
PACIFIC Study Topline Data January 2, 2024 Exhibit 99.2
Forward-Looking Statements This presentation contains forward-looking statements about Longboard Pharmaceuticals, Inc. (“we,” “Longboard” or the “Company”), including statements regarding: the potential of LP352 (including to be best-in-class, to change the DEE landscape, to serve as the cornerstone of a potential world-class epilepsy franchise, to have intellectual property protection through 2041, and to move rapidly into a global Phase 3 program); the competitive landscape, commercial opportunities and analogs; our development approach; the prevalence of unmet need associated with, and market opportunity for, DEEs; and other statements that are not historical facts, including statements that may include words such as “will”, “may”, “can”, “would”, “intend”, “plan”, “expect”, “believe”, “potential”, “opportunity” and similar words. For such statements, we claim the protection of the Private Securities Litigation Reform Act of 1995. These forward-looking statements are subject to a number of risks, uncertainties and assumptions, including, but not limited to: topline data may not reflect the complete or final results of a particular study or trial, and are subject to change; nonclinical and clinical data are voluminous and detailed, and regulatory agencies may interpret or weigh the importance of data differently and reach different conclusions than Longboard or others, request additional information, have additional recommendations or change their guidance or requirements before or after approval; our limited operating history; our history of incurring net losses and expectation that we will continue to incur net losses for the foreseeable future, and that we may never be profitable; our need for additional funding and related risks for our business, product development programs and future commercialization activities; the timing and success of clinical trials and preclinical studies we conduct; the ability to obtain and maintain regulatory approval to conduct our clinical trials (in the manner we propose or at all) and, ultimately, to market our product candidates; the ability to commercialize our product candidates; our ability to compete in the marketplace; risks regarding our license and dependencies on others; our ability to obtain and maintain intellectual property protection and freedom to operate for our product candidates; our ability to manage our growth; and other risks and factors disclosed in our filings with the U.S. Securities and Exchange Commission, including our most recently filed Annual Report on Form 10-K, subsequently filed Quarterly Reports on Form 10-Q, and in our other filings. We operate in a very competitive and rapidly changing environment. New risks emerge from time to time. It is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements we may make. The forward-looking events and circumstances discussed in this presentation may not occur and actual results could differ materially and adversely from those anticipated or implied in the forward-looking statements. Except as required by law, we assume no responsibility for the accuracy and completeness of the forward-looking statements, and we undertake no obligation to update any forward-looking statements after the date of this presentation to conform these statements to actual results or to changes in our expectations. Certain information contained in this presentation and statements made orally during this presentation relate to or are based on studies, research, publications, surveys and other data obtained from third-party sources and Longboard’s own internal estimates and research. While Longboard believes these third-party studies, research, publications, surveys and other data to be reliable as of the date of this presentation, they have not been independently verified, and Longboard makes no representations as to the adequacy, fairness, accuracy or completeness of any information obtained from third-party sources. This presentation discusses product candidates that have not yet been approved for marketing by the U.S. Food and Drug Administration (the “FDA”).

PACIFIC Study Topline Data January 2, 2024

Bexicaserin Has the Potential to Change the DEE Landscape *In the PACIFIC Study **Composition of matter through 2036 with potential for PTE / PTA (2041) Bexicaserin provides the cornerstone to potentially build a world-class epilepsy franchise Studies to date highlight bexicaserin as potentially best-in-class Composition of matter IP protection through 2041** provides the opportunity to maximize the full potential of LP352 Rapidly moving forward with preparations for a global Phase 3 program 53.3% Median Reduction in Seizures* 72.1% Dravet 48.1% LGS 61.2% DEE Other

6 mg 9 mg 12 mg Participant remains on 6, 9 or 12 mg based on tolerability during up-titration 5 Wks Screening Period 30 Days Follow-up Period Randomization & Up-Titration Days 1-15 Maintenance* Days 16-75 Down- Titration Days 76- 80/90** Bexicaserin (LP352) Ph 1b/2a PACIFIC Study in Participants with DEEs * Maintenance Dose of bexicaserin (TID): 6 mg, 9 mg, 12 mg or placebo TID ** Up to a 15-day down-titration/taper period (reducing dose every 5 days) depending on the last maintenance dose Definitions: LGS = Lennox-Gastaut Syndrome, CDD = CDKL5 Deficiency Disorder Open- Label Extension Double-blind, placebo-controlled study to assess the safety, tolerability, pharmacokinetics and efficacy of bexicaserin Study Objectives: Evaluate reduction in countable motor seizures across a broad group of epilepsies Identify potential indications for pivotal studies Analyze concentration response to understand dosing in different seizure types and disorders Placebo (n=9) LP352 (n=43) Key Inclusion Criteria: DEEs with average of ≥ 4 motor seizures per 4-week period during the 12 weeks prior to screening and ≥ 4 motor seizures in the 4-week period of screening DEEs (multiple syndromes) including LGS, Dravet syndrome, SCN2A-related epilepsies, CDD, among others Key Exclusion Criteria: Use of fenfluramine & lorcaserin Basic Information: Sites: 34 sites Ages: ≥ 12 to ≤ 65 yrs old No Echocardiograms Required in PACIFIC
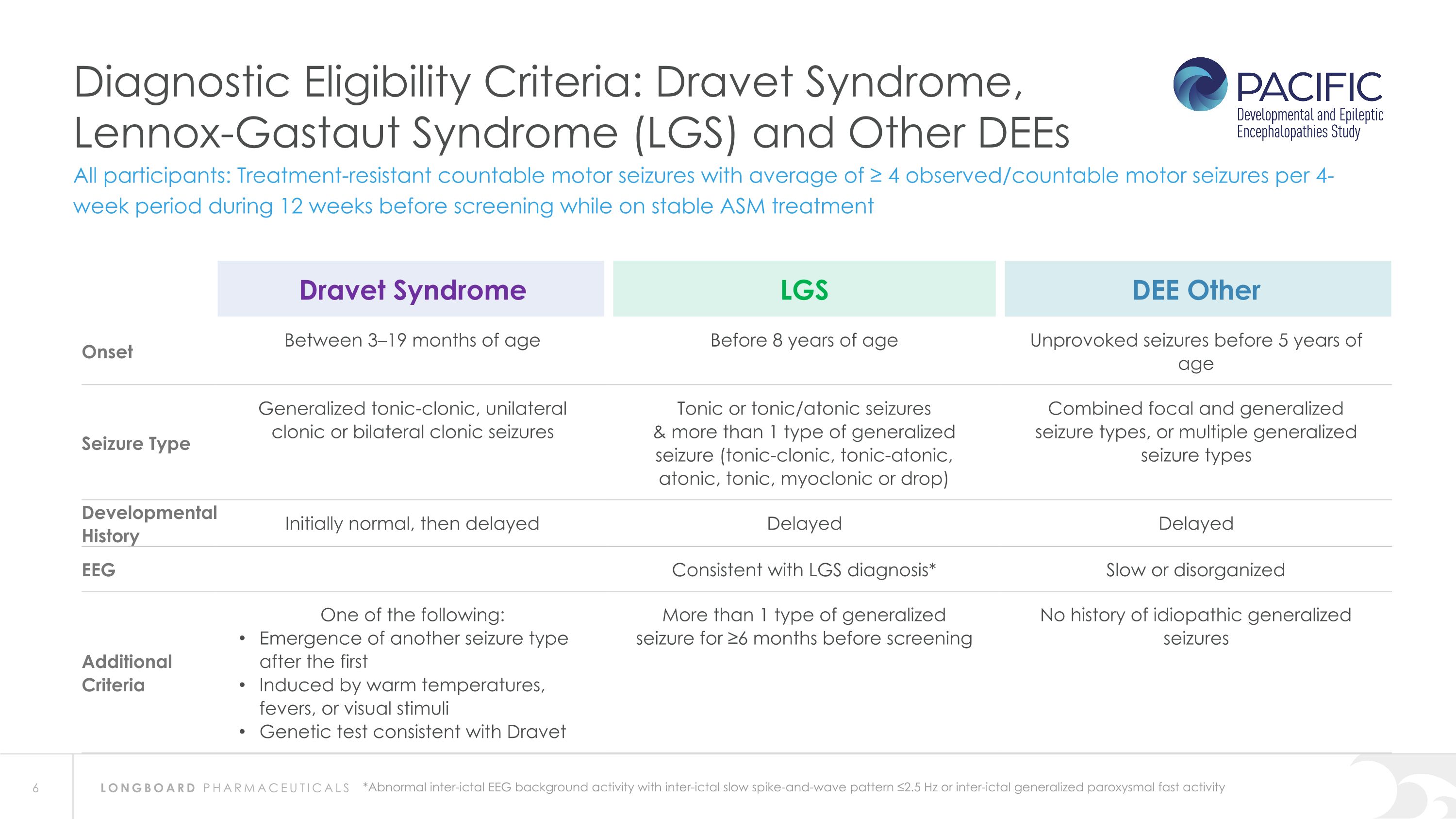
Diagnostic Eligibility Criteria: Dravet Syndrome, Lennox-Gastaut Syndrome (LGS) and Other DEEs *Abnormal inter-ictal EEG background activity with inter-ictal slow spike-and-wave pattern ≤2.5 Hz or inter-ictal generalized paroxysmal fast activity All participants: Treatment-resistant countable motor seizures with average of ≥ 4 observed/countable motor seizures per 4-week period during 12 weeks before screening while on stable ASM treatment Dravet Syndrome LGS DEE Other Onset Between 3–19 months of age Before 8 years of age Unprovoked seizures before 5 years of age Seizure Type Generalized tonic-clonic, unilateral clonic or bilateral clonic seizures Tonic or tonic/atonic seizures & more than 1 type of generalized seizure (tonic-clonic, tonic-atonic, atonic, tonic, myoclonic or drop) Combined focal and generalized seizure types, or multiple generalized seizure types Developmental History Initially normal, then delayed Delayed Delayed EEG Consistent with LGS diagnosis* Slow or disorganized Additional Criteria One of the following: Emergence of another seizure type after the first Induced by warm temperatures, fevers, or visual stimuli Genetic test consistent with Dravet More than 1 type of generalized seizure for ≥6 months before screening No history of idiopathic generalized seizures

Topline Participant Disposition & Safety Results Summary
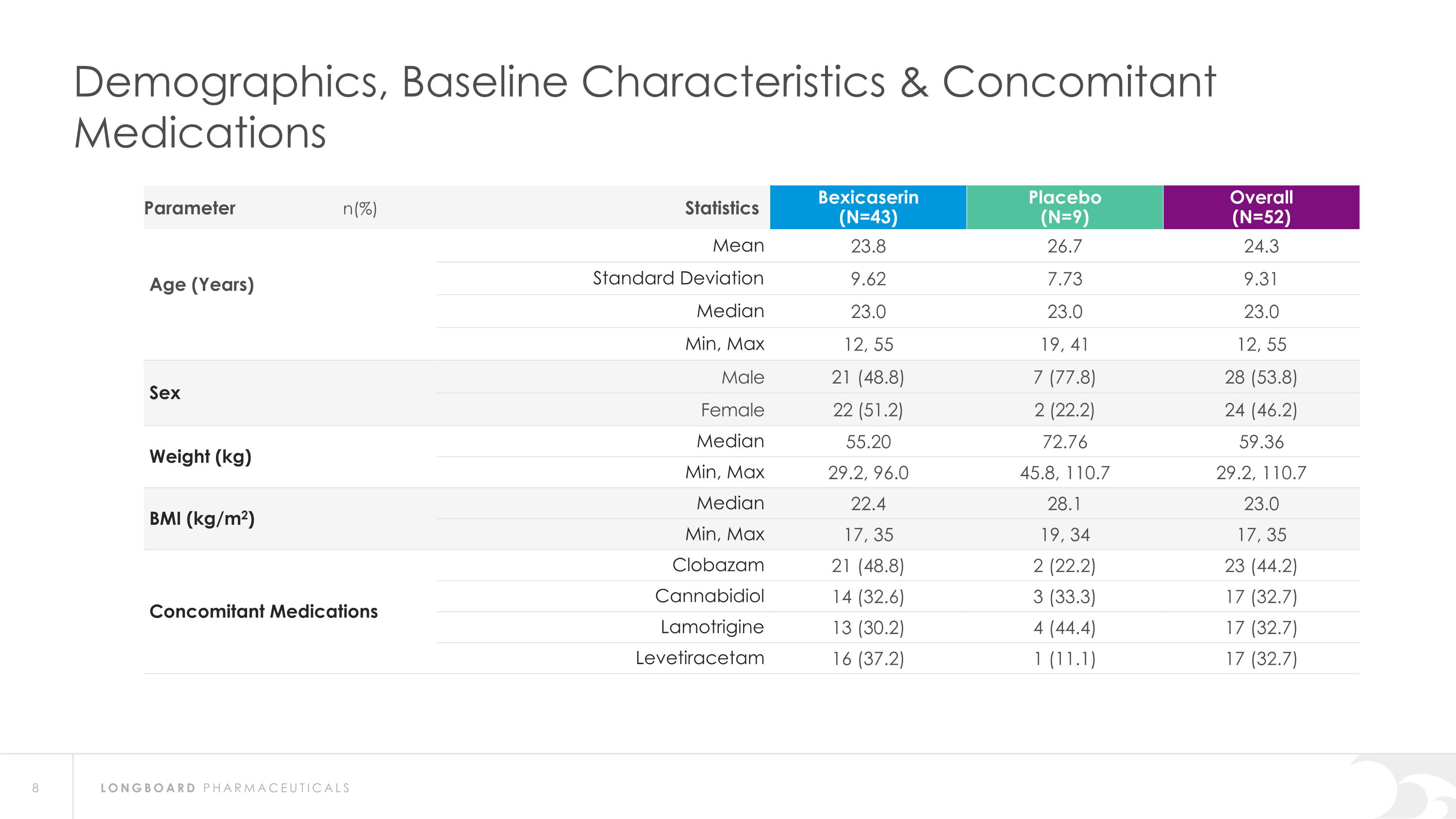
Demographics, Baseline Characteristics & Concomitant Medications Parameter n(%) Statistics Bexicaserin (N=43) Placebo (N=9) Overall (N=52) Age (Years) Mean 23.8 26.7 24.3 Standard Deviation 9.62 7.73 9.31 Median 23.0 23.0 23.0 Min, Max 12, 55 19, 41 12, 55 Sex Male 21 (48.8) 7 (77.8) 28 (53.8) Female 22 (51.2) 2 (22.2) 24 (46.2) Weight (kg) Median 55.20 72.76 59.36 Min, Max 29.2, 96.0 45.8, 110.7 29.2, 110.7 BMI (kg/m2) Median 22.4 28.1 23.0 Min, Max 17, 35 19, 34 17, 35 Concomitant Medications Clobazam 21 (48.8) 2 (22.2) 23 (44.2) Cannabidiol 14 (32.6) 3 (33.3) 17 (32.7) Lamotrigine 13 (30.2) 4 (44.4) 17 (32.7) Levetiracetam 16 (37.2) 1 (11.1) 17 (32.7)
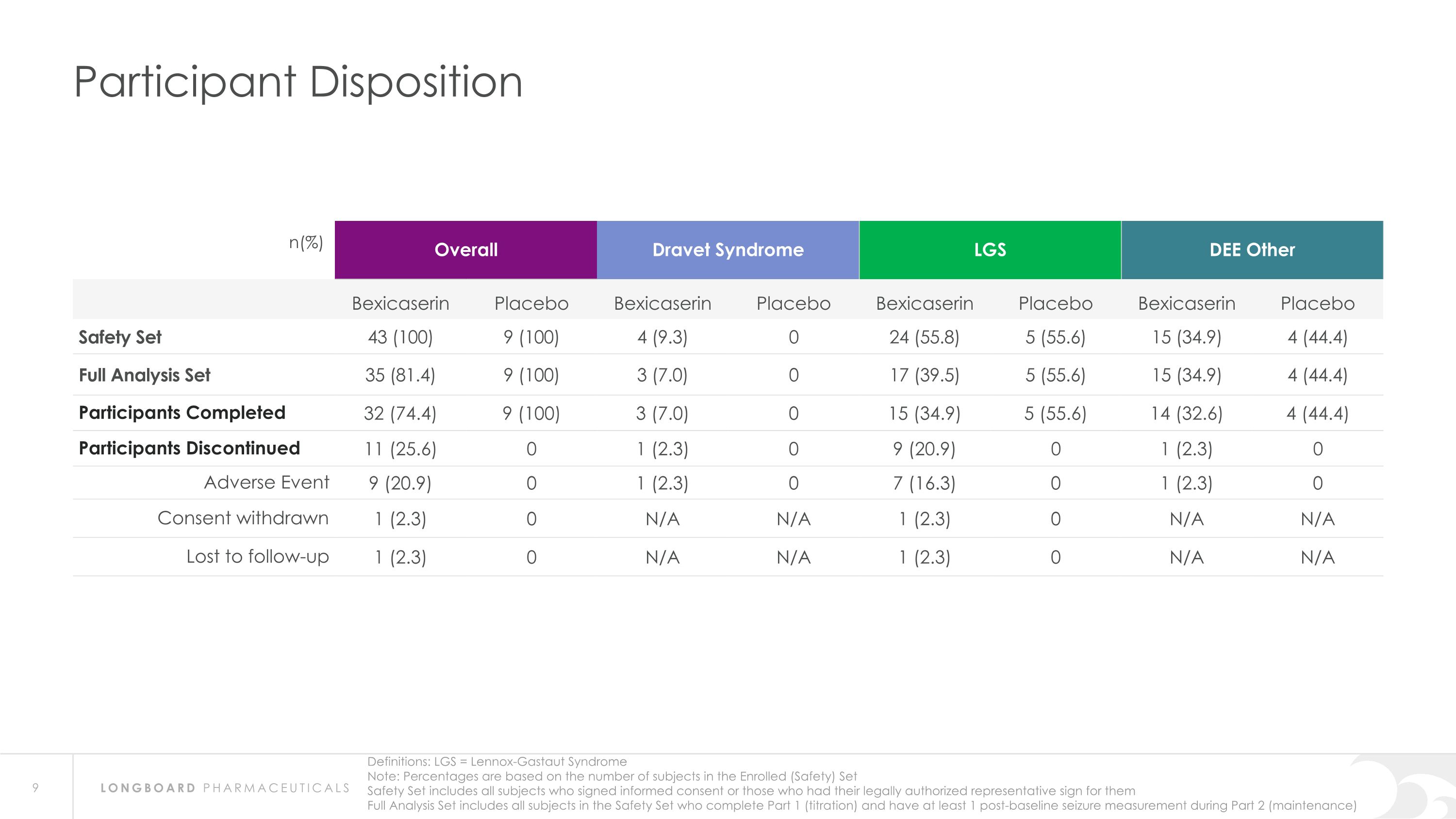
Participant Disposition n(%) Overall Dravet Syndrome LGS LGS DEE Other Bexicaserin Placebo Bexicaserin Placebo Bexicaserin Placebo Bexicaserin Placebo Safety Set 43 (100) 9 (100) 4 (9.3) 0 24 (55.8) 5 (55.6) 15 (34.9) 4 (44.4) Full Analysis Set 35 (81.4) 9 (100) 3 (7.0) 0 17 (39.5) 5 (55.6) 15 (34.9) 4 (44.4) Participants Completed 32 (74.4) 9 (100) 3 (7.0) 0 15 (34.9) 5 (55.6) 14 (32.6) 4 (44.4) Participants Discontinued 11 (25.6) 0 1 (2.3) 0 9 (20.9) 0 1 (2.3) 0 Adverse Event 9 (20.9) 0 1 (2.3) 0 7 (16.3) 0 1 (2.3) 0 Consent withdrawn 1 (2.3) 0 N/A N/A 1 (2.3) 0 N/A N/A Lost to follow-up 1 (2.3) 0 N/A N/A 1 (2.3) 0 N/A N/A Note: Percentages are based on the number of subjects in the Enrolled (Safety) Set Safety Set includes all subjects who signed informed consent or those who had their legally authorized representative sign for them Full Analysis Set includes all subjects in the Safety Set who complete Part 1 (titration) and have at least 1 post-baseline seizure measurement during Part 2 (maintenance) Definitions: LGS = Lennox-Gastaut Syndrome
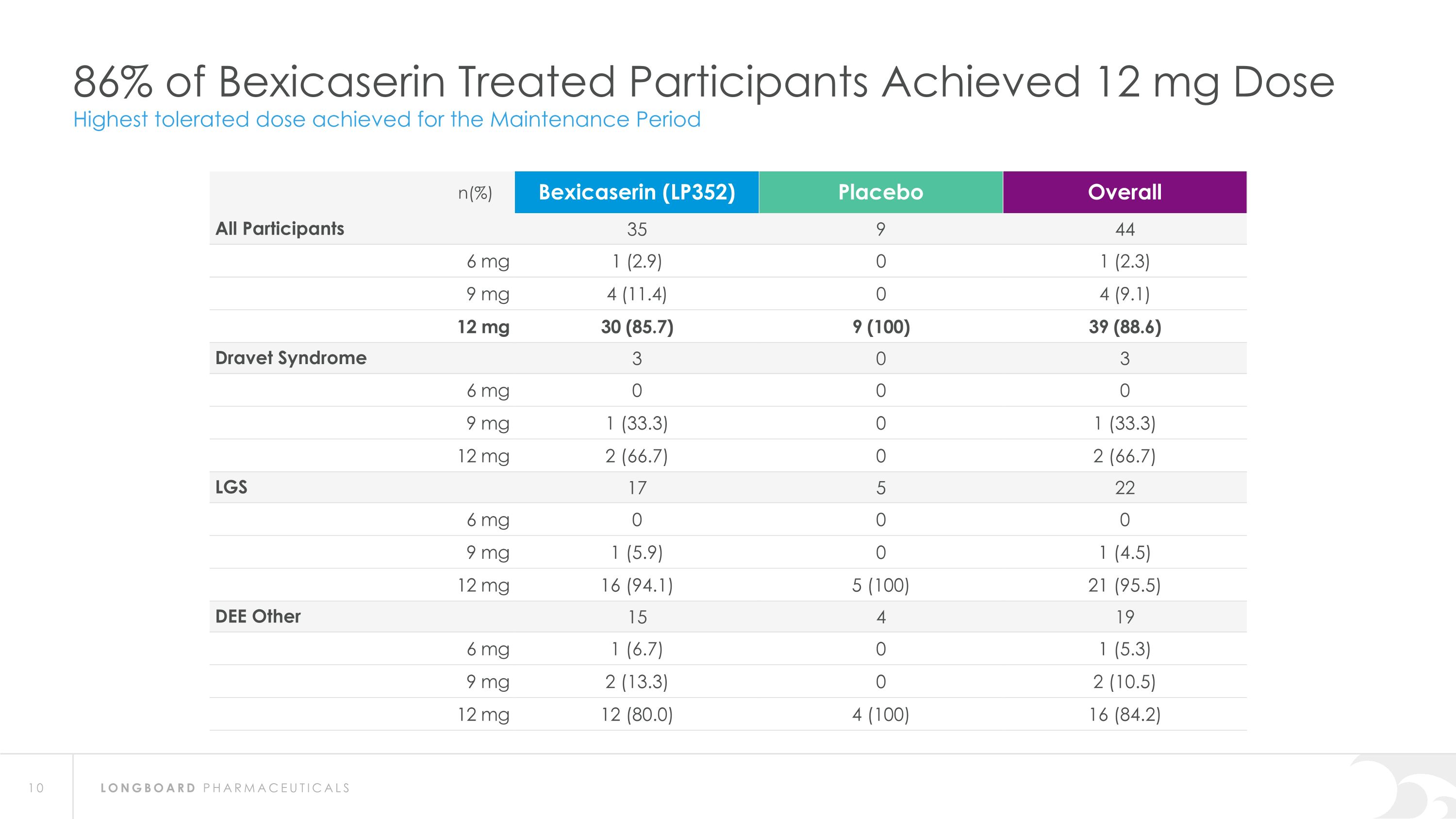
86% of Bexicaserin Treated Participants Achieved 12 mg Dose Highest tolerated dose achieved for the Maintenance Period n(%) Bexicaserin (LP352) Placebo Overall All Participants 35 9 44 6 mg 1 (2.9) 0 1 (2.3) 9 mg 4 (11.4) 0 4 (9.1) 12 mg 30 (85.7) 9 (100) 39 (88.6) Dravet Syndrome 3 0 3 6 mg 0 0 0 9 mg 1 (33.3) 0 1 (33.3) 12 mg 2 (66.7) 0 2 (66.7) LGS 17 5 22 6 mg 0 0 0 9 mg 1 (5.9) 0 1 (4.5) 12 mg 16 (94.1) 5 (100) 21 (95.5) DEE Other 15 4 19 6 mg 1 (6.7) 0 1 (5.3) 9 mg 2 (13.3) 0 2 (10.5) 12 mg 12 (80.0) 4 (100) 16 (84.2)
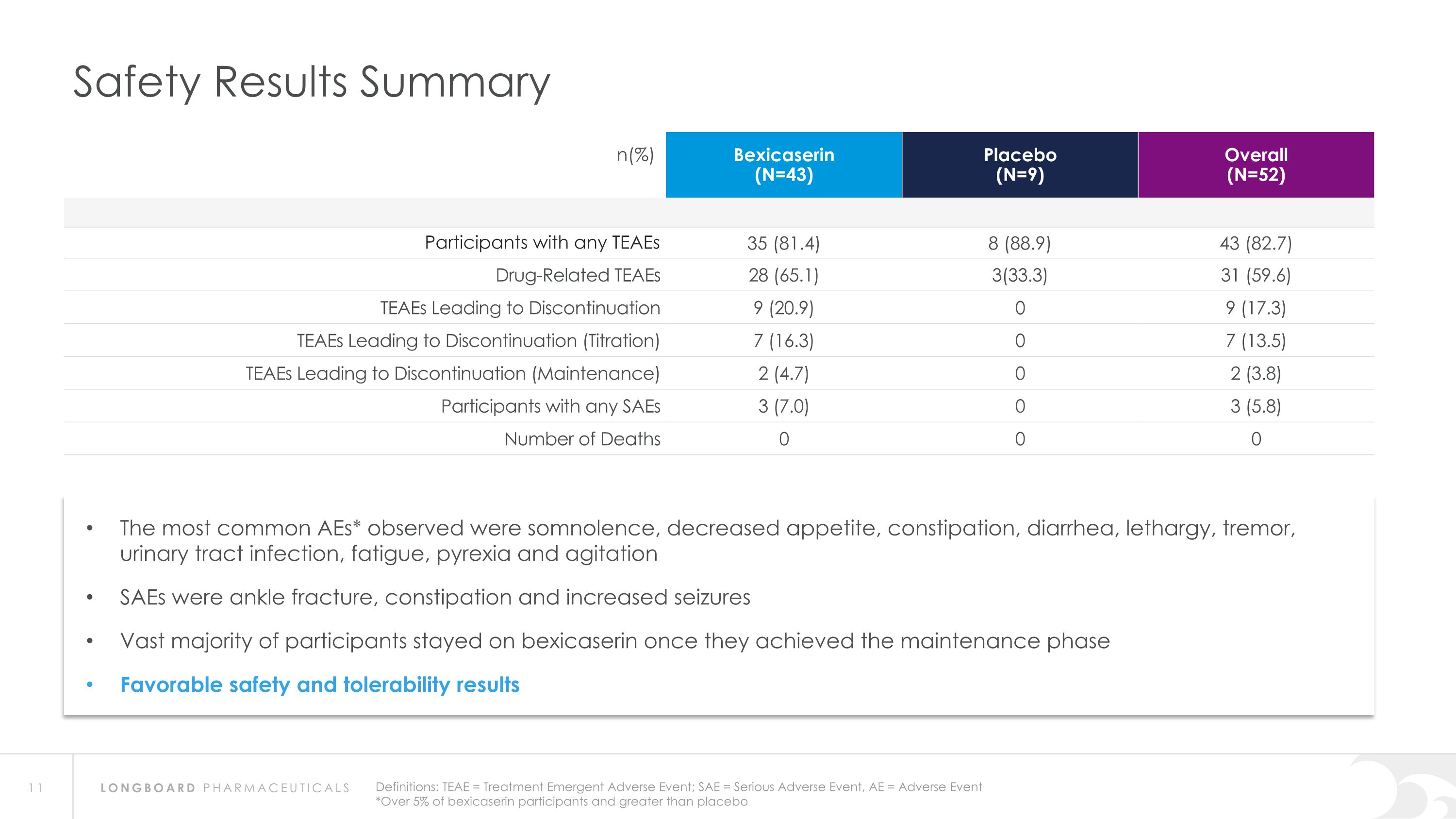
Safety Results Summary n(%) Bexicaserin (N=43) Placebo (N=9) Overall (N=52) Participants with any TEAEs 35 (81.4) 8 (88.9) 43 (82.7) Drug-Related TEAEs 28 (65.1) 3(33.3) 31 (59.6) TEAEs Leading to Discontinuation 9 (20.9) 0 9 (17.3) TEAEs Leading to Discontinuation (Titration) 7 (16.3) 0 7 (13.5) TEAEs Leading to Discontinuation (Maintenance) 2 (4.7) 0 2 (3.8) Participants with any SAEs 3 (7.0) 0 3 (5.8) Number of Deaths 0 0 0 Definitions: TEAE = Treatment Emergent Adverse Event; SAE = Serious Adverse Event, AE = Adverse Event *Over 5% of bexicaserin participants and greater than placebo The most common AEs* observed were somnolence, decreased appetite, constipation, diarrhea, lethargy, tremor, urinary tract infection, fatigue, pyrexia and agitation SAEs were ankle fracture, constipation and increased seizures Vast majority of participants stayed on bexicaserin once they achieved the maintenance phase Favorable safety and tolerability results

Topline Efficacy Results
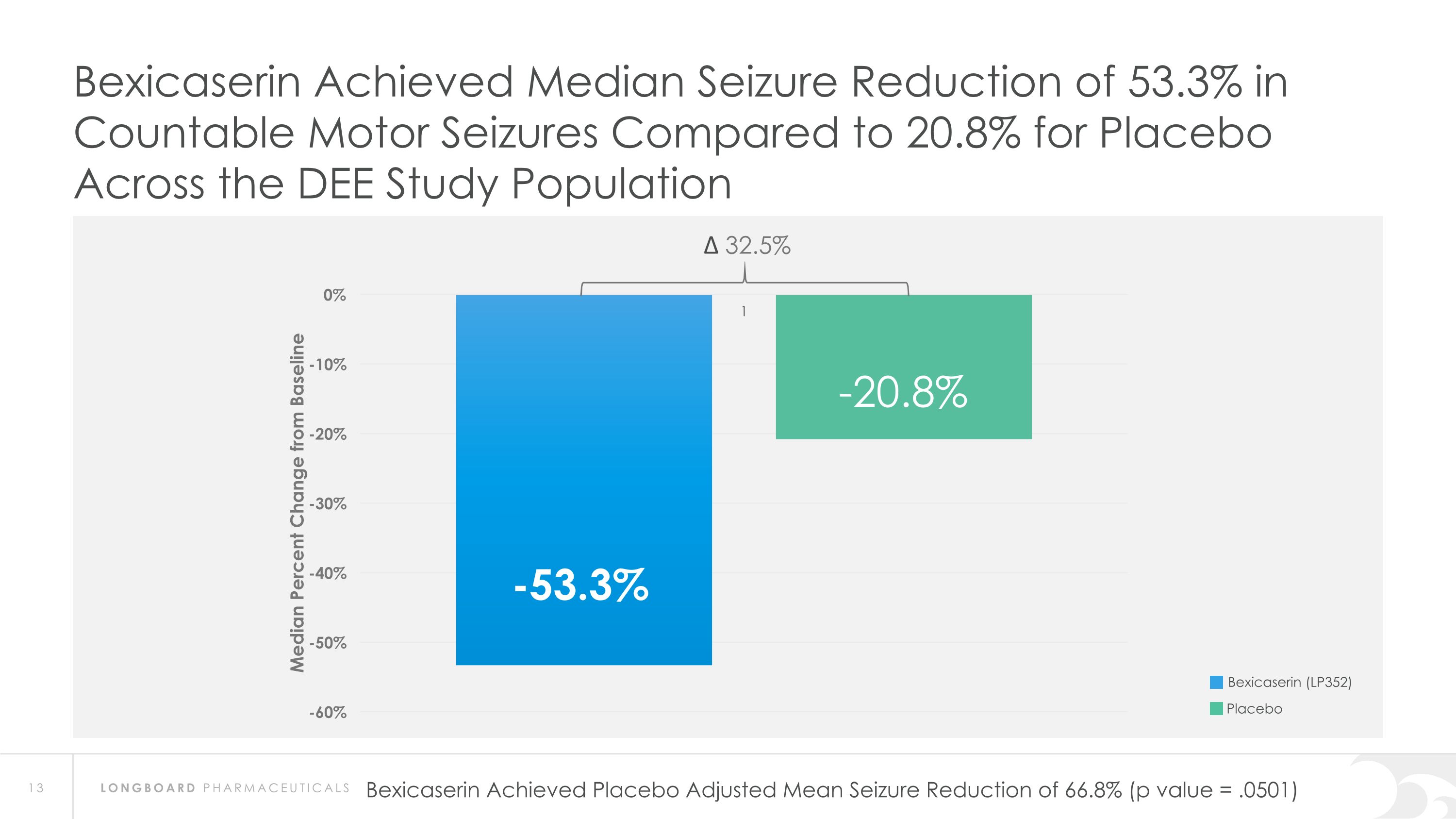
Bexicaserin Achieved Median Seizure Reduction of 53.3% in Countable Motor Seizures Compared to 20.8% for Placebo Across the DEE Study Population ∆ 32.5% Bexicaserin (LP352) Placebo Bexicaserin Achieved Placebo Adjusted Mean Seizure Reduction of 66.8% (p value = .0501)
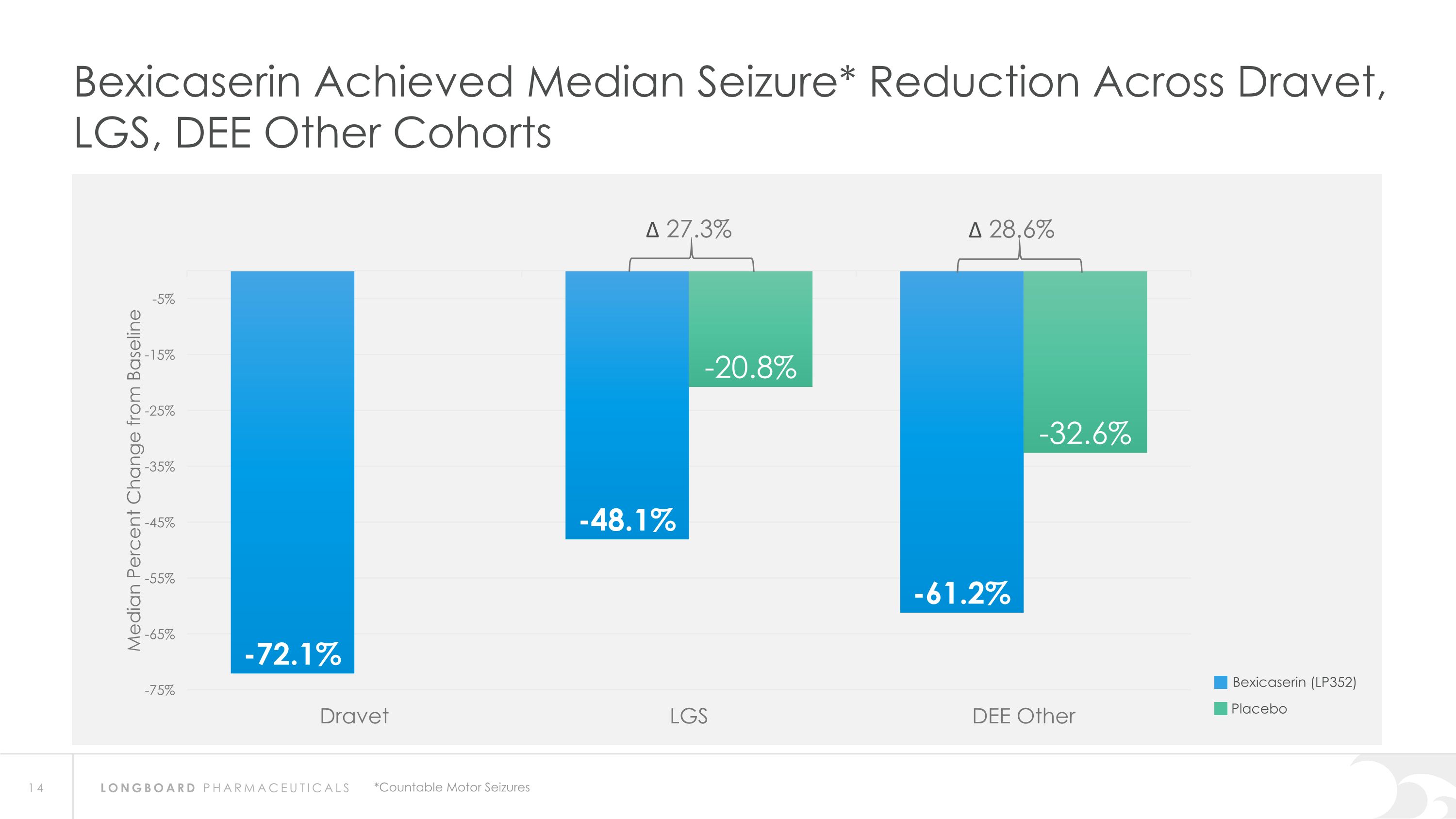
Bexicaserin Achieved Median Seizure* Reduction Across Dravet, LGS, DEE Other Cohorts Bexicaserin (LP352) Placebo ∆ 27.3% ∆ 28.6% *Countable Motor Seizures

PACIFIC Results Pave the Way for Global Phase 3 Program PACIFIC demonstrated meaningful efficacy results across a broad DEE population as well as in individuals with LGS and Dravet Bexicaserin (LP352) achieved a median percent reduction from baseline in seizure frequency during the treatment period of: 53.3% in broad DEE population (32.5% placebo-adjusted) 72.1% in Dravet cohort 48.1% in LGS cohort (27.3% placebo-adjusted) 61.2% in DEE Other cohort (28.6% placebo-adjusted) Results were demonstrated on top of a contemporary polytherapy background with multiple ASMs including cannabidiol (32.7% of participants were receiving cannabidiol) *Definitions: ASMs = Anti-Seizure Medications; UGT = Uridine Diphosphate Glucuronosyltransferase Favorable safety and tolerability results No echocardiograms required in PACIFIC study Metabolized via UGT pathway – potentially reduces risk of Drug-Drug Interactions 100% of PACIFIC participants who completed the study entered the Open Label Extension Study Awaiting analysis of the full PACIFIC dataset Utilizing key learnings for incorporation into the global Phase 3 program

Summary & Next Steps
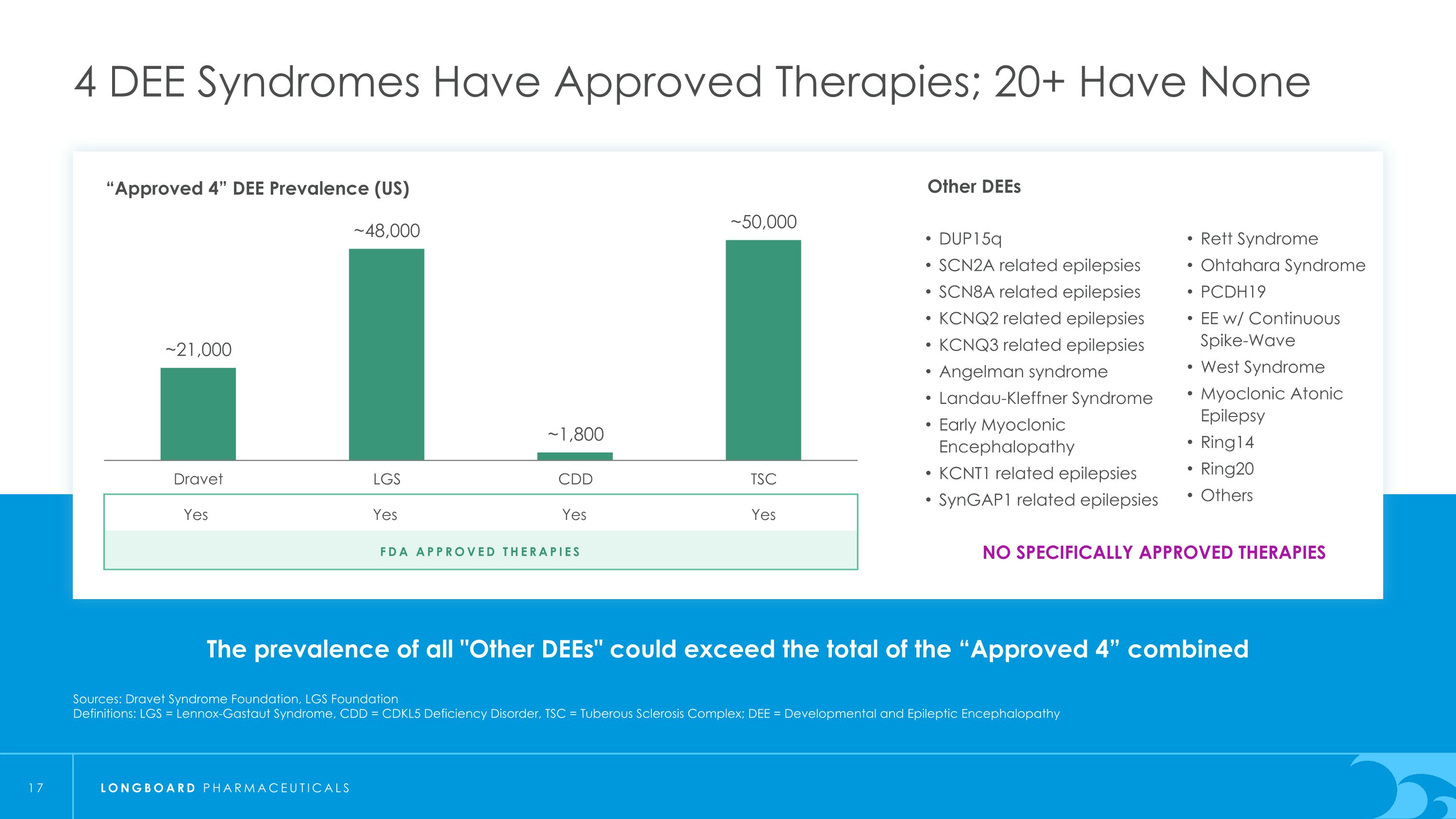
The prevalence of all "Other DEEs" could exceed the total of the “Approved 4” combined 4 DEE Syndromes Have Approved Therapies; 20+ Have None Sources: Dravet Syndrome Foundation, LGS Foundation Definitions: LGS = Lennox-Gastaut Syndrome, CDD = CDKL5 Deficiency Disorder, TSC = Tuberous Sclerosis Complex; DEE = Developmental and Epileptic Encephalopathy “Approved 4” DEE Prevalence (US) FDA Approved Therapies Yes Yes Yes Yes NO SPECIFICALLY APPROVED THERAPIES Other DEEs DUP15q SCN2A related epilepsies SCN8A related epilepsies KCNQ2 related epilepsies KCNQ3 related epilepsies Angelman syndrome Landau-Kleffner Syndrome Early Myoclonic Encephalopathy KCNT1 related epilepsies SynGAP1 related epilepsies Rett Syndrome Ohtahara Syndrome PCDH19 EE w/ Continuous Spike-Wave West Syndrome Myoclonic Atonic Epilepsy Ring14 Ring20 Others
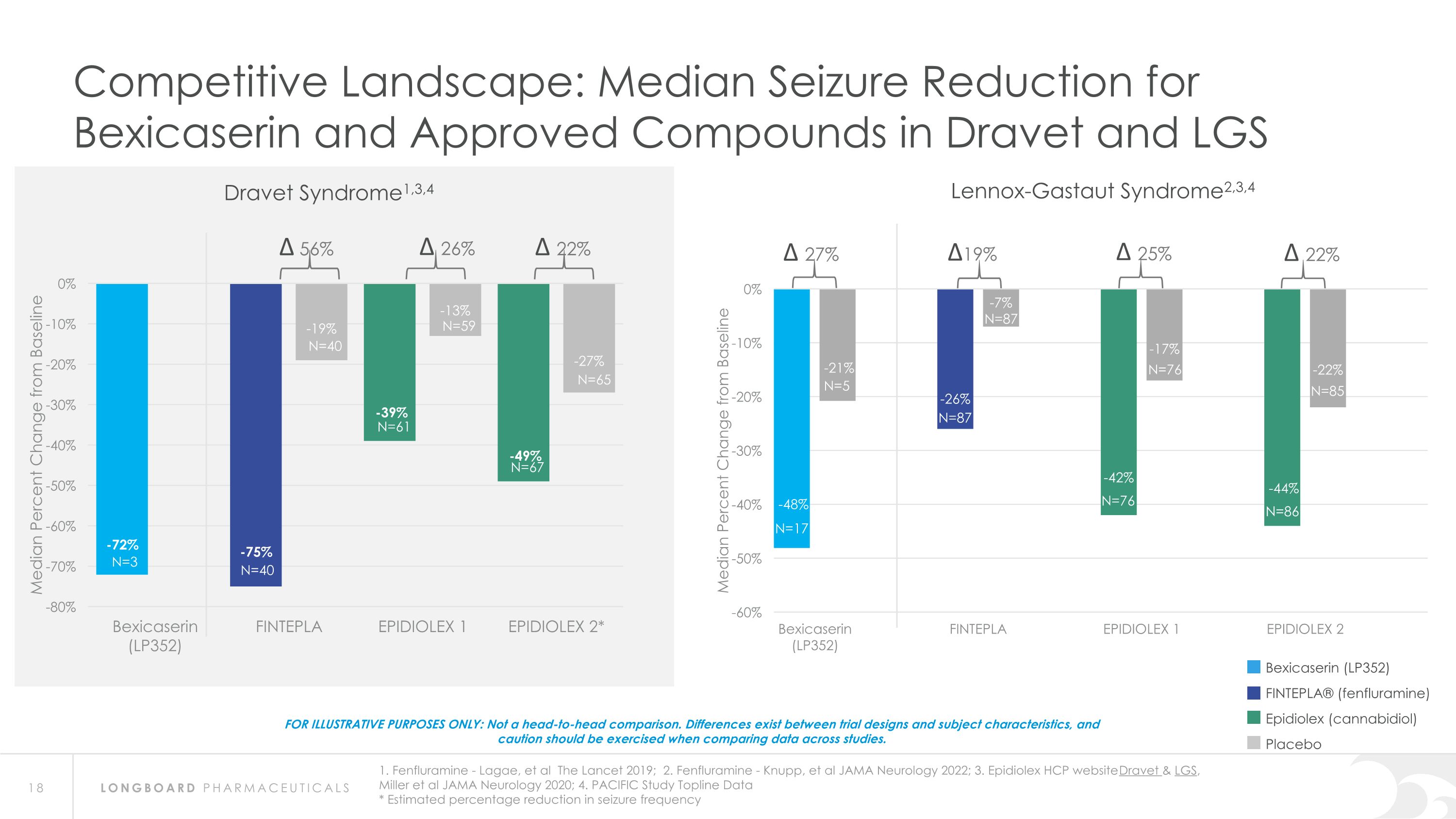
Competitive Landscape: Median Seizure Reduction for Bexicaserin and Approved Compounds in Dravet and LGS FOR ILLUSTRATIVE PURPOSES ONLY: Not a head-to-head comparison. Differences exist between trial designs and subject characteristics, and caution should be exercised when comparing data across studies. ∆ 56% ∆ 26% ∆ 22% ∆ 27% ∆19% Dravet Syndrome1,3,4 Lennox-Gastaut Syndrome2,3,4 Bexicaserin (LP352) FINTEPLA® (fenfluramine) Epidiolex (cannabidiol) Placebo 1. Fenfluramine - Lagae, et al The Lancet 2019; 2. Fenfluramine - Knupp, et al JAMA Neurology 2022; 3. Epidiolex HCP website Dravet & LGS, Miller et al JAMA Neurology 2020; 4. PACIFIC Study Topline Data * Estimated percentage reduction in seizure frequency N=3 N=40 N=61 N=67 N=40 N=59 N=65 N=17 N=87 N=76 N=86 N=5 N=76 N=85 N=87 ∆ 25% ∆ 22%
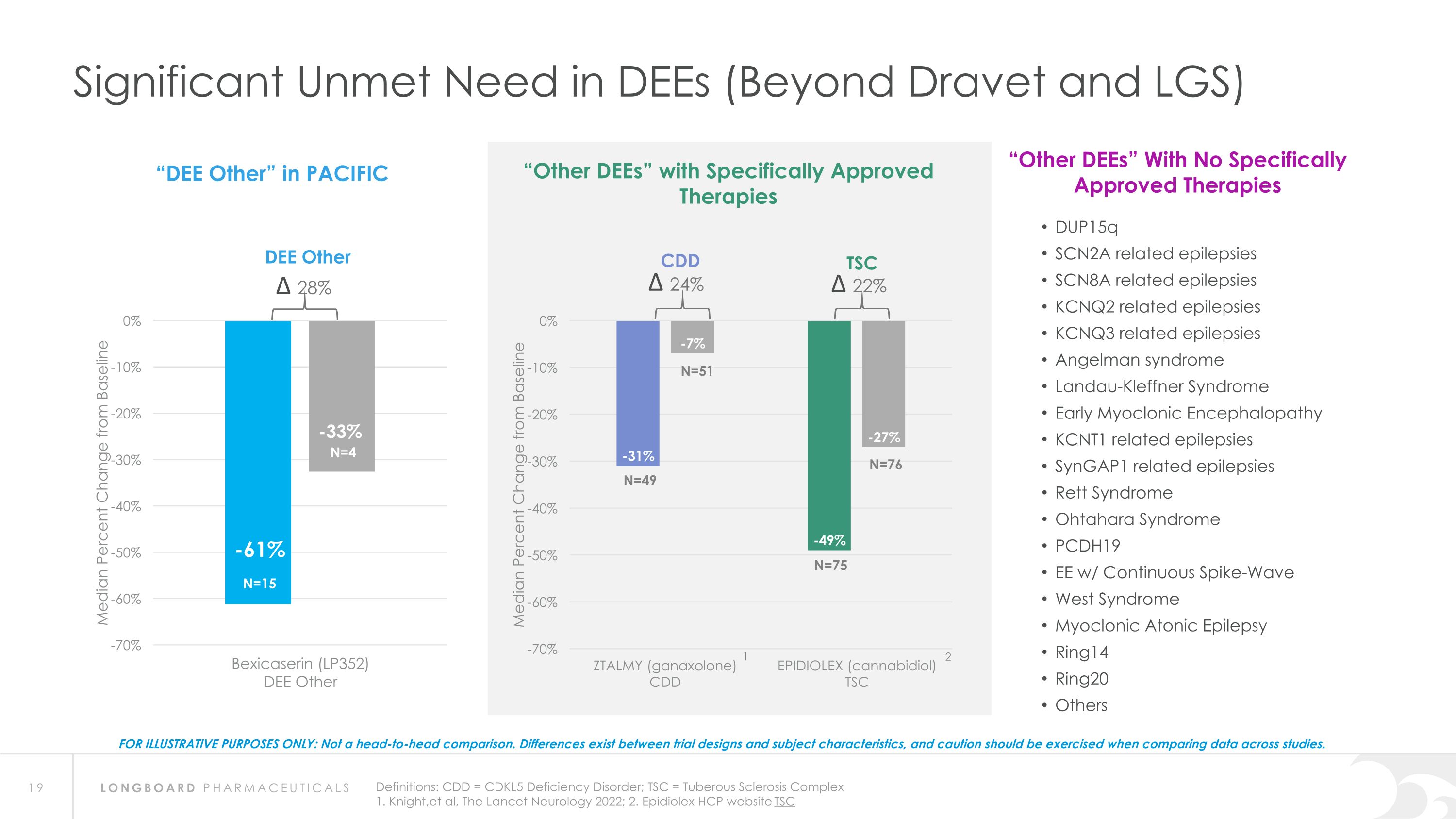
Significant Unmet Need in DEEs (Beyond Dravet and LGS) Definitions: CDD = CDKL5 Deficiency Disorder; TSC = Tuberous Sclerosis Complex 1. Knight,et al, The Lancet Neurology 2022; 2. Epidiolex HCP website TSC FOR ILLUSTRATIVE PURPOSES ONLY: Not a head-to-head comparison. Differences exist between trial designs and subject characteristics, and caution should be exercised when comparing data across studies. DUP15q SCN2A related epilepsies SCN8A related epilepsies KCNQ2 related epilepsies KCNQ3 related epilepsies Angelman syndrome Landau-Kleffner Syndrome Early Myoclonic Encephalopathy KCNT1 related epilepsies SynGAP1 related epilepsies Rett Syndrome Ohtahara Syndrome PCDH19 EE w/ Continuous Spike-Wave West Syndrome Myoclonic Atonic Epilepsy Ring14 Ring20 Others 2 “DEE Other” in PACIFIC “Other DEEs” With No Specifically Approved Therapies DEE Other “Other DEEs” with Specifically Approved Therapies TSC CDD 1 N=15 N=4 N=49 N=75 N=51 N=76 ∆ 28% ∆ 24% ∆ 22%

Bexicaserin Has the Potential to Change the DEE Landscape *In the PACIFIC Study **Composition of matter through 2036 with potential for PTE / PTA (2041) Bexicaserin provides the cornerstone to potentially build a world-class epilepsy franchise Studies to date highlight bexicaserin as potentially best-in-class Composition of matter IP protection through 2041** provides the opportunity to maximize the full potential of LP352 Rapidly moving forward with preparations for a global Phase 3 program 53.3% Median Reduction in Seizures* 72.1% Dravet 48.1% LGS 61.2% DEE Other

Thank you Nasdaq: LBPH IR@longboardpharma.com