Exhibit 99.1

Recro Pharma Reports Positive Top-Line Results from Pivotal Phase III Clinical Trial of IV Meloxicam
IV Meloxicam Achieves Primary Endpoint of Statistically Significant Reduction in SPID24 in Patients with Acute Postoperative Pain Following Abdominoplasty Surgery
Ten Secondary Endpoints Also Met
Company Estimates Filing US NDA in Summer 2017
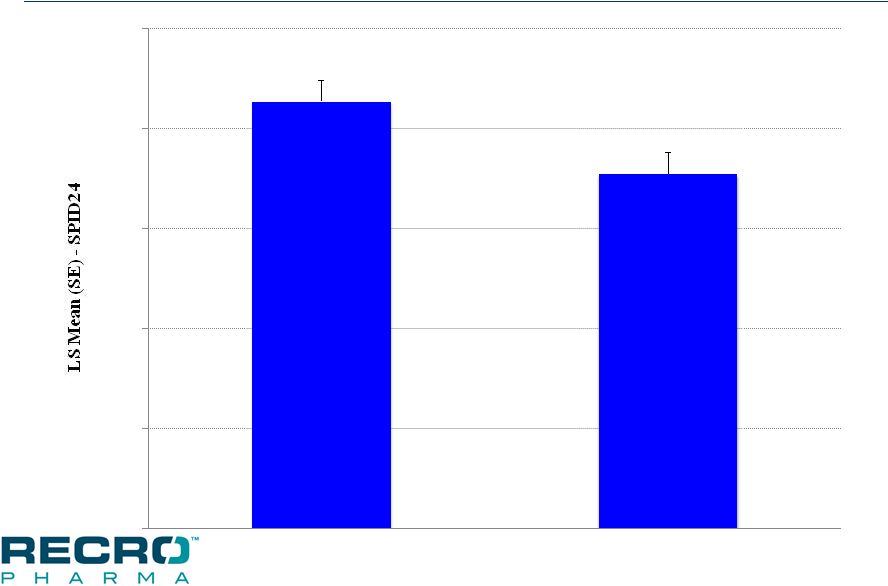
MALVERN, PA, November 28, 2016 – Recro Pharma, Inc. (Nasdaq:REPH), a revenue generating specialty pharmaceutical company focused on products for hospital and ambulatory care settings, currently developing non-opioid products for the treatment of serious acute pain, today announced positive results from its second of two Phase III clinical trials evaluating intravenous (IV) meloxicam for the treatment of acute postoperative pain. In this trial, IV meloxicam achieved the primary endpoint of a statistically significant difference in Summed Pain Intensity Difference (SPID) over the first 24 hours (SPID24), compared to placebo, in patients following abdominoplasty surgery. With the positive data from this study, the Company believes this completes the efficacy program for the IV meloxicam New Drug Application (NDA).
In this multicenter, randomized, double-blind, placebo-controlled clinical trial, 219 patients were enrolled and randomly assigned to receive a postoperative regimen of IV meloxicam (30mg bolus injection) or placebo in a 1:1 ratio, once every 24 hours. The IV meloxicam treatment arm demonstrated a statistically significant reduction in SPID24 (p=0.0145) compared to the placebo arm. The study also achieved statistical significance for 10 of the secondary endpoints, including statistically significant differences in SPID12 (p=0.0434), time to perceptible pain relief (p=0.0050), subjects with ³30% improvement at 24 hours (p=0.0178), number of times patients required rescue in the first 24 hours after randomization (p=0.0275), as well as number of times rescued from 24 to 48 hours (p=0.0009), and several other pain relief metrics, compared to placebo.
The safety results demonstrated that IV meloxicam was well tolerated with no difference in serious adverse events (SAEs) related to bleeding for IV meloxicam treated patients versus placebo (1 each). There were two additional SAEs observed in the placebo group. The most common (³2% in the IV meloxicam group) adverse events (AEs) were nausea, headache, vomiting, and dizziness. The incidence of these events was lower than those observed in the placebo group. The majority of AEs were mild in nature and one patient in the placebo group discontinued treatment due to an adverse event of post-procedural bleeding.
1

There were no meaningful differences between treatment groups in vital signs, ECGs or clinical lab assessments.
“The data from this trial demonstrated that IV meloxicam achieved a statistically significant difference in SPID24 pain relief following abdominoplasty surgery, a favorable safety and tolerability profile, and impressive impact on the number of times patients required rescue throughout the 0-24 and 24-48 hour periods, as well as the percent of subjects with ³30% improvement at 24 hours,” said Neil Singla, M.D., Chief Scientific Officer of Lotus Clinical Research. “These data are important because they show that, if approved, IV meloxicam has the potential to be a new, non-opioid alternative for patients with moderate-to-severe pain following soft tissue surgery.”
“The positive outcome from this second pivotal trial continues to demonstrate the efficacy of IV meloxicam in the acute postoperative setting, while reinforcing the favorable efficacy and safety profile observed in five prior studies,” said Gerri Henwood, Recro Pharma’s President and Chief Executive Officer. “Given the urgent need for non-opioid pain relief alternatives, we believe the data from this trial, together with the positive data from our previously reported pivotal Phase III trial in post-surgical bunionectomy patients, completes the efficacy platform for an NDA for IV meloxicam as a new, non-opioid analgesic option for acute moderate-to-severe postoperative pain. Enrollment in the remaining, ongoing safety study is expected to be complete by the end of the first quarter or early second quarter 2017, with an NDA filing expected to follow in summer 2017.”
The secondary endpoints of: SPID6, time to first rescue, number of subjects rescued 0-24 hours, number of subjects rescued 0-48 hours, time to meaningful pain relief, percent of subjects ³30% improved at 6 hours, percent of subjects ³50% improved at 6 or 24 hours, and Patient Global Assessment of pain control at 24 hours were not significantly different between treatment groups.
Recro plans to submit additional data from this Phase III clinical trial for presentation at a future scientific conference or in a journal publication.
About Abdominoplasty
Abdominoplasty surgery generally involves the removal of excess fat and skin and, in most cases, restores weakened or separated muscles from the abdominal area. According to the American Society for Aesthetic Plastic Surgery, abdominoplasty is among the top five most common cosmetic surgeries in the U.S., with more than 164,000 performed in 2014. Abdominoplasty surgery typically results in intense postoperative pain.
2

About IV/IM Meloxicam
Meloxicam is a long-acting, preferential COX-2 inhibitor that possesses anti-inflammatory, analgesic, and antipyretic activities, which are believed to be related to the inhibition of cyclooxygenase (COX) and subsequent reduction in prostaglandin biosynthesis. Meloxicam has been marketed by Boehringer Ingelheim Pharmaceuticals, Inc. since the 1990’s as an oral agent, Mobic®. IV/IM meloxicam was designed using NanoCrystal® platform, a technology that enables enhanced bioavailability of poorly water-soluble drug compounds. Recro acquired IV/IM meloxicam from Alkermes in April 2015.
About Recro Pharma, Inc.
Recro Pharma is a revenue generating specialty pharmaceutical company focused on products for hospital and ambulatory care settings that is currently developing non-opioid products for the treatment of serious acute pain. Recro Pharma is currently developing IV meloxicam, a proprietary, long-acting preferential COX-2 inhibitor for treatment of acute postoperative pain, which has completed four successful Phase II clinical trials in postoperative pain conditions and has reported positive results from its two pivotal Phase III clinical trials in patients following bunionectomy surgery, and in patients following abdominoplasty surgery. An additional development candidate, Dex-IN, a proprietary intranasal formulation of dexmedetomidine, is being pursued for the treatment of peri-procedural pain, and has had a past successful Phase II trial in Bunionectomy. As Recro Pharma’s product candidates are not in the opioid class of drugs, the Company believes its candidates would avoid many of the side effects associated with commonly prescribed opioid therapeutics, such as addiction, constipation and respiratory distress, while maintaining analgesic effect.
Recro Pharma also owns and operates a 97,000 square foot, DEA-licensed facility that manufactures five commercial products and receives manufacturing revenues and royalties associated with the sales of these products.
Cautionary Statement Regarding Forward Looking Statements
Any statements in this press release about future expectations, plans and prospects for the Company, including statements about the Company’s strategy, future operations, clinical development plans and other statements containing the words “anticipate,” “believe,” “estimate,” “upcoming,” “plan,” “target”, “intend,” “expect” and similar expressions, constitute forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including: results and timing of the clinical trials of injectable meloxicam and Dex-IN; unfavorable new clinical data and additional analyses of existing clinical data; whether
3

results of early clinical trials will be indicative of the results of future trials and whether interim results from a clinical trial will be predictive of the final results of the trial; the ability to obtain and maintain regulatory approval of injectable meloxicam and Dex-IN, and the labeling under any such approval; regulatory developments in the United States and foreign countries; the Company’s ability to raise future financing for continued development; the Company’s ability to pay its debt; the performance of third-party suppliers and manufacturers; the Company’s ability to obtain, maintain and successfully enforce adequate patent and other intellectual property protection; the successful commercialization of injectable meloxicam and Dex-IN and other factors discussed in the Risk Factors set forth in the Company’s Annual Report on Form 10-K and Quarterly Reports on Form 10-Q filed with the Securities and Exchange Commission (SEC) and in other filings the Company makes with the SEC from time to time. In addition, the forward-looking statements included in this press release represent the Company’s views only as of the date of this press release. Important factors could cause our actual results to differ materially from those indicated or implied by forward-looking statements, and as such we anticipate that subsequent events and developments will cause our views to change. However, while we may elect to update these forward-looking statements at some point in the future, we specifically disclaim any obligation to do so. These forward-looking statements should not be relied upon as representing our views as of any date subsequent to the date of this press release.
CONTACT:
Media Contact:
Argot Partners
Eliza Schleifstein
(973) 361-1546
eliza@argotpartners.com
Investor Relations Contact:
Argot Partners
Susan Kim/Natalie Wildenradt
(212) 600-1902
susan@argotpartners.com
natalie@argotpartners.com
Recro Pharma, Inc.
Michael Celano
(484) 395 2413
mcelano@recropharma.com
4