SECURITIES AND EXCHANGE COMMISSION
PURSUANT TO RULE 13a-16 OR 15d-16 OF THE
SECURITIES EXCHANGE ACT OF 1934
Parc de Negocis Can Sant Joan
Barcelona, Spain
(Address of registrant’s principal executive office)
| Item | Sequential Page Number | |||
| 2 | ||||
| 9 | ||||

| ▪ | Grifols completes the acquisition of Talecris and initiates the integration process | |
| ▪ | 80% of the new Grifols’ sales are generated in international markets. It is expected sales in US and Canada will account for 60% of revenues in the medium term | |
| ▪ | First step towards the realization of operating synergies: Grifols obtains FDA approval for the utilization of Fraction II+III of the Los Angeles plant (intermediate product) in the production of Gamunex®, Talecris’ IVIG | |
| ▪ | Recurring2 EBITDA increases by 8.8% and reaches 162.7 million Euros, representing 25.6% on sales. | |
| ▪ | Net Recurring2 profit up 12.4% to reach 76.4 million Euros1 | |
| ▪ | Net Financial Debt at lower levels than expected for the completion of the acquisition. Net Debt over Recurring2 EBITDA stands at 4.4x compared to the expected level of 5.2x. | |
| ▪ | Grifols average workforce exceeds 11,100 employees |
| 1 | Includes Talecris’ results for June 2011, first month consolidated | |
| 2 | Excluding costs associated to the transaction of Talecris and non recurring costs | |
| 3 | Pro-forma unaudited figures obtained from the consolidated statements of both companies for the 6 months period to 30 June 2011. Provided for guidance purposes only. |
1

| 1 | Includes Talecris’ results for June 2011, first month consolidated | |
| 2 | Excluding costs associated to the transaction of Talecris and non recurring costs | |
| 3 | Pro-forma unaudited figures obtained from the consolidated statements of both companies for the 6 months period to 30 June 2011. Provided for guidance purposes only. |
2

| 1 | Includes Talecris’ results for June 2011, first month consolidated | |
| 2 | Excluding costs associated to the transaction of Talecris and non recurring costs | |
| 3 | Pro-forma unaudited figures obtained from the consolidated statements of both companies for the 6 months period to 30 June 2011. Provided for guidance purposes only. |
3

(Millions of US dollars)
| Senior secured debt | Amount | Term | Conditions | |||||||||
Tranche A |
$ | 1,500 | 5 years | 3.75% / 4.00 | % | |||||||
Tranche B |
$ | 1,600 | 6 years | 4.25% / 4.50 | % | |||||||
Revolving credit facility |
$ | 300 | — | 3.75% / 4.00 | % | |||||||
Total |
$ | 3,400 | ||||||||||
| Senior unsecured debt | Amount | Term | Conditions | |||||||||
Corporate bond issue |
$ | 1,100 | 7 years | 8.25% | ||||||||
| 1 | Includes Talecris’ results for June 2011, first month consolidated | |
| 2 | Excluding costs associated to the transaction of Talecris and non recurring costs | |
| 3 | Pro-forma unaudited figures obtained from the consolidated statements of both companies for the 6 months period to 30 June 2011. Provided for guidance purposes only. |
4

| Million Euros | H1 — 20111 | H1 — 2010 | % var | |||||||||
Total revenues |
635.3 | 487.8 | +30.2 | % | ||||||||
Bioscience Division |
521.5 | 380.1 | +37.2 | % | ||||||||
Diagnostic Division |
56.8 | 54.4 | +4.4 | % | ||||||||
Hospital Division |
49.3 | 45.1 | +9.2 | % | ||||||||
Raw Materials & Others Division |
7.7 | 8.1 | -5.9 | % | ||||||||
Recurring EBITDA2 |
162.7 | 149.6 | +8.8 | % | ||||||||
% on sales |
25.6 | % | 30.7 | % | ||||||||
Recurring Net profit2 |
76.4 | 67.9 | +12.4 | % | ||||||||
% on sales |
12.0 | % | 13.9 | % | ||||||||
EBITDA |
96.9 | 147.6 | -34.4 | % | ||||||||
% on sales |
15.2 | % | 30.2 | % | ||||||||
Net profit |
19.3 | 66.4 | -71.0 | % | ||||||||
% on sales |
3.0 | % | 13.6 | % | ||||||||
| 1 | Includes Talecris’ results for June 2011, first month consolidated | |
| 2 | Excluding costs associated to the transaction of Talecris and non recurring costs | |
| 3 | Pro-forma unaudited figures obtained from the consolidated statements of both companies for the 6 months period to 30 June 2011. Provided for guidance purposes only. |
5

(data in million Euros)
| Million Euros | H1 — 20113 | H1 — 2010 | % var | |||||||||
Total revenues |
1,139.0 | 1,063.5 | +7.1 | % | ||||||||
Recurring EBITDA2 |
305.6 | 314.1 | -2.7 | % | ||||||||
% on sales |
26.8 | % | 29.5 | % | ||||||||
Recurring Net Profit2 |
151.9 | 146.5 | 3.6 | % | ||||||||
% on sales |
13.3 | % | 13.8 | % | ||||||||
| 1 | Includes Talecris’ results for June 2011, first month consolidated | |
| 2 | Excluding costs associated to the transaction of Talecris and non recurring costs | |
| 3 | Pro-forma unaudited figures obtained from the consolidated statements of both companies for the 6 months period to 30 June 2011. Provided for guidance purposes only. |
6

| 1 | Includes Talecris’ results for June 2011, first month consolidated | |
| 2 | Excluding costs associated to the transaction of Talecris and non recurring costs | |
| 3 | Pro-forma unaudited figures obtained from the consolidated statements of both companies for the 6 months period to 30 June 2011. Provided for guidance purposes only. |
7
for the six month period ended 30 June 2011
filed with the CNMV. In case of discrepancies, the Spanish
version will prevail
| • | Condensed Consolidated Interim Financial Statements |
| ▪ | Balance Sheet | ||
| ▪ | Income Statement | ||
| ▪ | Consolidated Comprehensive Income Statement | ||
| ▪ | Statement of Cash Flows | ||
| ▪ | Statement of Changes in Net Equity |
| • | Notes to Condensed Consolidated Interim Financial Statements |
| (1) | General Information | ||
| (2) | Basis of Presentation and Accounting Principles Applied | ||
| (3) | Changes in the composition of the Group | ||
| (4) | Financial Risk Management Policy | ||
| (5) | Segment Reporting | ||
| (6) | Goodwill | ||
| (7) | Other Intangible Assets and Property, Plant and Equipment | ||
| (8) | Trade Receivables | ||
| (9) | Other current assets | ||
| (10) | Cash and Cash equivalents | ||
| (11) | Capital and Reserves | ||
| (12) | Financial Liabilities | ||
| (13) | Financial Income and Expenses | ||
| (14) | Income Tax | ||
| (15) | Average Workforce | ||
| (16) | Discontinued Operation | ||
| (17) | Commitments and Contingencies | ||
| (18) | Related Parties | ||
| (19) | Subsequent Events |
I
at 30 June 2011 and 31 December 2010
| Assets | 30/06/11 | 31/12/10 | ||||||
| (unaudited) | ||||||||
| (expressed in thousands of euros) | ||||||||
Non-current assets |
||||||||
Intangible assets |
||||||||
Goodwill (note 6) |
2,281,696 | 189,448 | ||||||
Other intangible assets (note 7) |
128,474 | 78,299 | ||||||
Total intangible assets |
2,410,170 | 267,747 | ||||||
Property, plant and equipment (note 7) |
639,735 | 434,131 | ||||||
Investments in equity accounted investees |
3,546 | 598 | ||||||
Non-current financial assets (note 12) |
41,667 | 7,535 | ||||||
Deferred tax assets |
139,435 | 34,889 | ||||||
Total non-current assets |
3,234,553 | 744,900 | ||||||
Current assets |
||||||||
Inventories |
997,826 | 527,865 | ||||||
Trade and other receivables |
||||||||
Trade receivables (note 8) |
405,450 | 224,355 | ||||||
Other receivables |
48,971 | 44,032 | ||||||
Current income tax assets |
41,029 | 14,607 | ||||||
Trade and other receivables |
495,450 | 282,994 | ||||||
Other current financial assets |
19,254 | 12,946 | ||||||
Other current assets (note 9) |
13,344 | 80,628 | ||||||
Cash and cash equivalents (note 10) |
583,792 | 239,649 | ||||||
Total current assets |
2,109,666 | 1,144,082 | ||||||
Total assets |
5,344,219 | 1,888,982 | ||||||
at 30 June 2011 and 31 December 2010
| Equity and liabilities | 30/06/11 | 31/12/10 | ||||||
| (unaudited) | ||||||||
| (expressed in thousands of euros) | ||||||||
Equity |
||||||||
Share capital (note 11) |
114,914 | 106,532 | ||||||
Share premium (note 11) |
890,355 | 121,802 | ||||||
Reserves (note 11) |
||||||||
Accumulated gains |
519,788 | 350,543 | ||||||
Other reserves |
49,894 | 53,061 | ||||||
Total reserves |
569,682 | 403,604 | ||||||
Own shares (note 11) |
(1,927 | ) | (1,927 | ) | ||||
Profit for the period / year attributable to the Parent |
19,269 | 115,513 | ||||||
Total |
1,592,293 | 745,524 | ||||||
Available-for-sale financial assets |
(575 | ) | — | |||||
Cash flow hedges |
(2,331 | ) | (1,751 | ) | ||||
Translation differences |
(88,734 | ) | (50,733 | ) | ||||
Other comprehensive income |
(91,640 | ) | (52,484 | ) | ||||
Equity attributable to the Parent |
1,500,653 | 693,040 | ||||||
Non-controlling interests |
12,941 | 14,350 | ||||||
Total equity |
1,513,594 | 707,390 | ||||||
Liabilities |
||||||||
Non-current liabilities |
||||||||
Grants |
1,815 | 2,088 | ||||||
Provisions |
10,461 | 1,378 | ||||||
Non-current financial liabilities |
||||||||
Loans and borrowings, bonds and other
marketable securities |
2,642,944 | 665,385 | ||||||
Other financial liabilities |
72,400 | 10,474 | ||||||
Total non-current financial liabilities (note 12) |
2,715,344 | 675,859 | ||||||
Deferred tax liabilities |
140,075 | 79,141 | ||||||
Total non-current liabilities |
2,867,695 | 758,466 | ||||||
Current liabilities |
||||||||
Provisions |
35,828 | 4,365 | ||||||
Current financial liabilities |
||||||||
Loans and borrowings, bonds and other marketable securities |
507,374 | 191,635 | ||||||
Other financial liabilities |
17,336 | 18,236 | ||||||
Total current financial liabilities (note 12) |
524,710 | 209,871 | ||||||
Debts with associates |
2,352 | 1,162 | ||||||
Trade and other payables |
||||||||
Suppliers |
266,393 | 160,678 | ||||||
Other payables |
25,618 | 11,928 | ||||||
Current income tax liabilities |
27,227 | 4,172 | ||||||
Total trade and other payables |
319,238 | 176,778 | ||||||
Other current liabilities |
80,802 | 30,950 | ||||||
Total current liabilities |
962,930 | 423,126 | ||||||
Total liabilities |
3,830,625 | 1,181,592 | ||||||
Total equity and liabilities |
5,344,219 | 1,888,982 | ||||||
for the Six Month Period Ended 30 June 2011 and 2010
| 30/06/11 | 30/06/10 | |||||||
| (unaudited) | ||||||||
| (expressed in thousands of euros) | ||||||||
Continuing Operations |
||||||||
Revenues (note 5) |
635,341 | 487,809 | ||||||
Changes in inventories of finished goods and work in progress |
2,757 | 41,209 | ||||||
Self-constructed non-current assets |
32,346 | 16,051 | ||||||
Supplies |
(175,142 | ) | (157,107 | ) | ||||
Other operating income |
1,009 | 631 | ||||||
Personnel expenses |
(183,727 | ) | (141,972 | ) | ||||
Other operating expenses |
(155,532 | ) | (98,279 | ) | ||||
Amortisation and depreciation (note 7) |
(28,156 | ) | (21,434 | ) | ||||
Transaction costs of Talecris business combination (note 3 & 9) |
(38,607 | ) | (2,019 | ) | ||||
Non-financial and other capital grants |
742 | 550 | ||||||
Impairment and gains/(losses) on disposal of fixed assets (notes 6 & 7) |
(22,302 | ) | 681 | |||||
Results from operating activities |
68,729 | 126,120 | ||||||
Finance income |
1,761 | 2,179 | ||||||
Finance expenses (notes 8 & 13) |
(55,546 | ) | (25,285 | ) | ||||
Change in fair value of financial instruments (note 13) |
13,945 | (15,404 | ) | |||||
Exchange gains / (losses) |
(2,122 | ) | 1,970 | |||||
Finance expense |
(41,962 | ) | (36,540 | ) | ||||
Share of loss of equity accounted investees |
(807 | ) | (728 | ) | ||||
Profit before income tax |
25,960 | 88,852 | ||||||
Income tax expense (note 14) |
(7,347 | ) | (23,022 | ) | ||||
Consolidated profit for the period |
18,613 | 65,830 | ||||||
Profit attributable to equity holders of the Parent |
19,269 | 66,408 | ||||||
Loss attributable to non-controlling interests |
(656 | ) | (578 | ) | ||||
Basic earnings per share (Euros) |
0.09 | 0.31 | ||||||
Diluted earnings per share (Euros) |
0.09 | 0.31 | ||||||
for the Six Month Period Ended 30 June 2011 and 2010
| 30/06/11 | 30/06/10 | |||||||
| (unaudited) | ||||||||
| (expressed in thousands of euros) | ||||||||
Consolidated profit for the period |
18,613 | 65,830 | ||||||
Other comprehensive income |
||||||||
Income and expenses generated during the period |
||||||||
Measurement of financial instruments |
(575 | ) | 0 | |||||
Available-for-sale financial assets |
(822 | ) | 0 | |||||
Tax effect |
247 | 0 | ||||||
Cash flow hedges |
(2,331 | ) | 0 | |||||
Cash flow hedges |
(3,829 | ) | 0 | |||||
Tax effect |
1,498 | 0 | ||||||
Translation differences |
(38,541 | ) | 74,874 | |||||
Income and expenses generated during the period |
(41,447 | ) | 74,874 | |||||
Income and expense recognised in the income statement: |
||||||||
Cash flow hedges |
1,751 | 99 | ||||||
Cash flow hedges |
2,870 | 159 | ||||||
Tax effect |
(1,119 | ) | (60 | ) | ||||
Income and expense recognised in the income statement: |
1,751 | 99 | ||||||
Other comprehensive income and expenses for the period |
(39,697 | ) | 74,973 | |||||
Total comprehensive income and expenses for the period |
(21,083 | ) | 140,803 | |||||
Total comprehensive income / (losses) attributable to the Parent |
(19,887 | ) | 139,935 | |||||
Total comprehensive income / (losses) attributable to non-controlling interests |
(1,196 | ) | 868 | |||||
Total comprehensive income for the period |
(21,083 | ) | 140,803 | |||||
for the Six Month Period Ended 30 June 2011 and 2010
| 30/06/11 | 30/06/10 | |||||||
| (unaudited) | ||||||||
| (expressed in thousands of euros) | ||||||||
Cash flows from operating activities |
||||||||
Profit before tax |
25,960 | 88,852 | ||||||
Adjustments for: |
92,638 | 53,782 | ||||||
Amortisation and depreciation |
28,156 | 21,434 | ||||||
Other adjustments: |
64,482 | 32,348 | ||||||
Losses on equity accounted investments |
807 | 728 | ||||||
Exchange differences |
2,122 | (1,970 | ) | |||||
Net provision charges |
14,454 | 129 | ||||||
(Profit) / loss on disposal of fixed assets |
9,416 | (681 | ) | |||||
Government grants taken to income |
(742 | ) | (550 | ) | ||||
Finance expense / income |
37,130 | 33,386 | ||||||
Other adjustments |
1,295 | 1,306 | ||||||
Changes in capital and assets |
(65,159 | ) | 13,700 | |||||
Change in inventories |
752 | (11,982 | ) | |||||
Change in trade and other receivables |
(66,961 | ) | 20,239 | |||||
Change in current financial assets and other current assets |
(451 | ) | (3,875 | ) | ||||
Change in current trade and other payables |
1,501 | 9,318 | ||||||
Other cash flows from operating activities |
(36,745 | ) | (34,465 | ) | ||||
Interest paid |
(34,021 | ) | (19,801 | ) | ||||
Interest recovered |
999 | 3,861 | ||||||
Income tax recovered |
(3,723 | ) | (18,525 | ) | ||||
Net cash from operating activities |
16,694 | 121,869 | ||||||
Cash flows from investing activities |
||||||||
Payments for investments |
(1,669,390 | ) | (56,997 | ) | ||||
Group companies and business units (note 3) |
(1,615,417 | ) | (3,727 | ) | ||||
Property, plant and equipment and intangible assets |
(52,838 | ) | (49,151 | ) | ||||
Property, plant and equipment |
(42,841 | ) | (43,146 | ) | ||||
Intangible assets |
(9,997 | ) | (6,005 | ) | ||||
Other financial assets |
(1,135 | ) | (4,119 | ) | ||||
Proceeds from the sale of property, plant and equipment |
69,151 | 2,863 | ||||||
Property, plant and equipment |
69,151 | 2,863 | ||||||
Net cash used in investing activities |
(1,600,239 | ) | (54,134 | ) | ||||
Cash flows from financing activities |
||||||||
Proceeds from and payments for equity instruments |
(2,264 | ) | (1,250 | ) | ||||
Issue |
(2,264 | ) | (1,250 | ) | ||||
Proceeds from and payments for financial liability instruments |
2,235,339 | (8,671 | ) | |||||
Issue |
2,982,877 | 51,067 | ||||||
Redemption and repayment |
(747,538 | ) | (59,738 | ) | ||||
Dividends and interest on other equity instruments paid |
0 | (53 | ) | |||||
Other cash flows from financing activities |
(287,203 | ) | 323 | |||||
Transaction costs of financial instruments issued in the acquisition of Talecris |
(287,550 | ) | 0 | |||||
Other amounts received from financing activities |
347 | 323 | ||||||
Net cash from / (used in) financing activities |
1,945,872 | (9,651 | ) | |||||
Effect of exchange rate fluctuations on cash |
(18,184 | ) | 42,684 | |||||
Net increase in cash and cash equivalents |
344,143 | 100,768 | ||||||
Cash and cash equivalents at beginning of the period |
239,649 | 249,372 | ||||||
Cash and cash equivalents at end of period |
583,792 | 350,140 | ||||||
Condensed Statement of Changes in Consolidated Equity
for the Six Month Period Ended 30 June 2011
| Attributable to equity holders of the Parent | ||||||||||||||||||||||||||||||||||||||||||||||||
| Other comprehensive income | ||||||||||||||||||||||||||||||||||||||||||||||||
| Available-for | Equity | |||||||||||||||||||||||||||||||||||||||||||||||
| Profit attributable | sale | attributable | ||||||||||||||||||||||||||||||||||||||||||||||
| Share | Share | to | Interim | Own | Translation | Cash flow | financial | to | Non-controlling | |||||||||||||||||||||||||||||||||||||||
| capital | premium | Reserves (*) | Parent | dividend | Shares | differences | hedges | assets | Parent | interests | Equity | |||||||||||||||||||||||||||||||||||||
| (unaudited) | ||||||||||||||||||||||||||||||||||||||||||||||||
| (expressed in thousands of euros) | ||||||||||||||||||||||||||||||||||||||||||||||||
Balances at 31 December 2009 |
106,532 | 121,802 | 314,903 | 147,972 | (31,960 | ) | (677 | ) | (90,253 | ) | (1,948 | ) | 0 | 566,371 | 12,157 | 578,528 | ||||||||||||||||||||||||||||||||
Translation differences |
— | — | — | — | — | — | 73,428 | — | — | 73,428 | 1,446 | 74,874 | ||||||||||||||||||||||||||||||||||||
Cash flow hedges |
— | — | — | — | — | — | — | 99 | — | 99 | — | 99 | ||||||||||||||||||||||||||||||||||||
Other comprehensive income for the period |
0 | 0 | 0 | 0 | 0 | 0 | 73,428 | 99 | 0 | 73,527 | 1,446 | 74,973 | ||||||||||||||||||||||||||||||||||||
Profit/(loss) for the period |
— | — | — | 66,408 | — | — | — | — | — | 66,408 | (578 | ) | 65,830 | |||||||||||||||||||||||||||||||||||
Total comprehensive
income for the period |
0 | 0 | 0 | 66,408 | 0 | 0 | 73,428 | 99 | 0 | 139,935 | 868 | 140,803 | ||||||||||||||||||||||||||||||||||||
Operations with own shares |
— | — | — | — | — | (1,250 | ) | — | — | — | (1,250 | ) | — | (1,250 | ) | |||||||||||||||||||||||||||||||||
Other changes |
— | — | 1 | — | — | — | — | — | — | 1 | — | 1 | ||||||||||||||||||||||||||||||||||||
Distribution of 2009 profit |
||||||||||||||||||||||||||||||||||||||||||||||||
Reserves |
— | — | 88,783 | (88,783 | ) | — | — | — | — | — | 0 | — | 0 | |||||||||||||||||||||||||||||||||||
Dividends |
— | — | — | (27,229 | ) | — | — | — | — | — | (27,229 | ) | (53 | ) | (27,282 | ) | ||||||||||||||||||||||||||||||||
Interim dividend |
— | — | — | (31,960 | ) | 31,960 | — | — | — | — | 0 | — | 0 | |||||||||||||||||||||||||||||||||||
Operations with equity holders or owners |
0 | 0 | 88,784 | (147,972 | ) | 31,960 | (1,250 | ) | 0 | 0 | 0 | (28,478 | ) | (53 | ) | (28,531 | ) | |||||||||||||||||||||||||||||||
Balances at 30 June 2010 |
106,532 | 121,802 | 403,687 | 66,408 | 0 | (1,927 | ) | (16,825 | ) | (1,849 | ) | 0 | 677,828 | 12,972 | 690,800 | |||||||||||||||||||||||||||||||||
Balances at 31 December 2010 |
106,532 | 121,802 | 403,604 | 115,513 | 0 | (1,927 | ) | (50,733 | ) | (1,751 | ) | 0 | 693,040 | 14,350 | 707,390 | |||||||||||||||||||||||||||||||||
Translation differences |
— | — | — | — | — | — | (38,001 | ) | — | (38,001 | ) | (540 | ) | (38,541 | ) | |||||||||||||||||||||||||||||||||
Cash flow hedges |
— | — | — | — | — | — | — | (580 | ) | — | (580 | ) | — | (580 | ) | |||||||||||||||||||||||||||||||||
Available-for-sale financial assets Gains/(losses) |
— | — | — | — | — | — | — | — | (575 | ) | (575 | ) | — | (575 | ) | |||||||||||||||||||||||||||||||||
Other comprehensive income for the period |
0 | 0 | 0 | 0 | 0 | 0 | (38,001 | ) | (580 | ) | (575 | ) | (39,156 | ) | (540 | ) | (39,696 | ) | ||||||||||||||||||||||||||||||
Profit/(loss) for the period |
— | — | — | 19,269 | — | — | — | — | — | 19,269 | (656 | ) | 18,613 | |||||||||||||||||||||||||||||||||||
Total comprehensive income for the period |
0 | 0 | 0 | 19,269 | 0 | 0 | (38,001 | ) | (580 | ) | (575 | ) | (19,887 | ) | (1,196 | ) | (21,083 | ) | ||||||||||||||||||||||||||||||
Other changes |
— | — | (35 | ) | — | — | — | — | — | — | (35 | ) | (213 | ) | (248 | ) | ||||||||||||||||||||||||||||||||
Capital Increase (note 11) |
8,382 | 768,553 | (2,264 | ) | — | — | — | — | — | — | 774,671 | — | 774,671 | |||||||||||||||||||||||||||||||||||
Other movements (note 11) |
— | — | 52,864 | — | — | — | — | — | — | 52,864 | — | 52,864 | ||||||||||||||||||||||||||||||||||||
Distribution of 2010 profit |
||||||||||||||||||||||||||||||||||||||||||||||||
Reserves |
— | — | 115,513 | (115,513 | ) | — | — | — | — | — | 0 | — | 0 | |||||||||||||||||||||||||||||||||||
Operations with equity holders or owners |
8,382 | 768,553 | 166,078 | (115,513 | ) | 0 | 0 | 0 | 0 | 0 | 827,500 | (213 | ) | 827,287 | ||||||||||||||||||||||||||||||||||
Balance at 30 June 2011 |
114,914 | 890,355 | 569,682 | 19,269 | 0 | (1,927 | ) | (88,734 | ) | (2,331 | ) | (575 | ) | 1,500,653 | 12,941 | 1,513,594 | ||||||||||||||||||||||||||||||||
| (*) Reserves include accumulated earnings and other reserves | ||
for the six month period ended 30 June 2011
1
| - | IAS 24 Revised Related Party Disclosures (effective date: 1 January 2011). | ||
| - | Amendment to IFRIC 14: Prepayment of a minimum funding requirement (effective date: 1 January 2011). | ||
| - | IFRS 7 Amendments resulting from May 2010 Annual Improvements (effective date: 1 January 2011). | ||
| - | Amendment to IFRIC 13 Customer Loyalty Programmes (effective date: 1 January 2011). | ||
| - | IAS 34 Amendments resulting from May 2010 Annual Improvements (effective date: 1 January 2011). | ||
| - | IAS 1 Amendments resulting from May 2010 Annual Improvements (effective date: 1 January 2011). |
2
| - | Amendment to IAS 12 Deferred tax: recovery of underlying assets (effective date: 1 January 2012) | ||
| - | Amendment to IFRS 1 Severe Hyperinflation and Removal of Fixed Dates for First-time Adopters (effective date: 1 July 2011) | ||
| - | Amendment to IFRS 7 Financial Instrument: Disclosures — Transfer of Financial Assets (effective date: 1 July 2011) | ||
| - | IFRS 9 Financial instruments (effective date: 1 January 2013) | ||
| - | IFRS 10 Consolidated Financial Statements (effective date: 1 January 2013) | ||
| - | IFRS 11 Joint Arrangements (effective date: 1 January 2013) | ||
| - | IFRS 12 Disclosures of Interests in Other Entities (effective date: 1 January 2013) | ||
| - | IFRS 13 Fair Value Measurement (effective date: 1 January 2013) | ||
| - | IAS 27 Separate Financial Statements (effective date: 1 January 2013) | ||
| - | IAS 28 Investments in Associates and Joint Ventures (effective date: 1 January 2013) | ||
| - | IAS 19 Employee Benefits (effective date: 1 January 2013) |
| - | The corporate tax expense which, according to IAS 34, is recognised in interim periods based on the best estimate of the average tax rate that the Group expects for the annual period. |
3
| - | The useful lives of property, plant, and equipment and intangible assets. | ||
| - | Measurement of assets and goodwill to determine any related impairment losses. | ||
| - | Evaluation of the capitalisation of development costs. | ||
| - | Evaluation of provisions and contingencies. | ||
| - | The assumptions used for calculation of the fair value of financial instruments. | ||
| - | Evaluation of the effectiveness of hedging. | ||
| - | Evaluation of the nature of leases (operating or financial). |
4
| - | Kedrion and Grifols entered into a contract manufacturing agreement to fractionate and purify Kedrion’s plasma to deliver IVIG and Albumin under Kedrion’s private label, and Factor VIII under the trade name Koate, all of them for sale only in the United States. | ||
| - | Grifols is committed to sell to Kedrion the Melville fractionation facility. Grifols lease from Kedrion the Melville fractionation facility being the lease term 3 years with an optional extension of up to 1 year at Grifols request. | ||
| - | Grifols transfer to Kedrion all Koate (factor VIII) technology and commercial agreements for the US market. Grifols will produce Koate for Kedrion up to a period of 7 years. | ||
| - | Grifols is committed to sell to Kedrion two plasma collection centers. In addition Grifols committed to sell 200.000 liters of source plasma to Kedrion at a fixed price. | ||
| - | Grifols authorizes Kedrion to market and sell in the US, IVIG and albumin manufactured by Grifols for Kedrion. |
5
| Thousands of | Thousands of | |||||||
| Euros | USD | |||||||
Cost of business combination (valuation of Class B Shares) |
829,799 | 1,195,574 | ||||||
Cash paid (19 USD per share) |
1,763,601 | 2,540,997 | ||||||
Total cost of business combination |
2,593,400 | 3,736,571 | ||||||
Book value of net assets acquired (provisional) |
469,318 | 676,193 | ||||||
Goodwill (excess of the cost of the business combination
over the fair value of identifiable net assets acquired) |
2,124,082 | 3,060,378 | ||||||
| (see note 6) | ||||||||
Cash paid |
1,763,601 | 2,540,996 | ||||||
Cash and cash equivalents of the acquired company |
(149,693 | ) | (215,678 | ) | ||||
Cash outflow for the acquisition |
1,613,908 | 2,325,319 | ||||||
6
| Book value | ||||||||
| Thousands of | ||||||||
| Thousands of Euros | USD | |||||||
Intangible assets (note 7) |
50,621 | 72,936 | ||||||
Property, plant and equipment (note 7) |
306,401 | 441,462 | ||||||
Non — current financial assets |
3,720 | 5,359 | ||||||
Deferred tax assets |
80,115 | 115,429 | ||||||
Inventories |
490,976 | 707,398 | ||||||
Trade and other receivables |
126,772 | 182,653 | ||||||
Other assets |
3,683 | 5,307 | ||||||
Cash and cash equivalents Total assets |
149,693 | 215,678 | ||||||
Total assets |
1,211,981 | 1,746,222 | ||||||
Non — current provisions |
9,250 | 13,327 | ||||||
Non — current financial liabilities |
6,289 | 9,061 | ||||||
Current financial liabilities |
473,085 | 681,621 | ||||||
Current provisions |
31,180 | 44,924 | ||||||
Trade and other payables |
158,113 | 227,809 | ||||||
Other current liabilities |
44,055 | 63,475 | ||||||
Deferred tax liabilities |
20,691 | 29,812 | ||||||
Total liabilities and contingent liabilities |
742,663 | 1,070,029 | ||||||
Total net assets acquired |
469,318 | 676,193 | ||||||
7
| Net revenues (Thousands of Euros) | ||||||||
| Six months ended 30 | Six months ended 30 | |||||||
| SEGMENTS | June 2011 | June 2010 | ||||||
Bioscience |
521,538 | 380,081 | ||||||
Hospital |
49,289 | 45,146 | ||||||
Diagnostic |
56,831 | 54,413 | ||||||
Raw materials + Other |
7,683 | 8,169 | ||||||
TOTAL |
635,341 | 487,809 | ||||||
| Consolidated | ||||||||
| Income/(loss) (Thousands of Euros) | ||||||||
| Six months ended 30 | Six months ended 30 | |||||||
| SEGMENTS | June 2011 | June 2010 | ||||||
Bioscience |
186,521 | 162,938 | ||||||
Hospital |
4,786 | 5,196 | ||||||
Diagnostic |
(11,264 | ) | 4,798 | |||||
Raw materials + Other |
3,694 | 4,763 | ||||||
Total income of reported segments |
183,737 | 177,695 | ||||||
Unallocated expenses plus net financial result |
(157,777 | ) | (88,843 | ) | ||||
Profit before income tax from
continuing operations |
25,960 | 88,852 | ||||||
8
| Thousand Euros | ||||||||||||||||||||
| Balance at | Business | Translation | Balance at | |||||||||||||||||
| 31/12/10 | Combination | Impairment | differences | 30/06/11 | ||||||||||||||||
Net value |
||||||||||||||||||||
Grifols UK,Ltd. (UK) |
7,982 | 0 | 0 | (370 | ) | 7,612 | ||||||||||||||
Grifols Italia,S.p.A. (Italy) |
6,118 | 0 | 0 | 0 | 6,118 | |||||||||||||||
Biomat USA, Inc. (USA) |
113,052 | 0 | 0 | (8,534 | ) | 104,518 | ||||||||||||||
Plasmacare, Inc. (USA) |
38,464 | 0 | 0 | (2,903 | ) | 35,561 | ||||||||||||||
Woolloomooloo Holdings Pty
Ltd. (Australia) |
23,832 | 0 | (13,000 | ) | (415 | ) | 10,417 | |||||||||||||
Talecris Biotherapeutics (USA) |
0 | 2,124,082 | 0 | (6,612 | ) | 2,117,470 | ||||||||||||||
| 189,448 | 2,124,082 | (13,000 | ) | (18,834 | ) | 2,281,696 | ||||||||||||||
| (note 3) | ||||||||||||||||||||
| - | UK: bioscience segment | ||
| - | Italy: bioscience segment | ||
| - | USA: bioscience segment | ||
| - | Australia: mainly to diagnostic segment. |
9
| 31/12/2010 | ||||||||
| Growth rate | Pre- tax discount rate | |||||||
Bioscience |
2.0% — 3.0 | % | 10.5% — 10.9 | % | ||||
Diagnostic |
2.0% | 10.4% | ||||||
| 30/06/2011 | ||||||||
| Growth rate | Pre - tax discount rate | |||||||
Bioscience |
N/A | N/A | ||||||
Diagnostic |
2.0% | 11.5% | ||||||
10
| Other intangible | Property, plant | |||||||||||
| Assets | and equipment | Total | ||||||||||
Total Cost at 31/12/2010 |
151,861 | 656,295 | 808,156 | |||||||||
Total dep. & amort. At 31/12/2010 |
(73,562 | ) | (221,515 | ) | (295,077 | ) | ||||||
Impairment at 31/12/2010 |
0 | (649 | ) | (649 | ) | |||||||
Balance at 31/12/2010 |
78,299 | 434,131 | 512,430 | |||||||||
Cost |
||||||||||||
Additions |
9,997 | 43,101 | 53,098 | |||||||||
Business Combination |
50,621 | 306,401 | 357,022 | |||||||||
Disposals |
(588 | ) | (123,965 | ) | (124,553 | ) | ||||||
Transfers |
(126 | ) | (885 | ) | (1,011 | ) | ||||||
Translation differences |
(2,515 | ) | (19,327 | ) | (21,842 | ) | ||||||
Total Cost at 30/06/2011 |
209,250 | 861,620 | 1,070,870 | |||||||||
Depreciation & amortization |
||||||||||||
Additions |
(8,630 | ) | (19,526 | ) | (28,156 | ) | ||||||
Disposals |
0 | 13,727 | 13,727 | |||||||||
Transfers |
600 | 411 | 1,011 | |||||||||
Translation differences |
816 | 5,553 | 6,369 | |||||||||
Total dep. & amort. At 30/06/2011 |
(80,776 | ) | (221,350 | ) | (302,126 | ) | ||||||
Impairment |
||||||||||||
Additions |
0 | 114 | 114 | |||||||||
Impairment at 30/06/2011 |
0 | (535 | ) | (535 | ) | |||||||
Balance at 30/06/2011 |
128,474 | 639,735 | 768,209 | |||||||||
11
Pre-tax discount rate: 10.9%
| (a) | Sale of Spanish properties and lease back |
| - | Compulsory initial term of five years, | ||
| - | Initial rent established at market prices and will be reviewed annually, based on the percentage variation in the Spanish Consumer Price Index (CPI), | ||
| - | Automatic extensions of five-year periods that can be avoided by both parties by a six month anticipated notice. | ||
| - | Upon vacating the premises, the lessor will reimburse Grifols for the remaining value of leasehold improvements Grifols made. |
12
| - | Compulsory initial term of 20 years. | ||
| - | Initial rent has been established at market prices and will be reviewed annually with a 3% increase. On the first day of the sixth year, the remaining rents until year twenty will be paid in advance in a lump sum. | ||
| - | Renewal option to extend for a ten-year period at Grifols Group election. | ||
| - | Purchase options granted during the sixth year and in year twenty (20) at market value, to be estimated by independent appraisers. |
13
| Thousand Euros | ||||
| 30/06/11 | ||||
Maturity: |
||||
Up to 1 year |
20,990 | |||
Between 1 and 5 years |
87,742 | |||
More than 5 years |
17,293 | |||
Total future minimum payments |
126,025 | |||
| Thousand Euros | ||||||||
| 30/06/11 | ||||||||
| Current | Non-current | |||||||
Minimum payments |
4,659 | 17,295 | ||||||
Interest |
(1,932 | ) | (3,553 | ) | ||||
Present value |
2,727 | 13,742 | ||||||
| Thousand Euros | ||||||||||||
| 30/06/11 | ||||||||||||
| Minimum payments | Interest | Present value | ||||||||||
Maturity at: |
||||||||||||
Less than one year |
4,659 | 1,932 | 2,727 | |||||||||
Two years |
4,391 | 1,486 | 2,905 | |||||||||
Three years |
4,391 | 1,119 | 3,272 | |||||||||
Four years |
4,391 | 706 | 3,685 | |||||||||
Five years |
4,124 | 241 | 3,883 | |||||||||
Total |
21,956 | 5,484 | 16,472 | |||||||||
14
| • | The Group has sold properties in Spain amounting to Euros 80.4 million which together with its related mortgage loan of Euros 53.5 million resulted in a net cash inflow of Euros 26.9 million (see note 7). | ||
| • | Part of the consideration paid in the acquisition of Talecris Group has been realized by delivery of Class B shares (see note 3). The issue of Class B shares has had no cash impacts. |
15
| • | This amount includes a decrease in profit before tax due to the transaction costs incurred by the Group during the six month period ended 30 June 2011 amounting to Euros 38,607 thousand (2,019 thousand for the six months ended at 30 June 2010) that have been paid in this period. | ||
| • | Change in current trade and other payables includes Euros 19,516 thousand corresponding to business combination costs accrued by Talecris companies prior to acquisition date and paid during June 2011. |
| (a) | Share Capital and Share Premium |
| • | Class A Shares: 213,064,899 ordinary shares of 0.50 Euros nominal value each, fully subscribed and paid up, of the same class and series being the ordinary shares of the Company. | ||
| • | Class B Shares: 83,811,688 preference non-voting shares of 0.10 Euros nominal value each, of the same class and series, and with the preferential rights set forth in the Company’s by laws. |
16
for the six month period ended 30 June 2011
| - | Each Class B share entitles its holder to receive a minimum annual preferred dividend out of the distributable profits at the end of each fiscal year equal to a 0.01 Euros per Class B share if the aggregate preferred dividend does not exceed the distributable profits of that fiscal year. This preferred dividend is not cumulative if no sufficient distributable profits are obtained in the year. | ||
| - | Each Class B share is entitled to receive, in addition to the preferred dividend referred to above, the same dividends and other distributions as one Grifols ordinary share. | ||
| - | Each Class B share entitles its holder to have it redeemed under certain circumstances, if a tender offer for all or part of the shares in the Company is made and settled except if holders of Class B shares have been entitled to participate in such offer and have their shares acquired in such offer equally and on the same terms as holders of Class A shares. Terms and conditions of redemption incorporated in by laws limit the amounts to be redeemed to the existence of distributable reserves and limit the percentage of shares to be redeemed to a relation to the ordinary shares to which the offer is addressed. | ||
| - | Each Class B shares has the right to receive prior to the ordinary shares, upon the winding-up and liquidation of Grifols, an amount equal to the sum of (i) the nominal value of each Class B share, and (ii) the share premium paid-up for such Class B share when it was subscribed for. Each holder is entitled to receive, in addition to the Class B liquidation amount, the same liquidation amount that is paid to each Grifols ordinary share. |
17
| Num. of shares | Thousand Euros | |||||||
Balance at 1 January 2010 |
53,326 | 677 | ||||||
Acquisitions |
105,000 | 1,250 | ||||||
Balance at 30 June 2010
and 30 June 2011 |
158,326 | 1,927 | ||||||
18
| Thousand Euros | ||||||||
| 30/06/11 | 31/12/10 | |||||||
Non-current financial liabilities |
||||||||
Issue of Corporate bonds (a) |
0 | 446,918 | ||||||
Issue High Yied Bonds (a) |
761,088 | 0 | ||||||
Transaction costs on bonds |
(110,542 | ) | (5,715 | ) | ||||
Non-current promissory notes (a) |
650,546 | 441,203 | ||||||
Tranche A (US Dollars) |
830,277 | 0 | ||||||
Tranche B (US Dollars) |
892,721 | 0 | ||||||
Tranche A (Euros) |
213,125 | 0 | ||||||
Tranche B (Euros) |
217,800 | 0 | ||||||
Implicit Floor and swap floor |
(19,565 | ) | 0 | |||||
Transaction costs on loans and borrowings |
(185,314 | ) | (1,365 | ) | ||||
Club Deal |
0 | 100,000 | ||||||
Other loans |
18,391 | 120,813 | ||||||
Finance lease liabilities |
24,963 | 4,734 | ||||||
Loans and borrowings (b) |
1,992,398 | 224,182 | ||||||
Loans and borrowings and bonds or other non
current marketable securities |
2,642,944 | 665,385 | ||||||
Financial derivatives |
61,685 | 0 | ||||||
Other non-current financial liabilities |
10,715 | 10,474 | ||||||
| 2,715,344 | 675,859 | |||||||
19
| Redemption | Exchange | Final | ||||||||||||||||||
| Initial balance | and | differences an | balance at | |||||||||||||||||
| at 01/01/11 | Issue | Repayments | others | 30/06/11 | ||||||||||||||||
Debt securities issued in a member state of the European Union that have not
required the registration of a prospectus (nominal value) |
8,373 | 9,990 | (8,373 | ) | 0 | 9,990 | ||||||||||||||
Other debt securities issued ouside a member state of the European Union
(nominal value) |
446,918 | 761,088 | (415,270 | ) | (31,648 | ) | 761,088 | |||||||||||||
TOTAL |
455,291 | 771,078 | (423,643 | ) | (31,648 | ) | 771,078 | |||||||||||||
20
| Redemption | Exchange | Final | ||||||||||||||||||
| Initial balance | and | differences an | balance at | |||||||||||||||||
| at 01/01/11 | Issue | Repayments | others | 30/06/11 | ||||||||||||||||
Debt securities issued in a member state of the European Union that have not
required the registration of a prospectus (nominal value) |
6,510 | 8,334 | (6,510 | ) | 0 | 8,334 | ||||||||||||||
Other debt securities issued ouside a member state of the European Union
(nominal value) |
416,147 | 0 | 0 | 72,404 | 488,551 | |||||||||||||||
TOTAL |
422,657 | 8,334 | (6,510 | ) | 72,404 | 496,885 | ||||||||||||||
| • | Company name : Grifols Inc. | ||
| • | Relationship with the company that publishes : Subsidiary | ||
| • | Country: United States of America | ||
| • | Rating: Standard & Poors: B; Moody’s: B3 | ||
| • | ISIN code : not aplicable as unquoted | ||
| • | Value Type : unsecured bond | ||
| • | Operation Date : 13 January 2011 | ||
| • | Issue amount : US Dollars 1.100 million | ||
| • | Balance : US Dollars 1.100 million | ||
| • | Interest rate : 8.25% | ||
| • | Unquoted | ||
| • | Guaranted by Grifols, SA, Grifols Inc., and other subsidiaries listed as guarantors in credit agreement. |
| (b) | Loans and borrowings |
| O | Non-current syndicated financing Tranche A: Senior Debt Loan repayable in five years divided into two tranches: U.S Tranche A and Foreign Tranche A. |
| ▪ | U.S Tranche A : |
| • | Aggregate Principal Amount of US Dollares 1,200 million. | ||
| • | Applicable margin of 375 basic points (bp) linked to US Libor. | ||
| • | Floor over US Libor of 1.75% |
| ▪ | Foreign Tranche A : |
| • | Aggregate Principal Amount of Euros 220 million. | ||
| • | Applicable margin of 400 basic points (bp) linked to Euribor. |
21
| • | Floor over Euribor of 1.75% |
| US Tranche A | Foreign Tranche A | |||||||||||||||||||
| Amortization in | Amortization in | |||||||||||||||||||
| thousands of US | thousands of | Amortization in | ||||||||||||||||||
| Currency | Dollar | Euros | Currency | thousands of Euros | ||||||||||||||||
Maturity |
||||||||||||||||||||
2012 |
US Dollars | 112,500 | 77,839 | Euros | 20,625 | |||||||||||||||
2013 |
US Dollars | 127,500 | 88,217 | Euros | 23,375 | |||||||||||||||
2014 |
US Dollars | 180,000 | 124,542 | Euros | 33,000 | |||||||||||||||
2015 |
US Dollars | 585,000 | 404,760 | Euros | 107,250 | |||||||||||||||
2016 |
US Dollars | 195,000 | 134,920 | Euros | 35,750 | |||||||||||||||
Total |
US Dollars | 1,200,000 | 830,277 | Euros | 220,000 | |||||||||||||||
| ▪ | U.S Tranche B : |
| • | Aggregate Principal Amount of US Dollars 1,300 million. | ||
| • | Applicable margin of 425 basic points (bp) linked to US Libor. | ||
| • | Floor over US Libor of 1.75% |
| ▪ | Foreign Tranche B : |
| • | Aggregate Principal Amount of Euros 220 million. | ||
| • | Applicable margin of 450 basic points (bp) linked to Euribor. | ||
| Floor over Euribor of 1.75% |
22
| US Tranche B | Foreign Tranche B | |||||||||||||||||||
| Amortization in | Amortization in | |||||||||||||||||||
| thousands of US | thousands of | Amortization in | ||||||||||||||||||
| Currency | Dollar | Euros | Currency | thousands of Euros | ||||||||||||||||
Maturity |
||||||||||||||||||||
2011 |
US Dollars | 6,500 | 4,497 | Euros | 1,100 | |||||||||||||||
2012 |
US Dollars | 13,000 | 8,995 | Euros | 2,200 | |||||||||||||||
2013 |
US Dollars | 13,000 | 8,995 | Euros | 2,200 | |||||||||||||||
2014 |
US Dollars | 13,000 | 8,995 | Euros | 2,200 | |||||||||||||||
2015 |
US Dollars | 13,000 | 8,995 | Euros | 2,200 | |||||||||||||||
2016 |
US Dollars | 9,750 | 6,746 | Euros | 1,650 | |||||||||||||||
2017 |
US Dollars | 1,231,750 | 852,245 | Euros | 208,450 | |||||||||||||||
Total |
US Dollars | 1,300,000 | 899,467 | Euros | 220,000 | |||||||||||||||
| ▪ | U.S Revolving Credit Facility : |
| • | Commited Amount: US Dollars 50 million | ||
| • | Applicable margin of 375 basis point (bp). |
| ▪ | U.S. Multicurrency Revolving Credit Facility: |
| • | Commited Amount: US Dollars 200 million | ||
| • | Applicable margin of 375 basis point (bp) |
| ▪ | Foreign Revolving Credit Facility : |
| • | Commited Amount : US Dollars 50 million. | ||
| • | Applicable margin of 400 basis point (bp). |
23
| Thousands of Euros | ||||||||||||
| Tranche A and B Senior | ||||||||||||
| High Yield Bond | Loan | |||||||||||
Maturity |
||||||||||||
2011 |
59,301 | 80,474 | ||||||||||
2012 |
62,790 | 235,792 | ||||||||||
2013 |
62,790 | 241,228 | ||||||||||
2014 |
62,790 | 279,372 | ||||||||||
2015 |
62,790 | 613,129 | ||||||||||
2016 |
62,790 | 248,557 | ||||||||||
2017 |
62,790 | 1,087,608 | ||||||||||
2018 |
763,994 | 0 | ||||||||||
| Total | 1,200,034 | 2,786,160 | ||||||||||
24
| Thousands of euros | ||||||||||||||||
| Notional at | Notional at | Value at | ||||||||||||||
| 30/06/11 | 31/12/10 | 30/06/11 | Value at 31/12/10 | |||||||||||||
| Financial Derivatives | ||||||||||||||||
Interest Rate Swap |
50,000 | 50,000 | (1,146 | ) | (1,809 | ) | ||||||||||
Interest Rate Swap (Cash flow hedge) |
1,072,442 | — | (3,829 | ) | — | |||||||||||
Implicit Floor |
3,113,540 | — | (54,364 | ) | — | |||||||||||
Currency Rate Swap |
47,800 | — | (2,346 | ) | — | |||||||||||
Liability |
4,283,782 | 50,000 | (61,685 | ) | (1,809 | ) | ||||||||||
Unquoted future |
17,416 | 23,221 | 3,344 | (2,821 | ) | |||||||||||
Unquoted future |
26,370 | 26,370 | 4,078 | (3,930 | ) | |||||||||||
Swap floor |
1,072,442 | — | 32,558 | — | ||||||||||||
Assets |
1,116,227 | 49,591 | 39,980 | (6,751 | ) | |||||||||||
25
| Thousand Euros | ||||||||
| Current financial liabilities | 30/06/11 | 31/12/10 | ||||||
Talecris bonds (note 10) |
427,691 | 0 | ||||||
Transaction costs High Yield Bonds |
(18,032 | ) | 0 | |||||
Interest accrued on bonds |
27,907 | 7,207 | ||||||
Promisory notes |
9,586 | 8,235 | ||||||
Bonds |
447,152 | 15,442 | ||||||
Tranche A (US Dollars) |
0 | 0 | ||||||
Tranche B (US Dollars) |
6,746 | 0 | ||||||
Tranche A (Euros) |
6,875 | 0 | ||||||
Tranche B (Euros) |
2,200 | 0 | ||||||
Transaction costs on loans and borrowings |
(37,216 | ) | (708 | ) | ||||
Club Deal |
0 | 66,667 | ||||||
Other loans |
75,079 | 106,954 | ||||||
Finance lease liabilities |
6,538 | 3,280 | ||||||
Loans and borrowings |
60,222 | 176,193 | ||||||
Loans and borrowings and bonds or other current
marketeable securities |
507,374 | 191,635 | ||||||
Financial derivatives |
7,320 | 8,560 | ||||||
Other current financial liabilities |
10,016 | 9,676 | ||||||
Other current financial liabilities |
17,336 | 18,236 | ||||||
| 524,710 | 209,871 | |||||||
26
| Consolidated | ||||||||
| June 2011 | June 2010 | |||||||
Men |
5,283 | 2,831 | ||||||
Women |
5,891 | 3,050 | ||||||
Total |
11,174 | 5,881 | ||||||
27
28
29
| Thousand Euros | ||||||||||||||||
| Key management | Other related | Board of directors | ||||||||||||||
| Associates | personnel | parties | of the company | |||||||||||||
Net sales
|
21 | — | — | — | ||||||||||||
Other service expenses
|
(1,690 | ) | (15,045 | ) | (120 | ) | ||||||||||
Personnel expenses
|
— | (3,250 | ) | — | (1,168 | ) | ||||||||||
Sales of Property |
||||||||||||||||
Plant and Equipment
|
— | — | 80,393 | — | ||||||||||||
| (1,669 | ) | (3,250 | ) | 65,348 | (1,288 | ) | ||||||||||
| Thousand Euros | ||||||||||||
| Key management | Other related | Board of directors | ||||||||||
| personnel | parties | of the company | ||||||||||
Other service expenses |
— | (5,912 | ) | (90 | ) | |||||||
Personnel expenses |
(2,931 | ) | — | (1,033 | ) | |||||||
| (2,931 | ) | (5,912 | ) | (1,123 | ) | |||||||
30
31


| DISCLAIMER The facts and figures contained in this report which do not refer to historical data are “projections and forward-looking statements”. The words and expressions like “believe”, “hope”, “anticipate”, “predict”, “expect”, “intend”, “should”, “try to achieve”, “estimate”, “future” and similar expressions, insofar as they are related to Grifols Group, are used to identify projections and forward-looking statements. These expressions reflect the assumptions, hypothesis, expectations and anticipations of the management team at the date of preparation of this report, which are subject to a number of factors that could make the real results differ considerably. The future results of Grifols Group could be affected by events related to its own activity, such as shortages of raw materials for the manufacture of its products, the launch of competitive products or changes in the regulations of markets in which it operates, among others. At the date of preparation of this report Grifols Group has adopted the measures it considers necessary to offset the possible effects of these events. Grifols, S.A. does not assume any obligation to publicly inform, review or update any projections and forward-looking statements to adapt them to facts or circumstances following the preparation of this report, except as specifically required by law. This document does not constitute an offer or invitation to purchase or subscribe shares, in accordance with the provisions of the Spanish Securities Market Law 24/1988, of July 28, the Royal Decree-Law 5/2005, of March 11, and/or Royal Decree 1310/2005, of November 4, and its implementing regulations. |

| GRIFOLS COMPLETES BIOSCIENCE GENERATES OVER 80% OF THE NEW GRIFOLS’ |
| THE ACQUISITION OF 80%1 OF THE NEW SALES ARE GENERATED IN |
| TALECRIS AND INITIATES THE GROUP’S INCOME INTERNATIONAL MARKETS. |
| INTEGRATION PROCESS. IT IS EXPECTED SALES IN US |
| THE HIGHER SALE VOLUMES OF AND CANADA WILL ACCOUNT |
| SALES INCREASE IN ALL PLASMA DERIVED PRODUCTS FOR 60% OF THE REVENUES |
| DIVISIONS1 OF THE NEW GROUP SUCH AS IVIG, FACTOR VIII IN THE MEDIUM TERM |
| AND ALBUMIN, CONFIRM THE |
| FIRST STEP TOWARDS THE UPWARD TREND OF NET FINANCIAL DEBT |
| REALIZATION OF OPERATING THE SECTOR. ALPHA AT LOWER LEVELS THAN |
| SYNERGIES: GRIFOLS HAS 1-ANTITRYPSIN GAINS EXPECTED FOR THE CLOSING |
| OBTAINED FDA APPROVAL PROMINENCE IN OF THE TRANSACTION. |
| FOR THE UTILIZATION OF THE THE SALES MIX NET DEBT OVER |
| LOS ANGELES PLANT FRACTION RECURRING2 EBITDA |
| II+III (INTERMEDIATE PRODUCT) SALES IN THE US AND IN STANDS AT 4.4 X |
| IN THE PRODUCTION OF CANADA, A NEW MARKET IN COMPARED TO THE |
| GAMUNEX ®, TALECRIS’ IVIG. THE MIX, GROW 70% EXPECTED LEVEL OF 5.2 X |
| GRIFOLS AVERAGE |
| WORKFORCE EXCEEDS |
| 11,100 EMPLOYEES |
| 1 Includes Talecris’ results for June 2011, first month consolidated |
| 2 Excluding costs associated to the transaction of Talecris and non recurring costs |
| 3 Pro-forma unaudited figures obtained from the consolidated statements of both companies for the 6 months period to 30 June 2011. Provided for guidance purposes only. |
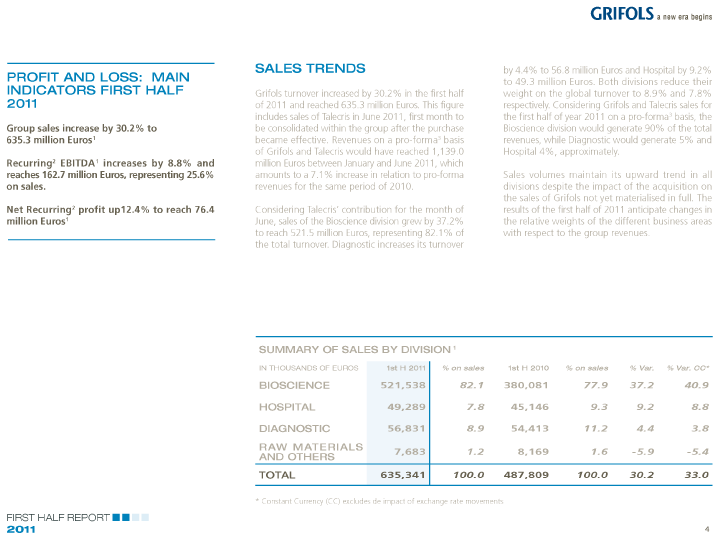
| SALES TRENDS by 4.4% to 56.8 million Euros and Hospital by 9.2% |
| PROFIT AND LOSS: MAIN to 49.3 million Euros. Both divisions reduce their |
| INDICATORS FIRST HALF Grifols turnover increased by 30.2% in the first half weight on the global turnover to 8.9% and 7.8% |
| 2011 of 2011 and reached 635.3 million Euros. This figure respectively. Considering Grifols and Talecris sales for |
| includes sales of Talecris in June 2011, first month to the first half of year 2011 on a pro-forma3 basis, the |
| Group sales increase by 30.2% to be consolidated within the group after the purchase Bioscience division would generate 90% of the total |
| 635.3 million Euros1 became effective. Revenues on a pro-forma3 basis revenues, while Diagnostic would generate 5% and |
| of Grifols and Talecris would have reached 1,139.0 Hospital 4%, approximately. |
| Recurring2 EBITDA1 increases by 8.8% and million Euros between January and June 2011, which |
| reaches 162.7 million Euros, representing 25.6% amounts to a 7.1% increase in relation to pro-forma Sales volumes maintain its upward trend in all |
| on sales. revenues for the same period of 2010. divisions despite the impact of the acquisition on |
| the sales of Grifols not yet materialised in full. The |
| Net Recurring2 profit up12.4% to reach 76.4 Considering Talecris’ contribution for the month of results of the first half of 2011 anticipate changes in |
| million Euros1 June, sales of the Bioscience division grew by 37.2% the relative weights of the different business areas |
| to reach 521.5 million Euros, representing 82.1% of with respect to the group revenues. |
| the total turnover. Diagnostic increases its turnover |
| SUMMARY OF SALES BY DIVISION 1 |
| IN THOUSANDS OF EUROS 1st H 2011% on sales 1st H 2010% on sales % Var. % Var. CC* |
| BIOSCIENCE 521,538 82.1 380,081 77.9 37.2 40.9 |
| HOSPITAL 49,289 7.8 45,146 9.3 9.2 8.8 |
| DIAGNOSTIC 56,831 8.9 54,413 11.2 4.4 3.8 |
| RAW MATERIALS |
| AND OTHERS 7,683 1.2 8,169 1.6 -5.9 -5.4 |
| TOTAL 635,341 100.0 487,809 100.0 30.2 33.0 |
| * Constant Currency (CC) excludes de impact of exchange rate movements |

| The acquisition further modifies the geographic Considering the geographical fit of the markets of |
| mix of income. In the first half of 2011, 42% of both companies prior to the integration, it is worth |
| sales have been generated in the US and Canada, a noting the significant growth of revenues from the |
| new significant market for the group, while 38.7% US and Canada. During the first half of 2011, sales in |
| of sales were generate in Europe. Areas such as these regions increased by 69.1% and exceeded 266 |
| Australia gain prominence. In this respect, we note million Euros1. In Europe, sales increased by 10.4% |
| how over 80% of Grifols’ activities are generated and reached 246.1 million Euros1 as expected; with |
| outside of Spain, whose relative weight decreases increase in market share in Germany and Portugal |
| to 19% as compared to 24.5% in the same period and significant growth in Australia. |
| of 2010. |
| SUMMARY OF SALES BY REGION1 |
| IN THOUSANDS OF EUROS 1st H 2011% on sales 1st H 2010% on sales % Var % Var. CC* |
| EU 246,144 38.7 223,019 45.7 10.4 10.1 |
| US+CANADA 266,547 42.0 157,620 32.3 69.1 79.3 |
| R.O.W. 120,992 19.0 105,335 21.6 14.9 13.0 |
| SUBTOTAL 633,683 99.7 485,974 99.6 30.4 33.2 |
| RAW MATERIALS 1,658 0.3 1,835 0.4 -9.6 -6.8 |
| TOTAL 635,341 100.0 487,809 100.0 30.2 33.0 |

| MARGIN ANALYSIS Pro-forma3 results of Grifols and Talecris show how FINANCIAL EXPENSES |
| recurrent2 EBITDA between January and June 2011 ARISING FROM THE NEW |
| The effect of healthcare reforms that had not would reach 305.6 million Euros, or 26.8% of sales, FINANCING STRUCTURE |
| yet affected first half 2010 results, the negative down by 2.7% with respect to the pro-forma gross |
| contribution of prices to the performance of revenues operating result for the same period of 2010. |
| and the impact of higher costs of raw materials Financial expenses increased in the first six months of |
| (plasma), have all had a direct impact on the gross Subsequent to the closing of the first half of 2011 in line with expectations, reaching 55 million |
| margin, which was 45% over sales1 . 2011, Grifols has obtained FDA approval for the Euros1 .The increase from 25.8 million Euros in the |
| utilization of Fraction II+III of the Los Angeles’ plant fist half of 2010, is the result of new financing |
| The recurring EBITDA2 grew by 8.8% to 162.7 million (intermediate product) in the purification of the IVIG raised through syndicated loans and a new bond |
| Euros1, during the first half of 2011, representing to obtain Talecris’ final product, Gamunex®. This issued in 2011 to meet the cost of the acquisition of |
| 25.6% of sales. approval will enable to increase production with a Talecris. It also includes previously capitalized costs |
| higher yield that will bring margin improvements in related to the group’s debt cancelled as a result of |
| Transaction costs related to the acquisition of Talecris the medium term. the purchase. |
| and non recurring impact the gross operating results |
| of the period by over 65 million Euros, resulting in Thus, Grifols’ recurring2 net profit increases by 12.4% |
| an EBITDA of 96.9 million Euros1. to 76.4 million Euros1, which amounts to 12.0% of |
| revenues. Considering the expenses relating to the |
| acquisition, net profit for the first 6 months to June |
| 2011 reached 19.3 million Euros1, representing 3% |
| on sales. |
| IN MILLIONS OF EUROS 1st H 2011 1st H 2010% var. |
| EBITDA 96.9 147.6 -34.4 |
| % ON SALES 15.2 30.2 |
| ADJUSTED EBITDA2162.7149.6 8.8 |
| % ON SALES 25.6 30.7 |
| NET PROFIT 19.3 66.4 -71.0 |
| % ON SALES 3.0 13.6 |
| ADJUSTED NET PROFIT2 76.4 67.9 12.4 |
| % ON SALES 12.0 13.9 |

| BALANCE SHEET: building works of a new fractionation plant in Parets MAIN INDICATORS del Vallès (Barcelona, Spain), which will have capacity to fractionate 1 million liters per year (expandable to PROJECTED CAPITAL 2 million); the investments undertaken in the new INVESTMENT (CAPEX) albumin production plant in Los Angeles (USA) and MAINTAINED. the completion in Barcelona, Spain, of the “Grifols Academy”, a meeting point for advanced training on all processes related to hemoderivatives production. Total consolidated assets as at June 2011 reached 5.344,2 million Euros, as compared to 1,889.0 The increase in intangible fixed assets mainly relates million Euros reported at year end 2010. to the goodwill generated through the acquisition of Talecris for an estimated amount of 2,124 million Tangible fixed assets have increased by over 200 Euros. At the time of publication, this is a provisional million Euros, as a result of the acquisition of amount as there was not yet sufficient information Talecris assets, including the plasma fractionation to determine the fair value of the relevant balance plant, located in Clayton (North Carolina, USA) sheet items. and several collection centers. In addition, Grifols has continued with the projected investment plan During the period, the Australian market performed (CAPEX), allocating over 31 million Euros to the worse than expected. As a result, an evaluation was expansion and improvement of its production carried out of the goodwill relating to investments facilities, given that the investments planned for in the country that triggered a 13 million Euros 2011 and 2012 are independent from the Talecris adjustment to its value. This resulted in a lower acquisition. Among the above, the start of the profit. |

| NET FINANCIAL DEBT NEW FINANCING STRUCTURE |
| BEATS ESTIMATES |
| SENIOR SECURED DEBT AMOUNT TERM CONDITIONS |
| Grifols’ net financial debt stand, at the end of the MILLIONS OF DOLLARS |
| first half of 2011, at 2,595.3 million Euros, 4.4 x over |
| recurring2 EBITDA, lower than the 5.2 x anticipated TRANCHE A $1,500 5 YEARS 3.75 / 4.00% |
| at the closing of the transaction. In this respect, the TRANCHE B $1,600 6 YEARS 4.25 / 4.50% |
| projected increase in short term cash flows to reduce |
| leverage in a swift manner is confirmed. Grifols REVOLVING CREDIT FACILITY $300 6 YEARS 3.75 / 4.00% |
| estimates that the net financial debt to EBITDA ratio |
| will return to levels previous to the purchase once TOTAL $3,400 |
| synergies have been achieved. |
| SENIOR UNSECURED DEBT |
| CORPORATE BOND ISSUANCE $1,100 7 YEARS 8.25% |
| In addition, the geographic redistribution of sales |
| following the acquisition will allow for an increase |
| of the group’s exposure to countries with shorter |
| collection periods, and it is anticipated that this will |
| contribute to gradually optimizing the short term |
| financing needs and to improve working capital. |
| Inventory levels have decrease moderately during |
| the first half of 2011 as a result of measures |
| implemented by Grifols. This trend, which started |
| in the first quarter of 2011, will be reinforced along |
| the year as a result of the acquisition of Talecris. |

| GRIFOLS’ NET EQUITY As a result of the new share issue, and in addition DOUBLES to increasing the share capital of the company, the share premium has also increased by 768.5 The acquisition of Talecris has entailed a significant million Euros, reaching 890.3 million Euros. Grifols increase of the group’s equity, as a result of the shareholders approved at the Annual Shareholders issue of new non-voting shares (Class B) of Grifols Meeting the allocation to reserves of 2010 net profit to cover the non-cash consideration portion. As at in its entirety thus increasing Equity funds by 115.5 June 2011, Grifols’ net equity amounts to 1,513.6 million Euros. million Euros that compared to the 707.4 million Euros reported at year end 2010, shows an increase As at 2011, the share capital of the company was exceeding 806 million Euros. 114.91 million Euros, represented by 213,064,899 ordinary shares (Class A) and 83,822,688 non-voting shares (Class B). |

| ANALYSIS BY BUSINESS Bioscience Division: 82% of revenues1 such as Germany, Chile and Argentina. In global AREAS: terms, Australia and Canada join in as important Sales of the Bioscience divisions, including June generators of hemoderivatives demand from Grifols, FAVORABLE EVOLUTION IN 2011 Talecris’ sales, increased by 37.2% to 521.5 whereas in terms of products, sales of alpha -1 ALL DIVISIONS million Euros. The increase in sales volumes of antitrypsin gain prominence. hemoderivatives has been the main driver of the The recent acquisition will allow Grifols to division’s growth, with a negative impact of prices in significantly expand its fractionation installed The operating results obtained by the group1 some countries. In addition the portfolio of available capacity. Following completion of the transaction, witness to the positive evolution of sales in all hemoderivative products is expanded with new trade the group has 4 facilities available in the US and in divisions, and confirm Grifols’ leadership in the references, which are maintained to meet the specific Spain, allowing for the fractionation of a maximum plasma proteins industry, as the third company needs of patients and healthcare professionals in the of 8.5 million liters of plasma per year in aggregate. by sales volume world wide. The integration plan various markets. Furthermore, Grifols has become the world leader in currently underway will help to obtain the anticipated terms of plasma collection capacity. It currently has synergies, on the basis of cost optimization and By products, the sales of intravenous immunoglobulin 147 plasmapheresis centers in the US, from which it increased efficiency in all stages of the production (IVIG) should be highlighted, boosted by significant can obtain over 6.5 million liters of plasma per year, processes. Grifols consolidates its basis for future increases in the US, Asia-Pacific and Australia among thus maximizing and ensuring self-sufficient supply of growth by maintaining its internationalization, others. Similarly, sales of factor VIII and albumin raw materials necessary to produce plasma-derived product diversification, promoting R+D and planning increased, with relevant growths posted by countries, protein therapies. investments as the strategic management pillars. |

| Diagnostic Division: 8.9% of sales1 of Immunohemathology and Blood Bank in the Hospital Division: 7.8% of turnover1 |
| so-called Transfusion Medicine area. Highlights of |
| Diagnostic increases its turnover by 4.4% to 56.8 the period are the test implementation of the Wi-Fi Revenues from the Hospital division have increased |
| million Euros. Significant are the increases in version of the Gricode® transfusion safety system at by 9.2% until June 2011, reaching 49.2 million |
| the blood bank (10.1%); pathogen inactivation the Legnano hospital (Italy), allowing for data read Euros. The increase in sales of I.V. therapies (13.4%), |
| (28.4%); and new technologies (20.4%) areas. This with the Gricode® reader to be downloaded to the medical devices (10.7%) and hospital logistics area |
| division counts with an international footprint as blood bank automatically and in real time. (7.8%) in an environment of budgetary contention |
| well as multiple potential growth paths. With this on the part of hospitals, have been driving factors |
| objective in mind, Grifols has grouped the areas for the good performance of revenues. In addition, |
| we should highlight the international and the |
| geographical diversification strategy initiated for |
| MAIN RESULTS OF GRIFOLS IN THE FIRST HALF OF 20111 the division through 3rd party agreements. Among |
| these, a 5 year period agreement with CareFusion, |
| MILLIONS OF EUROS 1st H 2011 1sr H 2010% Var. a global leader in medical technology, to distribute |
| the BlisPack® system, throughout several countries |
| RECURRING EBITDA2 162.7 149.6 +8.8 of Europe, Middle East, Africa and Asia. |
| % ON SALES 25.6 30.7 The BlisPack® is a system designed by Grifols to |
| RECURRING NET PROFIT276.4 67.9 +12.4 automate blister cutting, and identify drugs for |
| hospital use by electronic means. |
| % ON SALES 12.0 13.9 |
| EBITDA 96.9 147.6 -34.4 |
| % ON SALES 15.2 30.2 |
| NET PROFIT 19.3 66.4 -71.0 |
| % ON SALES 3.0 13.6 |

| SECOND QUARTER OF 2011 HIGHLIGHTS |
| Grifols successfully completes the purchase of |
| Talecris |
| On 2 June 2011, Grifols concluded the acquisition of 100% of Talecris shares, becoming the third producer of plasma derivatives worldwide by sales volumes. Grifols is, in addition, the leading European company in the industry, with a well-balanced and diversified range of products. After the acquisition, Grifols has direct commercial presence through subsidiaries in 24 countries. |
| Grifols non-voting shares listed in NASDAQ and in the |
| Spanish market |
| From June 2011, Grifols non-voting shares (Class B) are listed in the Spanish market (GRF.P) and in NASDAQ (GRFS) via ADSs (American Depositary Shares). Since 2006, Grifols’ ordinary stock (Class A) is listed in the Spanish stock exchange, and since 2008 it is part of the Ibex-35 index (GRF). |
| Grifols starts the integration process First step towards the realization of operating synergies: Grifols has obtained FDA approval Grifols has already defined its new operations for the utilization of an intermediate product steering committee for the US operations, through in the production of Gamunex® which the integration process will be fostered. |
| Grifols has also set up several task forces with a view Subsequent to the closing of the first half of 2011, to assessing and combining the best expertise and Grifols has obtained FDA approval for the utilization implement the best practices. of Los Angeles’ plant Fraction II+III (intermediate product) in the purification of IVIG to obtain Talecris’ final product, Gamunex®. This approval is an important step towards achieving the operating synergies designed by the group, in particular those relating to cost optimization of raw materials, as it will enable to increase the yield per liter of plasma utilized in the medium term. |

| Opening of Grifols’ new corporate headquarters The US Chamber of Commerce in Spain award in Spain to Mr. Víctor Grifols Roura, Chairman and Chief Executive Officer of Grifols |
| In June, Grifols opened its new head office in San Cugat del Vallès (Barcelona, Spain), in an act chaired Víctor Grifols has been awarded the Global by Miguel Sebastián, Ministry of Industry, Tourism Business Leader Award 2011 by the US Chamber and Commerce of the Spanish Government. The of Commerce in Spain. The award recognizes the ministry and other authorities visited the facilities achievements of business leaders contributing to a accompanied by Víctor Grifols and other directors responsible economic globalization. and management of the group. The opening of the new headquarters coincides with the group’s 70th anniversary and in the construction of the building; top environmental standards have been applied regarding lighting, HVAC and use of water. |

| CORPORATE Grifols has approved the new Environmental |
| RESPONSIBILITY: Programme for the period 2011-2013, focused on |
| the following strategic lines: |
| Research as a commitment • Consumptions: Foster a reduction of |
| consumption of raw materials such as alcohol |
| In the first half of 2011 Grifols’ investments in R+D, or acetone, up to an aggregate reduction of raw |
| including the technical area, exceeded 30 million materials procurement by 2,000 T. |
| Euros, 4,7% of revenues obtained, and doubling • Water cycle: Optimize consumption and reuse |
| the amount allocated to research in the first half of of clean waters for ancillary processes such as |
| 2010. This emphasizes the commitment of the new refrigeration, with a target of reducing overall |
| group with scientific development and society. Grifols water consumption by 20,000 m3/year. |
| features a significant portfolio of R+D projects and • Waste: Enhance value of waste in production |
| has the necessary resources to ensure the group’s areas and warehouses by over 3,000 T, reaching |
| continuing research activity in the long term. up to 70% of all waste produced. |
| Furthermore, the new group has announced that • Energy: A Corporate Plan of strategic actions |
| it will foster research in other fields with projection in energy for the period 2010-2012 has been |
| of future, such as regenerative medicine, through developed. The plan includes measures for the |
| Gri-cel. design and implementation of more efficient |
| production processes and optimizing energy |
| Environmental management. use in the new ancillary facilities. Measures |
| undertaken shall entail power savings of 3,600 |
| The “Environmental targets Programme 2008- MWh per year, and natural gas savings of 6,000 |
| 2010” was completed in the first half of year MWh/year. |
| 2011, including the relevant follow-up by the • The programme further includes other |
| Environmental Committees. The overall achievement objectives, such as land pollution prevention |
| rate of the targets was 85%. measures, extending environmental |
| In addition, emissions generated by the facilities considerations to projects developed for |
| of the Bioscience division in Spain, subject to the third parties and extending environmental |
| European CO2 Emissions Trading System were management to all production centers of the |
| verified. In 2010, emissions reached 19,764 t, i.e. Company. |
| 14.5% less than in the prior year. |

| Firm commitments towards Human Resources years and always committed to equal opportunities associations and hospitals, schools and universities, employment for men and women alike (Gender split: among others. This approach stems from drawing In June 2011, with the Talecris acquisition, the 46% men and 54% women) a parallel between business and academy. average aggregate headcount at Grifols raised to 11,174 employees, increasing by 87% with respect From the point of view of training, the Academy However Grifols’ commitment to fostering training to the end of 2010. The detail of the average has been opened in Spain during the quarter. It initiatives consistent with the degree of specialization headcount evolution is as follows: is a meeting point for advanced training on all required by the hemoderivatives industry is not new. processes related to the preparation and production In 2009, the group opened the “Grifols Academy of Over 74% of Grifols’ employees are based in North of plasma-derived medicines. In addition, the Plasmaphaeresis” in Phoenix (Arizona, USA), which America, geographically distributed as follows: “Grifols Academy” shall be a dissemination centre has already been attended by 905 participants and for scientific and business knowledge, fostering at which over 8,628 hours of training have been Grifols is currently a working place of reference a continued exchange among experts and other taught in 2010. worldwide, reaching an average seniority of over 6 external bodies, such as healthcare professional |
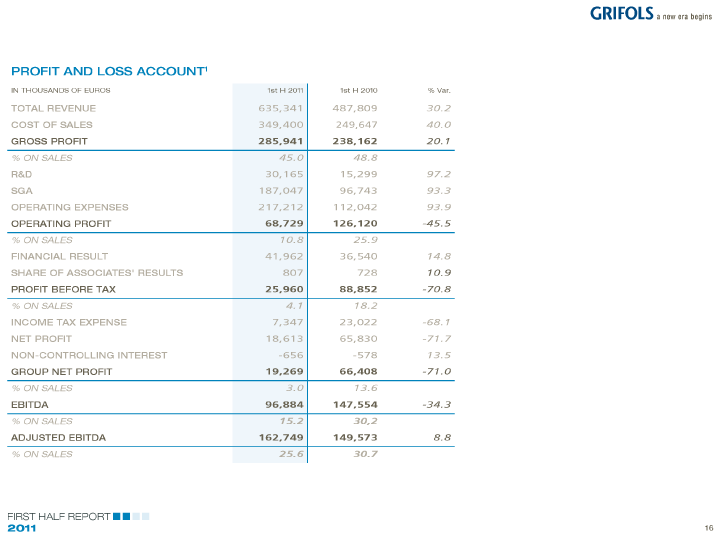
| PROFIT AND LOSS ACCOUNT1 |
| IN THOUSANDS OF EUROS 1st H 2011 1st H 2010% Var. |
| TOTAL REVENUE 635,341 487,809 30.2 |
| COST OF SALES 349,400 249,647 40.0 |
| GROSS PROFIT 285,941 238,162 20.1 |
| % ON SALES 45.0 48.8 |
| R&D 30,165 15,299 97.2 |
| SGA 187,047 96,743 93.3 |
| OPERATING EXPENSES 217,212 112,042 93.9 |
| OPERATING PROFIT 68,729 126,120 -45.5 |
| % ON SALES 10.8 25.9 |
| FINANCIAL RESULT 41,962 36,540 14.8 |
| SHARE OF ASSOCIATES’ RESULTS 807 728 10.9 |
| PROFIT BEFORE TAX 25,960 88,852 -70.8 |
| % ON SALES 4.1 18.2 |
| INCOME TAX EXPENSE 7,347 23,022 -68.1 |
| NET PROFIT 18,613 65,830 -71.7 |
| NON-CONTROLLING INTEREST -656 -578 13.5 |
| GROUP NET PROFIT 19,269 66,408 -71.0 |
| % ON SALES 3.0 13.6 |
| EBITDA 96,884 147,554 -34.3 |
| % ON SALES 15.2 30,2 |
| ADJUSTED EBITDA 162,749 149,573 8.8 |
| % ON SALES 25.6 30.7 |

| CASH FLOW1 |
| IN THOUSANDS OF EUROS 1st H 2011 1st H 2010 |
| NET PROFIT 19,269 66,408 |
| DEPRECIATION AND AMORTITZATION 28,156 21,434 |
| NET PROVISIONS 14,455 129 |
| OTHER ADJUSTMENTS 30,818 11,936 |
| CHANGES IN INVENTORIES 752 (11,982) |
| CHANGES IN TRADE RECEIVABLES (67,041) 25,966 |
| CHANGES IN TRADE PAYABLES (9,715) 7,978 |
| CHANGE IN OPERATING WORKING CAPITAL (76,004) 21,962 |
| NET CASHFLOW FROM OPERATING ACTIVITIES* 16,694 121,869 |
| BUSINESS COMBINATIONS AND INVESTMENTS IN GROUP COMPANIES (1,615,417) (3,727) |
| CAPEX (PROPERTY.PLANT & EQUIP) (47,838) (45,600) |
| R&D/OTHER INTANGIBLE ASSETS (5,000) (3,551) |
| OTHER CASH INFLOW /(OUTFLOW) 68,016 (1,256) |
| NET CASHFLOW FROM INVESTING ACTIVITIES (1,600,239) (54,134) |
| FREE CASH FLOW (1,583,546) 67,735 |
| CAPITAL INCREASES (2,264) - |
| ISSUE (PURCHASE) OF TREASURY STOCK - (1,250) |
| ISSUE (REPAYMENT) OF DEBT 1,947,789 (8,671) |
| DIVIDENDS - (53) |
| OTHER 347 323 |
| NET CASHFLOW FROM FINANCING ACTIVITIES 1,945,872 (9,651) |
| TOTAL CASH FLOW 362,327 58,084 |
| CASH AND CASH EQUIVALENTS AT THE START OF THE YEAR 239,649 249,372 |
| EFFECT OF EXCHANGE RATE CHANGES IN CASH AND CASH EQUIVALENTS (18,184) 42,684 |
| CASH AND CASH EQUIVALENTS AT THE END OF THE PERIOD 583,792 350,140 |
| *Net cash flow from operating activities calculated with adjusted items would total 74.8 million Euros |
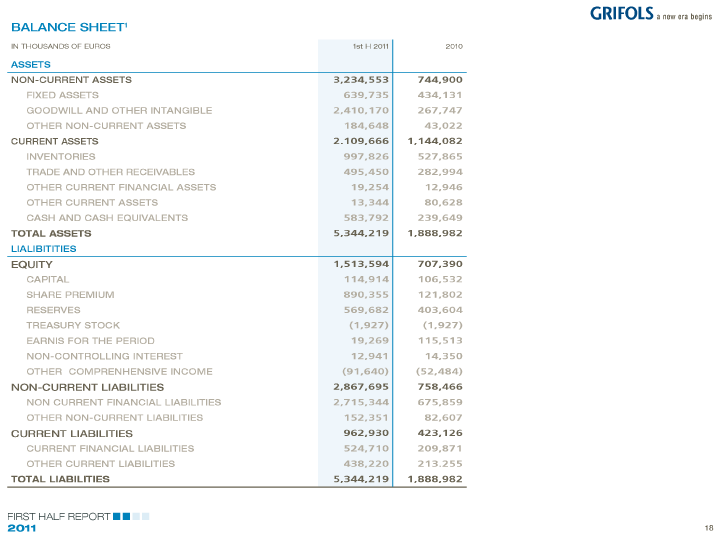
| BALANCE SHEET 1 |
| IN THOUSANDS OF EUROS 1st H 2011 2010 |
| ASSETS |
| NON-CURRENT ASSETS 3,234,553 744,900 |
| FIXED ASSETS 639,735 434,131 |
| GOODWILL AND OTHER INTANGIBLE 2,410,170 267,747 |
| OTHER NON-CURRENT ASSETS 184,648 43,022 |
| CURRENT ASSETS 2.109,666 1,144,082 |
| INVENTORIES 997,826 527,865 |
| TRADE AND OTHER RECEIVABLES 495,450 282,994 |
| OTHER CURRENT FINANCIAL ASSETS 19,254 12,946 |
| OTHER CURRENT ASSETS 13,344 80,628 |
| CASH AND CASH EQUIVALENTS 583,792 239,649 |
| TOTAL ASSETS 5,344,219 1,888,982 |
| LIALIBITITIES |
| EQUITY 1,513,594 707,390 |
| CAPITAL 114,914 106,532 |
| SHARE PREMIUM 890,355 121,802 |
| RESERVES 569,682 403,604 |
| TREASURY STOCK (1,927) (1,927) |
| EARNIS FOR THE PERIOD 19,269 115,513 |
| NON-CONTROLLING INTEREST 12,941 14,350 |
| OTHER COMPRENHENSIVE INCOME (91,640) (52,484) |
| NON-CURRENT LIABILITIES 2,867,695 758,466 |
| NON CURRENT FINANCIAL LIABILITIES 2,715,344 675,859 |
| OTHER NON-CURRENT LIABILITIES 152,351 82,607 |
| CURRENT LIABILITIES 962,930 423,126 |
| CURRENT FINANCIAL LIABILITIES 524,710 209,871 |
| OTHER CURRENT LIABILITIES 438,220 213.255 |
| TOTAL LIABILITIES 5,344,219 1,888,982 |
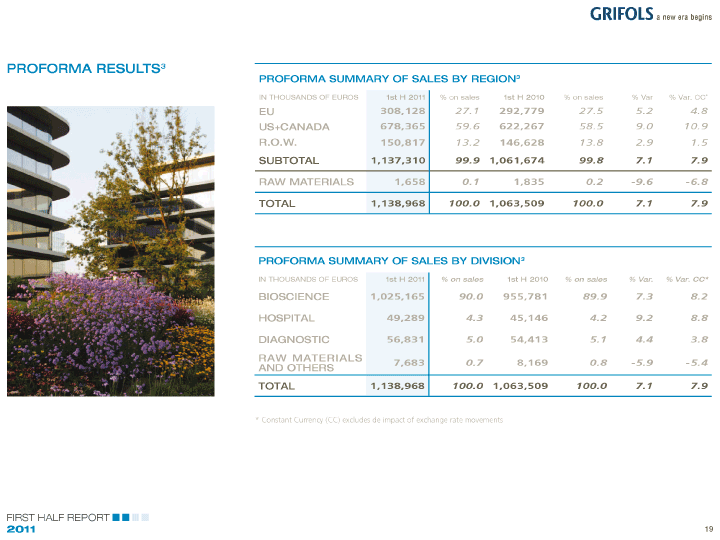
| PROFORMA SUMMARY OF SALES BY REGION3 |
| IN THOUSANDS OF EUROS 1st H 2011 % on sales 1st H 2010 % on sales % Var % Var. CC* |
| EU 308,128 27.1 292,779 27.5 5.2 4.8 |
| US+CANADA 678,365 59.6 622,267 58.5 9.0 10.9 |
| R.O.W. 150,817 13.2 146,628 13.8 2.9 1.5 |
| SUBTOTAL 1,137,310 99.9 1,061,674 99.8 7.1 7.9 |
| RAW MATERIALS 1,658 0.1 1,835 0.2 -9.6 -6.8 |
| TOTAL 1,138,968 100.0 1,063,509 100.0 7.1 7.9 |
| PROFORMA SUMMARY OF SALES BY DIVISION3 |
| IN THOUSANDS OF EUROS 1st H 2011% on sales 1st H 2010% on sales % Var. % Var. CC* |
| BIOSCIENCE 1,025,165 90.0 955,781 89.9 7.3 8.2 |
| HOSPITAL 49,289 4.3 45,146 4.2 9.2 8.8 |
| DIAGNOSTIC 56,831 5.0 54,413 5.1 4.4 3.8 |
| RAW MATERIALS |
| AND OTHERS 7,683 0.7 8,169 0.8 -5.9 -5.4 |
| TOTAL 1,138,968 100.0 1,063,509 100.0 7.1 7.9 |
| * Constant Currency (CC) excludes de impact of exchange rate movements |
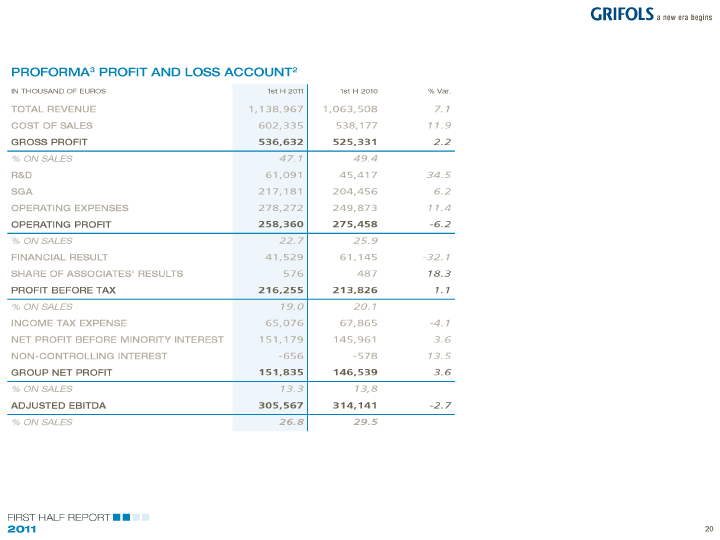
| PROFORMA3 PROFIT AND LOSS ACCOUNT2 |
| IN THOUSAND OF EUROS 1st H 2011 1st H 2010% Var. |
| TOTAL REVENUE 1,138,967 1,063,508 7.1 |
| COST OF SALES 602,335 538,177 11.9 |
| GROSS PROFIT 536,632 525,331 2.2 |
| % ON SALES 47.1 49.4 |
| R&D 61,091 45,417 34.5 |
| SGA 217,181 204,456 6.2 |
| OPERATING EXPENSES 278,272 249,873 11.4 |
| OPERATING PROFIT 258,360 275,458 -6.2 |
| % ON SALES 22.7 25.9 |
| FINANCIAL RESULT 41,529 61,145 -32.1 |
| SHARE OF ASSOCIATES’ RESULTS 576 487 18.3 |
| PROFIT BEFORE TAX 216,255 213,826 1.1 |
| % ON SALES 19.0 20.1 |
| INCOME TAX EXPENSE 65,076 67,865 -4.1 |
| NET PROFIT BEFORE MINORITY INTEREST 151,179 145,961 3.6 |
| NON-CONTROLLING INTEREST -656 -578 13.5 |
| GROUP NET PROFIT 151,835 146,539 3.6 |
| % ON SALES 13.3 13,8 |
| ADJUSTED EBITDA 305,567 314,141 -2.7 |
| % ON SALES 26.8 29.5 |
| GRIFOLS’ DAILY ORDINARY SHARE PRICE VS IBEX 35 |
| (BASE 100, FROM JANUARY 1 TO JUNE 30 2011) |
| 1 Includes Talecris’ results for June 2011, first month consolidated 2 Excluding costs associated to the transaction of Talecris and non recurring costs |
| 3 Pro-forma unaudited figures obtained from the consolidated statements of both companies for the 6 months period to 30 June 2011. Provided for guidance purposes only. |
| Grifols, S.A. |
||||
| By: | /s/ David I. Bell | |||
| Name: | David I. Bell | |||
| Title: | Authorized Signatory | |||