Exhibit 99.1

VYNE Therapeutics Reports Third Quarter 2021 Financial Results and Provides Business Update
Lead BET Inhibitor Candidate, VYN201, Demonstrates Potent Anti-Inflammatory Effect in Preclinical Studies
VYN201 Expected to Enter Clinic in 2022
Phase 2a Results for FMX114 in Mild-to-Moderate Atopic Dermatitis Expected Early Q1 2022
Process for Sale of Topical Minocycline Franchise Continues to Advance
Conference call and Live Webcast Today at 8:30 am Eastern Time
BRIDGEWATER, N.J., November 10, 2021 -- VYNE Therapeutics Inc. (Nasdaq: VYNE) (“VYNE” or the “Company”) today announced financial results for the third quarter ended September 30, 2021 and provided a business update.
“The third quarter marks an important turning point for VYNE, as our company is now fully engaged in the development of next-generation therapeutics designed to address multiple immuno-inflammatory diseases,” said David Domzalski, CEO of VYNE. “Based upon a growing body of preclinical data, we believe the evolving therapeutic profile for the BET inhibitor drug class represents a potential breakthrough in the treatment of immuno-inflammatory diseases. Our lead candidate, VYN201, is demonstrating a strong ability to effect multiple pro-inflammatory pathways while also appearing to be well tolerated in animal models. As we move toward 2022, creating shareholder value remains front and center for our company and we look forward to our Phase 2a data readout for FMX114 in Q1 2022 and advancing our lead BET inhibitor candidates.”
Pipeline Update
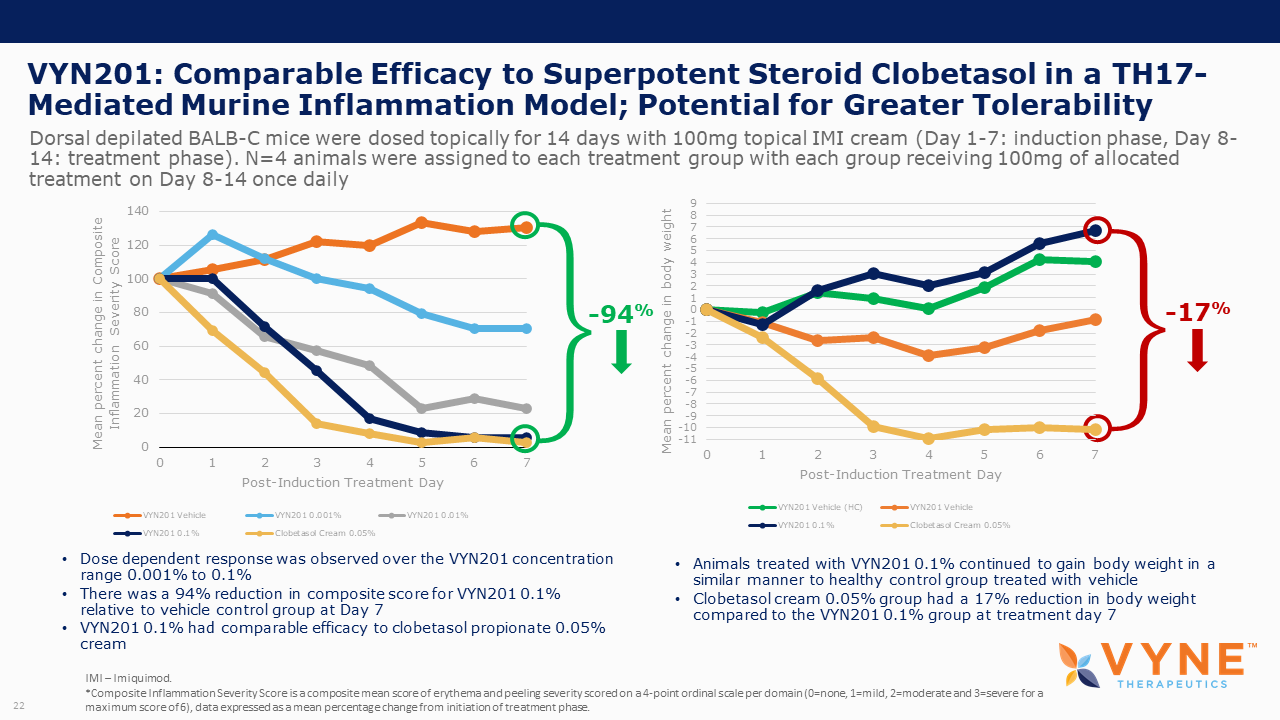
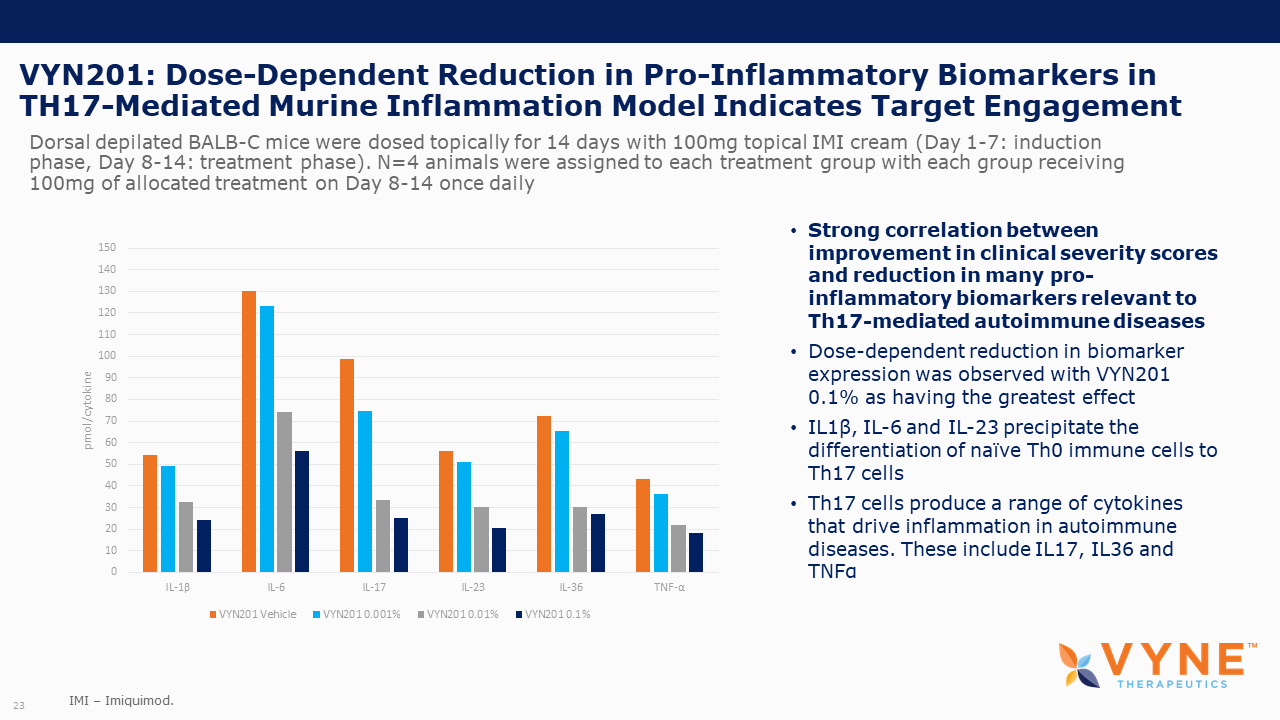
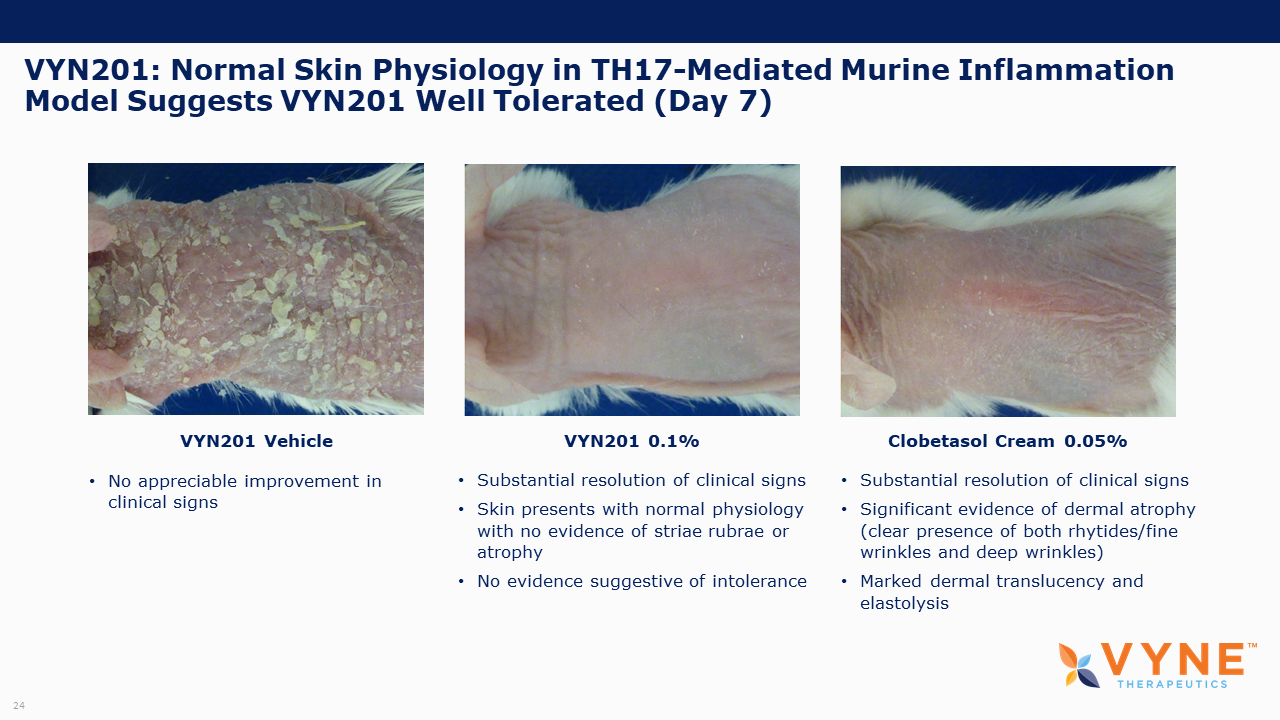
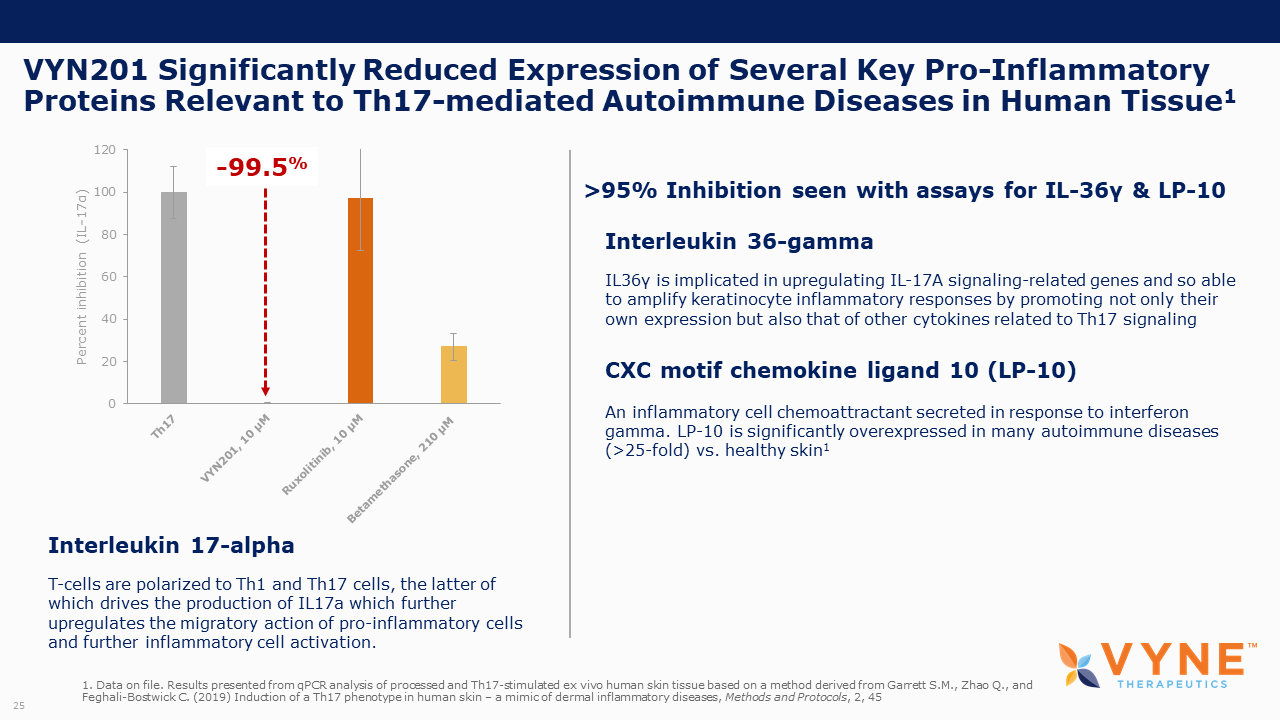
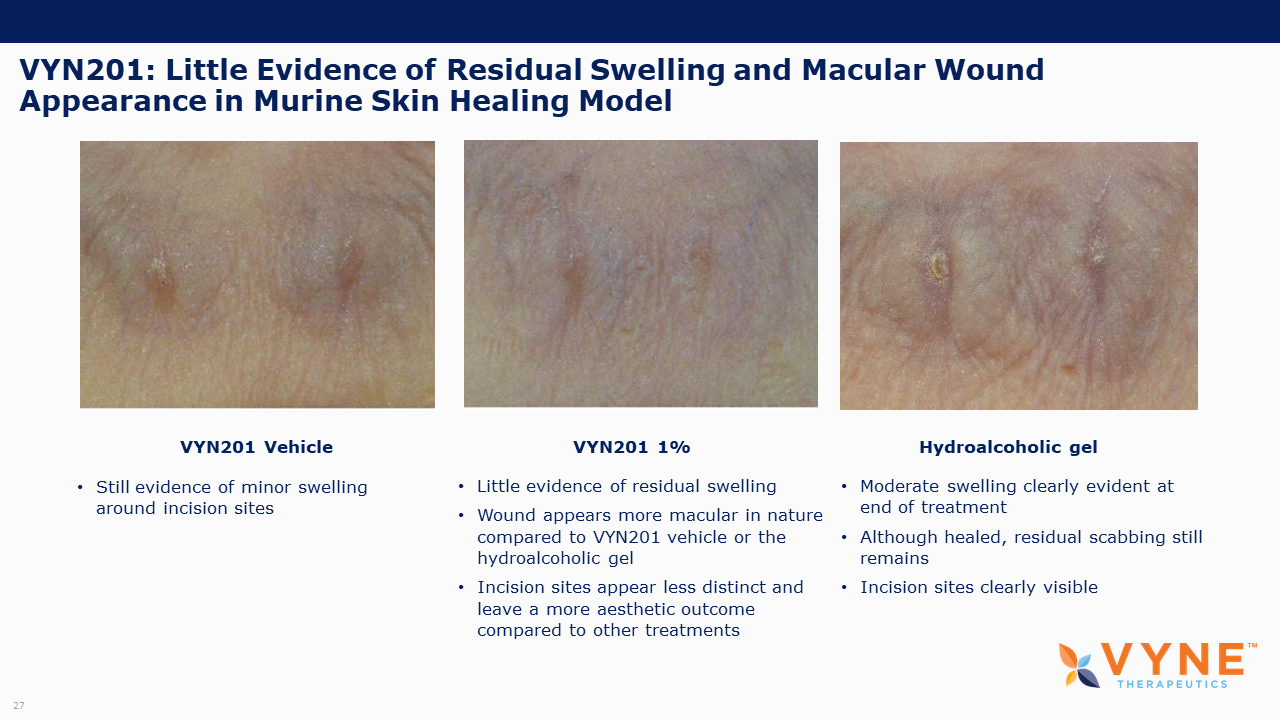
VYN201, Topical BET Inhibitor Program: On October 26, 2021 and November 4, 2021, VYNE announced data from preclinical studies showing that its pan-bromodomain BET inhibitor, VYN201, significantly reduced the expression of several key pro-inflammatory cytokines relevant to Th17-mediated autoimmune diseases and demonstrated improvement in reducing fibrotic tissue mass and overall skin repair outcomes. Key highlights from the preclinical studies include:
| • | VYN201 exhibited anti-inflammatory effect similar to super-potent glucocorticosteroids. | |
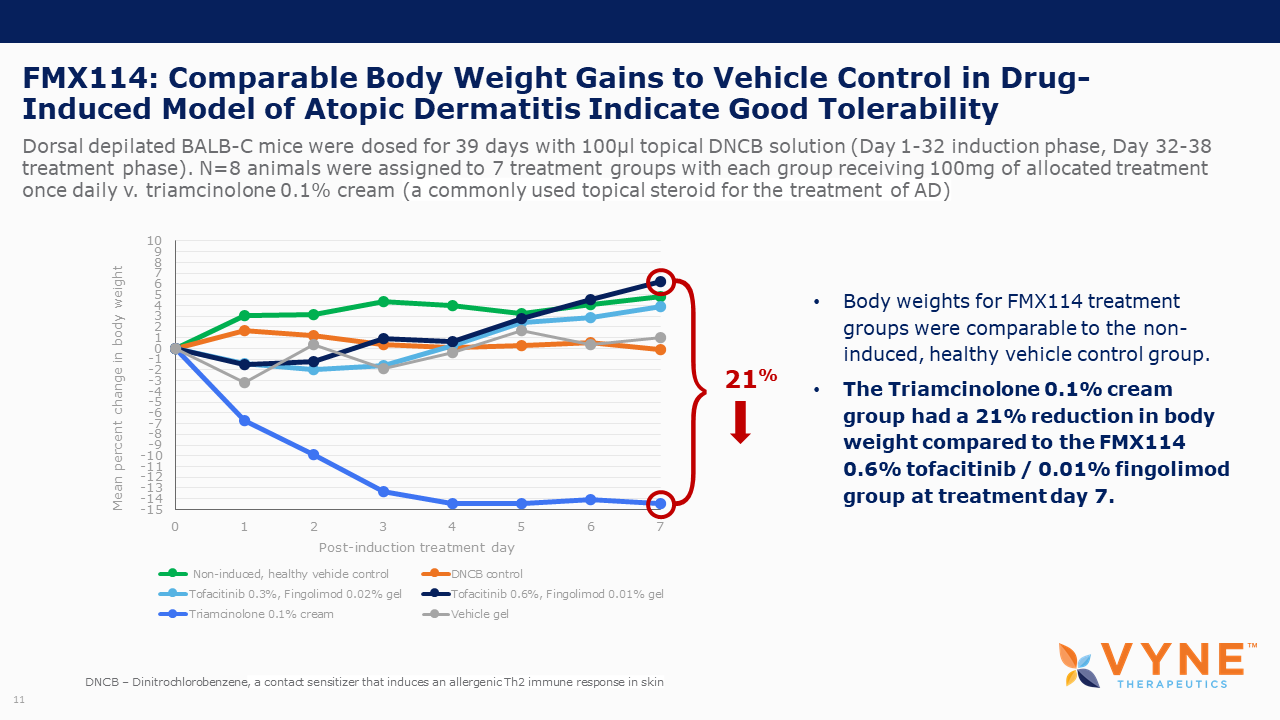
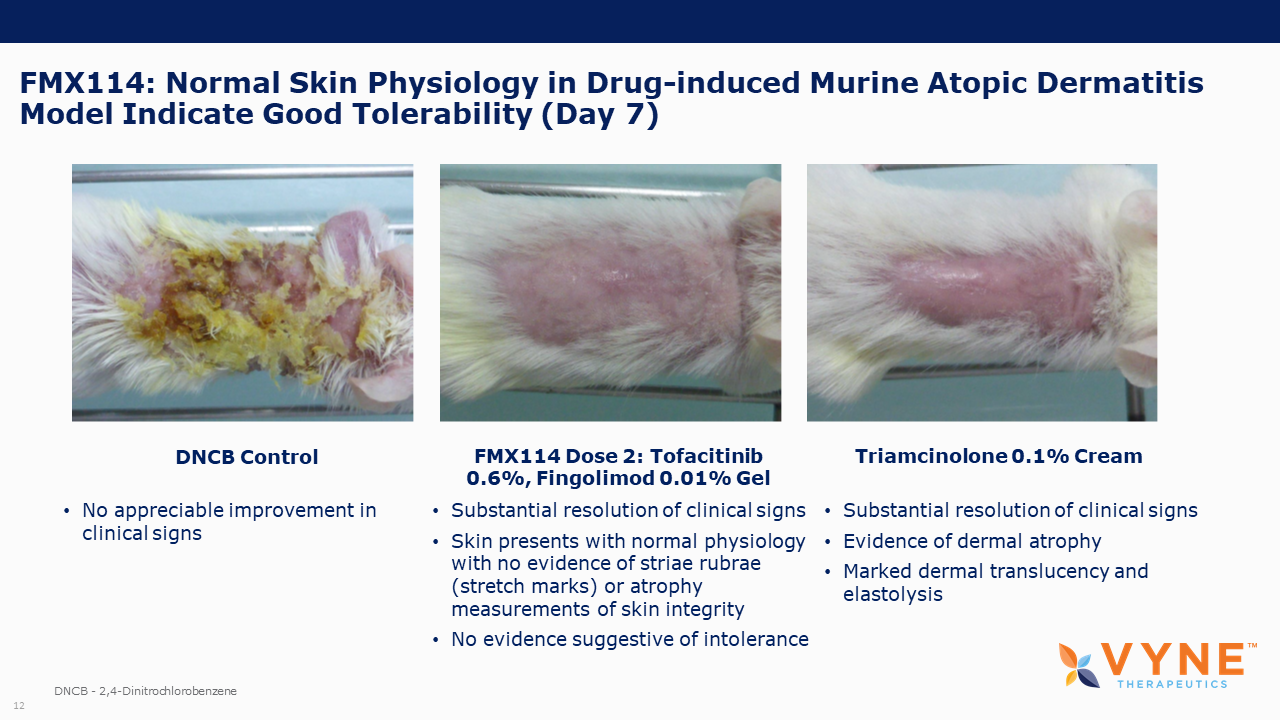
| • | VYN201 appeared well-tolerated in mice, as seen through animal body weight and skin condition. | |
| • | VYN201 also demonstrated stronger inhibition of key Th17 cytokines in ex vivo data with human skin tissue when directly compared to JAK1/2 inhibitor, ruxolitinib. | |
| • | VYN201, demonstrated improvements in reducing fibrotic tissue mass and overall skin repair outcomes with no negative impact on healing time, supporting the continued progression of the VYN201 development program. |
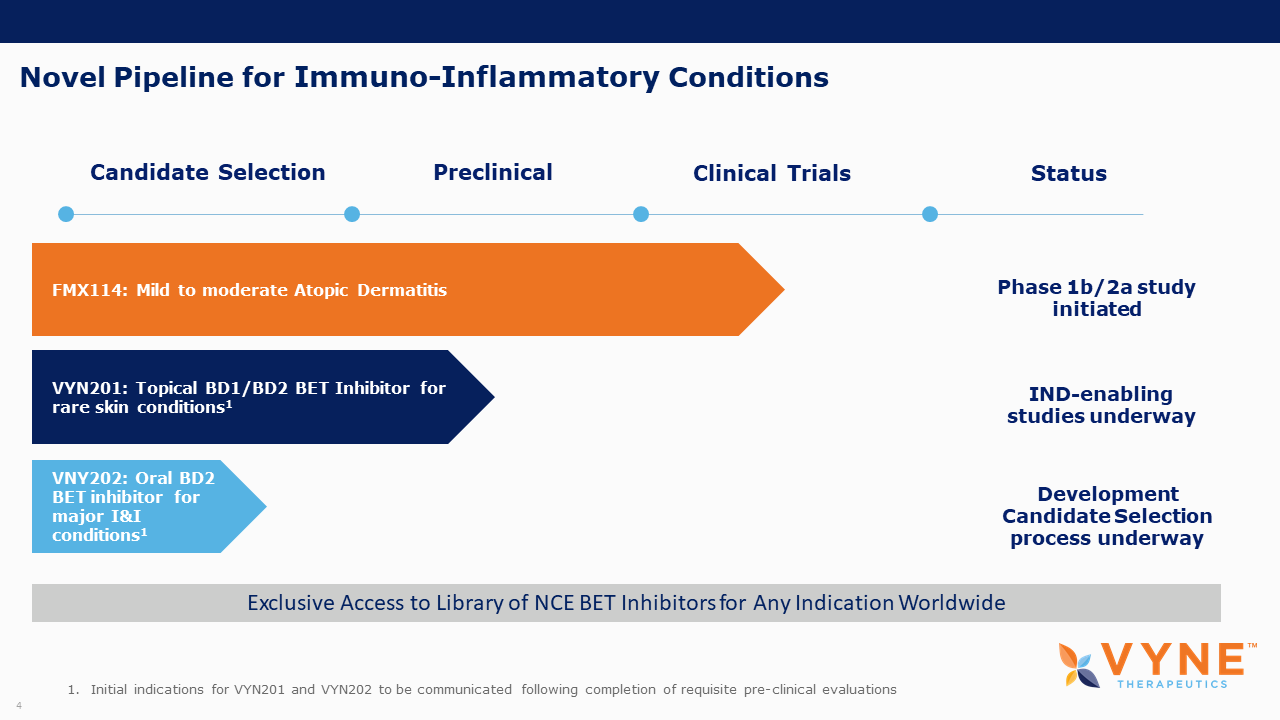
Following the completion of the prerequisite IND-enabling non-clinical safety assessments, the Company intends to communicate the initial indication it will be pursuing for VYN201 and expects to enter the program into the clinic in 2022.
VYN202, Oral BET Inhibitor Program: VYN202 is an orally delivered, first-in-class BET inhibitor that is highly selective for Bromodomain 2 (“BD2”). By selectively inhibiting BD2, the Company believes VYN202 could have a more targeted anti-inflammatory effect with an improved benefit/risk profile. Upon the selection of a lead candidate, VYNE intends to exercise its exclusive option with In4Derm Limited and commence an IND-enabling non-clinical safety program.
FMX114 for Atopic Dermatitis (AD): In October, VYNE announced the first patient had enrolled in its Phase 1b/2a study comparing the safety and efficacy of FMX114 gel with vehicle gel in patients with mild-to-moderate AD. Topline results from the Phase 2a portion of the study are anticipated early in the first quarter of 2022.
Strategic Process to Sell Topical Minocycline Franchise
The Company continues to execute its process to divest its topical minocycline franchise, including AMZEEQ, ZILXI and FCD105 (the Company’s Phase 3 proprietary novel topical combination foam formulation of minocycline and adapalene for the treatment of moderate-to-severe acne vulgaris), as well as the underlying Molecule Stabilizing Technology (MST) platform specific to this portfolio.
“We believe our minocycline franchise is a high-quality commercial platform that has significant value,” said Mr. Domzalski. “We are encouraged by the level of interest we have received and will provide additional details as we continue to progress the sale process.”
Recent Highlights:
| • | On October 5, 2021, we announced that the first patient was enrolled in Cutia's Phase 3 study in China evaluating AMZEEQ for the purposes of seeking regulatory approval in China. |
| • | On October 19, 2021, we announced that the first patient was enrolled in our Phase 1b/2a clinical trial evaluating FMX114 for the treatment of mild-to-moderate atopic dermatitis. We expect topline results from the study early in the first quarter of 2022. |
| • | On October 21, 2021, we announced the formation of a scientific advisory board ("SAB") composed of leading scientists and academics specializing in immunological and inflammatory diseases. The SAB will provide scientific expertise and guidance to the VYNE management team and Board, as the Company progresses its pipeline of innovative treatments for immuno-inflammatory conditions. |
| • | On October 26, 2021, we announced preclinical data showing that VYN201 significantly reduced the expression of several key pro-inflammatory cytokines relevant to Th17-mediated autoimmune diseases in an animal model and an ex vivo human tissue study. |
| • | On November 4, 2021, we announced preclinical data showing that VYN201 demonstrated improvements in reducing fibrotic tissue mass and overall skin repair outcomes with no negative impact on healing time, the results of which support the continued progression of VYN201 development program for autoimmune skin diseases. |
| Financial Performance (in thousands, except per share amounts) |
Three Months Ended September 30, |
Nine Months Ended September 30, |
||||||||||||||
| 2021 | 2020 | 2021 | 2020 | |||||||||||||
| Total Revenues | $ | 4,086 | $ | 3,269 | $ | 12,463 | $ | 16,707 | ||||||||
| Net Loss | $ | (21,285 | ) | $ | (24,714 | ) | $ | (61,759 | ) | $ | (232,387 | ) | ||||
| Diluted Net Loss per Share | $ | (0.41 | ) | $ | (0.59 | ) | $ | (1.22 | ) | $ | (7.98 | ) | ||||
| Adjusted Net Loss* | $ | (18,880 | ) | $ | (22,094 | ) | $ | (55,011 | ) | $ | (78,169 | ) | ||||
| Adjusted Diluted Net Loss per Share* | $ | (0.36 | ) | $ | (0.53 | ) | $ | (1.09 | ) | $ | (2.68 | ) | ||||
| Adjusted EBITDA* | $ | (15,378 | ) | $ | (20,934 | ) | $ | (49,318 | ) | $ | (74,944 | ) | ||||
* See "Note Regarding the Use of Non-GAAP Financial Measures" elsewhere in this earnings release.
Cash and Cash Equivalents
As of September 30, 2021, VYNE had cash, cash equivalents and restricted cash of $52.9 million. On August 11, 2021, the Company paid its lenders approximately $36.5 million in respect of its outstanding indebtedness and the Credit Agreement was terminated. See Note 1 to our unaudited interim condensed consolidated financial statements included in VYNE’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2021 for additional discussion on liquidity.
Financial Results for the Third Quarter Ended September 30, 2021
Revenues
Revenues totaled $4.1 million for the three months ended September 30, 2021 compared to $3.3 million for the three months ended September 30, 2020. For the three months ended September 30, 2021, our revenue consisted of $4.0 million of product sales and $0.1 million of royalty revenue. For the three months ended September 30, 2020, revenues consisted of $2.9 million of product sales, and $0.4 million of royalty revenue.
Cost of Goods Sold
Cost of goods sold was $1.0 million and $0.4 million for the three months ended September 30, 2021 and 2020, respectively. The increase in cost of goods sold was primarily due to an increase in sales volume.
Our gross margin percentage was 73% for the three months ended September 30, 2021. Our gross margin percentage of 87% for the three months ended September 30, 2020 was favorably impacted by product sales with certain materials produced prior to FDA approval and therefore expensed in prior periods.
Research and Development Expenses
Our research and development expenses for the three months ended September 30, 2021 were $7.0 million, representing an increase of $0.4 million, or 5.4%, compared to $6.6 million for the three months ended September 30, 2020. Employee-related expenses, including stock-based compensation, increased by $1.0 million. Expenditures for FMX114 and our BET inhibitor programs increased by $1.8 million, including fees due to In4Derm upon the execution of the Option Agreement and in connection with the exercise of the Topical BETi Option. The increases were offset by a decrease in clinical trial and manufacturing expenses due to the completion of FCD105 and serlopitant clinical trials and the product launches of AMZEEQ and ZILXI during 2020.
Selling, General and Administrative Expenses
Our selling, general and administrative expenses for the three months ended September 30, 2021 were $13.8 million, representing a decrease of $5.9 million, or 30.0%, compared to $19.8 million for the three months ended September 30, 2020. Employee-related expenses decreased by $2.0 million primarily due to lower headcount in 2021. Commercial operations and marketing expenditures decreased by $2.5 million as a result of the strategic shift of the business announced in August 2021. The balance of the decrease was primarily due to corporate costs incurred during 2020 following the merger between Menlo Therapeutics Inc. and Foamix Pharmaceuticals Ltd. that were eliminated or reduced in 2021. See Note 1 to our unaudited interim condensed consolidated financial statements included in VYNE’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2021 for additional information regarding the merger.
Conference Call and Live Webcast
VYNE management will host a conference call and live webcast today, Thursday, November 10, 2021, at 8:30 am Eastern Time to discuss the financial results and provide a business update.
| Toll Free: | 855-327-6837 |
| International: | 631-891-4304 |
| Conference ID: | 10017178 |
| Webcast: | https://viavid.webcasts.com/starthere.jsp?ei=1512469&tp_key=ee68066700 |
A replay of the call will be archived on the Company’s website at www.vynetherapeutics.com promptly after the conference call.
Note Regarding the Use of Non-GAAP Financial Measures
The Company has provided certain non-GAAP financial information as additional information to measure operating performance, including, among others, Adjusted Total Operating Expenses, Adjusted Net Loss, Adjusted Diluted Net Loss per Share and Adjusted EBITDA. The Company believes that its presentation of such non-GAAP measures provides useful information to management and investors regarding its financial condition and operations. Specifically, these measures exclude, among other things, share-based compensation. The Company does not believe that such expenses accurately reflect the Company's ongoing operations, and the Company does not expect to incur similar expenses in future periods. These measures are not in accordance with, or an alternative for, generally accepted accounting principles in the United States (“GAAP”) and may be different from similarly titled non-GAAP measures reported by other companies. The Company has provided required reconciliations to the most comparable GAAP measures elsewhere in the document.
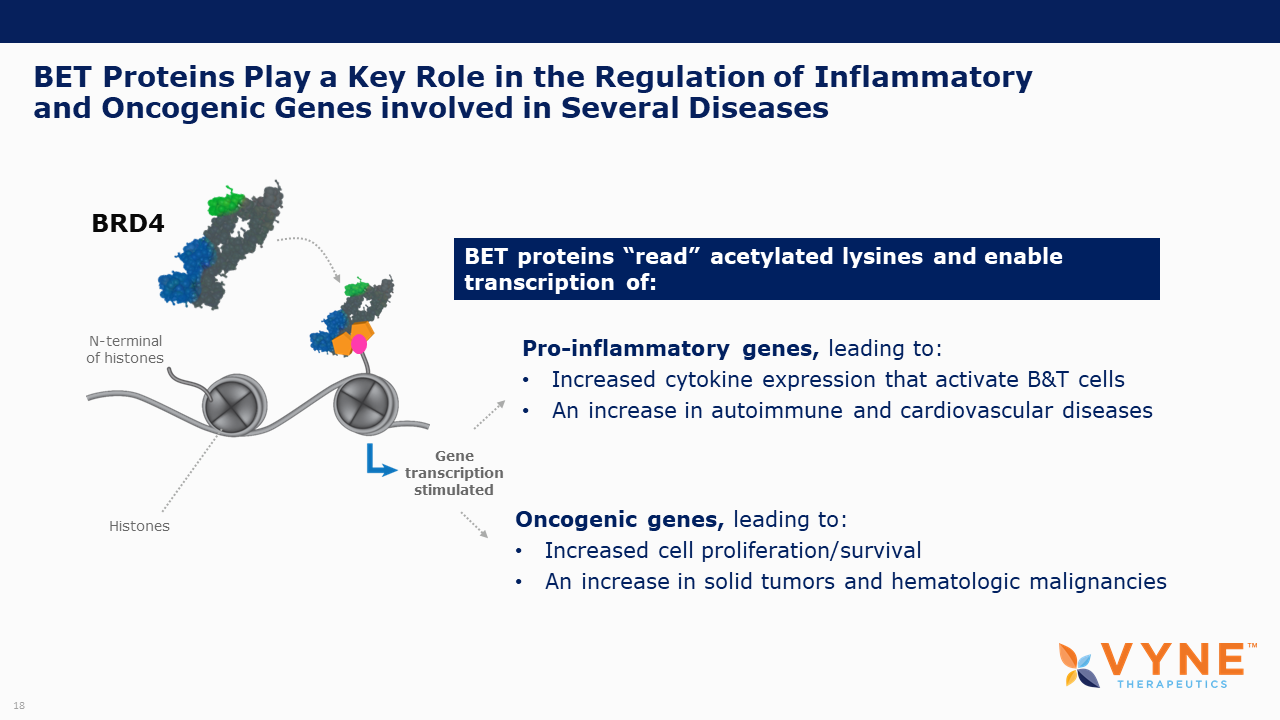
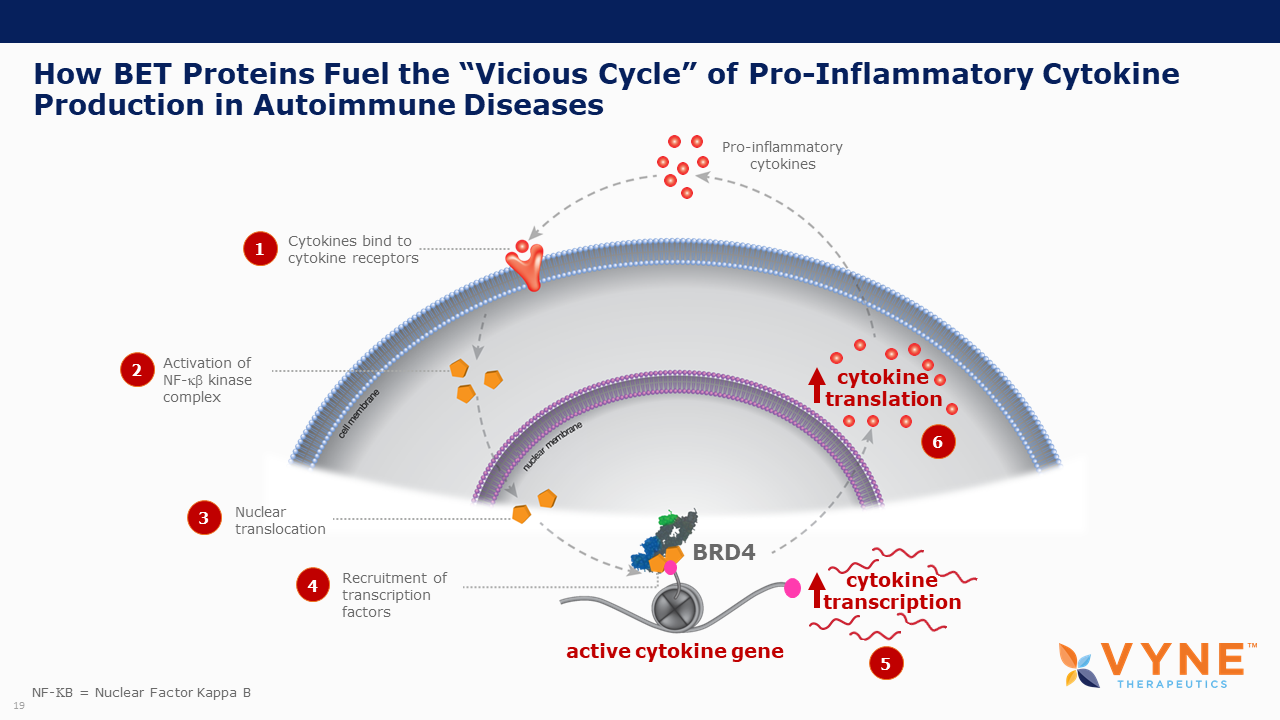
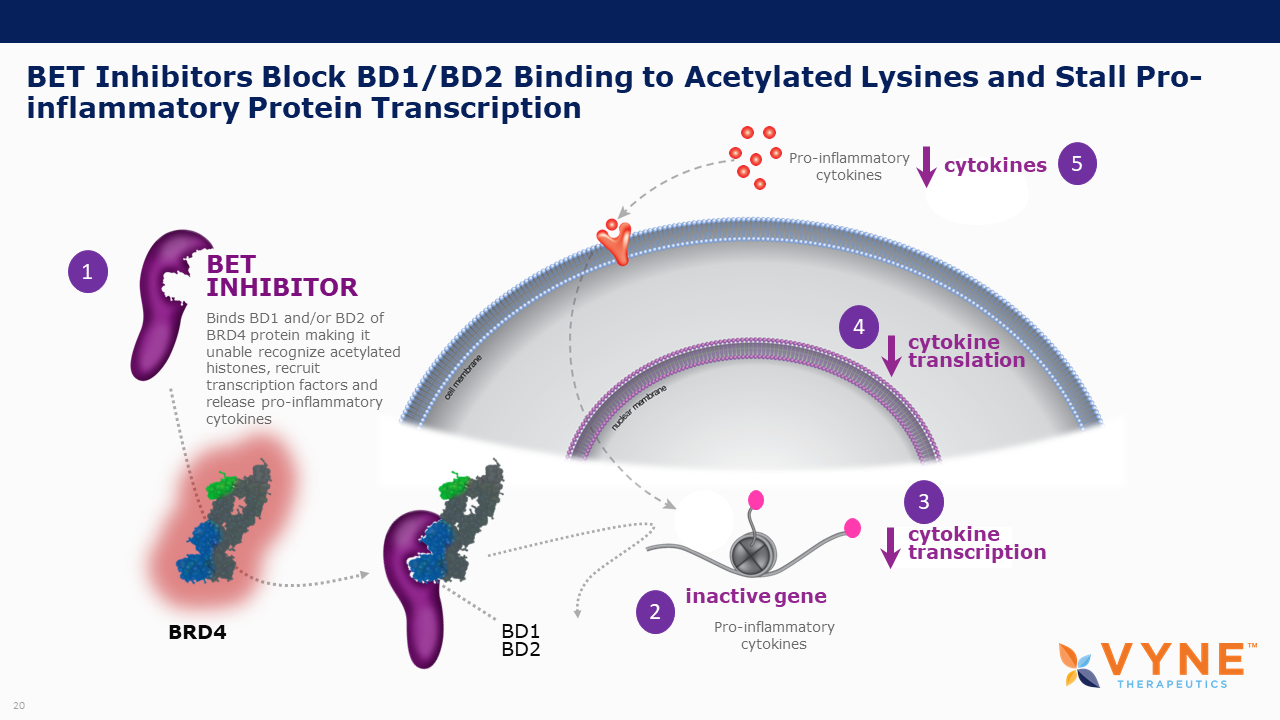
About Bromodomain and Extra-Terminal Domain (BET) Inhibitors
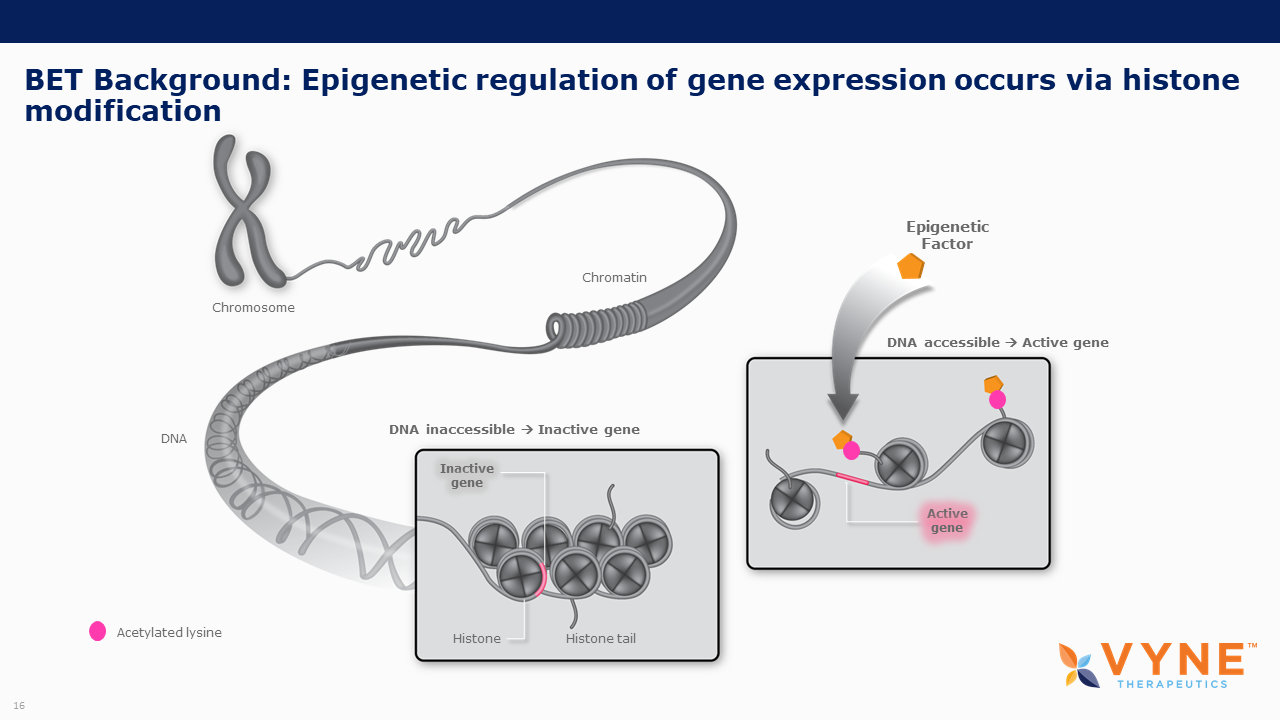
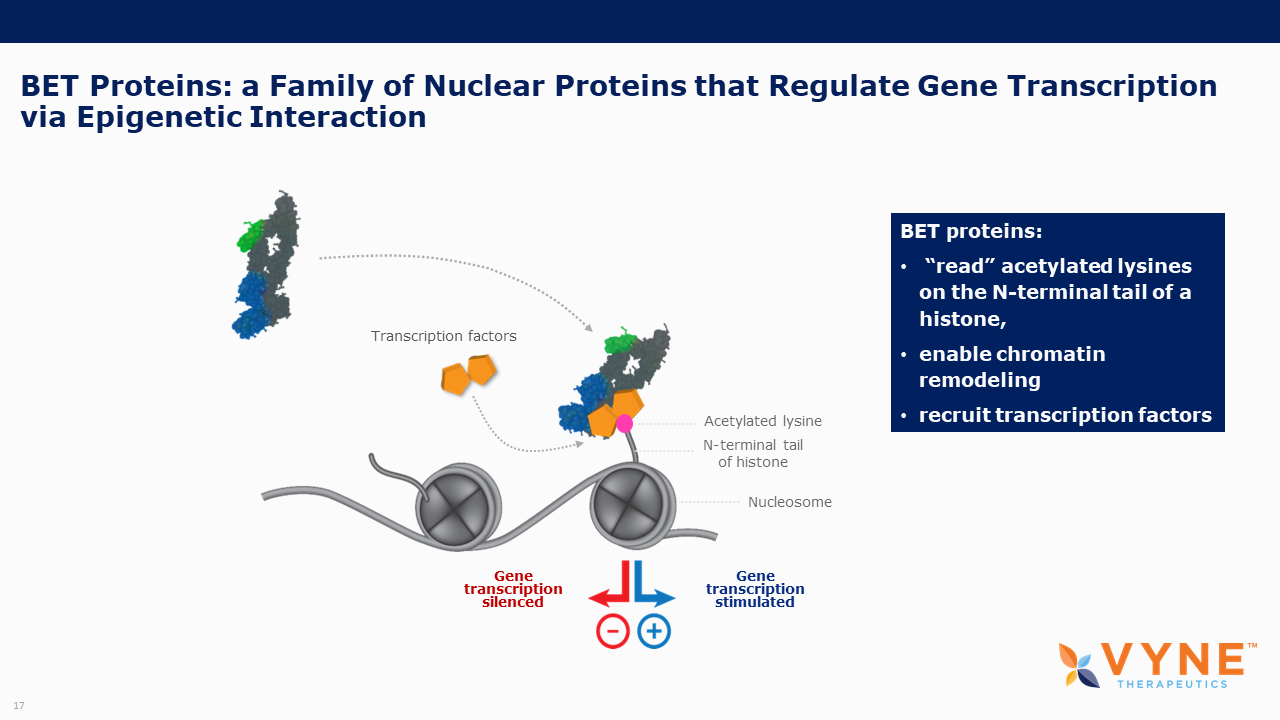
BET proteins play a key role in regulating gene transcription via epigenetic interactions (“reading”), and recent research has determined a key role for these BET proteins in regulating B cell and T cell activation and subsequent inflammatory processes. As epigenetic readers, BET proteins regulate the recruitment of transcriptional factors that are key to the production of several pro-inflammatory cytokines. Inhibiting BET proteins blocks cytokine transcription and therefore may have significant therapeutic potential across a wide variety of immuno-inflammatory/fibrotic and myeloproliferative neoplastic disorders. A topically applied BET inhibitor product has the possibility to positively impact diseases involving multiple, diverse inflammatory cell signaling pathways that are active in rare neutrophilic dermatoses. Furthermore, bromodomain 2 selective oral BET inhibitors may present as more conveniently-administered non-biologic treatment options for both acute control and chronic management of immuno-inflammatory indications, where the damaging effects of unrestricted inflammatory signaling activity is common.
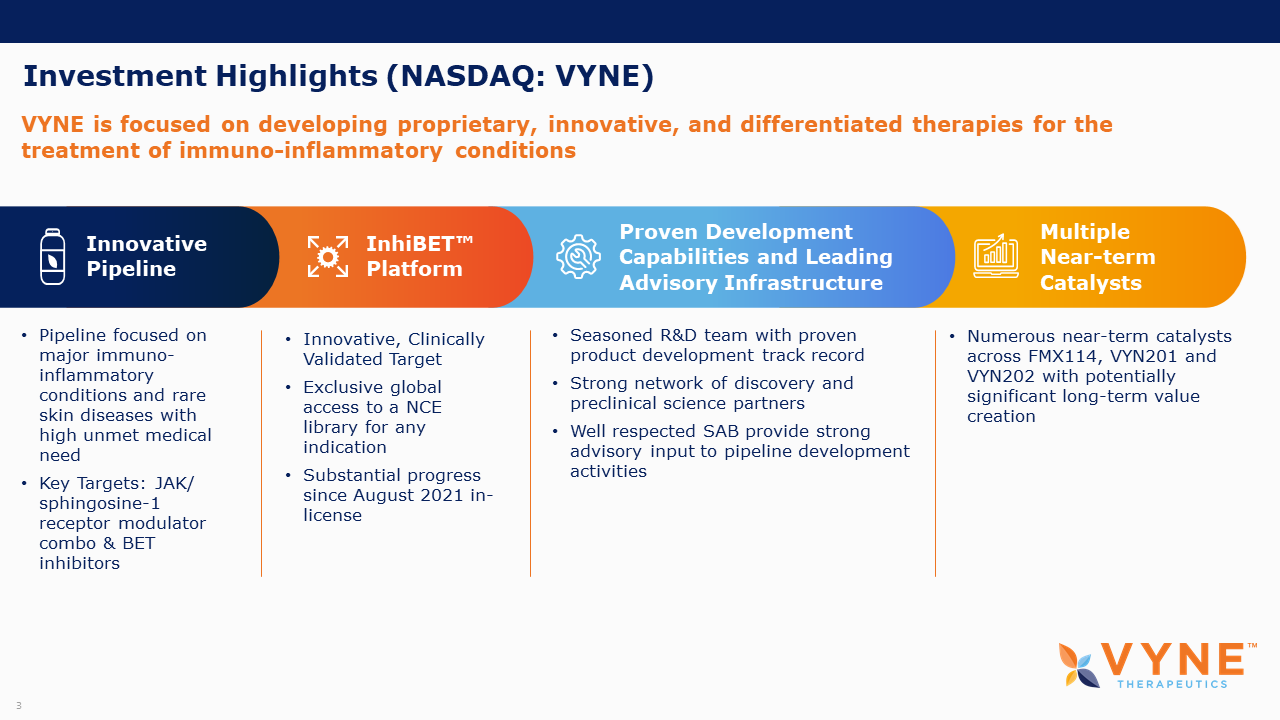
About VYNE Therapeutics Inc.
VYNE’s mission is to improve the lives of patients by developing proprietary, innovative, and differentiated therapies for the treatment of immuno-inflammatory conditions. VYNE is working to develop and commercialize a variety of therapies for major immuno-inflammatory conditions and rare skin diseases with high unmet medical need. The Company’s unique and proprietary pipeline includes FMX114 for the potential treatment of mild-to-moderate atopic dermatitis and access to a library of bromodomain & extra-terminal (BET) domain inhibitors in both topical and oral forms for the potential treatment of major immuno-inflammatory conditions and rare skin diseases.
For more information about VYNE Therapeutics Inc. or its investigational products, visit www.vynetherapeutics.com or follow VYNE on Twitter. VYNE may use its website to comply with its disclosure obligations under Regulation FD. Therefore, investors should monitor VYNE’s website in addition to following its press releases, filings with the U.S. Securities and Exchange Commission, public conference calls, and webcasts.
Investor Relations:
Joseph Ranieri
LifeSci Advisors, LLC
617-430-7582
joer@lifesciadvisors.com
Tyler Zeronda
Chief Financial Officer
VYNE Therapeutics
908-458-9106
Tyler.Zeronda@vynetx.com
Cautionary Statement Regarding Forward-Looking Statements
This release includes forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, including, but not limited to statements regarding the development of VYNE’s product candidates and other statements regarding the future expectations, plans and prospects of VYNE. All statements in this presentation which are not historical facts are forward-looking statements. Any forward-looking statements are based on VYNE’s current knowledge and its present beliefs and expectations regarding possible future events and are subject to risks, uncertainties and assumptions that could cause actual results to differ materially and adversely from those set forth or implied by such forward-looking statements. These risks and uncertainties include, but are not limited to, VYNE’s ability to complete a sale or out-license of its minocycline franchise on terms acceptable to VYNE in a timely manner or at all; VYNE’s ability to successfully develop its product candidates; the timing of commencement of future non-clinical studies and clinical trials; VYNE’s ability to successfully progress, complete, and receive favorable results in, clinical trials for its product candidates; VYNE’s ability to exercise its exclusive option with respect to an oral BETi candidate pursuant to the terms of the option agreement with In4Derm Limited; VYNE’s intentions and its ability to obtain additional funding, either through equity or debt financing transactions or collaboration arrangements; disruptions related to COVID-19 or another pandemic, epidemic or outbreak of a contagious disease, on the ability of VYNE’s suppliers to manufacture and provide materials for our product candidates, initiating and retaining patients in clinical trials, operating results, liquidity and financial condition; supply chain disruptions; the regulatory approval process for VYNE’s product candidates, including any delay or failure in obtaining requisite approvals; the potential market size of treatments for any diseases and market adoption of products, if approved or cleared for commercial use, by physicians and patients; developments and projections relating to competitors and the pharmaceuticals industry, including competing drugs and therapies; the timing or likelihood of regulatory filings and approvals or clearances for product candidates; VYNE’s ability to comply with various regulations applicable to its business; VYNE’s ability to create intellectual property and the scope of protection it is able to establish and maintain for intellectual property rights covering its product candidates, including the projected terms of patent protection; risks that any of VYNE’s patents may be held to be narrowed, invalid or unenforceable or one or more of VYNE’s patent applications may not be granted and potential competitors may also seek to design around VYNE’s granted patents or patent applications; the timing, costs or results of litigation, including litigation to protect its intellectual property, including the matter with Padagis Israel Pharmaceuticals Ltd.; VYNE’s ability to successfully challenge intellectual property claimed by others; estimates of VYNE’s expenses, capital requirements, its needs for additional financing and its ability to obtain additional capital on acceptable terms or at all; VYNE’s ability to attract and retain key scientific or management personnel; VYNE’s defense of any litigation that may be initiated against it; VYNE’s expectations regarding licensing, business transactions and strategic operations; VYNE’s future financial performance and liquidity; and volatility in VYNE’s stock price may result in rapid and substantial increases or decreases in the stock price that may or may not be related to the company’s operating performance or prospects. For a discussion of other risks and uncertainties, and other important factors, any of which could cause VYNE’s actual results to differ from those contained in the forward-looking statements, see the section titled “Risk Factors” in VYNE’s Annual Report on Form 10-K for the year ended December 31, 2020, as well as discussions of potential risks, uncertainties, and other important factors in VYNE’s subsequent filings with the U.S. Securities and Exchange Commission. Although VYNE believes these forward-looking statements are reasonable, they speak only as of the date of this announcement and VYNE undertakes no obligation to update publicly such forward-looking statements to reflect subsequent events or circumstances, except as otherwise required by law. Given these risks and uncertainties, you should not rely upon forward-looking statements as predictions of future events.
VYNE THERAPEUTICS INC.
CONDENSED CONSOLIDATED BALANCE SHEETS
(U.S. dollars in thousands, except per share data)
(Unaudited)
| September 30 | December 31 | |||||||
| 2021 | 2020 | |||||||
| Assets | ||||||||
| Current Assets: | ||||||||
| Cash and cash equivalents | $ | 52,306 | $ | 57,563 | ||||
| Restricted cash | 605 | 855 | ||||||
| Investment in marketable securities | — | 1,027 | ||||||
| Trade receivables, net of allowances | 10,084 | 15,819 | ||||||
| Prepaid and other assets | 5,064 | 4,591 | ||||||
| Inventory | 8,070 | 7,404 | ||||||
| Total Current Assets | 76,129 | 87,259 | ||||||
| Property and equipment, net | 472 | 555 | ||||||
| Operating lease right-of-use assets | 1,036 | 1,583 | ||||||
| Prepaid and other assets | 3,678 | 4,345 | ||||||
| Total Assets | $ | 81,315 | $ | 93,742 | ||||
| Liabilities and shareholders’ equity | ||||||||
| Current Liabilities: | ||||||||
| Trade payables | $ | 7,621 | $ | 4,780 | ||||
| Accrued expenses | 9,613 | 11,452 | ||||||
| Debt | — | — | ||||||
| Employee related obligations | 3,382 | 4,360 | ||||||
| Operating lease liabilities | 277 | 757 | ||||||
| Other | 104 | 104 | ||||||
| Total Current Liabilities | 20,997 | 21,453 | ||||||
| Liability for employee severance benefits | 206 | 312 | ||||||
| Operating lease liabilities | 775 | 853 | ||||||
| Long-term debt | — | 33,174 | ||||||
| Other liabilities | 451 | 457 | ||||||
| Total Liabilities | 22,429 | 56,249 | ||||||
| Commitments and Contingencies | — | — | ||||||
| Shareholders' Equity: | ||||||||
| Preferred stock: $0.0001 par value; 20,000,000 shares authorized at September 30, 2021 and December 31, 2020, respectively; no shares issued and outstanding at September 30, 2021 and December 31, 2020, respectively | — | — | ||||||
| Common stock: $0.0001 par value; 150,000,000 shares and 75,000,000 shares authorized at September 30, 2021 and December 31, 2020, respectively; 53,510,599 and 43,205,221 shares issued and outstanding at September 30, 2021 and December 31, 2020, respectively | 5 | 4 | ||||||
| Additional paid-in capital | 686,836 | 603,685 | ||||||
| Accumulated deficit | (627,955 | ) | (566,196 | ) | ||||
| Total Shareholders' Equity | 58,886 | 37,493 | ||||||
| Total Liabilities and Shareholders' Equity | $ | 81,315 | $ | 93,742 | ||||
VYNE THERAPEUTICS INC.
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS
(U.S. dollars in thousands, except per share data)
(Unaudited)
| Three months ended
September 30 | Nine months ended September 30 | |||||||||||||||
| 2021 | 2020 | 2021 | 2020 | |||||||||||||
| Revenues | ||||||||||||||||
| Product sales | $ | 3,953 | $ | 2,863 | $ | 11,805 | $ | 6,096 | ||||||||
| License revenues | — | — | — | 10,000 | ||||||||||||
| Royalty revenues | 133 | 406 | 658 | 611 | ||||||||||||
| Total Revenues | 4,086 | 3,269 | 12,463 | 16,707 | ||||||||||||
| Cost of goods sold | 1,049 | 371 | 2,445 | 858 | ||||||||||||
| Operating Expenses: | ||||||||||||||||
| Research and development | 6,981 | 6,623 | 19,723 | 35,695 | ||||||||||||
| Selling, general and administrative | 13,832 | 19,766 | 46,283 | 71,640 | ||||||||||||
| Goodwill and in-process research & development impairments | — | — | — | 54,345 | ||||||||||||
| Contingent Stock Right Remeasurement | — | — | — | 84,726 | ||||||||||||
| Total Operating Expenses | 20,813 | 26,389 | 66,006 | 246,406 | ||||||||||||
| Operating Loss | 17,776 | 23,491 | 55,988 | 230,557 | ||||||||||||
| Interest expense | 3,474 | 1,092 | 5,610 | 3,229 | ||||||||||||
| Other expense (income), net | 35 | 130 | 161 | (1,141 | ) | |||||||||||
| Loss Before Income Tax | 21,285 | 24,713 | 61,759 | 232,645 | ||||||||||||
| Income tax expense (benefit) | — | 1 | — | (258 | ) | |||||||||||
| Net Loss | $ | 21,285 | $ | 24,714 | $ | 61,759 | $ | 232,387 | ||||||||
| Loss per share basic and diluted | $ | 0.41 | $ | 0.59 | $ | 1.22 | $ | 7.98 | ||||||||
| Weighted average shares outstanding - basic and diluted | 52,028 | 41,934 | 50,776 | 29,132 | ||||||||||||
Non-GAAP Financial Measures
The following tables reconcile non-GAAP financial measures presented in this press release or that may be presented on the Company’s conference call with analysts and investors. The Company believes that these non-GAAP financial measures provide management, analysts, investors and other users of the Company’s financial information with meaningful supplemental information regarding the performance of the Company’s business. These non-GAAP financial measures should not be considered superior to, but rather in addition to, other financial measures prepared by the Company in accordance with GAAP, including the year-to-year results. The Company’s method of determining these non-GAAP financial measures may be different from other companies’ methods and, therefore, may not be comparable to those used by other companies, and the Company does not recommend the sole use of these non-GAAP measures to assess its financial and earnings performance. For reasons noted above, the Company is presenting certain non-GAAP financial measures for the three and nine months ended September 30, 2021 and September 30, 2020. In order for investors to be able to more easily compare the Company’s performance across periods, the Company has included comparable reconciliations for the 2020 period in the reconciliation tables below.
Reconciliation of EBITDA and Adjusted EBITDA (non-GAAP)
The following table provides a reconciliation of Net loss (GAAP) to EBITDA (non-GAAP) and Adjusted EBITDA (non-GAAP) for the three and nine months ended September 30, 2021 and 2020 (in thousands):
| Three Months Ended September 30, | Nine Months Ended September 30, | |||||||||||||||
| 2021 | 2020 | 2021 | 2020 | |||||||||||||
| Net loss (GAAP) | (21,285 | ) | (24,714 | ) | (61,759 | ) | (232,387 | ) | ||||||||
| Income tax expense (benefit) | — | 1 | — | (258 | ) | |||||||||||
| Interest expense, net | 3,474 | 1,092 | 5,610 | 3,229 | ||||||||||||
| Depreciation and amortization | 28 | 67 | 83 | 254 | ||||||||||||
| EBITDA (non-GAAP) | (17,783 | ) | (23,554 | ) | (56,066 | ) | (229,162 | ) | ||||||||
| Share based compensation expense | 2,405 | 2,620 | 6,748 | 15,147 | ||||||||||||
| Goodwill and in-process research & development impairments | — | — | — | 54,345 | ||||||||||||
| Contingent Stock Right Remeasurement | — | — | — | 84,726 | ||||||||||||
| Adjusted EBITDA (non-GAAP) | (15,378 | ) | (20,934 | ) | (49,318 | ) | (74,944 | ) | ||||||||
Reconciliation of Adjusted Net Loss and other non-GAAP metrics
The following table provides detailed reconciliations of various other income statement data between GAAP and non-GAAP amounts for the three and nine months ended September 30, 2021 and 2020 (in thousands, except per share data):
| Three months ended September 30 | Nine months ended September 30 | |||||||||||||||
| 2021 | 2020 | 2021 | 2020 | |||||||||||||
| Net Loss (GAAP) | (21,285 | ) | (24,714 | ) | (61,759 | ) | (232,387 | ) | ||||||||
| Share based compensation expense | 2,405 | 2,620 | 6,748 | 15,147 | ||||||||||||
| Goodwill and in-process research & development impairments | — | — | — | 54,345 | ||||||||||||
| Contingent Stock Right Remeasurement | — | — | — | 84,726 | ||||||||||||
| Adjusted Net Loss (non-GAAP) | (18,880 | ) | (22,094 | ) | (55,011 | ) | (78,169 | ) | ||||||||
| Research and development expense (GAAP) | 6,981 | 6,623 | 19,723 | 35,695 | ||||||||||||
| Share based compensation expense | (435 | ) | (361 | ) | (1,335 | ) | (3,927 | ) | ||||||||
| Adjusted Research and development expense (non-GAAP) | 6,546 | 6,262 | 18,388 | 31,768 | ||||||||||||
| Selling, general and administrative expense (GAAP) | 13,832 | 19,766 | 46,283 | 71,640 | ||||||||||||
| Share based compensation expense | (1,970 | ) | (2,259 | ) | (5,413 | ) | (11,220 | ) | ||||||||
| Adjusted Selling, general and administrative expense (non-GAAP) | 11,862 | 17,507 | 40,870 | 60,420 | ||||||||||||
| Total Operating Expenses (GAAP) | 20,813 | 26,389 | 66,006 | 246,406 | ||||||||||||
| Share based compensation expense | (2,405 | ) | (2,620 | ) | (6,748 | ) | (15,147 | ) | ||||||||
| Goodwill and in-process research & development impairments | — | — | — | (54,345 | ) | |||||||||||
| Contingent Stock Right Remeasurement | — | — | — | (84,726 | ) | |||||||||||
| Adjusted Total Operating Expenses | 18,408 | 23,769 | 59,258 | 92,188 | ||||||||||||
| Total Operating Loss (GAAP) | (17,776 | ) | (23,491 | ) | (55,988 | ) | (230,557 | ) | ||||||||
| Share based compensation expense | 2,405 | 2,620 | 6,748 | 15,147 | ||||||||||||
| Goodwill and in-process research & development impairments | — | — | — | 54,345 | ||||||||||||
| Contingent Stock Right Remeasurement | — | — | — | 84,726 | ||||||||||||
| Adjusted Total Operating Loss (non-GAAP) | (15,371 | ) | (20,871 | ) | (49,240 | ) | (76,339 | ) | ||||||||
| Net loss per common share - basic and diluted (GAAP) | (0.41 | ) | (0.59 | ) | (1.22 | ) | (7.98 | ) | ||||||||
| Share based compensation expense | 0.05 | 0.06 | 0.13 | 0.52 | ||||||||||||
| Goodwill and in-process research & development impairments | — | — | — | 1.87 | ||||||||||||
| Contingent Stock Right Remeasurement | — | — | — | 2.91 | ||||||||||||
| Adjusted Net loss per share - basic and diluted (non-GAAP) | (0.36 | ) | (0.53 | ) | (1.09 | ) | (2.68 | ) | ||||||||
| Weighted average number of common shares outstanding - basic and diluted | 52,028 | 41,934 | 50,776 | 29,132 | ||||||||||||