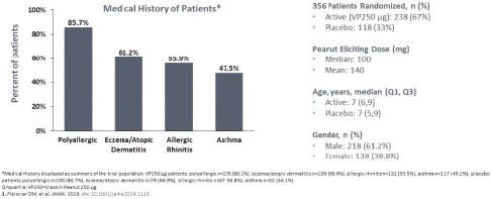
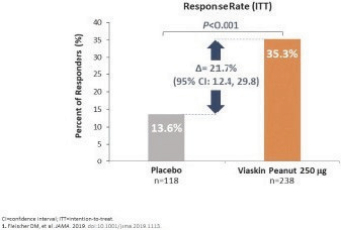
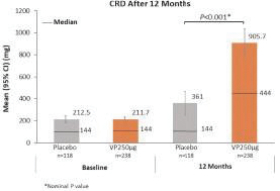
Additionally, in the United States, there have been several recent Congressional inquiries and federal and state legislative activity designed to, among other things, bring more transparency to drug pricing, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for drugs. At the federal level, the Trump administration used several means to propose or implement drug pricing reform, including through federal budget proposals, executive orders and policy initiatives. For example, on July 24, 2020 and September 13, 2020, the Trump administration announced several executive orders related to prescription drug pricing that seek to implement several of the administration’s proposals. As a result, the FDA concurrently released a final rule and guidance in September 2020, providing pathways for states to build and submit importation plans for drugs from Canada. Further, on November 20, 2020, the U.S. Department of Health and Human Services, or HHS, finalized a regulation removing safe harbor protection for price reductions from pharmaceutical manufacturers to plan sponsors under Medicare Part D, either directly or through pharmacy benefit managers, unless the price reduction is required by law. The implementation of the rule has been delayed by the Biden administration from January 1, 2022 to January 1, 2023 in response to ongoing litigation. The rule also creates a new safe harbor for price reductions reflected at the point-of-sale, as well as a new safe harbor for certain fixed fee arrangements between pharmacy benefit managers and manufacturers, the implementation of which have also been delayed until January 1, 2023. On November 20, 2020, CMS issued an interim final rule implementing the Trump administration’s Most Favored Nation executive order, which would tie Medicare Part B payments for certain physician-administered drugs to the lowest price paid in other economically advanced countries, effective January 1, 2021. As a result of litigation challenging the Most Favored Nation model, on December 27, 2021, CMS published a final rule that rescinded the Most Favored Nation model interim final rule. In July 2021, the Biden administration released an executive order “Promoting Competition in the American Economy,” with multiple provisions aimed at prescription drugs. In response to Biden’s executive order, on September 9, 2021, HHS released a Comprehensive Plan for Addressing High Drug Prices that outlines principles for drug pricing reform and sets out a variety of potential legislative policies that Congress could pursue as well as potential administrative actions HHS can take to advance these principles. No legislation or administrative actions have been finalized to implement these principles. It is unclear whether these or similar policy initiatives will be implemented in the future. At the state level, legislatures have increasingly passed legislation and implemented regulations designed to control pharmaceutical and biological product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing. Additional legislative proposals to reform healthcare and government insurance programs, along with the trend toward managed healthcare in the United States, could influence the purchase of medicines and reduce demand and prices for our products, if approved. This could harm our or our collaborators’ ability to market any products and generate revenues. Cost containment measures that healthcare payors and providers are instituting and the effect of further healthcare reform could significantly reduce potential revenues from the sale of any of our product candidates approved in the future, and could cause an increase in our compliance, manufacturing, or other operating expenses. It is also possible that additional governmental action will be taken in response to the COVID-19 pandemic.
In addition, in some foreign countries, the proposed pricing for a drug must be approved before it may be lawfully marketed. The requirements governing drug pricing vary widely from country to country. For example, the European Union provides options for its Member States to restrict the range of medicinal products for which their national health insurance systems provide reimbursement and to control the prices of medicinal products for human use. A Member State may approve a specific price for the medicinal product or it may instead adopt a system of direct or indirect controls on the profitability of the company placing the medicinal product on the market. In France, for example, effective access to the market can be achieved either at a free price, decided by the pharmaceutical company, or with a system of cover/reimbursement with a price regulated by the authorities. In this case, the future products must be included, for coverage by hospitals, on the list of proprietary medicinal