Exhibit 99.1

Fusion Pharmaceuticals to Present Interim Data from Phase 1 Trial of FPI-1434 in
Patients with Solid Tumors Expressing IGF-1R
Preliminary Clinical Data with Cold/Hot Dosing Regimen Demonstrate Potential to Improve Therapeutic Index and Safety Profile
Company Prioritizing Cold/Hot Dosing Regimen in Phase 1 Trial; Cohort 2 in this Arm is Currently Enrolling
Data to be Presented at the Society of Nuclear Medicine and Molecular Imaging (SNMMI) Annual Meeting
Fusion to Host Conference Call Tomorrow, Tuesday, June 27th at 4:00 p.m. ET/3:00 p.m. CT
Hamilton, ON & Boston, MA, June 26, 2023 – Fusion Pharmaceuticals Inc. (Nasdaq: FUSN), a clinical-stage oncology company focused on developing next-generation radiopharmaceuticals as precision medicines, today announced the presentation of interim data from the dose escalation portion of the Phase 1 trial of [225Ac]-FPI-1434 (FPI-1434) in patients with solid tumors expressing IGF-1R. The data will be presented by Neeta Pandit-Taskar, M.D., of Memorial Sloan Kettering Cancer Center, tomorrow at the Society of Nuclear Medicine and Molecular Imaging (SNMMI) Annual Meeting and will include molecular imaging, safety and pharmacokinetics (PK) from two dosing regimens: one with FPI-1434 alone (“hot only”), and another in which a small dose of cold antibody (naked IGF-1R antibody without the isotope) is administered prior to each dose of FPI-1434 (“cold/hot”). The poster presentation is available via the SNMMI Annual Meeting mobile application.
“Antibody drug conjugates (ADCs) have shown tremendous success recently, driven in part by the benefits of using more potent payloads. We believe that the high potency of alpha emitters in place of conventional chemical toxins represents the evolution of the ADC field and is a significant and underexploited opportunity. While much is known about optimal dosing levels and paradigms for small molecule radiopharmaceuticals against well-known targets like PSMA, there is comparatively less experience for antibody-based alpha therapies in solid tumors. Consequently, in the Phase 1 study of FPI-1434, we undertook a stepwise dose exploration, in patients with various cancer types expressing IGF-1R, that included three stages: (i) hot only regimen in a single ascending dose escalation; (ii) hot only regimen in a multiple ascending dose escalation; and (iii) cold/hot regimen in a multiple ascending dose escalation. Learnings from this multi-part study have taken us a step further towards unlocking the potential of antibody-based TATs as next-generation ADCs,” said Chief Executive Officer John Valliant, Ph.D.
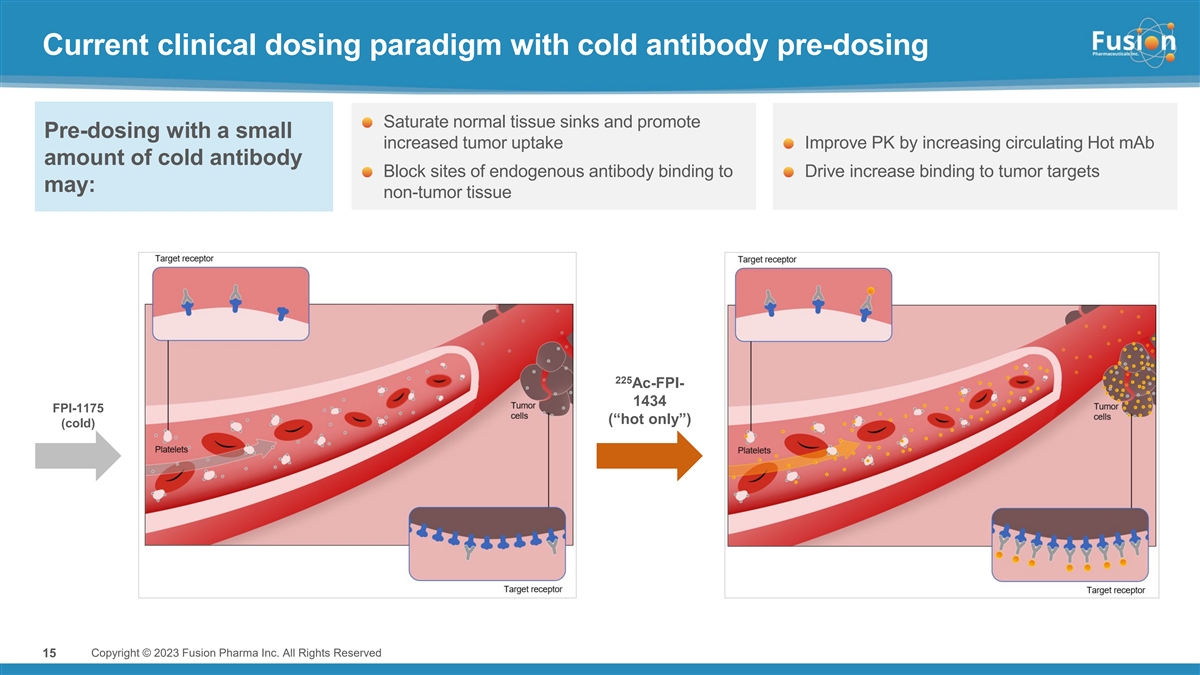
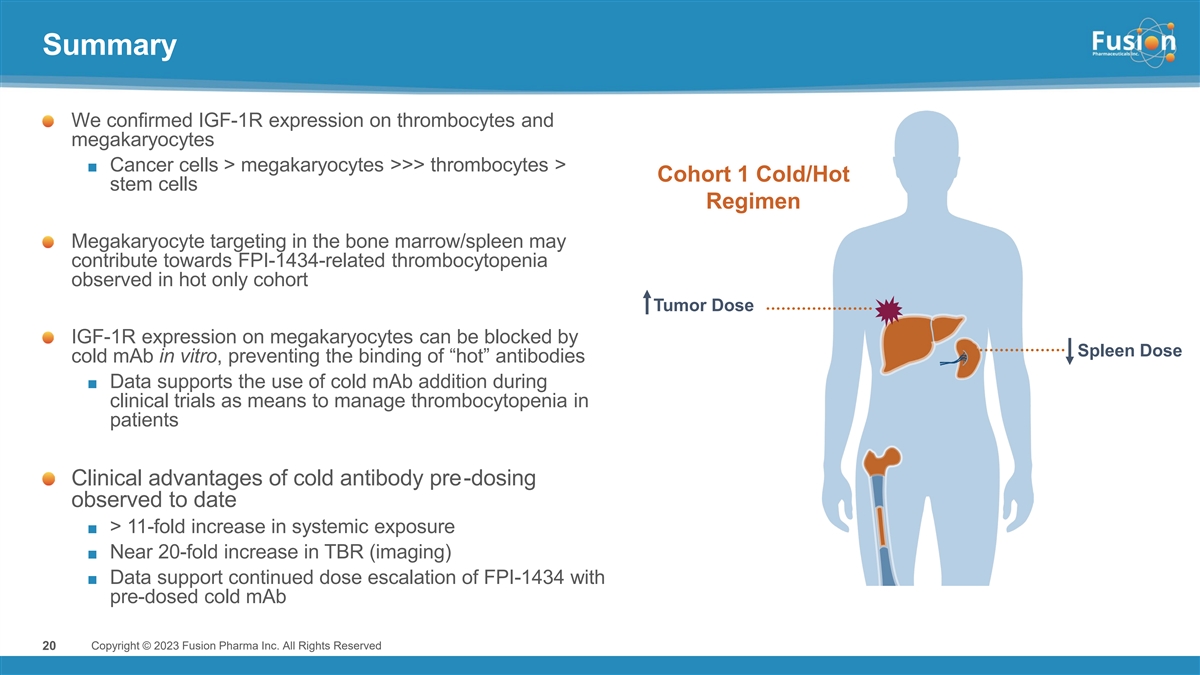
Dr. Valliant continued, “We are pleased to report data and key learnings from our first antibody targeted alpha therapy (TAT) program which showed pre-administration of cold antibody prior to administering radiolabeled FPI-1434 has the potential to significantly enhance the therapeutic index by driving more active drug to tumor sites with an improved safety profile compared to hot-only dosing. In addition, with pre-administration of cold antibody, only a fraction of the injected amount of radiation is needed to achieve comparable exposures to hot-only cohorts. Results show the pharmacokinetic profile of 15 kBq/kg in the cold/hot regimen is comparable to 40 kBq/kg hot only, with a safety profile that made it possible to deliver up to five cycles thus far. With encouraging safety, dosimetry, and PK results, as well as stable disease observed in two heavily pretreated patients despite a low dose, we are continuing evaluation of the cold/hot regimen at a higher dose level in Cohort 2. We expect to share those results around the end of 2023.”
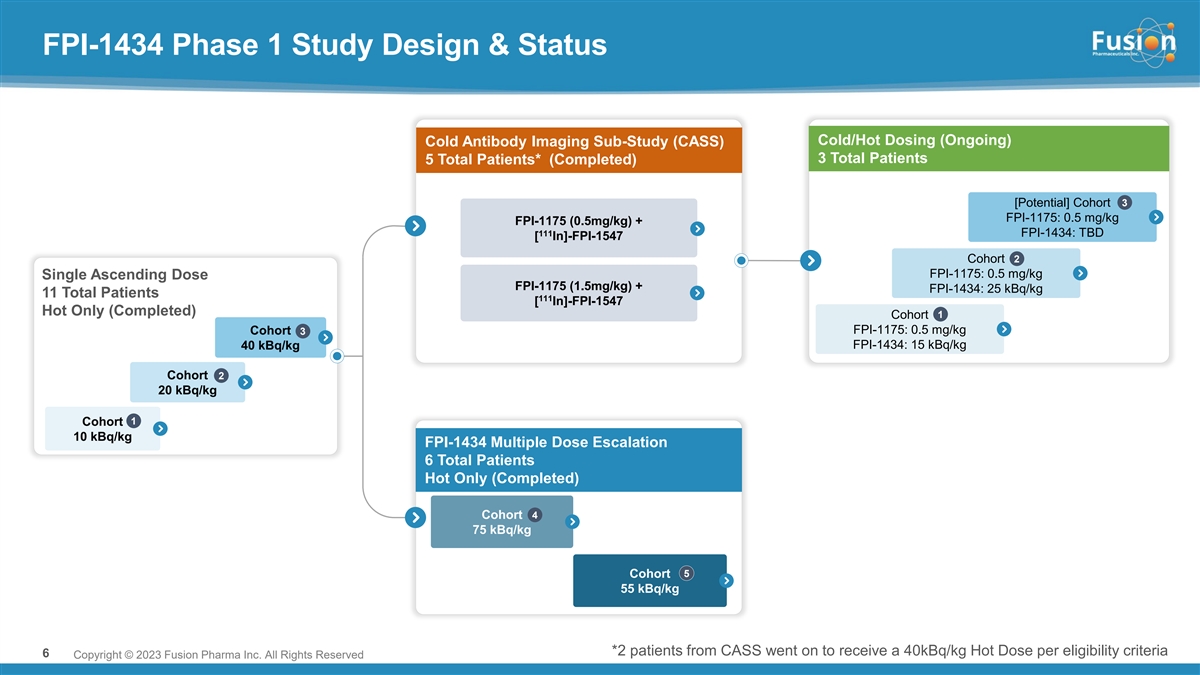
The Phase 1, multi-center, open-label clinical trial is designed to investigate the safety, tolerability and pharmacokinetics of FPI-1434 in patients with solid tumors expressing IGF-1R. The trial is also designed to establish the maximum tolerated dose for FPI-1434 and the recommended Phase 2 dose. As part of the precision medicine approach, prior to receiving the therapeutic injection of FPI-1434, patients are administered an indium-111 imaging analogue, [111In]-FPI-1547 (FPI-1547). The images collected are used to confirm the presence of tumor uptake and ensure that estimated radiation doses to organs and tissues are below protocol-specified safety limits.
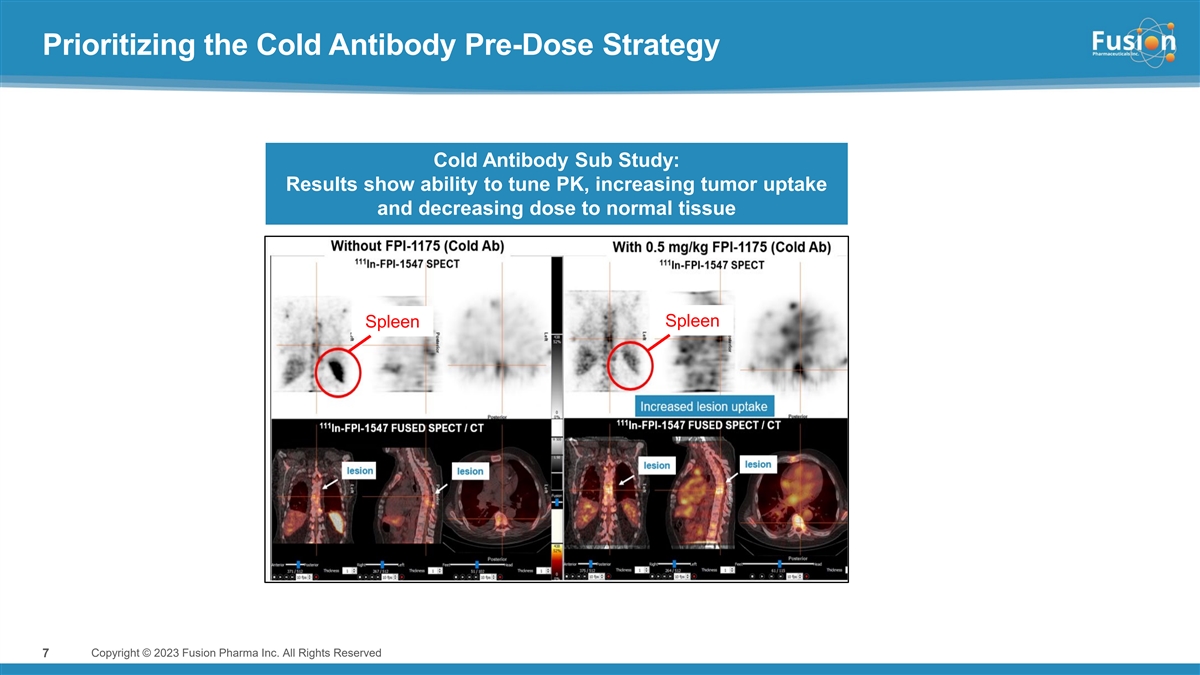
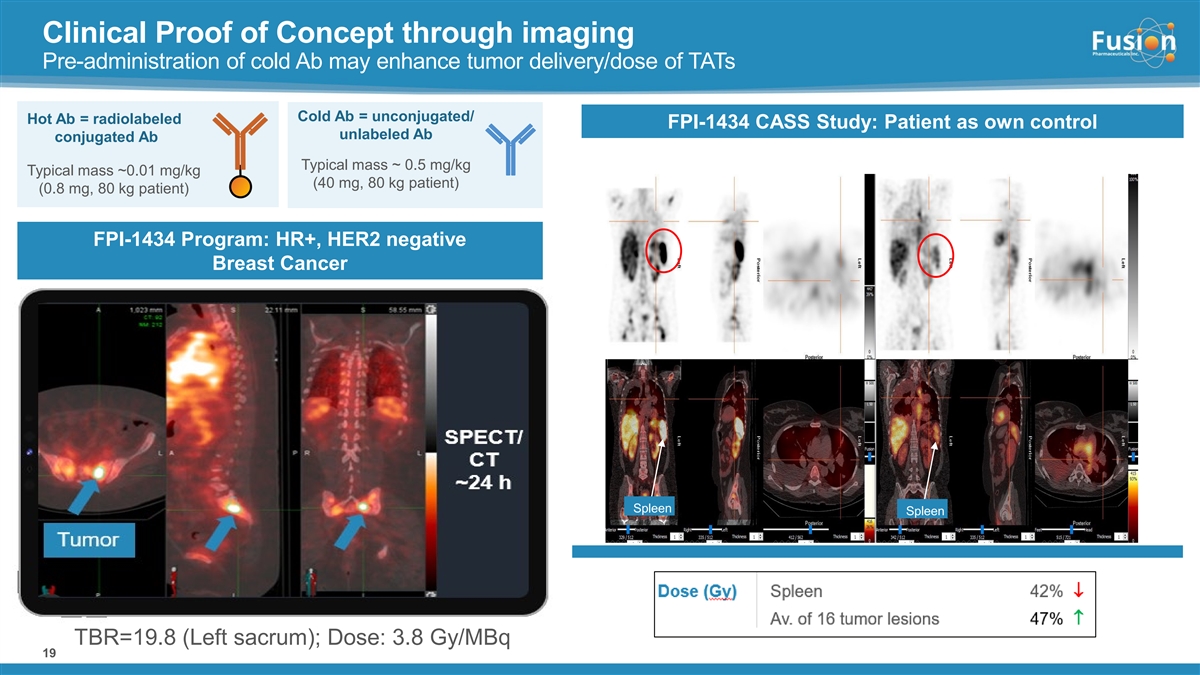
Based on results from an imaging sub-study evaluating pre-administration of cold antibody prior to each dose of FPI-1547 that demonstrated a favorable gain in tumor lesion uptake versus normal tissue, the Company amended the Phase 1 trial protocol to evaluate both the hot only and cold/hot dosing regimens.
Cold/Hot Dosing Regimen
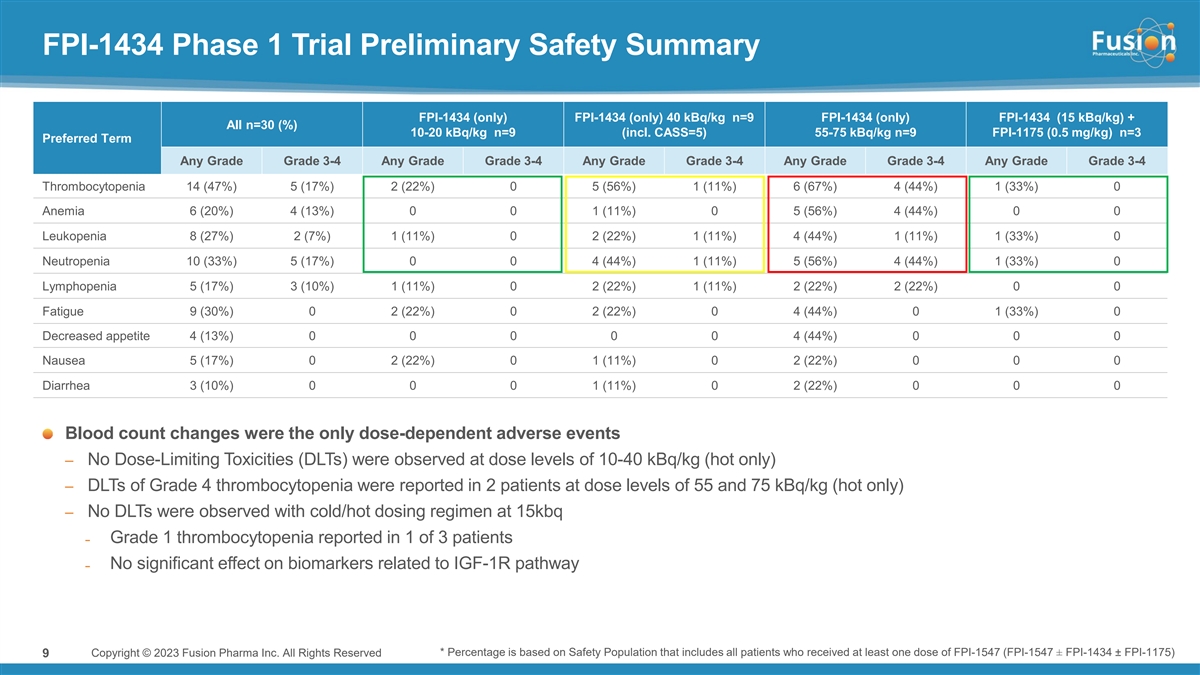
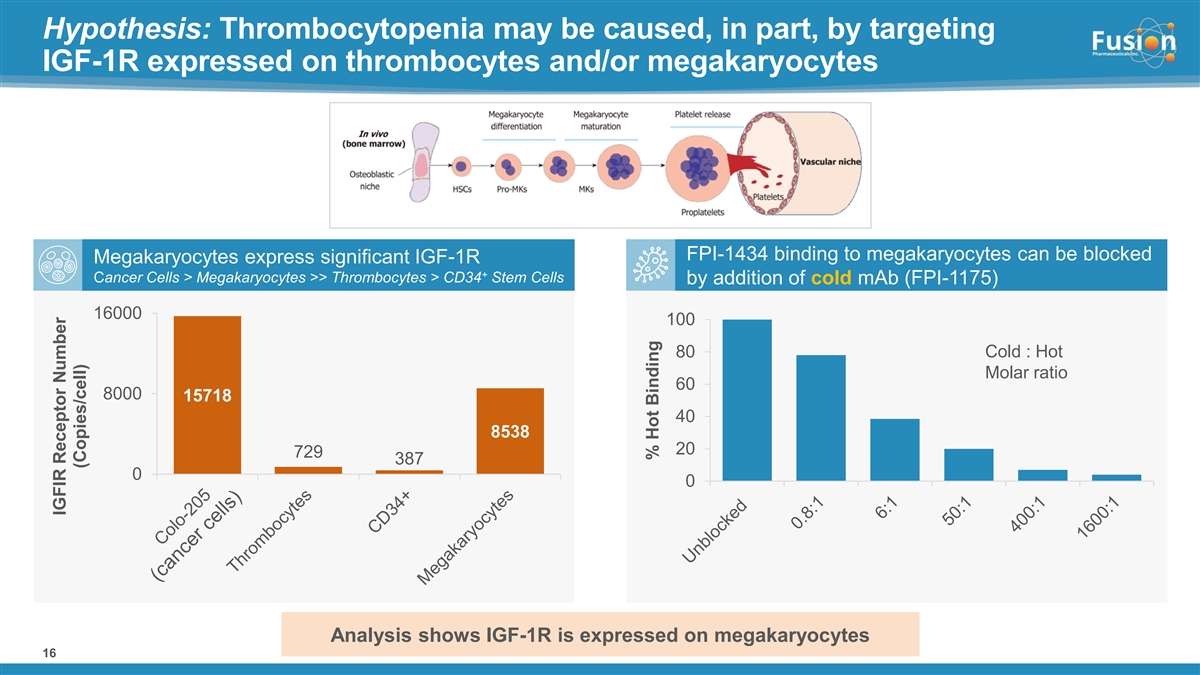
In the cold/hot arm, three patients were dosed in Cohort 1 at a dose of 15 kBq/kg following pre-administration of cold antibody. In Cohort 1, cold/hot dosing was observed to be safe with no treatment-related serious adverse events (SAEs) or dose limiting toxicities (DLTs). Absorbed dose to critical organs (kidney, liver and lungs) in this dosing regimen were less than 7% of defined limits, supporting continued dose escalation.
Pre-administration of cold antibody demonstrated improved tumor uptake while also reducing hematological toxicity observed in the hot only dosing arm. When normalized to 15 kBq/kg, the average lesion absorbed dose and dose/volume in the cold/hot arm were nearly double the level compared to hot only. Further, 15 kBq/kg in the cold/hot dosing arm shows comparable systemic exposure to approximately 40 kBq/kg of a hot only dose, but shows an improved hematological safety profile as measured by changes in platelet count.
Two heavily pre-treated patients from the cold/hot dosing arm received three and five cycles of treatment, with both achieving durable stable disease as their best response. The dose level from Cohort 1, while sufficient to demonstrate marked differences in tumor uptake and achieve stable disease, was expected to be sub-optimal with respect to efficacy.
Fusion is currently enrolling Cohort 2 in the cold/hot dosing regimen at 25 kBq/kg and expects to report data from this cohort around year-end 2023.
Hot Only Dosing Regimen
In the hot only multiple ascending arm, six patients were dosed across two cohorts. Dose-dependent decrease in blood counts, particularly thrombocytopenia, was identified as the most common type of adverse event related to FPI-1434, with Grade 4 thrombocytopenia observed at 75 kBq/kg and 55 kBq/kg. The Company has discontinued dosing in the hot only dosing arm and no longer plans to use this method of FPI-1434 administration.
“Given the known expression of IGF-1R on multiple cancers, Fusion believes in the opportunity for FPI-1434 to treat a broad array of cancer types,” said Chief Medical Officer Dmitri Bobilev, M.D. “We believe that thrombocytopenia observed at higher dose levels in the hot only part of the trial, may result at least in part from direct effect of FPI-1434 on IGF-1R-expressing megakaryocytes and from Fc-mediated binding in normal tissues. The ability to use pre-administration of cold antibody to block the IGF-1R receptors on normal cells and non-target specific binding and deliver higher doses of radioactivity to tumor cells is highly encouraging. We look forward to sharing results from the second cohort.”
Following the conclusion of the SNMMI Annual Meeting, the presentation can be found at https://fusionpharma.com/fusion-scientific-presentations/.
Fusion Conference Call Information
Fusion will host a live conference call and webcast tomorrow beginning at 4:00 p.m. ET/3:00 p.m. CT to discuss the data presentation. To access the live call, please dial 1-877-870-4263 (U.S.), 1-855-669-9657 (Canada) or 1-412-317-0790 (international) and reference Fusion Pharmaceuticals. A webcast of the conference call will be available under “Events and Presentations” in the Investors & Media section of Fusion’s website at https://ir.fusionpharma.com/overview. The archived webcast will be available on Fusion’s website shortly after the conclusion of the call and will be available for 90 days following the event.
About FPI-1434
FPI-1434 is a radioimmunoconjugate designed to target and deliver alpha emitting medical isotopes to cancer cells expressing IGF-1R, a receptor that is overexpressed on many tumor types. FPI-1434 utilizes Fusion’s Fast-Clear linker to connect a human monoclonal antibody that targets IGF-1R with actinium-225, a powerful alpha-emitting isotope with desirable half-life and decay chain properties.
About Fusion
Fusion Pharmaceuticals is a clinical-stage oncology company focused on developing next-generation radiopharmaceuticals as precision medicines. Fusion connects alpha particle emitting isotopes to various targeting molecules to selectively deliver the alpha emitting payloads to tumors. Fusion’s clinical portfolio includes: FPI-2265 targeting prostate specific membrane antigen (PSMA) for metastatic castration resistant prostate cancer currently in a Phase 2 trial with data from 20-30 patients anticipated in the first quarter of 2024; FPI-1434 targeting insulin-like growth factor 1 receptor currently in a Phase 1 trial; and FPI-2059, a small molecule targeting neurotensin receptor 1 (NTSR1), currently in a Phase 1 trial. In addition to a robust proprietary pipeline, Fusion has a collaboration with AstraZeneca to jointly develop novel targeted alpha therapies (TATs) and combination programs between Fusion’s TATs and AstraZeneca’s DNA Damage Response Inhibitors (DDRis) and immuno-oncology agents. The Company recently received IND clearance for FPI-2068, the first novel TAT under the collaboration, which targets EGFR-cMET. Fusion has also entered into a collaboration with Merck to evaluate FPI-1434 in combination with Merck’s KEYTRUDA® (pembrolizumab) in patients with solid tumors expressing IGF-1R. To support Fusion’s growing pipeline of TATs, the Company has signed strategic actinium supply agreements with TRIUMF, Niowave, Inc. and BWXT Medical.
Forward-Looking Statements
This press release contains “forward-looking statements” for purposes of the safe harbor provisions of The Private Securities Litigation Reform Act of 1995, including but not limited to the statements regarding Fusion Pharmaceuticals Inc.’s (the “Company”) future business. For this purpose, any statements contained herein that are not statements of historical fact may be deemed forward-looking statements. Without limiting the foregoing, the words “expect,” “plans,” “anticipates,” “intends,” “will,” and similar expressions are also intended to identify forward-looking statements, as are expressed or implied statements with respect to the Company’s potential drug candidates, including any expressed or implied statements regarding the successful development of its product candidates. Actual results may differ materially from those indicated by such forward-looking statements as a result of risks and uncertainties, including but not limited to the following: there can be no guarantees that the Company will advance any clinical product candidate or other component of its potential pipeline to the clinic, to the regulatory process or to commercialization; management’s expectations could be affected by unexpected patient recruitment delays, regulatory actions or delays, or changes in the competitive landscape; uncertainties relating to, or unsuccessful results of, clinical trials, including additional data relating to the ongoing clinical trials evaluating its product candidates; and competition in general. Such forward-looking statements involve known and unknown risks, uncertainties and other factors that may cause actual results to be materially different from any future results, performance or achievements expressed or implied by such statements. These and other risks which may impact management’s expectations are described in greater detail under the heading “Risk Factors” in the Company’s quarterly report on Form 10-Q for the year ended March 31, 2023, as filed with the SEC and in any subsequent periodic or current report that the Company files with the SEC. All forward-looking statements reflect the Company’s estimates only as of the date of this release (unless another date is indicated) and should not be relied upon as reflecting the Company’s views, expectations or beliefs at any date subsequent to the date of this release. While Fusion may elect to update these forward-looking statements at some point in the future, it specifically disclaims any obligation to do so, even if the Company’s estimates change.
Investors and others should note that Fusion communicates with its investors and the public using the Fusion website, www.fusionpharma.com, including, but not limited to, company disclosures, investor presentations, SEC filings, and press releases. The information that Fusion posts on this website could be deemed to be material information. As a result, Fusion encourages investors, media and others interested to review the information that Fusion posts there on a regular basis.
Contact:
Amanda Cray
Senior Director of Investor Relations & Corporate Communications
(617) 967-0207
cray@fusionpharma.com
# # #