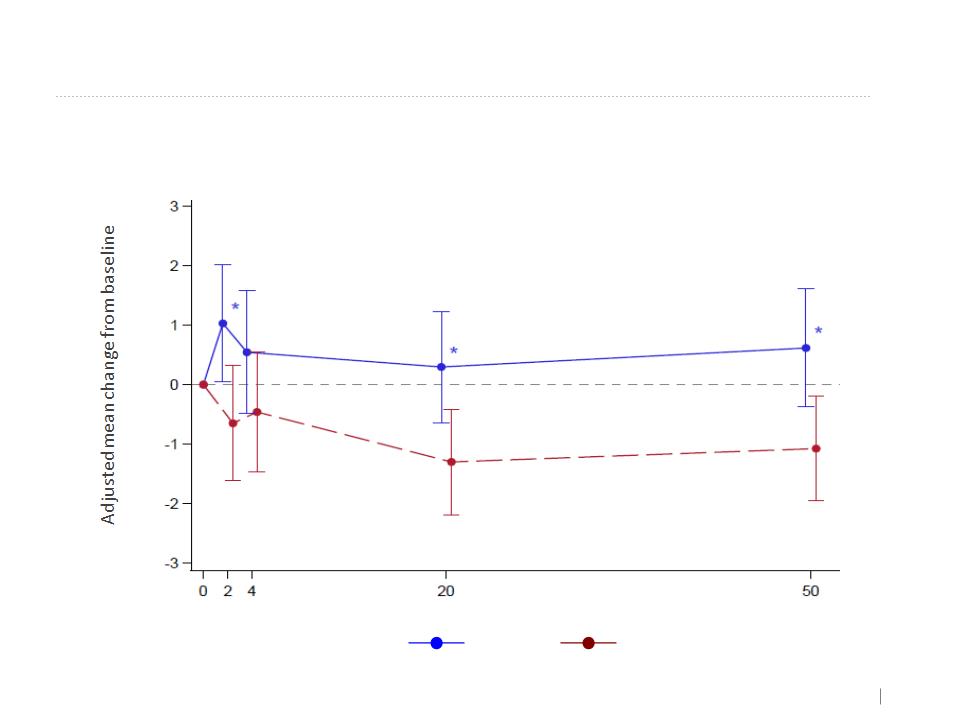
*=2"DDDCAB:25U1%&69C@"A(/
M4DJO=ZC::?%YEU.D8[;CR?H*X_6?&Y^:#2QQT,[#^0_QKD)[F:ZE,L\K22$\
MLQR:[Z.!G+6>AY>(S.$/=IZL[J]\>V\9*V5L\I[/)\H_+K6%<^,=8N#\LZPK
MZ1H/YG-<^/>NDT?PE-J]@MW'=1QJQ(VLA/0UV^PP]!;]9Q6(ERP?R1ER
MZOJ,Y_>WUPWUD-1?:[C_`)[R_P#?9KIY/`-S'$\AOHCM4MC8>U0:9X,GU+3X
MKM+R-%D!(4H3CG%6J]"UTU8SEA<5S7"GVD-:-MXLUJW((O
M&D7TD`:K]YX&N+.QGN6O8F$,;.5"'G`SBN6JX>QK+2S,JGUG#OWFT=S9?$%@
M0+ZT!'=H3_0_XUU6G:[IVJ#_`$:Y4O\`\\W^5ORKQVG*S*P93A@>".U8UD_>1[EWI:\TT;QK>6)6&^SOJ#[BO(KX:I1W6A]!A<;2Q"]UZ]BP>E,/2GFF&NJ
M*\.WTJ[-WJFH/F-]*Q^T>C3^$[3PPW_$L7_>-=3#)@`5R7AP[=.`]&KI().G
M-=C7NGA57;$2]39B.15N/CO6;#)QU_2KB25SS1Z%&6A/45BT=
M<6.?I3`W%-+^]0E\&E8OF)F(JM(:<7J"1Z+!S%6=N*YW79,:?*/[W%;EP]<[
MKIS8M]16B6AA*6J.7L&,><^M;D$^WYJP;<_,1[UH,^V#\:4'>FQ5X_O+E#59
MMTF?4UM:-M:P0D`\GK7-:@^2/J*Z'0V_XEZ_4U&&?[T6+36'1MQI'_<7\JM1
MQP<`PQG_`("*IQM5E6KTK(\5SE%[FA%!;_\`/"/_`+Y%6EB@"\0QC_@(JA#)
M5M7XKGFCLHU+CV5`.$4?05Y)XCXUNY_WS_.O6';BO)_$W&N7'^\:Y:S]T]3"
M.]0H1FK,.M8([)HTEG&Y2?2NLUKQ#I]CHGVC3II#*R!4W@9#]
MS^`KBXMKLN6'4<>M5)XK9=6\FY2:"-6Y*J6Q[XXISDXJ_W!Z-S@?6MOPO
MXC?1;EM/OLM8R'DCK&?[R^WJ*ZR\O/#4G`'XMK8CW4FZF9I,UVW."Q)FDW4S-)NI7"Q)FDW4S-)FF/E)-U&ZH]U)N
MHN'*2[J,U%NHW4KCY23=1NJ/-)NHN'*2[J-U1;J-U%PY27=1NJ+=1NHN'*2[
MJ-U1;J,T7#E)=U&ZHLT;J5PY23=1FHMU&ZBX[$FZC=4>:,T7"Q)FDS4>:,T7
M'8DS29IF?>C(I7"P_-&:CS1F@=A^:,U'FBD%B3-)FF9I,T#L/S1FF9HS0%B4
M=!]*<*:O0?2E%*V@>:9PL:#+$]A22;=D#:2NQMY?6^GVKW%RX2-1^?L/>O-M;\17.L3%O:W-K-WN.5MT.(X_3W/O637LX7"*FN:6Y\_C<=*J^2&P[-%%
M%=QY@ZM6Q\0:EI]L+>UN/+B!)"[0>364*<*4HJ2M((SE!WB['KFG7,MWX9BN
M)FW226Y9CZG!IOA;CPQ:'T0_S-8^E^(M-M_#D-K))()5@V$>4Q&<'OBG:#XB
MTZST*WMII9!(JD$")B.I[@5XLJ4[.RZGT<:\.:+;^RSF9_%&KW$$D$MUNCD4
MHR[!R#QZ5CT_R9"<^6_7^Z:#%(.J,/P->S",(JRT/G*LJE1WDVQ*!0**T.=B
MU=TW5+K2;H7%I)M/\2GHP]"*I45,HJ2L]APG*#YHNS/7=$UZVUNUWQ_),H_>
M1$\J?\*TFKQBPOKC3;Q+FV?;(A_`CT/M7JVD:M#K&GIJB\2-]*MS$QUF@G%C_`,"K;CD(
M-8.AG%E_P*MA&KT(O0\'%+][)FO#-Q5Q)AZUB1RD<596?WK.432E5MH:XN!3
MO.K+$_<&I!<#UK%Q.V-5%_S?K3&DJH)^.M-,WO4V-?:(L&6H'ESWJ)Y<]ZKR
M2YZ4U$3J(2>0\\U@:T^;-A[BM::3@YK#U=O]$<^XJ[:,PY[S2.=M7S(?K5^X
M?;:Y]Q67:-B4_6K]Z"UB0/45E#^&SNJ+]XKF1>2`D#/>NFT(_P#$N7_>-/]XUAA'^]*QT4J!MH:L*:K)4ZFO5N?/R1:B8@]:MJ_O6>IJ0.
M1WJ)*X4Y=>)?\`D+R&N/$*T#U\NGS5?D9T
M=2['E^11GBHH>@K3LT&"W>L:24G9GJ5)+[
M\_\`+0C\:E8;%7OS(X98["=F67^%CE<*EFI]0['_`-EK.O/A#+*=XF@W=\Y-
M*_BO4#_RV/YU!)XGU$_\O+?G6JP^)ZR7W&/UW#KX8O[S*U+P$=(@W>3Y;@<-
M`\;K@FM;\ONO=&L(>UCS-W3[[HZ^Y01W#HI^4'CZ5#FH8)3+"K-UQS4F:[
MD]#S'&SL+FDS2;J3-.X6'9I,TW-&:+CL.S29IN:3-(+#\T9IF:,T7'8?FDS3
ME
M;%N*S+5>E:UN.E<"/5-&`=#6C".E48!P*T(ATI,I%R.K*56CZ59CJ&43+7"^
M,M:^T7/]G0-^ZB/[TC^)O3\*ZG6M2&EZ3-<9_>8VQC_:/2O*V=I&+,GZ5T_AG_D5+;_`*YM_,UY
M4X-8>+?5GN4ZBEBI16T4/'BS0_\`G\'_`'P?\*T-/U.RU57-J_FHO#'80/U%
M>>>&_#$NL$3SYCLE/4=7]A_C7?W-WI_AW306"PQ*,)&HY8^@]Z5:E3@U"%W(
MK#5ZLXNI5242EK7A.RU*%G@C6WN@,JZ#`)]"*\SG@DM;B2"52LD;%6![$5Z#
MX9\2W&LZMOX5E4WO6OFZM-
MTYN+/M*%95J:FNI6>JL@JVXJK)UK(V*,HJE,*T)!5&44`9LXH[+_`+H_E3IA
M43G:!ENP_E7?@/C?H>=F7\-$P<"E,GH:J;Z7?CI7J6/)YR8N:JSOP>:'FQ5&
MXN.*J,=3.=5)$-P_!K.E+X6:T-:BNN7$_`>IEC_>_(RXZT[-L1M69'6C:?ZLUQTGJ>
MQ6V*\Q^-;/ADXFF_W:UH_P`4Y\4O]G9U>_GK32U,SS29
MKT;GSUAY-,)XI-U,)-.XTC)UNW-R8@#C&:SI[7[-I[*3G=S6S>Y\R/\`&J.J
M_P#'H/I7/."UEU/4H5))1AT+]A_QZ(?:K!JMIY_T./Z59-;Q?NHY*GQL2DI<
MTW-5&J+H:NZG!JSEU:V/57%
M3)J%J?\`EH1]13YDS)T9KH70U/5L566>W;I,OXU*K(>DB'\:9FXM="UOS&WT
M-9:#,R_05H!6V-T/!Z&J40_>+]!7G8[H>QE=];FG;#I6M`.E9EN.E:D`Z5YZ
M/81HPU?B[51AJ_%VI,I%M.E6$J!*G2H*.'\<7QEO8;)6^6)=[?[Q_P#K?SKE
MA5K5KK[9JUU<=GD./H.!_*JHKWZ$.2FDCY?%5'4JN0M+24HK8YA:6@44"8X5
MZCX4LUT[P\DDGRM*#,Y/IV_2O.=+M&O]2M[4?\M)`&]AW_2O1_$]P;/0OLUN
MI,D^(8U4HWC7^HW%TW61R1[#M^E>E^&?
M^13MO^N;?S-8^B^$DLK.2[OU#W/EL4CZA./U-;/AK_D5;?\`ZYM_,UEBJL)P
M48=&C;!4*E.HYU-Y)F-H^OVNB^$H"Y#SLS[(@>3R>3Z"N2U+4[K5+IKBZ?'+\Z?KMO+G".WEO]#Q7JS=*\5R1R.H
MKU_2KG[9I-I<9R7B4GZXY_6O'S*G9J9]%DU6\94WZDD@JI)UJX_6JLG6O+/<
M*4G0U2FJ]+5*:@#.FJA,WS8]!5^>LRJI^M:@
MKOA\*/%Q/\1DH-.S4=.K0YFAV:*;2YH%8*2C--)J1I`3S3&-*:C8T%I"$UFZ
MJ?\`0GJ^QK/U,_Z&]1/X6=-!>^CG+8_O#]:W;#_CX2L.`?.?K6YI_P#KEKFH
M.QZ>*V+EV,9-1Z>?E?\`WJ+PX4Y-,TSF.0_[5=-_>.%+]TS44FI5-0+4RUI<
MXI$H:EW4T=*6BYD-8UR&O#_3G/L*Z\UR.O<7[_2N?$_`>CEW\4R4J9;PV^5V
MY!%0I22IO?`->>FUJCVVE)V8UKEY7.W;]":T]'O)[-W8P,P88X%8[V,W55)^
ME1^7=Q?=##\:A5)PES6+E2ISCRG9'6YC_P`NDOY&E36)2XWVLP7N50D_E7(I
M=7R]))!_P(U)]NU#H99/^^C6ZQC?EE+^
M*FN7^V7QX,LG_?1IC/>2=G8T_K;\Q+`T^MCHIKZ9QF0QQ[?5QG\NM9]SJ3SI
MY2R"3`Q6>FGWT_6)L4&U:"38W#J>:AUJDEL;0H48Z)G6:+,\UEESG!P*TCTK
M(\/$FRD_W_Z5KXKTZ3]Q'AXE6K20VD-/IM:7,;C:*=3:+E"4E.[TF*+C&T4=
MZ6BX"44M%*X"44N*7%%P&TM+BC%%P$HIV*,4[A<2EHQ3L4KBN-Q2XI<4N*+B
MN)2XI:7%,5QN*7%.Q1BF*XF*,4[%+B@5QM%.Q1B@5QN**?MHQ0%QE%/Q1BD%
MQE%.HQ0%S)5V`&">E3+.Q&#S]:JJV1^%2*U8)G=*)*Q4]47\J84B/6-?RH)S
M247$KH/)@/6,4HMH#VQ0.M.!HT#FEW%6RB[-3Q8@'AJ0-4Z/S1H+FD^HS[*X
M&0U2Q??7_=%6%;@].E5X3EU_W17#C.AZ&77O*YK6_:M6#M67;]JU8.UG_IBW\J(*\D%1V@
MV>5CGDTZFTX5]$?*,44ZFBG4$CA0*!2T$G8>`K#S+V>]=?EB78A_VC_];^==
MZ88VF29E!>,':3_#GKBL;PU:+I?AZ$R_(64RR$]L_P#UL5:U^*>;0;H6TC)(
M$W`J<$@65BTCDLS'N37J.@7*ZUX71)3N8H89/KC
M'\L5YC=6[VEW-;OG=$Y0Y]C1@$HN4'NA9LW.,*B^%HAHI:*](\02FFG4TTF-
M##Q7I7@R;S?#<0[QNR'\\_UKS0UZ%X#/_$EG'I.?Y"N#,5>E?S/8RAVQ%O)G
M1R55DJW)562O"/IRE)TJE-5V6J4U`S.FK)N?]I`2>E1M4AIC=*]<^?97EJJ_2K4O2JK5:,);D#U7?K5IN:K2"AFD"!SQ
M5.6K;CBJDM92V.NF4Y>]5U^]5B456_CKGEN=T-CI-%_X]F^M:H%9.A\V[_6M
M@5WP^%'BXG^*PI:4"EVU9S-B44[&*3%`AM--2$4TCFD-$;5&U3$5$PI&D60M
M5#4/^/1ZT&K/U#_CU>HELSIH_&CGH#^\/UK9L#B:/W.*PXSME/UK3M),2QD=
MB#7)2D>IB(W1IWG!(--TK_5R$=-]/OIXI(5Q][;S2:4NV!A_M9KI^VC@VHNY
MHK4RBF**F45L<$F*!2XIP6G;:&97(2*Y'Q`,7[?2NR*UQWB(?Z>?I7/B/@/1
MRU_O3'2GX_>BFH*>?]8*X%L>X]S2MQ6Q`@>V;Y`V#W%8L$F*V]*N5B:3>,J5
MZ5W4VCS,3>UT58H(FOU0QJ>#D$5JK86QZP1_]\BJ$!#ZH748&,5MQBM816IQ
MXBI)6L^A"MC;#I!&/^`BG^1&OW8U'T%6@O%-=>*JR.+VLF]S/F&!7':A_P`A
M.0>M=I<#@UQNH@C5)*Y<2O=1Z^7.\GZ&WX>'^B2C_;_I6OBLKPWS;3#T!3PU5U8\?2G;ZY$SV'$L;CZTN\^M5P].#T
M[DN!8WGVIX>JP:GAJ+D.):#5*C#(JH&J9&IF35C01ACGH15>U/(^E*LGRX/I
M3+4\K]*XL8M$=^7WO(W+?M6K!VK(MSTK5@/2N$]0TX>E7HNM4(3Q5Z(]*EEH
MNITJ#5UWZ)>*.\+?RJ9.E221B:WDB;HZE3^(J8NTDQS5XM'CXIPI&4H[(>JG
M!I17T2/E)*S%%.%-%.%,D6M#1K(ZCJUM:X)#O\WT')_2L^NU\!6.Z6XOF'W1
MY:'W/)_I6.(J>SIN1OA:7M:RB:_C2^^R:(+9#A[AMN!_='7^E:?AR]&I:#;R
M-AF"^7)]1Q7"^,K_`.UZXT2GY+==@],]3_GVK3\`W^RXN+!CPX\Q![CK7GRP
M]L*GUW/3ABD\8X]'H_4<,/*?'J.G]:Z3PM_P`BO:?[A_F:K$5/:8>$O,G"4O98NK'9'@2W.R\`7_`)=[/8LWRRKO0>XZ_I_*H/'5A]FUE;E0=EPF
M2?\`:'!_I6#I=XVGZG;72DCRW!./3O\`I7HGC"S74/#IN8_F:'$J8[KW_0UY
M]7]SB5/I(]:C_M&"E3ZQU1YCVHHHKT3Q1*8U/IAZ4AH8:]$\"(1H>'UKU'PG#Y'AFUXP7R_P"9/_UJ\_,7:E;S/9R>-Z]^R-22JLO2K4E5)3Q7
MAGTY3FJC-5V7O5"IVJ!Z&5$@8<53E6KK<55E-9R.J
MF]2A**J?QU;G-5,_/7-+<]"GL='H/,,@]"*V11X(\.7&KQS%)8H@,'YR>:[1
M?AY=MTOK;/XUU*O3@K2=CRJV'J5*C<%+U3_P`!_P#KU/UV
MA_,/^SL3_*<"13,5WK?#JX'2\CQWXJM+\/[E!Q>0_C36+I/J'U#$+>)Q)%1L
M*ZR7P5=Q_P#+S":S;CPW>0@G,9QZ-6D:D9;,S=&I#XD<^P-4=07_`$23Z5N3
M:;)1B__``KMV6N-\3+_`*?_`,!KGQ'P'I99+]\8<8IS
M<.*12%&2<4N1(WRGI7"CZ![W+4/6M"(D*<54@2KT:<&NJ".*J]2S8C_2`:Z"
M):P[)?WPKH85X_"NF"LCR,7+4>%XICKQ5@+Q377BJ.!2U,NY'!KC-3_Y";_A
M7;70X-<)KA(OG"G!.*Y<3I$]S*]9-'0^%QF"X_WA_*MTK6%X.4FVNL\_,N/R
M-=(5K:B[P1R8]VQ$D5MM)MJP4II2M3E4BN5I-M3[:3;3*YB`K2;:FVTFV@?,
M0[:-M2[:-M(?,1;:-M2[:-M,.8CVT;:DQ2[:=@YB(+2[:DVT8HL+F(\4NVI-
MM&*+!S$>*7;3\48H%<9BEQ3\48]J87&XHVTZC&:+!<3%&*6B@!N*7%+10`VB
MEH[T`)1110`E%*:2@8E%%%`'+$;1SV%1;P:C:9F'(-(&%<',>^HOJ3*P-.!X
MQ4(*YIP*T^831,#4@:H`1ZT\'WIIF;1.K5*K567ZU(IJDS.42XK?+3K8C*_2
MH$8[2*?;GE?I7)C'HCJP"M*1OVQX%:L!'%8MJW2M:!NE<)Z-S6A-7XC6;">*
MOQ-TJ66F7XSQ5A#51#5E#4,M'FGB.T^QZ]Q\RWA
MOT7F,['^AZ?Y]ZXD&O=PU3GIIGS>+I^SJM#A3A30:<*W.445ZEI<:Z%X6620
M8*1&5Q[GG']*\^T&Q_M#6K:`KE-^Y_\`=')KL?'-]Y&F16:'YIVRW^Z/_KXK
M@Q?[RI&DCTL$O9TYUWTV.#DD::5Y7.7=BQ/O5O2;UM/U6WN@>(W&[Z=ZH4X&
MNYQ3CRGF*;C+F6YZ[X@LQJ>@W$:89@GF1GW'--\+_P#(KVO^XW\S47A._P#M
M^@0[CF2+]VX^G3],5JV=HME9&!/NJ6(]@23BO`DW%.D^C/J:<5.2KKJCQL_>
M/UH%*?O&D%?0QV1\A+<<*]1\*7:ZIX;6"4[C&#"X/<8X_0_I7EPKK/`=_P#9
M]7>T8_)<)P/]H<_RS7)C:?-2NMUJ=V65N2NHO:6ASM]:/8WT]J_WHG*_6J]=
M=X\L/)U.*\4?+.N&/^T/_K8KDL5M0J>TIJ1RXJE[*M*`TTTT^FFM3!!%$TT\
M<*C+.P4#W->QV\`MK.&W7[L2!!^`Q7GO@W3OMFM"=US';#?_`,"[5Z0U>+F-
M2\U!=#Z;)J/+3=1]2O)TJI)TJW)TJG+TKS3V2G,>M9TQJ_,>*S9Z`,ZX-4I6
M5=I<.%Q]X+FK%RW%/3#QJH1"0HR&')^F*[L"_?9Y^9J]->I15HY`3'(K?0\_
ME3FM+C&1#(1_NU!JEO\`998Y(482,>%QUJ"+5YXALD2=<<_*.U>OTNCY]*[M
M(DFM;CG]Q+_WR:I/;7`_Y82?]\U+<:Y$Z#RO-#>[#'XTB>)8XH#&VDP3RYX=
MYAK.4D=E.BEO8AEN"3SFDLGCEO8TD?:N>2:BN77'`(/J2/\*JQ,@N%+
M-GD9Q7)*=IH[E37*['I6B:[IUM*ZRW$ZIM"J(!S750^*=#\K;_:.H(<8!:,'
M%<1H-]H<$C2SVEQ&&7D1'(_#-=-'K/A,!3-;ZBN?85VSDGJ_P/%=-QDU&/WF
MJ-?TQ@-GBBYC(_OVV<_K3EUH-L,/B\9!^;?`0#^&,C\ZC%[X.B`:6UU-><9:
M,5;BO?!LL>Z/3M2D7U$6:Q;79_+K0@@[@T17Z8YI/.
MU"5OF\5:7(H/'++Q^!JH+WPH,*-#U1\\@&$?RS4;7/A;/R^&=1![GR57^M9<
MNNS^Y&J;MJU][--X=:&]4*^J$?_`!54+Z32K?!72-6M_3-SY?Z9-4E*^GY+_,EV:U?XLDDO
M/$D)*3:0[8_B24-5.>[O)%_>V/>LG_A)=6_Y:W,/
MX[:!KLT[".:6*3)Y15`!_&M56B]#&&"J0DG9:'/E6^U/O^]GFM"#C%59G,M]
M-)@#!Z.6U%&NKG!R,-QRPXJ:R8E\J,]A]:KW,:K)A?7O5G3LH<@@<\<5Y4+^T
ML?6R2]G=&Q;]:OH.#5&#EB?4UH)7IP6AX]9ZENR7]Z*Z&%>!6#8C]ZM='"O%
M;)61X^,EJ2!:9(O%6HUR>U,G4"E@W8X-<)KR8
MU!6/<5ABE>F?093+WSH/!R_Z-<_5?Y&NE*5S_@X?NKL>A7^M=,5K2E\"//S"
M7^TR*^RFE:L%:816IQJ1`5INVK!7-,*TRU(KE:-M3;::13+4B$K2;:E(I,4#
MYB/;1MJ3%&*+#N,Q1MI^*,4PN,Q1BGXHQ0%QF*,4_%)B@+C<48IV*,4!<;13
ML44P&T4M)0,3%&*6DH`*2EI#0-!2444`)1110,0T44E`PS29HHH&<@)W(&>:
M7S0>JJ?PJ(*>/I2[:\S4]^R'ED/5!283L"/QINVEQ1J`N!V)IRDYZTT"EH$R
M0$U(KGI4`-/5C5)D-%I7-2V[MFK7MVZ5@VS
MUKV[\"N0[#9A>M")JR(GK0A>DRDS4C;BK*-5&)JLHU9LT3)KFWCO+26VE&4D
M4J?:O*KVTEL+V6UE&'C;'U'8UZNC5S_BO1?M]L+R!`[_`,C4I;-F^2==RC_:'_UJ]$/W&^E>
M+V%TUE?072'#1.&_#O7I@\6Z*4_X_`"1R-C0W$?WHG#"H!3A0TFK,E2<7='J'B:V3
M5O"[3Q#<443QX],<_H37E_-=YX;\26$.AK9ZA/L9"4`*D[E/T^N*XJZ2);N5
M8'WPASL;'49XKAP<94W*FUML>GFK6I6Q2!2Q`4$DG``I^*ZSP9H!
MN9QJ5RG[F(_N@1]YO7\*Z:U54H.3.+#4)5ZBA$Z;PWI/]D:1'&X'GR?/*?<]
MOPK3:IFJ%Z^;G-SDY,^WI4U3@H1V17DZ52EJY+TJG+4%%"8UFW'>M*;O67J_/GUQ65==_I5RI1?
MF:DK*;TRL4.WY0">E5I+Z*&[>4HK%C@@'M6+-<$L1NK/FDD'(8_G7K\B1X,:
MSET.DO)M-N$S+$0Q[X!K!N[/3F<^6V.:HR7$Q'4U2FEF]Z:21:A.3O>Q:DTZ
M'/R3D9]ZKO9R*/DN#]*J-<2@5`UU+GI2-ZM]15:2VG<'H/
MH:C^V2CKFGIJ#KP0>:F\6;*-2.UC.N-$G8Y9IN?]G--@LC;;2"H;/.00:Z:#
M5T#!V7C/(QUK6\_3-0MEAN-BD*2)-O(/:L_JT+\R&\;5C[LXZ'/PQVSHNZ50
MYX(W<5HQ:5YR@(5<=0%<&E_L^U%;\8Q8W/MA35>.QO[8#[-?SHI["2K"WOB6`J!?32+']W
MH]],NN?]AJA_X2/Q$@P9
MK@@=Q*U,;Q7K.,/<7?/JV:7O]D5R=I?B/D\):AVLKQ,>@(JK)X/NC]^WN_\`
M@342^);^1?GO)_HP_P#K54DU>>3K=]>QQ_A1RWWL4O:+9L27PDJ9WQRCZRU7
M/ARR7[R_G-3W:Z(.6)S51X[@]7;\Z'"/\I49UG]LD;2=+CX;'_?PFJ[6NEQN
M"@.0IK-JVT3HC?K-BPK]HN9'0<%L@5L6UB[$#:>:P
M[1FM+DK)QN/'O73PWL:P$B0+)M^7ZUM0BG&_4SQ;FFN4273Y8>67]15G3H]^
M[';K5$ZAJX#!&`#6B:N<%=3C3;DUW')S751Z/9
M(%S/#SW!!JPNG:<@R9XC]#FN:6+@=E+*ZN[:.(EC!.5!_*N;\42)!IX5QRQX
MKUEHM+''F+Q_LFO-?B%HUSW"<`SJI!^A-"Q*DK'31RYTZJDY'DUT
MZ%V`/4U;TC;YH+\@-WJO=03V4A\Z*+\)`W\B:DM)X]XR=H[\=*XX?Q+L^CJ+
M]W9&Y#C><=,U>48JE92PRR;4))K?CLX@@,LL:9'\35Z]-:7/#KRY968EB/W@
MKI(%^45EV-I;&08O;?\`%L5TMK8+(HVW=JZB.)/
M:F7"<<"M^'0KO9E!&P]0XYK/O(H87:.:XMXW!^8&0$_I6*K1;T9E]5JQAJCE
M[Q3M-<3KJYN0.G%>H/I*W5LTT%[;2(O7;)S7`>(+3;/@2Q@@=UJJEJD&HGI9
M"N8[OGNO]:ZDK7,^"(F2*\+U34N/>AP
M%S,K[/:EV5/BE"BER!SE?93@E6`HS3P@HY"7,KA:;$WS"KNP8-9D;?-CWKBQ
MBLD=>#ES-F[:OTK6MWX%8%L_2M:W?I7"CL;-N%^*T(7Z5CPO6A"_%,$S7B>K
MD;5EQ/5R-ZEEIFBK5*K531ZG5JFQ=SD/$_ATQL]_9)F,\RQJ/N^X]JY1>:]?
M!!&#BN2UWPF)"]UIJ@,>7A'0_3_"O1PV+7P3/+Q>#^W3#0;3PYJ5LBS0B.Z`
MPZM,PW>XYK>7PGH1Y^S'_O\`-_C7F)5HW*,I5@>01@BI5FE`XE
MK_\`?1IPGF_YZO\`]]&I^J5?^?A7UZA_SZ1Z;_GJ_\`WT:/JE;_`)^$O'T/^?2/3!X/T+_G
MU/\`W^?_`!J0>#]!_P"?4_\`?Y_\:\R$\W_/63_OHT\3S?\`/63_`+Z-'U2M
M_P`_"?[1P_\`SY1Z8/!^@_\`/J?^_P`_^-+_`,(=H/\`SZG_`+_/_C7FGGS?
M\]9/^^C3O/F_YZO_`-]&E]4K?\_!?VEAO^?*.]U#1/"VF0-+<1`$#A!,Q9CZ
M`9K@)2CSNT,?EQEB53).T=ADT$EC\Q)/J:Z70?"4VHLMQ>!H;7J`>&?_``%:
MQ2PT7*I*YSSE+&S4*--+T_4I^'?#LNL7.^0,EHA^=_[WL*]-CAC@A2*)0L:#
M"J.PI8;>*V@2&"-4C0855'`IQKR,3B)5I7Z'TF!P4,+"V[>[(VJ%^E3-4+=*
MYCN*\E4Y15R2JDM`&=/WK*N#UK5G[UDW1X-`C'NCUK.NKH`A-HX`Y_"KUT>M
M85VK&=B/0?RKT
M=1C<:]>YX:A9>ZPD@Q_>_*JDB-Z&K#W4V.OZ55>YF]ORI-HWIJ1"RL/6H74U
M*US)Z#\JA:Y?^X/RJ&T=<5(85II0'TI3(S_=)SZ5
M/'E"N8WXZ\56$X_YYU8AO-I_U9_.G%Q[DS4K;$JO(N2$<+V.*MQ7FT[BS!AZ
MBE@U.)/O1-5Y-7LC@/`3]0*V5NC..HY?R$:ZF.2)2#FK2:L^`?-4@\$4JZAI
M+_?MA_WP*<9M#?K$5^@Q3NX!(!]0#230#M6C%%$D"1Q,&51@$=ZBD3GI6R6AQNK>;9DO%CM5648[5KR(
M/[M49D&#Q0XG53J7,"[\Q^.U01B[484\>_-:S0@GI3UC`7I7*\/=WNST%72C
M:Q6M+N]AD4B*-B/7O716^MWAC*MIT3-C&]7VD?I65&F#TK9TU0?O"MHTW;5L
MXL34A;F<436>HZI&04M8C@YPS`C]16U%K6J_]`RT8D8.2/\`"EMA&`/E%;]A
M+:*!O1=U152MJKGEQQ/-.R27WF.FK:^RG;I]F,\_='_Q-21:EXEB4EK.R*^Z
M#(_\=KL(KFW"\*M.DN(2O`'Y5QN<;VY#T8QG:_.OZ^9Y_=:IXE;)$D,9']Q5
M'_LM9%QXXUK28GCN(+6Z+#!-PN_].GZ5VVK[64E0`:\H\7*_VI>N,5K.,72N
MD&#JREB/9RLS#U;4WU6X,LR1)S]V)`H_2J"A!T_G04YIR1Y/2N#EE)W9]"DH
MQLA\5S-;.&BY]16E:ZM//,%EA1?]MWX'Z&JL,(S6A%$F.5KKI4ZG\VASU9PZ
MQ-JSNF#-MU&"+W*;LGV^6NALKJY<`?VY,@/=)5'Z<5S6GPQM*H*#'TKK+.TM
MP!^[7\JZY47):L\7$8N%%_#,,<#[1BJUS?03Q2--JH$BC`6XC
M:0M]"":TX+>TV_-$GXBJ=Y;VYSM1,>PK&-!EA\5
M2FK\J3.P\/R62:@J
MU2J:Z*6)J4]%JCGK82E5U>C/(<$'!!!'8T\`UZIQ@_G6
M5-X'L9,F">:(^APPKNACZ;^+0\RKEM5?!J<(HJ0"NK;P''=+L2&BM%9QT:3YC^M:Z@`8`QBN2IF72"^\]"ADG6K+[CG=(\(V6G%9I_\`
M2;@;4J3J.\G<]RC0IT8\M-6$---.---9FY&U0L*G:HFZ4
M#*LE4Y:O/5*;O0!FSUC79ZUKW)P*Q+MNO-,1C73=:K20!BI]5!_04^[?DU<$
M8:")C_SS7^0KT,N=IL\3/+JC'U,HVZ]Q4;6RGM6F\8!..E0,E>TCYE5&9E:[K561:=D=-.JS(DMU]*KM;KZ5JR+5=EI\B.R%5F<;<>E-^S#TJ\
M4HV5/LT;>U946V'I5B.S4]JF5:LQK5*FC*=5D*6"GJ*LII:'M5J("KL2^U/E
M1Q5,1-=2BFDQD=*D&CQ?W:U$'M4RJ?2BR.26*J=S'_L:+^[36T6''W:W-OM0
M4I:$_6ZG:,4'5S"(G-:EF,"J*"M"WXIHYJS
MNC5@;IS5Z-SBLV%JN1M421Y-2.IIQW;J,;CBI'OFQU[5F[Z:SG%8\B;)C.:T
M3)+FX+9^8UR6O1K<###-;\SG!K"OSDFM5%6L=N#NIJ1QLUH48XZ5&L>#TK9N
M(P3TJHT0SQ7+*BD]#Z6%9M:D<2UC25D7;$8-:R5E6M:2=*3.2OK(L`T^H@:7-2]%.KF/2N-VCWIVT44M%Q7#:*4"BEHN*XHI[)TN9%Z`5,NI3+T5/Q!_QJG13]A3[$^VJ=S376KE>B1?D?\:F7Q%>+TCA_
M[Y/^-8]+1["GV#V]3N;:^*+X=(K?_OEO\:D'BV_'_+*V_P"^6_QK!HH^KTNP
M>WJ=SH1XQU$?\L;7_OEO_BJD7QIJ0_Y8VO\`WPW_`,57-TM+ZO2_E%]8J_S'
M2CQQJ8_Y86G_`'PW_P`53QX[U0?\L+/_`+X;_P"*KEZ6CZO2_E#ZS5_F.J'C
M[51_R[V?_?#?_%4\?$'5A_R[V7_?#_\`Q52(9]`?\
M:S**/JM'^4/KV(_G9/+
MI65JCN7#J,Q_AC_(_P"-,-[(?X4_(U6I:V1S>SCV)CH7\J86+=0*92T[L
M:BD-*`TTVZ'UJ2G4797,T0?9(SW;\Z3[)'ZM^=6**+L.>7R_E5>BBX>S
MCV)S<.>H6HRY/I3:*=V59(0H&ZYIAMT;J6J6BG=E*36Q7^Q1G^)_SI?L4?\`
M>?\`,58I:+L/:2[D`M(QW;\ZE1`G3-.I:.9BE-HHNR'KN2>?:DS24M`!1113N`8HVBEHHN%Q-
M@]Z-@]Z=11<+L9Y8]31Y2^II]+1<+LC\E?4TGD+ZFI:6BXK4?9T]6
MJ6BF+F9#]F3U:C[*GJWYU-10/GEW(/LJ>K?G1]DC_O-^=3TM`<\NY7^QQ^K?
MG1]BC_O/^8JQ2T"]I+N5?L,7]Y_S%'V&+^\_YBK5%`_:2[E7[!%_>?\`,?X4
MG]GQ?WG_`#'^%6Z*`]I/N5/[.A_O/^8_PI/[.A_O2?F/\*N44![6?>?\*<\/?\`/YJG
M_?V/_P"(I?\`A3GA[_G\U3_O['_\17H5%/ZW7_F8?4Z'\J//?^%.^'O^?S5/
M^_L?_P`11_PIWP]_S^:I_P!_8_\`XBO0J*/K=?\`F8?4Z'\J//?^%.^'O^?S
M4_\`O['_`/$4O_"GO#W_`#^:G_W]C_\`B*]!HH^MU_YF'U+#_P`J//O^%/>'
M_P#G\U/_`+^Q_P#Q%'_"GO#_`/S^:G_W]C_^(KT&BCZW7_F8OJ6'_E1Y]_PI
M_P`/_P#/YJ?_`']C_P#B*/\`A3_A_P#Y_-3_`._L?_Q%>@T4?6Z_\S#ZEA_Y
M$>??\*?\/_\`/YJ?_?V/_P"(I?\`A4'A_P#Y_-3_`._L?_Q%>@44?6Z_\S#Z
MEA_Y$>?_`/"H/#__`#^:G_W]C_\`B*/^%0>'_P#G\U/_`+^Q_P#Q%>@44?6Z
M_P#,P^I8?^1'`?\`"H?#_P#S^:G_`-_8_P#XBC_A4.@?\_FI_P#?V/\`^(KO
MZ*/K=?\`F8?4L/\`R(X#_A4.@?\`/YJ?_?V/_P"(H_X5#H'_`#^:G_W]C_\`
MB*[^BCZW7_F8?444?7*_\S#ZAAOY$<'_`,*FT'_G[U+_`+^)_P#$4?\`"IM!_P"?
MO4O^_B?_`!%=Y11]H_P#?Q/\`XBNZHH^N5_YF']GX;^1'"_\`"J="_P"?O4?^_B?_`!%'_"J=
M"_Y^]1_[^)_\17=44?7*_P#,P_L_#?R(X7_A5.A?\_>H_P#?Q/\`XBE_X55H
M7_/WJ/\`W\3_`.(KN:*/KE?^9A_9^%_D1PW_``JK0_\`G[U'_OXG_P`11_PJ
MK0_^?O4?^_B?_$5W-%'URO\`S,/[/PO\B.&_X55H?_/WJ/\`W\3_`.(H_P"%
M5:'_`,_>H_\`?Q/_`(BNYHH^N5_YF']GX7^1'#?\*KT/_G[U'_OXG_Q%'_"J
M]#_Y^M1_[^)_\17#_Y]HX__A6^C_\`/S??]_$_^)H_X5OH_P#S\WW_`'\3_P")KL**/KN(
M_G8?V7@_^?:./_X5OH__`#\WW_?Q/_B:/^%;Z/\`\_-]_P!_$_\`B:["BCZ]
MB/YV']EX/_GVCC_^%;Z/_P`_-]_W\3_XFC_A7&C_`//S??\`?Q/_`(FNPHH^
MNXC^=A_9>#_Y]H****Y3O"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`H
MHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BB
MB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****
M`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`
M****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`H
MHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BB
MB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****
M`"BBB@`HHHH`****`"BBB@`HHHH`***.U`!1165KNO66@6)N;Q^IPD8^\Y]!
M3C%R=EN3*2BKRV-3)-P4
M#UYJKI'B6UUIO]$@N3&.#*T>%!],YIN:W
M;Z#IYN[@,PW!55>K$]OTJ];7"75M'/$P:.10RD=P:.5VYK:#4XN7+?4FS1FH
M+J?[-`TOE22[?X(QEC]!67I7BC3]7OI+.W:19XP2R2)M/!P?RIJ,FKH4JD8M
M1;U9MT4@-1W$\=K;R3RL%CC4NQ/8"I+;MN2T5B:5XEM-:;%G#%7Z
MFMG/%.47%V9,)QFKQ>@N12UBR>([./Q%%HI#>>ZD[L<`XR!^(K8H<7'=!&<9
M7Y7L.HK'U?Q#;:+\UY#<"+@>:L>5R>VC;*L/P/-:=2XN+LRXRC)7B[BT
M55O[V'3K*:[G.(HEW-@5!HVK0ZSID=]`K*CY^5NHP<4^5VYN@<\>;EOJ:-%%
M%24%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!111
M0`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`
M!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%
M%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4'
MI12'I0!%<3QVUO)/*P6.-2S,>P%>`>(]?F\1:U)=R$B$';#&?X%_Q]:]1^)N
MHM9>%6@1]KW4@BX[KU/\L?C7BJ??KV,MHKE=5GB9I7?,J2V-.WZ"NO\`"D9A
MCO\`4V9U2U@(!5L;F/05R$'2O1K#0[B3PM9VD1\M;IC=7,K=%0=![YX/X5T8
MJ:C&SZG+AH.4KK6R-0V=A<36-O-:7-S,UJC;P_RHOOS]:S=TM(_M-A+OA
M63RG7=G:V,]:N:I+J206L=C#*4FLX@\D:$GC/&1]:J3P26/A40W*&.6:XWJC
M=<`8Z5P4W)6:?R.RLH-2BX[+:Z`?#^HY_P"?:3_T$US'@J_M]-\&-<3G
M`$S`*!DL>,`#N374:Y_R+^H_]>TG_H)KS#3[35(?#MIK-C*9%LYW+08R!T^;
M'?C@UQX>"G1<6^J/2Q4W3KJ25]'^:/0]-TZ>YNO[5U,?OR/W$'58%/\`[,>Y
MK;Q63H&O6VNZ>L\)VR+Q+&>J'_"M5FVJ23@`9-)MT*G>\1=(B9@94::9AVC7M^)P/SK-\!:SNTV?3;LE)K,D
M_-V3_P"L<_I6KID;75E?ZK*,/>J3&#_#$`0H_'K^-)T_95'?IL/VOMZ22Z[F
M=\-<'P_-Z_:&_D*[%F55)/`'4FN,^&A_XD5R/2Y/_H*UL^*+J6#19(K==US=
M,+>)0>26X_EFGB$Y5VO,,+)0PJ?9'%Z_%+`;/Q7%DO)=%L=MF?D_,+^M>E6M
MPEW:PW$9RDJ!U/L1FN4U*/5;WP])I8T'9'Y01#]I0XQTX_"G?#W4/M.AM9R9
M\VTD*$'T/(_J*NJN>DI?RNWR,J#5.OR])*_S)?B#_P`BK)_UU3^=:OAO_D6M
M-_Z]D_E65\0O^15E_P"NJ?SK6\-_\BSIO_7LG\JA_P"[+U9K'_>Y>B,CQCHD
M=Q8/J=J/)OK0>:)(^"P'4&KOA+6FUO1$GEQY\9\N7'22'
MY%A@G[F*2C]I:_(WO$$8OS:Z2?N
MW4F9@/\`GFO)_7:/QKG_``',]A?ZEH4['="Y>,'OV/\`0U?%YJ$_B6]N[33_
M`+7#`HM483!,'[S=?<@?A6!JL][I?C*RUF[LC:),P1QY@?(Q@]/8C\JTI1O!
MTNZO\S"M42J*LNCM\CTT=**0'-+7">J%%%%`!1110`4444`%%%%`!1110`44
M44`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!111
M0`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`
M!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%
M%%%`!1110`4444`%%%%`!1110`4'I10>E`'F/Q>)\G2AVW2?^RUY>GWJ]F^*
M&GF[\,+X-O,B]"!P/PZ?G7)>$`MC9ZEK;@
M_P"C0^7"?^FC\#_/O6CX.D^V0ZAH\C#;=Q%H\]G'^?TK'$^]/G_EM_P36A[L
M.3K+^D;&L:K>V7]G16UP\:&S1B!CD\_X5#J$C:GH%K>NQ::!S#*3WSR#4?B=
M&CN]/1@0RV:`@]CDT:`PG^UZ:Q`6YB.P'^^.16,4E3C42VU"C?V?<(\=P9&(CD0C<#CUZUVF`
M?K1\OM7CJK:FX6\SZ*5%NJJE]M#SS5])O/">L+K.CHSVDC8F@7)QGJ,>GIZ5
MHZSXK@NO#LHLHK@W,Z;/+\ILIGJ3QZ5V>!28'053KJ5G-7:(6&<.94Y63Z&?
MI,]I-I\2639BB4(`5((P/0UQ_CXJ;RPFLP[WULW(2,G`X(S^/\Z]``%&!4TZ
MO)4YTBZM'VE+DO8PXO%%@VG+TU+@&JG6<^7FZ$PPRASQRP,\H=&>-@&R`,9Q[5?N=;MKOQ=9EO-%E:H["4Q-M,AX]/3-=E@>M
M)M'M3E64IN=MR88>4*:IJ6B\O^",\Q/*\W/[O;NW>U><:7?1Z/XWO)HDG.FW
M).7$3%03SGIV)(KTL`48%33J\B::O%=#2V@1R40^5&BE
MF=O?'O6]B@BE[711MH7[&SU=)30!VIU1.2E)M(UIQ<8J+=[!1114EA1110`4444`%
M%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`44
M44`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!111
M0`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`
M!1110`4444`%%%%`!1110`4444`%%%%`!1110`4'I11VH`@N[:.\M);:90T4
MJ%'7U!&*^?-8\CM<7.P'@_P@_A3-
M&N)=/U.VN@C_`+IP3@=N_P"E:DD_BCPB@M/,62T0XCD\L.N/J1D?0T1>-=;?
MK+#^$*UZ"YW%J*33\SRY\BDG-M->78T_%%W#J&J0RVK>8@A`)4'@Y)Q^M9MI
M++:7D-PJ-F-PW2KL7BW5F',D7_?L5.OB;5I6"1LC,>!MB!-9*,X0Y+*WJ$I4
MIU.>[N_(I:SY!U2:2V<-%(=XQV)Y(_/-=7X3TEK6W-Y.F)91\H/9?_KU!I6@
M37=R+_5GWR=1'G^?^%=8`!T'X5R5Z_N*G$[L)A7SNK)>AEZO=36L^G^7(RI)
M4KY6#D$#(R.>M7[VP-Y/:OYA3[/
M+YH`&=QP1S^!-,N-+\^_-UYQ5C`T&T+P`3G/UKG3C;4[)QG=M%?^WK:.",[9
MY?W<;,P49&_A]:*-\?*GR@A?YU6ETB\BN/\`1B^8
MH(XH69$96V]"<\CG_P"M6B-&PQ...15'S!_NY'UXXJY/IV^
M\%Y!(8;D)Y9;&5=>P8>WX=:SK;2[BYMYENU:&[^T+,TZD,LA4C!`[#`Q@_KU
MH7*UJ-\ZEH7CK4`M99UAG;RG=&15&1L^\>N,?C5>>_"2W=QYTP@^S0R+L`)7
M<6&0#]!0V@!HFC-W*5>221@54Y+]>,=NWUI[:'N@DA:YQ9([UL/.+D`&```9P%SD^P%2G3@='.GAV56B,9<\GD[HT/]Y9I
MF.U]?D.3)(8%^SL)`F"69P&08'(Q_.M5=9A,$KB"M/
MDL&DL(K7S2/+*'?CD[2"/U`JL^BEI)6%RX66;S73:,'Y0N/T%.\7N)1J1VU)
M$UNWEFMXHHIW,\2S*53@*3C)J*/Q%;RJK);71WQO(OR#Y@IPW?J*DLM&^Q/"
MZ3NYBMQ;CX'[TF.M6^VU94E:.X
MV;7"\#=]W/U]J4ZO`JO(4D%NH)\_`V'!P1USU]JJ+X>VI`@NY-L"Q*@*@XV'
MC\^]2+H0%M/:&[F-K)DK&0/W9)SP<9X/(HM`=ZM]B2'7+:=HE"2AI)3$.`1N
M"[NH..@[4R/78IY;5(;:=Q<*Y!`'R[6VG//K3Y-*EE2W+WLC302^8LA11_"5
MQ@#'0U';:']E-JT=S(6@$@RRCY@YR0?Q%'N!^]'P:M&;5&C6>Y"2"\ECDC4QDA00ZEMV"
M#W!/!IPT,_;/M)NY68,Y&Y5/##&,]<#M1:`?O+6L1Z-JN_3=.BE\R:YDMHY'
M;@GGC<><]?2MP=*Q8M`CCBLXC+N6T"",E!N&WT/;/0UM#I4SM>Z+I\R5I"T4
M45)H%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!11
M10`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%
M`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`
M%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4
MA&:6B@"O-90S*5=<@]161/X.TB=BQMPK'J5X_E6_151G*.S)E",_B5SGH_!N
MEQGA&^A8UH6VBV=J/W487Z"M&BFZDI;LF-*G%WBAB1*@X%.I:*@T"DI:*`"B
MBB@`I,4M%`!1110`4E+10`F.:6BB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`*&ALS:!IK,26-K$22>3\HJ_6?H/_
M`"+VF?\`7I%_Z`*T*`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH
M`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`
MHHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"B
MBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`***
M*`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH
M`****`"BBB@`HHHH`****`"BBB@`HHHH`15"J%4`*!@`#@4M%%`!1110`444
,4`%%%%`!1110!__9
`
end
GRAPHIC
4
exhibit_99-12.jpg
begin 644 exhibit_99-12.jpg
M_]C_X``02D9)1@`!`0$`8`!@``#_VP!#``@&!@<&!0@'!P<)"0@*#!0-#`L+
M#!D2$P\4'1H?'AT:'!P@)"XG("(L(QP<*#7J#A(6&AXB)BI*3E)66EYB9FJ*CI*6FIZBIJK*SM+6VM[BYNL+#Q,7&
MQ\C)RM+3U-76U]C9VN'BX^3EYN?HZ>KQ\O/T]?;W^/GZ_\0`'P$``P$!`0$!
M`0$!`0````````$"`P0%!@<("0H+_\0`M1$``@$"!`0#!`<%!`0``0)W``$"
M`Q$$!2$Q!A)!40=A<1,B,H$(%$*1H;'!"2,S4O`58G+1"A8D-.$E\1<8&1HF
M)R@I*C4V-S@Y.D-$149'2$E*4U155E=865IC9&5F9VAI:G-T=79W>'EZ@H.$
MA8:'B(F*DI.4E9:7F)F:HJ.DI::GJ*FJLK.TM;:WN+FZPL/$Q<;'R,G*TM/4
MU=;7V-G:XN/DY>;GZ.GJ\O/T]?;W^/GZ_]H`#`,!``(1`Q$`/P#W^BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"HU@A39MBC79G9A1\N>N/2I**`(U@A39MBC79G9A1\N>N/2A8(4V
M;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]
M*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N/2I**`(U@A39MBC7
M9G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%
M-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N
M/2I**`(U@A39MBC79G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8H
MUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*%@
MA39MBC79G9A1\N>N/2I**`(U@A39MBC79G9A1\N>N/2A8(4V;8HUV9V84?+G
MKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]*DHH`C6"%-FV
M*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N/2I**`(U@A39MBC79G9A1\N>N/2A
M8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=F%'R
MYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N/2I**`(U@A39
MMBC79G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCT
MH6"%-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1
M\N>N/2I**`(U@A39MBC79G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4
MV;8HUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX
M]*%@A39MBC79G9A1\N>N/2I**`(U@A39MBC79G9A1\N>N/2A8(4V;8HUV9V8
M4?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]*DHH`C6"
M%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N/2I**`(U@A39MBC79G9A1\N>
MN/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=
MF%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N/2I**`(U
M@A39MBC79G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+
MGKCTH6"%-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79
MG9A1\N>N/2I**`(U@A39MBC79G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"
M-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'
MRYZX]*%@A39MBC79G9A1\N>N/2I**`(U@A39MBC79G9A1\N>N/2A8(4V;8HU
MV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]*DHH
M`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N/2I**`(U@A39MBC79G9A
M1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*
M-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N/2I*
M*`(U@A39MBC79G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V
M84?+GKCTH6"%-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39M
MBC79G9A1\N>N/2I**`(U@A39MBC79G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ
M2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F
M=F%'RYZX]*%@A39MBC79G9A1\N>N/2I**`(U@A39MBC79G9A1\N>N/2A8(4V
M;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]
M*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N/2I**`(U@A39MBC7
M9G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%
M-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*DHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`H:&S-H&FLQ)8VL1))Y/RBK]9^@_\`(O:9_P!>D7_H`K0H`****`"B
MBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`***
M*`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH
M`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`
MHHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"B
MBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`***
F*`"BBB@!%4*H50`H&``.!2T44`%%%%`!1110`4444`%%%%`'_]D_
`
end
GRAPHIC
5
exhibit_99-13.jpg
begin 644 exhibit_99-13.jpg
M_]C_X``02D9)1@`!`0$`8`!@``#_VP!#``@&!@<&!0@'!P<)"0@*#!0-#`L+
M#!D2$P\4'1H?'AT:'!P@)"XG("(L(QP<*#7J#A(6&AXB)BI*3E)66EYB9FJ*CI*6FIZBIJK*SM+6VM[BYNL+#Q,7&
MQ\C)RM+3U-76U]C9VN'BX^3EYN?HZ>KQ\O/T]?;W^/GZ_\0`'P$``P$!`0$!
M`0$!`0````````$"`P0%!@<("0H+_\0`M1$``@$"!`0#!`<%!`0``0)W``$"
M`Q$$!2$Q!A)!40=A<1,B,H$(%$*1H;'!"2,S4O`58G+1"A8D-.$E\1<8&1HF
M)R@I*C4V-S@Y.D-$149'2$E*4U155E=865IC9&5F9VAI:G-T=79W>'EZ@H.$
MA8:'B(F*DI.4E9:7F)F:HJ.DI::GJ*FJLK.TM;:WN+FZPL/$Q<;'R,G*TM/4
MU=;7V-G:XN/DY>;GZ.GJ\O/T]?;W^/GZ_]H`#`,!``(1`Q$`/P#W^BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"HU@A39MBC79G9A1\N>N/2I**`(U@A39MBC79G9A1\N>N/2A8(4V
M;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]
M*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N/2I**`(U@A39MBC7
M9G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%
M-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N
M/2I**`(U@A39MBC79G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8H
MUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*%@
MA39MBC79G9A1\N>N/2I**`(U@A39MBC79G9A1\N>N/2A8(4V;8HUV9V84?+G
MKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]*DHH`C6"%-FV
M*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N/2I**`(U@A39MBC79G9A1\N>N/2A
M8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=F%'R
MYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N/2I**`(U@A39
MMBC79G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCT
MH6"%-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1
M\N>N/2I**`(U@A39MBC79G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4
MV;8HUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX
M]*%@A39MBC79G9A1\N>N/2I**`(U@A39MBC79G9A1\N>N/2A8(4V;8HUV9V8
M4?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]*DHH`C6"
M%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N/2I**`(U@A39MBC79G9A1\N>
MN/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=
MF%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N/2I**`(U
M@A39MBC79G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+
MGKCTH6"%-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79
MG9A1\N>N/2I**`(U@A39MBC79G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"
M-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'
MRYZX]*%@A39MBC79G9A1\N>N/2I**`(U@A39MBC79G9A1\N>N/2A8(4V;8HU
MV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]*DHH
M`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N/2I**`(U@A39MBC79G9A
M1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*
M-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N/2I*
M*`(U@A39MBC79G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V
M84?+GKCTH6"%-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39M
MBC79G9A1\N>N/2I**`(U@A39MBC79G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ
M2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F
M=F%'RYZX]*%@A39MBC79G9A1\N>N/2I**`(U@A39MBC79G9A1\N>N/2A8(4V
M;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%-FV*-=F=F%'RYZX]
M*DHH`C6"%-FV*-=F=F%'RYZX]*%@A39MBC79G9A1\N>N/2I**`(U@A39MBC7
M9G9A1\N>N/2A8(4V;8HUV9V84?+GKCTJ2B@"-8(4V;8HUV9V84?+GKCTH6"%
M-FV*-=F=F%'RYZX]*DHH`C6"%-FV*-=F=F%'RYZX]*DHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`
M"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*
M***`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HH
MHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`****`"BBB
M@`HHHH`****`"BBB@`HHHH`****`"BBB@`HHHH`*#10:`"BL[5=1FLECCM+8
MW%S*<(F<`>Y/I26JZDZ[KJ902,[88PH!]/FS3L*^MB]/<0VL+37$J11*,L[M
M@#\:Y&_^*'A6QF:'[>9Y`.!"A()],USWBWP3XUURWNP->@DM68M':"/!([#=
MZUYC'\(_%SE`W#YBV!185V>B6_QTLGO'BETMQ&&QE)?F'X8KO++Q#<:
MQ8PW>F6B^3*N0TS<_P#?*_XUY]8?!$#4VN;V]B$;$$K$"2?7KBO6;"QM]-L8
M;.UC$<,2A54?YZT@5SCK36?&]SK$T%UHT=K8IG;.@W%O3`)JU'?^+AKTD!M8
M3IHB#+<-$=Q?TP"*["BGLPLT#%9DXE@<8>,^X_J.*+`F:5%%%(
M84444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!
M1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%
M%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`444
M4`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110`4444`%%%%`!1110
M`4444`%%%%`!1110`4444`%%%%`!1110`4444`%!HH-`%2[C.])EX9>C8Z?7
MVIRWL7`D_=GU/3\ZLU"]M$X^[CZ4`2@@C(.0>]+6:UA-"=UM*5]E./TZ5'_:
M5U;'%S!O]"ORG\CP?SH%OX5=H&%%%%`!111
M0`5S>L>&!-*M]IDAM;V/E6C./\_0\&NDHH$U`[5E/RQR
M_B?NM['\*ZFLC7-"@UFWVN$W@8^9 |