Exhibit 99.1

“A New Paradigm in Drug Delivery” Annual Meeting Presentation Highlights from 2015

This Presentation contains forward - looking statements with respect to business conducted by Bio - Path Holdings, Inc . By their nature, forward - looking statements and forecasts involve risks and uncertainties because they relate to events and depend on circumstances that will occur in the future . The Company does not undertake to update any forward - looking statements . There are a number of factors that could cause actual results and developments to differ materially and investors should use their own judgment to evaluate risks . 2 Forward Looking Statements

2015 Overview □ Core Organization » Scientific Advisory Board □ Corporate Development » PR and Marketing » New Patent » DNAbilize ™ □ BP1001 (Liposomal Grb2) in the clinic □ ASH 2015 □ Plans and milestones □ Financial snapshot 3

Core Organization Peter Nielsen Co - Founder, President, Chief Executive Officer and Chief Financial Officer • Officer and D irector since founding Company in 2007 Ulrich Mueller, PhD Chief Operating Officer • P reviously Vice President at the Fred Hutchinson Center Research Center • Former Managing Director Office of Technology Commercialization at MD Anderson Ana M. Tari, PhD & MBA Director, Preclinical Operations & Research • Key member of the research team that developed our liposomal delivery technology Tara Sadeghi, PhD Director, Clinical Operations • Assistant Director of the Cord Blood Bank Regulatory and Quality Assurance office at The University of Texas M. D. Anderson • Over 24 years of drug development and clinical operations experience across all phases of clinical development (Phases I through III ) Suzanne Kennedy, PhD Director, Corporate Development • Over 15 years of marketing, business development, and research & development experience in the biotech industry 4

Jorge Cortes, M.D . Chairman • M.D . from la Facultad de Medicina , Universidad Nacional Autónoma de México • Jane and John Justin Distinguished Chair in Leukemia Research, Chief of the AML and CML sections , and Deputy Chair of the Department of Leukemia at The University of Texas MDACC • Has consulted leading pharmaceutical companies such as AstraZeneca on development of prenyltransferase inhibitors, GlaxoSmithKline on the use of topotecan in MDS and CMML, and Rhône - Poulenc Rorer on the use of PEG - Asparaginase in adult ALL. Amy P. Sing, M.D. Member, Bio - Path’s Board of Directors • M.D. from the Stanford University School of Medicine • Currently Senior Director of Medical Affairs at Genomic Health, Inc. • Former Senior Medical Director at Genentech, Inc., had an integral role in the Avastin ™ program. • Former Senior Director of Medical and Regulatory Affairs at Seattle Genetics Recruiting additional members Scientific Advisory Board 5

□ Brought on corporate marketing and branding Firm, 6 Degrees, to increase Bio - Path profile and outreach » Increase Bio - Path visibility in media » Develop partnerships with influential journalists » Strategic planning for conferences and investor presentations □ Development of drug product marketing plan □ Website upgrade with improved SEO and branding □ Increasing press releases as Bio - Path reaches milestones □ New composition of matter and process patent filed □ New tradename and trademark for DNAbilize™ technology and for Bio - Path Holdings logo Corporate Development 6

□ Bio - Path Holdings’ Data from Phase I and Safety Segment of Phase II Clinical Trials in Blood Cancers Presented at 57th American Society of Hematology (ASH) Annual Meeting □ Bio - Path Holdings Reports Third Quarter 2015 Operational and Financial Results □ Bio - Path Holdings’ Data to be presented at 57th American Society of Hematology (ASH) Annual Meeting □ Bio - Path Holdings to Present at the 14th Annual BIO Investor Forum □ Bio - Path Holdings Announces Successful Completion of Cohort 7 of Phase Ib of Clinical Trial Evaluating Liposomal Grb - 2 in Acute Myeloid Leukemia □ Bio - Path Holdings to Present at the 17th Annual Rodman & Renshaw Global Investment Conference □ Bio - Path Holdings Forms Scientific Advisory Board to Support Advancement of Liposomal Grb - 2 in Blood Cancers □ Bio - Path Holdings Reports Second Quarter 2015 Operational and Financial Results □ Bio - Path Holdings Announces "At the Market" Offering with Cantor Fitzgerald □ Bio - Path Holdings Reports First Quarter 2015 Operational and Financial Results □ Bio - Path Holdings Appoints Tara Sadeghi as Director of Clinical Operations □ Bio - Path Holdings Granted Orphan Drug Designation for Liposomal Grb - 2 in Acute Myeloid Leukemia □ Bio - Path Holdings' Drug Delivery Technology to be featured in a Presentation at IBC's 17th Annual TIDES Oligonucleotide and Peptide Therapeutics Conference □ Bio - Path Holdings Reports Fiscal Year 2014 Operational and Financial Results □ Bio - Path Holdings Starts Phase II Study with Liposomal Grb - 2 in AML Patients □ Bio - Path Holdings' Liposomal Grb - 2 Featured in Peer - Reviewed Journal "Expert Opinion on Drug Delivery" □ Bio - Path Holdings to Present at the Biotech Showcase 2015 Conference in San Francisco, CA Press Releases for 2015 (17) 7

□ Biotech Showcase EDB Group - January □ The 18th Annual BIO CEO & Investor Conference – February □ 28 th Annual ROTH Conference - March □ TIDES - Oligonucleotides in Therapeutics conference - May □ BIO International Convention - June □ ASCO 2016 - June □ Rodman & Renshaw 18th Annual Healthcare Conference - Sept □ BIO Investor Forum - October □ Therapeutic Area Partnerships (TAP) - November □ ASH 2016 - December Conference Program 2016 8
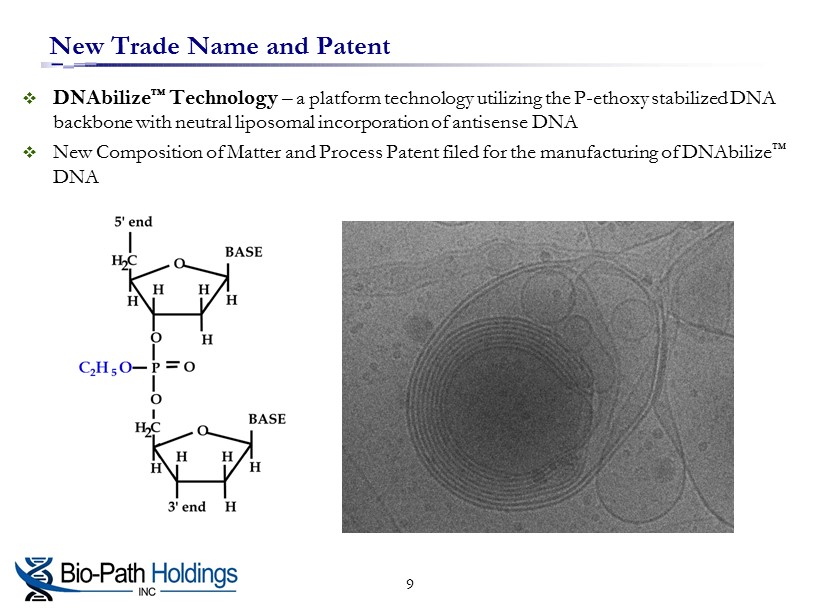
New Trade Name and Patent □ DNAbilize ™ Technology – a platform technology utilizing the P - ethoxy stabilized DNA backbone with neutral liposomal incorporation of antisense DNA □ New Composition of Matter and Process Patent filed for the manufacturing of DNAbilize ™ DNA 9

BP1001 in Clinical Testing □ Grb2 is a protein that bridges activated tyrosine kinases to the Ras signaling pathway □ Phase I clinical trial: » First 6 cohorts completed » P reliminary results show drug has been well tolerated in patients with AML, CML, and MDS with no signs of toxicity and signs of anti - leukemia activity □ Safety segment of P hase II (Phase Ib ) trial for Acute Myeloid Leukemia (AML) in combination with frontline therapy ongoing » Phase II efficacy trial to follow » Phase II trial in Chronic Myelogenous Leukemia (CML) planned □ Additional indications in triple negative and inflammatory breast cancers being developed and in other solid tumors 10
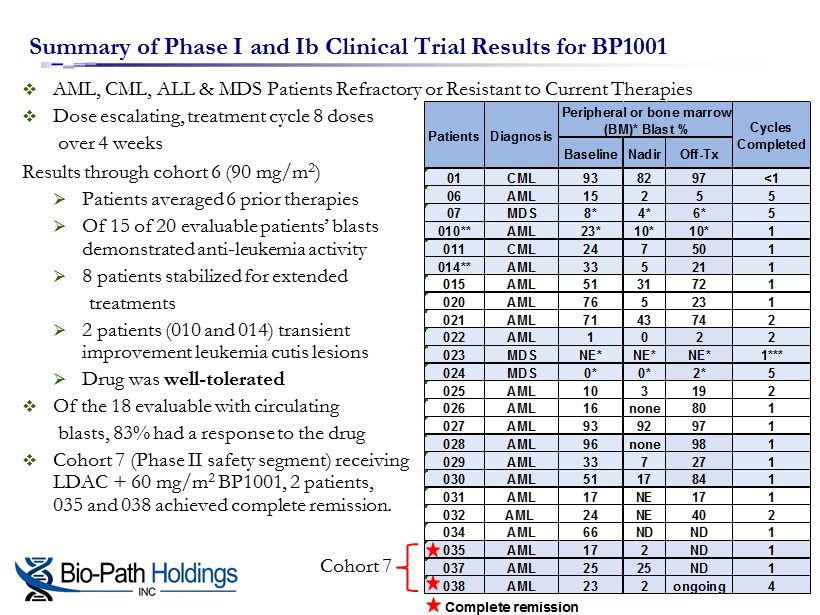
□ AML, CML, ALL & MDS Patients Refractory or Resistant to Current Therapies □ Dose escalating, treatment cycle 8 doses over 4 weeks Results through cohort 6 ( 90 mg/m 2 ) » Patients averaged 6 prior therapies » O f 15 of 20 evaluable patients’ blasts demonstrated anti - leukemia activity » 8 patients stabilized for extended treatments » 2 patients (010 and 014) transient improvement leukemia cutis lesions » D rug was well - tolerated □ Of the 18 evaluable with circulating blasts, 83% had a response to the drug □ Cohort 7 (Phase II safety segment) receiving LDAC + 60 mg/m 2 BP1001, 2 patients, 035 and 038 achieved complete remission . Summary of Phase I and Ib Clinical Trial Results for BP1001 Complete remission Baseline Nadir Off-Tx 01 CML 93 82 97 <1 06 AML 15 2 5 5 07 MDS 8* 4* 6* 5 010** AML 23* 10* 10* 1 011 CML 24 7 50 1 014** AML 33 5 21 1 015 AML 51 31 72 1 020 AML 76 5 23 1 021 AML 71 43 74 2 022 AML 1 0 2 2 023 MDS NE* NE* NE* 1*** 024 MDS 0* 0* 2* 5 025 AML 10 3 19 2 026 AML 16 none 80 1 027 AML 93 92 97 1 028 AML 96 none 98 1 029 AML 33 7 27 1 030 AML 51 17 84 1 031 AML 17 NE 17 1 032 AML 24 NE 40 2 034 AML 66 ND ND 1 035 AML 17 2 ND 1 037 AML 25 25 ND 1 038 AML 23 2 ongoing 4 Peripheral or bone marrow (BM)* Blast % Cycles Completed Patients Diagnosis Cohort 7
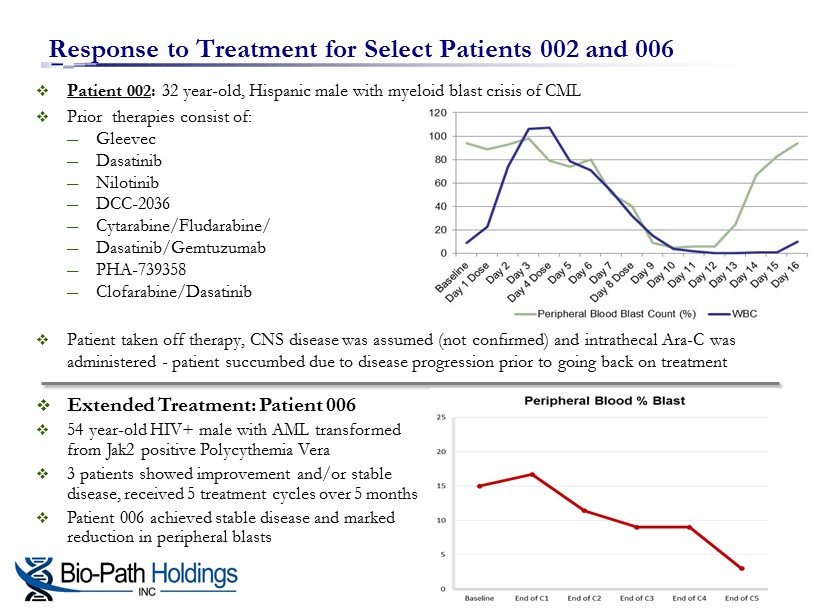
□ Patient 002 : 32 year - old, Hispanic male with myeloid blast crisis of CML □ Prior therapies consist of: ― Gleevec ― Dasatinib ― Nilotinib ― DCC - 2036 ― Cytarabine/Fludarabine/ ― Dasatinib/Gemtuzumab ― PHA - 739358 ― Clofarabine/Dasatinib □ Patient taken off therapy, CNS disease was assumed (not confirmed) and intrathecal Ara - C was administered - patient succumbed due to disease progression prior to going back on treatment Response to Treatment for Select Patients 002 and 006 □ Extended Treatment: Patient 006 □ 54 year - old HIV+ male with AML transformed from Jak2 positive Polycythemia Vera □ 3 patients showed improvement and/or stable disease, received 5 treatment cycles over 5 months □ Patient 006 achieved stable disease and marked reduction in peripheral blasts
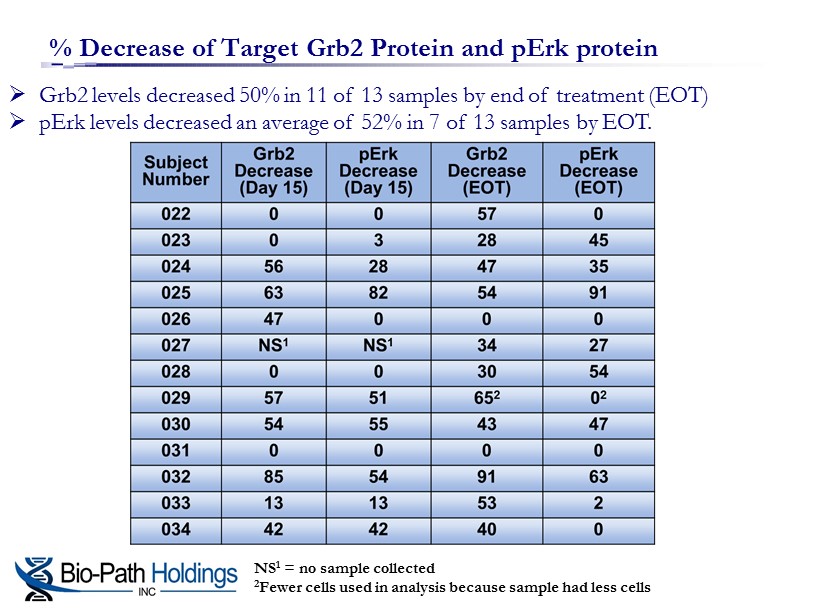
% Decrease of Target Grb2 Protein and pErk protein » Grb2 levels decreased 50% in 11 of 13 samples by end of treatment (EOT) » pErk levels decreased an average of 52% in 7 of 13 samples by EOT. NS 1 = no sample collected 2 Fewer cells used in analysis because sample had less cells

Results of Phase II Cohort 7 Combination LDAC + BP1001 Therapy □ S afety evaluation of combination l ow - dose cytarabine (LDAC) with BP1001 in refractory and relapsed patients. □ Three (3) evaluable patients were treated twice a week for 4 weeks with 60 mg/m 2 of BP1001 and LDAC □ T otal of 8 doses in combination with the standard regimen of LDAC. » Two out of three patients achieved complete remission □ Cohort 8 patients being treated with 90 mg/m 2 of BP1001 in combination with frontline LDAC is ongoing. Results were consistent with previous cohorts, showing BP1001 to be safe, well tolerated with significant anti - leukemia activity 14
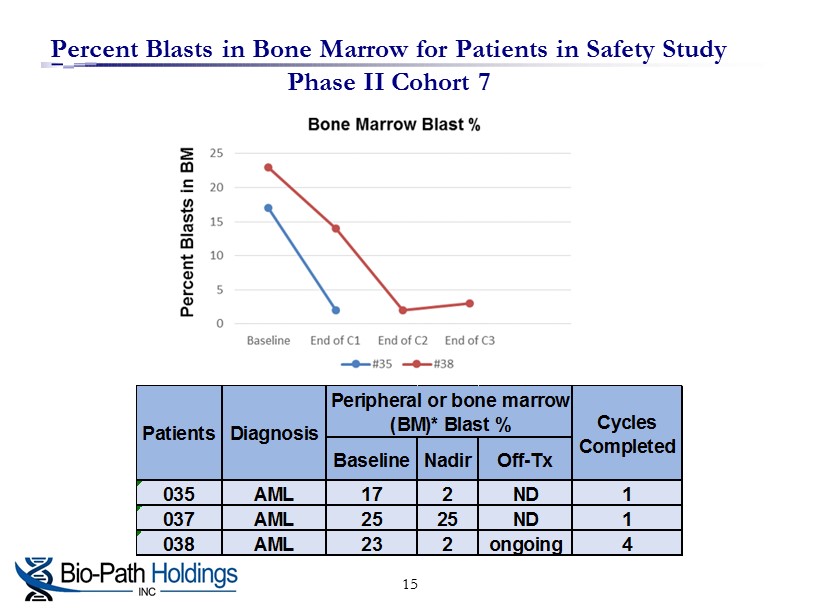
Percent Blasts in Bone Marrow for Patients in Safety Study Phase II Cohort 7 15 Baseline Nadir Off-Tx 035 AML 17 2 ND 1 037 AML 25 25 ND 1 038 AML 23 2 ongoing 4 Peripheral or bone marrow (BM)* Blast % Cycles Completed Patients Diagnosis

Safety , Pharmacokinetics, and Efficacy Of BP - 100 - 1 . 01 (Liposomal Grb - 2 Antisense Oligonucleotide) In Patients With Refractory Or Relapsed Acute Myeloid Leukemia (AML), Philadelphia Chromosome Positive Chronic Myelogenous Leukemia (CML), Acute Lymphoblastic Leukemia (ALL), and Myelodysplastic Syndrome (MDS ) Maro Ohanian, DO 1* , Hagop M. Kantarjian , MD 2 , Farhad Ravandi , MD 1 , Gautam Borthakur , MD 3 , Guillermo Garcia - Manero , MD 1 , Michael Andreeff , MD, PhD 4 , Elias Jabbour , MD 1 , Marina Konopleva , MD, PhD 1* , Miranda Lim , Sherry Pierce, Susan O'Brien, MD 1 , Alfonso Quintas - Cardama , MD 1,3* , Bradley G. Somer , MD 5* , Ana Tari, PhD 6* , Srdan Verstovsek , MD, PhD 7 , William G. Wierda , MD 8* and Jorge Cortes, MD 1 Highlights: • Patient 035, an 86 - year old female patient with AML, achieved CR after 1 cycle of BP - 100 - 1.01 and Ara - C drug combination. • Patient 038, a 69 - year old male patient with AML, achieved CR after 2 cycles of BP - 100 - 1.01 and Ara - C drug combination. ASH 2015 Presentation 16

Plans For Phase II Clinical Trials of BP1001 □ The proposed clinical program is to evaluate BP1001 in AML first in a Phase II clinical trial in combination with frontline therapy □ The current safety segment of the Phase II (Phase Ib) trial is evaluating 2 cohorts at 2 dose levels, 3 patients per cohort, to test for any potential negative synergies of using BP1001 together with frontline therapy □ Phase II efficacy trial: » Planned to have approximately 55 patients with an interim analysis after 19 patients » If successful, the trial will be rolled into a pivotal trial for accelerated approval » Expected to be conducted at leading cancer centers in the US, including the MD Anderson Cancer Center » Primary endpoint for the study is the number of patients who achieve complete remission ▪ Includes CRi (incomplete blood count recovery) and CRp (platelet recovery) □ Phase II trial in CML expected to start in early 2016 17

Accomplishments 2015 and Future Development for 2016 □ Significant corporate development in 2015: » Phase II safety segment (Phase Ib ) dosing for the combination therapy for BP1001 near completion » IND package for BP1002 (Liposomal Bcl2) in preparation to begin a clinical trial » Promising new targets in preclinical development (lymphoma, pancreatic, brain) » Expanding pipeline with new target drug candidates, new drug indication outside of cancer » BP1001 being developed for triple negative and inflammatory breast cancers » $25 Million ATM financing in place □ Value propositions for 2016: » Enrollment of first 19 patients in Phase II with an interim analysis to be completed with potential for switch to registration trial for accelerated approval » Initiation of safety segment for Phase II (Phase Ib ) in CML for combination therapy » Expanding pipeline and collaborations on new and creative drug candidates » Continued new manufacturing and target IP 18
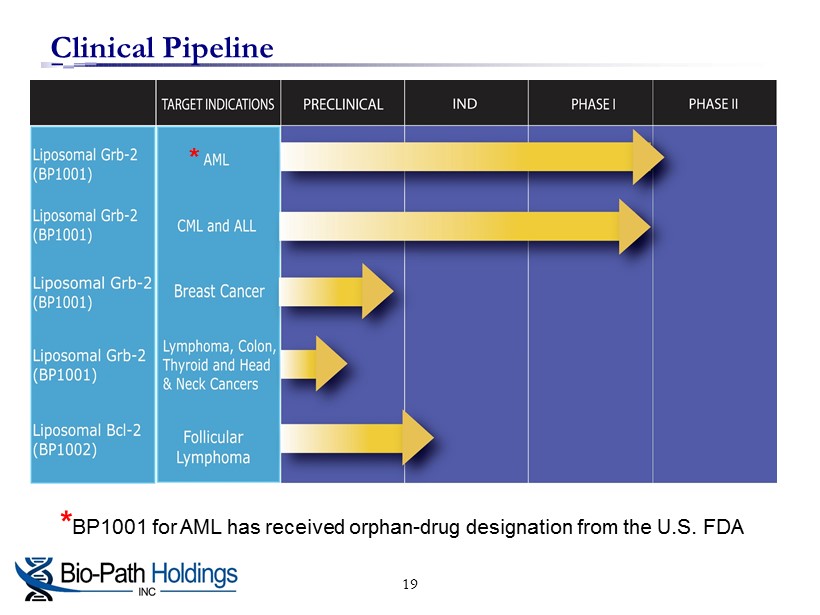
Clinical Pipeline * BP1001 for AML has received orphan - drug designation from the U.S. FDA * 19

Ticker: NASDAQ: BPTH Shares: 89.8M shares outstanding Sept 30, 2015 Market Cap: approximately $145 MM Capital Raised: $30.6 MM Cash: $9.9 MM as of Sept 30, 2015 Burn rate: approximately $1,000,000 per quarter excluding the clinical trial costs Financing: $25 million ATM program in place Financial Snapshot 20