Document
UNITED STATES SECURITIES AND EXCHANGE COMMISSION
Washington, DC 20549
Form 10-Q
|
| |
þ | QUARTERLY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the quarterly period ended March 31, 2017
OR |
| |
¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from_____________to___________
Commission File Number 333-199861
MYLAN N.V.
(Exact name of registrant as specified in its charter)
|
| | |
The Netherlands | | 98-1189497 |
(State or other jurisdiction of incorporation or organization) | | (I.R.S. Employer Identification No.) |
Building 4, Trident Place, Mosquito Way, Hatfield, Hertfordshire, AL10 9UL, England
(Address of principal executive offices)
+44 (0) 1707-853-000
(Registrant’s telephone number, including area code)
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes þ No ¨
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§ 232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes þ No ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act. (Check one):
|
| | | | | | |
Large accelerated filer | | þ | | Accelerated filer | | ¨ |
| | | | | | |
Non-accelerated filer | | ¨ (Do not check if a smaller reporting company) | | Smaller reporting company | | ¨ |
| | | | | | |
| | | | Emerging growth company | | ¨ |
| | | | | | |
If an emerging growth company, indicate by check mark if the registrant has elected not to us the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨ |
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ¨ No þ
Indicate the number of shares outstanding of each of the issuer’s classes of common stock, as of the latest practicable date.
As of May 4, 2017, there were 535,950,491 of the issuer’s €0.01 nominal value ordinary shares outstanding.
MYLAN N.V. AND SUBSIDIARIES
INDEX TO FORM 10-Q
For the Quarterly Period Ended
March 31, 2017
|
| | |
| | Page |
| PART I — FINANCIAL INFORMATION | |
ITEM 1. | Condensed Consolidated Financial Statements (unaudited) | |
| | |
| | |
| | |
| | |
| | |
| | |
| | |
ITEM 2. | | |
| | |
ITEM 3. | | |
| | |
ITEM 4. | | |
| | |
| PART II — OTHER INFORMATION | |
ITEM 1. | | |
| | |
ITEM 1A. | | |
| | |
ITEM 6. | | |
| | |
| | |
PART I — FINANCIAL INFORMATION
MYLAN N.V. AND SUBSIDIARIES
Condensed Consolidated Statements of Operations
(Unaudited; in millions, except per share amounts)
|
| | | | | | | |
| Three Months Ended |
| March 31, |
| 2017 | | 2016 |
Revenues: | | | |
Net sales | $ | 2,687.4 |
| | $ | 2,176.1 |
|
Other revenues | 32.1 |
| | 15.2 |
|
Total revenues | 2,719.5 |
| | 2,191.3 |
|
Cost of sales | 1,634.5 |
| | 1,284.3 |
|
Gross profit | 1,085.0 |
| | 907.0 |
|
Operating expenses: | | | |
Research and development | 217.5 |
| | 253.6 |
|
Selling, general and administrative | 631.3 |
| | 549.3 |
|
Litigation settlements and other contingencies, net | 9.0 |
| | (1.5 | ) |
Total operating expenses | 857.8 |
| | 801.4 |
|
Earnings from operations | 227.2 |
| | 105.6 |
|
Interest expense | 138.2 |
| | 70.3 |
|
Other expense, net | 17.4 |
| | 16.3 |
|
Earnings before income taxes | 71.6 |
| | 19.0 |
|
Income tax provision | 5.2 |
| | 5.1 |
|
Net earnings | $ | 66.4 |
| | $ | 13.9 |
|
Earnings per ordinary share: | | | |
Basic | $ | 0.12 |
| | $ | 0.03 |
|
Diluted | $ | 0.12 |
| | $ | 0.03 |
|
Weighted average ordinary shares outstanding: | | | |
Basic | 534.5 |
| | 489.8 |
|
Diluted | 536.9 |
| | 509.6 |
|
See Notes to Condensed Consolidated Financial Statements
3
MYLAN N.V. AND SUBSIDIARIES
Condensed Consolidated Statements of Comprehensive Earnings
(Unaudited; in millions)
|
| | | | | | | |
| Three Months Ended |
| March 31, |
| 2017 | | 2016 |
Net earnings | $ | 66.4 |
| | $ | 13.9 |
|
Other comprehensive earnings (loss), before tax: | | | |
Foreign currency translation adjustment | 434.2 |
| | 502.0 |
|
Change in unrecognized loss and prior service cost related to defined benefit plans | — |
| | (0.3 | ) |
Net unrecognized gain (loss) on derivatives in cash flow hedging relationships | 32.4 |
| | (49.1 | ) |
Net unrecognized loss on derivatives in net investment hedging relationships | (9.9 | ) | | — |
|
Net unrealized gain on marketable securities | 7.7 |
| | 4.4 |
|
Other comprehensive earnings, before tax | 464.4 |
| | 457.0 |
|
Income tax provision (benefit) | 13.7 |
| | (16.8 | ) |
Other comprehensive earnings, net of tax | 450.7 |
| | 473.8 |
|
Comprehensive earnings | $ | 517.1 |
| | $ | 487.7 |
|
See Notes to Condensed Consolidated Financial Statements
4
MYLAN N.V. AND SUBSIDIARIES
Condensed Consolidated Balance Sheets
(Unaudited; in millions, except share and per share amounts)
|
| | | | | | | |
| March 31,
2017 | | December 31,
2016 |
ASSETS |
Assets | | | |
Current assets: | | | |
Cash and cash equivalents | $ | 723.8 |
| | $ | 998.8 |
|
Accounts receivable, net | 2,872.0 |
| | 3,310.9 |
|
Inventories | 2,547.8 |
| | 2,456.4 |
|
Prepaid expenses and other current assets | 921.9 |
| | 756.4 |
|
Total current assets | 7,065.5 |
| | 7,522.5 |
|
Property, plant and equipment, net | 2,338.0 |
| | 2,322.2 |
|
Intangible assets, net | 14,370.0 |
| | 14,447.8 |
|
Goodwill | 9,394.1 |
| | 9,231.9 |
|
Deferred income tax benefit | 564.0 |
| | 633.2 |
|
Other assets | 541.0 |
| | 568.6 |
|
Total assets | $ | 34,272.6 |
| | $ | 34,726.2 |
|
| | | |
LIABILITIES AND EQUITY |
Liabilities | | | |
Current liabilities: | | | |
Trade accounts payable | $ | 1,141.4 |
| | $ | 1,348.1 |
|
Short-term borrowings | 31.0 |
| | 46.4 |
|
Income taxes payable | 31.0 |
| | 97.7 |
|
Current portion of long-term debt and other long-term obligations | 294.4 |
| | 290.0 |
|
Other current liabilities | 3,026.4 |
| | 3,258.5 |
|
Total current liabilities | 4,524.2 |
| | 5,040.7 |
|
Long-term debt | 14,700.8 |
| | 15,202.9 |
|
Deferred income tax liability | 2,019.1 |
| | 2,006.4 |
|
Other long-term obligations | 1,372.5 |
| | 1,358.6 |
|
Total liabilities | 22,616.6 |
| | 23,608.6 |
|
Equity | | | |
Mylan N.V. shareholders’ equity | | | |
Ordinary shares — nominal value €0.01 per ordinary share | | | |
Shares authorized: 1,200,000,000 | | | |
Shares issued: 537,237,925 and 536,639,291 as of March 31, 2017 and December 31, 2016 | 6.0 |
| | 6.0 |
|
Additional paid-in capital | 8,522.0 |
| | 8,499.3 |
|
Retained earnings | 5,008.5 |
| | 4,942.1 |
|
Accumulated other comprehensive loss | (1,813.0 | ) | | (2,263.7 | ) |
| 11,723.5 |
| | 11,183.7 |
|
Noncontrolling interest | — |
| | 1.4 |
|
Less: Treasury stock — at cost |
| | |
Ordinary shares: 1,311,193 as of March 31, 2017 and December 31, 2016 | 67.5 |
| | 67.5 |
|
Total equity | 11,656.0 |
| | 11,117.6 |
|
Total liabilities and equity | $ | 34,272.6 |
| | $ | 34,726.2 |
|
| | | |
See Notes to Condensed Consolidated Financial Statements
5
MYLAN N.V. AND SUBSIDIARIES
Condensed Consolidated Statements of Cash Flows
(Unaudited; in millions)
|
| | | | | | | |
| Three Months Ended |
| March 31, |
| 2017 | | 2016 |
Cash flows from operating activities: | | | |
Net earnings | $ | 66.4 |
| | $ | 13.9 |
|
Adjustments to reconcile net earnings to net cash provided by operating activities: | | | |
Depreciation and amortization | 415.5 |
| | 297.1 |
|
Share-based compensation expense | 23.1 |
| | 26.5 |
|
Deferred income tax expense | 35.6 |
| | 38.5 |
|
Loss from equity method investments | 33.2 |
| | 30.9 |
|
Other non-cash items | 98.8 |
| | 81.0 |
|
Litigation settlements and other contingencies, net | 8.9 |
| | 0.3 |
|
Changes in operating assets and liabilities: | | | |
Accounts receivable | 286.7 |
| | 83.5 |
|
Inventories | (105.6 | ) | | (222.8 | ) |
Trade accounts payable | (242.7 | ) | | (57.2 | ) |
Income taxes | (175.0 | ) | | (84.7 | ) |
Other operating assets and liabilities, net | 8.0 |
| | (126.5 | ) |
Net cash provided by operating activities | 452.9 |
| | 80.5 |
|
Cash flows from investing activities: | | | |
Cash paid for acquisitions, net | (71.6 | ) | | — |
|
Capital expenditures | (58.4 | ) | | (51.8 | ) |
Proceeds from sale of assets | 31.1 |
| | — |
|
Change in restricted cash | 12.7 |
| | — |
|
Purchase of marketable securities | (2.3 | ) | | (8.5 | ) |
Proceeds from sale of marketable securities | 2.3 |
| | 5.9 |
|
Payments for product rights and other, net | (77.9 | ) | | (105.6 | ) |
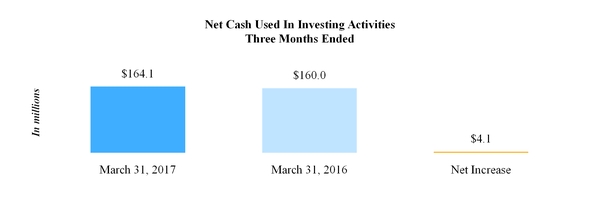
Net cash used in investing activities | (164.1 | ) | | (160.0 | ) |
Cash flows from financing activities: | | | |
Payments of long-term debt | (550.0 | ) | | — |
|
Change in short-term borrowings, net | (17.6 | ) | | 65.1 |
|
Taxes paid related to net share settlement of equity awards | (6.1 | ) | | (6.9 | ) |
Contingent consideration payments | (3.8 | ) | | — |
|
Payments of financing fees | (3.7 | ) | | (31.6 | ) |
Proceeds from exercise of stock options | 5.0 |
| | 3.6 |
|
Other items, net | 0.5 |
| | 0.3 |
|
Net cash (used in) provided by financing activities | (575.7 | ) | | 30.5 |
|
Effect on cash of changes in exchange rates | 11.9 |
| | 12.4 |
|
Net decrease in cash and cash equivalents | (275.0 | ) | | (36.6 | ) |
Cash and cash equivalents — beginning of period | 998.8 |
| | 1,236.0 |
|
Cash and cash equivalents — end of period | $ | 723.8 |
| | $ | 1,199.4 |
|
See Notes to Condensed Consolidated Financial Statements
6
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited)
The accompanying unaudited Condensed Consolidated Financial Statements (“interim financial statements”) of Mylan N.V. and subsidiaries (“Mylan” or the “Company”) were prepared in accordance with accounting principles generally accepted in the United States of America (“U.S. GAAP”) and the rules and regulations of the U.S. Securities and Exchange Commission (the “SEC”) for reporting on Form 10-Q; therefore, as permitted under these rules, certain footnotes and other financial information included in audited financial statements were condensed or omitted. The interim financial statements contain all adjustments (consisting of only normal recurring adjustments) necessary to present fairly the interim results of operations, comprehensive earnings, financial position and cash flows for the periods presented.
These interim financial statements should be read in conjunction with the Consolidated Financial Statements and Notes thereto in Mylan N.V.’s Annual Report on Form 10-K for the year ended December 31, 2016, as amended. The December 31, 2016 Condensed Consolidated Balance Sheet was derived from audited financial statements.
The interim results of operations, comprehensive earnings and cash flows for the three months ended March 31, 2017 are not necessarily indicative of the results to be expected for the full fiscal year or any other future period.
| |
2. | Revenue Recognition and Accounts Receivable |
The Company recognizes net sales when title and risk of loss pass to its customers and when provisions for estimates, including discounts, sales allowances, price adjustments, returns, chargebacks and other promotional programs are reasonably determinable.
Accounts receivable are presented net of allowances relating to these provisions. No significant revisions were made to the methodology used in determining these provisions or the nature of the provisions during the three months ended March 31, 2017. Such allowances were $2.14 billion and $2.05 billion at March 31, 2017 and December 31, 2016, respectively. Other current liabilities include $616.5 million and $809.0 million at March 31, 2017 and December 31, 2016, respectively, for certain sales allowances and other adjustments that are settled in cash.
Accounts receivable, net was comprised of the following at March 31, 2017 and December 31, 2016, respectively:
|
| | | | | | | |
(In millions) | March 31,
2017 | | December 31,
2016 |
Trade receivables, net | $ | 2,568.2 |
| | $ | 3,015.4 |
|
Other receivables | 303.8 |
| | 295.5 |
|
Accounts receivable, net | $ | 2,872.0 |
| | $ | 3,310.9 |
|
Through its wholly owned subsidiary Mylan Pharmaceuticals Inc. (“MPI”), the Company has access to a $400 million accounts receivable securitization facility (the “Receivables Facility”). The receivables underlying any borrowings are included in accounts receivable, net, in the Condensed Consolidated Balance Sheets. There were $854.8 million and $1.13 billion of securitized accounts receivable at March 31, 2017 and December 31, 2016, respectively.
| |
3. | Recent Accounting Pronouncements |
In March 2017, the Financial Accounting Standards Board (the “FASB”) issued Accounting Standards Update 2017-07, Compensation - Retirement Benefits (Topic 715): Improving the Presentation of Net Periodic Pension Cost and Net Periodic Postretirement Benefit Cost (“ASU 2017-07”), which requires companies to disaggregate the service cost component from the other components of net benefit cost and disclose the amount of net benefit cost that is included in the income statement or capitalized in assets, by line item. This guidance requires companies to report the service cost component in the same line item(s) as other compensation costs and to report other pension-related costs (which include interest costs, amortization of pension-related costs from prior periods and gains or losses on plan assets) separately and exclude them from the subtotal of operating income. This guidance also allows only the service cost component to be eligible for capitalization when applicable. This guidance is effective for fiscal years, and interim periods within those fiscal years, beginning after December 15, 2017. This guidance should be applied retrospectively for the presentation of the service cost component and the
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
other components of net periodic pension cost and net periodic postretirement benefit cost in the income statement and prospectively, on and after the effective date, for the capitalization of the service cost component of net periodic pension cost and net periodic postretirement benefit in assets. The update allows a practical expedient that permits a company to use the amounts disclosed in its pension and other postretirement plan note for the prior comparative periods as the estimation basis for applying the retrospective presentation requirements. The Company is currently assessing the impact of the adoption of this guidance on its consolidated financial statements and disclosures.
In January 2017, the FASB issued Accounting Standards Update 2017-04, Intangibles - Goodwill and Other (Topic 350): Simplifying the Test for Goodwill Impairment (“ASU 2017-04”), which simplifies the subsequent measurement of goodwill by eliminating Step 2 from the goodwill impairment test which previously required measurement of any goodwill impairment loss by comparing the implied fair value of a reporting unit’s goodwill with the carrying amount of that goodwill. Under ASU 2017-04, an entity should perform its annual, or interim, goodwill impairment test by comparing the fair value of a reporting unit with its carrying value and recognize an impairment charge for the amount by which the carrying amount exceeds the reporting unit’s fair value; without exceeding the total amount of goodwill allocated to that reporting unit. This guidance is effective for fiscal years, and interim periods within those fiscal years, beginning after December 15, 2019, with early adoption permitted. The Company has elected to early adopt this guidance as of January 1, 2017 and will apply it on a prospective basis. The adoption did not have a material impact on its condensed consolidated financial statements.
In January 2017, the FASB issued Accounting Standards Update 2017-01, Business Combinations (Topic 805) Clarifying the Definition of a Business (“ASU 2017-01”), which narrows the definition of a business and requires an entity to evaluate if substantially all of the fair value of the gross assets acquired is concentrated in a single identifiable asset or a group of similar identifiable assets, which would not constitute the acquisition of a business. The guidance also requires a business to include at least one substantive process and narrows the definition of outputs. This guidance is effective for fiscal years beginning after December 15, 2017, and interim periods within those fiscal years, with early adoption permitted. The Company has elected to early adopt this guidance as of January 1, 2017 and will apply it on a prospective basis. The adoption did not have a material impact on its condensed consolidated financial statements.
In March 2016, the FASB issued Accounting Standards Update 2016-09, Compensation - Stock Compensation (Topic 718) (“ASU 2016-09”), which simplifies the accounting for share-based compensation payments. The new standard requires all excess tax benefits and tax deficiencies (including tax benefits of dividends on share-based payment awards) to be recognized as income tax expense or benefit on the income statement. The tax effects of exercised or vested awards should be treated as discrete items in the reporting period in which they occur. ASU 2016-09 also addresses the classification of excess tax benefits in the statement of cash flows. As required, the Company applied the provisions of ASU 2016-09 on a prospective basis as of January 1, 2017 and the adoption did not have a material impact on its condensed consolidated financial statements.
In May 2014, the FASB issued Accounting Standards Update 2014-09, Revenue from Contracts with Customers (“ASU 2014-09” updated with “ASU 2015-14”, “ASU 2016-08”, “ASU 2016-10”, “ASU 2016-12” and “ASU 2016-20”), which revises accounting guidance on revenue recognition that will supersede nearly all existing revenue recognition guidance under U.S. GAAP. The core principal of this guidance is that an entity should recognize revenue when it transfers promised goods or services to customers in an amount that reflects the consideration which the entity expects to receive in exchange for those goods or services. This guidance also requires additional disclosure about the nature, amount, timing and uncertainty of revenue and cash flows arising from customer contracts, including significant judgments and changes in judgments and assets recognized from costs incurred to obtain or fulfill a contract. This guidance is effective for fiscal years beginning after December 15, 2017, and for interim periods within those fiscal years, and can be applied using a full retrospective or modified retrospective approach. The Company continues to review specific revenue arrangements, including customer and collaboration contracts, and expects to complete the review in the third quarter of 2017. The Company is still evaluating the adoption method it will elect upon implementation.
| |
4. | Acquisitions and Other Transactions |
Meda AB
On February 10, 2016, the Company issued an offer announcement under the Nasdaq Stockholm’s Takeover Rules and the Swedish Takeover Act (collectively, the “Swedish Takeover Rules”) setting forth a public offer to the shareholders of Meda AB (publ.) (“Meda”) to acquire all of the outstanding shares of Meda (the “Offer”), with an enterprise value, including the net debt of Meda, of approximately Swedish kronor (“SEK” or “kr”) 83.6 billion (based on a SEK/USD exchange rate of 8.4158) or $9.9 billion at announcement. On August 2, 2016, the Company announced that the Offer was accepted by Meda
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
shareholders holding an aggregate of approximately 343 million shares, representing approximately 94% of the total number of outstanding Meda shares, as of July 29, 2016, and the Company declared the Offer unconditional. On August 5, 2016, settlement occurred with respect to the Meda shares duly tendered by July 29, 2016 and, as a result, Meda became a controlled subsidiary of the Company. Pursuant to the terms of the Offer, each Meda shareholder that duly tendered Meda shares into the Offer received at settlement (1) in respect of 80% of the number of Meda shares tendered by such shareholder, 165kr in cash per Meda share, and (2) in respect of the remaining 20% of the number of Meda shares tendered by such shareholder, 0.386 of the Company’s ordinary shares per Meda share (subject to treatment of fractional shares as described in the offer document published on June 16, 2016). The non-tendered shares were required to be acquired for cash through a compulsory acquisition proceeding, in accordance with the Swedish Companies Act (Sw. aktiebolagslagen (2005:551)). The compulsory acquisition proceeding price accrued interest as required by the Swedish Companies Act. Meda’s shares were delisted from the Nasdaq Stockholm exchange on August 23, 2016.
On November 1, 2016, the Company made an offer to the remaining Meda shareholders to tender all their Meda shares for cash consideration of 161.31kr per Meda share (the “November Offer”) to provide such remaining shareholders with an opportunity to sell their shares in Meda to the Company in advance of the automatic acquisition of their shares for cash in connection with the compulsory acquisition proceeding. At the end of November 2016, Mylan completed the acquisition of approximately 19 million Meda shares duly tendered for aggregate cash consideration of approximately $330.3 million. In March 2017, the Company received full legal ownership to the remaining non-tendered Meda shares in exchange for a cash payment of approximately $71.6 million, equal to the uncontested portion of the compulsory acquisition price plus statutory interest, and the Company’s arrangement of a customary bank guarantee to secure the payment of any additional cash consideration that may be awarded to the former Meda shareholders in the compulsory acquisition proceeding. The arbitration tribunal conducting the compulsory acquisition proceeding will determine whether to award any such additional cash consideration at the completion of the compulsory acquisition proceeding, which is currently expected to occur in 2017 or 2018. As of March 31, 2017, the Company continues to maintain the bank guarantee as required by Swedish law. The Company does not expect that any additional payments in connection with the compulsory acquisition proceeding would be material to the consolidated financial statements.
On August 5, 2016, the total purchase price was approximately $6.92 billion, net of cash acquired, which includes cash consideration paid of approximately $5.3 billion, the issuance of approximately 26.4 million Mylan N.V. ordinary shares at a fair value of approximately $1.3 billion based on the closing price of the Company’s ordinary shares on August 5, 2016, as reported by the NASDAQ Global Select Stock Market (“NASDAQ”) and an assumed liability of approximately $431.0 million related to the November Offer and the compulsory acquisition proceeding for the non-tendered Meda shares. In accordance with U.S. GAAP, the Company used the acquisition method of accounting to account for this transaction. Under the acquisition method of accounting, the assets acquired and liabilities assumed in the transaction have been recorded at their respective estimated fair values at the acquisition date.
During the three months ended March 31, 2017, adjustments were made to the preliminary purchase price and are reflected as “Measurement Period Adjustments” in the table below. The preliminary allocation of the $6.92 billion purchase price to the assets acquired and liabilities assumed for Meda is as follows:
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
|
| | | | | | | | | | | |
(In millions) | Preliminary Purchase Price Allocation as of December 31, 2016 (a) | | Measurement Period Adjustments (b) | | Preliminary Purchase Price Allocation as of March 31, 2017 (as adjusted) |
Current assets (excluding inventories and net of cash acquired) | $ | 482.5 |
| | $ | — |
| | $ | 482.5 |
|
Inventories | 463.1 |
| | — |
| | 463.1 |
|
Property, plant and equipment | 177.5 |
| | — |
| | 177.5 |
|
Identified intangible assets | 8,060.7 |
| | — |
| | 8,060.7 |
|
Goodwill | 3,676.9 |
| | 1.7 |
| | 3,678.6 |
|
Other assets | 9.5 |
| | — |
| | 9.5 |
|
Total assets acquired | 12,870.2 |
| | 1.7 |
| | 12,871.9 |
|
Current liabilities | (1,105.9 | ) | | — |
| | (1,105.9 | ) |
Long-term debt, including current portion | (2,864.6 | ) | | — |
| | (2,864.6 | ) |
Deferred tax liabilities | (1,613.9 | ) | | (1.7 | ) | | (1,615.6 | ) |
Pension and other postretirement benefits | (322.3 | ) | | — |
| | (322.3 | ) |
Other noncurrent liabilities | (42.4 | ) | | — |
| | (42.4 | ) |
Net assets acquired | $ | 6,921.1 |
| | $ | — |
| | $ | 6,921.1 |
|
| |
(a) | As previously reported in the Company’s December 31, 2016 Annual Report on Form 10-K, as amended. |
| |
(b) | The measurement period adjustments were recorded in the first quarter of 2017 and are primarily related to certain income tax adjustments to reflect facts and circumstances that existed as of the acquisition date. |
The preliminary fair value estimates for the assets acquired and liabilities assumed were based upon preliminary calculations, valuations and assumptions that are subject to change as the Company obtains additional information during the measurement period (up to one year from the acquisition date). The primary areas subject to change relate to the finalization of the working capital components and income taxes.
The acquisition of Meda created a more diversified and expansive portfolio of branded and generic medicines along with a strong and growing portfolio of over-the-counter (“OTC”) products. The combined company has a balanced global footprint with significant scale in key geographic markets, particularly the U.S. and Europe. The acquisition of Meda also expanded our presence in emerging markets, which includes countries in Africa, as well as countries throughout Asia and the Middle East, and is complemented by Mylan’s presence in India, Brazil and Africa (including South Africa). The Company recorded a step-up in the fair value of inventory of approximately $107 million at the acquisition date, which was fully amortized as of December 31, 2016.
The identified intangible assets of $8.06 billion are comprised of product rights and licenses that have a weighted average useful life of 20 years. Significant assumptions utilized in the valuation of identified intangible assets were based on company specific information and projections which are not observable in the market and are thus considered Level 3 measurements as defined by U.S. GAAP. The goodwill of $3.68 billion arising from the acquisition consisted largely of the value of the employee workforce and the expected value of products to be developed in the future. The final allocation of goodwill to Mylan’s reportable segments has not been completed; however, the majority of goodwill is expected to be allocated to the Europe segment. None of the goodwill recognized in this transaction is currently expected to be deductible for income tax purposes.
Renaissance Topicals Business
On June 15, 2016, the Company completed the acquisition of the non-sterile, topicals-focused business (the “Topicals Business”) of Renaissance Acquisition Holdings, LLC (“Renaissance”) for approximately $1.0 billion in cash at closing, including amounts deposited into escrow for potential contingent payments, subject to customary adjustments. The Topicals Business provided the Company with a complementary portfolio of approximately 25 products, an active pipeline of approximately 25 products, and an established U.S. sales and marketing infrastructure targeting dermatologists. The Topicals Business also provided an integrated manufacturing and development platform. In accordance with U.S. GAAP, the Company
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
used the acquisition method of accounting to account for this transaction. Under the acquisition method of accounting, the assets acquired and liabilities assumed in the transaction were recorded at their respective estimated fair values at the acquisition date. The U.S. GAAP purchase price was $972.7 million, which includes estimated contingent consideration of approximately $16 million related to the potential $50 million payment contingent on the achievement of certain 2016 financial targets. The $50 million contingent payment remains in escrow and is classified as restricted cash included in prepaid expenses and other current assets on the Condensed Consolidated Balance Sheets at March 31, 2017 and December 31, 2016.
The preliminary allocation of the $972.7 million purchase price to the assets acquired and liabilities assumed for the Topicals Business is as follows: |
| | | |
(In millions) | |
Current assets (excluding inventories) | $ | 57.7 |
|
Inventories | 74.2 |
|
Property, plant and equipment | 54.8 |
|
Identified intangible assets | 467.0 |
|
In-process research and development | 275.0 |
|
Goodwill | 318.6 |
|
Other assets | 0.1 |
|
Total assets acquired | 1,247.4 |
|
Current liabilities | (74.2 | ) |
Deferred tax liabilities | (194.6 | ) |
Other noncurrent liabilities | (5.9 | ) |
Net assets acquired | $ | 972.7 |
|
The preliminary fair value estimates for the assets acquired and liabilities assumed were based upon preliminary calculations, valuations and assumptions that are subject to change as the Company obtains additional information during the measurement period (up to one year from the acquisition date). The primary areas subject to change relate to the finalization of the working capital components and income taxes.
The acquisition of the Topicals Business broadened the Company’s dermatological portfolio. The amount allocated to in-process research and development (“IPR&D”) represents an estimate of the fair value of purchased in-process technology for research projects that, as of the closing date of the acquisition, had not reached technological feasibility and had no alternative future use. The fair value of IPR&D of $275.0 million was based on the excess earnings method, which utilizes forecasts of expected cash inflows (including estimates for ongoing costs) and other contributory charges. A discount rate of 12.5% was utilized to discount net cash inflows to present values. IPR&D is accounted for as an indefinite-lived intangible asset and will be subject to impairment testing until completion or abandonment of the projects. Upon successful completion and launch of each product, the Company will make a determination of the estimated useful life of the individual asset. The acquired IPR&D projects are in various stages of completion and the estimated costs to complete these projects total approximately $59 million, which is expected to be incurred through 2018. There are risks and uncertainties associated with the timely and successful completion of the projects included in IPR&D, and no assurances can be given that the underlying assumptions used to estimate the fair value of IPR&D will not change or the timely completion of each project to commercial success will occur.
The identified intangible assets of $467.0 million are comprised of $454.0 million of product rights and licenses that have a weighted average useful life of 14 years and $13.0 million of contract manufacturing agreements that have a weighted average useful life of five years. Significant assumptions utilized in the valuation of identified intangible assets were based on company specific information and projections which are not observable in the market and are thus considered Level 3 measurements as defined by U.S. GAAP.
The goodwill of $318.6 million arising from the acquisition consisted largely of the value of the employee workforce and the expected value of products to be developed in the future. All of the goodwill was assigned to the North America segment. None of the goodwill recognized in this transaction is currently expected to be deductible for income tax purposes. The acquisition did not have a material impact on a pro forma basis for the three month period ended March 31, 2016.
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
Unaudited Pro Forma Financial Results
The following table presents supplemental unaudited pro forma information for the acquisition of Meda, as if it had occurred on January 1, 2015. The unaudited pro forma results reflect certain adjustments related to past operating performance and acquisition accounting adjustments, such as increased amortization expense based on the fair value of assets acquired, the impact of transaction costs and the related income tax effects. The unaudited pro forma results do not include any anticipated synergies which may be achievable, or have been achieved, subsequent to the closing of the Meda transaction. Accordingly, the unaudited pro forma results are not necessarily indicative of the results that actually would have occurred had the acquisitions been completed on the stated dates above, nor are they indicative of the future operating results of Mylan N.V. and its subsidiaries. |
| | | |
| Three Months Ended |
| March 31, |
(Unaudited, in millions, except per share amounts) | 2016 |
Total revenues | $ | 2,687.7 |
|
Net earnings | $ | 10.1 |
|
Earnings per ordinary share: | |
Basic | $ | 0.02 |
|
Diluted | $ | 0.02 |
|
Weighted average ordinary shares outstanding: | |
Basic | 518.0 |
|
Diluted | 537.8 |
|
Other Transactions
On March 29, 2017, the Company announced that it had completed its acquisition of the global rights to the Cold-EEZE® brand cold remedy line from ProPhase Labs, Inc. for approximately $50 million in cash. The Company accounted for this transaction as an asset acquisition and the asset is being amortized over a useful life of 15 years. On February 14, 2017, the Company entered into a joint development and marketing agreement for a respiratory product that resulted in approximately $50 million in research and development (“R&D”) expense in the first quarter of 2017.
| |
5. | Share-Based Incentive Plan |
The Company’s shareholders have approved the 2003 Long-Term Incentive Plan (as amended, the “2003 Plan”). Under the 2003 Plan, 55,300,000 ordinary shares are reserved for issuance to key employees, consultants, independent contractors and non-employee directors of the Company through a variety of incentive awards, including: stock options, stock appreciation rights (“SAR”), restricted ordinary shares and units, performance awards (“PSU”), other stock-based awards and short-term cash awards. Stock option awards are granted with an exercise price equal to the fair market value of the ordinary shares underlying the options at the date of the grant, generally become exercisable over periods ranging from three to four years, and generally expire in ten years. Since approval of the 2003 Plan, no further grants of stock options have been made under any other previous plan.
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
The following table summarizes stock option and SAR (together, “stock awards”) activity:
|
| | | | | | |
| Number of Shares Under Stock Awards | | Weighted Average Exercise Price per Share |
Outstanding at December 31, 2016 | 7,699,441 |
| | $ | 33.38 |
|
Granted | 706,995 |
| | 45.02 |
|
Exercised | (242,795 | ) | | 21.27 |
|
Forfeited | (161,159 | ) | | 50.43 |
|
Outstanding at March 31, 2017 | 8,002,482 |
| | $ | 34.43 |
|
Vested and expected to vest at March 31, 2017 | 7,723,468 |
| | $ | 33.98 |
|
Exercisable at March 31, 2017 | 5,976,527 |
| | $ | 30.24 |
|
As of March 31, 2017, stock awards outstanding, stock awards vested and expected to vest, and stock awards exercisable had average remaining contractual terms of 5.9 years, 5.8 years and 4.9 years, respectively. Also, at March 31, 2017, stock awards outstanding, stock awards vested and expected to vest and stock awards exercisable had aggregate intrinsic values of $73.5 million, $73.4 million and $73.0 million, respectively.
A summary of the status of the Company’s nonvested restricted ordinary shares and restricted stock unit awards, including PSUs (collectively, “restricted stock awards”), as of March 31, 2017 and the changes during the three months ended March 31, 2017 are presented below:
|
| | | | | | |
| Number of Restricted Stock Awards | | Weighted Average Grant-Date Fair Value per Share |
Nonvested at December 31, 2016 | 5,667,830 |
| | $ | 42.46 |
|
Granted | 1,255,062 |
| | 45.17 |
|
Released | (483,902 | ) | | 52.54 |
|
Forfeited | (117,259 | ) | | 49.99 |
|
Nonvested at March 31, 2017 | 6,321,731 |
| | $ | 42.09 |
|
As of March 31, 2017, the Company had $181.8 million of total unrecognized compensation expense, net of estimated forfeitures, related to all of its stock-based awards, which will be recognized over the remaining weighted average vesting period of 2.2 years. The total intrinsic value of stock awards exercised and restricted stock units released during the three months ended March 31, 2017 and 2016 was $26.1 million and $40.1 million, respectively.
| |
6. | Pensions and Other Postretirement Benefits |
Defined Benefit Plans
The Company sponsors various defined benefit pension plans in several countries. Benefits provided generally depend on length of service, pay grade and remuneration levels. The Company maintains two fully frozen defined benefit pension plans in the U.S., and employees in the U.S. and Puerto Rico are generally provided retirement benefits through defined contribution plans.
The Company also sponsors other postretirement benefit plans including plans that provide for postretirement supplemental medical coverage. Benefits from these plans are provided to employees and their spouses and dependents who meet various minimum age and service requirements. In addition, the Company sponsors other plans that provide for life insurance benefits and postretirement medical coverage for certain officers and management employees.
Net Periodic Benefit Cost
Components of net periodic benefit cost for the three months ended March 31, 2017 and 2016 were as follows:
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
|
| | | | | | | |
| Pension and Other Postretirement Benefits |
| March 31, |
(In millions) | 2017 | | 2016 |
Service cost | $ | 5.0 |
| | $ | 3.9 |
|
Interest cost | 3.7 |
| | 1.5 |
|
Expected return on plan assets | (3.5 | ) | | (2.0 | ) |
Amortization of prior service costs | 0.1 |
| | 0.1 |
|
Recognized net actuarial losses | 0.2 |
| | 0.2 |
|
Net periodic benefit cost | $ | 5.5 |
| | $ | 3.7 |
|
The Company is making the minimum mandatory contributions to its U.S. defined benefit pension plans in the 2017 plan year. The Company expects to make total benefit payments of approximately $30.4 million and contributions to pension and other postretirement benefit plans of approximately $30.2 million in 2017.
| |
7. | Balance Sheet Components |
Selected balance sheet components consist of the following:
Inventories
|
| | | | | | | |
(In millions) | March 31,
2017 | | December 31,
2016 |
Raw materials | $ | 833.8 |
| | $ | 783.4 |
|
Work in process | 427.3 |
| | 436.0 |
|
Finished goods | 1,286.7 |
| | 1,237.0 |
|
Inventories | $ | 2,547.8 |
| | $ | 2,456.4 |
|
Prepaid and other current assets |
| | | | | | | |
(In millions) | March 31,
2017 | | December 31, 2016 |
Prepaid expenses | $ | 177.0 |
| | $ | 169.1 |
|
Restricted cash | 135.8 |
| | 148.1 |
|
Available-for-sale securities | 91.3 |
| | 83.7 |
|
Fair value of financial instruments | 88.2 |
| | 62.2 |
|
Trading securities | 30.7 |
| | 29.6 |
|
Other current assets | 398.9 |
| | 263.7 |
|
Prepaid expenses and other current assets | $ | 921.9 |
| | $ | 756.4 |
|
Prepaid expenses consist primarily of prepaid rent, insurance and other individually insignificant items.
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
Property, plant and equipment, net
|
| | | | | | | |
(In millions) | March 31,
2017 | | December 31, 2016 |
Machinery and equipment | $ | 2,245.3 |
| | $ | 2,227.9 |
|
Buildings and improvements | 1,124.8 |
| | 1,106.5 |
|
Construction in progress | 330.4 |
| | 328.8 |
|
Land and improvements | 147.6 |
| | 144.7 |
|
Gross property, plant and equipment | 3,848.1 |
| | 3,807.9 |
|
Accumulated depreciation | 1,510.1 |
| | 1,485.7 |
|
Property, plant and equipment, net | $ | 2,338.0 |
| | $ | 2,322.2 |
|
Other assets
|
| | | | | | | |
(In millions) | March 31,
2017 | | December 31, 2016 |
Equity method investments, clean energy investments | $ | 305.6 |
| | $ | 320.6 |
|
Equity method investments, Sagent Agila | 58.6 |
| | 75.8 |
|
Other long-term assets | 176.8 |
| | 172.2 |
|
Other assets | $ | 541.0 |
| | $ | 568.6 |
|
Trade accounts payable
|
| | | | | | | |
(In millions) | March 31,
2017 | | December 31,
2016 |
Trade accounts payable | $ | 749.6 |
| | $ | 939.5 |
|
Other payables | 391.8 |
| | 408.6 |
|
Trade accounts payable | $ | 1,141.4 |
| | $ | 1,348.1 |
|
Other current liabilities
|
| | | | | | | |
(In millions) | March 31,
2017 | | December 31, 2016 |
Accrued sales allowances | $ | 616.5 |
| | $ | 809.0 |
|
Legal and professional accruals, including litigation accruals | 723.2 |
| | 720.4 |
|
Payroll and employee benefit plan accruals | 326.8 |
| | 409.8 |
|
Contingent consideration | 244.8 |
| | 256.9 |
|
Accrued interest | 130.7 |
| | 41.0 |
|
Restructuring | 77.2 |
| | 138.6 |
|
Equity method investments, clean energy investments | 65.3 |
| | 64.7 |
|
Fair value of financial instruments | 8.5 |
| | 15.3 |
|
Compulsory acquisition proceeding | — |
| | 70.2 |
|
Other | 833.4 |
| | 732.6 |
|
Other current liabilities | $ | 3,026.4 |
| | $ | 3,258.5 |
|
On March 31, 2017, the Company announced that Meridian Medical Technologies (“Meridian”), a Pfizer company that manufactures for the EpiPen® Auto-Injector, expanded a voluntary recall of select lots of EpiPen® Auto-Injector and EpiPen Jr® Auto-Injector to include additional lots distributed in the U.S. and other markets in consultation with the U.S. Food and Drug Administration (“FDA”) (the “EpiPen® Auto-Injector Recall”). This recall was conducted as a result of the receipt of two previously disclosed reports outside of the U.S. of the failure to activate the device due to a potential defect in a supplier component. Both reports were related to the single lot that was previously recalled. The expanded voluntary recall was initiated
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
in the U.S. and also extends to additional markets in Europe, Asia, North and South America. The Company is replacing recalled devices at no cost to the consumer. Estimated costs to Mylan related to product recalls are based on a formal campaign soliciting return of the product and are accrued when they are deemed to be probable and can be reasonably estimated. As of March 31, 2017, the Company recorded an accrual with respect to the recall but there can be no assurance that future costs related to the recall will not exceed amounts recorded. In addition, Meridian is contractually obligated to reimburse Mylan for costs related to the EpiPen® Auto-Injector Recall, and the Company has recorded an asset for the recovery of such costs.
In March 2017, the Company completed the compulsory acquisition proceeding and settled the associated liability. The Meda shareholders whose shares were subject to the compulsory acquisition proceeding received cash consideration plus statutory interest for their Meda shares totaling approximately $71.6 million. Refer to Note 4 Acquisitions and Other Transactions for additional information.
Other long-term obligations |
| | | | | | | |
(In millions) | March 31,
2017 | | December 31, 2016 |
Employee benefit liabilities | $ | 387.1 |
| | $ | 396.7 |
|
Contingent consideration | 321.2 |
| | 307.7 |
|
Equity method investments, clean energy investments | 288.5 |
| | 302.3 |
|
Tax contingencies | 240.5 |
| | 239.3 |
|
Other | 135.2 |
| | 112.6 |
|
Other long-term obligations | $ | 1,372.5 |
| | $ | 1,358.6 |
|
| |
8. | Equity Method Investments |
The Company has five equity method investments in limited liability companies that own refined coal production plants (the “clean energy investments”), whose activities qualify for income tax credits under Section 45 of the U.S. Internal Revenue Code of 1986, as amended.
Since December 2013, the Company held a 50% interest in Sagent Agila LLC (“Sagent Agila”), which was a joint venture established to develop, manufacture and distribute certain generic injectable products in the U.S. In April 2017, the Company and Sagent Pharmaceuticals Inc. (“Sagent”) finalized an agreement to dissolve the joint venture. Under the terms of the agreement, Mylan received Sagent’s interest in the joint venture in exchange for an approved product right. The assets in the joint venture consisted entirely of product rights for commercialized generic injectables. As a result of this transaction, during the three months ended March 31, 2017, the Company recognized a loss of $5.7 million as a component of net losses from equity method investments. Additionally, during the three months ended March 31, 2017, the Company received a dividend payment of $8.4 million from Sagent Agila, which reduced the carrying value of the equity investment. In the second quarter of 2017, the Company will reclassify its investment in Sagent Agila to product rights and licenses and amortize the amount over the remaining estimated useful lives of the products.
Summarized financial information, in the aggregate, for the Company’s significant equity method investments on a 100% basis for the three months ended March 31, 2017 and 2016 are as follows:
|
| | | | | | | |
| Three Months Ended |
| March 31, |
(In millions) | 2017 | | 2016 |
Total revenues | $ | 122.9 |
| | $ | 144.0 |
|
Gross loss | (2.7 | ) | | (0.3 | ) |
Operating and non-operating expense | 5.8 |
| | 5.7 |
|
Net loss | $ | (8.5 | ) | | $ | (6.0 | ) |
The Company’s net losses from the six equity method investments includes amortization expense related to the excess of the cost basis of the Company’s investment to the underlying assets of each individual investee. For the three months ended March 31, 2017 and 2016, the Company recognized net losses from equity method investments of $33.2 million and $30.9
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
million, respectively, which was recognized as a component of other expense, net in the Condensed Consolidated Statements of Operations. The Company recognizes the income tax credits and benefits from the clean energy investments as part of its provision for income taxes.
| |
9. | Earnings per Ordinary Share |
Basic earnings per ordinary share is computed by dividing net earnings by the weighted average number of ordinary shares outstanding during the period. Diluted earnings per ordinary share is computed by dividing net earnings by the weighted average number of ordinary shares outstanding during the period increased by the number of additional shares that would have been outstanding related to potentially dilutive securities or instruments, if the impact is dilutive.
On April 15, 2016, in connection with the expiration and settlement of the Company’s equity classified warrants, the Company issued approximately 17.0 million Mylan N.V. ordinary shares. The dilutive impact of the warrants, prior to settlement, is included in the calculation of diluted earnings per ordinary share based upon the average market value of the Company’s ordinary shares during the period as compared to the exercise price. For the three months ended March 31, 2016, 16.7 million warrants were included in the calculation of diluted earnings per ordinary share.
Basic and diluted earnings per ordinary share are calculated as follows:
|
| | | | | | | |
| Three Months Ended |
| March 31, |
(In millions, except per share amounts) | 2017 | | 2016 |
Basic earnings (numerator): | | | |
Net earnings | $ | 66.4 |
| | $ | 13.9 |
|
Shares (denominator): | | | |
Weighted average ordinary shares outstanding | 534.5 |
| | 489.8 |
|
Basic earnings per ordinary share | $ | 0.12 |
| | $ | 0.03 |
|
|
| | | | | | | |
Diluted earnings (numerator): | | | |
Net earnings | $ | 66.4 |
| | $ | 13.9 |
|
Shares (denominator): | | | |
Weighted average ordinary shares outstanding | 534.5 |
| | 489.8 |
|
Share-based awards and warrants | 2.4 |
| | 19.8 |
|
Total dilutive shares outstanding | 536.9 |
| | 509.6 |
|
Diluted earnings per ordinary share | $ | 0.12 |
| | $ | 0.03 |
|
Additional stock awards and restricted stock awards were outstanding during the three months ended March 31, 2017 and 2016, but were not included in the computation of diluted earnings per ordinary share for each respective period because the effect would be anti-dilutive. Excluded shares at March 31, 2017 include certain share-based compensation awards and restricted ordinary shares whose performance conditions had not been fully met. Such excluded shares and anti-dilutive awards represented 4.4 million shares and 6.2 million shares for the three months ended March 31, 2017 and 2016, respectively.
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
| |
10. | Goodwill and Intangible Assets |
The changes in the carrying amount of goodwill for the three months ended March 31, 2017 are as follows:
|
| | | | | | | | | | | | | | | |
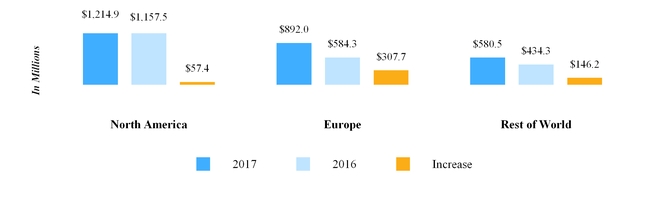
(In millions) | North America Segment | | Europe Segment | | Rest of World Segment | | Total |
Balance at December 31, 2016: | | | | | | | |
Goodwill | $ | 3,990.4 |
| | $ | 3,859.1 |
| | $ | 1,767.4 |
| | $ | 9,616.9 |
|
Accumulated impairment losses | (385.0 | ) | | — |
| | — |
| | (385.0 | ) |
| 3,605.4 |
| | 3,859.1 |
| | 1,767.4 |
| | 9,231.9 |
|
Reclassifications(1) | (199.0 | ) | | 373.2 |
| | (174.2 | ) | | — |
|
Measurement period adjustments | — |
| | 1.7 |
| | — |
| | 1.7 |
|
Divestiture | — |
| | (1.3 | ) | | — |
| | (1.3 | ) |
Foreign currency translation | 6.6 |
| | 77.7 |
| | 77.5 |
| | 161.8 |
|
| $ | 3,413.0 |
| | $ | 4,310.4 |
| | $ | 1,670.7 |
| | $ | 9,394.1 |
|
Balance at March 31, 2017: | | | | | | | |
Goodwill | $ | 3,798.0 |
| | $ | 4,310.4 |
| | $ | 1,670.7 |
| | $ | 9,779.1 |
|
Accumulated impairment losses | (385.0 | ) | | — |
| | — |
| | (385.0 | ) |
| $ | 3,413.0 |
| | $ | 4,310.4 |
| | $ | 1,670.7 |
| | $ | 9,394.1 |
|
| |
(1) | The reclassifications in the current quarter relate to the allocation of goodwill for the Meda acquisition. |
Intangible assets consist of the following components at March 31, 2017 and December 31, 2016:
|
| | | | | | | | | | | | | |
(In millions) | Weighted Average Life (Years) | | Original Cost | | Accumulated Amortization | | Net Book Value |
March 31, 2017 | | | | | | | |
Amortized intangible assets: | | | | | | | |
Product rights and licenses | 15 | | $ | 17,300.9 |
| | $ | 3,986.2 |
| | $ | 13,314.7 |
|
Patents and technologies | 20 | | 116.6 |
| | 109.6 |
| | 7.0 |
|
Other (1) | 6 | | 468.8 |
| | 354.7 |
| | 114.1 |
|
| | | 17,886.3 |
| | 4,450.5 |
| | 13,435.8 |
|
In-process research and development | | | 934.2 |
| | — |
| | 934.2 |
|
| | | $ | 18,820.5 |
| | $ | 4,450.5 |
| | $ | 14,370.0 |
|
December 31, 2016 | | | | | | | |
Amortized intangible assets: | | | | | | | |
Product rights and licenses | 15 | | $ | 16,968.4 |
| | $ | 3,585.7 |
| | $ | 13,382.7 |
|
Patents and technologies | 20 | | 116.6 |
| | 108.5 |
| | 8.1 |
|
Other (1) | 6 | | 465.9 |
| | 330.0 |
| | 135.9 |
|
| | | 17,550.9 |
| | 4,024.2 |
| | 13,526.7 |
|
In-process research and development | | | 921.1 |
| | — |
| | 921.1 |
|
| | | $ | 18,472.0 |
| | $ | 4,024.2 |
| | $ | 14,447.8 |
|
____________
| |
(1) | Other intangible assets consist principally of customer lists, contractual rights and other contracts. |
In December 2011, the Company completed the acquisition of the exclusive worldwide rights to develop, manufacture and commercialize a generic equivalent to GlaxoSmithKline’s Advair® Diskus and Seretide® Diskus incorporating Pfizer
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
Inc.’s proprietary dry powder inhaler delivery platform (the “respiratory delivery platform”). The Company accounted for this transaction as a purchase of a business and utilized the acquisition method of accounting. In conjunction with the Company’s Generic Drug User Fee Agreement goal date, on March 28, 2017, the Company received a complete response letter from the FDA regarding its Abbreviated New Drug Application for the respiratory delivery platform. As of March 31, 2017, the Company has an IPR&D asset of $347.2 million and related contingent consideration liability of $436.1 million. The Company performed an analysis and valuation of the IPR&D asset and the fair value of the related contingent consideration liability through the use of a discounted cash flow model. The model contained certain key assumptions including: the expected product launch date, the number of competitors, the timing of competition and a discount factor based on an industry specific weighted average cost of capital. Based on the analysis performed, the Company determined that the IPR&D asset was not impaired at March 31, 2017. Additionally, no significant fair value adjustment was required for the contingent consideration. However, resolution of the matters with the FDA, market conditions and other factors may result in significant changes in the projections and assumptions utilized in the discounted cash flow model, which could lead to material adjustments to the recorded amounts.
Amortization expense, which is classified primarily within cost of sales in the Condensed Consolidated Statements of Operations for the three months ended March 31, 2017 and 2016 totaled:
|
| | | | | | | |
| Three Months Ended |
| March 31, |
(In millions) | 2017 | | 2016 |
Intangible asset amortization expense | $ | 342.4 |
| | $ | 242.3 |
|
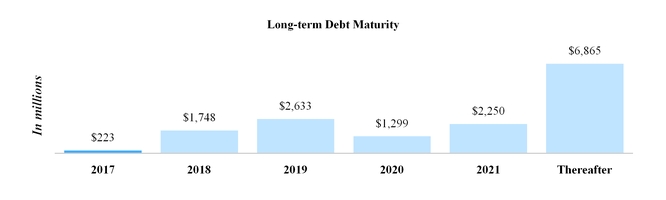
Intangible asset amortization expense over the remainder of 2017 and for the years ended December 31, 2018 through 2021 is estimated to be as follows:
|
| | | |
(In millions) | |
2017 | $ | 942 |
|
2018 | 1,220 |
|
2019 | 1,130 |
|
2020 | 1,010 |
|
2021 | 936 |
|
| |
11. | Financial Instruments and Risk Management |
The Company is exposed to certain financial risks relating to its ongoing business operations. The primary financial risks that are managed by using derivative instruments are foreign currency risk and interest rate risk.
Foreign Currency Risk Management
In order to manage foreign currency risk, the Company enters into foreign exchange forward contracts to mitigate risk associated with changes in spot exchange rates of mainly non-functional currency denominated assets or liabilities. The foreign exchange forward contracts are measured at fair value and reported as current assets or current liabilities on the Condensed Consolidated Balance Sheets. Any gains or losses on the foreign exchange forward contracts are recognized in earnings in the period incurred in the Condensed Consolidated Statements of Operations.
The Company has also entered into forward contracts to hedge forecasted foreign currency denominated sales from certain international subsidiaries. These contracts are designated as cash flow hedges to manage foreign currency transaction risk and are measured at fair value and reported as current assets or current liabilities on the Condensed Consolidated Balance Sheets. Any changes in fair value are included in earnings or deferred through accumulated other comprehensive earnings (“AOCE”), depending on the nature and effectiveness of the offset. Any ineffectiveness in a cash flow hedging relationship is recognized immediately in earnings in the Condensed Consolidated Statements of Operations.
In the first quarter of 2017, the Company designated certain Euro borrowings as a hedge of its investment in certain Euro-functional currency subsidiaries in order to manage foreign currency translation risk. The notional amount of the net investment hedges was €1.4 billion and consists of €604 million of the €1.0 billion aggregate principal amount of 2.250% Senior Notes due 2024 (the “2024 Euro Notes”) and €750 million aggregate principal amount of 3.125% Senior Notes due 2028
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
(the “2028 Euro Notes”). Borrowings designated as net investment hedges are marked to market using the current spot exchange rate as of the end of the period, with gains and losses included in the foreign currency translation component of AOCE until the sale or substantial liquidation of the underlying net investments. The Company recorded no ineffectiveness from its net investment hedges for the three months ended March 31, 2017. In addition, the Company manages the related foreign exchange risk of the €500 million aggregate principal amount of floating rate Senior Notes due 2018 (the “Floating Rate Euro Notes”), €750 million aggregate principal amount of 1.250% Euro Senior Notes due 2020 (the “2020 Euro Notes”) and the remaining portion of the 2024 Euro Notes through certain Euro denominated financial assets.
Interest Rate Risk Management
The Company enters into interest rate swaps in order to manage interest rate risk associated with the Company’s fixed-rate and floating-rate debt. These derivative instruments are measured at fair value and reported as current assets or current liabilities in the Condensed Consolidated Balance Sheets.
Cash Flow Hedging Relationships
The Company’s interest rate swaps designated as cash flow hedges fix the interest rate on a portion of the Company’s variable-rate debt or hedge part of the Company’s interest rate exposure associated with variability in future cash flows attributable to changes in interest rates. Any changes in fair value are included in earnings or deferred through AOCE, depending on the nature and effectiveness of the offset. Any ineffectiveness in a cash flow hedging relationship is recognized immediately in earnings in the Condensed Consolidated Statements of Operations.
Fair Value Hedging Relationships
The Company’s interest rate swaps designated as fair value hedges convert the fixed rate on a portion of the Company’s fixed-rate senior notes to a variable rate. Any changes in the fair value of these derivative instruments, as well as the offsetting change in fair value of the portion of the fixed-rate debt being hedged, is included in interest expense.
The Company regularly reviews the creditworthiness of its financial counterparties and does not expect to incur a significant loss from the failure of any counterparties to perform under any agreements. The Company is not subject to any obligations to post collateral under derivative instrument contracts. Certain derivative instrument contracts entered into by the Company are governed by master agreements, which contain credit-risk-related contingent features that would allow the counterparties to terminate the contracts early and request immediate payment should the Company trigger an event of default on other specified borrowings. The Company records all derivative instruments on a gross basis in the Condensed Consolidated Balance Sheets. Accordingly, there are no offsetting amounts that net assets against liabilities.
The Effect of Derivative Instruments on the Condensed Consolidated Balance Sheets
Fair Values of Derivative Instruments
Derivatives Designated as Hedging Instruments
|
| | | | | | | | | | | |
| Asset Derivatives |
| March 31, 2017 | | December 31, 2016 |
(In millions) | Balance Sheet Location | | Fair Value | | Balance Sheet Location | | Fair Value |
Interest rate swaps | Prepaid expenses and other current assets | | $ | 23.9 |
| | Prepaid expenses and other current assets | | $ | 26.2 |
|
Foreign currency forward contracts | Prepaid expenses and other current assets | | 47.5 |
| | Prepaid expenses and other current assets | | 21.9 |
|
Total | | | $ | 71.4 |
| | | | $ | 48.1 |
|
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
The Effect of Derivative Instruments on the Condensed Consolidated Balance Sheets
Fair Values of Derivative Instruments
Derivatives Not Designated as Hedging Instruments
|
| | | | | | | | | | | |
| Asset Derivatives |
| March 31, 2017 | | December 31, 2016 |
(In millions) | Balance Sheet Location | | Fair Value | | Balance Sheet Location | | Fair Value |
Foreign currency forward contracts | Prepaid expenses and other current assets | | $ | 16.9 |
| | Prepaid expenses and other current assets | | $ | 14.0 |
|
Total | | | $ | 16.9 |
| | | | $ | 14.0 |
|
|
| | | | | | | | | | | |
| Liability Derivatives |
| March 31, 2017 | | December 31, 2016 |
(In millions) | Balance Sheet Location | | Fair Value | | Balance Sheet Location | | Fair Value |
Foreign currency forward contracts | Other current liabilities | | $ | 8.5 |
| | Other current liabilities | | $ | 15.3 |
|
Total | | | $ | 8.5 |
| | | | $ | 15.3 |
|
The Effect of Derivative Instruments on the Condensed Consolidated Statements of Operations
Derivatives in Fair Value Hedging Relationships
|
| | | | | | | | | |
| Location of (Loss) Gain Recognized in Earnings on Derivatives | | Amount of (Loss) Gain Recognized in Earnings on Derivatives |
(In millions) | | Three Months Ended |
| March 31, |
| 2017 | | 2016 |
Interest rate swaps | Interest expense | | $ | (2.4 | ) | | $ | 29.6 |
|
Total | | | $ | (2.4 | ) | | $ | 29.6 |
|
|
| | | | | | | | | |
| Location of Gain (Loss) Recognized in Earnings on Hedged Items | | Amount of Gain (Loss) Recognized in Earnings on Hedged Items |
(In millions) | | Three Months Ended |
| March 31, |
| 2017 | | 2016 |
2023 Senior Notes (3.125% coupon) | Interest expense | | $ | 2.4 |
| | $ | (29.6 | ) |
Total | | | $ | 2.4 |
| | $ | (29.6 | ) |
The Effect of Derivative Instruments on the Condensed Consolidated Statements of Comprehensive Earnings
Derivatives in Cash Flow Hedging Relationships
|
| | | | | | | | |
| | Amount of Gain (Loss)
Recognized in AOCE
(Net of Tax) on Derivative
(Effective Portion) |
| | Three Months Ended |
| | March 31, |
(In millions) | | 2017 | | 2016 |
Foreign currency forward contracts | | $ | 14.1 |
| | $ | (4.4 | ) |
Interest rate swaps | | 0.7 |
| | (35.9 | ) |
Total | | $ | 14.8 |
| | $ | (40.3 | ) |
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
The Effect of Derivative Instruments on the Condensed Consolidated Statements of Comprehensive Earnings
Derivatives in Net Investment Hedging Relationships
|
| | | | | | | | |
| | Amount of Loss Recognized in AOCE
(Net of Tax) on Derivative
(Effective Portion) |
| | Three Months Ended |
| | March 31, |
(In millions) | | 2017 | | 2016 |
Foreign currency borrowings and forward contracts | | $ | (9.9 | ) | | $ | — |
|
Total | | $ | (9.9 | ) | | $ | — |
|
The Effect of Derivative Instruments on the Condensed Consolidated Statements of Operations
Derivatives in Cash Flow Hedging Relationships
|
| | | | | | | | | |
| Location of Loss Reclassified from AOCE into Earnings (Effective Portion) | | Amount of (Loss) Gain Reclassified from AOCE into Earnings (Effective Portion) |
| | Three Months Ended |
| | March 31, |
(In millions) | | 2017 | | 2016 |
Foreign currency forward contracts | Net sales | | $ | (5.2 | ) | | $ | (10.6 | ) |
Interest rate swaps | Interest expense | | (1.8 | ) | | 0.9 |
|
Total | | | $ | (7.0 | ) | | $ | (9.7 | ) |
|
| | | | | | | | | |
| Location of (Loss) Gain Excluded from the Assessment of Hedge Effectiveness | | Amount of (Loss) Gain Excluded from the Assessment of Hedge Effectiveness |
| | Three Months Ended |
| | March 31, |
(In millions) | | 2017 | | 2016 |
Foreign currency forward contracts | Other expense, net | | $ | (0.8 | ) | | $ | 7.3 |
|
Total | | | $ | (0.8 | ) | | $ | 7.3 |
|
At March 31, 2017, the Company expects that approximately $1 million of pre-tax net losses on cash flow hedges will be reclassified from AOCE into earnings during the next twelve months.
The Effect of Derivative Instruments on the Condensed Consolidated Statements of Operations
Derivatives Not Designated as Hedging Instruments
|
| | | | | | | | | |
| Location of Loss Recognized in Earnings on Derivatives | | Amount of Loss Recognized in Earnings on Derivatives |
| | Three Months Ended |
| | March 31, |
(In millions) | | 2017 | | 2016 |
Foreign currency option and forward contracts | Other expense, net | | $ | (0.3 | ) | | $ | (15.0 | ) |
Total | | | $ | (0.3 | ) | | $ | (15.0 | ) |
Fair Value Measurement
Fair value is based on the price that would be received from the sale of an identical asset or paid to transfer an identical liability in an orderly transaction between market participants at the measurement date. In order to increase consistency and
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
comparability in fair value measurements, a fair value hierarchy has been established that prioritizes observable and unobservable inputs used to measure fair value into three broad levels, which are described below:
| |
• | Level 1: Quoted prices (unadjusted) in active markets that are accessible at the measurement date for identical assets or liabilities. The fair value hierarchy gives the highest priority to Level 1 inputs. |
| |
• | Level 2: Observable market-based inputs other than quoted prices in active markets for identical assets or liabilities. |
| |
• | Level 3: Unobservable inputs are used when little or no market data is available. The fair value hierarchy gives the lowest priority to Level 3 inputs. |
In determining fair value, the Company utilizes valuation techniques that maximize the use of observable inputs and minimize the use of unobservable inputs to the extent possible, as well as considers counterparty credit risk in its assessment of fair value.
Financial assets and liabilities carried at fair value are classified in the tables below in one of the three categories described above:
|
| | | | | | | | | | | | | | | |
| March 31, 2017 |
(In millions) | Level 1 | | Level 2 | | Level 3 | | Total |
Recurring fair value measurements | | | | | | | |
Financial Assets | | | | | | | |
Cash equivalents: | | | | | | | |
Money market funds | $ | 275.8 |
| | $ | — |
| | $ | — |
| | $ | 275.8 |
|
Total cash equivalents | 275.8 |
| | — |
| | — |
| | 275.8 |
|
Trading securities: | | | | | | | |
Equity securities — exchange traded funds | 30.7 |
| | — |
| | — |
| | 30.7 |
|
Total trading securities | 30.7 |
| | — |
| | — |
| | 30.7 |
|
Available-for-sale fixed income investments: | | | | | | | |
Corporate bonds | — |
| | 18.3 |
| | — |
| | 18.3 |
|
U.S. Treasuries | — |
| | 6.0 |
| | — |
| | 6.0 |
|
Agency mortgage-backed securities | — |
| | 3.8 |
| | — |
| | 3.8 |
|
Asset backed securities | — |
| | 1.6 |
| | — |
| | 1.6 |
|
Other | — |
| | 2.2 |
| | — |
| | 2.2 |
|
Total available-for-sale fixed income investments | — |
| | 31.9 |
| | — |
| | 31.9 |
|
Available-for-sale equity securities: | | | | | | | |
Marketable securities | 59.4 |
| | — |
| | — |
| | 59.4 |
|
Total available-for-sale equity securities | 59.4 |
| | — |
| | — |
| | 59.4 |
|
Foreign exchange derivative assets | — |
| | 64.4 |
|
| — |
|
| 64.4 |
|
Interest rate swap derivative assets | — |
| | 23.9 |
| | — |
| | 23.9 |
|
Total assets at recurring fair value measurement | $ | 365.9 |
|
| $ | 120.2 |
|
| $ | — |
|
| $ | 486.1 |
|
Financial Liabilities | | | | | | | |
Foreign exchange derivative liabilities | $ | — |
| | $ | 8.5 |
| | $ | — |
| | $ | 8.5 |
|
Contingent consideration | — |
| | — |
| | 566.0 |
| | 566.0 |
|
Total liabilities at recurring fair value measurement | $ | — |
| | $ | 8.5 |
| | $ | 566.0 |
| | $ | 574.5 |
|
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
|
| | | | | | | | | | | | | | | |
| December 31, 2016 |
(In millions) | Level 1 | | Level 2 | | Level 3 | | Total |
Recurring fair value measurements | | | | | | | |
Financial Assets | | | | | | | |
Cash equivalents: | | | | | | | |
Money market funds | $ | 433.7 |
| | $ | — |
| | $ | — |
| | $ | 433.7 |
|
Total cash equivalents | 433.7 |
| | — |
| | — |
| | 433.7 |
|
Trading securities: | | | | | | | |
Equity securities — exchange traded funds | 29.6 |
| | — |
| | — |
| | 29.6 |
|
Total trading securities | 29.6 |
| | — |
| | — |
| | 29.6 |
|
Available-for-sale fixed income investments: | | | | | | | |
Corporate bonds | — |
| | 17.5 |
| | — |
| | 17.5 |
|
U.S. Treasuries | — |
| | 6.0 |
| | — |
| | 6.0 |
|
Agency mortgage-backed securities | — |
| | 4.0 |
| | — |
| | 4.0 |
|
Asset backed securities | — |
| | 1.6 |
| | — |
| | 1.6 |
|
Other | — |
| | 2.3 |
| | — |
| | 2.3 |
|
Total available-for-sale fixed income investments | — |
| | 31.4 |
| | — |
| | 31.4 |
|
Available-for-sale equity securities: | | | | | | | |
Marketable securities | 52.3 |
| | — |
| | — |
| | 52.3 |
|
Total available-for-sale equity securities | 52.3 |
| | — |
| | — |
| | 52.3 |
|
Foreign exchange derivative assets | — |
| | 35.9 |
| | — |
| | 35.9 |
|
Interest rate swap derivative assets | — |
| | 26.2 |
| | — |
| | 26.2 |
|
Total assets at recurring fair value measurement | $ | 515.6 |
| | $ | 93.5 |
| | $ | — |
| | $ | 609.1 |
|
Financial Liabilities | | | | | | | |
Foreign exchange derivative liabilities | $ | — |
| | $ | 15.3 |
| | $ | — |
| | $ | 15.3 |
|
Contingent consideration | — |
| | — |
| | 564.6 |
| | 564.6 |
|
Total liabilities at recurring fair value measurement | $ | — |
| | $ | 15.3 |
| | $ | 564.6 |
| | $ | 579.9 |
|
For financial assets and liabilities that utilize Level 2 inputs, the Company utilizes both direct and indirect observable price quotes, including the LIBOR yield curve, foreign exchange forward prices and bank price quotes. Below is a summary of valuation techniques for Level 1 and Level 2 financial assets and liabilities:
| |
• | Cash equivalents — valued at observable net asset value prices. |
| |
• | Trading securities — valued at the active quoted market price from broker or dealer quotations or transparent pricing sources at the reporting date. |
| |
• | Available-for-sale fixed income investments — valued at the quoted market price from broker or dealer quotations or transparent pricing sources at the reporting date. |
| |
• | Available-for-sale equity securities — valued using quoted stock prices from public exchanges at the reporting date. |
| |
• | Interest rate swap derivative assets and liabilities — valued using the LIBOR/EURIBOR yield curves at the reporting date. Counterparties to these contracts are highly rated financial institutions. |
| |
• | Foreign exchange derivative assets and liabilities — valued using quoted forward foreign exchange prices and spot rates at the reporting date. Counterparties to these contracts are highly rated financial institutions. |
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
Contingent Consideration
The fair value measurement of contingent consideration is determined using Level 3 inputs. The Company’s contingent consideration represents a component of the total purchase consideration for the respiratory delivery platform, the acquisition of Agila Specialties (“Agila”), the acquisition of certain female healthcare businesses from Famy Care Limited (such businesses “Jai Pharma Limited”), the acquisition of the Topicals Business and certain other acquisitions. The measurement is calculated using unobservable inputs based on the Company’s own assumptions. For the respiratory delivery platform, Jai Pharma Limited, the Topicals Business and certain other acquisitions, significant unobservable inputs in the valuation include the probability and timing of future development and commercial milestones and future profit sharing payments. When valuing the contingent consideration related to the respiratory delivery platform and Jai Pharma Limited, the value of the obligations are derived from a probability assessment based on expectations of when certain milestones or profit share payments occur which are discounted using a market rate of return. At March 31, 2017 and December 31, 2016, discount rates ranging from 0.9% to 10.0% were utilized in the valuations. Significant changes in unobservable inputs could result in material changes to the contingent consideration liability.
A rollforward of the activity in the Company’s fair value of contingent consideration from December 31, 2016 to March 31, 2017 is as follows:
|
| | | | | | | | | | | |
(In millions) | Current Portion (1) | | Long-Term Portion (2) | | Total Contingent Consideration |
Balance at December 31, 2016 | $ | 256.9 |
| | $ | 307.7 |
| | $ | 564.6 |
|
Payments | (16.1 | ) | | (0.2 | ) | | (16.3 | ) |
Accretion | — |
| | 7.8 |
| | 7.8 |
|
Fair value loss(3) | 4.0 |
| | 5.9 |
| | 9.9 |
|
Balance at March 31, 2017 | $ | 244.8 |
| | $ | 321.2 |
| | $ | 566.0 |
|
| |
(1) | Included in other current liabilities on the Condensed Consolidated Balance Sheets. |
| |
(2) | Included in other long-term obligations on the Condensed Consolidated Balance Sheets. |
| |
(3) | Included in litigation settlements and other contingencies, net in the Condensed Consolidated Statements of Operations. |
2017 Changes to Contingent Consideration: During the three months ended March 31, 2017, the Company recorded a fair value loss of $9.9 million related to Jai Pharma Limited contingent consideration. In addition, the Company made payments of approximately $12.5 million related to the settlement reached with Strides Arcolab Limited in November 2016.
Although the Company has not elected the fair value option for other financial assets and liabilities, any future transacted financial asset or liability will be evaluated for the fair value election.
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
Long-Term Debt
A summary of long-term debt is as follows: |
| | | | | | | | | | |
(In millions) | Coupon | | March 31,
2017 | | December 31,
2016 |
Current portion of long-term debt: | | | | | |
Meda Bank Loans (a) | | | $ | 222.9 |
| | $ | 219.6 |
|
Other | | | 4.3 |
| | 3.7 |
|
Current portion of long-term debt | | | $ | 227.2 |
| | $ | 223.3 |
|
| | | | | |
Non-current portion of long-term debt: | | | | | |
2016 Term Loans (b) ** | | | $ | 1,050.0 |
| | $ | 1,600.0 |
|
Meda Medium Term Notes (c) | | | 148.6 |
| | 146.4 |
|
2018 Euro Senior Notes (d) ** | | | 532.8 |
| | 526.0 |
|
2018 Senior Notes (e) * | 2.600 | % | | 649.6 |
| | 649.6 |
|
2018 Senior Notes (e) ** | 3.000 | % | | 499.6 |
| | 499.6 |
|
2019 Senior Notes (f) ** | 2.500 | % | | 999.2 |
| | 999.1 |
|
2019 Senior Notes (g) * | 2.550 | % | | 499.5 |
| | 499.5 |
|
2020 Euro Senior Notes (h) ** | 1.250 | % | | 796.0 |
| | 785.7 |
|
2020 Senior Notes (i) ** | 3.750 | % | | 499.9 |
| | 499.9 |
|
2021 Senior Notes (j) ** | 3.150 | % | | 2,247.8 |
| | 2,247.7 |
|
2023 Senior Notes (g) * | 3.125 | % | | 772.9 |
| | 775.3 |
|
2023 Senior Notes (k) * | 4.200 | % | | 498.6 |
| | 498.6 |
|
2024 Euro Senior Notes (l)** | 2.250 | % | | 1,062.8 |
| | 1,049.2 |
|
2026 Senior Notes (m) ** | 3.950 | % | | 2,233.9 |
| | 2,233.5 |
|
2028 Euro Senior Notes (n) ** | 3.125 | % | | 791.3 |
| | 781.1 |
|
2043 Senior Notes (o) * | 5.400 | % | | 497.1 |
| | 497.0 |
|
2046 Senior Notes (p) ** | 5.250 | % | | 999.8 |
| | 999.8 |
|
Other | | | 7.1 |
| | 7.1 |
|
Deferred financing fees | | | (85.7 | ) | | (92.2 | ) |
Long-term debt | | | $ | 14,700.8 |
| | $ | 15,202.9 |
|
| |
(a) | Represents a bank loan of 2.0kr billion with AB Svensk Exportkredit (publ), as lender (“Svensk Exportkredit”), which matures in October 2017, and accordingly is included in current portion of long-term debt and other long-term obligations in the Condensed Consolidated Balance Sheets at March 31, 2017 and December 31, 2016. |
| |
(b) | The 2016 Term Loans mature on November 22, 2019 and bear interest at LIBOR (determined in accordance with the 2016 Term Credit Agreement) plus 1.375% per annum. At March 31, 2017, the weighted average interest rate of the 2016 Term Loans was approximately 2.35%. |
| |
(c) | Swedish medium term notes (“MTN”) program with an upper limit of 7kr billion. Of the total amount outstanding of 1.3kr billion, 588.0kr million matures on April 5, 2018 and 745.0kr million matures on May 21, 2019. At March 31, 2017, the weighted average interest rate of the MTNs was approximately 2.01%. |
| |
(d) | Instrument bears interest at a rate of three-month EURIBOR plus 0.870% per annum, reset quarterly. |
| |
(e) | Instrument is callable by the Company at any time at the greater of 100% of the principal amount and the sum of the present values of the remaining scheduled payments of principal and interest discounted at the U.S. Treasury rate plus 0.30% plus, in each case, accrued and unpaid interest. |
| |
(f) | Instrument is callable by the Company at any time at the greater of 100% of the principal amount and the sum of the present values of the remaining scheduled payments of principal and interest discounted at the U.S. Treasury rate plus 0.25% plus, in each case, accrued and unpaid interest. |
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
| |
(g) | Instrument is callable by the Company at any time at the greater of 100% of the principal amount and the sum of the present values of the remaining scheduled payments of principal and interest discounted at the U.S. Treasury rate plus 0.20% plus, in each case, accrued and unpaid interest. |
| |
(h) | Instrument is callable by the Company at any time prior to the date that is one month prior to the instrument’s maturity date at the greater of 100% of the principal amount and the sum of the present values of the remaining scheduled payments of principal and interest discounted to the redemption date on an annual basis, at a rate equal to the applicable Bund Rate (as defined in the Euro Notes Indenture), plus 0.30% plus, in each case, accrued and unpaid interest. On or after such date, the instrument is callable by the Company at 100% of the principal amount plus accrued and unpaid interest. |
| |
(i) | Instrument is callable by the Company at any time prior to the date that is one month prior to the instrument’s maturity date at the greater of 100% of the principal amount and the sum of the present values of the remaining scheduled payments of principal and interest discounted at the U.S. Treasury rate plus 0.35% plus, in each case, accrued and unpaid interest. On or after such date, the instrument is callable by the Company at 100% of the principal amount plus accrued and unpaid interest. |
| |
(j) | Instrument is callable by the Company at any time prior to the date that is one month prior to the instrument’s maturity date at the greater of 100% of the principal amount and the sum of the present values of the remaining scheduled payments of principal and interest discounted at the U.S. Treasury rate plus 0.30% plus, in each case, accrued and unpaid interest. On or after such date, the instrument is callable by the Company at 100% of the principal amount plus accrued and unpaid interest. |
| |
(k) | Instrument is callable by the Company at any time prior to August 29, 2023 at the greater of 100% of the principal amount and the sum of the present values of the remaining scheduled payments of principal and interest discounted at the U.S. Treasury rate plus 0.25% plus, in each case, accrued and unpaid interest. On or after such date, the instrument is callable by the Company at 100% of the principal amount plus accrued and unpaid interest. |
| |
(l) | Instrument is callable by the Company at any time prior to the date that is two months prior to the instrument’s maturity date at the greater of 100% of the principal amount and the sum of the present values of the remaining scheduled payments of principal and interest discounted to the redemption date on an annual basis, at a rate equal to the applicable Bund Rate (as defined in the Euro Notes Indenture), plus 0.35% plus, in each case, accrued and unpaid interest. On or after such date, the instrument is callable by the Company at 100% of the principal amount plus accrued and unpaid interest. |
| |
(m) | Instrument is callable by the Company at any time prior to the date that is three months prior to the instrument’s maturity date at the greater of 100% of the principal amount and the sum of the present values of the remaining scheduled payments of principal and interest discounted at the U.S. Treasury rate plus 0.35% plus, in each case, accrued and unpaid interest. On or after such date, the instrument is callable by the Company at 100% of the principal amount plus accrued and unpaid interest. |
| |
(n) | Instrument is callable by the Company at any time prior to the date that is three months prior to the instrument’s maturity date at the greater of 100% of the principal amount and the sum of the present values of the remaining scheduled payments of principal and interest discounted to the redemption date on an annual basis, at a rate equal to the applicable Bund Rate (as defined in the Euro Notes Indenture), plus 0.45% plus, in each case, accrued and unpaid interest. On or after such date, the instrument is callable by the Company at 100% of the principal amount plus accrued and unpaid interest. |
| |
(o) | Instrument is callable by the Company at any time prior to May 29, 2043 at the greater of 100% of the principal amount and the sum of the present values of the remaining scheduled payments of principal and interest discounted at the U.S. Treasury rate plus 0.25% plus, in each case, accrued and unpaid interest. On or after such date, the instrument is callable by the Company at 100% of the principal amount plus accrued and unpaid interest. |
| |
(p) | Instrument is callable by the Company at any time prior to the date that is six months prior to the instrument’s maturity date at the greater of 100% of the principal amount and the sum of the present values of the remaining scheduled payments of principal and interest discounted at the U.S. Treasury rate plus 0.40% plus, in each case, accrued and unpaid interest. On or after such date, the instrument is callable by the Company at 100% of the principal amount plus accrued and unpaid interest. |
| |
* | Instrument was issued by Mylan Inc. |
| |
** | Instrument was issued by Mylan N.V. |
Receivables Facility
The Receivables Facility has a committed balance of $400 million, although from time-to-time, the available amount of the Receivables Facility may be less than $400 million based on accounts receivable concentration limits and other
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
eligibility requirements. As of March 31, 2017 and December 31, 2016, the Company had no short-term borrowings under the Receivables Facility in the Condensed Consolidated Balance Sheets.
2016 Revolving Credit Agreement
On November 22, 2016, the Company entered into a revolving credit agreement (the “2016 Revolving Credit Agreement”) among the Company, as borrower, Mylan Inc., as a guarantor (the “Guarantor”), certain lenders and issuing banks and Bank of America, N.A., as the administrative agent (in such capacity, the “Revolving Administrative Agent”). The 2016 Revolving Credit Agreement contains a revolving credit facility (the “2016 Revolving Facility”) under which the Company may obtain extensions of credit in an aggregate principal amount not to exceed $2.0 billion, subject to the satisfaction of customary conditions, in U.S. Dollars or alternative currencies including Euro, Sterling, Yen and any other currency that is approved by the Revolving Administrative Agent and each lender under the 2016 Revolving Facility. The 2016 Revolving Facility includes a $200 million subfacility for the issuance of letters of credit and a $175 million sublimit for swingline borrowings.
The current interest rate under the 2016 Revolving Facility is LIBOR (determined in accordance with the 2016 Revolving Credit Agreement) plus 1.200% per annum, if the Company chooses to make LIBOR borrowings, or at a base rate (determined in accordance with the 2016 Revolving Credit Agreement) plus 0.200% per annum. In addition, the 2016 Revolving Facility has a facility fee which is currently 0.175% of the daily amount of the aggregate revolving commitments. The applicable margins over LIBOR and the base rate for the 2016 Revolving Facility can fluctuate based on the long term unsecured senior, non-credit enhanced debt rating of the Company by S&P Global Ratings. Moody’s Investors Service, Inc. and Fitch Ratings, Inc.
Amounts drawn on the 2016 Revolving Facility become due and payable on November 22, 2021 and may be voluntarily prepaid without penalty or premium, other than customary breakage costs related to prepayments of LIBOR borrowings. At March 31, 2017 and December 31, 2016, the Company had no amounts outstanding under the 2016 Revolving Facility.
2016 Term Credit Agreement
On November 22, 2016, the Company entered into a term loan credit agreement (the “2016 Term Credit Agreement”) among the Company, as borrower, the Guarantor, as a guarantor, certain lenders and Goldman Sachs Bank USA, as administrative agent pursuant to which the Company borrowed $2.0 billion in term loans denominated in U.S. dollars (the “2016 Term Loans”).
The 2016 Term Loans currently bear interest at LIBOR (determined in accordance with the 2016 Term Credit Agreement) plus 1.375% per annum, if the Company chooses to make LIBOR borrowings, or at a base rate (determined in accordance with the 2016 Term Credit Agreement) plus 0.375% per annum. The applicable margins over LIBOR and the base rate for the 2016 Term Loans can fluctuate based on the long term unsecured senior, non-credit enhanced debt rating of the Company by the S&P Global Ratings, Moody’s Investors Service, Inc. and Fitch Ratings, Inc.
The 2016 Term Loans mature on November 22, 2019 and have no required amortization payments. The entire principal amount on the 2016 Term Loans will be due and payable on November 22, 2019. The 2016 Term Loans may be voluntary prepaid without penalty or premium, other than customary breakage costs related to prepayments of LIBOR borrowings. The Company voluntarily prepaid $400 million of the aggregate principal amount of the 2016 Term Loans in the fourth quarter of 2016 and $550 million in the first quarter of 2017. As such, at March 31, 2017, the Company had an aggregate principal amount of $1.05 billion outstanding under the 2016 Term Loans. As a result of the voluntary prepayment, the Company expensed approximately $1.9 million of deferred financing costs during the three months ended March 31, 2017.
Euro Notes
On November 22, 2016, the Company completed its offering of the Floating Rate Euro Notes, the 2020 Euro Notes, the 2024 Euro Notes and the 2028 Euro Notes pursuant to the indenture dated November 22, 2016 (the “Euro Notes Indenture”) among the Company, Mylan Inc., as guarantor, and Citibank N.A., London Branch, as trustee, paying agent, transfer agent, registrar and calculation agent. The Floating Rate Euro Notes, the 2020 Euro Notes, 2024 Euro Notes and 2028 Euro Notes, together, are referred to as the “Euro Notes.”
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
At March 31, 2017, the outstanding balance of the Floating Rate Euro Notes, 2020 Euro Notes, 2024 Euro Notes and 2028 Euro Notes was approximately $532.8 million, $796.0 million, $1,062.8 million and $791.3 million, respectively, converted at the March 31, 2017 EUR to USD spot exchange rate. At March 31, 2017, discounts on the 2020 Euro Notes, 2024 Euro Notes and 2028 Euro Notes were approximately $3.1 million, $2.7 million and $7.9 million, respectively, converted at the March 31, 2017 EUR to USD spot exchange rate. During the three months ended March 31, 2017, the Company recorded mark-to-market losses related to the Floating Rate Euro Notes, 2020 Euro Notes, 2024 Euro Notes and 2028 Euro Notes of approximately $6.8 million, $10.2 million, $13.6 million and $10.2 million, respectively. Refer to Note 11 Financial Instruments and Risk Management for further discussion of the foreign currency risk management of these instruments.
Fair Value
At March 31, 2017 and December 31, 2016, the fair value of the Company’s 2.600% Senior Notes due 2018, 3.000% Senior Notes due 2018, 2.500% Senior Notes due 2019, 2.550% Senior Notes due 2019, 3.750% Senior Notes due 2020, 3.150% Senior Notes due 2021, 3.125% Senior Notes due 2023, 4.200% Senior Notes due 2023, 3.950% Senior Notes due 2026, 5.400% Senior Notes due 2043 and 5.250% Senior Notes due 2046 (collectively, the “Senior Notes”) and Euro Notes was approximately $13.7 billion and $13.2 billion, respectively. The fair values of the Senior Notes and Euro Notes were valued at quoted market prices from broker or dealer quotations and were classified as Level 2 in the fair value hierarchy. Based on quoted market rates of interest and maturity schedules of similar debt issues, the fair values of the Company’s 2016 Term Loans and the Meda borrowings, determined based on Level 2 inputs, approximate their carrying values at March 31, 2017 and December 31, 2016.
Mandatory minimum repayments remaining on the notional amount of outstanding long-term debt at March 31, 2017 are as follows for each of the periods ending December 31:
|
| | | |
(In millions) | Total |
2017 | $ | 223 |
|
2018 | 1,748 |
|
2019 | 2,633 |
|
2020 | 1,299 |
|
2021 | 2,250 |
|
Thereafter | 6,865 |
|
Total | $ | 15,018 |
|
| |
13. | Comprehensive Earnings |
Accumulated other comprehensive loss, as reflected on the Condensed Consolidated Balance Sheets, is comprised of the following:
|
| | | | | | | |
(In millions) | March 31,
2017 | | December 31,
2016 |
Accumulated other comprehensive loss: | | | |
Net unrealized gain on marketable securities, net of tax | $ | 19.4 |
| | $ | 14.5 |
|
Net unrecognized losses and prior service cost related to defined benefit plans, net of tax | (0.3 | ) | | (0.5 | ) |
Net unrecognized losses on derivatives in cash flow hedging relationships, net of tax | (17.3 | ) | | (38.6 | ) |
Net unrecognized losses on derivatives in net investment hedging relationships, net of tax | (11.3 | ) | | (1.4 | ) |
Foreign currency translation adjustment | (1,803.5 | ) | | (2,237.7 | ) |
| $ | (1,813.0 | ) | | $ | (2,263.7 | ) |
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
Components of accumulated other comprehensive loss, before tax, consist of the following, for the three months ended March 31, 2017 and 2016:
|
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Three Months Ended March 31, 2017 |
Gains and Losses on Derivatives in Cash Flow Hedging Relationships | | Gains and Losses on Net Investment Hedges | | Gains and Losses on Marketable Securities | | Defined Pension Plan Items | | Foreign Currency Translation Adjustment | | Totals |
(In millions) | Foreign Currency Forward Contracts | | Interest Rate Swaps | | Total | | | | | | | | | | |
Balance at December 31, 2016, net of tax | | | | | $ | (38.6 | ) | | $ | (1.4 | ) | | $ | 14.5 |
| | $ | (0.5 | ) | | $ | (2,237.7 | ) | | $ | (2,263.7 | ) |
Other comprehensive earnings (loss) before reclassifications, before tax | | | | | 25.4 |
| | (9.9 | ) | | 7.7 |
| | (0.3 | ) | | 434.2 |
| | 457.1 |
|
Amounts reclassified from accumulated other comprehensive earnings (loss), before tax: | | | | | | | | | | | | | | | |
Loss on foreign exchange forward contracts classified as cash flow hedges, included in net sales | 5.2 |
| | | | 5.2 |
| | | | | | | | | | 5.2 |
|
Loss on interest rate swaps classified as cash flow hedges, included in interest expense | | | 1.8 |
| | 1.8 |
| | | | | | | | | | 1.8 |
|
Amortization of prior service costs included in SG&A | | | | | | | | | | | 0.1 |
| | | | 0.1 |
|
Amortization of actuarial loss included in SG&A | | | | | | | | | | | 0.2 |
| | | | 0.2 |
|
Net other comprehensive earnings (loss), before tax | | | | | 32.4 |
| | (9.9 | ) | | 7.7 |
| | — |
| | 434.2 |
| | 464.4 |
|
Income tax provision (benefit) | | | | | 11.1 |
| | — |
| | 2.8 |
| | (0.2 | ) | | — |
| | 13.7 |
|
Balance at March 31, 2017, net of tax | | | | | $ | (17.3 | ) | | $ | (11.3 | ) | | $ | 19.4 |
| | $ | (0.3 | ) | | $ | (1,803.5 | ) | | $ | (1,813.0 | ) |
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
|
| | | | | | | | | | | | | | | | | | | | | | | | | |
| Three Months Ended March 31, 2016 |
Gains and Losses on Derivatives in Cash Flow Hedging Relationships | | Gains and Losses on Marketable Securities | | Defined Pension Plan Items | | Foreign Currency Translation Adjustment | | Totals |
(In millions) | Foreign Currency Forward Contracts | | Interest Rate Swaps | | Total | | | | | | | | |
Balance at December 31, 2015, net of tax | | | | | $ | (18.1 | ) | | $ | (1.0 | ) | | $ | (14.9 | ) | | $ | (1,730.3 | ) | | $ | (1,764.3 | ) |
Other comprehensive (loss) earnings before reclassifications, before tax | | | | | (39.4 | ) | | 4.4 |
| | (0.6 | ) | | 502.0 |
| | 466.4 |
|
Amounts reclassified from accumulated other comprehensive (loss) earnings, before tax: | | | | | | | | | | | | | |
Loss on foreign exchange forward contracts classified as cash flow hedges, included in net sales | (10.6 | ) | | | | (10.6 | ) | | | | | | | | (10.6 | ) |
Gain on interest rate swaps classified as cash flow hedges, included in interest expense | | | 0.9 |
| | 0.9 |
| | | | | | | | 0.9 |
|
Amortization of prior service costs included in SG&A | | | | | | | | | 0.1 |
| | | | 0.1 |
|
Amortization of actuarial gain included in SG&A | | | | | | | | | 0.2 |
| | | | 0.2 |
|
Net other comprehensive (loss) earnings, before tax | | | | | (49.1 | ) | | 4.4 |
| | (0.3 | ) | | 502.0 |
| | 457.0 |
|
Income tax (benefit) provision | | | | | (18.3 | ) | | 1.6 |
| | (0.1 | ) | | — |
| | (16.8 | ) |
Balance at March 31, 2016, net of tax | | | | | $ | (48.9 | ) | | $ | 1.8 |
| | $ | (15.1 | ) | | $ | (1,228.3 | ) | | $ | (1,290.5 | ) |
A summary of the changes in shareholders’ equity for the three months ended March 31, 2017 and 2016 is as follows:
|
| | | | | | | | | | | |
(In millions) | Total
Mylan N.V.
Shareholders' Equity | | Noncontrolling Interest | | Total |
December 31, 2016 | $ | 11,116.2 |
| | $ | 1.4 |
| | $ | 11,117.6 |
|
Net earnings | 66.4 |
| | — |
| | 66.4 |
|
Other comprehensive earnings, net of tax | 450.7 |
| | — |
| | 450.7 |
|
Stock option activity | 5.2 |
| | — |
| | 5.2 |
|
Share-based compensation expense | 23.1 |
| | — |
| | 23.1 |
|
Issuance of restricted stock, net of shares withheld | (5.6 | ) | | — |
| | (5.6 | ) |
Other | — |
| | (1.4 | ) | | (1.4 | ) |
March 31, 2017 | $ | 11,656.0 |
| | $ | — |
| | $ | 11,656.0 |
|
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
|
| | | | | | | | | | | |
(In millions) | Total
Mylan N.V.
Shareholders' Equity | | Noncontrolling Interest | | Total |
December 31, 2015 | $ | 9,764.4 |
| | $ | 1.4 |
| | $ | 9,765.8 |
|
Net earnings | 13.9 |
| | — |
| | 13.9 |
|
Other comprehensive earnings, net of tax | 473.8 |
| | — |
| | 473.8 |
|
Stock option activity | 3.5 |
| | — |
| | 3.5 |
|
Share-based compensation expense | 26.5 |
| | — |
| | 26.5 |
|
Issuance of restricted stock, net of shares withheld | (9.9 | ) | | — |
| | (9.9 | ) |
Tax benefit of stock option plans | 1.2 |
| | — |
| | 1.2 |
|
Other | — |
| | 0.1 |
| | 0.1 |
|
March 31, 2016 | $ | 10,273.4 |
| | $ | 1.5 |
| | $ | 10,274.9 |
|
As a result of our acquisition of Meda and the integration of our portfolio across our branded, generics and over-the-counter (“OTC”) platforms in all of our regions, effective October 1, 2016, the Company expanded its reportable segments. The Company has three reportable segments on a geographic basis as follows: North America, Europe and Rest of World. Our North America segment is made up of our operations in the U.S. and Canada and includes the operations of our previously reported Specialty segment. Our Europe segment is made up of our operations in 35 countries within the region. Our Rest of World segment is primarily made up of our operations in India, Australia, Japan and New Zealand. Also included in our Rest of World segment are our operations in emerging markets, which includes countries in Africa (including South Africa) as well as Brazil and other countries throughout Asia and the Middle East. Comparative segment financial information has been recast for prior periods to conform to this revised segment reporting.
The Company’s chief operating decision maker is the Chief Executive Officer, who evaluates the performance of its segments based on total revenues and segment profitability. Segment profitability represents segment gross profit less direct R&D expenses and direct SG&A expenses. Certain general and administrative and R&D expenses not allocated to the segments, net charges for litigation settlements and other contingencies, impairment charges and other expenses not directly attributable to the segments and certain intercompany transactions, including eliminations, are reported in Corporate/Other. Additionally, amortization of intangible assets and other purchase accounting related items, as well as certain other significant special items, are included in Corporate/Other. Items below the earnings from operations line on the Company’s Condensed Consolidated Statements of Operations are not presented by segment, since they are excluded from the measure of segment profitability. The Company does not report depreciation expense, total assets and capital expenditures by segment, as such information is not used by the chief operating decision maker.
The accounting policies of the segments are the same as those described in the “Summary of Significant Accounting Policies” included in Mylan N.V.’s Annual Report on Form 10-K for the year ended December 31, 2016, as amended. Intersegment revenues are accounted for at current market values and are eliminated at the consolidated level.
Presented in the table below is segment information for the periods identified and a reconciliation of segment information to total consolidated information.
|
| | | | | | | | | | | | | | | | | | | |
(In millions) | North America | | Europe | | Rest of World | | Corporate /
Other | | Consolidated |
Three Months Ended March 31, 2017 | | | | | | | | | |
Third party net sales | $ | 1,214.9 |
| | $ | 892.0 |
| | $ | 580.5 |
| | $ | — |
| | $ | 2,687.4 |
|
Other revenue | 23.4 |
| | 6.7 |
| | 2.0 |
| | — |
| | 32.1 |
|
Intersegment | 13.1 |
| | 42.9 |
| | 99.1 |
| | (155.1 | ) | | — |
|
Total | $ | 1,251.4 |
| | $ | 941.6 |
| | $ | 681.6 |
| | $ | (155.1 | ) | | $ | 2,719.5 |
|
| | | | | | | | | |
Segment profitability | $ | 589.7 |
| | $ | 233.8 |
| | $ | 76.6 |
| | $ | (672.9 | ) | | $ | 227.2 |
|
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
|
| | | | | | | | | | | | | | | | | | | |
(In millions) | North America | | Europe | | Rest of World | | Corporate /
Other | | Consolidated |
Three Months Ended March 31, 2016 | | | | | | | | | |
Third party net sales | $ | 1,157.5 |
| | $ | 584.3 |
| | $ | 434.3 |
| | $ | — |
| | $ | 2,176.1 |
|
Other revenue | 14.0 |
| | 0.3 |
| | 1.1 |
| | — |
| | 15.2 |
|
Intersegment | 6.3 |
| | 25.2 |
| | 85.1 |
| | (116.6 | ) | | — |
|
Total | $ | 1,177.8 |
| | $ | 609.6 |
| | $ | 520.5 |
| | $ | (116.6 | ) | | $ | 2,191.3 |
|
| | | | | | | | | |
Segment profitability | $ | 573.8 |
| | $ | 124.6 |
| | $ | 29.6 |
| | $ | (622.4 | ) | | $ | 105.6 |
|
The following tables present condensed consolidating financial information for (a) Mylan N.V., the issuer of the 3.000% Senior Notes due 2018, 2.500% Senior Notes due 2019, 3.750% Senior Notes due 2020, 3.150% Senior Notes due 2021, 3.950% Senior Notes due 2026 and 5.250% Senior Notes due 2046 (collectively, the “Mylan N.V. Senior Notes”), which are guaranteed on a senior unsecured basis by Mylan Inc.; (b) Mylan Inc., the issuer of the 2.600% Senior Notes due 2018, 2.550% Senior Notes due 2019, 3.125% Senior Notes due 2023, 4.200% Senior Notes due 2023 and 5.400% Senior Notes due 2043 (collectively, the “Mylan Inc. Senior Notes”), which are guaranteed on a senior unsecured basis by Mylan N.V.; and (c) all other subsidiaries of the Company on a combined basis, none of which guarantee the Mylan N.V. Senior Notes or guarantee the Mylan Inc. Senior Notes (“Non-Guarantor Subsidiaries”). The consolidating adjustments primarily relate to eliminations of investments in subsidiaries and intercompany balances and transactions. The condensed consolidating financial statements present investments in subsidiaries using the equity method of accounting.
The following financial information presents the unaudited Condensed Consolidating Statements of Operations for the three months ended March 31, 2017 and 2016, the unaudited Condensed Consolidating Statements of Comprehensive Earnings for the three months ended March 31, 2017 and 2016, the unaudited Condensed Consolidating Balance Sheets as of March 31, 2017 and December 31, 2016 and the unaudited Condensed Consolidating Statements of Cash Flows for the three months ended March 31, 2017 and 2016. This unaudited condensed consolidating financial information has been prepared and presented in accordance with SEC Regulation S-X Rule 3-10 “Financial Statements of Guarantors and Issuers of Guaranteed Securities Registered or Being Registered.”
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
UNAUDITED CONDENSED CONSOLIDATING STATEMENT OF OPERATIONS
Three Months Ended March 31, 2017
|
| | | | | | | | | | | | | | | | | | | | | | | |
(In millions) | Mylan N.V. | | Mylan Inc. | | Guarantor Subsidiaries | | Non-Guarantor Subsidiaries | | Eliminations | | Consolidated |
Revenues: | | | | | | | | | | | |
Net sales | $ | — |
| | $ | — |
| | $ | — |
| | $ | 2,687.4 |
| | $ | — |
| | $ | 2,687.4 |
|
Other revenues | — |
| | — |
| | — |
| | 32.1 |
| | — |
| | 32.1 |
|
Total revenues | — |
| | — |
| | — |
| | 2,719.5 |
| | — |
| | 2,719.5 |
|
Cost of sales | — |
| | — |
| | — |
| | 1,634.5 |
| | — |
| | 1,634.5 |
|
Gross profit | — |
| | — |
| | — |
| | 1,085.0 |
| | — |
| | 1,085.0 |
|
Operating expenses: | | | | | | | | | | | |
Research and development | — |
| | — |
| | — |
| | 217.5 |
| | — |
| | 217.5 |
|
Selling, general and administrative | 12.6 |
| | 156.5 |
| | — |
| | 462.2 |
| | — |
| | 631.3 |
|
Litigation settlements and other contingencies, net | — |
| | — |
| | — |
| | 9.0 |
| | — |
| | 9.0 |
|
Total operating expenses | 12.6 |
| | 156.5 |
| | — |
| | 688.7 |
| | — |
| | 857.8 |
|
(Losses) earnings from operations | (12.6 | ) | | (156.5 | ) | | — |
| | 396.3 |
| | — |
| | 227.2 |
|
Interest expense | 97.6 |
| | 25.4 |
| | — |
| | 15.2 |
| | — |
| | 138.2 |
|
Other (income) expense, net | (95.5 | ) | | (57.3 | ) | | — |
| | 170.2 |
| | — |
| | 17.4 |
|
(Loss) earnings before income taxes | (14.7 | ) | | (124.6 | ) | | — |
| | 210.9 |
| | — |
| | 71.6 |
|
Income tax provision (benefit) | (1.6 | ) | | 3.2 |
| | — |
| | 3.6 |
| | — |
| | 5.2 |
|
Earnings (loss) of equity interest subsidiaries | 79.5 |
| | 214.0 |
| | — |
| | — |
| | (293.5 | ) | | — |
|
Net earnings | $ | 66.4 |
| | $ | 86.2 |
| | $ | — |
| | $ | 207.3 |
| | $ | (293.5 | ) | | $ | 66.4 |
|
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
UNAUDITED CONDENSED CONSOLIDATING STATEMENT OF OPERATIONS
Three Months Ended March 31, 2016
|
| | | | | | | | | | | | | | | | | | | | | | | |
(In millions) | Mylan N.V. | | Mylan Inc. | | Guarantor Subsidiaries | | Non-Guarantor Subsidiaries | | Eliminations | | Consolidated |
Revenues: | | | | | | | | | | | |
Net sales | $ | — |
| | $ | — |
| | $ | — |
| | $ | 2,176.1 |
| | $ | — |
| | $ | 2,176.1 |
|
Other revenues | — |
| | — |
| | — |
| | 15.2 |
| | — |
| | 15.2 |
|
Total revenues | — |
| | — |
| | — |
| | 2,191.3 |
| | — |
| | 2,191.3 |
|
Cost of sales | — |
| | — |
| | — |
| | 1,284.3 |
| | — |
| | 1,284.3 |
|
Gross profit | — |
| | — |
| | — |
| | 907.0 |
| | — |
| | 907.0 |
|
Operating expenses: | | | | | | | | | | | |
Research and development | — |
| | — |
| | — |
| | 253.6 |
| | — |
| | 253.6 |
|
Selling, general and administrative | 13.2 |
| | 176.0 |
| | — |
| | 360.1 |
| | — |
| | 549.3 |
|
Litigation settlements and other contingencies, net | — |
| | — |
| | — |
| | (1.5 | ) | | — |
| | (1.5 | ) |
Total operating expenses | 13.2 |
| | 176.0 |
| | — |
| | 612.2 |
| | — |
| | 801.4 |
|
(Losses) earnings from operations | (13.2 | ) | | (176.0 | ) | | — |
| | 294.8 |
| | — |
| | 105.6 |
|
Interest expense | 13.3 |
| | 41.5 |
| | — |
| | 15.5 |
| | — |
| | 70.3 |
|
Other expense, net | — |
| | — |
| | — |
| | 16.3 |
| | — |
| | 16.3 |
|
(Losses) earnings from operations | (26.5 | ) | | (217.5 | ) | | — |
| | 263.0 |
| | — |
| | 19.0 |
|
Income tax provision (benefit) | — |
| | 9.0 |
| | — |
| | (3.9 | ) | | — |
| | 5.1 |
|
Earnings of equity interest subsidiaries | 40.4 |
| | 264.8 |
| | — |
| | — |
| | (305.2 | ) | | — |
|
Net earnings | $ | 13.9 |
| | $ | 38.3 |
| | $ | — |
| | $ | 266.9 |
| | $ | (305.2 | ) | | $ | 13.9 |
|
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
UNAUDITED CONDENSED CONSOLIDATING STATEMENT OF COMPREHENSIVE EARNINGS
Three Months Ended March 31, 2017
|
| | | | | | | | | | | | | | | | | | | | | | | |
(In millions) | Mylan N.V. | | Mylan Inc. | | Guarantor Subsidiaries | | Non-Guarantor Subsidiaries | | Eliminations | | Consolidated |
Net earnings | $ | 66.4 |
| | $ | 86.2 |
| | $ | — |
| | $ | 207.3 |
| | $ | (293.5 | ) | | $ | 66.4 |
|
Other comprehensive earnings (loss), before tax: | | | | | | | | | | | |
Foreign currency translation adjustment | 434.2 |
| | — |
| | — |
| | 434.2 |
| | (434.2 | ) | | 434.2 |
|
Change in unrecognized gain (loss) and prior service cost related to defined benefit plans | — |
| | 0.1 |
| | — |
| | (0.1 | ) | | — |
| | — |
|
Net unrecognized gain on derivatives in cash flow hedging relationships | 32.4 |
| | 1.8 |
| | — |
| | 30.6 |
| | (32.4 | ) | | 32.4 |
|
Net unrecognized loss on derivatives in net investment hedging relationships | (9.9 | ) | | — |
| | — |
| | (9.9 | ) | | 9.9 |
| | (9.9 | ) |
Net unrealized gain (loss) on marketable securities | 7.7 |
| | 7.8 |
| | — |
| | (0.1 | ) | | (7.7 | ) | | 7.7 |
|
Other comprehensive earnings, before tax | 464.4 |
| | 9.7 |
| | — |
| | 454.7 |
| | (464.4 | ) | | 464.4 |
|
Income tax provision | 13.7 |
| | (3.6 | ) | | — |
| | 17.3 |
| | (13.7 | ) | | 13.7 |
|
Other comprehensive earnings, net of tax | 450.7 |
| | 13.3 |
| | — |
| | 437.4 |
| | (450.7 | ) | | 450.7 |
|
Comprehensive earnings (loss) | $ | 517.1 |
| | $ | 99.5 |
| | $ | — |
| | $ | 644.7 |
| | $ | (744.2 | ) | | $ | 517.1 |
|
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
UNAUDITED CONDENSED CONSOLIDATING STATEMENT OF COMPREHENSIVE EARNINGS
Three Months Ended March 31, 2016
|
| | | | | | | | | | | | | | | | | | | | | | | |
(In millions) | Mylan N.V. | | Mylan Inc. | | Guarantor Subsidiaries | | Non-Guarantor Subsidiaries | | Eliminations | | Consolidated |
Net earnings | $ | 13.9 |
| | $ | 38.3 |
| | $ | — |
| | $ | 266.9 |
| | $ | (305.2 | ) | | $ | 13.9 |
|
Other comprehensive earnings (loss), before tax: | | | | | | | | | | | |
Foreign currency translation adjustment | 502.0 |
| | — |
| | — |
| | 502.0 |
| | (502.0 | ) | | 502.0 |
|
Change in unrecognized (loss) gain and prior service cost related to defined benefit plans | (0.3 | ) | | — |
| | — |
| | (0.3 | ) | | 0.3 |
| | (0.3 | ) |
Net unrecognized (loss) gain on derivatives | (49.1 | ) | | (58.4 | ) | | — |
| | 9.3 |
| | 49.1 |
| | (49.1 | ) |
Net unrealized gain on marketable securities | 4.4 |
| | 3.8 |
| | — |
| | 0.6 |
| | (4.4 | ) | | 4.4 |
|
Other comprehensive earnings (loss), before tax | 457.0 |
| | (54.6 | ) | | — |
| | 511.6 |
| | (457.0 | ) | | 457.0 |
|
Income tax (benefit) provision | (16.8 | ) | | (20.2 | ) | | — |
| | 3.4 |
| | 16.8 |
| | (16.8 | ) |
Other comprehensive earnings (loss), net of tax | 473.8 |
| | (34.4 | ) | | — |
| | 508.2 |
| | (473.8 | ) | | 473.8 |
|
Comprehensive earnings | $ | 487.7 |
| | $ | 3.9 |
| | $ | — |
| | $ | 775.1 |
| | $ | (779.0 | ) | | $ | 487.7 |
|
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
UNAUDITED CONDENSED CONSOLIDATING BALANCE SHEET
As of March 31, 2017
|
| | | | | | | | | | | | | | | | | | | | | | | |
(In millions) | Mylan N.V. | | Mylan Inc. | | Guarantor Subsidiaries | | Non-Guarantor Subsidiaries | | Eliminations | | Consolidated |
ASSETS | | | | | | | | | | | |
Assets | | | | | | | | | | | |
Current assets: | | | | | | | | | | | |
Cash and cash equivalents | $ | — |
| | $ | 6.3 |
| | $ | — |
| | $ | 717.5 |
| | $ | — |
| | $ | 723.8 |
|
Accounts receivable, net | — |
| | 5.4 |
| | — |
| | 2,866.6 |
| | — |
| | 2,872.0 |
|
Inventories | — |
| | — |
| | — |
| | 2,547.8 |
| | — |
| | 2,547.8 |
|
Intercompany receivables | 220.1 |
| | 427.3 |
| | — |
| | 11,317.7 |
| | (11,965.1 | ) | | — |
|
Prepaid expenses and other current assets | 6.4 |
| | 245.9 |
| | — |
| | 669.6 |
| | — |
| | 921.9 |
|
Total current assets | 226.5 |
| | 684.9 |
| | — |
| | 18,119.2 |
| | (11,965.1 | ) | | 7,065.5 |
|
Property, plant and equipment, net | — |
| | 351.9 |
| | — |
| | 1,986.1 |
| | — |
| | 2,338.0 |
|
Investments in subsidiaries | 16,047.7 |
| | 8,589.3 |
| | — |
| | — |
| | (24,637.0 | ) | | — |
|
Intercompany notes and interest receivable | 7,555.4 |
| | 9,928.1 |
| | — |
| | 16.7 |
| | (17,500.2 | ) | | — |
|
Intangible assets, net | — |
| | — |
| | — |
| | 14,370.0 |
| | — |
| | 14,370.0 |
|
Goodwill | — |
| | 17.1 |
| | — |
| | 9,377.0 |
| | — |
| | 9,394.1 |
|
Other assets | 5.3 |
| | 37.4 |
| | — |
| | 1,062.3 |
| | — |
| | 1,105.0 |
|
Total assets | $ | 23,834.9 |
| | $ | 19,608.7 |
| | $ | — |
| | $ | 44,931.3 |
| | $ | (54,102.3 | ) | | $ | 34,272.6 |
|
| | | | | | | | | | | |
LIABILITIES AND EQUITY | | | | | | | | | | | |
Liabilities | | | | | | | | | | | |
Current liabilities: | | | | | | | | | | | |
Trade accounts payable | $ | 0.5 |
| | $ | 38.5 |
| | $ | — |
| | $ | 1,102.4 |
| | $ | — |
| | $ | 1,141.4 |
|
Short-term borrowings | — |
| | — |
| | — |
| | 31.0 |
| | — |
| | 31.0 |
|
Income taxes payable | — |
| | — |
| | — |
| | 31.0 |
| | — |
| | 31.0 |
|
Current portion of long-term debt and other long-term obligations | — |
| | 0.2 |
| | — |
| | 294.2 |
| | — |
| | 294.4 |
|
Intercompany payables | 427.3 |
| | 11,537.8 |
| | — |
| | — |
| | (11,965.1 | ) | | — |
|
Other current liabilities | 102.7 |
| | 354.3 |
| | — |
| | 2,569.4 |
| | — |
| | 3,026.4 |
|
Total current liabilities | 530.5 |
| | 11,930.8 |
| | — |
| | 4,028.0 |
| | (11,965.1 | ) | | 4,524.2 |
|
Long-term debt | 11,648.4 |
| | 2,896.7 |
| | — |
| | 155.7 |
| | — |
| | 14,700.8 |
|
Intercompany notes payable | — |
| | 3,449.4 |
| | — |
| | 14,050.8 |
| | (17,500.2 | ) | | — |
|
Other long-term obligations | — |
| | 57.9 |
| | — |
| | 3,333.7 |
| | — |
| | 3,391.6 |
|
Total liabilities | 12,178.9 |
| | 18,334.8 |
| | — |
| | 21,568.2 |
| | (29,465.3 | ) | | 22,616.6 |
|
| | | | | | | | | | | |
Total equity | 11,656.0 |
| | 1,273.9 |
| | — |
| | 23,363.1 |
| | (24,637.0 | ) | | 11,656.0 |
|
| | | | | | | | | | | |
Total liabilities and equity | $ | 23,834.9 |
| | $ | 19,608.7 |
| | $ | — |
| | $ | 44,931.3 |
| | $ | (54,102.3 | ) | | $ | 34,272.6 |
|
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
UNAUDITED CONDENSED CONSOLIDATING BALANCE SHEET
As of December 31, 2016
|
| | | | | | | | | | | | | | | | | | | | | | | |
(In millions) | Mylan N.V. | | Mylan Inc. | | Guarantor Subsidiaries | | Non-Guarantor Subsidiaries | | Eliminations | | Consolidated |
ASSETS | | | | | | | | | | | |
Assets | | | | | | | | | | | |
Current assets: | | | | | | | | | | | |
Cash and cash equivalents | $ | 0.3 |
| | $ | 12.3 |
| | $ | — |
| | $ | 986.2 |
| | $ | — |
| | $ | 998.8 |
|
Accounts receivable, net | — |
| | 12.3 |
| | — |
| | 3,298.6 |
| | — |
| | 3,310.9 |
|
Inventories | — |
| | — |
| | — |
| | 2,456.4 |
| | — |
| | 2,456.4 |
|
Intercompany receivables | 215.9 |
| | 416.0 |
| | — |
| | 10,506.6 |
| | (11,138.5 | ) | | — |
|
Prepaid expenses and other current assets | — |
| | 256.4 |
| | — |
| | 500.0 |
| | — |
| | 756.4 |
|
Total current assets | 216.2 |
| | 697.0 |
| | — |
| | 17,747.8 |
| | (11,138.5 | ) | | 7,522.5 |
|
Property, plant and equipment, net | — |
| | 360.3 |
| | — |
| | 1,961.9 |
| | — |
| | 2,322.2 |
|
Investments in subsidiaries | 15,606.2 |
| | 8,277.8 |
| | — |
| | — |
| | (23,884.0 | ) | | — |
|
Intercompany notes and interest receivable | 7,952.3 |
| | 9,817.3 |
| | — |
| | 16.7 |
| | (17,786.3 | ) | | — |
|
Intangible assets, net | — |
| | — |
| | — |
| | 14,447.8 |
| | — |
| | 14,447.8 |
|
Goodwill | — |
| | 17.1 |
| | — |
| | 9,214.8 |
| | — |
| | 9,231.9 |
|
Other assets | 5.2 |
| | 51.9 |
| | — |
| | 1,144.7 |
| | — |
| | 1,201.8 |
|
Total assets | $ | 23,779.9 |
| | $ | 19,221.4 |
| | $ | — |
| | $ | 44,533.7 |
| | $ | (52,808.8 | ) | | $ | 34,726.2 |
|
| | | | | | | | | | | |
LIABILITIES AND EQUITY | | | | | | | | | | | |
Liabilities | | | | | | | | | | | |
Current liabilities: | | | | | | | | | | | |
Trade accounts payable | $ | 3.9 |
| | $ | 69.6 |
| | $ | — |
| | $ | 1,274.6 |
| | $ | — |
| | $ | 1,348.1 |
|
Short-term borrowings | — |
| | — |
| | — |
| | 46.4 |
| | — |
| | 46.4 |
|
Income taxes payable | — |
| | — |
| | — |
| | 97.7 |
| | — |
| | 97.7 |
|
Current portion of long-term debt and other long-term obligations | — |
| | 0.2 |
| | — |
| | 289.8 |
| | — |
| | 290.0 |
|
Intercompany payables | 416.0 |
| | 10,722.5 |
| | — |
| | — |
| | (11,138.5 | ) | | — |
|
Other current liabilities | 90.9 |
| | 388.8 |
| | — |
| | 2,778.8 |
| | — |
| | 3,258.5 |
|
Total current liabilities | 510.8 |
| | 11,181.1 |
| | — |
| | 4,487.3 |
| | (11,138.5 | ) | | 5,040.7 |
|
Long-term debt | 12,151.5 |
| | 2,897.6 |
| | — |
| | 153.8 |
| | — |
| | 15,202.9 |
|
Intercompany notes payable | — |
| | 3,870.9 |
| | — |
| | 13,915.4 |
| | (17,786.3 | ) | | — |
|
Other long-term obligations | — |
| | 58.1 |
| | — |
| | 3,306.9 |
| | — |
| | 3,365.0 |
|
Total liabilities | 12,662.3 |
| | 18,007.7 |
| | — |
| | 21,863.4 |
| | (28,924.8 | ) | | 23,608.6 |
|
| | | | | | | | | | | |
Total equity | 11,117.6 |
| | 1,213.7 |
| | — |
| | 22,670.3 |
| | (23,884.0 | ) | | 11,117.6 |
|
| | | | | | | | | | | |
Total liabilities and equity | $ | 23,779.9 |
| | $ | 19,221.4 |
| | $ | — |
| | $ | 44,533.7 |
| | $ | (52,808.8 | ) | | $ | 34,726.2 |
|
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
UNAUDITED CONDENSED CONSOLIDATING STATEMENT OF CASH FLOWS
Three Months Ended March 31, 2017
|
| | | | | | | | | | | | | | | | | | | | | | | |
(In millions) | Mylan N.V. | | Mylan Inc. | | Guarantor Subsidiaries | | Non-Guarantor Subsidiaries | | Eliminations | | Consolidated |
Cash flows from operating activities: | | | | | | | | | | | |
Net cash (used in) provided by operating activities | $ | (27.4 | ) | | $ | (192.7 | ) | | $ | — |
| | $ | 673.0 |
| | $ | — |
| | $ | 452.9 |
|
Cash flows from investing activities: | | | | | | | | | | | |
Capital expenditures | — |
| | (18.3 | ) | | — |
| | (40.1 | ) | | — |
| | (58.4 | ) |
Change in restricted cash | — |
| | — |
| | — |
| | 12.7 |
| | — |
| | 12.7 |
|
Purchase of marketable securities | — |
| | — |
| | — |
| | (2.3 | ) | | — |
| | (2.3 | ) |
Proceeds from sale of assets | — |
| | — |
| | — |
| | 31.1 |
| | — |
| | 31.1 |
|
Proceeds from sale of marketable securities | — |
| | — |
| | — |
| | 2.3 |
| | — |
| | 2.3 |
|
Cash paid for acquisitions, net | (71.6 | ) | | — |
| | — |
| | — |
| | — |
| | (71.6 | ) |
Investments in affiliates | — |
| | (7.2 | ) | | — |
| | — |
| | 7.2 |
| | — |
|
Dividends from affiliates | 52.4 |
| | — |
| | — |
| | — |
| | (52.4 | ) | | — |
|
Loans to affiliates | (100.2 | ) | | (111.1 | ) | | — |
| | (977.5 | ) | | 1,188.8 |
| | — |
|
Repayments of loans from affiliates | 701.3 |
| | 0.3 |
| | — |
| | 188.8 |
| | (890.4 | ) | | — |
|
Payments for product rights and other, net | — |
| | (0.1 | ) | | — |
| | (77.8 | ) | | — |
| | (77.9 | ) |
Net cash (used in) provided by investing activities | 581.9 |
| | (136.4 | ) | | — |
| | (862.8 | ) | | 253.2 |
| | (164.1 | ) |
Cash flows from financing activities: | | | | | | | | | | | |
Payments of financing fees | (3.7 | ) | | — |
| | — |
| | — |
| | — |
| | (3.7 | ) |
Change in short-term borrowings, net | — |
| | — |
| | — |
| | (17.6 | ) | | — |
| | (17.6 | ) |
Payments of long-term debt | (550.0 | ) | | — |
| | — |
| | — |
| | — |
| | (550.0 | ) |
Proceeds from exercise of stock options | 5.0 |
| | — |
| | — |
| | — |
| | — |
| | 5.0 |
|
Taxes paid related to net share settlement of equity awards | (6.1 | ) | | — |
| | — |
| | — |
| | — |
| | (6.1 | ) |
Contingent consideration payments | — |
| | — |
| | — |
| | (3.8 | ) | | — |
| | (3.8 | ) |
Capital contribution from affiliates | — |
| | — |
| | — |
| | 7.2 |
| | (7.2 | ) | | — |
|
Capital payments to affiliates | — |
| | — |
| | — |
| | (52.4 | ) | | 52.4 |
| | — |
|
Payments on borrowings from affiliates | — |
| | (648.3 | ) | | — |
| | (242.1 | ) | | 890.4 |
| | — |
|
Proceeds from borrowings from affiliates | — |
| | 977.5 |
| | — |
| | 211.3 |
| | (1,188.8 | ) | | — |
|
Other items, net | — |
| | (6.1 | ) | | — |
| | 6.6 |
| | — |
| | 0.5 |
|
Net cash provided by financing activities | (554.8 | ) | | 323.1 |
| | — |
| | (90.8 | ) | | (253.2 | ) | | (575.7 | ) |
Effect on cash of changes in exchange rates | — |
| | — |
| | — |
| | 11.9 |
| | — |
| | 11.9 |
|
Net (decrease) increase in cash and cash equivalents | (0.3 | ) | | (6.0 | ) | | — |
| | (268.7 | ) | | — |
| | (275.0 | ) |
Cash and cash equivalents — beginning of period | 0.3 |
| | 12.3 |
| | — |
| | 986.2 |
| | — |
| | 998.8 |
|
Cash and cash equivalents — end of period | $ | — |
| | $ | 6.3 |
| | $ | — |
| | $ | 717.5 |
| | $ | — |
| | $ | 723.8 |
|
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
UNAUDITED CONDENSED CONSOLIDATING STATEMENT OF CASH FLOWS
Three Months Ended March 31, 2016
|
| | | | | | | | | | | | | | | | | | | | | | | |
(In millions) | Mylan N.V. | | Mylan Inc. | | Guarantor Subsidiaries | | Non-Guarantor Subsidiaries | | Eliminations | | Consolidated |
Cash flows from operating activities: | | | | | | | | | | | |
Net cash (used in) provided by operating activities | $ | (27.1 | ) | | $ | (139.4 | ) | | $ | — |
| | $ | 247.0 |
| | $ | — |
| | $ | 80.5 |
|
Cash flows from investing activities: | | | | | | | | | | | |
Capital expenditures | — |
| | (20.6 | ) | | — |
| | (31.2 | ) | | — |
| | (51.8 | ) |
Purchase of marketable securities | — |
| | (0.5 | ) | | — |
| | (8.0 | ) | | — |
| | (8.5 | ) |
Proceeds from sale of marketable securities | — |
| | — |
| | — |
| | 5.9 |
| | — |
| | 5.9 |
|
Investments in affiliates | — |
| | (11.3 | ) | | — |
| | — |
| | 11.3 |
| | — |
|
Loans to affiliates | (3.6 | ) | | (1,465.6 | ) | | — |
| | (1,699.6 | ) | | 3,168.8 |
| | — |
|
Repayments of loans from affiliates | 32.8 |
| | 12.2 |
| | — |
| | 7.2 |
| | (52.2 | ) | | — |
|
Payments for product rights and other, net | — |
| | (0.1 | ) | | — |
| | (105.5 | ) | | — |
| | (105.6 | ) |
Net cash provided by (used in) investing activities | 29.2 |
| | (1,485.9 | ) | | — |
| | (1,831.2 | ) | | 3,127.9 |
| | (160.0 | ) |
Cash flows from financing activities: | | | | | | | | | | | |
Payments of financing fees | (31.6 | ) | | — |
| | — |
| | — |
| | — |
| | (31.6 | ) |
Change in short-term borrowings, net | — |
| | — |
| | — |
| | 65.1 |
| | — |
| | 65.1 |
|
Proceeds from exercise of stock options | 3.6 |
| | — |
| | — |
| | — |
| | — |
| | 3.6 |
|
Taxes paid related to net share settlement of equity awards | (6.9 | ) | | — |
| | — |
| | — |
| | — |
| | (6.9 | ) |
Capital contribution from affiliates | — |
| | — |
| | — |
| | 11.3 |
| | (11.3 | ) | | — |
|
Payments on borrowings from affiliates | — |
| | (40.0 | ) | | — |
| | (12.2 | ) | | 52.2 |
| | — |
|
Proceeds from borrowings from affiliates | 31.6 |
| | 1,703.2 |
| | — |
| | 1,434.0 |
| | (3,168.8 | ) | | — |
|
Other items, net | 1.2 |
| | — |
| | — |
| | (0.9 | ) | | — |
| | 0.3 |
|
Net cash (used in) provided by financing activities | (2.1 | ) | | 1,663.2 |
| | — |
| | 1,497.3 |
| | (3,127.9 | ) | | 30.5 |
|
Effect on cash of changes in exchange rates | — |
| | — |
| | — |
| | 12.4 |
| | — |
| | 12.4 |
|
Net increase (decrease) in cash and cash equivalents | — |
| | 37.9 |
| | — |
| | (74.5 | ) | | — |
| | (36.6 | ) |
Cash and cash equivalents — beginning of period | — |
| | 870.5 |
| | — |
| | 365.5 |
| | — |
| | 1,236.0 |
|
Cash and cash equivalents — end of period | $ | — |
| | $ | 908.4 |
| | $ | — |
| | $ | 291.0 |
| | $ | — |
| | $ | 1,199.4 |
|
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
On December 5, 2016, the Company announced restructuring programs in certain locations representing initial steps in a series of actions that are anticipated to further streamline its operations globally. Since 2015, the Company has made a number of significant acquisitions, and as part of the holistic, global integration of these acquisitions, the Company is focused on how to best optimize and maximize all of its assets across the organization and across all geographies.
Charges for restructuring and ongoing cost reduction initiatives are recorded in the period the Company commits to a restructuring or cost reduction plan, or executes specific actions contemplated by the plan and all criteria for liability recognition have been met.
The Company continues to develop the details of the cost reduction initiatives, including workforce actions and other potential restructuring activities beyond the programs announced, including potential shutdown or consolidation of certain operations. The continued restructuring actions are expected to be implemented through fiscal year 2018. For the restructuring activities that have been initiated to date, the Company estimates that it will incur aggregate pre-tax charges ranging between $175.0 million and $225.0 million, inclusive of the 2016 and year to date 2017 restructuring charges of $172.8 million. As additional restructuring activities are undertaken, the Company expects to incur additional costs including employee related costs, such as severance and continuation of healthcare and other benefits; asset impairments; accelerated depreciation; costs associated with contract terminations; and other closure costs. At this time, the expenses related to the additional restructuring activities cannot be reasonably estimated.
The following table summarizes the restructuring charges and the reserve activity from December 31, 2016 to March 31, 2017:
|
| | | | | | | | | | | |
(In millions) | Employee Related Costs | | Other Exit Costs | | Total |
Balance at December 31, 2016: | $ | 138.6 |
| | $ | 1.6 |
| | $ | 140.2 |
|
Charges (1) | 9.6 |
| | 13.5 |
| | 23.1 |
|
Reclassifications | (8.3 | ) | | 8.3 |
| | — |
|
Cash payment | (54.2 | ) | | (1.0 | ) | | (55.2 | ) |
Utilization | — |
| | (19.8 | ) | | (19.8 | ) |
Foreign currency translation | (9.8 | ) | | — |
| | (9.8 | ) |
Balance at March 31, 2017: | $ | 75.9 |
| | $ | 2.6 |
| | $ | 78.5 |
|
| |
(1) | For the three months ended March 31, 2017, total restructuring charges in North America, Europe, Rest of World and Corporate / Other were approximately $6.4 million, $3.6 million, $12.8 million and $0.3 million, respectively. |
At March 31, 2017 and December 31, 2016, accrued liabilities for restructuring and other costs reduction programs were included in other current liabilities on the Condensed Consolidated Balance Sheets.
| |
18. | Collaboration and Licensing Agreements |
We periodically enter into collaboration and licensing agreements with other pharmaceutical companies for the development, manufacture, marketing and/or sale of pharmaceutical products. Our significant collaboration agreements are focused on the development, manufacturing, supply and commercialization of multiple, high-value generic biologic compounds, insulin analog products and respiratory products. Under these agreements, we have future potential milestone payments and co-development expenses payable to third parties as part of our licensing, development and co-development programs. Payments under these agreements generally become due and are payable upon the satisfaction or achievement of certain developmental, regulatory or commercial milestones or as development expenses are incurred on defined projects. Milestone payment obligations are uncertain, including the prediction of timing and the occurrence of events triggering a future obligation and are not reflected as liabilities in the Condensed Consolidated Balance Sheets, except for milestone and royalty obligations reflected as acquisition related contingent consideration. Refer to Note 11 Financial Instruments and Risk Management for contingent consideration amounts recorded. These agreements may also include potential sales-based milestones and call for us to pay a percentage of amounts earned from the sale of the product as a royalty or a profit share. The
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
amounts disclosed do not include sales based milestones or royalty obligations on future sales of product as the timing and amount of future sales levels and costs to produce products subject to these obligations is not reasonably estimable. These sales-based milestones or royalty obligations may be significant depending upon the level of commercial sales for each product. There have been no significant changes to our collaboration and licensing agreements as disclosed in our Annual Report on Form 10-K for the year ended December 31, 2016, as amended.
The Company is involved in various disputes, governmental and/or regulatory inquiries and proceedings, tax proceedings and litigation matters that arise from time to time, some of which are described below. The Company is also party to certain litigation matters including those for which Merck KGaA, Abbott Laboratories or Strides Arcolab has agreed to indemnify the Company, pursuant to the respective sale and purchase agreements.
While the Company believes that it has meritorious defenses with respect to the claims asserted against it and intends to vigorously defend its position, the process of resolving matters through litigation or other means is inherently uncertain, and it is not possible to predict the ultimate resolution of any such proceeding. It is possible that an unfavorable resolution of any of the matters described below, or the inability or denial of Merck KGaA, Abbott Laboratories, Strides Arcolab, or another indemnitor or insurer to pay an indemnified claim, could have a material effect on the Company’s business, financial condition, results of operations, cash flows and/or ordinary share price. Unless otherwise disclosed below, the Company is unable to predict the outcome of the respective litigation or to provide an estimate of the range of reasonably possible losses. Legal costs are recorded as incurred and are classified in SG&A in the Company’s Condensed Consolidated Statements of Operations.
Lorazepam and Clorazepate
On June 1, 2005, a jury verdict was rendered against Mylan, MPI, and co-defendants Cambrex Corporation and Gyma Laboratories in the U.S. District Court for the District of Columbia in the amount of approximately $12.0 million, which has been accrued for by the Company. The jury found that Mylan and its co-defendants willfully violated Massachusetts, Minnesota and Illinois state antitrust laws in connection with active pharmaceutical ingredient supply agreements entered into between the Company and its API supplier (Cambrex) and broker (Gyma) for two drugs, Lorazepam and Clorazepate, in 1997, and subsequent price increases on these drugs in 1998. The case was brought by four health insurers who opted out of earlier class action settlements agreed to by the Company in 2001 and represents the last remaining antitrust claims relating to Mylan’s 1998 price increases for Lorazepam and Clorazepate. Following the verdict, the Company filed a motion for judgment as a matter of law, a motion for a new trial, a motion to dismiss two of the insurers and a motion to reduce the verdict. On December 20, 2006, the Company’s motion for judgment as a matter of law and motion for a new trial were denied and the remaining motions were denied on January 24, 2008. In post-trial filings, the plaintiffs requested that the verdict be trebled and that request was granted on January 24, 2008. On February 6, 2008, a judgment was issued against Mylan and its co-defendants in the total amount of approximately $69.0 million, which, in the case of three of the plaintiffs, reflects trebling of the compensatory damages in the original verdict (approximately $11.0 million in total) and, in the case of the fourth plaintiff, reflects their amount of the compensatory damages in the original jury verdict plus doubling this compensatory damage award as punitive damages assessed against each of the defendants (approximately $58.0 million in total), some or all of which may be subject to indemnification obligations by Mylan. Plaintiffs are also seeking an award of attorneys’ fees and litigation costs in unspecified amounts and prejudgment interest of approximately $8.0 million. The Company and its co-defendants appealed to the U.S. Court of Appeals for the D.C. Circuit and have challenged the verdict as legally erroneous on multiple grounds. The appeals were held in abeyance pending a ruling on the motion for prejudgment interest, which has been granted. Mylan has contested this ruling along with the liability finding and other damages awards as part of its appeal, which was filed in the Court of Appeals for the D.C. Circuit. On January 18, 2011, the Court of Appeals issued a judgment remanding the case to the District Court for further proceedings based on lack of diversity with respect to certain plaintiffs. On June 13, 2011, Mylan filed a certiorari petition with the U.S. Supreme Court requesting review of the judgment of the D.C. Circuit. On October 3, 2011, the certiorari petition was denied. The case is now proceeding before the District Court. On January 14, 2013, following limited court-ordered jurisdictional discovery, the plaintiffs filed a fourth amended complaint containing additional factual averments with respect to the diversity of citizenship of the parties, along with a motion to voluntarily dismiss 775 (of 1,387) self-funded customers whose presence would destroy the District Court’s diversity jurisdiction. The plaintiffs also moved for a remittitur (reduction) of approximately $8.1 million from the full damages award. Mylan’s brief in response to the new factual averments in the complaint was filed on February 13, 2013. On July 29, 2014, the court granted both plaintiffs’ motion to amend the complaint and their motion to dismiss 775 self-funded customers.
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
In connection with the Company’s appeal of the judgment, the Company submitted a surety bond underwritten by a third-party insurance company in the amount of $74.5 million in February 2008. On May 30, 2012, the District Court ordered the amount of the surety bond reduced to $66.6 million.
Pricing and Medicaid Litigation
Dey L.P. (now known as Mylan Specialty L.P. and herein as “Mylan Specialty”), a wholly owned subsidiary of the Company, was named as a defendant in several class actions brought by consumers and third-party payors. Mylan Specialty reached a settlement of these class actions, which was approved by the court and all claims have been dismissed. Additionally, a complaint was filed under seal by a plaintiff on behalf of the United States of America against Mylan Specialty in August 1997. In August 2006, the Government filed its complaint-in-intervention and the case was unsealed in September 2006. The Government asserted that Mylan Specialty was jointly liable with a co-defendant and sought recovery of alleged overpayments, together with treble damages, civil penalties and equitable relief. Mylan Specialty completed a settlement of this action in December 2010. These cases all have generally alleged that Mylan Specialty falsely reported certain price information concerning certain drugs marketed by Mylan Specialty, that Mylan Specialty caused false claims to be made to Medicaid and to Medicare, and that Mylan Specialty caused Medicaid and Medicare to make overpayments on those claims.
Under the terms of the purchase agreement with Merck KGaA, Mylan is fully indemnified for the claims in the preceding paragraph and Merck KGaA is entitled to any income tax benefit the Company realizes for any deductions of amounts paid for such pricing litigation. Under the indemnity, Merck KGaA is responsible for all settlement and legal costs, and, as such, these settlements had no impact on the Company’s Consolidated Statements of Operations. At March 31, 2017, the Company has accrued approximately $63.3 million in other current liabilities, which represents its estimate of the remaining amount of anticipated income tax benefits due to Merck KGaA. We are not aware of any outstanding claims related to Merck KGaA.
Modafinil Antitrust Litigation and FTC Inquiry
Beginning in April 2006, Mylan and four other drug manufacturers have been named as defendants in civil lawsuits filed in or transferred to the U.S. District Court for the Eastern District of Pennsylvania by a variety of plaintiffs purportedly representing direct and indirect purchasers of the drug modafinil and in a lawsuit filed by Apotex, Inc., a manufacturer of generic drugs. These actions allege violations of federal antitrust and state laws in connection with the generic defendants’ settlement of patent litigation with Cephalon relating to modafinil. Discovery is closed. On June 23, 2014, the court granted the defendants’ motion for partial summary judgment dismissing plaintiffs’ claims that the defendants had engaged in an overall conspiracy to restrain trade (and denied the corresponding plaintiffs’ motion). On January 28, 2015, the District Court denied the defendants’ summary judgment motions based on factors identified in the Supreme Court’s Actavis decision. In an order on June 1, 2015, vacated and reissued on June 11, 2015, the District Court denied the indirect purchaser plaintiffs’ motion for class certification. The indirect purchaser plaintiffs filed a petition for leave to appeal the certification decision, which was denied by the Court of Appeals for the Third Circuit on December 21, 2015. On July 27, 2015, the District Court granted the direct purchaser plaintiffs’ motion for class certification. On October 9, 2015, the Third Circuit granted defendants’ petition for leave to appeal the class certification decision. On October 16, 2015, defendants filed a motion to stay the liability trial, which had been set to begin on February 2, 2016, with the District Court pending the appeal of the decision to certify the direct purchaser class; this motion was denied on December 17, 2015. On December 17, 2015, the District Court approved the form and manner of notice to the certified class of direct purchasers; the notice was subsequently issued to the class. On December 21, 2015, the defendants filed a motion to stay with the Court of Appeals for the Third Circuit, which was granted on January 25, 2016; accordingly, the trial was stayed and the case was placed in suspense. On September 13, 2016, the Third Circuit reversed the district court’s certification order and remanded for further proceedings. On October 14, 2016 direct purchaser plaintiffs filed a petition seeking rehearing. On October 31, 2016 the petition seeking rehearing was denied. On December 12, 2016, the District Court removed the case from suspense and set the trial for June 5, 2017. On March 24, 2015, Mylan reached a settlement in principle with the putative indirect purchasers, and on November 20, 2015, Mylan entered into a settlement agreement with the putative indirect purchasers for approximately $16 million. Plaintiffs have not yet moved for preliminary approval of that settlement. In December 2016, Mylan reached a settlement with the putative direct purchaser class and the retailer opt-out plaintiffs for $165 million, of which approximately $68.5 million was paid before December 31, 2016. The settlement with the retailer opt-out plaintiffs has been completed. The settlement with the putative direct purchaser class will undergo the court approval process. On February 3, 2017, the putative direct purchaser class moved for preliminary approval. The Company has accrued approximately $112.5 million related to this matter at March 31, 2017 and December 31, 2016.
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
On June 29, 2015, the City of Providence, Rhode Island filed suit in the District of Rhode Island against the same parties named as defendants in litigation pending in the Eastern District of Pennsylvania, including Mylan, asserting state law claims based on the same underlying allegations. All defendants, including Mylan, moved to dismiss the suit on October 15, 2015, and the case was subsequently settled.
On July 10, 2015, the Louisiana Attorney General filed in the 19th Judicial District Court in Louisiana a petition against Mylan and three other drug manufacturers asserting state law claims based on the same underlying allegations as those made in litigation pending in the Eastern District of Pennsylvania. The petition was filed by the State of Louisiana purportedly in its capacity as an indirect purchaser. On May 16, 2016, the Judicial District Court deferred Mylan’s declinatory exception of no personal jurisdiction and its peremptory exception of prescription, and granted in part and denied in part Mylan’s peremptory exceptions of no cause of action and no right of action. On June 30, 2016, the plaintiff filed a supplemental and amended petition. The defendants filed a motion to strike and joint peremptory exceptions to the amended petition. On July 21, 2016, the plaintiff filed in the First Circuit Court of Appeal its application for a supervisory writ regarding the granting of defendant’s exceptions, which the defendants opposed. The appeal was denied on October 31, 2016. On April 20, 2016, the State of Louisiana filed a motion to consolidate the pending action with four other actions against other pharmaceutical manufacturers concerning products not related to modafinil, which Mylan opposed. On June 27, 2016, the Judicial District Court declined to consolidate Mylan’s case with the other four actions, with leave to renew the consolidation request after filing the above-referenced amended petition. On July 21, 2016, the plaintiff filed a motion to reurge consolidation. Subsequently, the action to which plaintiff seeks to join Mylan was stayed, resulting in a stay of the consolidation motion. On December 8, 2016, Mylan’s peremptory exceptions of no cause of action with respect to the supplemental and amended petition were granted in their entirety and with prejudice and judgment was entered. On February 17, 2017, the plaintiff filed in the 19th Judicial District Court a motion for appeal, which the Judicial District Court granted on February 21, 2017. The appeal was lodged with the First Circuit Court of Appeal on April 4, 2017.
On July 28, 2016, United Healthcare filed a complaint against Mylan and four other drug manufacturers in the United States District Court for the District of Minnesota, asserting state law claims based on the same underlying allegations as those made in litigation pending in the Eastern District of Pennsylvania. On January 6, 2017, the case was transferred to the Eastern District of Pennsylvania. Mylan filed its answer to the complaint on March 31, 2017.
The Company believes that it has strong defenses to these remaining cases. Although it is reasonably possible that the Company may incur additional losses from these matters, any amount cannot be reasonably estimated at this time.
In addition, by letter dated July 11, 2006, Mylan was notified by the U.S. Federal Trade Commission (“FTC”) of an investigation relating to the settlement of the modafinil patent litigation. In its letter, the FTC requested certain information from Mylan, MPI and Mylan Technologies, Inc. pertaining to the patent litigation and the settlement thereof. On March 29, 2007, the FTC issued a subpoena, and on April 26, 2007, the FTC issued a civil investigative demand to Mylan, requesting additional information from the Company relating to the investigation. Mylan has cooperated fully with the government’s investigation and completed all requests for information. On February 13, 2008, the FTC filed a lawsuit against Cephalon in the U.S. District Court for the District of Columbia and the case was subsequently transferred to the U.S. District Court for the Eastern District of Pennsylvania. On July 1, 2010, the FTC issued a third party subpoena to Mylan, requesting documents in connection with its lawsuit against Cephalon. Mylan has responded to the subpoena. The lawsuit against Cephalon settled and a Stipulated Order for Permanent Injunction and Equitable Monetary Relief was entered by the Court on June 17, 2015.
Pioglitazone
Beginning in December 2013, Mylan, Takeda, and several other drug manufacturers have been named as defendants in civil lawsuits consolidated in the U.S. District Court for the Southern District of New York by plaintiffs which purport to represent indirect purchasers of branded or generic Actos® and Actoplus Met®. These actions allege violations of state and federal competition laws in connection with the defendants’ settlements of patent litigation in 2010 relating to Actos and Actoplus Met®. Plaintiffs filed an amended complaint on August 22, 2014. Mylan and the other defendants filed motions to dismiss the amended complaint on October 10, 2014. Two additional complaints were subsequently filed by plaintiffs purporting to represent classes of direct purchasers of branded or generic Actos® and Actoplus Met®. On September 23, 2015, the District Court granted defendants’ motions to dismiss the indirect purchasers amended complaints with prejudice. The indirect purchasers filed a notice of appeal on October 22, 2015; however they have since abandoned and dismissed their appeal of the District Court’s dismissal of claims asserted against Mylan. The putative direct purchaser class filed an amended complaint on January 8, 2016. Defendants’ motion to dismiss was filed on January 28, 2016 and the briefing has been completed. The case was stayed pending the resolution of the indirect purchasers’ appeal against the defendants remaining in
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
that case. A decision was issued by the Second Circuit on February 8, 2017, reversing in part and affirming in part, the District Court’s decision as to the remaining defendants. Following this decision, the direct purchasers filed an amended complaint and the Court has set a schedule for briefing on Supplemental Motions to Dismiss.
SEC Investigation
On September 10, 2015, Mylan N.V. received a subpoena from the SEC seeking documents with regard to certain related party matters. Mylan is fully cooperating with the SEC in its investigation, and we are unable to predict the outcome of this matter at this time.
EpiPen® Auto-Injector and Certain Congressional Matters
Classification of EpiPen® Auto-Injector and EpiPen Jr® Auto-Injector
In November 2014, the Company received a subpoena from the U.S. Department of Justice (“DOJ”) related to the classification of the EpiPen® Auto-Injector for purposes of the Medicaid Drug Rebate Program. The Company complied with various information requests received from the DOJ pursuant to the subpoena. The question in the underlying matter was whether EpiPen® Auto-Injector was properly classified with the Centers for Medicare and Medicaid Services (“CMS”) as a non-innovator drug under the applicable definition in the Medicaid Rebate statute and subject to the formula that is used to calculate rebates to Medicaid for such drugs. EpiPen® Auto-Injector has been classified with CMS as a non-innovator drug since before Mylan acquired the product in 2007 based on longstanding written guidance from the federal government. Beginning in August 2016, questions regarding the pricing of the EpiPen® Auto-Injector significantly increased and the Company has received or has been the subject of additional inquiries, including with respect to the classification of EpiPen® Auto-Injector for purposes of the Medicaid Drug Rebate Program and certain other federal programs, from committees and members of Congress and from other federal and state governmental agencies.
Subsequent to these developments, on October 7, 2016, Mylan agreed to the terms of a $465 million settlement with the DOJ and other government agencies related to the classification of the EpiPen® Auto-Injector for purposes of the Medicaid Drug Rebate Program. The terms of the settlement do not provide for any finding of wrongdoing on the part of Mylan Inc. or any of its affiliated entities or personnel. The settlement terms provide for resolution of all potential Medicaid rebate liability claims by federal and state governments as to whether the product should have been classified as an innovator drug for Medicaid Drug Rebate Program purposes, and subject to a higher rebate formula. In connection with the settlement, Mylan expects to enter into a corporate integrity agreement with the Office of Inspector General of the Department of Health and Human Services. Mylan continues to work with the government to finalize the settlement. When the settlement is finalized, Mylan expects to classify the EpiPen® Auto-Injector as an innovator drug effective April 1, 2017. During the year ended December 31, 2016, the Company recorded an accrual of $465 million related to the DOJ settlement which is included in other current liabilities in the Condensed Consolidated Balance Sheets.
SEC Document Request/Subpoena
On October 7, 2016, Mylan received a document request from the Division of Enforcement at the SEC seeking communications with CMS and documents concerning Mylan products sold and related to the Medicaid Drug Rebate Program, and any related complaints. On November 15, 2016, Mylan received a follow-up letter, modifying the initial document request, seeking information on and public disclosures regarding the settlement with the DOJ announced on October 7, 2016 and the classification of the EpiPen® Auto-Injector under the Medicaid Drug Rebate Program. On February 6, 2017, Mylan received a subpoena from the SEC in this matter, seeking additional documents. Mylan is fully cooperating with the SEC’s inquiry.
FTC Request for Information
On November 18, 2016, Mylan received a request from the FTC Bureau of Competition seeking documents and information relating to its preliminary investigation into potential anticompetitive practices in the market for epinephrine auto-injectors. Mylan is fully cooperating with the FTC’s inquiry.
Federal Securities Litigation
Purported class action complaints were filed in October 2016 against Mylan N.V., Mylan Inc. and certain of their current and former directors and officers (collectively, for purposes of this paragraph, the “defendants”) in the United States
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
District Court for the Southern District of New York on behalf of certain purchasers of securities of Mylan N.V. and/or Mylan Inc. on the NASDAQ. The complaints alleged that defendants made false or misleading statements and omissions of purportedly material fact, in violation of federal securities laws, in connection with disclosures relating to Mylan N.V. and Mylan Inc.’s classification of their EpiPen® Auto-Injector as a non-innovator drug for purposes of the Medicaid Drug Rebate Program. The complaints sought damages, as well as the plaintiffs’ fees and costs. On March 20, 2017, after the actions were consolidated, a consolidated amended complaint was filed, alleging substantially similar claims and seeking substantially similar relief, but adding allegations that defendants made false or misleading statements and omissions of purportedly material fact in connection with allegedly anticompetitive conduct with respect to EpiPen® Auto-Injector and certain generic drugs, and alleging violations of both federal securities laws (on behalf of a purported class of certain purchasers of securities of Mylan N.V. and/or Mylan Inc. on the NASDAQ) and Israeli securities laws (on behalf of a purported class of certain purchasers of securities of Mylan N.V. on the Tel Aviv Stock Exchange). Defendants’ response to the consolidated amended complaint is due May 30, 2017. We believe that the claims in the consolidated amended complaint are without merit and intend to defend against them vigorously.
Israeli Securities Litigation
On October 13, 2016, a purported shareholder of Mylan N.V. filed a lawsuit, together with a motion to certify the lawsuit as a class action on behalf of certain Mylan N.V. shareholders on the Tel Aviv Stock Exchange, against Mylan N.V. and four of its directors and officers (collectively, for purposes of this paragraph, the “defendants”) in the Tel Aviv District Court (Economic Division). The plaintiff alleges that the defendants made false or misleading statements and omissions of purportedly material fact in Mylan N.V.’s reports to the Tel Aviv Stock Exchange regarding Mylan N.V.’s classification of its EpiPen® Auto-Injector for purposes of the Medicaid Drug Rebate Program, in violation of both U.S. and Israeli securities laws, the Israeli Companies Law and the Israeli Torts Ordinance. The plaintiff seeks damages, among other remedies. On January 19, 2017, the Court stayed this case until a final judgment is issued in the securities litigation currently pending in the United States District Court for the Southern District of New York. On April 30, 2017, another purported shareholder of Mylan N.V. filed a separate lawsuit, together with a motion to certify the lawsuit as a class action on behalf of certain Mylan N.V. shareholders on the Tel Aviv Stock Exchange, in the Tel Aviv District Court (Economic Division), alleging substantially similar claims and seeking substantially similar relief against the defendants and other directors and officers of Mylan N.V., but alleging also that this group of defendants made false or misleading statements and omissions of purportedly material fact in connection with allegedly anticompetitive conduct with respect to EpiPen® Auto-Injector and certain generic drugs, and alleging violations of both U.S. federal securities laws and Israeli law. Service of process has not been effected in the April 30, 2017 lawsuit. We believe that the claims in these lawsuits are without merit and intend to defend against them vigorously.
EpiPen® Auto-Injector Civil Litigation
Beginning in August 2016, Mylan Specialty L.P. and other Mylan-affiliated entities have been named as defendants in thirteen putative class actions relating to the pricing and/or marketing of the EpiPen® Auto-Injector. A Mylan officer and other non-Mylan affiliated companies also have been named as defendants in some of the class actions. These lawsuits were filed in the U.S. District Courts for the Northern District of California, Northern District of Illinois, District of Kansas, Eastern District of Michigan, Western District of Washington, District of New Jersey and the Western District of Pennsylvania, as well as the Hamilton County, Ohio Court of Common Pleas (later removed to the Southern District of Ohio). All but four of these lawsuits (one in Illinois, one in Washington and two in New Jersey) have either been dismissed or consolidated into a single putative class action pending in the U.S. District Court for the District of Kansas. The plaintiffs in these cases assert violations of various federal and state antitrust and consumer protection laws, the Racketeer Influenced and Corrupt Organizations Act (“RICO”), as well as common law claims. Plaintiffs’ claims include purported challenges to the prices charged for the EpiPen® Auto-Injector and/or the marketing of the product in packages containing two auto-injectors, as well as allegedly anti-competitive conduct. Plaintiff in one of the putative class action lawsuits in New Jersey has filed a request with the Judicial Panel on Multidistrict Litigation to transfer all of the cases into a multidistrict litigation (“MDL”) proceeding in the District of New Jersey. We believe that the claims in these lawsuits are without merit and intend to defend against them vigorously.
On April 24, 2017, Sanofi-Aventis U.S., LLC (“Sanofi”) filed a lawsuit against Mylan Inc. and Mylan Specialty L.P. in the U.S. District Court for the District of New Jersey. In this lawsuit, Sanofi alleges exclusive dealings and anti-competitive marketing practices in violation of the antitrust laws in connection with the sale and marketing of the EpiPen® Auto-Injector. We believe that the claims in this lawsuit are without merit and intend to defend against them vigorously.
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
EpiPen® Auto-Injector State AG Investigations
Beginning in August 2016, the Company and certain of its affiliated entities received subpoenas and informal requests from various state attorneys general seeking information and documents relating to the pricing and/or marketing of the EpiPen® Auto-Injector. The Company is fully cooperating with these inquiries.
U.S. Congress/State Requests for Information and Documents
Beginning in August 2016, Mylan has received several requests for information and documents from various Committees of the U.S. Congress and federal and state lawmakers concerning the marketing, distribution and sales of Mylan products. Mylan has cooperated and intends to continue cooperating with federal and state lawmakers as appropriate in response to their requests.
The Company believes that it has strong defenses to current and future potential civil litigation, as well as governmental investigations and enforcement proceedings, discussed in this “EpiPen® Auto-Injector and Certain Congressional Matters” section of this Note 19 Litigation. Although it is reasonably possible that the Company may incur additional losses from these matters, any amount cannot be reasonably estimated at this time. In addition, the Company expects to incur additional legal and other professional service expenses associated with such matters in future periods and will recognize these expenses as services are received. The Company believes that the ultimate amount paid for these services and claims could have a material effect on the Company's business, consolidated financial condition, results of operations, cash flows and/or ordinary share price in future periods.
Drug Pricing Matters
Department of Justice/Connecticut Subpoenas
On December 3, 2015, a subsidiary of Mylan N.V. received a subpoena from the Antitrust Division of the U.S. DOJ seeking information relating to the marketing, pricing, and sale of our generic Doxycycline products and any communications with competitors about such products.
On September 8, 2016, a subsidiary of Mylan N.V., as well as certain employees and a member of senior management, received subpoenas from the DOJ seeking additional information relating to the marketing, pricing and sale of our generic Cidofovir, Glipizide-metformin, Propranolol and Verapamil products and any communications with competitors about such products. Related search warrants also were executed. The Company is fully cooperating with the DOJ’s inquiry.
On December 21, 2015, the Company received a subpoena and interrogatories from the Connecticut Office of the Attorney General seeking information relating to the marketing, pricing and sale of certain of the Company’s generic products (including Doxycycline) and communications with competitors about such products. The Company is fully cooperating with Connecticut’s inquiry.
Civil Litigation
Twenty-two putative class action complaints are pending against Mylan Inc., Mylan Pharmaceuticals Inc. and other pharmaceutical manufacturers in a MDL in the United States District Court for the Eastern District of Pennsylvania; plaintiff indirect purchasers, direct purchasers and independent pharmacies generally allege anticompetitive conduct with respect to certain Doxycycline and Digoxin products. Mylan and its subsidiary believe that the claims in these lawsuits are without merit and intend to deny liability and to defend against them vigorously.
Twelve putative class action complaints have been filed against Mylan Inc., Mylan Pharmaceuticals Inc. and other pharmaceutical manufacturers in the United States District Court for the Eastern District of Pennsylvania; plaintiff indirect and direct purchasers generally allege anticompetitive conduct with respect to certain Pravastatin products. These cases have been transferred to the MDL. Mylan and its subsidiary believe that the claims in these lawsuits are without merit and intend to deny liability and to defend against them vigorously.
Eight putative class action complaints have been filed against Mylan Inc., Mylan Pharmaceuticals Inc. and another pharmaceutical manufacturer in the United States District Court for the Eastern District of Pennsylvania; plaintiff indirect and direct purchasers generally allege anticompetitive conduct with respect to certain Divalproex products. These cases have been
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
transferred to the MDL. Mylan and its subsidiary believe that the claims in these lawsuits are without merit and intend to deny liability and to defend against them vigorously.
Ten putative class action complaints have been filed against Mylan Pharmaceuticals Inc. and other pharmaceutical manufacturers in the United States District Courts for the Southern District of New York and the Eastern District of Pennsylvania; plaintiff indirect and direct purchasers generally allege anticompetitive conduct with respect to certain Levothyroxine products. These cases have been transferred to the MDL. Mylan Pharmaceuticals Inc. believes that the claims in these lawsuits are without merit and intends to deny liability and to defend against them vigorously.
Ten putative class action complaints have been filed against Mylan Inc., Mylan Pharmaceuticals Inc., UDL Laboratories, Inc. and other pharmaceutical manufacturers in the United States District Courts for the Southern District of New York and the Eastern District of Pennsylvania; plaintiff indirect and direct purchasers generally allege anticompetitive conduct with respect to certain Propranolol products. The Defendants’ Motions to Dismiss the South District of New York cases was granted as to some state law claims but otherwise denied on April 6, 2017. These cases have been transferred to the MDL. Mylan and its subsidiaries believe that the claims in these lawsuits are without merit and intend to deny liability and to defend against them vigorously.
Eight putative class action complaints have been filed against Mylan Inc., Mylan Pharmaceuticals Inc., Mylan N.V. and another pharmaceutical manufacturer in the United States District Court for the District of Puerto Rico, the District of New Jersey and the Eastern District of Pennsylvania; plaintiff indirect and direct purchasers generally allege anticompetitive conduct with respect to certain Clomipramine products. These cases have been transferred to the MDL. Mylan and its subsidiaries believe that the claims in these lawsuits are without merit and intend to deny liability and to defend against them vigorously.
Four putative class action complaints have been filed against Mylan Inc., Mylan Pharmaceuticals Inc. and another pharmaceutical manufacturer in the U.S. District Court for the Eastern District of Pennsylvania; plaintiff indirect and direct purchasers generally allege anticompetitive conduct with respect to certain Albuterol products. These cases have been transferred to the MDL. Mylan and its subsidiary believe that the claims in these lawsuits are without merit and intend to deny liability and to defend against them vigorously.
One putative class action complaint has been filed against Mylan Inc., Mylan Pharmaceuticals Inc. and another pharmaceutical manufacturer in the U.S. District Court for the Eastern District of Pennsylvania; plaintiff indirect purchaser generally alleges anticompetitive conduct with respect to certain Benazepril HCTZ products. This case has been transferred to the MDL. Mylan and its subsidiary believe that the claims in this lawsuit are without merit and intend to deny liability and to defend against them vigorously.
Four putative class action complaints have been filed against Mylan Inc., Mylan Pharmaceuticals Inc. and other pharmaceutical manufacturers in the U.S. District Court for the Eastern District of Pennsylvania and the U.S. District Court for the Southern District of New York; plaintiff indirect and direct purchasers generally allege anticompetitive conduct with respect to certain Amitriptyline products. These cases have been transferred to the MDL. Mylan and its subsidiary believe that the claims in these lawsuits are without merit and intend to deny liability and to defend against them vigorously.
A complaint was filed on January 31, 2017 by putative classes of direct and indirect purchasers against Mylan Pharmaceuticals Inc. and other pharmaceutical manufacturers in the United States District Court for the District of Connecticut. Plaintiffs generally allege anticompetitive conduct and RICO violations with respect to, among other things, certain Doxycycline products. This case has been transferred to the MDL. Mylan Pharmaceuticals Inc. believes that the claims in this lawsuit are without merit and intends to deny liability and to defend against them vigorously.
Attorney General Litigation
On December 14, 2016, attorneys general of twenty states filed a complaint in the United States District Court for the District of Connecticut against several generic pharmaceutical drug manufacturers, including Mylan, alleging anticompetitive conduct with respect to, among other things, Doxycycline Hyclate Delayed Release. On March 1, 2017, the complaint was amended to add the attorneys general of twenty additional states; the complaint alleges violation of federal and state antitrust laws, as well as violation of various states’ consumer protection laws. Certain of the Defendants have filed a request to transfer the case to the MDL. A decision on the request to transfer the case to the MDL remains pending. We believe that the claims in this lawsuit against Mylan are without merit and intend to defend against them vigorously.
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
European Commission Proceedings
Perindopril
On or around July 8, 2009, the European Commission (the “Commission”) stated that it had initiated antitrust proceedings pursuant to Article 11(6) of Regulation No. 1/2003 and Article 2(1) of Regulation No. 773/2004 to explore possible infringement of Articles 81 and 82 EC and Articles 53 and 54 of the European Economic Area Agreement by Les Laboratoires Servier (“Servier”) as well as possible infringement of Article 81 EC by the Company’s Indian subsidiary, Mylan Laboratories Limited, and four other companies, each of which entered into agreements with Servier relating to the product Perindopril. On July 30, 2012, the Commission issued a Statement of Objections to Servier SAS, Servier Laboratories Limited, Les Laboratories Servier, Adir, Biogaran, Krka, d.d. Novo mesto, Lupin Limited, Mylan Laboratories Limited, Mylan, Niche Generics Limited, Teva UK Limited, Teva Pharmaceutical Industries Ltd., Teva Pharmaceuticals Europe B.V. and Unichem Laboratories Limited. Mylan Inc. and Mylan Laboratories Limited filed responses to the Statement of Objections. On July 9, 2014, the Commission issued a decision finding that Mylan Laboratories Limited and Mylan, as well as the companies noted above (with the exception of Adir, a subsidiary of Servier), had violated European Union competition rules and fined Mylan Laboratories Limited approximately €17.2 million, including approximately €8.0 million jointly and severally with Mylan Inc. The Company paid approximately $21.7 million related to this matter during the fourth quarter of 2014. In September 2014, the Company filed an appeal of the Commission’s decision to the General Court of the European Union. The briefing on appeal is complete. A hearing on the appeal before the General Court of the European Union has been scheduled for June 27, 2017.
Citalopram
On March 19, 2010, Mylan and Generics [U.K.] Limited, a wholly owned subsidiary of the Company, received notice that the Commission had opened proceedings against Lundbeck with respect to alleged unilateral practices and/or agreements related to Citalopram in the European Economic Area. On July 25, 2012 a Statement of Objections was issued to Lundbeck, Merck KGaA, Generics [U.K.] Limited, Arrow, Resolution Chemicals, Xelia Pharmaceuticals, Alpharma, A.L. Industrier and Ranbaxy. Generics [U.K.] Limited filed a response to the Statement of Objections and vigorously defended itself against allegations contained therein. On June 19, 2013, the Commission issued a decision finding that Generics [U.K.] Limited, as well as the companies noted above, had violated European Union competition rules and fined Generics [U.K.] Limited approximately €7.8 million, jointly and severally with Merck KGaA. Generics [U.K.] Limited has appealed the Commission’s decision to the General Court of the EU. Briefing on the appeal has been completed and a hearing took place on October 8, 2015. On September 8, 2016, the General Court dismissed all appeals against the European Commission’s decision. Mylan filed an appeal of the decision on November 18, 2016 to the European Court of Justice. The Company has accrued approximately $8.3 million and $8.2 million as of March 31, 2017 and December 31, 2016, respectively, related to this matter. It is reasonably possible that we will incur additional losses above the amount accrued but we cannot estimate a range of such reasonably possible losses at this time. There are no assurances, however, that settlements reached and/or adverse judgments received, if any, will not exceed amounts accrued. Generics [U.K.] Limited has also sought indemnification from Merck KGaA with respect to the €7.8 million portion of the fine for which Merck KGaA and Generics [U.K.] Limited were held jointly and severally liable. Merck KGaA has counterclaimed against Generics [U.K.] Limited seeking the same indemnification.
U.K. Competition and Markets Authority
Paroxetine
On August 12, 2011, Generics [U.K.] Limited received notice that the Office of Fair Trading (subsequently changed to the Competition and Markets Authority (the “CMA”)) was opening an investigation to explore the possible infringement of the Competition Act 1998 and Articles 101 and 102 of the Treaty on the Functioning of the European Union, with respect to alleged agreements related to Paroxetine. On April 19, 2013, a Statement of Objections was issued to Beecham Group plc, GlaxoSmithKline UK Limited, GlaxoSmithKline plc and SmithKline Beecham Limited (formerly, SmithKline Beecham plc) (together, “GlaxoSmithKline”), Generics [U.K.] Limited, Merck KGaA, Actavis UK Limited (formerly, Alpharma Limited), Xellia Pharmaceuticals ApS (formerly, Alpharma ApS) and Alpharma LLC (formerly, Zoetis Products LLC, Alpharma LLC, and Alpharma Inc.) (together, “Alpharma”), and Ivax LLC (formerly, Ivax Corporation) and Norton Healthcare Limited (which previously traded as Ivax Pharmaceuticals UK) (together, “Ivax”). Generics [U.K.] Limited filed a response to the Statement of Objections, defending itself against the allegations contained therein. The CMA issued a Supplementary Statement of Objections (“SSO”) to the above-referenced parties on October 21, 2014 and a hearing with regard to the SSO took place on December 19, 2014. The CMA issued a decision on February 12, 2016, finding that GlaxoSmithKline, Generics [U.K.] Limited, Merck KGaA and Alpharma, were liable for infringing EU and U.K. competition rules. With respect to Merck KGaA and
MYLAN N.V. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial Statements (Unaudited) - Continued
Generics [U.K.] Limited, the CMA issued a penalty of approximately £5.8 million, for which Merck KGaA is liable for the entire amount; and of that amount Generics [U.K.] Limited is jointly and severally liable for approximately £2.7 million, which was accrued for at March 31, 2017. Generics [U.K.] Limited has appealed the decision. The hearing before the Competition Appeals Tribunal concluded on March 30, 2017 and the parties are presently awaiting a decision.
Product Liability
The Company is involved in a number of product liability lawsuits and claims related to alleged personal injuries arising out of certain products manufactured and/or distributed by the Company, including but not limited to Phenytoin, Alendronate Sodium and Reglan. The Company believes that it has meritorious defenses to these lawsuits and claims and is vigorously defending itself with respect to those matters. From time to time, the Company has agreed to settle or otherwise resolve certain lawsuits and claims on terms and conditions that are in the best interests of the Company. The Company has accrued approximately $31.4 million and $31.5 million at March 31, 2017 and December 31, 2016, respectively. It is reasonably possible that we will incur additional losses and fees above the amount accrued but we cannot estimate a range of such reasonably possible losses or legal fees related to these claims at this time. There are no assurances, however, that settlements reached and/or adverse judgments received, if any, will not exceed amounts accrued.
Intellectual Property
In certain situations, the Company has used its business judgment to decide to market and sell products, notwithstanding the fact that allegations of patent infringement(s) or other potential third party rights have not been finally resolved by the courts. The risk involved in doing so can be substantial because the remedies available to the owner of a patent for infringement may include, a reasonable royalty on sales or damages measured by the profits lost by the patent owner. If there is a finding of willful infringement, damages may be increased up to three times. Moreover, because of the discount pricing typically involved with bioequivalent products, patented branded products generally realize a substantially higher profit margin than bioequivalent products. An adverse decision could have an adverse effect that is material to our business, financial condition, results of operations, cash flows and/or ordinary share price.
Other Litigation
The Company is involved in various other legal proceedings that are considered normal to its business, including but not limited to certain proceedings assumed as a result of the acquisition of the former Merck Generics business, Meda, Agila and the acquired EPD Business. The Company has approximately $20 million accrued related to these various other legal proceedings at March 31, 2017. While it is not possible to predict the ultimate outcome of such other proceedings, the ultimate outcome of any such proceeding is not currently expected to be material to the Company’s business, financial condition, results of operations, cash flows and/or ordinary share price.
ITEM 2. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion and analysis addresses material changes in the financial condition and results of operations of Mylan N.V. and subsidiaries for the periods presented. Unless context requires otherwise, the “Company”, “Mylan”, “our”, or “we” refer to Mylan N.V. and its subsidiaries. This discussion and analysis should be read in conjunction with the Consolidated Financial Statements, the related Notes to Consolidated Financial Statements and Management’s Discussion and Analysis of Financial Condition and Results of Operations included in Mylan N.V.’s Annual Report on Form 10-K for the year ended December 31, 2016, as amended, the unaudited interim financial statements and related Notes included in Part I — ITEM 1 of this Quarterly Report on Form 10-Q (“Form 10-Q”) and our other Securities and Exchange Commission (the “SEC”) filings and public disclosures. The interim results of operations, comprehensive earnings and cash flows for the three months ended March 31, 2017 are not necessarily indicative of the results to be expected for the full fiscal year or any other future period.
This Form 10-Q contains “forward-looking statements.” These statements are made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Such forward-looking statements may include, without limitation, statements about the acquisition of Meda AB (publ.) (“Meda”) by Mylan (the “Meda Transaction”), Mylan’s acquisition (the “EPD Transaction”) of Mylan Inc. and Abbott Laboratories’ non-U.S. developed markets specialty and branded generics business (the “EPD Business”), the potential benefits and synergies of the EPD Transaction and the Meda Transaction, future opportunities for Mylan and products, and any other statements regarding Mylan’s future operations, anticipated business levels, future earnings, planned activities, anticipated growth, market opportunities, strategies, competition, and other expectations and targets for future periods. These may often be identified by the use of words such as “will,” “may,” “could,” “should,” “would,” “project,” “believe,” “anticipate,” “expect,” “plan,” “estimate,” “forecast,” “potential,” “intend,” “continue,” “target” and variations of these words or comparable words. Because forward-looking statements inherently involve risks and uncertainties, actual future results may differ materially from those expressed or implied by such forward-looking statements. Factors that could cause or contribute to such differences include, but are not limited to: the ability to meet expectations regarding the accounting and tax treatments of the EPD Transaction and the Meda Transaction; changes in relevant tax and other laws, including but not limited to changes in the U.S. tax code and healthcare and pharmaceutical laws and regulations in the U.S. and abroad; actions and decisions of healthcare and pharmaceutical regulators; the integration of the EPD Business and Meda being more difficult, time-consuming, or costly than expected; operating costs, customer loss, and business disruption (including, without limitation, difficulties in maintaining relationships with employees, customers, clients, or suppliers) being greater than expected following the EPD Transaction and the Meda Transaction; the retention of certain key employees of the EPD Business and Meda being difficult; the possibility that Mylan may be unable to achieve expected synergies and operating efficiencies in connection with the EPD Transaction the Meda Transaction, and the December 2016 announced restructuring program in certain locations, within the expected time-frames or at all and to successfully integrate the EPD Business and Meda; with respect to the Medicaid Drug Rebate Program Settlement (as defined below), the inability or unwillingness on the part of any of the parties to finalize the settlement, any legal or regulatory challenges to the settlement, and any failure by third parties to comply with their contractual obligations; expected or targeted future financial and operating performance and results; the capacity to bring new products to market, including but not limited to where Mylan uses its business judgment and decides to manufacture, market, and/or sell products, directly or through third parties, notwithstanding the fact that allegations of patent infringement(s) have not been finally resolved by the courts (i.e., an “at-risk launch”); any regulatory, legal, or other impediments to Mylan’s ability to bring new products, including but not limited to generic Advair, to market; success of clinical trials and Mylan’s ability to execute on new product opportunities, including but not limited to generic Advair; any changes in or difficulties with our inventory of, and our ability to manufacture and distribute, the EpiPen® Auto-Injector and EpiPen Jr® Auto-Injector (collectively, “EpiPen® Auto-Injector”) to meet anticipated demand; the potential impact of any change in patient access to the EpiPen® Auto-Injector and the introduction of a generic version of the EpiPen® Auto-Injector; the scope, timing, and outcome of any ongoing legal proceedings, including government investigations, and the impact of any such proceedings on financial condition, results of operations, and/or cash flows; the ability to protect intellectual property and preserve intellectual property rights; the effect of any changes in customer and supplier relationships and customer purchasing patterns; the ability to attract and retain key personnel; changes in third-party relationships; the impact of competition; changes in the economic and financial conditions of the businesses of Mylan; the inherent challenges, risks, and costs in identifying, acquiring, and integrating complementary or strategic acquisitions of other companies, products, or assets and in achieving anticipated synergies; uncertainties and matters beyond the control of management; and inherent uncertainties involved in the estimates and judgments used in the preparation of financial statements, and the providing of estimates of financial measures, in accordance with accounting principles generally accepted in the United States of America (“U.S. GAAP”) and related standards or on an adjusted basis. For more detailed information on the risks and uncertainties associated with Mylan’s business activities, see the risks described in Mylan’s Annual Report on Form 10-K for the year ended
December 31, 2016, as amended and our other filings with the SEC. You can access Mylan’s filings with the SEC through the SEC website at www.sec.gov, and Mylan strongly encourages you to do so. Mylan undertakes no obligation to update any statements herein for revisions or changes after the filing date of this Form 10-Q.
Executive Overview
Mylan is a leading global pharmaceutical company, which develops, licenses, manufactures, markets and distributes generic, brand name and over-the-counter (“OTC”) products in a variety of dosage forms and therapeutic categories. Mylan is committed to setting new standards in healthcare by creating better health for a better world, and our mission is to provide the world’s 7 billion people access to high quality medicine. To do so, we innovate to satisfy unmet needs; make reliability and service excellence a habit; do what’s right, not what’s easy; and impact the future through passionate global leadership.
Mylan offers one of the industry’s broadest product portfolios, including more than 7,500 marketed products globally, to customers in more than 165 countries and territories. We operate a global, high quality vertically-integrated manufacturing platform around the world and one of the world’s largest active pharmaceutical ingredient (“API”) operations. We also operate a strong and innovative research and development (“R&D”) network that has consistently delivered a robust product pipeline, including complex products such as injectables.
Effective October 1, 2016, the Company expanded its reportable segments and now reports in three segments on a geographic basis as follows: North America, Europe and Rest of World. Comparative segment financial information have been recast for prior periods to conform to this revised segment structure.
In the fourth quarter of 2016, the Company announced restructuring programs in certain locations representing initial steps in a series of actions that are anticipated to further streamline our operations globally. The Company continues to develop the details of the cost reduction initiatives, including workforce actions and other potential restructuring activities beyond the programs already announced. During the three months ended March 31, 2017, the Company recorded pre-tax charges of $23.1 million, of which $11.5 million were non-cash asset impairment charges. The continued restructuring actions are expected to be implemented through fiscal year 2018. For the restructuring activities that have been undertaken to date, the Company estimates that it will incur aggregate pre-tax charges ranging between $175.0 million and $225.0 million, inclusive of the 2016 and year to date 2017 restructuring charges of $172.8 million. In addition, as a result of the restructuring activities that have been undertaken to date, management believes the potential annual savings will be between approximately $175.0 million and $225.0 million once fully implemented, with the majority of these savings improving operating cash flow. At this time, the expenses related to the additional restructuring activities cannot be reasonably estimated.
Financial Summary
The tables below are a summary of the Company’s financial results for the three months ended March 31, 2017 compared to the prior year period:
|
| | | | | | | | | | | | | | |
| Three Months Ended | | | | |
| March 31, | | | | |
(In millions, except per share amounts) | 2017 | | 2016 | | Change | | % Change |
Total revenues | $ | 2,719.5 |
| | $ | 2,191.3 |
| | $ | 528.2 |
| | 24 | % |
Gross profit | 1,085.0 |
| | 907.0 |
| | 178.0 |
| | 20 | % |
Earnings from operations | 227.2 |
| | 105.6 |
| | 121.6 |
| | 115 | % |
Net earnings | 66.4 |
| | 13.9 |
| | 52.5 |
| | 378 | % |
Diluted earnings per ordinary share | $ | 0.12 |
| | $ | 0.03 |
| | $ | 0.09 |
| | 300 | % |
A detailed discussion of the Company’s financial results can be found below in the section titled “Results of Operations.” As part of this discussion, we also report sales performance using the non-GAAP financial measure of “constant currency” third party net sales and total revenues. This measure provides information on the change in net sales assuming that foreign currency exchange rates had not changed between the prior and current period. The comparisons presented at constant currency rates reflect comparative local currency sales at the prior year’s foreign exchange rates. We routinely evaluate our third party net sales performance at constant currency so that sales results can be viewed without the impact of foreign currency exchange rates, thereby facilitating a period-to-period comparison of our operational activities, and believe that this
presentation also provides useful information to investors for the same reason. The following table compares third party net sales on an actual and constant currency basis for each reportable segment and consolidated total revenues on an actual and constant currency basis for the three months ended March 31, 2017 and 2016.
More information about non-GAAP measures used by the Company as part of this discussion, including adjusted cost of sales, adjusted gross margins, adjusted earnings and adjusted EPS (all of which are defined below) can be found in “Item 2. Management’s Discussion and Analysis of Financial Condition and Results of Operations - Results of Operations - Use of Non-GAAP Financial Measures.”
Results of Operations
Three Months Ended March 31, 2017 Compared to Three Months Ended March 31, 2016
|
| | | | | | | | | | | | | | | | | | | | | |
| Three Months Ended |
| March 31, |
(In millions) | 2017 | | 2016 | | % Change | | 2017 Currency Impact (1) | | 2017 Constant Currency Revenues | | Constant Currency % Change (2) |
Third party net sales | | | | | | | | | | | |
North America (3) | $ | 1,214.9 |
| | $ | 1,157.5 |
| | 5 | % | | $ | (2.2 | ) | | $ | 1,212.7 |
| | 5 | % |
Europe (3) | 892.0 |
| | 584.3 |
| | 53 | % | | 24.3 |
| | 916.3 |
| | 57 | % |
Rest of World (3) | 580.5 |
| | 434.3 |
| | 34 | % | | (12.7 | ) | | 567.8 |
| | 31 | % |
Total third party net sales (3) | 2,687.4 |
| | 2,176.1 |
| | 23 | % | | 9.4 |
| | 2,696.8 |
| | 24 | % |
| | | | | | | | | | | |
Other third party revenues | 32.1 |
| | 15.2 |
| | 111 | % | | 0.2 |
| | 32.3 |
| | 113 | % |
Consolidated total revenues | $ | 2,719.5 |
| | $ | 2,191.3 |
| | 24 | % | | $ | 9.6 |
| | $ | 2,729.1 |
| | 25 | % |
____________
| |
(1) | Currency impact is shown as unfavorable (favorable). |
| |
(2) | The constant currency percentage change is derived by translating third party net sales or revenues for the current period at prior year comparative period exchange rates, and in doing so shows the percentage change from 2017 constant currency third party net sales or revenues to the corresponding amount in the prior year. |
| |
(3) | Effective October 1, 2016, the Company expanded its reportable segments as follows: North America, Europe and Rest of World. As a result, the amounts previously reported under the Specialty segment have been recast to North America and amounts related to Brazil are included in Rest of World for all periods presented. |
Total Revenues
For the current quarter, Mylan reported total revenues of $2.72 billion, compared to $2.19 billion for the comparable prior year period, representing an increase of $528.2 million, or 24%. Total revenues include both net sales and other revenues from third parties. Third party net sales for the current quarter were $2.69 billion, compared to $2.18 billion for the comparable prior year period, representing an increase of $511.3 million, or 23%. Other third party revenues for the current quarter were $32.1 million, compared to $15.2 million for the comparable prior year period, an increase of $16.9 million. The increase in other third party revenues was principally the result of an increase in royalty income we are now receiving as a result of the acquisition of Meda.
The increase in total revenues was the result of third party net sales growth in all segments. Contributing to this increase were net sales from the acquisitions of Meda and the non-sterile, topicals-focused business (the “Topicals Business”), together totaling approximately $606.6 million. This increase was partially offset by a net decrease in net sales from new product introductions and existing products of approximately $85.8 million. The decrease from existing products was due to a combination of lower pricing and volumes in the current period. Mylan’s current quarter total revenues was unfavorably impacted by the effect of foreign currency translation. The unfavorable impact of foreign currency translation on current quarter revenues was approximately $10 million. As such, constant currency total revenues increased $537.8 million, or 25%.
From time to time, a limited number of our products may represent a significant portion of our net sales, gross profit and net earnings. Generally, this is due to the timing of new product introductions and the amount, if any, of additional competition in the market. Our top ten products in terms of sales, in the aggregate, represented approximately 19% and 26% of the Company’s net sales for the three months ended March 31, 2017 and 2016, respectively.
Mylan offers one of the industry’s broadest product portfolios, including generic, brand name and over-the-counter (“OTC”) products in a variety of dosage forms and therapeutic categories. Generic products, particularly in the U.S., generally contribute most significantly to revenues and gross margins at the time of their launch, even more so in periods of market exclusivity, or in periods of limited generic competition. As such, the timing of new product introductions can have a significant
impact on the Company’s financial results. The entrance into the market of additional competition generally has a negative impact on the volume and pricing of the affected products. Additionally, pricing is often affected by factors outside of the Company’s control.
For branded products, the majority of the product’s commercial value is usually realized during the period in which the product has market exclusivity. In the U.S. and some other countries, when market exclusivity expires and generic versions of a product are approved and marketed, there can often be very substantial and rapid declines in the branded product’s sales. OTC products also participate in a competitive environment that includes both branded and private label products. In the OTC space, value is realized through innovation, access and consumer activation.
Third party net sales are derived from our three geographic reporting segments: North America, Europe and Rest of World. The graph below shows third party net sales by segment for the three months ended March 31, 2017 and 2016 and the increase period over period:
North America Segment
Third party net sales from North America increased by $57.4 million or 5% during the three months ended March 31, 2017 when compared to the prior year period. This increase was primarily due to net sales from the acquisitions of Meda and the Topicals Business which totaled approximately $182.1 million. These increases were partially offset by a net decrease in net sales from new product introductions and lower volume and pricing on existing products. As anticipated, the U.S. generics products experienced price erosion in the mid-single digits. Sales of the EpiPen® Auto-Injector declined in the current quarter as a result of increased competition and the impact of the launch of the authorized generic. The impact of foreign currency translation on current period third party net sales was less than 1% within North America.
Europe Segment
Third party net sales from Europe increased by $307.7 million or 53% during the three months ended March 31, 2017 when compared to the prior year period. The increase was primarily the result of net sales from the acquisition of Meda which totaled approximately $337.8 million. This increase was partially offset by a net decrease in net sales from new product introductions and lower volume and pricing on existing products. The unfavorable impact of foreign currency translation on current period third party net sales was $24 million, or 4% within Europe. As such, constant currency third party net sales increased by approximately $332 million, or 57% when compared to the prior year period.
The acquisition of Meda significantly increased our operations and revenues throughout Europe, but particularly in France, Italy, Germany and Sweden. In France, we remain the generics market leader.
Certain markets within Europe in which we do business have undergone government-imposed price reductions, and further government-imposed price reductions are expected in the future. Such measures, along with the tender systems discussed below, are likely to have a negative impact on sales and gross profit in these markets. However, government initiatives in certain markets that appear to favor generic products could help to mitigate this unfavorable effect by increasing rates of generic substitution and penetration.
A number of markets in which we operate in Europe have implemented, or may implement, tender systems for generic pharmaceuticals in an effort to lower prices. Generally speaking, tender systems can have an unfavorable impact on sales and
profitability. Under such tender systems, manufacturers submit bids that establish prices for generic pharmaceutical products. Upon winning the tender, the winning company will receive preferential reimbursement for a period of time. The tender system often results in companies underbidding one another by proposing low pricing in order to win the tender. The loss of a tender by a third party to whom we supply API can also have a negative impact on our sales and profitability. Sales continue to be negatively affected by the impact of tender systems.
Rest of World Segment
Third party net sales from Rest of World increased by $146.2 million, or 34% during the three months ended March 31, 2017 when compared to the prior year period. This increase was primarily driven by the acquisition of Meda which contributed net sales of approximately $86.7 million. In addition, net sales from existing products increased principally as a result of higher sales from our anti-retroviral (“ARV”) franchise. Throughout the segment, sales from new products and higher volumes on existing products more than offset lower pricing. Third party net sales from Rest of World were favorably impacted by the effect of foreign currency translation by approximately $13 million, or 3% during the three months ended March 31, 2017. As such, constant currency third party net sales increased by approximately $133 million, or 31%.
In addition to third party net sales, the Rest of World segment supplies finished dosage form (“FDF”) generic products and API, primarily from Mylan India, to Mylan subsidiaries in conjunction with the Company’s vertical integration strategy. Intercompany sales related to this strategy were approximately $89.8 million and $71.2 million in the three months ended March 31, 2017 and 2016, respectively. These intercompany sales eliminate within, and therefore are not included in the consolidated third party net sales.
In Australia, third party net sales increased primarily as a result of new product introductions, and to a lesser extent, the acquisition of Meda. Third party net sales in Japan were essentially flat when compared to the prior year period. As in Europe, both Australia and Japan have undergone government-imposed price reductions that have had, and could continue to have, a negative impact on sales and gross profit in these markets.
As a result of the acquisition of Meda, we have significantly expanded and strengthened our presence in emerging markets including China, Southeast Asia and the Middle East. These markets provide opportunities for future growth and expansion and are complemented by Mylan’s historical presence in India, Brazil and certain countries in Africa (including South Africa).
Cost of Sales and Gross Profit
Cost of sales increased from $1.28 billion for the three months ended March 31, 2016 to $1.63 billion for the three months ended March 31, 2017, corresponding to the increase in sales. Cost of sales was primarily impacted by purchase accounting related amortization of acquired intangible assets, acquisition related costs and restructuring and other special items, which are described further in the section titled Use of Non-GAAP Financial Measures. Gross profit for the three months ended March 31, 2017 was $1.09 billion and gross margins were 40%. For the three months ended March 31, 2016, gross profit was $907.0 million and gross margins were 41%. Gross margins were negatively impacted in the current quarter by increased amortization expense as a result of the acquisitions of Meda and the Topicals Business by approximately 365 basis points, lower gross profit from the sales of existing products in North America, including the EpiPen® Auto-Injector, by approximately 250 basis points, partially offset by the contributions from the acquired businesses noted above. Adjusted gross margins were approximately 53% for the three months ended March 31, 2017, compared to approximately 54% for the three months ended March 31, 2016. For the quarter ended March 31, 2017, adjusted gross margins were negatively impacted in the current quarter as a result of lower gross profit from the sales of existing products in North America, including the EpiPen® Auto-Injector, by approximately 150 basis points, partially offset by the contributions from the acquired businesses noted above.
A reconciliation between cost of sales, as reported under U.S. GAAP, and adjusted cost of sales and adjusted gross margin for the three months ended March 31, 2017 compared to the three months ended March 31, 2016 is as follows:
|
| | | | | | | |
| Three Months Ended |
| March 31, |
(In millions) | 2017 | | 2016 |
U.S. GAAP cost of sales | $ | 1,634.5 |
| | $ | 1,284.3 |
|
Deduct: | | | |
Purchase accounting amortization and other related items | (343.3 | ) | | (243.6 | ) |
Restructuring related costs | (12.9 | ) | | (1.4 | ) |
Acquisition related items | (5.9 | ) | | (18.5 | ) |
Other special items | (7.1 | ) | | (13.8 | ) |
Adjusted cost of sales | $ | 1,265.3 |
| | $ | 1,007.0 |
|
| | | |
Adjusted gross profit (a) | $ | 1,454.2 |
| | $ | 1,184.3 |
|
|
| | | | | |
Adjusted gross margin (a) | 53 | % | | 54 | % |
____________
| |
(a) | Adjusted gross profit is calculated as total revenues less adjusted cost of sales. Adjusted gross margin is calculated as adjusted gross profit divided by total revenues. |
Operating Expenses
Research & Development Expense
R&D expense for the three months ended March 31, 2017 was $217.5 million, compared to $253.6 million for the comparable prior year period, a decrease of $36.1 million. The decrease was due to lower expenditures totaling approximately $47 million principally related to the Company’s respiratory and biologics programs due to the timing of clinical activities when compared to the prior year period. Partially offsetting these decreases was the impact of acquisitions which increased R&D expense by approximately $16 million in the current quarter.
In addition, in the first quarter of 2017, the Company entered into a joint development and marketing agreement for a respiratory product resulting in approximately $50 million in R&D expense. In the first quarter of 2016, the Company made an upfront payment for $45 million related to the Company’s collaboration agreement entered into on January 8, 2016 with Momenta Pharmaceuticals, Inc. (“Momenta”).
Selling, General & Administrative Expense
Selling, general and administrative expense (“SG&A”) for the current quarter was $631.3 million, compared to $549.3 million for the comparable prior year period, an increase of $82.0 million. The increase in SG&A is primarily due to the additional expense related to acquisitions which increased SG&A by approximately $132.1 million in the current quarter. Partially offsetting this increase were lower acquisition related costs, including consulting and legal costs, in the current quarter.
Litigation Settlements and Other Contingencies, Net
During the three months ended March 31, 2017 and 2016, the Company recorded a charge, net of $9.0 million and a gain, net of $1.5 million, respectively. During the three months ended March 31, 2017, the Company recorded a loss of $9.9 million to the fair value of contingent consideration related to the Jai Pharma Limited acquisition. In the prior year period, the gain was primarily related to the settlement of an intellectual property matter.
Interest Expense
Interest expense for the three months ended March 31, 2017 totaled $138.2 million, compared to $70.3 million for the three months ended March 31, 2016, an increase of $67.9 million. The increase in the current quarter is primarily due to $74.6 million of interest expense related to the issuance of the senior notes in June of 2016 and the Euro senior notes issued in December of 2016. This increase was partially offset by the repayment of the 1.800% Senior Notes due 2016 and the 1.350% Senior Notes due 2016 in June and November of 2016, respectively.
Other Expense, Net
Other expense, net, was $17.4 million in the current quarter, compared to $16.3 million for the comparable prior year period. Other expense, net, includes losses from equity affiliates, foreign exchange gains and losses and interest and dividend income. Other expense, net was comprised of the following for the three months ended March 31, 2017 and 2016, respectively:
|
| | | | | | | |
| Three Months Ended March 31, |
(In millions) | 2017 | | 2016 |
Losses from equity affiliates, primarily clean energy investments | $ | 33.2 |
| | $ | 30.9 |
|
Foreign exchange gains, net | (10.3 | ) | | (14.2 | ) |
Other gains, net | (5.5 | ) | | (0.4 | ) |
Other expense, net | $ | 17.4 |
| | $ | 16.3 |
|
Income Tax Provision
For the three months ended March 31, 2017, the Company recognized an income tax provision of $5.2 million, compared to $5.1 million for the comparable prior year period. The effective tax rate was 7.3% and 26.8% for the three months ended March 31, 2017 and 2016, respectively. The effective tax rate for the three months ended March 31, 2017 versus the comparable prior year period was impacted by the changing mix of income earned in jurisdictions with differing tax rates, statutory releases of certain tax uncertainties and the revaluation of deferred tax assets and liabilities in countries that changed their statutory corporate tax rate.
Use of Non-GAAP Financial Measures
Whenever the Company uses non-GAAP financial measures, we provide a reconciliation of the non-GAAP financial measures to their most directly comparable U.S. GAAP financial measure. Investors and other readers are encouraged to review the related U.S. GAAP financial measures and the reconciliation of non-GAAP measures to their most directly comparable U.S. GAAP measure and should consider non-GAAP measures only as a supplement to, not as a substitute for or as a superior measure to, measures of financial performance prepared in accordance with U.S. GAAP. Additionally, since these are not measures determined in accordance with U.S. GAAP, non-GAAP financial measures have no standardized meaning across companies, or as prescribed by U.S. GAAP and, therefore, may not be comparable to the calculation of similar measures or measures with the same title used by other companies.
Management uses these measures internally for forecasting, budgeting, measuring its operating performance, and incentive-based awards. In addition, primarily due to acquisitions, we believe that an evaluation of our ongoing operations (and comparisons of our current operations with historical and future operations) would be difficult if the disclosure of our financial results was limited to financial measures prepared only in accordance with U.S. GAAP. We believe that non-GAAP financial measures are useful supplemental information for our investors and when considered together with our U.S. GAAP financial measures and the reconciliation to the most directly comparable U.S. GAAP financial measure, provide a more complete understanding of the factors and trends affecting our operations. The financial performance of the Company is measured by senior management, in part, using adjusted metrics as described below, along with other performance metrics. Management’s annual incentive compensation is derived, in part, based on the adjusted EPS metric.
Adjusted Cost of Sales and Adjusted Gross Margin
We use the non-GAAP financial measure “adjusted cost of sales” and the corresponding non-GAAP financial measure “adjusted gross margin.” The principal items excluded from adjusted cost of sales include restructuring, acquisition related and other special items and purchase accounting amortization and other related items, which are described in greater detail above.
Adjusted Earnings and Adjusted EPS
Adjusted net earnings (“adjusted earnings”) is a non-GAAP financial measure and provides an alternative view of performance used by management. Management believes that, primarily due to acquisition activity, an evaluation of the Company’s ongoing operations (and comparisons of its current operations with historical and future operations) would be difficult if the disclosure of its financial results were limited to financial measures prepared only in accordance with U.S. GAAP. Management believes that adjusted earnings and adjusted earnings per diluted share (“adjusted EPS”) are two of the most important internal financial metrics related to the ongoing operating performance of the Company, and are therefore useful to investors and that their understanding of our performance is enhanced by these adjusted measures. Actual internal and forecasted operating results and annual budgets used by management include adjusted earnings and adjusted EPS.
The significant items excluded from adjusted cost of sales, adjusted earnings and adjusted EPS include:
Purchase Accounting Amortization and Other Related Items
The ongoing impact of certain amounts recorded in connection with acquisitions is excluded from adjusted cost of sales, adjusted earnings and adjusted EPS. These amounts include the amortization of intangible assets, inventory step-up and intangible asset impairment charges, including in-process research and development.
Upfront and Milestone-Related R&D Expenses
These expenses and payments are excluded from adjusted earnings and adjusted EPS because they generally occur at irregular intervals and are not indicative of the Company’s ongoing operations. Also included in this adjustment are certain expenses related to the Company’s collaboration agreement with Momenta including certain milestone related costs. Such costs include payments related to Mylan’s future decisions, on a product by product basis, to continue with the development of such product in the collaboration after certain R&D work is performed. Related amounts are excluded from adjusted earnings as Mylan considers such payments as additional upfront buy-in payments for the products.
Accretion of Contingent Consideration Liability and Other Fair Value Adjustments
The impact of changes to the fair value of contingent consideration and accretion expense are excluded from adjusted earnings and adjusted EPS because they are not indicative of the Company’s ongoing operations due to the variability of the amounts and the lack of predictability as to the occurrence and/or timing and management believes it is helpful to understanding the underlying, ongoing operational performance of the business.
Restructuring, Acquisition Related and Other Special Items
Costs related to restructuring, acquisition and integration activities and other actions are excluded from adjusted cost of sales, adjusted earnings and adjusted EPS, as applicable. These amounts include items such as:
| |
• | Costs related to formal restructuring programs and actions, including costs associated with facilities to be closed or divested, employee separation costs, impairment charges, accelerated depreciation, incremental manufacturing variances, equipment relocation costs and other restructuring related costs; |
| |
• | Certain acquisition related remediation and integration and planning costs, as well as other costs associated with acquisitions such as advisory and legal fees and certain financing related costs, and other business transformation and/or optimization initiatives, which are not part of a formal restructuring program, including employee separation and post-employment costs; |
| |
• | The pre-tax loss of the Company’s clean energy investments, whose activities qualify for income tax credits under Section 45 of the U.S. Internal Revenue Code of 1986, as amended (the “Code”); only included in adjusted earnings and adjusted EPS is the net tax effect of the entity’s activities; and |
| |
• | Certain costs to further develop and optimize our global enterprise resource planning systems, operations and supply chain. |
The Company has undertaken restructurings and other optimization initiatives of differing types, scope and amount during the covered periods and, therefore, these charges should not be considered non-recurring; however, management excludes these amounts from adjusted earnings and adjusted EPS because it believes it is helpful to understanding the underlying, ongoing operational performance of the business.
Litigation Settlements, Net
Charges and gains related to legal matters, such as those discussed in the Notes to interim financial statements — Note 19 Litigation are generally excluded from adjusted earnings and adjusted EPS. Normal, ongoing defense costs of the Company made in the normal course of our business are not excluded.
Reconciliation of Adjusted Earnings and Adjusted EPS
A reconciliation between net earnings and diluted earnings per share, as reported under U.S. GAAP, and adjusted earnings and adjusted EPS for the periods shown follows:
|
| | | | | | | | | | | | | | | |
| Three Months Ended March 31, |
(In millions, except per share amounts) | 2017 | | 2016 |
U.S. GAAP net earnings and U.S. GAAP diluted earnings per share | $ | 66.4 |
| | $ | 0.12 |
| | $ | 13.9 |
| | $ | 0.03 |
|
Purchase accounting related amortization (primarily included in cost of sales) | 349.2 |
| | | | 249.3 |
| | |
Litigation settlements, net | (0.9 | ) | | | | (1.5 | ) | | |
Interest expense (primarily related to clean energy investment financing) | 7.3 |
| | | | 5.7 |
| | |
Accretion of contingent consideration liability and other fair value adjustments | 17.7 |
| | | | 10.0 |
| | |
Clean energy investments pre-tax loss (a) | 22.3 |
| | | | 25.5 |
| | |
Acquisition related costs (primarily included in SG&A and cost of sales) (b) | 31.3 |
| | | | 53.2 |
| | |
Restructuring related costs (c) | 23.1 |
| | | | 9.8 |
| | |
Other special items included in: | | | | | | | |
Cost of sales | 7.1 |
| | | | 13.8 |
| | |
Research and development expense (d) | 65.1 |
| | | | 66.1 |
| | |
Selling, general and administrative expense | 5.9 |
| | | | 6.8 |
| | |
Other expense, net | 6.1 |
| | | | 2.2 |
| | |
Tax effect of the above items and other income tax related items | (100.8 | ) | | | | (68.5 | ) | | |
Adjusted net earnings and adjusted EPS | $ | 499.8 |
| | $ | 0.93 |
| | $ | 386.3 |
| | $ | 0.76 |
|
Weighted average diluted ordinary shares outstanding | 536.9 |
| | | | 509.6 |
| | |
____________
Significant items for the three months ended March 31, 2017 include the following:
| |
(a) | Adjustment represents exclusion of the pre-tax loss related to Mylan's clean energy investments and related financing, excluding interest expense, the activities of which qualify for income tax credits under Section 45 of the Code. The amount is included in other expense, net in the Condensed Consolidated Statements of Operations. |
| |
(b) | Acquisition related costs primarily relate to acquisition and integration activities, including ongoing activities. Included in SG&A for the three months ended March 31, 2017 is approximately $24.1 million, primarily related to consulting, professional and legal costs. |
| |
(c) | Refer to Note 17 Restructuring included in Item 1 in this Form 10-Q. Of the total amount, approximately $12.9 million is included in cost of sales, $1.3 million is included in R&D and $8.9 million is included in SG&A. |
| |
(d) | R&D expense includes an upfront expense of approximately $50 million related to a joint development and marketing agreement for a respiratory product, $5.8 million related to Momenta collaboration expense and other similar smaller agreements. |
Liquidity and Capital Resources
Our primary source of liquidity is cash provided by operations, which was $452.9 million for the three months ended March 31, 2017. We believe that cash provided by operating activities and available liquidity will continue to allow us to meet our needs for working capital, capital expenditures and interest and principal payments on debt obligations. Nevertheless, our ability to satisfy our working capital requirements and debt service obligations, or fund planned capital expenditures, will substantially depend upon our future operating performance (which will be affected by prevailing economic conditions), and financial, business and other factors, some of which are beyond our control.
Operating Activities
Net cash provided by operating activities increased by $372.4 million to $452.9 million for the three months ended March 31, 2017, as compared to net cash provided by operating activities of $80.5 million for the three months ended March 31, 2016. Cash provided by operating activities is derived by net earnings adjusted for non-cash operating items, gains and losses attributed to investing and financing activities and changes in operating assets and liabilities resulting from timing differences between the receipts and payments of cash, including changes in cash primarily reflecting the timing of cash collections from customers, payments to vendors and employees and tax payments in the ordinary course of business.
The net increase in cash provided by operating activities was principally due to the following:
| |
• | net earnings for the three months ended March 31, 2017 increased $52.5 million when compared to the prior year period, principally as a result of an increase in earnings from operations. Other significant factors impacting cash provided by operating activities in the current year include the following: |
| |
◦ | an increase in non-cash expenses of $140.8 million, principally a result of increased depreciation and amortization as a result of recent acquisitions of approximately $118.4 million; |
| |
◦ | a net increase in the amount of cash provided by changes in accounts receivable, including estimated sales allowances, of $203.2 million, reflecting the timing of sales, cash collections and customer credits issued related to sales allowances; |
| |
◦ | a net decrease of $117.2 million in the amount of cash used through changes in inventory balances; and |
| |
◦ | an net increase in the amount of cash provided by changes in other operating assets and liabilities of $134.5 million, primarily due to higher amounts of accrued interest as a result of the long-term debt issuances during 2016 and the timing of interest payments related to those debt instruments. |
These items were partially offset by the following:
| |
• | a net increase in the amount of cash used through changes in trade accounts payable of $185.5 million as a result of the timing of cash payments; and |
| |
• | a net increase in the amount of cash used through changes in income taxes of $90.3 million as a result of the level and timing of estimated tax payments made during the current period. |
Investing Activities
Cash used in investing activities was $164.1 million for the three months ended March 31, 2017, as compared to $160.0 million for the three months ended March 31, 2016, a net increase of $4.1 million.
In 2017, significant items in investing activities included the following:
| |
• | cash paid for acquisitions, net totaling approximately $71.6 million related to the acquisition of the remaining non-tendered shares of Meda in the compulsory acquisition proceeding; |
| |
• | payments for product rights and other, net totaled approximately $77.9 million, which included a payment of $50 million related to the acquisition of intellectual property rights for the Cold-EEZE® brand cold remedy line; |
| |
• | proceeds from the sale of certain European assets for approximately $31.1 million; |
| |
• | a decrease in restricted cash of $12.7 million in the current quarter due to amounts released from escrow for the payment of certain claims related to the Agila contingent consideration; and |
| |
• | capital expenditures, primarily for equipment and facilities, totaled approximately $58.4 million. While there can be no assurance that current expectations will be realized, capital expenditures for the 2017 calendar year are expected to be approximately $400 million to $500 million. |
In 2016, significant items in investing activities included the following:
| |
• | payments for product rights and other, net totaled approximately $105.6 million which included a payment of $90 million related to the acquisition of certain European intellectual property rights and marketing authorizations; and |
| |
• | capital expenditures, primarily for equipment and facilities, totaled approximately $51.8 million. |
Financing Activities
Cash used in financing activities was $575.7 million for the three months ended March 31, 2017, compared to cash provided by financing activities of $30.5 million for the three months ended March 31, 2016, a net decrease of $606.2 million.
In 2017, significant items in financing activities included the following:
| |
• | the Company voluntarily prepaid $550 million of the aggregate principal amount of the 2016 Term Loans: and |
| |
• | the Company had net repayments of short-term borrowings of $17.6 million. |
In 2016, significant items in financing activities included the following:
| |
• | the Company had net short-term borrowings of $65.1 million; partially offset by |
| |
• | payments of financing fees which totaled $31.6 million primarily related to a bridge credit agreement related to the Meda acquisition. |
Capital Resources
Our cash and cash equivalents at our non-U.S. operations totaled $700.9 million at March 31, 2017. The majority of these funds represented earnings considered to be permanently reinvested to support the growth strategies of our non-U.S. subsidiaries. The Company anticipates having sufficient U.S. liquidity, including existing borrowing capacity under the 2016 Revolving Facility and the Receivables Facility combined with cash to be generated from operations, to fund foreseeable U.S. cash needs without requiring the repatriation of non-U.S. cash. If these funds are ultimately needed for the Company’s operations in the U.S., the Company may be required to accrue and pay U.S. taxes to repatriate these funds. If funds are needed from the Company’s subsidiaries that do not have an ultimate U.S. parent, the Company will generally not be required to accrue and pay taxes to repatriate these funds because its foreign parent would not be subject to tax on receipt of these distributions.
The Company has access to $2.0 billion under the 2016 Revolving Facility which also includes a $200 million subfacility for the issuance of letters of credit and a $175 million sublimit for swingline borrowings. As of March 31, 2017, we had $193.7 million available under the $200 million subfacility on our 2016 Revolving Facility for the issuance of letters of credit.
In addition to the 2016 Revolving Facility, Mylan Pharmaceuticals Inc. (“MPI”), a wholly owned subsidiary of the Company, has a $400 million receivables facility (the “Receivables Facility”), which will expire in January 2018. Although from time-to-time, the available amount of the Receivables Facility may be less than $400 million based on accounts receivable concentration limits and other eligibility requirements. Under the terms of the Receivables Facility, MPI sells certain accounts receivable to Mylan Securitization LLC, a wholly owned special purpose entity which in turn sells a percentage of ownership interest in the receivables to financial institutions and commercial paper conduits sponsored by financial institutions. As of March 31, 2017, the Company had no amounts outstanding under the Receivables Facility.
At March 31, 2017, our long-term debt totaled $14.70 billion, as compared to $15.20 billion at December 31, 2016. The decrease in long-term debt was due to the prepayment of a portion of the 2016 Term Loans during the three months ended March 31, 2017. The total long-term debt balance at March 31, 2017 was comprised primarily of $1.05 billion of term loans, $148.6 million of Medium Term Notes acquired from Meda, $13.05 billion of fixed rate senior notes and $532.8 million of floating rate senior notes. In addition, at March 31, 2017, we had $227.2 million of long-term debt classified as current and payable within the next twelve months, as compared to $223.3 million at December 31, 2016. In addition to the current portion of long-term debt, the Company has significant debt maturities in the second and fourth quarters of 2018, as the 2.600% Senior Notes due 2018 mature in June 2018, the Floating Rate Euro Notes mature in November 2018 and the 3.000% Senior Notes due 2018 mature in December 2018. The Company intends to utilize available liquidity to fund these repayments.
For additional information regarding our debt agreements refer to Note 12 Debt in Item 1 in this Form 10-Q.
Long-term Debt Maturity
Mandatory minimum repayments remaining on the outstanding notional amount of long-term debt at March 31, 2017 are as follows for each of the periods ending December 31:
The Company’s 2016 Term Loans and 2016 Revolving Facility contain customary affirmative covenants for facilities of this type, including among others, covenants pertaining to the delivery of financial statements, notices of default and certain material events, maintenance of corporate existence and rights, property, and insurance and compliance with laws, as well as customary negative covenants for facilities of this type, including limitations on the incurrence of subsidiary indebtedness, liens, mergers and certain other fundamental changes, investments and loans, acquisitions, transactions with affiliates, payments of dividends and other restricted payments and changes in our lines of business. The 2016 Term Loans and 2016 Revolving Facility contain a maximum consolidated leverage ratio financial covenant. We are compliant with these financial covenants as of March 31, 2017, and we expect to remain in compliance for the next twelve months.
Collaboration and Licensing Agreements
We periodically enter into collaboration and licensing agreements with other pharmaceutical companies for the development, manufacture, marketing and/or sale of pharmaceutical products. Our significant collaboration agreements are focused on the development, manufacturing, supply and commercialization of multiple, high-value generic biologic compounds, insulin analog products and respiratory products. Under these agreements, we have future potential milestone payments and co-development expenses payable to third parties as part of our licensing, development and co-development programs. Payments under these agreements generally become due and are payable upon the satisfaction or achievement of certain developmental, regulatory or commercial milestones or as development expenses are incurred on defined projects. Milestone payment obligations are uncertain, including the prediction of timing and the occurrence of events triggering a future obligation and are not reflected as liabilities in the Condensed Consolidated Balance Sheets, except for milestone and royalty obligations reflected as acquisition contingent consideration. These agreements may also include potential sales based milestones and call for us to pay a percentage of amounts earned from the sale of the product as a royalty or a profit share. These sales based milestones or royalty obligations may be significant depending upon the level of commercial sales for each product.
Our most significant contingent payment relates to the potential future consideration related to our December 2011 acquisition of the exclusive worldwide rights to develop, manufacture and commercialize a generic equivalent to GlaxoSmithKline’s Advair® Diskus and Seretide® Diskus incorporating Pfizer Inc.’s proprietary dry powder inhaler delivery platform (the “respiratory delivery platform”). These payments are contingent upon the occurrence of certain future events and the ultimate success of the respective projects. Given the inherent uncertainty of these events, it is unclear when, if ever, we may be required to pay such amounts or pay amounts in excess of those accrued. The Company has also recorded contingent consideration related to the acquisition of the Topicals Business, the acquisition of certain female healthcare businesses from Famy Care Limited (such businesses, “Jai Pharma Limited”), the acquisition of Agila Specialties Private Limited (“Agila”) and certain other acquisitions. The amount of contingent consideration recorded was $566.0 million and $564.6 million at March 31, 2017 and December 31, 2016, respectively. In addition, the Company expects to incur approximately $35 million to $40 million of annual accretion expense related to the increase in the net present value of the contingent consideration liability.
In conjunction with the Company’s Generic Drug User Fee Agreement goal date, on March 28, 2017, the Company received a complete response letter from FDA regarding its Abbreviated New Drug Application for the respiratory delivery platform. As of March 31, 2017, the Company has an IPR&D asset of $347.2 million and related contingent consideration liability of $436.1 million. The Company performed an analysis and valuation of the IPR&D asset and the fair value of the related contingent consideration liability through the use of a discounted cash flow model. The model contained certain key assumptions including: the expected product launch date, the number of competitors, the timing of competition and a discount
factor based on an industry specific weighted average cost of capital. Based on the analysis performed, the Company determined that the IPR&D asset was not impaired at March 31, 2017. Additionally, no significant fair value adjustment was required for the contingent consideration. However, resolution of the matters with the FDA, market conditions and other factors may result in significant changes in the projections and assumptions utilized in the discounted cash flow model, which could lead to material adjustments to the recorded amounts.
We are actively pursuing, and are currently involved in, joint projects related to the development, distribution and marketing of both generic and branded products. Many of these arrangements provide for payments by us upon the attainment of specified milestones. While these arrangements help to reduce the financial risk for unsuccessful projects, fulfillment of specified milestones or the occurrence of other obligations may result in fluctuations in cash flows.
Other Commitments
We are involved in various legal proceedings that are considered normal to our business. While it is not possible to predict the outcome of such proceedings, an adverse outcome in any of these proceedings could materially affect our business, financial condition, results of operations, and cash flows and could cause the market value of our ordinary shares to decline. We have approximately $650 million accrued for legal contingencies. For certain contingencies assumed in conjunction with the acquisition of the former Merck Generics business, Merck KGaA, the seller, has agreed to indemnify Mylan. Strides has also agreed to indemnify Mylan for certain contingencies related to our acquisition of Agila. The inability or denial of Merck KGaA, Strides Arcolab, or another indemnitor or insurer to pay on an indemnified claim could have a material adverse effect on our business, financial condition, results of operations, cash flows and/or ordinary share price.
We are continuously evaluating the potential acquisition of products, as well as companies, as a strategic part of our future growth. Consequently, we may utilize current cash reserves or incur additional indebtedness to finance any such acquisitions, which could impact future liquidity. In addition, on an ongoing basis, we review our operations including the evaluation of potential divestitures of products and businesses as part of our future strategy. Any divestitures could impact future liquidity.
Other Developments
Effective April 22, 2017, the Company entered into a six-year collective bargaining agreement with the United Steel, Paper and Forestry, Rubber, Manufacturing, Energy, Allied Industrial and Service Workers International Union and its Local Union 8-957 AFL-CIO which agreement governs certain production and maintenance employees at the Company’s largest manufacturing site in Morgantown, West Virginia. The agreement expires March 17, 2023.
| |
ITEM 3. | QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK |
For a discussion of the Company’s market risk, see “Item 7A. Quantitative and Qualitative Disclosures about Market Risk” in Mylan N.V.’s Annual Report filed on Form 10-K for the year ended December 31, 2016, as amended.
| |
ITEM 4. | CONTROLS AND PROCEDURES |
An evaluation was performed under the supervision and with the participation of the Company’s management, including the Principal Executive Officer and the Principal Financial Officer, of the effectiveness of the design and operation of the Company’s disclosure controls and procedures as of March 31, 2017. Based upon that evaluation, the Principal Executive Officer and the Principal Financial Officer concluded that the Company’s disclosure controls and procedures were effective.
Management identified the following change in the Company’s internal control over financial reporting (“ICFR”) that occurred during the quarter that has materially affected, or is reasonably likely to materially affect, the Company’s ICFR. During the quarter ended March 31, 2017, the Company continued to implement and utilize a new Enterprise Resource Planning (“ERP”) system in certain countries, which, when completed, will handle the business, financial and administrative processes for the Company. The Company has modified and will continue to modify its internal controls relating to its business and financial processes throughout the entire ERP system implementation, which is expected to progress through the end of 2017. While the Company believes that this new system and the related changes to internal controls will ultimately strengthen its ICFR, there are inherent risks in implementing any new ERP system and the Company will continue to evaluate and test control changes in order to provide certification as of its fiscal year ending December 31, 2017 on the effectiveness of its ICFR.
PART II — OTHER INFORMATION
For information regarding legal proceedings, refer to Note 19 Litigation, in the accompanying Notes to interim financial statements in this Quarterly Report.
There have been no material changes in the Company’s risk factors from those disclosed in Mylan’s Annual Report on Form 10-K for the year ended December 31, 2016, as amended.
|
| |
ITEM 6. EXHIBITS |
10.1 | Amendment to Amended and Restated 2003 Long-Term Incentive Plan, adopted as of February 23, 2017* |
| |
10.2 | Form of Restricted Stock Unit Award Agreement under the 2003 Long-Term Incentive Plan for Heather Bresch and Rajiv Malik for awards granted on or after February 23, 2017* |
| |
10.3 | Form of Performance-Based Restricted Stock Unit Award Agreement under the 2003 Long-Term Incentive Plan for Heather Bresch and Rajiv Malik for awards granted on or after February 23, 2017* |
| |
10.4 | Form of Waiver Letter with respect to Specified Award Agreements by and between Mylan N.V and Heather Bresch and Rajiv Malik, February 23, 2017* |
| |
10.5 | Executive Employment Agreement, dated March 24, 2017 and effective April 1, 2017, between Mylan Inc. and Daniel M. Gallagher* |
| |
10.6 | Transition and Succession Agreement, dated March 24, 2017, between Mylan Inc. and Daniel M. Gallagher* |
| |
10.7 | Form of Performance-Based Restricted Stock Unit Award Agreement under the One-Time Special Five-Year Performance-Based Realizable Value Incentive Program for Daniel M. Gallagher* |
| |
31.1 | Certification of Principal Executive Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. |
| |
31.2 | Certification of Principal Financial Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. |
| |
32 | Certification of Principal Executive Officer and Principal Financial Officer pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. |
| |
101.INS | XBRL Instance Document |
| |
101.SCH | XBRL Taxonomy Extension Schema |
| |
101.CAL | XBRL Taxonomy Extension Calculation Linkbase |
| |
101.DEF | XBRL Taxonomy Definition Linkbase |
| |
101.LAB | XBRL Taxonomy Extension Label Linkbase |
| |
101.PRE | XBRL Taxonomy Extension Presentation Linkbase |
| |
* | Denotes management contract or compensatory plan or arrangement. |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
|
| | |
| | Mylan N.V. (Registrant) |
| | |
| By: | /s/ HEATHER BRESCH |
| | Heather Bresch |
| | Chief Executive Officer |
| | (Principal Executive Officer) |
May 10, 2017
|
| | |
| | /s/ KENNETH S. PARKS |
| | Kenneth S. Parks |
| | Chief Financial Officer |
| | (Principal Financial Officer) |
May 10, 2017
EXHIBIT INDEX
|
| |
10.1 | Amendment to Amended and Restated 2003 Long-Term Incentive Plan, adopted as of February 23, 2017* |
| |
10.2 | Form of Restricted Stock Unit Award Agreement under the 2003 Long-Term Incentive Plan for Heather Bresch and Rajiv Malik for awards granted on or after February 23, 2017* |
| |
10.3 | Form of Performance-Based Restricted Stock Unit Award Agreement under the 2003 Long-Term Incentive Plan for Heather Bresch and Rajiv Malik for awards granted on or after February 23, 2017* |
| |
10.4 | Form of Waiver Letter with respect to Specified Award Agreements by and between Mylan N.V and Heather Bresch and Rajiv Malik, February 23, 2017* |
| |
10.5 | Executive Employment Agreement, dated March 24, 2017 and effective April 1, 2017, between Mylan Inc. and Daniel M. Gallagher* |
| |
10.6 | Transition and Succession Agreement, dated March 24, 2017, between Mylan Inc. and Daniel M. Gallagher* |
| |
10.7 | Form of Performance-Based Restricted Stock Unit Award Agreement under the One-Time Special Five-Year Performance-Based Realizable Value Incentive Program for Daniel M. Gallagher* |
| |
31.1 | Certification of Principal Executive Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. |
| |
31.2 | Certification of Principal Financial Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. |
| |
32 | Certification of Principal Executive Officer and Principal Financial Officer pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. |
| |
101.INS | XBRL Instance Document |
| |
101.SCH | XBRL Taxonomy Extension Schema |
| |
101.CAL | XBRL Taxonomy Extension Calculation Linkbase |
| |
101.DEF | XBRL Taxonomy Definition Linkbase |
| |
101.LAB | XBRL Taxonomy Extension Label Linkbase |
| |
101.PRE | XBRL Taxonomy Extension Presentation Linkbase |
| |
* | Denotes management contract or compensatory plan or arrangement. |