Exhibit 99.1
|
|

|
SQZ Biotechnologies Announces First Autoimmune Disease Indication
for Tolerizing Antigen Carrier (TAC) Platform
IND filing for Celiac Disease Anticipated in Third Quarter 2022
Clinical Translation Supported by Existing Red Blood Cell-Based Manufacturing
Capabilities and Preclinical Models Demonstrating Treg Mediated Tolerance
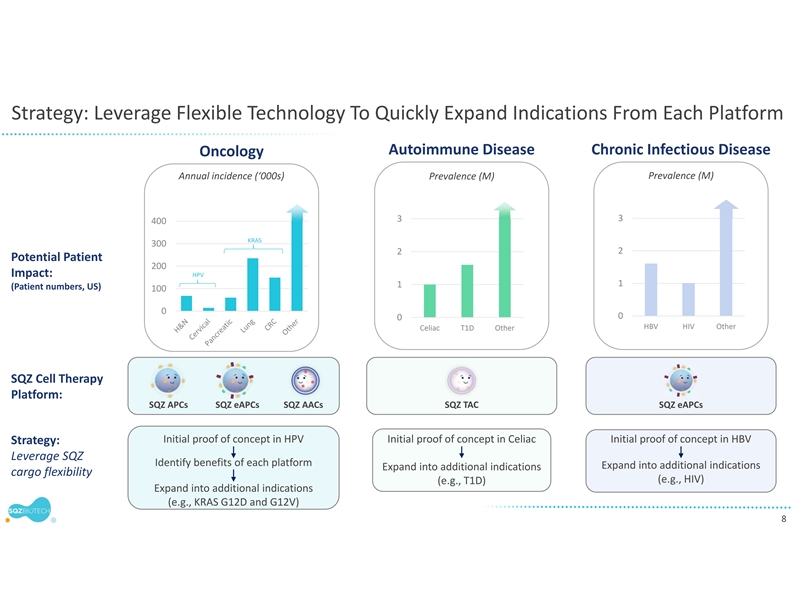
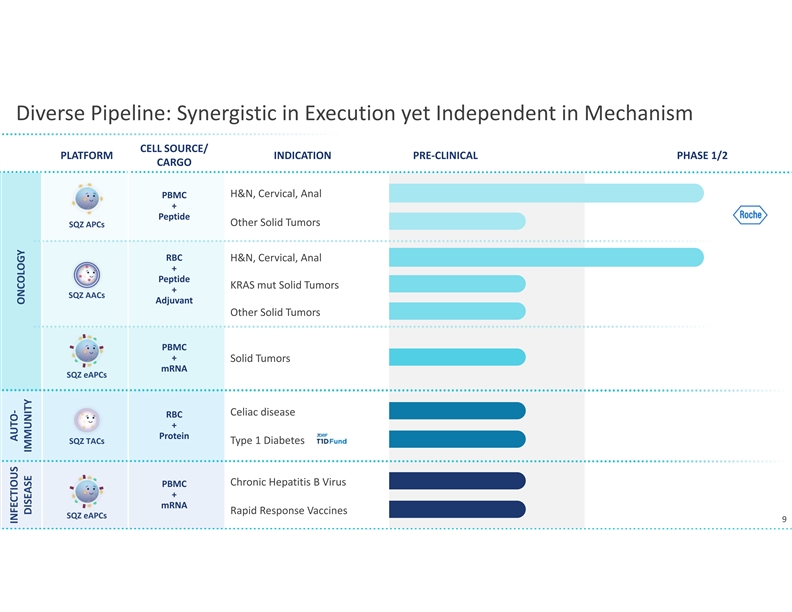
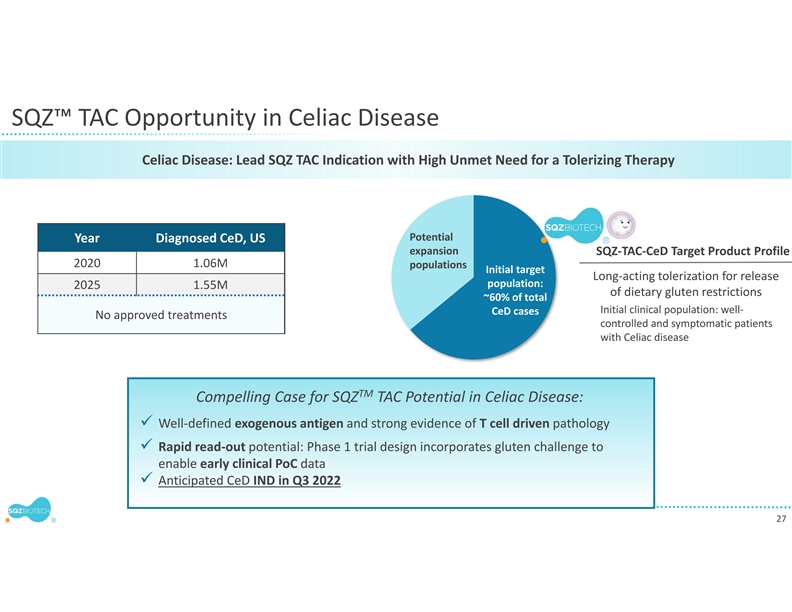
WATERTOWN, Mass., September 13, 2021 – SQZ Biotechnologies (NYSE: SQZ), focused on unlocking the full potential of cell therapies for multiple therapeutic areas, today announced that the first clinical translation of the company’s Tolerizing Antigen Carrier (TAC) platform in autoimmune diseases will be for celiac disease, a chronic autoimmune disorder that afflicts millions of patients and has no approved drug treatment. The company anticipates an IND filing in the third quarter of 2022. In preclinical models, SQZ™ TACs have demonstrated the ability to induce multi-mechanism antigen-specific tolerance relevant to many immune mediated disorders. The company’s celiac disease program may support expansion into additional autoimmune diseases.
“There is significant unmet need for patients with celiac disease. Its acute symptoms, potential for long-term complications, and necessity for a strict gluten free diet can all be extremely difficult,” said Armon Sharei, Ph.D., Chief Executive Officer at SQZ Biotechnologies. “We are excited to apply our TAC platform’s broad potential in immune tolerance, coupled with our existing rapid red blood cell-based manufacturing capabilities, to create a therapeutic that could provide a meaningful benefit to patients with celiac disease. In addition to our existing oncology clinical trials, implementation of our first immune tolerance clinical program will be an important step towards expanding our potential to impact patient lives.”
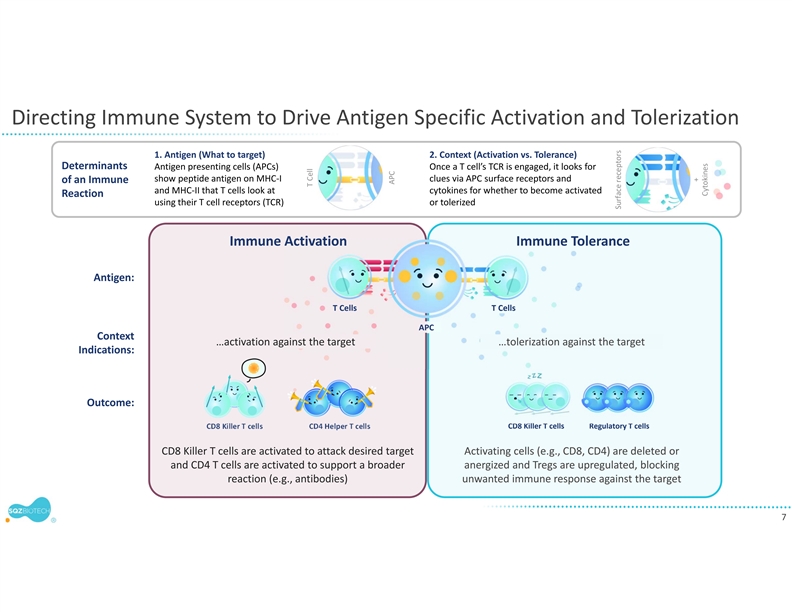
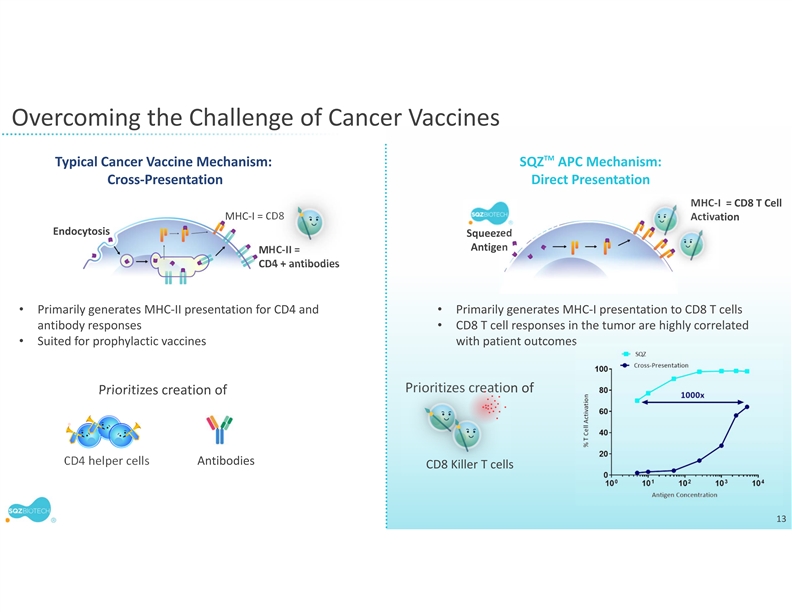
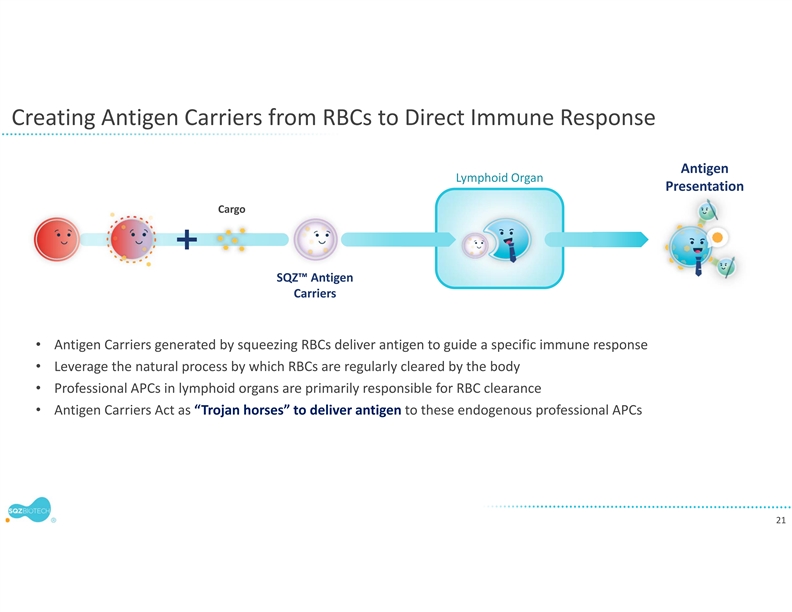
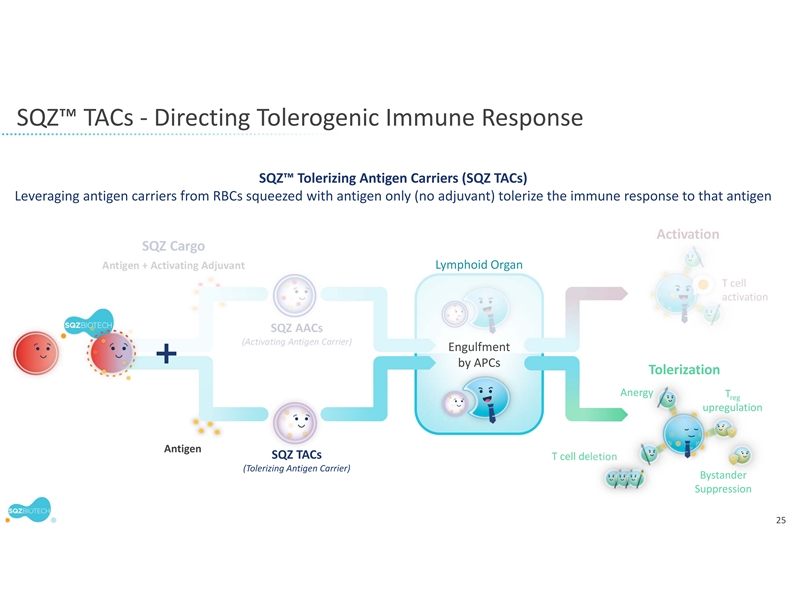
SQZ TACs leverage the body’s natural mechanisms of red blood cell (RBC) clearance and antigen presentation to induce immune tolerance. TACs are derived from patient RBCs and are designed to carry disease-specific antigen cargos to specialized immune cells that are capable of inducing tolerance through multiple mechanisms.
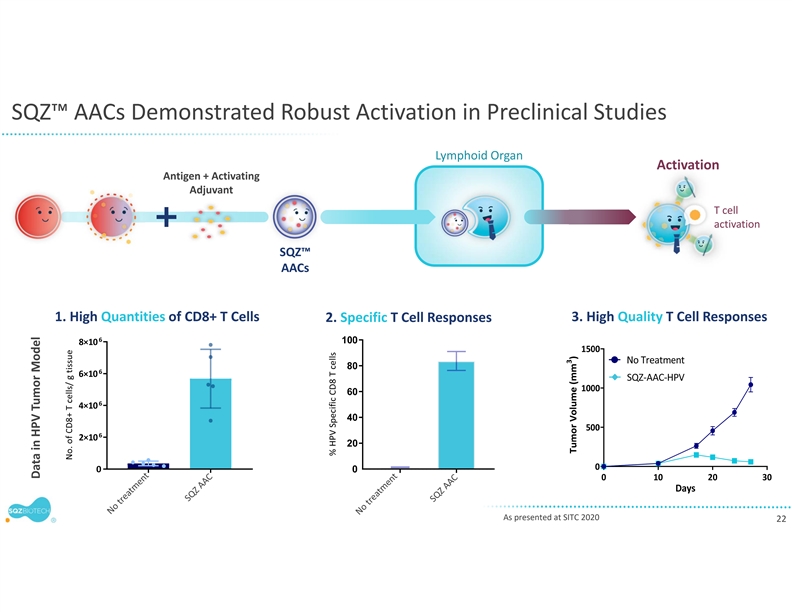
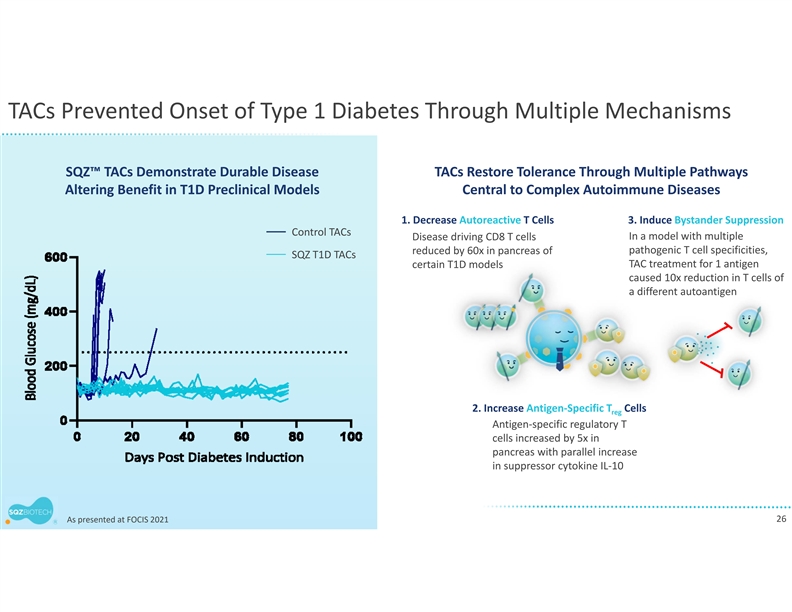
Earlier this year, the company presented preclinical findings at the Antigen-Specific Immune Tolerance Digital Summit and the Federation of Clinical Immunology Societies demonstrating that its engineered TACs can drive antigen-specific immune tolerance in complex models of autoimmune disease. SQZ TACs were shown to delete antigen-specific T cells, without causing broad immune suppression. Importantly, SQZ TACs also increased antigen-specific regulatory T cells (Tregs) that exerted potent bystander suppression, showing the ability to suppress pathogenic T cells with different autoantigen specificities.
“Our preclinical research has found that SQZ TACs can leverage physiological processes to induce T cell tolerance to disease-driving antigens through multiple
|
|

|
mechanisms,” said Howard Bernstein, M.D., Ph.D., Chief Scientific Officer at SQZ Biotechnologies. “We look forward to advancing this promising immune tolerance platform into the clinic for the treatment of celiac disease while in parallel continuing preclinical development for additional autoimmune indications, such as Type 1 diabetes.”
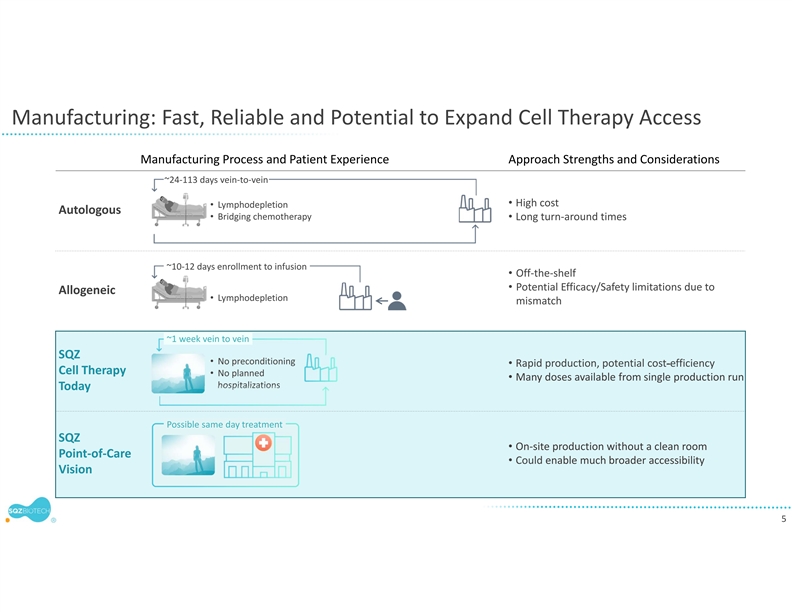
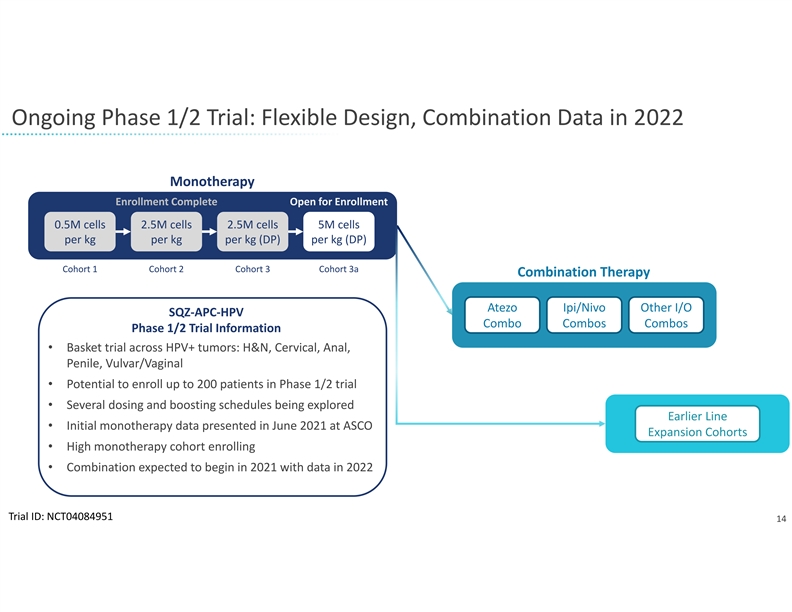
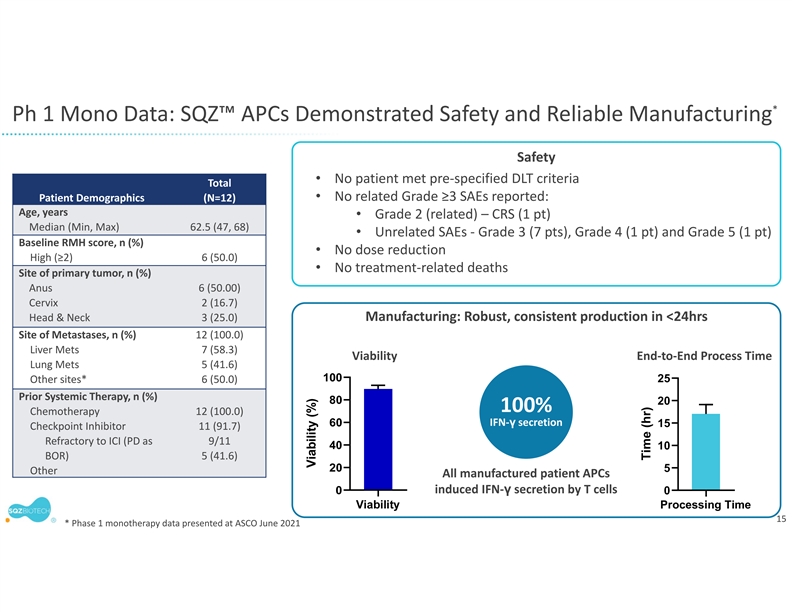
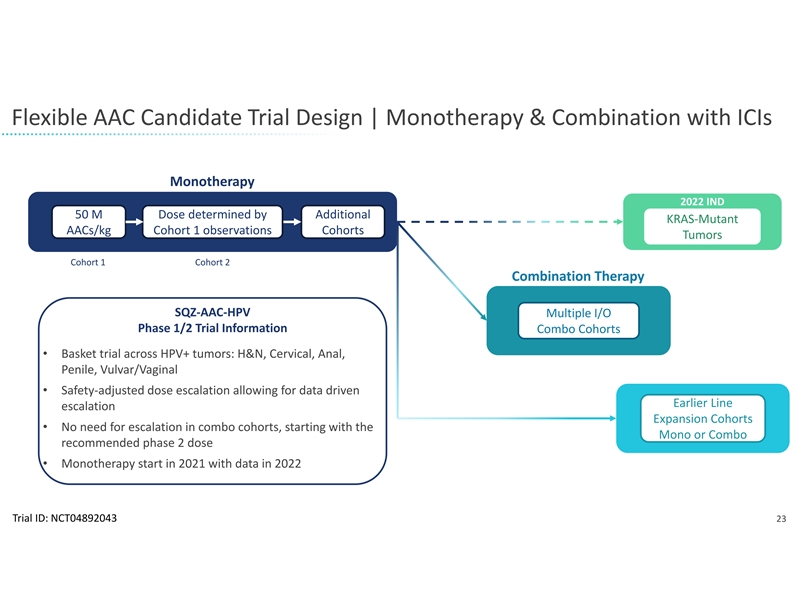
The company will leverage its rapid manufacturing system for the development of SQZ TACs. Earlier this year, the company presented clinical trial findings at the American Society of Clinical Oncology demonstrating the clinical feasibility of its manufacturing system. In the Phase 1 trial of the company’s APC therapeutic candidate for HPV positive tumors, patient batches of the investigational cell therapy were produced in less than 24 hours. The flexibility and scalability of the company’s manufacturing system allows it to be used for both the TAC and Oncology platforms despite different cell types and antigen cargos. SQZ TAC therapeutic candidates are also expected to be compatible with potential future point of care implementations.
About Celiac Disease
Celiac disease is a chronic autoimmune disorder that occurs in genetically predisposed people.1 2 The disease is triggered by eating foods containing gluten, which is found in wheat, barley, and rye. Disease symptoms can include abdominal pain, diarrhea, nausea, vomiting, and other common signs. When gluten is ingested the body mounts an immune response that attacks and damages the villi that line the small intestine, which can impact nutrient absorption.3 Many people who have celiac disease have not been diagnosed,4 however population-based studies indicate that the disease affects about 2 million people in the United States and approximately 1% of the population worldwide, with regional differences.5 6 There is currently no approved drug treatment and patients must therefore maintain a gluten-free diet, which involves strict, lifelong avoidance of exposure to gluten proteins. Long-term complications of celiac disease may include malnutrition, accelerated osteoporosis, nervous system problems and issues related to reproduction. Rare complications can include cancer of the small intestine, cirrhosis, and non-Hodgkin lymphoma.
About SQZ TACs
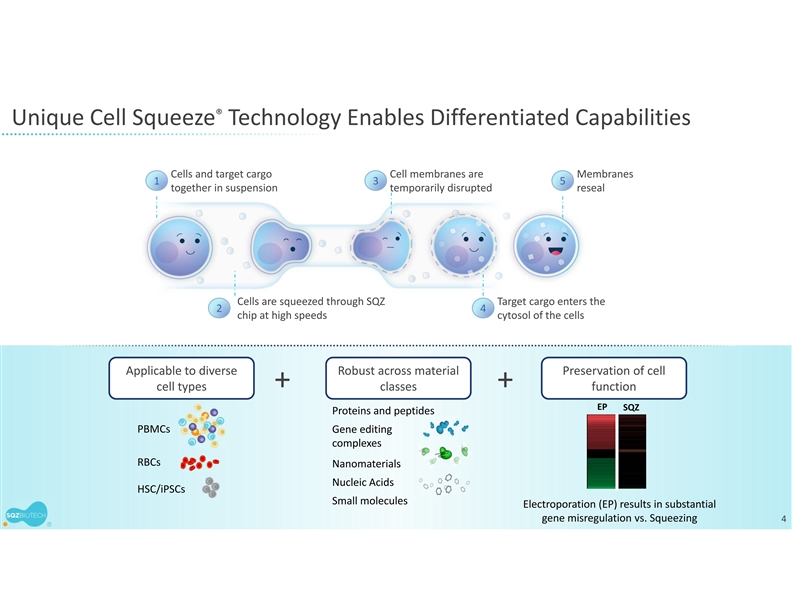
SQZ TACs are a red blood cell-derived cell therapy platform being developed as an antigen-specific immune tolerance approach for autoimmune diseases. The platform is designed to leverage the natural process of RBC clearance by professional antigen presenting cells (APCs) in the lymphoid organs. This physiological mechanism is tolerogenic by default, instructing the immune system not to mount an attack. SQZ TACs are generated by squeezing RBCs with disease-specific antigens and are made to appear aged. SQZ TACs are designed to be rapidly engulfed by professional APCs and to act as a “Trojan horse” to present antigens in a non-inflammatory context, inducing tolerization of the patient’s T cells against the specific target. Preclinical data has demonstrated deletion and anergy of antigen-specific T cells as well as induction of disease specific Tregs capable of bystander suppression.
|
|

|
About SQZ Biotechnologies
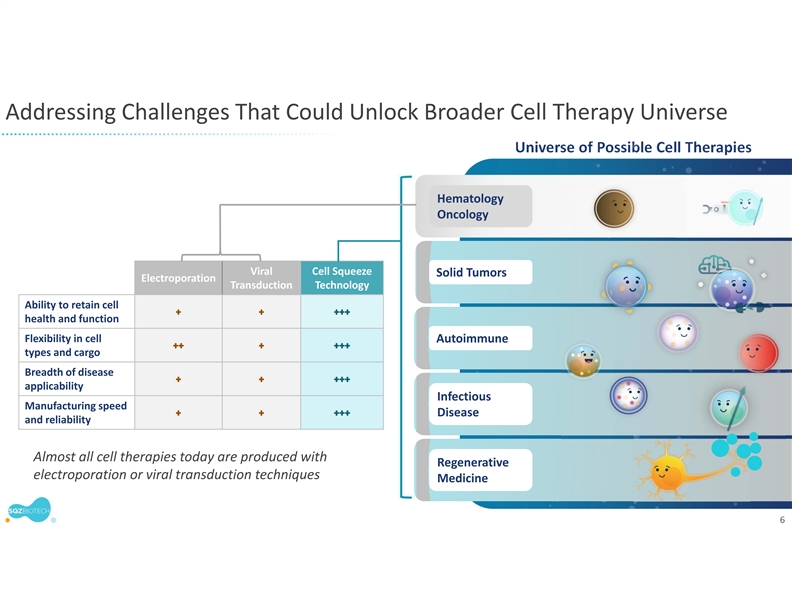
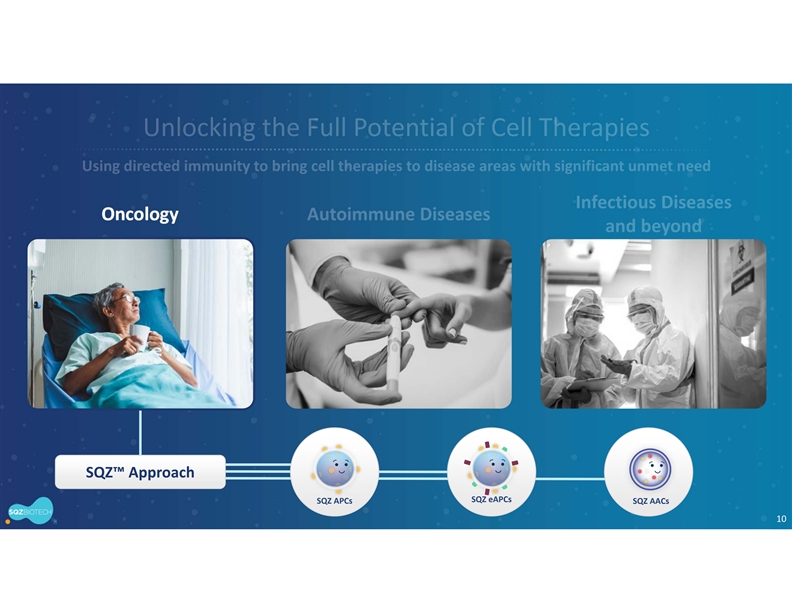
SQZ Biotechnologies Company is a clinical-stage biotechnology company focused on unlocking the full potential of cell therapies for patients around the world and has active programs in Oncology, Autoimmune and Infectious Diseases, as well as additional exploratory initiatives to support future pipeline growth. The company’s proprietary Cell Squeeze® technology offers the unique ability to deliver multiple biological materials into many cell types to engineer what we believe can be a broad range of potential therapeutics. With demonstrated production timelines under 24 hours and the opportunity to eliminate preconditioning and lengthy hospital stays, our approach could significantly broaden the therapeutic range and accessibility of cell therapies. The company’s first therapeutic applications seek to generate target-specific immune responses, both in activation for the treatment of solid tumors and infectious diseases, and in immune tolerance for the treatment of unwanted immune reactions and autoimmune diseases. For more information, please visit www.sqzbiotech.com.
Forward Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements contained that do not relate to matters of historical fact should be considered forward-looking statements, including without limitation statements relating to our platform development, manufacturing capabilities, product candidates, preclinical and clinical activities and outcomes, development plans and progress, clinical efficacy, regulatory submissions, therapeutic impact and market opportunities. These forward-looking statements are based on management’s current expectations. Actual results could differ from those projected in any forward-looking statements due to several risk factors. Such factors include, among others, risks and uncertainties related to our limited operating history; our significant losses incurred since inception and expectation to incur significant additional losses for the foreseeable future; the development of our initial product candidates, upon which our business is highly dependent; the impact of the COVID-19 pandemic on our operations and clinical activities; our need for additional funding and our cash runway; the lengthy, expensive, and uncertain process of clinical drug development, including uncertain outcomes of clinical trials and potential delays in regulatory approval; our ability to maintain our relationships with our third party vendors; and protection of our proprietary technology, intellectual property portfolio and the confidentiality of our trade secrets. These and other important factors discussed under the caption “Risk Factors” in our Annual Report on Form 10-K and other filings with the U.S. Securities and Exchange Commission could cause actual results to differ materially from those indicated by the forward-looking statements. Any forward-looking statements represent management’s estimates as of this date and SQZ undertakes no duty to update these forward-looking statements, whether as a result of new information, the occurrence of current events, or otherwise, unless required by law.
Certain information contained in this press release relates to or is based on studies, publications, surveys and other data obtained from third-party sources and our own internal estimates and research. While we believe these third-party sources to be reliable as of the date of this press release, we have not independently verified, and we make no representation as to the adequacy, fairness, accuracy or completeness of any information obtained from third-party sources.
|
|

|
SQZ Biotechnologies IR Contact:
investors@sqzbiotech.com
SQZ Biotechnologies Media Contact:
John Lacey
Corporate Communications
john.lacey@sqzbiotech.com
781-392-5514
| 1 | Leonard MM, Sapone A, Catassi C, et al. Celiac Disease and Nonceliac Gluten Sensitivity: A Review. JAMA 2017;318:647-656 |
| 2 | Celiac Disease Foundation website (as of Sept 13,2021) |
| 3 | Beyond Celiac website (as of Sept 13, 2021) |
| 4 | National Institute of Diabetes and Digestive and Kidney Diseases website (as of Sept 13, 2021) |
| 5 | Lionetti E, Gatti S, Pulvirenti A, et al. Celiac disease from a global perspective. Best Pract Res Clin Gastroenterol 2015;29:365-79. |
| 6 | Leonard MM, Sapone A, Catassi C, et al. Celiac Disease and Nonceliac Gluten Sensitivity: A Review. JAMA 2017;318:647-656 |