Exhibit 99.2









UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM
CURRENT REPORT PURSUANT
TO SECTION 13 OR 15(d) OF THE
SECURITIES EXCHANGE ACT OF 1934
Date
of Report (Date of earliest event reported):
(Exact name of registrant as specified in charter)
| (State or other jurisdiction of incorporation) | (Commission File Number) |
(IRS Employer Identification No.) |
| (Address of principal executive offices) | (Zip Code) |
Registrant’s
telephone number, including area code:
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) | |
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) | |
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) | |
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading symbol(s) | Name of each exchange on which registered | ||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1934 (§240.12b-2 of this chapter)
Emerging
growth company
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 2.02. Results of Operations and Financial Condition.
On March 31, 2022, INVO Bioscience, Inc. (the “Company”) issued a press release announcing financial results for the year ended December 31, 2021. The text of the press release is furnished as Exhibit 99.1 to this current report.
The information in this Item 2.02 and Exhibit 99.1 hereto shall not be deemed “filed” for the purposes of or otherwise subject to the liabilities under Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). Unless expressly incorporated into a filing of the Company under the Securities Act of 1933, as amended, or the Exchange Act, the information contained in this Item 2.02 and Exhibit 99.1 hereto shall not be incorporated by reference into any Company filing, whether made before or after the date hereof, regardless of any general incorporation language in such filing.
Item 8.01 Other Events
Exhibit 99.2 hereto contains a presentation by INVO Bioscience, Inc. entitled “INVO Center Review-March 2022” discussing the current status of its INVO Centers and plans for establishment of additional INVO Centers.
.
Item 9.01 Financial Statements and Exhibits
(d) Exhibits.
| Exhibit No. | Description | |
| 99.1 | Press Release dated March 31, 2022. | |
| 99.2 | Presentation entitled “INVO Center Review-March 2022” |
| -2- |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
| INVO BIOSCIENCE, INC. | ||
| By: | /s/ Steven Shum | |
| Name: | Steven Shum | |
| Title: | Chief Executive Officer | |
| Dated March 31, 2022 | ||
| -3- |
Exhibit 99.1
INVO Bioscience Reports 2021 Financial Results
Company to Host Conference Call Today at 4:30pm ET; Slide Presentation Also Available
SARASOTA, Fla., March 31, 2022 -- INVO Bioscience, Inc. (Nasdaq: INVO) (“INVO” or the “Company”), a commercial-stage fertility company focused on expanding access to advanced treatment worldwide with its INVOcell® medical device and the intravaginal culture (“IVC”) procedure it enables, today announced financial results for the year ended December 31, 2021, and provided a business update.
Recent Operational Highlights
| ● | Regained full U.S. commercialization rights to sell its INVOcell® solution directly into existing in vitro fertilization (IVF) clinics, to expand the number of U.S. INVO Centers free of any limitations, and to pursue its market expansion strategy focused on increasing access to care and democratizing fertility care for the underserved patient population. | |
| ● | Opened three INVO Center’s in 2021, including Birmingham, Alabama, Atlanta, Georgia, and Monterrey, Mexico with another clinic scheduled to open in the San Francisco area. The Company recently identified Tampa, Florida as the location of the next INVO Center. | |
| ● | Entered into an agreement with Ovo in Spain to expand INVOcell® commercialization in all four of their Ovoclinics, establish a European center of excellence and training site for INVOcell® and the IVC procedure, and leverage Ovobank’s fertility clinic relationships across Europe. | |
| ● | Completed the acquisition of a Canadian-based entity, originally formed to offer INVOcell®, to advance distribution efforts in Canada, where the product is already approved by Health Canada and cleared for importing. | |
| ● | Obtained regulatory approval to commercialize INVOcell® in Thailand and Nigeria. | |
| ● | Presented four poster abstracts discussing INVOcell® at the 77th Scientific Congress & Expo of the American Society for Reproductive Medicine held in Baltimore, Maryland in October 2021. | |
| ● | Revenue was $4.2 million for the year ended December 31, 2021, including $2.9 million due to the accelerated recognition of deferred revenue from the Ferring license, and compared to $1.0 million for the previous year. | |
| ● | Adjusted EBITDA for 2021 was $(2.8) million, which included a $0.7 million loss attributable to the Company’s joint ventures, compared to $(3.7) million in the prior year (see Adjusted EBITDA Table). |
Management Commentary
“2021 was an exciting year for INVO as we opened our first INVO Center clinics,” commented Steve Shum, CEO of INVO. “Our INVO Center model represents a strategy aimed at expanding adoption of INVOcell® and allowing us to capture a significantly larger percentage of the per-cycle IVC procedure revenue it enables. The INVO Centers are designed to offer attractive economics for our shareholders, medical practitioner partners and patients, and we believe are key to our efforts to address the fertility industry’s biggest challenges of cost and capacity. In addition to our announced plans to open additional centers in the San Francisco Bay Area and Tampa, Florida, we are evaluating over 20 additional US locations which represent attractive opportunities to open new INVO Centers. We also are in discussions for similar ventures across the globe. On the distribution side of our business, during the first quarter of 2022, we began selling directly to existing IVF fertility clinics in the U.S., after regaining full domestic commercialization rights to the INVOcell® upon termination of our prior agreements with Ferring. We are also seeing increased distribution activity from some of our partners in Europe and Africa.”
“We are enthusiastic about our multi-channel strategy of supporting, servicing, and expanding across existing IVF clinic networks and building new, dedicated INVO Centers,” Shum continued. “We expect this strategy to drive increased market awareness and utilization of our revolutionary technology, and provide an affordable and equally effective fertility treatment alternative to the large, underserved patient population.”
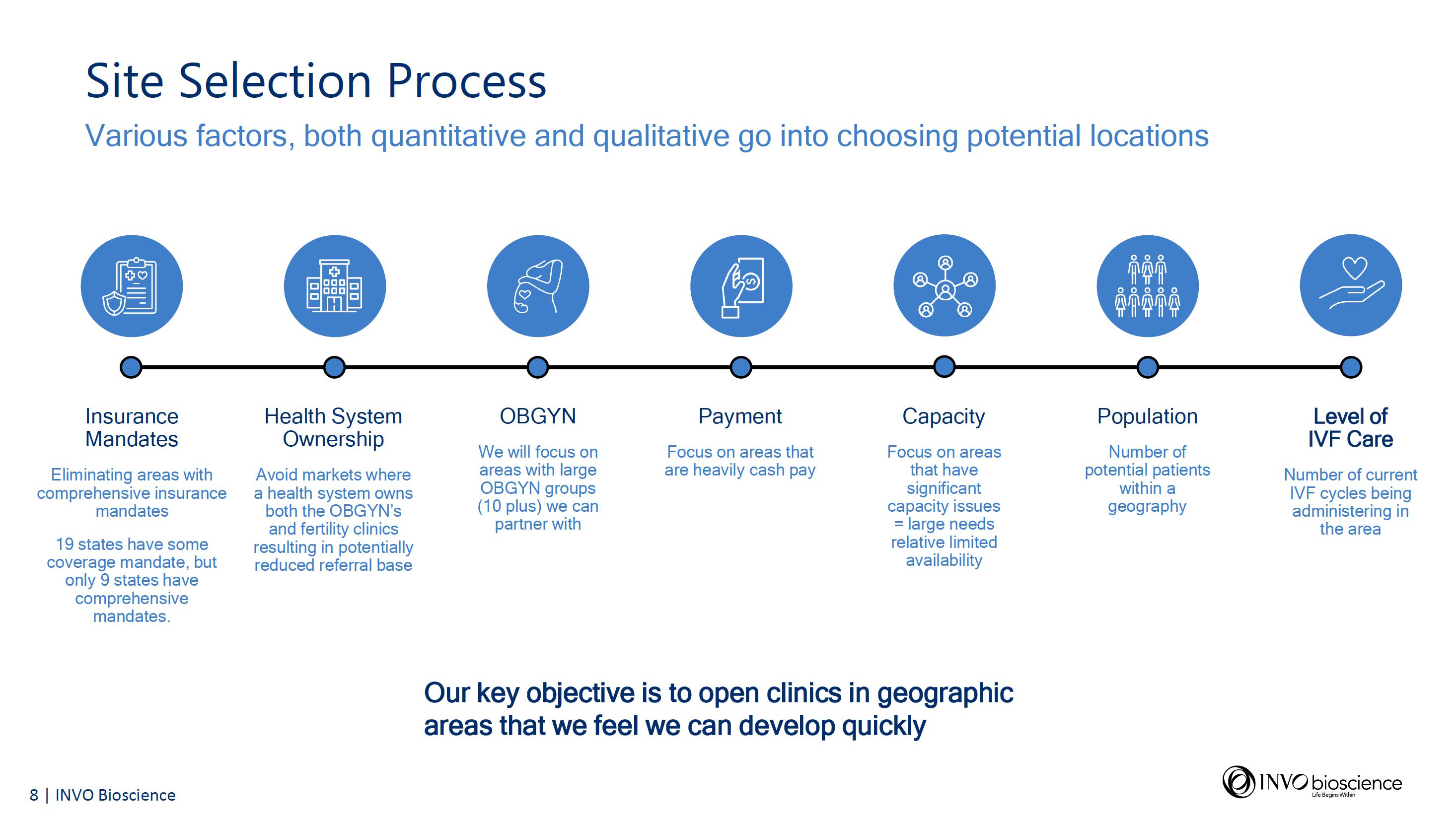
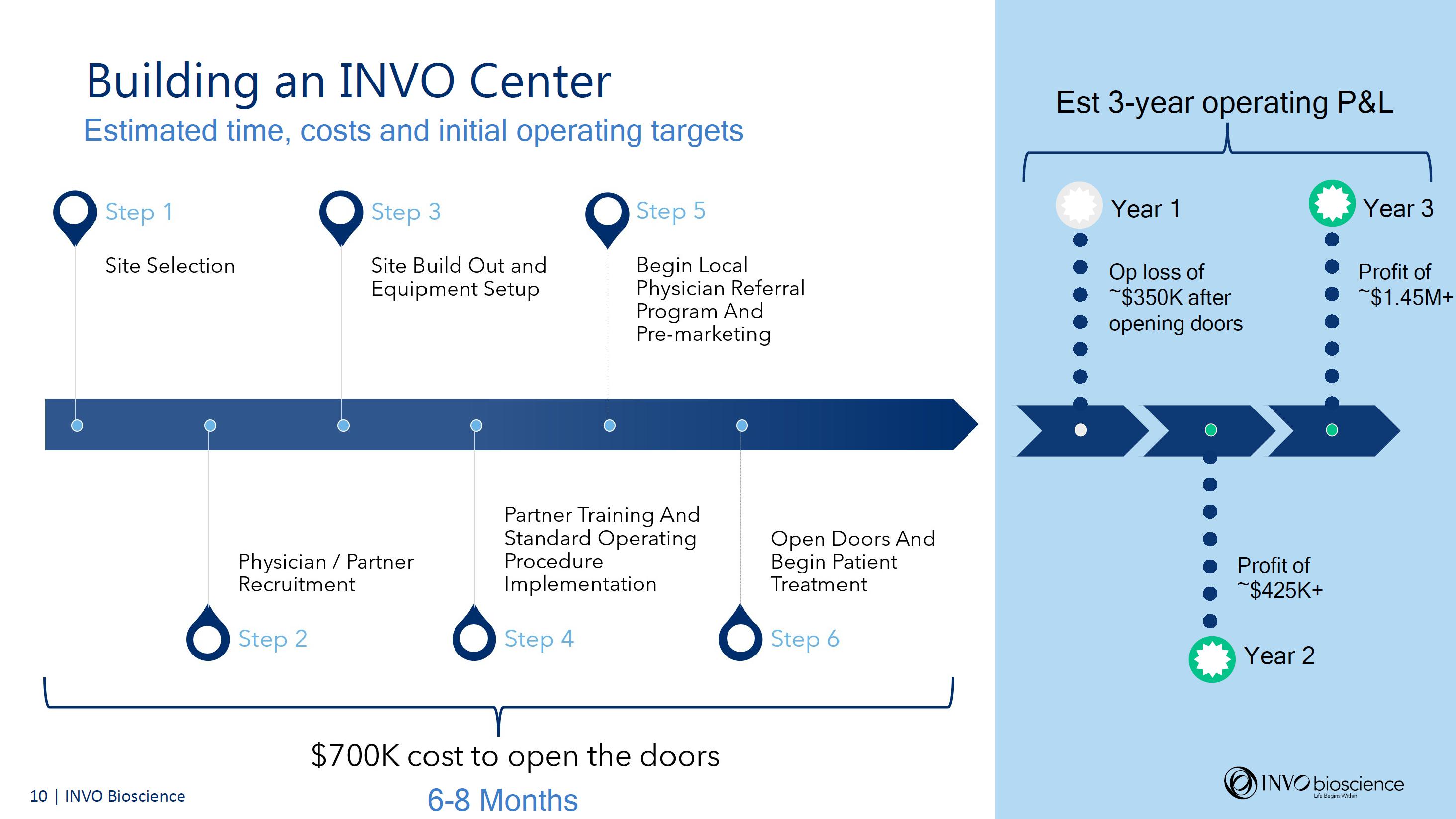
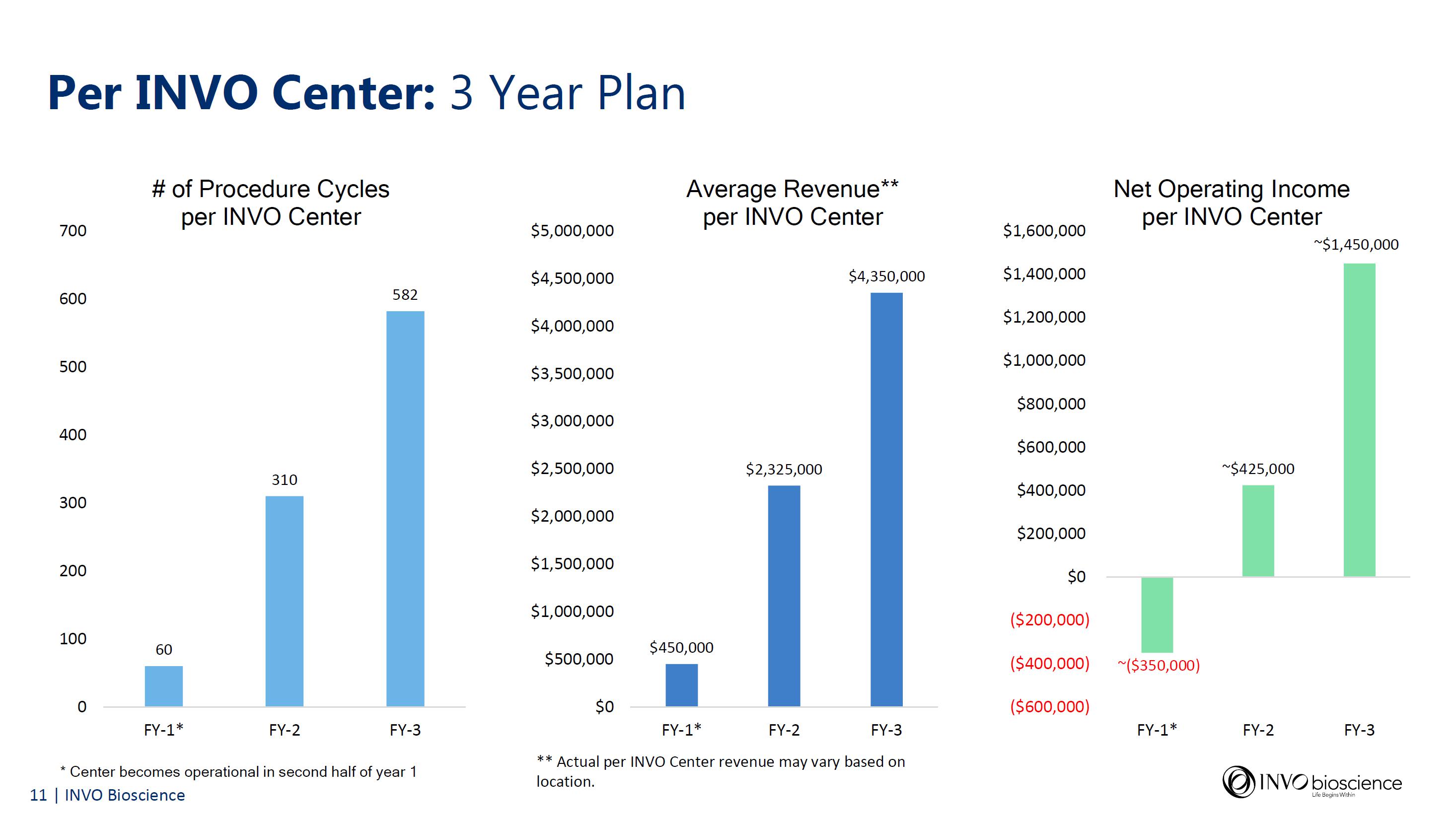
INVO Center Slide Presentation
Accompanying today’s financial results, the company has issued a slide presentation further discussing its important INVO Center model and approach which will be accessible on the Investor Relations section of the Company’s website at https://www.invobioscience.com/investors/.
Financial Results
Revenue for 2021 was $4.2 million, compared to $1.0 million in 2020. Of the $4.2 million in revenue for 2021, $2.9 million was due to the accelerated recognition of deferred revenue related to the early termination of the U.S. distribution agreement with Ferring.
Gross margin in 2021 was 97% compared to approximately 91% in 2020.
Selling, general and administrative expenses for the years ended December 31, 2021 and 2020 were $9.0 million and $6.1 million, respectively, of which $2.7 million and $1.8 million, respectively, was for non-cash, stock-based compensation expense. The increase of approximately $2.9 million, or 49%, in selling general and administrative expenses was primarily the result of approximately $0.9 million in increased non-cash, stock-based compensation, approximately $0.7 million in increased personnel expenses, approximately $0.5 million in increased expenses related to the operations of the consolidated Atlanta JV and approximately $0.4 million in legal and startup costs related to new and potential INVO Centers.
We began funding additional research and development (“R&D”) efforts in 2020 as part of our 5-day FDA label expansion efforts. R&D expenses for the years ended December 31, 2021 and 2020 were $0.2 million and $0.4 million, respectively.
Loss from equity investments for the years ended December 31, 2021 and 2020, was $0.3 million and $0, respectively. The increase in loss is due to the investment in the Alabama JV becoming operational in the third quarter of 2021. The clinic was only open for part of the period and was still in the early ramp up phase.
Interest expense and financing fees for the years ended December 31, 2021 and 2020 were $1.3 million and $2.8 million, respectively. The decrease of approximately $1.5 million, or approximately 55%, was primarily due to a decrease in amortization of discount, debt issuance cost and interest on the 2020 convertible notes.
Adjusted EBITDA (see Adjusted EBITDA Table) for the year ended December 31, 2021, was $(2.8) million, which included a $0.7 million loss attributable to our joint ventures, compared to adjusted EBITDA of $(3.7) million for the year ended December 31, 2020.
As of December 31, 2021, the Company had approximately $5.7 million in cash.
Use of Non-GAAP Measure
Adjusted EBITDA is a non-GAAP measure. This measure is not intended to be a substitute for those financial measures reported in accordance with GAAP. Adjusted EBITDA has been included because management believes that, when considered together with the GAAP figures, it provides meaningful information related to our operating performance and liquidity and can enhance an overall understanding of financial results and trends. Adjusted EBITDA may be calculated by us differently than other companies that disclose measures with the same or similar terms. See our attached financials for a reconciliation of this non-GAAP measure to the nearest GAAP measure.
Conference Call Details
INVO Bioscience has scheduled a conference call for Thursday, March 31, 2022, at 4:30 pm ET (1:30 pm PT) to review these results and recent events. Interested parties can access the conference call by dialing (833) 756-0861 or (412) 317-5751 or can listen via a live Internet webcast at https://app.webinar.net/ELRqV6y23Y9, which is also available in the Investor Relations section of the Company’s website at https://www.invobioscience.com/investors/. A teleconference replay of the call will be available through April 7, 2022, at (877) 344-7529 or (412) 317-0088, confirmation #1918743. A webcast replay will be available in the Investor Relations section of the Company’s website at https://www.invobioscience.com/investors/ for 90 days.
About INVO Bioscience
We are a commercial-stage fertility company dedicated to expanding the assisted reproductive technology (“ART”) marketplace by making fertility care accessible and inclusive to people around the world. Our flagship product is INVOcell®, a revolutionary medical device that allows fertilization and early embryo development to take place in vivo within the woman’s body. Our primary mission is to implement new medical technologies aimed at increasing the availability of affordable, high-quality, patient-centered fertility care. This treatment solution is the world’s first intravaginal culture technique for the incubation of oocytes and sperm during fertilization and early embryo development. This technique, designated as “IVC”, provides patients a more natural, intimate, and more affordable experience in comparison to other ART treatments. The IVC procedure can deliver comparable results at a fraction of the cost of traditional in vitro fertilization (“IVF”) and is a significantly more effective treatment than intrauterine insemination (“IUI”). Our commercialization strategy is focused on the opening of dedicated “INVO Centers” offering the INVOcell® and IVC procedure (with three centers in North America now operational), in addition to continuing to sell our technology solution into existing fertility clinics. For more information, please visit www.invobio.com.
Safe Harbor Statement
This release includes forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. The Company invokes the protections of the Private Securities Litigation Reform Act of 1995. All statements regarding our expected future financial position, results of operations, cash flows, financing plans, business strategies, products and services, competitive positions, growth opportunities, plans and objectives of management for future operations, as well as statements that include words such as “anticipate,” “if,” “believe,” “plan,” “estimate,” “expect,” “intend,” “may,” “could,” “should,” “will,” and other similar expressions are forward-looking statements. All forward-looking statements involve risks, uncertainties, and contingencies, many of which are beyond our control, which may cause actual results, performance, or achievements to differ materially from anticipated results, performance, or achievements. Factors that may cause actual results to differ materially from those in the forward-looking statements include those set forth in our filings at www.sec.gov. We are under no obligation to (and expressly disclaim any such obligation to) update or alter our forward-looking statements, whether as a result of new information, future events or otherwise.
INVO BIOSCIENCE, INC.
CONSOLIDATED BALANCE SHEETS
| December 31, 2021 | December 31, 2020 | |||||||
| ASSETS | ||||||||
| Current assets | ||||||||
| Cash | $ | 5,684,871 | $ | 10,097,760 | ||||
| Accounts receivable | 50,470 | 21,699 | ||||||
| Inventory | 287,773 | 265,372 | ||||||
| Prepaid expenses and other current assets | 282,751 | 157,700 | ||||||
| Total current assets | 6,305,865 | 10,542,531 | ||||||
| Property and equipment, net | 501,436 | 132,206 | ||||||
| Intangible assets, net | 132,093 | 94,963 | ||||||
| Lease right of use | 2,037,052 | 79,319 | ||||||
| Other assets | - | 240 | ||||||
| Investment in joint ventures | 1,489,934 | 98,084 | ||||||
| Total assets | $ | 10,466,380 | $ | 10,947,343 | ||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY | ||||||||
| Current liabilities | ||||||||
| Accounts payable and accrued liabilities | $ | 443,422 | $ | 328,927 | ||||
| Accrued compensation | 581,689 | 527,326 | ||||||
| Deferred revenue, current portion | 5,900 | 714,286 | ||||||
| Lease liability, current portion | 221,993 | 22,707 | ||||||
| Note payable – Payroll Protection Program | - | 157,620 | ||||||
| Convertible notes, net | - | 536,063 | ||||||
| Income taxes payable | - | 1,062 | ||||||
| Total current liabilities | 1,253,004 | 2,287,991 | ||||||
| Lease liability, net of current portion | 1,901,557 | 58,634 | ||||||
| Deferred revenue, net of current portion | - | 2,857,143 | ||||||
| Deferred tax liability | 1,139 | 469 | ||||||
| Total liabilities | 3,155,700 | 5,204,237 | ||||||
| Stockholders’ equity | ||||||||
| Common Stock, $.0001 par value; 125,000,000 shares authorized; 11,929,147 and 9,639,268 issued and outstanding as of December 31, 2021 and December 31, 2020, respectively | 1,193 | 964 | ||||||
| Additional paid-in capital | 46,200,509 | 37,978,224 | ||||||
| Accumulated deficit | (38,891,022 | ) | (32,236,082 | ) | ||||
| Total stockholders’ equity | 7,310,680 | 5,743,106 | ||||||
| Total liabilities and stockholders’ equity | $ | 10,466,380 | $ | 10,947,343 | ||||
INVO BIOSCIENCE, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS
| For the Year Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Revenue: | ||||||||
| Product revenue | $ | 544,942 | $ | 323,000 | ||||
| Clinic revenue | 43,745 | - | ||||||
| License revenue | 3,571,429 | 714,286 | ||||||
| Total revenue | $ | 4,160,116 | $ | 1,037,286 | ||||
| Cost of goods sold: | ||||||||
| Production costs | 126,326 | 79,035 | ||||||
| Depreciation | 18,726 | 9,725 | ||||||
| Total cost of goods sold | 145,052 | 88,760 | ||||||
| Gross profit | 4,015,064 | 948,526 | ||||||
| Operating expenses | ||||||||
| Selling, general and administrative | 9,015,158 | 6,065,066 | ||||||
| Research and development | 216,430 | 398,426 | ||||||
| Total operating expenses | 9,231,588 | 6,463,492 | ||||||
| Loss from operations | (5,216,524 | ) | (5,514,966 | ) | ||||
| Other income (expense): | ||||||||
| Loss from equity method joint ventures | (327,542 | ) | - | |||||
| Gain on extinguishment of debt | 159,126 | - | ||||||
| Interest income | 3,657 | 4,190 | ||||||
| Interest expense | (1,265,359 | ) | (2,836,504 | ) | ||||
| Foreign currency exchange loss | (3,534 | ) | - | |||||
| Total other expense | (1,433,652 | ) | (2,832,314 | ) | ||||
| Net loss before income taxes | (6,650,176 | ) | (8,347,280 | ) | ||||
| Income taxes | 4,764 | 36 | ||||||
| Net loss | $ | (6,654,940 | ) | $ | (8,347,316 | ) | ||
| Net loss per common share: | ||||||||
| Basic | $ | (0.63 | ) | $ | (1.52 | ) | ||
| Diluted | $ | (0.63 | ) | $ | (1.52 | ) | ||
| Weighted average number of common shares outstanding: | ||||||||
| Basic | 10,632,413 | 5,489,738 | ||||||
| Diluted | 10,632,413 | 5,489,738 | ||||||
Adjusted EBITDA
| Years Ended | ||||||||
| December 31 | ||||||||
| 2021 | 2020 | |||||||
| Net loss | $ | (6,654,940 | ) | $ | (8,347,316 | ) | ||
| Gain on extinguishment of debt | (159,126 | ) | - | |||||
| Interest expense | 80,706 | 346,458 | ||||||
| Foreign currency exchange loss | 3,534 | - | ||||||
| Stock-based compensation | 1,164,277 | 616,002 | ||||||
| Stock option expense | 1,543,912 | 1,140,989 | ||||||
| Amortization of debt discount | 1,188,310 | 2,494,236 | ||||||
| Depreciation and amortization | 27,761 | 11,917 | ||||||
| $ | (2,805,566 | ) | $ | (3,737,714 | ) | |||
| Loss from equity method JV | $ | 327,542 | $ | - | ||||
| Loss from consolidated JV (less depreciation) | 368,959 | - | ||||||
| $ | (2,109,065 | ) | $ | (3,737,714 | ) | |||
INVO Center Results
The following tables summarize the combined financial information of our consolidated and equity method joint venture INVO Centers:
| Year Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Statements of operations: | ||||||||
| Operating revenue | $ | 195,417 | $ | - | ||||
| Operating expenses* | (1,192,601 | ) | - | |||||
| Net income | $ | (997,184 | ) | $ | - | |||
| December 31, 2021 | December 31, 2020 | |||||||
| Balance sheets: | ||||||||
| Current assets | $ | 660,877 | $ | - | ||||
| Long-term assets | 2,374,471 | - | ||||||
| Current liabilities | (585,226 | ) | - | |||||
| Long-term liabilities | (743,972 | ) | - | |||||
| Net assets | $ | 1,706,150 | $ | - | ||||
*2021 operating expenses include approximately $0.5 million of one time and pre-revenue startup and labor expenses
CONTACT: INVO Bioscience, Steve Shum, CEO, 978-878-9505, sshum@invobio.com; or Investor, Lytham Partners, LLC, Robert Blum, 602-889-9700, INVO@lythampartners.com
Exhibit 99.2











