Exhibit 99.1

Recro Pharma Reports Second Quarter 2016 Financial Results
Announced Positive Top-line Results from Pivotal Phase III Trial of
IV Meloxicam Following Bunionectomy Surgery
Pivotal Phase III Trial of IV Meloxicam in Abdominoplasty Ongoing with Top-line Data Expected by the End of 2016
Manufacturing Business Continues Solid Performance
MALVERN, PA, August 11, 2016 – Recro Pharma, Inc. (Nasdaq: REPH), a revenue generating specialty pharmaceutical company focused on products for hospital and ambulatory care settings, currently developing non-opioid products for the treatment of serious acute pain, today reported financial results for the second quarter ended June 30, 2016.
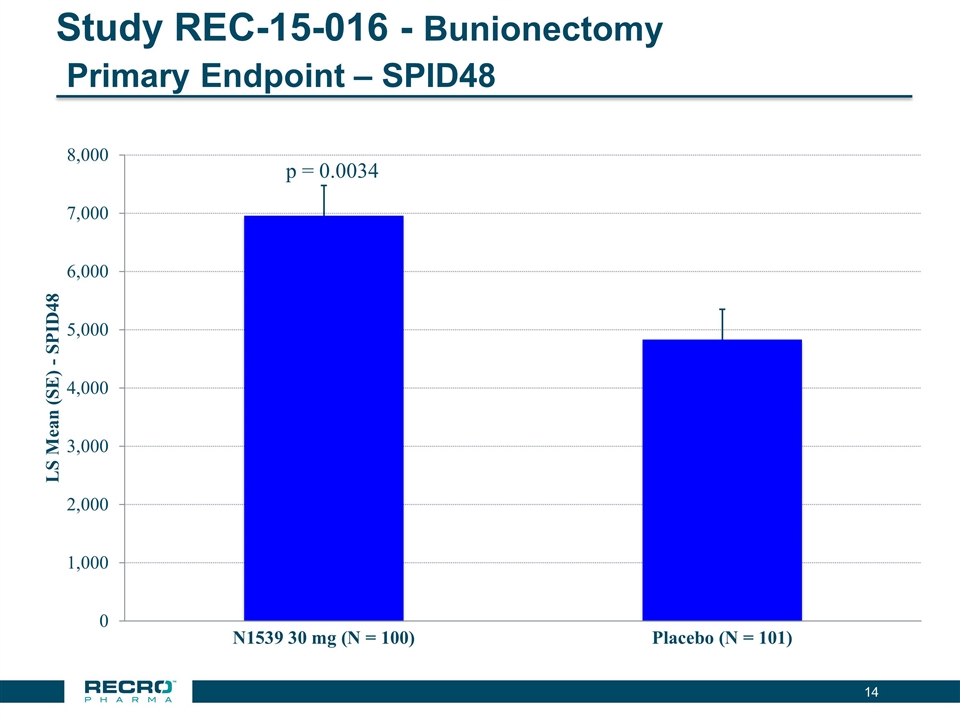
“We had a strong first half of 2016, highlighted by positive top-line results recently reported from one of our pivotal Phase III trials of intravenous (IV) meloxicam for acute postoperative pain in patients following bunionectomy surgery,” said Gerri Henwood, President and Chief Executive Officer of Recro Pharma. “In addition to achieving its primary endpoint, which was a statistically significant reduction in SPID over the first 48 hours versus placebo, IV meloxicam also achieved fifteen of nineteen secondary endpoints, suggesting potential for the broad clinical utility of this promising candidate in a post-operative setting. There exists an urgent and growing need for non-opioid alternatives to address moderate-to-severe pain, and we look forward to reporting top-line results from our second pivotal study of IV meloxicam in mini abdominoplasty surgery by the end of 2016. Meloxicam represents a significant near-term commercial opportunity for our Company, and assuming positive data from our second pivotal study, we anticipate filing an NDA mid-summer 2017.”
Second Quarter 2016 and Recent Highlights
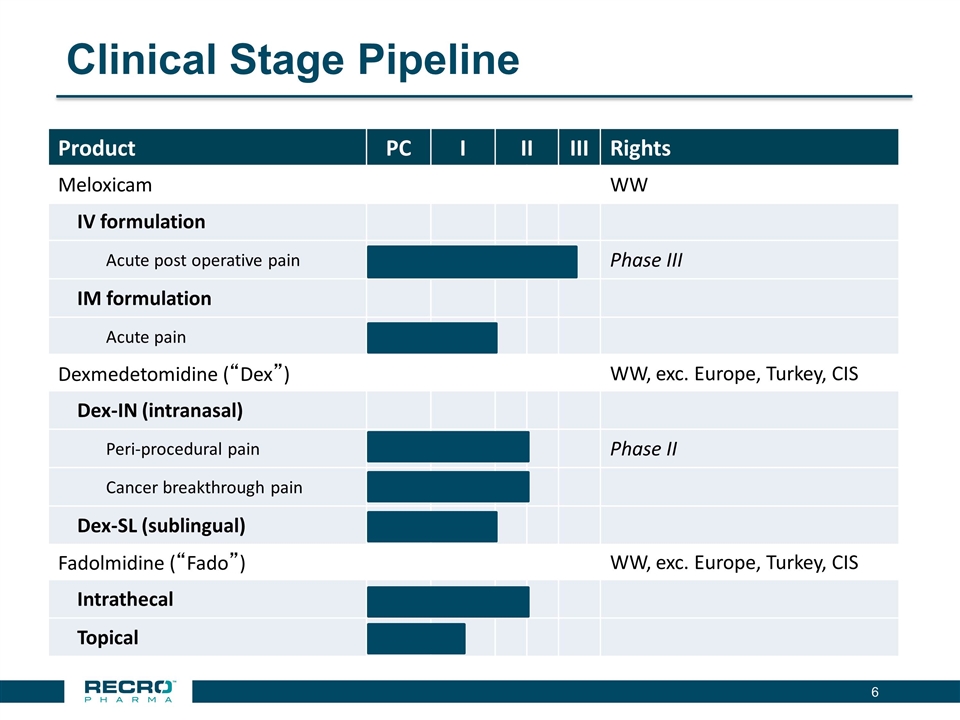
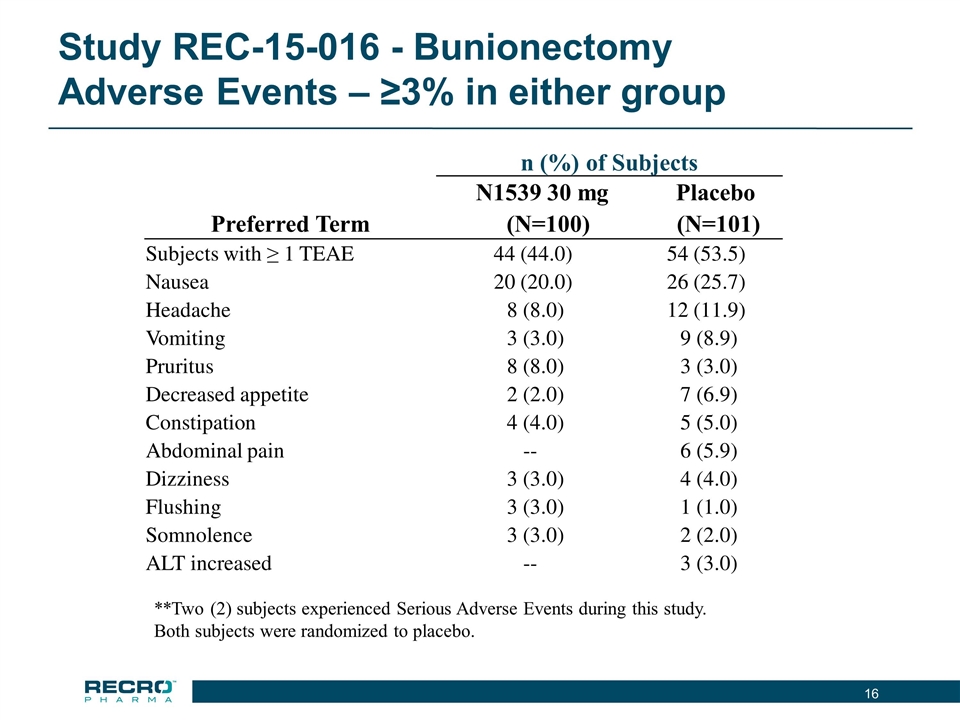
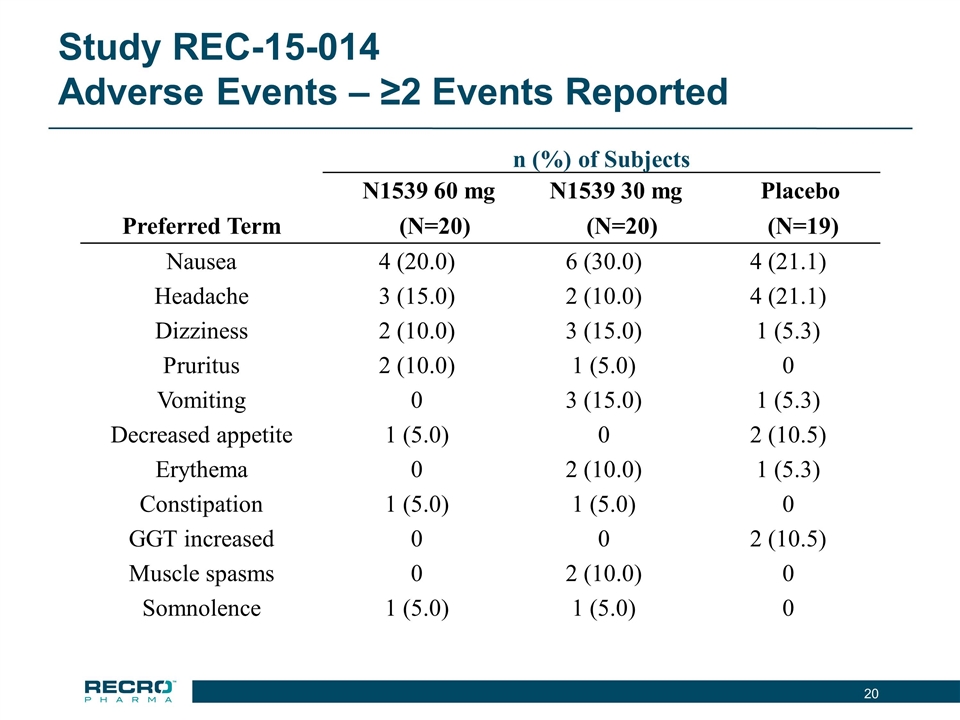
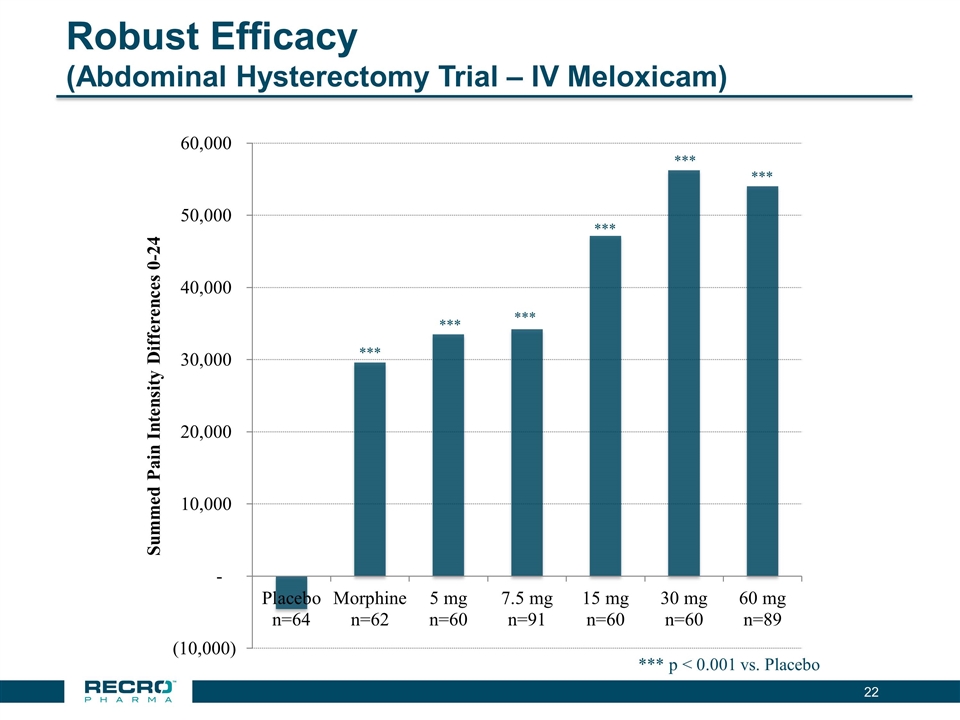
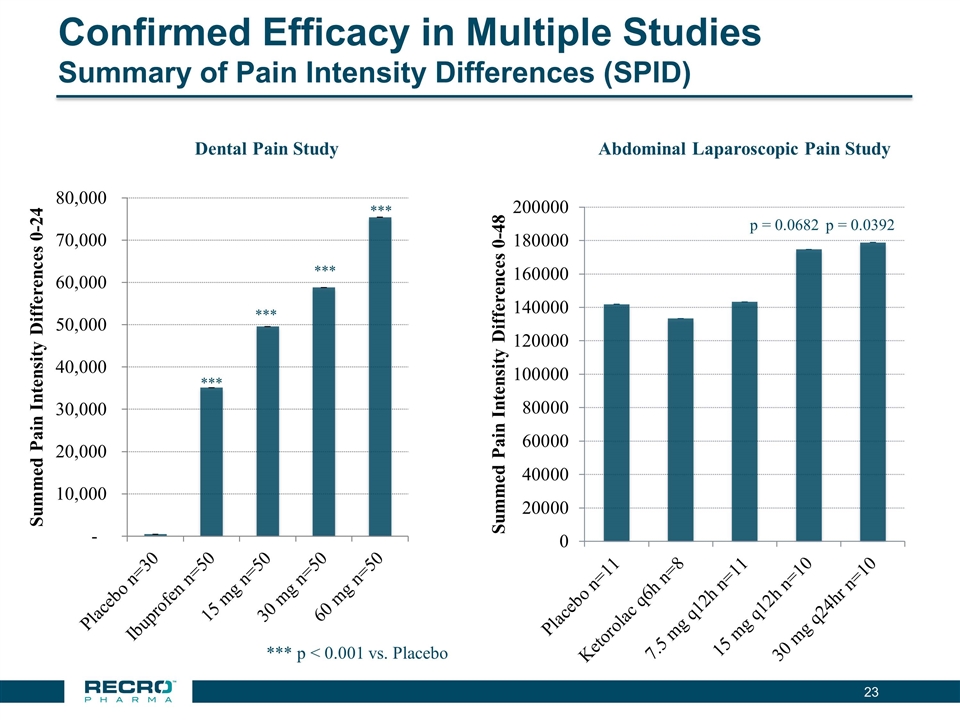
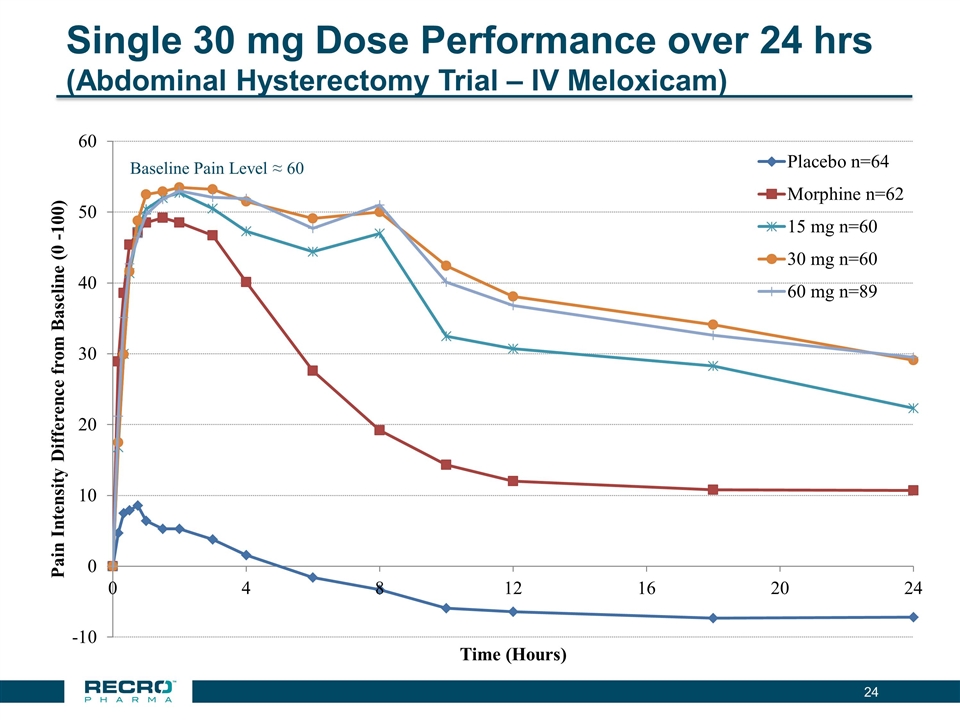
| • | Reported Positive Top-line Phase III Results from First of Two Pivotal Intravenous (IV) Meloxicam Trials. In July 2016, Recro reported positive results from its Phase III clinical trial evaluating IV meloxicam for the treatment of acute postoperative pain in patients following bunionectomy surgery. In the trial, IV meloxicam achieved the primary endpoint of a statistically significant difference in Summed Pain Intensity Difference (SPID) over the first 48 hours (SPID48) compared to placebo. The study also achieved fifteen of nineteen secondary endpoints, including statistically significant differences in SPID6, SPID12, SPID24, SPID24-48, time to first use of rescue medication, and several other rescue use and pain relief metrics during the first 48 hours, compared to placebo. |
| • | Ongoing Execution of Pivotal Phase III Trial of IV Meloxicam in Abdominoplasty. Enrollment continues in Recro’s second pivotal Phase III clinical trial evaluating IV meloxicam for acute postoperative pain in patients following mini abdominoplasty surgery. |
1

| The Company remains on track to report top-line results by the end of 2016. Assuming positive results from this second Phase III trial, the Company plans to file a New Drug Application for IV meloxicam in mid-summer 2017. |
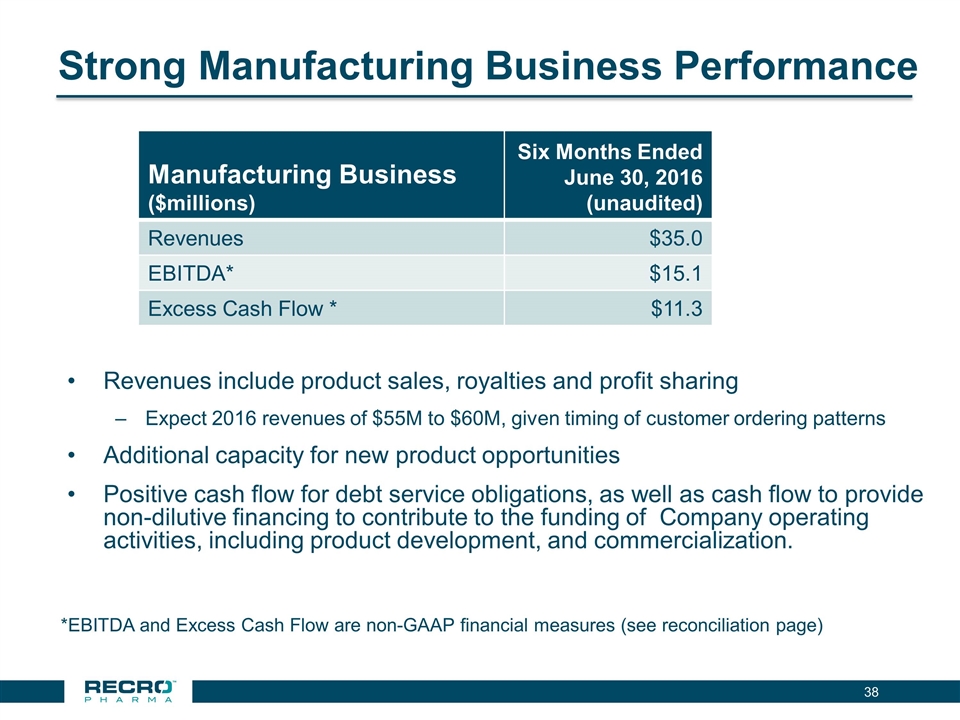
| • | Manufacturing Business Continues Solid Performance. Our manufacturing business performed well with revenues of $35 million for the six months ended June 30, 2016, generating positive cash flow for the Company. We continue to expect 2016 revenues of $55 million to $60 million, given the timing of customer ordering patterns. |
| • | Strengthened Management Team with the Appointment of Michael Celano as Chief Financial Officer. Mr. Celano brings more than 35 years of financial leadership experience in the life sciences and biopharmaceutical industries to Recro, including serving in executive roles at Kensey Nash, BioRexis, and DrugScan. |
Financial Results
As of June 30, 2016, Recro Pharma had cash and cash equivalents of $14.8 million.
Pursuant to the terms of the Company’s credit agreement with OrbiMed, OrbiMed has the option to require the Company to pay down debt with excess free cash flow generated from the Recro Gainesville manufacturing business. The Company made principal payments of approximately $2.6 million to OrbiMed during the six months ended June 30, 2016. Since its acquisition in April 2015 of the manufacturing business, the Company has paid down approximately $19.0 million, or 38%, of the original $50.0 million of senior secured term loan from the business’s excess cash flow generated. In August 2016, we will make an additional payment of $3.7 million which was 50% of the excess cash flow generated during the second quarter of 2016.
For the three months ended June 30, 2016, Recro Pharma reported a net loss applicable to common shareholders of $7.9 million, or $0.83 per share, compared to a net loss applicable to common shareholders of $1.3 million, or $0.17 per share, for the comparable period in 2015. For the six months ended June 30, 2016, Recro Pharma reported a net loss applicable to common shareholders of $14.4 million, or $1.53 per share, compared to a net loss applicable to common shareholders of $5.5 million, or $0.70 per share, for the comparable period in 2015.
Revenues for the three months ended June 30, 2016 were $17.3 million, compared to $18.7 million for the same period in 2015. The decrease in revenues is a result of the completion of research and development projects in 2015. COGS for the three months ended June 30, 2016 were $9.5 million, compared to $9.4 million for the same period in 2015.
Revenues and COGS for the six months ended June 30, 2016 were $35.0 million and $19.8 million, respectively, were higher than revenues and COGS of $18.7 million and $9.4 million, respectively, for the comparable period in 2015, as we owned the manufacturing business for three months in 2015 compared to six months in 2016.
2

Research and development expenses for the three months ended June 30, 2016 were $8.3 million, compared to $2.8 million for the same period in 2015. Research and development expenses for the six months ended June 30, 2016 were $16.1 million, compared to $4.6 million for the same period in 2015. The increases in research and development expenses were primarily due to the Company’s injectable meloxicam Phase III trials clinical expenses as well as increased research and development costs incurred at the Recro Gainesville facility, offset by a decrease in Dex clinical expenses.
General and administrative expenses for the three months ended June 30, 2016 were $2.8 million, compared to $2.6 million for the same period in 2015. General and administrative expenses for the six months ended June 30, 2016 were $5.4 million, compared to $5.0 million for the same period in 2015. The increases in general and administrative expenses were primarily due to an increase in salaries, benefits and stock compensation as a result of additional headcount.
Amortization of intangibles for the three months ended June 30, 2016 were $0.6 million, compared to $0.6 million for the same period in 2015. Amortization of intangibles for the six months ended June 30, 2016 were $1.3 million, compared to $0.6 million for the same period in 2015, as we owned the manufacturing business for three months in 2015 compared to six months in 2016.
Interest expense was $1.3 million for the three months ended June 30, 2016, compared to $1.7 million for the same period in 2015. Interest expense for the six months ended June 30, 2016 were $2.8 million, compared to $1.7 million for the same period in 2015, as we owned the manufacturing business for three months in 2015 compared to six months in 2016. Interest expense consists of interest incurred on our OrbiMed senior secured term loan used to purchase the manufacturing business and amortization of related financing costs.
About IV/IM Meloxicam
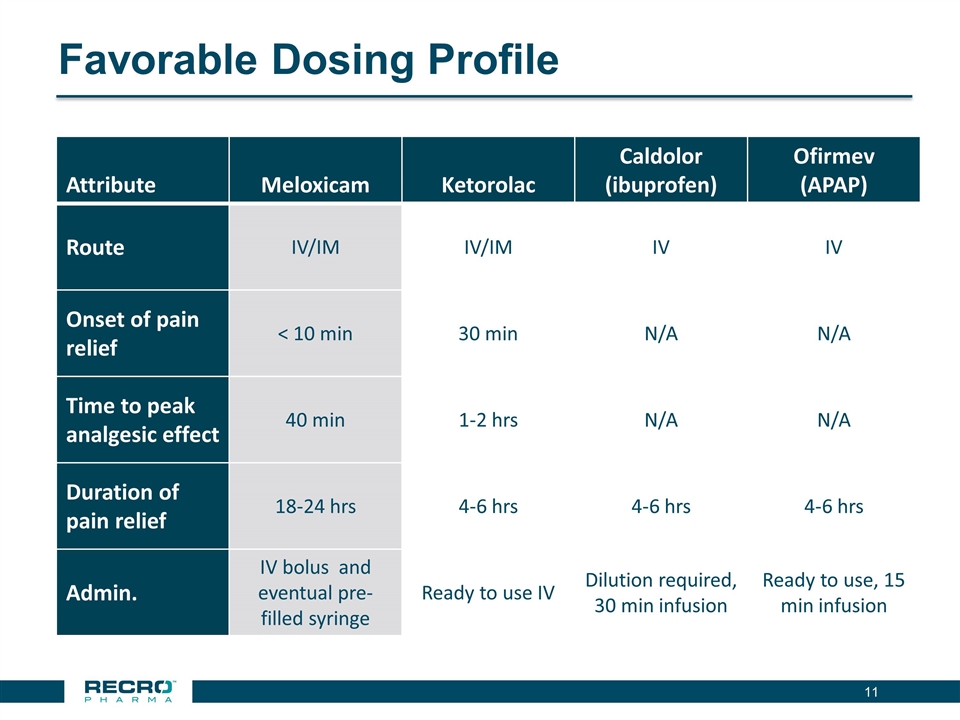
Meloxicam is a long-acting, preferential COX-2 inhibitor that possesses anti-inflammatory, analgesic, and antipyretic activities, which are believed to be related to the inhibition of cyclooxygenase (COX) and subsequent reduction in prostaglandin biosynthesis. Meloxicam has been marketed by Boehringer Ingelheim Pharmaceuticals, Inc. since the 1990’s as an oral agent, Mobic®. IV/IM meloxicam was designed using NanoCrystal® platform, a technology that enables enhanced bioavailability of poorly water-soluble drug compounds. Recro acquired IV/IM meloxicam from Alkermes in April 2015.
About Recro Pharma, Inc.
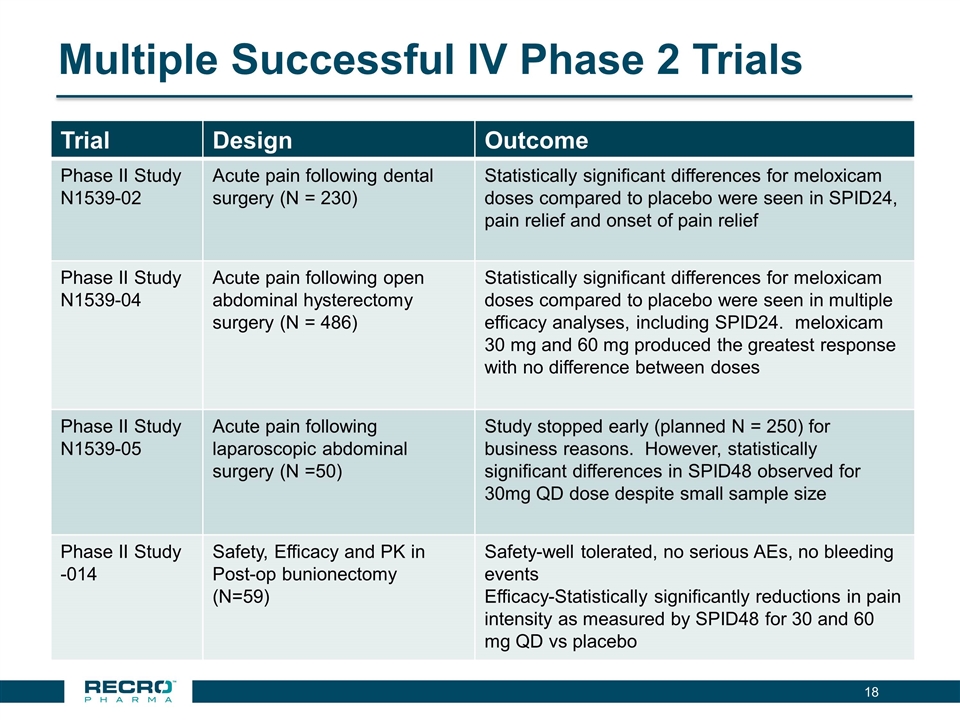
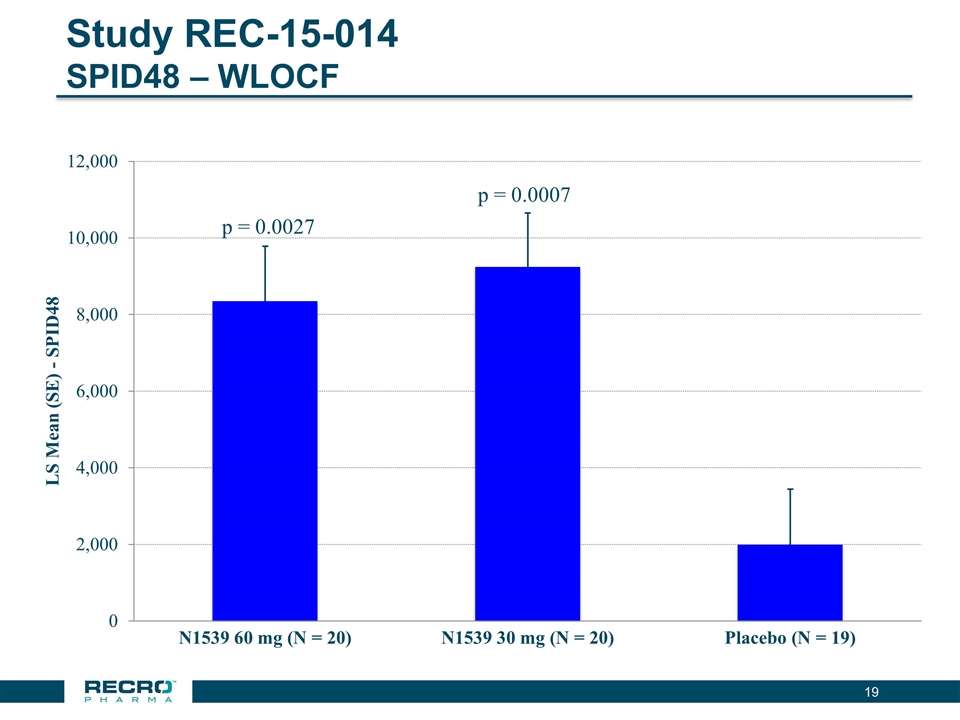
Recro Pharma is a revenue generating specialty pharmaceutical company focused on products for hospital and ambulatory care settings and is currently developing non-opioid products for the treatment of serious acute pain. Recro Pharma is currently developing IV meloxicam, a proprietary, long-acting preferential COX-2 inhibitor for treatment of acute postoperative pain, which has completed four successful Phase II clinical trials in postoperative pain conditions and
3

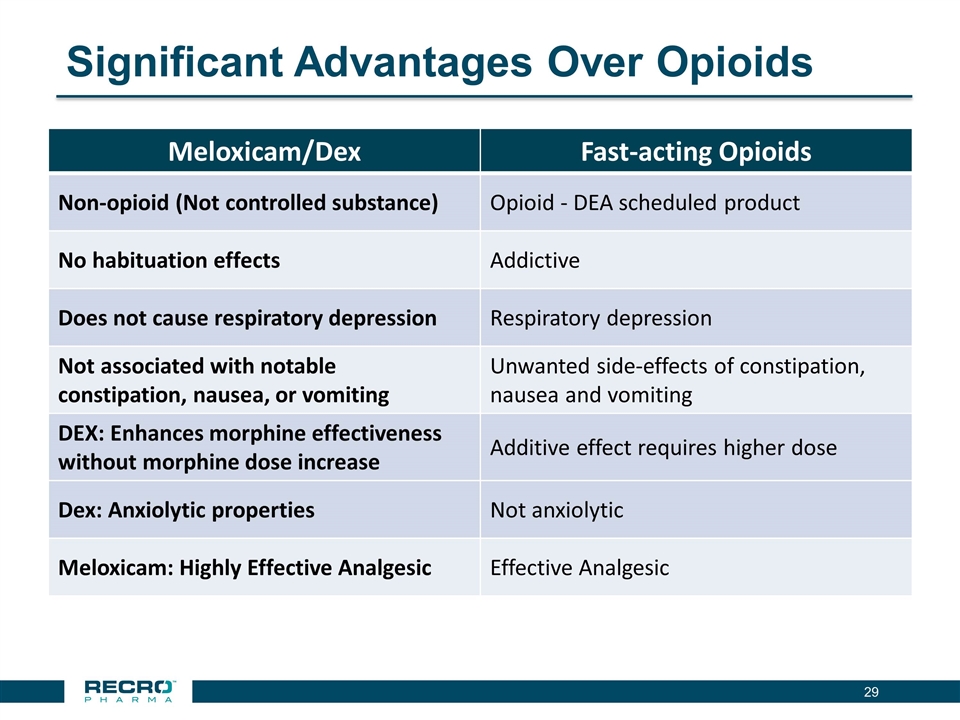
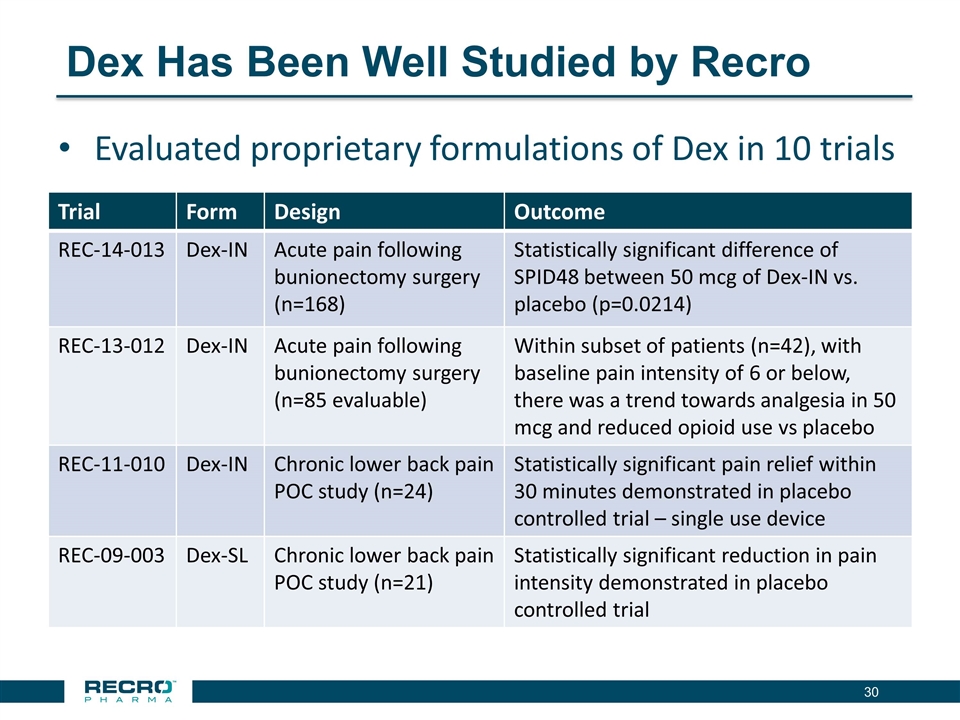
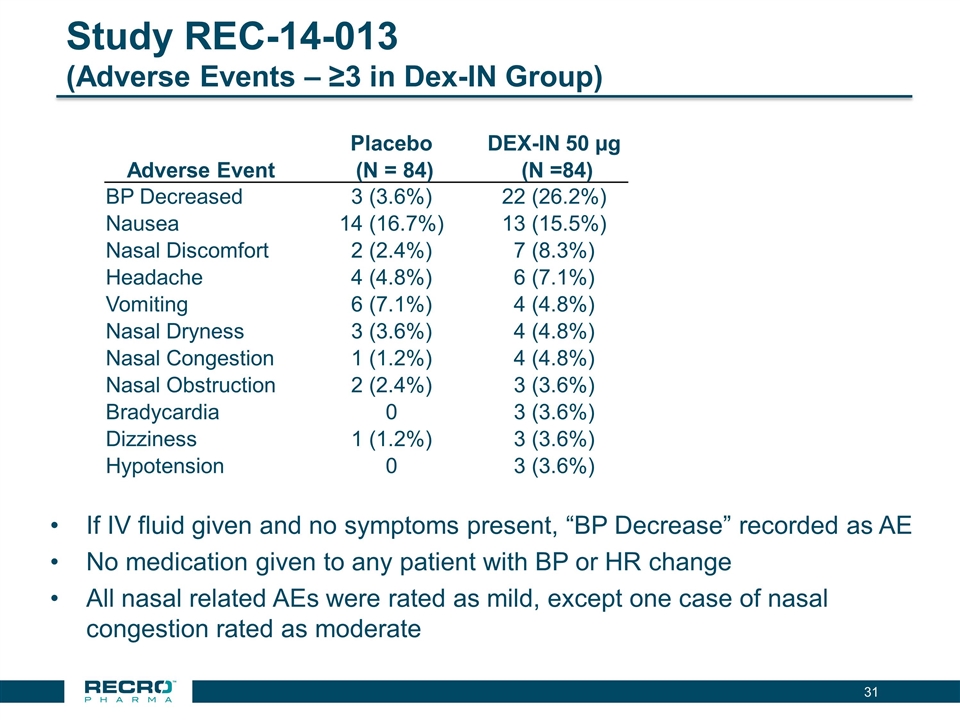
has reported positive results from its first pivotal Phase III clinical trial in patients following bunionectomy surgery. An additional development candidate, Dex-IN, a proprietary intranasal formulation of dexmedetomidine, is being pursued for the treatment of peri-procedural pain, and has had a past successful Phase II trial in bunionectomy. As Recro Pharma’s product candidates are not in the opioid class of drugs, the Company believes its candidates would avoid many of the side effects associated with commonly prescribed opioid therapeutics, such as addiction, constipation and respiratory distress, while maintaining analgesic effect.
Recro Pharma also owns and operates a 97,000 square foot, DEA-licensed facility that manufactures five commercial products and receives manufacturing revenues and royalties associated with the sales of these products.
Cautionary Statement Regarding Forward Looking Statements
Any statements in this press release about future expectations, plans and prospects for the Company, including statements about the Company’s strategy, future operations, clinical development plans and other statements containing the words “anticipate,” “believe,” “estimate,” “upcoming,” “plan,” “target”, “intend,” “expect” and similar expressions, constitute forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including: results and timing of the clinical trials of injectable meloxicam and Dex-IN; unfavorable new clinical data and additional analyses of existing clinical data; whether results of early clinical trials will be indicative of the results of future trials and whether interim results from a clinical trial will be predictive of the final results of the trial; the ability to obtain and maintain regulatory approval of injectable meloxicam and Dex-IN, and the labeling under any such approval; regulatory developments in the United States and foreign countries; the Company’s ability to raise future financing for continued development; the Company’s ability to pay its debt; the performance of third-party suppliers and manufacturers; the Company’s ability to obtain, maintain and successfully enforce adequate patent and other intellectual property protection; the successful commercialization of injectable meloxicam and Dex-IN and other factors discussed in the Risk Factors set forth in the Company’s Annual Report on Form 10-K and Quarterly Reports on Form 10-Q filed with the Securities and Exchange Commission (SEC) and in other filings the Company makes with the SEC from time to time. In addition, the forward-looking statements included in this press release represent the Company’s views only as of the date of this press release. Important factors could cause our actual results to differ materially from those indicated or implied by forward-looking statements, and as such we anticipate that subsequent events and developments will cause our views to change. However, while we may elect to update these forward-looking statements at some point in the future, we specifically disclaim any obligation to do so. These forward-looking statements should not be relied upon as representing our views as of any date subsequent to the date of this press release.
4

CONTACT:
Media Contact:
Argot Partners
Eliza Schleifstein
(973) 361-1546
eliza@argotpartners.com
Investor Relations Contact:
Argot Partners
Susan Kim
(212) 600-1902
susan@argotpartners.com
5

RECRO PHARMA, INC. AND SUBSIDIARIES
Consolidated Balance Sheets
(unaudited)
| (amounts in thousands, except share and per share data) | ||||||||
| June 30, 2016 | December 31, 2015 | |||||||
| Assets | ||||||||
| Current assets: |
||||||||
| Cash and cash equivalents |
$ | 14,845 | $ | 19,779 | ||||
| Accounts receivable |
11,480 | 8,580 | ||||||
| Other receivables |
23 | 36 | ||||||
| Inventory |
6,728 | 8,982 | ||||||
| Prepaid expenses |
1,855 | 757 | ||||||
| Deferred equity costs |
316 | 542 | ||||||
|
|
|
|
|
|||||
| Total current assets |
35,247 | 38,676 | ||||||
| Property, plant and equipment, net |
36,621 | 37,922 | ||||||
| Deferred income taxes |
16,644 | 15,637 | ||||||
| Intangible assets, net |
38,725 | 40,016 | ||||||
| Goodwill |
6,446 | 6,446 | ||||||
|
|
|
|
|
|||||
| Total assets |
$ | 133,683 | $ | 138,697 | ||||
|
|
|
|
|
|||||
| Liabilities and Shareholders’ Equity | ||||||||
| Current liabilities: |
||||||||
| Accounts payable |
$ | 1,752 | $ | 1,553 | ||||
| Accrued expenses |
5,274 | 3,418 | ||||||
| Current portion of long-term debt |
5,351 | 4,516 | ||||||
|
|
|
|
|
|||||
| Total current liabilities |
12,377 | 9,487 | ||||||
| Long-term debt |
22,225 | 25,244 | ||||||
| Warrants |
3,416 | 3,770 | ||||||
| Contingent consideration |
64,358 | 59,846 | ||||||
|
|
|
|
|
|||||
| Total liabilities |
102,376 | 98,347 | ||||||
|
|
|
|
|
|||||
| Shareholders’ equity: |
||||||||
| Preferred stock, $0.01 par value. Authorized, 10,000,000 shares; none issued and outstanding. |
— | — | ||||||
| Common stock, $0.01 par value. Authorized, 50,000,000 shares, issued and outstanding, 9,868,255 shares at June 30, 2016 and 9,224,315 shares at December 31, 2015 |
99 | 92 | ||||||
| Additional paid-in capital |
76,695 | 71,321 | ||||||
| Accumulated deficit |
(45,487 | ) | (31,063 | ) | ||||
|
|
|
|
|
|||||
| Total shareholders’ equity |
31,307 | 40,350 | ||||||
|
|
|
|
|
|||||
| Total liabilities and shareholders’ equity |
$ | 133,683 | $ | 138,697 | ||||
|
|
|
|
|
|||||
6

RECRO PHARMA, INC. AND SUBSIDIARIES
Consolidated Statements of Operations
(unaudited)
| (amounts in thousands, except share and per share data) | ||||||||||||||||
| Three Months Ended | Six Months Ended | |||||||||||||||
| June 30, | June 30, | |||||||||||||||
| 2016 | 2015 | 2016 | 2015 | |||||||||||||
| Revenue: |
||||||||||||||||
| Manufacturing, royalty and profit sharing revenue |
$ | 16,933 | $ | 16,704 | $ | 34,072 | $ | 16,704 | ||||||||
| Research and development revenue |
346 | 1,956 | 949 | 1,956 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total revenue |
17,279 | 18,660 | 35,021 | 18,660 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Operating expenses: |
||||||||||||||||
| Cost of sales (excluding amortization of intangible assets) |
9,547 | 9,395 | 19,818 | 9,395 | ||||||||||||
| Research and development |
8,320 | 2,821 | 16,129 | 4,575 | ||||||||||||
| General and administrative |
2,763 | 2,597 | 5,421 | 4,986 | ||||||||||||
| Amortization of intangible assets |
646 | 592 | 1,291 | 592 | ||||||||||||
| Change in warrant valuation |
1,240 | 882 | (354 | ) | 882 | |||||||||||
| Change in contingent consideration valuation |
1,534 | 2,000 | 4,512 | 2,000 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total operating expenses |
24,050 | 18,287 | 46,817 | 22,430 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Operating income (loss) |
(6,771 | ) | 373 | (11,796 | ) | (3,770 | ) | |||||||||
| Other income (expense): |
||||||||||||||||
| Interest income |
8 | 4 | 17 | 8 | ||||||||||||
| Interest expense |
(1,317 | ) | (1,688 | ) | (2,829 | ) | (1,688 | ) | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net loss before income taxes |
(8,080 | ) | $ | (1,311 | ) | (14,608 | ) | (5,450 | ) | |||||||
| Income tax benefit |
195 | 184 | ||||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net loss applicable to common shareholders |
$ | (7,885 | ) | $ | (1,311 | ) | (14,424 | ) | (5,450 | ) | ||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Basic and diluted net loss per common share |
$ | (0.83 | ) | $ | (0.17 | ) | $ | (1.53 | ) | $ | (0.70 | ) | ||||
|
|
|
|
|
|
|
|
|
|||||||||
| Weighted average basic and diluted common shares outstanding |
9,544,629 | 7,829,536 | 9,398,288 | 7,799,282 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
7