Exhibit 99.1

| CONTACT | ||
| DR. REDDY'S LABORATORIES LTD. | Investor relationS | Media relationS |
| 8-2-337, Road No. 3, Banjara Hills, | AMIT AGARWAL | APARNA TEKURI |
| Hyderabad - 500034. Telangana, India. | amita@drreddys.com | aparnatekuri@drreddys.com |
| (Ph: +91-40-4900 2135) | (Ph: +91-40-4900 2446) | |
Dr. Reddy’s Q1 FY21 Financial Results
Hyderabad, India, July 29, 2020: Dr. Reddy’s Laboratories Ltd. (BSE: 500124 | NSE: DRREDDY | NYSE: RDY) today announced its consolidated financial results for the quarter ended June 30, 2020. The information mentioned in this release is on the basis of consolidated financial statements under International Financial Reporting Standards (IFRS).
|
Revenues
|
Rs. 4,418 Cr [YoY: Up 15%; QoQ: Down 0.3%] |
|
Gross Margin
|
56.0% [Q1 FY20: 51.7%; Q4 FY20: 51.5%] |
|
SG&A Expenses
|
Rs. 1,279 Cr [YoY: Up 6%; QoQ: Up 5%] |
|
R&D Expenses
|
Rs. 398 Cr [9.0% of Revenues] |
|
EBITDA*
|
Rs. 1,162 Cr [YoY: Up 2%; QoQ: Up 16%] |
|
Profit before Tax
|
Rs. 879 Cr [19.9% of Revenues] |
|
Profit after Tax
|
Rs. 579 Cr [13.1% of Revenues] |
* Q1 FY20 includes a settlement income of Rs. 346 Cr, adjusted for which the EBITDA YoY growth is 47%
Commenting on the results, Co-chairman & MD, G V Prasad said “the current quarter’s financial performance has been strong across all parameters. I am glad that we have been able to serve our patients well and ensured continuity of business operations despite the challenging times. We have started integration of the acquired business from Wockhardt and executed two important licencing arrangements for treatment options for COVID-19. Currently, we are working towards bringing both these drugs to multiple markets”.
| 1 |
| All amounts in millions, except EPS | All US dollar amounts based on convenience translation rate of I USD = Rs. 75.53 |
Dr. Reddy’s Laboratories Limited and Subsidiaries
Consolidated Income Statement
| Q1 FY21 | Q1 FY20 | YoY | Q4 FY20 | QoQ | ||||||||||||||||||||||||||||
| Particulars | ($) | (Rs.) | ($) | (Rs.) | Gr % | ($) | (Rs.) | Gr% | ||||||||||||||||||||||||
| Revenues | 585 | 44,175 | 509 | 38,435 | 15 | 587 | 44,318 | (0 | ) | |||||||||||||||||||||||
| Cost of Revenues | 257 | 19,420 | 246 | 18,576 | 5 | 285 | 21,510 | (10 | ) | |||||||||||||||||||||||
| Gross Profit | 328 | 24,755 | 263 | 19,859 | 25 | 302 | 22,808 | 9 | ||||||||||||||||||||||||
| Operating Expenses | ||||||||||||||||||||||||||||||||
| Selling, General & Administrative expenses | 169 | 12,786 | 160 | 12,065 | 6 | 161 | 12,177 | 5 | ||||||||||||||||||||||||
| Research and Development expenses | 53 | 3,980 | 48 | 3,609 | 10 | 55 | 4,190 | (5 | ) | |||||||||||||||||||||||
| Impairment of non-current assets | 0 | 7 | (100 | ) | ||||||||||||||||||||||||||||
| Other operating income | (2 | ) | (118 | ) | (50 | ) | (3,759 | ) | (97 | ) | (2 | ) | (168 | ) | (30 | ) | ||||||||||||||||
| Results from operating activities | 107 | 8,107 | 105 | 7,944 | 2 | 87 | 6,602 | 23 | ||||||||||||||||||||||||
| Net finance income | (8 | ) | (605 | ) | (5 | ) | (393 | ) | 54 | (6 | ) | (435 | ) | 39 | ||||||||||||||||||
| Share of profit of equity accounted investees | (1 | ) | (77 | ) | (2 | ) | (163 | ) | (53 | ) | (1 | ) | (105 | ) | (27 | ) | ||||||||||||||||
| Profit before Income Tax | 116 | 8,789 | 113 | 8,500 | 3 | 95 | 7,142 | 23 | ||||||||||||||||||||||||
| Income tax | 40 | 2,996 | 25 | 1,872 | 60 | (7 | ) | (500 | ) | (699 | ) | |||||||||||||||||||||
| Profit for the period | 77 | 5,793 | 88 | 6,628 | (13 | ) | 101 | 7,642 | (24 | ) | ||||||||||||||||||||||
| Diluted Earnings Per Share (EPS) | 0.46 | 34.86 | 0.53 | 39.91 | (13 | ) | 0.61 | 46.01 | (24 | ) | ||||||||||||||||||||||
| As a % to Revenues | Q1 FY21 | Q1 FY20 | Q4 FY20 | |||||||||
| Gross Profit | 56.0 | 51.7 | 51.5 | |||||||||
| SG&A | 28.9 | 31.4 | 27.5 | |||||||||
| R&D | 9.0 | 9.4 | 9.5 | |||||||||
| EBITDA | 26.3 | 29.5 | 22.6 | |||||||||
| PBT | 19.9 | 22.1 | 16.1 | |||||||||
| PAT | 13.1 | 17.2 | 17.2 | |||||||||
EBITDA Computation
| Q1 FY21 | Q1 FY20 | Q4 FY20 | ||||||||||||||||||||||
| Particulars | ($) | (Rs.) | ($) | (Rs.) | ($) | (Rs.) | ||||||||||||||||||
| Profit before Income Tax | 116 | 8,789 | 113 | 8,500 | 95 | 7,142 | ||||||||||||||||||
| Interest income (net)* | (4 | ) | (306 | ) | (3 | ) | (239 | ) | (1 | ) | (100 | ) | ||||||||||||
| Depreciation | 28 | 2,120 | 28 | 2,124 | 28 | 2,080 | ||||||||||||||||||
| Amortization | 14 | 1,020 | 13 | 958 | 12 | 885 | ||||||||||||||||||
| Impairment | 0 | 0 | 0 | 0 | 0 | 7 | ||||||||||||||||||
| EBITDA | 154 | 11,622 | 150 | 11,343 | 133 | 10,013 | ||||||||||||||||||
* Includes income from Investments
| 2 |
| All amounts in millions, except EPS | All US dollar amounts based on convenience translation rate of I USD = Rs. 75.53 |
Key Balance Sheet Items
As on 30th Jun 2020 | As on 31st Mar 2020 | As on 30th Jun 2019 | ||||||||||||||||||||||
| Particulars | ($) | (Rs.) | ($) | (Rs.) | ($) | (Rs.) | ||||||||||||||||||
| Cash and cash equivalents and other investments | 374 | 28,227 | 345 | 26,068 | 377 | 28,439 | ||||||||||||||||||
| Trade receivables (current & non-current) | 640 | 48,316 | 689 | 52,015 | 503 | 37,961 | ||||||||||||||||||
| Inventories | 518 | 39,148 | 464 | 35,066 | 465 | 35,137 | ||||||||||||||||||
| Property, plant and equipment* | 717 | 54,183 | 693 | 52,332 | 716 | 54,083 | ||||||||||||||||||
| Goodwill and Other Intangible assets* | 609 | 45,991 | 419 | 31,653 | 633 | 47,821 | ||||||||||||||||||
| Loans and borrowings (current & non-current) | 418 | 31,582 | 293 | 22,102 | 455 | 34,389 | ||||||||||||||||||
| Trade payables | 252 | 19,038 | 221 | 16,659 | 197 | 14,842 | ||||||||||||||||||
| Equity | 2,142 | 1,61,748 | 2,052 | 1,54,988 | 1,936 | 1,46,209 | ||||||||||||||||||
*On the basis of provisional allocation of purchase price for the business acquired from Wockhardt
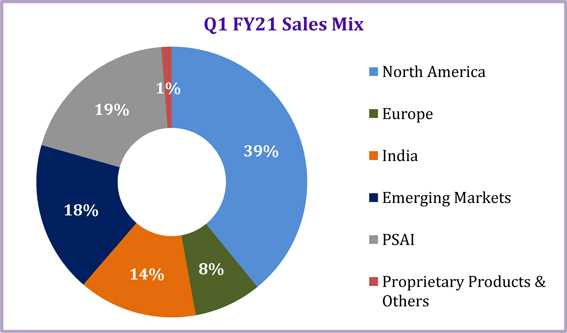
Revenue Mix by Segment
| Q1 FY21 | Q1 FY20 | YoY | Q4 FY20 | QoQ | ||||||||||||||||
| Particulars | (Rs.) | (Rs.) | Growth % | (Rs.) | Growth % | |||||||||||||||
| Global Generics | 35,075 | 32,982 | 6 | % | 36,398 | (4 | %) | |||||||||||||
| North America | 17,282 | 16,323 | 6 | % | 18,072 | (4 | %) | |||||||||||||
| Europe | 3,551 | 2,404 | 48 | % | 3,446 | 3 | % | |||||||||||||
| India | 6,260 | 6,960 | (10 | %) | 6,839 | (8 | %) | |||||||||||||
| Emerging Markets | 7,982 | 7,296 | 9 | % | 8,042 | (1 | %) | |||||||||||||
| Pharmaceutical Services and Active Ingredients (PSAI) | 8,553 | 4,539 | 88 | % | 7,195 | 19 | % | |||||||||||||
| Proprietary Products & Others | 547 | 914 | (40 | %) | 725 | (25 | %) | |||||||||||||
| Total | 44,175 | 38,435 | 15 | % | 44,318 | 0 | % | |||||||||||||
| 3 |
COVID-19 Update
In the current challenging times due to the COVID-19 pandemic, we are undertaking reasonable precautions to ensure the health and safety of our employees, including adhering to the social distancing norms, sanitization of our premises, usage of masks, gloves and other protective wears.
Our operations have continued without much impact. We continued our engagement with doctors through digital channels, ensured regular supplies of our products to meet with the market demand and continued our R&D activities including few projects pertaining to COVID-19.
While the sales volume were impacted in some of our markets due to lower prescriptions generated and fall in patient footfalls in pharmacies / clinics due to Covid-19, the pricing environment was relatively stable, new products launches continued and depreciation of rupee against the US dollar and Euro supported the business.
Revenue Analysis
Global Generics (GG)
Revenues from GG segment at Rs. 35.1 billion:
| Ø | Year-on-year growth of 6% driven primarily by Europe and Emerging Markets. This was offset partially by decline in India. The overall growth was on account of volume traction in the base business, new product launches and aided by favorable forex rates, though offset partially due to price erosion. |
| Ø | Sequential quarter decline of 4%, which is attributable to lower volumes across markets. |
North America
Revenues from North America at Rs. 17.3 billion:
| Ø | Year-on-year growth of 6%, driven by contribution from new products launched and aided by a favorable forex rate, which was partially offset by price erosion. |
| Ø | Sequential decline of 4%, on account of lower sales of certain key molecules. |
| Ø | We launched six new products (Fenofibrate Tablets, Nitroglycerin Patch, Amphetamine Sulfate Tablets, Desmopressin Acetate Ampules, Colchicine Tablets and Abiraterone Acetate Tablets) |
| Ø | We filed five new ANDAs during the quarter. As of 30th June 2020, cumulatively 101 generic filings are pending for approval with the USFDA (99 ANDAs and 2 NDAs under 505(b)(2) route). Of the 99 ANDAs, 54 are Para IVs and we believe 28 have ‘First to File’ status. |
| 4 |
Emerging Markets
Revenues from Emerging Markets at Rs. 8.0 billion. Year-on-year growth of 9%. Sequential decline of 1%:
| Ø | Revenues from Russia at Rs. 3.3 billion. Year-on-year decline of 17% and sequential decline of 16%. Decline primarily on account of lower volumes due to lower prescriptions generated and fall in patient footfalls in pharmacies / clinics due to Covid-19. |
| Ø | Revenues from other CIS countries and Romania market at Rs. 1.4 billion. Year-on-year growth of 15% driven by higher volumes and new product launches. Sequential decline of 22% on account of lower volumes. |
| Ø | Revenues from Rest of World (RoW) territories at Rs. 3.3 billion. Year-on-year growth of 56% & sequential growth of 41%, primarily driven by new products and volume traction in base business. The growth was offset partially due to price erosion in some molecules. |
India
Revenues from India at Rs. 6.3 billion:
| Ø | Year-on-year decline of 10% and sequential decline of 8%. The decline was on account of lower sales volume due to lower prescriptions generated and fall in patient footfalls in pharmacies / clinics due to Covid-19. |
| Ø | We launched four new brands during the period. |
| Ø | We completed the acquisition of select business from Wockhardt including the manufacturing plant located in Baddi, Himachal Pradesh in the quarter. |
Europe
Revenues from Europe at Rs. 3.6 billion:
| Ø | Year-on-year growth of 48%, on account of new product launches and volume traction across markets. |
| Ø | Sequential growth of 3%, aided by contribution by new products launched and favorable forex, offset partially by lower volumes. |
Pharmaceutical Services and Active Ingredients (PSAI)
Revenues from PSAI at Rs. 8.6 billion:
| Ø | Year-on-year growth of 88% and sequential growth of 19% on account of higher volumes of certain products, increase in new product sales and favorable forex. | |
| Ø | During the quarter we filed DMF for one product in the US. |
Proprietary Products (PP)
Revenues from PP at Rs. 56 million:
| Ø | Year-on-year decline of 80% due to absence of the Neurology franchise products (the US and select territory rights of which were sold in the previous year). |
| 5 |
Income Statement Highlights:
| Ø | Gross profit margin at 56.0%: |
| - | Increased by ~430 bps over previous year and by ~450 bps sequentially, primarily on account of a favorable product mix and forex benefit. |
| - | Gross profit margin for GG and PSAI business segments are at 61.4% and 33.4% respectively. |
| Ø | SG&A expenses at Rs. 12.8 billion, increased by 6% on a year-on-year basis and by 5% sequentially. The increase was primarily attributable to higher freight cost due to shortage of carriers for shipping the goods from India to other countries due to COVID-19 related disruptions. |
| Ø | R&D expenses at Rs. 4.0 billion. As % to revenues- Q1 FY21: 9.0% | Q4 FY 20: 9.5% | Q1 FY20: 9.4%. Our focus continues on building complex generics, bio-similars and differentiated products pipeline. We are also undertaking development of a few projects pertaining to COVID-19 related drugs. |
| Ø | Other operating income at Rs. 118 million compared to Rs. 3.8 billion in Q1 FY20. Previous year included Rs. 3.5 billion received from Celgene pursuant to an agreement entered towards settlement of any claim the Company or its affiliates may have had for damages under section 8 of the Canadian Patented Medicines (Notice of Compliance) Regulations in regard to the Company’s ANDS for a generic version of REVLIMID brand capsules, (Lenalidomide) pending before Health Canada. |
| Ø | Net Finance income at Rs. 605 million compared to Rs. 393 million in Q1 FY20. The increase is primarily on account of higher foreign exchange gain as compared to the previous year. |
| Ø | Profit before Tax at Rs. 8.8 billion, increased by 3% year-on-year and by 23% sequentially. Adjusted for the Rs. 3.5 billion received from Celgene last year, the year-on-year growth is at 74%. |
| Ø | Profit after Tax at Rs. 5.8 billion. The effective tax rate is around 34% for the quarter. The higher tax rate was primarily due to discontinuation of weighted deduction on R&D and completion of tax holiday for one of our plants. |
| Ø | Diluted earnings per share is at Rs. 34.86. |
Other Highlights:
| Ø | Capital expenditure is at Rs. 1.5 billion. |
| Ø | Free cash-flow generated during the quarter stood at Rs. 9.3 billion (before acquisition related payout to Wockhardt of Rs. 15 billion). |
| Ø | Net debt of the company is at Rs. 3.4 billion as on June 30, 2020. Consequently, net debt to equity ratio is 0.02. |
| 6 |
Earnings Call Details (05:30 pm IST, 08:00 am EDT, July 29, 2020)
The management of the Company will host an earnings call to discuss the Company’s financial performance and answer any questions from the participants.
| Conference Joining Information |
| Option 1: Express Join with DiamondPass™ |
Pre-register with the below link and join without waiting for the operator.
https://services.choruscall.in/DiamondPassRegistration/register?confirmationNumber=122477&linkSecurityString=5070ddaf
Option 2: Join through below Dial-In Numbers
| |
|
Universal Access Number:
|
+91 22 6280 1219 +91 22 7115 8120 |
|
Local Access Number: Available all over India |
+91 70456 71221 |
| International Toll Free Number: |
USA: 1 866 746 2133 UK: 0 808 101 1573 Singapore: 800 101 2045 Hong Kong: 800 964 448 |
No password/pin number is necessary to dial in to any of the above numbers. The operator will provide instructions on asking questions before and during the call.
Play Back: The play back will be available after the earnings call, till August 6th, 2020. For play back dial in phone No: +91 22 7194 5757 | +91 22 6663 5757, and Playback Code is 58336.
Transcript: Transcript of the Earnings call will be available on the Company’s website: www.drreddys.com
About Dr. Reddy’s: Dr. Reddy’s Laboratories Ltd. (BSE: 500124, NSE: DRREDDY, NYSE: RDY) is an integrated pharmaceutical company, committed to providing affordable and innovative medicines for healthier lives. Through its three businesses - Pharmaceutical Services & Active Ingredients, Global Generics and Proprietary Products – Dr. Reddy’s offers a portfolio of products and services including APIs, custom pharmaceutical services, generics, biosimilars and differentiated formulations. Our major therapeutic areas of focus are gastrointestinal, cardiovascular, diabetology, oncology, pain management and dermatology. Dr. Reddy’s operates in markets across the globe. Our major markets include – USA, India, Russia & CIS countries, and Europe. For more information, log on to: www.drreddys.com.
Disclaimer: This press release may include statements of future expectations and other forward-looking statements that are based on the management’s current views and assumptions and involve known or unknown risks and uncertainties that could cause actual results, performance or events to differ materially from those expressed or implied in such statements. In addition to statements which are forward-looking by reason of context, the words "may", "will", "should", "expects", "plans", "intends", "anticipates", "believes", "estimates", "predicts", "potential", or "continue" and similar expressions identify forward-looking statements. Actual results, performance or events may differ materially from those in such statements due to without limitation, (i) general economic conditions such as performance of financial markets, credit defaults , currency exchange rates , interest rates , persistency levels and frequency / severity of insured loss events (ii) mortality and morbidity levels and trends, (iii) changing levels of competition and general competitive factors, (iv) changes in laws and regulations and in the policies of central banks and/or governments, (v) the impact of acquisitions or reorganization , including related integration issues, (vi) the susceptibility of our industry and the markets addressed by our, and our customers’, products and services to economic downturns as a result of natural disasters, epidemics, pandemics or other widespread illness, including coronavirus (or COVID-19), and (vii) other risks and uncertainties identified in our public filings with the Securities and Exchange Commission, including those listed under the "Risk Factors" and "Forward-Looking Statements" sections of our Annual Report on Form 20-F for the year ended March 31, 2020. The company assumes no obligation to update any information contained herein.
| 7 |







