conditions. The future minimum lease payments for this property are included in the table above.
McKellar Court Lease Obligation—During 1999, the Company completed a sale and leaseback transaction of its San Diego facility at McKellar Court. The facility was sold for $15.0 million, of which $3.8 million was capital contributed by the Company. The sale was an all cash transaction, netting the Company approximately $7.0 million. The Company is a 25% limited partner in the partnership that acquired the facility. The transaction was deemed a financing transaction under the guidance in ASC Topic 840-40, Accounting for Sales of Real Estate. The assets sold remain on the books of the Company and will continue to be depreciated over the estimated useful life. In December 2009, the Company amended the terms of its lease agreement for the McKellar Court property, which had no significant impact on the Company’s financial statements. The amended terms included a new ten-year lease term through December 31, 2019, with options to extend the lease for up to three additional five-year periods. In the fourth quarter of 2015, the Company amended the terms of its lease agreement to extend the lease term through December 31, 2020. The options to extend the lease for up to three additional five-year periods commence at the new lease term date of December 31, 2020. The Company is amortizing the lease obligation over the new estimated lease term, including extensions. As the Company accounts for the lease as a financing transaction, the Company adjusted the implied interest rate so that the existing lease obligation is amortized to the end of the estimated lease term, including extensions. The Company has determined that the partnership is a variable interest entity (VIE). The Company is not, however, the primary beneficiary of the VIE as it does not absorb the majority of the partnership’s expected losses or receive a majority of the partnership’s residual returns. The Company made lease payments to the partnership of approximately $0.9 million, $0.9 million and $1.1 million for the years ended December 31, 2017, 2016 and 2015, respectively.
Purchase Commitments
The Company has $5.9 million in firm purchase commitments with respect to planned inventory and capital expenditures as of December 31, 2017.
Litigation and Other Legal Proceedings
In Beckman Coulter Inc. v. Quidel Corporation, which was filed in the Superior Court for the County of San Diego, California, on November 27, 2017, Beckman Coulter (“Beckman”) alleges that a provision of an agreement between Quidel and Beckman Coulter violates state antitrust laws. Our acquisition of the BNP Business consisted of assets and liabilities relating to a contractual arrangement with Beckman (the “Beckman Agreement”) for the supply of antibodies and other inputs related to, and distribution of, the Triage® BNP Test for the Beckman Coulter Access Family of Immunoassay Systems. The Beckman Agreement further provides that Beckman, for a specified period, cannot research or develop an assay for use in the diagnosis of cardiac diseases that measures or detects the presence or absence of BNP or NT-pro-BNP (a related biomarker). In the lawsuit, Beckman asserts that this provision violates certain state antitrust laws and is unenforceable. Beckman contends that it has suffered damages due to this provision and seeks a declaration that this provision is void.
We deny that the contractual provision is unlawful, deny any liability with respect to this matter, and intend to vigorously defend ourselves. There are multiple factors that prevent us from being able to estimate the amount of loss, if any, that may result from this matter including: (1) we are vigorously defending ourselves and believe that we have a number of meritorious legal defenses; (2) there are unresolved questions of law and fact that could be important to the ultimate resolution of this matter; and (3) discovery is in the very early stages. Accordingly, at this time, we are not able to estimate a possible loss or range of loss that may result from this matter or to determine whether such loss, if any, would have a material adverse effect on our financial condition, results of operations or liquidity.
From time to time, the Company is involved in other litigation and proceedings, including matters related to product liability claims, commercial disputes and intellectual property claims, as well as regulatory, employment, and other claims related to our business. The Company accrues for legal claims when, and to the extent that, amounts associated with the claims become probable and are reasonably estimable. The actual costs of resolving legal claims may be substantially higher or lower than the amounts accrued for those claims. For those matters as to which we are not able to estimate a possible loss or range of loss, we are not able to determine whether the loss will have a material adverse effect on our business, financial condition or results of operations or liquidity. No accrual has been recorded as of December 31, 2017 and December 31, 2016 related to such
73
matters as they are not probable and/or reasonably estimable.
Management believes that all such current legal actions, in the aggregate, will not have a material adverse effect on the Company. However, the resolution of, or increase in any accruals for, one or more matters may have a material adverse effect on the Company's results of operations and cash flows.
The Company also maintains insurance, including coverage for product liability claims, in amounts which management believes are appropriate given the nature of its business.
Licensing Arrangements
The Company has entered into various licensing and royalty agreements, which largely require payments by the Company based on specified product sales as well as the achievement of specified milestones. The Company had royalty and license expenses relating to those agreements of approximately $0.6 million, $0.8 million and $0.5 million for the years ended December 31, 2017, 2016 and 2015, respectively.
Note 9. Industry and Geographic Information
The Company operates in one reportable segment. Sales to customers outside the U.S. represented 18%, 17% and 14% of total revenue for the years ended December 31, 2017, 2016 and 2015, respectively. As of December 31, 2017 and 2016, balances due from foreign customers, in U.S. dollars, were $18.8 million and $6.8 million, respectively.
The Company had sales to individual customers in excess of 10% of total revenue, as follows:
Year ended December 31, | ||||||||
2017 | 2016 | 2015 | ||||||
Customer: | ||||||||
A | 21 | % | 16 | % | 20 | % | ||
B | 20 | % | 15 | % | 17 | % | ||
C | 13 | % | 13 | % | 11 | % | ||
54 | % | 44 | % | 48 | % | |||
As of December 31, 2017 and 2016, accounts receivable from individual customers with balances due in excess of 10% of total accounts receivable totaled $44.4 million and $13.9 million, respectively.
The following presents long-lived assets (excluding intangible assets) and total net revenue by geographic territory (in thousands):
Long-lived assets as of December 31, | Total revenue for the years ended December 31, | ||||||||||||||||||
2017 | 2016 | 2017 | 2016 | 2015 | |||||||||||||||
Domestic | $ | 59,833 | $ | 50,774 | $ | 227,611 | $ | 158,244 | $ | 168,809 | |||||||||
Foreign | 1,752 | 84 | 50,132 | 33,359 | 27,320 | ||||||||||||||
Total | $ | 61,585 | $ | 50,858 | $ | 277,743 | $ | 191,603 | $ | 196,129 | |||||||||
Consolidated net revenues by product category are as follows (in thousands):
Year ended December 31, | |||||||||||
2017 | 2016 | 2015 | |||||||||
Rapid Immunoassay | $ | 165,099 | $ | 121,416 | $ | 130,348 | |||||
Cardiac Immunoassay | 47,030 | — | — | ||||||||
Specialized Diagnostic Solutions | 51,978 | 60,681 | 60,358 | ||||||||
Molecular Diagnostic Solutions | 13,636 | 9,506 | 5,423 | ||||||||
Total revenues | $ | 277,743 | $ | 191,603 | $ | 196,129 | |||||
74
Note 10. Fair Value Measurement
The following table presents the Company’s hierarchy for its assets and liabilities measured at fair value on a recurring basis as of the following periods (in thousands):
December 31, 2017 | December 31, 2016 | ||||||||||||||||||||||||||||||
Level 1 | Level 2 | Level 3 | Total | Level 1 | Level 2 | Level 3 | Total | ||||||||||||||||||||||||
Assets: | |||||||||||||||||||||||||||||||
Cash equivalents | 36,086 | — | — | 36,086 | 133,540 | — | — | 133,540 | |||||||||||||||||||||||
Total assets at fair value | $ | 36,086 | $ | — | $ | — | $ | 36,086 | $ | 133,540 | $ | — | $ | — | $ | 133,540 | |||||||||||||||
Liabilities: | |||||||||||||||||||||||||||||||
Contingent consideration | — | — | 24,301 | 24,301 | — | — | 5,175 | 5,175 | |||||||||||||||||||||||
Deferred consideration | — | 223,158 | — | 223,158 | — | — | — | — | |||||||||||||||||||||||
Total liabilities at fair value | $ | — | $ | 223,158 | $ | 24,301 | $ | 247,459 | $ | — | $ | — | $ | 5,175 | $ | 5,175 | |||||||||||||||
There were no transfers of assets or liabilities between Level 1, Level 2 and Level 3 categories of the fair value hierarchy during the years ended December 31, 2017 and 2016.
The Company used Level 1 inputs to determine the fair value of its cash equivalents, which primarily consist of funds held in government money market accounts and commercial paper. As such, the carrying value of cash equivalents approximates fair value. As of December 31, 2017 and 2016, the carrying value of cash equivalents was $36.1 million and $133.5 million, respectively.
In connection with the acquisition of the BNP Business, the Company will pay up to $280.0 million in cash, of which $256.0 million is guaranteed and is considered deferred consideration and $24.0 million is contingent consideration. The fair value of the deferred consideration was determined to be $220.6 million on the acquisition date based on the net present value of cash payments using an estimated borrowing rate using a quoted price for a similar liability. The Company recorded 2.6 million for the accretion of interest on the deferred consideration in the fourth quarter of 2017. The fair value of contingent consideration on the acquisition date was $19.7 million and was calculated using a discounted probability weighted valuation model.
In conjunction with the acquisitions of BioHelix Corporation in May 2013, AnDiaTec GmbH & Co. KG in August 2013 and Immutopics, Inc. in March 2016, the Company recorded contingent consideration of $4.6 million as of December 31, 2017 and $5.2 million as of December 31, 2016. The Company assesses the fair value of contingent consideration to be settled in cash related to these prior acquisitions using a discounted revenue model. Significant assumptions used in the measurement include revenue projections and discount rates. This fair value measurement of contingent consideration is based on significant inputs not observed in the market and thus represent Level 3 measurements. Due to changes in the estimated payments and a shorter discounting period related to the various contingent consideration liabilities, the fair value of the contingent consideration changed during the years ended 2017, 2016 and 2015. These changes resulted in gains of $0.1 million, $0.5 million and $0.1 million recorded to cost of sales in the Consolidated Statements of Operations during the years ended December 31, 2017, 2016 and 2015, respectively.
Changes in estimated fair value of contingent consideration liabilities from December 31, 2016 through December 31, 2017 are as follows (in thousands):
Contingent consideration liability (Level 3 measurement) | |||
Balance at December 31, 2016 | $ | 5,175 | |
Cash payments | (498 | ) | |
Net gain recorded for fair value adjustments | (80 | ) | |
Additional liability recorded for the BNP Business | 19,700 | ||
Unrealized loss on foreign currency translation | 4 | ||
Balance at December 31, 2017 | $ | 24,301 | |
75
Note 11. Employee Benefit Plan
The Company has a defined contribution 401(k) plan (the “401(k) Plan”) covering all employees who are eligible to join the 401(k) Plan upon employment. Employee contributions are subject to a maximum limit by federal law. This Plan includes an employer match of 50% on the first 6% of pay contributed by the employee. The Company contributed approximately $1.5 million, $1.5 million and $1.3 million to the 401(k) Plan during the years ended December 31, 2017, 2016 and 2015, respectively.
Note 12. Acquisition
On October 6, 2017, the Company acquired the Triage and BNP Businesses. The acquisition has been accounted for in conformity with ASC Topic 805, Business Combinations. In connection with the acquisition of the Triage Business, the Company paid $399.8 million in cash and assumed certain liabilities. These acquisitions enhance the Company's revenue profile and expand the Company's geographic footprint and product diversity. The Company used proceeds from the Term Loan (defined and discussed in Note 3) of $245.0 million and cash on hand to pay (i) the consideration for the Triage Business and (ii) fees and expenses incurred in connection with the acquisition of the Triage and BNP Businesses. In connection with the acquisition of the BNP Business, the Company: (i) will pay (A) $16.0 million in cash plus up to an additional $24.0 million in contingent consideration, payable in five annual installments of up to $8.0 million, the first of which will be paid on April 30, 2018, (B) $240.0 million in cash, payable in six annual installments of $40.0 million each, the first of which will be paid on April 30, 2018 and (C) $0.2 million in cash for certain inventory related adjustments; and (ii) assumed certain liabilities.
The purchase price consideration is as follows (in thousands):
Cash consideration—Triage Business | $ | 399,798 | |
Deferred consideration—BNP Business | 220,550 | ||
Contingent consideration—BNP Business | 19,700 | ||
Inventory related adjustment | 205 | ||
Net consideration | $ | 640,253 | |
The fair value of the deferred consideration was determined to be $220.6 million on the acquisition date based on the net present value of cash payments using an estimated borrowing rate using a quoted price for a similar liability. The fair value of contingent consideration on the acquisition date was $19.7 million and was calculated using a discounted probability weighted valuation model.
The Company is still finalizing the allocation of the purchase price, therefore, the purchase price allocation or the provisional measurements of intangible assets, goodwill and deferred income tax assets or liabilities may be adjusted if the Company recognizes additional assets or liabilities to reflect new information obtained about facts and circumstances that existed as of the acquisition date that, if known, would have affected the measurement of the amounts recognized as of that date. The Company expects to complete the allocation of purchase price during fiscal year 2018.
The components of the preliminary purchase price allocation at the acquisition date and the purchase price consideration transferred at December 31, 2017 are as follows:
Prepaid expenses and other current assets | $ | 796 | |
Assets held for sale | 146,540 | ||
Inventories | 52,205 | ||
Property, plant and equipment | 10,608 | ||
Intangible assets | 184,900 | ||
Goodwill | 245,531 | ||
Other non-current assets | 182 | ||
Total assets acquired | $ | 640,762 | |
Other current liabilities | (509 | ) | |
Total net assets and liabilities acquired | $ | 640,253 | |
76
Goodwill represents the excess of the total purchase price over the fair value of the underlying net assets, largely arising from synergies expected to be achieved by the combined Company and the expanded revenue profile and product diversity. The goodwill is expected to be fully deductible for tax purposes.
The following sets forth results of the amounts assigned to the identifiable intangible assets acquired (in thousands):
Intangible Asset | Amortization period | Fair value of assets acquired | ||||
Purchased technology | 10 years | $ | 52,400 | |||
Customer relationships | 7 years | 115,000 | ||||
Trademarks | 10 years | 17,500 | ||||
Total intangible assets | $ | 184,900 | ||||
The fair value of the identified intangible assets was determined primarily using an income based approach. Intangible assets are amortized on a straight-line basis over the amortization periods noted above.
The following unaudited pro forma financial information shows the combined results of operations of the Company, including the Triage and BNP Businesses, as if the acquisition had occurred as of the beginning of the periods presented:
Year ended December 31, | |||||||
(in thousands, except per share data) | 2017 | 2016 | |||||
Pro forma total revenues | $ | 468,577 | $ | 437,525 | |||
Pro forma net income (1) | $ | 41,296 | $ | 28,045 | |||
Pro forma basic earnings per share | $ | 1.22 | $ | 0.86 | |||
Pro forma diluted net earnings per share | $ | 1.18 | $ | 0.84 | |||
Total revenue included in the Consolidated Statement of Operations since acquisition | $ | 47,030 | |||||
Net loss included in the Consolidated Statement of Operations since acquisition (2) | $ | (4,776 | ) | ||||
(1) | Includes the reversal of non-recurring transaction costs totaling $11.7 million directly attributable to the acquisition incurred during the year ended December 31, 2017. Pro forma net income also includes the reversal of the amortization of the fair value step-up of inventory of $11.0 million because it does not have a continuing impact. |
(2) | Net loss of $4.8 million includes amortization of the fair value write-up of inventory of $11.0 million, amortization of acquired intangible assets of $5.9 million, cash and non-cash interest expense on the Term Loan of $3.5 million and accretion of interest on the deferred consideration of $2.6 million. |
77
Note 13. Selected Quarterly Financial Data (unaudited)
First Quarter | Second Quarter | Third Quarter | Fourth Quarter | ||||||||||||
(in thousands, except per share data) | |||||||||||||||
2017 | |||||||||||||||
Total revenues | $ | 73,692 | $ | 38,267 | $ | 50,894 | $ | 114,890 | |||||||
Gross profit (1) | $ | 48,499 | $ | 18,826 | $ | 29,690 | $ | 59,127 | |||||||
Operating income (loss) | $ | 19,229 | $ | (11,027 | ) | $ | (2,537 | ) | $ | 3,887 | |||||
Net income (loss) | $ | 14,290 | $ | (11,842 | ) | $ | (5,525 | ) | $ | (5,088 | ) | ||||
Basic income (loss) per share | $ | 0.43 | $ | (0.35 | ) | $ | (0.16 | ) | $ | (0.15 | ) | ||||
Diluted income (loss) per share | $ | 0.42 | $ | (0.35 | ) | $ | (0.16 | ) | $ | (0.15 | ) | ||||
2016 | |||||||||||||||
Total revenues | $ | 50,321 | $ | 39,133 | $ | 49,341 | $ | 52,808 | |||||||
Gross profit (1) | $ | 29,482 | $ | 20,225 | $ | 30,023 | $ | 32,001 | |||||||
Operating income (2) | $ | (3,460 | ) | $ | (9,019 | ) | $ | 2,125 | $ | 5,915 | |||||
Net loss | $ | (3,446 | ) | $ | (7,840 | ) | $ | (572 | ) | $ | (1,950 | ) | |||
Basic and diluted loss per share (3) | $ | (0.11 | ) | $ | (0.24 | ) | $ | (0.02 | ) | $ | (0.06 | ) | |||
(1) | Includes reclassification of $1.6 million, $1.7 million and $1.8 million from amortization of intangible assets from acquired business and technology to cost of sales expense for the first, second and third quarters, respectively, in the year ended December 31, 2017. Includes reclassification of $1.6 million, $1.6 million, $1.6 million and $1.7 million from amortization of intangible assets from acquired business and technology to cost of sales expense for the first, second, third and fourth quarters in the year ended December 31, 2016, respectively. |
(2) | Includes reclassification of $0.7 million from amortization of intangible assets from acquired business and technology to sales and marketing expense for each of the first, second and third quarters in the year ended December 31, 2017. Includes reclassification of $0.6 million, $0.7 million, $0.7 million and $0.6 million from amortization of intangible assets from acquired business and technology to sales and marketing expense for the first, second, third and fourth quarters, respectively, in the year ended December 31, 2016. |
(3) | Basic and diluted EPS amounts in each quarter are computed using the weighted-average number of shares outstanding during that quarter, while basic and diluted EPS for the full year is computed using the weighted-average number of shares outstanding during the year. Therefore, the sum of the four quarters’ basic or diluted EPS may not equal the full year basic or diluted EPS. |
Note 14. Subsequent Events
On January 5, 2018, the Company entered into a sale and leaseback transaction for the San Diego property on Summers Ridge Road that was acquired as part of the Triage Business from Alere discussed in Note 12 and is included as assets held for sale on the Consolidated Balance Sheet as of the year ended December 31, 2017. The Company sold the Summers Ridge property for net consideration of $146.6 million. In addition, the Company entered into a lease agreement with the buyer to lease two of the four buildings on the Summers Ridge campus for a term of 15 years.
On January 11, 2018, the Company used $100.0 million of net cash proceeds from the sale and leaseback transaction to pay down a portion of the existing Term Loan under its Credit Agreement described in Note 3. Following the payment, the Company has approximately $145.0 million remaining in Term Loan outstanding. Separately, the Company also repaid the entire outstanding $10.0 million balance on its Revolving Credit Facility under the Credit Agreement.
78
SCHEDULE II
QUIDEL CORPORATION
CONSOLIDATED VALUATION AND QUALIFYING ACCOUNTS
Description | Balance at beginning of period | Additions charged to expense or as reductions to revenue (1) | Deductions (2) | Balance at end of period | ||||||||||||
(in thousands) | ||||||||||||||||
Year ended December 31, 2017 | ||||||||||||||||
Accounts receivable allowance | $ | 7,165 | $ | 36,449 | $ | (31,305 | ) | $ | 12,309 | |||||||
Year ended December 31, 2016: | ||||||||||||||||
Accounts receivable allowance | $ | 7,488 | $ | 28,329 | $ | (28,652 | ) | $ | 7,165 | |||||||
Year ended December 31, 2015: | ||||||||||||||||
Accounts receivable allowance | $ | 8,221 | $ | 31,532 | $ | (32,265 | ) | $ | 7,488 | |||||||
(1) | Represents charges associated primarily to accruals for early payment discounts, volume discounts and contract rebates recorded as reductions to revenue. Additions to allowance for doubtful accounts are recorded to sales and marketing expenses. |
(2) | The deductions represent actual charges against the accrual described above. |
79
Item 9. Changes in and Disagreements With Accountants on Accounting and Financial Disclosure
None.
Item 9A. Controls and Procedures
Evaluation of disclosure controls and procedures: We have performed an evaluation under the supervision and with the participation of our management, including our Chief Executive Officer (“CEO”) and Chief Financial Officer (“CFO”), of the effectiveness of our disclosure controls and procedures, as defined in Rule 13a-15(e) under the Securities Exchange Act of 1934 (the “Exchange Act”). Based on that evaluation, our CEO and CFO concluded that our disclosure controls and procedures were effective as of December 31, 2017 at a reasonable assurance level to ensure that information required to be disclosed by us in the reports filed or submitted by us under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms.
Changes in internal control over financial reporting: There was no change in our internal control over financial reporting during the quarter ended December 31, 2017 that materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
Management’s report on internal control over financial reporting: Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Exchange Act Rule 13a-15(f). Our internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of our financial statements for external purposes in accordance with U.S. generally accepted accounting principles. Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Therefore, even those systems determined to be effective can provide only reasonable assurance with respect to financial statement preparation and presentation. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate. Under the supervision and with the participation of our management, including our CEO and CFO, we conducted an evaluation of the effectiveness of our internal control over financial reporting based on the framework in Internal Control—Integrated Framework (2013), issued by the Committee of Sponsoring Organizations of the Treadway Commission. Management has excluded from its assessment the internal control over financial reporting for the Triage and BNP Businesses acquired from Alere Inc. on October 6, 2017. The Triage and BNP Businesses constitute 8% of total assets and 17% of total revenues of Quidel’s consolidated financial statement amounts as of and for the year ended December 31, 2017. These businesses will be in scope for management’s assessment as of December 30, 2018. Based on our evaluation under the framework in Internal Control—Integrated Framework, our management concluded that our internal control over financial reporting was effective as of December 31, 2017.
The effectiveness of our internal control over financial reporting as of December 31, 2017 has been audited by Ernst & Young LLP, our independent registered public accounting firm, as stated in their report which is included in this Item 9A.
80
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Shareholders and the Board of Directors of Quidel Corporation
Opinion on Internal Control over Financial Reporting
We have audited Quidel Corporation’s internal control over financial reporting as of December 31, 2017, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework), (the COSO criteria). In our opinion, Quidel Corporation (the Company) maintained, in all material respects, effective internal control over financial reporting as of December 31, 2017, based on the COSO criteria.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the consolidated balance sheets of the Company as of December 31, 2017 and 2016, the related consolidated statements of operations, comprehensive loss, stockholders‘ equity, and cash flows, for each of the three years in the period ended December 31, 2017, and the related notes and schedule listed in the Index at Item 15(a)(2) and our report dated February 27, 2018 expressed an unqualified opinion thereon.
As indicated in the accompanying Management’s Report on Internal Control over Financial Reporting, management’s assessment of and conclusion on the effectiveness of internal control over financial reporting did not include the internal controls of the Triage and BNP businesses, which are included in the December 31, 2017 consolidated financial statements of the Company and constituted 8% of total assets, as of December 31, 2017 and 17% of total revenues for the year then ended. Our audit of internal control over financial reporting of the Company also did not include an evaluation of the internal control over financial reporting of the Triage and BNP businesses.
Basis for Opinion
The Company’s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management’s Report on Internal Control Over Financial Reporting. Our responsibility is to express an opinion on the Company’s internal control over financial reporting based on our audit. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects.
Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
Definition and Limitations of Internal Control Over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
/s/ Ernst & Young LLP
San Diego, California
February 27, 2018
81
Item 9B. Other Information
2018 Annual Meeting of Stockholders
The Company’s 2018 Annual Meeting of Stockholders will be held on Tuesday, May 15, 2018, beginning at 8:30 a.m. (local time) at the San Diego Marriott Del Mar, 11966 El Camino Real, San Diego, California 92130.
82
Part III
Item 10. Directors, Executive Officers and Corporate Governance
The information required by this item is incorporated by reference to our 2018 proxy statement, which will be filed with the SEC no later than April 30, 2018 (the "2018 Proxy Statement"). Information with respect to the Company's executive officers is included under Part 1 of this Annual Report.
Item 11. Executive Compensation
The information required by this item is incorporated by reference from our 2018 Proxy Statement.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The information required by this item is incorporated by reference from our 2018 Proxy Statement.
Item 13. Certain Relationships and Related Transactions, and Director Independence
The information required by this item is incorporated by reference from our 2018 Proxy Statement.
Item 14. Principal Accountant Fees and Services
The information required by this item is incorporated by reference from our 2018 Proxy Statement.
83
Part IV
Item 15. Exhibits and Financial Statement Schedules
The following documents are filed as part of this Form 10-K:
(a) | (1) Financial Statements |
The Consolidated Financial Statements required by this Item are submitted in Part II, Item 8 of this form 10-K.
(2) Financial Statement Schedules
The following Financial Statement Schedule of Quidel Corporation for the years ended December 31, 2017, 2016 and 2015 is filed as part of this Annual Report in Part II, Item 8 and should be read in conjunction with the Consolidated Financial Statements of Quidel Corporation:
Schedule II. Consolidated Valuation and Qualifying Accounts.
Financial Statement Schedules not listed above have been omitted because of the absence of conditions under which they are required or because the required information is included in the Consolidated Financial Statements or the Notes thereto.
(3) Exhibits. See Paragraph 15(b) below.
(b) | Exhibits |
The exhibits listed on the accompanying Exhibit Index immediately following this Item 15 are filed as part of, and incorporated by reference into, this Annual Report on Form 10-K.
(c) | Financial Statements required by Regulation S-X which are excluded from this Annual Report on Form 10-K by Rule 14(a)-3(b). |
Not applicable.
84
EXHIBIT INDEX
Exhibit Number | Description |
85
86
101.INS* | XBRL Instance Document |
101.SCH* | XBRL Taxonomy Extension Schema Document |
101.CAL* | XBRL Taxonomy Calculation Linkbase Document |
101.DEF* | XBRL Taxonomy Extension Definition Linkbase Document |
101.LAB* | XBRL Taxonomy Label Linkbase Document |
101.PRE* | XBRL Taxonomy Presentation Linkbase Document |
* | Filed / furnished herewith |
(1) | Indicates a management plan or compensatory plan or arrangement. |
87
Item 16. Form 10-K Summary
Not applicable.
88
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this Report to be signed on its behalf by the undersigned, thereunto duly authorized.
QUIDEL CORPORATION | ||
By | /s/ DOUGLAS C. BRYANT | |
Date: February 27, 2018 | Douglas C. Bryant President, Chief Executive Officer (Principal Executive Officer) | |
Pursuant to the requirements of the Securities Exchange Act of 1934, this Report has been signed below by the following persons on behalf of the Registrant and in the capacities and on the dates indicated.
Signature | Title | Date | ||
/s/ DOUGLAS C. BRYANT | Director, President, Chief Executive Officer (Principal Executive Officer) | February 27, 2018 | ||
Douglas C. Bryant | ||||
/s/ RANDALL J. STEWARD | Chief Financial Officer, (Principal Financial and Accounting Officer) | February 27, 2018 | ||
Randall J. Steward | ||||
/s/ KENNETH F. BUECHLER | Chairman of the Board | February 27, 2018 | ||
Kenneth F. Buechler | ||||
/s/ THOMAS D. BROWN | Director | February 27, 2018 | ||
Thomas D. Brown | ||||
/s/ MARY LAKE POLAN | Director | February 27, 2018 | ||
Mary Lake Polan | ||||
/s/ JACK W. SCHULER | Director | February 27, 2018 | ||
Jack W. Schuler | ||||
/s/ CHARLES P. SLACIK | Director | February 27, 2018 | ||
Charles P. Slacik | ||||
/s/ KENNETH J. WIDDER | Director | February 27, 2018 | ||
Kenneth J. Widder | ||||
89