Exhibit 99.1

|
For media: |
For investors: |
|
Megan Lustig |
Jonathan Poole |
|
Spectrum Science Communications |
Genocea Biosciences |
|
O: 202-955-6222 |
O: 617-876-8191 |
|
mlustig@spectrumscience.com |
jonathan.poole@genocea.com |
Genocea Announces Positive Top-Line Phase 2 Data
for Genital Herpes Immunotherapy GEN-003
- Highly statistically significant 55 percent reduction in viral shedding rate with best dose -
- Company to host conference call at 9 a.m. EDT today including guest speaker Peter A. Leone, MD -
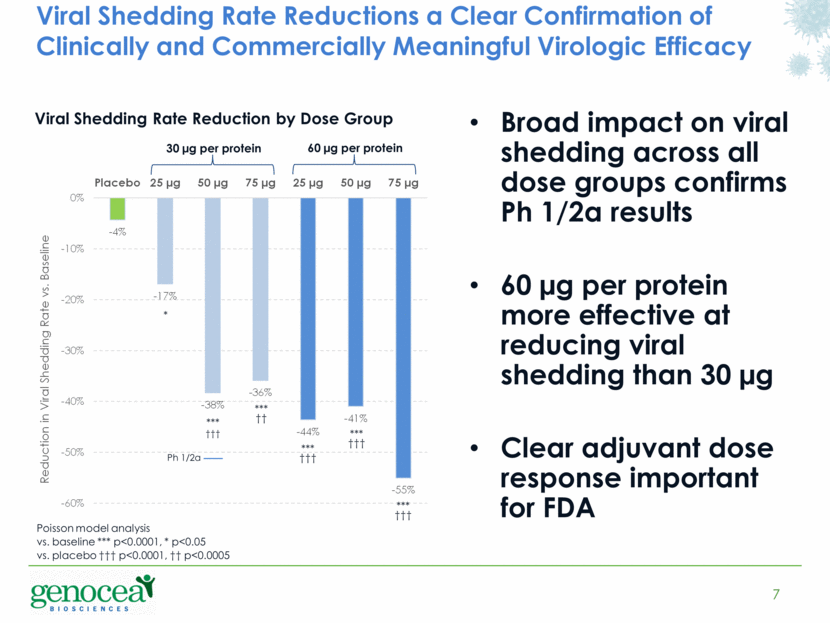
CAMBRIDGE, MA, May 20, 2015 —(BUSINESS WIRE)— Genocea Biosciences, Inc. (NASDAQ:GNCA), a biopharmaceutical company developing T cell-directed vaccines and immunotherapies, today announced positive top-line data from a Phase 2 dose optimization trial evaluating GEN-003 for the treatment of genital herpes. During the 28-day observation period immediately after completion of dosing, the best dose of 60 µg per protein / 75 µg of Matrix-M2TM adjuvant demonstrated a highly statistically significant (p<0.0001) 55 percent reduction from baseline in the viral shedding rate, the primary endpoint of the trial and a measure of anti-viral activity. All dose combinations tested, including the successful 30 µg per protein / 50 µg of adjuvant dose from the prior Phase 1/2a trial, demonstrated a statistically significant viral shedding rate reduction versus baseline and only the lowest dose combination did not demonstrate a statistically significant reduction versus placebo.
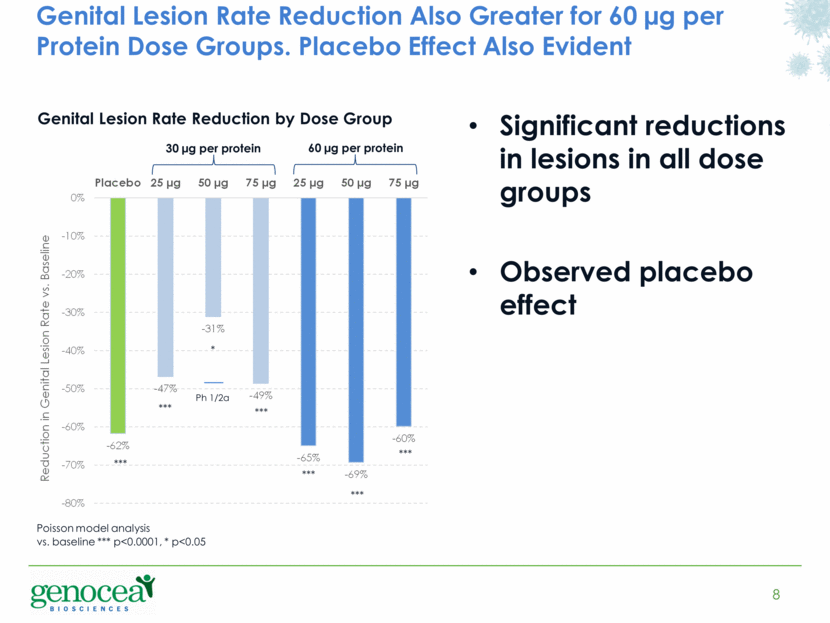
In a planned secondary analysis to assess impact on patient-reported genital lesion rates, a self-assessed measurement of clinical disease, all dose groups, including the placebo group, demonstrated a statistically significant reduction from baseline.
“We are extremely pleased with these positive top-line results which have successfully allowed us to identify the optimal dose to advance into further trials,” said Chip Clark, president and chief executive officer. “The results strengthen the product profile from our Phase 1/2a trial, which we have shown in market research to be highly clinically meaningful and commercially attractive, providing further evidence of the strong value proposition of GEN-003 for patients, physicians and payers.”
The Phase 2 study showed that GEN-003 was generally safe and well tolerated by patients, with no serious adverse events related to the vaccine. Safety data throughout the trial is monitored by an independent data safety monitoring board. Although reactogenicity increased with adjuvant dose, there was no difference in discontinuations in patient dosing due to adverse events across the different treatment arms.
“People living with genital herpes are seeking treatments that deliver on efficacy and provide them with peace of mind that they are at less risk of transmitting the infection to their sexual partners,” said Peter Leone, MD, Professor of Medicine and Adjunct Professor of Epidemiology at the University of North Carolina, and an investigator in the GEN-003 dose optimization trial. “We know that asymptomatic viral shedding is the driver of herpes transmission and GEN-003’s demonstrated antiviral efficacy data reported to date suggest this immunotherapy can be an effective future treatment.”
About the GEN-003 Phase 2 Clinical Trial
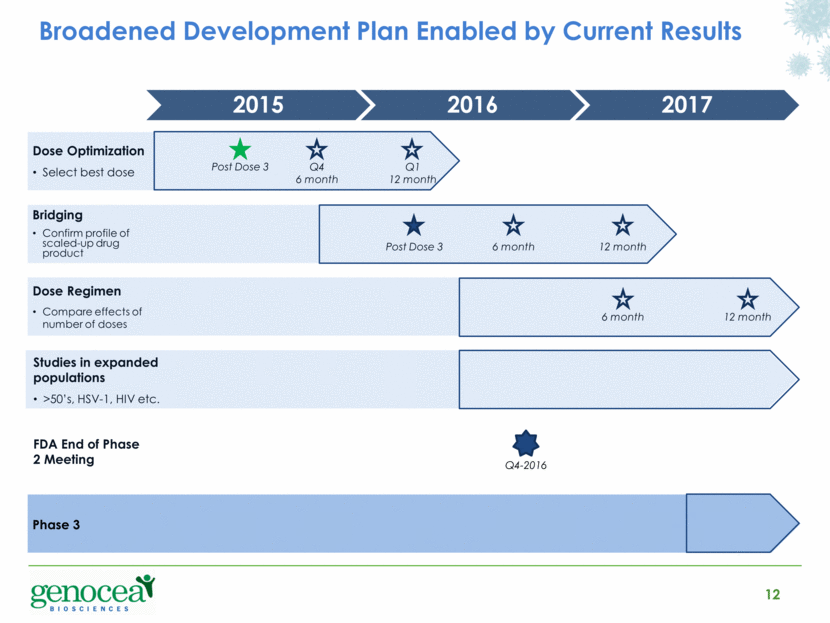
The Phase 2 study enrolled 310 subjects from 17 institutions in the United States. Subjects were randomized to one of six dosing groups of either 30 µg or 60 µg per protein paired with one of three adjuvant doses (25 µg, 50 µg, or 75 µg). A seventh group received placebo. Subjects received three doses of GEN-003 or placebo at 21-day intervals. Baseline viral shedding and genital lesion rates were established for each subject in a 28-day observation period prior to the commencement of dosing by collecting 56 genital swab samples (two per day), which were analyzed for the presence of HSV-2 DNA, and by recording the days on which genital lesions were present. This 28-day observation period was repeated immediately after the completion of dosing and will be repeated two more times over the course of this trial, at six and twelve months following dosing. No booster doses will be given.
A prior Phase 1/2a clinical trial demonstrated, at the corresponding immediate post dosing 28-day observation period, a highly statistically significant 52 percent reduction in the viral shedding rate and a highly statistically significant 48 percent reduction in the genital lesion rate compared to baseline at a dose of 30 µg per protein/50 µg of Matrix-M2TM adjuvant.
For more information about this clinical study of GEN-003 please visit www.clinicaltrials.gov.
Conference Call
Genocea management will host a conference call and webcast today, at 9 a.m. EDT. Peter A. Leone, MD, Professor of Medicine and Adjunct Professor of Epidemiology at the University of North Carolina, will also join management for the call. Dr. Leone is an expert in the surveillance, prevention, and control of sexually transmitted diseases and is an investigator in the GEN-003 dose optimization trial. The conference call may be accessed by dialing (844) 826-0619 for domestic participants and (315) 625-6883 for international callers (reference conference ID 52068310). A live webcast of the conference call will be available online from the investor relations section of the Company’s website at http://ir.genocea.com. A webcast replay of the conference call will be available on the Genocea website beginning approximately two hours after the event, and will be archived for 30 days.
About GEN-003
Inducing a T cell response against HSV-2 is critical to treating the clinical symptoms of disease and controlling transmission of the infection. GEN-003 is a first-in-class T-cell directed immunotherapy designed to elicit both a T cell and B cell (antibody) immune response. The immunotherapy was designed using Genocea’s ATLAS™ platform, which profiles the comprehensive spectrum of actual T cell responses mounted by humans in response to disease, to identify antigen targets that drive T cell response. GEN-003 includes the antigens ICP4 and gD2 along with Matrix-M2TM adjuvant, which Genocea licensed from Novavax, Inc.
For more information about GEN-003, please visit http://www.genocea.com/platform-pipeline/pipeline/gen003-for-genital-herpes/
About Genital Herpes
Genital Herpes affects more than 400 million people worldwide and causes recurrent, painful genital lesions. It can be transmitted to sexual partners, even when the disease is asymptomatic. Current genital herpes therapies only partially control clinical symptoms and viral shedding, a process which drives disease transmission. Incomplete control of genital lesions and transmission risk, expense and the perceived inconvenience of taking a daily medication are hurdles for long-term disease management. Immunity through T cells is believed to be particularly critical to the control and possible prevention of genital herpes infections.
About Genocea
Genocea is harnessing the power of T cell immunity to develop life-changing vaccines and immunotherapies. T cells are increasingly recognized as a critical element of protective immune responses to a wide range of diseases, but traditional discovery methods have proven unable to identify the targets of such protective immune response. Using ATLAS™, its proprietary technology platform, Genocea identifies these targets to potentially enable the rapid development of medicines to address critical patient needs. Genocea’s pipeline of novel clinical stage T cell-enabled product candidates includes GEN-003 for genital herpes, GEN-004 for the prevention of infection by all serotypes of pneumococcus, and earlier-stage programs in chlamydia, genital herpes prophylaxis, malaria and cancer immunotherapy. For more information, please visit the company’s website at www.genocea.com.
Forward Looking Statements
Statements herein relating to future business performance, conditions or strategies and other financial and business matters, including expectations regarding clinical developments, are forward-looking statements within the meaning of the Private Securities Litigation Reform Act. Genocea cautions that these forward-looking statements are subject to numerous assumptions, risks and uncertainties, which change over time. Factors that may cause actual results to differ materially from the results discussed in the forward-looking statements or historical experience include risks and uncertainties, including Genocea’s ability to progress any product candidates in preclinical or clinical trials; the scope, rate and progress of its preclinical studies and clinical trials and other research and development activities; anticipated clinical trial results; that current results may not be predictive of future results; even if the data from preclinical studies or clinical trials is positive, regulatory authorities may require additional studies for approval and the product may not prove to be safe and efficacious; Genocea’s ability to enter into future collaborations with industry partners and the government and the terms, timing and success of any such collaboration; risks associated with the manufacture and supply of clinical and commercial product; the cost of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights; Genocea’s ability to obtain rights to technology; competition for clinical resources and patient enrollment from drug candidates in development by other companies with greater resources and visibility; the rate of cash utilized by Genocea in its business and the period for which existing cash will be able to fund such operation; Genocea’s ability to obtain adequate financing in the future through product licensing, co-promotional arrangements, public or private equity or debt financing or otherwise; general business conditions; competition; business abilities and judgment of personnel; the availability of qualified personnel and other factors set forth under “Risk Factors” in Genocea’s Annual Report on Form 10-K for the fiscal year ended December 31, 2014 and other filings with the Securities and Exchange Commission (the “SEC”). Further information on the factors and risks that could affect Genocea’s business, financial conditions and results of operations is contained in Genocea’s filings with the SEC, which are available at www.sec.gov. These forward-looking statements speak only as of the date of this press release and Genocea assumes no duty to update forward-looking statements.
## ## ##