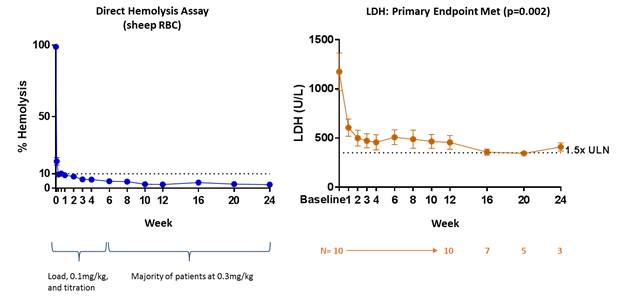
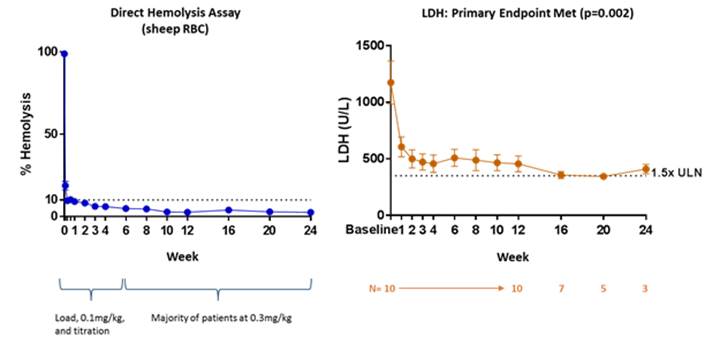
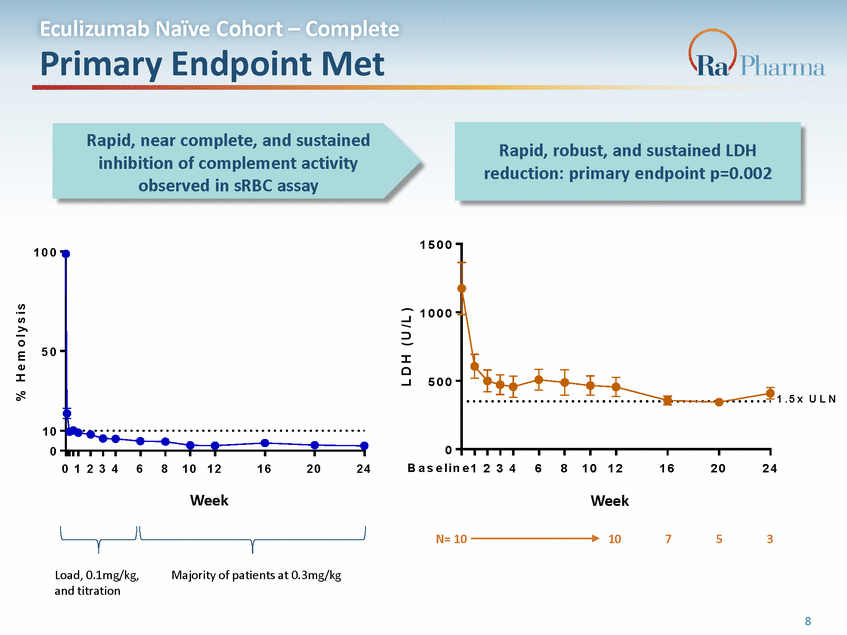
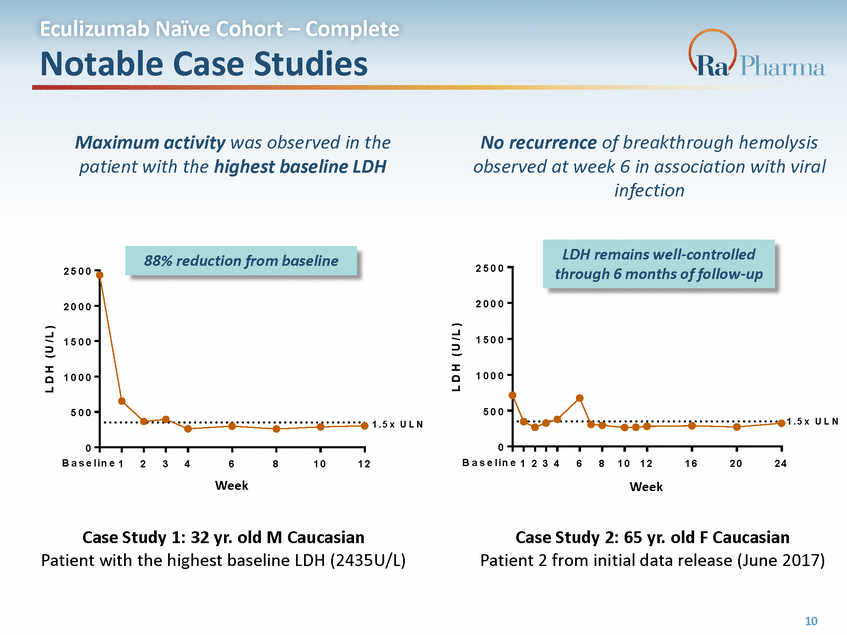
L
M%==P]7IT%6^TADXPB88P![LK28LP2$.OO8:H+I2S"5=PQZKS!*LSRHKPR1XP
M")]S2X^T2+.TNE:Q1Z]T3P]U2A?L3J>TWFYT1Z.P$S/U3 >U4J>T"M?T4GP3B?UO :Q-$!P37-U"!.Q5D?U=KVT4_?T3W-T5I/T6)?^[%13<0S7\$O+
MK#KS[$[?]2GWXN55;F/@6O]$@A6 C(P X 9A%$ 7@<'F'>@ 54P V(@1W[
ML[2XD13, 2\ "$F@0Y8Z0% SP \ P0F0 AD !/$QX?4DID>I#$4H_\=
M P@; P@C]00?0[O"I_*"7N%Z,D8H1%>]"!/!SYS^Z66RZ]XE@)QBYFR"@&!;F F
MA((GB.73^"8;T,'-D"(9C($3X(!)LS5;&S54#WA+PW3!;C55/V^.@S1,Z_@"
MHS '"[%&8ZQ(MS.0%[6/@_!:3SE64SD-CW63&S%'\WB.1_D[P[51'[E0#_E:
M1S!.1_%7_W@'5[4%I[G%RG0%!SE-0_@Y$W&3O[F6[[299VQT52X.M88*- *\, B18$4*#84&-)3V $2O$!-X8 :Z 'Z
M9H/Y2L$3)% '$.(/[( @'\ !&$!FC46\J( 7@,;EKD)C@-Z-!F5M#WIMXWG)
M(OJ$S^G^;E]D\L*"67D"*0 &PC2&G[J/XR:Z1%JFGQ+JGHW5>5(C:&&"0.?&
M$ A!!@'''!@'\NWR)I W*IP[NI=W>3=GIBVG',D1I>&!("4S[A;F$Q@!?=^
M#-QW"(R?->MW P "^C+:-TI)X>S-+CAEA=QF0>YE;=U.YPQR+Y@V\YCN<"-SAUG*MUFQMX26.Y1M=UG/\X
M"O_\T)^YFVLUT<UW?YT2O]EDN]O4K]5]\YD:]TU9_]TE?^?:_7=-"C\,YS
MM-BOO=Q3]8]+><5R
M0X]O_>+O^JY_<257'T#UU A:@P ,TK2%' :5# >Z28BG>P ODP P8
M01MTK?ER@A0@00^\P O$ ^81V57
M@]Z?]--7+"TD?'-G?,;O66@TPA<,:"-H0BK42H30Z3R' 1#\@!J-@1F805_YP3I:6KF? KK? BKX KJ?.T"8VF0*4!^#>_KL
M4:B0#QZ'>.3,B7/&C)DR9<1 >?+^Q$@1(D1VR*@Q8L2'#B(Z0'"P@"4#+)=$
MU9(92Q:L6+!HY:0EC58N:3Y_ A4JC:A/H4=_%B6JU.A2H+J60FU*-%K5IE6I
MYK):-=JO7,=HQ0I+TU.G*R@:/!#!(\D1*:$XD;+5S1RY;KM(77*398B0)E^^
M' F2! H;.G7HP($C9HR3(C=FO !*5-?D:-*P/KV\.2EE;M-\0L4<5>E/T=*@
M;L:*^O+4TZ6#.E5Z>G+GU%11RXXZM'.NT[='_UYJ&S/GU:5]$^66'/93IL.+
M)U3AMXM#/X[[N''KN^;2Y
M"1_NOO[^L>)[KK7K*%NJ-
MZ<3#2%0P@004=B"BB"B@2#%%.\1X0PTIH"CB@@MD>(*--G!T XDD:' !@A!X
MV"&&$41XX( %-##!!!16P$(2F6IYY9=:9!&+%F-RJ0; _[A*,$$ ?3HP%YUH
M@>67FC)\Q95.(%F""2""8"*(+D+QQ!9/8*E)+"MU&C":6;32:A9CXV_]:SZ:;-J
MCCF&T&-BH8D62IA *ZVUCD BC49"L26;;LHQIQM=2,GD#C ^^N&'*M[(U%8T
MT""CUQXBVT"#^J:I3^21BPJO09)Q&T]EDDU&V65J;CMNY)!=KKFHVAP\V>;A
M0FZ9&^E0HT_DEH=C$#J:@7;9Z*%17GIDHI\>V6BH*?..:O5P%CEKE(<"ZKQX
MS>6I2Z/C(]GIG%/^*A=""Y5E0T\^Z4021S1@X(01/CI1Q17)4$.--Z1P8H8)
M+K@!B2;^ 10EVBTI*H:
M".6#[ZN?=M(IEENO&EF_111675
M/EAM%=8]\)"UCCGRF$/_73,:PU=@02(#8A') QU@%K,,L( N*044%+3+\*2
M$[#XR3O4T!K/ZG,UI>UL@P":4+V,88Q\90A22R"! 0P 1WLX A-<$,H"K8+
MNJR/%YS0!!? <(0JN($.<9!#''3EOQ[T@ 8OL$ "*L!!)4J#9DMT(F6:*+(H
M/I&*3HS^H@:KJ)LE3M&*6:P9%I\(1NB(L3X_\Z(2UZ.R!^4B;&:K8M8*=)EZ
MY4(L;G/;*VK1B5I8H@L/4*$,3'0B%(E!#+I"PQO*\(8W0.$'%U# !236@R#T
M 804( "(# "'^C@!"+8 A0< 5+6$)NM9C$3#@T/.*!D'1=VAF#Q/03L,@N
M7Z][Q25V]Y9$<:(-31""$#2QBE<,3Y6T.!!7=&&,Y/W)0H6"Q2>,\0I0X%&/
MKBB%)JRP."(@00IO@,,!9#WB:3U7O
M.\A"PO>0.N3!#V> @QG(0 ;&0*%7CO'(#H14+!%X@ ,;8);^ YP5@ &L(!*?
M^,1,]$4F-DKG;&?TZ!)?LQ77U=$5KOC$)1@!A P,P(\GZ$$3FM (3:!S?>6@
M(2?8D 8VZ&%3M2
MN*K$+IT.,\I8SG)\HHQCE(XZ7C1C>(X)EK9MB$.U\(0E@ !$0C01%&0PD8R
MDY 00@( !#F#:31()! T @14L,8I.@**!%U43[/9D
MS)YLB4'96,I<258O:2AV3ZY04RW^&I&]4&0C&S4,!1N"<((L6$)-^1J+3I*G
ME6AD(QJ\J(PQ[.BV6'RBL9\012E0"H8A^( (51 #IN)@!SDXA _F/&L5R[&%?9V"L:8)EE!.UH7%%0
M8A$L<(!H(<"#[+D0+KN083=Z<0H_)(8-<#B#&,(@!20(E0>F=0 *F%")D(4L
M/], VKBF03-=>'E?.;/(J#%5:F!5.EM.,W2FVL1L5#7,
MTO M6K'^NN6X)LW-@(;.7/OL5>"6*ZXT2S/0I+,-J.J9STSLLW+*!94^S_6J
M26,SSYJ8GT:#]31/-?5=G6I!M#+ZMUBE1IAU\=8F6QEQ7310M):-HT_#*SWXQ#66-"$@JF3EVD( @J FXY>MS&JT*+]96%9PXDR;1E.8G/''^"4AX00@^P!X2
MPL &_?U0#N ;WR9LD709EJ,< '8Z@-\Q3U^D:B#WI-]#(!*'!IN!#0'-2$$G
M[)$>"# &DPM!!S@P@08T(($!$( )F*#'#=4B@GTBSUH%O65B8YK%HAX.I,,\
MZV,/GJJX-GRXJ5(@W3:3I,?]1"26P -F258'06BA&\YYBU[LXA2<@ ,;NA[O
M*B3A!U5629"[( E1)-K/7'8STB#-C32'V=.8COW/!%TN24OZJD\%VE.Y/.9+
M2T/2OP8:N*63'V[L'M=^/_95!Y_H+C-Z^M*QX*T9#?WK,]K,/Q/-4WF?'TF7
M%==:U>KXRR7FY;/:S4S$_C2RH??^7JNUUL1GXK"['+)?LW[-["?\FJW/]NA,
M%Y@/_<+,U0+MUKCAU.)L\ )P\+!JS"Q-U6H-N%YM&B3-S"Q-TM!*TPB-_AQP
MQ?)*&O@J&MJ(VL[,C YM^#+M[T"0!8>+)\#")HQ+F#QD$4R+!P")""8L"IP@
M"@AI#,J LZ @8\0@#9"@"&+@D@X@ B+@DF H71@!$A QH@!;[@$LK"MCCD
M%:PD%FQ,-DZ'0;KM^5+M_U;G*W+"NL9"32P!Y"H%+CC!#MQ "GX !'S@$<#+
M"_-E%O+E& :%*WCA3];PCC@$%"QJ%4#AL7SI!W0H#,3 P>9 #O(@#SX%Z0PF
M&YBN'O+^H1,]T>G,H1?:!Q $PNJNCNC^Z<'B30Q\T D,B@AZ("2*!090PJ$\
MA@$"( * .XH@;SP"((B2+CJ1>.4;1HH\/[,3*ND \Q:T,\:L,^R81LPT/Y0
M[>(<>80E0P &*"@1TX/+:@!-.P18V@0\2
MPPQ8<0I^H 9"X $RT?X44#H>C1K-D" %$M.@C]N>;ZYN
MS;>D,?G8+&E^30&S$2HDTMM@#[B83R$YL-AX+R&W3_OL[]?V; $9TO8 TOL,
M$34@S?[J;-<><,YV;2;Y#FB"C0)-;2AW3=7^8(;Y(DTDH4HE
MB3(I>1("25*MC)(:O8PH>;(!$Y+Z(HW8>@\CYV\;0K+WS# GXXK9+(X:5D<:
ME&$IDJ':V(H:MHT:1S(AI?'2IL$L#V26:*+=J7>
MRH ,H" ,TB ,CH (9& &8. %8"!($"H&0@ $0. $EB 3/&$15\&V:LFZ;,P_
MB%'-YJSWB+*,RL7&UM F8B%**"$%0"#DZM!2GN (=& S&P%XO) /=0)+KD0G
M#*5M&DLF/&&/&L%Z@B (! ;T%*,2%V(3J/,4;F'I^LL3/S$? &S Z,E]7F4/
M9N4A\.?# *@.4Q:(#^!L@N!,ZN Q*@ 3JL 1@ $@ "R A-+N0
M#7OBKHPQ(&4R^4[R)H/-\+ O*<\L9+!RR\I0&;%O_%AP&DI0;;YQ+.RH%E;!
M%5C!$A@A"$A@[3K@!'8@"9K ,#CA?N 8QJCB);% 6J3$2R!X5:A%L"2^ JP
MUW*406]M]WYO&ZZ2 S?2SWK4_DSR(5-31W%2.=;O9RIR]S@P^60OTA9P2D=R
MSA90)9-O(Q60^;1O_< T! L2 I>4&@H02&G/^*A/.7+4M] 4_&(R&D,R*844
M27LOT@J0(IDT1_TT2WE/-'%"H#;QI.,QE(IQI.1QF,HD"4K1KBK\_:S_JX
M+$U+LM.23U.A*A>4X1MM"3D9H;1.0 =ZP$2, =^< R@ %F',$4N0J 8$PF@
MM0AZ( F28(@*#C-!( ,4SD81\8[^4(*P#:I0U=(&-$D74#55TBE,4R_S14W\
M 11X 6$"D4M97%&X %&@!% 11>YR;$:S@!EE!H@B;FCKSRB(^"@ =^P G2
MX&^^Z3#\0 _\X.BL\\C,X1WPH1/U(1\V]A/[RSM3I1033,'RP,'HD9!2Q!5=
M$0>(:.S*[NR4Y0'6K@!HE@$R@#\]X16$J4J0(2=V-4S\@V;^&M!1M^])+])-
MHY%3Q;3_,#+1@"_W0K#0C'3WO&Q4W\PLD\$UP9$FNI#AV$123&#M-B *DR -
MV@ .Z T*<, &]DVT3$ )&J%&+THF.) IU$U*54N*?6JA#0U\Y0I]U90A512
MT11([W1PF;0;B._ZKN]/?\]OGX_W[O0SMD%/4S(F*[=R,TUSV:Q')_=, S5+
MSQ5-%_=,35=O3;=OT=3[-!=PQ9)QD=12XQ1O!Q7\Y/353)5=0
M_=88>[=0]U9V\7;.A!=ODQ=Y4S-YDQ=(.34EOVUT>7) R54MF^@8FJ$:3)5>
MN )5LV%<737^Z&K37JU?E^=,XXL)5I&4]_QF^3E@-IV9:UM&]C4I:W;?>B<5QJ^90K]V>6;_FR
M 99GUY8EUY=]%TJEV7>C47UEN8Y5-Y6GV9FU% .UE)8+MQM@N9I?V75GUYM5
M.7/%>9W;69WA.36_K98+]TAS&9!?K=H";SB8;;"2H2V5B73B"'RQX54/T$(O
ML,^.M/M,699E&5QX\C/F2)';-7H^S@%$8"V&-7^/50CI#6, .$5\!:C"6%IC
M44@Z( .40.4\09BBI)9*G-?P:OF('@T:9A#0C./TBL%?"!@TL"'-V6^^*D/AA@04*%4
MZ"*)E[B).Y8[Y2D4;^$63D$@-B%\^( 2O^D\TW,5H< 5B\ )WA,^+8P6/4EF
M/:P !N#*NJ 3< YV9($88H%MA$OO !F#U7EP3[ET?;=T_SC,FH&/I4VS:]H8
ME]=W/=MJ#3D9)HZXPD*Q?P&/8@*]O, *4B E7J (T@"FXW2;5QDLR5E+H;2==31X6;EPD5F>5?=*
M!U>>EYE.63F9I;>AEYF:K92;R9G^N1%UE2-7N:U;N)];F_44?<6;N0/UO)=Y
ME=&T&[:Y[E_&;P*EWP-\9OZDWEYN[;UO9P=D[P=,9O5?7
MNV5Y<*6Q%_8,P'%7M$E5&K"6>[OQ5%&UQ,$7_C2;EJ/QPK][^0C0E'/4@I3A
MTFZLL3K$$I0 HU=HH]\MWJ @LS*BWI(561%'"BPKC(=H!R[L 4R@L T;HV Z
M)QADV )O=+4;E27<=ZMMQH=1+W\S%1[!"D8TA3K@!3(LPUH+ AP!%!8%O*K$
M"ZMDL:'Z6F!G%1@%$KA@"'[ \L+@;^"+JZ]X8DGQB,W!Z9K8K)48K1-&%&\!
M'CG!BK'^6']PQ2+J>@R^. HZ0EII("1.>'*0!; 7H D*@-28!$,6T/XD%"(
M 2S\*J[T^5P/G,(!7$A_QKL)7)4Y6Z=SW6KCN<,#;\8->:<5^;1I013*[;PX
MQQ*^( A$ 9Z( W@@%/PX S"P 9"()1*X4DLRF Y)'._N[J7;[E5MQLNG+M?
M^1O\VY1GE\6Q&RRCL9ZKNY>Q7-+T^]N)NW#IM)ZYH=[MG96]V]T!WMYU%RS1
M/97O?<[Z&Y7/%<,57IH9GI59'$H7OI[;?70-?KI7-YL9OG#E_<'AW>"Q7+MS
M&=R_N[@Y'E'Y.\Y 'L)!?L('',/E&;MO'<:_'4W5MW+KF(_^U9*NA/&?_SF@
MDV=TJJ(:F"$9]IF/*[[%[5U]@]>0$=DFO+ 6) $2@OH$HC!O7+%7Y,U_AQ"D
MB9#(C5P*VA.!8\"TE" 2GASB8+I>E"$:6OU)/[OC5U[AX\P8K6&?U;)>P+$6
M(,$*5. !4"@!,HP#3L*30*D19('AC.%=<6LT%=N6CM.B(N$*?* &A. ((/&;
M[,![YFLA"L*(P]H7X*G0\^'0G;@309%]DHXZXW&<)/T\Y2W>5!986):(!,CL
M0&!9UHXE%@C+T!YNXERQ0V@8?X+*+S"Y:][!67X;R/W"E]F6/T.?=WWZ0[N;
M;QUYZ_+H@[W+Q4*QN[ 68J(6%O'^$7P@!'Z@F\()(L3@"&) !;S E"2A$[J0
M0WZAW[^=Q5N+)A%R.[D1HLR')ZW1O#F0Y3:!&QD2
M/'ERFK5ITYHEDS8MF3*DN:0=2\8LV3%CT:)1C78LFK1DR9(V(TITJ$V=(FD2
MY:9LFM-CQV+%>O6JEBA&*C+ T+&C1Y$B1IR,<0)%C!@H9:"@.5PXRA@H3_KN
M+4*DAPP8,#J<8"2ITR>XKV0IBT6+[3%IRKA%:^@3-5,&G>
MP)D3)WP>/'OZF#=E"M4M7[ZZF3-7+]^^^?3GSRM7+ENW7;9L;?J_"1]UU#%'
M@7:844898BP6A1-&[-4#$330(,-D,$ @P@8/-,! 0LLX( *BU#2V7+$Q$*,
M,5F--DU:85%CC4_;K%;112%U0U)! X&U#5@^_@@DD ]-0].,,)WD36LP_MB5
M,DLI,Q4ML2#SEBQQ??()*)YX(@0,/TC!!AU\Y'$&&T?H 0CG4A2RW*ON!6+
M0M\4-"=%W'Q49YX$Y2D022EMTTV=!PF:$*';,$0H0V09:>C^H"/)6>A.D8P7%6^\LDJLE#7R!(I_/#$$V&084:!XOV1
M1Q[E];$)>NKYP@U\]="3CWSUT1/^#W[5\L=)U__5(: <<\"Q+QGZ1@&%$T6H
M'5D/DDUV0@@0//" PXPL$ #)C 1"3'62D+BL80PQ977/EHUJ(5$?LI-Z'>
ME%&RW@@%%E));0765V%-?BQ-ONIX9&L^5B,-4VS%TM9L<+792>N6*$&"#%JP
M <=X>:AQ1 Q*/-))FU@.XY8QL? YT9QZ?H-\JGL:OZFFGM9*:O34&YI\\HY;S#BY!>&XO:3?Y,A\J8)2KW[V?%9<4/>QOO_]_-Y;
MK'[[L\;?OP!FSW'^DQ2>\L>__QV/7-2P5?^T1Z?O58\@&E%4 X&P!!
M!C( ,YGA;&:R$ 9GGJ,S3T"B"[E)01">(!@QO.$,X8E#TI1F'J:E1SV_Z 5\
MX%,U^MP#:UK+1G]LT;6N#8@.<[ #8@K3KR@T" >0<5N%8D 9N76@;@LH0 $&
MX( 4>,$2<8%+X(A!N&,@XRG<\M%+K &I_:$14-.A@=G 8TQS40+(@-*(3KYC$)][R
M1<*%#WG<\X:M\D=0@LHOH0M%WO0:NCR%#M1Q.))H0@=Z489F=* -Q='T-KK1
MAZ4N^9%*8$-"A+.2I1E<;T?CC%J$NC%S_X
M*21)GC(?4#.J4YEHY"Q@64ILIF(,6N2B&KGPA"4:\85&6,(3M%B+BS3W(@VJ
M]*,$_>A.2-6KUSRK+4*L12U4H $(Z$ ';JN7+/NU&,(81J]E4$P8_E)%(NQ@
M!W%S ! >(0E1%!-%M* %E+@AC8G^@6I0.]W?\V:4N6.TR(VQ\,J>3'-,PS-P"G^6E*)1C,R9A/\
M9@1YCF.RDB\:K&]4D'KF@E&WCL(M:72KOD$B"N4#M)RC#6F'RW'1J$>4JO'5.L)Q3I2CWYCKB<]H6)WZ,,SVHUQ1)V@
M7F?[V=N./+&7?>E=S^B$6RHIFQ 51C,BE48FCCSHW=FE6Z8@YYK133"WI2VY
MD$4DK+"$+'SA"TU(,Q,@$0LQI@6LR3KKD.%^T3RCDS0;?DLM(@%7",A++VL+
M@V+R11BSF>U?3_B+@W" QO8( ;^F$:!FCR-.B@E@R&2SQW@J:U**5[
MEEL,N0@4P& '.GC -GZDFPW\@4JI1 =Z, =T0 =V <(8C:P
M)$MJHQ<3(@,Q@$LBD"$;8 &T$L#H 'K!0E8XC&Q$().\A1+40W-(!;81$&#
M%W89E7Q(E214IB0!EC'UM16D43I)H8.;(SG8-&$LB%0Z,F%"P17<8A5O) S&
M,$BKP A H -@4&)^0"9M4',HT 6K(!>?\#$?@Q5S1W1E)PY?>%%0AW5<1PY+
MYW5>&2%8.:M=TWY"&%Z6&:X<\:F=U=#@.X9!1<6AU6$>'
M!&5U3A=32@=UA:AU4&<.T(,?AK@-H%>((&4..W&'8VB(9 =VD?ACH"=Q4#9A
M5?9W$Q8C4[9E$%8GQW=CF\)@A^(C3H4NLJ$NV6 )9^8&H= UH> &QF8%DJ!Y
M22$6I/835Y=00 =W&3$Y10%F55(+C' "&6 ">-$#].($LC0&B:974/ &D?17
MMB(C@.*2_8\Z)064[%JL6 =
M(L #)] !X!O(I RJHXA#_- #]QF#OH1@./&"?_!!WI0!W! DV8@!MLE
M&+%TC=_U S] ;^-U@1O" PP ,!D DK ");P6L&3(NBR%<=@0PPV$-Z #=\0
M#I6X=- CAA=UAB_X#O46;
M679:YYM!9X>6:95<)W9 AYIUJ(AQ=YN\:8>P29FBB3R N%%3"4V30V$Q^(K0
M)(HE19[D*9>A.#DIF(_/0H+/%PN>\ 4C$PJ[$("
M1Y7'UYMO&(:422JN\68I]Q:=L @GH $B,'T[L!?V@HT+DB"%H5V1Q$1C, 6W
M=P.Z%P+PL@*1<$B$$WS*D%]TQHK?<(90UY5U]X6<.9K),X2: R6K!A>3< 4\
M ( *9 ;H#(=H'KD]0!6" J2( F3D!G^] FZUCOHEQL[$ 1($!AO@ 9GT&QR
M( =X8*;^>- 'F%1F!N!;* V@4P8O" :H,#
M$C*!XC4<=,, !N!+ Y !"0E^KV!(L2 ,R<06.'A??O>#X5"5XMFCS3D.+\BC
M2@>?\ EE-$ARTZ MS904]&661[$53M(B8#$4#)$D@;=1\)E0HHI?KD$-V**/
MQ" ,_B""E* $.M $:D '>/ '>/ &-;C7\F'L(FM-CJ:<[B)W;F=W8F'=-B:F*F;[8H\R,B= MNCOKF59'B9YTJ9K4FNFXJ;L-G^FIXYFM3YE40'
MF>*:F;^I=-V*KV4XG FUKW$I=)XHK]S9FEX'/=@ /7>'GN@Y:D3"GNBIC$4F
M<>>Y34KQ5")AYNYL)PHFBOK
M&D;!%E1B>E> AF C$@ W7U@(K1+XN&!F] F-H5&'_!1'MQ S\@ ^6( E?0
M:8>D#([5%41"$)D*G_SJK1A+KZ?YL7>7H%\!)6ZA.OX "4& B2 J'&EH:I'
M&>5E DL0I56Z)N[4"?[4"8PP!"?@ S]P!) D!F9P!I04!V5:IF:Z!TK3--;6
M"\K%7/% I^)&;GFJIW 0NO]B+X#^500XD**W! ,A (=@&\;DJ@:H )8 FB
M\!R"(QO%,!I.DA9#067M:975*ZXZ*IIBN+$]BA\0VZ/P694908/88*K?PBU*
MF$P%-U]/DA0NDE\3UCW0\YY/^Z L54&[*AK(Y!R@8 E"$ -'D 8#@JQT4)A7
M, MPHD*%,PT$E:UMV'7LVINDB9D\VJWUR['A*H8D:[#?BJUAV(:EV: ]"IX1
MNZ\E.YK^^J!N. [@::-A>+*3:9E0UZ[UV\)L9[%L!YWF>KW(F*V7,R(6VT1JK^B6*BMX\@L.%P6_=T<-RN(:70&5RO0QJS8;DM $
M/Y $;4!N=2 %2, #(G %L9 +NFHD*RMX1,>:&#O!3EN(9LQ\RP0GL= )6 L"
M(A #>=&AAY:-#*PZ6=($)J ! 2 !I !&**AE.$"Y<4$EI 9F7&E
MDD );-()CK ")W "06 $84 8AS&ZWS$'>'"Z,F<>:[I_YG"2\@ /65,MV< +
MJ 22 G))X#&Z:+ @4H0#8X![O3N!(U!K^)9O'%( #-"!2N#^!9WPK&^"3,A@
M#,A@.'6K+'3V/,B##;ELE1Q]FSWYJPD,ZW-AH
M[=:)3<. &,<(9I;L] J2P 3$(0)X000WL!=1\ 3M1AAV4 :E3,KMQD11<*)%
M< ,[( ,BL*AQ"ZF,I0S5P T!=JG5"['Y^MJL;:/%B73#+7$",12">T+%3 N1
MD+4" .\ G4%XN\+CE!017@ 65$ E2>J512@F,H 0J< )#$ 15D 90-+IG
MP$H"C+I[4![_80O)Q6WC\ [E@ W54L^!7 =X4"!G, ?[K"#X8B]58 0!;0,S
M, .XM$?^]]8 88ZH# ":=(P[0(GQ+!,%,T,S* Y4ME0[\G1CVS:2-R@V"IT
MI!G;9XCAY#F7*NLMW/(*E( %*V#H*\!>_S05A0,E+=(MN3J5%S6_,QSA@FS579#54R+1V_(5B1W6B6W%+BRO#=S8>DW9KK[%
MB&VOM/[9E ZPRNG8^5K">MZ57LSK1*>OSBG9:7V:O>[!XJJ<0CS"]DK2$ZS7
MH*W6R[ZN.1H.>MW8M5[2I.G%;VW:9'WM#/OJE?V@*PRPL-[#^=K73]S $1S%
M)]OM6US"]"[9RCGMI$G9JM[8T%[AGQV7P^W1&0$.ILC;O W^3J.�$FBHSL
MYQG.QO+9V\F >,C@O+(1@K) "XZ7 A:I!FH@!5 @!2.?!#SP !G "+$0#=:0
M%@N_#57I#1[]A7W]ZM\)P1=NU$2!L\>PL['P9_#"R?-"2]<8!3EI-GM%MH0!
M2_^M-@,> P:> BYJ,R?B)"C(90)OA^$P[.:Z[*O^[2?;Y\>]JS@+HS0C"5B0
M 0 ]07I3QN"X0 CIP!$A0!560!5?0"%KE"9C0"+CUS7.?!F&@+Z([NF2:
MSD'^'R*97.Y1D@%H2@-H775 IG!P!F9@!K,W!F'P5Y.&>[DW X,*O'VT 1S"
M /R6 2LP(K5PP' B&X2##*[*#&#^H8J7BO5:G\O6;N_KWG2@_>O=/IW[ZM'A
MP,@5)!36< R\ E+L 1-T 5:D 7*W]"Y@,?VF0P9DPTUD1$8WM%9#]=?R9HX
MGQ'*0H+%@ R^*@N6P 2OI@9P('-0+@5& )=("61JD*D(Q1S..PN[.Q?[,#-
MWL/7"A#BRHD;1^[;0'$$S1E$F%#<-W("'3HT2*ZB18CF)GY+6 YBQ(GE+(($
M^1&B28OB0 JL.+&CQ88JQR6$*-*DRW(V5[[TF%(E2(07(>+4J=)AT(,4.;XL
M"!/GQ)$(8\;\:1!JPW$+4XH<]W%FR(@#EQXM^M.A1H$?<7X4*RXL#HM 3*E3!DQ4)X4Z<&#!PH'*"(IJU:MV>'$CM^>_&CUZ,B5Y>*&
MH^MMF[5IU90=0T8L5BQ(*AIL("%#Q@T<.)PX@0)%BA@Q9,2<'M_]C13O4)P4
M8;\C!H@,2B+Y&T;,/G-EA)'7)?Z0Y42TF'(IK865"42HA(8 #%("A@Q=>\* ##D+P00LVV( B"B26:**++]S0P@<18O@A"220
M4$,-,LI XXPXY)"C1SGPP(.//?C@HX]--CGEEEUZV:6;77;^L<463JHT$H\\
M\ICCC"W-@.,-\,)X8CLCC,"!B!MDB"&&$$0080,X'6!@@0'H9&&12%X99D)A
M[".&N6.426::9I*AAJ]MMK'++N*(XPBCBW82)T"U)J4()'.6ZBDXCCJ=RZ[B
MN!'TF%":2"(-3D[9A),TC@C"BU:.^5.9Y03-9L%;N4D05.&P6>JQ<(0[RJ''
M'*N+T&22-6888V0!Q8H:@@B##3^RA$.-(D"XPA5_H.&LFFQNW0:XLT9**"+@
M4BH(JG/-O30JBHR"*J5TTVVW*I]$@O3<1^M-=Z"5R!UP0(O00A>XK%QR=]YS
M+V47X0#;W?==DMP]N-*&!\Z8($G^Z26I*H2:,HHM2U+B^^JK&&F8T*X8S
M8CZ3!19>/.D"B"7&0.W$(HCH00<('C"!B4Z2%=2P;8IKM+^* X84H6"!_69I
M:YI9SKGG($%!@PU$L X['*)XXHGN*.[[J*(8KT>W!,!!"4@&>:3
M5ZH^)EEFIE%,KK?BI4DE?;\Y66&7K>K*KFT02RZ98XR)11@*/Y$D$A4R(,$'
M&3JXX((/8:CAB#;TT*...-IHI(DE@O!!AP<>X""&)(X((PTUWBC##O"!Q$..
M.H3$8X_^/9)4\I0HI9RRRBHWX6/Y+<\PPPPRS A33">,8.^&&]A !C. @8?@
MM $'-&!."W# V2HAB0F]0ABQ0,:# D6K9.1G/]98VG" 5;JYM,1<#5&+R_QS
M+]8YQ26/>9UB!J,,:]#B$4 X@AYLX8M=;*(-2-#!$"PA0?O$PH+ANI4WIL$-
MQG@D6')Q2^D&))SAU 6)T\#@U#Y#"T]D(0;2@D.6_D '-1B!!%?PQ"NFA@QC
M@(L:WMB*Q,RE%7_%<83B>$<91CP:"XQ[E:# _QBV0_I))'P=I+SFZ
M\9 1J:.[##F21OKDD9.JER(3N*LI"%)WR)B48D(0A9 (\8QM2#'L#@
M 0UH0 HB$8MDJ1(;<:',<-YRR(Q9LRZ5:09S;A<+2JA R @00QL@)TB;"=Q
MW2%#CAH'!3M X7&JB0)[B* F$:"@"Q!\A3'^A(Q!\84QPY$7N0[F+WVUZR3^
M:4QA9I=+?\9B^M"'/?B!#FUH
MPQI:=8,9=&@&(G6"&-XPASG8(0X]ZI'YSH<^]0652O!;51W*AU,XV ]_Q>2?
M_\(6P '" 8AZ,#E'K# !3# !$#^<$0GAH&[5\2"&&@,%.T&!31K&,T;D $'
M0>-BR3C"=80&S>/IHGBWRO3L%8PXP1)"P;Y;<*(-5? !#[( BJK9!QD5K VA
MCM@,QAS$FF^=BD_>XBC(;$,_RBI&+)S5A2V& 0YSR,,?YJ &)Y!@": 8QC"F
MQ@R>'=$BD23D(=_1E$;&#)*:'"0=>1D[1C)IU+
M7.AJ:$ 3\'D$+83"F$\0& P@P$P6+6$4%DP$TQ,SE;7!5KKE HDT.
M78X$H2Y"'C0.(U" /;"MI.0A.R1!>68(5'M (7F>#"$&PP4QI$
MH:1%.BDG^J"'*OFA#6FH@IEH0(,B1($,:)C#3G?J(Q^9#WU[Z ,?DM0'3B0)
M?E6B0QWL,-JDF@$-J!G#& ZW&GL*4$T$I"H'-L#, A1 JURUA"R^.=8*&CB:
M^O&&9-;Z06LV!#@XCFZ.'1*7M3ZDTM901C-JT843!$$-H:B2'MR A!V,@ >0
MB(4R_%0,QI;ZB$@\;UQR4F."H:R)CD).U*;VK4^ EHM^P$/^'*[E!!%T 11F
M?&UMD%.O=RBWCHWD-#G>L9!N>WNW)LOV;#<][D$VF-NT97>YTZ5M<:-;W.R.
M=W)O'&YZ^^O;[99WOI$;;G-C^]WQUC?!<7ON;P^SG4
M-*+A"4LTHA&/N(0G9I&-:M#N9[$EC'Y&:=Y0$B89>4M6LM!HC&/,PA*+6$(*
MA""$&"0 !D?@,O^*0(,==&"9($";,9(!T+Y#K0H<]CR)\9N!0''J'9IT):LY'\L"H^<$)Y=:!#3N'P>A&/8>QCZ%_8B(#B
M&!20JF5CY@*RF@$57 $2GZ %+8C1IP?1JM2J%*ADK,'6MP^,D0*']\*WO? ;
MS_;3;WLE-@C%C%2.XZ]O #=RT=9LM#FRX>@LX<]LVN2LW%/2V;#/!@&-!A"M!#VRW%,Q =XND
M&53!W7+^N.LK&! 401J$I!#,0:VP01'LP!\$P1GL0!,,07Y;0>M;01;\0"($
M0B=TP8&K(SK:/LB C+3BBP61!4OX@OO"KR5P@TS@A6QXD&0).5R"+=@:C)(;
M#/%J!C8\.C2J&EH0C278PR9H B*(@1R0@2<8 S%!@B38 1D(@;)Q "6P!)C[
M+AB2C+AHJY5)P2&<-WD0M^W+.RJ:.N>0!-C0 ,&1 1O :WS,!SYNL9Y U:$
MG!-9CS2) ;1[A$\8AML9*UK9#_YPF$S401SLP8C(Q)V HF^P-,<2%-J)!= P
M!E!@A"!(@E @/'/HAEO(A#7H,B(H@B=@ S$P@\?SQBS)DI/^0BDD*1(YT)(M
M,;,S$Q\U,Q\B*1(^,"K6>SU6]!X=D;TQ(#O_N3U$'($0^,\J>Q8HX).S#G<[YP:"N"TIB?\$4JK#<7U$$FG$&[DXMRN ;*L(Q8
M8 03(($=^($:,<0D^,,08 )'_ 1E%*MCR(66RP]NT(:V*@=L",FW$P<,TF<843,(DRU#50$>5PDF%U+W$
M5$<]0D:53%*5U%-=U1J$3%?U2R"LS+8:M:"YA"MH@E#PA"K9LB/P@='TC&2P
M(CM,U#J,FD)IN41]+[Y11ED8#2 (@A8Y@IDJ D*4/4/<@1/^$(%E.IM.@*\)
M YK1 38;I#?'W+:/K+3D6([;.89.Z!T'P#H:N 'V< (/6U$1.P,T&,]Y6M$7
MM8$8( $4\ ))P!VQ0@8-$JB*O$A:Q51Y28C+]*!M(I2I>SE:X 50H 3["@5;
MR(9II!(Z2%!99#\9F9'-&X/[\8,_\(.48K,C.9)-0*D]D ,>X2F>RI(XT- @
M*3.K-+,> 4LR #V 8D($=
MH($N%1O7: 18D(55Z(2,"P5+ 7^7J@&;$ PP_"(MQM;@>$DD P'J,F,S2C3
M+A"!'Y"G.O 1JH2"&!A-?R &:-A*9&"&4D-!)K5+U7W22]7((JS+((3"2(74
M*FPX#E1;L,S=V#U,4Y7"=4U5Q73+N9S=5JW/)Z7=UFU2MI3=NU1=Y/W Y!V'
M'92'.I('WWK4LYT)=H X6#&*!@#(O@!'7@3!M 9QJ&Q6K#
M@+(&O'O8B^Q 823,E9C(_-S/YC"&3E@!^""!&2!%?-T.[Y0"',F1^[$#-$"#
M>'J4]&*A;A^$0*,M0OL_X#% H
MLBYP! +M!BC9!#"2@B20@0TH@0:H$PB0JE(T@BE8639X/3\P$C).$O1IMIS]
M$9U51R$Y S=V8SLX S I _&0/;([$QSH 1I0$QWX1Q 0>0$P88 8 OD6@
M!&6<(*WERF;5C_CT!G!HJ[::"2>R"#PJVW(H-^LEAUY$2]L]WKSD5"V,!_VC
M#,QHAEC@'08X@ W(3NUDCQX0@1/H@D[HA$;(@C[,94IXO_S0!FXXB/VSS-*A
MWB3-PN%H*V#.&P)$ACZ)A6+P A'P@:>D@Y]]G!$( EH(9S*(;G
M74+9]<7ZA%XHK,+5A5VQ;-Y2'=ZT1<+;Q.#$MVX$OEG>---7=A>?HY62ZZ\7KQ5VPU,)('C55\H9:N (@^ +"DQ).
M>#4?"($3P().$ :2)NG[Q;4"M,,"Y$\_D1 )<99&" (>Z$,B&&$C +$HF *7
M? #T #B#"MG-DKX-*^[1<&@:W#Z;Z8NIP]]/P (4^&-7-L6M
M^S H<"=^-L$L_+0F
M2HRZ: 91H3!CF 59H(0^; 10$%G^ ]4#-0B#*NZ GM8 J66F!OBH+\L.'!@#
MU1B#,(8#/S JTN,#$-6I..A<,VNV/\"#-_;7%F[AT^BS$Y&<['@J-5F3-A$!
M" ADJ@&9==);KV\JV3,;"
M=^A><8@'D>S)=-@+O5$&6A %+ "! DB $,C.;!2;$]A1+R -(&""/F0"'@@"
M1_"'0-'KMJFTQZU(ZC7J8WZ,;+@E9CA*9G8.9/ "$)#F-Z!F/*B#-WB"+7H$
M?YB:K?06S/!GW-7>NOS%O9S=?U[N"5]7Y\W=(![G>N[GW:7=A';GA"9Q
M) 3QA%O^019?V^!=;A+/< G_9X4><2"\\10_9RIT4@UG:!^/".EVB\N\6&3I
M!"M( 3=@M4W0@S90 R2PX@90@4>0H-LI::U%Z6.K(*KY43V1A;#B!5YX!)E^
M1AB8@2*XD5?$ 6\%Y$+^AX0]OO?U;=0='4I,B.[=MA>G\4@2![AV"YL!AU!J
MA@KJDT]8A LKIQA84",P'*YKI]- STS U9L)_ 8:QRP ;..CSPQZ?@F#+$M
MG;(%2V*>+6)&P8D@XF%+C&;0!N5[$&.@A55XA, F4'/H!5MHW9II, & 48()*Z(3.>8[WQ;4#:^OF*]++! >W< FD
M7D+LY7&T%6?EM5[&9&>V'.*.($1& $0B $D
M< /PD0(IZ $F<(1<4 9NX(:(9*OA^//[Y _*8+EE,$"2GI K\( :J (VH ,A
M^1(HD($@: 1/2.*IZ69F.&B ]^>AKV=V#G'F'N@2YW-S?G%Y $R 7E)-E7$*
M%^<-AVBD9WH>+WKH?7IYGFMO._7B)7JU%7$;S_&HAV@D#.?KPUZF%DLIK:Y"
MT9M.4((3:((EUP,V..PBB $(6.#^%*@$41"&UOHJDOZJ_[:/9LY?EQ:R7O*$
M+^ !J8JI&W@"-1 #VBN[& #7!5:!2FC/- T=2QN'BO7A'2?>?OX*4!-TU/5$
M8I@$1X"-BS>G/&90[P"QQG%A20=KU#"F,,"! *KA(O X0:)$@Q]IVM2I(T4*W$6SB(E5
MYDVFM9KAP(7C*6[<.L0YP]4DW"P9LF(J8[UZU85$#"1LZ.#!,P?.F!]"OGR*
MY<\8LV+&7"-5>C2HT-A!JUXMBIL<[JFU8]=^%_6W5:;FY#&U'5O><*G-W\D#
M3KOHSZ5(@4OG?1VI.:*RNWJSFP9'7YPXU?2VA;]G.H[\5.OQDTNM
MK]]J\//%C:;GVU/128743C@A1I@US2@SC3*K7$&"#T&*,+/Z $B,DC=#8B!9!Z "#CC0@(44:3SSA
M1!$TP !1R1<(0IF)KF63#6$038.3HI]5Q2!\D1'#G*S*985.)')E Q*L0SC
MB H.@ !###+8FE&+B/!8.6(4Q.%8NKT"RQ&F>=%...;9LP@D;:R01
M0P88<8<=LQ!
M$)[,HM%L&=>6D1 44;QI1!%%$''^PT0SQ'!11B!PM$ "YB0 A:4I&:,22L=
MPU(RS!!VS93.--S"-58PP2_Z2Q0@Q'"$&:'OX0<<3/P312"NT@))--L:\
M]@Q0O25E7G@) ^6=@ #NU[: U[$M'#MKNUW;PDV979Y_^E4%-W36NT[%C1Y]R=V'W]@(GYT;>7L#54]UZD&7)>7D*(;330LR
MX^!8LG3^8<())^CP Q$[Q/!"!PDTP!$*6) DRRNR +]*)YX07_PEEH 22B@T
M=K%$$,\_OT,/TQ?1HQHS?]O##,$^D($2DF0FC##%%',O-O@6-HYC ._47U+R
ME#.=5>+( ^F4X&RSH%B5D4E)"F@^C5@X$)(3$B(%/]7)# 2Q0QG@\ 9L 2H)
M-H@!"4[@B$_(BQCE:XE,#-,32,7C<.]@QW ,]*\#:C#I.8H+'O<\69%(&,LJ7&9>XIV^%<
MI8#2R'?X!%/A6$=7-K4_9,1B$HMP0 $:H*.V#&H(->! FX0!CO882\SVP 0
M+O&*P.2,9S?A5Z8<,YB>(8U!Y!,19KH0@QH800QLP$,>],"&-#SO"Y[@139
M ;9K0JYNV;3D-HTRN2IUT$00I" 2M1#1^,B7NDUY0TJ)^> \H7/._: 0,H012S% ^0K_
M90 $(5@3#;[UIB= 00QR*D.U"F('.'R1@660TQBB8(0;Q $(%C$)V2A*)0D
M8QH=],;Z%$G/I/!D'8\1VF!8J PGR2)42Y!F-QJ["TZ$@@Y02((,/*"!$3#A
M@D>$$1I% '*$AA"C0 @15&$8L_!L8:>I7)EW+2KW#X3"8K#(P?ES8,S+C""\\(9'.?M\L7@(RA2#\2>2?UVG
MC3.W3L>UN&_B;;&+_T8W@O*XGKMQI);:EI[XY:8ZP2$*.<:17:$=1D%BH0PR
MC/$*1\BW 8P0 ,2@+L&%+=##3A!$(1P! IE00L4TH(6FM $*32!ICY*@QO<
MT(8_ZR$4=> $'3B1E^OM!0FS\^D#'G""+I3B%:)(E*)84XW^;'P%'-^XB=!.
M^*_IO.><_P2L.'SR&'!,-1F*&L8D_J=5&-"@6&XJ8%CG1"=I35/<;J%X0-2[-"\5^@139, <.">V&
M-"3!!QW0 FP$ E:H?L3Z@:%(U(PVN$ L4ISH8_DOR.<2#L<"(<6XQI?.2\#1G&
M02EOW]M9#\6%]VXY[C%5%%_D@89WG+$YCHUC WAVCL<_XQR<;P[&>':2_',_
MN>XZEAUR5'])P2VIC&7 !HK0+@ 0B F,F\D0;@_@$Q(,(1+D13F?ZY#708
M_O#U4 <]\$$/RM>#'_C@_#TXOPYO",,3?MT#BVX@ 1!8PB4\(6E*"T,9QL#&
MI;%QC:0JE2??\2MZIY+(A$[;&@SU8RQ"J00T550'0A?20J!01EZC@;1XT6CD
MR5J)@5O^%<$.G $, $D@$*B=%G.<,/'Y00[7!?HO U3F%I.V,1@3 ,V9$,,
M+4$32%,V=(,YW (GZ,$:N,$1[ $@, )*,&YO4(1<=8D?$(G7($)5) /# 'O
MO9D17 B&% &0>$L!!1P;\)J$0&J, BG!MFE(1*H 2"Q43/G-]C(%*I:1?D:=/=447@V(UW==/*&&!
M@(7.<,-8:- P^ ,EK, &( &?$ -V%?"*=P%Y 1J $4%($,0 )> &E^=&]
M5 ,S, /Y$4;J[8M,<-R$C<4Q7),QQ$@H-$$03(\4B($?[('^*=C"+(;"%V0!
M&%C!%RB/)V0#+ZP8D=T8'7Z>RG6.W2W>?N#3?=@&XA5>-E4.5!19C0E>.OW5
M?/ -X_T8.1FCD[G<)6%><$2*'BZ;Z2W&.@A6R$F5@BT(2AR#950:(3I ,A>
M 902FN$>1T! #[Q!&X0"H(6"'G"""B(?)_ !03I?\AF?'^0!'^!!'<1!7M !
M'*A!&!2!#0#719U $W1?^#17+#!4-7Q%QFA:3F22P!R%"96!#2^ 0B,T 1/(X7N? YWWBIW#H&@K42#Z(Q2!E=J_, 9D)%5D3$,N1()*-0(GE(HY/)8?K$$/^8 )9$"L@ (OR$)E#H,H
M6.HG>((F"$%KW
M3]0/P#B83?QATD"#(!:#A2Y "7 #/3 6R3L.0 B#)D:."!BL9!
MTN;!&>"!C!9$ !X$&D#!M8P!% C^R:!@39T=@0^D0!=(PJ0Q5Y>!9$B:G_H8
MTB$1!5&(0W8!UE2BE_IABCCP54W !$N0!31\@B.<@ -0E Y(1 \TQ$Y.+1DT
MRQF0:5#.@1P 94%<"Q3H%&D]0 I8PG+%0OEDIB[I1$]$H\J^+)@DP^MUY5=R
M&R?< 1OTD U\ '(Q@26D&"^LZROXP_=-0BIHPA#DP P\Q$,XP12\EA,([I\\
M@<"- 13^B:JVU6$&IZSNP/+J" QTP//J:@&T0 $P@ 8 2- @YFQG'>56#
M1,^$PS?8*=RB+%.PW\>2$W*4[#IQ'JEATCID!8)8@S;4'/D4 S08D2B10 8
MP : ZQ3^@,L/3% ,? '/$ )!)6ZLBMKR"9+Y R^, J=!@:7W1R*H*(C7 &\
MP9T:M$&>$*X<<((MG""W=<,M8$(H]&L0+,$_H*^1X6>6C&S9!,[&RD/& JC9
M9&Q]&,S"Q+ _T5,/4RP0WRC 88G=4;P$$9J$&_\=H6
MC8$34-^;[*X:.42H/D$8J$&'J8$/_8 72((KD(DP0,-*J$A(9L,W($CZ9=50L+]B8 (E&L/X$ 1.,'^$T3!GQ"NL\2!
M'0BEXOXD',R!&=@!&20$AB3E"3 "251N8&B#-G@#-L3M5(1L>:FM3_@$8EQE
M9-!"(P!!$_"B8]E"**R!G/D "(C6/U!"+-#"B0B#3,*N*&BQ)S3""C$.L'42A>(DF))M'M)ZUK,22T_Z!%"91R
M#\@J(]J #%" !@S!(R@7SM*F_6:BO7@O@] O@W#BUY"/*29/$,!J&K !1+X!
M&_R)7[*!)G#"+?0">/:"+7 "=@K^@1*\SP\3L(0SE VY2,0UW7-;]8 SA\R34X R 1E4 0X( 1=@PBO\0J)457*"9#:4PS=L6OC6]6)8
MH"0GJ3E:(% T\0F59#/O"W1]LFJ\G@.<0)K(0*RQ;6]':C'LYBRF9@$()*+TN 4[\W$JJS*KJBD]Z]1=
M5D%=ANJWR.H5ZHBY3%03+(064G9SY%H?%8$HYA"\IUJPP&-$G@,(59$ ., #P(#TH/03_/$:A $1
M?, 0@,$C9.\GT.:Z,D,FMJN*X))8+,,R(,.E90,S9&L.-L(1)($3E($
MV#.X" $2:($;: (I,+4YF$-30[4UPV=:EP?B$6B3 ?II>NSE>?59_W!6=[56
MDVRC1ZQX$<7A74?%R'4XE,.E.YB47*5Q/R>^1!TS5,-8-%?^. LBC&"G\Q@V
M8M->9*)9#DP!6$%X]>FQ/D]/#XAV\SKO\VY !4R !\A $825:1]M+:=!%=P7
M%X#/+QQG99@/-GP#QM"$9\:O3C &<)=:Z:ELZ4B)*-:K,O 16S*!9$+ 338X
ML@ F&,6R',A!'L"!N].R&;S!&SBNHNE !UA0G(8?, OSE)0#.51,D5U90D&&
M7G^#-%R"%=PW)YP"JG!"&TC!$>@ $)$ $T@"^>"3"9!R;UA7&8&'R"'W^6D7K\)\SJY0MQSH93L647DYD^J:M$(,L
M \Z5R!9' @IXH0.4:UL@0:&A2BB0IQ58@1)D%D(;D3'X@S!8<+LZL'3ZD3%X
MC4A/PEL"-1*(0>*^3<\-2>T,,0B<=0'3@Z7
M=7O?YS827AX6>J0;<6D2J.:+_E^9?I'A6(P)A7>D[0>Y)H(XQKYTH" I"*BK
M2#70PLW%@BSH*V';F11\VPL>0!2)6>XI?F+N@&?O@ S0#FC'0(Y
M[29A(*>/6C)LU;^#$E1,7
MCQRY=T+?%35Z]-VXHN+8B0L7#AQ4<-N201)RXD@H3J':N$G3Q <)#28621J&
MK" Q8;&$#8OU:M@P69\L*3$1P\<.&SV(1#12Q(B3P$X(%RYL)$F1OGUO_-BQ
M0P8,R3!$@'C0 /.! PU4_%M42=(D2J! '?0G2YBQ8LE8*VO63">XG4_%U1;W
M[O91HDAY]_;M6YY1>?+&[28J]+8XH4'+D5.N5"DYI4N7)J_^S3-<.6S67B]3
MAI;8L%>?9*VZDT)$!PB29?Q8DVE7MVZW4''2XX9+DR!-NE@B;2P:6HP9<,!B
MD$$KE@.%0:88?P8$I9$F)$RC#3KJF",..U1R@@@;0MB@ 05>D($(+=QH(Q1;
MXC/''/E\JZ>H>I0*#L;?>@M..!S?J?&=X7(;#K?@=-2QQQYQ)+(H)(4KDC<>
MB3QRR2=C9(2I<<@8QY$RR:.0!!(I+I?L>448
MG8U)1AG^1*TA5-QQ1AYYSC"?U!%&(.T,<\Z_>U-NJ*)&3HZZ/)G*;1T\#U_G
M\.S".?0U9@XLQJU7+'UDB2""."((&6RH80@NX.NFQ5YNN<463MJ0,(DF''$D
M%%",J2:;:HSQ1V=A>%9+9W\LD?"(E^FPT([DH8BBB!YX".&R!1)0P 42D6C"
MC5!"X44^%NU44O E^QY.R#BA-+-'T<^QMWK8$9S&HJB,4TT[W:^S
MQ_WY7G(>:7K3 %7#Z$0\'M,>M+\TO0^\I6)?X!CDOJ*M#[\L:E+X<-34I(S
MCMJL8QP;@PJ[!G4-1#%*44=S3.<"*[FW22HZ?;/.Z53MD8HZ;!C&=8;F=R
MD84EEN"#(W!!$VNH@A&.4 4NM($3J/!%+^33C6S8HG6AR!03FL ITO""0&Q1
M2WC4X@]_5&()P92"&]APO#J\00Q2B((3HB8"68$ !3R @:PZ$(,?'*%86ME%
M-KJAP(-*21X!W%\LD>*F-ZE/?>2CGT$C**<* FZ Z\N;D!&-:"03&3"A24+4VV#FRL@U G,R%L6C.0
MM,#3VI%&;:( VMN2D3&=S(8VB;L1^&Z4035%Z4Y*T5.>].1!R#4%
ME*]_�* "6K4#JA"7[W&TIC^1I8^[G5;T*J
M![G+EV[(/O!\!X73WL"M[0I>Z=WQ#AP&O_? ]\&H;WX>():^_0ZUQFC??BXW
MD1KK[[]A&]P*#=]$E?+!<:A#*H#^FMMG85/:TB[CR*=E)H%:ZXD(@34-:G@#
M'6Q;ZB@D@0B1Z< #%G" !4 JRA8@1*PL A'1"(2DH!$)*0K";19M]8#TAV!
MC.&6 T.""21X AT<(0*FX&\== Z&U;B,DMX@A?'2! RFL&::B2C472D!KNL
ML?9%R:9= A;PW"C7*&NT!BT#>H7:/B&)13C S">(P0Y^0 0C& $*B2]#&="0
MH0P7K,-_*%@53P2(
MW$!JUIS^@61"8-4-:,!6!@A 31 A4H 0A*Z((4'"&A("PB%/Z0Q/I#(RUC
M=#09=-1)R2Z:>N$$,$?]/BR9$JAPAR/%.7!C<6K#T#Q('#Q(.L9A'%SI &EC
M9%R/6TPH49I!Y K"&#SA$81@/]I@$T[!%/I $]P@#(P@(B2I0D)A%&RA%UBD
M%WIA%W!*>[Y 0I@ BT9C;=S/$SP!$JZ "5;@"=! Z^9 V&Z+JG8@5B# !%;
M$29A&- &$I9 !;"/ 4" !SY'AT(A@NZM@>RJK_@*LA1.@+Y-?K(P#+]M3;[P
M2^B*L.X-XOX,?2)KH=#$W?RO2\9P_P0+?++$V[!-@^(0@C6[YEB(?A6 QE88S54@T&*034*Q!_:HBW>0A*N
M 4:C >*P-?>H".TC@XT1#&.H!%XX6*( ;YJL?=@8QJJP1JF0?X0!66PH1N]
MD5U&JW*2(9DD!9'49A+\;LPVX,>D+"*<( HDZ23(BR,TK,,B[P\J+Q2C =X
M 6 P!(&1!HQYF1,YBD@QRCLKTI0[W V"AN^X1NVH1:\8 5(@ 0R &P8 13<
MY5J<8;2 [W(L\!.ZH/@:0@=D(&K^?H O%H,E^X(OHH8'I&8RS D$KB_,N&8
M"J !INB*EL ++($3/"$4I" (?* +(L$+L +K"">;,>NDF1_G,0W@B)/<"/11@8[7&\=L /CS(4GSN7UO@&78"A18(@9=)&U2.H(
MCN .^J#3 &$/Z$ -FF<&9H!$BD<+4$H32.$64(<%?4$7:D@&EZ )EJ +'L'H
M7*$6.J$1LB"'C$4D\N ,*FQYFH<'1, !&FS9Y.S 7D$2*@$+5" #&(D*]T,*
M D[B *W=O-!\!(ZPV =.[JHVS9 W\CANF0>9A,Y^0W?
MM!!^BL3^..'-_^"0'1INWH)S#A,(V_R'38CB*@&1#Y'".B#'8R#'A/SISE[C
MA5XC&1S%#@#."@#,A #)Z 0VA@!D( H2(
M 3) !:Y $MY"%(;A9X#F9Z3QAE&!M
M!G "0#&DN*@#N) "J@J"1H!E5()CJB1'%F#&>ILTF"H&EZ#7;*A71;%(UDC
MF8B!+>YI[UZA%C[!$79, VJ&FB(""?@E\=Z C.H1#_8@#_J@2^\Q'\U@Q*(@
M-%.@"X!J)EP(2*]!'>Y&3_#&L'C#H<"23\(E'J!" ;7!%5+^\PIZCA(\0;^<
MP1FNX36F@3V?,B2#AA&*#U5.\C&(0"^HP=@X%(G ^=D12$:H "V
MKP2Z+_P@TQ):8:Q:IPV"X,R$ M28 F6@*>^(";H"#8 1:)&)DX%+MUNDS?W
M\ V#8W[.L' 4$#?8P6X@IP'#(83^Z(\P#E R+EU&:#M.AAD\TFT>9!.1H WT
MH-/Z(!_)(+UFP 5B102J:#^R0 M0Q#!]@5U]X1=:S1:TYRLDY H8X1&0,IA(
M32,6)L1P*PJB!@8< .=08!$H012:R1=_1A124PE4 / @X 2"( FTLS8=3O\D
MSF+AS5>ULU=GTWSDB@LC2U?Y+2O^NY-D>P3_N UC099CJ;,YM3"O>A-_=I5]
M-/:P^-!F;W8XN(UD<;9CY0$>? -9QR4>PB7 $I&T=O0]#>E="B)!8@%J;S 6
M:N$2+L$+P.!SLN!$0'$?I^H&:. %L,8 %F GL2 2$(PMH+8F?&<:#R3D>@^&
MBL&TEB$N*2T7=\9WC@$4%F$8.4 &D@ *5(X>-J_2P(-$;W!MC X2NDL#0$ $!N\'DL9*H> D.F(.
MZJ!@N-1+#89+\Z DSH#QIB (>$#:/ $UXHAN-ZY'.KTBO7B
MG,)C#M#^&\;!&[B!&Y1!&J2A&M8N[D +M$C+4']/08SA$QB!!T @!!Q)!D[R
M!Z2,!J#/!MH74FF RR(#!D)SJV:N 4J@!-9I^Q94VH"@";1(%\K!IN*U"78
M!,#&"AK!$D*0"8* "2*!&PQ58[P!3XM7V_;0KVQ3.GVU?^Y'H7+5*,B!'1*M
M*<)!>)>56Z0BXYZU6R9X6J>5<@G$4KK@5;5B$SR-=M^ >7K@!10@6((%8JNP
M";1@,.\@$V0*=5@MI]Q@"91@!:P@"( @"4J-$UQW818O'@$V5D" "1S!02;T
M9Y#!'(%QUYSM 41 !]RM9O=/C6U39"^6.BNV8N?-.2FVW1#^R&?C&&>Y$^+@
ML&1Q4S>!!/]N\XVALV:OD_\RJ O%$Q#9F/_X$!YJ=J$<*\; LFCO5+-THAFT
M41MS!W=P)QND(1=FH15@015*H10@X0Z\P NX( NJH"ZK@*F6Y2)&4#%D+D%U
M,@-6H"S89D(1-A)1Z_?J[(4HUY@IS;22:6Z09=8156X1*N
M 0:H#)090=0E\+L W,H"/P8$NY%!#ZH \*N@^XU \61L22( A0159GB.QZ
M3^[ 07@%"GD)[2@RZ_3"Q= *\2G^M.5YH9<;O&$;<(E=KB$=3F9NO*'CU/0[
MW"9\:Z8A8,!1I8SP*C5]96#P;%HR>(!<8Z6)&.!3!6#[HM1_.86L6N0%]0 )
M9* AP, -,J&UXM4-F@ (O 68.-0-L8 AP*$QQ-(XNHWX7B/"XY(A,*#FD)X
MNT59OT%=P/%DUJ7T_$1C#D4"L_$M8^AVC"$4&"%VW( 33N$4-L$/+ \*^09<6SN/\PN(,;N#NVD97[K-S8*&BI
M6&UCA.,!&]+!O[9C&]J.&J3A&&A!%G*PAC"E$1KA$1JA"[K "L!@",! "(*I
MI=B@0M1 #0;T+VX@!EZ@MPK 1J4$CYA0J&!;0]DGY#!.UY(PRLGA>(F:4M+
M:44./'217DX@:Z+Y"2[B##(LP]X@FWE@"1H!% H"&N*B;1KD\XK!$9-!9WB\
M;4,4&AQW9_))C&,!P3ZA$SK!$BS!$;S "H! G4AW?JLI:2AL\4#Q!Z
M$_#2H&-W#N" #'R(9H+@$3P!%ZMQ4,%!L\(2+!,2EIB"HQ?^,#O(Q8^>5UMD
M8V[695#40< *!6D/E4$&Y!.6 500 3*]_BDC-''B:<=J7Q#H/J^K(G$C*@%
M ( #<:0F^(!1T(1L$N!=X(13"@ AJ8 9*)Q-T03YN:BND0.=JX1>T8<^E
M4J"^DKJ'@X%FMF0O%F9WG3R;PFY,V%L Q<4.1;\0)5"N05L2L=D/A1DZ"H:0
M0>DLY3&K;1-LP;#] )1P( 9 P 120",IP1&N( 5:4V#GMP0Z49<2>P AN@)025 /$71)>02T2)(QO9[5T)QIN)Q<4
M[ I80 /N&&05V>+-D#M_]H(QG@[^)1D-RTV\)]FZ#1F#^=CB@19CD9.[DT3<
M=/.X=Y7C_=B0$WD\>R22Y>& *E[C=7YF[:0I,FMDXD'H]0L;0AD3,4')ESR_
M,T5"KF>()40+P* *J$ +TL#JEP4.#!P*_B+F8H #)"!4S P(&.$3>.>7IY&?
M:!5;#F4;LX4[N'H"W5,^E6% : %"4J $V+&RGP#+Y4##G@H*'/H(-#(6XB(6
M#J(3H,L_0(%W!X3:Z5,^?\%QD6%M^@X2+ $2\EL3-*$1OH"5-0T(3J !#N !
M2$ RJBD)"$/QRL#R/(P/]J /-@$0.HWV"[O++X0-/G3QY'#]J!"GR
M8\F0(CF&'+ER9GKXT":*%$R=;FSCY@3-FRHP,#E(P F5,EC]0E!8QX9&A
M@0$%$';T*/)$#9L[G$C]ZD707"^P=>K,F0-'S)@P2(K\D-'^X4$#$$H@?3*F
MC!@Q9,B40?MEC!FS:JB-'>-\[-BK2%A(JIS-4K;)C2=3F@0YCR1(EBI/N@P^
MTO9'>/!VU]:87+;+W,51ZGX>7'9S>-4_D@,._5WRZ+=_[U9>4GQ+XK['%\\-
MDE_ZX>:5HY\=?WUXX!O9O1/';MPZ<.NL@TTY V9CC">/="%$$$L%2221!BB?O"+^RRN4=)$"$$ HL40CCWB"U2RRT!(+,J_L
M^0LTR CSRBN=/++($DL$<002C#Z!1!5<< $&&$>(<, #$(2@@PX]$)&$$4Z,
M 8489< Q1X+X
M]\XZ&EUT$4T8B7-3@.6$XRPXX-PDD3?6"KO.-=7BE)-$VB3EC3+-)&.C,9\P
M@0(((H00 @PPZ!"O#C'4&\,(,(P0@@@@;."O!@TPT$# !QP P,$'C-!$([SL
MTDTYW=C"B1M)V%"##57<(5 OOO2RB\2<6-@#""I HTVS5C3;;'[L:.?1Q[9
M)IUSOK'^A!QUY;74T3OZD7/1.C2%0RTX1'V3#3;93)---ML;"-1--[Z 2,=89HPQQA%!^$#"P!F@<(4DP@A#C# X
M/OFD:: 7DS4RFQ.C3"RBC#=>;ZJW;I)[TME\G.H<-?<.Z^R%=Y]XMJN7W7C\
MP)<;[.G),P]].L_SG7SA'>_Z[[WOAMU[)4W_O''EJ5>\2]_-X_S,)F$'/NVN
M"Z>1RQ:%$V \\1 U"R-79+%$$UEHH84;^(?B1BC\U\&)_P#^)!R($C.'-Y H
M"E$HP@Q@ ($'&* &C#!"ASQ"7\8PT:<6X8&4S:-:O3(&D2YAE B4JUN8:N$
M1!L*-L!A#:1$S6I7,X8P8A$))6@@ 1T@PA-&98=3U0$.;5 #$HC ($JH"12+
M0,&;XJ2$("A!"4Q@1"1 $8M8#$H4J!.&*'XA"DG(!5%::,+]!,<&-ZRA#6W@
M7R."X, -B&!3>B&"$YP0A2Z5P0YPP,.'-F&*5KW*%X#\F,3T0(U@M>, A!!S8 @0T\X)@#8X #
M&! 0! !'0P)SL]+!N[()_6DB"#(B0,5+<8B ]< WOM95AS8;4:6S-P0DV.($(5@2"%4#B%1+U#&@8J@QM
M, ,;IH%J+,T%FLP)(WOR+!_YLNH\K9+'JV -:SVOE]7;C!6K8@VK6KTZ,Y2<
MU9YL_>K^6G575I6X;#_- E E"22+1LRO$:%H QT$RPDZ>.5_A*M#6#;A(3YP
MB$.-@4("<4 #"#20 98JRU *,S[HRA1B:RC#2NTHNS6@FQRI
M1U Q33*4$:A(I, !#QC!3,,@AC?080X;@H,:G% $!GG"$Y980@R"("$I/"@(
M/_B!HIK@A49 HA:NH 4L8O&+6D"B"TI8&/]"H0><_H\/>@@+X4*1A X!M-=(K"LOXPYL($]L %,$ )&K !'7Q!%P[KAD%"<80?^. '9N+$
M+7I!95_AM;SJ ,N#8,+CRLH$-8GB"$W!P
M@QB@A0%?I@2$0?.994R-:M4@BFFLT8QIB):V61-&,NSRV>?
M:>-NVL]^JU>O/>UFRW4WW)9VFL_^\[),"DT=Y<@&).87"EN0C1.-=6R'YOW8
M.H1)#G,(4V-D/04:+! "!@A #*P@D5,@K/#Z.P%LU8NGV1X 3>DN'K<. #X0#LBVX,F"#6Y,7_[, &+OO@!$* 1"Q6$Y71@E H
M_42E."IB\8NOX^+O4%^Q1EP19&4\'C]SV8FM%0Y8*F4J4^K""4 WW>)P)SO
MVE<'X$ 09V" (5="$+1QFF%N@8A-M
M8(,4N,R#R:-@!9&(A9-4IK+"R^1E&ZE=/->LO;F:VU)(*T<-#"$DRX
M:!.$P.@K[%D6T2@I,O9454 %2DK%\(RBK7"$-8Q]$WT0+A*2P ,25$[3O##0
M!7EEHQ:K9DK#( J3H B*L (B@#<-@&J+$@9J0 >25BNSA@,SL $#$$&+@#FQ
MH#DX!O,] ! "=Z Z "E3 H,M)9HL,,.M)"2%%\I%1Q&(9A
M,J$LQ[(.Y1 /&2<.T8)"(J@4U\ ,S_ ,O 8))+U!>>M &:; #=[$!\24#\T4$H/($8_!U9C '>8!3IT![!%$/R%@/Z28Q
MFR '\A<$)W "5U +O'(UI\$C*T1"U$(1JU5*.#%]9(9AZN!A&!8/Y&".[[ =
M_:%XPV(D3%(NLR0+BS!Y,>8NF9+^*1V0CQQ 3,AD&080, Q0,-!D "<@(="E
M VV "9G@!D=0+S^0,9F09.9@9:80"K#6"#('F*SWR@>%@#7U)
M%"WTCFJX#,F05;?#?&H5$N$V@SM85O(0#]1V@\WGF#A8@Y5I@W!UF63%5BUI
M;M:"#=3^( M=P .-X&Y@P0=R$ >J&0=GT)IFD :PEE_Y)2I1X 1&X',_4 ,B
ML %V<0 %((8H4(8)IVQ2@AI)DPW4@!1#,TJ9I$IE1I+EAA&BQ#[F*$K_$6?.
MD [-\ S.\ S+ W%, R=@ 4G4 (<@)95, :FD@=X "8>]98T$ 5DT$.M:0?U
MB09O( 8D(@7SEP0]\)\[D'5NP FA4%ALX <(^H2LN1BSD@8VH (D"GQI76W
MN4/ZM8AM@%.\0!COD(SX@ ]XN#2;@ =UP 904 4^, )&)0M38HW!-B1$L9R4
M-$HSBDK?*"TC9A'2B8?IV*,]JGC.>!VS@3_($?/R@\S&:9Y<$.,>&2FT1)((@TH' %2] %LJ<+"2$_
M/+ NTF@)LA"4,T24@C(,KS ,G\ (2B $69!>IM '?? '9R &1F #(5 Y!E<,
M%L0,GG674>$-"T4561-#*44)E? /5\( 5^H"3J &=I 8%F($55=4E# ,Q/ *
MPL!4'0B"W)(.HJ1Q_N0-,SD-((<,8;68Y8.MFCF#VOHZ8*6"T+:MJM.MX/:M
MYDIM[K%MXSI7RI.8X9H2+^,R 7+^$<*R#:(9 S/G%7@P!V> !OV*!F@P!@'K
M*+9I!%-@!#B0L#=P S90+QZP 4.V L0 1([ "; HL@"M P#-"0#,:)- 1B
M+>O@3LNB>.. ?'<%-#SC$?!$G8C'+.H0L\&2#L]P#MZY#,S@8EA B5@ 3:
M V,@!JVIFHR1!E)0!$2 U'@KZHYM%8X!V@ !QAB2)KHGT ' S,@ SWP*4:0
MGHXQ!F8 L '[&$\@ PJ0 G@+CJ@M?1E!$\0!6-0!F5 !H\1!FTP"KI@#LCH
M/?20#\A8#KNP"2/Z:EH@!"=@ I?S"M4@.JB!#=SP<-[ <> @0B*4#MB0#I/;
M3S1*CI[^) Z(1YWE-$)&V0',[4'\@H $=N0)8$ E&=*F8^@D)-R.?T%) D 6:P FF ']Y
M< :.
M3Q5L/5%"?W>R0*-X[( ==P5/Y'N2[S"^X^N<-Z$.P)*=W2D5JI$,I;"S)> !
M-? WO64'JDD'LQ)K40 %BP$FJTFT<8!@=E!(;/ &Y 0%3U $_QF],S #T4L#
M-Y"PQIRP1& #/_ !#1 !!N!&,;"V-E $;CL%:""W93 &1E $&9-Z>CL/'YH/
M^7 \W 6>C 6:5 %/W "#K89"Q?^%*B#2W$+41CN7<8#GM(+3=:$V5&
M$QJ!CI"9?/)PLOWA+)3TCK3%)TS"PBA/UHP?SL0 RX ='(!-C+L$##\=#U.HA/I0<(*0DO\5F2T" J G1RT30!$?@ _="
M B$]U&J9QR!L+#:@3"5Z0 C%P U(@(@#T!D7P DM "6-MK,H6&A\$
M@B.[#O'^ "WE3
M/,GDFZ?YH1^JY!]_1RV4.X)-837%\ J2@ 58D@$Z$ 1/D 9OT(@_),%+6P>J
MB0>I.O
MC 90(+<_A0-%X :G\##(F _X(,[Y8 _CH-*<,(ML0 9.\ /^+W $E, +Q9!!
MLZ4-317"X0(.Z9 .UV"Y2W,TY? -U1(@EN0L\ZH?&=&2*_L;QLW93Z'DM!4+
MCJ ""]!C$K ! /-Y#""Q#B"Q:5XP$1 !#P #"ZNP#"L#,_ "+I!+/Z %GL -
M2J9VG%! 4N $*'* &J "_^ (DB C,N)93V)L;)A:<<@^XML.R-(.[1 /1GV^
M7"S;<"7)Q_(.G_YWK41L5K,(*G "2Y &3D #4U &>ITR3 (Z.4N8X6DC
M 2@*E\#"@0X'OVIO<""L2A CPY#^@9Q#-0I%)"/IR/.J2MPB@D9A&KXMV^&6
MKN&&.]ZSV[7-VE0,\*.NK:P#W%0<\/DN#_20\*VSQ:/.Q+'- <74QRG@"*61#._(EY?$24"]'NV OB+1'$OOY<>B'SDZ-,"R
MWU7%15A@ AJ :7P!!6@0!QP2!V7@]:DI!P[.GG&0!ZJ9!W,PRV=@R]C\!A02
M!;)F!$3PEB_0 0E@ 'Q_ +G3 0 ;P )JBM3U 1U%@S7,K!K49!@5*>WM[
M#_AP#\'^TPLZU0=CT1C:Q -7 H<>Y=3PPV/^[C <@Y_B#1PLPO9P&'JD Y_
M-X0D>1&G3M <\?2_@1'98@U0LCF0H (W3]$58!<2BUEQ+N=3&@&7$@(_4,QM
MB[1['@-Y3])-\ 6:$ J-X 9V4 8F M<-E %*4 F4, D']PHR4B/5$$O%=B2O
M-#0Z2H?OV]X!;;ZD/0ZT ZZ_+3WY=,DTP4)#.L*.D ( <4('$1DTILPY==NYDEBRH,F3%C/J#]NL5EA0RCKRI$[7^CIDC,YBX'#:L
M&#)ERYCI!(>M7+APXMBQ&R=.W+MUZ]2& P?76K-FRN3-NRM/[]Z]>//F]RT_ON';NVM($""QR@0)YBA1(LS;$P8K1"
M@P Q8L8)#@P?8'#AB86/2)&;.YUJYYNQUNW=G9[."9AA>[77QY]$WO?=?N
M_FS99]F)(PL<<-0!YYJYE$&0J%A$640%$$"0@8@GWG@#CCKHF$,,*-"0(PX\
M/L0CCCSBZ#"/#DF,(T4[[#AC13O^S*!0#1F?>**((F28X04(%(B@1P,V ,&W
M#3H8(089;K#1B2C$8%(,-<)(HPU.>.FE&W.NK"=+<^KIQ9=3-MFC)#.J*$('
M(!KY1)AAA$&&JYN:F>:K9YRY9AI0'&FDB29""064;-:Y1L!PQAG4/_YDNP\_
M1-^))QYQR@%PI[H0% :2% )@H($&@F/ :N^_33ZPXH(((#$A!!AQV(P('5
M)(QXM8@DDNAAAQF,U.&''W2HX:$:0GB -Q6PB$222281Y9-7LGK%J&22H>M-
M:P2,"ZZWQ'&+K?4.96?11.5)--S*XCN4+0##.4\;!)=)1A(L3 #A 0AB>(*3
M6W99J(W^-&QX@0@]--'DBRR:$**)+IH(8HDT_-C$%%/ZH(.--) @8H0'1K@"
M$F.0(0:99F8:#YOSK@E4P&D+Q&8N9A"LJ\UBU!1&%$94D"$)->2H P\YX*AB
M!B$J*28KCI%15QMKL"'++6S'Z6\V;-T*,*SQ6.NL,]!(Z^N=QA;+"QZNY5'M
MZ]7F\9IJL &+SS"S ^-Z,;'O*JQMT;1^&Z]Z$%-[
MM+@W$ZQLQ0Z_C#'+\ H-\<$,I6T"TP $%$XX(844@ ""
M"4;PA*0325Z1)99:7HE%DA4R""#W 0XP( $#&M# .P>8^*3^&KI(#G10RPV-
MS;7(]D.TV[;*6D<= N?RYB:B.([%D10T4""&))Z @@PXZ("###+*J$,.$#UT
M'T420TRQCA3CF,.._.%X0T8GTT@#C9"0!"+,P 4/0!7I$L ["'@ !C,X4A&,
M (4Q0($-;) 1'#3!"5OLHA=7 B$(>W&+4X@$#WF @QJ>L(,3+ $2PHB%,#KF
MIFG4A2[-8,8G'+&$)32AAT%PA)\&1*VGK8,_9WG/8=X!#^EA"RX&4A>"7B&)
M2S$@4\+QU'6T>)U2E:H!(,H )%!")22QQV,=RQ_+THI1BL'^+FB11QN!P@:UDA:/LF ++=ABBVR,*(]Q
M\$4V]3'-._CQ+7FTXS69I(W2!@4.;=RP*\2(A.<*8 (-($3G-C$)AA2!1ET
MX >:4$@F0M$(+2 L"#]H0AL:)A(_L"$,5?A!#"" @B5T@A;'0$9,O!(R\YQ,
M0.J BX!(YI-H)>AEPA#&*RJ1@A@.,:
MD()D]([8%D(1B&38<%Q?_$8XL^6MH*8I##T":C7+&-1YA/':UP(ZML)$SJ%8
M6VC5$.JVPJ6&-1]%S-WTHM#!T(=L& WI126*FK_8S:$3G2CZ 05 &$)T($$*#02#5EXPABQ.\8Q8C'58\B0%K7[
MAP,& ( K#(XP/.."8B'#9V4;!RUZ19L9!,?>-!G;'E3G%Y"XTE$Z2>MZR'4
M.L!13VY,2AEL0@8O&H$""'1 !D5 A0J] 8-S2%G.O/0B$+D/CETR++WBX,=
M\*<_"I%!1I]5@QBD@(0F(.&7.H@!"0Y@J@0HH ,OD($-;D $)Y#/21?40RA"
M@HI;5*D;ONA2EWQ!PH>=T QP"$,2>."#14PBAL0@AK,0]*SQZ%!UH;!$*+[@
MPT88(QU$5,?3TKK$)=:GD_4AEVS4L@ZY7(,;VA.&[01PG4W^K;(XQ=EB 0+0
M(WF-0 =#^,$1<& C(Q2XP$A <(*/D(0?R$ &(^ @YP !)8(1+%VB,?7R&*
MK&3E9@;Z5'/>O^C-*9URS3CX$7'UU1SMI2J#6XJ_0Q-6U.8N+[^%6NN9IO8Z!89>+@4U@X--4$+)S>Y;AK5
M%P6IJC/-EUSO!:?L08L[>&H] ?GD&,90EE.-40UF5*,8TT[&M*/:)C;)$)PP
ME$16!T 73*4PY -PH6,8ROV%-0:1DO>ZU:A&,HR2#5E H@EI" 4O%A**)B2A"99@1O+>
M?-;VS)C^K<]CM%H M-<>-Z,8M5!" +A('+HOX+[7Z5$$6+G[)-"(4T^"5&$ 5N4*2T,"(7
MNXQ,6@='"9"4^0IEB(578 +KF+LMNHZNTJ\(6("#0ZT:T)68LQ$!(A]!)#DH
M> (I$" DJ (A\ $=(!T>0($48()_6 1*,!91,,AAR+R7V#P08Y?Q&(_CJ2=_
MVZMP* >QR#%LP*O:6(O_<#07DSU$ 9PZ%KNS9_\ =0N!,ET( ,
M. $?*",C( (;D($74( 0^ )/Z* GXX2(T8,LVP0Z2 -DT@$(R @< 1/H(5"
MLR&?4"3=4SNU,\.UHQ;Y.S_@DR%W"8$?4(,^TYDY."828 )":Q.6,0^D60MR
M0 MC@P_^<#D+RW$+D\E!8&-#D6+#$_1 #&2,JE&UE0+!2ENI%?Q+N:E!3VN-
M"^Q+D#H-DLH;P>&+ND2I#&3!6 .7Q>A A:(IQX%!P)BH#G2<48M!PBS!V'@/
M)NH/(23"(OPG;U S-2,/9J"3\7B6VB2*[4&0LX2&26 "$]@J 0@ 1B %B"!
M11"&GS"]MI"->$ 4MV(-S:S!"E2B&?-(M- ]!QP/KU@&HS"&57 $)2"!#9@!
M'(@")Q@#-HC$#YD#%)&LBT-$1K0?S<*?_#F#282#,X 1]5&L-T@#*="")T""
M!;L!&YB!', "\B!%YB!,3*"*CC/.\BYD "$"04$4ZA0AXG^I5K,@PT] S+H
MLAH B\ P&$@*)&/83J%P2+IH-YRX
MAF_ AG@0BXKD4[40BQXE!P!A3DF2'FZ1O>K$JW'H/6^HH61S/-->&8BB]>4%O
MU1K ^4"K$<%?*U?1.!R.6IO6T$S2L$Q[,QS(^$NXJ4N_J(=X;4&V2'?$CM29 Z(XJ9F(1_, $-\,*79 $L$ 4=V[%J68ML
M+-2VT@P>%$QZB(>M8(,Y@(,\"!&=
M@:S*P@,_T[]'))$3R:PY.(,SB(,S6$_\1 /\I) R #E!O$=6>8B;S(&RG8$9
ML $<<%#^-O #/^B#"7U;6GQ;39#;/MC0/,#/,3!*,7,JE0P:HQ"&2^B"( B%
M6_BM4V ()- !'O "83 : 5FO:EPK>8B J_9([
M)%W2)BV !^@ $0@!*8VM&T 2"2J"*(B"> 2Y2H2",H 1QA*#,B@#_H&"38P"
M)'B"6MD!'0@"(>@"+V@$2)B=91E&[BP&*627\W/(G1"+;#BQ@#;/0%MLP
M&6](M&?9SIAAA!'P@2?HL_;Y S88 P;^A&-(AL_KJY)II'+1"^>,/3F$,W#8
M#,XTJ%C3UY'BJ,7D80_D-H'#/,)N0AF70X!^11,VOXBALBY+K0AFBBB6$@5J98 4:@A%C@AMUS
MLT%1SOM@JX@B8L^DATW>P+CY8K)9HOA@FMG(5G2Y!C2+"9Y=DU?H!"] 0Z8
M@2EP C& @UIVK,MBQ,IJG\IR1*G^E<_[69&KI=JJ/0,TX-V."SDQH+DG". J
M ,)N@$<*5"S3=LJ" .VI45 <-NW)06YW8-O1EHX8 ,C& +C#0]C>*>@&09_
M^ 1&0%Y.^"!?.+(T2 (P8H1?>%QK&=_92*(7M"3F9,YS6=^6B05*6($!R)W<
M$=T!*(X( ( ":( 'V #595T'LQ&9HY'9!3D[ +DWX%TFR4^.(]H5V<22[KH@
MX $=(($3X $KD#Q*8,EGC043W1A:;6'M(8_7S(9MP(9LZ&G,V5.R>)1R2 ?M
M51H;]0_*":4+EN2(;89A!(7!;8(O<(,\ 8('((X2>($7B($>Z $9>"/@B $=
M."PH@(/^838#- @#!?8 $5#*9R6T8@@Q9? W])C6I($DG6(+M-!C^/,WG:#-
MHLB-I0N#$L8#<7:"#UB!%2Z_0VN&0,$FR=6D-GP-VL@F3G:<<,WD-+X;3RX<
M,I9B*_[A?)UBF5+7S3QMSF:,'$SCS=3ATE9MO(ABV?;,C)H')";,QTP-,W[M
MVO;M+T9BQ@#-T#;8)Y;.X,Z+<%U!L*'M+0Y)?6H'_^"GIHZS0+F&/T:D0#8:
MG9 _''(&:Z"VG?!&49"$6A &9=@QG*7&VKBD0C7!OBQ!)\Y7R9GAM#J+@P\A\# X GYD@DY8YZP0
M1DI8 B' I5[H!5/0 S>H A_8 !5XA'EZM^69C>;1"^2F7-MK%$9B+U(J)62(
MA4Y@ @"H#H2N#KL+@"8% .0:!'H@!"84@BR :_N@<*+ BG0TI&[W?4I S20
MASTM:A\-D+/BIZ26;C/$IO0EF4-BAFFHACMA@B2XLE>B@S6P 0Q@
M.0X( 0B2@1W8 4_EG0F( 2=0@_S^V=HQ& ,;23D(X %& (5AB(6L@ ED^(JC
M2=FDJ<;Q]8_ULDK>.R1LJ(9K6 :N$ 9'\($40 (+640X& ,16 %0((9DZ*M2
M E*A9I1+(M?S>H^S4 \<[N1YV.+1OHO@Q@M,ANU];^W-7D'HU%?'9.+:YFW7
MGBB76BA.=LQO[>TO=DSEIO?G;'@T)EC1-.V\,.+B]F=^@'B]S'?GCOAZSVV*
M5V,OMGC).4V]@#1'FAXX&Z4X4R3KYN.9%U)L^&Z2X8::^IKJ63FG.%/
M@^,BSK2(W^S @)[[=K-39H9GL8! 5^ $SFN5:OL](Q"RI39&+NY\Z
MD,\3>7#^J\5:"?^#.$>#M.>SCH8"#0F#VCJ)OS,"#P?Q%PCQV"*" Z#-;@#
M"/7F$#$#8T*"'QB!\/L$=E[GE>P"Y/T%X>($,4"F#] +*B%G%>T-U/.;K$Q
M(?X+G&IR4[XACOF$*[#R ,!RN1-=)Y7H(0F!#^!J![.!BW['-/]:"W_SM$_[
MLH_S2-0L%_%]"@&Y*, !I=N!(#B"+/""+W@$2P"%J3)1HXB)9KAV-QG 0SOE
M?TH'/@7?H,:F.