UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM
(Mark One)
OR
for
the fiscal year ended
OR
OR
Date of event requiring this shell company report: _________
for the transition period from _________ to _________
Commission File Number:
(Exact name of registrant as specified in its charter)
(Jurisdiction of incorporation or organization)
CAP Business Center
(Address of principal executive offices)
Chief Executive Officer
MDxHealth, Inc.
United States
(Name, Telephone, E-mail and/or Facsimile number and Address of Company Contact Person)
Securities registered or to be registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol | Name of each exchange on which registered | ||
| The |
Securities registered or to be registered pursuant to Section 12(g) of the Act. None
Securities for which there is a reporting obligation pursuant to Section 15(d) of the Act. None
Indicate
the number of outstanding shares of each of the issuer’s class of capital or common stock as of the close of the period covered
by the annual report. Ordinary shares, no nominal value per share:
Indicate
by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐
If
this report is an annual or transition report, indicate by check mark if the registrant is not required to file reports pursuant to Section
13 or 15(d) of the Securities Exchange Act of 1934. Yes ☐
Note - Checking the box above will not relieve any registrant required to file reports pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 from their obligations under those Sections.
Indicate
by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange
Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2)
has been subject to such filing requirements for the past 90 days.
Indicate
by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule
405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant
was required to submit such files).
Indicate by check mark whether the registrant is a large accelerated filer, accelerated filer, a non-accelerated filer, or an emerging growth company. See definition of “large accelerated filer”, “accelerated filer” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer | ☐ | Accelerated filer | ☐ | |
| ☒ | Emerging Growth Company | |||
If
an emerging growth company that prepares its financial statements in accordance with U.S. GAAP, indicate by check mark if the registrant
has elected not to use the extended transition period for complying with any new or revised financial accounting standards† provided
pursuant to Section 13(a) of the Exchange Act.
| † | The term “new or revised financial accounting standard” refers to any update issued by the Financial Accounting Standards Board to its Accounting Standards Codification after April 5, 2012. |
Indicate
by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness
of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered
public accounting firm that prepared or issued its audit report.
If
securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant
included in the filing reflect the correction of an error to previously issued financial statements.
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate by check mark which basis of accounting the registrant has used to prepare the financial statements included in this filing:
| U.S. GAAP ☐ | as issued by the International Accounting Standards Board ☒ | Other ☐ |
If “Other” has been checked in response to the previous question, indicate by check mark which financial statement item the registrant has elected to follow: Item 17 ☐ Item 18 ☐
If
this is an annual report, indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange
Act). Yes ☐ No
TABLE OF CONTENTS
i
INTRODUCTION
Unless otherwise indicated or the context otherwise requires, references in this annual report to “we,” “our,” “us,” “MDxHealth,” or the “Company” refer to MDxHealth SA and its wholly owned subsidiaries.
We were incorporated on January 10, 2003 as a company with limited liability (naamloze vennootschap/société anonyme) incorporated and operating under the laws of Belgium. We are registered with the legal entities register (Liège) under enterprise number 0479.292.440. In October 2010, the Company’s name was changed from OncoMethylome Sciences SA to MDxHealth SA. We have two directly held, wholly owned subsidiaries: MDxHealth, Inc., a Delaware company incorporated in April 2003, and MDxHealth B.V., a Dutch company incorporated in September 2015.
Our headquarters and principal executive offices are located at CAP Business Center, Zone Industrielle des Hauts-Sarts, Rue d’Abhooz 31, 4040 Herstal, Belgium, our telephone number is +32 4 257 70 21 and our email is info@mdxhealth.com. Our website address is www.mdxhealth.com. The information contained on, or accessible through, our website is not incorporated by reference into this annual report, and you should not consider any information contained in, or that can be accessed through, our website as part of this annual report.
American Depositary Shares (“ADS”), each representing 10 ordinary shares of the Company, began trading on the Nasdaq Capital Market on November 4, 2021. On November 13, 2023, we completed a 1-for-10 reverse stock split of our ordinary shares, after which each ADS represented one ordinary share. On November 27, 2023, we completed the mandatory exchange of all of our ADSs for one ordinary share each and subsequently terminated the Company’s ADS facility, at which time the ordinary shares were admitted to listing on the Nasdaq Capital Market under the symbol “MDXH”. Following a transition period of three weeks, the Company de-listed its ordinary shares from Euronext Brussels and, as of December 18, 2023, our ordinary shares began solely trading on the Nasdaq Capital Market. All references in this annual report to “$” are to U.S. dollars and all references to “€” are to Euros. Solely for the convenience of the reader, certain Euro amounts herein have been translated into U.S. dollars at the official exchange rate quoted as of December 31, 2023, by the European Central Bank of €1.00 to $1.105 for assets and liabilities and €1.00 to $1.081 for income and expenses. These translations should not be considered representations that any such amounts have been, could have been or could be converted into U.S. dollars at that or any other exchange rate as at that or any other date. The disclosures in this report give retroactive effect to these changes, with all references to shares representing ordinary shares of the Company.
Trademarks and Service Marks
We own various trademark registrations and applications, and unregistered trademarks and service marks. “MDxHealth,” “Confirm mdx,” “Select mdx,” “Resolve mdx,” “Genomic Prostate Score,” “GPS,” “Monitor mdx,” the MDxHealth logo and other trademarks or service marks of MDxHealth SA appearing in this annual report are the property of MDxHealth SA or its subsidiaries. Solely for convenience, the trademarks, service marks and trade names referred to in this annual report are listed without the ® and ™ symbols, but such references should not be construed as any indicator that their respective owners will not assert, to the fullest extent under applicable law, their rights thereto. All other trademarks, trade names and service marks appearing in this annual report are the property of their respective owners. We do not intend to use or display other companies’ trademarks and trade names to imply any relationship with, or endorsement or sponsorship of us by, any other companies.
ii
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This annual report contains forward-looking statements. All statements other than statements of historical facts contained in this annual report, including statements regarding our strategy, future operations, future financial position, future revenue, projected costs, prospects, plans, objectives of management and expected market growth are forward-looking statements. Forward-looking statements give our current expectations or forecasts of future events. You can find many (but not all) of these statements by looking for words such as “approximates,” “believes,” “hopes,” “expects,” “anticipates,” “estimates,” “projects,” “intends,” “plans,” “would,” “should,” “could,” “may”, “potential”, “will”, “goal”, “next”, “aim”, “explore” or other similar expressions in this annual report. These forward-looking statements are subject to certain risks and uncertainties that could cause actual results to differ materially from our historical experience and our present expectations or projections, including, but not limited to, those identified under Item 3D. “Risk Factors” in this annual report. Actual results may differ materially from those discussed as a result of various factors, including, but not limited to:
| ● | our plans relating to commercializing our tests and related diagnostic products and services (collectively “tests”, “testing solutions” or “solutions”) and the rate and degree of market acceptance of our solutions; |
| ● | the size of the market opportunity for our Confirm mdx, Select mdx, Resolve mdx, Monitor mdx and Genomic Prostate Score (“GPS”) tests and other future tests and solutions we commercialize or may develop; |
| ● | the acceptance of our testing solutions by healthcare providers; |
| ● | the willingness of health insurance companies and other payers to cover our testing solutions and adequately reimburse us for such solutions; |
| ● | our plans relating to the further development of testing solutions; |
| ● | existing regulations and regulatory developments in the United States, Europe and other jurisdictions; |
| ● | our ability to obtain and maintain regulatory approvals and comply with applicable regulations; |
| ● | timing, progress and results of our research and development programs; |
| ● | the period over which we estimate our existing cash will be sufficient to fund our future operating expenses and capital expenditure requirements; |
| ● | our ability to attract and retain qualified employees and key personnel; |
| ● | the scope of protection we are able to establish and maintain for intellectual property rights covering our testing solutions and technology; |
| ● | our ability to operate our business without infringing the intellectual property rights and proprietary technology of third parties; |
| ● | the possibility that the anticipated benefits from our business acquisitions will not be realized in full or at all or may take longer to realize than expected; |
| ● | cost associated with defending intellectual property infringement, product liability and other claims; |
| ● | uncertainties associated with global macroeconomic conditions; and |
| ● | other risks and uncertainties, including those listed under Item 3D. “Risk Factors.” |
These statements reflect our views with respect to future events as of the date of this annual report and are based on assumptions and subject to risks and uncertainties. Given these uncertainties, you should not place undue reliance on these forward-looking statements. These forward-looking statements represent our estimates and assumptions only as of the date of this annual report and, except as required by law, we undertake no obligation to update or review publicly any forward-looking statements, whether as a result of new information, future events or otherwise after the date of this annual report. We anticipate that subsequent events and developments will cause our views to change. You should read this annual report and the documents referenced in this annual report and filed as exhibits to the annual report, completely and with the understanding that our actual future results may be materially different from what we expect. We qualify all of our forward-looking statements by these cautionary statements.
iii
MARKET AND INDUSTRY DATA
Unless otherwise indicated, information contained in this annual report concerning our industry and the markets in which we operate, including our general expectations and market opportunity, is based on information from our own management estimates and research, as well as from industry and general publications, research, surveys and studies conducted by third parties. Management estimates are derived from publicly available information, our knowledge of our industry and assumptions based on such information and knowledge, which we believe to be reasonable. Where information has been sourced from third parties, this information has been accurately reproduced. As far as we are aware and are able to ascertain from information published by those third parties, no facts have been omitted which would render the reproduced information inaccurate or misleading. The industry publications and third-party studies generally state that the information that they contain has been obtained from sources believed to be reliable, although they do not guarantee the accuracy or completeness of such information. Forecasts and other forward-looking information obtained from these sources are subject to the same qualifications and uncertainties as the other forward-looking statements in this annual report. See “Special Note Regarding Forward-Looking Statements.” These forecasts and forward-looking information are subject to uncertainty and risk due to a variety of factors, including those described under “Risk Factors.” These and other factors could cause results to differ materially from those expressed in our forecasts or estimates or those of independent third parties.
iv
PART I
ITEM 1. IDENTITY OF DIRECTORS, SENIOR MANAGEMENT AND ADVISERS
Not applicable.
ITEM 2. OFFER STATISTICS AND EXPECTED TIMETABLE
Not applicable.
ITEM 3. KEY INFORMATION
A. [Reserved]
B. Capitalization and Indebtedness
Not applicable.
C. Reasons for the Offer and Use of Proceeds
Not applicable.
D. Risk Factors
Our business and our industry are subject to significant risks. You should carefully consider the risks and uncertainties described below, together with all of the other information in this annual report, including our audited consolidated financial statements and related notes. This annual report also includes forward-looking statements that involve risks and uncertainties. See “Special Note Regarding Forward-Looking Statements.” If any of the following risks are realized, our business, financial condition, operating results and prospects could be materially and adversely affected.
Summary of Risk Factors
| ● | We have a history of losses and expect to incur net losses in the future and may never achieve profitability. |
| ● | We might require substantial additional funding to continue our operations and to respond to business needs or take advantage of new business opportunities, which may not be available on acceptable terms, or at all. |
| ● | Our loan facility contains restrictions that limit our flexibility in operating our business, and if we fail to comply with the covenants and other obligations under our loan agreement, the lenders may be able to accelerate amounts owed under the facility and may foreclose upon the assets securing our obligations. |
| ● | We may engage in acquisitions that are not successful and which could disrupt our business, cause dilution to our stockholders and reduce our financial resources. |
| ● | Our operating results could be subject to significant fluctuation, which could increase the volatility of our stock price and cause losses to our shareholders. |
| ● | The molecular diagnostics industry is highly competitive and characterized by rapid technological changes and we may be unable to keep pace with our competitors. |
| ● | Our financial results are largely dependent on sales of two tests, Confirm mdx and GPS, and we will need to generate sufficient revenues from these tests and other future solutions to grow our business. |
| ● | We face uncertainties over the reimbursement of our tests by third party payors. |
| ● | Our business may be adversely affected by global macroeconomic conditions and volatility in the capital markets. |
| ● | If we are unable to retain intellectual property protection in relation to our tests or if we are required to expend significant resources to protect our intellectual property position, our competitive position could be undercut. |
| ● | We may be subject to substantial costs and liabilities or be prevented from using technologies incorporated in our tests as a result of litigation or other proceedings relating to patent rights. |
1
| ● | We rely on strategic collaborative and license arrangements with third parties to develop critical intellectual property. We may not be able to successfully establish and maintain such intellectual property. |
| ● | Due to billing complexities in the diagnostic and laboratory service industry, we may have difficulties receiving timely payment for the tests we perform, and may face write-offs, disputes with payors and patients, and long collection cycles. |
| ● | We face an inherent risk of product liability claims. |
| ● | Failure to attract or retain key personnel or to secure the support of key scientific collaborators could materially adversely impact our business. |
| ● | Our business and reputation will suffer if we are unable to establish and comply with, stringent quality standards to assure that the highest level of quality is observed in the performance of our tests. |
| ● | Our laboratory facilities may become inoperable due to natural or man-made disasters or regulatory sanctions. |
| ● | We rely on a limited number of third-party suppliers for services and items used in the production and operation of our testing solutions, and some of those services and items are supplied from a single source. Disruption of the supply chain, unavailability of third-party services required for the performance of the tests, modifications of certain items or failure to achieve economies of scale could have a material adverse effect on us. |
| ● | Failures in our information technology, storage systems, or our clinical laboratory equipment could significantly disrupt our operations and our research and development efforts. |
| ● | We expect to make significant investments to research and develop new tests, which may not be successful. |
| ● | Our research and development efforts will be hindered if we are not able to obtain samples, contract with third parties for access to samples or complete timely enrollment in future clinical trials. |
| ● | Failure to comply with governmental payor regulations could result in us being excluded from participation in Medicare, Medicaid or other governmental payor programs, which would adversely affect our business. |
| ● | Failure to comply with federal, state and foreign laboratory licensing and related requirements could cause us to lose the ability to perform our tests, experience disruptions to our business, or become subject to administrative or judicial sanctions. |
| ● | The FDA may change its position with respect to its regulation of the laboratory developed tests we offer or may seek to offer in the future, causing us to incur substantial costs and time delays associated with meeting requirements for pre-market clearance or approval or we could experience decreased demand for or reimbursement of our tests. |
| ● | Delays in receipt of, or failure to obtain, required FDA clearances or approvals for our products in development, or improvements to or expanded indications for our current offerings, could materially delay or prevent us from commercializing or otherwise adversely impact future product commercialization. |
2
| ● | We expect to rely on third parties to conduct any future studies of our technologies that may be required by the FDA or other U.S. or foreign regulatory bodies, and those third parties may not perform satisfactorily. |
| ● | We conduct business in a heavily regulated industry, and changes in regulations or violations of regulations may, directly or indirectly, adversely affect our results of operations and financial condition and harm our business. |
| ● | Our business is subject to various complex laws and regulations applicable to providers of clinical diagnostic products and services. |
| ● | Failure to comply with privacy, security, and consumer protection laws and regulations could result in fines, penalties and damage to our reputation and have a material adverse effect on our business. |
| ● | Our employees, independent contractors, consultants, commercial partners, and vendors may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements. |
| ● | Our operating results could be materially adversely affected by unanticipated changes in tax laws and regulations, adjustments to our tax provisions, exposure to additional tax liabilities, or forfeiture of our tax assets. |
| ● | Certain of our significant shareholders may have different interests from us and may be able to control us, including the outcome of shareholder votes. |
| ● | Holders of our ordinary shares should be aware that the rights provided to holders of our ordinary shares under Belgian corporate law and our Articles of Association differ in certain respects from the rights that you would typically enjoy as a shareholder of a U.S. company under applicable U.S. federal and state laws. |
| ● | Concentration of ownership of our ordinary shares among our existing executive officers, directors and principal shareholders may prevent holders of our ordinary shares from influencing significant corporate decisions. |
| ● | As a foreign private issuer and as permitted by the listing requirements of Nasdaq, we rely on certain home country corporate governance practices rather than the corporate governance requirements of Nasdaq. |
| ● | We may lose our foreign private issuer status in the future, which could result in significant additional costs and expenses. |
| ● | We incur significant costs as a result of operating as a company that is publicly listed on Nasdaq, and our management is required to devote substantial time to compliance initiatives. |
| ● | If we fail to implement and maintain effective internal controls over financial reporting, our ability to produce accurate financial statements on a timely basis could be impaired. |
Risks Related to Our Business and Industry
We have a history of losses and expect to incur net losses in the future and may never achieve profitability.
We have incurred substantial net losses since our inception, and there can be no assurance that we will achieve profitability. As of December 31, 2023, we had an accumulated deficit of $331.4 million and for the year ended December 31, 2023, we had a net loss of $43.1 million and net cash outflows from operating activities of $21.5 million. We expect our losses to continue as a result of costs relating to ongoing research and development and for increased selling and marketing costs for existing and planned testing solutions. These losses have had, and will continue to have, an adverse effect on our working capital, total assets, and stockholders’ equity. Even if we achieve significant revenues, we may not become profitable, and even if we achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis. Failure to become and remain consistently profitable could adversely affect the market price of our common stock and could significantly impair our ability to raise capital or expand our business in accordance with our growth strategy. Historically, we have been able to raise capital at regular occasions. If we are unable to continue to do this, our ability to operate as a going concern could be seriously compromised.
3
We may not be able to continue as a going concern.
Our consolidated financial statements have been prepared on a going concern basis, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business. We have a history of operating losses and management expects the Company to continue to incur net losses and have significant cash outflows for at least the next twelve months. While these conditions, among others, raise substantial doubt about our ability to continue as a going concern, these consolidated financial statements have been prepared assuming that the Company will continue as a going concern.
We may require substantial additional funding to continue our operations and to respond to business needs (including repayment of our outstanding debt) or take advantage of new business opportunities, which may not be available on acceptable terms, or at all.
Our capital outlays and operating expenditures are expected to increase over the next several years as commercial operations expand. We expect that we may require additional equity or debt funding from time to time in case of a shortfall in cash inflows from operations or to respond to business needs (including repayment of our outstanding debt) or take advantage of new business opportunities, which may not be available at acceptable terms, or at all. For more information about our cash and cash equivalent position or total liquidity position, see also Item 5B. “Liquidity and Capital Resources.”
Additionally, under the terms of the Asset Purchase Agreement pursuant to which we acquired the GPS prostate cancer business of Exact Sciences Corporation (“Exact Sciences”), we will be required to make additional earnout payments to Exact Sciences in 2025, 2026, and 2027, based upon a portion of the reported revenues attributable to the GPS business in each of 2024, 2025 and 2023, respectively, in an aggregate amount of up to $82.5 million , with the maximum earnout payable in 2025 not to exceed $40 million. At our option, the earnout amounts can be settled in cash or through the issuance of shares to Exact Sciences, provided that the aggregate number of shares held by Exact Sciences will not exceed more than 7.5% of our outstanding shares.
If additional funds are raised through the sale of equity, convertible debt or other equity-linked securities, our securityholders’ ownership will be diluted. Any equity securities issued also may provide for rights, preferences or privileges senior to those of holders of shares. If additional funds are raised by issuing debt securities, these debt securities would have rights, preferences and privileges senior to those of shareholders, and the terms of the debt securities issued could impose significant restrictions on our operations.
If adequate funds are not available, we may have to scale back our operations or limit our research and development activities, which may cause us to grow at a slower pace, or not at all, and our business could be adversely affected.
Our loan facility contains restrictions that limit our flexibility in operating our business, and if we fail to comply with the covenants and other obligations under our loan agreement, the lenders may be able to accelerate amounts owed under the facility and may foreclose upon the assets securing our obligations.
In August 2022, we entered into a $35 million loan and security agreement with an affiliate of Innovatus Capital Partners, LLC (“Innovatus”). Under this agreement, at the option of the Company, an additional $35 million can be drawn from Innovatus, consisting of a $20 million term B loan and a $15 million term C loan, which can be drawn in 2024 and 2025 respectively, subject to certain conditions. There can be no assurance that these conditions will be satisfied and that we will be able to draw any further term loan amounts under this facility.
The loans accrue interest at a floating per annum rate equal to the sum of (a) the greater of (i) the prime rate published in The Wall Street Journal in the “Money Rates” section or (ii) 4.00%, plus (b) 4.25%, and require interest-only payments for the initial four years. At our election, a portion of the interest may be payable in-kind by adding an amount equal to 2.25% of the outstanding principal amount to the then outstanding principal balance on a monthly basis until August 2025. The loans mature in August 2027. The lenders have the right to convert, prior to August 2025, up to 15% of the outstanding principal amount of the loans into shares of the Company at a price per share of $11.21.
The loans are secured by substantially all assets of the Company, including intellectual property related to our Confirm mdx, Select mdx and Genomic Prostate Score (“GPS”) tests. The loan agreement also subjects us to certain affirmative and negative covenants, including limitations on our ability to transfer or dispose of assets, merge with or acquire other companies, make investments, pay dividends, incur additional indebtedness and liens and conduct transactions with affiliates. As a result of these covenants, we have certain limitations on the manner in which we can conduct our business, and we may be restricted from engaging in favorable business activities or financing future operations or capital needs until our current debt obligations are paid in full or we obtain the consent of Innovatus, which we may not be able to obtain. We cannot be certain that we will be able to generate sufficient cash flow or revenue to meet the financial covenants or pay the principal and accrued interest on the debt.
4
In addition, upon the occurrence of an event of default or a mandatory prepayment event, Innovatus, among other things, can declare all indebtedness due and payable immediately, which would adversely impact liquidity and reduce the availability of cash flows to fund working capital needs, capital expenditures and other general corporate purposes. An event of default or a mandatory prepayment event includes, but is not limited to, our failure to pay any amount due and payable under the loan agreement, the breach of any representation or warranty in the loan agreement, the breach of any covenant in the loan agreement (subject to a cure period in some cases), a change of control as defined in the loan agreement, certain defaults on loan agreements with third parties and insolvency events and proceedings. If an event of default or a mandatory prepayment event occurs and we are unable to repay amounts due under the loan agreement, Innovatus could foreclose on substantially all of our assets, including intellectual property. We cannot be certain that future working capital, borrowings or equity financings will be available to repay or refinance our debt to Innovatus or any other debt we may incur in the future.
Furthermore, we must meet certain covenants in order to draw down additional term loans that may remain available under the $70 million Innovatus facility. If we do not have sufficient working capital to fund our earnout payment obligations to Exact Sciences if and when they become due, and we are also unable to meet loan covenants necessary to draw down one or more of the additional Innovatus term loans, we may be unable to satisfy our contractual obligations to Exact Sciences, resulting in a material breach under the Asset Purchase Agreement with Exact Sciences as well as an event of default under the loan agreement with Innovatus, and our business could be adversely affected.
Our acceptance of a Paycheck Protection Program loan subjects us to a variety of federal regulations and although we may apply for forgiveness of this loan, it may not be forgiven.
In April 2020, we qualified for a $2.3 million loan through the Paycheck Protection Program (the “PPP”) of the U.S. Coronavirus Aid, Relief and Economic Security Act (the “CARES Act”), under a loan agreement administered by the U.S. Small Business Administration. By participating in a federal loan program, we become subject to increased governmental oversight and federal regulatory compliance obligations, including potential civil and criminal liability for making false claims or statements under the U.S. False Claims Act, 31 USC. § 3729 et seq. (the “FCA”). Liability under the FCA and similar federal statutes can carry significant potential monetary penalties and potential jail time, and can arise from both “knowing” and “willful” misstatements. FCA violations will result in a civil penalty per false claim, of not less than $11,181 and not more than $22,363, plus treble the government’s actual damages. A person who violates § 3729 will also be held liable for the government’s costs for bringing a civil action to recover any penalty or damages. If, despite our good faith belief that we satisfied all eligibility requirements for the PPP loan, we are found to have been ineligible to receive the PPP loan or in violation of any of the laws or regulations that apply to us in connection with the PPP loan, we may be subject to penalties, including under the FCA, and could be required to repay the PPP loan. Additionally, a review or audit by the U.S. Small Business Administration (“SBA”) or other government entity in connection with any future forgiveness application (if we chose to apply for forgiveness) or claims under the False Claims Act could consume significant financial and management resources. Any of these events could harm our business, results of operations and financial condition.
We may engage in acquisitions that are not successful and which could disrupt our business, cause dilution to our stockholders and reduce our financial resources.
In addition to our acquisition of NovioGendix, a privately held company based in Nijmegen, The Netherlands, in September 2015 and our acquisition of our GPS test from Genomic Health, Inc., a subsidiary of Exact Sciences, in August 2022, we may enter into other transactions in the future to acquire other businesses, products or technologies. We may be unable to realize the anticipated benefits of the acquisitions or do so within the anticipated timeframe. Any acquisitions may not strengthen our competitive position, and these transactions may be viewed negatively by customers or investors. We could incur losses resulting from known or unknown liabilities of the acquired business that are not sufficiently covered by the indemnification we may obtain from the seller. In addition, we may not be able to successfully integrate the acquired personnel, technologies and operations into our existing business in an effective, timely and non-disruptive manner. If we are unable to do so, the disruption to our operations could result in additional costs or could distract management’s attention from other initiatives.
Our operating results could be subject to significant fluctuation, which could increase the volatility of our stock price and cause losses to our shareholders.
Our revenues and results of operations have historically fluctuated significantly and may do so in the future, depending on a variety of factors, including the following:
| ● | our success in marketing and selling, and changes in demand for, our tests, and the level of reimbursement and collection obtained for such tests; |
| ● | seasonal variations or non-seasonal events or circumstances affecting healthcare provider recommendations for our tests and patient compliance with healthcare provider recommendations, including without limitation, holidays, weather events, and circumstances such as the outbreak of influenza that may limit patient access to medical practices or institutions for diagnostic tests and preventive services; |
| ● | our success in collecting payments from third-party and other payors, patients and collaborative partners, variation in the timing of these payments and recognition of these payments as revenues; |
| ● | the pricing of our tests, including potential changes in the reimbursement rates for claims submitted to the U.S. Centers for Medicare & Medicaid Services (“CMS”) or other healthcare payors; |
5
| ● | circumstances affecting our ability to provide our tests, including weather events, supply shortages, or regulatory or other circumstances that adversely affect our ability to manufacture our tests or process tests in our clinical laboratories; |
| ● | fluctuations in the amount and timing of our selling and marketing costs and our ability to manage costs and expenses and effectively implement our business; and |
| ● | our research and development activities, including the timing, size, complexity, and cost of clinical studies. |
If our revenues or operating results fall below the expectations of investors or public market analysts, the trading price of our common stock could decline substantially.
The molecular diagnostics industry is highly competitive and characterized by rapid technological changes and we may be unable to keep pace with our competitors.
The molecular diagnostics field is characterized by rapid technological changes, frequent new product introductions, changing customer preferences, emerging competition, evolving industry and regulatory compliance standards, reimbursement uncertainty and price competition. Moreover, the molecular diagnostics field is intensely competitive both in terms of service and price, and continues to undergo significant consolidation, permitting larger clinical laboratory service providers to increase cost efficiencies and service levels, resulting in more intense competition.
The market for assessing men at risk for prostate cancer diagnosis or aggressiveness is large. As a result, this market has attracted competitors, some of which possess substantially greater financial, selling, logistical and laboratory resources, more experience in dealing with third-party payors, and greater market penetration, purchasing power and marketing budgets, as well as more experience in providing diagnostic services. Some companies and institutions are developing serum-based tests and diagnostic tests based on the detection of proteins, nucleic acids or the presence of fragments of mutated genes in the blood that are associated with prostate cancer. These competitors could have technological, financial, reputational, and market access advantages over us.
Some of our current and potential competitors may have significant competitive advantages over us, which may make them more attractive to hospitals, clinics, group purchasing organizations and physicians. See “Item 4.B. Business Overview — Competition” in this Annual Report on Form 20-F for additional information regarding our competitors and the effects of competition on our business.
We may be unable to compete effectively against our competitors either because their products and services are superior or because they are more effective in developing or commercializing competing products and services. Furthermore, even if we do develop new marketable products or services, our current and future competitors may develop products and services that are more clinically or commercially attractive than ours, and they may bring those products and services to market earlier or more effectively than us. If we are unable to compete successfully against current or future competitors, we may be unable to increase market acceptance for, and sales of, our tests, which could prevent us from increasing or sustaining our revenues or achieving sustained profitability and could cause the market price of our common stock to decline.
Our commercial success will depend on the market acceptance and adoption of our current and future tests.
Healthcare providers typically take a long time to adopt new products, testing practices and clinical treatments, partly because of perceived liability risks and the uncertainty of third-party coverage and reimbursement. It is critical to the success of our sales efforts that we educate enough patients, clinicians and administrators about molecular diagnostics testing, in general, as well as about our testing solutions, and demonstrate their clinical benefits. It is likely that clinicians may not adopt, and third-party payors may not cover or adequately reimburse for, our tests unless they determine, based on published peer-reviewed journal articles and the experience of other clinicians, that they provide accurate, reliable, and cost-effective information.
As the healthcare reimbursement system in the United States evolves to place greater emphasis on comparative effectiveness and outcomes data, we cannot predict whether we will have sufficient data, or whether the data we have will be presented to the satisfaction of any payors seeking such data, in the process of determining and maintaining coverage for our diagnostic tests. The administration of clinical and economic utility studies is expensive and demands significant attention from the management team. There can be no assurance that our clinical studies will be successfully initiated, enrolled or completed. Also, data collected from these studies may not be positive or consistent with our existing data or may not be statistically significant or compelling to the medical community. If the results obtained from ongoing or future studies are inconsistent with certain results obtained from previous studies, adoption of diagnostic services would suffer, and our business would be harmed.
If our tests or the technology underlying our current or future tests do not receive sufficient favorable exposure in peer-reviewed publications, the rate of clinician adoption of our tests and positive reimbursement coverage decisions for our tests could be negatively affected. See “Risk Factors — We face uncertainties over the reimbursement of our tests by third party payors.” The publication of clinical data in peer-reviewed journals is a crucial step in commercializing and obtaining reimbursement for diagnostic tests, and our inability to control when, if ever, our results are published may delay or limit our ability to derive sufficient revenue from any product that is the subject of a study.
6
Our financial results are largely dependent on sales of two tests, Confirm mdx and GPS, and we will need to generate sufficient revenues from these tests and other future solutions to grow our business.
Currently, we rely significantly on the sales of Confirm mdx and GPS tests in the United States for our revenues, with these tests combined accounting for approximately 79% of total revenue in 2023 and 84% of total revenue in 2022. In 2021, which was prior to our acquisition of the GPS test, Confirm mdx accounted for approximately 91% of total revenues.
We have diversified our revenue through the launch and commercialization of additional precision diagnostic test offerings, including our Select mdx and Resolve mdx tests. However, sales of Confirm mdx and GPS are expected to continue to account for a substantial portion of total revenues for at least the next several years. If reimbursement for our tests were to be revoked or limited either by CMS or commercial payors, this could have an immediate impact on our revenues. While we do not believe that revocation of Medicare reimbursement for Confirm mdx or GPS tests is likely, if this were to occur, the impact could be severe.
The commercial success of our testing solutions and our ability to generate sales will depend on several factors, including:
| ● | acceptance by the medical community; |
| ● | the number of patients undergoing a prostate biopsy procedure; |
| ● | acceptance, endorsement and formal policy approval of favorable reimbursement for the test by Medicare and other third-party payors; |
| ● | our ability to successfully market the tests; |
| ● | the amount of and nature of competition from other prostate cancer products and procedures; |
| ● | maintaining and defending patent protection for the intellectual property relevant to our products and services; and |
| ● | our ability to establish and maintain adequate commercial distribution, sales force and laboratory testing capabilities. |
If we are unable to increase sales and reimbursement of our current testing solutions or successfully develop and commercialize other solutions or enhancements, our revenues and our ability to achieve profitability would be impaired, and the market price of our shares could decline.
We face uncertainties concerning the coverage and reimbursement of our tests by third-party payors.
Successful commercialization of our testing solutions depends, in large part, on the availability of coverage and adequate reimbursement from government and private payors. Favorable third-party payor coverage and reimbursement are essential to meeting our immediate objectives and long-term commercial goals. In the United States, for new diagnostic tests, each private and government payor decides whether to cover the test, the amount it will reimburse clinical laboratories or other providers for a covered test, and any specific conditions for coverage and reimbursement. Healthcare providers may be unlikely to order a specific diagnostic test unless an applicable third-party payor offers meaningful reimbursement for the test. Therefore, adequate coverage and reimbursement is critical to the commercial success of a diagnostic product, and if we are unable to secure and maintain favorable coverage determinations and reimbursement, this will undermine our ability to earn revenue from our products.
Medicare
Reimbursement for diagnostic tests furnished to Medicare beneficiaries (typically patients aged 65 or older) is usually based on a fee schedule set by the CMS, a division of the U.S. Department of Health and Human Services (“HHS”). As a Medicare-enrolled provider with our primary laboratory based in California, we bill Noridian Healthcare Solutions (“Noridian”), the Medicare Administrative Contractor (“MAC”) for California, and our Select mdx, Confirm mdx, and GPS tests are subject to Noridian’s local coverage and reimbursement policies. Noridian participates in the Molecular Diagnostic Services Program (“MolDX”), administered by Palmetto GBA, which handles technical assessments for U.S. laboratories that perform molecular diagnostic testing. The Confirm mdx test obtained a positive Medicare local coverage determination (“LCD”) under the MolDX program in 2014, the GPS test obtained a positive Medicare coverage LCD in 2015, and the Select mdx test obtained a positive Medicare coverage LCD in April 2023, each of which provides coverage for Medicare patients throughout the United States. As a Medicare-enrolled provider with a secondary laboratory based in Texas, we bill Novitas Solutions (“Novitas”), the MAC covering Texas, for tests that are offered by our Texas laboratory, including our Resolve mdx test, claims for which tests are therefore subject to Novitas’ local coverage and reimbursement policies. Novitas does not at this time participate in the MolDx program, nor does it in practice issue LCDs for all molecular tests that it may reimburse. As a result, molecular tests offered by our Texas laboratory may in certain cases be billed to Novitas using industry-standard coding terms that describe the procedures performed, pursuant to guidance and instructions set forth by the American Medical Association “CPT codebook” as well as associated guidance set forth in the Policy Manual for Medicare Services published by the National Correct Coding Initiative (NCCI). Because of the highly technical nature of interpreting complex and disparate coding policies applicable to certain of the Company’s tests, there can be no assurance that Medicare, or the MACs that determine local Medicare coverage, will continue to issue or follow positive coverage and reimbursement policies and practices and, if issued or followed, that such policies or reimbursement practices will be maintained in the future.
Medicare accounted for approximately 45% of MDxHealth’s revenues in 2023, compared to 43% in 2022. See Note 4 in the Notes to Consolidated Financial Statements included in Part III for further detail.
7
Commercial payors
Obtaining coverage and reimbursement by commercial payors is a time-consuming and costly process, without a guaranteed outcome, since each commercial payor makes its own decision with respect to whether to cover a particular test and, if so, at what rate to reimburse providers for that test. In addition, several payors and other entities conduct technology assessments of new medical tests and devices and provide the results of these assessments for informational purposes to other parties. These assessments may be used by third-party payors and healthcare providers as grounds to deny coverage for a particular test, or to refuse to use or order a particular test or procedure. Our tests have received initial negative technology assessments from several of these entities and are likely to receive more negative technology assessments. We continue to work with third-party payors to obtain coverage and reimbursement for our tests and to appeal coverage denial decisions based on existing and ongoing studies, peer reviewed publications, and support from physician and patient groups. There are no assurances that commercial payors will continue to issue positive coverage and reimbursement policies and/or contracts and, if issued, that such policies and/or contracts will be maintained in the future. If our tests are considered on a policy-wide level by major third-party payors, whether at our request or on the payor’s own initiative, and the payor determines that such tests are ineligible for coverage and reimbursement, our revenue potential could be adversely impacted.
Our business may be adversely affected by global macroeconomic conditions and volatility in the capital markets.
The growth of our business is, and will continue to be, affected by changes in the overall global economy. Various macroeconomic factors could adversely affect our business and the results of our operations and financial condition, including changes in inflation, high interest rates, foreign currency exchange rates, weakness in general economic conditions and threatened or actual recessions, including those resulting from the current and future conditions in the global financial markets, and budgeting constraints of governmental entities. Cost inflation, including increases in raw material prices, labor rates, and transportation costs, may continue to impact our profitability. Our ability to recover these cost increases through price increases is significantly limited by the process by which we are reimbursed for our products and services by government and private payors. In addition, disruptions in the U.S., Europe or other economies, including due to geopolitical conflict, could disrupt global markets, interrupt global supply chains, and have other potential inflationary or recessionary effects on the global economy.
The volatility of the capital markets could also affect the value of our investments and our ability to liquidate our investments in order to fund our operations. The high interest rate environment and reduced access to capital markets could also adversely affect the ability of our suppliers, distributors, licensors, collaborators, contract manufacturers and other commercial partners to remain effective business partners or to remain in business. The loss of a critical business partner, or a failure to perform by a critical business partner, could have a disruptive effect on our business and could adversely affect our results of operations.
Public health crises, such as the COVID-19 pandemic, have had, and could in the future have, adverse effects on our business and financial results.
Pandemics or disease outbreaks, such as the COVID-19 pandemic, have created and may continue to create significant volatility, uncertainty and economic disruption in the markets in which we sell or plan to sell our current or future tests and in which we operate, and may negatively impact business and healthcare activity globally. For example, in response to the COVID-19 pandemic, governments around the world imposed measures designed to reduce the transmission of COVID-19, patients postponed visits to healthcare providers, certain healthcare providers temporarily closed their offices or restricted patient visits, healthcare provider employees became generally unavailable and there were disruptions in the operations of payors, suppliers and other third parties that are necessary for our tests to be administered. The extent to which fear of exposure to or actual effects of COVID-19, new variants, disease outbreak, epidemic or a similar widespread health concern impacts our business will depend on future developments, which are highly uncertain and cannot be predicted with confidence, such as the speed and extent of geographic spread of the disease, the duration of the outbreak, travel restrictions, the efficacy of vaccination and treatment; impact on the U.S. and international healthcare systems, the U.S. economy and worldwide economy; the timing, scope and effectiveness of U.S. and international governmental response; and the impact on the health, well-being and productivity of our employees; and short- and long-term changes in the behaviors of medical professionals and patients resulting from any such pandemic, outbreak, epidemic or other health concern.
Risks Related to Our Intellectual Property
If we are unable to retain intellectual property protection in relation to our tests or if we are required to expend significant resources to protect our intellectual property position, our competitive position could be undercut.
Our ability to protect our discoveries, know-how and technologies affects our ability to compete and to achieve profitability. We rely on a combination of U.S. and foreign patents and patent applications, copyrights, trademarks and trademark applications, confidentiality or non-disclosure agreements, material transfer agreements, licenses and consulting agreements to protect our intellectual property rights. We also maintain certain company know-how, algorithms, and technological innovations designed to provide us with a competitive advantage in the marketplace as trade secrets. As of April 1, 2024, we owned or had exclusive rights to more than 18 patent families related to our molecular technology and cancer-specific biomarkers. Specifically, there are 198 granted or pending patent applications in this group comprised of 15 issued or allowed U.S. patents, 5 pending U.S. provisional or non-provisional applications, 8 pending international patent applications filed under the Patent Cooperation Treaty (“PCT”) and 170 granted or allowed patents in jurisdictions outside the United States, including Japan, Canada, Israel and the major European countries. Our issued U.S. patents expire at various times between 2024 and 2036. Of these issued patents, 20 cover intellectual property used in our Select mdx test, the last of which expires in 2036, and 97 cover intellectual property used in our GPS test, the last of which expires in 2038. When these patents expire other companies will no longer be prohibited from incorporating the subject intellectual property into competing tests they may seek to develop.
8
While we intend to pursue additional and future patent applications, it is possible that pending patent applications and any future applications may not result in issued patents. Even if patents are issued, third parties may independently develop similar or competing technology that avoids our patents. Third parties may also assert infringement or other intellectual property claims against us or against our licensors, licensees, suppliers or strategic partners. Any actions regarding patents could be costly and time-consuming and could divert the attention of management and key personnel from other areas of our business. Further, we cannot be certain that the steps we have taken will prevent the misappropriation of our trade secrets and other confidential information as well as the misuse of our patents and other intellectual property, particularly in foreign countries with no patent protection.
Although we have licensed and own issued patents in the United States and foreign countries, we cannot be certain the claims will continue to be considered patentable by the U.S. Patent and Trademark Office (the “USPTO”), U.S. courts patent offices and courts in other jurisdictions. The U.S. Supreme Court, other federal courts and/or the USPTO, may change the standards of patentability and any such changes could have a negative impact on our business. For instance, the Federal Circuit has recently ruled on several patent cases, such as Univ. of Utah Research Found. v. Ambry Genetics Corp., 774 F.3d 755 (Fed. Cir. 2014), Ariosa Diagnostics, Inc. v. Sequenom, Inc., 788 F.3d 1371 (Fed. Cir. 2015), Genetic Tech. Ltd. v. Merial LLC, 818 F.3d 1369 (Fed. Cir. 2016), and Cleveland Clinic Found. v. True Health Diagnostics, 859 F.3d 1352 (Fed. Cir. 2017), that some diagnostic method claims are not patent eligible. These decisions have narrowed the scope of patent protection available in certain circumstances or weakened the rights of patent owners in certain situations. Some aspects of our technology involve processes that may be subject to this evolving standard and we cannot guarantee that any of our issued or pending process claims will be patentable as a result of such evolving standards. In addition, this combination of decisions has created uncertainty as to the value of certain issued patents, in particular in the detection of prostate cancer and other cancers.
We may be subject to substantial costs and liabilities or be prevented from using technologies incorporated in our tests as a result of litigation or other proceedings relating to patent rights.
Third parties may assert infringement or other intellectual property claims against us or our licensors, licensees, suppliers or strategic partners. We pursue a patent strategy that we believe provides us with a competitive advantage in the assessment of prostate cancer and is designed to maximize patent protection against third parties in the United States and, potentially, in certain foreign countries. In order to protect or enforce our patent rights, we may have to initiate actions against third parties. Any actions regarding patents could be costly and time-consuming and could divert the attention of management and key personnel from other areas of our business. Additionally, such actions could result in challenges to the validity or applicability of our patents. Because the USPTO maintains patent applications in secrecy until a patent application is published or the patent is issued, we have no way of knowing if others may have filed patent applications covering technologies used by us or our partners. Additionally, there may be third-party patents, patent applications and other intellectual property relevant our technologies that may block or compete with our technologies. Even if third-party claims are without merit, defending a lawsuit may result in substantial expense to us and may divert the attention of management and key personnel. In addition, we cannot provide assurance that we would prevail in any such suits or that the damages or other remedies, if any, awarded against us would not be substantial. Claims of intellectual property infringement may require us, or our strategic partners, to enter into royalty or license agreements with third parties that may not be available on acceptable terms, if at all. These claims may also result in injunctions which could prevent us from further developing and commercializing services or products containing our technologies, which could in turn adversely affect our ability to earn revenues from these services or products.
Also, patents and patent applications owned by us may become the subject of post-grant challenges or interference proceedings in the USPTO to determine validity and the priority of invention, which could result in substantial cost as well as a possible adverse decision as to the validity or priority of invention of the patent or patent application involved. An adverse decision in an interference proceeding may result in the loss of rights under a patent or patent application subject to such a proceeding.
Ultimately, the potential weakening of our intellectual property position as a result of the evolution of case law or otherwise may make us more vulnerable to competition. While we are unable to quantify the impact of this risk given that our patents remain untested in the courts, the impact could be severe if our competitors are able to take advantage of any weakening of our intellectual property position.
We rely on strategic collaborative and license arrangements with third parties to develop critical intellectual property. We may not be able to successfully establish and maintain such intellectual property.
The development and commercialization of our products and services rely, directly or indirectly, upon strategic collaborations and license agreements with third parties. We have a license agreement with an academic institution pursuant to which we have incorporated licensed technology into our Confirm mdx test and may incorporate licensed technology into our pipeline products. Our dependence on license, collaboration and other similar agreements with third parties may subject us to a number of risks. There can be no assurance that any current contractual arrangements between us and third parties or between our strategic partners and other third parties will be continued on materially similar terms and will not be breached or terminated early. Any failure to obtain or retain the rights to necessary technologies on acceptable commercial terms could require us to re-configure our products and services, which could negatively impact their commercial sale or increase the associated costs, either of which could materially harm our business and adversely affect our future revenues and ability to achieve sustained profitability.
9
We expect to continue and expand our reliance on collaboration and license arrangements. Establishing new strategic collaborations and license arrangements is difficult and time-consuming. Discussions with potential collaborators or licensors may not lead to the establishment of collaborations on favorable terms, if at all. To the extent we agree to work exclusively with one collaborator in a given area, our opportunities to collaborate with other entities could be limited. Potential collaborators or licensors may reject collaborations with us based upon their assessment of our financial, regulatory or intellectual property position or other factors. Even if we successfully establish new collaborations, these relationships may never result in the successful commercialization of any product or service. In addition, the success of the projects that require collaboration with third parties will be dependent on the continued success of such collaborators. There is no guarantee that our collaborators will continue to be successful and, as a result, we may expend considerable time and resources developing products or services that will not ultimately be commercialized.
If patent regulations or standards are modified, such changes could have a negative impact on our business.
From time to time, the U.S. Supreme Court, other federal courts, the U.S. Congress, or the USPTO may change the standards of patentability and validity of patents within the cancer screening and diagnostics space, and any such changes could have a negative impact on our business.
There have been several cases involving “gene patents” and diagnostic claims that have been considered by the U.S. Supreme Court that have affected the legal concept of subject matter eligibility by seemingly narrowing the scope of the statute defining patentable inventions.
Additionally, in December 2014 and again in 2019, the USPTO published revised guidelines for patent examiners to apply when examining process claims that narrow the scope of patentable subject matter. While these guidelines may be subject to review and modification by the USPTO over time, we cannot assure you that our patent portfolio will not be negatively impacted by the decisions mentioned above, rulings in other cases, or changes in guidance or procedures issued by the USPTO.
Additional substantive changes to patent law, whether new or associated with the America Invents Act — which substantially revised the U.S. patent system — may affect our ability to obtain, enforce or defend our patents. Accordingly, it is not clear what, if any, impact these substantive changes will ultimately have on the cost of prosecuting our patent applications, our ability to obtain patents based on our discoveries, and our ability to enforce or defend our issued patents, all of which could have a material adverse effect on our business.
Risks Related to Our Operations
Due to billing complexities in the diagnostic and laboratory service industry, we may have difficulties receiving timely payment for the tests we perform, and may face write-offs, disputes with payors and patients, and long collection cycles.
Billing for diagnostic and laboratory services is a complex process. We bill many different payors including patients, private insurance companies, Medicare, Medicaid, and employer groups, all of which have different billing requirements.
We are often obligated to bill services in the specific manner required by each particular third-party payor. Failure to comply with these complex billing requirements (including complex federal and state regulations related to billing government health care programs, e.g., Medicare and Medicaid) may significantly hinder our collection and retention efforts, including not only potential write-offs of doubtful accounts and long collection cycles for accounts receivable, but also the potential disgorgement of previously paid claims based on third-party payor program integrity investigations into billing discrepancies, fraud, waste and abuse. With CMS’s recent implementation of a comprehensive oversight regime that consolidated program integrity powers into a single Unified Program Integrity Contractor (“UPIC”), audit and investigatory activity into potential billing fraud, waste and abuse in the industry has in recent years significantly increased. Responding to requests from a UPIC, or other auditor, is often time-consuming and requires dedication of internal, and sometimes external, resources. UPICs also have the authority to implement Medicare payment suspensions during the pendency of an audit, which could significantly impact cash flows, even where no improper billing is ultimately found to have occurred. Commercial payors may also engage in audit activity, requiring timely production of medical documentation in support of billed claims.
We may face patient dissatisfaction, complaints or lawsuits, including to the extent our tests are not fully covered by insurers and patients become responsible for all or part of the price of the test. As a result, patient demand for our tests could be adversely affected. To the extent patients express dissatisfaction with our billing practices to their healthcare providers, those healthcare providers may be less likely to prescribe our tests for other patients, and our business would be adversely affected.
Even if payors agree to cover our tests, our billing and collections process may be complicated by the following and other factors, which may be beyond our control:
| ● | complex and disparate reimbursement rules and requirements; |
| ● | disputes among payors as to which payor is responsible for payment; |
| ● | disparity in coverage among various payors or among various healthcare plans offered by a single payor; |
| ● | payor medical management requirements, including prior authorization requirements; |
| ● | differing information and billing requirements among payors; |
10
| ● | failure by patients or healthcare providers to provide complete and correct billing information; and |
| ● | limitations and requirement for patient billing, including those related to deductibles, co-payments, and co-insurance originating from contracts with commercial payors. |
Similarly, when we have a contract with a commercial payor to cover our tests, we are not permitted to bill patients insured by that payor for amounts beyond deductibles, co-payments, and co-insurance as prescribed in the coverage agreement between the payor and the patients. Therefore, when such contracted payors do not pay us our full, contracted rate for a test, for example, for failure to satisfy prior-authorization or other payor medical management requirements, we may not be permitted to collect the balance from the patient and our business is adversely impacted.
As a result of the above, we may face write-offs of doubtful accounts, payment suspensions and disgorgement of previously paid claims, disputes with payors and patients, and long collection cycles.
We face an inherent risk of product liability claims.
The marketing, sale and use of our tests could lead to product or professional liability claims against us if someone were to allege that our tests failed to perform as they were designed, or if someone were to misinterpret test results or improperly rely on them for clinical decisions. Although we maintain product and professional liability insurance which is deemed to be appropriate and adequate, it may not fully protect us from the financial impact of defending against product liability or professional liability claims or any judgments, fines or settlement costs arising out of any such claims. Furthermore, any product liability lawsuit, with or without merit, could increase our insurance rates or prevent us from securing insurance coverage in the future. Additionally, any product liability lawsuit could harm our reputation, which could impact our results of operations, or cause collaboration partners to terminate existing agreements and potential partners to seek alternate partners, any of which could negatively impact our results of operations.
Failure to attract or retain key personnel or to secure the support of key scientific collaborators could materially adversely impact our business.
Our success in implementing our business strategy depends largely on the skills, experience, and performance of key members of our executive management team and others in key management positions, including Michael McGarrity, our Chief Executive Officer. The collective efforts of our executive management team are critical to us as we continue to develop our technologies, tests, and R&D and sales programs. The loss or incapacity of existing members of our executive management team could adversely affect our operations. If we were to lose one or more of these key employees, we could experience difficulties in finding qualified successors, competing effectively, developing our technologies and implementing our business strategy. Our executives have employment agreements; however, the existence of an employment agreement does not guarantee retention of members of our executive management team. We do not maintain “key person” life insurance on any of our employees.
We have established relationships with leading key opinion leaders and scientists at important research and academic institutions that we believe are key to establishing tests using our technologies as a standard of care for cancer assessment and diagnosis. If our collaborators determine that cancer testing using our technologies are not appropriate options for prostate cancer diagnosis, or superior to available prostate cancer methods, or that alternative technologies would be more effective in the early diagnosis of prostate cancer, we would encounter significant difficulty establishing tests using our technologies as a standard of care for prostate cancer diagnosis, which would limit our revenue growth and profitability.
Our results of operations can be adversely affected by labor shortages, turnover and labor cost increases.
Labor is a significant component of operating our business. A number of factors may adversely affect the labor force available to us or increase labor costs, including high employment levels, federal unemployment subsidies, increased wages offered by other employers, vaccine mandates and other government regulations and our responses thereto. As more employers offer remote work, we may have more difficulty recruiting for jobs that require on-site attendance, such as certain clinical laboratory and sales roles. Although we have not experienced any material labor shortage to date, we have recently observed an overall tightening and increasingly competitive labor market. A sustained labor shortage or increased turnover rates within our employee base, caused by a pandemic or as a result of general macroeconomic factors, could lead to increased costs, such as increased overtime or financial incentives to meet demand and increased wage rates to attract and retain employees, and could negatively affect our ability to efficiently operate our clinical laboratories and overall business. If we are unable to hire and retain employees capable of performing at a high-level, or if mitigation measures we may take to respond to a decrease in labor availability have unintended negative effects, our business could be adversely affected.
Additionally, the operations of our vendors and partners could also suffer from labor shortages, turnover and labor cost increases which could result in supply change disruptions and increases in the costs of the products and services we purchase, each of which could adversely affect our operations.
Our business and reputation will suffer if we are unable to establish and comply with stringent quality standards to assure that the highest level of quality is observed in the performance of our tests.
Inherent risks are involved in providing and marketing cancer tests and related services. Patients and healthcare providers rely on us to provide accurate clinical and diagnostic information that may be used to make critical healthcare decisions. As such, users of our testing solutions may have a greater sensitivity to errors than users of some other types of products and services.
11
Past or future performance or accuracy defects, incomplete or improper quality and process controls, excessively slow turnaround times, unanticipated uses of our tests or mishandling of samples or test results (whether by us, patients, healthcare providers, courier delivery services or others) can lead to adverse outcomes for patients and interruptions to our services. These events could lead to voluntary or legally mandated safety alerts relating to our tests or our laboratory facilities and could result in the removal of our products and services from the market or the suspension of our laboratories’ operations. Insufficient quality controls and any resulting negative outcomes could result in significant costs and litigation, as well as negative publicity that could reduce demand for our tests and payors’ willingness to cover our tests. Even if we maintain adequate controls and procedures, damaging and costly errors may occur.
Our laboratory facilities may become inoperable due to natural or man-made disasters or regulatory sanctions.
We currently perform testing services in our laboratory facilities located in Irvine, California and Plano, Texas, and our GPS test is currently performed by Exact Sciences in its laboratory facility in Redwood City, California. These laboratory facilities could become inoperable due to circumstances that may be beyond our control, and such inoperability could adversely affect our business and operations. The facilities, equipment and other business process systems would be costly to replace and could require substantial time to repair or replace. The inability to perform our tests or the backlog of tests that could develop if any of our facilities become inoperable for even a short period of time may result in the loss of customers or harm our reputation, and we may be unable to regain those customers or rebuild our reputation in the future. Although we possess insurance for damage to our property and the disruption of our business, this insurance may not be sufficient to cover all of our potential losses and may not continue to be available to us on acceptable terms, if at all.
The facilities may be damaged or destroyed by natural or man-made disasters, including earthquakes, wildfires, floods, outbreak of disease, acts of terrorism or other criminal activities and power outages, which may render it difficult or impossible for us to perform our tests for some period.
The facilities may also be rendered inoperable because of regulatory sanction. In the United States, we are subject to federal and state laws and regulations regarding the operation of clinical laboratories. Our U.S. laboratory facilities in Irvine, California and Plano, Texas are certified under the Clinical Laboratory Improvement Amendments (“CLIA”). CLIA and the laws of California and certain other states, impose certification requirements for clinical laboratories, and establish standards for quality assurance and quality control, among other things. Clinical laboratories are subject to inspection by regulators, and to sanctions for failing to comply with applicable requirements. Sanctions available under CLIA include prohibiting a laboratory from running tests, requiring a laboratory to implement a corrective action plan, and imposing civil monetary penalties. Our U.S. laboratory facilities hold certificates of accreditation from CMS to perform high-complexity testing. To renew these certificates, the facilities are subject to survey and inspection every two years. We also hold a certificate of accreditation from the College of American Pathologists (“CAP”), which sets standards that are higher than those contained in the CLIA regulations. CAP is an independent, non-governmental organization of board-certified pathologists that accredits laboratories nationwide on a voluntary basis. Sanctions for failure to comply with CAP or CLIA requirements, including proficiency testing violations, may include suspension, revocation, or limitation of a laboratory’s CLIA certificate, which is necessary to conduct business, as well as the imposition of significant fines or criminal penalties. In addition, our U.S. facilities are subject to regulation under state laws and regulations governing laboratory licensure. Certain states have enacted state licensure laws that are more stringent than CLIA. Failure to maintain CLIA certification, CAP accreditation, or required state licenses could have a material adverse effect on the sales of our tests and results of operations. Many states maintain independent licensure, registration, or certification procedures with which our U.S. facilities must maintain compliance in order to receive and test samples from that location. Maintaining compliance with the myriad of governmental requirements is time and resource intensive, and failure to maintain compliance could result in sanctions.
In order to rely on a third party to perform certain of our tests, we could only use another facility with established state licensure and CLIA accreditation following validation and other required procedures. We cannot assure you that we would be able to find another CLIA certified facility willing to comply with the required procedures, that this laboratory would be willing to perform the tests for us on commercially reasonable terms, or that it would be able to meet our quality or regulatory standards. Alternatively, establishing a redundant facility for certain of our testing would require considerable time and money to secure adequate space, construct the facility, recruit and train employees, and establish the additional operational and administrative infrastructure necessary to support this facility. We also may not be able, or it may take considerable time, to replicate our testing processes or results in a new facility. Additionally, any such new facility would be subject to certification under CLIA and licensing by several states, including California and New York, which could take a significant amount of time and result in delays in our ability to resume operations.
12
We rely on a limited number of third-party suppliers for services and items used in the production and operation of our testing solutions, and some of those services and items are supplied from a single source. Disruption of the supply chain, unavailability of third-party services required for the performance of the tests, modifications of certain items or failure to achieve economies of scale could have a material adverse effect on us.
To provide our testing services, we are required to obtain customized components and services that are currently available from a limited number of sources. Most of these components and services are sourced externally from approximately 40 external suppliers. Many of the consumable supplies and reagents used as raw materials in our testing process are procured from a limited number of suppliers, some of which are single source. In addition, we rely on a limited number of suppliers, or in some cases a single supplier (for example, for the automation of our deparaffination steps for our Confirm mdx test), for certain services and equipment with which we provide testing services. If we have to switch to a replacement supplier for any of these items that are sub-components or for certain services required for the performance of our tests, or if we have to commence our own manufacturing or testing services to satisfy market demand, we may face delays. For example, in the past, a supplier has delivered critical non-conforming components that failed our acceptance testing, requiring us to audit the supplier and assist the supplier in improving its internal quality processes. In addition, third party suppliers may be subject to circumstances which impact their ability to supply, including enforcement action by regulatory authorities, natural disasters, epidemics, labor disputes, financial difficulties including insolvency, among a variety of other internal or external factors. Any such supply disruptions could in turn result in service disruptions for an extended period of time, which could delay completion of our clinical studies or commercialization activities and prevent us from achieving or maintaining profitability. While we were able to qualify alternative suppliers to address COVID-19 related disruptions, in the future alternative suppliers may be unavailable, may be unwilling to supply, may not have the necessary regulatory approvals, or may not have in place an adequate quality management systems.
Exact Sciences currently performs our GPS test pursuant to a reference laboratory services agreement we entered into with Exact Sciences in connection with our acquisition of the GPS test. Until we are able to transition performance of the GPS test to our laboratory facilities we will be reliant on this contractual relationship to generate revenues from this test.
Modifications to a service or items, such as modifications to the assembly and packaging of items for our testing services supplied to healthcare providers, or inclusions of certain services or items made by a third-party supplier could require new approvals from the relevant regulatory authorities before the modified service or item may be used. While we have not experienced any material supply chain disruptions to date, if we were to experience such disruptions it could have an immediate impact on revenues, and the impact could be material depending on the length of the supply disruption.
Failures in our information technology, storage systems, or our clinical laboratory equipment could significantly disrupt our operations and our research and development efforts.
Our ability to execute our business strategy depends, in part, on the continued and uninterrupted performance of our information technology (“IT”) systems, which support our operations, including at our clinical laboratories, and our research and development efforts. We depend on our IT systems to receive and process test orders, securely store patient health records and deliver the results of our tests. The integrity and protection of our own data, and that of our customers and employees, is critical to our business. IT systems are vulnerable to damage from a variety of sources, including telecommunications or network failures, malicious human acts from criminal hackers, hacktivists, state-sponsored intrusions, industrial espionage and employee malfeasance, breaches due to employee error and natural disasters. Moreover, despite network security and back-up measures, some of our servers are potentially vulnerable to physical or electronic break-ins, computer viruses, and similar disruptive problems. Cyber-attacks are becoming more sophisticated and frequent, and in some cases have caused significant harm at other companies.
We face four primary risks relative to protecting sensitive and critical personally identifiable information, intellectual property or other proprietary business information about our customers, payors, recipients and collaboration partners, including test results: (1) loss of access risk, (2) inappropriate disclosure or access risk, (3) inappropriate modification risk, and (4) the risk of being unable to identify and audit controls over the first three risks. While we devote significant resources to protect the security of our IT systems, including the personal data and other information that we receive and store, there can be no assurance that any security measures will be effective against current or future security threats. We have experienced and expect to continue to experience attempted cyber-attacks on our IT systems and networks. To date, none of these attempted cyber-attacks has had a material effect on our operations or financial condition. However, any such breach or interruption could compromise our networks and the information stored therein could be accessed by unauthorized parties, publicly disclosed, lost or stolen. Despite the precautionary measures we have taken to prevent unanticipated problems that could affect our IT systems, unauthorized access, loss or disclosure could also disrupt our operations, including our ability to:
| ● | process tests, provide test results, bill payors or patients; |
| ● | process claims and appeals; |
| ● | provide customer assistance services; |
| ● | conduct research and development activities; |
| ● | collect, process and prepare company financial information; |
13
| ● | provide information about our tests and other patient and healthcare provider education and outreach efforts through our website; and |
| ● | manage the administrative aspects of our business. |
Any such access, disclosure or other loss of information could result in legal claims or proceedings, liability under laws that protect the privacy of personal information, such as the Health Insurance Portability and Accountability Act of 1996 (“HIPAA”), as amended by the Health Information Technology for Economic and Clinical Health Act (“HITECH”), similar U.S. state data protection regulations, the European Union’s General Data Protection Regulation (“GDPR”), and other regulations, the breach of which could result in significant penalties and damage to our reputation. In addition, disruptions to our business occurring as a result of system updates and enhancements, such as our efforts to move our precision oncology tests to our technology and services platform, could have a material adverse effect on our financial condition and operating results. There can be no assurance that our process of improving existing systems, developing new systems to support our expanding operations, protecting confidential patient information, and improving service levels will not be delayed or will not give rise to additional systems issues in the future. Although we carry insurance for this purpose, failure to adequately protect and maintain the integrity of our information systems and data, including as a result of a security breach, may result in significant losses that exceed our insurance coverage limits and have a material adverse effect on our financial position, results of operations and cash flows.
We expect to make significant investments to research and develop new tests, which may not be successful.
We are seeking to improve the performance of our existing testing solutions and to continue to expand our menu of products and services. For example, in August 2022, we acquired the GPS test from Exact Sciences and in September 2022, we developed and launched Resolve mdx which is a non-invasive urine test that identifies and quantifies infectious bacteria and their antibiotics susceptibility to help ensure patients receive the correct diagnosis and treatment as quickly as possible. In addition, we are currently developing an additional product, Monitor mdx, as a non-invasive test to risk stratifies patients for continued active surveillance versus intervention.
Developing new or improved diagnostic tests is a speculative and risky endeavor. Candidate products and services that may initially show promise may fail to achieve the desired results in larger clinical validation studies or may not achieve acceptable levels of clinical accuracy. Results from early studies or trials are not necessarily predictive of future clinical validation or clinical trial results, and interim results of a validation study or trial are not necessarily indicative of final results. From time to time, we may publicly disclose then-available data from clinical validation studies before completion, and the results and related findings and conclusions may be subject to change following the final analysis of the data related to the particular study. As a result, such data should be viewed with caution until the final data are available. Additionally, such data from clinical trials are subject to the risk that one or more of the clinical outcomes may materially change as patient enrollment and/or follow-up continues, and more patient data become available. Significant differences between initial or interim data and final data from either our clinical validation studies or clinical trials could significantly alter our plans to proceed with additional studies or trials, and harm our reputation and business prospects. If we determine that any of our current or future development programs is unlikely to succeed, we may abandon it without any return on our investment into the program. We may need to raise additional capital to bring any new products or services to market, which may not be available on acceptable terms, if at all.
Our research and development efforts will be hindered if we are not able to obtain samples, contract with third parties for access to samples or complete timely enrollment in future clinical trials.
Access to human sample types, such as blood, tissue, stool, or urine is necessary for our research and product development. Acquiring samples from individuals with clinical diagnoses or associated clinical outcomes through purchase or clinical studies is necessary. Lack of available samples can delay development timelines and increase costs of development. Generally, the agreements under which we gain access to human samples are non-exclusive. Other companies may compete with us for access. Additionally, the process of negotiating access to samples can be lengthy and it may involve numerous parties and approval levels to resolve complex issues such as usage rights, institutional review board approval and patient informed consent, privacy rights, publication rights, intellectual property ownership and research parameters. If we are not able to negotiate access to clinical samples with research institutions, hospitals, clinical partners, pharmaceutical companies, or companies developing therapeutics on a timely basis, or at all, or if other laboratories or our competitors secure access to these samples before us, our ability to research, develop and commercialize future products will be limited or delayed. Finally, we may not be able to conduct or complete clinical trials on a timely basis if we are not able to enroll sufficient numbers of patients in such trials, and our failure to do so could have an adverse effect on our research and development and product commercialization efforts.
Risks Related to Regulation of Our Business
Failure to comply with governmental payor regulations could result in us being excluded from participation in Medicare, Medicaid or other governmental payor programs, which would adversely affect our business.
Failure to comply with applicable Medicare, Medicaid and other governmental payor rules could result in our exclusion from participation in one or more governmental payor programs, a requirement to return funds already paid, civil monetary penalties, criminal penalties and/or limitations on the operational function of our laboratories. Additionally, with the recent implementation by CMS of a comprehensive oversight regime that consolidates program integrity powers into a single UPIC, audit and investigatory activity into potential billing fraud, waste and abuse in the industry has increased. These changes have adversely affected and may in the future adversely affect coverage and reimbursement for laboratory services, including the molecular diagnostics testing services we provide. If we were unable to receive reimbursement under a governmental payor program, this would have a severe impact on our revenues, given the importance of reimbursement under these programs in our revenue base.
14
Failure to comply with federal, state and foreign laboratory licensing and related requirements could cause us to lose the ability to perform our tests, experience disruptions to our business, or become subject to administrative or judicial sanctions.
We are subject to CLIA, a federal law that regulates clinical laboratories that perform testing on specimens derived from humans for the purpose of providing information for the diagnosis, prevention or treatment of disease. CLIA regulations establish specific standards with respect to personnel qualifications, facility administration, proficiency testing, quality control, quality assurance and inspections. Any testing subject to CLIA regulation must be performed in a CLIA certified laboratory. CLIA certification is also required in order for us to be eligible to bill state and federal healthcare programs, as well as commercial payors, for our tests. In addition, some states, including California and New York, require that we hold licenses or permits to test samples from patients in those states, even if our laboratory facilities are not located in those states, and as a result we are also required to maintain standards related to those states’ licensure requirements to conduct testing in our laboratories.
Failure to comply with applicable clinical laboratory licensure requirements may result in a range of enforcement actions, including suspension, limitation or revocation of our CLIA certification and/or state licenses, imposition of a directed plan of action, on-site monitoring, civil monetary penalties, criminal sanctions, inability to receive reimbursement from Medicare, Medicaid and commercial payors, as well as significant adverse publicity. Any sanction imposed under CLIA, its implementing regulations, or state or foreign laws or regulations governing clinical laboratory licensure or our failure to renew our CLIA certification, a state or foreign license or accreditation, could have a material adverse effect on our business, financial condition and results of operations. Even if we were able to bring our laboratory back into compliance, we could incur significant expenses and potentially lose revenue in doing so.
The FDA may change its position with respect to its regulation of the laboratory developed tests we offer or may seek to offer in the future, causing us to incur substantial costs and time delays associated with meeting requirements for pre-market clearance or approval or we could experience decreased demand for or reimbursement of our tests.
Our current tests are regulated as laboratory developed tests (“LDTs”) and we may seek to commercialize future products as LDTs. LDTs are clinical laboratory tests that are developed and validated by a laboratory for its own use. The FDA historically has taken the position that it has the authority to regulate such tests as medical devices under the FDCA but has for the most part exercised enforcement discretion and has not required clearance, de novo classification, or approval of LDTs prior to marketing.
At various times since 2006, the FDA has issued documents outlining its intent to require varying levels of FDA oversight of many LDTs, including our tests. In October 2023, the FDA announced a proposed rule seeking to regulate LDTs as medical devices under the FDCA. The comment period for the FDA’s proposed rule closed in December 2023 and the FDA has indicated its desire to issue a final rule on LDTs in April 2024 or shortly thereafter. Even if FDA does not finalize its proposed rule, the U.S. Congress may enact statutory changes that could alter or eliminate FDA’s current LDT enforcement policy. It is therefore unclear at this time what form that final regulation may take, or if FDA will finalize the regulation at all. It is also unclear what additional obligations might be placed on us as we continue to offer LDTs in light of the FDA’s renewed interest in greater oversight into this class of tests and the potential for legislative changes. Action by the FDA or Congress to phase out the FDA’s current policy of enforcement discretion over LDTs may materially impact our development and commercialization of our tests. If pre-market review is required for our current LDTs, the FDA could require that we stop selling our tests pending pre-market clearance or approval and our business could be negatively impacted in the U.S. until such review is completed and clearance or approval is obtained. The regulatory approval process may involve, among other things, successfully completing additional clinical studies and submitting a pre-market clearance notice or filing a pre-market approval application with the FDA. Such pre-market clinical testing could delay the commencement or completion of other clinical testing, significantly increase our test development costs, delay commercialization of any future LDTs, and interrupt sales of our current LDTs. Many of the factors that may cause or lead to a delay in the commencement or completion of clinical studies may also ultimately lead to delay or denial of regulatory clearance or approval. If pre-market review is required by the FDA, there can be no assurance that our LDTs will be cleared or approved on a timely basis, if at all, nor can there be assurance that the labeling claims cleared or approved by the FDA will be consistent with our current claims or adequate to support continued adoption of and reimbursement for our LDTs.
If our tests are allowed to remain on the market but there is uncertainty about the regulatory status of such tests, if they are labeled investigational by the FDA, or if labeling claims the FDA allows us to make are more limited than the claims we currently make, orders or reimbursement may decline.
Delays in receipt of, or failure to obtain, required FDA clearances or approvals for our products in development, or improvements to or expanded indications for our current offerings, could materially delay or prevent us from commercializing or otherwise adversely impact future product commercialization.
Unless otherwise exempted or subject to enforcement discretion, medical devices, which include diagnostic tests, must receive either FDA regulatory approval or clearance before being marketed in the U.S. Our tests and tests that we may develop may be deemed medical devices and require FDA clearance or approval. The FDA determines whether a medical device will require either regulatory approval or clearance based on statutory criteria that include the risk associated with the device and whether the device is similar to an existing, legally marketed product. The process to obtain either regulatory approval or clearance is typically costly, time-consuming, and uncertain. The regulatory approval process is generally more challenging than the clearance process. Even if we design a product that we expect to be eligible for the regulatory clearance process, the FDA may require that the product undergo the regulatory approval process. There can be no assurance that the FDA will ever permit us to market any new product that we develop. Even if regulatory approval or clearance is granted, such approval may include significant limitations on indicated uses, which could materially and adversely affect the prospects of any new medical device.
15
FDA regulatory approval or clearance is also required for certain enhancements we may make to future FDA-approved or FDA-cleared tests. FDA approval or clearance may also be required to make changes to the processes, equipment, reagents, and other consumables used in connection with a test. The FDA’s approval pathway can be time-consuming and costly and there can be no assurance that the FDA will ultimately approve any premarket approval submitted by us in a timely manner or at all.
In addition, the FDA’s ability to review and clear or approve new products or changes to existing products can be affected by a variety of factors, including government budget and funding levels, statutory, regulatory, and policy changes, the FDA’s ability to hire and retain key personnel and accept the payment of user fees, and other events that may otherwise affect the FDA’s ability to perform routine functions. Average review times at the agency have fluctuated in recent years as a result. In addition, prolonged government shutdowns or global health concerns may prevent or delay the FDA or other regulatory authorities from conducting, at all or in a timely manner, their regular inspections, reviews, or other regulatory activities (including pre-submission engagements). Any such delay in the ability of the FDA or other regulatory authorities to timely review and process our regulatory submissions could have a material adverse effect on our business.
Delays in receipt of, or failure to obtain, clearances or approvals could materially delay or prevent us from commercializing our products or result in substantial additional costs that could decrease our profitability. In addition, even if we receive FDA clearance or approval for a new or enhanced product, the FDA may condition, withdraw, or materially modify its clearance or approval.
We expect to rely on third parties to conduct any future studies of our technologies that may be required by the FDA or other U.S. or foreign regulatory bodies, and those third parties may not perform satisfactorily.
We expect to rely on third parties, such as contract research organizations, medical institutions and clinical investigators to conduct studies of our technologies that may be required by the FDA or other U.S. or foreign regulatory bodies. Our reliance on these third parties for clinical development activities will reduce our control over these activities. These third parties may not complete activities on schedule or conduct studies in accordance with regulatory requirements or our study design. Our reliance on these third parties will not relieve us of our requirement to prepare, and ensure our compliance with, various procedures required under good clinical practices, even though third-party contract research organizations may prepare and comply with their own, comparable procedures. If these third parties do not successfully carry out their contractual duties or regulatory obligations or meet expected deadlines, if the third parties need to be replaced or if the quality or accuracy of the data they obtain is compromised due to the failure to adhere to our clinical protocols or regulatory requirements or for other reasons, our studies may be extended, delayed, suspended or terminated, the study data may be invalidated, and we may not be able to obtain a required regulatory approval.
We conduct business in a heavily regulated industry, and changes in, or violations of, applicable regulations may, directly or indirectly, adversely affect our operational results and financial condition, which could harm our business.
Our business operations and activities may be subject to a range of local, state, federal, and international healthcare laws and regulations, including investigatory and program integrity audits and other oversight federal and state health care programs. These laws and regulations currently include, among others:
| ● | CLIA (which requires laboratories to obtain certification from the federal government) and state laboratory licensure laws; |
| ● | Federal Trade Commission standards regarding advertising and business practices; |
| ● | FDA laws and regulations; |
| ● | HIPAA (which imposes comprehensive federal standards with respect to the privacy and security of protected health information, and requirements for the use of certain standardized electronic transactions), and the amendments to HIPAA under HITECH (which strengthened and expanded HIPAA privacy and security compliance requirements, increased penalties for violators, extended enforcement authority to state attorneys general and imposed requirements for breach notification); |
| ● | state laws regulating genetic testing and the privacy protection of genetic test results, as well as state laws protecting the privacy and security of health information and personal data and mandating reporting of breaches to affected individuals and state regulators; |
| ● | the federal Anti-Kickback Statute (which prohibits knowingly and willfully offering, paying, soliciting, receiving, or providing remuneration, directly or indirectly, to induce either the referral of an individual, or the furnishing, arranging for, or recommending of an item or service that is reimbursable, in whole or in part, by a federal health care program) and parallel state anti-kickback laws (which contain similar prohibitions on remuneration between referral sources, although these state laws are not always limited in application to items or services reimbursable by federal or state health care programs); |
| ● | the federal False Claims Act (which imposes liability on any person or entity that, among other things, knowingly presents, or causes to be presented, a false or fraudulent claim for payment to the federal government or the improper retention of identified overpayments or other financial obligations to the federal government) and parallel state false claims acts (which contain similar prohibition on presenting false or fraudulent claims, although these state may extend to items or services by any third-party payor, including commercial insurers); |
| ● | the federal Civil Monetary Penalties Law, which prohibits, among other things, the offering or transferring of remuneration to a Medicare or state health care program (e.g., Medicaid) beneficiary if the person knows or should know it is likely to influence the beneficiary’s selection of a particular provider, practitioner, or supplier of services reimbursable by Medicare or a state health care program, unless an exception applies; |
16
| ● | the federal physician self-referral law, commonly known as the “Stark Law,” which prohibits a physician from making a referral to an entity for certain “designated health services” (“DHS”) payable by Medicare if the physician, or an immediate family member of the physician, has a financial relationship with that entity, unless an exception applies. The Stark Law further prohibits the entity from billing the Medicare program for DHS furnished pursuant to a prohibited referral. In addition, the Stark Law, through the addition of section 1903(s) to the Social Security Act, prohibits the federal government from making federal financial participation payments to state Medicaid programs for DHS furnished as a result of a referral that would violate the Stark Law if Medicare “covered the service to the same extent and under the same conditions” as the state Medicaid Program. The U.S. Department of Justice (“DOJ”) and several state agencies have successfully argued that Section 1903(s) expands the Stark Law to Medicaid-covered claims, even absent a separate state self-referral law prohibiting the same conduct; |
| ● | other federal and state fraud and abuse laws, including (i) the state anti-kickback laws described above, (ii) the state physician self-referral laws, and (iii) the state false claims acts described above; |
| ● | Section 216 of the Protecting Access to Medicare Act of 2014, which requires applicable laboratories to report commercial payor data in a timely and accurate manner beginning in 2017 and every three years thereafter (and in some cases annually); |
| ● | federal and state laws that impose reporting and other compliance-related requirements; and |
| ● | similar foreign laws and regulations that apply to us in the countries in which we operate. |
In addition, in October 2018, the Eliminating Kickbacks in Recovery Act of 2018 (“EKRA”), was enacted by the U.S. Congress as part of the Substance Use-Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities Act. EKRA is an all-payor anti-kickback law that makes it a criminal offense to pay any remuneration to induce referrals to, or in exchange for, patients using the services of a recovery home, a substance use clinical treatment facility, or laboratory. Although it appears that EKRA was intended to reach patient brokering and similar arrangements to induce patronage of substance use recovery and treatment, the language in EKRA is broadly written. Further, certain of EKRA’s exceptions, such as the exception applicable to relationships with employees that effectively prohibits incentive compensation, are inconsistent with the federal anti-kickback statute and regulations, which permit payment of employee incentive compensation, a practice that is common in the industry. Significantly, EKRA permits the DOJ to issue regulations clarifying EKRA’s exceptions or adding additional exceptions, but such regulations have not yet been issued. Laboratory industry stakeholders are reportedly seeking clarification regarding EKRA’s scope and/or amendments to its language.
Our business practices, in operating U.S. clinical laboratories, may face heightened scrutiny from U.S. government enforcement agencies such as the DOJ, the HHS Office of Inspector General (“OIG”), and CMS. The OIG has issued fraud alerts in recent years that identify certain arrangements between clinical laboratories and referring physicians as implicating the federal Anti-Kickback Statute. The OIG has stated that it is particularly concerned about these types of arrangements because the choice of laboratory, as well as the decision to order laboratory tests, typically are made or strongly influenced by the physician, with little or no input from the patient. Moreover, the provision of payments or other items of value by a clinical laboratory to a referring physician could be prohibited under the Stark Law, unless the arrangement meets all criteria of an applicable exception. The government has actively enforced these laws against clinical laboratories in recent years.
These U.S. laws and regulations are complex and are subject to interpretation by the U.S. courts and government agencies. Our failure to comply with such laws and regulations could lead to significant civil or criminal penalties, exclusion from participation in state and federal health care programs, individual imprisonment, disgorgement of profits, contractual damages, reputational harm, diminished profits and future earnings, additional reporting or oversight obligations if we become subject to a corporate integrity agreement or other agreement to resolve allegations of non-compliance with the law, curtailment or restructuring of our operations, or prohibitions or restrictions on our laboratories’ ability to provide or receive payment for our services, any of which could adversely affect our ability to operate our business and pursue our strategy. Even where we are able to successfully defend against any such claims, any potential audit, enforcement action, or litigation would involve substantial internal and external resources, detract from our executives’ day to day responsibilities, and result in legal expenditures, all of which could materially adversely affect our results of operations. While we believe that we are in material compliance with all applicable laws and regulations, there remains a risk that one or more government agencies could take a contrary position, or that a private party could file suit under the qui tam provisions of the federal False Claims Act or a similar state law. Such occurrences, regardless of their outcome, could damage our reputation and adversely affect important business relationships with third parties, including managed care organizations, and other private third-party payors.
Our business is subject to various complex laws and regulations applicable to providers of clinical diagnostic products and services.
As a provider of clinical diagnostic products and services, we and our partners are subject to extensive and frequently changing federal, state, local and foreign laws and regulations governing various aspects of our business. In particular, the clinical laboratory and healthcare industry is subject to significant governmental certification and licensing regulations, as well as federal, state and foreign laws regarding:
| ● | test ordering and billing practices; |
17
| ● | marketing, sales and pricing practices; |
| ● | health information privacy and security, including HIPAA and comparable state and foreign laws; |
| ● | insurance, including foreign public reimbursement; |
| ● | anti-markup legislation; and |
| ● | consumer protection |
We are also required to comply with FDA regulations, including with respect to our labeling and promotion activities. In addition, advertising of our tests is subject to regulation by the Federal Trade Commission, or FTC, and advertising of laboratory services is regulated by certain state laws. Violation of any FDA requirement could result in enforcement actions, such as seizures, injunctions, civil penalties and criminal prosecutions, and violation of any FTC or state law requirement could result in injunctions and other associated remedies, all of which could have a material adverse effect on our business. Most states also have similar regulatory and enforcement authority for medical devices. Additionally, most foreign countries have authorities comparable to the FDA and processes for obtaining marketing approvals. In particular, the entry into application of the European Union’s In Vitro Diagnostic Device Regulation will impose new requirements and create new compliance risks. Obtaining and maintaining these approvals, and complying with all laws and regulations, may subject us to similar risks and delays as those we could experience under FDA, FTC and state regulation. We incur various costs in complying and overseeing compliance with these laws and regulations. The growth of our business and sales organization, the acquisition of additional businesses or products and services and our expansion outside of the U.S. may increase the potential of violating these laws, regulations or our internal policies and procedures.
Healthcare policy has been a subject of extensive discussion in the executive and legislative branches of the federal and many state governments, and healthcare laws and regulations are subject to change. Development of the existing commercialization strategy for our tests and planned development of products in our pipeline has been based on existing healthcare policies. We cannot predict what additional changes, if any, will be proposed or adopted or the effect that such proposals or adoption may have on our business, financial condition and results of operations.
If we, or our partners, fail to comply with these laws and regulations, we could incur significant fines and penalties and our reputation and prospects could suffer. Additionally, any such partners could be forced to cease offering our products and services in certain jurisdictions, which could materially disrupt our business.
Failure to comply with privacy, security, and consumer protection laws and regulations could result in fines, penalties and damage to our reputation and have a material adverse effect on our business.
We are subject to a number of foreign, federal and state laws and regulations protecting the use, disclosure, and confidentiality of certain patient health and personal information, including patient records, and restricting the use and disclosure of that protected information, including state breach notification laws, HIPAA, as amended by HITECH, the European Union’s GDPR, and the California Consumer Privacy Act (“CCPA”), among others.
HIPAA extensively regulates the use and disclosure of individually identifiable health information, known as “protected health information,” and require covered entities, including health plans and most health care providers, to implement administrative, physical and technical safeguards to protect the security of such information. Covered entities must report breaches of unsecured protected health information to affected individuals without unreasonable delay and notification must also be made to the U.S. Department of Health & Human Services, Office for Civil Rights (the “OCR”) and, in certain situations involving large breaches, to the media. Various U.S. state laws and regulations may also require us to notify affected individuals and state agencies in the event of a data breach involving individually identifiable information.
Violations of the HIPAA privacy and security regulations may result in criminal and civil penalties. The OCR enforces the regulations and performs compliance audits. In addition to enforcement by OCR, state attorneys general are authorized to bring civil actions seeking either injunction or damages in response to violations that threaten the privacy of state residents. We follow and maintain a HIPAA compliance program, which we believe complies with the HIPAA privacy and security regulations, but there can be no assurance that OCR or other regulators will agree. The HIPAA privacy regulations and security regulations have and will continue to impose significant costs on us in order to comply with these standards.
We also remain subject to state privacy-related laws, such as the CCPA, that are more restrictive than the privacy regulations issued under HIPAA. These laws vary and could impose additional penalties.
We are also subject to laws and regulations in foreign countries covering data privacy and other protection of health and employee information that may be more onerous than corresponding U.S. laws, including in particular the laws of Europe.
For instance, the GDPR applies across the European Union and includes, among other things, a requirement for prompt notice of data breaches to data subjects and supervisory authorities in certain circumstances and significant fines for non-compliance. The GDPR also requires companies processing personal data of individuals residing in the European Union to comply with EU privacy and data protection rules, even if we do not have a physical presence in the European Union. Noncompliance could result in the imposition of fines, penalties, or orders to stop noncompliant activities.
18
These laws and regulations, in addition to similar laws and regulations being enacted by other states and counties, impose stringent cybersecurity standards and potentially significant non-compliance penalties, involve the expenditure of significant resources, the investment of significant resources and the investment of significant time and effort to comply. As these laws and regulations continue develop in the United States and internationally, we may be required to expend significant time and resources in order to update existing processes or implement additional mechanisms as necessary to ensure compliance with such cybersecurity laws.
Our employees, independent contractors, consultants, commercial partners, and vendors may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements.
We are exposed to the risk of fraud, misconduct, or other illegal activity by our employees, independent contractors, consultants, commercial partners, and vendors. Misconduct by these parties could include intentional, reckless and negligent conduct that fails to: comply with the rules and regulations of the CMS, FDA, and other federal and state government agencies as well as comparable foreign regulatory authorities; provide true, complete and accurate information to such regulatory authorities; comply with manufacturing and clinical laboratory standards; comply with healthcare fraud and abuse laws in the United States and similar foreign fraudulent misconduct laws; or report financial information or data accurately or to disclose unauthorized activities to us. In particular, research, sales, marketing, education, and other business arrangements in the healthcare industry are subject to extensive laws designed to prevent fraud, kickbacks, self-dealing, and other abusive practices, as well as off-label product promotion. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, educating, marketing and promotion, sales and commission, certain customer incentive programs, and other business arrangements generally. Activities subject to these laws also involve the improper use of information obtained in the course of participant recruitment for clinical studies, which could result in regulatory sanctions and cause serious harm to our reputation. We have adopted a code of business conduct and ethics and provide compliance training to our workforce members upon onboarding and annually thereafter, but it is not always possible to identify and deter misconduct by employees and third parties, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to be in compliance with such laws. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business, including the imposition of significant fines or other sanctions. Even if it is later determined after an action is instituted against us that we were not in violation of these laws, we may be faced with negative publicity, incur significant expenses defending our actions, and have to divert significant management resources from other matters.
Our operating results could be materially adversely affected by unanticipated changes in tax laws and regulations, adjustments to our tax provisions, exposure to additional tax liabilities, or forfeiture of our tax assets.
We are subject to laws and regulations on tax levies and other charges or contributions in different countries, including transfer pricing and tax regulations for the compensation of personnel and third parties. Our tax structure involves several transfers and transfer price determinations between our parent company and our subsidiaries or other affiliates. Our effective tax rates could be adversely affected by changes in tax laws, treaties and regulations, both internationally and domestically. An increase of the effective tax rates could have an adverse effect on our business, financial position, results of operations and cash flows.
The net operating loss carry forwards of our corporate subsidiaries may be unavailable to offset future taxable income because of restrictions under U.S. tax law. As of December 31, 2023, consolidated net tax loss carry forwards amounted to $308.7 million. Our NOLs generated in tax years ending on or prior to December 31, 2017 are only permitted to be carried forward for 20 taxable years under applicable U.S. federal tax law, and therefore could expire unused. We consider that it is highly likely that we will be unable to use at least a portion of these NOLs, in light of our continued losses. Under tax legislation commonly referred to as the Tax Cuts and Jobs Act (“TCJA”), as modified by the CARES Act, our federal NOLs generated in tax years ending after December 31, 2017 may be carried forward indefinitely and NOLs arising in taxable years beginning after December 31, 2017 and before January 1, 2021 may be carried back to each of the five taxable years preceding the tax year of such loss, but NOLs arising in taxable years beginning after December 31, 2020 may not be carried back. In addition, under the TCJA, as modified by the CARES Act, for taxable years beginning after December 31, 2020, the deductibility of federal NOLs generated in taxable years beginning after December 31, 2017 is limited to 80% of current year taxable income. It is uncertain if and to what extent various states will conform to the TCJA, as modified by the CARES Act.
In addition, under sections 382 and 383 of the Internal Revenue Code of 1986, as amended, or the Code, if a corporation undergoes an “ownership change” (generally defined as a cumulative change in ownership by “5-percent shareholders” that exceeds 50 percentage points over a rolling three-year period), the corporation’s ability to use its pre-change NOLs and certain other pre-change tax attributes to offset post-change income and taxes may be limited. Similar rules may apply under state tax laws. Our existing NOLs and other certain tax attributes may be subject to limitations arising from previous ownership changes. In addition, future changes in our stock ownership, many of which are outside of our control, could result in an ownership change under Sections 382 and 383 of the Code. We have not conducted any studies to determine annual limitations, if any, that could result from such changes in the ownership. Our ability to utilize those NOLs and certain other tax attributes could be limited by an “ownership change” as described above and consequently, we may not be able to utilize a material portion of our NOLs and certain other tax attributes, which could have a material adverse effect on our cash flows and results of operations by effectively increasing our future tax obligations.
Also under Belgian tax law, certain restrictions regarding the use of Belgian tax losses carried forward apply and these losses may also be forfeited upon certain changes of control over Belgian corporate taxpayers.
19
Given that we have historically generated operating losses, any change in our ability to use NOLs could have a severe impact on us if and when we become profitable. As of December 31, 2023, we had an accumulated deficit of $331.4 million and for the year ended December 31, 2023, we had a net loss of $43.1 million.
Risks Related to Ownership of Our Ordinary Shares
The trading price of our ordinary shares may be volatile due to factors beyond our control, and purchasers of our ordinary shares could incur substantial losses.
The market price of our ordinary shares may be volatile. The stock market in general and the market for biotechnology companies in particular have experienced extreme volatility that has often been unrelated to the operating performance of particular companies. As a result of this volatility, investors may not be able to sell their shares at or above the price originally paid for the security. The market price for our ordinary shares may be influenced by many factors, including:
| ● | actual or anticipated fluctuations in our financial condition and operating results; |
| ● | the release of new data from our clinical trials; |
| ● | actual or anticipated changes in our growth rate relative to our competitors; |
| ● | competition from existing products or new products that may emerge; |
| ● | announcements by us or our competitors of significant acquisitions, strategic partnerships, joint ventures, collaborations or capital commitments; |
| ● | failure to meet or exceed financial estimates and projections of the investment community or that we provide to the public; |
| ● | issuance of new or updated research or reports by securities analysts; |
| ● | fluctuations in the valuation of companies perceived by investors to be comparable to us; |
| ● | currency fluctuations; |
| ● | additions or departures of key management or scientific personnel; |
| ● | disputes or other developments related to proprietary rights, including patents, litigation matters and our ability to obtain patent protection for our technologies; |
| ● | changes to coverage policies or reimbursement levels by commercial third-party payors and government payors and any announcements relating to coverage policies or reimbursement levels; |
| ● | announcement or expectation of additional debt or equity financing efforts; |
| ● | issuances or sales of ordinary shares by us, our insiders or our other holders; and |
| ● | general economic and market conditions. |
These and other market and industry factors may cause the market price and demand for our ordinary shares to fluctuate substantially, regardless of our actual operating performance, which may limit or prevent investors from readily selling shares and may otherwise negatively affect the liquidity of the trading market for our ordinary shares.
Certain of our significant shareholders may have different interests from us and may be able to control us, including the outcome of shareholder votes.
As of March 31, 2024, (i) MVM Partners LLP beneficially owned approximately 16.7% of our ordinary shares and has one representative at the board level (Dr. Eric Bednarski), (ii) Bleichroeder LP owned approximately 14.2% of our ordinary shares, and (iii) Valiance Asset Management beneficially owned approximately 7.7% of our ordinary shares. In addition, as long as two of MVM Partners LLP’s funds (MVM V LP and MVM GP (No.5) LP) hold in aggregate 5% of our company’s outstanding shares, they are entitled to have one observer at the board level (see Item 7B. “Related party transactions — MVM Subscription Agreement”). As a result, these shareholders will be able to exercise a significant level of control over all matters requiring stockholder approval, including the election of directors, amendment of our Articles of Association and approval of certain significant corporate transactions. This control could have the effect of delaying or preventing a change of control of the Company or changes in management, in each case, which other shareholders might find favorable, and will make the approval of certain transactions difficult or impossible without the support of these significant shareholders.
20
If securities or industry analysts do not publish research or publish inaccurate or unfavorable research about our business, the price of our ordinary shares and their trading volume could decline.
The trading market for our ordinary shares depends in part on the research and reports that securities or industry analysts publish about us or our business. If no or only limited securities or industry analysts cover our company, the trading price for our ordinary shares could be negatively impacted. If one or more of the analysts who cover us downgrades our equity securities or publishes inaccurate or unfavorable research about our business, the price of our ordinary shares would likely decline. If one or more of these analysts ceases coverage of our company or fails to publish reports on us regularly, or downgrades our securities, demand for our ordinary shares could decrease, which could cause the price of our ordinary shares or their trading volume to decline.
We intend to retain all available funds and any future earnings and, consequently, the ability of holders of our ordinary shares to achieve a return on their investment will depend on appreciation in the price of our ordinary shares.
We have never declared or paid any cash dividends on our ordinary shares, and we intend to retain all available funds and any future earnings to fund the development and expansion of our business. In addition, our loan agreement with Innovatus limits our ability to pay any such dividends. Therefore, holders of our ordinary shares are not likely to receive any dividends for the foreseeable future and the success of an investment in our ordinary shares will depend upon any future appreciation in their value. Consequently, investors may need to sell all or part of their holdings of our ordinary shares after price appreciation, which may never occur, as the only way to realize any future gains on their investment. There is no guarantee that our ordinary shares will appreciate in value or even maintain the price at which our investors have purchased them. Investors seeking cash dividends should not purchase our ordinary shares.
In addition, if we choose to pay dividends in the future, exchange rate fluctuations may affect the amount of Euros that we are able to distribute, and the amount in U.S. dollars that our shareholders receive upon the payment of cash dividends or other distributions we declare and pay in euros, if any. Any dividends will generally be subject to Belgian withholding tax. These factors could harm the value of our ordinary shares.
Holders of our ordinary shares should be aware that the rights provided to holders of our ordinary shares under Belgian corporate law and our Articles of Association differ in certain respects from the rights that you would typically enjoy as a shareholder of a U.S. company under applicable U.S. federal and state laws.
We are a Belgian company with limited liability. Our corporate affairs are governed by our Articles of Association and by the laws governing companies incorporated in Belgium. The rights of shareholders and the responsibilities of members of our Board of Directors may be different from the rights and obligations of shareholders and boards of directors in companies governed by the laws of U.S. jurisdictions. In the performance of its duties, our Board is required by Belgian law to consider the interests of our company, its shareholders, its employees, and other stakeholders. It is possible that some of these parties will have interests that are different from, or in addition to, the interests of our shareholders. See Item 10B. “Memorandum and Articles of Association.”
Concentration of ownership of our ordinary shares among our existing executive officers, directors and principal shareholders may prevent holders of our ordinary shares from influencing significant corporate decisions.
Our executive officers, directors, greater than three percent shareholders and their affiliates beneficially owned approximately 46.6% of our outstanding ordinary shares as of March 31, 2024. Depending on the level of attendance at our general meetings of shareholders, these shareholders, either alone or voting together as a group, will be in a position to determine the outcome of decisions taken at any such general meeting. Any shareholder or group of shareholders controlling more than 50% of the share capital present and voting at our general meetings of shareholders may control any shareholder resolution requiring a simple majority, including the appointment of Board members, as well as certain decisions relating to our capital structure, the approval of certain significant corporate transactions and amendments to our Articles of Association. Among other consequences, this concentration of ownership may prevent or discourage unsolicited acquisition proposals that shareholders may believe are in the best interest of the Company. Some of these persons or entities may have interests different than those of our shareholders.
Future sales, or the perception of future sales, of a substantial number of our ordinary shares could adversely affect the price of our ordinary shares, and actual sales of our equity will dilute current holders of our ordinary shares.
Future sales of a substantial number of our ordinary shares, or the perception that such sales will occur, could cause a decline in the market price of our ordinary shares. Approximately 12.7 million ordinary shares are held by our directors, executive officers and greater than five percent shareholders. If one or more of these securityholders sell substantial amounts of ordinary shares in the public market, or the market perceives that such sales may occur, the market price of our ordinary shares and our ability to raise capital through an issue of equity securities in the future could be adversely affected.
If we issue ordinary shares in future financings, shareholders may experience dilution and, as a result, the price of our ordinary shares may decline.
We may from time-to-time issue additional ordinary shares at a discount from the trading price of our ordinary shares. As a result, holders of our ordinary shares would experience immediate dilution upon the issuance of any of our ordinary shares at such discount. In addition, as opportunities present themselves, we may enter into financing or similar arrangements in the future, including the issuance of debt securities, preference shares or shares. If we issue ordinary shares or other equity or equity-linked securities, holders of our ordinary shares would experience additional dilution and, as a result, the price of our ordinary shares may decline.
21
It may be difficult for holders of our ordinary shares outside Belgium to serve process on, or enforce foreign judgments against, us or our directors and senior management.
We are a Belgian limited liability company. Less than a majority of the members of our Board of Directors are residents of the United States. All or a substantial portion of the assets of such non-resident persons and a significant portion of our assets are located outside the United States. As a result, it may not be possible for holders of our ordinary shares to effect service of process upon such persons or on us or to enforce against them or us a judgment obtained in U.S. courts. Original actions or actions for the enforcement of judgments of U.S. courts relating to the civil liability provisions of the federal or state securities laws of the United States are not directly enforceable in Belgium.
The United States and Belgium do not currently have a multilateral or bilateral treaty providing for reciprocal recognition and enforcement of judgments, other than arbitral awards, in civil and commercial matters. In order for a final judgment for the payment of money rendered by U.S. courts based on civil liability to produce any effect on Belgian soil, it is accordingly required that this judgment be recognized or be declared enforceable by a Belgian court in accordance with Articles 22 to 25 of the 2004 Belgian Code of Private International Law. Recognition or enforcement does not imply a review of the merits of the case and is irrespective of any reciprocity requirement. A U.S. judgment will, however, not be recognized or declared enforceable in Belgium, unless (in addition to compliance with certain technical provisions) the Belgian courts are satisfied of the following:
| ● | the effect of the enforcement judgment is not manifestly incompatible with Belgian public policy; |
| ● | the judgment did not violate the rights of the defendant; |
| ● | the judgment was not rendered in a matter where the parties transferred rights subject to transfer restrictions with the sole purpose of avoiding the application of the law applicable according to Belgian international private law; |
| ● | the judgment is not subject to further recourse under U.S. law; |
| ● | the judgment is not incompatible with a judgment rendered in Belgium or with a subsequent judgment rendered abroad that might be recognized in Belgium; |
| ● | the claim was not filed outside Belgium after the same claim was filed in Belgium, while the claim filed in Belgium is still pending; |
| ● | the Belgian courts did not have exclusive jurisdiction to rule on the matter; |
| ● | the U.S. court did not accept its jurisdiction solely on the basis of the presence of the plaintiff or the location of goods not direct linked to the dispute in the United States; |
| ● | the judgment did not concern the deposit or validity of intellectual property rights when the deposit or registration of those intellectual property rights was requested, done or should have been done in Belgium pursuant to international treaties; |
| ● | the judgment did not relate to the validity, operation, dissolution, or liquidation of a legal entity that has its main seat in Belgium at the time of the petition of the U.S. court; |
| ● | if the judgment relates to the opening, progress or closure of insolvency proceedings, it is rendered on the basis of the European Insolvency Regulation (EC Regulation No. 1346/2000 of May 29, 2000) or, if not, that (a) a decision in the principal proceedings is taken by a judge in the state where the most important establishment of the debtor was located or (b) a decision in territorial proceedings was taken by a judge in the state where the debtor had another establishment than its most important establishment; and |
| ● | the judgment submitted to the Belgian court is authentic under the laws of the state where the judgment was issued; in case of a default judgment, it can be shown that under locally applicable laws the invitation to appear in court was properly served on the defendant; a document can be produced showing that the judgment is, under the rules of the state where it was issued, enforceable and was properly served on the defendant. |
In addition to recognition or enforcement, a judgment by a federal or state court in the United States against us may also serve as evidence in a similar action in a Belgian court if it meets the conditions required for the authenticity of judgments according to the law of the state where it was rendered. The findings of a federal or state court in the United States will not, however, be taken into account to the extent they appear incompatible with Belgian public policy.
Based on the lack of a treaty as described above, U.S. investors may not be able to enforce against us or members of our Board of Directors or our executive management any judgments obtained in U.S. courts in civil and commercial matters, including judgments under the U.S. federal securities laws.
22
We are an “emerging growth company” and as a result of the reduced disclosure and governance requirements applicable to emerging growth companies, our ordinary shares may be less attractive to investors.
We are an “emerging growth company” as defined in the Jumpstart Our Business Startups Act of 2012 (the “JOBS Act”). For as long as we continue to be an emerging growth company, we may take advantage of exemptions from various reporting requirements that are applicable to other public companies that are not emerging growth companies, including not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act of 2002 (the “Sarbanes-Oxley Act”), exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder approval of any golden parachute payments not previously approved. We may take advantage of these exemptions until we are no longer an emerging growth company. We could be an emerging growth company for up to five years, although circumstances could cause us to lose that status earlier, including if the aggregate market value of our ordinary shares held by non-affiliates exceeds $700 million as of the end of our second fiscal quarter before that time, in which case we would no longer be an emerging growth company as of the following December 31st (the last day of our fiscal year). We cannot predict if investors will find our ordinary shares less attractive because we may rely on these exemptions. If some investors find our ordinary shares less attractive as a result, there may be a less active trading market for our ordinary shares and the price of our ordinary shares may be more volatile.
As a foreign private issuer and as permitted by the listing requirements of Nasdaq, we rely on certain home country corporate governance practices rather than the corporate governance requirements of Nasdaq.
We qualify as a foreign private issuer and our ordinary shares are listed on Nasdaq. In accordance with the listing requirements of Nasdaq, we rely on home country governance requirements and certain exemptions thereunder rather than relying on the corporate governance requirements of Nasdaq. For example, we are exempt from certain rules under the Exchange Act that regulate disclosure obligations and procedural requirements related to the solicitation of proxies, consents or authorizations applicable to a security registered under the Exchange Act, including the U.S. proxy rules under Section 14 of the Exchange Act. In addition, our officers and directors are exempt from the reporting and “short-swing” profit recovery provisions of Section 16 of the Exchange Act and related rules with respect to their purchases and sales of our securities. Moreover, while we currently publish annual and semi-annual reports on our website and file such financial reports with the SEC, we are not required to file periodic reports with the SEC as frequently or as promptly as U.S. public companies. Specifically, we are not required to file quarterly reports on Form 10-Q or current reports on Form 8-K that a domestic company would be required to file under the Exchange Act. Accordingly, there may be less publicly available information concerning our company than there would be if we were not a foreign private issuer.
In addition, the Listing Rules of the Nasdaq Stock Market require a majority of the directors of a listed U.S. company to be independent, whereas under the applicable rules in Belgium, there is no requirement to appoint independent directors. The Listing Rules of the Nasdaq Stock Market further require that each of the nominating, compensation and audit committees of a listed U.S. company be comprised entirely of independent directors. At present, our Audit Committee is composed entirely of independent directors, whereas our Nomination and Remuneration Committee is composed of three independent directors out of four members. However, the Belgian Companies and Associations Code does not require that we have a nominating, compensation or audit committee and does not have independence requirements for such committees. Therefore, our shareholders may be afforded less protection than they otherwise would have under corporate governance listing standards applicable to U.S. domestic issuers.
We may lose our foreign private issuer status in the future, which could result in significant additional costs and expenses.
As a foreign private issuer, we are not required to comply with all the periodic disclosure and current reporting requirements of the Exchange Act and related rules and regulations. The determination of foreign private issuer status is made annually on the last business day of our most recently completed second fiscal quarter. Accordingly, we will next make a determination with respect to our foreign private issuer status on June 30, 2024. There is a risk that we will lose our foreign private issuer status in the future.
We would lose our foreign private issuer status if, for instance more than 50% of our ordinary shares are owned by U.S. residents or persons and more than 50% of our assets are located in the United States and we continue to fail to meet additional requirements necessary to maintain our foreign private issuer status. The regulatory and compliance costs to us under U.S. securities laws as a U.S. domestic issuer may be significantly greater than the costs we incur as a foreign private issuer. If we are not a foreign private issuer, we will be required to file periodic reports and registration statements on U.S. domestic issuer forms with the SEC, which are more detailed and extensive in certain respects than the forms available to a foreign private issuer. We would be required under current SEC rules to prepare our financial statements in accordance with U.S. GAAP and modify certain of our policies to comply with corporate governance practices associated with U.S. domestic issuers. Such conversion and modifications would involve additional costs. In addition, we may lose our ability to rely upon exemptions from certain corporate governance requirements on U.S. stock exchanges that are available to foreign private issuers, which could also increase our costs.
U.S. holders of our ordinary shares may suffer adverse tax consequences if we are characterized as a passive foreign investment company, or PFIC.
In general, a non-U.S. corporation is a PFIC for U.S. federal income tax purposes for any taxable year in which (i) 50% or more of value of its assets (based on an average of the quarterly values of the assets during such taxable year) consists of assets that produce, or are held for the production of, passive income, or (ii) 75% or more of its gross income consists of passive income. A separate determination must be made after the close of each fiscal year as to whether a non-U.S. corporation is a PFIC for that year. For purposes of the above calculations, a non-U.S. corporation that owns, directly or indirectly, at least 25% by value of the shares of another corporation is treated as if it held its proportionate share of the assets of the other corporation and received directly its proportionate share of the income of the other corporation. Passive income generally includes dividends, interest, investment gains and certain rents and royalties. Cash is generally a passive asset for these purposes. The value goodwill is generally treated as an active asset if it is associated with business activities that produce active income.
23
If we are a PFIC for any taxable year during which a U.S. Holder (as defined under Item 10E. “Taxation”) holds our ordinary shares, we will continue to be treated as a PFIC with respect to such U.S. Holder in all succeeding years during which the U.S. Holder owns our ordinary shares regardless of whether we continue to meet the PFIC test described above, unless the U.S. Holder makes a specified election once we cease to be a PFIC. If we are classified as a PFIC for any taxable year during which a U.S. Holder holds our ordinary shares, the U.S. Holder may be subject to adverse tax consequences regardless of whether we continue to qualify as a PFIC, including ineligibility for any preferred tax rates on capital gains or on actual or deemed dividends, interest charges on certain taxes treated as deferred, and additional reporting requirements.
Based on the current estimates, and expected future composition, of our income and the value of our assets, including goodwill, we do not expect to be a PFIC for our current taxable year. However, our PFIC status for any taxable year is an annual determination that can be made only after the end of that year and will depend on the composition of our income and assets and the value of our assets from time to time. The determination of whether we are a PFIC is fact-intensive and the applicable law is subject to varying interpretation. There can be no assurance that the U.S. Internal Revenue Service, or IRS, will agree with our position or that the IRS will not successfully challenge our position including our classification of certain income and assets as non-passive or our valuation of our tangible and intangible assets.
A U.S. Holder may in certain circumstances mitigate the adverse tax consequences of the PFIC rules by filing an election to treat the PFIC as a Qualified Electing Fund (“QEF”) or, if shares of the PFIC are “marketable stock” for purposes of the PFIC rules, by making a mark-to-market election with respect to the shares of the PFIC. However, we do not currently intend to provide the information necessary for U.S. Holders to make a QEF election if we were treated as a PFIC for any taxable year and prospective investors should assume that a QEF election will not be available. Furthermore, if a U.S. Holder were to make a mark-to-market election with respect to our ordinary shares, the U.S. Holder would be required to include annually in its U.S. federal taxable income (taxable at ordinary income rates) an amount reflecting any year end increase in the value of its ordinary shares. For further discussion of the PFIC rules and the adverse U.S. federal income tax consequences in the event we are classified as a PFIC, see Item 10E. “Taxation.”
The U.S. federal income tax rules relating to PFICs are very complex. Current and prospective U.S. Holders are strongly urged to consult their own tax advisors with respect to the impact of PFIC status on the purchase, ownership and disposition of our ordinary shares, the consequences to them of an investment in a PFIC, any elections available with respect to our ordinary shares and the IRS information reporting obligations with respect to the purchase, ownership and disposition of our ordinary shares of a PFIC.
If a U.S. Holder is treated as owning at least 10% of our ordinary share capital, such holder may be subject to adverse U.S. federal income tax consequences.
If a U.S. Holder (as defined below under Item 10E. “Taxation”) is treated as owning, directly, indirectly or constructively, at least 10% of the value or voting power of our share capital, such U.S. Holder may be treated as a “U.S. shareholder” with respect to each “controlled foreign corporation” in our group, if any. Because our group currently includes at least one U.S. subsidiary, under current law, any of our current non-U.S. subsidiaries and any future newly formed or acquired non-U.S. subsidiaries will be treated as controlled foreign corporations, regardless of whether we are treated as a controlled foreign corporation. A U.S. shareholder of a controlled foreign corporation may be required to annually report and include in its U.S. taxable income its pro rata share of “Subpart F income,” “global intangible low-taxed income” and investments in U.S. property by controlled foreign corporations, regardless of whether we make any distributions. An individual that is a U.S. shareholder with respect to a controlled foreign corporation generally would not be allowed certain tax deductions or foreign tax credits that would be allowed to a U.S. shareholder that is a U.S. corporation. Failure to comply with controlled foreign corporation reporting obligations may subject a U.S. shareholder to significant monetary penalties. We cannot provide any assurances that we will furnish to any U.S. shareholder information that may be necessary to comply with the reporting and tax paying obligations applicable under the controlled foreign corporation rules of the Code. U.S. Holders should consult their tax advisors regarding the potential application of these rules to their investment in our ordinary shares. See Item 10E. “Taxation” for a more detailed discussion.
We incur significant costs as a result of operating as a company that is publicly listed on Nasdaq, and our management is required to devote substantial time to compliance initiatives.
As a company that is publicly listed on Nasdaq, we are subject to the reporting requirements of the Exchange Act, the Sarbanes-Oxley Act, the Dodd-Frank Wall Street Reform and Consumer Protection Act, the Nasdaq listing requirements and other applicable securities rules and regulations. Compliance with these rules and regulations is costly, and will become even more costly after we are no longer an “emerging growth company” and/or a foreign private issuer. Further, as Belgian limited liability company listed in the U.S., we are subject to potentially overlapping and disparate multi-jurisdictional laws and rules that potentially impact the disclosure of information. From time to time, this may result in uncertainty regarding compliance matters and result in higher costs necessitated by legal analysis of dual legal regimes, ongoing revisions to disclosure and adherence to heightened governance practices. Moreover, these rules and regulations increase our legal and financial compliance costs and make some activities more time-consuming and costly. For example, we expect that these rules and regulations may make it more difficult and more expensive for us to obtain director and officer liability insurance, which in turn could make it more difficult for us to attract and retain qualified senior management personnel or members for our Board of Directors. These rules and regulations are often subject to varying interpretations, in many cases due to their lack of specificity, and, as a result, their application in practice may evolve over time as new guidance is provided by regulatory and governing bodies. This could result in continuing uncertainty regarding compliance matters and higher costs necessitated by ongoing revisions to disclosure and governance practices.
24
As a result of being a U.S. public company, we are subject to regulatory compliance requirements, including Section 404 of the Sarbanes-Oxley Act (“Section 404”), and if we fail to maintain an effective system of internal controls, we may not be able to accurately report our financial results or prevent fraud.
Pursuant to Section 404, our management is required to assess and attest to the effectiveness of our internal control over financial reporting in connection with issuing our consolidated financial statements as of and for each fiscal year. Section 404 also requires an attestation report on the effectiveness of internal control over financial reporting be provided by our independent registered public accounting firm beginning with our annual report following the date on which we are no longer an “emerging growth company.”
The cost of complying with Section 404 significantly increases and management’s attention may be diverted from other business concerns, which could adversely affect our results. We may need to hire more employees in the future or engage outside consultants to comply with these requirements, which will further increase expenses. If we fail to comply with the requirements of Section 404 in the required timeframe, we may be subject to sanctions or investigations by regulatory authorities, including the SEC and Nasdaq. Furthermore, if we are unable to attest to the effectiveness of our internal control over financial reporting, we could lose investor confidence in the accuracy and completeness of our financial reports, and the market price of our ordinary shares could decline. Failure to implement or maintain effective internal control over financial reporting could also restrict our future access to the capital markets and subject each of us, our directors and our officers to both significant monetary and criminal liability. In addition, changing laws, regulations and standards relating to corporate governance and public disclosure are creating uncertainty for public companies, increasing legal and financial compliance costs and making some activities more time consuming. These laws, regulations and standards are subject to varying interpretations, in many cases due to their lack of specificity, and, as a result, their application in practice may evolve over time as new guidance is provided by regulatory and governing bodies. This could result in continuing uncertainty regarding compliance matters and higher costs necessitated by ongoing revisions to disclosure and governance practices. We intend to invest resources to comply with evolving laws, regulations and standards, and this investment may result in increased general and administrative expense and a diversion of management’s time and attention from revenue generating activities to compliance activities. If our efforts to comply with new laws, regulations and standards differ from the activities intended by regulatory or governing bodies due to ambiguities related to their application and practice, regulatory authorities may initiate legal proceedings against us and our business, financial position, results and prospects may be adversely affected.
If we fail to implement and maintain effective internal controls over financial reporting, our ability to produce accurate financial statements on a timely basis could be impaired.
We are subject to reporting obligations under U.S. securities laws and the Sarbanes-Oxley Act of 2002. Section 404 of the Sarbanes-Oxley Act requires that we include a report from management on the effectiveness of our internal control over financial reporting in our annual report on Form 20-F for the year ended December 31, 2023. If we fail to implement and maintain adequate disclosure controls and procedures, our management may conclude that our internal control over financial reporting is not effective. This conclusion could adversely impact the market price of our ordinary shares due to a loss of investor confidence in the reliability of our reporting processes.
During 2023, management identified a material weakness related to deficiencies related to the design of our controls over logical access and segregation of duties, at the application control level, in certain IT environments that are relevant to the preparation of our financial statements. Specifically, (i) periodic user access reviews of roles and permissions were not performed sufficiently throughout the period for certain key IT systems, and (ii) certain key IT systems were not logically restricted, resulting in improper segregation of duties for certain business processes. These IT deficiencies did not result in a misstatement to the financial statements, however, the deficiencies, when aggregated, could impact our ability to maintain effective segregation of duties, as well as the effectiveness of IT-dependent controls (such as automated controls that address the risk of material misstatement to one or more assertions, along with the IT controls and underlying data that support the effectiveness of system-generated data and reports) that could result in misstatements potentially impacting all financial statement accounts and disclosures that would result in a material misstatement to the annual or interim financial statements that would not be prevented or detected. Management took steps to remediate the material weakness, including redesigning the logical access and placing enhanced segregation of duties, enhancing its internal documentation and monitoring approach to ensure that all procedures designed to restrict access to applications and data, are operating in an optimal manner in order to provide management with comfort that access is properly limited to the appropriate internal personnel. Although remediation of the identified IT deficiencies is now complete (as of the date of this annual report), our management concluded that, as of December 31, 2023, our internal control over financial reporting was not effective due to the material weakness described above. Although we were able to remediate these issues, these efforts may not be sufficient to avoid similar material weaknesses in the future.
Undetected material weaknesses in our internal controls could lead to financial statement restatements and require us to incur remediation costs. To the extent we experience additional future material weaknesses, investors could lose confidence in the accuracy or completeness of our reported financial information, which could have a negative effect on the trading price of our ordinary shares. Failure to implement or maintain effective internal control over financial reporting could also restrict our future access to the capital markets and subject each of us, our directors and our officers to both significant monetary and criminal liability.
25
We are required to perform system and process evaluations and testing of our internal controls over financial reporting, to allow our management and our independent public registered accounting firm to report on the effectiveness of our internal control over financial reporting. In addition, our compliance with Section 404 of the Sarbanes-Oxley Act will require that we incur substantial accounting expense, expend significant management effort and we may need to hire additional accounting and financial staff with the appropriate experience and technical accounting knowledge, and compile the system and process documentation necessary to perform the evaluation needed to comply with Section 404 of the Sarbanes-Oxley Act. We may not be able to complete our evaluation, testing and any required remediation in a timely fashion. Any failure to implement required new or improved controls, or difficulties encountered in their implementation could cause us to fail to meet our reporting obligations. In addition, any testing by us conducted in connection with Section 404 of the Sarbanes-Oxley Act, or any subsequent testing by our independent registered public accounting firm, may reveal additional deficiencies in our internal controls over financial reporting that are deemed to be material weaknesses or that may require prospective or retroactive changes to our financial statements or identify other areas for further attention or improvement. We cannot assure you that there will not be additional material weaknesses or significant deficiencies in our internal control over financial reporting in the future.
We may be subject to securities litigation, which is expensive and could divert management’s attention.
The market price of our ordinary shares may be volatile and, in the past, companies that have experienced volatility in the market price of their stock have been subject to securities class action litigation. We may be the target of this type of litigation in the future. Securities litigation against us could result in substantial costs and divert our management’s attention from other business concerns, which could seriously harm our business.
Investors residing in countries other than Belgium may suffer dilution if they are unable to participate in future preferential subscription rights offerings.
Under Belgian law and our constitutional documents, shareholders have a waivable and cancellable preferential subscription right to subscribe pro rata to their existing shareholdings to the issuance, against a contribution in cash, of new shares or other securities entitling the holder thereof to new shares, unless such rights are limited or cancelled by resolution of our general shareholders’ meeting or, if so authorized by a resolution of such meeting, our Board of Directors. The exercise of preferential subscription rights by certain shareholders not residing in Belgium (including those in the United States, Australia, Israel, Canada or Japan as a result of the offering and taking into account the current shareholding and international network of our current Board of Directors) may be restricted by applicable law, practice or other considerations, and such shareholders may not be entitled to exercise such rights, unless the rights and shares are registered or qualified for sale under the relevant legislation or regulatory framework. In particular, we may not be able to establish an exemption from registration in the United States under the Securities Act of 1933, as amended (the “Securities Act”), and we are under no obligation to file a registration statement with respect to any such preferential subscription rights or underlying securities or to endeavor to have a registration statement declared effective under the Securities Act. Shareholders in jurisdictions outside Belgium who are not able or not permitted to exercise their preferential subscription rights in the event of a future preferential subscription rights, equity or other offering may suffer dilution of their shareholdings.
Takeover provisions in the national law of Belgium may make a takeover difficult.
Public takeover bids on our shares and other voting securities, such as warrants or convertible bonds, if any, are subject to the Belgian Act of April 1, 2007 on public takeover bids, as amended and implemented by the Belgian Royal Decree of April 27, 2007, or Royal Decree, and to the supervision by the Belgian Financial Services and Markets Authority, or FSMA. Public takeover bids must be made for all of our voting securities, as well as for all other securities that entitle the holders thereof to the subscription to, the acquisition of or the conversion into voting securities. Prior to making a bid, a bidder must issue and disseminate a prospectus, which must be approved by the FSMA. The bidder must also obtain approval of the relevant competition authorities, where such approval is legally required for the acquisition of our company. However, as the Company no longer qualifies a listed company under Belgian law following de-listing from Euronext Brussels in December 2023, the requirement, provided for by the Belgian Act of April 1, 2007, to launch a mandatory bid for all of our outstanding shares and securities giving access to shares if a person, as a result of its own acquisition or the acquisition by persons acting in concert with it or by persons acting on their account, directly or indirectly holds more than 30% of the voting securities in a company that has its registered office in Belgium and of which at least part of the voting securities are traded on a regulated market or on a multilateral trading facility designated by the Royal Decree no longer applies. This may allow existing shareholders or new investors to acquire significant influence or control over the Company by acquiring the shares in the market without being required to acquire the other outstanding voting securities, as well as for all other securities that entitle the holders thereof to the subscription to, the acquisition of or the conversion into voting securities.
There are several provisions of Belgian company law and certain other provisions of Belgian law, such as merger control, that may apply to us and which may make an unfriendly tender offer, merger, change in management or other change in control, more difficult. These provisions could discourage potential takeover attempts that third parties may consider and thus deprive the shareholders of the opportunity to sell their shares at a premium (which is typically offered in the framework of a takeover bid).
26
ITEM 4. INFORMATION ON THE COMPANY
A. History and Development of the Company
We were incorporated on January 10, 2003 as a company with limited liability (naamloze vennootschap/société anonyme) incorporated and operating under the laws of Belgium. We are registered with the legal entities register (Liège) under enterprise number 0479.292.440. We were publicly listed on Euronext Brussels in June 2006 and we had ADSs listed on the Nasdaq Capital Market in November 2021. In November 2023, we completed a share consolidation of our ordinary shares by means of a 1-for-10 reverse split, such that following the share consolidation each ADS represented one ordinary share (instead of 10 ordinary shares previously). Following the share consolidation and also during November 2023, we completed the mandatory exchange of all of our ADSs for one ordinary share each and subsequently terminated our ADS facility. Following a transition period, the Company de-listed its ordinary shares from Euronext Brussels and as of December 18, 2023, our ordinary shares began solely trading on the Nasdaq Capital Market under the symbol “MDXH.” In October 2010, the Company’s name was changed from OncoMethylome Sciences SA to MDxHealth SA. We have two directly held, wholly owned subsidiaries: MDxHealth, Inc., a Delaware company incorporated in April 2003, and MDxHealth B.V., a Dutch company incorporated in September 2015. Our agent for service of process in the United States is MDxHealth, Inc., which is located at 15279 Alton Parkway — Suite 100, Irvine, CA 92618. MDxHealth, Inc.’s telephone number is +1 949-812-6979.
Our headquarters and principal executive offices are located at CAP Business Center, Zone Industrielle des Hauts-Sarts, Rue d’Abhooz 31, 4040 Herstal, Belgium, our telephone number is +32 4 257 70 21 and our email is info@mdxhealth.com. Our website address is www.mdxhealth.com. The information contained on, or accessible through, our website is not incorporated by reference into this annual report, and you should not consider any information contained in, or that can be accessed through, our website as part of this annual report or in deciding whether to purchase our ordinary shares.
Our capital expenditures for the years ended December 31, 2023, 2022 and 2021 amounted to $2.7, $2.8 million and $0.9 million, respectively. These capital expenditures primarily consisted of laboratory equipment, information technology equipment, and leasehold improvements. Our research and development costs for the years ended December 31, 2023, 2022 and 2021 amounted to $6.4 million, $5.5 million and $5.6 million, respectively. These research and development costs primarily consisted of expenses incurred in connection with the development of our pipeline products, such as labor costs (including salaries, bonuses, benefits, and stock-based compensation), reagents and supplies, clinical studies, outside services, patent expenses, depreciation of laboratory equipment, facility occupancy and information technology costs. We recognize research and development expenses in the period in which they are incurred, except for those development expenses that qualify for capitalization (refer to Note 11 in the consolidated financial statements). We expect that our research and development expenses will increase in absolute dollars as we continue to develop additional products, however, we expect that these expenses will decrease as a percentage of revenue over the long term, though they may fluctuate as a percentage from period to period due to the timing and extent of these expenses. For the near future, our investments will mainly remain in the United States where our molecular laboratory facility is currently located.
The SEC maintains a website that contains reports, proxy and information statements, and other information regarding issuers that file electronically with the SEC at http://www.sec.gov. We also maintain a website at http://www.mdxhealth.com/. The reference to our website is an inactive textual reference only and the information contained in, or that can be accessed through, our website or any other website cited in this annual report is not a part of this annual report.
B. Business Overview
Overview
We are a commercial-stage precision diagnostics company providing non-invasive, clinically actionable and cost-effective urologic solutions to improve patient care. Our core menu of testing solutions, Select mdx for Prostate Cancer (“Select mdx”), Confirm mdx for Prostate Cancer (“Confirm mdx”), and Genomic Prostate Score (“GPS”), provide personalized genomic insights to both physicians and patients navigating the complexities of prostate cancer diagnosis and treatment. Our other testing solutions address related urologic diseases and conditions that are often managed by the same specialists who utilize our core prostate cancer tests. Each of our cutting-edge molecular diagnostic technologies provides patient-specific results enabling tailored approaches that enhance patient well-being while minimizing the need for invasive and unnecessary treatments and procedures.
More than 7,000 physicians have ordered over 250,000 mdxhealth tests. We have established a systematic approach to bring our precision diagnostic solutions to market, centered on proactive engagement, education, and market expansion aimed at healthcare professionals and their patients. Each of our core tests, Select mdx, Confirm mdx and GPS, have been included in prostate cancer detection and treatment guidelines published by the National Comprehensive Cancer Network (“NCCN”), a non-profit alliance of the 31 leading cancer centers in the United States, and each core test has also successfully completed formal technical assessment review for Medicare reimbursement, culminating in positive final local coverage determinations (“LCDs”).
Building from the foundation of our complementary marketed products, we are committed to sustained growth, with our core management principles defined by a commitment to focus, commercial execution and operating discipline throughout our organization. Our dedicated commercial team concentrates on cultivating relationships with major community urology centers, prioritizing those with significant patient volumes. We foster enduring connections with key physicians and practice groups who serve populations eligible for our solutions. Our overarching objective is to provide comprehensive support to physicians utilizing our tests throughout the entire patient journey, from initial diagnosis to advanced prostate cancer management.
27
Our Product Portfolio
Our core commercial tests address a substantial unmet clinical need in the prostate cancer decision pathway. According to the American Cancer Society, prostate cancer is the most common, and second deadliest, form of cancer in males in the United States. Prior to the emergence of precision diagnostic solutions, existing tests were critically flawed, leading to overdiagnosis and inappropriate treatment decisions.
To screen at-risk men for prostate cancer, approximately 25 million prostate-specific antigen (“PSA”) tests are performed each year, and over 15% of those reveal heightened levels of PSA. An elevated PSA level can be caused by many different sources, the majority of which are not cancer. Current clinical guidelines suggest that men with an elevated PSA should be considered for a prostate biopsy, so that a pathologist can visually inspect the sampled tissue to identify any sign of malignancy. However, 60% of biopsies are negative, not revealing any cancer, and as many as a third of these negative biopsies are false negatives, providing limited comfort to patients and their physicians that cancer was not missed. The relatively modest sensitivity and specificity of these current standard-of-care tests and procedures has led to increased patient anxiety, potentially unnecessary, invasive and costly interventions, and increased complications and hospitalizations.
Our Select mdx test — which is a noninvasive urine test with 95% Negative Predictive Value (“NPV”) for clinically significant prostate cancer — can be used to help physicians determine whether a costly, painful and complication-prone needle-core biopsy is advisable when a patient presents with an elevated PSA level or an abnormal digital rectal exam (“DRE”). For those men who proceed to a biopsy procedure, our Confirm mdx test, which measures biomarker signals in the biopsied tissue, provides additional information to physicians and increases the accuracy of the biopsy, with a 96% NPV for clinically significant disease.
Upon diagnosis of localized prostate cancer, our GPS test — which measures the expression of a panel of genes in prostate cancer tissue to predict the likelihood of having adverse pathology, risk of metastasis, and prostate cancer death — helps to inform treatment decisions and to identify patients who may avoid unnecessary interventions.

To further supplement our prostate cancer menu, with Resolve mdx we have developed a novel, advanced urinary tract infection (“UTI”) test that delivers patient-specific antimicrobial treatment options within 48 hours (standard urine cultures can take up to 5 to 7 days). Developed especially for patients with recurrent, persistent, and complicated UTIs, Resolve mdx combines precise pathogen identification and resistance gene detection with a proprietary susceptibility methodology that identifies personalized oral antibiotic options for fast resolution and improved patient outcomes.
Our Competitive Strengths
We believe we have the following competitive strengths, which underpin our commercial execution success and will position us for sustainable growth:
| ● | Targeted Menu Improving Prostate Cancer Diagnosis and Treatment Decisions. We offer a menu of tests that provide clinically actionable results for men along the prostate cancer pathway, which includes men at-risk for, as well as men newly diagnosed with, prostate cancer. Collectively, Select mdx, Confirm mdx and GPS provide urologists with a clear clinical pathway to accurately identify and appropriately treat prostate cancer while minimizing the use of aggressive procedures and treatments, improving health outcomes and significantly lowering costs to the healthcare system. |
| ● | Strong Commercial Focus and Presence. We aim to increase adoption of our commercial tests by leveraging our direct sales force in the United States to continue to market and sell to our urology-focused network. We have significant experience in building effective commercial teams consisting of sales reps, strategic account managers, and medical science liaisons led by a management team with a track record of success. In addition, our payor and reimbursement, revenue cycle management and client services groups provide expert support for our field sales team as well as our patients and customer base. We believe we can leverage these groups to explore additional opportunities for growth based on this commercial channel. Outside the United States, we will continue to evaluate distribution partners to drive adoption in markets where our menu is best suited. |
28
| ● | Commercial Channel Advantage. Building from the launch of our first commercial test in 2012, we have established MDxHealth as an industry leader in precision diagnostics for prostate cancer detection and treatment. We intend to take advantage of our established urology and pathology relationships to support menu expansion and additional growth opportunities as appropriate and within our focus. |
| ● | Compelling Reimbursement Strategy. Adoption of our Confirm mdx, Select mdx and GPS tests has been supported by LCDs issued via the MolDX Program, their inclusion in NCCN and other U.S. and internationally recognized clinical-practice guidelines, as well as consistent expansion of coverage by commercial payors. Our Resolve mdx UTI test is currently reimbursed by Medicare and most private insurance payors, based on nationally recognized Current Procedural Terminology (“CPT”) codes. |
| ● | Robust and Reliable Technology. We possess a proprietary know-how and intellectual property portfolio capable of advancing our precision diagnostics pathway as well as high quality laboratory operations, including our CAP accredited, CLIA certified and New York State Department of Health (“NYSDOH”) approved molecular laboratory facilities. We also have an extensive library of biomarkers that can be applied in additional urology and men’s health diagnostics. |
| ● | Proven Leadership with Industry Expertise. Our management team members have proven track records of execution and value creation across medical devices, diagnostics and biotech. We believe we have built a culture of performance, responsibility and accountability — from research and development, to selling and marketing, and operations and management — and are committed to building value for all of our stakeholders, including patients, customers, employees and shareholders. |
Our Strategy
Our ultimate goal with our core testing solutions is to take an at-risk patient from prostate cancer screening all the way through the diagnostic and therapeutic pathway of prostate cancer. As such, we are focused on continuing to drive adoption of our Select mdx, Confirm mdx, and GPS tests and expand our product offerings. The key elements of our strategy include:
| ● | Physician and Patient Education. One important component of our efforts to successfully penetrate the urology market and promote clinical adoption of our Select mdx, Confirm mdx, GPS and Resolve mdx tests is to drive awareness of these tests. We educate physicians and patients through a variety of channels including by supporting clinical studies for the publication of peer reviewed journals and abstracts at key scientific conferences, forging relationships with the leading medical and scientific opinion leaders in urology, developing strategic partnerships with leading pathology laboratories with large urology client bases and via public relations and advertising campaigns. |
| ● | Expand Test Menu. We intend to build on our leadership in the prostate cancer diagnostic space by expanding our existing menu of tests. We are currently developing a candidate test, Monitor mdx, for the prostate cancer diagnostic and treatment pathway. Monitor mdx is intended to function as a non-invasive solution that risk stratifies patients for continued active surveillance versus intervention, while also improving patient compliance with active surveillance protocols. |
| ● | Expand Reimbursement. An important component of our commercial strategy is to expand reimbursement for our tests. Our Select mdx, Confirm mdx and GPS tests have been covered by Medicare MolDX LCDs since 2023, 2014 and 2015, respectively. Our managed care team continues to pursue adoption of positive coverage and reimbursement policies and contracts by other payors. We believe the clinical utility and actionability of our tests, combined with our experience and knowledge of the complex coverage and reimbursement landscape in the United States, will enable us to expand coverage and reimbursement among the commercial payor market. We continue to build upon our successful strategy, supported by governmental and commercial coverage policies, as a foundation to secure additional contracts from major payors. |
Market Opportunity
Prostate cancer is the most diagnosed cancer and the second leading cause of cancer death in men. According to the NIH National Cancer Institute, in 2024, approximately 299,000 men are expected to be diagnosed with prostate cancer in the United States, with more than 35,000 dying from the disease.
29
There are currently significant challenges with diagnosing and treating prostate cancer in the United States. Approximately 25 million PSA tests are performed each year, and over 15% of those reveal elevated levels of PSA. Current clinical guidelines suggest that men with an elevated PSA should be considered for a prostate biopsy; however, approximately 50-60% of biopsies do not reveal any cancer, and as many as a third are false negatives, providing limited comfort to patients and their physicians that cancer was not missed. For those men whose biopsies reveal cancer, the majority will harbor indolent cancer, but traditional methods are unable to accurately identify which of these men might safely avoid invasive and costly interventions. In addition, for patients diagnosed with localized prostate cancer who are on Active Surveillance, management of their disease relies on the measurement of PSA levels, digital rectal exam (“DRE”) results, and surveillance biopsies, which can be unreliable and lead to overdiagnosis and overtreatment.
The relatively modest sensitivity, specificity and prognostic ability of current standard-of-care tests and procedures has led to increased patient anxiety, unnecessary, invasive and costly interventions, and increased complications and hospitalizations. Our suite of commercial products addresses these issues, presenting a substantial market opportunity. The Company has calculated approximate addressable market opportunities for our menu of tests, based on the estimated:
| ● | 3 million men screened for prostate cancer annually; |
| ● | 500,000 men who undergo prostate biopsies annually; |
| ● | 299,000 men diagnosed prostate cancers annually; and |
| ● | 2 million UTI cases managed by urologists annually. |

* Monitor mdx test in development (all figures based on management estimates).
Commercial Products
Select mdx for Prostate Cancer Urine Test
The current standard for prostate cancer screening is the PSA blood test. Unfortunately, the PSA is not specific to clinically significant prostate cancer — it is more of an indicator of prostate health. There are many factors such as benign prostatic hyperplasia, inflammation, prostatitis and a naturally occurring enlarged prostate that can cause an elevated PSA. In men with an elevated PSA level between 3-10 ng/mL, only 25-40% of biopsies reveal cancer — and the majority of these identified cancers are indolent. Also, following a prostate biopsy procedure, around 18% of men suffer complications (blood in urine) and around 3% are hospitalized for infection (sepsis). Select mdx helps physicians determine if a patient is at higher or lower risk for prostate cancer and which men can safely avoid biopsy.
Select mdx is a non-invasive urine test that measures the expression of two mRNA cancer-related biomarkers (HOXC6 and DLX1) combined with an advanced clinical model incorporating traditional risk factors. The test provides a personalized risk profile that helps the physician determine whether the patient may benefit from a biopsy and early prostate cancer detection or whether the patient can avoid a biopsy and return to routine screening.
Men identified by the test as having a high likelihood of clinically significant cancer can, upon biopsy, be diagnosed and treated sooner, while men identified as having very low risk may avoid biopsy.
30
The following chart depicts the functioning of the Select mdx test:

Guidelines Inclusion
Select mdx has been included in the NCCN Prostate Cancer Early Detection guidelines since 2020. Select mdx has also been included in the EAU Prostate Cancer guidelines since 2018.
Clinical Validation Studies
The use of Select mdx as a predictive test to identify men at low risk for aggressive prostate cancer has been well validated in both scientific and clinical studies.
Results from the clinical validation study for Select mdx confirmed its superior performance compared to other commonly used biomarker tests and risk calculators. The test’s NPV of 95% in the validation study means that if the test identifies a very low risk, the physician and patient can be 95% sure that a subsequent biopsy will not detect Gleason score ≥7 prostate cancer, information that may provide a level of confidence needed to avoid a biopsy. The test has a very high predictive accuracy (AUC 0.85) for high-grade prostate cancer, which is significantly better than the Prostate Cancer Prevention Trial Risk Calculator (“PCPTRC”) version 2.
There are twelve published studies assessing the Select mdx test and which together demonstrate its analytical validity, clinical validity, clinical utility and positive health economic outcomes. These studies, all of which have been published in peer-reviewed publications, evaluated more than 4,500 patients in the aggregate.
The following is a summary that highlights key findings from some of these studies.
| ● | Analytical validity. A study published in 2017 illustrated, in an independent laboratory, the performance characteristics and robustness of the Select mdx mRNA assay, covering all aspects of analytical method validation including assay sensitivity, specificity, linearity, precision, repeatability and reproducibility using pre-specified acceptance criteria. |
| ● | Clinical validity. In a study published in 2019, the Select mdx test demonstrated an NPV of 95%. Urine samples were collected from 1,955 men from The Netherlands, France and Germany prior to an initial prostate biopsy. Select mdx molecular biomarker results were combined with other risk factors in a clinical model optimized to detect International Society of Urological Pathology Grade Group 2 or greater prostate cancer in men. Results in the validation cohort were compared with the independent PCPTRC version 2. The full validation cohort of 916 men including all prostate specific antigen levels yielded an AUC of 0.85 with 93% sensitivity, 47% specificity and 95% negative predictive value. The PCPTRC AUC was 0.76. In the 715-patient validation cohort, limited to subjects with PSA less than 10 ng/ml, the AUC was 0.82 with 89% sensitivity, 53% specificity and 95% negative predictive value. The PCPTRC AUC was 0.70. |
| ● | Clinical utility. In a 2019 study, Select mdx had a significant impact on initial prostate biopsy decision-making in a U.S. community urology setting. Biopsy rates in Select mdx positive men were 5-fold higher than in Select mdx negatives. |
31
| ● | Health economic outcomes. A 2018 study demonstrated that routine use of the Select mdx test to guide biopsy decision making improved health outcomes and significantly lowered costs in American men at risk for prostate cancer. Compared to the current standard of care, Select mdx implementation would result in an average of 0.045 quality-adjusted life years (“QALYs”) gained at a cost savings of $1,694 per patient. Assuming approximately 300,000 men are biopsied each year, this translates to an incremental 14,000 QALYs gained at cost savings of $500,000 annually. |

Confirm mdx for Prostate Cancer Tissue Test
Approximately 30% of men with a cancer-negative prostate biopsy actually have cancer. Prostate cancer is difficult to diagnose because it is both heterogenous and multi-focal. The standard of care for diagnosing prostate cancer is a transrectal ultrasound guided biopsy. However, this procedure samples less than 1% of the entire gland, leaving men at risk for undetected prostate cancer.
Confirm mdx is a well-validated epigenetic test that guides the detection of occult (hidden) prostate cancer on a patient’s previously biopsied negative tissue. The test can help urologists determine a man’s risk for harboring clinically significant prostate cancer despite having a cancer-negative biopsy result, and it has a number of unique features/advantages.
For patients with an initial negative biopsy, few options are currently available to guide an urologist in determining whether or when an additional biopsy procedure is warranted. Fear of occult prostate cancer leads to additional procedures, leading many men to receive multiple follow-up biopsy procedures to rule out the presence of cancer.
The Confirm mdx test addresses prostate biopsy sampling concerns, helping urologists to:
| ● | “Rule-out” men from undergoing potentially unnecessary repeat biopsies and screening procedures, helping to reduce complications, patient anxiety and excessive healthcare expenses associated with these procedures; and |
| ● | “Rule-in” high-risk men with a previous negative biopsy result who may be harboring undetected cancer (false negative biopsy result) and therefore may benefit from a repeat biopsy and potentially treatment. |
For men with a negative biopsy, independently published clinical studies have shown that the Confirm mdx test is the most significant, independent predictor of prostate biopsy outcomes relative to other available clinical factors such as age, PSA and DRE results. Incorporating Confirm mdx into clinical practice can reduce the number of unnecessary repeat biopsies, yielding clinical and economic value for healthcare providers, patients and payors. Confirm mdx can aid urologists with patient management decisions regarding the need for follow-up testing and procedures with the identification of low-risk patients testing negative for DNA hypermethylation.
The use of Confirm mdx for prostate cancer detection using methylation-specific PCR and cancer-associated epigenetic biomarkers to improve upon histopathology has been well validated in both scientific and clinical studies. DNA methylation, the most common and useful measure of epigenetic abnormality testing, is responsible for the silencing of key tumor suppressor genes. DNA methylation biomarkers associated with prostate cancer have been extensively evaluated.
GSTP1 is a widely studied and reported epigenetic biomarker associated with prostate cancer diagnosis, encoding the glutathione S-transferase Pi 1 (GSTP1) protein involved in detoxification, due to its high sensitivity and specificity. Complementing GSTP1, methylation of the APC and RASSF1 genes is frequently found in prostate cancer, and these markers have demonstrated a “field effect” aiding in the identification of biopsies with false-negative histopathological results.
The epigenetic field effect is a molecular mechanism whereby cells adjacent to cancer foci can contain DNA methylation changes, which may be indistinguishable by histopathology, but detectable by methylation-specific PCR testing. The presence of epigenetic field effects associated with prostate cancer has been widely published and is the basis of activity for the Confirm mdx assay to aid in the detection of occult prostate cancer on previously biopsied, histopathologically negative tissue.
32
The following image depicts how the Confirm mdx test identifies false-negative biopsies:
Confirm mdx Field Effect
 |
 |
Guidelines Inclusion
Confirm mdx has been included in the NCCN Prostate Cancer Early Detection guidelines since 2016. Confirm mdx has also been included in the EAU Prostate Cancer guidelines since 2018.
Confirm mdx Clinical Validation Studies
The use of Confirm mdx for prostate cancer detection to improve upon histopathology has been well validated in both scientific and clinical studies.
There are more than 55 published studies on the genes and technology used in the Confirm mdx test. Among these, studies demonstrating the analytical validity, clinical validity, clinical utility and positive health economic outcomes of the Confirm mdx test evaluated more than 1,200 patients in the aggregate.
The following is a summary that highlights key findings from some of these studies.
| ● | Analytical validity. A study published in 2012 illustrated the performance characteristics and robustness of the Confirm mdx multiplex DNA methylation assay, covering the analytical method including assay sensitivity, specificity, linearity, precision, repeatability and reproducibility using pre-specified acceptance criteria. |
| ● | Clinical validity. The clinical validity of the Confirm mdx test has been demonstrated in two large, blinded clinical validation studies published in 2013 and 2014, yielding a NPV of ~90% for all prostate cancer, which is significantly higher (p < 0.001) than that afforded by standard histopathology review, as well as a NPV of 96% for clinically significant prostate cancer. Further, when compared to all pertinent risk factors for prostate cancer detection (patient’s age, serum PSA level, DRE, histopathological findings on the previous cancer-negative biopsy and the epigenetic assay), Confirm mdx was shown to be the most significant, independent predictor for prostate cancer in a repeat biopsy with an odds ratio of 3.24 (and a p-value < 0.001). An additional clinical validity study published in 2017 demonstrated that the Confirm mdx test improved the identification of African American men at risk for aggressive cancer missed by a prostate biopsy, with accuracy equivalent to prior studies in predominantly Caucasian populations. |
| ● | Clinical utility. A 2014 study reported on the real-world use of the Confirm mdx assay, demonstrating that the test impacts physician behavior. A very low rate of repeat biopsies (4.4%) was observed in the Confirm mdx negative men, as compared to the expected 43% rate of repeat biopsy reported in a large population-based randomized trial sponsored by the National Cancer Institute. |
33
| ● | Health economic outcomes. In a study published in 2013, a budget impact model developed to evaluate the effect of the Confirm mdx assay on healthcare spending demonstrated significant potential healthcare savings associated with the reduction of repeat biopsies and complications avoided. Under the study’s model, utilization of Confirm mdx would bring approximately $500,000 in annual savings per 1 million covered patients. |
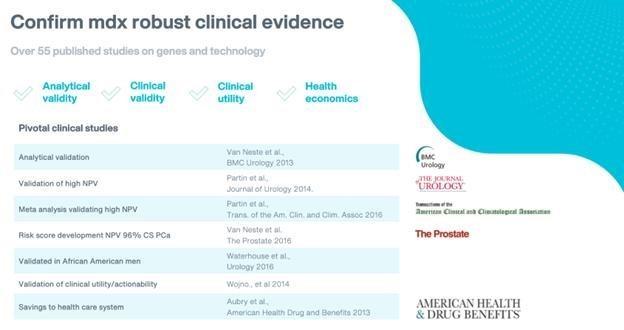
GPS Tissue Test
Currently, most cases of detected prostate cancer remain indolent and men with the disease often die from other causes. Patients who have indolent prostate cancer may be appropriately managed with observation or active surveillance (“AS”), while those with aggressive cancers may benefit from immediate treatment. Internationally recognized clinical organizations have implemented guidelines stressing the importance of discerning favorable from unfavorable disease features to guide personalized management for patients diagnosed with low- and high-risk prostate cancer, including among others NCCN, the American Urology Association, American Society for Radiation Oncology, Society of Urologic Oncology, the American Society of Clinical Oncology, and the National Institute for Health and Care Excellence. The use of AS has increased in recent years and it is now estimated that up to 50% of clinically low-risk patients choose AS, while the remainder choose some type of immediate treatment.
The tissue-based GPS test assesses 17 genes in total — 12 cancer-related genes representing 4 important biologic pathways (androgen signaling, cellular organization, stromal response, and proliferation) together with 5 references genes (to control for RNA quantity and quality). The test uses reverse transcription polymerase chain reaction (“RT-PCR”) to measure gene expression in very small amounts of prostate tumor tissue (requiring as little as 5 ng of RNA). Genetic expression of the 12 cancer-related genes, normalized by the 5 reference genes, is used in an algorithm to generate a GPS result that ranges from 0 to 100, with higher scores associated with more aggressive disease. The GPS test, in conjunction with clinical risk factors, is predictive of a finding of adverse pathology (“AP”) upon a radical prostatectomy (“RP”) and clinical recurrence following RP, and consequently provides clinicians and patients with information about the likely aggressiveness of their cancer to help guide initial treatment decisions.
For patients with NCCN very low- to favorable intermediate- prostate cancer, the GPS test provides information on the risk of AP to help physicians guide personalized treatment for patients at the initial decision point. For the unfavorable intermediate- and high-risk groups, the GPS test helps inform decisions on the intensity of definitive treatment. The patient results report gives the risk of a patient developing metastasis within 10 years, risk of PCD (death from prostate cancer) within 10 years, and risk of tumor aggressiveness based on AP result outlining the clinical characteristics of each NCCN risk group.
The GPS test is intended for men with clinically localized prostate cancer who have undergone biopsy within 3 years and have not yet started treatment. Patients with any NCCN risk category between very low- and high-risk are eligible for GPS testing.
Guidelines Inclusion
GPS has been included in the NCCN Prostate Cancer guidelines since 2019.
Clinical Validation Studies
The use of GPS as a predictive test to identify men at low risk for aggressive prostate cancer has been well validated in both scientific and clinical studies.
34
The following is a summary that highlights key findings from clinical studies regarding the GPS test.
| ● | Clinical validity. In a prospective and retrospective study on 402 patients published in 2015, the GPS test demonstrated its ability to discriminate prostate cancer aggressiveness in biopsy tissue despite tumor heterogeneity and multifocality. Further, the test demonstrated its ability to improve prediction of adverse pathology. |
| ● | Clinical utility. Numerous clinical studies have shown the impact of GPS on treatment recommendations and have demonstrated that generally, use of GPS increases the proportion of men for whom AS is recommended, when the background AS rates are similar to the national average for these risk groups. |
| o | In a prospective decision impact study by Badani et al. (2015) of 158 patients with NCCN very low-, low- and low intermediate-risk disease, the GPS test resulted in a 26% change in recommended treatment modality or intensity and patients who received GPS testing had a 24% relative increase in AS recommendations. |
| o | In a chart review study by Dall’Era et al. (2015) of 211 patients with NCCN very low- or low risk-disease, biological risk predicted by GPS differed from NCCN clinical risk alone in 62 men (39%). AS use increased by 24% in patients who received the GPS test versus patients who did not receive the GPS test. |
| o | In a study by Eure et al. (2017) of 297 patients, 23% of patients’ risk was restratified. In the NCCN low-risk group, the change in the management plan between AS and immediate treatment was 28%; 51% of the men who were initially recommended immediate treatment pre-GPS testing switched to AS post-GPS testing, while 14% of those initially recommended AS switched to immediate treatment post-GPS testing. Among the men who elected AS, their one-year AS persistence rates remained high at 89%. A fourth study by Lynch et al. (2017) demonstrated utility in a US Department of Veterans Affairs population, an equal-access healthcare system with high baseline AS rates without testing. The study compared management patterns for men with NCCN very low-, low-, and intermediate-risk PCa with and without molecular profiling. Overall, use of AS was 12% higher (absolute; relative increase 19%) in GPS-tested versus untested men, with the biggest increases observed in low-risk patients (90% versus 72% for tested versus untested, respectively) and patients under the age of 60 (75% versus 42% for tested versus untested, respectively). |
| ● | Health economic outcomes. A 2016 publication in Reviews in Urology presented a comprehensive economic analysis of the GPS test in low-risk prostate cancer patients. Results showed that use of the GPS test results in a net savings of $2,286 USD per patient — including the cost of the test — by decreasing unnecessary immediate invasive treatments. The study demonstrated that incorporation of the GPS test as part of the treatment decision algorithm for patients with NCCN very low- and low-risk disease (64% of the study population) led to a 21% net increase in the use of AS. Of these, treatment patterns and cost for 80 men tested with GPS were compared to 100 patients in the same practice without genomic testing. Based on a real-world practice setting in the U.S. Northeast with a contemporary patient population and using current treatment cost averages, these results demonstrated that the use of the GPS test represented a reduction in cost of unnecessary intermediate interventions by more than 50% over a 6-month period. Additional savings could also be expected by removing the cost of management of associated side effects of treatment such as impotence and incontinence. |

35
Resolve mdx for Urinary Tract Infection
UTIs affect around 10 million people who seek medical attention every year. It is estimated that 2-3 million of these cases lead to emergency department visits. UTIs can be complicated and recurrent, resulting in painful symptoms such as abdominal and rectal pain, frequent urination, burning or pain during urination, and fatigue. Antibiotic resistance is a significant issue, observed in up to one-third of UTI infections, causing about 2.8 million infections and 35,000 deaths annually, according to the CDC.
The traditional method of conducting a UTI test, urine culture, can take up to 3 to 5 days to produce results. Unfortunately, relying solely on culture-based testing may produce equivocal results of “mixed flora” in up to 30% of cases. Often, clinicians will rely on empiric therapy to treat UTIs, which can lead to overuse/misuse of antibiotics. Through the use of Resolve mdx, we help support antibiotic stewardship initiatives, as our test identifies personalized antibiotic options that would be expected to be more effective against the patient’s infection.
To address this unmet clinical need, with Resolve mdx we developed an advanced urine test that utilizes Polymerase Chain Reaction (“PCR”) technology to detect and quantify both infectious pathogens and resistance genes. Resolve mdx also includes susceptibility testing to identify the antibiotics that may be best suited to resolve the urinary tract infection. This approach provides prompt and accurate pathogen identification and personalized antibiotic recommendations. PCR-based testing offers improved sensitivity and specificity in identifying pathogens, addressing the problem of “mixed flora” results associated with traditional testing methods. This increased accuracy provides physicians with clinically actionable information to guide their decision-making for patient care.
Our proprietary Antibiotic Susceptibility Testing method included with the Resolve mdx test, called ASTX, determines how each pathogen responds to the 26 antibiotics tested. The unique aspect of ASTX is that it tests whole urine samples, ensuring accurate results and identifying the most effective treatment options for patients.
Resolve mdx Pathogens Tested, Resistance Genes and Antibiotics
Our goal is to help pinpoint not only the offending organisms, regardless of how many are identified, but also the most likely oral antibiotics capable of clearing the entire infection. We estimate the addressable market in the United States for UTI testing at approximately 2 million cases annually, or $1 billion.
Hereditary Prostate Cancer Genetic Test
In March 2024, we announced that we had added to our offerings a comprehensive hereditary prostate cancer genetic test. With a simple saliva sample, this test provides insights into a patient’s risk of developing cancer, disease progression, and response to prostate cancer therapies. Germline testing of prostate cancer patients is recommended in the NCCN guidelines and is covered by Medicare and many commercial insurance plans, for eligible patients.
Pipeline
We intend to build on our leadership in the urologic diagnostic space by expanding our menu of tests beyond Select mdx, Confirm mdx, GPS and Resolve mdx. We are currently developing an additional product for the prostate cancer diagnostic and treatment pathway. AS is a management approach for prostate cancer that involves closely monitoring the cancer’s progression through regular tests and imaging, without immediately initiating active treatment such as surgery or radiation therapy. Since not all prostate cancers progress the same manner, there is a significant unmet clinical need to help physicians identify which patients may benefit from AS, distinguishing those at risk for disease progression.
36
Men on AS are monitored using PSA, MRI and periodic biopsies to determine if their prostate cancer has progressed and whether definitive treatment is necessary. We are actively analyzing urine and blood biomarker panels with the goal of developing a non-invasive test for monitoring these patients that we call Monitor mdx. A testing solution that allows a physician to forego or delay their patient’s surveillance biopsy would represent a significant business opportunity with little or no direct competition.
If our development efforts are successful, MDxHealth would have a full offering of biomarker-based prostate cancer tests from early detection to treatment and management. Select mdx and Confirm mdx help determine which patients should (or should not) undergo a prostate biopsy, while our GPS test guides the decision to enter into AS upon an initial diagnosis of prostate cancer. In the AS setting, Monitor mdx would provide methods to identify and monitor patients who could remain on AS as a treatment option.
Laboratory Operations
We currently process tests at our 38,000 square foot, CAP-accredited, CLIA-certified, and NYSDOH-approved molecular laboratory and office facility located at our U.S. headquarters in Irvine, California; and through our 8,000 square foot, CAP-accredited, CLIA-certified laboratory facility in Plano, Texas. We also operate a 7,800 square foot office and a laboratory facility located in Nijmegen, The Netherlands, where we primarily conduct research and development activities. Our GPS test is currently performed by Exact Sciences in its laboratory facility in Redwood City, California. Our current clinical reference laboratory has excess processing expansion capacity with incremental increases in laboratory personnel and equipment, including expansion capacity for laboratory facilities. We believe that we currently have sufficient capacity to process all of our tests. We may require additional facilities in the future as we expand our business and believe that additional space, when needed, will be available on commercially reasonable terms.
Sales and Marketing
Our sales approach focuses on the clinical and economic benefits of our tests as supported by peer-reviewed literature covering the clinical validation and utility of these tests. Our sales and marketing team includes molecular diagnostic specialists, reimbursement account managers, clinical liaisons and client service personnel. Sales personnel are primarily field-based, while client service and marketing personnel are primarily based in our California headquarters.
Our sales team is trained to address the clinical, economic and reimbursement questions associated with selling our tests. Our sales force focuses on educating its primary and secondary clientele, which consists of urologists and their clinical staff, including nurses, laboratory and pathology personnel, finance administrators and billing personnel, and secondarily the pathology and laboratory staff who fulfil test requests on behalf of their clinician clients. Our current urology sales force consists of direct sales representatives, strategic account managers and regional sales managers.
Our sales efforts are directed towards increasing adoption and utilization of our tests in clinical practice. The strategy entails:
| ● | working with community-based, large group practices and academic urologists to educate them on the clinical and economic benefits provided by our tests; |
| ● | nurturing and strengthening relationships with KOLs in urology; |
| ● | supporting ongoing collaborations with leading universities and research institutions that have generated clinical validation data supporting our tests; and |
| ● | encouraging ongoing exploration and studies of expanded indications for our tests. |
Successful penetration of the urology market and clinical adoption of our tests has been achieved with a multi-faceted approach to build brand recognition and raise awareness of the tests. Our efforts and programs include sharing information with leaders in the medical community on a national and regional scale, supporting clinical studies for the publication of peer reviewed journals and abstracts at key scientific conferences, development of tools for our customers to interact with patients and consumers (doctor-to-consumer education), providing support to patient advocacy groups like Prostate Conditions Education Council, developing strategic partnerships with leading pathology laboratories with large urology client bases, participation in industry trade shows and the implementation of public relations, media and advertising campaigns.
37
Reimbursement
Reimbursement of our tests by third-party payors is essential to our commercial success. Payment for our testing services may come from, in some cases:
| ● | third-party payors that provide health care coverage to the patient (e.g., commercial health insurance companies or managed care organizations); |
| ● | federal health care programs, such as Medicare, state Medicaid programs, the Department of Defense and Veterans Affairs hospitals in the United States; |
| ● | other government agencies or laboratories that order the testing service; and |
| ● | patients in cases where the patient has no insurance or coverage benefit, is underinsured or has insurance with cost sharing benefits whereby the insurance covers a percentage of testing costs, and the patients are responsible for a co-payment, co-insurance and/or deductible amount. |
Reimbursement for diagnostic tests furnished to Medicare beneficiaries (typically patients aged 65 or older) is typically based on a fee schedule set by CMS, a division of the HHS.
In 2014, 2015 and 2023, respectively, our Confirm mdx test, our GPS test and our Select mdx test received positive Medicare LCDs under the MolDX Program, which provide coverage and reimbursement for Medicare beneficiaries throughout the United States. Our Resolve mdx UTI test is currently reimbursed by Medicare and most private insurance payors, based on nationally recognized CPT codes.
We believe the clinical utility and actionability of our tests, combined with our experience and knowledge of the complex coverage and reimbursement landscape in the United States will enable us to expand coverage and reimbursement of our tests among the commercial payor market. We continue to build upon our successful strategy, using our Medicare LCDs for Confirm mdx, Select mdx and GPS and existing commercial payor contracts as a foundation to secure additional contracts from major payors.
Where there is a payor policy or contract in place, we bill in accordance with the terms of that policy or contract. Where there is no payor policy or contract in place, we pursue third-party reimbursement on behalf of each patient on a case-by-case basis. Our efforts on behalf of these patients involve a substantial amount of time and expense, and bills may not be paid for many months, if at all. Furthermore, if a third-party payor denies coverage after final appeal, it may take a substantial amount of time to collect from the patient, if we are able to collect at all.
Materials Needed for Our Laboratory Services
In connection with our role as a CLIA-certified provider of laboratory services, we assist healthcare providers with certain logistics related to the collection and return of samples for testing.
The processing of testing solutions requires items and services, the majority of which are sourced from multiple suppliers. Certain of the consumable supplies and reagents used in our testing process are procured from a limited number of suppliers, some of which are single source. In addition, we rely on a limited number of suppliers, or in some cases a single supplier (for example, for the automation of our deparaffination steps for the Confirm mdx test), for certain equipment with which we perform testing services. To date, we have acquired all of our equipment and the majority of our materials on a purchase order basis, and we generally do not have language included in contracts with our suppliers and manufacturers that commit them to supply equipment and materials to us.
Competition
The molecular diagnostics field is characterized by rapid technological changes, frequent new product introductions, changing customer preferences, emerging competition, evolving industry standards, reimbursement uncertainty and price competition. Moreover, the molecular diagnostics field is intensely competitive both in terms of service and price, and continues to undergo significant consolidation, permitting larger clinical laboratory service providers to increase cost efficiencies and service levels, resulting in more intense competition.
The market for assessing men at risk for prostate cancer is large. As a result, this market has attracted competitors, some of which possess substantially greater financial, selling, logistical and laboratory resources, more experience in dealing with third-party payors, and greater market penetration, purchasing power and marketing budgets, as well as more experience in providing diagnostic services. Some companies and institutions are developing liquid biopsy (blood and urine)-based tests and diagnostic tests based on the detection of proteins, mRNA, nucleic acids or the presence of fragments of mutated genes that are associated with prostate cancer. These competitors could have technological, financial, reputational, and market access advantages over us.
38
In regard to our Select mdx test, several directly competitive products are currently commercially available. In 2014, OPKO Health, a NYSE listed company, launched the 4Kscore test, a blood based 4-plex test which combines the results of the blood test with clinical information in an algorithm that calculates a patient’s percent risk for aggressive prostate cancer prior to an initial or repeat biopsy (no previous diagnosis of prostate cancer). The 4Kscore test received marketing approval from the FDA in December 2021. OPKO Health operates one of the largest clinical laboratories in the United States. The 4Kscore test competes directly with Select mdx. In 2016, ExosomeDx launched the ExoDx (Intelliscore), a urine-based test designed to assess whether patient presenting for an initial or repeat biopsy is at greater risk for high-grade prostate cancer. The ExoDx test competes directly with Select mdx. In 2018, Bio-Techne Corporation, a large U.S.-based, diversified life sciences company, acquired the ExoDx test. In addition to the ExoDx and the 4Kscore tests, the Prostate Health Index test (“phi score”) offered by Beckman Coulter, competes directly with the Select mdx test. In addition, each of these tests may also provide a competitive advantage since, unlike the Select mdx test, they do not require a digital rectal procedure as part of their specimen collection process. Each of Bio-Techne, OPKO Health and Beckman Coulter have greater resources and significantly larger sales and marketing teams than MDxHealth. As a result of these significantly greater resources, these competitors are able to make larger investments into the tests they produce and the sales and marketing of these tests, which may cause us to lose market share.
Regarding our GPS test, acquired in August 2022 from Exact Sciences, several directly competitive products are currently commercially available. Myriad Genetics offers the Prolaris test, a tissue-based genetic test to help identify those men who need treatment versus those who can choose active surveillance. Additionally, Veracyte offers the Decipher test, a tissue-based genetic test to help identify those men who need treatment versus those who can choose active surveillance. In addition to directly competitive genomic tests, traditional methods used by pathologists and clinicians to estimate risk for disease progression also pose competitive threats. Companies combining these traditional methods with artificial intelligence could potentially emerge as competitors, though most of these technologies are currently in the research stage.
In regard to our Confirm mdx test, several competitive products are currently commercially available. Both the 4Kscore test, offered by OPKO Health, and the ExoDx (Intelliscore) test offered by ExosomeDx, compete with the Confirm mdx test. Offered at a lower price point, both the 4Kscore and ExoDx tests offer a competitive price advantage over the Confirm mdx test. In addition, both the 4Kscore test and the PCA-3 test from Hologic, a urine-based test, are on the U.S. market as FDA-approved tests, which may be perceived as providing a competitive advantage since the Confirm mdx for Prostate Cancer test is not FDA approved. The PCA-3 test is intended for the same patient population as Confirm mdx for Prostate Cancer, but its performance has only been established in men who were already recommended by urologists for repeat biopsy.
Regarding our Resolve mdx test, several directly competitive products are commercially available. In 2016, Pathnostics, based in Irvine, California, launched its Guidance UTI test that combines PCR-based identification with antibiotic susceptibility testing to identify antibiotic options for the treatment of UTI. An early market mover, Pathnostics is currently the Company’s largest direct competitor in molecular testing for UTI. Pathnostics has patented its methodology for testing antibiotic susceptibility and has made substantial investments in clinical research to demonstrate its UTI test’s performance. In addition, UroKEY, a molecular and next-generation sequencing UTI test commercialized by Microgen dx, and Urine-ID, a UTI test commercialized by Vikor Scientific, each also competes directly with our Resolve mdx test.
In addition to competitive products, the Confirm mdx, Select mdx and GPS tests also face competition from mpMRI, a clinical diagnostic imaging procedure available to and used by physicians for many years, which focuses on visual tissue analysis. The mpMRI procedure can visually reveal potential locations of abnormal and potentially cancerous prostate tissue characteristics that distinguish tumors from healthy tissue. The visual aspect of diagnostic imaging may feel more accessible and be considered preferable by some physicians over molecular analysis, and there likely is an economic incentive for some physicians to earn a professional fee from the performance of mpMRI procedures. It may be difficult to change the methods or behavior of physicians to incorporate our testing solutions into their practices in conjunction with, or instead of, mpMRI clinical diagnostic imaging procedures. In addition, companies developing or offering capital equipment or point-of-care kits to physicians represent another source of potential competition. These devices are used directly by the physicians or their institutions, which can facilitate adoption.
Intellectual Property
Our intellectual property and its protection are crucial to the operation of our business. We are committed to developing and protecting our intellectual property and, where appropriate, filing patent applications or securing licenses to patents to protect our technology. We rely on a combination of patent, copyright, trademark and other agreements with employees and third parties to establish and protect our proprietary intellectual property rights. We also rely upon trade secret laws to protect unpatented know-how and continuing technological innovation. However, trade secrets can be difficult to protect, and do not provide protection against a third party independently discovering or recreating the information for which trade secret protection is claimed. We seek to protect our proprietary unpatented technology, know how, algorithms and processes, in part, by entering into confidentiality agreements with our employees, consultants, scientific advisors and contractors and limit access to, and distribution of, our proprietary information. We also require our officers, employees and consultants to enter into standard agreements containing provisions requiring confidentiality of proprietary information and assignment to us of all inventions made during the course of their employment or consulting relationship. We also enter into nondisclosure agreements with our commercial counterparties and limit access to, and distribution of, our proprietary information. We also seek to preserve the integrity and confidentiality of our data and trade secrets by maintaining physical security of our premises and physical and electronic security of our information technology systems.
39
We believe that our patent portfolio places us in a competitive position in the realm of molecular cancer diagnostics. We own or hold exclusive rights to a range of issued and pending patents in multiple countries worldwide covering our epigenetic and molecular tests and associated biomarkers, their methods of use and any other inventions that are important to the development of our business. Many of our commercially important technology and inventions are in-licensed from academic and commercial collaborators. Through our internal R&D programs, together with our academic and commercial collaborations, we continue to be at the forefront of researching and understanding the link between cancer and methylation (epigenetics), and how this link can be translated into meaningful clinical molecular diagnostic solutions. We consider patent protection of the technologies on which our products are based to be a key factor to our success.
Our intellectual property portfolio is managed by an in-house intellectual property team, which works in close collaboration with qualified external patent attorneys both in Europe and the United States. As of April 1, 2024, we own or have exclusive rights to more than 18 patent families related to our molecular technology and cancer-specific biomarkers. Specifically, there are 198 granted or pending patent applications in this group comprised of 15 issued or allowed U.S. patents, 5 pending U.S. provisional or non-provisional applications, 8 pending international patent applications filed under the PCT and 170 granted or allowed patents in jurisdictions outside the United States, including Japan, Canada, Israel and certain European countries. Our issued U.S. patents expire at various times between 2024 and 2036. Of these issued patents, 20 cover intellectual property used in our Select mdx test, the last of which expires in 2036, and 97 cover intellectual property used in our GPS test, the last of which expires in 2038. When these patents expire other companies will no longer be prohibited from incorporating the subject intellectual property into competing tests they may seek to develop. Nevertheless, given the significant unpatented proprietary and confidential intellectual property that we have developed and that is used in our tests, together with the clinical performance characteristics reported in published clinical studies that are specific to these branded tests, we believe there will be significant barriers to any competitors’ ability to use such previously patent-protected intellectual property to develop competitive tests.
In most countries in which we file, our patents have a life of 20 years from the date of the filing of the nonprovisional application. In the United States, a patent’s term may be lengthened by patent term adjustment, which compensates a patentee for administrative delays by the USPTO in granting a patent, or may be shortened if a patent is terminally disclaimed over an earlier-expiring patent. The expected expiration dates listed above assume that no terminal disclaimers will be filed. A patent term can also be lengthened in certain countries, including the United States, under circumstances where there is a delay associated with approval from a governmental regulatory agency.
We may in the future receive notices of claims of infringement and misappropriation or misuse of other parties’ proprietary rights and may from time to time receive additional notices. Assertions of misappropriation, infringement or misuse, or actions seeking to establish the validity of our patents could materially or adversely affect our business, financial condition and results of operations.
Collaboration and License Agreements
Collaborative research agreements and clinical research agreements
We have entered into agreements with universities, medical centers and external researchers for research and development work and for the validation of our technology and products. These agreements typically have durations of one to three years. In certain circumstances, we pay fixed fees to the collaborators and in exchange typically receive access and rights to the results of the work. MDxHealth has collaborated on research and clinical development with a number of the world’s leading academic and government cancer research institutes. These important relationships have provided us with additional resources and expertise for clinical marker validation as well as access to patient samples for testing.
Commercial and intellectual property licensing agreements
We have entered into agreements with universities and companies for in-licensing intellectual property. These agreements typically require us to pay an up-front fee, annual maintenance fees and/or minimum annual royalty fees, legal fees related to the patents, and certain milestone and royalty fees if the patents are eventually used in a commercialized product. In addition, we must provide the licensor with periodic reports.
In regard to our developed tests, we have entered into a range of marketing and sales arrangements with commercial entities in the normal course of our business. These relationships provide us with additional resources and infrastructure to expand the geographic reach and awareness of our solutions.
Government Regulations
Certain of our activities are subject to regulatory oversight by CMS pursuant to CLIA, as well as agencies in various states, including New York. We are subject to many other federal, state and foreign laws, including anti-fraud and abuse, anti-kickback and patient privacy. Failure to comply with applicable requirements can lead to sanctions, including withdrawal of products from the market, recalls, refusal to authorize government contracts, product seizures, exclusion from participation in federal and state healthcare programs, civil money penalties, injunctions, and criminal prosecution.
40
Laboratory Certification, Accreditation, and Licensing
We are also subject to U.S. and state laws and regulations regarding the operation of clinical laboratories. CLIA requirements and laws of certain states, including those of California, New York, Maryland, Pennsylvania and Rhode Island, impose certification requirements for clinical laboratories, and establish standards for quality assurance and quality control, among other things. CLIA provides that a state may adopt different or more stringent regulations than federal law and permits states to apply for exemption from CLIA if the state’s laboratory laws are equivalent to or more stringent than CLIA. For example, the State of New York’s clinical laboratory regulations, which have received an exemption from CLIA, contain provisions that are in certain respects more stringent than federal law. Therefore, as long as New York maintains a licensure program that is CLIA-exempt, we will need to comply with New York’s clinical laboratory regulations in order to offer our clinical laboratory products and services in New York.
Our U.S. laboratory facilities in Irvine, California and Plano, Texas are certified under CLIA. Clinical laboratories are subject to inspection by regulators and to sanctions for failing to comply with applicable requirements. Sanctions available under CLIA and certain state laws include prohibiting a laboratory from running tests, requiring a laboratory to implement a corrective plan, and imposing civil monetary penalties.
HIPAA and Other Privacy Laws
The Health Insurance Portability and Accountability Act of 1996, as amended by HITECH, established comprehensive protection for the privacy and security of health information. The HIPAA standards apply to three types of organizations, or “Covered Entities”: (1) health plans, (2) healthcare clearinghouses, and (3) healthcare providers that conduct certain healthcare transactions electronically. Covered Entities and their business associates must have in place administrative, physical, and technical standards to guard against the misuse of individually identifiable health information. As a clinical laboratory that engages in HIPAA-covered standard transactions, we are a Covered Entity subject to regulation under HIPAA. We have implemented privacy and security policies and procedures, provided required training to our personnel, and ensure that we enter into Business Associate Agreements with our vendors who have access to our protected health information. Penalties for violations of HIPAA include civil money and criminal penalties.
HIPAA establishes a federal “floor” with respect to privacy, security, and breach notification requirements and does not supersede any state laws insofar as they are broader or more stringent than HIPAA. Numerous state and certain other federal laws protect the confidentiality of health information and other personal information, including but not limited to state medical privacy laws, state laws protecting personal information, state data breach notification laws, state genetic privacy laws, human subjects research laws and federal and state consumer protection laws. These additional federal and state privacy and security-related laws may be more restrictive than HIPAA and could impose additional penalties. For example, the Federal Trade Commission uses its consumer protection authority under Section 5 of the Federal Trade Act to initiate enforcement actions in response to alleged privacy violations and data breaches. California recently enacted the California Consumer Privacy Act (“CCPA”), which went into effect on January 1, 2020. The CCPA, among other things, creates new data privacy obligations for covered companies and provides new privacy rights to California residents, including the right to opt out of certain disclosures of their information. The CCPA also creates a private right of action with statutory damages for certain data breaches, thereby potentially increasing risks associated with a data breach. California recently amended and expanded the CCPA through another ballot initiative, the California Privacy Rights Act (“CPRA”), passed on November 3, 2020. It remains unclear what, if any, additional modifications will be made to the CPRA by the California legislature or how it will be interpreted. In addition to California, other states have strengthened their data security laws and others have indicated their intention to do so as well. We expect that other states will follow with their own comprehensive data privacy legislation as well. Our activities must therefore comply with these other applicable privacy laws, which impose further restrictions on the access, use and disclosure of personal information. Further, we are required to comply with international personal data protection laws and regulations, including the European Union’s General Data Protection Regulation (“GDPR”). The GDPR is a prescriptive, detailed regulation that provides extensive powers to public authorities to sanction and stop use of personal data. While companies are afforded some flexibility in determining how to comply with the GDPR’s various requirements, the GDPR has and will continue to require significant effort and expense to ensure compliance. All of these laws may impact our business and may change periodically, which could adversely affect our business operations.
In Vitro Regulation
Under current law, in vitro diagnostics that the FDA regulates as medical devices must undergo premarket review prior to commercialization, unless the device is exempt from such review. The particular premarket requirements that must be met to market a medical device in the United States will depend on the classification of the device under FDA regulations. Medical devices are categorized into one of three classes, based on the degree of risk they present. Devices that pose the lowest risk are designated as Class I devices; devices that pose moderate risk are designated as Class II devices and are subject to general controls and special controls; and the devices that pose the highest risk are designated as Class III devices and are subject to general controls and premarket approval. As a CLIA-certified laboratory, we offer our testing solutions as LDTs, and we may seek to commercialize future testing services in development as LDTs. LDTs are generally defined as clinical laboratory tests that are developed and validated by a laboratory for its own use. The FDA historically has taken the position that it has the authority to regulate such tests as medical devices under the FDCA but has for the most part exercised enforcement discretion and has not required laboratories that offer LDTs to comply with the FDA requirements for medical devices, such as registration, device listing, quality systems regulations, premarket clearance or premarket approval, and post-market controls.
41
At various times since 2006, the FDA has issued documents outlining its intent to require varying levels of FDA oversight of many LDTs, including our tests. In October 2023, the FDA published a proposed rule that, if finalized, would regulate LDTs as medical devices under the FDCA. The comment period for the FDA’s proposed rule closed in December 2023 and the FDA has indicated its desire to issue a final rule on LDTs in April 2024 or shortly thereafter. Even if the FDA does not finalize its proposed rule, the U.S. Congress may enact statutory changes that could alter or eliminate the FDA’s current LDT enforcement policy. It is therefore unclear at this time what form that final regulation may take, or if the FDA will finalize the regulation at all. It is also unclear what additional obligations might be placed on us as we continue to offer LDTs in light of the FDA's renewed interest in greater oversight into this class of tests. Action by the FDA to phase out its current policy of enforcement discretion over LDTs may materially impact our development and commercialization of LDTs, including our current tests.
Until regulatory requirements suggested by the FDA or required by any new legislation are phased in, we do not consider our current LDTs to require FDA filings or clearance or approval before launch and we will continue to follow the CLIA certification and inspection pathway.
If the new requirements are phased in or if we elect to develop IVDs, our future offerings may require a 510(k) submission or a Premarket Approval (“PMA”) application to the FDA. In a 510(k) submission, the device sponsor must demonstrate that the new device is “substantially equivalent” to a predicate device in terms of intended use, technological characteristics, and performance testing. A 510(k) requires demonstration of substantial equivalence to another device that is legally marketed in the United States. Substantial equivalence means that the new device is at least as safe and effective as the predicate. A device is substantially equivalent if, in comparison to a predicate it (a) has the same intended use as the predicate and has the same technological characteristics as the predicate; or (b) has the same intended use as the predicate, has different technological characteristics, and the information submitted to the FDA does not raise new questions of safety and effectiveness, and is demonstrated to be at least as safe and effective as the legally marketed predicate device.
A claim of substantial equivalence does not mean the new and predicate devices must be identical. Substantial equivalence is established with respect to intended use, design, energy used or delivered, materials, chemical composition, manufacturing process, performance, safety, effectiveness, labeling, biocompatibility, standards, and other characteristics. A device may not be marketed in the United States until the submitter receives a letter declaring the device substantially equivalent. If the FDA determines that a device is not substantially equivalent, the applicant may resubmit another 510(k) with new data, or request a Class I or II designation through the FDA’s de novo process that allows a new device without a valid predicate to be classified into Class I or II if it meets certain criteria, or file a reclassification petition, or submit a PMA.
Manufacturers of medical devices must comply with various regulatory requirements under the FDCA and regulations thereunder, including, but not limited to, quality system regulations, unless they are exempt, facility registration, product listing, labeling requirements, and certain post-market surveillance requirements. Entities that fail to comply with FDA requirements can be liable for criminal or civil penalties, such as recalls, detentions, orders to cease manufacturing, and restrictions on labeling and promotion, among other potential sanctions.
The regulatory review and approval process for medical devices can be costly, timely, and uncertain. This process may involve, among other things, successfully completing additional clinical trials and submitting a premarket clearance notice or filing a premarket approval application with the FDA. If premarket review is required by the FDA, there can be no assurance that our tests will be cleared or approved on a timely basis, if at all. In addition, there can be no assurance that the labeling claims cleared or approved by the FDA will be consistent with our current claims or adequate to support continued adoption of and reimbursement for our products. Ongoing compliance with FDA regulations could increase the cost of conducting our business, subject us to FDA inspections and other regulatory actions, and potentially subject us to penalties in the event we fail to comply with such requirements.
Federal and State Fraud and Abuse Laws
False Claims and Overpayments
We are subject to numerous federal and state fraud and abuse laws, including the federal False Claims Act. Many of these fraud and abuse laws are broad in scope, and, at least with respect to the state laws, neither the courts nor relevant government agencies have extensively interpreted these laws. Prohibitions under some of these laws include:
| ● | the submission of false claims or false information to government programs, |
| ● | the retention of any overpayments received from governmental payors, |
| ● | deceptive or fraudulent conduct, |
| ● | excessive or unnecessary services or services at excessive prices, and |
| ● | defrauding commercial health insurers. |
42
Numerous federal and state agencies enforce these fraud and abuse laws. In addition, commercial insurers may also bring private actions. In some circumstances, private whistleblowers are authorized to bring lawsuits on behalf of the government against providers and are entitled to receive a portion of any final recovery.
In addition, amendments to the FCA impose severe penalties for the knowing and improper retention of overpayments collected from governmental payors. Within sixty days of identifying and quantifying an overpayment, a provider is required to notify CMS or the relevant MAC of the overpayment (and the reason for it) and to return the overpayment; failure to do so may result in a separate basis for liability under the FCA. These amendments could subject our procedures for identifying and processing payments to greater scrutiny. Overpayments may occur from time to time in the healthcare industry without any fraudulent intent. For example, overpayments may result from mistakes in reimbursement claim forms or from improper processing by governmental payors. We maintain protocols intended to identify any overpayments and to make timely refunds as appropriate. From time to time, we have identified overpayments and made appropriate refunds to government payors.
To avoid liability, we must carefully and accurately code claims for reimbursement, proactively monitor the accuracy and appropriateness of Medicare claims and payments received, diligently investigate any credible information indicating that we may have received an overpayment, and promptly return any overpayments.
Federal and State “Self-Referral” and “Anti-Kickback” Restrictions
Anti-Kickback Statute
The federal Anti-Kickback Statute prohibits persons from knowingly and willfully soliciting, receiving, offering, or paying remuneration, directly or indirectly, to induce either the referral of an individual, or the furnishing, recommending, or arranging for an item or service, for which payment may be made under a federal health care program, such as the Medicare and Medicaid programs, unless an exception or “safe harbor” applies. The term “remuneration” is not defined in the federal Anti-Kickback Statute and has been broadly interpreted to include anything of value, including for example, gifts, discounts, the furnishing of supplies or equipment, credit arrangements, payments of cash, waivers of payment, ownership interests and providing anything at less than its fair market value. Sanctions for violations of the federal Anti-Kickback Statute may include imprisonment and other criminal penalties, civil monetary penalties, and exclusion from participation in federal health care programs. Further, the Affordable Care Act made clear that claims for items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the federal civil FCA. Many states have also adopted laws similar to the federal Anti-Kickback Statute, some of which are not limited in application to only items or services reimbursable by federal health care programs, and do not contain identical safe harbors.
In addition, the Eliminating Kickbacks in Recovery Act of 2018 (“EKRA”) imposes criminal penalties for knowing or willful payment or offer, or solicitation or receipt, of any remuneration, whether directly or indirectly, overtly or covertly, in cash or in kind, in exchange for the referral or inducement of laboratory testing (among other healthcare services) unless a specific exception applies. Although it appears that EKRA was intended to reach patient brokering and similar arrangements to induce patronage of substance use recovery and treatment, the language in EKRA is broadly written and could apply to laboratory services covered under public or private payer arrangements.
Medicare Physician Self-Referral Law
The federal “self-referral” law, commonly referred to as the “Stark” law, provides that healthcare providers who, personally or through a family member, have ownership interests in or compensation arrangements with a laboratory are prohibited from making a referral to that laboratory for laboratory tests reimbursable by Medicare, and also prohibits laboratories from submitting a claim for Medicare payments for laboratory tests referred by healthcare providers who, personally or through a family member, have ownership interests in or compensation arrangements with the testing laboratory. The Stark law contains a number of specific exceptions which, if met, permit healthcare providers who have ownership or compensation arrangements with a testing laboratory to make referrals to that laboratory and permit the laboratory to submit claims for Medicare payments for laboratory tests performed pursuant to such referrals. We are subject to comparable state laws, some of which apply to all payers regardless of source of payment, and do not contain identical exceptions to the Stark law.
Compliance with federal fraud and abuse laws such as the Anti-Kickback Statute and the Stark law involve constant monitoring for regulatory changes, agency and court interpretations, and revisiting of arrangements based on new interpretations or clarifications, all of which will require ongoing compliance costs. In addition, these laws and their exceptions and safe harbors are complex and clear interpretations are not always available. Despite our best efforts to comply, we cannot guarantee that a government agency will necessarily agree with our interpretations or that one or more of our arrangements will not be subject to challenge, nor can we provide any assurance that they will not have an adverse effect on our business, financial condition, results of operations, and cash flows.
43
Sunshine Act
In 2010, Congress enacted the Physician Payments Sunshine Act (“Sunshine Act”), which aims to promote transparency around financial relationships between physicians, teaching hospitals and certain life sciences industry manufacturers. The Sunshine Act requires manufacturers of drugs, devices, biologicals, and medical supplies covered by Medicare, Medicaid, or the Children’s Health Insurance Program to report annually to CMS any payments or other transfers of value made to healthcare providers and teaching hospitals, unless an exception applies. Manufacturers must also disclose to CMS any healthcare provider ownership or investment interests. Some states have similar transparency laws. Although we do not consider our laboratory services to be covered devices under the Sunshine Act, the laws and regulations are continually evolving and in the future we could be required to comply with transparency requirements in the future, or we could otherwise be subject to scrutiny for the nature and amount of our payments and transfers of value in the healthcare industry.
International Regulatory Requirements and FCPA
When marketing our tests outside of the United States, we are subject to foreign regulatory requirements governing human clinical testing, export of tissue, marketing approval for our products, and performance and reporting of tests in each market. These requirements vary by jurisdiction, differ from those in the United States, and may require us to perform additional pre-clinical or clinical testing. In many countries outside of the United States, coverage, pricing, and reimbursement approvals are also required in order for our tests to be made available to patients in substantial volume.
Many countries in which we offer our tests have anti-kickback regulations prohibiting providers, as well as medical and in vitro diagnostic device manufacturers, from offering, paying, soliciting, or receiving remuneration, directly or indirectly, or providing a benefit to a healthcare professional in order to induce business that is reimbursable under any national healthcare program. In situations involving healthcare providers employed by public or state-funded institutions or national healthcare services, violation of the local anti-corruption or anti-gift laws may also constitute a violation of the U.S. Foreign Corrupt Practices Act (“FCPA”).
The FCPA prohibits any U.S. individual, business entity, or employee of a U.S. business entity from offering or providing, directly or through a third party, including the distributors we rely on in certain markets, anything of value to a foreign government official with corrupt intent to influence an award or continuation of business or to gain an unfair advantage, whether or not such conduct violates local laws. In addition, it is illegal for a company that reports to the SEC to have false or inaccurate books or records or to fail to maintain a system of internal accounting controls. We are also required to maintain accurate information and control over sales and distributors’ activities that may fall within the purview of the FCPA, its books and records provisions, and its anti-bribery provisions.
Other Laws
Occupational Safety and Health
In addition to its comprehensive regulation of health and safety in the workplace in general, the Occupational Safety and Health Administration has established extensive requirements aimed specifically at laboratories and other healthcare-related facilities. In addition, because our operations require employees to use certain hazardous chemicals, we also must comply with regulations on hazard communication and hazardous chemicals in laboratories. These regulations require us, among other things, to develop written programs and plans, which must address methods for preventing and mitigating employee exposure, the use of personal protective equipment, and training.
Specimen Transportation
Our commercialization activities subject us to regulations of the Department of Transportation, the U.S. Postal Service, and the Centers for Disease Control and Prevention that apply to the surface and air transportation of clinical laboratory specimens.
C. Organizational Structure
MDxHealth SA is a company with limited liability (naamloze vennootschap/société anonyme) incorporated and operating under the laws of Belgium.
44
The following chart shows our organizational structure as of December 31, 2023:
| Subsidiary Name | Jurisdiction of Organization | Ownership & Voting Interest Held by MDxHealth SA | ||
| MDxHealth, Inc. | Delaware | 100% (held directly) | ||
| MDxHealth BV | The Netherlands | 100% (held directly) | ||
| MDxHealth Servicelab BV | The Netherlands | 100% (held through MDxHealth BV) | ||
| MDxHealth Research BV | The Netherlands | 100% (held through MDxHealth BV) | ||
| Delta Laboratories LLC | Texas | 100% (held through MDxHealth, Inc.) |
D. Property, Plant and Equipment
We process our tests, and conduct research and development, at our 38,000 square foot U.S. headquarters and laboratory facility in Irvine, California pursuant to a lease that is currently scheduled to expire in 2026. This laboratory facility is certified pursuant to CLIA and accredited by CAP. We also maintain an 8,000 square foot laboratory facility in Plano, Texas pursuant to a lease currently scheduled to expire in May 2025. This laboratory facility is certified pursuant to CLIA.
Our headquarters, which we own, is located in the CAP Business Center, Herstal, Belgium. We maintain a 7,800 square foot office and a laboratory facility located in Nijmegen, The Netherlands, pursuant to a lease that is currently scheduled to expire in 2027, where we conduct research and development activities.
We believe that our existing facilities are adequate for our near-term needs, and we believe that suitable additional or alternative office and manufacturing space will be available as required in the future on commercially reasonable terms.
ITEM 4A. UNRESOLVED STAFF COMMENTS
Not applicable.
ITEM 5. OPERATING AND FINANCIAL REVIEW AND PROSPECTS
You should read the following discussion of our operating and financial review and prospects in conjunction with our audited consolidated financial statements and the related notes thereto included elsewhere in this annual report. In addition to historical information, the following discussion and analysis contains forward-looking statements that reflect our current plans, estimates, expectations and beliefs and involve risks and uncertainties. Our actual results and the timing of events could differ materially from those anticipated in the forward-looking statements as a result of various factors. Factors that could cause or contribute to these differences include, but are not limited to, those discussed below and elsewhere in this annual report, particularly in sections titled “Risk Factors” and “Special Note Regarding Forward-Looking Statements.”
Overview
We are a commercial-stage precision diagnostics company committed to providing non-invasive, clinically actionable and cost-effective urologic solutions to improve patient care. Our novel genomic testing solutions combine advanced clinical modeling with genomic data to provide each patient with a personalized risk profile, which provides more accurate and actionable information than traditional clinical risk factors used by clinicians. Our Select mdx and Confirm mdx tests address men at risk for undetected prostate cancer, providing physicians with a clear clinical pathway to accurately identify clinically significant prostate cancer while reducing the use of invasive procedures that are prone to complications. Our GPS test addresses men newly diagnosed with localized prostate cancer, providing physicians with a clear clinical pathway to make the most informed treatment decision for their individual disease. Our Resolve mdx test provides patients with recurrent, persistent and complicated UTIs with a test that is able to quickly deliver patient-specific antimicrobial treatment options for fast resolution and improved patient outcomes. Our team’s collective decades of experience in precision diagnostics and our portfolio of novel biomarkers for diagnostic, prognostic and predictive molecular assays supports our active pipeline of new testing solutions for urologic diseases.
We have experienced net losses and significant cash used in operating activities since inception in 2003. To date, our primary sources of capital have been public offerings of our ordinary shares and private placements, debt financing agreements, and revenue from the sale of our products. As of December 31, 2023, we had cash and cash equivalents of $22.4 million, long-term loans and borrowings of $35.6 million and an accumulated deficit of $331.4 million. During the years ended December 31, 2023, 2022 and 2021, we generated revenue of $70.2 million, $37.1 million, $22.2 million, respectively, with a net loss of $43.1 million, $44.0 million, $29.0 million, respectively.
In February 2023, the Company raised $40 million in gross proceeds by means of a public offering of 10,000,000 shares at an issue price of $4.00 per share through a public offering. In March 2023, the Company received additional gross proceeds of $3.0 from the underwriters’ exercise of their overallotment option.
Management expects the Company to continue to incur net losses and have significant cash outflows for at least the next twelve months. While these conditions, among others, could raise doubt about our ability to continue as a going concern, these consolidated financial statements have been prepared assuming that the Company will continue as a going concern. This basis of accounting contemplates the recovery of our assets and the satisfaction of liabilities in the normal course of business. A successful transition to attaining profitable operations is dependent upon achieving a level of positive cash flows adequate to support the Company’s cost structure.
45
Acquisition of Genomic Prostate Score® (GPS) test (formerly Oncotype DX GPS) from Exact Sciences
On August 2, 2022, we announced that we have entered into an agreement with Genomic Health, Inc., a subsidiary of Exact Sciences, to acquire the GPS test from Exact Sciences. We acquired GPS in order to expand our menu of tests targeted into urology and prostate cancer and in order to position the Company as one of the leaders in the urology and prostate cancer space with one of the most comprehensive menus of precision diagnostics.
Under the terms of the agreement, we acquired the GPS prostate cancer business of Exact Sciences for an aggregate purchase price of up to $100 million, of which an amount of $25 million was paid in cash and an amount of $5 million was settled through the delivery of 691,171 shares of the Company, at a price of $7.23 per share. Following the closing, which took place on August 2, 2022, an additional aggregate earnout amount of up to $70 million was to be paid by us to Exact Sciences over a three year period, commencing in 2024, in tranches equal to a portion of the annual revenues attributable to the GPS prostate cancer business for the preceding fiscal year; provided, in each instance, that such revenues exceed certain minimum revenue milestones for such fiscal year.
On August 23, 2023, we entered into an amendment to the asset purchase agreement with Exact Sciences, deferring the Company’s initial earnout payment by 3 years, from 2024 to 2027, in consideration for an amendment fee of $250,000 in cash and 250,000 of the Company’s shares, a 5-year subscription right (warrant) to acquire up to 1,000,000 of the Company’s shares at an exercise price of $5.265 per share, and an increase in the potential aggregate earnout amount from $70 million to $82.5 million. The Company agreed to convene a general shareholders’ meeting to approve the subscription right. Under the terms of the amended asset purchase agreement, we have agreed to make earnout payments to Exact Sciences in each of fiscal years 2025, 2026 and 2027, based upon certain revenues related to fiscal years 2024, 2025 and 2023, respectively. At our option, the earnout amounts can be settled in cash or through the issuance of additional shares of the Company (valued in function of a volume weighted average trading price of the Company’s shares at the end of the relevant earnout period) to Exact Sciences, provided that the aggregate number of shares held by Exact Sciences shall not exceed more than 7.5%.
Financial Operations Overview
Revenues and Other Income
Revenues
The majority of our revenue is derived from laboratory services with revenue recognized at a point in time when control of the services has transferred to the customer. This is generally when the test results are delivered to the customer. We derive a small amount of additional revenue from license fees, royalties and government grants.
A large portion of our revenues are either derived from Medicare, which has set a fixed price pursuant to a LCD for the Company’s Confirm mdx, Select mdx and GPS tests, or are established with reference to the Medicare fixed price level. Therefore, the revenue recognized from Medicare (as well certain contracted commercial insurance companies) for Confirm mdx, Select mdx and GPS is determined by reference to a fixed price pursuant to the respective LCD. For reimbursement claims made to other commercial insurance companies, where there is no certainty of the amount that will be paid for services rendered, the Company uses historical collection data — on an individual payor basis — to estimate its future collection and corresponding revenues that should be recognized for each of our testing solutions.
We analyze historical collection data on a monthly basis and make monthly adjustments to our estimates. In accordance with IFRS 15, revenue is recognized where such a variable consideration is included in the transaction price only to the extent that it is highly probable that the amount of revenue recognized will not be subject to significant future reversals as a result of subsequent re-estimation. When historical collection data is insufficient to estimate future collections, recognize revenue on a cash basis, meaning that revenues will not be recognized until actual cash payment is received from the payor.
Cost of Sales (exclusive of amortization of intangible assets)
Cost of sales includes the cost of materials, labor (including salaries, bonuses, and benefits), transportation, collection kits, and allocated overhead costs associated with processing samples. Allocated overhead costs include depreciation of laboratory equipment, facility occupancy and information technology costs. Costs associated with processing samples are expensed when incurred, regardless of the timing of revenue recognition. As such, cost of sales and related volume do not always trend in the same direction as revenue recognition.
Gross Profit and Gross Margin
We calculate gross profit as revenue less cost of sales, and gross margin as gross profit divided by revenue. Our gross margin has and will continue to be affected by a variety of factors, primarily average selling prices and ordering volumes. We expect our gross profit to increase in the foreseeable future as our revenue grows, our average selling price improves — based on broader commercial coverage for our tests — and as we take advantage of economies of scale as we grow test volume.
46
Operating Expenses
Research and Development Expenses
Research and development expenses consist of costs incurred for the development of our products. These expenses consist primarily of labor costs (including salaries, bonuses, benefits, and stock-based compensation), reagents and supplies, clinical studies, outside services, patent expenses, depreciation of laboratory equipment, facility occupancy and information technology costs. Research and development expenses also include costs associated with assay improvements and automation workflow for our current suite of products. We expense our research and development expenses in the period in which they are incurred, except for those development expenses that qualify for capitalization.
We expect that our research and development expenses will increase in absolute dollars as we continue to develop additional products, however, we expect that these expenses will decrease as a percentage of revenue over the long term, though they may fluctuate as a percentage from period to period due to the timing and extent of these expenses.
Selling and Marketing Expenses
Our selling and marketing expenses are expensed as incurred and include costs associated with our sales organization, including our direct clinical sales force and sales management, medical affairs, client services, marketing and managed care, as well as technical lab support and administration. These expenses consist primarily of labor costs (including salaries, bonuses, benefits, and stock-based compensation), customer education and promotional expenses, market analysis expenses, conference fees, travel expenses and allocated overhead costs.
While there are no immediate plans to grow the size of our commercial organization, our selling and marketing expenses may increase in absolute dollars as we increase our marketing activities to drive further awareness and adoption of our products. However, we expect that these expenses will decrease as a percentage of revenue over the long term, though they may fluctuate as a percentage from period to period due to the timing and extent of these expenses.
General and Administrative Expenses
Our general and administrative expenses include costs for certain executives, accounting and finance, legal, revenue cycle management, information technology, human resources, and administrative functions. These expenses consist primarily of labor costs (including salaries, bonuses, benefits, and stock-based compensation), professional service fees such as consulting, accounting, legal, general corporate costs, and public-company costs associated with our listing on the Nasdaq Capital Market, as well as allocated overhead costs (rent, utilities, insurance, etc.).
We expect that our general and administrative expenses will continue to increase in absolute dollars primarily due to increased headcount (some of it related to volume, such as revenue cycle management) and costs associated with operating as a public company, including expenses related to legal, accounting, regulatory, tax, maintaining compliance with our listing on the Nasdaq Capital Market and requirements of the SEC, director and officer insurance and investor relations.
Amortization of Intangible Assets
Amortization of intangible assets primarily relates to the acquired intellectual property, brand, and customer relationships of the GPS business combination, as detailed in Note 3 to the consolidated financial statements.
Other Operating (Expense) Income, net
Other operating (expense) income, net is comprised of fair-value adjustment of our loan facility, revaluation of the contingent consideration related to the acquisition of GPS and NovioGendix in 2015, and grant income. We do not expect the revaluation of the contingent consideration to be a significant source of income or expense going forward.
Financial Income/Expenses
Financial income/expense is comprised of fair-value adjustments related to the GPS contingent consideration, the NovioGendix contingent consideration, the Innovatus derivative instrument, and Exact Sciences’ 5-year warrant. In addition, financial expense also includes interest income/expense, debt extinguishment expenses, as well as foreign exchange gain/loss and other financial gain/loss. Interest income consists primarily of interest earned on our deposits. Our interest expense is primarily related to our current $35 million long-term debt facility with Innovatus Capital Partners, which replaced our previous €9 million debt facility with Kreos Capital VI (UK) Limited (“Kreos Capital”) in 2022.
Foreign exchange gains/losses are derived from our operating in two different currencies (Euro and U.S. dollar) for our European and U.S. operations. These gains and losses are not expected to be significant and we maintain reserves in both currencies to offset extreme fluctuations in the dollar/euro exchange rate.
Other financial loss is derived from accrued interest charges on the fair value of the NovioGendix contingent liability.
47
A. Operating results
Comparison of the years ended as of December 31, 2023 and 2022
Our results of operations for the years ended as of December 31, 2023 and 2022 are summarized in the tables below:
| Year Ended December 31, | Year-Over-Year Change | |||||||||||||||
| (in Thousands) | 2023 | 2022 | $ | % | ||||||||||||
| Services | $ | 69,965 | $ | 36,965 | $ | 33,000 | 89 | % | ||||||||
| Royalties and other revenues | 228 | 89 | 139 | 156 | ||||||||||||
| Revenues | 70,193 | 37,054 | 33,139 | 89 | ||||||||||||
| Cost of sales (exclusive of amortization of intangible assets) | (26,264 | ) | (17,835 | ) | (8,429 | ) | 47 | |||||||||
| Gross Profit | 43,929 | 19,219 | 24,710 | 129 | ||||||||||||
| Research and development expenses | (6,376 | ) | (5,497 | ) | (879 | ) | 16 | |||||||||
| Selling and marketing expenses | (36,915 | ) | (25,704 | ) | (11,211 | ) | 44 | |||||||||
| General and administrative expenses | (23,010 | ) | (23,308 | ) | 298 | (1 | ) | |||||||||
| Amortization of intangible assets | (4,494 | ) | (3,169 | ) | (1,325 | ) | 42 | |||||||||
| Other operating (expense) income, net | (461 | ) | 559 | (1,020 | ) | (182 | ) | |||||||||
| Operating loss | (27,327 | ) | (37,900 | ) | 10,573 | (28 | ) | |||||||||
| Financial income | 2,570 | 241 | 2,329 | 966 | ||||||||||||
| Financial expenses | (18,342 | ) | (6,385 | ) | (11,957 | ) | 187 | |||||||||
| Loss before income tax | (43,099 | ) | (44,044 | ) | 945 | (2 | ) | |||||||||
| Income tax | (1 | ) | — | (1 | ) | n/a | ||||||||||
| Loss for the year | (43,100 | ) | (44,044 | ) | 944 | (2 | ) | |||||||||
| Earnings per share attributable to parent (EPS) | ||||||||||||||||
| Basic and Diluted | (1.66 | ) | (2.78 | ) | 1.12 | (40 | ) | |||||||||
Revenue
Revenue increased $33.1 million, or 89% for the year ended December 31, 2023, compared to the year ended December 31, 2022, due to the acquisition of the GPS, an increase in our test volumes for Confirm mdx and Resolve mdx, as well as the initiation of Medicare reimbursement for Select mdx. Excluding revenues from the recently acquired GPS test, total revenue for 2023 was $39.3 million, an increase of 42% versus 2022.
2023 revenues were comprised of $24.8 million from Confirm mdx, $30.9 million from GPS, $9.7 million from Resolve mdx, with the remaining revenues from Select mdx and other. 2022 revenues were comprised of $21.8 million from Confirm mdx, $9.3 million from GPS, $4.9 million from Resolve mdx, with the remaining revenues from Select mdx and other.
Cost of Sales
Cost of sales increased $8.4 million, or 47% for the year ended December 31, 2023 compared to the year ended December 31, 2022, primarily as a result of the increase in sales and unit volumes, as well as product mix and the addition of GPS to our product menu.
Gross margins were 62.6% compared to 51.9% for the prior year, an improvement of 10.7 percentage points attributed to contribution from Select mdx reimbursement, cost management, expanded coverage across our menu, and a higher contribution from GPS to total revenue.
Amortization of intangible assets are excluded from cost of sales and are presented separately in the statement of profit and loss.
Research and Development Expenses
Research and development expenses increased $0.9 million, or 16% for the year ended December 31, 2023, compared to the year ended December 31, 2022, primarily due to annual compensation increases, as well as an increase in ongoing clinical studies, partially offset by savings in patent expenses, lab consumables, and external collaborator fees.
Selling and Marketing Expenses
Selling and marketing expenses increased $11.2 million, or 44%, for the year ended December 31, 2023, compared to the year ended December 31, 2022, primarily due to the additional field sales personnel associated with the GPS business as well as an increase in direct marketing expenses, travel expenses, facilities expenses, and depreciation offset by a decrease in outside professional fees.
48
General and Administrative Expenses
General and administrative expense decreased $0.3 million, or 1% for the year ended December 31, 2023, compared to the year ended December 31, 2022. Despite an increase in personnel costs of $1.2 million, there were decreases in public company expenses as well as a decrease in professional fees from the 2022 acquisition of GPS. Professional fees for 2023 included one-time expenses related to the transition of its sole listing on NASDAQ, the amended asset purchase agreement with Exact Sciences, and the capital increase in early 2023.
Amortization of Intangible Assets
Amortization of intangible assets primarily relates to the acquired intellectual property, brand, and customer relationships of the GPS business combination and was comprised of:
| Thousands of $ For the years ended December 31 |
2023 |
2022 |
2021 | |||||||||
| Research and development | 3,157 | 2,060 | 1,084 | |||||||||
| Selling and marketing | 1,315 | 878 | 292 | |||||||||
| General and administrative | 22 | 231 | 234 | |||||||||
| Total amortization of intangible assets | 4,494 | 3,169 | 1,610 | |||||||||
In 2023, the Company segregated “amortization of intangible assets” from other operating categories in the statement of profit or loss and is presenting amortization of intangible assets as a separate category. Prior periods balances have been reclassified to conform to current period presentation.
Other Operating (Expense) Income, net
Other operating (expense) income, net, decreased by $1.0 million, or 182% for the year ended December 31, 2023, compared to the year ended December 31, 2022, primarily due a negative fair value adjustment related to the GPS contingent consideration, partially offset by a positive fair-value adjustment related to the NovioGendix contingent liability.
Financial Income/Expense
Financial income increased 2.3 million, or 966% for the year ended December 31, 2023, compared to the year ended December 31, 2022, due to interest income from our cash deposits as well as the fair value of our option to pay the GPS earnout in shares of the company.
Financial expenses increased by $12.0 million, or 187% for the year ended December 31, 2023, compared to the year ended December 31, 2022, primarily due to increases in the fair-value adjustment for the GPS contingent consideration of $6.7 million, interest charges for our Innovatus debt facility of $3.6 million, and fair value adjustment of Exact Sciences’ 5-year warrant of $2.2 million.
Comparison of the years ended as of December 31, 2022 and 2021
Our results of operations for the years ended as of December 31, 2022 and 2021 are summarized in the tables below:
| Year Ended December 31, | Year-Over-Year Change | |||||||||||||||
| (in Thousands) | 2022 | 2021 | $ | % | ||||||||||||
| Services | $ | 36,965 | $ | 21,937 | $ | 15,028 | 69 | % | ||||||||
| Royalties and other revenues | 89 | 302 | (213 | ) | (71 | ) | ||||||||||
| Revenues | 37,054 | 22,239 | 14,815 | 67 | ||||||||||||
| Cost of sales (exclusive of amortization of intangible assets) | (17,835 | ) | (11,675 | ) | (6,160 | ) | 53 | |||||||||
| Gross Profit | 19,219 | 10,564 | 8,655 | 82 | ||||||||||||
| Research and development expenses | (5,497 | ) | (5,589 | ) | 92 | (2 | ) | |||||||||
| Selling and marketing expenses | (25,704 | ) | (17,452 | ) | (8,252 | ) | 47 | |||||||||
| General and administrative expenses | (23,308 | ) | (13,915 | ) | (9,393 | ) | 68 | |||||||||
| Amortization of intangible assets | (3,169 | ) | (1,610 | ) | (1,559 | ) | 97 | |||||||||
| Other operating income, net | 559 | 1,161 | (602 | ) | (52 | ) | ||||||||||
| Operating loss | (37,900 | ) | (26,841 | ) | (11,059 | ) | 41 | |||||||||
| Financial expense, net | (6,144 | ) | (2,161 | ) | (3,983 | ) | 184 | |||||||||
| Loss before income tax | (44,044 | ) | (29,002 | ) | (15,042 | ) | 52 | |||||||||
| Income tax | — | — | — | — | ||||||||||||
| Loss for the year | (44,044 | ) | (29,002 | ) | (15,042 | ) | 52 | |||||||||
| Earnings per share attributable to parent (EPS) | ||||||||||||||||
| Basic and Diluted, | (2.78 | ) | (2.38 | ) | (0.4 | ) | 17 | |||||||||
49
Revenue
Revenue increased $14.8 million, or 67% for the year ended December 31, 2022, compared to the year ended December 31, 2021, due to both the acquisition of the GPS test as well as an increase in our test volumes for Confirm mdx and Resolve mdx. Excluding revenues from the recently acquired GPS test, total revenue for 2022 was $27.7 million, an increase of 25% versus 2021.
For the year ended December 31, 2022 and 2021, Confirm mdx accounted for 59% and 91% of total revenue, respectively. 2022 revenues were comprised of $21.8 million from Confirm mdx, $9.3 million from GPS, $4.9 million from Resolve mdx, with the remaining revenues from Select mdx and other.
Cost of Sales (exclusive of amortization of intangible assets)
Cost of sales increased $6.1 million, or 53% for the year ended December 31, 2022 compared to the year ended December 31, 2021, primarily as a result of the increase in sales and unit volumes, however, gross margins increased from 47.5% for the year ended December 31, 2021 to 51.9% for the year ended December 31, 2021, a 4.4 percentage point improvement primarily related to our product mix and the addition of GPS to our product menu.
Research and Development Expenses
Research and development expenses decreased $0.1 million, or 2% for the year ended December 31, 2022, compared to the year ended December 31, 2021, primarily due to decreases in collaborator fees, patent expenses and clinical validation, partially offset by increased personnel costs.
Selling and Marketing Expenses
Selling and marketing expenses increased $8.3 million, or 47%, for the year ended December 31, 2022, compared to the year ended December 31, 2021, primarily due to the additional field sales personnel associated with the GPS business as well as an increase in marketing expense.
General and Administrative Expenses
General and administrative expense increased $9.4 million, or 68% for the year ended December 31, 2022, compared to the year ended December 31, 2021, primarily due to $3.7 million of one-time expenses related to the GPS acquisition (included in Professional fees), with the remaining $5.7 million increase primarily related to higher insurance, professional fees and public company expenses.
Other Operating Income, net
Other operating income, net, decreased by $0.6 million, or 52% for the year ended December 31, 2022, compared to the year ended December 31, 2021, primarily due to the reduction of grant income from the U.S. Department of Health and Human Services and the Dutch government NOW grants, partially offset by an increase in fair-value adjustment related to the acquisition of NovioGendix in 2015.
Financial Income/Expense
Financial expenses, net, increased by $4.0 million, or 184% for the year ended December 31, 2022, compared to the year ended December 31, 2021, primarily due to fair-value adjustment for the GPS contingent consideration of $2.4 million, interest charges and extinguishment expenses of $1.6 million for the loan facility with Kreos Capital, and interest charges of $1.6 million related to the Innovatus debt facility.
B. Liquidity and Capital Resources
We have incurred net losses in each quarter since our inception. For the years ended December 31, 2023, 2022, and 2021, we incurred net losses of $43.1 million, $44.0 million, and $29.0 million, respectively. We expect to continue to incur significant expenses for the foreseeable future and to incur operating losses in the near term while we make investments to support our anticipated growth.
As of December 31, 2023, we have been financed primarily through net proceeds of approximately $360 million from the sale of our equity securities and long-term debt facilities. Our primary source of cash from operations is cash receipts on accounts receivable from our revenue. As of December 31, 2023, we had cash and cash equivalents of $22.4 million and an accumulated deficit of $331.4 million.
On February 3, 2023, we announced the pricing of a registered public offering of 10,000,000 shares at a price to the public of $4.00 per share for total gross proceeds of $40.0 million before deducting commissions and estimated offering expenses.
50
On March 6, 2023, we announced that, in the context of the above-mentioned offering, the underwriters exercised the option to purchase additional shares, on the same terms and conditions as stated above, in the amount of 750,000 shares for gross proceeds of $3.0 million, bringing the aggregate gross proceeds from this transaction to $43.0 million.
Our primary uses of cash are to fund operating expenses, service debt and acquire equipment. Cash used to fund operating expenses excludes the impact of non-cash items such as depreciation and stock-based compensation and is impacted by the timing of when we pay our operating expenses as reflected in the change in our outstanding accounts payable and accrued expenses. Debt service primarily consists of interest payments on our outstanding debt. Acquisitions of property and equipment primarily consist of purchases of laboratory equipment.
On August 2, 2022, we entered into a $70 million loan and security agreement with Innovatus, which replaced the Company’s €9 million debt facility with Kreos Capital. At closing, an amount of $35 million was drawn, with an additional $35 million remaining available as a $20 million term B loan and a $15 million term C loan that can be drawn in 2024 and 2025 respectively, subject to certain conditions. The loans are secured by our assets including intellectual property rights. Remaining proceeds of the loans will be used for working capital purposes and to fund general business requirements.
The loans accrue interest at a floating per annum rate equal to the sum of (a) the greater of (i) the prime rate published in The Wall Street Journal in the “Money Rates” section or (ii) 4.00%, plus (b) 4.25%, and require interest-only payments for the initial four years. As contractually agreed, and at our election, a portion of the interest becomes payable in-kind by adding an amount equal to 2.25% of the outstanding principal amount to the then outstanding principal balance on a monthly basis until August 2, 2025. The loans mature on August 2, 2027. The lenders shall have the right to convert, prior to August 2, 2025, up to 15% of the outstanding principal amount of the loans into shares of the Company at a price per share equal to $11.21, reflecting a substantial premium to the trading price prior to the announcement of the acquisition. Amounts converted into shares of the Company will be reduced from the principal amount outstanding under the loan. Notable fees payable to Innovatus consist of a facility fee equal to 1% of the total loan commitment, due on the funding date of the relevant loans, and an end-of-loan fee equal to 5% of the amount drawn, payable upon final repayment of the relevant loans.
In April 2020, the Company, through our U.S. subsidiary, MDxHealth Inc., entered into a PPP loan with the SBA in the amount of $2,316,000 as part of the CARES Act. The loan has a term of five years and carries an interest rate of 1.0% per year. Payments on the loan are deferred for the first eighteen months following disbursement of the loan, with principal and interest payments beginning in the nineteenth month. Interest on the loan continues to accrue during the eighteen-month deferment period. Cash proceeds from the loan were received in July 2020. Of the $2.3 million PPP loan, $1.6 million, $1.0 million and $0.3 million has been recorded as long-term debt on the company’s consolidated balance sheet as of December 31, 2021, December 31, 2022, and December 31, 2023, respectively.
Funding Requirements
As of December 31, 2023, we had cash and cash equivalents of $22.4 million. Based on our current business plan, including the Company’s expected ability to access additional cash through debt, equity, or other means, we estimate that our current cash and cash equivalents and our anticipated cash flows generated from sales of our products, will be sufficient to meet our anticipated cash requirements over at least the next 12 months from the date of this annual report. This assessment is based on forecasts and projections within management’s most recent business plan as well as our expected ability to access additional cash through debt, equity or other means, for which at this moment a material uncertainty exists that casts substantial doubt on our ability to continue as a going concern. To meet our long-term financial needs, we may consider raising additional capital to expand our business, to pursue strategic investments, to take advantage of financing opportunities or for other reasons. As a result of our expected revenue growth, we expect our accounts receivable and inventory balances to increase. Any increase in accounts receivable and inventory may not fully cover corresponding increases in accounts payable and accrued expenses, which could result in greater working capital requirements. We have based these estimates on assumptions that may prove to be wrong, and we could utilize our available capital resources sooner than we expect.
Until such time, if ever, as we can generate revenue to support our cost structure, we expect to finance our operations through equity offerings or debt financings, or other capital resources, including potentially collaborations or licensing arrangements. The sale of equity and convertible debt securities may result in dilution to our shareholders and the terms of these securities could provide for rights, preferences or privileges senior to those of our common stock. The terms of debt securities issued or borrowings pursuant to a credit agreement could impose significant restrictions on our operations. If we raise funds through collaborations and licensing arrangements, we might be required to relinquish significant rights to our technologies or products or grant licenses on terms that are not favorable to us. Additional capital may not be available on reasonable terms, or at all.
Our ability to generate sufficient revenue to achieve profitability will be heavily dependent on the successful commercialization of our currently marketed products and our anticipated future products, as well as obtaining favorable reimbursement. We anticipate that a substantial portion of our capital resources and efforts in the foreseeable future will be focused on the commercialization of our existing products and the development of future products.
51
Our operating results may fluctuate significantly from period to period, depending on the timing of our planned development activities, clinical studies, and the growth of our sales and marketing activities. We expect our expenses will increase substantially for the foreseeable future as we:
| ● | attract, hire and retain qualified personnel; |
| ● | continue to develop additional solutions and generate any evidence required to support expanded reimbursement of our solutions; |
| ● | expand our sales force and territories and increase our marketing activities to drive further awareness and adoption of our solutions; |
| ● | protect and defend our intellectual property; |
| ● | invest in processes, infrastructure to support the growth of our business; and |
| ● | operate as a publicly listed company. |
The table below summarizes our cash flows information for the years ended December 31, 2023 and 2022.
| For the year ended December 31, | ||||||||
| (in Thousands) | 2023 | 2022 | ||||||
| Net cash used in operations | $ | (21,497 | ) | $ | (34,118 | ) | ||
| Net cash used in investing activities | (3,931 | ) | (29,038 | ) | ||||
| Net cash from financing activities | 32,280 | 20,716 | ||||||
| Effects of exchange rate changes | 25 | (555 | ) | |||||
| Change in cash and cash equivalents | $ | 6,877 | $ | (42,995 | ) | |||
Net cash used in operations was $21.5 million for year ended December 31, 2023, compared to $34.1 million for the year ended December 31, 2022. The decrease of cash used in operations of $12.6 million was primarily due to a lower operating loss of $10.6 million as well as a higher adjustment for non-cash related items such depreciation and amortization.
Net cash used in investing activities for the year ended December 31, 2023, was $3.9 million compared to $29.0 million for the year ended December 31, 2022. The decrease in net cash from investing activities primarily related to the acquisition of the GPS business which occurred during 2022.
Net cash from financing activities for year ended December 31, 2023, was $32.3 million compared to $20.7 million for the year ended December 31, 2022. Cash from financing activities for the year ended December 31, 2023, were primarily derived from net proceeds of $39.6 million from our registered public offering in March 2023. Cash from financing activities for the year ended December 31, 2022, were primarily derived from the $35.0 million debt facility from Innovatus, partially offset by the repayment of $10.8 million to Kreos.
Cash Flows
The table below summarizes our cash flows information for the years ended December 31, 2022 and 2021.
| For the year ended December 31, | ||||||||
| (in Thousands) | 2022 | 2021 | ||||||
| Net cash used in operations | $ | (34,118 | ) | $ | (22,548 | ) | ||
| Net cash used in investing activities | (29,038 | ) | (885 | ) | ||||
| Net cash from financing activities | 20,716 | 66,498 | ||||||
| Effects of exchange rate changes | (555 | ) | (520 | ) | ||||
| Change in cash and cash equivalents | $ | (42,995 | ) | $ | (42,545 | ) | ||
52
Net cash used in operations was $34.1 million for year ended December 31, 2022, compared to $22.5 million for the year ended December 31, 2021. The increase of cash used in operations of $11.6 million was primarily due to a higher operating loss as well as negative variation in our working capital.
Net cash used in investing activities for the year ended December 31, 2022, was $29.1 million compared to $0.9 million for the year ended December 31, 2021. The increase in net cash used in investing activities primarily related to the acquisition of the GPS business as well as to purchases of property, plant, and equipment during 2022.
Net cash from financing activities for year ended December 31, 2022, was $20.7 million compared to $66.5 million for the year ended December 31, 2021. Cash from financing activities for the year ended December 31, 2022 were primarily derived from the $35 million debt facility from Innovatus, partially offset by the repayment of $10.8 million to Kreos. Cash from financing activities for the year ended December 31, 2021 were primarily from an equity financing of €25.0 million ($30.4 million) in gross proceeds by means of a private placement as well as from an initial public offering in the United states of 3,750,000 shares for gross proceeds of $45.0 million.
Contractual Obligations and Commitments
Our principal obligations consist of a lease liability, financial debt and trade and other payables. The following table sets out, as of December 31, 2023, our contractual obligations and commitments due by period:
| Payments Due by Period | ||||||||||||||||||||
| (in thousands) | Total | Less than 1 Year | 1 – 3 Years | 4 – 5 Years | More than 5 Years | |||||||||||||||
| Loans and borrowings | $ | 36,207 | $ | 643 | $ | 35,564 | $ | 0 | $ | 0 | ||||||||||
| Lease liabilities | 5,058 | 1,480 | 3,476 | 102 | 0 | |||||||||||||||
| Total | $ | 41,265 | $ | 2,123 | $ | 39,040 | $ | 102 | $ | 0 | ||||||||||
The contractual obligations table does not include any additional potential contingent payments upon the future achievement by us of specified sales-based and other milestones, or royalty payments we may be required to make under license agreements we have entered into pursuant to which we have in-licensed certain intellectual property or under the agreement pursuant to which we acquired the GPS test. See “Collaboration and License Agreements” under Item 4B. “Business Overview” for additional information. The timing of when these additional payments will actually be made is uncertain and the payments are contingent upon the completion of future activities.
C. Research and development, patents and licenses
Our research and development teams utilize our deep expertise to contribute to the growth of our business. In the years ended December 31, 2023, 2022 and 2021, R&D expenses were $6.4 million, $5.5 million and $5.6 million, respectively, on research and development. For a discussion of our research and development activities, see Item 4B. “Business Overview” and Item 5A. “Operating Results.”
D. Trend information
Key Factors and Trends
Ability to Attract New Ordering Physicians and Increase Our Penetration with Existing Physicians
Revenue growth for our products will depend on our ability to continue to expand our base of ordering physicians, increase our penetration with existing physician customers, and increase the number of physicians who consistently order our tests. We do not have immediate plans to expand our direct sales force and believe that we have the ability to increase our base of ordering physicians with our current structure.
Reimbursement for Genomic Testing from Third-Party Payors
Successful commercialization of our tests depends, in large part, on the availability of coverage and adequate reimbursement from government and private payors. Favorable third-party payor coverage and reimbursement are essential to meeting the Company’s immediate objectives and long-term commercial goals. In the United States, for new diagnostic solutions, each private and government payor decides whether to cover the test, the amount it will reimburse for a covered test, and any the specific conditions for reimbursement. Providers may be unlikely to order a specific diagnostic test unless an applicable third-party payor offers meaningful reimbursement for the test. Therefore, adequate coverage and reimbursement is critical to the commercial success of a diagnostic product, and if the Company is unable to secure and maintain favorable coverage determinations and reimbursement levels, this will compromise its ability to earn revenues from its products.
53
Medicare
Reimbursement for diagnostic tests furnished to Medicare beneficiaries is typically based on a fee schedule set by CMS. As a Medicare-enrolled clinical services provider, the Company bills Noridian for its core tests offered from its California laboratory facility, and is subject to Noridian’s local coverage and reimbursement policies applicable to Medicare patients. Noridian participates in the MolDX program, which handles technical assessments for U.S. laboratories that perform molecular diagnostic testing. In 2014, 2015 and 2023, respectively, our Confirm mdx test, our GPS test and our Select mdx test received positive Medicare LCDs under the MolDX program, which provide coverage and reimbursement for Medicare beneficiaries throughout the United States. As a Medicare-enrolled provider with a secondary laboratory based in Texas, we bill Novitas, the MAC covering Texas, for tests that are offered by our Texas laboratory, including our Resolve mdx test, claims for which tests are therefore subject to Novitas’ local coverage and reimbursement policies. Our Resolve mdx UTI test is currently reimbursed by Novitas and most private insurance payors, based on nationally recognized CPT codes.
Commercial payors
Obtaining coverage and reimbursement by commercial payors is a time-consuming and costly process, without a guaranteed outcome, since each commercial payor makes its own decision with respect to whether to cover a particular test, and, if so, at what rate to reimburse providers for such test. In addition, several payors and other entities conduct technology assessments of new medical tests and devices and provide the results of these assessments for informational purposes to other parties. These assessments may be used by third-party payors and healthcare providers as grounds to deny coverage for a particular test, or to refuse to use or order a particular test or procedure. The Company’s tests have received initial negative technology assessments from several of these entities and are likely to receive more negative technology assessments. The Company continues to work with third-party payors to obtain coverage for its tests and to appeal denial decisions based on existing and ongoing studies, peer reviewed publications, and support from physician and patient groups. There are no assurances that commercial payors will continue to issue positive coverage and reimbursement policies and/or contracts, and, if issued, that such policies and/or contracts will be maintained in the future. If the Company’s tests are considered on a policy-wide level by major third-party payors, whether at the Company’s request or on their own initiative, and the tests are determined to be ineligible for coverage and reimbursement by such payors, the Company’s collection efforts and potential for revenue growth could be adversely impacted.
Increasing Market Acceptance and Adoption of our Tests
Healthcare providers typically take a long time to adopt new products, testing practices and clinical treatments, partly because of perceived liability risks and the uncertainty of third-party coverage and reimbursement. It is critical to the success of our sales efforts that we educate enough patients, clinicians and administrators about molecular diagnostics testing, in general, as well as about our testing solutions, and demonstrate our clinical benefits. It is likely that clinicians may not adopt, and third-party payors may not cover or adequately reimburse for, the Company’s tests unless they determine, based on published peer-reviewed journal articles and the experience of other clinicians, that they provide accurate, reliable and cost-effective information.
Menu Expansion
We intend to build on our leadership in the prostate cancer diagnostic space by expanding our existing menu of tests. We are currently developing a candidate test, Monitor mdx, for the prostate cancer diagnostic and treatment pathway. Monitor mdx is intended to function as a non-invasive solution that risk stratifies patients for continued AS versus intervention, while also improving patient compliance with AS protocols. Our pipeline products in active surveillance are still under development and may or may not make it to market, depending on results of our research and clinical studies. The completion of these research and development activities is difficult to predict, and the related expenses may vary significantly by quarter. We expect to increase our research and development expense during this time. We may also take advantage of our strong commercial channel into urology to introduce complimentary tests outside of our current pipeline products.
While these factors may present significant opportunities for us, they also pose significant risks and challenges that we must address. See Item 3D. “Risk Factors” for more information.
E. Critical Accounting Estimates
Critical Accounting Policies and Estimates
Refer to Note 2.4 to our consolidated financial statements found elsewhere in this annual report, for a discussion on the critical accounting policies, estimates, assumptions, and judgments that we believe to have the most significant impact on our consolidated financial statements.
54
ITEM 6. DIRECTORS, SENIOR MANAGEMENT AND EMPLOYEES
A. Directors and Senior Management
Our Board of Directors
The following table sets forth certain information relating to our Board of Directors as of March 31, 2024.
| Name | Age | Position(s) | Term | |||
| Koen Hoffman(1) | 55 | Independent Non-Executive Director (Chairperson of the Board of Directors) | Until 2024(2) | |||
| Michael K. McGarrity | 61 | Executive Director (Chief Executive Officer) | Until 2026 | |||
| Dr. Lieve Verplancke(3) | 64 | Independent Non-Executive Director | Until 2024(2) | |||
| Hilde Windels(4) | 58 | Independent Non-Executive Director | Until 2025 | |||
| Dr. Regine Slagmulder(5) | 57 | Independent Non-Executive Director | Until 2025 | |||
| Dr. Eric Bednarski | 52 | Non-Executive Director | Until 2025 | |||
| Donnie (Don) M. Hardison | 73 | Independent Non-Executive Director | Until 2025 |
| (1) | Acting through Ahok BV. |
| (2) | Director is up for reelection to a new mandate in May 2024 at our shareholder meeting. |
| (3) | Acting through Qaly-Co BV. |
| (4) | Acting through Hilde Windels BV. |
| (5) | Acting through Regine Slagmulder BV. |
Unless otherwise stated, the address for our directors is CAP Business Center, Zone Industrielle des Hauts-Sarts, Rue d’Abhooz 31, 4040 Herstal, Belgium.
Our Board of Directors has determined that five out of seven of the members of the Board are independent under Belgian law and the Nasdaq Stock Market listing requirements.
Except for our employment agreement with Michael K. McGarrity, our Executive Director and Chief Executive Officer, as described in “Item 7B. Related Party Transactions,” there are no service contracts between us and any of our directors providing for benefits upon termination of employment.
Board Diversity Matrix
| (as of April 30, 2024) | ||
| Country of Principal Executive Offices: | Belgium | |
| Foreign Private Issuer | Yes | |
| Disclosure Prohibited under Home Country Law | Yes | |
| Total Number of Directors | 7 | |
| Female | Male | Non-Binary | Did Not Disclose Gender | |||||||||||||
| Part I: Gender Identity | ||||||||||||||||
| Directors | 3 | 4 | ||||||||||||||
| Part II: Demographic Background | ||||||||||||||||
| Underrepresented Individual in Home Country Jurisdiction | ||||||||||||||||
| LGBTQ+ | ||||||||||||||||
| Did Not Disclose Demographic Background | 3 | 4 | ||||||||||||||
| (as of April 25, 2023) | ||
| Country of Principal Executive Offices: | Belgium | |
| Foreign Private Issuer | Yes | |
| Disclosure Prohibited under Home Country Law | Yes | |
| Total Number of Directors | 8 | |
| Female | Male | Non-Binary | Did Not Disclose Gender | |||||||||||||
| Part I: Gender Identity | ||||||||||||||||
| Directors | 3 | 5 | ||||||||||||||
| Part II: Demographic Background | ||||||||||||||||
| Underrepresented Individual in Home Country Jurisdiction | ||||||||||||||||
| LGBTQ+ | ||||||||||||||||
| Did Not Disclose Demographic Background | 3 | 5 | ||||||||||||||
55
The following sets forth the biographical information of the members of our board of directors:
Koen Hoffman, Chairperson of our Board of Directors, obtained a Master in Applied Economics and an MBA at Vlerick Business School. Between 1992 and July 2016, he worked at KBC Group, where he started his career in the corporate finance department and later became the CEO of KBC Securities in October 2012. Since August 2016, he is the CEO of Value Square asset management. Mr. Hoffman serves also as board member at Fagron NV (Chair), Greenyard (Chair), and SnowWorld.
Michael K. McGarrity, Chief Executive Officer and Executive Director of our Company, has more than 25 years of experience in the healthcare industry with a unique combination of device, diagnostics and biotechnology experience. Michael was most recently the CEO of Sterilis Medical from November 2017 to February 2019. Prior to Sterilis Michael was the CEO of Nanosphere (Nasdaq: NSPH), a nanotechnology-based molecular diagnostics company, where he engineered an operational and strategic turnaround that resulted in its successful sale to Luminex (Nasdaq: LMNX) in 2016. Prior to Nanosphere, McGarrity spent 13 years at Stryker Corporation (NYSE: SYK). Mr. McGarrity holds a BA from the University of Notre Dame.
Dr. Lieve Verplancke, MD, Independent Non-Executive Director of our Company, a Belgian national, began her career in 1984 with The Beecham Group (now part of GlaxoSmithKline), and has since held key management positions with Merck & Co., as well as Bristol-Myers Squibb, where she served as Managing Director, leading their Belgian/GDL subsidiary, until 2012. Ms. Verplancke also serves as a Board Member for Brussels-based Europe Hospitals; the Imelda Hospital in Bonheiden; and the Euronext fund, Quest for Growth and Materialise. She is also the Founder and Managing Director of Qaly@Beersel, an elderly care center in Belgium. In addition to being a medical doctor (MD–KULeuven University), Ms. Verplancke holds a postgraduate degree in Economics and an MBA from the University of Antwerp. She has also completed courses at INSEAD, CEDEP, Columbia University and the Vlerick Business School, and is a certified Executive Coach (PCC).
Hilde Windels, Independent Non-Executive Director of our Company, has over 20 years of experience in the biotechnology sector with a track record of building and structuring organizations, fundraising, M&A, public capital markets and corporate strategies. Currently, Mrs. Windels serves as a board member at PHAXIAM Therapeutics, GIMV NV, MCROPHYT SA, and Celyad. Previously, she served as CEO of immunodiagnostic company Antelope Dx BV, and as CEO ad interim and Deputy CEO of Biocartis from September 2015 until September 2017 and as CFO of Biocartis from 2011 until September 2015. Mrs. Windels has also worked as independent CFO for several private biotech companies and from 1999 to 2008 she was CFO of Devgen. In the past, she also served on the boards of Devgen, Biocartis, Ablynx, VIB and FlandersBio. Mrs. Windels holds a Masters in Economics (commercial engineer) from the University of Leuven, Belgium.
Dr. Regine Slagmulder, Independent Non-Executive Director of our Company, is a full professor in management accounting & control at Vlerick Business School,a visiting professor of accounting & control at INSEAD, and the Knowledge &Academic Director (a.i.) at Guberna, the Belgian Institute for Directors. Previously, she worked as a strategy practice consultant at McKinsey & Company. She also previously worked as a professor of management accounting at INSEAD and at the University of Tilburg. She serves as an independent director and member of the audit committee on the board of the investment company Quest for Growth (since 2011) and as an independent director and chair of the audit committee of Ekopak (since 2021), both listed on Euronext. Dr. Slagmulder graduated in civil electrotechnical engineering and industrial management from the University of Gent, after which she received a management doctorate at Vlerick Business School. As part of her research activities, she was a research fellow attached to INSEAD, Boston University (USA) and the P. Drucker Graduate Management Center at Claremont University (USA). She is an INSEAD certified director (IDP-C).
Dr. Eric Bednarski, Non-Independent Non-Executive Director of our Company, currently serves as a Partner of MVM Partners LLP. Before joining MVM in 2008, he was a Partner at Advent Healthcare Ventures and a Principal at Advent International Corporation. Prior to Advent, he was a Director in the Corporate Finance Group of Silicon Valley Bank. Dr. Bednarski has a B.S. degree in Neural Science from Brown University and a Ph.D. in Biological Sciences from the University of California, Irvine.
Donnie (Don) M. Hardison, Independent Non-Executive Director of our Company, currently is the sole proprietor of DMH Consulting, a management consulting firm that he founded and previously operated from April 2016 to January 2017. He was most recently the President and Chief Executive Officer, and served on the board of directors, of Biotheranostics, Inc., a molecular diagnostic company focused on oncology, from February 2017 until it was acquired by Hologic, Inc. in February 2021. From April 2010 to March 2016, Mr. Hardison was the President and Chief Executive Officer of Good Start Genetics, a molecular genetic testing and information company. For more than 20 years prior to that, Mr. Hardison held various executive and senior management positions at companies including Laboratory Corporation of America (LabCorp) a clinical laboratory company, Exact Sciences Corporation, a molecular diagnostics company, OnTarget, Inc., a sales and marketing consulting company, Quest Diagnostics Inc., a clinical laboratory company, SmithKline Beecham Corporation, a pharmaceutical company, and others. He served on the board of directors of Exact Sciences Corporation (Nasdaq: EXAS) from May 2000, through its initial public offering in February 2001, until August 2007. Mr. Hardison received his Bachelor of Arts degree, in political science, from the University of North Carolina, Chapel Hill.
56
Our Executive Management
The following table sets forth certain information relating to our executive management as of December 31, 2023.
| Name | Age | Position(s) | ||
| Michael K. McGarrity | 61 | Chief Executive Officer and Executive Director | ||
| John Bellano | 55 | Chief Commercial Officer | ||
| Ron Kalfus | 49 | Chief Financial Officer | ||
| Joseph Sollee | 59 | Executive Vice President of Corporate Development and General Counsel |
Unless otherwise stated, the address for our executive management is CAP Business Center, Zone Industrielle des Hauts-Sarts, Rue d’Abhooz 31, 4040 Herstal, Belgium.
The following is the biographical information of those members of our executive management who do not also serve on our Board of Directors:
John Bellano, our Chief Commercial Officer, joined MDxHealth in June 2019. He has more than 25 years of experience in the healthcare industry. Mr. Bellano started his career in pharmaceuticals and transitioned to molecular diagnostics where he has spent the past 20 years of his career, most recently as Chief Commercial Officer of Sterilis Solutions. Prior to Sterilis Solutions, he served as the commercial leader for pharmacogenomic companies Assurex Health and AltheaDx. Mr. Bellano holds a degree from Allentown College.
Ron Kalfus, our Chief Financial Officer, joined MDxHealth in July 2019. He has over 20 years of leadership experience in both public and private companies within diagnostics/biotech and other sectors, and brings extensive knowledge in financial operations and management. Mr. Kalfus joined MDxHealth from Rosetta Genomics, where he helped lead efforts to reposition the company for commercial success with its oncology diagnostic products. Prior to Rosetta, Mr. Kalfus served as the CFO and Treasurer of MabCure, a Belgium-based publicly-traded biotechnology start up in the field of early cancer detection using antibodies. Mr. Kalfus holds a MS in Accounting from Fairleigh Dickinson University and a BBA in Finance from the University of Georgia and is a CPA licensed in New Jersey.
Joseph Sollee, our Executive Vice President of Corporate Development and General Counsel, has provided legal counsel to MDxHealth since its inception in 2003, and in April 2008 joined our management team. Prior to joining the Company, Mr. Sollee served as Special Counsel with the law firm of Kennedy Covington (now K&L Gates), where he led the Life Sciences Practice Group. Mr. Sollee has more than 20 years of experience in the life sciences industry, and has held senior legal and management positions at Triangle Pharmaceuticals and TherapyEdge. In addition, he has practiced as a corporate attorney in the Washington D.C. legal firm Swidler & Berlin and in investment banking at Smith Barney in New York. Mr. Sollee received a Juris Doctorate in Law (JD) and a Master’s degree in International & Comparative Law (LLM) from Duke University, a BA degree from Harvard University, and has been certified into the legal bars of New York, Washington D.C. and North Carolina.
Family Relationships
There are no family relationships among any of the members of our executive management and/or our Board of Directors.
B. Compensation
Compensation of Our Directors and Executive Management
Our current remuneration policy is based on meritocracy and a sense of ownership and is designed to reward performance in order to motivate members of the Board of Directors and the executive management of the Company in order to deliver increased shareholder value through superior business results. Since de-listing from Euronext Brussels in December 2023, the Company is no longer required to have a remuneration policy within the sense of the Belgian Companies and Associations Code.
57
Compensation of Our Board of Directors
Upon recommendation and proposal of the Nomination and Remuneration Committee, our Board of Directors determines the remuneration of the directors to be proposed to the general shareholders’ meeting. Pursuant to Belgian law, the general shareholders’ meeting approves the remuneration of the directors. The level and structure of the remuneration of the members of the Board of Directors are determined based on their general and specific responsibilities and market practice. Our shareholders approved the following annual remuneration and compensation of the members of the Board of Directors:
| Compensation | ||||||||
| (in EUR) | (in USD) | |||||||
| Chairman – Non-Executive Director | 59,500 | 64,300 | ||||||
| Non-Executive Director (including Independent Directors) | 35,000 | 37,800 | ||||||
| Additional fee for the Chair of the Audit Committee | 17,500 | 18,900 | ||||||
| Additional fee for a Member of the Audit Committee (other than the Chair of the Audit Committee) | 9,000 | 9,700 | ||||||
| Additional fee for the Chair of the Nomination and Remuneration Committee | 17,500 | 18,900 | ||||||
| Additional fee for a Member of the Nomination and Remuneration Committee Member (other than the Chair of the Nomination and Remuneration Committee) | 5,500 | 5,900 | ||||||
The abovementioned remuneration can be reduced pro rata temporis depending on the duration of the mandate, chairpersonship or membership of a director during a given year. The abovementioned amounts are exclusive of VAT and similar charges.
Directors are not entitled to any kind of performance cash bonus or other kind of variable remuneration. Mr. McGarrity, our Chief Executive Officer, Executive Director and a member of our Board of Directors, does not receive any compensation for his service as a director. Additionally, directors are not entitled to any kind of compensation when their mandate ends. For 2023, the following remuneration or compensation was due to the directors (excluding Mr. McGarrity):
| Compensation | ||||||||
| (in EUR) | (in USD) | |||||||
| Koen Hoffman(1) | 100,000 | 108,100 | ||||||
| Jan Pensaert(2) | — | — | ||||||
| Dr. Lieve Verplancke(3) | 49,500 | 53,500 | ||||||
| Hilde Windels(4) | 44,000 | 47,600 | ||||||
| Dr. Regine Slagmulder(5) | 52,500 | 56,800 | ||||||
| Dr. Eric Bednarski | — | — | ||||||
| Donnie (Don) M. Hardison | 52,500 | 56,800 | ||||||
| (1) | Acting through Ahok BV. |
| (2) | Acting through Valiance Advisors LLP. Mr. Pensaert resigned from the Board of Directors on February 15, 2024. |
| (3) | Acting through Qaly-Co BV. |
| (4) | Acting through Hilde Windels BV. |
| (5) | Acting through Regine Slagmulder BV. |
58
The table below provides an overview as of December 31, 2023, of the subscription rights, or options, held by the non-executive directors.
| Options | ||||||||||
| Name | Number
of Ordinary Shares Underlying the Options | Option Exercise Price per Ordinary Share Underlying the Options (in EUR) | Option Expiration Date | |||||||
| Koen Hoffman(1) | 1,000 2,000 | 49.70 12.80 | June 18, 2027 June 20, 2029 | |||||||
| Jan Pensaert(2) | 400 1,000 1,000 1,000 1,000 1,000 1,000 1,000 1,000 1,000 | 41.30 49.10 41.30 49.70 49.70 12.80 12.80 13.75 7.97 3.00 | June 22, 2024 June 22, 2024 June 22, 2024 June 18, 2027 June 18, 2027 June 20, 2029 June 20, 2029 May 26, 2031 May 25, 2032 July 4, 2033 | |||||||
| Dr. Lieve Verplancke(3) | 1,000 1,000 1,000 | 49.70 49.70 12.80 | June 18, 2027 June 18, 2027 June 20, 2029 | |||||||
| Hilde Windels(4) | 1,000 1,000 | 49.70 12.80 | June 18, 2027 June 20, 2029 | |||||||
| Dr. Regine Slagmulder(5) | None | |||||||||
| Dr. Eric Bednarski | None | |||||||||
| Donnie (Don) M. Hardison | None | |||||||||
| (1) | Acting through Ahok BV. |
| (2) | Acting through Valiance Advisors LLP. Mr. Pensaert resigned from the Board of Directors on February 15, 2024. |
| (3) | Acting through Qaly-Co BV. |
| (4) | Acting through Hilde Windels BV. |
| (5) | Acting through Regine Slagmulder BV. |
Compensation of Our Executive Management
The remuneration of our executive management is determined by our Board of Directors. The remuneration of the Chief Executive Officer and the other members of our executive management is based on recommendations made by our Nomination and Remuneration Committee. The Chief Executive Officer can, and will in principle be invited to, participate in an advisory capacity at the meetings of the committee when it deals with the remuneration of other executive managers.
59
Each member of the executive management is entitled to a basic fixed remuneration designed to fit responsibilities, relevant experience and competences, in line with market rates for equivalent positions. The majority of the annual remuneration is a fixed compensation amount. There is no minimum or maximum variable bonus. The Chief Executive Officer has a fixed remuneration, a fixed bonus and variable bonus linked to the performance of the Company and to his capacity to manage remuneration costs. The other management team members receive a fixed remuneration plus a variable bonus that is linked to their personal achievements (i.e., experience, know-how, education, skills, responsibilities, and performance) and the achievements of the Company. The remuneration is closely linked to performance. Bonuses, if any, are linked to identifiable objectives and to special projects and are set and measured on a calendar-year basis. Non-performers are not retained in the Company. The performance objectives of the management team members are primarily evaluated with regard to the following criteria: (i) respect of the board-approved annual budget, and (ii) meeting measurable operational targets. The various objectives and their weighting may differ for the individual managers.
The Nomination and Remuneration Committee of the Board of Directors meets annually to review the performance of the executive managers, to compare the actual measurable results to the objectives that were pre-defined by the committee, and to establish the measurable objectives for the ensuing calendar year.
Each member of the executive management is in principle entitled to receive share options or subscription rights. Each member of the executive management who is a salaried employee may be entitled to a number of fringe benefits, which may include participating in a defined contribution pension or retirement scheme, disability insurance, a company car, a mobile telephone, internet access and/or a laptop computer according to general Company policy, and other collective benefits (such as hospitalization insurance and meal vouchers). Executive members who are engaged on the basis of a services contract do not receive fringe benefits, except that they may be provided with a mobile phone and laptop computer according to general Company policy, and they qualify for reimbursement of expenses incurred while carrying out their professional responsibilities. Executive managers of the Company that are employed under employee contracts are entitled to enroll in defined-contribution type pension plans (such as 401(k) plans in the United States). Executive managers of the Company that are engaged on the basis of a service agreement are not entitled to any pension plans or pension plan contributions from the Company. Furthermore, the Company has entered into indemnification arrangements with the members of the executive management and has implemented directors’ and officers’ insurance coverage in order to cover liability they may incur in the exercise of their mandates.
Mr. McGarrity is remunerated on the basis of his executive management position. As CEO, Mr. McGarrity is entitled to a gross annual base salary of $535,000, which will be reviewed by the Board of Directors (or the Nomination and Remuneration Committee) on an annual basis, and an annual bonus of up to 50% of the then applicable base salary. Furthermore, Mr. McGarrity is entitled to the reimbursement of expenses, and he and his dependents are eligible to participate in all group health, medical, dental, disability and insurance plans, incentive, savings and retirement plans, and other employee benefits that are established by the Company for its executives.
Excluding the value of subscription rights (employee share options), the remuneration and benefits provided to Mr. McGarrity in 2023 were composed of the following:
| Compensation | ||||||||
| (in EUR) | (in USD) | |||||||
| Fixed gross remuneration(1) | 494,789 | 535,015 | ||||||
| Supplementary paid compensation(2) (gross) | 148,432 | 160,500 | ||||||
| Pension benefits | 12,208 | 13,200 | ||||||
| Other benefits(3) | 53,839 | 58,216 | ||||||
| Total | 709,268 | 766,931 | ||||||
| (1) | Total cost to the Company, including employer social security contributions and vacation pay accrual. |
| (2) | Excludes value of 100,000 subscription rights (employee share options) already created, issued, and accepted in 2023 under the Company’s 2023 Share Option Plan. |
| (3) | Includes Company-paid and other similar benefits, such as the employer’s payroll taxes, meal tickets and health insurances. Excludes reimbursement of normal professional expenses such as telephone and Company travel expenses. |
The 2023 combined remuneration package of the other executive management team members in office in 2023 (excluding the CEO)—i.e., John Bellano, Joseph Sollee and Ron Kalfus—including employer taxes, was $1,464,149 composed of the following:
| Compensation | ||||||||
| (in EUR) | (in USD) | |||||||
| Fixed gross remuneration(1) | 994,195 | 1,075,023 | ||||||
| Bonuses paid and awarded (2) (gross) | 181,957 | 196,750 | ||||||
| Pension benefits | 32,139 | 34,752 | ||||||
| Other benefits(3) | 145,773 | 157,624 | ||||||
| Total | 1,354,064 | 1,464,149 | ||||||
| (1) | Includes employer taxes and vacation pay accrual. Excludes VAT. |
60
| (2) | Excludes value of subscription rights (employee share options) already created, issued, and accepted in 2023 by certain other executive managers under the Company’s 2023 Share Option Plan. |
| (3) | Includes for some individuals a Company car, meal vouchers, and other similar benefits. Excludes reimbursement of normal professional expenses such as telephone and Company travel expenses. |
The total remuneration and benefits paid to the executive management team members (including the Chief Executive Officer) in 2023 and 2022 was €2,063,332 ($2,231,081) and €1,729,842 ($1,822,389) (gross amount, excluding VAT and share based compensation), respectively. In the aforementioned figures, the service fees of the managers hired on the basis of a service agreement are included with the salaries of the other management team members.
The primary performance objectives for the bonuses of the above management team members in 2023 were the following:
| ● | respect of the board-approved annual budget, with a focus on cash-flow management; and | |
| ● | meeting measurable operational targets, including total revenues and margins for the Company’s testing solutions and achievement of certain strategic and investor relations metrics. |
Each of these foregoing targets was based on minimum percentage attainment of defined, objective outcomes for the full calendar year, with the revenue, margin and cash flow targets tied to items that are reviewed by the Company’s independent auditors in the ordinary course. Additionally, each performance target was assigned a pre-defined weighting as a percentage of the bonus eligibility applicable to each member of executive management, with the CEO being treated consistently with other members of the executive management team.
The table below provides an overview as of December 31, 2023, of the subscription rights (employee share options) held by the members of the executive management team. The options with an expiration date of June 30, 2033 were granted in 2023.
| Options | ||||||||||
| Name | Number of Ordinary Shares Underlying the Options | Option Exercise Price per Ordinary Share Underlying the Options (in EUR) | Option Expiration Date | |||||||
| Michael K. McGarrity | 150,000 45,000 100,000 100,000 100,000 | 14.90 8.00 13.75 6.84 2.90 | June 18, 2027 June 20, 2029 May 26, 2031 May 25, 2032 June 30, 2033 | |||||||
| Ron Kalfus | 20,000 34,700 40,000 40,000 40,000 | 12.40 8.00 13.75 6.84 2.90 | June 20, 2029 June 20, 2029 May 26, 2031 May 25, 2032 June 30, 2033 | |||||||
| Joseph Sollee | 4,000 4,000 4,000 23,000 9,800 35,000 40,000 40,000 | 44.90 37.80 53.50 12.40 8.00 13.75 6.84 2.90 | June 22, 2024 June 22, 2024 June 22, 2024 June 20, 2029 June 20, 2029 May 26, 2031 May 25, 2032 June 30, 2033 | |||||||
| John Bellano | 40,000 28,800 45,000 40,000 40,000 | 12.40 8.00 13.75 6.84 2.90 | June 20, 2029 June 20, 2029 May 26, 2031 May 25, 2032 June 30, 2033 | |||||||
61
C. Board Practices
The following table sets forth certain information relating to our Board of Directors as of December 31, 2023.
| Name | Age | Position(s) | Year of Initial Appointment | Current Term Expiration Year | |||||||||
| Koen Hoffman(1) | 55 | Independent Non-Executive Director (Chairperson of the Board of Directors) | 2018 | 2024(2) | |||||||||
| Michael K. McGarrity | 61 | Executive Director (Chief Executive Officer) | 2019 | 2026 | |||||||||
| Jan Pensaert(3) | 51 | Non-Executive Director | 2014 | 2024 | |||||||||
| Dr. Lieve Verplancke(4) | 64 | Independent Non-Executive Director | 2017 | 2024(2) | |||||||||
| Hilde Windels(5) | 58 | Independent Non-Executive Director | 2017 | 2025 | |||||||||
| Dr. Regine Slagmulder(6) | 57 | Independent Non-Executive Director | 2020 | 2025 | |||||||||
| Dr. Eric Bednarski | 52 | Non-Executive Director | 2020 | 2025 | |||||||||
| Donnie (Don) M. Hardison | 73 | Independent Non-Executive Director | 2021 | 2025 | |||||||||
| (1) | Acting through Ahok BV. |
| (2) | Director is up for reelection to a new mandate in May 2024 at our shareholder meeting. |
| (3) | Acting through Valiance Advisors LLP. Mr. Pensaert resigned from the Board of Directors on February 15, 2024. |
| (4) | Acting through Qaly-Co BV. |
| (5) | Acting through Hilde Windels BV. |
| (6) | Acting through Regine Slagmulder BV. |
Unless otherwise stated, the address for our directors is CAP Business Center, Zone Industrielle des Hauts-Sarts, Rue d’Abhooz 31, 4040 Herstal, Belgium.
Committees of our Board of Directors
Our Board of Directors is assisted by a number of specialized committees in order to advise the board in respect of decisions to be taken, to give comfort to the Board of Directors that certain issues have been adequately addressed and, if necessary, to bring specific issues to the attention of the Board of Directors. The decision-making remains the collegial responsibility of the Board of Directors.
Our Board of Directors has established, in its midst and under its responsibility, two board committees which are responsible for assisting the Board of Directors and making recommendations in specific fields: an Audit Committee and a Nomination and Remuneration Committee. The terms of reference of these board committees are primarily set out in the Corporate Governance Charter of the Company.
Audit Committee
As of the date of this annual report, our Audit Committee consists of three directors: Regine Slagmulder, Lieve Verplancke, and Hilde Windels. Our Board of Directors has determined that all members of our Audit Committee are independent under Rule 10A-3 of the Exchange Act and the applicable listing standards of Nasdaq.
The members of the Audit Committee must have a collective expertise relating to the activities of the Company, and at least one member of the Audit Committee must have the necessary competence in accounting and auditing, including qualifying as an “audit committee financial expert” as defined under the Exchange Act. Our Board of Directors has determined that the members of the Audit Committee satisfy the competency requirement, and our Board of Directors has further determined that Regine Slagmulder and Hilde Windels each qualifies as an “audit committee financial expert” as defined under the Exchange Act.
The Audit Committee is governed by a charter that complies with Nasdaq listing rules to the extent applicable to the Company. The members of the Audit Committee shall have unrestricted access to the offices and all information and papers kept by the company and its subsidiaries. Each member of the Audit Committee may ask the executive management or any other staff member of the Company or its subsidiaries to submit the information that he or she deems useful, appropriate or necessary to perform his or her tasks within the framework of the Audit Committee.
Nomination and Remuneration Committee
As of the date of this annual report, our Nomination and Remuneration Committee consists of four directors: Eric Bednarski, Don Hardison, Koen Hoffman, and Lieve Verplancke. Our Board of Directors has determined that three members of our Nomination and Remuneration Committee are independent under the applicable listing standards of Nasdaq.
62
The role of the Nomination and Remuneration Committee is to make recommendations to the Board of Directors with regard to the appointment and remuneration of directors and members of the executive management and, in particular, to:
| ● | identify, recommend and nominate, for the approval of the Board of Directors, candidates to fill vacancies in the Board of Directors and executive management positions as they arise. In this respect, the Nomination and Remuneration Committee must consider and advise on proposals made by relevant parties, including management and shareholders; |
| ● | advise the Board of Directors on any proposal for the appointment of the Chief Executive Officer and on the Chief Executive Officer’s proposals for the appointment of other members of the executive management; |
| ● | draft appointment procedures for members of the Board of Directors and the Chief Executive Officer; |
| ● | ensure that the appointment and re-election process is organized objectively and professionally; |
| ● | periodically assess the size and composition of the Board of Directors and make recommendations to the Board of Directors with regard to any changes; and |
| ● | consider issues related to succession planning; |
D. Employees
As of December 31, 2023, we had 300 employees, all of whom are employed on a full-time basis. Of these employees, 287 are located in the United States and 13 in Europe. None of our employees is covered by a collective bargaining agreement, and we believe our relationship with our employees is good.
Our human capital resources objectives include, as applicable, identifying, recruiting, retaining, incentivizing and integrating our existing and new employees, advisors and consultants. The principal purposes of our equity incentive plans are to attract, retain and reward personnel through the granting of stock-based compensation awards, in order to increase stockholder value and the success of our company by motivating such individuals to perform to the best of their abilities and achieve our objectives.
E. Share Ownership
For information regarding the share ownership of our supervisory and executive board members, see Item 6B. “Compensation” and Item 7A. “Major Shareholders.”
F. Disclosure of a registrant’s action to recover erroneously awarded compensation
Not applicable.
ITEM 7. MAJOR SHAREHOLDERS AND RELATED PARTY TRANSACTIONS
A. Major Shareholders
Except as specifically noted, the following table sets forth information with respect to the beneficial ownership of our ordinary shares as of March 31, 2024:
| ● | each of our directors and executive officers; |
| ● | each person known to us to beneficially own more than 3% of our ordinary shares; and |
| ● | all of our supervisory board members and executive board members as a group. |
As of March 31, 2024, there were four record holders of our ordinary shares, two of whom were residents of the United States.
All of the ordinary shares have the same voting rights and no major shareholders of the Company have different voting rights.
Beneficial ownership is determined according to the rules of the SEC and generally means that a person has beneficial ownership of a security if he, she or it possesses sole or shared voting or investment power of that security, including ordinary shares that can be acquired within 60 days of March 31, 2024. Ordinary shares subject to derivative securities currently exercisable or exercisable within 60 days of March 31, 2024 are deemed to be outstanding for computing the percentage ownership of the person holding these securities and the percentage ownership of any group of which the holder is a member, but are not deemed outstanding for computing the percentage of any other person.
The percentage ownership information shown in the table is based upon 27,288,093 ordinary shares outstanding as of March 31, 2024.
63
Except as indicated by the footnotes below, we believe, based on the information furnished to us, that the persons named in the table below have sole voting and investment power with respect to all ordinary shares shown that they beneficially own, subject to community property laws where applicable. The information does not necessarily indicate beneficial ownership for any other purpose, including for purposes of Sections 13(d) and 13(g) of the Exchange Act.
Except as otherwise indicated in the table below, addresses of the directors, members of the executive management team and named beneficial owners are in care of MDxHealth SA, CAP Business Center, Zone Industrielle des Hauts-Sarts, Rue d’Abhooz 31, 4040 Herstal, Belgium.
| Name of beneficial owner | Number of Ordinary Shares Beneficially Owned | Percentage of Ordinary Shares Beneficially Owned | ||||||
| 3% or Greater Shareholders: | ||||||||
| MVM Partners LLP(1) | 4,550,458 | 16.7 | % | |||||
| Bleichroeder LP(2) | 3,878,334 | 14.2 | % | |||||
| Valiance Asset Management Limited(3) | 2,093,108 | 7.7 | % | |||||
| Biovest NV(4) | 1,192,358 | 4.4 | % | |||||
| Genomic Health, Inc.(5) | 941,171 | 3.4 | ||||||
| Executive Officers and Directors: | ||||||||
| Michael K. McGarrity(6) | 357,707 | 1.3 | ||||||
| Ron Kalfus(6) | 117,694 | * | ||||||
| Joseph Sollee(6) | 117,515 | * | ||||||
| John Bellano(6) | 136,699 | * | ||||||
| Koen Hoffman (acting through Ahok BV)(6) | 3,000 | * | ||||||
| Dr. Lieve Verplancke (acting through Qaly-Co BV)(6) | 3,000 | |||||||
| Hilde Windels (acting through Hilde Windels BV)(6) | 2,000 | * | ||||||
| Dr. Regine Slagmulder (acting through Regine Slagmulder BV) | 59,020 | * | ||||||
| Dr. Eric Bednarski | — | * | ||||||
| Donnie (Don) M. Hardison | — | * | ||||||
| All current directors and executive management as a group (10 persons) | 796,635 | 2.8 | % | |||||
| * | Less than one percent. |
| (1) | Includes 4,458,529 shares held by MVM V LP and 91,928 shares held by MVM GP (No. 5) (collectively, the “MVM Entities”). The Company has been informed that voting and investment power over the shares held by the MVM Entities is exercised jointly by three or more natural persons and voting and disposition decisions require the approval of a majority of such persons. Accordingly, no single natural person has voting or dispositive power over such shares. The MVM Entities are party to the MVM Subscription Agreement, providing for certain rights to appoint a member of our Board of Directors. See Item 7B. “Related party transactions — MVM Subscription Agreement.” This information has been obtained from Schedule 13D/A filed by the MVM Entities with the SEC on February 28, 2023. |
| (2) | Includes 2,034,216 shares held by 21 April Fund Ltd., a Cayman Islands company for which Bleichroeder LP acts as investment adviser. This information has been obtained from Schedule 13G/A filed by Bleichroeder Entities with the SEC on February 14, 2024. |
| (3) | Includes 1,233,929 shares held by TopMDx Ltd. and 859,179 shares held by Valiance Life Sciences Growth Investments SICAV-SIF (collectively, the “Valiance Entities”). The Company has been informed that voting and investment power over the shares held by the Valiance Entities is exercised jointly by three or more natural persons and voting and disposition decisions require the approval of a majority of such persons. Accordingly, no single natural person has voting or dispositive power over such shares. This information has been obtained from Schedule 13D/A filed by the Valiance Entities with the SEC on April 14, 2023. |
| (4) | Consists of shares held by Biovest NV (“Biovest”). RMM, S.A. (“RMM” and together with Biovest, the “Biovest Entities”) is the sole owner of Biovest and pursuant to an understanding with Biovest, decisions relating to the voting and dispositive power of the shares are shared between Biovest and the RMM’s Board of Directors (the “RMM Board”). Voting and investment power over the shares managed by the RMM Board is exercised jointly by more than three natural persons and voting and disposition decisions require the approval of a majority of such persons. Accordingly, no single natural person has a controlling decision and no individual director of the RMM should be deemed to be a beneficial owner of the shares. This information has been obtained from Schedule 13G/A filed by the Biovest Entities with the SEC on March 17, 2023. |
| (5) | Reflects shares issued to Exact Sciences in connection with the acquisition of its GPS prostate cancer business, which is described above in Item 5. “Operating and Financial Review and Prospects.” This information is based solely on the Company’s shareholder records. |
| (6) | Reflects shares acquirable upon the exercise of warrants, which are described above in Item 6.B. “Compensation.” |
64
From December 31, 2020 to December 31, 2023, the only significant changes related to the beneficial owners listed in the above table of which we have been notified of through Shareholder Transparency Declarations in accordance with Article 14 of the Belgian Act of May 2, 2007 on the disclosure of important participations in issuers of which shares are admitted to trading on a regulated market and regarding miscellaneous provisions (the “Declarations” and each a “Declaration”) are listed below:
| ● | April 7, 2023 Declaration: MDxHealth was notified that the number of shares with respect to which Valiance Asset Management Limited can exercise voting rights passively crossed below the threshold of 10% of the outstanding shares and voting rights of MDxHealth on February 7, 2023. | |
| ● | March 2, 2023 Declaration: MDxHealth was notified that the number of shares with respect to which MVM Partners, LLC can exercise voting rights passively crossed below the threshold of 20% of the outstanding shares and voting rights of MDxHealth on February 7, 2023 and that the that the number of shares with respect to which Biovest NV can exercise voting rights passively crossed below the threshold of 5% of the outstanding shares and voting rights of MDxHealth on February 7, 2023. | |
| ● | February 21, 2023 Declaration: MDxHealth was notified that the number of shares with respect to which Bleichroeder LP can exercise voting rights crossed below the threshold of 15% of the outstanding shares and voting rights of MDxHealth on February 3, 2023. | |
| ● | December 10, 2021 Declaration: MDxHealth was notified that the number of shares with respect to which Scorpiaux can exercise voting rights passively crossed below the threshold of 3% of the outstanding shares and voting rights of MDxHealth on November 8, 2021. | |
| ● | November 22, 2021 Declaration: MDxHealth was notified that the number of shares with respect to which Bleichroeder LP can exercise voting rights actively crossed above the threshold of 15% of the outstanding shares and voting rights of MDxHealth on November 19, 2021. | |
| ● | November 15, 2021 Declaration: MDxHealth was notified that the number of shares with respect to which Bleichroeder LP can exercise voting rights actively crossed above the threshold of 10% of the outstanding shares and voting rights of MDxHealth on November 4, 2021. | |
| ● | February 3, 2021 Declaration: Biovest NV notified MDxHealth that the number of shares with respect to which Biovest NV can exercise voting rights passively crossed below the threshold of 10% of the outstanding shares and voting rights of MDxHealth on January 26, 2021. |
B. Related Party Transactions
Agreements with the Members of the Board of Directors and the Executive Management
Each non-executive director exercises his/her mandate as a self-employed worker. According to our Articles of Association, the term of a directors’ mandate cannot exceed four years, but may be renewed. The directors’ mandates may be terminated ad nutum (at any time) without any form of compensation. There is no specific agreement between us and non-executive directors which waives or restrains this right of the Company to terminate ad nutum the mandates of the directors.
Currently, all the members of the executive management are engaged on the basis of an employment agreement. The employment agreements are for an indefinite term. The employment agreements include, where appropriate, non-competition undertakings, as well as confidentiality and intellectual property transfer undertakings (which are intended to obtain maximum protection of our interests, under applicable laws and subject to the employee’s agreement).
| ● | We hired Mr. Michael K. McGarrity, acting in the role of Chief Executive Officer, effective as of February 18, 2019. The executive employment agreement with Mr. McGarrity provides that if we terminate the employment agreement without cause or if Mr. McGarrity resigns for good reason, Mr. McGarrity shall be eligible to receive as severance an amount equal to twelve months of base salary in effect at the time of the separation. In addition, we have the right, exercisable at any time, to terminate the executive employment agreement with immediate effect for cause (as defined in the employment agreement) by providing written notice. | |
| ● | Acting under the direction of the Board of Directors, we hired Mr. Ron Kalfus, acting in the role of Chief Financial Officer, effective as of July 22, 2019. The executive employment agreement with Mr. Kalfus provides that if we terminate the employment agreement without cause or if Mr. Kalfus resigns for good reason, Mr. Kalfus shall be eligible to receive as severance an amount equal to six months of base salary in effect at the time of the separation, which amount will increase to twelve months of base salary for a termination that occurs after July 22, 2020. In addition, we have has the right, exercisable at any time, to terminate the executive employment agreement with immediate effect for cause (as defined in the employment agreement) by providing written notice. | |
| ● | Acting under the direction of the Board of Directors, we hired Mr. John Bellano, acting in the role of Chief Commercial Officer, effective as of June 19, 2019. The executive employment agreement with Mr. Bellano provides that if we terminate the employment agreement without cause or if Mr. Bellano resigns for good reason, Mr. Bellano shall be eligible to receive as severance an amount equal to six months of base salary in effect at the time of the separation, which amount will increase to twelve months of base salary for a termination that occurs after June 19, 2020. In addition, we have the right, exercisable at any time, to terminate the executive employment agreement with immediate effect for cause (as defined in the employment agreement) by providing written notice. | |
| ● | The executive employment contract with Mr. Joe Sollee provides that if his employment is terminated for a reason other than serious misconduct or if Mr. Sollee resigns for good reason, he will be entitled to severance pay of nine months gross remuneration and benefits. In addition, we have the right, exercisable at any time, to terminate the executive employment agreement with immediate effect for cause (as defined in the employment agreement) by providing written notice. |
65
Options Granted to Our Board Directors and Executive Management
We have granted subscription rights, or options, to certain members of our Board of Directors and executive management. For more information regarding the options granted to our Board of Directors and executive management. See Item 6B. “Compensation.”
MVM Subscription Agreement
As part of an investment by MVM V LP and MVM GP (No. 5) LP, or collectively MVM, into our share capital, our company entered in April 2020 into a subscription agreement with MVM, or the Subscription Agreement. Pursuant to the Subscription Agreement, MVM is entitled to have one observer at the Board of Directors of our Company for as long as MVM holds in aggregate 5% of our company’s outstanding shares. At the date of this annual report, the observer of MVM at our Board of Directors is Dr. Kyle Dempsey. In addition, we agreed that MVM could propose to our 2020 general shareholders’ meeting to appoint Dr. Eric Bednarski as director of the Company. Our general shareholders’ meeting held on July 30, 2020 approved the appointment of Dr. Eric Bednarski as a director for a term of three years, up to and including the closing of our annual general shareholders’ meeting held on May 25, 2023, at which meeting he was re-elected for two years with a term ending on the date of our annual shareholders’ meeting in 2025.
Offering Purchases
Pursuant to our February 2023 follow-on public equity offering in the United States, (1) the Bleichroeder Entities (as defined above under “A. Major Shareholders”) purchased 1,500,000 shares for an aggregate purchase price of $6,000,000, (2) the MVM Entities (as defined above under “A. Major Shareholders”) purchased 1,000,000 shares for an aggregate purchase price of $4,000,000, and (3) the Valiance Entities (as defined above under “A. Major Shareholders”) purchased and 190,408 shares for an aggregate purchase price of $761,632.
C. Interests of Experts and Counsel
Not applicable.
ITEM 8. FINANCIAL INFORMATION
A. Consolidated Statements and Other Financial Information
Consolidated Financial Statements
Our audited consolidated financial statements are appended at the end of this annual report starting at page F-1, and form a part hereof.
Legal Proceedings
We are not a party to any pending material legal proceedings.
Dividend Policy
We have never declared or paid any cash dividends on our ordinary shares. We do not anticipate paying cash dividends on our equity securities in the foreseeable future and intend to retain all available funds and any future earnings for use in the operation and expansion of our business. In general, distributions of dividends proposed by our Board of Directors require the approval of our shareholders at a meeting of shareholders with a simple majority vote, although our Board of Directors may declare interim dividends without shareholder approval, subject to the terms and conditions of the Belgian Code of Companies and Associations, or CCA. See Item 10B. “Memorandum and Articles of Association.”
Our ability to distribute dividends is subject to availability of sufficient distributable profits as defined under Belgian law on the basis of our stand-alone statutory accounts prepared in accordance with Belgian GAAP. In particular, dividends can only be distributed if following the declaration and issuance of the dividends the amount of our net assets on the date of the closing of the last financial year as follows from the statutory non-consolidated financial statements (i.e., summarized, the amount of the assets as shown in the balance sheet, decreased with provisions and liabilities, all in accordance with Belgian accounting rules), and, save in exceptional cases, to be mentioned and justified in the notes to the annual accounts, decreased with the non-amortized costs of incorporation and extension and the non-amortized costs for research and development, does not fall below the amount of the paid-up capital (or, if higher, the issued capital), increased with the amount of non-distributable reserves (which include, as the case may be, the unamortized part of any revaluation surpluses).
In addition, pursuant to Belgian law and our Articles of Association, we must allocate an amount of 5% of our Belgian GAAP annual net profit to a legal reserve in its stand-alone statutory accounts, until the legal reserve amounts to 10% of our share capital. Our legal reserve currently does not meet this requirement nor will it meet the requirement at the time of the closing. Accordingly, 5% of our Belgian GAAP annual net profit during future years will need to be allocated to the legal reserve, further limiting our ability to pay out dividends to its shareholders.
For information regarding the Belgian withholding tax applicable to dividends and related U.S. reimbursement procedures, see Item 9E. “Taxation — Material Belgian Tax Consequences.”
66
B. Significant Changes
None.
ITEM 9. THE OFFER AND LISTING
A. Offer and Listing Details
Our ADSs were listed on Nasdaq Capital Market under the symbol “MDXH” beginning on November 4, 2021. Prior to that date, there was no public trading market for our ADSs. Our ordinary shares began trading on the regulated market of Euronext in Brussels under the symbol “MDXH” in June 2006. Prior to that date, there was no public trading market for our ADSs or our ordinary shares.
The Company announced on October 2, 2023, that its board of directors had determined that it is in the best interest of the Company, its investors and other stakeholders to consolidate all trading of the Company’s securities on one exchange in the United States. This transaction involved (1) a share consolidation with respect to all outstanding Company shares by means of a 1-for-10 reverse stock split, which was completed on November 13, 2023 (the “Share Consolidation”), after which ten former Company shares are represented by one new Company ordinary share, and each ADS represents one new ordinary share, (2) listing the new ordinary shares on Nasdaq, (3) the mandatory exchange of each outstanding ADS for one new ordinary share (the “Mandatory ADS Exchange”), (4) a repositioning of the new ordinary shares from the Euronext Brussels trading system to the Nasdaq trading system, and (5) following a transition period of at least three weeks after the Mandatory ADS Exchange, the de-listing of the ordinary shares from listing and trading on Euronext Brussels. On November 3, 2023, an extraordinary general shareholders’ meeting of the Company approved the transition from a dual listing of the Company’s ADSs on Nasdaq and ordinary shares on Euronext Brussels to a sole listing of shares on Nasdaq, as well as the Share Consolidation.
Following the completion of the Share Consolidation on November 13, 2023, the Company completed the Mandatory ADS Exchange, effective November 27, 2023. As a result of the Mandatory ADS Exchange, ADS holders received ordinary shares that are listed on Nasdaq in exchange for their ADSs on the basis of a ratio of one ADS for one ordinary share, the ADS program was terminated, and the ADSs ceased to be listed and traded on Nasdaq.
Effective as of the opening of trading on Nasdaq on November 13, 2023, the ordinary shares were admitted to listing and trading on Nasdaq. During a transition period, which ended December 15, 2023, the ordinary shares were listed on both Euronext Brussels and Nasdaq. Effective as of close of trading on Euronext Brussels on December 15, 2023, the ordinary shares are no longer listed and traded on Euronext Brussels and as of that moment on, the ordinary shares have been solely listed and traded on Nasdaq.
No significant trading suspensions have occurred in the prior three years.
B. Plan of Distribution
Not applicable.
C. Markets
For information regarding the stock exchange and regulated market on which our ordinary shares are listed, see Item 9A. “Offer and Listing Details.”
D. Selling Shareholders
Not applicable.
E. Dilution
Not applicable.
F. Expenses of the Issuer
Not applicable.
67
ITEM 10. ADDITIONAL INFORMATION
A. Share Capital
Not applicable.
B. Memorandum and Articles of Association
Corporate Profile
Our legal and commercial name is MDxHealth SA. We are a public limited liability company incorporated in the form of a naamloze vennootschap/société anonyme under Belgian law. We are registered with the Register of Legal Entities (RPM Liège) under the enterprise number 0479.292.440. Our principal executive and registered offices are located at CAP Business Center, Zone Industrielle des Hauts-Sarts, Rue d’Abhooz 31, 4040 Herstal, Belgium and our telephone number is +32 4 257 70 21. Our agent for service of process in the United States is MDxHealth, Inc., whose address is 15279 Alton Parkway, Suite 100, Irvine, CA 92618, United States.
We were incorporated in Belgium on January 10, 2003 for an unlimited duration. Our fiscal year ends December 31.
Corporate Purpose
Our corporate purpose as set forth in Article 3 of our articles of association is as follows:
“The Company’s corporate purpose is to engage in Belgium and abroad, in its own name and on behalf of third parties, alone or in collaboration with third parties, in the following activities:
All forms of research and development into or involving biological cells and organisms (including gene methylation) and chemical compounds, as well as the industrialization and commercialization of the results thereof;
| ● | Research and development into biotechnological or derivative products that could have a market value in applications related to human and animal healthcare, diagnostics, pharmacogenomics and therapeutics, based amongst other things on the technology of genetics, genetic engineering and detention, chemistry and cell biology; |
| ● | Commercialization of the aforementioned products and application domains; |
| ● | Acquisition, disposal, exploitation, commercialization and management of intellectual property, property and usage rights, trademarks, patents, drawings, licenses and any other form of know how. |
The Company is also authorized to engage in all commercial, industrial, financial and real estate transactions which are directly or indirectly related to or which may be beneficial to the achievement of its corporate purpose.
It may, by means of subscription, contribution, merger, collaboration, financial participation or otherwise, take interests or participate in any company, existing or to be incorporated, undertakings, businesses and associations in Belgium or abroad. The company may manage, re-organize or sell these interests and can also, directly or indirectly, participate in the board of directors, management, control and winding-up of companies, undertakings, business and associations in which it has an interest or a participation. The company may provide guarantees and security interests for the benefit of these companies, undertakings, businesses and associations, act as their agent or representative, and grant advances, credit, mortgages or other securities.”
Board of Directors
Belgian law does not specifically regulate the ability of directors to borrow money from us.
Directors are expected to arrange their personal and business affairs so as to avoid conflicts of interest with our Company. When the board takes a decision, board members should disregard their personal interests. They should not use business opportunities intended for the Company for their own benefit.
In accordance with article 7:96 of the Belgian Companies and Associations Code, all directors must inform the board of directors and the statutory auditor of the Company of conflicts of interest as they arise and abstain from voting on the matter involved in accordance with the relevant provisions of the Belgian Companies and Associations Code.
68
Each board member should place the Company’s interests above his/her own. The board members have the duty to look after the interests of all shareholders on an equivalent basis. Each board member should act in accordance with the principles of reasonableness and fairness.
Each board member should inform the board of any conflict of interests that could in their opinion affect their capacity of judgement. In particular, at the beginning of each board or committee meeting, board members should declare whether they have any conflict of interests regarding the items on the agenda.
Each board member should, in particular, be attentive to conflicts of interests that may arise between the Company, its board members, its significant or controlling shareholder(s) and other shareholders. The board members who are proposed by significant or controlling shareholder(s) should ensure that the interests and intentions of these shareholder(s) are sufficiently clear and communicated to the board in a timely manner.
The board should act in such a manner that a conflict of interest, or the appearance of such a conflict, is avoided. In the possible case of a conflict of interest, the board should, under the lead of its chair, decide which procedure it will follow to protect the interests of the Company and all its shareholders. In the next annual report, the board should explain why they chose this procedure. However, where there is a substantial conflict of interests, the board should carefully consider communicating as soon as possible on the procedure followed, the most important considerations and the conclusions.
There are no outstanding loans granted by our Company to any of the members of the board of directors and members of the executive management, nor are there any guarantees provided by our Company for the benefit of any of the members of the board of directors and members of the executive management.
None of the members of the board of directors and members of the executive management has a family relationship with any other of the members of the board of directors and members of the executive management.
The DGCL generally permits transactions involving a Delaware corporation and an interested director of that corporation if (i) the material facts as to the director’s relationship or interest and as to the transaction are disclosed and a majority of disinterested directors consent, (ii) the material facts are disclosed as to the director’s relationship or interest and a majority of shares entitled to vote thereon consent or (iii) the transaction is fair to the corporation at the time it is authorized by the board of directors, a committee of the board of directors or the shareholders.
We rely on a provision in the listing rules of the Nasdaq Stock Market that allows us to follow Belgian corporate law with respect to certain aspects of corporate governance. This allows us to continue following certain corporate governance practices that differ in significant respects from the corporate governance requirements applicable to U.S. companies listed on the Nasdaq Capital Market. In particular, the listing rules of the Nasdaq Stock Market require a majority of the directors of a listed U.S. company to be independent, whereas pursuant to Belgian law, there is no longer a requirement for the Company to have independent directors since the de-listing from Euronext Brussels in December 2023. The listing rules of the Nasdaq Stock Market further require that each of the nominating, compensation and audit committees of a listed U.S. company be comprised entirely of independent directors. However, the Belgian Companies and Associations Code does not require companies that are not listed in the sense of the Belgian Companies and Associations Code to have an audit committee or a nominating committee. At present, our audit committee is composed of three independent directors out of three members. Our nomination and remuneration committees are composed of three independent directors out of five members. Our board of directors currently has no plan to change the composition of our nomination and remuneration committee.
Form and Transferability of Our Shares
All our ordinary shares are fully paid and rank pari passu in all respects with all other existing and outstanding shares of the Company. All of our shares belong to the same class of securities and are in registered form or in dematerialized form. All of our outstanding shares are fully paid-up and freely transferable, subject to any contractual restrictions. Belgian company law and our articles of association entitle shareholders to request, in writing and at their expense, the conversion of their dematerialized shares into registered shares and vice versa. Any costs incurred as a result of the conversion of shares into another form will be borne by the shareholder. For shareholders who opt for registered shares, the shares will be recorded in our shareholder register.
69
Currency
Our share capital, which is represented by our outstanding ordinary shares, is denominated in Euros.
Changes to the share capital decided by the shareholders
In principle, changes to our share capital are decided by our shareholders. Our general shareholders’ meeting may at any time decide to increase or reduce the share capital of the Company. Such resolution must satisfy the quorum and majority requirements that apply to an amendment of the articles of association, as described below under “— Right to attend and vote at general shareholders’ meetings” and “— Quorum and majorities.”
Capital increases decided by the board of directors
Subject to the quorum and majority requirements described below under subsection “— Right to attend and vote at general shareholders’ meetings” and subsection “— Quorum and majorities”, the general shareholders’ meeting may authorize our board of directors, within certain limits, to increase our share capital without any further approval of our shareholders. This is the so-called authorized capital. This authorization needs to be limited in time (i.e. it can only be granted for a renewable period of maximum five years).
By virtue of the resolution of the extraordinary general shareholders’ meeting of the Company held on June 30, 2023, as published by excerpt in the Annexes to the Belgian Official Gazette (Belgisch Staatsblad/Moniteur belge) on July 7, 2023 under number 23368447, which entered into force on July 7, 2023, the board of directors of the Company has been granted certain powers to increase our share capital in the framework of the authorized capital. The powers under the authorized capital have been set out in article 6 of the Company’s articles of association. Pursuant to the authorization granted by the extraordinary general shareholders’ meeting, the board of directors was authorized to increase the share capital of the Company on one or several occasions by a maximum aggregate amount of EUR 163,471,629.58 (excluding issue premium, as the case may be).
The board of directors may increase the share capital by contributions in cash or in kind, by capitalization of reserves, whether available or unavailable for distribution, and capitalization of issue premiums, with or without the issuance of new shares, with or without voting rights, that will have the rights as will be determined by the board of directors. The board of directors is also authorized to use this authorization for the issuance of convertible bonds or subscription rights, bonds with subscription rights or other securities.
In the event of a capital increase decided by the board of directors within the framework of the authorized capital, all issue premiums booked, if any, will be accounted for in accordance with the provisions of these articles of association.
The board of directors is authorized, when exercising its powers within the framework of the authorized capital, to restrict or cancel, in the interest of the company, the preferential subscription rights of the shareholders. This restriction or cancellation of the preferential subscription rights can also be done in favor of members of the personnel of the Company or of its subsidiaries, or in favor of one or more persons other than members of the personnel of the Company or of its subsidiaries.
The board of directors is authorized, with the right of substitution, to amend the articles of association, after each capital increase that has occurred within the framework of the authorized capital, in order to bring them in conformity with the new situation of the share capital and the shares.
So far, the board of directors has used its powers under the authorized capital on October 20, 2023, by issuing 250,000 new shares to Exact Sciences in connection with an amendment to their GPS prostate cancer business asset purchase agreement, deferring the Company’s initial earnout payment by 3 years, from 2024 to 2027, for an aggregate amount of EUR 831,123.31. As a result, the board of directors still has the authority under the authorized capital to increase the share capital of the Company by an aggregate amount of EUR 162,640,506.27 (excluding issue premium, as the case may be).
70
Preferential Subscription Rights
In the event of a capital increase for cash with the issue of new shares, or in the event of an issue of convertible bonds or subscription rights, the existing shareholders have a preferential right to subscribe, pro rata, to the new shares, convertible bonds or subscription rights. These preferential subscription rights are transferable during the subscription period.
Our general shareholders’ meeting may decide to limit or cancel this preferential subscription right, subject to special reporting requirements. Such decision by the general shareholders’ meeting needs to satisfy the same quorum and majority requirements as the decision to increase our share capital.
The shareholders may also decide to authorize our board of directors to limit or cancel the preferential subscription right within the framework of the authorized capital, subject to the terms and conditions set forth in the Belgian Companies and Associations Code. As mentioned above, our board of directors of the Company has been granted certain powers to increase our share capital in the framework of the authorized capital and to cancel the statutory preferential subscription rights of the shareholders (within the meaning of articles 7:191 and 7:193 of the Belgian Companies and Associations Code). The powers under the authorized capital have been set out in article 6 of the Company’s articles of association.
Generally, unless expressly authorized in advance by the general shareholders’ meeting, the authorization of the board of directors to increase our share capital through contributions in cash with cancellation or limitation of the preferential subscription right of the existing shareholders is suspended as of the notification to us by the Belgian Financial Services and Markets Authority, or the FSMA, of a public takeover bid on our financial instruments. Our general shareholders’ meeting did not grant such express authorization to our board of directors. See also “— Capital increases decided by the board of directors” above.
Under Delaware General Corporation Law (the “DGCL”), shareholders of a Delaware corporation have no pre-emptive rights to subscribe for additional issues of stock or to any security convertible into such stock unless, and to the extent that, such rights are expressly provided for in the corporation’s certificate of incorporation.
Acquisition and Sale of own Shares
We may acquire, pledge and dispose of our own shares, profit certificates or associated certificates at the conditions provided for by articles 7:215 and following of the Belgian Companies and Associations Code. These conditions include a prior special shareholders’ resolution approved by at least 75% of the votes validly cast at a general shareholders’ meeting (whereby abstentions are not included in the numerator nor in the denominator) where at least 50% of the share capital and at least 50% of the profit certificates, if any, are present or represented.
Furthermore, shares can only be acquired with funds that would otherwise be available for distribution as a dividend to the shareholders and the transaction must relate to fully paid-up shares or associated certificates. Furthermore, an offer to purchase shares must be made by way of an offer to all shareholders under the same conditions.
Generally, the general shareholders’ meeting or the articles of association determine the amount of shares, profit certificates or certificates that can be acquired, the duration of such an authorization which cannot exceed five years as from the publication of the proposed resolution as well as the minimum and maximum price that the board of directors can pay for the shares. The prior approval by the shareholders is not required if we purchase the shares to offer them to our personnel, in which case the shares must be transferred within a period of 12 months as from their acquisition.
We may, without prior authorization by the general shareholders’ meeting, dispose of the Company’s own shares, profit certificates or associated certificates in the limited number of situations set out in article 7:218 of the Belgian Companies and Associations Code.
As of the date of this annual report, our company does not hold any own shares.
Under the DGCL, a Delaware corporation may purchase or redeem its own shares, unless the capital of the corporation is impaired or the purchase or redemption would cause an impairment of the capital of the corporation.
71
Description of the Rights and Benefits Attached to Our Shares
Right to attend and vote at general shareholders’ meetings
Annual meetings of shareholders
Our annual general shareholders’ meeting is held at the registered office of our Company (in Belgium) or at the place determined in the notice convening the general shareholders’ meeting. The meeting is held every year on the last Thursday of May at 15:00 p.m. (Belgian time). If this day would be a Belgian public holiday, the annual general shareholders’ meeting shall be held on the previous business day. At our annual general shareholders’ meeting, the board of directors submits to the shareholders the audited non-consolidated and consolidated annual financial statements and the reports of the board of directors and of the statutory auditor with respect thereto.
The general shareholders’ meeting then decides on the approval of the statutory annual financial statements, the proposed allocation of the Company’s profit or loss, the release from liability of the directors and the statutory auditor, and, when applicable, the (re-)appointment or dismissal of the statutory auditor and/or of all or certain directors. In addition, as relevant, the general shareholders’ meeting must also decide on the approval of the remuneration of the directors and statutory auditor for the exercise of their mandate (see also “— Voting rights attached to the ordinary shares” below).
Special and extraordinary general shareholders’ meetings
Our board of directors or the statutory auditor (or the liquidators, if appropriate) may, whenever the interest of our Company so requires, convene a special or extraordinary general shareholders’ meeting. Pursuant to article 7:126 of the Belgian Companies and Associations Code, such general shareholders’ meeting must also be convened every time one or more shareholders holding, alone or together, at least 10% of our company’s share capital so request. Shareholders that do not hold at least 10% of our share capital do not have the right to have the general shareholders’ meeting convened.
Under the DGCL, special meetings of the shareholders of a Delaware corporation may be called by such person or persons as may be authorized by the certificate of incorporation or by the bylaws of the corporation, or if not so designated, as determined by the board of directors. Shareholders generally do not have the right to call meetings of shareholders, unless that right is granted in the certificate of incorporation or the bylaws.
Notices convening the general shareholders’ meeting
The notice convening the general shareholders’ meeting must state the place, date and hour of the meeting, must include an agenda indicating the items to be discussed and the proposed resolutions, and must be published at least 15 calendar days prior to the general shareholders’ meeting in the Belgian Official Gazette (Belgisch Staatsblad/Moniteur Belge), in a newspaper that is published nation-wide in Belgium, in paper or electronically, and on our company’s website. A publication in a nation-wide newspaper is not needed for annual general shareholders’ meetings taking place on the date, hour and place indicated in the articles of association of the Company if the agenda is limited to the treatment and approval of the financial statements, the annual report of the board of directors, the report of the statutory auditor, and the discharge from liability of the directors and statutory auditor. See also “— Voting Rights attached to the ordinary shares” below. In addition to this publication, the notice has to be distributed at least 15 calendar days prior to the meeting via the normal publication means that the Company uses for the publication of press releases. The term of 15 calendar days prior to the general shareholders’ meeting for the publication and distribution of the convening notice can be reduced to 10 calendar days for a second meeting if, as the case may be, the applicable quorum for the meeting is not reached at the first meeting, the date of the second meeting was mentioned in the notice for the first meeting and no new item is put on the agenda of the second meeting. See also further below under “— Quorum and majorities.”
At the same time as its publication, the convening notice must also be sent to the holders of registered shares, holders of registered convertible bonds, holders of registered subscription rights, holders of registered certificates issued with the co-operation of the Company (if any), and, as the case may be, to the directors and statutory auditor of the Company. This communication needs to be made by e-mail unless the addressee has informed the Company that it wishes to receive the relevant documentation by another equivalent means of communication. If the relevant addressee does not have an e-mail address or if it did not inform the Company thereof, the relevant documentation will be sent by ordinary mail.
Under the DGCL, unless otherwise provided in the certificate of incorporation or bylaws, written notice of any meeting of the shareholders of a Delaware corporation must be given to each shareholder entitled to vote at the meeting not less than ten nor more than sixty days before the date of the meeting and shall specify the place, date, hour and, in the case of a special meeting, the purpose of the meeting.
72
Formalities to attend the general shareholders’ meeting
All holders of shares, warrants, profit-sharing certificates, non-voting shares, convertible bonds, subscription rights or other securities issued by our company, as the case may be, and all holders of certificates issued with the co-operation of our company (if any) can attend the general shareholders’ meetings insofar as the law or the articles of association entitles them to do so and, as the case may be, gives them the right to participate in voting. The articles of association determine the formalities that shareholders need to fulfill to be admitted to the general shareholders’ meeting. As the case may be, the formalities for the registration of securities holders, and the notification of our company must be described in the notice convening the general shareholders’ meeting.
Electronic participation
Our board of directors has the possibility to organize the general shareholders’ meeting by means of electronic communication which must (i) allow the Company to verify the capacity and identity of the shareholders using it; (ii) at least enable (a) the securities holders to directly, simultaneously and continuously follow the discussions during the meeting and (b) the shareholders to exercise their voting rights on all points on which the general shareholders’ meeting is required to take a decision; and (iii) allow the securities holders to actively participate to the deliberations and to ask questions during the meeting.
Voting by proxy or remote voting
Each shareholder has, subject to compliance with the requirements set forth above under “— Formalities to attend the general shareholders’ meeting”, the right to attend a general shareholders’ meeting and to vote at the general shareholders’ meeting in person or through a proxy holder, who need not be a shareholder. The appointment of a proxy holder must be made in accordance with the applicable rules of Belgian law, including in relation to conflicts of interest and the keeping of a register.
The notice convening the meeting may allow shareholders to vote remotely in relation to the general shareholders’ meeting, by sending a paper form or, if specifically allowed in the notice convening the meeting, by sending a form electronically (in which case the form shall be signed by means of an electronic signature in accordance with applicable Belgian law). These forms shall be made available by our company. The original signed paper form must be received by our company within the term specified by the articles of association. Voting through the signed electronic form may occur until the last calendar day before the meeting.
Our company may also organize a remote vote in relation to the general shareholders’ meeting through other electronic communication methods, such as, among others, through one or several websites. Our company shall specify the practical terms of any such remote vote in the convening notice.
When votes are cast electronically, an electronic confirmation of receipt of the votes is sent to the relevant shareholders that cast the vote. After the general shareholders’ meeting, shareholders can obtain, at least upon request (which must be made no later than three months after the vote), the confirmation that their votes have been validly recorded and taken into account by the Company, unless that information is already available to them.
Holders of securities who wish to be represented by proxy or vote remotely must, in any case comply with the formalities to attend the meeting, as explained under “— Formalities to attend the general shareholders’ meeting.” Holders of shares without voting rights, profit-sharing certificates without voting rights, convertible bonds, warrants or certificates issued with the cooperation of our company may attend the general shareholders’ meeting but only with an advisory vote.
Voting rights attached to the ordinary shares
Each shareholder of the Company is entitled to one vote per ordinary share. Shareholders may vote by proxy, subject to the rules described below in “— Right to attend and vote at general shareholders’ meetings” and “— Voting by proxy or remote voting.”
Voting rights can be mainly suspended in relation to shares:
| ● | which are not fully paid up, notwithstanding the request thereto of the board of directors of the Company; |
| ● | to which more than one person is entitled or on which more than one person has rights in rem (zakelijke rechten/droits réels) on, except in the event a single representative is appointed for the exercise of the voting right vis-à-vis the Company; |
73
| ● | which entitle their holder to voting rights above the threshold of 25% of the total number of voting rights attached to the outstanding financial instruments of our company on the date of the relevant general shareholders’ meeting, in the event that the relevant shareholder has not notified us at least 20 calendar days prior to the date of the general shareholders’ meeting in accordance with the applicable rules of the Belgian Companies and Associations Code; or |
| ● | of which the voting right was suspended by a competent court. |
Pursuant to the Belgian Companies and Associations Code, the voting rights attached to shares owned by the Company, or a person acting in its own name but on behalf of the Company, or acquired by a subsidiary of the Company, as the case may be, are suspended. Generally, the general shareholders’ meeting has sole authority with respect to:
| ● | the approval of the annual financial statements of the Company; |
| ● | the distribution of profits (except interim dividends (see “— Dividends” below)); |
| ● | the appointment and dismissal of directors of the Company; |
| ● | the appointment and dismissal of the statutory auditor of the Company; |
| ● | the granting of release from liability to the directors and the statutory auditor of the Company; |
| ● | the determination of the remuneration of the directors and of the statutory auditor for the exercise of their mandate; |
| ● | the filing of a claim for liability against directors; |
| ● | the decisions relating to the dissolution, merger and certain other reorganizations of the Company; and |
| ● | the approval of amendments to the articles of association. |
Quorum and majorities
In general, there is no attendance quorum requirement for a general shareholders’ meeting and decisions are generally passed with a simple majority of the votes of the shares present or represented. However, capital increases (other than those decided by the board of directors pursuant to the authorized capital), decisions with respect to the Company’s dissolution, mergers, de-mergers and certain other reorganizations of the Company, amendments to the articles of association (other than an amendment of the corporate purpose), and certain other matters referred to in the Belgian Companies and Associations Code do not only require the presence or representation of at least 50% of the share capital of our Company but also a majority of at least 75% of the votes cast (whereby abstentions are not included in the numerator nor in the denominator). An amendment of our company’s corporate purpose requires the approval of at least 80% of the votes cast at a general shareholders’ meeting (whereby abstentions are not included in the numerator nor in the denominator), which can only validly pass such resolution if at least 50% of the share capital of the Company and at least 50% of the profit certificates, if any, are present or represented. In the event where the required quorum is not present or represented at the first meeting, a second meeting needs to be convened through a new notice. The second general shareholders’ meeting may validly deliberate and decide regardless of the number of shares present or represented. The special majority requirements, however, remain applicable.
Under the DGCL, the certificate of incorporation or bylaws of a Delaware corporation may specify the number of shares required to constitute a quorum but in no event shall a quorum consist of less than one-third of shares entitled to vote at a meeting. In the absence of such specifications, a majority of shares entitled to vote shall constitute a quorum.
Right to ask questions
Within the limits of article 7:139 of the Belgian Companies and Associations Code, security holders have a right to ask questions to the directors in connection with the report of the board of directors or the items on the agenda of such general shareholders’ meeting. However, directors may, in the interest of the Company, refuse to answer questions when the communication of certain information or facts could cause prejudice to the Company or is contrary to the obligations of confidentiality entered into by them or by the Company.
Shareholders can also ask questions to the statutory auditor in connection with its report. Such questions can be submitted in writing prior to the meeting or can be asked at the meeting. Written questions to the statutory auditor must be submitted to the Company at the same time. The statutory auditor may, in the interest of the Company, refuse to answer questions when the communication of certain information or facts could cause prejudice to the Company or is contrary to its professional secrecy or to obligations of confidentiality entered into by the Company. The statutory auditor has the right to speak at the general meeting in connection with the performance of its duties.
Written and oral questions will be answered during the meeting concerned in accordance with applicable law. In addition, in order for written questions to be considered, the shareholders who submitted the written questions concerned must comply with the formalities to attend the meeting, as explained under “— Formalities to attend the general shareholders’ meeting.”
74
Dividends
All shares participate equally in the Company’s profits (if any). Pursuant to the Belgian Companies and Associations Code, the shareholders can in principle decide on the distribution of profits with a simple majority vote at the occasion of the annual general shareholders’ meeting, based on the most recent statutory audited financial statements, prepared in accordance with Belgian GAAP and based on a (non-binding) proposal of the Company’s board of directors. The Belgian Companies and Associations Code and the Company’s articles of association also authorize the board of directors to declare interim dividends without shareholder approval. The right to pay such interim dividends is, however, subject to certain legal restrictions.
Our company’s ability to distribute dividends is subject to availability of sufficient distributable profits as defined under Belgian law on the basis of our stand-alone statutory accounts prepared in accordance with Belgian GAAP. In particular, dividends can only be distributed if following the declaration and issuance of the dividends the amount of our net assets on the date of the closing of the last financial year as follows from the statutory non-consolidated financial statements (i.e. summarized, the amount of the assets as shown in the balance sheet, decreased with provisions and liabilities, all in accordance with Belgian accounting rules), decreased with, except in exceptional circumstances, to be disclosed and justified in the notes to the annual accounts, the non-amortized costs of incorporation and extension and non-amortized costs for research and development, does not fall below the amount of the paid-up capital (or, if higher, the issued capital), increased with the amount of non-distributable reserves.
In addition, pursuant to Belgian law and our articles of association, the Company must allocate an amount of 5% of our Belgian GAAP annual net profit (nettowinst/bénéfices nets) to a legal reserve in its stand-alone statutory accounts, until the legal reserve amounts to 10% of our share capital. Our legal reserve currently does not meet this requirement. Accordingly, 5% of our Belgian GAAP annual net profit during future years will need to be allocated to the legal reserve, limiting our ability to pay out dividends to our shareholders.
Under the loan and security agreement entered into between Innovatus Life Sciences Lending Fund I, LP (“Innovatus”) and MDxHealth on August 2, 2022, no distributions can be declared or made without the consent of Innovatus.
In addition, further financial restrictions and other limitations may be contained in future credit agreements.
The right to payment of dividends expires five years after the board of directors declared the dividend payable.
Under the DGCL, a Delaware corporation may pay dividends out of its surplus (the excess of net assets over capital), or in case there is no surplus, out of its net profits for either or both of the fiscal year in which the dividend is declared and the preceding fiscal year (provided that the amount of the capital of the corporation is not less than the aggregate amount of the capital represented by the issued and outstanding stock of all classes having a preference upon the distribution of assets). Dividends may be paid in the form of shares, property or cash.
Appointment of Directors
Pursuant to the Belgian Companies and Associations Code and the articles of association, the board of directors must consist of at least three directors. Our Company’s Corporate Governance Charter provides that the board of directors should have a composition appropriate to the Company’s purpose, its operations, phase of development, structure of ownership and other specifics. Pursuant to the Belgian Companies and Associations Code and the articles of association of the company, the board of directors should be composed of at least three directors.
Liquidation Rights
Our company can only be voluntarily dissolved by a shareholders’ resolution passed with a majority of at least 75% of the votes cast at a meeting of shareholders where at least 50% of the share capital is present or represented. In the event the required quorum is not present or represented at the first meeting, a second meeting needs to be convened through a new notice. The second meeting of shareholders can validly deliberate and decide regardless of the number of shares present or represented.
Under the DGCL, unless the board of directors approves the proposal to dissolve, dissolution of a Delaware corporation must be approved by shareholders holding 100% of the total voting power of the corporation. Only if the dissolution is initiated by the board of directors may it be approved by a simple majority of the corporation’s outstanding shares. The DGCL allows a Delaware corporation to include in its certificate of incorporation a supermajority voting requirement in connection with dissolutions initiated by the board.
75
In the event of the dissolution and liquidation of our company, the assets remaining after payment of all debts and liquidation expenses will be distributed to the holders of our shares, each receiving a sum on a pro rata basis.
Pursuant to article 7:228 of the Belgian Companies and Associations Code, if, as a result of losses incurred, the ratio of our company’s net assets (determined in accordance with Belgian legal and accounting rules for non-consolidated financial statements) to share capital is less than 50%, the board of directors must convene an extraordinary general shareholders’ meeting within two months as of the date upon which the board of directors discovered or should have discovered this undercapitalization. At this general shareholders’ meeting the board of directors needs to propose either the dissolution of the Company or the continuation of the Company, in which case the board of directors must propose measures to ensure the Company’s continuity. The board of directors must justify its proposals in a special report to the shareholders. Shareholders representing at least 75% of the votes validly cast at this meeting have the right to dissolve the Company, provided that at least 50% of our share capital is present or represented at the meeting.
If, as a result of losses incurred, the ratio of the Company’s net assets to share capital is less than 25%, the same procedure must be followed, it being understood, however, that in that event shareholders representing 25% of the votes validly cast at the meeting can decide to dissolve the Company.
Pursuant to article 7:229 of the Belgian Companies and Associations Code, if the amount of the Company’s net assets has dropped below €61,500 (the minimum amount of share capital of a corporation with limited liability organised under the laws of Belgium (naamloze vennootschap/société anonyme)), any interested party is entitled to request the competent court to dissolve the Company. The court can order the dissolution of the Company or grant a grace period within which the Company is to remedy the situation.
If the Company is dissolved for any reason, the liquidation must be carried out by one or more liquidators appointed by the general shareholders’ meeting and whose appointment has been ratified by the enterprise court. Any balance remaining after discharging all debts, liabilities and liquidation costs must first be applied to reimburse, in cash or in kind, the paid-up capital of the shares not yet reimbursed. Any remaining balance shall be equally distributed amongst all the shareholders.
On the date of this report, the Company’s net equity is positive and thus does not fall within the scope of the articles 7:228 or 7:229 of the Belgian Companies and Associations Code.
Specific legislation and jurisdiction
Notification of significant shareholdings
Pursuant to Article 7:83 of the Belgian Companies and Associations Code, a notification to the Company is required by all natural persons and legal persons that directly or indirectly acquire dematerialized voting securities representing the share capital or not of a limited liability company, at the latest within five working days following the day of acquisition, of the number of securities it holds, when the voting rights attached to these securities reach 25% or more of the total voting rights at the time of the transaction requiring notification. This notification is also compulsory within the same period in case of a transfer of securities when as a result the voting rights fall below the 25% threshold mentioned above.
The obligation to disclose significant shareholdings as well as certain other provisions of Belgian law (e.g., merger control and authorized capital) that may apply to the Company, may make an unsolicited tender offer, merger, change in management or other change in control, more difficult. Such provisions could discourage potential takeover attempts that third parties may consider and that other shareholders may consider to be in their best interest and could adversely affect the market price of the shares. These provisions may also deprive shareholders of the opportunity to sell their shares at a premium (which is typically offered in the context of a takeover bid).
In accordance with U.S. federal securities laws, holders of our ordinary shares will be required to comply with disclosure requirements relating to their ownership of our securities. Any person that, after acquiring beneficial ownership of our ordinary shares, is the beneficial owners of more than 5% of our outstanding ordinary shares must file with the SEC a Schedule 13D or Schedule 13G, as applicable, disclosing the information required by such schedules, including the number of our ordinary shares that such person has acquired (whether alone or jointly with one or more other persons). In addition, if any material change occurs in the facts set forth in the report filed on Schedule 13D (including a more than 1% increase or decrease in the percentage of the total shares beneficially owned), the beneficial owner must promptly file an amendment disclosing such change.
Public Takeover Bids
Public takeover bids for the Company’s shares and other securities giving access to voting rights (such as subscription rights or convertible bonds, if any) are subject to supervision by the FSMA. Any public takeover bid must be extended to all of the Company’s voting securities, as well as all other securities giving access to voting rights. Prior to making a bid, a bidder must publish a prospectus which has been approved by the FSMA prior to publication.
76
Belgium has implemented the Thirteenth Company Law Directive (European Directive 2004/25/EC of 21 April 2004) by the Belgian Act of 1 April 2007 on public takeover bids, as amended, or the Belgian Takeover Act, and the Belgian Royal Decree of 27 April 2007 on public takeover bids, as amended, or the Belgian Takeover Decree. As the Company no longer qualifies a listed company under Belgian law since its de-listing from Euronext Brussels in December 2023, the requirement, provided for by the Belgian Act of April 1, 2007, to launch a mandatory bid for all of our outstanding shares and securities giving access to shares if a person, as a result of its own acquisition or the acquisition by persons acting in concert with it or by persons acting on their account, directly or indirectly holds more than 30% of the voting securities in a company that has its registered office in Belgium and of which at least part of the voting securities are traded on a regulated market or on a multilateral trading facility designated by the Belgian Royal Decree of 27 April 2007 no longer applies. This may allow existing shareholders or new investors to acquire significant influence or control over the Company by acquiring the shares in the market without being required to acquire the other outstanding voting securities, as well as for all other securities that entitle the holders thereof to the subscription to the acquisition of or conversion into voting securities.
There are several provisions of Belgian company law and certain other provisions of Belgian law, such as merger control, that may apply towards the Company and which may create hurdles to an unsolicited tender offer, merger, change in management or other change in control. These provisions could discourage potential takeover attempts that other shareholders may consider to be in their best interest and could adversely affect the market price of the shares of the Company. These provisions may also have the effect of depriving the shareholders of the opportunity to sell their shares at a premium.
In addition, pursuant to Belgian company law, the board of directors of Belgian companies may in certain circumstances, and subject to prior authorization by the shareholders, deter or frustrate public takeover bids through dilutive issuances of equity securities (pursuant to the “authorized capital”) or through share buy-backs (i.e. purchase of own shares). In principle, the authorization of the board of directors to increase the share capital of the Company through contributions in kind or in cash with cancellation or limitation of the preferential subscription right of the existing shareholders is suspended as of the notification to the Company by the FSMA of a public takeover bid on the securities of the Company. The general shareholders’ meeting can, however, under certain conditions, expressly authorize the board of directors to increase the capital of the Company in such case by issuing shares in an amount of not more than 10% of the existing shares at the time of such a public takeover bid. (see also “— Rights attached to the ordinary shares”, “— Changes to the share capital” and “— Capital increases decided by the board of directors”).
The Company’s articles of association do not provide for any specific protective mechanisms against public takeover bids.
Squeeze-out
Pursuant to article 7:82 of the Belgian Companies and Associations Code or the regulations promulgated thereunder, a person or legal entity, or different persons or legal entities acting alone or in concert, who own, at least 95% of the securities with voting rights in a limited liability company are entitled to acquire the totality of the securities with voting rights in that company following a squeeze-out offer. With the exception of the securities for which the owner has expressly indicated in writing that he does not wish to relinquish them, the securities not offered at the end of the procedure shall be deemed to have passed automatically to the person making a squeeze-out offer with consignment of the price.
Limitations on the Right to Own Securities
Neither Belgian law nor our articles of association impose any general limitation on the right of non-residents or foreign persons to hold our securities or exercise voting rights on our securities other than those limitations that would generally apply to all shareholders.
Exchange Controls and Limitations Affecting Shareholders
There are no Belgian exchange control regulations that impose limitations on our ability to make, or the amount of, cash payments to residents of the United States.
We are in principle under an obligation to report to the National Bank of Belgium certain cross-border payments, transfers of funds, investments and other transactions in accordance with applicable balance-of-payments statistical reporting obligations. Where a cross-border transaction is carried out by a Belgian credit institution on our behalf, the credit institution will in certain circumstances be responsible for the reporting obligations.
Securities Exercisable for Ordinary Shares (Equity Incentives)
See “Management — Compensation of Our Directors and Executives — Warrant Plans” for a description of securities granted by our board of directors to our directors, members of the executive management team, employees and other service providers.
Listing
Our ordinary shares are listed on the Nasdaq Capital Market under the symbol “MDXH.”
Transfer Agent and Registrar
The transfer agent and registrar for our ordinary shares is Computershare.
77
C. Material Contracts
For information on our material contracts entered into during the two years immediately preceding the date of the filing of this annual report, please refer to Item 4B. “Business Overview” and Item 7B. “Related Party Transactions” of this annual report.
D. Exchange Controls
There are no Belgian exchange control regulations that impose limitations on our ability to make, or the amount of, cash payments to residents of the United States.
We are in principle under an obligation to report to the National Bank of Belgium certain cross-border payments, transfers of funds, investments and other transactions in accordance with applicable balance-of-payments statistical reporting obligations. Where a cross-border transaction is carried out by a Belgian credit institution on our behalf, the credit institution will in certain circumstances be responsible for the reporting obligations.
E. Taxation
The discussion below is for general information only and is not, and should not be interpreted to be, tax advice to any holder of our ordinary shares. Each holder or prospective holder of our ordinary shares is urged to consult his, her or its own tax advisor.
Material U.S. Federal Income Tax Consequences
General
The following is a discussion of the material U.S. federal income tax consequences to U.S. Holders and Non-U.S. Holders, both as defined below, of the ownership and disposition of our ordinary shares as of the date of this report. This discussion is based on the provisions of the Internal Revenue Code of 1986, as amended, or the “Code,” the applicable U.S. Treasury regulations promulgated and proposed thereunder, judicial decisions and current administrative rulings and guidance, all of which are subject to change, possibly on a retroactive basis. This discussion applies to you only if you acquire our ordinary shares and hold such shares as a capital asset within the meaning of Section 1221 of the Code (generally, held for investment). The U.S. Internal Revenue Service, or the “IRS,” may challenge the tax consequences described below, and we have not requested, nor will we request, a ruling from the IRS or an opinion of counsel with respect to the U.S. federal income tax consequences of acquiring, holding or disposing of our ordinary shares. This discussion does not purport to deal with all aspects of U.S. federal income taxation that may be relevant to the ownership of our ordinary shares. In particular, the discussion does not address tax consequences that depend upon an investor’s particular tax circumstances, nor does it cover any state, local or foreign law, the possible application of the U.S. federal estate or gift tax laws. You are urged to consult your own tax advisor regarding the application of the U.S. federal income tax laws to your particular situation as well as any state, local, foreign and U.S. federal estate and gift tax consequences resulting from the ownership and disposition of our ordinary shares. In addition, this discussion does not take into account special U.S. federal income tax rules that apply to particular categories of holders of our ordinary shares, including, without limitation, the following:
| ● | dealers, brokers or traders in securities electing to use a mark-to-market method of accounting; |
| ● | banks, thrifts or other financial institutions; |
| ● | individual retirement or tax-deferred accounts; |
| ● | insurance companies; |
| ● | tax-exempt organizations; |
| ● | regulated investment companies or real estate investment trusts; |
| ● | persons holding our ordinary shares as part of a hedging, straddle, or conversion transaction for U.S. federal income tax purposes; |
| ● | persons required for U.S. federal income tax purposes to conform the timing of income accruals to their financial statements under Section 451 of the Code; |
| ● | persons whose functional currency for U. S. federal income tax purposes is not the U.S. dollar; |
| ● | persons subject to the alternative minimum tax; |
| ● | persons that own, or are treated as owning, 10% or more, by voting power or value, of our outstanding common stock; |
78
| ● | certain former U.S. citizens and residents who have expatriated; or |
| ● | persons receiving our ordinary shares pursuant to the exercise of employee stock options or otherwise as compensation. |
U.S. Holders
For purposes of the discussion below, you are a “U.S. Holder” if you are a beneficial owner of our ordinary shares that is:
| ● | an individual United States citizen or resident alien of the United States (as specifically defined for United States federal income tax purposes); |
| ● | a corporation, or other entity treated as a corporation for U.S. federal income tax purposes, created or organized in or under the laws of the United States, any state thereof or the District of Columbia; |
| ● | an estate whose income is subject to United States federal income tax regardless of its source; or |
| ● | a trust (x) if a U.S. court can exercise primary supervision over the trust’s administration and one or more U.S. persons are authorized to control all substantial decisions of the trust or (y) that, if it was in existence on August 20, 1996, was treated as a U.S. person prior to that date and has a valid election in effect under applicable United States Treasury regulations to be treated as a U.S. person. |
If a partnership (including any entity or arrangement treated as a partnership for U.S. federal income tax purposes) holds our ordinary shares, the tax treatment of a partner in such partnership will generally depend upon the status of the partner and upon the activities of the partnership. If you are a partnership holding our ordinary shares or a partner in such partnership, you should consult your tax advisor with respect to the U.S. federal income tax consequences of the ownership and disposition of our ordinary shares by the partnership.
Distributions
Subject to the “passive foreign investment company”, or PFIC, rules discussed below, the amount of any cash distribution (other than in liquidation) that you receive with respect to our ordinary shares including the amount of any Belgian taxes actually withheld therefrom (described below in “— Material Belgian Tax Consequences”) generally will be taxed to a U.S. Holder as dividend income to the extent such distribution does not exceed our current or accumulated earnings and profits, or E&P, as calculated for U.S. federal income tax purposes. Such income will be includable in your gross income as ordinary income on the date of receipt by the Depositary. Dividends received by individuals and certain other non-corporate U.S. Holders from “qualified foreign corporations” are taxed at the rate of either 0 percent, 15 percent or 20 percent, depending upon the particular taxpayer’s U.S. federal income tax bracket; provided that the recipient-shareholder has held his or her shares as a beneficial owner for more than 60 days during the 121-day period beginning on the date which is 60 days before the shares’ ex-dividend date. A foreign corporation is a “qualified foreign corporation” if the stock with respect to which it pays dividends is traded on an established securities market in the United States, provided that the foreign corporation is not a PFIC.
Our ordinary shares are traded on an established securities market in the United States, although we cannot guarantee that our ordinary shares will be so traded in the future. If we are not a PFIC and we are treated as a qualified foreign corporation, dividends we pay with respect to our ordinary shares would be eligible for the reduced rates of taxation described in this paragraph. We do not expect to be treated as a PFIC for our current taxable year. However, our PFIC status for any taxable year is an annual determination that can be made only after the end of that year and will depend on the composition of our income and assets and the value of our assets from time to time. No assurance can be given that the IRS will not disagree and seek to treat us as a PFIC. If we are a PFIC with respect to a particular U.S. Holder, dividends received from us would be taxed at regular ordinary income tax rates and certain other rules will apply. See “Passive Foreign Investment Company (PFIC),” below. Holders of our ordinary shares should consult their own tax advisors regarding the availability of a reduced dividend tax rate in light of their own particular circumstances.
To the extent any distribution exceeds our E&P, the distribution will first be treated as a tax-free return of capital to the extent of your adjusted tax basis in our ordinary shares and will be applied against and reduce such basis on a dollar-for-dollar basis (thereby increasing the amount of gain and decreasing the amount of loss recognized on a subsequent disposition of such ordinary shares). To the extent that such distribution exceeds your adjusted tax basis, the distribution will be taxed as gain recognized on a sale or exchange of such ordinary shares. However, because we do not maintain calculations of our E&P under U.S. federal income tax principles, it is expected that distributions will generally be reported to U.S. Holders as dividends. Because we are not a U.S. corporation, no dividends-received deduction will be allowed to corporations with respect to dividends paid by us.
79
For U.S. foreign tax credit limitation purposes, dividends received on our ordinary shares will be treated as foreign source income and will generally constitute “passive category income,” or in the case of certain holders, “general category income.” You may be eligible, subject to a number of complex limitations, to claim a foreign tax credit in respect of Belgian taxes actually withheld on dividends paid on our ordinary shares. A U.S. Holder who does not elect to claim a foreign tax credit for foreign tax withheld may instead claim a deduction, for U.S. federal income tax purposes, in respect of such withholding, but only for a year in which such U.S. Holder elects to do so for all creditable foreign income taxes. However, if we are a “U.S.-owned foreign corporation,” solely for foreign tax credit purposes, a portion of the dividends allocable to our U.S. source earnings and profits may be re-characterized as U.S. source. A “U.S.-owned foreign corporation” is any foreign corporation in which U.S. persons own, directly or indirectly, 50% or more (by vote or by value) of the stock. In general, U.S.-owned foreign corporations with less than 10% of earnings and profits attributable to sources within the United States are excepted from these rules. Although we don’t believe we are currently a “U.S.-owned foreign corporation,” we may become one in the future. In such case, if 10% or more of our earnings and profits are attributable to sources within the United States, a portion of the dividends paid on our ordinary shares allocable to our U.S. source earnings and profits will be treated as U.S. source, and, as such, a U.S. Holder may not offset any foreign tax withheld as a credit against U.S. federal income tax imposed on that portion of dividends. The rules governing U.S. foreign tax credits are complex, and we recommend that you consult your tax advisor regarding the applicability of such rules to you.
Sale, Exchange or Other Disposition of Our Ordinary Shares
Subject to the PFIC rules discussed below, generally, in connection with the sale, exchange or other disposition of our ordinary shares:
| ● | you will recognize capital gain or loss equal to the difference (if any) between the amount realized on such sale, exchange or other disposition and your adjusted tax basis in such ordinary shares; |
| ● | such gain or loss will be long-term capital gain or loss if your holding period for such ordinary shares is more than one year at the time of the sale or other disposition; |
| ● | such gain or loss will generally be treated as U.S. source for U.S. foreign tax credit purposes; and |
| ● | your ability to deduct capital losses is subject to limitations. |
Long-term capital gains recognized by individuals and certain other non-corporate taxpayers are taxed at preferential rates. If the consideration received upon the sale or other taxable disposition of our ordinary shares is paid in foreign currency, the amount realized will be the U.S. dollar value of the payment received, translated at the spot rate of exchange on the date of taxable disposition. If ordinary shares are treated as traded on an established securities market, a cash basis U.S. Holder and an accrual basis U.S. Holder who has made a special election (which must be applied consistently from year to year and cannot be changed without the consent of the IRS) will determine U.S. dollar value of the amount realized in foreign currency by translating the amount received at the spot rate of exchange on the settlement date of the sale. An accrual basis United States Holder that does not make the special election will recognize exchange gain or loss to the extent attributable to the difference between the exchange rates on the sale date and the settlement date, and such exchange gain or loss generally will constitute ordinary income or loss.
Passive Foreign Investment Company (PFIC)
In general, a non-U.S. corporation is a PFIC for U.S. federal income tax purposes for any taxable year in which (i) 50% or more of value of its assets (based on an average of the quarterly values of the assets during such taxable year) consists of assets that produce, or are held for the production of, passive income, or (ii) 75% or more of its gross income consists of passive income. A separate determination must be made after the close of each fiscal year as to whether a non-U.S. corporation is a PFIC for that year. For purposes of the above calculations, a non-U.S. corporation that owns, directly or indirectly, at least 25% by value of the shares of another corporation is treated as if it held its proportionate share of the assets of the other corporation and received directly its proportionate share of the income of the other corporation. Passive income generally includes dividends, interest, investment gains and certain rents and royalties. Cash is generally a passive asset for these purposes. The value goodwill is generally treated as an active asset if it is associated with business activities that produce active income.
Based on the current estimates, and expected future composition, of our income and the value of our assets, including goodwill, we do not expect to be a PFIC for our current taxable year. However, our PFIC status for any taxable year is an annual determination that can be made only after the end of that year and will depend on the composition of our income and assets and the value of our assets from time to time. The determination of whether we are a PFIC is fact-intensive and the applicable law is subject to varying interpretation. There can be no assurance that the IRS will agree with our position or that the IRS will not successfully challenge our position including our classification of certain income and assets as non-passive or our valuation of our tangible and intangible assets.
80
If we are treated as a PFIC, gain realized on the sale, exchange or other disposition of your ordinary shares would in general not be treated as capital gain. Instead, such gain would be allocated ratably over your holding period for such ordinary shares. The amounts allocated to the taxable year of the sale, exchange or other disposition and to any year before we became a PFIC would be taxed as ordinary income. The amount allocated to each other taxable year would be subject to tax at the highest rate in effect for such year, together with an interest charge on the tax attributable to each such year. If we are a PFIC for any year during a U.S. Holder’s holding period for our ordinary shares, we generally will continue to be treated as a PFIC with respect to such U.S. Holder for all succeeding years during which such U.S. Holder owns our ordinary shares. Dividends received from ordinary shares will not be eligible for the special tax rates applicable to qualified dividend income for certain non-corporate U.S. Holder if we were treated as a PFIC with respect to the U.S. Holder, either in the taxable year of the distribution or the preceding taxable year, but instead will be taxable at rates applicable to ordinary income. Further, any distribution in respect of our ordinary shares in excess of 125 percent of the average annual distributions on such ordinary shares received by a U.S. Holder during the preceding three years or such U.S. Holder’s holding period, whichever is shorter, would be allocated ratably over the U.S. Holder’s holding period for such ordinary shares and subject to taxation as described with respect to sales, exchanges or other dispositions above. Certain elections may be available that would result in alternative treatments such as mark-to-market treatment of our ordinary shares.
3.8% Medicare Tax on “Net Investment Income”
Certain U.S. Holders that are individuals, estates, and certain trusts are subject to a 3.8% tax on all or a portion of their “net investment income,” which may include any gain realized or amounts received with respect to our ordinary shares, to the extent of their net investment income that, when added to other modified adjusted gross income, exceeds $200,000 for a single taxpayer (or a qualifying head of household), $250,000 for married taxpayers filing a joint return (or a qualifying widower), or $125,000 for a married taxpayer filing a separate return. U.S. Holders should consult their own tax advisors with respect to the applicability of the net investment income tax.
Information Reporting and Backup Withholding
Except in the case of corporations or other exempt holders, amounts received by a U.S. Holder in connection with distributions, if any, paid by Company with respect to ordinary our shares and proceeds from the sale, exchange or other disposition of ordinary shares may be subject to U.S. information reporting requirements and backup withholding unless the U.S. Holder provides an accurate taxpayer identification number and complies with certain certification procedures or otherwise establishes an exemption from backup withholding. Backup withholding is not an additional tax and amounts withheld may be allowed as a credit against the U.S. Holder’s U.S. federal income tax liability and may entitle the U.S. Holder to a refund, provided that certain required information is timely furnished to the IRS.
U.S. Holders who are individuals (and under proposed regulations, certain entities) and who own “specified foreign financial assets” with an aggregate value in excess of $50,000 on the last day of the tax year (or more than $75,000 at any time during the tax year) are generally required to file an information statement along with their tax returns, currently on IRS Form 8938, with respect to such assets, subject to certain exceptions (including an exception for shares held in custodial accounts maintained with a U.S. financial institution) “Specified foreign financial assets” include securities issued by a non-U.S. issuer (which would include our ordinary shares) that are not held in accounts maintained by financial institutions. Higher reporting thresholds apply to certain individuals living abroad and to certain married individuals. Individuals who fail to report the required information could be subject to substantial penalties, and such individuals should consult their own tax advisors concerning the application of these rules to their investment in our ordinary shares.
TAX MATTERS CAN BE COMPLICATED. THE FOREGOING DISCUSSION OF MATERIAL U.S. FEDERAL INCOME TAX CONSEQUENCES IS NOT INTENDED TO BE A COMPLETE ANALYSIS OR DESCRIPTION OF ALL U.S. FEDERAL INCOME TAX CONSEQUENCES OF THE OWNERSHIP AND DISPOSITION OF OUR ORDINARY SHARES. IN ADDITION, THE DISCUSSION DOES NOT ADDRESS TAX CONSEQUENCES THAT DEPEND UPON INDIVIDUAL CIRCUMSTANCES. THIS DISCUSSION DOES NOT ADDRESS ANY U.S. FEDERAL TAX CONSEQUENCES OTHER THAN INCOME TAX OR ANY FOREIGN, STATE OR LOCAL TAX CONSIDERATIONS, NOR ANY TAX CONSEQUENCES OF ANY TRANSACTION OTHER THAN THE OWNERSHIP AND DISPOSITION OF OUR ORDINARY SHARES. ACCORDINGLY, YOU ARE STRONGLY URGED TO CONSULT YOUR OWN TAX ADVISOR TO DETERMINE THE PARTICULAR U.S. FEDERAL, STATE, LOCAL, OR FOREIGN INCOME OR OTHER TAX CONSEQUENCES OF THE OWNERSHIP AND DISPOSITION OF OUR ORDINARY SHARES TO YOU.
Non-United States Holders
For purposes of this discussion, if you are not a U.S. Holder (as defined above), you are a “Non-U.S. Holder”.
Distributions on our ordinary Shares
You generally will not be subject to U.S. federal income tax or withholding on distributions made on our ordinary shares unless:
| ● | you conduct a trade or business in the U.S., and |
| ● | the distributions are effectively connected with the conduct of that trade or business (or, under certain income tax treaties, such distributions are attributable to a permanent establishment that you maintain in the United States). |
If you meet the two tests above, you generally will be subject to tax in respect of such distributions in the same manner as a U.S. Holder, as described above. In addition, any effectively connected distributions received by a non-U.S. corporation may also, under certain circumstances, be subject to an additional “branch profits tax” at a 30-percent rate or such lower rate as may be provided by an applicable income tax treaty.
81
Sale, Exchange or Other Disposition of Our Ordinary Shares
Generally, you will not be subject to U.S. federal income tax or withholding in respect of gain recognized on a sale, exchange or other disposition of our ordinary shares unless:
| ● | your gain is effectively connected with a trade or business that you conduct in the United States (or, under certain income tax treaties, such gain is attributable to a permanent establishment that you maintain in the United States), or |
| ● | you are an individual Non-U.S. Holder and are present in the United States for at least 183 days in the taxable year of the sale, exchange or other disposition, and certain other conditions exist. |
If you meet either of the two tests above, you will be subject to tax in respect of any gain effectively connected with your conduct of a trade or business in the United States generally in the same manner as a U.S. Holder, as described above. Effectively connected gains realized by a non-U.S. corporation may also, under certain circumstances, be subject to an additional “branch profits tax” at a rate of 30-percent or such lower rate as may be provided by an applicable income tax treaty.
Backup Withholding and Information Reporting
Payments, including distributions and proceeds from sales, exchanges or other dispositions in respect of our ordinary shares that are made in the United States or by a U.S.-related financial intermediary will be subject to U.S. information reporting rules. In addition, such payments may be subject to U.S. federal backup withholding. You will not be subject to backup withholding provided that:
| ● | you are a corporation or other exempt recipient, or |
| ● | you provide your correct U.S. federal taxpayer identification number and certify, under penalties of perjury, that you are not subject to backup withholding. |
Amounts withheld under the backup withholding rules may be credited against your U.S. federal income tax, and you may obtain a refund of any excess amounts withheld under the backup withholding rules by filing the appropriate claim for refund with the IRS in a timely manner.
Material Belgian Tax Consequences
General
The following paragraphs are a summary of material Belgian tax consequences of the ownership of our ordinary shares by an investor. The summary is based on laws, treaties and regulatory interpretations in effect in Belgium on the date of this document, all of which are subject to change, including changes that could have retroactive effect.
The summary only discusses Belgian tax aspects which are relevant to U.S. holders of our ordinary shares (“Holders”). This summary does not address Belgian tax aspects which are relevant to persons who are residents in Belgium or engaged in a trade or business in Belgium through a permanent establishment or a fixed base in Belgium. This summary does not purport to be a description of all of the tax consequences of the ownership of our ordinary shares and does not take into account the specific circumstances of any particular investor, some of which may be subject to special rules, or the tax laws of any country other than Belgium. This summary does not describe the tax treatment of investors that are subject to special rules, such as banks, insurance companies, collective investment undertakings, dealers in securities or currencies, persons that hold, or will hold, ordinary shares in a position in a straddle, share-repurchase transaction, conversion transactions, synthetic security or other integrated financial transactions. Investors should consult their own advisers regarding the tax consequences of an investment in ordinary shares in the light of their particular circumstances, including the effect of any state, local or other national laws, treaties and regulatory interpretation thereof.
Dividend Withholding Tax
As a general rule, under current Belgian law, a withholding tax of 30% is levied on the gross amount of dividends paid on or attributed to our ordinary shares, subject to such relief as may be available under applicable domestic or tax treaty provisions. Dividends subject to the dividend withholding tax include all benefits attributed to our ordinary shares. A reimbursement of fiscal capital made in accordance with the Belgian Companies and Associations Code is partly considered to be a distribution of the existing taxed reserves (irrespective whether incorporated into the capital or not) and/or the tax-free reserves incorporated into the capital. The proportion is determined on the basis of the ratio between certain taxed reserves and tax-free reserves incorporated into the capital on the one hand and, on the other hand, the aggregate of such reserves and the fiscal capital. In principle, fiscal capital includes paid-up statutory share capital, and subject to certain conditions, the paid-up issue premiums and the cash amounts subscribed to at the time of the issue of profit sharing certificates.
In case of a redemption by us of own shares, the redemption distribution (after deduction of the portion of fiscal capital represented by the redeemed shares) can be treated as a dividend which in certain circumstances may be subject to a withholding tax of 30%, subject to such relief as may be available under applicable domestic or tax treaty provisions. In case of a liquidation of our Company, any amounts distributed in excess of the fiscal capital will be subject to a 30% withholding tax, subject to such relief as may be available under applicable domestic or tax treaty provisions.
For non-residents, the dividend withholding tax will be the only tax on dividends in Belgium, unless the non-resident holds ordinary shares in connection with a business conducted in Belgium, through a fixed base in Belgium or a Belgian permanent establishment.
82
Relief of Belgian Dividend Withholding Tax
Under the Belgium-United States Tax Treaty (the “Treaty”), there is a reduced Belgian withholding tax rate of 15% on dividends paid by us to a U.S. resident which beneficially owns the dividends and is entitled to claim the benefits of the Treaty under the limitation of benefits article included in the Treaty, (a “Qualifying Holder”). If such Qualifying Holder is a company that owns directly at least 10% of our voting stock, the Belgian withholding tax rate is further reduced to 5%. No withholding tax is however applicable if the Qualifying Holder, is: (i) a company that is a resident of the United States that has owned directly ordinary shares representing at least 10% of our capital for a 12-month period ending on the date the dividend is declared, or (ii) a pension fund that is a resident of the United States, provided that such dividends are not derived from the carrying on of a business by the pension fund or through an associated enterprise.
Under the normal procedure, we or our paying agent must withhold the full Belgian withholding tax (without taking into account the Treaty rate). Qualifying Holders may make a claim for reimbursement for amounts withheld in excess of the rate defined by the Treaty. The reimbursement form (Form 276 Div-Aut.) may be obtained online on the website of the Belgian tax authorities. Qualifying Holders may also, subject to certain conditions, obtain the reduced Treaty rate at source. Qualifying Holders should deliver a duly completed Form 276 Div-Aut. no later than ten days after the date on which the dividend is paid or attributed. U.S. holders should consult their own tax advisors as to whether they qualify for reduction in withholding tax upon payment or attribution of dividends, and as to the procedural requirements for obtaining a reduced withholding tax upon the payment of dividends or for making claims for reimbursement.
Withholding tax is also not applicable, pursuant to Belgian domestic tax law, on dividends paid to certain U.S. pension funds provided that the U.S. pension fund (i) qualifies as a non-resident saver for Belgian withholding tax purposes (i.e., it has a separate legal personality and fiscal residence outside of Belgium and without a permanent establishment or fixed base in Belgium), (ii) has a corporate purpose that consists solely in managing and investing funds collected in order to pay legal or complementary pensions, (iii) has activity that is limited to the investment of funds collected in the exercise of its statutory purpose, without any profit making activity and (iv) is exempt from income taxes in the United States. Furthermore, such pension fund may not contractually be obligated to redistribute the dividends to any beneficial owner of such dividends for whom it would manage our ordinary shares nor obligated to pay a manufactured dividend with respect to our ordinary shares under a securities borrowing transaction (save in certain particular cases as described in Belgian law) and subject to certain procedural formalities. A pension fund not holding the shares — which give rise to dividends — for an uninterrupted period of 60 days in full ownership amounts to a rebuttable presumption that the arrangement or series of arrangements which are connected to the dividend distributions, are not genuine. The withholding tax exemption will in such case be rejected, unless counterproof is provided by the OFP that the arrangement or series of arrangements are genuine.
Under Belgian domestic tax law, a withholding tax exemption is available to dividends paid to a non-resident corporate shareholder (located in a Member State of the European Union or in a country with which Belgium has entered in a double tax treaty including sufficient information exchange provisions) provided that (i) at the date of payment or attribution of the dividend it holds a participation in our company representing at least 10% of our share capital, (ii) this holding is held or will be held in full ownership for an uninterrupted period of at least one year, (iii) this non-resident corporate shareholder is tax resident of the country where it is established according to the tax laws of and the bilateral tax treaties established by such country, (iv) this non-resident corporate shareholder is subject to a corporate income tax regime similar to Belgian corporate income tax regime without benefitting from a tax regime that derogates from our ordinary tax regime and (v) its legal form is (similar to one of the legal forms) listed in the annex of the E.U. directive dated July 23, 1990 (90/435/EC) as amended by the directive of December 22, 2003 (2003/123/EC). The withholding tax exemption will apply provided that certain procedural formalities are complied with.
Finally, a withholding tax exemption is available, pursuant to Belgian domestic tax law, to dividends paid to a non-resident corporate shareholder (located in the European Economic Area or in a country with which Belgium has entered in a double tax treaty including sufficient information exchange provisions) to the extent that at the date of payment or attribution of the dividend it holds a participation in our company representing less than 10% of our share capital but the acquisition value of which is at least €2.5 million and provided that certain other conditions are met, i.e., that (i) this holding is held or will be held in full ownership for an uninterrupted period of at least one year (ii) this non-resident corporate shareholder is subject to a corporate income tax regime similar to Belgian corporate income tax regime without benefitting from a tax regime that derogates from our ordinary tax regime, and (iii) its legal form is (similar to one of the legal forms) listed in the annex I, part A, of the E.U. directive dated November 30, 2011 (2011/96/EU) as amended by the directive of July 8, 2014 (2014/86/EU). This reduced withholding tax will apply only if and to the extent that our ordinary Belgian withholding tax cannot be credited or reimbursed to the non-resident corporate shareholder referred to above and subject to certain procedural formalities.
83
Capital Gains and Losses
Pursuant to the Treaty, capital gains and/or losses realized by a Qualifying Holder from the sale, exchange or other disposition of ordinary shares do not fall within the scope of application of Belgian domestic tax law.
Capital gains realized on ordinary shares by a corporate Holder which is not entitled to claim the benefits of the Treaty under the limitation of benefits article included in the Treaty are generally not subject to taxation in Belgium unless the corporate Holder is acting through a Belgian permanent establishment or a fixed place in Belgium to which our ordinary shares are effectively connected. Capital losses are not deductible.
Private individual Holders who are not entitled to claim the benefits of the Treaty under the limitation of benefits article included in the Treaty and which are holding ordinary shares as a private investment will, as a rule, not be subject to tax on any capital gains arising out of a disposal of ordinary shares. Losses will, as a rule, not be deductible in Belgium. However, if the gain realized by such individual Holders on ordinary shares is deemed to be realized outside the scope of the normal management of such individual’s private estate and the capital gain is obtained or received in Belgium, the gain will in principle be taxable at 33%. The Official Commentary to the ITC 1992 stipulates that occasional transactions on a stock exchange regarding ordinary shares should not be considered as transactions realized outside the scope of normal management of one’s own private estate.
Capital gains realized by such individual Holders on the disposal of ordinary shares for consideration, outside the exercise of a professional activity, to a non-resident company (or a body constituted in a similar legal form), to a foreign state (or one of its political subdivisions or local authorities) or to a non-resident legal entity who is established outside the European Economic Area, are in principle taxable at a rate of 16.5% if, at any time during the five years preceding the sale, such individual Holders has owned directly or indirectly, alone or with his/her spouse or with certain relatives, a substantial shareholding in us (that is, a shareholding of more than 25% of our shares).
Capital gains realized by a Holder upon the redemption of ordinary shares or upon our liquidation will generally be taxable as a dividend. See section “Dividend Withholding Tax.”
Estate and Gift Tax
There is no Belgian estate tax on the transfer of ordinary shares upon the death of a Belgian non-resident.
Donations of ordinary shares made in Belgium may or may not be subject to gift tax in Belgium depending on the modalities under which the donation is carried out.
Belgian Tax on Stock Exchange Transactions
A tax on stock exchange transactions (taxe sur les opérations de bourse/taks op de beursverrichtingen) is generally levied on the purchase and the sale and on any other acquisition and transfer for consideration of existing ordinary shares on the secondary market carried out by a Belgian resident investor through a professional intermediary if (i) executed in Belgium through a professional intermediary, or (ii) deemed to be executed in Belgium, which is the case if the order is directly or indirectly made to a professional intermediary established outside of Belgium, either by private individuals having their usual residence in Belgium, or legal entities for the account of their seat or establishment in Belgium.
The applicable rate amounts to 0.35% of the consideration paid but with a cap of €1,600 per transaction and per party. The tax is due separately from each party to any such transaction, i.e., the seller (transferor) and the purchaser (transferee), both collected by the professional intermediary.
However, if the intermediary is established outside of Belgium, the tax will in principle be due by the ordering private individual or legal entity, unless that individual or entity can demonstrate that the tax has already been paid. Professional intermediaries established outside of Belgium can, subject to certain conditions and formalities, appoint a Belgian representative for tax purposes, which will be liable for the tax on stock exchange transactions in respect of the transactions executed through the professional intermediary.
84
Belgian non-residents who purchase or otherwise acquire or transfer, for consideration, ordinary shares in Belgium for their own account through a professional intermediary may be exempt from the tax on stock exchange transactions if they deliver a sworn affidavit to the intermediary in Belgium confirming their non-resident status.
No stock exchange tax is payable by: (i) professional intermediaries described in Article 2, 9° and 10° of the Belgian Act of August 2, 2002 acting for their own account, (ii) insurance companies described in Article 6 of the Belgian Act of 13 March 2016 on the status and control of insurance and reinsurance companies, (iii) professional retirement institutions referred to in Article 2, 1° of the Belgian Act of October 27, 2006 relating to the control of professional retirement institutions acting for their own account, (iv) collective investment institutions acting for their own account, or (v) regulated real estate companies (for the stock exchange tax only).
No stock exchange tax will thus be due by Holders on the subscription, purchase or sale of ordinary shares, if the Holders are acting for their own account. In order to benefit from this exemption, the Holders must file with the professional intermediary in Belgium a sworn affidavit evidencing that they are non-residents for Belgian tax purposes.
Belgian Annual Tax on Securities Accounts
The Belgian Act of February 17, 2021 introduced a new annual tax on securities accounts due on securities accounts held through an intermediary if the average value of the taxable financial instruments held on this securities account exceeds €1 million during a reference period of 12 consecutive months. This new annual tax on securities accounts is introduced because the previous tax on securities accounts was annulled by the Belgian Constitutional Court.
The annual tax on securities accounts is due irrespective of whether the holder of a securities account is a physical person or a legal entity. If the holder of a securities account is a Belgian resident, the annual tax on securities accounts will be applicable both to securities accounts held in Belgium as well as securities accounts held abroad. For non-residents, only securities accounts held in Belgium fall in scope of the annual tax on securities accounts. A double tax treaty could prevent Belgium to levy the annual tax on securities accounts.
Certain exemptions exist to mitigate the impact of the annual tax on securities accounts on the financial sector. As such, securities accounts held by certain financial undertakings are exempt.
All securities held on a securities account are targeted, such as shares, bonds, participations in investment funds and investment companies, but also derived products, such as index trackers, turbos, real estate certificates and cash. The rate of the annual tax on securities accounts amounts to 0.15% on securities accounts of which the average value exceeds €1 million during a reference period of 12 consecutive months. In order to avoid that the payment of the tax would result in a decrease of the average value below the €1 million threshold, the rate is limited to 10% of the difference between the taxable base and €1 million in those cases. The reference period is a subsequent period of 12 months starting on October 1 and ending September 30 of the subsequent year subject to certain changes in specific circumstances. The average value is calculated by taking the average of the securities accounts values on December 31, March 31, June 30 and September 30.
The tax must be declared and paid by the Belgian resident intermediary with whom the securities account is held. If a securities account is held with a non-resident intermediary, the holder of the securities account itself is responsible for the declaration and the payment of the annual tax on securities accounts. Alternatively, the foreign intermediary could also voluntarily appoint a recognized responsible representative in Belgium to declare and pay the tax.
In case of non-declaration, late, inaccurate or incomplete declaration, as well as non-payment or late payment, a penalty varying from 10% to 200% of the tax due can be imposed. Every holder of the securities account is jointly and severally liable to pay these penalties. The Act furthermore includes a general anti-abuse provision pursuant to which the following is not allowed: (i) distributing taxable financial instruments over different securities accounts to avoid the threshold of €1 million for an individual account, (ii) converting taxable financial instruments into nominative securities (the latter are out of scope of the tax); (iii) transferring a securities account to a foreign legal entity which then transfers the securities to a foreign securities account, etc. In the aforementioned circumstances, there is a refutable presumption that abuse exists. However, the Act also includes situations in which there is an irrefutable presumption of abuse (specific anti-abuse provisions). As such, the following transactions taking place as of October 30, 2020 onwards will be considered to constitute abuse: (i) splitting of a securities account into multiple securities accounts held by the same intermediary; and (ii) the conversion of taxable financial instruments held in a securities account to nominal financial instruments. However, in its judgment of October 27, 2022, the Belgian Constitutional Court annulled the specific anti-abuse provisions as well as the retroactive effect up to October 30, 2020 of the general anti-abuse provision. As a result, only the general anti-abuse provision can still be validly applied and, moreover, only as of February 26, 2021.
Prospective Holders should consult their own tax advisors as to whether they are subject to the new annual tax on securities accounts.
85
Proposed Financial Transactions Tax
On 14 February 2013, the EU Commission adopted the Draft Directive on a common Financial Transaction Tax ("FTT"). Earlier negotiations for a common transaction tax among all 28 EU Member States had failed. The current negotiations between the Participating Member States (i.e. Austria, Belgium, France, Germany, Greece, Italy, Portugal, Slovakia, Slovenia and Spain) are seeking a compromise under "enhanced cooperation" rules, which require consensus from at least nine nations. Estonia already left the negotiations by declaring it would not introduce the FTT.
The Draft Directive currently stipulates that once the FTT enters into force, the Participating Member States shall not maintain or introduce taxes on financial transactions other than the FTT (or VAT as provided in the Council Directive 2006/112/EC of 28 November 2006 on the common system of value added tax). For Belgium, the tax on stock exchange transactions should thus be abolished once the FTT enters into force.
Pursuant to the Draft Directive, the FTT would be payable on financial transactions provided at least one party to the financial transaction is established or deemed established in a Participating Member State and there is a financial institution established or deemed established in a Participating Member State which is a party to the financial transaction, or is acting in the name of a party to the transaction. The FTT would, however, not apply to (inter alia) primary market transactions referred to in article 5(c) of Regulation (EC) No 1287/2006, including the activity of underwriting and subsequent allocation of financial instruments in the framework of their issue.
The rates of the FTT would be fixed by each Participating Member State but for transactions involving financial instruments other than derivatives shall amount to at least 0.1% of the taxable amount. The taxable amount for such transactions would in general be determined by reference to the consideration paid or owed in return for the transfer or the market price (whichever is higher). The FTT should be payable by each financial institution established or deemed established in a Participating Member State which is either a party to the financial transaction, or acting in the name of a party to the transaction or where the transaction has been carried out on its account. Where the FTT due has not been paid within the applicable time limits, each party to a financial transaction, including persons other than financial institutions, would become jointly and severally liable for the payment of the FTT due.
In case of implementation any sale, purchase or exchange of shares would become subject to the FTT at a minimum rate of 0.1% provided the above mentioned prerequisites are met. The issuance of new shares would not be subject to the FTT.
In January 2019, Germany and France proposed that a French-style FTT be levied on the acquisition of shares of listed companies whose head office is in a Member State of the European Union and whose market capitalization exceeds EUR 1 billion on 1 December of the preceding year. The tax should be levied on the transfer of ownership when shares of listed public limited companies are acquired. Initial public offerings, market making and intraday trading should not be taxable.
The tax rate should be no less than 0.2 per cent.
On 11 March 2019, the finance ministers of the Participating Member States met in the margins of the Ecofin meeting. There is consensus among the ministers that the FTT should continue to be negotiated according to the Franco-German proposal.
However, the introduction of the FTT remains subject to negotiations between the Participating Member States. It may therefore be altered prior to any implementation, of which the eventual timing and fate remains unclear. Additional EU Member States may decide to participate or drop out of the negotiations. The project will be terminated if the number of Participating Member States falls below nine.
In the framework of the Multiannual Financial Framework (MFF)/Own Resources negotiations, the European Parliament supported the introduction of the FTT as an Own Resource. The Commission agreed to issue a declaration as part of the overall political agreement. The Commission has recently clarified that "should there be an agreement on this Financial Transaction Tax, the Commission will make a proposal in order to transfer revenues from this Financial Transaction Tax to the EU budget as an own resource. If there is no agreement by end of 2022, the Commission will, based on impact assessments, propose a new own resource, based on a new Financial Transaction Tax. The Commission shall endeavor to make these proposals by June 2024 in view of its introduction by 1 January 2026".
In February 2021, EU Member States have been consulted on their current position regarding the FTT.
On 18 May 2021, the Commission again mentioned in a Communication that it will propose additional new own resources, which could include a Financial Transaction Tax.
Prospective investors should consult their own professional advisors in relation to the FTT.
86
F. Dividends and Paying Agents
Not applicable.
G. Statement by Experts
Not applicable.
H. Documents on Display
We are subject to the information reporting requirements of the Exchange Act applicable to foreign private issuers and under those requirements file reports with the SEC. Those reports may be inspected without charge at the locations described below. As a foreign private issuer, we are exempt from the rules under the Exchange Act related to the furnishing and content of proxy statements, and our supervisory and executive board members and principal shareholders are exempt from the reporting and short-swing profit recovery provisions contained in Section 16 of the Exchange Act. In addition, we are not required under the Exchange Act to file periodic reports and financial statements with the SEC as frequently or as promptly as United States companies whose securities are registered under the Exchange Act. Nevertheless, we file with the SEC an annual report containing financial statements that have been examined and reported on, with and opinion expressed by an independent registered public accounting firm, and we submit quarterly interim consolidated financial data to the SEC under cover of the SEC’s Form 6-K.
We maintain a corporate website at www.mdxhealth.com. We intend to post our annual report on our website promptly following it being filed with the SEC. Information contained on, or that can be accessed through, our website does not constitute a part of this annual report. We have included our website address in this annual report solely as an inactive textual reference.
The SEC maintains a website (http://www.sec.gov) that contains reports, proxy and information statements and other information regarding registrants, such as MDxHealth, that file electronically with the SEC.
With respect to references made in this annual report to any contract or other document of MDxHealth, such references are not necessarily complete and you should refer to the exhibits attached or incorporated by reference to this annual report for copies of the actual contract or document.
I. Subsidiary Information
Not applicable.
J. Annual Report to Security Holders
If we are required to provide an annual report to security holders in response to the requirements of Form 6-K, we will submit the annual report to security holders in electronic format in accordance with the EDGAR Filer Manual.
87
ITEM 11. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
Credit Risk
Credit risk arises from cash and cash equivalents, short-term bank deposits, as well as credit exposure to collaboration partners. Credit risk refers to the risks that counterparty will default on its contractual obligations resulting in financial loss to the Company or its subsidiaries.
At the end of 2023, the Company operated with more than 1,000 different customers, systematically reducing credit risk compared to prior periods.
In the U.S. healthcare system, and particularly within the molecular diagnostic CLIA-laboratory industry, where there are rapid technological advances in diagnostic services, companies provide services to healthcare professionals and their patients, while being reimbursed from commercial and governmental insurance systems. Often these services are provided out-of-network and without supplier contracts. As a result, there is reimbursement risk, separate from credit risk that is characterized by uncertainty in reimbursement value, delays in payment, and ultimately non-payment. This impacts the Company’s revenue recognition and cash collections.
In addition to reimbursement risk associated with commercial third-party payors, credit risk may also arise from amounts due directly from patients. In many cases, payors will cover the entire cost of testing. For example, for tests that fall under the Clinical Laboratory Fee Schedule, there is no co-payment, co-insurance or deductible for patients covered under traditional Medicare. However, patients covered by commercial insurance companies may be responsible for a co-payment, co-insurance, and/or deductible depending on the health insurance plan and individual patient benefit. Credit risk exists for those patients who cannot meet their co-payment or deductible portions.
Customers’ compliance with agreed credit terms is regularly and closely monitored. Trade accounts receivable amounted to $11.1 million as of December 31, 2023, and no allowance for expected credit loss was recorded. The Company applies the simplified approach to providing for expected credit losses (“ECL”) prescribed by IFRS 9, which requires the use of the lifetime expected loss provision for all trade receivables. No ECL has been recorded for other financial assets carried at amortized cost as there is no related credit risk.
The credit risk on cash and cash equivalents of $22.4 million is limited given that the counterparties are banks with high credit scores attributed by international rating agencies.
Interest Rate Risk
Our cash consists of cash in readily available checking accounts and money market accounts. As a result, the fair value of our portfolio is relatively insensitive to interest rate changes. Our long-term debt bears interest at a fixed rate and therefore has no exposure to changes in interest rates.
Foreign Currency
Our European operations, including all sales and expenses, are denominated in Euros. At the end of each reporting period, these assets and liabilities are converted to U.S. dollars at the then-applicable foreign exchange rate. As a result, our business is affected by fluctuations in exchange rates between the U.S. dollar and foreign currencies. We enter into limited foreign currency hedging transactions to mitigate our exposure to foreign currency exchange risks. Exchange rate fluctuations may adversely affect our expenses, results of operations, financial position and cash flows. However, to date, these fluctuations have not been significant and a movement of 10% in U.S. dollar to the Euro exchange rate would not have a material effect on our results of operations.
ITEM 12. DESCRIPTION OF SECURITIES OTHER THAN EQUITY SECURITIES
A. Debt Securities
Not applicable.
B. Warrants and Rights
Not applicable.
C. Other Securities
Not applicable.
D. American Depositary Shares
Not applicable.
88
PART II
ITEM 13. DEFAULTS, DIVIDENDS ARREARAGES AND DELINQUENCIES
Not applicable.
ITEM 14. MATERIAL MODIFICATIONS TO THE RIGHTS OF SECURITY HOLDERS AND USE OF PROCEEDS
For information regarding material modifications to the rights of our security holders, see Item 9A. “Offer and Listing Details.”
ITEM 15. CONTROLS AND PROCEDURES
Disclosure Controls and Procedures
We are responsible for maintaining disclosure controls and procedures, as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act. Disclosure controls and procedures are controls and other procedures designed to ensure that the information required to be disclosed by us in the reports that we file or submit under the Exchange Act is recorded, processed, summarized, and reported within the time periods specified in the SEC’s rules and forms. Our management, with the participation of our Chief Executive Officer (principal executive officer) and our Chief Financial Officer (principal financial officer), has evaluated the effectiveness of our disclosure controls and procedures, as of the end of the period covered by this annual report on Form 20-F. During the year, control deficiencies in certain information technology (“IT”) environments were identified, at which time our management immediately implemented a remediation plan. Although remediation of the identified IT deficiencies is now complete (as of the date of this annual report), our Chief Executive Officer (principal executive officer) and Chief Financial Officer (principal financial officer) concluded that, as of December 31, 2023, our disclosure controls and procedures were not effective due to the material weakness as described in the paragraph “Changes in Control over Financial Reporting”.
Management’s Annual Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over our financial reporting (as such term is defined in Rule 13a-15(f) and 15d-15(f) under the Exchange Act). Our management, with the participation of the Chief Executive Officer and the Chief Financial Officer, conducted an assessment of the effectiveness of our internal control over financial reporting as of December 31, 2023, based on the criteria set forth in “Internal Control — Integrated Framework (2013)” issued by the Committee of Sponsoring Organizations of the Treadway Commission. During the year, control deficiencies in certain IT environments were identified, at which time our management immediately implemented a remediation plan. Although remediation of the identified IT deficiencies is now complete (as of the date of this annual report), our management concluded that, as of December 31, 2023, our internal control over financial reporting was not effective due to the material weakness as described in the paragraph “Changes in Control over Financial Reporting”.
Attestation Report of the Registered Public Accounting Firm
This annual report does not include an attestation report of the company’s registered public accounting firm because MDxHealth is an emerging growth company under the JOBS Act.
Changes in Control over Financial Reporting
Other than as set forth below, there has been no change in our internal control over financial reporting (as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act) during the year ended December 31, 2023 that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
During the year, Management identified deficiencies related to the design of our controls over logical access and segregation of duties, at the application control level, in certain IT environments that are relevant to the preparation of our financial statements. Specifically, (i) periodic user access reviews of roles and permissions were not performed sufficiently throughout the period for certain key IT systems, and (ii) certain key IT systems were not logically restricted, resulting in improper segregation of duties for certain business processes.
These IT deficiencies did not result in a misstatement to the financial statements, however, the deficiencies, when aggregated, could impact our ability to maintain effective segregation of duties, as well as the effectiveness of IT-dependent controls (such as automated controls that address the risk of material misstatement to one or more assertions, along with the IT controls and underlying data that support the effectiveness of system-generated data and reports) that could result in misstatements potentially impacting all financial statement accounts and disclosures that would result in a material misstatement to the annual or interim financial statements that would not be prevented or detected. Accordingly, management has determined these deficiencies in the aggregate constituted a material weakness.
Once identified, management began taking steps to remediate these deficiencies, including redesigning the logical access and placing enhanced segregation of duties, enhancing its internal documentation and monitoring approach to ensure that all procedures designed to restrict access to applications and data, are operating in an optimal manner in order to provide management with comfort that access is properly limited to the appropriate internal personnel. Although management has determined that remediation of the identified IT deficiencies is now complete (as of the date of this annual report), our management concluded that, as of December 31, 2023, our internal control over financial reporting was not effective due to the ongoing remediation efforts still in process at that date.
89
ITEM 16. [RESERVED]
Not applicable.
ITEM 16A. AUDIT COMMITTEE FINANCIAL EXPERT
Currently, our Audit Committee consists of three directors: Regine Slagmulder, Lieve Verplancke and Hilde Windels. The members of the Audit Committee must have a collective expertise relating to the activities of the Company, and at least one member of the Audit Committee must have the necessary competence in accounting and auditing, including qualifying as an “audit committee financial expert” as defined under the Exchange Act. Our Board of Directors has determined that Regine Slagmulder and Hilde Windels each qualify as an “audit committee financial expert,” as defined by SEC rules and regulations. Regine Slagmulder, Lieve Verplancke, and Hilde Windels are independent under Belgian law and the Nasdaq Stock Market listing requirements.
ITEM 16B. CODE OF ETHICS
We have adopted a Code of Business Conduct and Ethics that is applicable to all of our, and our subsidiaries’, directors, officers and employees. The Code of Business Conduct and Ethics is available on our website at www.mdxhealth.com. Our Board is responsible for overseeing the Code of Business Conduct and Ethics. Any waiver of the Code of Business Conduct and Ethics for our directors or executive officers (including our principal financial officers) may be made only by the Board and will be disclosed to the public as required by law or under applicable listing rules. Waivers of the Code of Business Conduct and Ethics for other employees may be made only by our Chief Executive Officer or Compliance Officer and will be reported to our Audit Committee. All amendments to Code of Business Conduct and Ethics Code must be approved by the Board or a committee thereof and, if applicable, shall be promptly disclosed to the public as required by law or under applicable listing rules.
ITEM 16C. PRINCIPAL ACCOUNTANT FEES AND SERVICES
BDO Réviseurs d’Entreprises SRL, has served as our independent registered public accounting firm for 2023 and 2022. Our accountants billed the following fees to us for professional services in each of those fiscal years:
| Thousands of $ | Year ended December 31, | |||||||
| 2023 | 2022 | |||||||
| Audit fees for statutory and consolidated financials | $ | 408 | $ | 239 | ||||
| Other audit fees | $ | - | $ | 191 | ||||
| Audit-related and other fees | $ | 40 | $ | 42 | ||||
| Tax Fees | $ | — | $ | — | ||||
| Total | $ | 448 | $ | 472 | ||||
“Audit Fees” are the aggregate fees billed for the audit of our annual financial statements.
“Other Audit Fees” are mainly the aggregate fees billed in the relation with the Company’s follow-on public offerings and capital raise processes.
“Audit-Related Fees” are the aggregate fees billed for assurance and related services that are reasonably related to the performance of the audit and are not reported under Audit Fees. This category also includes services that generally the independent accountant provides, such as consents and assistance with and review of documents filed with the SEC, as well as the aggregate fees billed for statutory audit services as requested by the Belgian Company Code.
“Tax Fees” are the aggregate fees billed for professional services rendered by the principal accountant for tax compliance, tax advice and tax planning related services.
Audit and Non-Audit Services Pre-Approval Policy
The Audit Committee’s policy is to pre-approve all audit and permissible non-audit services to be performed by the independent registered public accounting firm. These services may include audit services, audit-related services, tax services and other services. All such services provided in fiscal 2023 were pre-approved by the Audit Committee. The Audit Committee has delegated pre-approval authority to its chairman when necessary due to timing considerations. Any services pre-approved by such chairman must be reported to the full Audit Committee at its next scheduled meeting.
90
ITEM 16D. EXEMPTIONS FROM THE LISTING STANDARDS FOR AUDIT COMMITTEES
Not applicable.
ITEM 16E. PURCHASES OF EQUITY SECURITIES BY THE ISSUER AND AFFILIATED PURCHASERS
There were no repurchases of our equity securities during the year ended December 31, 2023.
ITEM 16F. CHANGE IN REGISTRANT’S CERTIFYING ACCOUNTANT
Not applicable.
ITEM 16G. CORPORATE GOVERNANCE
Differences between Our Corporate Governance Practices and the Listing Rules of the Nasdaq Stock Market
The listing rules of the Nasdaq Stock Market include certain accommodations in relation to corporate governance requirements that allow foreign private issuers, to follow “home country” corporate governance practices in lieu of the otherwise applicable corporate governance standards of the Nasdaq Stock Market. The application of such exceptions requires that we disclose each instance of non-compliance with the Nasdaq Stock Market listing rules that we do not follow and describe the Belgian corporate governance practices that we do follow in lieu of the relevant Nasdaq Stock Market corporate governance standard.
We intend to continue to follow Belgian corporate governance practices in lieu of the corporate governance requirements of the Nasdaq Stock Market in respect of the following:
| ● | Quorum at Shareholder Meetings. Nasdaq Stock Market Listing Rule 5620(c) requires that for any meeting of shareholders, the quorum must be no less than 33.33% of the outstanding shares of common voting stock. There is no general quorum requirement under Belgian law for ordinary meetings of shareholders, except in relation to decisions regarding certain matters. See Item 10B. “Memorandum and Articles of Association.” |
| ● | Nomination and Remuneration Committee. Nasdaq Stock Market Listing Rule 5605(d)(2) requires that compensation of officers must be determined by, or recommended to, the Board of Directors for determination, either by a majority of the independent directors, or a compensation committee comprised solely of independent directors. Nasdaq Stock Market Listing Rule 5605(e) requires that director nominees be selected, or recommended for selection, either by a majority of the independent directors or a nominations committee comprised solely of independent directors. Under Belgian law, we are not subject to any such requirements. In particular, we are not required by Belgian law to set up any compensation or nominations committees within our Board of Directors and are therefore not subject to any Belgian legal requirements as to the composition of such committees. However, our Articles of Association provide that our Board of Directors may form committees from among its members. Accordingly, our Board of Directors has set up and appointed a Nomination and Remuneration Committee. |
| ● | Charters. Nasdaq Stock Market Listing Rules 5605(c)(1), (d)(1) and (e)(2) require that each committee of the Board of Directors must have a formal written charter, while under Belgian law this is no longer required for the Company since the de-listing from Euronext Brussels in December 2023 |
| ● | Independent Director Majority. Nasdaq Stock Market Listing Rules 5605(b)(1) and (2) require that a majority of the Board of Directors must be comprised of independent directors and that independent directors must have regularly scheduled meetings at which only independent directors are present. We are not required under Belgian law to have a majority of independent directors on our Board of Directors. However, our Articles of Association provide that our Board of Directors must be comprised of at least three directors. |
| ● | Meetings of Independent Directors. Nasdaq Stock Market Listing Rule 5605(b)(2) requires that independent directors must have regularly scheduled meetings at which only independent directors are present. We do not intend to require our independent directors to meet separately from the full Board of Directors on a regular basis or at all, although the Board of Directors is supportive of its independent members voluntarily arranging to meet separately from the other members of our Board of Directors when and if they wish to do so. |
| ● | Stockholder Approval of Certain Share Issuances. Nasdaq Stock Market Listing Rule 5635(a) requires shareholder approval prior to the issuance of securities in connection with the acquisition of the stock or assets of another company if (1)(A) the common stock has or will have upon issuance voting power equal to or in excess of 20% of the voting power outstanding before the issuance of stock or securities convertible into or exercisable for common stock or (B) the number of shares of common stock to be issued is or will be equal to or in excess of 20% of the number of shares of common stock outstanding before the issuance of the stock or securities; or (2) any director, officer or Substantial Shareholder (as defined in the Nasdaq rules) has a 5% or greater interest (or all such parties have a 10% or greater interest in the aggregate) in the Company or assets to be acquired or in the consideration to be paid in the transaction or series of transactions and the transaction or series of transactions results in an increase in the outstanding common shares or voting power of 5% or more. However, as permitted by Belgian law, we do not need prior stockholder approval to issue shares of authorized stock. |
91
| ● | Stockholder Approval of Equity Compensation Arrangements. Nasdaq Stock Market Listing Rule 5635(c) requires shareholder approval when a plan or other equity compensation arrangement is established or materially amended. Under Belgian law the establishment or amendment of equity compensation arrangements does not require a prior approval by the general shareholders’ meeting. However, pursuant to Belgian law our shareholders must decide any issuance of new equity, as a general matter. As mentioned in Item 10B. “Memorandum and Articles of Association,” the shareholders may also authorize the Board of Directors, within certain limits, to issue new equity (including equity compensation arrangements) in the framework of the so-called authorized capital. By virtue of a resolution of our extraordinary general shareholders’ meeting of June 30, 2023, our Board of Directors was authorized to issue equity (including equity compensation arrangements) in the framework of the authorized capital. Furthermore, the compensation of director mandates is subject to an approval by the general shareholders’ meeting. See also Item 6B. “Management — Compensation of Our Directors and Executive Management.” In the future, we intend to keep following Belgian home country rules and practice. |
ITEM 16H. MINE SAFETY DISCLOSURE
Not applicable.
ITEM 16I. DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS
Not applicable.
ITEM 16J. INSIDER TRADING POLICIES
Not applicable.
ITEM 16K. CYBERSECURITY
Risk Management and Strategy
We operate in the clinical molecular diagnostics laboratory sector, which is subject to various cybersecurity risks that could adversely affect our business, financial condition, and results of operations, including intellectual property theft; fraud; extortion; harm to employees or customers; violation of privacy and healthcare laws and other litigation and legal risk; and reputational risk. We have implemented a risk-based approach to identify and assess the cybersecurity threats that could affect our business and information systems. Our processes also include assessing cybersecurity threat risks associated with our use of third-party services providers in normal course of business use. Third-party risks are included within our cybersecurity risk management processes discussed above. In addition, we assess cybersecurity considerations in the selection and oversight of our third-party services providers, including due diligence on the third parties that have access to our systems and facilities that house systems and data.
Our business depends on the availability, reliability, and security of our information systems, networks, data, and intellectual property. Any disruption, compromise, or breach of our systems or data due to a cybersecurity threat or incident could adversely affect our operations, customer service, product development, and competitive position. They may also result in a breach of our contractual obligations or legal duties to protect the privacy and confidentiality of our stakeholders. Such a breach could expose us to business interruption, lost revenue, ransom payments, remediation costs, liabilities to affected parties, cybersecurity protection costs, lost assets, litigation, regulatory scrutiny and actions, reputational harm, customer dissatisfaction, or harm to our vendor relationships.
Cybersecurity Governance and Oversight
Our board of directors addresses our cybersecurity risk management as part of its general oversight function. We maintain security controls that are continuously reviewed to protect against emerging cyber threats. Our Compliance Department, in collaboration with our IT Department, monitors these security controls and risks and regularly reports to senior management and the board of directors on material developments.
To manage our material risks from cybersecurity threats and to protect against, detect, and prepare to respond to cybersecurity incidents, we undertake the below listed activities:
| ● | Monitor emerging data protection laws in conjunction with our advisors and implement changes to our processes to comply; |
| ● | Maintain firewall and virus protection software; and |
| ● | Maintain a cybersecurity insurance policy. |
Our cybersecurity incident response processes are designed to escalate certain cybersecurity incidents to our Chief Compliance Officer and the other members of management depending on the circumstances, including in some cases to our executive team. The board of directors receives regular reports from management concerning our cybersecurity risk management program. The board also receives various summaries and/or presentations related to cybersecurity threats, risks and mitigation.
As of the date of this Annual Report on Form 20-F, we are not aware of any cybersecurity threats that have materially affected, or are reasonably likely to materially affect, our business strategy, results of operations or financial position.
92
PART III
ITEM 17. FINANCIAL STATEMENTS
Not applicable.
ITEM 18. FINANCIAL STATEMENTS
See pages F-1 through F-43 of this annual report.
ITEM 19. EXHIBITS
Exhibit Index
The following exhibits are filed as part of this annual report:
93
| * | Indicates management compensatory plan, contract or arrangement. |
| # | Certain confidential portions of this Exhibit were omitted by means of marking such portions with brackets (“[***]”) because the identified confidential portions (i) are not material and (ii) would be competitively harmful if publicly disclosed. |
94
SIGNATURES
The registrant hereby certifies that it meets all of the requirements for filing on Form 20-F and that it has duly caused and authorized the undersigned to sign this annual report on its behalf.
| MDXHEALTH SA | ||
| /s/ Michael McGarrity | ||
| By: | Michael McGarrity | |
| Title: | Chief Executive Officer | |
Date: April 30, 2024
95
MDxHealth SA
INDEX TO THE CONSOLIDATED FINANCIAL STATEMENTS
| Page | ||
| Audited Consolidated Financial Statements for the Years Ended December 31, 2023 and 2022 | ||
| Report of Independent Registered Public Accounting Firm PCAOB ID #1432 | F-2 | |
| Consolidated Statement of Profit or Loss | F-3 | |
| Consolidated Statement of Comprehensive Income | F-4 | |
| Consolidated Statement of Financial Position | F-5 | |
| Consolidated Statement of Changes in Equity | F-6 | |
| Consolidated Statement of Cash Flow | F-7 | |
| Notes to Consolidated Financial Statements | F-8 |
F-1
Report of Independent Registered Public Accounting Firm
Shareholders and Board of Directors
MDxHealth SA
Herstal, Belgium
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated statements of financial position of MDxHealth SA (the “Company”) as of December 31, 2023 and 2022, the related consolidated statements of profit or loss, comprehensive income, changes in equity, and cash flows for each of the three years in the period ended December 31, 2023, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company at December 31, 2023 and 2022, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2023, in conformity with the International Financial Reporting Standards (IFRS) as issued by the International Accounting Standards Board (IASB).
Material Uncertainty Related to Going Concern
We draw attention to Note 2.3 in the financial statements, which indicates that the Company incurred net losses and significant cash used in operating activities since its inception through 2023, and is expecting the same for at least the next twelve months. As stated in Note 2.3, the ability to realize the business plan including the Company’s ability to access additional cash through debt, equity or other means, along with the matters set forth in the Note, indicate that a material uncertainty exists that raises substantial doubt on the Company’s ability to continue as a going concern. Management’s plans as regards to these matters are also discussed in Note 2.3 The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/
We have served as the Company’s auditor since 2003.
April 30, 2024
PCAOB
ID #
F-2
Consolidated statement of profit or loss
| Thousands of $ (except per share amounts) For the years ended December 31 |
Notes |
2023 |
2022 |
2021 | ||||||||||
| Services | 4 | |||||||||||||
| Royalties and other revenues | 4 | |||||||||||||
| Revenues | ||||||||||||||
| Cost of sales (exclusive of amortization of intangible assets) | 4 | ( | ) | ( | ) | ( | ) | |||||||
| Gross profit | ||||||||||||||
| Research and development expenses | 5 | ( | ) | ( | ) | ( | ) | |||||||
| Selling and marketing expenses | 5 | ( | ) | ( | ) | ( | ) | |||||||
| General and administrative expenses | 5 | ( | ) | ( | ) | ( | ) | |||||||
| Amortization of intangible assets | 5 | ( | ) | ( | ) | ( | ) | |||||||
| Other operating (expense) income, net | 7 | ( | ) | |||||||||||
| Operating loss | ( | ) | ( | ) | ( | ) | ||||||||
| Financial income | 8 | |||||||||||||
| Financial expenses | 8 | ( | ) | ( | ) | ( | ) | |||||||
| Loss before income tax | ( | ) | ( | ) | ( | ) | ||||||||
| Income tax | 9 | ( | ) | |||||||||||
| Loss for the year | ( | ) | ( | ) | ( | ) | ||||||||
| Loss per share attributable to parent | ||||||||||||||
| 20 | ( | ) | ( | ) | ( | ) | ||||||||
F-3
Consolidated statement of comprehensive income
Thousands
of $ |
Notes | 2023 | 2022 | 2021 | ||||||||||
| Loss for the year | ( | ) | ( | ) | ( | ) | ||||||||
| Other comprehensive income (loss) | ||||||||||||||
| Items that will be reclassified to profit or loss: | ||||||||||||||
| Exchange differences arising from translation of foreign operations | ( | ) | ||||||||||||
| Total other comprehensive income (loss) | ( | ) | ||||||||||||
| Total comprehensive loss for the year (net of tax) | ( | ) | ( | ) | ( | ) | ||||||||
F-4
Consolidated statement of financial position
Thousands of $ For the years ended December 31 | Notes | 2023 | 2022 | |||||||
| ASSETS | ||||||||||
| Non-current assets | ||||||||||
| Goodwill | 3/10 | |||||||||
| Intangible assets | 11 | |||||||||
| Property, plant and equipment | 12 | |||||||||
| Right-of-use assets | 12 | |||||||||
| Financial assets | - | |||||||||
| Total non-current assets | ||||||||||
| Current assets | ||||||||||
| Inventories | 13 | |||||||||
| Trade receivables | 14/19 | |||||||||
| Prepaid expenses and other current assets | 14 | |||||||||
| Cash and cash equivalents | 15/19 | |||||||||
| Total current assets | ||||||||||
| TOTAL ASSETS | ||||||||||
| EQUITY | ||||||||||
| Share capital | 22 | |||||||||
| Issuance premium | 22 | |||||||||
| Accumulated deficit | ( | ) | ( | ) | ||||||
| Share-based compensation | 24 | |||||||||
| Translation reserve | ( | ) | ( | ) | ||||||
| Total equity | ||||||||||
| LIABILITIES | ||||||||||
| Non-current liabilities | ||||||||||
| Loans and borrowings | 16/19 | |||||||||
| Lease liabilities | 16 | |||||||||
| Other non-current financial liabilities | 16/19 | |||||||||
| Total non-current liabilities | ||||||||||
| Current liabilities | ||||||||||
| Loans and borrowings | 16/19 | |||||||||
| Lease liabilities | 16 | |||||||||
| Trade payables | 18/19 | |||||||||
| Other current liabilities | 18 | |||||||||
| Other current financial liabilities | 16/19 | |||||||||
| Total current liabilities | ||||||||||
| Total liabilities | ||||||||||
| TOTAL EQUITY AND LIABILITIES | ||||||||||
F-5
Consolidated statement of changes in equity
| Attributable to owners of mdxhealth sa | ||||||||||||||||||||||||
| Thousands
of $ (except number of shares) | Number of shares | Share capital
& issuance premium | Accumulated Deficit | Share-based compensation | Translation reserve | Total equity | ||||||||||||||||||
| Notes | 22 | 24 | ||||||||||||||||||||||
| Balance at January 1, 2021 | ( | ) | ( | ) | ||||||||||||||||||||
| Loss for the year | ( | ) | ( | ) | ||||||||||||||||||||
| Other comprehensive income | ||||||||||||||||||||||||
| Total comprehensive income for the year | ( | ) | ( | ) | ||||||||||||||||||||
| Transactions with owners in their capacity as owners: | ||||||||||||||||||||||||
| Issuance of shares, net of transaction costs | ||||||||||||||||||||||||
| Share-based compensation costs | ||||||||||||||||||||||||
| Balance at December 31, 2021 | ( | ) | ( | ) | ||||||||||||||||||||
| Balance at January 1, 2022 | ( | ) | ( | ) | ||||||||||||||||||||
| Loss for the year | ( | ) | ( | ) | ||||||||||||||||||||
| Other comprehensive income | ||||||||||||||||||||||||
| Total comprehensive income for the year | ( | ) | ( | ) | ||||||||||||||||||||
| Transactions with owners in their capacity as owners: | ||||||||||||||||||||||||
| Issuance of shares as part of GPS acquisition | ||||||||||||||||||||||||
| Share-based compensation costs | ||||||||||||||||||||||||
| Balance at December 31, 2022 | ( | ) | ( | ) | ||||||||||||||||||||
| Balance at January 1, 2023 | ( | ) | ( | ) | ||||||||||||||||||||
| Loss for the year | ( | ) | ( | ) | ||||||||||||||||||||
| Other comprehensive income | ( | ) | ( | ) | ||||||||||||||||||||
| Total comprehensive income for the year | ( | ) | ( | ) | ( | ) | ||||||||||||||||||
| Transactions with owners in their capacity as owners: | ||||||||||||||||||||||||
| Issuance of shares, net of transaction costs | ||||||||||||||||||||||||
| Issuance of shares as part of amended GPS asset purchase agreement | ||||||||||||||||||||||||
| Share-based compensation costs | ||||||||||||||||||||||||
| Balance at December 31, 2023 | ( | ) | ( | ) | ||||||||||||||||||||
| * | The company completed a share consolidation with respect to all its outstanding shares by means of a 1-for-10 reverse stock split (the “Share Consolidation”) as of November 13, 2023. All share amounts and the EPS were adjusted retroactively to reflect the reverse stock-split. |
F-6
Consolidated statement of cash flow
| Thousands of $ For the years ended December 31 | Notes | 2023 | 2022 | 2021 | ||||||||||
| CASH FLOWS FROM OPERATING ACTIVITIES | ||||||||||||||
| Operating loss | ( | ) | ( | ) | ( | ) | ||||||||
| Depreciation | 12 | |||||||||||||
| Amortization of intangible assets | 11 | |||||||||||||
| Impairment | 11 | |||||||||||||
| Share-based compensation | 24 | |||||||||||||
| Other non-cash transactions | ( | ) | ( | ) | ||||||||||
| Cash used in operations before working capital changes | ( | ) | ( | ) | ( | ) | ||||||||
| Increase (-) / decrease (+) in inventories | 13 | ( | ) | ( | ) | |||||||||
| Increase (-) in receivables | 14 | ( | ) | ( | ) | ( | ) | |||||||
| Increase (+) in payables | 18/19 | |||||||||||||
| Net cash outflow from operating activities | ( | ) | ( | ) | ( | ) | ||||||||
| CASH FLOWS FROM INVESTING ACTIVITIES | ||||||||||||||
| Purchase of property, plant and equipment | 12 | ( | ) | ( | ) | ( | ) | |||||||
| Acquisition and generation of intangible assets | 11 | ( | ) | ( | ) | |||||||||
| Acquisition of Genomic Prostate Score Business | 11 | ( | ) | |||||||||||
| Interests received | 8 | |||||||||||||
| Net cash outflow from investing activities | ( | ) | ( | ) | ( | ) | ||||||||
| CASH FLOWS FROM FINANCING ACTIVITIES | ||||||||||||||
| Proceeds from issuance of shares, net of transaction costs | 22 | |||||||||||||
| Proceeds from loan obligation | 16 | |||||||||||||
| Repayment of loan obligation and debt extinguishment costs | 16 | ( | ) | ( | ) | |||||||||
| Amendment fee related to GPS Asset Purchase Agreement | 16 | ( | ) | |||||||||||
| Payment of lease liability | 16 | ( | ) | ( | ) | ( | ) | |||||||
| Payment of interest | ( | ) | ( | ) | ( | ) | ||||||||
| Other financial expenses | 8 | ( | ) | |||||||||||
| Net cash inflow from financing activities | ||||||||||||||
| Net increase (+) / decrease (-) in cash and cash equivalents | ( | ) | ||||||||||||
| Cash and cash equivalents at beginning of the financial year | ||||||||||||||
| Effect on exchange rate changes | ( | ) | ( | ) | ||||||||||
| Cash and cash equivalents at end of the financial year | 14/18 | |||||||||||||
F-7
NOTE 1: Status and principal activity
When used in this report, all references to “MDxHealth”, the “company”, “we”, “our” and “us” refer to MDxHealth, SA and its subsidiaries. MDxHealth is a limited liability company domiciled in Belgium, with offices and labs in Belgium, the United States and The Netherlands.
MDxHealth is a commercial-stage precision diagnostics company committed to providing non-invasive, clinically actionable and cost-effective urologic solutions to improve patient care. The Company’s novel prostate cancer genomic testing solutions combine advanced clinical modeling with genomic data to provide each patient with a personalized cancer risk profile, which provides more accurate and actionable information than standard risk factors (e.g., PSA, DRE, age) used by clinicians.
The Company’s Select mdx and Confirm mdx solutions address men at risk for developing prostate cancer, providing physicians with a clear clinical pathway to accurately identify clinically significant prostate cancer while minimizing the use of invasive procedures that are prone to complications. The Company’s Genomic Prostate Score (GPS) solution addresses men newly diagnosed with prostate cancer, providing physicians with a clear clinical pathway to make the most informed treatment decision for their individual disease, including active surveillance. The Company’s collective decades of experience in precision diagnostics and its portfolio of novel biomarkers for diagnostic, prognostic and predictive molecular assays supports its active pipeline of new testing solutions for prostate and other urologic diseases.
MDxHealth
offers its laboratory solutions from its state-of-the-art,
The Company is headquartered in Belgium. The parent company, MDxHealth SA, has its registered and corporate office in Cap Business Center, Rue d’Abhooz 31, 4040 Herstal, Belgium. MDxHealth, Inc., the Company’s U.S. subsidiary, is located at 15279 Alton Parkway, Suite 100, Irvine, CA 92618, United States. MDxHealth B.V., the Company’s Dutch subsidiary, is located at Transistorweg 5, 6534 Nijmegen, The Netherlands.
American
Depositary Shares (“ADS”), each representing
NOTE 2: Summary of Material Accounting Policies
2.1. Basis of preparation and statement of compliance
MDxHealth’s consolidated financial statements have been prepared in accordance with the International Financial Reporting Standards (IFRS) and interpretations issued by the IFRS Interpretation Committee (IFRS-IC) applicable to companies reported under IFRS. The financial statements comply with IFRS as issued by the International Accounting Standards Board (IASB), collectively “IFRS”. In addition the financial statements are also prepared in accordance with IFRS as adopted by the EU (“EU IFRS”).
The principal accounting policies applied in the preparation of the consolidated financial statements are set out below. These policies have been consistently applied to all the years presented, unless otherwise stated. The functional and presentation currency is the U.S. Dollar ($) and all amounts are presented in thousands of U.S. Dollars, rounded to the nearest thousand, unless otherwise indicated.
Reclassifications
Certain prior period balances have been reclassified to conform to current period presentation of the Company’s consolidated financial statements and accompanying notes. Such reclassifications have no effect on previously reported results of operations, accumulated deficit, subtotals of operating, investing or financing cash flows or consolidated balance sheet totals.
2.2. Basis of consolidation
The consolidated financial statements incorporate the financial statements of MDxHealth SA (Belgium) and its wholly owned subsidiaries, including MDxHealth Inc. (United States), and MDxHealth BV (The Netherlands) for each fiscal year ending on December 31.
Subsidiaries are all entities over which the Company has control. The Company controls an entity when the Company is exposed to, or has rights to, variable returns from its involvement with the entity and has the ability to affect those returns through its power to direct the activities of the entity. Subsidiaries are fully consolidated from the date on which control is transferred to the Company. The acquisition method of accounting is used to account for business combinations by the Company.
All intercompany balances, profits and transactions are eliminated upon consolidation.
F-8
2.3. Going Concern
The
Company has experienced net losses and significant cash used in operating activities since its inception in 2003, and as of December
31, 2023, had an accumulated deficit of $
As of December 31, 2023, the Company had cash and cash equivalents
of $
Macroeconomic Factors
The Company does not believe that the Ukraine war or the war in Israel/Gaza has an impact on the Company’s ability to continue as a going concern. There is no direct or indirect impact of these conflicts on the day-to-day business of the Company. The Company is not materially impacted by inflation, supply disruption or cyber-attacks due to the current geopolitical conflict.
With regards to climate-related matters, the Company is not impacted in a material way by extreme weather conditions.
2.4. Use of estimates and judgments
Management makes certain critical accounting estimates and management judgments when applying the Company’s accounting policies, which affect the reported amounts of assets and liabilities, the disclosure of contingent assets and liabilities at the date of the financial statements, and the reported amounts of revenue and expenses during the reporting period. Estimates and judgments are continuously evaluated based on historical experience and other factors, including expectations of future events, which are believed to be reasonable under the circumstances. In the future, actual experience may differ from these estimates and assumptions.
Judgements:
Financial Liabilities and Assets (Note 16)
The warrant issued to Exact Sciences as well as the right held by MDxHealth to pay the earnout obligation either in cash or in shares, up to certain limits, were considered as embedded derivatives of the host financial (earnout) liability and not closely related to the host financial liability given the differences in risk and exposure:
| ● | The
fair value of the Company’s option to settle the earnout obligation in cash or through the issuance of additional shares of the
Company was measured based on a Monte Carlo valuation model which takes into account several factors including the expected evolution
in Company’s share price as well as the |
| ● | The
fair value of the warrant held by Exact Sciences to acquire up to |
Revenue recognition (Note 4):
As further described in Note 2.7 (paragraph “Determining the Transaction Price”), the Company analyzes historical collection data on a quarterly basis and makes adjustments to its estimates. In accordance with IFRS 15, revenue is recognized where such a variable consideration is included in the transaction price only to the extent that it is highly probable that the amount of revenue recognized will not be subject to significant future reversals as a result of subsequent re-estimation.
Going Concern (Note 2.3)
Management needs to make significant judgements whether the Company will have sufficient liquidity to continue operations during the next twelve months. Refer to Note 2.3 for management assessment.
F-9
Estimates:
The areas where assumptions and estimation uncertainties in the financial statements have potentially the most significant effect in 2023, are listed below:
Financial Liabilities and Assets (Note 16)
Other financial assets and liabilities are accounted for at fair value through the statement of profit or loss and include the following:
The fair value of the contingent consideration payable to Exact Sciences (for the GPS acquisition) and to NovioGendix, which are presented in the yearend statement of financial position under “other non-current financial liabilities” and “other current financial liabilities” are based on an estimated outcome of the conditional purchase price and contingent payments arising from contractual obligations (level 3 input). These were initially recognized as part of the purchase price and then subsequently measured for fair value. Any changes to fair value are recorded in the statement of profit or loss through either “other operating (expense) income”, “financial income” or “financial expense” depending on the underlying driver for the fair value adjustment.
The fair value of the GPS contingent consideration is based on the estimated timing and amount of the earnout payments. This estimate is then discounted to its net present value, taking into account the expected time when the earnout would become payable in 2025, 2026, and 2027. This contingent consideration was initially recorded along with the purchase price allocation of this business combination as detailed in Note 3.
In
August 2023, the Company and Exact Sciences amended their existing GPS asset purchase agreement in consideration for an amendment fee
of $
The
amendment resulted in a fair value adjustment to the contingent consideration liability of $
The
fair value of the contingent consideration related to NovioGendix is based on a risk-adjusted future cash flows of different scenarios
discounted using an interest rate of
The
fair value of the derivative financial liabilities related to the Innovatus derivative call option (as detailed in Note 16) was performed
using a binomial pricing model which takes into account several factors including the expected evolution in Company’s share price
and are considered as level 3 input. The fair value of the liability for the year ended December 31, 2023, is estimated at $
Impairment Testing (Notes 3, 10 and 11)
The
Company recorded Goodwill of $
The
impairment testing is based on a discounted cash flow (DCF) model, with cash flows for the next
Key underlying estimates are considered to be the estimated cashflows and the weighted-average cost of capital, and are further described in Note 10.
Share-Based Payments (Note 24)
Management estimates the fair value of the equity-settled share-based payment transactions by using the Black-Scholes option valuation model:
| ● | The dividend return is estimated by reference to the historical dividend payment of the Company; currently, this is estimated to be zero as no dividends have been paid since inception; |
| ● | The expected volatility was determined using the average volatility of the stock over the last two years at the date of grant; |
| ● | Risk-free interest rate is based on the interest rate applicable for the |
F-10
2.5. New Standards, Interpretations and Amendments
2.5.1. New Standards, Interpretations and Amendments adopted by the Company
The accounting policies have been consistently applied by the Company and are consistent with those used in previous years.
The following amendments and interpretations issued by the IASB and IFRIC apply for the first time in 2023, but do not have a significant impact on the consolidated financial statements of the Group.
| ● | IFRS 17 Insurance Contracts (issued on 18 May 2017); including Amendments to IFRS 17 (issued on 25 June 2020) |
| ● | Amendments to IAS 8 Accounting policies, Changes in Accounting Estimates and Errors: Definition of Accounting Estimates (issued on 12 February 2021) |
| ● | Amendments to IAS 1 Presentation of Financial Statements and IFRS Practice Statement 2: Disclosure of Accounting policies (issued on 12 February 2021) |
| ● | Amendments to IAS 12 Income Taxes: Deferred Tax related to Assets and Liabilities arising from a Single Transaction (issued on 7 May 2021) |
| ● | Amendments to IFRS 17 Insurance contracts: initial application of IFRS 17 and IFRS 9 – Comparative information (issued on 9 December 2021 |
| ● | Amendments to IAS 12 Income taxes: International Tax Reform – Pillar Two Model Rules (issued on 23 May 2023) |
This adoption has not had any material impact on the disclosures or on the amounts reported in these financial statements.
2.5.2. Standards and Interpretations issued but not yet effective in the current period
Certain new accounting standards and amendments to standards have been published, but were not mandatory for the December 31, 2023, reporting period.
No amendments to standards that are issued but not yet effective are considered to materially affect the Company’s accounting policies or any of the disclosures when applied for the first time.
The following amendments have been issued, but are not mandatory for the financial year beginning January 1, 2023:
| ● | Amendments to IAS 1 ‘Presentation of Financial Statements: Classification of Liabilities as current or non-current’ (effective January 1, 2024), affect only the presentation of liabilities in the statement of financial position — not the amount or timing of recognition of any asset, liability income or expenses, or the information that entities disclose about those items. The amendments: |
| o | clarify that the classification of liabilities as current or non-current should be based on rights that are in existence at the end of the reporting period and align the wording in all affected paragraphs to refer to the “right” to defer settlement by at least twelve months and make explicit that only rights in place “at the end of the reporting period” should affect the classification of a liability; |
| o | clarify that classification is unaffected by expectations about whether an entity will exercise its right to defer settlement of a liability; and make clear that settlement refers to the transfer to the counterparty of cash, equity instruments, other assets or services; and |
| o | clarify how conditions with which an entity must comply within 12 months after the reporting period, such as covenants, affect the corresponding liability’s classification. |
| ● | Amendments to IFRS 16 Leases: Lease Liability in a Sale and Leaseback (effective January 1, 2024). The amendments explain how an entity accounts for a sale and leaseback after the date of the transaction, specifically where some or all the lease payments are variable lease payments that do not depend on an index or rate. They state that, in subsequently measuring the lease liability, the seller-lessee determines ‘lease payments’ and ‘revised lease payments’ in a way that does not result in the seller-lessee recognizing any amount of the gain or loss that relates to the right of use it retains. Any gains and losses relating to the full or partial termination of a lease continue to be recognized when they occur as these relate to the right of use terminated and not the right of use retained. |
| ● | Amendments to IAS 7 ‘Statement of Cash Flows’ and IFRS 7 ‘Financial Instruments: Disclosures’1: Supplier Finance Arrangements, effective 1 January 2024 |
| ● | Amendments to IAS 21 ‘The Effects of Changes in Foreign Exchange Rates: Lack of Exchangeability’1, effective 1 January 2025 |
The Company is analyzing the impact, if any, on its consolidated financial statements in view of the application of the amendment of IAS 1.
F-11
2.6. Foreign currency translation
Functional and presentation currency
Items included in the financial statements of each of the Company’s entities are measured using the currency of the primary economic environment in which the entity operates (the functional currency). The Company’s functional and presentation currency is the U.S. dollar based on the continuing development of the commercial activities in the U.S. market.
Foreign currency transactions are translated into the functional currency using the exchange rates at the date of the transactions. Foreign exchange gains and losses resulting from the settlement of such transactions and from the translation of monetary assets and liabilities denominated in foreign currencies at year-end exchange rates are generally recognized in profit or loss.
The results and financial positions of foreign operations that have a functional currency different from the presentation currency are translated into the presentation currency as follows:
| ● | Assets and liabilities for each statement of financial position presented are translated at the closing rate at the date of that balance sheet. At December 31, 2023, the official exchange rate applied for assets and liabilities was € |
| ● | Income and expenses for each statement of profit or loss and statement of comprehensive income are translated at average exchange rates. At December 31, 2023, the official exchange rate applied for income and expenses was € |
| ● | All resulting exchange differences are recognized in other comprehensive income. |
2.7. Revenue recognition
Performance obligations and timing of revenue recognition
The majority of the Company’s revenue is derived from laboratory services with revenue recognized at a point in time when control of the services has transferred to the customer. This is generally when the test results are delivered to the customer.
Minor other Company’s revenue is derived from license fees and royalties:
| ● | License fees are recognized when the Company has fulfilled all conditions and obligations. If the Company has continuing performance obligations towards the fees, the fee will be recognized on a straight-line basis over the contractual performance period. |
| ● | Royalties are recognized as revenue once the amounts due can be reliably estimated based on the sale of the underlying products and services and when the collection of the royalties can be reasonably assured. |
License up-front (signature fees) and non-refundable fees for access to prior research results and databases are recognized when earned, if the Company has no continuing performance obligations and all conditions and obligations are fulfilled. If the Company has continuing performance obligations towards the fees, the fee will be recognized on a straight-line basis over the contractual performance period.
Royalties are generated from the sales by third parties of products or services which incorporate the Company’s proprietary technology. Royalties are recognized as revenue once the amounts due can be reliably estimated based on the sale of the underlying products and services and when the collection of the royalties can be reasonably assured.
Determining the transaction price
A large portion of the Company’s revenues are derived from Medicare, which reimburses the Company for tests performed on its insured patients. Medicare has set a fixed price (via a Local Coverage Determination or “LCD”) for the Company’s Confirm mdx, Select mdx, and GPS tests. Therefore, the amount of revenue recognized from Medicare for these tests is determined by reference to the LCD pricing.
For other patients insured by commercial insurance companies where there is no certainty of the amount that will be paid for services rendered, the Company uses historical collection data – on an individual payor basis – to estimate its future collection and corresponding revenues that should be recognized for each type of test.
The Company analyzes historical collection data on a monthly basis and makes monthly adjustments to its estimates. In accordance with IFRS 15, revenue is recognized where such a variable consideration is included in the transaction price only to the extent that it is highly probable that the amount of revenue recognized will not be subject to significant future reversals as a result of subsequent re-estimation.
F-12
When historical collection data is insufficient to estimate future collections, the Company defaults to cash basis, meaning that revenues will not be recognized until actual cash payment is received from the payor.
Total revenue in any given year includes amounts related to tests performed in previous years that relate to:
| ● | revenue from transactions in previous years that did not previously meet the revenue recognition criteria; |
| ● | differences between the revenue recognized in previous years and the actual amount received; and |
| ● | reversals of revenue relating to balances that are outstanding for more than 9 months. |
2.8. Segment information
Information for the Company’s operating segments has been determined by reference to the information used by the chief operating decision maker (“CODM”) of the Company to review the performance of the Company and in making decisions on allocation of resources, the nature of the activities and the management structure and accountabilities. The Company’s CEO has been identified as the chief operating decision maker in accordance with his designated responsibility for the allocation of resources to operating segments and assessing their performance through periodic reporting. The CODM periodically reviews the Company’s performance based on information at a company level.
The Company monitors the profitability of the group as a whole given revenues are generated from clinical laboratory service testing and does accordingly not distinguish different business segments.
2.9. intangible assets
Initial measurement:
Externally acquired
Intangible assets are recognized on business combinations if they are separable from the acquired entity or give rise to other contractual/legal rights. The amounts ascribed to such intangibles are determined using appropriate valuation techniques.
Intangible assets are recognized on the business combinations of NovioGendix in 2015 (Select mdx) and GPS in 2022 and include:
| ● | Externally acquired intellectual property, including patents, technology and related IP; and |
| ● | Customers. |
All were valued through application of the relief from royalty method, except for the customers which were valued on the basis of multi-period excess earnings method.
Externally acquired intangible assets also include patents and software licenses which are initially recognized at cost.
Internally generated intangible assets (development costs)
Development costs are capitalized when requirements for capitalization during the development phase have been met. In absence of meeting the requirements, these are expensed in the period in which they were incurred as research and development expenses.
Internally generated intangible assets relate to Confirm mdx, Select mdx, Resolve mdx and GPS.
Subsequent measurement
Intangible assets are carried at their cost less any accumulated amortization and any accumulated impairment losses amortized on a straight-line basis over their estimated useful lives on the following basis:
| ● | Patents & software: shorter of (a) 5 years; or (b) the software license period / patent life |
| ● | Intellectual property: |
| ● | Customers: |
| ● | Capitalized development costs: |
Amortization over the asset’s useful life shall begin when the asset is available for use. Amortization of intangible assets are presented as a separate line in the consolidated statement of profit or loss.
F-13
2.10. Property, plant and equipment
Property, plant and equipment are stated at historical cost less accumulated depreciation and impairment. Repair and maintenance costs are charged to the income statement as incurred. Depreciation is charged to write off the cost or valuation of assets over their useful lives, using the straight-line method, on the following basis:
| ● | Equipment: |
| ● | IT hardware and software: |
| ● | Furniture: |
| ● | Vehicles: |
| ● | Leasehold improvements: in line with the non-cancellable lease period of the related lease |
2.11. Right-of-use assets and liabilities
Right-of-use assets:
The
Company recognizes right-of-use assets at the commencement date of the lease (i.e., the date the underlying asset is available for use).
Right-of-use assets are measured at cost, less any accumulated depreciation and impairment losses, and adjusted for any remeasurement
of lease liabilities. The cost of right-of-use assets includes the amount of lease liabilities recognized, initial direct costs incurred,
and lease payments made at or before the commencement date less any lease incentives received. Unless the Company is reasonably certain
to obtain ownership of the leased asset at the end of the lease term, the recognized right-of-use assets are depreciated on a straight-line
basis over the shorter of its estimated useful life and the lease term. Depreciation periods range between
Lease liabilities:
At the commencement date of the lease, the Company recognizes lease liabilities measured at the present value of lease payments to be made over the lease term. The lease payments include fixed payments (including in substance fixed payments) less any lease incentives receivable, variable lease payments that depend on an index or a rate, and amounts expected to be paid under residual value guarantees. The lease payments also include the exercise price of a purchase option reasonably certain to be exercised by the Company and payments of penalties for terminating a lease, if the lease term reflects the Company exercising the option to terminate. The variable lease payments that do not depend on an index or a rate are recognized as expense in the period on which the event or condition that triggers the payment occurs. In calculating the present value of lease payments, the Company uses the incremental borrowing rate at the lease commencement date which is in the following ranges:
| ● | Buildings: |
| ● | Vehicles: |
| ● | Materials: |
After the commencement date, the amount of lease liabilities is increased to reflect the accretion of interest and reduced for the lease payments made. In addition, the carrying amount of lease liabilities is remeasured if there is a modification, a change in the lease term, a change in the in-substance fixed lease payments or a change in the assessment to purchase the underlying asset.
2.12. Impairment of assets
Goodwill and intangible assets that have an indefinite useful life are not subject to amortization and are tested annually for impairment, or more frequently if events or changes in circumstances indicate that they might be impaired. Other assets are tested for impairment whenever events or changes in circumstances indicate that the carrying amount may not be recoverable. An impairment loss is recognized for the amount by which the asset’s carrying amount exceeds its recoverable amount. The recoverable amount is the higher of an asset’s fair value less costs of disposal and value in use. The Company monitors its Goodwill at consolidated Company level which is the level of the Company of cash-generating units (CGUs) benefiting from the synergies. Non-financial assets other than Goodwill that suffered an impairment are reviewed for possible reversal of the impairment at the end of each reporting period.
2.13. Inventories
Inventories are initially recognized at cost, and subsequently at the lower of cost and net realizable value. Cost comprises merely purchase costs, as the inventory consists solely of raw materials. Raw materials are not ordinarily interchangeable, and they are as such accounted for using the specific identification of their individual cost.
The Company does not account for work in progress and finished products.
2.14. Cash and cash equivalents
Cash and cash equivalents are carried on the consolidated statement of financial position at nominal value. For the purposes of the cash flow statements, cash and cash equivalents comprise cash on hand, deposits held on call with banks, other short-term highly liquid investments and bank overdrafts. Bank overdrafts, if any, are included in borrowings included in current liabilities.
F-14
2.15. Taxation
Current tax assets and liabilities for the current period are measured at the amount expected to be recovered from or paid to the taxation authorities. The tax rates and tax laws used to compute the amount are those that are enacted or substantively enacted, at the reporting date.
Deferred income tax is provided in full using the “balance sheet liability method”, on temporary differences between the carrying amount of assets and liabilities for financial reporting purposes and the amounts used for taxation purposes.
Deferred tax liabilities are recognized for all taxable differences. Deferred tax assets are recognized for all deductible temporary differences, carry forward of unused tax credits and unused tax losses, to the extent that it is probable that taxable profit will be available against which the deductible temporary differences, and the carry forward of unused tax credits and unused tax losses can be utilized.
The carrying amount of deferred tax assets is reviewed at each reporting date and reduced to the extent that it is no longer probable that sufficient taxable profit will be available to allow all or part of the deferred tax asset to be utilized. Unrecognized deferred tax assets are reassessed at each reporting date and are recognized to the extent that it has become probable that future taxable profits will allow the deferred tax asset to be recovered.
Deferred tax assets and liabilities are measured at the tax rates that are expected to apply in the year when the asset is realized or the liability is settled, based on tax rates (and tax laws) that have been enacted or substantively enacted at the reporting date.
Deferred tax assets and deferred tax liabilities are offset, if a legally enforceable right exists to set off current tax assets against current income tax liabilities and the deferred taxes relate to the same taxable entity and the same taxation authority.
2.16. Share Capital
Ordinary shares are classified as equity. Incremental costs directly attributable to the issue of new shares or options are shown in equity as a deduction, net of tax, from the proceeds.
2.17. Financial Assets
The financial assets consist mainly of trade receivables and other current assets (deposits) as well as the Company’s option to settle the earnout obligation to Exact Sciences in cash or through the issuance of additional shares of the Company (refer to Note 3 for additional details on the Company’s earnout obligation to Exact Sciences).
Trade receivables and other current assets (deposits)
Classification and measurement on initial recognition
The classification of financial assets at initial recognition depends on the financial asset’s contractual cash flow characteristics and the Company’s business model for managing them.
The company initially measures a financial asset at its fair value plus, in the case of a financial asset not at fair value through profit or loss, transaction costs.
For
both trade receivables that do not contain a significant financing component, and trade receivables for which the collection is expected
in less than
Trade receivables do not carry any interest and are recognized initially at fair value and subsequently measured at amortized cost using the effective interest method, less provision for impairment based on expected credit losses, where applicable.
Subsequent measurement
After initial recognition, trade receivables and some other current assets as listed in Note 14 are measured at amortized cost using the effective interest method, less provision for impairment based on expected credit losses.
Option to settle the earnout obligation to Exact Sciences in cash or through the issuance of additional shares of the Company
Classification and measurement on initial recognition
At
the option of MDxHealth, the earnout amounts to Exact Sciences can be settled in cash or through the issuance of additional shares of
the Company (valued in function of a volume weighted average trading price of the Company's shares at the end of the relevant earnout
period) to Exact Sciences, provided that the aggregate number of shares held by Exact Sciences shall not exceed more than
F-15
This option is considered as an embedded derivative of the host financial (earnout) liability and not closely related to the host financial liability and is recognized at fair value through the statement of profit or loss at each closing date using the Monte Carlo valuation model, as further detailed in Note 3.
2.18. Financial Liabilities
The financial liabilities consist mainly of loans and borrowings, lease liabilities, trade and other payables and other financial liabilities that include derivative financial liabilities and contingent consideration related to business combinations.
Measurement on initial recognition
At initial recognition, financial liabilities are measured at fair value less transaction costs unless the financial liability is carried at fair value through the statement of profit or loss, in which case the transaction costs are immediately recognized in the statement of profit or loss. The best estimate of the fair value at initial recognition is usually the transaction price, represented by the fair value of the consideration given or received in exchange for the financial instrument. Any difference between the fair value estimated by the entity and the transaction price (“day one gain or loss”) is recognized:
| ● | in the statement of profit or loss if the estimate is evidenced by a quoted price in an active market; and |
| ● | deferred as an adjustment to the carrying amount of the financial instrument in all other cases. |
The fair value of the contingent consideration payable at the date of acquisition is computed as the sum of the probability-weighted fair values of the purchase prices, as follows:
| ● | GPS: the liability recognized reflects a probability-weighted estimate at the current net present value at the date of acquisition, which is expected to become payable. |
| ● | NovioGendix: each of the potential product development paths. The fair value of each path is in turn computed as the sum of the survival probability discounted to present values of the contingent payments in each such path, including the milestone and commercialization payments. Any other financial liability included in the consideration payable for a business combination is recorded at fair value at the date of acquisition. |
The fair value of the derivative financial liabilities is determined as follows:
| ● | Exact Sciences 5-Year Warrants: The fair value of the warrant held by Exact Sciences to acquire up to |
| ● | Innovatus: The derivative financial instrument related to the Innovatus debt facility option to convert up to |
Subsequent measurement
After initial recognition, loans and borrowings, lease liabilities, trade and other payables, are measured at amortized cost using the effective interest method. Contingent considerations and derivative financial liabilities are measured at fair value and are reviewed at each reporting period, with any changes in fair value recorded in the statement of profit or loss in either operating results (e.g., for changes in internal forecasts and projections) or financial results (e.g., for changes in net present value), depending on the nature of the driver of the fair value adjustment.
2.19. Retirement benefit plans and employee savings plans
Payments to defined contribution employee savings plans are charged as an expense as they fall due. The Company does not offer nor operate any material defined benefit plans for its employees.
2.20. Share-based compensation plans for personnel, directors and business associates
The Company grants stock options in accordance with several share-based compensation plans in consideration for services performed by personnel, directors and business associates. The cost of the services rendered is measured at the fair value of the granted options and recognized as an expense in the statement of profit or loss. The corresponding credit is recorded directly into equity.
The estimate of the number of options which will ultimately vest is revised at each reporting date. The change in estimate is recorded as an expense with a corresponding correction in equity.
The received amount, less directly attributable transaction costs, will be recorded as share capital and share premium when the options are exercised.
F-16
NOTE 3: Business combination
Acquisition of Genomic Prostate Score® (GPS) test (formerly Oncotype DX GPS) from Exact Sciences
On August 2, 2022, the Company announced it has entered into an agreement with Genomic Health, Inc., a subsidiary of Exact Sciences Corporation (“Exact Sciences”), to acquire the GPS test from Exact Sciences. MDxHealth acquired GPS in order to expand its menu of tests targeted into urology and prostate cancer and in order to position the Company as one of the leaders in the urology and prostate cancer space with one of the most comprehensive menus of precision diagnostics.
Under
the terms of the agreement, the Company acquired the GPS prostate cancer business of Exact Sciences for an aggregate purchase price of
up to $
At
the option of MDxHealth, the earnout amounts can be settled in cash or through the issuance of additional shares of the Company (valued
in function of a volume weighted average trading price of the Company’s shares at the end of the relevant earnout period) to Exact
Sciences, provided that the aggregate number of shares held by Exact Sciences shall not exceed more than
The Acquisition was accounted for under the acquisition method of accounting and was being treated as a business combination in accordance with IFRS given that there are inputs from the intellectual property and customers acquired, a substantive process, consisting of a workforce that was hired from Exact Sciences, which allows the Company to generate outputs as from day 1 of the acquisition. The purchase price was allocated based on the estimated fair value of net assets acquired and liabilities assumed at the date of the acquisition.
| Cash | ||||
| Stock | ||||
| GPS Contingent consideration | ||||
| Total acquisition consideration |
The
purchase price in excess of the fair value of net assets acquired, has been considered as residual Goodwill for an amount of $
| Thousands of $ | Carrying value at acquisition date | Fair value adjustments | Fair value at acquisition date | |||||||||
| Intangible assets IP / Brand | ||||||||||||
| Intangible assets Customer relationships | ||||||||||||
| Total identified assets | ||||||||||||
| Goodwill | ||||||||||||
| Acquisition price | ||||||||||||
We have performed a fair value analysis of the business combination, with corresponding adjustments to the intangible assets.
The
accounting for the business combination resulted in fair values at date of acquisition of $
Following
the closing, an additional aggregate earnout amount of up to $
F-17
The net deferred tax asset resulting from this purchase price allocation was not recognized given insufficient future taxable profits. The recognized goodwill is expected to be fully tax deductible upon actual payment of the contingent consideration.
The
total acquisition-related costs recognized as an expense in general and administrative expenses for the year ended December 31, 2022,
were $
The GPS acquisition contributed $
The
Company financed the acquisition in part through a $
On
August 23, 2023, MDxHealth and Exact Sciences Corporation amended their existing Oncotype DX GPS prostate cancer business asset purchase
agreement, deferring the Company’s initial earnout payment by 3 years, from 2024 to 2027, in consideration for an amendment fee
of $
The
Company agreed to convene a general shareholders’ meeting to approve the subscription right. Under the terms of the amended asset
purchase agreement, MDxHealth has agreed to make earnout payments to Exact Sciences in each of fiscal years 2025, 2026 and 2027, based
upon certain revenues related to fiscal years 2024, 2025 and 2023, respectively. At the option of MDxHealth, the earnout amounts can
be settled in cash or through the issuance of additional shares of the Company (valued in function of a volume weighted average trading
price of the Company’s shares at the end of the relevant earnout period) to Exact Sciences, provided that the aggregate number
of shares held by Exact Sciences shall not exceed more than
NOTE 4: Revenue and cost of sales
Revenue
| Thousands of $ For the years ended December 31 |
2023 |
2022 |
2021 | |||||||||
| Services | ||||||||||||
| Royalties and other revenues | ||||||||||||
| Total revenue | ||||||||||||
Revenues related to royalties, licenses and other revenues are generally recognized over time as described in Note 2.7.
The Company did not recognize any contract assets or contracts liabilities.
Total
revenue for 2023 was $
Medicare
Reimbursement for diagnostic tests furnished to Medicare beneficiaries (typically patients aged 65 or older) is usually based on a fee schedule set by the U.S. Centers for Medicare & Medicaid Services (“CMS”), a division of the U.S. Department of Health and Human Services (“HHS”). As a Medicare-enrolled service provider, the Company bills the regional Medicare Administrative Contractor (“MAC”) for CMS that covers the region where the testing service is performed by the Company. The Confirm mdx test obtained a positive Medicare local coverage determination (“LCD”) in 2014, the GPS test obtained a positive Medicare coverage LCD in 2015, and the Select mdx test obtained a positive Medicare coverage LCD in 2023, each of which provides coverage for Medicare patients throughout the United States.
In
2023, Medicare represented the only payer generating over
At the end of 2023, the Company had concluded agreements with 140 commercial payors for Confirm mdx (2022: 129; 2021: 119), 84 commercial payors for Select mdx (2022: 62; 2021: 54) and 62 commercial payors for GPS (2022:29; 2021: 0).
F-18
Segment revenue
The Company does not distinguish different business segments since most revenues are generated from clinical laboratory service testing, or the out-licensing of the Company’s patented DNA methylation platform and biomarkers. However, the Company does distinguish different geographical operating segments based on revenue since the revenues are generated both in United States of America and Europe.
In
2023, the Company earned
| Thousands of $ For the years ended December 31 | 2023 | 2022 | 2021 | |||||||||
| United States of America | ||||||||||||
| Europe | ||||||||||||
| Rest of the world | ||||||||||||
| Total segment revenue | ||||||||||||
At
the end of 2023,
Cost of sales
| Thousands of $ For the years ended December 31 |
2023 | 2022 |
2021 | |||||||||
| Cost of sales | ||||||||||||
| Total cost sales | ||||||||||||
The costs of sales include the costs associated with providing testing services to third parties and include the cost of materials, labor (including salaries, bonuses, and benefits), transportation, collection kits, and allocated overhead costs associated with processing samples. Allocated overhead costs include depreciation of laboratory equipment, facility occupancy and information technology costs. Costs associated with processing samples are expensed when incurred, regardless of the timing of revenue recognition. Amortization of intangible assets are excluded from cost of sales and are presented separately in the statement of profit or loss, as further detailed in Note 5.
NOTE 5: Nature of expenses
| Thousands of $ For the years ended December 31 |
Notes |
2023 |
2022 |
2021 | ||||||||||
| Personnel costs | 6 | |||||||||||||
| Depreciation | 12 | |||||||||||||
| Impairment | 11 | |||||||||||||
| Lab consumables | ||||||||||||||
| Patent expenses | ||||||||||||||
| External collaborator fees | ||||||||||||||
| Clinical validation | ||||||||||||||
| Other expenses | ||||||||||||||
| Total research and development expenses | ||||||||||||||
Research and development expenses consist of costs incurred for the development and improvement of our products. These expenses consist primarily of labor costs (including salaries, bonuses, benefits, and share-based compensation), reagents and supplies, clinical studies, outside services, patent expenses, depreciation of laboratory equipment, facility occupancy and information technology costs. Research and development expenses also include costs associated with assay improvements and automation workflow for our current suite of products.
For
the year ended December 31, 2023, research and development expenses increased by $
F-19
For
the year ended December 31, 2022, research and development expenses decreased $
| Thousands of $ For the years ended December 31 | Notes |
2023 |
2022 |
2021 | ||||||||||
| Personnel costs | 6 | |||||||||||||
| Depreciation | 12 | |||||||||||||
| Professional fees | ||||||||||||||
| Marketing expenses | ||||||||||||||
| Travel expenses | ||||||||||||||
| Offices & facilities expenses | ||||||||||||||
| Other expenses | ||||||||||||||
| Total selling and marketing expenses | ||||||||||||||
The Company’s selling and marketing expenses are expensed as incurred and include costs associated with its sales organization, including its direct clinical sales force and sales management, medical affairs, client services, marketing and managed care, as well as technical lab support and administration. These expenses consist primarily of labor costs (including salaries, bonuses, benefits, and share-based compensation), customer education and promotional expenses, market analysis expenses, conference fees, travel expenses and allocated overhead costs.
For
the year ended December 31, 2023, selling and marketing expenses increased by $
For
the year ended December 31, 2022, selling and marketing expenses increased $
| Thousands of $ For the years ended December 31 | Notes |
2023 |
2022 |
2021 | ||||||||||
| Personnel costs | 6 | |||||||||||||
| Depreciation | 12 | |||||||||||||
| Professional fees | ||||||||||||||
| Public company expenses | ||||||||||||||
| Travel expenses | ||||||||||||||
| Offices & facilities expenses | ||||||||||||||
| Royalties to third parties | ||||||||||||||
| Board fees | ||||||||||||||
| Other expenses | ||||||||||||||
| Total general and administrative expenses | ||||||||||||||
General and administrative expenses include costs for certain executives, accounting and finance, legal, revenue cycle management, information technology, human resources, and administrative functions. These expenses consist primarily of labor costs (including salaries, bonuses, benefits, and share-based compensation), professional service fees such as consulting, accounting, legal, general corporate costs, and public-company costs associated with the Company’s listing, as well as allocated overhead costs (rent, utilities, insurance, etc.).
General
and administrative expenses decreased in 2023 by $
General
and administrative expenses increased in 2022 by $
F-20
| Thousands of $ For the years ended December 31 |
2023 |
2022 |
2021 | |||||||||
| Research and development | ||||||||||||
| Selling and marketing | ||||||||||||
| General and administrative | ||||||||||||
| Total amortization of intangible assets | ||||||||||||
Amortization of intangible assets primarily relates to the acquired intellectual property, brand, and customer relationships of the GPS business combination, as detailed in Note 3.
In 2023, the Company segregated “amortization of intangible assets” from other operating categories in the statement of profit or loss and is presenting amortization of intangible assets as a separate category. Prior periods balances have been reclassified to conform to current period presentation.
NOTE 6: Personnel costs
| Thousands of $ For the years ended December 31 |
2023 |
2022 |
2021 | |||||||||
| The number of employees at the end of the year was: | ||||||||||||
| Laboratory operations | ||||||||||||
| R&D staff | ||||||||||||
| S&M staff | ||||||||||||
| G&A staff | ||||||||||||
| Total number of employees | ||||||||||||
| Their aggregate remuneration comprised: | ||||||||||||
| Wages and salaries | ||||||||||||
| Social security costs | ||||||||||||
| Pension costs | ||||||||||||
| Health insurance expenses | ||||||||||||
| Share-based compensation | ||||||||||||
| Other costs | ||||||||||||
| Total personnel costs | ||||||||||||
The personnel numbers in the table reflect year-end numbers, with 42 sales and marketing employees hired in August 2022 as part of the GPS acquisition.
NOTE 7: Other operating (expense) income, net
| Thousands of $ For the years ended December 31 |
2023 |
2022 |
2021 | |||||||||
| Grant subsidies – The Netherlands | ||||||||||||
| Grant subsidies – USA | ||||||||||||
| Fair value adjustments | ( | ) | ||||||||||
| Other operating income | ||||||||||||
| Other operating expenses | ( | ) | ||||||||||
| Total other operating (expense) income, net | ( | ) | ||||||||||
Other
operating (expense) income, net for the year ended December 31, 2023, primarily consisted of a negative fair value adjustment of $
Other
operating (expense) income, net for the year ended December 31, 2022, primarily consisted of a positive fair value adjustment of $
Other
operating (expense) income, net for the year ended December 31, 2021, primarily consisted of a $
F-21
NOTE 8: Financial income and expense
| Thousands of $ For the years ended December 31 |
2023 |
2022 |
2021 | |||||||||
| Interest income | ||||||||||||
| Innovatus derivative instrument | ||||||||||||
| Fair value of option to pay GPS earnout in shares | ||||||||||||
| Financial income, net | ||||||||||||
| Thousands of $ For the years ended December 31 |
2023 |
2022 |
2021 | |||||||||
| Interest on Kreos loan | ( | ) | ( | ) | ||||||||
| Interest on Innovatus loan | ( | ) | ( | ) | ||||||||
| Interest on other loans and leases | ( | ) | ( | ) | ( | ) | ||||||
| Kreos settlement | ( | ) | ||||||||||
| Fair value adjustments | ||||||||||||
| GPS contingent consideration | ( | ) | ( | ) | ||||||||
| NovioGendix contingent consideration | ( | ) | ( | ) | ( | ) | ||||||
| Kreos derivative instrument | ( | ) | ( | ) | ||||||||
| Exact Sciences | ( | ) | ||||||||||
| Other financial loss | ( | ) | ( | ) | ( | ) | ||||||
| GPS amendment: additional consideration payment in cash | ( | ) | ||||||||||
| GPS amendment: additional consideration payment in shares | ( | ) | ||||||||||
| Financial expenses, net | ( | ) | ( | ) | ( | ) | ||||||
For
the year ended December 31, 2023, financial expenses, were primarily comprised of a negative fair value adjustment for the GPS contingent
consideration of $
Other financial loss relates to bank costs incurred during the year.
NOTE 9: Income Tax
No
income taxes were payable in view of the losses incurred by the Company. On December 31, 2023, the Company had a consolidated net tax
loss carried forward amounting to $
The
tax losses related to MDxHealth SA in Belgium are available for carry forward. Until
Tax credits
(investment deductions) amounted to $
F-22
It
is uncertain if the Company will have taxable profits in the near future to allow all or part of the deferred tax asset to be utilized
and as a result, no deferred tax asset was recognized in 2023, 2022, and 2021.
| Thousands of $ | Income Statement | |||||||||||
| For the years ended December 31 | 2023 | 2022 | 2021 | |||||||||
| Loss for the year | ( | ) | ( | ) | ( | ) | ||||||
| Income tax expense | ( | ) | ||||||||||
| Loss before income tax | ( | ) | ( | ) | ( | ) | ||||||
| Tax using the MDxHealth’s domestic tax rate | ||||||||||||
| Effect of unused tax losses not recognized as deferred tax assets | ( | ) | ( | ) | ( | ) | ||||||
NOTE 10: Goodwill
On
August 2, 2022, the Company acquired the GPS test from Exact Sciences (refer to Note 3 for further details). The purchase price in excess
of the fair value of the net assets acquired has been considered as residual goodwill for an amount of $
The Company is required to test Goodwill for impairment on an annual basis. The recoverable amount is determined based on a value-in-use calculation. The use of this method requires the estimation of future cash flows and the determination of a discount rate in order to calculate the present value of the cash flows.
The
company monitors its Goodwill at the consolidated company level, which is the level of its cash generating unit (CGU) benefiting from
the synergies. The recoverable amount of the CGU has been determined from the value-in-use calculation based on the Company’s cash
flow projections covering a period of
The
amount by which the CGU’s recoverable value exceeds its carrying value is $
| Thousands of $ | Goodwill | |||
| At January 1, 2022 | ||||
| Additions through business combination | ||||
| Impairment | ||||
| Currency translation adjustments | ||||
| Carrying value at December 31, 2022 | ||||
| At January 1, 2023 | ||||
| Additions through business combination | ||||
| Impairment | ||||
| Currency translation adjustments | ||||
| Carrying value at December 31, 2023 | ||||
| Assumptions used | December 31, 2023 | |||
| Discount rate (post-tax) | % | |||
| Terminal growth rate | % | |||
The discount rate is based on comparable companies in the industry together with company-specific risks. Terminal growth rate is based on management estimates and industry data.
The Company’s impairment review is sensitive to changes in the assumptions used, most notably the discount rate and the terminal growth rate.
An
increase of
Based on the above information, management concluded that there is no Goodwill impairment in 2023.
F-23
NOTE 11: Intangible assets
| Thousands of $ | Patents and Software licenses | Internally developed intangible assets | Externally acquired Intellectual property | Customers | TOTAL | |||||||||||||||
| Gross value | ||||||||||||||||||||
| At January 1, 2022 | ||||||||||||||||||||
| Additions | ||||||||||||||||||||
| Additions through business combination (Note 3) | ||||||||||||||||||||
| Currency translation adjustments | ||||||||||||||||||||
| Gross value at December 31, 2022 | ||||||||||||||||||||
| Accumulated amortization and impairment At January 1, 2022 | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||||
| Additions | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||
| Impairment | ( | ) | ( | ) | ||||||||||||||||
| Accumulated amortization and impairment at December 31, 2022 | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||
| Net value at December 31, 2022 | ||||||||||||||||||||
| Gross value | ||||||||||||||||||||
| At January 1, 2023 | ||||||||||||||||||||
| Additions | ||||||||||||||||||||
| Gross value at December 31, 2023 | ||||||||||||||||||||
| Accumulated amortization and impairment At January 1, 2023 | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||
| Additions | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||||
| Currency translation adjustments | ||||||||||||||||||||
| Accumulated amortization and impairment at December 31, 2023 | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||
| Net value at December 31, 2023 | ||||||||||||||||||||
Amortization of intangible assets is included as a separate line in the statement of profit or loss.
The
externally acquired intangible assets include technology acquired in the business combination with NovioGendix in 2015 and with the acquisition
of the GPS test in August 2022, and increased by $
Customer
relationships includes customers acquired in the GPS acquisition, resulting in the fair value at acquisition date of $
The
internally-developed intangible assets relate to the capitalized development expenses for Confirm mdx and Select mdx over the past years
as well as for the development of the GPS assay in-house and our Resolve mdx assay. The estimated remaining amortization period amounts
to
F-24
NOTE 12: Property, plant and equipment and right of-use assets
Property, plant and equipment
| Thousands of $ | Laboratory equipment |
Furniture | IT equipment | Leasehold improvements |
TOTAL | |||||||||||||||
| Gross value | ||||||||||||||||||||
| At January 1, 2022 | ||||||||||||||||||||
| Additions | ||||||||||||||||||||
| Disposals | ( | ) | ( | ) | ||||||||||||||||
| Exchange rate difference arising | ( | ) | ( | ) | ||||||||||||||||
| Gross value at December 31, 2022 | ||||||||||||||||||||
| Accumulated depreciation At January 1, 2022 | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||
| Additions | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||
| Disposals | ||||||||||||||||||||
| Exchange rate difference arising | ( | ) | ( | ) | ||||||||||||||||
| Accumulated depreciation at December 31, 2022 | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||
| Net value at December 31, 2022 | ||||||||||||||||||||
| Gross value | ||||||||||||||||||||
| At January 1, 2023 | ||||||||||||||||||||
| Additions | ||||||||||||||||||||
| Disposals | - | ( | ) | ( | ) | |||||||||||||||
| Transfer from leases | ||||||||||||||||||||
| Exchange rate difference arising | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||
| Gross value at December 31, 2023 | ||||||||||||||||||||
| Accumulated depreciation At January 1, 2023 | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||
| Additions | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||
| Disposals | ||||||||||||||||||||
| Transfer from leases | ( | ) | ( | ) | ||||||||||||||||
| Exchange rate difference arising | ||||||||||||||||||||
| Accumulated depreciation at December 31, 2023 | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||
| Net value at December 31, 2023` | ||||||||||||||||||||
F-25
During
2023, the Company acquired $
Right of-use assets
| Thousands of $ | buildings | vehicles | Equipment | TOTAL | ||||||||||||
| Gross value | ||||||||||||||||
| Balance at January 1, 2022 | ||||||||||||||||
| Additions | ||||||||||||||||
| Exchange rate differences | ( | ) | ( | ) | ||||||||||||
| Gross value at December 31, 2022 | ||||||||||||||||
| Accumulated depreciation | ||||||||||||||||
| Balance at January 1, 2022 | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||
| Additions | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||
| Exchange rate difference | ( | ) | ( | ) | ||||||||||||
| Accumulated depreciation on December 31, 2022 | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||
| Net value at December 31, 2022 | ||||||||||||||||
| Gross value | ||||||||||||||||
| Balance at January 1, 2023 | ||||||||||||||||
| Additions | ||||||||||||||||
| Disposals | ( | ) | ( | ) | ( | ) | ||||||||||
| Transfer to tangible assets | ( | ) | ( | ) | ||||||||||||
| Gross value on December 31, 2023 | ||||||||||||||||
| Accumulated depreciation | ||||||||||||||||
| Balance at January 1, 2023 | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||
| Additions | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||
| Disposals | ||||||||||||||||
| Transfer to tangible assets | ||||||||||||||||
| Exchange rate differences | ||||||||||||||||
| Accumulated depreciation on December 31, 2023 | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||
| Net value at December 31, 2023 | ||||||||||||||||
F-26
In
June 2022, the company entered into a 36-month lease agreement (the “Plano lease”) for approximately
In October 2022, the company renewed its lease agreement for a term of 60 months for its facilities in Nijmegen, The Netherlands.
The
new lease agreements from 2023 represent additional right of use assets of a total value of $
| Thousands of $ | 2023 | 2022 | 2021 | |||||||||
| Depreciation expense | ||||||||||||
| Interest expense on lease liabilities | ||||||||||||
NOTE 13: Inventories
| Thousands of $ For the years ended December 31 | 2023 | 2022 | ||||||
| Raw materials and consumables | ||||||||
| Total Inventories | $ | $ | ||||||
Inventories
are recognized at the lower of cost or net realizable value. Inventories recognized as an expense during the year ended December 31,
2023, amounted to $
NOTE 14: Trade and other receivables
Trade receivables
| Thousands of $ For the years ended December 31 |
2023 |
2022 | ||||||
| Trade receivables | ||||||||
| Total trade receivables | ||||||||
Trade receivables mainly consist of claims due from our patients’ insurance companies related to our diagnostic tests.
F-27
| A/R by claim date | Months | |||||||||||||||||||
| Thousands of $ For the year ended December 31, 2023 | 1-3 months | 4-6 months | 7-12 months | Not due | Total | |||||||||||||||
| Confirm mdx | - | |||||||||||||||||||
| Select mdx | (8 | ) | ||||||||||||||||||
| Resolve mdx | - | |||||||||||||||||||
| GPS | - | |||||||||||||||||||
| Total Trade Receivables | (8 | ) | ||||||||||||||||||
| A/R by claim date | Months | |||||||||||||||||||
| Thousands of $ For the year ended December 31, 2022 | 1-3 months | 4-6 months | 7-12 months | Not due | Total | |||||||||||||||
| Confirm mdx | ||||||||||||||||||||
| Select mdx | ||||||||||||||||||||
| Resolve mdx | ||||||||||||||||||||
| GPS | ||||||||||||||||||||
| Other | ||||||||||||||||||||
| Total Trade Receivables | ||||||||||||||||||||
Prepaid expenses and other current assets
Thousands
of $ |
2023 |
2022 | ||||||
| Prepayments | ||||||||
| Deposits | ||||||||
| Recoverable VAT | ||||||||
| Grants to be received | ||||||||
| Other | ||||||||
| Total prepaid expenses and other current assets | $ | $ | ||||||
Prepaid expenses mainly consist of prepaid insurance premiums and prepaid maintenance contracts.
All financial assets carried at amortized cost are shown net of expected credit losses which are not deemed material.
NOTE 15: Cash and cash equivalents
| Thousands of $ For the years ended December 31 |
2023 |
2022 | ||||||
| Cash and cash equivalents | ||||||||
| Total cash and cash equivalents | $ | $ | ||||||
The bank balances and cash held by the Company and short-term bank deposits have an original maturity of less than 3 months. The carrying amount of these assets approximates their fair value.
The Company had no restricted cash in 2023 and 2022.
F-28
NOTE 16: Loans, Borrowings, Leases obligations and other financial liabilities
| Thousands of $ For the years ended December 31 |
2023 |
2022 | ||||||
| Non-current loans and borrowings | ||||||||
| Loans | ||||||||
| Lease liabilities (*) | ||||||||
| Total non-current loans and borrowings | $ | $ | ||||||
| Thousands of $ For the years ended December 31 |
2023 |
2022 | ||||||
| Current loans and borrowings | ||||||||
| Loans | ||||||||
| Lease liabilities (*) | ||||||||
| Total current loans and borrowings | $ | $ | ||||||
| (*) |
Innovatus debt facility
On August 2, 2022, the Company entered into a
$
Security has been granted over all assets (including IP rights) owned by the Company and MDxHealth, Inc. The loan agreement contains customary financial covenants and general affirmative and negative covenants, including limitations on the Company’s ability to transfer or dispose of assets, change our business, merge with or acquire other companies, incur additional indebtedness and liens, make investments, pay dividends and conduct transactions with affiliates.
The
Innovatus debt facility has been accounted for as a hybrid financial instrument which includes a host financial liability as well as
an embedded derivative financial instrument being an equity conversion call option at a fixed rate of up to
The embedded derivative is not considered to be closely related to the host financial liability given the differences in economics and risks, and as such both are accounted for separately:
| ● | The host financial liability is recognized at amortized cost applying the effective interest rate method and has been accounted for as non-current loans and borrowings; |
| ● | The embedded derivative convertible (American) call option is recognized at fair value using a binomial tree option pricing model whereby the fair value is based on the actual stock price and the estimated volatility of the Company’s shares on Nasdaq since the Company’s IPO on November 4, 2021, and through the valuation date. The volatility measured on August 2, 2022, which was the closing date of the Innovatus debt facility, was |
F-29
Kreos debt facility
As
part of the new debt facility with Innovatus, the Company’s debt facility with Kreos for an outstanding principal amount of €
The
2022 repayment did not include the derivative financial liability for the initial Kreos drawdown fee which had an estimated fair value
on December 31, 2022, of $
In
June 2023, Kreos provided notice to the Company of the cancelation of the convertible loan associated with the initial drawdown fee and
requested repayment in cash. As such, Kreos was entitled to a cash repayment of €
On
April 20, 2020, the Company, through its U.S. subsidiary, MDxHealth Inc., has entered into a “Paycheck Protection Program”
(PPP) loan with the U.S. Small Business Administration (SBA) in the amount of $
In addition to the contracted loans, the Company has several lease obligations. The leases have terms of 3 to 5 years.
| Thousands of $ For the years ended December 31 |
2023 |
2022 | ||||||
| Loans | ||||||||
| Within one year | ||||||||
| Years two to five | ||||||||
| Leases | ||||||||
| Within one year | ||||||||
| Years two to five | ||||||||
Note: all figures shown in this table are undiscounted and reflect future cash payments (capital and interests)
Other financial liabilities
| Thousands of $ For the years ended December 31 |
2023 |
2022 | ||||||
| Other financial liabilities | ||||||||
| Other non-current financial liabilities | ||||||||
| Other current financial liabilities | ||||||||
| Total other financial liabilities | ||||||||
GPS Contingent consideration
As
part of the acquisition of the GPS business from Exact Sciences in August 2022, and the subsequent amended asset purchase agreement from
August 2023, an aggregate earnout amount of up to $
MDxHealth option to settle earnout obligation in shares
The
fair value of the Company’s option to settle the earnout obligation in cash or through the issuance of additional shares of the
Company, was measured using a Monte Carlo valuation model which takes into account several factors including the expected evolution in
Company’s share price. This valuation model is considered as level 3 input and was assessed at $
F-30
Exact
Sciences
The
fair value of the warrant held by Exact Sciences to acquire up to
Innovatus embedded derivative convertible call option
The
embedded derivative convertible (American) call option is recognized at fair value within other current financial liabilities and is
measured using a Binomial tree valuation model which takes into account several factors including the expected evolution in the Company’s
share price. The fair value of the liability is estimated at $
Kreos derivative financial instrument (“initial drawdown fee”)
As of December 31, 2023, the convertible loan associated with the initial drawdown fee payable to Kreos has been fully off in cash and as such, the fair value of the financial derivative has been reduced to zero as no further liabilities to Kreos exist.
As
of December 31, 2022, the fair value of the financial derivative related to the initial drawdown fee of the Kreos loan was computed as
the sum of the probability-weighted values of the fair values associated with each of the possible outcomes and amounted to $
Other financial liabilities
Other
financial liabilities include the contingent consideration related to the acquisition of NovioGendix in 2015 and amounts to $
Thousands of $ | Loans and borrowings | Other financial liabilities | ||||||||||||||
| For the years ended December 31 | 2023 | 2022 | 2023 | 2022 | ||||||||||||
| Beginning balance | ||||||||||||||||
| Cash movements | ||||||||||||||||
| Loans and borrowings repaid1 (Kreos / PPP) | ( | ) | ( | ) | ( | ) | ||||||||||
| Loans and borrowings received (Innovatus) | ||||||||||||||||
| Non-cash movements | ||||||||||||||||
| GPS Contingent Consideration | ||||||||||||||||
| Recognition of Innovatus embedded derivative convertible call option | ( | ) | ||||||||||||||
| Kreos effective interest rate adjustment and extinguishment costs | ||||||||||||||||
| Innovatus - effective interest rate adjustment | ||||||||||||||||
| Foreign exchange rate impact / other | ( | ) | ( | ) | ( | ) | ||||||||||
| Fair value changes through profit and loss | ||||||||||||||||
| Ending balance | $ | $ | $ | $ | ||||||||||||
| 1 |
| Thousands of $ For the years ended December 31 |
2023 | |||
| Increase of NovioGendix contingent consideration | ||||
| Increase of Kreos derivative financial instrument (“initial drawdown fee”) | ||||
| Increase of GPS contingent consideration | ||||
| Decrease of Innovatus embedded derivative convertible call option | ( | ) | ||
| Exact Sciences | ||||
| Total fair value adjustment | ||||
F-31
| Thousands of $ | Lease liabilities | |||||||
| For the years ended December 31 | 2023 | 2022 | ||||||
| Opening balance | ||||||||
| Cash movements | ||||||||
| Repayment of lease liabilities | ( | ) | ( | ) | ||||
| Non-cash movements | ||||||||
| Interest accretion | ||||||||
| New leases | ||||||||
| Closing balance | ||||||||
NOTE 17: Contractual obligations
| Thousands of $ For The Years ended December 31 |
2023 |
2022 | ||||||
| Outstanding commitments for future minimum rent payments, which fall due as follows: | ||||||||
| Less than one year | ||||||||
| Years 2-5 | ||||||||
| Total contractual obligations | ||||||||
For 2023 and 2022, we refer to Note 12 and 16 for the lease liabilities subsequent adoption and application of IFRS 16.
Outstanding
commitments for future minimum rent payments include rental fees related to leased facilities, and equipment for assets with a value
below $
NOTE 18: Trade and other payables
Trade accounts payable
| Thousands of $ For The Years ended December 31 |
2023 |
2022 | ||||||
| Trade accounts payable | ||||||||
| Accruals for invoices to be received | ||||||||
| Total trade accounts payable | ||||||||
Other current liabilities
| Thousands of $ For The Years ended December 31 |
2023 |
2022 | ||||||
| Payroll | ||||||||
| Other accruals | ||||||||
| Total other current liabilities | ||||||||
F-32
NOTE 19: Financial instruments and fair value
The table shows the Company’s significant financial assets and liabilities. All financial assets and liabilities are carried at amortized cost with the exception of the contingent considerations in relation to acquisitions and derivative financial instruments reported at fair value through the statement of profit or loss.
| Thousands of $ For The Years ended December 31 |
2023 |
2022 | Fair value hierarchy | |||||||
| Assets | ||||||||||
| At amortized cost | ||||||||||
| Trade receivables | ||||||||||
| Cash and cash equivalents | ||||||||||
| Total financial assets | ||||||||||
| Liabilities | ||||||||||
| At fair value: | ||||||||||
| Other financial liabilities | ||||||||||
| GPS contingent consideration | Level 3 | |||||||||
| Exact Sciences | Level 3 | |||||||||
| NovioGendix contingent consideration | Level 3 | |||||||||
| Innovatus derivative instrument | Level 3 | |||||||||
| Kreos derivative instrument | Level 3 | |||||||||
| Subtotal financial liabilities at fair value | ||||||||||
| At amortized cost: | ||||||||||
| Loans and borrowings | Level 2 | |||||||||
| Lease liabilities | ||||||||||
| Trade payables | ||||||||||
| Subtotal financial liabilities at amortized cost | ||||||||||
| Total financial liabilities | ||||||||||
Recognized fair value measurements – valuation technique and principal inputs
The fair value of the financial instruments has been determined on the basis of the following methods and assumptions:
| ● | The carrying value of the cash and cash equivalents, the trade receivables, other current assets and the trade payables approximate their fair value due to their short-term character; |
| ● | The fair value of loans and borrowings applying the Effective Interest Rate method approximates their carrying value (level 2). |
| ● | Innovatus debt facility: the host financial liability was obtained with a variable interest rate based upon the Prime Rate (with a floor of |
| ● | Paycheck Protection Program (PPP): applying a market rate would not result in a materially different fair value which carries an interest rate of |
| ● | Kreos debt facility: Given full repayment of the convertible loan associated with the initial drawdown fee, no fair value assessment was performed as of December 31, 2023 |
| ● | Leases are measured at the present value of the remaining lease payments, using a discount rate based on the incremental borrowing rate at the commencement date of these leases. Their fair value approximates their carrying value. |
| ● | The fair value of contingent consideration payable to Exact Sciences (for the GPS acquisition) and NovioGendix (presented in the yearend statement of financial position under “other non-current financial liabilities” and “other current financial liabilities”) is based on an estimated outcome of the conditional purchase price/contingent payments arising from contractual obligations (level 3). This is initially recognized as part of the purchase price and subsequently fair valued with changes recorded through other operating income in the statement of profit or loss. |
| o | GPS: The fair value of the contingent consideration payable to Exact Sciences
is based on a probability-weighted average estimate based on multiple scenarios varying in timing and amount of earnout payment. This
probability-weighted estimate of a payout of $ |
F-33
| o | NovioGendix: the Company used a discount rate of |
| ● | The fair value of the derivative financial liabilities related to the
Innovatus derivative call option (as detailed in Note 16) was performed using a binomial pricing model which takes into account several
factors including the expected evolution in share price and are considered as level 3 input. The fair value of the liability is estimated
at $ |
| ● | Exact Sciences |
| ● | Financial instruments are evaluated based on the mark-to-market report and the unrealized gains (loss) are recognized through the statement of profit or loss. |
Fair value hierarchy:
The Company uses the following hierarchy for determining and disclosing the fair value of financial instruments by valuation technique:
| ● | Level 1: quoted prices in active markets for identical assets and liabilities; |
| ● | Level 2: other techniques for which all inputs which have a significant effect on the recorded fair value are observable, either directly or indirectly; and |
| ● | Level 3: techniques which use inputs that have a significant effect on the recorded fair value that are not based on observable market data. |
No financial assets or financial liabilities have been reclassified between the valuation categories during the year.
| Thousands of $ | Financial Derivative Instruments (Kreos and Innovatus) | Contingent Consideration (NovioGendix and GPS) | ||||||||||||||
| For the years ended December 31 | 2023 | 2022 | 2023 | 2022 | ||||||||||||
| Beginning balance | ||||||||||||||||
| Cash movements | ||||||||||||||||
| Loans and borrowings repaid | ( | ) | ( | ) | ||||||||||||
| Non-cash movements | ||||||||||||||||
| GPS contingent consideration | ||||||||||||||||
| Exact Sciences | ||||||||||||||||
| Innovatus embedded derivative convertible call option | ||||||||||||||||
| Effective interest rate adjustment | ( | ) | ||||||||||||||
| Foreign exchange rate impact / other movements | ( | ) | ||||||||||||||
| Fair value changes through profit and loss | ( | ) | ||||||||||||||
| Change to level 1 fair value hierarchy | ||||||||||||||||
| Ending balance | ||||||||||||||||
NOTE 20: Loss per share
The basic loss per share is calculated by dividing the net result attributable to shareholders by the weighted average number of shares outstanding during the year, adjusted for the 1-for-10 reverse stock split that took place in November 2023.
| Years ended December 31 | 2023 | 2022 | 2021 | |||||||||
| Loss for the year, in thousands of $ | ( | ) | ( | ) | ( | ) | ||||||
| ( | ) | ( | ) | ( | ) | |||||||
| Weighted average number of shares | 2023 | 2022 | 2021 | ||||||||||
At December 31, 2023, 2022, and 2021, the Company had potential dilutive shares in the form of warrants, contingent considerations and convertible loans (see Note 16 and Note 24 for further details). Diluted loss per share considers the impact of potentially dilutive securities except in periods in which there is a loss because the inclusion of the potential common shares would have an anti-dilutive effect.
F-34
NOTE 21: Financial Risk Management
Capital management
Capital is comprised of equity attributable to shareholders, borrowings, and cash and cash equivalents. The Company aims to maintain a strong capital base in order to maintain investor and creditor confidence and to sustain the future development of the business. The Company’s objectives when managing capital are to maintain sufficient liquidity to meet its working capital requirements, fund capital investment and purchases, and safeguard its ability to continue operating as a going concern. The Company monitors capital regularly to ensure that the statutory capital requirements are met and may propose capital increases at shareholders’ meetings to ensure the necessary capital remains intact.
Credit risk
Credit risk arises from cash and cash equivalents, short-term bank deposits, as well as credit exposure to collaboration partners. Credit risk refers to the risks that counterparty will default on its contractual obligations resulting in financial loss to the Company.
At
the end of 2023, the Company operated with more than
In the U.S. healthcare system, and particularly within the molecular diagnostic CLIA laboratory industry, where there are rapid technological advances in diagnostic services, companies provide services to healthcare professionals and their patients, while being reimbursed from commercial and governmental insurance systems. Often these services are provided out of network and without supplier contracts. As a result, there is reimbursement risk, separate from credit risk that is characterized by uncertainty in reimbursement value, delays in payment, and ultimately non-payment. This impacts the Company’s revenue recognition and cash collections.
In addition to reimbursement risk associated with commercial third-party payors, credit risk may also arise from amounts due directly from patients. In many cases, payors will cover the entire cost of testing. For example, for tests that fall under the Clinical Laboratory Fee Schedule, there is no co-payment, co-insurance or deductible for patients covered under traditional Medicare. However, patients covered by commercial insurance companies may be responsible for a co-payment, co-insurance, and/or deductible depending on the health insurance plan and individual patient benefit. Credit risk exists for those patients who cannot meet their co-payment or deductible portions.
Customers’
compliance with agreed credit terms is regularly and closely monitored. Trade accounts receivable amounted to $
The
credit risk on cash and cash equivalents of $
Interest risk
During
2022, the Company entered into a
In
addition, on April 20, 2020, the Company, through its U.S. subsidiary, MDxHealth Inc., has entered into a “Paycheck Protection
Program” (PPP) loan with the U.S. Small Business Administration (SBA) in the amount of $
Currency risk
The functional currency changed from the EURO to the U.S. Dollar as of July 1, 2014. Consequently, the currency risk is concentrated on European operations.
As
of December 31, 2023, cash deposits in EURO amounted to €
The
Company performed a sensitivity analysis of an increase/decrease of exchange rate on operations of
F-35
Liquidity risk
The
Company manages liquidity risk by maintaining adequate reserves and by continuously monitoring forecast and actual cash flows and matching
the maturity profiles of financial assets and liabilities. At the date of this report, the Company has
For the years ended December 31, 2023 Thousands of $ |
Less than |
1-2 |
3-5 | Total contractual cash flows |
Carrying | |||||||||||||||
| Non derivatives | ||||||||||||||||||||
| Trade payables | ||||||||||||||||||||
| Loans | ||||||||||||||||||||
| Lease liabilities | ||||||||||||||||||||
| Total | ||||||||||||||||||||
For the years ended December 31, 2022 Thousands of $ | Less than 1 year | 1-2 years | 3-5 years | Total contractual cash flows | Carrying amount | |||||||||||||||
| Non derivatives | ||||||||||||||||||||
| Trade payables | ||||||||||||||||||||
| Loans | ||||||||||||||||||||
| Lease liabilities | ||||||||||||||||||||
| Total | ||||||||||||||||||||
Note: Except for carrying amount, all figures shown in this table are undiscounted and reflect future cash payments
The Company is also committed to a potential additional cash out of
an aggregate earnout amount of up to $
Other risks
The Company subscribes to certain insurance policies to cover matters such as (i) fire, theft, and other damage to its assets, (ii) product and liability insurance and clinical trial insurance, and (iii) D&O insurance. To date, no significant claims have been made under these insurance policies and there is no guarantee that the insurances will cover all damages if they should ever occur.
NOTE 22: Share capital and reserves
| For the Years ended December 31 | 2023 | 2022 | ||||||
| Common shares | ||||||||
| Total outstanding shares | ||||||||
On
August 11, 2022, to settle a portion of the purchase price for the acquisition by the Company of the GPS test from Genomic Health, Inc.
(a subsidiary of Exact Sciences Corporation) announced on August 2, 2022, the Company issued
In
March 2023, the Company completed a registered public offering of
On
August 23, 2023, the Company and Exact Sciences Corporation amended their existing Oncotype DX GPS prostate cancer business asset purchase
agreement, deferring the Company’s initial earnout payment by
F-36
| Thousands of $ | Thousands of € | |||||||||||||||
| For the Years ended December 31 | Share Capital | Issuance Premium | Share Capital | Issuance Premium | ||||||||||||
| As of January 1, 2022 | ||||||||||||||||
| August 2022 – Issuance of | ||||||||||||||||
| As of December 31, 2022 | ||||||||||||||||
| March 2023 – Issuance of | ||||||||||||||||
| August 2023 – Issuance of | ||||||||||||||||
| As of December 31, 2023 | ||||||||||||||||
| (*) |
| Thousands of $ | Thousands of € | |||||||||||||||
| For the Years ended December 31 | 2023 | 2022 | 2023 | 2022 | ||||||||||||
| Share Capital as per statutory accounts | ||||||||||||||||
| Capital increase costs | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||
| Share capital under IFRS | ||||||||||||||||
| Issuance premium | ||||||||||||||||
| Share capital and issuance premium | ||||||||||||||||
The history of the Share Capital can be found in “General Information; Capital and Shares”.
By virtue of the resolution of the extraordinary general shareholders’ meeting of the Company held on May 27, 2021, which entered into force on June 1, 2021, the board of directors of the Company has been granted certain powers to increase the Company’s share capital in the framework of the authorized capital. The powers under the authorized capital have been set out in article 6 of the Company’s articles of association.
Pursuant
to the authorization granted by the extraordinary general shareholders’ meeting of June 30, 2023, the board of directors was authorized
to increase the share capital of the Company on one or several occasions by a maximum aggregate amount of €
The
board of directors has used its powers under the authorized capital on the occasion of the amendment of its agreement with Exact Sciences,
by issuing
In addition to the outstanding shares of the Company:
| ● | under the loan and security agreement entered into by the Company and Innovatus Capital Partners in August 2022, Innovatus has the right to convert, prior to August 2, 2025, up to 15% of the outstanding principal amount of the loans (by means of a contribution in kind of the relevant payables due by the Company under the loans) into shares of the Company at a conversion price per share equal to $11.21 (see note 16 for further details). |
F-37
NOTE 23: Retirement benefit plans
The Company operates defined contribution plans for all its qualifying employees. The assets of these plans are held separately from those of the Company in designated funds.
A
total cost of $
The employees of the Company in Belgium are members of a state-managed retirement benefit plan operated by the government (i.e., legal pension) and are members of a bank-operated private pension plan. The Company is required to contribute a specified percentage of payroll costs to the retirement benefit plan to fund the benefits. The obligation of the Company with respect to the retirement benefit plan is to make the specified contributions.
Because the Company must guarantee the statutory minimum return on these plans, not all actuarial and investment risks relating to these plans are transferred to the insurance company or pension fund managing the plans. The Company has considered the potential impact of the employer’s obligation to guarantee a minimum return and that this was assessed not to be significant.
NOTE 24: Share-based payments
Warrants
are granted to employees, consultants or directors of the Company and its subsidiaries. Each warrant entitles its holders to subscribe
to one new share of the Company at a subscription price determined by the board of directors, within the limits decided upon at the occasion
of their issuance. The warrants issued generally have a term of
On November 13, 2023, the company completed a share consolidation with respect to all its outstanding shares by means of a 1-for-10 reverse stock split (the “Share Consolidation”). Although the number of warrants does not change, the reverse stock split affects the number of shares into which the original number of warrants are convertible. Therefore, all share amounts were adjusted to reflect the Share Consolidation.
This section provides an overview of the outstanding warrants as of December 31, 2023. The warrants were created within the context of share-based incentive plans for employees, directors and consultants of the Company.
The
Company has created several pools of warrants under stock option plans for grant to eligible employees, directors, and consultants. Stock
option plans were announced on June 23, 2014 (
| Outstanding warrants | 2023 | 2022 | ||||||
| Warrants created | ||||||||
| Warrants available for grant | ( | ) | ( | ) | ||||
| Warrants granted | ||||||||
| Warrants terminated or lapsed | ( | ) | ( | ) | ||||
| Warrants exercised | ( | ) | ||||||
| Total outstanding at December 31 | ||||||||
As
of December 31, 2023, there are
For
the year 2023,
| Number of potential shares from outstanding warrants | 2023 | 2022 | ||||||
| As of January 1 | ||||||||
| Number of warrants cancelled/forfeited during the year | ( | ) | ( | ) | ||||
| Number of warrants granted during the year | ||||||||
| As of December 31 | ||||||||
F-38
| Thousands of $ Years ended December 31 |
2023 |
2022 |
2021 | |||||||||
| Share-based compensation in consolidated statement of profit or loss | ||||||||||||
| Cumulated Share-based compensation in equity | ||||||||||||
The Cumulated Share-based compensation amount is part of the Total Shareholders’ Equity on the Consolidated statement of financial position. This amount is presented on the Consolidated statement of financial position for both exercised and non-exercised warrants.
In
general, the warrants vest in cumulative tranches of
| ● | The warrants granted to directors under the June 23, 2014 Stock Option Plan, the June 21, 2019 Stock Option Plan, the May 27, 2021 Stock Option Plan, the May 25, 2022, and the June 30, 2023 Stock Option Plan, all vest on the date of the annual meeting that takes place in the calendar year following the calendar year in which they were granted, provided that the mandate of the relevant director has not ended or been terminated. |
| ● | The warrants granted to beneficiaries who are not directors under the June 23, 2014 Stock Option Plan all vest in instalments of |
| ● | The warrants granted to beneficiaries who are not directors under the June 21, 2019 Stock Option Plan, the May 27, 2021 Stock Option Plan, the May 25, 2022, and the June 30, 2023 Stock Option Plan may adopt a manual or custom vesting procedure under certain conditions or a particular vesting period over 3 or 4 years. |
Warrants |
Weighted |
Potential | Weighted average exercise price per potential share (€) | |||||||||||||
| Granted in 2022 | ||||||||||||||||
| Outstanding at December 31, 2022 | ||||||||||||||||
| Granted in 2023 | ||||||||||||||||
| Outstanding at December 31, 2023 | ||||||||||||||||
| Exercisable at December 31, 2023 | ||||||||||||||||
| Category | 2023 | 2022 | ||||||
| Executive Director | ||||||||
| Non-Executive Directors | ||||||||
| Management team (excluding the Executive Director) | ||||||||
| Other employees, consultants, and former service providers | ||||||||
| Total outstanding at December 31 | ||||||||
The
weighted average exercise price of all outstanding warrants (vested and non-vested warrants; assuming 1 warrant = 1 share) is €
F-39
| Number of warrants granted | Expected | Expected | Risk-free | Expected duration (months) | ||||||||||||||||||||||||||||
| Dates | to Belgian benef. | to other benef. | Exercise price (€) | dividend Yield | stock price volatility | interest rate | to Belgian benef. | to other benef. | ||||||||||||||||||||||||
| 23-Jun-14 | € | % | % | |||||||||||||||||||||||||||||
| 10-Dec-14 | € | % | % | |||||||||||||||||||||||||||||
| 09-Feb-15 | € | % | % | |||||||||||||||||||||||||||||
| 01-Apr-15 | € | % | % | |||||||||||||||||||||||||||||
| 01-May-15 | € | % | % | |||||||||||||||||||||||||||||
| 29-May-15 | € | % | % | |||||||||||||||||||||||||||||
| 01-Jun-15 | € | % | % | |||||||||||||||||||||||||||||
| 01-Jul-15 | € | % | % | |||||||||||||||||||||||||||||
| 01-Aug-15 | € | % | % | |||||||||||||||||||||||||||||
| 01-Sep-15 | € | % | % | |||||||||||||||||||||||||||||
| 01-Oct-15 | € | % | % | |||||||||||||||||||||||||||||
| 01-Nov-15 | € | % | % | |||||||||||||||||||||||||||||
| 01-Dec-15 | € | % | % | |||||||||||||||||||||||||||||
| 01-Jan-16 | € | % | % | |||||||||||||||||||||||||||||
| 04-Feb-16 | € | % | % | |||||||||||||||||||||||||||||
| 04-Feb-16 | € | % | % | |||||||||||||||||||||||||||||
| 22-Apr-16 | € | % | % | |||||||||||||||||||||||||||||
| 27-May-16 | € | % | % | |||||||||||||||||||||||||||||
| 01-Jun-16 | € | % | % | |||||||||||||||||||||||||||||
| 01-Aug-16 | € | % | % | |||||||||||||||||||||||||||||
| 21-Oct-16 | € | % | % | |||||||||||||||||||||||||||||
| 22-Jan-16 | € | % | % | |||||||||||||||||||||||||||||
| 01-Dec-16 | € | % | % | |||||||||||||||||||||||||||||
| 01-Jan-17 | € | % | % | |||||||||||||||||||||||||||||
| 01-Mar-17 | € | % | % | |||||||||||||||||||||||||||||
| 01-Apr-17 | € | % | % | |||||||||||||||||||||||||||||
| 11-Apr-17 | € | % | % | |||||||||||||||||||||||||||||
| 1-Jun-17 | € | % | % | |||||||||||||||||||||||||||||
| 1-Jul-17 | € | % | % | |||||||||||||||||||||||||||||
| 29-Jul-17 | € | % | % | |||||||||||||||||||||||||||||
| 01-Sep-17 | € | % | % | |||||||||||||||||||||||||||||
| 01-Oct-17 | € | % | % | |||||||||||||||||||||||||||||
| 02-Nov-17 | € | % | % | |||||||||||||||||||||||||||||
| 1-Dec-17 | € | % | % | |||||||||||||||||||||||||||||
| 20-Jun-17 | € | % | % | |||||||||||||||||||||||||||||
| 27-Jun-17 | € | % | % | |||||||||||||||||||||||||||||
| 01-Apr-18 | € | % | % | |||||||||||||||||||||||||||||
| 01-May-18 | € | % | % | |||||||||||||||||||||||||||||
| 01-Jun-18 | € | % | % | |||||||||||||||||||||||||||||
| 01-Aug-18 | € | % | % | |||||||||||||||||||||||||||||
| 05-Dec-18 | € | % | % | |||||||||||||||||||||||||||||
| 24-Jan-19 | € | % | % | |||||||||||||||||||||||||||||
| 16-May-19 | - | € | % | % | ||||||||||||||||||||||||||||
| 01-Nov-19 | € | % | % | |||||||||||||||||||||||||||||
| 01-Dec-19 | € | % | % | |||||||||||||||||||||||||||||
F-40
| Number of warrants granted | Expected | Expected | Risk-free | Expected duration (months) | ||||||||||||||||||||||||||||
| Dates | to Belgian benef. | to other benef. | Exercise price (€) | dividend Yield | stock price volatility | interest rate | to Belgian benef. | to other benef. | ||||||||||||||||||||||||
| 01-Feb-20 | € | % | % | |||||||||||||||||||||||||||||
| 01-Jun-20 | € | % | % | |||||||||||||||||||||||||||||
| 01-Oct-20 | € | % | % | |||||||||||||||||||||||||||||
| 15-Jul-20 | € | % | % | |||||||||||||||||||||||||||||
| 01-Jul-19 | € | % | % | |||||||||||||||||||||||||||||
| 24-Jul-19 | € | % | % | |||||||||||||||||||||||||||||
| 15-Jul-20 | € | % | % | |||||||||||||||||||||||||||||
| 30-Jul-20 | - | € | % | % | ||||||||||||||||||||||||||||
| 01-Oct-20 | € | - | % | % | ||||||||||||||||||||||||||||
| 01-Mar-21 | € | - | % | % | ||||||||||||||||||||||||||||
| 03-May-21 | € | % | % | |||||||||||||||||||||||||||||
| 01-Jun-21 | € | % | % | |||||||||||||||||||||||||||||
| 27-Jul-21 | € | % | % | |||||||||||||||||||||||||||||
| 27-Jul-21 | € | % | % | |||||||||||||||||||||||||||||
| 24-Nov-21 | € | % | % | |||||||||||||||||||||||||||||
| 03-Jul-21 | € | % | % | |||||||||||||||||||||||||||||
| 07-Jul-21 | € | % | % | |||||||||||||||||||||||||||||
| 06-May-22 | € | % | % | |||||||||||||||||||||||||||||
| 04-Aug-22 | € | % | % | |||||||||||||||||||||||||||||
| 03-Aug-22 | € | % | % | |||||||||||||||||||||||||||||
| 03-Aug-22 | € | % | % | |||||||||||||||||||||||||||||
| 04-Aug-22 | € | % | % | |||||||||||||||||||||||||||||
| 01-Oct-22 | € | % | % | |||||||||||||||||||||||||||||
| 01-Dec-22 | € | % | % | |||||||||||||||||||||||||||||
| 25-Mar-23 | € | % | % | |||||||||||||||||||||||||||||
| 3-May-23 | € | % | % | |||||||||||||||||||||||||||||
| 27-Apr-23 | € | % | % | |||||||||||||||||||||||||||||
| 22-Jun-23 | € | % | % | |||||||||||||||||||||||||||||
| 30-Jun-23 | € | % | % | |||||||||||||||||||||||||||||
| 04-Jul-23 | € | % | % | |||||||||||||||||||||||||||||
| 05-Sep-23 | € | % | % | |||||||||||||||||||||||||||||
| 18-Sep-23 | € | % | % | |||||||||||||||||||||||||||||
| 18-Sep-23 | € | % | % | |||||||||||||||||||||||||||||
| 09-Nov-23 | € | % | % | |||||||||||||||||||||||||||||
| 09-Nov-23 | € | % | % | |||||||||||||||||||||||||||||
The above inputs for the Black-Scholes model have been determined based on the following:
| ● | The dividend return is estimated by reference to the historical dividend payment of the Company. Currently, this is estimated to be zero as no dividends have been paid since inception. |
| ● | The expected volatility was determined using the average volatility of the stock over the last |
| ● | On November 27, 2023 the Company announced its transition to a single listing on Nasdaq which repositioned the Company’s shares from the Euronext Brussels trading system to the Nasdaq trading system. Since there were no shares granted following this delisting date, the risk-free interest rate is based on the interest rate applicable for the 10-year Belgian government bond at the grant date. |
NOTE 25: Related parties
Transactions between the Company and its employees, consultants or Directors are described below. There were no other related party transactions.
Remuneration of key management personnel
During the year ended December 31, 2023, the executive management team included four members:
| 1. | Chief Executive Officer, Mr. Michael K. McGarrity |
| 2. | Executive Vice President of Corporate Development & General Counsel, Mr. Joseph Sollee |
| 3. | Chief Financial Officer, Mr. Ron Kalfus |
| 4. | Chief Commercial Officer, Mr. John Bellano |
F-41
Thousands of $ except per personnel, warrants & share amounts For The Years ended December 31 |
2023 |
2022 |
2021 | |||||||||
| Number of management members and Executive Directors | ||||||||||||
| Short-term employee benefits | ||||||||||||
| Post-employment benefits | ||||||||||||
| Other employment costs | ||||||||||||
| Total benefits | ||||||||||||
| IFRS share-based compensation expense | ||||||||||||
| Number of warrants offered | ||||||||||||
| Cumulative outstanding warrants | ||||||||||||
| Exercisable warrants | ||||||||||||
| 2023 | 2022 | 2021 | ||||||||||
| Number of warrants exercised | ||||||||||||
| Number of new warrants granted and accepted | ||||||||||||
| Annualized IFRS cost for existing warrants | $ | $ | $ | |||||||||
No loans, quasi-loans or other guarantees are outstanding with members of the executive management team.
Remuneration of the Board
The
total remuneration of the Board of Directors (including the Executive Director) in 2023, 2022, and 2021 was $
Transactions with Non-Executive Directors
Since
2012, the Non-Independent Directors do not receive a fee payment for attending and preparing for Board meetings or for assisting the
Company with Board matters. They receive reimbursement for expenses directly related to the Board meetings, totaling less than $
The
Independent Directors receive a fee for attending and preparing meetings of the Board of Directors and for assisting the Company with
Board matters, and they receive reimbursement for expenses directly related to the Board meetings. In 2023, 2022, and 2021, fees and
expense reimbursement in the amount of $
Warrants
to subscribe to
NOTE 26: Significant agreements, commitments and contingencies
Fair value of Other financial liabilities
Other
financial liabilities include the contingent consideration related to the acquisition of NovioGendix in 2015. The Company is contractually
required to pay at maturity to the holder of the obligation the amount of maximum $
The
contingent consideration related to the acquisition of the GPS business from Exact Sciences in August 2022 (as detailed in Note 3), has
been assessed at $
F-42
Collaborative research agreements and clinical research agreements
The Company
has entered into multiple agreements with universities, medical centers and external researchers for research and development work and
for the validation of the Company’s technology and products. These agreements typically have durations of
Intellectual property in-licensing agreements
The Company has entered into numerous agreements with universities and companies for in-licensing intellectual property. These agreements typically require the Company to pay an up-front fee, annual maintenance fees and/or minimum annual royalty fees, legal fees related to the patents, and certain milestone and royalty fees if the patents are eventually used in a commercialized product. In addition, the Company must provide the licensor with periodic reports.
Commercial and intellectual property sub-licensing agreements
The Company has entered into multiple partnering and sub-licensing agreements. With regards to the Company’s developed tests, the Company has entered into a range of marketing and sales arrangements with commercial entities. These important relationships provide the Company with additional resources and infrastructure to expand the geographic reach and awareness of the Company’s solutions, primarily in relation to the Confirm mdx and Select mdx tests.
In regard to intellectual property that MDxHealth has developed or improved, MDxHealth has sublicensed certain of its non-core technologies to commercial partners, several of whom have launched products that generate royalties and other fees. These sublicenses include an exclusive sublicense to Laboratory Corporation of America (LabCorp) for the MGMT test, which LabCorp began to commercialize in North America in 2008, and an exclusive sublicense to Vesica Health, Inc. for the Company’s patented AssureMDx test for the purpose of bladder cancer detection on a worldwide basis.
Litigation
As of the date of this document and as far as MDxHealth is aware, the Company is not involved in any material legal proceedings.
NOTE 27: Subsidiaries
| MDxHealth Inc. | ||
| Address | ||
| Incorporation Date | ||
| MDxHealth B.V. | ||
| Address | ||
| Incorporation Date | ||
| Incorporated into MDxHealth on |
NOTE 28: Principal audit fees and services
During the past fiscal year, in addition to their usual activity, the statutory auditor performed additional activities on behalf of the Company mainly for the issuance of special reports related to warrant plans, grant report certification, for participation to the audit committees and for participation to special projects.
| in thousands of $ | in thousands of € | ||||||||||||||||||||||||
| For the years ended December 31 | 2023 | 2022 | 2021 | 2023 | 2022 | 2021 | |||||||||||||||||||
| Audit fee for statutory and consolidated financials | |||||||||||||||||||||||||
| Other audit fees | |||||||||||||||||||||||||
| Audit related and other services | |||||||||||||||||||||||||
| Total | |||||||||||||||||||||||||
F-43