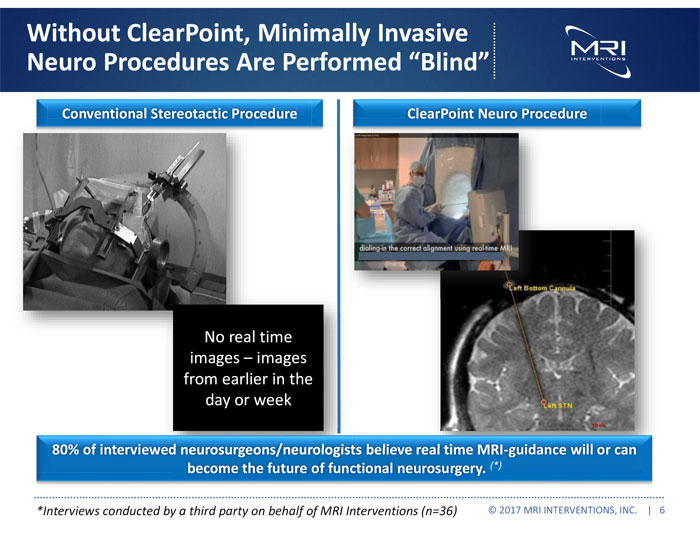
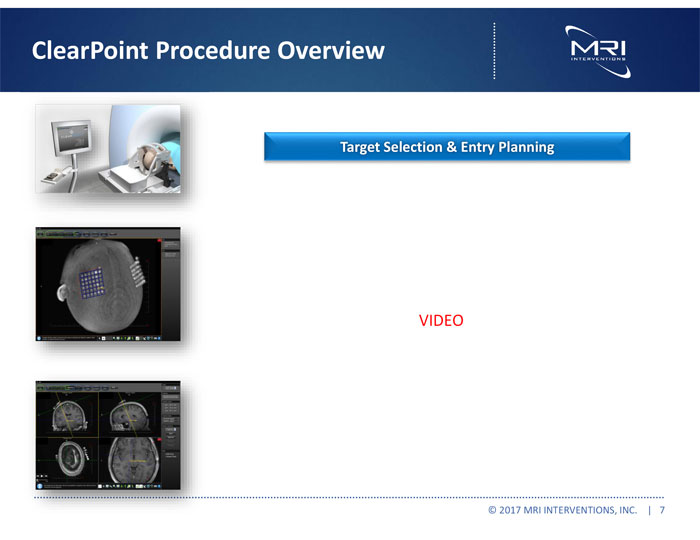
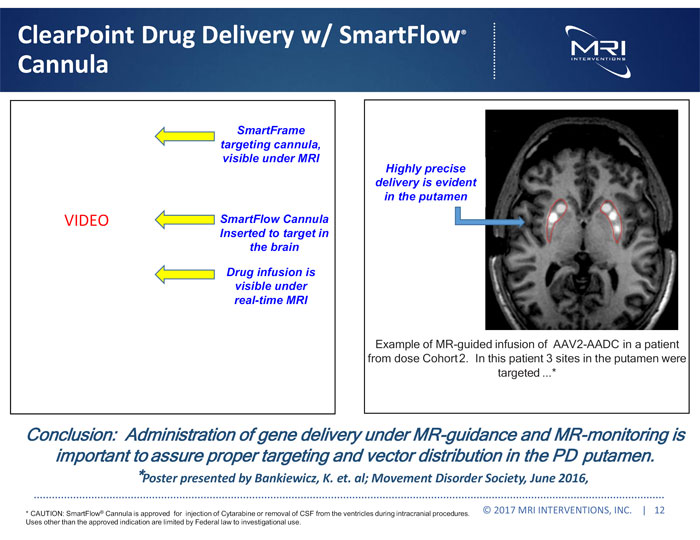
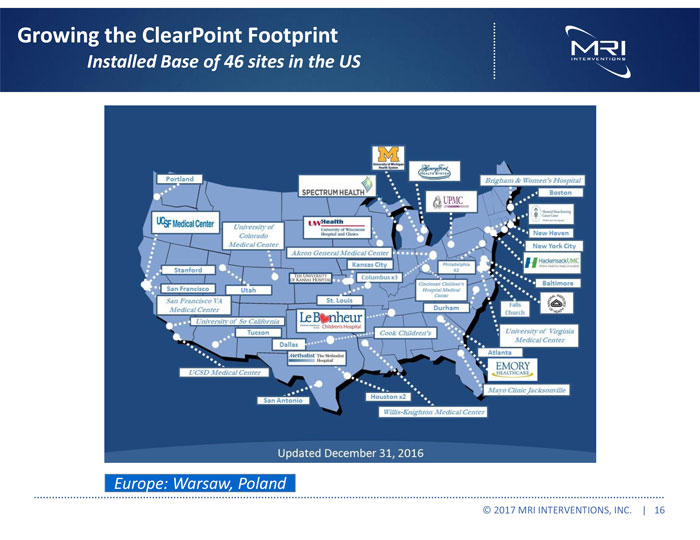
Exhibit 99.1

Contact: Harold A. Hurwitz, Chief Financial Officer
(949) 900-6833
For Immediate Release
MRI INTERVENTIONS, INC. ANNOUNCES 2016 FOURTH QUARTER
AND FULL YEAR RESULTS
Key
2016 accomplishments include year-over-year increases in revenue of 25%, ClearPoint®
procedures of 32% and disposable product revenue of 36%
IRVINE, CA, February 28, 2017 – MRI Interventions, Inc. (OTCQB: MRIC) today announced financial results for the fourth quarter and full year ended December 31, 2016.
2016 Highlights
“We were very pleased with fourth quarter results, as well as our accomplishments in 2016. During the last year, we established a solid foundation for the Company and a platform for continued growth. As we drive the adoption of our technology, we continue to focus on adding new sites, growing procedures, and tightly managing expenses,” said Frank Grillo, President and CEO of MRI Interventions. “Real time intra-operative MRI guidance is a significant advance in neurosurgery, and we are proud to be the leader in this field. The milestones we achieved include:
2016 Key Accomplishments:
| ● | Increased 2016 revenue by 25% over 2015, with a 36% increase in disposable products; |
| ● | Grew 2016 ClearPoint procedure volume 32% over 2015, and completed more than 500 procedures for the first time in a calendar year; |
| ● | Solidified our installed base of major academic medical centers and leading hospitals, ending 2016 with 47 accounts; |
| ● | Increased 2016 gross margin to 59%, with further leverage available as we grow the volume of procedures; |
| ● | Improved our balance sheet through reduction of debt and accumulated interest of almost $4.5 million; and |
| ● | Advanced our drug delivery strategy, including sale of our SmartFlow® cannulas to six companies for use in pharmaceutical and biotech trials. |
Fourth Quarter 2016 Key Accomplishments
| ● | Increased fourth quarter 2016 disposable product revenue by 35% over the same period in 2015; |
| ● | Set new Company records for quarterly revenue, disposable product revenue and ClearPoint procedures, which reached 130 in the fourth quarter of 2016; and |
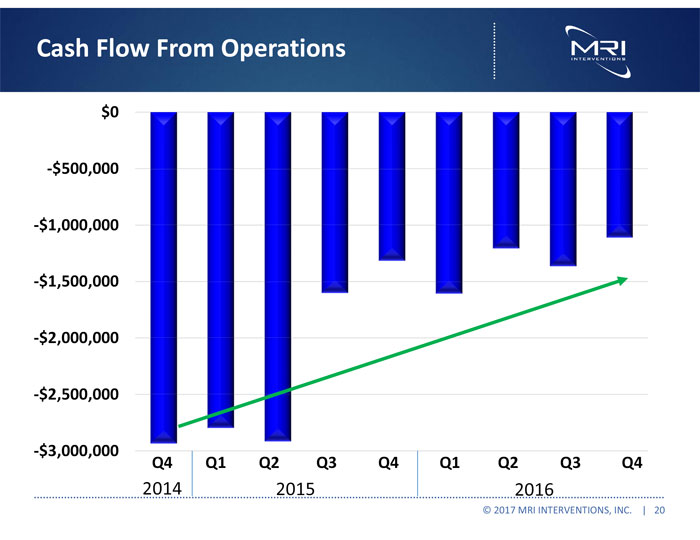
| ● | Tightly managed cash, resulting in a net cash burn of $1.1 million in the fourth quarter of 2016, our lowest quarterly burn rate since becoming a commercially focused company. |
5 Musick, Irvine, California 92618 949.900.6833 |
“2017 will be an exciting year for the Company and our technology,” Grillo continued. “Revenues are up, expenses are down, and adoption of the ClearPoint Neuro Navigation System continues to expand.”
Financial Results – Year Ended December 31, 2016
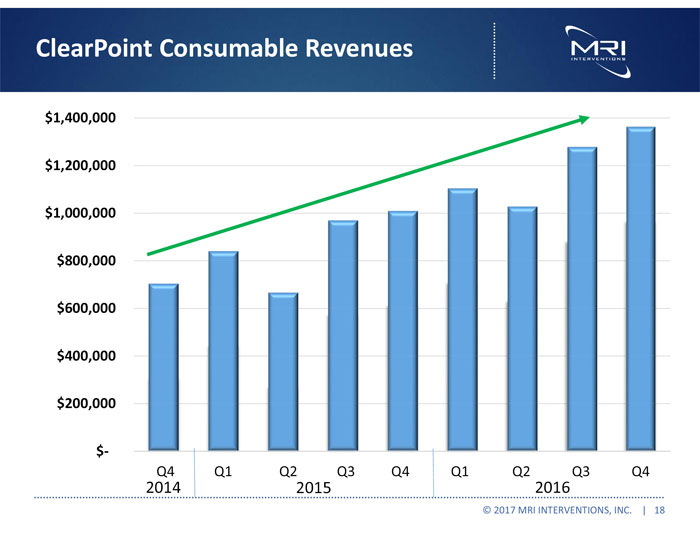
ClearPoint disposable product sales for the year ended December 31, 2016 were $4.8 million, compared with $3.5 million for the year ended December 31, 2015, representing an increase of $1.3 million, or 36%. This increase was due primarily to a greater number of procedures in 2016, which, as previously reported, exceeded 500 procedures for the first time in the Company’s history, using the ClearPoint Neuro Navigation System.
ClearPoint reusable product sales in 2016 were $831,000, compared with $907,000 in 2015. Reusable products consist primarily of computer hardware and software bearing sales prices that are appreciably higher than those for disposable products and sales volume has historically fluctuated from period to period.
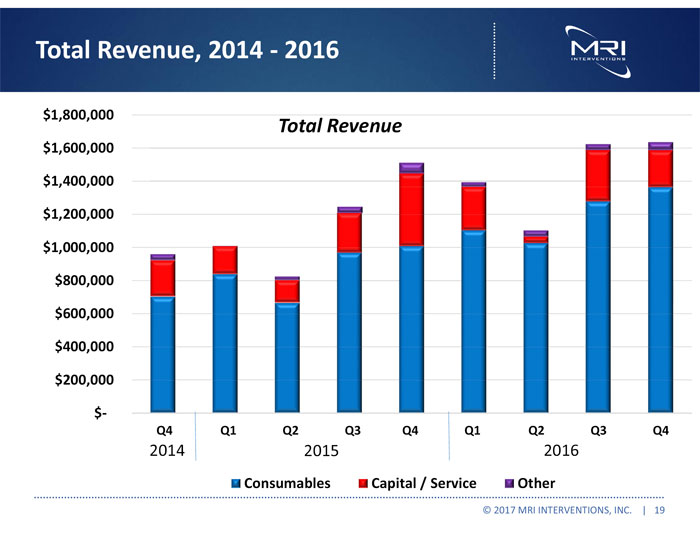
Total revenues were $5.7 million in 2016, and $4.6 million in 2015, an increase of $1.2 million, or 25%.
Gross margin on product revenues in 2016 was 53%, compared to gross margin of 55% in 2015. The decrease in gross margin was due primarily to: (a) product mix differences between 2015 and 2016 in the equipment configuration of hardware and software in ClearPoint systems sold during those respective periods; (b) an increase from 2015 to 2016 in charges to the provision for obsolete and expired product; and (c) an increase in 2016, relative to 2015, in the allocation of indirect labor to production activities, commensurate with the Company’s transition from research and development to commercial activities. These factors were partially offset by increases in 2016, relative to 2015, in average unit selling prices, and decreases in 2016, relative to 2015, in average unit costs due to more favorable pricing from vendors resulting from higher order quantities.
Research and development costs were $2.6 million in 2016, compared to $2.0 million in 2015, an increase of $671,000, or 34%. The increase was due primarily to increases in: (a) costs related to the Company’s development of the next generation of the ClearPoint operating system; (b) intellectual property costs; (c) professional fees and consultants; and (d) personnel costs. Partially offsetting these factors was an increase in departmental costs allocated to production activities.
Selling, general and administrative expenses were $8.0 million in 2016, compared with $8.4 million in 2015, a decrease of $403,000, or 5%. The decrease was primarily attributable to: (a) a decrease in personnel costs; (b) an increase in the allocation of departmental resources to production activities; (c) a decrease in medical device excise taxes, suspended by federal legislation for a two-year period beginning January 1, 2016; and (d) a decrease in non-personnel related marketing expenses. Partially offsetting these factors were increases in public company costs and professional fees. With respect to professional fees, in August 2016, the Company elected to suspend efforts then underway to sell equity units through a public offering and instead commenced a private placement of equity units that was completed in September 2016. Upon suspension of those public offering efforts, the Company capitalized certain related legal and other costs, amounting to $459,000, in anticipation of resuming public offering efforts within an estimated six-month time frame. In December 2016, the Company determined that a future public offering it might consider was not likely to be commenced within this six-month time frame, and accordingly, in the fourth quarter of 2016, the Company recorded a charge of $459,000 to general and administrative expense.
In March 2015, the Company announced the consolidation of all major business functions into its Irvine, California headquarters. In connection with this consolidation, the Company closed its Memphis, Tennessee office in May 2015, and did not retain any of its Memphis-based employees. A total of seven employees were impacted by the consolidation, including three executives. As a result, the Company incurred expense of $1.3 million primarily related to termination costs, including the modifications of option terms, in 2015.
In 2016 and 2015, the Company recorded gains of $1.1 million and $1.5 million, respectively, resulting from changes in the fair value of derivative liabilities. In 2016, such derivative liabilities related to: (a) the issuance of warrants in connection with 2012 and 2013 private placement transactions; and (b) amendments, in June and August 2016, of certain notes to add contingent conversion terms and potential down round pricing protection of warrants issued in connection with such notes. In 2015, derivative liabilities were limited to the issuance of warrants in connection with the 2012 and 2013 private placement transactions.
In April 2016, the Company entered into a securities purchase agreement with Brainlab AG (“Brainlab”) under which a note payable to Brainlab in the principal amount of $4.3 million (the “Brainlab Note”) was restructured and, among other items, the Company: (i) entered into a patent and technology license agreement with Brainlab for software relating to the Company’s SmartFrame® device, in consideration for the cancellation of $1.0 million of the principal amount of the Brainlab Note; and (ii) issued to Brainlab, in consideration for the cancellation of approximately $1.3 million of the principal amount of the Brainlab Note, equity units, consisting of shares of the Company’s common stock and warrants to purchase shares of common stock. As a result of the foregoing, the Company recorded a debt restructuring gain of $941,000 representing the difference between (a) the aggregate fair value of the license agreement, which had no cost basis on the Company’s consolidated balance sheets, and the equity units, and (b) the aggregate principal amount of the Brainlab Note cancelled as consideration.
On June 30, 2016, the Company entered into amendments (the “Amendments”) with Brainlab, with respect to the Brainlab Note, and with two holders of secured notes payable we executed in 2014 (the “2014 Secured Notes”). Pursuant to the Amendments, the parties agreed that, in the event the Company closes a qualified public offering: (i) $2,000,000 of the principal balance of those notes, plus all unpaid accrued interest on that amount, will automatically convert into the security offered in the qualified public offering; and (ii) the exercise price for 46,207 shares of common stock underlying warrants issued in connection with those notes will be reduced as provided in the Amendments. Based on the provisions of the Amendments, on June 30, 2016, the Company recorded a non-cash loss of $820,000 resulting from the restructuring of the Brainlab Note and those 2014 Secured Notes.
On August 31, 2016, the Company entered into second amendments with the two holders of the 2014 Secured Notes that provided, in the event the Company closes a private equity offering, for: (a) the conversion to equity of an aggregate of $1.75 million of principal based on the private offering price; and (b) a reduction in the exercise price for shares of common stock that may be purchased upon exercise of warrants issued in connection with the issuance of such notes based the private offering’s terms for warrant exercise pricing. Execution of the second amendments constituted a debt extinguishment under generally accepted accounting principles, necessitating the Company to record a non-cash loss on debt restructuring of approximately $933,000, representing the aggregate difference in the fair value of the derivative liabilities between the points in time (i) immediately preceding, and (ii) immediately subsequent to, the execution of the second amendments.
Financial Results – Quarter Ended December 31, 2016
ClearPoint disposable product sales for the three months ended December 31, 2016 were $1.4 million, compared with $1.0 million for the same period in 2015, representing an increase of $354,000, or 35%. This increase was due primarily to a greater number of procedures performed using the ClearPoint Neuro Navigation System in the fourth quarter of 2016, relative to the same period in 2015.
ClearPoint reusable product sales for the three months ended December 31, 2016 were $224,000, and $438,000 for the same period in 2015.
Total revenues were $1.6 million for the three months ended December 31, 2016, and $1.5 million for the same period in 2015, an increase of $124,000, or 8%.
Gross margin on product revenues was 59% for the three months ended December 31, 2016, compared to 57% for the same period in 2015. The improvement was attributable primarily to: (a) a greater portion in the fourth quarter of 2016, compared to the same period in 2015, of revenues represented by ClearPoint disposable products, which generally have higher gross margins relative to ClearPoint reusable products; and (b) a decrease in the fourth quarter of 2016, relative to the same period in 2015, in average unit costs due to more favorable pricing from vendors resulting from higher order quantities; partially offset by (c) an increase in charges to the provision for obsolete and expired product.
Research and development costs of $530,000 for the three months ended December 31, 2016, were substantially unchanged from $523,000 for the same period in 2015. Decreases in the fourth quarter of 2016, relative to the same period in 2015, in personnel costs and third-party testing costs, were offset by increases in intellectual property costs and consulting expenses.
Selling, general and administrative expenses were $2.2 million for the three months ended December 31, 2016, compared to $1.8 million in the same period in 2015, an increase of $457,000, or 21%. This increase was due primarily to the Company’s decision, in December 2016, to write off the previously capitalized public offering costs, amounting to $459,000, discussed above.
During the three months ended December 31, 2016 and 2015, the Company recorded gains of $318,000 and $559,000, respectively, from changes in the fair value of derivative liabilities.
Reverse Stock Split
As previously announced, on July 21, 2016, the Company’s Board of Directors approved a 1-for-40 reverse stock split of its issued common stock, which was effectuated on July 26, 2016. All disclosure of common shares and per share data in the accompanying condensed consolidated financial statements have been adjusted retroactively to reflect the reverse stock split for all periods presented.
Teleconference Information
Investors and analysts are invited to listen to a live broadcast review of the Company's 2016 fourth quarter and full year financial results today at 4:30 p.m. Eastern Time (1:30 p.m. Pacific Time) that may be accessed by visiting the Company's website at www.mriinterventions.com and selecting “Investors” / “News” / “IR Calendar.” The conference call may also be accessed at http://mriinterventions.equisolvewebcast.com/q4-2016. Investors and analysts who would like to participate in the conference call may do so via telephone at (877) 407-9034, or at (201) 493-6737 if calling from outside the U.S. or Canada.
For those who cannot access the live broadcast, a replay will be available shortly after the completion of the call until March 7, 2017 by calling (877) 660-6853, or (201) 612-7415 if calling from outside the U.S. or Canada, and then entering conference I.D. number 413671. An online archive of the broadcast will be available on the Company's website at www.mriinterventions.com, on the “Investor Relations” page.
About MRI Interventions, Inc.
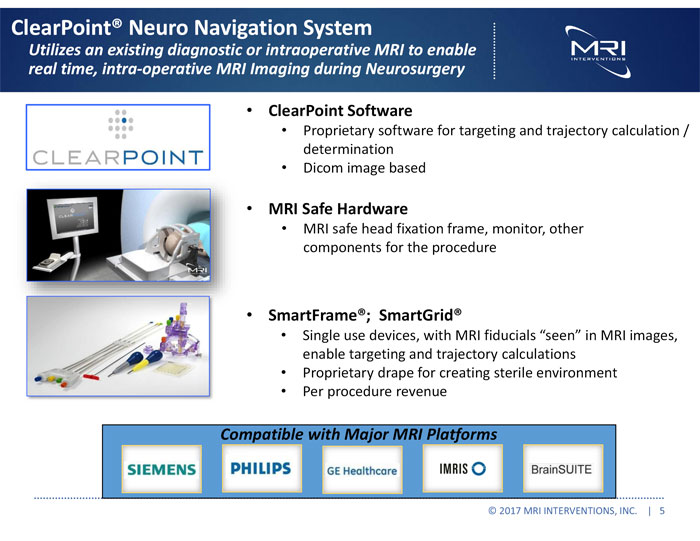
Building on the imaging power of magnetic resonance imaging (“MRI”), MRI Interventions is creating innovative platforms for performing the next generation of minimally invasive surgical procedures in the brain. The ClearPoint Neuro Navigation System, which has received 510(k) clearance and is CE marked, utilizes a hospital’s existing diagnostic or intraoperative MRI suite to enable a range of minimally invasive procedures in the brain. For more information, please visit www.mriinterventions.com.
Forward-Looking Statements
Statements herein concerning MRI Interventions, Inc. (the “Company”) plans, growth and strategies may include forward-looking statements within the context of the federal securities laws. Statements regarding the Company's future events, developments and future performance, as well as management's expectations, beliefs, plans, estimates or projections relating to the future, are forward-looking statements within the meaning of these laws. Uncertainties and risks may cause the Company's actual results to differ materially from those expressed in or implied by forward-looking statements. Particular uncertainties and risks include those relating to: the Company’s ability to obtain additional financing; estimates regarding the sufficiency of the Company’s cash resources; future revenues from sales of the Company’s ClearPoint Neuro Navigation System products; and the Company’s ability to market, commercialize and achieve broader market acceptance for the Company’s ClearPoint Neuro Navigation System products. More detailed information on these and additional factors that could affect the Company’s actual results are described in the “Risk Factors” section of the Company’s Annual Report on Form 10-K for the year ended December 31, 2015, and its Quarterly Report on Form 10-Q for the quarter ended September 30, 2016, both of which have been filed with the Securities and Exchange Commission, as well as the Company’s Annual Report on Form 10-K for the year ended December 31, 2016, which will be filed with the Securities and Exchange Commission on or before March 31, 2017.
(tables follow)
MRI INTERVENTIONS, INC.
Consolidated Statements of Operations
| For The Three Months Ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| Revenues: | ||||||||
| Product revenues | $ | 1,599,326 | $ | 1,452,963 | ||||
| Other service revenues | 35,779 | 47,088 | ||||||
| Development service revenues | - | 11,563 | ||||||
| Total revenues | 1,635,105 | 1,511,614 | ||||||
| Cost of product revenues | 676,924 | 646,812 | ||||||
| Research and development costs | 529,714 | 522,609 | ||||||
| Selling, general, and administrative expenses | 2,218,726 | 1,761,920 | ||||||
| Operating loss | (1,790,259 | ) | (1,419,727 | ) | ||||
| Other income (expense): | ||||||||
| Gain on change in fair value of derivative liabilities | 317,855 | 558,654 | ||||||
| Other income (expense), net | 6,571 | (12,630 | ) | |||||
| Interest income | 1,032 | 1,568 | ||||||
| Interest expense | (216,082 | ) | (326,300 | ) | ||||
| Net loss | $ | (1,680,883 | ) | $ | (1,198,435 | ) | ||
| Net loss per share attributable to common stockholders: | ||||||||
| Basic and diluted | $ | (0.47 | ) | $ | (0.62 | ) | ||
| Weighted average shares outstanding: | ||||||||
| Basic and diluted | 3,610,655 | 1,923,054 | ||||||
MRI INTERVENTIONS, INC.
Consolidated Balance Sheets
| December 31, | ||||||||
| 2016 | 2015 | |||||||
| ASSETS | ||||||||
| Current Assets: | ||||||||
| Cash and cash equivalents | $ | 3,315,774 | $ | 5,408,523 | ||||
| Accounts receivable | 865,943 | 1,218,043 | ||||||
| Inventory, net | 1,768,382 | 1,807,895 | ||||||
| Prepaid expenses and other current assets | 134,996 | 97,249 | ||||||
| Total current assets | 6,085,095 | 8,531,710 | ||||||
| Property and equipment, net | 328,249 | 440,606 | ||||||
| Software license inventory | 976,900 | 937,100 | ||||||
| Other assets | 10,641 | 27,306 | ||||||
| Total assets | $ | 7,400,885 | $ | 9,936,722 | ||||
| LIABILITIES AND STOCKHOLDERS’ DEFICIT | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 1,546,926 | $ | 697,807 | ||||
| Accrued compensation | 666,060 | 557,784 | ||||||
| Other accrued liabilities | 450,424 | 1,398,707 | ||||||
| Derivative liabilities | 131,173 | 658,286 | ||||||
| Deferred product and service revenues | 223,117 | 116,009 | ||||||
| Senior secured note payable, net of unamortized discount of $64,835 at December 31, 2015 | - | 4,224,609 | ||||||
| Total current liabilities | 3,017,700 | 7,653,202 | ||||||
| Accrued interest | 647,500 | 542,500 | ||||||
| Senior note payable | 2,000,000 | - | ||||||
| 2010 junior secured notes payable, net of unamortized discount of $2,302,472 and $2,535,230 at December 31, 2016 and 2015, respectively | 697,528 | 464,770 | ||||||
| 2014 junior secured 12% notes payable, net of unamortized discount and deferred issuance costs aggregating $180,774 and $467,611 at December 31, 2016 and 2015, respectively | 1,794,226 | 3,257,389 | ||||||
| Total liabilities | 8,156,954 | 11,917,861 | ||||||
| Commitments and contingencies | ||||||||
| Stockholders' deficit: | ||||||||
| Preferred stock, $0.01 par value; 25,000,000 shares authorized at December 31, 2016 and 2015; none issued and outstanding at December 31, 2016 and 2015 | - | - | ||||||
| Common stock, $0.01 par value; 200,000,000 shares authorized at December 31, 2016 and 2015; 3,622,032 and 2,284,537 shares issued and outstanding at December 31, 2016 and 2015, respectively | 36,220 | 22,845 | ||||||
| Additional paid-in capital | 93,076,475 | 83,722,596 | ||||||
| Accumulated deficit | (93,868,764 | ) | (85,726,580 | ) | ||||
| Total stockholders’ deficit | (756,069 | ) | (1,981,139 | ) | ||||
| Total liabilities and stockholders’ deficit | $ | 7,400,885 | $ | 9,936,722 | ||||
MRI INTERVENTIONS, INC.
Consolidated Statements of Operations
| Years Ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| Revenues: | ||||||||
| Product revenues | $ | 5,612,857 | $ | 4,416,036 | ||||
| Development service revenues | - | 37,405 | ||||||
| Other service revenues | 136,597 | 140,751 | ||||||
| Total revenues | 5,749,454 | 4,594,192 | ||||||
| Cost of product revenues | 2,642,763 | 1,987,636 | ||||||
| Research and development costs | 2,628,179 | 1,957,332 | ||||||
| Selling, general, and administrative expenses | 7,967,250 | 8,370,749 | ||||||
| Restructuring charges | - | 1,252,584 | ||||||
| Operating loss | (7,488,738 | ) | (8,974,109 | ) | ||||
| Other income (expense): | ||||||||
| Gain on change in fair value of derivative liabilities | 1,065,935 | 1,539,876 | ||||||
| Loss on debt restructuring | (811,909 | ) | - | |||||
| Other income, net | 216,075 | 230,875 | ||||||
| Interest income | 8,807 | 16,455 | ||||||
| Interest expense | (1,060,065 | ) | (1,262,343 | ) | ||||
| Net loss | $ | (8,069,895 | ) | $ | (8,449,246 | ) | ||
| Net loss per share attributable to common stockholders: | ||||||||
| Basic and diluted | $ | (2.93 | ) | $ | (4.48 | ) | ||
| Weighted average shares outstanding: | ||||||||
| Basic and diluted | 2,754,803 | 1,884,849 | ||||||
MRI INTERVENTIONS, INC.
Consolidated Statements of Cash Flows
| Years Ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| Cash flows from operating activities: | ||||||||
| Net loss | $ | (8,069,895 | ) | $ | (8,449,246 | ) | ||
| Adjustments to reconcile net loss to net cash flows from operating activities: | ||||||||
| Depreciation and amortization | 155,707 | 211,946 | ||||||
| Share-based compensation | 959,585 | 1,682,063 | ||||||
| Expenses paid through the issuance of common stock | 290,245 | 145,987 | ||||||
| Gain on change in fair value of derivative liabilities | (1,065,935 | ) | (1,539,876 | ) | ||||
| Loss on debt restructuring | 811,909 | - | ||||||
| Loss on retirement of equipment | 1,689 | 2,053 | ||||||
| Amortization of debt issuance costs and original issue discounts | 424,431 | 471,146 | ||||||
| Increase (decrease) in cash resulting from changes in: | ||||||||
| Accounts receivable | 352,100 | (749,094 | ) | |||||
| Inventory | 72,342 | 68,626 | ||||||
| Prepaid expenses and other current assets | (37,748 | ) | (68,029 | ) | ||||
| Other assets | - | 9,811 | ||||||
| Accounts payable and accrued expenses | 178,419 | (436,420 | ) | |||||
| Deferred revenue | 107,108 | 13,299 | ||||||
| Net cash flows from operating activities | (5,820,043 | ) | (8,637,734 | ) | ||||
| Cash flows from investing activities: | ||||||||
| Purchases of property and equipment | (101,002 | ) | (76,883 | ) | ||||
| Net cash flows from investing activities | (101,002 | ) | (76,883 | ) | ||||
| Cash flows from financing activities: | ||||||||
| Net proceeds from equity private placements | 3,833,052 | 4,879,134 | ||||||
| Cash paid in lieu of issuing fractional shares in reverse split of common stock | (4,756 | ) | - | |||||
| Net cash flows from financing activities | 3,828,296 | 4,879,134 | ||||||
| Net change in cash and cash equivalents | (2,092,749 | ) | (3,835,483 | ) | ||||
| Cash and cash equivalents, beginning of year | 5,408,523 | 9,244,006 | ||||||
| Cash and cash equivalents, end of year | $ | 3,315,774 | $ | 5,408,523 | ||||
| SUPPLEMENTAL CASH FLOW INFORMATION | ||||||||
| Cash paid for: | ||||||||
| Income taxes | $ | - | $ | - | ||||
| Interest | $ | 976,295 | $ | 223,500 | ||||