
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM
For the fiscal year ended
or
For the transition period from ______ to ______.
Commission File Number:
___________________________________________________
(Exact name of registrant as specified in its charter)
___________________________________________________
| (State of Incorporation) | (IRS Employer Identification No.) | |
|
|
||
| (Address of principal executive offices) | (Zip Code) |
Registrant's telephone number, including area code:
___________________________________________________
Securities registered under Section 12(b) of the Act:
| Title of each class | Name of each exchange on which registered |
| N/A | N/A |
Securities registered under Section 12(g) of the Act:
(Title of class)
___________________________________________________
Indicate by check mark if the registrant is a
well-known seasoned issuer, as defined in Rule 405 of the Securities Act. ☐ Yes ☒
Indicate by check mark if the registrant is not
required to file reports pursuant to Section 13 or Section 15(d) of the Act. ☐ Yes ☒
Indicate by check mark whether the registrant
(1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months
(or for such shorter period that the issuer was required to file such reports), and (2) has been subject to such filing requirements for
the past 90 days. ☒
Indicate by check mark whether the registrant
has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405
of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). ☒
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§ 229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant's knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ☒
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer”, “smaller reporting company”, and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer ☐ | Accelerated filer ☐ | |
| Smaller reporting company | ||
| Emerging growth company |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☐
Indicate by check mark whether the registrant is a shell company (as
defined in Rule 12b-2 of the Act). ☐ Yes ☒
On August 31st, 2022, the last business day of
the registrant’s most recently completed fourth quarter, the aggregate market value of the Common Stock held by non-affiliates
of the registrant was $
As of November 29, 2022, the registrant had shares of its Common Stock, $0.0001 par value, outstanding.
Table of Contents
| i |
FORWARD LOOKING STATEMENTS
Certain statements made in this Annual Report are “forward-looking statements” (within the meaning of the Private Securities Litigation Reform Act of 1995) regarding the plans and objectives of management for future operations. Such statements involve known and unknown risks, uncertainties and other factors that may cause actual results, performance or achievements of the Company to be materially different from any future results, performance or achievements expressed or implied by such forward-looking statements. The forward-looking statements made in this Report are based on current expectations that involve numerous risks and uncertainties. The Company’s plans and objectives are based, in part, on assumptions involving the growth and expansion of business. Assumptions relating to the foregoing involve judgments with respect to, among other things, future economic, competitive and market conditions and future business decisions, all of which are difficult or impossible to predict accurately and many of which are beyond the control of the Company. Although the Company believes that its assumptions underlying the forward-looking statements are reasonable, any of the assumptions could prove inaccurate and, therefore, there can be no assurance that the forward-looking statements made in this Report will prove to be accurate. In light of the significant uncertainties inherent in the forward-looking statements made in this Report, the inclusion of such information should not be regarded as a representation by the Company or any other person that the objectives and plans of the Company will be achieved.
As used in this Annual Report, the terms “we”, “us”, “our”, “Company”, and “CNBX” means CNBX Pharmaceuticals Inc., unless otherwise indicated.
| ii |
PART I
Item 1. Description of Business
CNBX PHARMACEUTICALS, INC. is based in Bethesda, Maryland, is a clinical stage company dedicated to discovery, development and commercialization of novel cannabinoid-based products and innovative technologies for the treatment of cancer. Our first lead product candidate is a Cannabics SR, the oral capsule developed for the treatment of patients with advanced cancer and cancer anorexia cachexia syndrome (CACS). Our leading anti-neoplastic drug candidate under development for Colorectal Cancer (CRC) is RCC-33.
Historically we were previously an exploration stage mining company which transitioned into a bio-tech company in 2014.
Our corporate address is #3 Bethesda Metro Center, Suite 700, Bethesda, Maryland, 20814; Telephone (877) 424-2429.
The Company was previously engaged in the oil and gas exploration business. On April 29th, 2014, the Company began a new direction and the majority of the Shareholders of the Company elected the Board of Directors and renamed the Company CNBX Pharmaceuticals Inc. The Company’s R&D is conducted in a Government licensed lab facility in Israel with the focus of development of cannabinoid-based therapies, medications and administration routes for treatment of cancer.
History:
CNBX Pharmaceuticals Inc. was incorporated on September 15, 2004, under the laws of the State of Nevada, as Thrust Energy Corp., for the purpose of acquiring undivided working interests in small oil and gas exploration properties and non-operating interests in both producing and exploration projects throughout the United States and Canada.
On September 30, 2010, we increased our authorized capital to 900 million shares of common stock (par value $0.0001) and 100 million shares of preferred stock (par value $0.0001) and effected a 20-for-1 reverse split of our issued and outstanding common stock. As a result of the reverse split, our issued and outstanding common stock was reduced from 13,604,000 shares to 680,202 shares and 5,000,000 preferred shares.
Due to our inability to earn any meaningful revenue from oil and gas exploration, our management determined in April 2011 that we should change our business plan to include toll milling and refining.
On May 5, 2011, we effected a change of name to American Mining Corp. by completing a short form merger with a wholly-owned subsidiary.
On April 25, 2014, Cannabics Inc., a Delaware Corporation, purchased 20,500,000 shares of restricted stock of the Company, thus acquiring control of the Company.
On June 3, 2014, the Company's Board of Directors declared a two-to-one forward stock split of all outstanding shares of common stock. The stock split was approved by FINRA on June 19th, 2014. The effect of the stock split increased the number of shares of common stock outstanding from 40,880,203 to 81,760,406. All common share and per common share data in these financial statements and related notes hereto have been retroactively adjusted to account for the effect of the stock split for all periods presented prior to June 3rd, 2014. The total number of authorized common shares and the par value thereof was not changed by the split.
On June 19, 2014, FINRA granted final approval of Change of Name & Ticker Symbol of the Corporation from American Mining Corporation to CNBX PHARMACEUTICALS INC., with the new Ticker Symbol of “CNBX”. Said approval was predicated upon CNBX Pharmaceuticals Inc.’s filing of Articles of Merger with American Mining Corporation with the Nevada Secretary of State on May 21st, 2014. Under the laws of the State of Nevada, CNBX Pharmaceuticals Inc. was merged with and into the Registrant, with the Registrant being the surviving entity. The Merger was completed under Section 92A.180 of the Nevada Revised Statutes, Chapter 92A, as amended, and as such, does not require the approval of the stockholders of either the Registrant or CNBX Pharmaceuticals Inc.
On July 24, 2014, the Company executed a Collaboration Agreement with Cannabics Inc. (“Cannabics”), a Delaware corporation and largest shareholder of the Company. Per the terms of the Agreement, the Company issued 18,239,594 shares of its common stock to acquire the entire institutional knowledge of Cannabics, Inc., which primarily consists of in-process Research & Development technology, the cumulative result of its years of scientific institutional knowledge in the fields of Molecular Biology, Cancer and Pharmacology research. Additionally, Cannabics tendered $150,000 to the Company specifically earmarked as working funds towards prospective projects of the Company. Per the Agreement, from that day forth they have carried forward their research and development as part of, and for the exclusive benefit of the Company, which initial findings have now branched out into new and divergent discoveries.
| 1 |
On August 25, 2014, CNBX Pharmaceuticals Inc. incorporated a wholly owned subsidiary in Israel, named “G.R.I.N Ultra Ltd”, dedicated to advanced research and development.
On July 3, 2018, the Company announced the conclusion of its Clinical Trial of Cannabics SR 5mg drug for Cancer Anorexia Cachexia Syndrome, as noted on the press release of that date.
On June 16th, 2020, the Company announced its appointment of Dr. Erez Scapa, MD, to its Scientific Board of Advisors. Dr. Scapa is an Expert in Invasive Gastroenterology in the Sourasky Medical Center in Tel-Aviv, Israel, where he is head of the Endoscopic Submucosal Dissection (ESD) program.
On August 5th, 2020, the Company announced its appointment of Dr. Dana Ben-Ami Shor to its Scientific Board of Advisors, where she will help lead the design and implementation of the company's clinical validation plan of its novel drug candidates for the treatment of colorectal cancer.
On August 20th, 2020, the Company announced the creation of a new Division for its Anti-Tumor drug candidate RCC-33, for the treatment of colorectal cancer. The emanates from the Company’s focus on a clinical validation path, including in-vivo experiments, collaborations with key medical centers, and the preparation of a product dossier with which the company plans to schedule a Pre IND-Meeting with the U.S. Food and Drug Administration (the “FDA”).
On October 18th, 2021, the Company filed 2 new Provisional Patent applications on Compositions and Methods for treating cancer, including colorectal cancer and early intervention therapy for colorectal cancer patients.
On May 10, 2022, the Company changed its name from “Cannabics Pharmaceuticals Inc.” to the current CNBX Pharmaceuticals Inc.; effectuated a one-for-one hundred twenty (1:120) reverse split of the issued and outstanding shares of common stock of the Company and decreased its authorized shares of preferred stock from 100,000,000 to 5,000,000 as noted in the 8K of May 13th, 2022.
On July 20th, 2022, Dr. Eyal Ballan resigned his position as Chief Technical Officer, and was replaced by Dr. Sanja Goldberg as noted in the 8K of July 24th, 2022.
On November 8th, 2022 the Company announced that it has acquired controlling interest in TaGeza Biopharmaceuticals Ltd. The TaGeza research team remains committed to the Company and co-founder Prof. Benjamin Dekel (MD, PhD) will continue to serve as the Company Chief Scientist; Gabriel Yariv, to assume deposition of TaGeza Biopharmaceuticals CEO.
Our Business:
Company Overview
We are a clinical-stage company specializing in the discovery, development and commercialization of novel cannabinoid-based products and innovative technologies for the treatment of cancer.
Our first lead product candidate is Cannabics SR the oral capsule developed for the treatment of patients with advanced cancer and cancer anorexia cachexia syndrome (CACS), showed promising results in a peer-reviewed clinical study that concluded the results justify a larger clinical study. For oncology, we intend to pursue a broad strategy of combining our technology platforms with conventional oncology therapies, based on their mechanisms of action, safety profiles and versatility. Our leading anti-neoplastic drug candidate under development for Colorectal Cancer (CRC) is RCC-33, a first-in-class therapy being developed primarily in two settings: one to reduce tumor cell activity in CRC patients as a standalone in neoadjuvant treatment or “window of opportunity” at the time after colonoscopy, prior to cancer staging; and another for patients with refractory to therapy and adjuvant to surgery also at the time after colonoscopy. Neoadjuvant treatment is the administration of antitumor therapy as a first step to shrink a cancerous tumor prior to surgical intervention. We intend to initiate Phase I/II clinical trials for both candidates in 2023.
| 2 |
1. General:
CNBX Pharmaceuticals Inc. is a clinical stage pharmaceutical company primarily focused on the development of novel cannabinoid-based products and innovative technologies for the treatment of cancer.
The company is currently preparing to launch Phase I/II (a) clinical study in 2023, for the evaluation of its lead drug candidates Cannabics SR for the treatment of patients with advanced cancer and cancer anorexia cachexia syndrome (CACS) and RCC-33 for the treatment of colorectal cancer. The company’s activities are centered around the company’s biological laboratory facilities located in Rehovot Israel, where our scientific team leads the company’s research and development efforts.
Our company’s core activities consist of:
| · | Drug Discovery: development of novel molecular formulations and drug candidates; | |
| · | Intellectual Property: filing of corresponding IP to protect our products; and | |
| · | Regulatory Affairs: initiation of the regulatory pathway for each drug candidate in our development pipeline |
Our current business model is to undertake an FDA regulatory pathway for each of the new drug candidates under IND (Investigational New Drug) classification and complete a successful Phase I/II(a) clinical study (toxicity and proof of concept in humans). In reaching this milestone, where an initial feasibility in humans was demonstrated, the company will have gained several commercial opportunities for capitalizing on each such product candidate, including entering into commercial agreements with larger pharma corporations. Accordingly, our company does not engage in any manufacturing, distribution, or sales of products, nor is it foreseeable to expect that we will in the near future.
2. Development pipeline:
2.1. Cannabics SR for Cancer Anorexia-Cachexia Syndrome
Overview
We are developing Cannabics SR as a product candidate for the treatment of CACS. Cannabics SR is a sustained-release oral capsule containing a standardized compound of cannabinoids that has demonstrated a clinically significant weight increase in CACS patients in a peer-reviewed pilot study conducted by Dr. Gil Bar-Sela of the Rambam Hospital Health Care Campus, Division of Oncology, in Haifa, Israel. Our patent-pending technology provides for a convenient, once or twice daily administration, with rapid onset and a steady state of therapeutic effect for a 6 to 8-hour duration.
Cancer Anorexia-Cachexia Syndrome
CACS is a common complication of cancer associated with high morbidity and mortality. It is a complex metabolic syndrome in which a persistently elevated basal metabolic rate is not compensated for by adequate calorie or protein intake, causing involuntary and progressive weight loss leading to increasing functional impairment in cancer patients, especially in advanced stages of the disease. Once established, CACS cannot presently be reversed using available pharmacological or nutritional support techniques.
Unlike starvation, body-weight loss in CACS patients arises mainly from loss of muscle mass, characterized by increased catabolism of skeletal muscle and decreased protein synthesis. This weight loss is associated with important clinical outcomes such as increased morbidity, diminished effectiveness of chemotherapy, muscle wasting, inflammation, fatigue, and reduced survival expectations. The impact of CACS on the patient is not, however, limited to the effect of weight loss. Quality of life, functional abilities, symptoms, psychological outcomes, and social aspects are all affected by CACS.
| 3 |
According to the National Cancer Institute, nearly one-third of cancer deaths can be attributed to the severe weight loss and “metabolic mutiny” associated with CACS, and more than 50% of patients with cancer die with cachexia being present. The overall prevalence of CACS is currently estimated to range from 40% at cancer diagnosis to 70-80% in advanced phases of the disease (Source: Critical Reviews in Oncology/Hematology, 2013;88(3):625-636), while the overall prevalence of weight loss in cancer patients may be as high as 86% in the last 1-2 weeks of life (Source: Journal of Pain and Symptom Management 2007;34:94–104).
The cause and subsequent development of CACS is still poorly understood, but several factors and biological pathways are known to be involved, including inflammation, decreased secretion of anabolic hormones, and altered metabolic response. While there have been important advances in the study of CACS over the past decade, including progress in understanding its mechanisms and the development of promising pharmacologic and supportive care interventions, there is presently no effective pharmacologic therapy for CACS.
Current treatments for CACS are generally based on nutritional support and CACS pathophysiology-modulating drugs, with the most common being the progestogens, megestrol and medroxyprogesterone, and corticosteroids. Progestogens appear to stimulate appetite and improvements in body weight by increasing adipose tissue, but have not been confirmed to augment lean body mass. Megestrol also carries an increased risk of mortality and thromboembolism. Nonetheless, megestrol is the only FDA approved treatment option for CACS and no drug to date has been shown to be superior to it in efficacy and tolerability. Corticosteroids are also considered effective in stimulating appetite and reducing fatigue but should only be used for short periods and in selected cases because of side effects from longer term use, such as insulin resistance, fluid retention, steroidal myopathy, skin fragility, adrenal insufficiency, and sleep and cognitive disorders. Other drugs are being investigated or are in development. Given the dearth of approved therapies, we believe that CACS remains a significant area of unmet medical need.
Cannabinoid Therapies for CACS
Cannabis has long been suggested as a well-tolerated, safe, and effective option to help patients cope with cancer related symptoms with fewer serious side effects than most prescription drugs currently used as anti-emetics, analgesics, and the like. As such, cannabinoids are finding application in palliative care for reducing nausea and vomiting, alleviating cancer pain, and stimulating appetite, as well as improving quality of life in cancer patients. Dronabinol (Marinol®) and nabilone (Cesamet®), two drugs based on synthetic cannabinoids, have each been approved by the FDA for the treatment of chemotherapy-related nausea in patients who do not respond to conventional antiemetic therapy. Another drug, nabiximols (Sativex®), a specific cannabis extract, is approved in Canada and the United Kingdom for symptomatic relief of pain in advanced cancer patients.
Despite interest in cannabinoid-based therapies as a treatment for CACS, their use has been limited by impediments beyond the legal status of cannabis. The most significant obstacle is the lack of clinical research demonstrating their efficacy. While there is evidence that cannabinoids improve appetite, body weight, body fat level, caloric intake, mood, and quality of life in cancer patients, the few studies on these effects have yielded mixed and inconclusive findings. In addition, some of these studies have suffered from methodological constraints that limit any ability to draw firm conclusions.
The therapeutic use of cannabinoids has also been inhibited by limitations associated with traditional administration routes that reduce their effectiveness. Smoking and ingestion of cannabis suffer from wide variability in potency due to a lack of standardized and reproducible formulations. The ingestion of unformulated cannabis has also been associated with poor absorption and low bioavailability versus other administration routes, requiring higher doses and a greater risk of negative side effects. Additionally, the lack of available information on cannabinoid strains has made it difficult for healthcare providers to establish dosing rates. In our experience, however, the principal concern of patients with respect to medical cannabis lies in the undesirable side effects, such as disorientation and dizziness, which result from significant variability in peak blood levels of active cannabinoids soon after administration. We further believe that these side effects, which are common among immediate release methods, are a significant factor in the failure of patients to adhere to recommended treatment regimens and are therefore a pervasive threat to their health and wellbeing.
| 4 |
Cannabics SR
Cannabics SR is an oral composition in the form of a hydroxypropylmethylcellulose (HPMC) capsule containing a patent-pending formulation of cannabinoid extracts suspended in a lipid emulsion. It provides a relatively rapid onset of action, typically within 30-40 minutes, followed by a gradual and sustained release of active cannabinoids, resulting in a steady state level of beneficial effects for up to 6 to 8 hours with each capsule. Cannabics SR provides a consistent, predictable concentration of cannabinoids with an absorption profile and bioavailability of active ingredients that we believe to be superior to other oral cannabinoid administrations. We believe that the multifactorial benefits of the active pharmaceutical ingredients in Cannabics SR address an unmet medical need for a safe and effective treatment of CACS, leading to improved patient adherence and better health outcomes.
Cannabics SR capsules contain only food grade materials without any artificial additives. The active ingredients of each capsule are standardized in composition, formulation, and dose, and are comprised of only pure, natural extracts of active cannabinoids from selected strains of medical cannabis. All excipients are recognized by the FDA as Generally Regarded as Safe.
In addition to the therapeutic potential of Cannabics SR as a treatment for CACS, we believe that our SR technology may be formulated to serve the unique needs of patients suffering from other indications for which a sustained release of a cannabinoid formulation may be beneficial.
Clinical Development
In 2016, we commenced a two-year pilot study to evaluate the influence of Cannabics SR capsules on CACS, and, in particular, on weight loss in advanced cancer patients. The study was led by Professor Gil Bar-Sela, the former Deputy Director of the Division of Oncology at Rambam Health Care Campus, Head of the Palliative and Supportive Oncology Unit, and Head of Service for Melanoma and Sarcoma Patients.
Patients were administered 2 × 10 mg of Cannabics SR per 24 hours for six months. During the study, after some patients reported several psychoactive side effects, the dosage of each capsule was reduced to 5 mg. Almost no side effects were reported with the 5 mg dosage. Participants were weighed at each physician visit. The primary objective of the study was a weight gain of ≥10% from baseline. Of 24 patients who agreed to participate in the study, 17 started the Cannabics SR treatment, but only 11 received the capsules for more than two weeks. Three of six patients who completed the study period met the primary end-point. The remaining three patients had stable weights. In quality-of-life questionnaires patients reported less appetite loss after the Cannabics SR treatment (p=0.05). According to patients’ self-reports, improvement in appetite and mood as well as a reduction in pain and fatigue was demonstrated.
Despite various limitations, the preliminary study demonstrated a weight increase of ≥10% in 3 out of 17 (17.6%) of patients with doses of 5 mg × 1 or 5 mg × 2 capsules daily, without significant side effects. The remaining patients had stable weights. Also, all patients who remained in the study for at least 4.5 months reported an increase in appetite, as did 83% of the patients who completed the study. For 50% of the patients who completed the study, there were reports of pain reduction and sleep improvement. Additional results showed a significant decrease of appetite loss complaints among 83% of the patients who completed the study. (See Bar-Sela, Gil et al. “The Effects of Dosage-Controlled Cannabis Capsules on Cancer-Related Cachexia and Anorexia Syndrome in Advanced Cancer Patients: Pilot Study.” Integrative Cancer Therapies vol. 18 (2019): 1534735419881498. doi:10.1177/1534735419881498.)
Figure 1: Appetite loss among the six patients who completed Cannabics SR treatment, as reported on European Organization of Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLC-C30)

| 5 |
Commercialization
The results of our planned pilot studies may permit us to commercialize Cannabics SR in Israel under license by the Israeli Ministry of Health. If we are granted such a permit, we intend to engage a GMP manufacturer in Israel to produce Cannabics SR capsules for national distribution.
On May 13, 2020, the Israeli Ministry of Economy signed a Free Export Order, authorizing the export of GMP certified medical cannabis products from Israel. We are currently evaluating our export opportunities and optimal commercialization path for Cannabics SR across all available international markets, particularly with regard to the European Union, Canada, and Australia.
Cancer and Cancer Anorexia Cachexia Syndrome (CACS) Market Analysis
The dynamics of the cancer cachexia market are expected to shift in the coming years as a result of the positive outcomes of some of the rare candidates during the development stage by key players that are in the early stages of clinical development have the potential to create a significant positive shift. The emerging therapies are projected to be launched during the forecast period. In addition, the dearth of effective therapies for this condition presents a great opportunity for pharma companies to create novel drugs because there is less competition in the cancer cachexia market. Moreover, the rising awareness about the condition is also impacting the growth of the cancer cachexia market positively.
The Global Cancer Cachexia Market Size is projected to reach USD 2.93 billion by 2027, exhibiting a CAGR of 4.8% during the forecast period [2020-2027].
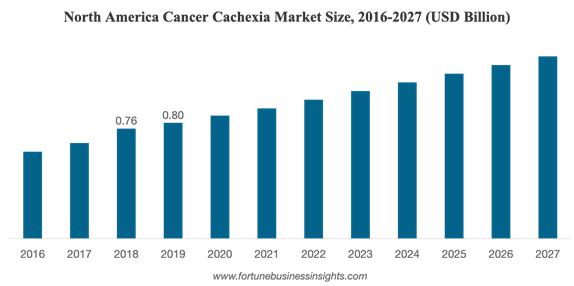
https://www.fortunebusinessinsights.com/cancer-cachexia-market-103262
North America region holds the largest market share of the global Cancer Anorexia-Cachexia Syndrome Drug North America is expected to hold a large market share in the global Cancer Anorexia-Cachexia Syndrome Drug Market due to the growing incidence of cancer cases. The International Agency for Research on Cancer (IARC) claims 13 million new cancer cases worldwide. The World Cancer Report provides that the incidence rate of new cancer cases is increased by 50% to 15 million in 2020. The existence of a highly developed healthcare system, the high degree of acceptance by medical practitioners of novel products, the total availability of advanced technological tools, FDA approval of new drugs and many companies are developing oncology products ( https://www.datamintelligence.com/research-report/ cancer-anorexia-cachexia-syndrome-drug-market)
| 6 |
2.2 RCC-33: colorectal cancer treatment drug candidate:
Our anti-neoplastic flagship product under development, RCC-33, is an antitumor drug candidate for the treatment of colorectal cancer, which is the 3rd most diagnosed and 2nd most lethal of all cancers, with approximately 2M new cases being diagnosed annually worldwide and a current market estimated at $12B, and which is expected to reach $17B by 2027.
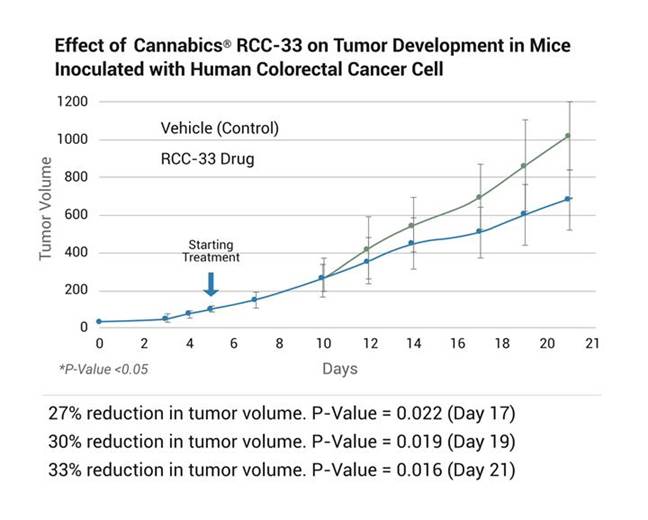
The RCC-33 proprietary formula consists of a specific synthetic cannabinoid molecular composition that has demonstrated the potential to reduce colorectal cancer tumor volume by over 30% in repeated in-vivo studies performed.
Overview
Cancer and Cannabinoids
Cancer is a general term used to describe a group of more than 100 related diseases characterized by uncontrolled growth and spread of abnormal cells, leading to the development of a mass commonly known as a tumor, followed by invasion of the surrounding tissues and subsequent spread, or metastasis, to other parts of the body. Despite enormous investment in research and the introduction of new treatments, cancer remains a critical area of unmet medical need. According to the World Health Organization, cancer is the second leading cause of mortality worldwide, responsible for an estimated nearly 10 million deaths in 2021. As of January 1, 2019, there were more than 16.9 million people with a history of cancer living in the United States, with 1.9 million new cases and 609,360 cancer deaths expected in 2022 (Source: American Cancer Society. Cancer Facts & Figures 2022).
Over the past decade, there has been growing interest in the therapeutic value of cannabinoid compounds in oncology. Cannabis has long been suggested as a well-tolerated, safe, and effective option to help patients cope with cancer related symptoms by reducing nausea and vomiting, alleviating cancer pain, stimulating appetite, and improving quality of life. Beyond their palliative benefits, however, cannabinoids have also been receiving increased attention for their anti-cancer potential, which we believe may one day revolutionize cancer therapy.
Cannabinoids are a diverse class of chemical compounds that occur naturally within cannabis plants and are pharmacologically similar to cannabinoids produced by the human body, known as endocannabinoids. Endocannabinoids form part of the human endocannabinoid system (ECS), a complex biological network that also includes cannabinoid receptors and enzymes involved in cannabinoid formation, transport, and degradation. The ECS is regarded as an important endogenous system implicated in regulation of the most vital biological processes to maintain homeostasis, assisting the body to remain stable and balanced despite external, or environmental, fluctuations (Source: Current Pharmaceutical Design, 2016;22(12):1756-1766).
Dysregulation of the ECS owing to variation in the expression and function of cannabinoid receptors or enzymes or the concentration of endocannabinoids has been associated with several diseases, including cancer (Source: International Journal of Molecular Sciences, 2020;21(3):747). Indeed, the mechanisms involved in the regulation of the ECS as well as the processes that it regulates include practically every pathway important in cancer biology. Expression of the ECS is altered in numerous types of tumors, compared to healthy tissue, and this aberrant expression has been related to cancer prognosis and disease outcome, depending on the origin of the cancer (Source: British Journal of Pharmacology, 2018;175(13):2566-2580). Recent studies suggest that endocannabinoids contribute to maintaining balance in cell proliferation and that targeting the ECS can affect cancer growth (Source: Canadian Urological Association Journal, 2017;11(3-4):E138-E142).
Cannabinoids can interact with the cannabinoid receptors in the ECS, sometimes with a higher affinity than endocannabinoids. As a consequence, all the processes regulated by endocannabinoids are susceptible to interference by cannabinoids. The ability to use cannabinoids to modulate the ECS encompasses several attractive pharmacotherapeutic targets for systemic anti-cancer treatment and has sparked considerable research examining cannabinoid action on cancer cells (Source: Pharmacological Reviews, 2006;58(3):389-462).
| 7 |
Cannabinoids have demonstrated selective anti-tumor properties in preclinical studies, exerting anti-proliferative, proapoptotic, anti-angiogenic, and anti-metastatic and anti-inflammatory effects depending on tumor type and specific setting (Source: Cancer Medicine, 2018:7(3):765-775). These effects appear to be more pronounced when cannabinoids are used together versus being administered separately, a mechanism known as the entourage effect. We believe, therefore, that cannabinoid combinations may hold promise for an improved anti-proliferative strategy for cancer management.
In addition to their potential role as anti-cancer agents, cannabinoids have been observed to act synergistically with some conventional antineoplastic drugs, such as chemotherapeutic agents, enhancing their effectiveness (Source: Cancer Medicine, 2018;7(3)765-775). This raises the potential for combinational therapies that may increase the range of chemotherapeutic options available to patients and enable targeting of tumor progression at different levels while also permitting dosages of cytotoxic drugs to be dramatically reduced without compromising efficacy.
Figure 2: Synergistic effects of cannabis extracts and chemotherapies on cancer biopsy after treatment with the same extract and three different chemotherapy combinations
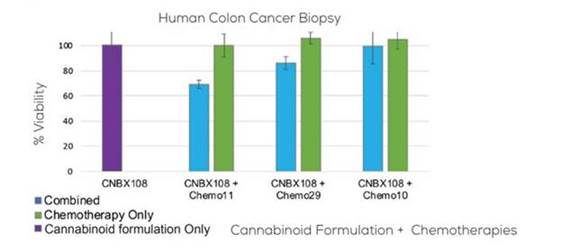
As of the date of this filing, we are not aware of any cannabinoid-based therapies approved for the anti-cancer treatment.
Our lead product candidate is RCC-33, which we are developing as a treatment for CRC. RCC-33 is an oral capsule containing a proprietary formulation of cannabinoids that have demonstrated synergistic efficacy in reducing the viability of human colon cancer cell lines in preclinical studies.
Colorectal Cancer
CRC is one of the more common forms of cancer worldwide, representing a significant challenge to the global healthcare system. According to the World Health Organization, CRC is the third most diagnosed cancer in the world and the second-leading cause of cancer-related mortality. In the United States, there were approximately 1,369,005 people living with CRC in 2019(Source: National Cancer Institute. “Cancer Stat Facts: Colorectal Cancer”). In 2022, an estimated 151,030 cases of colon cancer and 44,850 cases of rectal cancer will be diagnosed in the US, and a total of 52,580 people will die from these cancers (Source: American Cancer Society. “Cancer Facts & Figures 2022”).
Most CRCs begin as a noncancerous growth called a polyp that develops on the inner lining of the colon or rectum. The most common kind of polyp is called an adenomatous polyp or adenoma. According to the American Cancer Society, an estimated one-third to one-half of all individuals will eventually develop one or more adenomas. Although all adenomas have the capacity to become cancerous, fewer than 10% are estimated to progress to invasive cancer. The likelihood that an adenoma will evolve into cancer increases as it becomes larger or when it acquires certain histopathological characteristics. Adenomas that become cancerous, called adenocarcinomas, comprise nearly 96% of all CRCs (Source: American Cancer Society. “Colorectal Cancer Facts & Figures 2020-2022”). Adenocarcinomas may grow into blood vessels or lymph vessels, increasing the chance of metastasis to other anatomical sites.
| 8 |
CRC usually develops slowly, over a period of 10 to 20 years. The complex sequence of events occurring during initiation, development and propagation of adenocarcinomas is likely the result of a lifelong accumulation of mutations caused by both genetic and environmental factors known as the adenoma to carcinoma sequence. While the specific cause of any particular case of CRC is often unknown, more than one-half of all cases and deaths are attributable to lifestyle and environmental factors, such as smoking, unhealthy diet, high alcohol consumption, physical inactivity, and excess body weight (Source: American Cancer Society. “Cancer Facts & Figures 2020”).
CRC does not usually cause symptoms until the disease is advanced, therefore early detection of adenomas by screening is vital. If not treated or removed, an adenoma can become a potentially life-threatening cancer.
Current Standard of Care
Treatment options for CRC patients depend on several factors, including the type and stage of cancer, possible side effects, and the patient’s preferences and overall health. Surgical removal of the tumor is the most common form of treatment, particularly in the early stages of malignancy. Patients with more advanced stages of CRC may be given adjuvant chemotherapy to kill any cancer cells remaining after surgery, though standard chemotherapy is associated with severe side effects and provides marginal benefit to the majority of patients. While radiation therapy is often used to treat rectal cancer, it is not generally recommended for colon cancer patients except in the later stages of the disease (Source: American Cancer Society. “Treating Colorectal Cancer”).
CRC is a heterogeneous disease with distinct clinical, molecular, and pathophysiological characteristics. As a result, the response to treatment is variable between patients, even when they are diagnosed at the same clinical stage. Such heterogeneity remains an obstacle to the optimization of treatment for each individual. Researchers are continuing to investigate new treatment options, such as immunotherapy and targeted therapy, that focus upon the genes, proteins, and other factors in a particular tumor (Source: American Cancer Society. “Advances in Colorectal Research”).
Immunotherapy uses the body’s own immune system to kill cancer cells. There are already several FDA-approved immunotherapy options for CRC, such as pembrolizumab (Keytruda®), nivolumab (Opdivo®), and ipilimumab (Yervoy®). Many immunotherapies that have shown promise in addressing other types of cancer are also being tested for CRC. While immunotherapy has had some encouraging results, significant limitations remain. Its efficacy is often unpredictable, and the treatment can lead to the body becoming resistant or result in off-target toxicities where the body’s immune system attacks healthy tissue. Immunotherapy may take longer than other protocols and it is substantially more expensive than classical treatments (Source: Pharmacy & Therapeutics, 2017;42(8):514-521).
Targeted therapy uses drugs to target specific molecules inside cancer cells or on their surface to slow the growth of cancer, destroy cancer cells, and relieve cancer symptoms. There are different types of targeted therapy drugs, each working differently depending on what molecule the drug is targeting. A treatment is chosen based on the types of molecules expressed on the patient’s tumor cells, which allows doctors to tailor cancer treatment for each person. Several targeted therapy drugs, such as bevacizumab (Avasin®) and cetuximab (Erbitux®), are already used to treat advanced CRC. Despite showing clinical promise, targeted therapy has challenges, such as tumor heterogeneity, off-target toxicity, and acquired resistance (Source: Medical Research Journal, 2019;4(2):99-105). The lack of biomarkers by which to identify patients having a high probability of response is also a particularly significant obstacle. As with immunotherapy, the cost of targeted therapy is substantially higher than classical treatments.
We believe that there is no “magic bullet” to cure cancer and that a personalized combination of cancer treatments may be the best course for long term survival benefits in each case. To that end, the development of more prevention strategies and novel agents will be essential.
Cannabinoids and Colorectal Cancer
One area of increasing interest in the treatment of CRC lies in the development and use of cannabinoid therapeutics. The ECS is regarded as an important regulatory system in the gastrointestinal tract, being involved in several important functions such as motility, secretion, sensation, inflammation, and carcinogenesis. Recent studies advocate that the ECS plays a critical role in the development of CRC and should therefore be considered as an appropriate target for CRC inhibition (Source: Frontiers in Pharmacology, 2016;7:361). The expression of ECS components in CRC has been found to be increased and associated with poorer prognosis and advanced stages of disease (Source: Cannabis and Cannabinoid Research, 2018, 3(1):272-281). For example, cannabinoid receptors have been found to be overexpressed in tumor cells of the colon and this up-regulation has been postulated to be an indicator of cancer outcome (Source: British Journal of Pharmacology, 2018; 175(13): 2566-2580).
| 9 |
Research on the effects of cannabinoid compounds on CRC has demonstrated an ability to reduce the viability of CRC cell lines in vitro (Source: Cancer Medicine, 2018;7(3):765-775), while there is also convincing scientific evidence that cannabinoids are able to prevent or reduce carcinogenesis in different animal models of colon cancer (Source: Expert Review of Gastroenterology & Hepatology, 11:10, 871-873).
We believe that cannabinoids are a promising therapeutic agent for the treatment of CRC. We have conducted several in vitro unpublished studies using our bioinformatics platform to confirm that cannabinoids cause necrosis in colon cancer cells. While many cannabinoids demonstrate levels of toxicity on cancer cells, we have found that certain cannabinoid extracts and combinations show increased levels of toxicity relative to other isolated or combined cannabinoids. These findings have spurred the development of RCC-33, our product candidate for the treatment of CRC.
Figure 3: Synergistic effects of different cannabinoid combinations on viability of a colon cancer cell line.
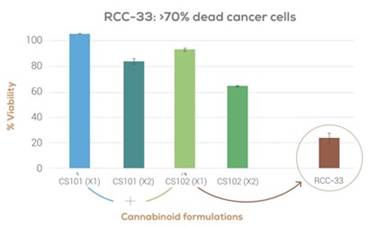
RCC-33
We are developing RCC-33 as an oral capsule or solution containing high concentrations of the cannabinoids CBDV and CBGA in a novel formulation, which we believe may be effective in the treatment of adenocarcinomas of the colon. The cannabinoids in RCC-33 have demonstrated complex synergistic anti-tumor effects in combination, with no psychoactive effect. In our preclinical in vitro studies evaluating the influence of 15 different cannabinoids on human colon cancer cell lines (RKO, HCT116), alone and in combination, RCC-33 demonstrated clear efficacy in reducing the viability of colon cancer cells versus alternative cannabinoid combinations. Importantly, we could detect significant reduction effect on tumour development in mice inoculated with human colorectal cancer cells.
| 10 |
Development Plan
The company is currently preparing to launch Phase I/II (a) clinical study in 2023, for the evaluation of its lead drug candidates Cannabics SR for the treatment of patients with advanced cancer and cancer anorexia cachexia syndrome (CACS) and RCC-33 for the treatment of colorectal cancer. We plan to conduct further preclinical studies to establish the safety and efficacy before proceeding with first-in-human clinical testing.
Preclinical Studies
We plan to conduct non-clinical safety studies following Good Laboratory Practice (GLP) to evaluate the systemic and local toxicity of escalating doses of RCC-33 and establish dosing parameters. The results of these preclinical studies, which are expected in 2023, will guide our planned Phase I/II(a) clinical trial. The non-clinical requirements to support the development program will be verified with the FDA at a pre-IND meeting. Such studies may include repeated dose toxicity studies, male and female fertility studies, embryofetal development studies, animal abuse related studies, pharmacokinetics studies, drug-drug interaction studies, and others.
Clinical Trials
We plan to evaluate the safety, tolerability, and pharmacokinetic properties of Cannabis SR and RCC-33 in a Phase I/II(a) ascending dose clinical trial in CRC patients, commencing in the first quarter 2023. The clinical trial will examine the tolerability, pharmacokinetics, pharmacodynamics, and efficacy of multiple doses of RCC-33 in CRC patients. We are currently identifying potential contract research organizations and clinical trial centers to conduct the Phase I/II(a) human proof of concept study, which is estimated to cost $6,500,000. As of the date of this filing, however, the Company does not have sufficient funds to complete the Phase I/II(a) study.
Subject to the results from our Phase I trials, we plan to submit an IND to the FDA for RCC-33 with the clinical protocol for a Phase II double-blind placebo controlled clinical trial evaluating RCC-33 in patients with CRC at various dosing levels versus placebo. The outcomes from the planned Phase II human proof of concept trial will inform our decision regarding further steps in the clinical development of RCC-33.
Our Pipeline:
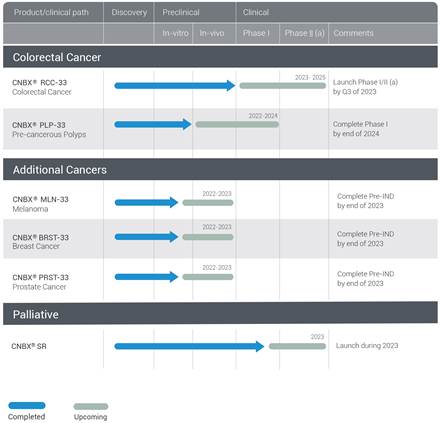
In addition to RCC-33, our colorectal cancer treatment drug candidate, the company has several other drug candidates under development, including PLP-33 for the local treatment of Lateral Spreading, or Sessile, colorectal polyps during colonoscopy, BRST-33 for the treatment of breast cancer, MLN-33 for the treatment of Melanoma and PRST-33 for the treatment of prostate cancer. These additional drug candidates are in the early stage of development and the company expects to complete the in-vivo research for each product by end of 2023. (see Fig. 4).
| 11 |
Fig. 4
2.3 Product lines currently not actively developed:
The company has several product lines that are currently not being actively developed following company’s decision to focus its resources and attention exclusively on the development of its FDA route drug candidates, and SR Capsule described above. The product lines not actively developed include:
Cannabics CDx (evaluate) Drug Sensitivity Test
Cannabics CDx is an ex-vivo drug sensitivity test under development to provide healthcare providers with clinical decision support data from which they can identify, for a particular cancer patient undergoing cannabinoid therapy, which cannabinoids or cannabinoid combinations may have the most beneficial anti-cancer effects, and which cannabinoids may be contraindicated.
Company may revisit this decision at a later stage after launching the first in human clinical studies for the validation of its colorectal cancer treatment drug candidate RCC-33.
| 12 |
3. Market opportunity for cancer treatment drug candidates:
3.1. Neoadjuvant therapy:
According to the National Cancer Institute, Neoadjuvant Therapy is a "treatment given as a first step to shrink a tumor before the main treatment, which is usually surgery”.
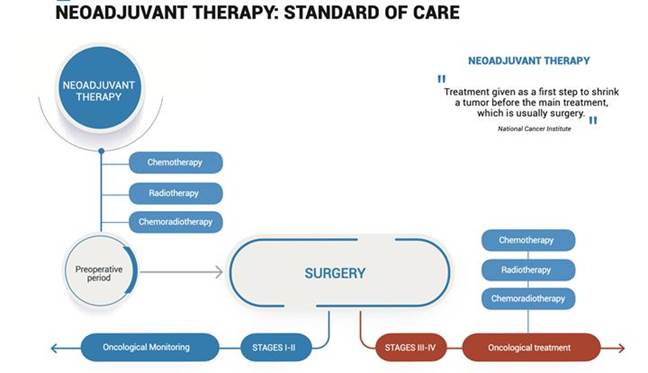
Limitations:
| · | Mild to severe side effects | |
| · | Suppressed immune system | |
| · | Potential resistance of tumor residues to postoperative chemotherapy * |
* “nCRT increases ITGH and may result in the expansion of resistant tumor cell populations in residual tumors”.
Frontiers in Oncology. 2019
The Effects of Neoadjuvant Chemoradiation in Locally Advanced Rectal Cancer—The Impact in Intratumoral Heterogeneity.
| 13 |
3.1.1 Neoadjuvant therapy in rectal cancer
Neoadjuvant chemoradiotherapy has become the standard treatment for locally advanced rectal cancer. Neoadjuvant chemoradiotherapy not only can reduce tumor size and recurrence, but also increase the tumor resection rate and anus retention rate with very slight side effect. Comparing with preoperative chemotherapy, preoperative chemoradiotherapy can further reduce the local recurrence rate and downstage. Middle and low rectal cancers can benefit more from neoadjuvant chemoradiotherapy than high rectal cancer.

3.1.2 Neoadjuvant therapy in breast cancer
In early breast cancer, surgery is the mainstay of curative treatment. Complementary local radiotherapy and systemic - adjuvant endocrine therapy or chemotherapy treatments are associated with the aim of reducing the risk of relapse according to the clinicopathological characteristics of the tumor. However, the possibility of administering these therapies prior to surgery in neoadjuvant setting offers several advantages:
| · | reduction in tumor size to improve respectability, | |
| · | increased rate of conservative surgery improving esthetic results, | |
| · | reduction in the extent of axillary surgery, | |
| · | early treatment of micrometastatic disease |
| 14 |
Fig5. Asco Guidelines for neoadjuvant therapy in breast cancer


According to ASCO guidelines most of the patients are eligible for neoadjuvant chemotherapy and are the end consumers of BRST-33, while the current treatment regimen negates severe side effects.
Side effects and risks of standard of care:
| · | nausea or vomiting | |
| · | hair loss | |
| · | nail or skin changes | |
| · | appetite loss | |
| · | weight changes | |
| · | diarrhea or constipation | |
| · | mouth sores | |
| · | fatigue |
| 15 |
3.2. Cannabinoid Neoadjuvant Therapy
For some time now, the FDA has promoted clinical studies on Cannabinoids as a growing range of stakeholders has expressed interest in development of drugs that contain cannabis and compounds found in cannabis. Recent legislative changes have also opened new opportunities for medical cannabis clinical research. As this body of research progresses and grows, the FDA is working to support drug development in this emerging scientific arena.
RCC-33 & BRST-33 – Potential safe drugs improving rectal and breast cancer neoadjuvant standard of care
RCC-33 & BRST-33 anticipated advantage over standard of care:
| · | Non-Suppressed immune system | |
| · | potential low toxicity which is even more important in neoadjuvant treatment since patients will suffer less side effects. Since the two drug candidates are based on two natural molecules (cannabinoids) found in the Cannabis plant, the safety of the molecules in the short and long run is potentially lower. Not like in a new drug entity in which toxicity could not be predicted. | |
| · |
Overcoming Potential resistance of tumor residues to postoperative chemotherapy |
3.3. Cancer Immunotherapy:
Our business strategy is to advance our programs through clinical studies with partners, and to opportunistically add programs in areas of high unmet medical needs through acquisition, collaboration, or internal development. Despite significant advances in oncology, there is still a large unmet need in treating patients with solid tumors, especially those with advanced disease who are refractory or unresponsive to currently available drugs. A business and clinical synergism of CNBX Pharmaceuticals and Tageza Pharmaceuticals is advancing towards unique therapeutics and optimizing their combination to expand oncology treatment options to cancer patients. The team work will be a unique collaboration of experienced scientist, physicians and exceptional healthcare research professionals. Within the partnership, CNBX Pharmaceuticals leverages it’s the cutting-edge infrastructure platform to support Tageza at various stages of drug development, including, target validation, compound validation and pre-clinical and clinical trial optimization. From Tageza Pharmaceutical partnership side, the clinical experience from one of the biggest Oncology Centers, access to data and insights as a result of large cohorts of patients during treatment, can be validated to determine patients who are most likely to benefit from novel cannabinoid treatments.
Patients undergoing most of currently existing treatments benefit a reduction in tumor size but often relapse and develop more aggressive disease. The residual population of chemotherapy-resistant tumor cells is enriched by Tumor Initiating Cells (TIC) previously known as cancer stem cells (CSC). The TaGeza technology is targeting TIC/ CSC by re- purposing small molecules and generating specific antibody generation for targeted immunotherapy ADC (Antibody drug conjugates).
CSCs were first found in colorectal cancer. Because of its heterogeneity, drug resistance etc., it was difficult to completely eliminate CSCs using traditional treatment methods. The remaining CSCs can cause tumor recurrence and invasion, and ultimately the death of the patient. Therefore, developing a therapy that can effectively target CSCs will greatly promote the treatment of GI cancers (Sun, Y., Li, B., Cao, Q. et al. Targeting cancer stem cells with polymer nanoparticles for gastrointestinal cancer treatment. Stem Cell Res Ther 13, 489 (2022). https://doi.org/10.1186/s13287-022-03180-9)
Antibody Drug Conjugates ADC
Antibody–drug conjugate (ADC) is a new emerging class of highly potent pharmaceutical drugs, which is a great combination of chemotherapy and immunotherapy. Antibody–drug conjugate (ADC) is typically composed of a monoclonal antibody (mAbs) covalently attached to a cytotoxic drug via a chemical linker. It combines both the advantages of highly specific targeting ability and highly potent killing effect to achieve accurate and efficient elimination of cancer cells, which has become one of the hotspots for the research and development of anticancer drugs. Since the first ADC, Mylotarg® (gemtuzumab ozogamicin), was approved in 2000 by the US Food and Drug Administration (FDA), there have been 14 ADCs received market approval so far worldwide. Moreover, over 100 ADC candidates have been investigated in clinical stages at present. This kind of new anti-cancer drugs, known as “biological missiles”, is leading a new era of targeted cancer therapy. The target antigen expressed on tumor cells is the navigation direction for ADC drugs to identify tumor cells and it also determines the mechanism (e.g., endocytosis) for the delivery of cytotoxic payloads into cancer cells. Hence, an appropriate selection of target antigen is the first consideration for the designation of ADC. In order to reduce off-target toxicity, the targeted antigen firstly should be expressed exclusively or predominantly in tumor cells, but rare or low in normal tissues.22 The antigen is ideally a surface (or extracellular) antigen (Fu, Z., Li, S., Han, S. et al. Antibody drug conjugate: the “biological missile” for targeted cancer therapy. Sig Transduct Target Ther 7, 93 (2022). https://doi.org/10.1038/s41392-022-00947-7) The ADC Market indicators
| 16 |
Global sales of currently marketed ADCs are forecast to exceed US$16.4 billion in 2026. The market for ADCs is set to grow further beyond 2026, as novel agents enter clinical practice across several oncology indications. https://www.nature.com/articles/d41573-021-
4. Outsourced GMP manufacturing and commercial operation:
4.1. Outsourced GMP manufacturing
Our current position is that all of our Chemistry Manufacturing and Controls (CMC) required for the approval process of our drug candidates is to be outsourced. The RCC-33 formulation, as well as all additional drug candidates in our pipeline, while inspired by natural molecules, could consist only of formulations made from chemically synthesized molecules, or APIs (Active Pharmaceutical Ingredients). Our Company is not engaged in the development of any botanical or botanically based product/s. Additionally, in view of our upcoming submission of a pre-IND meeting request with the FDA, the Company has entered an agreement with Purisys, a supplier of GMP (Good Manufacturing Practice) grade APIs suited for Clinical Stage Products. Purisys is a large and long-established US corporation with a long track record of working with the FDA. Accordingly, under said agreement, Purisys will also support CNBX throughout an IND filing process, including providing all necessary and related information concerning CMC in the form of a comprehensive technical package to be presented to the FDA. APIs supplied under said agreement will be used by Company in Phase I/II (a) clinical studies that it is planning to launch in 2023.
4.2. Commercial Operations
We have not established a sales, marketing, or product distribution infrastructure. We plan to commercialize any drugs we develop through licensing arrangements and strategic partnerships with established companies in the pharmaceutical industry having strong marketing capabilities and distribution networks. We generally intend to advance our drug candidates through Phase I and Phase II clinical trials as appropriate in order to establish their clinical and commercial potential before negotiating the terms of any licensing or collaboration. We believe that this approach will achieve the fullest marketing and distribution potential of any drugs or other products that we may develop in the short term.
5. Core activities:
5.1. Drug Discovery
Conduct all screening and pre-clinical research at in-house state of the art laboratory facilities
Our Research and Development:
To address these problems and improve clinical outcomes, CNBX Pharmaceuticals focuses on the development of diagnostics that monitor cancer progression and cannabinoid-cancer sensitivity tests to tailor treatment of cancer with cannabinoid medicine. Utilizing novel High-Throughput Screening (HTS) methods to perform studies on cancer cell lines and on circulating tumor cells (CTC) derived from cannabis medicated patients.
We aim to treat a wide scope of cancers both as the main treatment and as a conjugate to conventional chemotherapy. We believe a significant need remains for novel drugs for patients who do not respond to existing therapies or for whom these therapies bear undesirable side effects. We recognize the potential therapeutic applications of the synergistic effects of these active compounds thus building the methodology and procedures that decipher specific ratios of active compounds in regard to their antitumor activity.
Our government licensed laboratory operates a unique, custom designed and built research and development laboratory which combines high throughput screening, (HTS) capabilities with the most advanced data tools allowing us to enable miniaturization and automation of a variety of biological assays. The automated system is comprised of:
| 1. | High Content Screening (HCS) Platform, which is an automated cellular imaging and analysis platform designed for quantitative microscopy. |
| 2. | Flow Cytometry, which enables multi-parametric single cell analysis. |
| 3. | Automated workstation, for liquid handling for dispensing accurate and reproducible volumes of liquids and compounds. |
| 4. | Multimode microplate reader, designed for fast measurements of numerous biological reactions/processes. |
| 17 |
The integration of these instruments is enabled via a robotic arm, which allows a continuous process which utilizes all instruments.
Readouts generated from these instruments provide us with insights to the effect of our cannabinoid library on parameters such as, proliferation inhibition, apoptosis induction, angiogenesis prevention and toxicity on cancerous cells.
These experiments will produce multiplexed data composed of images of cells, cell specific markers and the extent/signal of the biological response. The biological response will be measured using different concentration of cannabinoids and their combinations, thus determining the most effective cannabinoid treatment for a specific cancer type.
In vitro Studies – Drug Screening
We have a proprietary procedure of high throughput screening (HTS) and high content screening (HCS) for the detection of correlations between cannabinoid ratios, dosages and anti-tumor activity using a growing library of human cancer cell lines and creating an enlarged variety of Cannabis-based compounds. We examine the biological activity of these compounds on tumor cell lines of distinct tissue lineage and creating a highly valuable therapeutic data. We Screen for the most potent cannabinoid/natural extracts. Our goal in the invitro studies is to build a library of both purified and natural cannabinoid extracts and to reveal their biological impact on a library of cancer cell lines. The HTS technology enables us to gain this data base in a faster manner and to reveal more mechanisms of action that are related to the genetics of the cancer. We are now in the process of merging our data with sophisticated data mining to help find meaningful insights of both treatment and outcome.
Our core technology is a continuously evolving bioinformatics platform that utilizes high-throughput screening technology, advanced data analytics, and proprietary methodologies to rapidly examine the physiologic effect of multiple cannabinoid compounds on tumor cells. This technology enables us to screen thousands of cannabinoid combinations, generating multiple datasets on the anti-tumor properties of different cannabinoid formulations and ratios. We conduct a broad range of preclinical research on cannabinoids through our bioinformatics platform, which informs the development of our product drug candidates.

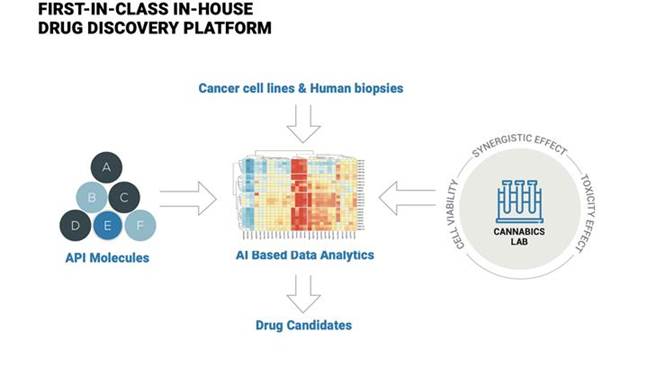
| 18 |
We have developed a continuously evolving preclinical bioinformatics platform that enables us to evaluate and classify the physiological impact of multiple cannabinoid compounds on various cancer cells. Utilizing state-of-the-art high-throughput screening and flow cytometry, our platform is capable of testing thousands of compounds weekly, allowing us to rapidly and effectively examine their interactions with a growing library of human cancer cell lines and biopsies. Through the large body of data generated by our platform, we are accumulating in-depth knowledge of the various therapeutic effects of cannabinoids and patterns of cannabinoid ratios that demonstrate meaningful physiologic impact on cancer.
Our bioinformatics platform includes the following:
| Ø | high-throughput screening, high content screening, flow cytometry, machine learning, robotics, and proprietary methodologies; |
| Ø | a library of human cancer cell lines and thousands of different combinations and ratios of cannabinoid compounds in a costumed matrix; |
| Ø | a growing database of biological response data; |
| Ø | in-house extraction, processing methodologies, and analytical techniques that yield well-characterized and standardized extracts; |
| Ø | collaborations with regulated cannabis producers that may expand our cannabinoid compound library and provide us with access for future proprietary cultivars; |
| Ø | fully integrated in-house research and development; and |
| Ø | regulatory expertise. |
Once a series of potentially active cannabinoids is identified for a specific cancer type, we then test and confirm their activity through in vitro and ex-vivo evaluation studies to determine their potential activity. Through this process, we are able to assess their therapeutic potential. The results of our pre-clinical experiments provide starting points for our clinical development programs.
5.2. Intellectual Property
Expand and protect the technology, inventions, and trade secrets that are critical for business development.
Intellectual Property
Our success depends in significant part on our ability to protect the proprietary nature of our Product Prospects, technology and know-how, to operate without infringing on the proprietary rights of others; and to defend challenges and oppositions from others and prevent others from infringing on our proprietary rights, including our provisional patents described below.
We plan to continue to seek patent protection in the United States and other countries for our proprietary technologies. To date, our intellectual property portfolio includes three provisional patents, filed with the USPTO, related to our line of activity (pharmaceutical formulations; drug delivery; therapeutic uses of cannabinoids and other cannabis compounds and personalized cannabinoid diagnostics), as well as know-how and trade secrets.
| 19 |
The Company’s current patent applications:
| Title | Application/Patent No. | Country | Status | Filing Date | ||||
| System and Method for High-throughput Screening of Cancer Cells |
Utility patent application no. 15/559,052 |
United States | Pending | 04/05/2016 | ||||
| System and Method for High-throughput Screening of Cancer Cells | Patent application no. 2983557 | Canada | Allowed | 04/05/2016 | ||||
| System and Method for High-throughput Screening of Cancer Cells | Patent application no. 201717032756 | India | Pending | 04/05/2016 | ||||
| System and Method for High-throughput Screening of Cancer Cells | Patent no. 254571 | Israel | Granted | 04/05/2016 | ||||
| System and Method for High-throughput Screening of Cancer Cells | Patent application no. 2016268526 | Australia | Granted | 04/05/2016 | ||||
| System and Method for High-throughput Screening of Cancer Cells | Regional patent no. 3304085 | Europe | Granted Validated in: France, Germany, Great Britain, Italy, Liechtenstein Netherlands, Switzerland |
04/05/2016 | ||||
| System and Method for High-throughput Screening of Cancer Cells | Patent application no. 1120170217678 | Brazil | Pending | 04/05/2016 | ||||
| System and Method for High-throughput Screening of Cancer Cells | Patent no. 382296 | Mexico | Granted | 04/05/2016 | ||||
| System and Method for High-throughput Screening of Cancer Cells | Patent no. 2017/06960 | South Africa | Granted | 04/05/2016 | ||||
| System and Method for High-throughput Screening of Cancer Cells | Patent no. 1253861 | Hong Kong | Granted | 04/05/2016 | ||||
| Cannabinoid Compositions, Methods of Manufacture and Use Thereof | Patent application no. 244278 | Israel | Pending | 24/02/2016 | ||||
| Cannabinoid Compositions, Methods of Manufacture and Use Thereof | Patent application no. 3054392 | Canada | Pending | 23/02/2017 | ||||
| Method for Sensitivity Testing of Cannabinoids on Patient-Derived Tumor Biopsies and CTCs | Patent application no. 273060 | Israel | Pending | 02/01/2018 | ||||
| Method for Sensitivity Testing of Cannabinoids on Patient-Derived Tumor Biopsies and CTCs | Patent application no. 16/644,393 | United States | Pending | 02/01/2018 | ||||
| Method for Sensitivity Testing of Cannabinoids on Patient-Derived Tumor Biopsies and CTCs | Patent application no. 3074819 | Canada | Pending | 02/01/2018 | ||||
| Novel System and Method for Microbiome Profiling and Modulation by Means of Cannabis Administration | Utility patent application no. 16/182,726 | United States | Pending | 07/11/2018 | ||||
| Composition and method for treating cancer with cannabinoids | Patent application no. 17/918,410 | United States | Pending | 13/04/2021 | ||||
| Composition and method for treating cancer with cannabinoids | Patent application no. 297246 | Israel | Pending | 13/04/2021 | ||||
| Composition and method for treating cancer with cannabinoids | Patent application no. 21789130.8 | Europe | Pending | 13/04/2021 | ||||
| Compositions Comprising Cannabinoids And Methods of Use Thereof in The Treatment of Cancer | PCT international patent application no. PCT/IL2022/051086 | WIPO | Pending | 12/10/2022 |
| 20 |
We anticipate that we will file additional patent applications in conjunction with our research, testing, and development of our cannabis-based Product Prospects.
Our policy is to seek patent protection for the technology, inventions and improvements that we consider important to the development of our business, but only in those cases where we believe that the costs of obtaining patent protection is justified by the scientific and commercial potential of the technology, and typically only in those jurisdictions that we believe present significant commercial opportunities. We anticipate that we will file additional patent applications in conjunction with our research, testing, and development of our cannabis-based products.
5.3. Scientific Advisory Board:
The development of our drug candidates is taking place with the close guidance and support of our clinical advisory team consisting of world-renowned experts, including doctors and scientists with specific expertise in the fields of cancer research, cancer treatment, Oncology, Hematology, Melanoma, Gastroenterology, AI and FDA regulation. Our team of experts is highly qualified and respected worldwide, some members having numerous publications in their respected names, including in high authority peer reviewed publications such as The New England Journal of Medicine and Nature, to name only a few.
Prof. Caroline Robert (MD)
Prof. Caroline Robert, M.D., Ph.D., is a world-renowned expert on Melanoma and is Head of the Dermatology Unit at Institut Gustave Roussy in Paris, France. She is also co-Director of the Melanoma Research Unit at INSERM 981 Paris-Sud University. Prof. Robert is the former Chair of the Melanoma group of the European Organization for the Research and Treatment of Cancer (EORTC), and is currently a Board member of the European Association of Onco-Dermatology (EADO), the European Society of Medical Oncology (ESMO), the European Association of Dermato-Venereology (EADV) the French Society of Dermatology and Venereology (SFD), the American Society of Oncology (ASCO) and the American Association of Clinical Research (AACR).
Prof. Amos Toren (MD)
Prof. Amos Toren is the Director of Pediatric Hemato-Oncology and BMT Department at the Sheba Medical Center since 2001 and a Professor in the division of Hematology, Sackler School of Medicine Tel-Aviv University. Prof. Toren is a specialist in Pediatrics, General Hematology and Pediatric Hemato-Oncology. He also has a PhD degree in genetics and qualified as a Master of Health Administration (MHA) at the Recannati Business School, Tel-Aviv University. Professor Toren runs numerous clinical studies, investigator initiated, company initiated, unicenter as well as multicenter. Prof. Amos Toren is the Director of Pediatric Hemato- Oncology and BMT department at the Sheba Medical Center since 2001 and a Professor in the division of Hematology, Sackler school of medicine tel-aviv university.
Prof. Zamir Halpern (MD)
Prof. Halpern, is a senior physician at the Gastroenterology Institute of the Sourasky Medical Center in Tel-Aviv, Israel, and current Chairman of the National Gastro Nutrition and Liver Diseases Council at the Israeli Ministry of Health. He has also served as Chairman of the Israeli Association of the Study of Liver, Chairman of the Israeli Gastroenterology Association and Chairman of the National Council for Food and Agriculture. Prof. Halpern, Senior Physician at the Gastroenterology institute in the Sourasky Medical Center in Tel-Aviv, Israel.
Prof. Noam Shomron (Ph.D.)
Professor Noam Shomron is passionate about using basic science to advance better healthcare. Professor Shomron heads the Functional Genomic Team at the Faculty of Medicine at Tel Aviv University, after training at MIT. Dr. Shomron leads a multidisciplinary team of scientists which develops computational methods for parsing big data in the bio-medical field using Artificial Intelligence. Shomron has over 30 patents and has published nearly 200 peer-reviewed publications on multiple genomic fields including medicine, agriculture, and business.
| 21 |
Dr. Sigal Tavor (MD)
Dr. Tavor currently serves as a Senior Physician at the Hemato Oncology Institute, Assuta Medical Center, Tel-Aviv, Israel. Dr. Tavor currently conducts gene Therapy research at the Weizmann Institute, Israel. Dr. Tavor is the former Acting Head of tthe Leukemia Unit, Department of Hematology and Bone Marrow Transplantation, Sourasky Medical Center, Tel Aviv. Dr. Tavor has two postdoctoral fellowships, at the Department of Hematology and Oncology, University of California Los Angeles, CA, and at the Department of Immunology, Weizmann Institute, Israel. Dr. Tavor has published extensively on Leukemia in peer reviewed journals and is a member of the Israel Society of Hematology and Blood Transfusion, the European Hematology Association (EHA), the American Society of Hematology (ASH), and the European Leukemia Network (ELN).
Dr. Yonina Tova (MD)
Dr. Yonina Tova (MD) is an internationally recognized Radiation Oncologist, certified by the American Board of Radiology (ABR), and currently serves as Director of the Radiotherapy Institute at Ziv Hospital in Israel. Dr. Tova obtained her Medical Degree from the College of Medicine, University of Illinoi, Chicago, in 1986, and specializes in treating cancer with radiation, using radiation therapy to treat a wide variety of cancers.
Dr. Erez Scapa (MD)
Dr. Scapa earned his M.D. in 2000 at the Technion Institute of Technology in Haifa, Israel, cum laude. He later held a Research Fellowship in Hepatology at the Brigham and Women's Hospital, Harvard University, Boston, Massachusetts, as well as a fellowship in Endoscopic Submucosal Dissection at the NTT Medical Center in Tokyo, Japan. He is an expert in invasive endoscopy and has extensive experience in preforming colonoscopies and gastroscopies. He is proficient in both diagnostic as well as invasive endoscopic ultrasound and has served as the head of the Endoscopic Submucosal Dissection program in Tel-Aviv Sourasky Medical Center since 2019. Dr. Scapa is a Senior Physician at the Gastroenterology institute in the Sourasky Medical Center in Tel-Aviv, Israel, who completed a Research Fellowship in Hepatology at the Brigham and Women’s Hospital, Harvard University.
Dr. Ilan Hochman, VP R&D
Dr. Hochman holds a PhD in cancer Immunology from the Sackler School of Medicine, Tel Aviv University. Dr. Hochman brings more than 20 years of experience in versatile R&D leadership and management positions in Innovative biotechnology and startup companies.
Dr. Lior Eshdat, VP IP
Dr. Eshdat holds a Ph.D. in organic chemistry and brings over 14 years of experience in intellectual property practice. Before he became an Israeli Patent Attorney Dr. Eshdat held a post-doctoral research fellowship at the University of Boulder and was a senior chemist in Dynamic Organic Light which developed OLED display technologies in Colorado.
Dr. Dana Ben-Ami Shor (MD)
Dr. Ben-Ami Shor is a recognized expert in invasive endoscopy and gastroenterology in the Sourasky Medical Center in Tel-Aviv, Israel. Dr. Ben-Ami Shor earned her M.D in 2009, graduating cum laude from the Sackler Faculty of Medicine, Tel Aviv University. She specialized in internal medicine and gastroenterology at the Sheba Medical Center, Israel. She also successfully completed an advanced endoscopy (ASGE) accredited fellowship within the Center for Interventional Endoscopy at AdventHealth, in Florida. Additionally, Dr. Ben-Ami Shor is proficient in both diagnostic and therapeutic endoscopic ultrasound (EUS), and endoscopic retrograde cholangiopancreatography (ERCP). Dr. Ben-Ami Shor is a recognized expert in invasive endoscopy and gastroenterology in the Sourasky Medical Center in Tel-Aviv, Israel.
| 22 |
Dr. Sigalit Arieli Portnoy (Ph.D.)
A Senior Advisor in the field of Regulation, Validation and Quality. Dr. Sigalit Ariely-Portnoy has over 17 years’ experience in the pharmaceutical industry. During this time, she has managed pharmaceutical and chemical plants at Taro pharmaceutical industries Ltd as Operation Group Vice president and in Teva Pharmaceutical industries Ltd as Kfar-Saba OSD plant manager. Dr. Ariely-Portnoy managed Teva's largest plant worldwide (9 billion tablets per annum and more than $2B revenues). During her career, she led more than 50 inspections by the US FDA, EMEA, Israeli MOH, and others. Dr. Ariely-Portnoy spearheaded the construction of a 200,000 sq ft pharmaceutical plant, several chemical plants and bio-warehouses, as well as many significant plant expansions for manufacturers of semisolids, liquids and oral solid dosage forms. Between the years 2003-2006, Dr. Ariely-Portnoy was the president of the Israel chapter of the PDA (Parenteral Drug Association). For the last 5 years, Dr. Ariely-Portnoy manages Gsap, a company which consults pharmaceutical, medical device and biotechnology companies in several major fields, including innovative product development, regulation, establishing quality systems and validation services. Dr. Ariely-Portnoy received her B.Sc., M.Sc., and D.Sc. from the Technion Institute of Technology in Haifa, Israel, in the fields of Chemical Engineering and Biomedical Engineering. Dr. Sigalit Ariely-Portnoy has vast experience in the pharmaceutical industry and managed pharmaceutical and chemical plants at Taro Pharmaceutical Industries LTD as Operation Group Vice President and in Teva Pharmaceutical industries LTD as OSD plant manager.
Prof. Benjamin Dekel (MD, PhD)
Prof. Dekel (MD, PhD) is the Head of the Israeli Stem Cell Society, and heads the Pediatric Nephrology Division and the Pediatric Stem Cell Research Institute at the Sheba Medical Center. He is also Associate Dean for Clinical Research Innovations and Development, and Chair of Nephrology and Director of the Center of Regenerative Medicine, at Tel Aviv University.
6. Competition
The biotechnology and pharmaceutical industries are characterized by rapidly advancing technologies, intense competition and a strong emphasis on proprietary products. We believe that our scientific knowledge, experience, technology and development capabilities provide us with competitive advantages, but we face competition from many different sources, including major pharmaceutical, specialty pharmaceutical and biotechnology companies, academic institutions, governmental agencies and public and private research institutions. Many of our current and potential competitors have longer operating histories and substantially greater financial, scientific, technical, intellectual property, regulatory and human resources than we have, as well as greater experience in developing and commercializing products, including obtaining FDA and other regulatory approvals.
As the medical use of cannabis increasingly receives government approval worldwide, we face growing competition from many new and existing companies seeking to develop cannabinoid-based therapies. Companies currently known to be developing cannabinoid-based human therapeutics include GW Pharmaceuticals PLC, Cannabis Science Inc., InMed Pharmaceuticals Inc., Emerald Bioscience Inc., Corbus Pharmaceuticals Holdings Inc., Zynerba Pharmaceuticals Inc., PharmaCyte Biotech, Inc., Tetra Bio-Pharma Inc. and Cure Pharmaceutical Holding Corp.
Many of our competitors are conducting research targeting the same technologies, applications, and markets as we are. Consequently, they may develop products for the same indications we are pursuing or may pursue in the future that are more effective, better tolerated, more widely-prescribed or accepted, more useful, and less costly. Any products that we successfully develop and commercialize will compete with existing products, as well as those currently in development or that may become available in the future.
In addition to competing for market position, we will also compete in terms of recruiting and retaining qualified personnel, acquiring intellectual property, establishing clinical trial sites, and enrolling patients for clinical trials and in obtaining funding.
Given the rapid changes affecting the global, national, and regional economies in general and cannabis-related medical research and development in particular, we may not be able to create and maintain a competitive advantage in the marketplace. Time-to-market is a critical factor in our industry and our success will depend on our ability to timely develop innovative technologies that will be accepted by patients. Our competitors may be better able to react to market changes, respond more rapidly to new regulations, or allocate greater resources to the development of their products than we can, which may result in our technologies and products becoming obsolete before we are able to enter the market, recover the expenses incurred to develop them, or generate significant revenue. Our success will depend, in part, upon our ability to develop our product candidates in a timely manner, keep our future products current with advancing technologies, achieve market acceptance of our future products, gain name recognition and a positive reputation in the healthcare industry, and establish successful marketing, sales, and distribution efforts. We cannot be certain that we will be able to compete against current or future competitors or that competitive pressure will not seriously harm our business prospects.
| 23 |
7. Competitive Factors
The Pharmaceutical industry is highly competitive and we will be competing with many other and better financed companies. We are a clinical stage and early stage-biotech pharmaceutical company, with relatively deminimis cash flow. We compete with other early-stage bio-tech and pharmaceutical companies for financing from a limited number of investors that are prepared to make investments in early-stage development companies. The presence of competing early-stage pharmaceutical companies may impact on our ability to raise additional capital in order to fund our research and development if investors are of the view that investments in competitors are more attractive based on their subjective analysis of our company, the general market conditions and the price of the investment offered to investors.
8. Regulations
CNBX Pharmaceuticals Inc. is purely a Bio-Technology Pharmaceutical company which licenses use of its Intellectual Property, it does not produce, manufacture or provide any product in any location. We are duly licensed by the Israeli Health Ministry for our research in Israel. Beyond the Israeli Health Ministry (by whom we are licensed) and the FDA regulatory pathway for our drug candidates as described above, we are not under the aegis of any Federal or State regulatory scheme as we have no manufacturing activity. Any licensee whom we engage must be duly licensed and certified according to all pertinent local government regulations in their jurisdiction. The Company is exploring drug development within approved laboratory clinical trial settings conducted within approved regulatory frameworks. Should any prescription drug product be developed by the Company, such drug product will not be commercialized prior to receipt of applicable regulatory approval, which will only be granted if clinical evidence of safety and efficacy for the intended use(s) is successfully developed.
United States
In the U.S., the FDA and other federal, state, local and foreign regulatory agencies impose substantial requirements upon the clinical development, approval, labeling, manufacture, marketing and distribution of drug products. These agencies regulate, among other things, research and development activities and the testing, approval, manufacture, quality control, safety, effectiveness, labeling, storage, record keeping, advertising and promotion of any prescription drug product candidates or commercial products. The regulatory approval process is generally lengthy and expensive, with no guarantee of a positive result. Moreover, failure to comply with applicable FDA or other requirements may result in civil or criminal penalties, recall or seizure of products, injunctive relief including partial or total suspension of production, or withdrawal of a product from the market.
Cannabis (other than hemp) is strictly controlled under the Controlled Substances Act (21 U.S.C. § 811) (the “CSA”) as a Schedule I substance. Schedule I substances by definition have no currently accepted medical use in the United States, a lack of accepted safety for use under medical supervision, and a high potential for abuse. Schedule I and II drugs are subject to the strictest controls under the CSA, including manufacturing and procurement quotas, security requirements and criteria for importation. Anyone wishing to conduct research on substances listed in Schedule I under the CSA must register with the U.S. Drug Enforcement Administration (the “DEA”), and obtain DEA approval of the research proposal. For any product containing cannabis to be available for commercial marketing in the United States, cannabis must be rescheduled, or the product itself must be scheduled, by the DEA to Schedule II, III, IV or V. Scheduling determinations by the DEA are dependent on FDA approval of a substance or a specific formulation of a substance.
The process required before a prescription drug product candidate may be marketed in the United States generally involves:
| · | completion of extensive non-clinical laboratory tests, animal studies and formulation studies, all performed in accordance with the FDA’s Good Laboratory, Good Clinical and/or Manufacturing Practice regulations; | |
| · | submission to the FDA of an IND, which must become effective before human clinical trials may begin; | |
| · | approval by an institutional review board or independent ethics committee at each clinical trial site before each trial may be initiated; | |
| · | for some products, performance of adequate and well-controlled human clinical trials in accordance with the FDA’s regulations, including Good Clinical Practices, to establish the safety and efficacy of the prescription drug product candidate for each proposed indication; | |
| · | submission to the FDA of a New Drug Application (“NDA”); | |
| · | Satisfactory completion of one or more FDA pre-approval inspections of the manufacturing facility or facilities where the drug will be produced to assess compliance with cGMP requirements to assure that the facilities, methods and controls are adequate to preserve the drug’s identity, strength, quality, and purity; and | |
| · | FDA review and approval of the NDA prior to any commercial marketing, sale or shipment of the drug. |
| 24 |
The testing and approval process requires substantial time, effort and financial resources, and the Company cannot be certain that any approvals for its prescription drug product candidates will be granted on a timely basis, if at all.
Non-clinical tests include laboratory evaluations of product chemistry, formulation and stability, as well as studies to evaluate toxicity in animals and other animal studies. The results of non-clinical tests, together with manufacturing information and analytical data, are submitted as part of an IND to the FDA. Some non-clinical testing may continue even after an IND is submitted. The IND also includes one or more protocols for the initial clinical trial or trials and an investigator’s brochure. An IND automatically becomes effective 30 days after receipt by the FDA, unless the FDA, within the 30-day time period, raises concerns or questions relating to the proposed clinical trials as outlined in the IND and places the clinical trial on a clinical hold. In such cases, the IND sponsor and the FDA must resolve any outstanding concerns or questions before any clinical trials can begin. Clinical trial holds also may be imposed at any time before or during studies due to safety concerns or non-compliance with regulatory requirements.
An independent institutional review board, at each of the clinical centers proposing to conduct the clinical trial, must review and approve the plan for any clinical trial before it commences at that center. An independent institutional review board considers, among other things, whether the risks to individuals participating in the trials are minimized and are reasonable in relation to anticipated benefits. The independent institutional review board also approves the consent form signed by the trial participants and must monitor the study until completed. The FDA, the independent institutional review board, or the sponsor may suspend or discontinue a clinical trial at any time on various grounds, including a finding that the subjects are being exposed to an unacceptable health risk. There also are requirements governing the reporting of ongoing clinical trials and completed clinical trials to public registries.
Clinical trials to support NDAs for marketing approval are typically conducted in three sequential phases, but the phases may overlap. In general in Phase 1, the initial introduction of the drug into healthy human subjects or patients, the drug is tested to assess metabolism, pharmacokinetics, pharmacological actions, side effects associated with increasing doses and, if possible, early evidence on effectiveness. Phase 2 usually involves trials in a limited patient population to determine the effectiveness of the drug for a particular indication, dosage tolerance and optimum dosage, and to identify common adverse effects and safety risks. If a compound demonstrates evidence of effectiveness and an acceptable safety profile in Phase 2 evaluations, Phase 3 trials are undertaken to obtain the additional information about clinical efficacy and safety in a larger number of patients, typically at geographically dispersed clinical trial sites, to permit the FDA to evaluate the overall benefit-risk relationship of the drug and to provide adequate information for the labeling of the drug. In most cases, the FDA requires two adequate and well-controlled Phase 3 clinical trials to demonstrate the efficacy of the drug. The FDA may, however, determine that a drug is effective based on one clinical study plus confirmatory evidence. Only a small percentage of investigational drugs complete all three phases and obtain marketing approval. In some cases, the FDA may require post-market studies, known as Phase 4 studies, to be conducted as a condition of approval in order to gather additional information on the drug’s effect in various populations and any side effects associated with long-term use. Depending on the risks posed by the drugs, other post-market requirements may be imposed.
After completion of the required clinical testing, an NDA is prepared and submitted to the FDA. The FDA approval of the NDA is required before marketing of the product may begin in the U.S. The NDA must include the results of all pre-clinical, clinical, and other testing and a compilation of data relating to the product’s pharmacology, chemistry, manufacture, and controls. The cost of preparing and submitting an NDA is substantial.
The FDA has 60 days from its receipt of an NDA to determine whether the application will be accepted for filing based on the agency’s threshold determination that it is sufficiently complete to permit substantive review. Once the submission is accepted for filing, the FDA begins an in-depth review. Under the statute and implementing regulations, the FDA has 180 days (the initial review cycle) from the date of filing to issue either an approval letter or a complete response letter, unless the review period is adjusted by mutual agreement between the FDA and the applicant or as a result of the applicant submitting a major amendment. In practice, the performance goals established pursuant to the Prescription Drug User Fee Act have effectively extended the initial review cycle beyond 180 days. The FDA’s current performance goals call for the FDA to complete review of 90 percent of standard (non-priority) NDAs within 10 months of receipt and within six months for priority NDAs, but two additional months are added to standard and priority NDAs for a new molecular entity, or NME.
| 25 |
The FDA may also refer applications for novel drug products, or drug products that present difficult questions of safety or efficacy, to an advisory committee, which is typically a panel that includes clinicians and other experts, for review, evaluation and a recommendation as to whether the application should be approved. The FDA is not bound by the recommendation of an advisory committee, but it generally follows such recommendations. Before approving an NDA, the FDA will typically inspect one or more clinical sites to assure compliance with GCP. Additionally, the FDA will inspect the facility or the facilities at which the drug is manufactured. The FDA will not approve the product unless compliance with current GMP is satisfactory and the NDA contains data that provide substantial evidence that the drug is safe and effective in the indication studied.
After the FDA evaluates the NDA and the manufacturing facilities, it issues either a n approval letter or a complete response letter. A complete response letter generally outlines the deficiencies in the submission and may require substantial additional testing, or information, in order for the FDA to reconsider the application. If, or when, those deficiencies have been addressed to the FDA’s satisfaction in a resubmission of the NDA, the FDA will issue an approval letter. The FDA has committed to reviewing 90 percent of resubmissions within two to six months depending on the type of information included.
An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications. As a condition of NDA approval, the FDA may require a risk evaluation and mitigation strategy, or REMS, to help ensure that the benefits of the drug outweigh the potential risks. REMS can include medication guides, communication plans for health care professionals, and elements to assure safe use, or ETASU. ETASU can include, but are not limited to, special training or certification for prescribing or dispensing, dispensing only under certain circumstances, special monitoring, and the use of patient registries. The requirement for a REMS can materially affect the potential market and profitability of the drug. Moreover, product approval may require substantial post-approval testing and surveillance to monitor the drug’s safety or efficacy. Once granted, product approvals may be withdrawn if compliance with regulatory standards is not maintained or problems are identified following initial marketing.
The FDA offers a number of regulatory mechanisms that provide expedited or accelerated approval procedures for selected drugs and indications which are designed to address unmet medical needs in the treatment of serious or life-threatening diseases or conditions. These include programs such as Breakthrough Therapy designations, Fast Track designations, Priority Review and Accelerated Approval, which the Company may need to rely upon in order to receive timely approval or to be competitive.
The Company may plan to seek orphan drug designation for certain indications qualified for such designation. Under the Orphan Drug Act, the FDA may grant orphan drug designation to drugs intended to treat a “rare disease or condition,” which, in the U.S., is generally a disease or condition that affects fewer than 200,000 individuals in the United States, or 200,000 or more individuals in the United States and for which there is no reasonable expectation that the cost of developing and making a drug available in the United States for this type of disease or condition will be recovered from sales of the product. Orphan drug designation must be requested before submitting an NDA. If a product that has an orphan drug designation subsequently receives the first regulatory approval for the indication for which it has such designation, the product is entitled to orphan exclusivity, meaning that the applicable regulatory authority may not approve any other applications to market the same drug for the same indication, except in very limited circumstances, for a period of seven years in the U.S. Orphan drug designation does not prevent competitors from developing or marketing different drugs for the same indication or the same drug for different indications. After orphan drug designation is granted, the identity of the therapeutic agent and its potential orphan use are publicly disclosed. Orphan drug designation does not convey an advantage in, or shorten the duration of, the development, review and approval process. However, this designation provides an exemption from marketing and authorization fees.
Drugs manufactured or distributed pursuant to FDA approvals are subject to continuing regulation by the FDA, including, among other things, requirements relating to recordkeeping, periodic reporting, product sampling and distribution, reporting of adverse experiences with the product, and complying with promotion and advertising requirements. The FDA may impose a number of post-approval requirements as a condition of approval of an NDA. For example, the FDA may require post-market testing, including phase IV clinical trials, and surveillance to further assess and monitor the product’s safety and effectiveness after commercialization. In addition, drug manufacturers and their subcontractors involved in the manufacture and distribution of approved drugs are required to register their establishments with the FDA and certain state agencies and are subject to periodic unannounced inspections by the FDA and certain state agencies for compliance with ongoing regulatory requirements, including current Good Manufacturing Practices, which impose certain procedural and documentation requirements. Failure to comply with statutory and regulatory requirements may subject a manufacturer to legal or regulatory action, such as warning letters, suspension of manufacturing, product seizures, injunctions, civil penalties or criminal prosecution. There is also a continuing, annual prescription drug product program user fee.
| 26 |
The FDA may withdraw approval if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, may result in revisions to the approved labeling to add new safety information, requirements for post-market studies or clinical trials to assess new safety risks, or imposition of distribution or other restrictions under a risk evaluation and mitigation strategy.
Controlled Substances
The CSA and its implementing regulations establish a “closed system” of regulations for controlled substances. The CSA imposes registration, security, recordkeeping and reporting, storage, manufacturing, distribution, importation and other requirements under the oversight of the DEA. The DEA is responsible for regulating controlled substances, and requires those individuals or entities that manufacture, import, export, distribute, research, or dispense controlled substances to comply with the regulatory requirements in order to prevent the diversion of controlled substances to illicit channels of commerce.
Facilities that manufacture, distribute, import or export any controlled substance must register annually with the DEA. The DEA registration is specific to the particular location, activity(ies) and controlled substance schedule(s). For example, separate registrations are needed for import and manufacturing, and each registration will specify which schedules of controlled substances are authorized.
The DEA inspects all manufacturing facilities to review security, recordkeeping, reporting and handling prior to issuing a controlled substance registration. The specific security requirements vary by the type of business activity and the schedule and quantity of controlled substances handled. The most stringent requirements apply to manufacturers of Schedule I and Schedule II substances. Required security measures commonly include background checks on employees and physical control of controlled substances through storage in approved vaults, safes and cages, and through use of alarm systems and surveillance cameras. Once registered, manufacturing facilities must maintain records documenting the manufacture, receipt and distribution of all controlled substances. Manufacturers must submit periodic reports to the DEA of the distribution of Schedule I and II controlled substances, Schedule III narcotic substances, and other designated substances. Registrants must also report any controlled substance thefts or significant losses, and must obtain authorization to destroy or dispose of controlled substances. Imports of Schedule I and II controlled substances for commercial purposes are generally restricted to substances not already available from a domestic supplier or where there is not adequate competition among domestic suppliers. In addition to an importer or exporter registration, importers and exporters must obtain a permit for every import or export of a Schedule I and II substance or Schedule III, IV and V narcotic, and submit import or export declarations for Schedule III, IV and V non-narcotics.
For drugs manufactured in the United States, the DEA establishes annually an aggregate quota for the number of substances within Schedules I and II that may be manufactured or produced in the United States based on the DEA’s estimate of the quantity needed to meet legitimate medical, scientific, research and industrial needs. The quotas apply equally to the manufacturing of the active pharmaceutical ingredient and production of dosage forms. The DEA may adjust aggregate production quotas a few times per year, and individual manufacturing or procurement quotas from time to time during the year, although the DEA has substantial discretion in whether or not to make such adjustments for individual companies.
Individual U.S. states also establish and maintain separate controlled substance laws and regulations, including licensing, recordkeeping, security, distribution, and dispensing requirements. State authorities, including boards of pharmacy, regulate use of controlled substances in each state. Failure to maintain compliance with applicable requirements, particularly as manifested in the loss or diversion of controlled substances, can result in enforcement action that could have a material adverse effect on the Company’s business, operations and financial condition. The DEA may seek civil penalties, refuse to renew necessary registrations, or initiate proceedings to revoke those registrations. In certain circumstances, violations could lead to criminal prosecution.
| 27 |
9. Employees
As of August 31, 2022, the Company had 5 employees, two of which were our Directors Gabriel Yariv and Eyal Barad, which along with our counsel and CFO were given monthly salaries. Aside from counsel, all employees reside in Israel.
Israeli labor laws principally govern the length of the workday, minimum wages for employees, procedures for hiring and dismissing employees, determination of severance pay, annual leave, sick days, advance notice of termination of employment, equal opportunity and anti-discrimination laws and other conditions of employment. Subject to certain exceptions, Israeli law generally requires severance pay upon the retirement, death or dismissal of an employee, and requires us and our employees to make payments to the National Insurance Institute, which is similar to the U.S. Social Security Administration. Our employees have defined benefit pension plans that comply with applicable Israeli legal requirements, which also include the mandatory pension payments required by applicable law and allocations for severance pay.
Item 1A. Risk Factors
Risk Factors
You should consider carefully the risks and uncertainties described below, together with all of the other information in this Annual Report on Form 10-K. If any of the following risks are realized, our business, financial condition, results of operations and prospects could be materially and adversely affected. The risks described below are not the only risks facing the Company. Risks and uncertainties not currently known to us or that we currently deem to be immaterial also may materially adversely affect our business, financial condition, results of operations and prospects.
Risks Related to Our Company and Business
Our independent auditors have expressed substantial doubt about our ability to continue operating as a going concern, which could prevent us from obtaining new financing on reasonable terms or at all.
Our independent registered public accountants have expressed substantial doubt about our ability to continue as a going concern. This opinion could materially limit our ability to raise additional funds by issuing new debt or equity securities or otherwise. If we fail to raise sufficient capital when needed, we will not be able to complete our proposed business. As a result we may have to liquidate our business and investors may lose their investments. Our ability to continue as a going concern is dependent upon our ability to successfully accomplish our plan of operations described in this filing, obtain financing and eventually attain profitable operations. Investors should consider our independent registered public accountant’s comments when deciding whether to invest in the Company.
We have not generated any significant revenue since our inception and we may never achieve profitability.
We are an early-stage biotechnology company and have not generated any significant revenue since we commenced our present operations in April 2014. At the present time, Cannabics SR is the only product that we have commercialized. To date, we have financed our operations primarily through private placements of common stock, warrants, and direct equity investments. As we continue our research and development of cannabinoid-based diagnostics, our expenses are expected to increase significantly. Accordingly, we will need to generate significant revenue to achieve profitability. Even as we begin to commercialize our technologies, we expect our losses to continue as a result of ongoing research and development expenses. These losses, among other things, have had and will continue to have an adverse effect on our working capital, total assets and stockholders’ equity. Because of the numerous risks and uncertainties associated with product development and commercialization efforts, we are unable to predict at what stage the Company will become profitable. We may never become profitable. Even if we do achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis. If we are unable to achieve and then maintain profitability, our business, financial condition and results of operations will be negatively affected, and the market value of our common stock will decline.
| 28 |
Since we have a limited operating history in our business, it is difficult for potential investors to evaluate our business.
We commenced operations as a biotechnology company in April 2014, and therefore have a relatively short operating history upon which an evaluation of our future success or failure can objectively be made. Our business is a highly speculative undertaking and involves a substantial degree of risk. We have not demonstrated an ability to successfully overcome many of the risks and uncertainties frequently encountered by early-stage companies in new and rapidly evolving competitive fields, including under-capitalization, cash shortages, limitations with respect to personnel, financial, and other resources and lack of revenue. The likelihood of our success must be considered in light of the early stage of our operations. There is no assurance that our business will ever be successful or that we will be able to attain profitability. Any failure by the Company to report profits may adversely affect the price of our common stock.
We will need to raise additional capital to meet our business requirements in the future, which may be costly or difficult to obtain and could dilute our stockholders’ ownership interests.
The Company has not yet generated significant revenue and will require additional capital to continue its research and development activities, conduct clinical trials, commercialize its products and otherwise fund its operations. Our ability to secure required financing will depend in part upon investor perception of our ability to create a successful business. Capital market conditions and other factors beyond our control may also play important roles in our ability to raise capital. There can be no assurance that debt or equity financing will be available or sufficient for our requirements or for other corporate purposes, or if debt or equity financing is available, that it will be on terms acceptable to us. Moreover, future activities may require us to alter our capitalization significantly. Our inability to access sufficient capital for our operations could have a material adverse effect on our financial condition, results of operations and prospects. If we are unable to obtain additional funding as needed, we may be required to reduce the scope of our research and development activities, which could harm our business plan, financial condition and operating results, or we may be required to cease our operations entirely, in which case, our investors will lose all of their investment.
Any additional capital raised through the sale of equity or equity-backed securities may dilute our stockholders’ ownership percentages and could also result in a decrease in the market value of our equity securities. The terms of any securities issued by us in future capital transactions may be more favorable to new investors, and may include preferences, superior voting rights and the issuance of warrants or other derivative securities, which may have a further dilutive effect on the holders of our securities then outstanding. Any debt financing secured in the future could involve restrictive covenants relating to capital raising activities and other financial and operational matters, which may make it more difficult for us to obtain additional capital and to pursue business opportunities.
In addition, we may incur substantial costs in pursuing future capital financing, including investment banking fees, legal fees, accounting fees, securities law compliance fees, printing and distribution expenses and other costs. We may also be required to recognize non-cash expenses in connection with certain securities we issue, such as convertible notes and warrants, which may have an adverse impact on our financial condition.
We are highly dependent on the success of cannabinoid technology, and we may not be able to develop the technology, successfully obtain regulatory or marketing approval for, or successfully commercialize, our products or product candidates.
Our business is focused entirely upon the research, development and commercialization of cannabinoid-based technologies for the detection and treatment of cancer. Our success is dependent upon the viability of this technology and the development of cancer diagnostics and therapies.
We have not received regulatory approval from the FDA to market any diagnostics or therapeutics based on botanical cannabinoids. The FDA has approved Epidiolex (CBD) oral solution for the treatment of seizures associated with two rare and severe forms of epilepsy, Lennox-Gastaut syndrome and Dravet syndrome, in patients two years of age and older. This is the first FDA-approved drug that contains a purified drug substance derived from cannabis. The FDA has also approved two drugs that contain a synthetic substance that acts similarly to cannabis compounds but is not present in the cannabis plant.
The scientific evidence underlying the feasibility of developing cannabinoid-based technologies for the detection and treatment of cancer is both preliminary and limited. In 2017, an ad hoc committee of the National Academies of Sciences, Engineering, and Medicine determined that while there is conclusive or substantial evidence that oral cannabinoids are effective antiemetics in the treatment of chemotherapy-induced nausea and vomiting, there was insufficient evidence to make any statement about the efficacy of cannabinoids as a treatment for cancer. The ad hoc committee went on to state that further clinical research into the anti-cancer effects of cannabinoids needs to be conducted.
| 29 |
If our cannabinoid technology is found to be ineffective or unsafe in humans, or if it never receives regulatory approval for commercialization, we may never be able bring our product candidates to market and may never become profitable. Further, our current business strategy, including all of our research and development, is focused on utilizing cannabinoid technology to detect and treat cancer. This lack of diversification increases the risk associated with the ownership of our common stock. If we are unsuccessful in developing and commercializing our cannabinoid-based technology and its application to the detection and treatment of cancer, we may be required to alter our scope and direction and steer away from the intellectual property we have developed as well as the core capabilities of our management team and advisory board. Without successful commercialization of our products and product candidates, we may never become profitable, which would have a material adverse effect on our business, results of operations and financial condition.
Our success depends upon our ability to retain our senior management and our ability to attract, retain and motivate other qualified personnel.
We are an early-stage biotechnology company. As of August 30, 2020, we had five employees and several key consultants. Our success materially depends upon the efforts of our management and other key personnel, including but not limited to Dr Sanja Goldberg, our Chief Technology Officer. If we lose the services of Dr. Goldberg’s or any other executive officers or significant employees, our business would likely be materially and adversely affected. At this time, we do not currently have “key man” life insurance for or any other executive officer.
Because of the specialized scientific and managerial nature of our business, we rely heavily on our ability to attract and retain qualified scientific, technical and managerial personnel. The competition for qualified personnel in the biotechnology industry is intense. Due to this intense competition, we may be unable to continue to attract and retain qualified personnel necessary for the development of our business or to recruit suitable replacement personnel. Any difficulties in obtaining and retaining qualified officers, employees and consultants could have a material adverse effect on our operations.
The relative lack of public company experience by our management team may put us at a competitive disadvantage.
As a company with a class of securities registered under the United States Securities Exchange Act of 1934, as amended (the “Exchange Act”), we are subject to reporting and other legal, accounting, corporate governance, and regulatory requirements imposed by the Exchange Act and rules and regulations promulgated under the Exchange Act. With the exception of our CFO, Uri Ben-Or, our management team lacks significant public company experience, which could impair our ability to comply with these legal, accounting, and regulatory requirements. Such responsibilities include complying with securities laws and making required disclosures on a timely basis. Our senior management may not be able to implement and effect programs and policies in an effective and timely manner that adequately respond to such increased legal and regulatory compliance and reporting requirements. Our failure to do so could lead to the imposition of fines and penalties and further result in the deterioration of our business.
If we are unable to enter into acceptable sales, marketing and distribution arrangements with third parties or establish sales, marketing and distribution capabilities, we may not be successful in commercializing any product candidate that we develop if and when a product candidate is approved.
We do not have any sales, marketing or distribution infrastructure and have no experience in the commercialization of biotechnology. To achieve commercial success for any product, we must develop a sales and marketing organization, outsource these functions to third parties or license our products to others.
In the United States, we intend to only commercialize our products by licensing them to organizations having greater resources and experience than we do. While we have already licensed our Cannabics SR medical cannabis capsules in Colorado to Mountain High Products LLC, and in states outside of Colorado to the Cima Group LLC, there can be no assurance that such licensing efforts will be successful, or that we will be able to license any future products on satisfactory terms, or at all. We do not presently have any other agreement or arrangement for the commercialization of our products in the United States or elsewhere.
While we generally intend to adopt a licensing model for the commercialization of our products, we may also seek one or more strategic partners for commercialization of our products outside the United States. As a result of entering into arrangements with third parties to perform sales, marketing and distribution services, our product revenue or the profitability of our product revenue may be lower, perhaps substantially lower, than if we were to directly market and sell products in those markets. Furthermore, we may be unsuccessful in entering into the necessary arrangements with third parties or may be unable to do so on terms that are favorable to us. In addition, we may have little or no control over such third parties and any of them may fail to devote the necessary resources and attention to sell and market our product candidates effectively.
| 30 |
If we do not license our products or outsource our commercialization efforts, we will be required to develop our own sales, marketing and distribution capabilities, which will require substantial resources and will be time-consuming, and could delay any product launch. Moreover, we may not be able to hire or retain a sales force that is sufficient in size or has adequate expertise in the consumer health markets that we plan to target. If we are unable to establish or retain a sales force and marketing and distribution capabilities, our operating results may be adversely affected.
If we do not successfully license our products or establish sales and marketing capabilities, either on our own or in collaboration with third parties, it is likely that we will be unable to commercialize any of our products.
We face intense competition, often from companies with greater resources and experience than we have, which may result in others developing or commercializing competing products before us or more successfully.
The market for cancer diagnostics and therapies is intensely competitive, subject to rapid change and significantly affected by new product introductions and other market activities of industry participants. Our competitors include large multinational corporations and their operating units, including Abbott Laboratories Inc., Cepheid Inc., Philips, GE Healthcare, Siemens, Gen-Probe Incorporated, MDxHealth SA, EpiGenomics AG, Roche Diagnostics, Exact Sciences Corporation, Sequenom, Inc. and several others. We also compete against pharmaceutical companies, specialty pharmaceutical companies and biotechnology companies worldwide, as well as smaller and other early-stage companies. Other potential competitors include academic institutions, government agencies and other public and private research organizations that conduct research, seek patent protection and establish collaborative arrangements for research, development, manufacturing and commercialization.
Many of our competitors and potential competitors have or will have substantially greater financial, technological, managerial and research and development resources and experience than we have, and many have been engaged in the biotechnology industry for a much longer time than we have. Many of our competitors spend significantly more funds on research, development, promotion and sale of new and existing products than we do, and may therefore be able to react more quickly to new or emerging technologies, shifting market conditions and regulatory changes.
There can be no assurance that any of our current or future products and technologies will have a competitive advantage in the marketplace, or that they will remain competitive following the introduction of competing products or technologies. Our commercial opportunity could be reduced or eliminated if our competitors develop and commercialize products that are safer, more effective, more convenient or less expensive. There can be no assurance that we will be successful in the face of increasing competition from new technologies or products introduced by existing companies in the industry or by new companies entering the market.
If we are unable to compete successfully, there may be a material and adverse effect on our business, financial condition and results of operations.
If the marketplace does not accept the products in our development pipeline or any other diagnostic products we might develop, we may be unable to generate sufficient revenue to sustain and grow our business.
Even if we are able to successfully develop and obtain regulatory approval of a product candidate, our ability to generate significant revenue will depend on the acceptance of our products by physicians and patients. Physicians, hospitals, clinical laboratories, researchers or others in the healthcare industry may not use our current or future diagnostic product candidates unless they are determined to be an effective and cost-efficient means of detecting and diagnosing cancer. Market acceptance of our current or future therapeutic products will depend on a number of factors, including the indication statement and warnings approved by regulatory authorities in the product label, continued demonstration of efficacy and safety in commercial use, physicians’ willingness to prescribe the product, reimbursement from third-party payers such as government healthcare systems and insurance companies, the price of the product, the nature of any post-approval risk management plans mandated by regulatory authorities, competition, marketing and distribution support. In addition, we will need to expend a significant amount of resources on marketing and educational efforts to create awareness of our products and to encourage their acceptance and adoption. If the market for our products does not develop sufficiently or the products are not accepted, our revenue potential will be harmed.
| 31 |
We are exposed to potential product liability risks that are inherent in the testing, manufacturing and marketing of cancer diagnostics, pharmaceuticals and dietary supplements.
While we do not presently carry any product liability insurance coverage, we intend to obtain such insurance in amounts we believe to be commercially reasonable for our current level of activity and exposure. There is no assurance, however, that we will be able to obtain or maintain insurance coverage that will be adequate to cover our potential liabilities, or that premiums will be commercially justifiable. Furthermore, insurance that might otherwise be readily available, may be more difficult for us to find and more expensive because we work with medicinal cannabis. If we are the subject of a successful product liability claim that exceeds the limits of, or is not otherwise covered by our insurance, or if we incur such liability at a time when we are not able to obtain liability insurance, we may incur substantial charges that adversely affect our earnings and require the commitment of capital resources that might otherwise be available for the development and commercial launch of our product programs.
If we fail to protect our intellectual property rights, our ability to pursue the development of our technologies and products would be negatively affected.
Our success will depend in part on our ability to protect our intellectual property. This is done, in part, by obtaining patents and trademarks and then maintaining adequate protection of our technologies, tradenames and products. If we do not adequately protect our intellectual property, competitors may be able to use our technologies to produce and market products in direct competition with us and erode our competitive advantage. Some foreign countries lack rules and methods for defending intellectual property rights and do not protect proprietary rights to the same extent as the United States. Many companies have had difficulty protecting their proprietary rights in these foreign countries. We may not be able to prevent misappropriation of our proprietary rights.
We are currently seeking patent protection for several processes and finished products. However, the patent process is subject to numerous risks and uncertainties, and there can be no assurance that we will be successful in protecting our products by obtaining and defending patents. These risks and uncertainties include the following:
| · | patents that may be issued or licensed may be challenged, invalidated, or circumvented, or otherwise may not provide any competitive advantage; |
| · | our competitors, many of which have substantially greater resources than us and many of which have made significant investments in competing technologies, may seek, or may already have obtained, patents that will limit, interfere with, or eliminate our ability to make, use, and sell our products and product candidates either in the United States or in international markets; |
| · | there may be significant pressure on the United States government and other international governmental bodies to limit the scope of patent protection both inside and outside the United States for treatments that prove successful as a matter of public policy regarding worldwide health concerns; |
| · | countries other than the United States may have less restrictive patent laws than those upheld by United States courts, allowing foreign competitors the ability to exploit these laws to create, develop, and market competing products. |
Any patents issued to us may not provide us with meaningful protection, and third parties may challenge, circumvent or narrow them. Third parties may also independently develop products similar to our products or product candidates, duplicate our unpatented product or product candidates, and design around any patents on product candidates we may develop.
Additionally, extensive time is required for development, testing and regulatory review of product candidates. While extension of a patent term due to regulatory delays may be available, it is possible that before any of our product candidates can be commercialized, any related patent, even with an extension, may expire or remain in force for only a short period following commercialization, thereby reducing any advantages of the patent.
In addition, the United States Patent and Trademark Office (the “USPTO”), and patent offices in other jurisdictions have often required that patent applications concerning biotechnology-related inventions be limited or narrowed substantially to cover only the specific innovations exemplified in the patent application, thereby limiting the scope of protection against competitive challenges. Thus, even if we or our licensors are able to obtain patents, the patents may be substantially narrower than anticipated.
| 32 |
In addition to patents, we rely on a combination of trade secrets, confidentiality, nondisclosure and other contractual provisions, and security measures to protect our confidential and proprietary information. These measures may not adequately protect our trade secrets or other proprietary information. If they do not adequately protect our rights, third parties could use our technology, and we could lose any competitive advantage we may have. In addition, others may independently develop similar proprietary information or techniques or otherwise gain access to our trade secrets, which could impair any competitive advantage we may have.
Costly litigation may be necessary to protect our intellectual property rights and we may be subject to claims alleging the violation of the intellectual property rights of others.
We may face significant expense and liability as a result of litigation or other proceedings relating to patents and other intellectual property rights of others. If another party has also filed a patent application or been issued a patent relating to an invention or technology claimed by us in pending applications, we may be required to participate in an interference proceeding declared by the USPTO to determine priority of invention, which could result in substantial uncertainties and costs, even if the eventual outcome were favorable to us. We could also be required to participate in interference proceedings involving issued patents and pending applications of another entity. An adverse outcome in an interference proceeding could require us to cease using the technology or to license rights from prevailing third parties.
The cost to us of any patent litigation or other proceeding relating to our patents or patent applications, even if resolved in our favor, could be substantial. Our ability to enforce our patent protection could be limited by our financial resources, and may be subject to lengthy delays.
A third party might claim that we are using inventions claimed by their patents and might go to court to stop us from engaging in our normal operations and activities, such as research, development and the sale of any future products. Such lawsuits are expensive and would consume time and other resources. There is a risk that the court will decide that we are infringing the third party’s patents and will order us to stop the activities claimed by the patents, redesign our products or processes to avoid infringement or obtain licenses (which may not be available on commercially reasonable terms). In addition, there is a risk that a court will order us to pay the other party damages for having infringed their patents.
There is no guarantee that any prevailing patent owner would offer us a license so that we could continue to engage in activities claimed by the patent, or that such a license, if made available to us, could be acquired on commercially acceptable terms. In addition, third parties may, in the future, assert other intellectual property infringement claims against us with respect to our products, technologies or other matters.
Failure in our information technology or storage systems could significantly disrupt our operations and our research and development efforts, which could adversely impact our revenues, as well as our research, development and commercialization efforts.
Our ability to execute our business strategy depends, in part, on the continued and uninterrupted performance of our information technology (“IT”), systems, which support our operations and our research and development efforts, as well as our storage systems. Due to the sophisticated nature of the technology we use in our products and service offerings, we are substantially dependent on our IT systems. IT systems are vulnerable to damage from a variety of sources, including telecommunications or network failures, malicious human acts and natural disasters. Moreover, despite network security and back-up measures, some of our servers are potentially vulnerable to physical or electronic break-ins, computer viruses and similar disruptive problems. Despite the precautionary measures we have taken to prevent unanticipated problems that could affect our IT systems, sustained or repeated system failures that interrupt our ability to generate and maintain data, could adversely affect our ability to operate our business.
We will need to grow the size of our organization, and we may experience difficulties in managing any growth we may achieve.
As of the date of this filing, we have two full-time employees. As our development and commercialization plans and strategies progress, we expect to need additional research, development, managerial, operational, sales, marketing, financial, accounting, legal and other resources. Future growth would impose significant added responsibilities on our management, which may not be able to accommodate those added responsibilities. If we fail to effectively manage our future growth, it could delay the execution of our business plan and disrupt our operations.
| 33 |
We are subject to financial reporting and other requirements that place significant demands on our resources.
We are subject to reporting and other obligations under the Securities Exchange Act of 1934, as amended, including the requirements of Section 404 of the Sarbanes-Oxley Act of 2002. Section 404 requires us to conduct an annual management assessment of the effectiveness of our internal controls over financial reporting. These reporting and other obligations place significant demands on our management, administrative, operational, internal audit and accounting resources. The costs of preparing and filing annual and quarterly reports, proxy statements and other information with the SEC and furnishing audit reports to stockholders causes our expenses to be higher than they would be if we remained a privately-held company. The increased costs associated with operating as a public company may decrease our net income or increase our net loss, and may cause us to reduce costs in other areas of our business or increase the prices of our product to offset the effect of such increased costs. Additionally, if these requirements divert our management’s attention from other business concerns, they could have a material adverse effect on our business, financial condition and results of operations.
Management assessed the effectiveness of the Company’s internal controls over financial reporting as of August 31, 2022, based on the framework in Internal Control—Integrated Framework (COSO 2013) issued by the Committee of Sponsoring Organization of the Treadway Commission. On the basis of that assessment, management determined that our internal controls over financial reporting were effective as of that date.
Effective internal controls are necessary for us to provide reasonable assurance with respect to our financial reports and to effectively prevent fraud. If we cannot provide reasonable assurance with respect to our financial reports and effectively prevent fraud, our reputation and operating results could be harmed. Pursuant to the Sarbanes-Oxley Act of 2002, we are required to furnish a report by management on internal control over financial reporting, including management’s assessment of the effectiveness of such control. Internal control over financial reporting may not prevent or detect misstatements because of its inherent limitations, including the possibility of human error, the circumvention or overriding of controls, or fraud. Therefore, even effective internal controls can provide only reasonable assurance with respect to the preparation and fair presentation of financial statements. In addition, projections of any evaluation of effectiveness of internal control over financial reporting to future periods are subject to the risk that the control may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate. If we fail to maintain the adequacy of our internal controls, including any failure to implement required new or improved controls, or if we experience difficulties in their implementation, our business and operating results could be adversely impacted, we could fail to meet our reporting obligations, and our business and stock price could be adversely affected.
At August 31, 2022, our Chief Executive Officer and Chief Financial Officer evaluated the effectiveness of the design and operation of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act) and concluded that, subject to the inherent limitations identified in Item 9A of Part II of our Annual Report on Form 10-K for the fiscal year ended August 31, 2022, our disclosure controls and procedures were effective as of that date. Our independent auditors issued an adverse attestation report regarding the effectiveness of our internal control over financial reporting as of August 31, 2022.
Since that time, we have implemented a remediation plan to address the previously reported material weakness in internal control over financial reporting, described in Part II, Item 9A, "Controls and Procedures" in our Annual Report on Form 10-K for the fiscal year ended August 31, 2022. Remediation measures include adding additional resources in our finance function, addition of qualified financial professionals and formation of our Audit Committee. The material weakness will not be considered remediated, until the applicable controls operate for a sufficient period of time and management has concluded, through testing, that these controls are operating effectively. We expect that the remediation of this material weakness will be completed prior to the end of fiscal year 2022. Notwithstanding the material weakness, we believe the financial statements in this report fairly present, in all material respects, our financial position, results of operations, and cash flows for the periods presented in conformity with U.S. GAAP.
Risks Related to Cannabis
Our failure to comply with controlled substance legislation could restrict or harm our ability to develop and commercialize our products.
Our business is, and will be, subject to wide-ranging laws and regulations of Israel, the United States (federal and state), the European Community and other governments in each of the countries where we may develop and market our products. We must comply with all regulatory requirements if we expect to be successful.
| 34 |
Most countries are parties to the Single Convention on Narcotic Drugs of 1961 as amended by the 1972 Protocol, which governs international trade and domestic control of narcotic substances, including cannabis extracts. Countries may interpret and implement their treaty obligations in a way that creates a legal obstacle to us obtaining marketing approval in those countries for any cannabinoid-based products we develop. These countries may not be willing or able to amend or otherwise modify their laws and regulations to permit our products to be marketed, or achieving such amendments to the laws and regulations may take a prolonged period of time. In the case of countries with similar obstacles, we would be unable to market our product candidates in countries in the near future or perhaps at all if the laws and regulations in those countries do not change.
Any cannabinoid-based product candidate that we may develop for use in the United States, will be subject to U.S. controlled substance laws and regulations that will require us, along with our collaborators and licensees, to expend time, money and effort in all areas of regulatory compliance, including, if applicable, manufacturing, production, quality control and assurance and clinical trials. Any failure to comply with these laws and regulations, or the cost of compliance with these laws and regulations, could adversely affect the results of our business operations and our financial condition.
The constant evolution of laws and regulations affecting the research and development of cannabis-based diagnostics and therapies could detrimentally affect our business. Laws and regulations related to the therapeutic uses of cannabis are subject to changing interpretations. These changes may require us to incur substantial costs associated with legal and compliance fees and ultimately require us to alter our business plan. Furthermore, violations or alleged violation of these laws could disrupt our business and result in a material adverse effect on our operations, including our ability to conduct clinical trials that are prerequisite to our ability to commercialize our cannabis-based medical products and therapies. We cannot predict the nature of any future laws, regulations, interpretations or applications of laws and regulations and it is possible that new laws and regulations may be enacted in the future that will be directly applicable to our business.
Cannabis remains illegal under U.S. federal law, and any change in the enforcement priorities of the federal government could render our current and planned future operations unprofitable or even prohibit such operations.
We are a biotechnology company focused on the research and development of cannabinoid-based diagnostics, anti-cancer pharmaceuticals and palliative therapies. The commercial viability of our products and technologies in the United States depends, in part, on state laws and regulations; however, Cannabis remains illegal under US federal law.
The United States federal government regulates drugs through the Controlled Substances Act of 1970 (the “CSA”), which places controlled substances, including cannabis, on one of five schedules. Cannabis is currently classified as a Schedule I controlled substance, which is viewed as having a high potential for abuse and no currently accepted medical use in treatment in the United States. No prescriptions may be written for Schedule I substances, and such substances are subject to production quotas, security requirements and criteria for importation imposed by the United States Drug Enforcement Administration (the “DEA”). Because of this, doctors may not prescribe cannabis for medical use under federal law, although they can recommend its use under the First Amendment.
Currently, thirty-seven U.S. states and the District of Columbia allow the use of medical cannabis. Eighteen states and the District of Columbia also allow its recreational use. Because cannabis is a Schedule I controlled substance, however, the development of a legal cannabis industry under the laws of these states is in conflict with the Federal Controlled Substances Act, which makes cannabis use and possession illegal on a national level. The United States Supreme Court has confirmed that the federal government has the right to regulate and criminalize cannabis, including for medical purposes, and that federal law criminalizing the use of cannabis pre-empts state laws that legalize its use.
In 2014, the United States House of Representatives passed an amendment (the “Rohrabacher-Farr Amendment”) to the Commerce, Justice, Science, and Related Agencies Appropriations Bill, which funds the United States Department of Justice (the “DOJ”). The Rohrabacher-Farr Amendment prohibits the DOJ from using funds to prevent states with medical cannabis laws from implementing such laws. In August 2016, a Ninth Circuit federal appeals court ruled in United States v. McIntosh that the Rohrabacher-Farr Amendment bars the DOJ from spending funds on the prosecution of conduct that is allowed by state medical cannabis laws, provided that such conduct is in strict compliance with applicable state law. While the Rohrabacher-Farr Amendment has been included as a rider to the Consolidated Appropriations Acts every year since 2014, there can be no assurances that the Rohrabacher-Farr Amendment will be included in future appropriations bills or budget resolutions. The federal government could at any time change its enforcement priorities against the cannabis industry. We do not grow or distribute cannabis, but our current and planned business operations may involve licensing cannabinoid-based products and technology. Any change in enforcement priorities could render such operations unprofitable or even prohibit such operations.
| 35 |
Our ability to earn revenue through licensing our product in the United States is dependent on additional states legalizing medical marijuana.
We are engaged in the business developing and commercializing cannabinoid-based products for the detection and treatment of cancer. Our ability to commercialize our products in the United States is dependent upon the continued progress of legislative authorization of cannabis at the state level for medical purposes and, in certain states, based on the specifics of the legislation passed in that state. Any number of factors could slow or halt the progress. Furthermore, progress, while encouraging, is not assured. The legislative process normally encounters set-backs before achieving success. While there may be ample public support for legislative proposals, there must be political will in the legislative committee or a bill may never advance to a vote. Numerous factors impact the legislative process. Any one of these factors could slow or halt the progress and adoption of cannabis for medical purposes, which would limit the market for our products and negatively impact our business and revenues.
Changes in consumer preferences and acceptance of medical cannabis, or any negative trends, will adversely affect our business.
Our business is substantially dependent on market acceptance of medical cannabis. Market perception of medical cannabis can be significantly influenced by a number of social, political and economic factors that are beyond our control, including scientific research or findings, regulatory investigations, litigation, media attention and other publicity regarding such products and treatments. There can be no assurance that future scientific research, findings, regulatory proceedings, litigation, media attention or other research findings or publicity will be favorable to the market for any of our current or future cannabinoid-based diagnostics or therapies. Future research reports, findings, regulatory proceedings, litigation, media attention or other publicity that are perceived as less favorable than, or that question, earlier research reports, findings or publicity could have a material adverse effect on the demand for our products, as well as our business, results of operations, financial condition and cash flows.
We believe that as cannabis-based biotechnology becomes more widely accepted by the U.S. medical community and the public at large, the stigma associated with medical cannabis will moderate and, as a result, consumer demand will likely continue to grow. There is, however, no assurance that such increase in demand will occur, that we will benefit from any demand increase or that our business will ever become profitable. We cannot predict the future growth rate and size of the market, assuming that the regulatory climate permits, of which there can be no assurance. Any negative outlook on medical cannabis will adversely affect our business prospects.
We also believe that large, well-funded pharmaceutical and other related businesses and industries may have economic reasons to oppose cannabinoid-based therapies. The pharmaceutical industry is well-funded with a strong and experienced lobby presence at both the federal and state levels, as well as internationally, that surpasses financial resources of the current group of medical cannabis research and development companies. Any effort by the pharmaceutical lobby to halt or delay cannabinoid-based medical products and therapies could have a detrimental impact on our business.
Risks Related to Product Development
If we fail to successfully develop and commercialize diagnostics, pharmaceutical or therapies, we may be unable to execute our plan of operations.
Our current business strategy focuses on discovering, developing and commercializing cannabinoid-based diagnostics, anti-cancer pharmaceuticals and palliative therapies. To date, we have only commercialized Cannabics SR, our non-pharmaceutical extended release capsules for palliative therapy. The success of our business will depend upon our ability to fully develop and commercialize the diagnostics and therapeutic product candidates in our current development pipeline as well as to continue the discovery and development of other products and technology.
| 36 |
Prior to commercializing our product candidates, we will be required to undertake time-consuming and costly development activities with uncertain outcomes, including conducting clinical studies and obtaining regulatory clearance or approval in Israel, the United States, the European Union and other countries where we may develop and market our product candidates. Delays in obtaining approvals and clearances could have material adverse effects on us and our ability to fully carry out our plan of operations. We have limited experience in taking products through these processes and there are considerable risks involved in these activities. The science and methods that we are employing are innovative and complex, and it is possible that our development programs will ultimately not yield products suitable for commercialization or government approval. Product candidates that appear promising in early development may fail to be validated in subsequent studies, and even if we achieve positive results, we may still fail to obtain the necessary regulatory clearances or approvals. Few research and development projects result in commercial products, and perceived viability in early clinical studies often is not replicated in later studies. At any point, we may abandon development of a product candidate, or we may be required to expend considerable resources obtaining additional clinical and nonclinical data, which would adversely impact the timing for generating potential revenue from those products. Further, our ability to develop and launch product candidates is dependent on our receipt of substantial additional funding. If our discovery and development programs yield fewer commercial product candidates than we expect, we may be unable to execute our business plan, and our business, financial condition and results of operations may be adversely affected.
If we fail to maintain or establish satisfactory arrangements for the supply of raw materials or the manufacture of our product candidates for preclinical or clinical trials, or if we experience an interruption of supply, we might not have sufficient quantities of our product candidates at an acceptable cost, which could delay, prevent or impair our development or commercialization efforts
We do not produce medical cannabis, and therefore our ability to research, develop and commercialize our cannabinoid-based diagnostics and therapeutic product candidates is dependent upon a sufficient supply of medical cannabis strains. Any significant interruption or negative change in the availability or economics of the supply chain for medical cannabis could materially impact our business, financial condition and operating results. Some strains of medical cannabis may only be available from a single supplier or a limited group of suppliers. If a sole source supplier were to go out of business, we might be unable to find a replacement source in a timely manner or at all. If a sole source supplier were to be acquired by a competitor, that competitor might elect not to supply us. Any inability to secure required supplies of medical cannabis or to do so on appropriate terms could have a materially adverse impact on our business, financial condition and operating results.
Our clinical diagnostics may never be validated.
The FDA regulates the sale and distribution, in interstate commerce, of in vitro diagnostic test kits, reagents and instruments used to perform diagnostic testing. To the extent that any diagnostic test we develop is regarded as an in vitro diagnostic test rather than as a Laboratory Developed Test (“LDT”), we will be subject to increased FDA regulation that will delay and add to the cost of commercialization of our diagnostic product candidates, which will have a material adverse effect on our business, results of operations and financial condition.
We are also subject to the United States Clinical Laboratory Improvement Amendments of 1988 (“CLIA”), federal regulatory standards that apply to all clinical laboratories that perform testing on specimens derived from humans in the United States for the purpose of providing information for the diagnosis, prevention or treatment of disease. CLIA is intended to ensure the quality and reliability of clinical laboratories in the United States by mandating specific standards in the areas of personnel qualifications, administration, and participation in proficiency testing, patient test management, quality control, quality assurance and inspections. Accreditation by the College of American Pathologists (“CAP”), one of six CLIA-approved accreditation organizations, is sufficient to satisfy the requirements of CLIA.
The validation for CLIA or CAP is a two-step process. The first step is optimization of all of the steps of the test protocol to show that the test is able to produce repeatable and consistent results. The second step is the clinical validation, in which statistically significant sensitivity and specificity of the test on the appropriate human samples are determined. Overall, the purpose of the validation process is to determine the accuracy, precision, sensitivity and specificity of the test. The time and cost to complete the validation process can vary widely, and it is possible that we would be unable to complete the validation process along the timeline and within the budget as planned.
As of the date of this filing, our clinical diagnostics have not yet been validated for commercialization in a CLIA or CAP laboratory, and we have not yet begun the validation process. We may be unable to enter into an agreement with a CLIA or CAP laboratory on favorable terms, or at all. Although we may be able to validate the tests, they might have sensitivity and specificity that is insufficient to bring the product to market. Any delays or incurrence of greater costs than budgeted in validating these tests may have a material adverse effect on our business, results of operations and financial condition.
| 37 |
The Federal Food and Drug Administration may impose additional regulatory obligations and costs upon the development of our diagnostics.
On October 3, 2014, the FDA issued draft guidance regarding oversight of LDTs, titled “Framework for Regulatory Oversight of Laboratory Developed Tests (LDTs).” According to this guidance, the FDA plans to take a phased-in risk-based approach to regulating LDTs. The FDA plans to phase in enforcement of LTD premarket review, quality system oversight and adverse event reporting over a number of years. The FDA would require that laboratories providing LDTs, subject to certain limited exemptions, within six months after the guidance documents are finalized to comply with (i) either a new notification procedure in which the laboratory must provide the FDA with certain basic information about each LDT offered by their laboratory or the FDA’s device registration and listing requirements, and (ii) the medical device reporting, or MDR, requirements for LDTs offered by that laboratory. Under this new risk-based approach, it is possible that some level of pre-market review may be required for our LDTs, which may require us to obtain additional clinical data.
The FDA draft guidance was subject to public comment until February 2, 2015. On January 13, 2017, the FDA issued a discussion paper on LDTs that does not represent the formal position of FDA and is not enforceable, but is intended to advance public discussion on future LDT oversight. At the present time, we cannot assess what the additional costs and regulatory burdens of any FDA final guidance or FDA enforcement will be, or the impact it may have on our business and operations.
If the FDA requires us to seek clearance or approval for any of our diagnostic products (as opposed to simply licensing our technology to a CLIA lab), we may not be able to obtain such approvals on a timely basis, or at all. The cost of conducting clinical trials and otherwise developing data and information to support any applications may be significant. Failure to comply with applicable regulatory requirements of the FDA could result in enforcement action, including receiving untitled or warning letters, fines, injunctions, or civil or criminal penalties. In addition, we could be subject to a recall or seizure of products, operating restrictions, partial suspension or total shutdown of production. Any such enforcement action would have a material adverse effect on our business, financial condition and operations.
Changes in laws and regulations concerning clinical diagnostic tests may adversely affect our business, financial condition and results of operations.
The clinical laboratory testing industry is highly regulated, and failure to comply with applicable regulatory, supervisory or licensing requirements may adversely affect our business, financial condition and results of operations. In particular, the laws and regulations governing the marketing and research of clinical diagnostic testing are extremely complex and in many instances there are no clear regulatory or judicial interpretations of these laws and regulations, which increase the risk that we may be found to be in violation of these laws.
The regulatory environment in which we operate may change significantly and adversely in the future. The molecular diagnostics industry as a whole is a growing industry and regulatory agencies such as the FDA may also apply heightened scrutiny to new developments in the field of molecular diagnostics. Should we be deemed to not be in compliance with regulatory requirements or any changes thereto, we may be subject to sanctions which could include required changes to our operations, adverse publicity, substantial financial penalties and criminal proceedings. Any change in the laws and the regulations relating to our business, whether in the form of new or amended laws or regulations or regulatory policies, or the application of any of the above, may adversely affect our business, financial condition and results of operations by increasing our costs to comply with the new laws or constraining our ability to develop, market and commercialize our diagnostic tests.
For example, a development affecting our industry is the increased enforcement of the federal False Claims Act and, in particular, actions brought pursuant to the False Claims Act's "whistleblower" or " qui tam " provisions. The False Claims Act imposes liability on any person or entity that, among other things, knowingly presents, or causes to be presented, a false or fraudulent claim for payment by a federal governmental payer program. The qui tam provisions of the False Claims Act allow a private individual to bring civil actions on behalf of the federal government for violations of the False Claims Act and permit such individuals to share in any amounts paid by the defendant to the government in fines or settlement. When an entity is determined to have violated the False Claims Act, it is subject to mandatory damages of three times the actual damages sustained by the government, plus mandatory civil penalties ranging from $5,500 to $11,000 for each false claim. In addition, various states have enacted false claim laws analogous to the federal False Claims Act, and in some cases go even further because many of these state laws apply where a claim is submitted to any third-party payer and not merely a governmental payer program.
| 38 |
In addition, there has been a recent trend of increased U.S. federal and state regulation of payments made to physicians, which are governed by laws and regulations including the Stark Law. Among other requirements, the Stark Law requires laboratories to track, and places a cap on, non-monetary compensation provided to referring physicians. While we have a compliance plan to address compliance with applicable fraud and abuse laws and regulations, the evolving commercial compliance environment and the need to build and maintain robust and expandable systems to comply with multiple jurisdictions with different compliance and reporting requirements increases the possibility that we could violate one or more of these requirements.
All of our diagnostics and therapeutic product candidates are in clinical and preclinical development, the validation of which may not be successful and may be subject to delays, which would have a material adverse effect on our business, results of operation and financial condition.
To date, we have devoted our resources towards developing the technology upon which we are building our clinical diagnostics and therapeutic product candidates. Our clinical diagnostic product candidates have yet to be validated and our clinical therapeutic product candidates are currently in a preclinical development phase. As of the date of this filing, only Cannabics SR, our non-pharmaceutical palliative therapy, has been commercialized.
We may be unable to successfully complete the clinical validation process for our diagnostic product candidates due to several factors, including our ability to acquire enough samples for full validation and the procurement of materials necessary to conduct testing.
We may not be able to successfully complete the preclinical testing necessary to advance our therapeutic product candidates into clinical development, including animal pharmacology and toxicity studies. The results of any preclinical work may indicate that our therapeutic product candidates do not have the safety or efficacy necessary to file an Investigational New Drug (“IND”) with the FDA in order to move our product on to the clinical development process.
Once we initiate the clinical development of our product candidates, it may be difficult to identify and qualify patients to participate in future clinical trials for our product candidates, and the timing of our clinical trials depends on the speed at which we can recruit patients to participate in testing as well as completion of required follow-up periods. If patients are unwilling to participate in our clinical trials due to concerns over the safety of the product candidate or for other reasons, the timeline for conducting the trials and obtaining regulatory approval may be delayed. Furthermore, we may also compete for patients with other companies conducting similar clinical trials. Any delays in our future clinical trials could result in increased costs, delays in product development or termination of the clinical trials altogether.
Any of these events could have a material adverse effect on our business, results of operations and financial condition.
We may fail to demonstrate the safety and efficacy of our therapeutic product candidates in accordance with regulatory standards and may incur delays and substantial costs in our clinical trials.
In order to commercialize our therapeutic product candidates, we must conduct extensive clinical trials demonstrating the safety and efficacy of our product candidates in humans. The clinical testing process is expensive, difficult to design and implement, takes many years to complete and is unpredictable in both its duration and outcome. A failure of one or more clinical trials can occur at any stage of testing. There is a high failure rate for drugs and biological products proceeding through clinical trials. The research, testing, manufacturing, labeling, packaging, storage, approval, sale, marketing, advertising and promotion, pricing, export, import and distribution of drug products are subject to extensive regulation by the FDA and other regulatory authorities in the United States and other countries, which regulations differ from country to country. We are not permitted to market our therapeutic product candidates as a prescription pharmaceutical product in the United States until we receive approval of a New Drug Application (“NDA”), from the FDA, or in any foreign countries until we receive the requisite approval from such countries. In the United States, the FDA generally requires the completion of pre-clinical testing and clinical trials of each drug to establish its safety and efficacy and extensive pharmaceutical development to ensure its quality before an NDA is approved. Regulatory authorities in other jurisdictions impose similar requirements. Of the large number of drugs in development, only a small percentage result in the submission of an NDA to the FDA and even fewer are eventually approved for commercialization. We have not submitted an NDA to the FDA or comparable applications to other regulatory authorities. Preclinical and clinical data is often susceptible to varying interpretations and types of analyses and regulatory authorities may fail to approve our product. In addition, even if we successfully complete early clinical trials, such results may not be indicative of the success or results of our later clinical trials.
| 39 |
Our successful completion of clinical trials may be materially adversely affected by many factors, including:
| · | ineffective trial design and disagreement with the FDA on final trial design; |
| · | imposition of a clinical hold following an inspection of our clinical trial operations by the FDA or other regulatory authorities; |
| · | difficulties or delays in reaching an agreement with a contract research organization, and clinical trial sites; |
| · | delays in obtaining required institutional review board approval for each trial site; |
| · | data collected from clinical trials may not be sufficient to support the submission of a NDA or other submission or to obtain regulatory approval in the United States or elsewhere; |
| · | delays or difficulties in recruiting suitable patients to participate in clinical trials; |
| · | delays in manufacturing or delivering products and materials to clinical trial sites; |
| · | delays or difficulties caused by lack of patient adherence to treatment or post-treatment follow-up; |
| · | delays caused by patients dropping out of a trial and the need for recruiting additional patients; and |
| · | delays caused by clinical sites dropping out of the trial and the time required to recruit a new site. |
Any of these delays or difficulties could cause us to be delayed in obtaining marketing approval from regulatory authorities, if at all, or allow us to obtain approval for specific indications or patient populations that are not as broad as currently targeted. In addition, such delays or difficulties may cause our development costs or our time to bring our product candidates to market to increase, may weaken our competitive positioning in the market and may have a material adverse effect on our business, results of operations and financial condition.
We cannot predict if or when we will receive regulatory approval to commercialize a therapeutic product candidate.
We cannot commercialize a therapeutic product candidate until the appropriate regulatory authorities, such as the FDA or a state regulating authority, have reviewed and approved the product candidate. Even if our therapeutic product candidates demonstrate safety and efficacy in clinical trials, regulatory agencies may not complete their review processes in a timely manner, and we may not be able to obtain timely regulatory approval. We may never be able to receive regulatory approval for our therapeutic product candidates at all. Additional delays may result if an FDA advisory committee or other regulatory authority recommends non-approval or restrictions on approval. In addition, we may experience delays or rejections based upon additional government regulation from future legislation or administrative action, or changes in regulatory agency policy during the period of product development, clinical trials and the review process. Regulatory agencies may also approve a product candidate for fewer or more limited indications than requested or may grant approval subject to the performance of post-marketing studies. In addition, regulatory agencies may not approve the labeling claims that are necessary or desirable for the successful commercialization of our product candidates. Delays or failure to obtain necessary regulatory approvals could have a material adverse effect on our business, results of operations and financial condition.
Even if we obtain regulatory approval for a therapeutic product candidate, we will remain subject to extensive regulatory scrutiny.
Even if we obtain regulatory approval in the United States for our therapeutic product candidates, the FDA and other appropriate regulatory agencies may still impose significant restrictions or delays, including restriction of patient population or indications or additional costly studies. Any changes to the approved product or its labeling or manufacturing process would require FDA approval. Any advertisements or promotions must comply with FDA regulations and are subject to FDA review as well as state and federal laws. Drug product manufacturers are subject to continual review and inspection by the FDA and other regulatory authorities to comply with Current Good Manufacturing Practice standards. If the FDA or other regulatory authority finds previously undiscovered compliance issues with products, such as unanticipated adverse effects or issues with the manufacturing facility, the FDA or other regulatory authority may:
| 40 |
| · | issue a warning letter asserting that we are in violation of law; |
| · | seek an injunction; |
| · | impose civil or criminal penalties or monetary fines; |
| · | suspend or withdraw regulatory approval; |
| · | suspend currently ongoing clinical trials; |
| · | refuse any pending applications; |
| · | seize product; or |
| · | prohibit us from entering into beneficial or necessary contracts such as supply or government contracts. |
Any government investigation of alleged violations of law could require us to expend significant time and resources in response, could result in litigation and litigation-related expense and could generate negative publicity. The occurrence of any event or penalty described above may inhibit our ability to commercialize our therapeutic product candidates and generate revenue, which would have a material adverse effect on our business, results of operations and financial condition.
In addition, even if we obtain regulatory approvals, the timing or scope of any approvals may prohibit or reduce our ability to commercialize our therapeutic product candidates successfully. For example, if the approval process takes too long, we may miss market opportunities and give other companies the ability to develop competing products or establish market dominance. Any regulatory approval that we ultimately obtain may be limited or subject to restrictions or post-approval commitments that render our products not commercially viable. For example, regulatory authorities may approve our therapeutic product candidates for fewer or more limited indications than we request, may not approve the price we intend to charge for our therapeutic product candidates, may grant approval contingent on the performance of costly post-marketing clinical trials, or may approve our therapeutic product candidates with a label that does not include the labeling claims necessary or desirable for the successful commercialization of that indication. Further, the FDA may place conditions on approvals including potential requirements or risk management plans and the requirement for a Risk Evaluation and Mitigation Strategy (“REMS”) to assure the safe use of the drug. If the FDA concludes a REMS is needed, the sponsor of the NDA must submit a proposed REMS; the FDA will not approve the NDA without an approved REMS, if required. A REMS could include medication guides, physician communication plans, or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. Any of these limitations on approval or marketing could restrict the commercial promotion, distribution, prescription or dispensing of our therapeutic product candidates. Moreover, product approvals may be withdrawn for non-compliance with regulatory standards or if problems occur following the initial marketing of the product. Any of the foregoing scenarios could materially harm the commercial success of our product candidates and have a material adverse effect on our business, results of operations and financial condition.
We may fail to obtain orphan drug status for our therapeutic product candidates.
We intend to seek orphan drug status from the FDA for those anti-cancer therapeutic product candidates we are presently developing to the extent such product candidates are eligible for orphan drug status under the Orphan Drug Act of 1983. The orphan drug status gives the manufacturer specific financial incentives to develop a pharmacological agent. If a product that has an orphan drug designation receives the first FDA approval for the disease for which it has such designation, the product is entitled to orphan drug exclusivity, which means that the FDA may not approve any other applications to market the same medication for the same indication, except in very limited circumstances, for seven years. Failure to obtain an orphan drug designation for our product candidates may have a material adverse effect on our business, results of operations and financial condition.
| 41 |
Any of our therapeutic product candidates may cause adverse effects or have properties that could delay or prevent their regulatory approval or limit the scope of any specific indications or market acceptance.
Adverse events caused by our therapeutic product candidates could cause interruptions, delays or the halting of our clinical trials. If adverse effects are observed in any clinical trials for our therapeutic product candidates, we may be unable to obtain timely, or any, regulatory approval of our therapeutic product candidates. Adverse effects caused by our therapeutic product candidates could also subject us to litigation and liability, which could have a material adverse effect on our business, results of operations and financial condition.
In addition, if any of our therapeutic product candidates are approved for commercialization and are found to cause serious or unpredicted side effects, serious consequences may result, including but not limited to, the withdrawal of marketing approval by regulatory authorities, restrictions on distribution by regulatory authorities, the need to conduct additional clinical trials, litigation and potential liability for personal injury to patients and damage to our reputation. Furthermore, our ability to achieve and maintain profitability may be permanently impaired. Any of these events could have a material adverse effect on our business, results of operations and financial condition.
Our dietary supplements are subject to government regulation, both in the United States and internationally, which could increase our costs significantly and limit or prevent the sale of our dietary supplements.
The manufacture, packaging, labeling, advertising, promotion, distribution and sale of Cannabics SR, and any other dietary supplements that we may develop and commercialize, is subject to regulation by numerous national and local governmental agencies in the United States and other countries, including the FDA and Federal Trade Commission in the United States, and the Ministry of Health in Israel. Failure to comply with these regulatory requirements may result in various types of penalties or fines. These include injunctions, product withdrawals, recalls, product seizures, fines and criminal prosecutions. Individual states also regulate dietary supplements. A U.S. state may interpret claims or products presumptively valid under federal law as illegal under that state’s regulations. In markets outside the United States, we will likely be required to obtain approvals, licenses, or certifications from a country’s ministry of health or comparable agency, as well as labeling and packaging regulations, all of which vary from country to country. Approvals or licensing may be conditioned on reformulation of products or may be unavailable with respect to certain products or product ingredients. Any of these government agencies, as well as legislative bodies, can change existing regulations, or impose new ones, or could take aggressive measures, causing or contributing to a variety of negative consequences, including:
| · | requirements for the reformulation of certain or all products to meet new standards; |
| · | the recall or discontinuance of certain or all products; |
| · | additional record keeping; |
| · | expanded documentation of the properties of certain or all products; |
| · | expanded or different labeling; |
| · | adverse event tracking and reporting; and |
| · | additional scientific substantiation. |
Any or all of these requirements could have a material adverse effect on us. There can be no assurance that the regulatory environment in which we operate will not change or that such regulatory environment, or any specific action taken against us, will not result in a material adverse effect on us.
| 42 |
Changes in legislation or regulation in the health care systems in the United States and foreign jurisdictions may affect us.
Our ability to successfully commercialize our cannabinoid-based products may depend on how the healthcare systems of the United States, the European Union and other governments provide coverage or reimbursement. Reimbursement and healthcare payment systems in international markets vary significantly by country, and include both government sponsored healthcare and private insurance. To obtain reimbursement or pricing approval in some countries, we may be required to produce clinical data, which may involve one or more clinical trials, that compares the cost-effectiveness of our products to other available therapies. We may not obtain international reimbursement or pricing approvals in a timely manner, if at all. Our failure to receive international reimbursement or pricing approvals would negatively impact market acceptance of our products in the international markets in which those approvals are sought.
We believe that future reimbursement may be subject to increased restrictions in the United States, the European Union and in other international markets. There is increasing pressure by governments worldwide to contain health care costs by limiting both the coverage and the level of reimbursement for therapeutic products and by refusing, in some cases, to provide any coverage for products that have not been approved by the relevant regulatory agency. Future legislation, regulation or reimbursement policies of third-party payers may adversely affect the demand for our product candidates currently under development and limit our ability to sell our product candidates on a profitable basis. In addition, third party payers continually attempt to contain or reduce the costs of healthcare by challenging the prices charged for healthcare products and services. If reimbursement for our products is unavailable or limited in scope or amount or if pricing is set at unsatisfactory levels, market acceptance of our products candidates will be impaired and future revenues, if any, will be adversely affected.
Risks Related to Our Dependence on Third Parties
We rely and expect to continue to rely heavily on third parties to conduct our preclinical studies and clinical trials, and those third parties may not perform satisfactorily, including failing to meet deadlines for the completion of such studies and trials.
We do not have in-house research facilities and, as a consequence, we must currently rely on third parties to conduct our clinical trials. We expect to continue to rely heavily on third parties, such as contract research organizations, clinical data management organizations, medical institutions, clinical investigators and others to conduct our clinical trials. Our agreements with these third parties generally allow the third party to terminate our agreement with them at any time. If we are required to enter into alternative arrangements because of any such termination, the introduction of our product candidates to market could be delayed.
Our reliance on third parties for research and development will reduce our control over such activities but will not relieve us of our responsibilities. Likewise, our reliance on third parties whom we do not control does not relieve us of our responsibility to comply with regulatory requirements to use Current Good Clinical Practice standards when conducting, recording and reporting the results of clinical trials in order to ensure that data and reported results are credible and accurate and that the rights, integrity and confidentiality of trial participants are protected. We are also required to register ongoing clinical trials and post the results of completed clinical trials on a government-sponsored database of regulatory agencies within specified timeframes. Failure to do so can result in fines, adverse publicity and civil and criminal sanctions.
The third parties on whom we rely may also have relationships with other entities, some of whom may be our competitors. If these third parties do not successfully carry out their contractual duties, meet expected deadlines or conduct our clinical trials in accordance with the requirements of a regulatory agency or our stated protocols, we will not be able to obtain, or may be delayed in obtaining, marketing approvals for our product candidates and will not be able to, or may be delayed in our efforts to, successfully commercialize our product candidates.
Collaboration agreements that we may enter into in the future may not be successful, which could adversely affect our ability to develop and commercialize our diagnostics and therapeutic product candidates.
We may enter into collaboration agreements with pharmaceutical companies and biotechnology institutions for the development or commercialization of our cannabinoid-based diagnostics and therapeutic product candidates, which agreements may contain provisions based upon, among other things, the merits of retaining certain rights. We will face significant competition in seeking appropriate collaborators and in negotiating agreements at acceptable terms, if at all. We may not be successful in our efforts to enter, implement and maintain collaboration agreements. Disagreements stemming from collaboration agreements concerning development, intellectual property, regulatory or commercialization matters can lead to delays and, in some cases, termination of our collaboration agreements or otherwise result in the potentially significant costs and fees in seeking to enforce or protect our rights, if any. Any such disagreements can be difficult if, in fact, neither of the parties has final decision-making authority. The resulting outcome of any disputes or disagreements would in all likelihood adversely affect our business.
| 43 |
Data provided by collaborators and others upon which we rely that has not been independently verified could turn out to be false, misleading, or incomplete.
We rely on third-party vendors, scientists, and collaborators to provide us with significant data and other information related to our projects, clinical trials, and our business. If such third parties provide inaccurate, misleading, or incomplete data, our business, prospects, and results of operations could be materially adversely affected.
Our business model is substantially dependent on third party licensees to market and sell our products, which will subject us to a number of risks.
We depend on third party licensees to sell, market, and service our products and current and future products in our intended markets. We are subject to a number of risks associated with reliance upon third party licensees, including:
| · | lack of day-to-day control over the activities of licensees; |
| · | third party licensees may not commit the necessary resources to market and sell our current and future products to our level of expectations; |
| · | third party licensees may terminate their arrangements with us on limited or no notice or may change the terms of these arrangements in a manner unfavorable to us; and |
| · | disagreements with our future licensees could result in costly and time-consuming litigation or arbitration which we could be required to conduct in jurisdictions with which we are not familiar. |
If we fail to establish and maintain satisfactory relationships with our future third party licensees, our revenue and market share may not grow as anticipated, and we could be subject to unexpected costs which could harm our results of operations and financial condition.
Risks Related To Operating In Israel
Failure to secure the necessary Israeli licenses to use cannabis for medical research could limit our ability to execute our research and development activities, delay the launch of our products and adversely affect the results of our business operations.
To date, we have only conducted our research in Israel and, in fact, have limited our operations to Israel. The biotechnologies that we are developing contain cannabis, a “controlled substance” as defined in the Israeli Dangerous Drugs Ordinance [New Version], 5733 - 1973. In Israel, licenses to cultivate, possess and to use cannabis for medical research are granted by the Ministry of Health, Israel Medical Cannabis Unit (“IMCU”), on an ad hoc basis. We have obtained all IMCU licenses that are necessary for us to carry out our research. Even though we have an established track record of successfully obtaining the requisite licenses as required, there can be no assurance that we will continue to be able to secure licenses in the future. If we fail to comply with Israeli rules and regulations related to the licensing of cannabis, we may not be able to research and develop our product candidates as we intend or at all.
We may become subject to claims for remuneration or royalties for assigned service invention rights by our employees, which could result in litigation and adversely affect our business.
A significant portion of our intellectual property has been developed by our Israeli employees in the course of their employment for us. Under the Israeli Patent Law, 5727-1967 (the “Israeli Patent Law”), inventions conceived of by an employee during the term and as part of the scope of his or her employment with a company are regarded as “service inventions,” which belong to the employer, absent a specific agreement between the employee and employer giving the employee service invention rights. The Israeli Patent Law also provides that if there is no such agreement between an employer and an employee, the Israeli Compensation and Royalties Committee (the “C&R Committee”), a body constituted under the Israeli Patent Law, shall determine whether the employee is entitled to remuneration for his or her inventions. The C&R Committee (decisions of which have been upheld by the Israeli Supreme Court) has held that employees may be entitled to remuneration for their service inventions despite having specifically waived any such rights. Further, the C&R Committee has not yet set specific guidelines regarding the method for calculating this remuneration or the criteria or circumstances under which an employee’s waiver of his or her right to remuneration will be disregarded. We generally enter into intellectual property assignment agreements with our employees pursuant to which such employees assign to us all rights to any inventions created in the scope of their employment or engagement with us. Although our employees have agreed to assign to us service invention rights and have specifically waived their right to receive any special remuneration for such assignment beyond their regular salary and benefits, we may face claims demanding remuneration in consideration for assigned inventions. As a consequence of such claims, we could be required to pay additional remuneration or royalties to our current or former employees, or be forced to litigate such claims, which could negatively affect our business.
| 44 |
We expect that our results of operations will be subject to fluctuations in currency exchange rates because a substantial portion of our anticipated revenue will be generated in U.S. dollars and Euros while a significant portion of our expenses will be incurred in New Israeli Shekels.
We expect a substantial portion of our revenue will be generated in U.S. dollars and Euros, while a significant portion of our expenses, principally salaries and related personnel expenses, is paid in New Israeli Shekels, or NIS. As a result, we are exposed to the risk that the rate of inflation in Israel will exceed the rate of devaluation of the NIS in relation to the Euro or the U.S. dollar, or that the timing of this devaluation will lag behind inflation in Israel. Because inflation has the effect of increasing the U.S. dollar and Euro costs of our operations, it would therefore have an adverse effect on our dollar-measured results of operations. The value of the NIS, against the Euro, the U.S. dollar, and other currencies may fluctuate and is affected by, among other things, changes in Israel’s political and economic conditions. Any significant revaluation of the NIS may materially and adversely affect our cash flows, revenues and financial condition. Fluctuations in the NIS exchange rate, or even the appearance of instability in such exchange rate, could adversely affect our ability to operate our business.
We may not be able to enforce covenants not-to-compete under current Israeli law.
We have non-competition agreements with most of our employees, all of which are governed by Israeli law. These agreements generally prohibit our employees from competing with us or working for our competitors for a specified period following termination of their employment. However, Israeli courts are reluctant to enforce non-compete undertakings of former employees and tend, if at all, to enforce those provisions for relatively brief periods of time in restricted geographical areas and only when the employee has unique value specific to that employer’s business and not just regarding the professional development of the employee. Any such inability to enforce non-compete covenants may cause us to lose any competitive advantage arising from confidential information known to such former employees.
It may be difficult for investors in the United States to enforce any judgments obtained against us or some of our directors or officers.
The majority of our assets are located outside the United States. In addition, certain of our officers are nationals or residents of countries other than the United States, and all or a substantial portion of such persons’ assets are located outside the United States. As a result, it may be difficult for investors to enforce within the United States any judgments obtained against us or any of our non-U.S. officers, including judgments predicated upon the civil liability provisions of the securities laws of the United States or any state thereof. It may also be difficult to assert claims under United States securities law in actions originally instituted outside of the United States. Moreover, Israeli courts may refuse to hear a United States securities law claim because Israeli courts may not be the most appropriate forums in which to bring such a claim. Even if an Israeli court agrees to hear a claim, it may determine that Israeli law, and not U.S. law, is applicable to the claim. Further, if U.S. law is found to be applicable, certain content of applicable U.S. law must be proved as a fact, which can be a time-consuming and costly process, and certain matters of procedure would still be governed by Israeli law. Consequently, our investors may be effectively prevented from pursuing remedies under U.S. federal and state securities laws against us or any of our non-U.S. directors or officers.
If there are significant shifts in the political, economic and military conditions in Israel and its neighbors, it could have a material adverse effect on our business relationships and profitability.
All of our research facilities and certain of our key personnel are located in Israel. Our business is directly affected by the political, economic and military conditions in Israel and its neighbors. Since the establishment of the State of Israel in 1948, a number of armed conflicts have occurred between Israel and its Arab neighbors. A state of hostility, varying in degree and intensity, has caused security and economic problems in Israel. Although Israel has entered into peace treaties with Egypt and Jordan, and various agreements with the Palestinian Authority, there has been a marked increase in violence, civil unrest and hostility, including armed clashes, between the State of Israel and the Palestinians since September 2000. The establishment in 2006 of a government in the Gaza Strip by representatives of the Hamas militant group has created heightened unrest and uncertainty in the region. In mid-2006, Israel engaged in an armed conflict with Hezbollah, a Shiite Islamist militia group based in Lebanon, and in June 2007, there was an escalation in violence in the Gaza Strip. From December 2008 through January 2009 and again in November and December 2012, Israel engaged in an armed conflict with Hamas, which involved missile strikes against civilian targets in various parts of Israel and negatively affected business conditions in Israel. In July 2014, Israel launched an additional operation against Hamas operatives in the Gaza strip in response to Palestinian groups launching rockets at Israel. Recent political uprisings and social unrest in Syria are affecting its political stability, which has led to the deterioration of the political relationship between Syria and Israel and have raised new concerns regarding security in the region and the potential for armed conflict. Similar civil unrest and political turbulence is currently ongoing in many countries in the region. The continued political instability and hostilities between Israel and its neighbors and any future armed conflict, terrorist activity or political instability in the region could adversely affect our operations in Israel and adversely affect the market price of our shares of common stock. In addition, several countries restrict doing business with Israel and Israeli companies have been and are today subjected to economic boycotts. The interruption or curtailment of trade between Israel and its present trading partners could adversely affect our business, financial condition and results of operations.
| 45 |
Risks Related to Our Stock
There can be no assurance of an active, liquid and orderly trading market for our common stock or that investors will be able to sell their shares of common stock.
At present, our common stock is quoted on the OTCQB tier of the marketplace maintained by OTC Markets Group Inc., under the symbol “CNBX.” There is only a limited, liquid public trading market for our common stock. There can be no assurance that a liquid market for our common stock will continue. Market liquidity will depend on the perception of our business and any steps that our management might take to bring public awareness of our business to the investing public within the parameters of the federal securities laws. There is no assurance that any such awareness will be generated or sustained. Therefore, investors may not be able to liquidate their investment or liquidate it at a price paid by investors equal to or greater than their initial investment in our common stock. Moreover, holders of our common stock may not find purchasers for their shares should they to decide to sell the common stock held by them at any particular time if ever. Our common stock should be purchased only by investors who have no immediate need for liquidity in their investment and who can hold our common stock, possibly for a prolonged period of time.
The price of our common stock is volatile, and the value of your investment could decline.
The market price of our common stock has been highly volatile. Between September 1st, 2021, and August 31st, 2022, the sales price of our stock on the OTCQB ranged from a low of $0.16 per share to a high of $0.45 per share. Accordingly, it is difficult to forecast the future performance of our common stock. The market price of our common stock may be higher or lower than the price you pay, depending on many factors, some of which are beyond our control and may not be related to our operating performance. These fluctuations could cause you to lose all or part of your investment in our common stock. Factors that could cause fluctuations in the trading price of our common stock include the following:
| · | technological innovations or new products and services by us or our competitors; |
| · | regulatory developments at the federal, state or local level; |
| · | additions or departures of key personnel; |
| · | our ability to execute our business plan; |
| · | operating results that fall below expectations; |
| · | loss of any strategic relationship; |
| · | industry developments; |
| · | economic, political and other external factors; and |
| · | period-to-period fluctuations in our financial results. |
The stock market generally and in particular, the market for stocks of biotechnology companies with lower market capitalizations, like us, have from time to time experienced, and likely will again experience significant price and volume fluctuations that are unrelated to the operating performance of a particular company. The trading price of our common stock might decline in reaction to events that affect other companies in our industry, even if these events do not directly affect us.
Periods of volatility in the market price of a company’s securities have often been followed by securities class action litigation against that company. If our stock price continues to be volatile, we may become the target of securities litigation, which could result in substantial costs and divert our management’s attention and resources from our business. This could have a material adverse effect on our business, operating results and financial condition.
| 46 |
We may never pay any dividends to our shareholders.
We currently intend to retain any future earnings for use in the operation and expansion of our business. Accordingly, we do not expect to pay any dividends in the foreseeable future, but will review this policy as circumstances dictate. The declaration and payment of all future dividends, if any, will be at the sole discretion of our board of directors, which retains the right to change our dividend policy at any time. Consequently, our stockholders must rely on sales of their common stock after price appreciation, which may never occur, as the only way to realize any future gains on their investment.
Our principal stockholders and management own a significant percentage of our stock and will be able to exert significant control over matters subject to stockholder approval.
As of August 31, 2022, Cannabics Inc., a Delaware corporation owns 57.6% of our common stock. Our three Directors are also holders of shares in Cannabics Inc., and therefore have substantial influence over it. Accordingly, the Company (and our management) may be able to control the outcome of stockholder votes, including votes concerning the election of directors, amendment of our organizational documents, approval of mergers, sales of assets and other significant corporate transactions. This concentration of ownership in Cannabics Inc. (and our management) may have the effect of delaying or preventing a change in our management and voting control of Cannabics Inc., including preventing or discouraging unsolicited acquisition proposals or offers for our common stock that some of our stockholders may believe is in their best interest.
We may issue shares of preferred stock with greater rights than our common stock, which may entrench management and result in dilution of our stockholders' investment.
Our Articles of Incorporation authorize the issuance of up to 100 million shares of preferred stock, par value $0.0001 per share. The authorized but unissued preferred stock may be issued by our board of directors from time to time on any number of occasions, without stockholder approval, as one or more separate series of shares comprised of any number of the authorized but unissued shares of preferred stock, designated by resolution of our board of directors stating the name and number of shares of each series and setting forth separately for such series the relative rights, privileges and preferences thereof, including, if any, the: (i) rate of dividends payable thereon; (ii) price, terms and conditions of redemption; (iii) voluntary and involuntary liquidation preferences; (iv) provisions of a sinking fund for redemption or repurchase; (v) terms of conversion to common stock, including conversion price, and (vi) voting rights. Such preferred stock may enable our board of directors to hinder or discourage any attempt to gain control of the Company by a merger, tender offer at a control premium price, proxy contest or otherwise. Consequently, the preferred stock could entrench our management. The market price of our common stock could be depressed by the existence of the preferred stock.
Nevada law and certain provisions of our Articles of Incorporation and bylaws may discourage mergers and other transactions.
Provisions of Nevada law, such as its business combination statute, and certain provisions of our Articles of Incorporation and by-laws could make it more difficult for someone to acquire control of the Company and limit the price that certain investors might be willing to pay for shares of our common stock. These provisions may make it more difficult for stockholders to take certain corporate actions and could delay or prevent someone from acquiring our business. The provisions could be beneficial to our management and the board of directors in a hostile tender offer, and could have an adverse impact on stockholders who might want to participate in such tender offer, or who might want to replace some or all of the members of the board of directors.
Our common stock may be subject to penny stock rules, which may make it more difficult for our investors to sell their common stock.
Our common stock is presently considered to be a "penny stock" and is subject to SEC rules and regulations that impose limitations upon the manner in which such shares may be publicly traded, and regulate broker-dealer practices in connection with transactions in "penny stocks." Penny stocks generally are equity securities with a price of less than $5.00 (other than securities registered on certain national securities exchanges or quoted on the NASDAQ system, provided that current price and volume information with respect to transactions in such securities is provided by the exchange or system). The penny stock rules require a broker-dealer, prior to a transaction in a penny stock not otherwise exempt from the rules, to deliver a standardized risk disclosure document that provides information about penny stocks and the risks in the penny stock market. The broker-dealer must also provide the customer with current bid and offer quotations for the penny stock, the compensation of the broker-dealer and its salesperson in the transaction, and monthly account statements showing the market value of each penny stock held in the customer's account. In addition, the penny stock rules generally require that prior to a transaction in a penny stock, the broker-dealer make a special written determination that the penny stock is a suitable investment for the purchaser and receive the purchaser's written agreement to the transaction. These disclosure requirements may have the effect of reducing the level of trading activity in the secondary market for a stock that becomes subject to the penny stock rules which may increase the difficulty investors may experience in attempting to liquidate such securities. These requirements could also hamper our ability to raise funds in the primary market for our shares of common stock.
| 47 |
Item 1B. Unresolved Staff Comments
None.
Item 2. Properties
Our executive offices and laboratory are located in Eli Horovitz Street, 12 Rehovot Israel. We lease the property of our laboratory in Rehovot, Israel, the monthly lease is $6,500 per month. Our current lease terminates at the end of February 2024, though we have a two additional one-year option, which management intends to execute prior to that time.
As security for its obligation under a property lease agreement, car lease and credit cards, the Company’s subsidiary provided a bank guarantee in the amount of $20,000.
Item 3. Legal Proceedings
None.
Item 4. Mine Safety Disclosures
Not applicable.
| 48 |
PART II
Item 5. Market For Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Market Information.
We have one class of securities, Common Voting Equity Shares ("Common Stock"). The holders of our common stock have equal ratable rights to dividends from funds legally available if and when declared by our Board of Directors and are entitled to share pro-rata in all of our available assets for distribution to holders of common stock upon liquidation, dissolution or winding up of our affairs; there are no preemptive, subscription or conversion rights and there are no redemption or sinking fund provisions or rights.
Our common stock is quoted on the NASDAQ OTC Bulletin Board (“OTCQB”) under the symbol "CNBX". As of October 14th, 2022, the Company’s common stock was held by 76 shareholders of record, which does not include shares that are held in street or nominee name.
Shareholders
Our shares of common stock are issued in registered form. The registrar and transfer agent for our shares of common stock is ClearTrust LLC, 16540 Pointe Village Dr. Suite 210, Lutz, FL 33558; (813) 235-4490.
On October 14th, 2022, the shareholders' list of our shares of common stock showed 76 registered holders of our shares of common stock and 148,661,247 shares of common stock outstanding. The number of record holders was determined from the records of our transfer agent and does not include beneficial owners of shares of common stock whose shares are held in the names of various security brokers, dealers, and registered clearing agencies, which as of last quarter was in excess of 35,000.
Issuer Purchases of Equity Securities
During the years ended August 31, 2022, and August 31, 2021, there were no repurchases of the Company’s common stock by the Company.
Dividend Policy
Our board of directors may declare and pay dividends on outstanding shares of common stock out of funds legally available there for in our sole discretion; however, to date no dividends have been declared or paid on common stock.
Indemnification of Directors and Officers
Nevada Corporation Law allows for the indemnification of officers, directors, and any corporate agents in terms sufficiently broad to indemnify such persons under certain circumstances for liabilities, including reimbursement for expenses, incurred arising under the 1933 Act. The Bylaws of the Company provide that the Company will indemnify its directors and officers to the fullest extent authorized or permitted by law and such right to indemnification will continue as to a person who has ceased to be a director or officer of the Company and will inure to the benefit of his or her heirs, executors and Consultants; provided, however, that, except for proceedings to enforce rights to indemnification, the Company will not be obligated to indemnify any director or officer in connection with a proceeding (or part thereof) initiated by such person unless such proceeding (or part thereof) was authorized by the Board of Directors. The right to indemnification conferred will include the right to be paid by the Company the expenses (including attorney’s fees) incurred in defending any such proceeding in advance of its final disposition.
The Company may, to the extent authorized from time to time by the Board of Directors, provide rights to indemnification and to the advancement of expenses to employees and agents of the Company similar to those conferred to directors and officers of the Company. The rights to indemnification and to the advancement of expenses are subject to the requirements of the 1940 Act to the extent applicable.
Furthermore, the Company may maintain insurance, at its expense, to protect itself and any director, officer, employee or agent of the Company or another company against any expense, liability or loss, whether or not the Company would have the power to indemnify such person against such expense, liability or loss under the Nevada General Corporation Law.
| 49 |
Anti-Takeover Provisions Under the Nevada Revised Statutes
Certain provisions of Nevada law, and our Articles of Incorporation and our Bylaws, each as amended and subject, where applicable as described below, our opting out of certain provisions of Nevada law, contain provisions that could make the following transactions more difficult: acquisition of us by means of a tender offer; acquisition of us by means of a proxy contest or otherwise; or removal of our incumbent officers and directors. It is possible that these provisions could make it more difficult to accomplish or could deter transactions that stockholders may otherwise consider to be in their best interest or in our best interests, including transactions that might result in a premium over the market price for our shares.
These provisions, summarized below, are expected to discourage coercive takeover practices and inadequate takeover bids. These provisions are also designed to encourage persons seeking to acquire control of us to first negotiate with our board of directors. We believe that the benefits of increased protection of our potential ability to negotiate with the proponent of an unfriendly or unsolicited proposal to acquire or restructure us outweigh the disadvantages of discouraging these proposals because negotiation of these proposals could result in an improvement of their terms.
Recent Sales of Unregistered Securities
None.
Penny Stock Regulation
Our shares must comply with the Penny Stock Reform Act of 1990, which may potentially decrease our shareholders’ ability to easily transfer their shares. Broker-dealer practices in connection with transactions in "penny stocks" are regulated. Penny stocks generally are equity securities with a price of less than $5.00. The penny stock rules require a broker-dealer, prior to a transaction in a penny stock not otherwise exempt from the rules, to deliver a standardized risk disclosure document that provides information about penny stocks and the risks in the penny stock market. The broker-dealer also must provide the customer with current bid and offer quotations for the penny stock, the compensation of the broker-dealer and its salesperson in the transaction, and monthly account statements showing the market value of each penny stock held in the customer's account. In addition, the penny stock rules generally require that prior to a transaction in a penny stock, the broker-dealer make a special written determination that the penny stock is a suitable investment for the purchaser and receive the purchaser's written agreement to the transaction. These disclosure requirements may have the effect of reducing the level of trading activity in the secondary market for a stock that must comply with the penny stock rules. Since our shares must comply with such penny stock rules, our shareholders will in all likelihood find it more difficult to sell their securities.
Item 6. Selected Financial Data
We are a smaller reporting company as defined by Rule 12b-2 of the Exchange Act and are not required to provide the information required under this item.
Item 7. Management's Discussion and Analysis of Financial Condition and Results of Operations
THE FOLLOWING DISCUSSION SHOULD BE READ IN CONJUNCTION WITH OUR AUDITED FINANCIAL STATEMENTS AND THE RELATED NOTES THAT APPEAR ELSEWHERE IN THIS ANNUAL REPORT. THE FOLLOWING DISCUSSION CONTAINS FORWARD-LOOKING STATEMENTS THAT REFLECT OUR PLANS, ESTIMATES AND BELIEFS. OUR ACTUAL RESULTS COULD DIFFER MATERIALLY FROM THOSE DISCUSSED IN THE FORWARD-LOOKING STATEMENTS. FACTORS THAT COULD CAUSE OR CONTRIBUTE TO SUCH DIFFERENCES INCLUDE THOSE DISCUSSED BELOW AND ELSEWHERE IN THIS ANNUAL REPORT.
| 50 |
FORWARD-LOOKING STATEMENTS
Certain statements made in this report may constitute “forward-looking statements on our current expectations and projections about future events”. These forward-looking statements involve known or unknown risks, uncertainties and other factors that may cause the actual results, performance, or achievements of the Company to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. In some cases you can identify forward-looking statements by terminology such as “may,” “should,” “potential,” “continue,” “expects,” “anticipates,” “intends,” “plans,” “believes,” “estimates,” and similar expressions. These statements are based on our current beliefs, expectations, and assumptions and are subject to a number of risks and uncertainties. Although we believe that the expectations reflected-in the forward-looking statements are reasonable, we cannot guarantee future results, levels of activity, performance or achievements. These forward-looking statements are made as of the date of this report, and we assume no obligation to update these forward-looking statements whether as a result of new information, future events, or otherwise, other than as required by law. In light of these assumptions, risks, and uncertainties, the forward-looking events discussed in this report might not occur and actual results and events may vary significantly from those discussed in the forward-looking statements.
Overview
The Company was incorporated in the State of Nevada, on September 15, 2004, as Thrust Energy Corp. On May 5, 2011, the Company changed its name to American Mining Company. Our principal offices are in Bethesda, Maryland. On May 21st, 2014 the Company changed its name to its current CNBX Pharmaceuticals Inc.
The Company was originally engaged in the exploration, development and production of oil and gas projects within North America, but was unable to operate profitably. In May 2011, the Company suspended its oil and gas operations and changed its business to toll milling and refining and mine development. As of April 2014, the Company has changed its course of business to Biotechnology Pharmaceutical development. As such, the Company has divested itself of its former mining properties.
Financing
We will require additional financing to implement our business plan, which may include joint venture projects and debt or equity financings. The nature of this enterprise and lack of positive cash flow places debt financing beyond the credit-worthiness required by most banks or typical investors of corporate debt until such time as an economically viable profits and losses can be demonstrated. Therefore any debt financing of our activities may be costly and result in substantial dilution to our stockholders.
Future financing through equity investments is likely to be dilutive to existing stockholders. Also, the terms of securities we may issue in future capital transactions may be more favorable for our new investors. Newly issued securities may include preferences, superior voting rights, and the issuance of warrants or other derivative securities, which may have additional dilutive effects. Further, we may incur substantial costs in pursuing future capital and financing, including investment banking fees, legal fees, accounting fees, and other costs. We may also be required to recognize non-cash expenses in connection with certain securities we may issue, such as convertible notes and warrants, which will adversely impact our financial condition.
Our ability to obtain needed financing may be impaired by such factors as the capital markets, both generally and specifically in the bio-pharma industry, and the fact that we have not been profitable to date, which could impact the availability or cost of future financings. If the amount of capital we are able to raise from financing activities, together with our revenue from operations, is not sufficient to satisfy our capital needs, even to the extent that we reduce our operations accordingly, we may be required to cease operations.
There is no assurance that we will be able to obtain financing on terms satisfactory to us, or at all. We do not have any arrangements in place for any future financing. If we are unable to secure additional funding, we may cease or suspend operations. We have no plans, arrangements or contingencies in place in the event that we cease operations.
| 51 |
Results of Operations
Year ended August 31, 2022 compared to the year ended August 31, 2021
Revenues
We had no revenues for the year's ended August 31, 2022, and August 31, 2021.
Operating and Other Expenses
For the year ended August 31, 2022, our total operating expenses were $2,953,970 compared to $2,600,177 for the year ended August 31, 2021. The increase is attributable mainly to general and administrative of $946,313 and decrease in research and development expenditures of $588,252. The increase in the general and administrative expenses attributed mainly to Share based payment expenses of $695,103, Salary expenses of $236,933, Insurance of $36,045, other expenses of $21,735 and decrease in Legal and professional fees of $43,503
We realized financial loss of $769,280 for year August 31, 2022, compared to financial loss of $594,270 for year August 31, 2021. The increase in financial expense was mainly attributable to exchange differences in total of$36,586 and a convertible loan valuation expense of $15,374 and decrease in capital gain of $183,168 and other expenses of $59,000. Asa result, the net loss was $3,723,250 for the year August 31, 2022, compared to a net loss of $3,194,447 for year August 31, 2021.
The net loss for the year ended August 31, 2022, was $3,723, 250 compared to net Income of $3,194, 447 for the year ended August 31, 2021.
Liquidity and Capital Resources
Overview
For the years ended August 31, 2022, as well as August 31, 2021, we funded our operations through issuance of common stock and advances from our majority shareholder. Our principal use of funds during the year ended August 31, 2022, has been for laboratory and clinical research relating to our proprietary materials normative corporate operating expenses.
Liquidity and Capital Resources during the year ended August 31, 2022 compared to the year ended August 31, 2021
As of August 31, 2022, we had $117,515 cash compared to $1,386,472 as of August 31, 2021. The Company used cash in operations of $1,679,192 for the year ended August 31, 2022, compared to cash used in operations of 2,492,913 for the year ended August 31, 2021.
During the year ended August 31, 2022, the Company's investing earned totaled $25,229. This compares to net cash used for investing activities in the year ended August 31, 2021, in the amount of $645,025. The difference reflects primarily of realized held for trading investments and purchase of fixed assets.
During the year ended August 31, 2022, the company had net cash earned from financing activities of $389,858. This compares to $2,456,750 in the year ended August 31, 2021.
Year ended August 31, 2021 compared to the year ended August 31, 2020
Revenues
The revenues for the year ended August 31, 2021 totaled to $7,157 compared to $9,843 for the year ended August 31, 2020.
| 52 |
Operating and Other Expenses
For the year ended August 31, 2021, our total operating expenses were $3,050,609 compared to $3,313,450 for the year ended August 31, 2020. The decrease is attributable mainly to sales and marketing expenses of $228,166, decrease in general and administrative of $173,378, and increase in research and development expenditures of $138,702. The decrease in the sales and marketing expenses attributed mainly to the company PR services of $21,665, had none travel abroad and conferences expenses for the year ended August 31, 2021 comparing to $125,672 and $51,817 respectively for the year ended August 31, 2020.
The net loss for the year ended August 31, 2021, was $7,467, 463 compared to net Income of $1,132,970 for the year ended August 31, 2020.
Liquidity and Capital Resources during the year ended August 31, 2021 compared to the year ended August 31, 2020
As of August 31, 2021, we had $777,611 in cash compared to $265,982 as of August 31, 2020. The Company used cash in operations of $2,774,954 for the year ended August 31, 2021 compared to cash used in operations of $3,093,782 for the year ended August 31, 2020.
During the year ended August 31, 2021, the Company's investing earned totaled $3,286,584. This compares to net cash used for investing activities in the year ended August 31, 2020 in the amount of $5,251,115. The difference reflects primarily of realized held for trading investments and purchase of fixed assets.
During the year ended August 31, 2021, the Company's had no financing activities. This compares to net cash earned from financing activities in the year ended August 31, 2020 in the amount of $7,217,270.
Going Concern
Our independent auditors included an explanatory paragraph in their report on the accompanying consolidated financial statements regarding concerns about our ability to continue as a going concern. Our financial statements contain additional note disclosures describing the circumstances that lead to this disclosure by our independent auditors.
Our financial statements have been prepared on a going concern basis, which assumes the realization of assets and settlement of liabilities in the normal course of business. Our ability to continue as a going concern is dependent upon our ability to generate profitable operations in the future and/or to obtain the necessary financing to meet our obligations and repay our liabilities arising from normal business operations when they become due. The outcome of these matters cannot be predicted with any certainty at this time and raise substantial doubt that we will be able to continue as a going concern. Our financial statements do not include any adjustments to the amount and classification of assets and liabilities that may be necessary should we be unable to continue as a going concern.
There is no assurance that our operations will be profitable. The Company has conducted private placements of its common stock, which have generated funds to satisfy the initial cash requirements of its planned Nevada exploration ventures. Our continued existence and plans for future growth depend on our ability to obtain the additional capital necessary to operate either through the generation of revenue or the issuance of additional debt or equity.
Off-Balance Sheet Arrangements
We currently have no off-balance sheet arrangements that have or are reasonably likely to have a current or future material effect on our financial condition, changes in financial condition, revenues or expenses, results of operations, liquidity, capital expenditures or capital resources.
Item 7A. Quantitative and Qualitative Disclosures About Market Risk.
We are a smaller reporting company as defined by Rule 12b-2 of the Exchange Act and are not required to provide the information required under this item.
| 53 |
Item 8. Financial Statements and Supplementary Data.
| 54 |

REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Shareholders and the Board of Directors of CNBX Pharmaceuticals Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of CNBX Pharmaceuticals Inc. (“the Company”) as of August 31, 2022, and 2021 and the related statements of operations, changes in stockholders' deficit and cash flows, for each of the years ended August 31, 2022 and 2021, and the related notes and schedules (collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of August 31, 2022, and 2021, and the results of its operations and its cash flows for each of the periods ended August 31, 2022 and 2021, in conformity with generally accepted accounting principles in the United States of America.
Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company's financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
Going Concern
The accompanying financial statements have been prepared assuming the Company will continue as a going concern. As more fully described in Note 1, the Company has incurred significant losses and needs to raise additional funds to meet its obligations and sustain its operations. These conditions raise substantial doubt about the Company's ability to continue as a going concern. Management's plans in regard to these matters are also described in Note 1. These financial statements do not include any adjustments that might result from the outcome of this uncertainty.
| F-1 |
Critical Audit Matters
The critical audit matters communicated below are matters arising from the current period audit of the financial statements that were communicated or are required to be communicated to the audit committee and that: (1) relate to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective, or complex judgments. The communication of critical audit matters does not alter in any way our opinion on the financial statements, taken as a whole, and we are not, by communicating the critical audit matters below, providing separate opinions on the critical audit matters or on the accounts or disclosures to which they relate.
Going concern- refer to note 1 of the financial statements
Critical audit matter description
The Company raised a substantial doubt about the entity's ability to continue as a going concern for a reasonable period of time. The financial statements for the years under audit have been prepared assuming that the Company will continue as a going concern, which contemplates the realization of assets and satisfaction of liabilities in the normal course of business. See the explanatory paragraph of the opinion paragraph.
How the Critical Audit Matter was addressed in the Audit
| - | We evaluated whether there is substantial doubt about the entity's ability to continue as a going concern for a reasonable period of time. | |
| - | We obtained information about management's plans that are intended to mitigate the effect of such conditions or events, and assess the likelihood that such plans can be effectively implemented. | |
| - | We added explanatory paragraph to the audit report. |
Convertible Loan-refer to note 6 of the financial Statements
Critical audit matter description
In order to raise funds, the company entered into a Securities Purchase Agreement (“SPA”) with an institutional investor for a private placement of senior secured convertible notes totaling up to an aggregate of $2,750,000 to be issued in three tranches subject to the achievement of certain milestones.
At this time a portion of the loan has been converted to common shares. While the loan is presented on the books as a liability, the company expects that the entire amount will be converted to share capital. If the lender were to request payment in kind the company would possibly not have sufficient to repay the loan, therefore we consider this a critical audit matter/
How the Critical Audit Matter was addressed in the Audit
| - | We evaluated whether it is reasonable to expect the loan to be converted to share capital or if the company will be required to pay cash |
/s/
We have served as the Company's auditor since 2019.
November 29, 2022
| F-2 |
CNBX PHARMACEUTICALS INC.
Audited Consolidated Balance Sheets
| August 31, | August 31, | |||||||
| 2022 | 2021 | |||||||
| ASSETS | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | $ | ||||||
| Prepaid expenses and other receivables | ||||||||
| Total current assets | ||||||||
| Available for sale Investment | ||||||||
| Equipment, net | ||||||||
| Total assets | $ | $ | ||||||
| LIABILITIES AND STOCKHOLDERS' EQUITY | ||||||||
| Current liabilities: | ||||||||
| Accounts payable and accrued liabilities | $ | $ | ||||||
| Convertible loan | ||||||||
| Due to a related party | ||||||||
| Total current liabilities | ||||||||
| Stockholders' equity (deficit): | ||||||||
| Preferred stock, $ par value, shares authorized shares issued and outstanding. | ||||||||
| Common stock, $ par value, shares authorized, and *shares issued and outstanding at August 31, 2022 and August 31, 2021 respectively. | * | |||||||
| Additional paid-in capital | ||||||||
| Issuance of warrants | ||||||||
| Other comprehensive loss | ( | ) | ( | ) | ||||
| Accumulated deficit | ( | ) | ( | ) | ||||
| Total stockholders' equity (deficit) | ( | ) | ||||||
| Total liabilities and stockholders' equity | $ | $ | ||||||
The accompanying notes are an integral part of the financial statements
* On May 12, 2022, the Company effected a reverse-split of its common stock on a 1:120 basis.
| F-3 |
CNBX PHARMACEUTICALS INC.
Audited Consolidated Statements of Operations
| For the Year Ended August 31, | ||||||||
| 2022 | 2021 | |||||||
| Operating expenses: | ||||||||
| Research and development expense | $ | $ | ||||||
| General and administrative expenses | ||||||||
| Total operating expenses | ||||||||
| Loss from operations | ( | ) | ( | ) | ||||
| Other income | ||||||||
| Capital Gain | ||||||||
| Financial (loss) income, net | ( | ) | ( | ) | ||||
| Net (loss) income | $ | ( | ) | $ | ( | ) | ||
| (Loss) Profit from available for sale assets | ( | ) | ||||||
| Total comprehensive (loss) income | ( | ) | ( | ) | ||||
| Net (loss) per share - basic and diluted: | $ | ) | $ | ) | ||||
| Weighted average number of shares outstanding - Basic and Diluted | * | |||||||
The accompanying notes are an integral part of the financial statements.
* On May 12, 2022, the Company effected a reverse-split of its common stock on a 1:120 basis.
| F-4 |
CNBX PHARMACEUTICALS INC.
Audited Consolidated Statements of Cash Flows
| For the year Ended August 31, | ||||||||
| 2022 | 2021 | |||||||
| Cash flows from operating activities: | ||||||||
| Net (Loss) Profit | $ | ( | ) | $ | ( | ) | ||
| Adjustments required to reconcile net loss to net cash used in operating activities: | ||||||||
| Depreciation | ||||||||
| Stock issued for services | ||||||||
| Convertible loan valuation | ||||||||
| Profit from held for trading investments | ||||||||
| Capital Gain | ( | ) | ( | ) | ||||
| Changes in operating assets and liabilities: | ||||||||
| Accounts Receivable and prepaid expenses | ( | ) | ||||||
| Accounts payable and accrued liabilities | ( | ) | ||||||
| Net cash used in operating activities | ( | ) | ( | ) | ||||
| Cash flows from investing activities: | ||||||||
| Sale of equipment | ||||||||
| Available for sale investments | ||||||||
| Acquisition of equipment | ( | ) | ( | ) | ||||
| Net cash used in investing activities | ||||||||
| Cash flows from financing activities: | ||||||||
| Proceeds from issuance of a Convertible loan | ||||||||
| Net cash provided by financing activities | ||||||||
| Net increase (decrease) in cash | ( | ) | ||||||
| Effect of exchange rate fluctuations on cash | ( | ) | ||||||
| Cash and cash equivalents at beginning of year | ||||||||
| Cash and cash equivalents at end of the year | $ | $ | ||||||
| Significant non-cash transactions: | ||||||||
| Exercise of a Convertible loan to shares of common stock. | $ | $ | ||||||
The accompanying notes are an integral part of the financial statements.
| F-5 |
CNBX PHARMACEUTICALS INC.
Audited Consolidated Statements of Stockholders' Equity (Deficit)
| Common stock | Additional paid in | Other comprehensive | Accumulated | Total stockholders’ equity | ||||||||||||||||||||||||
| *Shares | Amount | capital | Loss | Warrants | deficit | (deficit) | ||||||||||||||||||||||
| Balance, August 31, 2020 | $ | $ | $ | ( | ) | $ | $ | ( | ) | $ | ||||||||||||||||||
| Issuance of common stock for services | ||||||||||||||||||||||||||||
| Issuance of warrants | – | |||||||||||||||||||||||||||
| Other comprehensive profit | – | |||||||||||||||||||||||||||
| Exercise of a Convertible loan to shares of common stock. | ||||||||||||||||||||||||||||
| Net loss for the year ended August 31, 2021 | – | ( | ) | ( | ) | |||||||||||||||||||||||
| Balance, August 31, 2021 | $ | $ | $ | ( | ) | $ | $ | ( | ) | $ | ||||||||||||||||||
| Share based Payment | – | |||||||||||||||||||||||||||
| Exercise of a Convertible loan to shares of common stock. | ||||||||||||||||||||||||||||
| Post-split adjustment | ( | ) | ||||||||||||||||||||||||||
| Other comprehensive loss | – | ( | ) | ( | ) | |||||||||||||||||||||||
| Net loss for the year ended August 31, 2022 | – | ( | ) | ( | ) | |||||||||||||||||||||||
| Balance, August 31, 2022 | $ | $ | $ | ( | ) | $ | $ | ( | ) | $ | ( | ) | ||||||||||||||||
The accompanying notes are an integral part of the financial statements.
| F-6 |
CNBX PHARMACEUTICALS INC.
Notes to Consolidated Financial Statements
As of August 31, 2022
Note 1 – Nature of Business, Presentation and Going Concern
Organization
CNBX Pharmaceuticals Inc. (the "Company"), was incorporated in the State of Nevada, on September 15, 2004, under the name of Thrust Energy Corp. The Company was originally engaged in the exploration, exploitation, development and production of oil and gas projects within North America, but was unable to operate profitably.
In May 2011, the Company changed its name to American Mining Corporation, suspending its oil and gas operations and changing its business to toll milling and refining, mineral exploration and mine development.
On April 25, 2014, the Company experienced a change in control. Cannabics, Inc. (“Cannabics”) acquired a majority of the issued and outstanding common stock of the Company in accordance with stock purchase agreements by and between Cannabics and Thomas Mills (“Mills”). On the closing date, April 25, 2014, pursuant to the terms of the Stock Purchase Agreement, Cannabics purchased from Mills 41,000,000 shares of the Company’s outstanding restricted common stock for $198,000, representing 51%.
On May 21, 2014, the Company changed its name, via merger in the state of Nevada, to CNBX Pharmaceuticals Inc. The Company’s principal offices are in Bethesda, Maryland. As of May 21, 2014, the Company has changed its course of business to laboratory research and development.
On August 25, 2014, the Company organized G.R.I.N. Ultra Ltd. (“GRIN”), an Israeli corporation, as a wholly-owned subsidiary. GRIN will provide research and development activities for the Company’s products in Israel.
Stock Split
On June 3, 2014, the Company’s Board of Directors declared a two-to-one forward stock split of all outstanding shares of common stock. The stock split was approved by FINRA on June 25, 2014. The effect of the stock split increased the number of shares of common stock outstanding from 40,880,203 to 81,760,406. All common share and per common share data in these financial statements and related notes hereto have been retroactively adjusted to account for the effect of the stock split for all periods presented prior to June 3, 2014. The total number of authorized common shares and the par value thereof was not changed by the split.
Basis of Presentation
The accompanying consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America and the rules and regulations of the Securities and Exchange Commission (“SEC”).
Going Concern
The accompanying financial statements have been
prepared on a going concern basis, which contemplates the realization of assets and the satisfaction of liabilities in the normal course
of business. The Company has incurred a net loss of $
The Company’s continuation as a going concern is dependent upon its ability to generate revenues, its ability to continue to raise investment capital, and implementing its business plan. No assurance can be given that the Company will be successful in these efforts.
These financial statements do not include any adjustments relating to the recoverability and classification of recorded asset amounts or the amounts and classification of liabilities that might be necessary should the Company be unable to continue as a going concern. Management believes that actions presently being taken to obtain additional funding and implement its strategic plans provide the opportunity for the Company to continue as a going concern. No assurance can be given that the Company will be successful in these efforts.
| F-7 |
Note 2 – Summary of Significant Accounting Policies
Functional currency
The currency of the primary economic environment in which the operations of the Company and its Subsidiary are conducted is the U.S. dollar (“$” or “dollar”). Therefore, the functional currency of the Company and its Subsidiary is the dollar.
Transactions and balances denominated in dollars are presented at their original amounts. Non-dollar transactions and balances have been re-measured to dollars in accordance with the provisions of ASC 830-10 (formerly Statement of Financial Accounting Standard 52), "Foreign Currency Translation". All transaction gains and losses from re-measurement of monetary balance sheet items denominated in non-dollar currencies are reflected in the statement of operations as financial income or expenses, as appropriate.
Use of Estimates
The preparation of financial statements in conformity with accounting principles generally accepted in the United States of America requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from those estimates. Significant estimates in the accompanying financial statements include the amortization period for intangible assets, impairment valuation of intangible assets, valuation of share-based payments and the valuation allowance on deferred tax assets.
Principles of Consolidation
The consolidated financial statements include the accounts of Cannabics Pharmaceutical Inc. and its wholly-owned subsidiary, G.R.I.N. Ultra Ltd. All significant inter-company balances and transactions have been eliminated in consolidation.
Cash and Cash Equivalents
The Company considers all highly liquid temporary cash investments with an original maturity of three months or less to be cash equivalents. At August 31, 2022 and 2020, cash equivalents consisted of bank accounts held at financial institutions.
Concentration of Credit Risk
The Company places its cash and cash equivalents with high credit quality financial institutions. There is Federal Deposit Insurance on the Company’s U.S. bank accounts.
Equipment, net
Equipment at August 31, 2022 consists of computer equipment, office equipment and cars recorded at cost. Expenditures for major additions and betterments are capitalized. Maintenance and repairs are charged to operations as incurred. Depreciation of property and equipment is computed by the straight-line method (after taking into account their respective estimated residual values) over the assets estimated useful lives of 3 years for computer equipment, 14 years for office equipment and 7 years for cars. Upon sale or retirement of property and equipment, the related cost and accumulated depreciation are removed from the accounts and any gain or loss is reflected in the consolidated statements of operations.
Depreciation expense was $
Revenue recognition
Revenue is recognized when delivery has occurred, evidence of an arrangement exists, title and risks and rewards for the products are transferred to the customer, collection is reasonably assured and product returns can be reliably estimated.
Revenue from license agreements is recognized over the periods from which the Company is entitled to the respective payments.
| F-8 |
Impairment or Disposal of Long-Lived Assets
The Company accounts for the impairment or disposal of long-lived assets according to the Financial Accounting Standards Board’s (“FASB”) Accounting Standards Codification (“ASC”) 360 “Property, Plant and Equipment”. ASC 360 clarifies the accounting for the impairment of long-lived assets and for long-lived assets to be disposed of, including the disposal of business segments and major lines of business. Long-lived assets are reviewed when facts and circumstances indicate that the carrying value of the asset may not be recoverable. When necessary, impaired assets are written down to estimated fair value based on the best information available. Estimated fair value is generally based on either appraised value or measured by discounting estimated future cash flows. Considerable management judgment is necessary to estimate discounted future cash flows. Accordingly, actual results could vary significantly from such estimates.
Fair Value of Financial Instruments
The following provides an analysis of financial instruments that are measured subsequent to initial recognition at fair value, grouped into Levels 1 to 3 based on the degree to which fair value is observable:
Level 1 - fair value measurements are those derived from quoted prices (unadjusted) in active markets for identical assets or liabilities;
Level 2 - fair value measurements are those derived from inputs other than quoted prices included within Level 1 that are observable for the asset or liability, either directly (i.e. as prices) or indirectly (i.e. derived from prices); and
Level 3 - fair value measurements are those derived from valuation techniques that include inputs for the asset or liability that are not based on observable market data (unobservable inputs).
Fair value estimates discussed herein are based upon certain market assumptions and pertinent information available to management as of August 31, 2022 and 2021. The respective carrying value of certain on-balance-sheet financial instruments approximated their fair values due to the short-term nature of these instruments.
The Company applied ASC 820 for all non-financial assets and liabilities measured at fair value on a non-recurring basis. The adoption of ASC 820 for non-financial assets and liabilities did not have a significant impact on the Company’s financial statements.
As of August 31, 2022, the fair values of the
Company’s level 1 financial instruments assets are in total of $
As of August 31, 2022, the fair values of the Company’s financial instruments approximate their historical carrying amount.
Research and development, net
Research and development expenses include costs directly attributable to the conduct of research and development programs, including the cost of salaries, employee benefits, the cost of supplies, the cost of services provided by outside contractors, including services related to the Company’s clinical trials, clinical trial expenses and the full cost of manufacturing product for use in research and preclinical development. All costs associated with research and developments are expensed as incurred.
Clinical trial costs are a significant component of research and development expenses and include costs associated with third-party contractors. The Company outsources a substantial portion of its clinical trial activities, utilizing external entities such as Contract Research Organizations, independent clinical investigators, and other third-party service providers to assist the Company with the execution of its clinical studies. For each clinical trial that the Company conducts, clinical trial costs are expensed immediately.
| F-9 |
The Company accounts for Stock-Based Compensation under ASC 718 “Compensation – Stock Compensation”, which addresses the accounting for transactions in which an entity exchanges its equity instruments for goods or services, with a primary focus on transactions in which an entity obtains employee services in share-based payment transactions. ASC 718-10 requires measurement of the cost of employee services received in exchange for an award of equity instruments based on the grant-date fair value of the award. Incremental compensation costs arising from subsequent modifications of awards after the grant date must be recognized.
The Company accounts for stock-based compensation awards to non-employees in accordance with ASC 505-50, Equity-Based Payments to Non-Employees. Under ASC 505-50, the Company determines the fair value of the warrants or stock-based compensation awards granted as either the fair value of the consideration received or the fair value of the equity instruments issued, whichever is more reliably measurable. Any stock options or warrants issued to non-employees are recorded in expense and additional paid-in capital in shareholders' deficit over the applicable service periods using variable accounting through the vesting dates based on the fair value of the options or warrants at the end of each period.
The Company issues stock to consultants for various services. The costs for these transactions are measured at the fair value of the consideration received or the fair value of the equity instruments issued, whichever is more reliably measurable. The value of the common stock is measured at the earlier of (i) the date at which a firm commitment for performance by the counterparty to earn the equity instruments is reached or (ii) the date at which the counterparty's performance is complete. The Company recognized consulting expense and a corresponding increase to additional paid-in-capital related to stock issued for services.
Income Taxes
Income taxes are accounted for under the liability method of accounting for income taxes. Under the liability method, future tax liabilities and assets are recognized for the estimated future tax consequences attributable to differences between the amounts reported in the financial statement carrying amounts of assets and liabilities and their respective tax bases. Future tax assets and liabilities are measured using enacted or substantially enacted income tax rates expected to apply when the asset is realized or the liability settled. The effect of a change in income tax rates on future income tax liabilities and assets is recognized in income in the period that the change occurs. Future income tax assets are recognized to the extent that they are considered more likely than not to be realized.
The FASB has issued ASC 740 “Income Taxes”. ASC 740 clarifies the accounting for uncertainty in income taxes recognized in an enterprise’s financial statements. This standard requires a company to determine whether it is more likely than not that a tax position will be sustained upon examination based upon the technical merits of the position.
If the more-likely-than-not threshold is met, a company must measure the tax position to determine the amount to recognize in the financial statements.
As a result of the implementation of this standard, the Company performed a review of its material tax positions in accordance with recognition and measurement standards established by ASC 740 and concluded that the tax position of the Company has not met the more-likely-than-not threshold as of August 31, 2022.
Comprehensive Income
The Company adopted ASC 220, Comprehensive Income which establishes standards for reporting and display of comprehensive income, its components and accumulated balances. The Company is disclosing this information on its Statement of Stockholders' Equity. Comprehensive income comprises equity except those resulting from investments by owners and distributions to owners. The Company has no elements of “other comprehensive income” for the years ended August 31, 2022, and 2021.
| F-10 |
The Company computes income (loss) per share in accordance with ASC 260, “Earnings per Share”, which requires presentation of both basic and diluted earnings per share (“EPS”) on the face of the statement of operations. Basic EPS is computed by dividing income (loss) available to common shareholders by the weighted average number of shares outstanding during the period. Diluted EPS gives effect to all dilutive potential shares of common stock outstanding during the period using the treasury stock method and convertible preferred stock using the if-converted method. In computing diluted EPS, the average stock price for the period is used in determining the number of shares assumed to be purchased from the exercise of stock options or warrants. Diluted EPS excludes all dilutive potential shares if their effect is anti-dilutive. As of August 31, 2022, and 2020, the potentially dilutive shares were anti-dilutive.
Segment Information
In accordance with the provisions of ASC 280-10, “Disclosures about Segments of an Enterprise and Related Information”, the Company is required to report financial and descriptive information about its reportable operating segments. The Company does not consider itself to have any operating segments as of August 31, 2022, and 2021.
Reclassification
Certain amounts in the prior period financial statements have been reclassified to conform to the current period presentation.
Note 3 – Recent Accounting Pronouncements
In February 2015, the Financial Accounting Standards Board (the “FASB”) issued Accounting Standards Update (“ASU”) 2015-02, “Consolidation (Topic 810): Amendments to the Consolidation Analysis”, which provides guidance in evaluating entities for inclusion in consolidations. ASU 2015-02 is effective for fiscal years beginning after December 15, 2015. The Company does not believe the adoption of ASU 2015-02 will have a material effect on its consolidated financial statements.
In June 2014, the FASB issued Accounting Standards Update (“ASU”) No. 2014-10 (“ASU 2014-10”), Development Stage Entities (Topic 915), Elimination of Certain Financial Reporting Requirements, Including an Amendment to Variable Interest Entities Guidance in Topic 810, Consolidation. The objective of the amendments in this Update is to improve financial reporting by reducing the cost and complexity associated with incremental reporting requirements for development stage entities. The amendments in this Update also eliminate an exception provided to development stage entities in Topic 810, Consolidation, for determining whether an entity is a variable interest entity on the basis of the amount of investment equity at risk. The amendments related to the elimination of inception-to-date information and the other remaining disclosure requirements of Topic 915 should be applied retrospectively except for the clarification to Topic 275, which shall be applied prospectively. These amendments are effective for annual reporting periods beginning after December 15, 2014, and interim periods therein.
The amendment eliminating the exception to the sufficiency-of-equity-at-risk criterion for development stage entities in paragraph 810-10-15-16 should be applied retrospectively for annual reporting periods beginning after December 15, 2015 and interim periods therein. Early application of each of the amendments is permitted for any annual reporting period or interim period for which the entity’s financial statements have not yet been issued. The Company has adopted ASU 2014-10 in the fourth quarter of 2014 and does not expect this adoption to have a material impact on its consolidated financial condition, results of operations or cash flows.
In August 2014, the FASB issued ASU No. 2014-15, Presentation of Financial Statements-Going Concern (Subtopic 205-40), Disclosure of Uncertainties about an Entity’s Ability to Continue as a Going Concern. The objective of the amendments in this Update is to provide guidance on determining when and how to disclose going-concern uncertainties in the financial statements. The new standard requires management to perform interim and annual assessments of an entity’s ability to continue as a going concern within one year of the date the financial statements are issued. An entity must provide certain disclosures if “conditions or events raise substantial doubt about the entity’s ability to continue as a going concern.” The ASU applies to all entities and is effective for annual periods ending after December 15, 2017, and interim periods thereafter, with early adoption permitted. The Company is evaluating the impact of ASU 2014-15 on its consolidated financial condition, results of operations and cash flows.
| F-11 |
In September 2015, the Financial Accounting Standards Board (“FASB”) issued Accounting Standards Update No. 2015-16 (ASU 2015-16) "Simplifying the Accounting for Measurement Period Adjustments". ASU 2015-16 require that an acquirer recognize adjustments to provisional amounts that are identified during the measurement period in the reporting period in which the adjustment amounts are determined. The amendments in ASU 2015-16 require that the acquirer record, in the same period’s financial statements, the effect on earnings of changes in depreciation, amortization, or other income effects, if any, as a result of the change to the provisional amounts, calculated as if the accounting had been completed at the acquisition date. The amendments in ASU 2015-16 require an entity to present separately on the face of the income statement or disclose in the notes the portion of the amount recorded in current-period earnings by line item that would have been recorded in previous reporting periods if the adjustment to the provisional amounts had been recognized as of the acquisition date. For public business entities, the amendments in ASU 2015-16 are effective for fiscal years beginning after December 15, 2015, including interim periods within those fiscal years. The amendments in ASU 2015-16 should be applied prospectively to adjustments to provisional amounts that occur after the effective date of this update with earlier application permitted for financial statements that have not been issued. For all other entities, the amendments in ASU 2015-16 are effective for fiscal years beginning after December 15, 2017, and interim periods within fiscal years beginning after December 15, 2017. The amendments in ASU 2015-16 should be applied prospectively to adjustments to provisional amounts that occur after the effective date of this update with earlier application permitted for financial statements that have not yet been made available for issuance.
Note 4 – Related Party Transactions
During the year ended August 31, 2022, and August
31, 2021, the Company paid approximately of $
Cannabics Inc. (the parent company) balance as
of August 31, 2022, and at August 31, 2021 was $
During the year ended August 31, 2022, the Company recorded a non cash expense of $ as share based payment, to the company chairman, board members and advisor, compared to for the year ended August 31, 2021.
Note 5 – Commitments and Contingencies
As security for its obligation under a property
lease agreement, cars lease and credit cards of the Company’s subsidiary provided a bank guarantee in the amount of $
Note 6 – Stockholders’ Equity (Deficit)
Authorized Shares
The Company is authorized to issue up to shares of common stock par value $0 per share. Each outstanding share of common stock entitles the holder to one vote per share on all matters submitted to a stockholder vote. All shares of common stock are non-assessable and non-cumulative, with no pre-emptive rights. The Company’s initial Articles authorized preferred shares at par value, no other attributes have been assigned and no such shares have ever been issued.
Common Stock
On May 12, 2022, the Company effected a reverse-split
of its common stock on a
During the year ended August 31, 2022, the
Company issued
(reverse split) shares of its common stock to an investor as a result of a convertible loan exercise in the sum of $
| F-12 |
Stock Based Compensation
On October 6, 2021, the Compensation Committee adopted, and the Board ratified the Company’s Equity Incentive Plan. The Plan is intended to secure for the Company the benefits arising from ownership of the Company’s common stock by the employees, officers, directors and consultants of the Company, all of whom are, and will be, responsible for the Company’s future growth. The Plan is designed to help attract and retain for the Company, qualified personnel for positions of exceptional responsibility, to reward employees, officers, directors and consultants for their services to the Company and to motivate such individuals through added incentives to further contribute to the success of the Company.
The Plan provides an opportunity for an employee, officer, director or consultant of the Company, subject to certain national securities and taxation laws, to receive (i) incentive stock options, (ii) nonqualified stock options; (iii) restricted stock; (iv) stock awards; (v) shares in performance of services; or (vi) any combination of the foregoing. Incentive stock options granted under the Plan are intended to qualify as “incentive stock options” within the meaning of Section 422 of the Internal Revenue Code of 1986, as amended (the “Code”). Nonqualified (non-statutory stock options) granted under the Plan are not intended to qualify as incentive stock options under the Code. The Plan is administered by the Altshuler Shaham Benefits Firm, a licensed fiduciary.
During November 2021, the company issued options to directors
and a consultant, the fair value of the said issuance was $
The fair value of the warrants are estimated using the Black Scholes option-pricing model with the following assumptions:
| PV of exercise Share price | $ | |
| Expected Volatility | % | |
| Risk Free Interest Rate | % | |
| Expected Term (years) | - | |
| Expected Dividend Yield | % |
On May 2, 2022, the Compensation Committee approved amending the Plan to increase the number of shares reserved for issuance under the Plan to 15% of the common stock outstanding immediately following consummation of this offering.
Private Placement of Notes and Warrant
On December 16, 2020, we entered into a Securities
Purchase Agreement (“SPA”) with an institutional investor for a private placement of senior secured convertible notes totaling
up to an aggregate of $
On December 21, 2020, we closed the first tranche
and issued a note in the amount of $
On April 23, 2021, we entered into a senior secured
promissory note (the “Senior Secured Note”) for $
| F-13 |
On February 15, 2022, we entered into a forbearance agreements with the institutional investor relating to that certain Senior Secured Note. Pursuant to the forbearance agreement, the investor, through March 7, 2022, agreed to forbear from exercising any rights and remedies against the Company related to the outstanding payments and to waive certain other defaults under the Senior Secured Note and related rights pursuant to the registration rights agreement entered into in December 2020 between the Company and the investor.
On November 28, 2022, we entered into a forbearance agreements with the institutional investor relating to that certain Senior Secured Note. Pursuant to the forbearance agreement, the investor, through December 12, 2022, agreed to forbear from exercising any rights and remedies against the Company related to the outstanding payments and to waive certain other defaults under the Senior Secured Note and related rights pursuant to the registration rights agreement entered into in December 2020 between the Company and the investor.
On March 16, 2022, we issued to the investor a
demand promissory note (the “Demand Note”) in the principal amount of $
On November 28, 2022, we entered into a forbearance agreements with the institutional investor relating to that certain Senior Secured Note. Pursuant to the forbearance agreement, the investor, through December 12, 2022, agreed to forbear from exercising any rights and remedies against the Company related to the outstanding payments and to waive certain other defaults under the Senior Secured Note and related rights pursuant to the registration rights agreement entered into in May 16, 2022 between the Company and the investor.
On June 15, 2022, the Company entered into a Securities
Purchase Agreement providing for the issuance of the Convertible Promissory Note in the principal amount of $
Note 7 – Warrants
| 1. | On September
24, 2018, as part of a securities purchase agreement the company issued Warrants, to purchase common shares of the Company
at $1.00 per share; said Warrants are valid for five years, expiring on | |
| The fair value of each warrant is approximately $0.556 and the total value of the 5,000,000 warrants is $ | ||
| The fair value of the warrants are estimated using the Black Scholes option-pricing model with the following assumptions: |
| PV of exercise Share price | $ | |
| Expected Volatility | % | |
| Risk Free Interest Rate | % | |
| Expected Term (years) | ||
| Expected Dividend Yield | % |
| 1. | On December
16, 2020, as part of a convertible loan agreement the company issued Warrants, to purchase common shares of the Company
at $0.50 per share; said Warrants are valid for three years, expiring on | |
| The fair value of the first, second and third trenches are approximately $0.09, $0.09 and $0.1 respectively and the total value of the 5,500,000 warrants is $ | ||
| The fair value of the warrants are estimated using the Black Scholes option-pricing model with the following assumptions: |
| PV of exercise Share price | $ | |
| Expected Volatility | % | |
| Risk Free Interest Rate | % | |
| Expected Term (years) | ||
| Expected Dividend Yield | % |
| F-14 |
| 2022 | 2021 | 2020 | ||||||||||||||||||||||
| Weighted Average Exercise | Weighted Average Exercise | Weighted Average Exercise | ||||||||||||||||||||||
| Warrants | Price | Warrants | Price | Warrants | Price | |||||||||||||||||||
| Warrants outstanding as of September 1 | $ | $ | $ | |||||||||||||||||||||
| Issued | $ | $ | $ | |||||||||||||||||||||
| Exercised | $ | $ | $ | |||||||||||||||||||||
| Expired | $ | $ | ( | ) | $ | |||||||||||||||||||
| Warrants outstanding as of August 31 | $ | $ | $ | |||||||||||||||||||||
| Warrants exercisable as of August 31 | $ | $ | $ | |||||||||||||||||||||
The weighted average remaining contractual life for the options outstanding as of August 31, 2022, was years.
Note 8 – Income Taxes
Taxes on income included in the consolidated statements of operations represent current taxes due to taxable income of the Company and its Subsidiary.
Corporate taxation in the U.S.
The applicable corporate tax rate for the Company
is
No provision for income tax was made for the period
from September 15, 2004 (Inception) to August 31, 2022, as the Company had cumulative operating losses. For the years ended August 31,
2022, and 2021, the Company incurred net losses for tax purposes of $
Corporate taxation in Israel:
The Subsidiary is taxed in accordance with Israeli
tax laws. The corporate tax rate applicable is
As of August 31, 2022, the Subsidiary has an accumulated
tax loss carry forward of approximately $
The income tax expense (benefit) differs from the amount computed by applying the United States Statutory corporate income tax rate as follows:
| For the Year Ended August 31, | ||||||||
| 2022 | 2021 | |||||||
| United States statutory corporate income tax rate | ||||||||
| Change in valuation allowance on deferred tax assets | - | - | ||||||
| Provision for income tax | ||||||||
| F-15 |
Deferred income taxes reflect the tax effect of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for tax purposes. The components of the net deferred income tax assets are approximately as follows:
| August 31, | ||||||||
| 2022 | 2021 | |||||||
| US Deferred income tax assets: | ||||||||
| Net operating loss carry forwards benefit | $ | $ | ||||||
| Valuation allowance | ( | ) | ( | ) | ||||
| Net deferred income tax assets | $ | $ | ||||||
| Outside US Deferred income tax assets: | ||||||||
| Net operating loss carry forwards benefit | $ | $ | ||||||
| Valuation allowance | ( | ) | ( | ) | ||||
| $ | $ | |||||||
| August 31, | ||||||||
| 2022 | 2021 | |||||||
| Consolidated Deferred income tax assets: | ||||||||
| Net operating loss carry forwards benefit | $ | $ | ||||||
| Valuation allowance | ( | ) | ( | ) | ||||
| Net deferred income tax assets | $ | $ | ||||||
The amount taken into income as deferred income
tax assets must reflect that portion of the income tax loss carry forwards that is more likely than not to be realized from future operations.
The Company has established a full valuation allowance on its net deferred tax assets because of a lack of sufficient positive evidence
to support its realization. The valuation allowance increased by $
No provision for income taxes has been provided
in these financial statements due to the net loss for the years ended August 31, 2022 and 2021. At August 31, 2022, the Company has net
operating loss carry forwards of approximately $
IRS Section 382 places limitations (the “Section 382 Limitation”) on the amount of taxable income which can be offset by net operating loss carry forwards after a change in control (generally greater than 50% change in ownership) of a loss corporation. Generally, after a change in control, a loss corporation cannot deduct operating loss carry forwards in excess of the Section 382 Limitation. Due to these “change in ownership” provisions, utilization of the net operating loss and tax credit carry forwards may be subject to an annual limitation regarding their utilization against taxable income in future periods. The Company has not concluded its analysis of Section 382 through August 31, 2022, but believes the provisions will not limit the availability of losses to offset future income.
The Company is subject to income taxes in the U.S. federal jurisdiction and is subject to examination for a period of three years for current filings and indefinitely for any delinquent filings. The tax regulations within each jurisdiction are subject to interpretation of related tax laws and regulations and require significant judgment to apply. The Company estimates that the amount of penalties, if any, will not have a material effect on the results of operations, cash flows or financial position. No provisions have been made in the financial statements for such penalties, if any.
| F-16 |
Note 9 – Subsequent Events
On September 28th, 2022, the Company announced the grant of its Australian Patent “System and Method for High Throughput Screening of Cancer Cells”
On September 29th, 2022, the Company announced the peer-reviewed publication of its sponsored preclinical study titled "The Effect of Isolated Cannabinoid Extracts on Polypoid Colorectal Tissue" in the International Journal of Molecular Sciences;
On November 8th, 2022 the Company announced that it has acquired controlling interest in TaGeza Biopharmaceuticals Ltd. The TaGeza research team remains committed to the Company and co-founder Prof. Benjamin Dekel (MD, PhD) will continue to serve as the Company Chief Scientist; Gabriel Yariv, to assume deposition of TaGeza Biopharmaceuticals CEO.
The Company has evaluated subsequent events through the date the financial statements were issued and filed with the Securities and Exchange Commission and has determined that there are no other such events that warrant disclosure or recognition in the financial statements.
Note 10 – General & administrate expenses
| For the year Ended August 31, 2022 | For the year Ended August 31, 2021 | |||||||
| Salaries and related expenses | $ | $ | ||||||
| Share based payment | ||||||||
| Legal and professional fees | ||||||||
| Insurance | ||||||||
| Marketing expenses | ||||||||
| Other expenses | ||||||||
| Total general and administrative expenses | $ | $ | ||||||
Note 11 – Financial (Income) expenses
| For the year Ended August 31, 2022 | For the year Ended August 31, 2021 | |||||||
| Interest and bank charges | ||||||||
| Capital gain from selling of fixed assets | ( | ) | ||||||
| Capital gain from selling of available for sale investment | ( | ) | ||||||
| Loss (Gain) from convertible loan valuation | ||||||||
| Currency exchange differences loss (profit) | ||||||||
| Other financial expense | ||||||||
| Total other income (expense) | $ | $ | ||||||
| F-17 |
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
None.
Item 9A. Controls and Procedures
Evaluation of Disclosure Controls and Procedures
Disclosure Controls and Procedures
The Company maintains a set of disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) of the Exchange Act) designed to ensure that information required to be disclosed by the Company in reports that it files or submits under the Exchange Act, is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms and is accumulated and communicated to the Company’s management, including the Chief Executive Officer and Chief Financial Officer, as appropriate, to allow timely decisions regarding required disclosure. In accordance with Rule 13a-15(b) of the Exchange Act, as of the end of the period covered by this Annual Report on Form 10-K, an evaluation was carried out under the supervision and with the participation of the Company’s management, including its Chief Executive Officer, Chief Financial Officer and the Audit Committee, of the effectiveness of its disclosure controls and procedures. The Audit Committee assessed, reviewed and determined that the Company’s disclosure controls and procedures were effective as to this annual filing. Based on that evaluation, The Board accepted and ratified the findings of the Audit Committee that the Company’s disclosure controls and procedures, as of August 31, 2022, the end of the period covered by this Annual Report on Form 10-K, were effective to provide reasonable assurance that information required to be disclosed by the Company in reports that it files or submits under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms and is accumulated and communicated to the Company’s management, including the Chief Executive Officer, Chief Financial Officer, and Audit Committee as appropriate to allow timely decisions regarding required disclosure.
Management’s Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) of the Exchange Act). Internal control over financial reporting is a process designed by, or under the supervision of, the issuer’s principal executive and principal financial officers, or persons performing similar functions, and effected by the issuer’s board of directors, management and other personnel, to provide reasonable assurance regarding the reliability of our financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. Internal control over financial reporting includes those policies and procedures that (i) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the Company; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the Company are being made only in accordance with authorizations of management and directors of the Company; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the Company’s assets that could have a material effect on the financial statements. Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that our degree of compliance with the policies or procedures may deteriorate.
Management assessed the effectiveness of the Company’s internal controls over financial reporting as of August 1, 2022, based on the framework in Internal Control—Integrated Framework (COSO 2013) issued by the Committee of Sponsoring Organization of the Treadway Commission. On the basis of that assessment, management determined that our internal controls over financial reporting were effective as of that date.
Changes in Internal Control Over Financial Reporting
Since our previous annual report, the Company has created an Audit Committee to better review our internal financial reporting. There were no other changes in our internal control over financial reporting during the year ended August 31, 2022, that materially affected, or are reasonably likely to materially affect, our internal control over financial reporting
| 55 |
Limitations on the Effectiveness of Controls
Our disclosure controls and procedures and internal control over financial reporting are designed to provide reasonable assurance of achieving their objectives as specified above. Management does not expect, however, that our disclosure controls and procedures or our internal control over financial reporting will prevent or detect all error and fraud. Any control system, no matter how well designed and operated, is based upon certain assumptions and can provide only reasonable, not absolute, assurance that its objectives will be met. Further, no evaluation of controls can provide absolute assurance that misstatements due to error will not occur or that all control issues and instances of inaccurate entries, if any, within the Company have been detected.
Item 9B. Other Information
None.
| 56 |
PART III
Item 10. Directors, Executive Officers, Promoters and Control Persons; Compliance With Section 16(A) of the Exchange Act
The following individuals serves as Directors and Executive Officers of the Company as of the date of this Annual Report. Directors of the Company hold office until the next annual meeting of our shareholders or until their successors have been elected and qualified. Executive officers of the Company are appointed by our board of directors and hold office until their death, resignation or removal from office.
Director Independence
The Board of Directors annually determines the independence of each director and nominee for election as a director. The Board makes these determinations in accordance with The NASDAQ Capital Market’s (“NASDAQ’s”) listing standards for the independence of Directors and the SEC’s rules.
In assessing director independence, the Board considers, among other matters, the nature and extent of any business relationships, including transactions conducted, between the Company and each director and between the Company and any organization for which one of our Directors is a Director or executive officer or with which one of our directors is otherwise affiliated.
The Board has affirmatively determined that Mr. Shaul Yemal, Mr. Gil Feiler, and Inbar Maymon are independent.
In accordance with NASDAQ requirements, our Board is comprised of a majority of independent Directors and the Compensation and Audit Committees are comprised entirely of independent Directors.
| Name | Position | Age | Held Position Since | |||
| Sanja Goldberg | CTO | 46 | July 24, 2022 | |||
| Uri Ben-Or | CFO | 50 | November 05, 2016 | |||
| Inbar Maymon | Independent Director | 44 | July 1, 2021 | |||
| Gil Feiler | Independent Director | 62 | July 15, 2021 | |||
| Shaul Yemal | Independent Director | 76 | November 16, 2021 | |||
| Eyal Barad | Director, CEO | 57 | November 13, 2018 | |||
| Gabriel Yariv | Director, Chair of Board | 45 | October 2, 2019 |
Officer Qualifications:
Dr. Sanja Goldberg has held several Senior Researcher positions at Israel’s largest hospital, Sheba Medical Center’s Cancer Research Lab and Regenerative Research Lab. Additionally she has had several Executive positions, including at Compugen and at TaGeza Biopharma where she was VP of Research and Development. Dr. Goldberg obtained her Ph.D. in Cell Biology from the Department of Cell Biology and Genetics, Erasmus MC, Rotterdam, the Netherlands, in 2009, and has since led cutting edge science in her positions related to cancer and early-stage drug discovery activities.
Uri Ben-Or, is our CFO. Mr. Ben-Or is a licensed CPA, MBA and has More than 15 years of experience as CFO of public and private companies in the life science and Medical Device Industry. Uri has significant expertise in public Life Science companies traded on the TASE. In addition, Uri has strong finance, operation, and business development background in both startups and public global companies in the US, Europe, and Israel including developing strategic policy and guidance with respect to corporate structure and fundraising.
Eyal Barad, is a co-founder, and our Director and CEO. Mr. Barad brings over 20 years of executive managerial experience in successful technology ventures and was a pioneer in the digital publishing industry in NYC. He has a BA in Economics & International Relations from the Hebrew University in Jerusalem, and an MBA with Honors from Haifa University.
| 57 |
Gabriel Yariv is our Chair and brings over 20 years of successful executive experience in the medical industry. Mr. Yariv was part of the founding group of BreathID, an Oridion Medical business unit (now Medtronic) and its subsequent spinoff company, Exalenz Bioscience, which develops and manufactures advanced non-invasive diagnostic medical devices for gastrointestinal and liver conditions. Mr. Yariv also co-founded SimuTec, a medical simulation and training company in Brazil that develops and commercializes advanced personalized Virtual Reality training programs for physicians. Mr. Yariv is actively engaged in non-profit and philanthropic activities including ongoing business mentoring of entrepreneurs, founder of the Yariv Foundation for Leadership, and current member of the Friends of the Israel Museum society. Mr. Yariv holds a BA (Cum Laude) in History, Philosophy & Political science from Boston University.
Dr. Gil Feiler, is an Independent Director and sits on the Compensation Committee. Mr. Feiler is a Director of the Bank of Georgia, formerly Board member of First International Bank of Israel, advisor for the government of Ras Al Khaimah, UAE, Alumni of the US State Dept International Visitor Leadership Program, as well as known author of numerous publications on regional economics and business relations. Mr. Feiler holds a BA, master’s degree and PhD from University of Tel Aviv. Mr. Feiler is also the head of the company’s Advisory Board.
Dr. Inbar Maymon-Pomeranchik is an Independent Director and sits on both our Compensation and Audit Committees. Dr. Maymon-Pomeranchik is the founder & CEO of AgChimedes Group Ltd., a company focused on the convergence of science, innovation and investment in the field of food security; she also founded BioDiligence, a biotech investment consultancy; Executive Director of Ananda Developments Pls (UK), an investment and operational firm that targets medicinal cannabis derivatives for research and development; a Director of NRGene (Israel), a company that develops and commercializes cutting-edge AI based genomic tools, and an Advisory Board member of Avida Global Ltd. (UK/Columbia), Avida is a leading licensed producer of pharmaceutical grade cannabis extract for scientific and medical use. Dr. Maymon-Pomeranchik holds a PhD in plants science and molecular biology from the Hebrew University (2008) and a Post Doctorate from the Weizmann Institute (2010).
Shaul Yemal is an Independent Director and heads our Audit Committee. Mr. Yemal served as Senior Economist in the Division of Economic Consulting for the Ministry of Finance, Budget Division of the Minister of Finance and Manager of the Economic and Planning Division of the Ministry of Energy (Israel). Mr. Yemal was acting Chair of Financial, Auditor and Compensation Committees for numerous companies both public and private. Mr. Yemal was CEO of ICL in Santiago, Chile, Senior VP of Business Development for ISAL Amlat VC Fund and was responsible for their IPO in the Tel Aviv stock exchange; Senior VP Corporate Manager and Board Member of Oridion Medical, and catalyst for their successful IPO on the Swiss stock exchange. Mr. Yemal holds a Masters Degree in Economics from University of Rosario, Argentina.
All directors serve for terms of one year each and are subject to re-election at Annual Meeting of Shareholders, unless they earlier resign.
There are no material proceedings to which any of our directors, officers or affiliates, any owner of record or beneficially of more than five percent of any class of our voting securities, or any associate of any such director, officer, affiliate, or security holder is a party adverse to us or any of our subsidiaries or has a material interest adverse to us or any of our subsidiaries.
We have attempted and will continue to attempt to ensure that any transactions between we and our officers, directors, principal shareholders, or other affiliates have been and will be on terms no less favorable to us than could be obtained from unaffiliated third parties on an arm’s length basis.
Committees of the Board
We currently maintain an Audit Committee, and Compensation Committee, which have the committee members described supra. Our two standing Committees are comprised of senior executives and independent directors who are actively involved in the disclosure process, to specify, coordinate and oversee the public disclosure of information regarding the Company, including periodic and current report filings with the SEC.
Audit Committee
The Audit Committee is tasked with reviewing and approving any issues relating to conflicts of interests and all related party transactions of the Company (“Related Party Transactions”). The Audit Committee, in undertaking such review and approval, will analyze the following factors, in addition to any other factors the Audit Committee deems appropriate, in determining whether to approve a Related Party Transaction: (1) the fairness of the terms for the Company (including fairness from a financial point of view); (2) the materiality of the transaction; (3) bids / terms for such transaction from unrelated parties; (4) the structure of the transaction; (5) the policies, rules and regulations of the U.S. federal and state securities laws; (6) the policies of the Committee; and (7) interests of each related party in the transaction.
| 58 |
The Audit Committee will only approve a Related Party Transaction if the Audit Committee determines that the terms of the Related Party Transaction are beneficial and fair (including fair from a financial point of view) to the Company and are lawful under the laws of the United States. In the event multiple members of the Audit Committee are deemed a related party, the Related Party Transaction will be considered by the disinterested members of the board of directors in place of the Committee. Our Audit Committee is headed by Shaul Yemal, who, by virtue of his years of experience as a Senior Economist at the Ministry of Finance, is clearly a “financial expert" as defined in Item 407(d)(5) of Regulation S-K,
The Board has also determined that Mr. Yemal is an “audit committee financial expert” (as defined in the SEC rules) because he has the following attributes: (i) an understanding of generally accepted accounting principles in the United States of America (“GAAP”) and financial statements; (ii) the ability to assess the general application of such principles in connection with accounting for estimates, accruals and reserves; (iii) experience analyzing and evaluating financial statements that present a breadth and level of complexity of accounting issues that are generally comparable to the breadth and complexity of issues that can reasonably be expected to be raised by our financial statements; (iv) an understanding of internal control over financial reporting; and (v) an understanding of audit committee functions. Mr. Yemal has acquired these attributes by means of having held various positions that provided relevant experience, as described in his biographical supra.
Compensation Committee
The Compensation Committee, which is comprised exclusively of independent directors, is responsible for the administration of our stock compensation plans, approval, review and evaluation of the compensation arrangements for our executive officers and directors and oversees and advises the Board on the adoption of policies that govern the Company’s compensation and benefit programs. In addition, the Compensation Committee has the authority, at its discretion and at our expense, to retain special legal, accounting or other advisors to advise the Compensation Committee. The Compensation Committee convened, proposed, and the Board ratified the Compensation Committee Charter on November 25th, 2021.
Compensation Committee Interlocks and Insider Participation.
The current members of the Compensation Committee are Dr. Gil Feiler and Dr. Maymon, who are each independent members of our board of Directors. No member of the Compensation Committee is an employee or a former employee of the Company. During fiscal 2021, none of our executive officers served on the compensation committee (or its equivalent) or board of directors of another entity whose executive officer served on our Compensation Committee. Accordingly, the Compensation Committee members have no interlocking relationships required to be disclosed under SEC rules and regulations.
A shareholder who wishes to communicate with the Company’s board of directors may do so by directing a written request addressed to any of our Directors at the address appearing on the first page of this registration statement.
Involvement in Certain Legal Proceedings
Except as noted herein or below, during the last ten (10) years none of our directors or officers have:
(1) had any bankruptcy petition filed by or against any business of which such person was a general partner or executive officer either at the time of the bankruptcy or within two years prior to that time;
(2) been convicted in a criminal proceeding or subject to a pending criminal proceeding;
(3) been subject to any order, judgment, or decree, not subsequently reversed, suspended or vacated, of any court of competent jurisdiction, permanently or temporarily enjoining, barring, suspending or otherwise limiting his involvement in any type of business, securities or banking activities; or
| 59 |
(4) been found by a court of competent jurisdiction in a civil action, the Commission or the Commodity Futures Trading Commission to have violated a federal or state securities or commodities law, and the judgment has not been reversed, suspended, or vacated.
All of these filing requirements were satisfied by the Company’s officers, directors, and ten-percent holders.
In making these statements, we have relied on the written representation of our Directors and Officers or copies of the reports that they have filed with the Commission.
Code of Ethics
The Company has adopted a Code of Ethics for Senior Financial Officers that is applicable to our principal executive officer, principal financial officer, principal accounting officer or controller, or persons performing similar functions.
Indemnification
Under our Articles of Incorporation and Bylaws of the corporation, we may indemnify an officer or director who is made a party to any proceeding, including a law suit, because of his position, if he acted in good faith and in a manner he reasonably believed to be in our best interest. We may advance expenses incurred in defending a proceeding. To the extent that the officer or director is successful on the merits in a proceeding as to which he is to be indemnified, we must indemnify him against all expenses incurred, including attorney's fees. With respect to a derivative action, indemnity may be made only for expenses actually and reasonably incurred in defending the proceeding, and if the officer or director is judged liable, only by a court order. The indemnification is intended to be to the fullest extent permitted by the laws of the State of Nevada.
Regarding indemnification for liabilities arising under the Securities Act of 1933, which may be permitted to directors or officers under Nevada law, we have been advised that, in the opinion of the Securities and Exchange Commission, indemnification is against public policy, as expressed in the Act and is, therefore, unenforceable.
Item 11. Executive Compensation
We recorded consulting expenses fees to three of our Directors, CFO and salary to three officers. For the current year ending August 31, 2022, our Directors and officers received a yearly salary of $1,370,250.
Summary Compensation Table
| Executive Officer Compensation Schedules – See Section D of Main Questionnaire | ||||||||||||||||||||||||||||||||
| Summary Compensation Table in For Fiscal 2022($) | ||||||||||||||||||||||||||||||||
| Name | Salary | Consulting fees | Bonus | Stock awards | Share base payment | Cars Expenses | Nonqualified deferred compensation earnings ($) | Total ($) | ||||||||||||||||||||||||
| Eyal Barad | 246,167 | – | – | – | – | 22,982 | – | 269,149 | ||||||||||||||||||||||||
| Gabriel Yariv | 230,856 | – | – | – | 489,650 | 31,076 | – | 751,583 | ||||||||||||||||||||||||
| Dr. Inbar | – | 12,000 | – | – | 69,950 | – | – | 81,950 | ||||||||||||||||||||||||
| Dr. Gil Feiler | – | 72,000 | – | – | 56,104 | – | – | 128,104 | ||||||||||||||||||||||||
| Shaul Yemal | – | 9,000 | – | – | 55,146 | – | – | 64,146 | ||||||||||||||||||||||||
| Sanja Goldberg | 4,812 | – | – | – | – | – | – | |||||||||||||||||||||||||
| Uri Ben-Or | – | 75,318 | – | – | – | – | – | 75,318 | ||||||||||||||||||||||||
| 481,836 | 168,318 | – | – | 670,850 | 54,058 | – | 1,370,250 | |||||||||||||||||||||||||
| 60 |
The following table sets forth, as of August 31, 2022, information concerning ownership of our securities by (i) each director, (ii) each executive officer, (iii) all directors and executive officers as a group; and (iv) each person known to us to be the beneficial owner of more than five percent of each class:
The number and percentage of shares beneficially owned includes any shares as to which the named person has sole or shared voting power or investment power and any shares that the named person has the right to acquire within 60 days.
| Beneficial Ownership | ||||||||
| Name of Beneficial Owner | Common Shares | Percentage of class | ||||||
| Cannabics Inc. * | 724,499 | 57.6% | ||||||
____________________
*Our 2 Directors Eyal Barad and Gabriel Yariv are also holders of Cannabics, Inc. The mailing address for all directors, executive officers and beneficial owners of Cannabics Inc. is #3 Bethesda Metro Center, Suite 700, Bethesda, Maryland, 20814.
*The Directors of the Company hold positions in Cannabics, Inc., the majority holder. The relative positions of Cannabics, Inc. are listed below :
| Shareholder | Common Stock | % Issued | ||||||
| Eyal Barad, Director, CEO | 316 | 26.25% | ||||||
| Eyal Ballan, past Director, CTO | 141 | 11.71% | ||||||
| Gabriel Yariv, Director COO | 4 | 0.33% | ||||||
| Shay Avraham Sarid | 223 | 18.52% | ||||||
| Itamar Borochov | 222 | 18.44% | ||||||
| Seach Sarid Ltd. | 40 | 3.32% | ||||||
| Ariel Kirtchuk | 25 | 2.08% | ||||||
| J Reiger Ltd | 128 | 10.63% | ||||||
| Cannis Group | 68 | 5.64% | ||||||
| Gil Feiler | 37 | 3.07% | ||||||
| Total | 1,204 | 100.00% | ||||||
Unless otherwise noted, we believe that all persons or entities named in the table have sole voting and investment power with respect to all shares of common stock beneficially owned by them. For purposes hereof, a person is considered to be the beneficial owner of securities that can be acquired by such person within 60 days from the date hereof.
Equity Compensation Plan Information
On October 6th, 2021, the Compensation Committee adopted, and the Board ratified the Company’s Equity Incentive Plan. The Plan is intended to secure for the Company the benefits arising from ownership of the Company’s common stock by the employees, officers, directors and consultants of the Company, all of whom are, and will be, responsible for the Company’s future growth. The Plan is designed to help attract and retain for the Company, qualified personnel for positions of exceptional responsibility, to reward employees, officers, directors and consultants for their services to the Company and to motivate such individuals through added incentives to further contribute to the success of the Company.
The Plan provides an opportunity for an employee, officer, director or consultant of the Company, subject to certain national securities and taxation laws, to receive (i) incentive stock options, (ii) nonqualified stock options; (iii) restricted stock; (iv) stock awards; (v) shares in performance of services; or (vi) any combination of the foregoing. Incentive stock options granted under the Plan are intended to qualify as “incentive stock options” within the meaning of Section 422 of the Internal Revenue Code of 1986, as amended (the “Code”). Nonqualified (non-statutory stock options) granted under the Plan are not intended to qualify as incentive stock options under the Code. The Plan is administered by the Altshuler Shaham Benefits Firm, a licensed fiduciary.
| 61 |
Item 12. Security Ownership of Certain Beneficial Owners and Management Related Stockholder Matters.
None.
Item 13. Certain Relationships and Related Transactions, and Director Independence
None.
Item 14. Principal Accounting Fees and Services
Audit Fees
The aggregate fees billed by Weinstein International CPA for professional services rendered for the audit of our annual financial statements included in this Annual Report on Form 10-K for the fiscal year ended August 31, 2022, were $10,000.
All Other Fees
For the fiscal years ended August 31, 2022, and 2021, the Company did not pay any other fees.
Effective May 6, 2003, the Securities and Exchange Commission adopted rules that require that before Weinstein International CPA is engaged by us or our subsidiaries to render any auditing or permitted non-audit related service, the engagement be:
| · | approved by our audit committee; or | |
| · | entered into pursuant to pre-approval policies and procedures established by the audit committee, provided the policies and procedures are detailed as to the particular service, the audit committee is informed of each service, and such policies and procedures do not include delegation of the audit committee's responsibilities to management. |
We do not have an audit committee. Our Board of Directors pre-approves all services provided by our independent auditors. All of the above services and fees were reviewed and approved by our Board of Directors before the respective services were rendered.
| 62 |
PART IV
Item 15. Exhibits
| * | Filed herewith. |
| 63 |
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934 the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| Date: November 30, 2022 | By: | /s/ Eyal Barad |
|
Eyal Barad, Director Chief Executive Officer | ||
| /s/ Uri Ben-Or | ||
|
Uri Ben-Or, Chief Financial Officer Principal Accounting Officer | ||
| 64 |