SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
THE SECURITIES ACT OF 1933
(Exact Name of Registrant as Specified in its Charter)
State of Israel | | | 3844 | | | Not Applicable |
(State or Other Jurisdiction of Incorporation or Organization) | | | (Primary Standard Industrial Classification Code Number) | | | (I.R.S. Employer Identification No. ) |
Neve Ilan, Israel 9085000
+972 02 995 0506
(Address, including zip code, and telephone number, including
area code, of Registrant’s principal executive offices)
28 Liberty Street
New York, NY 10005
(212) 894-8940
Andrea L. Nicolás Yossi Vebman Skadden, Arps, Slate, Meagher & Flom LLP One Manhattan West New York, New York 10001 Tel: +1-212-735-3000 Fax: +1-212-735-2000 | | | Ian Rostowsky Amit, Pollak, Matalon & Co. APM House, 18 Raoul Wallenberg St. Building D. Ramat Hachayal Tel Aviv 6971915, Israel Tel: +972-3-568-9000 Fax: +972-73-297-8645 | | | Peter N. Handrinos Wesley C. Holmes Latham & Watkins LLP 200 Clarendon Street Boston, Massachusetts 02116 Tel: +1-617-948-6000 Fax: +1-617-948-6001 | | | Chaim Friedland Ari Fried Gornitzky & Co. Zion House 45 Rothschild Blvd. Tel Aviv 6578403, Israel Tel. +972-3-710-9191 Fax: +972-3-560-6555 |
Title of each Class of Securities to be Registered | | | Proposed Maximum Aggregate Offering Price(1)(2) | | | Amount of Registration Fee(3) |
Ordinary shares, par value NIS 0.01 per share | | | $125,000,000 | | | $16,225.00 |
(1) | Estimated solely for the purpose of calculating the registration fee pursuant to Rule 457(o) under the Securities Act of 1933, as amended (the “Securities Act”). |
(2) | Includes ordinary shares that the underwriters may purchase pursuant to their option to purchase additional ordinary shares. |
(3) | Calculated pursuant to Rule 457(o) under the Securities Act based on an estimate of the proposed maximum aggregate offering price. |

| | | Per share | | | Total | |
Initial public offering price | | | $ | | | $ |
Underwriting discounts and commissions(1) | | | $ | | | $ |
Proceeds to us (before expenses) | | | $ | | | $ |
(1) | Refer to “Underwriting” for additional information regarding underwriting compensation. |
Cantor | | | Oppenheimer & Co. | | | Berenberg | | | CIBC Capital Markets |
| | | Page | |
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | |
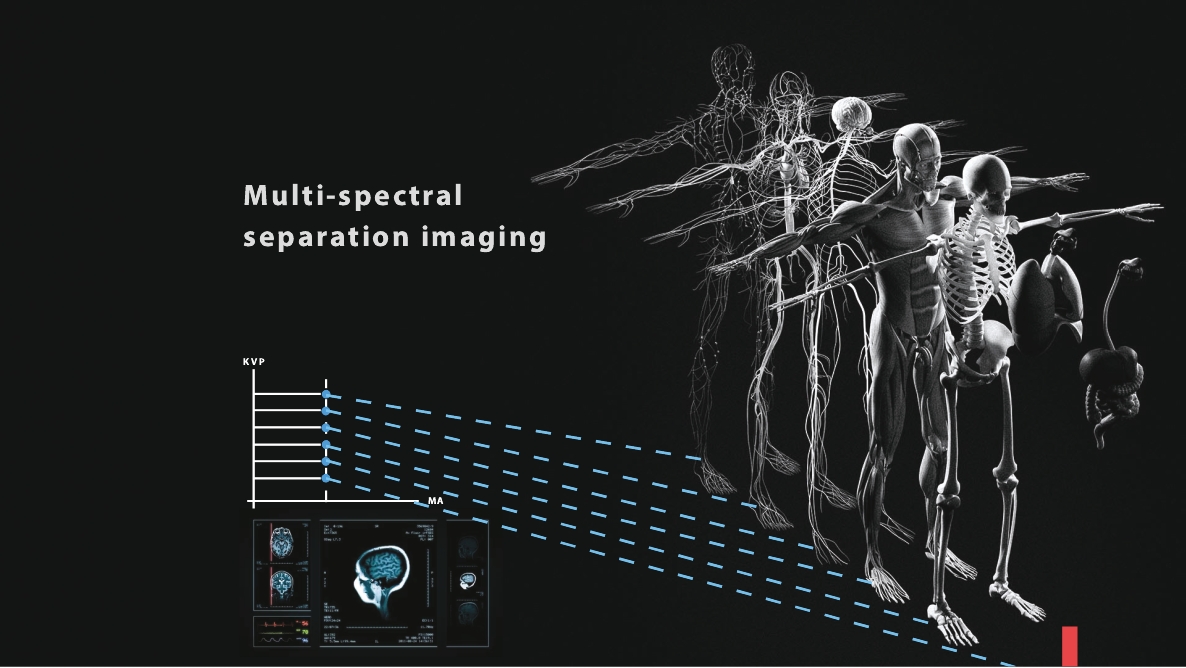
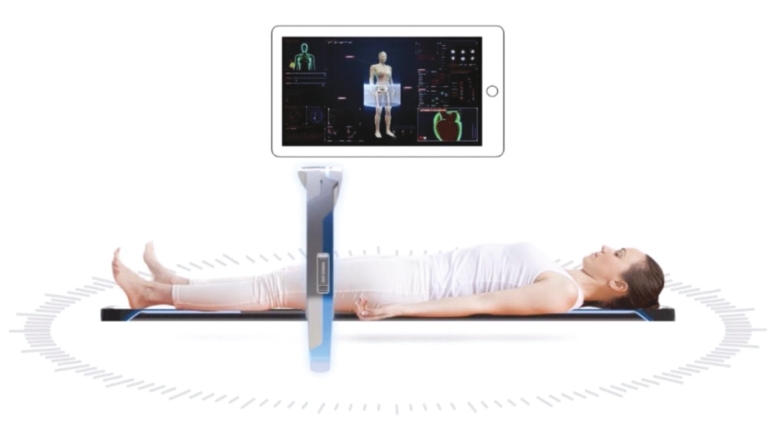
• | Digital X-ray source with the potential to significantly reduce the costs of medical imaging systems. We believe our digital X-ray source technology will allow us to manufacture the Nanox.ARC, if cleared, at substantially lower costs compared to medical imaging systems that use a legacy analog X-ray source without sacrificing imaging quality. A lower cost device has the potential to substantially increase medical imaging availability and improve accessibility of early-detection services broadly across the globe. |
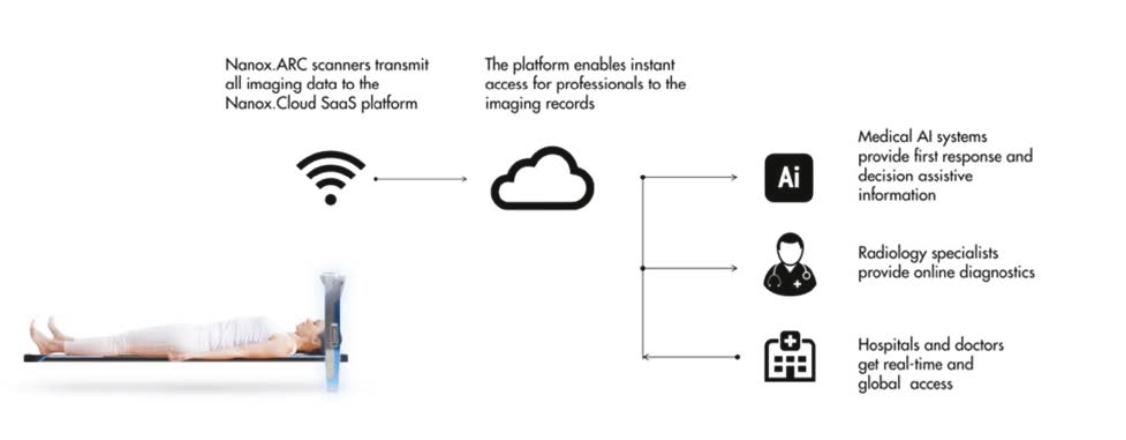
• | Technology designed to improve upon the industry standard with integrated radiology diagnostics via a cloud-based MSaaS platform. The Nanox.ARC employs our novel digital X-ray source that is designed to be energy-efficient, smaller and can be more precisely controlled compared to existing X-ray source. By integrating the Nanox.CLOUD, we believe the Nanox System could provide a streamlined process where each scanned image is uploaded automatically to the cloud system and matched to a human radiology expert and decision assistive AI algorithms to provide scan reviews and diagnostics in a significantly shorter time frame than current diagnostics, which could substantially reduce wait-times for imaging results and increase early detection rates compared to currently employed imaging process protocols. |
• | Business model designed to increase the availability of medical imaging. Our primary business model is based on a pay-per-scan pricing structure as opposed to the capital expenditure-based business model currently used by medical imaging manufacturing companies. We believe our business model will significantly reduce the price per scan compared to the current global average cost of $300 per scan, and has the potential to commoditize medical imaging services at prices that are affordable to a greater number of people. We believe our MSaaS business model has the potential to expand the total size of the X-ray-based medical imaging market. |
• | Secure regulatory clearance for our medical imaging system. We expect to take a multi-step approach to the regulatory clearance process. As a first step, we submitted a 510(k) application for a single-source version of the Nanox.ARC to an accredited Review Organization under the Third Party Review Program in January 2020. In response to the feedback we received from the reviewer, we are conducting standard functional and safety tests to support the 510(k) application and expect to submit the results from these tests in the third quarter of 2020. The timeline was delayed due to the impact of COVID-19 on the external labs we work with to complete these tests. We will continue to optimize and develop further features of the Nanox.ARC, and plan to submit an additional 510(k) application under the Third Party Review Program with respect to the multiple-source Nanox.ARC during the fourth quarter of 2020, which, if cleared, will be our commercial imaging system. |
• | Jumpstart the MSaaS-based medical imaging market with strategic partnerships. We plan to produce and deploy an initial wave of approximately 15,000 Nanox.ARC units over the next three to four years to jumpstart the MSaaS-based medical imaging market. We have entered into a contract manufacturing agreement with a subsidiary of Foxconn for the commercial production and assembly of the Nanox.ARC and we have entered into commercial agreements with strategic regional partners for the deployment, operation and marketing of the Nanox System broadly across the globe, including in the United States and certain countries in Asia, Europe, Africa and South America. We plan to work with these partners to achieve local integrations into health maintenance organizations, electronic health record systems, payment methods and insurance coverage companies. In addition, we have entered into collaboration agreements with cloud-based enterprises and are actively seeking collaboration opportunities, as we anticipate an industry shift to a digital and cloud-based subscription model will bring more digital healthcare disruptors into the market. |
• | Maximize the commercial potential of our technology with simultaneous business models. We plan to commercialize our novel X-ray source technology by pursuing three simultaneous business models, which we believe will provide us the flexibility and long-term sustainability to monetize our technology. |
• | Subscription Model: In certain countries, if permitted by the laws in the applicable jurisdiction, our primary sales strategy will be based on a pay-per-scan pricing structure, where we expect to sell the Nanox System at low cost or at no cost, with a suggested retail price per scan that is substantially lower than the current global average charge, and receive a portion of the proceeds from each scan as the right-to-use licensing fee and fees for usage of the Nanox.CLOUD, artificial intelligence capability and maintenance support. |
• | Sales Model: In certain countries, to accommodate specific local regulatory requirements, we expect to sell the Nanox.ARC for a one-time charge at a price that is substantially less than current market offerings. |
• | Licensing Model: For certain medical imaging market participants, we plan to tailor our X-ray source technology to their specific imaging systems to replace the legacy X-ray source or to license our X-ray source technology to them to develop new types of imaging systems. We expect to charge a one-time licensing fee upfront and receive recurring royalty payments for each system sold. |
• | Leverage the Nanox System to bring added value to our collaborators. We expect that the Nanox System will enable us to accumulate a significant number of medical images, which have the potential to be used by collaborators, such as medical AI-analytics companies, through machine learning algorithms to increase the probability of early disease detection. |
• | we are a development-stage company with limited operating history. We may never be able to effectuate our business plan or achieve any revenue or profitability. Therefore, at this stage of our business, potential investors have a high probability of losing their entire investment; |
• | our efforts may never demonstrate the feasibility of our X-ray source technology for commercial applications; |
• | we are highly dependent on the successful development, marketing and sale of our X-ray source technology and the related products and services; |
• | our business models depend on the successful commercial application of Nanox.CLOUD, which is subject to numerous risks and uncertainties; |
• | business interruptions resulting from the COVID-19 pandemic or similar public health crises could cause a disruption of the development, deployment or regulatory clearance of the Nanox System and adversely impact our business; |
• | products utilizing our technology may need to be approved or cleared by the FDA and similar regulatory agencies worldwide. We may not receive, or may be delayed in receiving, the necessary approval or clearance for our future products, which would adversely affect business, financial condition, results of operations and products; |
• | we may not be successful in implementing our business models; |
• | we expect to depend on third parties to manufacture the Nanox.ARC and to supply certain component parts; |
• | it is difficult and costly to protect our intellectual property and our proprietary technologies, and we may not be able to ensure their protection; |
• | patent terms may be inadequate to protect our competitive position on our future products for an adequate amount of time; |
• | our product candidates and operations are subject to extensive government regulation and oversight both in the United States and abroad, and our failure to comply with applicable requirements could harm our business; |
• | under applicable employment laws, we may not be able to enforce covenants not to compete and therefore may be unable to prevent our competitors from benefiting from the expertise of some of our former employees; and |
• | conditions in Israel could materially and adversely affect our business. |
• | the option to include in an initial public offering registration statement only two years of audited financial statements and selected financial data and only two years of related disclosure; |
• | reduced executive compensation disclosure; and |
• | an exemption from the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act of 2002 (the “Sarbanes-Oxley Act”) in the assessment of our internal control over financial reporting. |
• | the last day of our fiscal year during which we have total annual revenue of at least $1.07 billion; |
• | the last day of our fiscal year following the fifth anniversary of the closing of this offering; |
• | the date on which we have, during the previous three-year period, issued more than $1.0 billion in non-convertible debt securities; or |
• | the date on which we are deemed to be a “large accelerated filer” under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), which, among other things, would occur if the market value of our ordinary shares that are held by non-affiliates exceeds $700 million as of the last business day of our most recently completed second fiscal quarter. |
• | the sections of the Exchange Act regulating the solicitation of proxies, consents or authorizations in respect of a security registered under the Exchange Act; |
• | the sections of the Exchange Act requiring insiders to file public reports of their share ownership and trading activities and liability for insiders who profit from trades made in a short period of time; |
• | the rules under the Exchange Act requiring the filing with the Securities and Exchange Commission (the “SEC”) of quarterly reports on Form 10-Q containing unaudited financial and other specified information, or current reports on Form 8-K, upon the occurrence of specified significant events; and |
• | Regulation Fair Disclosure (“Regulation FD”), which regulates selective disclosures of material information by issuers. |
• | the majority of our executive officers or directors are U.S. citizens or residents; |
• | more than 50% of our assets are located in the United States; or |
• | our business is administered principally in the United States. |
We intend to use the net proceeds from this offering, together with cash on hand, cash equivalents, and short-term investments, for (i) the manufacturing of the initial wave of Nanox.ARC units planned for global deployment and investment in manufacturing capacities, (ii) shipping, installation and deployment costs of the Nanox.ARC, (iii) the continued research and development of the Nanox.ARC, the development of the Nanox.CLOUD and regulatory clearance in various regions and (iv) sales and marketing expenses, general and administrative expenses and other general corporate purposes. See “Use of Proceeds” for additional information.
• | ordinary shares issuable upon the exercise of options to purchase ordinary shares outstanding under the NANO-X Imaging Ltd. 2019 Equity Incentive Plan (the “2019 Equity Incentive Plan”) as of , 2020, at a weighted average exercise price of $ per share; |
• | additional ordinary shares reserved for future issuance under our 2019 Equity Incentive Plan as of , 2020; |
• | ordinary shares issuable upon the exercise of warrants to purchase ordinary shares as of , 2020, at a weighted average exercise price of $ per share, which warrants shall not expire upon the closing of this offering if not exercised; and |
• | ordinary shares issuable upon the exercise of options to purchase ordinary shares to be granted to A-Labs, which provided certain consulting services for this offering, at the closing of this offering, at an exercise price of $16.00 per share. |
• | the assumed exercise prior to the closing of this offering of certain outstanding warrants that shall otherwise expire upon such closing to purchase ordinary shares for an aggregate purchase price of approximately $ million; |
• | no exercise of the outstanding share options or warrants (other than as described above) after , 2020; |
• | no exercise by the underwriters of their option to purchase up to additional ordinary shares from us; and |
• | the adoption and effectiveness of our amended and restated articles of association, which will occur immediately prior to the closing of this offering. |
| | | Six months ended June 30, | | | Year ended December 31, | |||||||
| | | 2020 | | | 2019 | | | 2019 | | | 2018 | |
| | | ($ in thousands, except per share data) | ||||||||||
Consolidated Statement of Operations Data: | | | | | | | | | ||||
Research and development expenses | | | $4,152 | | | $340 | | | $2,717 | | | $672 |
Marketing expenses | | | 1,745 | | | 242 | | | 1,556 | | | 209 |
General and administrative expenses | | | 7,903 | | | 1,079 | | | 18,298 | | | 1,023 |
Operating loss | | | (13,800) | | | (1,661) | | | (22,571) | | | (1,904) |
Financial (income) expenses, net | | | (14) | | | 14 | | | (8) | | | 5 |
Net loss for the year | | | $(13,786) | | | $(1,675) | | | $(22,563) | | | $(1,909) |
Basic and diluted loss per ordinary share(1) | | | $(0.47) | | | $(0.07) | | | $(0.90) | | | $(0.09) |
Weighted average number of ordinary shares outstanding – basic and diluted(1) | | | 29,273 | | | 23,452 | | | 25,181 | | | 20,793 |
Pro forma basic and diluted loss per ordinary share(2) | | | $ | | | $ | | | $ | | | $ |
Pro forma weighted average number of ordinary shares outstanding – basic and diluted(2) | | | | | | | | | ||||
(1) | See Note 7 to our unaudited condensed consolidated financial statements and Note 11 to our audited consolidated financial statements appearing at the end of this prospectus for further details on the calculation of basic and diluted net loss per share. |
(2) | Pro forma loss per ordinary share is calculated by dividing loss for the year by the pro forma weighted average number of ordinary shares outstanding during the period, which gives effect to the Transactions (as defined and further described under “Capitalization”). |
| | | Actual | | | Pro forma(2) | | | Pro forma, as adjusted(3) | |
| | | As of June 30, 2020 | |||||||
| | | ($ in thousands) | |||||||
Consolidated Balance Sheet Data: | | | | | | | |||
Cash and cash equivalents | | | $39,524 | | | $ | | | $ |
Working capital(1) | | | 37,846 | | | | | ||
Total assets | | | 43,581 | | | | | ||
Total liabilities | | | 3,222 | | | | | ||
Accumulated deficit | | | (54,387) | | | | | ||
Total shareholders’ equity | | | 40,359 | | | | | ||
(1) | We define working capital as current assets less current liabilities. |
(2) | The summary pro forma balance sheet data gives effect to the Transactions (as defined and further described under “Capitalization”). |
(3) | The summary pro forma as adjusted balance sheet data gives effect to (i) the Transactions (as defined and further described under “Capitalization”), and (ii) the issuance of ordinary shares in this offering, at an assumed initial public offering price of $ per share, which is the midpoint of the price range set forth on the cover page of this prospectus, after deducting the estimated underwriting discounts and commissions and estimated offering expenses, including the fees payable to A-Labs, payable by us. |
• | the Nanox.CLOUD requires a considerable investment of technical, financial, and legal resources, which may not be available to us; |
• | it may require separate regulatory clearances or approvals; |
• | it may not be technically viable to integrate the Nanox.CLOUD with the businesses of our potential customers and collaborators, such as local operators, radiologists, cloud storage providers, medical AI software providers and others; |
• | market acceptance of the MSaaS model is affected by a variety of factors, including security, reliability, scalability, customization, performance, customer preference, patients’ concerns with entrusting a third party to store and manage their health data, public concerns regarding privacy and compliance with restrictive laws or regulations; |
• | our cloud-based service may raise concerns among our customer base, including concerns regarding changes to pricing over time, service availability, information security of a cloud-based solution and access to medical images while offline; |
• | the Nanox.CLOUD may be subject to computer system failures, cyber-attacks or other security breaches; |
• | incorrect or improper implementation or use of the Nanox.CLOUD by third-party cloud-service providers under our Sales Model could result in customer dissatisfaction and harm our business and reputation; |
• | undetected software errors or flaws in the Nanox.CLOUD could harm our reputation or decrease market acceptance of the MSaaS model; and |
• | we may incur higher costs than we expected as we expand our cloud-based services. |
• | our ability to achieve sufficient market acceptance by hospitals and clinics, providers of medical imaging services, medical professionals such as radiologists, third-party payors and others in the medical community; |
• | our ability to compete with existing medical imaging technology companies; |
• | our ability to establish, maintain and expand our sales, marketing and distribution networks; |
• | our ability to obtain and/or maintain necessary regulatory approvals; and |
• | our ability to effectively protect our intellectual property. |
• | the process of manufacturing and deploying the Nanox System is a complex, multi-step process that depends on factors outside our control, and could cause us to expend significant time and resources prior to earning associated revenues; |
• | the manufacturing cost of the Nanox.ARC may be higher than we expect, may increase significantly, or may increase at a higher rate than anticipated, and we may not be able to set or timely adjust our pay-per-scan pricing to compensate for any increased costs; |
• | the manufacturing of the Nanox.ARC may take longer than we expected, and we may have insufficient manufacturing capacity and experience delays in the manufacturing and deployment of the Nanox System, which would have a negative impact on the timing of our revenues; |
• | deployment and full utilization of the Nanox System may not be achieved or may take substantially longer than we expect, and we may not be able to deploy a sufficient number of units of the Nanox System to support our business or to effectively stimulate market interest; |
• | a Nanox System may perform fewer scans per day than our estimates due to a number of factors, including low market acceptance rate, technical failures and downtime, service disruptions, outages or other performance problems, which would have a negative impact on our revenues and our ability to recover costs; |
• | the implementation, integration and testing of the Nanox.CLOUD with our potential customers and collaborators can be complex, time-consuming and expensive for them, which may have a negative impact on the timing of our revenues; |
• | as part of the Subscription Model, we will be responsible for maintenance of the Nanox System units we deploy, which may be more costly and time-consuming than we expect; |
• | our customers may not be able to find or retain a sufficient number of radiologists to review the images generated by the Nanox System, especially as we deploy additional Nanox Systems and the volume of scans increases; |
• | the portion of our pay-per-scan pricing allocated to our collaborators may not be acceptable to them, either now or in the future, and pricing negotiations with such collaborators may be a complex and time-consuming process; |
• | our pay-per-scan pricing may not be sufficient to recover our costs and may not be adjusted in a timely manner, which could negatively affect our revenues or cause our revenues and results of operations to vary significantly from period to period; |
• | we may be unsuccessful in maintaining our target price per scan because we do not control the price charged by local operators and higher prices may adversely affect market acceptance of the Nanox System; and |
• | regulatory authorities may challenge our Subscription Model altogether, and impose significant civil, criminal, and administrative penalties, damages, fines, and/or exclusion from government funded healthcare programs, which could adversely affect our revenues and results of operations. |
• | generate widespread awareness, acceptance and adoption of our technology and future products or services; |
• | develop new or enhanced technologies or features that improve the convenience, efficiency, safety or perceived safety, and productivity of our technology and future products or services; |
• | properly identify customer needs and deliver new products or services or product enhancements to address those needs; |
• | limit the time required from prototype development to commercial production; |
• | limit the timing and cost of regulatory approvals; |
• | attract and retain qualified personnel and collaborators; |
• | protect our inventions with patents or otherwise develop proprietary products and processes; and |
• | secure sufficient capital resources to expand both our continued research and development, and sales and marketing efforts. |
• | insufficient capacity or delays in meeting our demand; |
• | inadequate manufacturing yields, inferior quality and excessive costs; |
• | inability to manufacture products that meet the agreed upon specifications; |
• | inability to obtain an adequate supply of materials; |
• | inability to comply with the relevant regulatory requirements for the manufacturing process; |
• | limited warranties on products supplied to us; |
• | inability to comply with our contractual obligations; |
• | potential increases in prices; and |
• | increased exposure to potential misappropriation of our intellectual property. |
• | effectiveness of the sales and marketing efforts of us, and our partners such as the local partners; |
• | perception by medical professionals and patients of the convenience, safety, efficiency and benefits of the Nanox.ARC, the Nanox.CLOUD or products using our technology, compared to competing methods of medical imaging; |
• | opposition from certain industry leaders, which may limit our ability to promote the Nanox.ARC or the Nanox.CLOUD and to penetrate into the medical imaging market in certain geographical areas; |
• | the existence of established medical imaging technology; |
• | willingness of market participants to accept the MSaaS model; |
• | the changing and volatile U.S. and global economic environments, including as a result of the COVID-19 pandemic; |
• | timing of market introduction of competing products, and the sales and marketing initiatives of such products; |
• | press and blog coverage, social media coverage, and other publicity and public relations factors by others; |
• | lack of financing or other resources to successfully develop and commercialize our technology and implement our business plan; |
• | the level of commitment and support that we receive from our partners, such as local operators, cloud storage providers and medical AI software providers, as well as medical professionals such as radiologists; and |
• | coverage determinations and reimbursement levels of third party payors. |
• | reimbursement and insurance coverage; |
• | our inability to find agencies, dealers or distributors in specific countries or regions; |
• | our inability to directly control commercial activities of third parties; |
• | limited resources to be deployed to a specific jurisdiction; |
• | the burden of complying with complex and changing regulatory, tax, accounting and legal requirements; |
• | different medical imaging practice and customs in foreign countries affecting acceptance in the marketplace; |
• | import or export licensing and other requirements; |
• | longer accounts receivable collection times; |
• | longer lead times for shipping; |
• | language barriers for technical training; |
• | reduced protection of intellectual property rights in some foreign countries; |
• | foreign currency exchange rate fluctuations; and |
• | interpretations of contractual provisions governed by foreign laws in the event of a contract dispute. |
• | limiting payments for imaging services in physician offices and free-standing imaging facility settings based upon rates paid to hospital outpatient departments; |
• | reducing payments for certain imaging procedures when performed together with other imaging procedures in the same family of procedures on the same patient on the same day in the physician office and free-standing imaging facility setting; |
• | making significant revisions to the methodology for determining the practice expense component of the Medicare payment applicable to the physician office and free-standing imaging facility setting which results in a reduction in payment; and |
• | revising payment policies and reducing payment amounts for imaging procedures performed in the hospital outpatient setting. |
• | disputes among payors as to which party is responsible for payment; |
• | disparity in coverage among various payors; |
• | disparity in information and billing requirements among payors; and |
• | incorrect or missing billing information, which is required to be provided by the ordering physician. |
• | we may not be able to control the amount and timing of resources that our collaborators may devote to our technology; |
• | should a collaborator fail to comply with applicable laws, rules or regulations when performing services for us, we could be held liable for such violations; |
• | our collaborators may have a shortage of qualified personnel, particularly radiologists who can review the medical images generated by the Nanox System, especially as we deploy additional Nanox Systems and the volume of scans increases; |
• | we may be required to relinquish important rights, such as marketing and distribution rights; |
• | business combinations or significant changes in a collaborator’s business strategy may adversely affect a collaborator’s willingness or ability to complete its obligations under any arrangement; |
• | under certain circumstances, a collaborator could move forward with a competing product developed either independently or in collaboration with others, including our competitors; |
• | our current or future collaborators may utilize our proprietary information in a way that could expose us to competitive harm; |
• | our collaborators could obtain ownership or other control over intellectual property that is material to our business; and |
• | collaborative arrangements are often terminated or allowed to expire, which could delay the ability to commercialize our technology. |
• | decreased demand for the Nanox System; |
• | injury to our reputation; |
• | costs of related litigation; |
• | substantial monetary awards to patients and others; |
• | loss of revenue; and |
• | the inability to commercialize future products. |
• | our inability to demonstrate to the satisfaction of the FDA or the applicable regulatory entity or notified body that our product candidates are safe or effective for their intended uses; |
• | the disagreement of the FDA or the applicable foreign regulatory body with the design or implementation of our clinical trials or the interpretation of data from pre-clinical studies or clinical trials; |
• | serious and unexpected adverse effects experienced by participants in our clinical trials; |
• | the data from our pre-clinical studies and clinical trials may be insufficient to support clearance or approval, where required; |
• | our inability to demonstrate that the clinical and other benefits of the device outweigh the risks; |
• | the manufacturing process or facilities we use may not meet applicable requirements; and |
• | the potential for approval policies or regulations of the FDA or applicable foreign regulatory bodies to change significantly in a manner rendering our clinical data or regulatory filings insufficient for clearance or approval. |
• | untitled letters or warning letters; |
• | fines, injunctions, consent decrees and civil penalties; |
• | recalls, termination of distribution, administrative detention, or seizure of our products; |
• | customer notifications or repair, replacement or refunds; |
• | operating restrictions or partial suspension or total shutdown of production; |
• | delays in or refusal to grant our requests for future clearances or approvals or foreign marketing authorizations of new products, new intended uses, or modifications to existing products; |
• | withdrawals or suspensions of product clearances or approvals, resulting in prohibitions on sales of our products; |
• | FDA refusal to issue certificates to foreign governments needed to export products for sale in other countries; and |
• | criminal prosecution. |
• | The U.S. federal healthcare program Anti-Kickback Statute prohibits, among other things, persons from knowingly and willfully soliciting, offering, receiving or providing remuneration, directly or indirectly, in cash or in kind, to induce or reward either the referral of an individual for, or the purchase, order or recommendation of, any good or service, for which payment may be made, in whole or in part, under federal healthcare programs such as Medicare and Medicaid. Although there are a number of statutory exemptions and regulatory safe harbors protecting certain common activities from prosecution, the exemptions and safe harbors are drawn narrowly and practices that involve remuneration to those who prescribe, purchase, or recommend medical devices, including certain discounts, or engaging consultants as speakers or consultants, may be subject to scrutiny if they do not fit squarely within the exemption or safe harbor. Our practices may not in all cases meet all of the criteria for safe harbor protection from anti-kickback liability. Moreover, there are no safe harbors for many common practices, such as educational and research grants. Liability may be established without a person or entity having actual knowledge of the federal Anti-Kickback Statute or specific intent to violate it. In addition, the government may assert that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the civil False Claims Act. Due to the breadth of these laws, the narrowness of statutory exceptions and regulatory safe harbors available, and the range of interpretations to which they are subject, it is possible that some of our current or future practices might be challenged under one or more of these laws, including, without limitation, our proposed Subscription Model, and our advisory, consulting and royalty agreements with certain physicians who receive compensation, in part, in the form of stock or stock options. |
• | The federal civil False Claims Act prohibits, among other things, any person from knowingly presenting, or causing to be presented, a false or fraudulent claim for payment of government funds, or knowingly making, using, or causing to be made or used, a false record or statement material to an |
• | The Health Insurance Portability and Accountability Act of 1996 (“HIPAA”), as amended by the Health Information Technology for Economic and Clinical Health Act of 2009 (“HITECH”), imposes criminal and civil liability for knowingly and willfully executing a scheme to defraud any healthcare benefit program, or knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false statement in connection with the delivery of or payment for healthcare benefits, items or services. In addition, HIPAA, as amended by HITECH, and their respective implementing regulations impose obligations, including mandatory contractual terms, on covered healthcare providers, health plans, as well as their business associates, with respect to safeguarding the privacy, security and transmission of individually identifiable health information. |
• | The Physician Payment Sunshine Act, implemented as the Open Payments program, requires manufacturers of certain products reimbursed by Medicare, Medicaid, or the Children’s Health Insurance Program to track and report to the federal government payments and transfers of value that they make to physicians and teaching hospitals, certain other healthcare professionals beginning in 2022, group purchasing organizations, and ownership interests held by physicians and their families, and provides for public disclosures of these data. Manufacturers are required to submit annual reports to the government and failure to do so may result in civil monetary penalties for all payments, transfers of value and ownership or investment interests not reported in an annual submission, and may result in liability under other federal laws and regulations. |
• | Many states have adopted laws and regulations analogous to the federal laws cited above, including state anti-kickback and false claims laws, which may apply to items or services reimbursed under Medicaid and other state programs or, in several states, regardless of the payer. Several states have enacted legislation requiring medical device companies to, among other things, establish marketing compliance programs; file periodic reports with the state, including reports on gifts and payments to individual health care providers; make periodic public disclosures on sales, marketing, pricing, clinical trials and other activities; and/or register their sales representatives. Some states prohibit specified sales and marketing practices, including the provision of gifts, meals, or other items to certain health care providers. |
• | strengthen the rules on placing devices on the market and reinforce surveillance once they are available; |
• | establish explicit provisions on manufacturers’ responsibilities for follow-up regarding the quality, performance and safety of devices placed on the market; |
• | improve the traceability of medical devices throughout the supply chain to the end-user or patient through a unique identification number; |
• | set up a central database to provide patients, healthcare professionals and the public with comprehensive information on products available in the EU; and |
• | strengthened rules for the assessment of certain high-risk devices, which may have to undergo an additional check by experts before they are placed on the market. |
• | Imposes an annual excise tax of 2.3% on any entity that manufactures or imports medical devices offered for sale in the United States, which, through a series of legislative amendments, was suspended, effective January 1, 2016 and subsequently repealed altogether on December 20, 2019; |
• | Establishes a new Patient-Centered Outcomes Research Institute to oversee and identify priorities in comparative clinical effectiveness research in an effort to coordinate and develop such research; and |
• | Implements Medicare payment system reforms including a national pilot program on payment bundling to encourage hospitals, physicians and other providers to improve the coordination, quality and efficiency of certain healthcare services through bundled payment models. |
• | actual or anticipated fluctuations in results of operations; |
• | actual or anticipated changes in our growth rate relative to our competitors, as well as announcements by us or our competitors of significant business developments, changes in relationships with our target customers, manufacturers or suppliers, acquisitions or expansion plans; |
• | failure to meet or exceed financial estimates and projections of the investment community or that we provide to the public, as well as variance in our financial performance from the expectations of market analysts; |
• | issuance of new or updated research or reports by securities analysts; |
• | share price and volume fluctuations attributable to inconsistent trading volume levels of our shares; |
• | additions or departures of key management or other personnel; |
• | our involvement in litigation; |
• | disputes or other developments related to proprietary rights, including patents, litigation matters, and our ability to obtain patent protection for our technology; |
• | announcement or expectation of additional debt or equity financing efforts; |
• | sales of our ordinary shares or other securities by us, our insiders or our other shareholders, or the perception that these sales may occur in the future; |
• | the trading volume of our ordinary shares; |
• | market conditions in our industry; |
• | changes in the estimation of the future size and growth rate of our markets; and |
• | general economic, market or political conditions in the United States or elsewhere. |
• | our ability to develop and commercialize our technology and future products or services; |
• | developments or disputes concerning our product’s intellectual property rights; |
• | our or our competitors’ technological innovations; |
• | fluctuations in the valuation of companies perceived by investors to be comparable to us; |
• | announcements by us or our competitors of significant contracts, acquisitions, strategic partnerships, joint ventures, capital commitments, new technologies or patents; |
• | failure to complete significant transactions or collaborate with vendors in manufacturing our product; and |
• | proposals for legislation that would place restrictions on the price of medical therapies. |
• | we have hired a corporate controller with U.S. GAAP and SEC reporting experience and are continuing to seek additional financial professionals to increase the number of qualified financial reporting personnel; |
• | we are selecting and implementing a new enterprise resource planning system; |
• | we are developing, communicating and implementing an accounting policy manual for our financial reporting personnel for recurring transactions and period-end closing processes; and |
• | we are establishing monitoring and oversight controls for non-recurring and complex transactions to ensure the accuracy and completeness of our consolidated financial statements and related disclosures. |
• | The initiation, timing, progress and results of our research and development, manufacturing and commercialization activities with respect to our X-ray source technology, the Nanox.ARC, the Nanox.CLOUD and the Nanox System; |
• | our ability to successfully demonstrate the feasibility of our technology for commercial applications; |
• | our expectations regarding the necessity of, timing of filing for, and receipt of, regulatory clearances or approvals regarding our technology, the Nanox.ARC and the Nanox.CLOUD; |
• | our ability to secure and maintain required FDA clearance and similar approvals from regulatory agencies worldwide and comply with applicable quality standards and regulatory requirements; |
• | our ability to manufacture the Nanox.ARC, if cleared, at substantially lower costs compared to medical imaging systems that use a legacy analog X-ray source; |
• | our ability to manufacture, market and deploy approximately 15,000 Nanox.ARC units within the contemplated timeframe; |
• | our ability to meet our planned deployment schedule for the Nanox System units within the contemplated timeframe; |
• | the pricing structure of our products and services, if such products and services receive regulatory clearance or approval; |
• | the implementation of our business models; |
• | our expectations regarding collaborations with third-parties and their potential benefits; |
• | our ability to enter into and maintain our arrangements with third-party manufacturers and suppliers; |
• | our ability to conduct business globally; |
• | our expectations regarding when certain patents may be issued and the protection and enforcement of our intellectual property rights; |
• | our ability to operate our business without infringing the intellectual property rights and propriety technology of third parties; |
• | regulatory developments in the United States and other jurisdictions; |
• | estimates of our expenses, future revenues, capital requirements and our needs for additional financing; |
• | the rate and degree of market acceptance of our technology and our products; |
• | development relating to our competitors and the medical imaging industry; |
• | our estimates of the adoption of the MSaaS-based model by market participants; |
• | our estimates regarding the market opportunities for our technology and our products; |
• | our ability to attract, motivate and retain key executive managers; |
• | our ability to comply with data protection laws, regulations and similar rules and to establish and maintain adequate cyber-security and data protection; |
• | our ability to obtain third-party payor coverage or reimbursement of our Nanox System; |
• | our expectation regarding the maintenance of our foreign private issuer and emerging growth company status; |
• | our expectations regarding the use of proceeds from this offering; |
• | the effect of the COVID-19 pandemic, including mitigation efforts and economic effects, on any of the foregoing or other aspects of our business operations, including but not limited to the development, deployment and regulatory clearance of the Nanox Systems; and |
• | our success at managing other risks and uncertainties, including those listed under “Risk Factors.” |
• | between $ million to $ million will be used for the manufacturing of the initial wave of Nanox.ARC units planned for global deployment and investment in manufacturing capacities; to the extent the cost-per-unit of the Nanox.ARC is higher than we expected or the amount of proceeds we receive is lower than we expected, we plan to reduce the number of units to be manufactured with such proceeds accordingly; |
• | between $ million to $ million will be used for the shipping, installation and deployment costs of the Nanox System; to the extent the number of units of the Nanox.ARC to be manufactured is reduced for the reasons described above, the amount of proceeds to be used for shipping, installation and deployment will be reduced accordingly; |
• | between $ million to $ million will be used for the continued research and development of the Nanox.ARC, the development of the Nanox.CLOUD and for regulatory clearance in various regions, which we expect will be sufficient for obtaining the 510(k) medical device clearance with respect to the Nanox.ARC with the FDA; and |
• | the remaining funds, if any, to be used for sales and marketing expenses, general and administrative expenses and general corporate purposes. |
• | the existence of unforeseen or other opportunities or the need to take advantage of changes in timing of our existing activities; |
• | the need or desire on our part to accelerate, increase, reduce or eliminate one or more existing initiatives due to, among other things, changing market conditions or competitive developments or interim results of research and development efforts; |
• | results from our business development and marketing efforts; |
• | the effect of federal, state, and local regulation on our business; and |
• | the presentation of strategic opportunities of which we are not currently aware (including acquisitions, joint ventures, licensing and other similar transactions). |
• | an actual basis; |
• | a pro forma basis to give effect to (i) the receipt of $66,000,000 and the issuance of 4,125,000 ordinary shares to certain investors after June 30, 2020 in connection with the Private Placement, and (ii) the receipt of $ pursuant to the exercise of warrants held by certain of our shareholders and the related issuance of ordinary shares as a result thereof immediately prior to the closing of this offering (collectively, the “Transactions”); and |
• | on a pro forma as adjusted basis to give further effect to the issuance and sale of ordinary shares by us in this offering at an assumed initial public offering price of $ per share, the midpoint of the price range set forth on the cover page of this prospectus, after deducting the estimated underwriting discounts and commissions and estimated offering expenses, including the fees payable to A-Labs, payable by us. |
| | | As of June 30, 2020 | |||||||
| | | Actual | | | Pro Forma | | | Pro Forma As Adjusted(1) | |
| | | ($ in thousands, except share and per share amounts) | |||||||
Cash and cash equivalents | | | $39,524 | | | $ | | | $ |
Shareholders’ equity: | | | | | | | |||
Ordinary Shares, par value NIS 0.01 per share; 40,000,000 shares authorized, actual; 100,000,000 shares authorized, pro forma and pro forma as adjusted; 30,679,965 shares issued and outstanding, actual; shares issued and outstanding, pro forma; shares issued and outstanding, pro forma as adjusted | | | 85 | | | — | | | — |
Additional paid-in capital | | | 94,661 | | | | | ||
Accumulated deficit | | | (54,387) | | | | | ||
Total shareholders’ equity | | | 40,359 | | | | | ||
Total capitalization | | | $40,359 | | | $ | | | $ |
(1) | Each $1.00 increase or decrease in the assumed initial public offering price of $ per share (which is the midpoint of the price range set forth on the cover page of this prospectus) would increase or decrease the amount of each of cash and cash equivalents, additional paid-in capital, total shareholders’ equity and total capitalization on a pro forma as adjusted basis by approximately $ million, assuming that the number of shares offered by us, as set forth on the cover page of this prospectus, remains the same and after deducting the estimated underwriting discounts and commissions and estimated offering expenses, including the fees payable to A-Labs, payable by us. Each increase or decrease of 1.0 million in the number of ordinary shares we are offering would increase or decrease the pro forma as adjusted amount of each of cash and cash equivalents, additional paid-in capital, total shareholders’ equity and total capitalization by approximately $ million, assuming no change in the assumed initial public offering price and after deducting the estimated underwriting discounts and commissions and estimated offering expenses, including the fees payable to A-Labs, payable by us. |
• | 4,586,424 ordinary shares issuable upon the exercise of options to purchase ordinary shares outstanding under the 2019 Equity Incentive Plan as of June 30, 2020, at a weighted average exercise price of $3.70 per share; |
• | 3,455,512 additional ordinary shares reserved for future issuance under our 2019 Equity Incentive Plan as of June 30, 2020; |
• | 5,496,984 ordinary shares issuable upon the exercise of warrants to purchase ordinary shares as of June 30, 2020, at a weighted average exercise price of $10.14 per share, which warrants shall not expire upon the closing of this offering if not exercised; and |
• | ordinary shares issuable upon the exercise of options to purchase ordinary shares to be granted to A-Labs, which provided certain consulting services for this offering, at the closing of this offering, at an exercise price of $16.00 per ordinary share. |
• | subtracting our liabilities from our tangible assets as of June 30, 2020; and |
• | dividing the difference by the number of ordinary shares outstanding as of June 30, 2020. |
Assumed initial public offering price per share | | | | | $ | |
Historical net tangible book value per share as of June 30, 2020 | | | $1.31 | | | |
Increase per share attributable to the Transactions | | | | | ||
Pro forma net tangible book value (deficit) per share as of June 30, 2020 | | | | | ||
Increase per share attributable to this offering | | | | | ||
Pro forma as adjusted net tangible book value per share after this offering | | | | | $ | |
Dilution per share to new investors in this offering | | | | | $ |
| | | Ordinary Shares Purchased | | | Total Consideration | | | Average Price Per Share | |||||||
| | | Number | | | % | | | Amount | | | % | | |||
Existing shareholders | | | | | | | $ | | | | | $ | |||
New investors | | | | | | | | | | | |||||
Total | | | | | 100% | | | | | 100% | | | |||
• | the percentage of ordinary shares held by existing shareholders will decrease to approximately % of the total number of our ordinary shares outstanding after this offering; and |
• | the number of shares held by new investors will increase to , or approximately % of the total number of our ordinary shares outstanding after this offering. |
• | 4,586,424 ordinary shares issuable upon the exercise of options to purchase ordinary shares outstanding under the 2019 Equity Incentive Plan as of June 30, 2020, at a weighted average exercise price of $3.70 per share; |
• | 3,455,512 additional ordinary shares reserved for future issuance under our 2019 Equity Incentive Plan as of June 30, 2020; |
• | 5,496,984 ordinary shares issuable upon the exercise of warrants to purchase ordinary shares as of June 30, 2020, at a weighted average exercise price of $10.14 per share, which warrants shall not expire upon the closing of this offering if not exercised; and |
• | ordinary shares issuable upon the exercise of options to purchase ordinary shares to be granted to A-Labs, which provided certain consulting services for this offering, at the closing of this offering, at an exercise price of $16.00 per ordinary share. |
| | | Six months ended June 30, | | | Year ended December 31, | |||||||
| | | 2020 | | | 2019 | | | 2019 | | | 2018 | |
| | | ($ in thousands, except per share data) | ||||||||||
Consolidated Statement of Operations Data: | | | | | | | | | ||||
Research and development expenses | | | $4,152 | | | $340 | | | $2,717 | | | $672 |
Marketing expenses | | | 1,745 | | | 242 | | | 1,556 | | | 209 |
General and administrative expenses | | | 7,903 | | | 1,079 | | | 18,298 | | | 1,023 |
Operating loss | | | (13,800) | | | (1,661) | | | (22,571) | | | (1,904) |
Financial (income) expenses, net | | | (14) | | | 14 | | | (8) | | | 5 |
Net loss for the year | | | $(13,786) | | | $(1,675) | | | $(22,563) | | | $(1,909) |
Basic and diluted loss per ordinary share(1) | | | (0.47) | | | (0.07) | | | $(0.90) | | | $(0.09) |
Weighted average number of ordinary shares outstanding – basic and diluted(1) | | | 29,273 | | | 23,452 | | | 25,181 | | | 20,793 |
(1) | Basic loss per share and diluted loss per share are the same because outstanding options would be anti-dilutive due to our net losses in these periods. See Note 7 to our unaudited condensed consolidated financial statements and Note 11 to our audited consolidated financial statements appearing at the end of this prospectus for further details on the calculation of basic and diluted net loss per share attributable to our ordinary shareholders. |
| | | As of June 30, | | | As of December 31, | ||||
| | | 2020 | | | 2019 | | | 2018 | |
| | | ($ in thousands) | |||||||
Consolidated Balance Sheet Data: | | | | | | | |||
Cash and cash equivalents | | | $39,524 | | | $8,072 | | | $5 |
Working capital(1) | | | 37,846 | | | (10,627) | | | (6,540) |
Total assets | | | 43,581 | | | 11,871 | | | 1,855 |
Total liabilities | | | 3,222 | | | 20,649 | | | 8,239 |
Accumulated deficit | | | (54,387) | | | (40,601) | | | (18,038) |
Total shareholders’ equity (deficit) | | | 40,359 | | | (8,778) | | | (6,384) |
(1) | We define working capital as current assets less current liabilities. |
• | expenses incurred in connection with the development of our products, including payments made pursuant to agreements with third parties, such as outside consultants related to process development and manufacturing activities, as well as patent registrations; |
• | costs of components and materials, including payments made pursuant to agreements with third parties; |
• | costs of laboratory supplies incurred for each program; |
• | facilities, depreciation and other expenses, including direct or allocated expenses for rent and maintenance of facilities, as well as insurance costs; |
• | costs related to compliance with regulatory requirements; and |
• | employee-related expenses, including salaries, related benefits and share-based compensation expenses for employees engaged in research and development activities. |
• | the timing and progress of development activities; |
• | our ability to maintain our current research and development programs and to establish new ones; |
• | the receipt of regulatory approvals from applicable regulatory authorities without the need for independent clinical trials or validation; |
• | the timing, receipt and terms of any marketing approvals from applicable regulatory authorities; |
• | our ability to establish new licensing or collaboration arrangements; |
• | the performance of our future collaborators, if any; |
• | establishing commercial manufacturing capabilities or making arrangements with third-party manufacturers; |
• | obtaining, maintaining, defending and enforcing patent claims and other intellectual property rights; |
• | launching commercial sales of our products, including the Nanox.ARC hardware and Nanox.CLOUD software, whether alone or in collaboration with others; and |
• | maintaining a continued acceptable safety profile of the products following approval. |
| | | Six months ended June 30, | | | |||||
| | | 2020 | | | 2019 | | | Change | |
| | | ($ in thousands) | |||||||
Operating expenses | | | | | | | |||
Research and development | | | $4,152 | | | $340 | | | $3,812 |
Marketing | | | 1,745 | | | 242 | | | 1,503 |
General and administrative | | | 7,903 | | | 1,079 | | | 6,824 |
Operating loss | | | (13,800) | | | (1,661) | | | (12,139) |
Financial (income) expenses, net | | | (14) | | | 14 | | | (28) |
Net loss | | | $(13,786) | | | $(1,675) | | | $(12,111) |
| | | Six months ended June 30, | ||||
| | | 2020 | | | 2019 | |
| | | ($ in thousands) | ||||
Research and Development Expenses: | | | | | ||
R&D - salaries and wages | | | $723 | | | $151 |
Share-based compensation | | | 1,917 | | | 0 |
R&D - professional services | | | 1,497 | | | 157 |
Other | | | 15 | | | 32 |
Total | | | $4,152 | | | $340 |
| | | Six months ended June 30, | ||||
| | | 2020 | | | 2019 | |
| | | ($ in thousands) | ||||
Marketing Expenses: | | | | | ||
Marketing – salaries and wages | | | $246 | | | $8 |
Marketing and business development | | | 855 | | | 234 |
Share-based compensation | | | 644 | | | 0 |
Total | | | $1,745 | | | $242 |
| | | Six months ended June 30, | ||||
| | | 2020 | | | 2019 | |
| | | ($ in thousands) | ||||
General and Administrative Expenses: | | | | | ||
G&A – salaries and wages | | | $783 | | | $26 |
Share-based compensation | | | 5,786 | | | 0 |
Management fee | | | 77 | | | 278 |
G&A – professional services | | | 765 | | | 524 |
Legal fees | | | 103 | | | 149 |
Rent and Maintenance | | | 233 | | | 58 |
Other | | | 156 | | | 44 |
Total | | | $7,903 | | | $1,079 |
| | | Year ended December 31, | | | |||||
| | | 2019 | | | 2018 | | | Change | |
| | | ($ in thousands) | |||||||
Operating expenses | | | | | | | |||
Research and development | | | $2,717 | | | $672 | | | $2,045 |
Marketing | | | 1,556 | | | 209 | | | 1,347 |
General and administrative | | | 18,298 | | | 1,023 | | | 17,275 |
Operating loss | | | (22,571) | | | (1,904) | | | (20,667) |
Financial (income) expenses, net | | | (8) | | | 5 | | | (13) |
Net loss | | | $(22,563) | | | $(1,909) | | | $(20,654) |
| | | Year ended December 31, | ||||
| | | 2019 | | | 2018 | |
| | | ($ in thousands) | ||||
Research and Development Expenses: | | | | | ||
R&D - salaries and wages | | | $437 | | | $131 |
Share-based compensation | | | 661 | | | — |
R&D - professional services | | | 1,450 | | | 519 |
Other | | | 169 | | | 22 |
Total | | | $2,717 | | | $672 |
| | | Year ended December 31, | ||||
| | | 2019 | | | 2018 | |
| | | ($ in thousands) | ||||
Marketing Expenses: | | | | | ||
Marketing – salaries and wages | | | $200 | | | $— |
Marketing and business development | | | $439 | | | $ 59 |
Share-based compensation | | | 617 | | | — |
Other | | | 300 | | | 150 |
Total | | | $1,556 | | | $209 |
| | | Year ended December 31, | ||||
| | | 2019 | | | 2018 | |
| | | ($ in thousands) | ||||
General and Administrative Expenses: | | | | | ||
G&A – salaries and wages | | | $461 | | | $88 |
Share-based compensation | | | 14,967 | | | 115 |
Management fee | | | 534 | | | 429 |
G&A – professional services | | | 1,470 | | | 84 |
Legal fees | | | 417 | | | 165 |
Other | | | 449 | | | 142 |
Total | | | $18,298 | | | $1,023 |
| | | Six months ended June 30, | | | Year ended December 31, | |||||||
| | | 2020 | | | 2019 | | | 2019 | | | 2018 | |
| | | ($ in thousands) | ||||||||||
Net cash used in operating activities | | | $(4,738) | | | $(1,060) | | | $(5,524) | | | $(3,671) |
Net cash used in investing activities | | | (244) | | | (80) | | | (125) | | | (73) |
Net cash provided by financing activities | | | 36,481 | | | 9,264 | | | 13,861 | | | 3,684 |
Net change in cash and cash equivalents and restricted cash | | | $31,499 | | | $8,124 | | | $8,212 | | | $(60) |
• | seek regulatory approvals for any additional products; |
• | seek to discover and develop additional products; |
• | establish a manufacturing, sales, marketing, medical affairs and distribution infrastructure to commercialize the Nanox System for which we may obtain marketing approval and intend to commercialize on our own or jointly; |
• | hire additional quality control and scientific personnel; |
• | expand our operational, financial and management systems and increase personnel, including personnel to support our clinical development, manufacturing and commercialization efforts and our operations as a public company; |
• | maintain, expand and protect our intellectual property portfolio; and |
• | acquire or in-license other products and technologies. |
• | the scope, progress, results and costs of researching and developing the Nanox System; |
• | the costs, timing and outcome of regulatory review of the Nanox.ARC; |
• | the costs of future activities, including product sales, medical affairs, marketing, manufacturing and distribution, for the Nanox System for which we receive marketing approval; |
• | commercial manufacturing of the Nanox System and sufficient inventory to support commercial launch; |
• | the revenue, if any, received from commercial sale of the Nanox System, should the Nanox.ARC receive marketing approval; |
• | the cost and timing of hiring new employees to support our continued growth; |
• | the costs of preparing, filing and prosecuting patent applications, maintaining and enforcing our intellectual property rights and defending intellectual property-related claims; |
• | the ability to establish and maintain collaborations on favorable terms, if at all; and |
• | the timing, receipt and amount of sales of the Nanox System, if any. |
| | | Payment due by period | |||||||||||||
| | | ($ in thousands) | |||||||||||||
Contractual Obligations | | | Total | | | Less than 1 year | | | 1-3 years | | | 3-5 years | | | More than 5 years |
Capital (Finance) Lease Obligations | | | — | | | — | | | — | | | — | | | — |
Operating Lease Obligations | | | $526 | | | $140 | | | $386 | | | — | | | — |
Purchase Obligations | | | — | | | — | | | — | | | — | | | — |
Total | | | $526 | | | $140 | | | $386 | | | — | | | — |
| | | % | |
Computers | | | 10-33 |
Office furniture and lab equipment | | | 10-20 |
Level 1: | Quoted prices (unadjusted) in active markets that are accessible at the measurement date for assets or liabilities. The fair value hierarchy gives the highest priority to Level 1 inputs. |
Level 2: | Observable prices that are based on inputs not quoted on active markets, but corroborated by market data. |
Level 3: | Unobservable inputs are used when little or no market data is available. The fair value hierarchy gives the lowest priority to Level 3 inputs. |
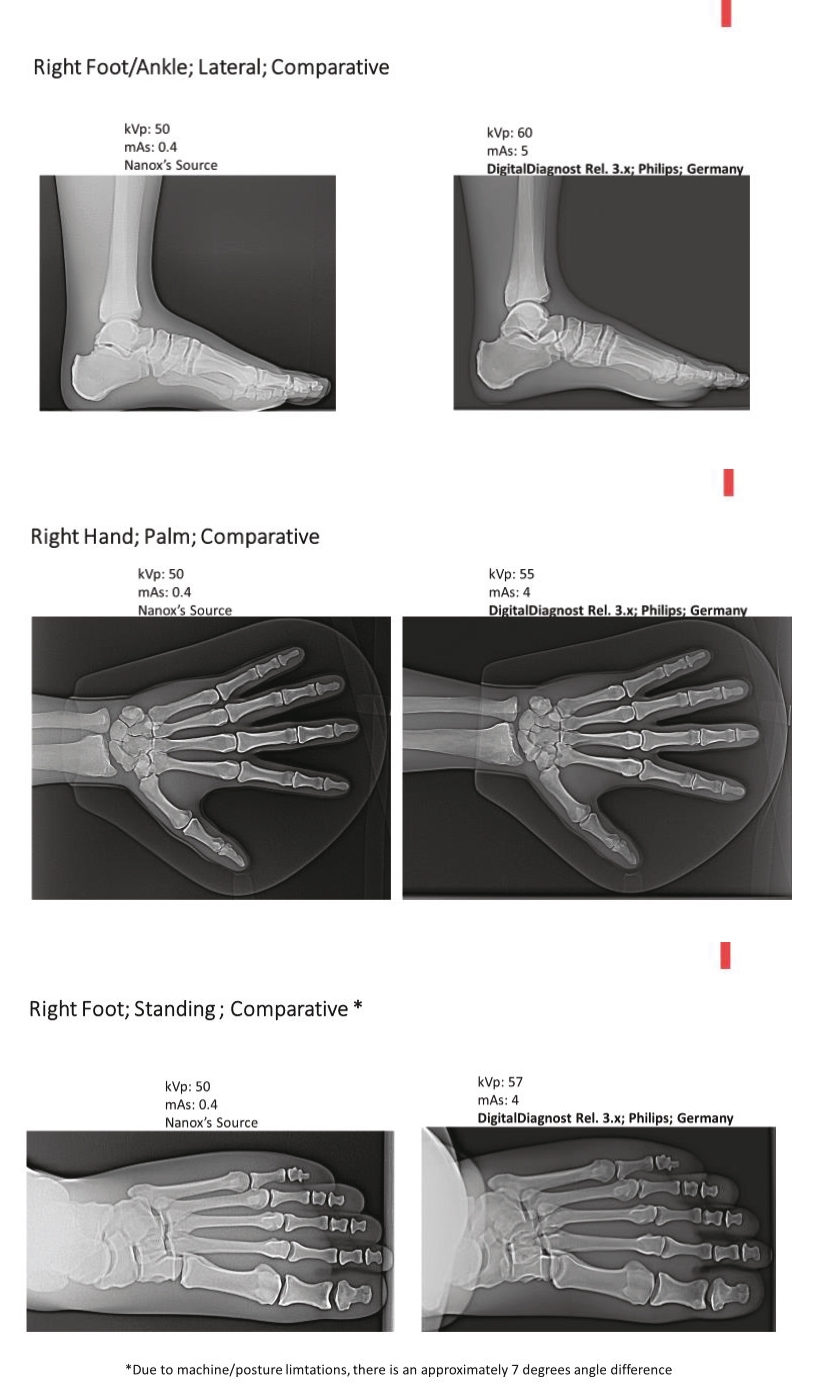
• | Digital X-ray source with the potential to significantly reduce the costs of medical imaging systems. We believe our digital X-ray source technology will allow us to manufacture the Nanox.ARC, if cleared, at substantially lower costs compared to medical imaging systems that use a legacy analog X-ray source without sacrificing imaging quality. A lower cost device has the potential to substantially increase medical imaging availability and improve accessibility of early-detection services broadly across the globe. |
• | Technology designed to improve upon the industry standard with integrated radiology diagnostics via a cloud-based MSaaS platform. The Nanox.ARC employs our novel digital X-ray source that is designed to be energy-efficient, smaller and can be more precisely controlled compared to existing X-ray source. By integrating the Nanox.CLOUD, we believe the Nanox System could provide a streamlined process where each scanned image is uploaded automatically to the cloud system and matched to a human radiology expert and decision assistive AI algorithms to provide scan reviews and diagnostics in a significantly shorter time frame than current diagnostics, which could substantially reduce wait-times for imaging results and increase early detection rates compared to currently employed imaging process protocols. |
• | Business model designed to increase the availability of medical imaging. Our primary business model is based on a pay-per-scan pricing structure as opposed to the capital expenditure-based business model currently used by medical imaging manufacturing companies. We believe our business model will significantly reduce the price per scan compared to the current global average cost of $300 per scan, and has the potential to commoditize medical imaging services at prices that are affordable to a greater number of people. We believe our MSaaS business model has the potential to expand the total size of the X-ray-based medical imaging market. |
• | Secure regulatory clearance for our medical imaging system. We expect to take a multi-step approach to the regulatory clearance process. As a first step, we submitted a 510(k) application for a single-source version of the Nanox.ARC to an accredited Review Organization under the Third Party Review Program in January 2020. In response to the feedback we received from the reviewer, we are conducting standard functional and safety tests to support the 510(k) application and expect to submit the results from these tests in the third quarter of 2020. The timeline was delayed due to the impact of COVID-19 on the external labs we work with to complete these tests. We will continue to optimize and develop further features of the Nanox.ARC, and plan to submit an additional 510(k) application under the Third Party Review Program with respect to the multiple-source Nanox.ARC during the fourth quarter of 2020, which, if cleared, will be our commercial imaging system. |
• | Jumpstart the MSaaS-based medical imaging market with strategic partnerships. We plan to produce and deploy an initial wave of approximately 15,000 Nanox.ARC units over the next three to four years to jumpstart the MSaaS-based medical imaging market. We have entered into a contract |
• | Maximize the commercial potential of our technology with simultaneous business models. We plan to commercialize our novel X-ray source technology by pursuing three simultaneous business models, which we believe will provide us the flexibility and long-term sustainability to monetize our technology. |
• | Subscription Model: In certain countries, if permitted by the laws in the applicable jurisdiction, our primary sales strategy will be based on a pay-per-scan pricing structure, where we expect to sell the Nanox System at low cost or at no cost, with a suggested retail price per scan that is substantially lower than the current global average charge, and receive a portion of the proceeds from each scan as the right-to-use licensing fee and fees for usage of the Nanox.CLOUD, artificial intelligence capability and maintenance support. |
• | Sales Model: In certain countries, to accommodate specific local regulatory requirements, we expect to sell the Nanox.ARC for a one-time charge at a price that is substantially less than current market offerings. |
• | Licensing Model: For certain medical imaging market participants, we plan to tailor our X-ray source technology to their specific imaging systems to replace the legacy X-ray source or to license our X-ray source technology to them to develop new types of imaging systems. We expect to charge a one-time licensing fee upfront and receive recurring royalty payments for each system sold. |
• | Leverage the Nanox System to bring added value to our collaborators. We expect that the Nanox System will enable us to accumulate a significant number of medical images, which have the potential to be used by collaborators, such as medical AI-analytics companies, through machine learning algorithms to increase the probability of early disease detection. |
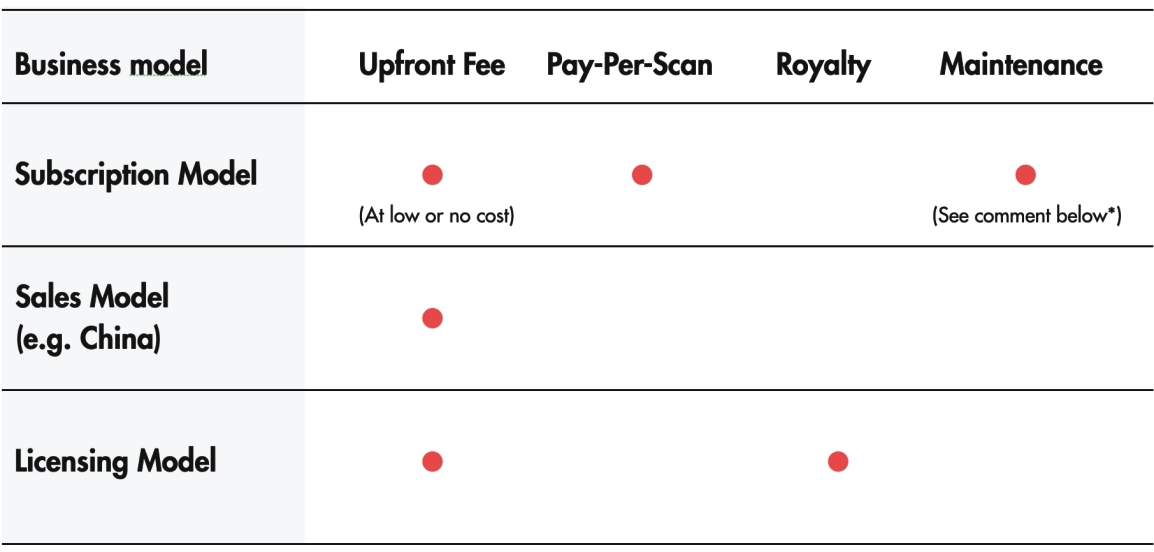
* | We expect to contract with third parties to provide maintenance and support services. |

Entity | | | Date of MSaaS Agreement | | | Region | | | Number of Nanox Systems to be Provided | | | Minimum Annual Fee and Amount of Letter of Credit (approximate) | | | Initial Term | | | Renewal Term |
The Gateway Group, Ltd. | | | February 11, 2020 | | | Australia, New Zealand and Norway | | | 1,000 | | | $58 million | | | 3 years | | | 3 years |
Golden Vine International Company, Ltd. | | | May 28, 2020 | | | Taiwan and Singapore | | | 500 | | | Up to $29 million | | | 5 years | | | 5 years* |
Promedica Bioelectronics s.r.l. | | | May 29, 2020 | | | Italy | | | 500 | | | $29 million | | | 4 years | | | 3 years |
JSC Roel Group | | | May 29, 2020 | | | Russian Federation | | | 500 | | | $12.6 million | | | 5 years | | | 5 years |
Clarity Medical Solution, a division of “Grodnobioproduct” LLC | | | June 4, 2020 | | | Belarus | | | 100 | | | $3.7 million | | | 3 years | | | 4 years |
Gold Rush | | | June 16, 2020 | | | South Africa | | | 500 | | | $15.5 million | | | 3 years | | | 3 years |
LATAM Business Development Group Ltd. | | | July 6, 2020 | | | Brazil | | | 1,000 | | | $4.8 million (9 million Letter of Credit) in Year 1 $14.5 million in Year 2 $24.2 million in Year 3*** | | | 6 years | | | 3 years |
APR 1998 S.L. | | | July 25, 2020 | | | Spain | | | 420 | | | $11.4 million | | | 5 years | | | 5 years** |
TOTAL | | | | | | | 4,520 | | | $163.8 million | | | | |
* | The MSaaS Agreement with Golden Vine International Company, Ltd. may also be terminated by either party upon notice stipulating that the notifying party has come to the conclusion, based on market evidence, that there is no business merit for the Nanox.ARC in Taiwan or Singapore. |
** | The MSaaS Agreement with APR 1998 S.L. may also be terminated by the service provider at the end of a six-month trial period by sending within five days a formal notice to the Company if trial results are not satisfactory. |
*** | The enforceability of the standby letter of credit from LATAM Business Development Group Ltd. in our favor is also conditioned upon the parties finalizing within 90 days of the date of the agreement, in mutually agreed form, the terms and conditions of the statement of work, the system requirement specifications and the service level agreement. |
• | Competing digital X-ray sources with same or better attributes; and |
• | Competing enterprises operating an MSaaS business model. |
• | establishment registration and device listing with the FDA; |
• | QSR requirements, which require manufacturers, including third-party manufacturers, to follow stringent design, testing, control, documentation and other quality assurance procedures during all aspects of the design and manufacturing process; |
• | labeling regulations and FDA prohibitions against the promotion of investigational products, or the promotion of “off-label” uses of cleared or approved products; |
• | requirements related to promotional activities; |
• | clearance or approval of product modifications to 510(k)-cleared devices that could significantly affect safety or effectiveness or that would constitute a major change in intended use of one of our cleared devices, or approval of certain modifications to PMA-approved devices; |
• | medical device reporting regulations, which require that a manufacturer report to the FDA if a device it markets may have caused or contributed to a death or serious injury, or has malfunctioned and the device or a similar device that it markets would be likely to cause or contribute to a death or serious injury, if the malfunction were to recur; |
• | correction, removal and recall reporting regulations, which require that manufacturers report to the FDA field corrections and product recalls or removals if undertaken to reduce a risk to health posed by the device or to remedy a violation of the FDCA that may present a risk to health; |
• | the FDA’s recall authority, whereby the agency can order device manufacturers to recall from the market a product that is in violation of governing laws and regulations; and |
• | post-market surveillance activities and regulations, which apply when deemed by the FDA to be necessary to protect the public health or to provide additional safety and effectiveness data for the device. |
• | warning letters, untitled letters, fines, injunctions, consent decrees and civil penalties; |
• | recalls, withdrawals or administrative detention or seizure of our products; |
• | operating restrictions or partial suspension or total shutdown of production; |
• | refusing or delaying requests for 510(k) marketing clearance or PMA approvals of new products or modified products; |
• | withdrawing 510(k) clearances or PMA approvals that have already been granted; |
• | refusal to grant export approvals for our products; or |
• | criminal prosecution. |
Area of Activity | | | As of June 30, 2020 |
General and Administrative | | | 11 |
Research, Development and Quality Assurance | | | 15 |
Sales and Marketing | | | 1 |
Total | | | 27 |
Name | | | Age | | | Position |
Executive Officers | | | | | ||
Ran Poliakine | | | 52 | | | Founder, Chief Executive Officer and Director* |
Itzhak Maayan | | | 55 | | | Chief Financial Officer |
Tal Shank | | | 42 | | | Vice President Corporate Development |
Yoel Raab | | | 65 | | | Chief Technology Officer |
Anat Kaphan | | | 50 | | | Vice President Product Marketing |
Shirly Kaufman-Kirshenbaum | | | 46 | | | Vice President Human Resources |
| | | | | |||
Non-Employee Directors and Director Nominees | | | | | ||
Onn Fenig | | | 45 | | | Director |
Floyd Katske | | | 69 | | | Director |
Erez Meltzer | | | 62 | | | Director |
Richard Stone | | | 77 | | | Director |
* | This individual will occupy the position of Chairman of the board of directors upon the closing of this offering. |
• | we intend to follow Israeli corporate governance practices instead of the Nasdaq requirements with regard to, among other things, the nomination committee and director nomination procedures. |
• | we intend to comply with Israeli law, which permits a company to determine in its articles of association the number of shareholders and percentage of holdings required for such a quorum. Our amended and restated articles of association provide that a quorum of two or more shareholders holding at least 25% of the voting rights in person or by proxy is required for commencement of business at a general shareholders meeting. While the quorum set forth in our amended and restated articles of association with respect to an adjourned meeting is identical to the quorum for any other meeting (as described in the initial sentence), if, within half an hour from the time appointed for the adjourned meeting, a quorum is not present, a quorum shall thereafter consist of one or more shareholders present in person or by proxy, regardless of the number or percentage of our outstanding shares held by them; |
• | with the exception of our external directors and directors elected by our board of directors due to a vacancy, in accordance with the staggered nomination as described under “—Board of Directors and Officers,” we intend to elect our directors to hold office until the annual general meeting of our shareholders that occurs in the third year following his or her election and until his or her successor shall be elected and qualified. The nominations for directors, which are presented to our shareholders by our board of directors, are generally made by the board of directors itself, in accordance with the provisions of our amended and restated articles of association and the Companies Law; |
• | we intend to adopt and approve material changes to equity incentive plans in accordance with the Companies Law, which does not impose a requirement of shareholder approval for such actions. In addition, we intend to follow Israeli corporate governance practice, which requires shareholder approval prior to an issuance of securities in connection with equity-based compensation of officers, directors, employees or consultants only under certain circumstances, in lieu of Nasdaq Marketplace Rule 5635(c); |
• | as opposed to making periodic reports to shareholders and proxy solicitation materials available to shareholders in the manner specified by the Nasdaq corporate governance rules, the Companies Law does not require us to distribute periodic reports directly to shareholders, and the generally accepted business practice in Israel is not to distribute such reports to shareholders but to make such reports available through a public website. We will only mail such reports to shareholders upon request. As a foreign private issuer, we are generally exempt from the SEC’s proxy solicitation rules; and |
• | we will follow Israeli corporate governance practices instead of Nasdaq requirements to obtain shareholder approval for all corporate actions requiring such approval under the requirements of the Companies Law such as (i) transactions with directors concerning the terms of their service or indemnification, exemption and insurance for their service (or for any other position that they may hold at our company), (ii) extraordinary transactions with controlling shareholders, (iii) terms of employment or other engagement of the controlling shareholder of the company or such controlling shareholder’s relative, (iv) private placements that will result in a change of control, (v) certain transactions, other than a public offering, involving issuances of a 20% or greater interest in us and (vi) certain acquisitions of the stock or assets of another company. |
• | an employment relationship; |
• | a business or professional relationship maintained on a regular basis; |
• | control; and |
• | service as an office holder, excluding service as a director in a private company prior to the first offering of its shares to the public if such director was appointed as a director of the private company in order to serve as an external director following the initial public offering. |
• | the majority of the shares voted at the meeting in favor of the election of the external director, excluding abstentions, include at least a majority of the votes of shareholders who are not controlling shareholders and do not have a personal interest in the appointment (excluding a personal interest that did not result from the shareholder’s relationship with the controlling shareholder); or |
• | the total number of shares held by non-controlling shareholders or any one on their behalf that are voted against the election of the external director does not exceed 2% of the aggregate voting rights in the company. |
• | his or her service for each such additional term is recommended by one or more shareholders holding at least 1% of the company’s voting rights and is approved at a shareholders meeting by a disinterested majority, where the total number of shares held by non-controlling, disinterested shareholders voting for such reelection exceeds 2% of the aggregate voting rights in the company and subject to additional restrictions set forth in the Companies Law with respect to the affiliation of the external director nominee; |
• | the external director proposed his or her own nomination, and such nomination was approved in accordance with the requirements described in the paragraph above; or |
• | his or her service for each such additional term is recommended by the board of directors and is approved at a meeting of shareholders by the same majority required for the initial election of an external director (as described above). |
• | the chairman of the board of directors; |
• | a controlling shareholder or a relative of a controlling shareholder; |
• | any director employed by the company or by one of its controlling shareholders or by an entity controlled by one of its controlling shareholders (other than as a member of the board of directors); |
• | any director who regularly provides services to the company, to one of its controlling shareholders or to an entity controlled by one of its controlling shareholders; or |
• | a director who derives most of his or her income from a controlling shareholder. |
• | recommending the retention and termination of our independent registered public accounting firm to the board of directors in accordance with Israeli law; |
• | recommending to the board of directors in accordance with Israeli law the appointment, compensation, retention and oversight of any accounting firm engaged for the purpose of preparing or issuing an audit report or performing other audit services; |
• | recommending the terms of audit and non-audit services to be provided by the independent registered public accounting firm for pre-approval by our board of directors; |
• | recommending the engagement or termination of the person filling the office of our internal auditor; |
• | reviewing with management and our independent directors our financial statements prior to their submission to the SEC; and |
• | approval of certain transactions with office holders and controlling shareholders, as described below, and other related party transactions. |
1. | to recommend to the board of directors the compensation policy for directors and officers, and, once every three years, or five years from a company’s initial public offering, to recommend to the board of directors, whether the compensation policy that had been approved should be extended for a longer period of time; |
2. | to recommend to the board of directors updates to the compensation policy, from time to time, and examine its implementation; |
3. | to decide whether to approve the terms of office and employment of directors and officers that require approval of the compensation committee; and |
4. | to decide whether the compensation terms of the chief executive officer, which were determined pursuant to the compensation policy, will be exempted from approval by the shareholders because such approval would harm the ability to engage the chief executive officer. |
• | the education, skills, experience, expertise and accomplishments of the relevant office holder; |
• | the office holder’s position, responsibilities and prior compensation agreements with him or her; |
• | the ratio between the cost of the terms of employment of an office holder and the cost of the employment of other employees of the company, including employees employed through contractors who provide services to the company, in particular the ratio between such cost, the average and median salary of the employees of the company, as well as the impact of such disparities on the work relationships in the company; |
• | if the terms of employment include variable components — the possibility of reducing variable components at the discretion of the board of directors and the possibility of setting a limit on the exercise value of non-cash variable equity-based components; and |
• | if the terms of employment include severance compensation — the term of employment or office of the office holder, the terms of his or her compensation during such period, the company’s performance during the such period, his or her individual contribution to the achievement of the company goals and the maximization of its profits and the circumstances under which the office holder is leaving the company. |
• | with regard to variable components: |
○ | with the exception of office holders who are subordinate to the chief executive officer, determining the variable components on long-term performance basis and on measurable criteria; however, the company may determine that an immaterial part of the variable components of the compensation package of an office holder shall be awarded based on non-measurable criteria, if such amount is not higher than three monthly salaries per annum while taking into account the office holder’s contribution to the company; |
○ | the ratio between variable and fixed components, as well as the limit of the values of variable components at the time of their grant. |
• | a condition under which the office holder will return to the company, according to conditions to be set forth in the compensation policy, any amounts paid as part of his or her terms of employment, if such amounts were paid based on information later to be discovered to be wrong, and such information was than re-presented in the company’s financial statements; |
• | the minimum holding or vesting period of variable equity-based components, while taking into consideration long-term incentives; and |
• | a limit to retirement grants. |
• | information on the business advisability of a given action brought for his or her approval or performed by virtue of his or her position; and |
• | all other important information pertaining to such action. |
• | refrain from any act involving a conflict of interest between the performance of his or her duties in the company and his or her other duties or personal affairs; |
• | refrain from any activity that is competitive with the business of the company; |
• | refrain from exploiting any business opportunity of the company for the purpose of gaining a personal advantage for himself or herself or others; and |
• | disclose to the company any information or documents relating to the company’s affairs which the office holder received as a result of his or her position as an office holder. |
• | the office holder’s relatives (spouse, siblings, parents, grandparents, descendants, spouse’s descendants and the spouses of any of these people); or |
• | any company in which the office holder or his or her relatives holds 5% or more of the shares or voting rights, serves as a director or general manager or has the right to appoint at least one director or the general manager. |
• | a transaction other than in the ordinary course of business; |
• | a transaction that is not on market terms; or |
• | a transaction that may have a material impact on the company’s profitability, assets or liabilities. |
• | a majority of the shares held by shareholders who have no personal interest in the transaction and are voting at the meeting must be voted in favor of approving the transaction, excluding abstentions; or |
• | the shares voted by shareholders who have no personal interest in the transaction who vote against the transaction represent no more than two percent (2%) of the voting rights in the company. |
• | at least a majority of the shares held by all shareholders who are not controlling shareholders and do not have a personal interest in such matter, present and voting at such meeting, are voted in favor of the compensation package, excluding abstentions; or |
• | the total number of shares of non-controlling shareholders and shareholders who do not have a personal interest in such matter voting against the compensation package does not exceed two percent (2%) of the aggregate voting rights in the company. |
• | an amendment to the articles of association; |
• | an increase in the company’s authorized share capital; |
• | a merger; and |
• | the approval of related party transactions and acts of office holders that require shareholder approval. |
• | a monetary liability incurred by or imposed on the office holder in favor of another person pursuant to a court judgment, including pursuant to a settlement confirmed as judgment or arbitrator’s decision approved by a competent court. However, if an undertaking to indemnify an office holder with respect to such liability is provided in advance, then such an undertaking must be limited to events which, in the opinion of the board of directors, can be foreseen based on the company’s activities when the undertaking to indemnify is given, and to an amount or according to criteria determined by the board of directors as reasonable under the circumstances, and such undertaking shall detail the abovementioned foreseen events and amount or criteria; |
• | reasonable litigation expenses, including reasonable attorneys’ fees, which were incurred by the office holder (i) as a result of an investigation or proceeding filed against the office holder by an authority authorized to conduct such investigation or proceeding; provided that such investigation or proceeding was either (a) concluded without the filing of an indictment against such office holder and without the imposition on him of any monetary obligation in lieu of a criminal proceeding; (b) concluded without the filing of an indictment against the office holder but with the imposition of a monetary obligation on the office holder in lieu of criminal proceedings for an offense that does not require proof of criminal intent; or (ii) in connection with a monetary sanction; |
• | a monetary liability imposed on the office holder in favor of an injured party at an Administrative Procedure (as defined below) as set forth in Section 52(54)(a)(1)(a) to the Securities Law; |
• | expenses expended by the office holder with respect to an Administrative Procedure under the Securities Law, including reasonable litigation expenses and reasonable attorneys’ fees; |
• | reasonable litigation expenses, including attorneys’ fees, incurred by the office holder or which were imposed on the office holder by a court (i) in a proceeding instituted against him or her by the company, on its behalf, or by a third party, (ii) in connection with criminal indictment of which the office holder was acquitted, or (iii) in connection with a criminal indictment which the office holder was convicted of an offense that does not require proof of criminal intent; and |
• | any other obligation or expense in respect of which it is permitted or will be permitted under applicable law to indemnify an office holder. |
• | a breach of the duty of loyalty to the company, provided that the office holder acted in good faith and had a reasonable basis to believe that the act would not harm the company; |
• | a breach of duty of care to the company or to a third party, to the extent such a breach arises out of the negligent conduct of the office holder; |
• | a monetary liability imposed on the office holder in favor of a third party; |
• | a monetary liability imposed on the office holder in favor of an injured party at an Administrative Procedure pursuant to Section 52(54)(a)(1)(a) of the Securities Law; and |
• | expenses incurred by an office holder in connection with an Administrative Procedure, including reasonable litigation expenses and reasonable attorneys’ fees. |
• | a breach of the duty of loyalty, except for indemnification and insurance for a breach of the duty of loyalty to the company to the extent that the office holder acted in good faith and had a reasonable basis to believe that the act would not prejudice the company; |
• | a breach of duty of care committed intentionally or recklessly, excluding a breach arising out of the negligent conduct of the office holder; |
• | an act or omission committed with intent to derive illegal personal benefit; or |
• | a fine or forfeit levied against the office holder. |
• | Forfeiture Upon Cause Termination. All awards held by a participant will be forfeited upon the participant’s termination for cause. |
• | No Repricing Without Shareholder Approval. Without prior shareholder approval, we will not (i) reduce the exercise price of a stock option, (ii) take any other action that is treated as repricing under U.S. GAAP or (ii) repurchase for cash or cancel a stock option when its exercise price is greater than the fair market value of the underlying shares in exchange for another, unless the cancellation and exchange occurs in connection with a change in capitalization or a similar change. |
• | No Transferability. Awards generally may not be transferred, except by will or the laws of descent and distribution, unless otherwise determined by the compensation committee. |
• | No Automatic Grants. The Plan does not provide for automatic grants to any participant. |
• | No Tax Gross-Ups. The Plan does not provide for any tax gross-ups. |
Name | | | Number of Options | | | Exercise Price | | | Date of Grant | | | Expiration Date |
Ran Poliakine | | | 1,206,290 | | | $2.21 | | | November 25, 2019 | | | November 25, 2029 |
Onn Fenig | | | 40,234 | | | $2.21 | | | November 25, 2019 | | | November 25, 2029 |
Floyd Katske | | | 0 | | | N/A | | | N/A | | | N/A |
Erez Meltzer | | | 0 | | | N/A | | | N/A | | | N/A |
Richard Stone | | | 100,584 | | | $2.21 | | | November 25, 2019 | | | November 25, 2029 |
Itzhak Maayan | | | 161,107 | | | $2.21 | | | November 25, 2019 | | | November 25, 2029 |
Anat Kaphan | | | 112,754 | | | $2.21 | | | November 25, 2019 | | | November 25, 2029 |
Yoel Raab | | | 152,754 | | | $2.21 | | | November 25, 2019 | | | November 25, 2029 |
Tal Shank | | | 74,362 | | | $2.21 | | | November 25, 2019 | | | November 25, 2029 |
Shirly Kaufman-Kirshenbaum | | | 0 | | | N/A | | | N/A | | | N/A |
• | each person or entity known by us to own beneficially more than 5% of our outstanding ordinary shares; |
• | each of our directors, executive officers and director nominees; and |
• | all of our executive officers, directors and director nominees as a group. |
| | | Shares Beneficially Owned Prior to the Offering | | | Shares Beneficially Owned After the Offering | |||||||
Name of Beneficial Owner | | | Number | | | Percentage | | | Number | | | Percentage |
5% or greater shareholders | | | | | | | | | ||||
Ran Poliakine(1) | | | | | | | | | ||||
Moshe Moalem(2) | | | | | | | | | ||||
SK Telecom TMT Investment Corp and Affiliates(3) | | | | | | | | | ||||
Tsuri Limited and Everhart Finance Limited(4) | | | | | | | | | ||||
Yozma Group Korea | | | | | | | | | ||||
Asia Beam Limited(5) | | | | | | | | | ||||
Directors, director nominees and executive officers | | | | | | | | | ||||
Ran Poliakine(1) | | | | | | | | | ||||
Onn Fenig | | | | | * | | | | | |||
Erez Meltzer | | | | | * | | | | | |||
Richard Stone(6) | | | | | | | | | ||||
Itzhak Maayan | | | | | * | | | | | |||
Yoel Raab | | | | | * | | | | | |||
Anat Kaphan | | | | | * | | | | | |||
Tal Shank | | | | | * | | | | | |||
Shirly Kaufman-Kirshenbaum | | | | | * | | | | | |||
All directors, director nominees and executive officers as a group (9 persons) | | | | | | | | | ||||
* | Amount represents less than 1% of outstanding ordinary shares. |
(1) | Represents (a) ordinary shares of the Company held by Ran Poliakine, (b) ordinary shares of the Company held in trust by Shay Zuckerman & Co. Law Firm (“Shay Zuckerman”), pursuant to an Escrow Agreement, dated February 3, 2020 (the “Escrow Agreement”), between Ran Poliakine, Moshe Moalem and Shay Zuckerman, as trustee, and (c) warrants to purchase ordinary shares held by Ran Poliakine. ordinary shares held by Shay Zuckerman are held in trust for the benefit of Ran Poliakine. Ran Poliakine has voting power of all the ordinary shares held in trust by Shay Zuckerman. Ran Poliakine and Moshe Moalem may be deemed to share the dispositive power over the ordinary shares held in trust by Shay Zuckerman as such ordinary shares may not be disposed of until a final settlement between Ran Poliakine and Moshe Moalem is reached with respect thereto. |
(2) | Represents (a) ordinary shares of the Company held by Moshe Moalem and (b) ordinary shares of the Company held in trust by Shay Zuckerman pursuant to the Escrow Agreement. Ran Poliakine and Moshe Moalem may be deemed to share the dispositive power over the ordinary shares held in trust by Shay Zuckerman as such ordinary shares may not be disposed of until a final settlement between Ran Poliakine and Moshe Moalem is reached with respect thereto. |
(3) | Represents ordinary shares held by SKT, ordinary shares held by Pureun Partners Asset Management Co., Ltd. (“Pureun”), ordinary shares held by EBEST-PPAM Fund No. 9 (“EBEST”), and warrants held by SKT to purchase ordinary shares. SKT has the voting and dispositive power of the shares held by Pureun and EBEST pursuant to a proxy. |
(4) | Represents ordinary shares held by Tsuri Limited and ordinary shares held by Everhart Finance Limited. The voting and dispositive power over such ordinary shares is ultimately held by Elie Douer and Marie Douer, and each of Elie Douer and Marie Douer may be deemed to share voting and dispositive power over the shares held by Tsuri Limited and Everhart Finance Limited. |
(5) | Represents ordinary shares held by Asia Beam Limited. The voting and dispositive power over such ordinary shares is ultimately held by Kasudjono Harianto. |
(6) | Consists of ordinary shares, options to purchase ordinary shares, and warrants to purchase ordinary shares held by Richard Stone, 696,196 ordinary shares and warrants to purchase ordinary shares held by Stone Isra Ventures LLC, and ordinary shares and warrants to purchase ordinary shares held by Adhoc Investors LLC. Richard Stone is the sole shareholder of Stone Isra Ventures LLC and Adhoc Investors LLC, and may be deemed to have voting and dispositive power of the ordinary shares held by Stone Isra Ventures LLC and Adhoc Investors LLC. |
• | amendments to our amended and restated articles of association; |
• | appointment or termination of our auditors; |
• | election of directors, including external directors (if applicable); |
• | approval of certain related party transactions; |
• | increases or reductions of our authorized share capital; |
• | mergers; and |
• | the exercise of our board of director’s powers by a general meeting, if our board of directors is unable to exercise its powers and the exercise of any of its powers is required for our proper management. |
• | 1% of the number of ordinary shares then outstanding; or |
• | the greater of 1% or the average weekly trading volume of our ordinary shares on the Nasdaq during the four calendar weeks preceding the filing of a notice on Form 144 with respect to the sale. |
• | persons other than affiliates, without restriction; and |
• | affiliates, subject to the manner-of-sale, current public information and filing requirements of Rule 144, in each case, without compliance with the six-month holding period requirement of Rule 144. |
• | Amortization over an eight-year period of the cost of purchased know-how and patents and rights to use a patent and know-how which are used for the development or advancement of the Industrial Enterprise, commencing from the tax year where the Industrial Enterprise began to use them. |
• | Under limited conditions, an election to file consolidated tax returns with related Israeli Industrial Companies; and |
• | Expenses related to a public offering are deductible in equal amounts from income attributed to the Industrial Enterprise over three years commencing in the year of the offering. |
• | The expenditures are approved by the relevant Israeli government ministry, determined by the field of research; or |
• | The research and development is for the promotion of the company and is carried out by or on behalf of the company seeking such tax deduction. |
• | banks and other financial institutions; |
• | insurance companies; |
• | pension plans; |
• | cooperatives; |
• | regulated investment companies; |
• | real estate investment trusts; |
• | broker-dealers; |
• | traders that elect to use a mark-to-market method of accounting; |
• | certain former U.S. citizens or long-term residents; |
• | tax-exempt entities (including private foundations); |
• | holders who acquire our ordinary shares pursuant to any employee share option or otherwise as compensation; |
• | investors that will hold our ordinary shares as part of a straddle, hedge, conversion, constructive sale or other integrated transaction for U.S. federal income tax purposes; |
• | persons holding our ordinary shares in connection with a trade or business outside the United States; |
• | persons that actually or constructively own 10% or more of our stock (by vote or value); |
• | investors that have a functional currency other than the U.S. dollar; |
• | partnerships or other entities taxable as partnerships for U.S. federal income tax purposes, or persons holding our ordinary shares through such entities, all of whom may be subject to tax rules that differ significantly from those discussed below. |
• | an individual who is a citizen or resident of the United States; |
• | a corporation (or other entity treated as a corporation for U.S. federal income tax purposes) created in, or organized under the law of, the United States or any state thereof or the District of Columbia; |
• | an estate the income of which is includible in gross income for U.S. federal income tax purposes regardless of its source; or |
• | a trust (A) the administration of which is subject to the primary supervision of a U.S. court and which has one or more U.S. persons who have the authority to control all substantial decisions of the trust or (B) that has otherwise validly elected to be treated as a U.S. person under the Code. |
• | the excess distribution or gain will be allocated ratably over the U.S. Holder’s holding period for the ordinary shares; |
• | the amount allocated to the taxable year of the excess distribution, sale or other disposition and to any taxable years in the U.S. Holder’s holding period prior to the first taxable year in which we are classified as a PFIC (each, a “pre-PFIC year”), will be taxable as ordinary income; |
• | the amount allocated to each prior taxable year, other than a pre-PFIC year, will be subject to tax at the highest tax rate in effect for individuals or corporations, as appropriate, for that year; and |
• | the interest charge generally applicable to underpayments of tax will be imposed on the tax attributable to each prior taxable year, other than a pre-PFIC year. |
Name | | | Number of shares |
Cantor Fitzgerald & Co. | | | |
Oppenheimer & Co. Inc. | | | |
Berenberg Capital Markets, LLC | | | |
CIBC World Markets Corp. | | | |
Total | | |
| | | Without option to purchase additional shares exercise | | | With full option to purchase additional shares exercise | |
Per Share | | | $ | | | $ |
Total | | | $ | | | $ |
• | offer, pledge, sell, contract to sell, sell any option or contract to purchase, purchase any option or contract to sell, grant any option, right or warrant to purchase, lend, or otherwise transfer or dispose of, directly or indirectly, any of our ordinary shares or any securities convertible into or exercisable or exchangeable for our ordinary shares (including without limitation, ordinary shares or such other securities which may be deemed to be beneficially owned by the such persons in accordance with the rules and regulations of the SEC and securities which may be issued upon exercise of a share option or warrant); |
• | enter into any hedging, swap or other agreement or transaction that transfers, in whole or in part, any of the economic consequences of ownership of our ordinary shares or such other securities; or |
• | make any demand for or exercise any right with respect to the registration of any of our ordinary shares or any security convertible into or exercisable or exchangeable for our ordinary shares, or publicly disclose the intention to do any of the foregoing. |
• | offer, pledge, sell, contract to sell, sell any option or contract to purchase, purchase any option or contract to sell, grant any option, right or warrant to purchase, lend, or otherwise transfer or dispose of, directly or indirectly, or submit to, or file with, the SEC a registration statement under the Securities Act relating to, any of our ordinary shares or any securities convertible into or exercisable or exchangeable for our ordinary shares, or publicly disclose the intention to undertake any of the foregoing; or |
• | enter into any swap or other agreement that transfers, in whole or in part, any of the economic consequences of ownership of our ordinary shares or any such other securities, without the prior written consent of Cantor Fitzgerald & Co. |
• | the information set forth in this prospectus and otherwise available to the representative; |
• | our prospects and the history and prospects for the industry in which we compete; |
• | an assessment of our management; |
• | our prospects for future earnings; |
• | the general condition of the securities markets at the time of this offering; |
• | the recent market prices of, and demand for, publicly traded equity securities of generally comparable companies; and |
• | other factors deemed relevant by the underwriters and us. |
(a) | to any legal entity which is a qualified investor as defined under the Prospectus Regulation; |
(b) | to fewer than 150 natural or legal persons (other than qualified investors as defined under the Prospectus Regulation), subject to obtaining the prior consent of the underwriters for any such offer; or |
(c) | in any other circumstances falling within Article 1(4) of the Prospectus Regulation, |
(a) | a corporation (which is not an accredited investor (as defined in Section 4A of the SFA)) the sole business of which is to hold investments and the entire share capital of which is owned by one or more individuals, each of whom is an accredited investor; or |
(b) | a trust (where the trustee is not an accredited investor) whose sole purpose is to hold investments and each beneficiary of the trust is an individual who is an accredited investor, securities (as defined in Section 239(1) of the SFA) of that corporation or the beneficiaries’ rights and interest (howsoever described) in that trust shall not be transferred within six months after that corporation or that trust has acquired the shares pursuant to an offer made under Section 275 of the SFA except: |
(a) | to an institutional investor or to a relevant person defined in Section 275(2) of the SFA, or to any person arising from an offer referred to in Section 275(1A) or Section 276(4)(i)(B) of the SFA; |
(b) | where no consideration is or will be given for the transfer; |
(c) | where the transfer is by operation of law; |
(d) | as specified in Section 276(7) of the SFA; or |
(e) | as specified in Regulation 32 of the Securities and Futures (Offers of Investments) (Shares and Debentures) Regulations 2005 of Singapore. |
Itemized expense | | | Amount |
SEC registration fee | | | $12,980 |
FINRA filing fee | | | 15,500 |
Nasdaq listing fee | | | * |
Printing and engraving expenses | | | * |
Legal fees and expenses | | | * |
Transfer agent and registrar fees | | | * |
Accounting fees and expenses | | | * |
Miscellaneous | | | * |
Total | | | $* |
* | To be filed by amendment. |
• | the judgment was rendered by a court which was, according to the laws of the state of the court, competent to render the judgment; |
• | the obligation imposed by the judgment is enforceable according to the rules relating to the enforceability of judgments in Israel and the substance of the judgment is not contrary to public policy; and |
• | the judgment is executory in the state in which it was given. |
• | the judgment was given in a state whose laws do not provide for the enforcement of judgments of Israeli courts (subject to exceptional cases); |
• | the enforcement of the judgment is likely to prejudice the sovereignty or security of the State of Israel; |
• | the judgment was obtained by fraud; |
• | the opportunity given to the defendant to bring its arguments and evidence before the court was not reasonable in the opinion of the Israeli court; |
• | the judgment was rendered by a court not competent to render it according to the laws of private international law as they apply in Israel; |
• | the judgment is contradictory to another judgment that was given in the same matter between the same parties and that is still valid; or |
• | at the time the action was brought in the foreign court, a lawsuit in the same matter and between the same parties was pending before a court or tribunal in Israel. |

/s/ Kesselman & Kesselman | | | |
Certified Public Accountants (Isr.) | | | |
A member firm of PricewaterhouseCoopers International Limited | | | |
| | | ||
Tel Aviv, Israel | | | |
February 18, 2020, except with respect to the matters which have removed the substantial doubt about the Company’s ability to continue as a going concern discussed in Note 1d and Note 12(d), (e), (f), (g) as to which the date is July 30, 2020 | | |
| | | December 31, | ||||
| | | 2019 (*) | | | 2018 (*) | |
| | | U.S. Dollars in thousands | ||||
Assets | | | | | ||
CURRENT ASSETS: | | | | | ||
Cash and cash equivalents | | | 8,072 | | | 5 |
Prepaid expenses and other current assets | | | 1,564 | | | — |
Related party prepaid expenses | | | — | | | 1,694 |
Total current assets | | | 9,636 | | | 1,699 |
| | | | | |||
NON-CURRENT ASSETS: | | | | | ||
Restricted cash | | | 145 | | | — |
Property and equipment, net | | | 228 | | | 156 |
Deferred offering costs | | | 1,197 | | | — |
Operating lease right-of-use asset | | | 526 | | | — |
Other non-current assets | | | 139 | | | — |
Total non-current assets | | | 2,235 | | | 156 |
Total assets | | | 11,871 | | | 1,855 |
| | | | | |||
Liabilities and Capital Deficiency | | | | | ||
CURRENT LIABILITIES: | | | | | ||
Accounts payable | | | 475 | | | 82 |
Accrued expenses and other liabilities | | | 1,828 | | | — |
Related party liability | | | 17,820 | | | 8,157 |
Current maturities of operating leases | | | 140 | | | — |
Total current liabilities | | | 20,263 | | | 8,239 |
| | | | | |||
NON-CURRENT LIABILITIES: | | | | | ||
Non-current operating leases | | | 386 | | | — |
Total non-current liabilities | | | 386 | | | — |
Total liabilities | | | 20,649 | | | 8,239 |
COMMITMENTS | | | | | ||
SHAREHOLDERS' DEFICIT: | | | | | ||
Ordinary Shares, par value NIS 0.01 per share, 40,000,000 and 30,000,000 shares authorized at December 31, 2019 and 2018, respectively; 27,150,080 and 21,924,208 issued and outstanding at December 31, 2019 and 2018, respectively | | | 75 | | | 58 |
Additional paid-in capital | | | 31,748 | | | 11,596 |
Accumulated deficit | | | (40,601) | | | (18,038) |
TOTAL SHAREHOLDERS' DEFICIT | | | (8,778) | | | (6,384) |
TOTAL LIABILITIES AND SHAREHOLDERS' DEFICIT | | | 11,871 | | | 1,855 |
(*) | The consolidated financial statements as of and for the years ended December 31, 2019 and 2018 reflect a retrospective application of a transaction under common control - see note 1c |
| | | Year ended December 31, | ||||
| | | 2019 (*) | | | 2018 (*) | |
| | | U.S. Dollars in thousands | ||||
OPERATING EXPENSES: | | | | | ||
Research and development | | | 2,717 | | | 672 |
Marketing | | | 1,556 | | | 209 |
General and administrative | | | 18,298 | | | 1,023 |
TOTAL OPERATING EXPENSES | | | 22,571 | | | 1,904 |
OPERATING LOSS | | | (22,571) | | | (1,904) |
FINANCIAL (INCOME) EXPENSES, net | | | (8) | | | 5 |
NET LOSS | | | (22,563) | | | (1,909) |
BASIC AND DILUTED LOSS PER SHARE | | | (0.90) | | | (0.09) |
THE WEIGHTED AVERAGE OF THE NUMBER OF ORDINARY SHARES (in thousands) | | | 25,181 | | | 20,793 |
(*) | The consolidated financial statements as of and for the years ended December 31, 2019 and 2018 reflect a retrospective application of a transaction under common control - see note 1c |
| | | Ordinary shares | | | Additional paid-in capital | | | Accumulated deficit | | | Total | ||||
| | | Number of shares | | | Amount | | | U.S. Dollars in thousands | |||||||
BALANCE AT JANUARY 1, 2018 | | | 20,257,434 | | | 41 | | | 7,814 | | | (16,129) | | | (8,274) |
CHANGES DURING 2018: | | ||||||||||||||
Issuance of ordinary shares | | | 1,666,774 | | | 17 | | | 3,667 | | | | | 3,684 | |
Share-based compensation | | | | | | | 115 | | | | | 115 | |||
Net loss for the year | | | | | | | | | (1,909) | | | (1,909) | |||
BALANCE AT DECEMBER 31, 2018 | | | 21,924,208 | | | 58 | | | 11,596 | | | (18,038) | | | (6,384) |
CHANGES DURING 2019: | | | | | | | | | | | |||||
Issuance of ordinary shares and warrants, net of issuance costs | | | 4,762,656 | | | 16 | | | 14,022 | | | | | 14,038 | |
Issuance of ordinary shares upon exercise of warrants | | | 454,166 | | | 1 | | | 136 | | | | | 137 | |
Issuance of ordinary shares to investors upon exercise of warrants | | | 9,050 | | | ** | | | 25 | | | | | 25 | |
Share-based compensation | | | | | | | 16,245 | | | | | 16,245 | |||
Additional consideration with respect to an assets purchase agreement, see note 1c and note 6 | | | | | | | (10,276) | | | | | (10,276) | |||
Net loss for the year | | | | | | | | | (22,563) | | | (22,563) | |||
BALANCE AT DECEMBER 31, 2019 | | | 27,150,080 | | | 75 | | | 31,748 | | | (40,601) | | | (8,778) |
(*) | The consolidated financial statements as of and for the years ended December 31, 2019 and 2018 reflect a retrospective application of a transaction under common control - see note 1c |
(**) | Less than 1 thousand US dollars. |
| | | Year ended December 31, | ||||
| | | 2019 (*) | | | 2018 (*) | |
| | | U.S. Dollars in thousands | ||||
CASH FLOWS FROM OPERATING ACTIVITIES: | | | | | ||
Net loss for the year | | | (22,563) | | | (1,909) |
Adjustments required to reconcile net loss to net cash used in operating activities: | | | | | ||
Share-based compensation | | | 16,245 | | | 115 |
Depreciation | | | 53 | | | 35 |
Changes in operating assets and liabilities: | | |||||
Prepaid expenses and other current assets | | | (1,564) | | | 66 |
Related party prepaid expenses | | | 1,081 | | | (1,844) |
Other non-current assets | | | (139) | | | — |
Accounts payable | | | 393 | | | (134) |
Operating lease | | | ** | | | — |
Accrued expenses and other liabilities | | | 970 | | | — |
Net cash used in operating activities | | | (5,524) | | | (3,671) |
| | | | | |||
CASH FLOWS FROM INVESTING ACTIVITIES: | | | | | ||
Purchase of property and equipment | | | (125) | | | (73) |
Net cash used in investing activities | | | (125) | | | (73) |
| | | | | |||
CASH FLOWS FROM FINANCING ACTIVITIES: | | | | | ||
Proceeds from issuance of ordinary shares and warrants , net of issuance costs | | | 14,038 | | | 3,684 |
Proceeds from issuance of ordinary shares upon exercise of warrants | | | 162 | | | — |
Deferred offering costs | | | (339) | | | — |
Net cash provided by financing activities | | | 13,861 | | | 3,684 |
| | | | | |||
NET CHANGE IN CASH AND CASH EQUIVALENTS AND RESTRICTED CASH | | | 8,212 | | | (60) |
CASH AND CASH EQUIVALENTS AND RESTRICTED CASH AT BEGINNING OF THE YEAR | | | 5 | | | 65 |
CASH AND CASH EQUIVALENTS AND RESTRICTED CASH AT END OF THE YEAR | | | 8,217 | | | 5 |
SUPPLEMENTARY INFORMATION ON ACTIVITIES NOT INVOLVING CASH FLOWS | | | | | ||
Unpaid offering costs | | | 858 | | | — |
Recognition of operating lease right-of-use asset against operating lease liabilities | | | 548 | | | — |
Additional consideration with respect to an assets purchase agreement, see note 1c | | | (10,276) | | | — |
(*) | The consolidated financial statements as of and for the years ended December 31, 2019 and |
(**) | Less than 1 thousand US dollars. |
a. | Nano-X Imaging Ltd., an Israeli company (hereinafter “the Company” or “Nanox IL” or “the Successor Company”), was incorporated on December 20, 2018 and commenced its operations on September 3, 2019. |
b. | Nanox Imaging PLC is a public limited company incorporated in Gibraltar in 2012 (hereinafter “Nanox PLC” or “the predecessor company”). |
c. | As of September 3 2019, Nanox IL and Nanox PLC had the same shareholders and, therefore, the transaction was treated as a transaction under common control for accounting purposes. |
1) | only the net assets that were transferred in the transaction according to the APA. Net assets which were not transferred in the transaction are not reflected in these consolidated financial statements. |
2) | no interests of Nanox Japan were transferred under the APA. The consolidated statements of operation include the costs incurred for services provided by Nanox Japan to Nanox PLC. |
NOTES TO THE FINANCIAL STATEMENTS(continued)
3) | the consideration in the transaction (the “Related Party Liability”) was recorded at the beginning of the earliest period presented against a decrease in shareholders' equity, with the exception of the cash consideration that was received by Nanox PLC from its equity financing activities in 2019, and which was recorded in 2019 (refer to note 8a). |
4) | all of the share-related information reflect the share information of Nanox IL. |
d. | In accordance with Accounting Standards Update (“ASU”) 2014-15, Presentation of Financial Statements–Going Concern (Subtopic 205-40): Disclosure of Uncertainties About an Entity’s Ability to Continue as a Going Concern, management is required to perform a two-step analysis of its ability to continue as a going concern. Management must first evaluate whether there are conditions and events that raise substantial doubt about the Company’s ability to continue as a going concern (step 1). If management concludes that substantial doubt is raised, management is also required to consider whether its plans alleviate that doubt (step 2). |
a. | Use of estimates in the preparation of financial statements |
b. | Functional currency |
NOTES TO THE FINANCIAL STATEMENTS(continued)
c. | Statement of Cash Flows |
d. | Cash and cash equivalents |
e. | Restricted Cash |
f. | Property and equipment, net |
| | | % | |
Computers | | | 10-33 |
Office furniture and lab equipment | | | 10-20 |
g. | Impairment of long-lived assets |
h. | Severance pay |
NOTES TO THE FINANCIAL STATEMENTS(continued)
i. | Legal and other contingencies |
j. | Research and development expenses |
k. | Marketing expenses |
l. | Income tax |
1) | The Company accounts for income taxes in accordance with ASC 740, “Income Taxes” (“ASC 740”). ASC 740 prescribes the use of the liability method whereby deferred tax asset and liability account balances are determined based on differences between the financial reporting and tax bases of assets and liabilities and are measured using the enacted tax rates and laws that will be in effect when the differences are expected to reverse. The Company provides a valuation allowance, if necessary, to reduce deferred tax assets to their estimated realizable value if it is more likely than not that a portion or all of the deferred tax assets will not be realized, based on the weight of available positive and negative evidence. Deferred tax liabilities and assets are classified as non-current in accordance with ASU 2015-17. |
2) | Taxes that would apply in the event of disposal of investments in foreign subsidiaries have not been taken into account in computing the deferred income taxes, as it is the Company’s intent and ability to hold these investments. |
NOTES TO THE FINANCIAL STATEMENTS(continued)
m. | Share-based compensation |
n. | Loss per share |
o. | Fair value measurement |
p. | Deferred Offering Costs |
NOTES TO THE FINANCIAL STATEMENTS(continued)
q. | Newly issued and recently adopted accounting pronouncements: |
(i) | In February 2016, the FASB issued ASU No. 2016-02, Leases (Topic 842), which supersedes the existing guidance for lease accounting, Leases (Topic 840). The guidance, along with amendments that were adopted thereafter, requires entities to record lease assets and lease liabilities on the balance sheet for all leases (unless an exception is applied) and disclose key information about leasing arrangements. The Company adopted the new lease standard on January 1, 2019 and used the effective date as the Company's date of initial application. |
(a) | Not reassess whether any existing contracts are or contain a lease; |
(b) | Not reassess the classification of leases that commenced before the effective date (for example, all existing leases that were classified as operating leases in accordance with Topic 840 will continue to be classified as operating leases, and all existing leases that were classified as capital leases in accordance with Topic 840 will continue to be classified as finance leases); |
(c) | Exclude initial direct costs from measurement of the right of use asset at the date of initial application. |
(ii) | See note 2(m) for other accounting pronouncement adopted during the year ended December 31, 2019. |
| | | December 31, | ||||
| | | 2019 | | | 2018 | |
| | | (U.S. Dollars in thousands) | ||||
Office furniture and lab equipment | | | 325 | | | 217 |
Computers | | | 39 | | | 22 |
| | | 364 | | | 239 | |
Less: accumulated depreciation | | | 136 | | | 83 |
Total property and equipment, net | | | 228 | | | 156 |
NOTES TO THE FINANCIAL STATEMENTS(continued)
| | | December 31, | ||||
| | | 2019 | | | 2018 | |
| | | (U.S. Dollars in thousands) | ||||
Cash and cash equivalents | | | 8,072 | | | 5 |
Restricted bank deposit | | | 145 | | | — |
Total cash, cash equivalents and restricted cash shown in the statement of cash flows | | | 8,217 | | | 5 |
| | | Year ended December 31, | |
| | | 2019 | |
| | | (U.S. Dollars in thousands) | |
Operating lease cost: | | | |
Fixed payments | | | 25 |
Short-term lease cost | | | 112 |
Total operating lease cost | | | 137 |
| | | Year ended December 31, | |
| | | 2019 | |
| | | (U.S. Dollars in thousands) | |
Cash paid for amounts included in the measurement of lease liabilities: | | | |
Operating cash flows from operating leases | | | 25 |
Right-of-use assets obtained in exchange for lease obligations (non-cash): | | | |
Operating leases | | | 548 |
| | | December 31, | |
| | | 2019 | |
| | | (U.S. Dollars in thousands) | |
Operating leases: | | | |
Operating lease right-of-use assets | | | 526 |
Current maturities of operating leases | | | 140 |
Non-current operating leases | | | 386 |
Total operating lease liabilities | | | 526 |
NOTES TO THE FINANCIAL STATEMENTS(continued)
| | | December 31, | |
| | | 2019 | |
Weighted average remaining lease term | | | |
Operating leases | | | 3.96 years |
| | | ||
Weighted average discount rate | | | |
Operating leases | | | 5.6% |
| | | December 31, | |
| | | 2019 | |
| | | (U.S. Dollars in thousands) | |
2020 | | | 146 |
2021 | | | 146 |
2022 | | | 146 |
2023 | | | 145 |
2024 and thereafter | | | — |
Total operating lease payments | | | 583 |
Less: imputed interest | | | 57 |
Present value of lease liabilities | | | 526 |
a. | Nanox Japan has been using two rooms and one clean room at the premises of the University of Tokyo since 2012. The total annual payments in 2019 were approximately $76 thousand. |
b. | As to the agreements for services with Six-Eye Interactive Ltd. (“Six-Eye”) – refer to note 10c. |
c. | Nanox IL entered into an advisory agreement with A-Labs Finance and Advisory Ltd. (“A-Labs”), effective February 1, 2019, as amended on October 18, 2019, pursuant to which A-Labs will provide the Company |
NOTES TO THE FINANCIAL STATEMENTS(continued)
d. | During September 2019, the Company entered into a Service Agreement with RMD AP Limited, a company registered under the laws of Hong Kong (RMD). RMD undertook to provide the Company with services related to the Asia Pacific region, including, among others, operational and business development related matters. The agreement is for a period of one year and the agreed commitment for the services is $800 thousand. RMD will bear all costs and expenses incurred beyond such agreed upon amount. |
e. | During September 2019, Nanox IL entered into a Collaboration Agreement with Hadasit Medical Research Services and Development Ltd. (“Hadasit”), a wholly owned subsidiary of the Hadassah Medical Organization. The parties agreed to collaborate with respect to the Company's medical imaging technology and resulting medical images devices (the “Company Products”), by way of (a) joint research and development projects (each, a “Research Project”), and (b) the provision by Hadasit of services in connection with the Company Products, such as testing and consulting work, where no innovative research will be carried out (each, a “Service”). Each Research Project and Service shall be rendered under a separate project agreement concluded between the parties in writing from time to time (collectively, the “Project Agreements”). The parties envisage the collaboration to continue over a period of five years, unless an extension is agreed by the parties in writing. Under such agreement, the Company paid Hadasit a non-refundable advance payment for the Research Projects and Services, in the amount of $ 250 thousand, which shall be credited against payments due from time to time to Hadasit under the Project Agreements. Nanox IL has no obligation to enter into Project Agreements with Hadasit in excess of such advanced payment. |
f. | On December 16, 2019, Nanox IL signed an agreement with Dr. Ilung Kim for provision of services to the Company. Dr. Kim will not receive any cash compensation, but will be granted options to purchase 1,206,290 ordinary shares with an exercise price of $2.21 per ordinary share. 301,572 of the options vested as of the grant date and the remaining 904,718 options will vest in equal monthly installments over a period of three years from the vesting commencement date. In the event of an IPO or Deemed Liquidation (as defined therein), all unvested options shall fully accelerate immediately prior to the consummation of the IPO. The vested options shall be exercisable until the earlier of (a) the second anniversary of termination of the engagement by and among the Company and Dr. Ilung Kim, or (b) the tenth anniversary from the date of grant. |
NOTES TO THE FINANCIAL STATEMENTS(continued)
a. | Share capital |
b. | Share based compensation |
1) | Share-based compensation to non-employees : |
NOTES TO THE FINANCIAL STATEMENTS(continued)
| | | Year ended December 31, | | | Year ended December 31, | |||||||
| | | 2019 | | | 2018 | |||||||
| | | Number of share-based payment awards | | | Weighted average exercise price | | | Number of share-based payment awards | | | Weighted average payment exercise price | |
Outstanding at beginning of year | | | 1,592,874 | | | $1.32 | | | 1,751,140 | | | $1.40 |
Changes during the year: | | |||||||||||
Granted | | | 2,271,698 | | | $2.77 | | | — | | | — |
Exercised | | | (454,166) | | | $0.3 | | | — | | | — |
Forfeited | | | — | | | — | | | — | | | — |
Expired | | | — | | | — | | | — | | | — |
Cancelled | | | — | | | — | | | (158,266) | | | $2.21 |
Outstanding at end of year | | | 3,410,406 | | | $1.89 | | | 1,592,874 | | | $1.32 |
Exercisable at end of year | | | 1,830,809 | | | $3.79 | | | 1,592,874 | | | $1.32 |
| | | 2019 | | | 2018 | |
Fair value of one ordinary share | | | 16 | | | 2.21 |
Dividend yield | | | 0 | | | 0 |
Expected volatility | | | 41.11% - 50.59% | | | 51.97% - 72.25% |
Risk-free interest rate | | | 1.55%-1.76% | | | 1.52%-2.94% |
Contractual term (years) | | | 0.50 – 10.00 | | | 1.05 – 6.00 |
NOTES TO THE FINANCIAL STATEMENTS(continued)
| | | December 31, 2018 | ||||||||||
Exercise price | | | Awards outstanding | | | Awards exercisable | ||||||
| | Number of awards outstanding at end of year | | | Weighted average remaining contractual life (years) | | | Number of awards exercisable at end of year | | | Weighted average remaining contractual life (years) | ||
$0.01 | | | 186,815 | | | 2.33 | | | 186,815 | | | 2.33 |
$0.30 | | | 454,166 | | | 0.77 | | | 454,166 | | | 0.77 |
$1.92 | | | 472,606 | | | 2.81 | | | 472,606 | | | 2.81 |
$2.21 | | | 479,287 | | | 3.37 | | | 479,287 | | | 3.37 |
| | | December 31, 2019 | ||||||||||
Exercise price | | | Awards outstanding | | | Awards exercisable | ||||||
| | Number of awards outstanding at end of year | | | Weighted average remaining contractual life (years) | | | Number of awards exercisable at end of year | | | Weighted average remaining contractual life (years) | ||
$0.01 | | | 186,815 | | | 1.33 | | | 186,815 | | | 1.33 |
$1.92 | | | 472,606 | | | 1.81 | | | 472,606 | | | 1.81 |
$2.21 | | | 2,727,028 | | | 5.27 | | | 1,163,402 | | | 5.27 |
$20.87 | | | 23,957 | | | 5.69 | | | 7,986 | | | 5.69 |
2) | Share-based compensation to employees, officers and directors |
| | | Year ended December 31, 2019 | ||||
| | | Number of share-based payment awards | | | Weighted average exercise price | |
Outstanding at beginning of year | | | — | | | — |
Changes during the year: | | |||||
Granted | | | 1,667,267 | | | 2.21 |
Exercised | | | — | | | — |
Forfeited | | | — | | | — |
Expired | | | — | | | — |
Cancelled | | | — | | | — |
Outstanding at end of year | | | 1,667,267 | | | 2.21 |
Exercisable at end of year | | | 450,557 | | | 2.21 |
NOTES TO THE FINANCIAL STATEMENTS(continued)
| | | 2019 | |
Fair value of one ordinary share | | | 16 |
Dividend yield | | | 0 |
Expected volatility | | | 46.1% - 50.65% |
Risk-free interest rate | | | 1.65% - 1.68% |
Contractual term (years) | | | 5.75 - 6.23 |
December 31, 2019 | ||||||||||||
Awards outstanding | | | Awards exercisable | |||||||||
Exercise price | | | Number of awards outstanding at end of year | | | Weighted average remaining contractual life (years) | | | Number of awards exercisable at end of year | | | Weighted average remaining contractual life (years) |
$2.21 | | | 1,667,267 | | | 9.90 | | | 450,557 | | | 9.90 |
3) | (i) Share-based compensation expenses for awards granted to non-employees, employees, officers and directors in the amount of $661 thousand included in the Company’s consolidated statements of operations for the year ended December 31, 2019 were recorded as research and development expenses. |
a. | Basis of taxation |
NOTES TO THE FINANCIAL STATEMENTS(continued)
b. | Tax assessments |
c. | Deferred tax assets |
a. | Balances with related parties: |
| | | December 31, | ||||
| | | 2019 | | | 2018 | |
| | | (U.S. Dollars in thousands) | ||||
Related party prepaid expenses – See d below | | | — | | | 1,694 |
Related party liability, refer to note 6 | | | 17,820 | | | 8,157 |
b. | Related parties transactions: |
| | | Year ended December 31, | ||||
| | | 2019 | | | 2018 | |
| | | (U.S. Dollars in thousands) | ||||
Research and development | | | 154 | | | 542 |
General and administrative | | | 5,824 | | | 892 |
c. | Six-Eye agreements for services |
NOTES TO THE FINANCIAL STATEMENTS(continued)
d. | The related party prepaid expenses reflect funds raised during 2018 (refer to note 8a) in an amount of $ 3,684 thousand from third party investors, less amounts payable in accordance with Six-Eye service agreement (see c above). The funds were received directly by Six-Eye (these funds were not received by the Company nor remitted from the Company to Six-Eye) less amounts payable in accordance with Six-Eye service agreement (see c above). |
e. | Effective from September 2019, Nanox IL signed an executive employment agreement with Ran Poliakine (“the CEO”) to serve as the Company’s CEO (“the CEO agreement”). According to the CEO agreement, the CEO will be entitled to a monthly gross salary of $40 thousand, which will be increased to $60 thousand upon the Company's consummation of an IPO. The CEO will be entitled to other benefits as described in the CEO agreement including an annual bonus subject to performance criteria. The CEO was granted options to purchase 1,206,290 ordinary shares with an exercise price of $2.21 per ordinary share. 301,572 of the options were vested as of the grant date and the remaining 904,718 options will be vested in equal monthly installments over a period of three years from the vesting commencement date. Share-based compensation expenses in an amount of $5,299 thousand included in the Company’s consolidated statements of operations for the year ended December 31, 2019 were recorded as general and administrative expenses for the abovementioned awards. |
a. | Basic |
| | | Year ended December 31, | ||||
| | | 2019 | | | 2018 | |
Net loss attributable to Company’s owners | | | $(22,563) | | | $(1,909) |
The weighted average of the number of ordinary shares (in thousands) | | | 25,181 | | | 20,793 |
Basic and diluted loss per share | | | $(0.90) | | | $(0.09) |
b. | Diluted |
NOTES TO THE FINANCIAL STATEMENTS(continued)
a. | During January 2020, subject to entering into a share purchase agreement in the aggregate amount of at least $6 million, and a pre-money valuation of more than $100 million, the Nanox IL’s Board approved the issuance and allotment of 1,109,245 ordinary shares to Nanox PLC with the purchase price of $12.00 per share, which reflects a discount of 25% from the price of the last financing round of the Company. As a result, on January 30, 2020 the related party liability was settled into equity at a price per share reflecting a discount of 25% from the price of the last financing round. |
b. | During January and February 2020 Nanox IL issued additional 257,723 ordinary shares to other investors for an aggregate purchase price of approximately $3.4 million at a $16 per ordinary share. |
c. | In February 2020, the Company granted a total of 407,868 awards with an exercise price of $2.21 per share to employees and service providers. |
d. | During the first half of 2020, the Company entered into share purchase agreements with certain investors (together, the “Investors”), under which the Company issued an aggregate of 2,368,250 ordinary shares to the Investors, at a price per share of $16.00, for an aggregate purchase price of approximately $37.9 million. |
e. | In July 2020, the Company issued 625,000 ordinary shares to one of its investors, as part of their $20 million equity investment at a price per share of $16.00, for a purchase price of $10 million, received in July 2020, which reflects the second portion of the investor’s $20 million investment. |
f. | In July 2020, the Company entered into additional share purchase agreements with certain investors for the issuance of 3,500,000 ordinary shares at a price per share of $16.00, for an aggregate purchase price of approximately $56 million. As of the date of the issuance of these financial statements, the funds were not received yet. |
g. | The Company has evaluated subsequent events through July 30, 2020, the date on which the consolidated financial statements were available to be issued. |
| | | June 30, 2020 | | | December 31, 2019 | |
| | | U.S. Dollars in thousands | ||||
Assets | | | | | ||
CURRENT ASSETS: | | | | | ||
Cash and cash equivalents | | | 39,524 | | | 8,072 |
Related party prepaid expenses | | | 35 | | | — |
Prepaid expenses and other current assets | | | 890 | | | 1,564 |
TOTAL CURRENT ASSETS | | | 40,449 | | | 9,636 |
| | | | | |||
NON-CURRENT ASSETS: | | | | | ||
Restricted cash | | | 192 | | | 145 |
Property and equipment, net | | | 439 | | | 228 |
Deferred offering costs | | | 1,469 | | | 1,197 |
Operating lease right-of-use asset | | | 934 | | | 526 |
Other non-current assets | | | 98 | | | 139 |
TOTAL NON-CURRENT ASSETS | | | 3,132 | | | 2,235 |
TOTAL ASSETS | | | 43,581 | | | 11,871 |
| | | | | |||
Liabilities and Shareholders' Equity (Capital Deficiency) | | | | | ||
CURRENT LIABILITIES: | | | | | ||
Accounts payable | | | 406 | | | 475 |
Accrued expenses and other liabilities | | | 1,690 | | | 1,828 |
Related party accrued liabilities | | | 192 | | | 72 |
Related party liability | | | — | | | 17,748 |
Current maturities of operating leases | | | 315 | | | 140 |
TOTAL CURRENT LIABILITIES | | | 2,603 | | | 20,263 |
| | | | | |||
NON-CURRENT LIABILITIES: | | | | | ||
Non-current operating leases | | | 619 | | | 386 |
TOTAL NON-CURRENT LIABILITIES | | | 619 | | | 386 |
TOTAL LIABILITIES | | | 3,222 | | | 20,649 |
| | | | | |||
COMMITMENTS SHAREHOLDERS' EQUITY (CAPITAL DEFICIENCY): | | | | | ||
Ordinary Shares, par value NIS 0.01 per share, 40,000,000 authorized at June 30, 2020 and December 2019, respectively; 30,679,965 and 27,150,080 issued and outstanding at June 30, 2020 and December 31, 2019, respectively | | | 85 | | | 75 |
Additional paid-in capital | | | 94,661 | | | 31,748 |
Accumulated deficit | | | (54,387) | | | (40,601) |
TOTAL SHAREHOLDERS’ EQUITY (CAPITAL DEFICIENCY) | | | 40,359 | | | (8,778) |
TOTAL LIABILITIES AND SHAREHOLDERS' EQUITY (CAPITAL DEFICIENCY) | | | 43,581 | | | 11,871 |
| | | Six Months Ended June 30, | | | Three Months Ended June 30, | |||||||
| | | 2020 | | | 2019 | | | 2020 | | | 2019 | |
| | | (U.S. dollars in thousands, except for per share data) | ||||||||||
OPERATING EXPENSES: | | | | | | | | | ||||
Research and development | | | 4,152 | | | 340 | | | 1,786 | | | 172 |
Marketing | | | 1,745 | | | 242 | | | 772 | | | 122 |
General and administrative | | | 7,903 | | | 1,079 | | | 3,871 | | | 659 |
OPERATING LOSS | | | (13,800) | | | (1,661) | | | (6,429) | | | (953) |
FINANCIAL EXPENSES (INCOME), NET | | | (14) | | | 14 | | | (65) | | | 11 |
NET LOSS | | | (13,786) | | | (1,675) | | | (6,364) | | | (964) |
| | | | | | | | | |||||
BASIC AND DILUTED LOSS PER SHARE | | | (0.47) | | | (0.07) | | | (0.21) | | | (0.04) |
WEIGHTED AVERAGE NUMBER OF | | | | | | | | | ||||
ORDINARY SHARES (in thousands) | | | 29,273 | | | 23,452 | | | 29,628 | | | 24,456 |
| | | Ordinary shares | | | Additional paid-in capital | | | Accumulated deficit | | | Total | ||||
| | | Number of shares | | | Amount | | |||||||||
| | | | | | | U.S. Dollars in thousands | |||||||||
BALANCE AT JANUARY 1, 2020 | | | 27,150,080 | | | 75 | | | 31,748 | | | (40,601) | | | (8,778) |
CHANGES DURING SIX MONTHS ENDED JUNE 30, 2020 | | | | | | | | | | | |||||
Issuance of ordinary shares, net of issuance costs | | | 2,368,250 | | | 10 | | | 36,690 | | | | | 36,700 | |
Conversion of related party liability to shareholders' equity | | | 1,109,245 | | | | | 17,748 | | | | | 17,748 | ||
Issuance of ordinary shares to employees and non-employees upon exercise of warrants | | | 29,766 | | | * | | | 66 | | | | | 66 | |
Issuance of ordinary shares to investors upon exercise of warrants | | | 22,624 | | | * | | | 62 | | | | | 62 | |
Share-based compensation | | | | | | | 8,347 | | | | | 8,347 | |||
Net loss | | | | | | | | | (13,786) | | | (13,786) | |||
BALANCE AT JUNE 30, 2020 | | | 30,679,965 | | | 85 | | | 94,661 | | | (54,387) | | | 40,359 |
| | | Ordinary shares | | | Additional paid-in capital | | | Accumulated deficit | | | Total | ||||
| | | Number of shares | | | Amount | | |||||||||
| | | | | | | U.S. Dollars in thousands | |||||||||
BALANCE AT JANUARY 1, 2019 | | | 21,924,208 | | | 58 | | | 11,596 | | | (18,038) | | | (6,384) |
CHANGES DURING SIX MONTHS ENDED JUNE 30, 2019 | | | | | | | | | | | |||||
Issuance of ordinary shares and warrants, net of issuance costs | | | 4,450,146 | | | 13 | | | 9,251 | | | | | 9,264 | |
Additional consideration for an asset purchase agreement | | | | | | | 174 | | | | | 174 | |||
Net loss | | | | | | | | | (1,675) | | | (1,675) | |||
BALANCE AT JUNE 30, 2019 | | | 26,374,354 | | | 71 | | | 21,021 | | | (19,713) | | | 1,379 |
(*) | Less than 1 thousand US dollars. |
| | | Ordinary shares | | | Additional paid-in capital | | | Accumulated deficit | | | Total | ||||
| | | Number of shares | | | Amount | | |||||||||
| | | | | | | U.S. Dollars in thousands | |||||||||
BALANCE AT APRIL 1, 2020 | | | 29,261,215 | | | 81 | | | 68,912 | | | (48,023) | | | 20,970 |
CHANGES DURING THREE MONTHS ENDED JUNE 30, 2020 | | | | | | | | | | | |||||
Issuance of ordinary shares, net of issuance costs | | | 1,418,750 | | | 4 | | | 22,252 | | | 22,256 | | | |
Share-based compensation | | | | | | | 3,497 | | | | | 3,497 | |||
Net loss | | | | | | | | | (6,364) | | | (6,364) | |||
BALANCE AT JUNE 30, 2020 | | | 30,679,965 | | | 85 | | | 94,661 | | | (54,387) | | | 40,359 |
| | | Ordinary shares | | | Additional paid-in capital | | | Accumulated deficit | | | Total | ||||
| | | Number of shares | | | Amount | | |||||||||
| | | | | | | U.S. Dollars in thousands | |||||||||
BALANCE AT APRIL 1, 2019 | | | 22,250,688 | | | 59 | | | 11,908 | | | (18,749) | | | (6,782) |
CHANGES DURING THREE MONTHS ENDED JUNE 30, 2019 | | | | | | | | | | | |||||
Issuance of ordinary shares and warrants, net of issuance costs | | | 4,123,666 | | | 12 | | | 9,103 | | | | | 9,115 | |
Additional consideration for an asset purchase agreement | | | | | | | 10 | | | | | 10 | |||
Net loss | | | | | (964) | | | (964) | | | | | |||
BALANCE AT JUNE 30, 2019 | | | 26,374,354 | | | 71 | | | 21,021 | | | (19,713) | | | 1,379 |
| | | Six Months Ended | ||||
| | | June 30, | ||||
| | | 2020 | | | 2019 | |
| | | U.S. Dollars in thousands | ||||
CASH FLOWS FROM OPERATING ACTIVITIES: | | | | | ||
Net loss | | | (13,786) | | | (1,675) |
Adjustments required to reconcile net loss to net cash used in operating activities: | | | | | ||
Share-based compensation | | | 8,347 | | | — |
Depreciation | | | 33 | | | 24 |
Changes in operating assets and liabilities: | | | | | ||
Prepaid expenses and other current assets | | | 782 | | | — |
Related party prepaid expenses | | | (35) | | | 654 |
Other non-current assets | | | 41 | | | — |
Accounts payable | | | (69) | | | (63) |
Operating leases | | | * | | | — |
Accrued expenses and other liabilities | | | (51) | | | — |
Net cash used in operating activities | | | (4,738) | | | (1,060) |
| | | | | |||
CASH FLOWS FROM INVESTING ACTIVITIES: | | | | | ||
Purchase of property and equipment | | | (244) | | | (80) |
Net cash used in investing activities | | | (244) | | | (80) |
| | | | | |||
CASH FLOWS FROM FINANCING ACTIVITIES: | | | | | ||
Proceeds from issuance of ordinary shares and warrants, net of issuance costs | | | 37,237 | | | 9,264 |
Proceeds from issuance of ordinary shares upon exercise of warrants | | | 128 | | | — |
Deferred offering costs | | | (884) | | | — |
Net cash provided by financing activities | | | 36,481 | | | 9,264 |
| | | | | |||
NET CHANGE IN CASH AND CASH EQUIVALENTS AND RESTRICTED CASH | | | 31,499 | | | 8,124 |
CASH AND CASH EQUIVALENTS AND RESTRICTED CASH AT BEGINNING OF THE PERIOD | | | 8,217 | | | 5 |
CASH AND CASH EQUIVALENTS AND RESTRICTED CASH AT END OF THE PERIOD | | | 39,716 | | | 8,129 |
SUPPLEMENTARY INFORMATION ON ACTIVITIES NOT INVOLVING CASH FLOWS: | | | | | ||
Unpaid offering costs | | | 138 | | | — |
Conversion of related party liability to shareholders' equity | | | 17,748 | | | — |
Additional consideration for an asset purchase agreement | | | — | | | 174 |
Operating lease liabilities arising from obtaining operating right-of-use assets | | | 486 | | | — |
Issuance costs | | | 537 | | | — |
(*) | Less than 1 thousand US dollars. |
a. | Nano-X Imaging Ltd., an Israeli Company (hereinafter “the Company” or “Nanox IL” or “the Successor Company”), was incorporated on December 20, 2018 and commenced its operations on September 3, 2019. |
b. | The Company's solution, referred to as the Nanox System, has two integrated components – “Nanox.ARC” and “Nanox.CLOUD”. Nanox.ARC is a medical imaging system incorporating the Company’s novel digital X-ray source. Nanox.CLOUD is a cloud-based system designed to provide end-to-end medical imaging services, including services such as image repository, radiologist matching, online and offline diagnostics review and annotation, connectivity to diagnostic assistive AI systems, billing and reporting. |
c. | In order to complete its technology development program, the Company will require significant funding. Moreover, the Company has experienced net losses and negative cash flows from operations since its inception and has relied on its ability to fund its operations primarily through equity financings. As of June 30, 2020, the Company had an accumulated deficit and negative cash flows from operations. The Company anticipates such losses will continue until its product candidates reach commercial profitability. |
d. | Current Impact of COVID-19 |
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
| | | Six months ended June 30, | | | Six months ended June 30, | |||||||
| | | 2020 | | | 2019 | |||||||
| | | Number of share-based payment Awards | | | Weighted average exercise price | | | Number of share-based payment awards | | | Weighted average exercise price | |
Outstanding, at beginning of period | | | 3,410,406 | | | $1.89 | | | 1,592,874 | | | $1.32 |
Changes during the period: | | | | | | | | | ||||
Granted | | | 121,840 | | | $8.91 | | | — | | | — |
Exercised | | | (29,766) | | | $2.21 | | | — | | | — |
Forfeited | | | — | | | — | | | — | | | — |
Expired | | | — | | | — | | | — | | | — |
Cancelled | | | — | | | — | | | — | | | — |
Outstanding at end of period | | | 3,502,480 | | | $2.07 | | | 1,592,874 | | | $1.32 |
Exercisable at end of period | | | 2,168,033 | | | $2.40 | | | 1,592,874 | | | $1.32 |
| | | Six months ended June 30, 2020 | |
Fair value of one ordinary share | | | 16 |
Dividend yield | | | 0 |
Expected volatility | | | 44.40% - 57.34% |
Risk-free interest rate | | | 0.34%-1.61% |
Contractual term (years) | | | 5.00-10.00 |
June 30, 2020 Awards outstanding | | | | | | | Awards exercisable | |||||
| | | Number of Awards outstanding at end of period | | | Weighted average remaining contractual life (years) | | | Number of Award exercisable at end of Period | | | Weighted average remaining contractual life (years) | |
$0.01 | | | 186,815 | | | 0.83 | | | 186,815 | | | 0.83 |
$1.92 | | | 472,606 | | | 1.31 | | | 472,606 | | | 1.31 |
$2.21 | | | 2,759,896 | | | 5.25 | | | 1,441,420 | | | 5.25 |
$16 | | | 59,206 | | | 4.96 | | | 59,206 | | | 4.96 |
$20.87 | | | 23,957 | | | 5.19 | | | 7,986 | | | 5.19 |
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
June 30, 2019 Awards outstanding | | | | | | | Awards exercisable | |||||
| | | Number of Awards outstanding at end of period | | | Weighted average remaining contractual life (years) | | | Number of Award exercisable at end of Period | | | Weighted average remaining contractual life (years) | |
$0.01 | | | 186,815 | | | 1.83 | | | 186,815 | | | 1.83 |
$0.30 | | | 454,166 | | | 0.27 | | | 454,166 | | | 0.27 |
$1.92 | | | 472,606 | | | 2.31 | | | 472,606 | | | 2.31 |
$2.21 | | | 479,287 | | | 2.87 | | | 479,287 | | | 2.87 |
| | | Six months ended June 30, 2020 | |||||||
| | | Number of share-based payment awards | | | Weighted average exercise price | | | Weighted average remaining contractual life (years) | |
Outstanding at beginning of period | | | 1,667,267 | | | 2.21 | | | 9.90 |
Changes during the period: | | | | | | | |||
Granted | | | 295,234 | | | 2.21 | | | 9.60 |
Exercised | | | — | | | — | | | — |
Forfeited | | | — | | | — | | | — |
Expired | | | — | | | — | | | — |
Cancelled | | | (9,375) | | | 2.21 | | | — |
Outstanding at end of period | | | 1,953,126 | | | 2.21 | | | 9.55 |
Exercisable at end of period | | | 726,880 | | | 2.21 | | | 9.55 |
| | | 2020 | |
Fair value of one ordinary share | | | $16 |
Dividend yield | | | 0 |
Expected volatility | | | 45.11% - 50.65% |
Risk-free interest rate | | | 1.45% - 1.61% |
Contractual term (years) | | | 10 |
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
| | | Six Months Ended June 30, | | | Three Months Ended June 30, | |||||||
| | | 2020 | | | 2019 | | | 2020 | | | 2019 | |
| | | (U.S. dollars in thousands) | ||||||||||
Research and development | | | 1,917 | | | — | | | 240 | | | — |
Marketing | | | 644 | | | — | | | 322 | | | — |
General and administrative | | | 5,786 | | | — | | | 2,935 | | | — |
| | | 8,347 | | | — | | | 3,497 | | | — | |
a. | On June 4, 2020, the Company signed a supplemental lease agreement to expand its facilities in Israel, which expires in June 2023 (“Supplemental Lease Agreement”). The monthly rate for the Supplemental Lease Agreement is approximately $14 thousand per month. |
b. | For services with SixAI Ltd. (“SixAI”), see Note 6c. |
a. | Related parties transactions: |
| | | Six Months Ended June 30, | | | Three Months Ended June 30, | |||||||
| | | 2020 | | | 2019 | | | 2020 | | | 2019 | |
| | | (U.S. Dollars in thousands) | ||||||||||
Research and development | | | 115 | | | 99 | | | 90 | | | 50 |
General and administrative | | | — | | | 278 | | | — | | | 148 |
b. | Six-Eye Interactive agreements for services |
c. | SixAI Ltd. Service agreement |
d. | Pursuant to the Asset Purchase Agreement between Nanox IL and Nanox PLC, as more fully described in the Company's annual financial statements, as of June 30, 2020, the Company has a related party liability balance of $192 thousand. |
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
a. | Basic |
| | | Six Months Ended June 30, | | | Three Months Ended June 30, | |||||||
| | | 2020 | | | 2019 | | | 2020 | | | 2019 | |
Net loss | | | (13,786) | | | (1,675) | | | $(6,364) | | | $(964) |
Weighted average number of ordinary shares (in thousands) | | | 29,273 | | | 23,452 | | | 29,628 | | | 24,456 |
Basic and diluted loss per share | | | (0.47) | | | (0.07) | | | $(0.21) | | | $(0.04) |
b. | Diluted |
a. | In July 2020, the Company issued 625,000 ordinary shares to one of its investors, at a price per share of $16.00, for a purchase price of $10 million, received in July 2020, which reflects the second portion of the investor’s $20 million investment. |
b. | In July 2020, the Company entered into additional share purchase agreements with certain investors for the issuance of 3,500,000 ordinary shares at a price per share of $16.00, for an aggregate purchase price of approximately $56 million. As of the date of the issuance of these financial statements, the funds were not received yet. |
c. | The Company has evaluated subsequent events through July 30, 2020, the date on which the consolidated financial statements were available to be issued. |

Oppenheimer & Co.
Berenberg
CIBC Capital Markets
INFORMATION NOT REQUIRED IN PROSPECTUS
Item 6. | Exculpation, Insurance and Indemnification of Office Holders (Including Directors and Officers). |
• | a monetary liability incurred by or imposed on the office holder in favor of another person pursuant to a court judgment, including pursuant to a settlement confirmed as judgment or arbitrator’s decision approved by a competent court. However, if an undertaking to indemnify an office holder with respect to such liability is provided in advance, then such an undertaking must be limited to events which, in the opinion of the board of directors, can be foreseen based on the company’s activities when the undertaking to indemnify is given, and to an amount or according to criteria determined by the board of directors as reasonable under the circumstances, and such undertaking shall detail the abovementioned foreseen events and amount or criteria; |
• | reasonable litigation expenses, including reasonable attorneys’ fees, which were incurred by the office holder (i) as a result of an investigation or proceeding filed against the office holder by an authority authorized to conduct such investigation or proceeding, provided that such investigation or proceeding; was either (a) concluded without the filing of an indictment against such office holder and without the imposition on him of any monetary obligation in lieu of a criminal proceeding; (b) concluded without the filing of an indictment against the office holder but with the imposition of a monetary obligation on the office holder in lieu of criminal proceedings for an offense that does not require proof of criminal intent; or (ii) in connection with a monetary sanction; |
• | a monetary liability imposed on the office holder in favor of an injured party at an Administrative Procedure (as defined below) as set forth in Section 52(54)(a)(1)(a) to the Securities Law; |
• | expenses expended by the office holder with respect to an Administrative Procedure under the Securities Law, including reasonable litigation expenses and reasonable attorneys’ fees; |
• | reasonable litigation expenses, including attorneys’ fees, incurred by the office holder or which were imposed on the office holder by a court (i) in a proceeding instituted against him or her by the company, on its behalf, or by a third party, (ii) in connection with criminal indictment of which the office holder was acquitted, or (iii) in connection with a criminal indictment which the office holder was convicted of an offense that does not require proof of criminal intent; and |
• | Any other obligation or expense in respect of which it is permitted or will be permitted under applicable law to indemnify an office holder. |
• | a breach of the duty of loyalty to the company, provided that the office holder acted in good faith and had a reasonable basis to believe that the act would not harm the company; |
• | a breach of duty of care to the company or to a third party, to the extent such a breach arises out of the negligent conduct of the office holder; |
• | a monetary liability imposed on the office holder in favor of a third party; |
• | a monetary liability imposed on the office holder in favor of an injured party at an Administrative Procedure pursuant to Section 52(54)(a)(1)(a) of the Securities Law; and |
• | expenses incurred by an office holder in connection with an Administrative Procedure, including reasonable litigation expenses and reasonable attorneys’ fees. |
• | a breach of the duty of loyalty, except for indemnification and insurance for a breach of the duty of loyalty to the company to the extent that the office holder acted in good faith and had a reasonable basis to believe that the act would not prejudice the company; |
• | a breach of duty of care committed intentionally or recklessly, excluding a breach arising out of the negligent conduct of the office holder; |
• | an act or omission committed with intent to derive illegal personal benefit; or |
• | a fine or forfeit levied against the office holder. |
Item 7. | Recent Sales of Unregistered Securities. |
Item 8. | Exhibits and Financial Statement Schedules. |
(a) | Exhibits |
(b) | Financial Statement Schedules |
Item 9. | Undertakings. |
(a) | The undersigned registrant hereby undertakes to provide to the underwriters at the closing specified in the underwriting agreement certificates in such denominations and registered in such names as required by the underwriters to permit prompt delivery to each purchaser. |
(b) | Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue. |
(c) | The undersigned registrant hereby undertakes that: |
1. | For purposes of determining any liability under the Securities Act of 1933, the information omitted from the form of prospectus filed as part of this registration statement in reliance upon Rule 430A and contained in a form of prospectus filed by the registrant pursuant to Rule 424(b)(1) or (4) or 497(h) under the Securities Act shall be deemed to be part of this registration statement as of the time it was declared effective. |
2. | For the purpose of determining any liability under the Securities Act of 1933, each post-effective amendment that contains a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
Exhibit No. | | | Description |
1.1* | | | Form of Underwriting Agreement |
2.1† | | | Asset Purchase Agreement, dated September 3, 2019, by and between the Registrant and Nanox Imaging PLC |
2.2† | | | Amendment to the Asset Purchase Agreement, dated December 3, 2019, by and between the Registrant and Nanox Imaging PLC |
2.3† | | | Amendment to the Asset Purchase Agreement, dated December 31, 2019, by and between the Registrant and Nanox Imaging PLC |
3.1† | | | Articles of Association of the Registrant |
3.2* | | | Form of Amended and Restated Articles of Association of the Registrant to become effective immediately prior to the closing of the offering |
4.1* | | | Specimen share certificate |
| | | Form of warrants to purchase ordinary shares, dated September 2, 2019, in connection with the warrants originally issued to certain investors by Nanox Imaging PLC in 2016 | |
| | | Form of warrants to purchase ordinary shares, dated September 2, 2019, in connection with the warrants originally issued to certain finders by Nanox Imaging PLC in 2015 | |
| | | Form of warrants to purchase ordinary shares, dated September 2, 2019, in connection with the warrants originally issued to certain finders and employee by Nanox Imaging PLC in 2014 and 2015 | |
| | | Form of warrants to purchase ordinary shares issued to A-Labs Finance and Advisory Ltd. | |
| | | Warrant to purchase ordinary shares, dated September 2, 2019, issued to SK Telecom TMT Investment Corp. | |
| | | Amendment to Warrant to purchase ordinary shares, dated June 4, 2020, issued to SK Telecom TMT Investment Corp. | |
5.1* | | | Opinion of Amit, Pollak, Matalon & Co., counsel to the Registrant, as to the validity of the ordinary shares (including consent) |
| | | Contract Manufacturing Agreement, dated May 26, 2020, by and between the Registrant and FoxSemicon Integrated Technology, Inc. | |
10.2* | | | Registration Rights Agreement by and among the Registrant and the certain shareholders named therein |
| | | 2019 Equity Incentive Plan | |
| | | U.S. Sub-Plan | |
10.5* | | | Form of Indemnification Agreement between the Registrant and each director and executive officer |
21.1† | | | List of subsidiaries of the Registrant |
| | | Consent of PricewaterhouseCoopers International Limited, an independent registered public accounting firm | |
23.2* | | | Consent of Amit, Pollak, Matalon & Co. (included in Exhibit 5.1) |
| | | Power of Attorney (included in signature page to Registration Statement) |
* | To be filed by amendment. |
† | Previously submitted. |
| | | NANO-X IMAGING LTD | ||||
| | | | | |||
| | | By | | | /s/ Ran Poliakine | |
| | | | | Name: Ran Poliakine | ||
| | | | | Title: Chief Executive Officer | ||
Signatures | | | Title | | | Date |
| | | | | |||
/s/ Ran Poliakine | | | Director and Chief Executive Officer (Principal Executive Officer) | | | July 30, 2020 |
Ran Poliakine | | |||||
| | | | | |||
/s/ Itzhak Maayan | | | Chief Financial Officer (Principal Financial Officer and Principal Accounting Officer) | | | July 30, 2020 |
Itzhak Maayan | | |||||
| | | | | |||
/s/ Onn Fenig | | | Director | | | July 30, 2020 |
Onn Fenig | | |||||
| | | | | |||
/s/ Floyd Katske | | | Director | | | July 30, 2020 |
Floyd Katske | | |||||
| | | | | |||
/s/ Erez Meltzer | | | Director | | | July 30, 2020 |
Erez Meltzer | | |||||
| | | | | |||
/s/ Richard Stone | | | Director | | | July 30, 2020 |
Richard Stone | | |||||
| | | | |
| | | By: | | | AUTHORIZED REPRESENTATIVE | ||||
| | | | | | | ||||
| | | By: | | | /s/ Richard Stone | ||||
| | | | | Name: | | | Richard Stone | ||
| | | | | Title: | | | Director | ||


