Delaware (State or other jurisdiction of incorporation or organization) | | | 8071 (Primary Standard Industrial Classification Code Number) | | | 27-1701898 (I.R.S. Employer Identification Number) |
Thomas Brida General Counsel and Secretary Invitae Corporation 1400 16th Street San Francisco, California 94103 (415) 374-7782 | | | Mike Hird Gabriella A. Lombardi Pillsbury Winthrop Shaw Pittman LLP 2550 Hanover Street Palo Alto, CA 94304-1115 (650) 233-4500 | | | Jason Myers President and Chief Executive Officer ArcherDX, Inc. 2477 55th Street, Suite 202 Boulder, Colorado 80301 (877) 771-1093 | | | Brent D. Fassett Laura Medina Cooley LLP 380 Interlocken Crescent, Suite 900 Broomfield, Colorado 80021 (720) 566-4000 |
Large accelerated filer ☒ | | | Accelerated filer ☐ | | | Non-accelerated filer ☐ | | | Smaller reporting company ☐ |
| | | | | | | Emerging growth company ☐ |
| | | Page | |
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | |
Q: | What is the merger? |
A: | Invitae, ArcherDX, Merger Sub A and Merger Sub B have entered into an Agreement and Plan of Merger and Plan of Reorganization, dated as of June 21, 2020, which (as the same may be amended from time to time) is referred to as the merger agreement. A copy of the merger agreement is attached as Annex A to this proxy statement/prospectus. The merger agreement contains the terms and conditions of the proposed acquisition of ArcherDX by Invitae. Under the merger agreement, subject to satisfaction or (to the extent permitted by law) waiver of the conditions set forth in the merger agreement and described hereinafter, Merger Sub A will merge with and into ArcherDX, with ArcherDX surviving the merger and becoming a wholly owned subsidiary of Invitae, referred to as the first merger. Promptly following the first merger, ArcherDX will merge with and into Merger Sub B, with Merger Sub B surviving as a wholly owned subsidiary of Invitae, referred to as the second merger. Such first merger and second merger are referred to collectively as the merger or the merger transactions. The merger is intended to be considered together as a single integrated transaction for U.S. federal income tax purposes and to qualify as a “reorganization” within the meaning of Section 368(a) of the Code. |
Q: | Why am I receiving these materials? |
A: | Invitae is sending these materials to its stockholders to help them decide how to vote their shares of common stock with respect to the merger and other matters to be considered at the special meeting. The special meeting will be held virtually. You are invited to attend the special meeting via live audio webcast to vote electronically on the proposals described in this proxy statement/prospectus. |
Q: | What will ArcherDX stockholders receive in the merger? |
A: | If the merger is completed, the aggregate consideration for the merger will consist of $325.0 million in cash and 30.0 million shares of Invitae common stock, plus up to an additional 27.0 million shares of Invitae common stock payable in connection with the achievement of certain milestones, which consideration is collectively referred to as the merger consideration. The actual merger consideration to be paid at the closing of the merger is subject to closing-related adjustments, based on ArcherDX’s cash, debt, net working capital, transaction expenses and other considerations at the closing of the merger. Subject to the terms and conditions of the merger agreement, including adjustment to the cash consideration payable, if the merger is completed, each share of ArcherDX capital stock (other than (1) shares held by ArcherDX as treasury stock and (2) shares held by a holder who has properly demanded and not effectively withdrawn or lost such holder’s appraisal, dissenters’ or similar rights under the General Corporation Law of the State of Delaware, or DGCL) will be converted into (a) approximately $9.05 in cash, without interest, plus (b) shares of Invitae |
Q: | How will Invitae pay the cash component of the merger consideration? |
A: | Invitae’s obligation to complete the merger is not conditioned upon obtaining financing. Invitae anticipates that approximately $325.0 million will be required to pay the aggregate cash portion of the merger consideration to ArcherDX stockholders. The merger will be financed in part by the use of Invitae’s cash on hand, in part by the private placement to accredited investors of up to an aggregate of approximately 16.3 million shares of Invitae common stock at a price of $16.85 per share, for gross proceeds to Invitae of approximately $275.0 million, and in part by the net proceeds of up to approximately $200.0 million from a new senior secured term loan facility. |
Q: | What equity stake will ArcherDX stockholders hold in Invitae immediately following the merger? |
A: | Upon the completion of the merger, the estimated number of shares of Invitae common stock issuable as a portion of the merger consideration will be 30.0 million shares and 57.0 million shares if the milestones are achieved in full, which will result in former ArcherDX stockholders holding up to approximately 12% and 23%, respectively, of the outstanding fully diluted Invitae common stock, based on the number of outstanding shares of common stock and outstanding stock-based awards of Invitae and ArcherDX as of August 19, 2020 and also assuming a closing date of October 2, 2020. |
Q: | When do Invitae and ArcherDX expect to complete the transaction? |
A: | Invitae and ArcherDX are working to complete the transaction as soon as practicable and expect that the transaction will be completed on October 2, 2020. Neither Invitae nor ArcherDX can predict, however, the actual date on which the transaction will be completed because it is subject to conditions beyond each company’s control. |
Q: | What are the conditions to completion of the merger? |
A: | In addition to the approval of the merger proposal by Invitae stockholders as described above, completion of the merger is subject to the satisfaction of a number of other conditions, including (1) the expiration or termination of the applicable waiting period under the Hart-Scott-Rodino Antitrust Improvements Act of 1976, as amended, which is referred to as the HSR Act and (2) no governmental authority of competent jurisdiction having issued or entered any order or enacted any law after June 21, 2020 having the effect of enjoining or otherwise prohibiting the completion of the merger. Termination of the applicable waiting period under the HSR Act occurred on July 15, 2020. In addition, holders of at least 75% of the outstanding shares of ArcherDX capital stock are required to approve the merger, the merger agreement and the transactions contemplated therein. ArcherDX has received votes which exceed the requisite stockholder vote required under Delaware law to approve the merger, and intends to solicit the additional votes necessary to satisfy the closing condition following the effectiveness of the registration statement of which this proxy statement/prospectus forms a part. |
Q: | What am I being asked to vote on, and why is this approval necessary? |
A: | Invitae stockholders are being asked to vote on the following proposals: |
1. | Adoption of the Merger Agreement. To vote on a proposal to adopt the merger agreement and approve the merger contemplated thereby, which is further described in “The Merger” and “The Merger Agreement,” beginning on pages 55 and 81, respectively, and a copy of which is attached as Annex A to the proxy statement/prospectus accompanying this notice, which is referred to as the merger proposal; |
2. | Issuance of Shares of Invitae Common Stock in Connection with a Private Placement to Accredited Investors and Issuance of Warrants to Purchase Invitae Common Stock in Connection with a Term Loan Facility. To vote on a proposal to approve, for the purposes of complying with the applicable provisions of NYSE Listing Rule 312.03 requiring stockholder approval of any issuance of securities that will have voting power of 20% or more of Invitae’s currently outstanding voting power, the issuance of up to an aggregate of approximately 16.3 million shares of Invitae common stock at a price of $16.85 per share, for gross proceeds to Invitae of approximately $275.0 million, in a private placement to accredited investors, referred to as the private placement, and the issuance of warrants to purchase 1.0 million shares of Invitae common stock at an exercise price of $16.85 per share pursuant to a new senior secured term loan facility, referred to as the term loan facility, which proceeds of the private placement and the term loan facility will be used for working capital and general corporate purposes, including to fund the cash consideration for the merger pursuant to the merger agreement, which is referred to as the securities issuance proposal; and |
3. | Adjournment or Postponement of the Special Meeting. To vote on a proposal to approve the adjournment of the special meeting to a later date or dates, if necessary or appropriate, to solicit additional proxies in the event there are not sufficient votes at the time of the special meeting to approve the merger proposal, which is referred to as the adjournment proposal. |
Q: | What vote is required to approve each proposal at the Special Meeting? |
A: | The merger proposal: The affirmative vote of holders of a majority of the shares of Invitae common stock represented (in person or by proxy) at the special meeting, assuming a quorum is present, is required to approve the merger proposal, which is referred to as the stockholder approval. |
Q: | How many votes do I have? |
A: | Each Invitae stockholder is entitled to one vote for each share of Invitae common stock held of record as of the record date. |
Q: | What constitutes a quorum? |
A: | The presence at the special meeting, in person or by proxy, of the holders of a majority of the shares of Invitae common stock issued and outstanding and entitled to vote on the record date for the special meeting will constitute a quorum for the transaction of business at the special meeting. Abstentions (which are described below) will count for the purpose of determining the presence of a quorum for the transaction of business at the special meeting. |
Q: | How does the Invitae Board recommend that I vote? |
A: | The Invitae Board unanimously recommends that stockholders vote: “FOR” the merger proposal, “FOR” the securities issuance proposal, and “FOR” the adjournment proposal. For information regarding the Invitae Board’s reasons for approving and recommending adoption of the merger agreement and the transactions contemplated by the merger agreement, including the merger, see “The Merger—Invitae Board of Directors’ Recommendation and Reasons for the Merger” beginning on page 63. |
Q: | What happens if the securities issuance proposal is not approved? |
A: | Approval of the securities issuance proposal is not a condition to the completion of the merger. Invitae is proposing the securities issuance proposal in order to comply with NYSE Listing Rule 312.03, which requires stockholder approval of certain transactions that result in the issuance of 20% or more of the outstanding voting power before the issuance of stock or securities. Invitae expects to issue approximately 16.3 million shares of Invitae common stock in a private placement to accredited investors and a warrant to purchase 1.0 million shares of Invitae common stock in connection with a new senior secured term loan facility in order to raise funds to pay the cash component of the merger consideration. If the securities issuance proposal is not approved, Invitae will need to use additional cash on hand to finance the cash component of the merger consideration, which may have an adverse effect on its business and financial condition. |
Q: | What do I need to do now? |
A: | After carefully reading and considering the information contained in this proxy statement/prospectus, please vote your shares as soon as possible so that your shares will be represented at the special meeting. Please follow the instructions set forth on the proxy card or on the voting instruction form provided by the record holder if your shares are held in the name of your broker, bank or other nominee. |
Q: | Does my vote matter? |
A: | Yes. The affirmative vote of holders of a majority of the shares of Invitae common stock represented (in person or by proxy) at the special meeting, assuming a quorum is present, is required to approve the merger agreement. If you abstain from voting, it will have the same effect as a vote cast “AGAINST” the merger proposal. If you fail to submit a proxy or to vote at the virtual special meeting, or you do not provide your bank, brokerage firm or other nominee with instructions, as applicable, it will have no effect on the vote count for the merger proposal, assuming a quorum is present. The Invitae Board unanimously recommends that stockholders vote “FOR” the proposal to adopt the merger agreement and approve the merger. |
Q: | How do I vote? |
A: | If you are a stockholder of record of Invitae as of the close of business on August 19, 2020, which is referred to as the record date, you are entitled to receive notice of, and cast a vote at, the virtual special meeting. Each holder of Invitae common stock is entitled to cast one vote on each matter properly brought before the virtual special meeting for each share of Invitae common stock that such holder owned of record as of the record date. You may submit your proxy before the special meeting in one of the following ways. |
• | Telephone voting—use the toll-free number and follow the telephone voting instructions shown on your proxy card—your vote must be received by 11:59 p.m., Eastern Time, on September 30, 2020 to be counted; |
• | Via the Internet—visit the website and follow the Internet voting instructions shown on your proxy card to vote via the Internet—your vote must be received by 11:59 p.m., Eastern Time, on September 30, 2020 to be counted; or |
• | Mail—complete, sign, date and return the enclosed proxy card in the enclosed postage-paid envelope. |
• | At the Virtual Meeting—Shares held in your name as the stockholder of record may be voted electronically at the special meeting by visiting www.virtualshareholdermeeting.com/NVTA2020SM and using the control number included on your proxy materials. The special meeting will be held at |
Q: | What is the difference between holding shares as a stockholder of record and as a beneficial owner? |
A: | Stockholder of Record. If your shares are registered directly in your name with Invitae’s transfer agent, American Stock Transfer & Trust Company, which is referred to as AST, you are considered, with respect to those shares, the stockholder of record. The proxy statement/prospectus and proxy card have been sent directly to you by Invitae. |
Q: | If my shares are held in “street name” by a broker, bank or other nominee, will my broker, bank or other nominee vote my shares for me? |
A: | If your shares are held in “street name” in a stock brokerage account or by a broker, bank or other nominee, you must provide the record holder of your shares with instructions on how to vote your shares. Please follow the voting instructions provided by your broker, bank or other nominee. Please note that you may not vote shares held in “street name” by returning a proxy card directly to Invitae or by voting in person at the special meeting unless you provide a “legal proxy,” which you must obtain from your broker, bank or other nominee. Your broker, bank or other nominee is obligated to provide you with a voting instruction card for you to use. |
• | your broker, bank or other nominee may not vote your shares on the merger proposal, which broker non-votes, if any, will have the no effect on the outcome of such proposal (assuming a quorum is present); |
• | your broker, bank or other nominee may not vote your shares on the securities issuance proposal, which broker non-votes, if any, will have no effect on the outcome of such proposal (assuming a quorum is present); and |
• | your broker, bank or other nominee may not vote your shares on the adjournment proposal, which broker non-votes, if any, will have no effect on the outcome of such proposal (regardless of whether a quorum is present). |
Q: | Why is Invitae holding a virtual special meeting? |
A: | After careful consideration, in light of the on-going developments related to the COVID-19 pandemic and to protect the health of Invitae employees, stockholders and the community, the special meeting will be a completely virtual meeting of stockholders conducted via live audio webcast. In addition, Invitae believes that the virtual meeting format will expand stockholder access and participation. You will not be able to attend the special meeting in person. |
Q: | How can I attend the virtual special meeting? |
A: | The special meeting will be a completely virtual meeting of stockholders conducted exclusively via live audio webcast. You will be able to attend the special meeting via live audio webcast by visiting www.virtualshareholdermeeting.com/NVTA2020SM. To participate in the special meeting, you will also need a control number, which is included on your proxy materials. The special meeting will begin promptly at 10:00 A.M., Pacific Time, on October 1, 2020. Invitae encourages you to access the virtual meeting website prior to the start time. Online check-in will begin at 9:45 A.M., Pacific Time, and you should allow ample time for the check-in procedures. |
Q: | What if I have technical difficulties accessing or participating in the virtual special meeting? |
A: | Invitae will have technicians ready to assist you with technical difficulties you may have accessing the virtual special meeting, voting at the special meeting or submitting questions at the special meeting. Please refer to the technical support information located at www.virtualshareholdermeeting.com/NVTA2020SM. |
Q: | What if I fail to vote or abstain? |
A: | For purposes of the special meeting, an abstention occurs when a stockholder attends the virtual special meeting and does not vote or returns a proxy with an “abstain” instruction. |
Q: | What will happen if I sign and return my proxy or voting instruction card without indicating how to vote? |
A: | If you sign and return your proxy or voting instruction card without indicating how to vote on any particular proposal, the common stock represented by your proxy will be voted as recommended by the Invitae Board with respect to that proposal. |
Q: | What happens if I sell my shares of Invitae common stock after the record date but before the special meeting? |
A: | The record date for the special meeting (the close of business on August 19, 2020) is earlier than the date of the special meeting and earlier than the date that the merger is expected to be completed. If you sell or otherwise transfer your shares of common stock after the record date but before the date of the special meeting, you will retain your right to vote at the special meeting. |
Q: | May I change or revoke my vote after I have delivered my proxy or voting instruction card? |
A: | Yes. If you are a record holder, you may change or revoke your vote before your proxy is voted at the special meeting. If you submitted your proxy by mail, you must file with the Secretary of Invitae a written notice of revocation or deliver, prior to the vote at the special meeting, a valid, later-dated proxy. If you submitted your vote by telephone or by the Internet, you may change your vote or revoke your proxy with a later telephone or Internet proxy, as the case may be. Please note that telephone and Internet voting have a deadline of 11:59 p.m., Eastern Time, on September 30, 2020 for any votes made through those methods to be counted. You may also attend the special meeting via live audio webcast, which would have the effect of revoking any proxy you provided before the special meeting. |
Q: | Where can I find the voting results of the special meeting? |
A: | The preliminary voting results will be announced at the special meeting. In addition, within four business days following certification of the final voting results, Invitae intends to file the final voting results with the SEC on a Current Report on Form 8-K. |
Q: | What are the U.S. federal income tax consequences of the merger? |
A: | The merger transactions are intended to be considered together as a single integrated transaction for U.S. federal income tax purposes which single integrated transaction is intended to qualify as a “reorganization” within the meaning of Section 368(a) of the Code. |
• | will recognize any gain, but not loss, realized as a result of the merger, but not in excess of the amount of cash received (excluding cash received in lieu of fractional Invitae shares, but including a U.S. ArcherDX holder’s share of the cash deposited in the Holders’ Representative expense fund), |
• | will have a tax basis for the Invitae shares (including fractional Invitae shares and Milestone Shares, but excluding the portion of Milestone Shares treated as interest income) that reflects a U.S. ArcherDX holder’s tax basis for the ArcherDX shares exchanged, and |
• | will have a holding period for the Invitae shares (including fractional Invitae shares and Milestone Shares, but excluding the portion of Milestone Shares treated as interest income) that includes a U.S. ArcherDX holder’s holding period for the ArcherDX shares exchanged. |
Q: | Are there any risks that I should consider in deciding whether to vote in favor of the merger proposal? |
A: | Yes. You should read and carefully consider the risk factors set forth in “Risk Factors” beginning on page 26. You also should read and carefully consider the risk factors of Invitae contained in the documents that are incorporated by reference into this proxy statement/prospectus. |
Q: | Do I have appraisal rights in connection with the merger? |
A: | Invitae stockholders do not have appraisal rights in connection with the merger. |
Q: | What will holders of ArcherDX equity-based awards receive in the merger? |
A: | Subject to the terms and conditions of the merger agreement, including adjustment to the cash consideration payable, if the merger is completed, (i) each outstanding and unexercised ArcherDX stock option will be converted into (a) approximately $9.05 in cash (less the applicable per share exercise price), without interest, plus (b) an option to (x) purchase shares of Invitae common stock plus (y) additional shares of Invitae common stock if certain milestones are achieved, subject, in the case of employees or consultants of ArcherDX at the time of the first merger, to continued service at the time of achievement of such milestone, and (ii) each unexpired, unexercised and outstanding ArcherDX warrant will be converted into (a) a number of shares of Invitae common stock equal to 30.0 million divided by the fully diluted shares of ArcherDX, plus (b) approximately $9.05 in cash (less the applicable per share exercise price), plus (c) additional shares of Invitae common stock if certain milestones are achieved, generally subject, in the case of employees or consultants of ArcherDX at the time of the merger, to continued service at the time of achievement of such milestone. For more details on the calculation of the Invitae stock price and the calculation of the exchange ratio, see “The Merger Agreement—Treatment of ArcherDX Options” beginning on page 84 and “The Merger Agreement—Treatment of ArcherDX Warrants” beginning on page 84. |
Q: | Whom should I contact if I have any questions about the proxy materials or voting? |
A: | If you have any questions about the proxy materials, or if you need assistance submitting your proxy or voting your shares or need additional copies of this proxy statement/prospectus or the enclosed proxy card, you should contact D.F. King & Co., Inc., the proxy solicitor for Invitae, at (866) 388-7535 or invitae@dfking.com. |
• | “FOR” the merger proposal; |
• | “FOR” the securities issuance proposal; and |
• | “FOR” the adjournment proposal. |
• | by either Invitae or ArcherDX: |
○ | upon written notice to the other party, if the merger has not been completed on or before March 20, 2021, which date may be extended from time to time by mutual written consent of Invitae and ArcherDX, except where the party seeking to terminate the merger agreement for this reason has failed to perform any of its obligations under the merger agreement and such failure to perform was the principal cause of or directly resulted in the failure of the completion of the merger on or before March 20, 2021; |
○ | if any governmental authority of competent jurisdiction has issued or entered any final and non-appealable order or enacted any law after the date of the merger agreement that has the effect of restraining, enjoining, preventing, making illegal or otherwise prohibiting the merger, except that the right to terminate the merger agreement is not available to a party if a material breach by such party of its obligations under the merger agreement has been the principal cause of or principally resulted in the issuance of such order; or |
○ | upon written notice to the other party, the Invitae special meeting of stockholders has concluded and the required stockholder approval by Invitae stockholders was not obtained; |
• | by ArcherDX: |
○ | if any of the representations or warranties of Invitae set forth in the merger agreement are not true and correct or if Invitae, Merger Sub A or Merger Sub B have failed to perform any covenant or agreement on the part of Invitae, Merger Sub A or Merger Sub B, as applicable, set forth in the merger agreement (including an obligation to consummate the closing) such that the conditions to closing set forth in the merger agreement would not be satisfied and the breach or breaches causing such representations or warranties not to be accurate, or the failures to perform any covenant or agreement, as applicable, are not cured on or prior to the earlier of (i) thirty (30) days after written notice thereof is delivered to Invitae and (ii) March 20, 2021; provided that this provision will not be available to ArcherDX if ArcherDX is then in material breach of the merger agreement; or |
○ | upon written notice to Invitae, if the Invitae Board has made an adverse recommendation change, as such term is defined in the merger agreement; |
• | by Invitae, if any of the representations or warranties of ArcherDX set forth in the merger agreement are not accurate or if ArcherDX has failed to perform any covenant or agreement on the part of ArcherDX set forth in the merger agreement (including an obligation to consummate the closing) such that the conditions to closing set forth in the merger agreement would not be satisfied and the breach or breaches causing such representations or warranties not to be accurate, or the failures to perform any covenant or agreement, as applicable, are not cured on or prior to the earlier of (i) thirty (30) days after written notice thereof is delivered to ArcherDX and (ii) March 20, 2021; provided that this provision will not be available to Invitae if Invitae is then in material breach of the merger agreement. |
• | each party shall remain liable for its willful breach of the merger agreement prior to termination; |
• | the termination provisions of the merger agreement will remain in effect; |
• | the Holders’ Representative will not be liable to the Holders for any action taken or omitted to be taken in connection with his services pursuant to the merger agreement or any ancillary agreements, except in certain limited cases; |
• | the merger agreement and claims or causes of action arising under the merger agreement will be governed by Delaware law; and |
• | any action relating to the merger agreement or the enforcement of any provision of the merger agreement will be brought exclusively in the Delaware Chancery Court, and to the extent that the Delaware Chancery Court rejects jurisdiction, in any state or federal court located in the Delaware county of New Castle. |
• | will recognize any gain, but not loss, realized as a result of the merger, but not in excess of the amount of cash received (excluding cash received in lieu of fractional Invitae shares, but including a U.S. ArcherDX holder’s share of the cash deposited in the Holders’ Representative expense fund), |
• | will have a tax basis for the Invitae shares (including fractional Invitae shares and Milestone Shares, but excluding the portion of Milestone Shares treated as interest income) that reflects a U.S. ArcherDX holder’s tax basis for the ArcherDX shares exchanged, and |
• | will have a holding period for the Invitae shares (including fractional Invitae shares and Milestone Shares, but excluding the portion of Milestone Shares treated as interest income) that includes a U.S. ArcherDX holder’s holding period for the ArcherDX shares exchanged. |
| | | Six Months Ended June 30, | | | Years Ended December 31, | ||||||||||||||||
(In thousands, except per share data) | | | 2020(1) | | | 2019(1) | | | 2019(1) | | | 2018(3) | | | 2017 | | | 2016 | | | 2015 |
| | | Unaudited | | | | | | | | | | | |||||||||
Consolidated Statements of Operations Data: | | | | | | | | | | | | | | | |||||||
Revenue | | | | | | | | | | | | | | | |||||||
Test revenue | | | $108,177 | | | $91,921 | | | $212,473 | | | $144,560 | | | $65,169 | | | $24,840 | | | $8,378 |
Other revenue | | | 2,262 | | | 2,107 | | | 4,351 | | | 3,139 | | | 3,052 | | | 208 | | | — |
Total revenue | | | 110,439 | | | 94,028 | | | 216,824 | | | 147,699 | | | 68,221 | | | 25,048 | | | 8,378 |
Cost of revenue | | | 83,374 | | | 49,260 | | | 118,103 | | | 80,105 | | | 50,142 | | | 27,878 | | | 16,523 |
Research and development | | | 130,631 | | | 43,296 | | | 141,526 | | | 63,496 | | | 46,469 | | | 44,630 | | | 42,806 |
Selling and marketing | | | 81,640 | | | 54,972 | | | 122,237 | | | 74,428 | | | 53,417 | | | 28,638 | | | 22,479 |
General and administrative | | | 54,660 | | | 34,593 | | | 79,070 | | | 52,227 | | | 39,472 | | | 24,085 | | | 16,047 |
Loss from operations | | | (239,866) | | | (88,093) | | | (244,112) | | | (122,557) | | | (121,279) | | | (100,183) | | | (89,477) |
Other expense, net | | | (16,728) | | | 2,019 | | | (3,891) | | | (2,568) | | | (303) | | | 348 | | | (94) |
Interest expense | | | (10,936) | | | (4,229) | | | (12,412) | | | (7,030) | | | (3,654) | | | (421) | | | (211) |
Net loss before taxes | | | (267,530) | | | (90,303) | | | (260,415) | | | (132,155) | | | (125,236) | | | (100,256) | | | (89,782) |
Income tax benefit | | | (2,600) | | | (3,950) | | | (18,450) | | | (2,800) | | | (1,856) | | | — | | | — |
Net loss | | | $(264,930) | | | $(86,353) | | | $(241,965) | | | $(129,355) | | | $(123,380) | | | $(100,256) | | | $(89,782) |
Net loss per share, basic and diluted | | | (2.35) | | | (1.01) | | | (2.66) | | | (1.94) | | | (2.65) | | | (3.02) | | | (3.18) |
Shares used in computing net loss per share, basic and diluted | | | 112,765 | | | 85,148 | | | 90,859 | | | 66,747 | | | 46,512 | | | 33,176 | | | 28,213 |
| | | As of June 30, | | | As of December 31, | ||||||||||||||||
(In thousands) | | | 2020(1) | | | 2019(1) | | | 2019(1)(2) | | | 2018(3) | | | 2017(1) | | | 2016 | | | 2015 |
| | | Unaudited | | | | | | | | | | | |||||||||
Consolidated Balance Sheet Data: | | | | | | | | | | | | | | | |||||||
Cash and cash equivalents | | | $168,203 | | | $247,000 | | | $151,389 | | | $112,158 | | | $12,053 | | | $66,825 | | | $73,238 |
Marketable securities | | | $253,933 | | | $800 | | | $240,436 | | | $13,727 | | | $52,607 | | | $25,798 | | | $53,780 |
Working capital | | | $344,225 | | | $247,827 | | | $360,538 | | | $129,127 | | | $53,294 | | | $87,047 | | | $120,433 |
Total assets | | | $969,875 | | | $502,902 | | | $781,601 | | | $282,959 | | | $211,078 | | | $130,651 | | | $156,676 |
Lease obligations | | | $50,329 | | | $53,159 | | | $50,071 | | | $3,312 | | | $5,412 | | | $1,575 | | | $3,164 |
Debt | | | $276,092 | | | $75,184 | | | $268,755 | | | $74,477 | | | $39,084 | | | $12,102 | | | $7,040 |
Total liabilities | | | $505,680 | | | $161,399 | | | $401,961 | | | $121,120 | | | $89,284 | | | $31,577 | | | $18,300 |
Accumulated deficit | | | $(1,023,607) | | | $(603,065) | | | $(758,677) | | | $(516,712) | | | $(398,598) | | | $(275,218) | | | $(174,962) |
Total stockholders’ equity | | | $464,195 | | | $341,503 | | | $379,640 | | | $161,839 | | | $121,794 | | | $99,074 | | | $138,376 |
(1) | In 2020, Invitae completed the acquisition of three businesses, in June 2019, Invitae completed the acquisition of Singular Bio, Inc., in July 2019, Invitae completed the acquisition of Jungla Inc., in November 2019, Invitae completed the acquisition of Clear Genetics, Inc., Invitae completed the acquisition of three businesses and in 2017, Invitae completed the acquisition of four businesses, all of which are included in Invitae’s selected consolidated financial data as of each acquisition date, as applicable. |
(2) | On January 1, 2019, Invitae adopted Accounting Standards Codification, or ASC, Topic 842 using the modified retrospective transition method which required the recognition of operating and finance lease right-of-use assets and operating lease liabilities to be recognized on its consolidated balance sheets. Prior period amounts are presented as originally reported based upon the accounting standards in effect for those periods. |
(3) | On January 1, 2018, Invitae adopted ASC Topic 606 using the modified retrospective transition method. Prior period amounts are presented as originally reported based upon the accounting standards in effect for those periods. |
| | | Six Months Ended June 30, | | | Year Ended December 31, | |||||||
| | | 2020 | | | 2019 | | | 2019 | | | 2018 | |
| | | (In thousands, except per share data) | ||||||||||
Consolidated Statements of Operations Data: | | | | | | | | | ||||
Revenue | | | | | | | | | ||||
Precision oncology products | | | $15,410 | | | $9,475 | | | $22,644 | | | $16,025 |
Pharma development services | | | 15,718 | | | 10,773 | | | 27,921 | | | 12,429 |
Total Revenue | | | 31,128 | | | 20,248 | | | 50,565 | | | 28,454 |
Costs & operating expenses | | | | | | | | | ||||
Cost of precision oncology products | | | 5,451 | | | 2,593 | | | 7,335 | | | 4,033 |
Cost of pharma development services | | | 7,655 | | | 3,446 | | | 9,212 | | | 6,230 |
Sales and marketing | | | 10,185 | | | 5,544 | | | 15.428 | | | 7.215 |
Research and development | | | 35,853 | | | 9,301 | | | 34.172 | | | 8.184 |
General and administrative | | | 18,518 | | | 5,709 | | | 15,875 | | | 7,700 |
Contingent consideration | | | 39,105 | | | 5,506 | | | 5,768 | | | — |
Total operating expenses | | | 116,767 | | | 32,099 | | | 87,790 | | | 33,362 |
Loss from operations | | | (85,639) | | | (11,851) | | | (37,225) | | | (4,908) |
Interest expense, net | | | (1,998) | | | (769) | | | (2,432) | | | (1,160) |
Other income (expense), net | | | (30,147) | | | (219) | | | (824) | | | 34 |
Loss before income taxes | | | (117,784) | | | (12,839) | | | (40,481) | | | (6,034) |
Income tax (benefit) expense | | | — | | | 7 | | | 497 | | | (481) |
Net loss and comprehensive loss | | | $(117,784) | | | $(12,846) | | | (40,978) | | | (5,553) |
Basic and diluted loss per common share | | | $(18.09) | | | $(2.01) | | | (6.39) | | | (0.87) |
Basic and diluted weighted-average common shares outstanding(1) | | | 6,511 | | | 6,377 | | | 6,415 | | | 6,377 |
(1) | See Note 15 to ArcherDX’s consolidated financial statements included elsewhere in this proxy statement/prospectus for a description of how ArcherDX computes basic and diluted net loss per share of common stock. The common shares outstanding gives effect to a 0.7039-for-1 reverse stock split of ArcherDX’s common and preferred stock, which was effective June 17, 2020. |
| | | Three Months Ended June 30, | | | Year Ended December 31, | ||||
| | | 2020 | | | 2019 | | | 2018 | |
| | | (In thousands, except per share data) | |||||||
Consolidated Balance Sheet Data: | | | | | | | |||
Cash and cash equivalents | | | $21,883 | | | $59,492 | | | $9,324 |
Total assets | | | 89,531 | | | 106,796 | | | 29,475 |
Total current assets | | | 59,078 | | | 83,290 | | | 19,231 |
Total current liabilities | | | 32,397 | | | 18,377 | | | 9,059 |
Working Capital(1) | | | 26,681 | | | 64,913 | | | 10,172 |
Long-term debt, net | | | 41,717 | | | 28,572 | | | — |
Convertible preferred stock | | | 115,347 | | | 110,154 | | | 42,180 |
Accumulated deficit | | | (187,027) | | | (69,243) | | | (28,265) |
Total stockholders’ deficit | | | (181,632) | | | (65,239) | | | (25,095) |
(1) | Working capital is defined as current assets less current liabilities. |
| | | As of June 30, 2020 | |
| | | (In millions) | |
Pro Forma Combined Balance Sheet Data: | | | |
Cash and cash equivalents | | | $233.9 |
Marketable securities | | | $253.9 |
Total assets | | | $3,186.3 |
Debt including convertible loans, net of unamortized discount and debt issuance costs | | | $384.5 |
Total stockholders' equity | | | $1,767.4 |
| | | Six Months Ended June 30, | | | Year Ended December 31, | |
| | | 2020 | | | 2019 | |
| | | (In millions, except per share data) | ||||
Pro Forma Combined Statement of Operations Data: | | | | | ||
Total revenues | | | $141.6 | | | $267.4 |
Loss from operations | | | $(319.3) | | | $(311.8) |
Net loss | | | $(353.6) | | | $(344.7) |
Net loss per share, basic and diluted | | | $(2.22) | | | $(2.48) |
| | | ArcherDX | | | Invitae | |||||||
| | | Historical | | | Pro Forma Equivalent(1) | | | Historical | | | Pro Forma Combined | |
(Loss) Income from Continuing Operations Per Share | | | | | | | | | ||||
Basic | | | | | | | | | ||||
Six Months Ended June 30, 2020 | | | $(18.09) | | | $(2.19) | | | $(2.35) | | | $(2.22) |
Year Ended December 31, 2019 | | | $(6.39) | | | $(2.45) | | | $(2.66) | | | $(2.48) |
Diluted | | | | | | | | | ||||
Six Months Ended June 30, 2020 | | | $(18.09) | | | $(2.19) | | | $(2.35) | | | $(2.22) |
Year Ended December 31, 2019 | | | $(6.39) | | | $(2.45) | | | $(2.66) | | | $(2.48) |
Cash Dividends Declared Per Share | | | | | | | | | ||||
Six Months Ended June 30, 2020 | | | None | | | None | | | None | | | None |
Year Ended December 31, 2019 | | | None | | | None | | | None | | | None |
Book Value Per Share | | | | | | | | | ||||
As of June 30, 2020 | | | $(27.41) | | | $9.81 | | | $3.54 | | | $9.94 |
(1) | Calculated by multiplying the unaudited pro forma combined per share amounts by the exchange ratio of 0.986771. |
• | the ability of Invitae and ArcherDX to satisfy the conditions to the closing of the merger on a timely basis or at all; |
• | the occurrence of events that may give rise to a right of one or both of Invitae and ArcherDX to terminate the merger agreement, including under circumstances that might require Invitae to compensate ArcherDX in the amount of $30.0 million; |
• | the possibility that the merger is delayed or does not occur; |
• | the possibility that the anticipated benefits from the merger cannot be realized in full or at all or may take longer to realize than expected, including risks associated with achieving expected synergies from the merger; |
• | negative effects of the announcement or the consummation of the merger on the market price of Invitae’s common stock and/or on the respective businesses, financial conditions, results of operations and financial performance of Invitae or ArcherDX; |
• | risks related to ArcherDX and Invitae being restricted in operating their businesses while the merger agreement is in effect; |
• | risks relating to the value of the Invitae shares to be issued in the merger, significant merger costs and/or unknown liabilities; |
• | risks associated with contracts containing consent and/or other provisions that may be triggered by the merger; |
• | risks associated with potential merger-related litigation; |
• | the possibility that costs or difficulties related to the integration of ArcherDX’s operations with those of Invitae will be greater than expected; |
• | the rate and degree of market acceptance of Invitae and ArcherDX’s tests and genetic testing generally; |
• | the ability to scale the infrastructure and operations of Invitae and ArcherDX in a cost-effective manner; |
• | the timing of and Invitae’s ability to introduce improvements to the combined company’s genetic testing platform and to expand its assays to include additional genes; |
• | the timing and results of studies with respect to Invitae and ArcherDX’s tests; |
• | developments and projections relating to competitors and the genomic testing industry; |
• | the degree to which individuals will share genetic information generally, as well as share any related potential economic opportunities with Invitae; |
• | the ability to obtain and maintain adequate reimbursement for Invitae and ArcherDX’s tests; |
• | regulatory developments in the United States and foreign countries; |
• | the ability to attract and retain key scientific or management personnel; |
• | the effect of the COVID-19 pandemic on Invitae’s or ArcherDX’s respective businesses and the success of any measures Invitae and ArcherDX have taken or may take in the future in response thereto; |
• | other risks as detailed from time to time in Invitae’s reports filed with the SEC, including Invitae’s annual report on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K and other documents filed with the SEC, including the risks and uncertainties set forth in or incorporated by reference into this proxy statement/prospectus in “Risk Factors” beginning on page 26. |
• | the market price of Invitae common stock could decline; |
• | if the merger agreement is terminated and the Invitae Board seeks another acquisition, stockholders cannot be certain that Invitae will be able to find a party willing to enter into a transaction with the same potential benefits or on terms equivalent to or more attractive than the terms that ArcherDX has agreed to in the merger agreement; |
• | time and resources, financial and other, committed by Invitae’s management to matters relating to the merger could otherwise have been devoted to pursuing other beneficial opportunities for Invitae; |
• | Invitae may experience negative reactions from the financial markets or from its customers, payers, suppliers, collaboration partners or employees; and |
• | Invitae will be required to pay its costs relating to the merger, such as legal, accounting, financial advisory and printing fees, whether or not the merger is completed. |
• | multiple, conflicting and changing laws and regulations such as privacy security and data use regulations, tax laws, export and import restrictions, economic sanctions and embargoes, employment laws, anti-corruption laws, regulatory requirements, reimbursement or payor regimes and other governmental approvals, permits and licenses; |
• | failure by ArcherDX or its distributors to obtain regulatory clearance, authorization or approval for the use of ArcherDX’s products and services in various countries; |
• | additional potentially relevant third-party patent rights; |
• | complexities and difficulties in obtaining intellectual property protection and maintaining, defending and enforcing ArcherDX’s intellectual property; |
• | difficulties in staffing and managing foreign operations; |
• | employment risks related to hiring employees outside the United States; |
• | complexities associated with managing multiple payor reimbursement regimes, government payers or patient self-pay systems; |
• | difficulties in negotiating favorable reimbursement negotiations with governmental authorities; |
• | logistics and regulations associated with shipping samples, including infrastructure conditions and transportation delays; |
• | limits in ArcherDX’s ability to penetrate international markets if it is not able to sell its products or conduct its testing or clinical diagnostic services locally; |
• | financial risks, such as longer payment cycles, difficulty collecting accounts receivable, the impact of local and regional financial crises on demand and payment for ArcherDX’s products and services and exposure to foreign currency exchange rate fluctuations; |
• | natural disasters, political and economic instability, including wars, terrorism and political unrest, outbreak of disease, boycotts, curtailment of trade and other business restrictions; |
• | regulatory and compliance risks that relate to maintaining accurate information and control over sales and distributors’ activities that may fall within the purview of the U.S. Foreign Corrupt Practices Act, or FCPA, its books and records provisions, or its anti-bribery provisions, or laws similar to the FCPA in other jurisdictions in which ArcherDX may now or in the future operate, such as the United Kingdom’s Bribery Act of 2010, or the U.K. Bribery Act; and |
• | onerous anti-bribery requirements of several member states in the EU, the United Kingdom, Japan, and other countries that are constantly changing and require disclosure of information to which U.S. legal privilege may not extend. |
• | A belief that uniting Invitae and ArcherDX would accelerate the ability of the combined company to offer comprehensive support for precision oncology, including due to the potential represented by the combination of (i) Invitae’s platform, including leadership in diagnostic and hereditary risk testing and strong relationships with clinicians caring for cancer patients, including cancer genetic counselors, oncologists and imaging centers, and (ii) ArcherDX’s platform, with its proprietary Anchored Multiplex PCR (AMPTM) chemistry at the core, which has enabled ArcherDX to develop products and services under investigation to optimize therapy and enable cancer monitoring across liquid and tissue samples; |
• | The expertise and market position of ArcherDX which has (i) developed and commercialized over 325 unique products, including research products and services in use by more than 300 laboratories worldwide, and (ii) collaborated with more than 50 biopharmaceutical companies and contract research organizations, providing services that enable biopharmaceutical companies, including partners such as Merck KGaA, Darmstadt, Germany, AstraZeneca PLC, Bristol Myers Squibb Company and Bayer AG, to cost-effectively accelerate drug development; |
• | The ongoing development efforts of ArcherDX, which is currently developing in-vitro diagnostic, or IVD, products for U.S. Food and Drug Administration, or FDA, submissions, including STRATAFIDE DX for therapy optimization and the broadly applicable PCM in development for disease recurrence monitoring and therapy optimization including selection, response and modulation, with both STRATAFIDE DX and PCM having received Breakthrough Device designation from the FDA; and |
• | A view that the combination of Invitae and ArcherDX will: (i) be ideally positioned to serve customers across the continuum, from individuals and community clinicians to biopharmaceutical partners, distribution partners, reference laboratories and academic centers, with Invitae’s central laboratory providing support for customers who prefer to send out and can benefit from a full suite of services including reporting, clinician consultation and genetic counseling for patients, while ArcherDX’s decentralized model supports geographies and customers where local control of patient reporting is either desired or required; (ii) with both centralized and local testing capabilities, offer breadth and flexibility in serving customers in more than 95 markets; and (iii) offer robust support for biopharmaceutical companies, from patient identification and screening, to biomarker identification and companion diagnostic development. |
• | reviewed certain internal financial statements, analyses and forecasts, and other financial and operating data relating to the business of ArcherDX, in each case, prepared by management of ArcherDX, and approved for Perella Weinberg’s use by Invitae, or the ArcherDX Forecasts; |
• | reviewed certain adjustments to the ArcherDX Forecasts prepared by management of Invitae, or the Adjusted ArcherDX Forecasts; |
• | reviewed certain publicly-available Wall Street research analyst estimates relating to Invitae approved for Perella Weinberg’s use by Invitae, or the Invitae Research Estimates; |
• | discussed the past and current business, operations, financial condition and prospects of ArcherDX with representatives of ArcherDX and Invitae; |
• | discussed the past and current business, operations, financial condition and prospects of Invitae with representatives of Invitae; |
• | compared the financial performance of ArcherDX and Invitae with that of certain publicly-traded companies which Perella Weinberg believed to be generally relevant; |
• | compared the financial terms of the merger with the publicly available financial terms of certain transactions which Perella Weinberg believed to be generally relevant; |
• | reviewed analyses and forecasts of the amount and timing of certain cost savings, operating efficiencies, financial synergies and other strategic benefits projected by Invitae to be achieved as a result of the merger, collectively referred to as the Synergies; |
• | participated in discussions among representatives of ArcherDX and Invitae and their respective advisors; |
• | reviewed a draft, dated June 20, 2020, of the merger agreement; and |
• | conducted such other financial studies, analyses and investigations, and considered such other factors, as Perella Weinberg deemed appropriate. |
Selected ArcherDX Publicly Traded Companies |
10x Genomics, Inc. |
Adaptive Biotechnologies Corporation |
CareDx, Inc. |
Guardant Health, Inc. |
NanoString Technologies, Inc. |
Natera, Inc. |
Veracyte, Inc. |
EV/2020E and 2021E Revenue Multiples | | | 2020E | | | 2021E |
Selected ArcherDX Publicly Traded Companies | | | | | ||
10x Genomics, Inc. | | | 35.2x | | | 20.7x |
Adaptive Biotechnologies Corporation | | | 63.8x | | | 33.3x |
CareDx, Inc. | | | 9.3x | | | 7.2x |
Guardant Health, Inc. | | | 25.4x | | | 19.0x |
NanoString Technologies, Inc. | | | 12.3x | | | 9.0x |
Natera, Inc. | | | 11.7x | | | 10.1x |
Veracyte, Inc. | | | 11.8x | | | 7.6x |
Median | | | 12.3x | | | 10.1x |
Invitae | | | 10.0x | | | 5.9x |
ArcherDX @ Aggregate consideration | | | 24.0x | | | 14.6x |
Announcement Date | | | Target | | | Acquiror | | | EV/ LTM Revenue Multiple |
March 2019 | | | Brammer Bio | | | Thermo Fisher Scientific Inc. | | | 12.1x |
November 2018* | | | Pacific Biosciences of California, Inc. | | | Illumina, Inc. | | | 13.8x |
June 2018 | | | Exosome Diagnostics, Inc. | | | Bio-Techne Corporation | | | NA |
June 2018 | | | Foundation Medicine, Inc. | | | F. Hoffmann-La Roche AG | | | 29.4x |
June 2017 | | | Spectrum, Inc. | | | Repligen Corporation | | | 8.9x |
July 2016 | | | Advanced Cell Diagnostics | | | Bio-Techne Corporation | | | 13.0x |
| | | | | | | ||||
Mean | | | | | | | 15.5x | ||
Median | | | | | | | 13.0x |
* | Transaction was terminated on January 2, 2020. |
• | calculating the present value as of June 19, 2020 of the estimated standalone unlevered free cash flows (calculated as operating income after taxes, plus depreciation and amortization, minus capital |
• | adding the present value as of June 19, 2020 of the terminal value of ArcherDX at the end of calendar year 2030 using perpetuity growth rates ranging from 2.5% to 3.5% and using discount rates ranging from 11.0% to 13.0% (assuming a perpetuity growth rate of 3.0% and a discount rate of 12.0%, Perella Weinberg noted that the present value as of June 19, 2020 of the terminal value of ArcherDX at the end of calendar year 2030 was approximately $1,923 million), and |
• | adding the approximately $23 million present value (using a discount rate of 12.0%) as of June 19, 2020 of ArcherDX’s net operating losses of ArcherDX included in the Adjusted ArcherDX Forecasts. |
Selected Invitae Publicly Traded Companies |
CareDx, Inc. |
Fluidigm Corporation |
Luminex Corporation |
Myriad Genetics, Inc. |
Natera, Inc. |
NeoGenomics, Inc. |
OraSure Technologies, Inc. |
Oxford Immunotec, Inc. |
Veracyte, Inc. |
EV/2020E and 2021E Revenue Multiples | | | 2020E | | | 2021E |
Selected Invitae Publicly Traded Companies | | | | | ||
CareDx, Inc. | | | 9.3x | | | 7.2x |
Fluidigm Corporation | | | 2.8x | | | 2.3x |
Luminex Corporation | | | 3.6x | | | 3.3x |
Myriad Genetics, Inc. | | | 1.4x | | | 1.3x |
Natera, Inc. | | | 11.7x | | | 10.1x |
NeoGenomics, Inc. | | | 7.1x | | | 5.9x |
OraSure Technologies, Inc. | | | 3.0x | | | 2.7x |
Oxford Immunotec, Inc. | | | 3.7x | | | 2.4x |
Veracyte, Inc. | | | 11.8x | | | 7.6x |
Median | | | 3.7x | | | 3.3x |
Invitae | | | 10.0x | | | 5.9x |
• | calculating the present value as of June 19, 2020 of the estimated standalone unlevered free cash flows (calculated as operating income after taxes, plus depreciation and amortization, minus capital expenditures, and adjusting for changes in net working capital) that Invitae could generate for the complete calendar years 2020 through 2030 using discount rates ranging from 10.5% to 12.5% based on estimates of the weighted average cost of capital of Invitae derived using the CAPM, |
• | adding the present value as of June 19, 2020 of the terminal value of Invitae at the end of calendar year 2030 using perpetuity growth rates ranging from 2.5% to 3.5% and using discount rates ranging from 10.5% to 12.5% (assuming a perpetuity growth rate of 3.0% and a discount rate of 11.5%, Perella Weinberg noted that the present value as of June 19, 2020 of the terminal value of Invitae at the end of calendar year 2030 was approximately $1,671 million), and |
• | adding the approximately $152 million present value (using a discount rate of 11.5%) as of June 19, 2020 of Invitae’s net operating losses as provided by management of Invitae based upon reported net operating losses as of December 31, 2019 and net operating losses projected to be generated during the applicable period. |
| | | Year Ended December 31, | |||||||||||||||||||||||||||||||
| | | 2020E | | | 2021E | | | 2022E | | | 2023E | | | 2024E | | | 2025E | | | 2026E | | | 2027E | | | 2028E | | | 2029E | | | 2030E | |
| | | ($ in millions) | |||||||||||||||||||||||||||||||
Revenues | | | $58 | | | $95 | | | $193 | | | $296 | | | $434 | | | $622 | | | $839 | | | $1,120 | | | $1,489 | | | $2,030 | | | $2,863 |
Operating Income (Loss) After Taxes | | | $(47) | | | $(5) | | | $32 | | | $71 | | | $115 | | | $187 | | | $231 | | | $305 | | | $363 | | | $438 | | | $541 |
Depreciation and Amortization | | | $2 | | | $5 | | | $8 | | | $11 | | | $14 | | | $11 | | | $16 | | | $23 | | | $32 | | | $46 | | | $69 |
Capital Expenditures | | | $(11) | | | $(6) | | | $(6) | | | $(10) | | | $(13) | | | $(15) | | | $(20) | | | $(27) | | | $(37) | | | $(51) | | | $(73) |
Change in Net Working Capital | | | $(5) | | | $1 | | | $(10) | | | $(13) | | | $(18) | | | $(24) | | | $(30) | | | $(38) | | | $(56) | | | $(86) | | | $(136) |
Unlevered Free Cash Flows(1) | | | $(61) | | | $(5) | | | $23 | | | $59 | | | $97 | | | $159 | | | $197 | | | $263 | | | $302 | | | $346 | | | $402 |
(1) | Non-GAAP financial measure calculated as operating income (loss) after taxes, plus depreciation and amortization, less capital expenditures, and less (plus) change in net working capital. |
| | | Year Ended December 31, | |||||||||||||||||||||||||||||||
| | | 2020E | | | 2021E | | | 2022E | | | 2023E | | | 2024E | | | 2025E | | | 2026E | | | 2027E | | | 2028E | | | 2029E | | | 2030E | |
| | | ($ in millions) | |||||||||||||||||||||||||||||||
Revenues | | | $262 | | | $447 | | | $597 | | | $806 | | | $1,082 | | | $1,396 | | | $1,728 | | | $2,050 | | | $2,325 | | | $2,516 | | | $2,591 |
Operating Income (Loss) After Taxes | | | $(288) | | | $(240) | | | $(103) | | | $(99) | | | $(80) | | | $(35) | | | $42 | | | $151 | | | $285 | | | $433 | | | $574 |
Depreciation and Amortization | | | $6 | | | $9 | | | $12 | | | $17 | | | $24 | | | $33 | | | $43 | | | $53 | | | $62 | | | $70 | | | $74 |
Capital Expenditures | | | $(12) | | | $(18) | | | $(21) | | | $(28) | | | $(37) | | | $(46) | | | $(56) | | | $(65) | | | $(73) | | | $(77) | | | $(78) |
Change in Net Working Capital | | | $(1) | | | $(7) | | | $(7) | | | $(12) | | | $(19) | | | $(24) | | | $(29) | | | $(31) | | | $(30) | | | $(22) | | | $(10) |
Unlevered Free Cash Flows(1) | | | $(295) | | | $(256) | | | $(119) | | | $(122) | | | $(111) | | | $(72) | | | $(12) | | | $67 | | | $171 | | | $290 | | | $411 |
(1) | Non-GAAP financial measure calculated as operating income (loss) after taxes, plus depreciation and amortization, less capital expenditures, and less (plus) change in net working capital. |
• | ArcherDX stockholders who are not U.S. ArcherDX holders, |
• | partnerships (or entities or arrangements treated as partnerships for U.S. federal income tax purposes) and partners therein, |
• | other pass-through entities or investors in such entities, |
• | financial institutions, |
• | insurance companies, |
• | regulated investment companies or real estate investment trusts, |
• | controlled foreign corporations, |
• | passive foreign investment companies, |
• | tax-exempt entities or governmental organizations, |
• | U.S. expatriates or former long-term residents of the United States, |
• | U.S. ArcherDX holders (as defined below) whose functional currency is not the U.S. dollar, |
• | dealers or brokers in securities or foreign currencies, |
• | traders in securities electing to apply a mark-to-market method of accounting, |
• | stockholders who hold ArcherDX shares as part of a hedge, appreciated financial position, straddle, constructive sale or conversion transaction, |
• | stockholders who acquired their ArcherDX shares pursuant to the exercise of employee stock options or otherwise as compensation, |
• | stockholders who acquired their ArcherDX shares pursuant to the exercise of warrants or conversion rights under convertible instruments, |
• | tax-qualified retirement plans, |
• | “qualified foreign pension funds” as defined in Section 897(l)(2) of the Code and entities all of the interests of which are held by qualified foreign pension funds, |
• | stockholders who hold their ArcherDX shares as “qualified small business stock” for purposes of Section 1202 of the Code, |
• | stockholders who hold their ArcherDX shares as “Section 1244 stock,” |
• | stockholders who acquired their ArcherDX shares in a transaction subject to the gain rollover provisions of Section 1045 of the Code, |
• | stockholders who are accrual method taxpayers subject to special tax accounting rules under Section 451(b) of the Code, |
• | stockholders deemed to hold ArcherDX shares under the constructive sale provisions of the Code, and |
• | stockholders who exercise dissenters’ rights. |
• | the gain, if any, realized by a U.S. ArcherDX holder who receives a combination of cash (excluding cash received in lieu of fractional Invitae shares, but including the U.S. ArcherDX holder’s share of the cash deposited in the Holders’ Representative expense fund) and Invitae shares (including fractional Invitae shares and Milestone Shares, but excluding the portion of Milestone Shares treated as interest income) in exchange for ArcherDX shares pursuant to the merger generally will be recognized, but not in excess of the amount of cash received (again, excluding cash received in lieu of fractional Invitae shares, but including the U.S. ArcherDX holder’s share of the cash deposited in the Holders’ Representative expense fund); |
• | no loss will be recognized by ArcherDX stockholders as a result of the merger; |
• | the aggregate tax basis of the Invitae shares received by a U.S. ArcherDX holder pursuant to the merger (including fractional Invitae shares and Milestone Shares, but excluding the portion of Milestone Shares treated as interest income) will be the same as the aggregate tax basis of the ArcherDX shares surrendered in exchange therefor, decreased by the amount of cash received |
• | the holding period of the Invitae shares (including fractional Invitae shares and Milestone Shares, but excluding the portion of Milestone Shares treated as interest income) received in exchange for ArcherDX shares will include the holding period of such ArcherDX shares; and |
• | a U.S. ArcherDX holder receiving cash instead of a fractional Invitae share will generally be treated as having received the fractional share pursuant to the merger and then as having sold that fractional Invitae share for cash. As a result, such U.S. ArcherDX holder will generally recognize gain equal to the difference between the amount of cash received and the tax basis allocated to such fractional Invitae share. |
• | the number of shares of Invitae common stock equal to the quotient of (i) 30.0 million shares of Invitae common stock (appropriately adjusted to reflect certain stock events as set forth in the merger agreement), divided by (ii) the fully diluted shares of ArcherDX (which shares of Invitae common stock will be subject to the Lock-Up Restriction (as defined below)); |
• | an amount of cash equal to the quotient of (i) the sum of (x) $325.0 million (subject to certain adjustments for ArcherDX’s debt, transaction expenses, net working capital amounts and the exercise of ArcherDX options and warrants), minus (y) the $1,000,000 expense fund set aside for the Holders’ Representative (as defined in the “Holders’ Representative” section below), divided by (ii) the fully diluted shares of ArcherDX; |
• | after any Milestone (as defined and set forth below) is achieved, a certificate or book entry reflecting the number of shares of Invitae common stock (any such shares, “Milestone Shares”) equal to the quotient of (i) the difference of (A) the Milestone Shares for each Milestone (as adjusted pursuant to any exercise of the Offset Right (as defined in the “Offset Right” section below) minus (B) a portion of such Milestone Shares representing the value of certain fees owed to ArcherDX’s financial advisor in connection with the merger, divided by (ii) (A) the fully diluted shares of ArcherDX minus (B) the shares of ArcherDX common stock that were subject to outstanding options upon closing of the merger to the extent such options were held by terminated service providers who will not participate in the distribution of such Milestone Shares (which shares of Invitae common stock will be subject to the Lock-Up Restriction, if applicable); and |
• | an amount of cash equal to the quotient of (i) the $1,000,000 expense fund set aside for the Holders’ Representative, when and to the extent released to ArcherDX stockholders, optionholders and warrantholders (each a “Holder” and collectively the “Holders”) as provided in the merger agreement, divided by (ii) the fully diluted shares of ArcherDX. |
• | 5.0 million shares upon achievement of a submission-related regulatory milestone for STRATAFIDE; |
• | 8.0 million shares upon achievement of an approval-related regulatory milestone for STRATAFIDE; |
• | 5.0 million shares upon achievement of a revenue-related commercial milestone for PCM; |
• | 4.0 million shares upon achievement of a submission-related regulatory milestone PCM; and |
• | 5.0 million shares upon achievement of an approval-related regulatory milestone for PCM. |
• | the estimated cash to shares consideration ratio of each option, rounded down to the nearest full share, will be automatically converted into the right to receive (without interest) with respect to each share of ArcherDX’s common stock subject to such portion of the option, a cash payment equal to the cash portion per company option. |
• | each option other than an option referred to in the first bullet point above (i.e., the remaining options after applying the estimated cash to shares consideration ratio to each option as contemplated in the first bullet point above) will be assumed by Invitae and converted into an option to purchase shares of Invitae’s common stock. Each such converted option will continue to have and be subject to substantially the same terms and conditions as were applicable to such option immediately before the effective time of the first merger, except that (i) for the avoidance of doubt, each converted option will be fully vested and exercisable at any time, (ii) each converted option will be exercisable for that number of shares of Invitae’s common stock equal to the product (rounded down to the nearest whole number) of (1) the number of shares of ArcherDX’s common stock subject to the option immediately before the effective time of the first merger and (2) the closing exchange ratio; and (iii) the per share exercise price for each share of Invitae’s common stock issuable upon exercise of the converted option will be equal to the quotient (rounded up to the nearest whole cent) obtained by dividing (1) the exercise price per share of ArcherDX’s common stock of such option immediately before the effective time of the first merger by (2) the closing exchange ratio. Shares of Invitae common stock issuable pursuant to the converted option will be subject to the Lock-Up Restriction. |
• | each option, whether subject to the first bullet point or second bullet point above, will be entitled to receive, (i) following each Milestone date, a number of shares of Invitae’s common stock equal to the applicable per share Milestone consideration, with the shares of Invitae’s common stock represented thereby subject to the Lock-Up Restriction if applicable and (ii) an amount of cash equal to up to the quotient of (x) the $1,000,000 expense fund set aside for the Holders’ Representative, when and to the extent released to the Holders, divided by (y) the fully diluted shares of company stock; provided, however, that, as to any terminated service provider as of a Milestone date, such terminated service provider will not receive any payment in respect of such Milestone. |
• | due organization, valid existence, good standing and qualification to do business; |
• | corporate authorization of the merger agreement and the transactions contemplated by the merger agreement and the valid and binding nature of the merger agreement; |
• | the absence of any conflicts or violations of organizational documents and other material agreements or laws; |
• | required consents and approvals from governmental entities; |
• | documents filed with the SEC and financial statements; |
• | internal controls and disclosure controls and procedures relating to financial reporting; and |
• | accuracy of information supplied or to be supplied in connection with this proxy statement/prospectus. |
• | scope of business operations and subsidiaries; |
• | accuracy of corporate documents made available to Invitae; |
• | the unanimous approval and recommendation by the ArcherDX Board of the merger agreement and the transactions contemplated by the merger agreement; |
• | ArcherDX stockholder approval; |
• | capitalization; |
• | options and warrants; |
• | the absence of undisclosed liabilities or off-balance-sheet arrangements; |
• | the absence of certain changes or events; |
• | the absence of certain legal proceedings, investigations and governmental orders; |
• | compliance with laws, permits and regulatory bodies; |
• | tax matters; |
• | employee benefit plans; |
• | employment and labor matters; |
• | environmental matters; |
• | material contracts and related party transactions; |
• | title to and condition of assets; |
• | intellectual property; |
• | privacy and data security; |
• | compliance with certain domestic and foreign corruption laws and customs and international trade laws; and |
• | broker’s, finder’s and financial advisor’s fees. |
• | that generally affects the industry in the same geography in which Invitae or ArcherDX, as applicable, operates; |
• | that results from general economic or political conditions in any country where Invitae or ArcherDX, as applicable, conducts its business; |
• | arising out of changes in financial, banking or securities markets in general; |
• | arising out of acts of war, armed hostilities, terrorism, sabotage, curfews, riots, demonstrations or public disorders or escalation of worsening of such; |
• | consisting of changes in applicable laws, regulations, orders or the like issued by any governmental authority or accounting rules (including GAAP) or the enforcement or implementation thereof; |
• | consisting of any natural or man-made disaster or acts of God (including COVID-19 and related measures); |
• | the announcement, pendency of or performance of the transactions contemplated under the merger agreement, including the impact of any of the foregoing on any relationships with customers, suppliers, distributors, collaboration partners, employees or regulators; |
• | the taking of any action required by the terms of the merger agreement or taken at the written request of, or with the prior written consent of, the other party; |
• | consisting of any failure by a party to meet any internal or published projections, forecasts or revenue or earnings predictions; or |
• | arising out of or resulting from the Natera Litigation (as defined in the “Natera Litigation” section below), |
• | conduct business in the ordinary course and in compliance with all applicable laws; |
• | maintain and preserve intact the business organization and goodwill of those having business relationships with it (including with respect to the value of ArcherDX’s assets and technology and its relationships with employees, customers, suppliers, strategic partners, licensors, licensees, regulators, landlords and others having business relationships with ArcherDX or its subsidiaries) and retain the services of all present officers, directors and employees; and |
• | maintain in full force and effect all insurance policies that are material to its business, as in effect on June 21, 2020. |
• | issue, sell, grant, dispose of, amend any term of, grant registration rights with respect to, pledge or otherwise encumber any shares of capital stock or other equity interests, or any securities or rights convertible into, exchangeable or exercisable for, or evidencing the right to subscribe for any shares of capital stock or other equity interests, or any rights, warrants, options, calls, commitments or any other agreements of any character to purchase or acquire any shares of capital stock or other equity interests or any securities or rights convertible into, exchangeable or exercisable for, or evidencing the right to subscribe for, any shares of capital stock or other equity interests; provided, however, that ArcherDX may issue shares of its capital stock upon the exercise of options or warrants that are outstanding on June 21, 2020, in each case in accordance with the terms thereof; |
• | other than accelerating all options effective as of immediately prior to the closing of the first merger, amend or waive any rights under, or accelerate the vesting under, any provision of the ArcherDX option plan or any agreement evidencing any option, warrant, convertible security or other right to acquire ArcherDX capital stock or any restricted stock purchase agreement or any similar or related contract; |
• | redeem, purchase or otherwise acquire or cancel any outstanding shares of capital stock or equity interests, or any rights, warrants, options, calls, commitments or any other agreements of any character to acquire any shares of capital stock or equity interests, except for forfeitures of unvested options upon termination of service of any service provider; |
• | declare, set aside funds for the payment of or pay any dividend on, or make any other distribution (whether in cash, stock or property) in respect of, any shares of capital stock or other equity interests or make any payments to the Holders in their capacity as stockholders of ArcherDX; |
• | split, combine, subdivide, reclassify or take any similar action with respect to any shares of ArcherDX capital stock; |
• | form any subsidiary; |
• | incur, guarantee, issue, sell, repurchase, prepay or assume any (i) ArcherDX debt, or issue or sell any options, warrants, calls or other rights to acquire any debt securities of ArcherDX or its subsidiaries; (ii) obligations ArcherDX or its subsidiaries issued or assumed as the deferred purchase price of property; (iii) conditional sale obligations of ArcherDX or its subsidiaries; (iv) obligations of ArcherDX or its subsidiaries under any title retention agreement (but excluding trade accounts payable and other accrued current liabilities arising in the ordinary course of business); (v) obligations of ArcherDX or its subsidiaries for the reimbursement of any obligor on any letter of credit; or (vi) obligations of the type referred to in clauses (i) through (v) of other persons for the payment of which ArcherDX or its subsidiaries is responsible or liable, directly or indirectly, as obligor, guarantor, surety or otherwise, including guarantees of such obligations; |
• | sell, transfer, lease, license, mortgage, encumber or otherwise dispose of or subject to any lien other than a permitted lien, any properties or assets, in each case, outside the ordinary course of business and involving properties or assets with an aggregate fair market value in excess of $10,000,000; |
• | make any capital expenditures in excess of $20,000,000 in the aggregate; |
• | acquire or agree to acquire in any manner (whether by merger or consolidation, the purchase of an equity interest in or a material portion of the assets of or otherwise) any business or any corporation, partnership, association or other business organization or division thereof other than the acquisition of inventory and equipment in the ordinary course of business; |
• | make any investment (by contribution to capital, property transfers, purchase of securities or otherwise) in, or loan or advance funds to any person (other than travel and similar advances to employees in the ordinary course of business); |
• | with respect to contracts, (i) enter into, adopt, terminate, modify, renew or amend (including by accelerating material rights or benefits under) any material contract other than in the ordinary course of business; (ii) enter into or extend the term or scope of any contract that purports to restrict ArcherDX or its subsidiaries, or any future subsidiary of a subsidiary, from engaging in any line of business or in any geographic area; (iii) enter into any material contract (or any contract that would constitute a material contract if in effect on June 21, 2020) that would be breached by, or require the consent of any third party in order to continue in full force following, consummation of the merger; or (iv) release any person from, or modify or waive any material provision of, any confidentiality or non-disclosure agreement; |
• | (i) hire or terminate any employees, except for the termination of any employee for legitimate business purposes; (ii) increase the annual level of compensation payable or to become payable by ArcherDX or its subsidiaries to any of its directors or current employees other than in the ordinary course of business; (iii) grant any bonus, benefit or other compensation to any current employee or current consultant, except as required by the terms of the merger agreement or in the ordinary course of business; (iv) increase the coverage or benefits available under or otherwise modify or amend or terminate any (or create any new) company benefit plan, except as required by the terms of the merger agreement, applicable law or by the terms of any company benefit plan; (v) enter into any employment, deferred compensation, severance, consulting, non-competition or similar agreement to which ArcherDX or its subsidiaries is a party (or amend any such agreement in any respect) with a current employee or current consultant, except, in each case, as required by the terms of the merger agreement, applicable law from time to time in effect or by the terms of any company benefit plan; or (vi) enter into any transactions pursuant to which any related party purchases any services, products or technology from, or sells or furnishes any services, products or technology to, ArcherDX or its subsidiaries; |
• | make, change or revoke any material election concerning taxes or tax returns, file any amended tax return or any tax return in a manner materially inconsistent with past practice, enter into any closing agreement or contract with any taxing authority with respect to taxes, settle any tax claim or assessment (other than by paying taxes in the ordinary course of business), take any affirmative action to surrender any right to claim a refund of a material amount of taxes, request any tax ruling or agree to an extension or waiver of the statute of limitations with respect to the assessment or determination of taxes; |
• | make any changes in financial or tax accounting methods, principles or practices (or change an annual accounting period), except as required by applicable law; |
• | amend its certificate of incorporation or bylaws; |
• | adopt a plan or agreement for or carry out any complete or partial liquidation, dissolution, restructuring, recapitalization, merger, consolidation or other reorganization other than as required by the provisions of the merger agreement; |
• | initiate, settle, agree to settle, waive or compromise any material action; |
• | accelerate, beyond the normal collection cycle, collection of accounts receivable or delay beyond normal payment terms payment of any accounts payable, other than in the ordinary course of business; |
• | accelerate or defer the construction of any premises; |
• | accelerate or defer the purchase of fixtures, equipment, leasehold improvements or other capital expenditures, other than in the ordinary course of business; |
• | grant or agree to grant any license to any intellectual property, other than in the ordinary course of business; |
• | hire, appoint or terminate any director or officer of ArcherDX or its subsidiaries; |
• | enter into any lease (either as lessor or lessee) or other form of use or occupancy agreement for the use or occupancy of any real property or amend, in any respect, or terminate any current real property leases; or |
• | obligate itself to take any of the foregoing actions. |
• | conduct its business in compliance with all applicable laws; |
• | maintain and preserve intact its business organization and the goodwill of those having business relationships with it (including by using commercially reasonable efforts to maintain the value of its assets and technology and preserve its relationships with employees, customers, suppliers, strategic partners, licensors, licensees, regulators, landlords and others having business relationships with it or any subsidiary); and |
• | maintain in full force and effect all insurance policies that are material to its business, as in effect on June 21, 2020. |
• | split, combine, subdivide, reclassify or take any similar action with respect to any shares of capital stock in a manner that has a material adverse effect on its ability to consummate the merger or transactions contemplated under the merger agreement; |
• | adopt a plan or agreement for, or carry out, (i) any complete or partial liquidation, dissolution, restructuring or recapitalization or (ii) to the extent it would reasonably be expected to have a material adverse effect on Invitae’s ability to consummate the transactions contemplated by the merger agreement, any other merger, consolidation or other reorganization; |
• | acquire or agree to acquire in any manner (whether by merger or consolidation, the purchase of an equity interest in or a material portion of the assets of or otherwise) any business or any corporation, partnership, association or other business organization or division thereof, to the extent it would reasonably be expected to have a material adverse effect on Invitae’s ability to consummate the merger or transactions contemplated under the merger agreement; |
• | declare, set aside funds for the payment of or pay any dividend on, or make any other distribution (whether in cash, stock or property) in respect of, any shares of Invitae capital stock or make any payments to Invitae stockholders in their capacity as such; |
• | make any investment (by contribution to capital, property transfers, purchase of securities or otherwise) in, or loan or advance funds to, any person to the extent it would reasonably be expected to have a material adverse effect on Invitae’s ability to consummate the transactions contemplated under the merger agreement, any merger, consolidation or other reorganization; or |
• | amend its organizational documents in a manner that has an adverse effect on Invitae’s ability to consummate the merger or transactions contemplated under the merger agreement. |
• | the obtaining of all actions or non-actions, consents, approvals, registrations, waivers, permits, authorizations, orders, expirations or terminations of waiting periods and other confirmations from any governmental authority or other person that are or may become necessary, proper or advisable in connection with the consummation of the transactions contemplated by the merger agreement; |
• | the preparation and making of all registrations, filings, forms, notices, petitions, statements, submissions of information, applications and other documents (including filings with governmental authorities) that are or may become necessary, proper or advisable in connection with the consummation of the transactions contemplated by the merger agreement; |
• | the taking of all steps as may be necessary, proper or advisable to obtain an approval from, or to avoid an action by, any governmental authority or other person in connection with the consummation of the transactions contemplated by the merger agreement; and |
• | the defending of any actions, whether judicial or administrative, challenging the merger agreement or that would otherwise prevent or delay the consummation of the transactions contemplated by the merger agreement, including the merger, performed or consummated by each party in accordance with the terms of the merger agreement, including seeking to have any stay, temporary restraining order or injunction entered by any governmental authority vacated or reversed. |
• | consultation and consent rights regarding any press releases or other public statements with respect to the merger agreement, the merger, or the other transactions contemplated by the merger agreement; |
• | certain additional employee and employee benefit matters; |
• | the conduct of Invitae, Merger Sub A and Merger Sub B; |
• | certain tax matters; |
• | certain reporting requirements under Section 16(a) of the Exchange Act; and |
• | the approval for the listing of the Invitae common stock to be issued in connection with the merger on NYSE. |
• | the representations and warranties of ArcherDX set forth in the merger agreement, without giving effect to qualifications in such representations and warranties with respect to materiality, material adverse or similar qualifiers, being true and correct at and as of the closing date of the merger (or, in the case of representations and warranties made as of a specific date, as of such date), and where the failure of the representations and warranties of ArcherDX set forth in the merger agreement to be accurate, individually or in the aggregate, has not had a material adverse effect; provided, however, that the representations and warranties of ArcherDX related to capitalization will be accurate in all respects at and as of the date of the merger agreement and the closing date, except for inaccuracies that are de minimis, both individually and in the aggregate; |
• | ArcherDX having performed in all material respects all covenants, agreements and obligations required to be performed under the merger agreement at or prior to the completion of the merger; |
• | ArcherDX having not experienced a material adverse effect since June 21, 2020; |
• | no action having been instituted, commenced, threatened or remaining pending that seeks to or would reasonably be expected to (i) restrain, prevent, enjoin, prohibit or make illegal the transactions, (ii) cause the transactions to be rescinded following the closing date of the merger, (iii) impose limitations on the ability of the surviving company to conduct its business following the closing date of the merger or (iv) compel Invitae or ArcherDX to dispose of any portion of the business of ArcherDX; |
• | any applicable waiting period (and any extension thereof) under the HSR Act relating to the completion of the merger having expired or early termination thereof having been granted; |
• | no governmental authority of competent jurisdiction having issued or entered any order or promulgated or enacted any law after the date of the merger agreement, in each case that is in effect, having the effect of enjoining, preventing or otherwise prohibiting the completion of the merger; |
• | Invitae having received a certificate from the Chief Executive Officer or the Chief Financial Officer of ArcherDX certifying that certain of the above conditions have been satisfied; |
• | Invitae having received a certificate from the Chief Executive Officer or the Chief Financial Officer of ArcherDX certifying that the allocation schedule delivered to Invitae is accurate; |
• | Invitae having received certificates of good standing with respect to ArcherDX issued by its jurisdiction of organization and the jurisdiction of its principal place of business, dated not more than ten business days prior to the closing date; |
• | Invitae having received a certificate from the Chief Executive Officer or the Chief Financial Officer of ArcherDX conforming to the requirements of Treasury Regulation Sections 1.897-2(h) and 1.1445-2(c)(3) and the notice to the IRS in accordance with the provisions of Treasury Regulations Section 1.897-2(h)(2) together with written authorization for Invitae to deliver such notice and a copy of such certificate to the IRS on behalf of ArcherDX upon the closing of the merger; |
• | Invitae having received the duly executed certificate of reverse merger from ArcherDX; |
• | Invitae having received from ArcherDX the resignations of each officer and director, other than those continuing officers and directors specified by Invitae, in form and substance reasonably satisfactory to Invitae; |
• | Invitae having received from ArcherDX payoff letters from each lender to ArcherDX evidencing the aggregate amount of company debt that represents indebtedness for borrowed money outstanding and owing to such lender as of the closing date and an agreement that, if such aggregate amount is paid to such lender on the closing date of the merger, such indebtedness will be repaid in full and that all related liens will be released; |
• | Invitae having received from ArcherDX evidence that any other outstanding liens, any related UCC filings and any related filings with the USPTO Assignment Division have been terminated; |
• | Invitae having received from ArcherDX’s outside legal counsel and any financial advisor or any other person who is entitled to compensation from ArcherDX in connection with services provided with respect to the merger agreement, written statements setting forth the total amount of unpaid expenses that remain payable with respect to services rendered through the closing date of the merger; |
• | ArcherDX having delivered copies of certain third-party notices and consents to Invitae; |
• | ArcherDX having obtained stockholder approval or disapproval (and therefore will not pay) of any payments and/or benefits that may constitute “parachute payments” under Section 280G of the Code; |
• | the Form S-4 Registration Statement having become declared effective under the Securities Act of 1933, and not being the subject of any stop order suspending the effectiveness of the Form S-4 Registration Statement; |
• | ArcherDX having obtained stockholder approval by the requisite stockholders and Holders of at least 75% of the outstanding shares of ArcherDX capital stock; and |
• | the shares of Invitae common stock to be issued in connection with the merger having been approved for the listing by the NYSE. |
• | the representations and warranties of Invitae set forth in the merger agreement, without giving effect to qualifications with respect to any materiality, material adverse or similar qualifiers, shall be accurate at and as of the closing date of the merger (or, in the case of representations and warranties made as of a specific date, as of such date), and where the failure of the representations and warranties of Invitae contained in the merger agreement to be so accurate, individually or in the aggregate, has not had a material adverse effect on Invitae; |
• | Invitae having performed or complied in all material respects with its obligations under the merger agreement that are required to be performed or complied with on or prior to the closing of the merger; |
• | the non-occurrence of a material adverse effect on Invitae being true and correct; |
• | no governmental authority of competent jurisdiction having issued or entered any order or promulgated or enacted any law after the date of the merger agreement, in each case that is in effect, having the effect of enjoining, preventing or otherwise prohibiting the completion of the merger; |
• | any applicable waiting period (and any extension thereof) under the HSR Act relating to the completion of the merger having expired or early termination thereof having been granted; |
• | the Form S-4 Registration Statement having become declared effective under the Securities Act of 1933, and not being the subject of any stop order suspending the effectiveness of the Form S-4 Registration Statement; |
• | the appointment of Jason Myers to the Invitae Board, with effect as of the completion of the merger; |
• | the shares of Invitae common stock to be issued in connection with the merger having been approved for listing by the NYSE; and |
• | ArcherDX having received a certificate certifying that certain of the above conditions have been satisfied from the Chief Executive Officer, the Chief Financial Officer or the General Counsel of Invitae. |
• | by the mutual written consent of Invitae and ArcherDX; |
• | by either Invitae or ArcherDX: |
○ | upon written notice to the other party, if the merger has not been completed on or before March 20, 2021, which date may be extended from time to time by mutual written consent of Invitae and ArcherDX; except where the party seeking to terminate the merger agreement for this reason failed to perform any of its obligations under the merger agreement and such failure to perform was the principal cause of or directly resulted in the failure of the completion of the merger on or before March 20, 2021; |
○ | if any governmental authority of competent jurisdiction has issued or entered any order or enacted any law after the date of the merger agreement that has the effect of restraining, enjoining, preventing, making illegal or otherwise prohibiting the merger, and in the case of such order, such order will have become final and non-appealable, provided that the right to terminate the merger agreement is not available to a party if a material breach by such party of its obligations under the merger agreement has been the principal cause of or principally resulted in the issuance of such order; or |
○ | upon written notice to the other party, if the Invitae stockholder meeting has concluded and the required stockholder approval by Invitae stockholders was not obtained; |
• | by Invitae, if any of the representations or warranties of ArcherDX set forth in the merger agreement are not accurate or if ArcherDX has failed to perform any covenant or agreement on the part of ArcherDX set forth in the merger agreement (including an obligation to consummate the closing) such that the conditions to closing set forth in the merger agreement would not be satisfied and the breach or breaches causing such representations or warranties not to be accurate, or the failures to perform any covenant or agreement, as applicable, are not cured on or prior to the earlier of (i) thirty (30) days after written notice thereof is delivered to ArcherDX and (ii) March 20, 2021; provided that this provision will not be available to Invitae if Invitae is then in material breach of the merger agreement; |
• | by ArcherDX: |
○ | if any of the representations or warranties of Invitae set forth in the merger agreement are not true and correct or if Invitae, Merger Sub A or Merger Sub B have failed to perform any covenant or agreement on the part of Invitae, Merger Sub A or Merger Sub B, as applicable, set forth in the merger agreement (including an obligation to consummate the closing) such that the conditions to closing set forth in the merger agreement would not be satisfied and the breach or breaches causing such representations or warranties not to be accurate, or the failures to perform any covenant or agreement, as applicable, are not cured on or prior to the earlier of (i) thirty (30) days after written notice thereof is delivered to Invitae and (ii) March 20, 2021; provided that this provision will not be available to ArcherDX if ArcherDX is then in material breach of the merger agreement; or |
○ | upon written notice to Invitae, if the Invitae Board has made an adverse recommendation change, as such term is defined in the merger agreement. |
• | each party shall remain liable for its willful breach of the merger agreement prior to termination; |
• | the termination provisions of the merger agreement will remain in effect; |
• | the Holders’ Representative will not be liable to the Holders for any action taken or omitted to be taken in connection with his services pursuant to the merger agreement or any ancillary agreements, except in certain limited cases; |
• | the merger agreement and claims or causes of action arising under the merger agreement will be governed by Delaware law; and |
• | any action relating to the merger agreement or the enforcement of any provision of the merger agreement will be brought exclusively in the Delaware Chancery Court, and to the extent that the Delaware Chancery Court rejects jurisdiction, in any state or federal court located in the Delaware county of New Castle. |
• | any claim for intentional fraud; |
• | any inaccuracy in the allocation schedule; |
• | any appraisal rights exercised by a Holder to the extent not covered by amounts returned to Invitae with respect to such Holder’s dissenting shares pursuant to the merger agreement, and any other action brought by a Holder or any other person claiming rights by, through or associated with such Holder that seeks to challenge any of the transactions based on adequacy of the consideration received by such Holder or claims that any director or officer of ArcherDX breached any fiduciary duty to ArcherDX or any of the Holders in connection with the negotiation, approval, execution or performance of the merger agreement or any of the transactions; |
• | any breach of or failure to perform any covenant or agreement of (A) ArcherDX provided for in the merger agreement with respect to covenants required to be performed before the completion of the merger or (B) the Holders’ Representative; |
• | any eligible post-closing adjustment, which is the amount equal to the final upfront cash consideration less the upfront cash consideration; |
• | any pre-closing taxes, but only to the extent that such taxes were not included in the computation of the closing net working capital or company transaction expenses, in each case, as finally determined pursuant to the merger agreement; and |
• | the Natera Litigation. |
• | The merger proposal requires the affirmative vote of holders of a majority of the shares of Invitae common stock represented (in person or by proxy) at the special meeting, assuming a quorum is present. |
• | The securities issuance proposal requires the affirmative vote of holders of a majority of the shares of Invitae common stock represented (in person or by proxy) at the special meeting, assuming a quorum is present. |
• | The adjournment proposal requires the affirmative vote of holders of a majority of the shares of Invitae common stock represented (in person or by proxy) at the special meeting, regardless of whether a quorum is present. |
• | To submit your proxy via the Internet, go to the website listed on your enclosed proxy card. Have your proxy card in hand when you access the website and follow the instructions to vote your shares. |
• | To submit your proxy by telephone, call 1-800-690-6903. Have your proxy card in hand when you call and then follow the instructions to vote your shares. |
• | If you vote via the Internet or by telephone, you must do so no later than 11:59 p.m., Eastern Time, on September 30, 2020. |
• | To submit your proxy by mail, simply mark your proxy card, date and sign it and return it in the postage-paid envelope. If you do not have the postage-paid envelope, please mail your completed proxy card to the following address: Vote Processing, c/o Broadridge, 51 Mercedes Way, Edgewood, NY 11717. |
• | If you vote by mail, you should mail your proxy card early enough so that it is received before the date of the special meeting. |
• | For the merger proposal, if an Invitae stockholder present at the virtual special meeting abstains from voting, or responds by proxy with an “abstain” vote, it will have the same effect as a vote cast “AGAINST” such proposal. If a stockholder is not present at the virtual special meeting and does not respond by proxy, it will have no effect on the vote count for such proposal (assuming a quorum is present). |
• | For the securities issuance proposal, if an Invitae stockholder present at the virtual special meeting abstains from voting, or responds by proxy with an “abstain” vote, it will have the same effect as a vote cast “AGAINST” such proposal. If a stockholder is not present at the virtual special meeting and does not respond by proxy, it will have the no effect on the vote count for such proposal (assuming a quorum is present). |
• | For the adjournment proposal, if an Invitae stockholder present at the virtual special meeting abstains from voting, or responds by proxy with an “abstain” vote, it will have the same effect as a vote cast “AGAINST” such proposal. If a stockholder is not present at the virtual special meeting and does not respond by proxy, it will have no effect on the vote count for such proposal (regardless of whether a quorum is present). |
• | your broker, bank or other nominee may not vote your shares on the merger proposal, which broker non-votes, if any, will have no effect on the vote count for such proposal (assuming a quorum is present); |
• | your broker, bank or other nominee may not vote your shares on the securities issuance proposal, which broker non-votes, if any, will have no effect on the vote count for such proposal (assuming a quorum is present); and |
• | your broker, bank or other nominee may not vote your shares on the adjournment proposal, which broker non-votes, if any, will have no effect on the vote count for such proposal (regardless of whether a quorum is present). |
• | By delivering, before 10:00 A.M. (Pacific Time) on October 1, 2020, to Invitae's Secretary (at Invitae's executive offices at 1400 16th Street, San Francisco, California 94103) a signed written notice of revocation bearing a later date than the proxy, stating that the proxy is revoked; |
• | By submitting a properly completed proxy card relating to the same shares of Invitae common stock with a later date; |
• | By duly submitting a subsequently dated proxy relating to the same shares of Invitae common stock by telephone or via the Internet (i.e., your most recent duly submitted voting instructions will be followed) before 11:59 p.m., Eastern Time, on September 30, 2020; or |
• | By attending the special meeting via live audio webcast and voting such shares during the virtual special meeting as described above, although attendance at the special meeting will not, by itself, revoke a proxy. |
• | the adjournment is for more than 30 days, in which case a notice of the adjourned meeting will be given to each stockholder of record entitled to vote at the meeting; or |
• | if, after the adjournment, a new record date for determination of stockholders entitled to vote is fixed for the adjourned meeting, in which case the Invitae Board will fix as the record date for determining stockholders entitled to notice of such adjourned meeting the same or an earlier date as that fixed for determination of stockholders entitled to vote at the adjourned meeting, and will give notice of the adjourned meeting to each stockholder of record as of such record date. |
Item 1. | The Merger Proposal |
Item 2. | The Securities Issuance Proposal |
Item 3. | The Adjournment Proposal |
• | The historical audited consolidated financial statements of Invitae as of and for the year ended December 31, 2019 included in Invitae’s Annual Report on Form 10-K for the year ended December 31, 2019 filed with the SEC on March 2, 2020; |
• | The historical unaudited consolidated financial statements of Invitae as of and for the six months ended June 30, 2020 included in Invitae’s Quarterly Report on Form 10-Q for the quarter ended June 30, 2020 filed with the SEC on August 4, 2020; |
• | ArcherDX’s audited financial statements and notes for the year ended December 31, 2019 and 2018 included elsewhere in this proxy statement/prospectus; |
• | ArcherDX’s unaudited financial statements and notes thereto for the six months ended June 30, 2020 included elsewhere in this proxy statement/prospectus; |
• | Singular Bio's financial statements and notes thereto for the three months ended March 31, 2019 included as Exhibit 99.2 to Amendment No. 1 to Invitae's Current Report on Form 8-K filed August 28, 2019; and |
• | Jungla's financial statements and notes thereto for the three months ended March 31, 2019 included as Exhibit 99.2 to Amendment No. 1 to Invitae’s Current Report on Form 8-K filed with the SEC on September 27, 2019. |
| | | Invitae Historical | | | ArcherDX Historical | | | Pro Forma Adjustments (Note 4) | | | Notes | | | Pro Forma Combined | |
Assets | | | | | | | | | | | |||||
Current assets: | | | | | | | | | | | |||||
Cash and cash equivalents | | | $168,203 | | | $21,883 | | | $43,807 | | | A, B, C, D | | | $233,893 |
Marketable securities | | | 253,933 | | | — | | | — | | | | | 253,933 | |
Accounts receivable | | | 27,905 | | | 20,828 | | | — | | | | | 48,733 | |
Prepaid expenses and other current assets | | | 21,081 | | | 16,367 | | | — | | | | | 37,448 | |
Total current assets | | | 471,122 | | | 59,078 | | | 43,807 | | | | | 574,007 | |
Property and equipment, net | | | 43,381 | | | 16,591 | | | — | | | | | 59,972 | |
Operating lease assets | | | 38,239 | | | 5,879 | | | — | | | | | 44,118 | |
Restricted cash | | | 6,343 | | | — | | | — | | | | | 6,343 | |
Intangible assets, net | | | 192,644 | | | 583 | | | 1,043,746 | | | E | | | 1,236,973 |
Goodwill | | | 211,225 | | | 4,972 | | | 1,039,357 | | | F | | | 1,255,554 |
Other assets | | | 6,921 | | | 2,428 | | | — | | | | | 9,349 | |
Total assets | | | $969,875 | | | $89,531 | | | $2,126,910 | | | | | $3,186,316 | |
Liabilities and stockholders' equity | | | | | | | | | | | |||||
Current liabilities: | | | | | | | | | | | |||||
Accounts payable | | | 20,091 | | | 12,898 | | | (4,795) | | | D, G | | | 28,194 |
Accrued liabilities | | | 99,490 | | | 18,117 | | | 1,059 | | | D, H | | | 118,666 |
Operating lease obligations | | | 6,339 | | | 1,382 | | | — | | | | | 7,721 | |
Finance lease obligations | | | 977 | | | — | | | — | | | | | 977 | |
Total current liabilities | | | 126,897 | | | 32,397 | | | (3,736) | | | | | 155,558 | |
Operating lease obligations, net of current portion | | | 42,134 | | | 4,918 | | | — | | | | | 47,052 | |
Finance lease obligations, net of current portion | | | 879 | | | — | | | — | | | | | 879 | |
Convertible senior notes, net | | | 276,092 | | | — | | | | | | | 276,092 | ||
Debt | | | — | | | 41,717 | | | 66,714 | | | B, G | | | 108,431 |
Deferred tax liability | | | 10,250 | | | — | | | 43,862 | | | I | | | 54,112 |
Other long-term liabilities | | | 49,428 | | | 76,784 | | | 650,601 | | | G, H | | | 776,813 |
Total liabilities | | | 505,680 | | | 155,816 | | | 757,441 | | | | | 1,418,937 | |
Contingent preferred stock | | | — | | | 115,347 | | | (115,347) | | | M | | | — |
Stockholders' equity: | | | | | | | | | | | |||||
Common stock | | | 13 | | | 66 | | | (62) | | | A, K, M | | | 17 |
Accumulated other comprehensive loss | | | 572 | | | — | | | — | | | | | 572 | |
Additional paid-in capital | | | 1,487,217 | | | 5,329 | | | 1,200,952 | | | A, J, K, L, M | | | 2,693,498 |
Accumulated deficit | | | (1,023,607) | | | (187,027) | | | 283,927 | | | C, D, I, L, M | | | (926,707) |
Total stockholders' equity | | | 464,195 | | | (181,632) | | | 1,484,817 | | | | | 1,767,380 | |
Total liabilities and stockholders' equity | | | $969,875 | | | $89,531 | | | $2,126,910 | | | | | $3,186,316 |
| | | Invitae Historical | | | ArcherDX Historical | | | Pro Forma Adjustments (Note 4) | | | Notes | | | Pro Forma Combined | |
Revenue: | | | | | | | | | | | |||||
Test revenue | | | $108,177 | | | $— | | | $— | | | | | $108,177 | |
Pharma development services | | | — | | | 15,718 | | | — | | | | | 15,718 | |
Precision oncology products | | | — | | | 15,410 | | | — | | | | | 15,410 | |
Other revenue | | | 2,262 | | | — | | | — | | | | | 2,262 | |
Total revenue | | | 110,439 | | | 31,128 | | | — | | | | | 141,567 | |
Cost of test and other revenue | | | 83,374 | | | — | | | — | | | | | 83,374 | |
Cost of pharma development services | | | — | | | 7,655 | | | 15,772 | | | E, N | | | 23,427 |
Cost of precision oncology products | | | — | | | 5,451 | | | 11,231 | | | E, N | | | 16,682 |
Research and development | | | 130,631 | | | 35,853 | | | 1,212 | | | N | | | 167,696 |
Selling and marketing | | | 81,640 | | | 10,185 | | | 1,307 | | | N | | | 93,132 |
General and administrative | | | 54,660 | | | 57,623 | | | (35,694) | | | D, E, G, N | | | 76,589 |
Loss from operations | | | (239,866) | | | (85,639) | | | 6,172 | | | | | (319,333) | |
Other income (expense), net | | | (16,728) | | | (30,147) | | | 31,992 | | | G | | | (14,883) |
Interest expense, net | | | (10,936) | | | (1,998) | | | (9,057) | | | B, G | | | (21,991) |
Net loss before taxes | | | (267,530) | | | (117,784) | | | 29,107 | | | | | (356,207) | |
Income tax benefit | | | (2,600) | | | — | | | — | | | | | (2,600) | |
Net loss | | | $(264,930) | | | $(117,784) | | | $29,107 | | | | | $(353,607) | |
Net loss per share, basic and diluted | | | $(2.35) | | | $(18.09) | | | | | | | $(2.22) | ||
Shares used in computing net loss per share, basic and diluted | | | 112,765 | | | 6,511 | | | 39,809 | | | M, O | | | 159,085 |
| | | Invitae Historical | | | Singular Bio Pro Forma (Note 5) | | | Jungla Pro Forma (Note 6) | | | Invitae, Singular Bio and Jungla Pro Form Combined | | | ArcherDX Historical | | | Pro Forma Adjustments (Note 4) | | | Notes | | | Pro Forma Combined | |
Revenue: | | | | | | | | | | | | | | | | | ||||||||
Test revenue | | | $212,473 | | | $— | | | $— | | | $212,473 | | | $— | | | $— | | | | | $212,473 | |
Pharma development services | | | — | | | — | | | — | | | — | | | 27,921 | | | | | | | 27,921 | ||
Precision oncology products | | | — | | | — | | | — | | | — | | | 22,644 | | | — | | | | | 22,644 | |
Other revenue | | | 4,351 | | | — | | | — | | | 4,351 | | | — | | | — | | | | | 4,351 | |
Total revenue | | | 216,824 | | | — | | | — | | | 216,824 | | | 50,565 | | | — | | | | | 267,389 | |
Cost of test and other revenue | | | 118,103 | | | — | | | 2,403 | | | 120,506 | | | — | | | — | | | | | 120,506 | |
Cost of pharma development services | | | — | | | — | | | — | | | — | | | 9,212 | | | 30,065 | | | E, N | | | 39,277 |
Cost of precision oncology products | | | — | | | — | | | — | | | — | | | 7,335 | | | 23,939 | | | E, N | | | 31,274 |
Research and development | | | 141,526 | | | (38,238) | | | 358 | | | 103,647 | | | 34,172 | | | 2,424 | | | N | | | 140,243 |
Selling and marketing | | | 122,237 | | | — | | | — | | | 122,237 | | | 15,428 | | | 2,616 | | | N | | | 140,281 |
General and administrative | | | 79,070 | | | (5,435) | | | (2,941) | | | 70,694 | | | 21,643 | | | 15,262 | | | E, G, N | | | 107,599 |
Loss from operations | | | (244,112) | | | 43,673 | | | 180 | | | (200,260) | | | (37,225) | | | (74,307) | | | | | (311,792) | |
Other income (expense), net | | | (3,891) | | | 26 | | | — | | | (3,865) | | | (824) | | | 1,552 | | | G | | | (3,137) |
Interest expense, net | | | (12,412) | | | 3 | | | — | | | (12,409) | | | (2,432) | | | (20,170) | | | B, G | | | (35,011) |
Net loss before taxes | | | (260,415) | | | 43,702 | | | 180 | | | (216,533) | | | (40,481) | | | (92,925) | | | | | (349,940) | |
Income tax expense / (benefit) | | | (18,450) | | | 3,950 | | | 8,750 | | | (5,750) | | | 497 | | | — | | | | | (5,253) | |
Net loss | | | $(241,965) | | | $39,752 | | | $(8,570) | | | $(210,783) | | | $(40,978) | | | $(92,925) | | | | | $(344,687) | |
Net loss per share, basic and diluted | | | $(2.66) | | | | | | | $(2.27) | | | $(6.39) | | | | | | | $(2.48) | ||||
Shares used in computing net loss per share, basic and diluted | | | 90,859 | | | 1,160 | | | 735 | | | 92,754 | | | 6,415 | | | 39,905 | | | M, O | | | 139,074 |
1. | Pending merger of ArcherDX |
| | | Purchase Price | | | Day 1 Expense | | | Post-Combination Expense | | | Total | |
Cash consideration | | | $313.5 | | | $21.1 | | | $— | | | $334.6 |
Liabilities incurred | | | 730.9 | | | — | | | 105.3 | | | 836.2 |
Equity consideration | | | 848.6 | | | 70.6 | | | — | | | 919.2 |
Total | | | $1,893.0 | | | $91.7 | | | $105.3 | | | $2,090.0 |
(in millions) | | | Pro Forma | | | -10% change | | | +10% change |
Price per share | | | $30.96 | | | $27.86 | | | $34.06 |
Purchase price | | | 1,893.0 | | | 1,735.6 | | | 2,050.6 |
Day 1 expense | | | 91.7 | | | 83.9 | | | 99.4 |
Post-close compensation expense | | | 105.3 | | | 94.7 | | | 115.8 |
Total | | | $2,090.0 | | | $1,914.2 | | | $2,265.8 |
$ change vs pro forma | | | n/a | | | (175.9) | | | 175.7 |
% change vs pro forma | | | n/a | | | (8%) | | | 8% |
Cash and cash equivalents | | | $21.8 |
Accounts receivable, net | | | 20.8 |
Other current assets | | | 16.4 |
Non-current assets | | | 24.9 |
Intangible assets | | | 1,044.3 |
Accounts payable | | | (9.8) |
Accrued liabilities | | | (11.8) |
Other short-term liabilities | | | (1.4) |
Deferred revenue | | | (6.3) |
Other long-term liabilities | | | (4.9) |
Deferred tax liability | | | (245.3) |
Estimated total net identifiable assets acquired | | | 848.7 |
Goodwill | | | 1,044.3 |
Estimated total purchase price | | | $1,893.0 |
2. | ArcherDX Accounting Policies Historical Financial Statement Reclassification |
3. | Financing Transactions |
4. | Pro Forma adjustments related to the pending merger of ArcherDX |
A. | To reflect the net cash generated of $263.6 million and related issuance of 16.3 million shares for the pending equity financing. |
B. | To reflect the net cash received and related obligation under the pending debt financing, using an estimated initial borrowing of $135.0 million, and debt discount and issuance costs of $26.6 million, including warrants issued to the lender (see Adjustment J). |
| | | Six months ended June 30, 2020 | | | Year ended December 31, 2019 | |
Principal amount | | | $135.0 | | | $135.0 |
Effective interest rate | | | 10.75% | | | 11.10% |
Interest on principal | | | 7.3 | | | 15.0 |
Amortization of debt issuance costs | | | 3.9 | | | 7.8 |
Total interest expense | | | $11.1 | | | $22.8 |
C. | To reflect the estimated cash to be paid to stockholders of ArcherDX of $334.6 million and the retained earnings impact for the non-recurring post-close compensation expense related to the accelerated vesting of equity awards. |
D. | To adjust the nonrecurring transaction expenses incurred by Invitae and reflect the estimated nonrecurring transaction expenses to be incurred by Invitae through the close of the pending merger. The transaction expenses incurred by Invitae of $3.6 million and by ArcherDX of $0.7 million for the six months ended June 30, 2020 are eliminated in the pro forma condensed combined statement of operations as an adjustment to general and administrative expense and a reclassification in the pro forma condensed combined balance sheet as an elimination from accounts payable for $1.7 million and accrued expenses of $2.5 million and a reduction to cash for $4.2 million. Additionally, the estimated nonrecurring transaction expenses to be incurred by Invitae are recorded as an adjustment to retained earnings and cash for $12.9 million. |
E. | To reflect an adjustment for the intangible assets acquired including in-process research and development (“IPR&D”), and finite life intangible assets (such as developed technology, customer relationships, and trademarks/trade names), valued at $1.0 billion; which as noted above is preliminary and subject to change once the pending merger is completed. Invitae estimates an average 10-year economic life of the finite intangibles based on benchmarking of comparable transactions. The finite life intangible assets are amortized to cost of pharma development services and cost of precision oncology products over the weighted-average useful life of 10-years resulting in amortization of $26.1 million for the six months ended June 30, 2020 and $52.2 million for the twelve months ended December 31, 2019. |
F. | To reflect estimated goodwill recognized from the merger, and the elimination of historical ArcherDX goodwill. |
G. | To eliminate ArcherDX’s historical outstanding debt and debt-like items, including (i) the historical loan outstanding, net of issuance costs of $41.7 million, (ii) accounts payable of $3.1 million, and (iii) other long-term liabilities of $76.8 million related to historical warrants issued and historical contingent consideration. |
H. | To reflect the estimated fair value of the contingent consideration of $730.9 million which will be settled with an estimated 26.8 million shares of Invitae common stock and the remaining portion in cash. The payments to be made within 12 months in cash of $3.6 million are classified as current liabilities and recorded in accrued liabilities. The remaining amount is classified as noncurrent and recorded in other long-term liabilities. |
I. | To reflect a net estimated $43.9 million increase in deferred tax liabilities as a result of the merger consisting of $245.3 million of deferred tax liabilities arising from estimated purchase adjustments in connection with the merger offset by an estimated $201.5 million decrease in the valuation allowance on Invitae’s deferred tax assets. Following the merger, ArcherDX and Invitae anticipate filing a consolidated federal return. As a result of the Tax Cuts and Jobs Act, federal net operating losses arising in 2018 and later years can only offset 80% of taxable income. As a result of the expected consolidated federal tax return filings, the deferred tax liabilities assumed in connection with the merger can serve as a source of income to support the realization of the Invitae’s deferred tax assets. In order to estimate the decrease in the valuation allowance on Invitae’s deferred tax assets, Invitae has assumed that only 80% of the federal deferred tax liabilities assumed or $201.5 million tax-effected may serve as a source of income as the majority of the Invitae’s net operating loss (“NOL”) carryover deferred tax assets were generated in 2018 and later years. |
J. | To reflect the warrants issued as part of the pending debt financing to the lender to purchase 1.0 million shares of Invitae common stock. The fair value of the warrants to be issued was estimated by using a Black-Scholes model at $23.5 million. This estimate is subject to change once the warrants are issued, and may differ materially. |
K. | To reflect the estimated issuance of 26.2 million shares of Invitae common stock in connection with the merger. |
L. | To reflect the estimated 3.8 million Invitae stock option awards, with an estimated fair value of $108.4 million based on Invitae’s closing share price as of August 6, 2020 less the estimated exercise price, granted in connection with the merger and the impact to retained earnings for the non-recurring post-close compensation expense related to the accelerated vesting. |
M. | To eliminate ArcherDX’s historical equity including shares outstanding. |
N. | To reflect the estimated post-combination expense related for consideration paid to ArcherDX stock option holders for meeting certain milestones and continuing employment. The estimated post-close expense will have a continuing impact to Invitae for a period longer than 12 months, will be recognized over the estimated period to achieve each milestone, and is allocated to each financial statement line item as follows (in millions): |
| | | Six months ended June 30, 2020 | | | Year ended December 31, 2019 | |
Cost of pharma development revenue | | | $0.5 | | | $1.0 |
Cost of precision oncology revenue | | | 0.4 | | | 0.8 |
Research and development expense | | | 1.2 | | | 2.4 |
Sales & marketing expense | | | 1.3 | | | 2.6 |
General & administrative expense | | | 11.0 | | | 22.0 |
Total | | | $14.4 | | | $28.8 |
O. | To reflect the additional 26.2 million shares of Invitae common stock to be issued and 3.8 million fully vested Invitae stock option awards granted as part of the merger and 16.3 million shares of Invitae common stock to be issued as part of the pending equity financing. Potentially dilutive securities, |
5. | Acquisition of Singular Bio and Pro Forma Adjustments |
| | | Singular Bio Historical (January 1, 2019 to June 19, 2019) | | | Pro Forma Adjustments | | | Note | | | Singular Bio Pro Forma | |
Revenue | | | | | | | | | ||||
Test revenue | | | $— | | | $— | | | | | $— | |
Other revenue | | | — | | | — | | | | | — | |
Total revenue | | | — | | | — | | | | | — | |
Cost of revenue | | | — | | | — | | | | | — | |
Research and development | | | 866 | | | (39,104) | | | B | | | (38,238) |
Selling and marketing | | | — | | | — | | | | | — | |
General and administrative | | | 1,752 | | | (7,187) | | | A, C, D | | | (5,435) |
Loss from operations | | | (2,618) | | | 46,291 | | | | | 43,673 | |
Other income (expense), net | | | 2,051 | | | (2,025) | | | C | | | 26 |
Interest expense | | | (744) | | | 777 | | | E | | | 3 |
Net loss before taxes | | | (1,341) | | | 45,043 | | | | | 43,702 | |
Income tax benefit | | | — | | | 3,950 | | | F | | | 3,950 |
Net loss | | | $(1,341) | | | $41,093 | | | | | $39,752 | |
Net loss per share, basic and diluted | | | | | | | | | ||||
Shares used in computing net loss per share, basic and diluted | | | | | 1,160 | | | G | | | 1,160 |
A. | To eliminate the nonrecurring post-combination expense of $3.2 million recorded due to the acceleration of unvested equity. |
B. | To remove the $39.1 million of stock-based compensation related to the time-based restricted stock units (“RSUs”) and performance-based RSUs granted in connection with the Singular Bio acquisition. |
C. | To eliminate the effects of the $3.0 million co-development agreement between Invitae and Singular Bio. Invitae recognized expense related to the co-development agreement from September 2018 through June 2019 through general and administrative expense with $2.0 million recognized during the six months ended June 30, 2019. This amount occurred prior to the close of the Singular Bio acquisition and therefore is added back to net income (loss). Singular Bio recognized other income (expense) related to this co-development agreement of $2.0 million during the six months ended June 30, 2019. As the other income related to this agreement would not have been recognized during this period had the acquisition closed on January 1, 2019 this adjustment reverses the other income recognized. |
D. | To eliminate $2.0 million of nonrecurring transaction costs incurred by both Invitae and Singular Bio that were incurred during the year ended December 31, 2019 and were directly related to the acquisition of Singular Bio. |
E. | To remove interest expenses related to the extinguishment of debt and debt-like items incurred by Singular Bio of $0.8 million during the year ended December 31, 2019. This debt was not assumed by Invitae in connection with the acquisition. |
F. | To remove the income tax benefit recognized due to net deferred tax liabilities assumed in connection with the acquisition of Singular Bio. |
G. | To reflect the 1.2 million adjustment to the weighted average of shares outstanding of Invitae common stock issued in connection with the acquisition of Singular Bio. |
6. | Acquisition of Jungla and Pro Forma Adjustments |
| | | Jungla Historical (January 1, 2019 to July 16, 2019) | | | Pro Forma Adjustments | | | Note | | | Jungla Pro Forma | |
Revenue | | | | | | | | | ||||
Test revenue | | | $— | | | $— | | | | | $— | |
Other revenue | | | — | | | — | | | | | — | |
Total revenue | | | — | | | — | | | | | — | |
Cost of revenue | | | — | | | 2,403 | | | C | | | 2,403 |
Research and development | | | 358 | | | — | | | | | 358 | |
Selling and marketing | | | — | | | — | | | | | — | |
General and administrative | | | 868 | | | (3,809) | | | A, B | | | (2,941) |
Loss from operations | | | (1,226) | | | 1,406 | | | | | 180 | |
Other income (expense), net | | | — | | | — | | | | | — | |
Interest expense | | | — | | | — | | | | | — | |
Net loss before taxes | | | (1,226) | | | 1,406 | | | | | 180 | |
Income tax benefit | | | — | | | 8,750 | | | D | | | 8,750 |
Net loss | | | $(1,226) | | | $(7,344) | | | | | $(8,570) | |
Net loss per share, basic and diluted | | | | | | | | | ||||
Shares used in computing net loss per share, basic and diluted | | | | | 735 | | | E | | | 735 |
A. | To eliminate the $2.9 million post-combination compensation expense recognized due to the acceleration of Jungla’s equity awards. |
B. | To eliminate $0.8 million of non-recurring transaction costs incurred by both Invitae and Jungla during the year ended December 31, 2019 and were directly related to the acquisition of Jungla. |
C. | To reflect the additional amortization of the acquired developed technology of $2.4 million for the year ended December 31, 2019. As the primary use of the developed technology is to generate revenue, the amortization is presented as cost of revenue. |
D. | To remove the income tax benefit recognized due to net deferred tax liabilities assumed in connection with the acquisition of Jungla. |
E. | To reflect the 0.7 million adjustment to the weighted-average of shares outstanding of Invitae common stock issued in connection with the acquisition of Jungla. |
• | Accuracy: Although current tests can accurately detect common or simple mutations, most cannot accurately detect complex mutations, resulting in a high rate of false negatives and positives. These tests are designed with inherent biases which fundamentally limit their ability to detect both known and unknown alterations. ArcherDX believes its purpose built AMP chemistry addresses these shortcomings by accurately detecting complex, as well as known and unknown, mutations. This allows ArcherDX to provide actionable information to clinicians, enabling them to select therapies targeted to the appropriate mutation, and thereby better informing treatment. |
• | Utility: Current tests generally take a “one-size-fits-all” approach, which limits their utility. Because ArcherDX’s products are not limited to blood and tissue, they enable testing across a multitude of sample types, targeting DNA, RNA, and ctDNA in order to provide higher resolution and a more comprehensive view of mutations. ArcherDX’s product development platform enables rapid and efficient customization of biomarker targets, which means clinicians can specify which of its products they want to use to identify mutations that they believe are important in treating each patient’s disease. Together, ArcherDX believes these features open additional areas of clinical application within therapy optimization for individual patients and cancer monitoring of early-stage cancer. |
• | Turnaround time: Results from centralized genomic tests can take well over 20 days to be returned, depending on complexity and sample type. As late-stage cancer patients are likely to experience superior survival rates and outcomes when treated as soon as possible after diagnosis, lengthy turnaround times can prevent timely treatment. ArcherDX’s products enable local testing with potential times to run the test in a matter of days, accelerating time to results while also allowing the original clinician to maintain sample custody. The reduction in time to actionable results accelerates clinical decisions, which is critical for cancer patients where immediate treatment is important to achieve optimal outcomes. |
• | Economics: The complexity of many genomic testing options requires significant resources and sophisticated infrastructure, which are typically limited to specialized academic or centralized facilities. As a result, hospitals often need to outsource their genomic testing, which incurs costs that are not reimbursed, presenting an additional hurdle to adoption. This particularly affects community and regional hospitals where approximately 85% of cancer patients are treated. Centralized labs that perform genomic testing receive the benefits of reimbursement and often retain possession of patients’ tumor biopsies and control patient data. ArcherDX believes its platform will empower more hospitals and clinics to practice precision oncology and receive reimbursement for in-house testing that they would otherwise not receive if they send the testing out to centralized labs. In addition, clinicians can retain possession of patients’ tumor biopsies and control the data, which can be matched with clinical data to generate revenue from collaborators, such as biopharmaceutical companies. ArcherDX believes this empowers providers in all patient care settings to participate in the economics of precision oncology testing. |
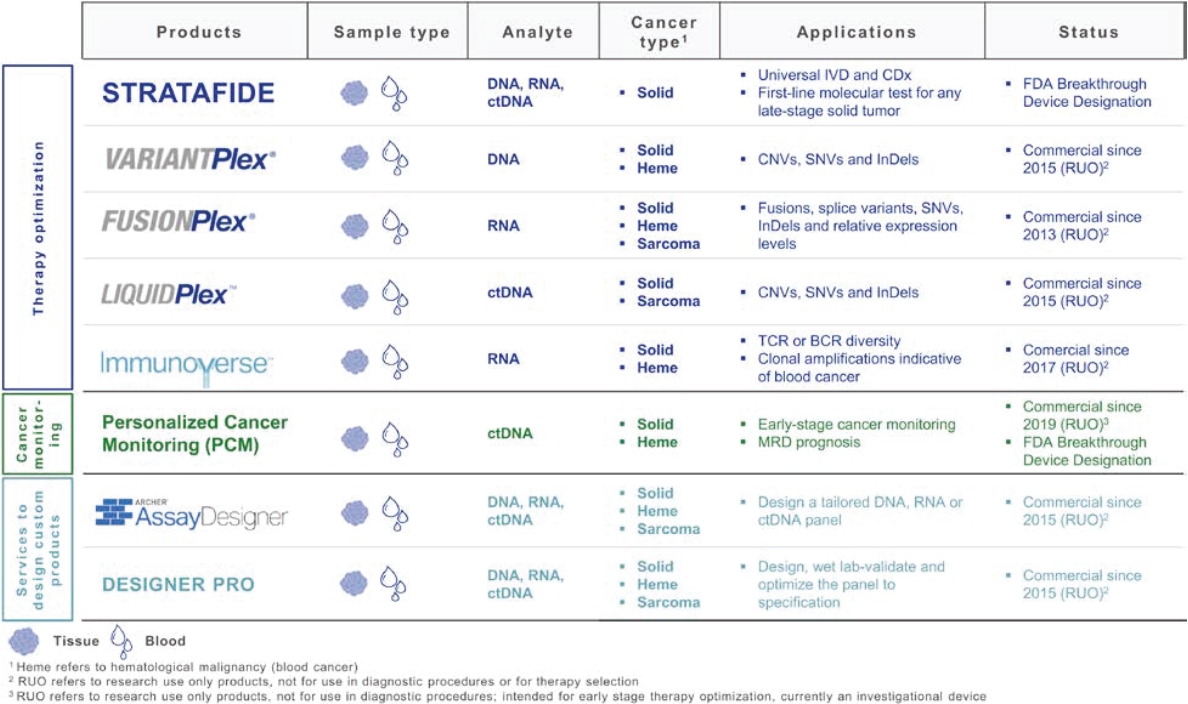
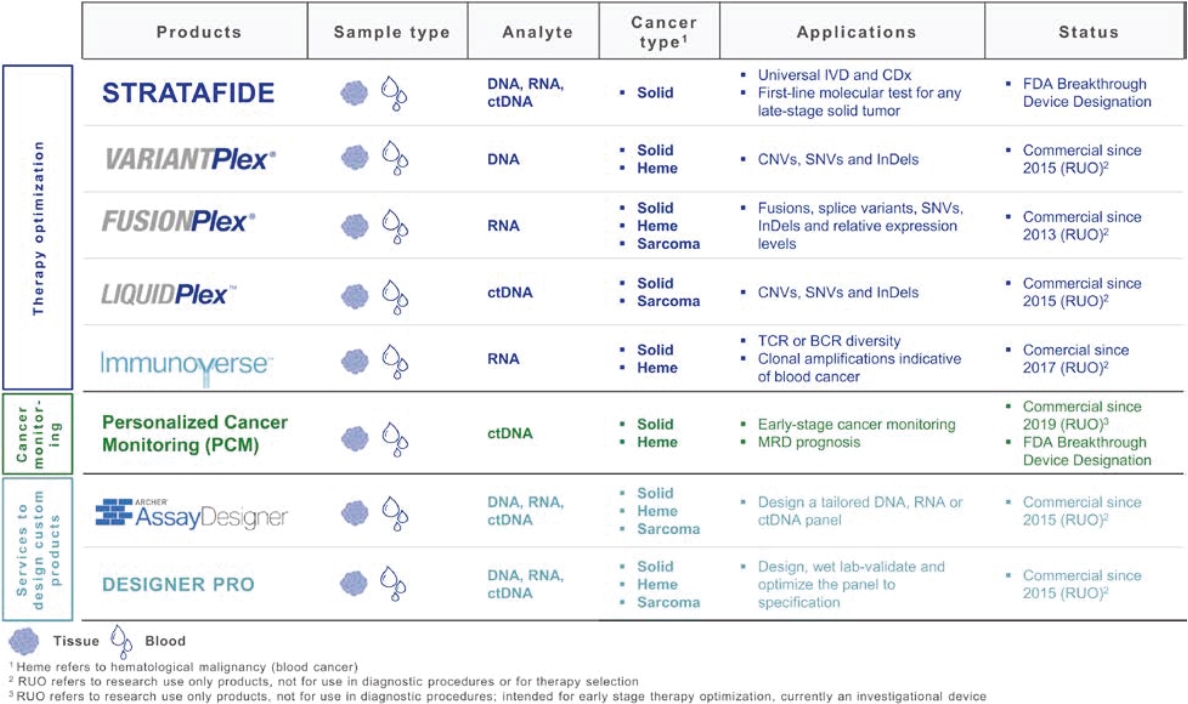
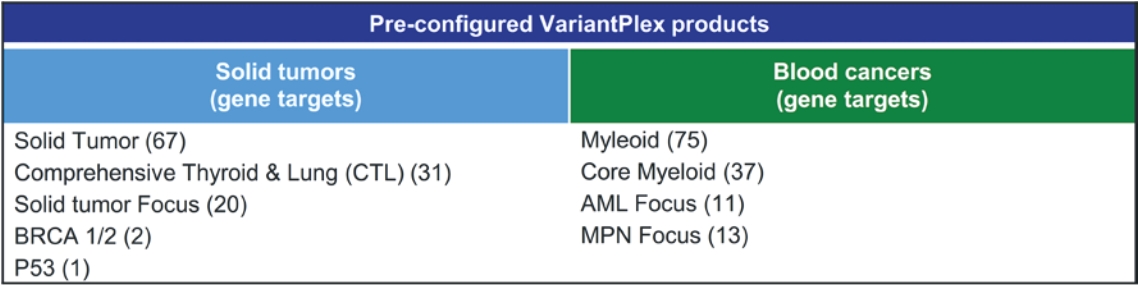
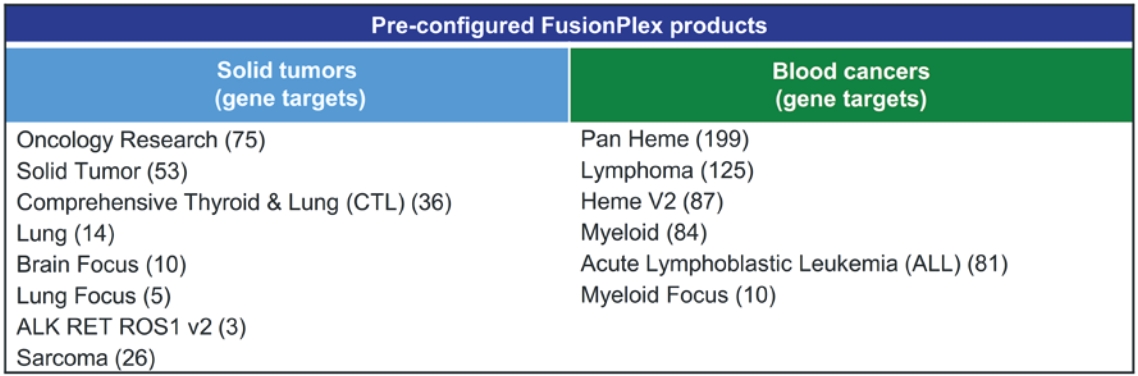

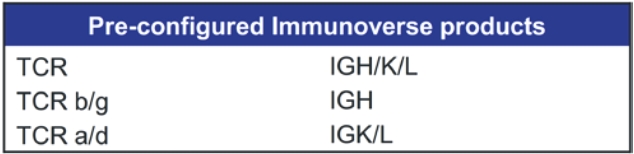
• | a custom VariantPlex product for MRD detection in pediatric patients with acute myeloid leukemia; |
• | a custom FusionPlex product for real-time sequencing and rapid identification of fusions in leukemias using rapid NGS technology; and |
• | a custom LiquidPlex product for tumor virus genome detection to track tumor burden and study viral oncogenicity and antiviral drug response. |
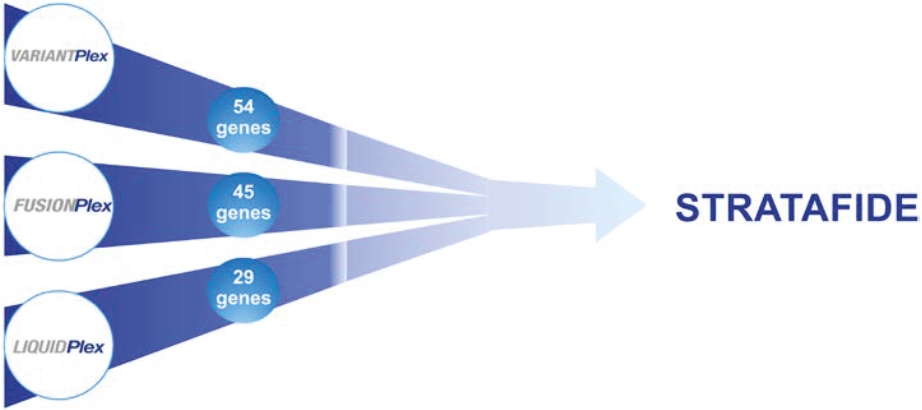
• | Measure disease progression (Recurrence Monitoring of Residual Disease) — ArcherDX can detect recurrence before it is symptomatic, allowing earlier intervention than standard of care. |
• | Measure therapy effectiveness (Therapy Response Monitoring) — Cancers are complex and contain multiple cell types. ArcherDX believes that longitudinal monitoring with PCM can identify if a therapy is truly effective or ineffective against all cell types in a person’s tumor. |
• | Determine treatment regimen (Therapy Optimization) — Patients who fail to respond to standard care are high-risk. ArcherDX believes PCM can identify previously undetectable high-risk patients in the adjuvant cancer setting earlier to bring therapies currently approved for late-stage patients into the early-stage setting, when the cancer is most curable. |
• | Refine therapy (Therapy Modulation) — ArcherDX believes a future application of PCM could be to identify patients cured by surgical resection of their cancer, thereby preventing unnecessary and potentially harmful adjuvant therapy, which could further limit long-term adverse events and healthcare costs. |
• | Accelerate clinical trial endpoints (Novel Surrogate Clinical Trial Endpoints) — ArcherDX believes PCM could potentially reduce the cost and length of trials and accelerate drug development, which can bring life-saving therapies to market faster and allow biopharmaceutical companies to complete more trials in the same finite time period. |
(1) | At initial diagnosis, exome sequencing of the surgically removed tumor or a tumor biopsy is used to identify the patient-specific cancer mutations. |
(2) | Data from the exome sequencing is analyzed with proprietary algorithms to select the patient-specific cancer mutations most likely to yield the greatest sensitivity for recurrence of the patient’s cancer. |
(3) | An enhanced version of Assay Designer is used to create a personalized LiquidPlex product, which is used to longitudinally track ctDNA and measure cancer burden. |
(4) | The personalized product is delivered to the laboratory affiliated with the patient’s care team. |
(5) | The clinicians use the personalized product to assess ctDNA taken from non-invasive peripheral blood draws at specified intervals, yielding a quantitative longitudinal view of the cancer’s evolution. |
• | High sensitivity and specificity — Archer AMP chemistry has demonstrated high sensitivity and specificity of simple and complex mutations compared to other methods. |
• | Chemistry modularity — Archer AMP chemistry provides ArcherDX with the ability to mix and match different sets of biomarker content without sacrificing performance. |
• | Known and unknown mutation detection — The accuracy of the Archer AMP chemistry has identified unknown RNA fusions for the very first time, which were unable to be detected given a lack of sensitivity in other testing methods. |
• | Improved clinical utility — Archer AMP chemistry has been proven to demonstrate improved clinical utility. For example, clinicians at Memorial Sloan Kettering profiled over 2,500 lung cancers using the MSK-IMPACT test. FusionPlex identified previously undetected, actionable mutations in 33 patients. Of these 33 patients, 10 received a matched targeted therapy, and 8 of these 10 achieved clinical benefit. |
• | Summary of study results: ArcherDX’s products provide highly sensitive (true positive) results, correctly identifying individuals with a genetic mutation, and highly specific (true negative) results, correctly identifying individuals without a genetic mutation across institutions. |
• | Summary of data: In a subset of 219 patient samples, multiple institutions achieved a sensitivity of 100% and specificity of 100% using ArcherDX’s FusionPlex product. In addition, investigators at Virginia Commonwealth University, Duke University and Genosity’s CAP-accredited laboratory also achieved sensitivity and specificity of 100% in over 515 patient samples with ArcherDX’s VariantPlex product. Finally, investigators at Massachusetts General Hospital interrogated 110 mutations using ArcherDX’s LiquidPlex product to identify mutations in solid tumors from blood samples with 90% sensitivity and 100% specificity. |


• | Summary of study result: Clinical validation of AMP as compared to fluorescent in situ hybridization, or FISH. The first validation of AMP demonstrated superior performance with respect to clinical sensitivity and specificity when compared to standard clinical FISH assays. The study also demonstrated AMP can reduce tissue sample quantity needed while still gathering high quality results. |
• | Summary of data: An AMP gene rearrangement panel using 319 FFPE samples showed 100% sensitivity (95% confidence limit: 96.5-100%) and 100% specificity (95% confidence limit: 99.3-100%) compared with reference methods. Based on performing AMP on 986 clinical FFPE samples, the study demonstrated AMP’s potential as both a robust clinical assay and a powerful discovery tool, which was used to identify new therapeutically important gene fusions: ARHGEF2-NTRK1 and CHTOP-NTRK1 in glioblastoma, MSN-ROS1, TRIM4-BRAF, VAMP2-NRG1, TPM3-NTRK1 and RUFY2-RET in lung cancer, FGFR2-CREB5 in cholangiocarcinoma and PPL-NTRK1 in thyroid carcinoma. |
• | Archer implication: Accuracy and Utility — AMP is a scalable and efficient next-generation sequencing target enrichment method for research and clinical applications. ArcherDX’s AMP chemistry is compatible with low nucleic acid input from FFPE specimens and is effective in detecting gene rearrangements (without prior knowledge of the fusion partners), single nucleotide variants, insertions, deletions and copy number changes. |
• | Summary of study result: Archer FusionPlex-based test is superior to FISH and PCR in identifying complex mutations and can also identify previously unknown fusion partners. |
• | Summary of data: ArcherDX’s AMP chemistry was used in a 26-gene FusionPlex Sarcoma panel and was evaluated and compared with FISH and reverse transcriptase-PCR (RT-PCR). Eighty-one samples were subjected to AMP-based targeted NGS, and 86% (n = 70) were successfully conducted and were either fusion positive (n = 48) or fusion negative but met all criteria for good quality (n = 22). A concordance of 90% was found between NGS and conventional techniques. AMP-based targeted NGS showed superior results compared to FISH and RT-PCR, which showed four cases that were false negative. The Archer assay also revealed COL1A1 and SEC31A as novel fusion partners for USP6 in nodular fasciitis, whereas FISH and RT-PCR did not detect these fusion partners. |
• | Archer implication: Accuracy and Utility —ArcherDX’s platform can potentially unlock new clinical applications demonstrated by the validation of ArcherDX’s AMP chemistry’s ability to detect complex mutations in RNA not possible with other technologies. This study provides clinical validation of AMP and demonstrates its superior utility compared to FISH and RT-PCR. |
• | Summary of study result: Large DNA-based sequencing tests lack adequate sensitivity for detecting all treatable cancer-related mutations, resulting in patients receiving non-specific, toxic chemotherapy. This study led to a new NCCN recommendation for concurrent RNA-based testing. |
• | Summary of data: As part of the prospective clinical genomic testing, clinicians at Memorial Sloan Kettering profiled 2,522 lung adenocarcinomas using the MSK-IMPACT test panel. This prospective clinical testing identified 195 (7.7%) fusions and 119 (4.7%) METex14 alterations. FusionPlex identified previously undetected mutations in 14% (36/254) of cases, 33 of which were actionable. Of these 33 patients, 10 then received matched targeted therapy, which achieved clinical benefit in 8 cases (80%). |
• | Archer implication: Utility and Flexibility —ArcherDX’s RNA-based FusionPlex product line can identify previously undetected mutations, providing the possibility of matching more patients with targeted therapy, thereby reducing the consequences of outdated, non-specific chemotherapy and toxic regimens. |
• | Summary of study result: Clinical validation of a large, custom FusionPlex test interrogating 106 cancer genes involving nearly 600 different fusions reported in hematological malignancies and solid tumors. This test identified fusions with 100% sensitivity and specificity, regardless of sample type. |
• | Summary of data: Gene fusions are one of the most common genomic alterations in pediatric cancer. Many fusions encode oncogenic drivers and play important roles in cancer diagnosis, risk stratification, and treatment selection. In the validation for this study, the test demonstrated perfect accuracy, with 100% sensitivity and 100% specificity on 60 pediatric tumor samples. In addition to identifying all known fusions in the validation samples, three previously unrecognized, yet clinically significant, fusions were also detected. Following assay validation, 276 additional cases were analyzed, identifying 51 different fusions. Of these, 16 were not previously identified and provided genomic information useful for clinical management. |
• | Archer implication: Accuracy and Utility —This study demonstrates that ArcherDX’s AMP chemistry enables custom FusionPlex products with many biomarker targets without sacrificing assay performance. In addition, FusionPlex products are able to detect the vast majority of known and previously unknown clinically-relevant fusions in pediatric cancers accurately and efficiently. |
• | Summary of study result: Clinical validation of a 28-gene Archer LiquidPlex cell-free DNA panel that targets the most common genetic alterations in solid tumors that demonstrated 100% specificity and over 89% sensitivity in identifying solid tumor driver mutations. |
• | Summary of data: The use of liquid biopsies to identify driver mutations in patients with solid tumors holds great promise for performing targeted therapy selection, monitoring disease progression, and detecting treatment resistance mechanisms. In this study, the panel and analytical tools developed were used to analyze commercially available controls, allowing establishment of a limit of detection allele fraction cutoff of 0.25%, with 100% (95% CI, 81.5%-100%) specificity and 89.8% (95% CI, 81.0%-94.9%) sensitivity. In addition, a total of 163 blood samples were analyzed from patients with metastatic cancer (n = 123) and demonstrated a >90% sensitivity for detecting previously identified expected mutations. Longitudinal monitoring of patients revealed a strong correlation of variant allele frequency changes and clinical outcome. Additional clinically relevant information included identification of resistance mutations in patients receiving targeted treatment and detection of complex patterns of mutational heterogeneity. |
• | Archer implication: Accuracy and Utility —Although tissue is the gold standard sample type, it may be difficult to obtain and not every patient is healthy enough to undergo an invasive biopsy. This study demonstrates the ability of ArcherDX’s LiquidPlex product to identify solid tumor driver mutations via liquid biopsy filling a critical need for patients without a tissue biopsy available. The high sensitivity and specificity and the correlation of variant allele frequency changes and clinical outcome strongly support clinical implementation of cell-free DNA panels in advanced cancer patients. ArcherDX designed the ctDNA LiquidPlex technology to enable efficient target capture of and variant detection in ctDNA from liquid biopsies. ArcherDX’s LiquidPlex technology uses AMP-based target enrichment to preferentially enrich for highly fragmented ctDNA over genomic DNA to reduce background noise and increase detection sensitivity of low-allelic fraction variants. |
• | Summary of study result: Clinical validation of a custom Archer panel to identify blood cancers by detecting MRD in patients with AML in remission. |
• | Summary of data: Patients with AML in remission remain at risk for relapse even after allogeneic hematopoietic cell transplantation. Ultra-deep, error-corrected sequencing for 13 commonly mutated genes in AML was performed on preconditioning blood from patients treated in a phase III clinical trial that randomly assigned adult patients with myeloid malignancy in morphologic complete remission to myeloablative conditioning, or MAC, or reduced-intensity conditioning, or RIC. This study provided evidence that MAC rather than RIC in patients with AML with genomic evidence of MRD before alloHCT can result in improved survival. |
• | Archer implication: Utility —This study underscores the power of the Archer platform to provide accurate genomic alteration detection regardless of sample sources (solid tumor or hematological malignancies), addressing the current limitations of “one-size-fits-all” approaches, and establishing the foundation for expansion of therapy optimization and cancer monitoring into blood cancers. |
• | Summary of study result: FusionPlex used to identify a novel fusion in a lung-cancer patient that responded to a MET-specific drug. |
• | Summary of data: The study describes the first case of a MET fusion in lung cancer identified and treated during course of clinical care, and the dramatic response of the patient’s tumor to crizotinib treatment. The authors utilized FusionPlex to detect a previously undescribed fusion of HLA-DRB1 exon 5 to MET exon 15, and this fusion was predicted to be in-frame and preserve the MET kinase domain. The finding was confirmed on a separate extraction and repeat analysis from the resection sample. The patient began treatment with off-label oral crizotinib 250mg twice daily. After 6 weeks of therapy, repeat CT showed complete resolution of the previously observed left upper lobe perihilar and right lower lobe lung nodules with no new lesions. This robust response remains as of 8 months on therapy. The recent rapid expansion in the number of characterized driver oncogenes, the increase in the number of approved therapies, and the large number of clinical studies now available testing drugs that target these novel oncogenes, necessitates evaluating multiple genes simultaneously. NGS-based assays are well suited to meet this requirement due to the ability to sequence in massively parallel fashion. This study employed AMP, an NGS-based assay designed specifically for gene fusion detection. The ability of this assay to detect fusions without prior knowledge of the fusion partner was critical in this case, as fusion of MET to HLA-DRB1 has not been reported previously. |
• | Archer implication: Accuracy and Utility —The accuracy of FusionPlex allows for the detection of novel fusions that expand the druggable targets within an indication. This study unlocks new applications for targeted therapies used by biopharmaceutical companies leading to more use in clinical trials and expanded clinical utility. |
• | Summary of study result: Utilizing ArcherDX’s AMP technology, ctDNA monitoring for MRD can detect relapse of NSCLC earlier than standard of care imaging surveillance in some instances. AMP personalized cell-free DNA, or cfDNA, enrichment can accurately detect low-frequency variant DNA at low assay DNA inputs consistent with an MRD setting. |
• | Summary of data: Post-operative timepoints were analyzed from 90 TRACERx patients. In patients whose cancer had relapsed and shed ctDNA, the ctDNA was detected at or before relapse with a median lead-time, or time from ctDNA detection to clinical relapse, of 164 days (range: 6 to 1,022 days) in the TRACERx study tracking a median of 200 variants per patient. Furthermore, in non-relapse patients, the assay demonstrated 99.3% clinical specificity within the research data set. Results from the analytical validation of a 50-variant version of the research assay demonstrated 100% specificity with detection down to 0.003% variant fractions at high cfDNA input levels. |
• | Archer implication: Accuracy and Utility — The study provides further evidence that ArcherDX’s AMP technology has the ability to potentially accurately detect exceedingly low levels of cancer-derived DNA from patient blood which is important for successful PCM. Furthermore, with more sensitive detection of ctDNA for MRD as a biomarker, it is possible for adjuvant clinical trials to be conducted in smaller and more relevant settings by only escalating therapy in patients who are set to relapse, thereby potentially reducing trial size, cost and time. |
• | the device may not be shown safe or effective to the FDA’s satisfaction; |
• | the data from pre-clinical studies and/or clinical trials may be found unreliable or insufficient to support approval; |
• | the manufacturing process or facilities may not meet applicable requirements; and |
• | changes in FDA clearance or approval policies or adoption of new regulations may require additional data. |
• | establishment registration and device listing with the FDA; |
• | QSR requirements, which require manufacturers, including third-party manufacturers, to follow stringent design, testing, control, documentation and other quality assurance procedures during all aspects of the design and manufacturing process; |
• | labeling regulations and FDA prohibitions against the promotion of investigational products, or the promotion of “off-label” uses of cleared or approved products; |
• | requirements related to promotional activities; |
• | clearance or approval of product modifications to 510(k)-cleared devices that could significantly affect safety or effectiveness or that would constitute a major change in intended use of one of ArcherDX’s cleared devices, or approval of certain modifications to PMA-approved devices; |
• | medical device reporting regulations, which require that a manufacturer report to the FDA if a device it markets may have caused or contributed to a death or serious injury, or has malfunctioned and the device or a similar device that it markets would be likely to cause or contribute to a death or serious injury, if the malfunction were to recur; |
• | correction, removal and recall reporting regulations, which require that manufacturers report to the FDA field corrections and product recalls or removals if undertaken to reduce a risk to health posed by the device or to remedy a violation of the FDCA that may present a risk to health; |
• | the FDA’s recall authority, whereby the agency can order device manufacturers to recall from the market a product that is in violation of governing laws and regulations; and |
• | post-market surveillance activities and regulations, which apply when deemed by the FDA to be necessary to protect the public health or to provide additional safety and effectiveness data for the device. |
• | issuance of warning letters, untitled letters, fines, injunctions, consent decrees and civil penalties; |
• | requesting or requiring recalls, withdrawals, or administrative detention or seizure of ArcherDX’s products; |
• | imposing operating restrictions or partial suspension or total shutdown of production; |
• | refusing or delaying requests for 510(k) marketing clearance or PMA approvals of new products or modified products; |
• | withdrawing 510(k) clearances or PMA approvals that have already been granted; |
• | refusal to grant export approvals for ArcherDX’s products; or |
• | criminal prosecution. |
| | | Three Months Ended June 30, | | | Change | |||||||
(in thousands, except percentages) | | | 2019 | | | 2020 | | | $ | | | % |
Revenue | | | | | | | | | ||||
Precision oncology products | | | $5,095 | | | $8,404 | | | $3,309 | | | 65% |
Pharma development services | | | 5,704 | | | 7,934 | | | 2,230 | | | 39% |
Total revenue | | | 10,799 | | | 16,338 | | | 5,539 | | | 51% |
Costs & operating expenses | | | | | | | | | ||||
Cost of precision oncology products | | | 1,525 | | | 3,138 | | | 1,613 | | | 106% |
Cost of pharma development services | | | 1,740 | | | 4,256 | | | 2,516 | | | 145% |
Sales and marketing | | | 2,900 | | | 4,861 | | | 1,961 | | | 68% |
Research and development | | | 5,006 | | | 22,116 | | | 17,110 | | | 342% |
General and administrative | | | 3,332 | | | 11,037 | | | 7,705 | | | 231% |
Contingent consideration | | | 2,790 | | | 39,140 | | | 36,350 | | | 1,303% |
Total operating expenses | | | 17,293 | | | 84,548 | | | 67,255 | | | 389% |
Loss from operations | | | (6,494) | | | (68,210) | | | (61,716) | | | 950% |
Interest expense, net | | | (769) | | | (1,105) | | | (336) | | | 44% |
Other expense, net | | | (211) | | | (29,152) | | | (28,941) | | | * |
Loss before income taxes | | | (7,474) | | | (98,467) | | | (90,993) | | | 1,217% |
Income tax expense | | | 6 | | | — | | | (6) | | | * |
Net loss | | | (7,480) | | | (98,467) | | | (90,987) | | | 1,216% |
* | Not meaningful |
| | | Six Months Ended June 30, | | | Change | |||||||
(in thousands, except percentages) | | | 2019 | | | 2020 | | | $ | | | % |
Revenue | | | | | | | | | ||||
Precision oncology products | | | $9,475 | | | $15,410 | | | $5,935 | | | 63% |
Pharma development services | | | 10,773 | | | 15,718 | | | 4,945 | | | 46% |
Total revenue | | | 20,248 | | | 31,128 | | | 10,880 | | | 54% |
Costs & operating expenses | | | | | | | | | ||||
Cost of precision oncology products | | | 2,593 | | | 5,451 | | | 2,858 | | | 110% |
Cost of pharma development services | | | 3,446 | | | 7,655 | | | 4,209 | | | 122% |
Sales and marketing | | | 5,544 | | | 10,185 | | | 4,641 | | | 84% |
Research and development | | | 9,301 | | | 35,853 | | | 26,552 | | | 285% |
General and administrative | | | 5,709 | | | 18,518 | | | 12,809 | | | 224% |
Contingent consideration | | | 5,506 | | | 39,105 | | | 33,599 | | | 610% |
Total operating expenses | | | 32,099 | | | 116,767 | | | 84,668 | | | 264% |
Loss from operations | | | (11,851) | | | (85,639) | | | (73,788) | | | 623% |
Interest expense, net | | | (769) | | | (1,998) | | | (1,229) | | | 160% |
Other expense, net | | | (219) | | | (30,147) | | | (29,928) | | | * |
Loss before income taxes | | | (12,839) | | | (117,784) | | | (104,945) | | | 817% |
Income tax expense | | | 7 | | | — | | | (7) | | | * |
Net loss | | | (12,846) | | | (117,784) | | | (104,938) | | | 817% |
* | Not meaningful |
| | | Six Months Ended June 30, | ||||
| | | 2019 | | | 2020 | |
Cash used in operating activities | | | $(10,606) | | | $(45,699) |
Cash used in investing activities | | | (1,349) | | | (7,254) |
Cash provided by financing activities | | | 42,266 | | | 15,344 |
| | | Payments due by period | |||||||||||||
| | | Total | | | Less than 1 year | | | 1-3 years | | | 3-5 years | | | More than 5 years | |
| | | (in thousands) | |||||||||||||
Lease obligations(1) | | | $6,034 | | | $953 | | | $2,421 | | | $2,555 | | | $105 |
Debt obligations(2) | | | 30,000 | | | — | | | — | | | 30,000 | | | — |
Interest obligations(2) | | | 11,092 | | | 3,300 | | | 6,600 | | | 1,192 | | | — |
Royalty obligation(3) | | | 10,229 | | | 1,491 | | | 2,947 | | | 5,791 | | | — |
Purchase obligations(4) | | | — | | | — | | | — | | | — | | | — |
Total | | | $57,355 | | | $5,744 | | | $11,968 | | | $39,538 | | | $105 |
(1) | ArcherDX leases office and laboratory space in St. Louis, Missouri; Boston, Massachusetts; Boulder, Colorado; and Golden, Colorado. Please see Note 8 to ArcherDX’s audited financial statements for additional information pertaining to its lease obligations. |
(2) | Interest obligations is based upon ArcherDX’s debt outstanding on June 30, 2020 of $45.0 million and the then effective interest rate of 11%. Please see Note 9 to its audited financial statements for additional information pertaining debt and interest obligations. |
(3) | Under certain of these agreements, ArcherDX is obligated to pay royalties ranging up to the mid-single digits of sales in which the patents are used in the product or service sold, subject to minimum annual royalties or fees in certain agreements. Please see note 16 to its audited financial statements for additional information pertaining to its royalty obligation. Only minimum royalty payments are listed. |
(4) | ArcherDX has contracts and purchase orders for limited future obligations related to continuing ongoing operations. Substantially all of its contracts and purchase orders maybe be canceled without significant penalty to ArcherDX. |
• | the prices, rights, preferences, and privileges of its Preferred Stock relative to those of its common stock; |
• | lack of marketability of the common stock; |
• | current business conditions and projections; |
• | hiring of key personnel and the experience of management; |
• | its stage of development; |
• | likelihood of achieving a liquidity event, such as an initial public offering, a merger or acquisition of its company given prevailing market conditions, or other liquidation event; |
• | the market performance of comparable publicly traded companies; |
• | the U.S. and global capital market conditions; and |
• | the magnitude of the impact of the COVID-19 pandemic on its productivity, results of operations and financial position, as well as on U.S. and global capital market conditions. |
• | each of ArcherDX’s named executive officers; |
• | each of ArcherDX’s directors; |
• | all of ArcherDX’s directors and executive officers as a group; and |
• | each person or entity known by ArcherDX to own beneficially more than 5% of ArcherDX’s common stock (by number or by voting power). |
Name of Beneficial Owner | | | Number of Shares Beneficially Owned | | | Percentage Ownership(1) |
5% Stockholders | | | | | ||
KV Enzymatics, LLC(2) | | | 3,673,682 | | | 13.8% |
Entities affiliated with Perceptive Life Sciences Master Fund LTD.(3) | | | 3,703,186 | | | 14.0% |
QIAGEN North American Holdings, Inc. | | | 2,403,024 | | | 9.1% |
Entities affiliated with Redmile Biopharma Investments II, L.P.(4) | | | 1,810,442 | | | 6.8% |
Stephen Picone(5) | | | 1,854,204 | | | 7.0% |
Entities affiliated with Boulder Ventures VII, L.P.(6) | | | 1,461,987 | | | 5.5% |
Named Executive Officers and Directors | | | | | ||
Jason Myers(7) | | | 1,238,663 | | | 4.6% |
Joshua Stahl(8) | | | 150,165 | | | * |
Britton Russell(9) | | | 7,444 | | | * |
Marvin Caruthers(10) | | | 101,407 | | | * |
Steven Kafka(11) | | | 220,843 | | | * |
Kyle Lefkoff(6) | | | 1,461,987 | | | 5.5% |
Paul Manning(12) | | | 4,864,453 | | | 18.3% |
Jason Ryan | | | — | | | |
All executive officers and directors as a group (8 persons)(13) | | | 8,044,962 | | | 29.7% |
* | Represents beneficial ownership of less than 1% of class |
(1) | Based on 26,541,272 shares of capital stock outstanding as of August 1, 2020. |
(2) | Mr. Manning, Ian Ratcliffe and Robert Harding are members of KV Enzymatics, LLC, or Enzymatics, and each may be deemed to indirectly own the shares held by Enzymatics. The address for Enzymatics is 200 Garrett Street, Suite S, Charlottesville, VA 22902. |
(3) | Consists of 2,413,922 shares of ArcherDX’s common stock held by Perceptive Life Sciences Master Fund LTD, or Perceptive Life, 527,421 shares of ArcherDX’s common stock held by Perceptive Credit Holdings II, LP, or Perceptive Credit, 79,060 shares of ArcherDX’s common stock held by PCOF EQ AIV II, LP, or, together with Perceptive Life and Perceptive Credit, the Perceptive Entities, and Series B Warrants held by Perceptive Credit to purchase an aggregate of 682,783 shares of ArcherDX’s common stock upon the exercise of the Series B Warrants in connection with the merger. The address for the Perceptive Entities is 51 Astor Place, 10th Floor, New York, NY 10003. |
(4) | Consists of 1,270,298 shares of ArcherDX’s common stock held by Redmile Biopharma Investments II, L.P., or Redmile Biopharma, and 540,144 shares of ArcherDX’s common stock held by Redmile Private Investments II, L.P., Redmile Investments, and together with Redmile Biopharma, Redmile Entities. Redmile Biopharma Investments II (GP), LLC is the general partner of Redmile Biopharma. Redmile Private Investments II (GP), LLC is the general partner of Redmile Investments. The address for the Redmile Entities is One Letterman Drive, Suite D3-300, San Francisco, CA 94129. |
(5) | Consists of 1,204,567 shares of ArcherDX’s common stock held by Stephen Picone, 383,860 shares of ArcherDX’s common stock held by the Stephen Picone Trust, of which Mr. Picone and his spouse are trustees, 195,387 shares of ArcherDX’s common stock to be issued to the Stephen Picone Trust pursuant to the terms of the Baby Genes Agreement in connection with this merger and 70,390 shares of ArcherDX’s common stock held by the Picone 2014 Irrevocable Trust, of which Mr. Picone’s spouse is trustee. The address for Mr. Picone is 507 Valley View Dr., Boulder, CO 80304. |
(6) | Consists of 735,299 shares of ArcherDX’s common stock held by Boulder Ventures VII, L.P., or BV VII, and 726,688 shares of ArcherDX’s common stock held by Boulder Ventures VI, L.P., or BV VI, and together with BV II, or BV Entities. BV Partners VII, LLC, or BVP VII, is the general partner of BV VII and BV Partners VI, LLC, or BVP VI, is the general partner of BV VI. BVP VII may be deemed to indirectly beneficially own the shares owned by BV VII and BVP VI and may be deemed to indirectly own the shares of BV VI. Kyle A. Lefkoff, a member of ArcherDX’s board, Peter A. Roshko and Jonathan L. Perl are managing members of BVP VII and Mr. Lefkoff, Mr. Roshko and Mr. Perl are managing members of BVP VI, and each share voting and dispositive power over the shares held by the applicable BV Entities. The principal business address of the BV Entities is 1941 Pearl Street, Suite 300, Boulder, CO 80302. |
(7) | Includes 187,706 shares Dr. Myers has the right to acquire through the exercise of stock options within 60 days of August 1, 2020 and 174,113 shares of ArcherDX’s common stock to be issued to Dr. Myers pursuant to the terms of the Baby Genes Agreement in connection with the merger. |
(8) | Includes 107,931 shares Mr. Stahl has the right to acquire through the exercise of stock options within 60 days of August 1, 2020. |
(9) | Consists of 7,444 shares Mr. Russell has the right to acquire through the exercise of stock options within 60 days of August 1, 2020. |
(10) | Consists of 8,799 shares Dr. Caruthers has the right to acquire through the exercise of stock options within 60 days of August 1, 2020 and 92,608 shares of ArcherDX’s common stock held by the J.P. Morgan Trust Company of Delaware, Trustee of The Marvin H. Caruthers 2008 Irrevocable Trust of which Dr. Caruthers is trustee. |
(11) | Consists of 220,843 shares Dr. Kafka has the right to acquire through the exercise of stock options within 60 days of August 1, 2020. |
(12) | Consists of 253,461 shares of ArcherDX’s common stock held by BKB Growth Investments LLC, or BKB Growth, 937,310 shares of ArcherDX’s common stock held by PBM and 3,673,682 shares of ArcherDX’s common stock held by Enzymatics. Tiger Lily Capital, LLC is the manager of BKB Growth. Paul B. Manning, one of ArcherDX’s directors, and Bradford Manning are managers of Tiger Lily Capital, LLC, and each share voting and dispositive power over the shares held by BKB Growth. Paul B. Manning, a manager of PBM, may be deemed to indirectly own the shares held by PBM. Mr. Manning, Ian Ratcliffe and Robert Harding are members of Enzymatics, and each may be deemed to indirectly own the shares held by Enzymatics. |
(13) | Includes 532,723 shares issuable upon exercise of stock options exercisable within 60 days of August 1, 2020 and 7,512,239 shares of common stock held by ArcherDX’s current directors and executive officers, including Jason Myers, Joshua Stahl, Britton Russell, Marvin Caruthers, Steven Kafka, Kyle Lefkoff, Paul Manning and Jason Ryan as more fully described above. |
| | | | Rights of Invitae Stockholders | | | Rights of ArcherDX Stockholders | | |
| | Authorized Capital Stock | | | The authorized capital stock of Invitae consists of 400,000,000 shares of common stock, par value $0.0001 per share, and 20,000,000 shares of preferred stock, par value $0.0001 per share. | | | The authorized capital stock of ArcherDX consists of 45,000,000 shares of common stock, par value $0.01 per share, and 28,293,525 shares of preferred stock, par value $0.001 per share. | |
| | Preferred Stock | | | Invitae’s certificate of incorporation provides that the Invitae Board may authorize the issuance of one or more series of preferred stock and fix by resolution (filed in accordance with the DGCL) the number of its shares, designations, preferences, voting powers (full or limited), relative, participating, optional or other special rights and privileges of each series and such qualifications, limitations or restrictions thereof. | | | ArcherDX’s certificate of incorporation does not provide that the ArcherDX Board may authorize the issuance of any series of preferred stock. | |
| | Dividends | | | Invitae’s certificate of incorporation provides that the Invitae Board may declare and pay dividends, from funds lawfully available, on the common stock subject to any preferential dividend rights of any then outstanding preferred stock. | | | ArcherDX’s certificate of incorporation provides that, prior to any payment of dividends on the shares of common stock, each series of preferred stock shall receive a non-cumulative dividend equal to 6% of the applicable original issue price of each such series of preferred stock. After the payment of the preferential dividend amounts described above, the ArcherDX Board may declare and pay dividends, from funds lawfully available, pro rata on the common stock and the preferred stock on a pari passu basis. | |
| | Special Meetings of Stockholders | | | Under the DGCL, a special meeting of stockholders may be called by the board of directors or by any other person authorized to do so in the certificate of incorporation or by-laws. | | | Under the DGCL, a special meeting of stockholders may be called by the board of directors or by any other person authorized to do so in the certificate of incorporation or by-laws. | |
| | | | Rights of Invitae Stockholders | | | Rights of ArcherDX Stockholders | | |
| | | | Invitae’s certificate of incorporation and bylaws provide that special meetings of stockholders may be called from time to time only by a majority of the Invitae Board, the Chief Executive Officer or the Chairman of the Invitae Board for the purpose specified in the notice of meeting. Any power of stockholders to call a special meeting of stockholders is specifically denied. | | | ArcherDX’s bylaws provide that special meetings of stockholders may be called from time to time only by the ArcherDX Board, the chairperson of the ArcherDX Board, or one or more stockholders holding shares in the aggregate entitled to cast not less than 10% of the votes at that meeting. | | |
| | Special Meetings of the Board | | | Invitae’s bylaws provide that special meetings of the Invitae Board may be called from time to time by the Secretary only at the request of the Chairman of the Board, the Chief Executive Officer, or by written consent of a majority of the directors then in office. | | | ArcherDX’s bylaws provide that special meetings of the ArcherDX Board may be called from time to time by the chairperson of the ArcherDX Board, the Chief Executive Officer, the President, the Secretary, or any two directors. | |
| | Stockholder Action by Written Consent | | | Under the DGCL, any action that can be taken at any annual or special meeting of stockholders of a corporation may also be taken by stockholders without a meeting, without prior notice and without a vote unless the certificate of incorporation provides otherwise. | | | Under the DGCL, any action that can be taken at any annual or special meeting of stockholders of a corporation may also be taken by stockholders without a meeting, without prior notice and without a vote unless the certificate of incorporation provides otherwise. | |
| | | | Invitae’s certificate of incorporation provides that stockholders may not take action by written consent. | | | ArcherDX’s certificate of incorporation does not prohibit stockholders from taking action by written consent. | | |
| | Stockholder Proposals and Nominations of Candidates for Election to the Board of Directors | | | Invitae’s bylaws allow stockholders who are entitled to vote at the meeting and who timely gave notice in writing to Invitae’s secretary prior to the meeting, to nominate candidates for election to the Invitae Board. Stockholders who are entitled to vote at the meeting and who timely gave notice in writing to Invitae’s secretary prior to the meeting may also propose business to be considered by stockholders at an annual meeting. | | | ArcherDX’s bylaws do not allow stockholders to (i) nominate candidates for election to the ArcherDX Board or (ii) propose business to be conducted at an annual meeting. | |
| | | | Such proposals and nominations (other than matters included in the notice of meeting pursuant to Rule 14a-8 promulgated under the Exchange Act) may only be brought in accordance with the applicable provision of Invitae’s bylaws. | | | |
| | | | Rights of Invitae Stockholders | | | Rights of ArcherDX Stockholders | | |
| | | | In connection with an annual meeting, to be timely, notice of such proposals and nominations must be delivered to the Secretary at Invitae’s principal executive office by a nationally recognized courier service or mailed by first class United States mail, postage or delivery charges prepaid not more than 120 days nor less than 90 days in advance of the anniversary of the date of Invitae’s proxy statement provided in connection with the previous year’s annual meeting of stockholders, or if no annual meeting was held in the previous year or the annual meeting is called for a date that is more than 30 days before or after the anniversary date of the previous year’s annual meeting, notice by the stockholder must be received by Invitae’s Secretary not later than the close of business on the later of (x) the 90th day prior to such annual meeting and (y) the 10th day following the day on which public announcement of the date of such meeting is first made by Invitae. In the case of a special meeting of stockholders called for the purpose of electing directors, to be timely, notice of such nominations must be delivered not later than the close of business on the 10th day following the day on which public announcement of the date of such meeting is first made by Invitae. | | | | ||
| | Proxy Access for Director Nominations | | | Neither the Invitae certificate of incorporation nor the Invitae bylaws contain proxy access provisions. | | | Neither the ArcherDX certificate of incorporation nor the ArcherDX bylaws contain proxy access provisions. | |
| | Number of Directors | | | The DGCL provides that the board of directors of a Delaware corporation must consist of one or more directors, each of whom must be a natural person, with the number of directors fixed by or in the manner provided in the corporation’s bylaws unless the certificate of incorporation fixes the number of directors. | | | The DGCL provides that the board of directors of a Delaware corporation must consist of one or more directors, each of whom must be a natural person, with the number of directors fixed by or in the manner provided in the corporation’s bylaws unless the certificate of incorporation fixes the number of directors. | |
| | | | Rights of Invitae Stockholders | | | Rights of ArcherDX Stockholders | | |
| | | | Invitae’s certificate of incorporation and bylaws provide that the number of directors will be determined by resolution of the majority of the Invitae directors then in office. | | | ArcherDX’s certificate of incorporation and bylaws provide that the number of directors will be determined by resolution of the ArcherDX Board. | | |
| | | | There are currently five directors serving on the Invitae Board. | | | There are currently six directors serving on the ArcherDX Board. | | |
| | Election of Directors | | | The DGCL provides that, unless the certificate of incorporation or bylaws provide otherwise, directors will be elected by a plurality of the votes of the shares present in person or represented by proxy at the meeting and entitled to vote on the election of directors. The DGCL also permits classified boards. | | | The DGCL provides that, unless the certificate of incorporation or bylaws provide otherwise, directors will be elected by a plurality of the votes of the shares present in person or represented by proxy at the meeting and entitled to vote on the election of directors. The DGCL also permits classified boards. | |
| | | | Invitae’s certificate of incorporation and bylaws provide that the Invitae Board is divided into three classes of directors, each class serving staggered three-year terms and being as nearly equal in number as possible. Each class holds office until the third annual meeting of stockholders following the most recent election of such class. | | | Neither ArcherDX’s certificate of incorporation nor bylaws provide for the division of the ArcherDX Board into classes of directors. | | |
| | | | Invitae’s directors are elected at the annual meeting of stockholders by such stockholders as have the right to vote on such election. An election of directors by the stockholders is determined by a plurality of the votes cast by the stockholders entitled to vote at such election. | | | ArcherDX’s directors can be elected at the annual meeting of stockholders by such stockholders as have the right to vote on such election or by written consent in lieu of an annual meeting. | | |
| | | | The Invitae Board has adopted a rule requiring any nominee for director who receives a greater number of votes “against” or “withheld” than votes “for” in an uncontested election to promptly tender his or her irrevocable resignation for consideration by the Invitae Board. If such director’s resignation is accepted by the Invitae Board, then the Invitae Board, in its sole discretion, may fill the resulting vacancy in accordance with Invitae’s bylaws. | | | The ArcherDX Board does not have a rule requiring any nominee for director who receives a greater number of votes “against” or “withheld” than votes “for” in an uncontested election to promptly tender his or her irrevocable resignation for consideration by the ArcherDX Board. | |
| | | | Rights of Invitae Stockholders | | | Rights of ArcherDX Stockholders | | |
| | Removal of Directors | | | The DGCL provides that directors serving on a classified board may be removed only for cause by the holders of a majority of the shares then entitled to vote in an election of directors, unless a higher threshold is provided in the certificate of incorporation. | | | The DGCL provides that directors serving on a classified board may be removed only for cause by the holders of a majority of the shares then entitled to vote in an election of directors, unless a higher threshold is provided in the certificate of incorporation. | |
| | | | Invitae’s bylaws provide that directors may be removed, but only for cause, by the holders of a majority of the voting power of the capital stock issued and outstanding then entitled to vote at an election of directors. | | | ArcherDX’s bylaws provide that directors may be removed, with or without cause, by the holders of a majority of the shares then entitled to vote at an election of directors. ArcherDX’s certificate of incorporation provides that the three directors elected by the holders of preferred stock, voting together as a single class on an as-converted basis, and the director elected by the common stock, exclusively and as a separate class, may be removed without cause by, and only by, the affirmative vote of the holders entitled to elect such director or directors. | | |
| | Vacancies of Directors | | | Invitae’s certificate of incorporation and bylaws provide that any vacancy in the Invitae Board may be filled by a majority of the directors then in office or by a sole remaining director. | | | ArcherDX’s bylaws provide that, unless otherwise provided in ArcherDX’s certificate of incorporation, any vacancy in the ArcherDX Board may be filled by a majority of the directors then in office or by a sole remaining director. | |
| | | | | | ArcherDX’s certificate of incorporation provides that if the holders of the shares of preferred stock or common stock, as the case may be, fail to elect a sufficient number of directors to fill all directorships for which they are entitled to elect directors, voting exclusively and as a separate class, pursuant to ArcherDX’s certificate of incorporation, then any directorship not so filled shall remain vacant until such time as the holders of the preferred stock or common stock, as the case may be, elect a person to fill such directorship by vote or written consent in lieu of a meeting; and no such directorship may be filled by stockholders of ArcherDX other than by the stockholders of ArcherDX that are entitled to elect a person to fill such directorship, voting exclusively and as a separate class. | |
| | | | Rights of Invitae Stockholders | | | Rights of ArcherDX Stockholders | | |
| | Limitation on Liability of Directors | | | The DGCL permits corporations to include provisions in their certificate of incorporation eliminating monetary damages for a director for any breach of fiduciary duty. A corporation may not eliminate liability for a director’s breach of the duty of loyalty to the corporation or its stockholders, for acts or omissions not in good faith or which involve intentional misconduct or a knowing violation of law, for unlawful dividends, stock purchases or redemptions, or for any transaction from which the director derived an improper personal benefit. | | | The DGCL permits corporations to include provisions in their certificate of incorporation eliminating monetary damages for a director for any breach of fiduciary duty. A corporation may not eliminate liability for a director’s breach of the duty of loyalty to the corporation or its stockholders, for acts or omissions not in good faith or which involve intentional misconduct or a knowing violation of law, for unlawful dividends, stock purchases or redemptions, or for any transaction from which the director derived an improper personal benefit. | |
| | | | In accordance with the DGCL, Invitae’s certificate of incorporation provides that no director will be personally liable to Invitae or its stockholders for monetary damages for breach of fiduciary duty as a director to the fullest extent permitted by the DGCL. | | | In accordance with the DGCL, ArcherDX’s certificate of incorporation provides that no director will be personally liable to ArcherDX or its stockholders for monetary damages for breach of fiduciary duty as a director to the fullest extent permitted by the DGCL. | | |
| | Indemnification of Directors and Officers | | | Under the DGCL, a Delaware corporation must indemnify its present or former directors and officers against expenses (including attorneys’ fees) actually and reasonably incurred to the extent that the officer or director has been successful on the merits or otherwise in defense of any action, suit or proceeding brought against him or her by reason of the fact that he or she is or was a director or officer of the corporation. | | | Under the DGCL, a Delaware corporation must indemnify its present or former directors and officers against expenses (including attorneys’ fees) actually and reasonably incurred to the extent that the officer or director has been successful on the merits or otherwise in defense of any action, suit or proceeding brought against him or her by reason of the fact that he or she is or was a director or officer of the corporation. | |
| | | | Delaware law provides that a corporation may indemnify its present and former directors, officers, employees and agents, as well as any individual serving with another corporation in that capacity at the corporation’s request against expenses (including attorney’s fees), judgments, fines and amounts paid in settlement of actions taken, if the individual acted in good faith and in a manner reasonably believed to be in, or not opposed to, the best interests of the corporation and, in the case of a criminal proceeding, the individual had no reasonable cause to | | | Delaware law provides that a corporation may indemnify its present and former directors, officers, employees and agents, as well as any individual serving with another corporation in that capacity at the corporation’s request against expenses (including attorney’s fees), judgments, fines and amounts paid in settlement of actions taken, if the individual acted in good faith and in a manner reasonably believed to be in, or not opposed to, the best interests of the corporation and, in the case of a criminal proceeding, the individual had no reasonable cause to | |
| | | | Rights of Invitae Stockholders | | | Rights of ArcherDX Stockholders | | |
| | | | believe the individual’s conduct was unlawful. However, no indemnification may be paid for judgments and settlements in actions by or in the right of the corporation. | | | believe the individual’s conduct was unlawful. However, no indemnification may be paid for judgments and settlements in actions by or in the right of the corporation. | | |
| | | | A corporation may not indemnify a current or former director or officer of the corporation against expenses to the extent the person is adjudged to be liable to the corporation unless a court approves the indemnity. | | | A corporation may not indemnify a current or former director or officer of the corporation against expenses to the extent the person is adjudged to be liable to the corporation unless a court approves the indemnity. | | |
| | | | The DGCL permits a corporation to purchase and maintain insurance on behalf of any person who is or was a director, officer, employee or agent of a corporation, or is or was serving at the request of the corporation as a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise against any liability asserted against such person and incurred by such person in any such capacity, or arising out of such person’s status as such. | | | The DGCL permits a corporation to purchase and maintain insurance on behalf of any person who is or was a director, officer, employee or agent of a corporation, or is or was serving at the request of the corporation as a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise against any liability asserted against such person and incurred by such person in any such capacity, or arising out of such person’s status as such. | | |
| | | | Invitae’s bylaws provide that it will indemnify any person who was or is made a party or is threatened to be made a party to or is involved in any actual or threatened action, suit or proceeding, whether civil, criminal, administrative or investigative, by reason of the fact that such person is or was a director or officer of Invitae, or is or was serving at the request of Invitae as a director, officer, employee or agent of another corporation, partnership, limited liability company, joint venture, trust, employee benefit plan sponsored or maintained by Invitae, or other enterprise, against all expense, liability and loss (including attorneys’ fees and related disbursements, judgments, fines, excise taxes or penalties and amounts paid or to be paid in settlement) actually and reasonably incurred by such person in connection with such action, suit or proceeding if such person acted in good faith and in a manner such person reasonably believed to be in or not opposed to the best interests of | | | ArcherDX’s bylaws provide that ArcherDX will indemnify any person who was or is a party or is threatened to be made a party to any threatened, pending or completed action, suit or proceeding, whether civil, criminal, administrative or investigative (other than an action by or in the right of ArcherDX) by reason of the fact that such person is or was a director or officer of ArcherDX, or is or was a director or officer of ArcherDX serving at the request of ArcherDX as a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise, against expenses (including attorneys’ fees), judgments, fines and amounts paid in settlement actually and reasonably incurred by such person in connection with such action, suit or proceeding if such person acted in good faith and in a manner such person reasonably believed to be in or not opposed to the best interests of ArcherDX, and, with respect to any criminal action or proceeding, had no | |
| | | | Rights of Invitae Stockholders | | | Rights of ArcherDX Stockholders | | |
| | | | Invitae, and, with respect to any criminal action or proceeding, had no reasonable cause to believe such person’s conduct was unlawful. | | | reasonable cause to believe such person’s conduct was unlawful. | | |
| | | | | | ArcherDX’s bylaws also provide that ArcherDX will indemnify any person who was or is a party or is threatened to be made a party to any threatened, pending or completed action or suit by or in the right of ArcherDX to procure a judgment in its favor by reason of the fact that such person is or was a director or officer of ArcherDX, or is or was a director or officer of ArcherDX serving at the request of ArcherDX as a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise against expenses (including attorneys’ fees) actually and reasonably incurred by such person in connection with the defense or settlement of such action or suit if such person acted in good faith and in a manner such person reasonably believed to be in or not opposed to the best interests of ArcherDX; except that no indemnification shall be made in respect of any claim, issue or matter as to which such person shall have been adjudged to be liable to ArcherDX unless and only to the extent that the Delaware Court of Chancery or the court in which such action or suit was brought shall determine upon application that, despite the adjudication of liability but in view of all the circumstances of the case, such person is fairly and reasonably entitled to indemnity for such expenses which the Delaware Court of Chancery or such other court shall deem proper. | | ||
| | | | Invitae’s bylaws further provide that a person entitled to indemnification will also have the right advancement of expenses incurred in defending any proceeding, provided that if the DGCL requires, the payment of such expenses incurred by a director or officer in his or her capacity as a | | | ArcherDX’s bylaws further provide that expenses (including attorneys’ fees) incurred by an officer or director of ArcherDX in defending any proceeding will be paid by ArcherDX in advance of the final disposition of such proceeding upon receipt of a written request therefor | |
| | | | Rights of Invitae Stockholders | | | Rights of ArcherDX Stockholders | | |
| | | | director or officer (and not in any other capacity in which service was or is rendered by such person while a director or officer, including, without limitation, service to an employee benefit plan) in advance of the final disposition of a proceeding, shall be made only upon delivery to Invitae of an undertaking by or on behalf of such director or officer to repay all amounts so advanced if it is ultimately determined that such director or officer is not entitled to be indemnified. In addition, no advance shall be made to an officer of Invitae (unless such officer is or was a director of Invitae) in any action, suit or proceeding, whether civil, criminal, administrative or investigative, if a determination is made by the disinterested members of Invitae’s board of directors or independent legal counsel, that such person acted in bad faith or in a manner that such person did not believe to be in or not opposed to the best interests of Invitae. | | | (together with documentation reasonably evidencing such expenses) and an undertaking by or on behalf of the person to repay such amounts if it is ultimately determined that the person is not entitled to be indemnified under ArcherDX’s bylaws or the DGCL. Such expenses (including attorneys’ fees) incurred by former directors and officers or other employees and agents of ArcherDX or by persons serving at the request of ArcherDX as directors, officers, employees or agents of another corporation, partnership, joint venture, trust or other enterprise may be so paid upon such terms and conditions, if any, as ArcherDX deems appropriate. | | |
| | Exclusive Forum | | | Invitae’s certificate of incorporation provides, unless Invitae consents in writing to the selection of an alternative forum, the Court of Chancery of the State of Delaware shall be the sole and exclusive forum for (i) any derivative action or proceeding brought on behalf of Invitae, (ii) any action asserting a claim of breach of a fiduciary duty owed by any of Invitae’s directors, officers or other employees to Invitae or its stockholders, (iii) any action asserting a claim arising pursuant to any provision of the DGCL, or (iv) any action asserting a claim governed by the internal affairs doctrine. | | | ArcherDX’s certificate of incorporation does not contain a provision for an exclusive forum. | |
| | | | Rights of Invitae Stockholders | | | Rights of ArcherDX Stockholders | | |
| | Amendments to Certificate of Incorporation | | | Under the DGCL, an amendment to the certificate of incorporation generally requires (1) the approval of the board of directors, (2) the approval of a majority of the voting power of the outstanding stock entitled to vote upon the proposed amendment and (3) the approval of the holders of a majority of the outstanding stock of each class entitled to vote thereon as a class, if any. | | | Under the DGCL, an amendment to the certificate of incorporation generally requires (1) the approval of the board of directors, (2) the approval of a majority of the voting power of the outstanding stock entitled to vote upon the proposed amendment and (3) the approval of the holders of a majority of the outstanding stock of each class entitled to vote thereon as a class, if any. | |
| | | | Invitae’s certificate of incorporation provides that the affirmative vote of the holders of at least 66-2/3% of the voting power of the shares of Invitae’s capital stock entitled to vote generally in the election of directors, voting together as a single class, shall be required to amend in any respect or repeal Paragraph A of Article V (Amendments to Bylaws), Article VI (Board of Directors; Newly Created Directorships), Article VII (Action By Written Consent; Meetings of Stockholders; Stockholder Nominations; Exclusive Forum), Article VIII (Limitation on Liability; Indemnification; Insurance) or Article IX (Amendment to Certificate of Incorporation). | | | ArcherDX’s certificate of incorporation contains “protective provisions” that require separate approvals of (i) a majority of the preferred stock, voting together as a single class on an as-converted basis, (ii) a majority of the Series A preferred stock, (iii) a majority of the Series B preferred stock and (iv) a majority of the Series C preferred stock, in each case in order to amend the charter in furtherance of any of several enumerated actions set forth in ArcherDX’s certificate of incorporation. | | |
| | Amendments to Bylaws | | | The Invitae certificate of incorporation authorizes the Invitae board of directors to adopt, amend or repeal Invitae’s bylaws, without any action on the part of the stockholders, by the vote of at least a majority of the directors then in office. In addition, Invitae’s bylaws may be adopted, amended or repealed by the affirmative vote of the holders of at least two-thirds of the voting power of the shares of Invitae’s capital stock entitled to vote in the election of directors, voting as one class; provided, however, that the affirmative vote of the holders representing only a majority of the voting power of the shares of Invitae’s capital stock entitled to vote in the election of directors, voting as one class, shall be required if such adoption, amendment or repeal of | | | ArcherDX’s bylaws may be adopted, amended or repealed by the vote of the stockholders entitled to vote thereon. In addition, ArcherDX’s certificate of incorporation authorizes the ArcherDX Board to make, repeal, alter, amend and rescind any or all of ArcherDX’s bylaws, without any action on the part of the stockholders; provided that pursuant to ArcherDX’s bylaws, a bylaw amendment adopted by stockholders which specifies the votes that shall be necessary for the election of directors shall not be further amended or repealed by the ArcherDX Board. | |
| | | | Rights of Invitae Stockholders | | | Rights of ArcherDX Stockholders | | |
| | | | Invitae’s bylaws has been previously approved by the affirmative vote of at least two-thirds of the directors then in office. | | | | ||
| | Certain Business Combinations | | | Section 203 of the DGCL prohibits a Delaware corporation from engaging in a business combination with a stockholder acquiring more than 15% but less than 85% of the corporation’s outstanding voting stock for three years following the time that person becomes an “interested stockholder” (a holder of more than 15% of the corporation’s outstanding shares), unless prior to the date the person becomes an interested stockholder, the board of directors approves either the business combination or the transaction which resulted in the stockholder becoming an interested stockholder or the business combination is approved by the board of directors and by the affirmative vote of at least two-thirds of the outstanding voting stock that is not owned by the interested stockholder or other specified exceptions are met. The DGCL allows a corporation’s certificate of incorporation to contain a provision expressly electing not to be governed by Section 203. | | | Section 203 of the DGCL prohibits a Delaware corporation from engaging in a business combination with a stockholder acquiring more than 15% but less than 85% of the corporation’s outstanding voting stock for three years following the time that person becomes an “interested stockholder” (a holder of more than 15% of the corporation’s outstanding shares), unless prior to the date the person becomes an interested stockholder, the board of directors approves either the business combination or the transaction which resulted in the stockholder becoming an interested stockholder or the business combination is approved by the board of directors and by the affirmative vote of at least two-thirds of the outstanding voting stock that is not owned by the interested stockholder or other specified exceptions are met. The DGCL allows a corporation’s certificate of incorporation to contain a provision expressly electing not to be governed by Section 203. | |
| | | | Although the DGCL permits a Delaware corporation’s certificate of incorporation to provide for a greater vote for a merger, consolidation or sale of substantially all the assets of a corporation than the vote described above, the Invitae certificate of incorporation does not require a greater vote. | | | Although the DGCL permits a Delaware corporation’s certificate of incorporation to provide for a greater vote for a merger, consolidation or sale of substantially all the assets of a corporation than the vote described above, the ArcherDX certificate of incorporation does not require a greater vote. | | |
| | Stockholder Rights Plan | | | The DGCL does not include a statutory provision expressly validating stockholder rights plans. However, such plans have generally been upheld by the decisions of courts applying Delaware law. | | | The DGCL does not include a statutory provision expressly validating stockholder rights plans. However, such plans have generally been upheld by the decisions of courts applying Delaware law. | |
| | | | Invitae does not have a stockholder rights plan currently in effect. | | | ArcherDX does not have a stockholder rights plan currently in effect. | |
• | its Annual Report on Form 10-K for the year ended December 31, 2019; |
• | its Quarterly Reports on Form 10-Q for the quarters ended March 31, 2020 and June 30, 2020; |
• | its Current Reports on Form 8-K filed with the SEC on March 10, 2020 (as amended on March 16, 2020), April 1, 2020 (to the extent filed under Items 8.01 and 9.01), April 6, 2020, May 22, 2020, June 12, 2020, June 18, 2020, June 22, 2020, June 24, 2020 and June 26, 2020; and |
• | The description of the Invitae common stock contained in Invitae’s Registration Statement on Form 8-A, filed with the SEC on February 11, 2015, including any further amendment or report filed hereafter for the purpose of updating such description. |
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | |
| | | ||
| | | ||
| | | ||
| | | ||
| | |
| | | 2018 | | | 2019 | |
ASSETS | | | | | ||
Current assets: | | | | | ||
Cash and cash equivalents | | | $9,324 | | | $59,492 |
Accounts receivable, net | | | 6,743 | | | 15,354 |
Inventories | | | 2,506 | | | 6,002 |
Prepaid expenses and other current assets | | | 658 | | | 2,442 |
Total current assets | | | 19,231 | | | 83,290 |
Property and equipment, net | | | 2,813 | | | 10,811 |
Right-of-use assets, net | | | — | | | 4,813 |
Intangible assets, net | | | 1,755 | | | 780 |
Goodwill | | | 5,453 | | | 4,972 |
Restricted cash | | | 150 | | | — |
Other assets | | | 73 | | | 2,130 |
Total assets | | | $29,475 | | | $106,796 |
LIABILITIES, CONVERTIBLE PREFERRED STOCK, AND STOCKHOLDERS’ DEFICIT | | | | | ||
Current liabilities | | | | | ||
Accounts payable | | | $3,540 | | | $5,657 |
Accrued expenses | | | 1,193 | | | 2,025 |
Accrued compensation | | | 595 | | | 2,824 |
Lease liabilities | | | — | | | 577 |
Other current liabilities | | | 27 | | | 1,060 |
Deferred revenue | | | 3,704 | | | 6,234 |
Total current liabilities | | | 9,059 | | | 18,377 |
Long-term lease liabilities - less current portion | | | — | | | 4,310 |
Long-term debt, net | | | — | | | 28,572 |
Other long-term liabilities | | | 3,331 | | | 10,622 |
Total liabilities | | | 12,390 | | | 61,881 |
Commitments and contingencies (Note 16) | | | | | ||
Convertible preferred stock, $0.001 par value, 15,000,000 and 28,293,525 shares authorized, 8,783,772 and 17,609,298 shares issued and outstanding with aggregate liquidation preference of $44,413 and $114,397 as of December 31, 2018 and 2019, respectively | | | 42,180 | | | 110,154 |
Stockholders’ deficit: | | | | | ||
Common stock, $0.01 par value, 30,000,000 and 45,000,000 shares authorized, 6,375,767 and 6,454,355 shares issued and outstanding as of December 31, 2018 and 2019, respectively | | | 64 | | | 65 |
Additional paid-in capital | | | 3,106 | | | 3,939 |
Accumulated deficit | | | (28,265) | | | (69,243) |
Total stockholders’ equity (deficit) | | | (25,095) | | | (65,239) |
Total liabilities, convertible preferred stock, and stockholders’ deficit | | | $29,475 | | | $106,796 |
| | | Year Ended December 31, | ||||
| | | 2018 | | | 2019 | |
Revenue | | | | | ||
Precision oncology products | | | $16,025 | | | $22,644 |
Pharma development services | | | 12,429 | | | 27,921 |
Total revenue | | | 28,454 | | | 50,565 |
Costs & operating expenses | | | | | ||
Cost of precision oncology products | | | 4,033 | | | 7,335 |
Cost of pharma development services | | | 6,230 | | | 9,212 |
Sales and marketing | | | 7,215 | | | 15,428 |
Research and development | | | 8,184 | | | 34,172 |
General and administrative | | | 7,700 | | | 15,875 |
Contingent consideration | | | — | | | 5,768 |
Total operating expenses | | | 33,362 | | | 87,790 |
Loss from operations | | | (4,908) | | | (37,225) |
Interest expense, net | | | (1,160) | | | (2,432) |
Other income (expense), net | | | 34 | | | (824) |
Loss before income taxes | | | (6,034) | | | (40,481) |
Income tax (benefit) expense | | | (481) | | | 497 |
Net loss and comprehensive loss | | | $(5,553) | | | $(40,978) |
Basic and diluted loss per common share | | | $(0.87) | | | $(6.39) |
Basic and diluted weighted-average common shares outstanding | | | 6,376,987 | | | 6,415,227 |
| | | Convertible Preferred Stock | | | Common Stock | | | Additional Paid-In Capital | | | Accumulated Deficit | | | Total Stockholders’ Deficit | |||||||
| | | Shares | | | Amount | | | Shares | | | Amount | | |||||||||
Balance - January 1, 2018 | | | — | | | — | | | 6,377,878 | | | $64 | | | $2,833 | | | $(22,712) | | | $(19,815) |
Issuance of Series A convertible preferred stock for cash and conversion of debt, net of issuance costs of $206 | | | 7,764,831 | | | 37,436 | | | — | | | — | | | — | | | — | | | — |
Issuance of Series A convertible preferred stock as merger consideration | | | 1,018,941 | | | 4,744 | | | — | | | — | | | — | | | — | | | — |
Issuance of common stock options as merger consideration | | | — | | | — | | | — | | | — | | | 47 | | | — | | | 47 |
Repurchase of restricted stock | | | — | | | — | | | (2,111) | | | — | | | — | | | — | | | — |
Stock-based compensation expense | | | — | | | — | | | — | | | — | | | 226 | | | — | | | 226 |
Net loss | | | — | | | — | | | — | | | — | | | — | | | (5,553) | | | (5,553) |
Balance - December 31, 2018 | | | 8,783,772 | | | 42,180 | | | 6,375,767 | | | 64 | | | 3,106 | | | (28,265) | | | (25,095) |
Issuance of Series B and Series C convertible preferred stock for cash, net of issuance costs of $2,010 | | | 8,828,807 | | | 67,990 | | | — | | | — | | | — | | | — | | | — |
Repurchase of Series A convertible preferred stock | | | (3,281) | | | (16) | | | — | | | — | | | — | | | — | | | — |
Stock-based compensation expense | | | — | | | — | | | — | | | — | | | 751 | | | — | | | 751 |
Proceeds from exercise of stock options | | | — | | | — | | | 78,588 | | | 1 | | | 82 | | | — | | | 83 |
Net loss | | | — | | | — | | | — | | | — | | | — | | | (40,978) | | | (40,978) |
Balance - December 31, 2019 | | | 17,609,298 | | | $110,154 | | | 6,454,355 | | | $65 | | | $3,939 | | | $(69,243) | | | $(65,239) |
| | | For the Years Ended December 31, | ||||
| | | 2018 | | | 2019 | |
Cash flows from operating activities | | | | | ||
Net loss | | | $(5,553) | | | $(40,978) |
Adjustments to reconcile net loss to net cash used in operating activities | | | | | ||
Depreciation and amortization | | | 849 | | | 2,687 |
Amortization of debt issuance costs and debt extinguishment | | | 739 | | | 197 |
Bad debt expense | | | 60 | | | (60) |
Stock-based compensation expense | | | 226 | | | 751 |
Change in fair value of contingent consideration | | | — | | | 5,768 |
Change in fair value of convertible preferred stock warrants | | | (64) | | | 802 |
Change in allowance for obsolete inventory | | | 188 | | | 37 |
Deferred tax (benefit) expense | | | (481) | | | 481 |
Changes in assets and liabilities | | | | | ||
Accounts receivable | | | (3,030) | | | (8,551) |
Inventories | | | (1,142) | | | (3,533) |
Prepaid expenses and other assets | | | (302) | | | (4,464) |
Accounts payable and accrued liabilities | | | 2,218 | | | 6,833 |
Lease liabilities | | | — | | | 45 |
Deferred revenue | | | 1,704 | | | 2,531 |
Deferred rent | | | (15) | | | — |
Net cash used in operating activities | | | (4,603) | | | (37,454) |
Cash flows from investing activities | | | | | ||
Purchase of property and equipment | | | (2,324) | | | (9,710) |
Cash acquired in merger transaction | | | 346 | | | — |
Net cash used in investing activities | | | (1,978) | | | (9,710) |
Cash flows from financing activities | | | | | ||
Proceeds from issuance of convertible preferred stock, net of issuance costs | | | 14,793 | | | 67,990 |
Proceeds from borrowing under credit facility, net of issuance costs | | | — | | | 29.125 |
Repayment of related party debt | | | (1,250) | | | — |
Payments on capital lease obligations | | | (15) | | | — |
Proceeds from exercise of stock options | | | — | | | 83 |
Repurchase of Series A convertible preferred stock | | | — | | | (16) |
Net cash provided by financing activities | | | 13,528 | | | 97,182 |
Net increase in cash, cash equivalents, and restricted cash | | | 6,947 | | | 50,018 |
Cash, cash equivalents, and restricted cash at beginning of year | | | 2,527 | | | 9,474 |
Cash, cash equivalents, and restricted cash at end of year | | | $9,474 | | | $59,492 |
Non-cash financing activities: | | | | | ||
Conversion of convertible debt and accrued interest into convertible preferred stock | | | $22,642 | | | $— |
Issuance of warrant with credit facility | | | — | | | $750 |
Issuance of convertible preferred stock for merger consideration | | | $4,745 | | | $— |
1. | Nature of Business |
2. | Summary of Significant Accounting Policies |
| | | December 31, | ||||
| | | 2018 | | | 2019 | |
Cash and cash equivalents | | | $9,324 | | | $59,492 |
Restricted cash | | | 150 | | | — |
Total cash and cash equivalents and restrictions | | | $9,474 | | | $59,492 |
| | | December 31, | ||||
| | | 2018 | | | 2019 | |
United States | | | $16,565 | | | $18,069 |
International | | | 11,889 | | | 32,496 |
Total revenue | | | $28,454 | | | $50,565 |
| | | Revenue for the Year Ended December 31 | | | Accounts Receivable as of December 31 | |||||||
| | | 2018 | | | 2019 | | | 2018 | | | 2019 | |
Customer A(1) | | | 22% | | | 5% | | | 13% | | | 2% |
Customer B(1) | | | 20% | | | 44% | | | 25% | | | 42% |
(1) | The country of headquarters of Customer A is the US, and the country of headquarters of Customer B is Germany |
Property and Equipment | | | Estimated Useful Life |
Furniture and fixtures | | | 7 years |
Manufacturing and lab equipment | | | 5 years |
Computer equipment | | | 3 years |
Leasehold improvements | | | Lesser of estimated useful life or remaining lease term |
| | | December 31, | ||||
| | | 2018 | | | 2019 | |
Deferred revenue, beginning balance | | | $2,000 | | | $3,704 |
Plus deferred revenue added during the year | | | 4,388 | | | 9,601 |
Less Revenue recognized from deferred revenue during the year | | | (2,684) | | | (7,071) |
Deferred revenue, ending balance | | | $3,704 | | | $6,234 |
3. | Business Combination |
Consideration | | | |
Series A convertible preferred stock | | | $4,745 |
Net working capital adjustment paid in cash | | | 81 |
Common stock options issued for vested Baby Genes options | | | 47 |
Contingent consideration | | | 2,559 |
Fair value of total consideration transferred | | | $7,432 |
Recognized amounts of identifiable assets acquired and liabilities assumed | | | |
Cash | | | $346 |
Other current assets | | | 228 |
Property and equipment, net | | | 80 |
Intangible assets | | | 1,929 |
Total assets acquired | | | 2,583 |
Current liabilities | | | (123) |
Total liabilities assumed | | | (123) |
Total identifiable net assets | | | 2,460 |
Goodwill | | | $4,972 |
| | | Unaudited Pro Forma Financial Information | |
Revenue | | | $29,151 |
Net loss and comprehensive loss | | | $(6,087) |
Basic and diluted loss per share | | | $(0.95) |
4. | Fair Value Measurements |
| | | Level 1 | | | Level 2 | | | Level 3 | |
December 31, 2018 | | | | | | | |||
Assets: | | | | | | | |||
Money market accounts | | | $80 | | | — | | | |
Liabilities: | | | | | | | |||
Warrant liability | | | — | | | — | | | $(742) |
Contingent consideration | | | — | | | — | | | $(2,559) |
December 31, 2019 | | | | | | | |||
Assets: | | | | | | | |||
Money market accounts | | | $43,154 | | | — | | | — |
Liabilities: | | | | | | | |||
Warrant liabilities | | | — | | | — | | | $(2,295) |
Contingent consideration | | | | | | | $(8,327) |
| | | Convertible Preferred Stock Warrants | | | Contingent Consideration | |
Balance as of December 31, 2017 | | | $806 | | | $— |
Exchange for Series A convertible preferred stock warrant | | | — | | | — |
Change in fair value of Series A convertible preferred stock warrant | | | (64) | | | — |
Addition of contingent consideration | | | — | | | 2,559 |
Balance as of December 31, 2018 | | | 742 | | | 2,559 |
Issuance of Series B convertible preferred stock warrant | | | 750 | | | — |
Change in fair value of Series A and B convertible preferred stock warrants | | | 803 | | | — |
Change in fair value of contingent consideration | | | — | | | 5,768 |
Balance as of December 31, 2019 | | | $2,295 | | | $8,327 |
5. | Inventories |
| | | December 31, | ||||
| | | 2018 | | | 2019 | |
Raw materials | | | $1,966 | | | $3,640 |
Work in process | | | 149 | | | 1,412 |
Finished goods | | | 391 | | | 950 |
Total inventories | | | $2,506 | | | $6,002 |
6. | Property and Equipment |
| | | December 31, | ||||
| | | 2018 | | | 2019 | |
Manufacturing and lab equipment | | | $4,136 | | | $490 |
Office equipment | | | 490 | | | 1,490 |
Leasehold improvements | | | 105 | | | 421 |
Construction in progress | | | — | | | 405 |
Property and equipment, gross | | | 4,731 | | | 14,441 |
Less accumulated depreciation and amortization | | | (1,918) | | | (3,630) |
Property and equipment, net | | | $2,813 | | | $10,811 |
7. | Goodwill and Intangible Assets |
| | | Estimated Useful Life in Years | | | December 31, | ||||
| | | 2018 | | | 2019 | ||||
Permits and licenses | | | 0.8 | | | $898 | | | $898 |
Trade name | | | 7.0 | | | 606 | | | 606 |
Non-competition agreement | | | 0.8 | | | 518 | | | 518 |
Intangible assets, gross | | | | | 2,022 | | | 2,022 | |
Less accumulated amortization | | | | | (267) | | | (1,242) | |
Intangible assets, net | | | | | $(1,755) | | | $780 | |
Year Ending December 31, | | | |
2020 | | | $315 |
2021 | | | $95 |
2022 | | | $95 |
2023 | | | $95 |
2024 | | | $95 |
Thereafter | | | $85 |
8. | Leases for Facilities and Equipment |
| | | Year Ended December 31, 2019 | |
Lease cost | | | |
Operating lease cost | | | $1,062 |
Variable lease cost | | | 94 |
Total lease cost | | | $1,156 |
Operating cash outflows from operating leases | | | $1,156 |
| | |
| | | December 31, 2019 | |
Weighted-average remaining lease term (years) | | | 5.0% |
Weighted-average discount rate | | | 8% |
Year Ending December 31, | | | |
2020 | | | $953 |
2021 | | | 1,182 |
2022 | | | 1,239 |
2023 | | | 1,272 |
2024 | | | 1,283 |
Thereafter | | | 105 |
Present value adjustment | | | (1,147) |
Total present value of lease payment | | | $4,887 |
9. | Convertible Notes Payable and Other Debt |
10. | Income Taxes |
| | | Year Ended December 31, | ||||
| | | 2018 | | | 2019 | |
| | | (in thousands) | ||||
Tax at the federal statutory rate | | | $(1,278) | | | $(8,501) |
Stock-based compensation | | | 39 | | | 118 |
Research and development credits | | | (444) | | | (65) |
Change in valuation allowance | | | 418 | | | 9,260 |
State taxes, net of federal benefits | | | (223) | | | (1,520) |
Fair value of contingent consideration | | | — | | | 1,211 |
Return to provision adjustment | | | 1,007 | | | — |
Other | | | — | | | (6) |
Total provision (benefit) for income taxes | | | $(481) | | | $497 |
| | | Year Ended December 31, | ||||
| | | 2018 | | | 2019 | |
| | | (in thousands) | ||||
Current: | | | | | ||
Federal | | | $— | | | $— |
State | | | — | | | 16 |
Total current tax expense | | | — | | | 16 |
Deferred: | | | | | ||
Federal | | | (405) | | | 405 |
State | | | (76) | | | 76 |
Total deferred tax expense | | | (481) | | | 481 |
Total provision for income taxes | | | $(481) | | | $497 |
| | | December 31, | ||||
| | | 2018 | | | 2019 | |
| | | (in thousands) | ||||
Deferred tax assets: | | | | | ||
Net operating loss carryforward | | | $5,271 | | | $14,589 |
Tax credit carryforwards | | | 861 | | | 926 |
Inventory | | | 112 | | | 120 |
Accrued expenses | | | 82 | | | 372 |
Deferred revenue | | | 499 | | | — |
Lease liability | | | — | | | 1,250 |
Other | | | 56 | | | 89 |
Total deferred tax assets | | | 6,881 | | | 17,346 |
Valuation allowance | | | (6,377) | | | (15,747) |
Deferred tax assets, net of valuation allowance | | | 504 | | | 1,599 |
Deferred tax liabilities: | | | | |||
Property, plant and equipment | | | (83) | | | (184) |
Right of use asset | | | — | | | (1,231) |
Intangible assets | | | (421) | | | (184) |
Total deferred tax liabilities | | | (504) | | | (1,599) |
Net deferred tax assets | | | $— | | | $— |
11. | Convertible Preferred Stock |
| | | Series A Convertible Preferred Stock | | | Series B Convertible Preferred Stock | | | Series C Convertible Preferred Stock | ||||||||||
| | | Shares | | | Amount | | | Shares | | | Amount | | | Shares | | | Amount | |
Balance – January 1, 2018 | | | — | | | $— | | | — | | | $— | | | — | | | $— |
Issuance of Series A convertible preferred stock | | | 7,764,831 | | | 37,436 | | | — | | | — | | | — | | | — |
Issuance of convertible preferred stock as merger | | | 1,018,941 | | | 4,744 | | | — | | | — | | | — | | | — |
Balance - December 31, 2018 | | | 8,783,772 | | | 42,180 | | | — | | | — | | | — | | | — |
Issuance of Series B convertible preferred stock | | | — | | | — | | | 2,190,546 | | | 13,150 | | | — | | | — |
Issuance of Series C convertible preferred stock | | | — | | | — | | | — | | | — | | | 6,638,261 | | | 54,840 |
Repurchase of Series A convertible preferred stock | | | (3,281) | | | (16) | | | — | | | — | | | — | | | — |
Balance - December 31, 2019 | | | 8,780,491 | | | $42,164 | | | 2,190,546 | | | $13,150 | | | 6,638,261 | | | $54,840 |
| | | December 31, 2019 | ||||||||||
| | | Shares Authorized | | | Shares Issued and Outstanding | | | Aggregate Liquidation Preference | | | Net Carrying Value | |
| | | | | | | (in thousands) | ||||||
Series A | | | 14,780,767 | | | 8,780,491 | | | $44,397 | | | $42,164 |
Series B | | | 4,082,031 | | | 2,190,546 | | | 15,000 | | | 13,150 |
Series C | | | 9,430,727 | | | 6,638,261 | | | 55,000 | | | 54,840 |
Total convertible preferred stock | | | 28,293,525 | | | 17,609,298 | | | $114,397 | | | $110,154 |
12. | Stockholders’ Deficit |
| | | As of December 31, | ||||
| | | 2018 | | | 2019 | |
Conversion of outstanding convertible preferred stock | | | 8,783,772 | | | 17,609,298 |
Conversion of contingent consideration | | | 1,463,758 | | | 1,463,758 |
Shares underlying outstanding equity awards | | | 2,553,440 | | | 4,848,949 |
Shares available for future equity award grants | | | 300,333 | | | 660,520 |
Exercise and conversion of preferred stock warrants | | | 159,866 | | | 615,054 |
Total | | | 13,261,169 | | | 25,197,579 |
13. | Stock-Based Compensation |
| | | Stock Options Outstanding | |||||||||||||
| | | Shares Available for Grant | | | Shares Subject to Options Outstanding | | | Weighted Average Exercise Price | | | Weighted Average Remaining Contractual Life (Years) | | | Average Intrinsic Value | |
Balance at January 1, 2019 | | | 292,245 | | | 1,380,554 | | | $1.05 | | | 9.8 | | | $588 |
Shares authorized | | | 2,823,293 | | | — | | | | | | | |||
Granted | | | (2,574,355) | | | 2,574,355 | | | $3.61 | | | | | ||
Exercised | | | — | | | (79,599) | | | $1.05 | | | | | ||
Canceled | | | 119,537 | | | (119,537) | | | $1.09 | | | | | ||
Balance at December 31, 2019 | | | 660,520 | | | 3,755,773 | | | $2.81 | | | 9.3 | | | $6,800 |
Vested at December 31, 2019 | | | | | 540,249 | | | $1.08 | | | 8.3 | | | $1,915 | |
| | | Year Ended December 31, | ||||
| | | 2018 | | | 2019 | |
Cost of precision oncology products revenue | | | $6 | | | $10 |
Sales and marketing expense | | | 30 | | | 95 |
Research and development expense | | | 90 | | | 94 |
General and administrative expense | | | 100 | | | 552 |
Total stock-based compensation expense | | | $226 | | | $751 |
| | | Year Ended December 31, | ||||
| | | 2018 | | | 2019 | |
Expected term (in years) | | | 6-7 | | | 6-7 |
Expected volatility | | | 80% | | | 80% |
Risk-free interest rate | | | 2.69% | | | 1.92% |
Expected dividend yield | | | — | | | — |
• | Expected Term. The expected term represents the period that the options granted are expected to be outstanding. After the adoption of ASU 2018-07 on January 1, 2019, the expected term of stock options issued to employees and nonemployee consultants is determined using the simplified method (based on the mid-point between the vesting date and the end of the contractual term) as ArcherDX has concluded that its stock option exercise history does not provide a reasonable basis upon which to estimate expected term. Prior to the adoption of ASU 2018-07, the expected term of stock options issued to employees was determined using the simplified method. |
• | Expected Volatility. Given that ArcherDX’s common stock is privately held, there is no active trading market for ArcherDX’s common stock. ArcherDX derived the expected volatility from the average historical volatilities over a period approximately equal to the expected term of comparable publicly traded companies within its peer group that were deemed to be representative of future stock price trends as ArcherDX has limited trading history for its common stock. |
• | Risk-Free Interest Rate. The risk-free interest rate is based on the U.S. Treasury rate, with maturities similar to the expected term of the stock options. |
• | Expected Dividend Yield. ArcherDX does not anticipate paying any dividends in the foreseeable future and, therefore, uses an expected dividend yield of zero. |
14. | Employee Benefit Plans |
15. | Net Loss Per Common Share |
| | | For The Year Ended December 31, | ||||
| | | 2018 | | | 2019 | |
Common shares under option plans | | | 2,593 | | | 5,194 |
Convertible preferred stock and warrants | | | 8,943 | | | 18,224 |
Total potential dilutive shares | | | 11,536 | | | 23,418 |
16. | Commitments and Contingencies |
17. | Subsequent Events |
18. | Return to Provision Adjustment |
| | | December 31, 2019 | | | June 30, 2020 | |
ASSETS | | | | | ||
Current assets: | | | | | ||
Cash and cash equivalents | | | $59,492 | | | $21,883 |
Accounts receivable, net | | | 15,354 | | | 20,828 |
Inventories | | | 6,002 | | | 11,435 |
Prepaid expenses and other current assets | | | 2,442 | | | 4,932 |
Total current assets | | | 83,290 | | | 59,078 |
Property and equipment, net | | | 10,811 | | | 16,591 |
Right-of-use assets, net | | | 4,813 | | | 5,879 |
Intangible assets, net | | | 780 | | | 583 |
Goodwill | | | 4,972 | | | 4,972 |
Other assets | | | 2,130 | | | 2,428 |
Total assets | | | $106,796 | | | $89,531 |
LIABILITIES, CONVERTIBLE PREFERRED STOCK, AND STOCKHOLDERS’ DEFICIT | | | | | ||
Current liabilities | | | | | ||
Accounts payable | | | $5,657 | | | $12,898 |
Accrued expenses | | | 2,025 | | | 6,698 |
Accrued compensation | | | 2,824 | | | 3,757 |
Lease liabilities | | | 577 | | | 1,382 |
Other current liabilities | | | 1,060 | | | 1,365 |
Deferred revenue | | | 6,234 | | | 6,297 |
Total current liabilities | | | 18,377 | | | 32,397 |
Long-term lease liabilities - less current portion | | | 4,310 | | | 4,918 |
Long-term debt, net | | | 28,572 | | | 41,717 |
Other long-term liabilities | | | 10,622 | | | 76,784 |
Total liabilities | | | 61,881 | | | 155,816 |
Commitments and contingencies (Note 15) | | | | | ||
Convertible preferred stock, $0.001 par value, 28,293,525 and 28,293,525 shares authorized, 17,609,298 and 18,393,441 shares issued and outstanding with aggregate liquidation preference of $114,397 and $118,362 as of December 31, 2019 and June 30, 2020, respectively | | | 110,154 | | | 115,347 |
Stockholders’ deficit: | | | | | ||
Common stock, $0.01 par value, 45,000,000 and 45,000,000 shares authorized, 6,454,355 and 6,627,583 shares issued and outstanding as of December 31, 2019 and June 30, 2020, respectively | | | 65 | | | 66 |
Additional paid-in capital | | | 3,939 | | | 5,329 |
Accumulated deficit | | | (69,243) | | | (187,027) |
Total stockholders’ deficit | | | $(65,239) | | | $(181,632) |
Total liabilities, convertible preferred stock, and stockholders’ deficit | | | $106,796 | | | $89,531 |
| | | Three months ended June 30, | | | Six months ended June 30, | |||||||
| | | 2019 | | | 2020 | | | 2019 | | | 2020 | |
Revenue | | | | | | | | | ||||
Precision oncology products | | | $5,095 | | | $8,404 | | | $9,475 | | | $15,410 |
Pharma development services | | | 5,704 | | | 7,934 | | | 10,773 | | | 15,718 |
Total revenue | | | 10,799 | | | 16,338 | | | 20,248 | | | 31,128 |
Operating expenses | | | | | | | | | ||||
Cost of precision oncology products | | | 1,525 | | | 3,138 | | | 2,593 | | | 5,451 |
Cost of pharma development services | | | 1,740 | | | 4,256 | | | 3,446 | | | 7,655 |
Sales and marketing | | | 2,900 | | | 4,861 | | | 5,544 | | | 10,185 |
Research and development | | | 5,006 | | | 22,116 | | | 9,301 | | | 35,853 |
General and administrative | | | 3,332 | | | 11,037 | | | 5,709 | | | 18,518 |
Contingent consideration | | | 2,790 | | | 39,140 | | | 5,506 | | | 39,105 |
Total operating expenses | | | 17,293 | | | 84,548 | | | 32,099 | | | 116,767 |
Loss from operations | | | (6,494) | | | (68,210) | | | 11,851 | | | 85,639 |
Interest expense, net | | | (769) | | | (1,105) | | | (769) | | | (1,998) |
Other expense, net | | | (211) | | | (29,152) | | | (219) | | | (30,147) |
Loss before income taxes | | | (7,474) | | | (98,467) | | | (12,839) | | | (117,784) |
Income tax expense | | | 6 | | | — | | | 7 | | | — |
Net loss and comprehensive loss | | | $(7,480) | | | $(98,467) | | | $(12,846) | | | $(117,784) |
Basic and diluted loss per common share | | | $(1.17) | | | $(15.04) | | | $(2.01) | | | $(18.09) |
Basic and diluted weighted-average common shares outstanding | | | 6,376,851 | | | 6,548,624 | | | 6,376,731 | | | 6,510,736 |
| | | Convertible Preferred Stock | | | Common Stock | | | Additional Paid-In Capital | | | Accumulated Deficit | | | Total Stockholders’ Deficit | |||||||
| | | Shares | | | Amount | | | Shares | | | Amount | | | | | | | ||||
Balance – April 1, 2019 | | | 8,781,124 | | | $42,167 | | | 6,376,642 | | | $64 | | | $3,175 | | | $(33,631) | | | $(30,392) |
Issuance of Series B convertible preferred stock for cash, net of issuance costs of $1,844 | | | 2,190,558 | | | 13,155 | | | — | | | — | | | — | | | — | | | — |
Proceeds from exercise of stock options | | | — | | | — | | | 178 | | | — | | | — | | | — | | | — |
Repurchase of Series A preferred stock | | | (567) | | | (3) | | | — | | | — | | | — | | | — | | | — |
Stock-based compensation expense | | | — | | | — | | | — | | | — | | | 70 | | | — | | | 70 |
Net loss | | | — | | | — | | | — | | | — | | | — | | | (8,230) | | | (8,230) |
Balance – June 30, 2019 | | | 10,971,115 | | | $55,319 | | | 6,376,820 | | | $64 | | | $3,245 | | | $(41,861) | | | $(38,552) |
Balance – April 1, 2020 | | | 18,393,441 | | | $115,347 | | | 6,508,640 | | | $65 | | | $4,374 | | | $(88,560) | | | $(84,121) |
Stock-based compensation expense | | | — | | | — | | | — | | | — | | | 661 | | | — | | | 661 |
Proceeds from exercise of stock options | | | — | | | — | | | 118,943 | | | 1 | | | 294 | | | — | | | 295 |
Net loss | | | — | | | — | | | — | | | — | | | — | | | (98,467) | | | (98,467) |
Balance – June 30, 2020 | | | 18,393,441 | | | $115,347 | | | 6,627,583 | | | $66 | | | $5,329 | | | $(187,027) | | | $(181,632) |
| | | Convertible Preferred Stock | | | Common Stock | | | Additional Paid-In Capital | | | Accumulated Deficit | | | Total Stockholders’ Deficit | |||||||
| | | Shares | | | Amount | | | Shares | | | Amount | | | | | | | ||||
Balance – January 1, 2019 | | | 8,783,772 | | | $42,180 | | | 6,375,767 | | | $64 | | | $3,106 | | | $(28,265) | | | $(25,095) |
Issuance of Series B convertible preferred stock for cash, net of issuance costs of $1,844 | | | 2,190,558 | | | 13,155 | | | | | | | | | | | |||||
Proceeds from exercise of stock options | | | — | | | — | | | 1,950 | | | — | | | 2 | | | — | | | 2 |
Repurchase of restricted stock | | | — | | | — | | | (897) | | | — | | | (1) | | | — | | | (1) |
Repurchase of Series A preferred stock | | | (3,215) | | | (16) | | | — | | | — | | | — | | | — | | | — |
Stock-based compensation expense | | | — | | | — | | | — | | | — | | | 138 | | | — | | | 138 |
Net loss | | | — | | | — | | | — | | | — | | | — | | | (13,596) | | | (13,596) |
Balance – June 30, 2019 | | | 10,971,115 | | | $55,319 | | | 6,376,820 | | | $64 | | | $3,245 | | | $(41,861) | | | $(38,552) |
Balance – January 1, 2020 | | | 17,609,298 | | | $110,154 | | | 6,454,355 | | | $65 | | | $3,939 | | | $(69,243) | | | $(65,239) |
Issuance of Series A convertible preferred stock for Baby Genes earnout | | | 624,277 | | | 4,142 | | | — | | | — | | | — | | | — | | | $— |
Proceeds from Warrant Exercise of Series A convertible preferred stock | | | 159,866 | | | 1,051 | | | — | | | — | | | — | | | — | | | — |
Stock-based compensation expense | | | — | | | — | | | — | | | — | | | 1,039 | | | — | | | 1,039 |
Proceeds from exercise of stock options | | | — | | | — | | | 173,228 | | | 1 | | | 351 | | | — | | | 352 |
Net loss | | | — | | | — | | | — | | | — | | | — | | | (117,784) | | | (117,784) |
Balance – June 30, 2020 | | | 18,393,441 | | | $115,347 | | | 6,627,583 | | | $66 | | | $5,329 | | | $(187,027) | | | $(181,632) |
| | | Six months ended June 30, | ||||
| | | 2019 | | | 2020 | |
Cash flows from operating activities | | | | | ||
Net loss | | | $(12,846) | | | $(117,784) |
Adjustments to reconcile net loss to net cash used in operating activities | | | | | ||
Depreciation and amortization | | | 1,021 | | | 2,080 |
Amortization of debt issuance costs | | | 69 | | | 273 |
Bad debt expense (recovery) | | | (26) | | | (6) |
Stock-based compensation expense | | | 138 | | | 1,039 |
Change in fair value of contingent consideration | | | 5,506 | | | 39,105 |
Change in fair value of convertible preferred stock warrants | | | 211 | | | 30,130 |
Changes in assets and liabilities | | | | | ||
Accounts receivable | | | (2,511) | | | (5,468) |
Inventories | | | (1,729) | | | (5,432) |
Prepaid expenses and other assets | | | (468) | | | (2,408) |
Accounts payable and accrued liabilities | | | 1,751 | | | 12,505 |
Lease liabilities | | | (15) | | | 204 |
Deferred revenue | | | (1,707) | | | 63 |
Net cash used in operating activities | | | (10,606) | | | (45,699) |
Cash flows from investing activities | | | | | ||
Purchase of property and equipment | | | (1,349) | | | (7,254) |
Net cash used in investing activities | | | (1,349) | | | (7,254) |
Cash flows from financing activities | | | | | ||
Proceeds from issuance of convertible preferred stock, net of issuance costs | | | 13,156 | | | — |
Proceeds from borrowing under credit facility, net of issuance costs | | | 29,125 | | | 15,000 |
Payment of contingent consideration | | | — | | | (10) |
Proceeds from exercise of stock options | | | 2 | | | 352 |
Repurchase of restricted stock | | | (1) | | | — |
Proceeds from exercise of Series A preferred stock warrants | | | — | | | 2 |
Repurchase of Series A convertible preferred stock | | | (16) | | | — |
Net cash provided by (used in) financing activities | | | 42,266 | | | 15,344 |
Net decrease in cash and cash equivalents | | | 30,311 | | | (37,609) |
Cash and cash equivalents at beginning of period | | | 9,474 | | | 59,492 |
Cash and cash equivalents at end of period | | | $39,785 | | | $21,883 |
Non-cash financing activities: | | | | | ||
Reclassification of Series A preferred stock warrants from liabilities to convertible preferred stock upon exercise | | | $— | | | $1,049 |
Issuance of warrant with credit facility | | | $750 | | | $— |
Issuance of Series A preferred stock for contingent consideration | | | $— | | | $4,142 |
Purchases of property and equipment included in accounts payable and accrued liabilities | | | $— | | | $390 |
1. | Nature of Business |
2. | Summary of Significant Accounting Policies |
| | | Three Months Ended June 30, | | | Six Months Ended June 30, | |||||||
| | | 2019 | | | 2020 | | | 2019 | | | 2020 | |
United States | | | $4,743 | | | $4,738 | | | $8,766 | | | $9,552 |
International | | | 6,056 | | | 11,600 | | | 11,482 | | | 21,576 |
Total revenue | | | $10,799 | | | $16,338 | | | $20,248 | | | $31,128 |
| | | Revenue for the Three Months Ended June 30, | | | Revenue for the Six Months Ended June 30, | | | Accounts Receivable as of | ||||||||||
| | | 2019 | | | 2020 | | | 2019 | | | 2020 | | | December 31, 2019 | | | June 30, 2020 | |
Customer A(1) | | | *% | | | *% | | | 10% | | | *% | | | *% | | | *% |
Customer B(1) | | | 40% | | | 38% | | | 39% | | | 35% | | | 41% | | | 26% |
Customer C(1) | | | *% | | | *% | | | *% | | | *% | | | *% | | | 18% |
Customer D(1) | | | *% | | | 10% | | | *% | | | 10% | | | *% | | | *% |
(1) | The country of headquarters of Customer A is the US, the country of headquarters of Customer B and D is Germany, and the country of headquarters for Customer C is the UK |
* | less than 10% |
Property and Equipment | | | Estimated Useful Life |
Furniture and fixtures | | | 7 years |
Manufacturing and lab equipment | | | 5 years |
Computer equipment | | | 3 years |
Leasehold improvements | | | Lesser of estimated useful life or remaining lease term |
3. | Business Combination |
4. | Fair Value Measurements |
| | | Level 1 | | | Level 2 | | | Level 3 | |
December 31, 2019 | | | | | | | |||
Assets: | | | | | | | |||
Money market accounts | | | $43,154 | | | — | | | — |
Liabilities: | | | | | | | |||
Warrant liabilities | | | — | | | — | | | $(2,295) |
Contingent consideration | | | $— | | | $— | | | $(8,327) |
| | | | | | | ||||
June 30, 2020 | | | | | | | |||
Assets: | | | | | | | |||
Money market accounts | | | $18,226 | | | $— | | | $— |
Liabilities: | | | | | | | |||
Warrant liabilities | | | $— | | | $— | | | $(33,504) |
Contingent consideration | | | $— | | | $— | | | $(43,280) |
| | | Convertible Preferred Stock Warrants | | | Contingent Consideration | |
Balance as of December 31, 2019 | | | $2,295 | | | $8,327 |
Issuance of Series A preferred stock for contingent consideration | | | — | | | (4,142) |
Issuance of Series B warrants | | | 2,128 | | | |
Cash payment for contingent consideration | | | — | | | (10) |
Exercise of preferred stock warrants | | | (1,049) | | | — |
Change in fair value | | | 30,130 | | | 39,105 |
Balance as of June 30, 2020 | | | $33,504 | | | $43,280 |
5. | Inventories |
| | | December 31, 2019 | | | June 30, 2020 | |
Raw materials | | | $3,640 | | | $6,071 |
Work in process | | | 1,412 | | | 4,618 |
Finished goods | | | 950 | | | 746 |
Total inventories | | | $6,002 | | | $11,435 |
6. | Property and Equipment |
| | | December 31, 2019 | | | June 30, 2020 | |
Manufacturing and lab equipment | | | $12,125 | | | $16,989 |
Office equipment | | | 1,490 | | | 2,151 |
Leasehold improvements | | | 421 | | | 1,930 |
Construction in progress | | | 405 | | | 924 |
Property and equipment, gross | | | 14,441 | | | 21,994 |
Less accumulated depreciation | | | (3,630) | | | (5,403) |
Property and equipment, net | | | $10,811 | | | $16,591 |
7. | Leases for Facilities and Equipment |
| | | Three Months Ended June 30, 2020 | | | Six Months Ended June 30, 2020 | |
Operating Lease cost | | | | | ||
Operating lease cost | | | $510 | | | $950 |
Variable lease cost | | | 68 | | | 256 |
Operating lease expense | | | $578 | | | $1,206 |
Short-term lease rent expense | | | $8 | | | $55 |
Total lease cost | | | $586 | | | $1,261 |
Operating cash outflows from operating leases | | | $406 | | | $743 |
| | | June 30, 2020 | |
Weighted-average remaining lease term (years) | | | 5.1 |
Weighted-average discount rate | | | 8% |
Year Ending December 31, | | | |
Remaining in 2020 | | | $938 |
2021 | | | 1,770 |
2022 | | | 1,655 |
2023 | | | 1,735 |
2024 | | | 1,757 |
Thereafter | | | 1,385 |
Present value adjustment | | | (1,773) |
Tenant improvement allowance receivable | | | (1,167) |
Total present value of lease payments | | | $6,300 |
8. | Convertible Notes Payable and Other Debt |
9. | Convertible Preferred Stock |
| | | Series A Convertible Preferred Stock | | | Series B Convertible Preferred stock | | | Series C Convertible Preferred stock | ||||||||||
| | | Shares | | | Amount | | | Shares | | | Amount | | | Shares | | | Amount | |
Balance - January 1, 2019 | | | 8,783,772 | | | $42,180 | | | — | | | $— | | | — | | | $— |
Issuance of Series B convertible preferred stock for cash, net of issuance costs of $1,850 | | | | | | | 2,190,546 | | | 13,150 | | | | | ||||
Repurchase of Series A convertible preferred stock | | | (3,281) | | | (16) | | | — | | | — | | | — | | | — |
Balance - June 30, 2019 | | | 8,780,491 | | | $42,164 | | | 2,190,546 | | | $13,150 | | | — | | | $— |
Balance - January 1, 2020 | | | 8,780,491 | | | 42,164 | | | 2,190,546 | | | 13,150 | | | 6,638,261 | | | 54,840 |
Exercise of Series A convertible preferred stock warrants | | | 159,866 | | | 1,051 | | | — | | | — | | | — | | | — |
Issuance of Series A convertible preferred stock for contingent consideration | | | 624,277 | | | 4,142 | | | — | | | — | | | — | | | — |
Balance - June 30, 2020 | | | 9,564,634 | | | $47,357 | | | 2,190,546 | | | $13,150 | | | 6,638,261 | | | $54,840 |
| | | June 30, 2020 | ||||||||||
| | | Shares Authorized | | | Shares Issued and Outstanding | | | Aggregate Liquidation Preference | | | Net Carrying Value | |
| | | | | | | (in thousands) | ||||||
Series A | | | 14,780,767 | | | 9,564,634 | | | $48,362 | | | $47,357 |
Series B | | | 4,082,031 | | | 2,190,546 | | | 15,000 | | | 13,150 |
Series C | | | 9,430,727 | | | 6,638,261 | | | 55,000 | | | 54,840 |
Total convertible preferred stock | | | 28,293,525 | | | 18,393,441 | | | $118,362 | | | $115,347 |
| | | Equity Upon Exercise | | | Exercise Price | | | Expiration Date | | | December 31, | | | June 30, | |
| | | 2019 | | | 2020 | ||||||||||
Warrants Issued in 2018 | | | Series A Preferred | | | $0.01 | | | 3/5/28 | | | 159,866 | | | — |
Warrants Issued in 2019 | | | Series B Preferred | | | 6.85 | | | 5/10/26 | | | 455,188 | | | 455,188 |
Warrants Issued in 2020 | | | Series B Preferred | | | 6.85 | | | 4/27/27 | | | — | | | 227,594 |
Total Warrants | | | | | | | | | 615,054 | | | 682,782 |
| | | Warrants | | | Weighted Average Stock Price | | | Weighted Average Remaining Contractual Life (in Years) | |
Balance - January 1, 2020 | | | 615,054 | | | $5.07 | | | 6.8 |
Issued | | | 227,594 | | | 6.85 | | | 6.8 |
Exercised | | | (159,866) | | | 0.01 | | | |
Balance - June 30, 2020 | | | 682,782 | | | $6.85 | | | 6.2 |
10. | Stockholders’ Deficit |
| | | As of December 31, | | | As of June 30, | |
| | | 2019 | | | 2020 | |
Conversion of outstanding convertible preferred stock | | | 17,609,298 | | | 18,393,441 |
Conversion of convertible preferred stock issuable for contingent consideration | | | 1,463,758 | | | 837,464 |
Shares underlying outstanding equity awards | | | 4,848,949 | | | 3,858,108 |
Shares available for future equity award grants | | | 660,520 | | | 385,987 |
Exercise and conversion of convertible preferred stock warrants | | | 615,054 | | | 682,782 |
Total | | | 25,197,579 | | | 24,157,782 |
11. | Stock-Based Compensation |
| | | | | Stock Options Outstanding | |||||||||||
| | | Shares Available for Grant | | | Shares Subject to Options Outstanding | | | Weighted Average Exercise Price | | | Weighted Average Remaining Contractual Life (Years) | | | Average Intrinsic Value | |
Balance at December 31, 2019 | | | 660,520 | | | 3,755,773 | | | $2.81 | | | 9.3 | | | $6,800 |
Shares authorized | | | — | | | — | | | | | | | |||
Granted | | | (852,312) | | | 852,312 | | | $4.62 | | | | | ||
Exercised | | | | | (172,198) | | | $1.99 | | | | | |||
Canceled | | | 577,779 | | | (577,779) | | | $3.24 | | | | | ||
Balance at June 30, 2020 | | | 385,987 | | | 3,858,108 | | | $3.18 | | | 9.1 | | | $5,388 |
Vested at June 30, 2020 | | | | | 811,379 | | | $1.55 | | | 8.5 | | | $2,409 | |
| | | Three Months Ended June 30, | | | Six Months Ended June 30, | |||||||
| | | 2019 | | | 2020 | | | 2019 | | | 2020 | |
Cost of precision oncology products revenue | | | $1 | | | $— | | | $1 | | | $— |
Sales and marketing expense | | | 12 | | | 41 | | | 24 | | | 52 |
Research and development expense | | | 14 | | | 55 | | | 29 | | | 107 |
General and administrative expense | | | 43 | | | 565 | | | 84 | | | 880 |
Total stock-based compensation expense | | | $70 | | | $661 | | | $138 | | | $1,039 |
| | | Six Months Ended June 30, | ||||
| | | 2019 | | | 2020 | |
Expected term (in years) | | | 6-7 | | | 6-7 |
Expected volatility | | | 80% | | | 80% |
Risk-free interest rate | | | 1.92% | | | 1.02% |
Expected dividend yield | | | — | | | — |
• | Expected Term. The expected term represents the period that the options granted are expected to be outstanding. The expected term of stock options issued to employees and nonemployee consultants is determined based on the mid-point between the vesting date and the end of the contractual term, as ArcherDX concluded that its stock option exercise history does not provide a reasonable basis upon which to estimate expected term. |
• | Expected Volatility. Given that ArcherDX’s common stock is privately held, there is no active trading market for ArcherDX’s common stock. ArcherDX derived the expected volatility from the average historical volatilities over a period approximately equal to the expected term of comparable publicly traded companies within its peer group that were deemed to be representative of future stock price trends as ArcherDX has limited trading history for its common stock. |
• | Risk-Free Interest Rate. The risk-free interest rate is based on the U.S. Treasury rate, with maturities similar to the expected term of the stock options. |
• | Expected Dividend Yield. ArcherDX does not anticipate paying any dividends in the foreseeable future and, therefore, uses an expected dividend yield of zero. |
12. | Net Loss Per Common Share |
| | | Three Months Ended June 30, | | | Six Months Ended June 30, | |||||||
| | | 2019 | | | 2020 | | | 2019 | | | 2020 | |
Numerator: | | | | | | | | | ||||
Net loss | | | $(7,480) | | | $(98,467) | | | $(12,846) | | | $(117,784) |
| | | | | | | | | |||||
Denominator: | | | | | | | | | ||||
Denominator for basic EPS - Weighted average shares outstanding | | | 6,376,851 | | | 6,548,624 | | | 6,376,731 | | | 6,510,736 |
Dilutive securities | | | | | | | | | ||||
Denominator for diluted EPS - Weighted average shares outstanding | | | 6,376,851 | | | 6,548,624 | | | 6,376,731 | | | 6,510,736 |
| | | | | | | | | |||||
Earnings per share: | | | | | | | | | ||||
Basic and diluted EPS | | | $(1.17) | | | $(15.04) | | | $(2.01) | | | $(18.09) |
| | | Six months ended June 30, | ||||
| | | 2019 | | | 2020 | |
Conversion of outstanding convertible preferred stock | | | 10,971,115 | | | 18,393,441 |
Shares of underlying outstanding equity awards | | | 1,557,775 | | | 3,858,108 |
Exercise and conversion of convertible preferred stock warrants | | | 615,054 | | | 682,782 |
Total | | | 13,143,944 | | | 22,934,331 |
13. | Commitments and Contingencies |
14. | Subsequent Events |
| | | |||||
| | | | | |||
| | | | | |||
| | | |||||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | |||||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | |||||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | |||||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | |||||
| | | | | |||
| | | | | |||
| | | | | |||
| | | |||||
| | | | | |||
| | | | | |||
| | | | | |||
| | | |||||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | |||||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | | |||
| | | | |
Exhibits | | | |
Exhibit A | | | - Irrevocable Written Consent of Stockholders |
Exhibit B-1 | | | - Mergers and Acquisitions Insurance Binder |
Exhibit B-2 | | | - Confirmation of Coverage |
Exhibit C | | | - Milestones |
Exhibit D | | | - Certificate of Reverse Merger |
Exhibit E | | | - Certificate of Forward Merger |
Exhibit F | | | - Third Party Consents and Notices |
| | | ||
Schedules | | | |
Schedule 1 | | | - Holders Executing Irrevocable Written Consent of Stockholders |
Term | | | Section |
“ACS” | | | Section 3.1(c) |
“Agreement” | | | Preamble |
“Agreement Date” | | | Preamble |
“Allocation Schedule” | | | Section 2.11 |
“Antitrust Laws” | | | Section 3.4 |
“Assets” | | | Section 3.13 |
“Certificate of Forward Merger” | | | Section 2.1 |
“Certificate of Reverse Merger” | | | Section 2.1 |
“Certificates of Merger” | | | Section 2.1 |
“Closing” | | | Section 2.2 |
“Closing Date” | | | Section 2.2 |
“Company” | | | Preamble |
“Company Charter Documents” | | | Section 3.1(d) |
“Company Group” | | | Section 3.1(c) |
“Company Option” | | | Section 1.1 |
“Company Stockholder Approvals” | | | Section 5.3(b) |
“Competing Transaction” | | | Section 5.11 |
“Conflict” | | | Section 3.2(d) |
“Consultant” | | | Section 3.1(b) |
“Continuing Employees” | | | Section 5.9 |
“Contracting Parties” | | | Section 9.16 |
“Current Consultant” | | | Section 3.1(b) |
“Current Employee” | | | Section 3.1(b) |
“DGCL” | | | Recitals |
“DLLCA” | | | Recitals |
“D&O Indemnified Persons” | | | Section 5.12 |
“D&O Tail Insurance” | | | Section 5.12 |
“Effective Time” | | | Section 2.1 |
“Eligible Post-Closing Adjustment” | | | Section 2.19(c)(ii) |
“Employee” | | | Section 3.1(b) |
“ERISA Affiliate” | | | Section 3.10(c) |
“Estimated Balance Sheet” | | | Section 2.11(e) |
“Exchange Act” | | | Section 4.4(a) |
“Exchange Agent” | | | Section 2.15(a) |
“Exchange Agreement” | | | Section 2.15(a) |
Term | | | Section |
“Expense Fund” | | | Section 8.4(g) |
“Final Calculation” | | | Section 2.19(a) |
“Financial Statements” | | | Section 3.5 |
“Forward Merger” | | | Recitals |
“Holders’ Representative” | | | Preamble |
“Inbound IP Contracts” | | | Section 3.14(c) |
“Initial Resolution Period” | | | Section 2.19(a) |
“Interim Balance Sheet” | | | Section 3.5 |
“Interim Balance Sheet Date” | | | Section 3.5 |
“IP Contracts” | | | Section 3.14(c) |
“Letter of Transmittal” | | | Section 2.15(b) |
“Material Contract” | | | Section 3.12(b) |
“Merger Sub A” | | | Preamble |
“Merger Sub B” | | | Preamble |
“Merger Subs” | | | Preamble |
“Mergers” | | | Recitals |
“Multiemployer Plan” | | | Section 3.10(c) |
“Non-Party Affiliates” | | | Section 9.15 |
“NYSE” | | | Section 5.15 |
“Objection Notice” | | | Section 2.19(a) |
“Objection Period” | | | Section 2.19(a) |
“Offset Certificate” | | | Section 8.3(b) |
“Offset Right” | | | Section 8.2 |
“Outbound IP Contracts” | | | Section 3.14(c) |
“Outside Date” | | | Section 7.1(b) |
“Parent” | | | Preamble |
“Parent Adverse Recommendation Change” | | | Section 5.4(c) |
“Parent Plan” | | | Section 5.10(a) |
“Parent Record Date” | | | Section 5.2(a) |
“Parent Stockholder Approvals” | | | Section 5.4(a) |
“Parent Stockholder Meeting” | | | Section 5.4(a) |
“Parent’s SEC Documents” | | | Section 4.4(a) |
“Parties” | | | Preamble |
“Payment Programs” | | | Section 3.8(e) |
“Payoff Amount” | | | Section 2.12(a) |
“PCM Revenue” | | | Exhibit C |
“Post-Closing Adjustment” | | | Section 2.19(c)(i) |
“Proxy Statement/Prospectus” | | | Section 5.3(a) |
“Requisite Stockholder Approval” | | | Section 3.2(b) |
“Reverse Merger” | | | Recitals |
“Reviewing Party” | | | Section 2.19(b) |
“Shrink Wrap Licenses” | | | Section 3.14(a)(i) |
“Stated Damages” | | | Section 8.3(b) |
“Support Agreement” | | | Recitals |
“Surviving Company” | | | Section 2.1 |
“Third Party Software” | | | Section 3.14(c) |
“Title IV Plan” | | | Section 3.10(c) |
“Transfer Taxes” | | | Section 5.9(e) |
“Written Consent” | | | Recitals |
PARENT: | | | INVITAE CORPORATION | |||
| | | | | |||
| | | By: | | | /s/ Sean George | |
| | | Name: | | | Sean George | |
| | | Title: | | | Chief Executive Officer | |
MERGER SUB A: | | | APOLLO MERGER SUB A INC. | |||
| | | | | |||
| | | By: | | | /s/ Tom Brida | |
| | | Name: | | | Tom Brida | |
| | | Title: | | | President | |
MERGER SUB B: | | | APOLLO MERGER SUB B LLC | |||
| | | | | |||
| | | By: | | | /s/ Tom Brida | |
| | | Name: | | | Tom Brida | |
| | | Title: | | | President | |
COMPANY: | | | ARCHERDX, INC. | |||
| | | | | |||
| | | By: | | | /s/ Jason Myers | |
| | | Name: | | | Jason Myers | |
| | | Title: | | | CEO | |
HOLDERS’ REPRESENTATIVE: | | | KYLE LEFKOFF, solely in his capacity as the Holders’ Representative | |||
| | | | | |||
| | | By: | | | /s/ Kyle LefKoff | |
| | | Name: | | | Kyle LefKoff | |
 | | | 767 Fifth Avenue New York, NY 10153 T 212.287.3200 F 212.287.3201 pwpartners.com |
Item 20. | Indemnification of Directors and Officers |
Item 21. | Exhibits and Financial Statement Schedules |
Exhibit | | | Description |
| | | Agreement and Plan of Merger and Plan of Reorganization, dated as of June 21, 2020, by and among Invitae Corporation, Apollo Merger Sub A Inc., Apollo Merger Sub B LLC, ArcherDX, Inc. and Kyle Lefkoff, solely in his capacity as holders’ representative (incorporated by reference to Exhibit 2.1 to Invitae Corporation’s Current Report on Form 8-K filed on June 24, 2020). | |
| | | ||
| | | Restated Certificate of Incorporation of Invitae Corporation (incorporated by reference to Exhibit 3.1 to Invitae Corporation's Current Report on Form 8-K filed February 23, 2015). | |
| | | ||
| | | Amended and Restated Bylaws of Invitae Corporation (incorporated by reference to Exhibit 3.2 to Invitae Corporation’s Current Report on Form 8-K filed February 23, 2015). | |
| | | ||
| | | Opinion of Pillsbury Winthrop Shaw Pittman LLP | |
| | | ||
| | | Consent of Pillsbury Winthrop Shaw Pittman LLP (included in the opinion filed as Exhibit 5.1). | |
| | | ||
| | | Consent of Ernst & Young LLP, independent registered public accounting firm of Invitae Corporation | |
| | | ||
| | | Consent of Ernst & Young LLP, independent registered public accounting firm of ArcherDX, Inc. | |
| | | ||
| | | Power of Attorney | |
| | | ||
| | | Form of Proxy Card | |
| | | ||
| | | Consent of Perella Weinberg Partners LP |
† | Schedules and certain exhibits have been omitted pursuant to Item 601(b)(2) of Regulation S-K. Invitae hereby undertakes to furnish supplementally copies of any of the omitted schedules and exhibits upon request by the U.S. Securities and Exchange Commission. |
* | Previously filed in the signature page to Form S-4. |
Item 22. | Undertakings |
| | | INVITAE CORPORATION | ||||
| | | | | |||
| | | By: | | | /s/ Sean E. George, Ph.D. | |
| | | | | Name: Sean E. George, Ph.D. | ||
| | | | | Title: President and Chief Executive Officer | ||
Signature | | | Title | | | Date |
| | | | | |||
/s/ Sean E. George, Ph.D. | | | President and Chief Executive Officer (Principal Executive Officer) and Director | | | August 25, 2020 |
Sean E. George, Ph.D. | | |||||
| | | | | |||
/s/ Shelly D. Guyer | | | Chief Financial Officer (Principal Financial Officer) | | | August 25, 2020 |
Shelly D. Guyer | | |||||
| | | | | |||
/s/ Robert F. Werner | | | Chief Accounting Officer (Principal Accounting Officer) | | | August 25, 2020 |
Robert F. Werner | | |||||
| | | | | |||
* | | | Director | | | August 25, 2020 |
Eric Aguiar, M.D. | | |||||
| | | | | |||
* | | | Director | | | August 25, 2020 |
Geoffrey S. Crouse | | |||||
| | | | | |||
* | | | Director | | | August 25, 2020 |
Christine M. Gorjanc | | |||||
| | | | | |||
* | | | Director | | | August 25, 2020 |
Chitra Nayak | |
* By: | | | /s/ Sean E. George, Ph.D. | | | |
| | | Sean E. George, Ph.D. Attorney-in-fact | | |

