Exhibit 99.1

iSpecimen Reports First Quarter 2022 Financial and Operating Results
- Recently Launched Enhanced iSpecimen Marketplace Platform to Help Scientists More Efficiently Search for Biospecimens -
LEXINGTON, MA, May 10, 2022 – iSpecimen Inc. (Nasdaq: ISPC) (“iSpecimen” or the “Company”), an online global marketplace that connects scientists in need of biospecimens for medical research with a network of healthcare specimen providers, today reported its financial and operating results for the three-month period ended March 31, 2022.
“Our team has been working diligently on expanding the capabilities of the iSpecimen Marketplace® platform, accelerating the growth and utilization of our global supply chain, and invigorating revenue growth for long term value. Recently, we saw the fruits of our labors with the first update to the enhanced iSpecimen Marketplace® platform, which delivers incremental, yet foundational, improvements to our software platform. This includes new data integration capabilities to further grow and federate our supply chain – such as automated data ingestion and data harmonization tools,” said Christopher Ianelli, MD, PhD, CEO and President of iSpecimen. “We expect that the enhanced iSpecimen Marketplace® platform, coupled with the investments we’ve made this past quarter to strengthen and expand our operations and sales teams globally, have positioned us to even better support our research clients’ needs for biospecimens going forward."
Dr. Ianelli concluded, “During the first quarter, iSpecimen demonstrated operational resiliency and flexibility despite a sudden shut down of our critical supply network in the Ukraine and Russia. Against the challenging backdrop of the conflict there, and a testament to the global reach of our network, we were able to quickly mobilize and transition biospecimen orders to other suppliers. The effect of this transition was a delay in the collection of specimens for approximately $1.0 million in open purchase orders, which had a corresponding negative impact to revenue in the first quarter. That said, our ability to shift these orders to other supply partners in our network highlights the adaptability of iSpecimen’s marketplace and our ability to address the fragmented global biospecimen procurement market and overcome supply headwinds. In spite of the setback this quarter, we remain extremely optimistic about our ability to gain additional market traction and help advance critical medical research.”
Q1 2022 Financial & Operational Highlights
| · | Revenue was approximately $2.5 million for the first quarter of 2022, compared to approximately $3.0 million for first quarter of 2021. The reduction in revenue during the quarter was primarily due to the Russia and Ukraine conflict that shut down supply sites in those regions and affected approximately $1.0 million in open purchase orders. | |
| · | Revenue mix in Q1 2022 was comprised of approximately $2.0 million in general specimen revenue and approximately $0.5 million in COVID-related revenue, or 81% and 19% of revenue, respectively, compared to approximately $2.3 million in general specimen revenue and $0.7 million in COVID-related revenue, or 77% and 23% of revenue, respectively the same period a year ago. | |
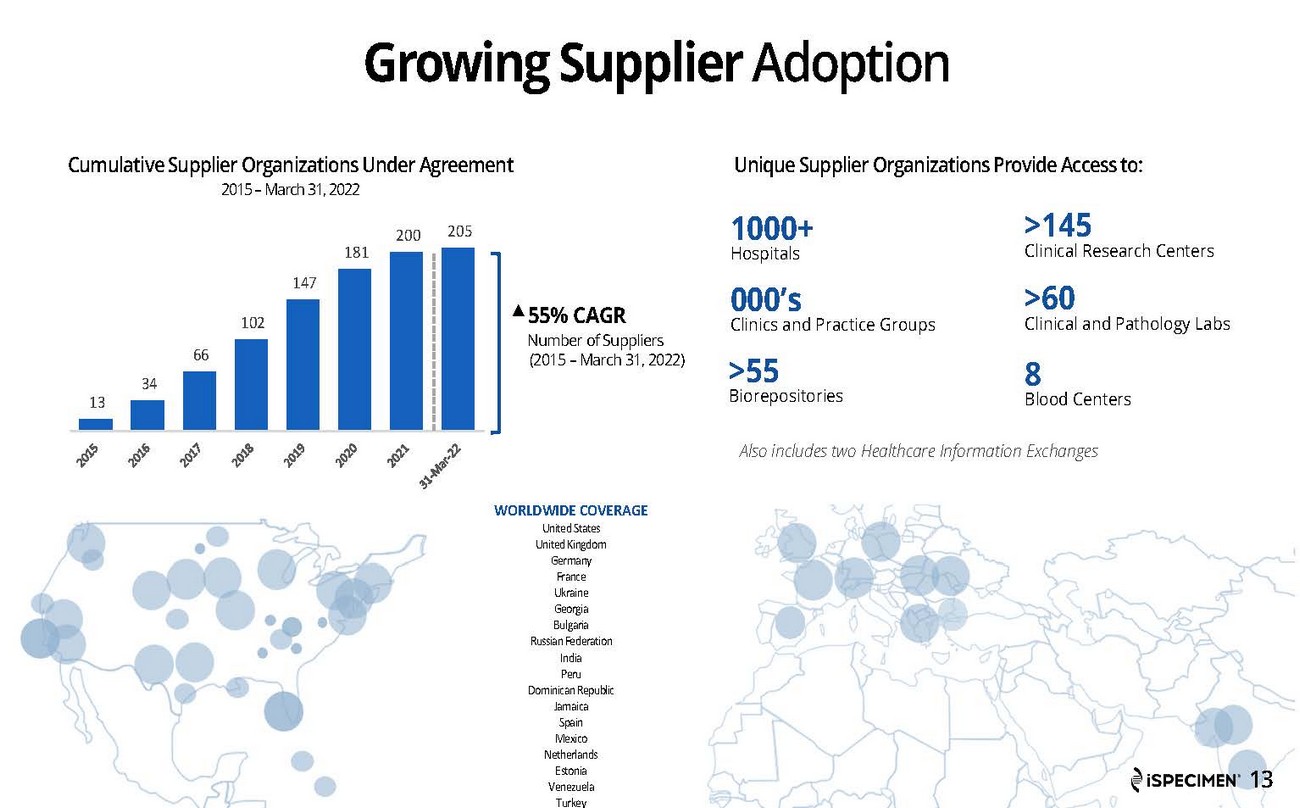
| · | Unique supplier organizations under agreement were 205 as of March 31, 2022, up from 189 as of March 31, 2021. |

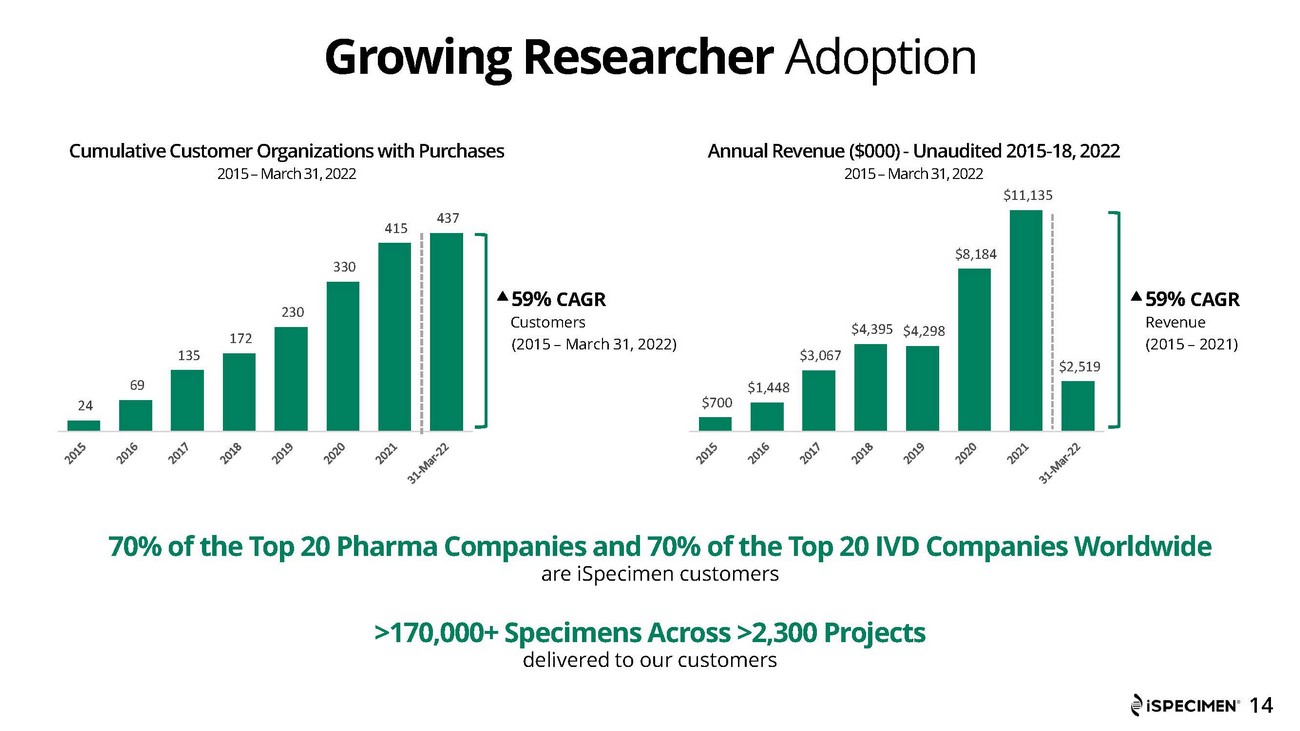
| · | Unique customer organizations who have purchased from iSpecimen totaled 437 as of March 31, 2022, an increase of 25% year over year. | |
| · | iSpecimen Marketplace had more than 5,600 registered research and supplier users as of March 31, 2022, up 34% year-over-year. |
Recent Corporate Updates
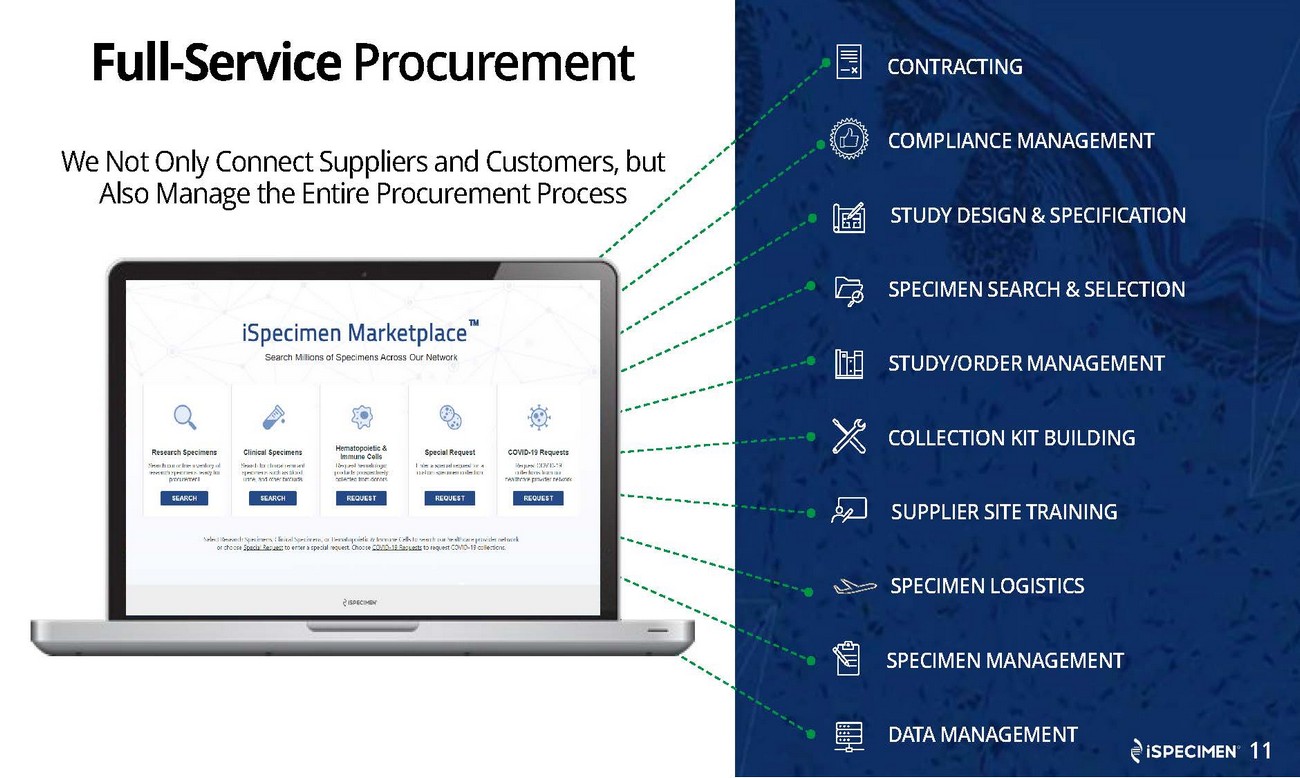
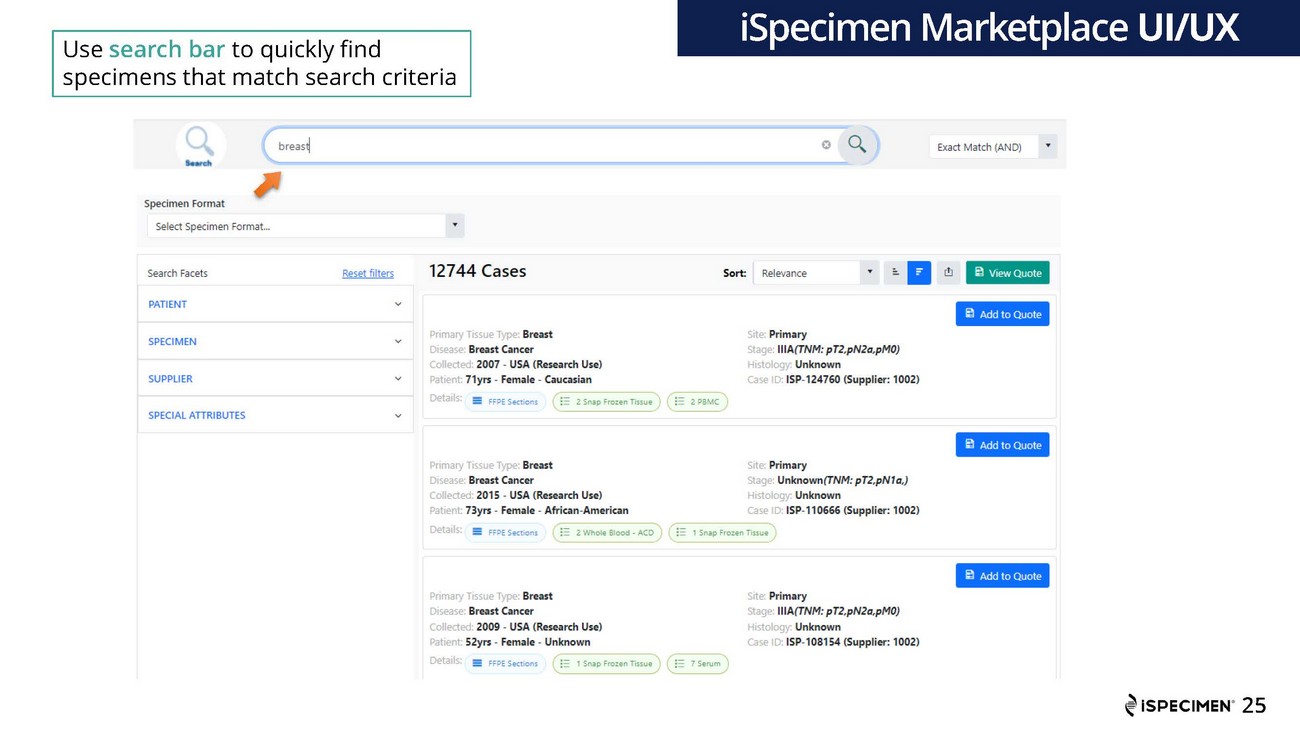
| · | Enhanced its iSpecimen Marketplace® platform with a redesigned platform with an easy-to-use interface that allows researchers to directly search for desired samples in iSpecimen’s provider network via a Google-like search bar. The new software includes improved search and supplier data integration capabilities that have been designed to efficiently connect scientists with well-matched samples for their research, as well as help to increase supplier inventory utilization. |
| · | Added Acutis Diagnostics (“Acutis”), a clinical diagnostics laboratory, to its platform as a laboratory services provider. Through the Acutis relationship, iSpecimen will now offer sequencing services to its research community to help researchers better understand genetic mutations and their impact on diseases such as COVID-19 and cancer. |
| · | Surpassed 200 healthcare providers in the iSpecimen Marketplace network. iSpecimen providers consist of hospitals, clinics, labs, biorepositories, and clinical trial management organizations dedicated to streamlining biospecimen procurement. |
| · | Expanded its supplier capabilities with the addition of a reference lab in New York to support customers focused on COVID-19 research, including analyzing the Omicron and Delta variants. |
Financial Results for the First Quarter of 2022
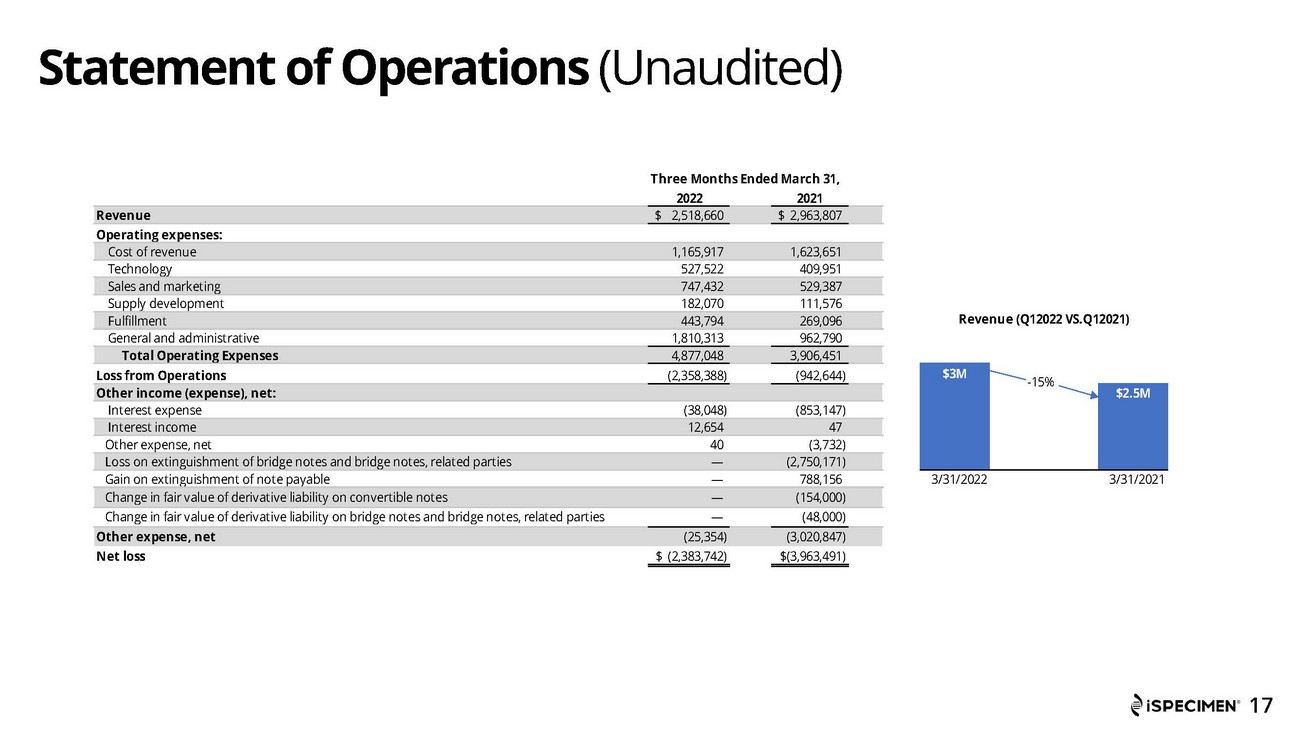
Revenue for the first quarter of 2022 was approximately $2.5 million, compared to approximately $3.0 million for the first quarter of 2021. The decrease in revenue was due to was primarily due to the Russia and Ukraine conflict which shut down supply sites in those regions and affected approximately $1.0 million in open purchase orders, along with a reduction in sales of COVID-19 specimens when compared to the prior year.
Cost of revenue was approximately $1.2 million for the first quarter of 2022, compared to approximately $1.6 million for the first quarter of 2021. The decrease was attributable both a decrease in the overall number of specimens that comprised the cost of revenue, along with an 11% decrease in the average cost per specimen.
General and administrative expenses were approximately $1.8 million for the first quarter of 2022, compared to approximately $0.9 million for the first quarter of 2021. The increase was primarily attributable to increased costs related to cash compensation, stock compensation, D&O insurance and operating and maintenance expenses.
Net loss was approximately $2.4 million or $(0.27) per share for the first quarter 2022, compared to net loss of approximately $4.0 million or $(4.23) per share for the same period in 2021.
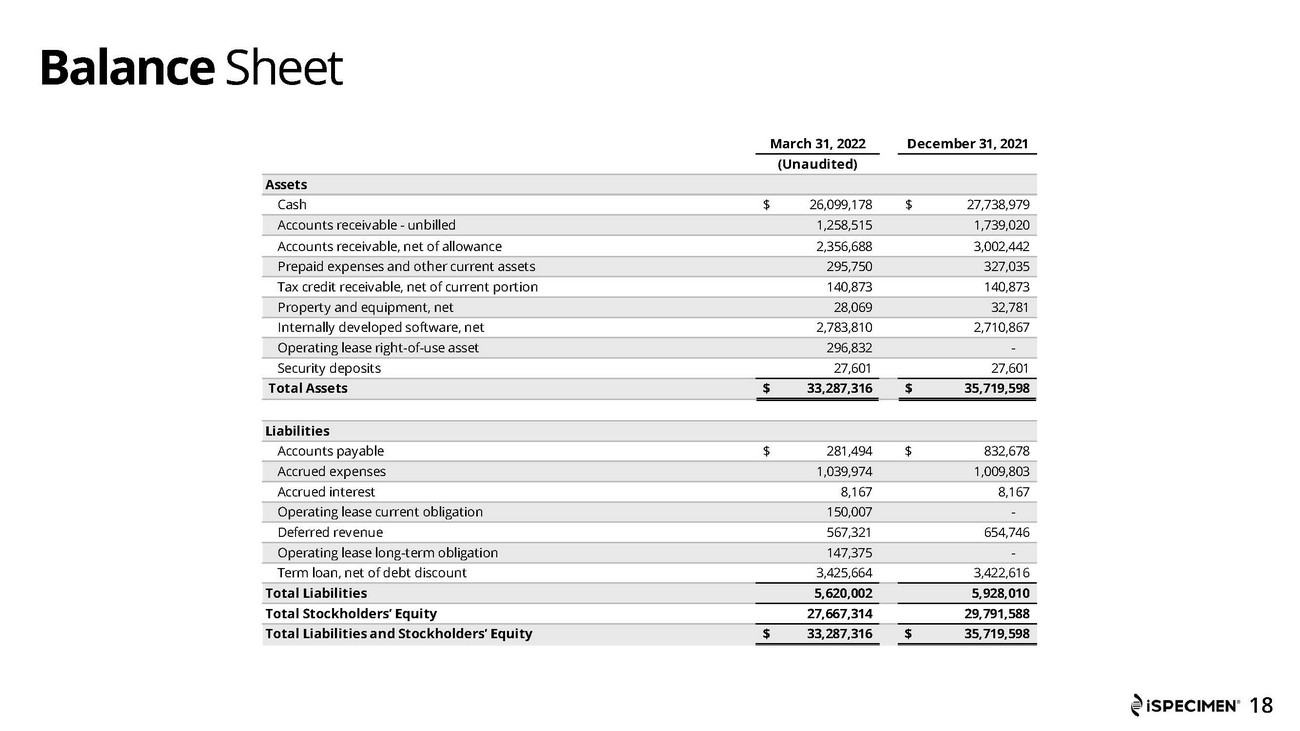
Cash was approximately $26.1 million as of March 31, 2022, compared to approximately 27.7 million as of December 31, 2021.

Conference Call and Webcast Information
The Company will host a conference call and audio webcast today, Tuesday, May 10th at 8:30 a.m. Eastern Time featuring remarks by Christopher Ianelli, MD, PhD, CEO and President, Tracy Curley, CFO and Treasurer, and Jill Mullan, COO and Secretary.
| Event: | iSpecimen First Quarter 2022 Results Conference Call |
| Date: | Tuesday, May 10, 2022 |
| Time: | 8:30 a.m. Eastern Time |
| Live Call: | +1-800-954-0654 (U.S. Toll Free) or +1-212-231-2933 (International) |
| Webcast: | https://viavid.webcasts.com/starthere.jsp?ei=1546450&tp_key=be907cc4df |
For interested individuals unable to join the conference call, a replay will be available through May 24, 2022, at +1-844-512-2921 (U.S. Toll Free) or +1-412-317-6671 (International). Participants must use the following code to access the replay of the call: 22018635. An archived version of the webcast will also be available on iSpecimen’s Investor Relations site: https://ispecimen.irpass.com/.
About iSpecimen
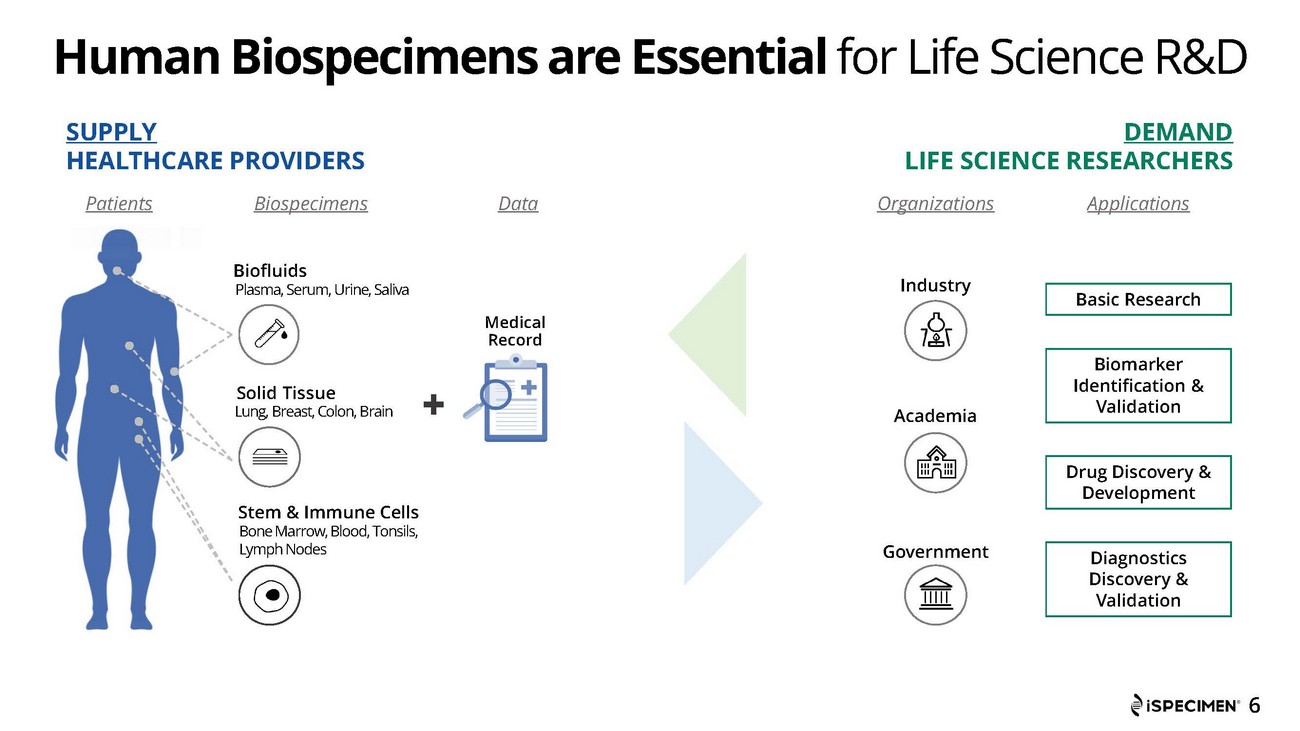
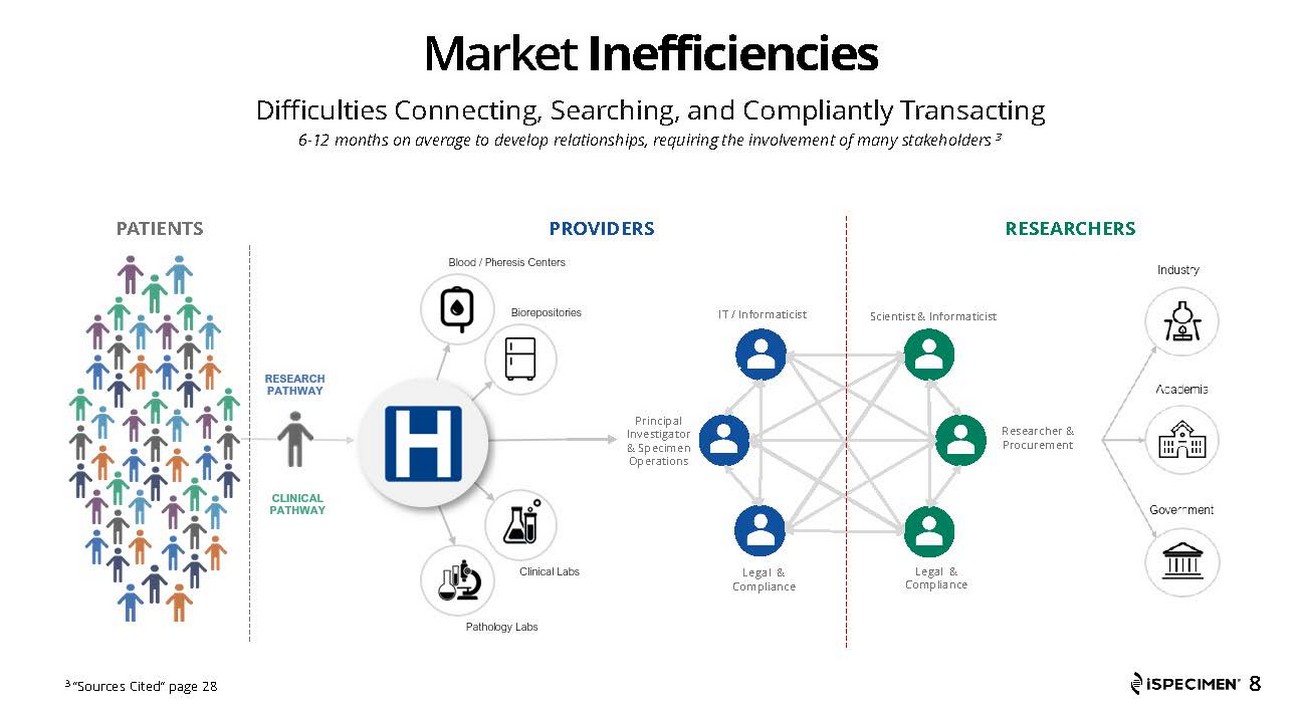
iSpecimen (Nasdaq: ISPC) offers an online marketplace for human biospecimens, connecting scientists in commercial and non-profit organizations with healthcare providers that have access to patients and specimens needed for medical discovery. Proprietary, cloud-based technology enables scientists to intuitively search for specimens and patients across a federated partner network of hospitals, labs, biobanks, blood centers and other healthcare organizations. For more information, please visit www.ispecimen.com.
Forward Looking Statements
This press release may contain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. Such forward-looking statements are characterized by future or conditional verbs such as "may," "will," "expect," "intend," "anticipate," believe," "estimate" and "continue" or similar words. You should read statements that contain these words carefully because they discuss future expectations and plans, which contain projections of future results of operations or financial condition or state other forward-looking information.
Forward-looking statements are predictions, projections and other statements about future events that are based on current expectations and assumptions and, as a result, are subject to risks and uncertainties. Many factors could cause actual future events to differ materially from the forward-looking statements in this press release, including but not limited to the risks factors contained in the Company's filings with the Securities and Exchange Commission, which are available for review at www.sec.gov. Forward-looking statements speak only as of the date they are made. New risks and uncertainties arise over time, and it is not possible for the Company to predict those events or how they may affect the Company. If a change to the events and circumstances reflected in the Company's forward-looking statements occurs, the Company's business, financial condition and operating results may vary materially from those expressed in the Company's forward-looking statements.
Readers are cautioned not to put undue reliance on forward-looking statements, and the Company assumes no obligation and do not intend to update or revise these forward-looking statements, whether as a result of new information, future events or otherwise.
For further information, please contact:
Investor Contact
KCSA Strategic Communications
Allison Soss
iSpecimen@kcsa.com
Media Contacts
KCSA Strategic Communications
Raquel Cona / Shana Marino
iSpecimen@kcsa.com

iSpecimen Inc.
BALANCE SHEETS
| March 31, 2022 | December 31, 2021 | |||||||
| (Unaudited) | ||||||||
| Assets | ||||||||
| Cash | $ | 26,099,178 | $ | 27,738,979 | ||||
| Accounts receivable - unbilled | 1,258,515 | 1,739,020 | ||||||
| Accounts receivable, net of allowance | 2,356,688 | 3,002,442 | ||||||
| Prepaid expenses and other current assets | 295,750 | 327,035 | ||||||
| Tax credit receivable, net of current portion | 140,873 | 140,873 | ||||||
| Property and equipment, net | 28,069 | 32,781 | ||||||
| Internally developed software, net | 2,783,810 | 2,710,867 | ||||||
| Operating lease right-of-use asset | 296,832 | - | ||||||
| Security deposits | 27,601 | 27,601 | ||||||
| Total Assets | $ | 33,287,316 | $ | 35,719,598 | ||||
| Liabilities | ||||||||
| Accounts payable | $ | 281,494 | $ | 832,678 | ||||
| Accrued expenses | 1,039,974 | 1,009,803 | ||||||
| Accrued interest | 8,167 | 8,167 | ||||||
| Operating lease current obligation | 150,007 | - | ||||||
| Deferred revenue | 567,321 | 654,746 | ||||||
| Operating lease long-term obligation | 147,375 | - | ||||||
| Term loan, net of debt discount | 3,425,664 | 3,422,616 | ||||||
| Total Liabilities | 5,620,002 | 5,928,010 | ||||||
| Total Stockholders’ Equity | 27,667,314 | 29,791,588 | ||||||
| Total Liabilities and Stockholders’ Equity | $ | 33,287,316 | $ | 35,719,598 | ||||

iSpecimen Inc.
STATEMENTS OF OPERATIONS
| Three Months Ended March 31, | ||||||||
| 2022 | 2021 | |||||||
| Revenue | $ | 2,518,660 | $ | 2,963,807 | ||||
| Operating expenses: | ||||||||
| Cost of revenue | 1,165,917 | 1,623,651 | ||||||
| Technology | 527,522 | 409,951 | ||||||
| Sales and marketing | 747,432 | 529,387 | ||||||
| Supply development | 182,070 | 111,576 | ||||||
| Fulfillment | 443,794 | 269,096 | ||||||
| General and administrative | 1,810,313 | 962,790 | ||||||
| Total Operating Expenses | 4,877,048 | 3,906,451 | ||||||
| Loss from Operations | (2,358,388 | ) | (942,644 | ) | ||||
| Other income (expense), net: | ||||||||
| Interest expense | (38,048 | ) | (853,147 | ) | ||||
| Interest income | 12,654 | 47 | ||||||
| Other expense, net | 40 | (3,732 | ) | |||||
| Loss on extinguishment of bridge notes and bridge notes, related parties | — | (2,750,171 | ) | |||||
| Gain on extinguishment of note payable | — | 788,156 | ||||||
| Change in fair value of derivative liability on convertible notes | — | (154,000 | ) | |||||
| Change in fair value of derivative liability on bridge notes and bridge notes, related parties | — | (48,000 | ) | |||||
| Other expense, net | (25,354 | ) | (3,020,847 | ) | ||||
| Net loss | $ | (2,383,742 | ) | $ | (3,963,491 | ) | ||