Exhibit 99.1

Oppenheimer 26th Annual Healthcare Conference
December 8, 2015 · New York, NY
DOUG GODSHALL
PRESIDENT AND CEO
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®

Safe Harbor Statement
Forward-Looking Statements
This announcement contains forward-looking statements that are based on management’s beliefs, assumptions and expectations and on information currently available to management. All statements that address operating performance, events or developments that we expect or anticipate will occur in the future are forward-looking statements, including without limitation our expectations with respect to the: commercialization of the HeartWare HVAD® System and introduction of the MVAD® System; timing, progress and outcomes of clinical trials; regulatory and quality compliance; research and development activities; consummation of our proposed acquisition of Valtech Cardio, Ltd. and our ability to take advantage of acquired and pipeline technology. Management believes that these forward-looking statements are reasonable as and when made. However, you should not place undue reliance on forward-looking statements because they speak only as of the date when made. HeartWare does not assume any obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as may be required by federal securities laws and the rules and regulations of the Securities and Exchange Commission. HeartWare may not actually achieve the plans, projections or expectations disclosed in forward-looking statements, and actual results, developments or events could differ materially from those disclosed in the forward-looking statements. Forward-looking statements are subject to a number of risks and uncertainties, including without limitation those described in Part I, Item 1A. “Risk Factors” in HeartWare’s Annual Report on Form 10-K filed with the Securities and Exchange Commission. HeartWare may update risk factors from time to time in Part II, Item 1A. “Risk Factors” in Quarterly Reports on Form 10-Q, Current Reports on Form 8-K, or other filings with the Securities and Exchange Commission.
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®
2

Additional Shareholder Information
Participants in the Solicitation
HeartWare, Valtech Cardio, Ltd. (“Valtech”) and their respective directors, executive officers, certain members of management and certain employees may be deemed to be participants in the solicitation of proxies in connection with the proposed acquisition of Valtech. A description of the interests in HeartWare of its directors and executive officers is set forth in HeartWare’s proxy statement for its 2015 Annual Meeting of Shareholders, which was filed with the Securities and Exchange Commission (the “SEC”) on April 30, 2015. Additional information regarding the persons who may, under the rules of the SEC, be deemed participants in the solicitation of proxies in connection with the proposed transaction, and a description of their direct and indirect interests in the proposed transaction, which may differ from the interests of HeartWare stockholders or Valtech shareholders generally, are set forth in a preliminary proxy statement/prospectus originally filed with the SEC on October 16, 2015 and amended on November 25, 2015.
Additional Information and Where To Find It
In connection with the proposed transactions, HW Global, Inc. (“Holdco”), has filed a Registration Statement on Form S-4 that contains a preliminary proxy statement/prospectus, which is not yet final and may be further amended. Holdco intends to file a final prospectus and other relevant materials and HeartWare intends to file a definitive proxy statement and other relevant materials with the SEC in connection with the proposed transactions. Investors and security holders of HeartWare and Valtech are urged to read these materials (when they become available) before making any voting or investment decision with respect to the transactions because they will contain important information about HeartWare, Valtech and the transactions. The proxy statement/prospectus and other relevant materials, and any other documents filed by Holdco or HeartWare with the SEC, may be obtained free of charge on the SEC website at www.sec.gov. In addition, investors and security holders may obtain free copies of these documents by directing a written request to HeartWare’s investor relations department at HeartWare International, Inc., 500 Old Connecticut Path, Framingham, MA 01701, Attention: Investor Relations.
This communication shall not constitute an offer to sell or the solicitation of an offer to sell or the solicitation of an offer to buy any securities, nor shall there be any sale of securities in any jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such jurisdiction. No offering of securities shall be made except by means of a prospectus meeting the requirements of Section 10 of the Securities Act of 1933, as amended (the “Securities Act”).
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®
3

HeartWare®
Mission
We create revolutionary technology for the treatment of heart failure to allow patients to get back to life.
hVAD®System
10,000+
Patients Implanted Globally
~125
U.S. Centers
47
Countries
~180
Intl. Centers
2009: CE Mark 2012: FDA Approval
HVAD® System - A Proven Track Record
Note: Data on Centers as of September 30, 2015
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®
4
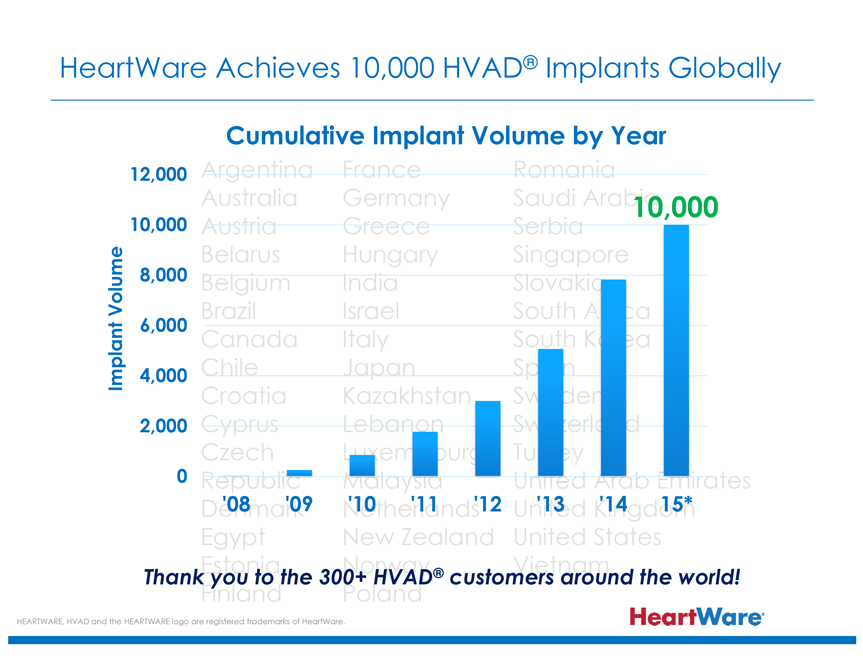
HeartWare Achieves 10,000 HVAD® Implants Globally
Cumulative Implant Volume by Year
Implant Volume
12,000
10,000
8,000
6,000
4,000
2,000
10,000
0
Argentina Australia France Germany Romania Saudi Arabia
Austria Greece Serbia
Belarus Hungary Singapore
Belgium India Slovakia
Brazil Israel South Africa
Canada Italy South Korea
Chile Japan Spain
Croatia Kazakhstan Sweden
Cyprus Lebanon Switzerland
Czech Luxembourg Turkey
Republic Malaysia United Arab Emirates
‘08 ‘09 ‘10 ‘11 ‘12 ‘13’14 15*
Denmark Netherlands United Kingdom
Egypt New Zealand United States
Estonia Norway Vietnam
Thank you to the 300+ HVAD® customers around the world!
Finland Poland
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®
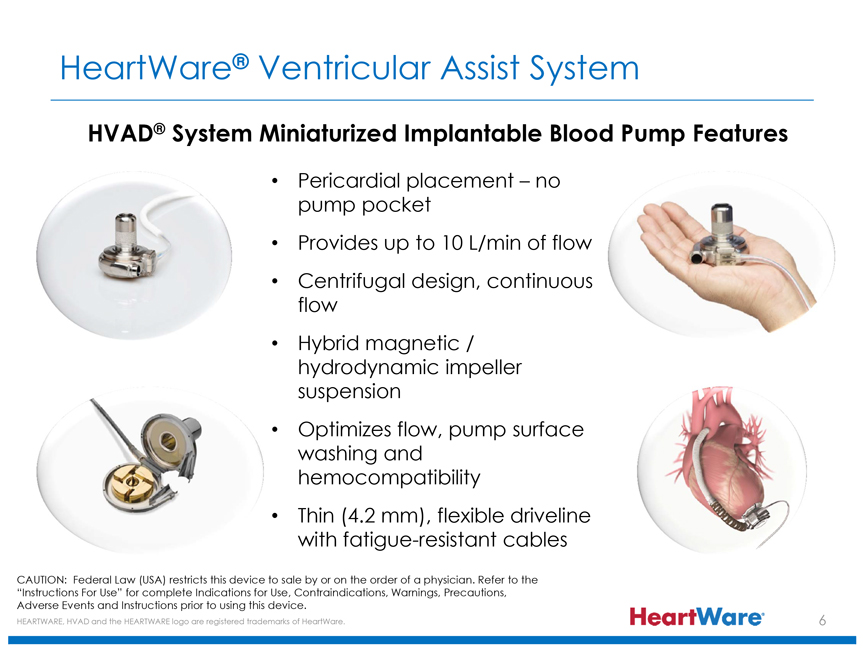
HeartWare® Ventricular Assist System
HVAD® System Miniaturized Implantable Blood Pump Features
Pericardial placement – no pump pocket
Provides up to 10 L/min of flow
Centrifugal design, continuous flow
Hybrid magnetic / hydrodynamic impeller suspension
Optimizes flow, pump surface washing and hemocompatibility
Thin (4.2 mm), flexible driveline with fatigue-resistant cables
CAUTION: Federal Law (USA) restricts this device to sale by or on the order of a physician. Refer to the
“Instructions For Use” for complete Indications for Use, Contraindications, Warnings, Precautions,
Adverse Events and Instructions prior to using this device.
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®
6

Enabling Patients to Get Back to Life
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®
7

Recent Milestones
Announced agreement to acquire Valtech Cardio
Commenced MVAD® System CE Mark international trial
Completed enrollment in ENDURANCE2 DT trial
Submitted MVAD IDE to FDA
Valtech’s Cardioband® awarded CE Mark approval
Continued ramping enrollment in HVAD® LATERAL; thoracotomy study ~60% enrolled (as of October 2015)
Surpassed 3 years of HVAD commercialization in U.S.
Longest-supported HVAD patient reached 8 years on support
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®
8

PIPELINE TECHNOLOGY
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®
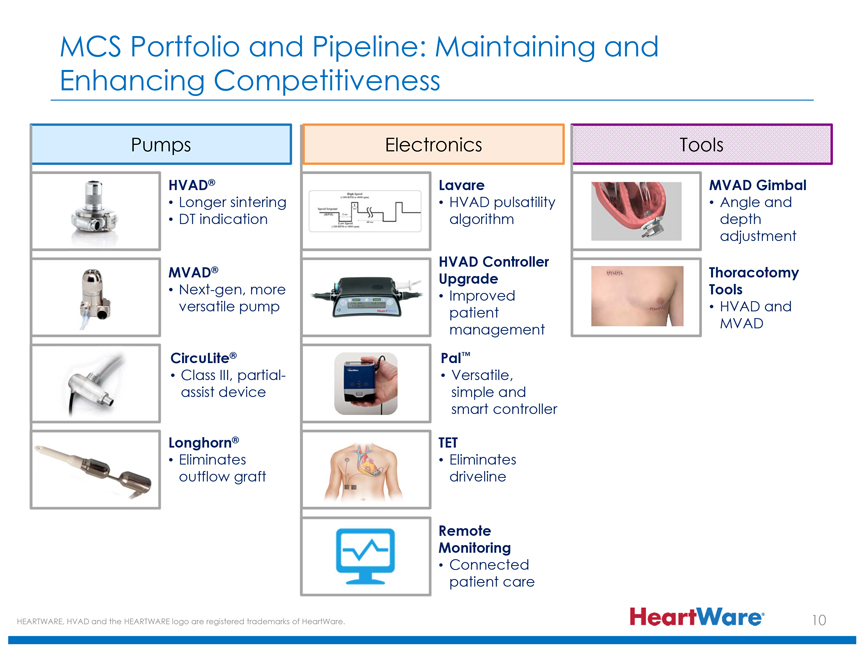
MCS Portfolio and Pipeline: Maintaining and Enhancing Competitiveness
Pumps
HVAD®
Longer sintering
DT indication
MVAD®
Next-gen, more versatile pump
CircuLite®
Class III, partial-assist device
Longhorn®
Eliminates outflow graft
Electronics
Lavare
HVAD pulsatility algorithm
HVAD Controller Upgrade
Improved patient management
Pal™
Versatile, simple and smart controller
TET
Eliminates driveline
Remote Monitoring
Connected patient care
Tools
MVAD Gimbal
Angle and depth adjustment
Thoracotomy Tools
HVAD and MVAD
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®
10

The MVAD® System Has Breakthrough Potential
MVAD® Pump
PalTM Patient Peripherals
Small size enables less invasive implant and improved anatomical fit
Advanced impeller technology for advanced hemocompatibility
Lighter and thoughtfully designed peripherals to enhance patient quality of life
Exclusively for Clinical Investigation.
Investigational device to be used by Qualified Investigators Only.
CAUTION: Investigational Device. Limited by Federal (or United States) law to investigational use.
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®
11
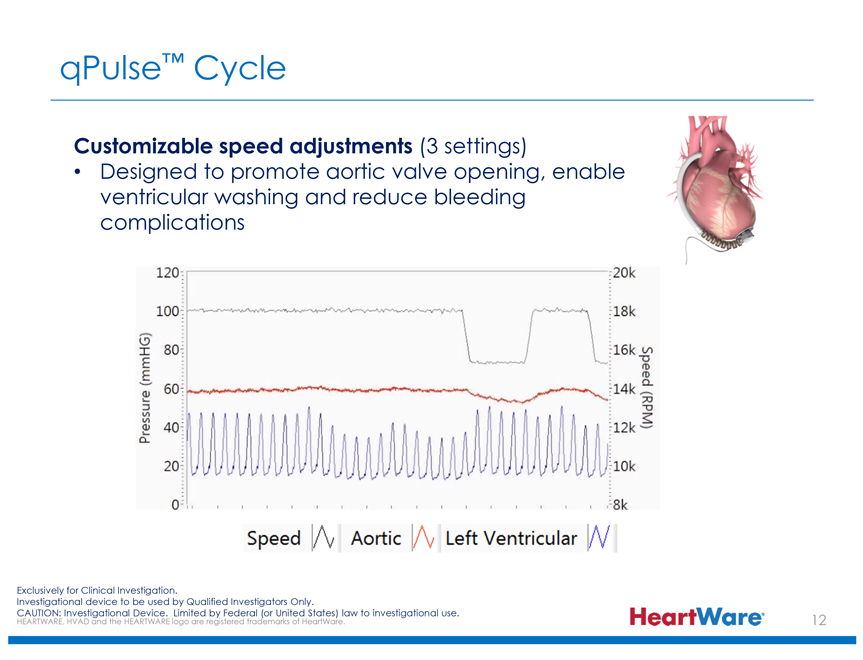
qPulse™ Cycle
Customizable speed adjustments (3 settings)
Designed to promote aortic valve opening, enable ventricular washing and reduce bleeding complications
Pressure (mmHG)
120 100 80 60 40 20 0
20k 18k 16k 14k 12k 10k 8k
Speed (RPM)
Speed Aortic Left Ventricular
Exclusively for Clinical Investigation.
Investigational device to be used by Qualified Investigators Only.
CAUTION: Investigational Device. Limited by Federal (or United States) law to investigational use.
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®
12
MVAD® Advantage™ CE Mark Trial
Multi-center, prospective, non-randomized, single-arm trial
70 patients at 11 sites in Australia, Austria, France, Germany & the UK
Primary Endpoint: Survival at 6 months
Implantation via thoracotomy or sternotomy
First patient enrolled July 2015
mVAD AdvantageTM Trial
Exclusively for Clinical Investigation.
Investigational device to be used by Qualified Investigators Only.
CAUTION: Investigational Device. Limited by Federal (or United States) law to investigational use.
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®
13
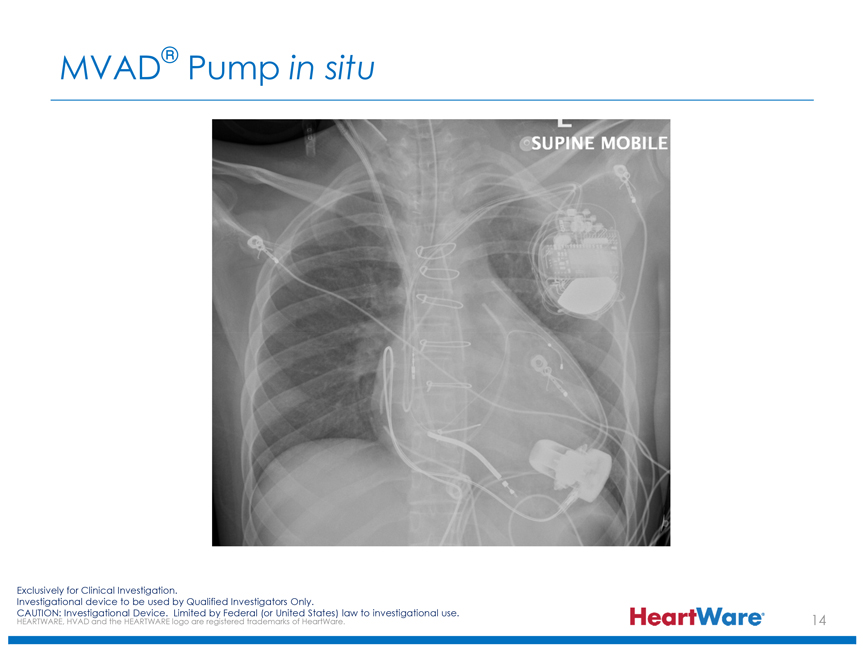
MVAD® Pump in situ
SUPINE MOBILE
Exclusively for Clinical Investigation.
Investigational device to be used by Qualified Investigators Only.
CAUTION: Investigational Device. Limited by Federal (or United States) law to investigational use.
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®
14

MVAD® System Commentary
Controller assembly fix complete, software patch progressing toward submission efficiently
Investigation continues with no anticipated design modifications
Narrowing our focus to specific areas within our manufacturing process, which we may elect to further tighten up
Working with investigators to review status and develop restart plan
Finalizing decision tree for return to the clinic
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®
15

BUSINESS COMBINATION
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare

HeartWare + Valtech: Creating the Technology Leader in Heart Failure
MVAD® System will lead a transformation of MCS portfolio and VAD market, picking up where HVAD® leaves off
Synergies with disease, customer, referral channel and delivery system will create compounding benefits over next decade
Adjustable repair of MR and TR expected to emerge as first-line option, whether surgical or interventional
Nearer-term commercial opportunity in established surgical and interventional mitral repair market with >$450M in sales
Sophisticated CardioValve™ design and delivery system to enable HeartWare leadership in mitral replacement
Combined pipeline creates unique opportunity for differentiated value creation
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®
17

Covering the Spectrum of Heart Failure
Heart Failure Continuum
Today
Cardioband Mitral HVAD
Tomorrow
Cardinal Mitral and Tricuspid
Cardioband Mitral and Tricuspid
MVAD HVAD
Future
Cardinal Mitral and Tricuspid
Cardioband Mitral and Tricuspid
Cardiovalve
CircuLite
MVAD
HVAD
Longhorn
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare® 18

Valtech: Consistent, Meaningful Milestone Cadence
2016 2017 2018 2019
Cardioband Launch
Cardioband IDE
Cardinal Launch EU
Cardioband TR FIM
Cardiovalve FIM
Cardioband TR IDE
Cardinal IDE
Cardioband PMA
Cardioband TR CE Mark
Cardinal PMA
Cardiovalve IDE
Cardiovalve CE
Cardioband TR PMA
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare® 19

Expanding Cardioband® Commercial Footprint
Cardioband® Center Locations in Europe & Israel
FRANCE
Paris – Hôpital Bichat-Claude Bernard
GERMANY
Bad Nauheim – Kerckhoff-Klinik
Berlin – Vivantes Klinikum Am Urban
Bonn – Universitätsklinikum Bonn
Frankfurt – CardioVasculäres Centrum Frankfurt
Hamburg – Asklepios Klinik St. Georg
Hamburg – HGH Universitäres Herz- und
Gefäßzentrum Hamburg GmbH
Hamburg – Universitätsklinikum Hamburg-Eppendorf (UKE)
Köln – Universität zu Köln
Mainz – Universitätsmedizin der Johannes Gutenberg
Universität Mainz
München – Ludwig Maximilians Universität (LMU) Klinikum
Rostock – Universitätsmedizin Rostock
ISRAEL
Haifa – Rambam Health Care Campus
Petah Tikva – Rabin Medical Center
ITALY
Catania – Ospedale Ferrarotto
Milan – Ospedale San Raffaele (OSR)
Rome – Policlinico Tor Vergata
NETHERLANDS
Nieuwegein – St. Antonius Ziekenhuis
SWITZERLAND
Zurich – UniversitätsSpital Zürich
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
Valtech HeartWare®
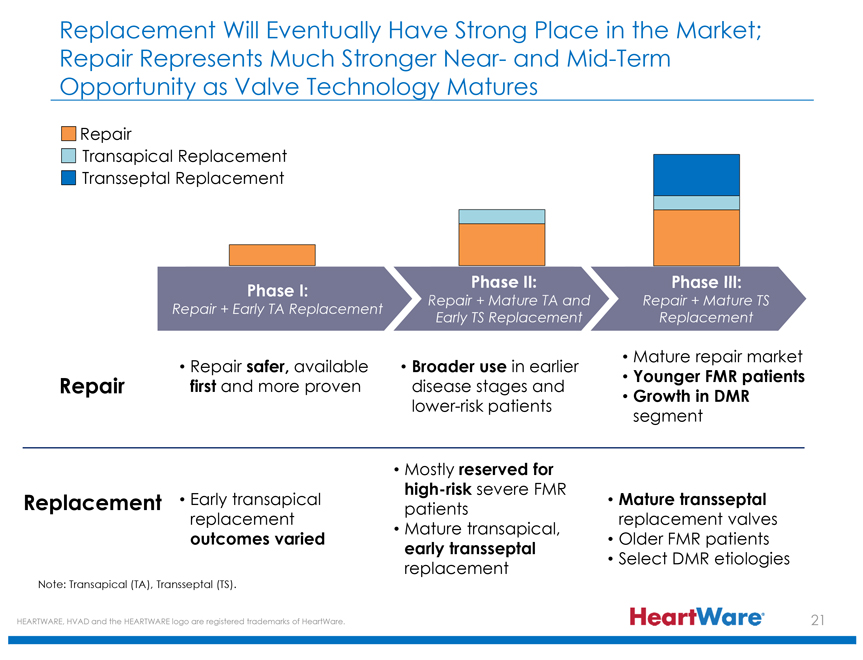
Replacement Will Eventually Have Strong Place in the Market; Repair Represents Much Stronger Near- and Mid-Term Opportunity as Valve Technology Matures
Repair
Transapical Replacement
Transseptal Replacement
Phase I: Repair + Early TA Replacement
Phase II: Repair + Mature TA and Early TS Replacement
Phase III: Repair + Mature TS Replacement
Repair Repair safer, available first and more proven
Broader use in earlier disease stages and lower-risk patients
Mature repair market
Younger FMR patients
Growth in DMR segment
Replacement
Early transapical replacement outcomes varied
Mostly reserved for high-risk severe FMR patients
Mature transapical, early transseptal replacement
Mature transseptal, replacement valves
Older FMR patients
Select DMR etiologies
Note: Transapical (TA), Transseptal (TS).
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare® 21
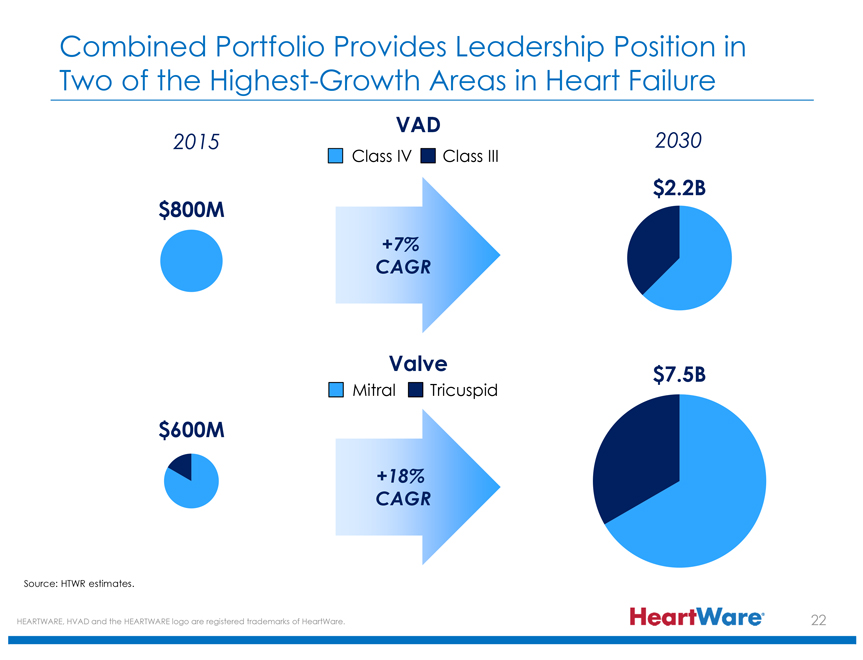
Combined Portfolio Provides Leadership Position in Two of the Highest-Growth Areas in Heart Failure
2015 VAD Class IV Class III 2030
$800M +7% CAGR $2.2B
Valve Mitral Tricuspid
$600M +18% CAGR $7.5B
Source: HTWR estimates.
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare® 22

Video and Slide Replay Available…
HeartWare®
HeartWare® International Analyst and Investor Meeting
Thursday, November 05, 2015 9:00 am EST
Webcast
00:33:19/03:47:33
Slide Search Slides Speakers Downloads
FMR / FTR Summary
Advanced CHF is a growing problem with significant mortality and repeat hospitalization
Most patients with CHF have FMR and in very advanced stages FTR
GDMT and resynchronization therapy work
Surgery is seldom used
Transcatheter therapy and MCS are major opportunities
A vertically integrated device company capable of managing different stages of CHF disease state makes total sense
Valtech HeartWare® 70
Slide 69 of 286
For more information and to view the Analyst & Investor Meeting video webcast replay, please visit HeartWare.com
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare® 23

Priorities for the Months Ahead…
Advance discussions with shareholders regarding proposed Valtech acquisition; facilitate diligence process
Complete MVAD® review and implement any appropriate actions identified
Resume enrollment in CE Mark clinical trial of MVAD System
Pursue initiation of MVAD System clinical trials in U.S. and Canada
Complete Warning Letter remediation process
Complete follow-up for ENDURANCE2 DT trial
Secure approval in U.S. for Lavare Cycle
Complete enrollment in HVAD® LATERAL thoracotomy IDE study
Advance CircuLite® System toward clinic
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare® 24

Thank You
HEARTWARE, HVAD and the HEARTWARE logo are registered trademarks of HeartWare.
HeartWare®