Exhibit 99.1

Nabriva Therapeutics to Participate in Two Investor Conferences in February
VIENNA, Austria, and KING OF PRUSSIA, Pa., February 8, 2017 — Nabriva Therapeutics AG (NASDAQ:NBRV), a clinical stage biopharmaceutical company engaged in the research and development of novel anti-infective agents to treat serious infections, with a focus on the pleuromutilin class of antibiotics, today announced that Colin Broom, M.D., Chief Executive Officer of Nabriva Therapeutics, will provide a company overview and business update at the following conferences in February.
· Leerink Partners 6th Annual Global Healthcare Conference
· Presentation Date: Wednesday, February 15, 2017
· Presentation Time: 11:30am ET
· Location: Lotte New York Palace Hotel, New York, NY
· RBC Capital Markets 2017 Global Healthcare Conference
· Presentation Date: Wednesday, February 22, 2017
· Presentation Time: 11:00am ET
· Location: Lotte New York Palace Hotel, New York, NY
The presentations will be webcast live and may be accessed by visiting the “Investors” section of the Company’s web site under the “Events and Presentations” tab at http://investors.nabriva.com. A replay of each webcast will be available for 90 days.
About Nabriva Therapeutics AG
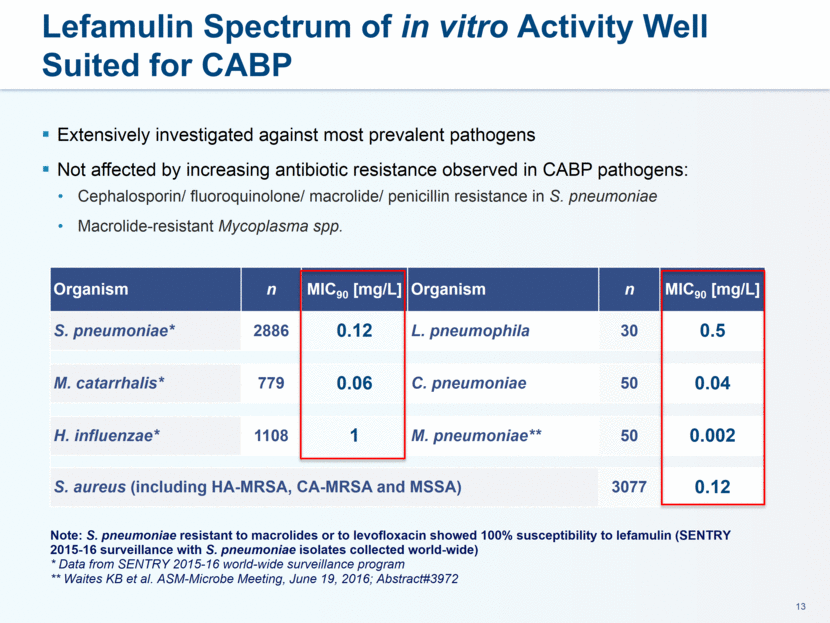
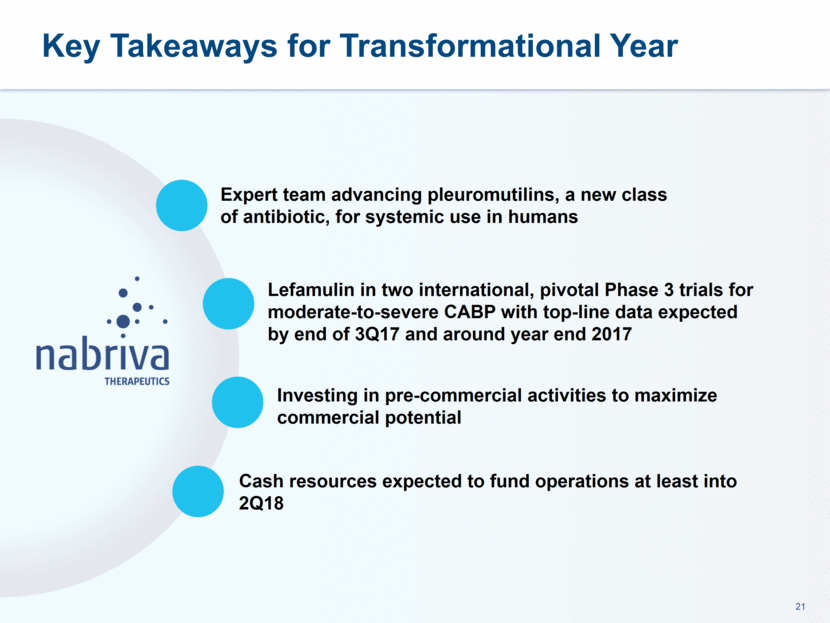
Nabriva Therapeutics is a clinical stage biopharmaceutical company engaged in the research and development of novel anti-infectives to treat serious bacterial infections, with a focus on the pleuromutilin class of antibiotics. Nabriva Therapeutics’ medicinal chemistry expertise has enabled targeted discovery of novel pleuromutilins, including both intravenous and oral formulations of its lead product candidate. Nabriva Therapeutics’ lead product candidate, lefamulin, is a novel semi-synthetic pleuromutilin antibiotic with the potential to be the first-in-class available for systemic
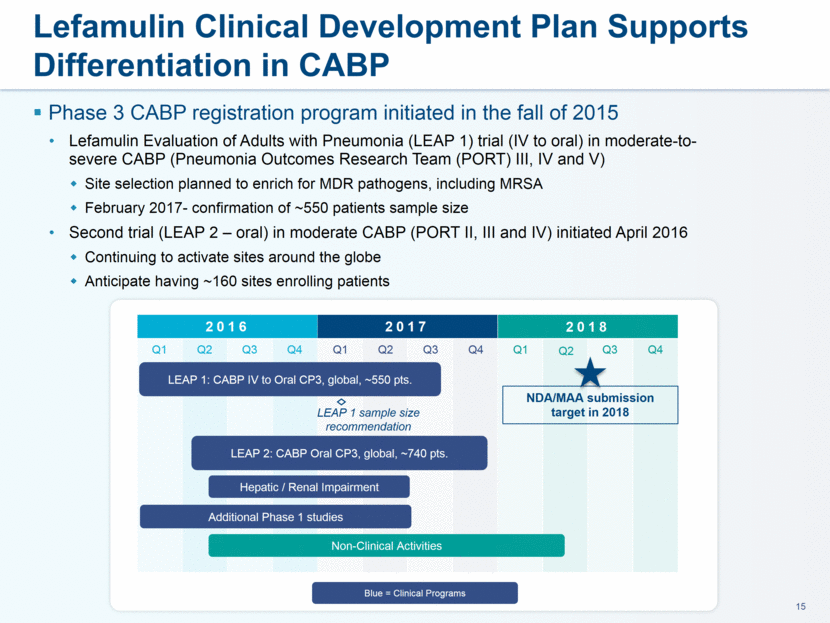
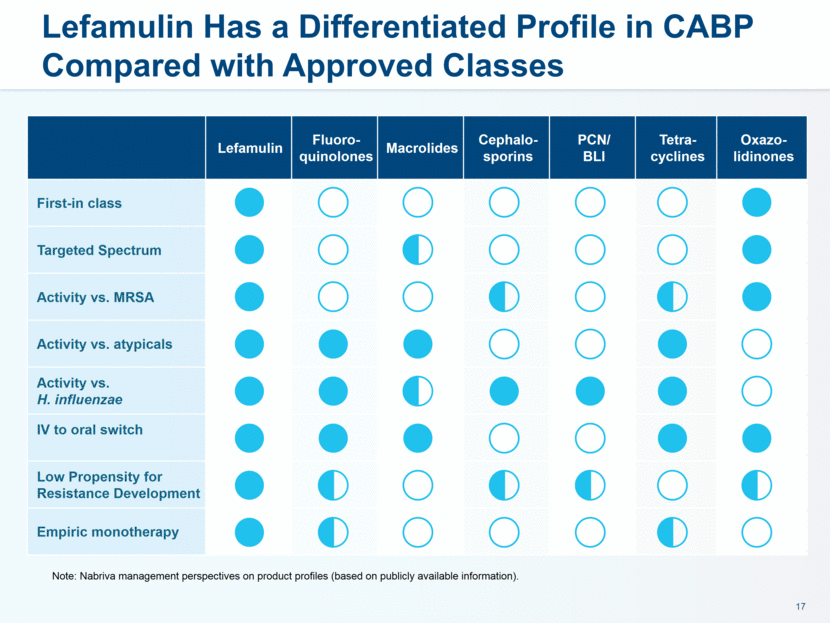
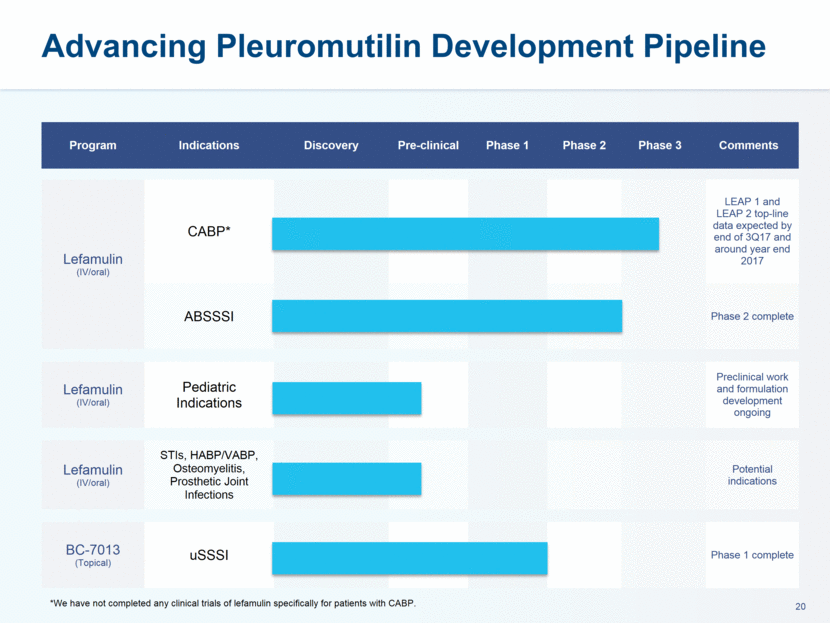
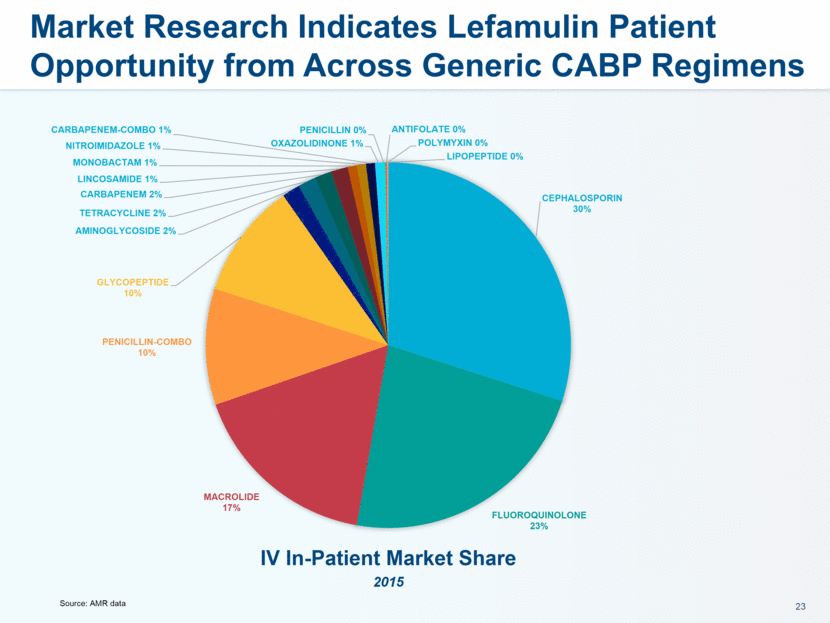
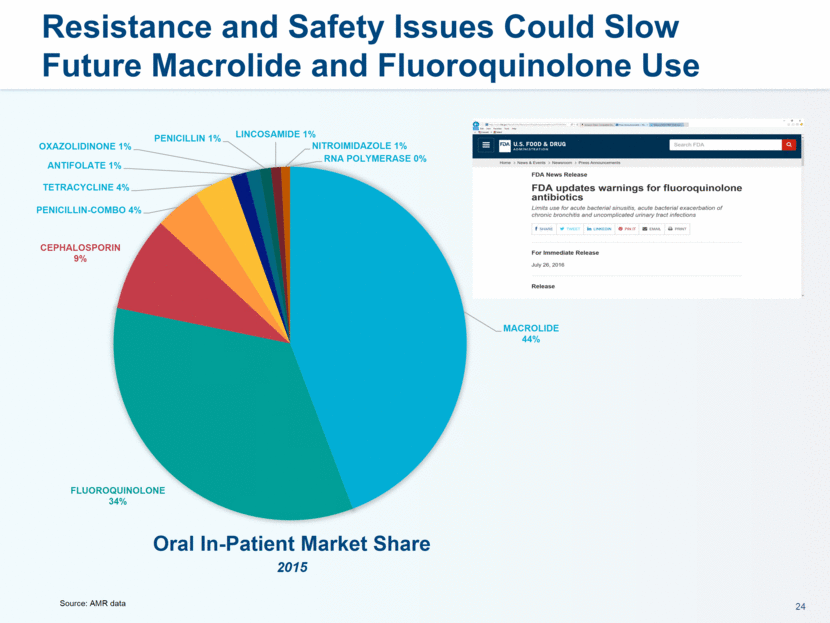
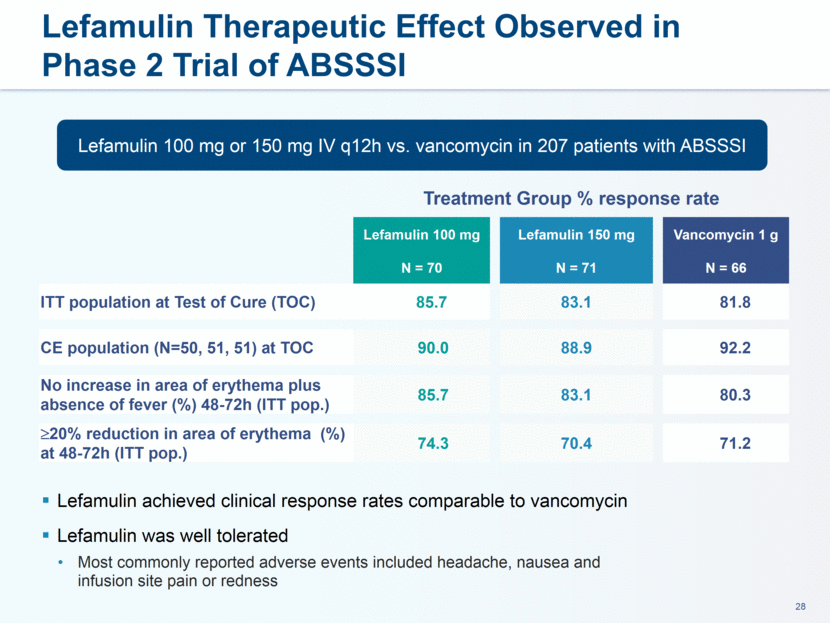
administration in humans. The company believes that lefamulin is the first antibiotic with a novel mechanism of action to have reached late-stage clinical development in more than a decade. Lefamulin is currently being evaluated in two global, registrational Phase 3 clinical trials in patients with moderate to severe CABP. Nabriva Therapeutics believes lefamulin is well positioned for use as a first-line empiric monotherapy for the treatment of moderate to severe CABP due to its novel mechanism of action, targeted spectrum of activity, resistance profile, achievement of substantial drug concentration in lung tissue and fluid, oral and IV formulations and a favorable tolerability profile. Nabriva Therapeutics intends to further pursue development of lefamulin for additional indications, including the treatment of acute bacterial skin and skin structure infections (ABSSSI), and is developing a formulation of lefamulin appropriate for pediatric use.
Nabriva Therapeutics owns exclusive, worldwide rights to lefamulin, which is protected by composition of matter patents issued in the United States, Europe and Japan.
Forward Looking Statements
Any statements in this press release about future expectations, plans and prospects for Nabriva, including but not limited to statements about the development of Nabriva’s product candidates, such as plans for the design, conduct and timelines of Phase 3 clinical trials of lefamulin for CABP, the clinical utility of lefamulin for CABP and Nabriva’s plans for filing of regulatory approvals and efforts to bring lefamulin to market, the development of lefamulin for additional indications, the development of additional formulations of lefamulin, plans to pursue research and development of other product candidates, and other statements containing the words “anticipate,” “believe,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “target,” “potential,” “likely,” “will,” “would,” “could,” “should,” “continue,” and similar expressions, constitute forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including: the uncertainties inherent in the initiation and conduct of clinical trials, availability and timing of data from clinical trials, whether results of early clinical trials or trials in different disease indications will be indicative of the results of ongoing or future trials, uncertainties associated with regulatory review of clinical trials and applications for marketing approvals, the availability or commercial potential of product candidates including lefamulin for use
as a first-line empiric monotherapy for the treatment of moderate to severe CABP, the sufficiency of cash resources and need for additional financing and such other important factors as are set forth under the caption “Risk Factors” in Nabriva’s annual report on Form 20-F and its prospectus supplement dated November 29, 2016 filed with the United States Securities and Exchange Commission. In addition, the forward-looking statements included in this press release represent Nabriva’s views as of the date of this release. Nabriva anticipates that subsequent events and developments will cause its views to change. However, while Nabriva may elect to update these forward-looking statements at some point in the future, it specifically disclaims any obligation to do so. These forward-looking statements should not be relied upon as representing Nabriva’s views as of any date subsequent to the date of this release.
CONTACT:
INVESTOR RELATIONS
Will Sargent
Nabriva Therapeutics AG
William.Sargent@nabriva.com
610-813-6406
MEDIA
Katie Engleman
Pure Communications, Inc.
Katie@purecommunicationsinc.com
910-509-3977