
November 2016 Developing Well-Differentiated Antibiotics Exhibit 99.1

Forward Looking Statement This presentation contains forward-looking statements regarding future events. These statements are just predictions and are subject to risks and uncertainties that could cause the actual events or results to differ materially. These risks and uncertainties include, among others: our and our strategic partners’ ability to obtain FDA and foreign regulatory approval of our product candidates; risks related to the costs, sources of funding, timing, regulatory review and results of our studies, clinical trials and regulatory applications, and those of our strategic partners; our need to obtain additional funding and our ability to obtain future funding on acceptable terms; our ability to commercialize and launch whether on our own or with a strategic partner any product that receives regulatory approval; our anticipated capital expenditures and our estimates regarding our capital requirements; our dependence on the success of solithromycin and fusidic acid; the unpredictability of the size of the markets for, and market acceptance of, any of our products, including solithromycin and fusidic acid; our ability to produce and sell any approved products and the price we are able to realize for those products; our ability to retain and hire necessary employees and to staff our operations appropriately; the possible impairment of, or inability to obtain, intellectual property rights and the costs of obtaining such rights from third parties; our ability to compete in our industry; innovation by our competitors; and our ability to stay abreast of and comply with new or modified laws and regulations that currently apply or become applicable to our business. Please refer to the documents that we file from time to time with the Securities and Exchange Commission.

Difficult few weeks for CEMP shareholders Share price collapse following manufacturing update, FDA briefing book and advisory committee meeting Investor uncertainty around approvability and potential market for solithromycin Significant management credibility concerns We will work every day to restore credibility with investors who expect: Unquestioned transparency Strong execution Our focus: Solithromycin approval Cempra committed to ensuring safe and appropriate use Drive progress at Uquifa to meet CMC requirements Advance pipeline of other anti-infectives, including fusidic acid The Path to Success for Cempra

Cempra, a Differentiated and Growing Company Focused on Anti-infectives Cempra – the leading antibacterial biotech Cempra has submitted NDAs/MAA for solithromycin to address significant need in CABP New macrolide is urgently needed - resistance to azithromycin and serious adverse event labeling of levofloxacin Validated by license for Japan, partnerships with BARDA and NIAID Patent protection extends to 2032 Cempra has a second antibiotic in fusidic acid in Phase 3 - ABSSSI and refractory BJI Only oral antibiotic for long term use with large market potential Cempra owns macrolide platform technology and discovery programs for non-antibiotic anti-inflammatories and motilin agonists Cempra has a strong team in place to support product launch, including: Commercialization, Medical Affairs, Regulatory, Clinical, Chemistry, Finance and IT ROW (except Japan) licensing potential is still open for solithromycin
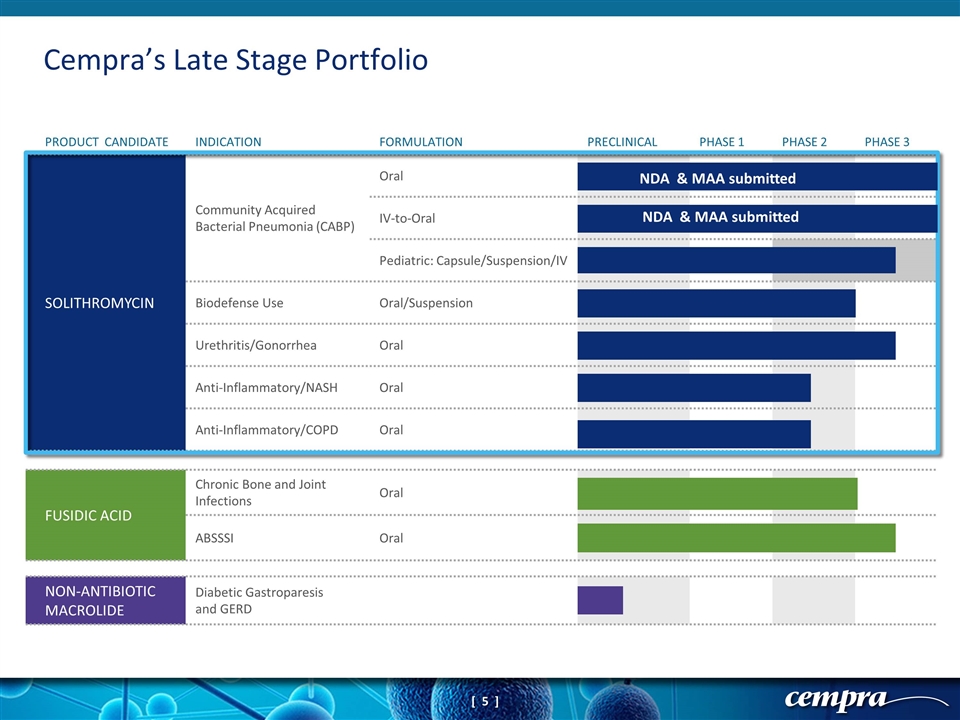
FUSIDIC ACID Chronic Bone and Joint Infections Oral ABSSSI Oral NON-ANTIBIOTIC MACROLIDE Diabetic Gastroparesis and GERD Cempra’s Late Stage Portfolio PRODUCT CANDIDATE INDICATION FORMULATION PRECLINICAL PHASE 1 PHASE 2 PHASE 3 SOLITHROMYCIN Community Acquired Bacterial Pneumonia (CABP) Oral IV-to-Oral Pediatric: Capsule/Suspension/IV Biodefense Use Oral/Suspension Urethritis/Gonorrhea Oral Anti-Inflammatory/NASH Oral Anti-Inflammatory/COPD Oral NDA & MAA submitted NDA & MAA submitted

Recent Events October 27: Cempra announces manufacturing update October 27: Cempra announces completion of enrollment in Phase 3 study of fusidic acid in Acute Bacterial Skin and Skin Structure Infections (ABSSSI) November 1: Cempra announces $10 M milestone payment from Toyama November 2: FDA publishes briefing materials for AMDAC meeting November 4: FDA Antimicrobial Drugs Advisory Committee (AMDAC) meeting

FDA Antimicrobial Drugs Advisory Committee (AMDAC) November 4, 2016 Majority (7-6) of committee votes that efficacy results outweigh the risks for the treatment of community-acquired bacterial pneumonia (CABP) Unanimous (13-0) vote that there was substantial evidence of the efficacy of solithromycin for CABP Majority (12-1) votes that the risk of hepatotoxicity had not been adequately characterized Committee discussed variety of potential approaches to further characterize the existing liver safety information Committee discussed unmet need for new antibiotic for CABP

Strong endorsement on efficacy of solithromycin in CABP Concern on liver safety data and appropriate use if approved Cempra committed to ensuring safe and appropriate use of solithromycin for 5-7 day course of therapy for CABP Next step—discussions with agency on their feedback post-ad com PDUFA Dates: December 27, 2016 (oral) December 28, 2016 (IV) Path to FDA Approval –Safety and Efficacy

August 2016: FDA announces import alert at Wockhardt October 2016: Based on meeting with FDA, Cempra believes FDA may not allow company to use the API produced by Wockhardt for approval or commercial supply Ongoing: Cempra accelerating activities with a secondary supplier (Uquifa) Transitioning from clinical scale to commercial scale Next steps—Continue discussions with FDA and deliver Uquifa data Current Timeline: Data from Uquifa material to FDA in several months to support launch (2017-18 pneumonia season) Path to FDA Approval –Manufacturing

November 2016 Developing Well-Differentiated Antibiotics
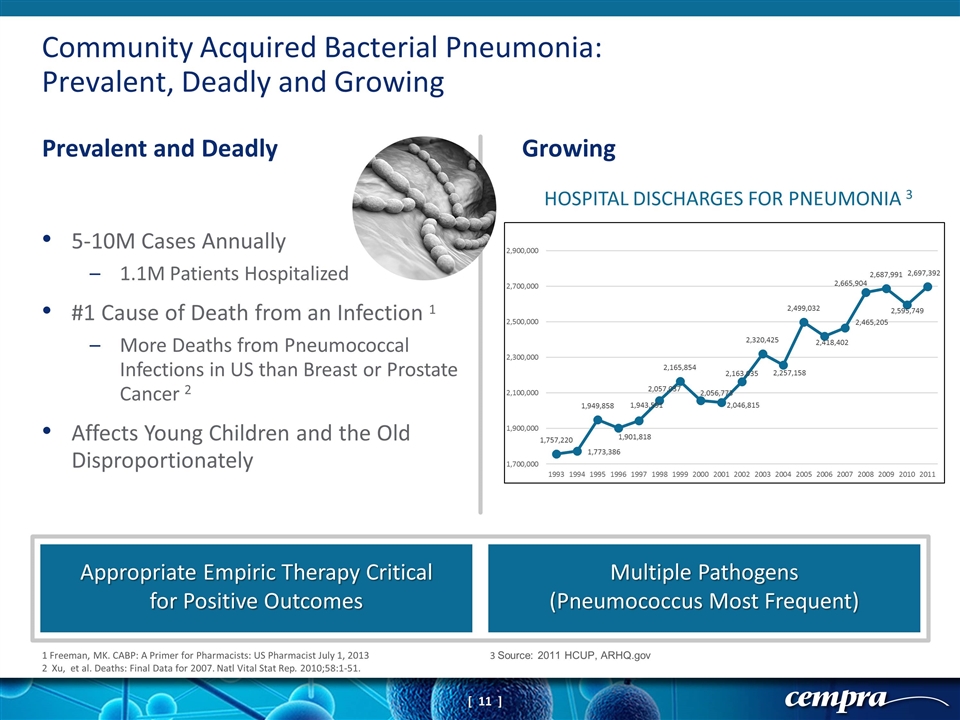
Community Acquired Bacterial Pneumonia: Prevalent, Deadly and Growing Prevalent and Deadly Growing Appropriate Empiric Therapy Critical for Positive Outcomes Multiple Pathogens (Pneumococcus Most Frequent) 1 Freeman, MK. CABP: A Primer for Pharmacists: US Pharmacist July 1, 2013 2 Xu, et al. Deaths: Final Data for 2007. Natl Vital Stat Rep. 2010;58:1-51. 3 Source: 2011 HCUP, ARHQ.gov 5-10M Cases Annually 1.1M Patients Hospitalized #1 Cause of Death from an Infection 1 More Deaths from Pneumococcal Infections in US than Breast or Prostate Cancer 2 Affects Young Children and the Old Disproportionately HOSPITAL DISCHARGES FOR PNEUMONIA 3

Solithromycin – Spectrum of Activity That Addresses CABP Pathogens GRAM ORGANISMS SOLITHROMYCIN AZITHROMYCIN CEFTRIAXONE LEVOFLOXACIN or MOXIFLOXACIN Positive Streptococcus pneumoniae P O P P Negative Haemophilus influenzae P P P P Positive Staphylococcus aureus P O P P Atypical Legionella pneumophila P P O P Atypical Mycoplasma pneumoniae P P/ O O P Atypical Chlamydophila pneumoniae P P O P Solithromycin Has Class-Leading Potency Against CABP Pathogens Overcomes macrolide resistance that limits existing macrolides Azithromycin Monotherapy not used to Treat Moderate to Severe Pneumonia – Potency, Spectrum and Resistance Allow Use Only in Simpler Infections or Add-On To Ceftriaxone
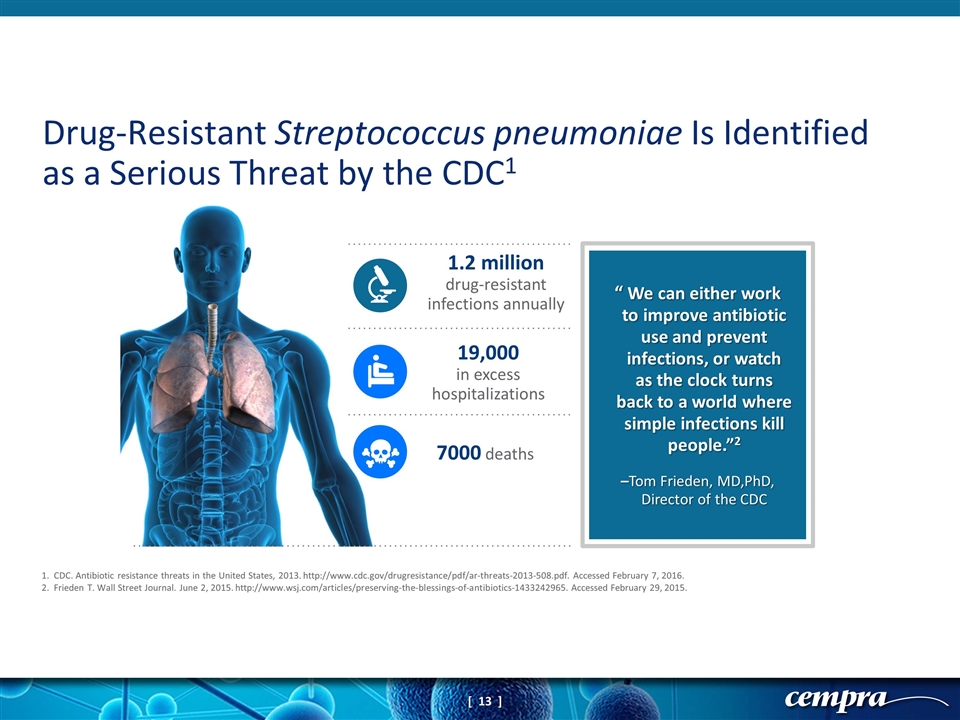
Drug-Resistant Streptococcus pneumoniae Is Identified as a Serious Threat by the CDC1 1.2 million drug-resistant infections annually 19,000 in excess hospitalizations 7000 deaths 1. CDC. Antibiotic resistance threats in the United States, 2013. http://www.cdc.gov/drugresistance/pdf/ar-threats-2013-508.pdf. Accessed February 7, 2016. 2. Frieden T. Wall Street Journal. June 2, 2015. http://www.wsj.com/articles/preserving-the-blessings-of-antibiotics-1433242965. Accessed February 29, 2015. “ We can either work to improve antibiotic use and prevent infections, or watch as the clock turns back to a world where simple infections kill people.”2 –Tom Frieden, MD,PhD, Director of the CDC
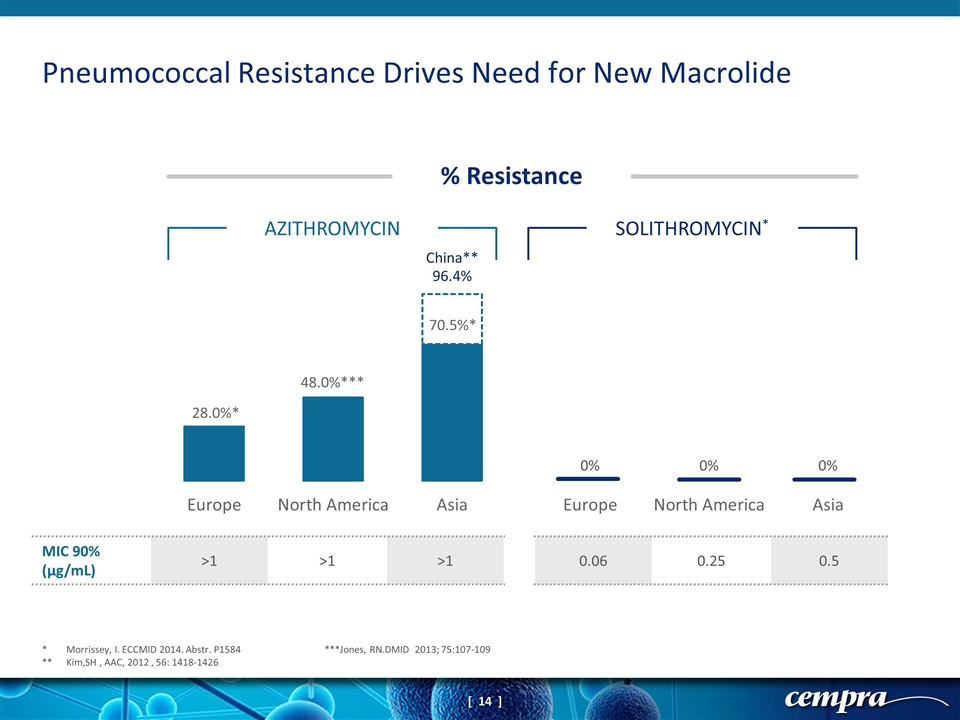
Pneumococcal Resistance Drives Need for New Macrolide * Morrissey, I. ECCMID 2014. Abstr. P1584 ***Jones, RN.DMID 2013; 75:107-109 ** Kim,SH , AAC, 2012 , 56: 1418-1426 % Resistance AZITHROMYCIN SOLITHROMYCIN* MIC 90% (µg/mL) >1 >1 >1 0.06 0.25 0.5 China** 96.4%

aIncludes Alaska and Hawaii. Data on file. Cempra Pharmaceuticals, Inc. Macrolide Resistance to S. Pneumoniea is on Average 49% Across The US MOUNTAIN 31% PACIFIC* 42% WEST NORTH CENTRAL 46% EAST NORTH CENTRAL 45% MID ATLANTIC 48% NEW ENGLAND 43% SOUTH ATLANTIC 53% EAST SOUTH CENTRAL 61% WEST SOUTH CENTRAL 64%
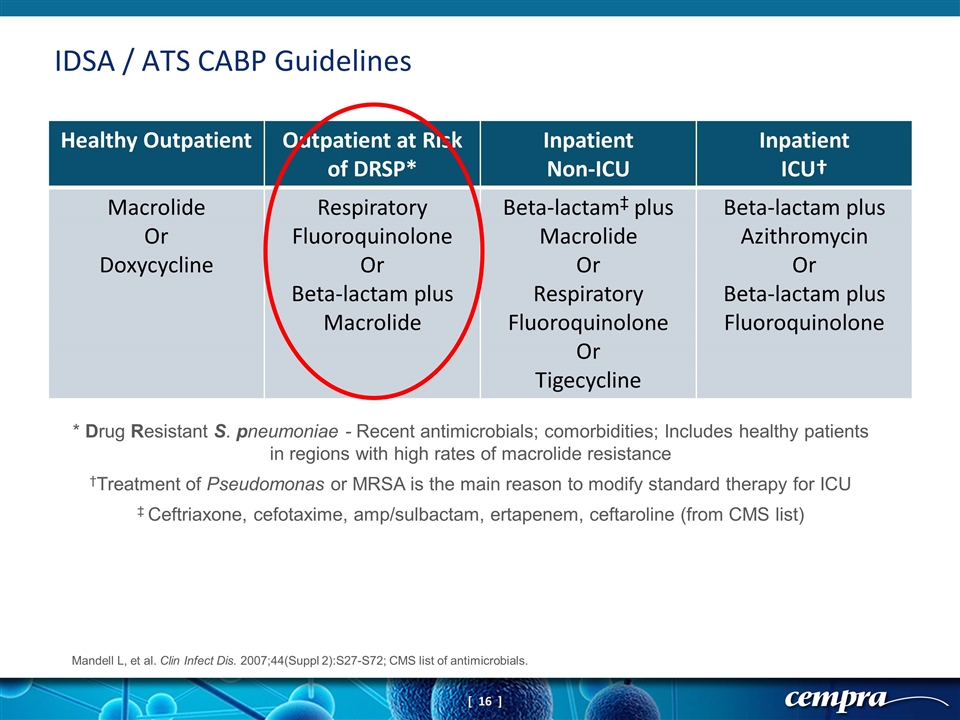
IDSA / ATS CABP Guidelines Healthy Outpatient Outpatient at Risk of DRSP* Inpatient Non-ICU Inpatient ICU† Macrolide Or Doxycycline Respiratory Fluoroquinolone Or Beta-lactam plus Macrolide Beta-lactam‡ plus Macrolide Or Respiratory Fluoroquinolone Or Tigecycline Beta-lactam plus Azithromycin Or Beta-lactam plus Fluoroquinolone * Drug Resistant S. pneumoniae - Recent antimicrobials; comorbidities; Includes healthy patients in regions with high rates of macrolide resistance †Treatment of Pseudomonas or MRSA is the main reason to modify standard therapy for ICU ‡ Ceftriaxone, cefotaxime, amp/sulbactam, ertapenem, ceftaroline (from CMS list) Mandell L, et al. Clin Infect Dis. 2007;44(Suppl 2):S27-S72; CMS list of antimicrobials.

July 2016: FDA Issues Updated Boxed Warning and Limitations for Use of Fluoroquinolones USDHHS. US Food and Drug Administration. FDA Drug Safety Communication: FDA updates warnings for oral and injectable fluoroquinolone antibiotics due to disabling side effects. http://www.fda.gov/downloads/Drugs/DrugSafety/UCM513019.pdf. Accessed August 12, 2016. FDA approved changes to the labels of fluoroquinolone antibacterial drugs for systemic use, ie, oral or injection Fluoroquinolones are associated with disabling and potentially permanent side effects of the tendons, muscles, joints, nerves, and central nervous system that can occur together in the same patient As a result, the FDA revised the Boxed Warning, FDA’s strongest warning, and added limitations of use to address these serious safety issues
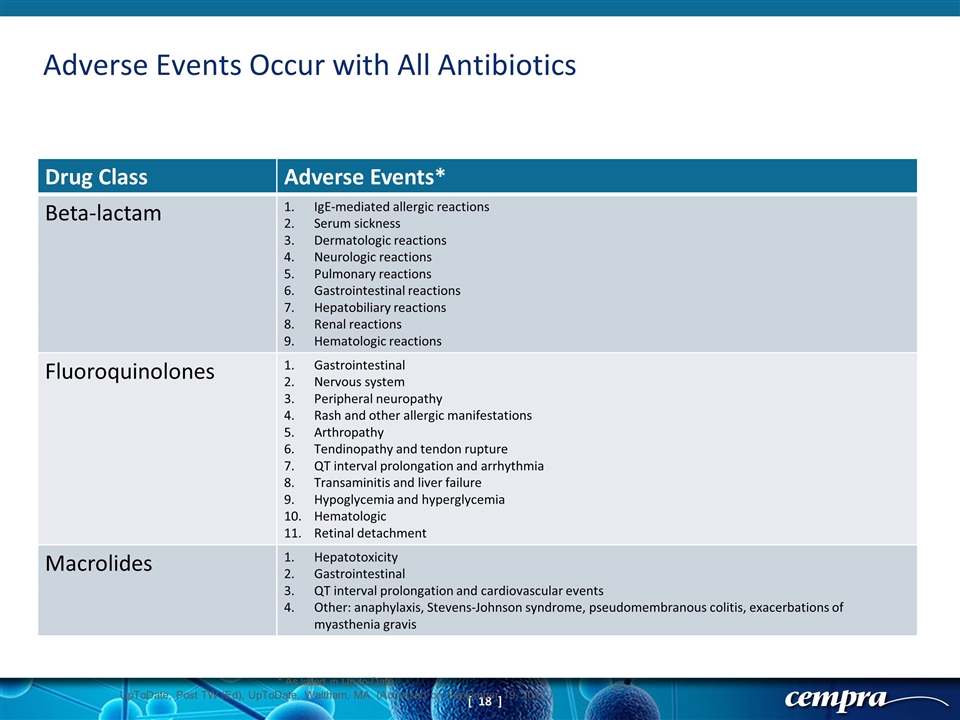
Adverse Events Occur with All Antibiotics Drug Class Adverse Events* Beta-lactam IgE-mediated allergic reactions Serum sickness Dermatologic reactions Neurologic reactions Pulmonary reactions Gastrointestinal reactions Hepatobiliary reactions Renal reactions Hematologic reactions Fluoroquinolones Gastrointestinal Nervous system Peripheral neuropathy Rash and other allergic manifestations Arthropathy Tendinopathy and tendon rupture QT interval prolongation and arrhythmia Transaminitis and liver failure Hypoglycemia and hyperglycemia Hematologic Retinal detachment Macrolides Hepatotoxicity Gastrointestinal QT interval prolongation and cardiovascular events Other: anaphylaxis, Stevens-Johnson syndrome, pseudomembranous colitis, exacerbations of myasthenia gravis * As listed in Up-to-Date UpToDate, Post TW (Ed), UpToDate, Waltham, MA. (Accessed on September 19, 2016.)
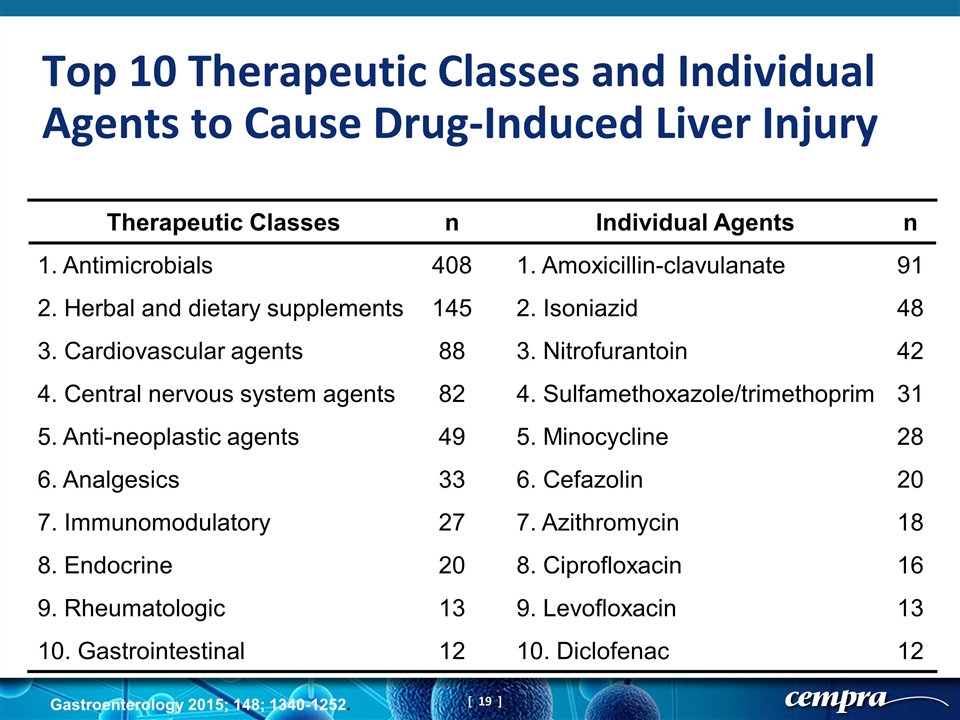
Therapeutic Classes n Individual Agents n 1. Antimicrobials 408 1. Amoxicillin-clavulanate 91 2. Herbal and dietary supplements 145 2. Isoniazid 48 3. Cardiovascular agents 88 3. Nitrofurantoin 42 4. Central nervous system agents 82 4. Sulfamethoxazole/trimethoprim 31 5. Anti-neoplastic agents 49 5. Minocycline 28 6. Analgesics 33 6. Cefazolin 20 7. Immunomodulatory 27 7. Azithromycin 18 8. Endocrine 20 8. Ciprofloxacin 16 9. Rheumatologic 13 9. Levofloxacin 13 10. Gastrointestinal 12 10. Diclofenac 12 Gastroenterology 2015; 148; 1340-1252. Top 10 Therapeutic Classes and Individual Agents to Cause Drug-Induced Liver Injury

Azithromycin (Z-Pak) Antibiotics Have Well Understood Class Effects Au

Solithromycin Addresses Significant Need Advisory Committee agreed (13-0)—Solithromycin effective against CABP Macrolide resistance at 50% and growing in U.S. Solithromycin has demonstrated activity against resistant strains Fluoroquinolone safety warnings recently strengthened by FDA Antibiotic stewardship a concern solithromycin can help to address Physicians need a new option for appropriate patients Cempra committed to ensuring safe and appropriate use for CABP for a 5-7 day course of therapy and post-marketing activities as agreed with FDA

Work every day to restore credibility with investors who expect: Unquestioned transparency Strong execution Engage with the FDA on path to approval Cempra committed to ensuring safe and appropriate use Drive progress at Uquifa to meet CMC requirements Get solithromycin in the toolbox for doctors to use with appropriate patients We have a vision for Cempra, a strategic focus on solithromycin and a defined plan to execute against Where We Are Headed

November 2016 Developing Well-Differentiated Antibiotics