Exhibit 99.1

Intrexon Announces Third Quarter 2017 Financial Results
– Quarterly GAAP revenues of $46.0 million and net loss attributable to Intrexon of $39.7 million
including non-cash charges of $24.0 million –
– Adjusted EBITDA of $(16.4) million –
GERMANTOWN, MD, November 9, 2017 – Intrexon Corporation (NYSE: XON), a leader in the engineering and industrialization of biology to improve the quality of life and health of the planet, today announced its third quarter financial results for 2017.
Business Highlights and Recent Developments:
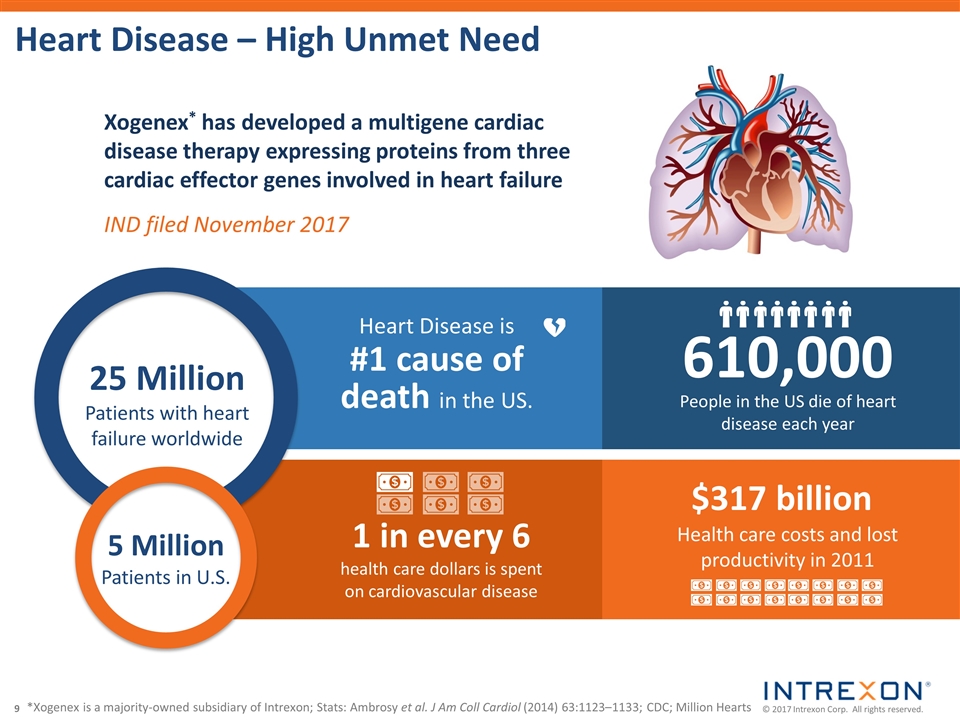
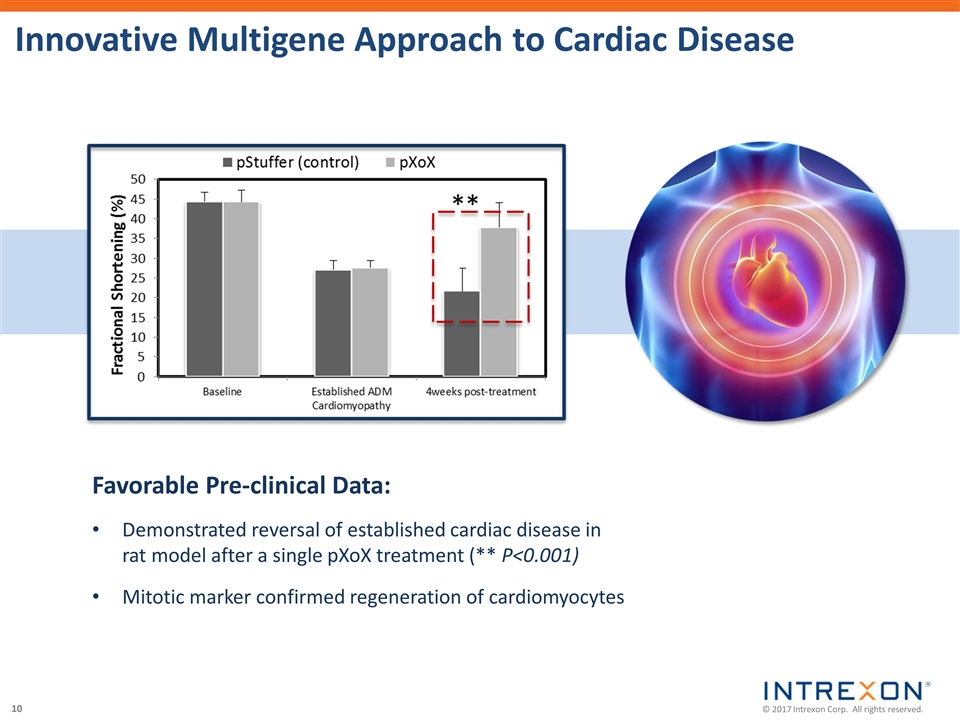
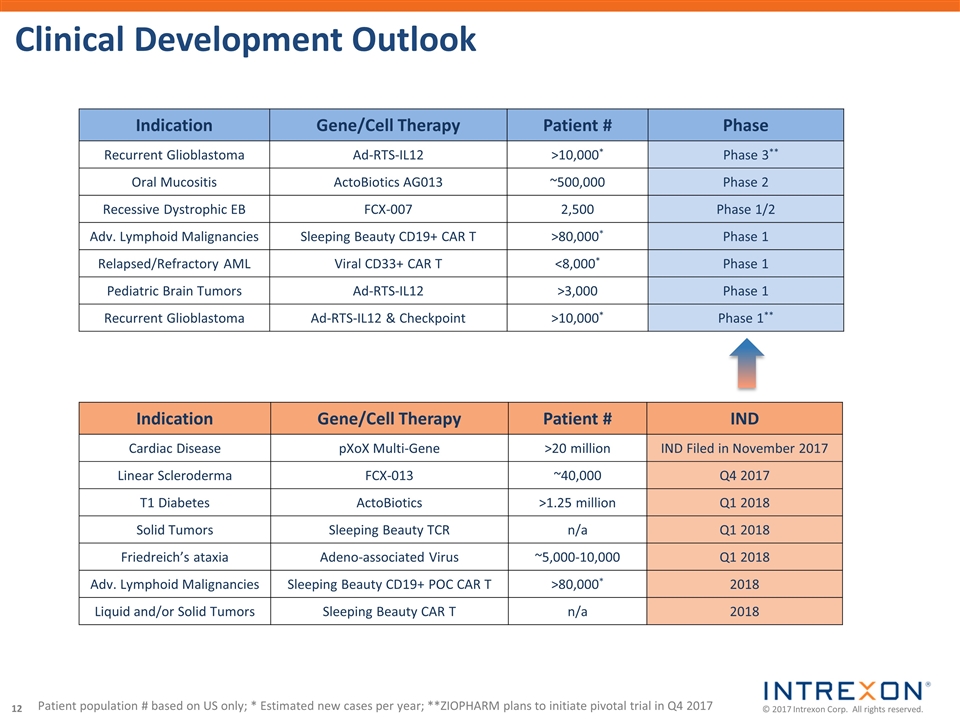
| • | Xogenex, a majority-owned subsidiary of Intrexon, filed an Investigational New Drug application with the U.S. Food and Drug Administration for a Phase 1 trial of the gene therapy INXN-4001, the world’s first multigene therapeutic candidate expressing proteins from three cardiac effector genes for the treatment of heart disease; |
| • | Intrexon’s proprietary methanotroph bioconversion platform continued to increase yield across multiple products including 2,3 butanediol, which increased approximately 15% during the quarter, and isobutanol, which increased 78%; |
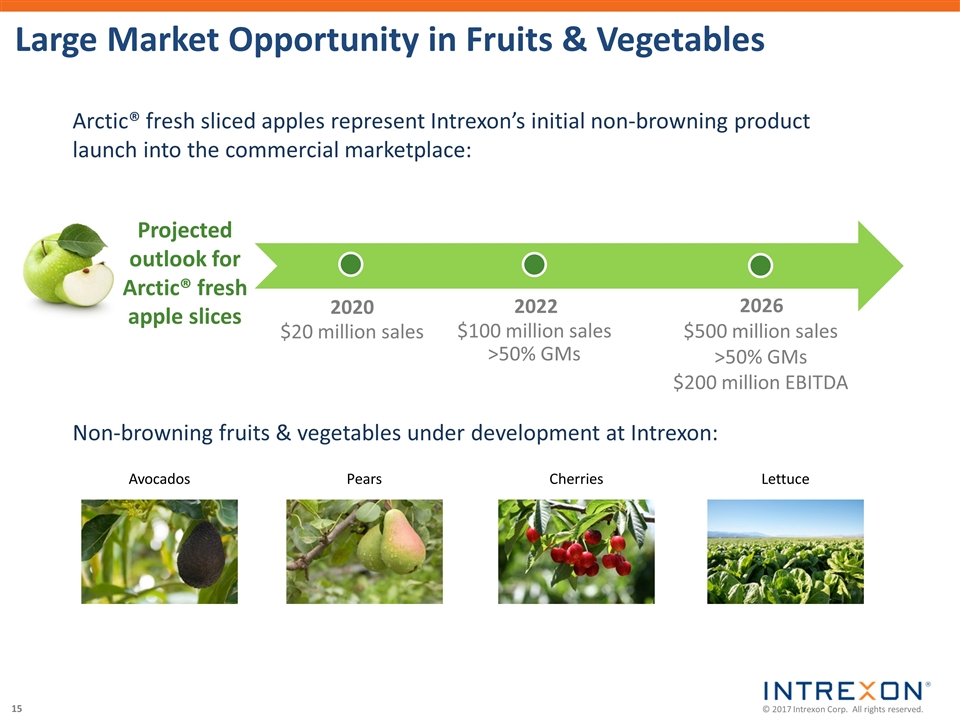
| • | Okanagan Specialty Fruits, a wholly-owned subsidiary of Intrexon, recently initiated the first commercial launch of its non-browning Arctic® Golden fresh sliced apples in stores across the mid-West and other regions; |
| • | Collaborator ZIOPHARM Oncology, Inc. (Nasdaq: ZIOP) announced that the first patient has been dosed in a new Phase 1 study of its gene therapy Ad-RTS-hIL-12 + veledimex for the treatment of pediatric brain tumors; |
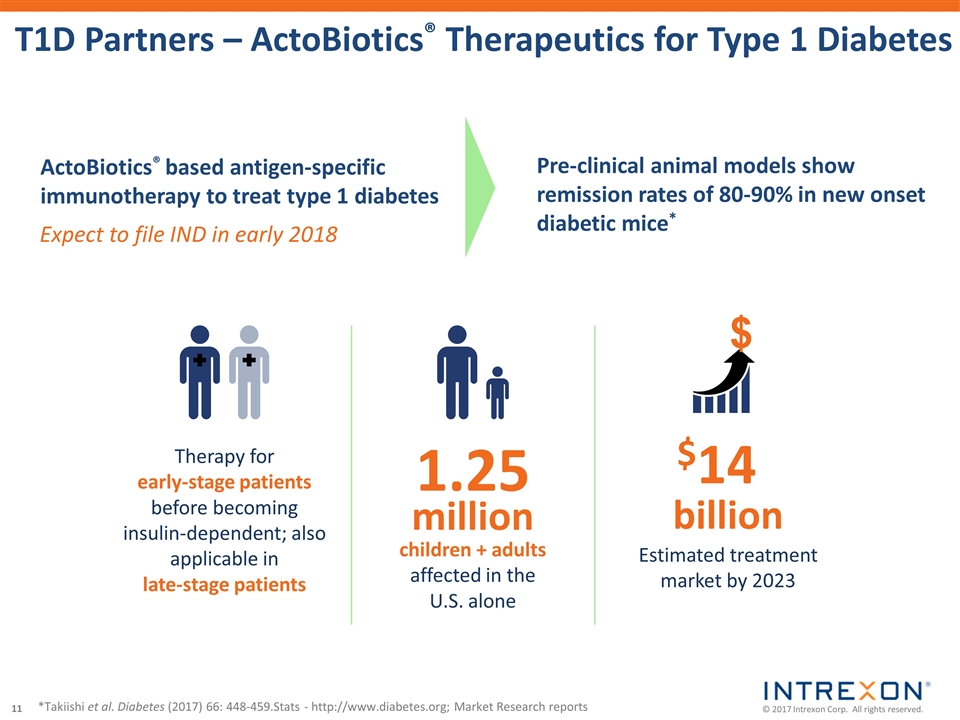
| • | Collaborator Oragenics, Inc. (NYSE MKT: OGEN) dosed the first patient in its Phase 2 clinical trial of AG013, an ActoBiotics® therapeutic candidate, for the treatment of oral mucositis; |
| • | Collaborator Fibrocell Science, Inc. (NASDAQ: FCSC) reported interim results in its Phase 1/2 clinical trial of its gene therapy FCX-007 for the treatment of recessive dystrophic epidermolysis bullosa with encouraging safety and positive early trends noted in wound healing and pharmacology signals; |
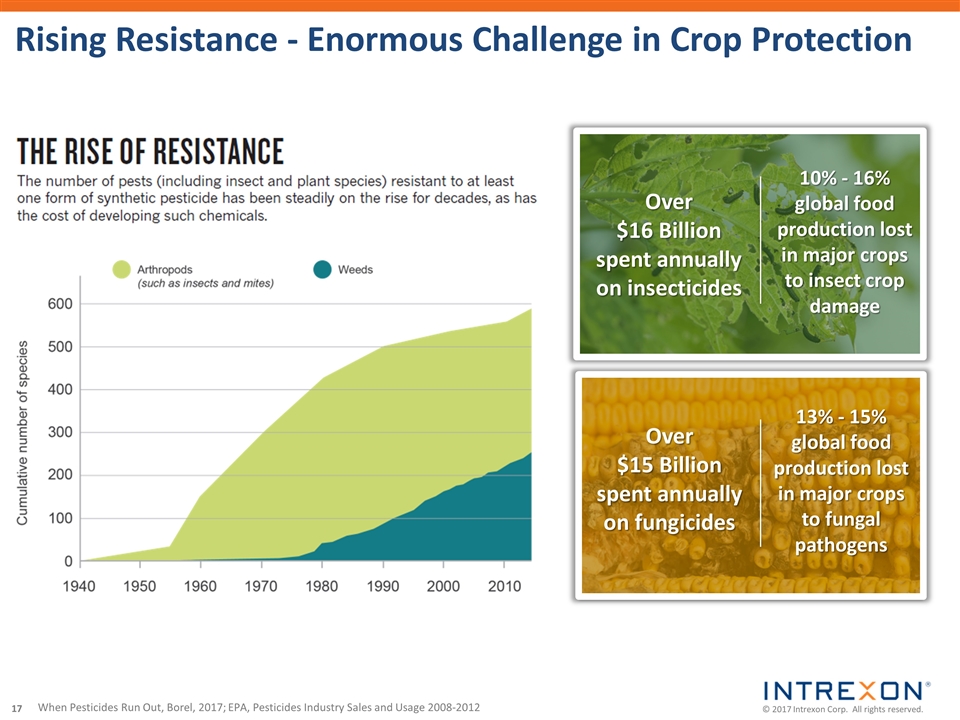
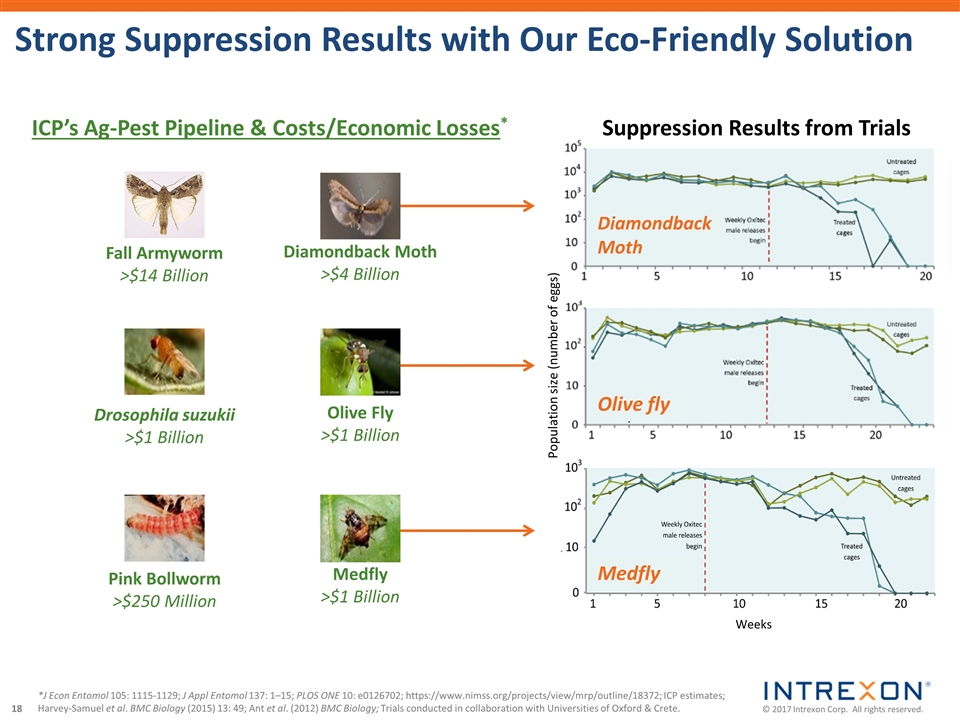
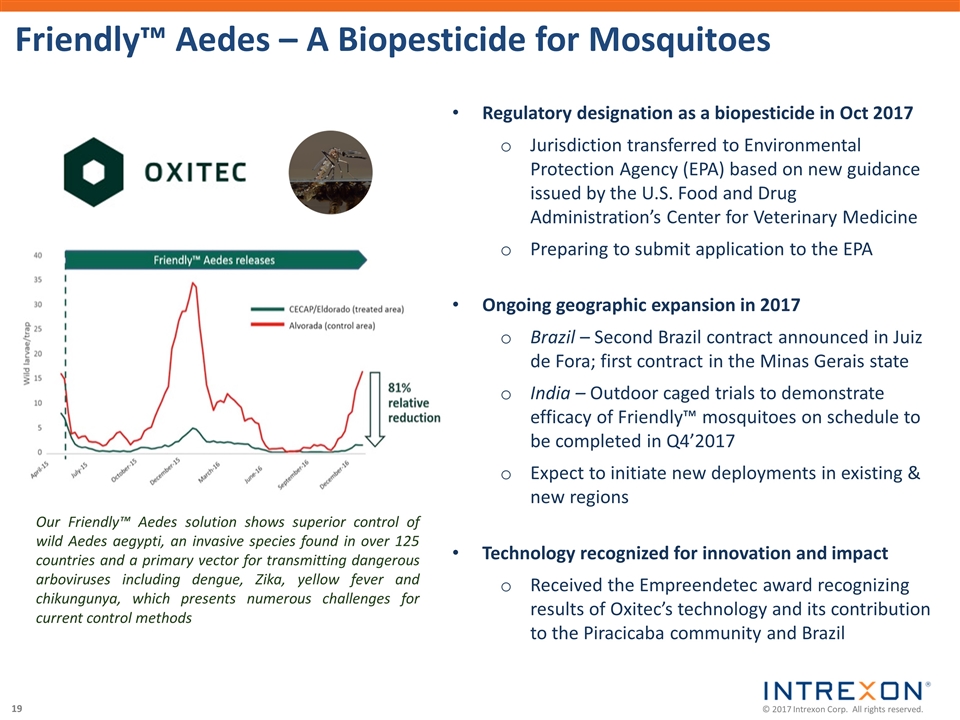
| • | Intrexon Crop Protection announced the achievement of a key milestone in its collaboration with a leading agricultural company on the development of an eco-friendly fall armyworm solution utilizing Oxitec’s self-limiting technology, the receipt of a milestone payment, and the continued advancement of the program. Native to the Americas, fall armyworm invaded Africa in 2016 and has rapidly spread to at least 28 countries causing an estimated $13.8 billion in losses of maize, sorghum, rice and sugarcane; |
| • | Oxitec’s innovative solution to suppress the diamondback moth (DBM), a major pest of brassica crops that costs farmers over $4 billion yearly in crop losses and control management, began field trials in the U.S. following the Finding of No Significant Impact issued by the U.S. Department of Agriculture. DBM is considered one of the most difficult pests to control because it has become resistant to dozens of chemical insecticides and has also evolved resistance in the field to Bacillus thuringiensis (Bt) proteins; |
| • | Intrexon announced a collaboration with Arch Pharmalabs, Ltd. to develop microbial strains for fermentation-based production of an active pharmaceutical ingredient currently sourced from animals; and |
| • | Intrexon entered into a Preferred Stock Equity Facility with an affiliate of Third Security, LLC, a venture capital firm founded by Randal J. Kirk, Intrexon’s Chairman and Chief Executive Officer, under which Intrexon may, from time to time at its discretion, sell to the investor up to $100 million of newly issued Series A Redeemable Preferred Stock. |
Third Quarter 2017 Financial Highlights:
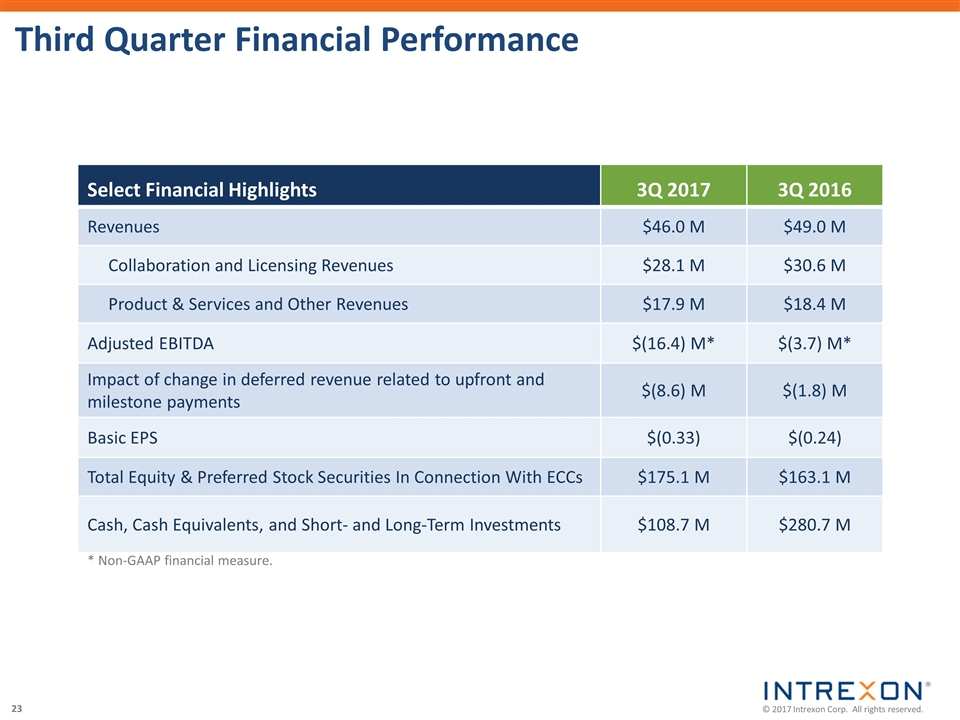
| • | Total revenues of $46.0 million, a decrease of 6% from the third quarter of 2016; |
| • | Net loss of $39.7 million attributable to Intrexon, or $(0.33) per basic share, including non-cash charges of $24.0 million; |
| • | Adjusted EBITDA of $(16.4) million, or $(0.14) per basic share; |
| • | The net change in deferred revenue related to upfront and milestone payments, which represents the cash and stock received from collaborators less the amount of revenue recognized during the period, was a decrease of $8.6 million compared to a decrease of $1.8 million in the third quarter of 2016; and |
| • | Cash, cash equivalents, and short-term investments totaled $108.7 million, the value of preferred shares totaled $148.5 million, and the value of common equity securities totaled $26.6 million at September 30, 2017. |
Year-to-Date 2017 Financial Highlights:
| • | Total revenues of $154.0 million, an increase of 6% over the nine months ended September 30, 2016; |
| • | Net loss of $89.8 million attributable to Intrexon, or $(0.75) per basic share, including non-cash charges of $66.0 million; |
| • | Adjusted EBITDA of $(25.5) million, or $(0.21) per basic share; and |
| • | The net change in deferred revenue related to upfront and milestone payments, which represents the cash and stock received from collaborators less the amount of revenue recognized during the period, was a decrease of $28.2 million compared to a net increase of $127.8 million in the nine months ended September 30, 2016. |
“I am proud of our team for its astonishing number of significant accomplishments. We continue our transition from a company with substantial potential to one that is realizing that promise scientifically and commercially,” commented Randal J. Kirk, Chairman and Chief Executive Officer of Intrexon. “Four years ago, we were focused almost exclusively on early stage programs but now are engaged in partnering efforts to capitalize on certain of our mature programs, including Methane Bioconversion Platform and Intrexon Crop Protection. We believe that these efforts will allow us to increase our investments in ground-breaking new programs while realizing, sometimes with partners, the available potential of our earlier work.
“Today, for example, with earlier-developed therapeutic candidates moving into Phase 3 and Phase 2 clinical trials, we mark a historic technical achievement in the fight against heart failure with the filing of an IND for the world’s first multigenic gene therapy targeting the leading cause of human death. This candidate generated promising safety and efficacy data in large animal models, and we are hopeful that it will greatly improve the prospects for the many patients — 5.7M adults in the U.S. alone — with this grim diagnosis. We shall be introducing additional complex, multigenic therapies into the clinic, both through our partners and on behalf of Precigen, in the near future.
“We see a similar pattern in our Energy and Food portfolios and look forward to developing this in Environment and Consumer as well and so believe that the balance of this year and 2018 will be validating for our strategy and ambition,” concluded Mr. Kirk.
Third Quarter 2017 Financial Results Compared to Prior Year Period
Total revenues decreased $3.0 million, or 6%, from the quarter ended September 30, 2016. Collaboration and licensing revenues decreased $2.4 million from the quarter ended September 30, 2016 due to a decrease in research and development services for certain of the Company’s collaborations as the Company temporarily redeployed certain resources towards supporting prospective new platforms and partnering opportunities. Product revenues decreased $1.6 million, or 17%, primarily due to lower customer demand for cows and live calves. Gross margin on products also decreased in the current period primarily due to customer demand. Service revenues increased $1.3 million, or 15%, due to an increase in the number of bovine in vitro fertilization cycles performed due to higher customer demand. Gross margin on these services was consistent period over period.
Research and development expenses increased $7.4 million, or 26%, due primarily to increases in (i) salaries, benefits and other personnel costs for research and development employees, (ii) depreciation and amortization, (iii) rent and utilities expenses, and (iv) lab supplies and consulting expenses. Salaries, benefits and other personnel costs increased $2.6 million due to an increase in research and development headcount necessary to invest in current or expanding platforms and to develop new prospective collaborations and other partnering opportunities. Depreciation and amortization increased $2.0 million primarily as a result of (i) the amortization of developed technology acquired from Oxitec, which began in November 2016 upon the completion of certain operational and regulatory events, and (ii) the amortization of developed technology acquired from GenVec in June 2017. Rent and utilities expenses increased $1.4 million primarily due to the expansion of certain facilities to support the Company’s increased headcount. Lab supplies and consulting expenses increased $1.1 million as a result of (i) the progression of certain programs into the preclinical and clinical phases with certain of Intrexon’s collaborators and (ii) the expansion or improvement of certain of the Company’s platform technologies. Selling, general and administrative (SG&A) expenses increased $5.5 million, or 16%. Salaries, benefits and other personnel costs increased $2.7 million primarily due to (i) increased headcount to support the Company’s expanding operations and (ii) increased stock-based compensation expense resulting from grants to certain of the Company’s officers in February 2017. Legal and professional fees increased $0.9 million primarily due to (i) increased legal fees to defend ongoing litigation and (ii) increased fees incurred for business development and prospective partnering efforts.
Total other income, net, increased $1.4 million, or 31%. This increase was primarily attributable to (i) increases in fair market value of the Company’s equity securities portfolio and (ii) dividend income from the Company’s investments in preferred stock.
Equity in net loss of affiliates, which includes the Company’s pro-rata share of the net losses of its investments accounted for using the equity method of accounting, decreased $3.3 million, or 52%. This decrease was primarily due to the temporary redeployment of certain of the Company’s resources away from these joint venture programs towards supporting prospective new platforms and additional collaborations.
Year-to-Date 2017 Financial Results Compared to Prior Year Period
Total revenues increased $9.0 million, or 6%, over the nine months ended September 30, 2016. Collaboration and licensing revenues increased $7.2 million, or 9%, over the nine months ended September 30, 2016, primarily due to the recognition of deferred revenue associated with the payment received in June 2016 from ZIOPHARM to amend the collaborations between the parties and increased revenues associated with collaborations entered into with the Harvest start-up entities in 2016. Product revenues decreased $2.9 million, or 10%, primarily due to lower customer demand for cows and live calves. Gross margin on products improved in the current period primarily due to a decline in the average cost of cows. Service revenues increased $4.6 million, or 14%, due to an increase in the number of bovine in vitro fertilization cycles performed due to higher customer demand. Gross margin on services decreased slightly in the current period primarily due to an increase in royalties and commissions due to vendors.
Research and development expenses increased $21.4 million, or 26%, due primarily to increases in (i) salaries, benefits and other personnel costs for research and development employees, (ii) lab supplies and consulting expenses, (iii) depreciation and amortization, and (iv) rent and utilities expenses. Salaries, benefits and other personnel costs increased $7.4 million due to an increase in research and development headcount necessary to invest in current or expanding platforms and to develop new prospective collaborations and other partnering opportunities. Lab supplies and consulting expenses increased $6.3 million as a result of (i) the progression of certain programs into the preclinical and clinical phases with certain of Intrexon’s collaborators and (ii) the expansion or improvement of certain of the Company’s platform technologies. Depreciation and amortization increased $4.3 million primarily as a result of (i) the amortization of developed technology acquired from Oxitec, which began in November 2016 upon the completion of certain operational and regulatory events, and (ii) the amortization of developed technology acquired from GenVec in June 2017. Rent and utilities expenses increased $2.5 million due to the expansion of certain facilities to support the Company’s increased headcount. SG&A expenses increased $6.3 million, or 6%. Salaries, benefits and other personnel costs increased $4.2 million primarily due to (i) increased headcount to support the Company’s expanding operations and (ii) increased stock-based compensation expense resulting from grants to certain of the Company’s officers in February 2017. Legal and professional fees increased $4.7 million primarily due to (i) increased legal fees to defend ongoing litigation, (ii) increased business development and public relations consulting expenses, and (iii) the Company’s acquisition of GenVec that was completed in June 2017. These increases were offset by $4.2 million in litigation expenses recorded in the prior period arising from the entrance of a court order in Trans Ova Genetics, L.C.’s trial with XY, LLC.
Total other income (expense), net, increased $66.8 million, or 171%. This increase was primarily attributable to (i) increases in fair market value of the Company’s equity securities portfolio, investments in preferred stock and other convertible instruments and (ii) dividend income from the Company’s investments in preferred stock.
Equity in net loss of affiliates, which includes the Company’s pro-rata share of the net losses of its investments accounted for using the equity method of accounting, decreased $5.7 million, or 33%. This decrease was primarily due to the temporary redeployment of certain of the Company’s resources away from these joint venture programs towards supporting prospective new platforms and additional collaborations.
Conference Call and Webcast
The Company will host a conference call today Thursday, November 9th, at 5:30 PM ET to discuss the third quarter 2017 financial results and provide a general business update. The conference call may be accessed by dialing 1-888-317-6003 (Domestic US), 1-866-284-3684 (Canada), and 1-412-317-6061 (International) and providing the number 7741944 to join the Intrexon Corporation Call. Participants may also access the live webcast through Intrexon’s website in the Investors section at http://investors.dna.com/events.
About Intrexon Corporation
Intrexon Corporation (NYSE: XON) is Powering the Bioindustrial Revolution with Better DNA™ to create biologically-based products that improve the quality of life and the health of the planet. Intrexon’s integrated technology suite provides its partners across diverse markets with industrial-scale design and development of complex biological systems delivering unprecedented control, quality, function, and performance of living cells. We call our synthetic biology approach Better DNA®, and we invite you to discover more at www.dna.com or follow us on Twitter at @Intrexon, on Facebook, and LinkedIn.
Non-GAAP Financial Measures
This press release presents Adjusted EBITDA and Adjusted EBITDA per share, which are non-GAAP financial measures within the meaning of applicable rules and regulations of the Securities and Exchange Commission (SEC). For a reconciliation of these measures to the most directly comparable financial measure calculated in accordance with generally accepted accounting principles and for a discussion of the reasons why the company
believes that these non-GAAP financial measures provide information that is useful to investors see the tables below under “Reconciliation of GAAP to Non-GAAP Measures.” Such information is provided as additional information, not as an alternative to Intrexon’s consolidated financial statements presented in accordance with GAAP, and is intended to enhance an overall understanding of the Intrexon’s current financial performance.
Trademarks
Intrexon, Arctic, ActoBiotics, RTS, Powering the Bioindustrial Revolution with Better DNA, and Better DNA are trademarks of Intrexon and/or its affiliates. Other names may be trademarks of their respective owners.
Safe Harbor Statement
Some of the statements made in this press release are forward-looking statements that involve a number of risks and uncertainties and are made pursuant to the Safe Harbor Provisions of the Private Securities Litigation Reform Act of 1995. These forward-looking statements are based upon Intrexon’s current expectations and projections about future events and generally relate to Intrexon’s plans, objectives and expectations for the development of Intrexon’s business. Although management believes that the plans and objectives reflected in or suggested by these forward-looking statements are reasonable, all forward-looking statements involve risks and uncertainties and actual future results may be materially different from the plans, objectives and expectations expressed in this press release. These risks and uncertainties include, but are not limited to, (i) Intrexon’s current and future collaborations and joint ventures; (ii) Intrexon’s ability to successfully enter new markets or develop additional products, whether with its collaborators or independently; (iii) actual or anticipated variations in Intrexon’s operating results; (iv) actual or anticipated fluctuations in Intrexon’s competitors’ or its collaborators’ operating results or changes in their respective growth rates; (v) Intrexon’s cash position; (vi) market conditions in Intrexon’s industry; (vii) the volatility of Intrexon’s stock price; (viii) Intrexon’s ability, and the ability of its collaborators, to protect Intrexon’s intellectual property and other proprietary rights and technologies; (ix) Intrexon’s ability, and the ability of its collaborators, to adapt to changes in laws or regulations and policies; (x) the outcomes of pending or future litigation; (xi) the rate and degree of market acceptance of any products developed by a collaborator under an ECC or through a joint venture; (xii) Intrexon’s ability to retain and recruit key personnel; (xiii) Intrexon’s expectations related to the use of proceeds from its public offerings and other financing efforts; (xiv) Intrexon’s estimates regarding expenses, future revenue, capital requirements and needs for additional financing; and (xv) Intrexon’s expectations relating to its subsidiaries and other affiliates. For a discussion of other risks and uncertainties, and other important factors, any of which could cause Intrexon’s actual results to differ from those contained in the forward-looking statements, see the section entitled “Risk Factors” in Intrexon’s Annual Report on Form 10-K, as well as discussions of potential risks, uncertainties, and other important factors in Intrexon’s subsequent filings with the Securities and Exchange Commission. All information in this press release is as of the date of the release, and Intrexon undertakes no duty to update this information unless required by law.
###
For more information regarding Intrexon Corporation, contact:
| Investor Contact: Christopher Basta Vice President, Investor Relations Tel: +1 (561) 410-7052 investors@intrexon.com |
Corporate Contact: Marie Rossi, Ph.D. Director, Technical Communications Tel: +1 (301) 556-9850 publicrelations@intrexon.com |
Intrexon Corporation and Subsidiaries
Consolidated Balance Sheets
(Unaudited)
| (Amounts in thousands) |
September 30, 2017 | December 31, 2016 | ||||||
| Assets |
||||||||
| Current assets |
||||||||
| Cash and cash equivalents |
$ | 64,216 | $ | 62,607 | ||||
| Restricted cash |
6,987 | 6,987 | ||||||
| Short-term investments |
44,502 | 174,602 | ||||||
| Receivables |
||||||||
| Trade, net |
18,134 | 21,637 | ||||||
| Related parties |
17,866 | 16,793 | ||||||
| Notes, net |
— | 1,500 | ||||||
| Other |
2,253 | 2,555 | ||||||
| Inventory |
17,730 | 21,139 | ||||||
| Prepaid expenses and other |
8,052 | 7,361 | ||||||
|
|
|
|
|
|||||
| Total current assets |
179,740 | 315,181 | ||||||
| Long-term investments |
— | 5,993 | ||||||
| Equity securities |
26,642 | 23,522 | ||||||
| Investments in preferred stock |
148,499 | 129,545 | ||||||
| Property, plant and equipment, net |
102,876 | 64,672 | ||||||
| Intangible assets, net |
240,897 | 225,615 | ||||||
| Goodwill |
166,821 | 157,175 | ||||||
| Investments in affiliates |
22,942 | 23,655 | ||||||
| Other assets |
9,844 | 3,710 | ||||||
|
|
|
|
|
|||||
| Total assets |
$ | 898,261 | $ | 949,068 | ||||
|
|
|
|
|
|||||
| Current liabilities |
||||||||
| Accounts payable |
$ | 7,852 | $ | 8,478 | ||||
| Accrued compensation and benefits |
11,206 | 6,540 | ||||||
| Other accrued liabilities |
18,960 | 15,776 | ||||||
| Deferred revenue |
48,289 | 53,364 | ||||||
| Lines of credit |
234 | 820 | ||||||
| Current portion of long term debt |
439 | 386 | ||||||
| Deferred consideration |
— | 8,801 | ||||||
| Related party payables |
816 | 440 | ||||||
|
|
|
|
|
|||||
| Total current liabilities |
87,796 | 94,605 | ||||||
| Long term debt, net of current portion |
7,673 | 7,562 | ||||||
| Deferred revenue, net of current portion |
227,998 | 256,778 | ||||||
| Deferred tax liabilities |
15,868 | 17,007 | ||||||
| Other long term liabilities |
5,747 | 3,868 | ||||||
|
|
|
|
|
|||||
| Total liabilities |
345,082 | 379,820 | ||||||
|
|
|
|
|
|||||
| Commitments and contingencies |
||||||||
| Total equity |
||||||||
| Common stock |
— | — | ||||||
| Additional paid-in capital |
1,370,917 | 1,325,780 | ||||||
| Accumulated deficit |
(820,554 | ) | (729,341 | ) | ||||
| Accumulated other comprehensive loss |
(16,750 | ) | (36,202 | ) | ||||
|
|
|
|
|
|||||
| Total Intrexon shareholders’ equity |
533,613 | 560,237 | ||||||
| Noncontrolling interests |
19,566 | 9,011 | ||||||
|
|
|
|
|
|||||
| Total equity |
553,179 | 569,248 | ||||||
|
|
|
|
|
|||||
| Total liabilities and total equity |
$ | 898,261 | $ | 949,068 | ||||
|
|
|
|
|
|||||
Intrexon Corporation and Subsidiaries
Consolidated Statements of Operations
(Unaudited)
| (Amounts in thousands, except share and per share data) |
Three months ended September 30, |
Nine months ended September 30, |
||||||||||||||
| 2017 | 2016 | 2017 | 2016 | |||||||||||||
| Revenues |
||||||||||||||||
| Collaboration and licensing revenues |
$ | 28,155 | $ | 30,590 | $ | 89,384 | $ | 82,144 | ||||||||
| Product revenues |
7,670 | 9,260 | 25,780 | 28,699 | ||||||||||||
| Service revenues |
9,975 | 8,706 | 37,890 | 33,298 | ||||||||||||
| Other revenues |
216 | 429 | 899 | 783 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total revenues |
46,016 | 48,985 | 153,953 | 144,924 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Operating Expenses |
||||||||||||||||
| Cost of products |
8,001 | 9,156 | 25,625 | 29,471 | ||||||||||||
| Cost of services |
7,013 | 5,803 | 21,805 | 17,807 | ||||||||||||
| Research and development |
36,472 | 29,035 | 104,663 | 83,266 | ||||||||||||
| Selling, general and administrative |
39,277 | 33,812 | 113,258 | 106,956 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total operating expenses |
90,763 | 77,806 | 265,351 | 237,500 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Operating loss |
(44,747 | ) | (28,821 | ) | (111,398 | ) | (92,576 | ) | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Other Income (Expense), Net |
||||||||||||||||
| Unrealized and realized appreciation (depreciation) in fair value of equity securities and preferred stock |
2,175 | 412 | 9,240 | (45,388 | ) | |||||||||||
| Interest expense |
(138 | ) | (227 | ) | (498 | ) | (759 | ) | ||||||||
| Interest and dividend income |
5,070 | 4,494 | 14,437 | 5,817 | ||||||||||||
| Other income, net |
(1,021 | ) | (32 | ) | 4,453 | 1,205 | ||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total other income (expense), net |
6,086 | 4,647 | 27,632 | (39,125 | ) | |||||||||||
| Equity in net loss of affiliates |
(2,993 | ) | (6,255 | ) | (11,273 | ) | (16,951 | ) | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Loss before income taxes |
(41,654 | ) | (30,429 | ) | (95,039 | ) | (148,652 | ) | ||||||||
| Income tax benefit |
818 | 418 | 2,164 | 3,290 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net loss |
$ | (40,836 | ) | $ | (30,011 | ) | $ | (92,875 | ) | $ | (145,362 | ) | ||||
| Net loss attributable to the noncontrolling interests |
1,147 | 1,029 | 3,123 | 2,887 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net loss attributable to Intrexon |
$ | (39,689 | ) | $ | (28,982 | ) | $ | (89,752 | ) | $ | (142,475 | ) | ||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net loss per share, basic and diluted |
$ | (0.33 | ) | $ | (0.24 | ) | $ | (0.75 | ) | $ | (1.21 | ) | ||||
|
|
|
|
|
|
|
|
|
|||||||||
| Weighted average shares outstanding, basic and diluted |
120,518,885 | 118,346,782 | 119,741,291 | 117,785,160 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
Intrexon Corporation and Subsidiaries
Reconciliation of GAAP to Non-GAAP Measures
(Unaudited)
Adjusted EBITDA and Adjusted EBITDA per share. To supplement Intrexon’s financial information presented in accordance with U.S. generally accepted accounting principles (“GAAP”), Intrexon presents Adjusted EBITDA and Adjusted EBITDA per share. A reconciliation of Adjusted EBITDA to net income or loss attributable to Intrexon under GAAP appears below. Adjusted EBITDA is a non-GAAP financial measure that Intrexon calculates as net income or loss attributable to Intrexon adjusted for income tax expense or benefit, interest expense, depreciation and amortization, stock-based compensation, shares issued as compensation for services, bad debt expense, litigation expense, realized and unrealized appreciation or depreciation in the fair value of equity securities and preferred stock, and equity in net loss of affiliates. Adjusted EBITDA and Adjusted EBITDA per share are key metrics for Intrexon’s management and Board of Directors for evaluating the Company’s financial and operating performance, generating future operating plans and making strategic decisions about the allocation of capital. Management and the Board of Directors believe that Adjusted EBITDA and Adjusted EBITDA per share are useful to understand the long-term performance of Intrexon’s core business and facilitate comparisons of the Company’s operating results over multiple reporting periods. Intrexon is providing this information to investors and others to assist them in understanding and evaluating the Company’s operating results in a manner similar to how its management and Board of Directors evaluate operating results (except for the impact of the change in deferred revenue related to upfront and milestone payments, which is adjusted in the measures evaluated by management and the Board of Directors as discussed below). While Intrexon believes that its non-GAAP financial measures are useful in evaluating its business, and may be of use to investors, this information should be considered as supplemental in nature and is not meant as a substitute for the related financial information prepared in accordance with GAAP. In addition, these non-GAAP financial measures may not be the same as non-GAAP financial measures presented by other companies. Adjusted EBITDA and Adjusted EBITDA per share are not measures of financial performance under GAAP, and are not intended to represent cash flows from operations nor earnings per share under GAAP and should not be used as an alternative to net income or loss as an indicator of operating performance or to represent cash flows from operating, investing or financing activities as a measure of liquidity. Intrexon compensates for the limitations of Adjusted EBITDA and Adjusted EBITDA per share by using them only to supplement the Company’s GAAP results to provide a more complete understanding of the factors and trends affecting the Company’s business. Adjusted EBITDA and Adjusted EBITDA per share have limitations as an analytical tool and you should not consider them in isolation or as a substitute for analysis of Intrexon’s results as reported under GAAP.
In addition to the reasons stated above, which are generally applicable to each of the items Intrexon excludes from its non-GAAP financial measure, Intrexon believes it is appropriate to exclude certain items from the definition of Adjusted EBITDA for the following reasons:
| • | Interest expense may be subject to changes in interest rates which are beyond Intrexon’s control; |
| • | Depreciation of Intrexon’s property and equipment and amortization of acquired identifiable intangibles can be affected by the timing and magnitude of business combinations and capital asset purchases; |
| • | Stock-based compensation expense is a noncash expense and may vary significantly based on the timing, size and nature of awards granted and also because the value is determined using formulas which incorporate variables, such as market volatility; |
| • | Shares issued as compensation for services and bad debt expense are noncash expenses which Intrexon excludes in evaluating its financial and operating performance; |
| • | Unrealized and realized appreciation or depreciation in the fair value of securities which Intrexon holds in its collaborators may be significantly impacted by market volatility and other factors which are outside of the Company’s control in the short term and Intrexon intends to hold these securities over the long term, except as otherwise disclosed; |
| • | Equity in net loss of affiliate reflects Intrexon’s proportionate share of the income or loss of entities over which the Company has significant influence, but not control, and accounts for using the equity method of accounting. Intrexon believes excluding the impact of such losses or gains on these types of strategic investments from its operating results is important to facilitate comparisons between periods; and |
| • | Litigation expense is an estimate of the net amount due, including prejudgment interest, as a result of the final court order from Trans Ova’s trial with XY, LLC. Intrexon believes it has compelling grounds to overturn the adverse rulings of the court order through appellate action and that, as a result, the amount of the damages could be reduced or eliminated. |
Furthermore, supplemental information about the impact of the change in deferred revenue related to upfront and milestone payments is provided below. GAAP requires Intrexon to account for its collaborations as multiple-element arrangements. As a result, the Company initially defers certain collaboration revenues because certain of its performance obligations cannot be separated and must be accounted for as one unit of accounting. The collaboration revenues that Intrexon so defers arise from upfront and milestone payments received from the Company’s collaborators, which Intrexon recognizes over the future performance period even though the Company’s right to such consideration is neither contingent on the results of Intrexon’s future performance nor refundable in the event of nonperformance. The supplemental information about the change in deferred revenue removes the noncash revenue recognized during the period and includes the cash and stock received from collaborators for upfront and milestone payments during the period. Management and the Board of Directors consider this information in evaluating Intrexon’s operating performance as they believe it permits the quarterly and annual comparisons of the Company’s ability to consummate new collaborations or to achieve significant milestones with existing collaborators.
The following table presents a reconciliation of net income (loss) attributable to Intrexon to EBITDA and also to Adjusted EBITDA, as well as the calculation of Adjusted EBITDA per share, for each of the periods indicated:
| Three months ended | Nine months ended | |||||||||||||||
| September 30, | September 30, | |||||||||||||||
| 2017 | 2016 | 2017 | 2016 | |||||||||||||
| (In thousands) | ||||||||||||||||
| Net loss attributable to Intrexon |
$ | (39,689 | ) | $ | (28,982 | ) | $ | (89,752 | ) | $ | (142,475 | ) | ||||
| Interest expense |
122 | 159 | 451 | 615 | ||||||||||||
| Income tax benefit |
(818 | ) | (418 | ) | (2,164 | ) | (3,290 | ) | ||||||||
| Depreciation and amortization |
7,866 | 5,858 | 22,502 | 17,292 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| EBITDA |
$ | (32,519 | ) | $ | (23,383 | ) | (68,963 | ) | (127,858 | ) | ||||||
| Stock-based compensation |
12,042 | 10,772 | 31,913 | 30,569 | ||||||||||||
| Shares issued as payment for services |
2,730 | 2,595 | 8,440 | 8,284 | ||||||||||||
| Bad debt expense |
511 | 426 | 1,093 | 1,609 | ||||||||||||
| Litigation expense |
— | — | — | 4,228 | ||||||||||||
| Unrealized and realized (appreciation) depreciation in fair value of equity securities and preferred stock |
(2,175 | ) | (412 | ) | (9,240 | ) | 45,388 | |||||||||
| Equity in net loss of affiliates |
2,993 | 6,255 | 11,273 | 16,951 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Adjusted EBITDA |
$ | (16,418 | ) | $ | (3,747 | ) | $ | (25,484 | ) | (20,829 | ) | |||||
|
|
|
|
|
|
|
|
|
|||||||||
| Weighted average shares outstanding, basic and diluted |
120,518,885 | 118,346,782 | 119,741,291 | 117,785,160 | ||||||||||||
| Adjusted EBITDA per share, basic and diluted |
$ | (0.14 | ) | $ | (0.03 | ) | $ | (0.21 | ) | $ | (0.18 | ) | ||||
|
|
|
|
|
|
|
|
|
|||||||||
| Supplemental information: |
||||||||||||||||
| Impact of change in deferred revenue related to upfront and milestone payments |
$ | (8,613 | ) | $ | (1,811 | ) | $ | (28,218 | ) | $ | 127,795 | |||||
|
|
|
|
|
|
|
|
|
|||||||||