|
Delaware
|
| |
001-40679
|
| |
86-2365445
|
|
(State or Other Jurisdiction of
Incorporation or Organization)
|
| |
(Primary Standard Industrial
Classification Code Number)
|
| |
(I.R.S. Employer
Identification No.)
|
|
Tonya Mitchem Grindon
Nathanael P. Kibler
Andrew T. Yonchak
Baker, Donelson, Bearman, Caldwell &
Berkowitz, PC
1600 West End Avenue, Suite 2000
Nashville, TN 37203
(615) 726-5600
(615) 744-5607 (fax)
|
| |
Ben A. Stacke
Jonathan L.H. Nygren
Griffin D. Foster
Faegre Drinker Biddle & Reath LLP
2200 Wells Fargo Center
90 South Seventh Street
Minneapolis, MN 55402
(612) 766-7000
(612) 766-1600 (fax)
|
|
Large accelerated filer ☐
|
| |
Accelerated filer ☐
|
|
Non-accelerated ☒
|
| |
Smaller reporting company ☒
|
|
|
| |
Emerging growth company ☒
|
|
|
| |
By Order of the Board of Directors,
|
|
|
| |
|
|
|
| |
/s/ R. Andrew White
|
|
|
| |
R. Andrew White
Chief Executive Officer
|
|
|
| |
By Order of the Board of Directors,
|
|
|
| |
|
|
|
| |
/s/ R. Andrew White
|
|
|
| |
R. Andrew White
Chief Executive Officer
|
|
(1)
|
| |
Proposal 1 — The NTA Proposal — To consider and vote on an amendment (the “NTA Amendment”) to the Amended and Restated Certificate of Incorporation of SEPA, dated July 27, 2021, as amended on December 20, 2022 and October 3, 2023 (the “Current Charter”),
which amendment shall be effective, if adopted and implemented by SEPA, prior to the consummation of the proposed Business Combination, to remove from the Current Charter the redemption limitation contained under Section 9.2(a) of the
Current Charter preventing SEPA from redeeming shares of SEPA’s Class A common stock, par value $0.0001 per share (“Class A Common Stock” or “Public Shares”), if it would have less than $5,000,001 of net tangible assets. A copy of the
NTA Amendment is attached to the accompanying proxy statement/prospectus as Annex C. The NTA Proposal is described in more detail in the accompanying proxy statement/prospectus under the heading “Proposal 1: The NTA Proposal.”
|
|
|
| |
|
|
(2)
|
| |
Proposal 2 — The Business Combination Proposal — To consider and vote upon a proposal to approve the Merger Agreement and the transactions contemplated thereby, including the Business Combination, pursuant to which Merger Sub will merge with and into
SANUWAVE, with SANUWAVE continuing as the surviving entity of the Business Combination and becoming a subsidiary of SEPA as described in more detail in the accompanying proxy statement/prospectus.
|
|
|
| |
|
|
|
| |
A copy of the Merger Agreement is appended to the accompanying proxy
statement/prospectus as Annex A. The Business Combination Proposal is described in more detail in the accompanying proxy statement/prospectus under the heading “Proposal 2: The Business
Combination Proposal.”
|
|
|
| |
|
|
(3)
|
| |
Proposal 3 — The Charter Proposal — To consider and vote on a proposal to approve, in connection with the Merger Agreement, the replacement of the Current Charter with the proposed new Second Amended and Restated Certificate of
Incorporation of SEPA (the “Proposed Charter”), in the form appended to the accompanying proxy statement/prospectus as Annex D, to be effective upon the filing with and acceptance by the Delaware Secretary of State, pursuant
to which, among other things, the name of SEPA will be changed to “SANUWAVE Health, Inc.” and certain blank check provisions will be removed from the Current Charter. The Charter Proposal is conditioned on the approval of the Business
Combination Proposal. Therefore, if the Business Combination Proposal is not approved, then the Charter Proposal will have no effect, even if approved by SEPA Stockholders. The Charter Proposal is described in more detail in the
accompanying proxy statement/prospectus under the heading “Proposal 3: The Charter Proposal.”
|
|
|
| |
|
|
(4) – (11)
|
| |
Proposals 4 – 11 — The Organizational Documents Proposals — To consider and vote, on an advisory and non-binding basis, on eight (8) separate proposals to approve certain governance provisions in the Proposed Charter. These separate votes are not otherwise
required by Delaware law, separate and apart from the Charter Proposal, but are required by Securities and Exchange Commission guidance requiring that stockholders have the opportunity to present their views on important corporate
governance provisions. The Merger is not conditioned on the separate approval of the Organizational Documents Proposals (separate and apart from approval of the Charter Proposal). The Organizational Documents Proposals are described
in more detail in the accompanying proxy statement/prospectus under the heading “Proposals 4 – 11: The Organizational Documents Proposals.”
|
|
|
| |
|
|
(12)
|
| |
Proposal 12 — The Nasdaq Proposal — To consider and vote upon a proposal to approve, for purposes of complying with Nasdaq Listing Rules 5635(a), (b) and (d), the issuance of more than 20% of the issued and outstanding shares of Class A
Common Stock and the resulting change in control in connection with the Business Combination, PIPE Investment, and transactions contemplated thereby. The Nasdaq Proposal is described in more detail in the accompanying proxy
statement/prospectus under the heading “Proposal 12: The Nasdaq Proposal.”
|
|
|
| |
|
|
(13)
|
| |
Proposal 13 — The Incentive Plan Proposal — To consider and vote on a proposal to approve the SANUWAVE Health, Inc. 2023 Equity Incentive Plan (the “Incentive Plan”), in the form appended to the accompanying proxy statement/prospectus as Annex E.
The board of directors of SEPA (the “SEPA Board”) intends to adopt the Incentive Plan, subject to approval from the SEPA Stockholders, effective immediately prior to the Closing, to be used by the post-Business Combination company
(the “Company” or “Combined Company”). The Incentive Plan Proposal is described in more detail in the accompanying proxy statement/prospectus under the heading “Proposal
13: The Incentive Plan Proposal.”
|
|
|
| |
|
|
(14)
|
| |
Proposal 14 — The Adjournment Proposal — To consider and vote upon a proposal to adjourn the Stockholder Meeting to a later date or dates, if necessary, to permit further solicitation and vote of proxies if it is determined by the SEPA Board
that more time is necessary or appropriate to approve one or more Proposals at the Stockholder Meeting. The Adjournment Proposal is described in more detail in the accompanying proxy statement/prospectus under the heading “Proposal 14: The Adjournment Proposal.”
|
|
(a)
|
hold Public Shares or hold Public Shares through Units and elect to separate their Units into the underlying Public Shares
and SEPA Public Warrants prior to exercising Redemption Rights with respect to the Public Shares; and
|
|
(b)
|
prior to 5:00 p.m., Eastern Time, on , 2023 (two business days prior to the vote at the Stockholder Meeting), (i) submit a
written request to the Transfer Agent (A) requesting that SEPA redeem the applicable Public Shares for cash and (B) identifying themselves as a beneficial holder and providing their legal name, phone number and address and (ii) deliver
share certificates (if any) and other redemption forms to the Transfer Agent, physically or electronically through The Depository Trust Company.
|
|
By Order of the Board of Directors
|
| |
|
|
|
| |
|
|
/s/ Andrew White
|
| |
|
|
Andrew White
|
| |
|
|
Chief Executive Officer
|
| |
|
|
(1)
|
Proposal 1 — The Warrant Amendment Proposal — To consider and vote upon
an amendment to the Warrant Agreement, dated as of July 21, 2021, a copy of which is attached as Annex B to the accompanying proxy statement/prospectus, to provide that, upon closing of the Business Combination (the
“Closing”), the then outstanding SEPA Public Warrants will be canceled and exchanged for the right to receive 450,336 shares of SEPA’s Class A common stock, par value $0.0001 per share, (i.e., approximately 0.0499 shares of Class A Common Stock for each public warrant) and the then outstanding SEPA Private Placement Warrants will be canceled and exchanged
for the right to receive 400,000 shares of Class A Common Stock (i.e., approximately 0.0499 shares of Class A Common Stock for
each private placement warrant). The Warrant Amendment Proposal is described in more detail in the accompanying proxy statement/prospectus under the heading “Warrant Holder
Proposal 1: The Warrant Amendment Proposal.”
|
|
(2)
|
Proposal 2 — The Warrant Holders Adjournment Proposal — To consider and
vote upon a proposal to approve the adjournment of the Warrant Holders Meeting to a later date or dates, if necessary, (a) to permit further solicitation and vote of proxies in the event that there are insufficient votes for the
approval of the Warrant Amendment Proposal at the Warrant Holders Meeting or (b) if the board of directors of SEPA determines before the Warrant Holders Meeting that it is not necessary or no longer desirable to proceed with the
proposals. The Warrant Amendment Proposal is described in more detail in the accompanying proxy statement/prospectus under the heading “Warrant Holder Proposal 2: The
Warrant Holders Adjournment Proposal.”
|
|
By Order of the Board of Directors
|
| |
|
|
|
| |
|
|
/s/ Andrew White
|
| |
|
|
Andrew White
|
| |
|
|
Chief Executive Officer
|
| |
|
|
(1)
|
“Aggregate Exercise Price” means the sum of (a) the sum of the exercise prices of all in-the-money SANUWAVE Options; and (b)
the sum of the exercise prices of all in-the-money SANUWAVE Warrants, in each case outstanding as of immediately prior to the Effective Time.
|
|
(2)
|
“Company Fully Diluted Shares” means the sum of (i) the number of shares of SANUWAVE Stock that are issued and outstanding
(other than any SANUWAVE securities cancelled pursuant to Section 1.09 of the Merger Agreement); (ii) the number of shares of SANUWAVE Stock issuable upon the exercise of in-the-money SANUWAVE Options outstanding (whether or not
then vested or exercisable); (iii) the number of shares of SANUWAVE Stock issuable upon the exercise of in-the-money SANUWAVE Warrants assumed by SEPA outstanding; and (iv) the number of shares of SANUWAVE Stock issuable upon conversion
of the SANUWAVE Convertible Notes assumed by SEPA, in each case as of immediately prior to the Effective Time.
|
|
•
|
the occurrence of any event, change or other circumstances that could give rise to the termination of the Merger Agreement;
|
|
•
|
the inability to consummate the Business Combination, including due to any failure to obtain approval of the SEPA
Stockholders or SANUWAVE Stockholders or other conditions to the Closing in the Merger Agreement, such as the requirements that SEPA shall have at least $12.0 million at Closing resulting from proceeds of (a) SEPA’s Class A Common Stock
that has not been redeemed and (b) a private placement;
|
|
•
|
delays in obtaining or the inability to obtain any necessary regulatory approvals required to complete the Business
Combination;
|
|
•
|
the inability to obtain or maintain the listing of the Class A Common Stock on Nasdaq following the Business Combination;
|
|
•
|
costs related to the Business Combination;
|
|
•
|
changes in applicable laws or regulations; and
|
|
•
|
the possibility that SEPA or SANUWAVE may be adversely affected by other economic, business, and/or competitive factors.
|
|
Q.
|
Why am I receiving this proxy statement/prospectus?
|
|
A.
|
You are receiving this proxy statement/prospectus in connection with the Stockholder Meeting. SEPA is holding the Stockholder
Meeting to consider and vote upon the Proposals described below. Your vote is important. You are encouraged to vote as soon as possible after carefully reviewing this proxy statement/prospectus.
|
|
Q.
|
Are the Proposals conditioned on one another?
|
|
A.
|
Each of the NTA Proposal, the Business Combination Proposal, the Charter Proposal, the Nasdaq Proposal, and the Incentive
Plan Proposal is interdependent upon the others and each must be approved in order for SEPA to complete the Business Combination contemplated by the Merger Agreement. The Organizational Documents Proposals are conditional upon the
Charter Proposal; the Adjournment Proposal is not conditioned on the approval of any other proposal.
|
|
Q.
|
Why is SEPA proposing the Business Combination?
|
|
A.
|
SEPA was incorporated to effect a merger, capital stock exchange, asset acquisition, stock purchase, reorganization or
similar business combination transaction. Since SEPA’s incorporation, the SEPA Board has sought to identify suitable candidates in order to effect such a transaction. In its review of SANUWAVE, the SEPA Board considered a variety of
factors weighing positively and negatively in connection with the Business Combination. After careful consideration, the SEPA Board has determined that the Business Combination presents a highly attractive business combination
opportunity. The SEPA Board believes that, based on its review and consideration, the Business Combination with SANUWAVE presents an opportunity to increase stockholder value. However, there can be no assurance that the anticipated
benefits of the Business Combination will be achieved.
|
|
Q.
|
What will happen in the Business Combination?
|
|
A.
|
Pursuant to the terms of the Merger Agreement, a business combination between SEPA and SANUWAVE will be effected. More
specifically, and as described in greater detail in this proxy statement/prospectus, at the Effective Time:
|
|
•
|
Merger Sub will merge with and into SANUWAVE, with SANUWAVE being the surviving company following the Merger and continuing
in existence as a wholly owned subsidiary of SEPA;
|
|
•
|
each issued and outstanding share of SANUWAVE Common Stock will be automatically converted into shares of Class A Common
Stock of SEPA at the Conversion Ratio (as defined in the Merger Agreement); and
|
|
•
|
outstanding Company Convertible Securities (as defined in the Merger Agreement) of SANUWAVE will be assumed by SEPA and will
be converted into the right to receive Class A Common Stock, reserved from issuance from the Merger Consideration, if not exercised or converted, as applicable, prior to the Effective Time.
|
|
Q.
|
What consideration will the SANUWAVE Securityholders receive in connection with the
acquisition of SANUWAVE by SEPA?
|
|
A.
|
Pursuant to the terms of the Merger Agreement, the Merger Consideration, consisting of 7,793,000 shares of Class A Common
Stock of SEPA, will be issued, or otherwise reserved for issuance, to the SANUWAVE Securityholders, as follows:
|
|
•
|
each share of SANUWAVE Common Stock that is outstanding will be automatically converted into shares of Class A Common Stock
of SEPA at the Conversion Ratio (as defined in the Merger Agreement);
|
|
•
|
each SANUWAVE Convertible Note that is outstanding, and has not been exchanged for shares of SANUWAVE Common Stock prior to
the Effective Time will, to the extent permitted by the terms of such convertible note, automatically be assumed by SEPA and convertible into the number of shares of Class A Common Stock of SEPA, reserved from issuance from the Merger
Consideration, equal to the product of (i) the number of shares of SANUWAVE Common Stock subject to such convertible note as of immediately prior to the Effective Time multiplied by (ii) the Conversion Ratio;
|
|
•
|
each outstanding SANUWAVE Option (whether vested or unvested, exercisable or unexercisable) that is outstanding as of
immediately prior to the Effective Time will be assumed by SEPA and automatically converted into the right to receive an option to acquire the number of shares of Class A Common Stock, reserved from issuance from the Merger
Consideration, equal to the product of (i) the number of shares of SANUWAVE Common Stock subject to such option as of immediately prior to the Effective Time multiplied by (ii) the Conversion Ratio; and
|
|
•
|
each SANUWAVE Warrant that is outstanding and unexercised, and has not been exchanged for shares of SANUWAVE Common Stock
prior to the Effective Time will, to the extent permitted by the terms of such warrant, automatically be assumed by SEPA and converted into a warrant to purchase the number of shares of Class A Common Stock, reserved from issuance from
the Merger Consideration, equal to the product of (1) the number of shares of SANUWAVE Common Stock subject to such warrant multiplied by (2) the Conversion Ratio.
|
|
Q.
|
What equity stake will current SEPA Stockholders and SANUWAVE Stockholders hold in the
Combined Company immediately after the completion of the Business Combination?
|
|
A.
|
Upon the completion of the Business Combination (assuming, among other things, that no SEPA Stockholders exercise Redemption
Rights with respect to their Class A Common Stock upon completion of the Business Combination), the SANUWAVE Stockholders are expected to own approximately 69.0% of the Combined Company’s outstanding Class A Common Stock and the current
holders of Class A Common Stock are expected to own approximately 11.5% of the Combined Company’s outstanding Class A Common Stock.
|
|
|
| |
No
Redemptions
|
| |
%
|
| |
10%
Redemptions
|
| |
%
|
| |
25%
Redemptions
|
| |
%
|
| |
50%
Redemptions
|
| |
%
|
| |
Maximum
Redemptions
|
| |
%
|
|
SANUWAVE Securityholders(1)
|
| |
7,793,000
|
| |
69.1%
|
| |
7,793,000
|
| |
69.5%
|
| |
7,793,000
|
| |
69.7%
|
| |
7,793,000
|
| |
69.7%
|
| |
7,793,000
|
| |
69.7%
|
|
SEPA Class A Stockholders (other than the Sponsor)(2)(3)
|
| |
1,304,259
|
| |
11.6%
|
| |
1,241,984
|
| |
11.1%
|
| |
1,148,572
|
| |
10.3%
|
| |
992,886
|
| |
8.9%
|
| |
681,512
|
| |
6.1%
|
|
Initial SEPA Class B Stockholders(3)(4)(5)
|
| |
1,232,241
|
| |
10.9%
|
| |
1,232,241
|
| |
11.0%
|
| |
1,232,241
|
| |
11.0%
|
| |
1,232,241
|
| |
11.0%
|
| |
1,232,241
|
| |
11.0%
|
|
SEPA Public Warrant Holders(6)
|
| |
450,336
|
| |
4.0%
|
| |
450,336
|
| |
4.0%
|
| |
450,336
|
| |
4.0%
|
| |
450,336
|
| |
4.0%
|
| |
450,336
|
| |
4.0%
|
|
SEPA Private Placement Warrant Holders(7)
|
| |
400,000
|
| |
3.5%
|
| |
400,000
|
| |
3.6%
|
| |
400,000
|
| |
3.6%
|
| |
400,000
|
| |
3.6%
|
| |
400,000
|
| |
3.6%
|
|
Sponsor Debt Conversion(8)
|
| |
100,000
|
| |
0.9%
|
| |
100,000
|
| |
0.9%
|
| |
100,000
|
| |
0.9%
|
| |
100,000
|
| |
0.9%
|
| |
100,000
|
| |
0.9%
|
|
PIPE Investors(9)
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
51,428
|
| |
0.5%
|
| |
207,114
|
| |
1.9%
|
| |
518,488
|
| |
4.6%
|
|
Total shares of Class A Common Stock
|
| |
11,279,836
|
| |
100%
|
| |
11,217,561
|
| |
100%
|
| |
11,175,577
|
| |
100%
|
| |
11,175,577
|
| |
100%
|
| |
11,175,577
|
| |
100%
|
|
Implied Per Share Value(10)
|
| |
$11.30
|
| |
—
|
| |
$11.37
|
| |
—
|
| |
$11.41
|
| |
—
|
| |
$11.41
|
| |
—
|
| |
$11.41
|
| |
—
|
|
(1)
|
SANUWAVE Securityholders consist of holders of shares of SANUWAVE Common Stock in addition to holders of in-the-money SANUWAVE
Warrants and in-the-money SANUWAVE Options that are not exercised prior the Closing and holders of SANUWAVE Convertible Notes that are not converted prior to the Closing. The table assumes that no SANUWAVE Warrants or SANUWAVE
Convertible Notes (including the PIPE Notes) are assumed by SEPA at the Closing (i.e., all outstanding SANUWAVE Warrants will be exercised, and all outstanding SANUWAVE Convertible Notes
(including the PIPE Notes) will be converted, prior to the Closing).
|
|
(2)
|
Pursuant to the Voting and Non-Redemption Agreements, certain SEPA Stockholders agreed not to redeem an aggregate of
681,512 shares of Class A Common Stock. As a result, the maximum number of shares of Class A Common Stock that may be redeemed in connection with the Business Combination is 622,747 shares of Class A Common Stock.
|
|
(3)
|
In order to bring SEPA into compliance with the Nasdaq listing standards, on October 2, 2023, the Sponsor elected to convert
2,415,375 of its shares of Class B Common Stock into 2,415,375 shares of Class A Common Stock (the “Conversion”). In order to maintain the same economics of the Business Combination for all SEPA Class B Stockholders, on October 2, 2023,
the Sponsor, SEPA and SANUWAVE entered into a Forfeiture and Redemption Agreement, pursuant to which the Sponsor agreed to forfeit 1,746,316 shares of Class A Common Stock contingent upon and effective immediately prior to the Closing
of the Business Combination. The “SEPA Class A Stockholders” row does not include the 2,415,375 shares of Class A Common Stock that the Sponsor currently holds following the Conversion. The “Initial SEPA Class B Stockholders” row
includes the 2,415,375 shares of Class B Common Stock that the Sponsor held prior to the Conversion. Following the Closing, the Sponsor will hold 1,059,909 shares of Class A Common Stock.
|
|
(4)
|
Reflects the conversion of Class B Common Stock to Class A Common Stock, pursuant to the Class B Charter Amendment, amending
the Class B Common Stock conversion ratio to 0.277 shares of Class A Common Stock for each share of Class B Common Stock.
|
|
(5)
|
Reflects the forfeiture of 61,850 shares of Class B Common Stock to the Company, in exchange for no consideration, by a
SEPA Class B Stockholder pursuant to a forfeiture and redemption agreement entered into on December 18, 2023, in connection with the liquidation of such stockholder. Following the forfeiture, the shares were cancelled.
|
|
(6)
|
Reflects the conversion of the SEPA Public Warrants into 450,336 shares of Class A Common Stock (i.e., approximately 0.0499 shares of Class A Common Stock for each public warrant) pursuant to the Warrant Agreement Amendment. All outstanding SEPA Public Warrants will be converted in
connection with the Business Combination, and none will be outstanding following the Closing.
|
|
(7)
|
Reflects the conversion of the SEPA Private Placement Warrants into 400,000 shares of Class A Common Stock (i.e., approximately 0.0499 shares of Class A Common Stock for each private placement warrant) pursuant to the Warrant Agreement Amendment. All outstanding SEPA Private Placement Warrants
will be converted in connection with the Business Combination, and none will be outstanding following the Closing.
|
|
(8)
|
On October 11, 2022, SEPA issued the Sponsor Note to the Sponsor in the original principal amount of up to $1,000,000
(together with all accrued but unpaid interest, fees, expenses and other amounts payable under the Sponsor Note, the “Outstanding Indebtedness”). In accordance with the Merger Agreement and as a part of the PIPE Investment, the
Sponsor has agreed to cancel and release the Outstanding Indebtedness in exchange for, and in consideration of, the issuance to the Sponsor by SEPA of 100,000 shares of Class A Common Stock.
|
|
(9)
|
The Merger Agreement provides that SANUWAVE’s obligation to consummate the Business Combination is conditioned on, among
other things, that as of the Closing, SEPA have at least $12,000,000 resulting from (i) proceeds from Class A Common Stock that has not been redeemed in the Redemption and (ii) proceeds of the PIPE Investment. Assuming SEPA
Stockholders redeem the maximum amount (622,747 shares of Class A Common Stock), then SEPA will need to obtain at least $5,184,880 (or 518,488 shares of Class A Common Stock at a price of $10.00 per share) in the PIPE Investment in
order to satisfy the Minimum Cash Condition. Assuming 50% redemptions of Class A Common Stock, then SEPA will need to receive at least $2,071,140 (or 207,114 shares of Class A Common Stock at a price of $10.00 per share) in the PIPE
Investment in order to satisfy the Minimum Cash Condition. Assuming 25% redemptions of Class A Common Stock, then SEPA will need to receive at least $514,280 (or 51,428 shares of Class A Common Stock at a price of $10.00 per share) in
the PIPE Investment in order to satisfy the Minimum Cash Condition. Thus, in summary, as the amount of redemptions of shares of Class A Common Stock increases, the amount that SEPA will need to receive in the PIPE Investment to
satisfy the Minimum Cash Condition also increases, which results in the implied per share value remaining relatively constant across the various redemption scenarios. Assuming 10% redemptions of Class A Common Stock or no redemptions
of Class A Common Stock, then SEPA will not need to raise any funds in the PIPE Investment in order to satisfy the Minimum Cash Condition. The actual amount that SEPA raises in the PIPE Investment will depend on the number of
redemptions of Class A Common Stock, market conditions, and other factors. As of the date hereof, SEPA does not have any commitments in the PIPE Investment.
|
|
(10)
|
Calculation of implied per share value assumes an enterprise value of $127.5 million of SANUWAVE upon consummation of the
Business Combination, which includes $12 million in proceeds from a combination of (A) proceeds from Class A Common Stock that have not been redeemed in the Redemption and (B) proceeds of the PIPE Investment.
|
|
Q.
|
Did the SEPA Board obtain a third-party valuation or fairness opinion in determining
whether or not to proceed with the Business Combination?
|
|
A.
|
Yes. The SEPA Board obtained a fairness opinion from of ValueScope, Inc. For a description of the opinion issued by
ValueScope, Inc. to the SEPA Board, please see “Proposal 2: The Business Combination Proposal — Opinion of ValueScope, Inc. as Fairness Opinion Provider.” The SEPA Board obtained and considered
a fairness opinion for a number of reasons: (i) to determine whether experienced, independent valuation specialists would judge the negotiated purchase price to be fair to SEPA, from a financial point of view, (ii) to help the SEPA
Board ensure that its own determination as to whether to accept and recommend the Merger was reasonable and in the best interests of SEPA Stockholders, (iii) to help the SEPA Board ensure that its own determination as to whether to
accept and recommend the Merger was the result of a reasonable and thorough examination of the relevant facts and (iv) to provide SEPA Stockholders with additional information to consider when deciding whether to vote in favor of the
Merger Agreement and the resulting Business Combination or not, and whether to redeem their shares in SEPA or not.
|
|
Q.
|
What happens to the funds deposited in the Trust Account after completion of the Business
Combination?
|
|
A.
|
After completion of the Business Combination, the funds in the Trust Account will be used to pay holders of the Class A
Common Stock who exercise Redemption Rights. As of November 30, 2023, there were cash and marketable securities held in the Trust Account of approximately $13.6 million. These funds will not be released until the earlier of the
completion of the Business Combination or the Redemption of the Class A Common Stock if SEPA is unable to complete a Business Combination by July 30, 2024 (except that interest earned on the amounts held in the Trust Account may be
released earlier as necessary to pay for any franchise or income taxes).
|
|
Q.
|
What happens if a substantial number of SEPA Stockholders vote in favor of the Business
Combination Proposal and exercise their Redemption Rights?
|
|
A.
|
SEPA Stockholders may vote in favor of the Business Combination and still exercise their Redemption Rights. The Business
Combination may be completed even though the funds available from the Trust Account and the number of SEPA Stockholders is substantially reduced as a result of Redemptions by SEPA Stockholders. Pursuant to Voting and Non-Redemption
Agreements, certain SEPA Stockholders agreed not to redeem (without receiving any cash consideration for this agreement) an aggregate of 681,512 shares of Class A Common Stock (representing approximately $7.1 million (calculated based
on the funds held in the Trust Account as of November 30, 2023) that SEPA would have otherwise been required to pay to redeem such shares in connection with the Business Combination).
|
|
Q.
|
What conditions must be satisfied to complete the Business Combination?
|
|
A.
|
Unless waived by the parties to the Merger Agreement, and subject to applicable law, the consummation of the Business
Combination is subject to a number of conditions set forth in the Merger Agreement including, among
|
|
Q.
|
When do you expect the Business Combination to be completed?
|
|
A.
|
It is currently expected that the Business Combination will be completed in the fourth quarter of 2023. This timing depends,
among other things, on the approval of the Proposals to be presented at the Stockholder Meeting. However, such meeting could be adjourned if the Adjournment Proposal is adopted at the Stockholder Meeting and SEPA elects to adjourn the
Stockholder Meeting to a later date or dates to permit further solicitation and vote of proxies if reasonably determined to be necessary or desirable by SEPA.
|
|
Q.
|
Will SEPA enter into any financing arrangements in connection with the Business
Combination?
|
|
A.
|
As of the date of this proxy statement/prospectus, there is no commitment for proposed financing from the PIPE Investment,
SEPA and SANUWAVE intend to enter into PIPE Subscription Agreements with PIPE Investors for an estimated aggregate amount of $5,184,880 for 518,488 shares of Class A Common Stock at a price of $10.00 per share in a private placement in
SEPA to be consummated simultaneously with the Closing. Except for the PIPE Investment described in the preceding sentence, SEPA does not currently expect to enter into any financing arrangements in connection with the Business
Combination.
|
|
Q.
|
Why is SEPA proposing the Incentive Plan Proposal?
|
|
A.
|
The purpose of the Incentive Plan is to enable the Combined Company to offer eligible employees, directors and consultants
cash and stock-based incentive awards in order to attract, retain and reward these individuals and strengthen the mutuality of interests between them and the Combined Company’s stockholders. For more information, see the section
entitled “Proposal 11: The Incentive Plan Proposal.”
|
|
Q.
|
What changes are being made to the Current Charter in connection with the Business
Combination?
|
|
A:
|
The NTA Amendment will remove from the Current Charter the redemption limitation contained under Section 9.2(a) of the
Current Charter preventing SEPA from redeeming shares of its Class A Common Stock if it would have less than $5,000,001 of net tangible assets.
|
|
Q.
|
Why is SEPA proposing the Adjournment Proposal?
|
|
A.
|
SEPA is proposing the Adjournment Proposal to allow the adjournment of the Stockholder Meeting to a later date or dates,
including if necessary to permit further solicitation and vote of proxies if it is determined by SEPA that more time is necessary or appropriate to approve one or more Proposals at the Stockholder Meeting.
|
|
Q.
|
When and where will the Stockholder Meeting be held?
|
|
A.
|
The Stockholder Meeting will be held virtually at a.m., Central Time, on , 2023. Only stockholders who held Common
Stock of SEPA at the close of business on , 2023 will be entitled to attend and vote at the Stockholder Meeting and at any adjournments and postponements thereof.
|
|
Q.
|
Who is entitled to vote at the Stockholder Meeting?
|
|
A.
|
SEPA has fixed , 2023 as the Record Date. If you were a SEPA Stockholder at the close of business on the Record Date, you
are entitled to vote on matters that come before the Stockholder Meeting. However, a SEPA Stockholder may only vote his, her or its shares if he, she or it is present in person virtually or is represented by proxy at the Stockholder
Meeting.
|
|
Q.
|
How do I vote?
|
|
A.
|
If you are a record owner of your shares of SEPA Common Stock, there are three ways to vote your shares at the Stockholder
Meeting:
|
|
Q.
|
What if I do not vote my SEPA shares or if I abstain from voting?
|
|
A.
|
At the Stockholder Meeting, SEPA will count a properly executed proxy marked “ABSTAIN” with respect to a particular Proposal
as present for purposes of determining whether a quorum is present. For purposes of approval, an abstention will have the same effect as a vote “AGAINST” the NTA Proposal, the Business Combination Proposal, and the Charter Proposal but
have no effect on the outcome of the other Proposals. However, if you do not submit a proxy or voting instruction, do not attend the Stockholder Meeting virtually or by proxy and your shares are not otherwise voted at the Stockholder
Meeting, your failure to do so will have the same effect as a vote “AGAINST” the NTA Proposal, the Business Combination Proposal, and the Charter Proposal but have no effect on the outcome of the other Proposals.
|
|
Q.
|
If my shares are held in “street name,” will my broker automatically vote them for me?
|
|
A.
|
No. Under the rules governing banks and brokers who submit a proxy card with respect to shares held in street name, such
banks and brokers have the discretion to vote on routine matters, but not on non-routine matters. The approval of the NTA Proposal, the Business Combination Proposal, the Charter Proposal, the Organizational Document Proposals, the
Nasdaq Proposal, the Incentive Plan Proposal, and the Adjournment Proposal (if presented) are non-routine matters.
|
|
Q.
|
What Proposals must be passed in order for the Business Combination to be completed?
|
|
A.
|
The Business Combination will not be completed unless the NTA Proposal, the Business Combination Proposal, the Charter
Proposal, the Nasdaq Proposal, and the Incentive Plan Proposal are approved. If SEPA does not complete a business combination by July 30, 2024, SEPA will be required to dissolve and liquidate itself and return the monies held within its
Trust Account to its SEPA Stockholders.
|
|
Q.
|
How does the SEPA Board recommend that I vote on the Proposals?
|
|
A.
|
The SEPA Board unanimously recommends that the SEPA Stockholders entitled to vote on the Proposals, vote as follows:
|
|
•
|
“FOR” approval of the NTA Proposal;
|
|
•
|
“FOR” approval of the Business Combination Proposal;
|
|
•
|
“FOR” approval of the Charter Proposal;
|
|
•
|
“FOR” approval of the Organizational Documents Proposals;
|
|
•
|
“FOR” approval of the Nasdaq Proposal;
|
|
•
|
“FOR” approval of the Incentive Plan Proposal; and
|
|
•
|
“FOR” approval of the Adjournment Proposal, if presented.
|
|
Q.
|
How many votes do I have?
|
|
A.
|
SEPA Stockholders have one vote per each share of SEPA Common Stock held by them on the Record Date for the Stockholder
Meeting.
|
|
Q.
|
How will the Sponsor and SEPA’s officers and directors vote in connection with the
Proposals?
|
|
A.
|
As of the Record Date, the Sponsor and SEPA’s officers and directors own approximately 76.4% of SEPA’s outstanding Common
Stock. Pursuant to the Sponsor Voting Agreement, the Sponsor has agreed to vote its shares (including the Founder Shares) in favor of the Proposals. Accordingly, it is more likely that the necessary stockholder approval of the Proposals
will be received at the Stockholder Meeting, than would be the case if the Sponsor and SEPA’s officers and directors had agreed to vote their shares of Common Stock in accordance with the majority of the votes cast by SEPA Stockholders.
|
|
Q.
|
What interests do the Sponsor and SEPA’s officers and directors have in the Business
Combination?
|
|
A.
|
In considering the recommendation of the SEPA Board to vote in favor of the Business Combination, SEPA Stockholders should be
aware that, aside from their interests as SEPA Stockholders, the Sponsor and SEPA’s directors and officers have interests in the Business Combination that are different from, or in addition to, those of other SEPA Stockholders
generally. SEPA’s directors were aware of and considered these interests, among other matters, in evaluating the Business Combination, and in recommending to the SEPA Stockholders that they approve the Business Combination. SEPA
Stockholders should take these interests into account in deciding whether to approve the Business Combination. These interests include, among other things:
|
|
•
|
Pursuant to the Letter Agreement, 5,031,250 Founder Shares owned by the Sponsor and other insiders became subject to a
lock-up whereby, subject to certain limited exceptions, they agreed not to transfer, assign or sell the Founder Shares until the earlier of (A) one year after the completion of SEPA’s initial business combination or (B) subsequent to
the Business Combination, (x) if the last sale price of Class A Common Stock equals or exceeds $12.00 per share (as adjusted for stock splits, stock dividends, reorganizations, recapitalizations and other similar transactions) for any
20 trading days within any 30-trading day period commencing at least 150 days after SEPA’s initial business combination or (y) the date on which SEPA completes a liquidation, merger, capital stock exchange, reorganization or other
similar transaction that results in all SEPA Stockholders having the right to exchange their shares of Class A Common Stock for cash, securities or other property; provided, however, upon approval of the Letter Agreement Amendment, this
period will be amended to until the earlier of (i) 180 days after the completion of the Business Combination or (ii) subsequent to the Business Combination, the date on which SEPA completes a liquidation, merger, capital stock exchange,
reorganization or other similar transaction that results in all SEPA Stockholders having the right to exchange their shares of Class A Common Stock for cash, securities or other property.
|
|
•
|
The Sponsor owns 2,415,375 shares of Class A Common Stock (the “Converted Shares”) (of which 1,746,316 shares will be
forfeited contingent upon and effective immediately prior to the Closing of the Business Combination pursuant to the Forfeiture and Redemption Agreement) and 1,050,000 Founder Shares, and SEPA’s officers and directors own 140,000
Founder Shares. On March 4, 2021, SEPA’s founder acquired 5,031,250 Founder Shares for an aggregate purchase price of $25,000, or approximately $0.005 per share. Also on March 24, 2021, SEPA’s founder assigned 160,000 Founder Shares
(40,000 Founder
|
|
•
|
The Sponsor and SEPA’s directors and officers may be incentivized to complete the Business Combination, or an alternative
initial business combination with a less favorable company or on terms less favorable to shareholders, rather than to liquidate, in which case the Sponsor would lose its entire investment. As a result, the Sponsor may have a conflict of
interest in determining whether SANUWAVE is an appropriate business with which to effectuate a business combination and/or in evaluating the terms of the Business Combination.
|
|
•
|
Simultaneously with the closing of the IPO, the Sponsor purchased 7,850,000 warrants at a price of $1.00 per warrant. Each
private placement warrant is exercisable to purchase one share of Class A Common Stock at a price of $11.50 per share. Such warrants will expire worthless if SEPA fails to complete an initial business combination by July 30, 2024.
|
|
•
|
Pursuant to the Sponsor Voting Agreement, the Sponsor has agreed to vote all of the Sponsor’s shares of SEPA in favor of the
Business Combination and to otherwise take (or not take, as applicable) certain other actions in support of the Business Combination and the other matters to be submitted to SEPA Stockholders for approval in connection with the Business
Combination. The Sponsor Voting Agreement also prevents transfers of SEPA shares held by the Sponsor between the date of the Sponsor Voting Agreement and the termination of such Sponsor Voting Agreement, except for certain permitted
transfers where the recipient also agrees to comply with the Sponsor Voting Agreement.
|
|
•
|
On October 11, 2022, SEPA issued the Sponsor Note to the Sponsor, pursuant to which SEPA could borrow up to $1,000,000 at
a 6% interest rate on or before October 11, 2024 to cover, among other things, expenses related to a business combination. As of December 20, 2023, SEPA has borrowed the full $1,000,000 under the Sponsor Note.
|
|
•
|
In order to finance transaction costs in connection with the Business Combination, the Sponsor or an affiliate of the
Sponsor, or certain of SEPA’s officers and directors may, but are not obligated to, loan SEPA the Working Capital Loans. If SEPA completes the Business Combination, SEPA would repay the Working Capital Loans out of the proceeds held in
the Trust Account released to SEPA. Otherwise, the Working Capital Loans would be repaid only out of funds held outside the Trust Account. In the event that the Business Combination is not completed, SEPA may use a portion of the
proceeds held outside the Trust Account to repay the Working Capital Loans but no proceeds held in the Trust Account would be used to repay the Working Capital Loans.
|
|
•
|
The Sponsor has agreed that it will be liable to SEPA if and to the extent any claims by a third party (other than SEPA’s
independent auditors) for services rendered or products sold to SEPA, or a prospective target business with which SEPA has discussed entering into a transaction agreement, reduce the amount of funds in the Trust Account to below (i)
$10.10 per Public Share or (ii) such lesser amount per Public Share held in the Trust Account as of the date of the liquidation of the Trust Account, due to reductions in value of
|
|
•
|
The fact that SEPA has provisions in its Current Charter which waive the corporate opportunities doctrine, which means that
SEPA’s officers and directors have not been obligated to bring all corporate opportunities to SEPA. The potential conflict of interest relating to the waiver of the corporate opportunities doctrine in the Current Charter did not, to
SEPA’s knowledge, impact the search for an acquisition target or prevent SEPA from reviewing any opportunities as a result of such waiver.
|
|
Q.
|
Do I have Redemption Rights?
|
|
A.
|
Pursuant to the Current Charter, SEPA Stockholders will be entitled to redeem their Class A Common Stock for a pro rata share
of the Trust Account (which would have amounted to approximately $10.46 per share of Class A Common Stock based on the amount in the Trust Account as of November 30, 2023) net of taxes payable. If a SEPA Stockholder exercises its
Redemption Rights, then such SEPA Stockholder will be exchanging its Class A Common Stock for cash.
|
|
Q.
|
How do I exercise my Redemption Rights?
|
|
A.
|
If you are a SEPA Stockholder and you seek to have your shares redeemed, you must, no later than 5:00 p.m., Eastern Time, on
, 2023 (two (2) business days before the Stockholder Meeting), (i) submit your request in writing to SEPA’s Transfer Agent (A) requesting that SEPA redeem your shares for cash, at the address listed at the end of this section and (B)
identifying yourself as a beneficial holder and providing your legal name, phone number and address and (ii) deliver your stock certificates (if any) and other redemption forms to SEPA’s Transfer Agent physically or electronically using
The Depository Trust Company’s DWAC system.
|
|
Q.
|
Is there a limit on the number of shares I may redeem?
|
|
A.
|
The Current Charter provides that each SEPA Stockholder, together with any affiliate or any other person with whom such SEPA
Stockholder is acting in concert or as a “group” (as defined in Section 13(d)(3) of the Exchange Act), will be restricted from seeking Redemption Rights with respect to 15% or more of the Class A Common Stock without the consent of
SEPA. Accordingly, any shares held by a SEPA Stockholder or “group” in excess of such 15% cap will not be redeemed by SEPA without SEPA’s consent. Any SEPA Stockholder who holds less than 15% of the Class A Common Stock may have all of
the Class A Common Stock held by him or her redeemed for cash.
|
|
Q.
|
What are the material U.S. federal income tax consequences to stockholders that exercise
their Redemption Rights?
|
|
A.
|
SEPA Stockholders that elect to exercise their Redemption Rights generally will recognize capital gain or loss equal to the
difference between the amount of cash received on the Redemption of the Class A Common Stock and such stockholder’s adjusted tax basis in such Class A Common Stock, which generally will be equal to the cost of such Class A Common Stock.
A SEPA Stockholder who purchased the Class A Common Stock in the IPO generally will have a tax basis in the shares of Class A Common Stock that were part of the Units equal to the portion of the purchase price of such Units allocated to
the Class A Common Stock (such allocation based on the relative fair market value of the Class A Common Stock and the warrants at the time). However, in certain circumstances, the cash paid to such stockholders will be treated as
dividend income for U.S. federal income tax purposes. For a more complete discussion of the material U.S. federal income tax consequences to SEPA Stockholders that elect to exercise their Redemption Rights, see the discussion in the
section entitled “Proposal 2: The Business Combination Proposal — Material U.S. Federal Income Tax Consequences to Redemption.”
|
|
Q.
|
What are the material U.S. federal income tax consequences to SEPA Stockholders that do
not exercise their Redemption Rights?
|
|
A.
|
The Business Combination will qualify as a “reorganization” within the meaning of Section 368(a) of the Code for U.S. federal
income tax purposes. As a result of the Business Combination qualifying as a reorganization, no gain or loss generally will be recognized by a U.S. Holder (as defined below) of SANUWAVE Common Stock for U.S. federal income tax purposes
on the exchange of its shares of SANUWAVE Common Stock for Class A Common Stock of SEPA in the Business Combination. For a more complete discussion of the material U.S. federal income tax consequences of the Business Combination, please
carefully review the information set forth in the section titled “Tax Treatment of Business Combination” of this proxy statement/prospectus. The tax consequences of the Business Combination to any particular SANUWAVE Stockholder will
depend on that stockholder’s particular facts and circumstances.
|
|
Q.
|
What happens if the Business Combination is not completed?
|
|
A.
|
If a SEPA Stockholder has tendered shares to be redeemed but the Business Combination is not completed, the Redemptions will
be canceled and the tendered shares will be returned to the relevant SEPA Stockholders as appropriate. The current deadline set forth in the Current Charter for SEPA to complete its initial Business Combination is July 30, 2024.
|
|
Q.
|
Why am I receiving this proxy statement/prospectus?
|
|
A.
|
You are receiving this proxy statement/prospectus in connection with the Warrant Holders Meeting. SEPA is holding the Warrant
Holders Meeting to consider and vote upon the Warrant Holder Proposals described below. Your vote is important. You are encouraged to vote as soon as possible after carefully reviewing this proxy
statement/prospectus.
|
|
Q.
|
Why is SEPA providing SEPA Public Warrant Holders with the opportunity to vote on the
Warrant Agreement Amendment?
|
|
A.
|
We are seeking approval of the Warrant Agreement Amendment, which will amend the Warrant Agreement to provide that, at
Closing, the then outstanding SEPA Public Warrants will be canceled and exchanged for the right to receive 450,336 shares of Class A Common Stock (i.e., approximately 0.0499 shares of Class A
Common Stock for each public warrant) and the then outstanding SEPA Private Placement Warrants will be canceled and exchanged for the right to receive 400,000 shares of Class A Common Stock (i.e.,
approximately 0.0499 shares of Class A Common Stock for each private placement warrant).
|
|
Q.
|
Why is SEPA proposing the Warrant Holders Adjournment Proposal?
|
|
A.
|
SEPA is proposing the Warrant Holders Adjournment Proposal to allow the adjournment of the Warrant Holders Meeting to a later
date or dates, including if necessary to permit further solicitation and vote of proxies if it is determined by SEPA that more time is necessary or appropriate to approve the Warrant Amendment Proposal at the Warrant Holders Meeting.
|
|
Q.
|
What constitutes a quorum at the Warrant Holders Meeting?
|
|
A.
|
A quorum will be present at the Warrant Holders Meeting if the holders of at least 50% of the outstanding SEPA Public
Warrants are represented in person (which would include presence at the virtual meeting) or by proxy. As of the Record Date , SEPA Public Warrants would be required to achieve a quorum.
|
|
Q.
|
What vote is required to approve each proposal at the Warrant Holders Meeting?
|
|
A.
|
The Warrant Amendment Proposal: The approval of the Warrant Amendment Proposal
requires the affirmative vote of the holders of at least 50% of the then outstanding SEPA Public Warrants. Abstentions and broker non-votes will count as a vote “AGAINST” the Warrant Amendment Proposal.
|
|
Q.
|
How does the SEPA Board recommend that I vote on the Warrant Holder Proposals?
|
|
A.
|
The SEPA Board has unanimously approved and unanimously recommends that the SEPA Public Warrant Holders entitled to vote on
the Warrant Holder Proposals, vote as follows:
|
|
•
|
“FOR” approval of the Warrant Amendment Proposal; and
|
|
•
|
“FOR” approval of the Warrant Holders Adjournment Proposal, if presented.
|
|
Q.
|
Will the Sponsor vote on the Warrant Holder Proposals?
|
|
A.
|
As of the Record Date, the Sponsor owns 100% of the outstanding SEPA Private Placement Warrants and none of the SEPA Public
Warrants. Because the Warrant Holder Proposals do not adversely affect the SEPA Private Placement Warrants in a different manner than the SEPA Public Warrants, or vice versa, the Sponsor is not entitled to vote on the Warrant Holder
Proposals.
|
|
Q.
|
When and where will the Warrant Holders Meeting be held?
|
|
A.
|
The Warrant Holders Meeting will be held in person or by proxy, on , 2023, in virtual format via live webcast at
https://www.cstproxy.com/sep-acquis/whm2023.
|
|
Q.
|
How many votes do I have?
|
|
A.
|
SEPA Public Warrant Holders have one vote per each SEPA Public Warrant held by them on , 2023.
|
|
Q.
|
How do I vote?
|
|
A.
|
If you are a record owner of your SEPA Public Warrants, there are three ways to vote your SEPA Public Warrants at the Warrant
Holders Meeting:
|
|
Q.
|
What if I do not vote my SEPA Public Warrants or if I abstain from voting?
|
|
A.
|
At the Warrant Holders Meeting, SEPA will count a properly executed proxy marked “ABSTAIN” with respect to a particular
Warrant Holder Proposal as present for purposes of determining whether a quorum is present. For purposes of approval, an abstention will have the same effect as a vote “AGAINST” the Warrant Amendment Proposal, but will have no effect on
the Warrant Holders Adjournment Proposal (if presented). However, if you do not submit a proxy or voting instruction, do not attend the Warrant Holders Meeting virtually or by proxy and your warrants are not otherwise voted at the
Warrant Holders Meeting, your failure to do so will have the same effect as a vote “AGAINST” the Warrant Amendment Proposal, but will have no effect on the Warrant Holders Adjournment Proposal (if presented).
|
|
Q.
|
If my warrants are held in “street name,” will my broker automatically vote them for me?
|
|
A.
|
No. Under the rules governing banks and brokers who submit a proxy card with respect to SEPA Public Warrants held in street
name, such banks and brokers have the discretion to vote on routine matters, but not on non-routine matters. The approval of the Warrant Amendment Proposal and Warrant Holders Adjournment Proposal (if presented) are non-routine matters.
|
|
•
|
NTA Proposal: The approval of the NTA Proposal will require the affirmative vote of
a majority of the outstanding shares of Common Stock as of the Record Date. Failure to vote by proxy or to vote virtually
|
|
•
|
Business Combination Proposal: The approval of the Business Combination Proposal
will require the affirmative vote of a majority of the outstanding shares of Common Stock as of the Record Date. Failure to vote by proxy or to vote virtually at the Stockholder Meeting or an abstention from voting will have the same
effect as a vote “AGAINST” the Business Combination Proposal. If any of the NTA Proposal, the Business Combination Proposal, the Charter Proposal, the Nasdaq Proposal, or the Incentive Plan Proposal fails to receive the required SEPA
Stockholder approval, the Business Combination will not be completed. Therefore, if any of the aforementioned Proposals are not approved, the Business Combination Proposal will have no effect, even if approved by the SEPA
Stockholders.
|
|
•
|
Charter Proposal: The approval of the Charter Proposal will require the affirmative
vote of a majority of the outstanding shares of Common Stock as of the Record Date. In addition, because the Charter Proposal will result in an increase in the number of authorized shares of Class A Common Stock, the Charter Proposal
will also require an affirmative vote by a majority of the outstanding shares of Class A Common Stock then outstanding as of the Record Date and voting separately as a single class. Failure to vote by proxy or to vote virtually at the
Stockholder Meeting or an abstention from voting will have the same effect as a vote “AGAINST” the Charter Proposal. Also, because they are cross-conditioned on each other, the Charter Proposal will be approved and adopted only if the
Business Combination Proposal is approved at the Stockholder Meeting by the affirmative vote of a majority of the outstanding shares of Common Stock as of the Record Date.
|
|
•
|
Organizational Documents Proposal: The approval of the Organizational Documents
Proposal will require the affirmative vote of a majority of the votes cast by SEPA Stockholders, present in person or represented by proxy, at the Stockholder Meeting and entitled to vote thereon. Failure to vote by proxy or to vote
virtually at the Stockholder Meeting or an abstention from voting will have no effect on the Organizational Documents Proposal. In addition, because they are cross-conditioned on each other, the Organizational Documents Proposal will
be approved and adopted only if the Business Combination Proposal is approved at the Stockholder Meeting by the affirmative vote of a majority of the outstanding shares of Common Stock as of the Record Date. As discussed above, the
Organizational Documents Proposals are advisory votes and therefore are not binding on SEPA or the SEPA Board. Furthermore, the Business Combination is not conditioned on the separate approval of the Organizational Documents Proposals
(separate and apart from approval of the Charter Proposal). Accordingly, regardless of the outcome of the non-binding advisory votes on the Organizational Documents Proposals, SEPA intends that the Proposed Charter will take effect
upon consummation of the Business Combination (assuming approval of the Charter Proposal).
|
|
•
|
Nasdaq Proposal: The approval of the Nasdaq Proposal will require the affirmative
vote of a majority of the votes cast by SEPA Stockholders, present in person or represented by proxy, at the Stockholder Meeting and entitled to vote thereon. Failure to vote by proxy or to vote virtually at the Stockholder Meeting or
an abstention from voting will have no effect on the Nasdaq Proposal. In addition, because they are cross-conditioned on each other, the Nasdaq Proposal will be approved and adopted only if the Business Combination Proposal is
approved at the Stockholder Meeting by the affirmative vote of a majority of the outstanding shares of Common Stock as of the Record Date.
|
|
•
|
Incentive Plan Proposal: The approval of the Incentive Plan Proposal will require
the affirmative vote of a majority of the votes cast by SEPA Stockholders, present in person or represented by proxy, at the Stockholder Meeting and entitled to vote thereon. Failure to vote by proxy or to vote virtually at the
Stockholder Meeting or an abstention from voting will have no effect on the Incentive Plan Proposal. In addition, because they are cross-conditioned on each other, the Incentive Plan Proposal will be approved and adopted only if the
Business Combination Proposal is approved at the Stockholder Meeting by the affirmative vote of a majority of the outstanding shares of Common Stock as of the Record Date.
|
|
•
|
Adjournment Proposal: The approval of the Adjournment Proposal will
require the affirmative vote of a majority of the votes cast by SEPA Stockholders, present in person or represented by proxy, at the Stockholder Meeting and entitled to vote thereon. Failure to vote by proxy or to vote virtually at
the Stockholder Meeting or an abstention from voting will have no effect on the Adjournment Proposal.
|
|
•
|
pursuant to the Letter Agreement, 5,031,250 Founder Shares owned by the Sponsor and other insiders became subject to a
lock-up whereby, subject to certain limited exceptions, they agreed not to transfer, assign or sell the Founder Shares until the earlier of (A) one year after the completion of SEPA’s initial business combination or (B) subsequent to
the Business Combination, (x) if the last sale price of SEPA’s Common Stock equals or exceeds $12.00 per share (as adjusted for stock splits, stock dividends, reorganizations, recapitalizations and other similar transactions) for any
20 trading days within any 30-trading day period commencing at least 150 days after SEPA’s initial business combination or (y) the date on which SEPA completes a liquidation, merger, capital stock exchange, reorganization or other
similar transaction that results in all of SEPA Stockholders having the right to exchange their shares of Common Stock for cash, securities or other property; provided, however, upon approval of the Letter Agreement Amendment, this
period will be amended to until the earlier of (i) 180 days after the completion of the Business Combination or (ii) subsequent to the Business Combination, the date on which SEPA completes a liquidation, merger, capital stock
exchange, reorganization or other similar transaction that results in all SEPA Stockholders having the right to exchange their shares of Common Stock for cash, securities or other property;
|
|
•
|
the Sponsor owns 2,415,375 shares of Class A Common Stock (the “Converted Shares”) (of which 1,746,316 shares will be
forfeited contingent upon and effective immediately prior to the Closing of the Business Combination pursuant to the Forfeiture and Redemption Agreement) and 1,050,000 Founder Shares, and SEPA’s officers and directors own 140,000
Founder Shares. On March 4, 2021, SEPA’s founder acquired 5,031,250 Founder Shares for an aggregate purchase price of $25,000, or approximately $0.005 per share. Also on March 24, 2021, SEPA’s founder assigned 160,000 Founder Shares
(40,000 Founder Shares each) to SEPA’s independent directors at their original purchase price, 35,000 Founder Shares (5,000 Founder Shares each) to SEPA’s advisors and 20,000 Founder Shares (10,000 Founder Shares each) to SEPA’s
Chief Financial Officer and Chief Strategy Officer. SEPA’s directors and officers also have a pecuniary interests in such Founder Shares through their ownership interest in the Sponsor. Upon conversion of the remaining Founder
Shares to Class A Common Stock and following the forfeiture of certain Converted Shares by the Sponsor pursuant to the Forfeiture and Non-Redemption Agreement, the Founder Shares and Converted Shares held by the Sponsor and SEPA’s
officers and directors would have an aggregate market value of approximately $10.5 million, based on the conversion ratio set forth in the Class B Charter Amendment and the last sale price of $10.55 per share of Class A Common Stock
on Nasdaq on December 20, 2023. If SEPA does not consummate the Business Combination or another initial business combination by July 30, 2024, and SEPA is therefore required to be liquidated, these shares would be worthless, as
Founder Shares are not entitled to participate in any redemption or liquidation of the Trust Account, and the Sponsor waived any right to the Trust Account with respect to the Converted Shares. Given the differential in the purchase
price that the Sponsor paid for the Founder Shares as compared to the price of the Units sold in SEPA’s IPO, the Sponsor and SEPA’s directors and officers may earn a positive rate of return on their investment even if SEPA’s Class A
Common Stock trades below the price initially paid for the Units in SEPA’s IPO and the public SEPA Stockholders experience a negative rate of return following the completion of the Business Combination;
|
|
•
|
the Sponsor and SEPA’s directors and officers may be incentivized to complete the Business Combination, or an alternative
initial business combination with a less favorable company or on terms less favorable to shareholders, rather than to liquidate, in which case the Sponsor would lose its entire investment. As a result, the Sponsor may have a conflict
of interest in determining whether SANUWAVE is an appropriate business with which to effectuate a business combination and/or in evaluating the terms of the Business Combination;
|
|
•
|
simultaneously with the closing of the IPO, the Sponsor purchased 7,850,000 SEPA Private Placement Warrants at a price of
$1.00 per warrant. Each private placement warrant is exercisable to purchase one share of Class A Common Stock at a price of $11.50 per share. Such SEPA Private Placement Warrants will expire worthless if SEPA fails to complete an
initial business combination by July 30, 2024;
|
|
•
|
pursuant to the Sponsor Voting Agreement, the Sponsor has agreed to vote all of the Sponsor’s shares of SEPA in favor of
the Merger Agreement and the Business Combination and to otherwise take (or not take, as applicable) certain other actions in support of the Merger Agreement and the Business Combination and the other matters to be submitted to SEPA
Stockholders for approval in connection with the Business Combination. The Sponsor Voting Agreement also prevents transfers of SEPA shares held by the Sponsor between the date of the Sponsor Voting Agreement and the termination of
such Sponsor Voting Agreement, except for certain permitted transfers where the recipient also agrees to comply with the Sponsor Voting Agreement;
|
|
•
|
on October 11, 2022, SEPA issued the Sponsor Note to the Sponsor, pursuant to which SEPA could borrow up to $1,000,000
at a 6% interest rate on or before October 11, 2024 to cover, among other things, expenses related to a business combination. As of December 20, 2023, SEPA has borrowed the full $1,000,000 under the Sponsor Note;
|
|
•
|
in order to finance transaction costs in connection with the Business Combination, the Sponsor or an affiliate of the
Sponsor, or certain of SEPA’s officers and directors may, but are not obligated to, loan SEPA additional funds as may be required. If SEPA completes the Business Combination, SEPA would repay the Working Capital Loans out of the
proceeds held in the Trust Account released to SEPA. Otherwise, the Working Capital Loans would be repaid only out of funds held outside the Trust Account. In the event that the Business Combination is not completed, SEPA may use a
portion of the proceeds held outside the Trust Account to repay the Working Capital Loans but no proceeds held in the Trust Account would be used to repay the Working Capital Loans;
|
|
•
|
the Sponsor has agreed that it will be liable to SEPA if and to the extent any claims by a third party (other than SEPA’s
independent auditors) for services rendered or products sold to SEPA, or a prospective target business with which SEPA has discussed entering into a transaction agreement, reduce the amount of funds in the Trust Account to below (i)
$10.10 per Public Share or (ii) such lesser amount per Public Share held in the Trust Account as of the date of the liquidation of the Trust Account, due to reductions in value of the Trust Account assets, in each case net of the
amount of interest which may be withdrawn to pay taxes as well as expenses relating to the administration of the Trust Account, except as to any claims by a third party who executed a waiver of any and all rights to seek access to the
Trust Account; and
|
|
•
|
the fact that SEPA has provisions in its Current Charter which waive the corporate opportunities doctrine, which means that
SEPA’s officers and directors have not been obligated to bring all corporate opportunities to SEPA. The potential conflict of interest relating to the waiver of the corporate opportunities doctrine in the Current Charter did not, to
SEPA’s knowledge, impact the search for an acquisition target or prevent SEPA from reviewing any opportunities as a result of such waiver.
|
|
•
|
“FOR” approval of the NTA Proposal;
|
|
•
|
“FOR” approval of the Business Combination Proposal;
|
|
•
|
“FOR” approval of the Charter Proposal;
|
|
•
|
“FOR” approval of the Organizational Documents Proposals;
|
|
•
|
“FOR” approval of the Nasdaq Proposal;
|
|
•
|
“FOR” approval of the Incentive Plan Proposal; and
|
|
•
|
“FOR” approval of the Adjournment Proposal, if presented.
|
|
•
|
“FOR” approval of the Warrant Amendment Proposal; and
|
|
•
|
“FOR” approval of the Warrant Holder Adjournment Proposal, if presented.
|
|
•
|
the Combined Company may be required to raise additional funds to finance operations and the Combined Company may not be
able to do so, and/or the terms of any financings may not be advantageous to the Combined Company;
|
|
•
|
SANUWAVE has a history of losses and the Combined Company may continue to incur losses and may not achieve or maintain
profitability;
|
|
•
|
the medical device/therapeutic product industries are highly competitive and subject to rapid technological change, so if
the Combined Company’s competitors are better able to develop and market products that are safer and more effective than any products the Combined Company may develop, the Combined Company’s commercial opportunities will be reduced or
eliminated;
|
|
•
|
if the Combined Company’s products and product candidates do not gain market acceptance among physicians, patients and
the medical community, the Combined Company may be unable to generate significant revenues, if any;
|
|
•
|
any product candidates of the Combined Company may not be developed or commercialized successfully;
|
|
•
|
the Combined Company may not successfully establish and maintain licensing and/or partnership arrangements for technology
for non-medical uses, which could adversely affect the Combined Company’s ability to develop and commercialize non-medical technology;
|
|
•
|
SANUWAVE’s product component materials are only produced by a single supplier for such product component. If the Combined
Company is unable to obtain product component materials and other products from SANUWAVE’s suppliers that the Combined Company will depend on for operations, or find suitable replacement suppliers, the Combined Company’s ability to
deliver products to market will likely be impeded, which could have a material adverse effect on the Combined Company;
|
|
•
|
SANUWAVE currently sells products through distributors and partners whose sales account for the majority of revenues and
accounts receivable. The Combined Company’s business and results of operations could be adversely affected by any business disruptions or credit or other financial difficulties experienced by such distributors or partners;
|
|
•
|
the Combined Company faces an inherent risk of liability in the event that the use or misuse of product candidates results
in personal injury or death;
|
|
•
|
actual or anticipated fluctuations in the Combined Company’s quarterly financial results or the quarterly financial
results of companies perceived to be similar to the Combined Company may negatively impact the trading price of the Combined Company’s securities;
|
|
•
|
the Combined Company will be dependent on information technology and the Combined Company’s systems and infrastructure face
certain risks, including from cybersecurity breaches and data leakage;
|
|
•
|
the Combined Company will generate a portion of revenue internationally and the Combined Company will be subject to various
risks relating to international activities which could adversely affect operating results;
|
|
•
|
results of Combined Company clinical trials may be insufficient to obtain regulatory approval for product candidates;
|
|
•
|
the Combined Company will be subject to extensive governmental regulation, including the requirement of FDA approval or
clearance, before any new product candidates may be marketed;
|
|
•
|
regulatory approval of the Combined Company’s product candidates may be withdrawn at any time;
|
|
•
|
federal regulatory reforms may adversely affect the Combined Company’s ability to sell products profitably;
|
|
•
|
failure to obtain regulatory approval in foreign jurisdictions may prevent the Combined Company from marketing products
abroad;
|
|
•
|
if the Combined Company fails to obtain an adequate level of reimbursement for approved products by third party payers,
there may be no commercially viable markets for approved products or the markets may be much smaller than expected;
|
|
•
|
uncertainty surrounding and future changes to healthcare law in the United States may have a material adverse effect on the
Combined Company;
|
|
•
|
if the Combined Company fails to comply with the United States Federal Anti-Kickback Statute, False Claims Act and similar
state laws, the Combined Company could be subject to criminal and civil penalties and exclusion from the Medicare and Medicaid programs, which would have a material adverse effect on the business and results of operations;
|
|
•
|
if the Combined Company fails to comply with the HIPAA Privacy, Security and Breach Notification Regulations, as such rules
become applicable to the Combined Company’s business, it may increase operational costs;
|
|
•
|
the Combined Company will face periodic reviews and billing audits from governmental and private payors and these audits
could have adverse results that may negatively impact the business;
|
|
•
|
product quality or performance issues may be discovered through ongoing regulation by the FDA and by comparable
international agencies, as well as through the Combined Company’s internal standard quality process;
|
|
•
|
the use of hazardous materials in Combined Company operation may subject the Combined Company to environmental claims or
liability;
|
|
•
|
the protection of the Combined Company’s intellectual property will be critical to the Combined Company’s success and any
failure on the Combined Company’s part to adequately protect those rights could materially adversely affect the business;
|
|
•
|
patent applications owned by or licensed to the Combined Company may not result in issued patents, and competitors may
commercialize discoveries the Combined Company attempts to patent;
|
|
•
|
the Combined Company’s patents may not be valid or enforceable and may be challenged by third parties;
|
|
•
|
issued patents and patent licenses may not provide the Combined Company with any competitive advantage or provide
meaningful protection against competitors;
|
|
•
|
the ability to market the products the Combined Company develops is subject to the intellectual property rights of third
parties;
|
|
•
|
changes in the market’s expectations about the Combined Company’s operating results;
|
|
•
|
success of competitors of the Combined Company;
|
|
•
|
the Combined Company’s operating results failing to meet the expectation of securities analysts or investors in a
particular period;
|
|
•
|
changes in financial estimates and recommendations by any securities analysts that may cover the Combined Company or the
industries in which the Combined Company operates in general;
|
|
•
|
operating and stock price performance of other companies that investors deem comparable to the Combined Company;
|
|
•
|
changes in laws and regulations affecting the Combined Company’s business;
|
|
•
|
commencement of, or involvement in, litigation involving the Combined Company;
|
|
•
|
changes in the Combined Company’s capital structure, such as future issuances of securities or the incurrence of additional
debt;
|
|
•
|
the volume of shares of Class A Common Stock available for public sale by the Combined Company;
|
|
•
|
any major change in the post-Closing board of directors or management of the Combined Company;
|
|
•
|
sales of substantial amounts of Common Stock by directors, executive officers or significant stockholders of the Combined
Company, or the perception that such sales could occur; and
|
|
•
|
general economic and political conditions such as recessions, pandemics, interest rates, fuel prices, international
currency fluctuations and acts of war or terrorism.
|
|
|
| |
For the Nine Months
Ended September 30,
|
| |
For the Year Ended
December 31,
|
| |
For the Period from
March 1, 2021
(inception) through
December 31,
|
|||
|
(in thousands)
|
| |
2023
|
| |
2022
|
| |
2022
|
| |
2021
|
|
Statement of Operations Data:
|
| |
|
| |
|
| |
|
| |
|
|
Loss from operations
|
| |
(1,764)
|
| |
(807)
|
| |
(1,174)
|
| |
(838)
|
|
Interest expense on promissory note — related party
|
| |
(55)
|
| |
—
|
| |
(2)
|
| |
—
|
|
Change in fair value of derivative liability
|
| |
(16)
|
| |
—
|
| |
—
|
| |
—
|
|
Expensed offering costs
|
| |
—
|
| |
—
|
| |
—
|
| |
(763)
|
|
Earnings on trading securities
|
| |
18
|
| |
—
|
| |
—
|
| |
—
|
|
Realized gain on investments held in Trust Account
|
| |
—
|
| |
521
|
| |
2,753
|
| |
—
|
|
Unrealized gain on investments held in Trust Account
|
| |
417
|
| |
679
|
| |
—
|
| |
30
|
|
Gain on waiver of deferred underwriting commissions by
underwriter
|
| |
300
|
| |
—
|
| |
—
|
| |
—
|
|
Gain from change in fair value of warrant liabilities
|
| |
—
|
| |
—
|
| |
6,643
|
| |
8,687
|
|
Unrealized (loss) gain from change in fair value of
warrant liabilities
|
| |
(755)
|
| |
6,473
|
| |
—
|
| |
—
|
|
Income tax expense
|
| |
—
|
| |
—
|
| |
(507)
|
| |
—
|
|
Net (loss) income
|
| |
(1,855)
|
| |
6,865
|
| |
7,713
|
| |
7,116
|
|
(in thousands)
|
| |
As of
September 30,
2023
|
| |
As of
December 31,
2022
|
|
Balance Sheet Data:
|
| |
|
| |
|
|
Total Current Assets
|
| |
$579
|
| |
$1,509
|
|
Total Assets
|
| |
14,248
|
| |
23,978
|
|
Total Liabilities
|
| |
3,893
|
| |
17,897
|
|
Class A common stock, $0.0001 par value, subject to
possible redemption; 1,304,259 shares at redemption value at September 30, 2023
|
| |
13,669
|
| |
13,333
|
|
Total stockholders’ deficit
|
| |
(3,314)
|
| |
(7,251)
|
|
|
| |
For the Nine Months Ended
September 30,
|
| |
For the Years Ended
December 31,
|
||||||
|
(in thousands)
|
| |
2023
|
| |
2022
|
| |
2022
|
| |
2021
|
|
Revenues:
|
| |
|
| |
|
| |
|
| |
|
|
Total Revenue
|
| |
$13,404
|
| |
$11,242
|
| |
$16,742
|
| |
$13,010
|
|
Cost of Revenues
|
| |
3,876
|
| |
3,141
|
| |
4,331
|
| |
4,986
|
|
Gross Margin
|
| |
9,528
|
| |
8,101
|
| |
12,411
|
| |
8,024
|
|
Operating Expenses:
|
| |
|
| |
|
| |
|
| |
|
|
General and administrative
|
| |
6,678
|
| |
9,484
|
| |
12,556
|
| |
11,690
|
|
Selling and marketing
|
| |
3,430
|
| |
5,037
|
| |
7,474
|
| |
8,591
|
|
Research and development
|
| |
436
|
| |
494
|
| |
567
|
| |
1,101
|
|
Depreciation and Amortization
|
| |
563
|
| |
575
|
| |
766
|
| |
784
|
|
Operating Loss
|
| |
(1,579)
|
| |
(7,489)
|
| |
(8,952)
|
| |
(14,142)
|
|
Other (Expense) Income, net
|
| |
(42,463)
|
| |
2,893
|
| |
(1,339)
|
| |
(13,089)
|
|
Income Tax Expense
|
| |
—
|
| |
—
|
| |
2
|
| |
28
|
|
Net Loss
|
| |
$(44,042)
|
| |
$(4,596)
|
| |
$(10,293)
|
| |
$(27,259)
|
|
(in thousands)
|
| |
As of
September 30,
2023
|
| |
As of
December 31,
2022
|
|
Total Current Assets
|
| |
$7,394
|
| |
$6,620
|
|
Total Assets
|
| |
20,342
|
| |
19,873
|
|
Total Liabilities
|
| |
86,296
|
| |
60,883
|
|
Total Stockholders’ Deficit
|
| |
(65,954)
|
| |
(41,010)
|
|
Total Liabilities and Stockholders’ Deficit
|
| |
20,342
|
| |
19,873
|
|
•
|
Assuming No Additional Redemptions: This pro forma presentation assumes that no
additional SEPA Stockholders exercise Redemption Rights with respect to their Class A Common Stock, subsequent to the redemptions that occurred on December 20, 2022.
|
|
•
|
Assuming Maximum Redemptions: This pro forma presentation assumes that 622,747
shares of Class A Common Stock subject to redemption are redeemed for an aggregate payment of approximately $6,526,686 (based on an estimated per share redemption price of approximately $10.48 that was calculated using the $16,669,258
in the Trust Account as of September 30, 2023 divided by 1,304,259 shares of Class A Common Stock subject to redemption assuming the pro forma maximum redemption scenario pursuant to the Merger Agreement). It is noted that 681,512
shares of Class A Common Stock are not being redeemed, pursuant to the Voting and Non-Redemption Agreements.
|
|
|
| |
Pro Forma Combined
|
|||
|
(in thousands, except share and per share amounts)
|
| |
Assuming No
Additional
Redemptions
|
| |
Assuming
Maximum
Redemptions
|
|
Summary Unaudited Pro Forma Condensed Combined
Statement of Operations Data For the Nine Months Ended September 30, 2023
|
| |
|
| |
|
|
Net loss
|
| |
$(9,693)
|
| |
$(9,693)
|
|
Net loss per share — basic and diluted
|
| |
$(0.86)
|
| |
$(0.87)
|
|
Weighted average shares outstanding of common stock - basic and diluted
|
| |
11,279,836
|
| |
11,175,577
|
|
|
| |
|
| |
|
|
Summary Unaudited Pro Forma Condensed Combined
Statement of Operations Data For the Year Ended December 31, 2022
|
| |
|
| |
|
|
Net loss
|
| |
$(9,980)
|
| |
$(9,980)
|
|
Net loss per share — basic and diluted
|
| |
$(0.88)
|
| |
$(0.89)
|
|
Weighted average shares outstanding of common stock - basic and diluted
|
| |
11,279,836
|
| |
11,175,577
|
|
|
| |
|
| |
|
|
Summary Unaudited Pro Forma Condensed Combined Balance
Sheet Data As of September 30, 2023
|
| |
|
| |
|
|
Total assets
|
| |
$28,717
|
| |
$27,375
|
|
Total liabilities
|
| |
$36,280
|
| |
$35,362
|
|
Total stockholders' deficit
|
| |
$(7,563)
|
| |
$(7,987)
|
|
Trading Date
|
| |
SNWV
Common
Stock (SNWV)
|
| |
SEPA Class A
Common
Stock (SEPA)
|
|
August 22, 2023
|
| |
$0.0185
|
| |
$10.62
|
|
December 20, 2023
|
| |
$0.0124
|
| |
$10.55
|
|
•
|
Prior to SEPA’s IPO, the Sponsor purchased 5,031,250 shares of Class B Common Stock for an aggregate purchase price of
$25,000, or approximately $0.005 per share. As a result of the significantly lower investment per share of the Sponsor as compared with the investment per share of SEPA Stockholders, a transaction which results in an increase in the
value of the investment of the Sponsor may result in a decrease in the value of the investment of SEPA Stockholders. In addition, if SEPA does not consummate a business combination by July 27, 2024, it would cease all operations except
for the purpose of winding up, redeeming all of the outstanding Public Shares for cash and, subject to the approval of its remaining shareholders and its board of directors, liquidating and dissolving, subject in each case to its
obligations under Delaware law to provide for claims of creditors and the requirements of other applicable law. In such event, the 5,031,250 shares of Class B Common Stock owned by the Sponsor would be worthless because following the
redemption of the Public Shares, SEPA would likely have few, if any, net assets if SEPA fails to complete a business combination within the required period. Additionally, in such event, the 7,850,000 SEPA Private Placement Warrants
purchased by the Sponsor simultaneously with the consummation of SEPA’s IPO for an aggregate purchase price of $7,850,000 and the Sponsor warrants (if issued at Closing at the option of the Sponsor) will also expire and be worthless.
SEPA’s directors and officers also have a direct or indirect economic interest in such SEPA Private Placement Warrants and in the 3,465,375 Class B Common Stock owned by the Sponsor.
|
|
•
|
SEPA’s existing directors and officers will be eligible for continued indemnification and continued coverage under SEPA’s
directors’ and officers’ liability insurance after the Business Combination and pursuant to the Merger Agreement.
|
|
•
|
The Sponsor and SEPA’s directors and officers may be incentivized to complete the Business Combination, or an alternative
initial business combination with a less favorable company or on terms less favorable to shareholders, rather than to liquidate, in which case the Sponsor would lose its entire investment. As a result, the Sponsor may have a conflict of
interest in determining whether SANUWAVE is an appropriate business with which to effectuate a business combination and/or in evaluating the terms of the Business Combination. The SEPA Board was aware of and considered these interests,
among other matters, in evaluating and unanimously approving the Business Combination and in recommending to SEPA Stockholders that they approve the Business Combination.
|
|
•
|
The fact that SEPA has provisions in its Current Charter which waive the corporate opportunities doctrine, which means that
SEPA’s officers and directors have not been obligated to bring all corporate opportunities to SEPA.
|
|
(i)
|
the date that is 180 days after the Closing; and
|
|
(ii)
|
the date on which the Combined Company completes a liquidation, merger, amalgamation, capital stock exchange, reorganization
or other similar transaction that results in all of the SEPA Stockholders having the right to exchange their shares of Class A Common Stock for cash, securities or other property.
|
|
•
|
its employees may experience uncertainty about their future roles, which might adversely affect the Combined Company’s
ability to retain and hire key personnel and other employees;
|
|
•
|
customers, suppliers, business partners and other parties with which SANUWAVE maintains business relationships may experience
uncertainty about its future and seek alternative relationships with third parties, seek to alter their business relationships with SANUWAVE or fail to extend an existing relationship with SANUWAVE; and
|
|
•
|
SANUWAVE continues to expand and will continue to expend significant costs, fees and expenses for professional services and
transaction costs in connection with the proposed Business Combination.
|
|
•
|
the Combined Company may be required to raise additional funds to finance operations and the Combined Company may not be
able to do so, and/or the terms of any financings may not be advantageous to the Combined Company;
|
|
•
|
SANUWAVE has a history of losses and the Combined Company may continue to incur losses and may not achieve or maintain
profitability;
|
|
•
|
the medical device/therapeutic product industries are highly competitive and subject to rapid technological change, so if the
Combined Company’s competitors are better able to develop and market products that are safer and more effective than any products the Combined Company may develop, the Combined Company’s commercial opportunities will be reduced or
eliminated;
|
|
•
|
if the Combined Company’s products and product candidates do not gain market acceptance among physicians, patients and the
medical community, the Combined Company may be unable to generate significant revenues, if any;
|
|
•
|
any product candidates of the Combined Company may not be developed or commercialized successfully;
|
|
•
|
the Combined Company may not successfully establish and maintain licensing and/or partnership arrangements for technology for
non-medical uses, which could adversely affect the Combined Company’s ability to develop and commercialize non-medical technology;
|
|
•
|
SANUWAVE’s product component materials are only produced by a single supplier for such product component. If the Combined
Company is unable to obtain product component materials and other products from SANUWAVE’s suppliers that the Combined Company will depend on for operations, or find suitable replacement suppliers, the Combined Company’s ability to
deliver products to market will likely be impeded, which could have a material adverse effect on the Combined Company;
|
|
•
|
SANUWAVE currently sells products through distributors and partners whose sales account for the majority of revenues and
accounts receivable. The Combined Company’s business and results of operations could be adversely affected by any business disruptions or credit or other financial difficulties experienced by such distributors or partners;
|
|
•
|
the Combined Company faces an inherent risk of liability in the event that the use or misuse of product candidates results in
personal injury or death;
|
|
•
|
actual or anticipated fluctuations in the Combined Company’s quarterly financial results or the quarterly financial
results of companies perceived to be similar to the Combined Company may negatively impact the trading price of the Combined Company’s securities;
|
|
•
|
the Combined Company will be dependent on information technology and the Combined Company’s systems and infrastructure face
certain risks, including from cybersecurity breaches and data leakage;
|
|
•
|
the Combined Company will generate a portion of revenue internationally and the Combined Company will be subject to various
risks relating to international activities which could adversely affect operating results;
|
|
•
|
results of Combined Company clinical trials may be insufficient to obtain regulatory approval for product candidates;
|
|
•
|
the Combined Company will be subject to extensive governmental regulation, including the requirement of FDA approval or
clearance, before any new product candidates may be marketed;
|
|
•
|
regulatory approval of the Combined Company’s product candidates may be withdrawn at any time;
|
|
•
|
federal regulatory reforms may adversely affect the Combined Company’s ability to sell products profitably;
|
|
•
|
failure to obtain regulatory approval in foreign jurisdictions may prevent the Combined Company from marketing products
abroad;
|
|
•
|
if the Combined Company fails to obtain an adequate level of reimbursement for approved products by third party payers, there
may be no commercially viable markets for approved products or the markets may be much smaller than expected;
|
|
•
|
uncertainty surrounding and future changes to healthcare law in the United States may have a material adverse effect on the
Combined Company;
|
|
•
|
if the Combined Company fails to comply with the United States Federal Anti-Kickback Statute, False Claims Act and similar
state laws, the Combined Company could be subject to criminal and civil penalties and exclusion from the Medicare and Medicaid programs, which would have a material adverse effect on the business and results of operations;
|
|
•
|
if the Combined Company fails to comply with the HIPAA Privacy, Security and Breach Notification Regulations, as such rules
become applicable to the Combined Company’s business, it may increase operational costs;
|
|
•
|
the Combined Company will face periodic reviews and billing audits from governmental and private payors and these audits
could have adverse results that may negatively impact the business;
|
|
•
|
product quality or performance issues may be discovered through ongoing regulation by the FDA and by comparable international
agencies, as well as through the Combined Company’s internal standard quality process;
|
|
•
|
the use of hazardous materials in Combined Company operation may subject the Combined Company to environmental claims or
liability;
|
|
•
|
the protection of the Combined Company’s intellectual property will be critical to the Combined Company’s success and any
failure on the Combined Company’s part to adequately protect those rights could materially adversely affect the business;
|
|
•
|
patent applications owned by or licensed to the Combined Company may not result in issued patents, and competitors may
commercialize discoveries the Combined Company attempts to patent;
|
|
•
|
the Combined Company’s patents may not be valid or enforceable and may be challenged by third parties;
|
|
•
|
issued patents and patent licenses may not provide the Combined Company with any competitive advantage or provide meaningful
protection against competitors;
|
|
•
|
the ability to market the products the Combined Company develops is subject to the intellectual property rights of third
parties;
|
|
•
|
changes in the market’s expectations about the Combined Company’s operating results;
|
|
•
|
success of competitors of the Combined Company;
|
|
•
|
the Combined Company’s operating results failing to meet the expectation of securities analysts or investors in a particular
period;
|
|
•
|
changes in financial estimates and recommendations by any securities analysts that may cover the Combined Company or the
industries in which the Combined Company operates in general;
|
|
•
|
operating and stock price performance of other companies that investors deem comparable to the Combined Company;
|
|
•
|
changes in laws and regulations affecting the Combined Company’s business;
|
|
•
|
commencement of, or involvement in, litigation involving the Combined Company;
|
|
•
|
changes in the Combined Company’s capital structure, such as future issuances of securities or the incurrence of additional
debt;
|
|
•
|
the volume of shares of Class A Common Stock available for public sale by the Combined Company;
|
|
•
|
any major change in the post-Closing board of directors or management of the Combined Company;
|
|
•
|
sales of substantial amounts of Common Stock by directors, executive officers or significant stockholders of the Combined
Company, or the perception that such sales could occur; and
|
|
•
|
general economic and political conditions such as recessions, pandemics, interest rates, fuel prices, international currency
fluctuations and acts of war or terrorism.
|
|
•
|
a requirement that the removal of directors for cause requires the affirmative vote of holders of at least two-thirds
(2/3) of the voting power of all then outstanding shares of capital stock of the Combined Company entitled to vote generally in the election of directors; and
|
|
•
|
Combined Company stockholders will no longer be able to take action by written consent, which would require all Combined
Company stockholder actions to be taken at a meeting of Combined Company stockholders; and
|
|
•
|
a requirement that to amend, alter, change or repeal certain provisions in the SEPA Bylaws requires the affirmative vote of
holders of at least two-thirds (2∕3) of the voting power of all then outstanding shares of capital stock of SEPA entitled to vote generally in the election of directors, which could
preclude SEPA Stockholders from bringing matters before annual or special meetings of SEPA Stockholders and delay changes in the Combined Company Board and also may inhibit the ability of an acquirer to effect such amendments to
facilitate an unsolicited takeover attempt.
|
|
•
|
Expertise and resources to analyze and properly apply U.S. GAAP to complex and non-routine transactions such as complex
financial instruments and derivatives and complex sales distributing agreements with select vendors.
|
|
•
|
A lack of internal resources to analyze and properly apply U.S. GAAP to accounting for financial instruments included in
service agreements with select vendors.
|
|
•
|
SANUWAVE has failed to design and implement controls around all accounting and IT processes and procedures and, as such,
SANUWAVE believes that all its accounting and IT processes and procedures need to be re-designed and tested for operating effectiveness.
|
|
•
|
unanticipated expenditures in research and development or manufacturing activities;
|
|
•
|
delayed market acceptance of any approved product;
|
|
•
|
unanticipated expenditures in the acquisition and defense of intellectual property rights;
|
|
•
|
the failure to develop strategic alliances for the marketing of some of SANUWAVE’s product candidates;
|
|
•
|
unforeseen changes in healthcare reimbursement for procedures using any of SANUWAVE’s approved products;
|
|
•
|
inability to train a sufficient number of physicians to create a demand for any of SANUWAVE’s approved products;
|
|
•
|
lack of financial resources to adequately support SANUWAVE’s operations;
|
|
•
|
difficulties in maintaining commercial scale manufacturing capacity and capability;
|
|
•
|
unforeseen problems with SANUWAVE’s third-party manufacturers, service providers or specialty suppliers of certain raw
materials;
|
|
•
|
unanticipated difficulties in operating in international markets;
|
|
•
|
unanticipated financial resources needed to respond to technological changes and increased competition;
|
|
•
|
unforeseen problems in attracting and retaining qualified personnel;
|
|
•
|
the impact of the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability
Reconciliation Act on SANUWAVE’s operations;
|
|
•
|
the impact of changes in U.S. health care law and policy on SANUWAVE’s operations;
|
|
•
|
enactment of new legislation or administrative regulations;
|
|
•
|
the application to SANUWAVE’s business of new court decisions and regulatory interpretations;
|
|
•
|
claims that might be brought in excess of SANUWAVE’s insurance coverage;
|
|
•
|
delays in timing of receipt of required regulatory approvals;
|
|
•
|
the failure to comply with regulatory guidelines; and
|
|
•
|
the uncertainty in industry demand and patient wellness behavior.
|
|
•
|
the reimbursement for its products is difficult to obtain or is too low, which can hinder the introduction and acceptance of
its products in the market;
|
|
•
|
it is unable to get its product candidates in commercial quantities at reasonable costs; and
|
|
•
|
the patient and physician community does not accept its product candidates.
|
|
•
|
required compliance with existing and changing foreign healthcare and other regulatory requirements and laws, such as those
relating to patient privacy or handling of bio-hazardous waste;
|
|
•
|
required compliance with anti-bribery laws, data privacy requirements, labor laws and anti-competition regulations;
|
|
•
|
export or import restrictions;
|
|
•
|
political and economic instability;
|
|
•
|
foreign exchange controls; and
|
|
•
|
difficulties protecting or procuring intellectual property rights.
|
|
•
|
advance notice of business to be brought is required for a meeting of SANUWAVE Stockholders;
|
|
•
|
no cumulative voting rights for the holders of SANUWAVE Common Stock in the election of directors; and
|
|
•
|
vacancies in the board of directors may be filled by the affirmative vote of a majority of directors then in office, even if
less than a quorum.
|
|
•
|
warning letters;
|
|
•
|
fines and other monetary penalties;
|
|
•
|
unanticipated expenditures;
|
|
•
|
product recall or seizure;
|
|
•
|
interruption of manufacturing;
|
|
•
|
operating restrictions;
|
|
•
|
injunctions; and
|
|
•
|
criminal prosecutions.
|
|
•
|
testing;
|
|
•
|
manufacturing;
|
|
•
|
quality control;
|
|
•
|
labeling;
|
|
•
|
advertising;
|
|
•
|
promotion;
|
|
•
|
distribution;
|
|
•
|
export;
|
|
•
|
reporting to the FDA certain adverse experiences associated with the use of the products; and
|
|
•
|
obtaining additional approvals or clearances for certain modifications to the products or their labeling or claims.
|
|
•
|
required refunding or retroactive adjustment of amounts SANUWAVE has been paid by governmental or private payors;
|
|
•
|
state or Federal agencies imposing fines, penalties and other sanctions on SANUWAVE;
|
|
•
|
loss of SANUWAVE’s right to participate in the Medicare program, state programs, or one or more private payor networks; or
|
|
•
|
damage to SANUWAVE’s business and reputation in various markets.
|
|
•
|
obtain and/or maintain protection for its products under the patent laws of the United States and other countries;
|
|
•
|
defend and enforce its patents once obtained;
|
|
•
|
obtain and/or maintain appropriate licenses to patents, patent applications or other proprietary rights held by others with
respect to its technology, both in the United States and other countries;
|
|
•
|
maintain trade secrets and other intellectual property rights relating to its products; and
|
|
•
|
operate without infringing upon the patents, trademarks, copyrights, and proprietary rights of third parties.
|
|
•
|
Within the U.S. and Canada: +1 800-450-7155 (toll-free)
|
|
•
|
Outside of the U.S. and Canada: +1 857-999-9155 (standard rates apply)
|
|
1.
|
A proposal to approve the NTA Amendment, which amendment shall be effective, if adopted and implemented by SEPA, prior to the
consummation of the proposed Business Combination, to remove from the Current Charter the redemption limitation contained under Section 9.2(a) of the Current Charter preventing SEPA from redeeming shares of Class A Common Stock, if it
would have less than $5,000,001 of net tangible assets. Please see the section entitled “Proposal 1: The NTA Proposal”;
|
|
2.
|
A proposal to approve the Merger Agreement and the transactions contemplated thereby, including the Business Combination,
pursuant to which Merger Sub will merge with and into SANUWAVE, with SANUWAVE continuing as the surviving entity of the Business Combination and becoming a subsidiary of SEPA as described in more detail in this proxy
statement/prospectus. Please see the section entitled “Proposal 2: The Business Combination Proposal”;
|
|
3.
|
A proposal to approve, in connection with the Merger, the replacement of the Current Charter with the Proposed Charter, in
the form appended to this proxy statement/prospectus as Annex D, to be effective upon the filing with and acceptance by the Delaware Secretary of State. Please see the section entitled “Proposal 3:
The Charter Proposal”;
|
|
4.
|
Proposals to vote upon, on a non-binding advisory basis, eight separate proposals with respect to certain governance
provisions in the Proposed Charter presented separately in accordance with SEC requirements. See the section entitled “Proposals 4 – 11: The Organizational Documents Proposals”;
|
|
5.
|
A proposal to approve, for purposes of complying with Nasdaq Listing Rules 5635(a), (b) and (d), the issuance of more than
20% of the issued and outstanding shares of Class A Common Stock and the resulting change in control in connection with the Business Combination, PIPE, and transactions contemplated thereby. Please see the section entitled “Proposal 12: The Nasdaq Proposal”;
|
|
6.
|
A proposal to approve the Incentive Plan, in the form appended to this proxy statement/prospectus as Annex E. Please
see the section entitled “Proposal 13: The Incentive Plan Proposal”; and
|
|
7.
|
A proposal to adjourn the Stockholder Meeting to a later date or dates, if necessary, to permit further solicitation and vote
of proxies if it is determined by the SEPA Board that more time is necessary or appropriate to approve one or more Proposals at the Stockholder Meeting. Please see the section entitled “Proposal 14: The
Adjournment Proposal”.
|
|
•
|
“FOR” approval of the NTA Proposal;
|
|
•
|
“FOR” approval of the Business Combination Proposal;
|
|
•
|
“FOR” approval of the Charter Proposal;
|
|
•
|
“FOR” approval of the Organizational Documents Proposals;
|
|
•
|
“FOR” approval of the Nasdaq Proposal;
|
|
•
|
“FOR” approval of the Incentive Plan Proposal; and
|
|
•
|
“FOR” approval of the Adjournment Proposal, if presented.
|
|
•
|
You Can Vote By Signing and Returning the Enclosed Proxy Card. If you vote by proxy
card, your “proxy,” whose name is listed on the proxy card, will vote your shares as you instruct on the proxy card. If you sign and return the proxy card but do not give instructions on how to vote your shares, your shares will be
voted as recommended by the SEPA Board “FOR” the NTA Proposal, the Business Combination Proposal, the Charter Proposal, the Organizational Documents Proposals, the Nasdaq Proposal, the Incentive Plan Proposal and the Adjournment
Proposal (if presented). Votes received after a matter has been voted upon at the Stockholder Meeting will not be counted.
|
|
•
|
You Can Vote Electronically. You can submit your proxy over the Internet in
accordance with the instructions on the enclosed proxy card.
|
|
•
|
You Can Attend the Stockholder Meeting and Vote in Person Virtually. You can attend
by logging into the website meeting portal with your control number. Once logged in you will be able to ask a question and or vote.
|
|
•
|
you may send another proxy card with a later date;
|
|
•
|
you may notify SEPA’s Secretary in writing before the Stockholder Meeting that you have revoked your proxy; or
|
|
•
|
you may attend the Stockholder Meeting and vote electronically by entering the control number found on your proxy card you
previously received. Attendance at the Stockholder Meeting will not, in and of itself, revoke a proxy.
|
|
(a)
|
Prior to the consummation of the initial Business Combination, the Corporation shall provide all holders of Offering Shares
with the opportunity to have their Offering Shares redeemed, out of the funds legally available therefor, upon the consummation of the initial Business Combination pursuant to, and subject to the limitations of, Section 9.2(b)
and Section 9.2(c) (such rights of such holders to have their Offering Shares redeemed pursuant to such Sections, the “Redemption Rights”) for cash equal to the
applicable redemption price per share determined in accordance with Section 9.2(b) (the “Redemption Price”) ;
provided, that the Corporation shall not redeem or repurchase Offering Shares to the
extent that such redemption would result in the Corporation’s failure to have net tangible assets (as determined in accordance with Rule 3a51-1(g)(1) of the Securities Exchange Act of 1934, as
amended (the “Exchange Act”) (or any successor rule)) in excess of
$5,000,000 or any greater net tangible asset or cash requirement upon consummation of the Corporation’s initial Business Combination which may be contained in the agreement relating to the initial
Business Combination (such limitation hereinafter called the “Redemption Limitation”). Notwithstanding anything to the contrary contained in this Amended and Restated Certificate, there shall be no Redemption Rights or liquidating
distributions with respect to any warrant issued pursuant to the Offering.
|
|
•
|
due incorporation, good standing, and that it has all requisite corporate power and authority to own, lease and operate its
properties and to carry on its business as now being conducted;
|
|
•
|
authorization, validity and enforceability of the Merger Agreement and power and authority to enter into the Merger Agreement
and to complete the transactions contemplated thereby;
|
|
•
|
capitalization, and in pertinent part, (i) the authorized amount of its equity interest and the amount issued and
outstanding, (ii) all outstanding equity interests have been duly authorized, are fully paid and non-assessable and not in violation of any purchase option, right of first refusal, preemptive right, subscription right or any similar
right under any provision of the BCA, any other applicable law, its organizational documents or any contract to which SANUWAVE is a party or by which it or its securities are bound, (iii) all of its securities have been granted,
offered, sold and issued in compliance with all
|
|
•
|
subsidiaries, and more specifically, each subsidiary’s jurisdiction of organization and capitalization;
|
|
•
|
except as described in certain disclosure schedules to the Merger Agreement, no conflict with any provision of its
organizational documents and no additional governmental approvals or filings or, except as has not been and would not reasonably be expected to have a Material Adverse Effect on SANUWAVE and its subsidiaries, any third-party consents
are required;
|
|
•
|
the filing of all forms, reports, schedules, statements, registration statements, prospectuses and other documents required
to be filed or furnished by the Combined Company with the SEC under the Securities Act and/or the Exchange Act, the compliance of such filings with the requirements of the Securities Act and the Exchange Act, as the case may be, and the
rules and regulations thereunder, and the failure, at the time such filing was made, of such filing to contain any untrue statement of a material fact or omit to state a material fact required to be stated therein or necessary in order
to make the statements made therein, in the light of the circumstances under which they were made, not misleading;
|
|
•
|
financial statements, indebtedness and the absence of undisclosed liabilities, and in pertinent part, (i) its financials (A)
were prepared in accordance with the books and records of SANUWAVE and its subsidiaries as of the times and for the periods referred to therein, (B) were prepared in accordance with GAAP, consistently applied throughout and among the
periods involved, (C) comply in all material respects with applicable accounting requirements under the Securities Act and the rules and regulations of the SEC thereunder, and (D) fairly present in all material respects the consolidated
financial position of SANUWAVE and its subsidiaries as of the respective dates thereof and the consolidated results of the operations and cash flows of SANUWAVE and its subsidiaries for the periods indicated; (ii) SANUWAVE and its
subsidiaries maintain accurate books and records reflecting their respective assets and liabilities and maintains proper and adequate internal accounting controls; (iii) all of the financial books and records of SANUWAVE and its
subsidiaries are complete and accurate in all material respects and have been maintained in the ordinary course consistent with past practice and in accordance with applicable laws, (iv) the absence of fraud or any written complaint,
allegation, assertion or claim regarding the accounting or auditing practices, procedures, methodologies or methods, or internal accounting controls of SANUWAVE and its subsidiaries, (v) except as described in certain disclosure
schedules to the Merger Agreement, SANUWAVE and its subsidiaries do not have any indebtedness; and (vi) except as described in certain disclosure schedules to the Merger Agreement, adequately reflected or reserved in SANUWAVE’s balance
sheet, incurred in the ordinary course of business or as would not, individually or in the aggregate, be expected to have a Material Adverse Effect, neither SANUWAVE nor any of its subsidiaries is subject to any liabilities or
obligations required to be reflected on a balance sheet prepared in accordance with GAAP;
|
|
•
|
the absence of a certain changes or events since December 31, 2022, and in pertinent part, each of SANUWAVE and its
subsidiaries, since December 31, 2022, has (i) conducted its business only in the ordinary course of business consistent with past practice, (ii) not been subject to a Material Adverse Effect, and (iii) not taken any action that would
be prohibited by the Merger Agreement if such action were taken on or after the date of the Merger Agreement without the consent of SEPA;
|
|
•
|
SANUWAVE’s and its subsidiaries’ compliance with laws, except for such non-compliance that would not reasonably be expected
to have a Material Adverse Effect;
|
|
•
|
permits, and in pertinent part, that each of SANUWAVE and its subsidiaries (and its employees who are legally required to be
licensed by a governmental authority in order to perform his or her duties with respect to his or her employment with any of SANUWAVE and its subsidiaries) holds all material permits necessary to lawfully conduct its business in all
material respects as presently conducted, and to own, lease and operate its assets and properties and all such material permits are in full force and effect, and no suspension or cancellation of any of such material permits is pending
or, to SANUWAVE’s knowledge, threatened, and none of SANUWAVE and its subsidiaries is in violation in any material respect of the terms of any such material permit, and none of SANUWAVE and its subsidiaries has received any written or,
to SANUWAVE’s knowledge, written notice of any actions relating to the revocation or material modification of any such material permit;
|
|
•
|
litigation, and in pertinent part, except as described in certain disclosure schedules to the Merger Agreement, (i) there is
no (a) action of any nature currently pending or, to SANUWAVE’s knowledge, threatened (and no such action has been brought or, to SANUWAVE’s knowledge, threatened in the past three (3) years), or (b) material order now pending or
outstanding or that was rendered by a governmental authority in the past three (3) years, in either case of (a) or (b) by or against any of SANUWAVE or its subsidiaries, its business, equity securities or assets, (ii) the items listed
on such disclosure schedules, if finally determined adversely to SANUWAVE or its subsidiaries, will not have, either individually or in the aggregate, a Material Adverse Effect upon any of SANUWAVE or its subsidiaries, and (iii) in the
past three (3) years, none of the current or former officers, senior management or directors of any of SANUWAVE or its subsidiaries have been charged with, indicted for, arrested for, or convicted of any felony or any crime involving
fraud;
|
|
•
|
material contracts, and in pertinent part, (i) any contracts that (a) contain covenants that materially limit the ability of
any of SANUWAVE or its subsidiaries (I) to compete in any line of business or with any person or in any geographic area or to sell, or provide any service or product, including any non-competition covenants, exclusivity restrictions,
rights of first refusal or most-favored pricing clauses or (II) to purchase or acquire an interest in any other person, (b) involves any joint venture, profit-sharing, partnership, limited liability company or other similar agreement or
arrangement relating to the formation, creation, operation, management or control of any partnership or joint venture, (c) involves any exchange traded, over the counter or other swap, cap, floor, collar, futures contract, forward
contract, option or other derivative financial instrument or contract, based on any commodity, security, instrument, asset, rate or index of any kind or nature whatsoever, whether tangible or intangible, including currencies, interest
rates, foreign currency and indices, (d) evidences indebtedness (whether incurred, assumed, guaranteed or secured by any asset) of any of SANUWAVE or its subsidiaries having an outstanding principal amount in excess of $500,000, (e)
involves the acquisition or disposition, directly or indirectly (by merger or otherwise), of assets with an aggregate value in excess of $500,000 (other than in the ordinary course of business consistent with past practice) or shares or
other equity interests of any of SANUWAVE or its subsidiaries or another person, (f) relates to any merger, consolidation or other business combination, with any other person or the acquisition or disposition of any other entity or its
business or material assets or the sale of any of SANUWAVE or its subsidiaries, its business or material assets, (g) by its terms, individually or with all related contracts, calls for aggregate payments or receipts by of SANUWAVE and
its subsidiaries under such contract or contracts of at least $500,000 in the 12-month period ended December 31, 2022, or during the 12-month period ending December 31, 2023, (h) is with any top customer or supplier, (i) is between any
of SANUWAVE or its subsidiaries and any present or former directors, officers, contractors or employees of SANUWAVE or its subsidiaries (other than at-will employment or consulting arrangements, confidentiality, non-solicitation,
non-competition, or intellectual property assignment agreements with employees and contractors entered into in the ordinary course of business consistent with past practice), including all non-competition, severance and indemnification
agreements, or any such related person, (j) obligates SANUWAVE or its subsidiaries to make any capital commitment or expenditure in excess of $500,000 (including pursuant to any joint venture), (k) relates to a material settlement in
excess of $500,000 entered into within two years prior to the date of Merger Agreement under which any of SANUWAVE or its subsidiaries has outstanding obligations other than customary confidentiality obligations, (l) provides another
person (other than another of SANUWAVE or its subsidiaries or any
|
|
•
|
intellectual property, and in pertinent part, (i) each of SANUWAVE and its subsidiaries owns, free and clear of all liens
(other than permitted liens), has valid and enforceable rights to use pursuant to valid contracts or licenses, or otherwise has the right to use all intellectual property currently used, licensed or held for use by such entity, except
as would not, individually or in the aggregate, reasonably be expected to have a Material Adverse Effect; (ii) the transactions do not and will not impair any of SANUWAVE’s and its subsidiaries’ intellectual property, except as would
not, individually or in the aggregate, reasonably be expected to have a Material Adverse Effect; (iii) none of SANUWAVE and its subsidiaries intellectual property has been found to be unenforceable or is the subject of any cancellation
or reexamination proceeding, and all registered intellectual property is valid and enforceable, except as would not, individually or in the aggregate, reasonably be expected to have a Material Adverse Effect; (iv) as of the date of the
Merger Agreement, no action is pending or, to SANUWAVE’s knowledge, threatened against SANUWAVE or its subsidiaries infringes upon or misappropriates or otherwise violates the intellectual property rights of any third party, except as
would not, individually or in the aggregate, reasonably be expected to have a Material Adverse Effect; (v) SANUWAVE and its subsidiaries taken reasonable steps in accordance with normal industry practice to maintain and protect the
confidentiality of all material intellectual property, except as would not, individually or in the aggregate, reasonably be expected to have a Material Adverse Effect; (vi) all persons who have contributed to the creation of any
intellectual property of SANUWAVE and its subsidiaries has assigned its rights to such intellectual property to SANUWAVE and its subsidiaries, except as would not, individually or in the aggregate, reasonably be expected to have a
Material Adverse Effect; (vii) the absence of any data breach; (viii) the compliance in all material respects, with all data privacy and security laws and contract requirements; and (ix) the absence of any indemnification obligations
relating to infringement;
|
|
•
|
taxes and tax returns;
|
|
•
|
real property;
|
|
•
|
personal property;
|
|
•
|
title to and sufficiency of SANUWAVE’s assets;
|
|
•
|
employee matters, and in pertinent part, (i) none of SANUWAVE or its subsidiaries is a party to any collective bargaining
agreement or other contract covering any group of employees, labor organization or other representative of any of the employees of any of SANUWAVE or its subsidiaries, and SANUWAVE has no knowledge of any activities or proceedings of
any labor union or other party to organize or represent such employees; (ii) there has not occurred or, to the knowledge of SANUWAVE, been threatened any
|
|
•
|
benefits plans, and in pertinent part, (i) with respect to each SANUWAVE benefit plan, there are no funded benefit
obligations for which contributions have not been made, or properly accrued and there are no unfunded benefit obligations that have not been accounted for by reserves, or otherwise properly footnoted in accordance with GAAP on the
company financials, except as would not, individually or in the aggregate, reasonably be expected to have a Material Adverse Effect; (ii) each company benefit plan is and has been operated at all times in compliance with all applicable
laws, including ERISA and the Code, except for any non-compliance that would not be reasonably expected to have a Material Adverse Effect; (iii) with respect to each SANUWAVE benefit plan: (A) to the knowledge of SANUWAVE, no breach of
fiduciary duty that could reasonably be expected to result in liability to SANUWAVE or any of its subsidiaries has occurred; (B) no action is pending, or to SANUWAVE’s knowledge, threatened; and (C) no prohibited transaction, as defined
in Section 406 of ERISA or Section 4975 of the Code, has occurred, except as would not, individually or in the aggregate, reasonably be expected to have a Material Adverse Effect; (iv) for the six-year period preceding the Effective
Time, no company benefit plan is a “defined benefit plan” (as defined in Section 414(j) of the Code), a “multiemployer plan” (as defined in Section 3(37) of ERISA) or a “multiple employer plan” (as described in Section 413(c) of the
Code) or is otherwise subject to Title IV of ERISA or Section 412 of the Code, and none of SANUWAVE or its subsidiaries has any outstanding liability under Title IV of ERISA and, to the knowledge of SANUWAVE, no condition presently
exists that is expected to cause such liability to be incurred; (v) the consummation of the transactions contemplated by the Merger Agreement and the ancillary documents will not: (A) entitle any individual to severance pay,
unemployment compensation or other benefits or compensation; or (B) accelerate the time of payment or vesting, or increase the amount of any compensation due, or in respect of, any individual; (vi) compliance with Section 409A of the
Code in all material respects; and (vii) the due qualification of Company Options intended to qualify as “incentive stock options”;
|
|
•
|
environmental matters;
|
|
•
|
transactions with related parties;
|
|
•
|
insurance;
|
|
•
|
books and records;
|
|
•
|
top customers and suppliers;
|
|
•
|
certain business practices;
|
|
•
|
none of SANUWAVE or its subsidiaries is an “investment company” or a person directly or indirectly “controlled” by or acting
on behalf of an “investment company”, or required to register as an “investment company”, in each case within the meaning of the Investment Company Act of 1940, as amended;
|
|
•
|
except as described in certain disclosure schedules to the Merger Agreement, none of SANUWAVE or its subsidiaries has
incurred or will incur any liability for any brokerage, finder’s or other fee or commission in connection with the transactions contemplated by the Merger Agreement;
|
|
•
|
healthcare industry matters;
|
|
•
|
product liability matters;
|
|
•
|
SANUWAVE’s Board taking action to ensure that the restrictions on business combinations contained in Section 203 of the DGCL
or contained in Section 78.411 through Section 78.444 of the BCA will not apply to the Merger Agreement, the Merger and the other transactions contemplated by the Merger Agreement, and the absence of a stockholder rights plan, “poison
pill” anti-takeover plan or other similar plan, device or arrangement;
|
|
•
|
independent investigation, and in pertinent part, SANUWAVE conducted its own independent investigation, review and analysis
of the business, results of operations, prospects, condition (financial or otherwise) or assets of SEPA;
|
|
•
|
none of the information supplied or to be supplied by SANUWAVE expressly for inclusion in any filing made with any
governmental authority or stock exchange with respect to the transactions contemplated by the Merger Agreement, this proxy statement/prospectus or in the mailings or other distributions to SEPA Stockholders and/or prospective
investors with respect to the consummation of the transactions contemplated by the Merger Agreement, will contain any untrue statement of a material fact or omit to state any material fact required to be stated therein or necessary in
order to make the statements therein, in light of the circumstances under which they are made, not misleading; and
|
|
•
|
no representations other than those specifically provided in the Merger Agreement.
|
|
•
|
due incorporation, good standing, and that it has all requisite corporate power and authority to own, lease and operate its
properties and to carry on its business as now being conducted;
|
|
•
|
authorization, validity and enforceability of the Merger Agreement, and power and authority to enter into the Merger
Agreement and to complete the transactions contemplated thereby;
|
|
•
|
except as described in certain disclosure schedules to the Merger Agreement, no conflict with any provision of its
organizational documents and no additional governmental approvals or filings or, except as has not been and would not reasonably be expected to have a Material Adverse Effect on SEPA, third-party consents required;
|
|
•
|
capitalization, and in pertinent part, (i) the authorized amount of its equity interest and the amount issued and
outstanding, (ii) all outstanding equity interests have been duly authorized, validly issued, fully paid and non-assessable and are not subject to or issued in violation of any purchase option, right of first refusal, preemptive right,
subscription right or any similar right under any provision of the DGCL, its organizational documents or any contract to which it is a party, (iii) except as set forth in certain disclosure schedules, there are no (a) equity
appreciation, phantom equity or similar rights, (b) outstanding options, warrants, puts, calls, convertible securities, preemptive or similar rights other than the Redemption, (c) bonds, debentures, notes or other indebtedness having
general voting rights or that are convertible or exchangeable into securities having such rights or (d) subscriptions or other rights, agreements, arrangements, contracts or commitments of any character (other than the Merger Agreement
and the ancillary documents), (I) relating to the issued or unissued shares of SEPA or (II) obligating SEPA to issue, transfer, deliver or sell or cause to be issued, transferred, delivered, sold or repurchased any options or shares or
securities convertible into or exchangeable for such shares, (III) obligating SEPA to grant, extend or enter into any such option, warrant, call, subscription or other right, agreement, arrangement or commitment for such capital shares,
or (IV) other than the Redemption or as expressly set forth in the Merger Agreement, obligating SEPA to redeem, repurchase or otherwise acquire any such shares or other equity interests, or provide an amount of funds to, or make any
investment (in the form of a loan, capital contribution or otherwise) in SEPA or any other person, (iv) except as set forth in certain disclosure schedules, there are no stockholders agreements, voting trusts or other agreements or
understandings to which SEPA is a party with respect to the voting of any shares of SEPA, (v) all Indebtedness (as defined
|
|
•
|
SEC filings and SEPA financials, and in pertinent part, (i) since SEPA’s IPO, it has filed all forms, reports, schedules,
statements, registration statements, prospectuses and other documents required to be filed or furnished by SEPA with the SEC under the Securities Act of 1933, as amended, and/or the Exchange Act of 1934, as amended, together with any
amendments, restatements or supplements thereto, and will file all such forms, reports, schedules, statements and other documents required to be filed subsequent to the date of the Merger Agreement, (ii) except for any changes to
SEPA’s historical accounting of SEPA’s warrants as equity rather than as liabilities that may be required as a result of the Staff Statement on Accounting and Reporting Considerations for Warrants Issued by Special Purpose Acquisition
Companies that was issued by the SEC on April 12, 2021 (the “SEC April Warrant Statement”), and related guidance by the SEC, SEPA’s financials fairly present in all material respects the financial position and the results of
operations, changes in stockholders’ equity, and cash flows of SEPA at the respective dates of and for the periods referred to in such financial statements, all in accordance with (a) GAAP methodologies applied on a consistent basis
throughout the periods involved, (b) Regulation S-X or Regulation S-K, as applicable (except as may be indicated in the notes thereto and for the omission of notes and audit adjustments in the case of unaudited quarterly financial
statements to the extent permitted by Regulation S-X or Regulation S-K, as applicable) and (c) the books and records of SEPA as of the times and for the periods referred to therein, and (iii) except for any changes to SEPA’s
historical accounting of SEPA’s warrants as equity rather than as liabilities that may be required as a result of the SEC April Warrant Statement, and as and to the extent reflected or reserved against in SEPA’s financials, SEPA has
not incurred any liabilities or obligations of the type required to be reflected on a balance sheet in accordance with GAAP that are not adequately reflected or reserved on or provided for in SEPA’s financials, other than liabilities
of the type required to be reflected on a balance sheet in accordance with GAAP that have been incurred since the most recent date of SEPA’s financials in the ordinary course of business;
|
|
•
|
absence of certain changes, and in pertinent part, SEPA has (i) since its formation, conducted no business other than its
formation, the public offering of its securities (and the related private offerings), public reporting and its search for an initial Business Combination as described in SEPA’s IPO prospectus (including the investigation of SANUWAVE and
its subsidiaries and the negotiation and execution of the Merger Agreement) and related activities, (ii) since December 31, 2022, has not been subject to a Material Adverse Effect on SEPA;
|
|
•
|
compliance with laws;
|
|
•
|
actions; orders and permits, and in pertinent part, (i) there is no pending or, to the knowledge of SEPA, threatened action
to which SEPA is subject which would reasonably be expected to have a Material Adverse Effect on SEPA (and no such action has been brought in the past three (3) years, or to the knowledge of SEPA, threatened in the past three (3)
years), (ii) SEPA is not subject to any material orders of any governmental authority, nor are any such orders pending, (iii) SEPA holds all material permits necessary to lawfully conduct its business as presently conducted, and to own,
lease and operate its assets and properties, all of which are in full force and effect, except where the failure to hold such permit or for such permit to be in full force and effect would not reasonably be expected to have a Material
Adverse Effect on SEPA, and (iv) none of the current or former officers, senior management or directors of SEPA have been charged with, indicted for, arrested for, or convicted of any felony or any crime involving fraud;
|
|
•
|
taxes and tax returns;
|
|
•
|
SEPA does not have any paid employees or maintain, sponsor, contribute to or otherwise have any liability under any benefit
plans;
|
|
•
|
SEPA does not own, license or otherwise have any right, title or interest in any material intellectual property and does not
own or lease any material real or personal property;
|
|
•
|
material contracts, and in pertinent part, (i) except as set forth on certain disclosure schedules, other than the Merger
Agreement and the ancillary documents, there are no contracts to which SEPA is a party or by which any of its properties or assets may be bound, subject or affected, which (a) creates or imposes a liability greater than $200,000, (b)
may not be cancelled by SEPA on less than sixty (60) days’ prior notice without payment of a material penalty or termination fee or (c) prohibits, prevents, restricts or impairs in any material respect any business practice of SEPA as
its business is currently conducted, any acquisition of material property by SEPA, or restricts in any material respect the ability of SEPA to engage in business as currently conducted by it or compete with any other person (each, a
“SEPA Material Contract”), (ii) all SEPA Material Contracts have been made available to SANUWAVE other than those that are exhibits to SEPA’s SEC reports, (iii) with respect to each SEPA Material Contract (a) the SEPA Material Contract
was entered into at arms’ length and in the ordinary course of business, (b) the SEPA Material Contract is legal, valid, binding and enforceable in all material respects against SEPA and, to the Knowledge of SEPA, the other parties
thereto, and is in full force and effect (except, in each case, as such enforcement may be limited by certain exceptions to enforceability), (c) SEPA is not in breach or default in any material respect, and no event has occurred that
with the passage of time or giving of notice or both would constitute such a breach or default in any material respect by SEPA, or permit termination or acceleration by the other party, under such SEPA Material Contract, and (d) to the
knowledge of SEPA, no other party to SEPA Material Contract is in breach or default in any material respect, and no event has occurred that with the passage of time or giving of notice or both would constitute such a breach or default
by such other party, or permit termination or acceleration or acceleration by SEPA under any SEPA Material Contract;
|
|
•
|
transactions with affiliates;
|
|
•
|
Merger Sub activities, and in pertinent part, Merger Sub has not engaged in any business activities other than as
contemplated by the Merger Agreement, does not own directly or indirectly any ownership, equity, profits or voting interest in any person and has no assets or liabilities except those incurred in connection with the Merger Agreement and
the ancillary documents to which it is a party, and is not a party to or bound by any contract;
|
|
•
|
SEPA is not an “investment company” or a person directly or indirectly “controlled” by or acting on behalf of an “investment
company”, or required to register as an “investment company”, in each case within the meaning of the Investment Company Act of 1940, as amended;
|
|
•
|
No broker, finder or investment banker is entitled to any brokerage, finder’s or other fee or commission from SEPA, SANUWAVE
or any of their respective affiliates in connection with the transactions contemplated by the Merger Agreement except as set forth on the disclosure schedules;
|
|
•
|
All Common Stock to be issued and delivered to SANUWAVE Stockholders shall be full paid and non-assessable, free and clear of
all liens, other than restrictions arising from applicable laws, applicable lock-up agreement, and any liens incurred by SANUWAVE Stockholders, and the issuance and transfer of such Common Stock pursuant to the Merger Agreement will not
be subject to or give rise to any purchase option, right of first refusal, preemptive right, subscription right or any similar right under any provision of the DGCL or any other applicable Law, SEPA’s organizational documents or any
contract to which SEPA is a party;
|
|
•
|
certain business practices;
|
|
•
|
insurance;
|
|
•
|
SEPA Trust Account, and in pertinent part as of June 30, 2023, the SEPA Trust Account has a specified balance and such monies
are invested solely in United States “government securities” within the meaning of Section 2(a)(16) of the Investment Company Act of 1940, as amended, or money market funds meeting certain conditions under Rule 2a-7 promulgated under
the Investment Company Act of 1940, as amended, and held in trust by the Transfer Agent pursuant to the Trust Agreement, which is valid and in full force and effect and enforceable in accordance with its terms (subject to certain
exceptions to enforceability) and has not been amended or modified;
|
|
•
|
The SEPA Board taking action to ensure that the restrictions on business combinations contained in Section 203 of the DGCL or
contained in Section 78.411 through Section 78.444 of the BCA will not apply to the Merger Agreement, the Merger and the other transactions contemplated by the Merger Agreement, and the absence of a stockholder rights plan, “poison
pill” anti-takeover plan or other similar plan, device or arrangement;
|
|
•
|
the delivery of an opinion of ValueScope to the effect that, as of the date of such opinion, and based on and subject to the
assumptions, limitations, qualifications and other matters set forth therein, the Merger is fair, from a financial point of view, to holders of Class A and Class B Common Stock;
|
|
•
|
independent investigation, and in pertinent part, SEPA conducted its own independent investigation, review and analysis of
the business, results of operations, prospects, condition (financial or otherwise) or assets of SANUWAVE and its subsidiaries;
|
|
•
|
none of the information supplied or to be supplied by SEPA expressly for inclusion in any filing made with any governmental
authority or stock exchange with respect to the transactions contemplated by the Merger Agreement, this Registration Statement or in the mailings or other distributions to SEPA Stockholders and/or prospective investors with respect to
the consummation of the transactions contemplated by the Merger Agreement, will contain any untrue statement of a material fact or omit to state any material fact required to be stated therein or necessary in order to make the
statements therein, in light of the circumstances under which they are made, not misleading; and
|
|
•
|
no other representations.
|
|
•
|
amend, waive or otherwise change, in any respect, its organizational documents, except as required by applicable law;
|
|
•
|
authorize for issuance, issue, grant, sell, pledge, dispose of or propose to issue, grant, sell, pledge or dispose of any of
its equity securities or any options, warrants, commitments, subscriptions or rights of any kind to acquire or sell any of its equity securities, or other securities, including any securities convertible into or exchangeable for any of
its shares or other equity securities or securities of any class and any other equity-based awards, except for certain exceptions as contemplated by the Merger Agreement, or engage in any hedging transaction with a third Person with
respect to such securities;
|
|
•
|
split, combine, recapitalize or reclassify any of its shares or other equity interests or issue any other securities in
respect thereof or pay or set aside any dividend or other distribution (whether in cash, equity or property or any combination thereof) in respect of its equity interests, or directly or indirectly redeem, purchase or otherwise acquire
or offer to acquire any of its securities;
|
|
•
|
incur, create, assume, prepay or otherwise become liable for any indebtedness (directly, contingently or otherwise) in excess
of $500,000 individually or $1,000,000 in the aggregate, make a loan or advance to or investment in any third party (other than advancement of expenses to employees in the ordinary course of business), or guarantee or endorse any
indebtedness, liability or obligation of any person in excess of $500,000 individually or $1,000,000 in the aggregate;
|
|
•
|
other than as required by applicable law or pursuant to the terms of any SANUWAVE benefit plans (A) increase the wages,
salaries or compensation of its senior vice presidents and above other than in the ordinary course of business, consistent with past practice, (B) make or commit to make any bonus payment (whether in cash, property or securities) other
than in the ordinary course of business consistent with past practice, (C) materially increase other benefits of employees generally other than in the ordinary course of business, or (D) enter into, establish, materially amend or
terminate any SANUWAVE benefit plan with, for or in respect of any current consultant, officer, manager director or other employee, other than in the ordinary course of business consistent with past practice;
|
|
•
|
make or rescind any material election relating to taxes, settle any claim, action, suit, litigation, proceeding, arbitration,
investigation, audit or controversy relating to material taxes, file any amended tax return or claim for refund, or make any material change in its method of tax accounting, in each case except as required by applicable law or in
compliance with GAAP;
|
|
•
|
take any action, or knowingly fail to take any action, which action or failure to act would reasonably be expected to prevent
or impede the Merger from qualifying as a “reorganization” within the meaning of Section 368(a) of the Code (and the Treasury Regulations) or, except as contemplated by the Merger Agreement, take any action, which action would
reasonably be expected to prevent or impede any other aspect of the intended tax treatment of the Merger;
|
|
•
|
transfer or license to any person or otherwise extend, materially amend or modify, permit to lapse or fail to preserve any
material SANUWAVE owned intellectual property (excluding lapses or terminations of contracts pursuant to the terms thereof), or disclose to any Person who has not entered into a confidentiality agreement any trade secrets;
|
|
•
|
terminate, waive or assign any material right under any SANUWAVE material contract or enter into any contract that would be a
SANUWAVE material contract, in any case outside of the ordinary course of business consistent with past practice;
|
|
•
|
fail to maintain its books, accounts and records in all material respects in the ordinary course of business consistent with
past practice;
|
|
•
|
establish any subsidiary or enter into any new line of business;
|
|
•
|
fail to use commercially reasonable efforts to keep in force material insurance policies or replacement or revised policies
providing insurance coverage with respect to its assets, operations and activities in such amount and scope of coverage substantially similar to that which is currently in effect;
|
|
•
|
revalue any of its material assets or make any material change in accounting methods, principles or practices, except to the
extent required to comply with GAAP and after consulting with the SANUWAVE’s outside auditors;
|
|
•
|
waive, release, assign, settle or compromise any claim, action or proceeding (including any suit, action, claim, proceeding
or investigation relating to the Merger Agreement or the transactions contemplated hereby), other than waivers, releases, assignments, settlements or compromises that involve only the payment of monetary damages (and not the imposition
of equitable relief on, or the admission of wrongdoing by SANUWAVE, its subsidiaries or its Affiliates) not in excess of $500,000 individually or $1,000,000 in the aggregate;
|
|
•
|
close or materially reduce its activities, or effect any layoff or other personnel reduction or change, at any of its
facilities;
|
|
•
|
acquire, including by merger, consolidation, acquisition of equity interests or assets, or any other form of business
combination, any corporation, partnership, limited liability company, other business organization or any division thereof, or any material amount of assets outside the ordinary course of business consistent with past practice;
|
|
•
|
make capital expenditures in excess of $500,000 individually for any project (or set of related projects) or $1,000,000 in
the aggregate;
|
|
•
|
voluntarily incur any liability or obligation (whether absolute, accrued, contingent or otherwise) in excess of $1,000,000
individually or $2,000,000 in the aggregate (excluding the incurrence of any expenses) other than pursuant to the terms of a SANUWAVE material contract or SANUWAVE benefit in existence at the date of the Merger Agreement or entered into
in the ordinary course of business;
|
|
•
|
adopt a plan of complete or partial liquidation, dissolution, Business Combination, consolidation, restructuring,
recapitalization or other reorganization;
|
|
•
|
enter into any agreement, understanding or arrangement with respect to the voting of equity securities of SANUWAVE;
|
|
•
|
take any action that would reasonably be expected to significantly delay or impair the obtaining of any consents of any
governmental authority to be obtained in connection with the Merger Agreement;
|
|
•
|
except as otherwise required by the Merger Agreement, enter into, amend, waive or terminate (other than terminations in
accordance with their terms) any transaction with any related person (other than compensation and benefits and advancement of expenses, in each case, provided in the ordinary course of business consistent with past practice); or
|
|
•
|
authorize or agree to do any of the foregoing actions.
|
|
•
|
amend, waive or otherwise change, in any respect, its organizational documents except as required by applicable law;
|
|
•
|
authorize for issuance, issue, grant, sell, pledge, dispose of or propose to issue, grant, sell, pledge or dispose of any of
its equity securities or any options, warrants, commitments, subscriptions or rights of any kind to acquire or sell any of its equity securities, or other securities, including any securities convertible into
|
|
•
|
split, combine, recapitalize or reclassify any of its shares or other equity interests or issue any other securities in
respect thereof or pay or set aside any dividend or other distribution (whether in cash, equity or property or any combination thereof) in respect of its shares or other equity interests, or directly or indirectly redeem, purchase or
otherwise acquire or offer to acquire any of its securities;
|
|
•
|
incur, create, assume, prepay or otherwise become liable for any indebtedness (directly, contingently or otherwise) in excess
of $500,000 individually or $1,000,000 in the aggregate, make a loan or advance to or investment in any third party, or guarantee or endorse any Indebtedness, Liability or obligation of any Person;
|
|
•
|
make or rescind any material election relating to material taxes, settle any claim, action, suit, litigation, proceeding,
arbitration, investigation, audit or controversy relating to taxes, file any amended tax return or claim for refund, or make any material change in its method of tax accounting, in each case except as required by applicable law or in
compliance with GAAP;
|
|
•
|
take any action, or knowingly fail to take any action, which action or failure to act would reasonably be expected to prevent
or impede the Merger from qualifying as a “reorganization” within the meaning of Section 368(a) of the Code (and the Treasury Regulations) or, except as contemplated by the Merger Agreement, take any action, which action would
reasonably be expected to prevent or impede any other aspect of the intended tax treatment of the Merger;
|
|
•
|
amend, waive or otherwise change the Trust Agreement in any manner adverse to SEPA;
|
|
•
|
terminate, waive or assign any material right under any SEPA material contract;
|
|
•
|
fail to maintain its books, accounts and records in all material respects in the ordinary course of business consistent with
past practice;
|
|
•
|
establish any subsidiary or enter into any new line of business;
|
|
•
|
fail to use commercially reasonable efforts to keep in force material insurance policies or replacement or revised policies
providing insurance coverage with respect to its assets, operations and activities in such amount and scope of coverage substantially similar to that which is currently in effect;
|
|
•
|
revalue any of its material assets or make any material change in accounting methods, principles or practices, except to the
extent required to comply with GAAP and after consulting SEPA’s outside auditors;
|
|
•
|
waive, release, assign, settle or compromise any claim, action or proceeding (including any suit, action, claim, proceeding
or investigation relating to the Merger Agreement or the transactions contemplated hereby), other than waivers, releases, assignments, settlements or compromises that involve only the payment of monetary damages (and not the imposition
of equitable relief on, or the admission of wrongdoing by, SEPA or its subsidiary) not in excess of $500,000 individually or $1,000,000 in the aggregate;
|
|
•
|
acquire, including by merger, consolidation, acquisition of equity interests or assets, or any other form of business
combination, any corporation, partnership, limited liability company, other business organization or any division thereof, or any material amount of assets outside the ordinary course of business;
|
|
•
|
make capital expenditures in excess of $500,000 individually for any project (or set of related projects) or $1,000,000 in
the aggregate (excluding for the avoidance of doubt, incurring any expenses);
|
|
•
|
adopt a plan of complete or partial liquidation, dissolution, merger, consolidation, restructuring, recapitalization or other
reorganization (other than with respect to the Merger);
|
|
•
|
voluntarily incur any liability or obligation (whether absolute, accrued, contingent or otherwise) in excess of $500,000
individually or $1,000,000 in the aggregate (excluding the incurrence of any expenses) other than pursuant to the terms of a contract in existence as of the date of the Merger Agreement or entered into in the ordinary course of
business;
|
|
•
|
enter into any agreement, understanding or arrangement with respect to the voting of SEPA securities;
|
|
•
|
take any action that would reasonably be expected to significantly delay or impair the obtaining of any consents of any
governmental authority to be obtained in connection with the Merger Agreement; or
|
|
•
|
authorize or agree to do any of the foregoing actions.
|
|
•
|
SEPA will file with the Delaware Secretary of State the Class B Charter Amendment, upon approval of the SEPA Class B
Stockholders, and the Net Tangible Assets Charter Amendment, upon approval by the SEPA Stockholders following approval at the Stockholder Meeting, and SEPA will file with the Delaware Secretary of State the SEPA Closing Date Charter
Amendment immediately after the filing of the Articles of Merger with the Secretary of State of the State of Nevada on the Closing Date;
|
|
•
|
SEPA and certain SANUWAVE Stockholders who will be Affiliates of SEPA immediately after the Closing will enter into a
registration rights agreement in a form to be reasonably acceptable to SEPA and SANUWAVE;
|
|
•
|
SEPA will use its commercially reasonable efforts to enter into voting and non-redemption agreements with such SEPA
Stockholders as mutually agreed by SEPA and SANUWAVE;
|
|
•
|
SEPA shall use its commercially reasonable efforts to obtain the approval of the Warrant Agreement Amendment from the
requisite holders of the SEPA Public Warrants;
|
|
•
|
SANUWAVE will use its commercially reasonable efforts to negotiate with the holders of the SANUWAVE Warrants and the SANUWAVE
Convertible Notes to cause such SANUWAVE Warrants and SANUWAVE Convertible Notes to be amended, exercised, converted or exchanged, as applicable, into shares of SANUWAVE Common Stock; and
|
|
•
|
SANUWAVE will pay off and satisfy in full certain indebtedness at or promptly following the Closing.
|
|
•
|
the Merger Agreement is terminated by SANUWAVE prior to the receipt of the required stockholder approval, if prior to or
substantially concurrent with such termination, SANUWAVE substantially concurrently with such termination enters into a definitive agreement with respect to a Superior Proposal that did not result from a material breach of Section 5.6
of the Merger Agreement, in which case payment shall be made before or concurrently with such termination;
|
|
•
|
the Merger Agreement is terminated by SEPA if SANUWAVE’s board effects a Company Adverse Recommendation Change, SANUWAVE’s
board approves, endorses or recommends a Superior Proposal, a tender offer or exchange offer for any outstanding shares of SANUWAVE capital stock is commenced before obtaining the required stockholder approval and the SANUWAVE board
fails to recommend against acceptance of such tender offer or exchange offer within 10 business days after commencement, or if there shall have been a material breach of Section 5.6 of the Merger Agreement, in which case payment shall
be made within two business days following such termination; or
|
|
•
|
after the date of the Merger Agreement, first, a Takeover Proposal is made, proposed or communicated to the SANUWAVE board or
management, or is publicly made, proposed or communicated or otherwise becomes publicly known; (B) second, the Merger Agreement is terminated by either SANUWAVE or SEPA pursuant to Section 7.1(b) or 7.1(i) of the Merger Agreement, or by
SEPA pursuant to Section 7.1(e) of the Merger Agreement; and (C) third, within twelve (12) months of such termination (1) any transaction included within the definition of a Takeover Proposal with respect to SANUWAVE is consummated or
(2) SANUWAVE enters into a definitive agreement providing for the consummation of any transaction within the definition of Takeover Proposal, in each case whether or not involving the same Takeover Proposal or the person or group making
the Takeover Proposal referred to in clause (A); provided, that for purposes of clause (C), the term “Takeover Proposal” shall have the meaning assigned to such term in Article X of the Merger Agreement, except that all
references to “15%” in such definition shall be deemed references to “50%”, in which case payment shall be made within two business days following such termination.
|
|
•
|
pursuant to the Letter Agreement, 5,031,250 Founder Shares owned by the Sponsor and other insiders became subject to a
lock-up whereby, subject to certain limited exceptions, agreed not to transfer, assign or sell the Founder Shares until the earlier of (A) one year after the completion of SEPA’s initial business combination or (B) subsequent to the
Business Combination, (x) if the last sale price of SEPA’s Common Stock equals or exceeds $12.00 per share (as adjusted for stock splits, stock dividends, reorganizations, recapitalizations and other similar transactions) for any 20
trading days within any 30-trading day period commencing at least 150 days after SEPA’s initial business combination or (y) the date on which SEPA completes a liquidation, merger, capital stock exchange, reorganization or other similar
transaction that results in all of SEPA Stockholders having the right to exchange their shares of Common Stock for cash, securities or other property; provided, however, upon approval of the Letter Agreement Amendment, this period will
be amended to until the earlier of (i) 180 days after the completion of the Business Combination or (ii) subsequent to the Business Combination, the date on which SEPA completes a liquidation, merger, capital stock exchange,
reorganization or other similar transaction that results in all of SEPA Stockholders having the right to exchange their shares of common stock for cash, securities or other property;
|
|
•
|
the Sponsor owns 2,415,375 shares of Class A Common Stock (the “Converted Shares”) (of which 1,746,316 shares will be
forfeited contingent upon and effective immediately prior to the Closing of the Business Combination pursuant to the Forfeiture and Redemption Agreement) and 1,050,000 Founder Shares, and SEPA’s officers and directors own 140,000
Founder Shares. On March 4, 2021, SEPA’s founder acquired 5,031,250 Founder Shares for an aggregate purchase price of $25,000, or approximately $0.005 per share. Also on March 24, 2021, SEPA’s founder assigned 160,000 Founder Shares
(40,000 Founder Shares each) to SEPA’s independent directors at their original purchase price, 35,000 Founder Shares (5,000 Founder Shares each) to SEPA’s advisors and 20,000 Founder Shares (10,000 Founder Shares each) to SEPA’s Chief
Financial Officer and Chief Strategy Officer. SEPA’s directors and officers also have a pecuniary interest in such Founder Shares through their ownership interest in the Sponsor. Upon conversion of the remaining Founders Shares to
Class A Common Stock and following the forfeiture of certain Converted Shares by the Sponsor pursuant to the Forfeiture and Non-Redemption Agreement, the Founder Shares and Converted Shares held by the Sponsor and SEPA’s officers and
directors would have an aggregate market value of approximately $10.5 million, based on the conversion ratio set forth in the Class B Charter Amendment and the last sale price of $10.55 per share of Class A Common Stock on Nasdaq on
December 20, 2023. If SEPA does not consummate the Business Combination or another initial business combination by July 30, 2024, and SEPA is therefore required to be liquidated, these shares would be worthless, as Founder Shares are
not entitled to participate in any redemption or liquidation of the Trust Account, and the Sponsor waived any right to the Trust Account with respect to the Converted Shares. Given the differential in the purchase price that the
Sponsor paid for the Founder Shares as compared to the price of the Units sold in SEPA’s IPO, the Sponsor and SEPA’s directors and officers may earn a positive rate of return on their investment even if SEPA’s Class A Common Stock
trades below the price initially paid for the Units in SEPA’s IPO and the public SEPA Stockholders experience a negative rate of return following the completion of the Business Combination;
|
|
•
|
the Sponsor and SEPA’s directors and officers may be incentivized to complete the Business Combination, or an alternative
initial business combination with a less favorable company or on terms less favorable to
|
|
•
|
simultaneously with the closing of the IPO, the Sponsor purchased 7,850,000 warrants at a price of $1.00 per warrant. Such
private placement warrant is exercisable to purchase one share of Class A Common Stock at a price of $11.50 per share. Such warrants will expire worthless if SEPA fails to complete an initial business combination by July 30, 2024;
|
|
•
|
pursuant to the Sponsor Voting Agreement, the Sponsor has agreed to vote all of the Sponsor’s shares of SEPA in favor of the
Merger Agreement and the Business Combination and to otherwise take (or not take, as applicable) certain other actions in support of the Merger Agreement and the Business Combination and the other matters to be submitted to SEPA
Stockholders for approval in connection with the Business Combination. The Sponsor Voting Agreement also prevents transfers of SEPA shares held by the Sponsor between the date of the Sponsor Voting Agreement and the termination of such
Sponsor Voting Agreement, except for certain permitted transfers where the recipient also agrees to comply with the Sponsor Voting Agreement;
|
|
•
|
on October 11, 2022, SEPA issued the Sponsor Note to the Sponsor, pursuant to which SEPA could borrow up to $1,000,000 at
a 6% interest rate on or before October 11, 2024 to cover, among other things, expenses related to a business combination. As of December 20, 2023, SEPA has borrowed the full $1,000,000 under the Sponsor Note;
|
|
•
|
in order to finance transaction costs in connection with the Business Combination, the Sponsor or an affiliate of the
Sponsor, or certain of SEPA’s officers and directors may, but are not obligated to, loan SEPA additional funds as may be required. If SEPA completes the Business Combination, SEPA would repay the Working Capital Loans out of the
proceeds held in the Trust Account released to SEPA. Otherwise, the Working Capital Loans would be repaid only out of funds held outside the Trust Account. In the event that the Business Combination is not completed, SEPA may use a
portion of the proceeds held outside the Trust Account to repay the Working Capital Loans but no proceeds held in the Trust Account would be used to repay the Working Capital Loans;
|
|
•
|
the Sponsor has agreed that it will be liable to SEPA if and to the extent any claims by a third party (other than SEPA’s
independent auditors) for services rendered or products sold to SEPA, or a prospective target business with which SEPA has discussed entering into a transaction agreement, reduce the amount of funds in the trust account to below (i)
$10.10 per Public Share or (ii) such lesser amount per Public Share held in the trust account as of the date of the liquidation of the trust account, due to reductions in value of the trust assets, in each case net of the amount of
interest which may be withdrawn to pay taxes as well as expenses relating to the administration of the trust account, except as to any claims by a third party who executed a waiver of any and all rights to seek access to the trust
account; and
|
|
•
|
The fact that SEPA has provisions in its Current Charter which waive the corporate opportunities doctrine, which means that
SEPA’s officers and directors have not been obligated to bring all corporate opportunities to SEPA. The potential conflict of interest relating to the waiver of the corporate opportunities doctrine in the Current Charter did not, to
SEPA’s knowledge, impact the search for an acquisition target or prevent SEPA from reviewing any opportunities as a result of such waiver.
|
|
•
|
Identified and evaluated over 100 potential acquisition target companies;
|
|
•
|
Participated in in-person or telephonic discussions with representatives of more than 50 potential acquisition targets; and
|
|
•
|
Signed 19 non-disclosure agreements and provided initial non-binding indications of interest to representatives of four
potential acquisition targets (other than SANUWAVE).
|
|
•
|
Large and Expanding Growth Industry. SANUWAVE is focused on the development of
regenerative medicine products that have the potential to address substantial unmet clinical needs across broad market indications. Based upon the commercial due diligence by SEPA, there are limited therapeutic treatments currently
available that directly and reproducibly activate healing processes in the areas in which SANUWAVE is focusing, particularly for wound care and repair of certain types of musculoskeletal conditions;
|
|
•
|
Platform Supports Further Growth Initiatives. SANUWAVE’s ultrasound and shock wave
technology uses patented systems of noninvasive, high-energy, acoustic shock waves or low intensity and non-contact ultrasound for regenerative medicine and other applications, and its two primary systems, UltraMIST® and PACE®,
are cleared by the FDA using directed energy for wound healing (non-contact ultrasound in the case of UltraMIST and focused shockwave in the case of dermaPACE);
|
|
•
|
Due Diligence. Due diligence examinations of SANUWAVE and discussions with
SANUWAVE’s management team and SEPA’s legal advisors concerning SEPA’s due diligence examination of SANUWAVE;
|
|
•
|
Stockholder Liquidity. The obligation in the Merger Agreement to have Class A Common
Stock issued as Merger Consideration listed on the Nasdaq, a major U.S. stock exchange, which the SEPA Board believes has the potential to offer SEPA Stockholders enhanced liquidity as a result of increased trading volume;
|
|
•
|
Financial Condition. The SEPA Board also considered factors such as SANUWAVE’s
historical financial results, outlook, financial plan, debt structure and unit economics as well as mergers and acquisitions activity for companies in the medical technology industry. In considering these factors, the SEPA Board
reviewed SANUWAVE’s historical growth and its current prospects for growth if SANUWAVE achieves its business plan and various historical and current balance sheet items of SANUWAVE. In reviewing these factors, the SEPA Board noted that
SANUWAVE will be well positioned to gain market share and expand its manufacturing capabilities while continuing to improve its margins;
|
|
•
|
Experienced Management Team. SANUWAVE has a strong management team with significant
operating experience. Morgan Frank, CEO, has over 25 years of experience advising and leading MedTech and other companies, and 30 years in capital markets with numerous successful exits. Andrew Walko, President of SANUWAVE, has deep
roots in the MedTech contract manufacturing industry and has several years of operational experience. He has scaled and launched several products at various companies in the Minnesota region. In addition, he has held leadership roles
for nearly 20 years in the MedTech space as well as the US Army. Tim Hendricks, Executive V.P. of Sales, brings 20 years of industry experience in building sales leadership teams in medical device, biologics, specialty pharmaceuticals
and durable medical equipment. Over a dozen of his years in the industry have been spent in the chronic wound space with start-ups and Fortune 500 companies such as Smith & Nephew (formerly Osiris Therapeutics) and Byram Healthcare.
The senior management of SANUWAVE intends to remain with SANUWAVE in the capacity of officers and/or directors, providing helpful continuity in advancing SANUWAVE’s strategic and growth goals;
|
|
•
|
Lock-Up. Certain SANUWAVE Stockholders (including Mr. Frank) have agreed to be
subject to a 180-day lockup in respect of their SEPA Class A Common Stock subject to certain customary exceptions, which will provide important stability to the leadership and governance of SANUWAVE;
|
|
•
|
Other Alternatives. The SEPA Board believes, after a thorough review of other
business combination opportunities reasonably available to SEPA, that the proposed Business Combination represents the best potential business combination for SEPA and the most attractive opportunity based upon the process utilized to
evaluate and assess other potential acquisition targets;
|
|
•
|
Negotiated Transaction. The financial and other terms of the Merger Agreement and
the fact that such terms and conditions are reasonable and were the product of arm’s length negotiations between SEPA and SANUWAVE; and
|
|
•
|
Fairness Opinion. The SEPA Board considered ValueScope’s fairness opinion, in which
ValueScope opined that the Business Combination is “fair” to the SEPA Stockholders from a financial perspective.
|
|
•
|
Business Plan and Projections May Not Be Achieved. The risk that SANUWAVE may not be
able to execute on the business plan, and realize the financial performance as set forth in the financial projections, in each case, presented to SEPA’s management team and SEPA Board;
|
|
•
|
Redemption Risk. The potential that a significant number of SEPA
Stockholders elect to redeem their shares prior to the consummation of the Business Combination and pursuant to SEPA’s Current Charter, which would potentially make the Business Combination more difficult to complete;
|
|
•
|
Stockholder and Warrantholder Vote. The risk that SEPA Stockholders and SEPA Public
Warrant Holders may fail to provide the respective votes necessary to effect the Business Combination;
|
|
•
|
Closing Conditions. The fact that the completion of the Business Combination is
conditioned on the satisfaction of certain closing conditions that are not within SEPA’s control;
|
|
•
|
Litigation. The possibility of litigation challenging the Business Combination or
that an adverse judgment granting permanent injunctive relief could indefinitely enjoin consummation of the Business Combination;
|
|
•
|
Listing Risks. The challenges associated with preparing SANUWAVE for the applicable
disclosure and listing requirements to which SANUWAVE will be subject as a publicly traded company on Nasdaq;
|
|
•
|
Benefits May Not Be Achieved. The risks that the potential benefits of the Business
Combination may not be fully achieved or may not be achieved within the expected timeframe;
|
|
•
|
Liquidation of SEPA. The risks and costs to SEPA if the Business Combination is not
completed, including the risk of diverting management focus and resources from other business combination opportunities, which could result in SEPA being unable to effect a business combination by July 30, 2024;
|
|
•
|
Growth Initiatives May Not be Achieved. The risk that SANUWAVE’s growth initiatives
may not be fully achieved or may not be achieved within the expected timeframe;
|
|
•
|
Board and Independent Committees. The risk that the post-Closing Combined Company
Board and independent committees do not possess adequate skill set within the context of SANUWAVE operating as a public company;
|
|
•
|
SEPA Stockholders Receiving a Minority Position in SANUWAVE. The risk that SEPA
Stockholders will hold a minority position in SANUWAVE;
|
|
•
|
Fees and Expenses. The fees and expenses associated with completing the Business
Combination; and
|
|
•
|
Other Risks Factors. Various other risk factors associated with the business of
SANUWAVE, as described in the section entitled “Risk Factors” appearing elsewhere in this proxy statement/prospectus.
|
|
•
|
NanoVibronix, Inc.
|
|
•
|
EDAP TMS S.A.
|
|
•
|
Organogenesis Holdings Inc.
|
|
•
|
MiMedx Group, Inc.
|
|
•
|
Integra LifeSciences Holdings Corporation
|
|
•
|
3M Company
|
|
•
|
Coloplast A/S
|
|
•
|
ConvaTec Group PLC
|
|
•
|
Smith & Nephew plc
|
|
•
|
Stryker Corporation
|
|
•
|
IDEXX Laboratories, Inc.
|
|
•
|
Intuitive Surgical, Inc.
|
|
•
|
ResMed Inc.
|
|
•
|
Teleflex Incorporated
|
|
•
|
Zimmer Biomet Holdings, Inc.
|
|
•
|
Becton, Dickinson and Company
|
|
•
|
InspireMD, Inc.
|
|
•
|
LeMaitre Vascular, Inc.
|
|
•
|
NuVasive, Inc.
|
|
•
|
Artivion, Inc.
|
|
•
|
Shockwave Medical, Inc.
|
|
•
|
Terumo Corporation
|
|
•
|
Sysmex Corporation
|
|
•
|
Olympus Corporation
|
|
•
|
Align Technology, Inc.
|
|
•
|
Baxter International Inc.
|
|
•
|
Boston Scientific Corporation
|
|
•
|
Edwards Life Scientific Corporation
|
|
•
|
Hologic, Inc.
|
|
•
|
AtriCure, Inc.
|
|
•
|
Medtronic plc
|
|
•
|
Abbot Laboratories
|
|
•
|
GE HealthCare Technologies Inc.
|
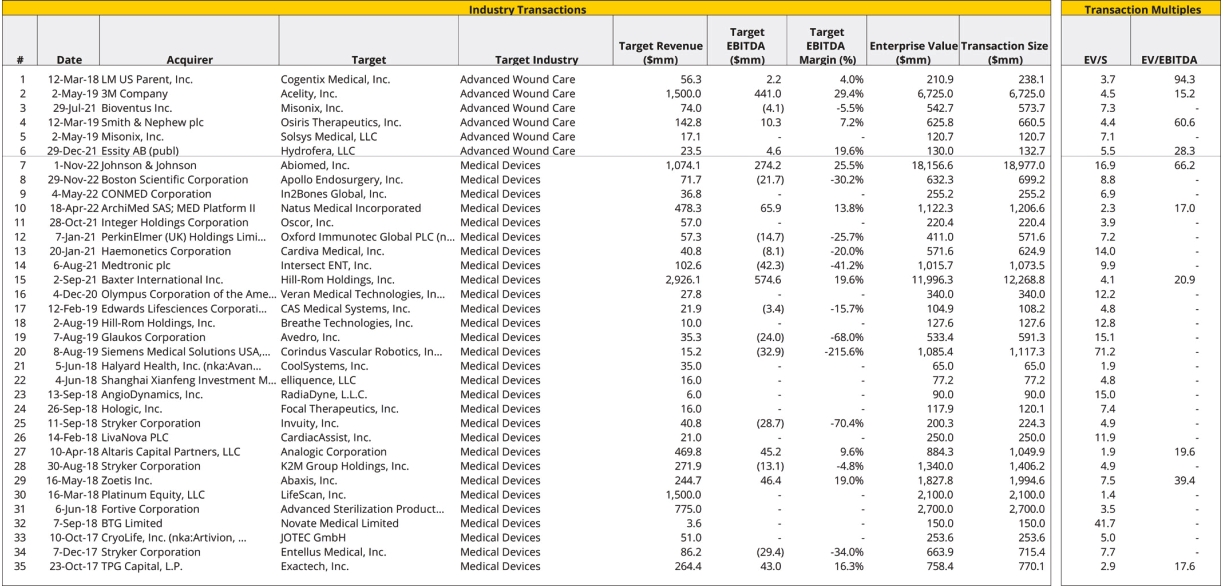
|
•
|
SEPA’s Sponsor or founder (or an officer, director, employee or affiliate thereof);
|
|
•
|
a government or agency or instrumentality thereof;
|
|
•
|
a tax-exempt entity;
|
|
•
|
a tax-qualified retirement plan or pension plan;
|
|
•
|
an S corporation, partnership, or other entity or arrangement treated as a partnership or other flow-through entity for U.S.
federal income tax purposes (and investors therein);
|
|
•
|
a dealer or trader in securities that uses a mark-to-market method of tax accounting with respect to the securities;
|
|
•
|
a regulated investment company;
|
|
•
|
a real estate investment trust;
|
|
•
|
a financial institution;
|
|
•
|
an insurance company;
|
|
•
|
a controlled foreign corporation or passive foreign investment company;
|
|
•
|
an expatriate or former long-term resident of the United States;
|
|
•
|
a person that actually or constructively owns 5% or more of our voting shares or 5% or more of the total value of our shares;
|
|
•
|
a person that acquired our shares pursuant to an exercise of employee share options, in connection with employee share
incentive plans or otherwise as compensation or in connection with services;
|
|
•
|
a person holding the shares as part of a hedge, straddle, constructive sale, or other risk reduction strategy or as part of a
conversion transaction or other integrated investment transaction;
|
|
•
|
a person subject to special tax accounting rules as a result of any item of gross income with respect to Public Shares being
taken into account in an applicable financial statement;
|
|
•
|
a person who elects to apply the provisions of Section 1400Z-2 of the Code to any gain realized;
|
|
•
|
a person holding our securities in connection with a trade or business outside the United States; or
|
|
•
|
a U.S. person whose functional currency is not the U.S. dollar.
|
|
•
|
an individual who is a citizen or resident of the United States;
|
|
•
|
a corporation (or other entity taxable as a corporation) organized in or under the laws of the United States, any state
thereof or the District of Columbia;
|
|
•
|
an estate the income of which is includible in gross income for U.S. federal income tax purposes regardless of its source;
|
|
•
|
a trust (i) the administration of which is subject to the primary supervision of a U.S. court and which has one or more U.S.
persons (within the meaning of the Code) who have the authority to control all substantial decisions of the trust or (ii) that has in effect a valid election under applicable Treasury regulations to be treated as a U.S. person.
|
|
•
|
a non-resident alien individual;
|
|
•
|
a foreign corporation; or
|
|
•
|
an estate or trust that is not a U.S. Holder.
|
|
•
|
the gain is effectively connected with the conduct of a trade or business by you within the United States (and, if required
by an applicable income tax treaty, is attributable to a U.S. permanent establishment or fixed base maintained by you);
|
|
•
|
you are a nonresident alien individual present in the United States for 183 days or more during the taxable year of the
disposition and certain other requirements are met; or
|
|
•
|
the Public Shares constitute a U.S. real property interest (“USRPI”) by reason of SEPA’s status as a “U.S. real property
holding corporation” (a “USRPHC”) for U.S. federal income tax purposes and, as a result, such gain is treated as effectively connected with a trade or business conducted by you in the United States.
|
|
•
|
Former SANUWAVE Securityholders are expected to own approximately 69.0% to 69.6% (assuming no additional Redemptions and
assuming maximum Redemptions, respectively) of the Common Stock outstanding immediately following the Closing (subject to adjustment in accordance with the Merger Agreement);
|
|
•
|
After the Closing, the Combined Company Board shall consist of seven directors: (i) six directors designated by SANUWAVE, and
(ii) one director designated by SEPA;
|
|
•
|
The executive officers of SANUWAVE will become the initial executive officers of the Combined Company;
|
|
•
|
The assets of SANUWAVE will represent a significant majority of the assets of the Combined Company (excluding cash formerly
held in the Trust Account); and
|
|
•
|
After the Closing, the business of the Combined Company will be the continued business of SANUWAVE. The business of the
Company will continue to focus on SANUWAVE’s core offerings.
|
|
Provision
|
| |
Current Charter
|
| |
Proposed Charter
|
|
Name of SEPA
|
| |
SEP ACQUISITION CORP.
See Article I.
|
| |
SANUWAVE Health, Inc.
See Article I.
|
|
|
| |
|
| |
|
|
Provisions Specific to a Blank Check Company and
Variation of Rights of Shares Prior to a Business Combination
|
| |
SEPA’s Current Charter contains language in Article II relating to the
purpose of SEPA as a blank check company.
SEPA’s Current Charter contains provisions in connection with the mechanics
and logistics relating to a business combination, including provisions relating to deposits to and distributions from the Trust Account, the Redemption Rights of holders of Common Stock included as part of the Units sold in the IPO,
share issuances, transactions with affiliates, transactions with other blank check companies, additional redemption rights, the minimum value of the target of the business combination, the appointment and removal of directors, and
approval of the business combination; and such provisions in Article IX cannot be amended prior to the consummation of the initial business combination unless approved by the affirmative vote of the holders of at least 65% of all then
outstanding shares of the Common Stock.
See Article II; Article IX.
|
| |
The Proposed Charter does not include blank check company provisions or
other provisions applicable prior to a business combination, because, upon consummation of the Business Combination, SEPA will cease to be a blank check company.
No corresponding provisions in the
Proposed Charter.
|
|
|
| |
|
| |
|
|
Provision
|
| |
Current Charter
|
| |
Proposed Charter
|
|
Common Stock; Preferred Stock
|
| |
SEPA’s authorized share capital consists of 171,000,000 shares of capital
stock, par value of $0.0001, consisting of (a) 170,000,000 shares of Common Stock, of which (i) 150,000,000 shares are Class A Common Stock and (ii) 20,000,000 shares are Class B Common Stock, and (b) 1,000,000 shares of preferred
stock, par value $0.0001 per share.
See Article IV, Section 4.1.
|
| |
The Proposed Charter will provide for, upon completion of the Business
Combination, the total number of shares of all classes of capital stock that SEPA is authorized to issue is 171,000,000 shares, consisting of (i) 170,000,000 shares of Class A Common Stock, par value $0.0001 per share, and
(ii) 1,000,000 shares of preferred stock, par value $0.0001 per share.
See Article IV, Section 4.1.
|
|
|
| |
|
| |
|
|
Number, Election, and Term of Directors
|
| |
The SEPA Board is divided into three classes, as nearly equal in number as
possible and designated Class I, Class II and Class III. The Board is authorized to assign members of the Board already in office to Class I, Class II or Class III.
See Article V, Section 5.2.
|
| |
The Proposed Charter will have one class of directors who will be elected at
each annual meeting of stockholders for a term of one year. The number of directors will be fixed solely and exclusively by resolution duly adopted from time to time by the Board.
See Article V, Section 5.2.
|
|
|
| |
|
| |
|
|
Removal of Directors
|
| |
Any or all of the directors may be removed from office at any time, but only
for cause and only by the affirmative vote of holders of a majority of the voting power of all then outstanding shares of capital stock of SEPA entitled to vote generally in the election of directors, voting together as a single class.
See Article V, Section 5.4.
|
| |
Any or all of the directors may be removed from office at any time, but only
for cause and only by the affirmative vote of holders of at least two-thirds (2∕3) of the voting power of all then outstanding shares of capital stock of SEPA entitled to vote generally in
the election of directors.
See Article V, Section 5.4.
|
|
|
| |
|
| |
|
|
Amendment of the Bylaws
|
| |
The Bylaws also may be adopted, amended, altered or repealed by the SEPA
Stockholders; provided, however, that in addition to any vote of the holders of any class or series of capital stock of SEPA required by law or by the Current Charter (including any Preferred
Stock Designation), the affirmative vote of the holders of a majority of the voting power of all then outstanding shares of capital stock of SEPA entitled to vote generally in the election of directors, voting together as a single
class, shall be required for the SEPA Stockholders to adopt, amend, alter or repeal the Bylaws; and provided
|
| |
The Bylaws also may be adopted, amended, altered or repealed by the SEPA
Stockholders; provided, however, that in addition to any vote of the holders of any class or series of capital stock of SEPA required by law or by the Proposed Charter (including any Preferred
Stock Designation), the affirmative vote of the holders of at least two-thirds (2∕3) of the voting power of all then outstanding shares of capital stock of SEPA entitled to vote generally
in the election of directors, shall be required for the
|
|
Provision
|
| |
Current Charter
|
| |
Proposed Charter
|
|
|
| |
further, however, that
no Bylaws hereafter adopted by the stockholders of SEPA shall invalidate any prior act of the Board that would have been valid if such Bylaws had not been adopted.
See Article VI.
|
| |
SEPA Stockholders to adopt, amend, alter or repeal the Bylaws; and provided further, however, that no Bylaws hereafter adopted by the SEPA Stockholders shall invalidate any prior act of the Board that would have been valid if such Bylaws had not been adopted.
See Article VI.
|
|
|
| |
|
| |
|
|
Stockholder Action by Written Consent
|
| |
Except as may be otherwise provided for or fixed pursuant to the Current
Charter (including any Preferred Stock Designation) relating to the rights of the holders of any outstanding series of preferred stock, subsequent to the consummation of SEPA’s IPO, any action required or permitted to be taken by the
SEPA Stockholders must be effected by a duly called annual or special meeting of such stockholders and may not be effected by written consent of the SEPA Stockholders other than with respect to its Class B Common Stock with respect to
which action may be taken by written consent.
See Article VII, Section 7.3.
|
| |
The Proposed Charter does not include a provision for stockholder action by
written consent for any class of SEPA Stockholders.
No corresponding provisions in the
Proposed Charter.
|
|
|
| |
|
| |
|
|
Corporate Opportunity Doctrine
|
| |
To the extent allowed by law, the doctrine of “corporate opportunity,” or
any other analogous doctrine, shall not apply with respect to SEPA or any of its officers or directors, and SEPA renounces any expectancy that any of the directors or officers of SEPA will offer any such corporate opportunity of which
he or she may become aware to SEPA, unless such opportunity was expressly offered to such person solely in his or her capacity as a director or officer of SEPA and such opportunity is one SEPA is legally and contractually permitted to
undertake and would otherwise be reasonable for SEPA to pursue.
See Article X.
|
| |
The Proposed Charter does not include a provision waiving the doctrine of
“corporate opportunity.”
No corresponding provisions in the
Proposed Charter.
|
|
•
|
a limited availability of market quotations for its Class A Common Stock;
|
|
•
|
reduced liquidity with respect to its Class A Common Stock;
|
|
•
|
determination that its shares are a “penny stock,” which will require brokers trading in its Class A Common Stock to adhere
to more stringent rules, possibly resulting in a reduced level of trading activity in the secondary trading market for its securities;
|
|
•
|
a limited amount of news and analyst coverage for the Combined Company; and
|
|
•
|
a decreased ability to issue additional securities or obtain additional financing in the future.
|
|
•
|
A “corporate transaction” generally means (i) a sale or other disposition of all or substantially all of the assets of the
Combined Company, or (ii) a merger, consolidation, share exchange or similar transaction involving the Combined Company.
|
|
•
|
A “change in control” generally refers to a corporate transaction (as defined above), the acquisition by a person or group of
beneficial ownership of 30% or more of the voting power of the Company’s stock, or stock equaling 50% or more of the Combined Company’s fair market value, a sale of 40% or more of the assets of the Combined Company, or the Combined
Company’s “continuing directors” ceasing to constitute a majority of its board.
|
|
Plan category
|
| |
Number of securities
to be issued upon
exercise of
outstanding options,
warrants, and rights
|
| |
Weighted
average
exercise price
of outstanding
options,
warrants and
rights
|
| |
Number of
securities
remaining
available for
future issuance
under equity
compensation
plans
|
|
Equity compensation plans approved by security holders
|
| |
—
|
| |
—
|
| |
—
|
|
Equity compensation plans not approved by security holders
|
| |
21,246,085
|
| |
0.28
|
| |
3,240,615
|
|
Total
|
| |
21,246,085
|
| |
0.28
|
| |
3,240,615
|
|
•
|
Within the U.S. and Canada: +1 800-450-7155 (toll-free)
|
|
•
|
Outside of the U.S. and Canada: +1 857-999-9155 (standard rates apply)
|
|
•
|
consider and vote upon an amendment to the Warrant Agreement, dated as of July 21, 2021 (the “Warrant Agreement Amendment”),
a copy of which is attached as Annex B to this proxy statement/prospectus, to provide that, upon Closing of the Business Combination, the then outstanding SEPA Public Warrants will be canceled and exchanged for the right to
receive 450,336 shares of Class A Common Stock (i.e., approximately 0.0499 shares of Class A Common Stock for each public warrant) and the then outstanding SEPA Private Placement Warrants will be
canceled and exchanged for the right to receive 400,000 shares of Class A Common Stock (i.e., approximately 0.0499 shares of Class A Common Stock for each private placement warrant) (the “Warrant
Amendment Proposal”); and
|
|
•
|
consider and vote upon a proposal to approve the adjournment of the Warrant Holders Meeting to a later date or dates, if
necessary, (a) to permit further solicitation and vote of proxies in the event that there are insufficient votes for the approval of the Warrant Amendment Proposal at the Warrant Holders Meeting or (b) if the SEPA Board determines
before the Warrant Holders Meeting that it is not necessary or no longer desirable to proceed with the proposals (the “Warrant Holders Adjournment Proposal” and, together with the Warrant Amendment Proposal, the “Warrant Holder
Proposals”).
|
|
•
|
sending another proxy card with a later date;
|
|
•
|
notifying SEPA’s Secretary in writing before the Warrant Holders Meeting that a SEPA Public Warrant Holder has revoked their
proxy; or
|
|
•
|
attending the Warrant Holders Meeting and voting electronically by entering the control number found on the proxy card they
previously received. Attendance at the Warrant Holders Meeting will not, in and of itself, revoke a proxy.
|
|
•
|
Former SANUWAVE Securityholders are expected to own approximately 69.0% to 69.6% (assuming no additional redemptions and
assuming maximum redemptions, respectively) of the Common Stock outstanding immediately following the Closing (subject to adjustment in accordance with the Merger Agreement);
|
|
•
|
After the Closing, the Combined Company Board shall consist of seven directors: (i) six directors designated by SANUWAVE, and
(ii) one director designated by SEPA;
|
|
•
|
The executive officers of SANUWAVE will become the initial executive officers of the Combined Company;
|
|
•
|
The assets of SANUWAVE will represent a significant majority of the assets of the Company (excluding cash formerly held in
the Trust Account); and
|
|
•
|
After the Closing, the business of the Company will be the continued business of SANUWAVE. The business of the Company will
continue to focus on SANUWAVE’s core offerings.
|
|
a.
|
each SANUWAVE Stockholder shall receive for each share of SANUWAVE Common Stock held (excluding any Treasury Stock), a number
of shares of Class A Common Stock equal to the Conversion Ratio (the aggregate portion of the Merger Consideration amount payable to all SANUWAVE Stockholders in respect of shares of SANUWAVE Common Stock (but excluding Merger
Consideration payable in respect of SANUWAVE Options, Assumed Warrants, and Assumed Convertible Notes) in accordance with the Merger Agreement is also referred to herein as the “Stockholder Merger Consideration”).
|
|
b.
|
the holders of in-the-money SANUWAVE Options shall receive, from the Merger Consideration, such number of SEPA Options as
at the Effective Time, such that each outstanding in-the-money SANUWAVE Option (whether vested or unvested, exercisable or unexercisable) that is outstanding as of immediately prior to the Effective Time shall, without any further
action on the part of the holder thereof, be assumed by the SEPA and automatically converted into the right to receive an option (each, a “SEPA Option”) that corresponds to an in-the-money SANUWAVE Option that was originally granted
pursuant to the Prior Plan will continue to be subject to the same terms and conditions set forth in the Prior Plan and the applicable award agreement as in effect immediately prior to the Effective Time (including, without
limitation, the vesting and acceleration provisions therein), to acquire shares of Class A Common Stock reserved for issuance from the Merger Consideration. Based on the price of SANUWAVE Common Stock on the date of this proxy
statement/prospectus, no in-the-money SANUWAVE Options are outstanding. Out-of-the-money SANUWAVE Options issued pursuant to the Prior Plan that are not exercised prior to the Effective Time will be assumed by SEPA and converted into
SEPA Options exercisable for shares of Class A Common Stock based on the Conversion Ratio; however, such out-of-the-money SANUWAVE Options shall not be reserved for issuance from the Merger Consideration. Out-of-the-money SANUWAVE
Options are currently exercisable for 19,136,150 shares of SANUWAVE Common Stock. Based upon the assumptions set forth in “Unaudited Pro Forma Condensed Combined Financial Information”
in this proxy statement/prospectus, such out-of-the-money SANUWAVE Options would be exercisable for approximately 60,514 shares of Class A Common Stock after the Closing of the Business Combination.
|
|
c.
|
the holders of each in-the-money SANUWAVE Warrant that is outstanding and unexercised, and has not been exchanged for shares
of SANUWAVE Common Stock, as of immediately prior to the Effective Time shall to the extent permitted by the terms of such in-the-money SANUWAVE Warrant, automatically, without any action on the part of the holder thereof and without
the issuance of a new warrant with respect thereto, be assumed by SEPA and converted into a warrant (each such warrant, an “Assumed Warrant”) to purchase that number of shares of Class A Common Stock reserved for issuance from the
Merger Consideration equal to the product of (1) the number of shares of SANUWAVE Stock subject to such in-the-money SANUWAVE Warrant multiplied by (2) the Conversion Ratio. Each Assumed Warrant shall be subject to the same terms and
conditions (including as to vesting and exercisability) as were applicable
|
|
d.
|
the holders of each SANUWAVE Convertible promissory note payable, Convertible promissory note payable, related parties,
Asset-backed secured promissory note, Asset-backed secured promissory note, related parties, the associated Accrued interest, and the associated Accrued interest, related parties, that is outstanding and unexercised, and has not been
exchanged for shares of SANUWAVE Common Stock, and has not been settled, as of immediately prior to the Effective Time (together, the “SANUWAVE Convertible Notes”) shall, to the extent permitted by the terms of such SANUWAVE
Convertible Note, automatically, without any action on the part of the holder thereof and without the issuance of a new convertible note with respect thereto, be assumed by the SEPA and converted into a convertible note (each such
assumed SANUWAVE Convertible Note, an “Assumed Convertible Note”) to purchase that number of shares of Class A Common Stock reserved for issuance from the Merger Consideration equal to the product of (i) the number of shares of
SANUWAVE Stock subject to such SANUWAVE Convertible Note as of immediately prior to the Effective Time multiplied by (ii) the Conversion Ratio. Each Assumed Convertible Note shall be subject to the same terms and conditions as were
applicable under the respective SANUWAVE Convertible Note immediately prior to the Effective Time. The holders of SANUWAVE Convertible Notes that have been exchanged for shares of SANUWAVE Common Stock, prior to the Effective Time,
shall receive the same Merger Consideration as the SANUWAVE Stockholders pursuant to subsection (a) above. Immediately after the Effective Time, there will be no outstanding SANUWAVE Convertible Notes.
|
|
•
|
Assuming No Additional Redemptions: This pro forma presentation assumes that no
additional SEPA Stockholders exercise Redemption Rights with respect to their Class A Common Stock, subsequent to the redemptions that occurred on December 20, 2022.
|
|
•
|
Assuming Maximum Redemptions: This pro forma presentation assumes that 622,747 shares
of Class A Common Stock subject to redemption are redeemed for an aggregate payment of approximately $6,526,686 (based on an estimated per share redemption price of approximately $10.48 that was calculated using the $13,669,258 in the
Trust Account as of September 30, 2023 divided by 1,304,259 shares of Class A Common Stock subject to redemption assuming the pro forma maximum redemption scenario pursuant to the Merger Agreement). It is noted 681,512 shares of Class A
Common Stock are not being redeemed, pursuant to the Voting and Non-Redemption Agreements.
|
|
|
| |
Assuming No
Additional
Redemptions
(Shares)
|
| |
%
|
| |
Assuming
Maximum
Redemptions
(Shares)
|
| |
%
|
|
Sanuwave Stockholders(1)
|
| |
7,793,000
|
| |
69.1%
|
| |
7,793,000
|
| |
69.7%
|
|
SEPA Class A Stockholders (other than the Sponsor)(2)(3)
|
| |
1,304,259
|
| |
11.6%
|
| |
681,512
|
| |
6.1%
|
|
Initial SEPA Class B Stockholders(3)(4)(5)
|
| |
1,232,241
|
| |
10.9%
|
| |
1,232,241
|
| |
11.0%
|
|
SEPA Public Warrant Holders(6)
|
| |
450,336
|
| |
4.0%
|
| |
450,336
|
| |
4.0%
|
|
SEPA Private Placement Warrant Holders(7)
|
| |
400,000
|
| |
3.5%
|
| |
400,000
|
| |
3.6%
|
|
Sponsor Debt Conversion(8)
|
| |
100,000
|
| |
0.9%
|
| |
100,000
|
| |
0.9%
|
|
PIPE Investors(9)
|
| |
—
|
| |
—%
|
| |
518,488
|
| |
4.6%
|
|
Pro forma Combined Company Common
Stock at September 30, 2023
|
| |
11,279,836
|
| |
100.0%
|
| |
11,175,577
|
| |
100.0%
|
|
(1)
|
Includes total Merger Consideration of 7,793,000 shares of Class A Common Stock issuable as follows:
(i) to SANUWAVE Stockholders of 3,244,730 shares of Class A Common Stock, and (ii) upon conversion of the in-the-money warrant liabilities into 3,029,722 shares of Class A Common Stock, and (iii) convertible promissory notes payable and
convertible promissory notes payable, related parties, and the associated accrued interest (together the “convertible promissory notes”) into 473,643 shares of Class A Common Stock, and (iv) Asset-Backed Secured Promissory Note
(“ASBPN”) into 414,359 shares of Class A Common Stock effected for the conversion of the notes into Common Stock, and conversion of the associated warrant liability described in adjustment A to the unaudited pro forma condensed combined
balance sheet, into 630,546 shares of Class A Common Stock respectively. These conversions are illustrated in the below table:
|
|
Security Type
|
| |
September 30, 2023
Sanuwave, pre-merger
|
| |
Merger
Consideration Shares
|
|
Sanuwave Common Stock
|
| |
1,026,078,464
|
| |
3,244,730
|
|
Warrant Liabilities
|
| |
958,086,482
|
| |
3,029,722
|
|
Convertible Promissory Notes and Accrued Interest
|
| |
149,779,681
|
| |
473,643
|
|
Convertible Notes, ABSPN
|
| |
131,032,424
|
| |
414,359
|
|
Warrants underlying the ABSPN
|
| |
199,397,171
|
| |
630,546
|
|
Total
|
| |
2,464,374,222
|
| |
7,793,000
|
|
Security Type
|
| |
SNWV
Common
Stock
|
| |
Theoretical
SEPA
Shares
|
| |
Maximum
Theoretical
Dilutive
Effect,
Total
|
|
(i)
|
| |
36,653,192
|
| |
115,907
|
| |
1.0%
|
|
(ii)
|
| |
19,136,150
|
| |
60,514
|
| |
0.5%
|
|
Total
|
| |
55,789,342
|
| |
176,421
|
| |
1.6%
|
|
(2)
|
Assumes redemption of 622,747 shares of Class A Common Stock because 681,512 shares of Class A Common
Stock are not being redeemed, pursuant to the Voting and Non-Redemption Agreements.
|
|
(3)
|
In order to bring SEPA into compliance with the Nasdaq listing standards, on October 2, 2023, the Sponsor
elected to convert 2,415,375 of its shares of Class B Common Stock into 2,415,375 shares of Class A Common Stock (the “Conversion”). In order to maintain the same economics of the Business Combination for all SEPA Class B Stockholders,
on October 2, 2023, the Sponsor, SEPA and SANUWAVE entered into a Forfeiture and Redemption Agreement, pursuant to which the Sponsor agreed to forfeit 1,746,316 shares of Class A Common Stock contingent upon and effective immediately
prior to the Closing of the Business Combination. The “SEPA Class A Stockholders” row does not include the 2,415,375 shares of Class A Common Stock that the Sponsor currently holds following the Conversion. The “Initial SEPA Class B
Stockholders” row includes the 2,415,375 shares of Class B Common Stock that the Sponsor held prior to the Conversion. Following the Closing, the Sponsor will hold 1,059,909 shares of Class A Common Stock.
|
|
(4)
|
Reflects the conversion of Class B Common Stock to Class A Common Stock, pursuant to the Class B Charter
Amendment to be entered into prior to Closing, amending the Class B Common Stock conversion ratio to 0.277 shares of Class A Common Stock. As of September 30, 2023, there were 4,510,375 shares of Class B Common Stock which will convert
to 1,249,374 shares of Class A Common Stock pursuant to the Class B Charter Amendment.
|
|
(5)
|
Reflects the forfeiture of 61,850 shares of Class B Common Stock to the Company, in exchange for no
consideration, by a SEPA Class B Stockholder pursuant to a forfeiture and redemption agreement entered into on December 18, 2023, in connection with the liquidation of such stockholder. Following the forfeiture, the shares were
cancelled.
|
|
(6)
|
Reflects the conversion of the SEPA Public Warrants into 450,336 shares of Class A Common Stock (i.e.,
approximately 0.0499 shares of Class A Common Stock for each public warrant) pursuant to the Warrant Agreement Amendment. All outstanding SEPA Public Warrants will be converted in connection with the Business Combination, and none
will be outstanding following the Closing.
|
|
(7)
|
Reflects the conversion of the SEPA Private Placement Warrants into 400,000 shares of Class A Common
Stock (i.e., approximately 0.0499 shares of Class A Common Stock for each private placement warrant) pursuant to the Warrant Agreement Amendment. All outstanding SEPA Private Placement Warrants will be converted in connection with the
Business Combination, and none will be outstanding following the Closing.
|
|
(8)
|
Assumes (i) issuance of 100,000 shares of Class A Common Stock in connection with the conversion of
SEPA’s Sponsor promissory note principal and accrued interest into an investment in the PIPE Investment, pursuant to the Sponsor Debt Conversion Agreement. The Sponsor Debt Conversion Agreement is contingent and effective upon
Closing.
|
|
(9)
|
Assumes (i) that under the No Additional Redemptions scenario, no SEPA Class A stockholders elect to
redeem and in this case the existing proceeds in the Trust Account satisfy the $12,000,000 Minimum Cash Condition in the Merger Agreement. Under this assumption the Company is not obligated to raise proceeds through the PIPE
Investment as the Minimum Cash Condition is assumed to be met by the absence of any redemptions.
|
|
|
| |
SEPA
(Historical)
|
| |
SANUWAVE
(Historical)
|
| |
Transaction
Accounting
Adjustments
(Assuming No
Additional
Redemptions)
|
| |
|
| |
Pro Forma
Combined
(Assuming No
Additional
Redemptions)
|
| |
Transaction
Accounting
Adjustments
(Assuming
Maximum
Redemptions)
|
| |
|
| |
Pro Forma
Combined
(Assuming
Maximum
Redemptions)
|
|
ASSETS
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Current assets:
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Cash and cash equivalents
|
| |
$518
|
| |
$1,095
|
| |
$13,669
|
| |
B
|
| |
$9,409
|
| |
$(6,527)
|
| |
J
|
| |
$8,067
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
5,185
|
| |
C
|
| |
|
|
|
| |
|
| |
|
| |
(2,500)
|
| |
D
|
| |
|
| |
|
| |
|
| |
|
|
|
| |
|
| |
|
| |
(3,373)
|
| |
K
|
| |
|
| |
|
| |
|
| |
|
|
Accounts receivable
|
| |
—
|
| |
3,231
|
| |
—
|
| |
|
| |
3,231
|
| |
—
|
| |
|
| |
3,231
|
|
Inventory
|
| |
—
|
| |
1,713
|
| |
—
|
| |
|
| |
1,713
|
| |
—
|
| |
|
| |
1,713
|
|
Prepaid expenses and other current assets
|
| |
61
|
| |
1,355
|
| |
—
|
| |
|
| |
1,416
|
| |
—
|
| |
|
| |
1,416
|
|
Total current assets
|
| |
579
|
| |
7,394
|
| |
7,796
|
| |
|
| |
15,769
|
| |
(1,342)
|
| |
|
| |
14,427
|
|
Cash and marketable securities held in Trust Account
|
| |
13,669
|
| |
—
|
| |
(13,669)
|
| |
B
|
| |
—
|
| |
—
|
| |
|
| |
—
|
|
Property and equipment, net
|
| |
—
|
| |
1,079
|
| |
—
|
| |
|
| |
1,079
|
| |
—
|
| |
|
| |
1,079
|
|
Intangible assets, net
|
| |
—
|
| |
4,609
|
| |
—
|
| |
|
| |
4,609
|
| |
—
|
| |
|
| |
4,609
|
|
Goodwill
|
| |
—
|
| |
7,260
|
| |
—
|
| |
|
| |
7,260
|
| |
—
|
| |
|
| |
7,260
|
|
Total assets
|
| |
$14,248
|
| |
$20,342
|
| |
$(5,873)
|
| |
|
| |
$28,717
|
| |
$(1,342)
|
| |
|
| |
$27,375
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
LIABILITIES, TEMPORARY EQUITY AND
STOCKHOLDERS’ EQUITY (DEFICIT)
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Current liabilities:
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Accounts payable and accrued expenses
|
| |
$832
|
| |
$10,982
|
| |
$(824)
|
| |
D
|
| |
$10,097
|
| |
$—
|
| |
|
| |
$10,097
|
|
|
| |
|
| |
|
| |
(893)
|
| |
K
|
| |
|
| |
|
| |
|
| |
|
|
Senior secured debt, in default
|
| |
—
|
| |
17,645
|
| |
—
|
| |
|
| |
17,645
|
| |
—
|
| |
|
| |
17,645
|
|
Convertible promissory notes payable
|
| |
—
|
| |
7,553
|
| |
(3,553)
|
| |
E
|
| |
—
|
| |
—
|
| |
|
| |
—
|
|
|
| |
|
| |
|
| |
(4,000)
|
| |
K
|
| |
|
| |
|
| |
|
| |
|
|
Convertible promissory notes payable, related parties
|
| |
—
|
| |
2,495
|
| |
(1,122)
|
| |
E
|
| |
—
|
| |
—
|
| |
|
| |
—
|
|
|
| |
|
| |
|
| |
(1,373)
|
| |
K
|
| |
|
| |
|
| |
|
| |
|
|
Income tax payable
|
| |
507
|
| |
—
|
| |
—
|
| |
|
| |
507
|
| |
—
|
| |
|
| |
507
|
|
Franchise tax payable
|
| |
30
|
| |
—
|
| |
—
|
| |
|
| |
30
|
| |
—
|
| |
|
| |
30
|
|
Asset-backed secured promissory notes
|
| |
—
|
| |
6,576
|
| |
(6,576)
|
| |
A
|
| |
—
|
| |
—
|
| |
|
| |
—
|
|
Asset-backed secured promissory notes, related parties
|
| |
—
|
| |
3,094
|
| |
(3,094)
|
| |
A
|
| |
—
|
| |
—
|
| |
|
| |
—
|
|
Promissory note - related party
|
| |
—
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
—
|
| |
|
| |
—
|
|
Convertible promissory note - related party, net of debt
discount
|
| |
729
|
| |
—
|
| |
—
|
| |
|
| |
729
|
| |
(729)
|
| |
C
|
| |
—
|
|
Derivative liability
|
| |
143
|
| |
—
|
| |
—
|
| |
|
| |
143
|
| |
(143)
|
| |
C
|
| |
—
|
|
Accrued interest
|
| |
—
|
| |
5,369
|
| |
(453)
|
| |
E
|
| |
2,938
|
| |
—
|
| |
|
| |
2,938
|
|
|
| |
|
| |
|
| |
(1,978)
|
| |
K
|
| |
|
| |
|
| |
|
| |
|
|
Accrued interest on promissory note - related party
|
| |
46
|
| |
—
|
| |
—
|
| |
|
| |
46
|
| |
(46)
|
| |
C
|
| |
—
|
|
Accrued interest, related parties
|
| |
—
|
| |
729
|
| |
(142)
|
| |
E
|
| |
—
|
| |
—
|
| |
|
| |
—
|
|
|
| |
|
| |
|
| |
(587)
|
| |
K
|
| |
|
| |
|
| |
|
| |
|
|
Factoring liabilities
|
| |
—
|
| |
1,814
|
| |
—
|
| |
|
| |
1,814
|
| |
—
|
| |
|
| |
1,814
|
|
Warrant liability
|
| |
—
|
| |
28,106
|
| |
(27,168)
|
| |
E
|
| |
938
|
| |
—
|
| |
|
| |
938
|
|
Current portion of contract liabilities
|
| |
—
|
| |
68
|
| |
—
|
| |
|
| |
68
|
| |
—
|
| |
|
| |
68
|
|
Other
|
| |
—
|
| |
1,003
|
| |
(540)
|
| |
K
|
| |
463
|
| |
—
|
| |
|
| |
463
|
|
Total current liabilities
|
| |
2,287
|
| |
85,434
|
| |
(52,303)
|
| |
|
| |
35,418
|
| |
(918)
|
| |
|
| |
34,500
|
|
Warrant liabilities - long term
|
| |
1,606
|
| |
—
|
| |
(1,606)
|
| |
F
|
| |
—
|
| |
—
|
| |
|
| |
—
|
|
Lease liabilities
|
| |
—
|
| |
550
|
| |
—
|
| |
|
| |
550
|
| |
—
|
| |
|
| |
550
|
|
|
| |
SEPA
(Historical)
|
| |
SANUWAVE
(Historical)
|
| |
Transaction
Accounting
Adjustments
(Assuming No
Additional
Redemptions)
|
| |
|
| |
Pro Forma
Combined
(Assuming No
Additional
Redemptions)
|
| |
Transaction
Accounting
Adjustments
(Assuming
Maximum
Redemptions)
|
| |
|
| |
Pro Forma
Combined
(Assuming
Maximum
Redemptions)
|
|
Contract liabilities
|
| |
—
|
| |
284
|
| |
—
|
| |
|
| |
284
|
| |
|
| |
|
| |
284
|
|
Deferred tax liability
|
| |
—
|
| |
28
|
| |
—
|
| |
|
| |
28
|
| |
—
|
| |
|
| |
28
|
|
Total liabilities
|
| |
3,893
|
| |
86,296
|
| |
(53,909)
|
| |
|
| |
36,280
|
| |
(918)
|
| |
|
| |
35,362
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Temporary Equity
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Class A common stock, $0.0001 par value, subject to
possible redemption; 1,304,259 shares at redemption value at September 30, 2023
|
| |
13,669
|
| |
—
|
| |
(13,669)
|
| |
G
|
| |
—
|
| |
—
|
| |
|
| |
—
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Stockholders’ Equity (Deficit)
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Preferred stock, $0.0001 par value; 1,000,000 shares
authorized; none issued and outstanding
|
| |
—
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
—
|
| |
|
| |
—
|
|
Preferred Stock, par value $0.001, 5,000,000 shares
authorized; 6,175 shares Series A, 293 shares Series B, 90 shares Series C and 8 shares Series D no shares issued and outstanding at September 30, 2023
|
| |
—
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
—
|
| |
|
| |
—
|
|
Class A Common stock, $0.0001 par value; 150,000,000
shares authorized; no shares issued and outstanding (excluding 1,304,259 shares subject to possible redemption at September 30, 2023)
|
| |
—
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
—
|
| |
|
| |
—
|
|
Class B common stock, $0.0001 par value; 20,000,000
shares authorized; 4,510,375 shares issued and outstanding at September 30, 2023
|
| |
—
|
| |
—
|
| |
—
|
| |
G
|
| |
—
|
| |
—
|
| |
|
| |
—
|
|
Common Stock, par value $0.001, 2,500,000,000 shares
authorized; 1,026,078,464 issued and outstanding at September 30, 2023
|
| |
—
|
| |
1,026
|
| |
—
|
| |
E
|
| |
1
|
| |
—
|
| |
C, J
|
| |
1
|
|
|
| |
|
| |
|
| |
(1,025)
|
| |
H
|
| |
|
| |
|
| |
|
| |
|
|
Additional paid-in capital
|
| |
115
|
| |
171,377
|
| |
(1,150)
|
| |
D
|
| |
226,415
|
| |
(6,527)
|
| |
J
|
| |
226,106
|
|
|
| |
|
| |
|
| |
32,974
|
| |
E
|
| |
|
| |
6,218
|
| |
C
|
| |
|
|
|
| |
|
| |
|
| |
1,606
|
| |
F
|
| |
|
| |
|
| |
|
| |
|
|
|
| |
|
| |
|
| |
13,669
|
| |
G
|
| |
|
| |
|
| |
|
| |
|
|
|
| |
|
| |
|
| |
1,025
|
| |
H
|
| |
|
| |
|
| |
|
| |
|
|
|
| |
|
| |
|
| |
(3,429)
|
| |
I
|
| |
|
| |
|
| |
|
| |
|
|
|
| |
|
| |
|
| |
10,228
|
| |
A
|
| |
|
| |
|
| |
|
| |
|
|
Accumulated deficit
|
| |
(3,429)
|
| |
(238,284)
|
| |
(526)
|
| |
D
|
| |
(233,906)
|
| |
(115)
|
| |
C
|
| |
(234,021)
|
|
|
| |
|
| |
|
| |
(536)
|
| |
E
|
| |
|
| |
|
| |
|
| |
|
|
|
| |
|
| |
|
| |
3,429
|
| |
I
|
| |
|
| |
|
| |
|
| |
|
|
|
| |
|
| |
|
| |
(558)
|
| |
A
|
| |
|
| |
|
| |
|
| |
|
|
|
| |
|
| |
|
| |
5,998
|
| |
K
|
| |
|
| |
|
| |
|
| |
|
|
Accumulated other comprehensive loss
|
| |
—
|
| |
(73)
|
| |
—
|
| |
|
| |
(73)
|
| |
—
|
| |
|
| |
(73)
|
|
Total stockholders’ deficit
|
| |
(3,314)
|
| |
(65,954)
|
| |
61,705
|
| |
|
| |
(7,563)
|
| |
(424)
|
| |
|
| |
(7,987)
|
|
Total liabilities, temporary equity
and stockholders’ equity (deficit)
|
| |
$14,248
|
| |
$20,342
|
| |
$(5,873)
|
| |
|
| |
$28,717
|
| |
$(1,342)
|
| |
|
| |
$27,375
|
|
|
| |
SEPA
(Historical)
|
| |
SANUWAVE
(Historical)
|
| |
Transaction
Accounting
Adjustments
(Assuming No
Additional
Redemptions)
|
| |
|
| |
Pro Forma
Combined
(Assuming No
Additional
Redemptions)
|
| |
Transaction
Accounting
Adjustments
(Assuming
Maximum
Redemptions)
|
| |
Pro Forma
Combined
(Assuming
Maximum
Redemptions)
|
|
Revenue, net
|
| |
$—
|
| |
$13,404
|
| |
$—
|
| |
|
| |
$13,404
|
| |
$—
|
| |
$13,404
|
|
Cost of revenue
|
| |
—
|
| |
3,876
|
| |
—
|
| |
|
| |
3,876
|
| |
—
|
| |
3,876
|
|
Gross profit
|
| |
—
|
| |
9,528
|
| |
—
|
| |
|
| |
9,528
|
| |
—
|
| |
9,528
|
|
Operating expenses
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Formation and operating costs
|
| |
1,610
|
| |
—
|
| |
—
|
| |
|
| |
1,610
|
| |
—
|
| |
1,610
|
|
Franchise tax
|
| |
154
|
| |
—
|
| |
—
|
| |
|
| |
154
|
| |
—
|
| |
154
|
|
General and administrative
|
| |
—
|
| |
6,678
|
| |
—
|
| |
|
| |
6,678
|
| |
—
|
| |
6,678
|
|
Selling and marketing
|
| |
—
|
| |
3,430
|
| |
—
|
| |
|
| |
3,430
|
| |
—
|
| |
3,430
|
|
Research and development
|
| |
—
|
| |
436
|
| |
—
|
| |
|
| |
436
|
| |
—
|
| |
436
|
|
Depreciation and amortization
|
| |
—
|
| |
563
|
| |
—
|
| |
|
| |
563
|
| |
—
|
| |
563
|
|
Total operating expenses
|
| |
1,764
|
| |
11,107
|
| |
—
|
| |
|
| |
12,871
|
| |
—
|
| |
12,871
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Loss from operations
|
| |
(1,764)
|
| |
(1,579)
|
| |
—
|
| |
|
| |
(3,343)
|
| |
—
|
| |
(3,343)
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Other income (expense):
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Interest expense
|
| |
—
|
| |
(10,125)
|
| |
4,620
|
| |
EE
|
| |
(5,505)
|
| |
—
|
| |
(5,505)
|
|
Interest expense on promissory note - related party
|
| |
(55)
|
| |
(2,379)
|
| |
2,434
|
| |
EE
|
| |
—
|
| |
—
|
| |
—
|
|
Change in fair value of derivative liability
|
| |
(16)
|
| |
—
|
| |
16
|
| |
DD
|
| |
—
|
| |
—
|
| |
—
|
|
Earnings on trading securities
|
| |
18
|
| |
—
|
| |
—
|
| |
|
| |
18
|
| |
—
|
| |
18
|
|
Unrealized gain on investments held in Trust Account
|
| |
417
|
| |
—
|
| |
(417)
|
| |
AA
|
| |
—
|
| |
—
|
| |
—
|
|
Gain on waiver of deferred underwriting commissions by
underwriter
|
| |
300
|
| |
—
|
| |
(300)
|
| |
CC
|
| |
—
|
| |
—
|
| |
—
|
|
Unrealized (loss) gain from change in fair value of
derivative liabilities
|
| |
(755)
|
| |
(29,943)
|
| |
29,851
|
| |
DD
|
| |
(847)
|
| |
—
|
| |
(847)
|
|
Other (expense) income
|
| |
—
|
| |
(16)
|
| |
—
|
| |
|
| |
(16)
|
| |
—
|
| |
(16)
|
|
Total other income (expense), net
|
| |
(91)
|
| |
(42,463)
|
| |
36,204
|
| |
|
| |
(6,350)
|
| |
—
|
| |
(6,350)
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Income (loss) before income taxes
|
| |
(1,855)
|
| |
(44,042)
|
| |
36,204
|
| |
|
| |
(9,693)
|
| |
—
|
| |
(9,693)
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Provision for income taxes
|
| |
—
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
—
|
| |
—
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Net income (loss)
|
| |
$(1,855)
|
| |
$(44,042)
|
| |
$36,204
|
| |
|
| |
$(9,693)
|
| |
$—
|
| |
$(9,693)
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Other Comprehensive Loss
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Foreign currency translation adjustments
|
| |
—
|
| |
(6)
|
| |
—
|
| |
|
| |
(6)
|
| |
—
|
| |
(6)
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Total Comprehensive (Loss)/Income
|
| |
$(1,855)
|
| |
$(44,048)
|
| |
$35,533
|
| |
|
| |
$(9,699)
|
| |
$—
|
| |
$(9,699)
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Net income (loss) per share (Note 4):
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Weighted average shares outstanding, Class A common
stock subject to possible redemption
|
| |
1,304,259
|
| |
—
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Basic and diluted net loss per share, Class A common
stock subject to possible redemption
|
| |
$(3.70)
|
| |
—
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
|
| |
SEPA
(Historical)
|
| |
SANUWAVE
(Historical)
|
| |
Transaction
Accounting
Adjustments
(Assuming No
Additional
Redemptions)
|
| |
|
| |
Pro Forma
Combined
(Assuming No
Additional
Redemptions)
|
| |
Transaction
Accounting
Adjustments
(Assuming
Maximum
Redemptions)
|
| |
Pro Forma
Combined
(Assuming
Maximum
Redemptions)
|
|
Weighted average shares outstanding, Class B common
stock
|
| |
4,510,375
|
| |
—
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Basic and diluted net income per share, Class B common
stock
|
| |
$0.66
|
| |
$—
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Weighted average shares outstanding
|
| |
—
|
| |
683,771,214
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Basic and diluted net loss per share
|
| |
—
|
| |
$(0.06)
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Weighted average shares outstanding - basic and diluted
|
| |
|
| |
|
| |
|
| |
|
| |
11,279,836
|
| |
|
| |
11,175,577
|
|
Pro forma net loss per share - basic and diluted
|
| |
|
| |
|
| |
|
| |
|
| |
$(0.86)
|
| |
|
| |
$(0.87)
|
|
|
| |
SEPA
(Historical)
|
| |
SANUWAVE
(Historical)
|
| |
Transaction
Accounting
Adjustments
(Assuming No
Additional
Redemptions)
|
| |
|
| |
Pro Forma
Combined
(Assuming No
Additional
Redemptions)
|
| |
Transaction
Accounting
Adjustments
(Assuming
Maximum
Redemptions)
|
| |
Pro Forma
Combined
(Assuming
Maximum
Redemptions)
|
|
Revenue, net
|
| |
$—
|
| |
$16,742
|
| |
$—
|
| |
|
| |
$16,742
|
| |
$—
|
| |
$16,742
|
|
Cost of revenue
|
| |
—
|
| |
4,331
|
| |
—
|
| |
|
| |
4,331
|
| |
—
|
| |
4,331
|
|
Gross profit
|
| |
—
|
| |
12,411
|
| |
—
|
| |
|
| |
12,411
|
| |
—
|
| |
12,411
|
|
Operating expenses
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Formation and operating costs
|
| |
974
|
| |
—
|
| |
—
|
| |
|
| |
974
|
| |
—
|
| |
974
|
|
Franchise tax
|
| |
201
|
| |
—
|
| |
|
| |
|
| |
201
|
| |
—
|
| |
201
|
|
General and administrative
|
| |
—
|
| |
12,556
|
| |
1,350
|
| |
CC
|
| |
13,906
|
| |
—
|
| |
13,906
|
|
Selling and marketing
|
| |
—
|
| |
7,474
|
| |
—
|
| |
|
| |
7,474
|
| |
—
|
| |
7,474
|
|
Research and development
|
| |
—
|
| |
567
|
| |
—
|
| |
|
| |
567
|
| |
—
|
| |
567
|
|
Depreciation and amortization
|
| |
—
|
| |
766
|
| |
—
|
| |
|
| |
766
|
| |
—
|
| |
766
|
|
Total operating expenses
|
| |
1,175
|
| |
21,363
|
| |
1,350
|
| |
|
| |
23,888
|
| |
—
|
| |
23,888
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Loss from operations
|
| |
(1,175)
|
| |
(8,952)
|
| |
(1,350)
|
| |
|
| |
(11,477)
|
| |
—
|
| |
(11,477)
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Other income (expense):
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Interest expense
|
| |
—
|
| |
(12,771)
|
| |
2,787
|
| |
EE
|
| |
(9,984)
|
| |
—
|
| |
(9,984)
|
|
Interest expense on promissory note - related party
|
| |
(2)
|
| |
(1,361)
|
| |
1,363
|
| |
EE
|
| |
—
|
| |
—
|
| |
—
|
|
Realized gain on investments held in Trust Account
|
| |
2,753
|
| |
—
|
| |
(2,753)
|
| |
AA
|
| |
—
|
| |
—
|
| |
—
|
|
Loss on issuance of debt
|
| |
—
|
| |
(3,434)
|
| |
—
|
| |
|
| |
(3,434)
|
| |
—
|
| |
(3,434)
|
|
Gain/(loss) on extinguishment of debt
|
| |
—
|
| |
(418)
|
| |
—
|
| |
|
| |
(418)
|
| |
—
|
| |
(418)
|
|
Change in fair value of derivative liabilities
|
| |
6,643
|
| |
16,654
|
| |
(7,446)
|
| |
DD
|
| |
15,851
|
| |
—
|
| |
15,851
|
|
Loss on foreign currency exchange
|
| |
—
|
| |
(9)
|
| |
—
|
| |
|
| |
(9)
|
| |
—
|
| |
(9)
|
|
Total other income (expense), net
|
| |
9,394
|
| |
(1,339)
|
| |
(6,049)
|
| |
|
| |
2,006
|
| |
—
|
| |
2,006
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Income (loss) before income taxes
|
| |
8,219
|
| |
(10,291)
|
| |
(7,399)
|
| |
|
| |
(9,471)
|
| |
—
|
| |
(9,471)
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Provision for income taxes
|
| |
(507)
|
| |
(2)
|
| |
—
|
| |
|
| |
(509)
|
| |
—
|
| |
(509)
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Net income (loss)
|
| |
$7,712
|
| |
$(10,293)
|
| |
$(7,399)
|
| |
|
| |
$(9,980)
|
| |
$—
|
| |
$(9,980)
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Other Comprehensive Loss
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Foreign currency translation adjustments
|
| |
—
|
| |
6
|
| |
—
|
| |
|
| |
6
|
| |
—
|
| |
6
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Total Comprehensive (Loss)/Income
|
| |
$7,712
|
| |
$(10,287)
|
| |
$(7,399)
|
| |
|
| |
$(9,974)
|
| |
$—
|
| |
$(9,974)
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Net loss per share (Note 4):
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Weighted average shares outstanding, Class A common
stock subject to possible redemption
|
| |
17,537,090
|
| |
—
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Basic and diluted net income per share, Class A common
stock subject to possible redemption
|
| |
$0.38
|
| |
—
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
|
| |
SEPA
(Historical)
|
| |
SANUWAVE
(Historical)
|
| |
Transaction
Accounting
Adjustments
(Assuming No
Additional
Redemptions)
|
| |
|
| |
Pro Forma
Combined
(Assuming No
Additional
Redemptions)
|
| |
Transaction
Accounting
Adjustments
(Assuming
Maximum
Redemptions)
|
| |
Pro Forma
Combined
(Assuming
Maximum
Redemptions)
|
|
Weighted average shares outstanding, Class B common
stock
|
| |
4,510,375
|
| |
—
|
| |
|
| |
|
| |
—
|
| |
|
| |
—
|
|
Basic and diluted net income per share, Class B common
stock
|
| |
$0.25
|
| |
—
|
| |
|
| |
|
| |
$—
|
| |
|
| |
$—
|
|
Weighted average shares outstanding
|
| |
—
|
| |
549,470,787
|
| |
|
| |
|
| |
11,279,836
|
| |
|
| |
11,175,577
|
|
Basic and diluted net loss per share
|
| |
$—
|
| |
$(0.02)
|
| |
|
| |
|
| |
$(0.88)
|
| |
|
| |
$(0.89)
|
|
A.
|
In evaluating the accounting for the Asset-backed secured promissory notes and Asset-backed secured promissory notes, related
parties (together, the “ABSPN”) and Side Letter, SANUWAVE first considered pursuant to the relevant guidance under ASC 815, Derivatives and Hedging, the unit of account, determining the Side
Letter does not represent a freestanding financial instrument as it is not legally detachable and separately exercisable. The redemption features under the Side Letter are considered embedded derivatives, including a put right for
contingent redemption upon event of default, automatic redemption upon maturity of the ABSPN, and redemption is triggered upon a Fundamental Transaction, as SANUWAVE determined each of these features are not clearly and closely related
to the host instrument (the ABSPN), therefore each of the features met the criteria of an embedded derivative. As of September 30, 2023, all proceeds were received under the ABSPN reflecting total of approximately $4.6 million gross
principal balance, which includes $1.5 million in gross principal balance from related parties, total debt discount of $(0.6) million, and total conversion option of $5.6 million, reflecting total carrying value of $9.6 million on the
condensed consolidated balance sheets of SANUWAVE at September 30, 2023. Immediately prior to Closing, pursuant to the redemption features of the Side Letter, upon the Fundamental Transaction, the outstanding principal balance and
conversion option above of approximately $10.2 million is recorded to additional paid-in capital, and the associated debt discount, of $(0.6) million is recorded to accumulated deficit. The transaction accounting adjustments reflect as
noted above, pursuant to the Side Letter upon a Fundamental Transaction, noting the Merger met the definition of a Fundamental Transaction, the redemption features of the Side Letter immediately prior to Closing are converted into the
underlying warrants representing 199,397,171 shares of SANUWAVE Common Stock and the notes are converted into 131,032,424 shares of SANUWAVE Common Stock. Immediately prior to Closing, these are converted into 630,546 shares of Class A
Common Stock and 414,359 shares of Class A Common Stock, respectively, totaling 1,044,905 shares of Class A Common Stock. The 1,044,905 shares of Class A Common Stock are included within the total Merger Consideration of 7,793,000
shares of Class A Common Stock.
|
|
B.
|
Reflects the reclassification of marketable securities held in the Trust Account to cash and cash equivalents.
|
|
C.
|
Represents under the maximum redemption scenario (i) estimated cash proceeds of $5,184,880 in connection with the assumed
purchase of 518,488 shares of Class A Common Stock at a purchase price of $10.00 per share, pursuant to the PIPE Investment if consummated immediately prior to the Closing on the Closing Date as described in the Merger Agreement and
(ii) conversion of SEPA’s Sponsor promissory note of approximately $1 million including accrued interest into 100,000 shares of Class A Common Stock at a purchase price of $10.00 per share, pursuant to the Sponsor Debt Conversion
Agreement and the Merger Agreement. The Merger Agreement provides that SANUWAVE’s obligation to consummate the Business Combination is conditioned on, among other things, that as of Closing, SEPA have at least $12,000,000 resulting from
(i) proceeds from Class A Common Stock that has not been redeemed in the Redemption,
|
|
D.
|
Represents estimated total transaction costs of $2,500,000 in relation to the Merger. Certain estimated transaction costs of
$1,350,000 comprising $700,000 of legal services, $200,000 of accounting and auditing services, and $450,000 of advisory, printer fees and other services related to the Merger incurred by SEPA of which $526,479 are recorded to SEPA’s
accumulated deficit as of September 30, 2023 and $823,521 is in Accounts Payable and Accrued Expenses of SEPA as of September 30, 2023. SANUWAVE expects to incur transaction costs of approximately $1,150,000, comprising $600,000 of
legal, $100,000 of accounting and auditing services, and $450,000 of advisory, printer fees and other services and relate to the equity issuance which are recognized in additional paid-in capital due to the treatment of the accounting
for a reverse recapitalization. SEPA and SANUWAVE continue to evaluate eligible costs that may need to be assumed pursuant to the Merger.
|
|
E.
|
Reflects (i) the elimination of the SANUWAVE derivative warrant liabilities in the amount of $27.2 million and convertible
promissory notes payable of $3.6 million, convertible promissory notes payable, related parties of $1.1 million, accrued interest of $0.5 million and accrued interest, related parties of $0.1 million, together assumed by SEPA in the
aggregate amount of $5.3 million and converted at Closing into 958,086,482 and 149,779,681 shares of SANUWAVE pre-merger Common Stock respectively, which then converted into 3,029,722 and 473,643 shares respectively, of Class A Common
Stock pursuant to the Merger Agreement. Approximately $2.9 million of Accrued interest remains accrued as of the Closing as this is associated with the $17.6 million of SANUWAVE Senior Secured Debt, in default, which also remains
outstanding at Closing. It is further noted that there is $0.9 million of out-of-the-money warrant liabilities associated with the above convertible notes that are not converting at close and remain outstanding on the unaudited pro
forma condensed combined balance sheet, and underlying these warrant liabilities are 36,653,192 shares of SANUWAVE Common Stock.
|
|
F.
|
Reflects the elimination of the SEPA Public Warrant and Private Warrant liabilities in the amount of $1.61 million, each of
which are not exercisable to purchase shares of Class A Common Stock and instead, effective as of immediately prior to the Effective Time of the Merger, are assumed to be automatically converted solely into the right to receive 450,336
and 400,000 shares of Class A Common Stock, respectively, pursuant to the Merger Agreement.
|
|
G.
|
Reflects (i) the reclassification to permanent equity of approximately $13.7 million of Class A Common Stock subject to
possible redemption pursuant to the Merger Agreement, and (ii) conversion of Class B Common Stock to Class A Common Stock, pursuant to the Class B Charter Amendment to be entered into prior to Closing, amending the Class B Common Stock
conversion ratio to 0.277 shares of Class A Common Stock.
|
|
H.
|
Represents recapitalization of SANUWAVE’s outstanding equity as a result of the reverse recapitalization and the issuance of
SANUWAVE Common Stock to SANUWAVE equity as consideration for the reverse recapitalization.
|
|
I.
|
Reflects the reclassification of SEPA’s historical accumulated deficit into additional paid-in capital as part of the reverse
recapitalization.
|
|
J.
|
Reflects pro forma presentation under the scenario of assuming maximum redemptions, in which 622,747 shares of Class A Common
Stock are redeemed in connection with the Merger, as SEPA stockholders have agreed not to redeem an aggregate of 681,512 outstanding Public Shares of SEPA Class A Common Stock pursuant to the Voting and Non-Redemption Agreements. The
aggregate payments to the redeeming public stockholders will be approximately $6.5 million (assuming a redemption price of $10.48 per share), reducing Class A Common Stock and additional paid-in capital. Pursuant to the Voting and
Non-Redemption Agreements, the SEPA Stockholders party thereto are entitled to receive shares of Class A Common Stock in accordance with the formula set forth in the Voting and Non-Redemption Agreements if the price of the shares of
Class A Common Stock sold in the PIPE Investment is below $10.00 per share. It is anticipated that the price of the shares of Class A Common Stock sold in the PIPE investment will be $10.00 per share. As a result, it is anticipated that
no additional shares of Class A Common Stock will be issued pursuant to the Voting and Non-Redemption Agreements. It is anticipated that 622,747 shares of Class A Common Stock will be redeemed and the Company will issue 518,488 shares
of Class A Common Stock under the PIPE Investment in order to satisfy the Minimum Cash Condition at Closing.
|
|
K.
|
Reflects pro forma impact of the settlement of the SANUWAVE Celularity Note and HealthTronics Note. It is noted the
SANUWAVE Celularity Note and SANUWAVE HealthTronics Note are not converting as of the Closing and will not be allocated any portion of the Merger Consideration. There was $4.0 million of convertible promissory notes payable and $2.0
million of accrued interest, with respect to the Celularity Note. On December 18, 2023, SANUWAVE entered into a Forbearance Letter with Celularity, pursuant to which SANUWAVE agreed to pay Celularity half of the remaining unpaid
principal amount under the Celularity Note, representing $2.0 million, within five business days following the consummation of the Merger Agreement, in exchange for a mutual release of all claims between the parties, other than
SANUWAVE's obligations arising under the Forbearance Letter. Those claims include the release of approximately $2 million of accrued interest, $0.9 million of accrued expenses and $0.5 of other liabilities. The transaction accounting
adjustment for the settlement resulted in a gain of approximately $5.4 million recorded to SANUWAVE’s accumulated deficit. There is $1.4 million of convertible promissory notes payable, related parties and $0.6 million of accrued
interest, related parties, with respect to the HealthTronics Note. On October 31, 2023, SANUWAVE entered into a letter agreement with HealthTronics, pursuant to which SANUWAVE agreed to pay HealthTronics the remaining unpaid principal
amount under the HealthTronics Note by the earlier of the Closing or March 31, 2024 in exchange for HealthTronics’ agreement to release all claims against SANUWAVE related to the HealthTronics Note. The transaction accounting
adjustment for the settlement resulted in a gain of approximately $0.6 million, reflecting a total gain recognized to accumulated deficit of approximately $6.0 million for the settlement of both notes.
|
|
AA.
|
Reflects elimination of investment income on the Trust Account.
|
|
BB.
|
Reflects the estimated transaction costs of SEPA of $1,350,000 as if incurred on January 1, 2022, the date the Merger
occurred for the purposes of the unaudited pro forma condensed combined statement of operations. This is a non-recurring item.
|
|
CC.
|
On June 30, 2023, SEPA’s underwriter agreed to waive its rights to its portion of the fee payable by the Company for
deferred underwriting commissions, with respect to any potential business combination of the Company. This adjustment eliminates the gain recognized upon forgiveness and is a non-recurring item.
|
|
DD.
|
Reflects (i) the elimination of the change in fair value of the SANUWAVE derivative warrant liabilities that will not be
assumed by SEPA and were required to be converted into SEPA warrants at Closing pursuant to the Merger Agreement, and where the remaining warrants will be converted post-close, associated gains and losses are retained on the unaudited
pro forma condensed combined statement of operations, and
|
|
EE.
|
Reflects (i) the elimination of interest expense on SEPA’s Sponsor promissory note, pursuant to the Sponsor Debt
Conversion Agreement effective upon the Closing, and (ii) the elimination of all SANUWAVE’s Convertible Notes’ historical interest expense, converted to SANUWAVE Common Stock prior to Closing, pursuant to the Merger Agreement.
SANUWAVE assumed the conversion of all convertible notes outstanding as of the period presented, except for the HealthTronics Note and the Celularity Note, with aggregate principal balances totaling $5.4 million, which will be settled
as described above. For previous convertible promissory notes which were settled in prior years and were no longer outstanding as of September 30, 2023, the associated interest expense is retained on the unaudited pro forma condensed
combined statement of operations as of December 31, 2022.
|
|
|
| |
For the Nine Months Ended
September 30, 2023
|
| |
For the Year Ended
December 31, 2022
|
||||||
|
|
| |
Assuming No
Additional
Redemptions
|
| |
Assuming
Maximum
Redemptions
|
| |
Assuming No
Additional
Redemptions
|
| |
Assuming
Maximum
Redemptions
|
|
Pro forma net loss (in thousands)
|
| |
$(9,693)
|
| |
$(9,693)
|
| |
$(9,980)
|
| |
$(9,980)
|
|
Weighted average shares outstanding of common stock(1)
|
| |
11,279,836
|
| |
11,175,577
|
| |
11,279,836
|
| |
11,175,577
|
|
Net loss per share - basic and diluted
|
| |
$(0.86)
|
| |
$(0.87)
|
| |
$(0.88)
|
| |
$(0.89)
|
|
Excluded securities:(2)
|
| |
|
| |
|
| |
|
| |
|
|
SANUWAVE Options(3)
|
| |
60,514
|
| |
60,514
|
| |
67,186
|
| |
67,186
|
|
SANUWAVE Common Stock underlying Warrant Liabilities
not converting at Closing(4)
|
| |
115,907
|
| |
115,907
|
| |
115,907
|
| |
115,907
|
|
(1)
|
Note that the maximum redemption scenario results in the reduction of 104,259 shares due to assumed
redemption of 622,747 shares of Class A Common Stock because 681,512 shares of Class A Common Stock are not being redeemed, pursuant to the Voting and Non-Redemption Agreements, is greater than the assumed issuance of 518,488 shares of
Class A Common Stock pursuant to the purchase under the PIPE Investment..
|
|
(2)
|
The potentially dilutive outstanding securities were excluded from the computation of pro forma net loss
per share, basic and diluted, because their effect would have been anti-dilutive, issuance or vesting of such shares is contingent upon the satisfaction of certain conditions which were not satisfied by the end of the periods presented.
|
|
(3)
|
Represents all outstanding SANUWAVE Options which are out-of-the-money options based on the price of
SANUWAVE Common Stock on the date of this proxy statement/prospectus. These out-of-the-money SANUWAVE Options would be exercisable for approximately 60,514 shares of Class A Common Stock after the Closing of the Business Combination.
No in-the-money SANUWAVE Options are outstanding.
|
|
(4)
|
Represents all outstanding SANUWAVE Warrant liabilities which are all out-of-the money based on the
price of SANUWAVE Common Stock on the date of this proxy statement/prospectus. These out-of-the-money SANUWAVE Warrants would be exercisable for approximately 115,907 shares of Class A Common Stock after the Closing of the Business
Combination. Immediately prior to the Effective Time, no in-the-money SANUWAVE Warrants will be outstanding, and as a result, no in-the-money SANUWAVE Warrants will convert into SEPA Assumed Warrants.
|
|
Name
|
| |
Age
|
| |
Position
|
|
M. Blair Garrou
|
| |
51
|
| |
Chairman
|
|
R. Andrew White
|
| |
51
|
| |
President and Chief Executive Officer, and Director
|
|
Winston Gilpin
|
| |
66
|
| |
Chief Financial Officer and Secretary
|
|
Christy Cardenas
|
| |
37
|
| |
Vice President and Chief Strategy Officer
|
|
Jay Gardner
|
| |
67
|
| |
Director
|
|
Mia Mends
|
| |
48
|
| |
Director
|
|
Carolyn Rodz
|
| |
43
|
| |
Director
|
|
•
|
the appointment, compensation, retention, replacement, and oversight of the work of the independent auditors and any other
independent registered public accounting firm engaged by SEPA;
|
|
•
|
pre-approving all audit and permitted non-audit services to be provided by the independent auditors or any other registered
public accounting firm engaged by SEPA, and establishing pre-approval policies and procedures;
|
|
•
|
reviewing and discussing with the independent auditors all relationships the auditors have with SEPA in order to evaluate
their continued independence;
|
|
•
|
setting clear hiring policies for employees or former employees of the independent auditors;
|
|
•
|
setting clear policies for audit partner rotation in compliance with applicable laws and regulations;
|
|
•
|
obtaining and reviewing a report, at least annually, from the independent auditors describing (i) the independent auditor’s
internal quality-control procedures and (ii) any material issues raised by the most recent internal quality-control review, or peer review, of the audit firm, or by any inquiry or investigation by governmental or professional
authorities within the preceding five years respecting one or more independent audits carried out by the firm and any steps taken to deal with such issues;
|
|
•
|
reviewing and approving any related party transaction required to be disclosed pursuant to Item 404 of Regulation S-K
promulgated by the SEC prior to SEPA entering into such transaction; and
|
|
•
|
reviewing with management, the independent auditors, and SEPA’s legal advisors, as appropriate, any legal, regulatory or
compliance matters, including any correspondence with regulators or government agencies and any employee complaints or published reports that raise material issues regarding SEPA’s financial statements or accounting policies and any
significant changes in accounting standards or rules promulgated by the Financial Accounting Standards Board, the SEC or other regulatory authorities.
|
|
•
|
reviewing and approving on an annual basis the corporate goals and objectives relevant to SEPA’s Chief Executive Officer’s
compensation, evaluating SEPA’s Chief Executive Officer’s performance in light of such goals and objectives and determining and approving the remuneration (if any) of SEPA’s Chief Executive Officer based on such evaluation;
|
|
•
|
reviewing and approving on an annual basis the compensation of all of SEPA’s other officers;
|
|
•
|
reviewing on an annual basis SEPA’s executive compensation policies and plans;
|
|
•
|
implementing and administering SEPA’s incentive compensation equity-based remuneration plans;
|
|
•
|
assisting management in complying with SEPA’s proxy statement and annual report disclosure requirements;
|
|
•
|
approving all special perquisites, special cash payments and other special compensation and benefit arrangements for SEPA’s
officers and employees;
|
|
•
|
if required, producing a report on executive compensation to be included in SEPA’s annual proxy statement; and
|
|
•
|
reviewing, evaluating and recommending changes, if appropriate, to the remuneration for directors.
|
|
•
|
The dermaPACE® System was determined to be a Class II device by the FDA under the generic name extracorporeal
shock wave device for treatment of chronic wounds per De Novo filing/order DEN160037. As a result of this order, SANUWAVE was immediately able to market dermaPACE as described in the De Novo request subject to the general control
provisions of the Federal Food, Drug, and Cosmetic Act and the special controls identified in the order. Besides having permission to market in the U.S., dermaPACE is licensed for distribution and sale in the following jurisdictions:
the European Union (CE Mark), Canada, Brazil, Egypt, Singapore and the United Arab Emirates.
|
|
•
|
Profile by SANUWAVE System is marketed only within the U.S. and is listed as a Class I device with the FDA (listing number
D065463). Profile by SANUWAVE System is not approved/cleared/licensed in any other jurisdiction.
|
|
•
|
The orthoPACE® System is not marketed in the U.S. It is marketed in the European Union (CE Mark) and licensed for
sale/distribution in South Korea and Taiwan.
|
|
•
|
Growth and expansion of sales across the United States;
|
|
•
|
Improvement of the functionality and ease-of-use for both medical personnel and patients;
|
|
•
|
Finding antibacterial and anti-biofilm solutions to replace the saline solution used to produce the mist used by this system
to conduct the ultrasound toward its target, which we believe could make the system more effective in treating bacterial infections associated with skin conditions; and
|
|
•
|
Design of new applicators capable of treating large skin conditions, for improved efficiency in such cases.
|
|
•
|
Acute and chronic wound conditions, including diabetic foot ulcers, venous and arterial ulcers, pressure sores, burns and
other skin eruption conditions;
|
|
•
|
Orthopedic applications, such as eliminating chronic pain in joints from trauma, arthritis or tendons/ligaments inflammation
or tendinopathies, speeding the healing of fractures (including nonunion or delayed-union conditions), improving bone density in osteoporosis, fusing bones in the extremities and spine, and other potential sports injury applications;
|
|
•
|
Plastic/cosmetic applications such as cellulite smoothing, graft and transplant acceptance, skin tightening, scarring and
other potential aesthetic uses; and
|
|
•
|
Cardiovascular applications for removing plaque due to atherosclerosis, eliminating occlusions and blood clots, and improving
heart muscle and cardiac valves performance.
|
|
Treatment Indication
|
| |
LEVEL OF EVIDENCE
|
| |
Publication
|
| |
WOUND
POPULATION
|
| |
MEASUREMENT
|
| |
PACE RESULT
|
| |
STANDARD OF CARE RESULT
|
| |
TREATMENT DURATION
|
| |
Reported Serious Adverse Events
|
| |
STATISTICS and Conclusions
|
| |
Author
|
| |||||||||||||||||||||||||
| |
Journal
|
| |||||||||||||||||||||||||
|
Diabetic Foot Ulcer
Trials from 2007 - 2012
|
| |
Ib
|
| |
Diabetic Foot Ulcer Treatment With Focused Shockwave Treatment: Two
Multicentre, Prospective, Controlled, Double-Blinded, Randomised, Phase III Clinical Trials
Robert Snyder,1 DPM, MSc, CWS, FFPM RCPS (Glasg); Robert Galiano,2 MD; Perry
Mayer,3 MB, BCh, BAO BSci (hon); Lee C. Rogers,4 DPM; Oscar Alvarez,5 PhD, CCT, FAPWCA; The Sanuwave Trial Investigators
Journal of Wound Care, 2018
December; Vol 27, NO 12
|
| |
N=336
Diabetic foot ulcers
(which had not reduced in volume by >50% over two weeks of receiving
standard care alone)
|
| |
Wound Closure (100% wound area reduction defined as epithelialization without
drainage or dressing requirements)
|
| |
22.7%
35.5%
37.8%
|
| |
18.3%
24.4%
26.2%
|
| |
12 weeks
20 weeks
24 weeks
|
| |
Throughout the 24-week follow-up, the adverse event rates between the
dermaPACE® and control subjects were similar with no statistical significance between the two cohorts in treatment-emergent adverse events (55.8% vs. 51.2%), device-related treatment emergent adverse events (5.2% vs. 2.4%), or all
adverse events (73.2% vs. 68.9%).
The percentage of patients that had to undergo full or partial amputation of
the target ulcer was lower in dermaPACE®, when compared to the control (2.3% vs. 6.63%, p-value= 0.066, respectively). The recurrence rate for dermaPACE® patients was 7.7% compared to 11.6% in the control group
(p-value=0.490).
|
| |
PACE® therapy trended toward significance at 12 weeks
p=0.027
p=0.023
Shockwave treatment demonstrated statistically significant results in wound
area reduction, wound closure, and demonstrated suggestive significance in reduction of wound recurrence
|
|
Treatment Indication
|
| |
LEVEL OF EVIDENCE
|
| |
Publication
|
| |
WOUND
POPULATION
|
| |
MEASUREMENT
|
| |
PACE RESULT
|
| |
STANDARD OF CARE RESULT
|
| |
TREATMENT DURATION
|
| |
Reported Serious Adverse Events
|
| |
STATISTICS and Conclusions
|
| |
Author
|
| |||||||||||||||||||||||||
| |
Journal
|
| |||||||||||||||||||||||||
|
Diabetic Foot Ulcer
Trials from 2007 - 2012
|
| |
Ib
|
| |
Focused Shockwave Therapy in Diabetic Foot Ulcers: Secondary Endpoints of Two
Multicentre Randomised, Controlled Trials
Robert Snyder,1 DPM, MSc, CWS, FFPM RCPS (Glasg); Robert Galiano,2 MD; Perry
Mayer,3 MB, BCh, BAO BSci (hon); Lee C. Rogers,4 DPM; Oscar Alvarez,5 PhD, CCT, FAPWCA; The Sanuwave Trial Investigators
Journal of Wound Care, 2019 June;
Vol 28, NO 6
|
| |
N=336
Diabetic foot ulcers
(which had not reduced in volume by >50% over two weeks of receiving
standard care alone)
|
| |
Wound Area Reduction
Perimeter Reduction
Time to Wound Closure (25% of subjects)
|
| |
48.6%
46.4%
84 days
|
| |
10.7%
25.0%
112 days
|
| |
Efficacy-related secondary endpoints were measured at 12, 20, and 24 weeks
|
| |
Same as prior
|
| |
p=0.015
p=0.022
p=0.0346
Shockwave treatment demonstrated statistically significant results in wound
area reduction, wound closure, and demonstrated suggestive significance in reduction of wound recurrence.
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Plantar Fasciitis
Trials from 1996 - 1998
|
| |
Ib
|
| |
High-Energy shock wave treatment for chronic plantar
fasciitis.
Ogden J, Alvarez R, et
al. Electrohydraulic
Journal of Bone and
Joint Surgery, Vol 86-A, No 10, October 2004, 2216-2228
|
| |
N=235
Multi-center, randomized, placebo-controlled clinical trial to determine the
safety and efficacy of extracorporeal shock wave (ESW) treatment for chronic heel pain syndrome
|
| |
Primary success criteria was minimum 50% improvement and VAS score of 4.0 or
less.
|
| |
74 subjects (62.18%) met the success criteria; subjects improved from a mean
baseline VAS score of 7.68 to 3.13 at week 12.
For the placebo patients who failed the criteria and elected for active
treatment, 52 of the 81 54.1% of the re-treated patients met the success criteria following their primary active treatment.
|
| |
69.83% of the placebo treatment subjects failed to meet success criteria by
week twelve and were therefore eligible for re-treatment with an active ESW procedure.
|
| |
Efficacy-related secondary endpoints were measured at 12, 24 and 52 weeks
|
| |
There were no complications in the Phase 1 patients. The most frequent phase
2 related complications in all groups were pain after treatment and mil neurologic symptoms (numbness or dysesthesia principally related to the anle-block anesthesia. All patients had complete resolution of the post-treatment
neurologic symptoms by the 3 month evaluation, and no patient had neurologic complaints at 12 months.
|
| |
(p-0.008).
The directed application electrohydraulic-generated shock waves to the
insertion of the plantar fascia onto the calcaneus is a safe and effective nonsurgical method for treating chronic, recalcitrant heel pain syndrome that has been present for at least 6 months and has been refractory to other commonly
used nonoperative therapies.
|
|
Treatment Indication
|
| |
LEVEL OF EVIDENCE
|
| |
Publication
|
| |
WOUND
POPULATION
|
| |
MEASUREMENT
|
| |
PACE RESULT
|
| |
STANDARD OF CARE RESULT
|
| |
TREATMENT DURATION
|
| |
Reported Serious Adverse Events
|
| |
STATISTICS and Conclusions
|
| |
Author
|
| |||||||||||||||||||||||||
| |
Journal
|
| |||||||||||||||||||||||||
|
Lateral Epicondylitis
Trial from 2000 - 2002
|
| |
IIb
|
| |
Premarket Approval (PMA) Application; Extracorporeal Shock wave treatment of
chronic lateral epicondylitis, PMA P090086S3, Approved 2003. On file at SANUWAVE.
|
| |
N=183 randomized; N=42 non-randomized.
Multi-center, randomized, placebo-controlled clinical trial to determine the
safety and efficacy of ESW treatment for chronic lateral epicondylitis.
|
| |
Primary success criteria was minimum 50% improvement and VAS score of 4.0 or
less.
|
| |
82 subjects were randomized to active ESW treatment. These subjects improved
from a mean baseline VAS score of 7.73 to 3.64 at week 12. 43 subjects (52.4%) met the success criteria.
|
| |
83 subjects were randomized to placebo treatment. These subjects improved
from a mean baseline score VAS of 7.81 to 5.17 at 12 weeks. 26 subjects (31.3%) met the success criteria
|
| |
12 weeks
|
| |
There were no unanticipated adverse effects or any serious adverse effects
(SAEs). Most commonly reported device related complications were localized swelling, bruising or petechiae at the treatment site or reaction to anesthetic. Other complications including numbness or tingling and post-treatment pain.
|
| |
results demonstrate that low-dose shock wave therapy without anesthetic is a
safe and effective treatment for chronic lateral epicondylitis.
|
|
Treatment Indication
|
| |
LEVEL OF EVIDENCE
|
| |
Publication
|
| |
WOUND
POPULATION
|
| |
MEASUREMENT
|
| |
PACE RESULT
|
| |
STANDARD OF CARE RESULT
|
| |
TREATMENT DURATION
|
| |
Reported Serious Adverse Events
|
| |
STATISTICS and Conclusions
|
| |
Author
|
| |||||||||||||||||||||||||
| |
Journal
|
| |||||||||||||||||||||||||
|
Diabetic foot ulcer
2010
|
| |
IIb
|
| |
Treatment of diabetic foot ulcers: a comparative study of
extracorporeal shockwave therapy and hyperbaric oxygen therapy.
Wang CJ, Wu RW, Yang
YJ.
Diabetes
Res Clin Pract. 2011 May;92(2):187-93. doi: 10.1016/j.diabres.2011.01.019. Epub 2011 Apr 6.
|
| |
ESWT Group N=39 patients/44 feet
HBOT Group N=38 patients/40 feet
Diabetic foot ulcers
|
| |
Completely Healed
>/50% Improved Ulcers
|
| |
57%
32%
|
| |
25%
15%
|
| |
Three weeks (two times/week; six total treatments)
Three weeks (daily; 20 total treatments)
|
| |
There were no device related adverse events.
|
| |
p=0.0003
p<.0.001
The blood flow perfusion rates were comparable between the two groups before
treatment (P=0.245); however, significant differences were noted after treatment favoring the ESWT group (P=0.002). Histopathological examination revealed considerable increases in cell proliferation and decreases in cell apoptosis in
the ESWT group as compared to the HBOT group.
|
|
Treatment Indication
|
| |
LEVEL OF EVIDENCE
|
| |
Publication
|
| |
WOUND
POPULATION
|
| |
MEASUREMENT
|
| |
PACE RESULT
|
| |
STANDARD OF CARE RESULT
|
| |
TREATMENT DURATION
|
| |
Reported Serious Adverse Events
|
| |
STATISTICS and Conclusions
|
| |
Author
|
| |||||||||||||||||||||||||
| |
Journal
|
| |||||||||||||||||||||||||
|
Diabetic foot ulcer
2010
|
| |
IIb
|
| |
Molecular changes in diabetic foot ulcers. Wang CJ, M.D., Ko JY, M.D., Kuo YR, M.D., Yang YJ, M.D., Diabetes Res Clin Pract. 2011 Oct;94(1):105-10.doi: 0.1016/j.diabres.2011.06.016. Epub 2011 Jul
13.
(Follow on analysis of:
Treatment of diabetic foot ulcers: a comparative study of extracorporeal shockwave therapy and hyperbaric oxygen therapy.)
|
| |
ESWT Group N=39 patients/44 feet
HBOT Group N=38 patients/40 feet
Diabetic foot ulcers
|
| |
Gene expression after treatment with ESWT (dermaPACE) or HBOT
vWF
VEGF
eNOS
PCNA
EGF
TUNEL (cell apoptosis)
|
| |
Pretreatment mean value
ESWT = 13.5
HBOT = 19.4
ESWT = 39.3
HBOT = 44.1
ESWT = 22.4
HBOT = 27.5
ESWT = 27.8
HBOT = 28.9
ESWT = 29.1
HBOT = 32.5
ESWT = 67.6
HBOT = 63.0
|
| |
Post-treatment mean value
ESWT = 49.7
HBOT = 21.17
ESWT = 65.7
HBOT = 45.8
ESWT = 54.64
HBOT = 37.1
ESWT = 60.5
HBOT = 36.7
ESWT = 68.9
HBOT = 40.8
ESWT = 31.9
HBOT = 57.6
|
| |
ESWT: three weeks (two times/week; six total treatments)
HBOT: three weeks (daily; 20 total treatments)
|
| |
There were no device related adverse events.
|
| |
ESWT: p=0.038
HBOT: p=0.893
ESWT: p<0.001
HBOT: p=0.090
ESWT: p=0.001
HBOT: p=0.400
ESWT: p=0.001
HBOT: p=0.061
ESWT: p=0.002
HBOT: p=0.051
ESWT: p=0.005
HBOT: p=0.161
PACE treatment increased perfusion response; and an immediate inflammatory
reaction due to the high-pressure waves that put mechanical stress on the microcirculatory system. PACE Technology has been shown to cause mechanical forces within tissues that initiate a biological response at a cellular level
producing angiogenic growth factors, including eNOS, VEGF, and PCNA.
|
|
Treatment Indication
|
| |
LEVEL OF EVIDENCE
|
| |
Publication
|
| |
WOUND
POPULATION
|
| |
MEASUREMENT
|
| |
PACE RESULT
|
| |
STANDARD OF CARE RESULT
|
| |
TREATMENT DURATION
|
| |
Reported Serious Adverse Events
|
| |
STATISTICS and Conclusions
|
| |
Author
|
| |||||||||||||||||||||||||
| |
Journal
|
| |||||||||||||||||||||||||
|
Venous Leg Ulcer
Pressure Ulcers
Diabetic Foot Ulcers
2007
|
| |
|
| |
Saggini et al. Extracorporeal Shock
Wave Treatment for Management of Chronic Ulcers in the Lower Extremities. Ultrasound in Medicine and Biology. 2008; 34: 1-11.
|
| |
N=40
Active: n=32
Control: n=8
Areas treated: ulcers in malleolar region; anterior aspect of the medial
third of leg; posterior aspect of the distal third of leg; lateral plantar side of foot; calcaneal region
|
| |
Determination of the effects of shockwave on the treated wound.
|
| |
32 wounds treated with ESW:
16 (50%) healed completely
All non-healed wounds showed decrease of exudates, increased percentage of
granulation tissue compared with fibrin/necrotic tissue and decrease in wound size statistically significant.
|
| |
1/8 (12.5%) wounds healed during the trial period
|
| |
12 week treatment period with 12 week followup
|
| |
No adverse effects such as bleeding, petechiae, hematoma or seroma formation
were reported.
|
| |
p<0.01
In patients who have had failure of conventional treatment, ESWT seems to be
a valid support to accelerate the healing process of these chronic wounds. The go on to state that ESWT used without local anesthesia may be a safe, feasible and cost-effective treatment in the management of chronic wounds in the
lower extremities.
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Ischemia
Oxygenation
2006
|
| |
IIb
|
| |
Extracorporeal Shockwave Enhanced Extended Skin Flap
Tissue Survival via Increase of Topical Blood Perfusion and Associated with Suppression of Tissue Pro-inflammation. Yur-Ren Kuo, M.D., Ph.D., F.A.C.S.,Wen-Sheng Wu, D.V.M.,
M.S., Yu-Ling Hsieh, B.S.,Feng-Sheng Wang, Ph.D., Chun-Ting Wang, B.S., Yuan-Cheng Chiang, M.D., and Ching-Jen Wang, M.D. J
Surg Res. 2007 Dec;143(2):385-92. Epub 2007 Aug 27.
|
| |
N=36 male Sprague Dawley rats (divided into three groups)
Group II
Group III
|
| |
|
| |
30 ESW
10 Control
32 wounds treated with ESW: 16 (50%) healed completely
All non-healed wounds showed decrease of exudates, increased percentage of
granulation tissue compared with fibrin/necrotic tissue and decrease in wound size statistically significant.
Necrotic area of the flaps in Group II was significantly reduced compared
with that in the control group (13 +/- 2.6% versus 42 +/- 5.7%, P < 0.01).
Flap tissue blood perfusion was significantly increased postoperatively.
There was small and insignificant reduction in the necrotic area in Group III
compared with the controls.
|
| |
Group I – control/no treatment
|
| |
500 impulses postoperatively
500 impulses postoperatively and one day post-op
|
| |
N/A
|
| |
Histological staining indicated that ESW treatment substantially
increased vascular endothelial growth factor and proliferating cell nuclear
antigen expressions, and reduced leukocyte infiltration and suppression of tumor necrosis factor alpha expression in flap tissue ischemic zones in Group II compared with that in controls.
|
|
Treatment Indication
|
| |
LEVEL OF EVIDENCE
|
| |
Publication
|
| |
WOUND
POPULATION
|
| |
MEASUREMENT
|
| |
PACE RESULT
|
| |
STANDARD OF CARE RESULT
|
| |
TREATMENT DURATION
|
| |
Reported Serious Adverse Events
|
| |
STATISTICS and Conclusions
|
| |
Author
|
| |||||||||||||||||||||||||
| |
Journal
|
| |||||||||||||||||||||||||
|
Ischemia
2006
|
| |
IIb
|
| |
Shockwave treatment reduces necrotic flap zones and
induces VEGF expression in animal epigastric skin flap model. Meirer R. Brunner, Deibl (2007): J Reconstructive Microsurgery; 23(4):231-6.
|
| |
N=40 Sprague-Dawley rats (randomly assigned into two groups)
|
| |
Areas of necrotic zones
VEG-F-positive cells
FGF-2-positive staining cells
|
| |
Areas of necrotic zones were significantly smaller (median 4.2%, range 1.9 –
17.3%)
Median 84.5%, range 57.4 – 94.5%
Not meaningful (Median 90.5%, range 67.1 – 96.0%
|
| |
(Median 18.3%, range 8.8 – 31.1%)
Median 46.7%, range 29.1 – 93.1%
Median 89.0%, range 74.6 – 94.0%
|
| |
500 impulses ESWT immediately post-op
|
| |
N/A
|
| |
p<0.01
p<0.1
p>0.1
ESWT’s potential to reduce necrosis in skin flaps and modulate the in-situ
expression of the angiogenic cytokines basic fibroblast factor (FGF-2) and VEGF in tissue abutting the necrotic area.
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Ischemia
2006
|
| |
IIb
|
| |
Pulsed Acoustic Cellular Treatment Induces Expression of
Proangiogenic Factors and Chemokines in Muscle Flaps. Lukasz Krokowicz, MD, Joanna Cwykiel, MSc, Aleksandra Klimczak, PhD, Mariusz Mielniczuk, MD, and Maria
Siemionow, MD, PhD, DSc. Journal of Trauma, Injury, Infection, and Critical Care, DOI: 10.1097/TA.0b013e3181ddd067
|
| |
N=42 Lewis rats
Group 1: control (N=10); acute PACE treatment 15 minutes before surgery
Group 2: 200 impulses (N=8)
Group 3: 500 impulses (N=8); and PACE treatment 24 hours before surgery Group
4: 200 impulses (N=8)
Group 5: 500 impulses (N=8)
|
| |
Red Blood Cell Velocity
Expression levels of:
iNOS (inductible nitric oxide synthase);
eNOS (endothelial nitric oxide synthase);
VEGF (vascular endothelial growth factor);
Von vWF (Wilenbrand factor);
CD31
Chemokines CXCL5, CCL2, CCR2
|
| |
PACE treatment resulted in an increase of arteriolar diameters in acute
groups 2 and 3 (p < 0.05). In group 5, vessel densities assessed by CD31, VEGF, and vWF expression increased significantly 24 hours after PACE treatment compared with control (p<0.05). PACE application downregulated
proinflammatory iNOS gene expression and upregulated proangiogenic genes expression of eNOS, VEGF, CXCL5, and CCL2.
|
| |
N/A
|
| |
2 – Acute PACE – 200 impulses 15 min pre-op
3 – Acute PACE – 500 impulses 15 min pre-op
4 – PACE – 200 impulses 24 hours pre-op
5 – PACE – 500 impulses 24 hours pre-op
|
| |
N/A
|
| |
RBC velocity/arteriolar diameter increase p=0.05
Vessel densities (CD31, VEGF, vWF) p<0.05
Downregulation of iNOS, upregulation of eNOS, VEGF, CXCL5, and CCL2
p=0.05
|
|
Treatment Indication
|
| |
LEVEL OF EVIDENCE
|
| |
Publication
|
| |
WOUND
POPULATION
|
| |
MEASUREMENT
|
| |
PACE RESULT
|
| |
STANDARD OF CARE RESULT
|
| |
TREATMENT DURATION
|
| |
Reported Serious Adverse Events
|
| |
STATISTICS and Conclusions
|
| |
Author
|
| |||||||||||||||||||||||||
| |
Journal
|
| |||||||||||||||||||||||||
|
Ischemia
2006
|
| |
IIb
|
| |
Pulsed acoustic cellular expression as a protective
therapy against I/R injury in a cremaster muscle flap model, Lukasz Krokowicz, Aleksandra Klimczak, Joanna Cwykiel, Mariusz Mielniczuk, Christopher Grykien ,
Maria Siemionow. Microvascular Research 83 (2012) 213–222
|
| |
N=34 rat cremaster muscle flaps
-
|
| |
Pre-ischemic PACE conditioning (group 3) resulted in increased RBC velocity
of:
Second order arterioles (A-2)
Third order arterioles (A-3)
Venule (V-1)
Post-ischemic PACE conditioning (group 4) revealed:
Increase in RBC velocity in second order arterioles (A-2)
Third order arterioles (A-3)
First order arterioles diameter (A-1) (p<0.05) compared to ischemic
controls
3) Number of functional capillaries
Upregulation of proangiogenic factors vWF and VEGF protein expression
|
| |
40%
15%
24%
65%
31%
33% increase
21% increase
Immunostaining assays showed that PACE post-conditioning upregulated
proangiogenic factors vWF and VEGF protein expression. This correlated with increased gene expression of VEGF (up to 180%). In contrast, gene expression of pro-inflammatory factors (iNOS, CCL2, CXCL5) decreased compared to ischemic
controls. Pre-ischemic PACE conditioning decreased gene expression of proinflammatory chemokines (CCL2 and CXCL5), compared to ischemic controls without conditioning.
|
| |
Comparing to ischemic group without conditioning.
Comparing to ischemia without conditioning (group 2).
Compared to ischemia without conditioning (group 2).
|
| |
Group 1: non-ischemic controls (N=10), Group 2: 5h ischemia without
conditioning (N=8), Group 3: pre- ischemic (5h) PACE conditioning (N=8), Group 4: post-ischemic (5h) PACE conditioning (N=8)
|
| |
N/A
|
| |
(p<0.05)
(p<0.05)
(p<0.05)
(p<0.05)
(P<0.05)
(P<0.05)
(P<0.05)
|
|
Treatment Indication
|
| |
LEVEL OF EVIDENCE
|
| |
Publication
|
| |
WOUND
POPULATION
|
| |
MEASUREMENT
|
| |
PACE RESULT
|
| |
STANDARD OF CARE RESULT
|
| |
TREATMENT DURATION
|
| |
Reported Serious Adverse Events
|
| |
STATISTICS and Conclusions
|
| |
Author
|
| |||||||||||||||||||||||||
| |
Journal
|
| |||||||||||||||||||||||||
|
Ischemia
2006
|
| |
IIb
|
| |
Pre- and post-ischemic pulsed acoustic cellular expression
conditioning modulates expression of inflammation factors in cremaster ischemia/reperfusion injury model. Cwykiel JM, Klimczak A, Krokowicz L, Siemionow M. Microsurgery. 2013 Feb;33(2):134-40. doi: 10.1002/micr.22048. Epub 2012 Nov 14.
|
| |
N=17 cremaster muscle flaps
|
| |
Expression of pro-inflammatory cytokines (TNFα, IL-6, IL-1α, IL-1β, GM-CSF)
and chemokines (CCL3, CCL4, CXCL4) was assessed using TaqMan® real-time PCR.
Expression of ELAM-1, VCAM-1, and ICAM-1 was assessed by immunostaining.
|
| |
Pre-ischemic PACE conditioning upregulated expression of IL-6, CCL3, CCL4,
and CXCL4, and downregulated expression of TNFα, GM-CSF, and IL-1α. Postischemic PACE conditioning significantly decreased expression of all evaluated genes. Pre- and postischemic PACE conditioning decreased expression of ELAM-1 and
ICAM-1.
Postischemic PACE conditioning revealed anti-inflammatory effect as confirmed
by decreased expression of inflammatory cytokines, chemokines, and cell adhesion molecules (ELAM-1 and ICAM-1).
|
| |
|
| |
Group 1: non-ischemic controls (N=5),
Group 2: five-hour ischemia controls (N=4),
Group 3: pre-ischemic (five-hour) PACE conditioning (N=4), and
Group 4: post-ischemic (five-hour) PACE conditioning (N=4)
|
| |
N/A
|
| |
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Ischemia
2005
|
| |
IIb
|
| |
Extracorporeal shockwave may enhance skin flap survival in
an animal model. Meirer R, Kamelger F, Huemer G, Wanner S, Piza, H British Assoc. of Plastic
Surgeons (2005); 58 53-57.
|
| |
N=20 Sprague-Dawley rats
Necrotic zone of skin flaps
|
| |
Surface area – standard deviation
|
| |
2.2±1.9%
|
| |
17.4±4.4%
|
| |
2500 impulses given to left upper corner of the flat
|
| |
N/A
|
| |
p<0.01
|
|
Treatment Indication
|
| |
LEVEL OF EVIDENCE
|
| |
Publication
|
| |
WOUND
POPULATION
|
| |
MEASUREMENT
|
| |
PACE RESULT
|
| |
STANDARD OF CARE RESULT
|
| |
TREATMENT DURATION
|
| |
Reported Serious Adverse Events
|
| |
STATISTICS and Conclusions
|
| |
Author
|
| |||||||||||||||||||||||||
| |
Journal
|
| |||||||||||||||||||||||||
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Ischemia
2008
|
| |
IIb
|
| |
Extracorporeal shockwave treatment modulates skin
fibroblast recruitment and leukocyte infiltration for enhancing extended skin-flap survival. Yur-Ren Kuo, MD, PhD; Chun-Ting Wang, BS1; Feng-Sheng Wang, PhD; Kuender
D. Yang, MD, PhD; Yuan-Cheng Chiang, MD; Ching-Jen Wang, MD Wound Repair Regen. 2009 Jan-Feb;17(1):80-7. doi: 10.1111/j.1524-475X.2008.00444.
|
| |
N=36 Sprague-Daley rats
Necrotic zone of skin flaps; random pattern extended dorsal skin flap
|
| |
Necrotic area of flaps
Number of apoptotic cells
Hydrogen peroxide expression
PCNA
rPH
8-OG
eNOS expression
|
| |
Group II area = 13 ± 2.6%
58%
23 MFI
97%
78%
65%
70%
|
| |
Group I area = 42±5.7%
80%
5 MFI
42%
40%
98%
38%
|
| |
Group 1: control group, no treatment
Group II: one session/500 impulses immediately post-op
Group III: two sessions/500 impulses, one immediately post-op and one one-day
post-op
|
| |
N/A
|
| |
P=0.003
P<0.001
P<0.01
P<0.001
P<0.001
P<0.001
P<0.001
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Ischemia
Oxygenation
2010
|
| |
IIb
|
| |
Extracorporeal shockwave treatment protects skin flaps
against ischemia-reperfusion injury. Reichenberger MA, Heimer S, Schaefer A, Lass U, Gebhard MM, Germann G, Engel H, Köllensperger E, Leimer U, Mueller W. Injury 2012 Mar;43(3):374-80. doi: 10.1016/j.injury.2011.11.019. Epub 2011 Dec 18.
|
| |
N=24 rats
|
| |
Mean area of flap necrosis
|
| |
27.2% ±9.6%
|
| |
(46.1% ± 7.9%
|
| |
Group I (sham, no ischemic insult)
Group II (ischemia control)
Group III (ESWT)
|
| |
N/A
|
| |
Study 15
(P=0.05)
|
|
Treatment Indication
|
| |
LEVEL OF EVIDENCE
|
| |
Publication
|
| |
WOUND
POPULATION
|
| |
MEASUREMENT
|
| |
PACE RESULT
|
| |
STANDARD OF CARE RESULT
|
| |
TREATMENT DURATION
|
| |
Reported Serious Adverse Events
|
| |
STATISTICS and Conclusions
|
| |
Author
|
| |||||||||||||||||||||||||
| |
Journal
|
| |||||||||||||||||||||||||
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Ischemia
2005
|
| |
IIb
|
| |
Extracorporeal shockwave treatment in ischemic tissues:
What is the appropriate number of shockwave impulses? Kamelger F, Oehlbauer M, Piza H, Meirer R:. J. Reconstr Microsurg; doi 10.1055/s-002901234296[49]
|
| |
N=42 Sprague-Dawley rats
Epigastric skin flap
|
| |
Area of necrotic tissue after seven days, post-treatment
|
| |
Group 1 (200 shocks) = 17.22%
Group 2 (500 shocks) = 6.64%
Group 3 (1500 shocks) = 12.49%
Group 4 (2500 shocks) = 10.62%
Group 5 (5000 shocks) = 30.04%
|
| |
26.3%
|
| |
Group 1: 200 impulses at 0.11 mJ/mm2
Group 2: 500 impulses at 0.11 mJ/mm2
Group 3: 1500 impulses at 0.11 mJ/mm2
Group 4: 2500 impulses at 0.11 mJ/mm2
Group 5: 5000 impulses at 0.11 mJ/mm2
Group 6:control; no treatment
|
| |
N/A
|
| |
P=0.228
P=0.008
P=0.047
P=0.006
P=0.995
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Plantar Fasciitis
2003-2005
|
| |
Ib
|
| |
Long-term results of Extracorporeal Shock wave Treatment
for Plantar Fasciitis;
Wang CJ et al.
Am. Journal Sports Medicine
(2006), 34:4 592-596
|
| |
N=149
Prospective placebo-controlled clinical trial to determine the safety and
efficacy of ESW treatment for chronic heel pain syndrome
|
| |
Investigate the effects of ESW on patients with an established diagnosis of
chronic heel pain
|
| |
69.1% excellent, 13.6% good, 6.2% fair and 11.1% poor
|
| |
0% excellent, 55% good, 36% fair and 9% poor
|
| |
Efficacy-related secondary endpoints were measured at 3, 12, 60, and 72
months.
|
| |
There were no device related complications. No systemic or local
complications such as hematoma or ecchymosis occurred for which the patient required special treatment.
|
| |
p<0.001.
The directed application electrohydraulic-generated shock waves to the
insertion of the plantar fascia onto the calcaneus is a safe and effective nonsurgical method for treating chronic, recalcitrant heel pain syndrome that has been present for at least 6 months and has been refractory to other commonly
used nonoperative therapies.
|
|
Treatment Indication
|
| |
LEVEL OF EVIDENCE
|
| |
Publication
|
| |
WOUND
POPULATION
|
| |
MEASUREMENT
|
| |
PACE RESULT
|
| |
STANDARD OF CARE RESULT
|
| |
TREATMENT DURATION
|
| |
Reported Serious Adverse Events
|
| |
STATISTICS and Conclusions
|
| |
Author
|
| |||||||||||||||||||||||||
| |
Journal
|
| |||||||||||||||||||||||||
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Plantar Fasciitis
2002-2004
|
| |
Ib
|
| |
Treatment of Painful Heel Syndrome with Shock Waves.
Chen HS, Chen LM, Wuang
TW.
Clinical Orthopedics
and Related Research, No. 387, pp 41-46.
|
| |
N=80
Prospective clinical trial to determine the safety and efficacy of ESW
treatment for chronic heel pain syndrome
|
| |
Investigate the effects of ESW on heel pain involved 80 patients; six of whom
received bi-lateral treatment. Pain intensity was measured via VAS scale of 1-10.
|
| |
Significantly better in 52.9%, slightly better in 17.6%, and unchanged in
8.8% of patients (68 heels) at 12 week follow-up. At 24 week follow-up, no complaints in 59.3%, significantly better in 27.7%, slightly better in 13% (54 heels). No patient reported worsening symptoms. 17 patients (18 heels) who did
not respond favorably to the first treatment had significantly better results after a second treatment.
|
| |
N/A
|
| |
Efficacy-related secondary endpoints were measured at 12 and 24 weeks.
|
| |
There were no device related complications. No systemic or local
complications such as hematoma or ecchymosis occurred for which the patient required special treatment.
|
| |
p<0.001
Treatment of painful heel syndrome with
shock waves has produced a high rate of success
in pain relief and functional restoration
with negligible complications.
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Delayed Union/Non-union fracture
1996-1999
|
| |
|
| |
Treatment of nonuions of long bone fractures with shock
waves. Wang CJ, Chen HS, et al (2001):
Clin Orth & Related
Res; 387, 95-101.
|
| |
N=130
|
| |
Percent callus formation (closing of fracture gap);
Consolidation of fracture (closure)
|
| |
% callus formation: at 3 mos, 25% of fractures had >75% callus formation
in the fracture gap, while by 12 months fully two thirds of fractures had >75% callus in the fracture gap.
Fracture healing: 3 months of the initial treatment, 28% of the fractures
were consolidated, 72% were unchanged, and none were worsened. By the end of 12 months, 71% were considered consolidated, 29% were unchanged, and none were considered worsened.
|
| |
N/A
|
| |
3 months and 12 months
|
| |
Petechial bleeding and hematomas were the predominant reported adverse events
with all self-resolving.
|
| |
p<0.01
Treatment of chronic bone fractures with
shock waves has produced a high rate of success
in pain relief and functional restoration
with negligible complications.
|
|
Treatment Indication
|
| |
LEVEL OF EVIDENCE
|
| |
Publication
|
| |
WOUND
POPULATION
|
| |
MEASUREMENT
|
| |
PACE RESULT
|
| |
STANDARD OF CARE RESULT
|
| |
TREATMENT DURATION
|
| |
Reported Serious Adverse Events
|
| |
STATISTICS and Conclusions
|
| |
Author
|
| |||||||||||||||||||||||||
| |
Journal
|
| |||||||||||||||||||||||||
|
Delayed Union/Non-union fracture
2003-2006
|
| |
|
| |
Extracorporeal Shock Wave Treatment of Non- or Delayed
Union of Proximal Metatarsal Fractures
Richard G. Alvarez, MD;
Brandon Cincere, MD; Chandra Channappa, MD; Richard Langerman, DO; Robert Schulte, DPM; Juha Jaakkola, MD; Keith Melancon, MD; Michael Shereff, MD; G. Lee Cross III, MD
American Orthopaedic
Foot & Ankle Society DOI: 10.3113/FAI.2011.0746
|
| |
N=31
Nonunion or a delayed healing of a proximal metatarsal, and in the fifth
metatarsal excluded to be fractured in Zone 2 and/or Zone 3.
|
| |
Collect safety and effectiveness data about ESW treatment for a nonunion or a
delayed healing of the 2nd, 3rd, or 4th metatarsal and Zone 2 and/or Zone 3 of the fifth metatarsal.
|
| |
12-week visit was 71.0% (22/31) with 9 treatments failing to respond. None of
the subjects who failed treatment chose to be retreated. The success/fail criteria was evaluated at the 6- and 12-month follow-up, showing continued treatment success rates of 88.5% (23/26) and 90.0% (18/20), respectively. Twenty-four
(24) subjects experienced continued success with 5 subjects experiencing continued failure. Eighteen (18) subjects assessed by X-ray at 6-months showed successful healing and 6 subjects were classified as partially healed. The
12-month X-ray follow-up showed 19 subjects as either healed (n=18) or partially healed (n=1).
|
| |
N/A
|
| |
12 week with followup at 12 and 24 months
|
| |
Adverse events which occurred within 48 hours post-treatment and associated
with the device were 11.8% (4/34) which included: two subjects with mild swelling, one with minor bruising (subcutaneous hemorrhage), and one showing petechiae. Two additional events attributed to the device but that occurred 7-10
days post-treatment were mild swelling and tenderness in the treated foot.
There were no unanticipated adverse device effects reported during this study
and no SAEs reported.
|
| |
With a high success rate and low adverse effect profile,
we believe ESWT was a safe, noninvasive and effective
alternative to surgical management of delayed union and
nonunion of proximal metatarsal fractures and Zone 2 fifth
metatarsal fractures.
|
|
Treatment Indication
|
| |
LEVEL OF EVIDENCE
|
| |
Publication
|
| |
WOUND POPULATION
|
| |
MEASUREMENT
|
| |
PACE RESULT
|
| |
STANDARD OF CARE RESULT
|
| |
TREATMENT DURATION
|
| |
Reported Serious Adverse Events
|
| |
STATISTICS and Conclusions
|
|||
| |
Author
|
| ||||||||||||||||||||||||||||
| |
Journal
|
| ||||||||||||||||||||||||||||
|
Burns
Oxygenation
2008
|
| |
IIIb
|
| |
Extracorporeal shockwaves, a new non-surgical method to
treat severe burns. Arnó A, García O, Hernán I, Sancho J, Acosta A, Barret JP. Burns. 2010 Sep;36(6):844-9. doi: 10.1016/j.burns.2009.11.012. Epub 2010 Jan 13
|
| |
N=15
Patients with <5% TBSA (total body surface area) deep partial/full
thickness burns
|
| |
Effect of ESWT on deep partial/full thickness burns
|
| |
Healed uneventfully prior to three weeks
|
| |
80%
|
| |
N/A
|
| |
Two ESWT sessions performed on third and fifth day after injury, LDI used
prior to each session
|
| |
No device related adverse events were reported
|
| |
After one session, burns had a significant increase in perfusion, as
evidenced by the LDI images.
|
| |
Required surgical debridement and grafting
|
| |
As many as 15%
|
| |||||||||||||||||||||||||
| |
Developed hypertrophic scarring
|
| |
5%
|
| |||||||||||||||||||||||||
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| ||
|
Venous leg ulcers
Diabetic foot ulcers
2005
|
| |
IIIb
|
| |
The effect of ESWT on chronic leg ulcers in diabetic
patients: A Clinical Trial. Presented at Wound Care Congress, Kamelger F, Meirer R, Piza H Nov 2006, Colorado Springs, CO
|
| |
N=9
Diabetic foot ulcers
|
| |
Wound area
Average wound closure rate
|
| |
Decreased from 5.1±5.5 cm2 to 0.4±0.5 cm2
4.5±3.1% per day
|
| |
N/A
|
| |
12 weeks
|
| |
No device related adverse events were reported
|
| |
p<0.05
Shockwave led to improved vascularization in ischemic tissue, which resulted
in more rapid wound closure.
|
|||
|
Treatment Indication
|
| |
LEVEL OF EVIDENCE
|
| |
Publication
|
| |
WOUND POPULATION
|
| |
MEASUREMENT
|
| |
PACE RESULT
|
| |
STANDARD OF CARE RESULT
|
| |
TREATMENT DURATION
|
| |
Reported Serious Adverse Events
|
| |
STATISTICS and Conclusions
|
| |
Author
|
| |||||||||||||||||||||||||
| |
Journal
|
| |||||||||||||||||||||||||
|
BURNS
2007
|
| |
IVb
|
| |
A Pilot Study of Pulsed Acoustic Wave Therapy (dermaPACE®)
in the Treatment of Mid-Deep Dermal Burns, Aslam R, Rashid A, Moiemen. ISBS, Montreal, Canada, Sept 2008. Poster and presentation.
|
| |
N=10
Mid-dermal burns; patients serving as own control
|
| |
Healing rate (complete reepithelialization)
|
| |
14 days (median, range = 14-30) to healing
|
| |
17.5 days (median, range = 14-25) to healing
|
| |
dermaPACE 500 impulses to half of affected area at initial visit and 48 hours
later.
|
| |
No device related adverse events were reported.
|
| |
20% faster healing rate (days)
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
VENOUS LEG ULCERS
ARTERIAL ULCERS
2006
|
| |
IVb
|
| |
Effects on Healing on Split Skin Grafting, Petschke, et al Poster presented at Wound Care Congress, Houston, TX 2007
|
| |
N=19
Arterial or venous ulcers
|
| |
Graft uptake complete (100%)
Graft uptake incomplete
|
| |
84.2% of patients (16/19); mean healing 12.2 days
15.8% of patients (3/19)
|
| |
N/A
|
| |
One treatment of 500 impulses of ESWT (one patient received two treatments);
|
| |
No device related adverse events were reported.
|
| |
Treating grafts with ESWT suggests possible beneficial results in uptake of
skin grafting.
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
BURNS
2006
|
| |
IVb
|
| |
Effects on Healing Deep Partial Thickness Burns Using ESW, Petschke, et al
Abstract on file
|
| |
N=5
Deep partial thickness burns
|
| |
Complete healing
Healing time frame
|
| |
100%
Mean=13.2 days (range seven to 20 days)
|
| |
N/A
|
| |
One treatment of 500 impulses of ESWT
|
| |
No device related adverse events were reported.
|
| |
ESWT treatment suggests beneficial results in healing and healing times.
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
PRESSURE ULCERS
2007
|
| |
IVb
|
| |
Pulse Acoustic Cellular Expression Technology for Chronic
Soft Tissue Wounds – Application Study in 10 patients, poster presented at WUWHS 2008
|
| |
N=10
Chronic soft tissue wounds
|
| |
Observe response of wounds to ESWT (dermaPACE) in conjunction with standard
of care
|
| |
Wound improvement seen in all 10 patients. Three of 10 patients’ wounds
healed completely.
|
| |
N/A
|
| |
Multiple, days 1, 4, 7, 11, and then weekly thereafter (as many as nine
treatments).
Shock count varied depending on wound size.
|
| |
No device related adverse events were reported.
|
| |
ESWT treatment suggests beneficial results in healing and healing times.
|
|
Treatment Indication
|
| |
LEVEL OF EVIDENCE
|
| |
Publication
|
| |
WOUND POPULATION
|
| |
MEASUREMENT
|
| |
PACE RESULT
|
| |
STANDARD OF CARE RESULT
|
| |
TREATMENT DURATION
|
| |
Reported Serious Adverse Events
|
| |
STATISTICS and Conclusions
|
| |
Author
|
| |||||||||||||||||||||||||
| |
Journal
|
| |||||||||||||||||||||||||
|
OXYGENATION
DIABETIC FOOT ULCER
2019
|
| |
IVb
|
| |
A Non-invasive Focused Extracorporeal Shock Wave Therapy
System Promotes Increased Tissue Oxygen Saturation in Chronic Wounds in Persons with Diabetes Windy Cole, Stacey Coe, Greg Maislin, Valerie Marmolejo American Journal of
Nursing Science 2021; 10(3): 166-172 http://www.sciencepublishing
group.com/j/ajns doi: 10.11648/j.ajns.20211003.14
ISSN: 2328-5745 (Print); ISSN: 2328-5753
|
| |
N=15
|
| |
Change in oxygen saturation pre- and post-treatment
Change in wound volume from baseline
|
| |
Final StO2% = 75.7% ± 11.1%
Seven of 15 wounds fully resolved (healed)
Excluding wounds that resolved, median volume reduction = 4.54cm2
|
| |
Baseline StO2% =
60.7%±18.9%
|
| |
Four ESWT treatments (one/week for four weeks); number of shocks based upon
wound area
Treatment and observation period = four weeks
|
| |
No device related adverse events were reported.
|
| |
P=0.0008
The results suggest that ESWT promotes formation of granulation tissue and
increased tissue oxygenation saturation as determined with a hand-held NIRS device.
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
VENOUS LEG ULCER
2016
|
| |
IVb
|
| |
Treating venous leg ulcers with Extracorporeal Shockwave
Technology (ESWT), Miller c, Kapp S, Green J, McGuiness W, Woddward M, Wounds International 2017,
Vol 8 Iss 3
|
| |
N=6
|
| |
Change in wound area
|
| |
A reduction in wound size was observed for five patients. Only one patient,
while achieving improved tissue quality, had an increased wound size and an alternative.
|
| |
N/A
|
| |
Pain was reported by one patient. No other device related adverse events.
|
| |
No device related adverse events were reported.
|
| |
This case series presents limited level of evidence; however results are
suggestive of further clinical evaluation.
|
|
•
|
Commercialize and support the domestic distribution of our UltraMIST and PACE systems to treat wounds;
|
|
•
|
Reduce and normalize operating costs to support growth;
|
|
•
|
Develop and commercialize our noninvasive biological response activating devices in the regenerative medicine area for the
treatment of skin, musculoskeletal tissue, and vascular structures; and
|
|
•
|
Support the global distribution of our products.
|
|
Patent #/
Application #
|
| |
Title
|
| |
Expiration Date
|
| |
Jurisdiction
|
|
US 7,867,178
|
| |
Apparatus for generating shock waves with piezoelectric fibers integrated in a composite
|
| |
Sep 29, 2027
|
| |
USA
|
|
US 8,088,073
|
| |
Device for the application of acoustic shock waves
|
| |
Jun 23, 2025
|
| |
USA
|
|
US 8,092,401
|
| |
Method and apparatus for producing shock waves for medical applications
|
| |
Feb 21, 2027
|
| |
USA
|
|
US 8,556,813
|
| |
Extracorporeal pressure shock wave device
|
| |
Sept 12, 2031
|
| |
USA
|
|
US 8,961,441
|
| |
Medical treatment system including an ancillary medical treatment apparatus with an associated
data storage medium
|
| |
March 13, 2032
|
| |
USA
|
|
US 9,161,768
|
| |
Extracorporeal pressure shock wave devices with reversed applicators and methods for using
these devices
|
| |
Aug 16, 2030
|
| |
USA
|
|
US 9,198,825
|
| |
Increase electrode life in devices used for extracorporeal shockwave therapy (ESWT)
|
| |
Aug 24, 2033
|
| |
USA
|
|
US 9,522,011
|
| |
Shock wave applicator with movable electrode
|
| |
July 8, 2030
|
| |
USA
|
|
US 9,566,209
|
| |
Shock wave electrodes with fluid holes
|
| |
June 21, 2033
|
| |
USA
|
|
US 10,058,340
|
| |
Extracorporeal pressure shock wave devices with multiple reflectors and methods for using these
devices
|
| |
Nov 9, 2033
|
| |
USA
|
|
US 10,769,249
|
| |
Distributor product programming system
|
| |
Feb 24, 2038
|
| |
USA
|
|
US 11,666,348
|
| |
Intracorporeal expandable shock wave reflector
|
| |
July 8, 2030
|
| |
USA
|
|
EP 2451422
|
| |
Usage of extracorporeal and intracorporeal pressure shock waves in medicine
|
| |
July 8, 2030
|
| |
Great Britain, France, Germany, Italy and Spain
|
|
KR 10-2255975
|
| |
Distributor product programming system
|
| |
May 25, 2038 (app.)
|
| |
South Korea
|
|
Patent #/
Application #
|
| |
Title
|
| |
Expiration Date
|
| |
Jurisdiction
|
|
US 8,343,420
|
| |
Methods and devices for cleaning and sterilization with shock waves
|
| |
July 2, 2031
|
| |
USA
|
|
US 8,728,809
|
| |
Use of pressure waves for stimulation, proliferation, differentiation and post-implantation
viability of stem cells
|
| |
August 25, 2031
|
| |
USA
|
|
US 9,119,888
|
| |
Methods for cleaning and sterilization of implant tissue ex vivo with shock waves
|
| |
Sept 17, 2030
|
| |
USA
|
|
US 10,238,405
|
| |
Blood vessel treatment with intracorporeal pressure shock waves
|
| |
Jan 19, 2032
|
| |
USA
|
|
Patent #/
Application #
|
| |
Title
|
| |
Expiration Date
|
| |
Jurisdiction
|
|
US 10,569,106
|
| |
Tissue disinfection with acoustic pressure shock waves
|
| |
Sep 28, 2038
|
| |
USA
|
|
US 10,639,051
|
| |
Occlusion and clot treatment with intracorporeal pressure shock waves
|
| |
Sep 29, 2031
|
| |
USA
|
|
US 10,888,715
|
| |
Acoustic pressure shock waves used for personalized medical treatment of tissue conditions
|
| |
May 12, 2039
|
| |
USA
|
|
US 11,684,806
|
| |
Infected Prosthesis and Implant Treatment with Acoustic Pressure Shock Waves
|
| |
July 22, 2037
|
| |
USA
|
|
US 11,771,781
|
| |
Reprocessing of contaminated reusable devices with direct contact of pressure waves
|
| |
May 6, 2041
|
| |
USA
|
|
EP 3117784
|
| |
Usage of intracorporeal pressure shock waves in medicine
|
| |
July 8, 2030
|
| |
Great Britain, France, Germany, Italy, Spain, Finland, Belgium, Denmark, Ireland, the
Netherlands, Norway, and Sweden
|
|
AU 2016250668
|
| |
Tissue disinfection with acoustic pressure shock waves
|
| |
March 22, 2036
|
| |
Australia
|
|
EP 3461438
|
| |
Combined intracorporeal and extracorporeal shock wave treatment system
|
| |
July 8, 2030
|
| |
Great Britain, France, Germany, and the Netherlands
|
|
EP 3285661
|
| |
Tissue disinfection with acoustic pressure shock waves
|
| |
April 22, 2036
|
| |
Great Britain, France, Germany, Ireland, and the Netherlands
|
|
AU 2017387130
|
| |
Acoustic pressure shock waves used for personalized medical treatment of tissue conditions
|
| |
Dec 29, 2037
|
| |
Australia
|
|
IL 267661
|
| |
Acoustic pressure shock waves used for personalized medical treatment of tissue conditions
|
| |
Dec 29, 2037 (app.)
|
| |
Israel
|
|
BR 112017022768
|
| |
Tissue disinfection with acoustic pressure shock waves
|
| |
April 22, 2036
|
| |
Brazil
|
|
Patent #/
Application #
|
| |
Title
|
| |
Expiration Date
|
| |
Jurisdiction
|
|
US 7,713,218
|
| |
Removable applicator nozzle for ultrasound wound therapy device
|
| |
May 6, 2028
|
| |
USA
|
|
US 7,785,277
|
| |
Removable applicator nozzle for ultrasound wound therapy device
|
| |
Jun 27, 2025
|
| |
USA
|
|
US 7,914,470
|
| |
Ultrasonic method and device for wound treatment
|
| |
June 27, 2023
|
| |
USA
|
|
US 8,491,521
|
| |
Removable multi-channel applicator nozzle
|
| |
May 3, 2028
|
| |
USA
|
|
US 11,224,767
|
| |
Systems and methods for producing and delivering ultrasonic therapies for wound treatment and
healing
|
| |
Nov 18, 2034
|
| |
USA
|
|
US 11,331,520
|
| |
Systems and methods for producing and delivering ultrasonic therapies for wound treatment and
healing
|
| |
Sept 7, 2035
|
| |
USA
|
|
US D733,319
|
| |
Ultrasonic treatment wand
(Design patent)
|
| |
June 30, 2029
|
| |
USA
|
|
US D733,321
|
| |
Ultrasonic treatment device
(Design patent)
|
| |
June 30, 2029
|
| |
USA
|
|
Patent #/
Application #
|
| |
Title
|
| |
Expiration Date
|
| |
Jurisdiction
|
|
IN 228689
|
| |
Ultrasonic method and device for wound treatment
|
| |
April 5, 2024
|
| |
India
|
|
HK 1119926
|
| |
Removable applicator nozzle for ultrasound wound therapy device
|
| |
June 23, 2026
|
| |
Hong Kong
|
|
CA 2,463,600
|
| |
Device and method for ultrasound wound debridement
|
| |
Aug 4, 2023
|
| |
Canada
|
|
CA 2,521,117
|
| |
Ultrasonic method and device for wound treatment
|
| |
April 5, 2024
|
| |
Canada
|
|
CA 2,931,612
|
| |
Systems and methods for producing and delivering ultrasonic therapies for wound treatment and
healing
|
| |
Nov 18, 2034
|
| |
Canada
|
|
EP 1893104
|
| |
Removable applicator nozzle for ultrasound wound therapy device
|
| |
July 8, 2026
|
| |
Belgium, France, Germany, Ireland, and Great Britain
|
|
AU 2021201720
|
| |
Systems and methods for producing and delivering ultrasonic therapies for wound treatment and
healing
|
| |
Nov 18, 2034
|
| |
Australia
|
|
•
|
Class I: general controls, such as labeling and adherence to quality system regulations;
|
|
•
|
Class II: special controls, pre-market notification (510(k)), specific controls such as performance standards, patient
registries, and post market surveillance, and additional controls such as labeling and adherence to quality system regulations; and
|
|
•
|
Class III: special controls and approval of a pre-market approval (“PMA”) application.
|
|
•
|
the FDA quality systems regulation, which governs, among other things, how manufacturers design, test, manufacture, exercise
quality control over, and document manufacturing of their products;
|
|
•
|
labeling and claims regulations, which prohibit the promotion of products for unapproved or “off-label” uses and impose other
restrictions on labeling;
|
|
•
|
the Medical Device Reporting regulation, which requires reporting to the FDA of certain adverse experiences associated with
use of the product; and
|
|
•
|
post market surveillance, including documentation of clinical experience and follow-on, confirmatory studies.
|
|
•
|
Do not reflect every expenditure, future requirements for capital expenditures or contractual commitments.
|
|
•
|
Do not reflect all changes in working capital needs.
|
|
•
|
Do not reflect the interest expense, or the amount necessary to service outstanding debt.
|
|
|
| |
Three months ended
September 30,
|
| |
Nine months ended
September 30,
|
||||||
|
(in thousands)
|
| |
2023
|
| |
2022
|
| |
2023
|
| |
2022
|
|
Net (Loss)/Income
|
| |
$(23,700)
|
| |
$(1,139)
|
| |
$(44,042)
|
| |
$(4,596)
|
|
Non-GAAP Adjustments:
|
| |
|
| |
|
| |
|
| |
|
|
Interest expense
|
| |
3,845
|
| |
3,821
|
| |
12,504
|
| |
9,972
|
|
Depreciation and amortization
|
| |
266
|
| |
235
|
| |
780
|
| |
681
|
|
EBITDA
|
| |
(19,589)
|
| |
2,917
|
| |
(30,758)
|
| |
6,057
|
|
Non-GAAP Adjustments for Adjusted EBITDA:
|
| |
|
| |
|
| |
|
| |
|
|
Change in fair value of derivative liabilities
|
| |
19,325
|
| |
(5,252)
|
| |
29,943
|
| |
(16,597)
|
|
Other non-cash or one-time charges:
|
| |
|
| |
|
| |
|
| |
|
|
Release of historical accrued employee compensation
expenses
|
| |
—
|
| |
—
|
| |
(1,250)
|
| |
—
|
|
Shares for Services
|
| |
—
|
| |
—
|
| |
224
|
| |
888
|
|
Loss on issuance of debt
|
| |
—
|
| |
—
|
| |
—
|
| |
3,434
|
|
Loss on extinguishment of debt
|
| |
—
|
| |
86
|
| |
—
|
| |
297
|
|
Adjusted EBITDA
|
| |
$(264)
|
| |
$(2,249)
|
| |
$(1,841)
|
| |
$(5,921)
|
|
|
| |
For the Three Months Ended
|
| |
For the Nine Months Ended
|
||||||||||||||||||
|
|
| |
September 30,
|
| |
Change
|
| |
September 30,
|
| |
Change
|
||||||||||||
|
|
| |
2023
|
| |
2022
|
| |
$
|
| |
%
|
| |
2023
|
| |
2022
|
| |
$
|
| |
%
|
|
Revenues:
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Total Revenue
|
| |
$4,953
|
| |
$4,166
|
| |
$787
|
| |
19%
|
| |
$13,404
|
| |
$11,242
|
| |
$2,162
|
| |
19%
|
|
Cost of Revenues
|
| |
1,412
|
| |
1,157
|
| |
255
|
| |
22%
|
| |
3,876
|
| |
3,141
|
| |
735
|
| |
23%
|
|
Gross Margin
|
| |
3,541
|
| |
3,009
|
| |
532
|
| |
18%
|
| |
9,528
|
| |
8,101
|
| |
1,427
|
| |
18%
|
|
Operating Expenses:
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
General and administrative
|
| |
2,681
|
| |
3,498
|
| |
(817)
|
| |
-23%
|
| |
6,678
|
| |
9,484
|
| |
(2,806)
|
| |
-30%
|
|
Selling and marketing
|
| |
1,039
|
| |
1,650
|
| |
(611)
|
| |
-37%
|
| |
3,430
|
| |
5,037
|
| |
(1,607)
|
| |
-32%
|
|
Research and development
|
| |
165
|
| |
157
|
| |
8
|
| |
5%
|
| |
436
|
| |
494
|
| |
(58)
|
| |
-12%
|
|
|
| |
For the Three Months Ended
|
| |
For the Nine Months Ended
|
||||||||||||||||||
|
|
| |
September 30,
|
| |
Change
|
| |
September 30,
|
| |
Change
|
||||||||||||
|
|
| |
2023
|
| |
2022
|
| |
$
|
| |
%
|
| |
2023
|
| |
2022
|
| |
$
|
| |
%
|
|
Depreciation and amortization
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
|
| |
187
|
| |
189
|
| |
(2)
|
| |
-1%
|
| |
563
|
| |
575
|
| |
(12)
|
| |
-2%
|
|
Operating Loss
|
| |
(531)
|
| |
(2,485)
|
| |
1,954
|
| |
-79%
|
| |
(1,579)
|
| |
(7,489)
|
| |
5,910
|
| |
-79%
|
|
Other Income (Expense), net
|
| |
(23,169)
|
| |
1,346
|
| |
(24,515)
|
| |
nm
|
| |
(42,463)
|
| |
2,893
|
| |
(45,356)
|
| |
nm
|
|
Net (Loss) Income
|
| |
$(23,700)
|
| |
$(1,139)
|
| |
(22,561)
|
| |
nm
|
| |
$(44,042)
|
| |
$(4,596)
|
| |
(39,446)
|
| |
nm
|
|
|
| |
For the
Three Months Ended
September 30,
|
| |
Change
|
| |
For the
Six Months Ended
September 30,
|
| |
Change
|
||||||||||||
|
|
| |
2023
|
| |
2022
|
| |
$
|
| |
%
|
| |
2023
|
| |
2022
|
| |
$
|
| |
%
|
|
Interest expense
|
| |
$(3,845)
|
| |
$(3,821)
|
| |
$(24)
|
| |
1%
|
| |
$(12,504)
|
| |
$(9,972)
|
| |
$(2,532)
|
| |
25%
|
|
Change in fair value of derivatives
|
| |
(19,325)
|
| |
5,252
|
| |
(24,577)
|
| |
-468%
|
| |
(29,943)
|
| |
16,597
|
| |
(46,540)
|
| |
-280%
|
|
Loss on issuance of debt
|
| |
—
|
| |
—
|
| |
—
|
| |
nm
|
| |
—
|
| |
(3,434)
|
| |
3,434
|
| |
nm
|
|
Loss on extinguishment of debt
|
| |
—
|
| |
(86)
|
| |
86
|
| |
nm
|
| |
—
|
| |
(297)
|
| |
297
|
| |
nm
|
|
Other expense
|
| |
1
|
| |
1
|
| |
—
|
| |
nm
|
| |
(16)
|
| |
(1)
|
| |
(15)
|
| |
nm
|
|
Other (expense)/income, net
|
| |
$(23,169)
|
| |
$1,346
|
| |
$(24,515)
|
| |
-1821%
|
| |
$(42,463)
|
| |
$2,893
|
| |
$(45,356)
|
| |
-1568%
|
|
|
| |
For the Years Ended
December 31,
|
| |
Change
|
||||||
|
(in thousands)
|
| |
2022
|
| |
2021
|
| |
$
|
| |
%
|
|
Revenue
|
| |
16,742
|
| |
$13,010
|
| |
$3,732
|
| |
29%
|
|
Cost of revenue
|
| |
4,331
|
| |
4,986
|
| |
(655)
|
| |
-13%
|
|
Gross margin
|
| |
12,411
|
| |
8,024
|
| |
4,387
|
| |
55%
|
|
Operating expenses:
|
| |
|
| |
|
| |
|
| |
|
|
General and administrative
|
| |
12,556
|
| |
11,690
|
| |
866
|
| |
7%
|
|
Selling and marketing
|
| |
7,474
|
| |
8,591
|
| |
(1,117)
|
| |
-13%
|
|
Research and development
|
| |
567
|
| |
1,101
|
| |
(534)
|
| |
-49%
|
|
Depreciation and amortization
|
| |
766
|
| |
784
|
| |
(18)
|
| |
-2%
|
|
Operating loss
|
| |
(8,952)
|
| |
(14,142)
|
| |
5,190
|
| |
-37%
|
|
Other income (expense), net
|
| |
(1,339)
|
| |
(13,089)
|
| |
11,750
|
| |
-90%
|
|
Income tax expense
|
| |
2
|
| |
28
|
| |
(26)
|
| |
-93%
|
|
Net loss
|
| |
$(10,293)
|
| |
$(27,259)
|
| |
$16,966
|
| |
-62%
|
|
|
| |
2022
|
| |
2021
|
| |
$
|
| |
%
|
|
Interest expense
|
| |
$(14,132)
|
| |
$(7,095)
|
| |
$(7,037)
|
| |
99%
|
|
Change in fair value of derivatives
|
| |
16,654
|
| |
(2,622)
|
| |
19,276
|
| |
nm
|
|
Loss on issuance of debt
|
| |
(3,434)
|
| |
(3,572)
|
| |
138
|
| |
-4%
|
|
Gain/(loss) on extinguishment of debt
|
| |
(418)
|
| |
204
|
| |
(622)
|
| |
nm
|
|
Loss on foreign currency exchange
|
| |
(9)
|
| |
(4)
|
| |
(5)
|
| |
125%
|
|
Other expense, net
|
| |
$(1,339)
|
| |
$(13,089)
|
| |
$11,750
|
| |
-90%
|
|
|
| |
For the nine months ended
September 30,
|
| |
For the years ended
December 31,
|
||||||
|
(in thousands)
|
| |
2023
|
| |
2022
|
| |
2022
|
| |
2021
|
|
Cash flows used by operating activities
|
| |
$(3,253)
|
| |
$(13,176)
|
| |
$(17,169)
|
| |
$(6,409)
|
|
Cash flows (used by) provided by investing activities
|
| |
$(156)
|
| |
$1,022
|
| |
$332
|
| |
$(529)
|
|
Cash flows provided by financing activities
|
| |
$3,356
|
| |
$12,664
|
| |
$17,384
|
| |
$5,121
|
|
Name
|
| |
Age
|
| |
Position Held
|
|
Morgan Frank
|
| |
51
|
| |
Director, Chairman of the Board, Chief Executive Officer
|
|
Toni Rinow
|
| |
58
|
| |
Chief Financial Officer
|
|
Andrew Walko
|
| |
39
|
| |
President
|
|
Kevin A. Richardson, II
|
| |
54
|
| |
Director, Chief Strategy Officer
|
|
Peter Stegagno
|
| |
63
|
| |
Chief Operating Officer
|
|
Iulian Cioanta, PhD
|
| |
60
|
| |
Chief Science and Technology Officer
|
|
Jeff Blizard
|
| |
54
|
| |
Director
|
|
Ian Miller
|
| |
47
|
| |
Director
|
|
Chantell Preston
|
| |
49
|
| |
Director
|
|
A. Michael Stolarski
|
| |
52
|
| |
Director
|
|
James Tyler
|
| |
65
|
| |
Director
|
|
•
|
appointing, compensating, retaining, evaluating, terminating and overseeing the Company’s independent registered public
accounting firm;
|
|
•
|
discussing with the Company’s independent registered public accounting firm their independence from management;
|
|
•
|
reviewing, with the Company’s independent registered public accounting firm, the scope and results of their audit;
|
|
•
|
approving all audit and permissible non-audit services to be performed by the Company’s independent registered public
accounting firm;
|
|
•
|
overseeing the financial reporting process and discussing with management and the Company’s independent registered public
accounting firm the quarterly and annual financial statements that the Combined Company files with the SEC;
|
|
•
|
overseeing the Company’s financial and accounting controls and compliance with legal and regulatory requirements;
|
|
•
|
reviewing the Company’s policies on risk assessment and risk management;
|
|
•
|
reviewing related person transactions; and
|
|
•
|
establishing procedures for the confidential anonymous submission of concerns regarding questionable accounting, internal
controls or auditing matters.
|
|
•
|
reviewing and approving the corporate goals and objectives, evaluating the performance of and reviewing and approving the
compensation of the Combined Company’s Chief Executive Officer, and the Chief Executive Officer may not be present during voting or deliberations on his or her compensation;
|
|
•
|
overseeing an evaluation of the performance of and reviewing and setting or making recommendations to the Combined Company
Board regarding the compensation of the Combined Company’s other executive officers;
|
|
•
|
reviewing and approving or making recommendations to the Combined Company Board regarding the Combined Company’s incentive
compensation and equity-based plans, policies and programs;
|
|
•
|
reviewing and approving all employment agreement and severance arrangements for the Combined Company’s executive officers;
|
|
•
|
making recommendations to the Combined Company Board regarding the compensation of the Combined Company’s directors; and
|
|
•
|
retaining and overseeing any compensation consultants.
|
|
•
|
identifying individuals qualified to become members of the Combined Company Board, consistent with criteria approved by the
Combined Company Board;
|
|
•
|
overseeing succession planning for the Combined Company’s Chief Executive Officer and other executive officers;
|
|
•
|
periodically reviewing the Combined Company Board’s leadership structure and recommending any proposed changes to the
Combined Company Board;
|
|
•
|
overseeing an annual evaluation of the effectiveness of the Combined Company Board and its committees; and
|
|
•
|
developing and recommending to the Combined Company Board a set of corporate governance guidelines.
|
|
•
|
Kevin A. Richardson, II, former Chief Executive Officer;
|
|
•
|
John Schlechtweg, former Chief Revenue Officer; and
|
|
•
|
Lisa Sundstrom, former Chief Talent Officer and Chief Financial Officer.
|
|
Name and Position
|
| |
Year
|
| |
Salary(1)
|
| |
All other
compensation(2)
|
| |
Total
|
|
Kevin A. Richardson, II,
former Chief Executive Officer(3)
|
| |
2022
|
| |
430,583
|
| |
175,000
|
| |
605,583
|
| |
2021
|
| |
350,000
|
| |
49,310
|
| |
399,310
|
||
|
John Schlechtweg,
former Chief Revenue Officer
|
| |
2022
|
| |
317,500
|
| |
90,483
|
| |
407,983
|
| |
2021
|
| |
—
|
| |
—
|
| |
—
|
||
|
Lisa Sundstrom,
former Chief Talent Officer and Chief Financial Officer
|
| |
2022
|
| |
208,333
|
| |
82,735
|
| |
291,068
|
| |
2021
|
| |
200,000
|
| |
52,023
|
| |
252,023
|
|
(1)
|
Amounts reflect the following:
|
|
(i)
|
the salary guaranteed by Mr. Richardson’s employment agreement with SANUWAVE and
|
|
(ii)
|
an aggregate amount of $60,000 for fees earned or paid in cash for Mr. Richardson’s service as a director in fiscal 2022.
|
|
(2)
|
Includes bonus, health, dental, life and disability insurance premiums and 401(k) matching contributions.
|
|
(3)
|
Mr. Richardson stepped down as Chief Executive Officer on May 23, 2023 to serve as SANUWAVE’s Chief Strategy Officer.
|
|
Name
|
| |
Number of
securities
underlying
unexercised
options
exercisable
|
| |
Number of
securities
underlying
options
unexercisable
|
| |
Equity incentive
plan awards
number of
securities
underlying
unexercised
unearned
options
|
| |
Exercise
price
($)
|
| |
Expiration
Date
|
|
Kevin A. Richardson, former Chief Executive Officer
|
| |
115,000
|
| |
—
|
| |
—
|
| |
$0.35
|
| |
2/21/2023
|
|
|
| |
452,381
|
| |
—
|
| |
—
|
| |
$0.11
|
| |
10/1/2025
|
|
|
| |
297,619
|
| |
—
|
| |
—
|
| |
$0.06
|
| |
10/1/2025
|
|
|
| |
700,000
|
| |
—
|
| |
—
|
| |
$0.04
|
| |
6/16/2026
|
|
|
| |
594,300
|
| |
—
|
| |
—
|
| |
$0.18
|
| |
11/9/2026
|
|
|
| |
900,000
|
| |
—
|
| |
—
|
| |
$0.11
|
| |
6/14/2027
|
|
|
| |
1,100,000
|
| |
—
|
| |
—
|
| |
$0.21
|
| |
9/20/2028
|
|
|
| |
50,000
|
| |
—
|
| |
—
|
| |
$0.15
|
| |
8/26/2029
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Lisa E. Sundstrom, Chief Talent Officer
|
| |
65,000
|
| |
—
|
| |
—
|
| |
$0.35
|
| |
2/21/2023
|
|
|
| |
25,000
|
| |
—
|
| |
—
|
| |
$0.55
|
| |
5/7/2024
|
|
|
| |
301,587
|
| |
—
|
| |
—
|
| |
$0.11
|
| |
10/1/2025
|
|
|
| |
198,413
|
| |
—
|
| |
—
|
| |
$0.06
|
| |
10/1/2025
|
|
|
| |
500,000
|
| |
—
|
| |
—
|
| |
$0.04
|
| |
6/16/2026
|
|
|
| |
424,500
|
| |
—
|
| |
—
|
| |
$0.18
|
| |
11/9/2026
|
|
|
| |
60,000
|
| |
—
|
| |
—
|
| |
$0.11
|
| |
6/14/2027
|
|
|
| |
750,000
|
| |
—
|
| |
—
|
| |
$0.21
|
| |
9/20/2028
|
|
|
| |
50,000
|
| |
—
|
| |
—
|
| |
$0.15
|
| |
8/26/2029
|
|
Director
|
| |
Fee Earned or
paid in cash
(in thousands)
|
|
Morgan Frank
|
| |
$43
|
|
A. Michael Stolarski
|
| |
$70
|
|
Jeff Blizzard
|
| |
$63
|
|
Ian Miller
|
| |
$63
|
|
James Tyler
|
| |
$63
|
|
•
|
SANUWAVE has been or is to be a participant;
|
|
•
|
the amount involved exceeded or exceeds the lesser of (a) $120,000 or (b) one percent of the average of SANUWAVE’s total
assets at year-end for the fiscal years ended December 31, 2022 and 2021; and
|
|
•
|
any of SANUWAVE’s directors, executive officers or holders of more than 5% of its capital stock prior to the Business
Combination, or any immediate family member of, or person sharing the household with, any of these individuals, had or will have a direct or indirect material interest.
|
|
•
|
any person who is, or at any time during the applicable period was, one of the Company’s executive officers or one of the
Company’s directors;
|
|
•
|
any person who is known by the Combined Company to be the beneficial owner of more than 5% of the Company’s voting shares;
|
|
•
|
any immediate family member of any of the foregoing persons, which means any child, stepchild, parent, stepparent, spouse,
sibling, mother-in-law, father-in-law, son-in-law, daughter-in-law, brother-in-law or sister-in-law of a director, executive officer or a beneficial owner of more than 5% of the Company’s voting shares, and any person (other than a
tenant or employee) sharing the household of such director, executive officer or beneficial owner of more than 5% of the Company’s voting shares; and
|
|
•
|
any firm, corporation or other entity in which any of the foregoing persons is a partner or principal, or in a similar
position, or in which such person has a 10% or greater beneficial ownership interest.
|
|
•
|
each person who is known to be the beneficial owner of more than 5% of SEPA’s Common Stock and is expected to be the
beneficial owner of more than 5% of shares of the Common Stock of the Combined Company;
|
|
•
|
each of SEPA’s current executive officers and directors;
|
|
•
|
each person who is expected to become an executive officer or director of the Combined Company; and
|
|
•
|
all executive officers and directors of SEPA as a group pre-Business Combination, and all executive officers and directors of
the Combined Company as a group post-Business Combination.
|
|
1.
|
a “No Redemption” scenario where (i) no public SEPA Stockholders exercise their Redemption Rights in connection with the
Business Combination, (ii) SEPA issues 7,793,000 shares of Class A Common Stock as the Merger Consideration pursuant to the Merger Agreement, (iii) the outstanding Class B Common Stock converts into Class A Common Stock at a ratio of
0.277, (iv) the outstanding SEPA Public Warrants convert into 450,336 shares of Class A Common Stock, (v) the outstanding SEPA Private Placement Warrants convert into 400,000 shares of Class A Common Stock, (vi) no SANUWAVE Warrants or
SANUWAVE Convertible Notes are assumed by SEPA at the Closing (i.e., all outstanding SANUWAVE Warrants will be exercised, and all outstanding SANUWAVE Convertible Notes will be converted, prior
to the Closing of the Business Combination), (vii) SEPA issues 100,000 shares of Class A Common Stock to the Sponsor pursuant to the Sponsor Debt Conversion Agreement, (viii) SEPA issues 518,488 shares of Class A Common Stock in the
PIPE Investment at $10.00 per share and (ix) no shares of Class A Common Stock are issued pursuant to the Voting and Non-Redemption Agreements; and
|
|
2.
|
a “Maximum Redemption” scenario where (i) 622,747 of SEPA’s outstanding Public Shares are redeemed in connection with the
Business Combination, (ii) SEPA issues 7,793,000 shares of Class A Common Stock as the Merger Consideration pursuant to the Merger Agreement, (iii) the outstanding Class B Common Stock converts into Class A Common Stock at a ratio of
0.277, (iv) the outstanding SEPA Public Warrants convert into 450,336 shares of Class A Common Stock, (v) the outstanding SEPA Private Placement Warrants convert into 400,000 shares of Class A Common Stock, (vi) no SANUWAVE Warrants or
SANUWAVE Convertible Notes are assumed by SEPA at the Closing (i.e., all outstanding SANUWAVE Warrants will be exercised, and all outstanding SANUWAVE Convertible Notes will be converted, prior
to the Closing of the Business Combination), (vii) SEPA issues 100,000 shares of Class A Common Stock to the Sponsor pursuant to the Sponsor Debt Conversion Agreement, (viii) SEPA issues 518,488 shares of Class A Common Stock in the
PIPE Investment at $10.00 per share and (ix) no shares of Class A Common Stock are issued pursuant to the Voting and Non-Redemption Agreements.
|
|
|
| |
|
| |
|
| |
Post-Business Combination
|
|||||||||
|
|
| |
Prior to the
Transactions
|
| |
Assuming
No Redemption
|
| |
Assuming
Maximum Redemption
|
|||||||||
|
Name and Address of
Beneficial Owners(1)
|
| |
Number of
Shares
(All Classes
(#))(2)
|
| |
Percentage of
Outstanding
Common
Stock
(%)
|
| |
Number of
Shares
(#)
|
| |
Percentage
(%)
|
| |
Number of
Shares
(#)
|
| |
Percentage
(%)
|
|
Executive Officers and Directors
Prior to the Transactions:
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
M. Blair Garrou
|
| |
4,260,375(3)(4)
|
| |
74.1%
|
| |
2,254,909(7)
|
| |
20.0%
|
| |
2,081,421(8)
|
| |
18.6%
|
|
R. Andrew White
|
| |
3,465,375(4)
|
| |
60.2%
|
| |
1,059,909(8)
|
| |
9.4%
|
| |
1,059,909(8)
|
| |
9.5%
|
|
Winston Gilpin(5)
|
| |
10,000(6)
|
| |
*%
|
| |
2,770
|
| |
*%
|
| |
2,770
|
| |
—%
|
|
Christy Cardenas
|
| |
10,000(6)
|
| |
*%
|
| |
2,770
|
| |
*%
|
| |
2,770
|
| |
—%
|
|
Jay Gardner
|
| |
40,000(6)
|
| |
*%
|
| |
11,080
|
| |
*%
|
| |
11,080
|
| |
—%
|
|
Mia Mends
|
| |
40,000(6)
|
| |
*%
|
| |
11,080
|
| |
*%
|
| |
11,080
|
| |
—%
|
|
Carolyn Rodz
|
| |
40,000(6)
|
| |
*%
|
| |
11,080
|
| |
*%
|
| |
11,080
|
| |
—%
|
|
All Directors and Executive Officers as a group
(7 Individuals)
|
| |
4,440,375
|
| |
77.2%
|
| |
2,293,689
|
| |
20.3%
|
| |
2,120,201
|
| |
19.0%
|
|
Executive Officers and Directors
After the Transactions:
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Morgan Frank(9)
|
| |
—
|
| |
—%
|
| |
1,201,248
|
| |
10.6%
|
| |
1,201,248
|
| |
10.7%
|
|
Kevin A. Richardson, II(10)
|
| |
—
|
| |
—%
|
| |
122,580
|
| |
1.1%
|
| |
122,580
|
| |
1.1%
|
|
Michael Stolarski
|
| |
—
|
| |
—%
|
| |
469,703
|
| |
4.2%
|
| |
469,703
|
| |
4.2%
|
|
Jeff Blizard
|
| |
—
|
| |
—%
|
| |
—
|
| |
—%
|
| |
—
|
| |
—%
|
|
Ian Miller
|
| |
—
|
| |
—%
|
| |
60,501
|
| |
*%
|
| |
60,501
|
| |
*%
|
|
James Tyler
|
| |
—
|
| |
—%
|
| |
11,694
|
| |
*%
|
| |
11,694
|
| |
*%
|
|
Chantell Preston
|
| |
—
|
| |
—%
|
| |
—
|
| |
—%
|
| |
—
|
| |
—%
|
|
Toni Rinow
|
| |
—
|
| |
—%
|
| |
—
|
| |
—%
|
| |
—
|
| |
—%
|
|
Andrew Walko
|
| |
—
|
| |
—%
|
| |
—
|
| |
—%
|
| |
—
|
| |
—%
|
|
Peter Stegagno
|
| |
—
|
| |
—%
|
| |
11,993
|
| |
*%
|
| |
11,993
|
| |
*%
|
|
Iulian Cioanta, PhD
|
| |
—
|
| |
—%
|
| |
9,537
|
| |
*%
|
| |
9,537
|
| |
*%
|
|
Tim Hendricks
|
| |
—
|
| |
—%
|
| |
—
|
| |
—%
|
| |
—
|
| |
—%
|
|
All Directors and Executive Officers as a group
(12 Individuals)
|
| |
—
|
| |
—%
|
| |
1,887,256
|
| |
16.7%
|
| |
1,887,256
|
| |
16.9%
|
|
Greater than 5% Holders:
|
| |
|
| |
|
| |
|
| |
|
| |
—
|
| |
—
|
|
Mercury Sponsor Group I LLC
|
| |
3,465,375(4)
|
| |
60.2%
|
| |
1,059,909
|
| |
9.4%
|
| |
1,059,909
|
| |
9.5%
|
|
(1)
|
Unless otherwise noted, the business address of the Sponsor and each executive officer and director prior to the transactions
is 3737 Buffalo Speedway, Suite 1750, Houston, TX 77098. Unless otherwise noted, the business address of each executive officer and director after the transactions is 11495 Valley View Road Eden Prairie, MN 55344.
|
|
(2)
|
Interests shown consist of Class A Common Stock and Founder Shares, classified as shares of Class B Common Stock.
|
|
(3)
|
Mercury Houston Partners, LLC is the record holder of 130,023 shares of Class A Common Stock reported herein and Mercury
Affiliates XI, LLC is the record holder of 664,977 shares of Class A Common Stock reported herein. M. Blair Garrou is the sole manager of Mercury
|
|
(4)
|
Mercury Sponsor Group I LLC is the record holder of 2,415,375 shares of Class A Common Stock and 1,050,000 Founder Shares.
Each of M. Blair Garrou and R. Andrew White are the managers of Mercury Sponsor Group I LLC. Affiliates of M. Blair Garrou and R. Andrew White each own 50% of the economic interest of Mercury Sponsor Group I LLC. As such, each of M.
Blair Garrou and R. Andrew White may be deemed to have beneficial ownership of the Class A Common Stock and Class B Common Stock held directly by Mercury Sponsor Group I LLC. Each of M. Blair Garrou and R. Andrew White disclaim
beneficial ownership over any securities owned by the Sponsor in which he does not have any pecuniary interest.
|
|
(5)
|
GSqr Consulting, LLC is the record holder of the shares reported herein. Mr. Gilpin is the manager of GSqr Consulting, LLC. As
such, Mr. Gilpin may be deemed to have beneficial ownership of the Class B Common Stock held directly by GSqr Consulting, LLC to the extent of his pecuniary interest.
|
|
(6)
|
Interests shown consists solely of Founder Shares, classified as shares of Class B Common Stock.
|
|
(7)
|
Represents shares of Class A Common Stock to be held by Mercury Houston Partners, LLC, Mercury Affiliates XI, LLC, and Mercury
Sponsor Group I LLC. Pursuant to the Voting and Non-Redemption Agreements, Mercury Houston Partners, LLC agreed not to redeem all of its 130,023 shares of Class A Common Stock, and Mercury Affiliates XI, LLC agreed not to redeem 491,489
shares out of its 664,977 shares of Class A Common Stock.
|
|
(8)
|
Represents shares of Class A Common Stock to be held by Mercury Sponsor Group I LLC.
|
|
(9)
|
Mr. James Besser is the managing member of Manchester Management PR, LLC (“Manchester”) and Manchester Management Company, LLC
(“GP”) and Morgan Frank serves as a portfolio manager and as a consultant for Manchester Explorer, L.P. (“Explorer”). Manchester is the investment manager of Explorer and GP is the general partner of Explorer. The parties together are
expected to own 1,263,661 shares in the Combined Company. The principal business address for each of Manchester, GP, Explorer and Messrs. Besser and Frank is 2 Calle Candina, #1701, San Juan, Puerto Rico, 00907.
|
|
(10)
|
Includes shares owned directly by Prides Capital Fund I, L.P. Prides Capital Partners LLC is the general partner of Prides
Capital Fund I, L.P. and Mr. Richardson is the controlling shareholder of Prides Capital Partners LLC. Mr. Richardson has also been deputized by Prides Capital Partners LLC to serve on the board of directors of the Company.
|
|
•
|
3,719,634 shares of SEPA’s Class A Common Stock (including those shares held as a constituent part of SEPA’s Units); and
|
|
•
|
2,033,150 shares of Class B Common Stock.
|
|
•
|
in whole and not in part;
|
|
•
|
at a price of $0.01 per warrant;
|
|
•
|
upon not less than 30 days’ prior written notice of redemption (the “30-day redemption period”) to each SEPA Public Warrant
holder; and
|
|
•
|
if, and only if, the last reported sale price of SEPA’s Class A Common Stock for any 20 trading days within a 30-trading day
period ending on the third trading day prior to the date on which SEPA sends the notice of redemption to the SEPA Public Warrant Holders (the “Reference Value”) equals or exceeds $18.00 per share (as adjusted for adjustments to the
number of shares issuable upon exercise or the exercise price of a warrant).
|
|
•
|
in whole and not in part;
|
|
•
|
at a price equal to a number of shares of Class A Common Stock to be determined by reference to the table below, based on the
redemption date and the “fair market value” of SEPA’s Class A Common Stock (as defined below) except as otherwise described below;
|
|
•
|
upon a minimum of 30 days’ prior written notice of redemption;
|
|
•
|
if, and only if, the Reference Value equals or exceeds $10.00 per share (as adjusted for adjustments to the number of shares
issuable upon exercise or the exercise price of a warrant) on the trading day prior to the date on which SEPA sends the notice of redemption to the warrant holders; and
|
|
•
|
if, and only if, the Reference Value is less than $18.00 per share (as adjusted for adjustments to the number of shares
issuable upon exercise or the exercise price of a warrant), the SEPA Private Placement Warrants must also concurrently be called for redemption on the same terms as the outstanding SEPA Public Warrants, as described above.
|
|
Redemption Date
(period to expiration of warrants)
|
| |
Fair Market Value of SEPA’s Class A Common Stock
|
||||||||||||||||||||||||
| |
$10.00
|
| |
$11.00
|
| |
$12.00
|
| |
$13.00
|
| |
$14.00
|
| |
$15.00
|
| |
$16.00
|
| |
$17.00
|
| |
$18.00
|
||
|
60 months
|
| |
0.261
|
| |
0.281
|
| |
0.297
|
| |
0.311
|
| |
0.324
|
| |
0.337
|
| |
0.348
|
| |
0.358
|
| |
0.361
|
|
57 months
|
| |
0.257
|
| |
0.277
|
| |
0.294
|
| |
0.310
|
| |
0.324
|
| |
0.337
|
| |
0.348
|
| |
0.358
|
| |
0.361
|
|
54 months
|
| |
0.252
|
| |
0.272
|
| |
0.291
|
| |
0.307
|
| |
0.322
|
| |
0.335
|
| |
0.347
|
| |
0.357
|
| |
0.361
|
|
51 months
|
| |
0.246
|
| |
0.268
|
| |
0.287
|
| |
0.304
|
| |
0.320
|
| |
0.333
|
| |
0.346
|
| |
0.357
|
| |
0.361
|
|
48 months
|
| |
0.241
|
| |
0.263
|
| |
0.283
|
| |
0.301
|
| |
0.317
|
| |
0.332
|
| |
0.344
|
| |
0.356
|
| |
0.361
|
|
45 months
|
| |
0.235
|
| |
0.258
|
| |
0.279
|
| |
0.298
|
| |
0.315
|
| |
0.330
|
| |
0.343
|
| |
0.356
|
| |
0.361
|
|
42 months
|
| |
0.228
|
| |
0.252
|
| |
0.274
|
| |
0.294
|
| |
0.312
|
| |
0.328
|
| |
0.342
|
| |
0.355
|
| |
0.361
|
|
39 months
|
| |
0.221
|
| |
0.246
|
| |
0.269
|
| |
0.290
|
| |
0.309
|
| |
0.325
|
| |
0.340
|
| |
0.354
|
| |
0.361
|
|
36 months
|
| |
0.213
|
| |
0.239
|
| |
0.263
|
| |
0.285
|
| |
0.305
|
| |
0.323
|
| |
0.339
|
| |
0.353
|
| |
0.361
|
|
33 months
|
| |
0.205
|
| |
0.232
|
| |
0.257
|
| |
0.280
|
| |
0.301
|
| |
0.320
|
| |
0.337
|
| |
0.352
|
| |
0.361
|
|
30 months
|
| |
0.196
|
| |
0.224
|
| |
0.250
|
| |
0.274
|
| |
0.297
|
| |
0.316
|
| |
0.335
|
| |
0.351
|
| |
0.361
|
|
27 months
|
| |
0.184
|
| |
0.214
|
| |
0.242
|
| |
0.268
|
| |
0.291
|
| |
0.313
|
| |
0.332
|
| |
0.350
|
| |
0.361
|
|
24 months
|
| |
0.173
|
| |
0.204
|
| |
0.233
|
| |
0.260
|
| |
0.285
|
| |
0.308
|
| |
0.329
|
| |
0.348
|
| |
0.361
|
|
21 months
|
| |
0.161
|
| |
0.193
|
| |
0.223
|
| |
0.252
|
| |
0.279
|
| |
0.304
|
| |
0.329
|
| |
0.347
|
| |
0.361
|
|
18 months
|
| |
0.146
|
| |
0.179
|
| |
0.211
|
| |
0.242
|
| |
0.271
|
| |
0.298
|
| |
0.322
|
| |
0.345
|
| |
0.361
|
|
15 months
|
| |
0.130
|
| |
0.164
|
| |
0.197
|
| |
0.230
|
| |
0.262
|
| |
0.291
|
| |
0.317
|
| |
0.342
|
| |
0.361
|
|
12 months
|
| |
0.111
|
| |
0.146
|
| |
0.181
|
| |
0.216
|
| |
0.250
|
| |
0.282
|
| |
0.312
|
| |
0.339
|
| |
0.361
|
|
9 months
|
| |
0.090
|
| |
0.125
|
| |
0.162
|
| |
0.199
|
| |
0.237
|
| |
0.272
|
| |
0.305
|
| |
0.336
|
| |
0.361
|
|
6 months
|
| |
0.065
|
| |
0.099
|
| |
0.137
|
| |
0.178
|
| |
0.219
|
| |
0.259
|
| |
0.296
|
| |
0.331
|
| |
0.361
|
|
3 months
|
| |
0.034
|
| |
0.065
|
| |
0.104
|
| |
0.150
|
| |
0.197
|
| |
0.243
|
| |
0.286
|
| |
0.326
|
| |
0.361
|
|
0 months
|
| |
—
|
| |
—
|
| |
0.042
|
| |
0.115
|
| |
0.176
|
| |
0.233
|
| |
0.281
|
| |
0.323
|
| |
0.361
|
|
•
|
If SEPA is unable to complete its initial business combination within 36 months from July 30, 2021, SEPA will (i) cease all
operations except for the purpose of winding up, (ii) as promptly as reasonably possible
|
|
•
|
Prior to SEPA’s initial business combination, SEPA may not issue additional shares of capital stock that would entitle the
holders thereof to (i) receive funds from the Trust Account or (ii) vote on any initial business combination;
|
|
•
|
Although SEPA does not intend to enter into a business combination with a target business that is affiliated with SEPA’s
Sponsor, SEPA’s directors or SEPA’s officers, SEPA is not prohibited from doing so. In the event SEPA enters into such a transaction, SEPA, or a committee of independent directors, will obtain an opinion from an independent investment
banking firm that is a member of FINRA or an independent accounting firm that such a business combination is fair to SEPA’s company from a financial point of view;
|
|
•
|
If a SEPA Stockholder vote on SEPA’s initial business combination is not required by law and SEPA does not decide to hold a
SEPA Stockholder vote for business or other legal reasons, SEPA will offer to redeem SEPA’s Public Shares pursuant to Rule 13e-4 and Regulation 14E of the Exchange Act, and will file tender offer documents with the SEC prior to
completing SEPA’s initial business combination which contain substantially the same financial and other information about SEPA’s initial business combination and the Redemption Rights as is required under Regulation 14A of the Exchange
Act;
|
|
•
|
SEPA’s initial business combination must occur with one or more target businesses that together have an aggregate fair market
value of at least 80% of SEPA’s net assets held in the Trust Account (net of amounts disbursed to management for working capital purposes and excluding the deferred underwriting commissions held in the Trust Account) at the time of the
agreement to enter into the initial business combination;
|
|
•
|
If SEPA Stockholders approve an amendment to SEPA’s Current Charter (A) to modify the substance or timing of SEPA’s
obligation to allow redemption in connection with SEPA’s initial business combination or redeem 100% of SEPA’s Public Shares if SEPA does not complete SEPA’s initial business combination within 18 months, or 24 months if SEPA has signed
a definitive agreement with respect to an initial business combination within such 18-month period (or up to 24 months if SEPA extends the period of time to consummate a business combination) from July 30, 2021 or (B) with respect to
any other provision relating to SEPA Stockholders’ rights or pre-initial business combination activity, SEPA will provide public SEPA Stockholders with the opportunity to redeem all or a portion of their shares of Class A Common Stock
upon such approval at a per-share price, payable in cash, equal to the aggregate amount then on deposit in the Trust Account, including interest earned on the funds held in the Trust Account and not previously released to SEPA to pay
SEPA’s franchise and income taxes as well as expenses relating to the administration of the Trust Account, divided by the number of then issued and outstanding Public Shares; and
|
|
•
|
SEPA will not complete SEPA’s initial business combination with another blank check company or a similar company with nominal
operations.
|
|
•
|
a stockholder who owns 15% or more of SEPA’s outstanding voting stock (otherwise known as an “interested stockholder”);
|
|
•
|
an affiliate of an interested stockholder; or
|
|
•
|
an associate of an interested stockholder, for three years following the date that the stockholder became an interested
stockholder.
|
|
•
|
the SEPA Board approves the transaction that made the stockholder an “interested stockholder,” prior to the date of the
transaction;
|
|
•
|
after the completion of the transaction that resulted in the stockholder becoming an interested stockholder, that stockholder
owned at least 85% of SEPA’s voting stock outstanding at the time the transaction commenced, other than statutorily excluded shares of common stock; or
|
|
•
|
on or subsequent to the date of the transaction, the business combination is approved by the SEPA Board and authorized at a
meeting of SEPA Stockholders, and not by written consent, by an affirmative vote of at least two-thirds of the outstanding voting stock not owned by the interested stockholder.
|
|
•
|
prior to the time of the transaction, the board of directors of the corporation approved either the business combination or
the transaction which resulted in the stockholder becoming an interested stockholder;
|
|
•
|
upon completion of the transaction that resulted in the stockholder becoming an interested stockholder, the stockholder owned
at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, excluding for purposes of determining the number of shares outstanding (i) shares
|
|
•
|
on or subsequent to the time of the transaction, the business combination is approved by the board and authorized at an
annual meeting stockholders, and not by written consent, by the affirmative vote of at least 662∕3% of the outstanding voting stock which is not owned by the interested stockholder.
|
|
SEPA
|
| |
SANUWAVE
|
| |
Combined Company
|
|
Authorized Capital Stock
|
||||||
|
SEPA’s authorized shares consist of 171,000,000 shares of capital stock par
value of $0.0001, consisting of (a) 170,000,000 shares of common stock, including (i) 150,000,000 shares of Class A Common Stock and (ii) 20,000,000 shares of Class B Common Stock, and (b) 1,000,000 shares of preferred stock, par value
$0.0001 per share.
|
| |
SANUWAVE’s authorized shares consist of 2,505,000,000 shares of capital
stock par value of $0.001, consisting of (a) 2,500,000,000 shares of common stock and (b) 5,000,000 shares of preferred stock, including (i) 6,175 shares of Series A Preferred Stock, (ii) 293 shares of Series B Preferred Stock, (iii) 90
shares of Series C Preferred Stock, and (iv) 8 shares of Series D Preferred Stock.
|
| |
The Proposed Charter will authorize the Combined Company to issue
171,000,000 shares, consisting of (i) 170,000,000 shares of Class A Common Stock, par value $0.0001 per share, and (ii) 1,000,000 shares of preferred stock, par value $0.0001 per share.
|
|
|
||||||
|
SEPA
|
| |
SANUWAVE
|
| |
Combined Company
|
|
Conversion
|
||||||
|
Class B Common Stock.
Shares of Class B Common Stock shall be convertible into shares of Class A Common Stock on a 1:0.277 basis, such that 1 share of Class B Common Stock is convertible to 0.277 shares of Class A Common Stock (as amended by the Class B
Charter Amendment), and shall be convertible (A) at any time and from time to time at the option of the holder thereof and (B) automatically on the closing of the Business Combination.
Common Stock. In the
event that a Business Combination is consummated by SEPA or SEPA holds a vote of its stockholders to amend its Certificate of Incorporation prior to the consummation of a Business Combination, any Stockholder who (i) followed the
procedures contained in the proxy materials to perfect the holder’s right to convert the holder’s Common Stock into cash, if any, or (ii) tendered the holder’s Common Stock as specified in the tender offer materials therefore, shall
be entitled to receive cash for the holder’s Common Stock.
SEPA shall, promptly
after the consummation of the Business Combination or the filing of the amendment to the Certificate of Incorporation with the Secretary of State of the State of Delaware, covert such shares into cash.
|
| |
Common Stock. The
holders of common stock do not have any conversion rights by virtue of their ownership of the common stock.
Series A Preferred Stock. Each share of preferred stock shall be convertible into that number of shares of common stock determined by dividing the stated value of $1,000.00 by the conversion price of $0.50 and shall be convertible at any time at the
option of the holder thereof.
Series B Preferred Stock. Each share of preferred stock shall be convertible into that number of shares of common stock determined by dividing the stated value of $1,000.00 by the conversion price of $0.50 and shall be convertible at any time at the
option of the holder thereof.
Series C Preferred Stock. Each share of preferred stock shall be convertible into that number of whole shares of common stock determined by dividing the stated value of $25,000.00 by the conversion price of $0.14 and shall be convertible at the
option of the holder thereof after the date that SANUWAVE shall have amended its charter to increase the number of shares of common stock authorized for issuance thereunder or effect a reverse stock split of the outstanding shares of
common stock by a sufficient amount to permit the conversion of all Series C Preferred Stock into shares of common stock.
Series D Preferred Stock. Each share of preferred stock shall be convertible into that number of whole shares of common stock determined by dividing the stated value of $25,000.00 by the conversion price of $0.14 and shall be convertible at the
option of the holder thereof after the date that
|
| |
There are no conversion provisions in the Proposed Charter or the SEPA
Bylaws.
|
|
SEPA
|
| |
SANUWAVE
|
| |
Combined Company
|
|
|
| |
SANUWAVE shall have amended its charter to increase the number of shares of
common stock authorized for issuance thereunder or effect a reverse stock split of the outstanding shares of common stock by a sufficient amount to permit the conversion of all Series D Preferred Stock into shares of common stock.
|
| |
|
|
|
||||||
|
Number and Qualification of Directors
|
||||||
|
The board of directors shall consist of one or more members. The number of
directors shall be fixed by the board of directors and may thereafter be changed from time to time by resolution of the board of directors.
|
| |
The entire board of directors shall not consist of less than one director or
more than nine directors. The number of directors constituting the entire board of directors shall be three until otherwise fixed by a majority of the entire board or directors.
|
| |
The number of directors which shall constitute the board of directors shall
be fixed exclusively by one or more resolutions adopted from time to time by the board of directors.
|
|
|
||||||
|
Structure of Board; Election of Directors
|
||||||
|
The Board is divided into three classes designated as Class I, Class II and
Class III, respectively. Each class consists, as nearly as may be possible, of one-third of the total number of directors constituting the whole board of directors. The bylaws provide that directors will be elected by a plurality of the
votes of the shares present in person or represented by proxy at the meeting and entitled to vote on the election of directors.
|
| |
The bylaws provide that directors will be elected at a meeting of the
stockholders and the directors to be elected at such meeting shall be elected by a plurality of votes given at such election.
|
| |
The Proposed Charter provides that, beginning with the Combined Company’s
first annual meeting of stockholders, the directors, including any of those elected by the holders of any series of Preferred Stock, shall be elected to hold office for a term expiring at the next annual meeting of the stockholders of
the Combined Company and until his or her successor shall be elected and qualified, or until his or her earlier death, resignation, retirement, disqualification or removal from office.
|
|
|
||||||
|
Removal of Directors
|
||||||
|
Any or all of the directors may be removed from office at any time, but only
for cause and only by the affirmative vote of holders of a majority of the voting power of all then outstanding shares of SEPA capital stock entitled to vote generally in the election of directors, voting together as a single class.
|
| |
Under the NRS, any director may be removed only by the vote of stockholders
representing not less than two-thirds (2∕3) of the voting power of the issued and outstanding stock entitled to vote.
|
| |
Subject to the special rights of the holders of one or more series of
preferred stock to elect directors, any director may be removed from office at any time by the affirmative vote of the holders of at least two-thirds (2∕3) of the voting power of all then
outstanding shares of capital stock of the Combined Company entitled to vote on the election of such director.
|
|
|
||||||
|
SEPA
|
| |
SANUWAVE
|
| |
Combined Company
|
|
Voting
|
||||||
|
Common Stock.
The holders of the Common Stock shall exclusively possess all voting power and each share of Common Stock shall have one vote.
Class A Common Stock. Unless provided under Delaware Law, the holders of Class A Common Stock do not hold separate voting rights from the holders of Class B Common Stock under the terms of the
Current Charter and the SEPA Bylaws.
Class B Common Stock. For so long as any shares of Class B Common Stock shall remain outstanding, SEPA shall not, without the prior vote or written consent of the holders of a majority of the shares of
Class B Common Stock then outstanding, voting separately as a single class, amend, alter or repeal any provision of the Current Charter, whether by merger, consolidation or otherwise, if
such amendment, alteration or repeal would alter or change the powers, preferences or relative, participating, optional or other or special rights of the Class B Common Stock.
|
| |
Each holder of shares of common stock is entitled to one vote per share
on all matters submitted to a vote of the common stockholders.
|
| |
The holders of the Class A Common Stock shall exclusively possess all
voting power and each share of Class A Common Stock shall have one vote.
|
|
|
||||||
|
Supermajority Voting Provisions
|
||||||
|
No amendment to Article IX of the Current Charter is effective prior to
the consummation of the initial business combination unless approved by the affirmative vote of the holders of at least 65% of all then outstanding shares of the Common Stock.
|
| |
The Bylaws of SANUWAVE may be adopted, amended, altered, changed,
rescinded or repealed in whole or in part at any annual or special meeting of the stockholders by the affirmative vote of two-thirds (2∕3) of the shares of
SANUWAVE outstanding and entitled to vote thereon.
Under the NRS, any director may be removed only by the vote of
stockholders representing not less than two-thirds (2∕3) of the voting power of the issued and outstanding stock entitled to vote.
|
| |
The Proposed Charter requires the approval by an affirmative vote of the
holders of at least two-thirds (2∕3) of the voting power of all then outstanding shares of capital stock of the Combined Company entitled to vote generally in
the election of directors to adopt, amend, alter or repeal the bylaws.
The Proposed Charter provides that directors may be removed from office
at any time, but only for cause and only by the affirmative vote of the holders of at least two-thirds (2∕3) of the voting power of the outstanding shares of
stock of the Combined Company entitled to vote on the election of such director.
|
|
SEPA
|
| |
SANUWAVE
|
| |
Combined Company
|
|
|
||||||
|
Vacancies on the Board of Directors
|
||||||
|
Subject to the rights, if any, of the holders of any outstanding series of
the Preferred Stock, newly created directorships resulting from an increase in the number of directors and any vacancies on the Board resulting from death, resignation, retirement, disqualification, removal or other cause may be filled
solely and exclusively by a majority vote of the remaining directors then in office, even if less than a quorum, or by a sole remaining director (and not by SEPA Stockholders), and any director so chosen shall hold office for the
remainder of the full term of the class of directors to which the new directorship was added or in which the vacancy occurred and until his or her successor has been elected and qualified, subject, however, to such director’s earlier
death, resignation, retirement, disqualification or removal.
|
| |
Any vacancy occurring in the board of directors for any cause other than by
reason of an increase in the number of directors may be filled by a majority of the remaining members of the board of directors , although such majority is less than a quorum, or by the SANUWAVE Stockholders. Any vacancy occurring by
reason of an increase in the number of directors may be filled by action of a majority of the entire board of directors or by the SANUWAVE Stockholders. A director elected by the board of directors to fill a vacancy shall be elected to
hold office until the expiration of the term for which they were elected and until his successor shall have been elected and shall have qualified.
|
| |
Subject to the rights, if any, of the holders of any outstanding series of
the Preferred Stock, newly created directorships resulting from an increase in the number of directors and any vacancies on the Board resulting from death, resignation, retirement, disqualification, removal or other cause may be filled
solely and exclusively by a majority vote of the remaining directors then in office, even if less than a quorum, or by a sole remaining director (and not by Combined Company stockholders), and any director so chosen shall hold office
for the remainder of the full term of the class of directors to which the new directorship was added or in which the vacancy occurred and until his or her successor has been elected and qualified, subject, however, to such director’s
earlier death, resignation, retirement, disqualification or removal.
|
|
|
||||||
|
Special Meeting of the Board of Directors
|
||||||
|
Subject to the rights, if any, of the holders of any outstanding series of
the preferred stock, and to the requirements of applicable law, special meetings of SEPA Stockholders may be called only by the Chairman of the Board, Chief Executive Officer, or the Board pursuant to a resolution adopted by a majority
of the Board, and the ability of SEPA Stockholders to call a special meeting is specifically denied. Except as provided in the foregoing sentence, special meetings of SEPA Stockholders may not be called by another person or persons.
|
| |
Special meetings of the board of directors for any purpose or purposes shall
be called at any time by the Chairperson of the board, the Chief Executive Officer, the President or, if the Chief Executive Officer and the President are absent or unable or refuse to act, by any Vice President or by any three
directors.
|
| |
Subject to the rights, if any, of the holders of any outstanding series of
the preferred stock, and to the requirements of applicable law, special meetings of Combined Company stockholders may be called only by the Chairman of the Board, Chief Executive Officer, or the Board pursuant to a resolution adopted by
a majority of the Board, and the ability of Combined Company stockholders to call a special meeting is specifically denied. Except as provided in the foregoing sentence, special meetings of Combined Company stockholders may not be
called by another person or persons.
|
|
|
||||||
|
SEPA
|
| |
SANUWAVE
|
| |
Combined Company
|
|
Amendment to Certificate/Articles of Incorporation
|
||||||
|
SEPA reserves the right at any time and from time to time to amend,
alter, change or repeal any provision contained in the Current Charter (including any Preferred Stock Designation), and other provisions authorized by the laws of the State of Delaware at the time in force that may be added or
inserted, in the manner now or hereafter prescribed by the Current Charter and the DGCL.
|
| |
Under the NRS, SANUWAVE’s Articles of Incorporation can be amended upon
the approval of SANUWAVE’s board of directors and SANUWAVE Stockholders holding shares representing at least a majority of the voting power of SANUWAVE.
|
| |
The Combined Company reserves the right at any time and from time to time
to amend, alter, change or repeal any provision contained in the Proposed Charter (including any Preferred Stock Designation), and other provisions authorized by the laws of the State of Delaware at the time in force that may be added
or inserted, in the manner now or hereafter prescribed by the Proposed Charter and the DGCL.
|
|
|
||||||
|
Amendment of Bylaws
|
||||||
|
The Current Charter requires the approval by an affirmative vote of the
holders of a majority of the voting power of all then outstanding shares of capital stock of SEPA entitled to vote generally in the election of directors to adopt, amend, alter or repeal the bylaws.
|
| |
The bylaws provide that the board of directors reserves the power to
amend the bylaws. The bylaws may also be adopted, amended, altered, changed, rescinded or repealed in whole or in part at any annual or special meeting of the stockholders by the affirmative vote of two-thirds (2∕3) of the shares of SANUWAVE outstanding and entitled to vote thereon.
|
| |
The Proposed Charter requires the approval by an affirmative vote of the
holders of at least two-thirds (2∕3) of the voting power of all then outstanding shares of capital stock of the Combined Company entitled to vote generally in
the election of directors to adopt, amend, alter or repeal the bylaws.
|
|
|
||||||
|
Quorum
|
||||||
|
Board of Directors. The bylaws provide that a majority of the directors then in office shall constitute a quorum for the transaction of business unless a greater number is required by law, by the
Current Charter or the bylaws. If a quorum shall not be present at any meeting of directors, the directors present thereat may adjourn the meeting from time to time, without notice other than
announcement at the meeting, until a quorum shall be present.
Stockholders. The
presence, in person or by proxy, at a stockholders meeting of the holders of shares of outstanding capital stock of SEPA representing a majority of the voting power of all outstanding shares
of capital stock of SEPA entitled to vote at such meeting shall constitute a quorum for the transaction of business at such meeting, except that when specified business is to be
|
| |
Board of Directors. One-half of the authorized number of directors shall be necessary to constitute a quorum for the transaction of business. Every act or decision done or made by a majority of the
directors present at a meeting duly held at which a quorum is present shall be regarded as the act of the board of directors, unless a greater number be required by law or by SANUWAVE’s Articles
of Incorporation.
Stockholders.
The presence in person or by proxy of persons entitled to vote a majority of the votes entitled to be cast by each separate class or voting group specified in SANUWAVE’s Articles
of Incorporation, as the same may be amended or supplemented from time to time, at any meeting shall constitute a quorum for the transaction of business.
|
| |
Board of Directors. The bylaws provide that a majority of the directors then in office shall constitute a quorum for the transaction of business unless a greater number is required by law, by the
Proposed Charter or the bylaws. If a quorum shall not be present at any meeting of directors, the directors present thereat may adjourn the meeting from time to time, without notice other than
announcement at the meeting, until a quorum shall be present.
Stockholders.
The presence, in person or by proxy, at a stockholders meeting of the holders of shares of outstanding capital stock of the Combined Company representing a majority of the voting power of all
outstanding shares of capital stock of the Combined Company entitled to vote at such meeting shall constitute a quorum for the transaction of business at such meeting, except that
|
|
SEPA
|
| |
SANUWAVE
|
| |
Combined Company
|
|
voted on by a class or series of stock voting as a class, the holders of
shares representing a majority of the voting power of the outstanding shares of such class or series shall constitute a quorum of such class or series for the transaction of such business.
|
| |
|
| |
when specified business is to be voted on by a class or series of stock
voting as a class, the holders of shares representing a majority of the voting power of the outstanding shares of such class or series shall constitute a quorum of such class or series for the transaction of such business.
|
|
|
||||||
|
Stockholder Action by Written Consent
|
||||||
|
Except as may be otherwise provided for or fixed pursuant to the Current
Charter (including any Preferred Stock Designation) relating to the rights of the holders of any outstanding series of preferred stock, subsequent to the consummation of the IPO, any action required or permitted to be taken by SEPA
Stockholders must be effected by a duly called annual or special meeting of such SEPA Stockholders and may not be effected by written consent of the SEPA Stockholders other than with respect to the Class B Common Stock with respect to
which action may be taken by written consent.
|
| |
Pursuant to SANUWAVE’s Bylaws, except where otherwise required by Nevada
law, action without a meeting is permitted to be taken by the SANUWAVE Stockholders.
|
| |
No action may be taken by written consent of the stockholders of the
Combined Company.
|
|
|
||||||
|
Special Stockholder Meetings
|
||||||
|
Subject to the rights, if any, of the holders of any outstanding series
of the preferred stock, and to the requirements of applicable law, special meetings of SEPA Stockholders may be called only by the Chairman of the Board, Chief Executive Officer, or the Board pursuant to a resolution adopted by a
majority of the Board, and the ability of SEPA Stockholders to call a special meeting is hereby specifically denied. Except as provided in the foregoing sentence, special meetings of SEPA Stockholders may not be called by another
person or persons.
|
| |
Special meetings of the stockholders, for any purpose or purposes
whatsoever, may be called by the Chairman of the Board of Directors, the Chief Executive Officer, the President, or the Board of Directors. Special meetings of SANUWAVE Stockholders may only be called by any other person or persons as
required by applicable law.
|
| |
Subject to the rights, if any, of the holders of any outstanding series
of the preferred stock, and to the requirements of applicable law, special meetings of stockholders of the Combined Company may be called only by the Chairman of the Board, Chief Executive Officer, or the Board pursuant to a
resolution adopted by a majority of the Board, and the ability of Combined Company stockholders to call a special meeting is hereby specifically denied. Except as provided in the foregoing sentence, special meetings of stockholders of
the Combined Company may not be called by another person or persons.
|
|
|
||||||
|
Notice of Stockholder Meetings
|
||||||
|
Written notice of the meeting stating the place, day and hour of the
meeting and, in the case of a special
|
| |
Written notice of each annual meeting shall be given to each SANUWAWE
Stockholder entitled
|
| |
Written or printed notice of the meeting stating the place, day and hour
of the meeting and, in the case
|
|
SEPA
|
| |
SANUWAVE
|
| |
Combined Company
|
|
meeting, stating the purpose or purposes for which the meeting is called,
and in case of a meeting held by remote communication stating such means, shall be delivered not less than 10 nor more than 60 days before the date of the meeting, either by hand delivery, mail, or overnight delivery service, or if
prior consent has been given by a stockholder by electronic transmission to each stockholder of record entitled to vote at such meeting.
|
| |
to vote, either personally or by mail or other means of written
communication, charges prepaid, addressed to such stockholder at stockholder’s address appearing on the books of SANUWAVE or given by a SANUWAVE Stockholder to SANUWAVE for the purpose of notice. All such notices shall be sent to each
SANUWAVE Stockholder entitled thereto not less than 10 nor more than 60 days before each annual meeting.
|
| |
of a special meeting, stating the purpose or purposes for which the meeting
is called, and in the case of a meeting held by remote communication stating such means, shall be delivered not less than 10 nor more than 60 days before the date of the meeting, either by hand delivery, mail or overnight delivery
service, or if prior consent has been given by a stockholder, by electronic transmission to each stockholder of record entitled to vote at such meeting.
|
|
|
||||||
|
Stockholder Proposals (Other than Nomination of Persons for Election as
Directors)
|
||||||
|
In addition to any other applicable requirements, for business (other than
nominations) to be properly brought before an annual meeting by a SEPA Stockholder, such SEPA Stockholder must have given timely notice thereof in proper written form to the Secretary and such business must otherwise be a proper matter
for SEPA Stockholder action. A SEPA Stockholder’s notice to the Secretary with respect to such business, to be timely, must be received by the Secretary at the principal executive offices of SEPA not later than the close of business on
the 90th day nor earlier than the opening of business on the 120th day before the anniversary date of the immediately preceding annual meeting of SEPA Stockholders; provided, however, that in the event that the annual meeting is more
than 30 days before or more than 60 days after such anniversary date, notice by the SEPA Stockholder to be timely must be so delivered not earlier than the close of business on the 120th day before the meeting and not later than the
later of (x) the close of business on the 90th day before the meeting or (y) the close of business on the 10th day following the day on which public announcement of the date of the annual meeting is first made by SEPA.
|
| |
Stockholders must properly bring a proposal before any meeting of SANUWAVE
Stockholders by such SANUWAVE Stockholder giving timely notice thereof in writing to the Secretary of SANUWAVE by delivering such notice to the principal executive offices of SANUWAVE not less than 50 days nor more than 75 days prior to
the meeting; provided, however, that in the event less than 60 days’ notice or prior public disclosure of the date of the meeting is given or made to SANUWAVE Stockholders, notice by the SANUWAVE Stockholder to be timely must be so
received not later than the close of business on the tenth day following the day on which such notice of the date of the meeting was mailed or such public disclosure was made.
|
| |
In addition to any other applicable requirements, for business (other than
nominations) to be properly brought before an annual meeting by a Combined Company stockholder, such Combined Company stockholder must have given timely notice thereof in proper written form to the Secretary and such business must
otherwise be a proper matter for Combined Company stockholder action. A Combined Company stockholder’s notice to the Secretary with respect to such business, to be timely, must be received by the Secretary at the principal executive
offices of the Combined Company not later than the close of business on the 90th day nor earlier than the opening of business on the 120th day before the anniversary date of the immediately preceding annual meeting of stockholders;
provided, however, that in the event that the annual meeting is more than 30 days before or more than 60 days after such anniversary date, notice by the Combined Company stockholder to be timely must be so delivered not earlier than the
close of business on the 120th day before the meeting and not later than the later of (x) the close of business on the 90th day before the meeting or (y) the close of business on the 10th day following the day on which public
announcement of the date of the annual meeting is first made by the Combined Company.
|
|
|
||||||
|
SEPA
|
| |
SANUWAVE
|
| |
Combined Company
|
|
Stockholder Nominations of Persons for Election as Directors
|
||||||
|
Nominations of persons for election to the Board at any annual meeting of
SEPA Stockholders, or at any special meeting of SEPA Stockholders called for the purpose of electing directors as set forth in SEPA’s notice of such special meeting, may be made (i) by or at the direction of the Board or (ii) by any
SEPA Stockholder (x) who is a SEPA Stockholder of record entitled to vote in the election of directors on the date of the giving of the notice provided for in the SEPA Bylaws and on the record date for the determination of SEPA
Stockholders entitled to vote at such meeting and (y) who complies with the notice procedures set forth in the SEPA Bylaws.
|
| |
SANUWAVE’s Articles of Incorporation and Bylaws do not have specific
provisions addressing the nomination of persons for election as directors.
|
| |
Nominations of persons for election to the Board at any annual meeting of
Combined Company stockholders, or at any special meeting of Combined Company stockholders called for the purpose of electing directors as set forth in the Combined Company’s notice of such special meeting, may be made (i) by or at the
direction of the Board or (ii) by any Combined Company stockholder (x) who is a Combined Company stockholder of record entitled to vote in the election of directors on the date of the giving of the notice provided for in the Combined
Company’s Bylaws and on the record date for the determination of Combined Company stockholders entitled to vote at such meeting and (y) who complies with the notice procedures set forth in the Combined Company’s Bylaws.
|
|
|
||||||
|
Exclusive Forum
|
||||||
|
Unless SEPA consents in writing to the selection of an alternative forum,
the Court of Chancery of the State of Delaware shall to the fullest extent permitted by law be the sole and exclusive forum for any SEPA Stockholder (including a beneficial owner) to bring (i) any derivative action or proceeding
brought on behalf of SEPA, (ii) any action asserting a claim of breach of a fiduciary duty owed by any director, officer or other employee of SEPA to SEPA or SEPA Stockholders, (iii) any action asserting a claim against SEPA, its
directors, officers or employees arising pursuant to any provision of the DGCL or the Current Charter or (iv) any action asserting a claim against SEPA, its directors, officers or employees governed by the internal affairs doctrine.
|
| |
SANUWAVE’s Articles of Incorporation and Bylaws do not have exclusive
forum provisions.
|
| |
Unless the Combined Company consents in writing to the selection of an
alternative forum, the Court of Chancery of the State of Delaware shall to the fullest extent permitted by law be the sole and exclusive forum for any Combined Company stockholder (including a beneficial owner) to bring (i) any
derivative action or proceeding brought on behalf of the Combined Company, (ii) any action asserting a claim of breach of a fiduciary duty owed by any director, officer or other employee of the Combined Company to the Combined Company
or stockholders, (iii) any action asserting a claim against the Combined Company, its directors, officers or employees arising pursuant to any provision of the DGCL or the Proposed Charter or (iv) any action asserting a claim against
SEPA, its directors, officers or employees governed by the internal affairs doctrine. Subject to the preceding provisions, the federal
|
|
SEPA
|
| |
SANUWAVE
|
| |
Combined Company
|
|
|
| |
|
| |
district courts of the United States of America shall be the exclusive forum
for the resolution of any complaint asserting a cause of action arising under the Securities Act or the Exchange Act.
|
|
|
||||||
|
Limitation of Liability of Directors and Officers
|
||||||
|
A director of SEPA shall not be personally liable to SEPA or SEPA
Stockholders for monetary damages for breach of fiduciary duty as a director, except to the extent such exemption from liability or limitation thereof is not permitted under the DGCL as the same exists or may hereafter be amended.
Any repeal or modification of the foregoing paragraph by the SEPA
Stockholders shall not adversely affect any right or protection of a director of SEPA with respect to events occurring prior to the time of such repeal or modification.
|
| |
Under the NRS, a director or officer is not individually liable for damages
as a result of an act or failure to act in their capacity as a director or officer unless (a) the presumption of good faith is rebutted or (b) such officer or director breached their fiduciary duty and such breach involved intentional
misconduct, fraud, or violation of law.
|
| |
A director of the Combined Company shall not be personally liable to the
Combined Company or stockholders for monetary damages for breach of fiduciary duty as a director, except to the extent such exemption from liability or limitation thereof is not permitted under the DGCL as the same exists or may
hereafter be amended.
Any repeal or modification of the foregoing paragraph by the Combined
Company stockholders shall not adversely affect any right or protection of a director of the Combined Company with respect to events occurring prior to the time of such repeal or modification.
|
|
|
||||||
|
Indemnification of Directors, Officers, Employees and Agents
|
||||||
|
SEPA, to the fullest extent permitted by applicable law, shall indemnify all
persons whom it may indemnify pursuant thereto. Expenses (including attorney’s fees) incurred by an officer or director in defending any civil, criminal, administrative, or investigative action, suit or proceeding for which such officer
or director may be entitled to indemnification hereunder shall be paid by SEPA in advance of the final disposition of such action, suit or proceeding upon receipt of an undertaking by or on behalf of such director or officer to repay
such amount if it shall ultimately be determined that he is not entitled to be indemnified by SEPA as authorized hereby.
|
| |
SANUWAVE, to the fullest extent authorized by Nevada law, shall indemnify
directors, officers, employees or agents against all expense, liability and loss (including attorneys’ fees, judgments, fines, ERISA excise taxes or penalties and amounts paid or to be paid in settlement) reasonably incurred or suffered
by such person in connection therewith. The right to indemnification shall be a contract right and shall include the right to be paid by SANUWAVE the expenses incurred in defending any such proceeding in advance of its final
disposition: provided, however, that, if Nevada law requires, the payment of such expenses incurred by a director or officer in their capacity as such in advance of the final disposition of a proceeding, shall be made only upon delivery
to SANUWAVE of an undertaking, by or on behalf of such director or
|
| |
The Combined Company, to the fullest extent permitted by applicable law,
shall indemnify all persons whom it may indemnify pursuant thereto. Expenses (including attorney’s fees) incurred by an officer or director in defending any civil, criminal, administrative, or investigative action, suit or proceeding
for which such officer or director may be entitled to indemnification hereunder shall be paid by the Combined Company in advance of the final disposition of such action, suit or proceeding upon receipt of an undertaking by or on behalf
of such director or officer to repay such amount if it shall ultimately be determined that he is not entitled to be indemnified by the Combined Company as authorized hereby.
|
|
SEPA
|
| |
SANUWAVE
|
| |
Combined Company
|
|
|
| |
officer, to repay all amounts so advanced if it shall ultimately be
determined that such director or officer is not entitled to be indemnified.
|
| |
|
|
|
||||||
|
Dividends, Distributions and Stock Repurchases
|
||||||
|
Distributions. A
holder of Offering Shares shall be entitled to receive distributions only as provided by Section 9.2(a), Section 9.2(b), Section 9.2(d) or Section 9.7 of the Current Charter. In no other circumstances shall a holder of Offering Shares
have any right or interest of any kind in or to the Trust Account.
Dividends. Subject to
applicable law, the rights, if any, of the holders of any outstanding series of the preferred stock and the provisions of Article IX of the Current Charter, the holders of shares of Common Stock shall be entitled to receive such
dividends and other distributions (payable in cash, property or capital stock of SEPA) when, as and if declared thereon by the Board from time to time out of any assets or funds of SEPA legally available therefor and shall share
equally on a per share basis in such dividends and distributions.
|
| |
Distributions. During
such time as the preferred stock is outstanding, if SANUWAVE declares or makes any dividend or other distribution of its assets (or rights to acquire its assets) to holders of shares of SANUWAVE Common Stock, by way of return of
capital or otherwise at any time after the issuance of the preferred stock, then, in each such case, the holder shall be entitled to participate in such distribution to the same extent that the holder would have participated therein
if the holder had held the number of shares of SANUWAVE Common Stock acquirable upon complete conversion of such holder’s preferred stock
Dividends. Holders of
preferred stock shall be entitled to receive dividends on shares of preferred stock equal (on an as-if-converted-to-common-stock basis) to and in the same form as dividends (other than dividends in the form of SANUWAVE Common Stock)
actually paid on shares of SANUWAVE Common Stock when, as and if such dividends (other than dividends in the form of SANUWAVE Common Stock) are paid on shares of SANUWAVE Common Stock. Other than as set forth in the previous sentence,
no other dividends shall be paid on shares of preferred stock.
SANUWAVE’s Board of
Directors reserves to itself alone the power to declare dividends.
|
| |
Dividends. Subject to
applicable law, the rights, if any, of the holders of any outstanding series of the preferred stock, the holders of shares of Class A Common Stock shall be entitled to receive such dividends and other distributions (payable in cash,
property or capital stock of the Combined Company) when, as and if declared thereon by the Board from time to time out of any assets or funds of the Combined Company legally available therefor and shall share equally on a per share
basis in such dividends and distributions.
|
|
|
||||||
|
Liquidation
|
||||||
|
Subject to applicable law, the rights, if any, of the holders of any
outstanding series of the preferred stock and the provisions of
|
| |
Upon any liquidation, dissolution or winding-up of SANUWAVE, whether
voluntary or involuntary, after the satisfaction in full of the
|
| |
Subject to applicable law, the rights, if any, of the holders of any
outstanding series of the preferred stock, in the event of any voluntary
|
|
SEPA
|
| |
SANUWAVE
|
| |
Combined Company
|
|
Article IX of the Current Charter, in the event of any voluntary or
involuntary liquidation, dissolution or winding up of SEPA, after payment or provision for payment of the debts and other liabilities of SEPA, the holders of shares of SEPA Common Stock shall be entitled to receive all the remaining
assets of SEPA available for distribution to SEPA Stockholders, ratably in proportion to the number of shares of Class A Common Stock (on an as converted basis with respect to the Class B Common Stock) held by them.
|
| |
debts of SANUWAVE and the payment of any liquidation preference owed to
the holders of shares of capital stock of SANUWAVE ranking prior to the preferred stock upon liquidation, the holders of the preferred stock shall participate pari passu with the holders of SANUWAVE Common Stock (on an
as-if-converted-to-common-stock basis without regard to any limitation on the conversion of the preferred stock) in the net assets of SANUWAVE.
|
| |
or involuntary liquidation, dissolution or winding up of the Combined
Company, after payment or provision for payment of the debts and other liabilities of the Combined Company, the holders of shares of Class A Common Stock shall be entitled to receive all the remaining assets of the Combined Company
available for distribution to stockholders, ratably in proportion to the number of shares of Class A Common Stock held by them.
|
|
|
||||||
|
Choice of Law
|
||||||
|
If any action, the subject matter of which is within the scope of
Section 12.1 of the Current Charter, is filed in a court other than a court located within the State of Delaware (a “Foreign Action”) in the name of any SEPA Stockholder, such SEPA Stockholder shall be deemed to have consented to (i)
the personal jurisdiction of the state and federal courts located within the State of Delaware in connection with any action brought in any such court to enforce Section 12.1 of the Current Charter (an “FSC Enforcement Action”) and
(ii) having service of process made upon such SEPA Stockholder in any such FSC Enforcement Action by service upon such SEPA Stockholder’s counsel in the Foreign Action as agent for such SEPA Stockholder. Any person or entity
purchasing or otherwise acquiring any interest in any security of SEPA shall be deemed to have notice of and consented to Article XII of the Current Charter.
|
| |
There are no choice of law provisions in SANUWAVE’s Articles of
Incorporation or Bylaws.
|
| |
If any action, the subject matter of which is within the scope of
Section 10.1 of the Proposed Charter, is filed in a court other than a court located within the State of Delaware (a “Foreign Action”) in the name of any Combined Company stockholder, such Combined Company stockholder shall be deemed
to have consented to (i) the personal jurisdiction of the state and federal courts located within the State of Delaware in connection with any action brought in any such court to enforce Section 10.1 of the Proposed Charter (an “FSC
Enforcement Action”) and (ii) having service of process made upon such Combined Company stockholder in any such FSC Enforcement Action by service upon such Combined Company stockholder’s counsel in the Foreign Action as agent for such
Combined Company stockholder. Any person or entity purchasing or otherwise acquiring any interest in any security of the Combined Company shall be deemed to have notice of and consented to Article X of the Proposed Charter.
|
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | |
| | | ||
| | | ||
| | | ||
| | | ||
| | |
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | |
| | | ||
| | | ||
| | | ||
| | | ||
| | |