0001935979false00019359792024-09-232024-09-23
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, DC 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of
The Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): September 23, 2024
Biohaven Ltd.
(Exact name of registrant as specified in its charter)
| | | | | | | | |
| British Virgin Islands | 001-41477 | Not applicable |
| (State or other jurisdiction of incorporation) | (Commission File Number) | (IRS Employer Identification No.) |
c/o Biohaven Pharmaceuticals, Inc.
215 Church Street
New Haven, Connecticut 06510
(Address of principal executive offices, including zip code)
(203) 404-0410
(Registrant’s telephone number, including area code)
Not applicable
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
☐ Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425)
☐ Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12)
☐ Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b))
☐ Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c))
Securities registered pursuant to Section 12(b) of the Act:
| | | | | | | | |
| Title of each class | Trading symbol | Name of each exchange on which registered |
| Common Shares, no par value | BHVN | New York Stock Exchange |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 7.01 Regulation FD Disclosure
On September 23, 2024, Biohaven Ltd. (the “Company”) issued a press release announcing topline data from the Company’s study to assess the effectiveness of troriluzole in Spinocerebellar Ataxia (“SCA”). A copy of the press release is furnished herewith as Exhibit 99.1 to this Current Report on Form 8-K.
In addition, on September 23, 2024, members of management of the Company held a conference call to discuss the results of the SCA study. A copy of the presentation that accompanied the conference call is available on the Company’s website at www.biohaven.com.
The information contained in this Item 7.01, including Exhibit 99.1, is being “furnished” and shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liability of that section or Sections 11 and 12(a)(2) of the Securities Act of 1933, as amended (the “Securities Act”). The information in this Item 7.01, including Exhibit 99.1, shall not be incorporated by reference into any registration statement or other document pursuant to the Securities Act or into any filing or other document pursuant to the Exchange Act, except as otherwise expressly stated in any such filing.
Item 8.01 Other Events
On September 23, 2024, the Company announced positive topline results from pivotal Study BHV4157-206-RWE (NCT06529146) demonstrating the efficacy of troriluzole on the mean change from baseline in the f-SARA after 3 years of treatment. The study achieved the primary endpoint and showed statistically significant improvements on the f-SARA at years 1 and 2 (Figure 1). SCA is a rare, progressively debilitating neurodegenerative disease that affects approximately 15,000 people in the United States and 24,000 in Europe and the United Kingdom. There are no U.S. Food and Drug Administration (the “FDA”) approved treatments for SCA.
Collectively, data across multiple analyses demonstrate a robust and clinically meaningful slowing of disease progression in SCA patients. These treatment benefits translate into a 50- 70% slower rate of decline compared to untreated patients, representing 1.5-2.2 years delay in disease progression over the 3-year study period. Additionally, in a responder sensitivity analysis, disease progression when defined by a 2 point or greater worsening on the f-SARA at 3 years showed an odds ratio (“OR”) of 4.1 (95% CI: 2.1, 8.1) for the untreated external control arm versus troriluzole treated subjects (p < 0.0001; pooled analysis).
Figure 1: f-SARA Change from baseline demonstrating troriluzole reduced SCA disease progression vs US Natural History External Control
Study BHV4157-206-RWE was designed, in discussion with the FDA, to assess the effectiveness of troriluzole in SCA after 3 years of treatment as measured by the change from baseline in the f-SARA. The study utilized Phase 3 data and an external control of matched, untreated SCA subjects from the US Clinical Research Consortium for the Study of Cerebellar Ataxia (“CRC-SCA”) in accordance with the FDA’s Guidance on Real-World Evidence (“RWE”) of effectiveness. All endpoints were prespecified, and both the study protocol and statistical analysis plan were submitted to, and reviewed by, the FDA prior to topline data analysis. The new analysis doubled the previously available 3 year data with 63 subjects now completing 3 years of treatment with troriluzole and matched to the external control arm. Propensity Score Matching was used to ensure that untreated patients from the CRC-SCA study were rigorously matched to treated patients from Study BHV4157-206 on baseline characteristics. The primary objective was to examine the treatment effects of troriluzole for up to 3 years, by comparing data on the f-SARA from patients treated with troriluzole in Study BHV4157-206 to untreated patients from the natural history study. Troriluzole-treated patients demonstrated statistically significant and sustained benefits at years 1, 2 and 3 on the f-SARA compared to a rigorously matched natural history control.
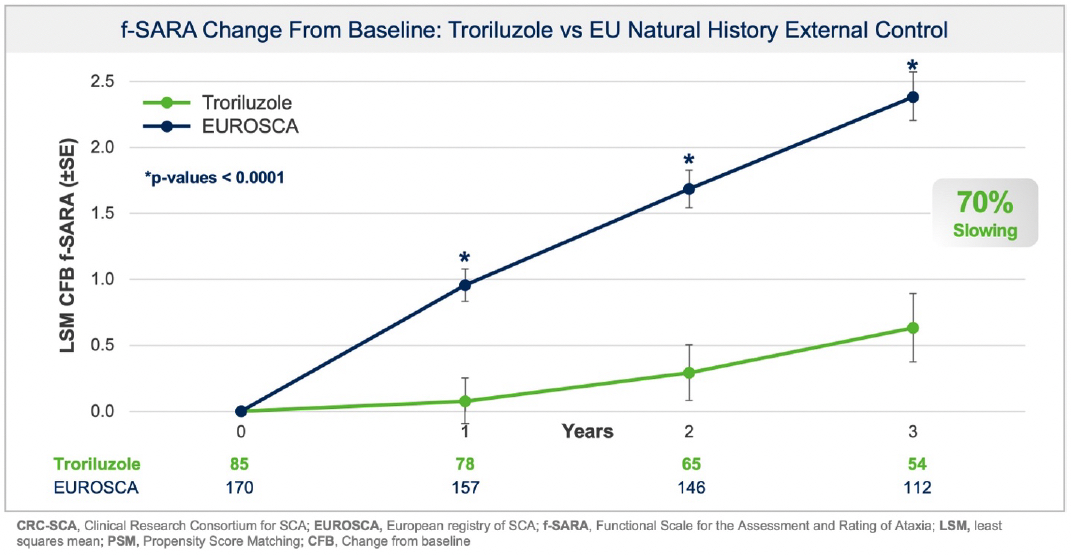
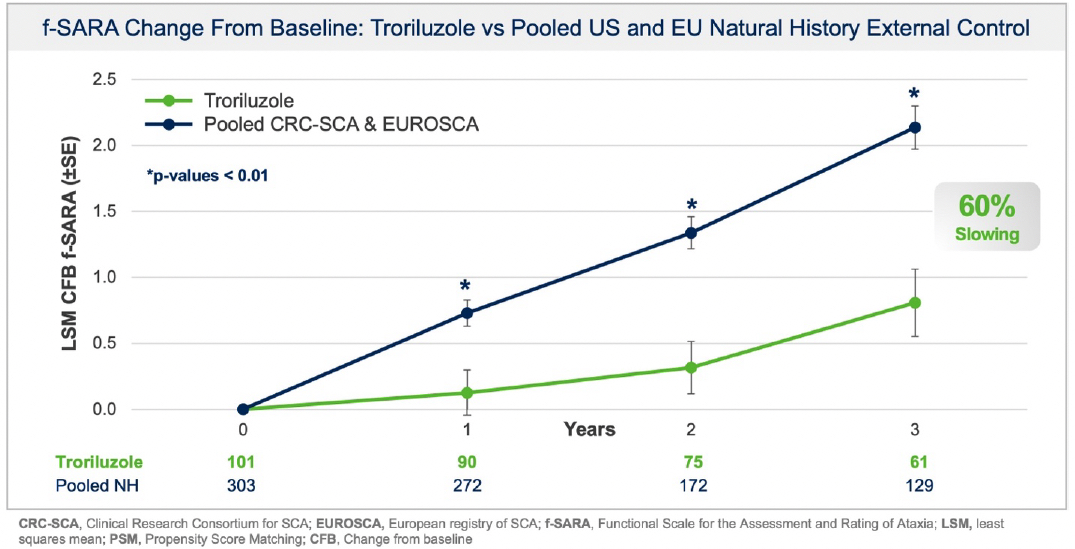
Additionally, prespecified analyses in the protocol employed a separate, independent natural history control from the European SCA natural history study (“EUROSCA”) for global regulatory purposes. Results using the EUROSCA patients, in addition to a pooled analysis using both CRC-SCA and EUROSCA patients, as the external controls were also statistically significant and consistent with the primary efficacy analysis at all timepoints (see Figure 2 and Figure 3). The addition of EUROSCA data increased the external control sample size and added to the robustness of the statistically significant treatment differences at years 1, 2, and 3, favoring troriluzole.
Figure 2: f-SARA change from baseline demonstrating troriluzole reduced SCA disease progression vs Independent EU Natural History External Control
Figure 3: f-SARA change from baseline demonstrating troriluzole reduced SCA disease progression vs Pooled US and EU Natural History External Control
Spinocerebellar ataxia is a group of dominantly inherited neurodegenerative disorders characterized by progressive loss of voluntary motor control and atrophy of the cerebellum, brainstem and spinal cord. Patients experience significant morbidity, including progression to a wheelchair, impaired gait leading to falls, inability to communicate due to speech impairment, difficulty swallowing, and premature death. While signs and symptoms can appear anytime from childhood to late adulthood, SCA typically presents in early adulthood and progresses over a number of years. Currently, there are no FDA-approved treatments and no cure for SCA.
Based upon the topline data from Study BHV4157-206-RWE, and previous safety and efficacy data from the troriluzole development program in SCA, the Company plans to submit a New Drug Application (“NDA”) to the FDA in the fourth quarter of 2024. The troriluzole development program has generated the largest clinical trial dataset in SCA and now has follow-up in some patients treated with troriluzole for over 5 years. The Company has previously received both Fast-Track and Orphan drug designation (“ODD”) from the FDA, and ODD from the European Medicines Agency, for troriluzole in SCA. An NDA with ODD is eligible for priority FDA review. The Company will be prepared to commercialize SCA in the United States in 2025, if ultimately approved, based on potential priority review timelines.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits
| | | | | | | | |
| Exhibit Number | | Exhibit Description |
| 99.1 | | |
| | |
| | |
| | |
| | |
| | |
| 104 | | The cover page of this Current Report on Form 8-K formatted as Inline XBRL. |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
Date: September 23, 2024
| | | | | |
| Biohaven Ltd. |
|
| By: | /s/ Matthew Buten |
| Matthew Buten |
| Chief Financial Officer |