8-K
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, DC 20549
FORM 8-K
CURRENT REPORT
Pursuant
to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): July 14, 2015
ZOGENIX, INC.
(Exact
Name of Registrant as Specified in its Charter)
|
|
|
|
|
| Delaware |
|
001-34962 |
|
20-5300780 |
| (State or Other Jurisdiction
of Incorporation) |
|
(Commission
File Number) |
|
(IRS Employer
Identification No.) |
|
|
| 12400 High Bluff Drive, Suite 650, San Diego, CA |
|
92130 |
| (Address of Principal Executive Offices) |
|
(Zip Code) |
Registrant’s telephone number, including area code: (858) 259-1165
(Former Name or Former Address, if Changed Since Last Report.)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the
following provisions (see General Instruction A.2. below):
| ¨ |
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ¨ |
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ¨ |
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ¨ |
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
| Item 7.01. |
Regulation FD Disclosure. |
On July 14, 2015, Zogenix, Inc. ( “Zogenix”) plans to host a
meeting featuring presentations by members of Zogenix’s management team and key opinion leaders in the field related to Dravet syndrome. The presentations will include the slides attached as Exhibit 99.1 to this Current Report on Form 8-K,
which is incorporated herein by reference.
The information in this Item 7.01, including Exhibit 99.1, is being furnished pursuant to Item 7.01
and shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended, or the Exchange Act, or otherwise subject to the liabilities of that section, and it shall not be deemed incorporated by
reference in any filing under the Securities Act of 1933, as amended, or under the Exchange Act, whether made before or after the date hereof, except as expressly set forth by specific reference in such filing to this item of this report.
Based on a recent meeting with the U.S. Food and Drug Administration (the
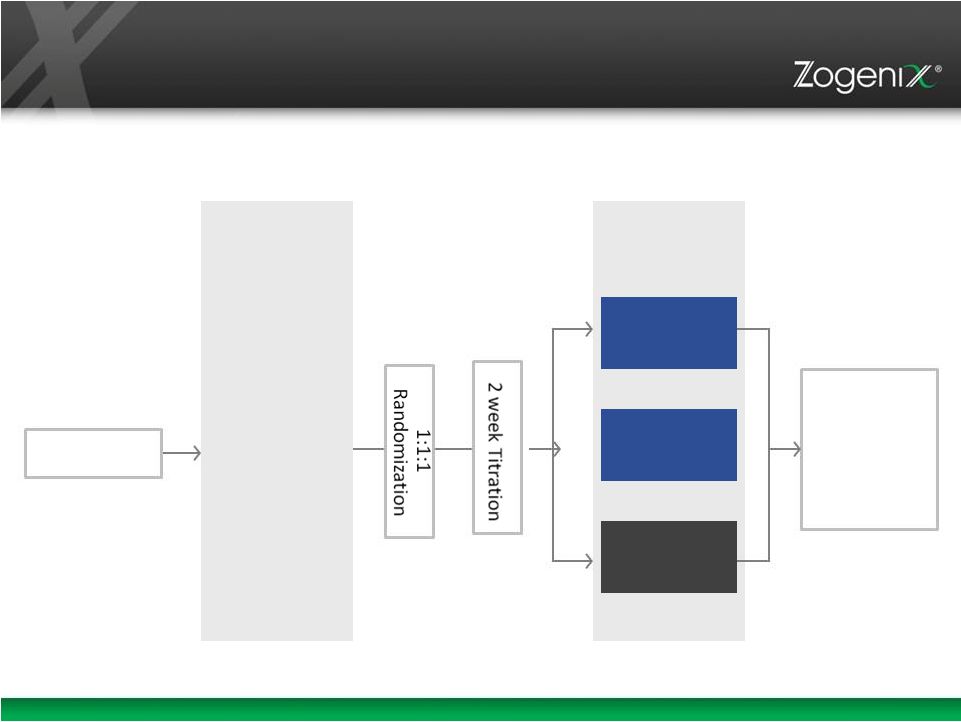
“FDA”), Zogenix has revised its clinical trial plans for ZX008, Zogenix’s investigational proprietary pediatric formulation of fenfluramine for the treatment of Dravet syndrome. Following FDA guidance, Zogenix will seek to enroll 105
patients in each of the two planned double-blind, randomized, controlled Phase 3 clinical trials for ZX008, and the baseline observation period will be eight weeks, followed by a two week titration period. Zogenix also plans to conduct an optional,
long-term, open label extension study. Zogenix currently expects to submit an investigational new drug (“IND”) application to initiate Phase 3 clinical trials for ZX008 to the FDA in August 2015, with Phase 3 clinical trials beginning in
the fourth quarter of 2015.
* * *
Zogenix
cautions you that statements included in this report that are not a description of historical facts are forward-looking statements. Words such as “plans,” “believes,” “expects,” “anticipates,” and
“will,” and similar expressions, are intended to identify forward-looking statements, and are based on Zogenix’s current beliefs and expectations. Such statements include, without limitation, statements regarding Zogenix’s
clinical development plans for ZX008; the timing of an IND submission for ZX008; and the commencement of related Phase 3 clinical trials for ZX008. You are cautioned not to place undue reliance on these forward-looking statements, which speak only
as of the date hereof. Zogenix’s actual future results may differ materially from its current expectations due to the risks and uncertainties inherent in its business. These risks include, but are not limited to: the inherent risks of clinical
development of ZX008, including potential delays in enrollment and completion of clinical trials, and Zogenix’s dependence on third parties in such development; risks and uncertainties associated with regulatory review and potential additional
requests or requirements from the FDA; other difficulties or delays relating to the development, testing, manufacturing and marketing of and obtaining regulatory approval for ZX008; and other risks detailed under “Risk Factors” and
elsewhere in Zogenix’s periodic reports and other filings made with the Securities and Exchange Commission from time to time. All forward-looking statements are qualified in their entirety by this cautionary statement, which is made under the
safe harbor provisions of Section 21E of the Private Securities Litigation Reform Act of 1995, and Zogenix undertakes no obligation to revise or update this report to reflect events or circumstances after the date hereof.
| Item 9.01. |
Financial Statements and Exhibits. |
(d) Exhibits.
|
|
|
| Exhibit
No. |
|
Description |
|
|
| 99.1 |
|
Slide Presentation |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned
hereunto duly authorized.
|
|
|
|
|
|
|
|
|
|
|
ZOGENIX, INC. |
|
|
|
|
| Date: July 14, 2015 |
|
|
|
By: |
|
/s/ Ann D. Rhoads |
|
|
|
|
Name: |
|
Ann D. Rhoads |
|
|
|
|
Title: |
|
Executive Vice President, Chief Financial Officer, Treasurer and Secretary |
EXHIBIT INDEX
|
|
|
| Exhibit
No. |
|
Description |
|
|
| 99.1 |
|
Slide Presentation |