|
Delaware
|
| |
3844
|
| |
84-4618156
|
|
(State or other jurisdiction of
incorporation or organization)
|
| |
(Primary Standard Industrial
Classification Code Number)
|
| |
(I.R.S. Employer
Identification Number)
|
|
Large accelerated filer
|
| |
☐
|
| |
Accelerated filer
|
| |
☐
|
|
Non-accelerated filer
|
| |
☒
|
| |
Smaller reporting company
|
| |
☒
|
|
|
| |
|
| |
Emerging growth company
|
| |
☐
|
|
(1)
|
up to 128,740,887 shares of the Company’s Class A common stock, par value $0.0001 per share (the “Class A common stock”)
consisting of (i) 114,940,887 shares of Class A common stock registered for sale by the selling securityholders named in the Registration Statement (the “Selling Securityholders”) (including the shares referred to in the following clause
(ii)), (ii) 6,853,333 shares of Class A common stock issuable upon the exercise of 6,853,333 warrants held by the Selling Securityholders (the “Private Placement Warrants”), and (iii) 13,800,000 shares of Class A common stock issuable
upon the exercise of 13,800,000 warrants included in the units issued by the Company in its initial public offering, each of which is exercisable for one share of Class A common stock, in accordance with its terms (the “Public Warrants”);
|
|
(2)
|
the resale of up to 26,426,937 shares of the Company’s Class B common stock, par value $0.0001 per share (the “Class B common
stock”) held by the Selling Securityholders, which are convertible into 26,426,937 shares of Class A common stock; and
|
|
(3)
|
the resale of up to 6,853,333 Private Placement Warrants, which represent warrants to acquire 6,853,333 shares of Class A
common stock.
|

| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | |
|
•
|
the success, cost and timing of our product development activities;
|
|
•
|
the potential attributes and benefits of our products and services;
|
|
•
|
our ability to obtain and maintain regulatory approval for our products, and any related restrictions and limitations of any
authorized product;
|
|
•
|
our ability to identify, in-license or acquire additional technology;
|
|
•
|
our ability to maintain our existing license, manufacturing and supply agreements;
|
|
•
|
our ability to compete with other companies currently marketing or engaged in the development of ultrasound imaging devices,
many of which have greater financial and marketing resources than us;
|
|
•
|
the size and growth potential of the markets for our products and services, and the ability of each to serve those markets,
either alone or in partnership with others;
|
|
•
|
our estimates regarding expenses, revenue, capital requirements and needs for additional financing;
|
|
•
|
our ability to raise financing in the future; and
|
|
•
|
our financial performance.
|
|
•
|
our rapid growth may not be sustainable and depends on our ability to attract and retain customers;
|
|
•
|
our business could be harmed if we fail to manage our growth effectively;
|
|
•
|
our projections are subject to risks, assumptions, estimates and uncertainties;
|
|
•
|
our business is subject to a variety of U.S. and foreign laws, which are subject to change and could adversely affect our
business;
|
|
•
|
the pricing of our products and services and reimbursement for medical procedures conducted using our products and services;
|
|
•
|
changes in applicable laws or regulations;
|
|
•
|
failure to protect or enforce our intellectual property rights could harm our business, results of operations and financial
condition;
|
|
•
|
the ability to maintain the listing of our Class A common stock on the NYSE; and
|
|
•
|
economic downturns and political and market conditions beyond our control could adversely affect our business, financial
condition and results of operations.
|
|
•
|
We have a limited operating history on which to assess the prospects for our business, we have generated limited revenue from
sales of our products, and we have incurred losses since inception. We anticipate that we will continue to incur significant losses for at least the next several years as we continue to commercialize our existing products and services
and seek to develop and commercialize new products and services.
|
|
•
|
We may need to raise additional funding to expand the commercialization of our products and services and to expand our
research and development (“R&D”) efforts. This additional financing may not be available on acceptable terms, or at all. Failure to obtain this necessary capital when needed may force us to delay, limit or terminate our product
commercialization or development efforts or other operations.
|
|
•
|
Our success depends upon market acceptance of our products and services, our ability to develop and commercialize existing
and new products and services and generate revenues, and our ability to identify new markets for our technology.
|
|
•
|
Medical device development is costly and involves continual technological change, which may render our current or future
products obsolete.
|
|
•
|
We will be dependent upon the success of our sales and customer acquisition and retention strategies. We have limited
experience in marketing and selling our products and related services, and if we are unable to successfully commercialize our products and related services, our business and operating results will be adversely affected.
|
|
•
|
If we do not successfully manage the development and launch of new products, we will not meet our long-term forecasts, and
operating and financial results and condition could be adversely affected.
|
|
•
|
We will need to expand our organization, and we may experience difficulties in recruiting needed additional employees and
consultants and retaining existing employees and consultants, which could disrupt our operations.
|
|
•
|
We have and may continue to experience pricing pressures from contract suppliers or manufacturers on which we rely.
|
|
•
|
We may experience manufacturing problems or delays that could limit the growth of our revenue or increase our losses.
|
|
•
|
We rely on limited or sole suppliers for some of the materials and components used in our products, and we may not be able to
find replacements or immediately transition to alternative suppliers, which could have a material adverse effect on our business, financial condition, results of operations and reputation.
|
|
•
|
If we do not successfully optimize and operate our sales and distribution channels or we do not effectively expand and update
infrastructure, our operating results and customer experience may be negatively impacted and we may have difficulty achieving market awareness and selling our products.
|
|
•
|
The market for our products and services is new, rapidly evolving, and increasingly competitive, as the healthcare industry
in the United States is undergoing significant structural change, which makes it difficult to forecast demand for our products and services.
|
|
•
|
We are subject to extensive government regulation, which could restrict the development, marketing, sale and distribution of
our products and could cause us to incur significant costs.
|
|
•
|
There is no guarantee that the United States Food and Drug Administration (“FDA”) will grant 510(k) clearance or pre-market
approval (“PMA”) of our future products, and failure to obtain necessary clearances or approvals for our future products would adversely affect our ability to grow our business.
|
|
•
|
If we fail to obtain marketing authorizations in other countries for existing products or products under development, we will
not be able to commercialize these products in those countries.
|
|
•
|
Because we do not require training for users of our current products, although they are limited under FDA’s marketing
clearances to use by trained healthcare practitioners, there exists a potential for misuse of these products, which could ultimately harm our reputation and business.
|
|
•
|
We are subject to complex and evolving U.S. and foreign laws and regulations regarding privacy, data protection, and other
matters. Many of these laws and regulations are subject to change and uncertain interpretation, and could result in claims, changes to our business practices, monetary penalties, increased cost of operations, or declines in customer
growth or engagement, or otherwise harm our business.
|
|
•
|
Cybersecurity risks and cyber incidents could result in the compromise of confidential data or critical data systems and give
rise to potential harm to customers, remediation and other expenses, expose us to liability under The Health Insurance Portability and Accountability Act of 1996 (“HIPAA”), as amended by the American Recovery and Reinvestment Act of
2009 and implementing regulations, consumer protection laws or other common law theories, subject us to litigation and federal and state governmental inquiries, damage our reputation and otherwise be disruptive to our business and
operations.
|
|
•
|
If we are unable to protect our intellectual property, our ability to maintain any technological or competitive advantage
over our competitors and potential competitors would be adversely impacted, and our business may be harmed.
|
|
•
|
We may need or may choose to obtain licenses from third parties to advance our research or allow commercialization of our
current or future products, and we cannot provide any assurances that we would be able to obtain such licenses.
|
|
•
|
The exercise of our outstanding warrants for our Class A common stock will increase the number of shares eligible for future
resale in the public market and result in dilution to our stockholders.
|
|
•
|
If we fail to maintain proper and effective internal controls, our ability to produce accurate and timely financial
statements could be impaired, which could harm our operating results, our ability to operate our business and investors’ views of us.
|
|
•
|
The valuation of our warrants could increase the volatility in our net income (loss) in our consolidated statements of
operations.
|
|
•
|
We face the risk of product liability claims and may be subject to damages, fines, penalties and injunctions, among other
things.
|
|
•
|
We are currently subject to a securities class action lawsuit, the unfavorable outcome of which may have a material adverse
effect on our financial condition, results of operations and cash flows.
|
|
(1)
|
Represents the number of shares of Class A common stock outstanding as of April 19, 2023.
Includes (i) 177,481,605 shares of Class A common stock and (ii) 26,426,937 shares of Class A common stock issuable upon conversion of outstanding Class B common stock. The number of issued and outstanding shares of Class A common
stock does not include the shares of Class A common stock reserved for issuance under the Butterfly Amended and Restated 2020 Equity Incentive Plan.
|
|
•
|
continue to build our sales, marketing and distribution infrastructure to commercialize our products and services;
|
|
•
|
continue to develop our products and services;
|
|
•
|
seek to identify, assess, acquire, license and/or develop other products and services and subsequent generations of our current
products and services;
|
|
•
|
seek to maintain, protect and expand our intellectual property portfolio;
|
|
•
|
seek to attract and retain skilled personnel; and
|
|
•
|
support our operations as a public company.
|
|
•
|
launching and commercializing current and future products and services, either directly or in conjunction with one or more
collaborators or distributors;
|
|
•
|
obtaining and maintaining marketing authorization with respect to each of our products and maintaining regulatory compliance
throughout relevant jurisdictions;
|
|
•
|
maintaining clinical and economical value for end-users and customers in changing environments;
|
|
•
|
addressing any competing technological and market developments;
|
|
•
|
negotiating favorable terms in any collaboration, licensing or other arrangements into which we may enter;
|
|
•
|
establishing and maintaining distribution relationships with third-parties that can provide adequate (in amount and quality)
infrastructure to support market demand for our products; and
|
|
•
|
maintaining, protecting and expanding our portfolio of intellectual property rights, including patents, trade secrets and
know-how.
|
|
•
|
challenges of developing (or acquiring externally-developed) technology solutions that are adequate and competitive in meeting
the requirements of next-generation design challenges; and
|
|
•
|
dependence upon physicians’ and other healthcare practitioners’ acceptance of our products.
|
|
•
|
customers increasingly engaging with competing products;
|
|
•
|
failure to introduce new and improved products and services;
|
|
•
|
inability to continue to develop products for mobile devices that customers find engaging, that work with a variety of mobile
operating systems and networks and that achieve a high level of market acceptance;
|
|
•
|
changes in customer sentiment about the quality or usefulness of our products and services or concerns related to privacy and
data sharing, safety, security or other factors;
|
|
•
|
inability to manage and prioritize information to ensure customers are presented with content that is engaging, useful and
relevant to them;
|
|
•
|
adverse changes in our products that are mandated by legislation or regulatory agencies, both in the United States and
internationally; or
|
|
•
|
technical or other problems preventing us from delivering products or services in a rapid and reliable manner or otherwise
affecting the user experience.
|
|
•
|
the challenges associated with building local brand awareness, obtaining local key opinion leader support and clinical support,
implementing reimbursement strategies and building local marketing and sales teams;
|
|
•
|
required compliance with foreign regulatory requirements and laws, including regulations and laws relating to patient data and
medical devices;
|
|
•
|
trade relations among the United States and those foreign countries in which our current or future customers, distributors,
manufacturers and suppliers have operations, including protectionist measures such as tariffs and import or export licensing requirements, whether imposed by the United States or such foreign countries, in particular the strained trade
relations between United States and China since 2018;
|
|
•
|
difficulties and costs of staffing and managing foreign operations;
|
|
•
|
difficulties protecting, procuring or enforcing intellectual property rights internationally;
|
|
•
|
required compliance with anti-bribery laws, such as the FCPA and the UK Bribery Act of 2010, data privacy requirements, labor
laws and anti-competition regulations;
|
|
•
|
laws regulating the confidentiality of sensitive personal information and the circumstances under which such information may be
released and/or collected, such as HIPAA, the HITECH Act, the GDPR and the UK GDPR;
|
|
•
|
laws and business practices that may favor local companies;
|
|
•
|
longer payment cycles and difficulties in enforcing agreements and collecting receivables through certain foreign legal
systems;
|
|
•
|
political and economic instability and war or other military conflict, including the ongoing conflict occurring in Ukraine,
which could have a material adverse impact on our sales in Europe and elsewhere; and
|
|
•
|
potentially adverse tax consequences, tariffs, customs charges, bureaucratic requirements and other trade barriers.
|
|
•
|
disruption in our relationships with customers, distributors, manufacturers or suppliers as a result of such a transaction;
|
|
•
|
unanticipated liabilities related to acquired companies;
|
|
•
|
difficulties integrating acquired personnel, technologies and operations into our existing business;
|
|
•
|
diversion of management’s time and focus away from operating our business to acquisition integration challenges;
|
|
•
|
increases in our expenses and reductions in our cash available for operations and other uses; and
|
|
•
|
possible write-offs or impairment charges relating to acquired businesses.
|
|
•
|
continue to recruit and retain adequate numbers of effective and experienced sales and marketing personnel;
|
|
•
|
effectively train our sales and marketing personnel in the benefits and risks of our products;
|
|
•
|
establish and maintain successful sales, marketing, training and education programs that educate health care professionals so
they can appropriately inform their patients about our products;
|
|
•
|
manage geographically dispersed sales and marketing operations; and
|
|
•
|
effectively train our sales and marketing personnel on the applicable fraud and abuse laws that govern interactions with
healthcare practitioners as well as current and prospective patients and maintain active oversight and auditing measures to ensure continued compliance.
|
|
•
|
design, development and manufacturing processes;
|
|
•
|
labeling, content and language of instructions for use and storage;
|
|
•
|
product testing, non-clinical studies and clinical trials;
|
|
•
|
regulatory authorizations, such as pre-market clearance or PMA;
|
|
•
|
establishment registration, device listing and ongoing compliance with the QSR requirements;
|
|
•
|
advertising and promotion;
|
|
•
|
marketing, sales and distribution;
|
|
•
|
conformity assessment procedures;
|
|
•
|
product traceability and record-keeping procedures;
|
|
•
|
review of product complaints, complaint reporting, recalls and field safety corrective actions;
|
|
•
|
post-market surveillance, including reporting of deaths or serious injuries and malfunctions that, if they were to recur, could
lead to death or serious injury;
|
|
•
|
post-market studies (if applicable); and
|
|
•
|
product import and export.
|
|
•
|
Untitled Letters, Warning Letters, fines, injunctions, consent decrees and civil penalties;
|
|
•
|
customer notification, or orders for repair, replacement or refunds;
|
|
•
|
voluntary or mandatory recall or seizure of our current or future products;
|
|
•
|
administrative detention by the FDA of medical devices believed to be adulterated or misbranded;
|
|
•
|
operating restrictions, suspension or shutdown of production;
|
|
•
|
refusal of our requests for 510(k) clearance or PMA of new products, new intended uses or modifications to existing products;
|
|
•
|
rescission of 510(k) clearance or suspension or withdrawal of PMAs that have already been granted; and
|
|
•
|
criminal prosecution.
|
|
•
|
harm to customers and end-users;
|
|
•
|
business interruptions and delays;
|
|
•
|
the loss, misappropriation, corruption or unauthorized access of data;
|
|
•
|
litigation, including potential class action litigation, and potential liability under privacy, security and consumer
protection laws or other applicable laws;
|
|
•
|
reputational damage;
|
|
•
|
increase to insurance premiums; and
|
|
•
|
foreign, federal and state governmental inquiries, any of which could have a material, adverse effect on our financial position
and results of operations and harm our business reputation.
|
|
•
|
We or our licensors might not have been the first to make the inventions covered by each of our pending patent applications or
granted patents;
|
|
•
|
We or our licensors might not have been the first to file patent applications for our inventions. To determine the priority of
these inventions, we may have to participate in interference proceedings or derivation proceedings declared by the U.S. Patent and Trademark Office (“USPTO”) that could result in substantial cost to us. No assurance can be given that our
patent applications or granted patents (or those of our licensors) will have priority over any other patent or patent application involved in such a proceeding;
|
|
•
|
Others may independently develop similar or alternative products and technologies or duplicate any of our products and
technologies;
|
|
•
|
It is possible that our owned or licensed pending patent applications will not result in granted patents, and even if such
pending patent applications grant as patents, they may not provide a basis for intellectual property protection of commercially viable products, may not provide us with any competitive advantages, or may be challenged and invalidated by
third parties;
|
|
•
|
We may not develop additional proprietary products and technologies that are patentable;
|
|
•
|
The patents of others may have an adverse effect on our business; and
|
|
•
|
While we apply for patents covering our products and technologies and uses thereof, as we deem appropriate, we may fail to
apply for patents on important products and technologies in a timely fashion or at all, or we may fail to apply for patents in potentially relevant jurisdictions
|
|
•
|
royalty payments;
|
|
•
|
meeting certain milestones pertaining to development, commercialization and sales of products using the licensed technology;
|
|
•
|
annual maintenance fees;
|
|
•
|
using commercially reasonable efforts to develop and sell a product using the licensed technology and developing a market for
such product; and
|
|
•
|
providing certain reports.
|
|
•
|
the scope of rights granted under the license agreement and other interpretation-related issues;
|
|
•
|
whether and the extent to which our technology and processes infringe on intellectual property of the licensor that is not
subject to the licensing agreement;
|
|
•
|
our right to sublicense patent and other rights to third parties under collaborative development relationships;
|
|
•
|
our diligence obligations with respect to the use of the licensed technology in relation to our development and
commercialization of our products, and what activities satisfy those diligence obligations; and
|
|
•
|
the ownership of inventions and know-how resulting from the joint creation or use of intellectual property by our licensors and
us and our partners.
|
|
•
|
seek licenses that may not be available on commercially reasonable terms, if at all;
|
|
•
|
abandon any infringing product or redesign our products or processes to avoid infringement;
|
|
•
|
pay substantial damages including, in an exceptional case, treble damages and attorneys’ fees, which we may have to pay if a
court decides that the product or proprietary technology at issue infringes upon or violates the third-party’s rights;
|
|
•
|
pay substantial royalties or fees or grant cross-licenses to our technology; or
|
|
•
|
defend litigation or administrative proceedings that may be costly whether we win or lose, and which could result in a
substantial diversion of our financial and management resources.
|
|
•
|
others may be able to develop and/or practice technology that is similar to our technology or aspects of our technology but
that is not covered by the claims of any patents that have issued, or may issue, from our owned or in-licensed patent applications;
|
|
•
|
we might not have been the first to make the inventions covered by a pending patent application that we own or license;
|
|
•
|
we might not have been the first to file patent applications covering an invention;
|
|
•
|
others may independently develop similar or alternative technologies without infringing our intellectual property rights;
|
|
•
|
pending patent applications that we own or license may not lead to issued patents;
|
|
•
|
patents, if issued, that we own or license may not provide us with any competitive advantages, or may be held invalid or
unenforceable, as a result of legal challenges by our competitors;
|
|
•
|
third parties may compete with us in jurisdictions where we do not pursue and obtain patent protection;
|
|
•
|
we may not be able to obtain and/or maintain necessary or useful licenses on reasonable terms or at all;
|
|
•
|
third parties may be able to also license the intellectual property that we have licensed nonexclusively;
|
|
•
|
third parties may assert an ownership interest in our intellectual property and, if successful, such disputes may preclude us
from exercising exclusive rights over that intellectual property;
|
|
•
|
we may not be able to maintain the confidentiality of our trade secrets or other proprietary information;
|
|
•
|
we may not develop or in-license additional proprietary technologies that are patentable; and
|
|
•
|
the patents of others may have an adverse effect on our business.
|
|
•
|
the ability of our board of directors to issue one or more series of preferred stock;
|
|
•
|
stockholder action by written consent only until the first time when Dr. Rothberg ceases to beneficially own a majority of the
voting power of our capital stock;
|
|
•
|
certain limitations on convening special stockholder meetings;
|
|
•
|
advance notice for nominations of directors by stockholders and for stockholders to include matters to be considered at our
annual meetings;
|
|
•
|
amendment of certain provisions of the organizational documents only by the affirmative vote of (i) a majority of the voting
power of our capital stock and (ii) at least two-thirds of the outstanding shares of our Class B common stock, voting as a separate class; and
|
|
•
|
a dual-class common stock structure with 20 votes per share of our Class B common stock, the result of which is that Dr.
Rothberg has the ability to control the outcome of matters requiring stockholder approval, even though Dr. Rothberg owns less than a majority of the outstanding shares of our capital stock.
|
|
•
|
the success of our or competing products or technologies;
|
|
•
|
developments or disputes concerning issued patents, patent applications or other intellectual property rights;
|
|
•
|
regulatory or legal developments in the U.S. and other countries;
|
|
•
|
the recruitment or departure of key personnel;
|
|
•
|
the level of expenses related to our products;
|
|
•
|
the results of our efforts to discover, develop, manufacture, acquire or in-license our current and additional products;
|
|
•
|
actual or anticipated changes in estimates as to financial results, timelines or recommendations by securities analysts;
|
|
•
|
variations in our financial results or the financial results of companies that are perceived to be similar to us;
|
|
•
|
sales of a substantial number of shares of our common stock in the public market, or the perception in the market that the
holders of a large number of shares intend to sell shares;
|
|
•
|
changes in the structure of healthcare payment systems;
|
|
•
|
general economic, industry and market conditions; and
|
|
•
|
the other factors summarized and described in this Risk Factors section.
|
|
1)
|
Hospitals and healthcare systems, initially focused in the United States;
|
|
2)
|
Expanding into international markets and driving global health equity to improve care across all settings;
|
|
3)
|
Moving Butterfly into home-based care, subject to appropriate authorizations; and
|
|
4)
|
Capturing opportunities in adjacent markets to drive growth.
|
|
•
|
Easy to use, enabling access to the most information with the least amount of effort
through education, an intuitive interface and AI.
|
|
•
|
Everywhere, the scope of our journey to change the standard of care. We are focused on
making Butterfly useful in more settings with cutting edge features and capabilities and building new business models to put Butterfly into every clinician’s pocket.
|
|
•
|
Economical, creating, capturing and delivering value and affordability for all. We are
focused on completing health economics studies to demonstrate that our system delivers better, more informed, lower cost care.
|
|
•
|
For the obstetric clinicians, the device tools can perform gestational age and amniotic fluid index calculations.
|
|
•
|
The device tools can provide automated bladder volume calculations with 3D visualizations and enable easier line placements
using NeedleVizTM technology and Biplane ImagingTM. These tools can be utilized across broad clinical applications and specialties.
|
|
•
|
Using TeleGuidanceTM, healthcare practitioners can perform ultrasound remotely, providing real-time guidance by
connecting with a novice user or peer directly from the Butterfly iQ+ app. Through our Teleguidance feature, healthcare practitioners can control the settings of the application while the device is in use and help the user identify the
image.
|
|
•
|
A targeted, regional, direct salesforce focused on large healthcare system-wide implementations.
|
|
•
|
An eCommerce website through which we sell our Butterfly iQ+ and iQ+ Vet to healthcare practitioners in these geographies,
where allowed by local law.
|
|
•
|
Distributor, veterinary and affiliate relationships to unlock additional channels to supplement our direct and eCommerce sales.
|
|
•
|
Conventional ultrasound systems; and
|
|
•
|
The development of other handheld ultrasound systems with the same or better attributes.
|


|
|
| |
Year ended December 31,
|
|||||||||||||||
|
|
| |
2022
|
| |
2021
|
| |
2020
|
|||||||||
|
(in thousands)
|
| |
Dollars
|
| |
% of
revenue
|
| |
Dollars
|
| |
% of
revenue
|
| |
Dollars
|
| |
% of
revenue
|
|
Revenue:
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Product
|
| |
$50,263
|
| |
68.5%
|
| |
$47,868
|
| |
76.5%
|
| |
$38,347
|
| |
82.9%
|
|
Software and other services
|
| |
23,127
|
| |
31.5
|
| |
14,697
|
| |
23.5
|
| |
7,905
|
| |
17.1
|
|
Total revenue
|
| |
73,390
|
| |
100.0
|
| |
62,565
|
| |
100.0
|
| |
46,252
|
| |
100.0
|
|
Cost of revenue:
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Product
|
| |
26,804
|
| |
36.5
|
| |
29,308
|
| |
46.8
|
| |
46,294
|
| |
100.1
|
|
Software and other services
|
| |
7,126
|
| |
9.7
|
| |
2,238
|
| |
3.6
|
| |
1,068
|
| |
2.3
|
|
Loss on product purchase commitments
|
| |
—
|
| |
—
|
| |
13,965
|
| |
22.3
|
| |
60,113
|
| |
130.0
|
|
Total cost of revenue
|
| |
33,930
|
| |
46.2
|
| |
45,511
|
| |
72.7
|
| |
107,475
|
| |
232.4
|
|
Gross profit (loss)
|
| |
39,460
|
| |
53.8
|
| |
17,054
|
| |
27.3
|
| |
(61,223)
|
| |
(132.4)
|
|
Operating expenses:
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Research and development
|
| |
89,121
|
| |
121.4
|
| |
74,461
|
| |
119.0
|
| |
49,738
|
| |
107.5
|
|
Sales and marketing
|
| |
59,888
|
| |
81.6
|
| |
49,604
|
| |
79.3
|
| |
26,263
|
| |
56.8
|
|
General and administrative
|
| |
83,471
|
| |
113.7
|
| |
85,717
|
| |
137.0
|
| |
24,395
|
| |
52.7
|
|
Total operating expenses
|
| |
232,480
|
| |
316.8
|
| |
209,782
|
| |
335.3
|
| |
100,396
|
| |
217.1
|
|
Loss from operations
|
| |
(193,020)
|
| |
(263.0)
|
| |
(192,728)
|
| |
(308.0)
|
| |
(161,619)
|
| |
(349.4)
|
|
Interest income
|
| |
3,384
|
| |
4.6
|
| |
2,573
|
| |
4.1
|
| |
285
|
| |
0.6
|
|
Interest expense
|
| |
(2)
|
| |
(0.0)
|
| |
(651)
|
| |
(1.0)
|
| |
(1,141)
|
| |
(2.5)
|
|
Change in fair value of warrant liabilities
|
| |
20,859
|
| |
28.4
|
| |
161,095
|
| |
257.5
|
| |
—
|
| |
—
|
|
Other income (expense), net
|
| |
98
|
| |
0.1
|
| |
(2,577)
|
| |
(4.1)
|
| |
(231)
|
| |
(0.5)
|
|
Loss before provision for income taxes
|
| |
(168,681)
|
| |
(229.8)
|
| |
(32,288)
|
| |
(51.6)
|
| |
(162,706)
|
| |
(351.8)
|
|
Provision for income taxes
|
| |
42
|
| |
0.1
|
| |
121
|
| |
0.2
|
| |
39
|
| |
0.1
|
|
Net loss
|
| |
$(168,723)
|
| |
(229.9)%
|
| |
$(32,409)
|
| |
(51.8)%
|
| |
$(162,745)
|
| |
(351.9)%
|
|
|
| |
Year ended December 31,
|
| |
|
| |
|
|||
|
(in thousands)
|
| |
2022
|
| |
2021
|
| |
Change
|
| |
% Change
|
|
Revenue:
|
| |
|
| |
|
| |
|
| |
|
|
Product
|
| |
$50,263
|
| |
$47,868
|
| |
$2,395
|
| |
5.0%
|
|
Software and other services
|
| |
23,127
|
| |
14,697
|
| |
8,430
|
| |
57.4%
|
|
Total revenue:
|
| |
$73,390
|
| |
$62,565
|
| |
$10,825
|
| |
17.3%
|
|
|
| |
Year ended December 31,
|
| |
|
| |
|
|||
|
(in thousands)
|
| |
2022
|
| |
2021
|
| |
Change
|
| |
% Change
|
|
Cost of revenue:
|
| |
|
| |
|
| |
|
| |
|
|
Product
|
| |
$26,804
|
| |
$29,308
|
| |
$(2,504)
|
| |
(8.5)%
|
|
Software and other services
|
| |
7,126
|
| |
2,238
|
| |
4,888
|
| |
218.4%
|
|
Loss on product purchase commitments
|
| |
—
|
| |
13,965
|
| |
(13,965)
|
| |
(100.0)%
|
|
Total cost of revenue:
|
| |
$33,930
|
| |
$45,511
|
| |
$(11,581)
|
| |
(25.4)%
|
|
Percentage of revenue
|
| |
46.2%
|
| |
72.7%
|
| |
|
| |
|
|
|
| |
Year ended December 31,
|
| |
|
| |
|
|||
|
(in thousands)
|
| |
2022
|
| |
2021
|
| |
Change
|
| |
% Change
|
|
Research and development
|
| |
$89,121
|
| |
$74,461
|
| |
$14,660
|
| |
19.7%
|
|
Percentage of revenue
|
| |
121.4%
|
| |
119.0%
|
| |
|
| |
|
|
|
| |
Year ended December 31,
|
| |
|
| |
|
|||
|
(in thousands)
|
| |
2022
|
| |
2021
|
| |
Change
|
| |
% Change
|
|
Sales and marketing
|
| |
$59,888
|
| |
$49,604
|
| |
$10,284
|
| |
20.7%
|
|
Percentage of revenue
|
| |
81.6%
|
| |
79.3%
|
| |
|
| |
|
|
|
| |
Year ended December 31,
|
| |
|
| |
|
|||
|
(in thousands)
|
| |
2022
|
| |
2021
|
| |
Change
|
| |
% Change
|
|
General and administrative
|
| |
$83,471
|
| |
$85,717
|
| |
$(2,246)
|
| |
(2.6)%
|
|
Percentage of revenue
|
| |
113.7%
|
| |
137.0%
|
| |
|
| |
|
|
|
| |
Year ended December 31,
|
| |
|
| |
|
|||
|
(in thousands)
|
| |
2021
|
| |
2020
|
| |
Change
|
| |
% Change
|
|
Revenue:
|
| |
|
| |
|
| |
|
| |
|
|
Product
|
| |
$47,868
|
| |
$38,347
|
| |
$9,521
|
| |
24.8%
|
|
Software and other services
|
| |
14,697
|
| |
7,905
|
| |
6,792
|
| |
85.9%
|
|
Total revenue:
|
| |
$62,565
|
| |
$46,252
|
| |
$16,313
|
| |
35.3%
|
|
|
| |
Year ended December 31,
|
| |
|
| |
|
|||
|
(in thousands)
|
| |
2021
|
| |
2020
|
| |
Change
|
| |
% Change
|
|
Cost of revenue:
|
| |
|
| |
|
| |
|
| |
|
|
Product
|
| |
$29,308
|
| |
$46,294
|
| |
$(16,986)
|
| |
(36.7)%
|
|
Software and other services
|
| |
2,238
|
| |
1,068
|
| |
1,170
|
| |
109.6%
|
|
Loss on product purchase commitments
|
| |
13,965
|
| |
60,113
|
| |
(46,148)
|
| |
(76.8)%
|
|
Total cost of revenue:
|
| |
$45,511
|
| |
$107,475
|
| |
$(61,964)
|
| |
(57.7)%
|
|
Percentage of revenue
|
| |
72.7%
|
| |
232.4%
|
| |
|
| |
|
|
|
| |
Year ended December 31,
|
| |
|
| |
|
|||
|
(in thousands)
|
| |
2021
|
| |
2020
|
| |
Change
|
| |
% Change
|
|
Research and development
|
| |
$74,461
|
| |
$49,738
|
| |
$24,723
|
| |
49.7%
|
|
Percentage of revenue
|
| |
119.0%
|
| |
107.5%
|
| |
|
| |
|
|
|
| |
Year ended December 31,
|
| |
|
| |
|
|||
|
(in thousands)
|
| |
2021
|
| |
2020
|
| |
Change
|
| |
% Change
|
|
Sales and marketing
|
| |
$49,604
|
| |
$26,263
|
| |
$23,341
|
| |
88.9%
|
|
Percentage of revenue
|
| |
79.3%
|
| |
56.8%
|
| |
|
| |
|
|
|
| |
Year ended December 31,
|
| |
|
| |
|
|||
|
(in thousands)
|
| |
2021
|
| |
2020
|
| |
Change
|
| |
% Change
|
|
General and administrative
|
| |
$85,717
|
| |
$24,395
|
| |
$61,322
|
| |
251.4%
|
|
Percentage of revenue
|
| |
137.0%
|
| |
52.7%
|
| |
|
| |
|
|
|
| |
Year ended December 31,
|
||||||
|
(in thousands)
|
| |
2022
|
| |
2021
|
| |
2020
|
|
Net cash used in operating activities
|
| |
$(169,115)
|
| |
$(189,187)
|
| |
$(81,700)
|
|
Net cash used in investing activities
|
| |
(93,779)
|
| |
(9,870)
|
| |
(2,376)
|
|
Net cash provided by financing activities
|
| |
2,881
|
| |
565,692
|
| |
54,280
|
|
Net (decrease) increase in cash, cash equivalents and restricted cash
|
| |
$(260,013)
|
| |
$366,635
|
| |
$(29,796)
|
|
•
|
Hardware devices and accessories;
|
|
•
|
Software subscriptions, including renewal subscriptions, which represent an obligation to provide the customer with ongoing
access to our cloud-hosted software applications on a continuous basis throughout the subscription period;
|
|
•
|
Implementation and integration services; and
|
|
•
|
Extended warranties.
|
|
(1)
|
Any sale, assignment, transfer, conveyance, hypothecation, or other transfer or disposition, directly or indirectly, of any
Class B common stock or any legal or beneficial interest in such share, whether or not for value and whether voluntary or involuntary or by operation of law (including by merger, consolidation, or otherwise), including, without limitation
the transfer of a share of Class B common stock to a broker or other nominee or the transfer of, or entering into a binding agreement with respect to, voting control over such share by proxy or otherwise, other than a permitted transfer.
|
|
(2)
|
Upon the first date on which Dr. Rothberg, together with all other qualified stockholders, collectively cease to beneficially
own at least 20% of the number of Class B common stock (as such number of shares is equitably adjusted in respect of any reclassification, stock dividend, subdivision, combination, or recapitalization of the Class B common stock)
collectively beneficially owned by Dr. Rothberg and permitted transferees of Class B common stock as of the effective time of the Merger.
|
|
(3)
|
Upon the date specified by the affirmative vote of the holders of at least two-thirds (2/3) of the outstanding shares of
Class B common stock, voting as a separate class.
|
|
•
|
in whole and not in part;
|
|
•
|
at a price of $0.01 per warrant;
|
|
•
|
upon not less than 30 days’ prior written notice of redemption (the “30-day redemption period”) to each warrant holder; and
|
|
•
|
if, and only if, the last reported sale price of the Class A common stock equals or exceeds $18.00 per share (as adjusted for
stock splits, stock dividends, reorganizations, recapitalizations and the like and for certain issuances of Class A common stock and equity-linked securities as described below) for any 20 trading days within a 30-trading day period
ending on the third trading day prior to the date Butterfly sends the notice of redemption to the warrant holders.
|
|
•
|
in whole and not in part;
|
|
•
|
at $0.10 per warrant upon a minimum of 30 days’ prior written notice of redemption provided that holders will be able to
exercise their warrants on a cashless basis prior to redemption and receive that number of shares determined by reference to the table below, based on the redemption date and the “fair market value” of the Class A common stock except as
otherwise described below;
|
|
•
|
if, and only if, the last reported sale price of the Class A common stock equals or exceeds $10.00 per share (as adjusted per
stock splits, stock dividends, reorganizations, reclassifications, recapitalizations and the like) on the trading day prior to the date on which Butterfly sends the notice of redemption to the warrant holders;
|
|
•
|
if, and only if, the private placement warrants are also concurrently exchanged at the same price (equal to a number of shares
of Class A common stock) as the outstanding public warrants, as described above; and
|
|
•
|
if, and only if, there is an effective registration statement covering the issuance of the shares of Class A common stock
issuable upon exercise of the warrants and a current prospectus relating thereto available throughout the 30-day period after written notice of redemption is given.
|
|
(period to expiration of warrants)
|
| |
≤ 10.00
|
| |
11.00
|
| |
12.00
|
| |
13.00
|
| |
14.00
|
| |
15.00
|
| |
16.00
|
| |
17.00
|
| |
≥ 18.00
|
|
57 months
|
| |
0.257
|
| |
0.277
|
| |
0.294
|
| |
0.310
|
| |
0.324
|
| |
0.337
|
| |
0.348
|
| |
0.358
|
| |
0.365
|
|
54 months
|
| |
0.252
|
| |
0.272
|
| |
0.291
|
| |
0.307
|
| |
0.322
|
| |
0.335
|
| |
0.347
|
| |
0.357
|
| |
0.365
|
|
51 months
|
| |
0.246
|
| |
0.268
|
| |
0.287
|
| |
0.304
|
| |
0.320
|
| |
0.333
|
| |
0.346
|
| |
0.357
|
| |
0.365
|
|
48 months
|
| |
0.241
|
| |
0.263
|
| |
0.283
|
| |
0.301
|
| |
0.317
|
| |
0.332
|
| |
0.344
|
| |
0.356
|
| |
0.365
|
|
45 months
|
| |
0.235
|
| |
0.258
|
| |
0.279
|
| |
0.298
|
| |
0.315
|
| |
0.330
|
| |
0.343
|
| |
0.356
|
| |
0.365
|
|
42 months
|
| |
0.228
|
| |
0.252
|
| |
0.274
|
| |
0.294
|
| |
0.312
|
| |
0.328
|
| |
0.342
|
| |
0.355
|
| |
0.364
|
|
39 months
|
| |
0.221
|
| |
0.246
|
| |
0.269
|
| |
0.290
|
| |
0.309
|
| |
0.325
|
| |
0.340
|
| |
0.354
|
| |
0.364
|
|
36 months
|
| |
0.213
|
| |
0.239
|
| |
0.263
|
| |
0.285
|
| |
0.305
|
| |
0.323
|
| |
0.339
|
| |
0.353
|
| |
0.364
|
|
33 months
|
| |
0.205
|
| |
0.232
|
| |
0.257
|
| |
0.280
|
| |
0.301
|
| |
0.320
|
| |
0.337
|
| |
0.352
|
| |
0.364
|
|
30 months
|
| |
0.196
|
| |
0.224
|
| |
0.250
|
| |
0.274
|
| |
0.297
|
| |
0.316
|
| |
0.335
|
| |
0.351
|
| |
0.364
|
|
27 months
|
| |
0.185
|
| |
0.214
|
| |
0.242
|
| |
0.268
|
| |
0.291
|
| |
0.313
|
| |
0.332
|
| |
0.350
|
| |
0.364
|
|
24 months
|
| |
0.173
|
| |
0.204
|
| |
0.233
|
| |
0.260
|
| |
0.285
|
| |
0.308
|
| |
0.329
|
| |
0.348
|
| |
0.364
|
|
21 months
|
| |
0.161
|
| |
0.193
|
| |
0.223
|
| |
0.252
|
| |
0.279
|
| |
0.304
|
| |
0.326
|
| |
0.347
|
| |
0.364
|
|
18 months
|
| |
0.146
|
| |
0.179
|
| |
0.211
|
| |
0.242
|
| |
0.271
|
| |
0.298
|
| |
0.322
|
| |
0.345
|
| |
0.363
|
|
15 months
|
| |
0.130
|
| |
0.164
|
| |
0.197
|
| |
0.230
|
| |
0.262
|
| |
0.291
|
| |
0.317
|
| |
0.342
|
| |
0.363
|
|
12 months
|
| |
0.111
|
| |
0.146
|
| |
0.181
|
| |
0.216
|
| |
0.250
|
| |
0.282
|
| |
0.312
|
| |
0.339
|
| |
0.363
|
|
9 months
|
| |
0.090
|
| |
0.125
|
| |
0.162
|
| |
0.199
|
| |
0.237
|
| |
0.272
|
| |
0.305
|
| |
0.336
|
| |
0.362
|
|
6 months
|
| |
0.065
|
| |
0.099
|
| |
0.137
|
| |
0.178
|
| |
0.219
|
| |
0.259
|
| |
0.296
|
| |
0.331
|
| |
0.362
|
|
3 months
|
| |
0.034
|
| |
0.065
|
| |
0.104
|
| |
0.150
|
| |
0.197
|
| |
0.243
|
| |
0.286
|
| |
0.326
|
| |
0.361
|
|
0 months
|
| |
—
|
| |
—
|
| |
0.042
|
| |
0.115
|
| |
0.179
|
| |
0.233
|
| |
0.281
|
| |
0.323
|
| |
0.361
|
|
•
|
Registration rights. Promptly, but in any event within 60 days following the closing
of the Business Combination, Butterfly was required to use its commercially reasonable efforts to file a registration statement under the Securities Act to permit the public resale of all registrable securities as permitted by Rule 415 of
the Securities Act and to cause such registration statement to be declared effective as soon as practicable after the filing thereof, but in no event later than 60 days following the filing deadline (or 90 days following the filing
deadline if the registration statement is reviewed by and receives comments from the SEC). As soon as practicable following the date of effectiveness of the registration statement, but in any event within two business days of such date,
Butterfly agreed to notify the holders of registrable securities of the effectiveness of such registration statement. At any time at which Butterfly has an effective shelf registration statement with respect to a holder’s registrable
securities, any such holder may request to sell all or a portion of their registrable securities pursuant to an underwritten offering pursuant to such shelf registration statement, provided that such holder(s) reasonably expect any such
sales to generate aggregate gross proceeds in excess of $50 million or reasonably expect to sell all of the registrable securities held by such holder, but in no event for aggregate gross proceeds of less than $10 million in gross
proceeds. Butterfly will enter into an underwriting agreement with a managing underwriter or underwriters selected by the initiating holder(s), after consultation with Butterfly, and will take all such other reasonable actions as are
requested by the managing underwriter to expedite or facilitate the disposition of such registrable securities.
|
|
•
|
Demand registration rights. At any time after the closing of the Business Combination,
if Butterfly does not have an effective registration statement outstanding, Butterfly will be required, upon the written request of the holders of at least a majority-in-interest of the then-outstanding registrable securities held by the
Sponsor Group Holders or the Butterfly Holders, as soon as practicable but not more than 45 days after receipt of such written request, to file a registration statement and to effect the registration of all or part of their registrable
securities. Butterfly is not obligated to effect more than an aggregate of three registrations pursuant to a demand registration request.
|
|
•
|
Piggyback registration rights. At any time after the closing of the Business
Combination, if Butterfly proposes to file a registration statement under the Securities Act to register any of its equity securities, or securities or other obligations exchangeable or convertible into equity securities, or to conduct a
public offering, either for its own account or for the account of any other person, subject to certain exceptions and reductions as described in the Amended and Restated Registration Rights Agreement, then Butterfly will give written
notice of such proposed filing to the holders of registrable securities as soon as practicable but not less than 10 days before the anticipated filing of such registration statement. Upon the written request of any holder of registrable
securities in response to such written notice, Butterfly will, in good faith, cause such registrable securities to be included in the registration statement and use its commercially reasonable efforts to cause the underwriters of any
proposed underwritten offering to include such holders’ registrable securities on the same terms and conditions as any similar securities of Butterfly included in such registration.
|
|
(1)
|
prior to such time the board of directors of the corporation approved either the business combination or the transaction which
resulted in the stockholder becoming an interested stockholder;
|
|
(2)
|
upon consummation of the transaction which resulted in the stockholder becoming an interested stockholder, the interested
stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, excluding for purposes of determining the voting stock outstanding (but not the outstanding voting stock owned by the
interested stockholder) those shares owned (i) by persons who are directors and also officers and (ii) employee stock plans in which employee participants do not have the right to determine confidentially whether shares held subject to
the plan will be tendered in a tender or exchange offer; or
|
|
(3)
|
at or subsequent to such time the business combination is approved by the board of directors and authorized at an annual or
special meeting of stockholders, and not by written consent, by the affirmative vote of at least 66 2∕3% of the outstanding voting stock which is not owned by the interested stockholder.
|
|
•
|
1% of the total number of shares of Butterfly Class A common stock then outstanding; or
|
|
•
|
the average weekly reported trading volume of Butterfly’s Class A common stock during the four calendar weeks preceding the
filing of a notice on Form 144 with respect to the sale.
|
|
•
|
the issuer of the securities that was formerly a shell company has ceased to be a shell company;
|
|
•
|
the issuer of the securities is subject to the reporting requirements of Section 13 or 15(d) of the Exchange Act;
|
|
•
|
the issuer of the securities has filed all Exchange Act reports and material required to be filed, as applicable, during the
preceding 12 months (or such shorter period that the issuer was required to file such reports and materials) other than Form 8-K reports; and
|
|
•
|
at least one year has elapsed from the time that the issuer filed current Form 10-type information with the SEC reflecting its
status as an entity that is not a shell company.
|
|
•
|
each person known to the Company to be the beneficial owner of more than 5% of outstanding Company common stock;
|
|
•
|
each of the Company’s NEOs and directors; and
|
|
•
|
all current executive officers and directors of the Company as a group.
|
|
Name and Address of
Beneficial Owner(1)
|
| |
Number of shares
of Class A
Common Stock
|
| |
%
|
| |
Number of shares of
Class B Common
Stock
|
| |
%
|
| |
% of Total
Voting
Power**
|
|
Directors and Executive Officers:
|
|||||||||||||||
|
Jonathan M. Rothberg, Ph.D.(2)
|
| |
10,474,911
|
| |
5.9
|
| |
26,426,937
|
| |
100
|
| |
76.3
|
|
Larry Robbins(3)
|
| |
17,404,767
|
| |
9.8
|
| |
—
|
| |
—
|
| |
2.5
|
|
Dawn Carfora(4)
|
| |
90,655
|
| |
*
|
| |
—
|
| |
—
|
| |
*
|
|
Elazer Edelman, M.D., Ph.D.(5)
|
| |
79,940
|
| |
*
|
| |
—
|
| |
—
|
| |
*
|
|
Gianluca Pettiti(6)
|
| |
96,660
|
| |
*
|
| |
—
|
| |
—
|
| |
*
|
|
S. Louise Phanstiel(7)
|
| |
138,695
|
| |
*
|
| |
—
|
| |
—
|
| |
*
|
|
Erica Schwartz, M.D., J.D., M.P.H.(8)
|
| |
56,199
|
| |
*
|
| |
—
|
| |
—
|
| |
*
|
|
Heather C. Getz(9)
|
| |
242,412
|
| |
*
|
| |
—
|
| |
—
|
| |
*
|
|
Andrei G. Stoica, Ph.D.(10)
|
| |
184,453
|
| |
*
|
| |
—
|
| |
—
|
| |
*
|
|
All Current Directors and Executive
Officers of the Company as a Group (10 Individuals)(11)
|
| |
28,941,389
|
| |
16.3
|
| |
26,426,937
|
| |
100
|
| |
79
|
|
Five Percent Holders:
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Jonathan M. Rothberg, Ph.D.(2)
|
| |
10,474,911
|
| |
5.9
|
| |
26,426,937
|
| |
100
|
| |
76.3
|
|
Fosun Industrial Co., Limited(12)
|
| |
10,716,630
|
| |
6
|
| |
—
|
| |
—
|
| |
1.5
|
|
Entities Affiliated with Glenview Capital Management(3)
|
| |
17,404,767
|
| |
9.8
|
| |
—
|
| |
—
|
| |
2.5
|
|
Blackrock, Inc.(13)
|
| |
10,448,729
|
| |
5.9
|
| |
—
|
| |
—
|
| |
1.5
|
|
The Vanguard Group(14)
|
| |
12,052,848
|
| |
6.8
|
| |
—
|
| |
—
|
| |
1.7
|
|
ARK Investment Management LLC(15)
|
| |
10,000,460
|
| |
5.6
|
| |
—
|
| |
—
|
| |
1.4
|
|
*
|
Indicates beneficial ownership of less than 1%.
|
|
**
|
Percentage of total voting power represents voting power with respect to all outstanding shares of our Class A common stock and
our Class B common stock as a single class. Each share of our Class B common stock is entitled to 20 votes per share and each share of our Class A common stock is entitled to one vote per share.
|
|
(1)
|
Unless otherwise indicated, the business address of each of these individuals is c/o Butterfly Network, Inc., 1600 District
Avenue, Burlington, MA 01803.
|
|
(2)
|
Consists of (i) 2,523,517 shares of Class A common stock held by Dr. Rothberg, (ii) 48,231 shares of Class A common stock
issuable upon the vesting of restricted stock units within 60 days of April 19, 2023, held by Dr. Rothberg, (iii) 21,645 shares of Class A common stock issuable upon the exercise of options exercisable within 60 days of April 19, 2023,
held by Dr. Rothberg, (iv) 6,202,545 shares of Class A common stock distributed from 2012 JMR Trust Common, LLC and held by entities owned by trusts created for the benefit of Dr. Rothberg's
|
|
(3)
|
Consists of (i) 4,546,687 shares of Class A common stock held by Longview Investors LLC, (ii) 8,033,501 shares of Class A common
stock held by Glenview Capital Partners, L.P., Glenview Institutional Partners, L.P., Glenview Capital Master Fund, LTD., Glenview Capital Opportunity Fund, L.P., Glenview Healthcare Master Fund, L.P. and Glenview Offshore Opportunity
Master Fund, LTD. (the “Glenview Investment Funds”), (iii) 3,032,600 shares underlying private placement warrants held by Longview Investors LLC that are exercisable within 60 days of April 19, 2023, (iv) 1,713,333 shares underlying
private placement warrants held by the Glenview Investment Funds that are exercisable within 60 days of April 19, 2023, (v) 8,770 shares of Class A common stock held by Mr. Robbins; (vi) 48,231 shares of Class A common stock issuable upon
the vesting of restricted stock units within 60 days of April 19, 2023 held by Mr. Robbins, and (vii) 21,645 shares of Class A common stock issuable upon the exercise of options exercisable within 60 days of April 19, 2023 held by
Mr. Robbins. Mr. Robbins is the managing member of Longview Investors LLC; the founder, portfolio manager and chief executive officer of Glenview Capital Management, LLC; and a member of our board of directors. Glenview Capital
Management, LLC serves as investment manager to each of the Glenview Funds. Mr. Robbins shares voting and dispositive power over the shares held by Longview Investors LLC, Glenview Capital Management, LLC and the Glenview Investment Funds
and may be deemed to beneficially own such shares. The address of the principal business office for Mr. Robbins, Longview Investors LLC and the Glenview Investment Funds is 767 Fifth Avenue, 44th Floor, New York, NY 10153.
|
|
(4)
|
Consists of (i) 20,779 shares of Class A common stock, (ii) 48,231 shares of Class A common stock issuable upon the vesting of
restricted stock units within 60 days of April 19, 2023, and (iii) 21,645 shares of Class A common stock issuable upon the exercise of options exercisable within 60 days of April 19, 2023.
|
|
(5)
|
Consists of (i) 10,064 shares of Class A common stock, (ii) 48,231 shares of Class A common stock issuable upon the vesting of
restricted stock units within 60 days of April 19, 2023, and (iii) 21,645 shares of Class A common stock issuable upon the exercise of options exercisable within 60 days of April 19, 2023.
|
|
(6)
|
Consists of (i) 26,784 shares of Class A common stock, (ii) 48,231 shares of Class A common stock issuable upon the vesting of
restricted stock units within 60 days of April 19, 2023, and (iii) 21,645 shares of Class A common stock issuable upon the exercise of options exercisable within 60 days of April 19, 2023.
|
|
(7)
|
Consists of (i) 60,049 shares of Class A common stock held by H.G. Phanstiel LP (ii) 8,770 shares of Class A common stock held
by Ms. Phanstiel, (iii) 21,645 shares of Class A common stock issuable upon the exercise of options exercisable within 60 days of April 19, 2023 and (iv) 48,231 shares of Class A common stock issuable upon the vesting of restricted stock
units within 60 days of April 19, 2023. Ms. Phanstiel is the Managing Member of H.G. Phanstiel LP, and therefore has voting and investment control over the shares.
|
|
(8)
|
Consists of (i) 7,968 shares of Class A common stock and (ii) 48,231 shares of Class A common stock issuable upon the vesting of
restricted stock units within 60 days of April 19, 2023.
|
|
(9)
|
Consists of (i) 157,123 shares of Class A common stock issuable upon the vesting of restricted stock units within 60 days of
April 19, 2023, and (ii) 85,289 shares of Class A common stock issuable upon the exercise of options exercisable within 60 days of April 19, 2023.
|
|
(10)
|
Consists of (i) 107,854 shares of Class A common stock, (ii) 25,862 shares of Class A common stock issuable upon the vesting of
restricted stock units within 60 days of April 19, 2023 and (iii) 50,737 shares of Class A common stock issuable upon the exercise of options exercisable within 60 days of April 19, 2023.
|
|
(11)
|
See footnotes 2 through 10; also includes 172,697 shares of Class A common stock issuable upon the vesting of restricted stock
units within 60 days of April 19, 2023 held by Larry Weiss, the Company’s Chief Legal Officer and Corporate Secretary.
|
|
(12)
|
Information is based on the Schedule 13G/A filed by Fosun Industrial Co., Limited (“Fosun Industrial”) and Shanghai Fosun
Pharmaceutical (Group) Co., Ltd. (“Fosun Pharma”) on February 14, 2023, Consists of shares of Class A common stock held by Fosun Industrial. Fosun Industrial is a wholly-owned subsidiary of Fosun Pharma. Fosun Pharma is a subsidiary of,
and is beneficially held approximately 35.82% by, Shanghai Fosun High Technology (Group) Co. Ltd. (“Fosun High Technology”) and 0.22% by Fosun International Limited (“Fosun International”). Fosun High Technology is a wholly-owned
subsidiary of Fosun International, which is a subsidiary of, and is beneficially held approximately 73.53% by, Fosun Holdings Limited (“Fosun Holdings”). Fosun Holdings is a wholly-owned subsidiary of Fosun International Holdings Ltd.
(“Fosun International Holdings”). Fosun International Holdings is beneficially held approximately 85.29% by Guo Guangchang and 14.71% by Wang Qunbin. Guo Guangchang controls Fosun International Holdings and could therefore be deemed the
beneficial owner of the securities held by Fosun Industrial. The address of the principal business office for Fosun Pharma is No. 1289 Yishan Road (Building A, Fosun Technology Park), Shanghai 200233, People’s Republic of China. The
address of the principal business office for Fosun Industrial is 5/F, Manulife Place, 348 Kwun Tong Road, Kowloon, Hong Kong.
|
|
(13)
|
Information is based on the Schedule 13G/A filed by BlackRock, Inc. on February 1, 2023, consists of shares of Class A common
stock beneficially owned, or that may be deemed to be beneficially owned, by BlackRock, Inc. and certain of its subsidiaries as of December 31, 2022. The principal business address of BlackRock, Inc. is 55 East 52nd Street, New York, NY
10055.
|
|
(14)
|
Information is based on the Schedule 13G/A filed by The Vanguard Group on February 9, 2023, consists of shares of Class A common
stock beneficially owned, or that may be deemed to be beneficially owned, by The Vanguard Group, Inc.’s clients, including investment companies registered under the Investment Company Act of 1940 and other managed accounts, as of
December 31, 2022. The principal business address of The Vanguard Group is 100 Vanguard Blvd., Malvern, PA 19355.
|
|
(15)
|
Information is based on the Schedule 13G filed by ARK Investment Management LLC on February 10, 2023, consists of shares of
Class A common stock beneficially owned, or that may be deemed to be beneficially owned, by ARK Investment Management LLC as of December 31, 2022. The principal business address of ARK Investment Management LLC is 200 Central Avenue, St.
Petersburg, FL 33701.
|
|
|
| |
Shares of
Class A
Common
Stock
Beneficially
Owned
Prior to
this Offering**
|
| |
Shares of
Class B
Common
Stock
Beneficially
Owned
Prior to
this Offering
|
| |
Private
Placement
Warrants
Beneficially
Owned Prior
to this
Offering
|
| |
Number of
shares of
Class A
Common
Stock Being
Offered
|
| |
Number of
Shares of
Class B
Common
Stock
Being
Offered***
|
| |
Number of
Private
Placement
Warrants
Being
Offered
|
| |
Shares of
Class A
Common
Stock
Beneficially
Owned
After this
Offered
Shares
of Class A
Common
Stock
are Sold
|
| |
Shares of
Class B
Common
Stock
Beneficially
Owned
After the
Offered
Shares
of Class B
Common
Stock
are Sold
|
| |
Private
Placement
Warrants
Beneficially
Owned
After
the Offered
Warrants
are Sold
|
||||||||||||||||||
|
|
| |
Shares
|
| |
Percent
|
| |
Shares
|
| |
Percent
|
| |
Shares
|
| |
Percent
|
| |
Shares
|
| |
Percent
|
| |
Shares
|
| |
Percent
|
| |
Shares
|
| |
Percent
|
|||||||||
|
Longview Investors LLC(1)
|
| |
4,546,687
|
| |
2.6%
|
| |
—
|
| |
—
|
| |
3,032,600
|
| |
44.3%
|
| |
4,546,687
|
| |
—
|
| |
3,032,600
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
4C Holdings I, LLC (2)(3)
|
| |
—
|
| |
—
|
| |
9,716,596
|
| |
36.8%
|
| |
—
|
| |
—
|
| |
—
|
| |
9,716,596
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Fosun Industrial Co., Limited(4)
|
| |
10,716,630
|
| |
6.0%
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
10,716,630
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Henry B. Rothberg (2)
|
| |
2,239,637
|
| |
1.3%
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
2,239,637
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
AJR 2012 Irrevocable Trust(2)
|
| |
696,851
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
696,851
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
4C Holdings V, LLC (2)(3)
|
| |
—
|
| |
—
|
| |
8,845,238
|
| |
33.5%
|
| |
—
|
| |
—
|
| |
—
|
| |
8,845,238
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
4C Holdings II, LLC (2)(3)
|
| |
—
|
| |
—
|
| |
2,621,701
|
| |
9.9%
|
| |
—
|
| |
—
|
| |
—
|
| |
2,621,701
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
4C Holdings III, LLC (2)(3)
|
| |
—
|
| |
—
|
| |
2,621,701
|
| |
9.9%
|
| |
—
|
| |
—
|
| |
—
|
| |
2,621,701
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
4C Holdings IV, LLC (2)(3)
|
| |
—
|
| |
—
|
| |
2,621,701
|
| |
9.9%
|
| |
—
|
| |
—
|
| |
—
|
| |
2,621,701
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Jonathan M. Rothberg, Ph.D. (2)(5)
|
| |
3,297,854
|
| |
1.9%
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
2,514,747
|
| |
—
|
| |
—
|
| |
783,107
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Deborah J. Rothberg (2)
|
| |
1,501,058
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
1,501,058
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Glenview Capital Master Fund, Ltd. (6)
|
| |
3,351,070
|
| |
1.9%
|
| |
—
|
| |
—
|
| |
825,682
|
| |
12.0%
|
| |
1,617,387
|
| |
—
|
| |
825,682
|
| |
1,733,683
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Laurent Faracci(7)
|
| |
1,580,802
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
1,580,802
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Glenview Capital Opportunity Fund, L.P.(6)
|
| |
2,320,123
|
| |
1.3%
|
| |
—
|
| |
—
|
| |
443,806
|
| |
6.5%
|
| |
1,333,067
|
| |
—
|
| |
443,806
|
| |
987,056
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
John Rodin(8)
|
| |
1,541,550
|
| |
*
|
| |
—
|
| |
—
|
| |
616,800
|
| |
9.0%
|
| |
1,541,550
|
| |
—
|
| |
616,800
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Lee Hathaway(9)
|
| |
770,625
|
| |
*
|
| |
—
|
| |
—
|
| |
514,000
|
| |
7.5%
|
| |
770,625
|
| |
—
|
| |
514,000
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Anil Fernando(10)
|
| |
1,284,625
|
| |
*
|
| |
—
|
| |
—
|
| |
514,000
|
| |
7.5%
|
| |
1,284,625
|
| |
—
|
| |
514,000
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Jeff Patterson(11)
|
| |
385,388
|
| |
*
|
| |
—
|
| |
—
|
| |
154,200
|
| |
2.3%
|
| |
385,388
|
| |
—
|
| |
154,200
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Mark Horowitz(12)
|
| |
462,375
|
| |
*
|
| |
—
|
| |
—
|
| |
308,400
|
| |
4.5%
|
| |
462,375
|
| |
—
|
| |
308,400
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
GIJAMI Trust(2)
|
| |
306,157
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
306,157
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
David A. Rothberg (2)
|
| |
1,097,581
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
1,097,581
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Glenview Offshore Opportunity Master Fund, Ltd.(6)
|
| |
1,886,131
|
| |
1.1%
|
| |
—
|
| |
—
|
| |
391,445
|
| |
5.7%
|
| |
1,144,874
|
| |
—
|
| |
391,445
|
| |
741,257
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Celia R. Meadow(2)
|
| |
711,472
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
711,472
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Darius Shahida (2)(13)
|
| |
959,915
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
959,915
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
UPMC(14)
|
| |
1,000,000
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
1,000,000
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Vanguard Health Management, Inc. (15)
|
| |
1,000,000
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
1,000,000
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
1997 JMR Trust Common, LLC(2)
|
| |
952,277
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
952,277
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Bonnie E Gould Rothberg MD(2)
|
| |
726,696
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
726,696
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Hartz VC Butterfly, LLC(16)
|
| |
100,000
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
100,000
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
FMR LLC(17)
|
| |
231,121
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
231,064
|
| |
—
|
| |
—
|
| |
57
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Stephanie Fielding (2)
|
| |
23,848
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
23,848
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
David A. Rothberg and Nan Birdwhistell (2)
|
| |
435,228
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
435,228
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
The Jeffrey S. Samberg Amended and Restated Revocable
Trust Indenture(18)
|
| |
30,000
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
30,000
|
| |
—
|
| |
—
|
| |
—
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Mary Miller(2)(19)
|
| |
415,320
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
415,320
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
TBC 222 LLC(20)
|
| |
250,000
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
250,000
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
The Joseph D. Samberg Revocable Trust (21)
|
| |
30,000
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
30,000
|
| |
—
|
| |
—
|
| |
—
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Alex Rothberg(2)
|
| |
323,463
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
323,463
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
|
| |
Shares of
Class A
Common
Stock
Beneficially
Owned
Prior to
this Offering**
|
| |
Shares of
Class B
Common
Stock
Beneficially
Owned
Prior to
this Offering
|
| |
Private
Placement
Warrants
Beneficially
Owned Prior
to this
Offering
|
| |
Number of
shares of
Class A
Common
Stock Being
Offered
|
| |
Number of
Shares of
Class B
Common
Stock
Being
Offered***
|
| |
Number of
Private
Placement
Warrants
Being
Offered
|
| |
Shares of
Class A
Common
Stock
Beneficially
Owned
After this
Offered
Shares
of Class A
Common
Stock
are Sold
|
| |
Shares of
Class B
Common
Stock
Beneficially
Owned
After the
Offered
Shares
of Class B
Common
Stock
are Sold
|
| |
Private
Placement
Warrants
Beneficially
Owned
After
the Offered
Warrants
are Sold
|
||||||||||||||||||
|
|
| |
Shares
|
| |
Percent
|
| |
Shares
|
| |
Percent
|
| |
Shares
|
| |
Percent
|
| |
Shares
|
| |
Percent
|
| |
Shares
|
| |
Percent
|
| |
Shares
|
| |
Percent
|
|||||||||
|
Judith Fae Laikin Rothberg Family Trust(2)
|
| |
244,862
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
244,862
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Albany Private Equity Holdings Trust(22)
|
| |
3,171,738
|
| |
1.8%
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
220,000
|
| |
—
|
| |
—
|
| |
2,951,738
|
| |
1.7%
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Elizabeth A. Whayland(2)
|
| |
271,320
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
114,946
|
| |
—
|
| |
—
|
| |
156,374
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Glenview Capital Partners, L.P.(6)
|
| |
281,584
|
| |
*
|
| |
—
|
| |
—
|
| |
52,400
|
| |
*
|
| |
201,220
|
| |
—
|
| |
—
|
| |
80,364
|
| |
*
|
| |
—
|
| |
—
|
| |
52,400
|
| |
*
|
|
Micol Molinari(2)
|
| |
51,000
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
51,000
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Ana Florez(2)
|
| |
222,359
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
143,762
|
| |
—
|
| |
—
|
| |
78,597
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Daniel B Rothberg 2021 Irrevocable Dir. Trust(2)
|
| |
132,343
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
132,343
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Jason B Rothberg 2021 Irrevocable Dir. Trust(2)
|
| |
132,343
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
132,343
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Jason Molinari(2)
|
| |
124,960
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
124,960
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Triumph Ventures LP (22)
|
| |
120,099
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
120,099
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
TRATE Ventures, LLC(23)
|
| |
129,000
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
129,000
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Simone A. Meadow Trust(2)
|
| |
77,322
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
77,322
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Averill L. Meadow Trust(2)
|
| |
77,322
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
77,322
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Herschel Meadow Trust(2)
|
| |
78,645
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
78,645
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
HAS 2012 GST Trust (2)
|
| |
77,322
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
77,322
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Samantha Rothberg (2)
|
| |
71,882
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
71,882
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Rebecca T. Rothberg (2)
|
| |
71,882
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
71,882
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Sheila Bennett Alderman(2)
|
| |
71,882
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
71,882
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Jason B. Rothberg 2012 Trust(2)
|
| |
56,180
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
56,180
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Daniel B. Rothberg 2012 Trust(2)
|
| |
70,224
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
70,224
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Jason B. Rothberg 2012 Irrevocable Trust(2)
|
| |
70,224
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
70,224
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Daniel B. Rothberg 2012 Irrevocable Trust(2)
|
| |
63,202
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
63,202
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Jason Emanuele Molinari as Cust for Sophia Alessandra
Molinari UTMA GA(2)
|
| |
61,181
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
61,181
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Jason Molinari as Cust for William Molinari UTMA GA(2)
|
| |
61,181
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
61,181
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
H.G. Phanstiel LP (2)(24)
|
| |
60,049
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
60,049
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Copper Beech Partners, LLC(25)
|
| |
35,109
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
10,000
|
| |
—
|
| |
—
|
| |
25,109
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Andrew Rothberg (2)
|
| |
17,500
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
17,500
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Michael J Rothberg Cust Justin Rothberg UTMA FL(2)
|
| |
34,504
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
34,504
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Gianluca Pettiti (2)(26)
|
| |
96,660
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
18,014
|
| |
—
|
| |
—
|
| |
78,646
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Dawn Carfora (2)(27)
|
| |
90,655
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
12,009
|
| |
—
|
| |
—
|
| |
78,646
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Westley Moore(28)
|
| |
25,000
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
25,000
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Derek Cribbs(29)
|
| |
25,000
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
25,000
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Randy Simpson(30)
|
| |
25,000
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
25,000
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Ali Tesoriero Saslafsk(2)
|
| |
2,250
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
2,250
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
|
| |
Shares of
Class A
Common
Stock
Beneficially
Owned
Prior to
this Offering**
|
| |
Shares of
Class B
Common
Stock
Beneficially
Owned
Prior to
this Offering
|
| |
Private
Placement
Warrants
Beneficially
Owned Prior
to this
Offering
|
| |
Number of
shares of
Class A
Common
Stock Being
Offered
|
| |
Number of
Shares of
Class B
Common
Stock
Being
Offered***
|
| |
Number of
Private
Placement
Warrants
Being
Offered
|
| |
Shares of
Class A
Common
Stock
Beneficially
Owned
After this
Offered
Shares
of Class A
Common
Stock
are Sold
|
| |
Shares of
Class B
Common
Stock
Beneficially
Owned
After the
Offered
Shares
of Class B
Common
Stock
are Sold
|
| |
Private
Placement
Warrants
Beneficially
Owned
After
the Offered
Warrants
are Sold
|
||||||||||||||||||
|
|
| |
Shares
|
| |
Percent
|
| |
Shares
|
| |
Percent
|
| |
Shares
|
| |
Percent
|
| |
Shares
|
| |
Percent
|
| |
Shares
|
| |
Percent
|
| |
Shares
|
| |
Percent
|
|||||||||
|
Eric S. Meadow and Jason Molinari, as Trustees of the CRM
2020 GST Trust B/O Simone Meadow(2)
|
| |
99,645
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
99,645
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Eric S. Meadow and Jason Molinari, as Trustees of the CRM
2020 GST Trust B/O Averill Meadow(2)
|
| |
103,644
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
103,644
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Eric S. Meadow and Jason Molinari, as Trustees of the CRM
2020 GST Trust B/O Herschel Meadow(2)
|
| |
101,645
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
101,645
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
AJR A Irrevocable Trust u/a/d November 1, 2021, Judith
Fae Laikin Rothberg and Jason Molinari, Trustees(2)
|
| |
1,000,000
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
1,000,000
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
AJR J Irrevocable Trust u/a/d November 1, 2021, Judith
Fae Laikin Rothberg and Jason Molinari, Trustees(2)
|
| |
1,000,000
|
| |
*
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
1,000,000
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Total
|
| |
53,681,201
|
| |
30.2%
|
| |
26,426,937
|
| |
100%
|
| |
6,853,333
|
| |
100%
|
| |
45,986,567
|
| |
26,426,937
|
| |
6,800,933
|
| |
7,694,634
|
| |
4.3%
|
| |
—
|
| |
—
|
| |
52,400
|
| |
*
|
|
*
|
Denotes less than 1%.
|
|
**
|
Certain Selling Securityholders may be deemed to beneficially own other shares reported herein.
|
|
***
|
The Class A common stock issuable upon conversion of shares of Class B common stock is also included in the Number of Shares of
Class A Common Stock Being Offered column immediately preceding.
|
|
(1)
|
Represents Class A common stock and private placement warrants held by Longview Investors LLC (“Longview”), or its affiliates.
Larry Robbins is the managing member of Longview. Mr. Robbins shares voting and dispositive power over the shares held by Longview and may be deemed to beneficially own such shares. As reported on Amendment No. 2 to Schedule 13D filed by
Longview Investors LLC, on March 11, 2022, Longview Investors LLC distributed in kind 3,159,563 shares of Class A common stock and 2,107,400 private placement warrants to certain of its members on a pro rata basis based on their ownership
interest in Longview Investors LLC for no consideration. The address of the principal business office for Longview is 767 Fifth Avenue, 44th Floor, New York, NY 10153.
|
|
(2)
|
Unless otherwise indicated, the business address of each of these holders is c/o Butterfly Network, Inc., 1600 District Avenue,
Burlington, MA 01803.
|
|
(3)
|
Represents Class B common stock, or Class A common stock issuable upon the conversion of Class B common stock, as the case may
be, held by 4C Holdings I, LLC, 4C Holdings II, LLC, 4C Holdings III, LLC, 4C Holdings IV, LLC, 4C Holdings V, LLC (the “4C Holdings LLCs”). Jonathan M. Rothberg, Ph.D., Butterfly’s founder, former Interim Chief Executive Officer and
member of the board of directors, is the sole manager of the 4C Holdings LLCs and therefore has sole voting and investment control over the shares.
|
|
(4)
|
Based on the Schedule 13G/A filed by Fosun Industrial Co., Limited (“Fosun Industrial”) and Shanghai Fosun Pharmaceutical
(Group) Co., Ltd. (“Fosun Pharma”) on February 14, 2023. Represents shares of Class A common stock held by Fosun Industrial. Fosun Industrial is a wholly-owned subsidiary of Fosun Pharma. Fosun Pharma is a subsidiary of, and is
beneficially held approximately 35.82% by, Shanghai Fosun High Technology (Group) Co. Ltd. (“Fosun High Technology”) and 0.22% by Fosun International Limited (“Fosun International”). Fosun High Technology is a wholly-owned subsidiary of
Fosun International, which is a subsidiary of, and is beneficially held approximately 73.53% by, Fosun Holdings Limited (“Fosun Holdings”). Fosun Holdings is a wholly-owned subsidiary of Fosun International Holdings Ltd. (“Fosun
International Holdings”). Fosun International Holdings is beneficially held approximately 85.29% by Guo Guangchang and 14.71% by Wang Qunbin. Guo Guangchang controls Fosun International Holdings and could therefore be deemed the
beneficial owner of the securities held by Fosun Industrial. The address of the principal business office for Fosun Pharma is No. 1289 Yishan Road (Building A, Fosun Technology Park), Shanghai 200233, People’s Republic of China. The
address of the principal business office for Fosun Industrial is 5/F, Manulife Place, 348 Kwun Tong Road, Kowloon, Hong Kong.
|
|
(5)
|
Represents (i) 2,523,517 shares ofClass A common stock (ii) 48,231 shares of Class A common stock that are issuable upon
exercise of options within 60 days of April 19, 2023, and (iii) 21,645 shares of Class A Common Stock issuable upon the vesting of restricted stock units within 60 days of April 19, 2023, held by Jonathan M. Rothberg, Ph.D.
|
|
(6)
|
Larry Robbins is Founder, Portfolio Manager and Chief Executive Officer of Glenview Capital Management, LLC, which serves as
investment manager to Glenview Capital Master Fund, Ltd., Glenview Capital Opportunity Fund, L.P., Glenview Offshore Opportunity Master Fund, Ltd., and Glenview Capital Partners, L.P. (the “Glenview Investment Funds”). Mr. Robbins shares
voting and dispositive power over the shares held by the Glenview Investment Funds and may be deemed to beneficially own such shares. The address of the principal business office for the Glenview Investment Funds is 767 Fifth Avenue, 44th
Floor, New York, NY 10153.
|
|
(7)
|
Represents 1,580,802 shares of Class A common stock that are issuable upon exercise of options held by Laurent Faracci. The
1,580,802 shares of Class A common stock are exercisable within 60 days of April 19, 2023. The business address of Mr. Faracci is 170 East 77th Street, Apt. 9A, New York, NY 10075.
|
|
(8)
|
Represents 1,541,550 shares of Class A common stock. Also includes 616,800 shares upon the exercise of private placement
warrants. The business address of such holder is 767 Fifth Avenue, 44th Floor, New York, NY 10153.
|
|
(9)
|
Represents 770,625 shares of Class A common stock. Also includes 514,000 shares upon the exercise of private placement warrants.
The business address of such holder is 767 Fifth Avenue, 44th Floor, New York, NY 10153.
|
|
(10)
|
Represents 1,284,625 shares of Class A common stock. Also includes 514,000 shares upon the exercise of private placement
warrants. The business address of such holder is 767 Fifth Avenue, 44th Floor, New York, NY 10153.
|
|
(11)
|
Represents 385,388 shares of Class A common stock. Also includes 154,200 shares upon the exercise of private placement warrants.
The business address of such holder is 767 Fifth Avenue, 44th Floor, New York, NY 10153.
|
|
(12)
|
Represents 462,375 shares of Class A common stock. Also includes 308,400 shares upon the exercise of private placement warrants.
The business address of such holder is 767 Fifth Avenue, 44th Floor, New York, NY 10153.
|
|
(13)
|
Represents (i) 424,242 shares of Class A common stock, (ii) 519,150 shares of Class A common stock that are issuable upon
exercise of options within 60 days of April 19, 2023, and (iii) 16,523 shares of Class A common stock issuable upon the vesting of restricted stock units within 60 days of April 19, 2023, held by Darius Shahida.
|
|
(14)
|
The business address of such holder is 6425 Penn Avenue, Suite 200, Pittsburg, PA 15206.
|
|
(15)
|
The business address of such holder is 14201 Dallas Parkway, Dallas, TX 75254.
|
|
(16)
|
The business address of such holder is 500 Plaza Drive, 6th Floor, Congers, New Jersey 07094.
|
|
(17)
|
These accounts are managed by direct or indirect subsidiaries of FMR LLC. Abigail P. Johnson is a Director, the Chairman, the
Chief Executive Officer and the President of FMR LLC. Members of the Johnson family, including Abigail P. Johnson, are the predominant owners, directly or through trusts, of Series B voting common shares of FMR LLC, representing 49% of
the voting power of FMR LLC. The Johnson family group and all other Series B shareholders have entered into a shareholders’ voting agreement under which all Series B voting common shares will be voted in accordance with the majority vote
of Series B voting common shares. Accordingly, through their ownership of voting common shares and the execution of the shareholders’ voting agreement, members of the Johnson family may be deemed, under the Investment Company Act of 1940,
as amended (the “Investment Company Act”), to form a controlling group with respect to FMR LLC. Neither FMR LLC nor Abigail P. Johnson has the sole power to vote or direct the voting of the shares owned directly by the various investment
companies registered under the Investment Company Act (“Fidelity Funds”) advised by Fidelity Management & Research Company, LLC (“FMR Co”), a wholly owned subsidiary of FMR LLC, which power resides with the Fidelity Funds’ Boards of
Trustees. Fidelity Management & Research Company, LLC carries out the voting of the shares under written guidelines established by the Fidelity Funds’ Boards of Trustees. The principal business address of FMR LLC is 245 Summer Street,
Boston, MA 02210.
|
|
(18)
|
The business address of such holder is 77 Bedford Road, Katonah, NY 10536.
|
|
(19)
|
Represents (i) 259,575 shares of Class A common stock that are issuable upon exercise of options and (ii) 155,745 shares of
Class A common stock issuable upon the vesting of restricted stock units held by Mary Miller. 81,113 shares of Class A common stock are exercisable within 60 days of April 19, 2023 held by Ms. Miller and 38,936 shares of Class A common
stock are issuable upon the vesting of restricted stock units within 60 days of April 19, 2023 held by Ms. Miller.
|
|
(20)
|
The business address of such holder is 8 Newbury Street, 5th Floor, Boston, MA 02116.
|
|
(21)
|
The business address of such holder is Level 1, 158 City Road, Southbank, Victoria, Australia 3006.
|
|
(22)
|
John Hammergren is the President of The Stoneyfield Group LLC, the General Partner of Triumph Ventures LP, and therefore has
voting and investment control over the shares held by the entity.
|
|
(23)
|
The business address of such holder is 17 Eugenia Avenue, Kiawah Island, SC 29455.
|
|
(24)
|
S. Louise Phanstiel is the Managing Member of H.G. Phanstiel LP, and therefore has voting and investment control over the shares
held by the entity.
|
|
(25)
|
Reflects 35,109 shares of Class A common stock held by Copper Beech Partners, LLC. Aryeh Davis is the managing member of Copper
Beech Partners, LLC and disclaims beneficial ownership of the shares held by the entity. The business address of such holder is 4 Copper Beech Lane, Lawrence, New York, 11559.
|
|
(26)
|
Represents (i) 26,784 shares of Class A common stock and (ii) 21,645 shares of Class A common stock that are issuable upon
exercise of options within 60 days of April 19, 2023, (iii) 48,231 shares of Class A common stock issuable upon the vesting of restricted stock units within 60 days of April 19, 2023, held by Darius Shahida.
|
|
(27)
|
Represents (i) 20,779 shares of Class A common stock and (ii) 21,645 shares of Class A common stock that are issuable upon
exercise of options within 60 days of April 19, 2023, (iii) 48,231 shares of Class A common stock issuable upon the vesting of restricted stock units within 60 days of April 19, 2023, held by Dawn Carfora.
|
|
(28)
|
The address of such holder is 767 Fifth Avenue, 44th Floor, New York, NY 10153.
|
|
(29)
|
The address of such holder is 15 Central Park West, New York, NY 10023.
|
|
(30)
|
The address of such holder is 313 Stanwich Road, Greenwich, CT 06830.
|
|
Names
|
| |
Ages
|
| |
Positions
|
|
Executive Officers:
|
| |
|
| |
|
|
Joseph DeVivo
|
| |
56
|
| |
President, Chief Executive Officer and Chairperson of the Board of Directors
|
|
Heather C. Getz
|
| |
48
|
| |
Chief Financial Officer
|
|
Lawrence Weiss
|
| |
53
|
| |
Chief Legal Officer and Corporate Secretary
|
|
Andrei G. Stoica
|
| |
50
|
| |
Chief Technology Officer
|
|
Non-Employee Directors:
|
| |
|
| |
|
|
Dawn Carfora
|
| |
51
|
| |
Director
|
|
Elazer Edelman, M.D., Ph.D.
|
| |
66
|
| |
Director
|
|
Gianluca Pettiti
|
| |
44
|
| |
Director
|
|
S. Louise Phanstiel
|
| |
64
|
| |
Director
|
|
Larry Robbins
|
| |
53
|
| |
Director
|
|
Erica Schwartz, M.D., J.D., M.P.H.
|
| |
51
|
| |
Director
|
|
Jonathan M. Rothberg, Ph.D.
|
| |
59
|
| |
Founder and Director
|
|
•
|
the achievement of corporate goals and individual performance;
|
|
•
|
the level of contributions made to the general management and leadership of the Company;
|
|
•
|
the appropriateness of salary increases;
|
|
•
|
the amount of bonuses to be paid, if any; and
|
|
•
|
whether or not stock option, restricted stock unit and/or other equity awards should be made.
|
|
•
|
all information relating to such person that would be required to be disclosed in a proxy statement;
|
|
•
|
certain biographical and share ownership information about the stockholder and any other proponent, including a description of
any derivative transactions in the Company’s securities;
|
|
•
|
a description of certain arrangements and understandings between the proposing stockholder and any beneficial owner and any
other person in connection with such stockholder nomination; and
|
|
•
|
a statement whether or not either such stockholder or beneficial owner intends to deliver a proxy statement and form of proxy
to holders of voting shares sufficient to carry the proposal.
|
|
•
|
certain biographical information concerning the proposed nominee;
|
|
•
|
all information concerning the proposed nominee required to be disclosed in solicitations of proxies for election of directors;
|
|
•
|
certain information about any other security holder of the Company who supports the proposed nominee;
|
|
•
|
a description of all relationships between the proposed nominee and the recommending stockholder or any beneficial owner,
including any agreements or understandings regarding the nomination; and
|
|
•
|
additional disclosures relating to stockholder nominees for directors, including completed questionnaires and disclosures
required by our bylaws.
|
|
Name and Principal Position
|
| |
Year
|
| |
Salary
($)
|
| |
Bonus
($)(1)
|
| |
Stock
Awards
($)(2)
|
| |
Option
Awards
($)(3)
|
| |
All Other
Compensation
($)
|
| |
Total
($)
|
|
Jonathan M. Rothberg,
Former Interim Chief Executive Officer(4)
|
| |
2022
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
217,498(5)
|
| |
217,498
|
|
Todd Fruchterman,
Former Chief Executive Officer(6)
|
| |
2022
|
| |
780,000
|
| |
2,655,706(7)
|
| |
989,449(8)
|
| |
2,215,456(9)
|
| |
6,640,611
|
| |
|
| |
2021
|
| |
687,500
|
| |
3,272,247
|
| |
17,025,602
|
| |
12,586,305
|
| |
1,244,510
|
| |
34,816,164
|
||
|
Heather Getz
Chief Financial Officer(10)
|
| |
2022
|
| |
316,667
|
| |
255,464(11)
|
| |
2,249,998
|
| |
749,458
|
| |
401,897(12)
|
| |
3,973,484
|
|
Andrei Stoica,
Chief Technology Officer
|
| |
2022
|
| |
475,000
|
| |
199,500
|
| |
1,714,139
|
| |
—
|
| |
319,131(13)
|
| |
2,707,770
|
| |
2021
|
| |
183,333
|
| |
749,452
|
| |
660,002
|
| |
662,844
|
| |
165,126
|
| |
2,420,757
|
|
(1)
|
The amounts in this column for 2022 reflect cash bonuses earned in 2022 and paid in 2023 as well as a $50,000 sign-on bonus paid
to Ms. Getz when she commenced employment with us.
|
|
(2)
|
The amounts in this column reflect the aggregate grant date fair value of stock awards granted during 2022 and 2021,
respectively, computed in accordance with Accounting Standards Codification, or ASC, Topic 718, Compensation—Stock Compensation, or Topic 718. Such grant date fair values do not take into account
any estimated forfeitures. Details as to the assumptions used to calculate the fair value of the option awards are included in Note 11 “Equity Incentive Plan” to our consolidated audited financial statements included in our Annual Report
on Form 10-K for the fiscal year ended December 31, 2022. The grant date fair value of each time-based RSU award is measured based on the closing price of our Class A common stock on the grant date. The value of the performance stock
unit, or PSU, awards granted on March 4, 2022 and March 22, 2022 to Dr. Fruchterman, based upon the then-probable outcome of the performance conditions, as computed in accordance with Topic 718, was $349,134 and $127,016 for each award,
respectively. Assuming that the maximum level of performance will be achieved, and assuming the $4.05 and $5.10 closing prices of our Class A common stock on March 4, 2022 and March 22, 2022, respectively, the value of each such PSU award
is $523,701 and $190,523, respectively. These amounts do not necessarily correspond to the actual value recognized or that may be recognized by the NEOs.
|
|
(3)
|
The amounts in this column reflect the aggregate grant date fair value of the option awards granted during 2022 and 2021,
respectively, computed in accordance with Topic 718, using the Black-Scholes option-pricing model. Such grant date fair values do not take into account any estimated forfeitures. Details as to the assumptions used to calculate the fair
value of the option awards are included in Note 11 “Equity Incentive Plan” to our consolidated audited financial statements included in our Annual Report on Form 10-K for the fiscal year ended December 31, 2022. These amounts do not
necessarily correspond to the actual value recognized or that may be recognized by the NEOs.
|
|
(4)
|
Dr. Rothberg served as our Interim Chief Executive Officer from December 31, 2022 to April 24, 2023 and received no additional
compensation for this role.
|
|
(5)
|
Amounts reported in this column represent $67,500 of director fees and $149,998 relating to a RSU award granted to Dr. Rothberg
in connection with his services as a director in 2022. The amount in this column relating to Dr. Rothberg’s RSU award reflects the aggregate grant date fair value of the stock award granted during 2022 computed in accordance with Topic
718. Such grant date fair value does not take into account any estimated forfeitures. The amount reported with respect to Dr. Rothberg’s RSU award does not necessarily correspond to the actual value recognized or that may be recognized by
him.
|
|
(6)
|
Dr. Fruchterman commenced employment with us on February 1, 2021 and departed on December 30, 2022.
|
|
(7)
|
For Dr. Fruchterman, includes $36,884 of incremental fair value recognized for a modification to his PSU award granted on
July 12, 2021, computed as of the modification date in accordance with Topic 718. The modification resulted in this award being fully accelerated to vest on December 30, 2022 at the time of Dr. Fruchterman’s separation. See discussion
below under – “Fruchterman Separation Agreement.”
|
|
(8)
|
For Dr. Fruchterman, includes $51,396 in incremental fair value, computed as of the modification date in accordance with Topic
718, with respect to Dr. Fruchterman’s options that were modified as of December 30, 2022 to extend the post-termination exercise period of such options from three months to one year. See discussion below under – “Fruchterman Separation
Agreement.”
|
|
(9)
|
Amounts in this column represent $960 of cell phone reimbursements, $269,530 of relocation-related reimbursements, $221,048 of
tax gross-ups and reimbursements, and $1,723,918 of severance that Dr. Fruchterman became entitled to on December 29, 2022 pursuant to the terms of his separation agreement, and was paid in 2023.
|
|
(10)
|
Ms. Getz commenced employment with us on May 2, 2022; her base salary was prorated accordingly.
|
|
(11)
|
Includes an amount of $21,290, which represents a prorated portion of an additional discretionary bonus paid to Ms. Getz in
connection with her services to the Office of the Chief Executive Officer.
|
|
(12)
|
Amounts reported in this column represent $640 of cell phone reimbursements, $230,000 of relocation-related reimbursements,
$169,861 of tax gross-ups and reimbursements, and $1,396 of 401(k) plan employer match contributions.
|
|
(13)
|
Amounts reported in this column represent $220,155 of relocation-related reimbursements, $92,877 of tax gross-ups and
reimbursements, and $6,100 of 401(k) plan employer match contributions.
|
|
Name
|
| |
2022 Base Salary
|
|
Todd M. Fruchterman, M.D., Ph.D.
|
| |
$780,000
|
|
Heather Getz(1)
|
| |
$475,000
|
|
Andrei Stoica, Ph.D.
|
| |
$475,000
|
|
Jonathan M. Rothberg, Ph.D.(2)
|
| |
—
|
|
(1)
|
Ms. Getz commenced employment with us on May 2, 2022. This amount reflects her annual base salary assuming employment for the
full year. Her actual base salary received was prorated according to her hire date.
|
|
(2)
|
Dr. Rothberg served as our Interim Chief Executive Officer effective December 31, 2022 to April 24, 2023 and received no
additional compensation for this role.
|
|
Name
|
| |
2022 Target Bonus
(% of Base Salary)
|
| |
2022 Target Bonus
($)
|
|
Todd M. Fruchterman, M.D., Ph.D.
|
| |
100%
|
| |
$780,000
|
|
Heather Getz
|
| |
70%
|
| |
$332,500
|
|
Andrei Stoica, Ph.D.
|
| |
50%
|
| |
$237,500
|
|
Jonathan M. Rothberg, Ph.D.(1)
|
| |
—
|
| |
—
|
|
(1)
|
Dr. Rothberg was not bonus eligible for 2022 and did not receive any bonus for his services as Interim Chief Executive Officer.
|
|
•
|
Achievement towards a formal revenue goal.
|
|
•
|
Achievement of non-financial strategic objectives.
|
|
•
|
Individual performance multipliers.
|
|
Name
|
| |
Target Bonus
Opportunity
|
| |
Annual Cash
Incentive Earned
|
| |
% of Target
|
|
Todd M. Fruchterman, M.D., Ph.D.(1)
|
| |
100%
|
| |
$0
|
| |
0%
|
|
Heather Getz(2)
|
| |
70%
|
| |
$234,174
|
| |
96%
|
|
Andrei Stoica, Ph.D.(3)
|
| |
50%
|
| |
$199,500
|
| |
96%
|
|
Jonathan M. Rothberg, Ph.D.(3)
|
| |
—
|
| |
—
|
| |
—
|
|
(1)
|
Dr. Fruchterman’s employment with us ended on December 30, 2022. Mr. Fruchterman earned a bonus equal to 80% of his Target
Opportunity, equal to $149,696; however, such amount was paid as severance and not as bonus compensation.
|
|
(2)
|
As described above, the Company exercised its negative discretion to reduce the overall payout of the bonus payable to Ms. Getz.
As a result, Ms. Getz’s bonus payment represents 96% of her target bonus or 84% of her 2022 salary.
|
|
(3)
|
As described above, the Company exercised its negative discretion to reduce the overall payout of the bonus payable to Dr.
Stoica. As a result, Dr. Stoica’s bonus payment represents 96% of his target bonus or 84% of his 2022 salary.
|
|
(4)
|
Dr. Rothberg served as our Interim Chief Executive Officer from December 31, 2022 to April 24, 2023 and received no additional
compensation for this role.
|
|
Award Type
|
| |
Description / Objective
|
|||
|
Stock Options
|
| |
•
|
| |
Vest over a three-year period from the grant date
|
| |
•
|
| |
Realized value strongly linked to share price appreciation following grant date
|
||
|
RSUs
|
| |
•
|
| |
Vest over a three or four-year period from the grant date
|
| |
•
|
| |
Realized value linked to share price while maintaining retentive glue during
times of volatility
|
||
|
Performance Stock Units
|
| |
•
|
| |
Awarded to select executives to further incentivize performance
|
| |
•
|
| |
May be earned from 0% - 150% of target units awarded based on a 1-year GAAP
organic revenue goal
|
||
| |
•
|
| |
Earned units vest on the third anniversary of the grant date
|
||
|
|
| |
|
| |
Option Awards
|
| |
Stock Awards
|
||||||||||||||||||
|
Name
|
| |
Grant Date
|
| |
Number of
Securities
Underlying
Unexercised
Options (#)
Exercisable
|
| |
Number of
Securities
Underlying
Unexercised
Options (#)
Unexercisable
|
| |
Option
Exercise
Price ($)
|
| |
Option
Expiration
Date(1)
|
| |
Number of
Shares or
Units of
Stock That
Have Not
Vested (#)
|
| |
Market
Value of
Shares or
Units of
Stock That
Have Not
Vested
($)(2)
|
| |
Equity
Incentive
Plan
Awards:
Number of
Unearned
Shares,
Units or
Other
Rights
That Have
Not
Vested (#)
|
| |
Equity
Incentive
Plan
Awards:
Market
or
Payout
Value of
Unearned
Shares,
Units or
Other
Rights
That
Have
Not
Vested
($)
|
|
Jonathan M. Rothberg,
Former Interim Chief Executive Officer
|
| |
2/16/2021(3)
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
8,772
|
| |
21,579
|
| |
—
|
| |
—
|
| |
7/1/2021
|
| |
21,645
|
| |
—
|
| |
14.25
|
| |
6/30/2031
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
||
| |
6/17/2022(4)
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
48,231
|
| |
118,648
|
| |
—
|
| |
—
|
||
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Todd Fruchterman,
Former Chief Executive Officer(5)
|
| |
2/1/2021
|
| |
1,135,640
|
| |
—
|
| |
15.29
|
| |
3/30/2023
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
7/12/2021
|
| |
176,604
|
| |
—
|
| |
12.48
|
| |
3/30/2023
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
||
| |
3/4/2022
|
| |
150,862
|
| |
—
|
| |
4.05
|
| |
12/31/2023
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
||
| |
3/22/2022
|
| |
51,767
|
| |
—
|
| |
5.10
|
| |
12/31/2023
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
||
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Heather Getz,
Chief Financial Officer
|
| |
5/2/2022(6)
|
| |
—
|
| |
314,915
|
| |
3.58
|
| |
5/1/2032
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
5/2/2022(7)
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
628,491
|
| |
1,546,088
|
| |
—
|
| |
—
|
||
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
|
Andrei Stoica,
Chief Technology Officer
|
| |
7/19/2021(8)
|
| |
38,053
|
| |
83,718
|
| |
10.68
|
| |
7/18/2031
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
7/19/2021(9)
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
42,487
|
| |
104,518
|
| |
—
|
| |
—
|
||
| |
3/4/2022(10)
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
310,344
|
| |
763,446
|
| |
—
|
| |
—
|
||
| |
3/22/2022(11)
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
89,656
|
| |
220,554
|
| |
—
|
| |
—
|
||
|
(1)
|
All option awards generally have a ten-year term from the grant date. Pursuant to our current equity plan, upon separation from
the Company, unexercised options expire 3 months after the departure date. Dr. Fruchterman’s March 4, 2022 and March 22, 2022 option awards were modified at the time of his departure to extend their expiration date from three months after
his departure on December 30, 2022 to one year after his departure.
|
|
(2)
|
The market value of the stock awards is based on the closing price of our Class A common stock of $2.46 per share on
December 31, 2022.
|
|
(3)
|
The shares underlying this RSU vest as to 33% of the award on February 16, 2022, with the remainder of the award vesting in 2
equal annual installments thereafter, subject to Dr. Rothberg’s continued service through the applicable vesting dates.
|
|
(4)
|
The shares underlying this RSU vest as to 100% of the award on June 15, 2023, subject to Dr. Rothberg’s continued service
through the applicable vesting dates.
|
|
(5)
|
All of Dr. Fruchterman’s unvested option awards and stock awards either had their vesting accelerated to his departure date of
December 30, 2022 or were forfeited as of his departure date. As a result, as of December 31, 2022, all of Dr. Fruchterman’s outstanding option awards are exercisable, and he has no outstanding unvested stock awards.
|
|
(6)
|
The shares underlying this option vest as to 25% of the award on May 2, 2023, with the remainder of the award vesting in 36
equal monthly installments thereafter, subject to Ms. Getz’s continued service through the applicable vesting dates.
|
|
(7)
|
The shares underlying this RSU vest as to 25% of the award on May 2, 2023, with the remainder of the award vesting in 3 equal
annual installments thereafter, subject to Ms. Getz’s continued service through the applicable vesting dates.
|
|
(8)
|
The shares underlying this option vest as to 25% of the award on September 30, 2022, with the remainder of the award vesting in
36 equal monthly installments thereafter, subject to Mr. Stoica’s continued service through the applicable vesting dates.
|
|
(9)
|
The shares underlying this RSU vest as to 25% of the award on September 30, 2022, with the remainder of the award vesting in 12
equal quarterly installments thereafter, subject to Mr. Stoica’s continued service through the applicable vesting dates.
|
|
(10)
|
The shares underlying this RSU vest as to 33% of the award on March 4, 2023, with the remainder of the award vesting in 8 equal
quarterly installments thereafter, subject to Mr. Stoica’s continued service through the applicable vesting dates.
|
|
(11)
|
The shares underlying this RSU vest as to 33% of the award on March 22, 2023, with the remainder of the award vesting in 8 equal
quarterly installments thereafter, subject to Mr. Stoica’s continued service through the applicable vesting dates.
|
|
•
|
Severance payable in the form of salary continuation. The severance amount is equal to participant’s then-current base salary
times a multiplier determined based on the participant’s title or role with us. The multiplier for Ms. Getz (executive vice president) is 1.0 and for Dr. Stoica (senior vice president) is .75.
|
|
•
|
We will pay for company contribution for continuation coverage under the Consolidated Omnibus Budget Reconciliation Act of
1985, as amended, or COBRA, during the severance period.
|
|
•
|
Severance payable in a single lump sum. The severance amount is equal to participant’s then-current base salary and
then-current target annual bonus opportunity, times a change in control multiplier determined based on the participant’s title or role with us. The multiplier for Ms. Getz and Dr. Stoica is 1.0.
|
|
•
|
We will pay for company contribution for continuation coverage under COBRA during the severance period.
|
|
•
|
Any outstanding unvested equity awards held by the participant under our then-current outstanding equity incentive plan(s) will
become fully vested on the date the termination of such participant’s employment becomes effective.
|
|
•
|
a material reduction of the participant’s base salary as in effect immediately prior to the reduction; or
|
|
•
|
a material reduction in the participant’s authority, duties or responsibilities.
|
|
•
|
willful misconduct or gross negligence in the performance of the participant’s duties;
|
|
•
|
refusal to follow the lawful directions of the Chief Executive Officer, which, if curable, has not been cured by the
participant within 30 days after he or she receives notice from the Chief Executive Officer;
|
|
•
|
breach of a fiduciary duty owed to us;
|
|
•
|
fraud, embezzlement or other material dishonesty with respect to us;
|
|
•
|
violation of applicable federal, state or local law or regulation governing our business;
|
|
•
|
commission, conviction, plea of nolo contendere, guilty plea, or confession to a crime based upon an act of fraud, embezzlement
or dishonesty or to a felony;
|
|
•
|
habitual abuse of alcohol or any controlled substance or reporting to work under the influence of alcohol or any controlled
substance (other than a controlled substance that the participant is properly taking under a current prescription);
|
|
•
|
misappropriation (or attempted misappropriation) by the participant of any material assets or business opportunities of us or
any of our subsidiaries or affiliates;
|
|
•
|
a material failure to comply with our written policies or rules, as they may be in effect from time to time during the
participant’s employment, including policies and rules prohibiting discrimination or harassment, which, if curable, has not been cured by the participant within 30 days after he or she receives notice from the Chief Executive Officer;
|
|
•
|
a material breach of the participant’s employment agreement or offer letter, the Non-Competition, Confidentiality and
Intellectual Property Agreement or any other written agreement between us or one of our subsidiaries and participant, which, if curable, has not been cured by the participant within 30 days after he or she receives notice from the Chief
Executive Officer.
|
|
(i)
|
any person or group of persons (other than us or our affiliates) becomes the owner, directly or indirectly, of our securities
representing more than 50% of the combined voting power of our then outstanding voting securities (the “Outstanding Company Voting Securities”) (but excluding any bona fide financing event in which securities are acquired directly from
us); or
|
|
(ii)
|
the consummation of a merger or consolidation of us with any other corporation, other than a merger or consolidation (i) that
results in the Outstanding Company Voting Securities immediately prior thereto continuing to represent (either by remaining outstanding or by being converted into voting securities of the surviving entity) at least 50% of the combined
voting power of the Outstanding Company Voting Securities (or such surviving entity or, if we or the entity surviving such merger is then a subsidiary, the ultimate parent thereof) outstanding immediately after such merger or
consolidation, or (ii) immediately following which the individuals who comprise the board of directors immediately prior thereto constitute at least a majority of the board of directors of the entity surviving such merger or consolidation
or, if we or the entity surviving such merger is then a subsidiary, the ultimate parent thereof; or
|
|
(iii)
|
the sale or disposition by us of all or substantially all of our assets, other than (i) a sale or disposition by us of all or
substantially all of our assets to an entity, at least 50% of the combined voting power of the voting securities of which are owned directly or indirectly by our stockholders following the completion of such transaction in substantially
the same proportions as their ownership of us immediately prior to such sale or (ii) a sale or disposition of all or substantially all of our assets immediately following which the individuals who comprise the board of directors
immediately prior thereto constitute at least a majority of the board of directors of the entity to which such assets are sold or disposed or, if such entity is a subsidiary, the ultimate parent thereof;
|
|
(iv)
|
provided that with respect to Sections (i), (ii) and (iii) above, a transaction or series of integrated transactions will not
be deemed a Change in Control (A) unless the transaction qualifies as a change in control within the meaning of Section 409A of the Code, or (B) if following the conclusion of the transaction or series of integrated transactions, the
holders of our Class B Common Stock immediately prior to such transaction or series of transactions continue to have substantially the same proportionate voting power in an entity which owns all or substantially all of our assets
immediately following such transaction or series of transactions.
|
|
Fiscal
Year
(a)(1)
|
| |
SCT
For PEO
Rothberg
(b)(2)
|
| |
CAP
For PEO
Rothberg
(c)(3)
|
| |
SCT
For PEO2
Fruchterman
(d)(2)
|
| |
CAP
For PEO2
Fruchterman
(e)(3)
|
| |
SCT
For PEO3
Faracci
(f)(2)
|
| |
CAP
For PEO3
Faracci
(g)(3)
|
| |
Average
SCT
for Other
NEOs
(h)(4)
|
| |
Average
CAP
For Other
NEOs
(i)(3)
|
| |
TSR
(j)(5)
|
| |
Net
Income
($M)
(k)(6)
|
|
2022
|
| |
$217,498
|
| |
($1,412,374)
|
| |
$6,640,611
|
| |
($2,988,104)
|
| |
—
|
| |
—
|
| |
$3,340,627
|
| |
$2,187,065
|
| |
$10.93
|
| |
($168.7)
|
|
2021
|
| |
—
|
| |
—
|
| |
$34,816,164
|
| |
$17,275,459
|
| |
$3,541,800
|
| |
($17,942,453)
|
| |
$2,875,233
|
| |
$2,441,247
|
| |
$29.73
|
| |
($32.4)
|
|
(1)
|
For 2022, Dr. Rothberg (“PEO 1”) and Dr. Fruchterman (“PEO 2”) each served as our PEO during part of the year, and Ms. Getz and
Mr. Stoica as our Other Non-PEO NEOs. For 2021, Dr. Fruchterman and Mr. Faracci (“PEO 3”) each served as our PEO during part of the year, and Ms. Fielding, Ms. Pugh, Mr. Shahida, and Mr. Stoica served as our Other Non-PEO NEOs.
|
|
(2)
|
The dollar amounts reported in columns (b), (d), and (e) are the amounts of total compensation reported for Dr. Rothberg, Dr.
Fruchterman, and Mr. Faracci for each corresponding year in the “Total” column of the Summary Compensation Table. Refer to the Summary Compensation Table as set forth on page 117 of this prospectus statement.
|
|
(3)
|
The dollar amounts reported in columns (c), (e), (g), and (i) represent the amount of CAP as computed in accordance with SEC
rules. CAP does not necessarily represent cash and/or equity value transferred to the applicable NEO without restriction, but rather is a value calculated under applicable SEC rules. We do not have a defined benefit plan so no adjustment
for pension benefits is included in the table below. Similarly, no adjustment is made for dividends as dividends are factored into the fair value of the award. The following table details these adjustments:
|
|
Fiscal
Year
|
| |
|
| |
SCT
(a)
|
| |
Grant
Date
Value of
New
Awards
(b)
|
| |
Year
End
Value of
New
Awards
(i)
|
| |
Change
in
Value of
Prior
Awards
(ii)
|
| |
Change
in
Value of
Vested
Awards
(iii)
|
| |
Change
in
Value of
Canceled
Awards
(iv)
|
| |
TOTAL
Equity
CAP
(c)=(i)+(ii)+(iii)
|
| |
CAP
(d) =(a)-(b)+(c)
|
|
2022
|
| |
PEO1
|
| |
$217,498
|
| |
($149,998)
|
| |
$118,648
|
| |
($37,106)
|
| |
($1,561,416)
|
| |
—
|
| |
($1,479,874)
|
| |
($1,412,374)
|
| |
PEO2
|
| |
$6,640,611
|
| |
($3,645,155)
|
| |
—
|
| |
—
|
| |
($4,873,998)
|
| |
($1,109,562)
|
| |
($5,983,560)
|
| |
($2,988,104)
|
||
| |
PEO3
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
||
| |
Non-PEO
NEOs
|
| |
$3,340,627
|
| |
($2,356,798)
|
| |
$1,446,322
|
| |
($189,039)
|
| |
($54,046)
|
| |
—
|
| |
$1,203,236
|
| |
$2,187,065
|
||
|
2021
|
| |
PEO1
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
PEO2
|
| |
$34,816,164
|
| |
($29,611,907)
|
| |
$12,071,202
|
| |
—
|
| |
—
|
| |
—
|
| |
$12,071,202
|
| |
$17,275,459
|
||
| |
PEO3
|
| |
$3,541,800
|
| |
—
|
| |
—
|
| |
—
|
| |
$6,021,275
|
| |
($27,505,528)
|
| |
($21,484,253)
|
| |
($17,942,453)
|
||
| |
Non-PEO
NEOs
|
| |
$2,875,233
|
| |
($1,762,952)
|
| |
$692,140
|
| |
$145,508
|
| |
$491,318
|
| |
—
|
| |
$1,328,966
|
| |
$2,441,247
|
|
(a)
|
The dollar amounts reported in the Summary Compensation Table for the applicable year.
|
|
(b)
|
The grant date fair value of equity awards represents the total of the amounts reported in the “Stock Awards” column in the
Summary Compensation Table for the applicable year.
|
|
(c)
|
The recalculated value of equity awards for each applicable year includes the addition (or subtraction, as applicable) of the
following:
|
|
(i)
|
the year-end fair value of any equity awards granted in the applicable year that are outstanding and unvested as of the end of
the year;
|
|
(ii)
|
the amount of change as of the end of the applicable year (from the end of the prior fiscal year) in fair value of any awards
granted in prior years that are outstanding and unvested as of the end of the applicable year;
|
|
(iii)
|
for awards that vest in the applicable year, the change in the fair value as of the vesting date from the beginning of the
applicable year.
|
|
(iv)
|
for awards that are cancelled in the applicable year, the fair value of awards from the beginning of the applicable year.
|
|
(4)
|
The dollar amounts reported in column (h) are the average amounts of total compensation reported for the other Non-PEO NEOs for
each corresponding year in the “Total” column of the Summary Compensation Table. Refer to the Summary Compensation Table as set forth on page 117 of this prospectus.
|
|
(5)
|
TSR determined in column (j) is based on the value of an initial fixed investment of $100 as of IPO on February 16, 2021.
|
|
(6)
|
The amounts in this column reflect net income as reported in the company’s Consolidated Statements of Operations and
Comprehensive Loss in the Annual Report on Form 10-K for the fiscal year ended December 31, 2022.
|
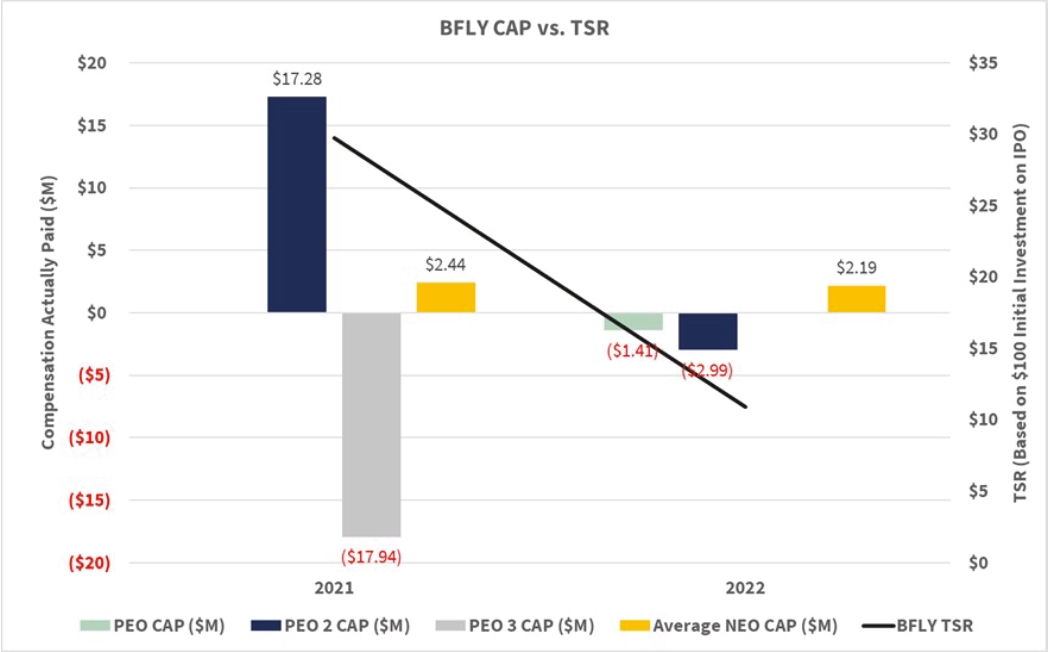
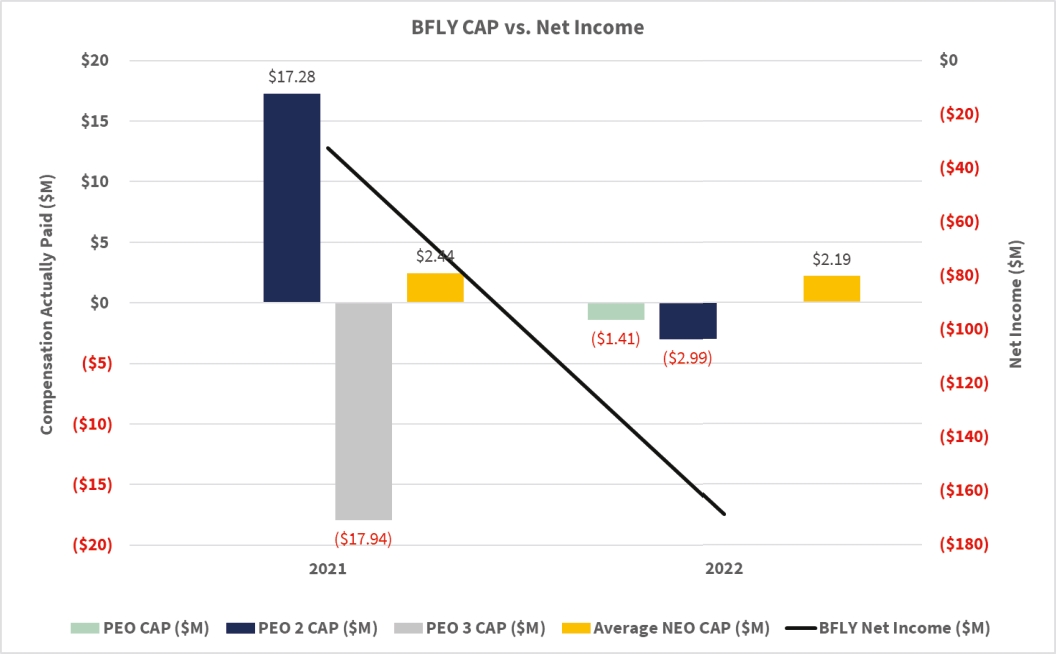
|
Name
|
| |
Fees
Earned or
Paid in
Cash ($)
|
| |
Stock
Awards(1)(2)
($)
|
| |
Option
Awards(2)(3)
($)
|
| |
All Other
Compensation
($)
|
| |
Total ($)
|
|
Dawn Carfora
|
| |
57,500
|
| |
149,998
|
| |
—
|
| |
—
|
| |
207,498
|
|
Elazer Edelman, M.D., Ph.D.
|
| |
65,000
|
| |
149,998
|
| |
—
|
| |
—
|
| |
214,998
|
|
John Hammergren
|
| |
65,000
|
| |
149,998
|
| |
—
|
| |
—
|
| |
214,998
|
|
Gianluca Pettiti
|
| |
75,000
|
| |
149,998
|
| |
—
|
| |
—
|
| |
224,998
|
|
S. Louise Phanstiel
|
| |
77,500
|
| |
149,998
|
| |
—
|
| |
—
|
| |
227,498
|
|
Larry Robbins
|
| |
62,500
|
| |
149,998
|
| |
—
|
| |
—
|
| |
212,498
|
|
Erica Schwartz, M.D., J.D., M.P.H.
|
| |
62,500
|
| |
149,998
|
| |
—
|
| |
—
|
| |
212,498
|
|
(1)
|
These amounts represent the aggregate grant date fair value of stock awards granted to each director in 2022 computed in
accordance with Topic 718. Such grant date fair values do not take into account any estimated forfeitures. Details as to the assumptions used to calculate the fair value of the stock awards are included in Note 11 “Equity Incentive Plan”
to our consolidated audited financial statements included in our Annual Report on Form 10-K for the fiscal year ended December 31, 2022. These amounts do not necessarily correspond to the actual value recognized or that may be recognized
by the directors.
|
|
(2)
|
The following table shows outstanding and unexercised options and unvested RSUs for each non-employee director as of
December 31, 2022, other than for Dr. Rothberg, whose outstanding equity is reported in the Outstanding Equity Awards at 2022 Fiscal Year-End.
|
|
Name
|
| |
Total Options
Outstanding
|
| |
Vested
Options
|
| |
Unvested RSUs
|
|
Dawn Carfora
|
| |
21,645
|
| |
21,645
|
| |
57,003
|
|
Elazer Edelman, M.D., Ph.D.
|
| |
21,645
|
| |
21,645
|
| |
58,297
|
|
John Hammergren
|
| |
21,645
|
| |
21,645
|
| |
57,003
|
|
Gianluca Pettiti
|
| |
21,645
|
| |
21,645
|
| |
57,003
|
|
S. Louise Phanstiel
|
| |
21,645
|
| |
21,645
|
| |
57,003
|
|
Larry Robbins
|
| |
21,645
|
| |
21,645
|
| |
57,003
|
|
Erica Schwartz, M.D., J.D., M.P.H.
|
| |
21,645
|
| |
21,645
|
| |
64,167
|
|
Name
|
| |
RSUs
Granted
(#)
|
| |
Options
Granted (#)
|
| |
Grant Date
|
| |
Grant Date
Fair Value
($)
|
|
Dawn Carfora
|
| |
48,231
|
| |
—
|
| |
6/17/2022
|
| |
149,998
|
|
Elazer Edelman, M.D., Ph.D.
|
| |
48,231
|
| |
—
|
| |
6/17/2022
|
| |
149,998
|
|
John Hammergren
|
| |
48,231
|
| |
—
|
| |
6/17/2022
|
| |
149,998
|
|
Gianluca Pettiti
|
| |
48,231
|
| |
—
|
| |
6/17/2022
|
| |
149,998
|
|
S. Louise Phanstiel
|
| |
48,231
|
| |
—
|
| |
6/17/2022
|
| |
149,998
|
|
Larry Robbins
|
| |
48,231
|
| |
—
|
| |
6/17/2022
|
| |
149,998
|
|
Erica Schwartz, M.D., J.D., M.P.H.
|
| |
48,231
|
| |
—
|
| |
6/17/2022
|
| |
149,998
|
|
Position
|
| |
Retainer
|
|
Audit committee chairperson
|
| |
$20,000
|
|
Audit committee member
|
| |
$10,000
|
|
Compensation committee chairperson
|
| |
$15,000
|
|
Compensation committee member
|
| |
$7,500
|
|
Nominating and corporate governance committee chairperson
|
| |
$10,000
|
|
Nominating and corporate governance committee member
|
| |
$5,000
|
|
Technology committee chairperson
|
| |
$15,000
|
|
Technology committee member
|
| |
$7,500
|
|
|
| |
(a)
|
| |
(b)
|
| |
(c)
|
|
Plan category
|
| |
Number of securities to
be issued upon exercise
of outstanding options,
warrants and rights
|
| |
Weighted-average
exercise price of
outstanding options,
warrants and rights
|
| |
Number of securities
remaining available for
future issuance under
equity compensation
plans (excluding
securities reflected in
column (a))
|
|
Equity compensation plans approved by security holders(1)
|
| |
22,533,203(2)
|
| |
$7.67(3)
|
| |
19,694,837(4)
|
|
Equity compensation plans not approved by security holders(5)
|
| |
—
|
| |
—
|
| |
—
|
|
Total
|
| |
22,533,203
|
| |
$7.67
|
| |
19,694,837(6)
|
|
(1)
|
These plans consist of our 2012 Employee, Director and Consultant Equity Incentive Plan, or 2012 Plan, and our Amended and
Restated 2020 Equity Incentive Plan, as amended, or 2020 Plan.
|
|
(2)
|
Consists of (i) 7,967,561 million shares to be issued upon exercise of outstanding options and RSUs under the 2012 Plan and
(ii) 14,565,642 million shares to be issued upon exercise of outstanding options and RSUs under the 2020 Plan.
|
|
(3)
|
Consists of the weighted-average exercise price of the 12,571,912 million stock options outstanding on December 31, 2022 and
does not include RSU’s.
|
|
(4)
|
Consists of shares that remained available for future issuance under the 2020 Plan as of December 31, 2022. No shares remained
available for future issuance under the 2012 Plan as of December 31, 2022.
|
|
(5)
|
We do not have any compensation plans that were not approved by shareholders nor have we granted any inducement awards.
|
|
(6)
|
The 2020 Plan has an evergreen provision that allows for an annual increase in the number of shares available for issuance under
the 2020 Plan to be added on the first day of each fiscal year, beginning in fiscal year 2021 and ending on the second day of fiscal year 2030. The evergreen provides for an automatic increase in the number of shares available for
issuance equal to the lesser of (i) 4% of the number of outstanding shares of common stock on such date and (ii) an amount determined by the plan administrator.
|
|
•
|
any person who is or was an executive officer, director, or director nominee of the Company at any time since the beginning of
the Company’s last fiscal year;
|
|
•
|
a person who is or was an Immediate Family Member (as defined below) of an executive officer, director, director nominee at any
time since the beginning of the Company’s last fiscal year;
|
|
•
|
any person who, at the time of the occurrence or existence of the transaction, is the beneficial owner of more than 5% of any
class of the Company’s voting securities (a “Significant Stockholder”); or
|
|
•
|
any person who, at the time of the occurrence or existence of the transaction, is an Immediate Family Member of a Significant
Stockholder of the Company.
|
|
•
|
the related person’s interest in the transaction;
|
|
•
|
the approximate dollar value of the amount involved in the transaction;
|
|
•
|
the approximate dollar value of the amount of the related person’s interest in the transaction without regard to the amount of
any profit or loss;
|
|
•
|
whether the transaction was undertaken in the ordinary course of business of the Company;
|
|
•
|
whether the transaction with the related person is proposed to be, or was, entered into on terms no less favorable to the
Company than terms that could have been reached with an unrelated third party;
|
|
•
|
the purpose of, and the potential benefits to the Company of, the transaction; and
|
|
•
|
any other information regarding the transaction or the related person in the context of the proposed transaction that would be
material to investors in light of the circumstances of the particular transaction.
|
|
•
|
banks, financial institutions, or financial services entities;
|
|
•
|
broker-dealers;
|
|
•
|
governments or agencies or instrumentalities thereof;
|
|
•
|
regulated investment companies;
|
|
•
|
real estate investment trusts;
|
|
•
|
expatriates, former citizens, or former long-term residents of the United States;
|
|
•
|
persons that actually or constructively own five percent or more (by vote or value) of our securities (except to the limited
extent set forth below);
|
|
•
|
persons that acquired our securities pursuant to an exercise of employee stock options, in connection with employee share
incentive plans or otherwise as compensation;
|
|
•
|
insurance companies;
|
|
•
|
dealers or traders subject to a mark-to-market method of accounting with respect to our securities (except to the limited
extent set forth below);
|
|
•
|
persons for whom our securities constitutes “qualified small business stock” within the meaning of Section 1202 of the Code or
as “Section 1244 stock” for purposes of Section 1244 of the Code;
|
|
•
|
persons holding our securities shares as part of a “straddle,” constructive sale, hedge, conversion or other integrated or
similar transaction;
|
|
•
|
U.S. Holders (as defined below) whose functional currency is not the U.S. dollar;
|
|
•
|
Subchapter S corporations, partnerships or entities or arrangements classified as partnerships or other pass-through entities
for
|
|
•
|
U.S. federal income tax purposes and any beneficial owners of such partnerships or pass-through entities;
|
|
•
|
tax-exempt entities;
|
|
•
|
controlled foreign corporations (including “specified foreign corporations”); and
|
|
•
|
passive foreign investment companies.
|
|
•
|
an individual who is a citizen or resident of the United States as determined for United States federal income tax purposes;
|
|
•
|
a corporation (or other entity taxable as a corporation) organized in or under the laws of the United States, any state thereof
or the District of Columbia;
|
|
•
|
an estate the income of which is includible in gross income for U.S. federal income tax purposes regardless of its source; or
|
|
•
|
a trust, if (i) a court within the United States is able to exercise primary supervision over the administration of the trust
and one or more United States persons (as defined in the Code) have authority to control all substantial decisions of the trust or (ii) it has a valid election in effect under Treasury Regulations to be treated as a United States person.
|
|
•
|
a non-resident alien individual (other than certain former citizens and residents of the United States subject to U.S. tax as
expatriates);
|
|
•
|
a foreign corporation; or
|
|
•
|
an estate or trust that is not a U.S. Holder;
|
|
•
|
the gain is effectively connected with the conduct by the Non-U.S. Holder of a trade or business within the United States (and,
under certain income tax treaties, is attributable to a United States permanent establishment or fixed base maintained by the Non-U.S. Holder); or
|
|
•
|
we are or have been a “United States real property holding corporation” for U.S. federal income tax purposes at any time during
the shorter of the five-year period ending on the date of disposition or the period that the Non-U.S. Holder held our securities, and, in the case where shares of our Butterfly Class A common stock are regularly traded on an established
securities market, as defined pursuant to applicable Treasury Regulations, the Non-U.S. Holder has owned, directly or constructively, more than 5% of our
|
|
•
|
purchases by a broker-dealer as principal and resale by such broker-dealer for its own account pursuant to this prospectus;
|
|
•
|
ordinary brokerage transactions and transactions in which the broker solicits purchasers;
|
|
•
|
block trades in which the broker-dealer so engaged will attempt to sell the shares as agent but may position and resell a
portion of the block as principal to facilitate the transaction;
|
|
•
|
an over-the-counter distribution in accordance with the rules of the NYSE;
|
|
•
|
through trading plans entered into by a Selling Securityholder pursuant to Rule 10b5-1 under the Exchange Act that are in place
at the time of an offering pursuant to this prospectus and any applicable prospectus supplement hereto that provide for periodic sales of their securities on the basis of parameters described in such trading plans;
|
|
•
|
short sales;
|
|
•
|
distribution to employees, members, limited partners or stockholders of the Selling Securityholders;
|
|
•
|
through the writing or settlement of options or other hedging transaction, whether through an options exchange or otherwise;
|
|
•
|
by pledge to secured debts and other obligations;
|
|
•
|
delayed delivery arrangements;
|
|
•
|
to or through underwriters or agents;
|
|
•
|
in “at the market” offerings, as defined in Rule 415 under the Securities Act, at negotiated prices, at prices prevailing at
the time of sale or at prices related to such prevailing market prices, including sales made directly on a national securities exchange or sales made through a market maker other than on an exchange or other similar offerings through
sales agents;
|
|
•
|
in privately negotiated transactions;
|
|
•
|
in options transactions; and
|
|
•
|
through a combination of any of the above methods of sale, as described below, or any other method permitted pursuant to
applicable law.
|
|
○
|
We tested the design and implementation over the establishment of reserves for losses on
product purchase commitments, including management’s controls over sales projections.
|
|
○
|
For recorded reserves for loss on product purchase commitments with suppliers, we performed the
following procedures:
|
|
—
|
Read relevant contracts and compared key provisions of the contracts to the Company’s analysis.
|
|
—
|
Recalculated the Company’s analysis of the losses including comparing the fixed minimum purchases to the contracts.
|
|
—
|
Obtained and evaluated the Company’s projected sales of inventory as it relates to the minimum purchase commitments by
performing the following:
|
|
•
|
Compared management’s prior-year assumptions of expected future sales to actual sales during the current year to identify
potential bias in the determination of the reserves.
|
|
•
|
Compared projections to recent sales history and related trends
|
|
•
|
Compared projections to industry information, market data, and peer group data.
|
|
•
|
Inspected minutes of the board of directors, regulatory and other public filings, and investor communications to identify any
evidence that may contradict management’s assertions.
|
|
•
|
Obtained evidence, including executed third party contracts used by management to support sales strategies reflected in the
analysis.
|
|
•
|
Inquired of sales and operations personnel regarding projections and strategies to determine whether it supported or
contradicted the conclusions reached by management in the analysis.
|
|
•
|
Inquired of operations personnel as to projected technology obsolescence to determine whether it supported or contradicted
the conclusions reached by management in the analysis.
|
|
|
| |
December 31,
|
|||
|
|
| |
2022
|
| |
2021
|
|
Assets
|
| |
|
| |
|
|
Current assets:
|
| |
|
| |
|
|
Cash and cash equivalents
|
| |
$
|
| |
$
|
|
Marketable securities
|
| |
|
| |
—
|
|
Accounts receivable, net
|
| |
|
| |
|
|
Inventories
|
| |
|
| |
|
|
Current portion of vendor advances
|
| |
|
| |
|
|
Prepaid expenses and other current assets
|
| |
|
| |
|
|
Total current assets
|
| |
|
| |
|
|
Property and equipment, net
|
| |
|
| |
|
|
Non-current portion of vendor advances
|
| |
—
|
| |
|
|
Operating lease assets
|
| |
|
| |
|
|
Other non-current assets
|
| |
|
| |
|
|
Total assets
|
| |
$
|
| |
$
|
|
Liabilities and stockholders’ equity
|
| |
|
| |
|
|
Current liabilities:
|
| |
|
| |
|
|
Accounts payable
|
| |
$
|
| |
$
|
|
Deferred revenue, current
|
| |
|
| |
|
|
Accrued purchase commitments, current
|
| |
|
| |
|
|
Accrued expenses and other current liabilities
|
| |
|
| |
|
|
Total current liabilities
|
| |
|
| |
|
|
Deferred revenue, non-current
|
| |
|
| |
|
|
Warrant liabilities
|
| |
|
| |
|
|
Accrued purchase commitments, non-current
|
| |
—
|
| |
|
|
Operating lease liabilities
|
| |
|
| |
|
|
Other non-current liabilities
|
| |
|
| |
|
|
Total liabilities
|
| |
|
| |
|
|
Commitments and contingencies (Note 19)
|
| | | | ||
|
Stockholders’ equity:
|
| |
|
| |
|
|
Class A common stock $
|
| |
|
| |
|
|
Class B common stock $
|
| |
|
| |
|
|
Additional paid-in capital
|
| |
|
| |
|
|
Accumulated deficit
|
| |
(
|
| |
(
|
|
Total stockholders’ equity
|
| |
|
| |
|
|
Total liabilities and stockholders’ equity
|
| |
$
|
| |
$
|
|
|
| |
Year ended December 31,
|
||||||
|
|
| |
2022
|
| |
2021
|
| |
2020
|
|
Revenue:
|
| |
|
| |
|
| |
|
|
Product
|
| |
$
|
| |
$
|
| |
$
|
|
Software and other services
|
| |
|
| |
|
| |
|
|
Total revenue
|
| |
|
|
|
| |
|
|
|
Cost of revenue:
|
| |
|
| |
|
| |
|
|
Product
|
| |
|
| |
|
| |
|
|
Software and other services
|
| |
|
| |
|
| |
|
|
Loss on product purchase commitments
|
| |
—
|
| |
|
| |
|
|
Total cost of revenue
|
| |
|
| |
|
| |
|
|
Gross profit (loss)
|
| |
|
| |
|
| |
(
|
|
Operating expenses:
|
| |
|
| |
|
| |
|
|
Research and development
|
| |
|
| |
|
| |
|
|
Sales and marketing
|
| |
|
| |
|
| |
|
|
General and administrative
|
| |
|
| |
|
| |
|
|
Total operating expenses
|
| |
|
| |
|
| |
|
|
Loss from operations
|
| |
(
|
| |
(
|
| |
(
|
|
Interest income
|
| |
|
| |
|
| |
|
|
Interest expense
|
| |
(
|
| |
(
|
| |
(
|
|
Change in fair value of warrant liabilities
|
| |
|
| |
|
| |
—
|
|
Other income (expense), net
|
| |
|
| |
(
|
| |
(
|
|
Loss before provision for income taxes
|
| |
(
|
| |
(
|
| |
(
|
|
Provision for income taxes
|
| |
|
| |
|
| |
|
|
Net loss and comprehensive loss
|
| |
$(
|
| |
$(
|
| |
$(
|
|
Net loss per common share attributable to Class A and B
common stockholders, basic and diluted
|
| |
$(
|
| |
$(
|
| |
$(
|
|
Weighted-average shares used to compute net loss per share
attributable to Class A and B common stockholders, basic and diluted
|
| |
|
| |
|
| |
|
|
|
| |
Convertible
Preferred
Stock
|
| |
Class A
Common
Stock
|
| |
Class B
Common
Stock
|
| |
Additional
Paid-In
Capital
|
| |
Accumulated
Deficit
|
| |
Total
Stockholders’
Equity (Deficit)
|
|||||||||
|
|
| |
Shares
|
| |
Amount
|
| |
Shares
|
| |
Amount
|
| |
Shares
|
| |
Amount
|
| ||||||||
|
December 31, 2019
|
| |
|
| |
$
|
| |
|
| |
$
|
| |
—
|
| |
$—
|
| |
$
|
| |
$(
|
|
$(
|
|
|
Net loss
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
(
|
| |
(
|
|
Common stock issued upon exercise of stock options
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
|
|
Stock-based compensation expense
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
|
|
December 31, 2020
|
| |
|
| |
|
| |
|
| |
|
| |
—
|
| |
—
|
| |
|
| |
(
|
| |
(
|
|
Net loss
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
(
|
| |
(
|
|
Common stock issued upon exercise of stock options and
warrants
|
| |
—
|
| |
—
|
| |
|
| |
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
|
|
Common stock issued upon vesting of restricted stock
units
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
|
Conversion of convertible preferred stock
|
| |
(
|
| |
(
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
—
|
| |
|
|
Conversion of convertible debt
|
| |
—
|
| |
—
|
| |
|
| |
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
|
|
Net equity infusion from the Business Combination
|
| |
—
|
| |
—
|
| |
|
| |
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
|
|
Stock-based compensation expense
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
|
|
December 31, 2021
|
| |
—
|
| |
—
|
| |
|
| |
|
| |
|
|
|
| |
|
| |
(
|
| |
|
|
|
Net loss
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
(
|
|
(
|
|
|
Common stock issued upon exercise of stock options and
warrants
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
|
|
Common stock issued upon vesting of restricted stock
units
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
—
|
| |
—
|
| |
(
|
| |
—
|
| |
(
|
|
Stock-based compensation expense
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
—
|
| |
|
| |
—
|
| |
|
|
December 31, 2022
|
| |
—
|
| |
$—
|
| |
|
| |
$
|
| |
|
| |
$
|
| |
$
|
| |
$(
|
| |
$
|
|
|
| |
Year ended December 31,
|
||||||
|
|
| |
2022
|
| |
2021
|
| |
2020
|
|
Cash flows from operating activities:
|
| |
|
| |
|
| |
|
|
Net loss
|
| |
$(
|
| |
$(
|
| |
$(
|
|
Adjustments to reconcile net loss to net cash used in operating activities:
|
| |
|
| |
|
| |
|
|
Depreciation and amortization
|
| |
|
| |
|
| |
|
|
Write-down of vendor advance
|
| |
—
|
| |
|
| |
|
|
Non-cash interest expense on convertible debt
|
| |
—
|
| |
|
| |
|
|
Write-down of inventories
|
| |
|
| |
|
| |
|
|
Stock-based compensation expense
|
| |
|
| |
|
| |
|
|
Change in fair value of warrant liabilities
|
| |
(
|
| |
(
|
| |
—
|
|
Other
|
| |
|
| |
|
| |
|
|
Changes in operating assets and liabilities:
|
| |
|
| |
|
| |
|
|
Accounts receivable
|
| |
(
|
| |
(
|
| |
(
|
|
Inventories
|
| |
(
|
| |
(
|
| |
(
|
|
Prepaid expenses and other assets
|
| |
|
| |
(
|
| |
(
|
|
Vendor advances
|
| |
|
| |
(
|
| |
|
|
Accounts payable
|
| |
|
| |
(
|
| |
|
|
Deferred revenue
|
| |
|
| |
|
| |
|
|
Accrued purchase commitments
|
| |
(
|
| |
(
|
| |
|
|
Change in operating lease assets and liabilities
|
| |
|
| |
|
| |
—
|
|
Accrued expenses and other liabilities
|
| |
|
| |
|
| |
|
|
Net cash used in operating activities
|
| |
(
|
| |
(
|
| |
(
|
|
|
| |
|
| |
|
| |
|
|
Cash flows from investing activities:
|
| |
|
| |
|
| |
|
|
Purchases of marketable securities
|
| |
(
|
| |
(
|
| |
—
|
|
Sales of marketable securities
|
| |
—
|
| |
|
| |
—
|
|
Purchases of property and equipment, including capitalized software
|
| |
(
|
| |
(
|
| |
(
|
|
Sales of property and equipment
|
| |
|
| |
—
|
| |
—
|
|
Net cash used in investing activities
|
| |
(
|
| |
(
|
| |
(
|
|
|
| |
|
| |
|
| |
|
|
Cash flows from financing activities:
|
| |
|
| |
|
| |
|
|
Proceeds from exercise of stock options and warrants
|
| |
|
| |
|
| |
|
|
Net proceeds from equity infusion from the Business Combination
|
| |
—
|
| |
|
| |
(
|
|
Proceeds from loan payable
|
| |
—
|
| |
—
|
| |
|
|
Proceeds from issuance of convertible debt
|
| |
—
|
| |
—
|
| |
|
|
Payment of loan payable
|
| |
—
|
| |
(
|
| |
—
|
|
Payments of debt issuance costs
|
| |
—
|
| |
(
|
| |
(
|
|
Other financing activities
|
| |
(
|
| |
—
|
| |
—
|
|
Net cash provided by financing activities
|
| |
|
| |
|
| |
|
|
Net (decrease) increase in cash, cash equivalents and
restricted cash
|
| |
(
|
| |
|
| |
(
|
|
Cash, cash equivalents and restricted cash, beginning of period
|
| |
|
| |
|
| |
|
|
Cash, cash equivalents and restricted cash, end of period
|
| |
$
|
|
$
|
| |
$
|
|
|
|
| |
|
| |
|
| |
|
|
Supplementary disclosure of non-cash investing and financing
activities
|
| |
|
| |
|
| |
|
|
Purchase of property and equipment
|
| |
$
|
| |
$
|
| |
$
|
|
Deferred offering costs and debt issuance costs
|
| |
—
|
| |
—
|
| |
|
|
•
|
revenue recognition, including determination of the timing and pattern of satisfaction of performance obligations and
determination of the standalone selling price (“SSP”) of performance obligations;
|
|
•
|
assumptions underlying the warranty liability calculation;
|
|
•
|
assumptions underlying the measurement of the purchase commitment loss;
|
|
•
|
measurement and allocation of capitalized costs, the net realizable value (the selling price as well as estimated costs of
completion, disposal and transportation) of inventory, and demand and future use of inventory;
|
|
•
|
assumptions underlying the incremental borrowing rate calculation;
|
|
•
|
assumptions underlying the warrant liability calculation;
|
|
•
|
valuation allowances with respect to deferred tax assets; and
|
|
•
|
assumptions underlying the fair value used in the stock-based compensation expense calculation.
|
|
•
|
Step 1: Identify Contracts with Customers: The Company typically enters into
contracts with customers through direct sales executed via signed contracts with payment terms of 60 days or less. Multi-year software subscriptions typically require advance payment for each annual subscription period.
|
|
•
|
Step 2: Identify Performance Obligations: The Company’s contracts with customers
often include multiple performance obligations. The Company has identified the following performance obligations in its contracts with customers:
|
|
•
|
Hardware devices and accessories
|
|
•
|
Software subscriptions, including renewal subscriptions, which represent an obligation to provide the customer with ongoing
access to the Company’s cloud-hosted software applications on a continuous basis throughout the subscription period
|
|
•
|
Implementation and integration services
|
|
•
|
Extended warranties and customer service
|
|
•
|
Step 3: Determine Transaction Price: The Company’s contracts with customers include
variable consideration in the form of refunds and credits for product returns and price concessions. The Company estimates variable consideration using the expected value method based on a portfolio of data from similar contracts.
|
|
•
|
Step 4: Allocate Transaction Price to Performance Obligations: The Company allocates
transaction price to the performance obligations in contracts with customers based on the relative SSPs of the goods and services. For the software subscriptions and renewal subscriptions, which the Company sells to customers on a
standalone basis, the Company uses the observable SSPs of new and renewal subscriptions, respectively. The Company’s sales of hardware devices and accessories represent single performance obligations.
|
|
•
|
Step 5: Recognize Revenue as Performance Obligations are Satisfied: Each sale of a
hardware device or accessory is a performance obligation satisfied at a point in time when control of the good transfers from the Company to the customer. The Company’s software subscriptions, extended warranties and customer service are
stand-ready performance obligations that are satisfied over time by providing the customer with ongoing access to the Company’s resources. The Company uses the time-elapsed (i.e., straight-line) measure of progress to recognize revenue as
these performance obligations are satisfied evenly over the respective service periods. The implementation and integration services are a performance obligation satisfied over time, and the Company uses the costs incurred as inputs into
the measure of progress to recognize revenue as it satisfies the performance obligation.
|
|
(in thousands)
|
| |
Fair Value
|
|
Allowance for doubtful accounts as of December 31, 2020
|
| |
$
|
|
Additions (recoveries)
|
| |
(
|
|
Deductions – write offs
|
| |
(
|
|
Allowance for doubtful accounts as of December 31, 2021
|
| |
$
|
|
Additions (recoveries)
|
| |
|
|
Deductions – write offs
|
| |
(
|
|
Allowance for doubtful accounts as of December 31, 2022
|
| |
$
|
|
Property and Equipment
|
| |
Estimated Useful Life
|
|
Software
|
| |
|
|
Machinery and equipment
|
| |
|
|
Furniture and fixtures
|
| |
|
|
Leasehold improvements
|
| |
Lesser of estimated useful life or remaining lease term
|
|
|
| |
Pattern of
Recognition
|
| |
2022
|
| |
2021
|
| |
2020
|
|
By Product Type:
|
| |
|
| |
|
| |
|
| |
|
|
Devices and accessories
|
| |
Point-in-time
|
| |
$
|
| |
$
|
| |
$
|
|
Software and other services
|
| |
Over time
|
| |
|
| |
|
| |
|
|
Total revenue
|
| |
|
| |
$
|
| |
$
|
| |
$
|
|
By Geographical Market:
|
| |
|
| |
|
| |
|
| |
|
|
United States
|
| |
|
| |
$
|
| |
$
|
| |
$
|
|
International
|
| |
|
| |
|
| |
|
| |
|
|
Total revenue
|
| |
|
| |
$
|
| |
$
|
| |
$
|
|
•
|
Level 1 — Valuations based on quoted prices in active markets for identical assets or
liabilities that an entity has the ability to access.
|
|
•
|
Level 2 — Valuations based on quoted prices for similar assets or liabilities, quoted
prices for identical assets or liabilities in markets that are not active, or other inputs that are observable or can be corroborated by observable data for substantially the full term of the assets or liabilities.
|
|
•
|
Level 3 — Valuations based on inputs that are supported by little or no market
activity and that are significant to the fair value of the assets or liabilities. The Company has no assets or liabilities valued with Level 3 inputs.
|
|
|
| |
|
| |
Fair Value Measurement Level
|
||||||
|
|
| |
Total
|
| |
Level 1
|
| |
Level 2
|
| |
Level 3
|
|
December 31, 2022:
|
| |
|
| |
|
| |
|
| |
|
|
Marketable securities:
|
| |
|
| |
|
| |
|
| |
|
|
Mutual funds
|
| |
$
|
| |
$
|
| |
$—
|
| |
$—
|
|
Total assets at fair value on a recurring basis
|
| |
$
|
| |
$
|
| |
$—
|
| |
$—
|
|
|
| |
|
| |
|
| |
|
| |
|
|
Warrants:
|
| |
|
| |
|
| |
|
| |
|
|
Public Warrants
|
| |
$
|
| |
$
|
| |
$—
|
| |
$—
|
|
Private Warrants
|
| |
|
| |
—
|
| |
|
| |
—
|
|
Total liabilities at fair value on a recurring basis
|
| |
$
|
| |
$
|
| |
$
|
| |
$—
|
|
|
| |
|
| |
|
| |
|
| |
|
|
December 31, 2021:
|
| |
|
| |
|
| |
|
| |
|
|
Warrants:
|
| |
|
| |
|
| |
|
| |
|
|
Public Warrants
|
| |
$
|
| |
$
|
| |
$—
|
| |
$—
|
|
Private Warrants
|
| |
|
| |
—
|
| |
|
| |
—
|
|
Total liabilities at fair value on a recurring basis
|
| |
$
|
| |
$
|
| |
$
|
| |
$—
|
|
|
| |
December 31,
2022
|
| |
December 31,
2021
|
|
Raw materials
|
| |
$
|
| |
|
|
Work-in-progress
|
| |
|
| |
|
|
Finished goods
|
| |
|
| |
|
|
Total inventories
|
| |
$
|
| |
$
|
|
|
| |
December 31,
|
|||
|
|
| |
2022
|
| |
2021
|
|
Reconciliation of cash, cash equivalents and restricted cash:
|
| |
|
| |
|
|
Cash and cash equivalents
|
| |
$
|
| |
$
|
|
|
| |
|
| |
—
|
|
|
| |
|
| |
|
|
Total cash, cash equivalents and
restricted cash shown in the consolidated statements of cash flows
|
| |
$
|
| |
$
|
|
|
| |
December 31,
2022
|
| |
December 31,
2021
|
|
Security deposits
|
| |
$
|
| |
$
|
|
|
| |
|
| |
|
|
Other long-term assets
|
| |
|
| |
|
|
Total other non-current assets
|
| |
$
|
| |
$
|
|
|
| |
December 31,
2022
|
| |
December 31,
2021
|
|
Software
|
| |
$
|
| |
$
|
|
Leasehold improvements
|
| |
|
| |
|
|
Machinery and equipment
|
| |
|
| |
|
|
Furniture and fixtures
|
| |
|
| |
|
|
Construction in progress
|
| |
|
| |
|
|
Other
|
| |
|
| |
|
|
|
| |
|
| |
|
|
Less: accumulated depreciation and amortization
|
| |
(
|
| |
(
|
|
Property and equipment, net
|
| |
$
|
| |
$
|
|
|
| |
2023
|
| |
2024
|
| |
2025
|
| |
2026
|
| |
2027
|
|
Software
|
| |
$
|
| |
$
|
| |
$
|
| |
$—
|
| |
$—
|
|
|
| |
December 31,
2022
|
| |
December 31,
2021
|
|
Employee compensation
|
| |
$
|
| |
$
|
|
Customer deposits
|
| |
|
| |
|
|
Accrued warranty liability
|
| |
|
| |
|
|
Non-income tax
|
| |
|
| |
|
|
Professional fees
|
| |
|
| |
|
|
|
| |
|
| |
|
|
Other
|
| |
|
| |
|
|
Total accrued expenses and other current liabilities
|
| |
$
|
| |
$
|
|
|
| |
2022
|
| |
2021
|
| |
2020
|
|
Balance, beginning of period
|
| |
$
|
| |
$
|
| |
$
|
|
Warranty provision charged to operations
|
| |
|
| |
|
| |
|
|
Warranty claims
|
| |
(
|
| |
(
|
| |
(
|
|
Balance, end of period
|
| |
$
|
| |
$
|
| |
$
|
|
(1)
|
Any sale, assignment, transfer, conveyance, hypothecation, or other transfer or disposition, directly or indirectly, of any
Class B common stock or any legal or beneficial interest in such share, whether or not for value and whether voluntary or involuntary or by operation of law (including by merger, consolidation, or otherwise), including, without limitation
the transfer of a share of Class B common stock to a broker or other nominee or the transfer of, or entering into a binding agreement with respect to, voting control over such share by proxy or otherwise, other than a permitted transfer.
|
|
(2)
|
Upon the first date on which Dr. Rothberg, together with all other qualified stockholders, collectively cease to beneficially
own at least
|
|
(3)
|
Upon the date specified by the affirmative vote of the holders of at least (2/3) of the outstanding shares of Class B common stock, voting as a separate class.
|
|
|
| |
Number of
Options
|
| |
Weighted
Average
Exercise
Price
|
| |
Weighted
Average
Remaining
Contractual
Term
|
| |
Aggregate
Intrinsic
Value
(in thousands)
|
|
Outstanding at December 31, 2020
|
| |
|
| |
|
| |
|
| |
|
|
Granted
|
| |
|
| |
|
| |
|
| |
|
|
Exercised
|
| |
(
|
| |
|
| |
|
| |
|
|
Forfeited
|
| |
(
|
| |
|
| |
|
| |
|
|
Outstanding at December 31, 2021
|
| |
|
| |
|
| |
|
| |
|
|
Granted
|
| |
|
| |
|
| |
|
| |
|
|
Exercised
|
| |
(
|
| |
|
| |
|
| |
|
|
Forfeited
|
| |
(
|
| |
|
| |
|
| |
|
|
Outstanding at December 31, 2022
|
| |
|
| |
|
| |
|
| |
|
|
Options exercisable at December 31, 2021
|
| |
|
| |
|
| |
|
| |
|
|
Options exercisable at December 31, 2022
|
| |
|
| |
|
| |
|
| |
|
|
Vested and expected to vest at December 31, 2021
|
| |
|
| |
|
| |
|
| |
|
|
Vested and expected to vest at December 31, 2022
|
| |
|
| |
|
| |
|
| |
|
|
|
| |
2022
|
| |
2021
|
| |
2020
|
|
Cash proceeds from the exercise of stock options (in millions)
|
| |
$
|
| |
$
|
| |
$
|
|
Total intrinsic value of stock options exercised (in millions)
|
| |
|
| |
|
| |
|
|
Weighted average grant date fair value of options granted
|
| |
|
| |
|
| |
|
|
|
| |
2022
|
| |
2021
|
| |
2020
|
|
Risk-free interest rate
|
| |
|
| |
|
| |
|
|
Expected dividend yield
|
| |
|
| |
|
| |
|
|
Expected term
|
| |
|
| |
|
| |
|
|
Expected volatility
|
| |
|
| |
|
| |
|
|
|
| |
2020
|
|
Risk-free interest rate
|
| |
|
|
Expected dividend yield
|
| |
|
|
Expected term
|
| |
|
|
Expected volatility
|
| |
|
|
|
| |
Number of
Restricted
Stock Units
|
| |
Weighted
Average
Grant Date
Fair Value
|
|
Outstanding at December 31, 2020
|
| |
|
| |
|
|
Granted
|
| |
|
| |
|
|
Vested
|
| |
(
|
| |
|
|
Forfeited
|
| |
(
|
| |
|
|
Outstanding at December 31, 2021
|
| |
|
| |
|
|
Granted
|
| |
|
| |
|
|
Vested
|
| |
(
|
| |
|
|
Forfeited
|
| |
(
|
| |
|
|
Outstanding at December 31, 2022
|
| |
|
| |
|
|
|
| |
Year ended December 31,
|
||||||
|
|
| |
2022
|
| |
2021
|
| |
2020
|
|
Cost of revenue – software and other services
|
| |
$
|
| |
$
|
| |
$
|
|
Research and development
|
| |
|
| |
|
| |
|
|
Sales and marketing
|
| |
|
| |
|
| |
|
|
General and administrative
|
| |
|
| |
|
| |
|
|
Total stock-based compensation expense
|
| |
$
|
| |
$
|
| |
$
|
|
Year ended December 31, 2022
|
| |
|
| |
|
| ||
|
|
| |
Class A
|
| |
Class B
|
| |
Total
Common Stock
|
|
Numerator:
|
| |
|
| |
|
| |
|
|
Allocation of undistributed earnings
|
| |
$(
|
| |
$(
|
| |
$(
|
|
Numerator for basic and diluted net loss
per share – loss available to common stockholders
|
| |
$(
|
| |
$(
|
| |
$(
|
|
Denominator:
|
| |
|
| |
|
| |
|
|
Weighted-average common shares outstanding
|
| |
|
| |
|
| |
|
|
Denominator for basic and diluted net
loss per share – weighted-average common stock
|
| |
|
| |
|
| |
|
|
Basic and diluted net loss per share
|
| |
$(
|
| |
$(
|
| |
$(
|
|
Year ended December 31, 2021
|
| |
|
| |
|
| ||
|
|
| |
Class A
|
| |
Class B
|
| |
Total
Common Stock
|
|
Numerator:
|
| |
|
| |
|
| |
|
|
Allocation of undistributed earnings
|
| |
$(
|
| |
$(
|
| |
$(
|
|
Numerator for basic and diluted net loss
per share – loss available to common stockholders
|
| |
$(
|
| |
$(
|
| |
$(
|
|
Denominator:
|
| |
|
| |
|
| |
|
|
Weighted-average common shares outstanding
|
| |
|
| |
|
| |
|
|
Denominator for basic and diluted net
loss per share – weighted-average common stock
|
| |
|
| |
|
| |
|
|
Basic and diluted net loss per share
|
| |
$(
|
| |
$(
|
| |
$(
|
|
Year ended December 31, 2020
|
| |
|
|
Numerator:
|
| |
|
|
Allocation of undistributed earnings
|
| |
$(
|
|
Numerator for basic and diluted net loss per share – loss
available to common stockholders
|
| |
$(
|
|
Denominator:
|
| |
|
|
Weighted-average common shares outstanding
|
| |
|
|
Denominator for basic and diluted net loss per share –
weighted-average common stock
|
| |
|
|
Basic and diluted net loss per share
|
| |
$(
|
|
|
| |
2022
|
| |
2021
|
| |
2020
|
|
Outstanding options to purchase common stock
|
| |
|
| |
|
| |
|
|
Outstanding restricted stock units
|
| |
|
| |
|
| |
|
|
Outstanding warrants
|
| |
|
| |
|
| |
—
|
|
Outstanding convertible preferred stock (Series A through D)
|
| |
—
|
| |
—
|
| |
|
|
Total anti-dilutive common equivalent shares
|
| |
|
| |
|
| |
|
|
|
| |
Year ended December 31,
|
||||||
|
|
| |
2022
|
| |
2021
|
| |
2020
|
|
Federal
|
| |
$(
|
| |
$(
|
| |
$(
|
|
Foreign
|
| |
|
| |
|
| |
|
|
Loss before provision for income taxes
|
| |
$(
|
| |
$(
|
| |
$(
|
|
|
| |
Year ended December 31,
|
||||||
|
|
| |
2022
|
| |
2021
|
| |
2020
|
|
Income at US statutory rate
|
| |
|
| |
|
| |
|
|
State taxes, net of federal benefit
|
| |
|
| |
|
| |
|
|
Stock compensation
|
| |
(
|
| |
(
|
| |
|
|
Change in fair value of warrants
|
| |
|
| |
|
| |
|
|
Tax credits
|
| |
|
| |
|
| |
|
|
Foreign rate differential
|
| |
|
| |
|
| |
|
|
Valuation allowance
|
| |
(
|
| |
(
|
| |
(
|
|
Other
|
| |
(
|
| |
(
|
| |
(
|
|
|
| |
(
|
| |
(
|
| |
(
|
|
|
| |
Year ended December 31,
|
|||
|
|
| |
2022
|
| |
2021
|
|
Deferred tax assets
|
| |
|
| |
|
|
Net operating loss carryforwards
|
| |
$
|
| |
$
|
|
Tax credits
|
| |
|
| |
|
|
Stock compensation
|
| |
|
| |
|
|
Accruals and reserves
|
| |
|
| |
|
|
Inventory reserve
|
| |
|
| |
|
|
Lease liability
|
| |
|
| |
|
|
Depreciation
|
| |
|
| |
|
|
Capitalized tax R&E
|
| |
|
| |
—
|
|
Other
|
| |
|
| |
|
|
Total deferred tax assets
|
| |
$
|
| |
$
|
|
Valuation allowance
|
| |
(
|
| |
(
|
|
Total deferred tax assets
|
| |
$
|
| |
$
|
|
Deferred tax liabilities
|
| |
|
| |
|
|
Right-of-use asset
|
| |
(
|
| |
(
|
|
Net deferred tax assets
|
| |
$—
|
| |
$—
|
|
|
| |
Year ended December 31,
|
||||||
|
|
| |
2022
|
| |
2021
|
| |
2020
|
|
Total incurred for operating expenses
|
| |
$
|
| |
$
|
| |
$
|
|
|
| |
December 31,
|
|||
|
|
| |
2022
|
| |
2021
|
|
Due from related parties
|
| |
$
|
| |
$—
|
|
Due to related parties
|
| |
—
|
| |
|
|
|
| |
Year ended
December 31, 2022
|
|
Research and development
|
| |
$
|
|
Sales and marketing
|
| |
|
|
General and administrative
|
| |
|
|
Total employee severance and benefits costs
|
| |
$
|
|
•
|
at a price of $
|
|
•
|
upon a minimum of
|
|
•
|
provided that the last reported sale price of the Class A common stock equals or exceeds $
|
|
•
|
provided that there is an effective registration statement covering the issuance of the shares of Class A common stock issuable
upon exercise of the warrants, and a current prospectus relating thereto, available through the
|
|
•
|
at $
|
|
•
|
upon a minimum of 30 days’ prior written notice of redemption;
|
|
•
|
provided that the last reported sale price of the Class A common stock equals or exceeds $
|
|
•
|
provided that the Private Warrants are also concurrently exchanged at the same price (equal to a number of shares of Class A
common stock) as the outstanding Public Warrants; and
|
|
•
|
provided that there is an effective registration statement covering the issuance of the shares of Class A common stock issuable
upon exercise of the warrants and a current prospectus relating thereto available throughout the
|
|
|
| |
Year ended December 31,
|
|||
|
|
| |
2022
|
| |
2021
|
|
Operating lease cost
|
| |
$
|
| |
$
|
|
Short-term lease cost
|
| |
|
| |
|
|
Variable lease cost
|
| |
|
| |
|
|
Total operating lease cost
|
| |
$
|
| |
$
|
|
Year ended December 31,
|
| |
Operating Lease Payments
|
|
2023
|
| |
$
|
|
2024
|
| |
|
|
2025
|
| |
|
|
2026
|
| |
|
|
2027
|
| |
|
|
2027 and thereafter
|
| |
|
|
Total gross operating lease payments
|
| |
|
|
Less: imputed interest
|
| |
(
|
|
Total operating lease liabilities,
reflecting the present value of net lease payments
|
| |
$
|
|
|
| |
December 31,
|
|||
|
|
| |
2022
|
| |
2021
|
|
Weighted average remaining lease term (in years)
|
| |
|
| |
|
|
Weighted average discount rate
|
| |
|
| |
|
|
|
| |
Year ended December 31,
|
|||
|
|
| |
2022
|
| |
2021
|
|
Cash paid for amounts included in the measurement of lease liabilities:
|
| |
|
| |
|
|
Operating lease payments, included in cash flows from operating activities
|
| |
$
|
| |
$
|
|
Non-cash additions to operating lease assets
|
| |
$—
|
| |
$
|
|
Item 13.
|
Other Expenses of Issuance and Distribution.
|
|
Expense
|
| |
Estimated
Amount
|
|
Securities and Exchange Commission registration fee
|
| |
$261,844.59
|
|
Accounting fees and expenses
|
| |
*
|
|
Legal fees and expenses
|
| |
*
|
|
Financial printing and miscellaneous expenses
|
| |
*
|
|
Total
|
| |
$*
|
|
*
|
These fees are calculated based on the securities offered and the number of issuances and accordingly cannot be defined at this
time.
|
|
Item 14.
|
Indemnification of Directors and Officers.
|
|
Item 15.
|
Recent Sales of Unregistered Securities.
|
|
Item 16.
|
Exhibits and Financial Statement Schedules.
|
|
(a)
|
Exhibits.
|
|
Exhibit
Number
|
| |
Exhibit Description
|
| |
Filed
Herewith
|
| |
Incorporated by
Reference herein
from Form or
Schedule
|
| |
Filing Date
|
| |
SEC File/
Reg.
Number
|
| | |
Business Combination Agreement, dated as of November 19, 2020, by and among
Butterfly Network, Inc. (formerly Longview Acquisition Corp.), Clay Merger Sub, Inc., and BFLY Operations, Inc. (formerly Butterfly Network, Inc.).
|
| |
|
| |
Form 8-K
(Exhibit 2.1)
|
| |
11/23/2020
|
| |
001-39292
|
|
| | |
Second Amended and Restated Certificate of Incorporation of Butterfly Network,
Inc.
|
| |
|
| |
Form 8-K
(Exhibit 3.1)
|
| |
2/16/2021
|
| |
001-39292
|
|
| | |
Amended and Restated Bylaws of Butterfly Network, Inc.
|
| |
|
| |
Form 8-K
(Exhibit 3.2)
|
| |
2/16/2021
|
| |
001-39292
|
|
| | |
Specimen Class A Common Stock Certificate.
|
| |
|
| |
Form 8-K
(Exhibit 4.1)
|
| |
2/16/2021
|
| |
001-39292
|
|
| | |
Specimen Class B Common Stock Certificate
|
| |
|
| |
POS AM
(Exhibit 4.2)
|
| |
4/19/2022
|
| |
333-254836
|
|
| | |
Warrant Agreement, dated as of May 20, 2020, by and between Butterfly Network,
Inc. (formerly Longview Acquisition Corp.) and Continental Stock Transfer & Trust Company.
|
| |
|
| |
Form 8-K
(Exhibit 4.1)
|
| |
5/27/2020
|
| |
001-39292
|
|
| | |
Opinion of Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C.
|
| |
|
| |
Form S-1/A
(Exhibit 5.1)
|
| |
5/12/2021
|
| |
333-254836
|
|
| | |
Exclusive (Equity) Agreement by and between BFLY Operations, Inc. (formerly
Butterfly Network, Inc.) and the Board of Trustees of the Leland Stanford Junior University, dated as of June 28, 2013.
|
| |
|
| |
Form S-4
(Exhibit 10.13.1)
|
| |
11/27/2020
|
| |
333-250995
|
|
| | |
Amendment No. 1, made effective as of April 23, 2019, to Exclusive (Equity)
Agreement, dated as of June 28, 2013, by and between BFLY Operations, Inc. (formerly Butterfly Network, Inc.) and the Board of Trustees of the Leland Stanford Junior University.
|
| |
|
| |
Form S-4
(Exhibit 10.13.2)
|
| |
11/27/2020
|
| |
333-250995
|
|
| | |
Manufacture and Supply Agreement, dated as of October 7, 2015, by and between
BFLY Operations, Inc. (formerly Butterfly Network, Inc.) and Benchmark Electronics, Inc.
|
| |
|
| |
Form S-4
(Exhibit 10.14.1)
|
| |
11/27/2020
|
| |
333-250995
|
|
| | |
Amendment No. 1, made effective as of August 2, 2019, to Manufacture and Supply
Agreement, dated as of October 7, 2015, by and between BFLY Operations, Inc. (formerly Butterfly Network, Inc.) and Benchmark Electronics, Inc.
|
| |
|
| |
Form S-4
(Exhibit 10.14.2)
|
| |
11/27/2020
|
| |
333-250995
|
|
Exhibit
Number
|
| |
Exhibit Description
|
| |
Filed
Herewith
|
| |
Incorporated by
Reference herein
from Form or
Schedule
|
| |
Filing Date
|
| |
SEC File/
Reg.
Number
|
| | |
Amendment No. 2, made effective as of February 26, 2021, to Manufacture and
Supply Agreement, dated as of October 7, 2015, by and between BFLY Operations, Inc. (formerly Butterfly Network, Inc.) and Benchmark Electronics, Inc.
|
| |
|
| |
Form 10-K/A
(Exhibit 10.6.3)
|
| |
5/12/2021
|
| |
001-39292
|
|
| | |
Distribution Agreement, dated as of July 11, 2018, by and between BFLY
Operations, Inc. (formerly Butterfly Network, Inc.) and Cardinal Health 105, Inc.
|
| |
|
| |
Form S-4
(Exhibit 10.15)
|
| |
11/27/2020
|
| |
333-250995
|
|
| | |
Foundry Service Agreement, dated as of March 31, 2019, by and between BFLY
Operations, Inc. (formerly Butterfly Network, Inc.) and Taiwan Semiconductor Manufacturing Company Limited.
|
| |
|
| |
Form S-4/A
(Exhibit 10.17.1)
|
| |
1/6/2021
|
| |
333-250995
|
|
| | |
Amendment No. 1, made effective as of October 1, 2020, to Foundry Service
Agreement, dated March 31, 2019, by and between BFLY Operations, Inc. (formerly Butterfly Network, Inc.) and Taiwan Semiconductor Manufacturing Company Limited.
|
| |
|
| |
Form S-4/A
(Exhibit 10.17.2)
|
| |
1/6/2021
|
| |
333-250995
|
|
| | |
Technology and Services Exchange Agreement, dated as of November 19, 2020,
between BFLY Operations, Inc. (formerly Butterfly Network, Inc.) and the participants named therein.
|
| |
|
| |
Form S-4/A
(Exhibit 10.18)
|
| |
1/6/2021
|
| |
333-250995
|
|
| | |
Office Lease Agreement, dated as of May 27, 2021, by and between Butterfly
Network, Inc. and NEEP Investors Holdings LLC.
|
| |
|
| |
Form 8-K/A
(Exhibit 10.1)
|
| |
3/28/2022
|
| |
001-39292
|
|
| | |
Advisory Agreement, dated as of February 12, 2021, by and between Butterfly
Network, Inc. and Jonathan Rothberg, Ph.D.
|
| |
|
| |
Form 10-K
(Exhibit 10.25)
|
| |
3/29/2021
|
| |
001-39292
|
|
| | |
Offer of Employment Letter, dated as of June 3, 2021, by and between Butterfly
Network, Inc. and Andrei G. Stoica.
|
| |
|
| |
Form 10-Q/A
(Exhibit 10.3)
|
| |
3/28/2022
|
| |
001-39292
|
|
| | |
Offer Letter, dated as of April 1, 2022, by and between Butterfly Network, Inc.
and Heather C. Getz.
|
| |
|
| |
Form 10-Q
(Exhibit 10.1)
|
| |
5/6/2022
|
| |
001-39292
|
|
| | |
Offer Letter, dated as of May 14, 2022, by and between Butterfly Network, Inc.
and Lawrence Weiss.
|
| |
|
| |
Form 10-Q
(Exhibit 10.2)
|
| |
8/3/2022
|
| |
001-39292
|
|
| | |
Executive Severance Plan, as amended.
|
| |
|
| |
Form 10-Q/A
(Exhibit 10.3)
|
| |
3/28/2022
|
| |
001-39292
|
|
| | |
Butterfly Network, Inc. Amended and Restated 2020 Equity Incentive Plan.
|
| |
|
| |
Form 10-K
(Exhibit 10.19.1)
|
| |
3/29/2021
|
| |
001-39292
|
|
| | |
Form of Stock Option Agreement under 2020 Equity Incentive Plan.
|
| |
|
| |
Form 8-K
(Exhibit 10.15.2)
|
| |
2/16/2021
|
| |
001-39292
|
|
| | |
Form of Restricted Stock Unit Agreement under 2020 Equity Incentive Plan.
|
| |
|
| |
Form S-8
(Exhibit 99.3)
|
| |
5/12/2021
|
| |
333-256044
|
|
Exhibit
Number
|
| |
Exhibit Description
|
| |
Filed
Herewith
|
| |
Incorporated by
Reference herein
from Form or
Schedule
|
| |
Filing Date
|
| |
SEC File/
Reg.
Number
|
| | |
BFLY Operations, Inc. 2012 Employee, Director and Consultant Equity Incentive
Plan, as amended.
|
| |
|
| |
Form 10-K
(Exhibit 10.20.1)
|
| |
3/29/2021
|
| |
001-39292
|
|
| | |
Form of Stock Option Agreement under 2012 Employee, Director and Consultant
Equity Incentive Plan, as amended.
|
| |
|
| |
Form 8-K
(Exhibit 10.16.2)
|
| |
2/16/2021
|
| |
001-39292
|
|
| | |
Form of Restricted Stock Unit Agreement under 2012 Employee, Director and
Consultant Equity Incentive Plan, as amended.
|
| |
|
| |
Form 8-K
(Exhibit 10.16.3)
|
| |
2/16/2021
|
| |
001-39292
|
|
| | |
Amended and Restated Nonemployee Director Compensation Policy
|
| |
|
| |
Form 10-Q
(Exhibit 10.4)
|
| |
8/3/2022
|
| |
001-39292
|
|
| | |
Form of Indemnification Agreement
|
| |
|
| |
Form 8-K
(Exhibit 10.18)
|
| |
2/16/2021
|
| |
001-39292
|
|
| | |
Amended and Restated Registration Rights Agreement, dated as of February 12,
2021, by and among Butterfly Network, Inc. (formerly Longview Acquisition Corp.), BFLY Operations, Inc. (formerly Butterfly Network, Inc.) and certain of their securityholders
|
| |
|
| |
Form 8-K
(Exhibit 10.19)
|
| |
2/16/2021
|
| |
001-39292
|
|
| | |
List of Subsidiaries
|
| |
|
| |
Form 8-K
(Exhibit 21.1)
|
| |
2/16/2021
|
| |
001-39292
|
|
| | |
Consent of Deloitte & Touche LLP, independent registered public accounting
firm
|
| |
X
|
| |
|
| |
|
| |
|
|
| | |
Consent of Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C. (included in
Exhibit 5.1)
|
| |
|
| |
|
| |
|
| |
|
|
| | |
Power of Attorney (included on the signature page to the initial Registration
Statement)
|
| |
|
| |
|
| |
|
| |
|
|
|
101.INS
|
| |
XBRL Instance Document
|
| |
|
| |
|
| |
|
| |
|
|
101.SCH
|
| |
XBRL Taxonomy Extension Schema Document
|
| |
|
| |
|
| |
|
| |
|
|
101.CAL
|
| |
XBRL Taxonomy Extension Calculation Linkbase Document
|
| |
|
| |
|
| |
|
| |
|
|
101.DEF
|
| |
XBRL Taxonomy Extension Definition Linkbase Document
|
| |
|
| |
|
| |
|
| |
|
|
101.LAB
|
| |
XBRL Taxonomy Extension Label Linkbase Document
|
| |
|
| |
|
| |
|
| |
|
|
101.PRE
|
| |
XBRL Taxonomy Extension Presentation Linkbase Document
|
| |
|
| |
|
| |
|
| |
|
|
104
|
| |
Cover Page Interactive Data File (formatted as Inline XBRL and contained in
Exhibit 101)
|
| |
|
| |
|
| |
|
| |
|
| | |
Filing Fee Table
|
| |
X
|
| |
|
| |
|
| |
|
|
†
|
Certain of the exhibits and schedules to this Exhibit have been omitted in accordance with Regulation S-K Item 601(a)(5). The
Registrant agrees to furnish a copy of all omitted exhibits and schedules to the SEC upon its request.
|
|
+
|
Management contract or compensatory plan or arrangement.
|
|
@
|
Certain confidential portions of this Exhibit were omitted by means of marking such portions with brackets (“[***]”) because the
identified confidential portions (i) are not material and (ii) would be competitively harmful if publicly disclosed.
|
|
Item 17.
|
Undertakings.
|
|
(a)
|
The undersigned registrant hereby undertakes:
|
|
(1)
|
To file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement:
|
|
(i)
|
To include any prospectus required by Section 10(a)(3) of the Securities Act;
|
|
(ii)
|
To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most
recent post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume
of securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the form of
prospectus filed with the Securities and Exchange Commission (the “Commission”) pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than a 20% change in the maximum aggregate offering price set
forth in the “Calculation of Registration Fee” table in the effective registration statement; and
|
|
(iii)
|
To include any material information with respect to the plan of distribution not previously disclosed in the registration
statement or any material change to such information in the registration statement; provided, however, that: Paragraphs (a)(1)(i), (a)(1)(ii) and (a)(1)(iii) of this section do not apply if the
information required to be included in a post-effective amendment by those paragraphs is contained in reports filed with or furnished to the Commission by the registrant pursuant to Section 13 or Section 15(d) of the Securities and
Exchange Act of 1934, as amended (the “Exchange Act”), that are incorporated by reference in the registration statement, or is contained in a form of prospectus filed pursuant to Rule 424(b) that is part of the registration statement.
|
|
(2)
|
That, for the purpose of determining any liability under the Securities Act, each such post-effective amendment shall be deemed
to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
|
|
(3)
|
To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold
at the termination of the offering.
|
|
(4)
|
That, for the purpose of determining liability under the Securities Act to any purchaser:
|
|
(i)
|
each prospectus filed by the registrant pursuant to Rule 424(b)(3) shall be deemed to be part of the registration statement as
of the date the filed prospectus was deemed part of and included in the registration statement; and
|
|
(ii)
|
Each prospectus required to be filed pursuant to Rule 424(b)(2), (b)(5) or (b)(7) as part of a registration statement in
reliance on Rule 430B relating to an offering made pursuant to Rule 415(a)(1)(i), (vii) or (x) for the purpose of providing the information required by Section 10(a) of the Securities Act shall be deemed to be part of and included in the
registration statement as of the earlier of the date such form of prospectus is first used after effectiveness or the date of the first contract of sale of securities in the offering described in the prospectus. As provided in Rule 430B,
for liability purposes of the issuer and any person that is at that date an underwriter, such date shall be deemed to be a new effective date of the registration statement relating to the securities in the registration statement to which
that prospectus relates, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. Provided, however, that no statement made in a registration
statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to
a purchaser with a time of contract of sale prior to such effective date, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document
immediately prior to such effective date.
|
|
(5)
|
That, for the purpose of determining liability of the registrant under the Securities Act to any purchaser in the initial
distribution of the securities, the undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant to this registration statement, regardless of the underwriting method used to sell the
securities to the purchaser, if the securities are offered or sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser and will be considered to offer or sell
such securities to such purchaser:
|
|
(i)
|
Any preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant
to Rule 424;
|
|
(ii)
|
Any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred
to by the undersigned registrant;
|
|
(iii)
|
The portion of any other free writing prospectus relating to the offering containing material information about the undersigned
registrant or its securities provided by or on behalf of the undersigned registrant; and
|
|
(iv)
|
Any other communication that is an offer in the offering made by the undersigned registrant to the purchaser.
|
|
|
| |
BUTTERFLY NETWORK, INC.
|
|||
|
|
| |
|
| |
|
|
|
| |
By:
|
| |
/s/ Joseph DeVivo
|
|
|
| |
|
| |
Joseph DeVivo
|
|
|
| |
|
| |
President, Chief Executive Officer and Chairperson of the Board
|
|
Signature
|
| |
Title
|
| |
Date
|
|
/s/ Joseph DeVivo
|
| |
President, Chief Executive Officer and
Chairperson of the Board
(Principal Executive Officer)
|
| |
May 2, 2023
|
|
Joseph DeVivo
|
| |||||
|
|
| |
|
| |
|
|
/s/ Heather C. Getz
|
| |
Chief Financial Officer
(Principal Financial Officer and Principal Accounting Officer)
|
| |
May 2, 2023
|
|
Heather C. Getz, CPA
|
| |||||
|
|
| |
|
| |
|
|
/s/ Dawn Carfora
|
| |
Director
|
| |
May 2, 2023
|
|
Dawn Carfora
|
| |||||
|
|
| |
|
| |
|
|
/s/ Elazer Edelman
|
| |
Director
|
| |
May 2, 2023
|
|
Elazer Edelman, M.D., Ph.D.
|
| |||||
|
|
| |
|
| |
|
|
/s/ Gianluca Pettiti
|
| |
Director
|
| |
May 2, 2023
|
|
Gianluca Pettiti
|
| |||||
|
|
| |
|
| |
|
|
/s/ S. Louise Phanstiel
|
| |
Director
|
| |
May 2, 2023
|
|
S. Louise Phanstiel
|
| |||||
|
|
| |
|
| |
|
|
/s/ Larry Robbins
|
| |
Director
|
| |
May 2, 2023
|
|
Larry Robbins
|
| |||||
|
|
| |
|
| |
|
|
/s/ Erica Schwartz
|
| |
Director
|
| |
May 2, 2023
|
|
Erica Schwartz, M.D., J.D., M.P.H.
|
| |||||
|
|
| |
|
| |
|
|
/s/ Jonathan M. Rothberg
|
| |
Director
|
| |
May 2, 2023
|
|
Jonathan M. Rothberg, Ph.D.
|
|