Exhibit 99.1
 Empowering Cells to Change Lives
Empowering Cells to Change Lives
SQZ Biotechnologies Initial First-In-Human Data Demonstrates Investigational Cell Therapy is Safe and Can Stimulate Immune Responses
in Certain Patients with Advanced or Metastatic HPV16+ Tumors
Doses of Autologous Cell Therapy Manufactured in Under 24 Hours
and Available to Patients in Approximately One Week
Clinical Trial Findings Presented at the
American Society of Clinical Oncology Annual Meeting
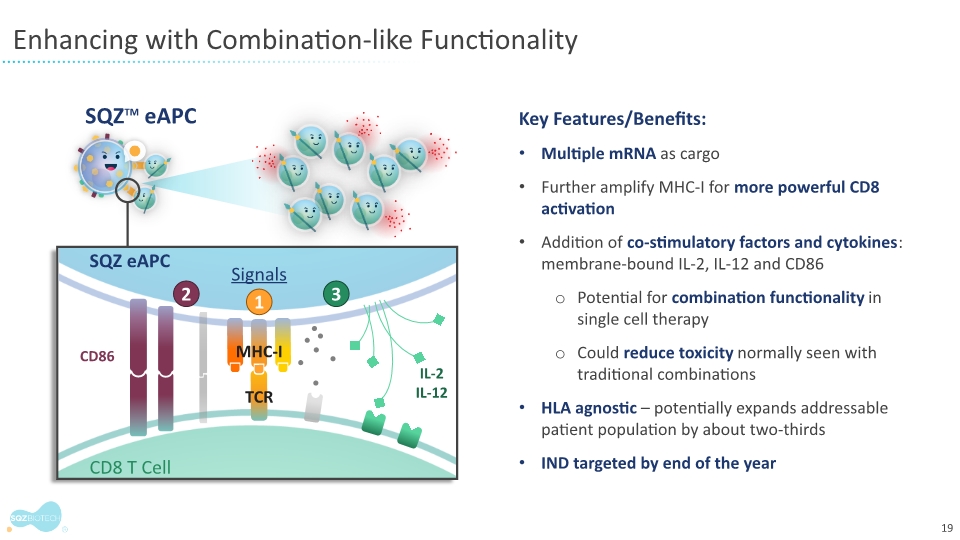
WATERTOWN, Mass., June 4, 2021 – SQZ Biotechnologies Company (NYSE: SQZ), focused on unlocking the full potential of cell therapies for multiple therapeutic areas, today presented initial results from its ongoing Phase 1 clinical trial of SQZ-PBMC-HPV demonstrating that the investigational cell therapy is safe and well-tolerated and can stimulate immune responses in certain patients with advanced or metastatic Human Papillomavirus positive (HPV16+) tumors. The trial also showed that the company’s clinical stage manufacturing process of its autologous cell therapy is fast and reliable. The monotherapy stage trial data of the company’s first Antigen Presenting Cell (APC) platform candidate was presented at the 2021 American Society of Clinical Oncology (ASCO) annual meeting; poster presentation 2536.
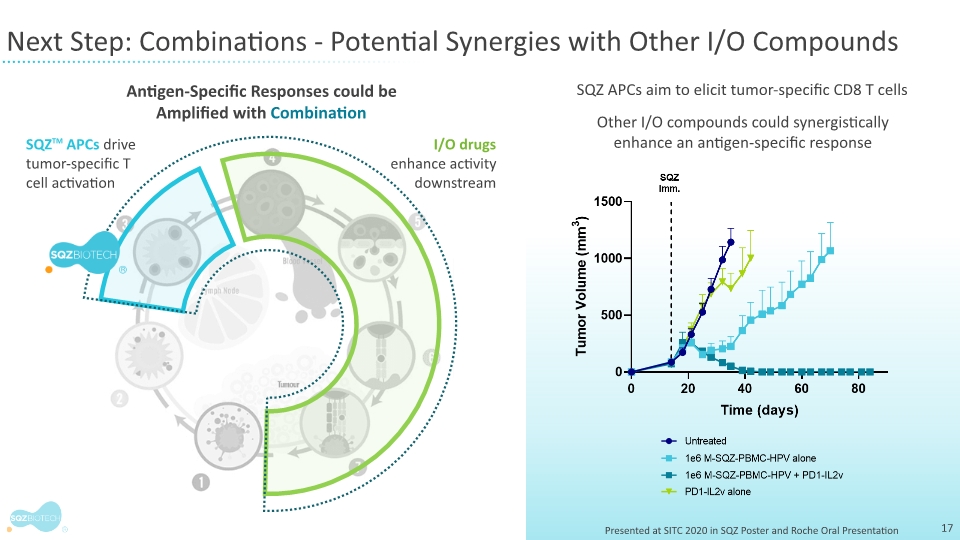
“Our vision is to make cell therapies that are safe and available with rapid turnaround times, allowing access to patients who need them,” said Oliver Rosen, M.D., chief medical officer at SQZ Biotechnologies. “The company’s first-in-human data of a cell-based therapeutic vaccine are encouraging and an important first step towards validation of our directed immunity approach. Within this small trial of patients with very advanced disease, four patients who had progressed after multiple prior therapies achieved stable disease. These early outcomes, combined with encouraging safety data and fast clinical-scale manufacturing times, support our plans to initiate the trial’s safety combination phase with immune checkpoint inhibitors.”
Safety & Tolerability
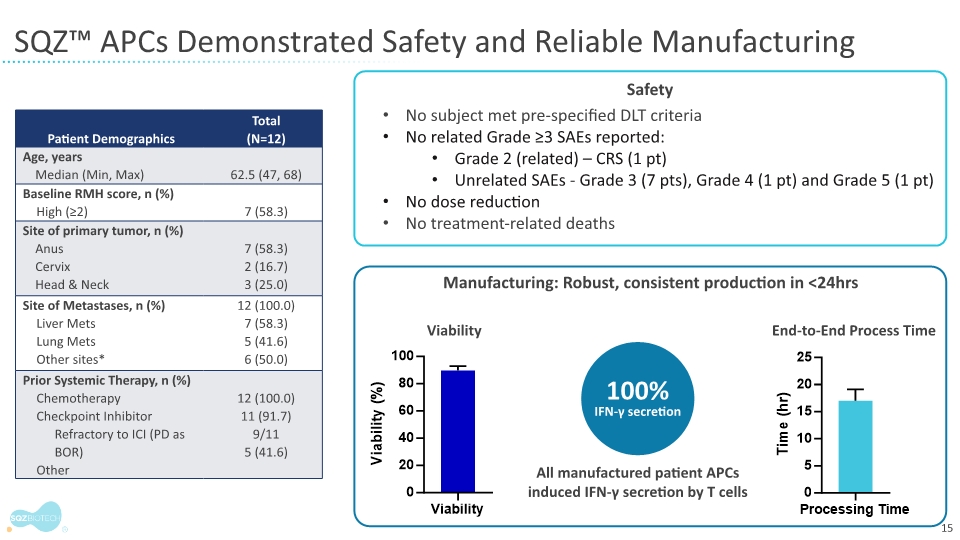
A primary outcome measure in the monotherapy dose escalation phase of the trial is safety and tolerability. Findings from the trial show that SQZ-PBMC-HPV was safe and well-tolerated at all tested dose levels with patients receiving 2 to 10 doses. No dose-limiting toxicities were observed.
“Overall, SQZ-PBMC-HPV has been safe and well tolerated by patients, even advanced patients as we have seen in this study,” said study author Antonio Jimeno, M.D., Ph.D., Professor of Medicine, Oncology and Otolaryngology, University of Colorado School of Medicine, and Co-Leader, Development Therapeutics Program, University of Colorado Cancer Center. “I look forward to completing the single agent portion of the trial and advancing into the combinations of SQZ-PBMC-HPV with immunotherapies.”
There were no grade 3 or higher treatment related serious adverse events (SAEs). In one patient, a grade 2 cytokine release syndrome and immune-related reaction was observed. A related grade 3 adverse event (AE, anemia) was observed in another patient.
Manufacturability
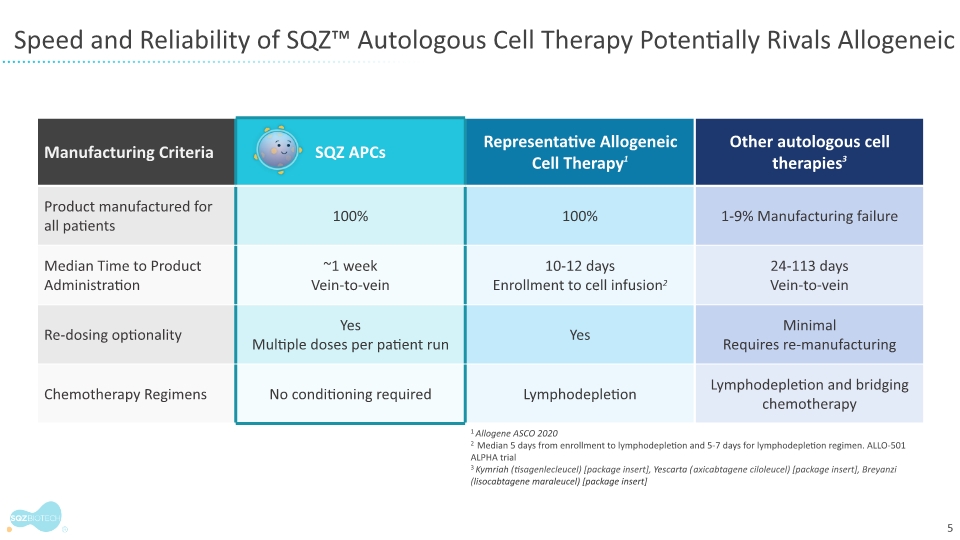
Manufacturing feasibility is a secondary outcome measure in the monotherapy phase of the trial. All patient batches were produced under current good manufacturing practice regulations, met specifications, and yielded multiple cryopreserved doses in less than 24 hours.
The findings show that doses of SQZ-PBMC-HPV were released and available for administration approximately one week from the time a patient’s cells were drawn. Antigen presentation was confirmed in all patient batches independent of individual patient medical history or prognostic score.
Patient Characteristics & Immune Response Biomarkers
The clinical trial enrolled patients with HPV16+ cancers progressing after unlimited prior lines of therapy. The 12 enrolled patients had very advanced disease:
|
|
• |
Median number of prior cancer treatments was four with one patient having received seven prior treatments |
|
|
• |
Eleven patients previously treated with an immune checkpoint inhibitor (ICI) |
|
|
• |
Six of the 12 patients had a Royal Marsden Hospital (RMH) score of 2. (RMH scores range from 0-to-3, with scores of 2 and higher predicting poor prognosis and short life expectancy) |
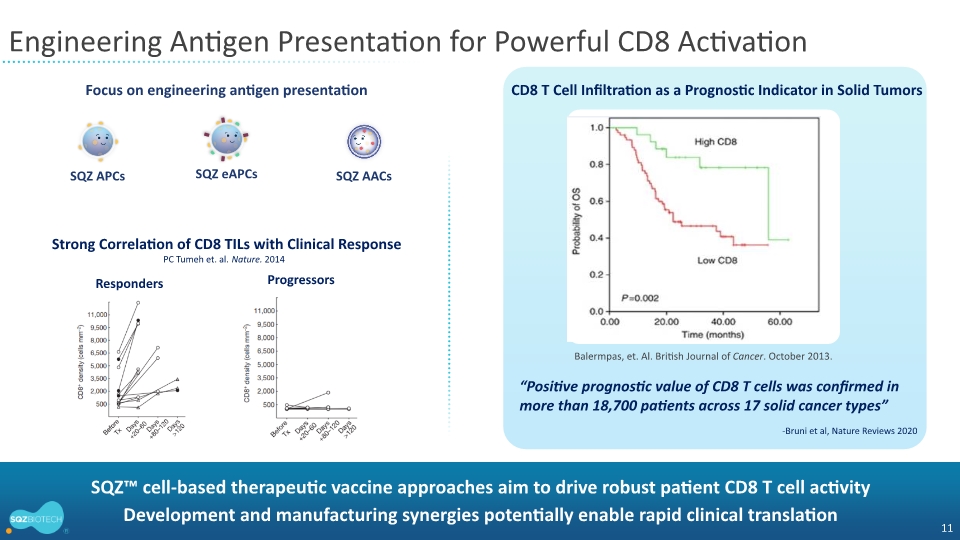
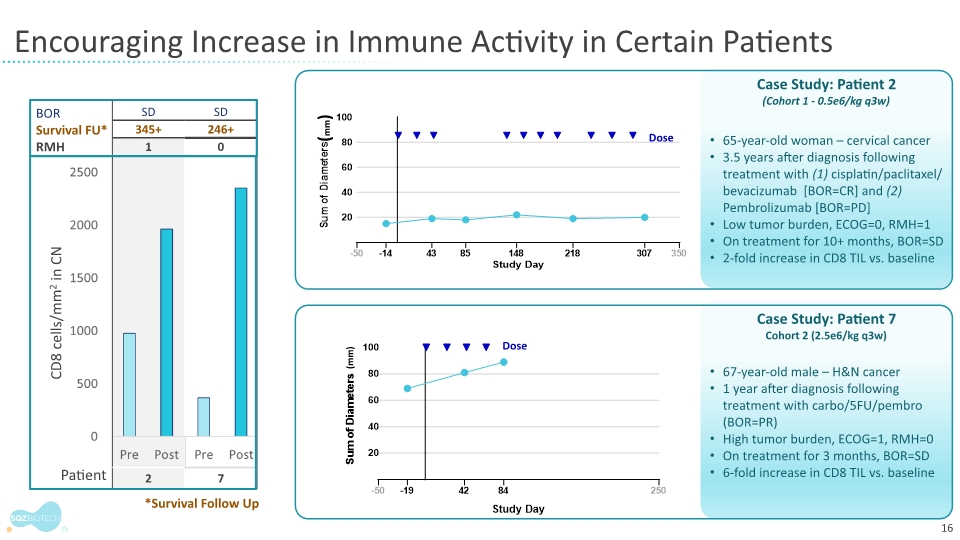
Despite the treatment refractory status of the enrolled patients, 4 out of 6 patients with RMH scores less than 2, reflecting less advanced disease, achieved stable disease as best overall response. Two of these patients showed an increase in CD8 tumor infiltrating lymphocytes (TILs), an important biomarker in immune-oncology therapy development.
The study authors highlighted two patients – Patients 2 and 7 detailed below – which suggested that less advanced patients with lower tumor burden, such as patient two, might have a higher likelihood of clinical benefit.
|
|
• |
Patient 2: Enrolled 3-and-half years after diagnosis and had a best overall response of progressive disease with ICI therapy. The patient had an RMH score of 1 and low tumor burden. She achieved stable disease while on the SQZ-PBMC-HPV-101 trial and remained on study for over 10 months. Image analysis of the central tumor 28 days after the first dose showed a 2-fold increase in CD8 TILs on treatment compared to baseline |
|
|
• |
Patient 7: Enrolled 1 year after diagnosis and had a partial response with chemotherapy in combination with ICI therapy but then progressed. He achieved stable disease after treatment on the SQZ-PBMC-HPV-101 trial and remained on study for three months. Image analysis of the central tumor showed a 6-fold increase in CD8 TILs on treatment compared to baseline |
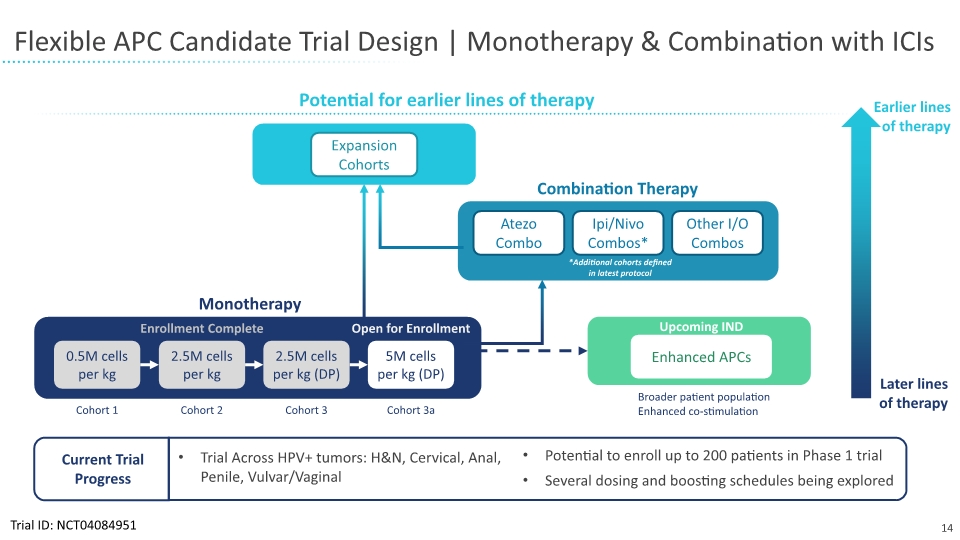
The company is now actively enrolling patients in the last monotherapy highest-dose cohort of the Phase 1 trial. These results will inform the dosage approach for the combination therapy phase of the clinical trial with immune checkpoint inhibitors.
Poster Presentation Details
Title: Initial Results of a first-in-human, dose escalation study of a cell-based vaccine in HLA-A* 02+ patients with recurrent, locally advanced or metastatic HPV16+ solid tumors
First Author: Antonio Jimeno, M.D., Ph.D., University of Colorado Cancer Center
Abstract Number: 2536
Poster Session: Developmental Therapeutics -- Immunotherapy
Date and Time: A copy of the poster is available on-demand via the ASCO virtual meeting website.
SQZ-PBMC-HPV-101 Trial Design
SQZ-PBMC-HPV is being evaluated in a Phase 1 clinical trial for the treatment of HPV16+ advanced or metastatic solid tumors. Patients must be positive for the human leukocyte antigen serotype HLA-A*02. The investigational candidate, which targets E6 and E7 oncoproteins, is being studied as a monotherapy and in combination with immuno-oncology agents. The study’s primary outcome measures in the monotherapy and combination phases of the trial include safety and tolerability. Antitumor activity is a secondary outcome measure in both the monotherapy and combination stages of the trial, and manufacturing feasibility is a secondary outcome measure in the monotherapy phase of the trial. The monotherapy phase of the study includes escalating dose cohorts with a DLT window of 28 days and the definition of a recommended phase 2 dose. The planned safety combination phase of the study will include SQZ-PBMC-HPV and checkpoint inhibitors that have previously received regulatory approval. DLT will be measured over 42 days in the safety combination phase.
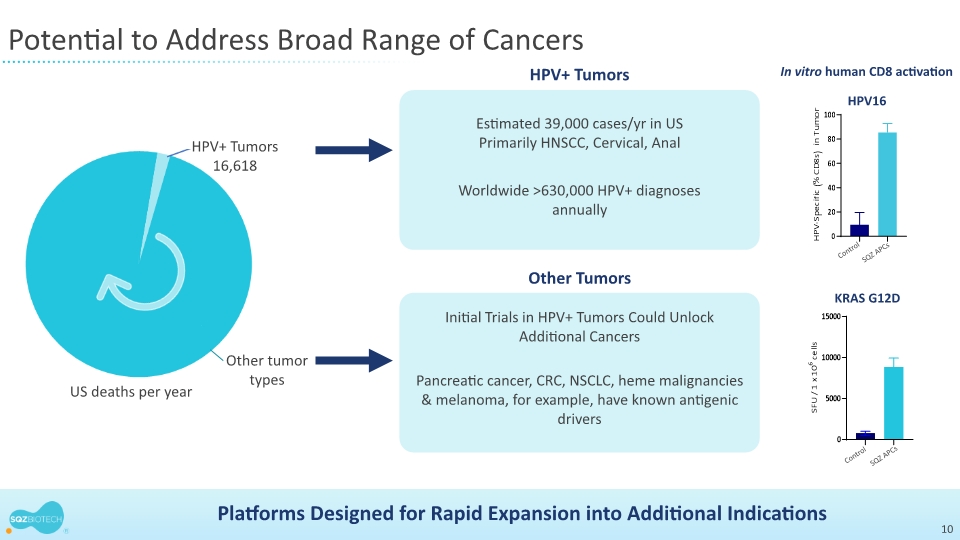
About Human Papillomavirus Positive Cancers
Human papillomavirus (HPV) is one of the most common viruses worldwide and certain strains persist for many years leading to cancer. According to the Centers for Disease Control (CDC), in the United States HPV+ tumors represent 3% of all cancers in women and 2% of all cancers in men, resulting in over 39,000 new cases of HPV+ tumors every year. HPV infection is larger outside of the U.S., and according to the International Journal of Cancer HPV+ tumors account for 4.5% of all cancers worldwide, resulting in approximately 630,000 new cases every year. According to the CDC, HPV infection plays a significant role in the formation of more than 90% of anal and cervical cancers, and most cases of vaginal (75%), oropharyngeal (70%), vulval (70%) and penile (60%) cancers.
About SQZ Biotechnologies
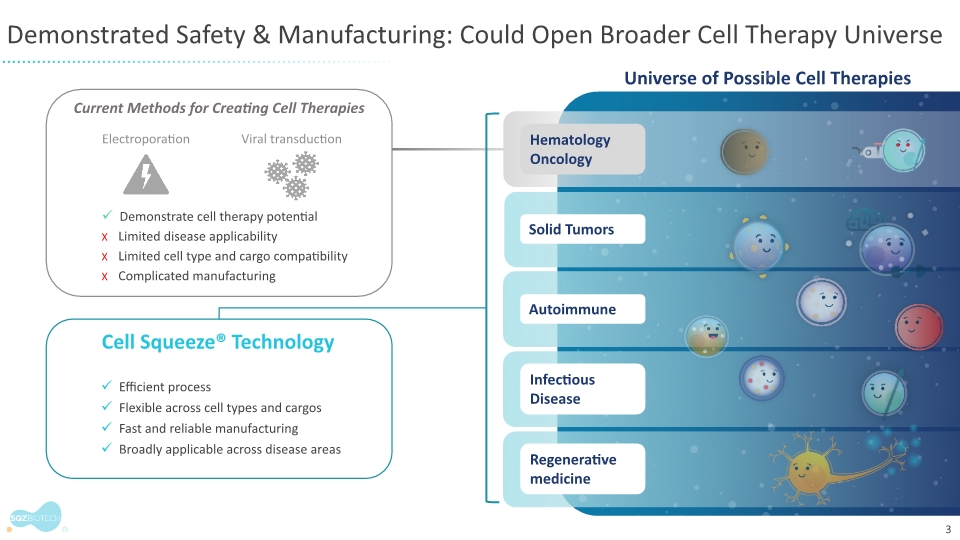
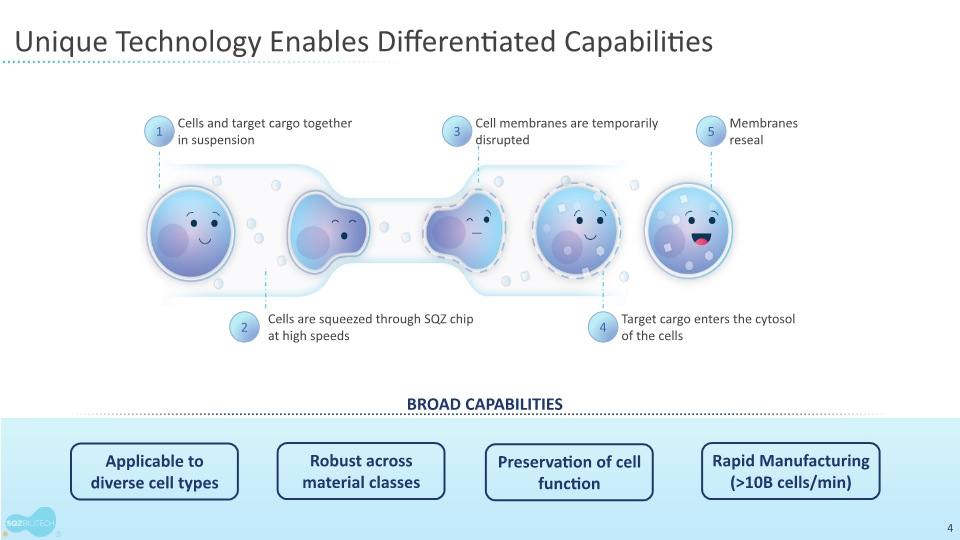
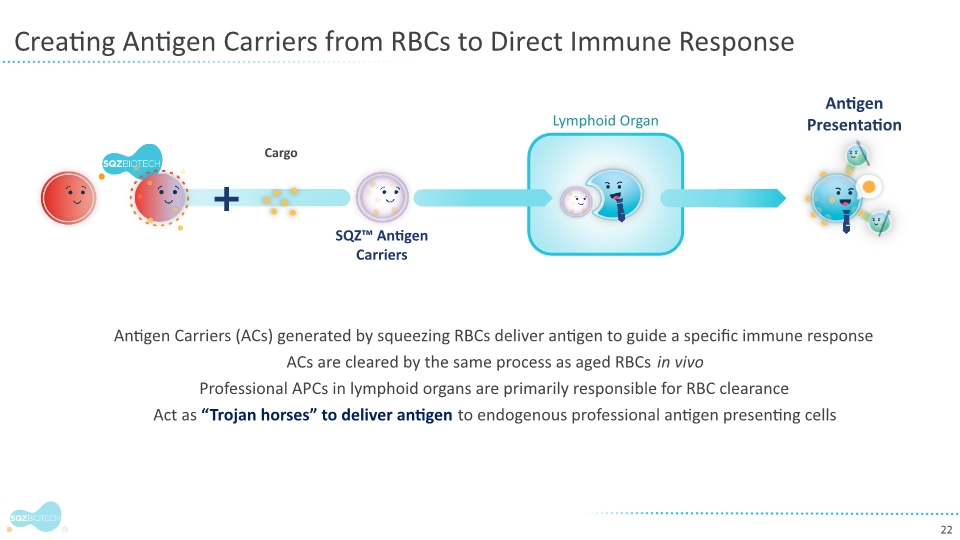
SQZ Biotechnologies is a clinical-stage biotechnology company focused on unlocking the full potential of cell therapies to benefit patients with cancer, autoimmune and infectious diseases. The company’s proprietary Cell Squeeze® technology offers the unique ability to deliver multiple biological materials into many patient cell types to engineer what we believe can be a broad range of potential therapeutics. Our goal is to create well-tolerated cell therapies that can provide therapeutic benefit for patients and improve the patient experience over existing cell therapy approaches. With accelerated production timelines under 24 hours and the opportunity to eliminate preconditioning and lengthy hospital stays, our approach could change the way people think about cell therapies. The company’s first therapeutic applications seek to generate target-specific immune responses, both in activation for the treatment of solid tumors and in immune tolerance for the treatment of unwanted immune reactions and autoimmune diseases. For more information, please visit www.sqzbiotech.com.
Forward Looking Statement
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements contained that do not relate to matters of historical fact should be considered forward-looking statements, including without limitation statements relating to events and presentations, our product candidates, preclinical and clinical activities, development plans, clinical safety and efficacy, regulatory compliance, and therapeutic impact. These forward-looking statements are based on management's current expectations. Actual results could differ from those projected in any forward-looking statements due to several risk factors. Such factors include, among others, risks and uncertainties related to our limited operating history; our significant losses incurred since inception and expectation to incur significant additional losses for the foreseeable future; the development of our initial product candidates, upon which our business is highly dependent; the impact of the COVID-19 pandemic on our operations and clinical activities; our need for additional funding and our cash runway; the lengthy, expensive, and uncertain process of clinical drug development, including uncertain outcomes of clinical trials and potential delays in regulatory approval; our ability to maintain our relationships with our third party vendors; and protection of our proprietary technology, intellectual property portfolio and the confidentiality of our trade secrets. These and other important factors discussed under the caption "Risk Factors" in our Annual Report on Form 10-K and other filings with the U.S. Securities and Exchange Commission could cause actual results to differ materially from those indicated by the forward-looking statements. Any forward-looking statements represent management's estimates as of this date and SQZ undertakes no duty to update these forward-looking statements, whether as a result of new information, the occurrence of current events, or otherwise, unless required by law.
Certain information contained in this press release relates to or is based on studies, publications, surveys and other data obtained from third-party sources and our own internal estimates and research. While we believe these third-party sources to be reliable as of the date of this press release, we have not independently verified, and we
make no representation as to the adequacy, fairness, accuracy, or completeness of any information obtained from third-party sources.
SQZ Biotechnologies IR Contact:
investors@sqzbiotech.com
SQZ Biotechnologies Media Contact:
John Lacey
Corporate Communications
john.lacey@sqzbiotech.com
781-392-5514