Exhibit 99.1
Kindred Biosciences Announces Second Quarter 2019 Financial Results and Unveils Positive Results from Pilot Efficacy Study of Parvovirus Monoclonal Antibody
– | Mortality benefit seen in both prophylaxis and treatment of parvovirus infection |
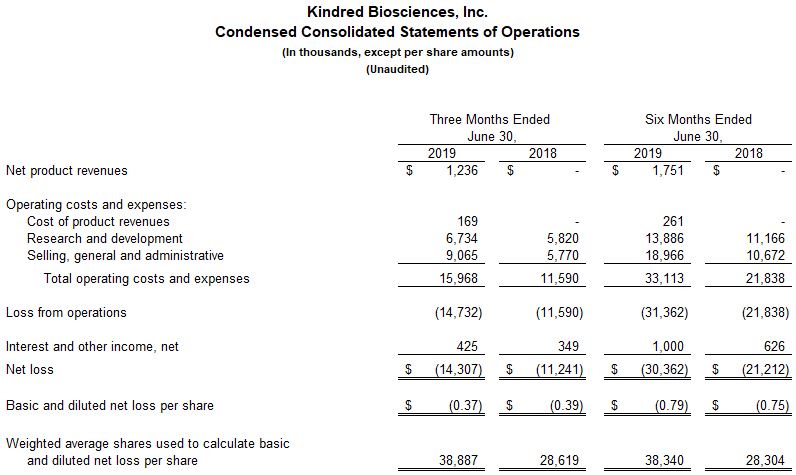
San Francisco, California (August 1, 2019) - Kindred Biosciences, Inc. (NASDAQ: KIN), a commercial-stage biopharmaceutical company focused on saving and improving the lives of pets, today announced financial results for the second quarter ended June 30, 2019 and provided updates on its programs, including positive results from a pilot efficacy study of its previously undisclosed parvovirus monoclonal antibody. For the second quarter 2019, KindredBio reported net product revenues of $1.2 million and a net loss of $14.3 million, or $0.37 per share. For the first six months of 2019, net product revenues were $1.8 million and the net loss was $30.4 million, or $0.79 per share.
“The increase in second quarter Mirataz revenues reflects broad-based customer adoption as expanded commercial activities drive continued growth in market penetration and re-order size,” said Richard Chin, CEO of KindredBio. “We are excited to announce our parvovirus antibody program, which targets a significant unmet medical need that results in very high mortality. We are pleased with the positive study results and are targeting approval of this product candidate by late 2020, or early 2021.”
“The second half of 2019 represents a catalyst rich period, and we are on track for key milestones. Earlier this week, we announced positive pilot field effectiveness study results for our lead canine atopic dermatitis biologic candidate, positioning it to become a key therapeutic in this large and growing market. We are also pleased to report that our first cGMP drug manufacturing campaigns at our plant in Burlingame have been successful, and that construction on our state-of-the-art Kansas biologics manufacturing facility has been completed on time and on budget. With end-to-end capabilities and a highly experienced biologics team that has worked on some of the most successful human drugs, we are well- positioned to be a leader in companion animal biologics. By year-end, we also anticipate announcement of results from additional pilot studies and the initiation of three pivotal studies. These catalysts have the potential to make 2019 a landmark year for KindredBio.”
Development and Corporate Updates
• | KindredBio today announced positive results from its pilot efficacy study of KIND-030, a chimeric, high-affinity monoclonal antibody targeting canine parvovirus. This was a 12-dog study, of which 4 dogs were treated prophylactically and 2 dogs were treated after establishment of the infection. All treated dogs survived, compared to none in the applicable placebo group. The effect was seen in both prophylaxis setting, as well as in a treatment setting after establishment of infection. Pivotal studies are expected to be conducted in 2020. |
Canine parvovirus (CPV) is the most significant contagious viral cause of enteritis in dogs, especially puppies, with mortality rates reportedly as high as 91%. There are currently no Food and Drug Administration (FDA) or United States Department of Agriculture (USDA) approved treatments for CPV, nor any other available treatment. Currently, owners spend thousands of dollars per puppy in supportive care for CPV.
• | The company recorded Mirataz® (mirtazapine transdermal ointment) net product revenues of $1.2 million in the second quarter, compared with net revenues of $0.5 million in the first quarter. Approximately 46% of veterinary clinics in the United States have purchased Mirataz since the product’s launch, and the reorder rate among participating clinics grew to approximately 65% in |
Exhibit 99.1
the quarter. The company expects ongoing commercial initiatives, the introduction of new marketing strategies and establishment of direct-to-consumer agreements to drive continued positive second-half momentum. KindredBio recently finalized agreements to stock Mirataz at leading eRetailers, such as Chewy.
The outcome of an oral hearing requested by the European Medicines Agency (EMA) and scheduled for September 2019 will determine the extent to which additional data may be needed to obtain approval in Europe. KindredBio will update the market on the path forward for Mirataz EU pending communication from the EMA post the hearing. Regulatory approval is subject to the typical risks inherent in such a process.
Mirataz is the first and only transdermal medication specifically developed, and FDA-approved, for the management of weight loss in cats. Weight loss in cats is a serious and potentially fatal condition that represents a leading cause of visits to the veterinarian for cats. The company's market research indicates that U.S. veterinarians see as many as nine million cats each year with unintended weight loss caused by varying underlying conditions, such as chronic kidney disease, cancer or diabetes. Mirataz, which is formulated with KindredBio's proprietary Accusorb™ technology, is applied topically to the cat's inner ear (pinna) once a day, providing a more attractive application route compared to oral administration. The product is classified as a weight gain drug and can be used in cats with various underlying diseases associated with unintended weight loss.
• | On July 29, 2019, KindredBio reported positive topline results from its pilot field effectiveness study of KIND-016, a fully caninized, high-affinity monoclonal antibody targeting interleukin-31, for the treatment of atopic dermatitis in dogs. At week 4, 60.7% of the KIND-016 group met treatment success criteria, vs. 33.3% of the placebo group. The reduction in itching, as measured by the PVAS score, peaked rapidly, showing significant efficacy as early as 24 hours with a trend as early as 4 hours, and the CADESI response was also very rapid. Treatment success rate reached 70% as early as week 1 in the KIND-016 group. While the study was not powered to demonstrate efficacy beyond week 4, the majority of dogs who were treatment successes at week 4 maintained response through week 8. In October 2018, KindredBio previously reported positive topline results from its laboratory pilot efficacy study of KIND-016. A pivotal study is expected to commence by year-end. |
• | The company also expects pilot effectiveness results for its canine anti-IL-4/IL-13 SINK molecule by year-end and is advancing other programs for atopic dermatitis. |
• | KindredBio is pursuing a multi-pronged approach toward atopic dermatitis, with a portfolio of promising biologics. Atopic dermatitis is an immune-mediated inflammatory skin condition in dogs. It is the leading reason owners take their dog to the veterinarian, and the current market size is more than $600 million annually and growing. |
• | The FDA has approved the safety and effectiveness technical sections for dipyrone injection for the control of pyrexia (fever) in horses. On May 16, 2019, KindredBio announced that it had been notified by its contract manufacturer of the active pharmaceutical ingredient (API) dipyrone that the FDA Center for Veterinary Medicine (CVM) had follow up questions, following an inspection in March 2019. Responses have since been submitted to the FDA by the API manufacturer, and the FDA has issued an Establishment Inspection Report indicating that the facility was compliant with good manufacturing practices. KindredBio has reactivated the New Animal Drug Application (NADA). The FDA granted a shortened timeline of 135 days for review of the NADA. Approval is |
Exhibit 99.1
expected in the fourth quarter of 2019 and is dependent on a product- and application-specific facility assessment of the API manufacturer by the CVM review office. Regulatory approval is subject to the typical risks inherent in such a process. Preparations for the commercial launch remain on track.
Dipyrone injection is expected to be the first FDA-approved product for the control of fever in horses. There are eight to nine million horses in the U.S. and currently more than one million are seen by a veterinarian for fever annually. Existing off-label treatments can have serious side effects.
• | The pivotal field effectiveness study for dipyrone oral gel has been completed with positive results. The target animal safety study is also complete, and dipyrone oral gel was found to be well-tolerated. KindredBio has agreed on a path forward with the FDA and bridging studies will likely commence in 2020. |
• | Dipyrone oral gel, which is a proprietary oral gel, is intended as a leave behind for owners to administer to their horse for continued care following dipyrone injection. Accordingly, it is expected to expand use of the drug and build upon the success of dipyrone injection. |
• | KindredBio’s feline recombinant erythropoietin cGMP drug substance met release criteria at the company’s plant in Burlingame, CA, and cGMP fill & finish will be undertaken at the Elwood, Kansas biologics manufacturing facility in the third quarter of 2019. Thereafter, a pivotal effectiveness study will commence before year-end. The product candidate is being developed for the management of non-regenerative anemia in cats. It has been engineered by the company to have a prolonged half-life compared to endogenous erythropoietin, a protein that regulates and stimulates production of red blood cells. KindredBio announced positive topline results from a pilot field effectiveness study of its feline recombinant erythropoietin in January 2019. |
• | Anemia is a common condition that is estimated to afflict millions of older cats. It is often associated with chronic kidney disease, because kidneys produce erythropoietin and chronic kidney disease leads to decreased levels of endogenous erythropoietin. Chronic kidney disease affects approximately half of older cats, making it a leading cause of feline mortality. Human erythropoietins, which are multi-billion dollar products in the human market, have been shown to be immunogenic in many cats. |
• | The pilot field effectiveness study for KindredBio's anti-TNF antibody for canine inflammatory bowel disease (IBD) has been initiated and is underway. Study results are expected by the end of 2019. |
• | The majority of canine IBD cases involve chronic states of diarrhea, vomiting, gastroenteritis, inappetence, and other symptoms, certain of which are cited as among the most frequent disorders impacting dogs. For certain dog breeds, the prevalence of diarrhea exceeds 5%. Alongside a high frequency of canine IBD, the duration of impact is also significant, as IBD is most commonly diagnosed in middle aged dogs yet can impact the animal for life. Existing treatments can have significant drawbacks, including limited diets and excessive antibiotic use, which can lead to owner frustration, lapses in treatment adherence, or poor quality of life for the affected animal. |
• | The pilot field effectiveness study of KIND-014 for the treatment of gastric ulcers in horses has been completed with positive results. The company has selected a formulation for development and both the pivotal field and pivotal safety studies will begin in the second half of 2019. |
Exhibit 99.1
• | Equine gastric ulcer syndrome (EGUS) is a common condition in horses. Prevalence estimates have been reported to range from 60% to 90% in adult horses, depending on age, performance, and evaluated populations. A variety of clinical signs are associated with EGUS, including poor appetite, poor condition, colic, and behavioral issues. |
• | The pilot field effectiveness study of KindredBio's anti-TNF monoclonal antibody targeting sick or septic foals has been completed, with positive results. KindredBio will initiate the next field study in 2020. |
• | Sepsis in foals can cause up to 50% mortality and is an important unmet medical need. There is currently no FDA-approved therapy. |
• | Construction to support initial production lines on KindredBio's biologics manufacturing facility in Elwood, Kansas is complete. The fill & finish equipment is installed and fully commissioned. The bioreactors are being used for production of IL-31 antibodies offsite and will be installed and commissioned in Kansas on or before the first quarter of 2020, once current production activities are complete. The facility includes approximately 180,000 square feet with clean rooms, utility, equipment, and related quality documentation suitable for small molecule and biologics manufacturing. KindredBio acquired the facility in August 2017. |
Second Quarter 2019 Financial Results
For the quarter ended June 30, 2019, KindredBio reported a net loss of $14.3 million or $0.37 per share, as compared to a net loss of $11.2 million or $0.39 per share, for the same period in 2018. For the six months ended June 30, 2019, the net loss was $30.4 million or $0.79 per share, as compared to a net loss of $21.2 million or $0.75 per share for the same period in 2018.
The company recorded $1.2 million in net product revenues for Mirataz for the second quarter and $1.8 million for the first six months of 2019. There were no net product revenues for the same periods in 2018 as Mirataz became commercially available in July 2018.
The cost of product sales totaled $0.2 million in the second quarter and $0.3 million for the six months in 2019, resulting in a gross margin of 86% and 85%, respectively.
Research and development expenses for the three and six months ended June 30, 2019 were $6.7 million and $13.9 million, respectively, compared to $5.8 million and $11.2 million for the same periods in 2018. Stock-based compensation expense included in research and development expense was $0.5 million and $0.9 million for the three and six months ended June 30, 2019, as compared to $0.4 million and $0.9 million for the same periods in 2018. The $2.7 million year-over-year increase in research and development expenses was primarily due to higher headcount and related expenses as the company focuses on advancing its biologics programs, and higher consulting expenses for quality assurance programs.
Selling, general and administrative expenses were $9.1 million and $19.0 million for the three and six months ended June 30, 2019, compared to $5.8 million and $10.7 million for the same periods in 2018. The $8.3 million year-over-year increase is the result of being a commercial company, as well as increased expenses incurred by the Elwood, Kansas plant in the lead up to its commissioning. In addition, higher corporate infrastructure costs and stock-based compensation expense also contributed to the increase in
Exhibit 99.1
expenses. Stock based compensation expense was $1.4 million and $2.8 million for the three and six months in the first half of 2019, versus $1.0 million and $2.0 million in the year-ago period.
As of June 30, 2019, KindredBio had $79.6 million in cash, cash equivalents and investments, compared with $73.9 million as of December 31, 2018. Net cash used in operating activities for the first six months of 2019 was approximately $31.8 million, offset by $43.1 million of net cash proceeds from an underwritten public offering of its common stock in the first quarter of 2019. The company also invested approximately $6.7 million in capital expenditures for the remaining portion of the build-out of its Elwood, Kansas manufacturing facility and the purchase of associated lab and manufacturing equipment for the facility.
With respect to spending in 2019, the company continues to expect operating expenses of between $57 million and $59 million, excluding the impact of stock-based compensation expense and the impact of acquisitions, if any. In addition, the company is on track with its $8.0 million to $10.0 million investment in capital expenditures for the year. KindredBio believes its existing cash, cash equivalents, restricted cash and short-term investments will be sufficient to fund the current operating plan at least through the end of 2020.
Webcast and Conference Call
KindredBio will host a conference call and webcast today at 4:30 p.m. Eastern time/1:30 p.m. Pacific time. Interested parties may access the call by dialing toll-free (855) 433-0927 from the US, or (484) 756-4262 internationally, and using conference ID 3784614. The call will be webcast live here, with a replay available at that link for 30 days.
Important Safety Information
Mirataz® (mirtazapine transdermal ointment) is for topical use in cats only under veterinary supervision. Do not use in cats with a known hypersensitivity to mirtazapine or any of the excipients or in cats treated with monoamine oxidase inhibitors (MAOIs). Not for human use. Keep out of reach of children. Wear gloves to apply and wash hands after. Avoid contact with treated cat for 2 hours following application. The most common adverse reactions include application site reactions, behavioral abnormalities (vocalization and hyperactivity) and vomiting. Please see the full Prescribing Information.
About Kindred Biosciences
Kindred Biosciences is a commercial-stage biopharmaceutical company focused on saving and improving the lives of pets. Its mission is to bring to pets the same kinds of safe and effective medicines that human family members enjoy. The company's strategy is to identify compounds and targets that have already demonstrated safety and efficacy in humans and to develop therapeutics based on these validated compounds and targets for dogs, cats, and horses. KindredBio has a deep pipeline of novel drugs and biologics in development across many therapeutic classes. Its first approved drug is Mirataz® (mirtazapine transdermal ointment) for the management of weight loss in cats.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the U.S. Private Securities Litigation Reform Act of 1995. All statements contained in this press release that do not relate to matters of historical fact should be considered forward-looking statements, including, but not limited to, statements regarding our expectations about the trials, regulatory approval, manufacturing, distribution
Exhibit 99.1
and commercialization of our current and future product candidates, and statements regarding our anticipated revenues, expenses, margins, profits and use of cash.
These forward-looking statements are based on our current expectations. These statements are not promises or guarantees, but involve known and unknown risks, uncertainties and other important factors that may cause our actual results to be materially different from any future results expressed or implied by the forward-looking statements. These risks include, but are not limited to, the following: our limited operating history and expectations of losses for the foreseeable future; the absence of significant revenue from our products and our product candidates for the foreseeable future; the likelihood that our revenue will vary from quarter to quarter; our potential inability to obtain any necessary additional financing; our substantial dependence on the success of our products and our lead product candidates which may not be successfully commercialized even if they are approved for marketing; the effect of competition; our potential inability to obtain regulatory approval for our existing or future product candidates; our dependence on third parties to conduct some of our development activities; our dependence upon third-party manufacturers for supplies of our products and our product candidates; uncertainties regarding the outcomes of trials regarding our product candidates; our potential failure to attract and retain senior management and key scientific personnel; uncertainty about our ability to develop a satisfactory sales organization; our significant costs of operating as a public company; our potential inability to obtain and maintain patent protection and other intellectual property protection for our products and our product candidates; potential claims by third parties alleging our infringement of their patents and other intellectual property rights; our potential failure to comply with regulatory requirements, which are subject to change on an ongoing basis; the potential volatility of our stock price; and the significant control over our business by our principal stockholders and management.
For a further description of these risks and other risks that we face, please see the risk factors described in our filings with the U.S. Securities and Exchange Commission (the SEC), including the risk factors discussed under the caption "Risk Factors" in our Annual Report on Form 10-K and any subsequent updates that may be contained in our Quarterly Reports on Form 10-Q filed with the SEC. As a result of the risks described above and in our filings with the SEC, actual results may differ materially from those indicated by the forward-looking statements made in this press release. Forward-looking statements contained in this press release speak only as of the date of this press release and we undertake no obligation to update or revise these statements, except as may be required by law.
The results stated in this press release have not been reviewed by the Food and Drug Administration or the United States Department of Agriculture Center for Veterinary Biologics, as applicable.
Contacts
For investor inquiries:
Katja Buhrer
Katja.buhrer@kindredbio.com
(917) 969-3438
Exhibit 99.1