Exhibit 99.1

Aphria Inc.
CONDENSED INTERIM CONSOLIDATED FINANCIAL STATEMENTS
FOR THE THREE MONTHS AND SIX MONTHS ENDED NOVEMBER 30, 2018 AND NOVEMBER 30, 2017
(Unaudited, expressed in thousands of Canadian Dollars, unless otherwise noted)
Aphria Inc.
Condensed Interim Consolidated Statements of Financial Position
(Unaudited - in thousands of Canadian dollars)
| Note | November 30, 2018 | May 31, 2018 | ||||||||||
| Assets | ||||||||||||
| Current assets | ||||||||||||
| Cash and cash equivalents | $ | 152,074 | $ | 59,737 | ||||||||
| Marketable securities | 4 | 32,747 | 45,062 | |||||||||
| Accounts receivable | 13,807 | 3,386 | ||||||||||
| Other current assets | 5 | 28,700 | 14,384 | |||||||||
| Inventory | 6 | 40,342 | 22,150 | |||||||||
| Biological assets | 7 | 6,096 | 7,331 | |||||||||
| Assets held for sale | – | 40,620 | ||||||||||
| Current portion of convertible notes receivable | 12 | 16,396 | 1,942 | |||||||||
| 290,162 | 194,612 | |||||||||||
| Capital assets | 9 | 413,963 | 303,151 | |||||||||
| Intangible assets | 10 | 425,234 | 226,444 | |||||||||
| Convertible notes receivable | 12 | 12,595 | 16,129 | |||||||||
| Interest in equity investees | 13 | 9,612 | 4,966 | |||||||||
| Long-term investments | 14 | 162,035 | 46,028 | |||||||||
| Promissory note receivable | 15 | 128,859 | – | |||||||||
| Goodwill | 11 | 656,039 | 522,762 | |||||||||
| $ | 2,098,499 | $ | 1,314,092 | |||||||||
| Liabilities | ||||||||||||
| Current liabilities | ||||||||||||
| Accounts payable and accrued liabilities | $ | 49,953 | $ | 31,517 | ||||||||
| Income taxes payable | 7,645 | 3,584 | ||||||||||
| Deferred revenue | 35,395 | 2,607 | ||||||||||
| Current portion of promissory note payable | 18 | 604 | 610 | |||||||||
| Current portion of long-term debt | 19 | 3,388 | 2,140 | |||||||||
| Current portion of option payment liability | 20 | 11,654 | – | |||||||||
| Current portion of derivative liability | – | 3,396 | ||||||||||
| 108,639 | 43,854 | |||||||||||
| Long-term liabilities | ||||||||||||
| Long-term debt | 19 | 51,165 | 28,337 | |||||||||
| Option payment liability | 20 | 32,722 | – | |||||||||
| Derivative liability | – | 9,055 | ||||||||||
| Deferred tax liability | 16 | 110,239 | 59,253 | |||||||||
| 302,765 | 140,499 | |||||||||||
| Shareholders’ equity | ||||||||||||
| Share capital | 21 | 1,650,077 | 1,113,981 | |||||||||
| Warrants | 22 | 1,336 | 1,375 | |||||||||
| Share-based payment reserve | 22,701 | 22,006 | ||||||||||
| Accumulated other comprehensive loss | – | (801 | ) | |||||||||
| Non-controlling interest | 24 | 18,612 | 9,580 | |||||||||
| Retained earnings | 103,008 | 27,452 | ||||||||||
| 1,795,734 | 1,173,593 | |||||||||||
| $ | 2,098,499 | $ | 1,314,092 | |||||||||
Nature of operations (Note 1)
Commitments and contingencies (Note 32)
Subsequent events (Note 33)
Approved on behalf of the Board:
| “John Cervini” | “Cole Cacciavillani” |
| Signed: Director | Signed: Director |
The accompanying notes are an integral part of these condensed interim consolidated financial statements
| 2 |
Aphria Inc.
Condensed Interim Consolidated Statements of Income and Comprehensive Income
(Unaudited - in thousands of Canadian dollars, except share and per share amounts)
| For
the three months ended November 30, | For
the six months ended November 30, | |||||||||||||||||||
| Note | 2018 | 2017 | 2018 | 2017 | ||||||||||||||||
| Revenue | $ | 24,524 | $ | 8,504 | $ | 37,816 | $ | 14,624 | ||||||||||||
| Excise taxes | (2,856 | ) | – | (2,856 | ) | – | ||||||||||||||
| Net revenue | 21,668 | 8,504 | 34,960 | 14,624 | ||||||||||||||||
| Production costs | 6 | 9,971 | 2,746 | 14,412 | 4,092 | |||||||||||||||
| Other costs of sales | 1,540 | – | 1,933 | – | ||||||||||||||||
| Gross profit before fair value adjustments | 10,157 | 5,758 | 18,615 | 10,532 | ||||||||||||||||
| Fair value adjustment on sale of inventory | 6 | 8,328 | 2,671 | 12,533 | 3,807 | |||||||||||||||
| Fair value adjustment on growth of biological assets | 7 | (4,154 | ) | (3,115 | ) | (13,665 | ) | (7,380 | ) | |||||||||||
| Gross profit | 5,983 | 6,202 | 19,747 | 14,105 | ||||||||||||||||
| Operating expenses: | ||||||||||||||||||||
| General and administrative | 25 | 12,276 | 1,973 | 21,127 | 3,708 | |||||||||||||||
| Share-based compensation | 26 | 2,574 | 2,200 | 8,696 | 4,709 | |||||||||||||||
| Selling, marketing and promotion | 8,336 | 2,819 | 13,077 | 4,767 | ||||||||||||||||
| Amortization | 2,617 | 276 | 5,891 | 515 | ||||||||||||||||
| Research and development | 612 | 80 | 874 | 170 | ||||||||||||||||
| Transaction costs | 1,123 | – | 1,988 | – | ||||||||||||||||
| 27,538 | 7,348 | 51,653 | 13,869 | |||||||||||||||||
| (21,555 | ) | (1,146 | ) | (31,906 | ) | 236 | ||||||||||||||
| Non-operating items: | ||||||||||||||||||||
| Consulting revenue | – | 183 | – | 476 | ||||||||||||||||
| Foreign exchange (loss) gain | (194 | ) | 282 | (253 | ) | 131 | ||||||||||||||
| Gain (loss) on marketable securities | 4 | 57 | 55 | (110 | ) | (1,691 | ) | |||||||||||||
| Loss on sale of capital assets | 9 | – | – | – | (7 | ) | ||||||||||||||
| Gain (loss) from equity investees | 13 | 46,896 | (457 | ) | 58,739 | (1,746 | ) | |||||||||||||
| Deferred gain on sale of intellectual property | 107 | 233 | 340 | 467 | ||||||||||||||||
| Finance income, net | 27 | 4,855 | 862 | 5,914 | 1,328 | |||||||||||||||
| Unrealized gain on convertible notes receivable | 12 | 2,567 | 665 | 2,862 | 1,212 | |||||||||||||||
| Gain on long-term investments | 28 | 30,503 | 6,075 | 53,203 | 25,157 | |||||||||||||||
| Unrealized loss on financial liabilities | (560 | ) | – | (975 | ) | – | ||||||||||||||
| 84,231 | 7,898 | 119,720 | 25,327 | |||||||||||||||||
| Income before income taxes | 62,676 | 6,752 | 87,814 | 25,563 | ||||||||||||||||
| Income taxes | 16 | 7,902 | 297 | 11,864 | 4,067 | |||||||||||||||
| Net income | 54,774 | 6,455 | 75,950 | 21,496 | ||||||||||||||||
| Other comprehensive gain (loss) | ||||||||||||||||||||
| Other comprehensive gain (loss) from equity investee | 13 | – | 520 | – | (801 | ) | ||||||||||||||
| Net comprehensive income | $ | 54,774 | $ | 6,975 | $ | 75,950 | $ | 20,695 | ||||||||||||
| Total comprehensive income is attributable to: | ||||||||||||||||||||
| Shareholders of Aphria Inc. | 54,970 | 6,975 | 76,357 | 20,695 | ||||||||||||||||
| Non-controlling interest | 24 | (196 | ) | – | (407 | ) | – | |||||||||||||
| $ | 54,774 | $ | 6,975 | $ | 75,950 | $ | 20,695 | |||||||||||||
| Weighted average number of common shares - basic | 244,873,891 | 138,839,530 | 235,166,745 | 138,775,253 | ||||||||||||||||
| Weighted average number of common shares - diluted | 249,303,182 | 145,878,443 | 239,417,492 | 146,075,449 | ||||||||||||||||
| Earnings per share - basic | 29 | $ | 0.22 | $ | 0.05 | $ | 0.32 | $ | 0.15 | |||||||||||
| Earnings per share - diluted | 29 | $ | 0.22 | $ | 0.04 | $ | 0.32 | $ | 0.15 | |||||||||||
The accompanying notes are an integral part of these condensed interim consolidated financial statements
| 3 |
Aphria Inc.
Condensed Interim Consolidated Statements of Changes in Equity
(Unaudited - in thousands of Canadian dollars, except share amounts)
Number of common shares | Share capital (Note 21) | Warrants (Note 22) | Share-based payment reserve | Accumulated other comprehensive loss | Non-controlling interest (Note 24) | Retained | Total | |||||||||||||||||||||||||
| Balance at May 31, 2017 | 138,628,704 | $ | 274,317 | $ | 445 | $ | 3,230 | $ | – | $ | – | $ | (4,123 | ) | $ | 273,869 | ||||||||||||||||
| Share issuance - November 2017 bought deal | 12,689,675 | 86,661 | – | – | – | – | – | 86,661 | ||||||||||||||||||||||||
| Share issuance - warrants exercised | 417,855 | 638 | – | – | – | – | – | 638 | ||||||||||||||||||||||||
| Share issuance - options exercised | 132,488 | 249 | – | (92 | ) | – | – | – | 157 | |||||||||||||||||||||||
| Share issuance - deferred share issuance costs | 2,525 | 15 | – | – | – | – | – | 15 | ||||||||||||||||||||||||
| Share-based payments | – | – | – | 4,495 | – | – | – | 4,495 | ||||||||||||||||||||||||
| Income tax recovery on share issuance costs | – | 1,412 | – | – | – | – | – | 1,412 | ||||||||||||||||||||||||
| Shares held in escrow for services not yet earned | – | 187 | – | – | – | – | – | 187 | ||||||||||||||||||||||||
| Net comprehensive income for the period | – | – | – | – | (801 | ) | – | 21,496 | 20,695 | |||||||||||||||||||||||
| Balance at November 30, 2017 | 151,871,247 | $ | 363,479 | $ | 445 | $ | 7,633 | $ | (801 | ) | $ | – | $ | 17,373 | $ | 388,129 | ||||||||||||||||
| Number of common shares | Share capital (Note 21) | Warrants (Note 22) | Share-based payment reserve | Accumulated other comprehensive loss | Non-controlling interest (Note 24) | Retained earnings | Total | |||||||||||||||||||||||||
| Balance at May 31, 2018 | 210,169,924 | $ | 1,113,981 | $ | 1,375 | $ | 22,006 | $ | (801 | ) | $ | 9,580 | $ | 27,452 | $ | 1,173,593 | ||||||||||||||||
| Share issuance - June 2018 bought deal | 21,835,510 | 245,925 | – | – | – | – | – | 245,925 | ||||||||||||||||||||||||
| Additional share issuance - Broken Coast acquisition | 19,963 | 297 | – | – | – | – | – | 297 | ||||||||||||||||||||||||
| Share issuance - LATAM acquisition | 15,678,310 | 273,900 | – | – | – | – | – | 273,900 | ||||||||||||||||||||||||
| Share issuance - warrants exercised | 316,063 | 1,409 | (39 | ) | – | – | – | – | 1,370 | |||||||||||||||||||||||
| Share issuance - options exercised | 1,911,974 | 11,139 | – | (7,390 | ) | – | – | – | 3,749 | |||||||||||||||||||||||
| Income tax recovery on share issuance costs | – | 3,426 | – | – | – | – | – | 3,426 | ||||||||||||||||||||||||
| Share-based payments | – | – | – | 8,085 | – | – | – | 8,085 | ||||||||||||||||||||||||
| Elimination of CTA on disposal of equity investee | – | – | – | – | 801 | – | (801 | ) | – | |||||||||||||||||||||||
| Non-controlling interest | – | – | – | – | – | 9,439 | – | 9,439 | ||||||||||||||||||||||||
| Net comprehensive income for the period | – | – | – | – | – | (407 | ) | 76,357 | 75,950 | |||||||||||||||||||||||
| Balance at November 30, 2018 | 249,931,744 | $ | 1,650,077 | $ | 1,336 | $ | 22,701 | $ | – | $ | 18,612 | $ | 103,008 | $ | 1,795,734 | |||||||||||||||||
The accompanying notes are an integral part of these condensed interim consolidated financial statements
| 4 |
Aphria Inc.
Condensed Interim Consolidated Statements of Cash Flows
(Unaudited - in thousands of Canadian dollars)
| For the six months ended November 30, | ||||||||||||
| Note | 2018 | 2017 | ||||||||||
| Cash generated from (used in) operating activities: | ||||||||||||
| Net income for the period | $ | 75,950 | $ | 21,496 | ||||||||
| Adjustments for: | ||||||||||||
| Future income taxes | 16 | 3,785 | 3,702 | |||||||||
| Fair value adjustment on sale of inventory | 6 | 12,533 | 3,807 | |||||||||
| Fair value adjustment on growth of biological assets | 7 | (13,665 | ) | (7,380 | ) | |||||||
| Loss on marketable securities | 4 | 110 | 1,691 | |||||||||
| Unrealized foreign exchange gain | (6 | ) | (77 | ) | ||||||||
| Amortization | 9,10 | 8,860 | 1,404 | |||||||||
| Loss on sale of capital assets | 9 | – | 7 | |||||||||
| Unrealized gain on convertible notes receivable | 12 | (2,862 | ) | (1,212 | ) | |||||||
| Gain on dilution of ownership in equity investee | 13 | (2,210 | ) | (7,535 | ) | |||||||
| Loss from equity investees | 13 | 822 | 9,281 | |||||||||
| Gain on sale of equity investee | 13 | (57,351 | ) | – | ||||||||
| Deferred gain recognized | (618 | ) | (467 | ) | ||||||||
| Consulting revenue | 18 | – | (476 | ) | ||||||||
| Other non-cash items | (1 | ) | 4 | |||||||||
| Share-based compensation | 26 | 8,696 | 4,709 | |||||||||
| Gain on long-term investments | 28 | (53,203 | ) | (25,157 | ) | |||||||
| Unrealized loss on financial liabilities | 13 | 975 | – | |||||||||
| Change in non-cash working capital | 30 | 2,013 | (3,382 | ) | ||||||||
| (16,172 | ) | 415 | ||||||||||
| Cash provided by financing activities: | ||||||||||||
| Share capital issued, net of cash issuance costs | 245,925 | 86,661 | ||||||||||
| Share capital issued on warrants and options exercised | 5,119 | 810 | ||||||||||
| Advances from related parties | 8 | 968 | 2,823 | |||||||||
| Repayment of amounts due to related parties | 8 | (968 | ) | (2,327 | ) | |||||||
| Proceeds from long-term debt | 19 | 24,927 | – | |||||||||
| Repayment of long-term debt | 19 | (863 | ) | (375 | ) | |||||||
| 275,108 | 87,592 | |||||||||||
| Cash used in investing activities: | ||||||||||||
| Investment in marketable securities | 4 | – | (5,000 | ) | ||||||||
| Proceeds from disposal of marketable securities | 4 | 12,205 | 34,801 | |||||||||
| Investment in capital and intangible assets, net of shares issued | 9,10 | (113,399 | ) | (59,023 | ) | |||||||
| Proceeds from disposal of capital assets | 9 | – | 200 | |||||||||
| Convertible notes advances | 12 | (10,000 | ) | (14,001 | ) | |||||||
| Repayment of convertible notes receivable | 1,942 | – | ||||||||||
| Investment in long-term investments and equity investees | (61,027 | ) | (10,897 | ) | ||||||||
| Proceeds from disposal of long-term investments and equity investees | 5,027 | 2,090 | ||||||||||
| Net cash paid on business acquisitions and investment in CannInvest Africa Ltd. | (1,347 | ) | – | |||||||||
| (166,599 | ) | (51,830 | ) | |||||||||
| Net increase in cash and cash equivalents | 92,337 | 36,177 | ||||||||||
| Cash and cash equivalents, beginning of period | 59,737 | 79,910 | ||||||||||
| Cash and cash equivalents, end of period | $ | 152,074 | $ | 116,087 | ||||||||
The accompanying notes are an integral part of these condensed interim consolidated financial statements
| 5 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
| 1. | Nature of operations |
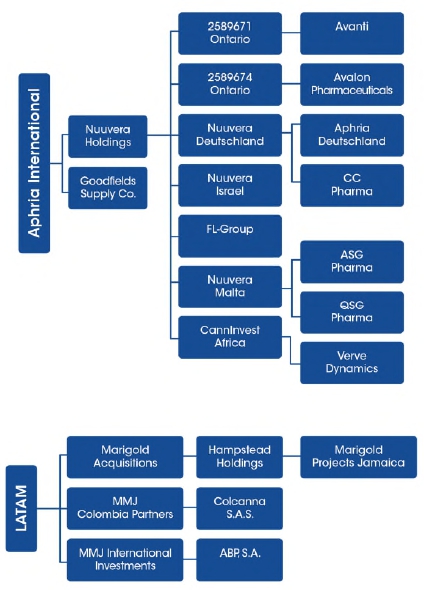
Aphria Inc. (the "Company" or “Aphria”) was continued in Ontario and is licensed to produce and sell cannabis under The Cannabis Act. In February 2018, the Company acquired Broken Coast Cannabis Ltd. (“Broken Coast”) (Note 11). Broken Coast is licensed to produce and sell cannabis under The Cannabis Act. In March 2018, the Company acquired Nuuvera Inc. (“Nuuvera”) (Note 11). Nuuvera is an international organization with a focus on building a global cannabis brand, with operations in Germany, Italy, Malta, and Lesotho. In July 2018, Aphria Inc. and its wholly-owned subsidiary, Pure Natures Wellness Inc. (o/a Aphria) amalgamated. In September 2018, the Company acquired LATAM Holdings Inc. (“LATAM”) (Note 11). This purchase provides Aphria an early foothold into the Latin American cannabis market whereby LATAM holds licenses and license applications presently in-process for production, import, export and sale of cannabis and cannabis derivatives in Colombia, Argentina and Jamaica.
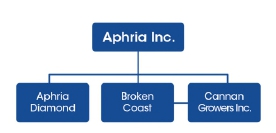
1974568 Ontario Ltd. (“Aphria Diamond”) is a 51% majority owned subsidiary of the Company, incorporated in November 2017. Aphria Diamond has applied for its cultivation licence under the provisions of The Cannabis Act.
The registered office of the Company is located at 5300 Commerce Court West, 199 Bay Street, Toronto, Ontario. However, the Company is in the process of registering a new address.
The Company’s common shares are listed under the symbol “APHA” on the Toronto Stock Exchange (“TSX”) in Canada and the New York Stock Exchange (“NYSE”) in the United States.
These condensed interim consolidated financial statements were approved by the Company’s Board of Directors on January 10, 2019.
| 2. | Basis of preparation |
| (a) | Statement of compliance |
The Company’s condensed interim consolidated financial statements have been prepared in accordance with IAS 34, “Interim Financial Reporting”. These condensed interim consolidated financial statements do not include all notes of the type normally included within the annual financial report and should be read in conjunction with the audited financial statements of the Company for the year ended May 31, 2018, which have been prepared in accordance with International Financial Reporting Standards (“IFRS”) as issued by the International Accounting Standards Board and Interpretations of the IFRS Interpretations Committee.
| (b) | Basis of measurement |
These condensed interim consolidated financial statements have been prepared on the going concern basis, under the historical cost convention except for certain financial instruments that are measured at fair value and biological assets that are measured at fair value less costs to sell, as detailed in the Company’s accounting policies.
| (c) | Functional currency |
The Company and its subsidiaries’ functional currency, as determined by management, is Canadian dollars. These condensed interim consolidated financial statements are presented in Canadian dollars.
| (d) | Foreign currency translation |
All figures presented in the condensed interim consolidated financial statements are reflected in Canadian dollars, which is the functional currency of the Company and all of its subsidiaries.
Foreign currency transactions are translated into Canadian dollars at exchange rates in effect on the date of the transactions. Monetary assets and liabilities denominated in foreign currencies are translated to Canadian dollars at the foreign exchange rate applicable at the statement of financial position date. Realized and unrealized exchange gains and losses are recognized through profit and loss.
The assets and liabilities of foreign operations, including marketable securities, long-term investments and promissory notes payable, are translated in Canadian dollars at period-end exchange rates. Income and expenses, and cash flows of foreign operations are translated into Canadian dollars using average exchange rates. Exchange differences resulting from translating foreign operations are recognized in other comprehensive income and accumulated in equity.
| 6 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
| (e) | Basis of consolidation |
Subsidiaries are entities controlled by the Company. Control exists when the Company has the power, directly and indirectly, to govern the financial and operating policies of an entity and be exposed to the variable returns from its activities. The financial statements of subsidiaries are included in the condensed interim consolidated financial statements from the date that control commences until the date that control ceases.
| Subsidiaries | Jurisdiction of incorporation | Ownership interest (1) |
| Aphria (Arizona) Inc.(2) | Arizona, United States | 100% |
| Cannan Growers Inc. | British Columbia, Canada | 100% |
| Nuuvera Inc. | Ontario, Canada | 100% |
| Nuuvera Holdings Ltd. | Ontario, Canada | 100% |
| ARA - Avanti Rx Analytics Inc. | Ontario, Canada | 100% |
| Avalon Pharmaceuticals Inc. | Ontario, Canada | 100% |
| 2589671 Ontario Inc. | Ontario, Canada | 100% |
| 2589674 Ontario Inc. | Ontario, Canada | 100% |
| Nuuvera Israel Ltd.(2) | Israel | 100% |
| Nuuvera Deutschland GmbH | Germany | 100% |
| Aphria Deutschland GmbH | Germany | 100% |
| FL-Group | Italy | 100% |
| Broken Coast Cannabis Ltd. | British Columbia, Canada | 100% |
| Goodfields Supply Co. Ltd. | United Kingdom | 100% |
| LATAM Holdings Inc. | British Columbia, Canada | 100% |
| MMJ Colombia Partners Inc. | Ontario, Canada | 100% |
| Marigold Acquisitions Inc. | British Columbia, Canada | 100% |
| Hampstead Holdings Ltd. | Bermuda | 100% |
| MMJ International Investments Inc. | British Columbia, Canada | 100% |
| ABP, S.A. | Argentina | 100% |
| Marigold Projects Jamaica Limited | Jamaica | 95% |
| Nuuvera Malta Ltd. | Malta | 90% |
| ASG Pharma Ltd. | Malta | 90% |
| QSG Health Ltd. | Malta | 90% |
| ColCanna S.A.S. | Colombia | 90% |
| 1974568 Ontario Ltd. | Ontario, Canada | 51% |
| Aphria Italy S.p.A. | Italy | 50.1% |
| CannInvest Africa Ltd. | South Africa | 50% |
| Verve Dynamics Incorporated (Pty) Ltd. | Lesotho | 30% |
(1) The Company defines ownership interest as the interest in which the Company is entitled a proportionate share of net income. Legal ownership of some subsidiaries may differ from ownership interest shown above.
(2) Represents inactive subsidiaries, which have no operations and does not own any assets, save and except for a related party balance owing to the entity related to a tax liability.
Intragroup balances, and any unrealized gains and losses or income and expenses arising from transactions with jointly controlled entities are eliminated to the extent of the Company’s interest in the entity.
The Company treats transactions with non-controlling interests that do not result in a loss of control as transactions with equity owners of the Company. A change in ownership interest results in an adjustment between the carrying amounts of the controlling and non-controlling interests to reflect their relative interests in the subsidiary. Any difference between the amount of the adjustment to non-controlling interests and any consideration paid or received is recognized in a separate reserve within equity attributable to the owners of the Company.
| 7 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
| (f) | Amalgamation |
Effective June 1, 2017, CannWay Pharmaceuticals Ltd. (“CannWay”), a wholly-owned subsidiary of the Company, was amalgamated with Pure Natures Wellness Inc. (o/a Aphria). The Company had historically presented all balances and activities of CannWay as a fully consolidated entity for financial statement presentation purposes. As of the date of amalgamation, CannWay did not have any assets or outstanding liabilities. There are no material changes to be considered prospectively or to the comparative consolidated statements as a result of the amalgamation.
Effective July 23, 2018, Pure Natures Wellness Inc. (o/a Aphria). (“PNW”), a wholly-owned subsidiary of the Company, was amalgamated with Aphria Inc. The Company had historically presented all balances and activities of PNW as a fully consolidated entity for financial statement presentation purposes. There were no material changes to be considered prospectively or to the comparative consolidated statements as a result of the amalgamation.
| (g) | Interest in equity investees |
The Company’s interest in equity investees is comprised of its interest in Althea Company Pty Ltd. (“Althea”).
In accordance with IFRS 10, associates are those in which the Company has significant influence, but not control or joint control over the financial and accounting policies.
Interests in associates are accounted for using the equity method in accordance with IAS 28. They are recognized initially at cost, which includes transaction costs. After initial recognition, the condensed interim consolidated financial statements include the Company’s share of the profit or loss and other comprehensive income (“OCI”) of equity investees until the date on which significant influence ceases.
If the Company’s share of losses in an equity investment equals or exceeds its interest in the entity, including any other unsecured long-term receivables, the group does not recognize further losses, unless it has incurred obligations or made payments on behalf of the other entity.
Unrealized gains on transactions between the Company and its associates are eliminated to the extent of the Company’s interest in these entities. Unrealized losses are also eliminated unless the transaction provides evidence of an impairment of the asset transferred.
The carrying amount of equity investments is tested for impairment in accordance with the policy described in the annual audited financial statements.
| 3. | Significant accounting policies |
These condensed interim consolidated financial statements have been prepared following the same accounting policies used in the preparation of the audited financial statements of the Company for the year ended May 31, 2018.
New standards applicable during the reporting period
IFRS 9 - Financial Instruments; Classification and Measurement, effective for annual periods beginning on or after January 1, 2018, with early adoption permitted, introduces new requirements for the classification, measurement and derecognition of financial instruments and introduces a new impairment model for financial assets.
Under IFRS 9, financial instruments are initially measured at fair value plus, in the case of a financial asset or financial liability not at fair value through profit or loss, transaction costs. Subsequently, all assets within scope of IFRS 9 are measured at:
| (i) | Amortized cost; |
| (ii) | Fair value through other comprehensive income (“FVOCI”); or |
| (iii) | Fair value through profit or loss (“FVTPL”). |
The classification is based on whether the contractual cash flows give rise to payments on specified dates that are solely payments of principal and interest (the “SPPI test”), and the objective of the Company’s business model is to hold assets only to collect cash flows, or to collect cash flows and to sell (the “Business Model test”). Financial assets are required to be reclassified only when the business model under which they are managed has changed. All reclassifications are to be applied prospectively from the reclassification date.
| 8 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
The impairment requirements under IFRS 9 are based on an expected credit loss model, replacing the IAS 39 incurred loss model. The expected credit loss model applies to debt instruments recorded at amortized cost or at FVOCI, such as loans, debt, securities and trade receivables, lease receivables and most loan commitments and financial guarantee contracts.
The following table summarizes the original measurement categories under IAS 39 and the new measurement categories under IFRS 9 for each class of the Company’s financial assets and financial liabilities:
| Financial assets/liabilities | IAS 39 Classification | IFRS 9 Classification |
| Cash and cash equivalents | FVTPL | FVTPL |
| Marketable securities | FVTPL | FVTPL |
| Accounts receivable | loans and receivables | amortized cost |
| Other receivables | loans and receivables | amortized cost |
| Convertible notes receivable | AFS | FVTPL |
| Long-term investments | FVTPL | FVTPL |
| Accounts payable and accrued liabilities | other financial liabilities | other financial liabilities |
| Income taxes payable | other financial liabilities | other financial liabilities |
| Promissory note payable | other financial liabilities | other financial liabilities |
| Long-term debt | other financial liabilities | other financial liabilities |
| Derivative liability | derivative financial instruments | FVTPL |
There were no other changes on adoption aside from the above classification changes.
IFRS 15 - Revenue from Contracts with Customers; effective for annual periods beginning on or after January 1, 2018, specifies how and when to recognize revenue, based on a five-step model, and enhances relevant disclosures to be applied to all contracts with customers.
The Company has applied IFRS 15 retrospectively and determined that there is no change to the comparative periods or transitional adjustments required as a result of the adoption of this standard. The Company’s accounting policy for revenue recognition under IFRS 15 is as follows:
To recognize revenue under IFRS 15, the Company applies the following five steps:
| 1. | Identify the contract(s) with a customer |
| 2. | Identify the performance obligations in the contract |
| 3. | Determine the transaction price |
| 4. | Allocate the transaction price to the performance obligations in the contract |
| 5. | Recognize revenue when or as the Company satisfies a performance obligation |
Revenue from the direct sale of cannabis to medical customers for a fixed price is recognized when the company transfers control of the good to the customer
New standards and interpretations issued but not yet adopted
IFRS 16 - Leases; in January 2016, the IASB issued IFRS 16, which specifies how an IFRS reporter will recognise, measure, present and disclose leases. The standard provides a single lessee accounting model, requiring lessees to recognise assets and liabilities for all leases unless the lease term is 12 months or less or the underlying asset has a low value. Lessors continue to classify leases as operating or finance, with IFRS 16’s approach to lessor accounting substantially unchanged from its predecessor, IAS 17. IFRS 16 is effective for annual reporting periods beginning on or after January 1, 2019, and a lessee shall either apply IFRS 16 with full retrospective effect or alternatively not restate comparative information but recognise the cumulative effect of initially applying IFRS 16 as an adjustment to opening equity at the date of initial application. Early adoption is permitted if IFRS 15 has also been adopted. Based on its current assets, interests and investments and review of existing lease arrangements, no significant impact is anticipated from the new standard.
| 9 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
There are no other standards that are not yet effective and that would be expected to have a material impact on the Company in the current or future reporting periods and on foreseeable future transactions.
The Company has reclassified certain immaterial items on the comparative consolidated statements of financial position, consolidated statements of income and comprehensive income, and consolidated statements of cash flows to improve clarity.
| 4. | Marketable securities |
Marketable securities are classified as fair value through profit or loss, and are comprised of:
| S&P rating at purchase | Interest rate |
Maturity date |
November 30, 2018 |
May 31, 2018 | ||
| Fixed Income: | ||||||
| Ford Motor Credit Co. LLC | BBB | 3.700% | 8/2/2018 | $ - | $ 1,015 | |
| Sobeys Inc. | BB+ | 3.520% | 8/8/2018 | - | 3,040 | |
| Canadian Western Bank | A- | 3.077% | 1/14/2019 | 1,519 | 1,528 | |
| Sun Life Financial Inc. | A | 2.770% | 5/13/2019 | 3,007 | 3,018 | |
| Ford Motor Credit Co. LLC | BBB | 3.140% | 6/14/2019 | 5,072 | 5,101 | |
| Canadian Western Bank | A- | 3.463% | 12/17/2019 | 1,020 | 1,025 | |
| Laurentian Bank of Canada | BBB | 2.500% | 1/23/2020 | - | 3,003 | |
| Enercare Solutions Inc. | BBB | 4.600% | 2/3/2020 | 3,930 | 3,974 | |
| Enbridge Inc. | BBB+ | 4.530% | 3/9/2020 | 5,145 | 5,203 | |
| Choice Properties REIT | BBB | 3.600% | 4/20/2020 | 5,045 | 5,091 | |
| Westcoast Energy Inc. | BBB+ | 4.570% | 7/2/2020 | - | 5,293 | |
| Citigroup Inc. (USD) | BBB+ | 2.050% | 12/17/2018 | 4,029 | 3,914 | |
| Royal Bank of Canada (USD) | AA- | 1.625% | 4/15/2019 | 3,980 | 3,857 | |
| $ 32,747 | $ 45,062 | |||||
The cost of marketable securities as at November 30, 2018 was $33,274 (May 31, 2018 - $45,863). During the three and six months ended November 30, 2018, the company divested of certain marketable securities for proceeds of $8,205 and $12,205 (2017 - $24,702 and $34,801), resulting in a loss on disposal of $91 and $146 (2017 - $256 and $387), and re-invested $nil and $nil (2017 - $nil and $5,000). During the three and six months ended November 30, 2018, the Company recognized a gain (loss) of $57 and $(110) (2017 - $55 and ($1,691)) on its marketable securities portfolio, of which $148 and $36 (2017 - $311 and ($1,304)) represented unrealized fair value adjustments.
| 5. | Other current assets |
Other current assets are comprised of:
| November 30, 2018 | May 31, 2018 | |||||||
| Sales tax receivable | $ | 13,198 | $ | 10,840 | ||||
| Accrued interest | 2,549 | 831 | ||||||
| Credit card receivable | 39 | 170 | ||||||
| Prepaid assets | 10,752 | 1,720 | ||||||
| Other | 2,162 | 823 | ||||||
| $ | 28,700 | $ | 14,384 | |||||
| 10 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
| 6. | Inventory |
Inventory is comprised of:
| Capitalized cost | Fair value adjustment | November 30, 2018 | May 31, 2018 | |||||||||||||
| Harvested cannabis | $ | 7,142 | $ | 8,355 | $ | 15,497 | $ | 12,331 | ||||||||
| Harvested cannabis trim | 1,593 | 1,605 | 3,198 | 2,277 | ||||||||||||
| Cannabis oil | 9,104 | 5,987 | 15,091 | 6,578 | ||||||||||||
| Softgel capsules | 136 | 112 | 248 | – | ||||||||||||
| Other inventory items | 6,308 | – | 6,308 | 964 | ||||||||||||
| $ | 24,283 | $ | 16,059 | $ | 40,342 | $ | 22,150 | |||||||||
During the three and six months ended November 30, 2018, the Company recorded $9,971 and $14,412 (2017 - $2,746 and $4,092) of production costs. Included in production costs for the three and six months ended November 30, 2018 is $1,047 and $1,194 of cannabis oil conversion costs (2017 - $54 and $95), $48 and $113 related to the cost of accessories (2017 - $61 and $98), and amortization of $1,020 and $1,533 (2017 - $500 and $889). During the three and six months ended November 30, 2018, the Company expensed $8,328 and $12,533 (2017 - $2,671 and $3,807) of fair value adjustments on the growth of its biological assets included in inventory sold.
During the three and six months ended November 30, 2018, the Company also disposed of nil and 13,642 plants prior to harvest. Included in production costs is $nil and $979 of accumulated costs relating to these plants which were not harvested.
The Company holds 4,229.8 kilograms of harvested cannabis (May 31, 2018 - 3,221.3 kgs), 1,057.9 kilograms of harvested cannabis trim (May 31, 2018 - 702.0 kgs), 19,079.7 litres of cannabis oils or 4,239.9 kilograms equivalent (May 31, 2018 - 7,724.7 litres or 1,716.6 kilograms equivalent), 278.3 litres of cannabis oils used in softgel capsules or 61.84 kilograms equivalent (May 31, 2018 - nil) at November 30, 2018.
| 7. | Biological assets |
Biological assets are comprised of:
| Amount | |||||
| Balance at May 31, 2018 | $ 7,331 | ||||
| Changes in fair value less costs to sell due to biological transformation | 13,665 | ||||
| Production costs capitalized | 14,469 | ||||
| Transferred to inventory upon harvest | (29,369) | ||||
| Balance at November 30, 2018 | $ 6,096 | ||||
The Company values medical cannabis plants at fair value. Management determined that cost approximates fair value from the date of initial clipping from mother plants until the fourth week prior to harvest. Measurement of the biological transformation of the plant at fair value less costs to sell begins in the fourth week prior to harvest and is recognized evenly until the point of harvest. The number of weeks in the growing cycle is between twelve and sixteen weeks from propagation to harvest. The Company has determined the fair value less costs to sell of harvested cannabis and harvested cannabis trim to be $3.50 and $2.75 per gram respectively (May 31, 2018 - $3.75 and $3.00 per gram), upon harvest for greenhouse produced cannabis and $4.00 and $3.25 per gram respectively (May 31, 2018 - $4.25 and $3.50 per gram), upon harvest for indoor produced cannabis.
The effect of the fair value less cost to sell over and above historical cost was an increase in non-cash value of biological assets and inventory of $4,154 and $13,665 during the three and six months ended November 30, 2018 (2017 - $3,115 and $7,380).
| 11 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
The fair value of biological assets is determined using a valuation model to estimate expected harvest yield per plant applied to the estimated price per gram less processing and selling costs. When there is a material change from the expected fair value used for cannabis, will the Company necessitate the fair value used in this calculation be adjusted. In this quarter, as a result of the newly enacted adult-use market along with the introduction of the excise duty tax, the Company determined a reduction of $0.25 per gram was warranted. The majority of the adult-use transactions are wholesale through provincial distribution agencies and as a result the net selling price and the selling costs are lower.
In determining the fair value of biological assets, management has made the following estimates in this valuation model:
| • | The harvest yield is between 40 grams and 80 grams per plant; |
| • | The selling price is between $3.00 and $7.00 per gram; |
| • | Processing costs include drying and curing, testing, post-harvest overhead allocation, packaging and labelling costs between $0.30 and $0.80 per gram; |
| • | Selling costs include shipping, order fulfilment, patient acquisition and patient maintenance costs between $0.00 and $1.50 per gram; |
Sales price used in the valuation of biological assets is based on the historical average selling price of all cannabis products and can vary based on different strains being grown as well as the proportion of sales derived from wholesale compared to retail. The low-end of the selling price, processing costs and selling costs are derived from historical wholesale sales, while the higher end prices and costs are from historical retail sales. Selling costs vary depending on methods of selling and are considered based on the expected method of selling and the determined additional costs which would be incurred. Expected yields for the cannabis plant is also subject to a variety of factors, such as strains being grown, length of growing cycle, and space allocated for growing. Management reviews all significant inputs, at each reporting period, based on historical information obtained as well as based on planned production schedules.
Management has quantified the sensitivity of the inputs and determined the following:
| • | Selling price per gram - a decrease in the average selling price per gram by 5% would result in the biological asset value decreasing by $318 (May 31, 2018 - $267) and inventory decreasing by $1,820 (May 31, 2018 - $1,040) |
| • | Harvest yield per plant - a decrease in the harvest yield per plant of 5% would result in the biological asset value decreasing by $155 (May 31, 2018 - $179) |
These inputs are level 3 on the fair value hierarchy, and are subject to volatility in market prices and several uncontrollable factors, which could significantly affect the fair value of biological assets in future periods.
| 8. | Related party transactions |
During the period, the Company disposed of its remaining shares in Liberty Health Sciences Inc. (“Liberty”) (note 13).
During the quarter, the Company appointed new board members. As a result of previous transactions, the Company held with related party corporations $900 in long-term investments for a U.S. legalization option (note 14), as at November 30, 2018.
The Company funded a portion of the Canadian operating costs of Liberty, for which Liberty reimburses the Company quarterly. Additionally, the Company purchases certain electrical generation equipment from and pays rent to a company owned by a director. These parties are related as they are corporations that are controlled by certain officers and directors of the Company.
During the three and six months ended November 30, 2018, related party corporations charged or incurred expenditures on behalf of the Company (including rent) totaling $53 and $138 (2017 - $54 and $93). Included in this amount was rent of $4 and $8 charged during the three and six months ended November 30, 2018 (2017 - $9 and $17).
| Amount | |||||
| Balance due to (from) related parties as at May 31, 2018 | $ - | ||||
| Related party charges in the period | 138 | ||||
| Payments to related parties in the period | (138) | ||||
| Payments made on behalf of related parties in the period | (830) | ||||
| Repayments made by related parties in the period | 830 | ||||
| Balance at November 30, 2018 | $ - | ||||
| 12 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
Key management personnel compensation for the six months ended November 30, 2018 and 2017 was comprised of:
| For the six months ended November 30, | ||||||||
| 2018 | 2017 | |||||||
| Salaries | $ | 1,677 | $ | 660 | ||||
| Short-term employment benefits (included in office and general) | 58 | 36 | ||||||
| Share-based compensation | 1,302 | 2,217 | ||||||
| $ | 3,037 | $ | 2,913 | |||||
Key management personnel compensation for the three months ended November 30, 2018 and 2017 was comprised of:
| For the three months ended November 30, | ||||||||
| 2018 | 2017 | |||||||
| Salaries | $ | 889 | $ | 354 | ||||
| Short-term employment benefits (included in office and general) | 31 | 18 | ||||||
| Share-based compensation | (678 | ) | 459 | |||||
| $ | 242 | $ | 831 | |||||
Directors and officers of the Company control 7.53% or 18,827,595 of the voting shares of the Company.
| 9. | Capital assets |
| Land | Production Facility | Equipment | Leasehold improvements | Construction in process | Total capital assets | ||||||||||||||||||||
| Cost | |||||||||||||||||||||||||
| At May 31, 2017 | $ | 10,829 | $ | 16,170 | $ | 5,340 | $ | 262 | $ | 42,159 | $ | 74,760 | |||||||||||||
| Business acquisitions | 854 | 6,992 | 2,860 | 1,388 | 5,947 | 18,041 | |||||||||||||||||||
| Additions | 12,716 | 47,149 | 4,759 | 15 | 151,899 | 216,538 | |||||||||||||||||||
| Transfers | 105 | 29,338 | 2,990 | – | (32,433 | ) | – | ||||||||||||||||||
| Disposals | – | (207 | ) | – | – | (415 | ) | (622 | ) | ||||||||||||||||
| At May 31, 2018 | 24,504 | 99,442 | 15,949 | 1,665 | 167,157 | 308,717 | |||||||||||||||||||
| Business acquisitions | 166 | 90 | 56 | 182 | – | 494 | |||||||||||||||||||
| Additions | 2,217 | 1,412 | 11,157 | 49 | 99,080 | 113,915 | |||||||||||||||||||
| Transfers | – | 1,737 | 1,247 | (1,389 | ) | (1,595 | ) | – | |||||||||||||||||
| At November 30, 2018 | $ | 26,887 | $ | 102,681 | $ | 28,409 | $ | 507 | $ | 264,642 | $ | 423,126 | |||||||||||||
| Accumulated depreciation | |||||||||||||||||||||||||
| At May 31, 2017 | $ | – | $ | 983 | $ | 1,260 | $ | 62 | $ | – | $ | 2,305 | |||||||||||||
| Amortization | – | 1,517 | 1,697 | 47 | – | 3,261 | |||||||||||||||||||
| At May 31, 2018 | – | 2,500 | 2,957 | 109 | – | 5,566 | |||||||||||||||||||
| Amortization | – | 1,587 | 1,995 | 15 | – | 3,597 | |||||||||||||||||||
| At November 30, 2018 | $ | – | $ | 4,087 | $ | 4,952 | $ | 124 | $ | – | $ | 9,163 | |||||||||||||
| Net book value | |||||||||||||||||||||||||
| At May 31, 2017 | $ | 10,829 | $ | 15,187 | $ | 4,080 | $ | 200 | $ | 42,159 | $ | 72,455 | |||||||||||||
| At May 31, 2018 | $ | 24,504 | $ | 96,942 | $ | 12,992 | $ | 1,556 | $ | 167,157 | $ | 303,151 | |||||||||||||
| At November 30, 2018 | $ | 26,887 | $ | 98,594 | $ | 23,457 | $ | 383 | $ | 264,642 | $ | 413,963 | |||||||||||||
| 13 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
| 10. | Intangible assets |
| Customer relationships | Corporate website | Licences, permits & applications | Non-compete agreements | Tokyo Smoke licensing agreement | Intellectual property, trademarks & brands | Total intangible assets | |||||||||||||||||||||||
| Cost | |||||||||||||||||||||||||||||
| At May 31, 2017 | $ | – | $ | 218 | $ | 1,250 | $ | – | $ | 459 | $ | 4,428 | $ | 6,355 | |||||||||||||||
| Business acquisitions | 11,730 | 39 | 137,920 | 1,930 | – | 76,190 | 227,809 | ||||||||||||||||||||||
| Additions | – | 152 | – | – | – | 9 | 161 | ||||||||||||||||||||||
| At May 31, 2018 | 11,730 | 409 | 139,170 | 1,930 | 459 | 80,627 | 234,325 | ||||||||||||||||||||||
| Business acquisitions | – | – | 191,066 | – | – | – | 191,066 | ||||||||||||||||||||||
| Additions | – | 79 | 11,703 | – | – | 1,205 | 12,987 | ||||||||||||||||||||||
| At November 30, 2018 | $ | 11,730 | $ | 488 | $ | 341,939 | $ | 1,930 | $ | 459 | $ | 81,832 | $ | 438,378 | |||||||||||||||
| Accumulated depreciation | |||||||||||||||||||||||||||||
| At May 31, 2017 | $ | – | $ | 156 | $ | 153 | $ | – | $ | – | $ | 4,155 | $ | 4,464 | |||||||||||||||
| Amortization | 1,274 | 100 | 124 | 314 | 92 | 1,513 | 3,417 | ||||||||||||||||||||||
| At May 31, 2018 | 1,274 | 256 | 277 | 314 | 92 | 5,668 | 7,881 | ||||||||||||||||||||||
| Amortization | 1,960 | 55 | 375 | 484 | 46 | 2,343 | 5,263 | ||||||||||||||||||||||
| At November 30, 2018 | $ | 3,234 | $ | 311 | $ | 652 | $ | 798 | $ | 138 | $ | 8,011 | $ | 13,144 | |||||||||||||||
| Net book value | |||||||||||||||||||||||||||||
| At May 31, 2017 | $ | – | $ | 62 | $ | 1,097 | $ | – | $ | 459 | $ | 273 | $ | 1,891 | |||||||||||||||
| At May 31, 2018 | $ | 10,456 | $ | 153 | $ | 138,893 | $ | 1,616 | $ | 367 | $ | 74,959 | $ | 226,444 | |||||||||||||||
| At November 30, 2018 | $ | 8,496 | $ | 177 | $ | 341,287 | $ | 1,132 | $ | 321 | $ | 73,821 | $ | 425,234 | |||||||||||||||
| 11. | Business Acquisitions |
Acquisition of Broken Coast Cannabis Ltd.
On February 13, 2018, the Company entered into a share purchase agreement to purchase all of the shares of Cannan Growers Inc. (“Cannan”), a holding company owning shares of Broken Coast Cannabis Ltd. (“Broken Coast”), and to acquire the remaining shares, for a combined total of 99.86%, of the issued and outstanding shares of Broken Coast. The combined purchase price was $214,168 satisfied through the issuance of an aggregate 14,373,675 common shares. The share purchase agreement entitled the Company to control Broken Coast effective on February 1, 2018, which became the effective acquisition date. In August 2018, the Company came to terms with the holder of the remaining 0.14% of the issued and outstanding shares of Broken Coast. In exchange for purchasing the remaining shares, the Company issued 19,963 shares to the holder.
| 14 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
The table below summarizes the fair value of the assets acquired and the liabilities assumed at the acquisition date:
| Note | Number of shares | Share price | Amount | |||||||||||||
| Consideration paid | ||||||||||||||||
| Shares issued | (i) | 14,393,638 | $ | 14.90 | $ | 214,465 | ||||||||||
| Total consideration paid | $ | 214,465 | ||||||||||||||
| Net assets acquired | ||||||||||||||||
| Current assets | ||||||||||||||||
| Cash and cash equivalents | 2,007 | |||||||||||||||
| Accounts receivable | 299 | |||||||||||||||
| Other current assets | 43 | |||||||||||||||
| Inventory | 2,572 | |||||||||||||||
| Biological assets | 826 | |||||||||||||||
| Long-term assets | ||||||||||||||||
| Capital assets | 13,298 | |||||||||||||||
| Customer relationships | 11,730 | |||||||||||||||
| Corporate website | 39 | |||||||||||||||
| Licences, permits & applications | 6,320 | |||||||||||||||
| Non-competition agreements | 1,930 | |||||||||||||||
| Intellectual property, trademarks & brands | 72,490 | |||||||||||||||
| Goodwill | 146,091 | |||||||||||||||
| Total assets | 257,645 | |||||||||||||||
| Current liabilities | ||||||||||||||||
| Accounts payable and accrued liabilities | 10,455 | |||||||||||||||
| Income taxes payable | 922 | |||||||||||||||
| Long-term liabilities | ||||||||||||||||
| Deferred tax liability | 25,889 | |||||||||||||||
| Long-term debt | 5,914 | |||||||||||||||
| Total liabilities | 43,180 | |||||||||||||||
| Total net assets acquired | $ | 214,465 | ||||||||||||||
| (i) | Share price based on the price of the shares on February 1, 2018. |
Net income and comprehensive net income for the Company would have been higher by approximately $567 and $1,134 for the three and six months ended November 30, 2017, if the acquisition had taken place on June 1, 2017. In connection with this transaction, the Company expensed transaction costs of $1,643.
Acquisition of Nuuvera Corp.
On March 23, 2018, the Company completed a definitive arrangement agreement (the “Arrangement Agreement”) pursuant to which the Company acquired, by way of a court-approved plan of arrangement, under the Business Corporations Act (Ontario) (the “Transaction”), 100% of the issued and outstanding common shares (on a fully diluted basis) of Nuuvera for a total consideration of $0.62 in cash plus 0.3546 of an Aphria share for each Nuuvera share held. All of Nuuvera’s outstanding options were exchanged for an equivalent option granted pursuant to Aphria’s stock option plan (each, a “Replacement Option”) to purchase from Aphria the number of common shares (rounded to the nearest whole share) equal to: (i) the exchange ratio multiplied by (ii) the number of Nuuvera shares subject to such Nuuvera Option. Each such Replacement Option shall provide for an exercise price per common share (rounded to the nearest whole cent) equal to: (i) the exercise price per Nuuvera share purchasable pursuant to such Nuuvera Option; divided by (ii) the exchange ratio.
| 15 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
The table below summarizes the fair value of the assets acquired and the liabilities assumed at the effective acquisition date:
| Note | Number of shares | Share price | Amount | |||||||||||||
| Consideration paid | ||||||||||||||||
| Cash | $ | 54,604 | ||||||||||||||
| Shares issued | (i) | 31,226,910 | $ | 13.17 | 411,258 | |||||||||||
| Warrants outstanding | (ii) | 1,345,866 | 1,015 | |||||||||||||
| Replacement options issued | (Ii) | 1,280,330 | 12,133 | |||||||||||||
| 479,010 | ||||||||||||||||
| Fair value of previously held investment | ||||||||||||||||
| Shares held by Aphria | (i) | 1,878,738 | $ | 14.92 | 28,028 | |||||||||||
| Warrants held by Aphria | (ii) | 322,365 | 243 | |||||||||||||
| 28,271 | ||||||||||||||||
| Total fair value of consideration | $ | 507,281 | ||||||||||||||
| Net assets acquired | ||||||||||||||||
| Current assets | ||||||||||||||||
| Cash and cash equivalents | 35,033 | |||||||||||||||
| Accounts receivable | 464 | |||||||||||||||
| Other current assets | 1,142 | |||||||||||||||
| Inventory | 401 | |||||||||||||||
| Long-term assets | ||||||||||||||||
| Capital assets | 4,743 | |||||||||||||||
| Intellectual property, trademarks & brands | 3,700 | |||||||||||||||
| Licences, permits & applications | 131,600 | |||||||||||||||
| Goodwilll | 377,221 | |||||||||||||||
| Total assets | 554,304 | |||||||||||||||
| Current liabilities | ||||||||||||||||
| Accounts payable and accrued liabilities | 11,000 | |||||||||||||||
| Long-term liabilities | ||||||||||||||||
| Deferred tax liability | 36,023 | |||||||||||||||
| Total liabilities | 47,023 | |||||||||||||||
| Total net assets acquired | $ | 507,281 | ||||||||||||||
| (i) | Share price based on the price of the shares on March 23, 2018; shares held by Aphria include the cash consideration paid. |
| (ii) | Options and warrants are valued using the Black-Scholes option pricing model using the following assumptions: the risk-free rate of 2.19%; expected life of 1- 10 years; volatility of 30% based on volatility used for similar instruments on the open market; forfeiture rate of nil; dividend yield of nil; and the exercise price of $2.52 - $20.30. |
Net income and comprehensive net income for the Company would have been lower by approximately $4,902 and $9,804 for the three and six months ended November 30, 2017, if the acquisition had taken place on June 1, 2017. In connection with this transaction, the Company expensed transaction costs of $3,439.
Acquisition of LATAM Holdings Inc.
On July 17, 2018, the Company signed a share purchase agreement with Scythian Biosciences Corp. (“Scythian”) to purchase 100% of the issued and outstanding shares of LATAM Holdings Inc. (“LATAM Holdings”); a direct wholly-owned subsidiary of Scythian. As outlined in the share purchase agreement, the negotiated purchase price was to be settled with the issuance of 15,678,310 shares of the Company valued on July 17, 2018 at $193,000 and the assumption of $1,000 short-term liabilities. The acquisition of LATAM Holdings closed on September 27, 2018. Therefore, in accordance with IFRS 3 - Business Combinations, the equity consideration transferred was measured at fair value at the acquisition date, which is the date control was obtained, which in this case was determined to be September 27, 2018. The fair value of the consideration shares on September 27, 2018 was $273,900.
| 16 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
LATAM Holdings, through other subsidiaries, provides the Company with access to the emerging cannabis markets in Latin America and the Caribbean. Through this acquisition, the Company has secured key licenses in Colombia, Argentina and Jamaica which is anticipated to provide substantial first mover advantage in these countries. In addition, the Company acquired an option and rights of first refusal to purchase a Brazilian incorporated entity, with the option and right of first refusal vesting only upon the entity obtaining a medical cannabis cultivation, processing and distribution license in Brazil.
The Company is in the process of assessing the fair value of the net assets acquired and, as a result, the fair value of the net assets acquired may be subject to adjustments pending completion of final valuations and post closing adjustments. The table below summarizes the preliminary estimated fair value of the assets acquired and the liabilities assumed at the effective acquisition date:
| Note | Number of shares | Share price | Amount | |||||||||||||
| Consideration paid | ||||||||||||||||
| Shares issued | (i) | 15,678,310 | $ | 17.47 | $ | 273,900 | ||||||||||
| Total consideration paid | $ | 273,900 | ||||||||||||||
| Net assets acquired | ||||||||||||||||
| Current assets | ||||||||||||||||
| Cash and cash equivalents | 2,704 | |||||||||||||||
| Accounts receivable | 571 | |||||||||||||||
| Other current assets | 106 | |||||||||||||||
| Inventory | 65 | |||||||||||||||
| Long-term assets | ||||||||||||||||
| Capital assets | 494 | |||||||||||||||
| Licences, permits & applications | 191,066 | |||||||||||||||
| Goodwill | 131,527 | |||||||||||||||
| Total assets | 326,533 | |||||||||||||||
| Current liabilities | ||||||||||||||||
| Accounts payable and accrued liabilities | 1,986 | |||||||||||||||
| Income taxes payable | 20 | |||||||||||||||
| Long-term liabilities | ||||||||||||||||
| Deferred tax liability | 50,627 | |||||||||||||||
| Total liabilities | 52,633 | |||||||||||||||
| Total net assets acquired | $ | 273,900 | ||||||||||||||
| (i) | Share price based on the price of the shares on September 27th, 2018. |
Net income and comprehensive net income for the Company would have been lower by approximately $1,139 and $2,278 for the three and six months ended November 30, 2017, if the acquisition had taken place on June 1, 2017. In connection with this transaction, the Company expensed transaction costs of $1,133.
Goodwill is comprised of:
| November 30, 2018 | May 31, 2018 | |||||||
| CannWay goodwill | $ | 1,200 | $ | 1,200 | ||||
| Broken Coast goodwill | 146,091 | 145,794 | ||||||
| Nuuvera goodwill | 377,221 | 375,768 | ||||||
| LATAM goodwill | 131,527 | – | ||||||
| $ | 656,039 | $ | 522,762 | |||||
| 17 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
| 12. | Convertible notes receivable |
| November 30, 2018 | May 31, 2018 | |||||||
| Copperstate Farms Investors, LLC | $ | – | $ | 1,942 | ||||
| HydRx Farms Ltd. (d/b/a Scientus Pharma) | 16,396 | 16,129 | ||||||
| Fire & Flower Inc. | 12,595 | – | ||||||
| 28,991 | 18,071 | |||||||
| Deduct - current portion | (16,396 | ) | (1,942 | ) | ||||
| $ | 12,595 | $ | 16,129 | |||||
Copperstate Farms Investors, LLC
As at November 30, 2018, this note was paid in full.
HydRx Farms Ltd. (d/b/a Scientus Pharma)
On August 14, 2017, Aphria purchased $11,500 in secured convertible debentures of Scientus Pharma (“SP”). The convertible debenture bears interest at 8%, paid semi-annually, matures in two years and includes the right to convert the debenture into common shares of SP at $2.75 per common share at any time before maturity. SP maintains the option of forced conversion of the convertible debenture if the common shares of SP trade on a stock exchange at a value of $3.02 or more for 30 consecutive days. The Company maintains a first charge on all assets of SP. In October 2018, the Company agreed to share its first charge on all assets of SP with a third party on a pari passu basis. As at November 30, 2018, the third party has not completed its investment.
During the three and six months ended November 30, 2018, the Company’s note receivable from SP increased by $112 and $267, representing the change in fair value on the note. As at November 30, 2018, the convertible note receivable totalled $16,396.
Fire & Flower Inc.
On July 26, 2018, Aphria purchased $10,000 in unsecured convertible debentures of Fire & Flower Inc. (“F&F”). The convertible debentures bear interest at 8% per annum compounded, accrued and paid semi-annually in arrears (the “Debentures”). The Debentures mature on the earlier of a public liquidity event or July 31, 2020, at which point, they automatically convert into common shares of F&F at the lower of $1.15 and the share price on July 31, 2020. The Debentures may also be converted into a loan on July 31, 2020 bearing interest at 12%, at the holder’s option.
During the three and six months ended November 30, 2018, the Company’s note receivable from F&F increased by $2,455 and $2,595, representing the change in fair value on the note. As at November 30, 2018, the convertible note receivable totalled $12,595.
Convertible notes receivable
During the period, the Company purchased a total of $10,000 in convertible notes. The unrealized gain on convertible notes receivable recognized in the results of operations amounts to $2,567 and $2,862 for the three and six months ended November 30, 2018 (2017 - $665 and $1,212).
The fair value was determined using the Black-Scholes option pricing model using the following assumptions: the risk-free rate of 0.85-1.51%; expected life of the convertible note; volatility of 70% based on comparable companies; forfeiture rate of nil; dividend yield of nil; and, the exercise price of the respective conversion feature.
| 18 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
| 13. | Interest in equity investees |
Summary of equity investees:
| November 30, 2018 | May 31, 2018 | |||||||
| Associated company | ||||||||
| Althea Company Pty Ltd. | $ | 9,612 | $ | 4,966 | ||||
| $ | 9,612 | $ | 4,966 | |||||
Summary of gain/loss from equity investees:
| For the three months ended November 30, | For the six months ended November 30, | |||||||||||||||
| 2018 | 2017 | 2018 | 2017 | |||||||||||||
| (Loss) gain on dilution of ownership in equity investee | $ | – | $ | (16 | ) | $ | 2,210 | $ | 7,535 | |||||||
| Loss from equity investees | (575 | ) | (441 | ) | (822 | ) | (9,281 | ) | ||||||||
| Gain on sale of equity investee | 47,471 | – | 57,351 | – | ||||||||||||
| $ | 46,896 | $ | (457 | ) | $ | 58,739 | $ | (1,746 | ) | |||||||
Liberty Health Sciences Inc.
In February 2018, the Company entered into a call/put obligation (“Obligation Agreement”) for the remaining shares held in Liberty, which were subject to CSE mandatory escrow requirements. As each new tranche of shares became freely trading, the Obligation Agreement resulted in the buyers acquiring the newly freely trading shares at an 18% discount to the market price of Liberty, based on Liberty’s 10 day volume weighted trading price.
The Obligation Agreement included an opt-out for Aphria’s benefit, in the event that the Toronto Stock Exchange amended their regulations such that it permitted investments by Canadian companies in U.S. based cannabis businesses, and in such instance, the Obligation Agreement would be automatically terminated. In exchange for the opt-out, the Company agreed to pay the buyers a $2,500 termination fee.
Based on the terms of the Obligation Agreement, the Company determined that the remaining shares held in Liberty met the requirements under IFRS 5 and were reclassified from interest in equity investees to assets held for sale. The Company ceased accounting for the investment as an equity investment as of November 30, 2017 and transferred the carrying value to assets held for sale.
In July 2018, 16,029,615 shares were released from escrow and sold as part of the Obligation Agreement. The Company received gross proceeds of $11,514 and recognized a gain on sale of equity investee of $9,880. As part of the transaction, the Company paid $480 in exchange for an option to buy back the shares at $1.00 a share, subject to certain downside risk protection which results in the purchaser sharing a portion of the difference between the share price on the day the option is exercised and the exercise price, provided the share price exceeds $1.25. The option to repurchase the shares is subject to the following conditions (collectively, the enumerated conditions (1) through (5), the “Conditions”):
| (1) | Cannabis becoming legalized federally in the United States; and |
One or more of the following conditions have been satisfied:
| (2) | The TSX has provided its approval for the purchase of the U.S. cannabis assets; |
| (3) | The TSX revises its rules such that it no longer has a prohibition against its listed companies having an interest in US assets which are involved in the cannabis business; |
| (4) | The common shares of the Company are voluntarily or involuntarily delisted from the TSX; and/or |
| (5) | The Company is acquired by another entity, provided that the common shares of the Company will be delisted from the TSX upon the change of control. |
This option has been included in long-term investments (Note 14).
| 19 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
During the period, the Company and the third party agreed to terminate the Obligation Agreement, in exchange for a $1,000 termination fee. The Company then entered into a share purchase agreement to divest of the remaining 64,118,462 Liberty shares in exchange for consideration in the form of a promissory note in the amount of $59,098, bearing interest at a rate of 12% due in 5 years (Note 15). As a security for the promissory note, the Liberty shares have been placed in trust with an escrow agent. The purchaser is able to remove the Liberty shares from the escrow at any time by paying off the promissory note. In the event that the Company enforces the security, the escrow agent will return the shares to the Company, provided that the Conditions are met. In the event they are not met, the escrow agent will transfer the securities to a third-party investment bank for liquidation, with the proceeds of liquidation delivered to the Company. Simultaneously with this sale, the Company entered into an option agreement to repurchase the Liberty shares for the amount of the promissory note (Note 14). The Company will pay an annual fee equal to 12.975% of the face value of the promissory note to maintain this option (Note 20). The option to repurchase the shares is subject to the Conditions described above. During the three and six months ended November 30, 2018, the Company reported a gain on sale of equity investee of $47,471 and $57,351.
During the three and six months ended November 30, 2017, the Company reported a total (loss) gain on dilution of ownership in equity investee of $(16) and $7,535. Prior to the Company no longer recording Liberty as an equity investee, Liberty reported a net loss of $23,493 and a net comprehensive loss of $27,001. In accordance with the equity method, the Company recorded a loss of $441 and $9,281 and other comprehensive gain (loss) of $520 and $(801).
Althea Company Pty Ltd. (“Althea”)
As at November 30, 2018 the Company held 50,750,000 common shares of Althea (May 31, 2018 - 4,500) representing an ownership interest of 25% (May 31, 2018 - 37.5%).
The following table summarizes, in aggregate, the financial information of the Company’s associate as included in their own financial statements.
| September 30, 2018 | March 31, 2018 | |||||
| Current assets | $ | 22,441 | $3,857 | |||
| Non-current assets | – | 3 | ||||
| Current liabilities | (141 | ) | (14) | |||
| Non-current liabilities | (13 | ) | - | |||
| Net assets | $ | 22,287 | $3,846 | |||
For the period from April 1 to September 30, 2018 the investee, Althea, reported a net loss of $2,327 AUD on its financial statements. In accordance with the equity method, the Company recorded a loss of $575 and $822 for the three and six months ended November 30, 2018 from its investee relative to its ownership of the outstanding common shares at the time.
During the period, Althea completed a share split of 7,500 shares for each existing share. Althea also issued 101,310,000 common shares for total proceeds of $19,650 AUD during the quarter. The Company participated in the financing of Althea contributing $3,400 AUD ($3,258 CAD) of the total $19,650 AUD raised. This additional raise reduced the Company’s ownership interest in Althea from 37.5% to 25% and accordingly, the Company recognized a gain on dilution of $2,210.
| November 30, 2018 | May 31, 2018 | |||||||
| Reconciliation to carrying amount: | ||||||||
| Opening balance | $ | 4,966 | $ | – | ||||
| Transfer from long-term investments | – | 2,483 | ||||||
| Cash contributions, net of share issuance costs | 3,258 | 2,497 | ||||||
| Gain on account of dilution of ownership | 2,210 | – | ||||||
| Share of reported net (loss) income | (822 | ) | (14 | ) | ||||
| Closing balance | $ | 9,612 | $ | 4,966 | ||||
| 20 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
| 14. | Long-term investments |
| Cost May 31, 2018 | Fair value May 31, 2018 | Investment | Divesture/ Transfer | Subtotal November 30, 2018 | Change in fair value | Fair value November 30, 2018 | ||||||||||||||||||||||
| Level 1 on fair value hierarchy | ||||||||||||||||||||||||||||
| CannaRoyalty Corp. | $ | 1,500 | $ | 3,765 | $ | – | $ | (3,765 | ) | $ | – | $ | – | $ | – | |||||||||||||
| MassRoots, Inc. | 304 | 164 | – | (164 | ) | – | – | – | ||||||||||||||||||||
| Tetra Bio-Pharma Inc. | 2,300 | 6,800 | 7,107 | – | 13,907 | 3,669 | 17,576 | |||||||||||||||||||||
| Hiku Brands Company Ltd. | 9,775 | 13,558 | – | (13,558 | ) | – | – | – | ||||||||||||||||||||
| Scythian Biosciences Corp. | 9,349 | 8,603 | 298 | (8,901 | ) | – | – | – | ||||||||||||||||||||
| National Access Cannabis Corp. | 1,093 | 710 | 10,481 | – | 11,191 | (4,384 | ) | 6,807 | ||||||||||||||||||||
| Emblem Corp. | – | – | 10,000 | – | 10,000 | (2,075 | ) | 7,925 | ||||||||||||||||||||
| 24,321 | 33,600 | 27,886 | (26,388 | ) | 35,098 | (2,790 | ) | 32,308 | ||||||||||||||||||||
| Level 2 on fair value hierarchy | ||||||||||||||||||||||||||||
| Hiku Brands Company Ltd. | 2,336 | 1,906 | 16,787 | (18,693 | ) | – | – | – | ||||||||||||||||||||
| Scythian Biosciences Corp. | 3,153 | 661 | – | (661 | ) | – | – | – | ||||||||||||||||||||
| 5,489 | 2,567 | 16,787 | (19,354 | ) | – | – | – | |||||||||||||||||||||
| Level 3 on fair value hierarchy | ||||||||||||||||||||||||||||
| Copperstate Farms, LLC | 1,755 | 5,300 | – | (5,300 | ) | – | – | – | ||||||||||||||||||||
| Copperstate Farms Investors, LLC | 9,407 | 14,700 | – | (14,700 | ) | – | – | – | ||||||||||||||||||||
| Resolve Digital Health Inc. | 718 | 3,300 | – | – | 3,300 | – | 3,300 | |||||||||||||||||||||
| Resolve Digital Health Inc. | 282 | 1,916 | – | – | 1,916 | 224 | 2,140 | |||||||||||||||||||||
| Green Acre Capital Fund I | 1,600 | 2,042 | 400 | – | 2,442 | 1,708 | 4,150 | |||||||||||||||||||||
| Green Acre Capital Fund II | – | – | 3,000 | – | 3,000 | (75 | ) | 2,925 | ||||||||||||||||||||
| Green Tank Holdings Corp. | 650 | 647 | – | – | 647 | 18 | 665 | |||||||||||||||||||||
| IBBZ Krankenhaus GmbH | 1,956 | 1,956 | – | – | 1,956 | 3 | 1,959 | |||||||||||||||||||||
| Greenwell Brands GmbH | – | – | 152 | – | 152 | – | 152 | |||||||||||||||||||||
| Rapid Dose Therapeutics Inc. | – | – | 5,400 | – | 5,400 | – | 5,400 | |||||||||||||||||||||
| HighArchy Ventures Ltd. | – | – | 9,995 | – | 9,995 | – | 9,995 | |||||||||||||||||||||
| Fire & Flower Inc. | – | – | 3,416 | – | 3,416 | – | 3,416 | |||||||||||||||||||||
| US legalization options | – | – | 54,762 | – | 54,762 | 40,863 | 95,625 | |||||||||||||||||||||
| 16,368 | 29,861 | 77,125 | (20,000 | ) | 86,986 | 42,741 | 129,727 | |||||||||||||||||||||
| Deduct - assets held for sale | (11,162 | ) | (20,000 | ) | – | 20,000 | – | – | – | |||||||||||||||||||
| $ | 35,016 | $ | 46,028 | $ | 121,798 | $ | (45,742 | ) | $ | 122,084 | $ | 39,951 | $ | 162,035 | ||||||||||||||
The fair value attached to warrants in both Level 2 and Level 3 were determined using the Black-Scholes option pricing model using the following assumptions: risk-free rate of 0.75-1.70% on the date of grant; expected life of 1 and 2 years; volatility of 70% based on comparable companies; forfeiture rate of 0%; dividend yield of nil; and, the exercise price of the respective warrant.
CannaRoyalty Corp. (“CR”)
During the period, the Company sold its remaining 750,000 shares of CR for proceeds of $4,111, resulting in an accounting gain of $346 (Note 28). The Company notes that as measured against the original cost of the shares, the Company recorded a total gain of $2,611 on its investment in CR.
MassRoots, Inc.
During the period, the Company sold its remaining 500,000 common shares in MassRoots, Inc. for proceeds of $1, resulting in a loss of $163 (Note 28).
Tetra Bio-Pharma Inc.
During the period, the Company purchased an additional 6,900,000 units of Tetra Bio-Pharma Inc. at a total cost of $7,107. Each unit is comprised of one Class A common share and one common share purchase warrant with an exercise price of $1.29 expiring November 2021. The Company owns 16,900,000 common shares and 6,900,000 warrants at a cost of $9,407, with a fair value of $17,576 as at November 30, 2018.
| 21 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
Hiku Brands Company Ltd. (“Hiku”)
During the period the Company exercised its 7,993,605 warrants at $2.10 per warrant. Subsequent to exercising its warrants the Company sold all of its shares of Hiku in exchange for $48,153, resulting in a gain on disposal of $15,902 (Note 28).
As part of the purchase of Hiku by a third party, the third party terminated the Hiku supply agreement with the Company. The third party purported to terminate the Tokyo Smoke supply agreement with the Company at approximately the same time. The Company does not believe that the agreement could be terminated by the third party and is pursuing all available legal options associated with the position taken by the third party.
Scythian Biosciences Inc. (“Scythian”)
During the period the Company purchased 123,800 common shares of Scythian at a total cost of $298. During the period, the Company sold its 2,812,300 common shares and 672,125 common share purchase warrants in Scythian in exchange for $6,609, resulting in a loss on disposal of $2,953 (Note 28).
National Access Cannabis Corp.
During the period, the Company purchased 10,344,505 common shares of National Access Cannabis Corp. at a total cost of $10,481. The Company owns 11,344,505 common shares in National Access Cannabis Corp. at a cost of $11,574, with a fair value of $6,807 as at November 30, 2018.
Emblem Corp. (“Emblem”)
During the period, the Company entered into a 5-year supply agreement with Emblem as part of the supply agreement the Company received 6,952,169 common shares to satisfy a deposit valued at $10,000. The Company owns 6,952,169 common shares in Emblem at a cost of $10,000, with a fair value of $7,925. The shares are subject to various hold restrictions tied to terms within the supply agreement.
US legalization options
During the period, the Company purchased an option to acquire 16,029,615 Liberty shares at $1.00 a share, expiring January 23, 2020. This option includes specific downside risk protection in which the purchaser will share a portion of the difference between the share price on the day the option is exercised and the exercise price, provided the share price exceeds $1.25. The cost of the option is $480. The option to repurchase the shares is subject to the Conditions described in note 13.
During the period, the Company entered into an option agreement to repurchase 64,118,462 Liberty shares in exchange for settlement of a promissory note receivable, expiring September 6, 2023 (Note 13 and Note 20). The cost of this option is a gross annual fee of $7,668, however the Company also receives $7,092 of interest income associated with the promissory note receivable, resulting in a net annual cost to the Company of $576. The option to repurchase the shares is subject to the Conditions described in note 13.
During the period, the Company contributed assets with a fair value of $55,000 to an investment fund. Simultaneously, the Company entered into an option agreement to purchase the Company’s proportionate share of the securities held in the investment fund at a cost of $55,000, expiring September 24, 2023 (Note 20). The cost of this option is a gross fee of $6,765, however the Company also receives $6,600 of interest income associated with the promissory note receivable, resulting in a net annual cost to the Company of $165. In the event the securities in the fund represent direct or indirect ownership of, or investment in, entities engaging in activities related to the cultivation, distribution or possession of cannabis in the United States, the option to purchase the securities is subject to the Conditions described in note 13.
The Company used the Black-Scholes option pricing model to determine the fair value of the options using the following assumptions: risk-free rate of 2.19-2.19%; expected life of 1.15 - 5 years; volatility of 70-90% based on comparable companies; forfeiture rate of 0%; dividend yield of nil; and, the exercise price of the respective option. The Company discounted the determined Black-Scholes value by 11.60% for the options expiring in January 2020, and 70.25% for the options expiring in September 2023, as the probability the Conditions described in note 13 will be met.
Copperstate Farms, LLC (“Copperstate”) and Copperstate Farms Investors, LLC (“CSF”)
During the period, the Company received the $20,000 from the sale of the shares of Copperstate and CSF, which were previously held as available for sale.
| 22 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
Resolve Digital Health Inc. (“Resolve”)
The Company owns 2,200,026 common shares and 2,200,026 warrants in Resolve at a total cost of $1,000, with a fair value of $5,440 as at November 30, 2018. The Company determined the fair value of its investment based on Resolve’s most recent financing. Each warrant is exercisable at $0.65 per warrant expiring December 1, 2021.
Green Acre Capital Fund I
The Company committed $2,000 to Green Acre Capital Fund I and, as of the balance sheet date, has funded the full $2,000. The Company determined the fair value of its investment, based on its proportionate share of net assets, to be $4,150 as at November 30, 2018. During the period, the Company received a dividend of $700 from its investment in Green Acre Capital Fund I.
Green Acre Capital Fund II
During the period, the Company committed to a $15,000 investment in Green Acre Capital Fund II, and as of the balance sheet date, has funded $3,000. The Company determined that the fair value of its investments, based on its proportionate share of net assets, was $2,925 as at November 30, 2018.
Green Tank Holdings Corp. (“Green Tank”)
The Company owns 98,425 preferred shares in Green Tank for a total cost of $500 USD ($650 CAD). The Company determined the fair value of its investment, based on Green Tank’s most recent financing at the same price, is equal to its carrying value. The Company recognized a gain from the change in fair value of $18 due to changes in the foreign exchange rate.
IBBZ Krankenhaus GmbH Klinik Hygiea (“Krankenhaus”)
The Company owns 25.1% of Krankenhaus, which is the owner and operator of Berlin-based Schöneberg Hospital, for €1,294 ($1,956 CAD). Through this investment, the Company is entitled to 5% of the net income (loss) for the years 2018 to 2021, and 10% of the net income (loss) for the period thereafter. The Company determined that the fair value of its investment, based on Krankenhaus’ most recent financing at the same price, is equal to its carrying value. The Company recognized a gain from the change in fair value of $3 due to changes in the foreign exchange rate.
Greenwell Brands GmbH (“Greenwell”)
In September 2018, the Company entered into an investment and shareholder agreement with Greenwell for the purchase of 1,250 common shares, for a total cost of €100 ($152CAD). The Company determined that the fair value of its investment, based on the most recent financing at the same price, is equal to its carrying value.
Rapid Dose Therapeutics Inc. (“RDT”)
In August 2018, the Company entered into a subscription agreement with RDT for the purchase of 7,200,000 common shares, for a total cost of $5,400. The Company determined that the fair value of its investment, based on the most recent financing at the same price, is equal to its carrying value. Subsequent to quarter-end, RDT’s common shares began trading on the Canadian Stock Exchange. The Company will reclassify this investment to Level 1 beginning next quarter.
HighArchy Ventures Ltd.
In October 2018, the Company entered into a subscription agreement with HighArchy Ventures Ltd. for the purchase of 1,999 Class A shares and 1,999 Class B shares, for a total cost of $9,995. The Company determined that the fair value of its investment, based on the most recent financing at the same price, is equal to its carrying value.
Fire & Flower Inc.
In October 2018, the Company entered into a subscription agreement with Fire & Flower Inc. for the purchase of 2,277,000 common shares, for a total cost of $3,416. The Company determined that the fair value of its investment, based on the most recent financing at the same price, is equal to its carrying value.
| 23 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
| 15. | Promissory note receivable |
| May 31, 2018 |
Additions | Payments | November 30, 2018 | ||
| September 6, 2018 - $59,098 - 12%, due | $ - | $ 59,098 | $ - | $ 59,098 | |
| September 6, 2023 | |||||
| September 24, 2018 - $55,000 - 12%, due | - | 55,000 | - | 55,000 | |
| September 24, 2023 | |||||
| October 11, 2018 - GBP £4,600 - 3.25%, due | - | 7,952 | - | 7,952 | |
| October 11, 2021 | |||||
| October 31, 2018 - $6,609 - 12%, due | - | 6,609 | - | 6,609 | |
| October 31, 2023 | |||||
| November 1, 2018 - $200 - interest free, due | - | 200 | - | 200 | |
| May 1, 2020 | |||||
| $ - | $ 128,859 | $ - | $ 128,859 | ||
| 16. | Income taxes and deferred income taxes |
A reconciliation of income taxes at the statutory rate with the reported taxes is as follows:
| For the six months ended November 30, | ||||||||
| 2018 | 2017 | |||||||
| Income before income taxes | $ | 87,814 | $ | 25,563 | ||||
| Statutory rate | 26.5 | % | 26.5 | % | ||||
| Expected income tax expense at combined basic federal and provincial tax rate | 23,271 | 6,774 | ||||||
| Effect on income taxes of: | ||||||||
| Foreign tax differential | (174 | ) | – | |||||
| Permanent differences | 1,055 | – | ||||||
| Non-deductible share-based compensation and other expenses | 2,717 | 1,267 | ||||||
| Non-taxable portion of losses (gains) | (15,035 | ) | (3,677 | ) | ||||
| Other | (104 | ) | (297 | ) | ||||
| Tax assets not recognized | 134 | – | ||||||
| $ | 11,864 | $ | 4,067 | |||||
| Income tax expense is comprised of: | ||||||||
| Current | $ | 8,079 | $ | 851 | ||||
| Future | 3,785 | 3,216 | ||||||
| $ | 11,864 | $ | 4,067 | |||||
| 24 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
The following table summarizes the components of deferred tax:
| November 30, 2018 | May 31, 2018 | |||||||
| Deferred tax assets | ||||||||
| Non-capital loss carry forward | $ | 7,137 | $ | 4,567 | ||||
| Capital loss carry forward | – | 405 | ||||||
| Share issuance and financing fees | 7,730 | 5,443 | ||||||
| Unrealized loss | – | 916 | ||||||
| Other | 941 | 27 | ||||||
| Deferred tax liabilities | ||||||||
| Net book value in excess of undepreciated capital cost | (719 | ) | (1,017 | ) | ||||
| Intangible assets in excess of tax costs | (113,767 | ) | (64,120 | ) | ||||
| Unrealized gain | (6,695 | ) | (1,097 | ) | ||||
| Biological assets and inventory in excess of tax costs | (4,866 | ) | (4,377 | ) | ||||
| Net deferred tax (liabilities) assets | $ | (110,239 | ) | $ | (59,253 | ) | ||
| 17. | Bank indebtedness |
The Company secured an operating line of credit in the amount of $1,000 which bears interest at the lender’s prime rate plus 75 basis points. As of the November 30, 2018, the Company has not drawn on the line of credit. The operating line of credit is secured by a first charge on the property at 265 Talbot St. West, Leamington, Ontario and a first ranking position on a general security agreement.
| 18. | Promissory note payable |
During the prior year, the Company entered into a promissory note with Althea for $700 AUD ($686), as part of the purchase of Althea common shares. The note is due and payable on December 31, 2020. The Company reached an agreement with Althea where the promissory note amount will be used by Althea to purchase products from the Company in connection with a supply agreement entered into in September 2017.
| November 30, 2018 |
May 31, 2018 | ||||
| Note payable to Althea Company Pty Ltd - $700 AUD ($686), opening balance, | $ 610 | $ 686 | |||
| non-interest bearing, due and payable on December 31, 2020 | |||||
| Reduction of Promissory note payable balance with respect to products provided | - | (63) | |||
| Foreign exchange (gain) loss | (6) | (13) | |||
| Balance remaining | 604 | 610 | |||
| Deduct - principal portion included in current liabilities | (604) | (610) | |||
| $ - | $ - | ||||
| 25 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
| 19. | Long-term debt |
| November 30, 2018 | May 31, 2018 | |||||||
| Term loan - $25,000 - Canadian Five Year Bond interest rate plus 2.73% with a minimum 4.50%, 5 year term, with a 15-year amortization, repayable in blended monthly payments sufficient to repay the loan by July 2033 | $ | 24,618 | $ | – | ||||
| Term loan - $25,000 - 3.95%, compounded monthly, 5 year term with a 15-year amortization, repayable in equal monthly instalments of $188 including interest, due in April 2022 | 24,023 | 24,107 | ||||||
| Term loan - $1,250 - 3.99%, 5-year term, with a 10-year amortization, repayable in equal monthly instalments of $13 including interest, due in July 2021 | 1,002 | 1,057 | ||||||
| Mortgage payable - $3,750 - 3.95%, 5-year term, with a 20-year amortization, repayable in equal monthly instalments of $23 including interest, due in July 2021 | 3,449 | 3,515 | ||||||
| Vendor take-back mortgage owed to related party - $2,850 - 6.75%, 5-year term, repayable in equal monthly instalments of $56 including interest, due in June 2021 | 1,593 | 1,869 | ||||||
| 54,685 | 30,548 | |||||||
| Deduct - unamortized financing fees | (132 | ) | (71 | ) | ||||
- principal portion included in current liabilities | (3,388 | ) | (2,140 | ) | ||||
| $ | 51,165 | $ | 28,337 | |||||
Total long-term debt repayments are as follows:
| Next 12 months | $ 3,388 | ||||
| 2 years | 3,547 | ||||
| 3 years | 3,434 | ||||
| 4 years | 3,182 | ||||
| Thereafter | 41,134 | ||||
| Balance of obligation | $ 54,685 |
The term loan of $24,618 was entered into on July 27, 2018 and is secured by a first charge on the property at 223, 231, 239, 265, 269, 271 and 275 Talbot Street West, Leamington Ontario, a first position on a general security agreement, and an assignment of fire insurance to the lender. Principal payments started on the term loan in August 2018. The effective interest rate during the period was 4.68%.
The term loan of $24,023 was entered into on May 9, 2017 and is secured by a first charge on the property at 265 Talbot Street West, Leamington Ontario, a first position on a general security agreement, and an assignment of fire insurance to the lender. Principal payments started on the term loan in March 2018.
The term loan of $1,002 and mortgage payable of $3,449 were entered into on July 22, 2016 and are secured by a first charge on the property at 265 Talbot Street West, Leamington, Ontario and a first position on a general security agreement.
The vendor take-back mortgage payable of $1,592, owed to a director of the Company, was entered into on June 30, 2016 in conjunction with the acquisition of the property at 265 Talbot Street West. The mortgage is secured by a second charge on the property at 265 Talbot Street West, Leamington, Ontario.
The Company acquired term loans of $3,000 and $1,201, and a mortgage payable of $1,713 as part of the acquisition of Broken Coast (Note 11). These loans and mortgages were paid in full during the prior year.
| 26 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
| 20. | Option payment liability |
| May 31, 2018 | Additions | Settlement | Change in fair value | November 30, 2018 | ||||||||||||||||
| US legalization options | $ | – | $ | 58,271 | $ | (14,433 | ) | $ | 538 | $ | 44,376 | |||||||||
| Deduct - current portion | – | (11,654 | ) | |||||||||||||||||
| $ | – | $ | 32,722 | |||||||||||||||||
During the period, the Company entered into an option agreement to repurchase 64,118,462 Liberty shares in exchange for settlement of a promissory note receivable, expiring September 6, 2023 (Note 13 and Note 14). The cost of this option is an annual fee of $7,668 paid at the beginning of each year; however, the Company also receives $7,092 of interest income associated with the promissory note receivable, resulting in a net annual cost to the Company of $576. The fair value of the payments from this contract was $30,958 at inception, and $23,660 as at November 30, 2018, using a discount rate of 12%.
During the period, the Company entered into an option agreement to repurchase the Company’s proportionate share of securities held by an investment fund at a cost of $55,000, expiring September 24, 2023 (Note 14). The cost of this option is an annual fee of $6,765 paid at the beginning of each year; however, the Company also receives $6,600 of interest income associated with the promissory note receivable, resulting in a net annual cost to the Company of $165. The fair value of the payments from this contract was $27,313 at inception, and $20,716 as at November 30, 2018, using a discount rate of 12%.
| 21. | Share capital |
The Company is authorized to issue an unlimited number of common shares. As at November 30, 2018, the Company has 249,931,744 shares issued and outstanding and 19,963 shares to be issued, of which 1,777,971 shares were held and subject to various escrow agreements.
| Common Shares | Number of shares | Amount | ||||||
| Balance at May 31, 2018 | 210,169,924 | $ | 1,113,981 | |||||
| June 2018 bought deal, net of cash issuance costs | 21,835,510 | 245,925 | ||||||
| Broken Coast acquisition | 19,963 | 297 | ||||||
| LATAM acquisition | 15,678,310 | 273,900 | ||||||
| Warrants exercised | 316,063 | 1,409 | ||||||
| Options exercised | 1,911,974 | 11,139 | ||||||
| Income tax recovery on share issuance costs | – | 3,426 | ||||||
| 249,931,744 | $ | 1,650,077 | ||||||
| a) | Throughout the period, 316,063 warrants with exercise prices ranging from $1.50 to $20.30 were exercised for a value of $1,409 including any cash consideration. |
| b) | Throughout the period, 1,911,974 shares were issued from the exercise of stock options with exercise prices ranging from $0.60 to $20.19 for a value of $11,139, including any cash consideration. |
| c) | In June 2018, the Company closed a bought deal financing in which it issued 21,835,510 common shares at a purchase price of $11.85 per share for $245,925 net of cash issuance costs. |
| d) | During the period, the Company agreed to terms to acquire the remaining 0.14% of Broken Coast (Note 11) and accordingly, the Company agreed to issue 19,963 shares. |
| e) | In September 2018, the Company completed the acquisition of LATAM (note 11) in which it issued 15,678,310 common shares at a purchase price of $17.47 per share for $273,900. |
| f) | During the period, the Company recognized a $3,426 income tax recovery on share issuance costs. |
| 27 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
| 22. | Warrants |
The warrant details of the Company are as follows:
| Type of warrant | Expiry date | Number of warrants | Weighted average price | Amount | ||||||||||
| Warrant | December 11, 2018 | 7,003 | 1.75 | $ | – | |||||||||
| Warrant | December 2, 2019 | 1,026,269 | 1.50 | – | ||||||||||
| Warrant | September 26, 2021 | 200,000 | 3.14 | 360 | ||||||||||
| Nuuvera warrant | February 14, 2020 | 1,293,803 | 20.30 | 976 | ||||||||||
| 2,527,075 | $ | 11.26 | $ | 1,336 | ||||||||||
| November 30, 2018 | May 31, 2018 | ||||||||||||||
| Number of warrants | Weighted average price | Number of warrants | Weighted average price | ||||||||||||
| Outstanding, beginning of the period | 2,843,138 | $ | 10.52 | 3,885,908 | $1.61 | ||||||||||
| Issued during the period | – | – | 1,345,866 | 20.30 | |||||||||||
| Exercised during the period | (316,063 | ) | 4.62 | (2,388,636 | ) | 1.54 | |||||||||
| Outstanding, end of the period | 2,527,075 | $ | 11.25 | 2,843,138 | $10.52 | ||||||||||
In March 2018, the Company completed the acquisition of Nuuvera (Note 11) in which it reserved 1,345,866 common shares for issuance to the holders of certain common share purchase warrants of Nuuvera (“Nuuvera Warrants”). There are 3,795,450 Nuuvera Warrants, exercisable for Nuuvera shares at an exercise price of $7.20 per share, the Nuuvera shares would convert to 0.3546 Aphria shares and $0.62 cash.
| 23. | Stock options |
The Company adopted a stock option plan under which it is authorized to grant options to officers, directors, employees and consultants enabling them to acquire common shares of the Company. The maximum number of common shares reserved for issuance of stock options that can be granted under the plan is 10% of the issued and outstanding common shares of the Company. The options granted can be exercised for up to a maximum of 10 years and vest as determined by the Board of Directors. The exercise price of each option can not be less than the market price of the common shares on the date of grant.
The Company recognized a share-based compensation expense of $3,910 and $8,085 during the three and six months ended November 30, 2018 (2017 - $2,055 and $4,495). The total fair value of options granted during the period was $10,884 (2017 - $6,091).
| November 30, 2018 | May 31, 2018 | |||||||||||||||
Number of options | Weighted average price | Number of options | Weighted average price | |||||||||||||
| Outstanding, beginning of the period | 8,956,195 | $ | 7.60 | 5,926,001 | $1.99 | |||||||||||
| Exercised during the period | (2,127,001 | ) | 3.72 | (2,637,363 | ) | 2.30 | ||||||||||
| Issued during the period | 1,400,000 | 13.61 | 6,703,330 | 11.12 | ||||||||||||
| Cancelled during the period | (96,037 | ) | 8.43 | (1,035,773 | ) | 11.77 | ||||||||||
| Outstanding, end of the period | 8,133,157 | $ | 9.63 | 8,956,195 | $7.60 | |||||||||||
| Exercisable, end of the period | 4,204,447 | $ | 6.63 | 3,919,542 | $1.36 | |||||||||||
In June 2018, the Company issued 250,000 stock options at an exercise price of $11.78 per share, exercisable for 3 years to officers of the Company. 83,331 vested immediately and the remainder vest over 2 years.
In July 2018, the Company issued 820,000 stock options at an exercise price between $11.51 and $11.85 per share, exercisable for 5 years to employees of the Company. 50,000 vested immediately and the remainder vest over 3 years.
| 28 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
In September 2018, the Company issued 250,000 stock options at an exercise price of $19.38, exercisable for 5 years to employees of the Company. Nil vested immediately and the remainder vest over 3 years.
In October 2018, the Company issued 80,000 stock options at an exercise price of $19.70, exercisable for 5 years to employees of the Company. Nil vested immediately and the remainder vest over 3 years.
The outstanding option details of the Company are as follows:
| Expiry date | Weighted average exercise price | Number of options | Vested and exercisable | |||||||||
| February 2019 | $ | 11.40 | 200,000 | – | ||||||||
| April 2019 | $ | 1.67 | 30,000 | 30,000 | ||||||||
| June 2019 | $ | 0.60 | 400,000 | 400,000 | ||||||||
| September 2019 | $ | 3.00 | 42,365 | 42,365 | ||||||||
| October 2019 | $ | 3.47 | 7,400 | 7,400 | ||||||||
| November 2019 | $ | 3.90 | 817,019 | 817,019 | ||||||||
| December 2019 | $ | 5.25 | 400,000 | 33,333 | ||||||||
| January 2020 | $ | 5.72 | 19,168 | 4,167 | ||||||||
| April 2020 | $ | 7.92 | 90,000 | 56,666 | ||||||||
| June 2020 | $ | 5.44 | 216,668 | 133,333 | ||||||||
| July 2020 | $ | 5.24 | 698,645 | 552,964 | ||||||||
| September 2020 | $ | 0.85 | 185,000 | 185,000 | ||||||||
| October 2020 | $ | 6.90 | 360,000 | 223,332 | ||||||||
| November 2020 | $ | 9.05 | 250,000 | 156,666 | ||||||||
| November 2020 | $ | 9.28 | 50,000 | 33,333 | ||||||||
| December 2020 | $ | 14.06 | 100,000 | 33,333 | ||||||||
| January 2021 | $ | 21.70 | 10,000 | 3,333 | ||||||||
| January 2021 | $ | 22.89 | 150,000 | 43,330 | ||||||||
| January 2021 | $ | 22.08 | 50,000 | 16,666 | ||||||||
| March 2021 | $ | 14.39 | 30,000 | 16,666 | ||||||||
| March 2021 | $ | 11.40 | 300,000 | 100,000 | ||||||||
| March 2021 | $ | 9.98 | 200,000 | 66,666 | ||||||||
| March 2021 | $ | 12.39 | 50,000 | 16,666 | ||||||||
| April 2021 | $ | 11.40 | 423,334 | 186,664 | ||||||||
| April 2021 | $ | 11.45 | 100,000 | 33,333 | ||||||||
| May 2021 | $ | 20.19 | 908,500 | 241,832 | ||||||||
| June 2021 | $ | 1.40 | 191,669 | 191,669 | ||||||||
| June 2021 | $ | 11.78 | 250,000 | 83,331 | ||||||||
| August 2021 | $ | 1.64 | 110,000 | 89,991 | ||||||||
| October 2022 | $ | 6.90 | 74,000 | 74,000 | ||||||||
| July 2023 | $ | 11.51 | 100,000 | – | ||||||||
| July 2023 | $ | 11.85 | 708,000 | 50,000 | ||||||||
| September 2023 | $ | 19.38 | 250,000 | – | ||||||||
| October 2023 | $ | 19.70 | 80,000 | – | ||||||||
| July 2027 | $ | 2.52 | 82,427 | 82,427 | ||||||||
| November 2027 | $ | 6.29 | 39,792 | 39,792 | ||||||||
| March 2028 | $ | 12.29 | 119,378 | 119,378 | ||||||||
| March 2028 | $ | 14.38 | 39,792 | 39,792 | ||||||||
| Outstanding, end of the period | $ | 9.63 | 8,133,157 | 4,204,447 | ||||||||
| 29 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
The Company used the Black-Scholes option pricing model to determine the fair value of options granted using the following assumptions: risk-free rate of 2.00-2.08% on the date of grant; expected life of 3 - 5 years; volatility of 70% based on comparable companies; forfeiture rate of 0%; dividend yield of nil; and, the exercise price of the respective option.
| 24. | Non-controlling interest |
The following tables summarise the information relating to the Company’s subsidiaries, 1974568 Ontario Ltd. (“Aphria Diamond”), CannInvest Africa Ltd., Verve Dynamics Incorporated (Pty) Ltd. (“Verve Dynamics”), Nuuvera Malta Ltd., Marigold Projects Jamaica Limited (“Marigold”), and ColCanna S.A.S. before intercompany eliminations.
| Aphria Diamond | CannInvest Africa Ltd. | Verve Dynamics | Nuuvera Malta Ltd. | Marigold | ColCanna S.A.S. | November 30, 2018 | ||||||||||||||||||||||
| Current assets | $ | 8,089 | $ | 34 | $ | – | $ | 2,506 | $ | 193 | $ | 8,995 | $ | 19,817 | ||||||||||||||
| Non-current assets | 139,312 | 3 | 13,503 | 448 | 337 | 298 | 153,901 | |||||||||||||||||||||
| Current liabilities | (8,109 | ) | (43 | ) | – | (270 | ) | – | (91 | ) | (8,513 | ) | ||||||||||||||||
| Non-current liabilities | (120,398 | ) | – | – | (3,056 | ) | (1,067 | ) | (9,509 | ) | (134,030 | ) | ||||||||||||||||
| Net assets | 18,894 | (6 | ) | 13,503 | (372 | ) | (537 | ) | (307 | ) | 31,175 | |||||||||||||||||
| Non-controlling interest % | 49 | % | 50 | % | 70 | % | 10 | % | 5 | % | 10 | % | ||||||||||||||||
| Non-controlling interest | $ | 9,258 | $ | (3 | ) | $ | 9,452 | $ | (37 | ) | $ | (27 | ) | $ | (31 | ) | $ | 18,612 | ||||||||||
| Aphria Diamond | CannInvest Africa Ltd. | Verve Dynamics | Nuuvera Malta Ltd. | Marigold | ColCanna S.A.S. | November 30, 2018 | ||||||||||||||||||||||
| Revenue | $ | – | $ | – | $ | – | $ | 141 | $ | – | $ | 2 | $ | 143 | ||||||||||||||
| Total expenses | 657 | 7 | – | 514 | 358 | 257 | 1,793 | |||||||||||||||||||||
| Non-controlling interest % | 49 | % | 50 | % | 70 | % | 10 | % | 5 | % | 10 | % | ||||||||||||||||
| $ | (322 | ) | $ | (4 | ) | $ | – | $ | (37 | ) | $ | (18 | ) | $ | (26 | ) | $ | (407 | ) | |||||||||
| Aphria Diamond | CannInvest Africa Ltd. | Verve Dynamics | Nuuvera Malta Ltd. | Marigold | ColCanna S.A.S. | May 31, 2018 | ||||||||||||||||||||||
| Current assets | $ | 7,313 | $ | – | $ | – | $ | – | $ | – | $ | – | $ | 7,313 | ||||||||||||||
| Non-current assets | 83,207 | – | – | – | – | – | 83,207 | |||||||||||||||||||||
| Current liabilities | (10,085 | ) | – | – | – | – | – | (10,085 | ) | |||||||||||||||||||
| Non-current liabilities | (60,884 | ) | – | – | – | – | – | (60,884 | ) | |||||||||||||||||||
| Net assets | 19,551 | – | – | – | – | – | 19,551 | |||||||||||||||||||||
| Non-controlling interest % | 49 | % | 0 | % | 0 | % | 0 | % | 0 | % | 0 | % | ||||||||||||||||
| Non-controlling interest | $ | 9,580 | $ | – | $ | – | $ | – | $ | – | $ | – | $ | 9,580 | ||||||||||||||
| 30 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
| 25. | General and administrative expenses |
| For the three months ended November 30, | For the six months ended November 30, | |||||||||||||||
| 2018 | 2017 | 2018 | 2017 | |||||||||||||
| Executive compensation | $ | 846 | $ | 354 | 1,681 | $ | 660 | |||||||||
| Consulting fees | 1,427 | 63 | 2,358 | 158 | ||||||||||||
| Office and general | 5,660 | 567 | 7,467 | 1,119 | ||||||||||||
| Professional fees | 485 | 480 | 2,047 | 697 | ||||||||||||
| Salaries and wages | 3,019 | 317 | 6,111 | 725 | ||||||||||||
| Travel and accommodation | 689 | 168 | 1,160 | 304 | ||||||||||||
| Rent | 150 | 24 | 303 | 45 | ||||||||||||
| $ | 12,276 | $ | 1,973 | $ | 21,127 | $ | 3,708 | |||||||||
| 26. | Share-based compensation |
Share-based compensation is comprised of:
| For the three months ended November 30, |
For the six months ended November 30, | |||||
| 2018 | 2017 | 2018 | 2017 | |||
| Amounts charged to share-based payment reserve in | $ 3,910 | $ 2,055 | $ 8,085 | $ 4,495 | ||
| respect of share-based compensation | ||||||
| Share-based compensation accrued in the prior period | - | - | - | (44) | ||
| Share-based compensation issued on behalf of a | - | - | - | (32) | ||
| related party | ||||||
| Shares for services compensation | - | 74 | - | 187 | ||
| Deferred share units expensed in the period | (1,336) | 71 | 611 | 103 | ||
| $ 2,574 | $ 2,200 | $ 8,696 | $ 4,709 | |||
During the period, the Company issued 18,925 deferred share units to certain directors of the Company under the terms of the Company’s Omnibus Long-Term Incentive Plan. In May 2018, directors and officers of the Company forfeited 312,000 deferred share units which were granted during the prior year.
As at November 30, 2018, the Company had 219,482 deferred share units outstanding.
| 27. | Finance income, net |
Finance income, net, is comprised of:
| For the three months ended November 30, | For the six months ended November 30, | |||||||||||||||
| 2018 | 2017 | 2018 | 2017 | |||||||||||||
| Interest income | $ | 5,603 | $ | 1,193 | $ | 7,095 | $ | 1,995 | ||||||||
| Interest expense | (748 | ) | (331 | ) | (1,181 | ) | (667 | ) | ||||||||
| $ | 4,855 | $ | 862 | $ | 5,914 | $ | 1,328 | |||||||||
| 31 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
| 28. | Gain on long-term investments |
Gain on long-term investments for the three and six months ended November 30, 2018 is comprised of:
| Investment | Proceeds | Opening fair value / cost | Gain (loss) on disposal | Change in fair value | Total | |||||||||||||||
| Level 1 on fair value hierarchy | ||||||||||||||||||||
| CannaRoyalty Corp. - shares | $ | 4,111 | $ | 3,765 | $ | 346 | $ | – | $ | 346 | ||||||||||
| MassRoots, Inc. - shares | 1 | 164 | (163 | ) | – | (163 | ) | |||||||||||||
| North Bud Farms - shares | 373 | 253 | 120 | – | 120 | |||||||||||||||
| Hiku Brands Company Ltd. - shares | 30,542 | 13,558 | 16,984 | – | 16,984 | |||||||||||||||
| Scythian Biosciences Corp. - shares | 6,609 | 8,901 | (2,292 | ) | – | (2,292 | ) | |||||||||||||
| Level 2 on fair value hierarchy | ||||||||||||||||||||
| Hiku Brands Company Ltd. - warrants | 17,611 | 18,693 | (1,082 | ) | – | (1,082 | ) | |||||||||||||
| Scythian Biosciences Corp. - warrants | – | 661 | (661 | ) | – | (661 | ) | |||||||||||||
| Level 3 on fair value hierarchy | ||||||||||||||||||||
| Copperstate Farms, LLC - shares | 5,300 | 5,300 | – | – | – | |||||||||||||||
| Copperstate Farms Investors, LLC - shares | 14,700 | 14,700 | – | – | – | |||||||||||||||
| Long-term investments (Note 14) | – | – | – | 39,951 | 39,951 | |||||||||||||||
| Six months ended November 30, 2018 | $ | 79,247 | $ | 65,995 | $ | 13,252 | $ | 39,951 | $ | 53,203 | ||||||||||
| Less transactions in previous quarters: | ||||||||||||||||||||
| August 31, 2018 | 24,112 | 23,929 | 183 | 22,517 | 22,700 | |||||||||||||||
| Three months ended November 30, 2018 | $ | 55,135 | $ | 42,066 | $ | 13,069 | $ | 17,434 | $ | 30,503 | ||||||||||
| 29. | Earnings per share |
The calculation of earnings per share for the three months ended November 30, 2018 was based on the net income attributable to common shareholders of $54,774 (2017 - $6,455) and a weighted average number of common shares outstanding of 244,873,891 (2017 - 138,839,530) calculated as follows:
| 2018 | 2017 | ||
| Basic earnings per share: | |||
| Net income for the period | $ 54,774 | $ 6,455 | |
| Average number of common shares outstanding during the period | 244,873,891 | 138,839,530 | |
| Earnings per share - basic | $ 0.22 | $ 0.05 | |
| 2018 | 2017 | ||||
| Diluted earnings per share: | |||||
| Net income for the period | $ 54,774 | $ 6,455 | |||
| Average number of common shares outstanding during the period | 244,873,891 | 138,839,530 | |||
| "In the money" warrants outstanding during the period | 1,109,499 | 2,735,654 | |||
| "In the money" options outstanding during the period | 3,319,792 | 4,303,259 | |||
| 249,303,182 | 145,878,443 | ||||
| Earnings per share - diluted | $ 0.22 | $ 0.04 | |||
| 32 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
The calculation of earnings per share for the six months ended November 30, 2018 was based on the net income attributable to common shareholders of $75,950 (2017 - $21,496) and a weighted average number of common shares outstanding of 235,166,745 (2017 - 138,775,253) calculated as follows:
| 2018 | 2017 | |||||||||
| Basic earnings per share: | ||||||||||
| Net income for the period | $ | 75,950 | $21,496 | |||||||
| Average number of common shares outstanding during the period | 235,166,745 | 138,775,253 | ||||||||
| Earnings per share - basic | $ | 0.32 | $0.15 | |||||||
| 2018 | 2017 | |||||||||
| Diluted earnings per share: | ||||||||||
| Net income for the period | $ | 75,950 | $21,496 | |||||||
| Average number of common shares outstanding during the period | 235,166,745 | 138,775,253 | ||||||||
| "In the money" warrants outstanding during the period | 1,096,145 | 2,796,468 | ||||||||
| "In the money" options outstanding during the period | 3,154,602 | 4,503,728 | ||||||||
| 239,417,492 | 146,075,449 | |||||||||
| Earnings per share - diluted | $ | 0.32 | $0.15 | |||||||
| 30. | Change in non-cash working capital |
Change in non-cash working capital is comprised of:
| For the six months ended November 30, | ||||||||
| 2018 | 2017 | |||||||
| Decrease (increase) in accounts receivable | $ | (9,850 | ) | $ | (1,840 | ) | ||
| Decrease (increase) in other current assets | (14,210 | ) | (3,212 | ) | ||||
| Decrease (increase) in inventory, net of fair value adjustment | (10,885 | ) | (8,626 | ) | ||||
| Decrease (increase) in biological assets, net of fair value adjustment | (4,875 | ) | 7,345 | |||||
| Increase (decrease) in accounts payable and accrued liabilities | 14,386 | 2,586 | ||||||
| Increase (decrease) in income taxes payable | 4,041 | 365 | ||||||
| Increase (decrease) in deferred revenue | 23,406 | – | ||||||
| $ | 2,013 | $ | (3,382 | ) | ||||
| 31. | Financial risk management and financial instruments |
Financial instruments
The Company has classified its cash and cash equivalents, marketable securities, convertible notes receivable, long-term investments, option payment liability and derivative liability as fair value through profit or loss (“FVTPL”), accounts receivable, other current assets and promissory note receivable as amortized cost, and accounts payable and accrued liabilities, income taxes payable, promissory notes payable, and long-term debt as other financial liabilities.
The carrying values of accounts receivable, other current assets, promissory notes receivable, accounts payable and accrued liabilities, and promissory notes payable approximate their fair values due to their short periods to maturity.
The Company’s long-term debt of $54,685 is subject to fixed interest rates. The Company’s long-term debt is valued based on discounting the future cash outflows associated with the long-term debt. The discount rate is based on the incremental premium above market rates for Government of Canada securities of similar duration. In each period thereafter, the incremental premium is held constant while the Government of Canada security is based on the then current market value to derive the discount rate. The fair value of the Company’s long-term debt in repayment as at November 30, 2018 was $52,465.
| 33 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
Fair value hierarchy
Financial instruments recorded at fair value are classified using a fair value hierarchy that reflects the significance of inputs used in making the measurements. Cash and cash equivalents are Level 1. The hierarchy is summarized as follows:
| Level 1 | quoted prices (unadjusted) in active markets for identical assets and liabilities |
| Level 2 | inputs that are observable for the asset or liability, either directly (prices) or indirectly (derived from prices) from observable market data |
| Level 3 | inputs for assets and liabilities not based upon observable market data |
| Level 1 | Level 2 | Level 3 | November 30, 2018 | |||||||||||||
| Financial assets at FVTPL | ||||||||||||||||
| Cash and cash equivalents | $ | 152,074 | $ | – | $ | – | $ | 152,074 | ||||||||
| Marketable securities | 32,747 | – | – | 32,747 | ||||||||||||
| Convertible notes receivable | – | – | 28,991 | 28,991 | ||||||||||||
| Long-term investments | 32,308 | – | 129,727 | 162,035 | ||||||||||||
| Outstanding, end of the period | $ | 217,129 | $ | – | $ | 158,718 | $ | 375,847 | ||||||||
| Level 1 | Level 2 | Level 3 | May 31, 2018 | |||||||||||
| Financial assets at FVTPL | ||||||||||||||
| Cash and cash equivalents | $ | 59,737 | $ | – | $ | – | $59,737 | |||||||
| Marketable securities | 45,062 | – | – | 45,062 | ||||||||||
| Convertible notes receivable | – | – | 18,071 | 18,071 | ||||||||||
| Long-term investments | 33,600 | 2,567 | 29,861 | 66,028 | ||||||||||
| Outstanding, end of the period | $ | 138,399 | $ | 2,567 | $ | 47,932 | $188,898 | |||||||
The following table presents the changes in level 3 items for the three and six months ended November 30, 2018:
| Unlisted equity securities | Trading derivatives | Total | ||||||||||
| Closing balance May 31, 2018 | $ | 29,861 | $ | 18,071 | $ | 47,932 | ||||||
| Acquisitions | 77,125 | 10,000 | 87,125 | |||||||||
| Disposals | (20,000 | ) | (1,942 | ) | (21,942 | ) | ||||||
| Unrealized gain on fair value | 42,741 | 2,862 | 45,603 | |||||||||
| Closing balance November 30, 2018 | $ | 129,727 | $ | 28,991 | $ | 158,718 | ||||||
Financial risk management
The Company has exposure to the following risks from its use of financial instruments: credit; liquidity; currency rate; and, interest rate price.
| (a) | Credit risk |
The maximum credit exposure at November 30, 2018 is the carrying amount of cash and cash equivalents, marketable securities, accounts receivable and other current assets and promissory notes receivable. The Company does not have significant credit risk with respect to customers. All cash and cash equivalents are placed with major Canadian financial institutions. Marketable securities are placed with major Canadian investment banks and are represented by investment grade corporate bonds.
| 34 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
The Company mitigates its credit risk and volatility on its marketable securities through its investment policy, which permits investments in Federal or Provincial government securities, Provincial utilities or bank institutions and Investment grade corporate bonds.
| Total | 0-30 days | 31-60 days | 61-90 days | 90+ days | ||||||||||||||||
| Trade receivables | $ | 13,807 | $ | 9,946 | $ | 2,198 | $ | 1,224 | $ | 439 | ||||||||||
| 72 | % | 16 | % | 9 | % | 3 | % | |||||||||||||
| (b) | Liquidity risk |
As at November 30, 2018, the Company’s financial liabilities consist of accounts payable and accrued liabilities, which have contractual maturity dates within one-year, promissory note payable, which have a contractual maturity within 15 months and long-term debt, which have contractual maturities over the next five years. The Company manages its liquidity risk by reviewing its capital requirements on an ongoing basis. Based on the Company’s working capital position at November 30, 2018, management regards liquidity risk to be low.
| (c) | Currency rate risk |
As at November 30, 2018, a portion of the Company’s financial assets and liabilities held in United States Dollars (“USD”) and Euros consist of cash and cash equivalents, marketable securities, convertible notes receivable, long-term investments and a promissory note payable. The Company’s objective in managing its foreign currency risk is to minimize its net exposure to foreign currency cash flows by transacting, to the greatest extent possible, with third parties in Canadian dollars. The Company does not currently use foreign exchange contracts to hedge its exposure of its foreign currency cash flows as management has determined that this risk is not significant at this point in time.
The Company is exposed to unrealized foreign exchange risk through its cash and cash equivalents. As at November 30, 2018 the majority of the Company’s cash and cash equivalents was in Canadian dollars, and therefore the Company did not have significant exposure to unrealized foreign exchange risk.
| (d) | Interest rate price risk |
The Company manages interest rate risk by restricting the type of investments and varying the terms of maturity and issuers of marketable securities. Varying the terms to maturity reduces the sensitivity of the portfolio to the impact of interest rate fluctuations.
| (e) | Capital management |
The Company’s objectives when managing its capital are to safeguard its ability to continue as a going concern, to meet its capital expenditures for its continued operations, and to maintain a flexible capital structure which optimizes the cost of capital within a framework of acceptable risk. The Company manages its capital structure and adjusts it in light of changes in economic conditions and the risk characteristics of the underlying assets. To maintain or adjust its capital structure, the Company may issue new shares, issue new debt, or acquire or dispose of assets. The Company is not subject to externally imposed capital requirements.
Management reviews its capital management approach on an ongoing basis and believes that this approach, given the relative size of the Company, is reasonable. There have been no changes to the Company’s capital management approach in the year. The Company considers its cash and cash equivalents and marketable securities as capital.
| 35 |
Aphria Inc. Notes to the Condensed Interim Consolidated Financial Statements For the three and six months ended November 30, 2018 and November 30, 2017 (Unaudited - in thousands of Canadian dollars, except share and per share amounts) |
| 32. | Commitments and contingencies |
The Company had a lease commitment until December 31, 2018 for rental of office space from a related party. On December 31, 2018, the Company extended the lease for an additional 5-years. The Company has an option to extend this lease for an additional 5-year period. The Company has lease commitments for the use of two motor vehicles expiring September 2019 and August 2020 in the amounts payable of $9 and $35, respectively. In April of 2017, the Company indemnified the landlord of the office space leased by Liberty with annual rent from $180 to $190 expiring June 2023. The Company has agreed to contribute $12,000 to Green Acre Capital Fund II. The Company has a lease for rental office space from December 2018 until November 30, 2028. The Company has committed purchase orders outstanding at November 30, 2018 related to capital asset expansion of $81,515, all of which are expected to be paid within the next year. Minimum payments payable over the next five years are as follows:
| Years ending May 31, | ||||||
| 2019 | $ | 93,854 | ||||
| 2020 | 325 | |||||
| 2021 | 331 | |||||
| 2022 | 331 | |||||
| 2023 | 3,384 | |||||
| $ | 98,225 | |||||
From time to time, the Company and/or its subsidiaries may become defendants in legal actions arising out of the ordinary course and conduct of its business.
| 33. | Subsequent events |
The following events occurred subsequent to November 30th, 2018:
(a) The Company became aware that its investee, Resolve, began sales of cannabis in the United States. The Company immediately notified Resolve that it needed to convert its common shares in Resolve into Exchangeable Shares in Resolve, which are exchangeable only on the sale of shares to a third party or upon the Conditions being met. Resolve held a shareholders meeting to approve changes to its capital structure and has swapped the Company’s common shares for Exchangeable Shares.
(b) The Company was either served or became aware of an intent to serve statements of claims in class action lawsuits against the Company and certain of its officers, related to the drop in its share price from December 3 to 5, 2018. At the present time, the Company is aware of six such claims, four of which are domiciled in the United States and two of which are domiciled in Canada. The total amounts claimed in each of the lawsuits have not been quantified at the present time. The Company intends on vigorously defending itself in the litigation. As at November 30, 2018, the Company has not recorded any uninsured amount related to this contingency.
(c) The Company completed its acquisition of CC Pharma GmbH, a leading distributor of pharmaceutical products to more than 13,000 pharmacies in Germany. The Company paid €18,920 in cash at closing, with an earn-out multiple on future EBITDA of up to another €23,500 following closing, if certain performance milestones are met.
36