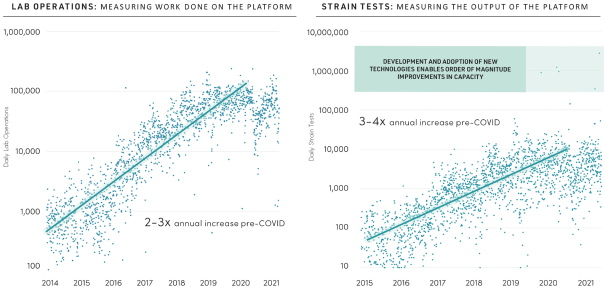
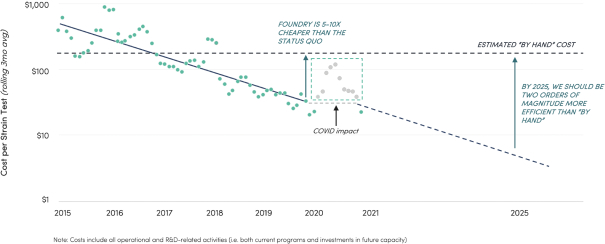
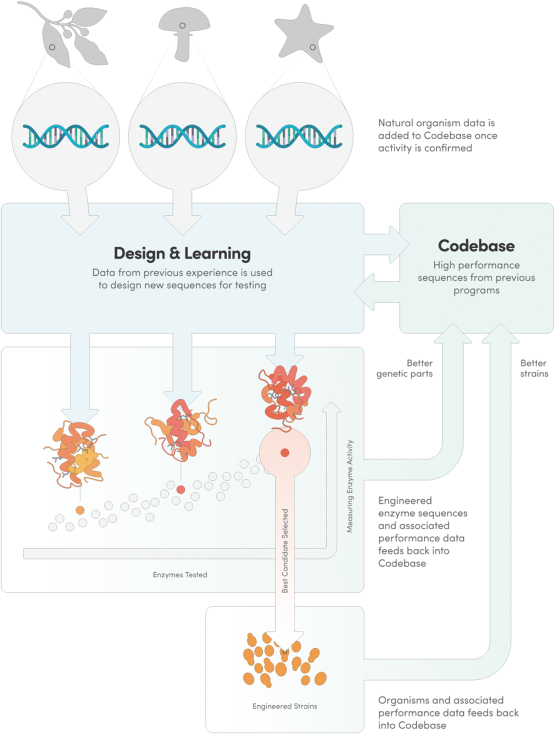
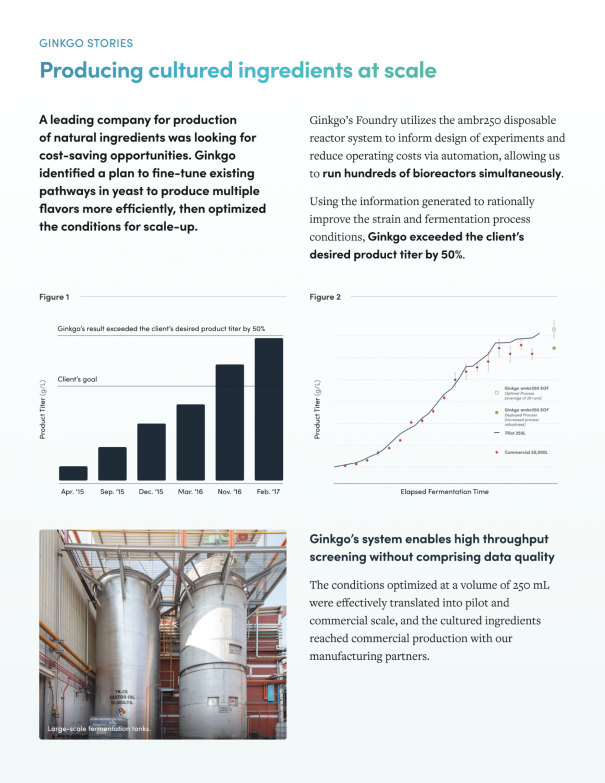
The information in this preliminary prospectus is not complete and may be changed. These securities may not be issued until the registration statement filed with the U.S. Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell these securities and does not constitute the solicitation of an offer to buy these securities in any jurisdiction where the offer or sale is not permitted.
SUBJECT TO COMPLETION, DATED NOVEMBER 23, 2021
PRELIMINARY PROSPECTUS

GINKGO BIOWORKS HOLDINGS, INC.
Prospectus for 84,346,092 Shares of
Class A Common Stock, 51,824,925 Shares of
Class A Common Stock Underlying Warrants and 17,325,000 Private Placement Warrants
of Ginkgo Bioworks Holdings, Inc.
This prospectus relates to (i) the resale of 84,346,092 shares of our Class A common stock, par value $0.0001 per share (“Class A common stock”), issued in connection with the Merger (as defined below), including 52,849,353 earn-out shares of New Ginkgo Class A common stock (the “Earn-Out Shares”), which are subject to forfeiture to the extent that certain vesting conditions are not satisfied on or before the fifth anniversary of the closing of the Merger, by certain of the selling securityholders named in this prospectus (each a “Selling Securityholder” and, collectively, the “Selling Securityholders”), (ii) the issuance by us and the resale of up to an aggregate of up to 17,325,000 shares of Class A common stock that are issuable upon the exercise of 17,325,000 private placement warrants (the “Private Placement Warrants”) originally issued in a private placement in connection with the IPO (as defined below) of Soaring Eagle Acquisition Corp., a Delaware corporation (“SRNG”), at an exercise price of $11.50 per share of Class A common stock, (iii) the issuance by us of up to 34,499,925 shares of Class A common stock that are issuable upon the exercise of 34,499,925 warrants issued in connection with the IPO (the “Public Warrants” and, together with the Private Placement Warrants, the “Warrants”) and the resale by the Selling Securityholders of an aggregate of up to 17,325,000 Private Placement Warrants.
On September 16, 2021, we consummated the transactions contemplated by that certain agreement and plan of merger, dated as of May 11, 2021, as amended on May 14, 2021 (the “Merger Agreement”), by and among SRNG, SEAC Merger Sub Inc., a Delaware corporation (“Merger Sub”), and Ginkgo Bioworks, Inc., a Delaware corporation (“Ginkgo”). As contemplated by the Merger Agreement, SRNG effected a deregistration under the Cayman Islands Companies Act (As Revised) and a domestication under Section 388 of the Delaware General Corporation Law, as amended (the “DGCL”), pursuant to which SRNG’s jurisdiction of incorporation was changed from the Cayman Islands to the State of Delaware (the “Domestication”), and, on the terms and subject to the conditions set forth in the Merger Agreement and in accordance with the DGCL, Merger Sub merged with and into Ginkgo, with Ginkgo surviving the merger as a wholly owned subsidiary of SRNG (the “Merger” and, together with the Domestication, the “Business Combination”). In addition, in connection with the consummation of the Business Combination, SRNG was renamed “Ginkgo Bioworks Holdings, Inc.” As used herein, “New Ginkgo” refers to SRNG after the consummation of the Business Combination.
This prospectus provides you with a general description of such securities and the general manner in which the Selling Securityholders may offer or sell the securities. More specific terms of any securities that the Selling Securityholders may offer or sell may be provided in a prospectus supplement that describes, among other things, the specific amounts and prices of the securities being offered and the terms of the offering. The prospectus supplement may also add, update or change information contained in this prospectus.
We will not receive any proceeds from the sale of shares of Class A common stock or Private Placement Warrants by the Selling Securityholders pursuant to this prospectus, except with respect to amounts received by us upon exercise of the Warrants. However, we will pay the expenses, other than any underwriting discounts and commissions, associated with the sale of securities pursuant to this prospectus.
We are registering certain of the securities for resale pursuant to the Selling Securityholders’ registration rights under certain agreements between us and the Selling Securityholders. Our registration of the securities covered by this prospectus does not mean that either we or the Selling Securityholders will issue, offer or sell, as applicable, any of the securities. The Selling Securityholders may offer and sell the securities covered by this prospectus in a number of different ways and at varying prices. We provide more information about how the Selling Securityholders may sell the shares or Private Placement Warrants in the section entitled “Plan of Distribution.”
You should read this prospectus and any prospectus supplement or amendment carefully before you invest in our securities.
Our Class A common stock and Public Warrants are listed on the NYSE under the symbols “DNA” and “DNA.WS,” respectively. On November 22, 2021, the closing price of our Class A common stock was $13.49 and the closing price for our Public Warrants was
$3.58.
We are an “emerging growth company” under applicable federal securities laws and will be subject to reduced public company reporting requirements.
INVESTING IN OUR SECURITIES INVOLVES RISKS THAT ARE DESCRIBED IN THE “RISK FACTORS” SECTION BEGINNING ON PAGE 10 OF THIS PROSPECTUS.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of the securities to be issued under this prospectus or determined if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
The date of this prospectus is , 2021.