000153412012/312024Q2FALSE0.00420.00420.0042http://fasb.org/us-gaap/2024#DerivativeLiabilitiesNoncurrenthttp://fasb.org/us-gaap/2024#DerivativeLiabilitiesNoncurrenthttp://www.avalotherapeutics.com/20240630#PropertyPlantAndEquipmentAndOperatingLeaseRightOfUseAssetAfterAccumulatedDepreciationAndAmortizationhttp://www.avalotherapeutics.com/20240630#PropertyPlantAndEquipmentAndOperatingLeaseRightOfUseAssetAfterAccumulatedDepreciationAndAmortizationhttp://fasb.org/us-gaap/2024#AccruedLiabilitiesAndOtherLiabilitieshttp://fasb.org/us-gaap/2024#AccruedLiabilitiesAndOtherLiabilitieshttp://fasb.org/us-gaap/2024#OtherLiabilitiesNoncurrenthttp://fasb.org/us-gaap/2024#OtherLiabilitiesNoncurrentP1Yxbrli:sharesiso4217:USDiso4217:USDxbrli:sharesxbrli:pureavtx:propertyavtx:renewal_optionavtx:class_of_stockavtx:milestoneavtx:unit00015341202024-01-012024-06-3000015341202024-08-0500015341202024-06-3000015341202023-12-310001534120us-gaap:SeriesCPreferredStockMember2023-12-310001534120us-gaap:SeriesCPreferredStockMember2024-06-300001534120us-gaap:SeriesDPreferredStockMember2024-06-300001534120us-gaap:SeriesDPreferredStockMember2023-12-310001534120us-gaap:SeriesEPreferredStockMember2023-12-310001534120us-gaap:SeriesEPreferredStockMember2024-06-300001534120us-gaap:ProductMember2024-04-012024-06-300001534120us-gaap:ProductMember2023-04-012023-06-300001534120us-gaap:ProductMember2024-01-012024-06-300001534120us-gaap:ProductMember2023-01-012023-06-3000015341202024-04-012024-06-3000015341202023-04-012023-06-3000015341202023-01-012023-06-3000015341202023-12-282023-12-2800015341202024-03-310001534120us-gaap:CommonStockMember2024-03-310001534120us-gaap:AdditionalPaidInCapitalMember2024-03-310001534120us-gaap:RetainedEarningsMember2024-03-310001534120us-gaap:AdditionalPaidInCapitalMember2024-04-012024-06-300001534120us-gaap:RetainedEarningsMember2024-04-012024-06-300001534120us-gaap:CommonStockMember2024-06-300001534120us-gaap:AdditionalPaidInCapitalMember2024-06-300001534120us-gaap:RetainedEarningsMember2024-06-300001534120us-gaap:CommonStockMember2023-12-310001534120us-gaap:AdditionalPaidInCapitalMember2023-12-310001534120us-gaap:RetainedEarningsMember2023-12-310001534120us-gaap:CommonStockMember2024-01-012024-06-300001534120us-gaap:CommonStockMemberavtx:AlmataBioTransactionMember2024-01-012024-06-300001534120us-gaap:AdditionalPaidInCapitalMemberavtx:AlmataBioTransactionMember2024-01-012024-06-300001534120avtx:AlmataBioTransactionMember2024-01-012024-06-300001534120us-gaap:SeriesCPreferredStockMemberavtx:AlmataBioTransactionMember2024-01-012024-06-300001534120us-gaap:SeriesCPreferredStockMemberus-gaap:PrivatePlacementMember2024-01-012024-06-300001534120us-gaap:SeriesDPreferredStockMemberus-gaap:PrivatePlacementMember2024-01-012024-06-300001534120us-gaap:SeriesEPreferredStockMemberus-gaap:PrivatePlacementMember2024-01-012024-06-300001534120us-gaap:AdditionalPaidInCapitalMember2024-01-012024-06-300001534120us-gaap:RetainedEarningsMember2024-01-012024-06-300001534120us-gaap:CommonStockMember2023-03-310001534120us-gaap:AdditionalPaidInCapitalMember2023-03-310001534120us-gaap:RetainedEarningsMember2023-03-3100015341202023-03-310001534120us-gaap:CommonStockMember2023-04-012023-06-300001534120us-gaap:AdditionalPaidInCapitalMember2023-04-012023-06-300001534120us-gaap:RetainedEarningsMember2023-04-012023-06-300001534120us-gaap:CommonStockMember2023-06-300001534120us-gaap:AdditionalPaidInCapitalMember2023-06-300001534120us-gaap:RetainedEarningsMember2023-06-3000015341202023-06-300001534120us-gaap:CommonStockMember2022-12-310001534120us-gaap:AdditionalPaidInCapitalMember2022-12-310001534120us-gaap:RetainedEarningsMember2022-12-3100015341202022-12-310001534120us-gaap:CommonStockMember2023-01-012023-06-300001534120us-gaap:AdditionalPaidInCapitalMember2023-01-012023-06-300001534120us-gaap:RetainedEarningsMember2023-01-012023-06-300001534120us-gaap:PreferredStockMemberus-gaap:PrivatePlacementMember2024-03-012024-03-310001534120us-gaap:PrivatePlacementMember2024-03-012024-03-310001534120us-gaap:WarrantMemberus-gaap:PrivatePlacementMember2024-06-300001534120avtx:AlmataBioTransactionMember2024-03-272024-03-270001534120us-gaap:PrivatePlacementMember2024-03-270001534120avtx:AlmataBioTransactionMember2024-03-282024-03-280001534120avtx:AlmataBioTransactionMember2024-04-012024-04-300001534120avtx:MilestoneOneMemberavtx:AlmataBioTransactionMember2024-03-280001534120avtx:MilestoneTwoMemberavtx:AlmataBioTransactionMember2024-03-280001534120avtx:AlmataBioTransactionMember2024-06-300001534120avtx:AlmataBioTransactionMember2024-03-270001534120avtx:TevaPharmaceuticalIndustriesLtd.Memberavtx:MillipredMember2021-07-012021-07-010001534120us-gaap:DiscontinuedOperationsDisposedOfBySaleMemberavtx:PediatricPortfolioMember2024-01-012024-06-300001534120us-gaap:DiscontinuedOperationsDisposedOfBySaleMember2024-06-300001534120avtx:EmployeeConsultantsAndDirectorsStockOptionsMember2024-04-012024-06-300001534120avtx:EmployeeConsultantsAndDirectorsStockOptionsMember2023-04-012023-06-300001534120avtx:EmployeeConsultantsAndDirectorsStockOptionsMember2024-01-012024-06-300001534120avtx:EmployeeConsultantsAndDirectorsStockOptionsMember2023-01-012023-06-300001534120avtx:WarrantCommonStockMember2024-04-012024-06-300001534120avtx:WarrantCommonStockMember2023-04-012023-06-300001534120avtx:WarrantCommonStockMember2024-01-012024-06-300001534120avtx:WarrantCommonStockMember2023-01-012023-06-300001534120us-gaap:SeriesCPreferredStockMember2024-04-012024-06-300001534120us-gaap:SeriesCPreferredStockMember2023-04-012023-06-300001534120us-gaap:SeriesCPreferredStockMember2024-01-012024-06-300001534120us-gaap:SeriesCPreferredStockMember2023-01-012023-06-300001534120avtx:PrefundedWarrantsMember2023-04-012023-06-300001534120avtx:PrefundedWarrantsMember2023-01-012023-06-300001534120avtx:PrefundedWarrantsMember2024-06-300001534120us-gaap:SeriesCPreferredStockMember2024-06-300001534120us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMember2024-06-300001534120us-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMember2024-06-300001534120us-gaap:FairValueInputsLevel3Memberus-gaap:FairValueMeasurementsRecurringMember2024-06-300001534120us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMember2023-12-310001534120us-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMember2023-12-310001534120us-gaap:FairValueInputsLevel3Memberus-gaap:FairValueMeasurementsRecurringMember2023-12-310001534120us-gaap:FairValueInputsLevel3Memberus-gaap:WarrantMember2024-03-310001534120us-gaap:FairValueInputsLevel3Memberus-gaap:DerivativeFinancialInstrumentsLiabilitiesMember2024-03-310001534120us-gaap:FairValueInputsLevel3Member2024-03-310001534120us-gaap:FairValueInputsLevel3Memberus-gaap:WarrantMember2024-04-012024-06-300001534120us-gaap:FairValueInputsLevel3Memberus-gaap:DerivativeFinancialInstrumentsLiabilitiesMember2024-04-012024-06-300001534120us-gaap:FairValueInputsLevel3Member2024-04-012024-06-300001534120us-gaap:FairValueInputsLevel3Memberus-gaap:WarrantMember2024-06-300001534120us-gaap:FairValueInputsLevel3Memberus-gaap:DerivativeFinancialInstrumentsLiabilitiesMember2024-06-300001534120us-gaap:FairValueInputsLevel3Member2024-06-300001534120us-gaap:FairValueInputsLevel3Memberus-gaap:WarrantMember2023-12-310001534120us-gaap:FairValueInputsLevel3Memberus-gaap:DerivativeFinancialInstrumentsLiabilitiesMember2023-12-310001534120us-gaap:FairValueInputsLevel3Member2023-12-310001534120us-gaap:FairValueInputsLevel3Memberus-gaap:WarrantMember2024-01-012024-06-300001534120us-gaap:FairValueInputsLevel3Memberus-gaap:DerivativeFinancialInstrumentsLiabilitiesMember2024-01-012024-06-300001534120us-gaap:FairValueInputsLevel3Member2024-01-012024-06-300001534120us-gaap:FairValueInputsLevel3Memberus-gaap:DerivativeFinancialInstrumentsLiabilitiesMember2023-03-310001534120us-gaap:FairValueInputsLevel3Member2023-03-310001534120us-gaap:FairValueInputsLevel3Memberus-gaap:DerivativeFinancialInstrumentsLiabilitiesMember2023-04-012023-06-300001534120us-gaap:FairValueInputsLevel3Member2023-04-012023-06-300001534120us-gaap:FairValueInputsLevel3Memberus-gaap:DerivativeFinancialInstrumentsLiabilitiesMember2023-06-300001534120us-gaap:FairValueInputsLevel3Member2023-06-300001534120us-gaap:FairValueInputsLevel3Memberus-gaap:DerivativeFinancialInstrumentsLiabilitiesMember2022-12-310001534120us-gaap:FairValueInputsLevel3Member2022-12-310001534120us-gaap:FairValueInputsLevel3Memberus-gaap:DerivativeFinancialInstrumentsLiabilitiesMember2023-01-012023-06-300001534120us-gaap:FairValueInputsLevel3Member2023-01-012023-06-300001534120us-gaap:SeriesCPreferredStockMemberus-gaap:PrivatePlacementMember2024-03-282024-03-280001534120us-gaap:WarrantMemberus-gaap:PrivatePlacementMember2024-03-282024-03-280001534120us-gaap:FairValueInputsLevel3Memberus-gaap:WarrantMember2024-06-300001534120us-gaap:FairValueInputsLevel3Memberus-gaap:MeasurementInputExpectedTermMemberus-gaap:WarrantMember2024-06-300001534120us-gaap:FairValueInputsLevel3Memberus-gaap:MeasurementInputPriceVolatilityMemberus-gaap:WarrantMember2024-06-300001534120us-gaap:FairValueInputsLevel3Memberus-gaap:MeasurementInputRiskFreeInterestRateMemberus-gaap:WarrantMember2024-06-300001534120us-gaap:FairValueInputsLevel3Memberus-gaap:MeasurementInputExpectedDividendRateMemberus-gaap:WarrantMember2024-06-300001534120us-gaap:WarrantMember2024-06-300001534120us-gaap:WarrantMember2024-03-310001534120avtx:ESTherapeuticsMemberus-gaap:DerivativeFinancialInstrumentsLiabilitiesMember2022-10-012022-12-310001534120avtx:AVTX501Member2022-12-310001534120avtx:MilestoneOneMemberavtx:AVTX007Member2022-12-310001534120avtx:MilestoneTwoMemberavtx:AVTX007Member2022-12-310001534120avtx:AVTX611Member2022-12-310001534120avtx:AVTX501AndAVTX007Memberus-gaap:DerivativeFinancialInstrumentsLiabilitiesMember2022-10-012022-12-310001534120avtx:AVTX501Memberus-gaap:DerivativeFinancialInstrumentsLiabilitiesMember2022-10-012022-12-310001534120avtx:AVTX007Memberus-gaap:DerivativeFinancialInstrumentsLiabilitiesMember2022-10-012022-12-310001534120avtx:AVTX007Memberus-gaap:DerivativeFinancialInstrumentsLiabilitiesMember2024-01-012024-06-300001534120avtx:AVTX501Memberus-gaap:DerivativeFinancialInstrumentsLiabilitiesMember2024-01-012024-06-300001534120us-gaap:DerivativeFinancialInstrumentsLiabilitiesMemberavtx:MeasurementInputProbabilityOfSuccessMemberavtx:AVTX501Member2022-12-310001534120us-gaap:DerivativeFinancialInstrumentsLiabilitiesMemberus-gaap:MeasurementInputExpectedTermMemberavtx:AVTX501Member2022-10-012022-12-310001534120us-gaap:DerivativeFinancialInstrumentsLiabilitiesMemberavtx:MeasurementInputSalesForecastPeakMemberavtx:AVTX007Member2022-10-012022-12-310001534120us-gaap:DerivativeFinancialInstrumentsLiabilitiesMemberavtx:MeasurementInputProbabilityOfSuccessMemberavtx:AVTX007Member2022-12-310001534120us-gaap:DerivativeFinancialInstrumentsLiabilitiesMemberus-gaap:MeasurementInputExpectedTermMemberavtx:AVTX007Member2022-10-012022-12-310001534120stpr:MDus-gaap:BuildingMember2024-06-300001534120stpr:MDus-gaap:BuildingMember2024-01-012024-06-300001534120stpr:PAus-gaap:BuildingMember2024-06-300001534120stpr:PAus-gaap:BuildingMember2024-01-012024-06-300001534120avtx:HorizonPowerscourtNotesMemberus-gaap:LoansPayableMember2021-06-040001534120avtx:HorizonPowerscourtNotesMemberus-gaap:LoansPayableMember2022-07-012022-09-300001534120avtx:HorizonPowerscourtNotesMemberus-gaap:LoansPayableMember2023-06-012023-06-300001534120avtx:HorizonPowerscourtNotesMemberus-gaap:LoansPayableMember2023-09-222023-09-220001534120us-gaap:WarrantMemberavtx:HorizonPowerscourtNotesMemberus-gaap:LoansPayableMember2021-06-042021-06-040001534120us-gaap:WarrantMemberavtx:HorizonPowerscourtNotesMemberus-gaap:LoansPayableMember2021-06-040001534120us-gaap:WarrantMemberavtx:HorizonPowerscourtNotesMemberus-gaap:LoansPayableMember2023-07-012023-09-300001534120us-gaap:WarrantMemberavtx:HorizonPowerscourtNotesMemberus-gaap:LoansPayableMember2024-04-012024-06-300001534120us-gaap:WarrantMemberavtx:HorizonPowerscourtNotesMemberus-gaap:LoansPayableMember2024-01-012024-06-300001534120us-gaap:PreferredStockMemberus-gaap:PrivatePlacementMember2024-03-282024-03-280001534120us-gaap:PrivatePlacementMember2024-03-282024-03-280001534120us-gaap:WarrantMemberus-gaap:PrivatePlacementMember2024-03-280001534120us-gaap:PrivatePlacementMember2024-01-012024-06-300001534120us-gaap:OtherExpenseMember2024-01-012024-06-300001534120us-gaap:OtherExpenseMemberus-gaap:PrivatePlacementMember2024-01-012024-06-300001534120us-gaap:PrivatePlacementMember2024-06-300001534120avtx:CommonStockWarrantsExpirationJune2031Memberus-gaap:CommonClassAMember2024-06-300001534120avtx:CommonStockWarrantsExercisableOnMarch282024Memberus-gaap:CommonClassAMember2024-06-300001534120us-gaap:CommonClassAMember2024-06-300001534120avtx:A2016ThirdAmendedPlanMember2024-01-012024-06-300001534120avtx:A2016ThirdAmendedPlanMember2024-01-012024-01-010001534120avtx:A2016ThirdAmendedPlanMember2024-06-300001534120us-gaap:StockOptionMemberavtx:A2016ThirdAmendedPlanMember2024-01-012024-06-300001534120us-gaap:ShareBasedPaymentArrangementEmployeeMemberavtx:A2016ThirdAmendedPlanMemberus-gaap:StockOptionMembersrt:MaximumMember2024-01-012024-06-300001534120avtx:A2016ThirdAmendedPlanMemberus-gaap:StockOptionMembersrt:DirectorMembersrt:MinimumMember2024-01-012024-06-300001534120avtx:A2016ThirdAmendedPlanMemberus-gaap:StockOptionMembersrt:DirectorMembersrt:MaximumMember2024-01-012024-06-300001534120us-gaap:ResearchAndDevelopmentExpenseMember2024-04-012024-06-300001534120us-gaap:ResearchAndDevelopmentExpenseMember2023-04-012023-06-300001534120us-gaap:ResearchAndDevelopmentExpenseMember2024-01-012024-06-300001534120us-gaap:ResearchAndDevelopmentExpenseMember2023-01-012023-06-300001534120us-gaap:GeneralAndAdministrativeExpenseMember2024-04-012024-06-300001534120us-gaap:GeneralAndAdministrativeExpenseMember2023-04-012023-06-300001534120us-gaap:GeneralAndAdministrativeExpenseMember2024-01-012024-06-300001534120us-gaap:GeneralAndAdministrativeExpenseMember2023-01-012023-06-300001534120avtx:ServiceBasedOptionsMember2023-12-310001534120avtx:ServiceBasedOptionsMember2023-01-012023-12-310001534120avtx:ServiceBasedOptionsMember2024-01-012024-06-300001534120avtx:ServiceBasedOptionsMember2024-06-300001534120avtx:ServiceBasedOptionsMember2024-06-242024-06-240001534120avtx:ServiceBasedOptionsMember2024-06-240001534120avtx:ServiceBasedOptionsMember2024-04-012024-06-300001534120avtx:ServiceBasedOptionsMemberus-gaap:SubsequentEventMember2024-07-152024-07-150001534120avtx:ServiceBasedOptionsMemberus-gaap:SubsequentEventMember2024-07-150001534120avtx:EmployeeStockPurchasePlanESPPMember2016-04-052016-04-050001534120avtx:EmployeeStockPurchasePlanESPPMember2016-04-050001534120avtx:EmployeeStockPurchasePlanESPPMember2024-01-012024-01-010001534120avtx:EmployeeStockPurchasePlanESPPMember2024-06-300001534120avtx:LillyLicenseAgreementMemberavtx:AVTX009LillyLicenseAgreementMember2024-03-270001534120avtx:KyowaKirinCoLtdKKCMemberavtx:AVTX002KKCLicenseAgreementMember2021-03-250001534120avtx:MilestoneOneMemberavtx:KyowaKirinCoLtdKKCMemberavtx:AVTX002KKCLicenseAgreementMember2021-03-250001534120avtx:MilestoneTwoMemberavtx:KyowaKirinCoLtdKKCMemberavtx:AVTX002KKCLicenseAgreementMember2021-03-250001534120avtx:KyowaKirinCoLtdKKCMemberavtx:AVTX002KKCLicenseAgreementMember2024-01-012024-06-300001534120avtx:KyowaKirinCoLtdKKCMemberavtx:AVTX002KKCLicenseAgreementMember2024-06-300001534120avtx:CHOPLicenseAgreementMemberavtx:AVTX002KKCLicenseAgreementMember2020-02-030001534120avtx:CHOPLicenseAgreementMemberavtx:AVTX002KKCLicenseAgreementMember2020-01-012020-12-310001534120avtx:SanfordBurnhamPrebysMedicalDiscoveryInstituteMemberavtx:AVTX008SanfordBurnhamPrebysLicenseAgreementMember2021-06-210001534120avtx:SanfordBurnhamPrebysMedicalDiscoveryInstituteMemberavtx:AVTX008SanfordBurnhamPrebysLicenseAgreementMember2021-06-300001534120avtx:MilestoneOneMemberavtx:SanfordBurnhamPrebysMedicalDiscoveryInstituteMemberavtx:AVTX008SanfordBurnhamPrebysLicenseAgreementMember2021-06-210001534120avtx:MilestoneTwoMemberavtx:SanfordBurnhamPrebysMedicalDiscoveryInstituteMemberavtx:AVTX008SanfordBurnhamPrebysLicenseAgreementMember2021-06-210001534120avtx:SanfordBurnhamPrebysMedicalDiscoveryInstituteMemberavtx:AVTX008SanfordBurnhamPrebysLicenseAgreementMember2024-01-012024-06-300001534120avtx:SanfordBurnhamPrebysMedicalDiscoveryInstituteMemberavtx:AVTX008SanfordBurnhamPrebysLicenseAgreementMember2024-06-300001534120avtx:AstellasPharmaIncAstellasMemberavtx:AVTX006AstellasLicenseAgreementMember2024-06-300001534120avtx:AstellasPharmaIncAstellasMemberavtx:AVTX006AstellasLicenseAgreementMember2024-01-012024-06-300001534120avtx:AltoMemberavtx:AVTX301OutLicenseMember2021-05-280001534120avtx:AltoMemberavtx:AVTX301OutLicenseMember2024-06-300001534120avtx:MilestoneOneMemberavtx:ESMemberavtx:AVTX406LicenseAssignmentMember2021-06-090001534120avtx:MilestoneTwoMemberavtx:ESMemberavtx:AVTX406LicenseAssignmentMember2021-06-090001534120avtx:ESMemberavtx:AVTX406LicenseAssignmentMember2024-06-300001534120avtx:AUGTherapeuticsLLCMemberavtx:AVTX800SeriesAssetSaleMemberavtx:PurchaseAgreementMember2023-10-272023-10-270001534120avtx:AUGTherapeuticsLLCMemberavtx:AVTX800SeriesAssetSaleMemberavtx:PurchaseAgreementMember2023-10-270001534120avtx:MilestoneTwoMemberavtx:AVTX009Member2024-06-300001534120avtx:AeviGenomicMedicineIncMember2020-01-012020-03-310001534120avtx:MilestoneOneMemberavtx:AeviGenomicMedicineIncMember2020-02-030001534120avtx:AeviGenomicMedicineIncMember2024-01-012024-06-300001534120avtx:MilestoneTwoMemberavtx:AeviGenomicMedicineIncMember2020-02-0300015341202019-07-012019-07-310001534120avtx:TRISPharmaMemberavtx:KarbinalAgreementMember2018-01-012018-12-31
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, DC 20549
FORM 10-Q
| | | | | |
☑
| QUARTERLY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
for the quarterly period ended June 30, 2024 |
| OR |
| ☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
COMMISSION FILE NUMBER: 001-37590
AVALO THERAPEUTICS, INC.
(Exact name of registrant as specified in its charter)
| | | | | |
Delaware (State of incorporation) | 45-0705648 (I.R.S. Employer Identification No.) |
540 Gaither Road, Suite 400 Rockville, Maryland 20850 (Address of principal executive offices) | (410) 522-8707 (Registrant’s telephone number, including area code) |
Securities registered pursuant to Section 12(b) of the Act:
| | | | | | | | |
| Title of each class | Trading Symbol | Name of each exchange on which registered |
| Common Stock, $0.001 par value | AVTX | Nasdaq Capital Market |
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☑ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☑ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b‑2 of the Exchange Act.
| | | | | | | | |
Large accelerated filer ☐ | | Accelerated filer ☐ |
Non-accelerated filer ☑ | | Smaller reporting company ☑ |
Emerging growth company ☐ | | |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b‑2 of the Exchange Act). Yes ☐ No ☑
As of August 5, 2024, the registrant had 1,034,130 shares of common stock outstanding.
AVALO THERAPEUTICS, INC.
FORM 10-Q
For the Quarter Ended June 30, 2024
TABLE OF CONTENTS
PART I - FINANCIAL INFORMATION
Item 1. Financial Statements.
AVALO THERAPEUTICS, INC. and SUBSIDIARIES
Condensed Consolidated Balance Sheets
(In thousands, except share and per share data)
| | | | | | | | | | | | | | |
| | June 30, 2024 | | December 31, 2023 |
| | (unaudited) | | |
| Assets | | | | |
| Current assets: | | | | |
| Cash and cash equivalents | | $ | 93,426 | | | $ | 7,415 | |
| | | | |
| Other receivables | | 33 | | | 136 | |
| | | | |
| Prepaid expenses and other current assets | | 2,435 | | | 843 | |
| Restricted cash, current portion | | — | | | 1 | |
| Total current assets | | 95,894 | | | 8,395 | |
| Property and equipment, net | | 1,780 | | | 1,965 | |
| Goodwill | | 10,502 | | | 10,502 | |
| Restricted cash, net of current portion | | 131 | | | 131 | |
| Total assets | | $ | 108,307 | | | $ | 20,993 | |
| Liabilities, mezzanine equity and stockholders’ (deficit) equity | | | | |
| Current liabilities: | | | | |
| Accounts payable | | $ | 654 | | | $ | 446 | |
| Accrued expenses and other current liabilities | | 7,888 | | | 4,172 | |
| Warrant liability | | 82,855 | | | — | |
| Contingent consideration | | 5,000 | | | — | |
| Total current liabilities | | 96,397 | | | 4,618 | |
| Royalty obligation | | 2,000 | | | 2,000 | |
| Deferred tax liability, net | | 168 | | | 155 | |
| Derivative liability | | 10,710 | | | 5,550 | |
| Other long-term liabilities | | 1,183 | | | 1,366 | |
| Total liabilities | | 110,458 | | | 13,689 | |
| Mezzanine equity: | | | | |
Series C Preferred Stock—$0.001 par value; 34,326 and 0 shares of Series C Preferred Stock authorized at June 30, 2024 and December 31, 2023, respectively; 22,358 and 0 shares of Series C Preferred Stock issued and outstanding at June 30, 2024 and December 31, 2023, respectively | | 11,457 | | | — | |
Series D Preferred Stock—$0.001 par value; 1 and 0 shares of Series D Preferred Stock authorized at June 30, 2024 and December 31, 2023, respectively; 1 and 0 shares of Series D Preferred Stock issued and outstanding at June 30, 2024 and December 31, 2023, respectively | | — | | | — | |
Series E Preferred Stock—$0.001 par value; 1 and 0 shares of Series E Preferred Stock authorized at June 30, 2024 and December 31, 2023, respectively; 1 and 0 shares of Series E Preferred Stock issued and outstanding at June 30, 2024 and December 31, 2023, respectively | | — | | | — | |
| Stockholders’ (deficit) equity: | | | | |
Common stock—$0.001 par value; 200,000,000 shares authorized at June 30, 2024 and December 31, 2023; 1,034,130 and 801,746 shares issued and outstanding at June 30, 2024 and December 31, 2023, respectively | | 1 | | | 1 | |
| Additional paid-in capital | | 344,352 | | | 342,437 | |
| Accumulated deficit | | (357,961) | | | (335,134) | |
| Total stockholders’ (deficit) equity | | (13,608) | | | 7,304 | |
| Total liabilities, mezzanine equity and stockholders’ (deficit) equity | | $ | 108,307 | | | $ | 20,993 | |
See accompanying notes to the unaudited condensed consolidated financial statements.
AVALO THERAPEUTICS, INC. and SUBSIDIARIES
Condensed Consolidated Statements of Operations and Comprehensive Income (Loss) (Unaudited)
(In thousands, except per share data)
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Three Months Ended | | Six Months Ended |
| | | June 30, | | June 30, |
| | | 2024 | | 2023 | | 2024 | | 2023 |
| Revenues: | | | | | | | | |
| Product revenue, net | | $ | — | | | $ | 643 | | | $ | — | | | $ | 1,117 | |
| | | | | | | | |
| Total revenues, net | | — | | | 643 | | | — | | | 1,117 | |
| | | | | | | | |
| Operating expenses: | | | | | | | | |
| Cost of product sales | | 343 | | | 708 | | | 263 | | | 1,259 | |
| Research and development | | 4,601 | | | 4,658 | | | 6,716 | | | 10,667 | |
| Acquired in-process research and development | | 103 | | | — | | | 27,641 | | | — | |
| General and administrative | | 4,528 | | | 2,427 | | | 7,721 | | | 5,134 | |
| | | | | | | | |
| Total operating expenses | | 9,575 | | | 7,793 | | | 42,341 | | | 17,060 | |
| | (9,575) | | | (7,150) | | | (42,341) | | | (15,943) | |
| Other income (expense): | | | | | | | | |
| Excess of initial warrant fair value over private placement proceeds | | — | | | — | | | (79,276) | | | — | |
| Change in fair value of warrant liability | | 112,046 | | | — | | | 112,046 | | | — | |
| Private placement transaction costs | | — | | | — | | | (9,220) | | | — | |
| Change in fair value of derivative liability | | (5,040) | | | (40) | | | (5,160) | | | (220) | |
| Interest income (expense), net | | 1,039 | | | (996) | | | 1,138 | | | (1,945) | |
| Other expense, net | | — | | | — | | | — | | | (25) | |
| Total other income (expense), net | | 108,045 | | | (1,036) | | | 19,528 | | | (2,190) | |
| Income (loss) before taxes | | 98,470 | | | (8,186) | | | (22,813) | | | (18,133) | |
| Income tax expense | | 7 | | | 7 | | | 14 | | | 15 | |
| Net income (loss) | | $ | 98,463 | | | $ | (8,193) | | | $ | (22,827) | | | $ | (18,148) | |
Net income (loss) per share of common stock1: | | | | | | | | |
| Basic | | $ | 4.21 | | | $ | (140.73) | | | $ | (24.11) | | | $ | (338.85) | |
| Diluted | | $ | (14.07) | | | $ | (140.73) | | | $ | (30.63) | | | $ | (338.85) | |
| | | | | | | | |
| | | | | | | | |
| | | | | | | | |
1 Amounts for prior periods presented have been retroactively adjusted to reflect the 1-for-240 reverse stock split effected on December 28, 2023. See Note 1 for details.
See accompanying notes to the unaudited condensed consolidated financial statements.
AVALO THERAPEUTICS, INC. and SUBSIDIARIES
Condensed Consolidated Statements of Preferred Stock and Changes in Stockholders’ Deficit (Unaudited)
(In thousands, except share amounts)
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Preferred Stock | | | Common stock | | | | Additional paid-in | | Accumulated | Total stockholders’ |
| | Shares | Amount | | | Shares | | Amount | | | | | | capital | | deficit | | (deficit) equity |
| Three Months Ended June 30, 2024 | | | | | | | | | | | | | | | | | |
| Balance, March 31, 2024 | 22,360 | | 11,457 | | | | 1,034,130 | | | $ | 1 | | | | | | | $ | 343,881 | | | $ | (456,424) | | | $ | (112,542) | |
| Stock-based compensation | — | | — | | | | — | | | — | | | | | | | 471 | | | — | | | 471 | |
| Net income | — | | — | | | | — | | | — | | | | | | | — | | | 98,463 | | | 98,463 | |
| Balance, June 30, 2024 | 22,360 | | $ | 11,457 | | | | 1,034,130 | | | $ | 1 | | | | | | | $ | 344,352 | | | $ | (357,961) | | | $ | (13,608) | |
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Preferred Stock | | | Common stock | | | | Additional paid-in | | Accumulated | Total stockholders’ |
| | Shares | Amount | | | Shares | | Amount | | | | | | capital | | deficit | | (deficit) equity |
| Six Months Ended June 30, 2024 | | | | | | | | | | | | | | | | | |
| Balance, December 31, 2023 | — | | — | | | | 801,746 | | | $ | 1 | | | | | | | $ | 342,437 | | | $ | (335,134) | | | $ | 7,304 | |
| Impact of reverse split fractional share round-up | — | | — | | | | 60,779 | | | — | | | | | | | — | | | — | | | — | |
| Issuance of common stock pursuant to Almata Transaction | — | | — | | | | 171,605 | | | — | | | | | | | 815 | | | — | | | 815 | |
| Issuance of Series C Preferred Stock pursuant to Almata Transaction | 2,412 | | 11,457 | | | | — | | | — | | | | | | | — | | | — | | | — | |
| Issuance of Series C Preferred Stock in private placement | 19,946 | | — | | | | — | | | — | | | | | | | — | | | — | | | — | |
| Issuance of Series D Preferred Stock in private placement | 1 | | — | | | | — | | | — | | | | | | | — | | | — | | | — | |
| Issuance of Series E Preferred Stock in private placement | 1 | | — | | | | — | | | — | | | | | | | — | | | — | | | — | |
| Stock-based compensation | — | | — | | | | — | | | — | | | | | | | 1,100 | | | — | | | 1,100 | |
| Net loss | — | | — | | | | — | | | — | | | | | | | — | | | (22,827) | | | (22,827) | |
| Balance, June 30, 2024 | 22,360 | | $ | 11,457 | | | | 1,034,130 | | | $ | 1 | | | | | | | $ | 344,352 | | | $ | (357,961) | | | $ | (13,608) | |
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Preferred Stock | | | Common stock | | | | Additional paid-in | | Accumulated | Total stockholders’ |
| | Shares | Amount | | | Shares1 | | Amount1 | | | | | | capital1 | | deficit | | deficit |
| Three Months Ended June 30, 2023 | | | | | | | | | | | | | | | | | |
| Balance, March 31, 2023 | — | | — | | | | 55,003 | | | $ | — | | | | | | | $ | 307,512 | | | $ | (313,779) | | | $ | (6,267) | |
| Issuance of common shares pursuant to ATM Program, net | — | | — | | | | 8,520 | | | — | | | | | | | 6,531 | | | — | | | 6,531 | |
| Retirement of common shares in exchange for pre-funded warrants | — | | — | | | | (5,417) | | | — | | | | | | | (3,874) | | | 234 | | | (3,640) | |
| Issuance of pre-funded warrants in exchange for retirement of common shares | — | | — | | | | — | | | — | | | | | | | 3,640 | | | — | | | 3,640 | |
| Exercise of pre-funded warrants for common shares | — | | — | | | | 301 | | | — | | | | | | | — | | | — | | | — | |
| Shares purchased through employee stock purchase plan | — | | — | | | | 82 | | | — | | | | | | | 67 | | | — | | | 67 | |
| Stock-based compensation | — | | — | | | | — | | | — | | | | | | | 893 | | | — | | | 893 | |
| Net loss | — | | — | | | | — | | | — | | | | | | | — | | | (8,193) | | | (8,193) | |
| Balance, June 30, 2023 | — | | $ | — | | | | 58,489 | | | $ | — | | | | | | | $ | 314,769 | | | $ | (321,738) | | | $ | (6,969) | |
| | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Preferred Stock | | | Common stock | | | | Additional paid-in | | Accumulated | Total stockholders’ |
| | Shares | Amount | | | Shares1 | | Amount1 | | | | | | capital1 | | deficit | | deficit |
| Six Months Ended June 30, 2023 | | | | | | | | | | | | | | | | | |
| Balance, December 31, 2022 | — | | — | | | | 39,294 | | | $ | — | | | | | | | $ | 292,909 | | | $ | (303,824) | | | $ | (10,915) | |
| Issuance of common stock and warrants in underwritten public offering, net | — | | — | | | | 15,709 | | | — | | | | | | | 13,748 | | | — | | | 13,748 | |
| Issuance of common shares pursuant to ATM Program, net | — | | — | | | | 8,520 | | | — | | | | | | | 6,531 | | | — | | | 6,531 | |
| Retirement of common shares in exchange for pre-funded warrants | — | | — | | | | (5,417) | | | — | | | | | | | (3,874) | | | 234 | | | (3,640) | |
| Issuance of pre-funded warrants in exchange for retirement of common shares | — | | — | | | | — | | | — | | | | | | | 3,640 | | | — | | | 3,640 | |
| Exercise of pre-funded warrants for common shares | — | | — | | | | 301 | | | — | | | | | | | — | | | — | | | — | |
| Shares purchased through employee stock purchase plan | — | | — | | | | 82 | | | — | | | | | | | 67 | | | — | | | 67 | |
| Stock-based compensation | — | | — | | | | — | | | — | | | | | | | 1,748 | | | — | | | 1,748 | |
| Net loss | — | | — | | | | — | | | — | | | | | | | — | | | (18,148) | | | (18,148) | |
| Balance, June 30, 2023 | — | | $ | — | | | | 58,489 | | | $ | — | | | | | | | $ | 314,769 | | | $ | (321,738) | | | $ | (6,969) | |
| | | | | | | | | | | | | | | | | |
1 Amounts for prior periods presented have been retroactively adjusted to reflect the 1-for-240 reverse stock split effected on December 28, 2023. See Note 1 for details.
See accompanying notes to the unaudited condensed consolidated financial statements.
AVALO THERAPEUTICS, INC. and SUBSIDIARIES
Condensed Consolidated Statements of Cash Flows (Unaudited)
(Amounts in thousands) | | | | | | | | | | | | | | |
| | | Six Months Ended June 30, |
| | | 2024 | | 2023 |
| Operating activities | | | | |
| Net loss | | $ | (22,827) | | | $ | (18,148) | |
| Adjustments to reconcile net loss used in operating activities: | | | | |
| Depreciation and amortization | | 67 | | | 74 | |
| Stock-based compensation | | 1,100 | | | 1,748 | |
| Acquired in-process research and development | | 27,641 | | | — | |
| Excess of initial warrant fair value over private placement proceeds | | 79,276 | | | — | |
| Change in fair value of warrant liability | | (112,046) | | | — | |
| Transaction costs paid pursuant to private placement | | 7,485 | | | — | |
| Contingent consideration paid pursuant to Almata Transaction | | (7,500) | | | — | |
| Transaction costs payable upon exercise of warrants issued in private placement | | 1,734 | | | — | |
| Change in fair value of derivative liability | | 5,160 | | | 220 | |
| Accretion of debt discount | | — | | | 699 | |
| Deferred taxes | | 14 | | | 15 | |
| Changes in assets and liabilities: | | | | |
| Accounts receivable, net | | — | | | (38) | |
| Other receivables | | 103 | | | 1,913 | |
| | | | |
| Inventory, net | | — | | | 2 | |
| Prepaid expenses and other assets | | (1,592) | | | 155 | |
| | | | |
| Lease incentive | | 107 | | | 158 | |
| Accounts payable | | 208 | | | (2,131) | |
| | | | |
| | | | |
| Deferred revenue | | — | | | (88) | |
| Accrued expenses and other liabilities | | (1,254) | | | (5,627) | |
| Lease liability, net | | (161) | | | (30) | |
| Net cash used in operating activities | | (22,485) | | | (21,078) | |
| Investing activities | | | | |
| Cash assumed from Almata Transaction | | 356 | | | — | |
| Leasehold improvements | | — | | | (158) | |
| Disposal of property and equipment | | — | | | 25 | |
| | | | |
| Net cash provided by (used in) investing activities | | 356 | | | (133) | |
| Financing activities | | | | |
| | | | |
| Proceeds from private placement investment, gross | | 115,625 | | | — | |
| Transaction costs paid pursuant to private placement | | (7,485) | | | — | |
| Proceeds from issuance of common stock and pre-funded warrants in underwritten public offering, net | | — | | | 13,748 | |
| Prepayment on Notes | | — | | | (6,000) | |
| Proceeds from issuance of common stock pursuant to ATM Program, net | | — | | | 6,531 | |
| | | | |
| | | | |
| Proceeds from issuance of common stock under employee stock purchase plan | | — | | | 67 | |
| Net cash provided by financing activities | | 108,140 | | | 14,346 | |
| Increase in cash, cash equivalents and restricted cash | | 86,011 | | | (6,865) | |
| Cash, cash equivalents, and restricted cash at beginning of period | | 7,546 | | | 13,318 | |
| Cash, cash equivalents, and restricted cash at end of period | | $ | 93,557 | | | $ | 6,453 | |
| Supplemental disclosures of cash flow information | | | | |
| Cash paid for interest | | $ | — | | | $ | 1,439 | |
| | | | |
| Supplemental disclosures of non-cash activities | | | | |
| Issuance of common stock and Series C Preferred Stock pursuant to Almata Transaction | | $ | 12,727 | | | $ | — | |
| Fair value of common stock retired in exchange for issuance of pre-funded warrants | | $ | — | | | $ | 3,640 | |
The following table provides a reconciliation of cash, cash equivalents and restricted cash reported within the condensed consolidated balance sheets that sum to the total of the same such amounts shown in the condensed consolidated statements of cash flows (in thousands):
| | | | | | | | | | | | | | |
| | June 30, |
| | 2024 | | 2023 |
| Cash and cash equivalents | | $ | 93,426 | | | $ | 6,307 | |
| Restricted cash, current | | — | | | 15 | |
| Restricted cash, non-current | | 131 | | | 131 | |
| Total cash, cash equivalents and restricted cash | | $ | 93,557 | | | 6,453 | |
See accompanying notes to the unaudited condensed consolidated financial statements.
AVALO THERAPEUTICS, INC. and SUBSIDIARIES
Notes to Unaudited Condensed Consolidated Financial Statements
1. Business
Avalo Therapeutics, Inc. (the “Company,” “Avalo” or “we”) is a clinical stage biotechnology company focused on the treatment of immune dysregulation. Avalo’s lead asset is AVTX-009, an anti-IL-1β monoclonal antibody (“mAb”), targeting inflammatory diseases. Avalo also has two additional drug candidates, which include quisovalimab (anti-LIGHT mAb) and AVTX-008 (BTLA agonist fusion protein).
Avalo was incorporated in Delaware and commenced operation in 2011, and completed its initial public offering in October 2015.
Liquidity
Since inception, we have incurred significant operating and cash losses from operations. We have primarily funded our operations to date through sales of equity securities, out-licensing transactions and sales of assets.
For the six months ended June 30, 2024, Avalo generated a net loss of $22.8 million and negative cash flows from operations of $22.5 million. As of June 30, 2024, Avalo had $93.4 million in cash and cash equivalents. In the first quarter of 2024, the Company closed a private placement investment consisting of an initial upfront gross investment of $115.6 million (net proceeds were $108.1 million after deducting transaction costs) and potentially up to an additional $69.4 million of gross proceeds upon the exercise of warrants issued in the financing.
Based on our current operating plans, we expect that our existing cash and cash equivalents are sufficient to fund operations for at least twelve months from the filing date of this Quarterly Report on Form 10-Q and we expect current cash on hand to fund operations into 2027. The Company closely monitors its cash and cash equivalents and seeks to balance the level of cash and cash equivalents with our projected needs to allow us to withstand periods of uncertainty relative to the availability of funding on favorable terms. We may satisfy any future cash needs through sales of equity securities under the Company’s at-the-market program or other equity financings, out-licensing transactions, strategic alliances/collaborations, sale of programs, and/or mergers and acquisitions. There can be no assurance that any financing or business development initiatives can be realized by the Company, or if realized, what the terms may be. To the extent that we raise capital through the sale of equity, the ownership interest of our existing stockholders will be diluted, and the terms may include liquidation or other preferences that adversely affect the rights of our stockholders. Further, if the Company raises additional funds through collaborations, strategic alliances or licensing arrangements with third parties, the Company might have to relinquish valuable rights to its technologies, future revenue streams, research programs or product candidates.
2. Basis of Presentation and Significant Accounting Policies
Basis of Presentation
The Company’s unaudited condensed consolidated financial statements have been prepared in accordance with U.S. generally accepted accounting principles (“GAAP”). Any reference in these notes to applicable guidance is meant to refer to the authoritative GAAP as found in the Accounting Standards Codification (“ASC”) and Accounting Standards Updates (“ASU”) of the Financial Accounting Standards Board (“FASB”).
In the opinion of management, the accompanying unaudited condensed consolidated financial statements include all adjustments, consisting of normal recurring adjustments, which are necessary to present fairly the Company’s financial position, results of operations, and cash flows. The condensed consolidated balance sheet at December 31, 2023 has been derived from audited financial statements at that date. The interim results of operations are not necessarily indicative of the results that may occur for the full fiscal year. Certain information and footnote disclosure normally included in the financial statements prepared in accordance with GAAP have been condensed or omitted pursuant to instructions, rules, and regulations prescribed by the United States Securities and Exchange Commission (“SEC”).
The Company believes that the disclosures provided herein are adequate to make the information presented not misleading when these unaudited condensed consolidated financial statements are read in conjunction with the December 31, 2023 audited consolidated financial statements.
On December 28, 2023, Avalo effected a 1-for-240 reverse stock split of the outstanding shares of the Company’s common stock and began trading on a split-adjusted basis on December 29, 2023. The Company retroactively applied the reverse stock split to common share and per share amounts for periods prior to December 28, 2023, including the unaudited condensed consolidated financial statements for the three and six months ended June 30, 2023. Additionally, pursuant to their terms, a proportionate adjustment was made to the per share exercise price and number of shares issuable under all of the Company’s outstanding options and warrants, and the number of shares authorized for issuance pursuant to the Company’s equity incentive plans have been reduced proportionately. Avalo retroactively applied such adjustments in the notes to consolidated financial statements for periods presented prior to December 28, 2023, including the three and six months ended June 30, 2023. The reverse stock split did not reduce the number of authorized shares of common and preferred stock and did not alter the par value.
Unless otherwise indicated, all amounts in the following tables are in thousands except share and per share amounts.
Significant Accounting Policies
During the three months ended June 30, 2024, there were no significant changes to the Company’s summary of significant accounting policies contained in the Company’s Annual Report on Form 10-K for the year ended December 31, 2023, as filed with the SEC on March 29, 2024, and the Company’s Quarterly Report on Form 10-Q for the quarter ended March 31, 2024, as filed with the SEC on May 13, 2024 (as amended on July 11, 2024).
3. Asset Acquisition
Almata Transaction
On March 27, 2024, the Company acquired AVTX-009, an anti-IL-1β mAb, through a merger with AlmataBio, Inc. (“AlmataBio”) with and into its wholly owned subsidiary (the “Almata Transaction”). The Company’s acquisition of AlmataBio was structured as a stock-for-stock transaction whereby all outstanding equity interests in AlmataBio were exchanged in a merger for a combination of the Company’s common stock and shares of the Company’s non-voting convertible preferred stock (the “Series C Preferred Stock”), resulting in the issuance of 171,605 shares of Company common stock and 2,412 shares of Series C Preferred Stock. Subject to Company stockholder approval (which is being sought at the Company’s annual meeting of stockholders scheduled to be held on August 13, 2024), each share of Company Series C Preferred Stock (i) issued to former AlmataBio stockholders and (ii) pursuant to the March 2024 financing will automatically convert to 1,000 shares of common stock, subject to certain beneficial ownership limitations. The Series C Preferred Stock holds no voting rights.
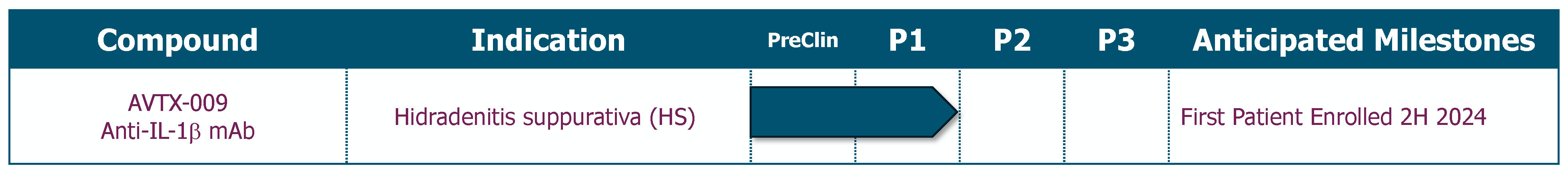
In addition to the shares issued, a cash payment of $7.5 million was due to the former AlmataBio stockholders upon the closing of a private placement investment. The private placement closed on March 28, 2024 and the Company paid the $7.5 million in April 2024. The Company is also required to pay potential development milestone payments to the former AlmataBio stockholders, including $5.0 million due upon the first patient dosed in a Phase 2 trial in patients with hidradenitis suppurativa (“HS”) for AVTX-009 and $15.0 million due upon the first patient dosed in a Phase 3 trial for AVTX-009, both of which are payable in cash, Avalo stock, or a combination thereof at the election of the former AlmataBio stockholders, subject to the terms and conditions of the definitive merger agreement.
The Company has been determined to be the acquiring company for accounting purposes. In connection with the Almata Transaction, substantially all of the consideration paid is allocable to the fair value of acquired in-process research and development (“IPR&D”), specifically AVTX-009, and as such the acquisition is treated as an asset acquisition. The Company initially recognized AlmataBio’s assets and liabilities by allocating the accumulated cost of the acquisition based on their relative fair values, as estimated by management. The net assets acquired as of the transaction date have been combined with the assets, liabilities, and results of operations of the Company on consummation of the Almata Transaction. In accordance with ASC 730, Research and Development, the portion of the consideration allocated to the acquired IPR&D, specifically AVTX-009, based on its relative fair value, is included as an operating expense as there is no alternative future use.
Below is a summary of the total consideration, assets acquired and the liabilities assumed in connection with the Almata Transaction (in thousands):
| | | | | | | | | | |
| | Six Months Ended June 30, 2024 |
Stock consideration1 | | $ | 12,272 | | | |
Milestone payment due upon close of private placement investment2 | | 7,500 | | | |
Milestone payment due upon first patient dosed in a Phase 2 trial2 | | 5,000 | | | |
| Transaction costs | | 2,402 | | | |
| Total GAAP Purchase Price at Close | | $ | 27,174 | | | |
| | | | |
| Acquired IPR&D | | $ | 27,641 | | | |
| Cash | | 356 | | | |
| Accrued expenses and other current liabilities | | (823) | | | |
| Total net assets acquired and liabilities assumed | | $ | 27,174 | | | |
1 Equal to the aggregate common shares issued of 171,605 and the aggregate preferred shares issued of 2,412 (as-convertible to 2,412,000 shares of common stock), multiplied by the Company’s closing stock price of $4.75 on March 27, 2024.
2 Avalo deemed these milestones probable and estimable as of the transaction close date and therefore included them as part of the GAAP purchase price at close. The first milestone payment due upon the close of the private placement investment was met on March 28, 2024 and was paid on April 1, 2024.
The cost to acquire the IPR&D asset related to AVTX-009 was expensed on the date of the Almata Transaction as it was determined to have no future alternative use. Accordingly, costs associated with the Almata Transaction to acquire the asset were expensed as incurred in acquired IPR&D.
4. Revenue
The Company’s license and supply agreement for Millipred®, an oral prednisolone indicated across a wide variety of inflammatory conditions, ended on September 30, 2023, and therefore there was no net product revenue for the six months ended June 30, 2024. Avalo considered Millipred® a non-core asset. Historically, the Company sold Millipred® in the United States primarily through wholesale distributors, who accounted for substantially all of the Company’s net product revenues and trade receivables. For the six months ended June 30, 2023, the Company recognized net product revenue of $1.1 million.
The Company will continue to monitor estimates for commercial liabilities for Millipred®, such as sales returns. As additional information becomes available, the Company could recognize expense (or a benefit) for differences between actuals or updated estimates to the reserves previously recognized. Pursuant the Millipred® license and supply agreement, Avalo was required to pay the supplier fifty percent of the net profit of the Millipred® product following each calendar quarter, subject to a $0.5 million quarterly minimum payment dependent on Avalo reaching certain net profit amounts as stipulated in the agreement. The profit share commenced on July 1, 2021 and ended on September 30, 2023. Within twenty-five months of September 30, 2023, the net profit share is subject to a reconciliation process where estimated deductions to arrive at net profit will be trued-up to actuals and could result in Avalo owing additional amounts to the supplier or vice versa, which would be recognized in cost of product sales.
Aytu BioScience, Inc. (“Aytu”), to which the Company sold its rights, title, and interests in assets relating to certain commercialized products in 2019 (the “Aytu Transaction”), managed Millipred® commercial operations until August 31, 2021 pursuant to a transition services agreements, which included managing the third-party logistics provider. As a result, Aytu collected cash on behalf of Avalo for revenue generated by sales of Millipred® from the second quarter of 2020 through the third quarter of 2021. The transition services agreement allows Aytu to withhold up to $1.0 million until December of 2024 and requires the retention amount to remain at $1.0 million as of June 1, 2024 pursuant to a reconciliation due on such date. In the second quarter of 2022, Avalo fully reserved the receivable as a result of Aytu’s conclusion within its Quarterly Report on Form 10-Q for the quarter ended June 30, 2022 that substantial doubt existed with respect to its ability to continue as a going concern within one year after the date those financial statements were issued. As of June 30, 2024, the total receivable balance was approximately $1.0 million and remains fully reserved. We will continue to reassess its collectability each reporting period. As Aytu incurs returns and other commercial liabilities on Avalo’s behalf, the receivable reserve is relieved, creating an offset to cost of product sales. Additionally, at certain points of time, the transition service agreement requires Avalo to pay up to the $1.0 million stated retention amount to provide Aytu with a balance against returns paid on Avalo’s behalf. Additional amounts owed increase the receivable and corresponding receivable reserve through an increase to cost of product sales.
5. Net Income (Loss) Per Share
The Company had two classes of stock outstanding during the three and six months ended June 30, 2024, common stock and preferred stock, and had only common stock outstanding during the three and six months ended June 30, 2023. The Company computes net income (loss) per share using the two-class method, as the Series C Preferred Stock participates in distributions with the Company’s common stock. The two-class method of computing net income (loss) per share is an earnings allocation formula that determines net income (loss) for common stock and any participating securities according to dividends declared and participation rights in undistributed earnings. As the Company had net income for the three months ended June 30, 2024, the two-class method of calculating net income per share allocates a portion of the net income to the participating securities. However, as the Company is in a cumulative net loss position for the six months ended June 30, 2024, the two-class method of computing net loss per share results in no allocation of undistributed losses to participating securities.
Basic net income (loss) per share for common stock is computed by dividing the sum of distributed and undistributed earnings by the weighted average number of shares outstanding for the period. The weighted average number of common shares outstanding as of June 30, 2023, includes the weighted average effect of pre-funded warrants, the exercise of which required nominal consideration for the delivery of the shares of common stock. There were no pre-funded warrants outstanding as of June 30, 2024.
Diluted net income (loss) per share includes the potential dilutive effect of common stock equivalents as if such securities were converted or exercised during the period, when the effect is dilutive. Common stock equivalents include: (i) outstanding stock options and restricted stock units, which are included under the “treasury stock method” when dilutive; (ii) common stock to be issued upon the exercise of outstanding warrants, which are included under the “treasury stock method” when dilutive, and (iii) preferred stock under the if-converted method. While the impact of these items are generally anti-dilutive during periods of net loss, the Company will determine whether the common stock equivalents should be included in diluted loss per share pursuant to sequencing rules.
The following tables set forth the computation of basic and diluted net income (loss) per share of common stock for the three and six months ended June 30, 2024 and June 30, 2023 (in thousands, except share and per share amounts):
| | | | | | | | | | |
| | Three Months Ended June 30, 2024 | | |
| | | Common stock | | |
| | | | |
| Basic income per share: | | | | |
| Net income | | $ | 98,463 | | | |
| Net income attributed to Series C Preferred Stock | | (94,109) | | | |
| Net income - basic | | $ | 4,354 | | | |
| Weighted average shares | | 1,034,130 | | | |
| Basic net income per share | | $ | 4.21 | | | |
| | | | |
| Diluted (loss) income per share: | | | | |
| Numerator: | | | | |
| Net income - basic | | $ | 4,354 | | | |
| Change in fair value of warrant liability | | (112,046) | | |
| Net loss - diluted | | $ | (107,692) | | | |
| | | | |
| Denominator: | | | | |
| Effect of dilutive securities: | | | | |
| Weighted average shares - basic | | 1,034,130 | | | |
| Common shares issuable for warrants | | 6,619,172 | | | |
| Weighted average shares - diluted | | 7,653,302 | | |
| Diluted net (loss) income per share | | $ | (14.07) | | | |
| | | | | | | | | | |
| | Six Months Ended June 30, 2024 |
| | | Common stock | | |
| | | | |
| Basic income per share: | | | | |
| Net loss | | $ | (22,827) | | | |
| Weighted average shares | | 946,756 | | | |
| Basic net loss per share | | $ | (24.11) | | | |
| | | | |
| Diluted loss per share: | | | | |
| Numerator: | | | | |
| Net loss - basic | | $ | (22,827) | | | |
| Change in fair value of warrant liability | | (112,046) | | |
| Net loss - diluted | | $ | (134,873) | | | |
| | | | |
| Denominator: | | | | |
| Effect of dilutive securities: | | | | |
| Weighted average shares - basic | | 946,756 | | | |
| Potentially dilutive shares | | 3,455,884 | | | |
| Weighted average shares - diluted | | 4,402,640 | | |
| Diluted net loss per share | | $ | (30.63) | | | |
As the Company is in a net loss position as of the six months ended June 30, 2024, the two-class method of computing net loss per share results in no allocation of undistributed losses to participating securities.
| | | | | | | | |
| | Three Months Ended June 30, 2023 |
| | | Common stock |
| | |
| Net loss | | $ | (8,193) | |
| Weighted average shares | | 58,217 | |
| Basic and diluted net loss per share | | $ | (140.73) | |
| | | | | | | | |
| | Six Months Ended June 30, 2023 |
| | | Common stock |
| | |
| Net loss | | $ | (18,148) | |
| Weighted average shares | | 53,557 | |
| Basic and diluted net loss per share | | $ | (338.85) | |
The following outstanding securities have been excluded from the computation of diluted weighted shares outstanding for the three and six months ended June 30, 2024 and 2023, as they could have been anti-dilutive:
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | Three Months Ended | | Six Months Ended |
| | June 30, | | June 30, |
| | | 20243 | | 2023 | | 20243 | | 2023 |
| Stock options | | 156,940 | | 7,706 | | 156,940 | | 7,706 |
Warrants on common stock1 | | 148 | | 17,237 | | 148 | | 17,237 |
Series C Preferred Stock (as-convertible to common stock)2 | | 22,357,897 | | — | | 22,357,897 | | — |
1 The weighted average number of common shares outstanding includes the weighted average outstanding pre-funded warrants for the period because their exercise price was nominal. The weighted average shares outstanding for the three and six months ended June 30, 2023 include the weighted average effect of 2,192 and 1,338 pre-funded warrants, respectively. There were no pre-funded warrants outstanding as of June 30, 2024.
2 Subject to stockholder approval, each share of the Company’s Series C Preferred Stock will automatically convert to 1,000 shares of common stock, subject to certain beneficial ownership limitations.
3 Pursuant to the Almata Transaction, the Company is required to pay potential development milestone payments to the former AlmataBio stockholders in cash, Avalo stock, or a combination thereof at the election of the former AlmataBio stockholders. In the event of share settlement, the number of Avalo shares delivered will vary based on the Company’s stock price. These additional shares are not included in the computation of basic and diluted net income (loss) per share for the three and six months ended June 30, 2024 pursuant to the guidance on contingently issuable shares.
6. Fair Value Measurements
ASC 820, Fair Value Measurements and Disclosures (“ASC 820”) defines fair value as the price that would be received to sell an asset, or paid to transfer a liability, in the principal or most advantageous market in an orderly transaction between market participants on the measurement date. The fair value standard also establishes a three‑level hierarchy, which requires an entity to maximize the use of observable inputs and minimize the use of unobservable inputs when measuring fair value. The valuation hierarchy is based upon the transparency of inputs to the valuation of an asset or liability on the measurement date. The three levels are defined as follows:
•Level 1—inputs to the valuation methodology are quoted prices (unadjusted) for an identical asset or liability in an active market.
•Level 2—inputs to the valuation methodology include quoted prices for a similar asset or liability in an active market or model‑derived valuations in which all significant inputs are observable for substantially the full term of the asset or liability.
•Level 3—inputs to the valuation methodology are unobservable and significant to the fair value measurement of the asset or liability.
The following table presents, for each of the fair value hierarchy levels required under ASC 820, the Company’s assets and liabilities that are measured at fair value on a recurring basis (in thousands):
| | | | | | | | | | | | | | | | | | | | |
| | | June 30, 2024 |
| | | Fair Value Measurements Using |
| | | Quoted prices in active markets for identical assets | | Significant other observable inputs | | Significant unobservable inputs |
| | | (Level 1) | | (Level 2) | | (Level 3) |
| Assets | | | | | | |
| Investments in money market funds* | | $ | 91,322 | | | $ | — | | | $ | — | |
| Liabilities | | | | | | |
| Warrant liability | | $ | — | | | $ | — | | | $ | 82,855 | |
| Derivative liability | | $ | — | | | $ | — | | | $ | 10,710 | |
| | | | | | | | | | | | | | | | | | | | |
| | | December 31, 2023 |
| | | Fair Value Measurements Using |
| | | Quoted prices in active markets for identical assets | | Significant other observable inputs | | Significant unobservable inputs |
| | | (Level 1) | | (Level 2) | | (Level 3) |
| Assets | | | | | | |
| Investments in money market funds* | | $ | 7,077 | | | $ | — | | | $ | — | |
| Liabilities | | | | | | |
| Derivative liability | | $ | — | | | $ | — | | | $ | 5,550 | |
*Investments in money market funds are reflected in cash and cash equivalents on the accompanying unaudited condensed consolidated balance sheets.
As of June 30, 2024, the Company’s financial instruments included cash and cash equivalents, restricted cash, other receivables, prepaid and other current assets, accounts payable, accrued expenses and other current liabilities, derivative liability, and warrant liability. As of December 31, 2023, the Company’s financial instruments included cash and cash equivalents, restricted cash, accounts receivable, other receivables, prepaid and other current assets, accounts payable, accrued expenses and other current liabilities, and derivative liability.
The carrying amounts reported in the accompanying unaudited condensed consolidated financial statements for cash and cash equivalents, restricted cash, accounts receivable, other receivables, prepaid and other current assets, accounts payable, and accrued expenses and other current liabilities approximate their respective fair values because of the short-term nature of these accounts.
Level 3 Valuation
The table presented below is a summary of changes in the fair value of the Company’s Level 3 valuations for the warrant liability and derivative liability for the three and six months ended June 30, 2024 and June 30, 2023:
| | | | | | | | | | | | | | | | | | | | |
| | | Warrant liability | | Derivative liability | | Total |
| Balance at March 31, 2024 | | $ | 194,901 | | | $ | 5,670 | | | $ | 200,571 | |
| Change in fair value | | (112,046) | | | 5,040 | | | (107,006) | |
| Balance at June 30, 2024 | | $ | 82,855 | | | $ | 10,710 | | | $ | 93,565 | |
| | | | | | | | | | | | | | | | | | | | |
| | | Warrant liability | | Derivative liability | | Total |
| Balance at December 31, 2023 | | $ | — | | | $ | 5,550 | | | $ | 5,550 | |
| Initial valuation of warrant liability | | 194,901 | | | — | | | 194,901 | |
| Change in fair value | | (112,046) | | | 5,160 | | | (106,886) | |
| Balance at June 30, 2024 | | $ | 82,855 | | | $ | 10,710 | | | $ | 93,565 | |
| | | | | | | | | | | | | | | | |
| | | | | Derivative liability | | Total |
| Balance at March 31, 2023 | | | | $ | 5,010 | | | $ | 5,010 | |
| | | | | | |
| Change in fair value | | | | 40 | | | 40 | |
| Balance at June 30, 2023 | | | | $ | 5,050 | | | $ | 5,050 | |
| | | | | | | | | | | | | | | | |
| | | | | Derivative liability | | Total |
| Balance at December 31, 2022 | | | | $ | 4,830 | | | $ | 4,830 | |
| | | | | | |
| Change in fair value | | | | 220 | | | 220 | |
| Balance at June 30, 2023 | | | | $ | 5,050 | | | $ | 5,050 | |
Warrant liability
On March 28, 2024, the Company closed a private placement investment with institutional investors in which the investors received (i) 19,946 shares of Series C Preferred Stock and (ii) warrants to purchase up to an aggregate of 11,967,526 shares of Avalo’s common stock (or a number of shares of Series C Preferred Stock convertible into the number of shares of common stock the warrant is then exercisable into). Refer to Note 10 - Capital Structure and sub-header “March 2024 Financing” for more information regarding the warrants.
The Company determined that the warrants do not satisfy the conditions to be accounted for as equity instruments. As the warrants do not meet the equity contract scope exception, the Company classified the warrants as a derivative liability upon issuance.
The Company’s warrant liability was measured at fair value on the issuance date and is measured at fair value each reporting period thereafter until the warrants are either exercised or expire utilizing the Black-Scholes option pricing model, which requires assumptions including the value of the stock on the measurement date, exercise price, expected term, expected volatility, and the risk-free interest rate. Certain assumptions, including the expected term and expected volatility, are subjective and require judgment to develop. As a result, if factors or expected outcomes change and we use significantly different assumptions or estimates, our warrant liability could be materially different. The warrant liability was classified as a Level 3 instrument as its value was based on unobservable market inputs. The inputs utilized in the valuation as of June 30, 2024 include the following:
| | | | | | | | |
| | As of June 30, 2024 |
| Common stock price | | $ | 12.47 | |
| Expected term (in years) | | 0.3 |
| Expected volatility | | 102 | % |
| Risk-free rate | | 5.47 | % |
| Exercise price | | $ | 5.796933 | |
| Dividend yield rate | | — | % |
The common stock price utilized is the closing stock price of Avalo’s common stock on the last trading day of the reporting period. This input is the main driver of the fair value of the warrant liability as of June 30, 2024. The closing stock price on the last day of the second quarter of 2024 was $12.47 per share compared to the closing stock price on the last day of the first quarter of 2024 of $21.75 per share. Subsequent increases or decreases to the stock price at each reporting period drives increases or decreases, will drive corresponding changes, to the fair value of the warrant liability.
As of June 30, 2024, the Company estimated the expected term based on thirty-one days post-expected Dosing Date (as defined below). If the stockholders approve the issuance of shares of common stock underlying the Warrants (the “Required Stockholder Approval”), the Warrants expire on the earlier of (i) the thirty-first day following the public announcement of the first patient dosed in a Phase 2 trial of AVTX-009 in hidradenitis suppurativa (the “Dosing Date”), or (ii) the fifth anniversary of the date of issuance. If the Required Stockholder Approval has not been received by the Dosing Date, then the Warrants expire on the earlier of (i) the thirty-first day following receipt of the Required Stockholder Approval or (ii) the fifth anniversary of the date of issuance. On July 9, 2024, the Company announced that its Investigational New Drug application for AVTX-009 is active, which allows the Company to commence its Phase 2 LOTUS Trial. The Company will seek the Required Stockholder Approval at its annual meeting of stockholders on August 13, 2024, and the expected term assumes that the approval will be obtained in that meeting. Subsequent increases or decreases to the expected term could increase or decrease, respectively, the fair value of the warrant liability.
The other inputs include expected volatility, which is a blend of the Company’s historical volatility and the volatility of comparable peer companies, and the risk-free interest rate, which is based on the implied yield available on U.S. treasury securities with a maturity equivalent to the expected term.
Derivative liability
In the fourth quarter of 2022, Avalo sold its economic rights to future milestone and royalty payments for previously out-licensed assets AVTX-501, AVTX-007, and AVTX-611 to ES Therapeutics, LLC (“ES”), an affiliate of Armistice Capital LLC (“Armistice”), in exchange for $5.0 million (the “ES Transaction”). At the time of the transaction, Armistice was a significant stockholder of the Company and whose chief investment officer, Steven Boyd, and managing director, Keith Maher, served on Avalo’s Board until August 8, 2022. The ES Transaction was approved in accordance with Avalo’s related party transaction policy.
The economic rights sold include (a) rights to a milestone payment of $20.0 million upon the filing and acceptance of an NDA for AVTX-501 pursuant to an agreement with Janssen Pharmaceutics, Inc. (the “AVTX-501 Milestone”) and (b) rights to any future milestone payments and royalties relating to AVTX-007 under a license agreement with Apollo AP43 Limited (“Apollo”), including up to $6.25 million of development milestones, up to $67.5 million in sales-based milestones, and royalty payments over a ten year period of a low single digit percentage of annual net sales (which percentage increases to another low single digit percentage if annual net sales exceed a specified threshold) (the “AVTX-007 Milestones and Royalties”). In addition, Avalo waived all its rights to AVTX-611 sales-based payments of up to $20.0 million that were payable by ES.
The exchange of the economic rights of the AVTX-501 Milestone and AVTX-007 Milestones and Royalties for cash met the definition of a derivative instrument. The fair value of the derivative liability is determined using a combination of a scenario-based method and an option pricing method (implemented using a Monte Carlo simulation). The significant inputs including probabilities of success, expected timing, and forecasted sales as well as market-based inputs for volatility, risk-adjusted discount rates and allowance for counterparty credit risk are unobservable and based on the best information available to Avalo. Certain information used in the valuation is inherently limited in nature and could differ from Janssen and Apollo’s internal estimates.
The fair value of the derivative liability as of the transaction date was approximately $4.8 million, of which $3.5 million was attributable to the AVTX-501 Milestone and $1.3 million was attributable to the AVTX-007 Milestones and Royalties. Subsequent to the transaction date, at each reporting period, the derivative liability is remeasured at fair value. As of June 30, 2024, the fair value of the derivative liability was $10.7 million, of which $6.9 million was attributable to the AVTX-007 Milestones and Royalties and $3.8 million was attributable to the AVTX-501 Milestone. For the six months ended June 30, 2024, the $5.2 million change in fair value was recognized in other income (expense), net in the accompanying unaudited condensed consolidated statements of operations and comprehensive income (loss).
The fair value of the AVTX-501 Milestone was primarily driven by an approximate 23% probability of success to reach the milestone in approximately 3.3 years. The fair value of AVTX-007 Milestones and Royalties was primarily driven by sales forecasts with peak annual net sales reaching $1.8 billion in atopic dermatitis, which is a much larger market opportunity than adult-onset Still’s disease, the previous indication being pursued, an approximate 17% probability of success, as well as time to commercialization of approximately 6.5 years. As discussed above, these unobservable inputs were estimated by Avalo based on limited publicly available information and therefore could differ from Janssen and Apollo’s internal development plans. Any changes to these inputs may result in significant changes to the fair value measurement. Notably, the peak annual net sales forecast (for the AVTX-007 Milestones and Royalties) and probability of successes (for both the AVTX-501 Milestone and the AVTX-007 Milestone and Royalties) are the largest drivers of the fair value and therefore changes to such inputs would likely result in significant changes to such fair value.
In the event that Janssen and/or Apollo are required to make payment(s) to ES Therapeutics pursuant to the underlying agreements, Avalo will recognize revenue under its existing contracts with those customers for that amount when it is no longer probable there would be a significant revenue reversal with any differences between the fair value of the derivative liability related to that payment immediately prior to the revenue recognition and revenue recognized to be recorded as other expense. However, given Avalo is no longer entitled to collect these payments, the potential ultimate settlement of the payments in the future from Janssen and/or Apollo to ES Therapeutics (and the future mark-to-market activity each reporting period) will not impact Avalo’s future cash flows.
No changes in valuation techniques occurred during the six months ended June 30, 2024 and 2023. No transfers of assets between Level 1 and Level 2 of the fair value measurement hierarchy occurred during the six months ended June 30, 2024 and 2023.
7. Leases
Avalo currently occupies two leased properties, both of which serve as administrative office space. The Company determined that both of these leases are operating leases based on the lease classification test performed at lease commencement.
The annual base rent for the Company’s office located in Rockville, Maryland is $0.2 million, subject to annual 2.5% increases over the term of the lease. The applicable lease provided for a rent abatement for a period of 12 months following the Company’s date of occupancy. The lease has an initial term of 10 years from the date the Company made its first annual fixed rent payment, which occurred in January 2020. The Company has the option to extend the lease two times, each for a period of five years, and may terminate the lease as of the sixth anniversary of the first annual fixed rent payment, upon the payment of a termination fee.
The initial annual base rent for the Company’s office located in Chesterbrook, Pennsylvania is $0.2 million and the annual operating expenses are approximately $0.1 million. The annual base rent is subject to periodic increases of approximately 2.4% over the term of the lease. The lease has an initial term of 5.25 years from the lease commencement on December 1, 2021.
The weighted average remaining term of the operating leases at June 30, 2024 was 4.2 years.
Supplemental balance sheet information related to the leased properties include (in thousands):
| | | | | | | | | | | | | | |
| | | As of |
| | | June 30, 2024 | | December 31, 2023 |
| Property and equipment, net | | $ | 1,211 | | | $ | 1,329 | |
| | | | |
| Accrued expenses and other current liabilities | | $ | 548 | | | $ | 537 | |
| Other long-term liabilities | | 1,183 | | | 1,366 | |
| Total operating lease liabilities | | $ | 1,731 | | | $ | 1,903 | |
The operating lease right-of-use (“ROU”) assets are included in property and equipment, net and the lease liabilities are included in accrued expenses and other current liabilities and other long-term liabilities in our unaudited condensed consolidated balance sheets. The Company utilized a weighted average discount rate of 9.0% to determine the present value of the lease payments.
The components of lease expense for the three and six months ended June 30, 2024 and 2023 were as follows (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | Three Months Ended June 30, | | Six Months Ended June 30, |
| | 2024 | | 2023 | | 2024 | | 2023 |
| Operating lease cost* | | $ | 113 | | | $ | 132 | | | $ | 222 | | | $ | 253 | |
*Includes short-term leases, which are immaterial.
The following table shows a maturity analysis of the operating lease liabilities as of June 30, 2024 (in thousands):
| | | | | | | | |
| | | |
| | | Undiscounted Cash Flows |
| July 1, 2024 through December 31, 2024 | | $ | 271 | |
| 2025 | | 553 | |
| 2026 | | 563 | |
| 2027 | | 259 | |
| 2028 | | 201 | |
| 2029 | | 207 | |
| Thereafter | | 17 | |
| Total lease payments | | $ | 2,071 | |
| Less implied interest | | (340) | |
| Total | | $ | 1,731 | |
8. Accrued Expenses and Other Current Liabilities
Accrued expenses and other current liabilities as of June 30, 2024 and December 31, 2023 consisted of the following (in thousands):
| | | | | | | | | | | | | | |
| | | As of |
| | | June 30, 2024 | | December 31, 2023 |
| Research and development | | $ | 732 | | | $ | 352 | |
| Compensation and benefits | | 2,412 | | | 580 | |
| General and administrative | | 578 | | | 830 | |
| Private placement investment transaction costs | | 1,734 | | | — | |
| | | | |
| Commercial operations | | 1,884 | | | 1,873 | |
| | | | |
| Lease liability, current | | 548 | | | 537 | |
| | | | |
| Total accrued expenses and other current liabilities | | $ | 7,888 | | | $ | 4,172 | |
9. Notes Payable
On June 4, 2021, the Company entered into a $35.0 million venture loan and security agreement (the “Loan Agreement”) with Horizon Technology Finance Corporation (“Horizon”) and Powerscourt Investments XXV, LP (“Powerscourt”, and together with Horizon, the “Lenders”). Between June and September of 2021, the Company borrowed the full $35.0 million (the “Note”) available under the Loan Agreement.
In the second quarter of 2022, the Company, as collectively agreed upon with the Lenders, prepaid $15.0 million of principal and accrued interest. In June of 2023, the Company, as collectively agreed upon with the Lenders, prepaid $6.0 million of principal. On September 22, 2023, the Company and the Lenders entered into a Payoff Letter (the “Payoff Letter”), pursuant to which the Company repaid all outstanding principal, inclusive of the final payment fee, and interest under the Loan Agreement in the aggregate amount of $14.3 million. As a result of the payment, all obligations of the parties under the Loan Agreement were deemed satisfied and terminated.
On June 4, 2021, pursuant to the Loan Agreement, the Company issued warrants to the Lenders to purchase 148 shares of the Company’s common stock with an exercise price of $7,488.00 per share (the “Loan Warrants”). The Loan Warrants are exercisable for ten years from the date of issuance. Pursuant to the Payoff Letter, Avalo’s obligations under the Loan Warrants shall survive pursuant to the original terms at issuance. The Loan Warrants, which met equity classification, were recognized as a component of permanent stockholders’ (deficit) equity within additional paid-in-capital and were recorded at the issuance date using a relative fair value allocation method. The Company recognized debt issuance costs and the amount allocated to the Loan Warrants as a debt discount on the date of issuance and amortized these costs to interest expense using the effective interest method over the original term of the loan. As a result of the payoff in the third quarter of 2023, the Company accelerated the remaining $0.9 million amortization of the debt discount, which was recognized as interest expense in the third quarter of 2023.
No Loan Warrants were exercised for the three and six months ended June 30, 2024.
10. Capital Structure
Pursuant to the Company’s amended and restated certificate of incorporation, the Company is authorized to issue two classes of stock, common stock and preferred stock. At June 30, 2024, the total number of shares of capital stock the Company was authorized to issue was 205,000,000, of which 200,000,000 was common stock and 5,000,000 was preferred stock. All shares of common and preferred stock have a par value of $0.001 per share.
Almata Transaction
On March 27, 2024, the Company acquired AlmataBio in which the former AlmataBio stockholders received (i) 171,605 shares of the Company’s common stock and (ii) 2,412 shares of the Company’s Series C Preferred Stock. Subject to the Required Stockholder Approval (the date Company stockholders approve the issuance of common stock for conversion of Series C Preferred Stock and for exercise of warrants), each share of the Series C Preferred Stock issued to former AlmataBio stockholders will automatically convert to 1,000 shares of common stock, subject to certain beneficial ownership limitations. The Series C Preferred Stock holds no voting rights. Refer to Note 3 - Asset Acquisition for more information regarding the acquisition and refer to sub-header “Series C Preferred Stock” within the “March 2024 Financing” section below for more information regarding the Series C Preferred Stock issued pursuant to the Almata Transaction.
March 2024 Financing
On March 28, 2024, the Company closed a private placement investment with institutional investors in which the investors received (i) 19,946 shares of non-voting convertible Series C Preferred Stock, and (ii) warrants to purchase up to an aggregate of 11,967,526 shares of Avalo’s common stock (or a number of shares of Series C Preferred Stock convertible into the number of shares of common stock the warrant is then exercisable into), resulting in upfront gross proceeds of $115.6 million. Net proceeds were $108.1 million after deducting transaction costs. The Company could receive up to an additional $69.4 million of gross proceeds upon the exercise of the warrants.
Warrants on common stock or Series C Preferred Stock issued in March 2024 Financing
The warrants are exercisable via gross physical settlement for $5.796933 per underlying share of common stock (or a number of shares of Series C Preferred Stock convertible into the number of shares of common stock the warrant is then exercisable into). The warrants will become exercisable on (i) March 28, 2024, if exercised for shares of Series C Preferred Stock, or (ii) upon receipt of the Required Stockholder Approval if exercised for shares of common stock. The warrants will expire on the earlier of (y) the fifth anniversary of the date of issuance or (z) the Dosing Date (as defined in Note 6 - Fair Value Measurements), provided that if the Required Stockholder Approval has not been received by the Dosing Date, then the warrants will expire on the earlier of the (A) the fifth anniversary of the date of issuance or (B) thirty-first day following receipt of the Required Stockholder Approval. The warrants include anti-dilution protection provisions.
The Company determined that the warrants do not satisfy the conditions to be accounted for as equity instruments. As the warrants do not meet the equity contract scope exception, the Company classified the warrants as a derivative liability upon issuance. The initial measurement of the warrant at fair value exceeded the proceeds received such that the difference between the initial fair value of the warrants and net upfront cash proceeds is recognized in the income statement as a loss. Subsequently, the warrants are carried at fair value with changes in fair value recognized in the Company’s unaudited condensed consolidated statements of operations and comprehensive income (loss) until either exercised or expired. The valuation of the warrants is considered under Level 3 of the fair value hierarchy due to the need to use assumptions in the valuation that are both significant to the fair value measurement and unobservable. See Note 6 - Fair Value Measurement for a description of the warrant’s valuation methodology.
No warrants were exercised for the six months ended on June 30, 2024.
Upon exercise of the warrants, the Company will pay an additional amount of transaction costs to a third-party financial institution, based on 2.5% gross proceeds received from the exercise. As the warrants are in the money as of June 30, 2024, the Company has recognized $1.7 million for transaction costs within other income (expense), net for the six months ended June 30, 2024. The Company also incurred an additional $7.5 million of transaction costs related to the private placement investment which were expensed within other income (expense), net for the six months ended June 30, 2024.
Series C Preferred Stock issued in the Almata Transaction and March 2024 Financing
As of June 30, 2024, the Company had 5,000,000 shares of Preferred Stock authorized, of which 34,326 have been designated as Series C Preferred Stock. As of June 30, 2024, there were 22,358 shares of Series C Preferred Stock outstanding, with a par value of $0.001 per share. The Series C Preferred Stock has no voting rights, no liquidation preference, and are not redeemable. In the event of any liquidation, dissolution or winding up of the Company, holders of Series C Preferred Stock are entitled to be paid out of the assets with the Company legally available for distribution to its stockholders on an as-converted and pari-passu basis with common stock. The Series C Preferred Stock is subject to broad-based weighted average anti-dilution protection for certain issuances of common stock and securities convertible into common stock. The Series C Preferred Stock is entitled to receive dividends equal to and in the same form, and in the same manner, based on the then-current conversion ratio as dividends actually paid on shares of the common stock, when, as and if such dividends are paid on shares of the common stock. Upon the Required Stockholder Approval, each share of Series C Preferred Stock (i) issued to the former AlmataBio stockholders (as discussed above) and (ii) pursuant to the March 2024 financing will automatically convert to 1,000 shares of common stock, subject to certain beneficial ownership limitations.
The Series C Preferred Stock is contingently redeemable outside the control of the Company such that the Series C Preferred Stock is recognized outside of permanent equity. The carrying value of $11.5 million of the Series C Preferred Stock issued to the former AlmataBio stockholders pursuant to the Almata Transaction is recognized outside of stockholder’s (deficit) equity on the Company’s unaudited condensed consolidated balance sheet. No amounts were allocated to the Series C Preferred Stock issued pursuant to the March 2024 financing because the initial fair value of the warrants exceeded gross proceeds received for the issuance of the private placement bundle that included both Series C Preferred Stock and warrants. The Series C Preferred Stock is not remeasured to redemption value until the shares are probable of becoming redeemable for cash. As of June 30, 2024, the Company expects to have sufficient authorized and unissued shares to settle the Series C Preferred Stock upon Required Stockholder Approval, and therefore it is not probable that the Series C Preferred Stock would be redeemable for cash as of the balance sheet date.
As of June 30, 2024, no Series C Preferred Stock were converted to common stock.
Series D and Series E Preferred Stock issued in the March 2024 Financing
As a condition to the March 2024 financing, a single share of Series D Preferred Stock and a single Series E Preferred Stock were issued to two institutional investors that participated in the private placement. Both the Series D and the Series E Preferred Stock have a par value and liquidation preference of $0.001 per share. The Series D and Series E Preferred Stock do not have voting rights, are not entitled to dividends, and are not convertible into common stock. The holders of the Series D and Series E Preferred Stock have the option to require the Company to redeem their shares at a price equal to the par value at any time. The Company retains the right to redeem the Series D and Series E Preferred Stock at a price equal to the par value if the holder owns less than a certain threshold of the Company’s outstanding common stock. While the Series D and Series E Preferred Stock do not provide the holders with substantive economics, the Series D and Series E Preferred Stock were issued solely to allow for the institutional investors to appoint a director to the Company’s board of directors.
Common Stock Warrants
At June 30, 2024, the following common stock warrants were outstanding:
| | | | | | | | | | | | | | |
| Number of common shares | | Exercise price | | Expiration |
| underlying warrants | | per share | | date |
| 148 | | $ | 7,488 | | | June 2031 |
| 11,967,526 | | $ | 5.80 | | (1) | (1) |
| 11,967,674 | | | | |
(1) The warrants will become exercisable (i) on March 28, 2024, if exercised for shares of Series C Preferred Stock, or (ii) upon receipt of the Required Stockholder Approval (the date Company stockholders approve the issuance of common stock for conversion of Series C Preferred Stock and for exercise of warrants), if exercised for shares of common stock. The warrants will expire on the earlier of (y) the fifth anniversary of the date of issuance or (z) the thirty-first day following the Dosing Date, provided that if the Required Stockholder Approval has not been received by the Dosing Date, then the warrants will expire on the earlier of the (A) the fifth anniversary of the date of issuance or (B) thirty-first day following receipt of the Required Stockholder Approval. The warrants include anti-dilution protection provisions.
11. Stock-Based Compensation
2016 Equity Incentive Plan
In April 2016, our board of directors adopted the 2016 Equity Incentive Plan, which was approved by our stockholders in May 2016 and which was subsequently amended and restated in May 2018 and August 2019 with the approval of our board of directors and our stockholders (the “2016 Third Amended Plan”). In June 2024, our board of directors approved a fourth amended and restated equity incentive plan, which is subject to Company stockholder approval. At the time of the filing of this Quarterly Report on Form 10-Q, the fourth amended and restated equity incentive plan has not been approved by our stockholders, which is being sought at the Company’s annual meeting of stockholders on August 13, 2024. As such, the 2016 Third Amended Plan continues to govern the share reserves disclosed below. During the term of the 2016 Third Amended Plan, the share reserve will automatically increase on the first trading day in January of each calendar year ending on (and including) January 1, 2026, by an amount equal to 4% of the total number of outstanding shares of common stock of the Company on the last trading day in December of the prior calendar year. On January 1, 2024, pursuant to the terms of the 2016 Third Amended Plan, an additional 32,070 shares were made available for issuance. As of June 30, 2024, there were 33,099 shares available for future issuance under the 2016 Third Amended Plan.
Option grants expire after ten years. Employee options typically vest over four years. Employees typically receive a new hire option grant, as well as an annual grant in the first or second quarter of each year. Options granted to directors typically vest either immediately or over a period of one or three years. Directors may elect to receive stock options in lieu of board compensation, which vest immediately. For stock options granted to employees and non-employee directors, the estimated grant date fair market value of the Company’s stock-based awards is amortized ratably over the individuals’ service periods, which is the period in which the awards vest. Stock-based compensation expense includes expense related to stock options and employee stock purchase plan shares. The amount of stock-based compensation expense recognized for the three and six months ended June 30, 2024 and 2023 was as follows (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | Three Months Ended June 30, | | Six Months Ended June 30, |
| | | 2024 | | 2023 | | 2024 | | 2023 |
| Research and development | | $ | 219 | | | $ | 331 | | | $ | 488 | | | $ | 658 | |
| General and administrative | | 252 | | | 562 | | | 612 | | | 1,090 | |
| Total stock-based compensation | | $ | 471 | | | $ | 893 | | | $ | 1,100 | | | $ | 1,748 | |
Stock options with service-based vesting conditions
The Company has granted options that contain service-based vesting conditions. The compensation cost for these options is recognized on a straight-line basis over the vesting periods. A summary of option activity for the six months ended June 30, 2024 is as follows:
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | Options Outstanding |
| | | Number of shares | | Weighted average exercise price per share | | Weighted average grant date fair value per share | | Weighted average remaining contractual term (in years) |
| Balance at December 31, 2023 | | 7,211 | | | $ | 3,191.97 | | | $ | 1,930.00 | | | 8.3 |
| Granted | | 150,000 | | | $ | 13.43 | | | $ | 10.94 | | | |
| | | | | | | | |
| | | | | | | | |
| Forfeited | | (13) | | | $ | 660.00 | | | $ | 472.94 | | | |
| Expired | | (202) | | | $ | 10,162.23 | | | $ | 6,254.04 | | | |
| Balance at June 30, 2024 | | 156,996 | | | $ | 146.25 | | | $ | 91.25 | | | 9.9 |
| | | | | | | | |
| Exercisable at June 30, 2024 | | 5,433 | | | $ | 3,462.88 | | | $ | 2,050.59 | | | 7.7 |
On June 24, 2024, the Company granted its newly appointed Chief Legal Officer options with service-based vesting conditions to purchase 150,000 shares of common stock with an exercise price of $13.43 as an inducement option grant pursuant to Nasdaq Listing Rule 5635(c)(4).
The aggregate intrinsic value of stock options is calculated as the difference between the exercise price of the stock options and the fair value of the Company’s common stock for those stock options that had exercise prices lower than the fair value of the Company’s common stock. As of June 30, 2024, the aggregate intrinsic value of options outstanding was minimal. There were 2,119 options that vested during the six months ended June 30, 2024 with a weighted average exercise price of $1,067.14 per share. The total grant date fair value of shares which vested during the six months ended June 30, 2024 was $1.6 million.
The Company recognized stock-based compensation expense of $0.5 million and $1.1 million related to stock options with service-based vesting conditions for the three and six months ended June 30, 2024. At June 30, 2024, there was $3.4 million of total unrecognized compensation cost related to unvested service-based vesting condition awards. The unrecognized compensation cost is expected to be recognized over a weighted-average period of 2.6 years.
Subsequently, on July 15, 2024, the Company granted its newly appointed Chief Medical Officer options with service-based vesting conditions to purchase 234,000 shares of common stock with an exercise price of $12.65 as an inducement option grant pursuant to Nasdaq Listing Rule 5635(c)(4).
Stock-based compensation assumptions
The following table presents the assumptions used to compute stock-based compensation for stock options with service-based vesting conditions granted under the Black-Scholes valuation model for the six months ended June 30, 2024.
| | | | | | | | |
| Service-based options | | |
| Expected term of option (in years) | | 6.25 |
| Expected stock price volatility | | 99.7% |
| Risk-free interest rate | | 4.26% |
| Expected annual dividend yield | | 0% |
| |
As of June 30, 2024, there were no outstanding stock options that contained market-based vesting conditions.
Employee Stock Purchase Plan
On April 5, 2016, the Company’s board of directors approved the 2016 Employee Stock Purchase Plan (the “ESPP”). The ESPP was approved by the Company’s stockholders and became effective on May 18, 2016 (the “ESPP Effective Date”). In June 2024, our board of directors approved an amended and restated employee stock purchase plan, which is subject to the Company’s stockholder approval. At the time of the filing of this Quarterly Report on Form 10-Q, the amended and restated employee stock purchase plan has not been approved by our stockholders, which is being sought at the Company’s annual meeting of stockholders on August 13, 2024. As such, the 2016 Employee Stock Purchase Plan continues to govern the share reserves disclosed below.
Under the ESPP, eligible employees can purchase common stock through accumulated payroll deductions at such times as are established by the administrator. The ESPP is administered by the compensation committee of the Company’s board of directors. Under the ESPP, eligible employees may purchase stock at 85% of the lower of the fair market value of a share of the Company’s common stock (i) on the first day of an offering period or (ii) on the purchase date. Eligible employees may contribute up to 15% of their earnings during the offering period. The Company’s board of directors may establish a maximum number of shares of the Company’s common stock that may be purchased by any participant, or all participants in the aggregate, during each offering or offering period. Under the ESPP, a participant may not accrue rights to purchase more than $25,000 of the fair market value of the Company’s common stock for each calendar year in which such right is outstanding.
The Company initially reserved and authorized up to 174 shares of common stock for issuance under the ESPP. On January 1 of each calendar year, the aggregate number of shares that may be issued under the ESPP automatically increases by a number equal to the lesser of (i) 1% of the total number of shares of the Company’s capital stock outstanding on December 31 of the preceding calendar year, (ii) 174 shares of the Company’s common stock, or (iii) a number of shares of the Company’s common stock as determined by the Company’s board of directors or compensation committee. On January 1, 2024, the number of shares available for issuance under the ESPP increased by 174. As of June 30, 2024, 958 shares remained available for issuance.
In accordance with the guidance in ASC 718-50, Employee Share Purchase Plans, the ability to purchase shares of the Company’s common stock at the lower of the offering date price or the purchase date price represents an option and, therefore, the ESPP is a compensatory plan under this guidance. Accordingly, stock-based compensation expense is determined based on the option’s grant-date fair value and is recognized over the requisite service period of the option. The Company used the Black-Scholes valuation model and recognized minimal stock-based compensation expense for the three and six months ended June 30, 2024.
12. Income Taxes
The Company recognized minimal income tax expense for the three and six months ended June 30, 2024 and 2023 due to the significant valuation allowance against the Company’s deferred tax assets and the current and prior period losses.
13. Commitments and Contingencies
Litigation
Litigation - General
The Company may become party to various contractual disputes, litigation, and potential claims arising in the ordinary course of business. Reserves are established in connection with such matters when a loss is probable and the amount of such loss can be reasonably estimated. The Company currently does not believe that the resolution of such matters will have a material adverse effect on its financial position or results of operations except as otherwise disclosed in this report.
Possible Future Milestone Payments for In-Licensed Compounds
General
Avalo is a party to license and development agreements with various third parties, which contain future payment obligations such as royalties and milestone payments. The Company recognizes a liability (and related expense) for each milestone if and when such milestone is probable and can be reasonably estimated. As typical in the biotechnology industry, each milestone has unique risks that the Company evaluates when determining the probability of achieving each milestone and the probability of success evolves over time as the programs progress and additional information is obtained. The Company considers numerous factors when evaluating whether a given milestone is probable including (but not limited to) the regulatory pathway, development plan, ability to dedicate sufficient funding to reach a given milestone and the probability of success.
AVTX-009 Agreements
On March 27, 2024, Avalo obtained the rights to an anti-IL-1β mAb (AVTX-009), including the world-wide exclusive license from Eli Lilly and Company (“Lilly”) (the “Lilly License Agreement”), pursuant to its acquisition of AlmataBio. AlmataBio had previously purchased the rights, title and interest in the asset from Leap Therapeutics, Inc. (“Leap”) in 2023, which have since been assumed by Avalo pursuant to its acquisition of AlmataBio (the “Leap Agreement”). Avalo is responsible for the development and commercialization of the program.
Avalo is required to pay up to $70.0 million based on the achievement of specified development and regulatory milestones to Lilly. Upon commercialization, the Company is required to pay sales-based milestones aggregating up to $650.0 million payable to Lilly and $70.0 million payable to Leap. There are no annual or maintenance fees payable under the Lilly License Agreement and Leap Agreement. Additionally, Avalo is required to pay royalties to Lilly during a country-by-country royalty term in which the low end and the high end of the range fall between 5% and 15% of Avalo or its sublicensees’ annual net sales. The royalty term due to Lilly commences on the date of first commercial sale of the licensed product in a given territory and expires on a county-by-country basis; on the latest of (a) the tenth (10th) anniversary of the date of the first commercial sale, (b) the expiration of the last-to-expire licensed patent in the given territory, or (c) the expiration of any data exclusivity period for the licensed product in the given territory.
The Lilly License Agreement remains in effect until the expiration of the last-to-expire royalty term of any licensed products. Each party may terminate for cause or by mutual agreement though the Company may terminate at its sole discretion by giving one-hundred twenty (120) days’ prior written notice to Lilly. There are no termination or expiration provisions under the Leap Agreement.
Avalo has not paid any milestones, royalties or any other amounts under the Lilly License Agreement or Leap Agreement.
No expense related to the agreements was recognized in the six months ended June 30, 2024. There has been no cumulative expense recognized as of June 30, 2024 under the agreements. The Company will continue to monitor the milestones and royalties at each reporting period.
Refer to the sub-header below entitled “Acquisition Related and Other Contingent Liabilities” for information regarding future development milestones that are payable to the former AlmataBio stockholders.
Quisovalimab (AVTX-002) Agreements
KKC License Agreement
On March 25, 2021, the Company entered into a license agreement with Kyowa Kirin Co., Ltd. (“KKC”) for exclusive worldwide rights to develop, manufacture and commercialize quisovalimab, KKC’s first-in-class fully human anti-LIGHT (TNFSF14) monoclonal antibody for all indications (the “KKC License Agreement”). The KKC License Agreement replaced the Amended and Restated Clinical Development and Option Agreement between the Company and KKC dated May 28, 2020. Avalo is responsible for the development and commercialization of quisovalimab in all indications worldwide (other than the option in the KKC License Agreement that, upon exercise by KKC, allows KKC to develop, manufacture and commercialize quisovalimab in Japan).
Under the KKC License Agreement, the Company paid KKC an upfront license fee of $10.0 million, which we recognized within research and development expenses in 2021. Avalo is also required to pay KKC up to an aggregate of $112.5 million based on the achievement of specified development and regulatory milestones. Upon commercialization, the Company is required to make milestone payments to KKC aggregating up to $75.0 million tied to the achievement of annual net sales targets. There are no annual or maintenance fees payable under the KKC License Agreement.
Additionally, the Company is required to pay KKC royalties during a country-by-country royalty term equal to a mid-teen percentage of annual net sales. The Company is required to pay KKC a mid-twenties percentage of the payments that the Company receives from sublicensing of its rights under the KKC License Agreement, subject to certain exclusions. The royalty term due to KKC commences on the date of first commercial sale of the licensed product in a given territory and expires on a county-by-country basis, on the latest of (a) the twelfth (12th) anniversary of the date of the first commercial sale, (b) the expiration of the last-to-expire licensed patent in the given territory, or (c) the expiration of any data exclusivity period for the licensed product in the given territory.
The KKC License Agreement remains in effect while the Company and its affiliates and sublicensees develop and commercialize quisovalimab subject to customary termination rights. Each party may terminate for cause though Avalo may terminate for convenience upon six (6) months’ prior written notice in the case where regulatory approval has not been obtained for the licensed product or upon twelve (12) months’ prior written notice where regulatory approval has been obtained for the licensed product.
As disclosed above, Avalo paid the $10.0 million upfront license fee in 2021. No further amounts have been paid related to the milestones, royalties or any other amounts under the agreement.
No expense related to the KKC License Agreement was recognized in the six months ended June 30, 2024. There has been no cumulative expense recognized as of June 30, 2024 related to the milestones, royalties or any other amounts other than the $10.0 million upfront license fee incurred in 2021 as disclosed above. The Company will continue to monitor the milestones and royalties at each reporting period.
CHOP License Agreement
Following its February 3, 2020 merger with Aevi Genomic Medicine, Inc. (“Aevi”), the Company became party to a license agreement with The Children’s Hospital of Philadelphia (“CHOP”) (as amended, the “CHOP License Agreement”). Quisovalimab became a covered product under this license agreement in 2021 and at that time became subject to the terms therein.
An initial upfront fee of $0.5 million was paid to CHOP by Aevi Genomic Medicine, LLC, which Avalo acquired in 2020. Avalo is required to pay an additional $1.0 million to CHOP based on the achievement of specified regulatory and commercial milestones Avalo is obligated to pay an annual license maintenance fee of $0.2 million to CHOP, of which Avalo has paid an aggregate of $0.9 million to-date.
The Company is also obligated to pay tiered royalties to CHOP on a country-to-country basis in which the low end and high end of the range are single-digit royalties based on the Company’s net sales of quisovalimab. The royalty term extends to the later of (a) fifteen years following the original date of the CHOP License Agreement, (b) the last-to-expire of the valid claims in the licensed patent rights covering the manufacture, sale, or use of quisovalimab and (c) the expiration of the regulatory exclusivity period for quisovalimab.
CHOP may terminate the CHOP License Agreement for the material default or insolvency of the Company, and the Company may terminate the CHOP License Agreement at will with six (6) months’ written notice.
As disclosed above, Aevi paid the $0.5 million upfront license fee and Avalo has paid $0.9 million of annual license fees. No further amounts have been paid related to the milestones, royalties or any other amounts under the agreement.
No expense related to the milestones and royalties due under the CHOP Agreement was recognized for the six months ended June 30, 2024. Avalo has not recognized any cumulative expense under the agreement related to the milestone or royalties as of June 30, 2024. The Company will continue to monitor the milestones and royalties at each reporting period.
AVTX-008 Sanford Burnham Prebys License Agreement
On June 21, 2021, the Company entered into an Exclusive Patent License Agreement with Sanford Burnham Prebys Medical Discovery Institute (the “Sanford Burnham Prebys License Agreement”) under which the Company obtained an exclusive license to a portfolio of issued patents and patent applications covering an immune checkpoint program (AVTX-008). Avalo is responsible for the development and commercialization of the program.
Under the terms of the Sanford Burnham Prebys License Agreement, the Company paid an upfront license fee of $0.4 million, as well as patent costs of $0.5 million, which we recognized within research and development expenses and within general and administrative expenses, respectively, in 2021. Additionally, Avalo pays a $40 thousand annual maintenance fee payable on the first anniversary of the effective date and each anniversary thereafter until the first commercial sale (of which Avalo has paid $0.1 million of annual maintenance fees as of the filing date of this Quarterly Report on Form 10-Q). The Company is required to pay Sanford Burnham Prebys up to approximately $24.2 million based on achievement of specified development and regulatory milestones. Upon commercialization, the Company is required to pay Sanford Burnham Prebys sales-based milestone payments aggregating up to $50.0 million tied to annual net sales targets.
Additionally, the Company is required to pay Sanford Burnham Prebys royalties during a country-by-country royalty term equal to a tiered low-to-mid single digit percentage of annual net sales. Avalo is also required to pay Sanford Burnham Prebys tiered payments in which the low end and high end of the range fall on or between 10-20% of what Avalo receives from sublicensing its rights under the Sanford Burnham Prebys License Agreement, subject to certain exclusions.
The Sanford Burnham Prebys License Agreement remains in effect until the expiration of the royalty term, which with respect to each product and country, continues until the expiration, invalidation or abandonment of the last of the licensed patent rights. Avalo may terminate the Sanford Burnham Prebys License Agreement at any time at its convenience upon providing at least ninety (90) days’ prior written notice. Sanford Burnham Medical Discovery Institute may terminate the Sanford Burnham Prebys License Agreement for cause.
As disclosed above, Avalo paid the $0.4 million upfront fee, as well as total patent costs of $0.5 million and $0.1 million of annual maintenance fees. No further amounts have been paid related to the milestones, royalties or any other amounts under the agreement.
No expense related to milestones or royalties pursuant to the Sanford Burnham Prebys License Agreement was recognized in the six months ended June 30, 2024. There has been no cumulative expense recognized as of June 30, 2024 related to the milestones or royalties under this license agreement other than the $0.4 million upfront fee incurred in 2021. The Company will continue to monitor the milestones and royalties at each reporting period.
AVTX-006 Astellas License Agreement
On July 15, 2019, the Company entered into an exclusive license agreement with OSI Pharmaceuticals, LLC, an indirect wholly owned subsidiary of Astellas Pharma, Inc. (“Astellas”), for the worldwide development and commercialization of the novel, second generation mTORC1/2 inhibitor (AVTX-006). Avalo is fully responsible for the development and commercialization of the program. Avalo considers AVTX-006 a non-core asset and is exploring strategic alternatives.
Under the terms of the license agreement, there was an upfront license fee of $0.5 million. The Company is required to pay Astellas up to an aggregate of $5.5 million based on the achievement of specified development and regulatory milestones. There are no annual maintenance fees payable under the Astellas license agreement. Additionally, the Company is required to pay Astellas a tiered mid-to-high single digit percentage of the payments that Avalo receives from any sublicensing of its rights under the Astellas license agreement, subject to certain exclusions. Upon commercialization, the Company is required to pay Astellas royalties during a country-by-country royalty term equal to a tiered mid-to-high single digit percentage of annual net sales the period beginning upon the date of the first commercial sale of such licensed product in such country and ending on the later to occur of (a) the expiry of the last valid claim of an OSI product patent covering such licensed product in such country, (b) expiration of regulatory exclusivity in such country, and (c) ten (10) years from the first commercial sale of such licensed product in such country.
The Astellas License Agreement remains in effect on a country-by-country and licensed product-by-licensed product basis (in the territory), unless the license agreement is terminated earlier in accordance with the license agreement. Avalo may terminate the agreement at any time upon providing sixty (60) days’ written notice to Astellas and may terminate the agreement in its entirety without cause.
As disclosed above, Avalo paid the $0.5 million upfront license fee. No further amounts have been paid related to the milestones, royalties or any other amounts under the agreement.
No expense related to this license agreement was recognized in the six months ended June 30, 2024. There has been $0.5 million of cumulative expense recognized as of June 30, 2024 related to the milestones under this license agreement. The Company will continue to monitor the remaining milestones and royalties at each reporting period. The Company will continue to monitor the remaining milestones and royalties at each reporting period.
Possible Future Milestone Proceeds for Out-Licensed Compounds
AVTX-301 Out-License
On May 28, 2021, the Company out-licensed its rights in respect of its non-core asset, AVTX-301, to Alto Neuroscience, Inc. (“Alto”). The Company initially in-licensed the compound from an affiliate of Merck & Co., Inc. in 2013. Alto is fully responsible for the development and commercialization of the program.
Under the out-license agreement, the Company received a mid-six digit upfront payment from Alto, which we recognized as license revenue in 2021. The Company is also eligible to receive up to an aggregate of $18.6 million based on the achievement of specified development, regulatory and commercial sales milestones. Additionally, the Company is entitled to a less than single digit percentage royalty based on annual net sales.
The out-license agreement remains in effect on a licensed product-by-licensed product and country-by-country basis until the later of (i) the expiration of the last to expire valid patent claim covering such licensed product in such country, or (ii) 10 (ten) years after the first commercial sale of such licensed product in such country. Upon expiration of the agreement, the licenses shall become a fully paid-up, royalty-free, irrevocable, perpetual non-exclusive license and sublicense.
The Company had not recognized any milestones as of June 30, 2024 or received any payments other than the upfront payment as disclosed above.
AVTX-406 License Assignment
On June 9, 2021, the Company assigned its rights, title, interest, and obligations under an in-license covering its non-core asset, AVTX-406, to ES, a wholly owned subsidiary of Armistice, who was a significant stockholder of the Company at the time of the transaction and whose chief investment officer, Steven Boyd, and managing director, Keith Maher, served on Avalo’s Board until August 8, 2022. The transaction with ES was approved in accordance with Avalo’s related party transaction policy. ES is fully responsible for the development and commercialization of the program.
Under the assignment agreement, the Company received a low-six digit upfront payment from ES, which we recognized as license revenue in 2021. The Company is also eligible to receive up to an aggregate of $6.0 million based on the achievement of specified development and regulatory milestones. Upon commercialization, the Company is eligible to receive sales-based milestone payments aggregating up to $20.0 million tied to annual net sales targets.
The Company had not recognized any milestones as of June 30, 2024 or received any payments other than the upfront payment as disclosed above.
AVTX-800 Series Asset Sale
On October 27, 2023, the Company sold its rights, title and interests in AVTX-801, AVTX-802 and AVTX-803 (collectively, the “800 Series”) to AUG Therapeutics, LLC (“AUG”). AUG is fully responsible for the development and commercialization of the program.
Pursuant to the Purchase Agreement with AUG, the Company received an upfront payment of $0.2 million. Additionally, AUG assumed aggregate liabilities of $0.4 million, which included certain liabilities incurred prior to the date of the Purchase Agreement, costs due and payable between the date of the Purchase Agreement and the closing date, and obligations under 800 Series contracts assumed by AUG. Avalo is also entitled to a contingent milestone payment of 20% of certain amounts, if any, granted to AUG upon sale of any priority review voucher related to the 800 Series compounds granted to AUG by the FDA, net of any selling costs, or $15.0 million for each compound (for a potential aggregate of $45.0 million) if the first FDA approval is for any indication other than a Rare Pediatric Disease (as defined in the Purchase Agreement).
The Company had not recognized any revenue related to the milestones as of June 30, 2024 or received any payments other than the upfront payment and reimbursement for certain liabilities as disclosed above.
Acquisition Related and Other Contingent Liabilities
Almata Transaction Possible Future Milestone Payments
On March 27, 2024, the Company acquired AVTX-009 through its acquisition of AlmataBio. Pursuant to the Almata Transaction, the Company made a cash payment of $7.5 million in April 2024 to the former AlmataBio stockholders, which was due upon the initial closing of the private placement investment on March 28, 2024 (the “Initial Milestone”). Further, a portion of the consideration for the AlmataBio transaction includes development milestones to the former AlmataBio stockholders including $5.0 million due upon the first patient dosed in a Phase 2 trial in patients with hidradenitis suppurativa for AVTX-009 (the “Second Milestone”) and $15.0 million due upon the first patient dosed in a Phase 3 trial for AVTX-009 (the “Third Milestone”), both of which are payable in cash or stock of Avalo (or combination thereof) at the election of the former AlmataBio stockholders. In the absence of timely notice of such election, Avalo may elect to pay the milestones in cash or common stock of Avalo or a combination thereof.
The Company paid the Initial Milestone payment in April 2024 and recognized the payment within acquired in-process research and development expense in the condensed consolidated statements of operations and comprehensive income (loss) for the six months ended June 30, 2024. In addition, the Company concluded the Second Milestone was probable as of the acquisition date and therefore recognized the $5.0 million milestone within acquired in-process research and development expense in the condensed consolidated statements of operations and comprehensive income (loss) for the six months ended June 30, 2024 and the corresponding liability as contingent consideration as of June 30, 2024. The Company will continue to monitor the Third Milestone each reporting period.
Aevi Merger Possible Future Milestone Payments
In the first quarter of 2020, the Company consummated its merger with Aevi, in which Avalo acquired the rights to quisovalimab, AVTX-006 and AVTX-007 (the “Merger” or the “Aevi Merger”). A portion of the consideration for the Aevi Merger included two future contingent development milestones worth up to an additional $6.5 million, payable in either shares of Avalo’s common stock or cash, at the election of Avalo.
The first milestone was the enrollment of a patient in a Phase 2 study related to quisovalimab (for treatment of pediatric onset Crohn’s disease), AVTX-006 (for treatment of any indication) or AVTX-007 (for treatment of any indication) prior to February 3, 2022, which would have resulted in a milestone payment of $2.0 million. The Company did not meet the first milestone prior to February 3, 2022. Therefore, no contingent consideration related to this milestone was recognized as of June 30, 2024 and no future contingent consideration will be recognized.
The second milestone is the receipt of an NDA approval for either AVTX-006 or AVTX-007 from the FDA on or prior to February 3, 2025. If this milestone is met, the Company is required to make a milestone payment of $4.5 million. The contingent consideration related to the second development milestone will be recognized if and when such milestone is probable and can be reasonably estimated. No contingent consideration related to the second development milestone had been recognized as of June 30, 2024. The Company will continue to monitor the second milestone each reporting period.
AVTX-006 Royalty Agreement with Certain Related Parties
In July 2019, Aevi entered into a royalty agreement, and liabilities thereunder were assumed by Avalo upon close of the Aevi Merger in February 2020. The royalty agreement provided certain investors, including LeoGroup Private Investment Access, LLC on behalf of Garry Neil, the Company’s Chief Executive Officer and Chairman of the Board, and Mike Cola, the Company’s former Chief Executive Officer (collectively, the “Investors”), a royalty stream, in exchange for a one-time aggregate payment of $2.0 million (the “Royalty Agreement”). Pursuant to the Royalty Agreement, the Investors will be entitled collectively to an aggregate amount equal to a low-single digit percentage of the aggregate net sales of the Company’s second generation mTORC1/2 inhibitor, AVTX-006 for a royalty term consistent with the royalty term disclosed in the AVTX-006 Astellas License Agreement section above. Avalo considers AVTX-006 a non-core asset and is exploring strategic alternatives. At any time beginning three years after the date of the first public launch of AVTX-006, Avalo may exercise, at its sole discretion, a buyout option that terminates any further obligations under the Royalty Agreement in exchange for a payment to the Investors of an aggregate of 75% of the net present value of the royalty payments. A majority of the independent members of the board of directors and the audit committee of Aevi approved the Royalty Agreement.
Avalo assumed this Royalty Agreement upon closing of the Aevi Merger and it is recorded as a royalty obligation within the Company's accompanying unaudited condensed consolidated balance sheet as of June 30, 2024 and December 31, 2023. Because there is a significant related party relationship between the Company and the Investors, the Company has treated its obligation to make royalty payments under the Royalty Agreement as an implicit obligation to repay the funds advanced by the Investors. As the Company makes royalty payments in accordance with the Royalty Agreement, it will reduce the liability balance. At the time that such royalty payments become probable and estimable, and if such amounts exceed the liability balance, the Company will impute interest accordingly on a prospective basis based on such estimates, which will result in a corresponding increase in the liability balance.
Karbinal Royalty Make-Whole Provision
In 2018, in connection with the acquisition of certain commercialized products, the Company entered into a supply and distribution agreement (the “Karbinal Agreement”) with TRIS Pharma Inc. (“TRIS”). As part of the Karbinal Agreement, the Company had an annual minimum sales commitment, which is based on a commercial year that spans from August 1 through July 31, of 70,000 units through 2025. The Company was required to pay TRIS a royalty make whole payment (“Make-Whole Payments”) of $30 for each unit under the 70,000 units annual minimum sales commitment through 2025.
As a part of the Aytu Transaction, the Company assigned all its payment obligations, including the Make-Whole Payments, under the Karbinal Agreement (collectively, the “TRIS Obligations”) to Aytu. However, under the original license agreement, the Company could ultimately be liable for the TRIS Obligations to the extent Aytu fails to make the required payments. The future Make-Whole Payments to be made by Aytu are unknown as the amount owed to TRIS is dependent on the number of units sold.
Item 2. Management’s Discussion and Analysis of Financial Condition and Results of Operations
This Quarterly Report on Form 10-Q and the information incorporated herein by reference contain forward-looking statements that involve a number of risks and uncertainties, as well as assumptions that, if they never materialize or prove incorrect, could cause our results to differ materially from those expressed or implied by such forward-looking statements. Forward-looking statements can be identified by the use of forward-looking words such as “projects,” “may,” “might,” “will,” “could,” “would,” “should,” “continue,” “seeks,” “aims,” “predicts,” “believes,” “expects,” “anticipates,” “estimates,” “intends,” “plans,” “potential,” “pro forma” or other similar words (including their use in the negative), or by discussions of future matters such as: the future financial and operational outlook; the development of product candidates; and other statements that are not historical. Although our forward-looking statements reflect the good faith judgment of our management, these statements can only be based on facts and factors currently known by us. Consequently, forward-looking statements are inherently subject to risks and uncertainties, and actual results and outcomes may differ materially from results and outcomes discussed in the forward-looking statements. Factors that could cause or contribute to these differences include those set out elsewhere in this Quarterly Report on Form 10-Q, particularly in Part II – Item 1A, “Risk Factors,” as well as in our Annual Report on Form 10-K filed with the Securities and Exchange Commission, or SEC, on March 29, 2024, and in our other filings with the SEC. Statements made herein are as of the date of the filing of this Quarterly Report on Form 10-Q with the SEC and should not be relied upon as of any subsequent date. Unless otherwise required by applicable law, we do not undertake, and we specifically disclaim any obligation to update any forward-looking statements to reflect occurrences, developments, unanticipated events or circumstances after the date of such statement.
The following discussion and analysis of our financial condition and results of operations should be read in conjunction with our unaudited financial statements and related notes that appear in Item 1 of this Quarterly Report on Form 10-Q and with our audited financial statements and related notes for the year ended December 31, 2023 appearing in our Annual Report on Form 10-K filed with the SEC on March 29, 2024.
Overview
Avalo Therapeutics, Inc. (the “Company,” “Avalo” or “we”) is a clinical stage biotechnology company focused on the treatment of immune dysregulation. Avalo’s lead asset is AVTX-009, an anti-IL-1β monoclonal antibody (“mAb”), targeting inflammatory diseases. Avalo has two additional drug candidates which include quisovalimab (anti-LIGHT mAb) and AVTX-008 (BTLA agonist fusion protein).
Management’s primary evaluation of the success of the Company is the ability to progress its pipeline forward toward commercialization or opportunistically out-licensing rights to indications or geographies. We believe the ability to achieve the anticipated milestone as presented in the following chart represents our most immediate evaluation point as to the progress of our goal to move the pipeline forward.
Recent Developments
In July 2024, Avalo announced that its Investigational New Drug (“IND”) for AVTX-009, an anti-IL-1β (mAb), for the treatment of hidradenitis suppurativa (“HS”) is now active, permitting the Company to commence its Phase 2 (“LOTUS”) clinical trial. Avalo expects to enroll its first patient in its Phase 2 LOTUS Trial, in the second half of 2024. The LOTUS Trial is a randomized, double-blind, placebo-controlled, parallel-group Phase 2 trial with two AVTX-009 dose regimens to evaluate the efficacy and safety of AVTX-009 in approximately 180 adults with moderate to severe HS. Avalo is the study sponsor and the current proposed trial locations include United States, Canada, France, Germany, Italy, Spain, Bulgaria, Czech Republic, Greece, Poland, Australia and Turkey. Subjects will be randomized (1:1:1) to receive either one of two doses of AVTX-009 or placebo. The primary efficacy endpoint is the proportion of subjects achieving Hidradenitis Suppurativa Clinical Response (HiSCR75) at Week 16.
On June 24, 2024, Avalo announced the appointment of Paul Varki as its Chief Legal Officer. Additionally, on July 16, 2024, Avalo announced the appointment of Mittie Doyle, MD, FACR as its Chief Medical Officer.
Liquidity
Since inception, we have incurred significant operating and cash losses from operations. We have primarily funded our operations to date through sales of equity securities, out-licensing transactions and sales of assets.
For the six months ended June 30, 2024, Avalo generated a net loss of $22.8 million and negative cash flows from operations of $22.5 million. As of June 30, 2024, Avalo had $93.4 million in cash and cash equivalents. In the first quarter of 2024, the Company closed a private placement investment consisting of an initial upfront gross investment of $115.6 million (net proceeds were $108.1 million after deducting transaction costs) and potentially up to an additional $69.4 million of gross proceeds upon the exercise of warrants issued in the financing.
Based on our current operating plans, we expect that our existing cash and cash equivalents are sufficient to fund operations for at least twelve months from the filing date of this Quarterly Report on Form 10-Q and we expect current cash on hand to fund operations into 2027. The Company closely monitors its cash and cash equivalents and seeks to balance the level of cash and cash equivalents with our projected needs to allow us to withstand periods of uncertainty relative to the availability of funding on favorable terms. We may satisfy any future cash needs through sales of equity securities under the Company’s at-the-market program or other equity financings, out-licensing transactions, strategic alliances/collaborations, sale of programs, and/or mergers and acquisitions. There can be no assurance that any financing or business development initiatives can be realized by the Company, or if realized, what the terms may be. To the extent that we raise capital through the sale of equity, the ownership interest of our existing stockholders will be diluted, and the terms may include liquidation or other preferences that adversely affect the rights of our stockholders. Further, if the Company raises additional funds through collaborations, strategic alliances or licensing arrangements with third parties, the Company might have to relinquish valuable rights to its technologies, future revenue streams, research programs or product candidates.
Our Strategy
Our strategy for increasing stockholder value includes:
•Advancing our pipeline of compounds through development and to regulatory approval;
•Developing the go-to-market strategy to quickly and effectively market, launch, and distribute each of our compounds that receive regulatory approval;
•Opportunistically out-licensing rights to indications or geographies; and
•Acquiring or licensing rights to targeted, complementary differentiated preclinical and clinical stage compounds.
There is no guarantee that our products will obtain regulatory approval by the United States Food and Drug Administration (the “FDA”) or comparable foreign regulatory authorities. The FDA approval process is complex, time-consuming, and expensive. It typically involves the following prior to submitting a new drug application (NDA) or biologics license application (BLA): preclinical laboratory and animal testing, submission of an IND application, and human clinical trials to establish safety and efficacy. Human clinical trials typically include: Phase 1 studies to evaluate the safety and tolerability of the drug, generally in normal, healthy volunteers; Phase 2 studies to evaluate safety and efficacy, as well as appropriate doses; these studies are typically conducted in patient volunteers who suffer from the particular disease condition that the drug is designed to treat; and Phase 3 studies to evaluate safety and efficacy of the product at specific doses in one or more larger pivotal trials. Upon submission of an NDA or BLA, the FDA reviews the application including potentially an FDA advisory committee review and inspects manufacturing facilities prior to FDA approval or rejection of the application. Even if a product receives FDA approval, the agency may impose post-approval requirements or withdraw approval if safety or efficacy issues arise. The processes for obtaining marketing approvals in foreign countries, along with subsequent compliance with applicable statutes and regulations, require the expenditure of substantial time and financial resources.
Results of Operations
Comparison of the Three Months Ended June 30, 2024 and 2023
Product Revenue, Net
There was no net product revenue for the three months ended June 30, 2024, compared to $0.6 million for the three months ended June 30, 2023. The decrease was driven by the planned expiration of our license and supply agreement for our only commercially marketed product, Millipred®, on September 30, 2023.
We do not expect gross product revenue for Millipred®, which the Company considered a non-core asset. However, the Company will continue to monitor estimates for commercial liabilities, such as sales returns. As additional information becomes available, the Company could recognize expense (or benefit) for differences between actuals or updated estimates to the reserves previously recognized.
Cost of Product Sales
Cost of product sales was $0.3 million for the three months ended June 30, 2024, compared to $0.7 million for the same period in 2023. The overall decrease was due to the expiration of Avalo’s license and supply agreement for Millipred® on September 30, 2023.
The amount recognized in the current period related to changes in the receivable reserve with Aytu BioScience, Inc. (“Aytu”), based upon the timing of reconciliations to the retention amount and the sales returns paid by Aytu on behalf of Avalo as stipulated in a transition services agreement.
The Company will continue to monitor estimates for commercial liabilities, such as sales returns, profit share with the supplier pursuant to the reconciliation process, and commercial activity with Aytu, who previously managed Millipred® commercial operations on our behalf for an interim period. As additional information becomes available, the Company could recognize expense (or a benefit) for differences between actuals or updated estimates to the reserves previously recognized, which could be recognized in cost of product sales.
Research and Development Expenses
The following table summarizes our research and development expenses for the three months ended June 30, 2024 and 2023 (in thousands):
| | | | | | | | | | | | | | |
| | | Three Months Ended June 30, |
| | | 2024 | | 2023 |
| Nonclinical expenses | | $ | 195 | | | $ | 211 | |
| Clinical expenses | | 1,579 | | | 1,697 | |
| CMC expenses | | 740 | | | 1,685 | |
| | | | |
| Internal expenses: | | | | |
| Salaries, benefits and related costs | | 1,810 | | | 679 | |
| Stock-based compensation expense | | 219 | | | 331 | |
| Other | | 58 | | | 55 | |
| | | $ | 4,601 | | | $ | 4,658 | |
Research and development expenses decreased $0.1 million for the three months ended June 30, 2024 compared to the three months ended June 30, 2023. This decrease was mainly driven by a $0.9 million decrease in chemistry, manufacturing, and controls (“CMC”) expenses and a $0.1 million decrease in clinical expenses, partially offset by a $1.1 million increase in salaries, benefits and related costs.
CMC and clinical expenses decreased due to the quisovalimab PEAK trial concluding in June 2023 and the corresponding timing of raw material orders that did not repeat in the current period. These decreases were partially offset by expenses incurred in the current period related to the development of AVTX-009 in HS, including trial enabling activities such as preparation of the IND (which was active in early July 2024) and preparation to enroll the first patient in the LOTUS Trial, which we expect to occur in the second half of 2024. Avalo acquired the rights to develop AVTX-009 in March 2024.
Salaries, benefits and related costs increased compared to the three months ended June 30, 2023 due to increased non-equity incentive plan compensation expense incurred in the current period.
We expect future research and development expenses to increase in 2024 as a result of our development plans for AVTX-009, including planned commencement of the Phase 2 LOTUS Trial in the second half of 2024.
Acquired in-process research and development
In the first quarter of 2024, we acquired AVTX-009, through a merger with AlmataBio, Inc. (“AlmataBio”) and its wholly owned subsidiary (the “Almata Transaction”), resulting in us acquiring $27.6 million of in-process research and development (“IPR&D”), of which $0.1 million was recognized in the three months ended June 30, 2024. There was no acquired IPR&D for the three months ended June 30, 2023.
General and Administrative Expenses
The following table summarizes our general and administrative expenses for the three months ended June 30, 2024 and 2023 (in thousands):
| | | | | | | | | | | | | | |
| | | Three Months Ended June 30, |
| | | 2024 | | 2023 |
| | | | |
| Salaries, benefits and related costs | | $ | 1,376 | | | $ | 431 | |
| Legal, consulting and other professional expenses | | 2,421 | | | 1,071 | |
| Stock-based compensation expense | | 252 | | | 562 | |
| Advertising and marketing expense | | 21 | | | 7 | |
| Other | | 458 | | | 356 | |
| | | $ | 4,528 | | | $ | 2,427 | |
General and administrative expenses increased $2.1 million for the three months ended June 30, 2024 compared to the prior period. The increase was primarily driven by a $1.4 million increase in legal, consulting and other professional expenses for activities incurred related to the legal, accounting, and reporting requirements following the Almata Transaction and private placement investment, which closed in late March 2024. Additionally, salaries, benefits and related costs increased $0.9 million due to increased non-equity incentive plan compensation expense incurred in the current period.
While we expect the majority of operating expense increases will be focused on research and development activities to progress AVTX-009, we also expect moderate increases to general and administrative expenses to support the AVTX-009 program.
Other Income (Expense), Net
The following table summarizes our other income (expense), net for the three months ended June 30, 2024 and 2023 (in thousands):
| | | | | | | | | | | | | | |
| | | Three Months Ended June 30, |
| | | 2024 | | 2023 |
| | | | |
| | | | |
| | | | |
| Change in fair value of warrant liability | | 112,046 | | | — | |
| Change in fair value of derivative liability | | (5,040) | | | (40) | |
| Interest income (expense), net | | 1,039 | | | (996) | |
| | | | |
| | $ | 108,045 | | | $ | (1,036) | |
Other income, net increased $109.1 million for the three months ended June 30, 2024 compared to prior period. The increase was primarily driven by a $112.0 million gain recognized on the change of fair value of the warrant liability associated with the warrants issued in the March 2024 financing. The warrant liability fair value was $82.9 million as of June 30, 2024, as compared to $194.9 million as of March 31, 2024. The decrease in the fair value was mainly driven by the decrease in the closing stock price on the last trading day of the second quarter of 2024 of $12.47 per share compared to the closing stock price on the last trading day of the first quarter of 2024 of $21.75 per share. As of June 30, 2024, the Company estimated the expected term based on thirty-one days post-expected Dosing Date (as defined below), which also assumes that the Required Stockholder Approval (as defined below) will be obtained at its upcoming annual meeting of stockholders on August 13, 2024.
As the warrants are carried at fair value, future changes in fair value will be recognized in other income (expense), net at each reporting period until the warrants are either exercised or expired. Notably, subsequent increase or decreases to the stock price at each reporting period or increases or decreases to the expected term, are expected to drive increases or decreases, respectively, to the fair value of the warrant liability. If the stockholders approve the issuance of shares of common stock underlying the warrants (the “Required Stockholder Approval”), the warrants expire on the earlier of (i) the thirty-first day following the public announcement of the first patient dosed in a Phase 2 trial of AVTX-009 in HS (the “Dosing Date”), or (ii) the fifth anniversary of the date of issuance. If the Required Stockholder Approval has not been received by the Dosing Date, then the warrants expire on the earlier of (i) the thirty-first day following receipt of the Required Stockholder Approval or (ii) the fifth anniversary of the date of issuance. Refer to Note 6 – Fair Value Measurements of the unaudited condensed consolidated financial statements for more information.
The increase to other income, net was partially offset by a $5.0 million increase in fair value of the derivative liability (representing a loss on the change in fair value), driven by changes in assumptions utilized in the valuation of the AVTX-007 Milestone and Royalties (as defined in Note 6 to the unaudited condensed consolidated finance statements) due to updated publicly available information of the indication and status of trial activities pursued for the asset by Apollo AP43 Limited (“Apollo”). The main driver of this increase was a significant increase to the peak annual net sales forecast as a result of Apollo developing the asset in atopic dermatitis, which is a much larger market opportunity than adult-onset Still’s disease, the previous indication being pursued. Refer to Note 6 - Fair Value Measurements of the unaudited condensed consolidated financial statements for more information.
Finally, interest income increased by $2.0 million given the Company’s increased cash position compared to the prior period, paired with no interest expense incurred in the current period given the Company’s full payoff of its former loan in the prior year.
Income Tax Expense
The Company recognized minimal income tax expense for both the three months ended June 30, 2024 and 2023.
Comparison of the Six Months Ended June 30, 2024 and 2023
Product Revenue, Net
There was no net product revenue for the six months ended June 30, 2024, compared to $1.1 million for the six months ended June 30, 2023. The decrease was driven by the planned expiration of our license and supply agreement for our only commercially marketed product, Millipred®, on September 30, 2023.
We do not expect future gross product revenue for Millipred®, which the Company considered a non-core asset. However, the Company will continue to monitor estimates for commercial liabilities, such as sales returns. As additional information becomes available, the Company could recognize expense (or benefit) for differences between actuals or updated estimates to the reserves previously recognized.
Cost of Product Sales
Cost of product sales were $0.3 million for the six months ended June 30, 2024, compared to $1.3 million for the same period in 2023. The overall decrease in cost of product sales during the period was related to the expiration of Avalo’s license and supply agreement for Millipred® on September 30, 2023.
The amount recognized in the current period related to changes in the receivable reserve with Aytu, based upon the timing of reconciliations to the retention amount and the sales returns paid by Aytu on behalf of Avalo pursuant to a transition services agreement.
The Company will continue to monitor estimates for commercial liabilities, such as sales returns, profit share with the supplier pursuant to the reconciliation process, and commercial activity with Aytu, who previously managed Millipred® commercial operations on our behalf for an interim period. As additional information becomes available, the Company could recognize expense (or a benefit) for differences between actuals or updated estimates to the reserves previously recognized, which could be recognized in cost of product sales.
Research and Development Expenses
The following table summarizes our research and development expenses for the six months ended June 30, 2024 and 2023 (in thousands):
| | | | | | | | | | | | | | |
| | | Six Months Ended June 30, |
| | | 2024 | | 2023 |
| Nonclinical expenses | | $ | 347 | | | $ | 575 | |
| Clinical expenses | | 1,641 | | | 4,472 | |
| CMC expenses | | 993 | | | 2,978 | |
| | | | |
| Internal expenses: | | | | |
| Salaries, benefits and related costs | | 3,134 | | | 1,873 | |
| Stock-based compensation expense | | 488 | | | 658 | |
| Other | | 113 | | | 111 | |
| | | $ | 6,716 | | | $ | 10,667 | |
Research and development expenses decreased $4.0 million for the six months ended June 30, 2024. This decrease was mainly driven by a $2.8 million decrease in clinical expenses and a $2.0 million decrease in CMC expenses. Clinical and CMC expenses decreased due to the quisovalimab PEAK trial concluding in June 2023 and the corresponding timing of raw material orders. These decreases were partially offset by increased expenses incurred in the second quarter of 2024 related to the development of AVTX-009 in HS including trial enabling activities such as preparation of the IND (which was active in early July 2024) and preparation to enroll the first patient in the LOTUS Trial, which we expect to occur in the second half of 2024. Avalo acquired the rights to develop AVTX-009 in March 2024.
These decreases were partially offset by a $1.3 million increase in salaries, benefits and related costs due to non-equity incentive plan compensation expense incurred in the current period.
We expect future research and development expenses to increase in 2024 as a result of our development plans for AVTX-009, including planned commencement of the Phase 2 LOTUS Trial in the second half of 2024.
Acquired in-process research and development
In the first quarter of 2024, we acquired AVTX-009 pursuant to the Almata Transaction, resulting in us acquiring $27.6 million of acquired IPR&D. The fair value of the IPR&D, substantially all of which is related to AVTX-009, was recognized as acquired IPR&D expense for the six months ended June 30, 2024 as there is no alternative future use. There was no acquired IPR&D for the six months ended June 30, 2023.
General and Administrative Expenses
The following table summarizes our general and administrative expenses for the six months ended June 30, 2024 and 2023 (in thousands):
| | | | | | | | | | | | | | |
| | | Six Months Ended June 30, |
| | | 2024 | | 2023 |
| | | | |
| Salaries, benefits and related costs | | $ | 2,286 | | | $ | 1,186 | |
| Legal, consulting and other professional expenses | | 3,996 | | | 2,253 | |
| Stock-based compensation expense | | 612 | | | 1,090 | |
| Advertising and marketing expense | | 28 | | | 20 | |
| Other | | 799 | | | 585 | |
| | | $ | 7,721 | | | $ | 5,134 | |
General and administrative expenses increased $2.6 million for the six months ended June 30, 2024 compared to the prior period. The increase was driven by a $1.7 million increase in legal, consulting and other professional expenses incurred related to the Almata Transaction and corresponding private placement investment. Additionally, salaries, benefits and related costs increased $1.1 million due primarily to non-equity incentive plan compensation expense incurred in the current period.
While we expect the majority of operating expense increases will be focused on research and development activities to progress AVTX-009, we also expect moderate increases to general and administrative expenses to support the AVTX-009 program.
Other Income (Expense), Net
The following table summarizes our other income (expense), net for the six months ended June 30, 2024 and 2023 (in thousands):
| | | | | | | | | | | | | | |
| | | Six Months Ended June 30, |
| | | 2024 | | 2023 |
| | | | |
| Excess of initial warrant fair value over private placement proceeds | | (79,276) | | | — | |
| Change in fair value of warrant liability | | 112,046 | | | — | |
| Private placement transaction costs | | (9,220) | | | — | |
| Change in fair value of derivative liability | | (5,160) | | | (220) | |
| Interest income (expense), net | | 1,138 | | | (1,945) | |
| Other expense, net | | — | | | (25) | |
| | $ | 19,528 | | | $ | (2,190) | |
Other income, net increased $21.7 million for the six months ended June 30, 2024 as compared to the prior period primarily driven by the impact of the warrant liability associated with the warrants issued in the March 2024 financing. For the six months ended June 30, 2024, we recognized a $79.3 million loss at issuance on the excess of initial warrant liability fair value ($194.9 million) over the private placement proceeds ($115.6 million). The loss was more than offset by a $112.0 million gain recognized on the change of fair value of the warrant liability from March 31, 2024 to June 30, 2024. The fair value of the warrant liability was estimated using a Black-Scholes option-pricing model and they key variable input driving the decrease in fair value as of June 30, 2024 compared to March 31, 2024 was the stock price movement ($12.47 as of the last trading day of the second quarter and $21.75 as of the last trading day of the first quarter). As of June 30, 2024, the Company estimated the expected term based on thirty-one days post-expected Dosing Date, which also assumes that the Required Stockholder Approval will be obtained at its upcoming annual meeting of stockholders on August 13, 2024.
As the warrants are carried at fair value, future changes in fair value will be recognized in other income (expense), net at each reporting period until the warrants are either exercised or expired. Notably, subsequent increase or decreases to the stock price at each reporting period or increases or decreases to the expected term, are expected to drive increases or decreases, respectively, to the fair value of the warrant liability. If the Required Stockholder Approval is obtained, the warrants expire on the earlier of (i) the thirty-first day following the Dosing Date, or (ii) the fifth anniversary of the date of issuance. If the Required Stockholder Approval has not been received by the Dosing Date, then the warrants expire on the earlier of (i) the thirty-first day following receipt of the Required Stockholder Approval or (ii) the fifth anniversary of the date of issuance. Refer to Note 6 – Fair Value Measurements of the unaudited condensed consolidated financial statements for more information.
The increase to other income, net was partially offset by the recognition of $9.2 million of private placement transaction costs, largely consisting of the placement agent fee of $7.0 million due on the transaction close date, and $1.7 million fee payable upon exercise of the warrants issued in the private placement investment.
Additionally, other income, net was partially offset by a $5.2 million increase in fair value of the derivative liability (representing a loss on the change in fair value) driven by changes in assumptions utilized in the valuation of the AVTX-007 Milestones and Royalties (as defined in Note 6 of the unaudited condensed consolidated financial statements) due to updated publicly available information of the indication and status of trial activities pursued for the asset by Apollo. The main driver of this increase was a significant increase to the peak annual net sales forecast as a result of Apollo developing the asset in atopic dermatitis, which is a much larger market opportunity than adult-onset Still’s disease, the previous indication being pursued. Refer to Note 6 - Fair Value Measurements of the unaudited condensed consolidated financial statements for more information.
Finally, interest income increased by $3.1 million given the Company’s increased cash position compared to the prior period, paired with no interest expense incurred in the current period given the Company’s full payoff of its former loan in the prior year.
Income Tax Expense
The Company recognized minimal income tax expense for both the six months ended June 30, 2024 and 2023.
Liquidity and Capital Resources
Uses of Liquidity
The Company uses cash to primarily fund the ongoing development of its research and development pipeline assets, mainly AVTX-009, and costs associated with its organizational infrastructure.
Cash Flows
The following table summarizes our cash flows for the three months ended June 30, 2024 and 2023 (in thousands):
| | | | | | | | | | | | | | |
| | | Six Months Ended June 30, |
| | | 2024 | | 2023 |
| | | | |
| Net cash (used in) provided by: | | | | |
| Operating activities | | $ | (22,485) | | | $ | (21,078) | |
| Investing activities | | 356 | | | (133) | |
| Financing activities | | 108,140 | | | 14,346 | |
| Net increase (decrease) in cash and cash equivalents | | $ | 86,011 | | | $ | (6,865) | |
Net cash used in operating activities
Net cash used in operating activities was $22.5 million for the six months ended June 30, 2024 and consisted primarily of a net loss of $22.8 million and adjustments to reconcile net loss to net cash used in operating activities including the change in fair value of the warrant liability of $112.0 million, excess of initial warrant fair value over private placement investment proceeds of $79.3 million, acquired IPR&D of $27.6 million, $7.5 million milestone payment made to the former AlmataBio stockholders upon the closing of a private placement investment, change in fair value of the derivative liability of $5.2 million, transaction costs payable upon exercise of the warrants issued pursuant to the private placement investment of $1.7 million, and stock-based compensation of $1.1 million. Prepaid expense increased $1.6 million primarily due to advances paid for AVTX-009 contracts and the timing of insurance prepayments.
Net cash used in operating activities was $21.1 million for the six months ended June 30, 2023, and consisted primarily of a net loss of $18.1 million and non-cash adjustments to reconcile cash used in operating activities including stock-based compensation expense of $1.7 million. Additionally, changes in net liabilities decreased $5.8 million driven by decreases in accrued expenses and other liabilities and accounts payable of $5.6 million and $2.1 million, respectively, partially offset by a $1.9 million decrease in other receivables.
We expect future cash used in operating activities to increase in 2024 as a result of acquiring AVTX-009 in March 2024 and our associated development plans.
Net cash provided by (used in) investing activities
Net cash provided by investing activities for the six months ended June 30, 2024 consisted of the cash acquired as part of the Almata Transaction. Net cash used in investing activities was minimal for the six months ended June 30, 2023.
Net cash provided by financing activities
Net cash provided by financing activities for the six months ended June 30, 2024 consisted of gross proceeds of $115.6 million from the private placement investment that closed on March 28, 2024, partially offset by transaction costs paid related to the private placement investment of $7.5 million.
Net cash provided by financing activities for the six months ended June 30, 2023 consisted of net proceeds of $13.7 million from an underwritten public offering that closed in February 2023 and net proceeds of $6.5 million from the sale of common stock pursuant to the Company’s at-the-market program in the second quarter of 2023, partially offset by the $6.0 million principal prepayment under the Loan Agreement (as defined in Note 9 to the unaudited condensed consolidated financial statements).
The Company could receive up to an additional $69.4 million of gross proceeds upon the exercise of the warrants that were issued pursuant to the private placement investment that closed on March 28, 2024. The warrants are exercisable for approximately $5.80 per underlying share of common stock (or a number of shares of Series C Preferred Stock convertible into the number of shares of common stock the warrant is then exercisable into). If the Required Stockholder Approval is obtained, the warrants expire on the earlier of (i) the thirty-first day following the Dosing Date, or (ii) the fifth anniversary of the date of issuance. If the Required Stockholder Approval has not been received by the Dosing Date, then the warrants expire on the earlier of (i) the thirty-first day following receipt of the Required Stockholder Approval or (ii) the fifth anniversary of the date of issuance.
Critical Accounting Policies, Estimates, and Assumptions
This Management’s Discussion and Analysis of Financial Condition and Results of Operations is based on our unaudited condensed consolidated financial statements included in this Quarterly Report on Form 10-Q, which have been prepared in accordance with GAAP. In preparing the financial statements in conformity with GAAP, the Company makes estimates and assumptions that have an impact on assets, liabilities, revenue and expenses reported. These estimates can also affect supplemental information disclosed by us, including information about contingencies, risk, and financial condition. In our unaudited condensed consolidated financial statements, estimates are used for, but not limited to, revenue recognition, cost of product sales, stock-based compensation, fair value measurements, the valuation of warrant liabilities, the valuation of derivative liabilities, cash flows used in management’s going concern assessment, income taxes, goodwill, and clinical trial accruals. The Company believes, given current facts and circumstances, that our estimates and assumptions are reasonable, adhere to GAAP and are consistently applied. Inherent in the nature of an estimate or assumption is the fact that actual results may differ from estimates, and estimates may vary as new facts and circumstances arise. Our most critical accounting estimates and assumptions are included in our Annual Report on Form 10-K for the year ended December 31, 2023 filed with the SEC on March 29, 2024 and Quarterly Report on Form 10-Q for the quarter ended March 31, 2024 filed with the SEC on May 13, 2024 (as amended on July 11, 2024). There have been no significant changes to our critical accounting policies during the three months ended June 30, 2024.
Off-Balance Sheet Arrangements
We do not have any off-balance sheet arrangements, as defined by applicable SEC rules and regulations.
Item 3. Quantitative and Qualitative Disclosures About Market Risk.
As a smaller reporting company, we are not required to provide the information required by this Item.
Item 4. Controls and Procedures.
Evaluation of Disclosure Controls and Procedures
As required by Rule 13a-15(b) and Rule 15d-15(b) of the Securities Exchange Act of 1934, as amended, or the Exchange Act, our management, including our principal executive officer and our principal financial officer, conducted an evaluation as of the end of the period covered by this Quarterly Report on Form 10-Q of the effectiveness of the design and operation of our disclosure controls and procedures. In designing and evaluating our disclosure controls and procedures, our management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired control objectives. Based on that evaluation, our principal executive officer and principal financial officer concluded that our disclosure controls and procedures were effective at the reasonable assurance level as of the end of the period covered by this Quarterly Report on Form 10-Q.
Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by us in the reports we file or submit under the Exchange Act is accumulated and communicated to our management, including our principal executive officer and principal financial officer, as appropriate to allow timely decisions regarding required disclosure.
Changes in Internal Control Over Financial Reporting
There were no changes in our internal control over financial reporting identified in management’s evaluation pursuant to Rules 13a-15(d) or 15d-15(d) of the Exchange Act during the period covered by this Quarterly Report on Form 10-Q that materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
PART II – OTHER INFORMATION
Item 1. Legal Proceedings.
The information set forth in Note 13 - Commitments and Contingencies, under the heading “Litigation” to our Unaudited Condensed Consolidated Financial Statements, included in Part I, Item 1 of this Quarterly Report on Form 10-Q, is incorporated herein by reference.
Item 1A. Risk Factors.
In addition to the other information set forth in this Quarterly Report on Form 10-Q, you should carefully consider the factors discussed in Part I, “Item 1A. Risk Factors” in our Annual Report on Form 10-K for the year ended December 31, 2023, filed with the SEC on March 29, 2024 (the “2023 10-K”), which could materially affect our business, financial condition, or future results. Our risk factors as of the date of this Quarterly Report on Form 10-Q have not changed materially from those described in the Form 10-K referenced above except as follows. These risks and those described in the Form 10-K referenced above, however, are not the only risks facing our Company. Additional risks and uncertainties not currently known to us or that we currently deem to be immaterial may also materially adversely affect our business, financial condition, or future results of operations and the trading price of our common stock.
If we are unable to obtain or maintain intellectual property rights, or if the scope of patent protection is not sufficiently broad, competitors could develop and commercialize products similar or identical to ours, and we might not be able to compete effectively in our market. Furthermore, certain of our composition of matter patents for AVTX-009 and AVTX-002 currently expire in 2026, 2027, or 2028.
Our success depends in significant part on our and our licensors’, licensees’ or collaborators’ ability to establish, maintain and protect patents and other intellectual property rights and operate without infringing the intellectual property rights of others. We have filed numerous patent applications both in the United States and in foreign jurisdictions to obtain patent rights to inventions we have discovered. We have also licensed from third parties’ rights to patent portfolios.
The patent prosecution process is expensive and time‑consuming, and we and our current or future licensors, licensees or collaborators might not be able to prepare, file and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner. It is also possible that we or our licensors, licensees or collaborators will fail to identify patentable aspects of inventions made in the course of development and commercialization activities before it is too late to obtain patent protection on them. Moreover, in some circumstances, we might not have the right to control the preparation, filing and prosecution of patent applications, or to maintain the patents, covering technology that we license from or license to third parties and are reliant on our licensors, licensees or collaborators. Therefore, these patents and applications might not be prosecuted and enforced in a manner consistent with the best interests of our business. If our current or future licensors, licensees or collaborators fail to establish, maintain or protect such patents and other intellectual property rights, such rights may be reduced or eliminated. If our licensors, licensees or collaborators are not fully cooperative or disagree with us as to the prosecution, maintenance or enforcement of any patent rights, such patent rights could be compromised.
The patent position of biotechnology and pharmaceutical companies generally is highly uncertain, involves complex legal and factual questions and has in recent years been the subject of much litigation. As a result, the issuance, scope, validity, enforceability and commercial value of our and our current or future licensors’, licensees’ or collaborators’ patent rights are highly uncertain. Our and our licensors’, licensees’ or collaborators’ pending and future patent applications might not result in patents being issued which protect our technology or products, in whole or in part, or which effectively prevent others from commercializing competitive technologies and products. The patent examination process may require us or our licensors, licensees or collaborators to narrow the scope of the claims of our or our licensors’, licensees’ or collaborators’ pending and future patent applications, which may limit the scope of patent protection that may be obtained. Our and our licensors’, licensees’ or collaborators’ patent applications cannot be enforced against third parties practicing the technology claimed in such applications unless and until a patent issues from such applications, and then only to the extent the issued claims cover the technology.
Furthermore, given the amount of time required for the development, testing and regulatory review of new product candidates, patents protecting such candidates might expire before or shortly after such candidates are commercialized. As a result, our owned and licensed patent portfolio might not provide us with sufficient rights to exclude others from commercializing products similar or identical to our products. We expect to seek extensions of patent terms where these are available in any countries where we are prosecuting patents. This includes in the United States under the Drug Price Competition and Patent Term Restoration Act of 1984, which permits a patent term extension of up to five years beyond the expiration of the patent. However, the applicable authorities, including the FDA in the United States, and any equivalent regulatory authority in other countries, might not agree with our assessment of whether such extensions are available, and may refuse to grant extensions to our patents, or may grant more limited extensions than we request.
Certain of our composition of matter patents for AVTX-009 and AVTX-002 currently expire in 2026, 2027, or 2028. If we are unable to obtain extensions to our patents or other means of regulatory exclusivity for our products, the expiration of patents might create opportunities for competitors to enter the market for our target indications, which could have a material negative impact on our financial results. Without patent protection, we are susceptible to competitors bringing similar products to market, obtaining FDA approval, and achieving regulatory exclusivity prior to us.
Both AVTX-009 and AVTX-002 are biologic products, which would allow the Company to receive biologics reference product exclusivity in both the United States (twelve years) and Europe (ten years) if and upon receiving marketing approval for the products. Once our composition of matter patents expire, we plan to rely on such exclusivity to protect our intellectual property, which has its associated risks. See the risk factor below titled “As appropriate, we intend to seek all available periods of regulatory exclusivity for our product candidates. However, there is no guarantee that we will be granted these periods of regulatory exclusivity or that we will be able to maintain these periods of exclusivity” for more information regarding the risks of relying on regulatory exclusivity.
As appropriate, we intend to seek all available periods of regulatory exclusivity for our product candidates. However, there is no guarantee that we will be granted these periods of regulatory exclusivity or that we will be able to maintain these periods of exclusivity.
The Biologics Price Competition and Innovation Act of 2009 (“BPCIA”) created an abbreviated pathway for the approval of biosimilar and interchangeable biological products. The regulatory pathway establishes legal authority for the FDA to review and approve biological products that are biosimilar to or interchangeable with an FDA-licensed reference biologic.
Under the BPCIA, a reference biological product is granted twelve years of exclusivity in the United States from the time of first licensure of the product (ten years of data and marketing exclusivity in Europe), and the FDA will not accept an application for a biosimilar or interchangeable product based on the reference biological product until four years after the date of first licensure of the reference product. In addition, the licensure of a biosimilar product may not be made effective by the FDA until twelve years from the date on which the reference product was first licensed. During this 12-year period of exclusivity, another company may still develop and receive approval for a competing version of the reference product if the FDA approves a full biologics license application for the competing product containing the sponsor’s own preclinical data and data from adequate and well-controlled clinical trials to demonstrate the safety, purity and potency of their product.
We believe that our current and any future product candidates we develop as biologic products should qualify for the twelve-year period of exclusivity in the United States (ten years in Europe). While we intend to apply for all periods of exclusivity that we may be eligible for, there is no guarantee that we will receive all such periods of exclusivity. Additionally, under certain circumstances, the FDA may revoke the period of exclusivity. As a result, there is no guarantee that we will be able to maintain a period of exclusivity, even if granted. Further, there is a risk that any exclusivity we receive is shortened due to Congressional action or otherwise, or that the FDA will not consider subject product candidates to be reference products for competing products, potentially creating the opportunity for biosimilar competition sooner than anticipated.
Pursuant to the terms of the securities purchase agreement for the March 2024 financing, we are required to recommend that our stockholders approve the conversion of all outstanding shares of our Series C non-voting convertible preferred stock into shares of our common stock and the ability to issue shares of common stock upon the exercise of the Warrants. We cannot guarantee that our stockholders will approve this matter, and if they fail to do so, it might have an adverse impact on our operations and the value and liquidity of your investment might be impaired.
Under the terms of the securities purchase agreement for the March 2024 financing, we agreed to use our best efforts to obtain the Required Stockholder Approval to allow (i) the conversion of all outstanding shares of Series C Preferred Stock issued in the AlmataBio Transaction and the March 2024 private placement and (ii) the ability of all Warrants issued in the March 2024 financing to be exercised for shares of common stock, as required by the Nasdaq listing rules, at a meeting of our stockholders and, if such approval is not obtained at that meeting, to seek to obtain such approval at a stockholders meeting to be held at least every 90 days thereafter until such approval is obtained. If the Required Stockholder Approval is not obtained at a meeting, the need to hold one or more additional meetings would be time consuming and costly. In addition, the failure to receive the Required Stockholder Approval could impair the public perception of our Company and our securities and could have an adverse impact on the liquidity and value of the Series C Preferred Stock and the Warrants, as well as on the value of our common stock.
The Series C non-voting convertible preferred stock and the Warrants are not convertible or exercisable for shares of our common stock without the approval of our stockholders and therefore may not have any value.
The Series C non-voting convertible preferred stock is convertible into shares of our common stock and the Warrants are exercisable for shares of our common stock only upon receipt of the Required Stockholder Approval. If we do not obtain the Required Stockholder Approval, then the Series C Preferred Stock will remain outstanding in accordance with its terms, the Warrants will remain outstanding and exercisable for shares of Series C Preferred Stock, and the AlmataBio Transaction milestone payments would only be payable in cash. If the Series C Preferred Stock cannot convert to shares of common stock, there may be less reason for the holders of the Warrants to exercise them as to our knowledge there currently is no market for the Series C Preferred Stock nor do we expect a robust market to develop. This would reduce the ability of the Company to benefit from the receipt of cash proceeds from the exercise of the Warrants.
A substantial number of shares of our common stock may be sold upon the conversion of our Series C Preferred Stock, which could cause the price of our common stock to decline.
If we receive the Required Stockholder Approval, the up to 34,325,423 shares of common stock issuable upon conversion of the Series C Preferred Stock and upon the exercise of the Warrants, subject to beneficial ownership limitations, will represent approximately 97% of the shares of common stock outstanding on August 5, 2024 on an as converted-basis. The sale of a substantial number of shares of our securities in the public market, or the perception that such sales may occur, could adversely affect the price of our common stock on the Nasdaq Capital Market. We cannot predict the effect, if any, that market sales of those shares of common stock or the availability of those shares of common stock for sale will have on the market price of our common stock.
In addition, in the future, we may also issue shares of our common stock in connection with investments or acquisitions. The amount of shares of our common stock issued in connection with an investment or acquisition could substantially increase our shares of common stock outstanding, which could adversely affect the price of our common stock on the Nasdaq Capital Market.
If we are not able to comply with the applicable continued listing requirements or standards of The Nasdaq Stock Market, Nasdaq could delist our common stock.
Our common stock is currently listed on The Nasdaq Stock Market. In order to maintain that listing, we must satisfy minimum financial and other continued listing requirements and standards, including those regarding director independence and independent committee requirements, minimum stockholders’ equity, a minimum closing bid price of $1.00 per share, and certain corporate governance requirements. There can be no assurances that we will be able to comply with the applicable listing standards. For example, on August 8, 2023, Nasdaq notified us that we failed the $1.00 minimum bid price requirement and the $35 million minimum Market Value of Listed Securities (“MVLS”) requirement. The Company affected a 1-for-240 reverse stock split on December 28, 2023, which has allowed its common stock to trade above $1.00 since December 29, 2023. On January 30, 2024, the Company received written notification from Nasdaq confirming that the Company had regained compliance with the Bid Price Rule. Nasdaq also notified the Company that it is subject to a mandatory panel monitor for a period of one year from January 30, 2024. If, within the one-year monitoring period, Nasdaq finds the Company again out of compliance with the Bid Price Rule, then notwithstanding Nasdaq Rule 5810(c)(2), the Company will not be permitted to provide Nasdaq with a plan of compliance with respect to that deficiency and Nasdaq will not be permitted to grant additional time for the Company to regain compliance with respect to that deficiency, nor will the Company be afforded an applicable cure or compliance period pursuant to Nasdaq Rule 5810(c)(3). Instead, Nasdaq will issue a Delist Determination Letter and the Company will have an opportunity to request a new hearing with the initial Nasdaq panel assigned to the Company for its recent noncompliance or newly convened hearings panel if the initial panel is unavailable. The Company will have the opportunity to respond to the hearings panel as provided by Nasdaq Rule 5815(d)(4)(C). If the Company fails to satisfy the Nasdaq panel, its securities would be delisted from Nasdaq. There can be no assurance that we will continue to maintain such requirement or remain in compliance with any other Nasdaq listing requirements.
Further, on May 20, 2024, we received a written notice from Nasdaq indicating that the Company no longer complies with the requirement under Nasdaq Listing Rule 5550(b)(1) to maintain a minimum of $2,500,000 in stockholders equity for continued listing on the Nasdaq Capital Market (the “Stockholders’ Equity Requirement) because the Company reported stockholders’ equity of negative $112.6 million in its Form 10-Q for the period ended March 31, 2024, and, as of the date of the written notice, the Company did not meet the alternatives of market value of listed securities or net income from continuing operations (together with the Stockholders’ Equity Requirement, the “Listing Rule”). In accordance with the Nasdaq Listing Rules, the Company submitted a plan to regain compliance (the “Plan of Compliance”) on July 1, 2024. On July 29, 2024, Nasdaq accepted the Company’s Plan of Compliance and granted the Company a 180-day extension (until November 18, 2024) to regain compliance with the Listing Rule. If the Company is unable to regain compliance within the extension period granted by the Staff, the Staff would be required to issue a delisting determination. There can be no assurance that the Company will evidence compliance with the Listing Rule by November 18, 2024.
In the event that our common stock is delisted from The Nasdaq Stock Market and is not eligible for quotation or listing on another market or exchange, trading of our common stock could be conducted only in the over‑the‑counter market or on an electronic bulletin board established for unlisted securities such as the Pink Sheets or the OTC Bulletin Board. In such event, it could become more difficult to dispose of, or obtain accurate price quotations for, our common stock, and there would likely also be a reduction in our coverage by securities analysts and the news media, which could cause the price of our common stock to decline further. Also, it may be difficult for us to raise additional capital if we are not listed on an exchange.
A delisting would also likely have a negative effect on the price of our common stock and would impair your ability to sell or purchase our common stock when you wish to do so. In the event of a delisting, we may take actions to restore our compliance with The Nasdaq Stock Market’s listing requirements, but we can provide no assurance that any such action taken by us would allow our common stock to become listed again, stabilize the market price or improve the liquidity of our common stock, prevent our common stock from dropping below The Nasdaq Stock Market minimum bid price requirement or prevent non‑compliance with The Nasdaq Stock Market’s listing.
Item 6. Exhibits.
| | | | | | | | |
Exhibit
Number | | Description of Exhibit |
| | |
| 10.1 | | |
| | |
| 10.2 | | |
| | |
| 10.3 | | |
| | |
| 31.1+ | | |
| | |
| 31.2+ | | |
| | |
| 32.1† | | |
| | |
| 101 | | Interactive data files pursuant to Rule 405 of Regulation S-T: (i) Condensed Consolidated Balance Sheets as of June 30, 2024 (Unaudited) and December 31, 2023; (ii) Condensed Consolidated Statements of Operations and Comprehensive Income (Loss) (Unaudited) for the Three and Six Months Ended June 30, 2024 and 2023; (iii) Condensed Consolidated Statements of Cash Flows (Unaudited) for the Six Months Ended June 30, 2024 and 2023; (iv) Condensed Consolidated Statements of Preferred Stock and Changes in Stockholders’ Deficit (Unaudited) for the Three and Six Months Ended June 30, 2024 and 2023; and (v) Notes to Unaudited Financial Statements. |
| | |
| 104 | | Cover Page Interactive Data File, formatted in XBRL (included in Exhibit 101). |
| | |
+ Filed herewith.
† This certification is being furnished solely to accompany this Quarterly Report on Form 10-Q pursuant to 18 U.S.C. Section 1350, and is not being filed for purposes of Section 18 of the Securities Exchange Act of 1934, as amended, and is not to be incorporated by reference into any filing of the registrant, whether made before or after the date hereof, regardless of any general incorporation language in such filing.
| | | | | | | | | | | |
| SIGNATURES |
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized. |
| | | |
| | | Avalo Therapeutics, Inc. |
| | | |
| Date: August 12, 2024 | | /s/ Christopher Sullivan |
| | | Christopher Sullivan |
| | | Chief Financial Officer |
| | | (on behalf of the registrant and as the registrant’s principal financial officer) |
| | |