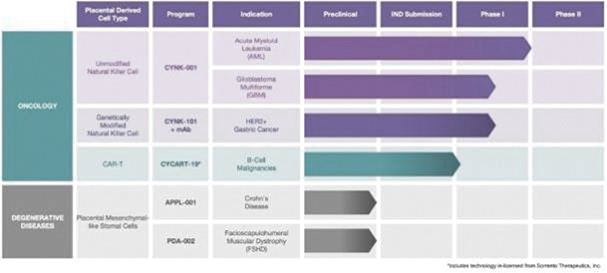
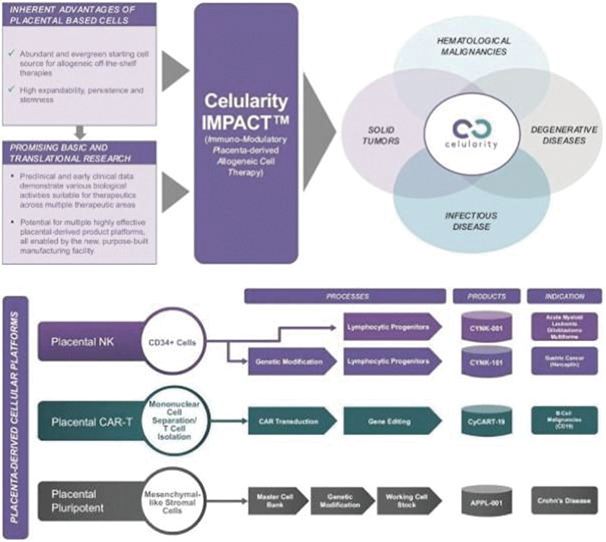
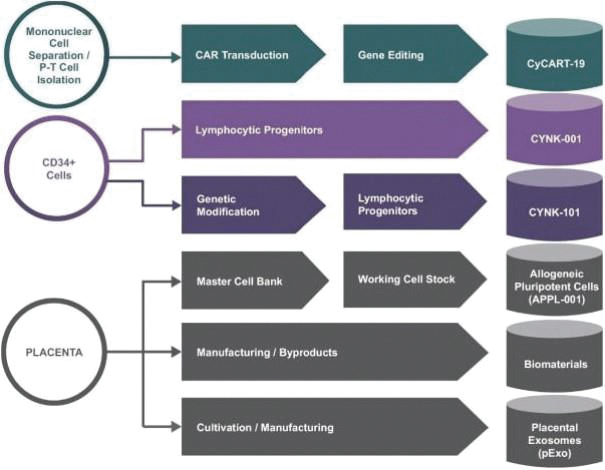
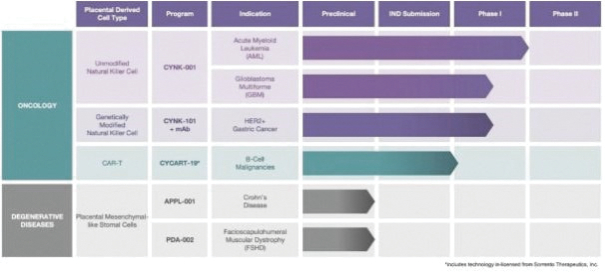
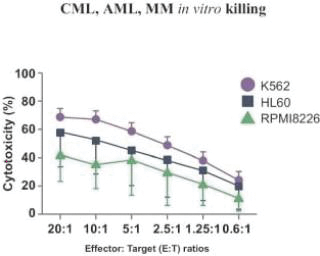
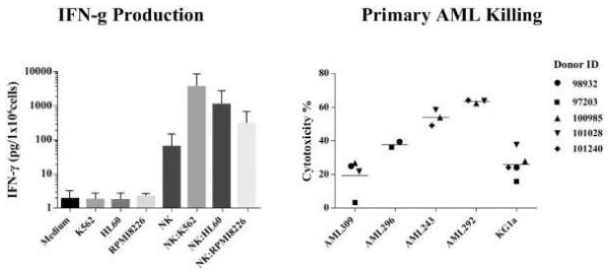
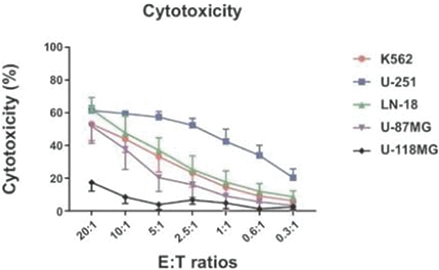
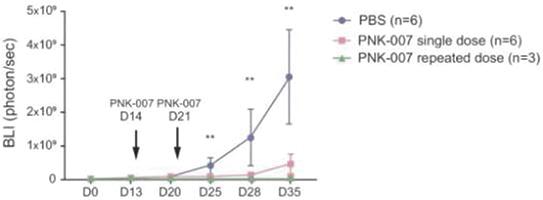
The information in this preliminary prospectus is not complete and may be changed. These securities may not be sold until the registration statement filed with the Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell these securities nor does it seek an offer to buy these securities in any jurisdiction where the offer or sale is not permitted.
Subject to Completion, Dated May 2
4
, 2022 PRELIMINARY PROSPECTUS

Up to 8,108,110 Shares of Class A Common Stock
This prospectus relates to the offer and sale from time to time by the selling securityholder named in this prospectus, or the Selling Securityholder, of an aggregate of up to 8,108,110 shares of our Class A common stock, $0.0001 par value per share, or Class A common stock, which consists of (i) up to 4,054,055 shares of our Class A common stock and (ii) up to 4,054,055 shares of our Class A common stock issuable upon the exercise of certain common stock purchase warrants, or the PIPE Warrants, which shares of Class A common stock and PIPE Warrants were originally issued in a private placement to the Selling Securityholder.
We will not receive any proceeds from the sale of shares of Class A common stock by the Selling Securityholder pursuant to this prospectus but we will receive the proceeds from any exercise of any PIPE Warrants for cash. We will pay the expenses, other than underwriting discounts and commissions and expenses incurred by the Selling Securityholder for brokerage, accounting, tax or legal services or any other expenses incurred by the Selling Securityholder in disposing of the securities, associated with the sale of securities pursuant to this prospectus.
We are registering the securities for resale pursuant to the Selling Securityholder’s registration rights under a registration rights agreement between us and the Selling Securityholder. Our registration of the securities covered by this prospectus does not mean that the Selling Securityholder will offer or sell any of the shares of Class A common stock. The Selling Securityholder may offer, sell or distribute all or a portion of its shares of Class A common stock publicly or through private transactions at prevailing market prices or at negotiated prices. We provide more information about how the Selling Securityholder may sell the Shares in the section entitled “.”
Plan of Distribution
We are an “emerging growth company” as defined under the federal securities laws and, as such, have elected to comply with certain reduced public company reporting requirements for this prospectus and our other filings with the Securities and Exchange Commission.
The Class A common stock and warrants issued as part of the units in our initial public offering, or the Public Warrants, are listed on The Nasdaq Capital Market, or the Nasdaq, under the symbols “CELU” and “CELUW,” respectively. On May 23, 2022, the closing price of the Class A common stock was $7.10 and the closing price for the Public Warrants was $0.62.
Investing in our securities involves a high degree of risk. You should review carefully the risks and uncertainties described in the section titled “
” beginning on page 5 of this prospectus, and under similar headings in any amendments or supplements to this prospectus.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities, or passed upon the accuracy or adequacy of this prospectus. Any representation to the contrary is a criminal offense.
The date of this prospectus is , 2022