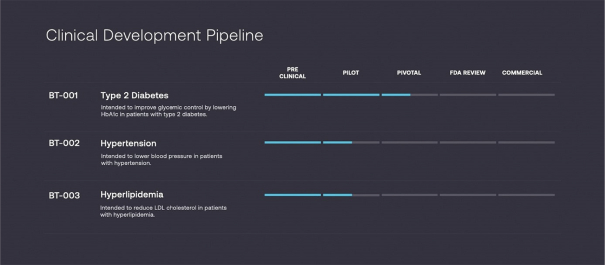
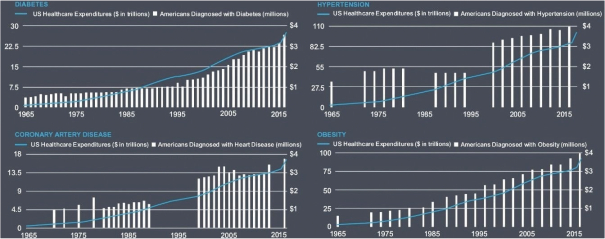
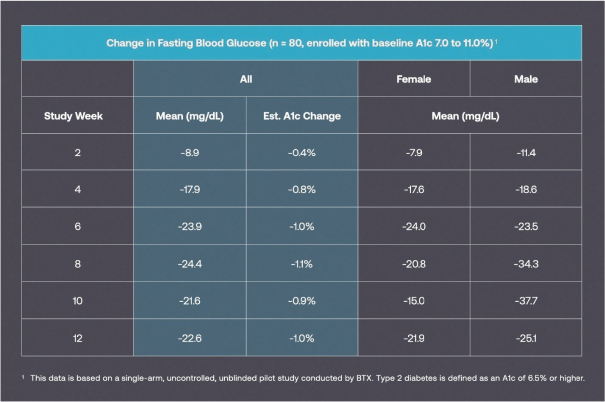
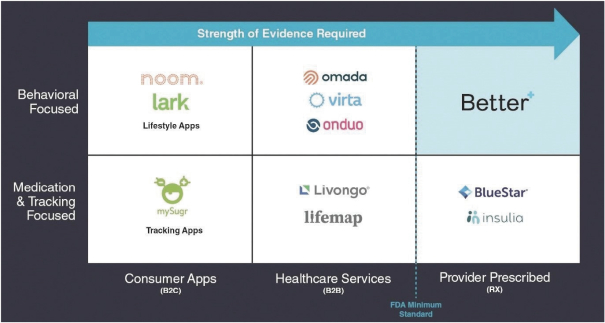
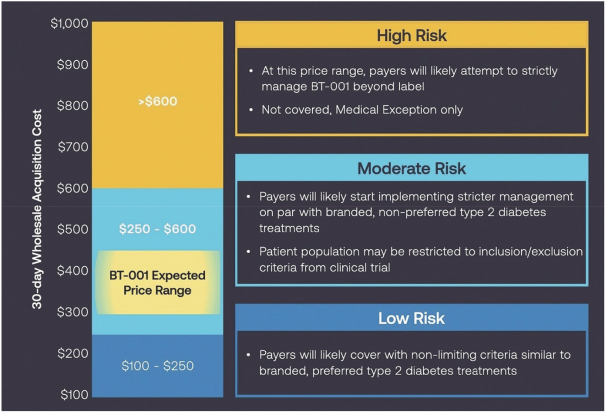
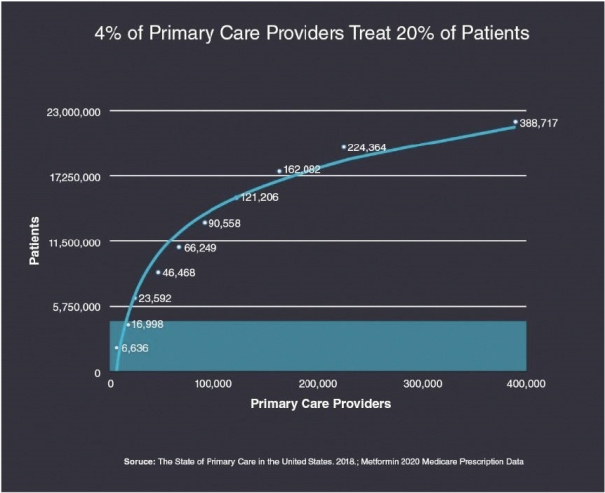
Information contained herein is subject to completion or amendment. A registration statement relating to these securities has been filed with the SEC. These securities may not be sold nor may offers to buy be accepted prior to the time the registration statement becomes effective. This prospectus shall not constitute an offer to sell or the solicitation of an offer to buy nor shall there be any sale of these securities in any jurisdiction in which such offer, solicitation or sale would be unlawful.
SUBJECT TO COMPLETION, DATED APRIL 1, 2022
PRELIMINARY PROSPECTUS

Up to 20,406,908 Shares of Common Stock
This prospectus relates to the offer and sale, from time to time, by the selling stockholders named in this prospectus (the “Selling Stockholders”), or any of their pledgees, donees, assignees and successors-in-interest (“permitted transferees”), of (i) up to an aggregate of 5,000,000 shares of our common stock that were issued to certain investors (collectively, the “PIPE Investors”) in a private placement in connection with the closing of the Business Combination (as defined below), and (ii) up to an aggregate of 15,406,908 shares of our common stock otherwise held by the Selling Stockholders. This prospectus also covers any additional securities that may become issuable by reason of share splits, share dividends or other similar transactions.
We will not receive any proceeds from the sale of shares of common stock by the Selling Stockholders pursuant to this prospectus. However, we will pay the expenses, other than underwriting discounts and commissions and certain expenses incurred by the Selling Stockholders in disposing of the securities, associated with the sale of securities pursuant to this prospectus.
We are registering the offer and sale of the securities described above to satisfy certain registration rights we have granted. Our registration of the securities covered by this prospectus does not mean that either we or the Selling Stockholders will issue, offer or sell, as applicable, any of the securities. The Selling Stockholders and any of their permitted transferees may offer and sell the securities covered by this prospectus in a number of different ways and at varying prices. Additional information on the Selling Stockholders, and the times and manner in which they may offer and sell the securities under this prospectus, is provided under “” and “” in this prospectus.
Selling Stockholders
Plan of Distribution
You should read this prospectus and any prospectus supplement or amendment carefully before you invest in our securities.
Our common stock is listed on Nasdaq under the symbol “BTTX”. On March 31, 2022, the closing price of our common stock was $1.98 per share.
We are an “emerging growth company,” as that term is defined under the federal securities laws and, as such, are subject to certain reduced public company reporting requirements.
Investing in our securities involves risks that are described in the “
” section beginning on page 10 of this prospectus.
Neither the SEC nor any state securities commission has approved or disapproved of the securities to be issued under this prospectus or determined if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
The date of this prospectus is , 2022.