Exhibit 99.2

CSE: PSYG | OTCQB: PSYGF Corporate Presentation

1 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Readers are therefore cautioned not to place undue reliance on such forward - looking statements . In addition, in considering any prior performance information contained in this presentation, readers should bear in mind that past results are not necessarily indicative of future results, and there can be no assurance that Psyence will achieve results comparable to those discussed in this presentation . This presentation speaks as of the date hereof and neither Psyence nor any affiliate or representative thereof assumes any obligation to provide any recipient of this presentation with subsequent revisions or updates to any historical or forward - looking information contained in this presentation to reflect the occurrence of events and/or changes in circumstances after the date hereof, except as may be required by law . Cautionary Statement Private & Confidential This presentation does not constitute an offer or invitation for the sale or purchase of securities and has been prepared solely for informational purposes. The information contained in this presentation (this “Presentation”) has been prepared for the exclusive use of the selected persons to whom it is addressed (“Recipients”), solely for the purpose of their own independent evaluation with respect to an investment (the “Proposed Investment”) in connection with the proposed business combination (the “Proposed Transaction”) between Newcourt Acquisition Corp (“Newcourt”) and a subsidiary of Psyence, and for no other purpose. This Presentation is subject to updating, completion, revision, verification and further amendment. None of Newcourt, Psyence, or their respective affiliates has authorized anyone to provide interested parties with additional or different information. No securities regulatory authority has expressed an opinion about the securities discussed in this Presentation and it is an offense to claim otherwise. The information contained herein does not purport to be all - inclusive. Nothing herein shall be deemed to constitute investment, legal, tax, financial, accounting or other advice. Neither this Presentation nor its delivery to Recipient shall constitute an offer to sell, invitation or other solicitation of an offer to buy any securities pursuant to the Proposed Investment or otherwise, nor shall there by any sale of securities in any jurisdiction in which the offer, solicitation or sale would be unlawful prior to the registration or qualification under the securities laws of any such jurisdiction. No offer shall be made except by means of a prospectus meeting the requirements of Section 10 of the Securities Act of 1933, as amended. Only the express provisions of any agreement, if and when it is executed, shall have any legal effect in connection with the Proposed Transaction between the parties thereto. This Presentation is not intended to form the basis of any investment decision. All information herein speaks only as of (1) the date of this Presentation, in the case of information about Psyence, or (2) the date of such information, in the case of information from persons other than Psyence. Additional Information about the Business Combination and Where to Find It A Registration Statement will be filed with the Securities and Exchange Commission (“SEC”), which will include a preliminary proxy statement / prospectus with respect to the Proposed Transaction. The definitive proxy statement / prospectus and other relevant documents will be mailed to shareholders of Newcourt as of a record date to be established for voting on the Business Combination. Shareholders of Newcourt and other interested persons are advised to read, when available, the preliminary proxy statement / prospectus, the definitive proxy statement/ prospectus and amendments thereto because these documents will contain important information about Newcourt, Psyence and the Proposed Transaction. These documents, once available, and Newcourt’s annual and other reports filed with the SEC can also be obtained, without charge, at the SEC’s internet site ( http://www.sec.gov ). Participants in the Solicitation Newcourt, Psyence and their respective directors and executive officers, other members of management and employees may be considered participants in the solicitation of proxies with respect to the Potential Transaction under the rules of the SEC. Information about the directors and executive officers of Newcourt is set forth in Newcourt’s filings with the SEC. Information regarding other persons who may, under the rules of the SEC, be deemed participants in the solicitation of the shareholders in connection with the potential transaction and a description of their interests will be set forth in the Registration Statement when it is filed with the SEC. These documents can be obtained free of charge from the sources indicated above.

2 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Executive Summary Licensed producer of natural psilocybin • Psyence Therapeutics is a wholly owned subsidiary of PSYENCE (CSE:PSYG) • Focussed on the development of natural psilocybin for the treatment of Adjustment disorder in palliative care • Strong Management with extensive Pharma drug development experience • Currently approved for Ph IIa clinical trials in UK • Key upcoming milestones: • Ph IIb clinical trial in Australia H1 2023, data read out Q1 2024 • Pre - IND FDA meeting in planning for Q1 2023 • EOP PhII FDA meeting , IND application for Phase III H1 2024 • Newcourt SPAC and Psyence Therapeutics have entered into a Business Combination agreement Jan 6 th 2023 • Resulting cash in the New Company will be $20MUSD: • Completion of Ph IIb • Open FDA IND • Initiate Ph III registrational studies Drug Development
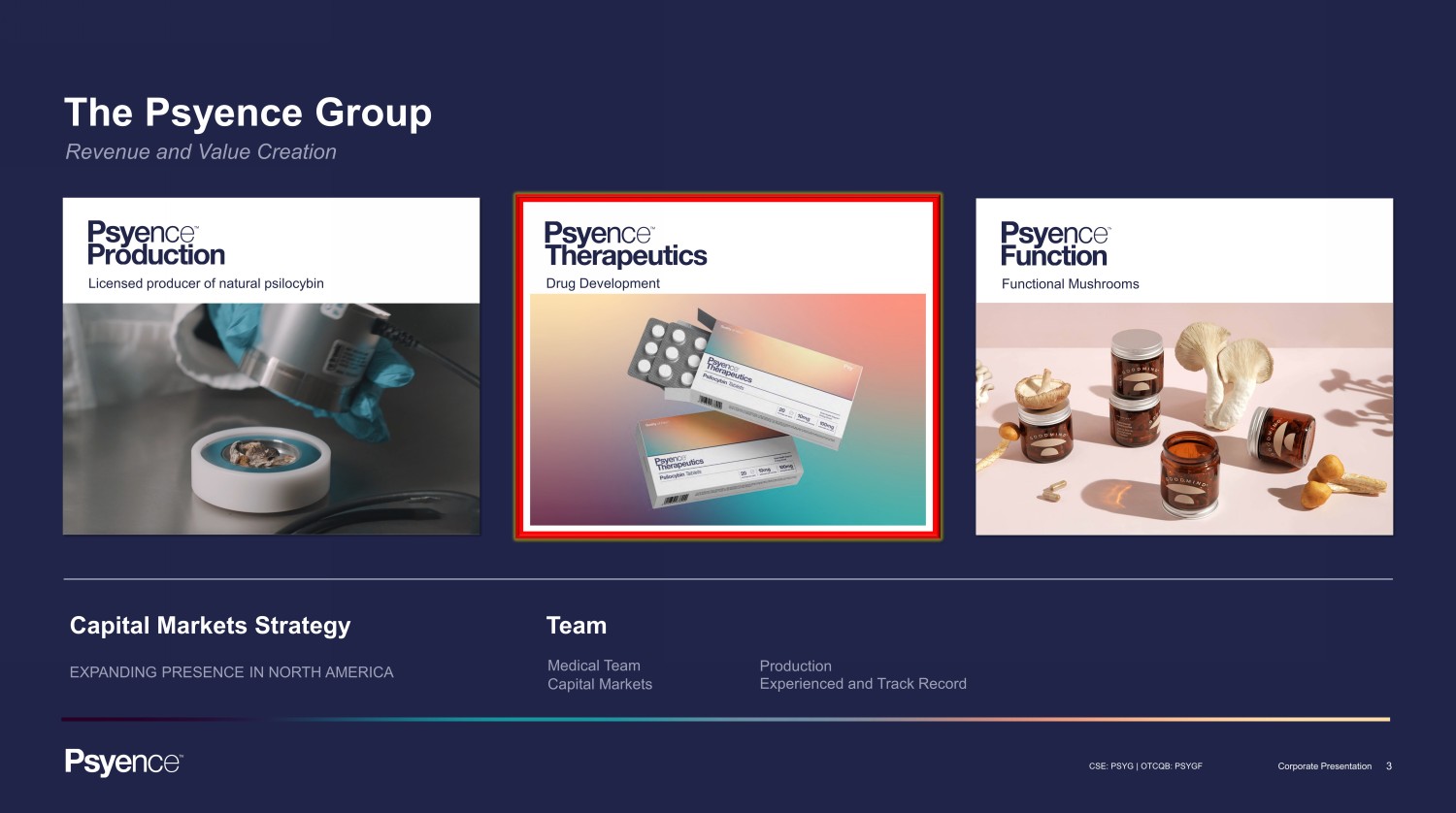
3 CSE: PSYG | OTCQB: PSYGF Corporate Presentation The Psyence Group Revenue and Value Creation Licensed producer of natural psilocybin Capital Markets Strategy EXPANDING PRESENCE IN NORTH AMERICA Team Functional Mushrooms Medical Team Capital Markets Production Experienced and Track Record Drug Development

4 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Acquisition Target Licensed producer of natural psilocybin Drug Development

5 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Xan Morgan DIRECTOR OF EUROPEAN OPERATIONS Successfully co - founded multinational businesses across water technology and electric sports cars, medical cannabis including holding Schedule 1 Controlled Drugs Licenses in UK. Tony Budden CHIEF STRATEGIC OFFICER Previous Director of Strategy: Canopy Growth Africa. Co - founded Hemporium, Africa’s oldest cannabis / hemp company. Recognised internationally as one of the top 100 Global Icons at the Cannabis Business Awards. Dr. Neil Maresky CEO Previous Vice President Scientific Affairs AstraZeneca Canada Inc. Trained Physician with over 25 years experience, leading Research and Development for Big Pharma, Responsible for the development and approval of over 30 New Medicines in Canada. Jody Aufrichtig CO - FOUNDER, PRESIDENT AND EXECUTIVE CHAIRMAN Multiple award - winning entrepreneur and business builder creating substantial shareholder value in multiple industries. Alan Friedman CAPITAL MARKET ADVISOR Capital Market Advisory, Bayline Capital Partners. Warwick Corden - Lloyd CHIEF FINANCIAL OFFICER Chartered Accountant and Certified Project Manager with over 17 years of experience in the UK, the USA, and South Africa. Taryn Vos GENERAL COUNSEL Corporate and legal attorney, previously Head of Legal for Canopy Growth Africa. Mary Elizabeth Gifford EVP PUBLIC AFFAIRS AND CSR Trusted voice for health justice, Chair of the Global Wellness Institute’s Psychedelics and Healing Initiative. Dr. Clive Ward - Able MEDICAL DIRECTOR Physician and pharmacist who has worked in the pharmaceutical industry for over 30 years. Experienced in international therapeutic drug commercialization and has been involved with the launch of over 16 products in multiple therapeutic areas. Experienced and complete Management Team in the Business of Medicine and Science and Capital Markets

6 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Dan Rogers CHIEF FIANNCIAL OFFICER • CEO of FintechForce, Inc. • Former CFO of Endpoint Clinical • Former CFO of Simplee • Former CFO of Fifth Third Processing Solutions • Former VP, Wells Fargo Merchant Services Marc Balkin CHIEF EXECUTIVE OFFICER • Partner, DiGame • Former Partner, Hasso Plattner Ventures / Capital Emerging Markets • Served on and chaired investment committees of Enablis, First National Bank Vumela Fund, Telkom FutureMakers, Alithea IDF Michael Jordaan CHAIRMAN • Former CEO of First National Bank, operating in 10 emerging markets in Asia and Africa • Co - Founder and Chairman of Bank Zero • Co - Founder of Rain Telco • Investment Committee Chair of SA SME Fund • Board Member at Several Disruptive Technology Companies Ryan Gilbert ADVISOR • Founder & General Partner, Launchpad Capital • Chairman of SmartBiz Loans, Director, River City Bank, Director, bKash • CEO of FTAC Zeus and FTAC Parnassus • Former CEO of FTAC Olympus, now merged with Payoneer Newcourt’s Management Team has extensive operating and investing experience across emerging markets
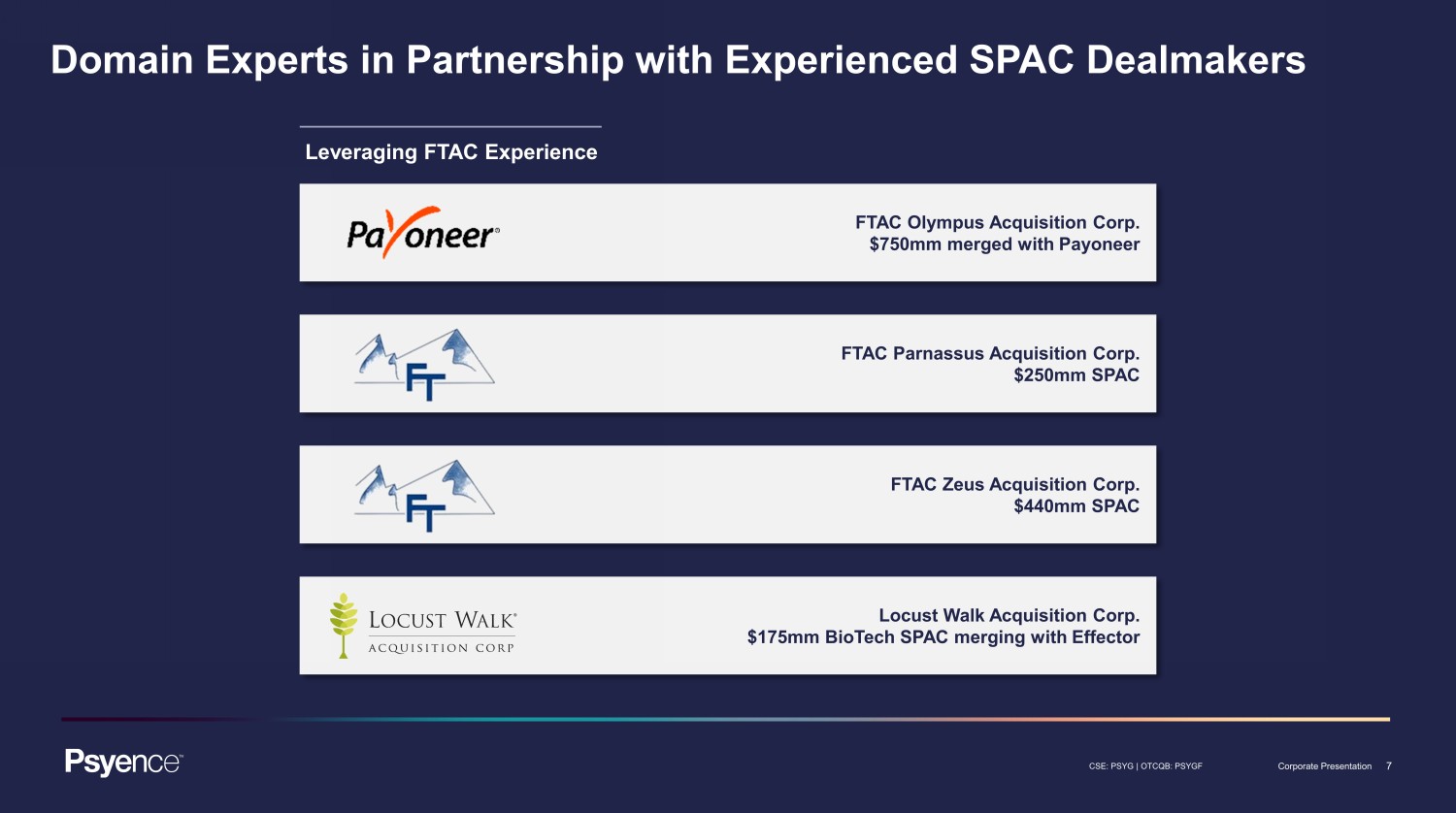
7 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Domain Experts in Partnership with Experienced SPAC Dealmakers Leveraging FTAC Experience FTAC Olympus Acquisition Corp. $750mm merged with Payoneer FTAC Parnassus Acquisition Corp. $250mm SPAC FTAC Zeus Acquisition Corp. $440mm SPAC Locust Walk Acquisition Corp. $175mm BioTech SPAC merging with Effector
8 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Psychedelics are making headlines: Market Disruptor
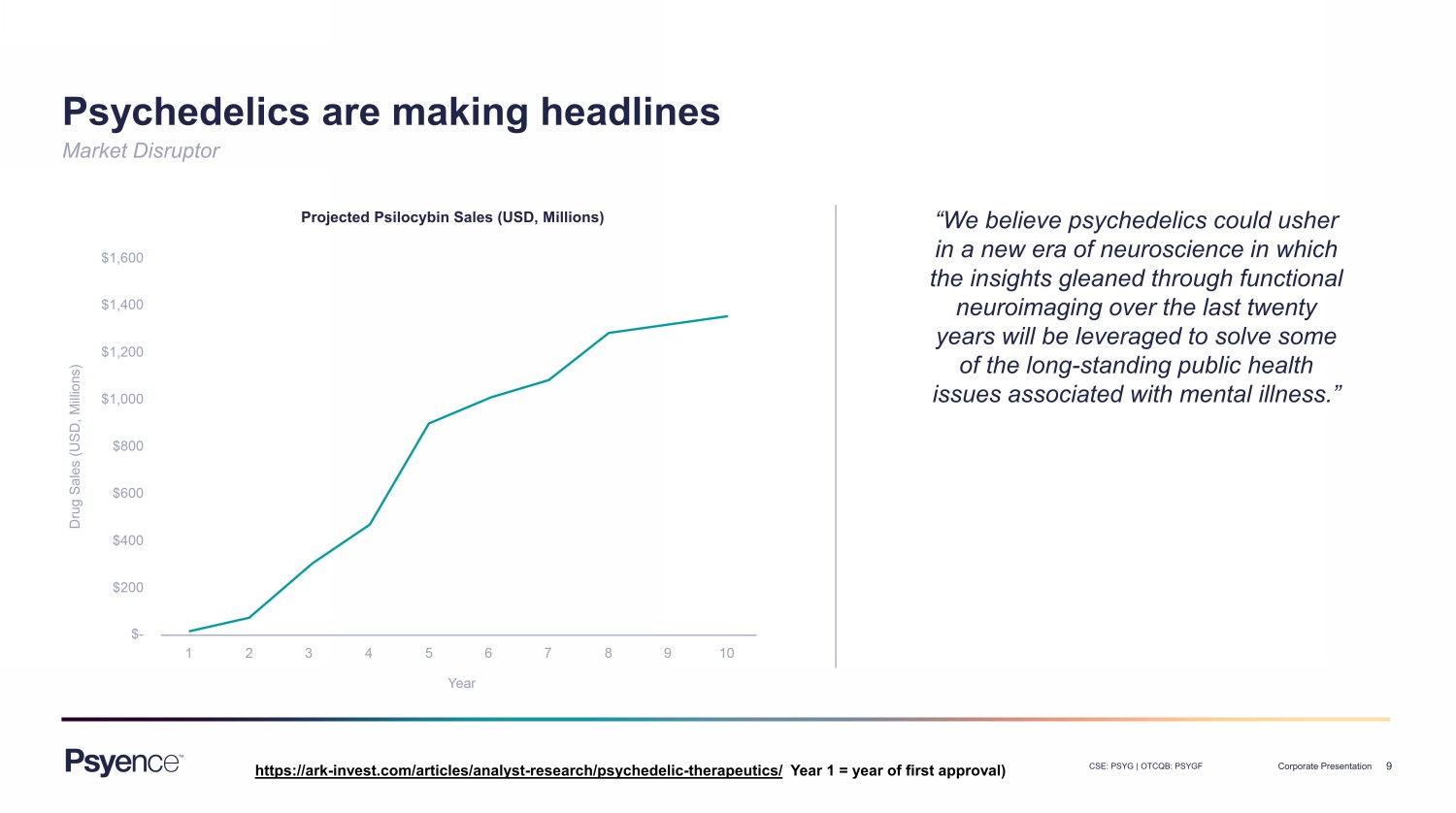
9 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Market Disruptor Psychedelics are making headlines “We believe psychedelics could usher in a new era of neuroscience in which the insights gleaned through functional neuroimaging over the last twenty years will be leveraged to solve some of the long - standing public health issues associated with mental illness.” Projected Psilocybin Sales (USD, Millions) https://ark - invest.com/articles/analyst - research/psychedelic - therapeutics/ Year 1 = year of first approval)
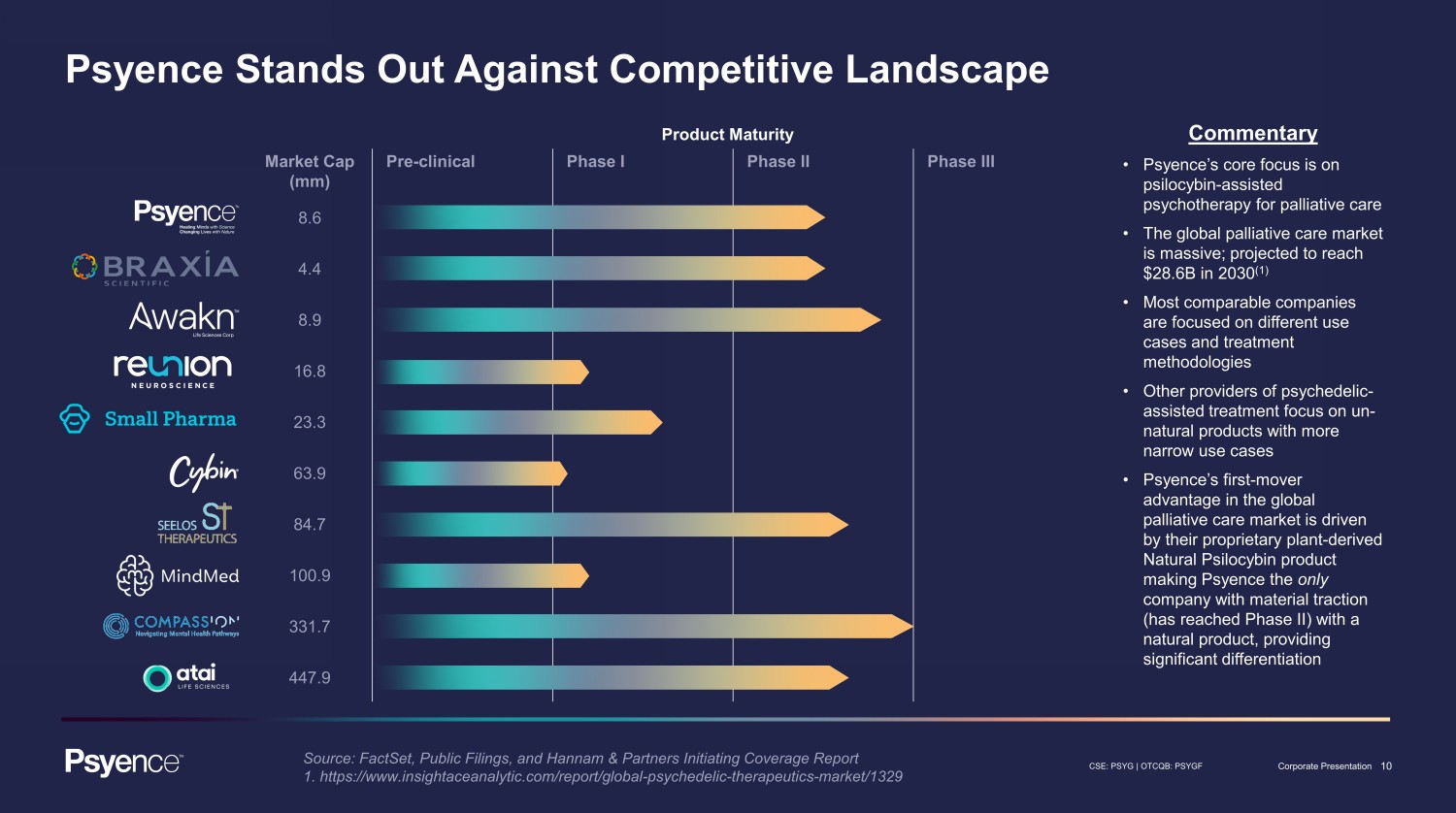
10 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Product Maturity Market Cap (mm) Pre - clinical Phase I Phase II Phase III 8.6 4.4 8.9 16.8 23.3 63.9 84.7 100.9 331.7 447.9 Commentary • Psyence’s core focus is on psilocybin - assisted psychotherapy for palliative care • The global palliative care market is massive; projected to reach $28.6B in 2030 (1) • Most comparable companies are focused on different use cases and treatment methodologies • Other providers of psychedelic - assisted treatment focus on un - natural products with more narrow use cases • Psyence’s first - mover advantage in the global palliative care market is driven by their proprietary plant - derived Natural Psilocybin product making Psyence the only company with material traction (has reached Phase II) with a natural product, providing significant differentiation Psyence Stands Out Against Competitive Landscape Source: FactSet, Public Filings, and Hannam & Partners Initiating Coverage Report 1. https://www.insightaceanalytic.com/report/global - psychedelic - therapeutics - market/1329

11 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Psyence (CSE:PSYG) is a life science biotechnology company pioneering the use of natural psychedelics in mental health and well - being. Our vision Psyence develops and will provide innovative, safe, and effective psychedelic and nature - centered solutions for mental health and palliative care. Our enduring work expands access to mental wellness and nurtures community. Psyence at a glance • First - mover advantage in global psychedelic medicine • Medical psilocybin production – Operational Facility & Laboratory • Clinical trials in process: MHRA (UK) • Pre - Clinical Program initiated in Canada • Listed on the CSE: PSYG and OTCQB: PSGYF • Strong path to revenue generation and value creation • Highly experienced medical and business team • Palliative Care

Developing proprietary natural psilocybin drug development and treatment protocols with a focus on palliative care
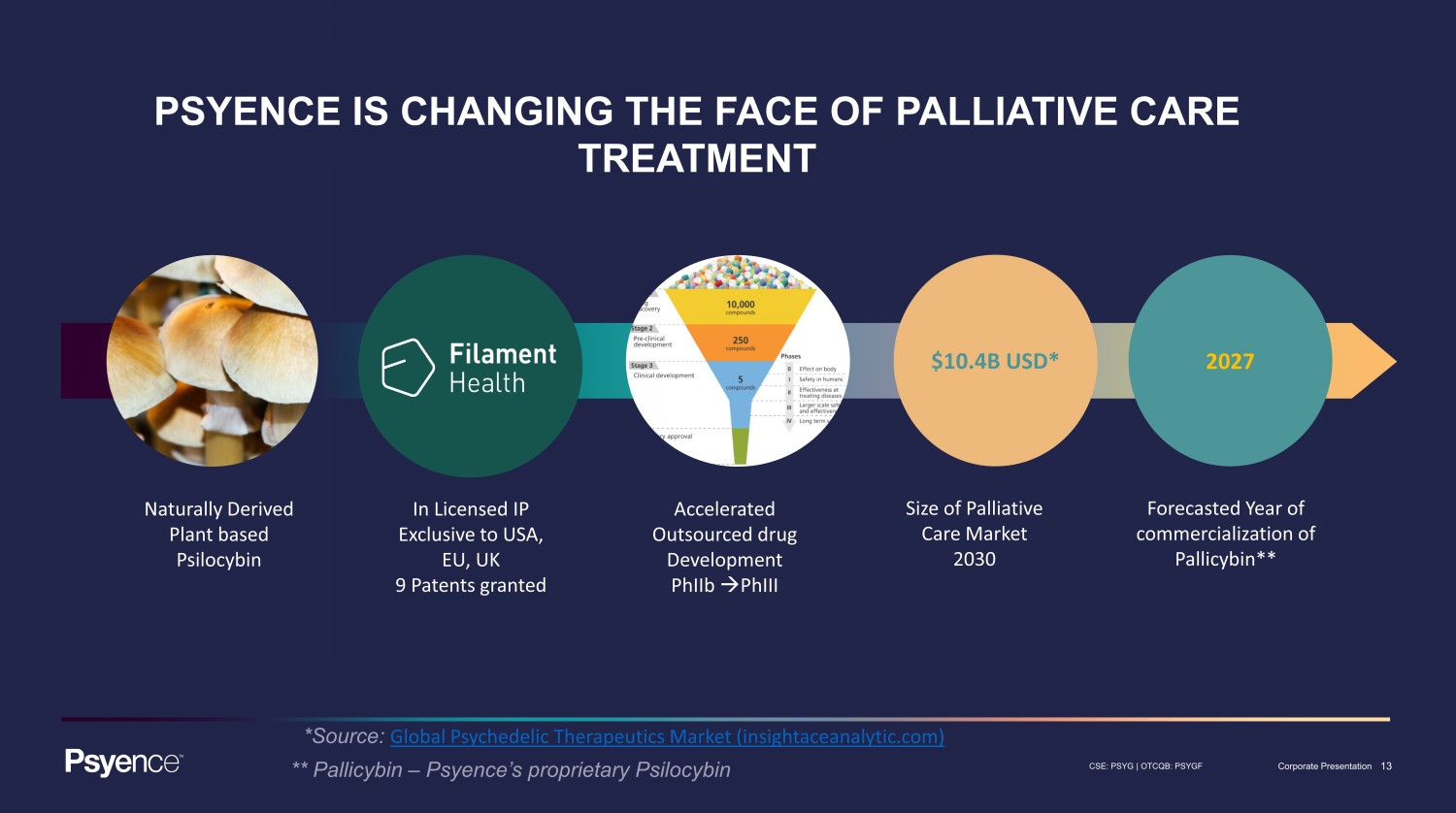
13 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Naturally Derived Plant based Psilocybin In Licensed IP Exclusive to USA, EU, UK 9 Patents granted Accelerated Outsourced drug Development PhIIb PhIII Size of Palliative Care Market 2030 Forecasted Year of commercialization of Pallicybin** $10.4B USD* 2027 PSYENCE IS CHANGING THE FACE OF PALLIATIVE CARE TREATMENT *Source: Global Psychedelic Therapeutics Market (insightaceanalytic.com) ** Pallicybin – Psyence’s proprietary Psilocybin

CSE: PSYG | OTCQB: PSYGF Corporate Presentation The Palliative Care (PC) Market Palliative care refers to care for patients and their families who are facing a serious, life - limiting illness. Palliative care is patient - centred coordinated care that aims to relieve suffering and improve quality of life for patients and their families at all stages of the illness. Psyence will focus on the psychological distress of palliative care, an important but often ignored and/or poorly treated area. https://health.gov.on.ca/en/public/programs/palliative/palliative_questionsandanswers.aspx
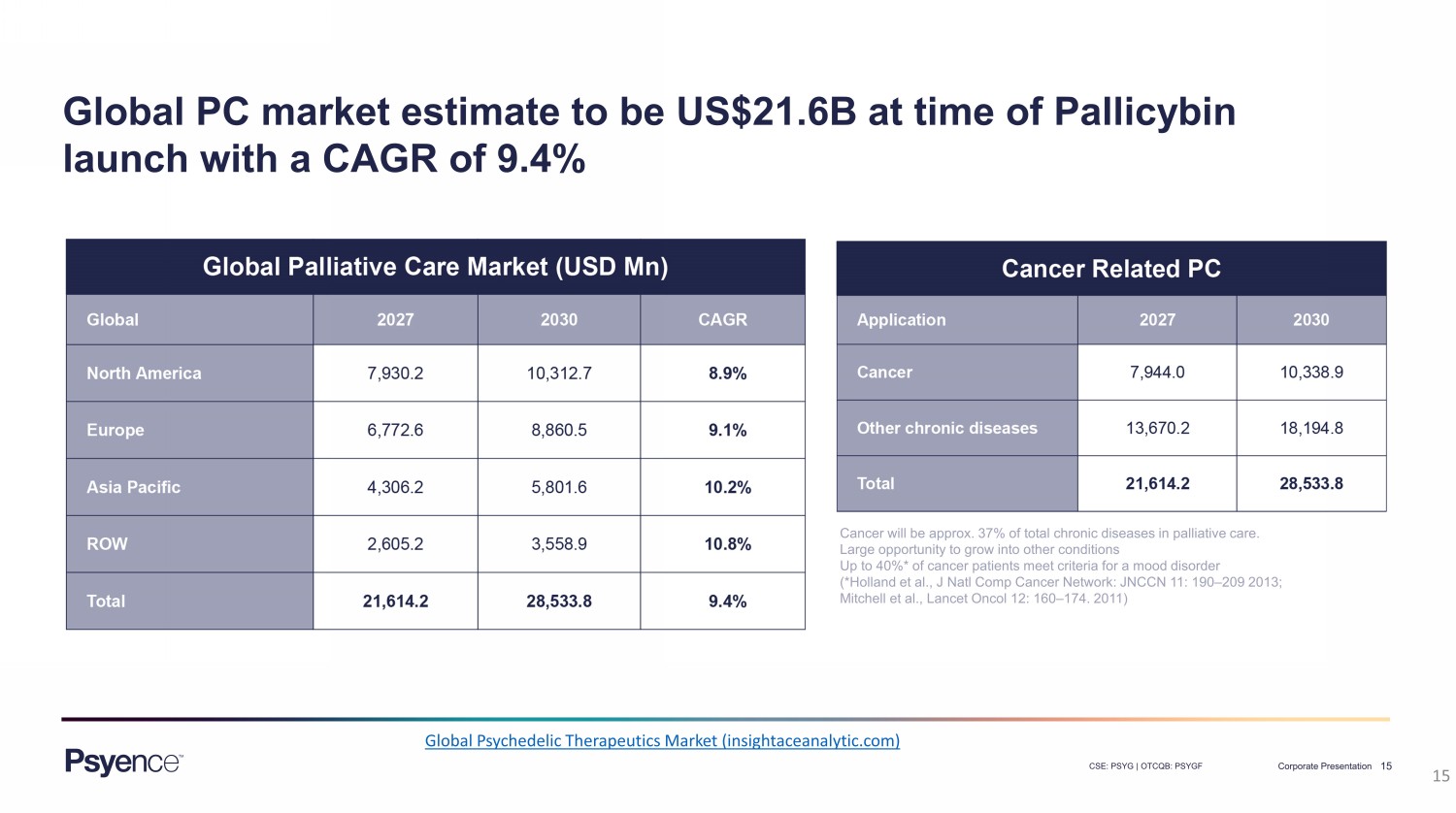
15 CSE: PSYG | OTCQB: PSYGF Corporate Presentation 15 Cancer will be approx. 37% of total chronic diseases in palliative care. Large opportunity to grow into other conditions Up to 40%* of cancer patients meet criteria for a mood disorder (*Holland et al., J Natl Comp Cancer Network: JNCCN 11: 190 – 209 2013; Mitchell et al., Lancet Oncol 12: 160 – 174 . 2011) Global Psychedelic Therapeutics Market (insightaceanalytic.com) Global PC market estimate to be US$21.6B at time of Pallicybin launch with a CAGR of 9.4%
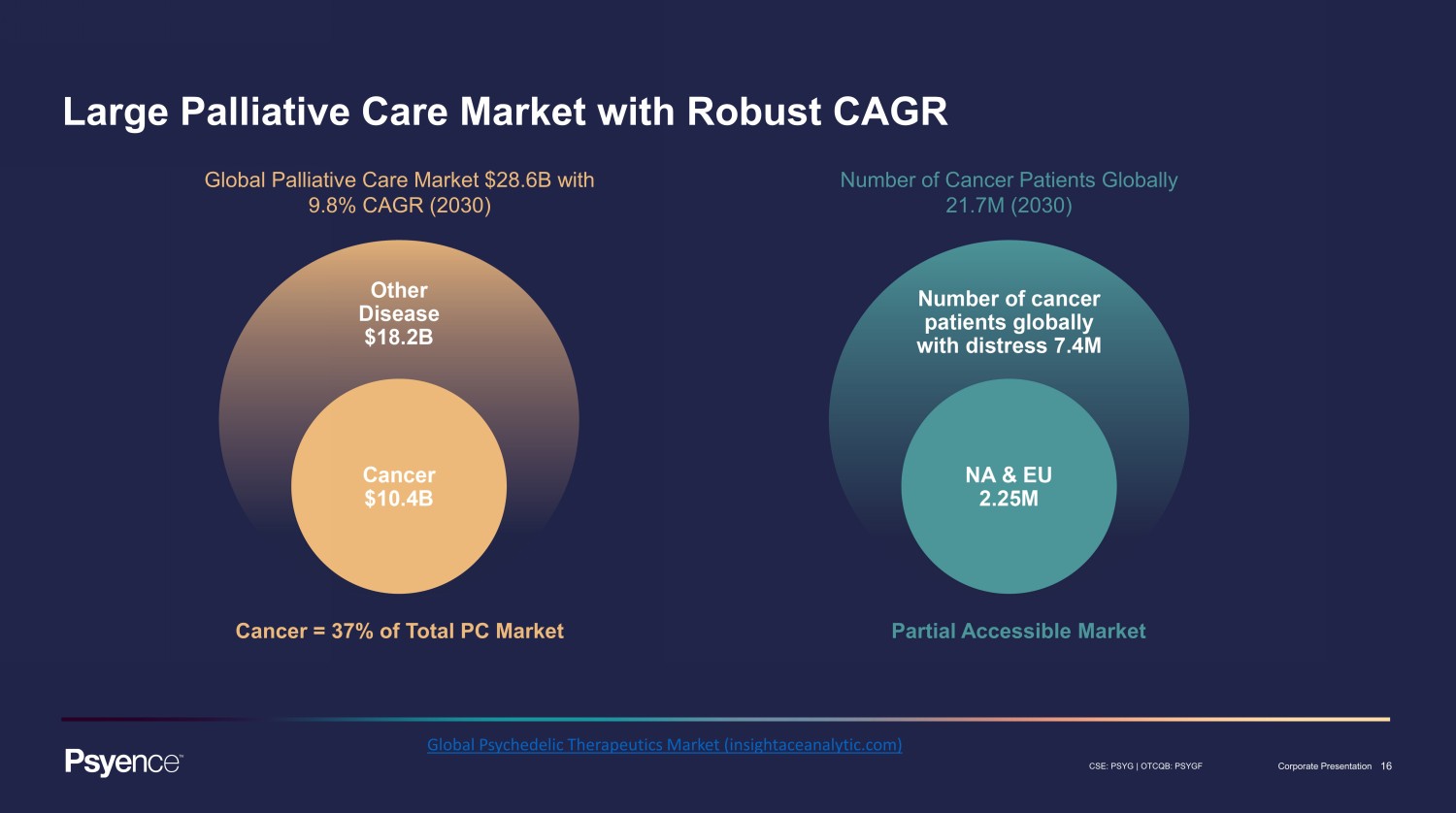
16 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Large Palliative Care Market with Robust CAGR Cancer = 37% of Total PC Market Partial Accessible Market Global Palliative Care Market $28.6B with 9.8% CAGR (2030) Number of Cancer Patients Globally 21.7M (2030) Global Psychedelic Therapeutics Market (insightaceanalytic.com)

17 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Large Palliative Care Market with Robust CAGR Market Share % PAP Pricing ** Psilocybin Cost only $ 6,000.00 $ 500.00 1% $135m $11.25m 2% $270m $22.5m 5% $675m $56.25m 10% $1.35 B $112.5m Estimated Total Addressable Market Global Palliative Care Market $28.6B with 9.8% CAGR (2030) Number of Cancer Patients Globally 21.7M (2030) Number of Cancer patients in North America & EU with Distress (2030) - 2.25M Market Size * 3 times larger including other palliative care patients • Performed by Insight Ace Analytic (insightaceanalytic.com) : September 2022 ** Psychedelic assisted psychotherapy * Global Psychedelic Therapeutics Market (insightaceanalytic.com) ** Psychedelic Assisted Psychotherapy

Palliative care reduces emergency room visits and total hospital days among patients with metastatic HPB and GI cancers | Scientific Reports (nature.com) Potential Health Economic Downstream Benefits

19 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Target Indication with Palliative Care Population • Pallicybin (Psilocybin) is indicated for the relief of symptoms of Adjustment Disorder caused by the diagnosis of a terminal illness, or • Pallicybin in conjunction with psychotherapy program is indicated for the treatment of Adjustment Disorder caused by the diagnosis of a terminal illness.

20 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Adjustment Disorder Adjustment Disorder falls into the wide categorical spectrum of anxiety and depressive disorders. The DSM - 5 - TR criteria describes adjustment disorder as a development of emotional or behavioural symptoms in response to an identifiable stressor within three months of onset. The type of stressor may vary in significance for children and adolescents versus adults. These stressors can include but are not limited to death, medical diagnoses, marital discord, unexpected life events, finances, sexuality discoveries, family problems, and the list goes on. The difference between adjustment disorder and other mental disorders lies in the impact it takes on one’s daily activities, with a true associated stressor for the cause. Symptoms Patients experiencing adjustment disorder may exhibit one or a combination of the following symptoms following an identifiable life event three months prior: • Depressed mood behavior such as dysthymia, tearfulness, hopelessness • Anxiety mood behavior such as excessive worrying, nervousness, uneasiness • Disturbance of conduct. For example, if a child is exhibiting this symptom, they may begin to misbehave in school or become aggressive • Unspecified due to maladaptive reactions * Adjustment disorder is a serious condition affecting 40% of patients with a terminal diagnosis https://pubmed.ncbi.nlm.nih.gov/36510761/ * Serious, Chronic, or Terminal Illnesses - Tips for (adaa.org)
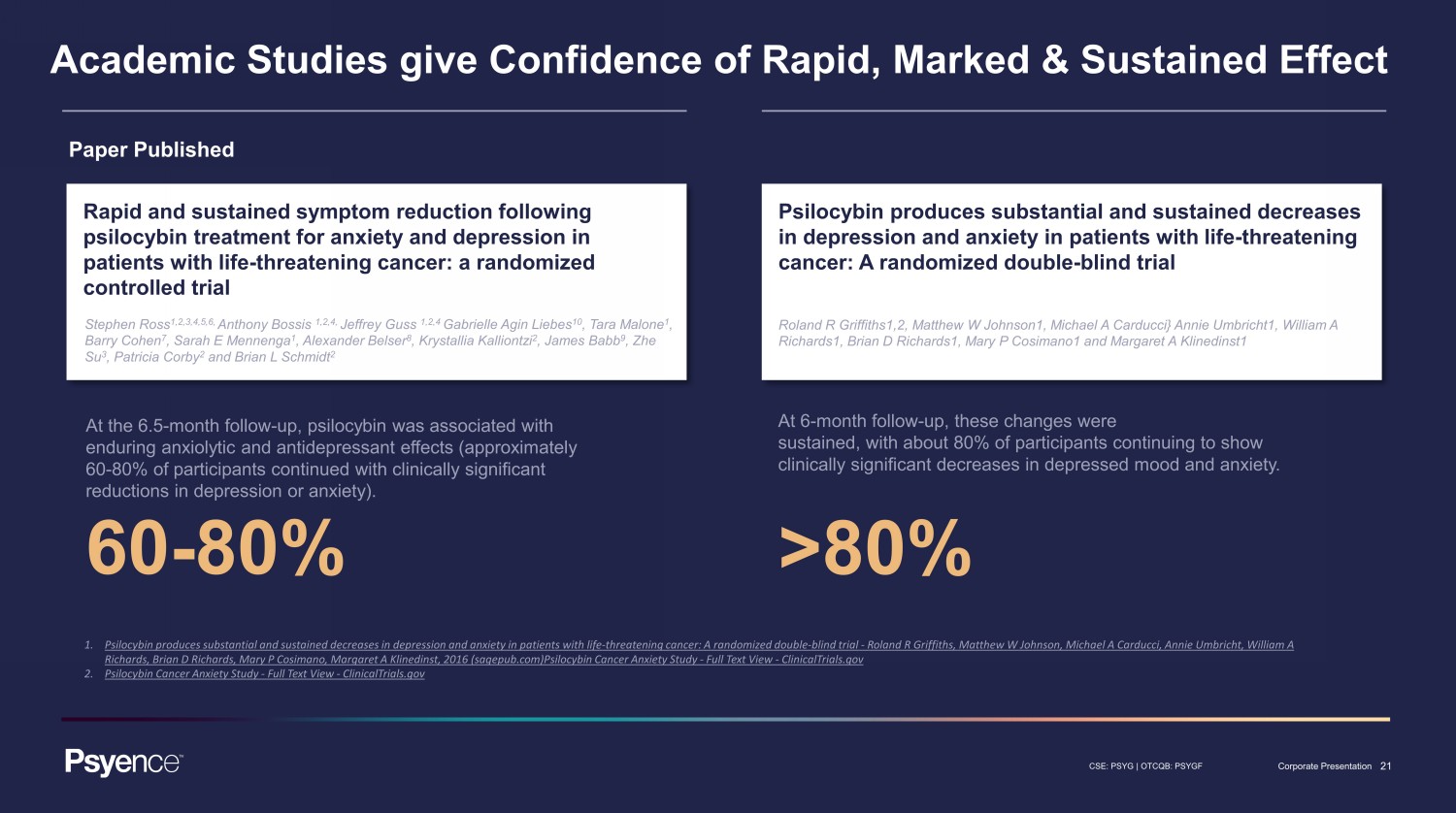
21 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Academic Studies give Confidence of Rapid, Marked & Sustained Effect Paper Published Rapid and sustained symptom reduction following psilocybin treatment for anxiety and depression in patients with life - threatening cancer: a randomized controlled trial Stephen Ross 1,2,3,4,5,6, Anthony Bossis 1,2,4, Jeffrey Guss 1,2,4 Gabrielle Agin Liebes 10 , Tara Malone 1 , Barry Cohen 7 , Sarah E Mennenga 1 , Alexander Belser 8 , Krystallia Kalliontzi 2 , James Babb 9 , Zhe Su 3 , Patricia Corby 2 and Brian L Schmidt 2 At the 6.5 - month follow - up, psilocybin was associated with enduring anxiolytic and antidepressant effects (approximately 60 - 80% of participants continued with clinically significant reductions in depression or anxiety). 60 - 80% 1. Psilocybin produces substantial and sustained decreases in depression and anxiety in patients with life - threatening cancer: A ra ndomized double - blind trial - Roland R Griffiths, Matthew W Johnson, Michael A Carducci, Annie Umbricht, William A Richards, Brian D Richards, Mary P Cosimano, Margaret A Klinedinst, 2016 (sagepub.com) Psilocybin Cancer Anxiety Study - Full Text View - ClinicalTrials.gov 2. Psilocybin Cancer Anxiety Study - Full Text View - ClinicalTrials.gov Psilocybin produces substantial and sustained decreases in depression and anxiety in patients with life - threatening cancer: A randomized double - blind trial Roland R Griffiths1,2, Matthew W Johnson1, Michael A Carducci} Annie Umbricht1, William A Richards1, Brian D Richards1, Mary P Cosimano1 and Margaret A Klinedinst1 At 6 - month follow - up, these changes were sustained, with about 80% of participants continuing to show clinically significant decreases in depressed mood and anxiety. >80%
22 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Identified Clinical Papers of Relevance Paper Published Source : https://clinicaltrials.gov/ct2/show/NCT05398484?term=NCT05398484&draw=2&rank=1
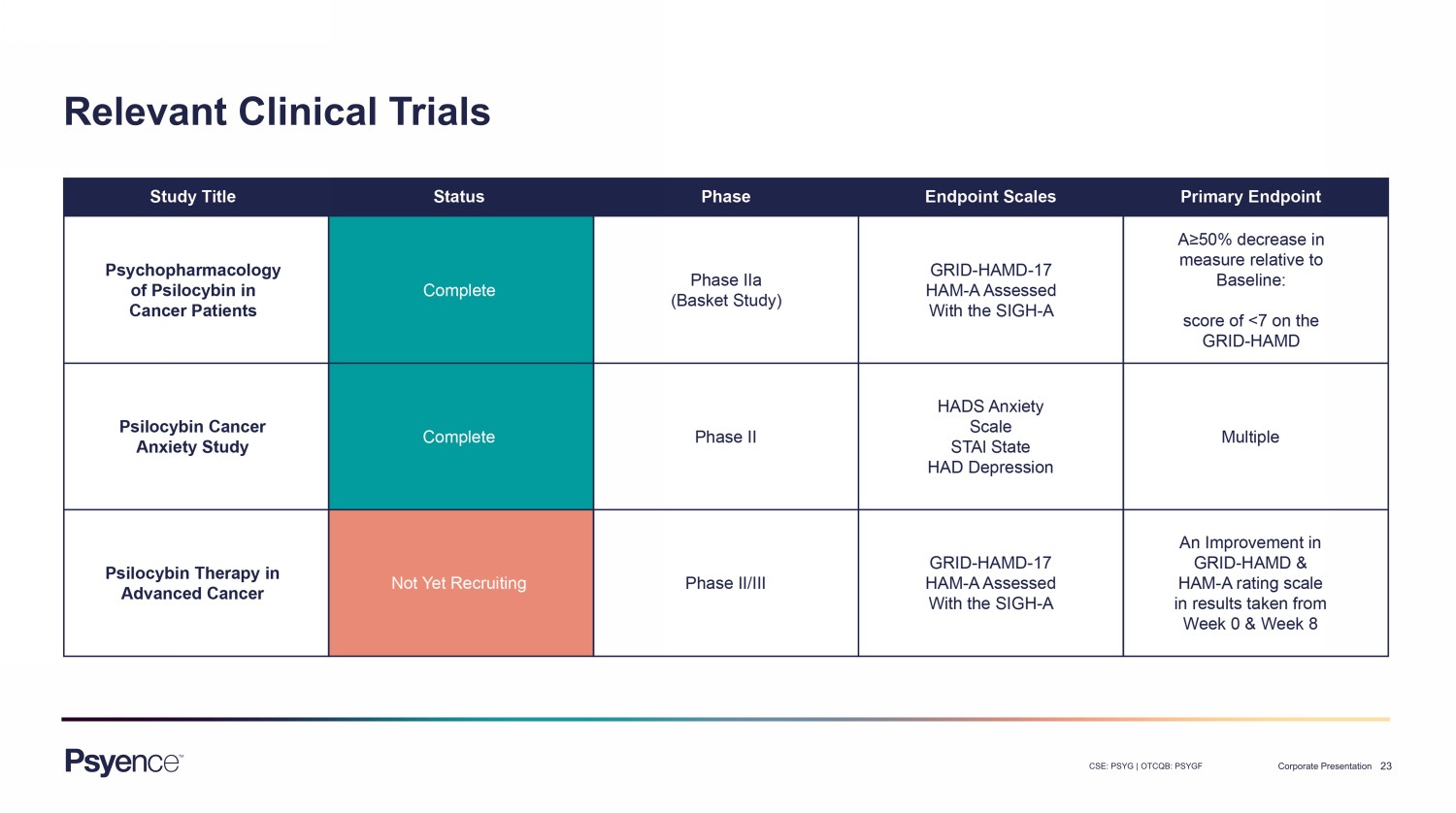
23 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Relevant Clinical Trials

24 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Product

25 CSE: PSYG | OTCQB: PSYGF Corporate Presentation • Psyence has Licensed in the Filament Health Patented technology • PEX 010 is the API for Development and commercialization • Naturally Derived Psilocybin • IP protected – 9 FDA and Health Canada granted Patents • Global Research and Commercial license granted for use in Palliative Care Psyence acquires Global License for Development and Commercialization https://psyence.com/2022/12/15/psyence - and - filament - health - announce - worldwide - licensing - agreement - of - psilocybin - capsule - for - pall iative - care/
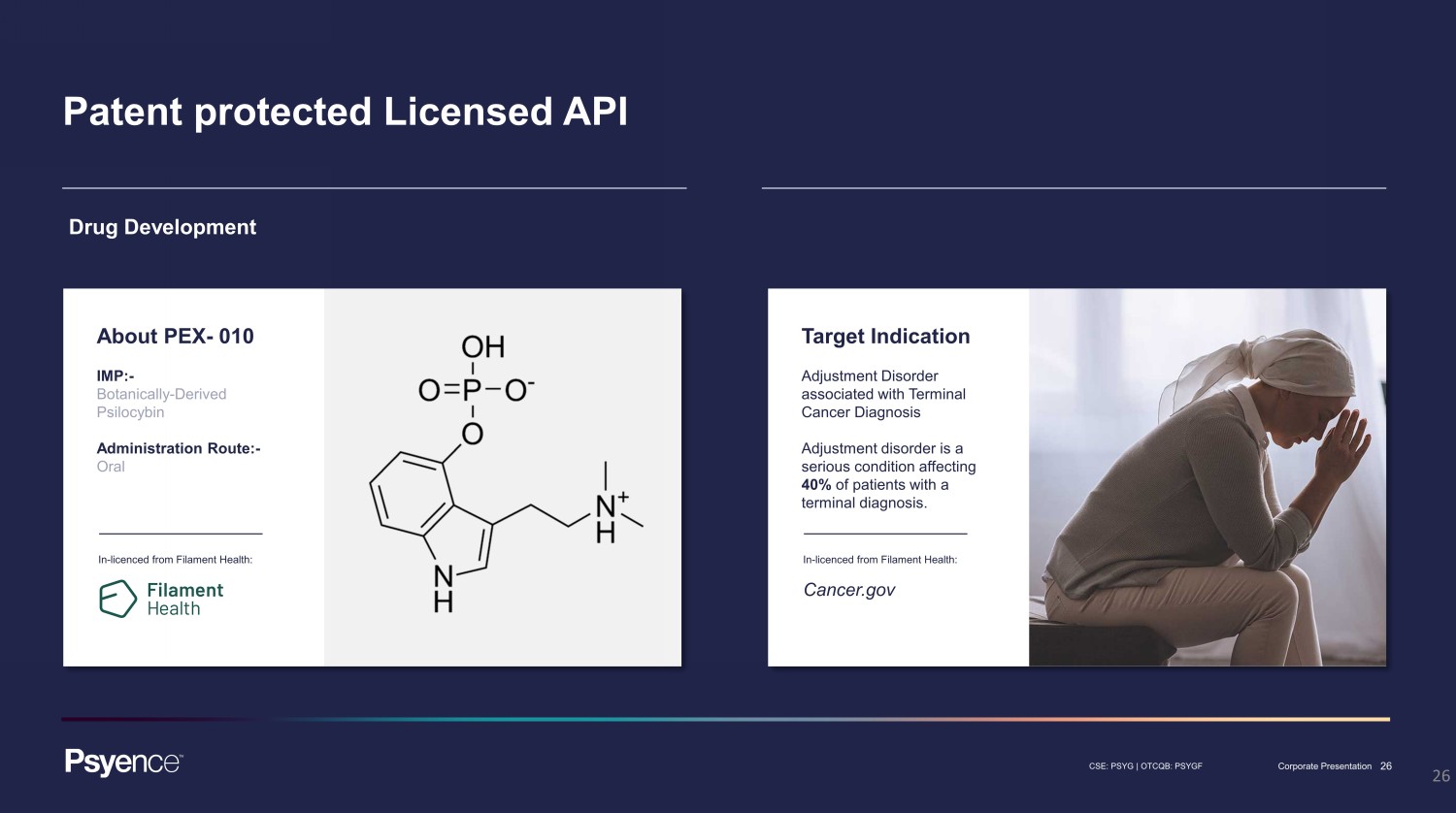
26 CSE: PSYG | OTCQB: PSYGF Corporate Presentation 26 Patent protected Licensed API Drug Development About PEX - 010 IMP: - Botanically - Derived Psilocybin Administration Route: - Oral In - licenced from Filament Health: Target Indication Adjustment Disorder associated with Terminal Cancer Diagnosis Adjustment disorder is a serious condition affecting 40% of patients with a terminal diagnosis. In - licenced from Filament Health: Cancer.gov

27 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Psyence is cultivating psilocybin mushrooms (psilocybe cubensis). Our natural psilocybin mushroom products are: 1 https://www.cbi.eu/market - information/natural - ingredients - health - products/trends 2 https://www.grandviewresearch.com/industry - analysis/pharmaceutical - manufacturing - market The Benefits of Natural Psilocybin Mushrooms Accessible Psyence has a federally licensed facility to cultivate natural psilocybin in Southern Africa. This product will be sent to our partners in Canada and the UK to extract and produce a standardized encapsulated product. Grown in an ISO 22000 certified facility Our facility is equipped with validated and calibrated specialized equipment to ensure optimal growing conditions and efficient harvesting and packaging. Nature - based and plant - base There is an increased consumer demand for nature - based medicine and plant - based products. This will likely incentivize the use of natural psilocybin as a therapeutic agent. Superior to synthetic molecules The psilocybin molecule from natural sources is the same as synthetically - derived products. However, the natural sourced product has other similar acting molecules to psilocybin which may have synergistic and desirable effects, known as the Entourage Effect. 27 CSE: PSYG | OTCQB: PSYGF Corporate Presentation

28 CSE: PSYG | OTCQB: PSYGF Corporate Presentation 28 Differentiated Strategy Psyence Target Indication Adjustment Disorder associated with Terminal Cancer Diagnosis Adjustment disorder is a serious condition affecting 40% of patients with a terminal diagnosis. Psychedelic Sector** • First mover in Palliative Care • Phase IIb Ready • Leading in Natural Psilocybin (Plant derived) vs Synthetic • Only Company in PhII with Natural • Entourage effect of Natural sustains efficacy* *https://entheonation.com/blog/entourage - effect - magic - mushrooms / **Psychedelics Drug Development Tracker - Psychedelic Alpha

Clinical Development Program for Palliative Care
30 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Driving our Clinical program: • Rigorous clinical trials to prove efficacy and safety • Partnership with Clerkenwell Health – MHRA [Medicines and Healthcare Products Regulatory Agency] for clinical trials in the UK • Large total market with robust CAGR (~8%) estimated to be US$25B by 2030. • New England Journal of Medicine: “Patients receiving Palliative Care had less depression, improved quality of life and survived 2.7 months longer” An opportunity for disruption in palliative care: With this condition, 39.8% were being treated with antidepressant medication, and 66.7% had been prescribed a benzodiazepine. Effective - but can diminish quality of life Centre to Advance Palliative Care [CAPC]: • 90 million Americans are living with serious illness – expected to double over the next 25 years • 6 million people in the United States could benefit from Palliative Care • Palliative Care integrated in hospitals could save $6 billion p.a. Proprietary Natural Psilocybin Drug Development And Treatment Protocols Focus on Palliative Care
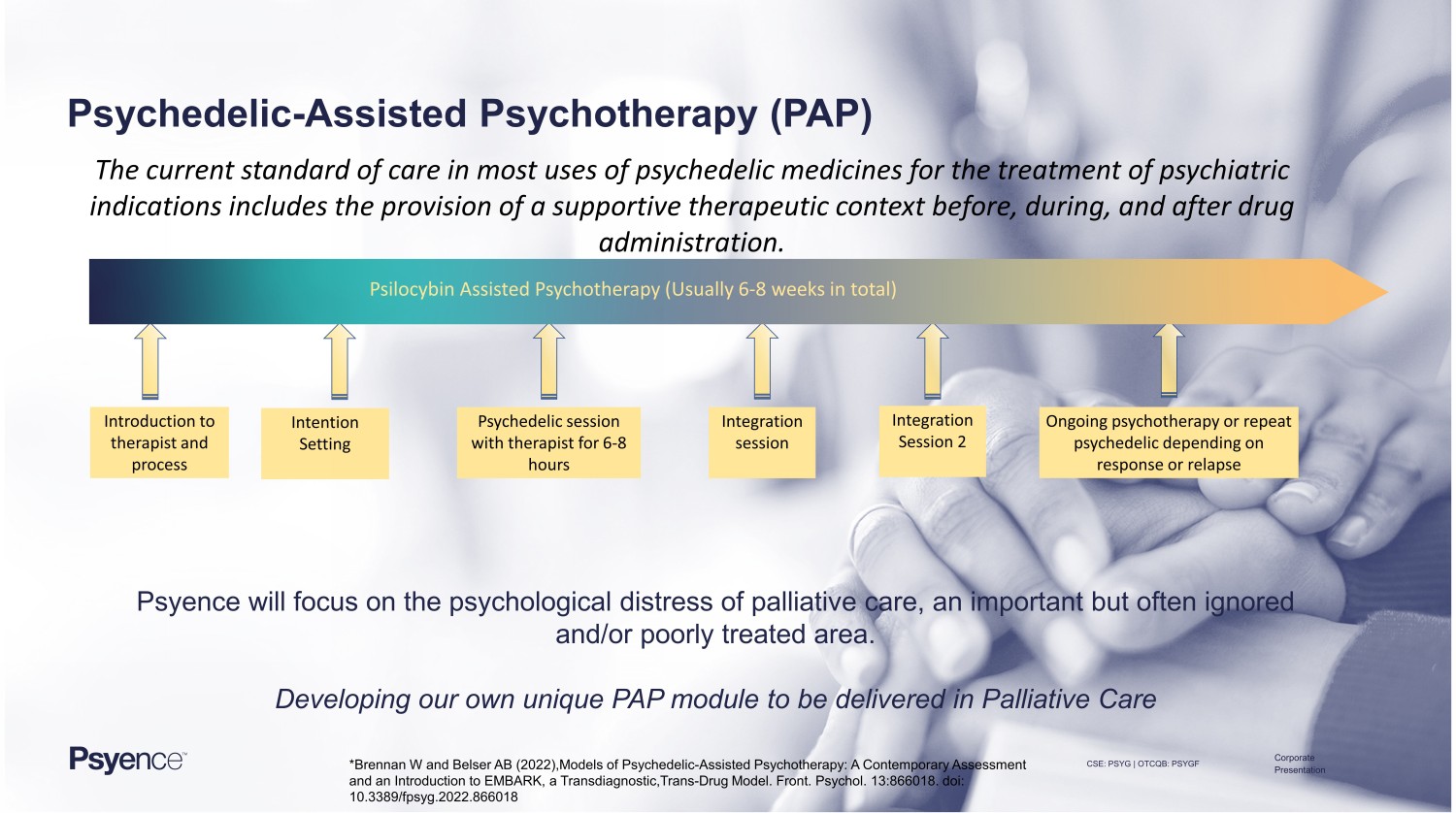
CSE: PSYG | OTCQB: PSYGF Corporate Presentation Psychedelic - Assisted Psychotherapy (PAP) The current standard of care in most uses of psychedelic medicines for the treatment of psychiatric indications includes the provision of a supportive therapeutic context before, during, and after drug administration. Psyence will focus on the psychological distress of palliative care, an important but often ignored and/or poorly treated area. Developing our own unique PAP module to be delivered in Palliative Care Introduction to therapist and process Psychedelic session with therapist for 6 - 8 hours Intention Setting Integration session Integration Session 2 Ongoing psychotherapy or repeat psychedelic depending on response or relapse Psilocybin Assisted Psychotherapy (Usually 6 - 8 weeks in total) *Brennan W and Belser AB (2022), Models of Psychedelic - Assisted Psychotherapy: A Contemporary Assessment and an Introduction to EMBARK, a Transdiagnostic,Trans - Drug Model. Front. Psychol. 13:866018. doi: 10.3389/fpsyg.2022.866018

32 CSE: PSYG | OTCQB: PSYGF Corporate Presentation UK Clinical Trial Phase IIa Psyence’s clinical trial program is focused on assessing the safety and efficacy of psilocybin - assisted psychotherapy for the treatment of adjustment disorder* due to an incurable cancer diagnosis versus psychotherapy alone. Our first clinical trial in the UK is pending MHRA approval. It is expected to commence in 2023. Our development strategy will include further studies including a pivotal study. Milestones Protocol submitted to the MHRA in May 2022 Trial Approved 15 th Sept - First patients expected to be screened in Q1 2023 Patients will be randomized to either placebo or treatment in Q1 * Adjustment disorder is under ICD - 11 and it’s a maladaptive reaction to an identifiable psychosocial stressor or multiple stressors that usually emerges within a month of the stressor. Overview Trial lead: Dr Emilio Arbe Location: St Pancras Hospital, London, plus one CRO site Stage: Phase IIa ( MHRA approved , expected to commence Q1, 2023) Description: A randomized, parallel, placebo - controlled, phase IIa, clinical trial to assess the efficacy of psilocybin - assisted psychotherapy for the treatment of adjustment disorder* due to an incurable cancer diagnosis versus psychotherapy alone. Partner: Clerkenwell (CRO) Product and Protocol The study protocol has been approved by the MHRA, using FH licensed product Expert and other reviews of the full protocol and patient materials completed Psilocybin mushrooms (psilocbybe cubensis) from our production facility in Southern Africa will be exported to our partner (Filament) in Canada. In parallel ,we are also considering a higher purity formulation (98%) to be developed in the UK by pharma CROs May 2022 Sept 2022 Q1 2023
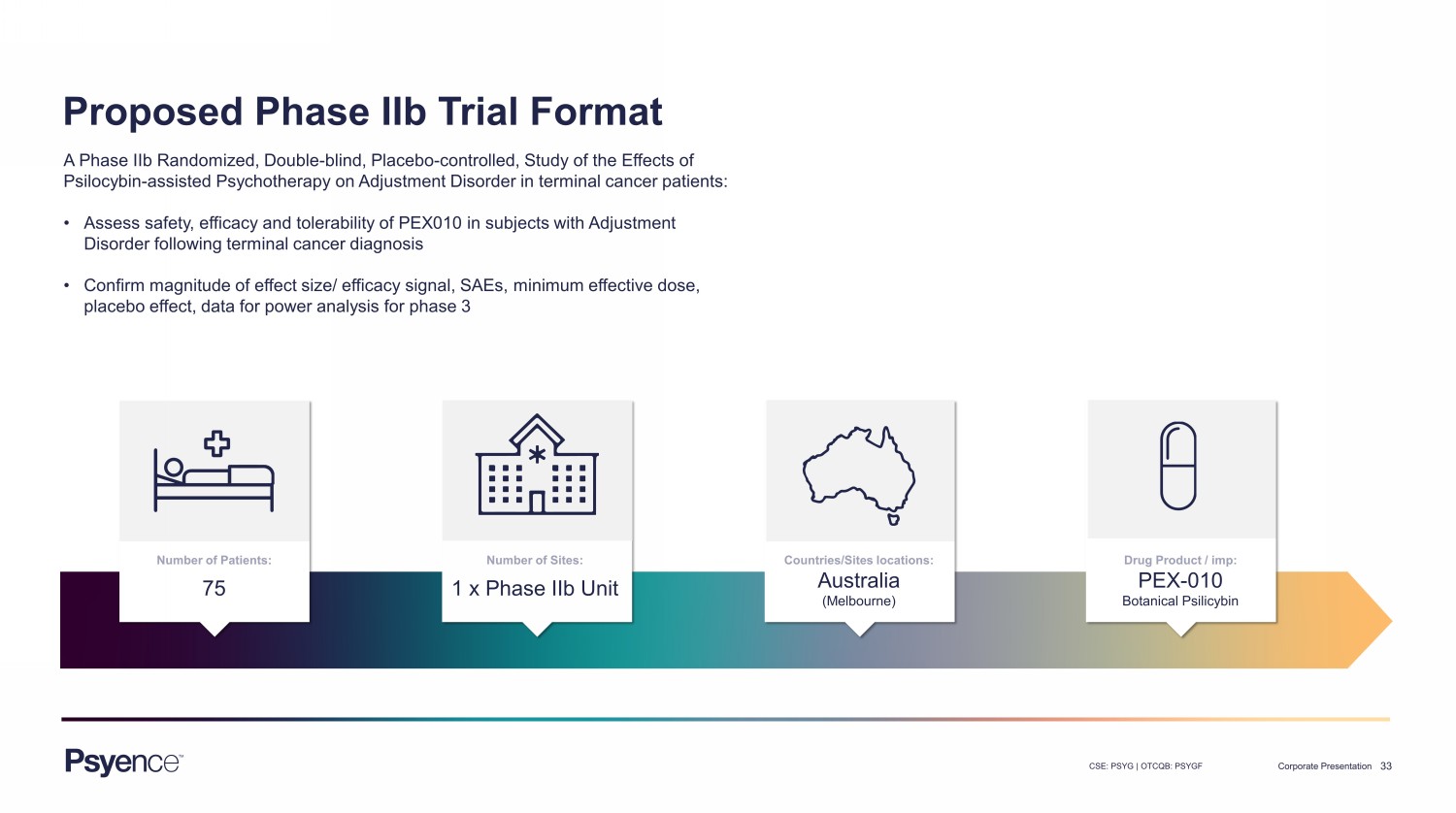
33 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Proposed Phase IIb Trial Format A Phase IIb Randomized, Double - blind, Placebo - controlled, Study of the Effects of Psilocybin - assisted Psychotherapy on Adjustment Disorder in terminal cancer patients: • Assess safety, efficacy and tolerability of PEX010 in subjects with Adjustment Disorder following terminal cancer diagnosis • Confirm magnitude of effect size/ efficacy signal, SAEs, minimum effective dose, placebo effect, data for power analysis for phase 3 Drug Product / imp: PEX - 010 Botanical Psilicybin Countries/Sites locations: Australia (Melbourne) Number of Sites: 1 x Phase IIb Unit Number of Patients: 75
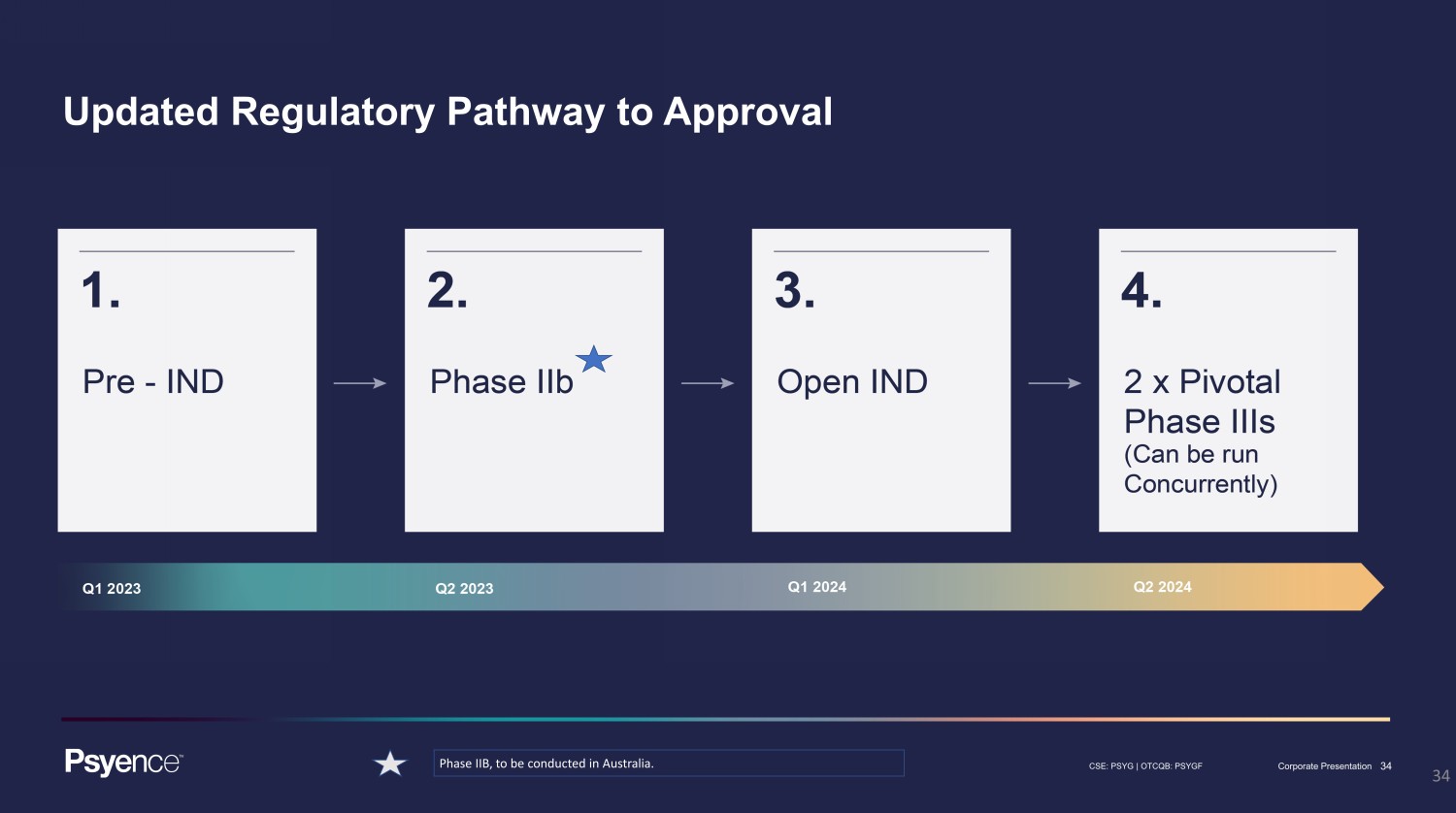
34 CSE: PSYG | OTCQB: PSYGF Corporate Presentation 34 Updated Regulatory Pathway to Approval Phase IIB, to be conducted in Australia.
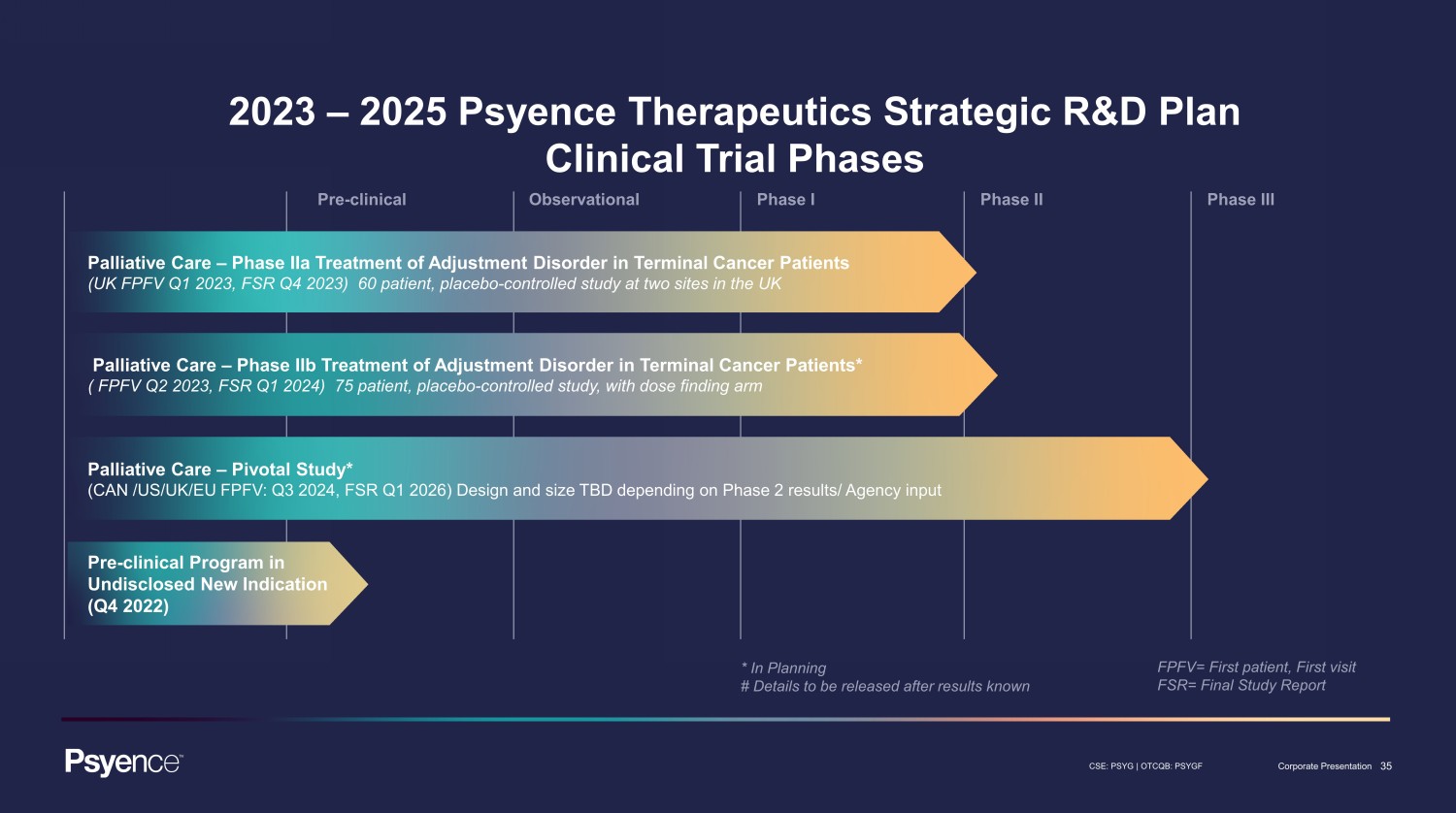
35 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Pre - clinical Observational Phase I Phase II Phase III 2023 – 2025 Psyence Therapeutics Strategic R&D Plan Clinical Trial Phases * In Planning # Details to be released after results known FPFV= First patient, First visit FSR= Final Study Report Palliative Care – Phase IIa Treatment of Adjustment Disorder in Terminal Cancer Patients (UK FPFV Q1 2023, FSR Q4 2023) 60 patient, placebo - controlled study at two sites in the UK Pre - clinical Program in Undisclosed New Indication (Q4 2022) Palliative Care – Pivotal Study* (CAN /US/UK/EU FPFV: Q3 2024, FSR Q1 2026) Design and size TBD depending on Phase 2 results/ Agency input Palliative Care – Phase IIb Treatment of Adjustment Disorder in Terminal Cancer Patients* ( FPFV Q2 2023, FSR Q1 2024) 75 patient, placebo - controlled study, with dose finding arm

36 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Psyence’s Intellectual Property within Drug development May 2022 Q4 2022 Q4 2022 Regulatory Protection Data and Marketing Exclusivity: • First indication with Molecule will be granted protection from any generic entry for periods up to 10 years • Upon approval, US FDA grants NCE protection for 5 years • Upon approval, EU grants equivalent protection for up to 10 years Intellectual Property Protection • Licensing and Exclusivity of Patent Protected PEX 010 9 granted patents • Psychedelic Assisted Therapy Module (PAPM). P roprietary psychedelic treatment modules for the delivery of Palliative Care Psychotherapy in conjunction with our Medication • University Collaborations o Generating Unique genetic Psilocybin mushroom strains for development by Psyence

37 CSE: PSYG | OTCQB: PSYGF Corporate Presentation In Summary Licensed producer of natural psilocybin • Psyence Therapeutics, developing natural psilocybin for the treatment of Adjustment Disorder in palliative care, is combining with NewCourt SPAC • The two companies have entered into a Business Combination agreement as of Jan 6 th 2023 • Resulting entity will have $20MUSD: • Completion of Ph IIb • Open FDA IND • Initiate Ph III registrational studies • Exploring 2 nd key indication Drug Development

38 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Readers are therefore cautioned not to place undue reliance on such forward - looking statements . In addition, in considering any prior performance information contained in this presentation, readers should bear in mind that past results are not necessarily indicative of future results, and there can be no assurance that Psyence will achieve results comparable to those discussed in this presentation . This presentation speaks as of the date hereof and neither Psyence nor any affiliate or representative thereof assumes any obligation to provide any recipient of this presentation with subsequent revisions or updates to any historical or forward - looking information contained in this presentation to reflect the occurrence of events and/or changes in circumstances after the date hereof, except as may be required by law . Risk Factors relating to Psyence’s business The Company’s operating and financial forecasts, which are subject to various known and unknown contingencies and factors outside of the Company’s control, may not prove accurate and the Company may not achieve results consistent with management’s expectations. The Company’s limited operating history and financial results make the Company’s future results, prospects and the risks the Company may encounter difficult to predict. The Company will require additional capital to support the growth of its business, which may not be available on terms acceptable to it, or at all. Because the Company will be initially focused on research and development, cultivation and production of psilocybin mushrooms, the prospects for the Company’s success will be dependent upon the future performance and market acceptance of the Company’s intended facilities, products, processes and services. Unlike certain entities that have the resources to develop and explore numerous product lines or operate in multiple industries, the Company does not anticipate having the ability to immediately diversify or benefit from the possible spreading of risks or offsetting of losses. The Company operates in the psychedelic areas of the mushroom industry. Again, the prospects for the Company’s success may become dependent upon the development or market acceptance of a very limited number of facilities, products, processes or services. There is no assurance as to whether the Company will become profitable or pay dividends. The Company has incurred and anticipates that it will continue to incur substantial expenses relating to the development and initial operations of its business. The payment and amount of any future dividends will depend upon, among other things, the Company’s results of operations, cash flow, financial condition and operating and capital requirements. There is no assurance that future dividends will be paid, and, if dividends are paid, there is no assurance with respect to the amount of any such dividends. The Company is reliant on employees and third - party consultants to assist in investigating the process of developing and commercializing its psilocybin mushroom products. No assurance can be given that the results of these investigations will determine that manufacturing and distribution of its products will be feasible or commercially viable. A failure to obtain satisfactory results on these investigations could have a material adverse effect on the Company’s business and may adversely affect the Company’s ability to begin earning revenue. Psyence is subject to risks associated with operating internationally. Several factors, including legal and regulatory compliance and weakened economic conditions in any of the international jurisdictions in which the Company expects to do business or have projects, could adversely affect such expansion and growth. Additionally, the Company’s entry into new international jurisdictions requires management attention and financial resources that would otherwise be spent on other parts of the business. Some of the countries in which the Company expects to sell products are to some degree subject to political, economic, and/or social instability. International business operations expose the Company to risks and expenses inherent in operating or selling products in foreign jurisdictions, and developing and emerging markets in particular, where these risks may be heightened. There is no guarantee that the Company’s intended project implementation timelines will be met as anticipated, or at all. The failure to achieve these milestones and deliverables could negatively impact the Company’s ability to raise additional funds required to fund its ongoing operations and research and development initiatives, ultimately impacting the financial viability of the Company. There is also no guarantee that the Company’s research and development efforts will result in commercially viable products, suitable for registration with the necessary authorities. The current global uncertainty with respect to the resurgence of COVID - 19 and its effect on the global economy, may have negative effects on the Company. Risk Factors Related to the Proposed Transaction The Proposed Transaction is subject to a number of conditions to closing including among others: obtaining Newcourt shareholder approval, Psyence Biomed, and, if necessary, Psyence shareholder approval of the Business Combination; the completion of regulatory review from the SEC and the Canadian Securities Exchange (the “CSE”); the filing effectiveness of the Registration Statement; obtaining required consents or approvals; and the resignation of certain Newcourt’s directors and officers. The obligations of each of Newcourt, Psyence Biomed, and Psyence to consummate the Business Combination are also conditioned on, among other things, the accuracy of the representations and warranties as set forth by the other parties in the Business Combination Agreement (subject to certain materiality qualifications) and the performance by the other parties, in all material respects, of their obligations under the Business Combination Agreement required to be performed at or prior to the closing. In addition, consummation of the Business Combination is subject to the parties being satisfied with their due diligence of the other parties, any conditions that the CSE may impose, including, if required, Psyence shareholder approval, and is also predicated on the parties settling and executing a number of ancillary agreements, including an amended and restated Newcourt shareholder rights agreement, lock - up agreements with Newcourt SPAC Sponsor LLC (the “Sponsor”), a support agreement with the Sponsor, and a support agreement with certain Psyence shareholders.

39 CSE: PSYG | OTCQB: PSYGF Corporate Presentation Back up Slides

Supportive literature PAP • “The first wave of psychedelic medicines in psychiatry will almost certainly hold as axiomatic that psychedelic medicines are best administered within a well - considered context of psychotherapy.” * Brennan W and Belser AB (2022), Models of Psychedelic - Assisted Psychotherapy: A Contemporary Assessment and an Introduction to EMBARK, a Transdiagnostic,Trans - D rug Model. Front. Psychol. 13:866018. doi: 10.3389/fpsyg.2022.866018 • PAP can help solve many pressing safety concerns in current psychopharmacological treatments. • PAP can reduce or even eliminate drug adherence problems and polypharmacy. * Schenberg EE (2018) Psychedelic - Assisted Psychotherapy: A Paradigm Shift in Psychiatric Research and Development. Front. Pharmacol. 9:733. doi: 10.3389/fphar.2018.00733 • Psychedelic Assisted Psychotherapy as it is practiced ………..cognitive - behavioral frameworks (i.e., CBT, DBT, and ACT) should be considered the default * Yaden DB, Earp D, Graziosi M, Friedman - Wheeler D, Luoma JB and Johnson MW (2022) Psychedelics and Psychotherapy: Cognitive - Behavioral Approaches as Default. Front. Psychol. 13:873279. doi: 10 .3389/fpsyg.2022.873279