EMERALD HEALTH THERAPEUTICS, INC.
Condensed Interim Consolidated Financial Statements
(Unaudited)
For the three and nine months ended September 30, 2020 and 2019
(In Canadian Dollars)
Table of Contents
| Condensed Interim Consolidated Statements of Financial Position | 2 |
| Condensed Interim Consolidated Statements of Loss and Comprehensive Loss | 3 |
| Condensed Interim Consolidated Statements of Changes in Equity | 4 |
| Condensed Interim Consolidated Statements of Cash Flows | 5 |
Notes to the Condensed Interim Consolidated Financial Statements
| Note 1 | Nature and Continuance of Operations | 6 | Note 11 | Convertible Debenture | 20 | |
| Note 2 | Significant Accounting Policies and Judgements | 6 | Note 12 | Share Capital | 20 | |
| Note 3 | Accounts Receivable | 9 | Note 13 | Share-based Compensation | 22 | |
| Note 4 | Biological Assets | 9 | Note 14 | Warrants | 24 | |
| Note 5 | Inventory | 11 | Note 15 | Leases | 25 | |
| Note 6 | Property, Plant and Equipment | 12 | Note 16 | Revenue | 26 | |
| Note 7 | Acquisitions | 13 | Note 17 | General and Administrative Expenses | 26 | |
| Note 8 | Intangible Assets | 14 | Note 18 | Segmented Information | 26 | |
| Note 9 | Investment in Joint Venture | 15 | Note 19 | Financial Instruments | 28 | |
| Note 10 | Related Party Transactions | 18 | Note 20 | Capital Management | 29 | |
| Note 21 | Subsequent Events | 30 |
| EMERALD HEALTH THERAPEUTICS, INC. |
| CONDENSED INTERIM CONSOLIDATED STATEMENTS OF FINANCIAL POSITION |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
| September 30 | December 31 | |||||
| 2020 | 2019 | |||||
| ASSETS | ||||||
| Current | ||||||
|
Cash and cash equivalents (Note 2 (f)) |
$ | 418 | $ | 2,525 | ||
|
Accounts receivable (Note 3) |
1,557 | 1,925 | ||||
|
Biological assets (Note 4) |
1,571 | 4,159 | ||||
|
Inventory (Note 5) |
7,765 | 6,588 | ||||
|
Prepaid expenses |
219 | 327 | ||||
|
Investment in joint venture - held for sale (Note 9) |
68,250 | - | ||||
|
Due from related parties (Note 10) |
207 | 201 | ||||
| Total current assets | 79,987 | 15,725 | ||||
|
Plant and equipment (Note 6) |
36,649 | 41,400 | ||||
|
Plant under construction (Note 6) |
8,795 | 8,407 | ||||
|
Deposits on materials and equipment |
531 | 886 | ||||
|
Refundable deposits |
196 | 196 | ||||
|
Intangible assets (Note 8) |
3,921 | 19,526 | ||||
|
Right-of-use assets (Note 15) |
3,824 | 5,628 | ||||
|
Goodwill |
169 | 169 | ||||
|
Long-term investment |
473 | 81 | ||||
|
Investment in joint venture (Note 9) |
- | 64,603 | ||||
| Total non-current assets | 54,558 | 140,896 | ||||
| TOTAL ASSETS | $ | 134,545 | $ | 156,621 | ||
| LIABILITIES | ||||||
| Current liabilities | ||||||
|
Accounts payable and accrued liabilities |
$ | 14,401 | $ | 10,941 | ||
|
Deferred payment (Note 7(b)(i)) |
9,000 | 7,818 | ||||
|
Payable to joint venture (Note 9) |
- | 710 | ||||
|
Due to related parties (Note 10) |
3,541 | 6,218 | ||||
| Total current liabilities | 26,942 | 25,687 | ||||
Lease liability (Note 15) |
4,198 | 5,944 | ||||
CEBA loan (Note 2(b)) |
40 | - | ||||
Convertible debenture (Note 11) |
23,197 | 21,823 | ||||
| TOTAL LIABILITIES | $ | 54,377 | $ | 53,454 | ||
| SHAREHOLDERS' EQUITY | ||||||
|
Share capital (Note 12) |
248,601 | 237,151 | ||||
|
Warrants (Note 14) |
1,718 | 2,449 | ||||
|
Convertible debt reserves (Note 11) |
383 | 383 | ||||
|
Contributed surplus |
29,908 | 28,146 | ||||
|
Accumulated deficit |
(199,283 | ) | (164,196 | ) | ||
| TOTAL SHAREHOLDERS' EQUITY | 81,327 | 103,933 | ||||
|
Non-controlling interest |
(1,159 | ) | (766 | ) | ||
| TOTAL LIABILITIES AND EQUITY | $ | 134,545 | $ | 156,621 |
Nature and continuance of operations (Note 1)
Events after the reporting period (Note 21)
On behalf of the Board of Directors:
| /s/ Punit Dhillon | /s/ Jim Heppell |
| Director | Director |
The accompanying notes form an integral part of these condensed interim consolidated financial statements
2
| EMERALD HEALTH THERAPEUTICS, INC. |
| CONDENSED INTERIM CONSOLIDATED STATEMENTS OF LOSS AND COMPREHENSIVE LOSS |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
| Three months | Three months | Nine months | Nine months | |||||||||
| ended | ended | ended | ended | |||||||||
| September 30 | September 30 | September 30 | September 30 | |||||||||
| 2020 | 2019 | 2020 | 2019 | |||||||||
| Revenue | ||||||||||||
|
Sales (Note 16) |
$ | 4,311 | $ | 9,718 | $ | 10,750 | $ | 17,398 | ||||
|
Excise taxes |
938 | 420 | 2,007 | 1,351 | ||||||||
| Net revenue | 3,373 | 9,298 | 8,743 | 16,047 | ||||||||
| Cost of sales | ||||||||||||
|
Cost of goods sold |
2,498 | 8,264 | 6,384 | 15,683 | ||||||||
|
Production costs |
2,177 | 1,412 | 3,596 | 2,869 | ||||||||
|
Amortization of Health Canada license (Note 8) |
29 | 920 | 458 | 2,760 | ||||||||
|
Inventory write-down (Note 5) |
1,819 | 2,824 | 2,874 | 2,824 | ||||||||
|
Loss (gain) on changes in fair value of biological assets (Note 4) |
2,339 | 433 | 825 | (545 | ) | |||||||
| Gross margin | (5,489 | ) | (4,555 | ) | (5,394 | ) | (7,544 | ) | ||||
| Expenses | ||||||||||||
|
General and administrative (Note 17) |
2,732 | 4,338 | 7,410 | 11,821 | ||||||||
|
Sales and marketing |
287 | 1,201 | 1,222 | 3,252 | ||||||||
|
Research and development |
267 | 1,179 | 975 | 3,413 | ||||||||
|
Depreciation and amortization (Note 6, 8 and 15) |
349 | 429 | 1,420 | 925 | ||||||||
|
Share-based payments (Note 13) |
755 | 2,993 | 2,585 | 11,437 | ||||||||
| 4,390 | 10,140 | 13,612 | 30,848 | |||||||||
| Loss from operations | 9,879 | 14,695 | 19,006 | 38,392 | ||||||||
|
Share of loss (income) from joint venture (Note 9) |
520 | 1,202 | (4,497 | ) | (19,099 | ) | ||||||
|
Interest and other income |
(29 | ) | (221 | ) | (851 | ) | (1,024 | ) | ||||
|
Finance costs and other expenses (Note 7(b)(i) and 11) |
1,531 | 2,489 | 3,954 | 3,030 | ||||||||
|
Impairment of assets (Note 6 and 8) |
112 | - | 17,175 | - | ||||||||
|
Loss on disposal of equipment |
32 | - | 235 | - | ||||||||
|
Loss on settlement of deferred payment |
- | - | - | 864 | ||||||||
|
Loss on dilution of joint venture ownership (Note 9) |
- | - | 850 | - | ||||||||
|
Fair value changes in financial assets (Note 19) |
(387 | ) | 101 | (392 | ) | 204 | ||||||
| Loss before income taxes | 11,658 | 18,266 | 35,480 | 22,367 | ||||||||
| Deferred income tax recovery | - | (805 | ) | - | (805 | ) | ||||||
| NET LOSS AND COMPREHENSIVE LOSS | 11,658 | 17,461 | 35,480 | 21,562 | ||||||||
| Net loss and comprehensive loss attributable to: | ||||||||||||
|
Emerald Health Therapeutics, Inc. |
11,491 | 17,253 | 35,087 | 20,971 | ||||||||
|
Non-controlling interest (Note 7(a)(i)) |
167 | 208 | 393 | 591 | ||||||||
| 11,658 | 17,461 | 35,480 | 21,562 | |||||||||
| Net loss per common share | ||||||||||||
|
Basic and diluted |
0.06 | 0.12 | 0.19 | 0.15 | ||||||||
| Weighted average number of common shares outstanding | ||||||||||||
|
Basic and diluted |
200,336,735 | 150,623,580 | 188,925,204 | 146,209,650 |
The accompanying notes form an integral part of these condensed interim consolidated financial statements
3
| EMERALD HEALTH THERAPEUTICS, INC. |
| CONDENSED INTERIM CONSOLIDATED STATEMENTS OF CHANGES IN EQUITY |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
| Share Capital | Warrants | ||||||||||||||||||||||||||
| Total | Non- | ||||||||||||||||||||||||||
| # of | # of | Contributed | Convertible | Accumulated | Shareholders' | Controlling | Total | ||||||||||||||||||||
| Shares | Amount | Warrants | Amount | Surplus | Debt Reserves | Deficit | Equity | Interest | Equity | ||||||||||||||||||
| Balance, January 1, 2020 | 160,986,373 | $ | 237,151 | 26,470,671 | $ | 2,449 | $ | 28,146 | $ | 383 | $ | (164,196 | ) | $ | 103,933 | $ | (766 | ) | $ | 103,167 | |||||||
| Shares issued on prospectus offering (Note 12) | 21,696,178 | 4,935 | - | - | - | - | - | 4,935 | - | 4,935 | |||||||||||||||||
| Shares issued on at-the-market offering (Note 12) | 1,312,500 | 287 | - | - | - | - | - | 287 | - | 287 | |||||||||||||||||
| Share issued on settlement of related party transaction (Note 10) | 9,713,666 | 2,914 | - | - | (97 | ) | - | - | 2,817 | - | 2,817 | ||||||||||||||||
| Share issued on settlement of convertible debt interest | 4,894,055 | 1,009 | - | - | - | - | - | 1,009 | - | 1,009 | |||||||||||||||||
| Warrants issued on prospectus offering (Note 12 and 14) | - | - | 21,696,178 | 164 | - | - | - | 164 | - | 164 | |||||||||||||||||
| Shares issued on restricted share unit vesting | 175,000 | 726 | - | - | (726 | ) | - | - | - | - | - | ||||||||||||||||
| Shares issued on warrant exercises (Note 12 and 14) | 6,250,000 | 1,958 | (6,250,000 | ) | (895 | ) | - | - | - | 1,063 | - | 1,063 | |||||||||||||||
| Share issuance costs | - | (379 | ) | - | - | - | - | - | (379 | ) | - | (379 | ) | ||||||||||||||
| Share-based payments (Note 13) | - | - | - | - | 2,585 | - | - | 2,585 | - | 2,585 | |||||||||||||||||
| Net loss and comprehensive loss | - | - | - | - | - | - | (35,087 | ) | (35,087 | ) | (393 | ) | (35,480 | ) | |||||||||||||
| Balance, September 30, 2020 | 205,027,772 | $ | 248,601 | $ | 41,916,849 | $ | 1,718 | $ | 29,908 | $ | 383 | $ | (199,283 | ) | $ | 81,327 | $ | (1,159 | ) | $ | 80,168 | ||||||
| Balance, January 1, 2019 | 141,443,116 | $ | 204,792 | 8,411,764 | $ | 4,360 | $ | 14,202 | $ | - | $ | (52,856 | ) | $ | 170,498 | $ | - | $ | 170,498 | ||||||||
| Adoption of IFRS 16 | - | - | - | - | - | - | (199 | ) | (199 | ) | - | (199 | ) | ||||||||||||||
| Balance, January 1, 2019, as restated | 141,443,116 | 204,792 | 8,411,764 | 4,360 | 14,203 | - | (53,055 | ) | 170,299 | - | 170,299 | ||||||||||||||||
| Shares issued on at-the-market offering (Note 12) | 5,936,500 | 18,768 | - | - | - | - | - | 18,768 | - | 18,768 | |||||||||||||||||
| Warrants issued on convertible debentures | - | - | 12,500,000 | 1,791 | - | - | - | 1,791 | - | 1,791 | |||||||||||||||||
| Convertible debt reserves | - | - | - | - | - | 383 | - | 383 | - | 383 | |||||||||||||||||
| Acquisition of Verdelite Sciences Inc. | 2,129,707 | 8,199 | - | - | - | - | - | 8,199 | - | 8,199 | |||||||||||||||||
| Acquisition of Emerald Health Naturals Inc. | - | - | - | - | - | - | - | - | 4,802 | 4,802 | |||||||||||||||||
| Shares issued on stock option exercises (Note 13) | 1,898,143 | 3,339 | - | - | (2,195 | ) | - | - | 1,144 | - | 1,144 | ||||||||||||||||
| Share issuance costs | - | (877 | ) | - | - | - | - | - | (877 | ) | - | (877 | ) | ||||||||||||||
| Share-based payments (Note 13) | - | - | - | - | 11,437 | - | - | 11,437 | - | 11,437 | |||||||||||||||||
| Net loss and comprehensive loss | - | - | - | - | - | - | (20,971 | ) | (20,971 | ) | (591 | ) | (21,562 | ) | |||||||||||||
| Balance, September 30, 2019 | 151,407,466 | $ | 234,222 | 20,911,764 | $ | 6,151 | $ | 23,444 | $ | 383 | $ | (74,027 | ) | $ | 190,173 | $ | 4,211 | $ | 194,384 | ||||||||
The accompanying notes form an integral part of these condensed interim consolidated financial statements
4
| EMERALD HEALTH THERAPEUTICS, INC. |
| CONDENSED INTERIM CONSOLIDATED STATEMENTS OF CASH FLOWS |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
| Nine months | Nine months | |||||
| ended | ended | |||||
| September 30 | September 30 | |||||
| 2020 | 2019 | |||||
| Operating activities | ||||||
|
Net loss |
$ | (35,480 | ) | $ | (21,562 | ) |
|
Items not involving cash |
||||||
|
Depreciation |
3,851 | 4,253 | ||||
|
Loss (gain) on changes in fair value of biological assets |
825 | (545 | ) | |||
|
Fair value changes in financial assets |
(392 | ) | 204 | |||
|
Inventory write-down |
2,874 | 2,824 | ||||
|
Share-based payments |
2,585 | 11,437 | ||||
|
Share of income from joint venture |
(4,497 | ) | (19,099 | ) | ||
|
Interest and accretion expense |
2,556 | 577 | ||||
|
Deferred income tax recovery |
- | (805 | ) | |||
|
Impairment of assets |
17,176 | - | ||||
|
Loss on disposal of asset |
218 | - | ||||
|
Loss on dilution of joint venture ownership |
850 | - | ||||
|
Loss on settlement of deferred payment |
- | 864 | ||||
|
Changes in non-cash operating working capital |
||||||
|
Accounts receivable |
368 | (2,548 | ) | |||
|
Due from related parties |
(6 | ) | 867 | |||
|
Prepaid expenses |
108 | 128 | ||||
|
Inventory and biological assets |
(1,905 | ) | (13,234 | ) | ||
|
Accounts payable and accrued liabilities |
5,517 | 3,767 | ||||
|
Due to related parties |
139 | 8,018 | ||||
|
Net cash flows used in operating activities |
(5,213 | ) | (24,854 | ) | ||
| Investing activities | ||||||
|
Investment in joint venture (Note 9) |
(710 | ) | (15,280 | ) | ||
|
Acquisition of asset (Note 7) |
(519 | ) | (1,100 | ) | ||
|
Deposits on material and equipment |
(93 | ) | 1,410 | |||
|
Sale of plant and equipment |
43 | - | ||||
|
Purchase of plant and equipment |
(782 | ) | (20,369 | ) | ||
|
Purchase of intangible assets |
(313 | ) | (1,562 | ) | ||
|
Net cash flows used in investing activities |
(2,374 | ) | (36,901 | ) | ||
| Financing activities | ||||||
|
Payment of lease liabilities |
(363 | ) | (362 | ) | ||
|
Repayment on long-term debt |
- | (2,503 | ) | |||
|
Repayment of deferred payment |
- | (8,000 | ) | |||
|
Proceeds from prospectus offering |
5,100 | 18,768 | ||||
|
Proceeds from at-the-market offering |
287 | - | ||||
|
Proceeds from convertible debenture financing |
- | 24,217 | ||||
|
Proceeds from CEBA loan (Note 2(b)) |
40 | - | ||||
|
Share issuance costs |
(380 | ) | (877 | ) | ||
|
Purchase warrants exercises |
1,063 | - | ||||
|
Stock option exercises |
- | 1,144 | ||||
|
Interest paid |
(267 | ) | (285 | ) | ||
|
Net cash flows provided by financing activities |
5,480 | 32,102 | ||||
| Decrease in cash and cash equivalents | (2,107 | ) | (29,653 | ) | ||
| Cash and cash equivalents, beginning of year | 2,525 | 36,042 | ||||
| Cash and cash equivalents, end of period | $ | 418 | $ | 6,389 | ||
| Supplemental Information: | ||||||
| Fair value of shares issued to settle outstanding loan amount and trade payables (Note 10) | $ | 2,914 | $ | - | ||
| Fair value of shares issued to settle interest on convertible debentures (Note 11) | $ | 1,009 | $ | - | ||
| Fair value of shares issued to settle portion of deferred payment | $ | - | $ | 8,199 |
The accompanying notes form an integral part of these condensed interim consolidated financial statements
5
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
| 1. | Nature and Continuance of Operations |
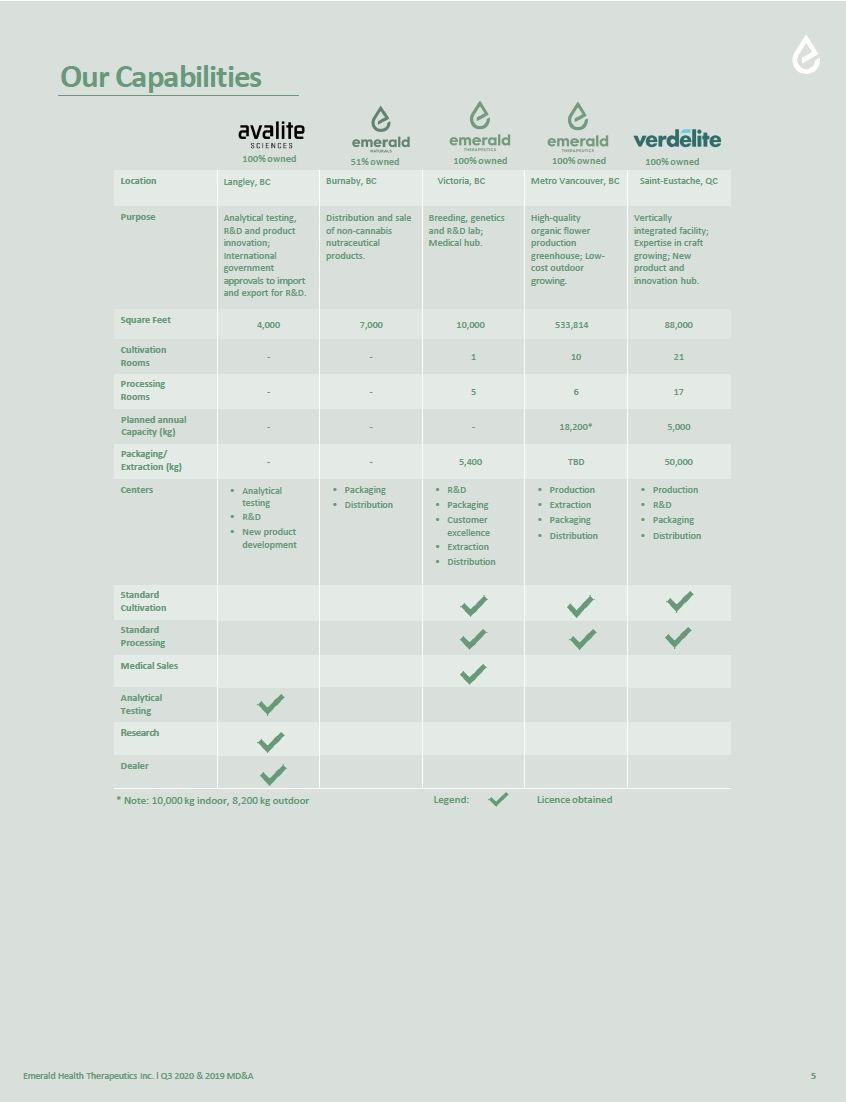
Emerald Health Therapeutics, Inc. (the "Company"), was incorporated pursuant to the Business Corporations Act (British Columbia) on July 31, 2007. The Company is classified as a Tier 1 Venture Issuer on the TSX Venture Exchange (the "TSXV”), with its common shares listed under the trading symbol “EMH.” The Company is also traded on the OTCQX, with its common shares listed under the trading symbol “EMHTF.”
The Company’s registered and records office is at Suite 2500 – 666 Burrard Street, Vancouver, British Columbia, V6C 2X8.
The Company’s principal business is the production, distribution, and sale of cannabis products in Canada, pursuant to the Cannabis Act (Canada) (the “Cannabis Act”).
These condensed interim consolidated financial statements have been prepared by management on a going concern basis which assumes that the Company will be able to realize its assets and discharge its liabilities in the normal course of business for the foreseeable future. As at September 30, 2020, the Company had not yet achieved profitable operations, had a loss for the nine months ended September 30, 2020 of $35,480 and accumulated losses since inception of $199,283. As at September 30, 2020, the Company had $418 in cash and cash equivalents. Subsequent to the quarter ended September 30, 2020, the Company completed the sale of its 41.28% interest in Pure Sunfarms Corp. for consideration of $79,900 on November 2, 2020. On the date of closing, the Company received a cash payment of $60,000 and received a secured promissory note in the principal amount of $19,900 (the “Note”). The Note will mature six months from the date of closing and bears interest of 12% per annum. This cash injection into the Company will help the continuing operations. The receipt of the $60,000 first payment provides the Company with positive working capital and cash on hand sufficient to fund planned operations.
| 2. | Significant Accounting Policies and Judgements |
| a) | Basis of Presentation and Measurement |
The condensed interim consolidated financial statements of the Company have been prepared in accordance with International Accounting Standard 34 - Interim Financial Reporting as issued by the International Accounting Standards Board (“IASB”) using accounting policies consistent with International Financial Reporting Standards (“IFRS”) as issued by the IASB. Unless otherwise noted, all amounts are presented in thousands of Canadian dollars, except share and per share data. These condensed interim financial statements were authorized for issue by the Audit Committee on November 25, 2020.
These condensed interim consolidated financial statements do not include all disclosures normally provided in annual financial statements and should be read in conjunction with the Company’s audited financial statements for the year ended December 31, 2019. Accordingly, accounting policies, estimates, and judgements applied are the same as those applied in the Company’s financial statements for the year ended December 31, 2019, unless otherwise indicated. The Company assesses its accounting estimates and judgements every reporting period. During the period, the Company applied an accounting policy for government grants in regard to the Canadian Emergency Wage Subsidy (“CEWS”) and the Canadian Emergency Business Account (“CEBA”).
6
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
| b) | Government Grants |
Government grants are recognized where there is reasonable assurance that the grant will be received, and all attached conditions will be complied with. When the grant relates to an expense item, it is recognized as income over the period necessary to match the grant on a systematic basis to the costs that it is intended to compensate. The CEWS and the CEBA are recognized as government grants. The Company applied for and received the CEWS, which provides a 75% wage subsidy effective March 15, 2020. During the nine months ended September 30, 2020, the Company determined that it had qualified for this subsidy, it applied for and received $787. The Company has recognized the $787 as income over the period necessary to match the grant on a systematic basis to the costs that it is intended to compensate.
On May 26, 2020, the Company obtained $40 in revolving credit from the Government of Canada under the CEBA COVID-19 Economic Response Plan. The funding is granted in the form of an interest-free revolving credit line of which up to $40 may be drawn. On January 1, 2021, any balance remaining on the revolving credit line will automatically convert to a non-revolving term loan. Effective January 1, 2023, any outstanding balance on the term loan shall bear interest at a rate of 5% per annum. The term loan matures on December 31, 2025. If 75% of the outstanding balance of the non-revolving term loan is repaid on or before December 31, 2022, the remaining 25% of the balance shall be forgiven.
| c) | Change in Estimates – Biological Assets |
During the three months ended September 30, 2020, the Company revised its estimates used in determining the average selling price per gram. One of the inputs used in determining the biological assets fair value less cost to sell was applied prospectively. Previously, the Company used the average retail price per gram of dried cannabis net of costs to sell for the period for all strains of cannabis sold at the finished good stage. The Company has revised its model to use the average wholesale market price per gram of dried bulk cannabis, based on quality (Tetrahydrocannabinol % and terpene profile). See Note 4.
| d) | COVID-19 Estimation Uncertainty |
During the three and nine months ended September 30, 2020, the global financial markets have been negatively impacted by the novel Coronavirus (“COVID-19”), which was declared a pandemic by the World Health Organization on March 11, 2020. This has led to significant global economic uncertainty, and the current outbreak of COVID-19 could have a material adverse effect on the Company’s operations and the operations of the Company’s suppliers and customers. At this time, the Company has not had any stoppages related to the production and sales of its cannabis, however, this could change based on future developments. The Company has taken what it believes to be appropriate safety precautions at its facilities to safeguard the health of its employees including remote work plans and additional protective measures on site, and there have been no outbreaks to date at any of the Company's facilities. The extent to which COVID-19 impacts the Company’s operations will depend on future developments, which continue to be highly uncertain and cannot be predicted with confidence.
In addition, it is possible that estimates in the Company’s financial statements will change in the near term as a result of COVID-19 and the effect of any such changes could be material, which could result in, among other things, impairment of long-lived assets including intangibles. The Company is closely monitoring the impact of the pandemic on all aspects of its business.
7
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
| e) | Basis of Consolidation |
These condensed interim consolidated financial statements include the accounts of the Company and its subsidiaries. All intercompany transactions and balances are eliminated on consolidation. Subsidiaries are all entities over which the Company has control. The Company controls an entity when the Company is exposed to, or has rights to, variable returns from its involvement with the entity and has the ability to affect those returns through its power over the entity.
The subsidiaries of the Company at September 30, 2020 include the following:
|
Ownership Interest |
Ownership Interest | ||
|
as at September 30 |
as at December 31 | ||
| Name of Entity |
2020 |
2019 | |
| Emerald Health Therapeutics Canada Inc. (EHTC) |
100% |
100% | |
| Emerald Health Naturals Inc. (Naturals) |
51% |
51% | |
| Avalite Sciences Inc. (Avalite) |
100% |
100% | |
| Pure Sunfarms Corp. (Pure Sunfarms) (Note 21) |
41.3% |
46.5% | |
| Verdélite Sciences Inc. (Verdélite) |
100% |
100% | |
| Verdélite Property Holdings Inc. |
100% |
100% |
| f) | Cash and Cash Equivalents |
Cash and cash equivalents are financial assets and the carrying amounts approximate fair value. Cash and cash equivalents include cash and redeemable short-term investment certificates held at major financial institutions as follows:
|
September 30 |
December 31 | |||
|
Interest Rate % |
2020 |
2019 | ||
|
$ |
$ | |||
| GIC - Maturing February 5, 2020 |
1.50% |
- |
200 | |
| GIC - Maturing September 11, 2020 |
2.25% |
- |
30 | |
| GIC - Maturing February 3, 2021 |
1.50% |
102 |
- | |
| GIC - Maturing March 11, 2022 | 0.50% |
31 |
- | |
| Total |
133 |
230 |
8
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
| 3. | Accounts Receivable |
The Company’s accounts receivable are comprised of:
| September 30 | December 31 | ||||
| 2020 | 2019 | ||||
| $ | $ | ||||
| Goods and services tax refund receivable | - | 1,181 | |||
| Trade receivables | 1,534 | 697 | |||
| Other receivables | 23 | 47 | |||
| 1,557 | 1,925 |
Credit risk is generally limited for receivables from government bodies, which generally have low default risk, and medical sales direct to patients, where payment is required prior to the delivery of goods. Credit risk for non-government wholesale customers is assessed on a case-by-case basis and a provision is recorded where required. As at September 30, 2020, 69% of the Company’s trade receivables are from two provincial government bodies.
| 4. | Biological Assets |
The Company’s biological assets consist of cannabis seeds and cannabis plants. Changes in the Company’s biological assets are as follows:
| September 30 | December 31 | ||||
| 2020 | 2019 | ||||
| $ | $ | ||||
| Carrying amount, beginning of year | 4,159 | 1,089 | |||
| Effect of unrealized changes in fair value of biological assets | 825 | 3,703 | |||
| Biological assets purchased | - | 197 | |||
| Biological asset expensed in research and development | - | (169 | ) | ||
| Deduct net abnormal plant destruction costs | (1,759 | ) | - | ||
| Transferred to inventory upon harvest | (1,654 | ) | (661 | ) | |
| Carrying amount, end of period | 1,571 | 4,159 |
During the three months ended September 30, 2020 the Company had to cull $1,759 worth of plants. This has been recorded as a loss on changes in fair value of biological assets.
As at September 30, 2020, included in the carrying amount of biological assets is $84 (December 31, 2019 -$85) in seeds and $1,487 (December 31, 2019 - $4,074) in live plants. The following inputs and assumptions are categorized within Level 3 on the fair value hierarchy, and are subject to volatility and several uncontrollable factors, which could significantly affect the fair value of the biological assets in future periods:
9
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
| Inputs and assumptions | Description | |
| Average market price per gram | Represents the the average wholesale market price per gram of dried bulk cannabis, based on quality (Tetrahydrocannabinol % and terpene profile). | |
| Average attrition rate | Represents the weighted average number of plants culled at each stage of production. | |
| Average yield per plant | Represents the average number of grams of dried cannabis inventory expected to be harvested from each cannabis plant. Based on historical yields. | |
| Average cost per plant | Represents costs incurred to grow plants at different stages of the production cycle. | |
| Stage of completion in the production process | Calculated by taking the weighted average number of days in production over a total average grow cycle of approximately 14 to 16 weeks based on location and strain. | |
| Production costs are capitalized to biological assets and include all direct and indirect costs relating to biological transformation. Costs include direct costs of production, such as labour, growing materials, as well as indirect cost s such as indirect labour, quality control costs, depreciation on production equipment, and overhead expenses including rent and utilities. | ||
The following table highlights the sensitivities and impact of changes in significant assumptions on the fair value of biological assets. As noted above in Note 2(c), the Company previously used the average retail price per gram of dried cannabis net of costs to sell for the period for all strains of cannabis sold at the finished good stage. The Company has revised its model to use the average wholesale market price per gram of dried bulk cannabis, based on quality (Tetrahydrocannabinol % and terpene profile).
| Range of inputs | Impact of fair value | ||||||||||||
| Significant inputs & assumptions | September 30, 2020 |
December 31, 2019 |
Sensitivity | September 30, 2020 |
December 31, 2019 |
||||||||
| Average market price per gram less cost to sell | $ | 1.14 | $ | 3.26 | Increase/decrease of $1 per gram (000's) | $ | 448 | $ | 1,031 | ||||
| Weighted average yield (gram per plant) | 116.55 | 92.13 | Increase/decrease by 10 gram per plant (000's) | $ | 9 | $ | 365 | ||||||
As of September 30, 2020, the weighted average stage of growth for the biological assets was 54% (December 31, 2019 – 45%). The average number of days from the point of propagation to harvest is 109 days.
The Company’s estimates are, by their nature, subject to change and changes in the significant assumptions will be reflected in the gain or loss on biological assets in future periods.
10
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
| 5. | Inventory |
The Company’s inventory is comprised of:
| September 30 | December 31 | ||||
| 2020 | 2019 | ||||
| $ | $ | ||||
| Dried bulk cannabis and packaged inventory | 4,818 | 3,545 | |||
| Cannabis oils | 2,378 | 2,465 | |||
| Supplies and consumables | 569 | 578 | |||
| 7,765 | 6,588 |
During the three and nine months ended September 30, 2020, inventory expensed to cost of goods sold was $2,498 and $6,384, respectively (September 30, 2019 - $8,264 and $15,683). The fair value change in biological assets included in cost of sale during the three and nine months ended September 30, 2020 was a loss of $2,339 and $825 (September 30, 2019 - (loss) gain of $(433) and $545).
During the three months and nine months ended September 30, 2020, a write-off of $1,819 and $2,874 respectively, were recognized for dried cannabis and packaged inventory (September 30, 2019 - $2,824 for the three and nine months) related to product deterioration and limited remaining shelf life. Included in this amount for the three and nine months ended September 30, 2020, is $420 of direct and indirect labour costs that were over and above product standard costs and were not capitalized into inventory.
11
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
| 6. | Property, Plant and Equipment |
The Company’s property, plant and equipment continuity is as follows:
| Production, Lab | |||||||||||||||
| Leasehold | and Growing | Other | |||||||||||||
| Land | Buildings | Improvement | Equipment | Computers | Equipment | Total | |||||||||
| $ | $ | $ | $ | $ | $ | $ | |||||||||
| Costs: | |||||||||||||||
| Balance, December 31, 2018 | 476 | 12,014 | 1,682 | 2,688 | 270 | 861 | 17,991 | ||||||||
| Acquired Through Naturals | - | - | 34 | - | 13 | 66 | 113 | ||||||||
| Additions | - | 20,612 | 758 | 4,203 | 73 | 526 | 26,172 | ||||||||
| Balance, December 31, 2019 | 476 | 32,626 | 2,474 | 6,891 | 356 | 1,453 | 44,276 | ||||||||
| Additions | - | - | - | 163 | 4 | 7 | 303 | ||||||||
| Disposals | - | - | - | (107 | ) | (13 | ) | - | (120 | ) | |||||
| Impairment (Note 8) | - | (2,354 | ) | - | - | - | - | (2,354 | ) | ||||||
| Balance, September 30, 2020 | 476 | 30,401 | 2,474 | 6,947 | 347 | 1,460 | 42,105 | ||||||||
| Production, Lab | |||||||||||||||
| Leasehold | and Growing | Other | |||||||||||||
| Land | Buildings | Improvement | Equipment | Computers | Equipment | Total | |||||||||
| $ | $ | $ | $ | $ | $ | $ | |||||||||
| Accumulated Depreciation: | |||||||||||||||
| Balance, December 31, 2018 | - | 154 | 155 | 548 | 76 | 92 | 1,025 | ||||||||
| Additions | - | 554 | 364 | 654 | 99 | 180 | 1,851 | ||||||||
| Balance, December 31, 2019 | - | 708 | 519 | 1,202 | 175 | 272 | 2,876 | ||||||||
| Additions | - | 982 | 433 | 929 | 72 | 198 | 2,614 | ||||||||
| Disposals | - | - | - | (28 | ) | (6 | ) | - | (34 | ) | |||||
| Balance, September 30, 2020 | - | 1,690 | 952 | 2,103 | 241 | 470 | 5,456 | ||||||||
| Production, Lab | |||||||||||||||
| Leasehold | and Growing | Other | |||||||||||||
| Land | Buildings | Improvement | Equipment | Computers | Equipment | Total | |||||||||
| $ | $ | $ | $ | $ | $ | $ | |||||||||
| Net book value: | |||||||||||||||
| September 30, 2020 | 476 | 28,711 | 1,522 | 4,844 | 106 | 990 | 36,649 | ||||||||
| December 31, 2019 | 476 | 31,918 | 1,955 | 5,689 | 181 | 1,181 | 41,400 |
Depreciation relating to manufacturing equipment and production facilities is initially expensed into production costs and then capitalized into inventory, and expensed to cost of sales upon the sale of goods. For the three and nine months ended September 30, 2020, $717 and $1,924 (September 30, 2019 - $272 and $493) of depreciation was recognized in cost of sales.
12
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
Plant under construction
During 2019, Phase 1 of construction on the Company’s new self-constructed production facility located in Metro Vancouver, British Columbia was completed. $14,343 was transferred from plant under construction to property, plant and equipment and began to be amortized. As at September 30, 2020, $8,795 of expenditures were capitalized to plant under construction relating to Phase 2 of the facility (December 31, 2019 - $8,407).
| 7. | Acquisitions |
a) Acquisitions completed during the year ended December 31, 2019
| (i) | Emerald Health Naturals |
On January 10, 2019, the TSXV approved the Company’s agreement with Emerald Health Bioceuticals, Inc. (“EHB”), an entity with common directors, to form Naturals. Naturals holds the Canadian distribution rights to EHB’s product line which focuses on health and wellness products that tap into the bodies Endocannabinoid System through the use of non cannabis-based ingredients. The Company controls Naturals and it has been consolidated, with a non-controlling interest recognized for the EHB owned portion.
b) Acquisitions completed during the year ended December 31, 2018
| (i) | Verdélite Sciences Inc. |
On May 30, 2019, the Company renegotiated the terms of the remaining $15,000 payable to the Vendors. $5,000 cash was paid in June 2019, with subsequent monthly payments of $1,000 commencing mid July through to mid November 2019, with a final payment of $5,000 plus accrued interest due on December 16, 2019. Interest on the full $15,000 balance began accruing May 31, 2019 at a rate of 10% per annum and accrues until such time as the entire balance is repaid. The amount outstanding as at September 30, 2020 was $9,000.
On July 31, 2020, the Company announced that they had entered into a share purchase agreement (the “Agreement”) in respect to the sale of the Company’s wholly-owned subsidiaries, Verdélite and Verdélite Property Holdings, Inc. (“Verdélite Property” and, together with Verdélite, the “Subsidiaries”). The Subsidiaries together own and operate a premium 88,000 square foot craft cannabis production indoor facility (the “Facility”) in St. Eustache, Québec.
Pursuant to the Agreement, the purchaser will purchase all of the issued and outstanding shares of the Subsidiaries in consideration for a cash purchase price of $21,000 subject to a 90-day working capital adjustment and certain other adjustments (the "Transaction"). The Agreement was negotiated at arm's length.
On the closing of the Transaction, the Subsidiaries will become wholly-owned subsidiaries of the purchaser, and the purchaser will continue the business of the Subsidiaries. Following closing of the Transaction, the Company will continue to sell its own products into the Québec market, subject to certain limited restrictions, and retains exclusive rights to its recently launched SouvenirTM brand.
The purchase price will be subject to a $750 holdback for the working capital adjustment and as an indemnity for certain pre-existing litigation. The Agreement contains representations and warranties,
13
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
covenants, conditions and indemnities for the benefit of each of the parties as are customary for transactions of this nature. Completion of the Transaction is subject to the completion of a number of conditions, including obtaining applicable consents and approval of the purchaser’s shareholders.
As a result of this, the Company assessed the Verdélite assets for impairment as at June 30, 2020 (Note 8).
| 8. | Intangible Assets |
The Company’s intangible assets continuity is as follows:
| Extraction | Health Canada | Computer | |||||||||
| Patents | Assets | Licence | Software | Total | |||||||
| Cost: | $ | $ | $ | $ | $ | ||||||
|
Balance, December 31, 2018 |
369 | - | 89,026 | 194 | 89,589 | ||||||
|
Acquired through Naturals (Note 7(a)(i)) |
89 | - | 149 | 11 | 249 | ||||||
|
Additions |
344 | 370 | - | 1,031 | 1,745 | ||||||
|
Impairment |
- | - | (65,122 | ) | - | (65,122 | ) | ||||
|
Balance, December 31, 2019 |
802 | 370 | 24,053 | 1,236 | 26,461 | ||||||
|
Additions |
34 | - | - | 44 | 78 | ||||||
|
Impairment |
- | - | (14,821 | ) | - | (14,821 | ) | ||||
| Balance, September 30, 2020 | 836 | 370 | 9,232 | 1,280 | 11,718 | ||||||
| Extraction | Health Canada | Computer | |||||||||
| Patents | Assets | Licence | Software | Total | |||||||
| Accumulated amortization: | $ | $ | $ | $ | $ | ||||||
|
Balance, December 31, 2018 |
- | - | 2,917 | 60 | 2,977 | ||||||
|
Additions |
- | - | 3,679 | 279 | 3,958 | ||||||
|
Balance, December 31, 2019 |
- | - | 6,596 | 339 | 6,935 | ||||||
|
Additions |
- | - | 458 | 404 | 862 | ||||||
| Balance, September 30, 2020 | - | - | 7,054 | 743 | 7,797 | ||||||
| Extraction | Health Canada | Computer | |||||||||
| Patents | Assets | Licence | Software | Total | |||||||
| Net book value: | $ | $ | $ | $ | $ | ||||||
|
September 30, 2020 |
836 | 370 | 2,178 | 537 | 3,921 | ||||||
|
December 31, 2019 |
802 | 370 | 17,457 | 897 | 19,526 |
14
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
On July 31, 2020, the Company announced that it had entered into a share purchase agreement relating to the sale of the Verdélite assets. The Company’s intention to sell the asset at June 30, 2020, as well as the continued decline in stock price and market capitalization of the Company were indicators of impairment at June 30, 2020. As a result of these factors, management evaluated its cash generating units (“CGU”) for impairment. It was determined that the Verdélite CGU was impaired at June 30, 2020. Taking into account the Company’s intention to sell the asset, the fair value less costs of disposal of Verdélite approximates its value in use at June 30, 2020, given that the value in use consists mainly of the net disposal proceeds. Management concluded that the carrying value was higher than the recoverable amount and recorded an impairment loss of $17,063. Management allocated the impairment loss based on the relative carrying amounts of the CGU’s assets at the impairment date which resulted in an impairment of $14,709 being allocated to the intangible assets and $2,354 being allocated to the building (production facility) of the Verdélite CGU.
Subsequent to the nine months ended September 30, 2020, the Company decreased the activity at Naturals. As a result of this, management evaluated the Naturals CGU and determined that certain assets of the CGU were impaired, resulting in an impairment of $112 that was recorded against certain licenses held by the subsidiary.
| 9. | Investment in Joint Venture – Held For Sale |
The Company’s investment in Pure Sunfarms is as follows:
| $ | |||
| Balance at December 31, 2019 | 64,603 | ||
| Equity adjustment due to change of ownership | (850 | ) | |
| Share of income | 4,497 | ||
| Balance at September 30, 2020 | 68,250 |
15
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
Summarized financial information for Pure Sunfarms is set out below:
| September 30 | December 31 | |||||
| 2020 | 2019 | |||||
| $ | $ | |||||
| Non current assets | 154,664 | 141,117 | ||||
| Current assets (a) | 100,060 | 82,340 | ||||
| Total assets | 254,724 | 223,457 | ||||
| Non current liabilities | 53,853 | 26,558 | ||||
| Current liabilities | 38,065 | 60,116 | ||||
| Total liabilities | 91,918 | 86,674 | ||||
| (a) includes cash and cash equivalents | 9,023 | 9,555 | ||||
| Nine months ended | Nine months ended | |||||
| September 30 | September 30 | |||||
| 2020 | 2019 | |||||
| $ | $ | |||||
| Sales | 53,533 | 71,548 | ||||
| Cost of Sales (b) | 32,027 | 18,785 | ||||
| Gross margin before fair value changes | 21,506 | 52,763 | ||||
| Change in fair value of biological asset | 3,271 | (15,025 | ) | |||
| Gross margin | 18,234 | 67,788 | ||||
| Selling, general and administrative expenses | 9,090 | 7,454 | ||||
| Income from operations | 9,144 | 60,334 | ||||
| Other (income) loss | (4,585 | ) | 723 | |||
| Income before taxes | 13,729 | 59,611 | ||||
| Provision for income taxes | 3,707 | 13,404 | ||||
| Net income | 10,022 | 46,207 | ||||
| (b) includes $2,668 of amortization expense (September 30, 2019 $1,791) | ||||||
| Net Income | 10,022 | 46,207 | ||||
| Elimination of transactions with the Company | 2,518 | (3,765 | ) | |||
| Fair value adjustment | (2,559 | ) | (4,243 | ) | ||
| Net income from Joint Venture for equity accounting purposes | 9,981 | 38,199 | ||||
| Emerald's share of income from Joint Venture (1) | 4,497 | 19,099 |
(1) During the year ended December 31, 2019, the Company's share of income from the Joint Venture was 50% up until November 18, 2019. Subsequently, the Company’s share of ownership decreased to 46.47%. During the nine months ended September 30, 2020, the Company's share of income from the Joint Venture was 46.47% up until February 29, 2020. Subsequently, share of ownership dropped to 42.60% up until March 31, 2020 and to 41.28% in April 2020. As at September 30, 2020 the Company's share of income from the Joint Venture was 41.28%.
16
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
A reconciliation of the summarized financial information to the carrying amount of the investment in Pure Sunfarms is set out below:
| September 30 | December 31 | |||||
| 2020 | 2019 | |||||
| $ | $ | |||||
| Total net assets of Pure Sunfarms | 162,806 | 136,784 | ||||
| Ownership interest held by the Company | 67,207 | 65,105 | ||||
| Cumulative adjustments carried forward | 480 | 2,669 | ||||
| Fair value adjustment recognized during the period | (1,106 | ) | (3,563 | ) | ||
| Elimination of transactions with the Company during the period | 1,149 | (128 | ) | |||
| Transaction costs cumulative | 520 | 520 | ||||
| Carrying amount of the investment | 68,250 | 64,603 |
As at September 30, 2020, Pure Sunfarms has not issued dividends. As a privately held company, there are no quoted market prices available for the shares of Pure Sunfarms.
On March 6, 2020, the Company completed a Settlement Agreement (the “Settlement Agreement”) with its Joint Venture partner. Pursuant to the Settlement Agreement, the Supply Agreements (collectively, the "Supply Agreements") entered into between the Company and Pure Sunfarms dated December 21, 2018 (the “2018 Supply Agreement”), which was for the 40% supply provision by Pure Sunfarms to the Company in 2019, at a guaranteed price for 2019, and dated March 29, 2019, which was for the 25% supply provision by Pure Sunfarms, at a guaranteed price adjusted on a semi-annual basis to the Company from 2020 to the end of 2022, respectively, were both terminated effective as of December 31, 2019, and the Company was released from all previous, current, and future obligations, liabilities and payments thereunder. The termination of the Supply Agreements removed the uncertainty regarding the potential obligation of the Company to make any payments to Pure Sunfarms under the terms of those Agreements and provided the Company with full flexibility regarding future access to, and supply of, wholesale cannabis. In exchange, the Company forfeited all amounts due from Pure Sunfarms pursuant to a shareholder’s loan of $13,000 plus accrued interest of $1,062 that the Company previously advanced to Pure Sunfarms. This was effective as of December 31, 2019.
On March 6, 2020, also as part of the Settlement Agreement, the Company issued a promissory note to Pure Sunfarms in the amount of $952 which bears interest at a rate of 6.2% per annum and will mature on the earlier of (a) December 31, 2020; (b) the Company ceasing to be a shareholder of Pure Sunfarms; or (c) the acquisition by any party of a majority of the outstanding shares of the Company. The principal amount is the amount owed by the Company to Pure Sunfarms as remittance for bulk sales under the 2018 Supply Agreement.
As part of the Settlement Agreement the Company also transferred 2.5% of its Pure Sunfarms equity to Village Farms. The Parties agreed that the $8,000 provided by Village Farms to Pure Sunfarms in 2020 was to be converted into additional common shares of Pure Sunfarms. Upon completion of this conversion and the other transactions referred to above, the Company’s share of income dropped to a 42.60% equity interest.
In April 2020, the Company announced that its Joint Venture had expanded its credit facility with its existing lender to $59,000 including accordion provisions of $20,000. The expanded credit facility (the “Credit Facility”) consists of a $7,500 revolving operating loan (the “Revolver”) and a $12,500 term loan (the “New Term Loan”), in addition to its existing $19,000 term loan (the “Existing Term Loan”). The New Term Loan
17
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
is specifically designated for the 1.1 million square foot Delta 2 greenhouse while the Existing Term Loan is specifically designated for the 1.1 million square foot Delta 3 greenhouse. The $7,500 Revolver and the $12,500 New Term Loan include an accordion provision that allows the Joint Venture to request additional lender commitments of up to an additional $7,500 and $12,500, respectively, subject to an additional lender entering the syndicate on or before May 30, 2020. Each of the components of the Credit Facility, including the existing term loan, mature on February 7, 2022. As part of this transaction, Village Farms completed an additional investment in the Joint Venture of $8,000, which reduced the Company’s equity position in the Joint Venture by 1.32% to 41.28%.
On July 1, 2020, the Company announced that its Joint Venture had further expanded its Credit Facility to its full $59,000 capacity with the completion of the Credit Facility’s accordion feature. The Credit Facility now consists of a $15,000 revolving operating loan and a $25,000 New Term Loan, in addition to its $19,000 Existing Loan. The New Term Loan is specifically designated for the 1.1 million square foot Delta 2 greenhouse while the Existing Term Loan is specifically designated for the 1.1 million square foot Delta 3 greenhouse facility. Each of the components of the Credit Facility, including the Existing Term Loan, mature on February 7, 2022.
On November 2, 2020, the Company completed a share purchase agreement (the “PSF Share Purchase Agreement”) with its Joint Venture partner, see Note 21 for details surrounding the sale. Due to the stage of the sale negotiations as of September 30, 2020, the Company determined that the Joint Venture met the criteria for IFRS 5 Non-current Asset held for Sale. Therefore, the Company measured the Joint Venture at the lower of its’ carrying amount and fair value less costs to sell. The carrying amount of the Joint Venture was the lower value, so this amount has been disclosed as a current asset on the statement of financial position.
| 10. | Related Party Transactions |
With Emerald Health Sciences Inc.
As of the nine months ended September 30, 2020, Emerald Health Sciences Inc. (“Sciences”) held an aggregate of 39,401,608 Common Shares, representing 20% (December 31, 2019 – 29,687,942 shares, representing 18%) of the issued and outstanding Common Shares and it also held 9,099,706 (December 31, 2019 – 9,099,706) common share purchase warrants of the Company.
Sciences charged the Company $Nil during the three and nine months ended September 30, 2020 (September 30, 2019 - $875 and $2,975) for services related to financing, business development, research and development, investor relations and acquisition activities. On October 1, 2019, the Company and Sciences amended the previously disclosed third amended and restated independent contractor agreement pursuant to which Sciences provided certain management services to the Company. No services were provided by Sciences in the three and nine months ended September 30, 2020. The amount owing at September 30, 2020 is $1,673 (December 31, 2019 - $1,846). This amount is included in the due to related parties caption on the condensed interim consolidated statements of financial position and is non-interest bearing. As at September 30, 2020, Sciences owed the Company $37 (December 31, 2019 – $31) for invoices paid on behalf of Sciences. This amount is included in the due from related parties caption on the condensed interim consolidated statements of financial position and is non-interest bearing.
In July 2019, the Company amended a previous loan agreement with Sciences pursuant to which Sciences agreed to loan up to $15,000 to the Company, in amounts and at times agreed to by the parties. Amounts loaned bear interest at 12% per annum and are repayable on demand. On January 31, 2020, the Company settled its outstanding balance related to the loan agreement and accrued loan interest totalling $794 as well as its outstanding balance related to trade payables for hemp purchased totalling $2,023, by way of
18
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
issuance of shares for debt. The Company issued 9,713,666 common shares of the Company at a deemed price of $0.29 per Common Share.
On May 27, 2020, the Company announced that it had terminated certain related party agreements pertaining to its cannabis cultivation operation in Metro Vancouver. Previously existing rights and continuing usage of the land and its cultivation operation in Metro Vancouver are unaffected by these terminations. Both the sublease agreement and cultivation agreement with Sciences were previously announced on October 4, 2019. The Company and Sciences agreed not to pursue the transactions contemplated by such agreements and agreed to terminate both the sublease agreement and the cultivation agreement in May 2020. The Company did not make any payments to Sciences under either the sublease agreement or the cultivation agreement. The Company’s previously existing rights related to the 12-acre parcel that was subject to the sublease agreement and the cultivation agreement are unaffected by these terminations.
On July 15, 2020, the Company announced that it had terminated certain related party agreements pertaining to consulting services and a loan facility. Sciences and the Company entered into an independent contractor agreement on May 1, 2015, as amended from time to time (the “Consulting Agreement”), and a loan agreement on August 25, 2015, as amended from time to time (the “Loan Agreement”). Sciences and the Company agreed to terminate the Consulting Agreement and Loan Agreement.
With the Company’s Joint Venture
As of September 30, 2020, Pure Sunfarms owes the Company $170 (December 31, 2019 - $170) for expenditures made on behalf of the Joint Venture. As at September 30, 2020, the Company owes to Pure Sunfarms $991 (December 31, 2019 - $957), of which $986 ($952 principle and $34 interest) is for a promissory note that was issued on March 6, 2020 as part of the Settlement Agreement with the Company’s Joint Venture. The note bears interest at a rate of 6.2% per annum, and will mature on the earlier of (a) December 31, 2020; (b) the Company ceasing to be a shareholder of Pure Sunfarms; or (c) the acquisition by any party of a majority of the outstanding shares of the Company. The principal amount is the amount owed by the Company to Pure Sunfarms as remittance for bulk sales under the 2018 Supply Agreement.
With a Company Controlled by the Company’s Executive Chairman
During the year ended December 31, 2017, the Company entered into a 30-year lease with a company (the “Landlord”) that is controlled by Avtar Dhillon, MD, the Executive Chairman of the Company with respect to land in Metro Vancouver, British Columbia on which the Company is constructing its new production facility. The lease amount was determined by an independent valuation, and was approved the non-conflicted directors of the Company. During the three and nine months ended September 30, 2020, the Company paid to the Landlord $99 and $269 (September 30, 2019 - $85 and $255) in rent. The Landlord was reimbursed by the Company for $Nil during the three and nine months ended September 30, 2020 (September 30, 2019 - $10 and $189) for development fees and services related to construction of the Company’s new facility. As at September 30, 2020, the Company recognized lease liabilities of $3,526 (December 31, 2019 - $3,554) relating to the land in Metro Vancouver with a corresponding right of use asset.
With a Company Whose CEO is Also a Director of the Company
The Company holds 1,666,667 common shares and 1,666,667 common share purchase warrants of Avricore Health Inc. (“Avricore”, formerly VANC Pharmaceuticals Inc.). Naturals holds 3,030,303 common share purchase warrants of Avricore. Subsequent to September 30, 2020, the Company sold its shares in Avricore, see Note 21.
19
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
With a Company Whose President is Also a CEO of the Subsidiary
Naturals owes to GAB Innovations, Inc. $47 (December 31, 2019 - $93) for expenditures made on behalf of Naturals.
Remuneration of directors and key management of the Company
| For the three | For the three | For the nine | For the nine | ||||||
| months ended | months ended | months ended | months ended | ||||||
| September 30 2020 | September 30 2019 | September 30 2020 | September 30 2019 | ||||||
| $ | $ | $ | $ | ||||||
| Wage and short term benefits | 366 | 506 | 1,271 | 1,067 | |||||
| Share-based compensation (Note 13) | 395 | 1,966 | 1,194 | 4,819 | |||||
| 761 | 2,472 | 2,465 | 5,886 |
The remuneration awarded to directors and to senior key management including the Executive Chairman, the Chief Executive Officer and President, the Chief Financial Officer, and the Chief Commercial Officer includes the following expenses recognized during the period:
Included in Due to Related Parties on the condensed interim consolidated statements of financial position at September 30, 2020 is $584 (December 31, 2019 - $50) due to related parties with respect to key management personnel and expense reimbursements and are non-interest bearing.
These transactions are recorded at the amounts agreed upon between the two parties.
| 11. | Convertible Debenture |
During the nine months ended September 30, 2020, interest expense of $384 which had accrued during the year ended December 31, 2019, was settled with the issuance of 1,322,627 common shares of the Company. Also during the nine months ended September 30, 2020, interest expense of $625 which had accrued during the six months ended June 30, 2020, was settled with the issuance of 3,571,428 common shares of the Company.
During the three and nine months ended September 30, 2020, interest expense of $315 and $939, and accretion expense of $467 and $1,374 were recorded.
| 12. | Share Capital |
Authorized
-
Unlimited number of Common Shares without par value
-
Unlimited number of preferred shares without par value, issuable in series
Issued
-
205,027,772 Common Shares (December 31, 2019 – 160,986,373)
-
Nil preferred shares (December 31, 2019 – Nil)
20
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
During the nine months ended September 30, 2020, the outstanding share capital increased by 44,041,399 Common Shares due to the following transactions:
-
The Company filed a prospectus supplement in connection with an at-the-market equity program (“ATM Program”) that it established with GMP Securities L.P. (the “Agent”) in the three months ended March 31, 2019. In connection with the ATM Program, the Company entered into an equity distribution agreement with the Agent. The ATM Program allows the Company to issue Common Shares from treasury having an aggregate gross sales price of up to $39,000 to the public from time to time, at the Company’s discretion, at the prevailing market price when issued on the TSXV or on any other marketplace for the common shares in Canada. The ATM Program is effective until the earlier of April 13, 2021 or completion of the sale of the maximum amount of shares thereunder. Sales of Common Shares will be made through “at-the-market distributions” as defined in National Instrument 44-102 – Shelf Distributions on the TSXV or on any other existing marketplace for the common shares in Canada. The Common Shares will be distributed at the prevailing market prices at the time of the sale and, as a result, prices may vary among purchasers and during the period of distribution. During the nine months ended September 30, 2020 the Company did not issue any Common Shares in connections with the ATM Program. This ATM Program was cancelled in August 2020 with the establishment of a new ATM Program;
-
On August 13, 2020, the Company announced that it had established an at-the-market equity program (“new ATM Program”) that allows the Company to issue common shares from treasury having an aggregate gross sales price of up to $3,250 to the public from time to time at the Company’s discretion, at the prevailing market price when issued. The new ATM Program is effective until the earlier of April 13, 2021 and completion of the sale of the maximum amount of shares thereunder and will be activated from time to time at the Company’s discretion if and when required based on the Company’s working capital requirements and capital expenditures and relative cost of other funding options. During the quarter 1,312,500 common shares were issued under the new ATM Program for gross proceeds of $287;
-
Issued 9,713,666 Common Shares with a value of $2,914 to settle an outstanding loan amount and trade payables with Sciences (see Note 10);
-
Issued 4,894,055 Common Shares with a value of $1,009 to settle outstanding interest on the convertible debentures (see Note 11);
-
During February 2020, the Company closed a private placement resulting in the issuance of 10,344,827 units at a price of $0.29 per unit, for gross proceeds of $3,000. Each unit consisted of one Common Share and one common share purchase warrant. The warrants were valued using the residual value method at $164 (Note 14);
-
During April 2020, 175,000 restricted stock units vested triggering the issuance of 175,000 Common Shares for no cash proceeds;
-
On April 9, 2020, the Company amended the terms of certain common share purchase warrants that were originally issued on September 9, 2019 (the “September Warrants”). An aggregate of 12,500,000 September Warrants were initially issued at an exercise price of $2.00 per Common Share of the Company in connection with a $25,000 convertible debenture financing. The Company amended the exercise price of the September Warrants such that: 6,250,000 September Warrants will have an exercise price of $0.17 per Common Share (the "$0.17 Warrants"). If, at any time prior to the expiry date of the $0.17 Warrants, the closing market price of the Common Shares on the TSXV is greater than $0.2125 for 10 consecutive trading days, the Company may deliver a notice to the holder of the $0.17 Warrants accelerating the expiry date of the $0.17 Warrants to the date that is 30 days following the date of such notice; and 6,250,000 Warrants will have an exercise price of $0.21 per Common Share (the "$0.21 Warrants"). If, at any time prior to the expiry date of the $0.21 Warrants, the closing market price of the Common Shares on the TSXV is greater than $0.2625 for 10 consecutive trading days, the Company may deliver a notice to the holder of the $0.21 Warrants
21
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
-
During June 2020, the Company closed a private placement resulting in the issuance of 11,351,351 units at a price of $0.185 per unit, for gross proceeds of $2,100. Each unit consisted of one Common Share and one common share purchase warrant. The warrants were valued using the residual value method at $Nil (Note 14).
accelerating the expiry date of the $0.21 Warrants to the date that is 30 days following the date of such notice (collectively, the “Warrant Repricing”). All other provisions of the September Warrants will remain the same and will still expire on September 9, 2021. Upon TSXV approval, the holders of the $0.17 Warrants immediately exercised all such warrants for proceeds of $1,063 to the Company; and
| 13. | Share-Based Compensation |
(a) Stock Options
The Board of Directors has the discretion to determine to whom options will be granted, the number and exercise price of such options and the terms and time frames in which the options will vest and be exercisable. The exercise price of the options must be no less than the closing market price of the Common Shares on the day preceding the grant. The maximum number of common shares issuable upon the exercise or redemption and settlement of all awards granted shall not exceed 10% of the issued and outstanding shares at the time of granting of such award less the number of shares reserved for issuance under all other security based compensation arrangements of the Company. The following types of awards can be issued: stock options, share appreciation rights, restricted share units and other performance awards.
The following table summarizes the stock options that remain outstanding as at September 30, 2020:
| Weighted Average | |||||
| Stock Options | Exercise Price | ||||
| # | $ | ||||
| Balance at December 31, 2018 | 9,894,211 | 2.31 | |||
|
Granted |
6,615,500 | 3.77 | |||
|
Forfeited |
(2,564,934 | ) | 3.77 | ||
|
Exercised |
(1,563,143 | ) | 0.74 | ||
| Balance at December 31, 2019 | 12,381,634 | 2.99 | |||
|
Granted |
9,877,500 | 0.24 | |||
|
Forfeited |
(3,187,472 | ) | 2.88 | ||
|
Expired |
(1,500,000 | ) | 0.45 | ||
| Balance at September 30, 2020 | 17,571,662 | 1.68 |
During the nine months ended September 30, 2020, the Company granted 9,877,500 stock options to employees, directors and consultants. The stock options granted had exercise prices between $0.17 and $0.32, have expiry dates of five years. Stock options issued to employees and consultants vest over three years and stock options issued to directors vest either immediately, or over twelve months. The weighted average fair value of the stock options granted was $0.14. There were no options exercised during the three and nine months ended September 30, 2020.
22
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
The fair values of the options granted during the nine months ended September 30, 2020 and 2019 were determined on the date of the grant using the Black-Scholes option pricing model with the following assumptions:
| September 30 | September 30 | |||||
| 2020 | 2019 | |||||
| Risk free interest rate | 0.25% - 1.65% | 1.29% - 1.85% | ||||
| Expected life of options (years) | 2.59 | 2.78 | ||||
| Expected annualized volatility | 93.07% - 104.54% | 95.99% - 105.68% | ||||
| Expected dividend yield | Nil | Nil | ||||
| Weighted average Black Scholes value of each option | $ | 0.14 | $ | 2.36 |
Volatility was determined by using the historical volatility of the Company. The expected life in years represents the period of time that options granted are expected to be outstanding. The risk-free rate is based on Canada government bonds with a remaining term equal to the expected life of the options.
Incentive stock options outstanding and exercisable at September 30, 2020 are summarized as follows:
| Outstanding | Exercisable | |||||
| Weighted | Weighted | |||||
| Range of | Remaining | average | average | |||
| exercise | contractual | exercise | exercise | |||
| prices | Quantity | life (years) | price | Quantity | price | |
| $ | $ | $ | ||||
| 0.165 - 0.29 | 8,970,000 | 4.60 | 0.24 | 3,364,711 | 0.21 | |
| 0.30 - 1.16 | 1,210,000 | 1.70 | 0.64 | 1,015,000 | 0.70 | |
| 1.17 - 1.47 | 843,662 | 3.60 | 1.26 | 758,662 | 1.24 | |
| 1.48 - 3.69 | 1,556,750 | 3.28 | 2.80 | 965,250 | 2.87 | |
| 3.70 - 4.60 | 4,766,250 | 3.17 | 4.18 | 3,391,250 | 4.18 | |
| 4.61 - 5.69 | 225,000 | 2.42 | 5.68 | 168,750 | 5.68 | |
| 17,571,662 | 3.82 | 1.68 | 9,663,623 | 2.10 | ||
The Company recorded share-based compensation expense related to the stock options of $532 and $1,843 for the three and nine months ended September 30, 2020 (September 30, 2019 - $2,495 and $10,258).
23
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
(b) Restricted Share Units (“RSUs”)
The Board of Directors has the discretion to determine to whom restricted share units (“RSUs”) will be granted, the number granted, and the terms and time frames in which the RSUs will vest and be settled.
| Weighted average | |||||
| fair value per | |||||
| Number of RSUs | unit at issue | ||||
| # | $ | ||||
| Balance at December 31, 2018 | 830,000 | 3.74 | |||
Granted |
475,000 | 4.15 | |||
Settled |
(355,000 | ) | 4.27 | ||
Forfeited |
(280,000 | ) | 2.06 | ||
| Balance at December 31, 2019 | 670,000 | 4.46 | |||
|
Granted |
550,000 | 0.27 | |||
|
Settled |
(175,000 | ) | 4.15 | ||
|
Forfeited |
(65,152 | ) | 3.25 | ||
| Balance at September 30, 2020 | 979,848 | 2.24 |
During the three and nine months ended September 30, 2020, the Company issued 550,000 RSUs to various employees and consultants, that vest 375,000 on February 6, 2021 and 175,000 on August 3, 2021 and settle in Common Shares. The Company recorded share-based compensation expense related to the RSUs of $223 and $742 for the three and nine months ended September 30, 2020 (September 30, 2019 – $498 and $1,178).
| 14. | Warrants |
For details of the issuance of warrants see Note 12.
| Weighted | |||||
| Number of | Average | ||||
| Warrants | Exercise Price | ||||
| # | $ | ||||
| Balance at December 31, 2018 | 8,411,764 | 2.92 | |||
| Issued in September 2019 | 12,500,000 | 0.17 | |||
| Issued in November 2019 | 4,385,965 | 0.75 | |||
| Issued in December 2019 | 5,172,942 | 0.39 | |||
| Expired | (4,000,000) | 5.20 | |||
| Balance at December 31, 2019 | 26,470,671 | 0.42 | |||
| Issued in February 2020 | 10,344,827 | 0.39 | |||
| Exercised in April 2020 | (6,250,000) | 0.17 | |||
| Issued in June 2020 | 11,351,351 | 0.27 | |||
| Balance at September 30, 2020 | 41,916,849 | 0.41 | |||
| Expiry: | |||||
| September 2021 | 6,250,000 | 0.17 | |||
| November 2021 | 4,411,764 | 0.85 | |||
| June 2023 | 11,351,351 | 0.27 | |||
| November 2024 | 4,385,965 | 0.75 | |||
| December 2024 | 5,172,942 | 0.39 | |||
| February 2025 | 10,344,827 | 0.39 | |||
| Balance at September 30, 2020 | 41,916,849 | 0.41 |
24
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
| 15. | Leases |
The Company’s leases consist primarily of land, office space, as well as miscellaneous production and other equipment. Information about the right-of-use assets and associated lease liabilities are seen below.
a) Right-of-Use Assets
| Land | Buildings | Equipment | Total | ||||||
| $ | $ | $ | $ | ||||||
| Costs: | |||||||||
|
Balance, applied January 1, 2019 |
3,634 | 1,372 | 178 | 5,184 | |||||
|
Additions |
- | 1,774 | 21 | 1,795 | |||||
|
Disposals |
- | - | (38 | ) | (38 | ) | |||
|
Balance, applied December 31, 2019 |
3,634 | 3,146 | 161 | 6,941 | |||||
|
Additions |
- | 57 | - | 57 | |||||
|
Disposals |
- | (1,774 | ) | - | (1,774 | ) | |||
| Balance, September 30, 2020 | 3,634 | 1,429 | 161 | 5,224 | |||||
| Accumulated Depreciation: | |||||||||
|
Balance, applied January 1, 2019 |
182 | 450 | 22 | 654 | |||||
|
Additions |
121 | 518 | 20 | 659 | |||||
|
Balance, applied December 31, 2019 |
303 | 968 | 42 | 1,313 | |||||
|
Additions |
91 | 396 | 33 | 520 | |||||
|
Disposals |
- | (433 | ) | - | (433 | ) | |||
| Balance, September 30, 2020 | 394 | 931 | 75 | 1,400 | |||||
| Carrying value: | |||||||||
|
September 30, 2020 |
3,240 | 498 | 86 | 3,824 | |||||
|
December 31 2019 |
3,331 | 2,178 | 119 | 5,628 |
b) Lease Liabilities
The following table reconciles the opening and ending balances of the lease liabilities:
| $ | |||
| Lease liabilities recognized at December 31, 2019 | 5,944 | ||
| Lease additions | 57 | ||
| Lease renewals | (83 | ) | |
| Lease disposals | (1,358 | ) | |
| Lease payments | (629 | ) | |
| Interest incurred | 267 | ||
| Balance, September 30, 2020 | 4,198 |
25
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
The Company expects the following maturities of its undiscounted lease liabilities:
| Contractual Undiscounted Cash Flows: | |||
| $ | |||
| Within 1 year | 652 | ||
| 1 <3 years | 1,027 | ||
| 3 <5 years | 665 | ||
| Over 5 years | 6,960 | ||
| Balance, September 30, 2020 | 9,304 |
For the three and nine months ended September 30, 2020, amounts of $52 and $168 respectively, (September 30, 2019 - $10 and $28) have been recorded in operating costs for the Company related to variable lease payments, and amounts relating to short term leases, and leases for low value assets.
| 16. | Revenue |
A summary of the Company’s sales by product line is provided in the table below:
| For the three | For the three | For the nine | For the nine | ||||||
| months ended | months ended | months ended | months ended | ||||||
| September 30 2020 | September 30 2019 | September 30 2020 | September 30 2019 | ||||||
| $ | $ | $ | $ | ||||||
| Dried Cannabis | 3,595 | 7,855 | 7,839 | 12,920 | |||||
| Cannabis Oils | 695 | 1,781 | 2,775 | 4,296 | |||||
| Other | 21 | 82 | 136 | 182 | |||||
| Total | 4,311 | 9,718 | 10,750 | 17,398 |
| 17. | General and Administrative Expenses |
| For the three | For the three | For the nine | For the nine | ||||||
| months ended | months ended | months ended | months ended | ||||||
| September 30 2020 | September 30 2019 | September 30 2020 | September 30 2019 | ||||||
| $ | $ | $ | $ | ||||||
| Professional, director and consulting fees | 1,416 | 1,387 | 2,631 | 3,780 | |||||
| Corporate communications and media | 54 | 345 | 198 | 1,330 | |||||
| Wages and benefits | 775 | 1,625 | 3,000 | 4,254 | |||||
| Office and general | 487 | 781 | 1,496 | 2,013 | |||||
| Travel and accommodations | - | 200 | 85 | 444 | |||||
| Total | 2,732 | 4,338 | 7,410 | 11,821 |
| 18. | Segmented Information |
Operating segments are components of the Company that engage in business activities which generate revenues and incur expenses (including intercompany revenues and expenses related to transactions conducted with other components of the Company). The operations of an operating segment are distinct and the operating results are regularly reviewed by the chief operating decision maker (“CODM”), the Company’s CEO, for the purposes of resource allocation decisions and assessing its performance. Reportable segments are Operating segments whose revenues or profit/loss or total assets exceed ten percent or more of those of the combined entity.
26
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
Key measures used by the CODM to assess performance and make resource allocation decisions include sales, income (loss) from operations and net (loss) income. The Company's operating results are divided into two reportable segments. The two reportable segments are (i) Cannabis; and (ii) Other. The Company primarily operates in the Cannabis segment.
| Cannabis | Other | Total | |||||
| $ | $ | $ | |||||
| For the three months ended September 30, 2020 | |||||||
| Sales | 4,290 | 21 | 4,311 | ||||
| Interest and other income | 16 | 13 | 29 | ||||
| Loss from operations | (6,251 | ) | (3,628 | ) | (9,879 | ) | |
| Net loss and comprehensive loss | (7,615 | ) | (4,043 | ) | (11,658 | ) | |
| Share of income (loss) from joint venture | (520 | ) | - | (520 | ) | ||
| For the nine months ended September 30, 2020 | |||||||
| Sales | 10,668 | 82 | 10,750 | ||||
| Interest and other income | 729 | 122 | 851 | ||||
| Loss from operations | (8,836 | ) | (10,170 | ) | (19,006 | ) | |
| Net loss and comprehensive loss | (23,640 | ) | (11,840 | ) | (35,480 | ) | |
| Share of income (loss) from joint venture | 4,497 | - | 4,497 | ||||
| Cannabis | Other | Total | |||||
| $ | $ | $ | |||||
| For the three months ended September 30, 2019 | |||||||
| Sales | 9,636 | 82 | 9,718 | ||||
| Interest and other income | 692 | (471 | ) | 221 | |||
| Loss from operations | (6,143 | ) | (8,552 | ) | (14,695 | ) | |
| Net loss and comprehensive loss | (9,364 | ) | (8,097 | ) | (17,461 | ) | |
| Share of income (loss) from joint venture | (1,202 | ) | - | (1,202 | ) | ||
| For the nine months ended September 30, 2019 | |||||||
| Sales | 17,222 | 176 | 17,398 | ||||
| Interest and other income | 644 | 380 | 1,024 | ||||
| Loss from operations | (20,248 | ) | (18,144 | ) | (38,392 | ) | |
| Net loss and comprehensive loss | (3,960 | ) | (17,602 | ) | (21,562 | ) | |
| Share of income (loss) from joint venture | 19,099 | - | 19,099 |
27
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
| 19. | Financial Instruments |
Financial instruments are measured either at fair value or at amortized cost.
The carrying value of the cash and cash equivalents, accounts receivable (excluding statutory receivable balances), due from related parties, refundable deposits, accounts payable and accrued liabilities, current portion of long-term debt, deferred payment, payable to joint venture and amounts due to related parties, approximate the fair value because of the short-term nature of these instruments. These are carried at amortized cost.
The Company’s financial instruments that are recorded at fair value are presented in the following table:
| Fair Value Measurement | ||||||||||
| Level 1 | Level 2 | Level 3 | Total | |||||||
| As at September 30, 2020 | $ | $ | $ | $ | ||||||
| Financial Assets | ||||||||||
|
Long-term investments |
250 | 223 | - | 473 | ||||||
| As at December 31, 2019 | ||||||||||
| Financial Assets | ||||||||||
|
Long-term investments |
50 | 31 | - | 81 | ||||||
Fair Value Hierarchy
Financial instruments recorded at fair value are classified using a hierarchy that categorizes into three levels the inputs to valuation techniques used to measure fair value. The three levels of hierarchy are:
Level 1 - Unadjusted quoted prices in active markets for identical assets or liabilities;
Level 2 - Inputs other than quoted prices that are observable for the asset or liability, either directly or indirectly; and
Level 3 - Inputs for the asset or liability that are not based on observable market data.
The Company is exposed to varying degrees to a variety of financial instrument related risks:
Currency risk
The Company’s functional and presentation currency is the Canadian dollar and major purchases are transacted in Canadian dollars. As a result, the Company’s exposure to foreign currency risk is minimal.
Credit risk
Credit risk is the risk of an unexpected loss to the Company if a customer or third-party to a financial instrument fails to meet its contractual obligations. The Company’s maximum exposure to credit risk as at September 30, 2020 is the carrying value of its financial assets. The Company’s cash and redeemable short-term investment certificates are largely held in large Canadian financial institutions. The Company maintains cash deposits with Schedule A financial institutions, which from time to time may exceed
28
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
federally insured limits. With regards to receivables, the Company is not exposed to significant credit risk as the Company’s sales are to government bodies or are typically paid at the time of the transaction. The Company provides credit to some of its customers in the normal course of business.
Interest rate risk
Interest rate risk is the risk the fair value or future cash flows of a financial instrument will fluctuate because of changes in market interest rates. Financial assets and liabilities with variable interest rates expose the Company to cash flow interest rate risk. The Company does not hold any financial liabilities with variable interest rates. The Company does maintain bank accounts and redeemable short-term investment certificates which earn interest at variable rates, but it does not believe it is currently subject to any significant interest rate risk.
Liquidity risk
Liquidity risk is the risk that the Company will not be able to meet its obligations associated with financial liabilities. The Company’s ability to continue as a going concern is dependent on management’s ability to raise required funding through future equity issuances and through short term borrowing. The Company manages its liquidity risk by forecasting cash flows from operations and anticipating any investing and financing activities. Management and the Board of Directors are actively involved in the review, planning and approval of significant expenditures and commitments. Please refer to Note 1 for discussion on the Company’s ability to cover its current liabilities.
COVID-19
The spread of COVID-19 is expected to have a material adverse effect on global and regional economies, and continues to negatively impact stock markets, including the trading price of the Company’s shares. The adverse effects on the economy, the stock market, and the Company’s share price could adversely impact the Company’s ability to raise capital, or its ability to pursue other strategic initiatives.
| 20. | Capital Management |
As at September 30, 2020, the capital structure of the Company consists of $134,545 (December 31, 2019 - $156,621) in shareholders’ equity and debt.
The Company’s objective when managing its capital is to ensure sufficient equity financing to fund its planned operations in a way that maximizes the shareholder return given the assumed risks of its operations. The Company considers shareholders’ equity as capital. Through the ongoing management of its capital, the Company will modify the structure of its capital based on changing economic conditions. In doing so, the Company may issue new shares. Annual budgeting is the primary tool used to manage the Company’s capital. Updates are made as necessary to both capital expenditure and operational budgets in order to adapt to changes in risk factors, proposed expenditure programs and market conditions.
29
| EMERALD HEALTH THERAPEUTICS, INC. |
| NOTES TO CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS |
| For the three and nine months ended September 30, 2020 and 2019 |
| (Unaudited – Amounts reflected in thousands of Canadian dollars, except share and per share amounts) |
| 21. | Subsequent Events |
Share Purchase Agreement for Sale of Pure Sunfarms
On November 2, 2020, the Company completed the sale of its 41.28% interest in its Joint Venture, Pure Sunfarms for consideration of $79,900. Pursuant to the PSF Share Purchase Agreement, the Company sold its 36,958,500 common shares to its Joint Venture Partner, representing all of the remaining shares of Pure Sunfarms not previously held by the Company’s Joint Venture partner, for an aggregate purchase price of $79,900 (the "Purchase Price"). The Company received $60,000 of the Purchase Price in cash at closing and acquired a secured promissory note to the Company in the principal amount of $19,900 (the "Note"). The Note will mature in six months from the date hereof, is secured against a certain number of common shares of Pure Sunfarms held by the Joint Venture partner and bears interest at a rate of 12% per annum. In addition, the Company's obligations under a promissory note in the principal amount of $952 that the Company had issued to PSF on March 6, 2020, have been settled without any payment.
Sale of Shares in Avricore
On October 5, 2020, the Company sold its 1,666,667 common shares in Avricore for $206.
Settlement of Convertible Debentures
On November 24, 2020, the Company fully prepaid in cash all amounts owing under the Convertible Debentures issued to certain Canadian institutional accredited investors (the “Debentureholders”) by the Company on September 9, 2019 (see Note 11). As a result, the Convertible Debentures are now terminated, and the Company has no further obligations thereunder.
The Convertible Debentures were due to mature on September 9, 2021, with no right for the Company to prepay amounts owing. However, the Debentureholders consented to the early repayment of all amounts owing under the Convertible Debentures in exchange for payment of a consent fee of $82 (the “Consent Fee”). A total of $25,568 in cash was paid by the Company to the Debentureholders, representing $25,000 in principal repayment, $486 in accrued interest and the Consent Fee.
30